Abstract
Pseudomonas aeruginosa is an opportunistic bacterial pathogen responsible for multiple hospital- and community-acquired infections, both in human and veterinary medicine. P. aeruginosa persistence in clinical settings is worrisome and is a result of its remarkable flexibility and adaptability. This species exhibits several characteristics that allow it to thrive under different environmental conditions, including the ability to colonize inert materials such as medical equipment and hospital surfaces. P. aeruginosa presents several intrinsic mechanisms of defense that allow it to survive external aggressions, but it is also able to develop strategies and evolve into multiple phenotypes to persevere, which include antimicrobial-tolerant strains, persister cells, and biofilms. Currently, these emergent pathogenic strains are a worldwide problem and a major concern. Biocides are frequently used as a complementary/combination strategy to control the dissemination of P. aeruginosa-resistant strains; however, tolerance to commonly used biocides has also already been reported, representing an impediment to the effective elimination of this important pathogen from clinical settings. This review focuses on the characteristics of P. aeruginosa responsible for its persistence in hospital environments, including those associated with its antibiotic and biocide resistance ability.
Keywords: antibiotic resistance, biocide tolerance, hospital environment, inert materials, Pseudomonas aeruginosa, virulence factors
1. Introduction
Pseudomonas aeruginosa, an aerobic, Gram-negative, rod-shaped bacterium, exhibits several characteristics that allow it to thrive under different environmental conditions. It is a ubiquitous microorganism that can be found both in abiotic and biotic environments, being able to tolerate a wide range of temperatures, varying from 4 °C to 42 °C [1,2]. Soil and water environments are common habitats for this bacterial species, whose opportunistic nature can partially explain its association with several hospital- and community-acquired infections [1,3,4,5,6].
P. aeruginosa is associated with high morbidity and mortality rates, both in human and veterinary medicine, especially in immunocompromised individuals [3,7]. In fact, according to the Centers for Disease Control and Prevention, in 2017 P. aeruginosa caused approximately 32,000 infections among hospitalized patients, being responsible for 2700 deaths in the United States alone [8]. This bacterium has been associated with several types of hospital-acquired infections, including ventilator-associated pneumonia, urinary tract infections in patients with long-term urinary catheterization, wound infections, bloodstream infections, and otitis [5,6,7,8,9,10,11,12]. P. aeruginosa infections can range from mild to life-threatening conditions, associated with treatment failure [1,5]. This species is also a major threat in veterinary medicine, being associated with chronic otitis externa, pyoderma, conjunctivitis, septicemia, lower urinary tract infections, pneumonia, and bacterial endocarditis [13,14,15,16,17,18,19,20,21,22,23,24,25].
The pathogenic potential of P. aeruginosa is related to its ability to express several virulence factors, including survival and stress-resistant mechanisms. In fact, this bacterial species frequently presents a multidrug-resistant profile and biofilm production ability, traits that render its elimination challenging. P. aeruginosa’s resistance to several antibiotics, including aminoglycosides, quinolones, and β-lactams, has been reported, and the emergence of new carbapenemase-producing strains is of major concern [23,26]. In fact, P. aeruginosa carbapenem-resistant strains have been classified by the World Health Organization since 2017 as critical-priority pathogens for the development of new antibiotics [27].
P. aeruginosa’s capacity to survive in different environments is also worrisome, as it can grow on a wide variety of surfaces, ranging from medical equipment and devices to sinks and drains from water systems [9,11,28,29,30,31,32]. The development of biofilms and persister cells poses a major challenge for surface disinfection and demands the characterization of the efficacy of disinfectants against these antimicrobial-resistant and -tolerant phenotypes [33,34,35,36,37,38].
This article reviews P. aeruginosa’s characteristics associated with its survival and persistence in hospital environments and surfaces, which represent a major concern in terms of nosocomial infections.
2. Genomic Structure
P. aeruginosa has the remarkable ability to adapt to a wide range of adverse and stressful environmental conditions. Having a large genome, of approximately 6.3 million base pairs in size and containing over 6000 protein-coding genes, this bacterial species presents a high genomic plasticity [39,40,41]. Its genetic material is divided into the core genome (~4 Mb) and the accessory genome, which mainly consists of variable-length and non-conserved extrachromosomal elements, such as plasmids. These mobile elements may include genomic islands [42], which are highly responsible for the versatility of this pathogen as they may contain genes related to virulence and antibiotic resistance abilities [40,42].
P. aeruginosa’s genome contains several genes involved in metabolic pathways, transport systems, and motility. The determinants responsible for the intrinsic resistance profile of this pathogen (i.e., associated with efflux pumps and antibiotic-inactivating enzymes) can be found in the core genome [39,41,42] and are also associated with its metabolic versatility and adaptability capacity [41].
3. Virulence
P. aeruginosa is well known for its high pathogenic potential, especially towards seriously ill and immunocompromised patients [43], which is related to its ability to express a wide variety of virulence factors [44]. A few of these traits also give the bacteria advantages in terms of environmental survival, allowing them to adhere to different surfaces and even form biofilms [33,45].
Some structural components of P. aeruginosa cells act as virulence determinants, but this species can also synthesize additional virulence factors, which can be excreted into the surrounding environment or directly into the host [44,46].
According to [47], virulence factors can be divided into three categories: cell surface structures, secreted factors, and factors involved in bacterial cell-to-cell interaction (Table 1) [44,46,47,48]. In this review, we discuss the most significant ones, especially those associated with P. aeruginosa’s ability to evade antimicrobial action.
Table 1.
P. aeruginosa virulence factors.
| Categories | Virulence Factors |
|---|---|
| Cell surface structures |
|
| Secreted factors |
|
| Bacterial cell-to-cell interaction |
|
3.1. Virulence Factors Related to the Cell Membrane
Like most Gram-negative bacteria, P. aeruginosa contains an outer membrane (OM) composed of lipopolysaccharide (LPS), pili, flagella, and about 300 proteins with several functions, most of which are still unknown. These structures confer advantages on this species, being involved in bacterial interaction with the host and in environmental survival [44,46].
LPS is fundamental to the integrity of the bacterial cell, but it also plays a relevant part in the host immune response and tissue damage, antibiotic resistance ability, and biofilm formation [46,49]. It consists of three domains: lipid A, core, and O-polysaccharide. Lipid A, a hydrophobic glycopeptide, is the inner portion of the LPS molecule, responsible for its endotoxicity. It promotes local and systemic inflammatory responses, necrosis, and septic shock through the disruption of cellular activities and macrophage activation [44,50]. The O-polysaccharide, or O-antigen, is the outer portion of the LPS, and is highly immunogenic, being responsible for bacterial interactions with the host or the environment, protecting it from phagocytosis and oxidative stress [49].
Motility is an important trait, as it allows bacteria to reach available nutrients. P. aeruginosa presents a single flagellum at one of its poles, as well as several pili and fimbriae. The flagellum is responsible for bacterial mobility, while the fimbriae allow it to adhere to host epithelial cells or environmental surfaces [44,47]. Additionally, these proteins can also initiate an inflammatory response in the host and have already been associated with drug resistance and biofilm formation ability in P. aeruginosa [45,49,51]. Pili play a crucial role in the initial stages of P. aeruginosa infections, as they contribute to bacterial twitching and swarming motility, the adhesion to surfaces, and biofilm formation [46,47].
OM proteins are involved in microbial nutrition by transporting amino acids, peptides, and carbon sources, but also antibiotics [44,52]. Additionally, they play a key role in bacterial adhesion and host recognition [44]. P. aeruginosa expresses twenty-six specialized channels for the transport of molecules, known as porins. These proteins can be divided into four categories, namely non-specific porins, responsible for the slow diffusion of small hydrophilic molecules; specific porins, which only allow the diffusion of particular molecules; gated porins, responsible for the uptake of ion complexes; and efflux porins, present in efflux pumps [4,12,53].
3.2. Secreted Virulence Factors
Most P. aeruginosa virulence factors are synthesized in its cytoplasm before being expelled to the extracellular space or introduced directly into the host cells through complex secretion systems, thereby avoiding elimination by the host immune system [46,54].
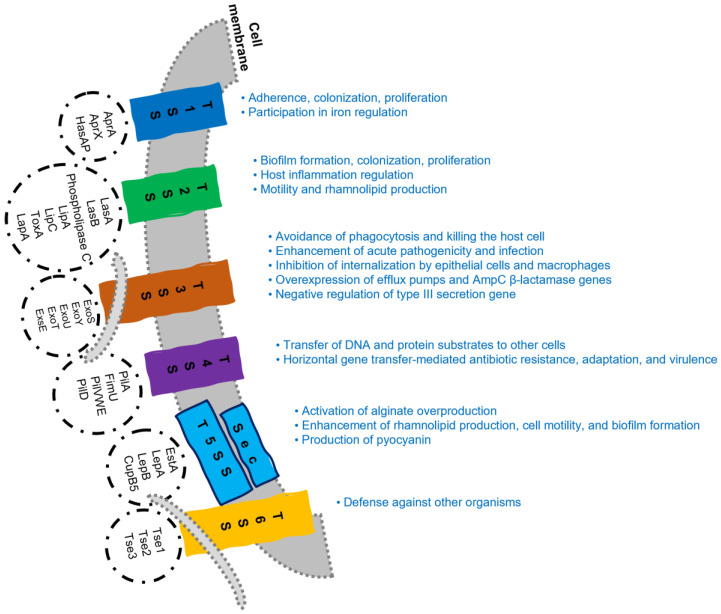
P. aeruginosa expresses six types of secretion systems (Figure 1) that participate in the transport of several virulence factors, including enzymes, exotoxins, and other proteins [20,44,48,54,55,56,57,58].
Figure 1.
Schematic representation of P. aeruginosa secretion systems (T1SS to T6SS) and their functions (original).
These systems are divided into two groups: one-step secretion systems (T1SS, T3SS, T4SS, and T6SS) and two-step secretion systems (T2SS and T5SS), which differ in the course of protein transport. In one-step secretion systems, the protein is directly transferred from the bacterial cytosol to its surface, crossing both the inner and outer membranes in a single step. In two-step secretion systems, the protein is temporarily stored in the periplasmic space located between the inner and outer membranes, before being released to the external environment (Figure 1). These differences affect the efficiency and specificity of protein transport [20,44,55,56,57,58].
T1SS has three structural elements, an ABC transporter protein, a membrane fusion protein, and an outer membrane factor, which participate in the one-step transfer of substrates across bacterial membranes [58,59,60]. T2SS secretes folded proteins, which are first transported through the inner membrane to the periplasm via the general secretory (Sec) or twin-arginine translocation (Tat) secretion mechanisms, after which they are secreted to the cell exterior. T2SS is mainly involved in nutrient acquisition [58,59,60]. T3SS transfers virulence proteins, named effectors, from the bacterial cytoplasm into the eukaryotic cell in a one-step process [58,59,60]. T4SS, unlike other secretion systems, is capable of transferring DNA in addition to proteins [57,61]. T5SS has a Sec translocase, which allows substrates to cross the inner membrane and reach the periplasm [55,59,60]. T6SS transports toxic substrates that can act on both eukaryotic [62] and prokaryotic cells [63], playing an important role in P. aeruginosa pathogenesis [59,60].
Beyond secretion systems, P. aeruginosa produces several other virulence determinants, including enzymes such as lipases, elastase A and B, and rhamnolipids [44,46,64]. Moreover, this species is known to produce several pigments that participate in iron scavenging, cell protection, and bacterial competition [1]. Pigments produced by P. aeruginosa include pyocyanin, a redox-active blue pigment that causes oxidative stress in the host by interfering with electron transport [64,65]; pyoverdine, a fluorescent siderophore that helps the bacterium to acquire iron—an essential nutrient for bacterial growth and virulence—from the surrounding environment [1]; and pyorubin, a red pigment that also participates in iron acquisition and in the maintenance of the redox equilibrium of the cell [44,46].
Some other relevant substances synthesized and secreted by P. aeruginosa include exotoxin A, a potent cytotoxin that leads to necrosis; ExoS, that has a negative impact on the host immune system; ExoU, associated with disease severity and increased mortality [12,66,67]; and exopolysaccharides, such as alginate, Psl, and Pel, responsible for bacterial tolerance to harsh conditions, such as desiccation and the presence of oxidizing agents, and for evasion of host defenses [47,68]. They are crucial for biofilm formation as they are the main components of its matrix [68,69].
In fact, besides having an obvious role in P. aeruginosa pathogenicity, some of these virulence factors contribute to P. aeruginosa’s resilience in a variety of ecological niches, helping its communication and competition with other bacteria.
3.3. Quorum Sensing Systems
To regulate all these virulence factors, P. aeruginosa relies on a highly coordinated communication system, known as quorum sensing (QS). Due to QS, bacteria are capable of sensing variations in population density and environmental conditions and of coordinating the behavior of the bacterial population [1,70]. This communication system is based on the expression of small signaling molecules. When the concentration of these signals reaches a certain threshold, they collectively activate specific transcriptional regulators that control gene expression [44,70].
P. aeruginosa has four distinct QS systems: the acyl-homoserine lactone QS systems (Las and Rhl), the quinolone QS system (Pqs), and the novel QS system (Iqs). The Las system positively controls the other three. Likewise, the Iqs system positively affects Pqs, which in turn stimulates Rhl, which has a negative effect on Pqs [46,70,71,72].
In the Las system, LasI binds to the transcriptional activator LasR, regulating the transcription of different virulent factors, such as LasA, LasB, AprA, PVD, and ETA, which are involved in host cell damage and acute infections. LasI induces the production of the autoinducer N-3-oxododecanoyl-L-homoserine lactone (C12HSL), creating an autoregulatory loop [46,73]. Together with pyocyanin, C12HSL is involved in the formation of persister cells by P. aeruginosa, which are recognized as responsible for recalcitrant chronic infections [74]. The Las system also induces apoptosis in airway epithelial cells by destroying their tight junctions, and, along with the Rhl and Pqs systems, affects T6SS [75].
RhlI is involved in the synthesis of the autoinducer N-butyryl-L-homoserine lactone (C4HSL). C4HSL forms a complex with the activator protein RhlR, participating in biofilm formation, LecA production, and the repression of genes involved in T3SS production [46,73].
The several alkyl-4(1H)-quinolones which participate in P. aeruginosa’s quinolone QS system are synthesized by enzymes coded in three gene clusters (pqsABCDE, phnAB, and pqsH) [76]. The most common is 2-heptyl-hydroxy-1H-quinolin-4-one (PQS), which acts as a mediator in iron acquisition, cytotoxicity, and the biogenesis of outer membrane vesicles. It also stimulates neutrophil chemotaxis, prompts the production of reactive oxygen species and tumor necrosis factor-α, and impairs the secretion of cytokines IL-2 and IL-12 [76]. The Pqs system binds to the pqsABCDE promoter, creating a positive feedback loop and promoting PqsE production, which is the major virulence effector of the quinolone system [76]. Together with the Rhl system, PqsE is involved in the regulation of pyocyanin synthesis, the expression of genes associated with iron starvation, and the formation of efflux pumps. The Pqs system also mediates the release of extracellular DNA (eDNA), which is vital for the development of a stable and mature biofilm [46,77].
In the novel P. aeruginosa QS system, Iqs is driven by the signal molecule 2-(2-hydroxyphenyl)-thiazole-4-carbaldehyde (IQS) [46,73]. Iqs monitors bacterial density and detects decreases in the concentration of phosphate, an indicator of infection-associated stress, in order to regulate the production of virulence factors [65]. It also controls Las system functions and, when disrupted, pyocyanin production [65]. Importantly, IQS inhibits host cell growth and stimulates apoptosis in a dosage-dependent manner [78].
The regulation of virulence factors depends on cell density and the release of the QS autoinducers Las, Rhl, Pqs, and Iqs. Together with other virulence factors, QS systems play a major role in P. aeruginosa virulence and survival [46].
4. Antibiotic Resistance
Antibiotics are a powerful tool in the treatment of any bacterial infection. However, the emergence of pathogens resistant to multiple antimicrobial agents has become a major public health threat worldwide [79,80,81,82]. Bacteria are classified as multidrug-resistant when they are not susceptible to at least one agent in three or more classes of antibiotics; as extensively drug-resistant when they are not susceptible to at least one agent in all categories but are susceptible to two or fewer antimicrobial categories; and as pan-drug-resistant when they are not susceptible to any agents in any antimicrobial categories [83].
P. aeruginosa is well known for its resistance capacity towards multiple antibiotics. In fact, the most recent report published by the European Center for Disease Control states that 30.1% of the P. aeruginosa isolates reported are resistant to at least 1 of the antimicrobial groups under surveillance [84].
P. aeruginosa presents three main types of resistance mechanisms: intrinsic, acquired, and adaptative.
4.1. Intrinsic Resistance
Intrinsic antibiotic resistance refers to the inherent characteristics of bacteria that allow them to survive the action of antibiotics and other antimicrobial agents [6,44]. This type of resistance depends on bacterial cell structures and does not develop due to previous contact with inhibitory compounds. For instance, P. aeruginosa is known for its intrinsic resistance to several antibiotics, associated with the decreased permeability of its OM, the expression of efflux pump systems, and the production of antibiotic-inactivating enzymes [67,85,86]. These mechanisms may be responsible for bacterial resistance towards β-lactams and quinolones, which penetrate the cell through porin channels, and towards aminoglycosides and polymyxins, whose uptake depends on the interaction with the bacterial OM [87,88].
4.1.1. OM Permeability
P. aeruginosa can decrease OM permeability by managing the number of non-specific porins present in the membrane [4,53]. OprF is the most prevalent porin in this bacterial species. Although non-selective, it is associated with low efficiency in antibiotic diffusion, since only a small fraction of OprF can form open channels [4,12]. OprD also participates in antibiotic uptake. It contains the binding sites for carbapenems, and low numbers of this porin confer a basal level of resistance to this class of antibiotics [89]. Additionally, OprH overexpression has been related to P. aeruginosa’s decreased susceptibility to polymyxin B and gentamicin, as it induces modifications in the LPS [12,90,91].
4.1.2. Efflux Systems
Another mechanism associated with antibiotic resistance is the presence of efflux pumps, which remove harmful agents from the interior of the bacterial cell, helping to maintain a stable internal environment [92]. P. aeruginosa has active multidrug efflux pumps that contribute significantly to its antibiotic resistance ability. These pumps can be classified into five different families: the resistance nodulation division family (RND), major facilitator superfamily, ATP-binding cassette superfamily, small multidrug resistance family, and multidrug and toxic compound extrusion family [12,92,93,94].
RND is particularly involved in antibiotic resistance in P. aeruginosa. This bacterial species expresses four well-known active efflux pumps belonging to the RND family, including MexAB-OprM responsible for the efflux of β-lactams, including carbapenems and quinolones; MexXY/OprM involved in intrinsic resistance to aminoglycosides; MexCD-OprJ also responsible for β-lactams elimination; and MexEF-OprN responsible for quinolone extrusion [92,93,95,96,97]. The overexpression of multiple efflux pumps by this species has been associated with multidrug resistance [12,95].
4.1.3. Antibiotic-Inactivating Enzymes
P. aeruginosa produces enzymes capable of selectively inactivating or modifying antibiotics, such as β-lactamases and aminoglycoside-modifying enzymes [98,99].
β-lactamases are hydrolytic enzymes, produced by most Gram-negative bacteria, which can inactivate β-lactam antibiotics by breaking the peptide bonds present in their molecules [100]. P. aeruginosa can express several β-lactamases belonging to different classes, such as penicillinases, cephalosporinases, cephamycinases, and carbapenemases, and was also found to be able to produce extended-spectrum β-lactamases, which confer resistance to an even wider range of antimicrobial agents [101,102].
P. aeruginosa’s resistance to aminoglycosides can be attributed to enzymatic modification that can occur through three different pathways: via aminoglycoside phosphotransferase, which can inactivate antibiotics such as neomycin and streptomycin by transferring a phosphoryl group to the 3-hydroxyl group; via aminoglycoside acetyltransferase, which inactivates gentamicin and tobramycin by transferring the acetyl group to the amino group; and via aminoglycoside nucleotidyltranferase that induce resistance to amikacin by transferring adenosine to either the amino or hydroxyl groups present in this antibiotic [103,104,105].
4.2. Acquired Resistance
The acquisition of new resistance determinants implies a change in the microorganism’s genetic material and can occur through gene mutation or the acquisition of new DNA by horizontal gene transfer [67,106]. Contrary to intrinsic resistance, the development of acquired resistance is highly influenced by external stressors, such as exposure to antibiotics. In the presence of such compounds, resistant bacteria present a selective advantage over susceptible strains; this way, the surviving bacteria will give rise to a resistant population [106,107,108,109].
Mutations can be induced or occur spontaneously and can be responsible for permanent changes in the bacterium gene structure. If its new traits are beneficial for the bacteria and contribute to its survival in adverse conditions, they will probably become predominant as they are transmitted to subsequent generations [110]. In P. aeruginosa, mutations can result in modifications of antibiotic targets or porin channels, and therefore in a reduced antibiotic uptake, or in the increased expression of resistance genes, and consequently in the overproduction of antibiotic-inactivating enzymes and multidrug efflux pumps [106,109,111].
Increased production of the AmpC enzyme is frequently observed in P. aeruginosa, being responsible for a high level of resistance to β-lactams, including to third- and fourth-generation cephalosporins [112,113,114]. It can occur due to mutations in the ampC gene itself or in the ampD gene, which codes for the cytosolic N-acetyl-anhydromuramil-l-alanine amidase, which is a repressor of ampC expression and contributes to the overproduction of β-lactamases [99,115,116].
The overexpression of efflux pump systems is another commonly acquired resistance trait in P. aeruginosa [117]. For example, the expression of the mexAB-oprM operon is negatively controlled by several regulatory loci, such as mexR, nalD, nalB, and nalC. Mutations in these loci can result in the overproduction of the MexAB-OprM efflux pump complex, leading to increased resistance to several antibiotics [92,96,117,118]. Similarly, the overexpression of MexXY-OprM, induced by mutations in mexZ, results in P. aeruginosa’s resistance to aminoglycosides, β-lactams, and fluoroquinolones [119,120,121].
Finally, mutations in genes coding for DNA gyrase (gyrA and gyrB) and topoisomerase IV (parC and parE) are associated with P. aeruginosa’s reduced susceptibility to quinolones. These mutations may lead to modifications of the target sites of these antibiotics, decreasing their binding affinity [122,123].
Bacteria can acquire exogenous DNA through three principal mechanisms: conjugation, in which DNA is transferred through direct cell-to-cell contact; transduction, in which bacteriophages are responsible for gene transfer between two bacterial cells; and transformation, which occurs when bacteria incorporate free DNA fragments present in the surrounding environment [12,124]. Different resistance genes can be present on plasmids, transposons, integrons, and prophages and can be transferred between bacteria belonging to the same or to different species [124,125]. For example, Liu et al. [126] have found evidence in E. coli of a plasmid-containing mcr-1 gene, responsible for resistance to colistin, a last-resort antibiotic for the treatment of infections promoted by carbapenem-resistant bacteria. Additionally, the authors observed that this plasmid could be mobilized into Klebsiella pneumoniae and P. aeruginosa via conjugation [126,127]. The fact that plasmids can be transferred among unrelated Gram-negative bacteria genera is of great concern, as it allows the quick spread of resistant traits between bacterial species associated with difficult-to-treat infections [127,128].
4.3. Adaptative Resistance
Adaptive resistance is a mechanism used by bacteria to temporarily increase their ability to resist the effects of antibiotics or other stressors. It involves changes in gene and protein expression in response to environmental stimuli. However, this type of resistance is usually reversible when the environmental conditions become favorable [12,67].
In P. aeruginosa, the formation of biofilms and of persister cells are examples of those strategies. Overall, biofilm formation is influenced by the autoinducer C4HSL from Rhl QS [46,73] and by eDNA released by the Pqs system [46,77]. Moreover, LPS [46,49], flagellum and fimbriae [45,49,51], pili [46,47], the secretion systems T2SS and T5SS [44,55,58], and exopolysaccharides such as alginate, Psl, and Pel [68,69] have also been associated with biofilm formation.
Biofilms are a significant problem for both human and veterinary medicine, since biofilm-associated bacteria are protected from the action of antimicrobials and disinfectants, and also from the action of the host immune system [3]. Biofilms, the main form of bacterial growth, are ubiquitous complex structures irreversibly attached to surfaces, consisting of an interactive polymicrobial community embedded in a self-produced matrix of polymeric extracellular substances, composed by polysaccharides, proteins, and eDNA [3,33,129]. These bacterial communities are responsible for more than 50% of nosocomial infections and are recalcitrant to most antimicrobials, due to several reasons, including the incapacity of these compounds to penetrate the biofilm matrix; the sequestering of antimicrobials within the biofilm structure; the metabolic latency of the bacteria present in the inner layers of the biofilm, due to poor nutrient and oxygen availability; the establishment of communication systems between biofilm-based bacteria via autoinducer expression; the increased rate of antimicrobial resistance gene transfer between biofilm bacteria; and the presence of high numbers of persister cells [130,131]. These are a small subpopulation of cells present within a biofilm that spontaneously enter a quiescent state, displaying no signs of cell division [130]. This bacterial phenotype corresponds to metabolically inactive bacteria that have entered in a dormant state, which allows them to survive prolonged exposure to high doses of antimicrobials [132,133]. Persister formation results from changes in gene expression, being triggered by stress factors, including exposure to antimicrobials; when the antimicrobial is removed from the environment, these cells recover their metabolic activity and switch back to their antimicrobial-susceptible wildtype [130,134,135].
These phenotypes allow P. aeruginosa to resist disinfection treatments, contributing to its persistence and survival in the hospital setting [136]. Indeed, studies have reported the high resistance ability of P. aeruginosa biofilms to several biocides, including quaternary ammonium compounds, chlorine, and aldehydes [38,136,137].
Moreover, P. aeruginosa’s ability to survive in the hospital environment has also been associated with its ability to form biofilms that can effectively colonize inert materials, such as medical equipment and devices, including urinary catheters and implants, and that have been isolated from different types of hospital surfaces, such as faucets and water systems [9,11,28,29,30,31,32,138,139,140]. P. aeruginosa is qualified as an “Opportunistic Premise Plumbing Pathogen” due to its high capacity to colonize and endure within hospital water networks [141]. Hence, biofilms present in such environments can be a reservoir of opportunistic strains, including P. aeruginosa [142,143]. This bacterium can survive different water treatments, including sub-chlorination, application of antibiotics and biocides, and thermal shock [142,144]. P. aeruginosa is well adapted to the adverse conditions promoted by these treatments due to its resistance mechanisms. For example, resistance to biocides has been previously associated with low membrane permeability, efflux pump expression, horizontal gene transfer of resistance genes, bacterial genomic plasticity, and the occurrence of adaptative mutations [142,145].
The emergence of MDR and XDR clinical isolates associated with difficult-to-treat infections, such as the epidemic P. aeruginosa high-risk clones ST235, ST111, ST233, ST244, ST357, ST308, ST175, ST277, ST654, and ST298 [146], has already been reported in different hospitals and represents a worldwide problem [145]. According to SENTRY, P. aeruginosa’s resistance to piperacillin–tazobactam and meropenem between 2019 and 2021 was similar in Western Europe and the United States (23%) and higher in Eastern Europe in patients hospitalized with pneumonia (34.7%) [147].
One of the most important measures to control the dissemination of nosocomial pathogens in hospitals is to avoid hand contact with surfaces, which should be frequently disinfected [139,148].
5. Biocide Tolerance
Environmental contamination in healthcare facilities has been thoroughly studied and related to the transmission of nosocomial infections [149,150,151,152,153,154,155,156,157,158,159,160,161,162,163,164,165]. Therefore, cleaning and disinfection are essential for limiting pathogen spread in hospital settings, with chemical disinfectants being the preferred biocides to be applied for this purpose [166,167,168,169,170].
Biocides have a multitarget antimicrobial activity that is usually independent of the metabolic state of the bacteria, allowing them to be effective against both active and dormant cells [171,172]. However, their excessive and improper use may result in biocide tolerance or even in antimicrobial cross-resistance [173,174,175,176,177,178,179,180,181,182]. The effectiveness of biocides can be impaired by several different factors, including those related to the biocide’s chemical properties, such as its concentration, pH, and composition, and those related to the conditions in which it is applied, such as the environmental temperature, presence of organic matter, and contact time [175,176,183]. Additionally, the antibiotic resistance traits displayed by bacteria, either inherent or acquired, can also influence their susceptibility to biocides. Disinfectants differ from antibiotics in their mode of action, which is usually non-specific, and target different processes or sites within bacterial cells, rendering these chemicals very effective.
As already discussed in this review, P. aeruginosa is equipped with a plethora of antimicrobial tolerance mechanisms, and the overexpression of efflux pumps is frequently associated with this problem [184,185,186,187]. In fact, efflux-pump-mediated resistance towards different classes of biocides, including phenolic compounds [188,189,190,191,192,193,194], cationic biocides [185,195,196,197,198,199,200], alkylating agents [201,202,203,204], and oxidizing compounds [173,205] has already been described.
In P. aeruginosa, the mexAB-oprM, mexCD-oprJ, and mexEF-oprN genes are among the most studied regulators of efflux pumps associated with tolerance to biocides [186,193,206]. The qac genes, especially qacE and qacEΔ1, coded in plasmids and integrons, are frequently associated with resistance to biocides, especially to quaternary ammonium compounds, and have already been described in P. aeruginosa, including in both clinical and environmental strains [200,207,208,209,210,211].
The possible correlation between biocide tolerance and antibiotic resistance has been a matter of concern in the scientific community [173,184,193]. This phenomenon could result in surface disinfection failures, leading to the spread of pathogens capable of resisting both antibiotics and disinfectants used in healthcare settings. In fact, this problem can be due to co-resistance, observed when biocide tolerance and antibiotic resistance genes are located on the same mobile genetic element [212,213], or to cross-resistance, which occurs when the same mechanism is responsible for both antibiotic resistance and biocide tolerance, as observed, for example, due to the expression of efflux pumps and changes in the outer membrane permeability [213,214,215,216].
In 2010, Mc Cay et al. [217] showed that P. aeruginosa’s continuous exposure to subinhibitory concentrations of benzalkonium chloride resulted in its increased tolerance to this chemical agent but also contributed to a significant increase in its resistance to fluoroquinolones through mutations in gyrA. Tandukar et al. [218] demonstrated that such exposure is also associated with increased resistance to other clinically relevant antibiotics, such as penicillin G and tetracycline.
Overall, selective pressure due to the repeated exposure of bacteria to subinhibitory concentrations of biocides seems to play a role in the emergence of antibiotic resistance, although further research is needed to completely understand its implications in co- and cross-resistance development [171].
6. Conclusions
P. aeruginosa is a highly resilient pathogen that not only possesses a wide variety of intrinsic mechanisms of defense against external aggressions but is also able to develop new strategies to survive. It seems that the virulence factors expressed by this bacterial species contribute not only to its pathogenicity but also to its high ability to adapt to different external aggressors, such as antibiotics and biocides, and to colonize inert materials, allowing P. aeruginosa to thrive in the adverse conditions observed in hospital settings. This bacterium is widely known for its multi-resistant profile towards several antibiotics, including last-generation compounds, rendering it a major concern for public health.
Additionally, biocide tolerance and possibly co- and cross-resistance to antibiotics have also been reported in P. aeruginosa. These phenomena seem to be related to the continuous exposure to subinhibitory concentrations of biocides, which frequently occurs due to their daily use in different settings, including in healthcare, industrial, and domestic facilities. All these aspects promote a selective pressure towards highly resistant strains that will continue to prevail in the environment and consequently be transmitted to different hosts.
Therefore, it is urgent to continue investing in scientific research in this field, in order to better understand P. aeruginosa’s resistance mechanisms and, consequently, to develop new control strategies against this pathogen, aiming to impair its dissemination in hospital environments.
Author Contributions
Conceptualization, C.V., I.S., L.T., S.G. and M.O.; formal analysis, C.V., I.S., L.T., S.G. and M.O.; investigation, C.V.; resources, L.T., S.G. and M.O.; writing—original draft preparation, C.V.; writing—review and editing, I.S., L.T., S.G. and M.O.; project administration, M.O. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Behzadi P., Baráth Z., Gajdács M. It’s Not Easy Being Green: A Narrative Review on the Microbiology, Virulence and Therapeutic Prospects of Multidrug-Resistant Pseudomonas aeruginosa. Antibiotics. 2021;10:42. doi: 10.3390/antibiotics10010042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.LaBauve A.E., Wargo M.J. Growth and Laboratory Maintenance of Pseudomonas aeruginosa. Curr. Protoc. Microbiol. 2012;25:6E.1.1–6E.1.8. doi: 10.1002/9780471729259.mc06e01s25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Thi M.T.T., Wibowo D., Rehm B.H. Pseudomonas aeruginosa Biofilms. Int. J. Mol. Sci. 2020;21:8671. doi: 10.3390/ijms21228671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chevalier S., Bouffartigues E., Bodilis J., Maillot O., Lesouhaitier O., Feuilloley M.G.J., Orange N., Dufour A., Cornelis P. Structure, function and regulation of Pseudomonas aeruginosa porins. FEMS Microbiol. Rev. 2017;41:698–722. doi: 10.1093/femsre/fux020. [DOI] [PubMed] [Google Scholar]
- 5.Azam M.W., Khan A.U. Updates on the pathogenicity status of Pseudomonas aeruginosa. Drug Discov. Today. 2018;24:350–359. doi: 10.1016/j.drudis.2018.07.003. [DOI] [PubMed] [Google Scholar]
- 6.Botelho J., Grosso F., Peixe L. Antibiotic resistance in Pseudomonas aeruginosa—Mechanisms, epidemiology and evolution. Drug Resist. Updat. 2019;44:100640. doi: 10.1016/j.drup.2019.07.002. [DOI] [PubMed] [Google Scholar]
- 7.Griffith S.J., Nathan C., Selander R.K., Chamberlin W., Gordon S., Kabins S., Weinstein R.A. The Epidemiology of Pseudomonas aeruginosa in Oncology Patients in a General Hospital. J. Infect. Dis. 1989;160:1030–1036. doi: 10.1093/infdis/160.6.1030. [DOI] [PubMed] [Google Scholar]
- 8.Centers for Disease Control and Prevention Pseudomonas aeruginosa in Healthcare Settings. [(accessed on 30 January 2023)]; Available online: https://www.cdc.gov/hai/organisms/pseudomonas.html.
- 9.Coppry M., Leroyer C., Saly M., Venier A.-G., Slekovec C., Bertrand X., Parer S., Alfandari S., Cambau E., Megarbane B., et al. Exogenous acquisition of Pseudomonas aeruginosa in intensive care units: A prospective multi-centre study (DYNAPYO study) J. Hosp. Infect. 2020;104:40–45. doi: 10.1016/j.jhin.2019.08.008. [DOI] [PubMed] [Google Scholar]
- 10.Sousa A. Master’s Thesis. Faculdade de Medicina Veterinária-Universidade de Lisboa; Lisbon, Portugal: 2021. Avaliação In Vitro de Estratégicas Antimicrobianas Inovadoras Para Controlo de Otites Externas Por Pseudomonas Aeruginosa No Cão. [Google Scholar]
- 11.Costa D., Bousseau A., Thevenot S., Dufour X., Laland C., Burucoa C., Castel O. Nosocomial outbreak of Pseudomonas aeruginosa associated with a drinking water fountain. J. Hosp. Infect. 2015;91:271–274. doi: 10.1016/j.jhin.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 12.Pang Z., Raudonis R., Glick B.R., Lin T.-J., Cheng Z. Antibiotic Resistance in Pseudomonas Aeruginosa: Mechanisms and Alternative Therapeutic Strategies. Biotechnol. Adv. 2019;37:177–192. doi: 10.1016/j.biotechadv.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 13.Petrov V., Tsachev I., Georgiev G.Z., Marutsov P. Otitis Externa in Dogs: Microbiology and Antimicrobial Susceptibility. Rev. Med. Vet. 2013;164:18–22. [Google Scholar]
- 14.Pye C. Pseudomonas otitis externa in dogs. Can. Vet. J. 2018;59:1231–1234. [PMC free article] [PubMed] [Google Scholar]
- 15.Fine D.M., Tobias A.H. Cardiovascular Device Infections in Dogs: Report of 8 Cases and Review of The Literature. J. Vet. Intern. Med. 2007;21:1265–1271. doi: 10.1111/j.1939-1676.2007.tb01948.x. [DOI] [PubMed] [Google Scholar]
- 16.Bubenik L.J., Hosgood G.L., Waldron D.R., Snow L.A. Frequency of urinary tract infection in catheterized dogs and comparison of bacterial culture and susceptibility testing results for catheterized and noncatheterized dogs with urinary tract infections. J. Am. Vet. Med. Assoc. 2007;231:893–899. doi: 10.2460/javma.231.6.893. [DOI] [PubMed] [Google Scholar]
- 17.Eugster S., Schawalder P., Gaschen F., Boerlin P. A Prospective Study of Postoperative Surgical Site Infections in Dogs and Cats. Vet. Surg. 2004;33:542–550. doi: 10.1111/j.1532-950X.2004.04076.x. [DOI] [PubMed] [Google Scholar]
- 18.Nuttall T., Cole L.K. Evidence-based veterinary dermatology: A systematic review of interventions for treatment of Pseudomonas otitis in dogs. Vet. Dermatol. 2007;18:69–77. doi: 10.1111/j.1365-3164.2007.00575.x. [DOI] [PubMed] [Google Scholar]
- 19.Hamilton E., Kruger J.M., Schall W., Beal M., Manning S.D., Kaneene J.B. Acquisition and persistence of antimicrobial-resistant bacteria isolated from dogs and cats admitted to a veterinary teaching hospital. J. Am. Vet. Med. Assoc. 2013;243:990–1000. doi: 10.2460/javma.243.7.990. [DOI] [PubMed] [Google Scholar]
- 20.Hayashi W., Izumi K., Yoshida S., Takizawa S., Sakaguchi K., Iyori K., Minoshima K.-I., Takano S., Kitagawa M., Nagano Y., et al. Antimicrobial Resistance and Type III Secretion System Virulotypes of Pseudomonas aeruginosa Isolates from Dogs and Cats in Primary Veterinary Hospitals in Japan: Identification of the International High-Risk Clone Sequence Type 235. Microbiol. Spectr. 2021;9:e00408-21. doi: 10.1128/Spectrum.00408-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Eliasi U.L., Sebola D., Oguttu J.W., Qekwana D.N. Antimicrobial resistance patterns of Pseudomonas aeruginosa isolated from canine clinical cases at a veterinary academic hospital in South Africa. J. S. Afr. Vet. Assoc. 2020;91:6. doi: 10.4102/jsava.v91i0.2052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gentilini F., Turba M.E., Pasquali F., Mion D., Romagnoli N., Zambon E., Terni D., Peirano G., Pitout J.D.D., Parisi A., et al. Hospitalized Pets as a Source of Carbapenem-Resistance. Front. Microbiol. 2018;9:2872. doi: 10.3389/fmicb.2018.02872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abraham S., Wong H.S., Turnidge J., Johnson J.R., Trott D.J. Carbapenemase-producing bacteria in companion animals: A public health concern on the horizon. J. Antimicrob. Chemother. 2014;69:1155–1157. doi: 10.1093/jac/dkt518. [DOI] [PubMed] [Google Scholar]
- 24.Haenni M., Bour M., Châtre P., Madec J.-Y., Plésiat P., Jeannot K. Resistance of Animal Strains of Pseudomonas aeruginosa to Carbapenems. Front. Microbiol. 2017;8:1847. doi: 10.3389/fmicb.2017.01847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wood S.J., Kuzel T.M., Shafikhani S.H. Pseudomonas aeruginosa: Infections, Animal Modeling, and Therapeutics. Cells. 2023;12:199. doi: 10.3390/cells12010199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.da Saúde D.-G. Programa de Prevenção e Controlo de Infeções e de Resistência aos Antimicrobianos. DGS; Lisboa, Portugal: 2017. Prevenção da Transmissão de Enterobacteriáceas Resistentes Aos Carbapenemos em Hospitais de Cuidados de Agudos; p. 20. [Google Scholar]
- 27.World Health Organization Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics. [(accessed on 1 February 2023)]; Available online: https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf?ua=1.
- 28.Trautmann M., Lepper P.M., Haller M. Ecology of Pseudomonas aeruginosa in the intensive care unit and the evolving role of water outlets as a reservoir of the organism. Am. J. Infect. Control. 2005;33:S41–S49. doi: 10.1016/j.ajic.2005.03.006. [DOI] [PubMed] [Google Scholar]
- 29.Tran-Dinh A., Neulier C., Amara M., Nebot N., Troché G., Breton N., Zuber B., Cavelot S., Pangon B., Bedos J., et al. Impact of intensive care unit relocation and role of tap water on an outbreak of Pseudomonas aeruginosa expressing OprD-mediated resistance to imipenem. J. Hosp. Infect. 2018;100:e105–e114. doi: 10.1016/j.jhin.2018.05.016. [DOI] [PubMed] [Google Scholar]
- 30.Asghari F.B., Nikaeen M., Mirhendi H. Rapid monitoring of Pseudomonas aeruginosa in hospital water systems: A key priority in prevention of nosocomial infection. FEMS Microbiol. Lett. 2013;343:77–81. doi: 10.1111/1574-6968.12132. [DOI] [PubMed] [Google Scholar]
- 31.Loveday H., Wilson J., Kerr K., Pitchers R., Walker J., Browne J. Association between healthcare water systems and Pseudomonas aeruginosa infections: A rapid systematic review. J. Hosp. Infect. 2014;86:7–15. doi: 10.1016/j.jhin.2013.09.010. [DOI] [PubMed] [Google Scholar]
- 32.Walker J., Moore G. Pseudomonas aeruginosa in hospital water systems: Biofilms, guidelines, and practicalities. J. Hosp. Infect. 2014;89:324–327. doi: 10.1016/j.jhin.2014.11.019. [DOI] [PubMed] [Google Scholar]
- 33.Sindeldecker D., Stoodley P. The many antibiotic resistance and tolerance strategies of Pseudomonas aeruginosa. Biofilm. 2021;3:100056. doi: 10.1016/j.bioflm.2021.100056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Geraldes C., Verdial C., Cunha E., Almeida V., Tavares L., Oliveira M., Gil S. Evaluation of a Biocide Used in the Biological Isolation and Containment Unit of a Veterinary Teaching Hospital. Antibiotics. 2021;10:639. doi: 10.3390/antibiotics10060639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mann R., Holmes A., McNeilly O., Cavaliere R., Sotiriou G.A., Rice S.A., Gunawan C. Evolution of biofilm-forming pathogenic bacteria in the presence of nanoparticles and antibiotic: Adaptation phenomena and cross-resistance. J. Nanobiotechnol. 2021;19:291. doi: 10.1186/s12951-021-01027-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vijayakumar R., Sandle T., Al-Aboody M.S., AlFonaisan M.K., Alturaiki W., Mickymaray S., Premanathan M., Alsagaby S.A. Distribution of biocide resistant genes and biocides susceptibility in multidrug-resistant Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumannii—A first report from the Kingdom of Saudi Arabia. J. Infect. Public Health. 2018;11:812–816. doi: 10.1016/j.jiph.2018.05.011. [DOI] [PubMed] [Google Scholar]
- 37.Goodarzi R., Yousefimashouf R., Taheri M., Nouri F., Asghari B. Susceptibility to biocides and the prevalence of biocides resistance genes in clinical multidrug-resistant Pseudomonas aeruginosa isolates from Hamadan, Iran. Mol. Biol. Rep. 2021;48:5275–5281. doi: 10.1007/s11033-021-06533-4. [DOI] [PubMed] [Google Scholar]
- 38.Smith K., Hunter I.S. Efficacy of common hospital biocides with biofilms of multi-drug resistant clinical isolates. J. Med. Microbiol. 2008;57:966–973. doi: 10.1099/jmm.0.47668-0. [DOI] [PubMed] [Google Scholar]
- 39.Silby M.W., Winstanley C., Godfrey S.A.C., Levy S.B., Jackson R.W. Pseudomonasgenomes: Diverse and adaptable. FEMS Microbiol. Rev. 2011;35:652–680. doi: 10.1111/j.1574-6976.2011.00269.x. [DOI] [PubMed] [Google Scholar]
- 40.Stover C.K., Pham X.Q., Erwin A.L., Mizoguchi S.D., Warrener P., Hickey M.J., Brinkman F.S.L., Hufnagle W.O., Kowalik D.J., Lagrou M., et al. Complete genome sequence of Pseudomonas aeruginosa PAO1, an opportunistic pathogen. Nature. 2000;406:959–964. doi: 10.1038/35023079. [DOI] [PubMed] [Google Scholar]
- 41.Eklockgether J., Ecramer N., Ewiehlmann L., Davenport C.F., Etümmler B. Pseudomonas aeruginosa Genomic Structure and Diversity. Front. Microbiol. 2011;2:150. doi: 10.3389/fmicb.2011.00150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.de Sousa T., Hébraud M., Dapkevicius M.L.N.E., Maltez L., Pereira J.E., Capita R., Alonso-Calleja C., Igrejas G., Poeta P. Genomic and Metabolic Characteristics of the Pathogenicity in Pseudomonas aeruginosa. Int. J. Mol. Sci. 2021;22:12892. doi: 10.3390/ijms222312892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Spagnolo A.M., Sartini M., Cristina M.L. Pseudomonas aeruginosa in the healthcare facility setting. Rev. Med. Microbiol. 2021;32:169–175. doi: 10.1097/MRM.0000000000000271. [DOI] [Google Scholar]
- 44.Qin S., Xiao W., Zhou C., Pu Q., Deng X., Lan L., Liang H., Song X., Wu M. Pseudomonas aeruginosa: Pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduct. Target. Ther. 2022;7:199. doi: 10.1038/s41392-022-01056-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.O’Toole G.A., Kolter R. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol. Microbiol. 1998;30:295–304. doi: 10.1046/j.1365-2958.1998.01062.x. [DOI] [PubMed] [Google Scholar]
- 46.Jurado-Martín I., Sainz-Mejías M., McClean S. Pseudomonas aeruginosa: An Audacious Pathogen with an Adaptable Arsenal of Virulence Factors. Int. J. Mol. Sci. 2021;22:3128. doi: 10.3390/ijms22063128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liao C., Huang X., Wang Q., Yao D., Lu W. Virulence Factors of Pseudomonas Aeruginosa and Antivirulence Strategies to Combat Its Drug Resistance. Front. Cell. Infect. Microbiol. 2022;12:926758. doi: 10.3389/fcimb.2022.926758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rocha A.J., Barsottini M.R.D.O., Rocha R.R., Laurindo M.V., De Moraes F.L.L., Da Rocha S.L. Pseudomonas Aeruginosa: Virulence Factors and Antibiotic Resistance Genes. Braz. Arch. Biol. Technol. 2019;62:e19180503. doi: 10.1590/1678-4324-2019180503. [DOI] [Google Scholar]
- 49.King J.D., Kocíncová D., Westman E.L., Lam J.S. Lipopolysaccharide biosynthesis in Pseudomonas aeruginosa. Innate Immun. 2009;15:261–312. doi: 10.1177/1753425909106436. [DOI] [PubMed] [Google Scholar]
- 50.Park W.S., Lee J., Na G., Park S., Seo S.-K., Choi J.S., Jung W.-K., Choi I.-W. Benzyl Isothiocyanate Attenuates Inflammasome Activation in Pseudomonas aeruginosa LPS-Stimulated THP-1 Cells and Exerts Regulation through the MAPKs/NF-κB Pathway. Int. J. Mol. Sci. 2022;23:1228. doi: 10.3390/ijms23031228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ozer E., Yaniv K., Chetrit E., Boyarski A., Meijler M.M., Berkovich R., Kushmaro A., Alfonta L. An inside look at a biofilm: Pseudomonas aeruginosa flagella biotracking. Sci. Adv. 2021;7:eabg8581. doi: 10.1126/sciadv.abg8581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sabnis A., Hagart K.L., Klöckner A., Becce M., Evans L.E., Furniss R.C.D., Mavridou D.A., Murphy R., Stevens M.M., Davies J.C., et al. Colistin kills bacteria by targeting lipopolysaccharide in the cytoplasmic membrane. Elife. 2021;10:e65836. doi: 10.7554/eLife.65836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hancock R.E.W., Brinkman F.S.L. Function of Pseudomonas Porins in Uptake and Efflux. Annu. Rev. Microbiol. 2002;56:17–38. doi: 10.1146/annurev.micro.56.012302.160310. [DOI] [PubMed] [Google Scholar]
- 54.Diggle S.P., Whiteley M. Microbe Profile: Pseudomonas aeruginosa: Opportunistic pathogen and lab rat. Microbiology. 2020;166:30–33. doi: 10.1099/mic.0.000860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Meuskens I., Saragliadis A., Leo J.C., Linke D. Type V Secretion Systems: An Overview of Passenger Domain Functions. Front. Microbiol. 2019;10:1163. doi: 10.3389/fmicb.2019.01163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tammam S., Sampaleanu L.M., Koo J., Manoharan K., Daubaras M., Burrows L.L., Howell P.L. PilMNOPQ from the Pseudomonas aeruginosa Type IV Pilus System Form a Transenvelope Protein Interaction Network That Interacts with PilA. J. Bacteriol. 2013;195:2126–2135. doi: 10.1128/JB.00032-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Juhas M., Crook D.W., Hood D.W. Type IV secretion systems: Tools of bacterial horizontal gene transfer and virulence. Cell. Microbiol. 2008;10:2377–2386. doi: 10.1111/j.1462-5822.2008.01187.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Filloux A. Protein Secretion Systems in Pseudomonas aeruginosa: An Essay on Diversity, Evolution, and Function. Front. Microbiol. 2011;2:155. doi: 10.3389/fmicb.2011.00155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Pena R.T., Blasco L., Ambroa A., González-Pedrajo B., Fernández-García L., López M., Bleriot I., Bou G., García-Contreras R., Wood T.K., et al. Relationship between Quorum Sensing and Secretion Systems. Front. Microbiol. 2019;10:1100. doi: 10.3389/fmicb.2019.01100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Green E.R., Mecsas J. Bacterial Secretion Systems: An Overview. Microbiol. Spectr. 2016;4:213–239. doi: 10.1128/microbiolspec.VMBF-0012-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cascales E., Christie P.J. The versatile bacterial type IV secretion systems. Nat. Rev. Microbiol. 2003;1:137–149. doi: 10.1038/nrmicro753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pukatzki S., Ma A.T., Revel A.T., Sturtevant D., Mekalanos J.J. Type VI secretion system translocates a phage tail spike-like protein into target cells where it cross-links actin. Proc. Natl. Acad. Sci. USA. 2007;104:15508–15513. doi: 10.1073/pnas.0706532104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Russell A.B., Peterson S.B., Mougous J.D. Type VI secretion system effectors: Poisons with a purpose. Nat. Rev. Microbiol. 2014;12:137–148. doi: 10.1038/nrmicro3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hall S., McDermott C., Anoopkumar-Dukie S., McFarland A.J., Forbes A., Perkins A.V., Davey A.K., Chess-Williams R., Kiefel M.J., Arora D., et al. Cellular Effects of Pyocyanin, a Secreted Virulence Factor of Pseudomonas aeruginosa. Toxins. 2016;8:236. doi: 10.3390/toxins8080236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lau G.W., Hassett D.J., Ran H., Kong F. The role of pyocyanin in Pseudomonas aeruginosa infection. Trends Mol. Med. 2004;10:599–606. doi: 10.1016/j.molmed.2004.10.002. [DOI] [PubMed] [Google Scholar]
- 66.Hilliam Y., Kaye S., Winstanley C. Pseudomonas aeruginosa and microbial keratitis. J. Med. Microbiol. 2020;69:3–13. doi: 10.1099/jmm.0.001110. [DOI] [PubMed] [Google Scholar]
- 67.Moradali M.F., Ghods S., Rehm B.H.A. Pseudomonas aeruginosa Lifestyle: A Paradigm for Adaptation, Survival, and Persistence. Front. Cell. Infect. Microbiol. 2017;7:39. doi: 10.3389/fcimb.2017.00039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Franklin M.J., Nivens D.E., Weadge J.T., Howell P.L. Biosynthesis of the Pseudomonas aeruginosa Extracellular Polysaccharides, Alginate, Pel, and Psl. Front. Microbiol. 2011;2:167. doi: 10.3389/fmicb.2011.00167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ghafoor A., Hay I.D., Rehm B.H.A. Role of Exopolysaccharides in Pseudomonas aeruginosa Biofilm Formation and Architecture. Appl. Environ. Microbiol. 2011;77:5238–5246. doi: 10.1128/AEM.00637-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lee J., Wu J., Deng Y., Wang J., Wang C., Wang J., Chang C., Dong Y., Williams P., Zhang L.-H. A cell-cell communication signal integrates quorum sensing and stress response. Nat. Chem. Biol. 2013;9:339–343. doi: 10.1038/nchembio.1225. [DOI] [PubMed] [Google Scholar]
- 71.Alfiniyah C., Bees M.A., Wood A.J. Quorum machinery: Effect of the las system in rhl regulation of P. aeruginosa. AIP Conf. Proc. 2019;2192:060001. doi: 10.1063/1.5139147. [DOI] [Google Scholar]
- 72.Lee J., Zhang L. The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell. 2015;6:26–41. doi: 10.1007/s13238-014-0100-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kariminik A., Baseri-Salehi M., Kheirkhah B. Pseudomonas aeruginosa quorum sensing modulates immune responses: An updated review article. Immunol. Lett. 2017;190:1–6. doi: 10.1016/j.imlet.2017.07.002. [DOI] [PubMed] [Google Scholar]
- 74.Möker N., Dean C.R., Tao J. Pseudomonas aeruginosa Increases Formation of Multidrug-Tolerant Persister Cells in Response to Quorum-Sensing Signaling Molecules. J. Bacteriol. 2010;192:1946–1955. doi: 10.1128/JB.01231-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sana T.G., Hachani A., Bucior I., Soscia C., Garvis S., Termine E., Engel J., Filloux A., Bleves S. The Second Type VI Secretion System of Pseudomonas aeruginosa Strain PAO1 Is Regulated by Quorum Sensing and Fur and Modulates Internalization in Epithelial Cells. J. Biol. Chem. 2012;287:27095–27105. doi: 10.1074/jbc.M112.376368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lin J., Cheng J., Wang Y., Shen X. The Pseudomonas Quinolone Signal (PQS): Not Just for Quorum Sensing Anymore. Front. Cell. Infect. Microbiol. 2018;8:230. doi: 10.3389/fcimb.2018.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.García-Reyes S., Soberón-Chávez G., Cocotl-Yanez M. The third quorum-sensing system of Pseudomonas aeruginosa: Pseudomonas quinolone signal and the enigmatic PqsE protein. J. Med. Microbiol. 2020;69:25–34. doi: 10.1099/jmm.0.001116. [DOI] [PubMed] [Google Scholar]
- 78.Wang J., Wang C., Yu H.B., Dela Ahator S., Wu X., Lv S., Zhang L.H. Bacterial quorum-sensing signal IQS induces host cell apoptosis by targeting POT1–p53 signalling pathway. Cell. Microbiol. 2019;21:e13076. doi: 10.1111/cmi.13076. [DOI] [PubMed] [Google Scholar]
- 79.Zarb P., Coignard B., Griskeviciene J., Muller A., Vankerckhoven V., Weist K., Goossens M.M., Vaerenberg S., Hopkins S., Catry B., et al. The European Centre for Disease Prevention and Control (ECDC) pilot point prevalence survey of healthcare-associated infections and antimicrobial use. Eurosurveillance. 2012;17:20316. doi: 10.2807/ese.17.46.20316-en. [DOI] [PubMed] [Google Scholar]
- 80.European Centre for Disease Prevention and Control Healthcare-Associated Infections—A Threat to Patient Safety in Europe. [(accessed on 20 January 2023)]; Available online: https://www.ecdc.europa.eu/sites/default/files/documents/healthcare-associated-infections-threat-patient-safety-infographic.pdf.
- 81.Lin M.Y., Weinstein R.A., Hayden M.K. Multidrug-Resistant Organisms: Epidemiology and Control. In: Jarvis W.R., editor. Bennett & Brachman’s Hospital Infections. Lippincott Williams & Wilkins; Philadelphia, PA, USA: 2014. pp. 181–207. [Google Scholar]
- 82.Bengtsson B., Greko C. Antibiotic resistance—Consequences for animal health, welfare, and food production. Upsala J. Med. Sci. 2014;119:96–102. doi: 10.3109/03009734.2014.901445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Magiorakos A.-P., Srinivasan A., Carey R.B., Carmeli Y., Falagas M.E., Giske C.G., Harbarth S., Hindler J.F., Kahlmeter G., Olsson-Liljequist B., et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012;18:268–281. doi: 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 84.European Centre for Disease Prevention and Control . Antimicrobial Resistance Surveillance in Europe 2022–2020 Data. European Centre for Disease Prevention and Control; Copenhagen, Denmark: 2022. [Google Scholar]
- 85.Quinn P.J., Markey B.K., Leonard F.C., FitzPatrick E.S., Fanning S., Hartigan P.J. Veterinary Microbiology and Microbial Disease. Wiley-Blackwell; Chichester, UK: 2011. Antibacterial Resistance. [Google Scholar]
- 86.Boerlin P., White D.G. Antimicrobial Resistance and Its Epidemiology. In: Giguère S., Prescott J.F., Dowling P.M., editors. Antimicrobial Therapy in Veterinary Medicine. Wiley-Blackwell; Ames, IA, USA: 2013. pp. 21–40. [Google Scholar]
- 87.Mohr K.I. History of Antibiotics Research. In: Stadler M., Dersch P., editors. How to Overcome the Antibiotic Crisis. Volume 398. Springer; Cham, Switzerland: 2016. pp. 237–272. [DOI] [PubMed] [Google Scholar]
- 88.Munita J.M., Arias C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016;4:464–473. doi: 10.1128/microbiolspec.VMBF-0016-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Li H., Luo Y.-F., Williams B.J., Blackwell T.S., Xie C.-M. Structure and function of OprD protein in Pseudomonas aeruginosa: From antibiotic resistance to novel therapies. Int. J. Med. Microbiol. 2012;302:63–68. doi: 10.1016/j.ijmm.2011.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Macfarlane E.L.A., Kwasnicka A., Ochs M.M., Hancock R.E.W. PhoP-PhoQ homologues in Pseudomonas aeruginosa regulate expression of the outer-membrane protein OprH and polymyxin B resistance. Mol. Microbiol. 1999;34:305–316. doi: 10.1046/j.1365-2958.1999.01600.x. [DOI] [PubMed] [Google Scholar]
- 91.Bell A., Bains M., Hancock R. Pseudomonas aeruginosa outer membrane protein OprH: Expression from the cloned gene and function in EDTA and gentamicin resistance. J. Bacteriol. 1991;173:6657–6664. doi: 10.1128/jb.173.21.6657-6664.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Poole K. Efflux pumps as antimicrobial resistance mechanisms. Ann. Med. 2007;39:162–176. doi: 10.1080/07853890701195262. [DOI] [PubMed] [Google Scholar]
- 93.Piddock L.J.V. Multidrug-resistance efflux pumps? Not just for resistance. Nat. Rev. Genet. 2006;4:629–636. doi: 10.1038/nrmicro1464. [DOI] [PubMed] [Google Scholar]
- 94.Sun J., Deng Z., Yan A. Bacterial multidrug efflux pumps: Mechanisms, physiology and pharmacological exploitations. Biochem. Biophys. Res. Commun. 2014;453:254–267. doi: 10.1016/j.bbrc.2014.05.090. [DOI] [PubMed] [Google Scholar]
- 95.Llanes C., Köhler T., Patry I., Dehecq B., van Delden C., Plésiat P. Role of the MexEF-OprN Efflux System in Low-Level Resistance of Pseudomonas aeruginosa to Ciprofloxacin. Antimicrob. Agents Chemother. 2011;55:5676–5684. doi: 10.1128/AAC.00101-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Masuda N., Sakagawa E., Ohya S., Gotoh N., Tsujimoto H., Nishino T. Substrate Specificities of MexAB-OprM, MexCD-OprJ, and MexXY-OprM Efflux Pumps in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2000;44:3322–3327. doi: 10.1128/AAC.44.12.3322-3327.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Dupont P., Hocquet D., Jeannot K., Chavanet P., Plésiat P. Bacteriostatic and bactericidal activities of eight fluoroquinolones against MexAB-OprM-overproducing clinical strains of Pseudomonas aeruginosa. J. Antimicrob. Chemother. 2005;55:518–522. doi: 10.1093/jac/dki030. [DOI] [PubMed] [Google Scholar]
- 98.Llanes C., Pourcel C., Richardot C., Plesiat P., Fichant G., Cavallo J.-D., Mérens A., Vu-Thien H., Leclercq R., Romaszko J.P., et al. Diversity of -lactam resistance mechanisms in cystic fibrosis isolates of Pseudomonas aeruginosa: A French multicentre study. J. Antimicrob. Chemother. 2013;68:1763–1771. doi: 10.1093/jac/dkt115. [DOI] [PubMed] [Google Scholar]
- 99.Wolter D.J., Lister P.D. Mechanisms of β-lactam Resistance among Pseudomonas aeruginosa. Curr. Pharm. Des. 2013;19:209–222. doi: 10.2174/138161213804070311. [DOI] [PubMed] [Google Scholar]
- 100.Majiduddin F.K., Materon I.C., Palzkill T.G. Molecular analysis of beta-lactamase structure and function. Int. J. Med. Microbiol. 2002;292:127–137. doi: 10.1078/1438-4221-00198. [DOI] [PubMed] [Google Scholar]
- 101.Hagel S., Makarewicz O., Hartung A., Weiß D., Stein C., Brandt C., Schumacher U., Ehricht R., Patchev V., Pletz M.W. ESBL colonization and acquisition in a hospital population: The molecular epidemiology and transmission of resistance genes. PLoS ONE. 2019;14:e0208505. doi: 10.1371/journal.pone.0208505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lemarié C., Legeay C., Lasocki S., Mahieu R., Kouatchet A., Bahier L., Onillon L., Corre M., Kempf M., Eveillard M. Extended-spectrum β-lactamase Enterobacteriaceae (ESBLE) in intensive care units: Strong correlation with the ESBLE colonization pressure in patients but not same species. J. Hosp. Infect. 2020;104:53–56. doi: 10.1016/j.jhin.2019.08.007. [DOI] [PubMed] [Google Scholar]
- 103.Ramirez M.S., Tolmasky M.E. Aminoglycoside modifying enzymes. Drug Resist. Updat. 2010;13:151–171. doi: 10.1016/j.drup.2010.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Poole K. Aminoglycoside Resistance in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2005;49:479–487. doi: 10.1128/AAC.49.2.479-487.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Subedi D., Vijay A.K., Willcox M. Overview of mechanisms of antibiotic resistance in Pseudomonas aeruginosa: An ocular perspective. Clin. Exp. Optom. 2017;101:162–171. doi: 10.1111/cxo.12621. [DOI] [PubMed] [Google Scholar]
- 106.Gellatly S.L., Hancock R.E. Pseudomonas aeruginosa: New insights into pathogenesis and host defenses. Pathog. Dis. 2013;67:159–173. doi: 10.1111/2049-632X.12033. [DOI] [PubMed] [Google Scholar]
- 107.Lynch J.P., Zhanel G.G. Pseudomonas aeruginosa Pneumonia: Evolution of Antimicrobial Resistance and Implications for Therapy. Semin. Respir. Crit. Care Med. 2022;43:191–218. doi: 10.1055/s-0041-1740109. [DOI] [PubMed] [Google Scholar]
- 108.Jeukens J., Freschi L., Kukavica-Ibrulj I., Emond-Rheault J.-G., Tucker N.P., Levesque R.C. Genomics of antibiotic-resistance prediction in Pseudomonas aeruginosa. Ann. N. Y. Acad. Sci. 2017;1435:5–17. doi: 10.1111/nyas.13358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Laborda P., Hernando-Amado S., Martínez J.L., Sanz-García F. Antibiotic Resistance in Pseudomonas. Adv. Exp. Med. Biol. 2022;1386:117–143. doi: 10.1007/978-3-031-08491-1_5. [DOI] [PubMed] [Google Scholar]
- 110.Chatterjee M., Anju C., Biswas L., Kumar V.A., Mohan C.G., Biswas R. Antibiotic resistance in Pseudomonas aeruginosa and alternative therapeutic options. Int. J. Med. Microbiol. 2016;306:48–58. doi: 10.1016/j.ijmm.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 111.Blair J.M.A., Webber M.A., Baylay A.J., Ogbolu D.O., Piddock L.J.V. Molecular mechanisms of antibiotic resistance. Nat. Rev. Microbiol. 2015;13:42–51. doi: 10.1038/nrmicro3380. [DOI] [PubMed] [Google Scholar]
- 112.Henrichfreise B., Wiegand I., Pfister W., Wiedemann B. Resistance Mechanisms of Multiresistant Pseudomonas aeruginosa Strains from Germany and Correlation with Hypermutation. Antimicrob. Agents Chemother. 2007;51:4062–4070. doi: 10.1128/AAC.00148-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Lister P.D., Wolter D.J., Hanson N.D. Antibacterial-Resistant Pseudomonas aeruginosa: Clinical Impact and Complex Regulation of Chromosomally Encoded Resistance Mechanisms. Clin. Microbiol. Rev. 2009;22:582–610. doi: 10.1128/CMR.00040-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Berrazeg M., Jeannot K., Enguéné V.Y.N., Broutin I., Loeffert S., Fournier D., Plésiat P. Mutations in β-Lactamase AmpC Increase Resistance of Pseudomonas aeruginosa Isolates to Antipseudomonal Cephalosporins. Antimicrob. Agents Chemother. 2015;59:6248–6255. doi: 10.1128/AAC.00825-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Tooke C.L., Hinchliffe P., Bragginton E.C., Colenso C.K., Hirvonen V.H.A., Takebayashi Y., Spencer J. β-Lactamases and β-Lactamase Inhibitors in the 21st Century. J. Mol. Biol. 2019;431:3472–3500. doi: 10.1016/j.jmb.2019.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Juan C., Maciá M.D., Gutiérrez O., Vidal C., Pérez J.L., Oliver A. Molecular Mechanisms of β-Lactam Resistance Mediated by AmpC Hyperproduction in Pseudomonas aeruginosa Clinical Strains. Antimicrob. Agents Chemother. 2005;49:4733–4738. doi: 10.1128/AAC.49.11.4733-4738.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Lorusso A.B., Carrara J.A., Barroso C.D.N., Tuon F.F., Faoro H. Role of Efflux Pumps on Antimicrobial Resistance in Pseudomonas aeruginosa. Int. J. Mol. Sci. 2022;23:15779. doi: 10.3390/ijms232415779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Janice J., Agyepong N., Owusu-Ofori A., Govinden U., Essack S.Y., Samuelsen Ø., Sundsfjord A., Pedersen T. Carbapenem Resistance Determinants Acquired through Novel Chromosomal Integrations in Extensively Drug-Resistant Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2021;65:e00289-21. doi: 10.1128/AAC.00289-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Guénard S., Muller C., Monlezun L., Benas P., Broutin I., Jeannot K., Plésiat P. Multiple Mutations Lead to MexXY-OprM-Dependent Aminoglycoside Resistance in Clinical Strains of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2014;58:221–228. doi: 10.1128/AAC.01252-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Hocquet D., Nordmann P., El Garch F., Cabanne L., Plésiat P. Involvement of the MexXY-OprM Efflux System in Emergence of Cefepime Resistance in Clinical Strains of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2006;50:1347–1351. doi: 10.1128/AAC.50.4.1347-1351.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Baum E.Z., Crespo-Carbone S.M., Morrow B.J., Davies T.A., Foleno B.D., He W., Queenan A.M., Bush K. Effect of MexXY Overexpression on Ceftobiprole Susceptibility in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2009;53:2785–2790. doi: 10.1128/AAC.00018-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Bruchmann S., Dötsch A., Nouri B., Chaberny I.F., Häussler S. Quantitative Contributions of Target Alteration and Decreased Drug Accumulation to Pseudomonas aeruginosa Fluoroquinolone Resistance. Antimicrob. Agents Chemother. 2013;57:1361–1368. doi: 10.1128/AAC.01581-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lee J.K., Lee Y.S., Park Y.K., Kim B.S. Alterations in the GyrA and GyrB subunits of topoisomerase II and the ParC and ParE subunits of topoisomerase IV in ciprofloxacin-resistant clinical isolates of Pseudomonas aeruginosa. Int. J. Antimicrob. Agents. 2005;25:290–295. doi: 10.1016/j.ijantimicag.2004.11.012. [DOI] [PubMed] [Google Scholar]
- 124.Arber W. Horizontal Gene Transfer among Bacteria and Its Role in Biological Evolution. Life. 2014;4:217–224. doi: 10.3390/life4020217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Breidenstein E.B., de la Fuente-Núñez C., Hancock R.E. Pseudomonas aeruginosa: All roads lead to resistance. Trends Microbiol. 2011;19:419–426. doi: 10.1016/j.tim.2011.04.005. [DOI] [PubMed] [Google Scholar]
- 126.Liu Y.-Y., Wang Y., Walsh T.R., Yi L.-X., Zhang R., Spencer J., Doi Y., Tian G., Dong B., Huang X., et al. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: A microbiological and molecular biological study. Lancet Infect. Dis. 2016;16:161–168. doi: 10.1016/S1473-3099(15)00424-7. [DOI] [PubMed] [Google Scholar]
- 127.Li B., Yin F., Zhao X., Guo Y., Wang W., Wang P., Zhu H., Yin Y., Wang X. Colistin Resistance Gene mcr-1 Mediates Cell Permeability and Resistance to Hydrophobic Antibiotics. Front. Microbiol. 2020;10:3015. doi: 10.3389/fmicb.2019.03015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Hong D.J., Bae I.K., Jang I.-H., Jeong S.H., Kang H.-K., Lee K. Epidemiology and Characteristics of Metallo-β-Lactamase-Producing Pseudomonas aeruginosa. Infect. Chemother. 2015;47:81–97. doi: 10.3947/ic.2015.47.2.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Tuon F.F., Dantas L.R., Suss P.H., Ribeiro V.S.T. Pathogenesis of the Pseudomonas aeruginosa Biofilm: A Review. Pathogens. 2022;11:300. doi: 10.3390/pathogens11030300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Fisher R.A., Gollan B., Helaine S. Persistent bacterial infections and persister cells. Nat. Rev. Genet. 2017;15:453–464. doi: 10.1038/nrmicro.2017.42. [DOI] [PubMed] [Google Scholar]
- 131.Vatan A., Saltoglu N., Yemisen M., Balkan I.I., Surme S., Demiray T., Mete B., Tabak F. Association between biofilm and multi/extensive drug resistance in diabetic foot infection. Int. J. Clin. Pract. 2018;72:e13060. doi: 10.1111/ijcp.13060. [DOI] [PubMed] [Google Scholar]
- 132.Liu Y., She P., Xu L., Chen L., Li Y., Liu S., Li Z., Hussain Z., Wu Y. Antimicrobial, Antibiofilm, and Anti-persister Activities of Penfluridol against Staphylococcus aureus. Front. Microbiol. 2021;12:727692. doi: 10.3389/fmicb.2021.727692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Scheper H., Wubbolts J.M., Verhagen J.A.M., de Visser A.W., van der Wal R.J.P., Visser L.G., de Boer M.G.J., Nibbering P.H. SAAP-148 Eradicates MRSA Persisters within Mature Biofilm Models Simulating Prosthetic Joint Infection. Front. Microbiol. 2021;12:625952. doi: 10.3389/fmicb.2021.625952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Iii M.C.W., Roe F., Bugnicourt A., Franklin M.J., Stewart P.S. Contributions of Antibiotic Penetration, Oxygen Limitation, and Low Metabolic Activity to Tolerance of Pseudomonas aeruginosa Biofilms to Ciprofloxacin and Tobramycin. Antimicrob. Agents Chemother. 2003;47:317–323. doi: 10.1128/aac.47.1.317-323.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Stewart P.S. Mechanisms of antibiotic resistance in bacterial biofilms. Int. J. Med. Microbiol. 2002;292:107–113. doi: 10.1078/1438-4221-00196. [DOI] [PubMed] [Google Scholar]
- 136.Bridier A., Dubois-Brissonnet F., Greub G., Thomas V., Briandet R. Dynamics of the Action of Biocides in Pseudomonas aeruginosa Biofilms. Antimicrob. Agents Chemother. 2011;55:2648–2654. doi: 10.1128/AAC.01760-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Campanac C., Pineau L., Payard A., Baziard-Mouysset G., Roques C. Interactions between Biocide Cationic Agents and Bacterial Biofilms. Antimicrob. Agents Chemother. 2002;46:1469–1474. doi: 10.1128/AAC.46.5.1469-1474.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Verdial C., Carneiro C., Machado I., Tavares L., Almeida V., Oliveira M., Gil S. Controlling bacteriological contamination of environmental surfaces at the biological isolation and containment unit of a veterinary teaching hospital. Ir. Vet. J. 2021;74:18. doi: 10.1186/s13620-021-00197-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kramer A., Schwebke I., Kampf G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect. Dis. 2006;6:130. doi: 10.1186/1471-2334-6-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lemmen S., Häfner H., Zolldann D., Stanzel S., Lütticken R. Distribution of multi-resistant Gram-negative versus Gram-positive bacteria in the hospital inanimate environment. J. Hosp. Infect. 2004;56:191–197. doi: 10.1016/j.jhin.2003.12.004. [DOI] [PubMed] [Google Scholar]
- 141.Falkinham J., Pruden A., Edwards M. Opportunistic Premise Plumbing Pathogens: Increasingly Important Pathogens in Drinking Water. Pathogens. 2015;4:373–386. doi: 10.3390/pathogens4020373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Virieux-Petit M., Hammer-Dedet F., Aujoulat F., Jumas-Bilak E., Romano-Bertrand S. From Copper Tolerance to Resistance in Pseudomonas aeruginosa towards Patho-Adaptation and Hospital Success. Genes. 2022;13:301. doi: 10.3390/genes13020301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Venier A.-G., Leroyer C., Slekovec C., Talon D., Bertrand X., Parer S., Alfandari S., Guerin J.-M., Megarbane B., Lawrence C., et al. Risk factors for Pseudomonas aeruginosa acquisition in intensive care units: A prospective multicentre study. J. Hosp. Infect. 2014;88:103–108. doi: 10.1016/j.jhin.2014.06.018. [DOI] [PubMed] [Google Scholar]
- 144.Quick J., Cumley N., Wearn C.M., Niebel M., Constantinidou C., Thomas C.M., Pallen M.J., Moiemen N.S., Bamford A., Oppenheim B., et al. Seeking the source of Pseudomonas aeruginosa infections in a recently opened hospital: An observational study using whole-genome sequencing. BMJ Open. 2014;4:e006278. doi: 10.1136/bmjopen-2014-006278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Oliver A., Mulet X., López-Causapé C., Juan C. The increasing threat of Pseudomonas aeruginosa high-risk clones. Drug Resist. Updat. 2015;21–22:41–59. doi: 10.1016/j.drup.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 146.del Barrio-Tofiño E., López-Causapé C., Oliver A. Pseudomonas aeruginosa epidemic high-risk clones and their association with horizontally-acquired β-lactamases: 2020 update. Int. J. Antimicrob. Agents. 2020;56:106196. doi: 10.1016/j.ijantimicag.2020.106196. [DOI] [PubMed] [Google Scholar]
- 147.Losito A.R., Raffaelli F., Del Giacomo P., Tumbarello M. New Drugs for the Treatment of Pseudomonas aeruginosa Infections with Limited Treatment Options: A Narrative Review. Antibiotics. 2022;11:579. doi: 10.3390/antibiotics11050579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Kampf G., Kramer A. Epidemiologic Background of Hand Hygiene and Evaluation of the Most Important Agents for Scrubs and Rubs. Clin. Microbiol. Rev. 2004;17:863–893. doi: 10.1128/CMR.17.4.863-893.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Joseph A. The Impact of the Environment on Infections in Healthcare Facilities. The Center for Health Design; Concord, CA, USA: 2006. [Google Scholar]
- 150.Greene C.E., Weese J.S., Calpin J.P. Environmental Factors in Infectious Disease. In: Greene C.E., editor. Infectious Diseases of the Dog and Cat. Elsevier Saunders; St. Louis, MO, USA: 2012. pp. 1078–1100. [Google Scholar]
- 151.Weber D.J., Anderson D., Rutala W.A. The role of the surface environment in healthcare-associated infections. Curr. Opin. Infect. Dis. 2013;26:338–344. doi: 10.1097/QCO.0b013e3283630f04. [DOI] [PubMed] [Google Scholar]
- 152.Soonthornsit J., Pimwaraluck K., Kongmuang N., Pratya P., Phumthanakorn N. Molecular epidemiology of antimicrobial-resistant Pseudomonas aeruginosa in a veterinary teaching hospital environment. Vet. Res. Commun. 2022;47:73–86. doi: 10.1007/s11259-022-09929-0. [DOI] [PubMed] [Google Scholar]
- 153.Boyce J.M. Environmental contamination makes an important contribution to hospital infection. J. Hosp. Infect. 2007;65:50–54. doi: 10.1016/S0195-6701(07)60015-2. [DOI] [PubMed] [Google Scholar]
- 154.Chaoui L., Mhand R., Mellouki F., Rhallabi N. Contamination of the Surfaces of a Health Care Environment by Multidrug-Resistant (MDR) Bacteria. Int. J. Microbiol. 2019;2019:3236526. doi: 10.1155/2019/3236526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Weese J.S., DaCosta T., Button L., Goth K., Ethier M., Boehnke K. Isolation of Methicillin-Resistant Staphylococcus aureus from the Environment in a Veterinary Teaching Hospital. J. Vet. Intern. Med. 2004;18:468–470. doi: 10.1111/j.1939-1676.2004.tb02568.x. [DOI] [PubMed] [Google Scholar]
- 156.Schimmunech M.S., Lima E.A., Silveira Â.V.B.D.A., de Oliveira A.F., Moreira C.N., de Souza C.M., de Paula E.M.N., Stella A.E. Pseudomonas aeruginosa Isolated from the Environment of a Veterinary Academic Hospital in Brazil-Resistance Profile. Acta Sci. Vet. 2022;50 doi: 10.22456/1679-9216.119471. [DOI] [Google Scholar]
- 157.Burgess B.A., Morley P.S., Hyatt D.R. Environmental surveillance for Salmonella enterica in a veterinary teaching hospital. J. Am. Vet. Med. Assoc. 2004;225:1344–1348. doi: 10.2460/javma.2004.225.1344. [DOI] [PubMed] [Google Scholar]
- 158.Dancer S.J. Controlling Hospital-Acquired Infection: Focus on the Role of the Environment and New Technologies for Decontamination. Clin. Microbiol. Rev. 2014;27:665–690. doi: 10.1128/CMR.00020-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Facciolà A., Pellicanò G.F., Visalli G., Paolucci I.A., Rullo E.V., Ceccarelli M., D’Aleo F., Di Pietro A., Squeri R., Nunnari G., et al. The role of the hospital environment in the healthcare-associated infections: A general review of the literature. Eur. Rev. Med. Pharmacol. Sci. 2019;23:1266–1278. doi: 10.26355/eurrev_201902_17020. [DOI] [PubMed] [Google Scholar]
- 160.Ghosh A., KuKanich K., Brown C.E., Zurek L. Resident Cats in Small Animal Veterinary Hospitals Carry Multi-Drug Resistant Enterococci and are Likely Involved in Cross-Contamination of the Hospital Environment. Front. Microbiol. 2012;3:62. doi: 10.3389/fmicb.2012.00062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Morgan D.J., Rogawski E., Thom K.A., Johnson J.K., Perencevich E., Shardell M., Leekha S., Harris A.D. Transfer of multidrug-resistant bacteria to healthcare workers’ gloves and gowns after patient contact increases with environmental contamination. Crit. Care Med. 2012;40:1045–1051. doi: 10.1097/CCM.0b013e31823bc7c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Murphy C.P., Reid-Smith R.J., Boerlin P., Weese J.S., Prescott J.F., Janecko N., Hassard L., McEwen S.A. Escherichia coli and selected veterinary and zoonotic pathogens isolated from environmental sites in companion animal veterinary hospitals in southern Ontario. Can. Vet. J. 2010;51:963–972. [PMC free article] [PubMed] [Google Scholar]
- 163.Donskey C.J. Does improving surface cleaning and disinfection reduce health care-associated infections? Am. J. Infect. Control. 2013;41:S12–S19. doi: 10.1016/j.ajic.2012.12.010. [DOI] [PubMed] [Google Scholar]
- 164.Protano C., Cammalleri V., Spica V.R. Hospital environment as a reservoir for cross transmission: Cleaning and disinfection procedures. Ann. Ig. 2019;31:436–448. doi: 10.7416/AI.2019.2305. [DOI] [PubMed] [Google Scholar]
- 165.de Abreu P.M., Farias P.G., Paiva G.S., Almeida A.M., Morais P.V. Persistence of microbial communities including Pseudomonas aeruginosa in a hospital environment: A potential health hazard. BMC Microbiol. 2014;14:118. doi: 10.1186/1471-2180-14-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Portner J.A., Johnson J.A. Guidelines for reducing pathogens in veterinary hospitals: Disinfectant selection, cleaning protocols, and hand hygiene. Compend. Contin. Educ. Vet. 2010;32:E1–E11. [PubMed] [Google Scholar]
- 167.Amini R., Moghadam R.H., Soleimani S., Madihi S.V., Jahani H., Heidarzadi K., Karami N., Karimi Z., Hematian A., Mohammadi J., et al. Disinfection and General Cleaning Practices Used in Health Care Centers and Hospitals. J. Basic Res. Med. Sci. 2014;1:1–13. [Google Scholar]
- 168.Traverse M., Aceto H. Environmental Cleaning and Disinfection. Vet. Clin. N. Am. Small Anim. Pract. 2014;45:299–330. doi: 10.1016/j.cvsm.2014.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Chemical Disinfection and Antiseptics: Quantitative Suspension Tests for the Evaluation of Bactericidal Activity of Chemical Disinfectants and Antiseptics Used in the Veterinary Area—Test Method and Requirements (Phase 2, Step 1) European Committee for Standardization; Bruxelles, Belgium: 2019. [Google Scholar]
- 170.Caveney L. Guidelines for Effective Cleaning and Disinfection. In: Caveney L., Jones B., Ellis K., editors. Veterinary Infection Prevention and Control. John Wiley & Sons, Inc.; Ames, IA, USA: 2016. pp. 107–127. [Google Scholar]
- 171.Jones I.A., Joshi L.T. Biocide Use in the Antimicrobial Era: A Review. Molecules. 2021;26:2276. doi: 10.3390/molecules26082276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Fernandes S., Gomes I.B., Sousa S.F., Simões M. Antimicrobial Susceptibility of Persister Biofilm Cells of Bacillus cereus and Pseudomonas fluorescens. Microorganisms. 2022;10:160. doi: 10.3390/microorganisms10010160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Martin D., Denyer S., McDonnell G., Maillard J.-Y. Resistance and cross-resistance to oxidising agents of bacterial isolates from endoscope washer disinfectors. J. Hosp. Infect. 2008;69:377–383. doi: 10.1016/j.jhin.2008.04.010. [DOI] [PubMed] [Google Scholar]
- 174.Maillard J. Russell, Hugo & Ayliffe’s. Wiley-Blackwell; Oxford, UK: 2012. Factors Affecting the Activities of Microbicides; pp. 71–86. [Google Scholar]
- 175.Maillard J.-Y. Resistance of Bacteria to Biocides. Microbiol. Spectr. 2018;6:6.2. doi: 10.1128/microbiolspec.ARBA-0006-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Russell A. Mechanisms of bacterial insusceptibility to biocides. Am. J. Infect. Control. 2001;29:259–261. doi: 10.1067/mic.2001.115671. [DOI] [PubMed] [Google Scholar]
- 177.Gerba C.P. Quaternary Ammonium Biocides: Efficacy in Application. Appl. Environ. Microbiol. 2015;81:464–469. doi: 10.1128/AEM.02633-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Molina-González D., Alonso-Calleja C., Alonso-Hernando A., Capita R. Effect of sub-lethal concentrations of biocides on the susceptibility to antibiotics of multi-drug resistant Salmonella enterica strains. Food Control. 2014;40:329–334. doi: 10.1016/j.foodcont.2013.11.046. [DOI] [Google Scholar]
- 179.Gadea R., Fuentes M.F., Pulido R.P., Gálvez A., Ortega E. Effects of exposure to quaternary-ammonium-based biocides on antimicrobial susceptibility and tolerance to physical stresses in bacteria from organic foods. Food Microbiol. 2017;63:58–71. doi: 10.1016/j.fm.2016.10.037. [DOI] [PubMed] [Google Scholar]
- 180.Tezel U., Pavlostathis S.G. Quaternary ammonium disinfectants: Microbial adaptation, degradation and ecology. Curr. Opin. Biotechnol. 2015;33:296–304. doi: 10.1016/j.copbio.2015.03.018. [DOI] [PubMed] [Google Scholar]
- 181.Webber M.A., Whitehead R.N., Mount M., Loman N.J., Pallen M.J., Piddock L.J.V. Parallel evolutionary pathways to antibiotic resistance selected by biocide exposure. J. Antimicrob. Chemother. 2015;70:2241–2248. doi: 10.1093/jac/dkv109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Capita R., Riesco-Peláez F., Alonso-Hernando A., Alonso-Calleja C. Exposure of Escherichia coli ATCC 12806 to Sublethal Concentrations of Food-Grade Biocides Influences Its Ability to Form Biofilm, Resistance to Antimicrobials, and Ultrastructure. Appl. Environ. Microbiol. 2014;80:1268–1280. doi: 10.1128/AEM.02283-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Geraldes C., Tavares L., Gil S., Oliveira M. Enterococcus Virulence and Resistant Traits Associated with Its Permanence in the Hospital Environment. Antibiotics. 2022;11:857. doi: 10.3390/antibiotics11070857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Gnanadhas D.P., Marathe S.A., Chakravortty D. Biocides—Resistance, cross-resistance mechanisms and assessment. Expert Opin. Investig. Drugs. 2012;22:191–206. doi: 10.1517/13543784.2013.748035. [DOI] [PubMed] [Google Scholar]
- 185.Fraud S., Campigotto A.J., Chen Z., Poole K. MexCD-OprJ Multidrug Efflux System of Pseudomonas aeruginosa: Involvement in Chlorhexidine Resistance and Induction by Membrane-Damaging Agents Dependent upon the AlgU Stress Response Sigma Factor. Antimicrob. Agents Chemother. 2008;52:4478–4482. doi: 10.1128/AAC.01072-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Morita Y., Murata T., Mima T., Shiota S., Kuroda T., Mizushima T., Gotoh N., Nishino T., Tsuchiya T. Induction of mexCD-oprJ operon for a multidrug efflux pump by disinfectants in wild-type Pseudomonas aeruginosa PAO1. J. Antimicrob. Chemother. 2003;51:991–994. doi: 10.1093/jac/dkg173. [DOI] [PubMed] [Google Scholar]
- 187.Rozman U., Pušnik M., Kmetec S., Duh D., Turk S. Reduced Susceptibility and Increased Resistance of Bacteria against Disinfectants: A Systematic Review. Microorganisms. 2021;9:2550. doi: 10.3390/microorganisms9122550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Shrestha P., Zhang Y., Chen W.-J., Wong T.-Y. Triclosan: Antimicrobial mechanisms, antibiotics interactions, clinical applications, and human health. J. Environ. Sci. Health Part C. 2020;38:245–268. doi: 10.1080/26896583.2020.1809286. [DOI] [PubMed] [Google Scholar]
- 189.Randall L.P., Cooles S.W., Piddock L., Woodward M.J. Effect of triclosan or a phenolic farm disinfectant on the selection of antibiotic-resistant Salmonella enterica. J. Antimicrob. Chemother. 2004;54:621–627. doi: 10.1093/jac/dkh376. [DOI] [PubMed] [Google Scholar]
- 190.Giuliano C.A., Rybak M.J. Efficacy of Triclosan as an Antimicrobial Hand Soap and Its Potential Impact on Antimicrobial Resistance: A Focused Review. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2015;35:328–336. doi: 10.1002/phar.1553. [DOI] [PubMed] [Google Scholar]
- 191.Mcmurry L.M., Oethinger M., Levy S.B. Overexpression of marA, soxS, or acrAB produces resistance to triclosan in laboratory and clinical strains of Escherichia coli. FEMS Microbiol. Lett. 1998;166:305–309. doi: 10.1111/j.1574-6968.1998.tb13905.x. [DOI] [PubMed] [Google Scholar]
- 192.Zeng W., Xu W., Xu Y., Liao W., Zhao Y., Zheng X., Xu C., Zhou T., Cao J. The prevalence and mechanism of triclosan resistance in Escherichia coli isolated from urine samples in Wenzhou, China. Antimicrob. Resist. Infect. Control. 2020;9:161. doi: 10.1186/s13756-020-00823-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Chuanchuen R., Beinlich K., Hoang T.T., Becher A., Karkhoff-Schweizer R.R., Schweizer H.P. Cross-Resistance between Triclosan and Antibiotics in Pseudomonas aeruginosa Is Mediated by Multidrug Efflux Pumps: Exposure of a Susceptible Mutant Strain to Triclosan Selects nfxB Mutants Overexpressing MexCD-OprJ. Antimicrob. Agents Chemother. 2001;45:428–432. doi: 10.1128/AAC.45.2.428-432.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Zhu L., Lin J., Ma J., Cronan J.E., Wang H. Triclosan Resistance of Pseudomonas aeruginosa PAO1 Is Due to FabV, a Triclosan-Resistant Enoyl-Acyl Carrier Protein Reductase. Antimicrob. Agents Chemother. 2010;54:689–698. doi: 10.1128/AAC.01152-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.ElDein M.A.T., Yassin A.S., El-Tayeb O., Kashef M.T. Chlorhexidine leads to the evolution of antibiotic-resistant Pseudomonas aeruginosa. Eur. J. Clin. Microbiol. Infect. Dis. 2021;40:2349–2361. doi: 10.1007/s10096-021-04292-5. [DOI] [PubMed] [Google Scholar]
- 196.Shepherd M., Moore G., Wand M., Sutton J.M., Bock L. Pseudomonas aeruginosa adapts to octenidine in the laboratory and a simulated clinical setting, leading to increased tolerance to chlorhexidine and other biocides. J. Hosp. Infect. 2018;100:e23–e29. doi: 10.1016/j.jhin.2018.03.037. [DOI] [PubMed] [Google Scholar]
- 197.Adair F.W., Geftic S.G., Gelzer J. Resistance of Pseudomonas to Quaternary Ammonium Compounds. Appl. Environ. Microbiol. 1971;21:1058–1063. doi: 10.1128/am.21.6.1058-1063.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Buffet-Bataillon S., Tattevin P., Maillard J.-Y., Bonnaure-Mallet M., Jolivet-Gougeon A. Efflux pump induction by quaternary ammonium compounds and fluoroquinolone resistance in bacteria. Futur. Microbiol. 2016;11:81–92. doi: 10.2217/fmb.15.131. [DOI] [PubMed] [Google Scholar]
- 199.Jaglic Z., Cervinkova D. Genetic basis of resistance to quaternary ammonium compounds—The qac genes and their role: A review. Vet. Med. 2012;57:275–281. doi: 10.17221/6013-VETMED. [DOI] [Google Scholar]
- 200.Romão C., Miranda C.A., Silva J., Clementino M.M., de Filippis I., Asensi M. Presence of qacEΔ1 Gene and Susceptibility to a Hospital Biocide in Clinical Isolates of Pseudomonas aeruginosa Resistant to Antibiotics. Curr. Microbiol. 2011;63:16–21. doi: 10.1007/s00284-011-9934-0. [DOI] [PubMed] [Google Scholar]
- 201.Charlebois A., Jacques M., Boulianne M., Archambault M. Tolerance of Clostridium perfringens biofilms to disinfectants commonly used in the food industry. Food Microbiol. 2017;62:32–38. doi: 10.1016/j.fm.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 202.Techtmann S.M., Baldwin A.L., Aluko D., Andersen J., Becker C., Chandler G., Forgrave S., Jones M., Klasner I., Martini J., et al. Draft Genome Sequencing of Three Glutaraldehyde-Tolerant Bacteria from Produced Water from Hydraulic Fracturing. Genome Announc. 2022;11:e01232-21. doi: 10.1128/mra.01232-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Pereira B.M.P., Salim M.A., Rai N., Tagkopoulos I. Tolerance to Glutaraldehyde in Escherichia coli Mediated by Overexpression of the Aldehyde Reductase YqhD by YqhC. Front. Microbiol. 2021;12:680553. doi: 10.3389/fmicb.2021.680553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Nomura K., Ogawa M., Miyamoto H., Muratani T., Taniguchi H. Antibiotic susceptibility of glutaraldehyde-tolerant Mycobacterium chelonae from bronchoscope washing machines. Am. J. Infect. Control. 2004;32:185–188. doi: 10.1016/j.ajic.2003.07.007. [DOI] [PubMed] [Google Scholar]
- 205.Walsh S., Maillard J.-Y., Russell A., Catrenich C., Charbonneau D., Bartolo R. Development of bacterial resistance to several biocides and effects on antibiotic susceptibility. J. Hosp. Infect. 2003;55:98–107. doi: 10.1016/S0195-6701(03)00240-8. [DOI] [PubMed] [Google Scholar]
- 206.Chuanchuen R., Narasaki C.T., Schweizer H.P. The MexJK Efflux Pump of Pseudomonas aeruginosa Requires OprM for Antibiotic Efflux but Not for Efflux of Triclosan. J. Bacteriol. 2002;184:5036–5044. doi: 10.1128/JB.184.18.5036-5044.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Namaki M., Habibzadeh S., Vaez H., Arzanlou M., Safarirad S., Bazghandi S.A., Sahebkar A., Khademi F. Prevalence of resistance genes to biocides in antibiotic-resistant Pseudomonas aeruginosa clinical isolates. Mol. Biol. Rep. 2021;49:2149–2155. doi: 10.1007/s11033-021-07032-2. [DOI] [PubMed] [Google Scholar]
- 208.Blanco P., Hernando-Amado S., Reales-Calderon J.A., Corona F., Lira F., Alcalde-Rico M., Bernardini A., Sanchez M.B., Martinez J.L. Bacterial Multidrug Efflux Pumps: Much More Than Antibiotic Resistance Determinants. Microorganisms. 2016;4:14. doi: 10.3390/microorganisms4010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Mahzounieh M., Khoshnood S., Ebrahimi A., Habibian S., Yaghoubian M. Detection of Antiseptic-Resistance Genes in Pseudomonas and Acinetobacter spp. Isolated from Burn Patients. Jundishapur J. Nat. Pharm. Prod. 2014;9:e15402. doi: 10.17795/jjnpp-15402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Bay D.C., Rommens K.L., Turner R.J. Small multidrug resistance proteins: A multidrug transporter family that continues to grow. Biochim. Biophys. Acta (BBA) Biomembr. 2008;1778:1814–1838. doi: 10.1016/j.bbamem.2007.08.015. [DOI] [PubMed] [Google Scholar]
- 211.Chowdhury C.S., Khan J.A., Khanam J., Nila S.S., Ahmed S., Haque N., Ahamad F., Paul A., Ara H., Paul S.K., et al. Detection of Biocide Resistance Genes (QacE and QacΔE1) in Pseudomonas spp. Isolated from Patients with CSOM at Mymensingh Medical College Hospital, Bangladesh. Mymensingh Med. J. 2021;30:954–959. [PubMed] [Google Scholar]
- 212.Kim M., Weigand M.R., Oh S., Hatt J.K., Krishnan R., Tezel U., Pavlostathis S.G., Konstantinidis K.T. Widely Used Benzalkonium Chloride Disinfectants Can Promote Antibiotic Resistance. Appl. Environ. Microbiol. 2018;84:e01201-18. doi: 10.1128/AEM.01201-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Chen B., Han J., Dai H., Jia P. Biocide-tolerance and antibiotic-resistance in community environments and risk of direct transfers to humans: Unintended consequences of community-wide surface disinfecting during COVID-19? Environ. Pollut. 2021;283:117074. doi: 10.1016/j.envpol.2021.117074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Gomaa F.A.M., Helal Z.H., Khan M.I. High Prevalence of blaNDM-1, blaVIM, qacE, and qacEΔ1 Genes and Their Association with Decreased Susceptibility to Antibiotics and Common Hospital Biocides in Clinical Isolates of Acinetobacter baumannii. Microorganisms. 2017;5:18. doi: 10.3390/microorganisms5020018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Winder C.L., Al-Adham I.S.I., Abdel Malek S.M.A., Buultjens T.E.J., Horrocks A.J., Collier P.J. Outer membrane protein shifts in biocide-resistant Pseudomonas aeruginosa PAO1. J. Appl. Microbiol. 2000;89:289–295. doi: 10.1046/j.1365-2672.2000.01119.x. [DOI] [PubMed] [Google Scholar]
- 216.Elekhnawy E., Sonbol F., Abdelaziz A., Elbanna T. Potential impact of biocide adaptation on selection of antibiotic resistance in bacterial isolates. Futur. J. Pharm. Sci. 2020;6:97. doi: 10.1186/s43094-020-00119-w. [DOI] [Google Scholar]
- 217.Mc Cay P.H., Ocampo-Sosa A.A., Fleming G.T.A. Effect of subinhibitory concentrations of benzalkonium chloride on the competitiveness of Pseudomonas aeruginosa grown in continuous culture. Microbiology. 2010;156:30–38. doi: 10.1099/mic.0.029751-0. [DOI] [PubMed] [Google Scholar]
- 218.Tandukar M., Oh S., Tezel U., Konstantinidis K.T., Pavlostathis S.G. Long-Term Exposure to Benzalkonium Chloride Disinfectants Results in Change of Microbial Community Structure and Increased Antimicrobial Resistance. Environ. Sci. Technol. 2013;47:9730–9738. doi: 10.1021/es401507k. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were created.