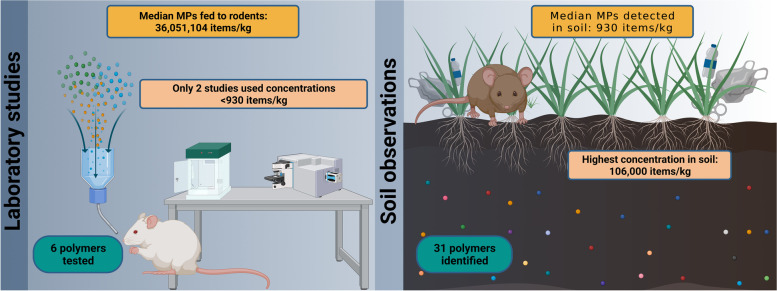
Abstract
Plastic pollution is now so widespread that microplastics are regularly detected in biological samples surveyed for their presence. Despite their pervasiveness, very little is known about the effects of microplastics on the health of terrestrial vertebrates. While emerging studies are showing that microplastics represent a potentially serious threat to animal health, data have been limited to in vivo studies on laboratory rodents that were force fed plastics. The extent to which these studies are representative of the conditions that animals and humans might actually experience in the real world is largely unknown. Here, we review 114 papers from the peer-reviewed literature in order to understand how the concentrations and types of microplastics being administered to rodents in lab studies compare to those found in terrestrial soils. From 73 in vivo lab studies, and 41 soil studies, we found that lab studies have heretofore fed rodents microplastics at concentrations that were hundreds of thousands of times greater than they would be exposed to in nature. Furthermore, health effects have been studied for only 20% of the microplastic polymers that are known to occur in soils. Plastic pollution is arguably one of the most pressing ecological and public health issues of our time, yet existing lab-based research on the health effects of terrestrial microplastics does not reflect the conditions that free-ranging vertebrates are actually experiencing. Going forward, performing more true-to-life research will be of the utmost importance to fully understand the impacts of microplastics and maintain the public’s faith in the scientific process.
Graphical Abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s43591-023-00059-1.
Keywords: Microplastics, Terrestrial, Health, Biomimetic, Mouse, Rat
Introduction
The invention of plastics in the early 1900s revolutionized human societies [1], yet the excessive consumption of short-lived and single-use plastics has resulted in plastics accumulating almost everywhere on Earth [2, 3]. Plastic pollution is now so widespread that microplastics – plastic particles between 1 µm and 5 mm – are regularly detected in biological samples surveyed for their presence [4, 5]. Their resistance to degradation and ubiquitous nature make microplastics a worrying environmental contaminant, yet, despite their pervasiveness, very little is known about how microplastics might be impacting the health of species living in terrestrial ecosystems. This stands in stark contrast to the fact that 80% of species live on land [6], and that the volume of microplastics in terrestrial systems may be greater than that in oceans [7, 8].
Though evidence is still extremely limited, emerging studies are attempting to identify the predicted no-effect concentrations (PNEC) of soil microplastics for different taxa [9, 10]. For terrestrial vertebrates, studies carried out on rodents are showing that microplastics represent a potentially serious health threat, and may impact an array of biological functions [11, 12]. For instance, recent work in mice and rats has demonstrated the detrimental effects of microplastics on sperm production [13]. Similarly, a study conducted by Wang et al. [14] indicated that mice exposed to microplastics experienced both necroptosis and inflammation within bladder epithelium, while Djouina et al. [15] found that microplastics can adversely affect the small intestine and colon of mice, causing histological and immune disturbances, as well as inflammation. Data have been primarily limited to in vivo studies on laboratory rodents that were force fed plastics, however, and there are currently no studies describing the health effects of microplastics exposure on terrestrial vertebrates outside of laboratory settings. Thus, although the findings from these studies are certainly worrying, the extent to which they are representative of the conditions that humans and animals are actually experiencing in the real world is largely unknown. Here, we review the peer-reviewed literature to explore the extent to which lab studies on the effects of microplastics are representative of the conditions that terrestrial animals are experiencing in the real world. In particular we focused on understanding how the concentrations of microplastics and types of polymers being administered to rodents in lab studies compared to those found in terrestrial soils. Although our focus was on microplastics in soils, this is not the only path of exposure to microplastics. For instance, plants can uptake microplastics [16], which can then be ingested by herbivorous/omnivorous species. Airborne microplastics can also be inhaled, with intake rates that may be comparable to ingestion [17]. Most studies on airborne microplastics quantify concentrations in terms of deposition rates [18], however, making direct comparisons to lab studies impossible, and there is little information on the microplastic exposure and ingestion rates of free-ranging terrestrial vertebrates. Nonetheless, air and waterborne microplastics will ultimately accumulate in soils [18, 19], and soils are at the base of many terrestrial food webs [7]. The concentrations of microplastics in soils are thus likely to be broadly representative of exposure levels. Our results can help provide much needed context to the findings of existing health studies, as well as an ecologically relevant baseline that can help guide future lab studies on the health effects of terrestrial microplastics.
Materials and methods
We first identified studies from the peer-reviewed literature that were focused on the health effects (i.e., changes in health resulting from exposure to a source) of microplastics on terrestrial rodents, or on microplastics in terrestrial soil environments. As our goal was to identify as many peer-reviewed articles as possible, we conducted a search for the terms “microplastics”, “microplastics” and “mice”, “microplastics” and “rats”, “microplastics” and “rodents”, “microplastics in lab”, and “microplastics in soil” on both Google Scholar and Scopus. We focused on microplastics in rodents as they are important model species for human and animal health research. Any in vivo lab studies not directly relating to the ingestion of microplastics were excluded as they were beyond the scope of our effort. Similarly, studies where soil samples were taken from lakes or river beds were excluded as our focus was on describing the conditions being experienced by terrestrial vertebrates. Through this initial search, 150 reviewed studies were compiled. For in vivo studies we extracted information on the polymer type, concentrations fed to laboratory rodents, and diameter, volume, and density of the microplastic particles. The microplastic type and final concentrations found in the soil environment were extracted from soil studies. There was very little consistency in the units across studies, and so to standardize microplastic measurements, all concentrations were converted to items/kg. To do this, polymer type was required to identify the density of the plastic, while diameter was required to calculate the volume. The known volume, density, and concentrations were then used in conjunction to calculate the number of particles and convert the data to items/kg. If any information required to make this conversion was absent from a study, it was excluded from subsequent analyses. Similarly, soil studies were excluded if information on the concentrations of microplastic were absent, or if they were experimental studies. This further narrowed the number of studies down to a total of 73 in vivo studies describing 183 experimental concentrations, and 41 soil studies with data on 93 sites. While it is possible that our literature search missed some relevant studies, the number of identified studies are in line with other recent reviews [20, 21] and our compiled dataset is thus likely to be representative of broad trends in the field. The full dataset is provided in Additional file 1 and the PRISMA checklist [22] in Additional file 2. The review process was not registered.
Results and discussion
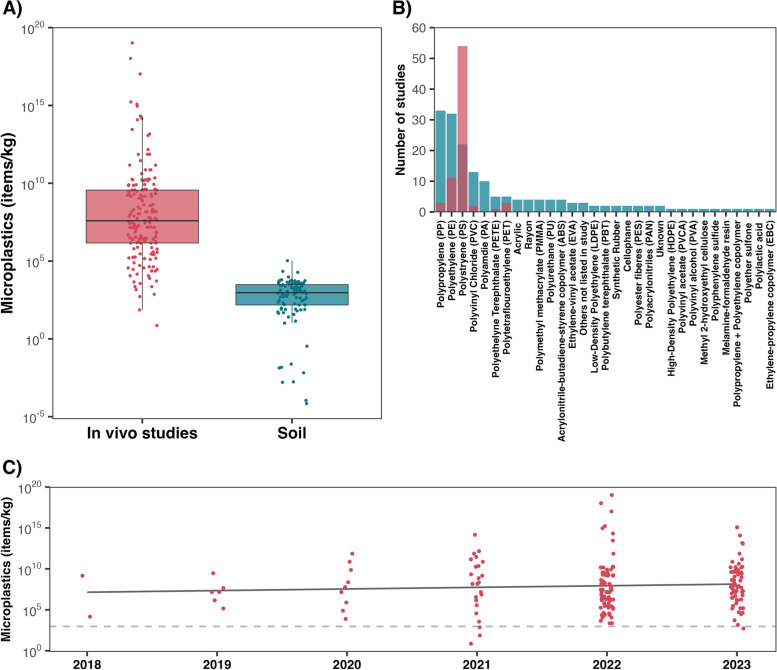
The median concentration of microplastics fed to laboratory rodents in in vivo studies was 39,051,103 items/kg. This was close to 42,000 times greater than the median concentration of 930 items/kg found in soil (Fig. 1A). Only two of the in vivo studies tested concentrations below this median value. The highest recorded mean concentration of microplastics in any soil sample was 106,000 items/kg which was found across various urban landscapes, including landfills, dumps, parking lots, industry and construction areas, urban parks, wetlands, forests, and agricultural areas in Coimbra, Portugal [23]; only 14 out of the 72 compiled lab studies used concentrations below this amount. We also found that while 31 different plastic polymers have been found to occur in soil, the health effects of only 6 polymers have been studied to date, with the overwhelming majority of in vivo experiments having focused on polystyrene (Fig. 1B). The stark contrast between the types and concentrations of microplastics being administered to lab rodents in in vivo studies versus the conditions these animals are likely to encounter in the wild illustrates the need for more ecologically realistic studies. Using a mixed effects regression model, we found that there was also no relationship between year of study and the concentrations of microplastics being administered to rodents in in vivo studies (βYear = 0.25, 95% CI = -0.34–0.84; Fig. 1C). In other words, there was no evidence that studies are becoming increasingly environmentally realistic over time.
Fig. 1.
The boxplot in A shows the concentrations of microplastics fed to rodents in in vivo lab studies, compared to those of MPs found in soils. In B the number of soil studies which identified different plastic polymers are shown in blue, whereas the number of polymers assessed via in vivo health studies are shown in red. In C, the concentrations of microplastics being administered to rodents in in vivo studies is shown as a function of year of publication. The dashed line in C represents the median concentration of microplastics found in soils, while the solid line represents the trend of a fitted regression model. Data were compiled from 114 peer-reviewed studies; 41 on microplastics in soil and 73 on the health effects of microplastics
Notably, and in light of this disconnect, a common trend across lab studies was the lack of any rationale for the concentrations of microplastic that were administered. The 31 studies that did provide justification chose concentrations that were based either on the concentrations of microplastic found in rivers [24–26], or on existing in vivo studies [12, 14, 27–51]. For instance, Yang et al. [34] and Mu et al. [30], both based their study designs on work on mice by Deng et al. [52]. Deng et al. [52], however, based their study on microplastics concentrations found in rivers, and therefore it does not accurately reflect exposure levels in terrestrial environments. In addition, while studies often use high concentrations of potential toxicants to establish dose-effect relationships, it is rare for acute or chronic ratios to exceed 100-fold greater than environmentally relevant concentrations [53]. In other words, the 4 order of magnitude difference between the microplastics administered in in vivo health studies and environmental concentrations can not be attributed to standard practice in dose-effect research. It is likely that the lack of any rationale contributed to the high variability in concentrations used between studies. Thus, while a handful of lab studies did provide some form of justification for their study design, the extent to which these studies are representative of the conditions that humans and animals are actually experiencing in the real world is limited.
Conclusions
Plastic pollution is arguably one of the most pressing ecological and public health issues of our time, yet existing research on the health effects of terrestrial microplastics does not accurately reflect the conditions that humans and animals are actually experiencing. This is in line with earlier findings from toxicology work on microplastics in aquatic environments [54–56], and our analyses showed no indication that exposure concentrations are becoming more realistic over time (Fig. 1C). Paired with this disconnect is the fact that 3,067 animals were sacrificed to generate the findings of these 73 studies, yet the majority of these animals were fed tens to hundreds of thousands of times more plastic than they would ever be exposed to in the wild. Because microplastics research also receives frequent media attention, performing true-to-life studies is of the utmost importance so as to not erode the public’s faith in the scientific process. It therefore falls on the scientific community to describe the ecologically realistic effects of microplastics on the health of terrestrial species in order for well-founded mitigation efforts to be launched.
Supplementary Information
Additional file 1. Compiled microplastics dataset.
Additional file 2. PRISMA 2020 Main Checklist.
Acknowledgements
Not applicable.
Authors’ contributions
MJN and MAMMF conceived of the study, CLM and JS conducted the literature review, CLM and MJN wrote the first manuscript draft, and all co-authors assisted with writing the final version of the manuscript. The author(s) read and approved the final manuscript.
Funding
This work was supported by an NSERC Discovery Grant RGPIN-2021-02758 to MJN, as well as the Canadian Foundation for Innovation. MAMMF was supported by LMUexcellent, funded by the Federal Ministry of Education and Research (BMBF) and the Free State of Bavaria under the Excellence Strategy of the Federal Government and the Länder.
Availability of data and materials
The data and R scripts used to carry out this study are openly available on GitHub at https://github.com/QuantitativeEcologyLab/MP_Disconnect.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Thompson RC, Swan SH, Moore CJ, Saal FS. Our plastic age. Philos Trans R Soc B Biol Sci. 2009;364(1526):1973–1976. doi: 10.1098/rstb.2009.0054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cole M, Lindeque P, Halsband C, Galloway TS. Microplastics as contaminants in the marine environment: a review. Mar Pollut Bull. 2011;62(12):2588–2597. doi: 10.1016/j.marpolbul.2011.09.025. [DOI] [PubMed] [Google Scholar]
- 3.Rochman CM, Hoellein T. The global odyssey of plastic pollution. Science (American Association for the Advancement of Science) 2020;368(6496):1184–5. doi: 10.1126/science.abc4428. [DOI] [PubMed] [Google Scholar]
- 4.Duis K, Coors A. Microplastics in the aquatic and terrestrial environment: sources (with a specific focus on personal care products), fate and effects. Environ Sci Eur. 2016;28(1):2–25. doi: 10.1186/s12302-015-0069-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bergami E, Rota E, Caruso T, Birarda G, Vaccari L, Corsi I. Plastics everywhere: First evidence of polystyrene fragments inside the common Antarctic collembolan Cryptopygus antarcticus. Biol Let. 2020;16(6):20200093. doi: 10.1098/rsbl.2020.0093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grosberg RK, Vermeij GJ, Wainwright PC. Biodiversity in water and on land. Curr Biol. 2012;22(1):900–903. doi: 10.1016/j.cub.2012.09.050. [DOI] [PubMed] [Google Scholar]
- 7.Souza Machado AA, Kloas W, Zarfl C, Hempel S, Rillig MC. Microplastics as an emerging threat to terrestrial ecosystems. Glob Change Biol. 2018;24(4):1405–1416. doi: 10.1111/gcb.14020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hurley RR, Nizzetto L. Fate and occurrence of micro(nano)plastics in soils: knowledge gaps and possible risks. Curr Opin Environ Sci Health. 2018;1:6–11.
- 9.Kim SW, Rillig MC. Research trends of microplastics in the soil environment: comprehensive screening of effects. Soil Ecol Lett. 2022;4(2):109–118. doi: 10.1007/s42832-021-0077-3. [DOI] [Google Scholar]
- 10.Tunali M, Adam V, Nowack B. Probabilistic environmental risk assessment of microplastics in soils. Geoderma. 2023;430:116315. doi: 10.1016/j.geoderma.2022.116315. [DOI] [Google Scholar]
- 11.Huang J, Dong G, Liang M, Wu X, Xian M, An Y, et al. Toxicity of microplastics with different size and surface charge on human nasal epithelial cells and rats via intranasal exposure. Chemosphere (Oxford). 2022;307. 10.1016/j.chemosphere.2022.136093. [DOI] [PubMed]
- 12.Luo T, Wang C, Pan Z, Jin C, Fu Z, Jin Y. Maternal polystyrene microplastic exposure during gestation and lactation altered metabolic homeostasis in the dams and their F1 and F2 offspring. Environ Sci Technol. 2019;53(18):10978–10992. doi: 10.1021/acs.est.9b03191. [DOI] [PubMed] [Google Scholar]
- 13.Jin H, Ma T, Sha X, Liu Z, Zhou Y, Meng X, et al. Polystyrene microplastics induced male reproductive toxicity in mice. J Hazard Mater. 2021;401:123430. doi: 10.1016/j.jhazmat.2020.123430. [DOI] [PubMed] [Google Scholar]
- 14.Wang Y, Wang S, Xu T, Cui W, Shi X, Xu S. A new discovery of polystyrene microplastics toxicity: the injury difference on bladder epithelium of mice is correlated with the size of exposed particles. Sci Total Environ. 2022;821:153413–153413. doi: 10.1016/j.scitotenv.2022.153413. [DOI] [PubMed] [Google Scholar]
- 15.Djouina M, Vignal C, Dehaut A, Caboche S, Hirt N, Waxin C, et al. Oral exposure to polyethylene microplastics alters gut morphology, immune response, and microbiota composition in mice. Environ Res. 2022;212(Pt B):113230. doi: 10.1016/j.envres.2022.113230. [DOI] [PubMed] [Google Scholar]
- 16.Azeem I, Adeel M, Ahmad MA, Shakoor N, Jiangcuo GD, Azeem K, et al. Uptake and accumulation of nano/microplastics in plants: a critical review. Nanomaterials. 2021;11(11):2935. doi: 10.3390/nano11112935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cox KD, Covernton GA, Davies HL, Dower JF, Juanes F, Dudas SE. Human consumption of microplastics. Environ Sci Technol. 2019;53(12):7068–7074. doi: 10.1021/acs.est.9b01517. [DOI] [PubMed] [Google Scholar]
- 18.Sridharan S, Kumar M, Singh L, Bolan NS, Saha M. Microplastics as an emerging source of particulate air pollution: a critical review. J Hazard Mater. 2021;418:126245. doi: 10.1016/j.jhazmat.2021.126245. [DOI] [PubMed] [Google Scholar]
- 19.Guo J, Huang X, Xiang L, Wang Y, Li Y, Li H, et al. Source, migration and toxicology of microplastics in soil. Environ Int. 2020;137:105263. doi: 10.1016/j.envint.2019.105263. [DOI] [PubMed] [Google Scholar]
- 20.Büks F, Kaupenjohann M. Global concentrations of microplastics in soils–a review. Soil. 2020;6(2):649–62. doi: 10.5194/soil-6-649-2020. [DOI] [Google Scholar]
- 21.Weber CJ, Bigalke M. Opening space for plastics—why spatial, soil and land use data are important to understand global soil (micro)plastic pollution. Microplastics. 2022;1:610–626. doi: 10.3390/microplastics1040042. [DOI] [Google Scholar]
- 22.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 2021;18(3):1003583–1003583. doi: 10.1371/journal.pmed.1003583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Leitão IA, Van Schaik L, Ferreira AJD, Alexandre N, Geissen V. The spatial distribution of microplastics in topsoils of an urban environment - Coimbra city case-study. Environ Res. 2023;218:114961. doi: 10.1016/j.envres.2022.114961. [DOI] [PubMed] [Google Scholar]
- 24.Liu S, Li H, Wang J, Wu B, Guo X. Polystyrene microplastics aggravate inflammatory damage in mice with intestinal immune imbalance. Sci Total Environ. 2022;833:155198–155198. doi: 10.1016/j.scitotenv.2022.155198. [DOI] [PubMed] [Google Scholar]
- 25.Shi C, Han X, Guo W, Wu Q, Yang X, Wang Y, et al. Disturbed Gut-Liver axis indicating oral exposure to polystyrene microplastic potentially increases the risk of insulin resistance. Environ Int. 2022;164:107273. doi: 10.1016/j.envint.2022.107273. [DOI] [PubMed] [Google Scholar]
- 26.Li S, Wang Q, Yu H, Yang L, Sun Y, Xu N, et al. Polystyrene microplastics induce blood–testis barrier disruption regulated by the MAPK-Nrf2 signaling pathway in rats. Environ Sci Pollut Res. 2021;28(35):47921–47931. doi: 10.1007/s11356-021-13911-9. [DOI] [PubMed] [Google Scholar]
- 27.Choi YJ, Park JW, Kim JE, Lee SJ, Gong JE, Jung Y, et al. Novel characterization of constipation phenotypes in ICR mice orally administrated with polystyrene microplastics. Int J Mol Sci. 2021;22(11):5845. doi: 10.3390/ijms22115845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hou J, Lei Z, Cui L, Hou Y, Yang L, An R, et al. Polystyrene microplastics lead to pyroptosis and apoptosis of ovarian granulosa cells via NLRP3/Caspase-1 signaling pathway in rats. Ecotoxicol Environ Saf. 2021;212:112012. doi: 10.1016/j.ecoenv.2021.112012. [DOI] [PubMed] [Google Scholar]
- 29.Li Z, Zhu S, Liu Q, Wei J, Jin Y, Wang X, et al. Polystyrene microplastics cause cardiac fibrosis by activating Wnt/β-catenin signaling pathway and promoting cardiomyocyte apoptosis in rats. Environ Pollut. 2020;265:115025. doi: 10.1016/j.envpol.2020.115025. [DOI] [PubMed] [Google Scholar]
- 30.Mu Y, Sun J, Li Z, Zhang W, Liu Z, Li C, et al. Activation of pyroptosis and ferroptosis is involved in the hepatotoxicity induced by polystyrene microplastics in mice. Chemosphere (Oxford) 2022;291(Pt 2):132944–132944. doi: 10.1016/j.chemosphere.2021.132944. [DOI] [PubMed] [Google Scholar]
- 31.Shi J, Deng H, Zhang M. Whole transcriptome sequencing analysis revealed key RNA profiles and toxicity in mice after chronic exposure to microplastics. Chemosphere (Oxford) 2022;304:135321–135321. doi: 10.1016/j.chemosphere.2022.135321. [DOI] [PubMed] [Google Scholar]
- 32.Wang Q, Wu Y, Zhang W, Shen T, Li H, Wu J, et al. Lipidomics and transcriptomics insight into impacts of microplastics exposure on hepatic lipid metabolism in mice. Chemosphere (Oxford) 2022;308:136591–136591. doi: 10.1016/j.chemosphere.2022.136591. [DOI] [PubMed] [Google Scholar]
- 33.Yang X, Jiang J, Wang Q, Duan J, Chen N, Wu D, et al. Gender difference in hepatic AMPK pathway activated lipid metabolism induced by aged polystyrene microplastics exposure. Ecotoxicol Environ Saf. 2022;245:114105. doi: 10.1016/j.ecoenv.2022.114105. [DOI] [PubMed] [Google Scholar]
- 34.Yang Y, Chen C, Lu T, Liao C. Toxicity-based toxicokinetic/toxicodynamic assessment for bioaccumulation of polystyrene microplastics in mice. J Hazard Mater. 2019;366:703–713. doi: 10.1016/j.jhazmat.2018.12.048. [DOI] [PubMed] [Google Scholar]
- 35.Wen S, Zhao Y, Liu S, Chen Y, Yuan H, Xu H. Polystyrene microplastics exacerbated liver injury from cyclophosphamide in mice: Insight into gut microbiota. Sci Total Environ. 2022;840:156668. doi: 10.1016/j.scitotenv.2022.156668. [DOI] [PubMed] [Google Scholar]
- 36.Sun H, Chen N, Yang X, Xia Y, Wu D. Effects induced by polyethylene microplastics oral exposure on colon mucin release, inflammation, gut microflora composition and metabolism in mice. Ecotoxicol Environ Saf. 2021;220:112340. doi: 10.1016/j.ecoenv.2021.112340. [DOI] [PubMed] [Google Scholar]
- 37.Lee CW, Hsu LF, Wu IL, Wang YL, Chen WC, Liu YJ, et al. Exposure to polystyrene microplastics impairs hippocampus-dependent learning and memory in mice. J Hazard Mater. 2022;430:128431. doi: 10.1016/j.jhazmat.2022.128431. [DOI] [PubMed] [Google Scholar]
- 38.Cao J, Xu R, Geng Y, Xu S, Guo M. Exposure to polystyrene microplastics triggers lung injury via targeting toll-like receptor 2 and activation of the NF-κB signal in mice. Environ Pollut. 2023;320:121068. doi: 10.1016/j.envpol.2023.121068. [DOI] [PubMed] [Google Scholar]
- 39.Zou H, Qu H, Bian Y, Sun J, Wang T, Ma Y, et al. Polystyrene microplastics induce oxidative stress in mouse hepatocytes in relation to their size. IJMS. 2023;24(8):7382. doi: 10.3390/ijms24087382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chen S, Yang JL, Zhang YS, Wang HY, Lin XY, Xue RY, et al. Microplastics affect arsenic bioavailability by altering gut microbiota and metabolites in a mouse model. Environ Pollut. 2023;324:121376. doi: 10.1016/j.envpol.2023.121376. [DOI] [PubMed] [Google Scholar]
- 41.Zhao T, Shen L, Ye X, Bai G, Liao C, Chen Z, et al. Prenatal and postnatal exposure to polystyrene microplastics induces testis developmental disorder and affects male fertility in mice. J Hazard Mater. 2023;445:130544. doi: 10.1016/j.jhazmat.2022.130544. [DOI] [PubMed] [Google Scholar]
- 42.Xie L, Chen T, Liu J, Hou Y, Tan Q, Zhang X, et al. Intestinal flora variation reflects the short-term damage of microplastic to the intestinal tract in mice. Ecotoxicol Environ Saf. 2022;246:114194. doi: 10.1016/j.ecoenv.2022.114194. [DOI] [PubMed] [Google Scholar]
- 43.Aghaei Z, Mercer GV, Schneider CM, Sled JG, Macgowan CK, Baschat AA, et al. Maternal exposure to polystyrene microplastics alters placental metabolism in mice. Metabolomics. 2022;19(1):1. doi: 10.1007/s11306-022-01967-8. [DOI] [PubMed] [Google Scholar]
- 44.Zheng J, Tan Z, Wu J, Liu J, Yang T, Yang H. Polystyrene microplastics aggravate acute pancreatitis in mice. Toxicology. 2023;491:153513. doi: 10.1016/j.tox.2023.153513. [DOI] [PubMed] [Google Scholar]
- 45.Feng Y, Yuan H, Wang W, Xu Y, Zhang J, Xu H, et al. Co-exposure to polystyrene microplastics and lead aggravated ovarian toxicity in female mice via the PERK/eIF2α signaling pathway. Ecotoxicol Environ Saf. 2022;243:113966. doi: 10.1016/j.ecoenv.2022.113966. [DOI] [PubMed] [Google Scholar]
- 46.Wang S, Chen L, Shi X, Wang Y, Xu S. Polystyrene microplastics-induced macrophage extracellular traps contributes to liver fibrotic injury by activating ROS/TGF-β/Smad2/3 signaling axis. Environ Pollut. 2023;324:121388. doi: 10.1016/j.envpol.2023.121388. [DOI] [PubMed] [Google Scholar]
- 47.Ijaz MU, Najam S, Hamza A, Azmat R, Ashraf A, Unuofin JO, et al. Pinostrobin alleviates testicular and spermatological damage induced by polystyrene microplastics in adult albino rats. Biomed Pharmacother. 2023;162:114686. doi: 10.1016/j.biopha.2023.114686. [DOI] [PubMed] [Google Scholar]
- 48.Wang W, Guan J, Feng Y, Nie L, Xu Y, Xu H, et al. Polystyrene microplastics induced nephrotoxicity associated with oxidative stress, inflammation, and endoplasmic reticulum stress in juvenile rats. Front Nutr. 2023;9:1059660. doi: 10.3389/fnut.2022.1059660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hassine MBH, Venditti M, Rhouma MB, Minucci S, Messaoudi I. Combined effect of polystyrene microplastics and cadmium on rat blood-testis barrier integrity and sperm quality. Environ Sci Pollut Res. 2023;30(19):56700–56712. doi: 10.1007/s11356-023-26429-z. [DOI] [PubMed] [Google Scholar]
- 50.Wang W, Guan J, Feng Y, Liu S, Zhao Y, Xu Y, et al. Polystyrene microplastics induced ovarian toxicity in juvenile rats associated with oxidative stress and activation of the PERK-eIF2α-ATF4-CHOP signaling pathway. Toxics. 2023;11(3):225. doi: 10.3390/toxics11030225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Deng Y, Chen H, Huang Y, Zhang Y, Ren H, Fang M, et al. Long-term exposure to environmentally relevant doses of large polystyrene microplastics disturbs lipid homeostasis via bowel function interference. Environ Sci Technol. 2022;56(22):15805–15817. doi: 10.1021/acs.est.1c07933. [DOI] [PubMed] [Google Scholar]
- 52.Deng Y, Zhang Y, Lemos B, Ren H. Tissue accumulation of microplastics in mice and biomarker responses suggest widespread health risks of exposure. Sci Rep. 2017;7(1):46687. doi: 10.1038/srep46687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Raimondo S, Montague BJ, Barron MG. Determinants of variability in acute to chronic toxicity ratios for aquatic invertebrates and fish. Environ Toxicol Chem. 2007;26(9):2019. doi: 10.1897/07-069R.1. [DOI] [PubMed] [Google Scholar]
- 54.Lenz R, Enders K, Nielsen TG. Microplastic exposure studies should be environmentally realistic. Proc Natl Acad Sci USA. 2016;113(29). Available from: https://pnas.org/doi/full/10.1073/pnas.1606615113. Cited 2023 May 5. [DOI] [PMC free article] [PubMed]
- 55.Connors KA, Dyer SD, Belanger SE. Advancing the quality of environmental microplastic research: advancing the quality of environmental microplastic research. Environ Toxicol Chem. 2017;36(7):1697–1703. doi: 10.1002/etc.3829. [DOI] [PubMed] [Google Scholar]
- 56.De Ruijter VN, Redondo-Hasselerharm PE, Gouin T, Koelmans AA. Quality criteria for microplastic effect studies in the context of risk assessment: a critical review. Environ Sci Technol. 2020;54(19):11692–11705. doi: 10.1021/acs.est.0c03057. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1. Compiled microplastics dataset.
Additional file 2. PRISMA 2020 Main Checklist.
Data Availability Statement
The data and R scripts used to carry out this study are openly available on GitHub at https://github.com/QuantitativeEcologyLab/MP_Disconnect.