Abstract
Background
Long‐acting beta‐agonists (LABAs), long‐acting muscarinic antagonists (LAMAs), and inhaled corticosteroids (ICSs) are inhaled medications used to manage chronic obstructive pulmonary disease (COPD). When two classes of medications are required, a LAMA plus an ICS (LABA+ICS) were previously recommended within a single inhaler as the first‐line treatment for managing stable COPD in people in high‐risk categories. However, updated international guidance recommends a LAMA plus a LABA (LAMA+LABA). This systematic review is an update of a Cochrane Review first published in 2017.
Objectives
To compare the benefits and harms of LAMA+LABA versus LABA+ICS for treatment of people with stable COPD.
Search methods
We performed an electronic search of the Cochrane Airways Group Specialised Register, ClinicalTrials.gov, and the World Health Organization Clinical Trials Search Portal, followed by handsearches. Two review authors screened the selected articles. The most recent search was run on 10 September 2022.
Selection criteria
We included parallel or cross‐over randomised controlled trials of at least one month's duration, comparing LAMA+LABA and LABA+ICS for stable COPD. We included studies conducted in an outpatient setting and irrespective of blinding.
Data collection and analysis
Two review authors independently extracted data and evaluated risk of bias. We resolved any discrepancies through discussion. We analysed dichotomous data as odds ratios (ORs), and continuous data as mean differences (MDs), with 95% confidence intervals (CIs) using Review Manager 5. Primary outcomes were: participants with one or more exacerbations of COPD; serious adverse events; quality of life, as measured by the St. George's Respiratory Questionnaire (SGRQ) total score change from baseline; and trough forced expiratory volume in one second (FEV1). We used the GRADE framework to rate our certainty of the evidence in each meta‐analysis as high, moderate, low or very low.
Main results
This review updates the first version of the review, published in 2017, and increases the number of included studies from 11 to 19 (22,354 participants). The median number of participants per study was 700. In each study, between 54% and 91% (median 70%) of participants were males. Study participants had an average age of 64 years and percentage predicted FEV1 of 51.5% (medians of study means). Included studies had a generally low risk of selection, performance, detection, attrition, and reporting biases. All but two studies were sponsored by pharmaceutical companies, which had varying levels of involvement in study design, conduct, and data analysis.
Primary outcomes
The odds of having an exacerbation were similar for LAMA+LABA compared with LABA+ICS (OR 0.91, 95% CI 0.78 to 1.06; I2 = 61%; 13 studies, 20,960 participants; moderate‐certainty evidence). The odds of having a serious adverse event were also similar (OR 1.02, 95% CI 0.91 to 1.15; I2 = 20%; 18 studies, 23,183 participants; high‐certainty evidence). Participants receiving LAMA+LABA had a similar improvement in quality of life, as measured by the SGRQ, to those receiving LABA+ICS (MD ‐0.57, 95% CI ‐1.36 to 0.21; I2 = 78%; 9 studies, 14,437 participants; moderate‐certainty evidence) but showed a greater improvement in trough FEV1 (MD 0.07, 95% CI 0.05 to 0.08; I2 = 73%; 12 studies, 14,681 participants; moderate‐certainty evidence).
Secondary outcomes
LAMA+LABA decreased the odds of pneumonia compared with LABA+ICS from 5% to 3% (OR 0.61, 95% CI 0.52 to 0.72; I2 = 0%; 14 studies, 21,829 participants; high‐certainty evidence) but increased the odds of all‐cause death from 1% to 1.4% (OR 1.35, 95% CI 1.05 to 1.75; I2 = 0%; 15 studies, 21,510 participants; moderate‐certainty evidence). The odds of achieving a minimal clinically important difference of four or more points on the SGRQ were similar between LAMA+LABA and LABA+ICS (OR 1.06, 95% CI 0.90 to 1.25; I2 = 77%; 4 studies, 13,614 participants; moderate‐certainty evidence).
Authors' conclusions
Combination LAMA+LABA therapy probably holds similar benefits to LABA+ICS for exacerbations and quality of life, as measured by the St George's Respiratory Questionnaire, for people with moderate to severe COPD, but offers a larger improvement in FEV1 and a slightly lower risk of pneumonia. There is little to no difference between LAMA+LABA and LAMA+ICS in the odds of having a serious adverse event. Whilst all‐cause death may be lower with LABA+ICS, there was a very small number of events in the analysis, translating to a low absolute risk. Findings are based on moderate‐ to high‐certainty evidence from heterogeneous trials with an observation period of less than one year. This review should be updated again in a few years.
Keywords: Female; Humans; Male; Middle Aged; Adrenal Cortex Hormones; Adrenal Cortex Hormones/therapeutic use; Adrenergic beta-2 Receptor Agonists; Adrenergic beta-2 Receptor Agonists/therapeutic use; Muscarinic Antagonists; Pneumonia; Pneumonia/drug therapy; Pulmonary Disease, Chronic Obstructive; Pulmonary Disease, Chronic Obstructive/drug therapy; Quality of Life
Plain language summary
Which combination of inhaled medications are safe and effective for chronic obstructive pulmonary disease (COPD)?
Key messages
• Using an inhaler containing a long‐acting muscarinic antagonist plus a long‐acting beta‐agonist (LAMA+LABA) for chronic obstructive pulmonary disease (COPD) probably improves respiratory function and reduces the risk of pneumonia compared to LABA plus an inhaled corticosteroid (LABA+ICS).
• LAMA+LABA and LABA+ICS probably work as well as each other in reducing COPD exacerbations and improving quality of life.
• The risk of death was slightly higher in people taking LAMA+LABA.
What is chronic obstructive pulmonary disease, and how is it treated?
Chronic obstructive pulmonary disease (COPD) is a long‐term lung condition characterised by cough, sputum production (fluids from the lungs, i.e. phlegm), and difficulty breathing.
COPD is treated with medications called 'bronchodilators' that make breathing easier by relaxing the muscles in the lungs and widening the airways. Two key types of bronchodilator medicines are long‐acting muscarinic antagonists (LAMAs) and long‐acting beta‐agonists (LABAs). Healthcare guidelines now recommend that people with stable, high‐risk COPD use inhalers containing either a combined LAMA+LABA medicine or a combined LABA plus inhaled corticosteroid (LABA+ICS) medicine. Corticosteroids are anti‐inflammatory medicines.
What did we want to find out?
We examined the findings of trials that randomised people to receive either LAMA+LABA or LABA+ICS to see how they compare.
What did we do?
We searched for trials that explored the benefits and harms of LAMA+LABA and LABA+ICS for the treatment of people with COPD. We summarised these results and rated our confidence in the evidence.
What did we find?
We included 19 studies involving 22,354 participants. The studies lasted from 6 to 52 weeks. The studies included more men than women (approximately 70% of participants were men), and the people in the studies were about 64 years old. Most studies included people with moderate to severe COPD. Pharmaceutical companies were involved in most of the studies, which might affect how much we can trust the results.
Compared to LABA+ICS, LAMA+LABA led to an improvement in lung function, reduced pneumonia from 5% to 3% but increased the risk of death from 1% to 1.4%. LAMA+LABA probably made little to no difference to COPD exacerbations (flare‐ups) compared to LABA+ICS. People in each of the treatment groups reported similar quality of life scores and were about as likely to experience serious side effects, which were rare.
What are the limitations of the evidence?
Our confidence in the evidence was moderate to high because the included studies were well‐designed and had a sufficient number of participants with mainly moderate to severe COPD.
How up to date is this evidence?
This review is up to date to 10 September 2022. The results from future or ongoing trials evaluating newly developed medicines are awaited. This review should be updated again in a few years.
Summary of findings
Summary of findings 1. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD).
| LAMA plus LABA versus LABA plus ICS for stable COPD | ||||||
| Population: people with stable COPD Setting: outpatient. Studies were conducted in > 50 countries including low‐, medium‐, and high‐income countries from all continents Intervention: LAMA+LABA Comparison: LABA+ICS | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effects (95% CI) | Number of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| LABA+ICS | LAMA+LABA | |||||
| Exacerbations (number of people experiencing ≥ 1 exacerbations) Follow‐up: 6 to 52 weeks |
291 per 1000 | 272 per 1000 (243 to 303) | OR 0.91 (0.78 to 1.06) | 20,960 (13 RCTs) | ⊕⊕⊕⊝ Moderatea‐e | Low OR means favourable outcome |
| Serious adverse events (number of people experiencing ≥ 1 SAEs) Follow‐up: 6 to 52 weeks |
148 per 1000 | 150 per 1000 (136 to 166) | OR 1.02 (0.91 to 1.15) | 23,183 (18 RCTs) | ⊕⊕⊕⊕ Highb,c,d,e | Low OR means favourable outcome. Herth 2020 was discarded as no events were reported in either arm. |
| Quality of life as measured by SGRQ total score change from baseline (MD) Follow‐up: 12 to 52 weeks Scale 0 to 100 |
‐ | ‐ | MD ‐0.57 (1.36 lower to 0.21 higher) |
14,437 (9 RCTs) |
⊕⊕⊕⊝ Moderatea‐e | Low MD means favourable outcome |
| Trough FEV1 change from baseline Follow‐up: 6 to 52 weeks |
‐ | ‐ | MD 0.07 L (0.05 to 0.08) |
14,681 (12 RCTs) |
⊕⊕⊕⊝ Moderatea,b,d,e | High MD means favourable outcome |
| Pneumonia Follow‐up: 12 to 52 weeks |
45 per 1000 | 28 per 1000 (24 to 33) | OR 0.61 (0.52 to 0.72) | 21,829 (14 RCTs) | ⊕⊕⊕⊕ Highb,d,e | Low OR means favourable outcome |
| All‐cause death Follow‐up: 6 to 52 weeks |
10 per 1000 | 14 per 1000 (11 to 18) | OR 1.35 (1.05 to 1.75) | 21,510 (15 RCTs) | ⊕⊕⊕⊝ Moderateb,d,e,f | Low OR means favourable outcome |
| SGRQ total score change from baseline (≥ 4 points, MCID) Follow‐up: 26 to 52 weeks |
373 per 1000 | 387 per 1000 (349 to 427) | OR 1.06 (0.90 to 1.25) |
13,614 (4 RCTs) |
⊕⊕⊕⊝ Moderatea‐e | High OR means favourable outcome |
| *The absolute risk (and its 95% CI) of LAMA+LABA group is based on the assumed risk in the LABA+ICS group and the OR of the intervention (and its 95% CI). CI: confidence interval; COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in one second; ICS: inhaled corticosteroid; LABA: long‐acting beta‐agonist; LAMA: long‐acting muscarinic antagonist; MCID: minimal clinically important difference; MD: mean difference; OR: odds ratio; RCT: randomised controlled trial; SGRQ: St. George's Respiratory Questionnaire. | ||||||
|
GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of effect but may be substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aThere was a considerable heterogeneity, I2 > 50%. bStudies varied in their inclusion criteria around recent history of exacerbation, baseline percentage predicted FEV1 and exclusion of asthma. No indirectness downgrades, but impacts interpretation and applicability of results (see Discussion). cConfidence interval includes benefit of either treatment but lies within predefined threshold for clinical importance (0.7 to 1.5 for OR outcomes; 4‐point MCID for SGRQ) ‐ no downgrade for imprecision dNo downgrades for publication bias. Funnel plots examined for primary outcomes (exacerbations, serious adverse events, SGRQ, FEV1) do not show obvious asymmetry (see Figure 3; Figure 3; Figure 4; Figure 6). Nearly all studies included in pneumonia and all‐cause death analyses. Only 4 studies included in SGRQ 4+ change analysis but not specified in most studies. eNo downgrades for risk of bias. Across outcomes, all or almost all studies were at high risk of 'other' bias due to conflicts of interest, and studies contributing between 18.5% and 35.9% of the analysis weight were rated high risk of bias in at least one other domain. We did not prespecify a threshold or specific domains for downgrading, but considered it insufficient to downgrade if studies contributing more than half the analysis weight were at low or unclear risk of bias in all but the 'other' domain. fThere was imprecision due to a low number of events.
Background
Description of the condition
Chronic obstructive pulmonary disease (COPD) is characterised by bronchial obstruction, systemic inflammation, and comorbidities. It is the third leading cause of death worldwide, with more than 3.23 million people dying due to COPD each year (WHO 2019). In addition to active tobacco smoking, indoor biomass smoke, outdoor air pollution, and occupational exposure also play a role in the development of COPD. The most common symptoms of COPD – shortness of breath on exertion and cough – are present for a prolonged period and typically worsen over time (GOLD 2023).
Since the late 1960s, the definition of COPD has repeatedly been modified. Early definitions of COPD included chronic bronchitis, which is clinically characterised by chronic cough, and emphysema, which is pathologically defined by damaged sacs or alveoli in the lungs (Burrows 1966). In 2001, the Global Initiative for Chronic Obstructive Lung Disease (GOLD) released its first report, Global Strategy for the Diagnosis, Management, and Prevention of COPD (Pauwels 2001), which supported the definition of COPD, indicating that the disorder is recognised primarily by chronic obstruction of lung airflow (Pauwels 2001).
If COPD is properly diagnosed and managed, symptoms can be ameliorated. Smoking cessation, smoke‐free cooking, vaccination, and an active lifestyle are the first steps in COPD management, and daily pharmacological treatment is required for most people with symptomatic stable COPD (GOLD 2023).
Description of the intervention
While asymptomatic individuals with mild airflow limitations can be treated with on‐demand short‐acting bronchodilators, key medications for symptomatic COPD management consist of three classes of inhaled medication: long‐acting beta‐agonists (LABAs), long‐acting muscarinic antagonists (LAMAs), and inhaled corticosteroids (ICSs) (GOLD 2023). If the disease cannot be adequately controlled with LAMA or LABA monotherapy, administration of two or more medications from different classes may prove beneficial. When two classes of medication are required, a LAMA plus a LABA (LAMA+LABA) or a LABA plus an ICS (LABA+ICS) are often selected because these combinations can be administered via a single medication device (Frampton 2014; Malerba 2014; Nannini 2013; Schachter 2013), which is most beneficial for improving patient adherence (Horita 2015a).
How the intervention might work
Currently, there are no medications that can cure COPD. Thus, the practical goal of COPD treatment is to control the symptoms, reduce the frequency of exacerbations, and improve exercise tolerance and quality of life. COPD treatment usually consists of smoking cessation, vaccination, inhaled bronchodilators, ICSs, oral medication, long‐term oxygen therapy, and pulmonary rehabilitation (GOLD 2023). According to the GOLD approach, people are classified into three categories depending on the degree of symptoms and the risk of exacerbations (GOLD 2023). Medications belonging to a specific class are recommended based on the following criteria:
Category A (low symptoms plus few exacerbations): bronchodilator (short‐ or long‐acting); consider switching to another depending on response;
Category B (high symptoms plus few exacerbations): LAMA+LABA;
Category E (high symptoms plus many exacerbations): LAMA+LABA; LAMA+LABA+ICS is considered if blood eosinophil count is high.
Long‐acting muscarinic antagonists
LAMAs dilate the airway by selectively blocking acetylcholine M3 receptors and inhibiting bronchoconstriction (Alagha 2014). Since the early 2000s, LAMAs, especially tiotropium, have been regarded as the first‐choice medication for treating COPD. LAMAs confer anti‐inflammatory effects and, more importantly, anti‐airway remodelling effects (Tashkin 2004).
Long‐acting beta‐agonists
LABAs widen the airway by relaxing the airway muscles. Studies have suggested that LABAs may also provide anti‐inflammatory and protective effects against bronchoconstrictive substances. Regular use of a short‐acting beta‐agonist that works quickly and lasts for four to six hours is not currently recommended for people with asthma or COPD. A LABA that lasts for approximately 12 to 24 hours is considered a maintenance medication (Anderson 2014; Tashkin 2004).
Inhaled corticosteroids
ICSs reduce airway inflammation. Although ICSs are indicated for bronchial asthma, in which eosinophils play a key role, they are not as effective when neutrophils are observed in the airways of people with COPD (Barnes 2010; Hanania 2008; Suissa 2009). The previous GOLD report recommended that ICSs be prescribed in combination with LABA for people with COPD with severe airflow limitation or with a high risk of exacerbations (GOLD 2016). Studies have suggested that LABA+ICS may be highly effective for people with a high sputum/blood eosinophil count (Pascoe 2015).
Why it is important to do this review
The previous GOLD report recommended the first‐line use of ICSs only for people with severe to very severe airflow limitation and two or more exacerbations per year, with one or more hospitalisations for exacerbations (GOLD 2016). That report suggested that ICSs reduce the risk of exacerbations (GOLD 2016). Nonetheless, prescription rates for ICSs and combined LABA+ICS agents are high (Drivenes 2014; White 2013). This is probably because many randomised controlled trials (RCTs) have supported the hypothesis that the salmeterol (LABA) plus fluticasone propionate (ICS) combination – the oldest combination treatment – can improve quality of life, especially for people with dyspnoea, and can also decrease acute exacerbations of COPD and reduce yearly declines in pulmonary function (GOLD 2016). Blood and sputum eosinophil counts can serve as predictive biomarkers for differentiating between people with COPD who will derive the greatest benefit from ICS administration and people who will not benefit from an ICS (Pascoe 2015). The GOLD 2023 report now recommends LAMA+LABA+ICS only for people with category E COPD who have a high eosinophil count, and recommends that ICSs should not be over‐prescribed in COPD cases.
Understanding which type of combination treatment (LAMA+LABA or LABA+ICS) is most beneficial for people with COPD, regardless of eosinophil count, is important, and this systematic review contributes to answering this question.
Objectives
To compare the benefits and harms of LAMA+LABA versus LABA+ICS for treatment of people with stable COPD.
Methods
Criteria for considering studies for this review
Types of studies
We planned to include individually‐randomised and cluster‐randomised controlled trials and cross‐over trials, but not quasi‐RCTs. We included studies reported as full text, those published as abstract only, and unpublished data. When we could not obtain sufficient data from published articles, we contacted authors and sponsors, and accessed trial registration websites. We included open‐label, single‐blinded, and double‐blinded studies. The minimum accepted trial duration was one month.
Types of participants
We included adults with a diagnosis of COPD according to GOLD guidelines (GOLD 2023). We did not set specific exclusion criteria involving comorbidities. We planned to exclude original studies focusing on asthma‐COPD overlap syndrome (ACOS).
Types of interventions
We included trials comparing LAMA+LABA versus LABA+ICS. We permitted treatments administered via a single combined device or via two separate devices. We excluded trials of short‐acting bronchodilators (e.g. ipratropium). We included co‐interventions when they were not part of the randomly assigned treatment.
Types of outcome measures
Primary outcomes
Exacerbations (participants with one or more)
Serious adverse events (participants with one or more)
Quality of life, as measured by the St. George's Respiratory Questionnaire (SGRQ) total score change from baseline
Trough forced expiratory volume in one second (FEV1) change from baseline
Secondary outcomes
Pneumonia (participants with one or more occurrences); assessed based on chest x‐ray
All‐cause death
SGRQ total score change from baseline (4 points or greater)
Hospitalisations for COPD exacerbations (participants with one or more occurrences)
Search methods for identification of studies
Electronic searches
We identified trials from the Cochrane Airways Group Specialised Register (CAGR), which is maintained by the Information Specialist for the Group. The Register contains trial reports identified through systematic searches of bibliographic databases, including the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, the Cumulative Index to Nursing and Allied Health Literature (CINAHL), the Allied and Complementary Medicine Database (AMED), and PsycINFO, and through handsearching of respiratory journals and conference abstracts (see Appendix 1 for details). We searched all records in the CAGR from the date it was last searched for the previous version of this review (2 February 2016) to 10 September 2022, using the search strategy provided in Appendix 2.
We conducted a search of ClinicalTrials.gov (www.ClinicalTrials.gov) and the World Health Organization (WHO) Clinical Trials Search Portal (www.who.int/ictrp/en/), from the date they were last searched (4 June 2016) to 10 September 2022 (Appendix 3; Appendix 4).
We searched all databases from their inception, and we imposed no restrictions on language of publication.
Searching other resources
We checked reference lists of all primary studies and review articles for additional references, and we searched relevant manufacturers' websites for trial information. We searched for errata or retractions from included studies published as full text on PubMed (www.ncbi.nlm.nih.gov/pubmed). Handsearches were done up to 1 November 2021.
Data collection and analysis
Selection of studies
We used Cochrane’s Screen4Me workflow to help assess the search results. Screen4Me comprises three components: (1) known assessments – a service that matches records in the search results to records that have already been screened in Cochrane Crowd and labelled as an RCT or 'Not an RCT'; (2) the RCT classifier – a machine learning model that distinguishes RCTs from non‐RCTs; and (3) if appropriate, Cochrane Crowd – Cochrane’s citizen science platform where the Crowd helps to identify and describe health evidence. For more information about Screen4Me and the evaluations that have been done, please visit the Screen4Me webpage on the Cochrane Information Specialists' portal: community.cochrane.org/organizational-info/resources/resources-groups/information-specialists-portal.
Two review authors (NF and NH) independently screened the titles and abstracts of all studies identified by the search for possible inclusion, and coded studies as 'retrieve' (eligible or potentially eligible/unclear) or 'do not retrieve'. We retrieved the full‐text publications. Two review authors (NF and NH) independently screened the full texts to identify studies for inclusion and recorded reasons for exclusion of ineligible studies. We resolved disagreements through discussion, or, when required, we consulted a third review author (TK). We identified and excluded duplicates and collated multiple reports of the same study, so that each study, rather than each report, was the unit of interest in the review. We recorded the selection process in sufficient detail to complete a PRISMA (Preferred Reporting Items for Systematic Reviews and Meta‐Analyses) flow diagram and a Characteristics of excluded studies table (Moher 2009).
Data extraction and management
We used a data collection form that had been piloted on at least one study in the review to document study characteristics and outcome data. Two review authors (NF and NH) extracted the following study characteristics from the included studies.
Methods: study design, duration of study follow‐up and 'run‐in' period, number of study centres and countries, and study start date.
Participants: number, mean and standard deviation (SD) age, gender, mean and SD of baseline FEV1 key inclusion criteria, number of participants randomised and completed, and follow‐up duration.
Interventions: intervention, comparison, and dosage of the intervention.
Outcomes: primary outcomes specified and collected and time points reported.
Notes: funding for trial and notable conflicts of interest (COI) of trial authors, trial registration, and other information if necessary.
Two review authors (NF and NH) independently extracted outcome data from the included studies. We noted in the Characteristics of included studies table if outcome data were not reported in a useable way. We resolved disagreements by consensus or by consultation with a third review author (TK). One review author (NF) transferred data into Review Manager 5 (Review Manager 2014). We double‐checked that data were entered correctly by comparing data presented in the systematic review versus data provided in study reports. A second review author (NH) spot‐checked study characteristics for accuracy against the trial report.
Assessment of risk of bias in included studies
Two review authors (NF and NH) independently assessed risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). We resolved disagreements by discussion or by consultation with another review author (EO). We assessed risk of bias according to the following domains:
random sequence generation;
allocation concealment;
blinding of participants and personnel;
blinding of outcome assessment;
incomplete outcome data;
selective outcome reporting;
other bias.
We graded each potential source of bias as high, low, or unclear and provided an explanation from the study report together with a justification for our judgement in the risk of bias table. We summarised risk of bias judgements across studies for each of the domains listed. We classified outcomes as subjective and objective to consider different risks of performance and selection bias for different blinding methods. All‐cause death is the only outcome of interest to this review that may be considered truly objective and the SGRQ is the only subjective, self‐reported measure. The other outcomes (exacerbations, serious adverse events, FEV1 and pneumonia) might be considered semi‐objective: they involve a degree of patient reporting, behaviour, or clinician judgement that could introduce bias (intentionally or otherwise) if the randomised treatment had been inadvertently detected through side effects or other means. For example, this could happen in the grading of an adverse event as serious or non‐serious, effort given when measuring FEV1, or the interpretation of a chest X‐ray to determine pneumonia. When we requested information on risk of bias related to unpublished data or corresponded with a trialist, we noted this in the risk of bias table.
When considering treatment effects, we took into account risk of bias for studies that contributed to that outcome.
Assessment of bias in conducting the systematic review
We conducted the review according to the previously published protocol (Horita 2016).
Measures of treatment effect
We analysed dichotomous data as odds ratios (ORs), and continuous data as MDs with 95% confidence intervals (CIs). We entered data presented as a scale with a consistent direction of effect (i.e. data in the LAMA+LABA arm minus data in the LABA+ICS arm). Although there is no universal rule to interpret the magnitude of the therapeutic effect from ORs, we believe that an OR greater than 1.5 and an OR of less than 0.7 mean that there is a considerable chance that the outcome is clinically important.
We undertook meta‐analyses only when this was meaningful (i.e. if treatments, participants, and the underlying clinical question were similar enough for pooling to make sense).
We had planned to describe skewed data using medians and interquartile ranges; however, we found no report describing skewed data.
According to the original protocol, when multiple trial arms were reported in a single trial, we included only the relevant arms.
Unit of analysis issues
We analysed the number of participants, not the number of events, as the unit of analysis for dichotomous data (i.e. participants with one or more events). For continuous data, we used MDs.
Dealing with missing data
We tried to contact investigators, study sponsors, and registration websites to verify key study characteristics and to obtain missing numerical outcome data when possible (e.g. when a study was only reported in an abstract format). When this was not possible, and when missing data were thought to introduce serious bias, we explored the impact of including such studies in the overall assessment of results by conducting a sensitivity analysis.
Assessment of heterogeneity
We used the I2 statistic to measure heterogeneity amongst the trials in each analysis: 0% to 40%: might not be important; 30% to 60%: might represent moderate heterogeneity; 50% to 90%: might represent substantial heterogeneity; 75% to 100%: might show considerable heterogeneity (Higgins 2011). When we identified considerable heterogeneity, we reported this and explored possible causes by performing a prespecified subgroup analysis.
Assessment of reporting biases
We created and examined a funnel plot to explore possible small‐study and publication biases for the primary outcomes.
Data synthesis
We used a random‐effects model and performed a sensitivity analysis by using a fixed‐effect model (see Sensitivity analysis).
Subgroup analysis and investigation of heterogeneity
We planned the following subgroup analyses for all primary and secondary outcomes:
LAMA+LABA: 'combined indacaterol + glycopyrronium bromide (IND/GLY)' versus 'combined umeclidinium + vilanterol (UMEC/VI)' versus 'other LAMA/LABA inhalers'.
COPD severity: 'including only mild or moderate (or both) (% predicted FEV1 50% or greater)' versus 'including severe and/or very severe (% predicted FEV1 less than 50%)' versus 'including both categories.'
We were unable to perform the COPD severity subgroup analysis because separate data for participants with different severities were not reported.
We used the I2 test to detect heterogeneity, as discussed in Higgins 2003. We used the formal test for subgroup differences provided in Review Manager 5 (Review Manager 2014).
Sensitivity analysis
We conducted sensitivity analyses using a fixed‐effect model for the primary outcomes and reported any important differences compared with results using the random‐effects model.
Summary of findings and assessment of the certainty of the evidence
We created a summary of findings table to present the following outcomes:
exacerbations (participants with one or more);
serious adverse events (participants with one or more);
quality of life, as measured by the St. George's Respiratory Questionnaire (SGRQ) total score change from baseline;
trough FEV1 change from baseline;
pneumonia (participants with one or more occurrences);
all‐cause death;
SGRQ total score change from baseline (4 points or greater).
We used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the certainty of a body of evidence (very low, low, moderate, and high certainty of evidence) as it related to studies that contributed data to meta‐analyses for prespecified outcomes (Guyatt 2008). We used the methods and recommendations described in Section 8.5 and Chapter 12 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011), with GRADEpro software (GRADEpro). We justified all decisions to downgrade or upgrade the certainty of the evidence by using footnotes, and we provided comments to aid readers' understanding of the review when necessary.
We did not prespecify specific domains or percentages for downgrading due to risk of bias, but considered it reasonable to downgrade if studies contributing more than half the analysis weight were at high risk of bias in the 'other bias' domain and at least one other domain. For inconsistency, we downgraded if I2 was greater than 50%. For imprecision, we based downgrade decisions on prespecified thresholds (clinical importance 0.7 to 1.5 for outcomes as odds ratios) or established minimal clinically important differences (MCIDs) (4 points for SGRQ).
Results
Description of studies
See Included studies (Table 2), Excluded studies, and Ongoing studies.
1. Summary of characteristics of included studies.
| Study | LAMA+LABA | LABA+ICS | Key inclusion criteria | Follow‐up duration (weeks) | Mean/median age (years) | Number randomised |
| Beeh 2016 | Tiotropium/olodaterol (2.5/5 μg) or tiotropium/olodaterol (5/5 μg) | Salmeterol/fluticasone (50/250 μg) twice daily or salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 30% to 80% Ex(‐) | 6 × 4 time periods (cross‐over) | 64 | 229 |
| Donohue 2015a | Umeclidinium/vilanterol (62.5/25 μg) | Salmeterol/fluticasone (50/250 μg) twice daily | %pred FEV1 30% to 70%, mMRC ≥ 2, Ex(‐) | 12 | 63 | 707 |
| Donohue 2015b | Umeclidinium/vilanterol (62.5/25 μg) | Salmeterol/fluticasone (50/250 μg) twice daily | %pred FEV1 30% to 70%, mMRC ≥ 2, Ex(‐) | 12 | 64 | 700 |
| Ferguson 2018 | Glycopyrronium/formoterol fumarate (18/9.6 μg) | Formoterol/budesonide/ fumarate (160/4.8 μg) twice daily | CAT ≥ 10, %pred FEV1 25% to 80%, not required to have exacerbation | 24 | 65 | 943 |
| Frith 2018 | Glycopyrronium/indacaterol (50/110 μg) | Salmeterol/fluticasone (50/500 μg) twice daily | CAT ≥ 10, %pred FEV1 30% to 80%, Ex(+) | 12 | 65 | 502 |
| Herth 2020 | Tiotropium/olodaterol (5/5 μg) | Salmeterol/fluticasone (50/500 μg) | %pred FEV1 < 70%, Ex(‐) | 6 | 62 | 76 |
| Hoshino 2015 | Tiotropium/indacaterol (18/150 μg) | Salmeterol/fluticasone (50/250 μg) twice daily | %pred FEV1 30% to 80%, Ex(‐) | 16 | 71 | 46 |
| Lipson 2018 | Umeclidinium/vilanterol (62.5/25 μg) | Vilanterol/fluticasone furoate (25/100 μg) | CAT ≥ 10, %pred FEV1 < 80%, Ex(+) | 52 | 65 | 6204 |
| Magnussen 2012 | Tiotropium/salmeterol (18/50 μg) twice daily | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 ≤ 65%, Ex(‐) | 8 x 2 time periods (cross‐over) | 61 | 344 |
| Mostafa 2021 | Tiotropium (18 μg) once daily/formoterol (9 μg) twice daily | Formoterol/budesonide (160/4.5 μg) twice daily | %pred FEV1 30% to 80%, Ex(‐) | 12 | 64 | 40 |
| NCT03240575 | Tiotropium (5μg)/olodaterol (5μg) once daily | Salmeterol/fluticasone (50μg/250μg) twice daily | %pred FEV1 30% to 80%, Ex(‐) | 12 | 64 | 302 |
| Rabe 2008 | Tiotropium/formoterol (18/24 μg) twice daily | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 ≤ 65%, Ex(‐) | 6 | 62 | 605 |
| Rabe 2020 | Glycopyrronium/formoterol (9/4.8 µg) twice daily. | Formoterol/budesonide (4.8/160 µg) twice daily. | %pred FEV1 25‐65%, CAT ≥ 10, Ex(+) | 52 | 65 | 4294 |
| Singh 2015 | Umeclidinium/vilanterol (62.5/25 μg) | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 30% to 70%, mMRC ≥ 2, Ex(‐) | 12 | 62 | 717 |
| Vogelmeier 2013 | Indacaterol/glycopyrronium bromide (110/50 μg) | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 30% to 80%, Ex(‐) | 26 | 63 | 523 |
| Vogelmeier 2016 | Aclidinium/formoterol (400/12 μg) twice daily | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 < 80%, CAT ≥ 10, Ex(‐) | 24 | 63 | 933 |
| Vogelmeier 2017 | Glycopyrronium/Indacaterol (50/110 μg) | any | %pred FEV1 50% ‐80%, mMRC ≥ 1, Ex(+) | 12 | 65 | 1083 |
| Wedzicha 2016 | Indacaterol/glycopyrronium bromide (110/50 μg) | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 25% to 60%, mMRC ≥ 2, Ex(+) | 52 | 65 | 3362 |
| Zhong 2015 | Indacaterol/glycopyrronium bromide (110/50 μg) | Salmeterol/fluticasone (50/500 μg) twice daily | %pred FEV1 30% to 80%, mMRC ≥ 2, Ex(‐) | 26 | 65 | 744 |
%pred FEV1: % predicted forced expiratory volume in one second; CAT: chronic obstructive pulmonary disease assessment test; Ex(‐): without recent exacerbation; Ex(+): with recent exacerbation; LABA: long‐acting beta‐agonist; LAMA: long‐acting muscarinic antagonist; mMRC: modified Medical Research Council dyspnoea scale
Results of the search
We identified a total of 2685 records from update searches of electronic databases conducted on 1 November 2021 and 10 September 2022, including ClinicalTrials.gov and the WHO Clinical Trials Search Portal. We found an additional 20 records through manual searches. After removing 1248 duplicate records, there were 1458 records eligible for screening. The Screen4Me workflow removed 376 records, and we excluded 1046 records after viewing titles and abstracts alone. We excluded a further 21 after viewing full texts, which comprised 17 new excluded studies and four records identified as duplicates of records that had already been included. The remaining 15 records met the inclusion criteria for the review, corresponding to eight new included studies and one ongoing study.
Thus, we have included a total of 19 studies in the quantitative synthesis in the review (11 studies in the previous version of the review plus eight new included studies; see Figure 1).
1.
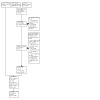
PRISMA study flow diagram
Included studies
The 19 studies included 22,354 participants. Seventeen studies used a parallel‐group design and two used a cross‐over design (Magnussen 2012; Vogelmeier 2017), with an 84‐day washout period between treatments. All studies delivered treatment in a double‐blind manner, except two open‐label studies (Hoshino 2015; Vogelmeier 2017).
The number of participants included in each study ranged from 40 to 6204, with a median of 700 participants per study. Thirteen studies included participants with moderate to severe COPD without recent exacerbation. Four trials that included only participants with a recent exacerbation accounted for 65% of the total participants. In each study, between 54% and 91% (median 70%) of participants were males. The mean age of the participants in each study ranged from 61 to 71 years (median 64 years). The percent predicted (%pred) FEV1 in each study ranged from 43% to 64%, with a median of 51.5%.
Treatment
Treatment duration ranged from six to 52 weeks. Of the LABA+ICS treatments used in these studies, 14 studies used combined salmeterol/fluticasone propionate (Beeh 2016; Donohue 2015a; Donohue 2015b; Frith 2018; Herth 2020; Hoshino 2015; Magnussen 2012; NCT03240575; Rabe 2008; Singh 2015; Vogelmeier 2013; Vogelmeier 2016; Wedzicha 2016; Zhong 2015), three studies used combined formoterol/budesonide (Ferguson 2018; Mostafa 2021; Rabe 2020), and one study used combined vilanterol/fluticasone furoate (Lipson 2018). One study permitted any type or dose of LABA+ICS (Vogelmeier 2017). Of the administered LAMA+LABA treatments, five studies used glycopyrronium/indacaterol (Frith 2018; Vogelmeier 2013; Vogelmeier 2017; Wedzicha 2016; Zhong 2015), four studies used umeclidinium/vilanterol (Donohue 2015a; Donohue 2015b; Lipson 2018; Singh 2015), two studies used tiotropium/formoterol (Rabe 2008; Mostafa 2021), three studies used tiotropium/olodaterol (Beeh 2016; Herth 2020; NCT03240575), two studies used glycopyrronium/formoterol (Ferguson 2018; Rabe 2020), one study used tiotropium/indacaterol (Hoshino 2015), one study used tiotropium/salmeterol (Magnussen 2012), and one study used aclidinium/formoterol (Vogelmeier 2016).
Outcomes
The reporting of outcomes across studies, organised by the type of LAMA+LABA combination investigated, is shown in Table 3. Thirteen studies reported exacerbations in a way that could be brought together in a meta‐analysis, 17 reported serious adverse events, 13 studies reported FEV1, 14 reported pneumonia, and 14 reported all‐cause death. Quality of life, as measured by the St. George's Respiratory Questionnaire (SGRQ), was less consistently reported, with nine studies reporting mean total scores and only four reporting the number of participants with a 4‐point or higher improvement on the scale. None of the included studies measured hospitalisations for COPD exacerbations.
2. ORBIT matrix of outcome reporting across included studies.
| Objective or semi‐objective outcomes (investigator‐assessed) | Subjective outcomes (patient‐reported) | ||||||
| Exacerbations | SAEs | Trough FEV1 | Pneumonia | All‐cause death | SGRQ (mean) | SGRQ ≥ 4 unit benefit | |
| Indacaterol/glycopyrronium studies | |||||||
| Frith 2018 | X | X | X | X | X | ||
| Vogelmeier 2013 | X | X | X | X | X | X | X |
| Vogelmeier 2017 | X | X | X | X | X | ||
| Wedzicha 2016 | X | X | X | X | X | X | X |
| Zhong 2015 | X | X | X | X | |||
| Umeclidinium/vilanterol studies | |||||||
| Donohue 2015a | X | X | X | X | X | X | |
| Donohue 2015b | X | X | X | X | X | X | |
| Lipson 2018 | X | X | X | X | X | X | X |
| Singh 2015 | X | X | X | X | X | X | |
| Other LAMA/LABA inhaler studies | |||||||
| Beeh 2016 | X | X | X | X | |||
| Ferguson 2018 | X | X | X | X | X | ||
| Herth 2020 | X | ||||||
| Hoshino 2015 | X | ||||||
| Magnussen 2012 | X | X | |||||
| Mostafa 2021 | X | X | |||||
| NCT03240575 | X | X | X | X | |||
| Rabe 2008 | X | X | |||||
| Rabe 2020 | X | X | X | X | X | X | |
| Vogelmeier 2016 | X | X | X | ||||
Outcomes shown are those included in the summary of findings table. Outcomes are classed as objective or subjective for the purposes of risk of bias assessment to consider differences in performance and detection bias.
FEV1: forced expiratory volume in one second; ORBIT: outcome reporting bias in trials; SAEs: serious adverse events; SGRQ: St George’s Respiratory Questionnaire
Excluded studies
We excluded 24 studies after viewing full texts (see Characteristics of excluded studies and Figure 1), of which 17 were new excluded studies in this update. Overall, 11 were not relevant because they did not compare LAMA+LABA versus LABA+ICS (Bruhn 2003; Calverley 2007; Knobil 2004a; Knobil 2004b; NCT00120978; Sciurba 2004; NCT03504527; NCT04320342; NCT04923347; NCT05097014; Papi 2018), three studies were not eligible because they were systematic reviews (Aziz 2018; Oba 2016; Pavord 2016), four studies were not eligible because they did not report an RCT (Anzueto 2017; Mahler 2016; NCT03376295; NCT04138758), two studies were not eligible because they reported cost‐effectiveness analyses (Price 2014; Skoupa 2018), two studies were letters or commentaries (Michael 2016; Singh 2019), one study was not eligible because it reported only patients with eosinophilic inflammation (UMIN000024905), and one study was not eligible because the data were only analysed for asthma‐COPD overlap (EUCTR2015‐002046‐31‐ES).
Ongoing studies
We found one ongoing study awaiting results (EUCTR2016‐004473‐41‐GB). The study, which started in 2016, is a trial comparing tiotropium/olodaterol with vilanterol/fluticasone furoate, sponsored by Mundipharma Research Limited (see Characteristics of ongoing studies). The primary end point is change from baseline in eosinophil count and one of the secondary end points is change from baseline in pre‐dose FEV1.
Risk of bias in included studies
Included studies had generally low risk of bias for random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective outcome reporting. The potential for performance and detection bias for different types of outcome (e.g. subjective, objective) is discussed under the Blinding heading. See Figure 2.
2.

Risk of bias summary: included studies had generally low risk of bias for random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective outcome reporting.
Allocation
Nine studies reported methods of random sequence generation that suggested a low risk of bias (Donohue 2015a; Donohue 2015b; Frith 2018; Magnussen 2012; NCT03240575; Singh 2015; Vogelmeier 2013; Wedzicha 2016; Zhong 2015). Ten studies had an unclear risk of bias in this domain because no details were reported (Beeh 2016; Ferguson 2018; Herth 2020; Hoshino 2015; Lipson 2018; Mostafa 2021; Rabe 2008; Rabe 2020 Vogelmeier 2016; Vogelmeier 2017). We rated 10 studies at low risk of bias relating to allocation concealment (Donohue 2015a; Donohue 2015b; Frith 2018; Magnussen 2012; Mostafa 2021; NCT03240575; Singh 2015; Vogelmeier 2013; Wedzicha 2016; Zhong 2015), two at high risk (Ferguson 2018; Herth 2020), and seven as unclear risk of bias because detailed methods were not provided (Beeh 2016; Hoshino 2015; Lipson 2018; Rabe 2008; Rabe 2020; Vogelmeier 2016; Vogelmeier 2017).
Blinding
We considered the impact of performance and detection bias on subjective and objective outcomes depending on the blinding methods used within studies. The reporting of objective, semi‐objective, and subjective outcomes across studies is shown in Table 3. Though bias is still possible in double‐blind studies for subjective and semi‐objective outcomes where blinding is broken unintentionally, there was no evidence that this had happened. Therefore, we rated all 17 double‐blind studies as low risk of performance and detection bias (Beeh 2016; Donohue 2015a; Donohue 2015b; Frith 2018; Ferguson 2018; Herth 2020; Lipson 2018; Magnussen 2012; Mostafa 2021; NCT03240575; Rabe 2008; Rabe 2020; Singh 2015; Vogelmeier 2013; Vogelmeier 2016; Wedzicha 2016;Zhong 2015).
Two studies adopted neither double‐ nor single‐blinding methods and we rated these high risk of bias for both blinding domains, because there is a risk of bias for all outcomes except all‐cause death (Hoshino 2015; Vogelmeier 2017).
Incomplete outcome data
Our prespecified criteria for high attrition bias was a dropout rate of more than 20% of randomised participants. Two trials had a dropout rate of more than 20% (Mostafa 2021; Rabe 2020), and we therefore rated them high risk of bias. We considered all other studies to be at low risk of attrition bias.
Selective reporting
Our criteria for rating a study as having a risk of selective reporting bias was if it was a non‐registered trial or if it considerably deviated from the registered protocol concerning outcome reporting. Two trials had a high risk of selective reporting bias due to non‐registration (Hoshino 2015; Mostafa 2021).
Other potential sources of bias
Seventeen out of 19 trials were sponsored by pharmaceutical companies (Beeh 2016; Donohue 2015a; Donohue 2015b; Frith 2018; Ferguson 2018; Herth 2020; Lipson 2018; Magnussen 2012; NCT03240575; Rabe 2008; Rabe 2020; Singh 2015; Vogelmeier 2013; Vogelmeier 2016; Vogelmeier 2017; Wedzicha 2016; Zhong 2015). Although pharmaceutical company sponsorship does not automatically signify a high risk of bias, scrutiny of disclosure statements revealed that all 17 studies had authors that were employed by the company, held shares, and/or received grants or fees for the work. In all cases, these authors were involved explicitly in the design of the study, its conduct (including data analysis), or interpretation and write up, which warranted high risk of bias judgements.
We found no other sources of bias apart from conflicts of interest.
Effects of interventions
See: Table 1
See Table 1.
Primary outcomes
Exacerbations (participants with one or more event)
Thirteen studies with 20,960 participants evaluated exacerbations over six to 52 weeks of observation. Participants randomised to LAMA+LABA had similar odds of experiencing one or more exacerbations to those randomised to LABA+ICS (OR 0.91, 95% CI 0.78 to 1.06; P = 0.22, I2 = 56%; moderate‐certainty evidence; Analysis 1.1). Our confidence in the result was reduced from high to moderate due to considerable heterogeneity.
1.1. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 1: Exacerbations
The test for subgroup differences indicated a difference between LAMA+LABA subgroups, with the largest benefit of LAMA+LABA versus LABA+ICS seen in the glycopyrronium/indacaterol studies (OR 0.72, 95% CI 0.63 to 0.83; P = 0.02, I2 = 0%) and the pooled effect for the umeclidinium/vilanterol subgroup laying in the opposite direction (OR 1.20, 95% CI 1.03 to 1.40; P < 0.001, I2 = 0%; test for subgroup differences P < 0.001). However, results are observational and should be interpreted with caution. The overall pooled result was similar to the random‐effects analysis when a fixed‐effect model was tested (OR 0.94, 95% CI 0.87 to 1.01; P = 0.08, I2 = 56%; Analysis 1.8).
1.8. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 8: Exacerbations (fixed‐effect sensitivity)
Serious adverse events (participants with one or more event)
Eighteen studies with 23,183 participants evaluated serious adverse events over six to 52 weeks of observation. We discarded data from one study because no serious adverse events were reported in either arm (Herth 2020). Participants randomised to LAMA+LABA had similar odds of having a serious adverse event as those randomised to LABA+ICS (OR 1.02, 95% CI 0.91 to 1.15; I2 = 20%; high‐certainty evidence; Analysis 1.2; test for subgroup differences P = 0.25). We did not downgrade the evidence in any of the GRADE domains and therefore have high confidence in the result.
1.2. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 2: Serious adverse events
Results were similar to the random‐effects analysis when a fixed‐effect model was tested (OR 1.03, 95% CI 0.95 to 1.12; I2 = 20%; Analysis 1.9).
1.9. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 9: Serious adverse events (fixed‐effect sensitivity)
Quality of life, as measured by the St. George's Respiratory Questionnaire (SGRQ) total score change from baseline
Nine studies with 14,437 participants assessed mean change in SGRQ scores over 12 to 52 weeks of observation. The benefit of LAMA+LABA versus LAMA+ICS was neither statistically nor clinically important (MD ‐0.57, 95% CI ‐1.36 to 0.21; I2 = 78%; moderate‐certainty evidence; Analysis 1.3; test for subgroup differences P < 0.004). Our confidence in the result was reduced from high to moderate due to considerable heterogeneity.
1.3. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 3: Quality of life as measured by the St George's Respiratory Questionnaire (SGRQ) total score change from baseline
The test for subgroup differences indicated a difference between LAMA+LABA subgroups, with the largest benefit of LAMA+LABA versus LABA+ICS seen in the indacaterol/glycopyrronium studies (MD ‐1.29, 95% CI ‐2.08 to ‐0.50; P = 0.001, I2 = 0%). The sensitivity analysis using a fixed‐effect model brought the point estimate from lying in favour of LAMA+LABA to zero (i.e. no effect) (MD 0.00, 95% CI ‐0.02 to 0.02; I2 = 78%; Analysis 1.10).
1.10. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 10: SGRQ mean change (fixed‐effect sensitivity)
Trough FEV1 mean change
In total, 12 studies with 14,681 participants reported change in trough FEV1 over six to 52 weeks. Compared to LABA+ICS, there was a significant increase in the trough FEV1 change from baseline with LAMA+LABA (MD 0.07 L, 95% CI 0.05 to 0.08; I2 = 73%; moderate‐certainty evidence; Analysis 1.4). This difference was less than the minimal clinically important difference (Donohue 2005). Our confidence in the result was reduced from high to moderate due to considerable heterogeneity.
1.4. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 4: Trough FEV1 mean change (litres)
In the LAMA+LABA subgroup analysis, each subgroup was consistently associated with an increase in trough FEV1 change from baseline (test for subgroup differences P = 0.93). Results from the sensitivity analysis using a fixed‐effect model were in line with the main random‐effects analysis (MD 0.06, 95% CI 0.05 to 0.07; I2 = 73%; Analysis 1.11).
1.11. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 11: Trough FEV1 mean change (fixed‐effect sensitivity)
Secondary outcomes
Pneumonia (participants with one or more event)
Fourteen studies with 21,829 participants evaluated pneumonia over 12 to 52 weeks of observation. Compared to LABA+ICS, there was a large reduction in the number of participants experiencing one or more episodes of pneumonia with LAMA+LABA (OR 0.61, 95% CI 0.52 to 0.72; I2 = 0%; high‐certainty evidence; Analysis 1.5; test for subgroup differences P = 0.90). We did not downgrade the evidence in any of the GRADE domains and therefore have high confidence in the result. Although it would be possible to calculate an absolute risk reduction, we decided not to do so, as the absolute effect size is highly dependent on the study duration.
1.5. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 5: Pneumonia
All‐cause death
Fifteen studies with 21,510 participants evaluated all‐cause death at six to 52 weeks of observation. There was an increased risk of all‐cause death with LAMA+LABA compared with LABA+ICS (OR 1.35, 95% CI 1.05 to 1.75; I2 = 0%; moderate‐certainty evidence; Analysis 1.6). Our confidence in the result was reduced from high to moderate due to imprecision in the effect.
1.6. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 6: All‐cause death
Point estimates and confidence intervals varied across subgroups containing studies of different LAMA+LABA combinations, but the test for subgroup differences indicated the differences were not statistically significant (test for subgroup differences P = 0.51).
SGRQ improvement of 4 points or greater
Four studies with 13,614 participants evaluated the SGRQ total score change from baseline (≥ 4 points) at 26 to 52 weeks of observation. There was little to no significant change in the SGRQ total score (4 points or greater) between LAMA+LABA and LABA+ICS (OR 1.06, 95% CI 0.90 to 1.25; I2 = 77%; moderate‐certainty evidence; Analysis 1.7; test for subgroup differences P = 0.002). Our confidence in the result was reduced from high to moderate due to considerable heterogeneity.
1.7. Analysis.

Comparison 1: Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS), Outcome 7: SGRQ improvement ≥ 4 points
Hospitalisations for COPD exacerbations
None of the included studies reported this outcome.
Discussion
Summary of main results
We conducted a systematic review and meta‐analysis to compare the efficacy and safety of LAMA+LABA and LABA+ICS therapy in people with stable COPD. The review includes 19 studies with 22,354 participants, including 11 studies from the previous version of the review. Most studies were parallel, double‐blind RCTs. We noted selection, attrition, and selective reporting bias in a small number of studies and there is a risk of bias from involvement of authors employed by the pharmaceutical sponsors in 17 of the 19 studies.
Compared to LABA+ICS, the review found there to be a greater improvement in FEV1 in the LAMA+LABA group (moderate‐certainty evidence), lower odds of pneumonia (high‐certainty evidence), and increased mortality (moderate‐certainty evidence). This update did not find a difference in exacerbations between LAMA+LABA and LABA+ICS (moderate‐certainty evidence), which was reported in the previous review (Horita 2017). The odds of having a serious adverse event are likely to be similar for LAMA+LABA and LABA+ICS (high‐certainty evidence), as were quality of life scores as measured by the St George's Respiratory Questionnaire (moderate‐certainty evidence).
Overall completeness and applicability of evidence
Most of the studies included in this analysis recruited people with moderate to severe COPD, according to the GOLD reports. Therefore, attention should be paid when applying our results to people with mild and very severe COPD.
Despite the apparent benefits of LAMA+LABA over LABA+ICS for trough FEV1 and reduced risk of pneumonia, the findings suggest little difference for exacerbations, serious adverse events, and quality of life as measured by the SGRQ. Although exacerbations and pneumonia should share risk factors, a paradox can occur in COPD treated with ICS where the risk of exacerbations and mortality reduces despite an increased risk of pneumonia. The apparent conflicting results of this review may partially relate to this relationship, and should also be interpreted with caution due to the limited follow‐up and number of observed events. It should also be noted that studies recruited outpatient populations and cases of pneumonia may have been mild. Variation in study results also reduced our confidence in some of these findings, and in some cases, such as the exacerbations analysis where Lipson 2018 reported a significant benefit in favour of LABA+ICS, outlier studies impacted on the overall findings.
There was also variation across the included studies in their exclusion of people with a history of asthma, and the reporting of blood eosinophil counts that may be indicative of ACOS was patchy. Given that ICSs are effective in preventing exacerbations in asthma (Reddel 2021), results may have been skewed in favour of the LABA+ICS group because people with ACOS or a high blood eosinophil sensitivity to ICS may have fared better than those receiving LAMA+LABA.
Studies further varied in their inclusion criteria around recent history of exacerbation, which is a known risk factor for COPD mortality (Esteban 2018). The variation may have impacted on analyses where studies that enroled participants with a history of exacerbations carried a large proportion of the weight within an analysis (e.g. Rabe 2020 in the all‐cause death analysis). Moreover, the % predicted FEV1 of participants in Rabe 2020 was as low as 25% to 65%, suggesting that participants' condition in this trial was poorer than those in other RCTs.
Limitations of the evidence include the lack of long‐term follow‐up data and the possibility that discontinuation of therapy may have contributed to early events such as withdrawal and death. For example, in Wedzicha 2016, all participants were treated with tiotropium during a one‐month run‐in period. Exacerbations in the LAMA+LABA groups might have occurred because participants who had been using ICSs prior to the trial had to discontinue them. As such, in studies where a recent exacerbation was not permitted, removal of these participants may have led to a biased population. In Lipson 2018, the participants who had been receiving ICSs before enroling in the trial were partly randomised to the LAMA+LABA group. These participants were actually stepping down in their treatment; this could lead to COPD exacerbations (Suissa 2018).
Certainty of the evidence
GRADE ratings of certainty in the evidence along with reasons for downgrading, or choosing not to downgrade, are summarised in Table 1. We downgraded most outcomes once to moderate certainty, meaning the true effect is likely to be close to the estimate of effect but may be substantially different. The pooled effects for serious adverse events and pneumonia were both rated high certainty, meaning the true effect lies close to that of the estimate of effect. The most common reason for downgrading was considerable heterogeneity (exacerbations, SGRQ mean change, FEV1 mean change and SGRQ improvement of 4 or more points), which may reflect the clinical heterogeneity between studies, discussed above. Only the all‐cause death analysis was downgraded due to imprecision, because there was a very small number of events in the analysis, leading to wide confidence intervals. The confidence intervals around the pooled result for several other outcomes include a potential benefit in either direction (exacerbations, serious adverse events, SGRQ mean change, SGRQ improvement of 4 or more points), but they lay within the threshold for clinical importance that was predefined in the protocol for this review, so we chose not to downgrade (0.7 to 1.5 for OR outcomes; 4‐point MCID for SGRQ).
We did not downgrade any outcomes due to indirectness of the contributing studies to the review question. However, variation in study inclusion criteria around recent history of exacerbation, baseline percentage predicted FEV1, and exclusion of asthma are noteworthy, and affect how the pooled results can be interpreted and applied to different subpopulations of people with COPD. Similarly, we did not downgrade any outcomes for suspected publication bias, and there was no obvious asymmetry on funnel plots for the primary outcomes.
The potential impact of risk of bias across analyses is difficult to quantify, and we did not prespecify a threshold for downgrading the evidence due to risk of bias in certain domains or in studies contributing a certain percentage of the analysis weight. Amongst the 19 included studies, only two with a few participants did not have any commercial sponsorship (Hoshino 2015; Mostafa 2021). In the remaining 17 studies, study authors involved in key aspects of the study were employed by, held shares for, or received fees from the sponsoring manufacturer (Table 4). Otherwise, the included studies were mostly well‐designed and conducted, with adequate sample sizes. Therefore, across outcomes, all or almost all studies are at high risk of 'other' bias due to conflicts of interest, and studies contributing between 18.5% and 35.9% of the analysis weight are rated high risk of bias in at least one other domain. We set a post hoc rule for downgrading, and considered it insufficient to downgrade if studies contributing more than half the analysis weight were at low or unclear risk of bias in all but the 'other' domain, meaning no outcomes were downgraded for this reason. There is a need for large, well‐conducted trials funded by national funding bodies to overcome the bias inherent in evidence funded by pharmaceutical companies.
3. Sponsor list for chronic obstructive pulmonary disease studies.
| Sponsor | Record count | % of 1723 |
| GlaxoSmithKline | 134 | 7.78 |
| Novartis | 128 | 7.43 |
| AstraZeneca | 122 | 7.08 |
| Boehringer Ingelheim | 113 | 6.56 |
| Pfizer | 84 | 4.88 |
| Nycomed | 49 | 2.84 |
| GSK | 45 | 2.61 |
| Chiesi | 41 | 2.38 |
| Almirall | 36 | 2.09 |
| Merck | 30 | 1.74 |
Web of Science Core Collection, advanced search for "TI=(COPD) AND TS=(inhal*)" without any restriction hit 1723 reports as of 13 June 2016. "Results analysis" > "Source Titles" output the table above.
Potential biases in the review process
Although we tried to extract data on exacerbations of any severity, some trials counted only moderate to severe exacerbations. In addition, some outcomes, such as exacerbation and adverse effects, were dependent on the threshold determined by the researchers of the original articles. Confidence intervals for the two cross‐over studies might be conservative in our analysis (Magnussen 2012; Vogelmeier 2017), due to the way we included the data in the analysis to avoid overestimating precision.
Agreements and disagreements with other studies or reviews
The previous version of this review, which included 11 studies with 9839 participants (Horita 2017), concluded there was no difference between the LAMA+LABA and LABA+ICS groups for all‐cause death (OR 1.01, 95% CI 0.61 to 1.67), whereas this update found a probable benefit of LABA+ICS over LAMA+LABA. As discussed above, this may be partially explained by differences in the presence and reporting of ACOS within the newer studies that carry a large proportion of the analysis weight.
A recent network meta‐analysis which included a LAMA+LABA versus LABA+ICS comparison looked at many of the same outcomes to this review in studies of low‐ and high‐risk COPD populations (Oba 2018). The review included many of the same studies (nine studies), and many others for comparisons not of interest to this review, but excluded those with a cross‐over design and those shorter than 12 weeks. Oba and colleagues also presented data separately for high‐ and low‐risk populations and looked at FEV1 and the SGRQ at three, six, and 12 months. Results for the high‐risk population are largely in line with this review: the finding relating to exacerbations was similar to ours (OR 0.89, 95% CI 0.78 to 1.04) and quality of life, as measured by the SGRQ (MD ‐1.47, 95% CI ‐3.74 to 0.45). It is noteworthy that there was no significant difference in all‐cause mortality (OR 1.15, 95% CI 0.70 to 1.95). A similarly large effect was seen for pneumonia (OR 0.61, 95% CI 0.34 to 1.01) and trough FEV1 (MD 0.05, 95% CI 0.03 to 0.07) in favour of LAMA+LABA, and effects were generally smaller or inconclusive in the low‐risk population studies (Oba 2018). A further review of incident pneumonia and mortality in people with COPD showed no difference between ICS and non‐ICS arms in pneumonia‐related mortality despite a significantly increased risk of pneumonia (Festic 2015).
Authors' conclusions
Implications for practice.
Combined long‐acting muscarinic antagonist (LAMA) and long‐acting beta‐agonist (LABA) inhalers probably hold similar benefits to LABA plus inhaled corticosteroid (ICS) inhalers for exacerbations and quality of life (as measured by the St George's Respiratory Questionnaire) for people with moderate to severe chronic obstructive pulmonary disease (COPD), but offer a larger improvement in forced expiratory volume in one second (FEV1)and a lower risk of pneumonia. Clinicians need to consider pneumonia as an underlying cause when people with COPD using LABA plus ICS (LABA+ICS) inhalers have exacerbations. All‐cause death may be lower with LABA+ICS. However, there was a very small number of events in the analysis, which means absolute risk was low. Findings are based on moderate‐ to high‐certainty evidence from heterogeneous trials with an observation period of less than one year.
The choice between LAMA+LABA and LABA+ICS may need to take account of the individual's condition, including blood eosinophil count, history of pneumonia, and recent exacerbations. This review should be updated again in a few years.
Implications for research.
Further research is indicated to clarify the relative positions of LABA+ICS and LAMA+LABA in the COPD treatment guidelines. Trials should exclude people with asthma and should rapidly switch participants from any prior treatment to trial treatment to reduce the number of people who withdraw from the study. Longer‐term follow‐up data would be beneficial, especially to identify any impact on serious adverse events or mortality. Results from future or ongoing trials evaluating newly developed bronchodilators are awaited. Meta‐analyses that access the data for each combined medication separately are also anticipated.
Feedback
Interpretation of SGRQ data and missing data, 20 March 2017
Summary
My colleagues and I thank Dr. Horita and colleagues for their efforts on their Cochrane Review (1). With the recent publication of the FLAME trial (2) that compared the utility of indacaterol‐glycopyrronium versus salmeterol‐fluticasone for chronic obstructive pulmonary disease (COPD) and its potential to change our current standard of care, we appreciate their work in evaluating the results of this trial and putting this evidence into perspective.
As we reviewed the manuscripts presented in the article, we focused on the applicability of the evidence. The authors reported that for the treatment of COPD, combination LAMA+LABA was associated with more frequent St. George’s Respiratory Questionnaire (SGRQ) total score improvement exceeding the minimal clinically important difference (4 points or greater).(1) Respectively, after critically appraising the data presented, we have reservations about the conclusions made regarding the impact of LABA+LAMA on the SGRQ. Please refer to our additional letter regarding the outcomes of rate of exacerbations and serious adverse events.
In this review, the outcome of SGRQ total score improvement (≥4 points) was based on two trials with the FLAME trial driving the results (Analysis 1.7).(1) We are concerned that the data set used for this outcome from FLAME is incomplete. Rather than using the modified intention to treat (mITT) population (N=3354) or even the per protocol population (N=3084), it appears that only patients who completed 52 weeks of treatment were included (N=2760).(2) It is possible that the exclusion of these 594 patients may have skewed the results of this analysis. Furthermore, it is unclear where the data used for this outcome came from, as neither the FLAME publication nor the supplemental appendices identify the SGRQ results for this population of patients who completed 52 weeks of treatment. This information is also not available through clinicaltrials.gov. In Figure 6 of this review, the outcome of mean difference in SGRQ from baseline was also driven by this same population.(1) Therefore, this begs the question of whether it is fair to base conclusions regarding the effects of LABA+LAMA versus LABA+ICS on SGRQ on the results of only two trials especially given that the larger trial’s data set does not appear to be complete.
In summary, with greater transparency and citation of the original data used for analyses, we believe that our conclusions potentially could align more closely with those identified by the Cochrane review authors. At present, perhaps the conclusions regarding the impact of these interventions on SGRQ could incorporate a greater sense of uncertainty and/or acknowledgement of the limitations of the available evidence. Overall, aside from the clarification of the data reported in the Cochrane review, we agree with the authors that a call for more research is warranted to ascertain the relative position of LAMA+LABA and LABA+ICS for COPD treatment and disaggregated data for participants in different COPD severity groups.
We hope that you will consider our constructive feedback and look forward to hearing from you soon.
References:
Horita N, Goto A, Shibata Y, Ota E, Nakashima K, Nagai K, Kaneko T. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev. 2017 Feb 10;2:CD012066.
Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, Thach C, Fogel R, Patalano F, Vogelmeier CF, FLAME Investigators. Indacaterol‐Glycopyrronium versus Salmterol‐Fluticasone for COPD. N Engl J Med. 2016 Jun 9;374(23):2222‐34.
Reply
Cochrane Airways awaits a final response from the author team.
Contributors
Stephanie Garland, BSc. Pharm, Pharmacy Resident Andrea Wan, BSc. Pharm, Pharmacy Resident Anna Yee, BSc. Pharm, Pharmacy Resident Grant Gill, BSc. Pharm, Pharmacy Resident Aaron Tejani, BSc. Pharm, PharmD
Exacerbations and missing data, 13 April 2017
Summary
With the recent publication of the FLAME trial (1) and its potential to change clinical practice, we would like to thank Dr. Horita and colleagues for their efforts in evaluating the results of this trial and putting this evidence into perspective in their Cochrane Review (2). The authors reported that for the treatment of COPD, combination LAMA+LABA was associated with fewer exacerbations, and a non‐significant decrease in serious adverse events (SAE) (2). Respectively, we have reservations in making similar claims and would like to address our chief concerns below.
First, we would like to focus on the reported reduction in the rate of COPD exacerbations and the implications based on the data set analysed. As illustrated in Figure 3, the FLAME trial was a primary driver of this finding, and the only trial to show a statistically significant reduction in the number of exacerbations.(2) When examining the FLAME data set, we were concerned that the data used to make this conclusion was incomplete. Of the 598 patients who did not complete 52 weeks of treatment, only 192 patients were continually followed with respect to exacerbations and adverse events (1). Thus, 406 patients were unaccounted for in both groups combined that may have experienced an exacerbation and unbalanced the intervention and control groups. Without delineating how these patients may have skewed the results, we are less confident that the results observed were the true effect. Moreover, of the 1680 patients randomised to indacaterol‐glycopyrronium and 1682 patients randomised to salmeterol‐fluticasone, 152 and 126 patients were unaccounted for in each group respectively in the per protocol analysis (1). Interestingly, the data from the per protocol analysis was used to quantify the primary outcomes in the FLAME trial, which complicates whether the observed effects are reliable. Additionally, when comparing the results of FLAME to those reported in this review, we found a discrepancy in exacerbation reporting. In Figure 3, 1290/1675 and 1377/1679 exacerbation events were reported in the LAMA+LABA and LABA+ICS groups respectively (2). However, it is unclear where this data originated from, as neither the FLAME publication, the supplemental appendices, nor the data from clinicaltrials.gov provided these exact results. Regarding the generalisability of the outcome data, patients in the FLAME trial experienced an average of 3.59 or 4.09 exacerbations during the 52 weeks of indacaterol‐glycopyrronium and salmeterol‐fluticasone treatment respectively, and these rates were primarily driven the total number of mild exacerbations recorded (1). Thus, it would be difficult to meaningfully apply this data to patients with chronic COPD who experience moderate to severe exacerbations. Given the potentially incomplete data from FLAME, the discrepancy in the data used, and the limited applicability of the primary outcome results, it is difficult to draw a definitive conclusion about the effects of LAMA+LABA compared to LABA+ICS on reducing the rate of COPD exacerbations at this time.
Second, despite the reduction in COPD exacerbations and the risk of pneumonia in patients randomised to the LAMA+LABA group, these differences failed to translate into statistically significant reductions in SAE and adverse events (AE), and produced no change in all cause mortality between the two treatment modalities. These discordant findings raise further questions about the validity of the data being meta‐analyzed. Predictably, we have identified the FLAME trial as a contributor to these potentially skewed results due to its ambiguous reporting of outcome data that does not outline the proportion of SAE, AE, or mortality following early discontinuation of study drugs and early withdrawal. As mentioned above, 406 patients were unaccounted for and without information on whether these patients may have experienced additional SAE or AE, it is difficult to interpret and make conclusions about the true effect.
In summary, with greater transparency and citation of the original data used for analyses, and acknowledgement of the limitations in the demographics of the studied patient population and reporting of primary outcomes, we believe that our conclusions could potentially more closely align with those identified by the Cochrane review authors. Acutely, perhaps the conclusions originally stated could incorporate a greater sense of uncertainty, and/or caveats to the generalizations provided. To close, we agree with the authors that a call for more research to ascertain the relative position of LAMA+LABA and LABA+ICS for COPD treatment and disaggregated data for participants in different COPD severity groups is warranted.
We hope that you will consider our constructive feedback and look forward to hearing from you soon.
The authors of this letter have no known conflicts of interest to declare.
References:
Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, Thach C, Fogel R, Patalano F, Vogelmeier CF, FLAME Investigators. Indacaterol‐Glycopyrronium versus Salmterol‐Fluticasone for COPD. N Engl J Med. 2016 Jun 9;374(23):2222‐34.
Horita N, Goto A, Shibata Y, Ota E, Nakashima K, Nagai K, Kaneko T. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev. 2017 Feb 10;2:CD012066.
Reply
Cochrane Airways awaits a final response from the author team.
Contributors
Andrea Wan, BSc. Pharm, Pharmacy Resident Anna Yee, BSc. Pharm, Pharmacy Resident Grant Gill, BSc. Pharm, Pharmacy Resident Stephanie Garland, BSc. Pharm, Pharmacy Resident Aaron Tejani, BSc. Pharm, PharmD
Questioning the pooling of exacerbation data, 18 December 2017
Summary
I read with great interest the systematic review on Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD) (1). The review is illustrated with a forest plot of the association of combined therapy with COPD exacerbation. This figure shows that indacaterol/glycopyrronium is the only combination able to prevent exacerbations.
The first step when a reader goes through a systematic review is to assess its credibility. In the case of this forest plot, this credibility is questioned when we inspect the rate of events. The indacaterol/glycopyrronium trials had a 66.4% of events in the control group, meanwhile it was 2.4% in the umeclidinium/vilanterol trials and 11.1% in the other inhalers. Although there is no statistically significant heterogeneity according to Cochrane Q test or I2, it is questionable to construct a forest plot in the presence of significant clinical heterogeneity (e.g. trials with exacerbation rates ranging between 2.4 to 66.4%).
The FLAME trial (2) was the only one designed to assess COPD exacerbations. This trial included patients with recent exacerbations and followed them for 52 weeks. In contrast, the other trials included patients without recent COPD exacerbations and followed them for only 6 to 12 weeks. It is nearly impossible to draw conclusions about the role of LAMA/LABA in the prevention of COPD exacerbation with these last trial designs. Neither the population, nor the follow‐up period were adequate to assess the prevention of COPD exacerbation. The importance of the follow‐up length could be illustrated in figure 2 of the own FLAME trial (2), where there were no differences in the rate of severe exacerbations until week 32.
We can be fairly confident about indacaterol/glycopyrronium preventing COPD exacerbations for patients with frequent exacerbations, but we do not have either enough evidence about the role of the others LABA/LAMA combinations to prevent COPD exacerbations, or the role of indacaterol/glycopyrronium in patients without frequent exacerbations.
In my opinion, the forest plot in question is misleading due its combination of apples and oranges.
Luis Corral‐Gudino
References
Horita N, Goto A, Shibata Y, Ota E, Nakashima K, Nagai K, Kaneko T. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev. 2017 Feb 10;2:CD012066.
Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, Thach C, Fogel R, Patalano F, Vogelmeier CF. Indacaterol–glycopyrronium versus salmeterol–fluticasone for COPD. N Engl J Med. 2016 Jun 9;2016(374):2222‐34.
Reply
We appreciate Dr. Corral‐Gudino's comments concerning the exacerbation forest plot in our review [1].
As correctly mentioned, the subgroup analysis of the exacerbation forest plot shows non‐negligible discrepancy. This planned subgroup analysis is impressive. While indacaterol/glycopyrronium clearly decreased the risk of exacerbation, medications in the other two subgroups did not. I2 for subgroup difference is as high as 69.7% with significant P value of 0.04. We agree with Dr. Corral‐Gudino that only indacaterol/glycopyrronium seemed to prevent exacerbations, which we discussed in the ‘Overall Completeness and Applicability of Evidence’ section of our review [1]. Given some limitations, the GRADE quality of evidence related to the outcome of exacerbations was downgraded to 'low.'[1]
Dr. Corral‐Gudino rightly commented that the study designs of included studies varied greatly. Thus, the between‐study heterogeneity should be assessed. It is reasonable to evaluate the heterogeneity using Cochrane Q test (P value = 0.29) and I2 statics (17%), which dispelled considerable inconsistency (Figure 1) [1].
The frequencies of exacerbation were indeed different between studies. Absolute risk reduction and number needed to treatment are meaningless if event frequencies are largely different among studies. However, odds ratios are known to be robust even when exacerbation frequencies vary greatly. For example, the incidence of exacerbations in Vogelmeier's, Wedzicha's and Zhong's studies were (44+62)/(258+264)=20%, (1290+1377)/(1675/1679)=80%, and (75+97)/(372/369)=23%, respectively. Yet the corresponding odds ratios were 0.67, 0.73, and 0.71 [1]. Compared to patients who were treated by control medication, those treated by indacaterol/glycopyrronium had a 30% lower chance of exacerbation regardless of absolute risk. As long as odds ratios are calculated, inconsistent frequencies between studies is not a considerable problem.
While Wedzicha's trial included patients with recent exacerbation, Vogeleier's and Zhong's trials included patients without recent exacerbation. Dr. Corral‐Gudino insists we do not have enough evidence about the role of indacaterol/glycopyrronium in patients without frequent exacerbations. However, our analysis clearly shows that the medication constantly decreased the risk of exacerbation in the three trials, with an odds ratio of approximately 0.7 without heterogeneity (I2=0%, P for heterogeneity = 0.92) regardless of recent exacerbations.
As Dr. Corral‐Gudino pointed out, observed exacerbations in each study were not sufficient in all studies, except Wedzicha's, due to small sample size, inclusion of patients without recent exacerbation, and short follow‐up periods. This is exactly why meta‐analysis is needed. A meta‐analysis can integrate many studies that do not have statistical power.
Reference
Horita N, Goto A, Shibata Y, Ota E, Nakashima K, Nagai K, Kaneko T. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database Syst Rev. 2017 Feb 10;2:CD012066.
Contributors
Feedback submitter
Luis Corral‐Gudino PhD MD, Internal Medicine Department. Hospital El Bierzo, Ponferrada Leon.
Do you have any affiliation with or involvement in any organisation with a financial interest in the subject matter of your comment?
I do not have any affiliation with or involvement in any organisation with a financial interest in the subject matter of my comment
Authors
Nobuyuki Horita, Atsushi Goto, Yuji Shibata, Erika Ota, Kentaro Nakashima, Kenjiro Nagai, Takeshi Kaneko
What's new
| Date | Event | Description |
|---|---|---|
| 5 June 2023 | New search has been performed | New search run September 10, 2022. |
| 5 June 2023 | New citation required and conclusions have changed | New studies identified; review redrafted; conclusions changed. |
History
Protocol first published: Issue 2, 2016 Review first published: Issue 2, 2017
| Date | Event | Description |
|---|---|---|
| 17 January 2018 | Amended | Feedback added. |
| 14 June 2017 | Feedback has been incorporated | Two pieces of feedback added to the review. Cochrane Airways awaits a full response from the author team. |
Acknowledgements
We would like to thank Ms Emma Barber from the National Center for Child Health and Development for editorial assistance. The authors and Cochrane Airways’ Editorial Team are grateful to the following peer and consumer reviewers for their time and comments: Brian Duncan (USA); Luis J Nannini MD, Hospital de G Baigorria, Universidad Nacional Rosario (Argentina); and Hilary Pinnock, Professor of Primary Care Respiratory Medicine, University of Edinburgh (UK).
We also would like to thank Mrs Elizabeth Stovold from the Cochrane Airways Group for constructing the search formula.
The Backgroundand Methods sections of this review are based on a standard template used by Cochrane Airways.
The views and opinions expressed are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS, or the Department of Health and Social Care.
Appendices
Appendix 1. Sources and search methods for the Cochrane Airways Group Specialised Register (CAGR)
Electronic searches: core databases
| Database | Frequency of search |
| MEDLINE (Ovid) | Weekly |
| Embase (Ovid) | Weekly |
| CENTRAL | Monthly |
| PsycINFO (Ovid) | Monthly |
| CINAHL (EBSCO) | Monthly |
| AMED (EBSCO) | Monthly |
Handsearches: core respiratory conference abstracts
| Conference | Years searched |
| American Academy of Allergy, Asthma and Immunology (AAAAI) | 2001 onwards |
| American Thoracic Society (ATS) | 2001 onwards |
| Asia Pacific Society of Respirology (APSR) | 2004 onwards |
| British Thoracic Society Winter Meeting (BTS) | 2000 onwards |
| Chest Meeting | 2003 onwards |
| European Respiratory Society (ERS) | 1992, 1994, 2000 onwards |
| International Primary Care Respiratory Group Congress (IPCRG) | 2002 onwards |
| Thoracic Society of Australia and New Zealand (TSANZ) | 1999 onwards |
Condition search
1. Lung Diseases, Obstructive/
2. exp Pulmonary Disease, Chronic Obstructive/
3. emphysema$.mp.
4. (chronic$ adj3 bronchiti$).mp.
5. (obstruct$ adj3 (pulmonary or lung$ or airway$ or airflow$ or bronch$ or respirat$)).mp.
6. COPD.mp.
7. COAD.mp.
8. COBD.mp.
9. AECB.mp.
10. or/1‐9
Filter to identify randomised controlled trials
1. exp "clinical trial [publication type]"/
2. (randomised or randomised).ab,ti.
3. placebo.ab,ti.
4. dt.fs.
5. randomly.ab,ti.
6. trial.ab,ti.
7. groups.ab,ti.
8. or/1‐7
9. Animals/
10. Humans/
11. 9 not (9 and 10)
12. 8 not 11
The MEDLINE strategy and RCT filter are adapted to identify trials in other electronic databases
Appendix 2. Search strategy to identify relevant trials from the Cochrane Airways Group Specialised Register (CAGR)
#1 MeSH DESCRIPTOR Pulmonary Disease, Chronic Obstructive Explode All
#2 MeSH DESCRIPTOR Bronchitis, Chronic
#3 (obstruct*) near3 (pulmonary or lung* or airway* or airflow* or bronch* or respirat*)
#4 COPD:MISC1
#5 (COPD OR COAD OR COBD OR AECOPD):TI,AB,KW
#6 #1 OR #2 OR #3 OR #4 OR #5
#7 MeSH DESCRIPTOR Muscarinic Antagonists
#8 muscarinic* NEXT antagonist*
#9 LAMA:ti,ab
#10 tiotropium*
#11 Spiriva
#12 glycopyrronium*
#13 NVA237
#14 Seebri
#15 umeclidinium*
#16 GSK573719
#17 Incruse
#18 aclidinium*
#19 LAS34273
#20 Turdorza
#21 Eklira
#22 #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20 or #21
#23 MeSH DESCRIPTOR Adrenergic beta‐2 Receptor Agonists
#24 long* NEAR beta* NEAR agonist*
#25 salmeterol*
#26 *formoterol*
#27 indacaterol*
#28 QAB149
#29 vilanterol*
#30 GW642444
#31 olodaterol*
#32 "BI 1744 CL"
#33 #23 or #24 or #25 or #26 or #27 or #28 or #29 or #30 or #31 or #32
#34 MeSH DESCRIPTOR Adrenal Cortex Hormones Explode All
#35 inhal* NEAR (corticosteroid* or steroid* or glucocorticoid*)
#36 fluticasone*
#37 budesonide*
#38 beclomethasone*
#39 ciclesonide*
#40 flunisolide*
#41 mometasone*
#42 triamcinolone*
#43 #34 or #35 or #36 or #37 or #38 or #39 or #40 or #41 or #42
#44 #6 AND #22 AND #33 AND #43
#45 Ultibro
#46 QVA149
#47 Stiolto
#48 Anoro
#49 #45 or #46 or #47 or #48
#50 Symbicort
#51 Viani
#52 Seretide
#53 Advair
#54 Atmadisc
#55 Adoair
#56 Foster or Fostair
#57 Inuvair
#58 Dulera
#59 Flutiform
#60 Breo
#61 #50 or #51 or #52 or #53 or #54 or #55 or #56 or #57 or #58 or #59 or #60
#62 #49 AND #61
#63 #49 AND #43
#64 #61 AND #22
#65 (#62 or #63 or #64) AND #6
#66 #44 or #65
[Note: in search line #4, MISC1 denotes the field in the record where the reference has been coded for condition, in this case, COPD]
Appendix 3. Search strategy to identify relevant trials from the ClinicalTrials.gov
Search term: (COPD OR chronic obstructive lung disease) AND (glycopyrrolate OR umeclidinium OR tiotropium OR aclidinium OR olodaterol OR QVA149) AND (indacaterol OR formoterol OR vilanterol OR salmeterol OR QVA149) AND (fluticasone OR budesonide) Study type: Intervention
Appendix 4. Search strategy to identify relevant trials from the World Health Organization search portal
Title search: (COPD OR chronic obstructive lung disease) AND (glycopyrrolate OR umeclidinium OR tiotropium OR aclidinium OR olodaterol OR QVA149) AND (indacaterol OR formoterol OR vilanterol OR salmeterol OR QVA149) AND (fluticasone OR budesonide)
Data and analyses
Comparison 1. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Exacerbations | 13 | 20960 | Odds Ratio (M‐H, Random, 95% CI) | 0.91 [0.78, 1.06] |
| 1.1.1 Indacaterol/glycopyrronium | 5 | 6200 | Odds Ratio (M‐H, Random, 95% CI) | 0.72 [0.63, 0.83] |
| 1.1.2 Umeclidinium/vilanterol | 4 | 8323 | Odds Ratio (M‐H, Random, 95% CI) | 1.20 [1.03, 1.40] |
| 1.1.3 Other LAMA/LABA inhalers | 4 | 6437 | Odds Ratio (M‐H, Random, 95% CI) | 0.97 [0.87, 1.08] |
| 1.2 Serious adverse events | 18 | 23158 | Odds Ratio (M‐H, Random, 95% CI) | 1.02 [0.91, 1.15] |
| 1.2.1 Indacaterol/glycopyrronium | 5 | 6204 | Odds Ratio (M‐H, Random, 95% CI) | 0.88 [0.76, 1.03] |
| 1.2.2 Umeclidinium/vilanterol | 4 | 8323 | Odds Ratio (M‐H, Random, 95% CI) | 1.07 [0.73, 1.55] |
| 1.2.3 Other LAMA/LABA inhalers | 9 | 8631 | Odds Ratio (M‐H, Random, 95% CI) | 1.04 [0.92, 1.19] |
| 1.3 Quality of life as measured by the St George's Respiratory Questionnaire (SGRQ) total score change from baseline | 9 | 14437 | Mean Difference (IV, Random, 95% CI) | ‐0.57 [‐1.36, 0.21] |
| 1.3.1 Indacaterol/glycopyrronium | 2 | 3693 | Mean Difference (IV, Random, 95% CI) | ‐1.29 [‐2.08, ‐0.50] |
| 1.3.2 Umeclidinium/vilanterol | 4 | 6615 | Mean Difference (IV, Random, 95% CI) | ‐0.00 [‐0.02, 0.02] |
| 1.3.3 Other LAMA/LABA inhalers | 3 | 4129 | Mean Difference (IV, Random, 95% CI) | ‐1.14 [‐4.06, 1.78] |
| 1.4 Trough FEV1 mean change (litres) | 12 | 14681 | Mean Difference (IV, Random, 95% CI) | 0.07 [0.05, 0.08] |
| 1.4.1 Indacaterol/glycopyrronium | 4 | 5843 | Mean Difference (IV, Random, 95% CI) | 0.07 [0.05, 0.09] |
| 1.4.2 Umeclidinium/vilanterol | 4 | 6669 | Mean Difference (IV, Random, 95% CI) | 0.08 [0.04, 0.11] |
| 1.4.3 Other LAMA/LABA inhalers | 4 | 2169 | Mean Difference (IV, Random, 95% CI) | 0.07 [0.02, 0.11] |
| 1.5 Pneumonia | 14 | 21829 | Odds Ratio (M‐H, Random, 95% CI) | 0.61 [0.52, 0.72] |
| 1.5.1 Indacaterol/glycopyrronium | 5 | 6204 | Odds Ratio (M‐H, Random, 95% CI) | 0.61 [0.44, 0.85] |
| 1.5.2 Umeclidinium/vilanterol | 4 | 8323 | Odds Ratio (M‐H, Random, 95% CI) | 0.63 [0.50, 0.80] |
| 1.5.3 Other LAMA/LABA inhalers | 5 | 7302 | Odds Ratio (M‐H, Random, 95% CI) | 0.58 [0.43, 0.78] |
| 1.6 All‐cause death | 15 | 21510 | Odds Ratio (M‐H, Random, 95% CI) | 1.35 [1.05, 1.75] |
| 1.6.1 Indacaterol/glycopyrronium | 5 | 6204 | Odds Ratio (M‐H, Random, 95% CI) | 1.02 [0.59, 1.76] |
| 1.6.2 Umeclidinium/vilanterol | 4 | 8324 | Odds Ratio (M‐H, Random, 95% CI) | 1.51 [1.00, 2.26] |
| 1.6.3 Other LAMA/LABA inhalers | 6 | 6982 | Odds Ratio (M‐H, Random, 95% CI) | 1.43 [0.94, 2.17] |
| 1.7 SGRQ improvement ≥ 4 points | 4 | 13614 | Odds Ratio (M‐H, Random, 95% CI) | 1.06 [0.90, 1.25] |
| 1.7.1 Indacaterol/glycopyrronium | 2 | 3192 | Odds Ratio (M‐H, Random, 95% CI) | 1.25 [1.09, 1.44] |
| 1.7.2 Umeclidinium/vilanterol | 1 | 6204 | Odds Ratio (M‐H, Random, 95% CI) | 1.00 [0.89, 1.12] |
| 1.7.3 Other LAMA/LABA inhalers | 1 | 4218 | Odds Ratio (M‐H, Random, 95% CI) | 0.89 [0.79, 1.01] |
| 1.8 Exacerbations (fixed‐effect sensitivity) | 13 | 20960 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.94 [0.87, 1.01] |
| 1.8.1 Indacaterol/glycopyrronium | 5 | 6200 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.72 [0.63, 0.83] |
| 1.8.2 Umeclidinium/vilanterol | 4 | 8323 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.20 [1.03, 1.40] |
| 1.8.3 Other LAMA/LABA inhalers | 4 | 6437 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.87, 1.08] |
| 1.9 Serious adverse events (fixed‐effect sensitivity) | 18 | 23158 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.03 [0.95, 1.12] |
| 1.9.1 Indacaterol/glycopyrronium | 5 | 6204 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.75, 1.03] |
| 1.9.2 Umeclidinium/vilanterol | 4 | 8323 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.99, 1.27] |
| 1.9.3 Other LAMA/LABA inhalers | 9 | 8631 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.05 [0.92, 1.20] |
| 1.10 SGRQ mean change (fixed‐effect sensitivity) | 9 | 14437 | Mean Difference (IV, Fixed, 95% CI) | ‐0.00 [‐0.02, 0.02] |
| 1.10.1 Indacaterol/glycopyrronium | 2 | 3693 | Mean Difference (IV, Fixed, 95% CI) | ‐1.29 [‐2.08, ‐0.50] |
| 1.10.2 Umeclidinium/vilanterol | 4 | 6615 | Mean Difference (IV, Fixed, 95% CI) | ‐0.00 [‐0.02, 0.02] |
| 1.10.3 Other LAMA/LABA inhalers | 3 | 4129 | Mean Difference (IV, Fixed, 95% CI) | ‐0.23 [‐1.01, 0.54] |
| 1.11 Trough FEV1 mean change (fixed‐effect sensitivity) | 12 | 14681 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [0.05, 0.07] |
| 1.11.1 Indacaterol/glycopyrronium | 4 | 5843 | Mean Difference (IV, Fixed, 95% CI) | 0.07 [0.05, 0.08] |
| 1.11.2 Umeclidinium/vilanterol | 4 | 6669 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [0.05, 0.07] |
| 1.11.3 Other LAMA/LABA inhalers | 4 | 2169 | Mean Difference (IV, Fixed, 95% CI) | 0.06 [0.04, 0.07] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Beeh 2016.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, cross‐over, double‐dummy, placebo‐controlled 4‐period 4‐arm trial Countries: 8 countries (mainly EU countries) Site: 29 centres Study duration: 6 weeks × 4 time periods Study start: October 2013 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 80%, without recent exacerbation Numbers of randomised and consequent‐treatment completed cases: 229 and 202 Age: 63.6 (SD 7.6) years Male/female: 148/81 %pred FEV1: unclear |
|
| Interventions | LAMA/LABA: tiotropium/olodaterol (2.5/5 μg) or tiotropium/olodaterol (5/5 μg) LABA/ICS: salmeterol/fluticasone (50/250 μg) twice daily or salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: change from baseline FEV1 AUC (0‐12 h) after 6 weeks of treatment Tiotropium/olodaterol (2.5/5 μg): 0.295 (SE 0.014) Tiotropium/olodaterol (5/5 μg): 0.317 (SE 0.014) Salmeterol/fluticasone (50/250 μg): 0.192 (SE 0.015) Salmeterol/fluticasone (50/500 μg): 0.188 (SE 0.014) |
|
| Notes | Registration: NCT01969721 COI: sponsored by Boehringer Ingelheim |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 202/229 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 6.9% with T+O 2.5/5 > T+O 5/5 > F+S 250/50 > F+S 500/50, 17.4% with T+O 5/5 > F+S 500/50 > T+O 2.5/5 > F+S 250/50, 10.0% with F+S 250/50 > T+O 2.5/5 > F+S 500/50 > T+O 5/5, and 11.5% with F+S 500/50 > F+S 250/50 > T+O 5/5 > T+O 2.5/5 Note: this was a four‐arm cross‐over study. Each arm used four consecutive treatments. For example, participants in the first arm were treated by T+O 2.5/5, then treated by T+O 5/5, then treated by F+S 250/50, then F+S 500/50. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described |
| Other bias | High risk | Study funding statement indicates that one or more authors were employees of the pharmaceutical sponsor (Boehringer Ingelheim) and were involved in the design, conduct and data analysis of the study. |
Donohue 2015a.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 7 countries (USA and European countries) Site: 63 centres Study duration: 12 weeks Study start: March 2013 Run‐in period: 7 to 14 days |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 70%, mMRC ≥ 2, no recent exacerbation Numbers of randomised and completed cases: 707 and 634 Age: 62.8 (SD 9.0) years Male/female: 497/213 %pred FEV1: 49.4% (SD 10.9) |
|
| Interventions | LAMA/LABA: umeclidinium/vilanterol (62.5/25 μg) LABA/ICS: salmeterol/fluticasone (50/250 μg) twice daily |
|
| Outcomes | Primary endpoint: change from baseline in 24‐h weighted‐mean serial FEV1 on day 84 Umeclidinium/vilanterol (62.5/25 μg): 0.165 (SE 0.0130) Salmeterol/fluticasone (50/250 μg) twice daily: 0.091 (SE 0.0131) |
|
| Notes | Registration: NCT01817764, GSK‐DB2114930 COI: sponsored by GlaxoSmithKline |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Central randomisation schedule was generated using a validated computer system (RanAll, GSK) |
| Allocation concealment (selection bias) | Low risk | Centralised randomisation prevented foreknowledge of intervention assignments by both researchers and participants |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 634/707 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 9.6% in umeclidinium/vilanterol arm and 10.8% in salmeterol/fluticasone arms. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Study funding statement indicates that all but one of the study authors were employees of and held stock in the pharmaceutical sponsor (GlaxoSmithKline) and were involved in the design, conduct and data analysis of the study. |
Donohue 2015b.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 7 countries (USA, European countries, and Russia) Site: 71 centres Study duration: 12 weeks Study start: June 2013 Run‐in period: 7 to 14 days |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 70%, mMRC ≥ 2, no recent exacerbation Numbers of randomised and completed cases: 700 and 638 Age: 63.6 (SD 8.9) years Male/female: 528/169 %pred FEV1: 49.5% (SD 10.9) |
|
| Interventions | LAMA/LABA: umeclidinium/vilanterol (62.5/25 μg) LABA/ICS: salmeterol/fluticasone (50/250 μg) twice daily |
|
| Outcomes | Primary endpoint: change from baseline in 24‐h weighted‐mean serial FEV1 on treatment day 84 Umeclidinium/vilanterol (62.5/25 μg): 0.213 (SE 0.0137) Salmeterol/fluticasone (50/250 μg) twice daily: 0.112 (SE 0.0139) |
|
| Notes | Registration: NCT01879410, GSK‐DB2114951 COI: sponsored by GlaxoSmithKline |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Central randomisation schedule was generated using a validated computer system (RanAll, GSK) |
| Allocation concealment (selection bias) | Low risk | Centralised randomisation prevented foreknowledge of intervention assignments by both researchers and participants. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 638/700 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 6.9% in umeclidinium/vilanterol arm and 10.9% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Study funding statement indicates that all but one of the study authors were employees of and held stock in the pharmaceutical sponsor (GlaxoSmithKline) and were involved in the design, conduct and data analysis of the study. |
Ferguson 2018.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 4 countries (Canada, China, Japan, USA) Site: 215 centres Study duration: 24 weeks Study start: August 2015 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: CAT ≥ 10, %pred FEV1 25% to 80%, not required to have exacerbation Numbers of randomised and consequent‐treatment completed cases: 943 and 790 Age: 65.1 (SD 7.4) years Male/female: 654/285 %pred FEV1: unclear |
|
| Interventions | LAMA/LABA: glycopyrronium/formoterol fumarate (18/9.6 μg) once daily LABA/ICS: formoterol fumarate/budesonide (4.8/160 μg) twice daily | |
| Outcomes | Primary outcome: change from baseline in morning pre‐dose FEV1 over 24 weeks LAMA/LABA: glycopyrronium/formoterol fumarate (18/9.6 μg): 125 mL (SD 6.6) LABA/ICS: formoterol fumarate/budesonide (9.6/320 μg): 601 mL (SD 9.2) |
|
| Notes | Registration: NCT02497001 COI: sponsored by AstraZeneca Notes: two of the references identified as relating to Ferguson 2018 (JPRN‐JapicCTI‐184080 and NCT03262012) are registry records for a 28‐week extension in the subset of participants recruited from Japanese centres (22% of total). We did not include extension data within the review because this would have caused double counting within the analysis. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | High risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 790/943 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 16.5% in glycopyrronium + formoterol fumarate arm and 15.9% in budesonide + formoterol fumarate arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Seven study authors who were involved in design, data management and interpretation were employees of AstraZeneca or its subsidiaries. |
Frith 2018.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, double‐dummy, active control, parallel group study Countries: 11 countries (mainly EU countries) Site: 60 centres Study duration: 12 weeks Study start: October 2015 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: CAT ≥ 10, %pred FEV1 30% to 80%, moderate to severe COPD and up to one exacerbation in the previous year Numbers of randomised and consequent‐treatment completed cases: 502 and 473 Age: 65.1 (SD 8.8) years Male/female: 444/54 %pred FEV1: 51.5 (SD 12.7) |
|
| Interventions | LAMA/LABA: glycopyrronium/indacaterol (50/110 μg) once daily LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: pre‐dose trough FEV1 at week 12 Mean difference: 0.045 L (95% CI 0.005 to 0.084) (P = 0.028) |
|
| Notes | Registration: NCT02516592 COI: sponsored by Novartis Pharmaceuticals |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | All eligible participants were randomised via interactive response technology. |
| Allocation concealment (selection bias) | Low risk | All eligible participants were randomised via interactive response technology. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 473/502 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 6.4% in glycopyrronium/indacaterol arm and 5.2% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Most or all authors received fees from the sponsor, Novartis Pharmaceuticals, during the conduct of the study. |
Herth 2020.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, cross‐over, double‐dummy trial Country: Germany Site: 10 centres Study duration: 6 weeks Study start: unclear Run‐in period: unclear |
|
| Participants | Key inclusion criteria: %pred FEV1 < 70%, without recent exacerbation Numbers of randomised and consequent‐treatment completed cases: 76 and 67 Age: 61.9 (SD 7.1) years Male/female: 45/31 %pred FEV1: 52.9 (SD12.1) |
|
| Interventions | LAMA/LABA: tiotropium/olodaterol (5/5 μg) once daily LABA/ICS: salmeterol/fluticasone propionate (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: change from baseline in left ventricular end‐diastolic volume index after 6 weeks of treatment Mean difference: ‐0.537 mL/m2 (95% CI ‐2.779 to 1.705) (P = 0.6331) | |
| Notes | Registration: NCT03055988 COI: sponsored by Boehringer Ingelheim |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | High risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 67/76 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Study funding statement indicates that one or more authors were employees of the pharmaceutical sponsor (Boehringer Ingelheim) and were involved in the design, conduct, and data analysis of the study. |
Hoshino 2015.
| Study characteristics | ||
| Methods | Design: randomised, parallel‐group, open‐label trial Countries: 1 country (Japan) Site: 1 centre Study duration: 16 weeks Study start: unclear Run‐in period: 21 days |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 80%, without recent exacerbation Numbers of randomised and completed cases: 46 and 43 Age: LAMA/LABA, 72 years (SD 7); LABA/ICS, 69 years (SD 6) Male/female: 36/7 %pred FEV1: LAMA/LABA, 61.9% (SD 16.3%); LABA/ICS, 60.8% (SD 16.4%) |
|
| Interventions | LAMA/LABA: tiotropium/indacaterol (18/150 μg) LABA/ICS: salmeterol/fluticasone (50/250 μg) twice daily |
|
| Outcomes | Primary outcomes: effects on airway dimensions | |
| Notes | Registration: none COI: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Study had an open‐label design. Knowledge of the randomised treatment could have affected participant or personnel behaviour that biased the results for subjective and semi‐objective outcomes. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Study had an open‐label design. Knowledge of the randomised treatment could have affected outcome assessor judgement for the subjective and semi‐objective outcomes. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 43/46 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 8.3% in tiotropium/indacaterol arm and 4.5% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | High risk | We could not rule out reporting bias because this trial was not registered. |
| Other bias | Low risk | We found no risk of other bias. |
Lipson 2018.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group trial Countries: 37 countries (mainly EU countries) Site: 29 centres Study duration: 52 weeks Study start: June 2014 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: CAT ≥ 10, %pred FEV1 that was less than 50% and at least one moderate or severe exacerbation in the previous year. %pred FEV1 50% to 80% and at least two moderate exacerbations or one severe exacerbation in the previous year. Numbers of randomised and consequent‐treatment completed cases: 6204 and 5373 Age: 65.3 (SD 8.3) years Male/female: 4104/2100 %pred FEV1: 45.5 (SD 14.7) |
|
| Interventions | LAMA/LABA: umeclidinium/vilanterol (62.5/25 μg) once daily LABA/ICS: vilanterol/fluticasone furoate (25/100 μg) once daily |
|
| Outcomes | Primary outcome: annual rate of moderate or severe exacerbations during treatment Umeclidinium/vilanterol: 1.21 Vilanterol/fluticasone furoate: 1.07 |
|
| Notes | Registration: NCT02164513 COI: sponsored by GlaxoSmith Kline |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation. |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 5373/6204 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 87.0% in umeclidinium/vilanterol arm and 85.7% in vilanterol/fluticasone furoate arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Authors declared a variety of conflicts of interest associated with the sponsor, GlaxoSmithKline, and employees were involved in the design and conduct of the study. |
Magnussen 2012.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, cross‐over, double‐dummy, placebo‐controlled trial Countries: 7 countries Site: 40 centres Study duration: 8 weeks Study start: September 2007 Run‐in period: 15 days |
|
| Participants | Key inclusion criteria: %pred FEV1 ≤ 65%, without recent exacerbation Numbers of randomised and completed cases: 344 and 300 Age: 61.0 years (SD 7.6) Male/female: 247/97 %pred FEV1: 47% (SD 12%) |
|
| Interventions | LAMA/LABA: tiotropium/salmeterol (18/50 μg) twice daily LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Co‐primary endpoint 1: post‐dose thoracic gas volume (functional residual capacity) (after 8 weeks) Mean difference ‐0.087 (SE 0.044). Co‐primary endpoint 2: endurance time (after 8 weeks) Mean difference 3.0 (95% CI ‐9.5 to 27.5) |
|
| Notes | Registration: NCT00530842 COI: sponsored by Boehringer Ingelheim and Pfizer |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Centralised randomisations of 4 blocks stratified according to sites. |
| Allocation concealment (selection bias) | Low risk | Centralised randomisations of 4 blocks stratified according to sites. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 300/344 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 13.4% in tiotropium/salmeterol > salmeterol/fluticasone arm and 12.2% in salmeterol/fluticasone > tiotropium/salmeterol arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. We found no risk of reporting bias. |
| Other bias | High risk | Contributing authors included those employed by the sponsors, Boehringer Ingelheim and Pfizer, and those who received grants, honoraria, and consultancy fees from the study sponsors. |
Mostafa 2021.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, prospective parallel study Countries: 1 country (Egypt) Site: 1 centre Study duration: 12 weeks Study start: November 2016 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 80%, without recent exacerbation Numbers of randomised and consequent‐treatment completed cases: 40 and 30 Age: 64.2 (SD 9.2) years Male/female: 21/9 %pred FEV1: 60.9 (SD 15.1) |
|
| Interventions | LAMA/LABA: tiotropium (18 μg) once daily + formoterol (9 μg) twice daily LABA/ICS: formoterol/budesonide (160/4.5 μg) twice daily |
|
| Outcomes | Primary outcome: changes that occurred in FEV1 as a percent of predicted after 12‐week treatment Tiotropium/formoterol: 77.61 (SD 21.34) mL Formoterol/budesonide: 73.83 (SD 15.97) mL |
|
| Notes | Registration: NCT04520230 | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Low risk | The independent researcher kept the original random allocation sequences in an inaccessible place. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 30/40 participants completed the study. Considerable attrition bias was suspected because attrition was > 20%. |
| Selective reporting (reporting bias) | High risk | There was no protocol. |
| Other bias | Low risk | No conflicts of interest or other sources of bias identified |
NCT03240575.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy trial Countries: 1 country (USA) Site: 45 centres Study duration: 12 weeks Study start: August 2017 Run‐in period: 4 weeks during which LABAs, LAMAs and ICSs were withdrawn prior to randomisation |
|
| Participants | Key inclusion criteria: outpatients of either sex; aged ≥ 40 years with a diagnosis of COPD; smoking history > 10 pack years; post‐bronchodilator 30% ≤ FEV1 predicted < 80%; postbronchodilator FEV1/FVC < 70%. Numbers of randomised and completed cases: 302 randomised; 291 in treated set (all who received at least one dose); 275 completed Age: LAMA/LABA 63.95 years (SD 8.32); LABA/ICS 64.74 years (SD 7.33) Male/female: LAMA/LABA 74/72; LABA/ICS 83/62 (54.0% male) %pred FEV1: not reported |
|
| Interventions | LAMA/LABA: tiotropium/olodaterol (5 μg/5 μg) once daily LABA/ICS: fluticasone Propionate/salmeterol (250 μg/50 μg) twice daily |
|
| Outcomes | Primary endpoint: FEV1 area under the curve from 0 to 24 hours (not relevant to review). Reports trough FEV1 change from baseline to 12 weeks, all‐cause mortality and serious adverse events (included pneumonia) | |
| Notes | Registration: NCT03240575; ENERGITO2 trial COI: sponsored by Boehringer Ingelheim |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | "Boehringer Ingelheim will generate the randomization scheme using validated randomization software, and prepare and code the medication in a blinded fashion" |
| Allocation concealment (selection bias) | Low risk | "An interactive response technology (IRT) system will be used for randomisation to treatment in this trial and for allocation of medication to patients throughout the treatment period." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | "Patients, investigators and everyone involved in analyzing or with an interest in this double‐blind study will remain blinded with regard to the randomized treatment assignments until after database lock." "Boehringer Ingelheim will...prepare and code the medication in a blinded fashion" |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | "Patients, investigators and everyone involved in analyzing or with an interest in this double‐blind study will remain blinded with regard to the randomized treatment assignments until after database lock." "Boehringer Ingelheim will...prepare and code the medication in a blinded fashion" |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 302 enrolled; 291 in treated set (at least one dose), 146 in the LAMA/LABA group and 145 in the LABA/ICS group. 2 participants in the LAMA/LABA group did not complete the study (1.4%) compared with 14 in the LABA/ICS group (9.7%), primarily due to participant withdrawal (n = 11). Analyses were conducted on the full analysis set, i.e. everyone who had baseline and at least one post‐baseline measurement for at least one efficacy parameter. The mixed‐effects repeated measurement (MMRM) model defined the participants who contributed to the model. The numbers and reasons for participant dropout varies between groups but is low and an appropriate method for imputing data for missing measurements was used and is unlikely to introduce bias. |
| Selective reporting (reporting bias) | Low risk | Outcomes in line with statistical analysis plan and protocol available on clinicaltrials.gov |
| Other bias | High risk | The trial sponsor was responsible for coordinating the trial, directing clinical trial personnel in conduct and reporting, training, monitoring, data management and statistical evaluation. |
Rabe 2008.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 8 countries, mainly EU Site: multicentre Study duration: 6 weeks Study start: November 2003 Run‐in period: 2 to 4 weeks |
|
| Participants | Key inclusion criteria: %pred FEV1 ≤ 65% without recent exacerbation Numbers of randomised and completed cases: 605 and unclear Age: 62 years (SD 9) Male/female: 414/191 %pred FEV1: 55% (SD 13%) |
|
| Interventions | LAMA/LABA: tiotropium/formoterol (18 μg/24 μg) twice daily LABA/ICS: salmeterol/fluticasone propionate (50 μg/500 μg) twice daily |
|
| Outcomes | Co‐primary endpoint 1: FEV1 AUC (0 to 12 h) after 6 weeks of treatment Mean difference 78 mL (95% CI 34 to 122) higher in tiotropium/formoterol arm Co‐primary endpoint 2: peak FEV1 measured after 6 weeks of treatment Mean difference 103 mL (95% CI 55 to 150) higher in tiotropium/formoterol arm |
|
| Notes | Registration: NCT00239421 COI: sponsored by Boehringer and Pfizer |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 592/605 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 2.3% in tiotropium/formoterol arms and 2.0% in salmeterol/fluticasone propionate arms. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. We found no risk of reporting bias. |
| Other bias | High risk | Contributing authors included those employed by the sponsors, Boehringer Ingelheim and Pfizer, and those who received grants, honoraria or consultancy fees from the study sponsors. |
Rabe 2020.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group trial Countries: 26 countries Study duration: 52 weeks Study start: June 2015 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: %pred FEV1 25% to 65%, CAT ≥ 10, at least one moderate or severe exacerbation in the year (%pred FEV1 < 50%), at least two moderate or at least one severe exacerbation in the year (%pred FEV1 ≥ 50%) Numbers of randomised and completed cases: 4294 and 3225 Age: 64.7 years (SD 7.6) Male/female: 2523/1728 %pred FEV1: 43.4 (SD 10.3) |
|
| Interventions | LAMA/LABA: glycopyrronium/formoterol (9/4.8 µg) twice daily LABA/ICS: formoterol/budesonide (4.8/160 µg) twice daily |
|
| Outcomes | Primary outcome: annual rate of moderate or severe COPD exacerbation Glycopyrronium/formoterol (9/4.8 µg): 1.42 Formoterol/budesonide (4.8/160 µg): 1.24 |
|
| Notes | Registration: NCT02465567 COI: sponsored by AstraZeneca |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation. |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 3225/4294 participants completed the study. Considerable attrition bias was suspected because attrition was > 20%. Attrition was 26.2% in glycopyrronium/formoterol arm and 23.6% in formoterol/budesonide arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Conflict of interest statements available on the New England Journal of Medicine website indicate most contributing authors were either employees of the sponsor, AstraZeneca, or received fees from the company outside the submitted work. |
Singh 2015.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 8 countries (mainly EU) Site: 79 centres Study duration: 12 weeks Study start: April 2013 Run‐in period: 7 to 14 days |
|
| Participants | Key inclusion criteria: %pred FEV1 30% to 70%, mMRC ≥ 2, without recent exacerbation Numbers of randomised and completed cases: 717 and 674 Age: 61.6 years (SD 8.0) Male/female: 515/201 %pred FEV1: 50.6% (SD 10.7%) |
|
| Interventions | LAMA/LABA: umeclidinium/vilanterol (62.5/25 μg) LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: change from baseline in 0 to 24 h weighted mean serial FEV1 at day 84 Mean difference 0.080 L (95% CI 0.046 to 0.113) (P < 0.001) |
|
| Notes | Registration: NCT01822899, GSK‐DB2116134 COI: sponsored by GlaxoSmithKline |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Validated computer system (RandAll) was used to generate central randomisation |
| Allocation concealment (selection bias) | Low risk | Validated computer system (RandAll) was used to generate central randomisation |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 674/717 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 6.7% in umeclidinium/vilanterol arm and 5.0% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. We found no risk of reporting bias. |
| Other bias | High risk | All but one author were employees and shareholders of the sponsor, GlaxoSmithKline, and the lead author received grants and personal payments from GSK. |
Vogelmeier 2013.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 10 countries (mainly EU) Site: 92 centres Study duration: 26 weeks Study start: March 2011 Run‐in period: 14 days |
|
| Participants | Key inclusion criteria: post‐bronchodilator 30% ≤ FEV1 predicted < 80%, without recent exacerbation Numbers of randomised and completed cases: 523 and 432 Age: LAMA/LABA, 63.2 years (SD 8.2); LABA/ICS, 63.4 years (SD 7.7) Male/female: LAMA/LABA, 181/77; LABA/ICS, 189/75 %pred FEV1: LAMA/LABA, 60.5% (SD 10.5%); LABA/ICS, 60.0% (SD 10.7%) |
|
| Interventions | LAMA/LABA: indacaterol/glycopyrronium (110/50 μg) LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: FEV1 AUC (0 to 12 h) LAMA/LABA: 1.69 (SE 0.027) LABA/ICS: 1.56 (SE 0.026) |
|
| Notes | Registration: NCT01315249 COI: sponsored by Novartis Study name: ILLUMINATE |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Investigators used an automated, interactive response technology to assign randomisation numbers to participants. |
| Allocation concealment (selection bias) | Low risk | Investigators used an automated, interactive response technology to assign randomisation numbers to participants. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 432/523 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 17.0% in indacaterol/glycopyrronium arm and 17.0% in salmeterol/fluticasone arms. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Five of the seven listed authors are employees of the sponsor, Novartis. |
Vogelmeier 2016.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 14 countries (mainly EU) Site: 126 centres Study duration: 24 weeks Study start: September 2013 Run‐in period: unclear |
|
| Participants | Key inclusion criteria: %pred FEV1 < 80%, CAT ≥ 10, without recent exacerbation Numbers of randomised and completed cases: 933 and 788 Age: 63.4 years (SD 7.8) Male/female: 607/326 |
|
| Interventions | LAMA/LABA: aclidinium/formoterol (400/12 μg) twice daily LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: peak FEV1 at week 24 LAMA/LABA: 1.655 (SE 0.011) LABA/ICS: 1.562 (SE 0.011) |
|
| Notes | Registration: NCT01908140, EudraCT 2013‐000116‐14 COI: sponsored by AstraZeneca |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 788/933 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 14.1% in aclidinium/formoterol arm and 17.0% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. We found no risk of reporting bias. |
| Other bias | High risk | Author disclosure statements could not be viewed on the publisher's website, but affiliations show that three authors involved in the design, conduct, or interpretation of the study were employed by the sponsors, Almirall or AstraZeneca. Writing support was provided by an AstraZeneca‐funded company. |
Vogelmeier 2017.
| Study characteristics | ||
| Methods | Design: randomised, open‐label, pragmatic study Countries: 23 countries (mainly Europe) Sites: 560 Study duration: 12 weeks Study start: June 2014 Run‐in period: April 2016 |
|
| Participants | Key inclusion criteria: %pred FEV1 50% to 80%, mMRC ≥ 1, without ≥ 2 exacerbation in the previous 12 months Numbers of randomised and completed cases: 1083 and 899 Age: 64.6 years (SD 8.85) Male/female: 692/388 %pred FEV1: 63.6% (SD 8.40%) |
|
| Interventions | LAMA/LABA: glycopyrronium/indacaterol (50/110 μg) LABA/ICS: any |
|
| Outcomes | Primary endpoints: trough FEV1 Mean difference: 71 mL (95% CI 36 to 107) higher in glycopyrronium/indacaterol arm |
|
| Notes | COI: sponsored by Novartis Pharmaceuticals Study name: CRYSTAL |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Study described as randomised but no details provided about methods of sequence generation |
| Allocation concealment (selection bias) | Unclear risk | Methods of allocation concealment not described |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Study was open‐label. Knowledge of the randomised treatment could have affected participant or personnel behaviour that biased the results for subjective and semi‐objective outcomes. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Study was open‐label. Knowledge of the randomised treatment could have affected outcome assessor judgement for the subjective and semi‐objective outcomes. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 899/1083 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 18.3% in glycopyrronium/indacaterol arm and 13.0% in LABA/ICS arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. We found no risk of reporting bias. |
| Other bias | High risk | Most authors were either employed by the sponsor, Novartis Pharmaceuticals, held shares, or received fees for the design and conduct of the study (but not for development of the manuscript). |
Wedzicha 2016.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 43 countries Site: 496 centres Study duration: 52 weeks Study start: July 2013 Run‐in period: 4 weeks |
|
| Participants | Key inclusion criteria: %pred FEV1 25% to 60%, mMRC ≥ 2, with recent exacerbation Numbers of randomised and completed cases: 3362 and 2760 Age: 64.6 years (SD 7.8) Male/female: 2557/805 %pred FEV1: 44.1% (SD 9.5%) |
|
| Interventions | LAMA/LABA: indacaterol/glycopyrronium (110/50 μg) LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: rate of COPD exacerbations per year LAMA/LABA: 3.59 (95% CI 3.28 to 3.94) LABA/ICS: 4.03 (95% CI 3.68 to 4.41) |
|
| Notes | Registration: NCT01782326 COI: sponsored by Novartis Study name: FLAME |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomised via interactive response technology to 1 of the treatment arms. |
| Allocation concealment (selection bias) | Low risk | Participants were randomised via interactive response technology to 1 of the treatment arms. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 2760/3362 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 16.6% in indacaterol/glycopyrronium arm and 19.0% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Author disclosure statements available on the New England Journal of Medicine website indicate that several authors involved in design, conduct, and interpretation were employees and shareholders of the sponsor, Novartis, and others received personal fees or grants from the sponsor. |
Zhong 2015.
| Study characteristics | ||
| Methods | Design: randomised, double‐blind, parallel‐group, double‐dummy, placebo‐controlled trial Countries: 4 countries (recruited mainly in China) Site: 56 centres Study duration: 26 weeks Study start: November 2012 Run‐in period: 14 days |
|
| Participants | Key inclusion criteria: post‐bronchodilator 30% ≤ FEV1 predicted < 80%, mMRC ≥ 2, without recent exacerbation Numbers of randomised and completed cases: 744 and 676 Age: LAMA/LABA 64.8 years (SD 7.8); LABA/ICS 65.3 years (SD 7.9) Male/female: 672/69 %pred FEV1: LAMA/LABA 51.6% (SD 12.8%), LABA/ICS 52.0% (SD 12.9%) |
|
| Interventions | LAMA/LABA: indacaterol/glycopyrronium (110/50 μg) LABA/ICS: salmeterol/fluticasone (50/500 μg) twice daily |
|
| Outcomes | Primary outcome: trough FEV1 following 26 weeks of treatment to demonstrate the non‐inferiority of indacaterol/glycopyrronium to salmeterol/fluticasone LAMA/LABA: 1.248 L (SE 0.0173) LABA/ICS: 1.176 L (SE 0.0172) |
|
| Notes | Registration: NCT01709903 COI: sponsored by Novartis Study name: LANTERN |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Participants were randomised via interactive response technology to 1 of the treatment arms. |
| Allocation concealment (selection bias) | Low risk | Participants were randomised via interactive response technology to 1 of the treatment arms. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study was double‐blinded. Some participants and/or personnel may have inadvertently detected the randomised treatment (e.g. through side effects), but there was no evidence to suggest this was the case or that it introduced bias in the subjective or semi‐objective outcome results. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Study was double‐blinded. Detection bias was not suspected. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | 676/744 participants completed the study. Considerable attrition bias was not suspected because attrition was < 20%. Attrition was 7.8% in indacaterol/glycopyrronium arm and 10.4% in salmeterol/fluticasone arm. |
| Selective reporting (reporting bias) | Low risk | Study was registered and the prespecified outcomes were appropriately described. |
| Other bias | High risk | Five of the nine listed authors, who were involved in the design, conduct, and interpretation of the study, were employees of the sponsor, Novartis. |
%pred FEV1: % predicted forced expiratory volume in one second; AUC: area under the curve; CAT: chronic obstructive pulmonary disease assessment test; CI: confidence interval; COI: conflicts of interest; COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in one second; FVC: forced vital capacity; h: hour; ICS: inhaled corticosteroid; LABA: long‐acting beta‐agonist; LAMA: long‐acting muscarinic antagonist; mMRC: modified Medical Research Council dyspnoea scale; SD: standard deviation; SE: standard error
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Anzueto 2017 | Not an RCT |
| Aziz 2018 | Systematic review |
| Bruhn 2003 | Not comparing LAMA+LABA versus LABA+ICS |
| Calverley 2007 | Not comparing LAMA+LABA versus LABA+ICS |
| EUCTR2015‐002046‐31‐ES | Results only reported for those with asthma‐COPD overlap syndrome |
| Knobil 2004a | Not comparing LAMA+LABA versus LABA+ICS |
| Knobil 2004b | Not comparing LAMA+LABA versus LABA+ICS |
| Mahler 2016 | Not an RCT |
| Michael 2016 | Commentary on included study (Wedzicha 2016) |
| NCT00120978 | Not comparing LAMA+LABA versus LABA+ICS |
| NCT03376295 | Not an RCT ‐ observational study |
| NCT03504527 | Not comparing LAMA+LABA versus LABA+ICS |
| NCT04138758 | Not an RCT ‐ retrospective study |
| NCT04320342 | Not comparing LAMA+LABA versus LABA+ICS |
| NCT04923347 | Not comparing LAMA+LABA versus LABA+ICS |
| NCT05097014 | Not comparing LAMA+LABA versus LABA+ICS |
| Oba 2016 | Systematic review |
| Papi 2018 | Not comparing LAMA+LABA versus LABA+ICS |
| Pavord 2016 | Systematic review |
| Price 2014 | Cost‐effectiveness analysis using previously published data |
| Sciurba 2004 | Not comparing LAMA+LABA versus LABA+ICS |
| Singh 2019 | Letter |
| Skoupa 2018 | Cost‐effectiveness analysis |
| UMIN000024905 | Only participants with eosinophilic inflammation |
COPD: chronic obstructive pulmonary disease; ICS: inhaled corticosteroids; LABA: long‐acting beta‐agonist; LAMA: long‐acting muscarinic antagonist; RCT: randomised controlled trial
Characteristics of ongoing studies [ordered by study ID]
EUCTR2016‐004473‐41‐GB.
| Study name | INCOGNITO |
| Methods | Design: randomised, open‐label, parallel group trial Countries: United Kingdom Study duration: 6 months |
| Participants | Key inclusion criteria: %pred FEV1 < 80%, without recent exacerbation |
| Interventions | LAMA/LABA: tiotropium/olodaterol (5/5 μg) once daily LABA/ICS: vilanterol/fluticasone furoate (22/92 μg) once daily |
| Outcomes | Primary end point: bacterial load of total respiratory pathogens determined by quantitative polymerase chain reaction |
| Starting date | Study start: March 2017 |
| Contact information | Chalmers 01382 383642 |
| Notes | Registration: EUCTR2016‐004473‐41‐GB COI: sponsored by Boehringer Ingelheim |
%pred FEV1: % predicted forced expiratory volume in one second; CAT: chronic obstructive pulmonary disease assessment test; COI: conflicts of interest; COPD: chronic obstructive pulmonary disease; FEV1: forced expiratory volume in one second; ICS: inhaled corticosteroid; LABA: long‐acting beta‐agonist; LAMA: long‐acting muscarinic antagonist
Differences between protocol and review
The original protocol prespecified that we would assess risk of bias separately for different outcomes where necessary, but did not specify how this would be accomplished. Therefore, we expanded the methods in this version by introducing a system to classify outcomes as subjective and objective to consider different risks of performance and selection bias for different blinding methods. All‐cause death is the only outcome of interest to this review that may be considered truly objective and the SGRQ is the only subjective, self‐reported measure. The other outcomes might be considered semi‐objective (exacerbations, serious adverse events, FEV1, and pneumonia) because they involve a degree of patient reporting, behaviour, or clinician judgement that could introduce bias.
We did not prespecify specific domains or percentages for GRADE assessments of risk of bias, but considered it reasonable to downgrade if studies contributing more than half the analysis weight were at high risk of bias in the 'other bias' domain and at least one other. For inconsistency, we downgraded if I2 was greater than 50%, and for imprecision, we based downgrade decisions on prespecified thresholds (clinical importance 0.7 to 1.5 for outcomes as odds ratios) or established MCIDs (4 points for SGRQ).
Contributions of authors
NF contributed to the conception of the work, data acquisition, data analysis, interpretation, drafting, and critical revision.
NH contributed to the conception of the work, data acquisition, data analysis, interpretation, and critical revision.
AK and AG performed interpretation and critical revision.
TK contributed to the conception of the work, interpretation, and critical revision.
EO contributed to data analysis, interpretation, and critical revision.
KK performed critical revision.
All review authors provided final approval and took accountability.
Contributions of editorial team
Sally Spencer (Co‐ordinating Editor) edited the review; advised on methodology, interpretation and content; approved the review prior to publication.
Ian Yang (Contact Editor): edited the review; advised on methodology, interpretation and content.
Emma Dennett (Deputy Co‐ordinating Editor): checked the data entry; advised on methodology, interpretation and content; edited the review.
Cathryn Broderick (Editor): undertook the MECIR check for the Airways Group.
Emma Jackson (Managing Editor): co‐ordinated the editorial process; conducted peer review; edited the review and reference sections of the review.
Elizabeth Stovold (Information Specialist): designed the search strategy; ran the searches.
Vittoria Lutje (Freelance Information Specialist): ran the updated search.
Sources of support
Internal sources
-
N/A, Other
The authors declare that no internal funding was received for this systematic review.
External sources
-
Cochrane Airways, UK
This project was supported by the National Institute for Health Research (NIHR), via Cochrane Infrastructure funding to the Cochrane Airways Group.
Declarations of interest
NF: none known. NH: personally received a lecture fee from AstraZeneca. AK: none known. AG: none known. TK: acted as an independent contractor, with monies paid to institution, for AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline and Novartis. TK also received grants with monies paid to host institution from Boehringer Ingelheim and Novartis. EO: none known. KK: worked as a freelance editor with Cochrane Airways at the time of writing but was not involved in the editorial process for this review.
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Beeh 2016 {published data only}
- Beeh KM, Derom E, Echave-Sustaeta J, Grönke L, Hamilton A, Zhai D, et al. The lung function profile of once-daily tiotropium and olodaterol via Respimat is superior to that of twice-daily salmeterol and fluticasone propionate via Accuhaler (ENERGITO study). International Journal of Chronic Obstructive Pulmonary Disease 2016;11:193-205. [DOI: 10.2147/COPD.S95055] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Derom E, Beeh K, Echave-Sustaeta J, Grönke L, Zhai D, Bjermer L. Tiotropium + olodaterol provides significant lung-function benefits compared to fluticasone + salmeterol regardless of prior bronchodilator use. European Respiratory Journal 2016;48:PA978. [DOI: 10.1183/13993003.congress-2016] [DOI] [Google Scholar]
Donohue 2015a {published data only (unpublished sought but not used)}
- Donohue J, Worsley S, Zhu CQ, Hardaker L, Church A. Efficacy and safety of umeclidinium/vilanterol (UMEC/VI) once daily (OD) vs fluticasone/salmeterol combination (FSC) twice daily (BD) in patients with moderate-to-severe COPD and infrequent COPD exacerbations. Chest 2014;146(4):73A. [Google Scholar]
- Donohue JF, Worsley S, Zhu CQ, Hardaker L, Church A. Improvements in lung function with umeclidinium/vilanterol versus fluticasone propionate/salmeterol in patients with moderate-to-severe COPD and infrequent exacerbations. Respiratory Medicine 2015;109(7):870-81. [DOI: 10.1016/j.rmed.2015.04.018] [PMID: ] [DOI] [PubMed] [Google Scholar]
Donohue 2015b {published data only (unpublished sought but not used)}
- Donohue J, Worsley S, Zhu CQ, Hardaker L, Church A. Efficacy and safety of umeclidinium/vilanterol (UMEC/VI) once daily (OD) vs fluticasone/salmeterol combination (FSC) twice daily (BD) in patients with moderate-to-severe COPD and infrequent COPD exacerbations. Chest 2014;146(4):73A. [Google Scholar]
- Donohue JF, Worsley S, Zhu CQ, Hardaker L, Church A. Improvements in lung function with umeclidinium/vilanterol versus fluticasone propionate/salmeterol in patients with moderate-to-severe COPD and infrequent exacerbations. Respiratory Medicine 2015;109(7):870-81. [DOI: 10.1016/j.rmed.2015.04.018] [PMID: ] [DOI] [PubMed] [Google Scholar]
Ferguson 2018 {published data only}
- Ferguson GT, Rabe KF, Martinez FJ, Fabbri LM, Wang C, Ichinose M, et al . Triple therapy with budesonide/glycopyrrolate/formoterol fumarate with co-suspension delivery technology versus dual therapies in chronic obstructive pulmonary disease (KRONOS): a double-blind, parallel-group, multicentre, phase 3 randomised controlled trial. Lancet Respiratory Medicine 2018;10:747-58. [DOI: 10.1016/S2213-2600(18)30327-8] [PMID: ] [DOI] [PubMed] [Google Scholar]
- JPRN-JapicCTI-184079. A Randomized, Double-Blind, Parallel-Group, 24-Week, Chronic-Dosing, Multi-Center Study to Assess the Efficacy and Safety of PT010, PT003, and PT009 Compared With Symbicort Turbuhaler as an Active Control in Subjects With Moderate to Very Severe Chronic Obstructive Pulmonary Disease (PT010006). www.who.int/trialsearch/Trial2.aspx?TrialID=JPRN-JapicCTI-184079.
- JPRN-JapicCTI-184080. A Randomized, Double-Blind, Parallel-Group, 28-Week, Chronic-Dosing, Multi-Center, Extension Study to Assess the Safety and Efficacy of PT010, PT003, and PT009 in Japanese Subjects With Moderate to Very Severe Chronic Obstructive Pulmonary Disease (COPD) Compared With Symbicort Turbuhaler as an Active Control (PT010007). www.who.int/trialsearch/Trial2.aspx?TrialID=JPRN-JapicCTI-184080.
- NCT03262012. Study to assess the safety and efficacy of PT010, PT003, and PT009 in Japanese subjects with COPD compared with Symbicort Turbohaler. ClinicalTrials.gov/show/NCT03262012 (first posted 25 August 2017).
Frith 2018 {published data only}
- Frith PA, Ashmawi S, Krishnamurthy S, Gurgun A, Hristoskova S, Pilipovic V, et al. Efficacy and safety of the direct switch to indacaterol/glycopyrronium from salmeterol/fluticasone in non-frequently exacerbating COPD patients: the FLASH randomized controlled trial. Respirology 2018;12:1152-9. [DOI: 10.1111/resp.13374] [PMID: ] [DOI] [PubMed] [Google Scholar]
Herth 2020 {published data only}
- Herth F, Hohlfeld JM, Haas J, Hoz A, Jin X, Kreitner K-F, et al. The effect of tiotropium/olodaterol versus fluticasone propionate/salmeterol on left ventricular filling and lung hyperinflation in patients with COPD. BMJ Open Respiratory Research 2020;7(1):e000741. [DOI: 10.1136/bmjresp-2020-000741] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hoshino 2015 {published data only}
- Hoshino M, Ohtawa J, Akitsu K. Comparison of airway dimensions with once daily tiotropium plus indacaterol versus twice daily Advair in chronic obstructive pulmonary disease. Pulmonary Pharmacology and Therapeutics 2015;30:128-33. [DOI: 10.1016/j.pupt.2014.08.002] [PMID: ] [DOI] [PubMed] [Google Scholar]
Lipson 2018 {published data only}
- Lipson DA, Barnhart F, Boucot I, Crim C, Brealey N, Criner GJ, et al. Comparison of LAMA/LABA vs ICS/LABA in high risk COPD patients: pre-specified analysis on lung function and health status from the IMPACT trial. European Respiratory Journal 2018;52:PA4385. [DOI: 10.1183/13993003.congress-2018.PA4385] [DOI] [Google Scholar]
- Lipson DA, Barnhart F, Boucot I, Crim C, Brealey N, Criner GJ, et al. Exacerbation outcomes with LAMA/LABA and ICS/LABA in high risk COPD patients in the IMPACT trial. European Respiratory Journal 2018;52:PA4384. [DOI: 10.1183/13993003.congress-2018.PA4384] [DOI] [Google Scholar]
- Lipson DA, Barnhart F, Boucot I, Crim C, Brealey N, Criner GJ. Comparison of LAMA/LABA vs ICS/LABA in high risk COPD patients: pre-specified analysis on lung function and health status from the IMPACT trial. Pneumologie 2019;73(Suppl 1):[no pagination]. [Google Scholar]
- Lipson DA, Barnhart F, Brealey N, Brooks J, Criner GJ, Day NC, et al. Once-daily single-inhaler triple versus dual therapy in patients with COPD. New England Journal of Medicine 2018;378(18):1671-80. [DOI: 10.1056/NEJMoa1713901] [PMID: ] [DOI] [PubMed] [Google Scholar]
Magnussen 2012 {published data only}
- Magnussen H, Maltais F, Schmidt H, Kesten S, Metzdorf N. Comparison of tiotropium+salmeterol vs. fluticasone+salmeterol on lung volumes, exercise tolerance and locus of symptom limitation. American Journal of Respiratory and Critical Care Medicine 2010;181:A4472. [Google Scholar]
- Magnussen H, Paggiaro P, Schmidt H, Kesten S, Metzdorf N, Maltais F. Effect of combination treatment on lung volumes and exercise endurance time in COPD. Respiratory Medicine 2012;106(10):1413-20. [DOI: 10.1016/j.rmed.2012.05.011] [PMID: ] [DOI] [PubMed] [Google Scholar]
Mostafa 2021 {published data only}
- Mostafa TM, El-Azab GA, Atia GA, Lotfy NS. The effectiveness of 3 combined therapeutic regimens in Egyptian patients with moderate-to-severe chronic obstructive pulmonary disease: a randomized double-blind prospective pilot study. Current Therapeutic Research Clinical and Experimental 2021;94:100625. [DOI: 10.1016/j.curtheres.2021.100625] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
NCT03240575 {published data only}
- NCT03240575. The ENERGITO 2 study compares 2 inhaled medicines for chronic obstructive pulmonary disease (COPD). One medicine is a combination of tiotropium and olodaterol (Stiolto) taken using the Respimat inhaler and the other medicine is a combination of fluticasone and salmeterol taken using the Diskus. ClinicalTrials.gov/show/NCT03240575 (first posted 7 August 2017).
Rabe 2008 {published data only (unpublished sought but not used)}
- Rabe KF, Timmer W, Sagkriotis A, Viel K. Comparison of a combination of tiotropium plus formoterol to salmeterol plus fluticasone in moderate COPD. Chest 2008;134(2):255-62. [DOI: 10.1378/chest.07-2138] [PMID: ] [DOI] [PubMed] [Google Scholar]
- Rabe KF, Timmer W, Sagriotis A, Viel K. Comparison of a combination of tiotropium and formoterol to salmeterol and fluticasone in moderate COPD. In: European Respiratory Society 15th Annual Congress; 2005 Sep 17-20; Copenhagen. 2005.
Rabe 2020 {published data only}
- Rabe KF, Martinez FJ, Ferguson GT, Wang C, Singh D, Wedzicha JA, et al. Triple inhaled therapy at two glucocorticoid doses in moderate-to-very-severe COPD. New England Journal of Medicine 2020;383(1):35-48. [DOI: 10.1056/NEJMoa1916046] [PMID: ] [DOI] [PubMed] [Google Scholar]
Singh 2015 {published data only (unpublished sought but not used)}
- Singh D, Worsley S, Zhu CQ, Hardaker L, Church A. Umeclidinium/vilanterol (UMEC/VI) once daily (OD) vs fluticasone/salmeterol combination (FSC) twice daily (BD) in patients with moderate-to-severe COPD and infrequent COPD exacerbations. European Respiratory Journal 2014;44(Suppl 58):P290. [Google Scholar]
- Singh D, Worsley S, Zhu CQ, Hardaker L, Church A. Umeclidinium/vilanterol versus fluticasone propionate/salmeterol in COPD: a randomised trial. BMC Pulmonary Medicine 2015;15:91. [DOI: 10.1186/s12890-015-0092-1] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Vogelmeier 2013 {published data only (unpublished sought but not used)}
- Bateman E, Vogelmeier C, Chen H, Banerji D. Comparison of COPD exacerbations with once-daily QVA149 versus twice-daily salmeterol/fluticasone combination: the ILLUMINATE study. American College of Chest Physicians 2014;145(3):409A. [Google Scholar]
- Bateman ED, Vogelmeier C, Pallante J, Bryant H, Alagappan V, D'Andrea P, et al. Once-daily QVA149 demonstrates superior lung function compared to twice-daily salmeterol/fluticasone in all subgroups of COPD patients: the ILLUMINATE study. American Journal of Respiratory and Critical Care Medicine 2013;187:A4273. [Google Scholar]
- Bateman ED, Vogelmeier C, Pallante J, Bryant H, Alagappan V, D'Andrea P, et al. Once-daily QVA149 improves breathlessness and reduces rescue medication use compared to twice-daily salmeterol/fluticasone in patients with COPD: the ILLUMINATE study. American Journal of Respiratory and Critical Care Medicine 2013;187:A2433. [Google Scholar]
- Mezzi K, Pallante J, Alagappan V, Chen H, Banerji D. Once-daily QVA149 demonstrates superior outcomes in COPD patients previously treated with fixed-dose long-acting β2-agonist/inhaled corticosteroid (LABA/ICS): the ILLUMINATE study. Chest 2014;145(3):424A. [Google Scholar]
- Vogelmeier C, Bateman E, Pallante J, Bryant H, Alagappan VD, Andrea P, et al. Once-daily QVA149 provides superior bronchodilation and improves lung function versus twice-daily fluticasone/salmeterol in COPD patients: the ILLUMINATE study. British Thoracic Society Winter Meeting 2012;67:A149, P194. [Google Scholar]
- Vogelmeier C, Bateman ED, Pallante J, Bryant H, Alagappan V, D'Andrea, et al. QVA149 once daily is safe and well tolerated in patients with COPD: the ILLUMINATE study. American Journal of Respiratory and Critical Care Medicine 2013;187:A1477. [Google Scholar]
- Vogelmeier CD, Bateman ED, D'Andrea P, Mezzi K, Chen H, Banerji D, et al. Once-daily QVA149 is more effective than twice-daily salmeterol/fluticasone in improving lung function, in patients with severe chronic obstructive pulmonary disease (COPD): the illuminate study. American Journal of Respiratory and Critical Care Medicine 2014;189:A3763. [Google Scholar]
- Vogelmeier CF, Bateman ED, Pallante J, Alagappan VK, D'Andrea P, Chen H, et al. Efficacy and safety of once-daily QVA149 compared with twice-daily salmeterol-fluticasone in patients with chronic obstructive pulmonary disease (ILLUMINATE): a randomised, double-blind, parallel group study. Lancet Respiratory Medicine 2013;1(1):51-60. [DOI: 10.1016/S2213-2600(12)70052-8] [PMID: ] [DOI] [PubMed] [Google Scholar]
Vogelmeier 2016 {published data only (unpublished sought but not used)}
- Greulich T, Kostikas K, Gaga M, Aalamian-Mattheis M, Lossi NS, Patalano F, et al. Indacaterol/glycopyrronium reduces the risk of clinically important deterioration after direct switch from baseline therapies in patients with moderate COPD: a post hoc analysis of the CRYSTAL study. International Journal of Chronic Obstructive Pulmonary Disease 2018;13:1229-37. [DOI: 10.2147/COPD.S159732] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogelmeier C, Paggiaro PL, Dorca J, Sliwinski P, Mallet M, Kirsten A, et al. The efficacy and safety of aclidinium/formoterol fixed-dose combination compared with salmeterol/fluticasone in patients with COPD: results from a phase III study. American Journal of Respiratory and Critical Care Medicine 2015;191:A3974. [Google Scholar]
- Vogelmeier C, Paggiaro PL, Dorca J, Sliwinski P, Mallet M, Kirsten AM, et al. Efficacy and safety of aclidinium/formoterol versus salmeterol/fluticasone: a phase 3 COPD study. European Respiratory Journal 2016;48(4):1030-9. [DOI: 10.1183/13993003.00216-2016] [PMID: ] [DOI] [PubMed] [Google Scholar]
Vogelmeier 2017 {published data only}
- Vogelmeier CF, Gaga M, Aalamian-Mattheis M, Greulich T, Marin JM, Castellani W, et al. Efficacy and safety of direct switch to indacaterol/glycopyrronium in patients with moderate COPD: the CRYSTAL open-label randomised trial. Respiratory Research 2018;18(1):140. [DOI: 10.1186/s12931-017-0622-x] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Wedzicha 2016 {published data only (unpublished sought but not used)}
- Kwaijtaal M, Wedzicha J, Decramer M, Vestbo J, Banerji D. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. Respirology 2014;TP:177. [DOI] [PubMed] [Google Scholar]
- Wedzicha J, Vestbo J, Gallagher N, Banerji D. A novel study design for the comparison between once-daily QVA149 and twice-daily salmeterol/fluticasone on the reduction of COPD exacerbations: the FLAME study. Chest 2014;145(3):408A. [Google Scholar]
- Wedzicha JA, Banerji D, Chapman KR, Vestbo J, Roche N, Ayers RT, et al. Indacaterol-glycopyrronium versus salmeterol-fluticasone for COPD. New England Journal of Medicine 2016;374(23):2222-34. [DOI: 10.1056/NEJMoa1516385] [PMID: ] [DOI] [PubMed] [Google Scholar]
Zhong 2015 {published data only (unpublished sought but not used)}
- Zhong N, Wang C, Zhou X, Zhang N, Humphries M, Wang L, et al. LANTERN: a randomized study of QVA149 versus salmeterol/fluticasone combination in patients with COPD. International Journal of Chronic Obstructive Pulmonary Disease 2015;10:1015-26. [DOI: 10.2147/COPD.S84436] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies excluded from this review
Anzueto 2017 {published data only}
- Anzueto AR, Vogelmeier CF, Kostikas K, Mezzi K, Fucile S, Bader G, et al. The effect of indacaterol/glycopyrronium versus tiotropium or salmeterol/fluticasone on the prevention of clinically important deterioration in COPD. International Journal of Chronic Obstructive Pulmonary Disease 2017;12:1325-37. [DOI: 10.2147/COPD.S133307] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Aziz 2018 {published data only}
- Aziz MIA, Tan LE, Wu DB, Pearce F, Chua GSW, Lin L, et al. Comparative efficacy of inhaled medications (ICS/LABA, LAMA, LAMA/LABA and SAMA) for COPD: a systematic review and network meta-analysis. International Journal of Chronic Obstructive Pulmonary Disease 2018;13:3203-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bruhn 2003 {published data only}
- Bruhn C. Chronic obstructive pulmonary disease: recommendation of salmeterol in fixed combination. Deutsche Apotheker-Zeitung 2003;143(11):48-51. [Google Scholar]
Calverley 2007 {published data only}
- Calverley P, Stockley R, Seemungal T, Hagan G, Wedzicha J. Adverse events and mortality in the INSPIRE study (Investigating New Standards for Prophylaxis In Reduction of Exacerbations). In: European Respiratory Society 17th Annual Congress; 2007 Sep 16-18; Stockholm. Vol. 30. 2007:125s P847.
EUCTR2015‐002046‐31‐ES {unpublished data only}
- EUCTR2015-002046-31-ES. A research study to compare two treatments for treating a respiratory disease known as asthma-COPD overlap syndrome (ACOS) [A randomised, single blind, cross-over study to compare a fixed dose combination of fluticasone propionate / formoterol fumarate (fluticasone /formoterol breath actuated inhaler (BAI)) with a fixed dose combination of indacaterol maleate / glycopyrronium bromide (Ultibro Breezhaler) in subjects with fixed airflow obstruction and elevated eosinophils]. trialsearch.who.int/?TrialID=EUCTR2015-002046-31-ES (first received 3 February 2016).
Knobil 2004a {published data only}
- Knobil K, Kalberg C, Merchant K, Emmett A, Cicale M. Maintenance of bronchodilator response for Advair Diskus 250/50 (fluticasone propionate/salmeterol) but not ipratropium/albuterol in patients with COPD. Chest 2004;126(Suppl 4):807S. [Google Scholar]
Knobil 2004b {published data only}
- Knobil K, Merchant K, Kalberg C, Emmett A, Cicale M. A comparison of patient perceived improvement in symptoms after initiating therapy with either Advair Diskus (fluticasone propionate/salmeterol) 250/50 or ipratropium/albuterol. Chest 2004;126(Suppl 4):806S-b-807S-b. [Google Scholar]
Mahler 2016 {published data only}
- Mahler DA, Keininger DL, Mezzi K, Fogel R, Banerji D. Efficacy of indacaterol/glycopyrronium in patients with COPD who have increased dyspnea with daily activities. Chronic Obstructive Pulmonary Disease 2016;3(4):758-68. [DOI: 10.15326/jcopdf.3.4.2016.0138] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Michael 2016 {published data only}
- Michael JA, Dana Z. Is combination long-acting beta-agonist and long-acting muscarinic antagonist therapy the future of COPD therapy? Clinical Pulmonary Medicine 2016;23(6):288-9. [DOI: 10.1097/CPM.0000000000000181] [DOI] [Google Scholar]
NCT00120978 {published data only}
- NCT00120978. Can advair and flovent reduce systemic inflammation related to chronic obstructive pulmonary disease (COPD)? A multi-center randomized controlled trial. clinicaltrials.gov/ct2/show/NCT00120978 (first received 19 July 2005).
NCT03376295 {unpublished data only}
- NCT03376295. Comparative effectiveness of COPD treatments. clinicaltrials.gov/show/NCT03376295 (first posted 18 December 2017).
NCT03504527 {unpublished data only}
- NCT03504527. Efficiency of budesonide combined with formoterol and tiotropium in the treatment of acute exacerbation of ACO. ClinicalTrials.gov/show/NCT03504527 (first posted 20 April 2018).
NCT04138758 {unpublished data only}
- NCT04138758. Comparative effectiveness and safety of tiotropium and olodaterol in comparison to LABA/ICS [Effectiveness and safety of maintenance treatment with combination of tiotropium and olodaterol in comparison to maintenance treatment with a combination of inhaled corticosteroids and long-acting β2 agonists in COPD patients]. clinicaltrials.gov/ct2/show/NCT04138758 (first posted 30 November 2020).
NCT04320342 {unpublished data only}
- NCT04320342. A Phase III Study Comparing Efficacy, Safety and Tolerability of the Fixed Dose Triple Combination CHF 5993 With the Fixed Dose Dual Combination CHF 1535 in Subjects With COPD. clinicaltrials.gov/show/NCT04320342 (first posted 25 March 2020).
NCT04923347 {unpublished data only}
- NCT04923347. A Study to Evaluate the Safety and Efficacy of Fluticasone Furoate (FF)/Umeclidinium(UMEC)/Vilanterol (VI) in Participants With Chronic Obstructive Pulmonary Disease (COPD). clinicaltrials.gov/ct2/show/NCT04923347 (first posted 11 June 2021).
NCT05097014 {unpublished data only}
- NCT05097014. CHF5993 and CHF1535 pMDI on Lung Hyperinflation and Exercise Endurance Time in Subjects With COPD. clinicaltrials.gov/show/NCT05097014 (first posted 27 October 2021).
Oba 2016 {published data only}
- Oba Y, Chandran AV, Devasahayam JV. Long-acting muscarinic antagonist versus inhaled corticosteroid when added to long-acting β-agonist for COPD: a meta-analysis. COPD 2016;13(6):677-85. [DOI: 10.3109/15412555.2016.1170799] [PMID: ] [DOI] [PubMed] [Google Scholar]
Papi 2018 {published data only}
- Papi A, Vestbo J, Fabbri L, Corradi M, Prunier H, Cohuet G. Extrafine inhaled triple therapy versus dual bronchodilator therapy in chronic obstructive pulmonary disease (TRIBUTE): a double-blind, parallel group, randomised controlled trial. Lancet 2018;391(10125):1076-84. [DOI: 10.1016/S0140-6736(18)30206-X] [PMID: ] [DOI] [PubMed] [Google Scholar]
Pavord 2016 {published data only}
- Pavord ID, Lettis S, Anzueto A, Barnes N. Blood eosinophil count and pneumonia risk in patients with chronic obstructive pulmonary disease: a patient-level meta-analysis. Lancet Respiratory Medicine 2016;4(9):731-41. [DOI: 10.1016/S2213-2600(16)30148-5] [PMID: ] [DOI] [PubMed] [Google Scholar]
Price 2014 {published data only}
- Price D, Keininger D, Costa-Scharplatz M, Mezzi K, Dimova M, Asukai Y, et al. Cost-effectiveness of the LABA/LAMA dual bronchodilator indacaterol/glycopyrronium in a Swedish healthcare setting. Respiratory Medicine 2014;108(12):1786-93. [DOI] [PubMed] [Google Scholar]
Sciurba 2004 {published data only}
- Sciurba FC, Kalberg C, Emmett A, Merchant K, Brown C, Knobil K. Efficacy of Advair Diskus 250/50 (fluticasone propionate/salmeterol) or ipratropium/albuterol in patients with COPD associated with chronic bronchitis and/or emphysema. Chest 2004;126(Suppl 4):807S-a-808S-a. [Google Scholar]
Singh 2019 {published data only}
- Singh D, Fabbri LM, Corradi M, Georges G, Guasconi A, Vezzoli S, et al. Extrafine triple therapy in patients with symptomatic COPD and history of one moderate exacerbation. European Respiratory Journal 2019;53(5):1900235. [DOI: 10.1183/13993003.00235-2019] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Skoupa 2018 {published data only}
- Skoupa J, Kasak V, Klimes J, Valena T. Indacaterol/glycopyrronium versus salmeterol/fluticasone in patients with COPD. A cost-effectiveness analysis in the Czech Republic. Value in Health Regional Issues 2018;16:112-8. [DOI: 10.1016/j.vhri.2018.09.002] [PMID: ] [DOI] [PubMed] [Google Scholar]
UMIN000024905 {unpublished data only}
- UMIN000024905. A randomized, open-label, crossover study to evaluate ICS/LABA treatment versus LAMA/LABA treatment in COPD with eosinophilic inflammation. center6.umin.ac.jp/cgi-open-bin/ctr_e/ctr_view.cgi?recptno=R000028656 (first posted 21 November 2016).
References to ongoing studies
EUCTR2016‐004473‐41‐GB {unpublished data only}
- EUCTR2016-004473-41-GB. Investigating whether two different COPD inhalers have different effects on chest infections [INvestigating COPD Outcomes, Genomics and Neutrophilic Inflammation with Tiotropium and Olodaterol (INCOGNITO trial) - INCOGNITO trial]. trialsearch.who.int/?TrialID=EUCTR2016-004473-41-GB (first received 21 March 2017).
Additional references
Alagha 2014
- Alagha K, Palot A, Sofalvi T, Pahus L, Gouitaa M, Tummino C, et al. Long-acting muscarinic receptor antagonists for the treatment of chronic airway diseases. Therapeutic Advances in Chronic Disease 2014;5(2):85-98. [DOI: 10.1177/2040622313518227] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Anderson 2014
- Anderson R, Theron AJ, Steel HC, Durandt C, Tintinger GR, Feldman C. The beta-2-adrenoreceptor agonists, formoterol and indacaterol, but not salbutamol, effectively suppress the reactivity of human neutrophils in vitro. Mediators of Inflammation 2014;2014:105420. [DOI: 10.1155/2014/105420] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Barnes 2010
- Barnes PJ. Inhaled corticosteroids in COPD: a controversy. Respiration 2010;80(2):89-95. [DOI: 10.1159/000315416] [PMID: ] [DOI] [PubMed] [Google Scholar]
Burrows 1966
- Burrows B, Fletcher CM, Heard BE, Jones NL, Wootliff JS. The emphysematous and bronchial types of chronic airways obstruction. A clinicopathological study of patients in London and Chicago. Lancet 1966;1(7442):830-5. [DOI: 10.1016/s0140-6736(66)90181-4] [PMID: ] [DOI] [PubMed] [Google Scholar]
Donohue 2005
- Donohue JF. Minimal clinically important differences in COPD lung function. COPD 2005;2(1):111-24. [DOI: 10.1081/copd-200053377] [PMID: ] [DOI] [PubMed] [Google Scholar]
Drivenes 2014
- Drivenes E, Ostrem A, Melbye H. Predictors of ICS/LABA prescribing in COPD patients: a study from general practice. BMC Family Practice 2014;15:42. [DOI: 10.1186/1471-2296-15-42] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Esteban 2018
- Esteban C, Castro-Acosta A, Alvarez-Martínez CJ, Capelastegui A, López-Campos JL, Pozo-Rodriguez F. Predictors of one-year mortality after hospitalization for an exacerbation of COPD. BMC Pulmonary Medicine 2018;18(1):18. [DOI: 10.1186/s12890-018-0574-z] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Festic 2015
- Festic E, Scanlon PD. Incident pneumonia and mortality in patients with chronic obstructive pulmonary disease. A double effect of inhaled corticosteroids? American Journal of Respiratory and Critical Care Medicine 2015;191(2):141-8. [DOI: 10.1164/rccm.201409-1654PP] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Frampton 2014
- Frampton JE. QVA149 (indacaterol/glycopyrronium fixed-dose combination): a review of its use in patients with chronic obstructive pulmonary disease. Drugs 2014;74(4):465-88. [DOI: 10.1007/s40265-014-0194-8] [PMID: ] [DOI] [PubMed] [Google Scholar]
GOLD 2016
- The Global Initiative for Chronic Obstructive Lung Disease. From the Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2016. www.goldcopd.org/Guidelines/guidelines-resources.html (accessed 7 December 2016).
GOLD 2023
- The Global Initiative for Chronic Obstructive Lung Disease. From the Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2023. www.goldcopd.org/2023-gold-report-2/ (accessed 15 December 2022).
GRADEpro [Computer program]
- GRADEpro GDT. Version 3.2 for Windows. Hamilton (ON): McMaster University, 2008. Available at gradepro.org.
Guyatt 2008
- Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336(7650):924-6. [DOI: 10.1136/bmj.39489.470347.AD] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hanania 2008
- Hanania NA. The impact of inhaled corticosteroid and long-acting beta-agonist combination therapy on outcomes in COPD. Pulmonary Pharmacology and Therapeutics 2008;21(3):540-50. [DOI: 10.1016/j.pupt.2007.12.004] [PMID: ] [DOI] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327(7414):557-60. [DOI: 10.1136/bmj.327.7414.557] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JP, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Horita 2015a
- Horita N, Kaneko T. Role of combined indacaterol and glycopyrronium bromide (QVA149) for the treatment of COPD in Japan. International Journal of Chronic Obstructive Pulmonary Disease 2015;10:813-22. [DOI: 10.2147/COPD.S56067] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Malerba 2014
- Malerba M, Morjaria JB, Radaeli A. Differential pharmacology and clinical utility of emerging combination treatments in the management of COPD - role of umeclidinium/vilanterol. International Journal of Chronic Obstructive Pulmonary Disease 2014;9:687-95. [DOI: 10.2147/COPD.S47792] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Moher 2009
- Moher D, Liberati A, Tetzlaff J, Altman D. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Medicine 2009;6(7):e1000097. [DOI: 10.1371/journal.pmed.1000097] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Nannini 2013
- Nannini LJ, Poole P, Milan SJ, Holmes R, Normansell R. Combined corticosteroid and long-acting beta2-agonist in one inhaler versus placebo for chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews 2013, Issue 11. Art. No: CD003794. [DOI: 10.1002/14651858.CD003794.pub4] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Oba 2018
- Oba Y, Keeney E, Ghatehorde N, Dias S. Dual combination therapy versus long-acting bronchodilators alone for chronic obstructive pulmonary disease (COPD): a systematic review and network meta-analysis. Cochrane Database of Systematic Reviews 2018, Issue 12. Art. No: CD012620. [DOI: 10.1002/14651858.CD012620.pub2] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pascoe 2015
- Pascoe S, Locantore N, Dransfield MT, Barnes NC, Pavord ID. Blood eosinophil counts, exacerbations, and response to the addition of inhaled fluticasone furoate to vilanterol in patients with chronic obstructive pulmonary disease: a secondary analysis of data from two parallel randomised controlled trials. Lancet Respiratory Medicine 2015;3(6):435-42. [DOI: 10.1016/S2213-2600(15)00106-X] [PMID: ] [DOI] [PubMed] [Google Scholar]
Pauwels 2001
- Pauwels RA, Buist AS, Calverley PM, Jenkins CR, Hurd SS, GOLD Scientific Committee. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Global Initiative for Chronic Obstructive Lung Disease (GOLD) Workshop summary. American Journal of Respiratory and Critical Care Medicine 2001;163(5):1256-76. [DOI: 10.1164/ajrccm.163.5.2101039] [PMID: ] [DOI] [PubMed] [Google Scholar]
Reddel 2021
- Reddel HK, Bacharier LB, Bateman ED, Brightling CE, Brusselle GG, Buhl R, et al. Global Initiative for Asthma Strategy 2021: executive summary and rationale for key changes. European Respiratory Journal 2021;59(1):2102730. [DOI: 10.1183/13993003.02730-2021] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Schachter 2013
- Schachter EN. Indacaterol/glycopyrronium bromide fixed-dose combination for the treatment of COPD. Drugs Today 2013;49(7):437-6. [DOI: 10.1358/dot.2013.49.7.1980496] [PMID: ] [DOI] [PubMed] [Google Scholar]
Suissa 2009
- Suissa S, Barnes PJ. Inhaled corticosteroids in COPD: the case against. European Respiratory Journal 2009;34(1):13-6. [DOI: 10.1183/09031936.00190908] [PMID: ] [DOI] [PubMed] [Google Scholar]
Suissa 2018
- Suissa S, Drazen JM. Making sense of triple inhaled therapy for COPD. New England Journal of Medicine 2018;378(18):1723-4. [DOI: 10.1056/NEJMe1716802.] [PMID: ] [DOI] [PubMed] [Google Scholar]
Tashkin 2004
- Tashkin DP, Cooper CB. The role of long-acting bronchodilators in the management of stable COPD. Chest 2004;125(1):249-59. [DOI: 10.1378/chest.125.1.249] [PMID: ] [DOI] [PubMed] [Google Scholar]
White 2013
- White P, Thornton H, Pinnock H, Georgopoulou S, Booth HP. Overtreatment of COPD with inhaled corticosteroids - implications for safety and costs: cross-sectional observational study. PLoS One 2013;8(10):e75221. [DOI: 10.1371/journal.pone.0075221] [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 2019
- World Health Organization. Global health estimates: life expectancy and leading causes of death and disability. www.who.int/data/gho/data/themes/mortality-and-global-health-estimates (accessed 1 January 2022).
References to other published versions of this review
Horita 2016
- Horita N, Goto A, Ota E, Nakashima K, Nagai K, Kaneko T. Long-acting muscarinic antagonist plus long-acting beta agonist versus long-acting beta agonist plus inhaled corticosteroid for stable chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews 2016, Issue 2. Art. No: CD012066. [DOI: 10.1002/14651858.CD012066] [DOI] [PMC free article] [PubMed] [Google Scholar]
Horita 2017
- Horita N, Goto A, Shibata Y, Ota E, Nakashima K, Nagai K, Kaneko T. Long‐acting muscarinic antagonist (LAMA) plus long‐acting beta‐agonist (LABA) versus LABA plus inhaled corticosteroid (ICS) for stable chronic obstructive pulmonary disease (COPD). Cochrane Database of Systematic Reviews 2017, Issue 2. Art. No: CD012066. [DOI: 10.1002/14651858.CD012066.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]


