Abstract
Background and study aims Novel topical hemostatic agents have shown promising results in treating patients with non-variceal upper gastrointestinal bleeding (NVUGIB). However, data are limited even in published meta-analyses as to their role, especially compared to conventional endoscopic approaches. The aim of this study was to perform a highly comprehensive systematic review assessing the effectiveness of topical hemostatic agents in UGIB in different clinical settings.
Methods We performed a literature search of OVID MEDLINE, EMBASE, and ISI Web of Knowledge databases through September 2021. Studies assessing the efficacy of topical hemostatic agents in UGIB were included. Main outcomes were immediate hemostasis and overall rebleeding.
Results A total of 980 citations were identified and 59 studies with a total of 3,417 patients were included in the analysis. Immediate hemostasis was achieved in 93 % (91 %; 94 %), with similar results according to etiology (NVUGIB vs. variceal), topical agent used, or treatment strategy (primary vs. rescue). The overall rebleeding rate was 18 % (15%; 21 %) with the majority of rebleeds occurring in the first 7 days. Among comparative studies, topical agents achieved immediate hemostasis more often than standard endoscopic modalities (OR 3.94 [1.73; 8.96), with non-different overall rebleeding odds (OR 1.06 [0.65; 1.74]). Adverse events occurred in 2 % (1 %; 3 %). Study quality was overall low to very low.
Conclusions Topical hemostatic agents are effective and safe in the management of UGIB with favorable outcomes when compared to conventional endoscopic modalities across a variety of bleeding etiologies. This is especially true in novel subgroup analyses that assessed immediate hemostasis and rebleeding among RCTs and in malignant bleeding. Due to methodological limitations of available data, additional studies are needed to ascertain their effectiveness more confidently in the management of patients with UGIB.
Introduction
Upper gastrointestinal bleeding (UGIB) is a common emergency condition associated with significant morbidity and mortality 1 . In the United States, UGIB results in more than 300,000 hospital admissions annually with significant associated costs 2 3 . Despite recent advances in the endoscopic management of both non-variceal upper gastrointestinal bleed (NVUGIB) and variceal upper gastrointestinal bleed, some patients fail conventional endoscopic therapy. Indeed, approximately 15 % of patients treated for UGIB fail primary endoscopic therapy, and re-bleeding can occur in up to 25 % of patients after initial successful therapy 4 with associated increased morbidity and mortality, especially in patients with variceal bleeding 5 .
Recently, novel topical endoscopic hemostatic agents were introduced for the management of UGIB 6 . These agents have shown promising results as a primary or salvage therapy while requiring less technical expertise 6 . Such agents include, among others the hemostatic powder TC-325 (Hemospray, Cook Medical, Bloomington, Indiana, United State), the starch derived polysaccharide hemostatic system (EndoClot, Endoclot Plus Inc., Santa Clara, California, United States), the biocompatible natural polymer UI-EWD (Nexpowder, NextBiomedical Co, Incheon, South Korea) and more recently the synthetic self-assembling peptide agent (PuraStat, 3D-Matrix, Europe Ltd., France) 7 8 ( Table 1 ). Previous studies have concluded that these hemostatic agents are effective and safe in the treatment of NVUGIB with low adverse event (AE) rates 9 10 11 12 . However, reported re-bleeding rates have been high (19 % at 72 hours) when used to treat NVUGIB 7 . As a result, the international guidelines for the management of ulcer bleeding suggested the hemostatic powder Tc-325 be used as temporizing measure when conventional endoscopic therapy fails but suggested against using it as a monotherapy 13 . In contradistinction, the more recent American College of Gastroenterology guidelines recently endorsed such a use 14 , bolstered by a recently published randomized trial exhibiting potentially limited external validity 15 16 . Unfortunately, preventing an adequate characterization of overall effectiveness, many of the published studies and systematic reviews did not completely assess the role of Tc-325 according to bleeding etiology which is thought to be a critical consideration when opting for such therapy. 16 Moreover, the published meta-analyses also failed to address other topical hemostatic agents in the management of UGIB or in certain situations such as variceal bleeding, even if data addressing these are sparse.
Table 1. Summary of included topical hemostatic agents.
| Agent | Trade Name | Composition | Mechanism of action | Approved human application | Formulation |
| TC-325 | Hemospray (Cook Medical, Winston-Salem, North Carolina, USA) |
Granular mineral-based |
Absorbs H
2
O
Forms mechanical tamponade Activates clotting cascade |
Nonvariceal gastrointestinal bleed | CO 2 pressurized handler canister (20 g) |
| EndClot | EndoClot (Endoclot Plus Inc., Santa Clara, California, United States) |
Absorbable starch-based modified polysaccharide |
Absorbs H
2
O and concentrates cells
Activates clotting cascade |
Upper and lower gastrointestinal bleeding | Pressurized air compressor |
| PuraStat | PuraStat (3D-Matrix, Europe Ltd., France) |
Synthetic self-assembling peptide agent | Forms a gel coat that induces hemostasis Promotes healing |
Bleeding from capillaries and oozing from capillaries of the gastrointestinal tract Prevention of delayed bleeding post colonic ESD |
Prefilled syringe |
| UI-EWD | Nexpowder (NextBiomedical Co, Incheon, South Korea) |
Biocompatible natural polymer | Forms adhesive hydrogel in the presence of water | Intended for upper gastrointestinal bleeding | Spraying device |
| CEGP-003 | CEGP-003 (CGBio, Seongnam, South Korea) |
Absorbable and adhesive macromolecules containing epidermal growth factor (EGF) | Forms adhesive gel upon contact with moist mucosa facilitating hemostasis Promotes wound healing |
Treatment of upper gastrointestinal bleeding Prevention of bleeding post EMR/ESD |
Spraying device |
ESD, endoscopic submucosal dissection; EMR, endoscopic mucosal resection.
The aim of this systematic review and meta-analysis is thus to more completely characterize the effectiveness and safety of hemostatic agents in achieving and maintaining hemostasis when managing patients with varying etiologies of UGIB in order to better and more comprehensively guide clinician decisions and future societal recommendations.
Methods
Search strategy
A comprehensive literature search was performed, from the recorded start of databases to September 2021 using OVID MEDLINE, EMBASE, and ISI Web of Knowledge databases, with a combination of MeSH term and controlled vocabulary to identify studies related to: 1) hemostatic agent or powder and; 2) gastrointestinal bleeding (Supplementary Table 1). Abstracts presented at major gastroenterology conferences (ACG, CDDW, DDW, UEGW, APDW) in the past 5 years were also hand-searched. Additional relevant studies were identified from cross-referencing and hand-searches of references of retrieved articles.
Validity assessment, data abstraction and rating of evidence
Two reviewers (AA, MM) evaluated the eligibility of all identified citations independently, with a third (AB) resolving disagreements. Study quality was assessed using the Cochrane Risk of bias tool for randomized trials 17 , and the Ottawa-Newcastle criteria for observational studies 18 . We used the GRADE rating to characterize the certainty of evidence 19 .
Inclusion and exclusion criteria
Studies assessing any hemostatic agent in UGIB of any etiology were considered for inclusion. Both manuscripts and abstracts from major gastroenterology societies (limited to the previous 5 years) were considered for inclusion. Only studies published in English were included. We excluded case reports, studies with less than 10 patients, review articles, and non-human studies. In addition, we excluded studies reporting on the hemostatic agent “Ankaferd” due to its limited availability globally 6 . Studies that used hemostatic agents prophylactically to prevent GI bleeding were also excluded.
Study definitions
“Immediate hemostasis” was defined as no further bleeding at least 3 minutes after application of the hemostatic agent. “Rebleeding” was defined as evidence of recurrent UGIB manifested as overt gastrointestinal bleeding or drop in hemoglobin ≥ 2 g/dL after achieving immediate hemostasis 20 . Rebleeding data were included only for patients following initial immediate hemostasis. “Technical success” was defined as successful use of the hemostatic agent during endoscopy without any technical problems (e. g. blockage of applications catheter). “Monotherapy” was defined as the use of a hemostatic agent alone, whereas “combination therapy” was defined as using the hemostatic agent in combination with adjunctive conventional endoscopic methods. “Rescue therapy” was defined as the use of a hemostatic agent when other conventional endoscopic methods had failed as evidenced by failure of immediate hemostasis. “Primary therapy” was defined as using the hemostatic agent as first-line endoscopic therapy for bleeding.
Outcomes
Immediate hemostasis and overall rebleeding (defined as any rebleeding during follow-up after the index endoscopy) were the outcomes of interest. Other outcomes assessed included 7-day and 30-day rebleeding, overall-mortality, bleeding-related mortality, technical success, and AEs related to application of the topical hemostatic agent. Data will be present in turn for a meta-analysis of proportions (purely descriptive) and in a meta-analysis assessing studies that compared hemostatic powders to a control therapeutic approach.
Sensitivity and subgroup analyses
Pre-planned possible subgroup and sensitivity analyses included assessments according to: type of hemostatic agent, type of therapies (primary therapy, rescue therapy, monotherapy, combination therapy), type of lesions (peptic ulcer disease (PUD), post endoscopic intervention, varices, malignant lesions), randomized-controlled trials alone; fully published articles alone, higher quality studies, year of publication, continent where the study was performed, performing a fixed rather than a random effect model (when appropriate), and when correcting for double-zero events.
Statistical analysis
Categorical estimates of outcomes were reported as proportions and 95 % confidence intervals (CI) using weighted random effects models. Continuous variables are reported as means and standard deviations medians were used if means were not available and standard deviations (SDs) were calculated or imputed when possible. For comparative studies, effect size was calculated with weighted mean differences (WMDs) for continuous variables. Odds ratios (ORs) are expressed for categorical variables. The DerSimonian and Laird method for random effect models was applied to all outcomes to determine corresponding overall effect sizes and their confidence intervals. Sensitivity analyses were performed using the Mantel-Haenszel method with random effect models; however fixed effects models were used when no statistical heterogeneity was noted. WMD were handled as continuous variables using the inverse variance approach. Presence of heterogeneity across studies was defined using a Chi-square test of homogeneity with a 0.10 significance level. The Higgins I 2 statistic was calculated to quantify the proportion of variation in treatment effects attributable to between-study heterogeneity, with values of 25 %, 50 %, and 75% representing low, moderate, and high heterogeneity, respectively. When heterogeneity was noted, prediction intervals were calculated and added to the forest plot. The prediction interval calculates the 95 % of where the effect size will be if a new study is randomly added to the meta-analysis. In order to ensure that zero event trials did not significantly affect the heterogeneity or p-values, sensitivity analyses were performed where a continuity correction was added to each trial with zero events using the reciprocal of the opposite treatment arm size. For all comparisons. Publication bias was evaluated using funnel plots if at least three citations were identified. All statistical analyses were done using Revman 5.4 and Meta package in R version 2.13.0, (R Foundation for Statistical Computing, Vienna, Austria, 2008).
Results
Included studies, quality assessment and publication bias
We initially identified 980 citations. After review of abstracts, a total of 59 studies were included ( Fig. 1 ). One study was excluded since it reported results as per episode of bleeding rather than per patient outcomes 21 .
Fig. 1.
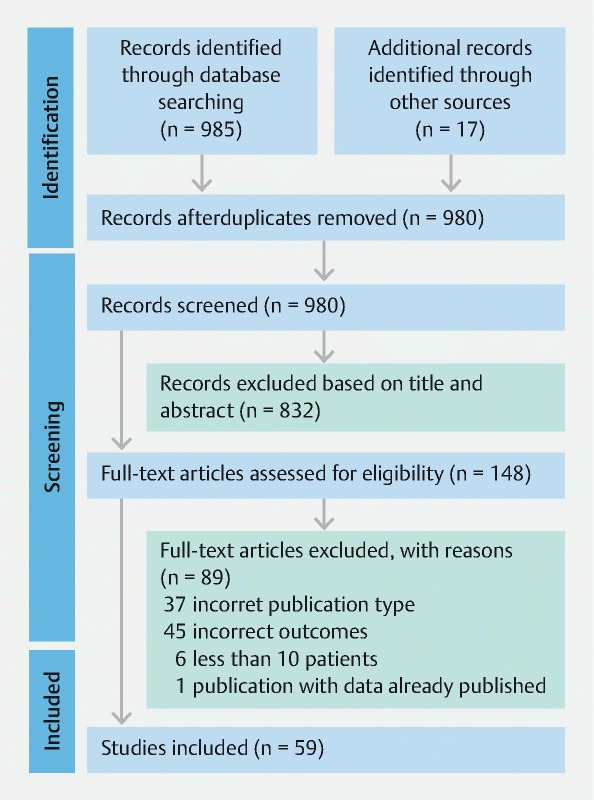
PRISMA diagram of included studies. From: Page MJ, McKenzie JE, Bossuyt PM et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021; 372:n71.
For the outcomes in the (solely descriptive) meta-analysis of proportions, moderate to high heterogeneity was present for immediate hemostasis, rebleeding (overall, 7 days and 30 days) and overall mortality. No significant heterogeneity was noted in the portion of the meta-analysis addressing between-group comparative results. No publication bias was observed (data available upon request).
Study quality was overall low-very low with the Ottawa Newcastle Scores (NOS) ranging from 4 to 8 stars out of a possible score of 9, with a mean of 5.9 ± 0.8 for observational studies. Assessing the individual domains of the NOS confirmed the low studies’ quality (Supplementary Table 2). The Cochrane risk bias tool revealed a high potential for performance bias across studies for randomized trials since all were single-blinded (Supplementary Fig. 1). The grading of the evidence was performed in studies that included a comparative arm and was found to be “very low” for all outcomes (Supplementary Table 3).
Patient and study characteristics
Overall, 59 studies were included (n = 3417 patients). Hemospray (TC-325) was the sole intervention in 44 studies 15 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 , EndoClot in five 65 66 67 68 69 , PuraStat in four 70 71 72 73 , Nexpowder in three 74 75 76 and CEGP-003 77 in one. In addition, two studies included both Hemospray and EndoClot 78 79 . Seven were RCTs, and 15 were prospective while 37 were retrospective cohort studies. Furthermore, 44 were fully published articles, while the remainder were sole abstracts ( Table 2 ). Ten studies included a comparison with standard endoscopic therapy 15 24 30 39 42 47 52 65 69 77 . Reports recruited patients between 2009 and 2021. Most of the bleeding lesions were classified as oozing bleeding (Forrest Ib). There were 11 studies 26 27 28 37 38 49 50 57 60 64 78 (n = 124 patients) that addressed patients with variceal bleeding. Full study and patient characteristics are shown in Table 2 .
Table 2. Patient and study characteristics.
|
Study
Country |
Intervention | Comparison | Sample size | Design | Study period | Indication | Etiology | Bleeding severity |
|
Arena et al. 2017
23
Italy |
Hemospray | None | 15 | Retrospective cohort | 2014–2015 | Primary therapy | Malignancy (100 %) | Oozing (100 %) |
|
Appleby et al. 2017
22
UK Abstract |
Hemospray | None | 32 | Retrospective cohort | 2014–2017 | Primary therapy Rescue therapy |
PUD (56 %) Malignancy (9 %) Dieulafoy lesion (28 %) Others (7 %) |
NR |
|
Baracat et al. 2020
24
Brazil |
Hemospray | Hemoclips | 39 | RCT | 2015–2017 | Primary therapy | PUD (43.6 %) Malignancy (12.8 %) Post intervention (10.3 %) Dieulafoy lesion (5.1 %) MalloryWeiss tear (7.7 %) Others (20.5 %) |
Spurting (10.3 %) Oozing (89.7 %) |
|
Bestari et al. 2020
26
Indonesia |
Hemospray | None | 30 | Retrospective cohort | 2016–2019 | Primary therapy Rescue therapy |
PUD (56.7 %) Malignancy (26.7 %) Variceal bleed (10 %) Portal hypertensive gastropathy (6.7 %) |
NR |
|
Becq et al. 2020
25
France |
Hemospray | None | 152 | Retrospective cohort | 2015–2018 | Primary therapy Rescue therapy |
PUD (47.7 %) Malignancy (22.2 %) Esophagitis (12.4 %) Other (17.7 %) |
Oozing (84.3 %) spurting (11.1 %) None active (2.6 %) Undetermined (2.0 %) |
|
Chahal et al. 2020
28
Canada |
Hemospray | None | 73 | Retrospective cohort | 2014–2018 | Primary therapy Rescue therapy |
PUD (67.1 %) Malignancy (5.5 %) Variceal bleed (5.5 %) MWT (8.2 %) Other (13.7 %) |
For PUD (n = 49) Forrest Ia (14.3 %) Forrest Ib (53.1 %) Forrest IIa (20.4 %) Forrest IIb (14.3 %) Forrest III (4.1 %) |
|
Cahyadi et al. 2017
27
Germany |
Hemospray | None | 52 | Retrospective cohort | 2013–2017 | Primary therapy Rescue therapy |
PUD (34.6 %) Malignancy (19.2 %) Post procedure (30.7 %) Dieulafoy lesion (1.9 %) GAVE (1.9 %) Variceal bleed (3.8 %) Other (7.6 %) |
Peptic ulcer only, n = 18 Forrest Ib (38.9 %) Forrest IIa (33.3 %) Forrest IIb (22.2 %) Forrest IIc (5.6 %) |
|
Chen et al. 2015
29
Canada |
Hemospray | None | 56 | Retrospective cohort | 2011–2013 | Primary therapy Rescue therapy |
Benign NVUGIB (37.5 %) Malignancy (33.9 %) Post intervention (28.6 %) |
For PUD (n = 13) Forrest Ia (23.1 %) Forrest Ib (76.9 %) |
|
Chen et al. 2020
30
Canada |
Hemospray | CHP | 17 | RCT | 2014–2016 | Primary therapy | Malignancy (100 %) | Spurting (5 %) Oozing (95 %) |
|
De Santiago et al. 2019
60
Spain |
Hemospray | None | 219 | Retrospective cohort | 2011–2018 | Primary therapy Rescue therapy |
PUD (33.3 %) Malignancy (21.0 %) Post-procedure (14.2 %) postsurgical (6.4 %) Dieulafoy lesion (3.7 %) Variceal bleed (3.2 %) GAVE (0.9 %) Others (17.4 %) |
Peptic ulcer only, n = 73 Forrest Ia (24.7 %) Forrest Ib (64.4 %) Forrest IIa (5.5 %) Forrest IIb (5.5 %) |
|
Disney et al. 2015
31
UK Abstract |
Hemospray | None | 19 | Retrospective cohort | NR | Primary therapy Rescue therapy |
Malignancy (100 %) | Oozing (100 %) |
|
Giles et al. 2016
32
New Zeland |
Hemospray | None | 36 | Retrospective cohort | 2013–2016 | Rescue Therapy | PUD (66.7 %) Malignant (5.6 %) post intervention (16.7 %) Others (11.0 %) |
Spurting (13.9 %) Oozing (69.4 %) Visible vessel (5.6 %) Adherent clot (5.6 %) Clean base (5.6 %) |
|
Gupta et al. 2018
33
UK Abstract |
Hemospray | None | 45 | Retrospective cohort | 2013–2017 | Primary therapy Rescue therapy |
PUD (41 %) Malignancy (15 %) Post-intervention (15 %) Other (29 %) |
Forrest Ia (15 %) Forrest Ib (46 %) Forrest IIa (15 %) Forrest IIb (15 %) Forrest IIc (9 %) |
|
Haddara et al. 2016
34
France |
Hemospray | None | 202 | Retrospective cohort | 2013–2015 | Primary therapy Rescue therapy |
PUD (37.1 %) Malignant (30.2 %) Post-intervention (17.3 %) Dieulafoy lesion (1.5 %) Others (3.9 %) |
For PUD (n = 75) Forrest Ia (20.0 %) Forrest Ib (57.3 %) Forrest II (18.7) Unclassified (4.0 %) |
|
Hagel et al. 2017
35
Germany |
Hemospray | None | 33 | Retrospective cohort | 2013–2014 | Rescue Therapy | PUD (48.5 %) Malignancy (12.1 %) Post intervention (12.1 %) Diffuse bleeding (18.2 %) Others (9.1 %) |
NR |
|
Holster et al. 2015
36
Netherland |
Hemospray | None | 16 | Retrospective cohort | 2011–2012 | Primary therapy Rescue therapy |
PUD (56.0 %) Malignant (13.0 %) Others (31.0 %) |
For PUD (n = 9) Forrest Ia (55.5 %) Forrest Ib (44.4 %) |
|
Hussein et al. 2021 (1)
62
UK |
Hemospray | None | 105 | Prospective study | 2016–2020 | Primary therapy Rescue therapy |
Malignancy (100 %) | Spurting (6 %) Oozing (77 %) Visible vessel/adherent clot (12 %) |
|
Hussein et al. 2021 (2)
61
UK |
Hemospray | None | 202 | Prospective cohort | 2016–2019 | Primary therapy Rescue therapy |
PUD (100 %) | Forrest Ia (19.0 %) Forrest Ib (58.0 %) Forrest IIa (12.0 %) Forrest IIb (10.0 %) |
|
Hussein et al. 2021 (3)
64
UK Abstract |
Hemospray | None | 12 | Prospective study | 2016–2019 | Primary therapy Rescue therapy |
Esophageal varices (83.3 %) Gastric varices (16.7 %) |
NR |
|
Hussein et al. 2020
63
UK |
Hemospray | None | 73 | Prospective cohort | 2016–2019 | Primary therapy Rescue therapy |
Post-procedure (100 %) | NR |
|
Ibrahim et al. 2015
37
Belgium |
Hemospray | None | 30 | Prospective cohort | 2013–2014 | Primary Therapy | Esophageal varices (83.4 %) Gastric varices (10 %) Duodenal varices (6.6 %) |
Spurting (43.4 %) Fibrin plug/red streaks (56.6 %) |
|
Ibrahim et al. 2019
38
Belgium |
Hemospray | Early elective endoscopy | 86 | RCT | 2014–2016 | Primary therapy | Variceal bleed (100 %) | Spurting (16.3 %) Blood in stomach (83.7 %) |
|
Kwek et al. 2017
39
Singapore |
Hemospray | Conventional endoscopic therapy | 20 | RCT | 2013–2015 | Primary therapy | PUD (100 %) | Forrest Ia (5.0 %) Forrest Ib (35.0 %) Forrest IIa (50.0) Forrest IIb (10.0 %) |
|
Lau et al. 2020
15
China |
Hemospray | Conventional endoscopic therapy | 224 | RCT | 2015–2018 | Primary therapy | PUD (58 %) Malignancy (14.3 %) Others (27.7 %) |
NR |
|
Leblanc et al. 2013
40
France |
Hemospray | None | 17 | Retrospective cohort | 2011–2012 | Primary therapy Rescue therapy |
Post intervention (70.6 %) Malignancy (29.4 %) |
For post-intervention (n = 12) pulsatile (8.3 %) oozing (91.7 %) |
|
Malik et al. 2015
41
UK Abstract |
Hemospray | None | 19 | Retrospective cohort | 2013–2014 | Primary therapy Rescue therapy |
PUD (53 %) Malignancy (21 %) Post intervention (21 %) Other (5 %) |
For PUD (n = 10) Forrest Ia (0 %) Forrest Ib (60 %) Forrest IIa (30 %) Forrest IIb (10 %) |
|
Martins et al. 2019
42
Brazil Abstract |
Hemospray | CHP | 36 | RCT | 2016–2017 | Primary therapy | Malignancy (100 %) | Active bleeding (63.9 %) |
|
Masci et al. 2014
43
Italy |
Hemospray | None | 13 | Prospective cohort | NR | Primary therapy | PUD (100 %) | Forrest Ia (35.7 %) Forrest Ib (64.3 %) |
|
Meng et al. 2018
44
Canada |
Hemospray | None | 25 | Retrospective study | 2010–2016 | Primary therapy Rescue therapy |
Malignancy (100 %) | Forrest Ia (8.0 %) Forrest Ib (76.0 %) unclassified (16.0 %) |
|
Min et al. 2018
45
UK Abstract |
Hemospray | None | 48 | Retrospective cohort | 2016–2017 | Primary therapy Rescue therapy |
PUD (69.0 %) Malignancy (2.0 %) Post- intervention (4.0 %) GAVE (4.0 %) Esophagitis (4.0 %) Others (16.0 %) |
NR |
|
Nasr et al. 2015
46
UK Abstract |
Hemospray | None | 26 | Retrospective cohort | 2013–2015 | Primary therapy Rescue therapy |
NVUGIB (65.4 %) Post-intervention (34.6 %) |
NR |
|
Paoluzi et al. 2021
79
Italy |
Hemospray EndoClot |
CHP | 108 | Retrospective cohort | 2017–2019 | Primary therapy Rescue therapy |
PUD (66.7 %) Malignancy (21.3 %) Post-intervention (6.4 %) Others (5.5 %) |
For PUD (n = 72) Forrest IA (20.8 %) Forrest IB (79.2 %) For malignancy (n = 23) oozing (100 %) |
|
Pittayanon et al. 2016
47
Thailand |
Hemospray | CHP | 20 | Prospective cohort | 2014–2015 | Primary therapy | Malignancy (100 %) | oozing blood (100 % |
|
Pittayanon et al. 2018
48
Canada |
Hemospray | None | 79 | Retrospective cohort | 2011–2016 | Primary therapy Rescue therapy |
Malignancy (100 %) | Adherent clot (4.5 %) Blood oozing (94.3 %) Blood spurting (1.1 %) |
|
Prentice et al. 2018
49
UK Abstract |
Hemospray | None | 47 | Retrospective cohort | 2014–2017 | Primary therapy Rescue therapy |
NVUGIB (78.7 %) Variceal bleed (21.3 %) |
NR |
|
Ramirez-Polo et al. 2019
50
Mexico |
Hemospray | None | 81 | Retrospective cohort | 2015–2017 | Primary therapy | PUD (17.3 %) Malignancy (43.2 %) post-procedure (14.8 %) Dieulafoy lesion (6.1 %) Variceal bleed (2.5 %) Postsurgical (2.5 %) Other (13.6 %) |
NR |
|
Shivaji et al. 2018
51
UK Abstract |
Hemospray | None | 45 | Retrospective cohort | 2013–2017 | Primary therapy Rescue therapy |
PUD (41.0 %) Malignancy (15.0 %) Post-intervention (15.0 %) Others (29.0 %) |
Peptic ulcer only, n = 22 Forrest Ia (15.0 %) Forrest Ib (46.0 %) Forrest IIa (15.0 %) Forrest IIb (15.0 %) Forrest IIc (9.0 %) |
|
Sinha et al. 2016
52
UK |
Hemospray | CHP | 40 | Retrospective cohort | 2013–2015 | Rescue therapy | PUD (65.0 %) Esophageal ulcer (20 %) Others (15.0 %) |
Forrest Ia (60.0 %) Forrest Ib (40.0 %) |
|
Smith et al. 2014
53
UK |
Hemospray | None | 63 | Prospective cohort | 2011 | Primary therapy Rescue therapy |
PUD (57.1 %) Malignancy (12.6 %) Post-procedure (15.9 %) Dieulafoy lesion (3.2 %) MWT (3.2 %) GAVE (3.2 %) Other (5.8 %) |
Peptic ulcer only, n = 30 Forrest Ia (36.7 %) Forrest Ib (53.3 %) Unclassified (10 %) |
|
Sulz et al. 2014
54
Switzerland |
Hemospray | None | 15 | Prospective cohort | 2013 | Primary therapy Rescue therapy |
PUD (25.0 %) Malignancy (18.8 %) Post-intervention (12.5 %) Others (43.8 %) |
Spurting (18.8 %) oozing (81.2 %) |
|
Sung et al. 2011
55
Hong Kong |
Hemospray | None | 20 | Prospective cohort | 2009–2010 | Primary Therapy | PUD (100 %) | Forrest Ia (5 %) Forrest Ib (95 %) |
|
Thayalasekaran et al. 2017
56
UK Abstract |
Hemospray | None | 44 | Retrospective cohort | 2014–2016 | Primary therapy Rescue therapy |
PUD (65.9 %) Malignancy (2.3 %) Variceal bleed (6.8 %) MWT (4.5 %) Post-intervention (12.3 %) Others (8.2 %) |
Forrest Ia (26.5 %) Forrest Ib (44.1 %) Forrest IIa (23.5 %) Forrest IIb (5.9 %) |
|
Vitali et al. 2019
78
Germany |
Hemospray | EndoClot | 127 | Prospective cohort | 2013–2017 | Primary therapy Rescue therapy |
PUD (38.6 %) Malignancy (11.8 %) Variceal bleed (10.2 %) Reflux esophagitis (9.4 %) Others (30.0 %) |
NR |
|
Weaver et al. 2019
57
USA Abstract |
Hemospray | None | 12 | Prospective cohort | 2018–2019 | Primary therapy Rescue therapy |
PUD (50.0 %) MWT (16.7 %) Variceal bleed (8.3 %) GAVE (8.3 %) Post-intervention (16.7 %) |
Peptic ulcer only, n = 6 Forrest Ia (50.0 %) Forrest Ib (50.0 %) |
|
Widlak et al. 2015
58
UK Abstract |
Hemospray | None | 48 | Retrospective cohort | 2013–2015 | Primary therapy Rescue therapy |
PUD (60.5 %) Other (27 %) Post banding ulcer (12.5 %) |
NR |
|
Yau et al. 2014
59
Canada |
Hemospray | None | 19 | Retrospective cohort | 2012–2013 | Primary therapy Rescue therapy |
PUD (63.2 %) Dieulafoy lesion (10.5 %) Mucosal erosion (5.3 %) Angiodysplasia (5.3 %) Post-intervention (10.6 %) Unknown (5.3 %) |
Spurting (21.1 %) oozing (57.9 %) No active bleeding (21.1 %) |
|
Branchi et al. 2021
72
Germany |
PuraStat | None | 78 | Prospective cohort | 2017–2018 | Primary therapy Rescue therapy |
PUD Post-intervention Malignancy Vascular lesions |
spurting (6 %) Oozing (69 %) Visible vessel (14 %) Adherent clot (5 %) Flat pigmented spot (5 %) |
|
de Nucci et al. 2020
70
Italy |
PuraStat | None | 41 | Retrospective cohort | 2017–2019 | Rescue therapy | PUD (46.3 %) Malignancy (7.3 %) Post-intervention (39.0 %) Other (7.4 %) |
Spurting (19.5 %) Oozing (80.5 %) |
|
Labianca et al. 2021
73
Italy Abstract |
PuraStat | None | 15 | Retrospective cohort | 2018–2020 | Primary therapy Rescue therapy |
PUD (86.7 %) Post-intervention (13.3 %) |
NR |
|
Subramaniam et al. 2019
71
UK |
PuraStat | None | 44 | Retrospective cohort | 2016–2017 | Primary therapy | Post-intervention (100 %) | NR |
|
Beg et al. 2015
65
UK |
EndoClot | CHP | 130 | Retrospective cohort | 2012–2014 | Rescue therapy | PUD (90 %) Esophageal ulcer (4.6 %) MWT (3.8 %) Other (1.6 %) |
NR |
|
Hagel et al. 2020
68
Germany |
EndoClot | None | 22 | Retrospective cohort | 2015–2020 | Primary therapy Rescue therapy |
PUD (40.9 %) Malignancy (40.9 %) Others (18.2 %) |
NR |
|
Kim et al. 2018
66
South Korea |
EndoClot | None | 12 | Retrospective cohort | 2016–2017 | Primary therapy | Malignancy (100 %) | Forrest Ib (100 %) |
|
Park et al. 2019
69
South Korea |
EndoClot | CHP | 176 | Retrospective cohort | 2012–2017 | Primary therapy Rescue therapy |
PUD (68.8 %) Malignancy (21.6 %) Post-intervention (5.7 %) Radiation gastritis (3.9 %) |
Forrest Ia (9.7 %) Forrest Ib (54.5 %) Forrest IIa (35.8 %) |
|
Prei et al. 2016
67
Germany |
EndClot | None | 58 | Prospective cohort | 2012–2014 | Primary therapy Rescue therapy |
PUD (46.6 %) Malignancy (17.2 %) Post-intervention (10.3 %) Esophagitis (10.3 %) Other (15.6 %) |
Forrest Ia (5.2 %) Forrest Ib (65.5 %) Forrest IIa (10.3 %) Forrest IIb (6.9 %) Forrest IIc (12.1 %) |
|
Park et al. 2019(1)
74
South Korea |
Nexpowder | None | 17 | Prospective cohort | 2016–2017 | Rescue therapy | PUD (29.4 %) Malignancy (23.5 %) Post-intervention (41.2 %) Other (5.9 %) |
Forrest Ia (11.8 %) Forrest Ib (88.2 %) |
|
Park et al 2019 (2)
75
South Korea Abstract |
Nexpowder | None | 56 | Retrospective cohort | NR | Primary therapy | PUD (14.3 %) Malignancy (1.8 %) Post-intervention (82.1 %) Other (1.8 %) |
NR |
|
Shin et al. 2021
76
South Korea |
Nexpowder | None | 41 | Retrospective cohort | 2016–2019 | Primary therapy Rescue therapy |
Malignancy (100 %) | Forrest Ia (7.3 %) Forrest Ib (92.7 %) |
|
Bang et al. 2018
77
South Korea |
CEGP-003 | Epinephrine injection | 72 | RCT | 2014–2015 | Primary therapy | PUD (22.2 %) Post-intervention (77.8 %) |
Forrest Ib (83.3 %) Forrest IIa (6.9 %) Forrest IIb (9.8 %) |
PUD, peptic ulcer disease; RCT, randomized controlled trial; CHP, conventional hemostatic procedures; MWT, Mallory-Weiss tearing; GAVE, gastric antral vascular ectasia; NVUGIB, non-variceal upper gastrointestinal bleeding.
Immediate hemostasis and overall rebleeding
Immediate hemostasis was reported in 59 studies (n = 2,919 patients) with a pooled success rate of 93 % (91 %; 94 %) ( Fig. 2 , Table 3 ). Overall rebleeding was included in 58 studies (n = 2,696) with a pooled overall rebleeding rate of 18 % (15 %; 21%) ( Fig. 3 , Table 3 ). Rebleeding rates at 7 and 30 days were 17 % (14 %; 20 %) and 21 % (17 %; 27 %), respectively ( Table 3 ).
Fig. 2.
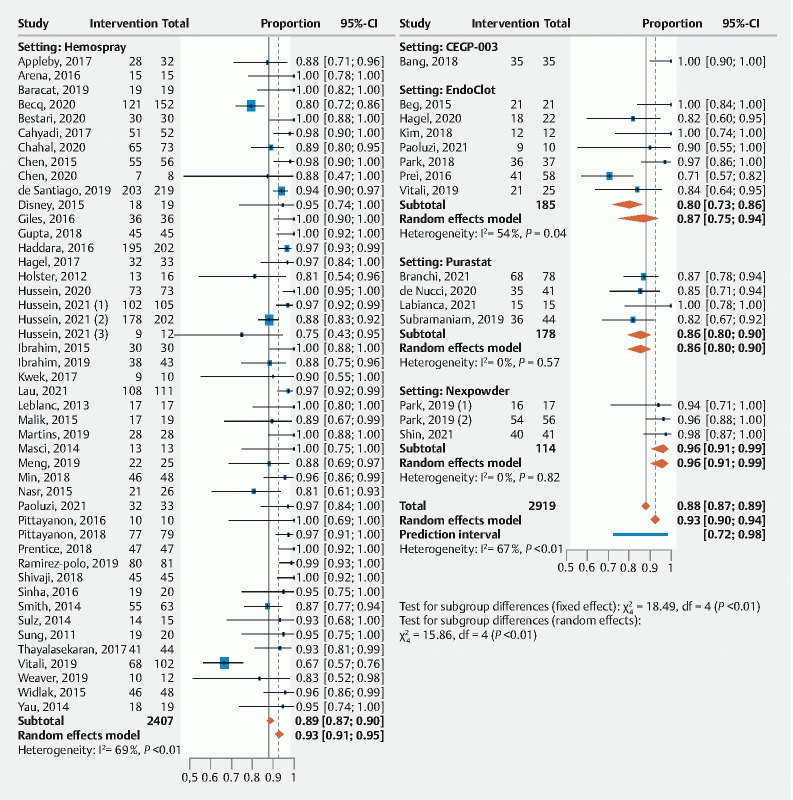
Forrest plot of primary outcome by proportion: immediate hemostasis.
Table 3. Primary and secondary outcome for proportions.
| No. studies | No. patients |
Proportion
(95 % CI) |
P
value
for heterogeneity |
I 2 | |
| Primary outcome | |||||
| Immediate hemostasis (overall UGIB) | 59 | 2919 | 0.93 (0.91; 0.94) | < 0.01 | 67 % |
| Overall rebleeding | 58 | 2696 | 0.18 (0.15; 0.21) | < 0.01 | 69 % |
| Rebleeding 7 days | 42 | 1943 | 0.17 (0.14; 0.20) | < 0.01 | 55 % |
| Rebleeding 30 days | 34 | 1692 | 0.21 (0.17; 0.26) | < 0.01 | 75 % |
| Secondary outcome | |||||
| Overall mortality | 45 | 2245 | 0.15 (0.12; 0.19) | < 0.01 | 64 % |
| Bleeding-related mortality | 34 | 1563 | 0.05 (0.04; 0.07) | 0.42 | 3 % |
| Technical success | 52 | 2392 | 0.97 (0.96; 0.98) | 0.99 | 0 % |
| Adverse events | 45 | 2111 | 0.02 (0.01; 0.03) | 0.99 | 0 % |
UGIB, upper gastrointestinal bleeding.
Fig. 3.
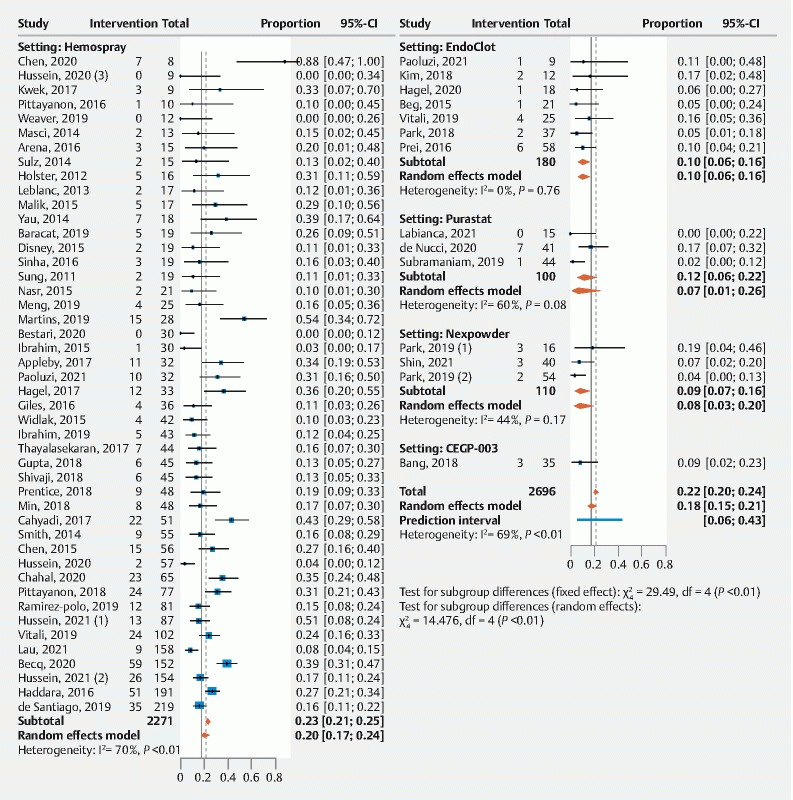
Forrest plot of primary outcome by proportion: overall rebleeding.
In the 10 studies comparing the topical hemostatic agent to a control intervention (n = 797), immediate hemostasis was more significantly achieved with the former (odds ratio [OR] = 3.94 (1.73; 8.96) ( Table 4 and Fig. 4 ). Among the comparative studies, the overall rebleeding risk was not significantly different between topical agents and conventional endoscopic therapy (OR = 1.06; 0.65–1.74, 10 studies, n = 775); this was also the case for rebleeding at 7 and 30 days (OR = 0.97; 0.43–2.16) and OR = 0.75; 0.39–1.45, respectively) ( Table 4 and Fig. 5 ).
Table 4. Primary and secondary outcome for comparative studies (comparing topical agent vs. conventional endoscopic therapy).
| No. studies | No. patients | Odds ratio (95 % CI) |
P
value
for heterogeneity |
I 2 | |
| Primary outcome | |||||
| Immediate hemostasis (overall UGIB) | 10 | 797 | 3.94 (1.73; 8.96) | 0.79 | 0 % |
| Overall rebleeding | 10 | 775 | 1.06 (0.65; 1.74) | 0.58 | 0 % |
| Rebleeding 7 days | 6 | 356 | 0.97 (0.43; 2.16) | 0.51 | 0 % |
| Rebleeding 30 days | 7 | 649 | 0.75 (0.39; 1.45) | 0.24 | 25 % |
| Secondary outcome (UGIB) | |||||
| Overall mortality | 9 | 621 | 1.05 (0.64; 1.70) | 0.79 | 0 % |
| Bleeding-related mortality | 5 | 347 | 0.62 (0.14; 2.71) | 0.45 | 0 % |
| Technical success | 9 1 | 573 | 0.30 (0.01; 8.33) | – | – |
| Adverse events | 5 2 | 185 | – | – | – |
Only one study with estimable data.
All were double zero-event.
Fig. 4.
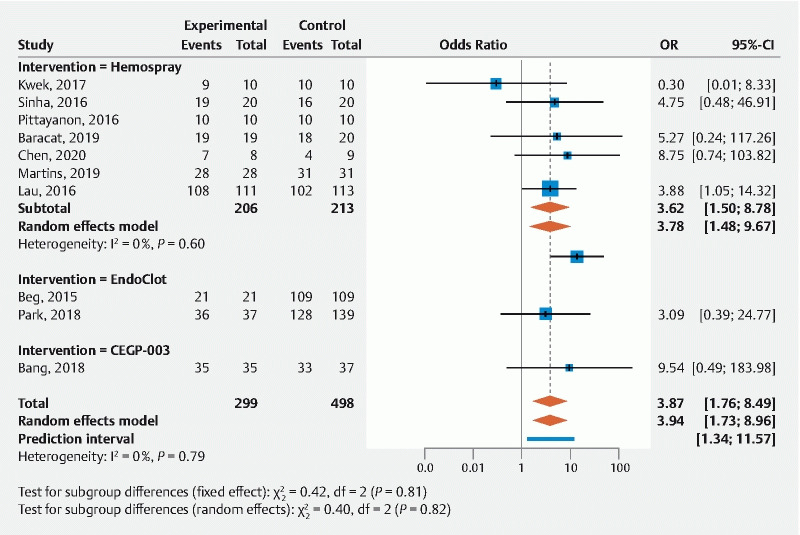
Forrest plot of primary outcome for comparative studies: immediate hemostasis.
Fig. 5.
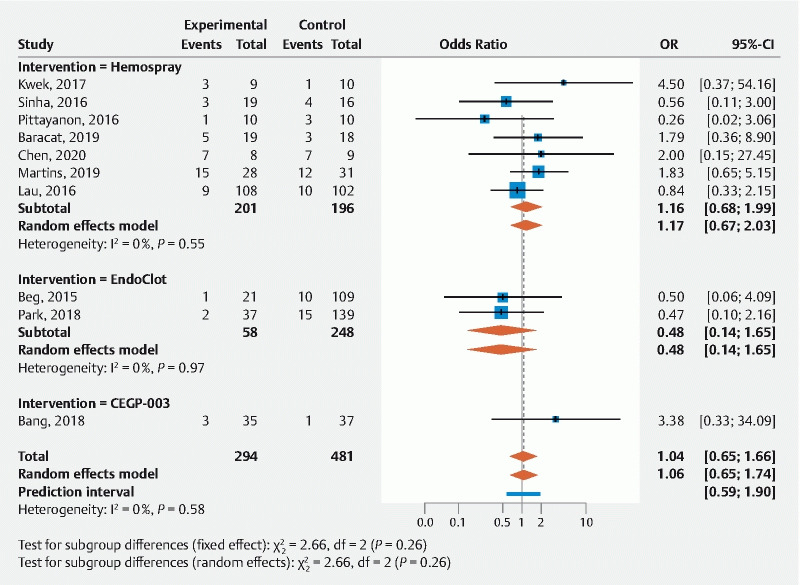
Forrest plot of primary outcome for comparative studies: overall rebleeding.
Other outcomes
Pooled proportions for overall mortality (45 studies, n = 2,245) and bleeding-related mortality (34 studies; n = 1,563) were 15% (12 %; 19 %) and 5 % (4 %; 7 %), respectively. In terms of technical success (52 studies, n = 2,392) and AEs (45 studies, n = 2,111), analyses yielded pooled proportions of 97 % (96 %; 98 %) and 2% (1 %; 3 %), respectively.
In studies that included a comparison group, none of these outcomes differed significantly when comparing the topical agent to the control hemostatic modality(ies) (table 4).
Sensitivity and subgroup analyses
Results for sensitivity analyses for immediate hemostasis and rebleeding are described in Appendix 1–4. Results confirm the robustness of the findings described in the primary analysis.
When analyzed according to etiology of UGIB, immediate hemostasis rates were high for all etiologies of UGIB including PUD (91 % [88 %; 93 %]) malignancy (94 % [91 %; 95 %]), post-endoscopic intervention (90 % [85 %; 93 %]), and a variceal etiology of bleeding (87 % [79 %; 92 %]). The risk of rebleeding for NVUGIB was 22 % (18 %; 27 %), for variceal bleeding was 23 % (10 %; 45 %), and for malignant bleeding, in particular, was 24% (19 %; 30 %) (Appendix 2).
When grouped according to indication, immediate hemostasis rates were similar when these agents were used as primary or rescue therapy (93 %, [89 %; 95 %] and 90 %, [85 %; 93%], respectively); these rates varied between 86 % to 99 % according to the topical agent assessed. The risk of rebleeding was 25 % (20 %; 30 %) when the topical agent was used as rescue therapy, and 18 % (14 %; 22 %) when used as primary therapy.
When studied according to topical agent studied, immediate hemostasis among the different agents was (Hemospray 93 % [91 %; 95 %]), EndoClot (87 % [75 %; 94 %]), PuraStat (86 % [80 %; 90 %]) Nexpowder (96 % [91 %; 99 %]) and CEGP-003 (99% [81 %; 100 %]).The risk of rebleeding was Hemospray 20 % (17 %; 24 %), EndoClot (10 % [6 %; 16 %]), PuraStat (7 % [1 %; 27%]) Nexpowder (8 % [3 %; 20 %]) and CEGP-003 (9 % [3 %; 23%]).
The subgroup analysis of sole RCTs yielded similar outcomes with a proportion of immediate hemostasis of 94 % (89 %; 97 %), and a rebleeding rate of 26 % (11 %; 48 %). Among Sensitivity analysis using comparative RCTs data confirmed that immediate hemostasis was more frequently achieved with topical hemostatic agents compared to conventional endoscopic therapy (3.62 [1.29; 10.11]), with similar odds of rebleeding (1.27 [0.62;2.59]). Topical agents were more effective when used as primary therapy (3.83 [1.59,9.24]) and when used for malignant lesions (14.74 [2.16,100.61]) (Appendix 3 and 4).
Discussion
To our knowledge, this is the largest, most granular and up-to-date meta-analysis that has characterized the efficacy and safety of different topical hemostatic agents according to various UGIB etiologies. Results confirm the effectiveness and safety of topical hemostatic agents when used to treat UGIB, however with a low to very low certainty of evidence for the data that these observations are based on, as is also the case for argon plasma coagulation and soft monopolar electrocoagulation for which there is also less published evidence 14 .
Overall, topical hemostatic agents achieve high immediate hemostasis rates (93 %, (91 %; 94 %)). This conclusion is true regardless of the cause of UGIB, including peptic ulcer disease, malignancy, post-endoscopic intervention, and even variceal bleeding. These impressive results remain robust whether the agent is used for primary therapy or rescue therapy. Furthermore, these results are comparable to published effectiveness for other conventional endoscopic modalities such as clips and thermocoagulation (98.5 % and 94.5 %, respectively) 80 . Direct comparisons between topical hemostatic agents and conventional endoscopic modalities were only available in 10 studies (n = 797), with topical agents achieving greater immediate hemostasis (OR 3.94 (1.73; 8.96)). This conclusion remained true when including only data derived from RCT (OR 4.01 (1.52; 10.60). This latter subgroup analysis yields a very important summary result previously not reported by other systematic reviews and for which the small number of patients is counterbalanced by the study quality and magnitude of treatment effect 10 11 12 . When different topical hemostatic agents were analyzed separately, all studied compounds appeared effective, although the number of patients studied varied greatly.
The major concern about the use of topical hemostatic agents expressed in previous studies has related to the risk of rebleeding. We found that the overall risk of rebleeding was 18 % (15 %; 21 %) among non-comparative studies. This risk is higher compared to conventional endoscopic approaches 80 81 . This elevated risk likely represents the inclusion of difficult-to-treat lesions (e. g. large ulcers or difficult position) in which these agents are best used as temporizing strategy until a more definitive endoscopic therapy can be employed. It may also relate to the mechanism of action of some of these agents that form a mechanical barrier to stop bleeding but washes off while a risk of rebleeding may persist (within 24 hours as in the case of Tc-325 and peptic ulcers for which the high-risk period post-hemostasis extends to at least 72 hours 29 ). However, studies that directly compared topical agents with conventional endoscopic modalities failed to show a difference in overall rebleeding between the two interventions (OR 1.06 [0.65; 1.74]). Similar conclusions were reached when RCT data only were included, even though the point estimate of overall rebleeding was actually greater for the topical agents, but with a large confidence interval owing to small patient numbers (1.27 [0.62; 2.59]). This observation is congruent with the hypothesis that the risk of rebleeding when using topical agents may have been over-estimated from previous studies, possibly reflecting selection and/or reporting bias. However, it is important to once again emphasize that RCTs comparing topical agents to conventional endoscopic modalities with regards to assessing rebleeding are few, with many exhibiting limitations. For NVUGIB, there were six RCTs with varying sample sizes. The main limitations with all these studies are two-fold: First, Kwek et al. 39 included a large proportion (60 %) of patients with non-bleeding lesions (Forrest IIa and IIb) – lesions to which Hemospray cannot adhere and thus are not indicated for its use. Second, the remaining studies (Baracat et al. 24 , Chen et al. 29 , Martins et al. 42 and Bang et al. 77 ) were small (20–72 patients in each study) and included a large proportion of patients with oozing stigmata (Forrest Ib). This category of lesions has recently been shown to exhibit a lower risk of re-bleeding than previously thought, possibly reflecting the inclusion of lower risk lesions than “true” Forrest Ib 82 . By far, the largest RCT assessing Hemospray, was recently published by Lau et al. 15 This study randomized 224 patients with NVUGIB to Hemospray or conventional endoscopic therapy. It concluded that Hemospray was non-inferior to conventional endoscopic hemostasis. However, the study was limited by the inclusion of a large proportion of Forrest Ib lesions. Furthermore, this study exhibited a marked imbalance in the malignant bleeding subgroup which were over-represented in the Hemospray arm and may have biased the results in favor of the topical agent 16 . Only one study assessed variceal bleeding (Ibrahim et al. 38 ) limiting the conclusions specifically addressing this subgroup of patients.
When the risk of rebleeding was stratified according to the agent used, it was numerically greater with Hemospray (20 % [17 %; 24 %]) compared to other agents (EndoClot 10 % [6 %; 16%], PuraStat 7 % [1 %; 27 %], or Nexpowder 8 % [3 %; 20 %]) and CEGP-003 (9 % [3 %; 23 %]). This difference may not be statistically significant (there are very few direct comparisons) and may only reflect low numbers of patients treated with the other agents or could alternately be a result of the inclusion of higher-risk lesions in the Hemospray studies. Indeed, only one study directly compared different topical hemostatic agents (Hemospray vs. EndoClot) 77 , concluding similar effectiveness, as well as rebleeding rates and safety profiles when used for UGIB of different etiologies in 127 patients.
When stratified by etiology, the risk of rebleeding was similar in NVUGIB, malignancy-related lesions, as well as in variceal bleeding (Appendix 2b). Even though the risk of rebleeding was numerically greater when the topical agent was used for rescue (25 % [20 %; 30 %]) compared to primary therapy (18 % [14 %; 22 %]); this difference is likely to be non-significant as suggested by marked overlapping confidence intervals. Given the ease of use of these topical agents, they comprise an attractive option to manage UGIB in emergency situations. These compounds may also prove to be the preferred endoscopic modalities when managing diffuse bleeding from fragile surfaces such as in malignancy, especially considering the difficulty and limited success when using conventional endoscopic options in such a scenario 1 . This is supported by the current meta-analysis results as we showed that topical agents were more effective in achieving immediate hemostasis than conventional endoscopic therapy when used for malignancy-related bleeding (OR = 14.74 [2.16;100.61]). This, too, is an important finding that bears important clinical implications and that has not been clearly identified previously using summary data.
One of the major strengths of the topical hemostatic agents is their remarkable safety profile. Indeed, the overall AE rate seen with these agents was 2 % (1;3 %). Some of the reported AEs were serious but remain rare, such as the three cases of perforations among 2111 patients. Another important advantage when using these agents is the high technical success rate noted (97 %, 97;98 %). The main technical problem reported was catheter blockage due to premature activation of the powder inside the delivery catheter. This problem was encountered with Hemospray only, and can be avoided by ensuring the endoscope channel and actual delivery catheter are cleared of any liquid (e. g. blood or fluid) before inserting the catheter.
Our study has a number of strengths. It is the largest and most comprehensive meta-analysis performed to-date assessing the effectiveness and safety of different topical hemostatic agents in the management of UGIB. It included 59 studies from different regions of the world published over the past decade, and assessed different agents, hence increasing generalizability. In addition, we assessed the effectiveness of these agents in UGIB of different etiologies (e. g. peptic ulcer disease, malignancy, variceal bleed) and in different settings (primary vs. rescue therapy), further increasing external validity. The main limitation of our analysis is the overall low to very low certainty of the evidence but such limitation exists also with many endoscopic therapeutic modalities yet have not deterred consideration for use in authoritative guidelines with adequate characterization of the data 14 . In addition to limitations in study design, the studies also display a lack of follow-up information in some reports which may have introduced reporting bias. Nevertheless, this systematic review addresses many of the limitations of previously published works that included small number of studies, limited inclusion criteria (limiting generalizability), and no formal assessment of the quality of the evidence using validated instruments such as the GRADE assessment tool 10 11 12 .
Conclusions
In conclusion, topical hemostatic agents are effective and safe in managing UGIB of different etiologies even when used as primary therapy and monotherapy. Patients treated with these agents should be monitored closely in the first few days due to a higher risk of rebleeding. More complete and novel subgroup analyses suggest these agents yield the highest immediate hemostatic rates, and are particularly effective in malignant bleeding, even when compared to other modalities. Future well designed studies should further compare hemostatic topical agents to conventional endoscopic modalities, and among each other to better inform recommendations as to their use, especially when chosen as sole first-line agent.
Acknowledgments
The authors extend their appreciation to the International Scientific Partnership Program ISPP at King Saud University for funding this research work through ISPP-21-156(1).
Footnotes
Competing interests The authors declare that they have no conflict of interest.
Supplementary material :
References
- 1.Lau L HS, Sung J JY. Treatment of upper gastrointestinal bleeding in 2020: New techniques and outcomes. Dig Endosc. 2021;33:83–94. doi: 10.1111/den.13674. [DOI] [PubMed] [Google Scholar]
- 2.Abougergi M S, Travis A C, Saltzman J R. The in-hospital mortality rate for upper GI hemorrhage has decreased over 2 decades in the United States: a nationwide analysis. Gastrointest Endosc. 2015;81:882–888. doi: 10.1016/j.gie.2014.09.027. [DOI] [PubMed] [Google Scholar]
- 3.Rotondano G. Epidemiology and diagnosis of acute nonvariceal upper gastrointestinal bleeding. Gastroenterol Clin North Am. 2014;43:643–663. doi: 10.1016/j.gtc.2014.08.001. [DOI] [PubMed] [Google Scholar]
- 4.Lau J Y, Barkun A, Fan D M et al. Challenges in the management of acute peptic ulcer bleeding. Lancet. 2013;381:2033–2043. doi: 10.1016/S0140-6736(13)60596-6. [DOI] [PubMed] [Google Scholar]
- 5.Hearnshaw S A, Logan R F, Lowe D et al. Acute upper gastrointestinal bleeding in the UK: patient characteristics, diagnoses and outcomes in the 2007 UK audit. Gut. 2011;60:1327–1335. doi: 10.1136/gut.2010.228437. [DOI] [PubMed] [Google Scholar]
- 6.Barkun A N, Moosavi S, Martel M. Topical hemostatic agents: a systematic review with particular emphasis on endoscopic application in GI bleeding. Gastrointest Endosc. 2013;77:692–700. doi: 10.1016/j.gie.2013.01.020. [DOI] [PubMed] [Google Scholar]
- 7.Chen Y I, Barkun A N. Hemostatic powders in gastrointestinal bleeding: A systematic review. Gastrointest Endosc Clin N Am. 2015;25:535–552. doi: 10.1016/j.giec.2015.02.008. [DOI] [PubMed] [Google Scholar]
- 8.Pioche M, Camus M, Rivory J et al. A self-assembling matrix-forming gel can be easily and safely applied to prevent delayed bleeding after endoscopic resections. Endosc Int Open. 2016;4:E415–E419. doi: 10.1055/s-0042-102879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.de Rezende D T, Brunaldi V O, Bernardo W M et al. Use of hemostatic powder in treatment of upper gastrointestinal bleeding: a systematic review and meta-analysis. Endosc Int Open. 2019;7:E1704–E1713. doi: 10.1055/a-0977-2897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mutneja H, Bhurwal A, Go A et al. Efficacy of Hemospray in upper gastrointestinal bleeding: A systematic review and meta-analysis. J Gastrointestin Liver Dis. 2020;29:69–76. doi: 10.15403/jgld-660. [DOI] [PubMed] [Google Scholar]
- 11.Aziz M, Weissman S, Mehta T I et al. Efficacy of Hemospray in non-variceal upper gastrointestinal bleeding: A systematic review with meta-analysis. Ann Gastroenterol. 2020;33:145–154. doi: 10.20524/aog.2020.0448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Facciorusso A, Straus T M, Eyileten P C et al. Efficacy of hemostatic powders in upper gastrointestinal bleeding: A systematic review and meta-analysis. Dig Liver Dis. 2019;51:1633–1640. doi: 10.1016/j.dld.2019.07.001. [DOI] [PubMed] [Google Scholar]
- 13.Barkun A N, Almadi M, Kuipers E J et al. Management of nonvariceal upper gastrointestinal bleeding: guideline recommendations from the International Consensus Group. Ann Intern Med. 2019;171:805–822. doi: 10.7326/M19-1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Laine L, Barkun A N, Saltzman J R et al. ACG Clinical Guideline: Upper Gastrointestinal and Ulcer Bleeding. Am J Gastroenterol. 2021;116:899–917. doi: 10.14309/ajg.0000000000001245. [DOI] [PubMed] [Google Scholar]
- 15.Lau J YW, Pittayanon R, Kwek A et al. Comparison of a hemostatic powder and standard treatment in the control of active bleeding from upper nonvariceal lesions: A multicenter, noninferiority, randomized trial. Ann Intern Med. 2022;175:171–178. doi: 10.7326/M21-0975. [DOI] [PubMed] [Google Scholar]
- 16.Barkun A N, Alali A. The role of hemostatic powder in endoscopic hemostasis of nonvariceal upper gastrointestinal bleeding. Ann Intern Med. 2022;175:289–290. doi: 10.7326/M21-4267. [DOI] [PubMed] [Google Scholar]
- 17.Higgins J P, Altman D G, Gotzsche P C et al. The Cochrane Collaborationʼs tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wells G, Shea B, OʼConnell D. The Newcastle – Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp
- 19.Guyatt G H, Oxman A D, Vist G E et al. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–926. doi: 10.1136/bmj.39489.470347.AD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Laine L, Spiegel B, Rostom A et al. Methodology for randomized trials of patients with nonvariceal upper gastrointestinal bleeding: Recommendations from an international consensus conference. Am J Gastroenterol. 2010;105:540–550. doi: 10.1038/ajg.2009.702. [DOI] [PubMed] [Google Scholar]
- 21.Subramaniam S, Kandiah K, Chedgy F et al. A novel self-assembling peptide for hemostasis during endoscopic submucosal dissection: A randomized controlled trial. Endoscopy. 2021;53:27–35. doi: 10.1055/a-1198-0558. [DOI] [PubMed] [Google Scholar]
- 22.Appleby R, Hoare J. Hemospray in a large tertiary NHS trust: A descriptive analysis of the first three years of use. Gut. 2017;66:A216. [Google Scholar]
- 23.Arena M, Masci E, Eusebi L H et al. Hemospray for treatment of acute bleeding due to upper gastrointestinal tumours. Dig Liver Dis. 2017;49:514–517. doi: 10.1016/j.dld.2016.12.012. [DOI] [PubMed] [Google Scholar]
- 24.Baracat F I, de Moura D TH, Brunaldi V O et al. Randomized controlled trial of hemostatic powder versus endoscopic clipping for non-variceal upper gastrointestinal bleeding. Surg Endosc. 2020;34:317–324. doi: 10.1007/s00464-019-06769-z. [DOI] [PubMed] [Google Scholar]
- 25.Becq A, Houdeville C, Tran Minh M L. Experience with the use of a hemostatic powder in 152 patients undergoing urgent endoscopy for gastrointestinal bleeding. Clin Res Hepatol Gastroenterol. 2020;45:101558. doi: 10.1016/j.clinre.2020.10.003. [DOI] [PubMed] [Google Scholar]
- 26.Bestari M B, Joewono I R, Girawan D et al. Hemospray during emergency endoscopy: indonesiaʼs first experience from 37 patients. Case Rep Gastroenterol. 2020;14:70–79. doi: 10.1159/000505775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cahyadi O, Bauder M, Meier B et al. Effectiveness of TC-325 (Hemospray) for treatment of diffuse or refractory upper gastrointestinal bleeding – a single center experience. Endosc Int Open. 2017;5:E1159–E1164. doi: 10.1055/s-0043-118794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chahal D, Lee J GH, Ali-Mohamad N et al. High rate of re-bleeding after application of Hemospray for upper and lower gastrointestinal bleeds. Dig Liver Dis. 2020;52:768–772. doi: 10.1016/j.dld.2020.01.009. [DOI] [PubMed] [Google Scholar]
- 29.Chen Y I, Barkun A, Nolan S. Hemostatic powder TC-325 in the management of upper and lower gastrointestinal bleeding: A two-year experience at a single institution. Endoscopy. 2015;47:167–171. doi: 10.1055/s-0034-1378098. [DOI] [PubMed] [Google Scholar]
- 30.Chen Y I, Wyse J, Lu Y et al. TC-325 hemostatic powder versus current standard of care in managing malignant GI bleeding: A pilot randomized clinical trial. Gastrointest Endosc. 2020;91:321–328 e1. doi: 10.1016/j.gie.2019.08.005. [DOI] [PubMed] [Google Scholar]
- 31.Disney B, Kurup A, Muhammad H et al. Hemospray use for the management of acute bleeding from upper gastrointestinal cancer: The Russells Hall experience. Gut. 2015;64:A71–A72. [Google Scholar]
- 32.Giles H, Lal D, Gerred S et al. Efficacy and safety of TC-325 (Hemospray) for non-variceal upper gastrointestinal bleeding at Middlemore Hospital: The early New Zealand experience. N Z Med J. 2016;129:38–43. [PubMed] [Google Scholar]
- 33.Gupta T, Leet F, Harborne M et al. Hemospray use in acute gastrointestinal bleeding – a 4 year single centre experience. Gut. 2018;67:A39. [Google Scholar]
- 34.Haddara S, Jacques J, Lecleire S et al. A novel hemostatic powder for upper gastrointestinal bleeding: a multicenter study (the “GRAPHE” registry) Endoscopy. 2016;48:1084–1095. doi: 10.1055/s-0042-116148. [DOI] [PubMed] [Google Scholar]
- 35.Hagel A F, Albrecht H, Nagel A et al. The application of Hemospray in gastrointestinal bleeding during emergency endoscopy. Gastroenterol Res Pract. 2017;2017:3.083481E6. doi: 10.1155/2017/3083481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Holster I L, Poley J W, Kuipers E J et al. Controlling gastric variceal bleeding with endoscopically applied hemostatic powder (Hemospray) J Hepatol. 2012;57:1397–1398. doi: 10.1016/j.jhep.2012.07.024. [DOI] [PubMed] [Google Scholar]
- 37.Ibrahim M, El-Mikkawy A, Abdalla H et al. Management of acute variceal bleeding using hemostatic powder. United European Gastroenterol J. 2015;3:277–283. doi: 10.1177/2050640615570148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ibrahim M, El-Mikkawy A, Abdel Hamid M et al. Early application of haemostatic powder added to standard management for oesophagogastric variceal bleeding: A randomised trial. Gut. 2019;68:844–853. doi: 10.1136/gutjnl-2017-314653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kwek B EA, Ang T L, Ong P LJ et al. TC-325 versus the conventional combined technique for endoscopic treatment of peptic ulcers with high-risk bleeding stigmata: A randomized pilot study. J Dig Dis. 2017;18:323–329. doi: 10.1111/1751-2980.12481. [DOI] [PubMed] [Google Scholar]
- 40.Leblanc S, Vienne A, Dhooge M et al. Early experience with a novel hemostatic powder used to treat upper GI bleeding related to malignancies or after therapeutic interventions (with videos) Gastrointest Endosc. 2013;78:169–175. doi: 10.1016/j.gie.2013.03.006. [DOI] [PubMed] [Google Scholar]
- 41.Malik A, Duane P, Eadala P. Use of Hemospray for non variceal upper gastrointestinal bleed in a district general hospital. Gut. 2015;64:A64. [Google Scholar]
- 42.Martins B, Corsato R, Batista Pires C et al. Preliminary results of a randomized controlled trial comparing hemostatic powder versus optimal clinical treatment in the management. United Europ Gastroenterol J. 2019;8S:P0135. [Google Scholar]
- 43.Masci E, Arena M, Morandi E et al. Upper gastrointestinal active bleeding ulcers: Review of literature on the results of endoscopic techniques and our experience with Hemospray. Scand J Gastroenterol. 2014;49:1290–1295. doi: 10.3109/00365521.2014.946080. [DOI] [PubMed] [Google Scholar]
- 44.Meng Z W, Marr K J, Mohamed R et al. Long-term effectiveness, safety and mortality associated with the use of TC-325 for malignancy-related upper gastrointestinal bleeds: A multicentre retrospective study. J Can Assoc Gastroenterol. 2019;2:91–97. doi: 10.1093/jcag/gwy031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Min T, Mothey M, Sharrack N et al. Efficacy of haemospray therapy on re-bleed and mortality rates – a UK single-centre experience. United Europ Gastroenterol J. 2018;8S:P0773. [Google Scholar]
- 46.Nasr I, De Martino S, Borrow D. PWE-007 When to use hemospray? A single centre experience. Gut. 2015;64 (supp1):A212–A213. [Google Scholar]
- 47.Pittayanon R, Prueksapanich P, Rerknimitr R. The efficacy of Hemospray in patients with upper gastrointestinal bleeding from tumor. Endosc Int Open. 2016;4:E933–E936. doi: 10.1055/s-0042-109863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pittayanon R, Rerknimitr R, Barkun A. Prognostic factors affecting outcomes in patients with malignant GI bleeding treated with a novel endoscopically delivered hemostatic powder. Gastrointest Endosc. 2018;87:994–1002. doi: 10.1016/j.gie.2017.11.013. [DOI] [PubMed] [Google Scholar]
- 49.Prentice E, Masegosa Ataz J, Barron M et al. A retrospective single-centre review of hemospray in GI bleeding. United Europ Gastroenterol J. 2018;8S:P1105. [Google Scholar]
- 50.Ramirez-Polo A I, Casal-Sanchez J, Hernandez-Guerrero A et al. Treatment of gastrointestinal bleeding with hemostatic powder (TC-325): A multicenter study. Surg Endosc. 2019;33:2349–2356. doi: 10.1007/s00464-019-06719-9. [DOI] [PubMed] [Google Scholar]
- 51.Shivaji U, Leet F, Gupta T et al. Hemospray use in acute gastrointestinal bleeding – a 4-year single-centre experience. United Europ Gastroenterol J. 2018;8S:P1726. [Google Scholar]
- 52.Sinha R, Lockman K A, Church N I et al. The use of hemostatic spray as an adjunct to conventional hemostatic measures in high-risk nonvariceal upper GI bleeding (with video) Gastrointest Endosc. 2016;84:900–906 e3. doi: 10.1016/j.gie.2016.04.016. [DOI] [PubMed] [Google Scholar]
- 53.Smith L A, Stanley A J, Bergman J J et al. Hemospray application in nonvariceal upper gastrointestinal bleeding: Results of the Survey to Evaluate the Application of Hemospray in the Luminal Tract. J Clin Gastroenterol. 2014;48:e89–e92. doi: 10.1097/MCG.0000000000000054. [DOI] [PubMed] [Google Scholar]
- 54.Sulz M C, Frei R, Meyenberger C et al. Routine use of Hemospray for gastrointestinal bleeding: Prospective two-center experience in Switzerland. Endoscopy. 2014;46:619–624. doi: 10.1055/s-0034-1365505. [DOI] [PubMed] [Google Scholar]
- 55.Sung J J, Luo D, Wu J C et al. Early clinical experience of the safety and effectiveness of Hemospray in achieving hemostasis in patients with acute peptic ulcer bleeding. Endoscopy. 2011;43:291–295. doi: 10.1055/s-0030-1256311. [DOI] [PubMed] [Google Scholar]
- 56.Thayalasekaran S, Bhandari P, Quine A et al. Hemospray use in the management of upper gastrointestinal haemorrhage: A 2-year experience across 2 teaching hospitals in the north and south of England. Gut. 2017;85:AB35. [Google Scholar]
- 57.Weaver M, Richard R, Rood M. Clinical outcomes and endoscopist’s perception of the use of hemospray for gastrointestinal bleeding: Outcomes from a U.S. academic tertiary care medical center. Am J Gastroenterol. 2019;114:S363. [Google Scholar]
- 58.Widlak M, Wijesinghe H, Siau K. PWE-032 Hemospray for acute upper gastrointestinal bleeding – a single centre experience. Gut. 2015;64 (supp1):A225. [Google Scholar]
- 59.Yau A H, Ou G, Galorport C et al. Safety and efficacy of Hemospray in upper gastrointestinal bleeding. Can J Gastroenterol Hepatol. 2014;28:72–76. doi: 10.1155/2014/759436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rodriguez de Santiago E, Burgos-Santamaria D, Perez-Carazo L et al. Hemostatic spray powder TC-325 for GI bleeding in a nationwide study: Survival and predictors of failure via competing risks analysis. Gastrointest Endosc. 2019;90:581–590 e6. doi: 10.1016/j.gie.2019.06.008. [DOI] [PubMed] [Google Scholar]
- 61.Hussein M, Alzoubaidi D, Lopez M F et al. Hemostatic spray powder TC-325 in the primary endoscopic treatment of peptic ulcer-related bleeding: multicenter international registry. Endoscopy. 2021;53:36–43. doi: 10.1055/a-1186-5360. [DOI] [PubMed] [Google Scholar]
- 62.Hussein M, Alzoubaidi D, OʼDonnell M et al. Hemostatic powder TC-325 treatment of malignancy-related upper gastrointestinal bleeds: International registry outcomes. J Gastroenterol Hepatol. 2021;36:3027–3032. doi: 10.1111/jgh.15579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hussein M, Alzoubaidi D, Serna A et al. Outcomes of Hemospray therapy in the treatment of intraprocedural upper gastrointestinal bleeding post-endoscopic therapy. United Europ Gastroenterol J. 2020;8:1155–1162. doi: 10.1177/2050640620938549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hussein M, Alzoubaidi D, Weaver M et al. Hemospray in the treatment of variceal bleeds: outcomes from the international Hemospray registry. Gut. 2021;70:A1–A262. [Google Scholar]
- 65.Beg S, Al-Bakir I, Bhuva M et al. Early clinical experience of the safety and efficacy of EndoClot in the management of non-variceal upper gastrointestinal bleeding. Endosc Int Open. 2015;3:E605–E609. doi: 10.1055/s-0034-1393087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kim Y J, Park J C, Kim E H et al. Hemostatic powder application for control of acute upper gastrointestinal bleeding in patients with gastric malignancy. Endosc Int Open. 2018;6:E700–E705. doi: 10.1055/a-0593-5884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Prei J C, Barmeyer C, Burgel N et al. EndoClot polysaccharide hemostatic system in nonvariceal gastrointestinal bleeding: Results of a prospective multicenter observational pilot study. J Clin Gastroenterol. 2016;50:e95–e100. doi: 10.1097/MCG.0000000000000615. [DOI] [PubMed] [Google Scholar]
- 68.Hagel A F, Raithel M, Hempen P. Multicenter analysis of endoclot as hemostatic powder in different endoscopic settings of the upper gastrointestinal tract. J Physiol Pharmacol. 2020;71:657–664. doi: 10.26402/jpp.2020.5.06. [DOI] [PubMed] [Google Scholar]
- 69.Park J C, Kim Y J, Kim E H et al. Effectiveness of the polysaccharide hemostatic powder in non-variceal upper gastrointestinal bleeding: Using propensity score matching. J Gastroenterol Hepatol. 2018;33:1500–1506. doi: 10.1111/jgh.14118. [DOI] [PubMed] [Google Scholar]
- 70.de Nucci G, Reati R, Arena I et al. Efficacy of a novel self-assembling peptide hemostatic gel as rescue therapy for refractory acute gastrointestinal bleeding. Endoscopy. 2020;52:773–779. doi: 10.1055/a-1145-3412. [DOI] [PubMed] [Google Scholar]
- 71.Subramaniam S, Kandiah K, Thayalasekaran S et al. Haemostasis and prevention of bleeding related to ER: The role of a novel self-assembling peptide. United Europ Gastroenterol J. 2019;7:155–162. doi: 10.1177/2050640618811504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Branchi F, Klingenberg-Noftz R, Friedrich K et al. PuraStat in gastrointestinal bleeding: results of a prospective multicentre observational pilot study. Surg Endosc. 2022;36:2954–2961. doi: 10.1007/s00464-021-08589-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Labianca O, Sica M, Zulli C. Use of Purastat in management of gastro-intestinal bleeding: our experience. Endoscopy. 2021;53:S97–S98. [Google Scholar]
- 74.Park J S, Bang B W, Hong S J et al. Efficacy of a novel hemostatic adhesive powder in patients with refractory upper gastrointestinal bleeding: A pilot study. Endoscopy. 2019;51:458–462. doi: 10.1055/a-0809-5276. [DOI] [PubMed] [Google Scholar]
- 75.Park J-S, Lee D-H, Hong S-J. Novel hemostatic adhesive powder application in nonvariceal upper gastrointestinal bleeding. UEG J. 2019;7:133–134. doi: 10.1055/a-0982-3194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Shin J, Cha B, Park J S et al. Efficacy of a novel hemostatic adhesive powder in patients with upper gastrointestinal tumor bleeding. BMC Gastroenterol. 2021;21:40. doi: 10.1186/s12876-021-01611-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Bang B W, Lee D H, Kim H K et al. CEGP-003 spray has a similar hemostatic effect to epinephrine injection in cases of acute upper gastrointestinal bleeding. Dig Dis Sci. 2018;63:3026–3032. doi: 10.1007/s10620-018-5208-z. [DOI] [PubMed] [Google Scholar]
- 78.Vitali F, Naegel A, Atreya R et al. Comparison of Hemospray and Endoclot for the treatment of gastrointestinal bleeding. World J Gastroenterol. 2019;25:1592–1602. doi: 10.3748/wjg.v25.i13.1592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Paoluzi O A, Cardamone C, Aucello A et al. Efficacy of hemostatic powders as monotherapy or rescue therapy in gastrointestinal bleeding related to neoplastic or non-neoplastic lesions. Scand J Gastroenterol. 2021;56:1–8. doi: 10.1080/00365521.2021.1974088. [DOI] [PubMed] [Google Scholar]
- 80.Sung J J, Tsoi K K, Lai L H et al. Endoscopic clipping versus injection and thermo-coagulation in the treatment of non-variceal upper gastrointestinal bleeding: A meta-analysis. Gut. 2007;56:1364–1373. doi: 10.1136/gut.2007.123976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bratanic A, Puljiz Z, Ljubicicz N et al. Predictive factors of rebleeding and mortality following endoscopic hemostasis in bleeding peptic ulcers. Hepatogastroenterology. 2013;60:112–117. doi: 10.5754/hge11838. [DOI] [PubMed] [Google Scholar]
- 82.Jensen D M, Eklund S, Persson T et al. Reassessment of rebleeding risk of forrest ib (oozing) peptic ulcer bleeding in a large international randomized trial. Am J Gastroenterol. 2017;112:441–446. doi: 10.1038/ajg.2016.582. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


