Abstract
In a prospective study, we measured the associations between three serum elements (Se, Zn and Cu) and the prognosis of 1475 patients with four different types of cancer (breast, prostate, lung and larynx) from University Hospitals in Szczecin, Poland. The elements were measured in serum taken after diagnosis and prior to treatment. Patients were followed from the date of diagnosis until death from any cause or until the last follow-up date (mean years of follow-up: 6.0–9.8 years, according to site). Kaplan–Meier curves were constructed for all cancers combined and for each cancer separately. Age-adjusted hazard ratios (HRs) were estimated using Cox regression. The outcome was all-cause mortality. A Se level in the highest quartile was also associated with a reduced mortality (HR = 0.66; 95%CI 0.49–0.88; p = 0.005) in all-cause mortality for all cancers combined. Zn level in the highest quartile was also associated with reduced mortality (HR = 0.55; 95%CI 0.41–0.75; p = 0.0001). In contrast, a Cu level in the highest quartile was associated with an increase in mortality (HR = 1.91; 95%CI 1.56–2.08; p = 0.0001). Three serum elements—selenium, zinc and copper—are associated with the prognosis of different types of cancer.
Keywords: copper, zinc, selenium, breast cancer, laryngeal cancer, lung cancer, prostate cancer, survival
1. Introduction
There is increasing interest in the role of serum/blood elements, including metals and metalloids, in the survival of cancer patients [1]. Cancers are heterogenous, but many cancers share genetic and epigenetic features in common and cancers at different sites and with different histologies may respond to the same chemotherapy. The fate of a patient with cancer is determined, in part, by the genetic and epigenetic makeup of the tumor itself (cell autonomous), but there are host factors as well (local and macro-environment) that influence the outcome (non-cell autonomous) [2]. Notable examples include tumor-infiltrating lymphocytes [3], the microbiome [4] and platelets [5]. It is possible that the metastatic niche permits the metastases and growth of more than one tumor type, and the metastatic potential of different cancers is under the influence of common factors. Among host factors, blood levels of several micro-elements have been implicated in cancer incidence and progression. We have previously reported on the effects of selenium, zinc and copper on the rates of cancer incidence [6,7,8] and cancer progression [9,10,11,12] in patients from our institution, but these studies have been site-specific.
1.1. Selenium
In a human body, selenium (Se) combines with proteins that are incorporated in the form of selenocysteine. As a component of selenoproteins, selenium plays both enzymatic and structural roles [13]. Some of the important functions of selenoproteins are the production of thyroid hormones, the stimulation of the immune system and protection against oxidative stress [14]. Reports on humans have matched well with the mechanisms of selenium activities described in animals [15,16]. Both a deficiency and excess of selenium can have adverse effects on the body, including, e.g., cardiac disorders, hypertension, reduced efficiency of the immune system, thyroid function disorders, disorders of bone mineralization, malformation of teeth and increased risks of cancer [17,18,19,20,21]. There are few studies which have evaluated the effect of selenium levels on cancer survival. Individual reports indicate that better survival is associated with higher serum selenium levels in patients with cancers of the breast, colon, malignant melanoma, larynx, lung and renal cell carcinoma [9,22,23,24,25,26,27,28].
1.2. Zinc
Zinc (Zn) is an essential element necessary for health which offers protection against free radicals. Zinc is a component of superoxide dismutase (SOD-2). It is also involved in the immunological processes and the function of the skin and mucous membranes. Zinc supports the storage and secretion of insulin from the pancreas and maintains normal levels of other micronutrients, including selenium, magnesium and copper. Zinc plays a role in the detoxification of heavy metals [29]. It has been observed that zinc accumulates in some cancer cells [30]. Normal prostate epithelial cells accumulate zinc; however, in prostate cancer cells, zinc concentration is reduced [31]. It has been proposed that zinc has anticancer effects, inhibiting the growth of cancer cells and activating apoptosis. Some studies have reported serum zinc levels to be higher in cancer patients than in others [32,33,34], whereas other studies have reported that it is lower [35]. Some studies suggest that dietary zinc has chemo-preventive properties. People with a diet rich in zinc have a lower risk of lung and colorectal cancers [36,37]. Moreover, some studies suggest that high dietary zinc intake [38] and higher zinc levels in the serum correlate with better survival in patients with prostate [38] and laryngeal cancers [26].
1.3. Copper
Copper (Cu) is an essential element that is involved in many physiological processes [5,6,7,8,9] and is a cofactor for numerous biological processes. Copper is a component of several metalloenzymes, including metalloproteinases, and plays roles in angiogenesis and oxidative phosphorylation. Several studies have reported high serum/blood copper levels in patients with prostate cancer [10,11,12] and other cancers, including lymphoma, reticulum cell sarcoma, laryngeal carcinomas, cervical, breast, pancreas, stomach and lung cancers [39,40,41]. We have recently shown that a high copper level was associated with an increased risk for the development of colon cancer [42]. In patients with breast and colorectal cancer, serum copper levels were correlated with disease stage [42,43]. There are few studies on the impact of circulating copper and cancer survival [44,45].
In this study, we evaluated the serum levels at diagnosis of three circulating elements as prognostic markers for four common cancers. We hypothesized that the elements’ levels are associated with survival are similar irrespective of the cancer site.
2. Materials and Methods
2.1. Study Group
We studied 1475 patients with four different types of cancer (breast, prostate, lung and larynx). All cases were unselected patients treated between 2009 and 2018 at the Clinical Hospitals of Pomeranian Medical University in Szczecin, Poland, or at an associated hospital. All patients were seen at these hospitals for the period specified and were enrolled into the study. Typically, these patients are offered genetic testing shortly after diagnosis during an out-patient visit to one of our clinics and are offered the opportunity to participate in other clinical research studies. Blood samples were taken from cases between 2009 and 2018 shortly after diagnosis and prior to treatment. Patients were restricted to those diagnosed at age 85 and younger. Medical charts were reviewed for date of diagnosis, smoking status (yes/no) and chemotherapy (yes/no). Additional information was collected for breast cancer (ER status, nodal status and tumor size) and for prostate cancer (Gleason grade, PSA (prostate-specific antigen) at diagnosis and prostatectomy (yes/no)). Tumor stage was assigned to one to four stages for larynx and lung cancer. The study was conducted in accordance with the Helsinki Declaration and with the consent of the Ethics Committee of Pomeranian Medical University in Szczecin under the number KB-0012/73/10 on 21 June 2010. All participants provided written informed consent to be enrolled in the study.
2.2. Sample Collection, and Storage and Measurement of Elements
Patients were asked to fast for six hours before sample collection. A total of 10 mL of peripheral blood was collected into a Beckton Dickinson Vacutainer tube [REF.367953] containing a clot activator. After the collection, the tubes were incubated at room temperature for a minimum 30 min to clot, but no longer than 120 min, and after which the tubes were centrifuged at 1300× g for 12 min. After the centrifugation, the serum was transferred into cryovials and deposited at −80 °C until analysis. On the day of analysis, the sera were thawed, vortexed and centrifuged at 5000× g for 5 min.
Determination of selenium (78Se), zinc (66Zn) and copper (65Cu) was performed using an ICP mass spectrometer ELAN DRC-e (PerkinElmer). Before each analytical run, the instrument was tuned to meet the manufacturer’s criteria. Oxygen was used as a reaction gas. Technical details are available upon request. The spectrometer was calibrated using external calibration. Calibration standards were prepared fresh daily from 10 µg/mL of Multi-Element Calibration Standard 3 (PerkinElmer, Waltham, MA, USA) by diluting it with blank reagent to the final concentration of 1, 2, 5, 10, 50 and 100 µg/L for Se, Zn and Cu determination. Correlation coefficients for calibration curves were always greater than 0.99. Matrix-matched calibration was used. The analysis protocol assumed 30-fold dilution of serum in a blank reagent. The blank reagent consisted of high-purity water (>18 MΩ), TMAH (Alfa Aesar, Tewksbury, MA, USA), Triton X-100 (PerkinElmer), n-butanol (Merck, Darmstadt, Germany) and EDTA (Sigma-Aldrich, St. Louis, MO, USA) [46,47,48]. The accuracy and precision of measurements were tested using the certified reference material (CRM) Clinchek Plasmonorm Serum Trace Elements Level 1 (Recipe, Munchen, Germany).
2.3. Statistical Analysis
For each of the three elements, patients were assigned to one of three categories based on the distribution of the values for the elements in the entire dataset. The cutoffs were low (<25th percentile), middle (25th to 75th percentile) and high (75th percentile). Cutoff values were determined separately for men and for women.
Patients in each of the four cancer site groups were compared based on a range of variables, including age, sex, the median level of each of the elements and five-year survival. We also conducted a correlation analysis for the levels of elements (two by two) using Pearson’s correlation coefficient.
The principal outcome was death from any cause. Death was ascertained by linking to the Polish Statistics Registry. Patients were followed for all-cause survival at diagnosis using survival analyses. Five-year survival was estimated for each site using the Kaplan–Meier method and crude statistical differences were assessed using the log-rank test. Statistical significance was set at p < 0.05 (two-sided).
To estimate the association of each of the three elements with survival in all patients combined, hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated using the Cox proportional hazards model. Patients were followed from the date of diagnosis to death or 1 January 2022. For each comparison the middle category was defined as the reference group. The initial analysis included all patients. The basic model was adjusted for age of diagnosis only. In the multivariable model, outcomes were adjusted for age at diagnosis, cancer site, current smoking status (yes/no), sex and the other two elements. The HRs were estimated for low and high levels of selenium, zinc and copper, and were compared to the middle level.
A series of site-specific analyses was then conducted. For each site, three analyses were conducted, one for each element. For prostate cancer, the multivariable HRs were adjusted for age at diagnosis, PSA (four levels) and Gleason grade (<7; 7+). For breast cancer, the multivariable HRs were adjusted for age at diagnosis, ER status, tumor size and nodal status (positive/negative). For lung and laryngeal cancer, the HRs were adjusted for sex, stage, age at diagnosis and current smoking status. Statistical analyses were conducted in SAS version 9.4.
3. Results
A total of 1475 cancer patients were enrolled in this study (794 men and 681 women). The patient characteristics are presented in Table 1. The mean levels of selenium, zinc and copper were compared with regard to cancer type (Table 1), age and sex (Table 2).
Table 1.
Characteristics of the 1475 patients in the study.
| Cancer Site | ||||
|---|---|---|---|---|
| Breast n = 531 |
Prostate n = 347 |
Lung n = 298 |
Larynx n = 299 |
|
| Age of diagnosis | ||||
| Years (mean, range) | 57.2 (26–84) | 66.0 (41–84) | 64.1 (43–84) | 61.0 (40–81) |
| Sex | ||||
| Male | 0 | 347 (100%) | 193 (65%) | 254 (85%) |
| Female | 531 (100%) | 0 | 105 (35%) | 45 (15%) |
|
Year of diagnosis (mean, range) |
2011 (2009–2016) | 2013 (2010–2015) | 2011 (2010–2012) | 2013 (2010–2018) |
| Selenium level (µg/L) (mean, range) |
86 (52–172) |
78 (42–138) |
62 (17–108) |
58 (21–105) |
| Zinc level (µg/L) (mean, range) |
867 (525–11045) |
847 (516–1340) |
718 (350–1071) |
640 (358–1318) |
| Copper level (µg/L) (mean, range) |
1153 (685–2153) |
1093 (460–2197) |
1146 (671–2866) |
1116 (436–2795) |
| Years of follow-up (mean) | 9.4 | 7.6 | 6.0 | 6.8 |
| 5- year survival | 85.9% | 83.3% | 45.3% | 63.2% |
Table 2.
Mean levels of copper, zinc and selenium according to age and sex.
| Men (n = 794) | Women (n = 681) | |||||
|---|---|---|---|---|---|---|
| Age of Diagnosis Frequency M/F |
Selenium (µg/L) Mean, Range |
Zinc (µg/L) Mean, Range |
Copper (µg/L) Mean, Range |
Selenium (µg/L) Mean, Range |
Zinc (µg/L) Mean, Range |
Copper (µg/L) Mean, Range |
| <50 20/139 |
1213 (839–1545) |
665 (377–884) |
61 (31–92) |
1150 (500–2049) |
840 (525–1254) |
83 (40–124) |
| 50–60 221/227 |
1124 (643–1391) |
729 (358–1244) |
65 (21–138) |
1158 (632–2151) |
867 (455–11045) |
83 (35–172) |
| 60–70 376/213 |
1090 (436–2197) |
761 (359–1340) |
69 (25–109) |
1172 (658–2866) |
810 (350–1389) |
80 (33–123) |
| 70+ 177/102 |
1092 (460–1842) |
745 (366–1190) |
67 (17–106) |
1161 (710–1910) |
794 (553–1128) |
76 (36–121) |
Across all age categories, zinc and selenium levels were higher in women than in men (Table 2). Copper levels were similar in men and women. There was little variation in the elements by age (Table 2). The cutoff levels for percentiles for men and women corresponding to the low, middle and high categories are presented in Table 3.
Table 3.
Cutoff levels used to determine low, middle and high categories for each element by sex.
| Men (n = 794) | Women (n = 681) | |||||
|---|---|---|---|---|---|---|
| Cutoff Level | Selenium (µg/L) |
Zinc (µg/L) |
Copper (µg/L) |
Selenium (µg/L) |
Zinc (µg/L) |
Copper (µg/L) |
| 25 percentile | 56 | 636 | 944 | 71 | 727 | 1011 |
| 75 percentile | 78 | 850 | 1230 | 93 | 899 | 1260 |
The mean levels of zinc were lower in patients with laryngeal and lung cancer than in patients with other cancer types. Only 7% of lung cancer patients and 3% of laryngeal cancer patients had a zinc level in the high category. The correlations between the serum levels of the three elements are presented in Table 4. There was a modest positive correlation between the serum zinc and selenium levels.
Table 4.
Correlation coefficients for three elements studied (n = 1475).
| Selenium | Zinc | Copper | |
|---|---|---|---|
| Selenium | - | ||
| Zinc | 0.195 p < 0.0001 |
- | |
| Copper | 0.059 p = 0.13 |
0.087 p = 0.02 |
- |
Five-year survival rates were 45% for lung cancer, 63% for laryngeal cancer, 83% for prostate cancer and 86% for breast cancer. The crude five-year survival rates by cancer site and serum levels of the three elements are presented in Figure 1, Figure 2, Figure 3 and Figure 4.
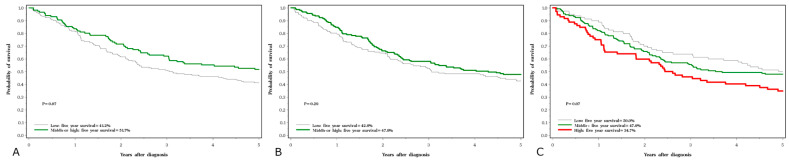
Figure 1.
Five-year survival associated with three element levels in lung cancer subjects; (A) selenium, (B) zinc, (C) copper.
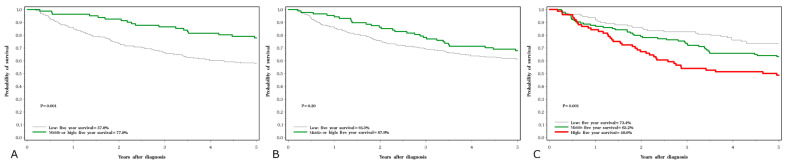
Figure 2.
Five-year survival associated with three element levels in larynx cancer subjects; (A) selenium, (B) zinc, (C) copper.
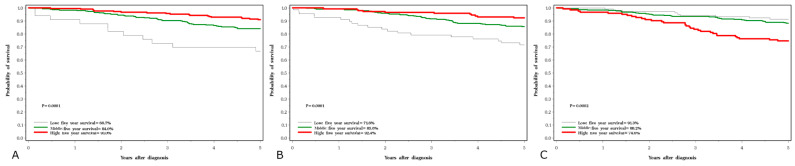
Figure 3.
Five-year survival associated with three element levels in breast cancer subjects; (A) selenium, (B) zinc, (C) copper.
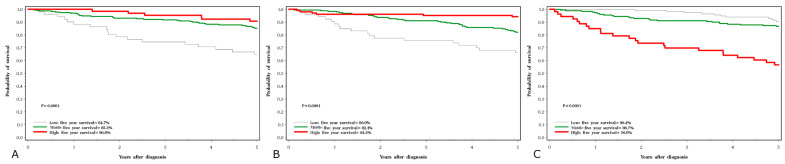
Figure 4.
Five-year survival associated with three element levels in prostate cancer subjects; (A) selenium, (B) zinc, (C) copper.
In the basic (age-adjusted) model, we sought to see if serum elements were predictive of survival in all cancer sites combined. In general, high levels of all three elements were predictive of survival. The associations persisted after adjusting for cancer site, age at diagnosis, smoking and age (Table 5). A low zinc level was associated with relatively poor survival and a high zinc level was associated with a relatively good survival. A low selenium level was associated with poor survival and a high selenium level was associated with good survival. In contrast, a low copper level was associated with good survival and a high copper level was associated with poor survival.
Table 5.
HRs associated with low and high serum metals all-cause mortality (all sites combined).
| Total /Deaths |
Age-Adjusted Only | Adjusted | |
|---|---|---|---|
| HR (95%CI) p-Value | HR (95%CI) p-Value | ||
| Selenium | |||
| Low | 484/250 | 2.18 (1.81–2.62) < 0.0001 | 1.61 (1.28–2.02) < 0.0001 |
| Middle | 702/206 | 1.0 | 1.0 |
| High | 289/58 | 0.66 (0.49–0.88) 0.005 | 0.71 (0.52–0.96) 0.03 |
| Zinc | |||
| Low | 473/224 | 1.68 (1.40–2.01) < 0.0001 | 1.19 (0.97–1.47) 0.10 |
| Middle | 737/240 | 1.0 | 1.0 |
| High | 265/50 | 0.55 (0.41–0.75) 0.0001 | 0.72 (0.52–0.99) 0.04 |
| Copper | |||
| Low | 406/115 | 0.86 (0.69–1.33) 0.19 | 0.81 (0.64–1.01) 0.07 |
| Middle | 746/239 | 1.0 | 1.0 |
| High | 323/160 | 1.91 (1.56–2.08) < 0.0001 | 1.72 (1.41–2.11) < 0.0001 |
| Current Smoker | |||
| No | 641/183 | 1 | 1 |
| Yes | 676/278 | 1.77 (1.46–2.13) < 0.0001 | 0.97 (0.76–1.25) 0.83 |
| Missing | 158/53 | 1.11 (0.81–1.51) 0.51 | 1.46 (0.99–2.15) 0.06 |
| Sex | |||
| Female | 681/214 | 1 | 1 |
| Male | 794/300 | 1.28 (1.06–1.53) 0.009 | 1.21 (0.91–1.60) 0.18 |
Adjusted model adjusted for age, sex and current smoking status in addition to the other elements and cancer sites.
HRs for the association between the three elements and all-cause mortality for the individual sites are presented in Table 6, Table 7, Table 8 and Table 9.
Table 6.
HRs associated with low and high serum metals and all-cause mortality (lung cancer subjects).
| Total /Deaths |
Age-Adjusted Only | Adjusted | |
|---|---|---|---|
| HR (95%CI) p-Value | HR (95%CI) p-Value | ||
| Selenium | |||
| Low | 182/109 | 1.34 (0.97–1.85) 0.08 | 1.16 (0.81–1.66) 0.43 |
| Middle | 111/55 | 1.0 | 1.0 |
| High | 5/2 | 0.68 (0.17–2.80) 0.59 | 1.14 (0.27–4.85) 0.86 |
| Zinc | |||
| Low | 141/83 | 1.25 (0.91–1.71) 0.17 | 1.17 (0.83–1.65) 0.37 |
| Middle | 145/74 | 1.0 | 1.0 |
| High | 12/9 | 1.73 (0.86–3.45) 0.12 | 1.33 (0.62–2.87) 0.47 |
| Copper | |||
| Low | 80/41 | 0.88 (0.61–1.29) 0.52 | 0.97 (0.65–1.46) 0.88 |
| Middle | 146/78 | 1.0 | 1.0 |
| High | 72/47 | 1.39 (0.97–2.01) 0.07 | 1.19 (0.82–1.73) 0.36 |
Adjusted model adjusted for age, sex, stage and current smoking status in addition to the other elements.
Table 7.
HRs associated with low and high serum metals and all-cause mortality (larynx cancer subjects).
| Total /Deaths |
Age-Adjusted Only | Adjusted | |
|---|---|---|---|
| HR (95%CI) p-Value | HR (95%CI) p-Value | ||
| Selenium | |||
| Low | 218/96 | 2.61 (1.51–4.50) 0.0006 | 2.19 (1.23–3.91) 0.008 |
| Middle | 72/15 | 1.0 | 1.0 |
| High | 9/5 | 3.32 (1.20–9.18) 0.002 | 2.10 (0.59–7.50) 0.26 |
| Zinc | |||
| Low | 212/87 | 1.46 (0.94–2.26) 0.09 | 1.03 (0.65–1.65) 0.89 |
| Middle | 82/26 | 1.0 | 1.0 |
| High | 5/3 | 2.35 (0.71–7.79) 0.16 | 1.46 (0.31–6.86) 0.63 |
| Copper | |||
| Low | 109/32 | 0.71 (0.45–1.12) 0.14 | 0.95 (0.59–1.53) 0.83 |
| Middle | 114/44 | 1.0 | 1.0 |
| High | 76/40 | 1.64 (1.06–2.52) 0.003 | 1.15 (0.74–1.79) 0.54 |
Adjusted model adjusted for age, sex stage and current smoking status in addition to the other elements.
Table 8.
HRs associated with low and high serum metals and all-cause mortality (breast cancer subjects).
| Total /Deaths |
Age-Adjusted Only | Adjusted | |
|---|---|---|---|
| HR (95%CI) p-Value | HR (95%CI) p-Value | ||
| Selenium | |||
| Low | 33/21 | 2.43 (1.49–3.97) 0.0004 | 2.55 (1.52–4.28) 0.0004 |
| Middle | 288/82 | 1.0 | 1.0 |
| High | 210/40 | 0.64 (0.44–0.94) 0.02 | 0.69 (0.46–1.03) 0.07 |
| Zinc | |||
| Low | 67/29 | 1.52 (0.99–2.34) 0.05 | 1.37 (0.87–2.18) 0.18 |
| Middle | 320/89 | 1.0 | 1.0 |
| High | 144/25 | 0.58 (0.37–0.90) 0.02 | 0.72 (0.46–1.14) 0.16 |
| Copper | |||
| Low | 103/21 | 0.83 (0.51–1.34) 0.44 | 0.87 (0.52–1.44) 0.59 |
| Middle | 306/78 | 1.0 | 1.0 |
| High | 122/44 | 1.70 (1.17–2.46) 0.005 | 1.55 (1.05–2.29) 0.03 |
Adjusted model adjusted for age, tumor size, nodes, ER status and current smoking status in addition to the other elements.
Table 9.
HRs associated with low and high serum metals and all-cause mortality (prostate cancer subjects).
| Total /Deaths |
Age-Adjusted Only | Adjusted | |
|---|---|---|---|
| HR (95%CI) p-Value | HR (95%CI) p-Value | ||
| Selenium | |||
| Low | 51/24 | 1.91 (1.16–3.14) 0.01 | 1.57 (0.94–2.65) 0.09 |
| Middle | 231/54 | 1.0 | 1.0 |
| High | 65/11 | 0.84 (0.44–1.61) 0.59 | 1.02 (0.51–2.02) 0.96 |
| Zinc | |||
| Low | 53/25 | 1.52 (0.93–2.48) 0.10 | 1.30 (0.77–2.19) 0.33 |
| Middle | 190/51 | 1.0 | 1.0 |
| High | 104/13 | 0.50 (0.27–0.93) 0.03 | 0.51 (0.27–0.96) 0.04 |
| Copper | |||
| Low | 114/21 | 0.84 (0.49–1.42) 0.51 | 0.93 (0.54–1.59) 0.78 |
| Middle | 180/39 | 1.0 | 1.0 |
| High | 53/29 | 2.73 (1.67–4.44) <0.0001 | 2.71 (1.64–4.48) 0.0001 |
Adjusted model adjusted for age, PSA level, Gleason grade and current smoking status in addition to the other elements.
For lung cancer, the associations were modest and were attenuated after adjusting for tumor stage (Table 6).
For laryngeal cancer, a low selenium level was predictive of poor survival in both the crude data (Figure 2C) and after adjusting for stage (Table 7). For each of the three elements, the quartile levels were based on the distribution of the entire patient cohort and were divided by sex. For both lung and laryngeal cancer there were few subjects with a zinc level or selenium level in the highest quartile, and for these evaluations the top three quartiles were merged.
For breast cancer and prostate cancer, the associations with all three elements were similar (Table 8 and Table 9). For breast cancer, both a high copper and a low selenium level were predictive of poor survival (Table 8, Figure 3B,C). There was also a trend related to poor survival with lower levels of zinc (Figure 3A). Similar effects were seen with prostate cancer (Table 9, Figure 4). For breast and prostate cancer, the HRs were attenuated slightly after adjusting for grade and the other elements.
4. Discussion
In this study, we evaluated the serum levels at diagnosis of three circulating elements as prognostic markers for various common cancers. The current study updates our previous reports for selenium and zinc [7,8,9,22,23,25,27] and extends the follow-up time by linking to the Polish Vital Statistics Registry. We have also added new data for copper.
In general, we can see that high selenium and zinc levels were predictive of a good prognosis and high copper levels were detrimental, but the associations and trends varied across the four cancer sites and were attenuated after adjusting for tumor stage. We report strong associations between a high copper level and poor survival for both breast and prostate cancer. The associations were present in the age-adjusted analysis and persisted after adjustment for other prognostic factors. A low serum selenium level was also associated with a poor prognosis for breast cancer patients, as reported previously [22,23].
Serum selenium and zinc predicted mortality for laryngeal and lung cancer patients, but the associations were modest and were attenuated after adjusting for stage [9,27]. Selenium and zinc levels in patients with larynx and lung cancer were low overall, suggesting that these elements play important roles in lung and laryngeal cancer incidence more than in cancer survival [26,27,49]. There were too few patients with laryngeal or lung cancer with high zinc or selenium levels to obtain precise HRs for these patient groups.
There are several limitations to our study. We did not include patients representing all cancer sites; notably, we did not include patients with gastrointestinal or gynecologic cancers. These will be the subject of future studies. We have recently reported that elevated blood copper was associated with the presence of colon cancer [8], but it is too early to assess survival in our cohort. Nevertheless, our patient population represented three of the four most common sites in Poland. We did not have details on the cause of death because the study was conducted by vital status linkage and the cause of death is not available in the Polish Death Registry. There are many factors which influence prognosis and there may be unmeasured confounding factors as well. Possible confounders include family histories, dietary supplements, comorbidity, exercise and BMI. Ongoing studies are underway to assess the ten-year follow up, to expand the range of cancer sites as well as to determine the specific cause of death.
The strength of this study is the large sample of patients (n = 1475), and all three elements were measured in all patients using standard methods in a single laboratory. For consistency and simplicity, we divided patients into three categories representing low, middle and high. These are constructed distinctions and are not intended to reflect current standards or recommendations. We elected to use the highest 75th percentile as the high category. This was not based on pre-existing standards but was chosen for statistical power and expediency. However, by using this definition very few of the lung and laryngeal cancer patients fell into the high category.
We measured the elements in the serum after diagnosis but prior to treatment. This method was chosen to exclude the effect of treatments on serum element levels. However, it would be of interest to see if changes in the serum level of copper and the other elements post-treatment are reflective of the presence of residual disease, metastatic disease, and/or if they predict prognosis post-treatment. If so, they may serve as useful tumor markers, as is the case for CA125 [50,51], PSA [52], platelets [53] and circulating tumor DNA [54].
The associations here are not proof of causality, but are hypothesis generating. The specific mechanisms by which the metals influence prognosis are uncertain. The levels of different metals may be a marker of risk, but not causally in the progression pathway. Selenium, zinc and copper are all involved in many metabolic processes that can impact prognosis. It is possible that the driver of progression is a conjugate protein activity and the metal level may be a consequence of the underlying physiological process—such that homeostasis is altered—with a resultant change in the serum level of the element. To provide a brief overview, selenium compounds can generate oxygen free radicals and various selenoproteins, including glutathione peroxidase 1–4 and 6, as well as thioredoxin reductase, which have also been linked to neoplastic growth [55,56,57]. Zinc is known to be involved in oxidative stress (including zinc/copper superoxide dismutase), DNA repair (replication and transcription via zinc finger proteins and controlling DNA binding activity, such as AP-1, NFκB and p53), cell signaling and apoptosis (also via NFκB and AP-1) [58,59]. Finally, copper plays a critical role in cellular oxygenation, the neutralization of free radicals, angiogenesis and cellular iron metabolism. A novel pathway of copper-dependent cell-death, cuproptosis [60,61], may also be linked to cancer prognosis.
The sheer number of proteins and processes which involve these metals present a challenge to clarify the mechanism. Nevertheless, we are encouraged to pursue clinical studies directed at lowering the serum copper level and raising the levels of zinc and selenium in selected patients. Zinc and selenium can be given as supplements, but to lower copper, it is necessary to give a chelating agent such as penicillamine or TNT. Ultimately, the utility of the treatment will depend on the mechanism of action. If the elements facilitate metastatic spread, it may be challenging because for breast and other cancers, the first progression (metastases) may precede the diagnosis. However, if the elements promote the growth of cancer cells within the metastatic niche or prevent grade progression, then providing the therapy post-diagnosis may be appropriate.
5. Conclusions
Three serum elements—selenium, zinc and copper—are associated with the prognosis of different types of cancer. A high copper level can be associated with an increase in all-cause mortality for the studied cancers (breast, prostate, lung and larynx). In contrast, high zinc and selenium levels can be associated with reduced mortality. Clinical studies directed at lowering serum copper levels and raising the levels of zinc and selenium in selected cancer patients are required.
Acknowledgments
We thank Renata Gibaszek and Ewa Putresza for their support of this study.
Author Contributions
Conceptualization, J.L. and S.A.N.; methodology, W.M., R.D., K.P. and P.S.; software, K.P. and P.S.; validation, W.M. and R.D.; formal analysis, K.P. and P.S.; investigation, M.R.L., W.M., S.P. and R.D.; resources, C.C., J.G., T.D. and T.H.; data curation, J.L., M.R.L., W.M., S.P., R.D., C.C., J.G., T.D., A.J., T.H., K.P. and P.S.; writing—original draft preparation, J.L., M.R.L., S.P. and S.A.N.; writing—review and editing, J.L., M.R.L., S.P., M.M., K.P. and S.A.N.; visualization, J.L., M.R.L., S.P. and S.A.N.; supervision, J.L. and S.A.N.; project administration, J.L., S.P. and S.A.N.; funding acquisition, J.L. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of the Pomeranian Medical University in Szczecin (Poland) under the number KB-0012/73/10.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data supporting the reported results are available from the first author upon request from all interested researchers.
Conflicts of Interest
Jan Lubiński is the CEO of Read-Gene SA, which offers measurements on a micro-and macroelement level. These authors are part-time employees of Read-Gene: W.M., R.D., C.C., J.G. and T.H. The other authors declare that they have no conflicts of interest.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Vella V., Malaguarnera R., Lappano R., Maggiolini M., Belfiore A. Recent views of heavy metals as possible risk factors and potential preventive and therapeutic agents in prostate cancer. Mol. Cell. Endocrinol. 2017;457:57–72. doi: 10.1016/j.mce.2016.10.020. [DOI] [PubMed] [Google Scholar]
- 2.Feunteun J., Ostyn P., Delaloge S. Tumor cell malignancy: A complex trait built through reciprocal interactions between tumors and tissue-body system. iScience. 2022;25:104217. doi: 10.1016/j.isci.2022.104217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.de Jong V.M., Wang Y., ter Hoeve N.D., Opdam M., Stathonikos N., Jóźwiak K., Hauptmann M., Cornelissen S., Vreuls W., Rosenberg E.H., et al. Prognostic Value of Stromal Tumor-Infiltrating Lymphocytes in Young, Node-Negative, Triple-Negative Breast Cancer Patients Who Did Not Receive (neo)Adjuvant Systemic Therapy. J. Clin. Oncol. 2022;40:2361–2374. doi: 10.1200/JCO.21.01536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sepich-Poore G.D., Zitvogel L., Straussman R., Hasty J., Wargo J.A., Knight R. The microbiome and human cancer. Science. 2021;371:6536. doi: 10.1126/science.abc4552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Giannakeas V., Kotsopoulos J., Cheung M.C., Rosella L., Brooks J.D., Lipscombe L., Akbari M.R., Austin P.C., Narod S.A. Analysis of Platelet Count and New Cancer Diagnosis Over a 10-Year Period. JAMA Netw. Open. 2022;5:e2141633. doi: 10.1001/jamanetworkopen.2021.41633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Białkowska K., Marciniak W., Muszyńska M., Baszuk P., Gupta S., Jaworska-Bieniek K., Sukiennicki G., Durda K., Gromowski T., Prajzendanc K., et al. Association of zinc level and polymorphism in MMP-7 gene with prostate cancer in Polish population. PLoS ONE. 2018;13:e0201065. doi: 10.1371/journal.pone.0201065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lener M.R., Scott R.J., Wiechowska-Kozłowska A., Serrano-Fernández P., Baszuk P., Jaworska-Bieniek K., Sukiennicki G., Marciniak W., Muszyńska M., Kładny J., et al. Serum Concentrations of Selenium and Copper in Patients Diagnosed with Pancreatic Cancer. Cancer Res. Treat. 2016;48:1056–1064. doi: 10.4143/crt.2015.282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Baszuk P., Marciniak W., Derkacz R., Jakubowska A., Cybulski C., Gronwald J., Dębniak T., Huzarski T., Białkowska K., Pietrzak S., et al. Blood Copper Levels and the Occurrence of Colorectal Cancer in Poland. Biomedicines. 2021;9:1628. doi: 10.3390/biomedicines9111628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pietrzak S., Wójcik J., Scott R.J., Kashyap A., Grodzki T., Baszuk P., Bielewicz M., Marciniak W., Wójcik N., Dębniak T., et al. Influence of the selenium level on overall survival in lung cancer. J. Trace Elem. Med. Biol. 2019;56:46–51. doi: 10.1016/j.jtemb.2019.07.010. [DOI] [PubMed] [Google Scholar]
- 10.Habib F., Dembinski T., Stitch S. The zinc and copper content of blood leucocytes and plasma from patients with benign and malignant prostates. Clin. Chim. Acta. 1980;104:329–335. doi: 10.1016/0009-8981(80)90390-3. [DOI] [PubMed] [Google Scholar]
- 11.Gupte A., Mumper R.J. Elevated copper and oxidative stress in cancer cells as a target for cancer treatment. Cancer Treat. Rev. 2009;35:32–46. doi: 10.1016/j.ctrv.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 12.Safi R., Nelson E.R., Chitneni S.K., Franz K.J., George D.J., Zalutsky M.R., McDonnell D.P. Copper Signaling Axis as a Target for Prostate Cancer Therapeutics. Cancer Res. 2014;74:5819–5831. doi: 10.1158/0008-5472.CAN-13-3527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hariharan S., Dharmaraj S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology. 2020;28:667–695. doi: 10.1007/s10787-020-00690-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Combs G.F., Clark L.C., Turnbull B.W. An analysis of cancer prevention by selenium. Biofactors. 2001;14:153–159. doi: 10.1002/biof.5520140120. [DOI] [PubMed] [Google Scholar]
- 15.Zhao L., Liu M., Sun H., Yang J.-C., Huang Y.-X., Huang J.-Q., Lei X., Sun L.-H. Selenium deficiency-induced multiple tissue damage with dysregulation of immune and redox homeostasis in broiler chicks under heat stress. Sci. China Life Sci. 2023 doi: 10.1007/s11427-022-2226-1. [DOI] [PubMed] [Google Scholar]
- 16.Xu Z.-J., Liu M., Niu Q.-J., Huang Y.-X., Zhao L., Lei X.G., Sun L.-H. Both selenium deficiency and excess impair male reproductive system via inducing oxidative stress-activated PI3K/AKT-mediated apoptosis and cell proliferation signaling in testis of mice. Free Radic. Biol. Med. 2023;197:15–22. doi: 10.1016/j.freeradbiomed.2023.01.024. [DOI] [PubMed] [Google Scholar]
- 17.Reddy V.N., Giblin F.J., Lin L.R., Dang L., Unakar N.J., Musch D.C., Boyle D.L., Takemoto L.J., Ho Y.S., Knoernschild T., et al. Glutathione peroxidase-1 deficiency leads to increased nuclear light scattering, membrane damage, and cataract formation in gene-knockout mice. Investig. Ophthalmol. Vis. Sci. 2001;42:3247–3255. [PubMed] [Google Scholar]
- 18.Kuria A., Fang X., Li M., Han H., He J., Aaseth J.O., Cao Y. Does dietary intake of selenium protect against cancer? A systematic review and meta-analysis of population-based prospective studies. Crit. Rev. Food Sci. Nutr. 2020;60:684–694. doi: 10.1080/10408398.2018.1548427. [DOI] [PubMed] [Google Scholar]
- 19.Jenkins D.J., Kitts D., Giovannucci E.L., Sahye-Pudaruth S., Paquette M., Mejia S.B., Patel D., Kavanagh M., Tsirakis T., Kendall C.W.C., et al. Selenium, antioxidants, cardiovascular disease, and all-cause mortality: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2020;112:1642–1652. doi: 10.1093/ajcn/nqaa245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schomburg L. The other view: The trace element selenium as a micronutrient in thyroid disease, diabetes, and beyond. Hormones. 2020;19:15–24. doi: 10.1007/s42000-019-00150-4. [DOI] [PubMed] [Google Scholar]
- 21.Kenfield S.A., Van Blarigan E.L., Dupre N., Stampfer M.J., Giovannucci E.L., Chan J.M. Selenium Supplementation and Prostate Cancer Mortality. J. Natl. Cancer Inst. 2014;107:dju360. doi: 10.1093/jnci/dju360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Szwiec M., Marciniak W., Derkacz R., Huzarski T., Gronwald J., Cybulski C., Dębniak T., Jakubowska A., Lener M., Falco M., et al. Serum Selenium Level Predicts 10-Year Survival after Breast Cancer. Nutrients. 2021;13:953. doi: 10.3390/nu13030953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lubinski J., Marciniak W., Muszynska M., Huzarski T., Gronwald J., Cybulski C., Jakubowska A., Debniak T., Falco M., Kladny J., et al. Serum selenium levels predict survival after breast cancer. Breast Cancer Res. Treat. 2018;167:591–598. doi: 10.1007/s10549-017-4525-9. [DOI] [PubMed] [Google Scholar]
- 24.Psathakis D., Wedemeyer N., Oevermann E., Krug F., Siegers C.-P., Bruch H.-P. Blood selenium and glutathione peroxidase status in patients with colorectal cancer. Dis. Colon Rectum. 1998;41:328–335. doi: 10.1007/BF02237487. [DOI] [PubMed] [Google Scholar]
- 25.Rogoża-Janiszewska E., Malińska K., Baszuk P., Marciniak W., Derkacz R., Lener M., Jakubowska A., Cybulski C., Huzarski T., Masojć B., et al. Serum Selenium Level and 10-Year Survival after Melanoma. Biomedicines. 2021;9:991. doi: 10.3390/biomedicines9080991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lubiński J., Marciniak W., Muszynska M., Jaworowska E., Sulikowski M., Jakubowska A., Kaczmarek K., Sukiennicki G., Falco M., Baszuk P., et al. Serum selenium levels and the risk of progression of laryngeal cancer. PLoS ONE. 2018;13:e0184873. doi: 10.1371/journal.pone.0184873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lubiński J., Jaworowska E., Derkacz R., Marciniak W., Białkowska K., Baszuk P., Scott R.J., Lubiński J.A. Survival of Laryngeal Cancer Patients Depending on Zinc Serum Level and Oxidative Stress Genotypes. Biomolecules. 2021;11:865. doi: 10.3390/biom11060865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Meyer H.A., Endermann T., Stephan C., Stoedter M., Behrends T., Wolff I., Jung K., Schomburg L. Selenoprotein P Status Correlates to Cancer-Specific Mortality in Renal Cancer Patients. PLoS ONE. 2012;7:e46644. doi: 10.1371/journal.pone.0046644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nasiadek M., Stragierowicz J., Klimczak M., Kilanowicz A. The Role of Zinc in Selected Female Reproductive System Disorders. Nutrients. 2020;12:2464. doi: 10.3390/nu12082464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.To P.K., Do M.H., Cho J.-H., Jung C. Growth Modulatory Role of Zinc in Prostate Cancer and Application to Cancer Therapeutics. Int. J. Mol. Sci. 2020;21:2991. doi: 10.3390/ijms21082991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zaichick V.Y., Sviridova T.V., Zaichick S.V. Zinc in the human prostate gland: Normal, hyperplastic and cancerous. Int. Urol. Nephrol. 1997;29:565–574. doi: 10.1007/BF02552202. [DOI] [PubMed] [Google Scholar]
- 32.El-Ahmady O., El-Maraghy A., Ibrahim A., Ramzy S. Serum copper, zinc, and iron in patients with malignant and benign pulmonary diseases. Nutrition. 1995;11:498–501. [PubMed] [Google Scholar]
- 33.Pasha Q., Malik S.A., Shah M.H. Statistical analysis of trace metals in the plasma of cancer patients versus controls. J. Hazard. Mater. 2008;153:1215–1221. doi: 10.1016/j.jhazmat.2007.09.115. [DOI] [PubMed] [Google Scholar]
- 34.Siddiqui M., Jyoti, Singh S., Mehrotra P., Singh K., Sarangi R. Comparison of some trace elements concentration in blood, tumor free breast and tumor tissues of women with benign and malignant breast lesions: An Indian study. Environ. Int. 2006;32:630–637. doi: 10.1016/j.envint.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 35.Kuo H.W., Chen S.F., Wu C.C., Chen D.R., Lee J.H. Serum and Tissue Trace Elements in Patients with Breast Cancer in Taiwan. Biol. Trace Element Res. 2002;89:1–11. doi: 10.1385/BTER:89:1:1. [DOI] [PubMed] [Google Scholar]
- 36.Zhou W., Park S., Liu G., Miller D.P., Wang L.I., Pothier L., Wain J.C., Lynch T.J., Giovannucci E., Christiani D.C. Dietary Iron, Zinc, and Calcium and the Risk of Lung Cancer. Epidemiology. 2005;16:772–779. doi: 10.1097/01.ede.0000181311.11585.59. [DOI] [PubMed] [Google Scholar]
- 37.Zhang X., Giovannucci E.L., Smith-Warner S.A., Wu K., Fuchs C.S., Pollak M., Willett W.C., Ma J. A prospective study of intakes of zinc and heme iron and colorectal cancer risk in men and women. Cancer Causes Control. 2011;22:1627–1637. doi: 10.1007/s10552-011-9839-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Epstein M.M., Kasperzyk J.L., Andrén O., Giovannucci E.L., Wolk A., Håkansson N., Andersson S.-O., Johansson J.-E., Fall K., Mucci L.A. Dietary zinc and prostate cancer survival in a Swedish cohort. Am. J. Clin. Nutr. 2011;93:586–593. doi: 10.3945/ajcn.110.004804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Lossow K., Schwarz M., Kipp A.P. Are trace element concentrations suitable biomarkers for the diagnosis of cancer? Redox Biol. 2021;42:101900. doi: 10.1016/j.redox.2021.101900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Arredondo M., Núñez M.T. Iron and copper metabolism. Mol. Asp. Med. 2005;26:313–327. doi: 10.1016/j.mam.2005.07.010. [DOI] [PubMed] [Google Scholar]
- 41.Yoshida Y., Furuta S., Niki E. Effects of metal chelating agents on the oxidation of lipids induced by copper and iron. Biochim. Et Biophys. Acta (BBA)—Lipids Lipid Metab. 1993;1210:81–88. doi: 10.1016/0005-2760(93)90052-B. [DOI] [PubMed] [Google Scholar]
- 42.Tapiero H., Townsend D., Tew K. Trace elements in human physiology and pathology. Copper. Biomed. Pharmacother. 2003;57:386–398. doi: 10.1016/S0753-3322(03)00012-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Linder M.C., Hazegh-Azam M. Copper biochemistry and molecular biology. Am. J. Clin. Nutr. 1996;63:797S–811S. doi: 10.1093/ajcn/63.5.797. [DOI] [PubMed] [Google Scholar]
- 44.Zabłocka-Słowińska K., Prescha A., Płaczkowska S., Porębska I., Kosacka M., Pawełczyk K. Serum and Whole Blood Cu and Zn Status in Predicting Mortality in Lung Cancer Patients. Nutrients. 2020;13:60. doi: 10.3390/nu13010060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fang A., Chen P., Wang X., Liu Z., Zhang D., Luo Y., Liao G., Long J., Zhong R., Zhou Z., et al. Serum copper and zinc levels at diagnosis and hepatocellular carcinoma survival in the Guangdong Liver Cancer Cohort. Int. J. Cancer. 2019;144:2823–2832. doi: 10.1002/ijc.31991. [DOI] [PubMed] [Google Scholar]
- 46.Jones D.R., Jarrett J.M., Tevis D.S., Franklin M., Mullinix N.J., Wallon K.L., Quarles C.D., Caldwell K.L., Jones R.L. Analysis of whole human blood for Pb, Cd, Hg, Se, and Mn by ICP-DRC-MS for biomonitoring and acute exposures. Talanta. 2017;162:114–122. doi: 10.1016/j.talanta.2016.09.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Clases D., de Vega R.G. Facets of ICP-MS and their potential in the medical sciences—Part 1: Fundamentals, stand-alone and hyphenated techniques. Anal. Bioanal. Chem. 2022;414:7337–7361. doi: 10.1007/s00216-022-04259-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhang G., Zhang F., Liu W., Liu C., You J., Tian M., Cao T., Jiang J., Yang Z., Wu H., et al. A simple, rapid method for simultaneous determination of multiple elements in serum by using an ICP-MS equipped with collision cell. BMC Chem. 2023;17:34. doi: 10.1186/s13065-023-00946-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jaworska K., Gupta S., Durda K., Muszyńska M., Sukiennicki G., Jaworowska E., Grodzki T., Sulikowski M., Woloszczyk P., Wójcik J., et al. A Low Selenium Level Is Associated with Lung and Laryngeal Cancers. PLoS ONE. 2013;8:e59051. doi: 10.1371/annotation/f777aaec-b6b8-4480-9cce-18e0f1b8e5d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Giamougiannis P., Martin-Hirsch P.L., Martin F.L. The evolving role of MUC16 (CA125) in the transformation of ovarian cells and the progression of neoplasia. Carcinogen. 2021;42:327–343. doi: 10.1093/carcin/bgab010. [DOI] [PubMed] [Google Scholar]
- 51.Zhang M., Cheng S., Jin Y., Zhao Y., Wang Y. Roles of CA125 in diagnosis, prediction, and oncogenesis of ovarian cancer. Biochim. Et Biophys. Acta (BBA)—Rev. Cancer. 2021;1875:188503. doi: 10.1016/j.bbcan.2021.188503. [DOI] [PubMed] [Google Scholar]
- 52.Dall’era M. Liquid biomarkers in active surveillance. World J. Urol. 2022;40:21–26. doi: 10.1007/s00345-021-03609-5. [DOI] [PubMed] [Google Scholar]
- 53.Augustine T.N. The aegis: Platelets as biomarkers of tumor progression. Biomark. Med. 2020;14:573–585. doi: 10.2217/bmm-2019-0514. [DOI] [PubMed] [Google Scholar]
- 54.Campos-Carrillo A., Weitzel J.N., Sahoo P., Rockne R., Mokhnatkin J.V., Murtaza M., Gray S.W., Goetz L., Goel A., Schork N., et al. Circulating tumor DNA as an early cancer detection tool. Pharmacol. Ther. 2020;207:107458. doi: 10.1016/j.pharmthera.2019.107458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Demircan K., Bengtsson Y., Sun Q., Brange A., Vallon-Christersson J., Rijntjes E., Malmberg M., Saal L.H., Rydén L., Borg Å., et al. Serum selenium, selenoprotein P and glutathione peroxidase 3 as predictors of mortality and recurrence following breast cancer diagnosis: A multicentre cohort study. Redox Biol. 2021;47:102145. doi: 10.1016/j.redox.2021.102145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chang C., Worley B.L., Phaëton R., Hempel N. Extracellular Glutathione Peroxidase GPx3 and Its Role in Cancer. Cancers. 2020;12:2197. doi: 10.3390/cancers12082197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Selenius M., Rundlöf A.-K., Olm E., Fernandes A.P., Björnstedt M. Selenium and the Selenoprotein Thioredoxin Reductase in the Prevention, Treatment and Diagnostics of Cancer. Antioxid. Redox Signal. 2010;12:867–880. doi: 10.1089/ars.2009.2884. [DOI] [PubMed] [Google Scholar]
- 58.Prasad A.S., Beck F.W.J., Snell D.C., Kucuk O. Zinc in Cancer Prevention. Nutr. Cancer. 2009;61:879–887. doi: 10.1080/01635580903285122. [DOI] [PubMed] [Google Scholar]
- 59.Ho E. Zinc deficiency, DNA damage and cancer risk. J. Nutr. Biochem. 2004;15:572–578. doi: 10.1016/j.jnutbio.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 60.Lelièvre P., Sancey L., Coll J.-L., Deniaud A., Busser B. The Multifaceted Roles of Copper in Cancer: A Trace Metal Element with Dysregulated Metabolism, but also a Target or a Bullet for Therapy. Cancers. 2020;12:3594. doi: 10.3390/cancers12123594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tsvetkov P., Coy S., Petrova B., Dreishpoon M., Verma A., Abdusamad M., Rossen J., Joesch-Cohen L., Humeidi R., Spangler R.D., et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375:1254–1261. doi: 10.1126/science.abf0529. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data supporting the reported results are available from the first author upon request from all interested researchers.