Abstract
Objective
To summarize the recommendations on folate intake and folic acid supplementation and fortification in the periconceptional period, aimed at prevention of neural tube defects (NTD), provided by official health organizations in different countries worldwide and WHO.
Design
Information on recommendations for folate and folic acid intake in the periconceptional period was gathered from the websites of official national health organizations of several countries worldwide and from the WHO website.
Setting
WHO, selected developed countries and emerging economies, totalling thirty-six countries worldwide (some European, BRICS, G8, Asian Tiger/Asian Dragon and Australia).
Results
Recommendations differ between countries, although the majority (69·4 %) recommend a healthy diet plus a folic acid supplement of 400 µg/d from preconception (4–12 weeks) until the end of the first trimester of pregnancy (8–12 weeks). The same recommendation is issued by the WHO. Dosages for women at high risk of NTD are up to 4–5 mg/d (for 41·7 % of studied countries). The recommended intake for folate is in the range of 300–400 µg/d for women of childbearing age and 500–600 µg/d for pregnant women in different countries and WHO. Five countries emphasize the importance of a healthy diet rendering supplementation needless. By contrast, five others advise a healthy diet and supplementation plus mandatory fortification. Only one mentions the importance of ensuring an adequate folate status and refers to checking with a health-care provider on the need for supplements.
Conclusions
Different recommendations regarding folate and folic acid, seeking NTD prevention, are available worldwide; however, most countries and WHO focus on a healthy diet and folic acid supplementation of 400 µg/d periconceptionally.
Keywords: Periconceptional period, Neural tube defects, Folate, Folic acid, Official health organizations’ recommendations
Worldwide, more than 10 % of infant deaths secondary to congenital anomalies are caused by nervous system anomalies( 1 ). Neural tube defects (NTD) are the most common major congenital anomalies of the central nervous system, constituting an important public health problem in terms of mortality, morbidity, social cost and human suffering( 2 ). The incidence of NTD ranges from 0·5 to 14 per 1000 live births( 3 ).
The findings obtained from randomized trials by the British Medical Research Council in 1991( 4 ) and by the Hungarian National Institute of Hygiene (a WHO Collaborating Centre for the Community Control of Hereditary Diseases) in 1992( 5 ) about the effect of periconceptional folic acid supplementation on NTD prevention were revolutionary and remain consensual to the present day( 6 ). Furthermore, folic acid seems to prevent other congenital anomalies (cleft lip( 7 , 8 ) and cardiovascular malformations), but the results are not consistent( 2 , 5 , 9 , 10 ). The relationships between maternal folate levels and other pregnancy outcomes, such as placental abruption, placental weight and gestational age, also remain inconclusive( 11 – 14 ).
Curiously, in two observational studies, folic acid supplementation did not show any influence on trends of incidence of NTD( 15 , 16 ), probably because most women started supplementation after the target period( 17 ). In order to prevent NTD, and considering that the neural tube closes by day 28( 18 ) when most women do not know they are pregnant( 2 ), even in planned pregnancies, folic acid supplementation should start before conception( 6 , 19 ). However, estimates indicate that 41 % pregnancies worldwide are unintended( 20 ).
Regarding these promising results provided by the randomized trials, the water-soluble nature of folic acid and the unawareness of adverse effects (apart from neurological complications in people with vitamin B12 deficiency( 6 )), combined with the difficulty of global supplementation in the desired period, fortification of foods with this vitamin has been considered safe( 21 ). This motivated some countries to opt for folic acid fortification of cereal products, namely flour. Nowadays, fortification of flour with folic acid is mandatory in forty-seven countries, in the continents of America and Oceania, in the Middle East and a few in Africa( 22 , 23 ). The effectiveness of mandatory folic acid fortification has been studied in some countries and a significant decline in prevalence of some NTD was observed; for instance, after mandatory folic acid fortification in the USA and Canada, the occurrence of some NTD declined by 28 %( 24 ) and 46 %( 25 ), respectively. Nevertheless, part of this decline can be explained by better prenatal screening and the termination of affected pregnancies( 26 ). Moreover, in a recent review, the authors defended that women of childbearing age may not yet be adequately targeted, while the general population may be over-fortified with folic acid( 27 ). Uncertainties remain over the minimum effective folate intake and status required for NTD prevention, and the safe upper folate level( 27 ). Voluntary fortification is permitted in most European countries, but none has mandatory fortification( 15 ). Recently the EU introduced new rules to regulate voluntary food fortification( 15 ).
After mandatory folic acid fortification, many questions arose regarding individual liberty (to choose fortified or non-fortified foods) and about feasible monitoring of potential hazards of the fortification( 15 ). The possible carcinogenicity of excessive intake of folic acid has been suggested( 2 , 28 ), supported by biologically plausible mechanisms: folate is essential in biological methylation reactions and nucleotide synthesis and impairment of these processes is thought to be involved in cancer development( 15 ). Some researchers suggested that a link could exist between high folic acid intake and cancer incidence( 29 , 30 ), but this is not consensual( 15 , 31 , 32 ).
Recently, a new concern has arisen regarding the potential adverse effects of universal maternal supplementation on adverse pregnancy outcomes, early or later in life, both in the mother and child( 13 , 33 ). Studies have associated maternal folic acid supplementation with increased birth weight( 11 – 14 ), insulin resistance in children( 34 ) and asthma in children( 35 , 36 ).
Overall, current evidence suggests that the relationship between maternal folic acid supplementation and pregnancy outcomes displays a U-shaped association, with adverse effects at both low and high dosages. Official health institutions should deliver clear and evidence-based messages regarding folic acid intake in order to maximize health gains and to minimize adverse effects.
The present study aimed to summarize the recommendations on folate intake and folic acid supplementation and fortification, in the periconceptional period, aimed at NTD risk reduction, provided by official health organizations in different countries worldwide and WHO. This work could constitute a first step of a global discussion about the divergences between recommendations, in order to improve them.
Methods
Data about folate (provided by diet) and folic acid (provided by supplements or fortified foods) recommendations in the periconceptional period were obtained from national health official websites across forty-six countries worldwide and from the WHO website. Eligibility criteria for country selection, defined by the research team, were the following: EU countries (Austria, Belgium, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Luxembourg, Malta, Netherlands, Poland, Portugal, Romania, Slovakia, Slovenia, Spain, Sweden and UK) and some European countries not belonging to the EU (Iceland, Liechtenstein, Norway, Switzerland and Turkey) that were mentioned on the European Commission website, on the web page entitled Public Health – Trustworthy websites on ‘Population groups’ ( 37 ); BRICS countries (Brazil, Russia, India, China and South Africa); Group of Eight (G8) countries (that include some EU and BRICS countries already stated; they are Canada, France, Germany, Italy, Japan, Russia, UK and USA); Asian Tiger/Asian Dragon countries (Hong Kong, Singapore, South Korea and Taiwan); and Australia.
Developed countries and emerging economies (according to the UN, International Monetary Fund, Organisation for Economic Co-operation and Development, and World Bank) in well-defined groups were selected because of the easiest access to data.
The keywords used for searching in each country’s websites were the following: folic acid, folate, pregnancy and nutrition. These terms and the information found in the countries’ websites were translated on Google® Translate, from English to the official languages and vice versa. Ministry of Health websites were chosen for their national coverage of the recommendations. However, for some countries we considered scientific organizations’ websites when mentioned or linked with the Ministry of Health website.
Data extracted from each website covered the following topics: (i) amount of recommended folate intake for women of childbearing age and pregnant women (measured in folate or dietary folate equivalents (DFE)); (ii) recommendation of a healthy diet and folate-rich foods; and (iii) folic acid supplementation for NTD risk reduction (recommended dosage, tolerable upper intake level (UL), dosage for high risk of NTD, when to start and to finish the supplementation). Data about mandatory folic acid fortification were obtained at the European Surveillance of Congenital Anomalies (EUROCAT) website( 23 ). The names of the official entities, the types of information (for health professionals or the general public) and the website addresses were also registered for further validation. Only nine countries (25·0 %) had all desired information available (Australia, Austria, Denmark, Estonia, Finland, Germany, Switzerland, Canada and USA).
Websites were chosen instead of collecting data from scientific articles or sending emails to each national health representative asking for information, because the found reviews( 16 , 38 ) did not include all the desired countries nor all information. We also intended to find which information is available through the World Wide Web, where many people (from health professionals to the general public) consult and follow health recommendations( 39 – 41 ). Information on mandatory fortification was collected only to complete this overview, since we intended to summarize information that could be searched by people and used to influence their behaviours. Mandatory fortification is a governmental decision that potentially affects the whole population.
Emails were sent for all forty-six selected countries in order to obtain data validation or to obtain missing data (data that could be on the website, but had not yet been found). In the validation process, the information was confirmed, corrected and/or completed. Data without official validation were equally considered for results and analysis, after confirmation from the websites on at least three different days.
Data were collected from May until October 2013, and updated in December 2014.
Results
From the forty-six countries initially considered, ten (21·7 %) were excluded – six in the EU (Croatia, Cyprus, Czech Republic, Greek, Lithuania and Slovakia), two European countries not belonging to the EU (Liechtenstein and Turkey), one BRICS country (India) and one Asian Tiger/Asian Dragon country (South Korea) – on the basis of data being unavailable or not found. Thus a final sample of thirty-six countries was obtained: twenty-two in the EU (Austria, Belgium, Bulgaria, Denmark, Estonia, Finland, France, Germany, Hungary, Ireland, Italy, Latvia, Luxembourg, Malta, Netherlands, Poland, Portugal, Romania, Slovenia, Spain, Sweden and UK), three European countries not belonging to the EU (Iceland, Norway and Switzerland), four BRICS countries (Brazil, Russia, China and South Africa), three more G8 countries (the other five were already stated in EU and BRICS; Canada, Japan and USA), three Asian Tiger/Asian Dragon countries (Hong Kong, Singapore and Taiwan) and Australia; see Fig. 1.
Fig. 1.
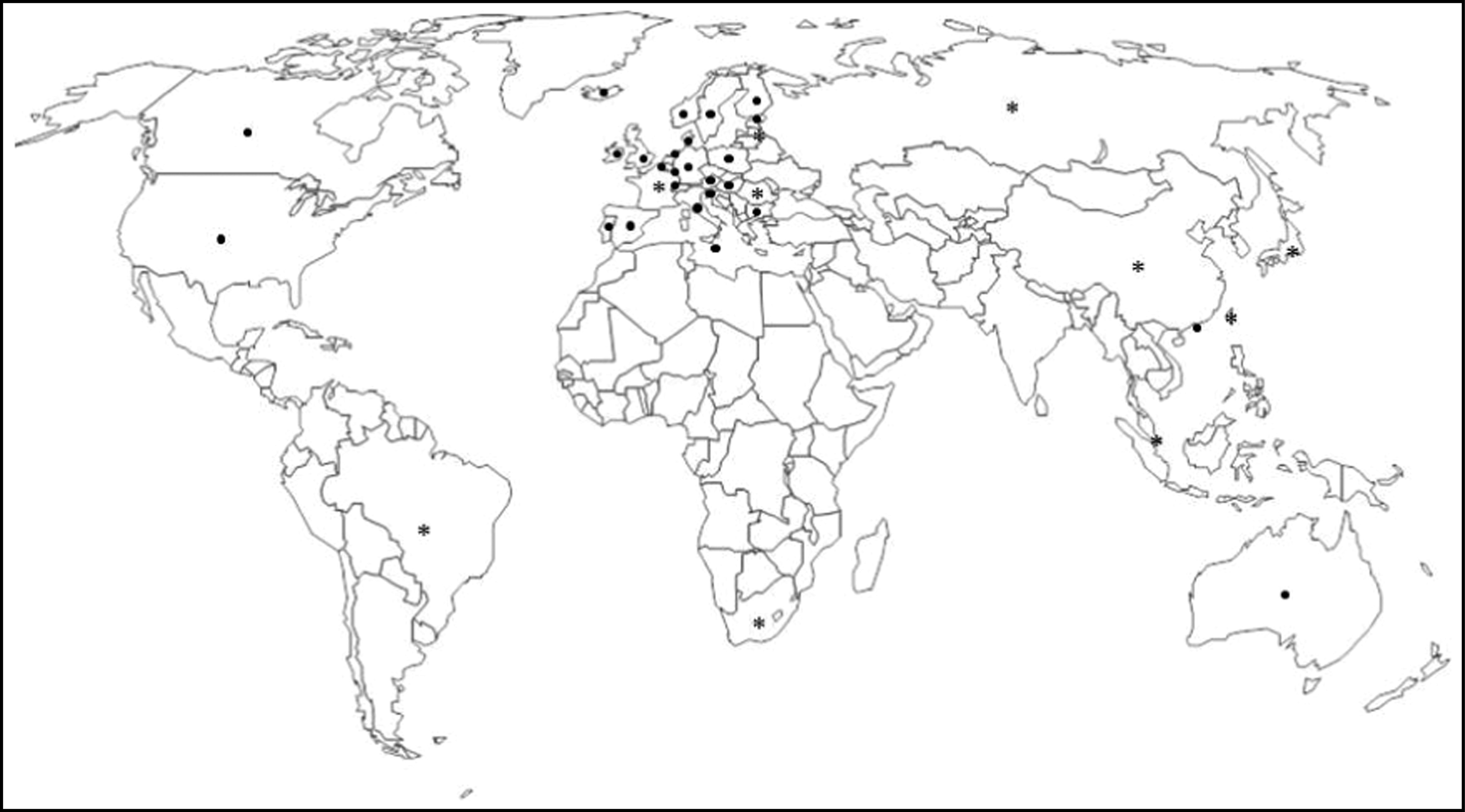
World map showing the thirty-six countries in which recommendations for folate and folic acid intake in the periconceptional period were analysed (●, country with official validation; ∗, country without official validation)
From the thirty-six countries included, twenty-six countries validated the information by email (72·2 %) and ten did not (Brazil, China, France, Japan, Latvia, Romania, Russia, Singapore, South Africa and Taiwan). From the other ten countries without data, only Liechtenstein validated the (missing) information. The WHO did not validate data.
The recommendations for folate and folic acid intake in the periconceptional period are synthesized in Table 1.
Table 1.
Folate and folic acid recommendations for women in the periconceptional period from official websites of national health organizations of some countries worldwide (n 36) and WHO
| Folic acid fortification | Folic acid supplementation for NTD risk reduction | Folate | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Country/entity | Is it mandatory?( 23 ) | Dosage | Begin | End | UL | Dosage for high risk of NTD | Is a healthy diet and folate-rich food recommended? | Recommended folate intake | Recommended DFE intake | Official entities |
| Australia | Yes | 400 µg/d (supplements)/at least 400 µg/d by health-care provider | Woman capable of, or planning, pregnancy at least 4 weeks/1 month before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | Higher dosages, to discuss with physician | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | National Health and Medical Research Council( 61 , 62 ) Food Standards Australia New Zealand( 63 ) |
| Austria | No | 400 µg/d | At least 4 weeks/1 month before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | To discuss with physician | Yes | – | Pregnant: 550 µg/d Women: 300 µg/d | Federal Ministry of Health, Austria – Public Health Portal( 64 ) D-A-CH( 65 ) |
| Belgium – Flanders | No | 400 µg/d | When the contraceptive is stopped | 12 weeks/3 months of pregnancy | 1 mg/d for adults | 4 mg/d | Yes | – | – | Flemish Government – Child and Family Support Centre for Policy Research – Welfare, Health and Family( 66 , 67 ) |
| Brazil* | Yes | 5 mg/d especially in women with history of NTD | 8–12 weeks/2–3 months before conception | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes | – | – | Ministry of Health( 68 , 69 ) |
| Bulgaria | No | 400 µg/d | At least 12 weeks/3 months before conception | 12 weeks/3 months of pregnancy | – | – | Yes | – | – | Official Bulgarian Patient Organization ‘Conception’( 70 ) |
| Canada | Yes | 400 µg/d (supplements) | Before conception by all women capable of becoming pregnant/at least 12 weeks/3 months before conception | Throughout pregnancy or early pregnancy/12 weeks/3 months of pregnancy | 1 mg/d for adults | 4–5 mg/d (higher dosages) | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | Health Canada( 71 – 74 ) |
| China* | No | 400 µg/d | At least 4 weeks/3 months before conception | Throughout the pregnancy | 1 mg/d for adults | – | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | E-health – Shanghai Jing’an District Health Promotion Board – Ministry of Health( 75 ) Chinese Nutrition Society( 76 ) |
| Denmark | No | 400 µg/d | When you start thinking about getting pregnant/for those who plan to become pregnant | 12 weeks/3 months after conception or 8 weeks/2 months (for high risk of NTD) | Not exceed recommendation | 5 mg prescribe by the physician | Yes | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | Danish Health and Medicines Authority( 77 , 78 ) Nordic Nutrition Recommendations( 79 ) |
| Estonia | No | 400 µg/d | At least 12 weeks/3 months before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | 4 mg/d | Yes | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | National Institute for Health Development( 80 , 81 ) Health Information Network( 82 ) Treatment Guide( 83 ) |
| Finland | No | 400 µg/d for woman with an unhealthy diet† | When the contraceptive is stopped/for those who plan to become pregnant | 12 weeks/3 months of pregnancy | 1 mg/d for adults | 4 mg/d | Yes† | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | Ministry of Social Affairs and Health( 84 ) Nordic Nutrition Recommendations( 79 ) |
| France* | No | 400 µg/d may be prescribed by health-care provider‡ | At pregnancy project | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes‡ | – | – | National Institute for Prevention and Health Education( 85 – 87 ) National Nutrition and Health Program( 88 ) |
| Germany | No | 400 µg/d | At least 4 weeks/1 month before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | To discuss with physician | Yes | – | Pregnant: 550 µg/d Women: 300 µg/d | Federal Centre for Health Education – Family Planning( 89 ) German Nutrition Society/D-A-CH( 65 ) |
| Hong Kong | No | 400 µg/d | 4 weeks/1 month before conception/when planning for pregnancy | 12 weeks/3 months of pregnancy (throughout pregnancy in women carrying thalassaemia trait) | – | – | Yes | – | – | Department of Health( 90 ) The Hong Kong College of Obstetricians and Gynaecologist( 91 ) |
| Hungary | No | 400 µg/d | At least 4 weeks/1 month before conception | 12 weeks/3 months of pregnancy | – | – | Yes | – | – | National Centre for Food Safety and Nutrition( 92 ) |
| Iceland | No | 400 µg/d | At least 4 weeks before conception/all women able to get pregnant | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | Directorate of Health( 93 , 94 ) Nordic Nutrition Recommendations( 79 ) |
| Ireland | No | 400 µg/d | 12 weeks/3 months before conception/all women of childbearing age | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes | – | – | Health Service Executive( 95 ) |
| Italy | No | 400 µg/d | Before conception | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes | – | – | Ministry of Health( 96 , 97 ) |
| Japan* | No | 400 µg/d | 12 weeks/3 months before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | – | Yes | – | – | Ministry of Health, Labour and Welfare( 98 ) |
| Latvia* | No | 400 µg/d | While trying to get pregnant | 12 weeks/3 months of pregnancy | – | – | Yes | – | – | Latvian Health Portal( 99 ) |
| Luxembourg | No | 400 µg/d | 4 weeks/1 month before conception | 12 weeks/3 months of pregnancy | – | Higher dosages (>400 µg/d) | Yes | – | – | Ministry of Health( 100 ) |
| Malta | No | 400 µg/d | Women who are planning a pregnancy/prior to conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | – | Yes | – | Women: 400 µg/d | Ministry for Health( 101 ) |
| Netherlands | No | 400 or 500 µg/d | 4 weeks/1 month before conception/when there is a chance of becoming pregnant | 10 weeks/2 months of pregnancy | – | Higher dosages (>400 or 500 µg/d) | Yes | – | – | National Institute for Public Health and Environment – Ministry of Health, Welfare and Sport( 102 ) Nutrition Centre( 103 ) |
| Norway | No | 400 µg/d | 4 weeks/1 month before conception/women who are planning to conceive | 8–12 weeks/2–3 months of pregnancy | – | 4 mg/d | Yes | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | National Institute of Public Health( 104 ) Nordic Nutrition Recommendations( 79 ) |
| Poland | No | 400 µg/d | While trying to get pregnant | 12 weeks/3 months of pregnancy | – | – | Yes | – | – | Ministry of Health( 105 ) |
| Portugal | No | – | At least 8 weeks/2 months before stopping using contraception | – | – | – | Yes | – | – | Ministry of Health( 106 ) |
| Romania* | No | 400 µg/d | – | – | 1 mg/d for adults | 5 mg/d | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | Ministry of Health – Romanian Nutrition Society( 107 ) |
| Russia* | No | – | – | – | – | – | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | Ministry of Health of the Russian Federation( 108 ) |
| Singapore* | No | Check with health-care provider‡ | Before conception/when trying to become pregnant | 12 weeks/3 months of pregnancy | – | – | Yes‡ | – | Pregnant: 600 µg/d Women: 400 µg/d | Health Promotion Board – Singapore Government( 109 ) |
| Slovenia | No | – | – | – | – | – | Yes | – | Pregnant, 2nd and 3rd trimester: 600 µg/d Women: 400 µg/d | Institute of Public Health of the Republic of Slovenia( 110 ) Ministry of Health( 111 ) |
| South Africa* | Yes | Recommended by health-care provider | When planning for pregnancy | – | Not exceed recommendation | – | Yes | – | – | The National Department of Health( 112 ) Association for Dietetics in South Africa( 113 ) |
| Spain | No | 400 µg/d | 12 weeks/3 months before stopping contraception/4–8 weeks/1–2 months before conception | 12 weeks/3 months of pregnancy | 1 mg/d for adults | 5 mg/d | Yes | – | – | Ministry of Health, Social Services and Equality – National Health System( 114 , 115 ) |
| Sweden | No | 400 µg/d except for women with high folate intake§ | 4 weeks/1 month before conception/woman who may get pregnant | 12 weeks/3 months of pregnancy | 1 mg/d for adults | – | Yes§ | Pregnant: 500 µg/d Women 18–30 years or childbearing age: 400 µg/d Women: 300 µg/d | – | National Food Agency( 116 , 117 ) Nordic Nutrition Recommendations( 79 ) European Food Safety Authority( 15 ) |
| Switzerland | No | 400 µg/d | At least 4 weeks/1 month before conception/women who wish or may fall pregnant | 12 weeks/3 months of pregnancy | 1 mg/d for adults | To discuss with physician/higher dosages (>400 µg/d) | Yes | – | Pregnant: 550 µg/d Women: 300 µg/d Women: 400 µg/d | Swiss Society of Nutrition( 118 )/D-A-CH( 65 ) Swiss Federal Office of Public Health( 119 ) |
| Taiwan* | No | –‡ | – | – | 1 mg/d for adults | – | Yes‡ | Pregnant: 600 µg/d Women: 400 µg/d | – | Health Promotion Administration, Ministry of Health and Welfare( 120 ) Food and Drug Administration( 121 , 122 ) |
| UK | No | 400 µg/d | While trying to get pregnant | 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes | – | – | National Health Service( 123 ) |
| USA | Yes | 400 µg/d (supplements or fortified foods) or 400–800 µg/d (supplements) to discuss with physician | Before conception by all women capable of becoming pregnant/at least 4 weeks/1 month before conception (for high risk of NTD) | 12 weeks/3 months of pregnancy(for high risk of NTD) or throughout pregnancy | 1 mg/d or adults | 4 mg/d | Yes | – | Pregnant: 600 µg/d Women: 400 µg/d | Institute of Medicine of the National Academies( 124 ) Centers for Disease Control and Prevention( 125 , 126 ) Women’s health – Department of Health and Human Services( 127 ) |
| WHO* | – | 400 µg/d | As early as possible (periconceptionally) or women trying to conceive/8 weeks/2 months before the planned pregnancy | Throughout pregnancy or 12 weeks/3 months of pregnancy | – | 5 mg/d | Yes (high risk of NTD) | – | Pregnant: 600 µg/d Women: 400 µg/d | WHO( 18 , 19 , 28 , 42 ) |
NTD, neural tube defects; UL, tolerable upper intake level; DFE, dietary folate equivalents; D-A-CH, Deutschland–Austria–Confoederatio Helvetica (reference values for nutrient intake by German Nutrition Society, Austrian Nutrition Society, Swiss Society for Nutrition Research and Swiss Society for Nutrition). – means that no information was available.
Without official validation.
‘Unhealthy diet means having less than 400 µg of folate.’
‘A varied diet may be enough’ (France)/‘Keep up your folate status by eating a healthy diet’ (Singapore)/‘A balanced diet is assumed to be enough to achieve the recommendations’ (Taiwan).
‘For women with a high intake of folate-rich foods, such as vegetarians and vegans eating large quantities of pulses and vegetables, folate intake from food may happen to be enough. For them there is no need for extra folic acid supplementation’.
A healthy diet and/or folate-rich food recommendations were issued by all countries (100·0 %), but the WHO website only mentioned it for women with high risk of NTD. Interestingly, five countries (13·9 %; Finland, France, Sweden, Singapore and Taiwan) considered that a healthy diet containing adequate amounts of folate may be enough, with no need for supplementation. Three countries (Hong Kong, Slovenia and Sweden) published e-leaflets for the general public with complete and detailed information about healthy diet during pregnancy, consumption of folate-rich foods, cooking methods and quantities. Eight more countries (Austria, Denmark, Estonia, France, Iceland, Luxembourg, Taiwan and Poland) also published e-leaflets with information for pregnant women including healthy diet. Some others mentioned it directly on the website.
Concerning recommended folate intake (and estimated average requirements) for women of childbearing age and pregnant women, respectively, we identified three main different recommendations: (i) Dietary Reference Intakes from the US Institute of Medicine (IOM; followed by 22·2 % of countries), namely 400 µg/d and 600 µg/d (320 µg/d and 520 µg/d); (ii) Nordic Nutrition Recommendations from the Nordic Council of Ministers (NNR; followed by 16·7 % of countries), namely 400 µg/d and 500 µg/d (200 µg/d); and (iii) reference values for the nutrient intake from the dominant states of the German language (Deutschland–Austria–Confoederatio Helvetica (D-A-CH); followed by 8·3 % of countries), namely 300 µg/d and 550 µg/d (220 µg/d and 420 µg/d). WHO adopted the folate recommendations from the IOM( 28 , 42 ). About half of the countries (52·8 %) did not have any references regarding recommended folate intake through diet. For Switzerland, different values were available in the two sources consulted (D-A-CH and those referred by the Swiss Federal Office of Public Health). Slovenia followed D-A-CH recommendations, with high values only after the first trimester. This appears more practicable as physiologically women have higher energetic needs in the second and third trimesters( 43 ), notwithstanding the role of folate after the first trimester is not completely established. It is also apparent that NNR presented the smallest increase from preconception to pregnancy, possibly more attainable. The highest values from the IOM seem less achievable than those from D-A-CH or NNR.
Regarding folic acid supplementation for NTD prevention, the majority (n 33, 91·7 %) of the analysed countries have some information on their national health official websites about this topic. Most countries (80·6 %) recommend a folic acid supplement of 400 µg/d, two countries state that the dosage should be recommended by a health-care provider (without mentioning any dosage) and one country recommends 5 mg/d. Concerning the time for initiating the supplementation, 33·3 % of countries state at least 4 weeks before conception, as settled by D-A-CH recommendations; 27·8 % of countries state that ‘supplementation should begin when planning pregnancy’ or ‘when there is a chance to become pregnant/capable of becoming pregnant’, as stressed by the IOM recommendations; 13·9 % of countries mention at least 12 weeks before pregnancy; two countries refer to 4–8 weeks or 8–12 weeks before conception; two advocate to start when contraception is stopped; and one country recommends at least 8 weeks before stopping contraception. Most countries (75·0 %) consider that supplementation should be maintained until the end of the first trimester (12 weeks) or almost (10 or 8–12 weeks), but three countries (Canada, China and USA) recommend supplementation until the end of pregnancy.
On the subject of the UL, 44·4 % of countries and two entities (D-A-CH and IOM) state 1 mg/d and two countries appeal for not exceeding the recommendations without mentioning the UL. Finally, the dosages recommended for women with high risk of NTD are 5 mg/d (adopted by 27·8 % of countries and by WHO recommendations) or 4 mg/d (considered by 13·9 % of countries). Some countries suggest these dosages should be discussed with a physician (8·3 % of countries and D-A-CH recommendations) and two countries refer to only ‘higher dosages’.
Although the observed recommendations differ between countries, the recommendation for folic acid of 400 µg/d before conception until the first trimester of pregnancy is common for most countries (69·4 %) and WHO.
Countries partially out of this common recommendation are those that recommended 5 mg/d (Brazil) or supplementation throughout pregnancy (Canada, China and USA) or that did not show the dosage, referring to check with a health-care provider on the need for taking supplements and to ensure an adequate folate status (Singapore). This resembles an attempt to personalize the intervention.
Some countries had incomplete information about supplementation as follows: dosage was missing in Portugal; time for initiation was absent in Romania; the end time was missing in South Africa, Portugal and Romania; and supplementation was not mentioned at all in Russia, Slovenia and Taiwan.
Taiwan’s website referred that the best method to ensure sufficient folate for fetal development is to consume foods high in folate on a frequent basis, giving examples. This perspective contrasted with that of countries with a focus mainly on supplementation, for example Netherlands and USA.
From the analysed countries, five had mandatory fortification (Australia, Brazil, Canada, South Africa and USA). All of these also advised folic acid supplementation.
Given the diversity of the recommendations for folate from international entities (from D-A-CH, NNR, IOM and WHO), we opted to build a diagram overlapping different recommendations with gestational age, periconceptional period and neurulation period, as shown in Fig. 2.
Fig. 2.
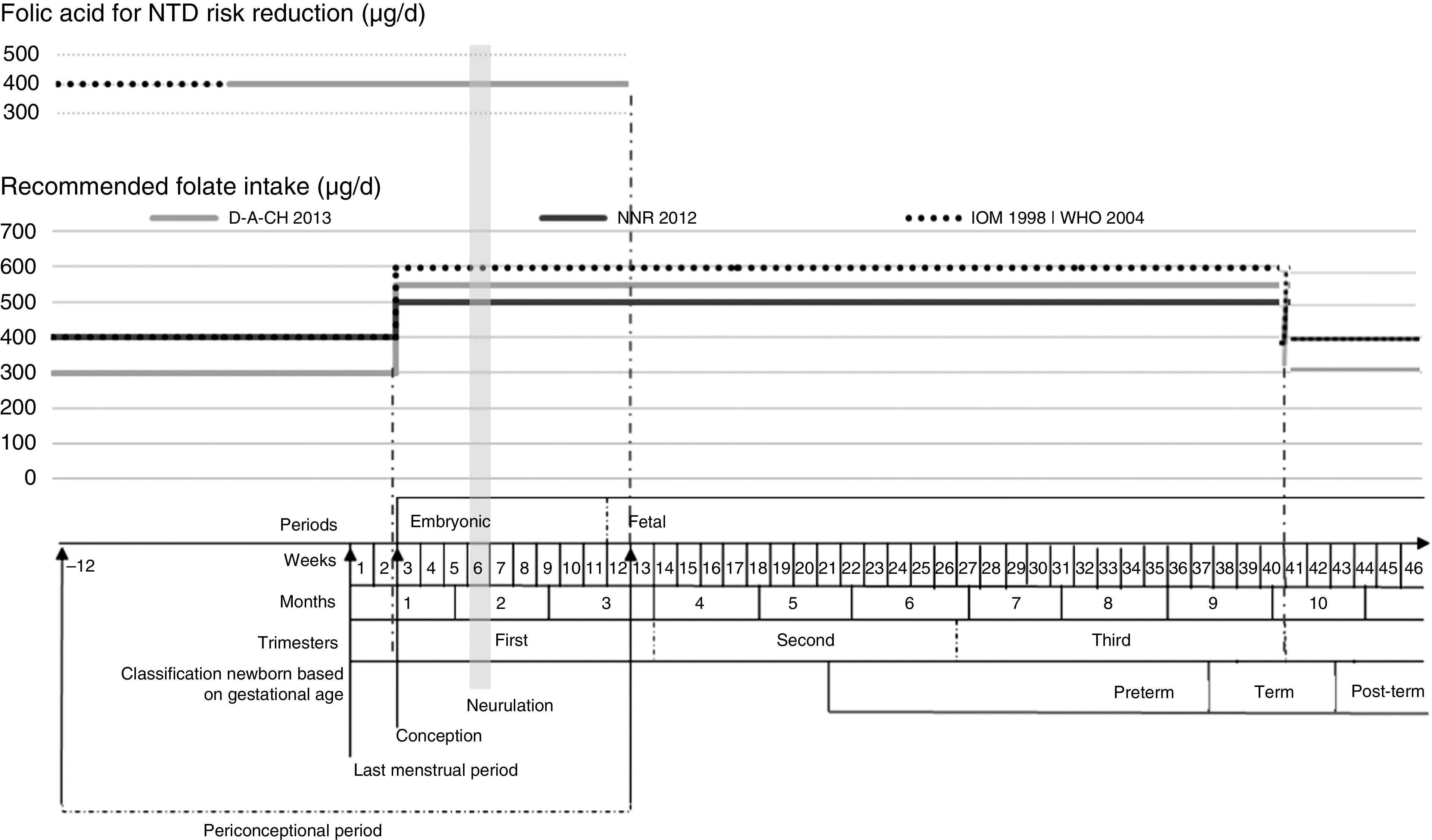
Recommended folate intake and recommended folic acid supplementation for prevention of NTD during the periconceptional period (NTD, neural tube defects; D-A-CH, Deutschland–Austria–Confoederatio Helvetica (reference values for nutrient intake by the German Nutrition Society, Austrian Nutrition Society, Swiss Society for Nutrition Research and Swiss Society for Nutrition); NNR, Nordic Nutrition Recommendations (by Denmark, Finland, Iceland, Norway and Sweden); IOM, Institute of Medicine, Dietary Reference Intakes; WHO, Vitamin and Mineral Requirements in Human Nutrition ( 42 )). Definition of periconceptional period adapted from Steegers-Theunissen et al.( 44 )
Discussion
The association between adequate maternal folate levels during the periconceptional period and the reduction in congenital anomalies, especially NTD, is well established. However, achieving an adequate folate status in the neurulation period is a challenge worldwide, according to prior studies( 2 , 6 , 44 ). To fill this gap, the WHO and many national health organizations have been developing recommendations related to folic acid supplementation and a folate-rich diet. Some countries have also implemented mandatory folic acid fortification.
In public health it is indispensable to communicate widespread clear and unique messages among health professionals and the general public, once the general public is becoming more interested in a topic, to judge the information provided by health professionals.
The question of folate adequacy seeking NTD prevention is very interesting, because the critical period for that adequacy is the first six gestational weeks( 2 ) when the majority of women do not know they are pregnant, even in planned pregnancies (exceptions would be medically assisted pregnancies). Some authors advocate that, ideally, a correct diet would supply adequate intakes, but this is not the reality for all( 17 ). Supplementation could be a solution for this deficiency, but it is not always effective because women are often unaware of their pregnancies during the target period( 15 , 16 ). Food fortification with folic acid appears a better solution, providing adequate intakes. So, recommendations should have a clear message regarding at least two of the three possibilities for an adequate folate intake: diet, fortification and supplements. Mandatory fortification is restricted to countries that opt for this measure. Others may have voluntary fortification and recent EU legislation was created in order to regulate this kind of fortification( 15 ).
Nowadays, the World Wide Web is available for the majority of the population in many countries worldwide. This is an excellent vehicle for giving messages to health professionals and the rest of the population. For this reason, this source of information was inspected. Initially, it was intended to obtain information for forty-six countries, but for ten of them it was impossible to reach any information on official websites. For the other thirty-six countries diverse information was obtained. Notwithstanding that differences should exist among countries regarding different dietary patterns and different disease prevalences, probably there is not a clear rational justification for the disparities observed between countries.
Folate intake recommendations differ between international entities: D-A-CH, NNR, Scientific Committee on Food, IOM( 2 , 16 , 38 , 45 , 46 ) and WHO( 28 , 42 ). A huge amount of work should be done regarding dietary advice. Public health efforts should incorporate practical advice on storage and cooking to increase folate intake, helping to optimize folate status( 47 ), as well as recommendations on dietary pattern( 48 ). A prudent dietary pattern, even in the era of fortification, may decrease the risk of NTD and some heart defects in non-users of folic acid( 48 ). In addition to folate, other micronutrients may decrease the risk of NTD occurrence( 49 ) and once again promoting a healthy diet has several benefits. Furthermore, the practice of a Western diet by mothers increases the risk of offspring with a cleft lip or cleft palate approximately twofold( 50 ). A high preconceptional intake of nutrients predominantly present in fruits and vegetables reduces the risk of offspring affected by orofacial cleft( 51 ). Fast food may increase the risk of NTD( 52 ).
With the present study we drew a roadmap of the recommendations about folate and folic acid intake before and during pregnancy in several countries worldwide (developed countries and emerging economies) and we also included WHO recommendations because it is plausible that many other countries consider them. The recommendations differ between countries, although the majority (66·7 %) recommend folic acid supplements of 400 µg/d, in the periconceptional period, combined with a folate-rich diet. A small number of countries emphasize the importance of a healthy diet naturally rich in folate with no need for folic acid supplementation. According to some authors( 48 ), this advice should be the main in all recommendations for NTD prevention( 47 ), namely because a healthy diet also supplies other micronutrients that may decrease the risk of NTD occurrence( 49 ). At the other end of the spectrum, some countries advise supplementation and have mandatory folic acid fortification.
It seems to be consensual that women at high risk of NTD benefit from higher dosages (4–5 mg/d) of folic acid supplementation( 4 , 26 , 53 , 54 ). However, not all cases of NTD are preventable by increasing the folic acid intake( 55 ). Several other lifestyle and environmental factors as well as genetic variations may have an influence, possibly by affecting one-carbon metabolism and thus epigenetic events( 27 ). Known risk factors for NTD are genetic (about 60 %( 2 )), chronic alcohol intake( 54 ), smoking( 53 ), use of certain drugs (e.g. carbamazepine, phenobarbital, methotrexate)( 53 , 54 ), some pathologies (e.g. epilepsy( 54 ), malabsorption disorders( 53 , 54 ), obesity( 26 , 53 , 54 ), pre-gestational diabetes( 26 , 53 , 54 )) and personal or family history of NTD( 54 ).
The current study found that the designations mentioned as the adequate period for supplementation differ between countries. It can be considered that the more adequate are the ones that include the general designation ‘before conception’ and that refer to ‘planning plus chance of becoming pregnant’. The designation ‘reproductive age’ is completely broad and if supplementation is suggested these women could maintain supplementation for large periods of time, unnecessarily.
Apart from recommendations about supplementation, some countries have introduced fortification and others not. Surprisingly, one of the countries with mandatory fortification (Brazil) recommends supplementation with high doses of folic acid (5 mg/d) for all women and not only for those at high risk of NTD. But no evidence sustains the recommendation of such doses for all pregnant women( 15 , 45 ). Moreover, nowadays the scientific community is concerned about potential adverse effects of folic acid fortification and supplementation( 2 , 15 , 28 – 32 ) and adverse pregnancy outcomes, early or later in life, both in the mother and child, such as increased birth weight( 11 – 14 ), insulin resistance in children( 34 ) and asthma in children( 35 , 36 ).
Although carcinogenicity induced by high doses of folic acid is not completely established, this issue should continue to be studied, since it constitutes a legitimate public health concern and needs careful monitoring( 56 ). It is known that through exposure to mandatory food fortification and vitamin supplement use, a large number of people have an unprecedented high folic acid intake( 57 ) and that the capacity of the body to convert folic acid to 5-methyltetrahydrofolate is limited( 58 ). An inverse association was also found between the presence of unmetabolized folic acid in plasma and natural killer cell cytotoxicity, but further studies on immune function and health are needed( 57 ). Given the possibility that excessive folic acid exposure may relate to cancer risk, monitoring the long-term effect should be warranted( 56 , 59 ) especially considering that the vast majority of the population is not at risk of NTD( 59 ).
In some countries, an effort to personalize intervention was observed. Some delegated to the physician or the health provider the decision for supplementation and/or dosage. In Singapore the importance of folate status, to be achieved by diet, was mentioned.
It is not easy to know the real intake of folic acid as supplements in different countries, because many scientific studies limit this kind of information, providing only the proportion of supplemented women without reference to the dose( 17 , 60 ). In order to measure accurately the real impact of such recommendations, systematic health registries are imperative. For instance, regarding congenital anomalies, it would be necessary to compile information regarding miscarriages, medical abortions, stillbirths and neonates with congenital information, information not always easy to aggregate. This is a critical issue in the comparison among countries with different levels of accuracy in their registries.
Strengths and weaknesses
As advantages of the present study we can point out its novelty, considering that all information was searched from official national health websites, a source of information that is widely used and accessible to health professionals and the general public. The previous studies found in the literature included fewer countries and did not analyse data available on the Internet( 16 , 38 ). Most data were officially validated, by email, ensuring a better accuracy. The present study allowed clustering of recommendations to check similarities and disparities within and between countries, and missing information. Countries that do not disseminate clear messages were also identified.
The study is limited by the lack of information about excluded countries, other countries worldwide and by having some countries without official validation, within which we cannot assure that other recommendations are emanated. The methodology underlying each recommendation was not analysed, nor was compliance or the impact of the recommendations in each country.
Public health implications
The role of folate and folic acid during pregnancy being unquestionable, the present study provides a huge amount of structured information about worldwide folate and folic acid recommendations in the periconceptional period and can form a basis for future discussions on these issues. Many questions arise at the end of the study, as follows. What are the reasons behind the different recommendations? Is there any association between recommendations and the epidemiology of NTD in the country? How can we optimize the recommendations, taking into account different sources (healthy diet, fortified foods and supplements)? How can we deal with unplanned pregnancies, other than to recommend supplementation during the entire childbearing age? Are fortified foods really a good solution when the entire population is considered (potential adverse effects)? How to reduce adverse effects in the mother, child and overall population? Is supplementation needed when women have adequate folate status before pregnancy (and considering adverse effects)? Will the future hold a place for personalized recommendations, namely based on blood analyses of folate status (serum and erythrocyte)?
Conclusions
We conclude that the recommendation more frequently used worldwide, from analysed countries and the WHO, about folic acid supplementation in the periconceptional period is 400 µg/d. The recommendation for folate intake is in the range of 300–400 µg/d for women of childbearing age and 500–600 µg/d for pregnant women.
In some countries there are clear indications for the use of higher dosages among women with higher risk of congenital anomalies. The recommendation of a healthy diet, naturally rich in folate, is unanimous. However, big disparities were seen for the recommendations between some countries: some recommend supplementation and a healthy diet plus mandatory fortification; in contrast, some recommend only a healthy diet.
Accurate and ideally evidence-based recommendations are needed regarding folate and folic acid intake during the periconceptional period.
Acknowledgements
Acknowledgements: The authors are grateful to the Ministries of Health of the following twenty-seven countries that validated data available in the websites: Australia, Austria, Belgium, Bulgaria, Canada, Denmark, Estonia, Finland, Germany, Hong Kong, Hungary, Iceland, Ireland, Italy, Liechtenstein, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Slovenia, Spain, Sweden, Switzerland, UK and USA. The authors are grateful to Yu-shuo Kuo who helped with research on Asian websites; and to Jessica Sheppard who helped with English revision. Financial support: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. The views expressed in this article are the authors’ own and do not reflect an official position of any institution. Conflict of interest: None. Authorship: E.P. and S.G. were responsible for designing the study. S.G. was responsible for data collection, the contacts with national entities and the statistical analyses. S.G. wrote the manuscript jointly with E.P. and C.L. All three authors contributed to the interpretation of the findings and approved the contents of this manuscript.
References
- 1. Rosano A, Botto LD, Botting B et al. (2000) Infant mortality and congenital anomalies from 1950 to 1994: an international perspective. J Epidemiol Community Health 54, 660–666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Institute of Medicine (1998) Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press. [PubMed] [Google Scholar]
- 3. Herrmann W & Obeid R (2011) The mandatory fortification of staple foods with folic acid: a current controversy in Germany. Dtsch Arztebl Int 108, 249–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. MRC Vitamin Study Research Group (1991) Prevention of neural tube defects: results of the Medical Research Council Vitamin Study. Lancet 338, 131–137. [PubMed] [Google Scholar]
- 5. Czeizel AE & Dudas I (1992) Prevention of the first occurrence of neural-tube defects by periconceptional vitamin supplementation. N Engl J Med 327, 1832–1835. [DOI] [PubMed] [Google Scholar]
- 6. Institute of Medicine (2006) Dietary Reference Intakes: The Essential Guide to Nutrient Requirements [JJ Otten, JP Hellwig and LD Meyers, editors]. Washington, DC: The National Academies Press.
- 7. Wilcox AJ, Lie RT, Solvoll K et al. (2007) Folic acid supplements and risk of facial clefts: national population based case–control study. BMJ 334, 464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kelly D, O’Dowd T & Reulbach U (2012) Use of folic acid supplements and risk of cleft lip and palate in infants: a population-based cohort study. Br J Gen Pract 62, e466–e472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. De-Regil LM, Fernandez-Gaxiola AC, Dowswell T et al. (2010) Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst Rev issue 10, CD007950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Rozendaal AM, van Essen AJ, Te Meerman GJ et al. (2013) Periconceptional folic acid associated with an increased risk of oral clefts relative to non-folate related malformations in the Northern Netherlands: a population based case–control study. Eur J Epidemiol 28, 875–887. [DOI] [PubMed] [Google Scholar]
- 11. Charles DH, Ness AR, Campbell D et al. (2005) Folic acid supplements in pregnancy and birth outcome: re-analysis of a large randomised controlled trial and update of Cochrane review. Paediatr Perinat Epidemiol 19, 112–124. [DOI] [PubMed] [Google Scholar]
- 12. de Benoist B (2008) Conclusions of a WHO Technical Consultation on folate and vitamin B12 deficiencies. Food Nutr Bull 29, 2 Suppl, S238–S244. [DOI] [PubMed] [Google Scholar]
- 13. Fekete K, Berti C, Trovato M et al. (2012) Effect of folate intake on health outcomes in pregnancy: a systematic review and meta-analysis on birth weight, placental weight and length of gestation. Nutr J 11, 75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Lassi ZS, Salam RA, Haider BA et al. (2013) Folic acid supplementation during pregnancy for maternal health and pregnancy outcomes. Cochrane Database Syst Rev 3, CD006896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. European Food Safety Authority (2009) Folic acid: an update on scientific developments. http://www.efsa.europa.eu/fr/supporting/doc/2e.pdf (accessed March 2015).
- 16. Botto LD, Lisi A, Robert-Gnansia E et al. (2005) International retrospective cohort study of neural tube defects in relation to folic acid recommendations: are the recommendations working? BMJ 330, 571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Pinto E, Barros H & dos Santos Silva I (2009) Dietary intake and nutritional adequacy prior to conception and during pregnancy: a follow-up study in the north of Portugal. Public Health Nutr 12, 922–931. [DOI] [PubMed] [Google Scholar]
- 18. World Health Organization (2012) WHO Guidelines Approved by the Guidelines Review Committee. Guideline: Daily Iron and Folic Acid Supplementation in Pregnant Women. Geneva: WHO. [PubMed] [Google Scholar]
- 19. World Health Organization (2006) Prevention of Neural Tube Defects. Standards for Maternal and Neonatal Care. Geneva: WHO, Department of Making Pregnancy Safer. [Google Scholar]
- 20. Singh S, Sedgh G & Hussain R (2010) Unintended pregnancy: worldwide levels, trends, and outcomes. Stud Fam Plann 41, 241–250. [DOI] [PubMed] [Google Scholar]
- 21. Butterworth CE Jr & Tamura T (1989) Folic acid safety and toxicity: a brief review. Am J Clin Nutr 50, 353–358. [DOI] [PubMed] [Google Scholar]
- 22. United Nations (2012) Composition of macro geographical (continental) regions, geographical sub-regions, and selected economic and other groupings.http://unstats.un.org/unsd/methods/m49/m49regin.htm (accessed October 2013).
- 23. EUROCAT (2013) World map of countries having mandatory fortification of food with folic acid. http://www.eurocat-network.eu/content/EUROCAT-Folic-Acid-Map.pdf (accessed September 2013).
- 24. Williams LJ, Rasmussen SA, Flores A et al. (2005) Decline in the prevalence of spina bifida and anencephaly by race/ethnicity: 1995–2002. Pediatrics 116, 580–586. [DOI] [PubMed] [Google Scholar]
- 25. De Wals P, Tairou F, Van Allen MI et al. (2007) Reduction in neural-tube defects after folic acid fortification in Canada. N Engl J Med 357, 135–142. [DOI] [PubMed] [Google Scholar]
- 26. Kondo A, Kamihira O & Ozawa H (2009) Neural tube defects: prevalence, etiology and prevention. Int J Urol 16, 49–57. [DOI] [PubMed] [Google Scholar]
- 27. Osterhues A, Ali NS & Michels KB (2013) The role of folic acid fortification in neural tube defects: a review. Crit Rev Food Sci Nutr 53, 1180–1190. [DOI] [PubMed] [Google Scholar]
- 28. Food and Agriculture Organization of the United Nations & World Health Organization (2001) Human Vitamin and Mineral Requirements. Report of a Joint FAO/WHO Expert Consultation, Bangkok, Thailand. Rome: FAO; available at http://www.fao.org/3/a-y2809e.pdf. [Google Scholar]
- 29. Cole BF, Baron JA, Sandler RS et al. (2007) Folic acid for the prevention of colorectal adenomas: a randomized clinical trial. JAMA 297, 2351–2359. [DOI] [PubMed] [Google Scholar]
- 30. Ebbing M, Bonaa KH, Nygard O et al. (2009) Cancer incidence and mortality after treatment with folic acid and vitamin B12 . JAMA 302, 2119–2126. [DOI] [PubMed] [Google Scholar]
- 31. Kennedy DA, Stern SJ, Moretti M et al. (2011) Folate intake and the risk of colorectal cancer: a systematic review and meta-analysis. Cancer Epidemiol 35, 2–10. [DOI] [PubMed] [Google Scholar]
- 32. Vollset SE, Clarke R, Lewington S et al. (2013) Effects of folic acid supplementation on overall and site-specific cancer incidence during the randomised trials: meta-analyses of data on 50,000 individuals. Lancet 381, 1029–1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. National Center for Biotechnology Information (2013) MeSH – Pregnancy Outcome. http://www.ncbi.nlm.nih.gov/mesh/?term=pregnancy+outcomes (accessed October 2013).
- 34. Yajnik CS, Deshpande SS, Jackson AA et al. (2008) Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: the Pune Maternal Nutrition Study. Diabetologia 51, 29–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Burdge GC & Lillycrop KA (2012) Folic acid supplementation in pregnancy: are there devils in the detail? Br J Nutr 108, 1924–1930. [DOI] [PubMed] [Google Scholar]
- 36. Whitrow MJ, Moore VM, Rumbold AR et al. (2009) Effect of supplemental folic acid in pregnancy on childhood asthma: a prospective birth cohort study. Am J Epidemiol 170, 1486–1493. [DOI] [PubMed] [Google Scholar]
- 37. European Commission (2013) Trustworthy websites on ‘Population groups’. http://ec.europa.eu/health/population_groups/portal/index_en.htm#tab_my_country (accessed October 2013).
- 38. European Food Safety Authority (2009) ESCO report prepared by the EFSA Scientific Cooperation Working Group on Analysis of Risks and Benefits of Fortification of Food with Folic Acid. http://www.efsa.europa.eu/en/scdocs/doc/3e.pdf (accessed October 2013).
- 39. Theodosiou L & Green J (2003) Emerging challenges in using health information from the internet. Adv Psychiatr Treat 9, 387–396. [Google Scholar]
- 40. Mo P (2012) The use of internet for health education. J Biosafety Health Educ 1, e102. [Google Scholar]
- 41. Houston TK & Allison JJ (2002) Users of Internet health information: differences by health status. J Med Internet Res 4, E7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. World Health Organization & Food and Agriculture Organization of the United Nations (2004) Vitamin and Mineral Requirements in Human Nutrition, 2nd ed. Geneva: WHO; available at http://whqlibdoc.who.int/publications/2004/9241546123.pdf [Google Scholar]
- 43. United Nations University/World Health Organization/Food and Agriculture Organization of the United Nations (2004) Human Energy Requirements: Report of a Joint FAO/WHO/UNU Expert Consultation, Rome, 17–24 October 2001. Rome: FAO; available at http://www.fao.org/docrep/007/y5686e/y5686e00.htm [Google Scholar]
- 44. Steegers-Theunissen RP, Twigt J, Pestinger V et al. (2013) The periconceptional period, reproduction and long-term health of offspring: the importance of one-carbon metabolism. Hum Reprod Update 19, 640–655. [DOI] [PubMed] [Google Scholar]
- 45. Institute of Medicine (2006) Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. In Dietary Reference Intakes Research Synthesis Workshop Summary, pp. 35–38 [CW Suitor and LD Meyers, rapporteurs]. Washington, DC: The National Academies Press. [PubMed]
- 46. Commission of the European Communities (1993) Reports of the Scientific Committee on Food (Thirty-first Series) Nutrition and Energy Intakes for the European Community. Luxembourg: Office for Official Publications of the European Communities. [Google Scholar]
- 47. McKillop DJ, Pentieva K, Daly D et al. (2002) The effect of different cooking methods on folate retention in various foods that are amongst the major contributors to folate intake in the UK diet. Br J Nutr 88, 681–688. [DOI] [PubMed] [Google Scholar]
- 48. Sotres-Alvarez D, Siega-Riz AM, Herring AH et al. (2013) Maternal dietary patterns are associated with risk of neural tube and congenital heart defects. Am J Epidemiol 177, 1279–1288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Chandler AL, Hobbs CA, Mosley BS et al. (2012) Neural tube defects and maternal intake of micronutrients related to one-carbon metabolism or antioxidant activity. Birth Defects Res A Clin Mol Teratol 94, 864–874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Vujkovic M, Ocke MC, van der Spek PJ et al. (2007) Maternal Western dietary patterns and the risk of developing a cleft lip with or without a cleft palate. Obstet Gynecol 110, 378–384. [DOI] [PubMed] [Google Scholar]
- 51. Krapels IP, van Rooij IA, Ocke MC et al. (2004) Maternal nutritional status and the risk for orofacial cleft offspring in humans. J Nutr 134, 3106–3113. [DOI] [PubMed] [Google Scholar]
- 52. Sumiyoshi Y (2007) How do we react on folic acid deficiency? Brain Spinal Cord 14, 1–3. [Google Scholar]
- 53. Kennedy D & Koren G (2012) Identifying women who might benefit from higher doses of folic acid in pregnancy. Can Fam Physician 58, 394–397. [PMC free article] [PubMed] [Google Scholar]
- 54. Talaulikar VS & Arulkumaran S (2011) Folic acid in obstetric practice: a review. Obstet Gynecol Surv 66, 240–247. [DOI] [PubMed] [Google Scholar]
- 55. Heseker HB, Mason JB, Selhub J et al. (2009) Not all cases of neural-tube defect can be prevented by increasing the intake of folic acid. Br J Nutr 102, 173–180. [DOI] [PubMed] [Google Scholar]
- 56. Kim YI (2004) Will mandatory folic acid fortification prevent or promote cancer? Am J Clin Nutr 80, 1123–1128. [DOI] [PubMed] [Google Scholar]
- 57. Troen AM, Mitchell B, Sorensen B et al. (2006) Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. J Nutr 136, 189–194. [DOI] [PubMed] [Google Scholar]
- 58. Kelly P, McPartlin J, Goggins M et al. (1997) Unmetabolized folic acid in serum: acute studies in subjects consuming fortified food and supplements. Am J Clin Nutr 65, 1790–1795. [DOI] [PubMed] [Google Scholar]
- 59. Bailey RL, Mills JL, Yetley EA et al. (2010) Unmetabolized serum folic acid and its relation to folic acid intake from diet and supplements in a nationally representative sample of adults aged ≥60 y in the United States. Am J Clin Nutr 92, 383–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Lunet N, Rodrigues T, Correia S et al. (2008) Adequacy of prenatal care as a major determinant of folic acid, iron, and vitamin intake during pregnancy. Cad Saude Publica 24, 1151–1157. [DOI] [PubMed] [Google Scholar]
- 61. National Health and Medical Research Council (2013) Australian Dietary Guidelines. http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/n55_australian_dietary_guidelines_0.pdf (accessed December 2014).
- 62. National Health and Medical Research Council (2005) Nutrient Reference Values for Australia and New Zealand. http://www.nhmrc.gov.au/_files_nhmrc/publications/attachments/n35.pdf (accessed December 2014).
- 63. Food Standards Australia New Zealand (2013) Folic acid/folate and pregnancy. http://www.foodstandards.gov.au/consumer/generalissues/pregnancy/folic/Pages/default.aspx (accessed December 2014).
- 64. Federal Ministry of Health, Austria – Public Health Portal (2014) Folat. https://www.gesundheit.gv.at/Portal.Node/ghp/public/content/Folsaeure_Folat_KH.html. Ernährung in der Schwangerschaft, https://www.gesundheit.gv.at/Portal.Node/ghp/public/content/Ernaehrung_SchwangerschaftLN.html (accessed December 2014).
- 65. D-A-CH (2013) Referenzwerte für die Nährstoffzufuhr Folat. http://relaunch-live.dge.de/fileadmin/public/doc/ws/ref/Referenzwerte-Folat.pdf, http://www.dge.de/wissenschaft/stellungnahmen/faq/folat/ (accessed December 2014).
- 66. Hoppenbrouwers K, Roelants M, Guérin C et al. (2010) Preventie van spina bifida en andere neuralebuisdefecten door foliumzuursuppletie tijdens de zwangerschap. http://www.kindengezin.be/img/foliumzuursuppletie.pdf (accessed December 2014).
- 67. Flemish Government – Child and Family Support Centre for Policy Research – Welfare, Health and Family (2014) Foliumzuur. http://www.kindengezin.be/zwangerschap-en-geboorte/zwanger/voeding-en-beweging/foliumzuur/ (accessed December 2014).
- 68. Ministério da Saúde (2012) Cadernos de Atenção Básica, nº 32 – Atenção ao pré-natal de baixo risco. Normas e Manuais Técnicos. http://189.28.128.100/dab/docs/publicacoes/geral/caderno_atencao_pre_natal_baixo_risco.pdf (accessed December 2014).
- 69. Ministério da Saúde (2006) Caderno nº 5–Manual Técnico – Pré-natal e puerpério – Atenção qualificada e humanizada. Série Direitos Sexuais e Direitos Reprodutivos. http://bvsms.saude.gov.br/bvs/publicacoes/manual_pre_natal_puerperio_3ed.pdf (accessed December 2014).
- 70. Official Bulgarian Patient Organization ‘Conception’ (2014) Folacid: Какво е фолиева киселина? http://www.zachatie.org/index.php?option=com_content&view=article&id=498:folacid----&catid=164:---sp-429&Itemid=135&highlight=WyJcdTA0NDRcdTA0M2VcdTA0M2JcdTA0MzhcdTA0MzVcdTA0MzJcdTA0MzAiLCJcdTA0M2FcdTA0MzhcdTA0NDFcdTA0MzVcdTA0M2JcdTA0MzhcdTA0M2RcdTA0MzAiLCJcdTA0NDRcdTA0M2VcdTA0M2JcdTA0MzhcdTA0MzVcdTA0MzJcdTA0MzAgXHUwNDNhXHUwNDM4XHUwNDQxXHUwNDM1XHUwNDNiXHUwNDM4XHUwNDNkXHUwNDMwIl0= (accessed December 2014).
- 71. Health Canada (2009) Prenatal Nutrition Guidelines for Health Professionals – Folate Contributes to a Healthy Pregnancy. http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/pubs/folate-fra.pdf (accessed December 2014).
- 72. Health Canada (2010) High Dose Folic Acid Supplementation – Questions and Answers for Health Professionals. http://www.hc-sc.gc.ca/fn-an/nutrition/prenatal/fol-qa-qr-fra.php (accessed December 2014).
- 73. Agence de la santé publique du Canada (2002) Pourquoi toutes les femmes qui pourraient devenir enceintes devraient prendre de l’acide folique. http://www.phac-aspc.gc.ca/fa-af/pdf/brochure_f.pdf (accessed December 2014).
- 74. Health Canada (2010) Dietary Reference Intakes – Definitions. http://www.hc-sc.gc.ca/fn-an/alt_formats/hpfb-dgpsa/pdf/nutrition/dri_tables-eng.pdf (accessed December 2014).
-
75.
 (E-health – Shanghai Jing’an District Health Promotion Board – Ministry of Health) (2014) http://www.ejiankang.org/articles/12, http://www.ejiankang.org/articles/225 (accessed December 2014).
(E-health – Shanghai Jing’an District Health Promotion Board – Ministry of Health) (2014) http://www.ejiankang.org/articles/12, http://www.ejiankang.org/articles/225 (accessed December 2014).
-
76.
 (Chinese Nutrition Society) (2007) http://www.cnsoc.org/cn/nutrition.asp (accessed December 2014).
(Chinese Nutrition Society) (2007) http://www.cnsoc.org/cn/nutrition.asp (accessed December 2014).
- 77. Danish Health and Medicines Authority (2014) Graviditet: Spis folsyre, jern, D-vitamin og eventuelt kalk som kosttilskud. http://sundhedsstyrelsen.dk/da/sundhed/graviditet/anbefalinger-til-gravide/folsyre,-jern,-d-vitamin-og-eventuelt-kalk (accessed December 2014).
- 78. Sundhedsstyrelsen (National Board of Health) (2010) Healthy habits before, during and after pregnancy. http://sundhedsstyrelsen.dk/publ/Publ2010/CFF/English/SundeVaner_en.pdf (accessed December 2014).
- 79. Nordic Council of Ministers (2014) Nordic Nutrition Recommendations 2012: Integrating nutrition and physical activity. 10.6027/Nord2014-002 (accessed December 2014). [DOI]
- 80. Tervise Arengu Instituut, Eesti Toitumisteaduse Selts (2006) Eesti toitumis – ja toidusoovitused. https://intra.tai.ee/images/prints/documents/130311768374_Eesti_toitumis_ja_toidusoovitused_est.pdf (accessed December 2014).
- 81. Tervise Arengu Instituut, Eesti Ämmaemandate Ühing (2008) Rasedate ja imetavate toitumis – ja toidusoovitused. http://www.kliinikum.ee/attachments/article/522/rasedate_ja_imetavate_emade_toitumis_ja_toidusoovitused.pdf (accessed December 2014).
- 82. Health Information Network (2011) Фолиевая кислота – что это и для чего?. http://www.terviseinfo.ee/et/component/search/?searchword=foolhape&x=-1383&y=-52&Itemid=131 (accessed December 2014).
- 83. Ravijuhend (2014) Foolhape – mis ja milleks? Foolhappe osatähtsus terve beebi sündimisel. http://ravijuhend.ee/kasutajale/pj/foolhape--mis-ja-milleks-foolhappe-osatahtsus-terve-beebi-syndimisel- (accessed December 2014).
- 84. Hasunen K, Kalavainen M, Keinonen H et al. (2004) Lapsi, perhe ja ruoka. http://www.stm.fi/c/document_library/get_file?folderId=28707&name=DLFE-3555.pdf&title=Lapsi__perhe_ja_ruoka_fi.pdf (accessed December 2014).
- 85. Programme National Nutrition Santé (2007) Le guide nutrition pendant et après la grossesse. http://www.inpes.sante.fr/cfesbases/catalogue/pdf/1059.pdf (accessed December 2014).
- 86. Institute national de prévention et d'éducation pour la santé (2014) Vous avez un projet de bébé? Pensez dès maintenant à la vitamine B9. http://www.inpes.sante.fr/CFESBases/catalogue/pdf/1429.pdf (accessed December 2014).
- 87. Institute national de prévention et d'éducation pour la santé (2011) Folates et désir de grossesse: informer et prescrire au bon moment. Les essentiels de l’INPES. http://www.inpes.sante.fr/professionnels-sante/pdf/folates-grossesses.pdf (accessed December 2014).
- 88. Manger Bouger – Programme National Nutrition Santé (2014) Les folates. http://www.mangerbouger.fr/pour-qui-242/future-maman/manger-de-facon-adaptee/avant-la-grossesse/les-folates.html (accessed December 2014).
- 89. Familienplanung – Bundeszentrale für gesundheitliche Aufklärung (2014) Lexikon – Folsäure-Prophylaxe. http://www.familienplanung.de/lexikon/folsaeure-prophylaxe/ (accessed December 2014).
-
90.
 (Department of Health) (2013) Healthy eating during pregnancy and breastfeeding. http://www.fhs.gov.hk/english/health_info/woman/20036.pdf (accessed December 2014).
(Department of Health) (2013) Healthy eating during pregnancy and breastfeeding. http://www.fhs.gov.hk/english/health_info/woman/20036.pdf (accessed December 2014).
- 91. The Hong Kong College of Obstetricians and Gynaecologists (2008) HKCOG Guidelines – Guidelines on Antenatal Care (Part I). http://hkcog.obg.cuhk.edu.hk/docs/college_guidelines/Guidelines_on_Antenatal_Care_%28Part_1%29_2008.pdf (accessed December 2014).
- 92. Agoston H, Bıcs E, Domonkos A et al. (2005) Táplálkozási ajánlások várandós és szoptató anyáknak. http://www.oeti.hu/download/taplalkozasi_ajanlasok_varandos.pdf (accessed December 2014).
- 93. Directorate of Health (2007) Fólat. http://www.landlaeknir.is/servlet/file/store93/item2792/version7/3252.pdf (accessed December 2014).
- 94. Directorate of Health (2004) Forvarnir með fólasíni. http://www.landlaeknir.is/servlet/file/store93/item2350/version4/2046.pdf (accessed December 2014).
- 95. Health Service Executive (2006) Training Programme for Public Health Nurses and Doctors in Child Health Screening, Surveillance and Health Promotion. Unit 7: Food & Nutrition. http://www.hse.ie/eng/services/Publications/Children/Unit_7_Food_and_Nutrition.pdf (accessed December 2014).
- 96. Ministero della Salute (2013) http://www.salute.gov.it/servizio/galleria.jsp?lang=italiano&id=543&dad=s&men=campagne07&label=genit (accessed December 2014).
- 97. Ministero della Salute (2009) 7 Azioni per la vita del tuo bambino. Genitori Più – Prendiamoci più cura della loro vita–Materiale informativo per gli operatori. http://www.salute.gov.it/imgs/C_17_opuscoliPoster_126_allegato.pdf (accessed December 2014).
-
98.
Ministry of Health Labour and Welfare (2009)
 (Folate). http://www.mhlw.go.jp/shingi/2009/05/dl/s0529-4u.pdf (accessed December 2014).
(Folate). http://www.mhlw.go.jp/shingi/2009/05/dl/s0529-4u.pdf (accessed December 2014).
- 99. Medicine – Latvijas veselibas portals (2013) Folijskābe grūtniecības laikā. http://www.medicine.lv/raksti/folijskabe-grutniecibas-laika (accessed December 2014).
- 100. Ministère de la Santé (2009) L’Alimentation saine pendant la grossesse. http://www.sante.public.lu/publications/rester-bonne-sante/alimentation/alimentation-saine-grossesse/alimentation-saine-grossesse-2009-fr.pdf (accessed December 2014).
- 101. Ministry for Health – Malta (2013) Registries. Birth defects. Malta Congenital Anomalies Registry. https://ehealth.gov.mt/HealthPortal/chief_medical_officer/healthinfor_research/registries/birth_defects.aspx (accessed December 2014).
- 102. National Institute for Public Health and Environment – Ministry of Health, Welfare and Sport (2014) Toolkits voor publiekscommunicatie. http://toolkits.loketgezondleven.nl/toolkits/?page_id=1754#link_5487 (accessed December 2014).
- 103. Voedingscentrum (2014) Zwanger. http://www.voedingscentrum.nl/nl/mijn-kind-en-ik/zwanger.aspx (accessed December 2014).
- 104. Folkehelseinstituttet (2014) Folat – Folattilskudd og svangerskap faktaark. http://www.fhi.no/eway/default.aspx?pid=239&trg=List_6212&Main_6157=6263:0:25,6665&MainContent_6263=6464:0:25,6667&List_6212=6218:0:25,6685:1:0:0:::0:0 (accessed December 2014).
- 105. Ministerstwo Zdrowia (2011) Żywienie w ciąży – porady żywieniowe dla przyszłych mam. http://www2.mz.gov.pl/wwwfiles/ma_struktura/docs/polzdrow_broszciaza_20120523zal12.pdf (accessed December 2014).
- 106. Direcção-Geral da Saúde (2006) Circular Normativa nº 2/DSMIA de 16/01/2006. http://www.dgs.pt/directrizes-da-dgs/normas-e-circulares-normativas/circular-normativa-n-2dsmia-de-16012006.aspx (accessed December 2014).
- 107. Societatea de Nutritie din Romania (2006) Ghid pentru alimentatia sanatoasa. http://www.ms.ro/documente/Ghid1_8318_6022.pdf (accessed December 2014).
- 108. здоровая россия (Healthy Russia) (2013) Фолиевая кислота. http://www.takzdorovo.ru/pitanie/zdorovoe-pitanie/folievaya-kislota/ (accessed December 2014).
- 109. Health Promotion Board (2012) Preganacy and diet, http://www.hpb.gov.sg/HOPPortal/health-article/3826. Conception to Birth: Eating For A Healthy Baby, http://www.hpb.gov.sg/HOPPortal/health-article/2740. Recommended Dietary Allowances, http://www.hpb.gov.sg/HOPPortal/article?id=2652 (accessed December 2014).
- 110. Institute za Varovanje Zdravja – Republike Slovenije (2013) Referencne vrednosti za vnos vitaminov in mineralov. http://www.ivz.si/prehrana?pi=5&_5_Filename=attName.png&_5_MediaId=6551&_5_AutoResize=false&pl=8-5.3 (accessed December 2014).
- 111. Ministrstvo za Zdravje – Republike Slovenije (2014) http://www.mz.gov.si/si/delovna_podrocja/javno_zdravje/sektor_za_krepitev_zdravja_in_zdrav_zivljenjski_slog/prehrana/publikacije_in_druga_gradiva/ (accessed December 2014).
- 112. Department of Health (2003) Human genetics policy guidelines for the management and prevention of genetic disorders, birth defects and disabilities. http://www.gov.za/sites/www.gov.za/files/humangenetics_0.pdf (accessed December 2014).
- 113. National Nutrition Week (2012) Nutritional tips before and during pregnancy. http://www.nutritionweek.co.za/pregnancy/31food.html (accessed December 2014).
- 114. Ministerio de Sanidad Política Social e Igualdad (2011) Estrategia Nacional de Salud Sexual y Reproductiva. http://www.msssi.gob.es/organizacion/sns/planCalidadSNS/pdf/equidad/ENSSR.pdf (accessed December 2014).
- 115. Ministerio de Sanidad Política Social e Igualdad (2006) Guía para la Prevención de Defectos Congénitos. http://www.msssi.gob.es/profesionales/prestacionesSanitarias/publicaciones/docs/GuiaPrevencionDDCC.pdf (accessed December 2014).
- 116. Swedish National Food Administration (2008) Advice about food for you who are pregnant. http://www.slv.se/upload/dokument/mat/kostrad/gravida_ammande/advice_about_food_pregnant.pdf (accessed December 2014).
- 117. Livsmedelsverket (2014) Råd om folsyra. http://www.slv.se/sv/grupp1/Mat-och-naring/Kostrad/Rad-om-folsyra/ (accessed December 2014).
- 118. Schweizerische Gesellschaft für Ernährung, Société Suisse de Nutrition, Società Svizzera di Nutrizione (2012) L’alimentation de la femme enceinte. http://www.sge-ssn.ch/media/medialibrary/2012/06/feuille_d_info_alimentation_de_la_femme_enceinte_2011.pdf (accessed December 2014).
- 119. Office Fédéral de la Santé Publique (2014) http://www.bag.admin.ch/suchen/index.html?lang=fr&keywords=grossesse+folate&search_mode=AND&from_day=&from_month=&from_year=&to_day=&to_month=&to_year=&column=&dokumenttyp=&site_mode=extern&nsb_mode=yes&Submit=rechercher#volltextsuche (accessed December 2014).
-
120.
Health Promotion Administration – Ministry of Health and Welfare (2009)
 (Maternal Health Handbook). http://www.hpa.gov.tw/bhpnet/Portal/file/ThemeULFile/200904290238200193/%E5%AD%95%E5%A9%A6%E5%81%A5%E5%BA%B7%E6%89%8B%E5%86%8A_%E8%8B%B1%E6%96%87%E7%89%881.pdf (accessed December 2014).
(Maternal Health Handbook). http://www.hpa.gov.tw/bhpnet/Portal/file/ThemeULFile/200904290238200193/%E5%AD%95%E5%A9%A6%E5%81%A5%E5%BA%B7%E6%89%8B%E5%86%8A_%E8%8B%B1%E6%96%87%E7%89%881.pdf (accessed December 2014).
-
121.
 (Food and Drug Administration) (2010) https://consumer.fda.gov.tw/Pages/List.aspx?nodeID=636 (accessed December 2014).
(Food and Drug Administration) (2010) https://consumer.fda.gov.tw/Pages/List.aspx?nodeID=636 (accessed December 2014).
-
122.
 (Food and Drug Administration) (2009) https://consumer.fda.gov.tw/Files/eweekly/%E7%AC%AC213%E6%9C%9F%E6%A0%B8%E5%AE%9A%E7%A8%BF.doc (accessed December 2014).
(Food and Drug Administration) (2009) https://consumer.fda.gov.tw/Files/eweekly/%E7%AC%AC213%E6%9C%9F%E6%A0%B8%E5%AE%9A%E7%A8%BF.doc (accessed December 2014).
- 123. NHS (2014) Vitamins and nutrition in pregnancy. http://www.nhs.uk/conditions/pregnancy-and-baby/pages/vitamins-minerals-supplements-pregnant.aspx#close (accessed December 2014).
- 124. Institute of Medicine (1998) Dietary Reference Intakes. http://www.iom.edu/Activities/Nutrition/SummaryDRIs/~/media/Files/Activity%20Files/Nutrition/DRIs/New%20Material/2_%20RDA%20and%20AI%20Values_Vitamin%20and%20Elements.pdf (accessed December 2014).
- 125. Centers for Disease Control and Prevention (2010) Folic Acid: Questions and Answers. http://www.cdc.gov/ncbddd/folicacid/documents/qanda_english.pdf (accessed December 2014).
- 126. Centers for Disease Control and Prevention (2014) Folic Acid Reccomendations. http://www.cdc.gov/ncbddd/folicacid/recommendations.html (accessed December 2014).
- 127. Women’s Health (2014) Folic acid fact sheet. http://www.womenshealth.gov/publications/our-publications/fact-sheet/folic-acid.html (accessed December 2014).


