Key Points
-
•
Given a growing antimicrobial resistance, the utility of fluoroquinolones prophylaxis in neutropenic transplant recipients is controversial.
-
•
Prophylaxis is preventive for blood-stream infection, not for mortality; its discontinuation concurred to reduce antimicrobial resistance.
Visual Abstract
Abstract
Fluoroquinolone prophylaxis’s (FQ-P) usefulness in patients with neutropenia is controversial. In recent decades, Italian epidemiological data has shown worrisome rates of FQ resistance. A single-center cohort study on 136 autologous stem cell transplantations (ASCTs) and 223 allogeneic hematopoietic stem cell transplantations (allo-HSCTs) was performed from January 2018 to December 2020. Piperacillin/tazobactam was the first-line therapy for febrile neutropenia (FN). Since February 2019, FQ-P has been omitted. We evaluated the day +30 posttransplant cumulative incidence function (CIF) of gram-negative bacteria pre-engraftment bloodstream infections (PE-BSIs) and any changes in antimicrobial resistance, FN, and infection-related mortality (IRM). In ASCTs, ≥1 FN episode occurred in 74.3% of transplants, without differences among groups (P = .66). CIF of gram-negative bacteria PE-BSI was 10.1%, with a significant difference according to FQ-P (0% [LEVO-group] vs 14.1% [NO-LEVO-group], P = .016). CIF of IRM was 0% in both groups. In allo-HSCTs, ≥1 FN episode occurred in 96.4% of transplants, without differences among groups (P = .72). CIF of gram-negative bacteria PE-BSI was 28%, significantly higher without FQ-P (14.7% [LEVO-group] vs 34.4% [NO-LEVO-group], P = .003). CIF of IRM was 5%, superimposable in both groups (P = .62). Comparing antimicrobial resistance among gram-negative bacteria of allo-HSCT setting, in the group without FQ-P, a significantly higher proportion of pathogens was susceptible to piperacillin/tazobactam (71% vs 30%, P = .026), FQ (49% vs 10%, P = .03), and carbapenems (95% vs 50%, P = .001). FQ-P discontinuation increased gram-negative bacteria PE-BSI but did not impact IRM, both in the ASCT and allo-HSCT settings; importantly, it concurred to significantly decrease antimicrobial resistance in gram-negative bacteria.
Introduction
Hematopoietic stem cell transplantation (HSCT) is an established treatment for many hematological diseases.1 Although over the past decades improvements have been made in transplant recipients’ care, transplant-related mortality still represents a relevant cause of death,2 particularly for infectious complications and gram-negative bacteria (GNB) pre-engraftment bloodstream infections (PE-BSI) during the early post-HSCT phase.3, 4, 5, 6
In this context, fluoroquinolone prophylaxis (FQ-P) has been largely adopted worldwide in hematological patients affected by acute leukemia or undergoing HSCT with expected profound and protracted neutropenia.7,8 Two randomized trials showed a significant decrease in febrile neutropenia (FN) and GNB BSIs in patients receiving FQ-P.9,10 Moreover, a meta-analysis reported lower mortality in this setting.11 Concerns were raised regarding the fact that an increasing antimicrobial resistance (AMR) in the community might have a negative impact on FQ-P efficacy in hematology; likewise, its extensive use could increase AMR.12,13 Consequentially, in 2015, the European Conference on Infections in Leukemia group decided to reassess the impact of FQ-P.14 Overall, according to the results from 2 randomized studies15,16 and several observational studies, FQ-P is preventive for FN and BSI; however, it has shown no improvement in mortality rates. A meta-regression analysis was also performed to investigate whether the background FQ resistance rate (which did not exceed 20% and 28% in the community and hospital, respectively) negatively affected FQ-P efficacy and found no effect. Thus, experts suggested weighing the benefit of FQ-P decreasing BSI rates against the disadvantages of its toxicity and ecological changes.17,18
Italian epidemiological data show worrisome rates of FQ resistance (2020 annual report19: 37.6% in Escherichia coli, 52.4% in Klebsiella pneumoniae, and 19.6% in Pseudomonas aeruginosa). Our Hematology and Bone Marrow Transplant Unit also displayed high AMR rates in isolates from allogeneic HSCT (allo-HSCT) recipients with GNB PE-BSIs (71% piperacillin/tazobactam-resistant, 82% FQ-resistant, and 29% carbapenem-resistant [CR]).20 Moreover, alterations of the intestinal microbiome have been reported in patients receiving FQ-P, potentially affecting the occurrence of BSI after allo-HSCT.21
Since February 2019, considering our local epidemiology while maintaining an active microbiological surveillance, we decided to discontinue FQ-P. Here, we continued to prospectively collect microbiological data from transplant recipients to analyze any changes in PE-BSI incidence and AMR of GNB, FN, and mortality.
Methods
This prospective cohort study included consecutive adults who underwent transplantation, either autologous stem cell transplant (ASCT) or allo-HSCT, at our institution from January 2018 to December 2020.
Patients were enrolled in the study upon written informed consent for transplant procedures and the use of medical records for research and were treated according to institutional standard of care.
We collected patients’ age, sex, diagnosis, diseases status at transplant, non–age-adjusted HSCT comorbidity index,22 multidrug resistant (MDR) GNB colonization, presence of neutropenia before transplant, antibiotics exposure and BSI episodes <3 months before transplant, conditioning regimen, donor type, neutrophils engraftment, data on BSI and antibiotic susceptibilities, presence of graft-versus-host disease (GVHD), disease relapse, overall survival, and cause of death.
Hematological treatment’s platforms were uniform throughout the overall study period.
The study outcomes included: (1) the cumulative incidence of PE-BSI, especially GNB PE-BSI, and AMR rates with and without FQ-P; (2) the cumulative incidence of FN and infection-related mortality (IRM); and (3) the evaluation of risk factors for GNB PE-BSIs.
Baseline (day 0) was defined as the date of ASCT or allo-HSCT graft infusion.
All patients were transplanted and hospitalized until neutrophil engraftment.
Follow-up was censored at the date of the occurrence of the event of interest or the competing event or the last available visit, whichever occurred first.
Definitions
Neutropenia was defined as an absolute neutrophils count (ANC) <500 cells/mm3.
Neutropenia before transplant was defined as ANC <500 cells/mm3 on the day of transplant for at least 7 days before, taking into account whether or not the conditioning regimen had begun in aplasia.
Engraftment was defined as the first of 3 consecutive days with ANC >500 cells/mm3.
PE-BSIs were defined as the isolation of a pathogen from ≥1 blood culture of a patient with neutropenia from the beginning of conditioning chemotherapy to neutrophil engraftment. In the case of common skin contaminants, BSIs were diagnosed if ≥2 consecutive blood cultures were positive for the same species. BSIs were considered polymicrobial if ≥2 pathogens were isolated from a single blood culture.
IRM was defined as the time from transplantation to death by an infectious cause without relapse/recurrence or GVHD.
Acute GVHD occurs within 100 days after HSCT and was defined and scored according to the criteria described in Harris et al.23
Transplant-related mortality was defined as the time from transplant to death by a transplant-related cause without relapse.
Patients were defined as MDR-GNB rectal carriers if they had a positive rectal swab within 30 days before transplant.
Underlying diseases were classified as follows: (1) myeloid disorders (acute myeloid leukemia, myelodysplastic syndrome, chronic myelomonocytic leukemia, and myeloproliferative disorders); (2) lymphoid disorders (acute lymphoblastic leukemia, non-Hodgkin lymphoma, Hodgkin lymphoma, multiple myeloma, chronic lymphocytic leukemia, amyloidosis, plasma cell leukemia, and POEMS syndrome); and (3) other disorders (severe aplastic anemia, inherited disorders, and neurological autoimmune disorders).
Infection prophylaxis and treatments
Anti-infectious prophylaxis was administered according to institutional protocols, based on international recommendations.24, 25, 26, 27, 28 With regard to antibacterial prophylaxis, from the onset of conditioning, patients received levofloxacin 500 mg daily until engraftment. According to a change in institutional guidelines, since February 2019, levofloxacin prophylaxis was omitted in hospitalized patients receiving ASCT and allo-HSCT.
During the overall study period, the institutional microbiological surveillance program included performing a rectal swab culture to detect any colonization by CR-GNB at admission and weekly thereafter (active surveillance) and collecting blood cultures at FN onset (passive surveillance).
Drug-resistant GNB were defined as follows: (1) extended-spectrum β-lactamases (ESBLs) Enterobacterales were defined as bacteria producing ESBLs enzymes that break down and destroy some commonly used antibiotics, including penicillin and cephalosporins; (2) carbapenem resistance was defined according to the 2015 Centers for Disease Control and Prevention criteria; briefly, Enterobacterales that test resistant to at least 1 of the carbapenem antibiotics or produce carbapenemase are called CR-Enterobacterales; and (3) GNB isolates were classified as MDR if they acquired nonsusceptibility to at least 1 agent in 3 or more antimicrobial categories, according to the criteria described by Magiorakos.29
Cultures for isolation of CR-GNB were performed on MacConkey agar plates containing a 10 μg disk of carbapenem. After 24 to 48 hours of incubation, colonies growing close to the disk were collected, and antimicrobial sensitivity testing was performed by automated microdilution using the Vitek-2 AST-GN202 card (replaced by AST-GN379 card from June 2018). The modified Hodge test was used to phenotypically detect carbapenemase production, and the synergy between phenylboronic acid and carbapenems in combined disk tests was used to detect K pneumoniae carbapenemase. Antimicrobial susceptibility testing was confirmed by broth microdilution Sensititre system and minimum inhibitory concentration values were interpreted according to the clinical break points by European Committee on Antimicrobial Susceptibility Testing.
For the whole study period, the first-line empiric therapy for uncomplicated FN was piperacillin/tazobactam; in the case of septic shock, it was meropenem plus an aminoglycoside. In MDR-GNB carriers, the empiric therapy for FN was designed to target such a strain in the case of severe clinical presentation and upon a physician’s evaluation in the event of an uncomplicated presentation (first-line empirical therapy or escalation therapy in case of fever persistence); a de-escalation strategy according to microbiological results and clinical course was gradually introduced in our institution starting in 2013.
Finally, there has been no difference in the use of broad-spectrum antibiotics for the empiric management of FN or PE-BSIs according to FQ-P.
Transplantation-related procedures
Transplantations were performed according to institutional guidelines.
In ASCT, patients received either a myeloablative or a reduced-intensity conditioning regimen.
In allo-HSCT, conditioning regimens were treosulfan-based,30 and posttransplant cyclophosphamide was used for in vivo T-cell depletion. Reduced toxicity regimens were based on treosulfan (14 g/m2 per day) on days 6 to 4 and fludarabine (30 mg/m2 per day) on days 6 to 2. In myeloablative regimens, patients also received melphalan (70 mg/m2 per day) on days 3 and 2 or thiotepa (5 mg/kg per day) on days 3 and 2, added to treosulfan and fludarabine. Radiotherapy was added according to local practice.
Donors were divided into matched related, matched unrelated, haploidentical, and cord blood unit.
The graft source was peripheral blood stem cells in nearly all transplants.
In most transplants, postgrafting immunosuppression consisted of posttransplant cyclophosphamide (50 mg/kg per day) on days 3 and 4. GVHD prophylaxis protocols were calcineurin inhibitor–free, based on sirolimus, withdrawn between 3 and 6 months after transplant in the absence of GVHD or relapse; mofetil mycophenolate was added for 30 days if the donor was not a matched related donor. Other immunosuppressive regimens were adopted according to local guidelines.
Further details are reported in the supplemental Materials.
Statistical analysis
The analyses were performed separately for ASCT and allo-HSCT.
Patient characteristics were described as the median (interquartile range, IQR) for continuous variables or proportions for categorical variables. Distributions of continuous variables were compared using the Wilcoxon-rank sum test; differences between proportions were tested by χ2 or Fisher exact test.
The cumulative incidence function (CIF) of any PE-BSI or GNB PE-BSI was calculated according to FQ-P use by Gray’s method,31 accounting for competing risk of pre-engraftment death, engraftment, and retransplantation, and compared; 95% confidence intervals (CIs) of the estimated cumulative incidences were also calculated.
CIFs were also estimated for IRM, considering relapse/progression, GVHD grade ≥2, and death from any other cause as competing risks for IRM.
Given the low incidence rates of GNB PE-BSIs among patients undergoing ASCT, multivariable analyses to assess factors associated with GNB PE-BSI were performed only for those undergoing allo-HSCT.
Univariable and multivariable Fine-Gray subdistribution hazard models were applied to estimate the relative change in the rate of the occurrence of GNB PE-BSI; these models included baseline factors known to have a potential effect on each outcome and other baseline covariates with a P < .2 at univariable analysis. Hazard ratios (HR) with the corresponding 95% CI were reported.
All statistical tests were 2-sided at 5% level and were performed using SAS statistical software version 9.4 (Statistical Analyses System Inc, Cary, NC).
Results
Study population
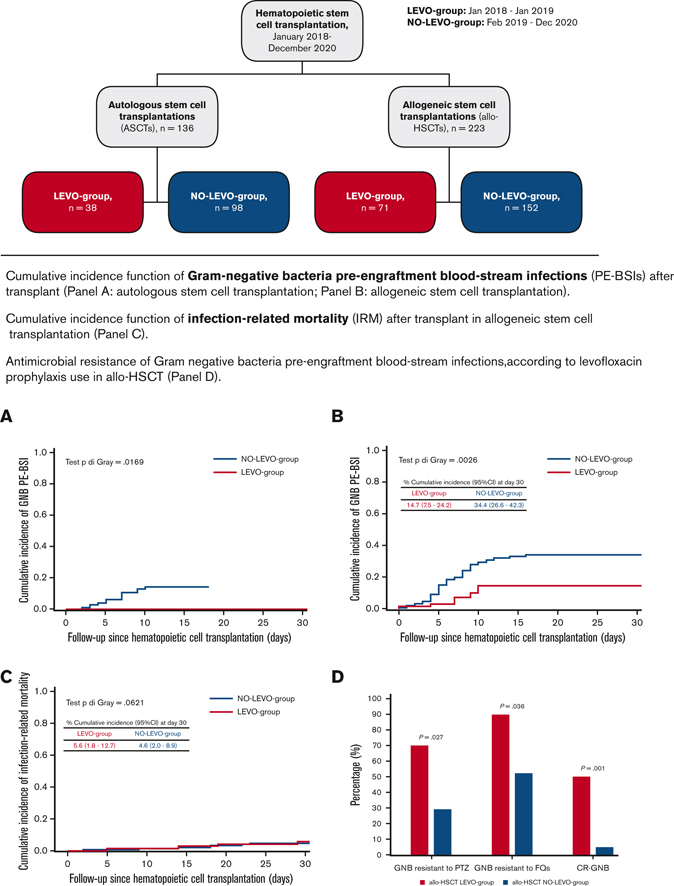
We collected data from 112 of 136 patients undergoing ASCT, including 38 receiving FQ-P (LEVO-group) and 98 not receiving FQ-P (NO-LEVO-group). Baseline patient characteristics were not significantly different between the groups, as shown in Table 1.
Table 1.
Characteristics of patients undergoing ASCT
| Patients’ characteristics | Overall (n = 136) | LEVO-group (n = 38) | NO-LEVO-group (n = 98) | P value |
|---|---|---|---|---|
| Baseline | ||||
| Age at ASCT, y, median (IQR) | 59 (52-66) | 63 (50-66) | 58 (52-65) | .86 |
| Male sex | 87 (64%) | 24 (63.2%) | 63 (64.3%) | .90 |
| Total number of ASCT for patient | .66 | |||
| First ASCT | 104 (76.5%) | 30 (78.9%) | 74 (75.5%) | |
| Second ASCT | 30 (22%) | 8 (21.1%) | 22 (22.5%) | |
| Third ASCT | 2 (1.5%) | 0 (0%) | 2 (2%) | |
| Diagnosis | .82 | |||
| Myeloid disorders | 4 (2.9%) | 1 (2.6%) | 3 (3.1%) | |
| Lymphoid disorders | 127 (93.4%) | 35 (92.1%) | 92 (93.8%) | |
| Other disorders | 5 (3.7%) | 2 (5.3%) | 3 (3.1%) | |
| Disease status at ASCT | .46 | |||
| Complete response | 86 (63.2%) | 21 (55.2%) | 65 (66.3%) | |
| Active disease | 45 (33.1%) | 15 (39.5%) | 30 (30.6%) | |
| Other | 5 (3.7%) | 2 (5.3%) | 3 (3.1%) | |
| Ongoing line of therapy | .75 | |||
| First line | 104 (76.5%) | 29 (76.3%) | 75 (76.5%) | |
| Second line | 21 (15.4%) | 5 (13.2%) | 16 (16.3%) | |
| Third line | 11 (8.1%) | 4 (10.5%) | 7 (7.2%) | |
| Conditioning chemotherapy | .75 | |||
| Myeloablative | 120 (88.2%) | 33 (86.8%) | 87 (88.8%) | |
| Reduced intensity | 16 (11.8%) | 5 (13.2%) | 11 (11.2%) | |
| ANC <500 cells/mm3 ≥ 7 d | 0 (0%) | 0 (0%) | 0 (0%) | - |
| MDR-GNB rectal carrier ≤30 d before ASCT | 1 (0.7%) | 0 (0%) | 1 (1%) | .53 |
| Antibiotics ≤90 d before ASCT | 12 (8.8%) | 0 (0%) | 12 (12.2%) | .02 |
| Follow-up | ||||
| Follow-up, d, median (IQR) | 318 (153-596) | 758 (188-964) | 256 (149-523) | .0001 |
| ANC engraftment | 136 (100%) | 38 (100%) | 98 (100%) | - |
| Time to engraftment, d, median (IQR) | 11 (10-11.5) | 11 (10-11) | 11 (10-12) | .82 |
| Relapse | 22 (16.2%) | 10 (26.3%) | 12 (12.2%) | .05 |
| Overall death | 13 (9.6%) | 7 (18.4%) | 6 (6.1%) | .03 |
| Causes of death | .13 | |||
| Disease | 8 (5.9%) | 5 (13.2%) | 3 (3.1%) | |
| IRM | 3 (2.2%) | 1 (2.6%) | 2 (2%) | |
| Others | 2 (1.5%) | 1 (2.6%) | 1 (1%) |
Moreover, we included data from 221 of 223 patients undergoing allo-HSCT, including 71 receiving FQ-P (LEVO-group) and 152 not receiving FQ-P (NO-LEVO-group). Baseline patient characteristics are shown in Table 2. The main differences between the groups were: the diagnosis, with a higher percentage of patients affected by myeloid disorders in the NO-LEVO-group; the conditioning chemotherapy, with the reduced toxicity regimen30 more represented in the NO-LEVO-group according to center policy; and the rate of MDR-GNB rectal carriers, which was significantly higher in the LEVO-group (8.5% vs 2%).
Table 2.
Characteristics of patients undergoing allo-HSCT
| Patient’ characteristics | Overall (n = 223) | LEVO-group (n = 71) | NO-LEVO-group (n = 152) | P value |
|---|---|---|---|---|
| Baseline | ||||
| Age at allo-HSCT, y, median (IQR) | 56 (41-65) | 52 (38-65) | 57 (44-66) | .20 |
| Male sex | 145 (65%) | 45 (63.4%) | 100 (65.8%) | .72 |
| ANC ≤500 cells/mm3 ≥ 7 d | 28 (12.6%) | 9 (12.7%) | 19 (12.5%) | .97 |
| Diagnosis | .003 | |||
| Myeloid disorders | 164 (73.5%) | 42 (59.2%) | 122 (80.3%) | |
| Lymphoid disorders | 54 (24.2%) | 26 (36.6%) | 28 (18.4%) | |
| Other disorders | 5 (2.3%) | 3 (4.2%) | 2 (1.3%) | |
| Disease status at allo-HSCT | .30 | |||
| Complete response | 140 (62.8%) | 46 (64.8%) | 94 (61.9%) | |
| Active disease and upfront | 78 (35%) | 22 (31%) | 56 (36.8%) | |
| Other | 5 (2.2%) | 3 (4.2%) | 2 (1.3%) | |
| Ongoing line of therapy | .56 | |||
| First line | 99 (44.4%) | 28 (39.4%) | 71 (46.7%) | |
| Second line | 69 (30.9%) | 23 (32.4%) | 46 (30.3%) | |
| ≥Third line | 55 (24.7%) | 20 (28.2%) | 35 (23%) | |
| Total number of allo-HSCT for patient | .15 | |||
| First allo-HSCT | 206 (92.4%) | 62 (87.3%) | 144 (94.7%) | |
| Second allo-HSCT | 15 (6.7%) | 8 (11.3%) | 7 (4.6%) | |
| Third allo-HSCT | 2 (0.9%) | 1 (1.4%) | 1 (0.7%) | |
| HCT-CI index (not age-adjusted) | .36 | |||
| 0-1 | 100 (44.8%) | 35 (49.3%) | 65 (42.8%) | |
| ≥2 | 123 (55.2%) | 36 (50.7%) | 87 (57.2%) | |
| Donor type | .18 | |||
| Matched related donor | 44 (19.7%) | 11 (15.5%) | 33 (21.7%) | |
| Matched unrelated donor | 105 (47.1%) | 33 (46.5%) | 72 (47.4%) | |
| Haploidentical donor | 61 (27.4%) | 25 (35.2%) | 36 (23.7%) | |
| Cord blood | 13 (5.9%) | 2 (2.8%) | 11 (7.2%) | |
| Stem cell source | .41 | |||
| Peripheral blood stem cells | 203 (91%) | 67 (94.4%) | 136 (89.5%) | |
| Bone marrow | 7 (3.1%) | 2 (2.8%) | 5 (3.3%) | |
| Cord blood | 13 (5.8%) | 2 (2.8%) | 11 (7.2%) | |
| Conditioning chemotherapy | .05 | |||
| Myeloablative | 136 (61%) | 50 (70.4%) | 86 (56.6%) | |
| Reduced toxicity | 87 (39%) | 21 (29.6%) | 66 (43.4%) | |
| GVHD prophylaxis | .03 | |||
| PT-Cy/sirolimus–based regimens | 186 (83.4%) | 66 (93%) | 120 (79%) | |
| PT-Cy/ATG or ATG-based regimens | 20 (9%) | 2 (2.8%) | 18 (11.8%) | |
| Other regimens | 17 (7.6%) | 3 (4.2%) | 14 (9.2%) | |
| MDR-GNB rectal carrier within 30 d before allo-HSCT | 9 (4%) | 6 (8.5%) | 3 (2%) | .02 |
| Antibiotics ≤90 d before allo-HSCT | 84 (37.7%) | 26 (36.6%) | 58 (38.2%) | .83 |
| Carbapenems | 43 (51.2%) | 15 (57.7%) | 28 (48.3%) | .48 |
| Fluoroquinolones | 8 (9.5%) | 2 (7.7%) | 6 (10.3%) | 1.00 |
| Glycopeptides | 40 (47.6%) | 15 (57.7%) | 25 (43.1%) | .25 |
| Penicillin/β-lactam inhibitors | 51 (60.7%) | 14 (53.8%) | 37 (63.8%) | .47 |
| Carbapenems or penicillin/β-lactam inhibitors | 68 (81.0%) | 21 (80.8%) | 47 (81.0%) | .99 |
| GNB BSI within 90 d before allo-HSCT | 26 (11.9%) | 5 (7.4%) | 21 (14.0%) | .18 |
| Follow-up | ||||
| Follow-up, days, median (IQR) | 367 (169-685) | 819 (239-966) | 300 (148-526) | <.0001 |
| ANC engraftment | 202 (92.7%) | 64 (90.1%) | 138 (93.9%) | .32 |
| Time to engraftment, d, median (IQR) | 21 (18-28) | 21 (18-28) | 21 (18-28) | .56 |
| Relapse | 39 (17.5%) | 17 (23.9%) | 22 (14.5%) | .08 |
| Time to relapse, d, median (IQR) | 163 (98-335) | 195 (137-368) | 116 (71-227) | .02 |
| Acute GVHD ≥2 | 53 (23.8%) | 13 (18.3%) | 40 (26.3%) | .19 |
| Time to acute GVHD ≥2, d, median (IQR) | 39 (27-54) | 43 (30-61) | 36 (23-49) | .29 |
| Overall death | 56 (25.1%) | 25 (35.2%) | 31 (20.4%) | .02 |
| Time to death, d, median (IQR) | 148 (45-246) | 187 (101-326) | 85 (23-181) | .03 |
| Cause of death | 0.03 | |||
| Disease | 24 (10.8%) | 13 (18.3%) | 11 (7.2%) | |
| IRM | 18 (8.1%) | 5 (7%) | 13 (8.6%) | |
| GVHD | 10 (4.5%) | 6 (8.5%) | 4 (2.6%) | |
| Other | 4 (1.8%) | 1 (1.4%) | 3 (2%) |
ATG, antithymocyte globulin; HCT-CI index, hematopoietic cell transplantation comorbidity index; PT-Cy, posttransplant cyclophosphamide.
Autologous stem cell transplantation
Overall, ≥1 FN episode occurred in 74.3% of ASCT (97/136 with 1 episode, 4/136 with 2 episodes) with a nonsignificant higher incidence in the NO-LEVO-group (71.1% [LEVO-group], 75.5% [NO-LEVO-group]; P = .66). In detail, among LEVO-group, 1 and 2 FN episodes happened in 68.4% and 2.6% of ASCT, respectively; among NO-LEVO-group, the incidence was of 72.5% and 3.1%, respectively (P = .86). The median time to FN onset was day +7 after transplant (IQR: 4-9) without difference between groups (P = .35). Details about the empiric antibiotic treatment for the first episode of FN, along with the duration of antibiotic therapy, are reported in Table 3.
Table 3.
Details on the length of hospital stay, the rate of hospital readmission within 100 day after transplant, antibiotic treatment’s characteristics of the first episode of FN, and the frequency of subsequent bloodstream infection in ASCT cohort
| LEVO-group | NO-LEVO-group | P value | |
|---|---|---|---|
| Duration of hospitalization (d) | 19 (15-27) | 17 (15-27) | .379 |
| Health care use (rehospitalization within 100 d from transplant) | 1 (3.2%)∗ | 3 (3.3%)† | .999 |
| Broad-spectrum antibiotics at the onset of the first FN episode | .026 | ||
| Ceftazidime | 1 (3.7%) | 0 (0%) | |
| Meropenem | 4 (14.8%) | 7 (9.5%) | |
| Piperacillin/tazobactam | 18 (66.7%) | 65 (87.8%) | |
| Others | 4 (14.8%) | 2 (2.7%) | |
| Escalation approach, yes | 12 (44.4%) | 34 (46.0%) | .999 |
| Days to antibiotic escalation since the onset of the first FN episode | 3 (2-3.5) | 2 (2-4) | .990 |
| Type of antibiotic escalation‡ | .345 | ||
| Vancomycin or daptomycin addition | 5 (41.7%) | 25 (73.5%) | |
| Amikacin or gentamycin addition | 0 (0%) | 0 (0%) | |
| Escalation to carbapenem | 7 (58.3%) | 18 (52.9%) | |
| Duration of antibiotic therapy to treat the first FN episode (d) | 9 (7-14) | 10 (8-14) | .645 |
| Reason of antibiotic therapy stop | 1.000 | ||
| Discontinuation (still in aplasia or after engraftment) | 24 (96.0%) | 66 (94.3%) | |
| Escalation for a second FN episode | 1 (4.0%) | 4 (5.7%) | |
| C difficile infection within 100 d | 0 (0%) | 2 (2.0%) | NA |
| ESBL-producing bacteria BSI within 100 d since transplant | 0 (0%) | 0 (0%) | NA |
| CR-GNB BSI within 100 d since transplant | 0 (0%) | 0 (0%) | NA |
Results reported as median (IQR) or frequency (%); comparisons by Wilcoxon rank-sum test or χ2 or Fisher exact test.
Reasons for rehospitalization: neuromotor rehabilitation 1.
Reasons for rehospitalization: pulmonary complications 2 (1 pneumonia and 1 pulmonary thromboembolism), disease progression 1.
Description of antibiotic escalation in supplemental Table 1.
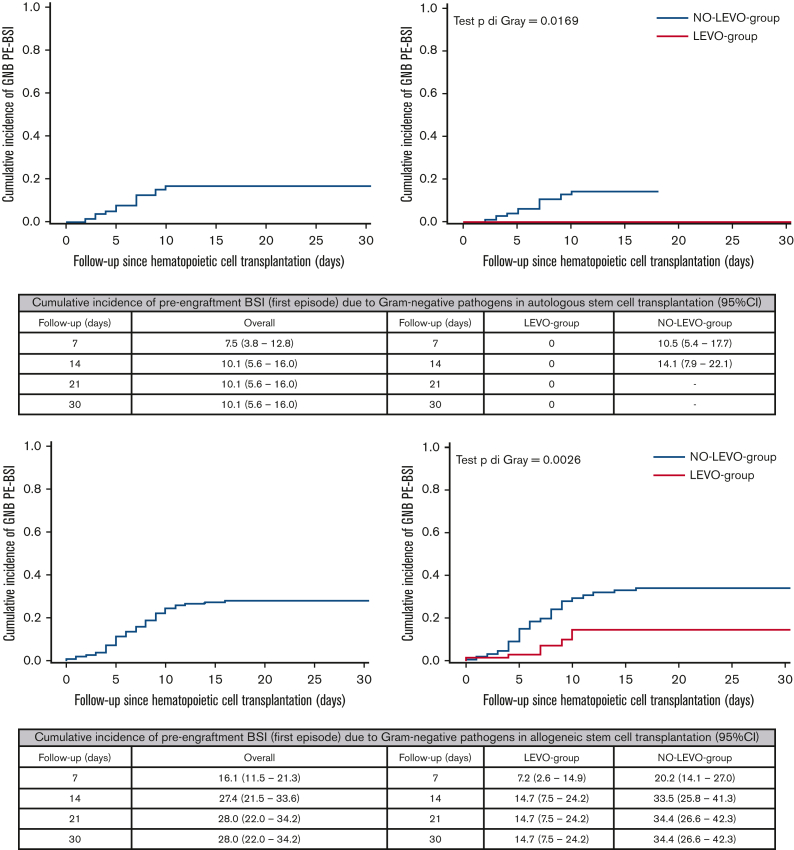
Overall, ≥1 PE-BSI occurred in 16.2% of ASCT (22/136 with 1 episode), and the median time to the first PE-BSI was day +6 after transplant (IQR = 4-7); the estimated CIF was 16.4% (95% CI, 10.7-23.1), both at day +14 and day +30 after transplant. A significant difference was observed in the day +30 CIF according to FQ-P (0% [LEVO-group], 22.6% [NO-LEVO-group]; P = .0017).
Overall, ≥1 GNB PE-BSI occurred in 9.6% of ASCT (13/136 with 1 episode) and the median time to the first GNB PE-BSI was day +7 after transplant (IQR = 4-7); overall, the estimated CIF was 10.1% both at day +14 and day +30 after transplant. A significant difference was observed in the day +30 CIF according to FQ-P (0% [LEVO-group], 14.1% [NO-LEVO-group]; P = .0169) (Figure 1).
Figure 1.
Cumulative incidence of PE-BSI (first episode) due to GNB in the overall sample and according to the use of prophylaxis in ASCT recipients (at the top) and in allo-HSCT recipients (at the bottom).
At day +30 after ASCT, IRM was 0% without differences according to FQ-P use.
With regard to PE-BSI etiology, among the NO-LEVO-group, PE-BSIs were sustained by single-species GNB and single-species gram-positive bacteria, respectively, in 55% (12/22) and 41% (9/22) of cases, whereas 4% (1/22) were polymicrobial. Details about the etiology and AMR of GNB and gram-positive bacteria are reported, respectively, in Table 4 and supplemental Table 4. Regarding MDR-GNB colonization before ASCT, only 1 patient in the NO-LEVO-group was colonized by CR-K pneumoniae and he did not develop PE-BSI. After ASCT, 1 patient in each group acquired colonization by MDR-GNB (1 CR-K pneumoniae and 1 CR-P aeruginosa) without developing signs of infection.
Table 4.
Etiology of GNB PE-BSI and AMR, according to levofloxacin prophylaxis use (group A, ASCT LEVO-group; group B, ASCT NO-LEVO-group; group C, allo-HSCT LEVO-group; group D, allo--HSCT NO-LEVO-group)
| Blood cultures’ isolates characteristics | Total bacterial isolates from blood cultures |
|||||
|---|---|---|---|---|---|---|
| Group A | Group B | P value | Group C | Group D | P value | |
| GNB | 0 | 13 | - | 10 | 58 | |
| GNB resistant to PTZ | 0 (0%) | 2 (15%) | - | 7 (70%) | 17 (29%) | .027 |
| GNB resistant to FQs | 0 (0%) | 2 (15%) | - | 9 (90%) | 30 (52%) | .036 |
| CR-GNB | 0 (0%) | 0 (0%) | - | 5 (50%) | 3 (5%) | .001 |
| E coli | 0 (0%) | 10 (77%) | - | 4 (40%) | 26 (45%) | |
| E coli ESBL-producing | 0 (0%) | 0 (0%) | 2 (50%) | 7 (27%) | ||
| E coli resistant to FQs | 0 (0%) | 1 (10%) | 4 (100%) | 15 (58%) | ||
| E coli resistant to PTZ | 0 (0%) | 2 (20%) | 2 (50%) | 6 (23%) | ||
| CR-E coli | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | ||
| K pneumoniae | 0 (0%) | 1 (8%) | - | 4 (40%) | 13 (22%) | .254 |
| K pneumoniae ESBL-producing | 0 (0%) | 1 (100%) | 0 (0%) | 7 (54%) | ||
| K pneumoniae resistant to FQs | 0 (0%) | 1 (100%) | 4 (100%) | 6 (46%) | ||
| K pneumoniae resistant to PTZ | 0 (0%) | 0 (0%) | 4 (100%) | 6 (46%) | ||
| CR-K pneumoniae | 0 (0%) | 0 (0%) | 4 (100%) | 0 (0%) | ||
| Other Enterobacteriaceae∗ | 0 (0%) | 0 (0%) | - | 0 (0%) | 3 (5%) | - |
| P aeruginosa | 0 (0%) | 2 (15%) | - | 2 (20%) | 8 (14%) | .634 |
| P aeruginosa resistant to FQs | 0 (0%) | 0 (0%) | 1 (50%) | 4 (50%) | ||
| P aeruginosa resistant to PTZ | 0 (0%) | 0 (0%) | 1 (50%) | 3 (37%) | ||
| CR-P aeruginosa | 0 (0%) | 0 (0%) | 1 (50%) | 3 (37%) | ||
| Other GNB† | 0 (0%) | 0 (0%) | - | 0 (0%) | 8 (14%) | - |
Bold values represent P values that reach statistical significance (P < 0.05). PTZ, piperacillin/tazobactam.
1 Klebsiella oxytoca, 1 Enterobacter cloacae, 1 Proteus mirabilis.
Two Ochrobactrum anthropi, 1 Stenotrophomonas maltophilia, 1 Achromobacter xylosoxidans, 1 Campylobacter jejuni, 1 Aeromonas sobrio, 1 Capnocytophaga species, 1 Sphingomonas paucimobilis.
Allogeneic hematopoietic stem cell transplantation
Overall, ≥1 FN episode occurred in 96.4% of those undergoing allo-HSCT (139/223 with 1 episode, 69/223 with 2 episodes, 7/223 with 3 episodes, and 3/223 with 4 episodes) without differences among groups (95.8% [LEVO-group], 96.7% [NO-LEVO-group]; P = .72). Among the LEVO-group, 1 and 2 FN episodes happened in 63.4% and 25.4% of those undergoing allo-HSCT, respectively; whereas among the NO-LEVO-group, the incidence of 1 and 2 FN episodes was in 61.8% and 33.6%, respectively (P = .05). The median time to FN onset was earlier in patients receiving FQ-P (day +2.5, IQR = 1-6 [LEVO-group]; day +5, IQR = 2 to 8 [NO-LEVO-group]; P = .009). Details about the empiric antibiotic treatment for the first episode of FN, along with the duration of antibiotic therapy, are reported in Table 5.
Table 5.
Details on the length of hospital stay, the rate of hospital readmission within 100 days after transplant, antibiotic treatment’s characteristics of the first episode of FN, and the frequency of subsequent bloodstream infection in allo-HSCT cohort
| LEVO-group | NO-LEVO-group | P value | |
|---|---|---|---|
| Duration of hospitalization (d) | 41 (31-55) | 37 (29-46) | .130 |
| Health care use (rehospitalization within 100 d from transplant) | 11 (25%)∗ | 21 (18.6%)† | .384 |
| Broad-spectrum antibiotics at the onset of the first FN episode | .002 | ||
| Ceftazidime | 9 (13.0%) | 2 (1.3%) | |
| Meropenem | 10 (14.5%) | 16 (10.6%) | |
| Piperacillin/tazobactam | 48 (69.6%) | 129 (85.4%) | |
| Others | 2 (2.9%) | 4 (2.7%) | |
| Escalation approach, yes | 44 (64%) | 74 (49%) | .058 |
| Days to antibiotic escalation since the onset of the first FN episode | 3 (2.5-5.5) | 2.5 (2-4) | .003 |
| Type of antibiotic escalation‡ | .646 | ||
| Vancomycin or daptomycin addition | 21 (47.7%) | 42 (56.8%) | |
| Amikacin or gentamycin addition | 5 (11.4%) | 12 (16.2%) | |
| Escalation to carbapenem | 31 (70.5%) | 48 (64.9%) | |
| Duration of antibiotic therapy to treat the first FN episode (d) | 17 (12-23) | 14 (10-20) | .002 |
| Reason of antibiotic therapy stop | .066 | ||
| Discontinuation (still in aplasia or after engraftment) | 47 (72.3%) | 86 (58.9%) | |
| Escalation for a second FN episode | 18 (27.7%) | 60 (41.1%) | |
| C difficile infection within 100 d | 11 (15.5%) | 18 (11.8%) | .522 |
| ESBL-producing bacteria BSI within 100 d since transplant§ | 1 (1.5%)‖ | 5 (3.5%)¶ | .667 |
| CR-GNB BSI within 100 d since transplant§ | 1 (1.5%)# | 1 (0.7%)∗∗ | .535 |
Results reported as median (IQR) or frequency (%); comparisons by Wilcoxon rank-sum test or χ2/Fisher exact test.
Reasons for rehospitalization: immune-mediated encephalopathy 1, possible hepatic-splenic candidiasis 1, clinically-relevant Cytomegalovirus infection 2, fever without microbiologically nor clinically documented infections 2, urinary tract infection 1, cutaneous lesions 1, pan-colitis 1, acute respiratory failure after central venous line removal 1, para-thyroidectomy intervention 1.
Reasons for rehospitalization: acute GVHD 3, viral infections 10 (2 COVID-19, 1 syncytial respiratory virus bronco-pneumonitis, 1 parainfluenza virus bronco-pneumonitis, 3 possible gastrointestinal HHV6 diseases, 1 HHV6 reactivation, 2 BK hemorrhagic cystitis, 1 adenovirus reactivation associated to urinary sepsis), bacterial infections 5 (1 ESBL-E coli BSI, 1 S. epidermidis BSI, 3 pneumonia), probable pulmonary aspergillosis 1, diarrhea 1, myocarditis 1.
Description of antibiotic escalation in supplemental Table 2.
Patients’ distribution according to ESBL BSI or CR BSI or acute GVHD occurrence within 100 days since HSCT and corresponding details in supplemental Table 3; denominators of these proportions did not include patients who died without achieving neutrophils engraftment.
One ESBL-E coli.
One ESBL-E coli, 4 ESBL-K pneumoniae.
One CR-Citrobacter freundii.
One CR-P aeruginosa.
Overall, ≥1 PE-BSI occurred in 44.8% of patients undergoing allo-HSCT (87/223 with 1 episode, 10/223 with 2 episodes, and 2/223 with 3 episodes). The median time to the first PE-BSI was earlier without FQ-P (day +10, IQR = 7-13 [LEVO-group]; day +7, IQR = 5 to 10 [NO-LEVO-group]; P = .028). The estimated CIF of PE-BSI was 41.3% (95% CI, 34.8-47.8) and 47% (95% CI, 39.9-53.8) at days +14 and +30 after transplant, respectively. A significant difference was observed in the day +30 CIF according to FQ-P (36.4% [LEVO-group], 51.9% [NO-LEVO-group]; P = .019).
Overall, ≥1 GNB PE-BSI occurred in 26.5% of patients undergoing allo-HSCT (54/223 with 1 episode, 4/223 with 2 episodes, and 1/223 with 3 episodes); the median time to the first GNB PE-BSI was day +6 after transplant (IQR = 4-9). The estimated CIF was 27.4% and 28% at days +14 and +30 after transplant, respectively. Again, a significant difference was observed in the day +30 CIF according to FQ-P (14.7% [LEVO-group], 34.4% [NO-LEVO-group]; P = .003) (Figure 1).
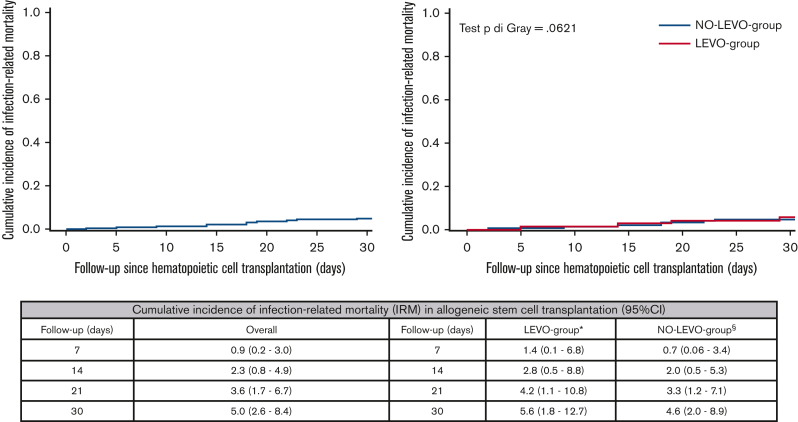
At day +30 after transplant, the estimated CIF of IRM was 5% (95% CI, 2.6-8.4), without differences according to FQ-P (P = .621) (Figure 2). At day +30 after transplant, all 4 patients who died of IRM in the LEVO-group experienced PE-BSI sustained almost exclusively by CR-GNB (2 CR-K pneumoniae and 1 CR-P aeruginosa). In contrast, in the NO-LEVO-group, among the 7 patients who died of IRM, 6 experienced PE-BSI; only 1 PE-BSI was sustained by CR-GNB pathogen (1 CR-P aeruginosa). Even at day +90, no differences in IRM emerged between the 2 groups (supplemental Materials).
Figure 2.
Cumulative incidence of IRM at day +30 after transplant in the overall sample and according to prophylaxis use in allo-HSCT recipients. The cumulative incidence of IRM was estimated according to the Fine-Gray method, with relapse/progression, GVHD grade ≥2, and death from any other cause as competing events for IRM. In this analysis, the time to IRM was censored at the earliest of relapse/progression, GVHD grade ≥2, and death from any other cause, as appropriate. ∗Four patients died of IRM at day +30 after transplant: 2 CR-Kp carriers who experienced CR-Kp PE-BSI achieving BSI clearance with an appropriate empiric antibiotic therapy (cause of death: 1 pneumonia, 1 probable invasive aspergillosis and HHV6 encephalitis); 1 CR-Pa carrier who experienced CR-Pa PE-BSI with an inappropriate empiric antibiotic therapy (cause of death: CR-Pa septic shock and pneumonia); 1 patient who experienced vancomycin-susceptible E faecium PE-BSI developing systemic complications of sepsis (cause of death: cardiac decompensation, acute kidney injury, multiorgan failure). §Seven patients died of IRM at day +30 after transplant: 1 patient died of disseminated Scedosporium prolificans infection; 2 patients experienced P aeruginosa PE-BSI, 1 CR-Pa with inappropriate empiric antibiotic therapy (acquisition of CR-Pa rectal colonization after HSCT [cause of death: CR-Pa septic shock]) and 1 carbapenem-susceptible Pa with appropriate empiric antibiotic therapy achieving BSI clearance (cause of death: pneumonia, multiorgan failure); 1 patient experienced K pneumoniae likely amp-C producer PE-BSI with appropriate empiric antibiotic therapy achieving BSI clearance (cause of death: further PE-BSI sustained by S maltophilia with associated pneumonia); 2 patients experienced not ESBL-producing E coli PE-BSI with appropriate empiric antibiotic therapy achieving BSI clearance (cause of death: 1 septic shock and pneumonia, 1 bowel perforation); 1 patient died of septic shock and pneumonia without the occurrence of PE-BSI.
Regarding PE-BSI etiology, among the LEVO-group, 28 PE-BSI episodes occurred in 25 patients, sustained by single-species GNB and single-species gram-positive bacteria in 36% (10/28) and 61% (17/28) of cases, respectively, and 4% (1/28) were poly-microbial. With regard to GNB, 10 single-species GNB PE-BSI cases occurred in 10 patients. In the NO-LEVO-group, 86 PE-BSI episodes occurred in 75 patients, sustained by single-species GNB and single-species gram-positive bacteria in 58% (50/86) and 30% (26/86) of cases, respectively; 10% (9/86) were poly-microbial, and 1% (1/86) was of fungal etiology (Scedosporium spp). Concerning GNB, 55 GNB PE-BSI occurred in 49 patients, and 5 were poly-microbial. Comparing AMR among GNB, in the NO-LEVO-group, a significantly higher proportion of pathogens was susceptible to piperacillin/tazobactam (71% vs 30%, P = .027), FQ (48% vs 10%, P = .036) and to carbapenems (95% vs 50%, P = .001). Details about the etiology and AMR of GNB and gram-positive bacteria are reported, respectively, in Table 4 and supplemental Table 4.
Focusing on MDR-GNB carriers, before allo-HSCT, in the LEVO-group, 6 patients were colonized (4 CR-K pneumoniae and 2 CR-P aeruginosa), 50% of whom developed PE-BSI sustained by the same pathogen (2/4 CR-K pneumoniae PE-BSI and 1/2 CR-P aeruginosa PE-BSI) and all died of IRM; the other 3 carriers did not experience any PE-BSI. In contrast, in the NO-LEVO-group, 3 patients were colonized (2 CR-K pneumoniae and 1 CR-E coli) and none developed any PE-BSI. After allo-HSCT, in the LEVO-group 17% of patients (11/65) acquired MDR-GNB colonization (6 CR-K pneumoniae, 3 CR-P aeruginosa, 1 CR-Citrobacter, and 1 CR-Enterobacter), with 2 patients developing PE-BSI from the same pathogen (2 CR-K pneumoniae, both survived). In contrast, in the NO-LEVO-group, 10% of patients (15/149) acquired MDR-GNB colonization (4 CR-K pneumoniae, 9 CR-P aeruginosa, 1 CR-E coli, and 1 CR-Enterobacter), with 3 patients experiencing PE-BSI by the same pathogen (2 CR-P aeruginosa, 1 CR-K pneumoniae [1 of the 2 patients with CR-P aeruginosa PE-BSI died of IRM]).
By multivariable analysis (Table 6), after adjustment for conditioning regimen and type of donor, having received FQ-P (adjusted HR [AHR], 0.40; 95% CI, 0.19-0.83) was a protective factor for GNB PE-BSI occurrence, whereas the presence of neutropenia before transplant (AHR, 2.52; 95% CI, 1.27-4.98) and a previous GNB BSI within 90 days before transplant (AHR, 2.15; 95% CI, 1.15-4.02) significantly increased the rate of GNB PE-BSI.
Table 6.
Univariate and multivariate Fine-Gray models to assess risk factors for GNB PE-BSIs in allo-HSCT recipients
| Characteristic | Risk categories | Univariate analysis |
Multivariate analysis (n = 218, 55 GNB PE-BSI, 5 competing events) |
||
|---|---|---|---|---|---|
| Unadjusted HR of GNB PE-BSI (95% CI) | P value | AHR of GNB PE-BSI (95% CI) | P value | ||
| Age at allo-HSCT | Per 5-y older | 1.08 (0.98-1.18) | .107 | 1.11 (1.00-1.24) | .061 |
| Use of prophylaxis | Yes vs no | 0.37 (0.18-0.74) | .005 | 0.40 (0.19-0.83) | .013 |
| ANC ≤500 for ≥ 7 d before allo-HSCT | Yes vs no | 2.67 (1.33-5.36) | .006 | 2.52 (1.27-4.98) | .008 |
| Acute leukemia | Yes vs no | 1.44 (0.75-2.76) | .275 | Not included | - |
| MDR-GNB rectal carrier within 30 d before allo-HSCT | Yes vs no | 1.39 (0.41-4.71) | .599 | Not included | - |
| Conditioning regimen | RTC vs MAC | 1.07 (0.63-1.83) | .793 | 0.70 (0.38-1.28) | .251 |
| GVHD prophylaxis | PT-Cy/ATG or ATG-based regimens vs PT-Cy/sirolimus–based regimens | 1.03 (0.34-3.07) | .963 | Not included | - |
| Other regimens vs PT-Cy/sirolimus–based regimens |
0.61 (0.20-1.93) | .404 | |||
| Type of donor | Haploidentical vs MRD | 0.78 (0.38-1.61) | .497 | 0.91 (0.41-2.03) | .817 |
| CB vs MRD | 1.26 (0.37-4.30) | .139 | 1.63 (0.46-5.74) | .447 | |
| MUD vs MRD | 0.85 (0.43-1.68) | .645 | 0.81 (0.39-1.71) | .587 | |
| Disease phase at allo-HSCT | >CR1 vs CR1 | 0.41 (0.16-1.04) | .059 | Not included | - |
| AD/PR vs CR1 | 1.36 (0.77-2.38) | .287 | |||
| Upfront vs CR1 | 1.13 (0.37-3.44) | .824 | |||
| GNB BSI within 90 d before allo-HSCT | Yes vs no | 2.35 (1.25-4.43) | .008 | 2.15 (1.15-4.02) | .016 |
Bold values represent P values that reach statistical significance (P < 0.05). AD, advanced disease; ATG, antithymocyte globulin; CB, cord blood; CR, complete remission; MAC, myeloablative conditioning; MRD, match-related donor; MUD, match-unrelated donor; PR, partial response; PT-Cy, posttransplant cyclophosphamide; RTC, reduced toxicity conditioning.
Both for the ASCT and allo-HSCT cohorts, we further analyzed any differences according to FQ-P use in the length of hospital stay, in the rate of hospital readmission within 100 days after transplant, and in the frequency of subsequent BSI sustained by ESBL-producing bacteria or CR-GNB (Tables 3 and 5).
Discussion
This study highlighted the impact of FQ-P discontinuation in HSCT recipients in a national and local setting of high FQ-resistance rate (largely exceeding 20%).
Regarding the impact of FQ-P on the incidence of BSI, FN, and mortality, the main findings of this study on 112 and 221 patients who underwent ASCT and allo-HSCT, respectively, can be summarized as follows: (1) PE-BSI incidence, including GNB PE-BSI, was significantly higher in patients who did not receive FQ-P, both in the ASCT and allo-HSCT settings; (2) FN occurrence was not influenced by FQ-P administration; and (3) FQ-P did not reduce IRM at day +30 after transplant. Interestingly, analyzing the differences in AMR within GNB sustaining PE-BSI, we observed the following main findings: (1) FQ-P discontinuation restored susceptibility of GNB to piperacillin/tazobactam, which is our first-line empiric therapy for uncomplicated FN, and (2) CR-GNB PE-BSI incidence was remarkably contained in the FQ-P free cohort.
As shown in recent studies,32,33 the mortality among patients with neutropenia is mainly driven by noncarbapenem β-lactams–resistant and CR-GNB BSI, along with receiving an inappropriate empiric antibiotic therapy for FN. Indeed, recent literature outlined the negative impact of an inappropriate empiric antibiotic therapy on mortality rates for patients with FN with P aeruginosa BSI who received an inappropriate empiric antibiotic therapy (48% vs 31%, P = .027)34 or with septic shock sustained by GNB and Candida.35 Given the negative impact of an inappropriate empiric antibiotic therapy for FN on mortality and the issue of AMR, empiric combination therapy could play a role in improving the appropriateness of empiric therapy and containing IRM in patients at high risk of MDR-GNB BSI, proceeding with a strict de-escalation approach if BSI is not documented.36 A recent propensity-matched cohort study pointed out a lower day 7 case-fatality rate for an empiric aminoglycosides-based combination therapy for FN vs β-lactam monotherapy in a cohort of 542 GNB BSI cases. Because P aeruginosa (including MDR-P aeruginosa) was significantly more represented in the combination therapy group, the greatest benefit is likely to emerge for this pathogen.37 FQ-P tends to increase the rate of FQ-resistant GNB,32 representing one of the predictors of MDR-P aeruginosa BSI in patients with neutropenia.38 Moreover, FQ-P is not always associated with a decreased rate of PE-BSI in HSCT recipients.33 The same relationship between mortality and MDR-GNB BSI also emerged in the setting of high-dose chemotherapy for acute leukemia.39 The feasibility of FQ-P omission has already been postulated in a study on ASCT,40 in which FQ-P seemed to confer a protective effect on FN and BSI without any benefit on mortality rates. Meanwhile, in a Belgian experience on first-induction chemotherapy and transplantation, they reported that FQ-P discontinuation may increase FN with comparable rates of BSI and IRM, and decrease FQ-resistant bacteria recovered from blood.41 Moreover, the gut microbiota plays a critical role in maintaining colonization resistance against intestinal pathogens, and alterations have been reported in patients receiving FQ-P after allo-HSCT.21
Our results confirmed that mortality rates did not increase after FQ-P discontinuation, which is undoubtedly the main safety concern in adopting a FQ-P–free policy in high-risk patients, despite the protective effect of FQ-P on PE-BSI occurrence. However, the increase in GNB PE-BSI did not translate into an excess of mortality, probably because the appreciable increase in GNB susceptible to our first-line empiric therapy for FN reduced the proportion of patients with GNB PE-BSI receiving an inappropriate empiric antibiotic therapy.
The omission of FQ-P leads to a reduction in antibiotic pressure, which is invariably linked to an increase in bacterial resistance. Moreover, previous studies clearly demonstrated that antibiotic use (particularly fluoroquinolones) is predictive for colonization and infection by ESBL-producing strains.42 So, it is plausible that FQ-P omission in the NO-LEVO-group promotes the biodiversity of the enteric microbiome composition,21 restoring the colonization by Enterobacterales susceptible to piperacillin/tazobactam and carbapenems.
Although indicators such as duration of hospitalization and rehospitalization within 100 days from HSCT did not differ among the LEVO-group and the NO-LEVO-group, the issue of MDR pathogens still represents a relevant topic in hematological patients undergoing multiple cycles of chemotherapy and subsequent neutropenic phases. In this regard, if the omission of FQ-P, along with active surveillance of MDR pathogen colonization, contributes to reducing the incidence of MDR-GNB PE-BSI, it is to be considered a valuable achievement.
Considering recent studies and our results, we suggest reconsidering the universal use of FQ-P in transplant recipients to stem the selective pressure that induces BSI sustained by noncarbapenem β-lactams–resistant and CR-GNB, particularly in countries with a high prevalence of AMR. In contrast, a tailored use of FQ-P could be considered for patients colonized by antibiotic-susceptible bacteria, as a recent study demonstrated that FQ-P was effective in preventing BSI in HSCT recipients who were not colonized by FQ-resistant Enterobacterales, whereas nearly one-third of FQ-resistant Enterobacterales carriers developed GNB BSI while receiving FQ-P.43
In the perspective of a tailored use of FQ-P, our data may suggest the use of FQ-P to be beneficial for preventing PE-BSI in ASCT, reducing the need for a full course of antibiotic therapy. Moreover, we urge being aware of one’s own national epidemiology because it is of paramount importance for assessing the translatability of trial results among different countries. For instance, data from the randomized placebo-controlled TEAMM trial, showing a significant reduction of FN episodes and deaths with FQ-P during the first 12 weeks of therapy in patients newly diagnosed with myeloma, may be extended to countries with an FQ-resistance rate in Enterobacterales similar to the United Kingdom (17.5%).44
Nevertheless, this study has some limitations that should be acknowledged.
Although AMR in GNB emerged as a risk factor for mortality in several studies,4,32, 33, 34,39 our single-center analysis was underpowered to assess risk factors for day +30 IRM.
Furthermore, the significant decrease in CR-GNB PE-BSI in NO-LEVO-group could be consequent also to the significantly lower proportion of CR-GNB carriers undergoing allo-HSCT in this patients’ group, as gastrointestinal colonization itself represents a well-known risk factor for CR-GNB PE-BSI4 in transplant setting. Nevertheless, the importance of FQ-P discontinuation in CR-GNB carriers has already been advocated because, in MDR-GNB carriers, FQ-P might reduce the number of susceptible pathogens and promote the selection and growth of MDR strains with an increased risk of subsequent infection due to an MDR pathogen.14
The study period includes the first and second pandemic waves of the coronavirus 2019 disease, which led to a strengthening of the use of personal protective equipment. In the Bone Marrow Transplant ward, this resulted in the systematic use of FFP2 masks while maintaining unchanged the contact isolation precautions for patients colonized by MDR-GNB or affected by Clostridium difficile infection. We previously demonstrated that this approach significantly reduced the crosstransmissions of MDR-GNB among transplant recipients,6 consequently reducing the attributable mortality thanks also to the implementation of a timely diagnosis of MDR-GNB BSI with molecular tests.45 During the pandemic, in the other units of the hospital, we did not observe an increase in infections sustained by antibiotic-susceptible bacteria as we did in the Bone Marrow Transplant Unit; in contrast, a significant increase in MDR bacterial infections was detected, particularly by MDR-P aeruginosa and MDR-Acinetobacter baumanii.
Then, we did not address the impact of inappropriate empiric antibiotic therapy on mortality. The follow-up of NO-LEVO-group was necessarily shorter than LEVO-group, but we think it did not affect study’s results because the impact of PE-BSI on IRM at day +30 after transplant is rightly assessable in the short-term follow-up.
Moreover, our active surveillance screening did not include the detection of colonization by FQ-susceptible GNB, preventing us from implementing a personalized, targeted use of FQ-P as suggested by Satlin.43
Finally, we did not yet analyze the impact of FQ-P in patients undergoing first-induction chemotherapy for acute leukemia, the setting that could benefit more from FQ-P as suggested by a recent randomized controlled trial in leukemic children where FQ-P was preventive on BSI occurrence during induction chemotherapy (but not in transplantation) and by the German Society of Hematology’s updated guidelines.46,47
In conclusion, our study, carried out in a country endemic for CR-GNB, confirms not only the feasibility and safety of FQ-P discontinuation, but also how this approach contributed to a significant reduction in AMR in GNB, even if it resulted in an increase in PE-BSI. This achievement is fundamental because AMR in GNB is the main determinant of mortality in patients with FN. Equally significant is to pursue active microbiological surveillance and a multidisciplinary approach to HSCT recipients, involving the transplant team, infectious disease specialists, and microbiologists.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Acknowledgments
The authors thank Rachel Applefield for the editing of the manuscript, Federica Ardizzoia for supporting data setting, the San Raffaele URC (Clinical Trial Office), the participating patients and their families, all nurses, and data managers who contributed to this study.
Authorship
Contribution: D.C., O.C., A.P.L, and F.E. collected the data; D.C. and O.C. interpreted the data and wrote the manuscript; L.G. performed statistical analysis and prepared figures; D.C., C.O., F.C., and A.C. designed the study; and all authors approved the final version of the manuscript and contributed to patient clinical care and data collection.
Footnotes
∗A.C. and C.O. are joint last authors.
Data are available on request from the corresponding author, Fabio Ciceri (ciceri.fabio@hsr.it).
The full-text version of this article contains a data supplement.
Supplementary Material
References
- 1.Passweg JR, Baldomero H, Chabannon C, et al. Hematopoietic cell transplantation and cellular therapy survey of the EBMT: monitoring of activities and trends over 30 years. Bone Marrow Transplant. 2021;56(7):1651–1664. doi: 10.1038/s41409-021-01227-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Penack O, Peczynski C, Mohty M, et al. How much has allogeneic stem cell transplant-related mortality improved since the 1980s? A retrospective analysis from the EBMT. Blood Adv. 2020;4(24):6283–6290. doi: 10.1182/bloodadvances.2020003418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Styczyński J, Tridello G, Koster L, et al. Death after hematopoietic stem cell transplantation: changes over calendar year time, infections and associated factors. Bone Marrow Transplant. 2020;55(1):126–136. doi: 10.1038/s41409-019-0624-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Girmenia C, Bertaina A, Piciocchi A, et al. Incidence, risk factors and outcome of pre-engraftment gram-negative bacteremia after allogenic and autologous hematopoietic stem cell transplantation: an Italian prospective multicenter survey. Clin Infect Dis. 2017;65(11):1884–1896. doi: 10.1093/cid/cix690. [DOI] [PubMed] [Google Scholar]
- 5.Forcina A, Lorentino F, Marasco V, et al. Clinical impact of pretransplant multidrug-resistant gram-negative colonization in autologous and allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant. 2018;24(7):1476–1482. doi: 10.1016/j.bbmt.2018.02.021. [DOI] [PubMed] [Google Scholar]
- 6.Forcina A, Baldan R, Marasco V, et al. Control of infectious mortality due to carbapenemase-producing Klebsiella pneumoniae in hematopoietic stem cell transplantation. Bone Marrow Transplant. 2017;52(1):114–119. doi: 10.1038/bmt.2016.234. [DOI] [PubMed] [Google Scholar]
- 7.Bucaneve G, Castagnola E, Viscoli C, Leibovici L, Menichetti F. Quinolone prophylaxis for bacterial infections in afebrile high risk neutropenic patients. Eur J Cancer Suppl. 2007;5(2):5–12. [Google Scholar]
- 8.Taplitz RA, Kennedy EB, Bow EJ, et al. Antimicrobial prophylaxis for adult patients with cancer-related immunosuppression: ASCO and IDSA clinical practice guideline update. J Clin Oncol. 2018;36(30):3043–3054. doi: 10.1200/JCO.18.00374. [DOI] [PubMed] [Google Scholar]
- 9.Bucaneve G, Micozzi A, Menichetti F, et al. Levofloxacin to prevent bacterial infection in patients with cancer and neutropenia. N Engl J Med. 2005;353(10):977–987. doi: 10.1056/NEJMoa044097. [DOI] [PubMed] [Google Scholar]
- 10.Cullen M, Steven N, Billingham L, et al. Antibacterial prophylaxis after chemotherapy for solid tumors and lymphomas. N Engl J Med. 2005;353(10):988–998. doi: 10.1056/NEJMoa050078. [DOI] [PubMed] [Google Scholar]
- 11.Gafter-Gvili A, Fraser A, Paul M, Leibovici L. Meta-analysis: antibiotic prophylaxis reduces mortality in neutropenic patients. Ann Intern Med. 2005;142(12):979–995. doi: 10.7326/0003-4819-142-12_part_1-200506210-00008. [DOI] [PubMed] [Google Scholar]
- 12.Lautenbach E, Strom BL, Bilker WB, Patel JB, Edelstein PH, Fishman NO. Epidemiological investigation of fluoroquinolone resistance in infections due to extended-spectrum beta-lactamase producing Escherichia coli and Klebsiella pneumoniae. Clin Infect Dis. 2001;33(8):1288–1294. doi: 10.1086/322667. [DOI] [PubMed] [Google Scholar]
- 13.Paterson DL, Mulazimoglu L, Casellas JM, et al. Epidemiology of ciprofloxacin resistance and its relationship to extended-spectrum beta-lactamase production in Klebsiella pneumoniae isolates causing bacteremia. Clin Infect Dis. 2000;30(3):473–478. doi: 10.1086/313719. [DOI] [PubMed] [Google Scholar]
- 14.Mikulska M, Averbuch D, Tissot F, et al. Fluoroquinolone prophylaxis in haematological cancer patients with neutropenia: ECIL critical appraisal of previous guidelines. J Infect. 2018;76(1):20–37. doi: 10.1016/j.jinf.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 15.Vehreschild JJ, Moritz G, Vehreschild MJGT, et al. Efficacy and safety of moxifloxacin as antibacterial prophylaxis for patients receiving autologous hematopoietic stem cell transplantation: a randomised trial. Int J Antimicrob Agents. 2012;39(2):130–134. doi: 10.1016/j.ijantimicag.2011.10.009. [DOI] [PubMed] [Google Scholar]
- 16.Laoprasopwattana K, Khwanna T, Suwankeeree P, Sujjanunt T, Tunyapanit W, Chelae S. Ciprofloxacin reduces occurrence of fever in children with acute leukemia who develop neutropenia during chemotherapy. Pediatr Infect Dis J. 2013;32(3):e94–e98. doi: 10.1097/INF.0b013e3182793610. [DOI] [PubMed] [Google Scholar]
- 17.Leibovici L, Paul M, Cullen M, et al. Antibiotic prophylaxis in neutropenic patients: new evidence, practical decisions. Cancer. 2006;107(8):1743–1751. doi: 10.1002/cncr.22205. [DOI] [PubMed] [Google Scholar]
- 18.Mikulska M, Cordonnier C. Fluoroquinolone prophylaxis during neutropenia: what can we expect nowadays? Clin Microbiol Infect. 2018;24(7):678–679. doi: 10.1016/j.cmi.2018.02.031. [DOI] [PubMed] [Google Scholar]
- 19.European Centre for Disease Prevention and Control Antimicrobial resistance in the EU/EEA (EARS-Net) – annual epidemiological report for 2020. https://www.ecdc.europa.eu/en/publications-data/surveillance-antimicrobial-resistance-europe-2020
- 20.Oltolini C, Greco R, Galli L, et al. Infections after allogenic transplant with post-transplant cyclophosphamide: impact of donor HLA matching. Biol Blood Marrow Transplant. 2020;26(6):1179–1188. doi: 10.1016/j.bbmt.2020.01.013. [DOI] [PubMed] [Google Scholar]
- 21.Mancini N, Greco R, Pasciuta R, et al. Enteric microbiome markers as early predictors of clinical outcome in allogeneic hematopoietic stem cell transplant: results of a prospective study in adult patient. Open Forum Infect Dis. 2017;4(4) doi: 10.1093/ofid/ofx215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sorror LM, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106(8):2912–2919. doi: 10.1182/blood-2005-05-2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Harris AC, Young R, Devine S, et al. International, multicenter standardization of acute graft-versus-host disease clinical data collection: a report from the Mount Sinai Acute GVHD International Consortium. Biol Blood Marrow Transplant. 2016;22(1):4–10. doi: 10.1016/j.bbmt.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tomblyn M, Chiller T, Einsele H, et al. Guidelines for preventing infectious complications among hematopoietic cell transplantation recipients: a global perspective. Biol Blood Marrow Transplant. 2009;15(10):1143–1238. doi: 10.1016/j.bbmt.2009.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Flowers CR, Seidenfeld J, Bow EJ, et al. Antimicrobial prophylaxis and outpatient management of fever and neutropenia in adults treated for malignancy: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol. 2013;31(6):794–810. doi: 10.1200/JCO.2012.45.8661. [DOI] [PubMed] [Google Scholar]
- 26.Freifeld AG, Bow EJ, Sepkowitz KA, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by Infectious Disease Society of America. Clin Infect Dis. 2011;52(4):e56–e93. doi: 10.1093/cid/cir073. [DOI] [PubMed] [Google Scholar]
- 27.Baden LR, Bensinger W, Angarone M, et al. Prevention and treatment of cancer-related infections. J Natl Compr Canc Netw. 2012;10(11):1412–1445. doi: 10.6004/jnccn.2012.0146. [DOI] [PubMed] [Google Scholar]
- 28.Carreras E, Dufour C, Mohty M, Kröger N. 7th ed. Springer; 2019. The EBMT Handbook. Hematopoietic Stem Cell Transplantation and Cellular Therapies. [PubMed] [Google Scholar]
- 29.Magiorakos AP, Srinivasan A, Carey RB, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect. 2012;18(3):268–281. doi: 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 30.Greco R, Lorentino F, Albanese S, et al. Post-transplantation cyclophosphamide- and sirolimus-based graft-versus-host-disease prophylaxis in allogeneic stem cell transplant. Transplant Cell Ther. 2021;27(9):776.e1–776.e13. doi: 10.1016/j.jtct.2021.05.023. [DOI] [PubMed] [Google Scholar]
- 31.Fine JP, Gray RJ. A proportional hazards model for the sub-distribution of a competing risk. J Am Stat Assoc. 1999;94(446):496–509. [Google Scholar]
- 32.Averbuch D, Tridello G, Hoek J, et al. Antimicrobial resistance in gram-negative rods causing bacteremia in hematopoietic stem cell transplant recipients: intercontinental prospective study of the Infectious Diseases Working Party of the European Bone Marrow Transplantation group. Clin Infect Dis. 2017;65(11):1819–1828. doi: 10.1093/cid/cix646. [DOI] [PubMed] [Google Scholar]
- 33.Averbuch D, Tridello G, Hoek J, et al. Intercontinental study on pre-engraftment and post-engraftment gram-negative rods bacteremia in hematopoietic stem cell transplantation patients: risk factors and association with mortality. J Infect. 2020;81(6):882–894. doi: 10.1016/j.jinf.2020.11.002. [DOI] [PubMed] [Google Scholar]
- 34.Martinez-Nadal G, Puerta-Alcalde P, Gudiol C, et al. Inappropriate empirical antibiotic treatment in high-risk neutropenic patients with bacteremia in the era of multidrug resistance. Clin Infect Dis. 2020;70(6):1068–1074. doi: 10.1093/cid/ciz319. [DOI] [PubMed] [Google Scholar]
- 35.Chumbita M, Puerta-Alcalde P, Gudiol C, et al. Impact of empirical antibiotic regimens on mortality in neutropenic patients with bloodstream infection presenting with septic shock. Antimicrob Agents Chemother. 2021;66(2) doi: 10.1128/AAC.01744-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gustinetti G, Raiola AM, Varaldo R, et al. De-escalation and discontinuation of empirical antibiotic treatment in a cohort of allogeneic hematopoietic stem cell transplantation recipients during the pre-engraftment period. Biol Blood Marrow Transplant. 2018;24(8):1721–1726. doi: 10.1016/j.bbmt.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 37.Albasanz-Puig A, Gudiol C, Puerta-Alcade P, et al. Impact of the inclusion of an aminoglycoside to the initial empirical antibiotic therapy for gram-negative bloodstream infections in hematological neutropenic patients: a propensity-matched cohort study (AMINOLACTAM study) Antimicrob Agents Chemother. 2021;65(8) doi: 10.1128/AAC.00045-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gudiol C, Albasanz-Puig A, Laporte-Amargós J, et al. Clinical predictive model of multidrug resistance in neutropenic cancer patients with bloodstream infection due to Pseudomonas aeruginosa. Antimicrob Agents Chemother. 2020;64(4) doi: 10.1128/AAC.02494-19. e02494-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhao Y, Lin Q, Liu L, et al. Risk factors and outcomes of antibiotic-resistant Pseudomonas aeruginosa bloodstream infection in adult patients with acute leukemia. Clin Infect Dis. 2020;71(4):S386–S393. doi: 10.1093/cid/ciaa1522. [DOI] [PubMed] [Google Scholar]
- 40.Signorelli J, Zimmer A, Liewer S, Shostrom VK, Freifeld A. Incidence of febrile neutropenia in autologous hematopoietic stem cell transplant (HSCT) recipients on levofloxacin prophylaxis. Transpl Infect Dis. 2020;22(2) doi: 10.1111/tid.13225. [DOI] [PubMed] [Google Scholar]
- 41.Verlinden A, Schroyens WA, Gadisseur AP. Clinical and microbiological impact of long-term discontinuation of fluoroquinolone prophylaxis in haematological patients with prolonged profound neutropenia. Eur J Haematol. 2021;107(3):377–379. doi: 10.1111/ejh.13670. [DOI] [PubMed] [Google Scholar]
- 42.Mendelson G, Hait V, Ben-Israel J, et al. Prevalence and risk factors of extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae in an Israeli long-term care facility. Eur J Clin Microbiol Infect Dis. 2005;24(1):17–22. doi: 10.1007/s10096-004-1264-8. [DOI] [PubMed] [Google Scholar]
- 43.Satlin MJ, Chen L, Douglass C, et al. Colonization with fluoroquinolone-resistant Enterobacterales decreases the effectiveness of fluoroquinolone prophylaxis in hematopoietic cell transplant recipients. Clin Infect Dis. 2021;73(7):1257–1265. doi: 10.1093/cid/ciab404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Drayson MT, Bowcock S, Planche T, et al. Levofloxacin prophylaxis in patients with newly diagnosed myeloma (TEAMM): a multicentre, double-blind, placebo-controlled, randomised, phase 3 trial. Lancet Oncol. 2019;20(12):1760–1772. doi: 10.1016/S1470-2045(19)30506-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Greco R, Barbanti MC, Mancini N, et al. Nanosphere's Verigene blood culture assay to detect multidrug-resistant gram-negative bacterial outbreak: a prospective study on 79 hematological patients in a country with high prevalence of antimicrobial resistance. Clin Hematol Int. 2019;1(2):120–123. doi: 10.2991/chi.d.190321.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Alexander S, Fisher BT, Gaur AH, et al. Effect of levofloxacin prophylaxis on bacteremia in children with acute leukemia or undergoing hematopoietic stem cell transplantation: a randomized clinical trial. JAMA. 2018;320(10):995–1004. doi: 10.1001/jama.2018.12512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Classen AY, Henze L, von Lilienfeld-Toal M, et al. Primary prophylaxis of bacterial infections and Pneumocystis jirovecii pneumonia in patients with hematologic malignancies and solid tumors: 2020 updated guidelines of the Infectious Diseases Working Party of the German Society of Hematology and Medical Oncology (AGIHO/DGHO) Ann Hematol. 2021;100(6):1603–1620. doi: 10.1007/s00277-021-04452-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.