Abstract
Background
Flavonols are phytoconstituents of biological and medicinal importance. In addition to functioning as antioxidants, flavonols may play a role in antagonizing diabetes, cancer, cardiovascular disease, and viral and bacterial diseases. Quercetin, myricetin, kaempferol, and fisetin are the major dietary flavonols. Quercetin is a potent scavenger of free radicals, providing protection from free radical damage and oxidation-associated diseases.
Main body of the abstract
An extensive literature review of specific databases (e.g., Pubmed, google scholar, science direct) were conducted using the keywords “flavonol,” “quercetin,” “antidiabetic,” “antiviral,” “anticancer,” and “myricetin.” Some studies concluded that quercetin is a promising antioxidant agent while kaempferol could be effective against human gastric cancer. In addition, kaempferol prevents apoptosis of pancreatic beta-cells via boosting the function and survival rate of the beta-cells, leading to increased insulin secretion. Flavonols also show potential as alternatives to conventional antibiotics, restricting viral infection by antagonizing the envelope proteins to block viral entry.
Short conclusion
There is substantial scientific evidence that high consumption of flavonols is associated with reduced risk of cancer and coronary diseases, free radical damage alleviation, tumor growth prevention, and insulin secretion improvement, among other diverse health benefits. Nevertheless, more studies are required to determine the appropriate dietary concentration, dose, and type of flavonol for a particular condition to prevent any adverse side effects.
Keywords: Flavonol, Quercetin, Myricetin, Kaempferol, Antioxidant
Background
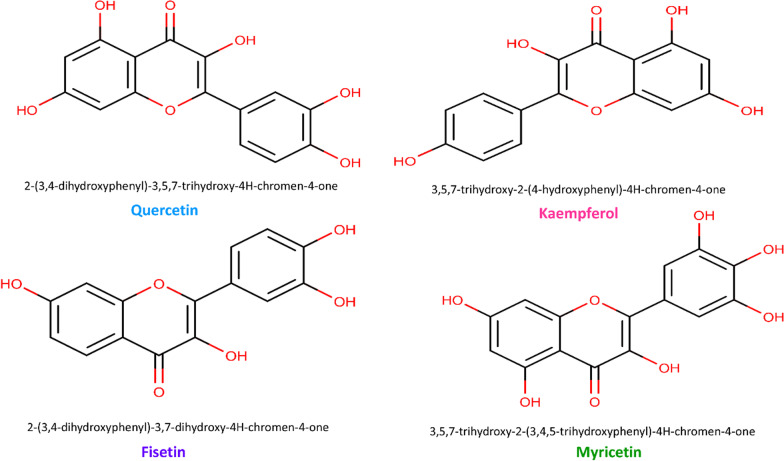
Flavonols are polyphenols that are especially abundant in broccoli, apples, grapes, tomatoes, onion, kale, broccoli, tea, red wine, olive, and citrus fruits. The structure of flavonols is represented by the C6–C3–C6 model in which two benzene rings are joined by a linear three-carbon chain (C2, C3, and C4) with a double bond between C2 and C3 and a carbonyl moiety on C4 (Fig. 1) [77, 167]. Structure–activity studies indicate that the number and arrangement of hydroxyl moieties in flavonols are important determinants of their antioxidant and biological activities [77, 86].
Fig. 1.
Structural formula of some promising medicinal flavanols compound
Flavonols act as antioxidants against a diverse range of stressors. Their photoprotective action is well reported [29, 127], yet how the biosynthesis of flavonols is regulated by ultraviolet (UV) light, the significance of their role in photoprotection, and whether the reactive oxygen species (ROS)-scavenging action of flavonols acts in concert with other signaling pathways in UV protection in planta remain unknown [55, 164]. Nonetheless, it is well established that flavonols control critical psychological functions in multicellular organisms when their redox balance is impaired by natural factors [162]. The transcriptional regulation of flavonol biosynthesis in plants is mediated by myeloblastosis (MYB) transcription factors driven by oxidation–reduction reaction; interestingly, the protective roles given to flavonols outweigh their ability to minimize various sources of reactive oxygen [121]. In addition to contributing to stress resistance in plants, flavonols behave as regulators of maturation and differentiation during plant development due to their affinity for a myriad of proteins involved in signaling pathways critical to cell growth and development [121, 149].
Plants universally increase their flavonol concentrations in response to environmental challenges, including high sunlight intensity, drought, and pathogen attack, creating an efficient strategy to regulate stress-induced ROS production [56, 87, 147]. Previous studies suggest that flavonol accumulation in plants may be induced by auxin and ethylene, key hormones that regulate plant growth and development, indicating associations between the auxin and ethylene signaling pathways and flavonoid regulatory and structural target genes [17, 89]. For example, in Arabidopsis, ethylene-induced flavonol production alleviates ROS in guard cells and affects stomatal conductance [141, 159]. Plant guard cells are essential for photosynthesis and transpiration, regulating the size of the tiny pores or stomata on the surface of leaves in response to environmental cues, such as water status, carbon dioxide concentration, calcium (Ca2+), and ROS.
Oxidative stress has been attributed to inflammation, atherosclerosis, ischemic damage, cancer, and neurodegenerative disorders, like Parkinson’s and Alzheimer’s. Myricetin, fisetin, quercetin, and kaempferol are the most common flavonol aglycones in fruits and vegetables and show high diversity in their methylation, hydroxylation, and glycosylation patterns [137].
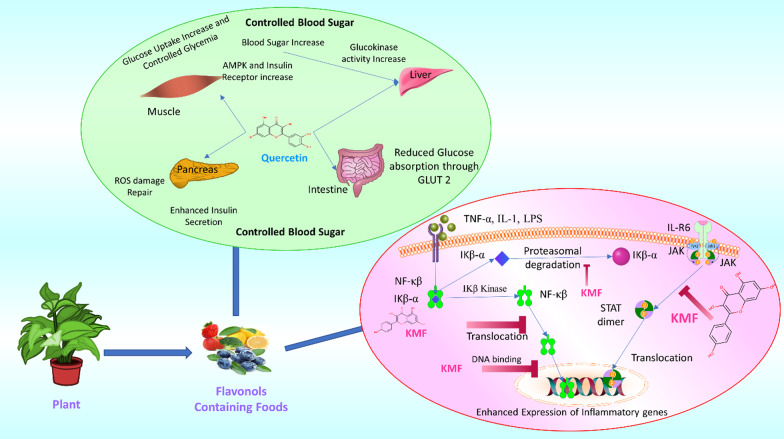
The hydroxyl group in the third position of the regular flavone structure is the key structural feature responsible for the antioxidant and biological action of flavonols. Flavonols are well known as antioxidants that help in scavenging free radicals that could cause serious heart diseases and cancer growth. Quercetin is used as a representative flavonol to illustrate the mechanistic action of scavenging toxic radicals, protecting the body from serious ailments (Fig. 2).
Fig. 2.
Plant-derived flavonol-containing foods have shown profound medicinal activity against disease and maintaining a healthy diet. Quercetin is found to be useful in homeostasis and as a modulator of controlled blood sugar levels in the body. Associate transporters and organs with enzymatic activity are regulated by quercetin, which controls the organ systems by either increasing or decreasing enzymatic activity, enzyme secretion, and expression of transporters, signaling pathways, and other aspects. It controls ROS (Reactive oxygen species) production in the absence of glucose in the pancreatic cell. Besides, quercetin also alleviates glucose assimilation via the GLUT2 receptor. Kaempferol (KMF) is observed in the anti-inflammatory response of cells against pathogen-mediated inflammation. Inflammation is a natural phenomenon that protects the host from damage by pathogens. Uncontrolled inflammation generates a cytokine storm that leads to potential damage to cells. Kaempferol inhibits many inflammatory signal transduction pathways at various levels (i.e., NF-kB, JAK-STAT pathway) NF-kB = Nuclear Factor kB
The perspective of using flavonols as pharmacophores is very intriguing and convincing because of their multipurpose therapeutic effects. However, low bioavailability and bioequivalence have impeded the large-scale production of flavonols as commercial “drugs.” Nanotechnology, such as nanotool delivery systems, may bridge this gap. However, along with this advanced technology, other factors have to be considered, such as the effective route of flavonol administration, the dose of the flavonols, suitability to patients, and the type and process of nanoparticle delivery system [61]. The biological pathways and associated functions of natural flavonols as medicinal compounds for treating various diseases will be illustrated in this review.
Main text
Classification of flavonols
Flavonols are a subclass of flavonoids characterized by a 3-hydroxyflavone backbone. In higher plants, flavonols are typically present in glycosylated forms, preferentially at C3 as mono-, di-, or triglycosides. The sugar residues commonly found in flavonols are glucose, galactose, rhamnose, and glucuronic acid. Flavonols can be classified based on their mechanistic actions.
Therapeutic aspects of flavonols
Research suggests that high consumption of flavonols can alleviate cancer, cardiovascular disease, diabetes, viral infections, bacterial infections, and other disease-related mortality threats [14, 27, 32, 150, 177]. For example, various mechanisms of flavonol action on the androgen receptor (AR)-mediated signaling cascades have been proposed for their chemopreventive action in castrate-resistant prostate cancer [14], and quercetin has undisputed endothelium-independent vasodilator actions, prevents endothelial dysfunction, and prevents myocardial ischemic injury [116] and it is well recognized for its antioxidant function, and it inhibits the action of a large variety of enzymes. As a result, quercetin is expected to impair a range of metabolic signaling mechanisms and biological and clinical processes. In the context of diabetes, there is mounting evidence that flavonols may influence carbohydrate metabolism by modulating α-amylase (the glucose-generating digestive enzyme) and the secondary active transporters (sodium-glucose cotransporter 1 [SGLT1] and glucose transporter 2 [GLUT2]) that mediate intestinal glucose absorption, among various other proposed mechanisms [53, 163]. Moreover, the use of flavonols for the treatment of various infectious diseases is associated with their ability to penetrate the bacterial cell membrane and interact with essential enzymes (e.g., DNA gyrase, DNA PriA helicase, ATPase) [1, 50, 58, 60, 169], including enzymes critical for viral replication, transcription, or integration into host cells (e.g., phosphatidylinositol 3-kinase, RNA polymerase, and 3C-like protease [3CLpro], a cysteine protease) [3, 106].
There is mounting evidence that medicinal plants have a large pool of antiviral compounds that can be developed into potential drug and vaccine candidates after further assessment and research via laboratory experiments and in silico investigations. These plants possess the structural diversity and strong adapting tendency to survive in changing environmental conditions, proving them to be a vital and sustainable source for creating potent drugs and vaccines [122]. Table 1 represents the most common flavonols pharmaceuticals properties with their natural sources.
Table 1.
Natural sources and pharmaceutical properties of flavonols
| Name | Pharmaceutical’s properties | Natural sources | Reference |
|---|---|---|---|
| Apigenin | Antioxidant, anti-inflammatory, and hepatoprotective | Chamomile, parsley, onions, oranges, wheat sprouts | [176] |
| Catechin | Antioxidant, anti-inflammatory, anticancer | Green tea, cocoa, apples, grapes, berries, and other fruits and vegetables | [11, 78, 103, 114] |
| Epicatechin | Antioxidant, anti-inflammatory, anticancer | Cocoa, tea, apples, grapes, berries, and other fruits and vegetables | [11, 20, 54] |
| Eriodictyol | Antioxidant, anti-inflammatory, anticancer, antiviral, and cardioprotective | Citrus fruits, parsley, and other fruits and vegetables | [35] |
| Fisetin | Antioxidant, anti-inflammatory, anticancer, neuroprotective, and cardioprotective | Strawberries, apples, persimmons, grapes, onions, cucumbers, and other fruits and vegetables | [83, 182, 183] |
| Hesperetin | Antioxidant, anti-inflammatory, anticancer, antiviral, neuroprotective, and cardioprotective | Citrus fruits, especially oranges | [129, 158, 182, 183] |
| Kaempferol | Antioxidant, anti-inflammatory, anticancer, antidiabetic, antimalarial, hepatoprotective neuroprotective, and cardioprotective | Tea, broccoli, kale, spinach, beans, berries, and other fruits and vegetables | [6, 119] |
| Myricetin | Antioxidant, anti-inflammatory, anticancer, antidiabetic, antimalarial, neuroprotective, and cardioprotective | Berries, grapes, onions, tea, and other fruits and vegetables | [90, 91, 143, 146] |
| Naringenin | Antioxidant, anti-inflammatory, anticancer, antidiabetic, neuroprotective, and cardioprotective | Citrus fruits, especially grapefruits | [9, 130] |
| Pinocembrin | Antioxidant, anti-inflammatory, anticancer, neuroprotective, and cardioprotective | Propolis, honey, and some fruits and vegetables | [39] |
| Procyanidins | Antioxidant, anti-inflammatory, anticancer | Cocoa, red wine, apples, grapes, berries, and other fruits and vegetables | [26] |
| Quercetin | Antioxidant, anti-inflammatory, anticancer, antiviral, anti-hypertension, antimalarial, neuroprotective, hepatoprotective and cardioprotective | Onions, apples, grapes, berries, broccoli, tea, red wine, and many other fruits and vegetables | [7, 40, 134] |
| Rutin | Antioxidant, anti-inflammatory, anticancer, antimalarial, neuroprotective, and cardioprotective | Buckwheat, asparagus, citrus fruits, and other fruits and vegetables | [46, 113, 131, 174] |
| Silymarin | Antioxidant, anti-inflammatory, anti-fibrotic, and hepatoprotective | Milk thistle | [2, 102] |
| Taxifolin | Antioxidant, anti-inflammatory, anticancer, antidiabetic, neuroprotective, and cardioprotective | Milk thistle, onions, citrus fruits, and other fruits and vegetables | [133, 151, 152] |
| Theaflavins | Antioxidant, anti-inflammatory, anticancer, antidiabetic, neuroprotective, and cardioprotective | Black tea | [88] |
| Thearubigin | Antioxidant, anti-inflammatory, anti tumor, anti-hypertensive | Black tea | [95] |
Anticancer activity of flavonols
The four most studied flavonols (kaempferol, quercetin, fisetin, and myricetin) associated with anticancer functionality are found in foods like olives, onions, various berries, and broccoli [14, 79]. Inhibition of 5-alpha reductase enzymes, interference with androgen and the androgen signaling axis, and suppression of the expression and activity of the AR complex by transactivation of coregulators specificity protein 1 (Sp1) and c-Jun transcription factors along with the phosphoinositide 3-kinase/ Ak strain transforming (PI3K/Akt) pathway are among various mechanisms underlying the chemopreventive effects of flavonols in prostate cancer [14]. Although cancer many react to androgen removal via operation and chemotherapy, the effect is temporary, with disease recurrence, eventually progressing to castrate-resistant prostate cancer, the lethal phenotype of the disease [16]. Tight regulation of the AR signaling homeostasis is critical to maintaining diverse cell functions because dysregulation of this homeostasis leads to aberrant androgen responses and promotes prostate cancer [13]. Some indicators of AR action are prostate-specific antigen (PSA), human protein kallikrein-2 (HK2), and 5α-reductase enzyme [14, 44, 49]. Transcriptional measurements in vitro show that quercetin inhibits the production of PSA and HK2 by suppressing AR function [170, 175]. Kaempferol (extracted from pomegranate extract) hinders dihydrotestosterone-stimulated AR deposition in the nucleus and the expression of PSA and 5α-reductase in vitro [48]. Fisetin, a flavonol found in apple, persimmon, strawberry, onion, kiwifruit, and cucumber, competes with androgen to specifically bind to the ligand-binding domain of AR in lymph node carcinoma of the prostate (LNCaP) cell line. This interaction decreases AR stability and amino-terminal/carboxyl-terminal (N–C) interaction of AR, thereby thwarting transactivation of AR target genes and downregulating AR protein levels. Moreover, in AR-positive CWR22rv1 prostate cancer cell-bearing mice, fisetin inhibits tumor growth and decreases serum PSA levels [73].
The extracellular signal-regulated kinase (ERK) signaling pathway is essential for cancer progression and programmed cell death [19]. By lowering B-cell lymphoma 2 (Bcl-2) expression and causing mitochondrial membrane potential (MMP) impairment, the total ERK1/2 protein expression and phosphorylated ERK functions are inhibited, causing HeLa cervical cancer cells to die [24]. Kaempferol and myricetin have been shown to exert anticancer properties by alleviating phosphorylated ERK activity rather than total ERK protein expression [25, 68, 85]. The ERK signals are also the molecular targets of quercetin in the prevention and treatment of cancer [76].
Nevertheless, the exact functional mechanism by which flavonols reduce the total ERK protein expression in HeLa cells is yet to be determined [15]. In an assessment of the cytotoxic effects of kaempferol, myricetin, and quercetin in rhesus monkey kidney epithelial cells- Lilly Laboratories Culture-Monkey Kidney 2 (LLC-MK2), 5 and 10 μM myricetin and 50 µM quercetin separately hindered normal epithelial cells by cytotoxic assays based on 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and sulforhodamine B, respectively, whereas kaempferol had no impact. However, in HeLa cells, kaempferol and quercetin, but not myricetin, showed marked cytotoxic activities on cancer cells and suppressed total ERK1/2 protein expression, with quercetin showing greater efficiency than kaempferol (Fig. 2) [15].
Fisetin induces apoptotic cell death in oral squamous cell carcinoma cell lines (Ca9-22 and CAL-27) via the mitochondrial pathway [110]. Notably, CAL-27 cells were more susceptible to fisetin, having a 50% inhibitory concentration (IC50) of 50 µM compared to 200 µM for Ca9-22 cells after incubation for 48 h [82]. Fisetin also induces apoptosis in several human laryngeal cancer cell lines (TU212, M2e, and Hep-2) via inhibiting tumor cell proliferation, inducing apoptosis and autophagy regulated by ERK1/2 and AKT/nuclear factor-kappa B (NF-κB)/mammalian target of rapamycin (mTOR) signaling pathways, with an IC50 of 10 µM [180]. In addition, chromatin contraction and nucleus shrinkage lead to programmed cell death and karyorrhexis (nucleus fragmentation), which occurred in oral squamous carcinoma cell lines (HSC-3, Ca9-22, and CAL-27) following fisetin treatment (B. S. [110, 139, 165]. The cytotoxic effects of fisetin also induce necrosis of TU212 cells [82].
Bcl-2 proteins, which include pro-apoptotic pore formers (BAX, BAK, and BOK) and pro-apoptotic BH3-only proteins (e.g., BAD, BID, BIK, BIM, BMF, HRK, NOXA, PUMA) as well as anti-apoptotic factors (e.g., BCL-2, BCL-XL, BCL-W, MCL-1, and BFL-1/A1), are key regulators of the intrinsic apoptotic pathway. Fisetin enhances the activity of pro-apoptotic proteins (e.g., BAD, BAX, NOXA, BOK) and caspase-3, caspase-8, and caspase-9 in head and neck cancer cells but alleviates the activity of anti-apoptotic BCL-2, MCL-1, XIAP, and BCL-X [82]. Moreover, fisetin escalates ROS levels in some oral cancer cell lines (HSC3 and SCC-4) to increase Ca2+ release and decrease the MMP, promoting programmed cell death [139, 144].
Kaempferol dose-dependently inhibits the growth of FaDu pharyngeal cancer cells, with 0.1 and 1 µM kaempferol [64, 64, 65, 65] showing a more potent effect than apigenin, a flavone found in tea [82]. Kaempferol also induces caspase-3-dependent apoptosis in SCC-1483 cells at various concentration doses [71]. In SSC-4 cells, kaempferol downregulates the ERK1/2 signaling pathway and inhibits activator protein-1 (AP-1) activity, markedly decreasing matrix metalloprotein-2 (MMP-2) expression, subsequently producing an antimetastatic effect [92]. The anticancer potency of kaempferol was further confirmed in a mice xenograft model, revealing the ability to significantly prevent the growth of tumor size coupled with a marked decrease in hexokinase-2 expression and epidermal growth factor receptor (EGFR) activity in tumor tissues [64, 65, 172].
Flavonols against cardiovascular disease
The vascular endothelium precisely regulates cardiovascular homeostatic mechanisms. To maintain cardiovascular conditions, the balance among antithrombotic and prothrombotic determinants, antiproliferative and proliferative determinants, and vasoconstrictors and vasodilators should be maintained. If not, platelet accumulation, atherosclerosis, hypertension, and myocardial infarction may occur. Consequently, compromised endothelium-dependent vasodilation is characterized by decreased nitric oxide (NO) activity and the conversion of a normal antithrombotic endothelial state into a prothrombotic and pro-inflammatory state [116]. In most coronary diseases, endothelial malfunction is an initial and independent predictor of poor prognosis [132, 160]. Thus, high blood pressure, arteriosclerosis, myocardial infarction, and various other disorders have been linked to changes in endothelial activity.
Cardiovascular dysfunction is considered one of the major threats for human health, which leads to cardiac diseases and vascular dysfunction. The loss of the regulatory role of the vascular endothelium acts as a diagnostic method to identify early signs of diabetes-induced vascular dysfunction. Flavonols have shown promising therapeutic effects as vasoprotective agents by acting as vasorelaxants [38]. In a study of healthy male subjects, acute consumption of quercetin improved endothelial function by augmenting endogenous NO (S-nitrosothiols, nitrite, and nitrate) and reducing endothelin-1 production [99]. Conversely, in the endothelium-independent process, flavonols prevent extracellular Ca2+ influx by blocking the voltage-gated or receptor-gated Ca2+ channels, hampering the release of Ca2+ from the sarcoplasmic reticulum channels thereby arresting contraction of the vascular endothelium, causing it to relax. Furthermore, quercetin has exhibited activating effects on BKCa by a hydrogen peroxide-dependent process in coronary arteries of rat models [86].
Quercetin’s impact on endothelial development and impairment has been extensively researched [117]. Studies have shown that quercetin exerts systemic and coronary vasodilator effects in vitro and reduces cardiac hypertrophy, endothelial dysfunction, vascular remodeling, oxidative status, and blood pressure in a number of rat models of hypertension [62, 63, 118]. Depending on the experimental conditions, quercetin may also act as a pro-oxidant and generate ROS [30]. However, quercetin (as the aglycone) is usually not present in plasma, but it is rapidly methylated, glucuronidated, and sulfated during absorption in vivo. Quercetin’s effect on NO is often quite complicated, and oxidative pressure markedly impacts the result. In the presence of oxygen, quercetin can auto-oxidize and produce superoxide (O2−) within an in vitro framework that efficiently interacts with and neutralizes NO, an effect that is not observed for the sulfated and glucuronidated derivatives [94, 96]. Amperometric measurements showed that quercetin might elevate the NO level if there is no oxidative pressure [148]. Quercetin has also been shown to alleviate endothelial dysfunction by increasing NO synthesis involving large-conductance Ca2+-activated K+ channel (BKCa)-dependent membrane hyperpolarization-induced capacitative Ca2+ entry [75, 148].
Flavonols can interfere with many biochemical factors implicated in the pathobiology of myocardial infarction and cardiovascular disease, as this section illustrated. Such multiple associations are often beneficial, but they can also be harmful [116].
Flavonols as antidiabetic agents
Diabetes mellitus is a major global health concern. It is defined as a chronic metabolic disorder characterized by the inability to maintain glucose homeostasis due to a malfunctioned glucose uptake mechanism. Diabetes mellitus is subdivided into type 1 diabetes mellitus (T1DM), which is classified as the absolute lack of insulin secretion due to the destruction of the beta-pancreatic cells, and type 2 diabetes mellitus (T2DM), in which insufficient insulin release occurs or the cells have developed resistance to insulin [153]. In addition, there is a phenomenon called “double diabetes” known for its persistent and unmanageable hyperglycemia, in which even a high dose of insulin fails to alleviate the blood glucose concentration. Ultimately, both T1DM and T2DM cause severe macrovascular dysfunction (cardiovascular disease, stroke) and microvascular dysfunction (nephropathy, neuropathy, retinal impairment) [61]. A clinical study has shown that long-term uncontrolled diabetes mellitus can also increase the risk of developing Alzheimer’s disease [4, 59]. Many therapeutic companies have developed drugs for the management of diabetes mellitus. However, the conventional methods have not generated the desired outcomes in patients, which have led scientists to study the reservoir of phytochemicals, more specifically, flavonols, as an alternative approach [38].
A growing body of evidence has exhibited an inverse correlation between the dietary intake of flavonols and the risk of T2DM. As already mentioned in this review, the antioxidant activity of flavonols can regulate the redox status and prevent damage caused by oxidative stress. Hyperglycemia causes increased ROS production due to the altered and high mitochondrial oxidation, resulting in high oxidative stress. ROS are constantly being generated in animals carrying out aerobic oxidation. ROS accompanied by poor antioxidant capacity results in oxidative stress [145]. Due to its lack of antioxidant enzymes, the pancreas is exceedingly vulnerable to the effects of oxidative stress, and the final consequence is the death of pancreas beta-cells, hence its contribution to the pathogenesis of diabetes [18, 145]. Kaempferol has been shown to improve cell viability and prevent pancreatic beta-cell apoptosis. When tested on cultured cells under high glucose concentrations, kaempferol promoted pancreatic beta-cell function and survival rate and insulin secretory function by restoring high-glucose-attenuated intracellular adenosine monophosphate (AMP) and adenosine triphosphate (ATP) production and improving the expression of anti-apoptotic genes Akt and Bcl-2 [86]. Additionally, kaempferol tended to be inversely related to T2DM risk, and myricetin showed a protective role against T2DM in men and women across European countries in a large, prospective case–cohort study [177]. Other studies have supported that quercetin induces insulin secretion and protects beta-cell function and the pancreas from oxidative damage and inflammatory cytokines [173]. Quercetin also indirectly neutralizes oxidative stress via the activation of the nuclear erythroid 2-related factor 2-antioxidant response element (Nrf2-ARE) antioxidant pathway and stimulates the catalase and superoxide dismutase antioxidant enzymes [66]. Besides hyperglycemia, lipid peroxidation aggravates oxidative stress by converting the free fatty acids to free radicals via hydrogen extraction. Quercetin has proved to be an efficient antioxidant phytochemical because of its free radical scavenging activity and chelation of transition metal ions [34]. It is proposed that quercetin arrests lipid peroxidation by inhibiting xanthine oxidase, the enzyme responsible for catalyzing the oxidation of xanthine to uric acid and simultaneous superoxide formation [34, 84]. The mechanism describes more than the quercetin itself becomes a radical but too low in energy to become a deteriorating reactive one by the reaction with another free radical alongside the donation of a proton [52].
Some of the long-term diabetic complications are cataracts (diabetic retinopathy), neuropathy, and nephropathy because of sorbitol accumulation in the body. Quercetin exerts an inhibitory action on the aldose reductase enzyme, which catalyzes the conversion of glucose to sorbitol (a sugar alcohol moiety) [99].
The insulin resistance along with hyperglycemia causes inflammation, activating the body’s various inflammatory mechanisms with subsequent release of cytokines and inflammatory mediators, leading to an aberrant uncontrolled response known as “cytokine storm.” One consequence of this phenomenon is organ damage or, worse still, multiple organ damage failure. Quercetin possesses antioxidant and inhibitory effects on the inflammatory response because of its high-affinity interactions and inhibition of enzymes playing a role in oxidative reactions and processes, such as cyclooxygenase and lipoxygenase, following the inhibition of leukotrienes and prostaglandins (inflammatory mediators) [138, 145].
Flavonols as antiviral agents
Flavonols may be considered an alternative treatment for various viral diseases due to the failure of conventional therapies to generate positive results. Experimental studies and in silico investigations have determined some promising therapeutic flavonols displaying the maximum antiviral effects [74, 101]. In this context, it is predicted that flavonols could become a consistent source of probable drugs and vaccines for various pandemic and epidemic diseases [150]. Even after the development of vaccines, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) has still managed to halt the world due to the emergence of its various strains. Polyphenolic compounds stand out as potential treatment methods and future imminent biopharmaceuticals [3]. In SARS-CoV-2, the cytokine storm results from over-production of the pro-inflammatory cytokines due to the stimulation of the Nod-like receptor family and pyrin domain-containing 3 (NLRP3) inflammatory pathway. Myricetin, rutin, kaempferol, fisetin, and astragalin are among numerous other flavonols that display inhibitory effects on the synthesis and expression of the inflammatory mediators and cytokines in the prevention of the cytokine storm syndrome [57, 101, 111].
Quercetin suppresses NLRP3 inflammasome activation and inhibits lipopolysaccharide-induced production of various cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin (IL)-6, IL-1β, and IL-8 in whole blood cells. Additionally, vitamin C and quercetin display a synergistic effect in combating viral infections [128]. The 3CLpro protease is a vital enzyme of coronaviruses. It cleaves the polyproteins (pp1a and pp1ab) into the individual nonstructural proteins (Nsps) that form the RNA replicase–transcriptase complex, which mediates viral transcription and replication. Finally, virions are released from the infected host cells. Quercetin exhibited > 80% inhibition activity with an IC50 of 73 μM on the 3CLpro protein product in vitro [31, 142]. The binding affinity of quercetin to the 3CLpro protease enzyme is likely a critical factor in its antiviral activity. Quercetin is a type of flavonoid that has been shown to have antiviral properties against coronaviruses. Quercetin has been shown to inhibit the 3CLpro protease enzyme of coronaviruses, which is essential for the cleavage the viral polyproteins into individual nonstructural proteins, enabling viral replication. The inhibition of this protease enzyme reduces viral replication and, hence, viral colonization load. The binding affinity of quercetin to the 3CLpro protease is vital in determining its antiviral activity. A higher binding affinity between quercetin and the 3CLpro protease can result in more robust inhibition of the enzyme’s activity, leading to more effective suppression of viral replication. Conversely, a lower binding affinity can result in weaker inhibition and may not be sufficient to control the virus’s spread.
Therefore, it can be inferred that the antiviral activity of quercetin is likely due to its ability to bind to the 3CLpro enzyme with high affinity and inhibit its activity, thereby reducing viral replication and load. However, it is essential to note that the in vitro studies do not necessarily indicate the same antiviral activity in vivo, and further studies are required to determine the potential therapeutic use of quercetin for viral infections. Isoquercitrin (quercetin 3-O-glycoside [Q3G]) possessed inhibitory effects on the 3CLpro of SARS-CoV [126]. Molecular modeling and docking studies asserted that Q3G interacts with Gln189, one of the key amino acid residues residing in the catalytic pocket of the 3CLpro enzyme, arresting the 3CLpro function [101]. Papain-like protease (PLpro) represents another therapeutic target for the pharmacophore because of its multifunctional roles during viral replication, specifically, its involvement in cleaving the N-terminal viral polyproteins to generate various Nsps (Nsp1, Nsp2, and Nsp3). The PLpro is also used as a means of immune escape by the virus via inactivation of the interferon regulatory factor-3 (IRF3) pathway, resulting in reduced production of antiviral interferons, hence causing immunosuppression [106]. Many studies claim that quercetin has an excellent binding affinity with RNA-dependent RNA polymerase (RdRp), host cell receptor angiotensin-converting enzyme 2 (ACE2), spike protein, and PLpro [70, 93]. Moreover, quercetin acts as a zinc ionophore, accelerating the transport of zinc ions across the lipid membranes. This action is crucial to blocking the entry of the virus into the host cells because coronavirus is vulnerable to the detrimental effects of zinc ions [3].
Acquired immunodeficiency syndrome (AIDS) is a chronic, potentially life-threatening health condition caused by the human immunodeficiency virus (HIV). The combined antiretroviral therapy (cART) introduced in 1996 constitutes several classes of drugs that act in concert to control and suppress HIV progression. The cornerstone of highly active antiretroviral therapy (HAART), which may also be called cART, is the co-administration of several different drugs that inhibit viral replication, an approach demonstrated to correlate with improved outcomes for patients with AIDS [80, 123, 135]. Nevertheless, the drugs pose long-term side effects. Moreover, emerging drug resistance, toxicity, lack of therapeutic effect, and restricted availability are among several disadvantages that have created avenues for the development of alternative medicines based on natural, medicinal compounds [122].
The HIV genome encodes for three enzymes essential for virus replication: protease (PR), reverse transcriptase (RT), and integrase (IN). The different subclasses and derivatives of flavonols act as multitarget agents by acting on these enzymes (PR, IN, and RT) while at the same time interfering and disrupting the steps in the viral replication process. The flavonol 7-O-glucoside herbacitrin, a kaempferol derivative with an additional hydroxyl group at the C-8 position, has shown such an effect, simultaneously inhibiting HIV-1 RT and HIV-IN. In an in vitro test designed to determine the potential target of herbacitrin, the data revealed marked inhibition of the HIV-1 RT at a high concentration of 21.5 μM herbacitrin compared to the inhibition of HIV-IN, which occurred at 2.15 μM [122]. RT was the first protein to be exploited for anti-HIV therapy. This multifunctional enzyme catalyzes the steps in the early stages of the viral replication process. It confers reverse transcriptase (RNA-dependent DNA polymerase, RDDP) activity, DNA-dependent DNA polymerase (DDDP), and the inherent ribonuclease H (RNase H) activity to catalyze the conversion of the viral genomic RNA into double-stranded DNA. Therefore, inhibition of each or any of the catalytic functions of RT will ultimately interfere with and disrupt the virus life cycle. Several quercetin- and kaempferol-based flavonol glycosides isolated from the leaves of Thevetia peruviana displayed notable inhibitory activity against RDDP, with IC50 of 20–43 μM, albeit the quercetin derivatives showing more activity than the kaempferol derivatives [150]. Kaempferol has displayed inhibiting effects on the RDDP of HIV-1 RT [112].
Furthermore, some flavonols block viral entry into the host cell by affecting the CD4 receptor and the CXCR4 and CCR5 co-receptors, eliminating the communication between the receptor and the cell’s internal pathways. In the TZM-bl cell line, a frequently used HIV-1 reporter cell line developed from HeLa cells, myricetin presented potent anti-HIV-1 activity, with > 87% inhibition of the infection and 49% inhibition against HIV-RT [112]. Thus, according to the World Health Organization (WHO) regulatory guidelines, myricetin has exhibited two crucial requirements to be developed as a microbicide: low toxicity and inhibitory activity against HIV infection [112].
Antibacterial activity of flavonols
Pathogenic bacteria have become a threat to human beings. Before the first effective antibiotic, penicillin was disclosed in 1928, followed by sulfonamide medications in 1930, bacterial infections were the cardinal cause of death. As such, antibiotics have played a key role in managing contagious bacterial infections over the last 60 years [50]. However, the latest antibiotics reservoir has been slowly depleting since the 1970s, with a rising prevalence of antibiotic-resistant microorganisms [136], leading to a prominent post-antibiotic age [8]. Nevertheless, it is anticipated that plant-derived flavonols will play a vital role in combating bacteria and may prove to be an alternative drug treatment against bacteria (Table 2).
Table 2.
Flavonols and their inhibitory dose concentrations against several types of bacteria
| Flavonol | Susceptible bacteria | Function | Dose concentration (mcg/mL) | Reference |
|---|---|---|---|---|
| Kaempferol | Helicobacter pylori | Bactericidal effect | 1000 | [102] |
| Quercetin | Pseudomonas aeruginosa | Antibiofilm activity | 20 | [107], [108], [123] |
| Shigella flexneri | No visible effect | 500 | ||
| Lactobacillus casei var. shirota | No visible effect | 500 | ||
| Proteus vulgaris | Damage to cell walls and membranes by enhancing alkaline phosphatase and β-galactosidase production | 300 | ||
| Escherichia coli | Damage to cell walls and membranes by enhancing alkaline phosphatase and β-galactosidase production | 400 | ||
| Staphylococcus aureus | Bacteriostatic effect via > 70% inhibition of biofilm formation | 20 | ||
| Fisetin | Streptococcus suis | Target suilysin to hinder apertures and hemolysis | 32 | [116] |
Kaempferol displays a dose-dependent inhibitory action against Helicobacter pylori, the bacterium responsible for human gastric cancer and peptic ulcer diseases worldwide [41]. Helicobacter pylori is a coil-shaped, neutralophilic, Gram-negative bacterium that inhabits the human bowel. Helicobacter pylori infection constitutes the predominant risk factor for gastric cancer and peptic ulcers combined, representing over a million annual fatalities [10, 104]. Combined treatments include proton-pump inhibitors and antibiotics (tetracycline, amoxicillin, clarithromycin, or metronidazole) [107]. Nevertheless, because of the high incidence of resistance to antibiotics, H. pylori elimination may not always be effective. According to a contemporary analysis in South American countries, antibiotic resistance trends differ dramatically by medication and region [21]. Kaempferol substantially inhibited two H. pylori strains (26,695 and 43,504) at 1000 mcg/mL in a liquid culture medium [41]. Moreover, a synergistic effect was observed after the combined application of kaempferol and (−)-epicatechin.
The omnipresent flavonol quercetin is considered to have antibacterial properties. Escherichia coli (Gram-negative bacterium) and Staphylococcus aureus (Gram-positive bacterium) were used to assess the antibacterial function of quercetin [157]. Gram-positive bacteria do not possess an outer lipopolysaccharide cell wall but harbor a thick peptidoglycan cell wall membrane. By contrast, Gram-negative bacteria have an outer lipopolysaccharide cell wall but a relatively thin peptidoglycan cell wall membrane. Quercetin damaged the cell walls and membranes of both E. coli and S. aureus via ameliorating the activity of the alkaline phosphatase and β-galactosidase enzyme concentrations in vitro, an effect that increased exponentially with increasing quercetin amounts [157]. It was also observed that quercetin had a greater bacteriostatic effect against Gram-positive bacteria than Gram-negative bacteria when tested against E. coli, Pseudomonas aeruginosa, Salmonella enterica Typhimurium, and S. aureus [157]. In another study, quercetin was tested for its antibacterial activity against P. aeruginosa, Shigella flexneri, Lactobacillus casei var shirota, Proteus vulgaris, E. coli, and S. aureus [67]. The minimum inhibitory concentrations of quercetin were 20 mcg/mL against S. aureus and P. aeruginosa, 300 mcg/mL against P. vulgaris, and 400 mcg/mL against E. coli [67, 161]. However, S. flexneri and L. casei var shirota were unaffected, even at 500 mcg/mL of quercetin [67].
Quercetin displays antiquorum sensing potential against P. aeruginosa. Research demonstrated the in vitro cytoprotective activity of quercetin against P. aeruginosa infection in human embryonic kidney cells (HEK 293 T), revealing 100% inhibition of the tested isolates by quercetin at 500 μg/mL, besides notable virulence attenuation and biofilm formation inhibition while also protecting the host epithelial cells [156]. These effects were observed even at a concentrated dose of 10,000 mcg/mL of quercetin because quercetin does not have major adverse effects on normal human cells [33, 120, 156]. In another example, four P. aeruginosa isolates (YU-V10, YU-V11, YU-V15, and YU-V28) with resistance to ciprofloxacin, gentamicin, amikacin, imipenem, and ceftazidime antibiotics were collected from urinary catheters to assess the antibiofilm efficacy of quercetin using the reference strain P. aeruginosa (PAO1), which was sensitive to all the tested antibiotics [156]. At a concentration of 250 mcg/mL, quercetin suppressed the biofilm formation of all isolates by about 43% to 79% compared with the reference strain. An initial determination indicated that although quercetin did not inhibit the growth of PAO1, it markedly inhibited biofilm formation by up to 51% at 8–64 mcg/mL and exerted a dramatic negative effect on the virulence factors [108]. Other studies have also supported the antibiofilm efficacy of quercetin [154, 178].
Streptococcus suis serotype 2 infection is a dangerous, porcine pathogen and zoonotic agent that has captured global attention since the first human case was reported in Denmark in 1968. Several virulence determinants are involved in the pathogenesis of the infection caused by S. suis serotype 2. New strategies for finding antivirulence molecules that can kill or hinder zoonotic bacterial infections have been published in recent decades. Fisetin was revealed as a potent antagonist to S. suis-mediated pathogenesis, specifically by suppressing the hemolytic activity of suilysin, a critical virulence factor [181]. To infect the host, S. suis must subvert epithelial blockades, evade the host’s immune system, replicate in the blood circulation, and infiltrate multiple organs, ultimately causing necrosis of the tissues, cells, or organs [43, 51]. The S. suis serotype 2 strain SC19 is known to secrete suilysin encoded by the sly gene and is highly noxious to pigs and mice [168, 179]. Suilysin exhibits antiphagocytic and antibactericidal properties in response to neutrophils and macrophages [12, 23, 43]. To achieve the maximal inhibition of suilysin in vitro, 32 mcg/mL of fisetin was required [181]. In vivo, 0.1 mcg/kg of fisetin showed therapeutic activity in SC19-infected mice, decreasing the bacterial loads and pro-inflammatory ability and improving the survival rate [181].
Other pharmaceuticals properties of flavonols
Antioxidant agents
Flavonols are a form of flavonoid that has been widely researched for their antioxidant qualities. Antioxidants are substances that protect cells from free radical damage, which is generated by highly reactive molecules in the body that create oxidative stress. Flavonols have been discovered to be powerful antioxidants capable of protecting cells from oxidative stress and lowering the risk of chronic illnesses such as cancer, heart disease, and diabetes [134]. Flavonols such as quercetin, kaempferol, and myricetin have been proved in vitro to effectively scavenge free radicals such as superoxide anion, hydroxyl radical, and peroxyl radical [69, 125]. Flavonols can also bind metal ions, which can limit free radical generation. Metal ions such as iron and copper can stimulate the generation of free radicals, which can be prevented by chelating them with flavonols [28, 125]. Flavonols can help renew antioxidants such as vitamin C and vitamin E in the body [125]. Vitamins C and E are key antioxidants that may protect cells from oxidative stress, and flavonols have been shown to boost their levels in the body while also improving their antioxidant activity [81, 105]. Flavonols can also stimulate the production of antioxidant enzymes such as superoxide dismutase (SOD) and catalase in the body [22, 81]. These enzymes are critical in preventing oxidative stress and lowering the risk of chronic illnesses.
Anti-neurodegenerative agents
Several studies have shown that flavonols benefit brain health, especially in preventing and treating neurodegenerative diseases like Alzheimer’s and Parkinson’s [72, 100]. Flavonols have strong antioxidant qualities that can help protect brain cells from oxidative stress, which has been linked to the onset and progression of neurodegenerative disorders [7, 40]. Flavonols’ anti-inflammatory qualities can aid in the reduction of inflammation in the brain [97]. The flavonol fisetin was discovered to lower brain inflammation and enhance cognitive performance [97]. Kaempferol has neuroprotective properties and might be utilized to protect neurons from injury [171]. As a result, it was discovered to preserve neurons in the brain and increase cognitive performance. By enhancing the production of brain-derived neurotrophic factor (BDNF), a protein involved in the development and survival of neurons, quercetin was discovered to increase spatial memory and learning [140]. Flavonols have anti-neurodegenerative properties through acting as antioxidants, lowering inflammation, providing neuroprotection, and boosting memory and learning.
Anti-inflammatory agents
Flavonols are being investigated extensively for their possible anti-inflammatory effects. Flavonols contain a number of anti-inflammatory qualities, including the ability to block pro-inflammatory enzymes, reduce cytokine synthesis, modulate immune cell activity, and inhibit the NF-B pathway. Flavonols have been shown to reduce the activity of pro-inflammatory enzymes such cyclooxygenase (COX) and lipoxygenase (LOX). These enzymes are essential in the synthesis of inflammatory mediators such as prostaglandins and leukotrienes. Flavonols like quercetin and kaempferol have been demonstrated to decrease COX and LOX activity, which can lower inflammatory mediator production and relieve inflammation [47]. Flavonols have also been shown to inhibit the production of pro-inflammatory cytokines including tumor necrosis factor-alpha (TNF-) and interleukin-6 (IL-6) [162]. These cytokines are important in the development of chronic inflammation, and inhibiting them with flavonols can help reduce inflammation [37, 98]. Flavonols have been proved to influence the activity of inflammatory immune cells such as macrophages and T lymphocytes [109]. Flavonols like quercetin and kaempferol can block macrophage activation, which reduces the generation of pro-inflammatory mediators. Flavonols can also affect T cell activity, which is important in the progression of chronic inflammation [109]. The nuclear factor-kappa B (NF-kB) pathway is an important inflammatory regulator, and its activation can result in the creation of pro-inflammatory mediators [155]. Flavonols like quercetin and kaempferol have been demonstrated to block the NF-B pathway, reducing the generation of pro-inflammatory mediators and alleviating inflammation [155].
Anti-osteoporotic agents
Flavonols can promote bone mineralization by stimulating osteoblasts, the cells responsible for bone production [124]. In research, flavonol quercetin boosted bone density and structure by promoting bone production [166]. Furthermore, Flavonols have the ability to suppress osteoclasts, the cells responsible for bone resorption, and hence prevent bone loss [124]. In research, flavonol kaempferol was discovered to decrease bone resorption and enhance the bone density by decreasing osteoclast activity. Quercetin’s anti-inflammatory effects decrease bone tissue inflammation and help prevent bone loss caused by estrogen shortage [124, 166]. Myricetin’s anti-oxidative property performs the same thing by lowering oxidative stress [146]. However, further study is needed to discover the appropriate amount and duration of flavonol supplementation in people to prevent and cure osteoporosis.
Hepatoprotective agents
Flavonoids have also been shown to have potential hepatoprotective properties and may help to prevent or treat liver damage and disease. The liver is an important organ that aids in detoxification and metabolism, and it is vulnerable to harm from many poisons and medicines. Silymarin, a flavonoid derived from milk thistle, has been shown to have potent hepatoprotective effects by reducing oxidative stress, inflammation, and liver fibrosis [2, 102]. Flavonols are powerful antioxidants that may scavenge free radicals and protect liver cells from oxidative damage [81]. Oxidative stress is a key source of liver damage and inflammation, and flavonols like quercetin and kaempferol have been shown in animal experiments to protect against oxidative stress-induced liver damage [36, 109, 152]. Chronic inflammation can contribute to the development of liver disease, and flavonols have been discovered to have anti-inflammatory characteristics that can aid in the relief of liver inflammation [6]. Flavonols like quercetin and apigenin have been shown in animal experiments to decrease liver inflammation by blocking the generation of pro-inflammatory cytokines and the activation of inflammatory cells [176]. Liver fibrosis is a frequent complication of chronic liver illness and is defined by the buildup of extracellular matrix proteins, which can compromise liver function. Flavonols with antifibrotic characteristics [11, 40, 134], such as quercetin and catechin, have been discovered to limit the deposition of extracellular matrix proteins and diminish liver fibrosis. Flavonols have been discovered to modulate lipid metabolism and enhance lipid profiles, which can contribute to the development of fatty liver disease. Flavonols such as quercetin and kaempferol have been shown to minimize liver fat buildup and enhance lipid profiles by influencing the expression of genes involved in lipid metabolism [6].
Antimalarial agents
Recent studies supported that several flavonols have potential applications regarding antimalarial response. Quercetin and kaempferol are the most extensively studied for their antimalarial properties [7, 119, 134]. Research showed they could inhibit the growth of Plasmodium falciparum (malaria parasite) both in vivo and in vitro [45, 115]. In addition, fisetin with quercetin and kaempferol are individually capable of inhibiting the formation of hemozoin [45, 115, 119, 182, 183] resulting in the parasite’s death. Moreover, quercetin, fisetin, and kaempferol’s antioxidant and anti-inflammatory roles were proved beneficial for malaria treatments. Another flavonol named myricetin has been proved to suppress Plasmodium falciparum development in vitro [146]. Myricetin accomplishes this by impairing the parasite’s capacity to use glucose, which is critical for survival. Furthermore, myricetin possesses antioxidant and anti-inflammatory characteristics that may be useful in the treatment of malaria [143]. Rutin is a flavonoid glycoside that has been proved to have antimalarial properties. Rutin does this by reducing the development of the Plasmodium berghei malaria parasite in vivo [5, 113]. Rutin does not appear to influence hemozoin production, although its antimalarial mechanism is unknown.
Future perspectives of Flavonols
Flavonols are a kind of flavonoid that has been intensively researched for their possible health advantages. Flavonols have been demonstrated in recent study to have a wide variety of uses, including the prevention and treatment of chronic illnesses.
Studies have shown flavonols to enhance endothelial function, decrease inflammation, and lower blood pressure, all of which are vital factors in preventing cardiovascular disease[7, 134]. Furthermore, flavonols may help prevent and manage type 2 diabetes by enhancing insulin sensitivity and decreasing inflammation [40]. New flavonol derivatives with better drug activity and pharmacokinetic characteristics have been developed thanks to advances in chemical synthesis and structural modification methods [113, 115]. These new flavonols may have the ability to overcome current medication limitations such as drug resistance and toxicity. Another interesting application for flavonols is cancer prevention and therapy [131]. Flavonols have been found in recent research to decrease cancer cell proliferation and trigger death in cancer cells, making them a promising choice for cancer therapy. Combining flavonols with other anticancer medicines may increase effectiveness and decrease drug resistance. This method might be employed more widely in the future to increase the efficacy of flavonols as anticancer medicines [39, 78, 151]. Furthermore, flavonols may have chemopreventive characteristics, lowering the risk of cancer development.
Finally, flavonols have been proved to have neuroprotective properties as well as the potential to boost cognitive performance [72, 100]. According to recent study, flavonols can improve memory, attention, and executive function, making them a possible therapy for neurodegenerative disorders like Alzheimer’s [72, 100]. Furthermore, flavonols may help protect the brain from oxidative stress and inflammation-related damage [78]. Flavonols may be used in conjunction with other antimalarial medications as an adjuvant treatment to minimize inflammation and oxidative stress associated with the condition [45, 115].
Conclusions
In the medicinal world, many drug compounds have significant mechanistic properties against various diseases. Most of these compounds are naturally acquired and have anticancer, antiviral, and antibacterial properties. Quercetin, myricetin, kaempferol, fisetin, rutin, and astragalin are examples of flavonols with high functionality, among other phytochemicals. Kaempferol, quercetin, fisetin, and myricetin show anticancer activity, and kaempferol is effective against H. pylori, a bacterium responsible for human gastric cancer. In addition, quercetin acts as an antioxidant that inhibits many enzymes, and kaempferol improves cell viability and prevents pancreatic beta-cell apoptosis. Kaempferol can also improve the function, survival rate, and insulin secretion of pancreatic beta-cells, supporting its antidiabetic properties.
Moreover, some flavonols block viral entry into the host cells and inhibit viral replication. There is also evidence that plant-derived flavonols might play a vital role in combating bacteria, providing alternative drug treatment. Based on these findings, we can conclude that flavonols have diverse biological properties of medicinal importance against various notorious diseases.
Acknowledgements
Not applicable.
Abbreviations
- UV
Ultraviolet
- ROS
Reactive oxygen species
- MYB
Myeloblastosis
- AR
Androgen receptor
- SGLT1
Sodium-glucose cotransporter 1
- GLUT2
Glucose transporter 2
- 3CLpro
3C-like protease
- Sp1
Specificity protein 1
- PI3K/Akt
Phosphoinositide 3-kinase/Ak strain transforming
- PSA
Prostate-specific antigen
- HK2
Human protein kallikrein 2
- LNCaP
Lymph node carcinoma of the prostate
- ERK
Extracellular signal-regulated kinase
- Bcl-2
B-cell lymphoma-2
- MMP
Mitochondrial membrane potential
- LLC-MK2
Lilly Laboratories Culture-Monkey Kidney 2
- MMP-2
Matrix metalloprotein-2
- AP-1
Activator protein-1
- EGFR
Epidermal growth factor receptor
- T1DM
Type 1 diabetes mellitus
- AMP
Adenosine monophosphate
- ATP
Adenosine triphosphate
- Nrf2-ARE
Nuclear erythroid 2-related factor 2-antioxidant response element
- SARS-CoV-2
Severe acute respiratory syndrome coronavirus-2
- NLRP3
Nod-like receptor family and pyrin domain-containing 3
- Nsps
Nonstructural proteins
- PLpro
Papain-like protease
- RdRp
RNA-dependent RNA polymerase
- ACE2
Angiotensin-converting enzyme 2
- RDDP
RNA-dependent DNA polymerase
- HEK
Human embryonic kidney cells
Author contributions
All authors confirm that the concept, design, data collection, analysis and interpretation, and writing are our own. We ensured that no other contributors were present. All authors read and approved the final manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Availability of data and materials
All data are available within this manuscript.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that there are no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Asif Shahriar, Email: abasifbl@gmail.com.
Md. Furkanur Rahaman Mizan, Email: rahaman4162@cau.ac.kr.
References
- 1.Adamczak A, Ożarowski M, Karpiński TM. Antibacterial activity of some flavonoids and organic acids widely distributed in plants. J Clin Med. 2019;9(1):109. doi: 10.3390/jcm9010109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Aghemo A, Alekseeva OP, Angelico F, Bakulin IG, Bakulina NV, Bordin D, Bueverov AO, Drapkina OM, Gillessen A, Kagarmanova EM, Korochanskaya NV, Kucheryavii UA, Lazebnik LB, Livzan MA, Maev IV, Martynov AI, Osipenko MF, Sas EI, Starodubova A, Yakovlev AA. Role of silymarin as antioxidant in clinical management of chronic liver diseases: a narrative review. Annals Med. 2022;54(1):1548–1560. doi: 10.1080/07853890.2022.2069854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Agrawal PK, Agrawal C, Blunden G. Quercetin: antiviral significance and possible COVID-19 integrative considerations. Nat Prod Commun. 2020 doi: 10.1177/1934578X20976293. [DOI] [Google Scholar]
- 4.Akomolafe A, Beiser A, Meigs JB, Au R, Green RC, Farrer LA, Wolf PA, Seshadri S. Diabetes mellitus and risk of developing Alzheimer disease: results from the Framingham Study. Arch Neurol. 2006;63(11):1551–1555. doi: 10.1001/archneur.63.11.1551. [DOI] [PubMed] [Google Scholar]
- 5.Al-Dhabi NA, Arasu MV, Park CH, Park SU (2015) An up-to-date review of rutin and its biological and pharmacological activities. EXCLI Journal. 10.17179/EXCLI2014-663 [DOI] [PMC free article] [PubMed]
- 6.Alkandahri MY, Pamungkas BT, Oktoba Z, Shafirany MZ, Sulastri L, Arfania M, Anggraeny EN, Pratiwi A, Astuti FD, Indriyani Dewi SY, Hamidah SZ. Hepatoprotective effect of kaempferol: a review of the dietary sources, bioavailability, mechanisms of action, and safety. Adv Pharmacol Pharm Sci. 2023 doi: 10.1155/2023/1387665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anand David AV, Arulmoli R, Parasuraman S. Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev. 2016;10(20):84. doi: 10.4103/0973-7847.194044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Appelbaum PC. 2012 and beyond: potential for the start of a second pre-antibiotic era? J Antimicrob Chemother. 2012;67(9):2062–2068. doi: 10.1093/jac/dks213. [DOI] [PubMed] [Google Scholar]
- 9.Arafah A, Rehman MU, Mir TM, Wali AF, Ali R, Qamar W, Khan R, Ahmad A, Aga SS, Alqahtani S, Almatroudi NM. Multi-therapeutic potential of naringenin (4′,5,7-trihydroxyflavonone): experimental evidence and mechanisms. Plants. 2020;9(12):1784. doi: 10.3390/PLANTS9121784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Axon A. Helicobacter pylori and public health. Helicobacter. 2014;19(S1):68–73. doi: 10.1111/hel.12155. [DOI] [PubMed] [Google Scholar]
- 11.Bae J, Kim N, Shin Y, Kim S-Y, Kim Y-J. Activity of catechins and their applications. Biomed Dermatol. 2020 doi: 10.1186/S41702-020-0057-8. [DOI] [Google Scholar]
- 12.Benga L, Fulde M, Neis C, Goethe R, Valentin-Weigand P. Polysaccharide capsule and suilysin contribute to extracellular survival of Streptococcus suis co-cultivated with primary porcine phagocytes. Vet Microbiol. 2008;132(1–2):211–219. doi: 10.1016/j.vetmic.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 13.Bennett NC, Gardiner RA, Hooper JD, Johnson DW, Gobe GC. Molecular cell biology of androgen receptor signalling. Int J Biochem Cell Biol. 2010;42(6):813–827. doi: 10.1016/j.biocel.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 14.Boam T. Anti-androgenic effects of flavonols in prostate cancer. Ecancermedicalscience. 2015;9:1–8. doi: 10.3332/ecancer.2015.585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boonmasawai S, Sariya L, Leesombun A, Chaichoun K. Anti-proliferative and total ERK1/2 inhibitory effects of plant flavonols on Human cervical cancer (HeLa) cells. Thai J Veterinary Med. 2018;48(4):541–549. [Google Scholar]
- 16.Bracarda S, De Cobelli O, Greco C, Prayer-Galetti T, Valdagni R, Gatta G, De Braud F, Bartsch G. Cancer of the prostate. Crit Rev Oncol Hematol. 2005;56(3):379–396. doi: 10.1016/j.critrevonc.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 17.Buer CS, Imin N, Djordjevic MA. Flavonoids: new roles for old molecules. J Integr Plant Biol. 2010;52(1):98–111. doi: 10.1111/j.1744-7909.2010.00905.x. [DOI] [PubMed] [Google Scholar]
- 18.Bule M, Abdurahman A, Nikfar S, Abdollahi M, Amini M. Antidiabetic effect of quercetin: a systematic review and meta-analysis of animal studies. Food Chem Toxicol. 2019;125:494–502. doi: 10.1016/j.fct.2019.01.037. [DOI] [PubMed] [Google Scholar]
- 19.Cagnol S, Chambard JC. ERK and cell death: mechanisms of ERK-induced cell death - apoptosis, autophagy and senescence. FEBS J. 2010;277(1):2–21. doi: 10.1111/j.1742-4658.2009.07366.x. [DOI] [PubMed] [Google Scholar]
- 20.Calderón-Oliver M, Ponce-Alquicira E (2018) Fruits: a source of polyphenols and health benefits. In: Natural and Artificial Flavoring Agents and Food Dyes, pp 189–228. 10.1016/B978-0-12-811518-3.00007-7
- 21.Camargo MC, García A, Riquelme A, Otero W, Camargo CA, Hernandez-García T, Candia R, Bruce MG, Rabkin CS. The problem of helicobacter pylori resistance to antibiotics: a systematic review in latin America. Am J Gastroenterol. 2014;109(4):485–495. doi: 10.1038/ajg.2014.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Carlsen MH, Halvorsen BL, Holte K, Bøhn SK, Dragland S, Sampson L, Willey C, Senoo H, Umezono Y, Sanada C, Barikmo I, Berhe N, Willett WC, Phillips KM, Jacobs DR, Blomhoff R. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutrit J. 2010 doi: 10.1186/1475-2891-9-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chabot-Roy G, Willson P, Segura M, Lacouture S, Gottschalk M. Phagocytosis and killing of Streptococcus suis by porcine neutrophils. Microb Pathog. 2006;41(1):21–32. doi: 10.1016/j.micpath.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 24.Chang HK, Kim DS, Chae JJ, Kim M, Myong J, Lee KH, Lee MW, Park TC. Inhibition of ERK activity enhances the cytotoxic effect of peroxisome proliferator-activated receptor γ ( PPARγ) agonists in HeLa cells. Biochem Biophys Res Commun. 2016 doi: 10.1016/j.bbrc.2016.11.122. [DOI] [PubMed] [Google Scholar]
- 25.Chen HJ, Lin CM, Lee CY, Shih NC, Peng SF, Tsuzuki M, Amagaya S, Huang WW, Yang JS. Kaempferol suppresses cell metastasis via inhibition of the ERK-p38-JNK and AP-1 signaling pathways in U-2 OS human osteosarcoma cells. Oncol Rep. 2013;30(2):925–932. doi: 10.3892/or.2013.2490. [DOI] [PubMed] [Google Scholar]
- 26.Chen H, Wang W, Yu S, Wang H, Tian Z, Zhu S. Procyanidins and their therapeutic potential against oral diseases. Molecules. 2022 doi: 10.3390/molecules27092932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cheng XQ, Chen X, Hughes RA, Williams SJ, Woodman OL. Understanding the cardioprotective effects of flavonols: discovery of relaxant flavonols without antioxidant activity. J Med Chem. 2008;51(6):1874–1884. doi: 10.1021/jm070352h. [DOI] [PubMed] [Google Scholar]
- 28.Čižmárová B, Hubková B, Tomečková V, Birková A. Flavonoids as promising natural compounds in the prevention and treatment of selected skin diseases. Int J Mole Sci. 2023 doi: 10.3390/ijms24076324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Close DC, Mcarthur C (2014) Rethinking the role of many plant phenolics : protection from photodamage not herbivores ? - protection from the role of many plant phenolics Rethinking not herbivores ? photodamage. 99(March), 166–172
- 30.Cogolludo A, Frazziano G, Briones AM, Cobeño L, Moreno L, Lodi F, Salaices M, Tamargo J, Perez-Vizcaino F. The dietary flavonoid quercetin activates BKCa currents in coronary arteries via production of H2O2. Role Vasodilatation Cardiovasc Res. 2007;73(2):424–431. doi: 10.1016/j.cardiores.2006.09.008. [DOI] [PubMed] [Google Scholar]
- 31.Colunga Biancatelli RML, Berrill M, Catravas JD, Marik PE. Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19) Front Immunol. 2020;11:1451. doi: 10.3389/fimmu.2020.01451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cushnie TPT, Hamilton VES, Chapman DG, Taylor PW, Lamb AJ. Aggregation of Staphylococcus aureus following treatment with the antibacterial flavonol galangin. J Appl Microbiol. 2007;103(5):1562–1567. doi: 10.1111/j.1365-2672.2007.03393.x. [DOI] [PubMed] [Google Scholar]
- 33.Cushnie TPT, Lamb AJ. Antimicrobial activity of flavonoids. Int J Antimicrob Agents. 2005;26(5):343–356. doi: 10.1016/j.ijantimicag.2005.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.David AVA, Arulmoli R, Parasuraman S. Overviews of biological importance of quercetin: a bioactive flavonoid. Pharmacogn Rev. 2016;10(20):84. doi: 10.4103/0973-7847.194044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Deng Z, Hassan S, Rafiq M, Li H, He Y, Cai Y, Kang X, Liu Z, Yan T. Pharmacological activity of eriodictyol: the major natural polyphenolic flavanone. Evid Based Complement Alternat Med ECAM. 2020 doi: 10.1155/2020/6681352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Drouet S, Hano C, Drouet S, Hano C. Biosynthesis and regulation of antioxidant flavonolignans in milk thistle. Antioxid Benefits Sour Mech Action. 2021 doi: 10.5772/INTECHOPEN.96783. [DOI] [Google Scholar]
- 37.Duda-Chodak A, Tarko T. Possible side effects of polyphenols and their interactions with medicines. Molecules. 2023;28(6):2536. doi: 10.3390/molecules28062536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ebrahimpour S, Zakeri M, Esmaeili A. Crosstalk between obesity, DIABETES, and Alzheimer’s disease: introducing quercetin as an effective triple herbal medicine. Ageing Res Rev. 2020 doi: 10.1016/j.arr.2020.101095. [DOI] [PubMed] [Google Scholar]
- 39.Elbatreek MH, Mahdi I, Ouchari W, Mahmoud MF, Sobeh M. Current advances on the therapeutic potential of pinocembrin: an updated review. Biomed Pharmacother. 2023;157:114032. doi: 10.1016/J.BIOPHA.2022.114032. [DOI] [PubMed] [Google Scholar]
- 40.El-Saber Batiha G, Beshbishy AM, Ikram M, Mulla ZS, Abd El-Hack ME, Taha AE, Algammal AM, Ali Elewa YH. The pharmacological activity, biochemical properties, and pharmacokinetics of the major natural polyphenolic flavonoid: quercetin. Foods. 2020;9(3):374. doi: 10.3390/FOODS9030374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Escandón RA, del Campo M, López-Solis R, Obreque-Slier E, Toledo H. Antibacterial effect of kaempferol and (−)-epicatechin on Helicobacter pylori. Eur Food Res Technol. 2016;242(9):1495–1502. doi: 10.1007/s00217-016-2650-z. [DOI] [Google Scholar]
- 42.Farhadi F, Khameneh B, Iranshahi M, Iranshahy M. Antibacterial activity of flavonoids and their structure–activity relationship: an update review. Phytother Res. 2019;33(1):13–40. doi: 10.1002/ptr.6208. [DOI] [PubMed] [Google Scholar]
- 43.Fittipaldi N, Segura M, Grenier D, Gottschalk M. Virulence factors involved in the pathogenesis of the infection caused by the swine pathogen and zoonotic agent Streptococcus suis. Future Microbiol. 2012;7(2):259–279. doi: 10.2217/fmb.11.149. [DOI] [PubMed] [Google Scholar]
- 44.Fontana F, Raimondi M, Marzagalli M, Di Domizio A, Limonta P. Natural compounds in prostate cancer prevention and treatment: mechanisms of action and molecular targets. Cells. 2020;9(2):460. doi: 10.3390/cells9020460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ganesh D, Fuehrer HP, Starzengrüber P, Swoboda P, Khan WA, Reismann JAB, Mueller MSK, Chiba P, Noedl H. Antiplasmodial activity of flavonol quercetin and its analogues in Plasmodium falciparum: evidence from clinical isolates in Bangladesh and standardized parasite clones. Parasitol Res. 2012;110(6):2289–2295. doi: 10.1007/S00436-011-2763-Z. [DOI] [PubMed] [Google Scholar]
- 46.Ganeshpurkar A, Saluja AK. The pharmacological potential of rutin. Saudi Pharmaceutical Journal. 2017;25(2):149–164. doi: 10.1016/J.JSPS.2016.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.García-Lafuente A, Guillamón E, Villares A, Rostagno MA, Martínez JA. Flavonoids as anti-inflammatory agents: implications in cancer and cardiovascular disease. Inflammat Res. 2009;58(9):537–552. doi: 10.1007/S00011-009-0037-3. [DOI] [PubMed] [Google Scholar]
- 48.Gasmi J, Sanderson JT. Growth inhibitory, antiandrogenic, and pro-apoptotic effects of punicic acid in LNCaP human prostate cancer cells. J Agric Food Chem. 2010;58(23):12149–12156. doi: 10.1021/jf103306k. [DOI] [PubMed] [Google Scholar]
- 49.Ghafouri-Fard S, Shabestari FA, Vaezi S, Abak A, Shoorei H, Karimi A, Taheri M, Basiri A. Emerging impact of quercetin in the treatment of prostate cancer. Biomed Pharmacother. 2021;138:111548. doi: 10.1016/j.biopha.2021.111548. [DOI] [PubMed] [Google Scholar]
- 50.Górniak I, Bartoszewski R, Króliczewski J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem Rev. 2019 doi: 10.1007/s11101-018-9591-z. [DOI] [Google Scholar]
- 51.Gottschalk M, Xu J, Calzas C, Segura M. Streptococcus suis: a new emerging or an old neglected zoonotic pathogen? Future Microbiol. 2010;5(3):371–391. doi: 10.2217/fmb.10.2. [DOI] [PubMed] [Google Scholar]
- 52.Gupta A, Birhman K, Raheja I, Sharma SK, Kar HK. Quercetin: a wonder bioflavonoid with therapeutic potential in disease management. Asian Pacific J Trop Dis. 2016;6(3):248–252. doi: 10.1016/S2222-1808(15)61024-6. [DOI] [Google Scholar]
- 53.Hanhineva K, Törrönen R, Bondia-Pons I, Pekkinen J, Kolehmainen M, Mykkänen H, Poutanen K. Impact of dietary polyphenols on carbohydrate metabolism. Int J Mol Sci. 2010;11(4):1365–1402. doi: 10.3390/ijms11041365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Haskell-Ramsay CF, Schmitt J, Actis-Goretta L. The impact of epicatechin on human cognition: the role of cerebral blood flow. Nutrients. 2018;10(8):986. doi: 10.3390/NU10080986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hernández I, Alegre L, Van Breusegem F, Munné-Bosch S. How relevant are flavonoids as antioxidants in plants? Trends Plant Sci. 2009;14(3):125–132. doi: 10.1016/j.tplants.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 56.Hichri I, Barrieu F, Bogs J, Kappel C, Delrot S, Lauvergeat V. Recent advances in the transcriptional regulation of the flavonoid biosynthetic pathway. J Exp Bot. 2011;62(8):2465–2483. doi: 10.1093/jxb/erq442. [DOI] [PubMed] [Google Scholar]
- 57.Higa S, Hirano T, Kotani M, Matsumoto M, Fujita A, Suemura M, Kawase I, Tanaka T. Fisetin, a flavonol, inhibits TH2-type cytokine production by activated human basophils. J Allergy Clin Immunol. 2003;111(6):1299–1306. doi: 10.1067/mai.2003.1456. [DOI] [PubMed] [Google Scholar]
- 58.Hossion AML, Zamami Y, Kandahary RK, Tsuchiya T, Ogawa W, Iwado A, Sasaki K. Quercetin diacylglycoside analogues showing dual inhibition of DNA gyrase and topoisomerase IV as novel antibacterial agents. J Med Chem. 2011;54(11):3686–3703. doi: 10.1021/jm200010x. [DOI] [PubMed] [Google Scholar]
- 59.Huang C-C, Chung C-M, Leu H-B, Lin L-Y, Chiu C-C, Hsu C-Y, Chiang C-H, Huang P-H, Chen T-J, Lin S-J. Diabetes mellitus and the risk of Alzheimer’s disease: a nationwide population-based study. PLoS ONE. 2014;9(1):e87095. doi: 10.1371/journal.pone.0087095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Huang YH, Huang CC, Chen CC, Yang KJ, Huang CY. Inhibition of Staphylococcus aureus PriA Helicase by Flavonol Kaempferol. Protein J. 2015;34(3):169–172. doi: 10.1007/s10930-015-9609-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hussain T, Tan B, Murtaza G, Liu G, Rahu N, Kalhoro MS, Kalhoro DH, Adebowale TO, Mazhar MU, ur Rehman, Z. Flavonoids and type 2 diabetes: evidence of efficacy in clinical and animal studies and delivery strategies to enhance their therapeutic efficacy. Pharmacol Res. 2020;152:104629. doi: 10.1016/j.phrs.2020.104629. [DOI] [PubMed] [Google Scholar]
- 62.Ibarra M, Moreno L, Vera R, Cogolludo A, Duarte J, Tamargo J, Perez-Vizcaino F. Effects of the flavonoid quercetin and its methylated metabolite Isorhamnetin in isolated arteries from spontaneously hypertensive rats. Planta Med. 2003;69(11):995–1000. doi: 10.1055/s-2003-45144. [DOI] [PubMed] [Google Scholar]
- 63.Ibarra M, Pérez-Vizcaíno F, Cogolludo A, Duarte J, Zaragozá-Arnáez F, López-López JG, Tamargo J. Cardiovascular effects of isorhamnetin and quercetin in isolated rat and porcine vascular smooth muscle and isolated rat atria. Planta Med. 2002;68(4):307–310. doi: 10.1055/s-2002-26752. [DOI] [PubMed] [Google Scholar]
- 64.Imran M, Rauf A, Shah ZA, Saeed F, Imran A, Arshad MU, Ahmad B, Bawazeer S, Atif M, Peters DG. Chemo-preventive and therapeutic effect of the dietary flavonoid kaempferol: a comprehensive review. Phytother Res. 2019;33(2):263–275. doi: 10.1002/ptr.6227. [DOI] [PubMed] [Google Scholar]
- 65.Imran M, Salehi B, Sharifi-Rad J, Aslam Gondal T, Saeed F, Imran A, Shahbaz M, Tsouh Fokou PV, Umair Arshad M, Khan H. Kaempferol: a key emphasis to its anticancer potential. Molecules. 2019;24(12):2277. doi: 10.3390/molecules24122277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Irmisch S, Ruebsam H, Jancsik S, Saint Yuen MM, Madilao LL, Bohlmann J. Flavonol biosynthesis genes and their use in engineering the plant antidiabetic metabolite montbretin A. Plant Physiol. 2019;180(3):1277–1290. doi: 10.1104/pp.19.00254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jaisinghani RN. Antibacterial properties of quercetin. Microbiol Res. 2017 doi: 10.4081/mr.2017.6877. [DOI] [Google Scholar]
- 68.Jeong JC, Kim MS, Kim TH, Kim YK. Kaempferol induces cell death through ERK and Akt-dependent down-regulation of XIAP and survivin in human glioma cells. Neurochem Res. 2009;34(5):991–1001. doi: 10.1007/s11064-008-9868-5. [DOI] [PubMed] [Google Scholar]
- 69.Jomová K, Hudecova L, Lauro P, Simunkova M, Alwasel SH, Alhazza IM, Valko M. A switch between antioxidant and prooxidant properties of the phenolic compounds myricetin, morin, 3’,4’-dihydroxyflavone, taxifolin and 4-hydroxy-coumarin in the presence of copper(II) ions: a spectroscopic, absorption titration and DNA damage study. Molecules. 2019 doi: 10.3390/molecules24234335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Joshi T, Joshi T, Sharma P, Mathpal S, Pundir H, Bhatt V, Chandra S. In silico screening of natural compounds against COVID-19 by targeting Mpro and ACE2 using molecular docking. Eur Rev Med Pharmacol Sci. 2020;24(8):4529–4536. doi: 10.26355/eurrev_202004_21036. [DOI] [PubMed] [Google Scholar]
- 71.Kang JW, Kim JH, Song K, Kim SH, Yoon J-H, Kim K-S. Kaempferol and quercetin, components of Ginkgo biloba extract (EGb 761), induce caspase-3-dependent apoptosis in oral cavity cancer cells. Phytother Res. 2010;24:S77–S82. doi: 10.1002/ptr.2913. [DOI] [PubMed] [Google Scholar]
- 72.Kaur R, Sood A, Lang DK, Bhatia S, Al-Harrasi A, Aleya L, Behl T. Potential of flavonoids as anti-Alzheimer’s agents: bench to bedside. Environ Sci Pollut Res. 2022;29(18):26063–26077. doi: 10.1007/S11356-021-18165-Z. [DOI] [PubMed] [Google Scholar]
- 73.Khan N, Asim M, Afaq F, Zaid MA, Mukhtar H. A novel dietary flavonoid fisetin inhibits androgen receptor signaling and tumor growth in athymic nude mice. Can Res. 2008;68(20):8555–8563. doi: 10.1158/0008-5472.CAN-08-0240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Khazeei Tabari MA, Iranpanah A, Bahramsoltani R, Rahimi R. Flavonoids as promising antiviral agents against SARS-CoV-2 infection: a mechanistic review. Molecules. 2021;26(13):3900. doi: 10.3390/molecules26133900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Khoo NKH, White CR, Pozzo-Miller L, Zhou F, Constance C, Inoue T, Patel RP, Parks DA. Dietary flavonoid quercetin stimulates vasorelaxation in aortic vessels. Free Radical Biol Med. 2010;49(3):339–347. doi: 10.1016/j.freeradbiomed.2010.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ki WL, Nam JK, Heo YS, Rogozin EA, Pugliese A, Mun KH, Bowden GT, Bode AM, Hyong JL, Dong Z. Raf and MEK protein kinases are direct molecular targets for the chemopreventive effect of quercetin, a major flavonol in red wine. Can Res. 2008;68(3):946–955. doi: 10.1158/0008-5472.CAN-07-3140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kim JD, Liu L, Guo W, Meydani M. Chemical structure of flavonols in relation to modulation of angiogenesis and immune-endothelial cell adhesion. J Nutr Biochem. 2006;17(3):165–176. doi: 10.1016/j.jnutbio.2005.06.006. [DOI] [PubMed] [Google Scholar]
- 78.Kim JM, Heo HJ. The roles of catechins in regulation of systemic inflammation. Food Sci Biotechnol. 2022;31(8):957–970. doi: 10.1007/S10068-022-01069-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.King A, Young G. Characteristics and occurrence of phenolic phytochemicals. J Am Diet Assoc. 1999;99(2):213–218. doi: 10.1016/S0002-8223(99)00051-6. [DOI] [PubMed] [Google Scholar]
- 80.Kitahata MM, Koepsell TD, Deyo RA, Maxwell CL, Dodge WT, Wagner EH. Physicians’ experience with the acquired immunodeficiency syndrome as a factor in patients’ survival. N Engl J Med. 1996;334(11):701–707. doi: 10.1056/NEJM199603143341106. [DOI] [PubMed] [Google Scholar]
- 81.Kotha RR, Tareq FS, Yildiz E, Luthria DL. Oxidative stress and antioxidants-a critical review on in vitro antioxidant assays. Antioxidants. 2022 doi: 10.3390/ANTIOX11122388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Kubina R, Iriti M, Kabala-Dzik A. Anticancer potential of selected Flavonols: fisetin, kaempferol, and quercetin on head and neck cancers. Nutrients. 2021;13(3):1–20. doi: 10.3390/nu13030845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kumar RM, Kumar H, Bhatt T, Jain R, Panchal K, Chaurasiya A, Jain V. Fisetin in cancer: attributes, developmental aspects, and nanotherapeutics. Pharmaceuticals. 2023;16(2):196. doi: 10.3390/PH16020196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kurutas EB. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: current state. Nutr J. 2015;15(1):1–22. doi: 10.1186/s12937-016-0186-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lee KW, Kang NJ, Rogozin EA, Kim HG, Cho YY, Bode AM, Lee HJ, Surh YJ, Bowden GT, Dong Z. Myricetin is a novel natural inhibitor of neoplastic cell transformation and MEK1. Carcinogenesis. 2007;28(9):1918–1927. doi: 10.1093/carcin/bgm110. [DOI] [PubMed] [Google Scholar]
- 86.Leo CH, Woodman OL. Flavonols in the prevention of diabetes-induced vascular dysfunction. J Cardiovasc Pharmacol. 2015;65(6):532–544. doi: 10.1097/FJC.0000000000000180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lepiniec L, Debeaujon I, Routaboul JM, Baudry A, Pourcel L, Nesi N, Caboche M. Genetics and biochemistry of seed flavonoids. Annu Rev Plant Biol. 2006;57:405–430. doi: 10.1146/annurev.arplant.57.032905.105252. [DOI] [PubMed] [Google Scholar]
- 88.Leung LK, Su Y, Chen R, Zhang Z, Huang Y, Chen ZY. Theaflavins in black tea and catechins in green tea are equally effective antioxidants. J Nutr. 2001;131(9):2248–2251. doi: 10.1093/JN/131.9.2248. [DOI] [PubMed] [Google Scholar]
- 89.Lewis DR, Ramirez MV, Miller ND, Vallabhaneni P, Keith Ray W, Helm RF, Winkel BSJ, Muday GK. Auxin and ethylene induce flavonol accumulation through distinct transcriptional networks. Plant Physiol. 2011;156(1):144–164. doi: 10.1104/pp.111.172502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Li J, Xiang H, Huang C, Lu J. Pharmacological actions of myricetin in the nervous system: a comprehensive review of preclinical studies in animals and cell models. Front Pharmacol. 2021;12:3717. doi: 10.3389/FPHAR.2021.797298/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Li T, Wang L, Wu L, Xie Y, Chang M, Wang D, Yi L, Zhu X, Mi M. Integrated metabolomics and network pharmacology investigation of cardioprotective effects of myricetin after 1-week high-intensity exercise. Nutrients. 2023;15(6):1336. doi: 10.3390/nu15061336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Lin CW, Chen PN, Chen MK, Yang WE, Tang CH, Yang SF, Hsieh YS. Kaempferol reduces matrix metalloproteinase-2 expression by down-regulating ERK1/2 and the activator protein-1 signaling pathways in oral cancer cells. PLoS ONE. 2013 doi: 10.1371/journal.pone.0080883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lingwan M, Shagun S, Pant Y, Kumari B, Nanda R, Masakapalli SK (2020) Antiviral phytochemicals identified in Rhododendron arboreum petals exhibited strong binding to SARS-CoV-2 MPro and Human ACE2 receptor
- 94.Lodi F, Jiménez R, Menendez C, Needs PW, Duarte J, Perez-Vizcaino F. Glucuronidated metabolites of the flavonoid quercetin do not auto-oxidise, do not generate free radicals and do not decrease nitric oxide bioavailability. Planta Med. 2008;74(7):741–746. doi: 10.1055/s-2008-1074525. [DOI] [PubMed] [Google Scholar]
- 95.Long P, Rakariyatham K, Ho CT, Zhang L. Thearubigins: formation, structure, health benefit and sensory property. Trends Food Sci Technol. 2023;133:37–48. doi: 10.1016/J.TIFS.2023.01.013. [DOI] [Google Scholar]
- 96.López-López G, Moreno L, Cogolludo A, Galisteo M, Ibarra M, Duarte J, Lodi F, Tamargo J, Perez-Vizcaino F. Nitric oxide (NO) scavenging and NO protecting effects of quercetin and their biological significance in vascular smooth muscle. Mol Pharmacol. 2004;65(4):851–859. doi: 10.1124/mol.65.4.851. [DOI] [PubMed] [Google Scholar]
- 97.Maher P, Dargusch R, Ehren JL, Okada S, Sharma K, Schubert D. Fisetin lowers methylglyoxal dependent protein glycation and limits the complications of diabetes. PLoS ONE. 2011;6(6):21226. doi: 10.1371/JOURNAL.PONE.0021226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mansuri ML, Parihar P, Solanki I, Parihar MS. Flavonoids in modulation of cell survival signalling pathways. Genes Nutr. 2014;9(3):400. doi: 10.1007/s12263-014-0400-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Miltonprabu S (2019) Quercetin: a flavonol with versatile therapeutic applications and its interactions with other drugs. In: Nonvitamin and Nonmineral Nutritional Supplements, pp. 75–83. Elsevier
- 100.Minocha T, Birla H, Obaid AA, Rai V, Sushma P, Shivamallu C, Moustafa M, Al-Shehri M, Al-Emam A, Tikhonova MA, Yadav SK, Poeggeler B, Singh D, Singh SK. Flavonoids as promising neuroprotectants and their therapeutic potential against alzheimer’s disease. Oxidative Med Cell Longev. 2022 doi: 10.1155/2022/6038996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Mouffouk C, Mouffouk S, Mouffouk S, Hambaba L, Haba H. Flavonols as potential antiviral drugs targeting SARS-CoV-2 proteases (3CLpro and PLpro), spike protein, RNA-dependent RNA polymerase (RdRp) and angiotensin-converting enzyme II receptor (ACE2) Eur J Pharmacol. 2021;891:173759. doi: 10.1016/j.ejphar.2020.173759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mukhtar S, Xiaoxiong Z, Qamer S, Saad M, Mubarik MS, Mahmoud AH, Mohammed OB. Hepatoprotective activity of silymarin encapsulation against hepatic damage in albino rats. Saudi J Biol Sci. 2021;28(1):717–723. doi: 10.1016/J.SJBS.2020.10.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Musial C, Kuban-Jankowska A, Gorska-Ponikowska M. Beneficial properties of green tea catechins. Int J Mole Sci. 2020 doi: 10.3390/IJMS21051744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Nagini S. Carcinoma of the stomach: a review of epidemiology, pathogenesis, molecular genetics and chemoprevention. World Journal of Gastrointestinal Oncology. 2012;4(7):156. doi: 10.4251/wjgo.v4.i7.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nayak B, Liu RH, Tang J. Effect of processing on phenolic antioxidants of fruits, vegetables, and grains—a review. Crit Rev Food Sci Nutr. 2015;55(7):887–918. doi: 10.1080/10408398.2011.654142. [DOI] [PubMed] [Google Scholar]
- 106.Nguyen TTH, Woo HJ, Kang HK, Nguyen VD, Kim YM, Kim DW, Ahn SA, Xia Y, Kim D. Flavonoid-mediated inhibition of SARS coronavirus 3C-like protease expressed in Pichia pastoris. Biotech Lett. 2012;34(5):831–838. doi: 10.1007/s10529-011-0845-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.O’Connor A, Vaira D, Gisbert JP, O’Morain C. Treatment of Helicobacter pylori Infection 2014. Helicobacter. 2014;19(S1):38–45. doi: 10.1111/hel.12163. [DOI] [PubMed] [Google Scholar]
- 108.Ouyang J, Sun F, Feng W, Sun Y, Qiu X, Xiong L, Liu Y, Chen Y. Quercetin is an effective inhibitor of quorum sensing, biofilm formation and virulence factors in Pseudomonas aeruginosa. J Appl Microbiol. 2016;120(4):966–974. doi: 10.1111/jam.13073. [DOI] [PubMed] [Google Scholar]
- 109.Pandey KB, Rizvi SI. Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev. 2009;2(5):270. doi: 10.4161/OXIM.2.5.9498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Park BS, Choi NE, Lee JH, Kang HM, Yu SB, Kim HJ, Kang HK, Kim IR. Crosstalk between fisetin-induced apoptosis and autophagy in human oral squamous cell carcinoma. J Cancer. 2019;10(1):138–146. doi: 10.7150/jca.28500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Park HH, Lee S, Son HY, Park SB, Kim MS, Choi EJ, Singh TSK, Ha JH, Lee MG, Kim JE. Flavonoids inhibit histamine release and expression of proinflammatory cytokines in mast cells. Arch Pharmacal Res. 2008;31(10):1303–1311. doi: 10.1007/s12272-001-2110-5. [DOI] [PubMed] [Google Scholar]
- 112.Pasetto S, Pardi V, Murata RM. Anti-HIV-1 activity of flavonoid myricetin on HIV-1 infection in a dual-chamber in vitro model. PLoS ONE. 2014;9(12):e115323. doi: 10.1371/journal.pone.0115323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Patel K, Patel DK. The beneficial role of rutin, a naturally occurring flavonoid in health promotion and disease prevention: a systematic review and update. Bioact Food Dietary Interv Arthritis Relat Inflamm Dis. 2019 doi: 10.1016/B978-0-12-813820-5.00026-X. [DOI] [Google Scholar]
- 114.Pedro AC, Maciel GM, Rampazzo Ribeiro V, Haminiuk CWI. Fundamental and applied aspects of catechins from different sources: a review. Int J Food Sci Technol. 2020;55(2):429–442. doi: 10.1111/IJFS.14371. [DOI] [Google Scholar]
- 115.Penna-Coutinho J, Aguiar ACC, Krettli AU. Commercial drugs containing flavonoids are active in mice with malaria and in vitro against chloroquine-resistant plasmodium falciparum. Mem Inst Oswaldo Cruz. 2018 doi: 10.1590/0074-02760180279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Perez-Vizcaino F, Duarte J. Flavonols and cardiovascular disease. Mol Aspects Med. 2010;31(6):478–494. doi: 10.1016/j.mam.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 117.Perez-Vizcaino F, Duarte J, Andriantsitohaina R. Endothelial function and cardiovascular disease: Effects of quercetin and wine polyphenols. Free Radical Res. 2006;40(10):1054–1065. doi: 10.1080/10715760600823128. [DOI] [PubMed] [Google Scholar]
- 118.Pérez-Vizcaíno F, Ibarra M, Cogolludo AL, Duarte J, Zaragozá-Arnáez F, Moreno L, López-López G, Tamargo J. Endothelium-independent vasodilator effects of the flavonoid quercetin and its methylated metabolites in rat conductance and resistance arteries. J Pharmacol Exp Ther. 2002;302(1):66–72. doi: 10.1124/jpet.302.1.66. [DOI] [PubMed] [Google Scholar]
- 119.Periferakis A, Periferakis K, Badarau, I. A., Petran, E. M., Popa, D. C., Caruntu, A., Costache, R. S., Scheau, C., Caruntu, C., & Costache, D. O. (2022). Kaempferol: Antimicrobial Properties, Sources, Clinical, and Traditional Applications. Int J Mole Sci. 10.3390/IJMS232315054 [DOI] [PMC free article] [PubMed]
- 120.Pernin A, Dubois-Brissonnet F, Roux S, Masson M, Bosc V, Maillard MN. Phenolic compounds can delay the oxidation of polyunsaturated fatty acids and the growth of Listeria monocytogenes: structure-activity relationships. J Sci Food Agric. 2018;98(14):5401–5408. doi: 10.1002/jsfa.9082. [DOI] [PubMed] [Google Scholar]
- 121.Pollastri S, Tattini M. Flavonols: old compounds for old roles. Ann Bot. 2011;108(7):1225–1233. doi: 10.1093/aob/mcr234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Prinsloo G, Marokane CK, Street RA. Anti-HIV activity of southern African plants: current developments, phytochemistry and future research. J Ethnopharmacol. 2018;210:133–155. doi: 10.1016/j.jep.2017.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rackal JM, Tynan A, Handford CD, Rzeznikiewiz D, Agha A, Glazier R (2011) Provider training and experience for people living with HIV/AIDS. Cochrane Database of Systematic Reviews, 6 [DOI] [PubMed]
- 124.Ramesh P, Jagadeesan R, Sekaran S, Dhanasekaran A, Vimalraj S. Flavonoids: classification, function, and molecular mechanisms involved in bone remodelling. Front Endocrinol. 2021 doi: 10.3389/FENDO.2021.779638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Rice-Evans C. Flavonoid antioxidants. Curr Med Chem. 2012;8(7):797–807. doi: 10.2174/0929867013373011. [DOI] [PubMed] [Google Scholar]
- 126.Rizzuti B, Grande F, Conforti F, Jimenez-Alesanco A, Ceballos-Laita L, Ortega-Alarcon D, Vega S, Reyburn HT, Abian O, Velazquez-Campoy A. Rutin is a low micromolar inhibitor of SARS-CoV-2 main protease 3CLpro: implications for drug design of quercetin analogs. Biomedicines. 2021;9(4):375. doi: 10.3390/biomedicines9040375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ryan KG, Swinny EE, Markham KR, Winefield C. Flavonoid gene expression and UV photoprotection in transgenic and mutant Petunia leaves. Phytochemistry. 2002;59(1):23–32. doi: 10.1016/S0031-9422(01)00404-6. [DOI] [PubMed] [Google Scholar]
- 128.Saakre M, Mathew D, Ravisankar V. Perspectives on plant flavonoid quercetin-based drugs for novel SARS-CoV-2. Beni-Suef Univer J Basic Appl Sci. 2021;10(1):1–13. doi: 10.1186/s43088-021-00107-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Salehi B, Cruz-Martins N, Butnariu M, Sarac I, Bagiu IC, Ezzat SM, Wang J, Koay A, Sheridan H, Adetunji COSemwal P, Schoebitz M, Martorell M, Sharifi-Rad J (2021) Hesperetin’s health potential: moving from preclinical to clinical evidence and bioavailability issues, to upcoming strategies to overcome current limitations. 62(16), 4449–4464. 10.1080/10408398.2021.1875979 [DOI] [PubMed]
- 130.Salehi B, Fokou PVT, Sharifi-Rad M, Zucca P, Pezzani R, Martins N, Sharifi-Rad J. The therapeutic potential of naringenin: a review of clinical trials. Pharmaceuticals. 2019 doi: 10.3390/PH12010011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Satari A, Ghasemi S, Habtemariam S, Asgharian S, Lorigooini Z. Rutin: a flavonoid as an effective sensitizer for anticancer therapy; insights into multifaceted mechanisms and applicability for combination therapy. Evid Based Complement Alternat Med. 2021 doi: 10.1155/2021/9913179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Schächinger V, Britten MB, Zeiher AM. Prognostic impact of coronary vasodilator dysfunction on adverse long-term outcome of coronary heart disease. Circulation. 2000;101(16):1899–1906. doi: 10.1161/01.CIR.101.16.1899. [DOI] [PubMed] [Google Scholar]
- 133.Seong EH, Gong DS, Shiwakoti S, Adhikari D, Kim HJ, Oak MH. Taxifolin as a major bioactive compound in the vasorelaxant effect of different pigmented rice bran extracts. Front Pharmacol. 2022;13:949. doi: 10.3389/FPHAR.2022.799064/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Shabir I, Kumar Pandey V, Shams R, Dar AH, Dash KK, Khan SA, Bashir I, Jeevarathinam G, Rusu AV, Esatbeyoglu T, Pandiselvam R. Promising bioactive properties of quercetin for potential food applications and health benefits: a review. Front Nutr. 2022;9:2587. doi: 10.3389/FNUT.2022.999752/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Shafer RW, Vuitton DA. Highly active antiretroviral therapy (HAART) for the treatment of infection with human immunodeficiency virus type 1. Biomed Pharmacother. 1999;53(2):73–86. doi: 10.1016/S0753-3322(99)80063-8. [DOI] [PubMed] [Google Scholar]
- 136.Shah PM. The need for new therapeutic agents: What is in the pipeline? Clin Microbiol Infect Suppl. 2005;11(3):36–42. doi: 10.1111/j.1469-0691.2005.01141.x. [DOI] [PubMed] [Google Scholar]
- 137.Sharma A, Sharma P, Singh Tuli H, Sharma AK. Phytochemical and pharmacological properties of flavonols. ELS. 2018 doi: 10.1002/9780470015902.a0027666. [DOI] [Google Scholar]
- 138.Shebeko SK, Zupanets IA, Popov OS, Tarasenko OO, Shalamay AS (2018) Effects of quercetin and its combinations on health. In: Polyphenols: mechanisms of action in human health and disease (pp. 373–394). Elsevier
- 139.Shih YL, Hung FM, Lee CH, Yeh MY, Lee MH, Lu HF, Chen YL, Liu JY, Chung JG (2017). Fisetin induces apoptosis of HSC3 human oral cancer cells through endoplasmic reticulum stress and dysfunction of mitochondria-mediated signaling pathways. In Vivo 31(6): 1103–1114. 10.21873/invivo.11176 [DOI] [PMC free article] [PubMed]
- 140.Sillapachaiyaporn C, Rangsinth P, Nilkhet S, Ung AT, Chuchawankul S, Tencomnao T. Neuroprotective effects against glutamate-induced HT-22 hippocampal cell damage and caenorhabditis elegans lifespan/healthspan enhancing activity of auricularia polytricha mushroom extracts. Pharmaceuticals. 2021 doi: 10.3390/PH14101001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Singh R, Singh S, Parihar P, Mishra RK, Tripathi DK, Singh VP, Chauhan DK, Prasad SM. Reactive oxygen species (ROS): beneficial companions of plants’ developmental processes. Front Plant Sci. 2016;7(September2016):1–19. doi: 10.3389/fpls.2016.01299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Solnier J, Fladerer JP (2020) Flavonoids: A complementary approach to conventional therapy of COVID-19? Phytochemistry Reviews, 1–23 [DOI] [PMC free article] [PubMed]
- 143.Song X, Tan L, Wang M, Ren C, Guo C, Yang B, Ren Y, Cao Z, Li Y, Pei J. Myricetin: a review of the most recent research. Biomed Pharmacother. 2021 doi: 10.1016/J.BIOPHA.2020.111017. [DOI] [PubMed] [Google Scholar]
- 144.Su CH, Kuo CL, Lu KW, Yu FS, Ma YS, Yang JL, Chu YL, Chueh FS, Liu KC, Chung JG. Fisetin-induced apoptosis of human oral cancer SCC-4 cells through reactive oxygen species production, endoplasmic reticulum stress, caspase-, and mitochondria-dependent signaling pathways. Environ Toxicol. 2017;32(6):1725–1741. doi: 10.1002/tox.22396. [DOI] [PubMed] [Google Scholar]
- 145.Survay NS, Upadhyaya CP, Kumar B, Young KE, Yoon D-Y, Park S-W. New genera of flavonols and flavonol derivatives as therapeutic molecules. J Korean Soc Appl Biol Chem. 2011;54(1):1–18. doi: 10.3839/jksabc.2011.001. [DOI] [Google Scholar]
- 146.Taheri Y, Suleria HAR, Martins N, Sytar O, Beyatli A, Yeskaliyeva B, Seitimova G, Salehi B, Semwal P, Painuli S, Kumar A, Azzini E, Martorell M, Setzer WN, Maroyi A, Sharifi-Rad J. Myricetin bioactive effects: Moving from preclinical evidence to potential clinical applications. BMC Complement Med Ther. 2020 doi: 10.1186/s12906-020-03033-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Tattini M, Galardi C, Pinelli P, Massai R, Remorini D, Agati G. Differential accumulation of flavonoids and hydroxycinnamates in leaves of Ligustrum vulgare under excess light and drought stress. New Phytol. 2004;163(3):547–561. doi: 10.1111/j.1469-8137.2004.01126.x. [DOI] [PubMed] [Google Scholar]
- 148.Taubert D, Berkels R, Klaus W, Roesen R. Nitric oxide formation and corresponding relaxation of porcine coronary arteries induced by plant phenols: essential structural features. J Cardiovasc Pharmacol. 2002;40(5):701–713. doi: 10.1097/00005344-200211000-00008. [DOI] [PubMed] [Google Scholar]
- 149.Taylor LP, Grotewold E. Flavonoids as developmental regulators. Curr Opin Plant Biol. 2005;8(3):317–323. doi: 10.1016/j.pbi.2005.03.005. [DOI] [PubMed] [Google Scholar]
- 150.Tewtrakul S, Nakamura N, Hattori M, Fujiwara T, Supavita T. Flavanone and flavonol glycosides from the leaves of Thevetia peruviana and their HIV-1 reverse transcriptase and HIV-1 integrase inhibitory activities. Chem Pharm Bull. 2002;50(5):630–635. doi: 10.1248/cpb.50.630. [DOI] [PubMed] [Google Scholar]
- 151.Thuan NH, Shrestha A, Trung NT, Tatipamula VB, Van Cuong D, Canh NX, Van Giang N, Kim TS, Sohng JK, Dhakal D. Advances in biochemistry and the biotechnological production of taxifolin and its derivatives. Biotechnol Appl Biochem. 2022;69(2):848–861. doi: 10.1002/BAB.2156. [DOI] [PubMed] [Google Scholar]
- 152.Topal F, Nar M, Gocer H, Kalin P, Kocyigit UM, Gülçin I, Alwasel SH. Antioxidant activity of taxifolin: an activity–structure relationship. J Enzyme Inhibit Med Chem. 2015;31(4):674–683. doi: 10.3109/14756366.2015.1057723. [DOI] [PubMed] [Google Scholar]
- 153.Torres-Piedra M, Ortiz-Andrade R, Villalobos-Molina R, Singh N, Medina-Franco JL, Webster SP, Binnie M, Navarrete-Vázquez G, Estrada-Soto S. A comparative study of flavonoid analogues on streptozotocin–nicotinamide induced diabetic rats: Quercetin as a potential antidiabetic agent acting via 11β-hydroxysteroid dehydrogenase type 1 inhibition. Eur J Med Chem. 2010;45(6):2606–2612. doi: 10.1016/j.ejmech.2010.02.049. [DOI] [PubMed] [Google Scholar]
- 154.Vasavi HS, Arun AB, Rekha PD. Anti-quorum sensing activity of Psidium guajava L. flavonoids against Chromobacterium violaceum and Pseudomonas aeruginosa PAO1. Microbiol Immunol. 2014;58(5):286–293. doi: 10.1111/1348-0421.12150. [DOI] [PubMed] [Google Scholar]
- 155.Vezza T, Rodríguez-Nogales A, Algieri F, Utrilla MP, Rodriguez-Cabezas ME, Galvez J. Flavonoids in inflammatory bowel disease: a review. Nutrients. 2016 doi: 10.3390/NU8040211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Vipin C, Mujeeburahiman M, Ashwini P, Arun AB, Rekha PD. Anti-biofilm and cytoprotective activities of quercetin against pseudomonas aeruginosa isolates. Lett Appl Microbiol. 2019;68(5):464–471. doi: 10.1111/lam.13129. [DOI] [PubMed] [Google Scholar]
- 157.Wang S, Yao J, Zhou B, Yang J, Chaudry MT, Wang M, Xiao F, Li Y, Yin W. Bacteriostatic effect of quercetin as an antibiotic alternative in vivo and its antibacterial mechanism in vitro. J Food Prot. 2018;81(1):68–78. doi: 10.4315/0362-028X.JFP-17-214. [DOI] [PubMed] [Google Scholar]
- 158.Wang Y, Liu XJ, Chen JB, Cao JP, Li X, Sun CD. Citrus flavonoids and their antioxidant evaluation. Crit Rev Food Sci Nutr. 2022;62(14):3833–3854. doi: 10.1080/10408398.2020.1870035. [DOI] [PubMed] [Google Scholar]
- 159.Watkins JM, Hechler PJ, Muday GK. Ethylene-induced flavonol accumulation in guard cells suppresses reactive oxygen species and moderates stomatal aperture. Plant Physiol. 2014;164(4):1707–1717. doi: 10.1104/pp.113.233528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Widlansky ME, Gokce N, Keaney JF, Vita JA. The clinical implications of endothelial dysfunction. J Am Coll Cardiol. 2003;42(7):1149–1160. doi: 10.1016/S0735-1097(03)00994-X. [DOI] [PubMed] [Google Scholar]
- 161.Wiegand I, Hilpert K, Hancock REW. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc. 2008;3(2):163–175. doi: 10.1038/nprot.2007.521. [DOI] [PubMed] [Google Scholar]
- 162.Williams RJ, Spencer JPE, Rice-Evans C. Flavonoids: antioxidants or signalling molecules? Free Radical Biol Med. 2004;36(7):838–849. doi: 10.1016/j.freeradbiomed.2004.01.001. [DOI] [PubMed] [Google Scholar]
- 163.Williamson G. Possible effects of dietary polyphenols on sugar absorption and digestion. Mol Nutr Food Res. 2013;57(1):48–57. doi: 10.1002/mnfr.201200511. [DOI] [PubMed] [Google Scholar]
- 164.Winkel-Shirley B. Biosynthesis of flavonoids and effects of stress. Curr Opin Plant Biol. 2002;5(3):218–223. doi: 10.1016/S1369-5266(02)00256-X. [DOI] [PubMed] [Google Scholar]
- 165.Won DH, Chung SH, Shin JA, Hong KO, Yang IH, Yun JW, Cho SD. Induction of sestrin 2 is associated with fisetinmediated apoptosis in human head and neck cancer cell lines. J Clin Biochem Nutrit. 2019;64(2):97–105. doi: 10.3164/jcbn.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Wong SK, Chin KY, Ima-Nirwana S. Quercetin as an agent for protecting the bone: a review of the current evidence. Int J Mol Sci. 2020;21(17):1–37. doi: 10.3390/IJMS21176448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Woodman OL, Chan ECH. Vascular and anti-oxidant actions of flavonols and flavones. Clin Exp Pharmacol Physiol. 2004;31(11):786–790. doi: 10.1111/j.1440-1681.2004.04072.x. [DOI] [PubMed] [Google Scholar]
- 168.Wu M, Peng A, Sun M, Deng Q, Hazlett LD, Yuan J, Liu X, Gao Q, Feng L, He J, Zhang P, Huang X. TREM-1 amplifies corneal inflammation after Pseudomonas aeruginosa infection by modulating toll-like receptor signaling and Th1/Th2-type immune responses. Infect Immun. 2011;79(7):2709–2716. doi: 10.1128/IAI.00144-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Xie Y, Yang W, Tang F, Chen X, Ren L. Antibacterial activities of flavonoids: structure-activity relationship and mechanism. Curr Med Chem. 2014;22(1):132–149. doi: 10.2174/0929867321666140916113443. [DOI] [PubMed] [Google Scholar]
- 170.Xing N, Chen Y, Mitchell SH, Young CYF. Quercetin inhibits the expression and function of the androgen receptor in LNCaP prostate cancer cells inhibited by quercetin in a dose-dependent manner. Carcinogenesis. 2001;22(3):409–414. doi: 10.1093/carcin/22.3.409. [DOI] [PubMed] [Google Scholar]
- 171.Yang EJ, Kim GS, Jun M, Song KS. Kaempferol attenuates the glutamate-induced oxidative stress in mouse-derived hippocampal neuronal HT22 cells. Food Funct. 2014;5(7):1395–1402. doi: 10.1039/C4FO00068D. [DOI] [PubMed] [Google Scholar]
- 172.Yao S, Wang X, Li C, Zhao T, Jin H, Fang W. Kaempferol inhibits cell proliferation and glycolysis in esophagus squamous cell carcinoma via targeting EGFR signaling pathway. Tumor Biology. 2016;37(8):10247–10256. doi: 10.1007/s13277-016-4912-6. [DOI] [PubMed] [Google Scholar]
- 173.Youl E, Bardy G, Magous R, Cros G, Sejalon F, Virsolvy A, Richard S, Quignard JF, Gross R, Petit P. Quercetin potentiates insulin secretion and protects INS-1 pancreatic β-cells against oxidative damage via the ERK1/2 pathway. Br J Pharmacol. 2010;161(4):799–814. doi: 10.1111/j.1476-5381.2010.00910.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Yu M, Gouvinhas I, Rocha J, Barros AIRNA. Phytochemical and antioxidant analysis of medicinal and food plants towards bioactive food and pharmaceutical resources. Sci Rep. 2021;11(1):1–14. doi: 10.1038/s41598-021-89437-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Yuan H, Gong A, Young CYF. Involvement of transcription factor Sp1 in quercetin-mediated inhibitory effect on the androgen receptor in human prostate cancer cells. Carcinogenesis. 2005;26(4):793–801. doi: 10.1093/carcin/bgi021. [DOI] [PubMed] [Google Scholar]
- 176.Yue S, Xue N, Li H, Huang B, Chen Z, Wang X. Hepatoprotective effect of apigenin against liver injury via the non-canonical NF-κB pathway in vivo and in vitro. Inflammation. 2020;43(5):1634–1648. doi: 10.1007/S10753-020-01238-5/FIGURES/6. [DOI] [PubMed] [Google Scholar]
- 177.Zamora-Ros R, Forouhi NG, Sharp SJ, González CA, Buijsse B, Guevara M, van der Schouw YT, Amiano P, Boeing H, Bredsdorff L, Fagherazzi G, Feskens EJ, Franks PW, Grioni S, Katzke V, Key TJ, Khaw KT, Kühn T, Masala G, Wareham NJ. Dietary intakes of individual flavanols and flavonols are inversely associated with incident type 2 diabetes in European populations. J Nutrit. 2014;144(3):335–343. doi: 10.3945/jn.113.184945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Zeng Y, Nikitkova A, Abdelsalam H, Li J, Xiao J. Activity of quercetin and kaemferol against Streptococcus mutans biofilm. Arch Oral Biol. 2019;98:9–16. doi: 10.1016/j.archoralbio.2018.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Zhang A, Chen B, Yuan Z, Li R, Liu C, Zhou H, Chen H, Jin M. HP0197 contributes to CPS synthesis and the virulence of streptococcus suis via CcpA. PLoS ONE. 2012 doi: 10.1371/journal.pone.0050987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Zhang XJ, Jia SS. Fisetin inhibits laryngeal carcinoma through regulation of AKT/NF-κB/mTOR and ERK1/2 signaling pathways. Biomed Pharmacother. 2016;83:1164–1174. doi: 10.1016/j.biopha.2016.08.035. [DOI] [PubMed] [Google Scholar]
- 181.Zhang Y, Zong B, Wang X, Zhu Y, Hu L, Li P, Zhang A, Chen H, Liu M, Tan C. Fisetin lowers streptococcus suis serotype 2 pathogenicity in mice by inhibiting the hemolytic activity of suilysin. Front Microbiol. 2018;9(July):1–13. doi: 10.3389/fmicb.2018.01723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhong G, Shen J, Chen Z, Lin Z, Long L, Wu J, Long C, Huang S, Lian P, Luo G. Antioxidant and antitumor activities of newly synthesized hesperetin derivatives. Molecules. 2022;27(3):879. doi: 10.3390/MOLECULES27030879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Zhong R, Farag MA, Chen M, He C, Xiao J. Recent advances in the biosynthesis, structure–activity relationships, formulations, pharmacology, and clinical trials of fisetin. EFood. 2022;3(1–2):e3. doi: 10.1002/EFD2.3. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data are available within this manuscript.