Abstract
Increasing utilization of nuclear power enhances the risks associated with industrial accidents, occupational hazards, and the threat of nuclear terrorism. Exposure to ionizing radiation interferes with genomic stability and gene expression resulting in the disruption of normal metabolic processes in cells and organs by inducing complex biological responses. Exposure to high dose radiation causes acute radiation syndrome, which leads to hematopoietic, gastrointestinal, cerebrovascular, and many other organ-specific injuries. Altered genomic variations, gene expression, metabolite concentrations, and microbiota profiles in blood plasma or tissue samples reflect the whole-body radiation injuries. Hence, multi-omic profiles obtained from high-resolution omics platforms offer a holistic approach for identifying reliable biomarkers to predict the radiation injury of organs and tissues resulting from radiation exposures.
In this review, we performed a literature search to systematically catalog the radiation-induced alterations from multi-omic studies and the radiation countermeasures. We covered the radiation-induced changes in the genomic, transcriptomic, proteomic, metabolomic, lipidomic, and microbiome profiles. Furthermore, we have covered promising multi-omic biomarkers, FDA-approved countermeasure drugs, and other radiation countermeasures that include radioprotectors and radiomitigators. This review presents an overview of radiation-induced alterations of multi-omics profiles and biomarkers, and associated radiation countermeasures.
Keywords: Radiation damage, DNA damage repair, radiomitigators, radioprotectors, multi-omics, radiation biomarkers
Introduction:
Exposure to high dose ionizing radiation (IR) causes complex cellular damage at the molecular level (1). Acute radiation syndrome (ARS) occurs due to total body exposure to high doses of IR or with long term exposure of significant doses to partial body condition. Radiation-induced DNA damage poses both direct and indirect consequences.
Direct consequences result when organic molecules absorb the radiation energy (DNA, proteins, or metabolites), leading to point mutations, DNA strand breaks, DNA crosslinks, chromosomal aberrations, protein modifications, and metabolite alterations (Figure 1). If such alterations are unrepaired, they can cause permanent cellular damage or cell death (2). Indirect biological effects of radiation exposure occur when water molecules in cells are oxidized and/or ionized through radiolysis. The free radicals and peroxides produced by this process interact with the surrounding cellular components causing cellular damage (3). Radiolysis results in the formation of free radicals, such as hydroxyl radical, superoxide radical anion, and hydrogen radicals, and their derivative non-radical species such as hydrogen peroxide, peroxynitrite, and ozone; all of which can lead to DNA damage. The extent of DNA damage caused by free radicals or reactive oxidants includes single-strand breaks, double-strand breaks, amplified damaged sites, base modifications, and adduct formation, which are often more significant than the direct action of radiation (4).
Figure 1:
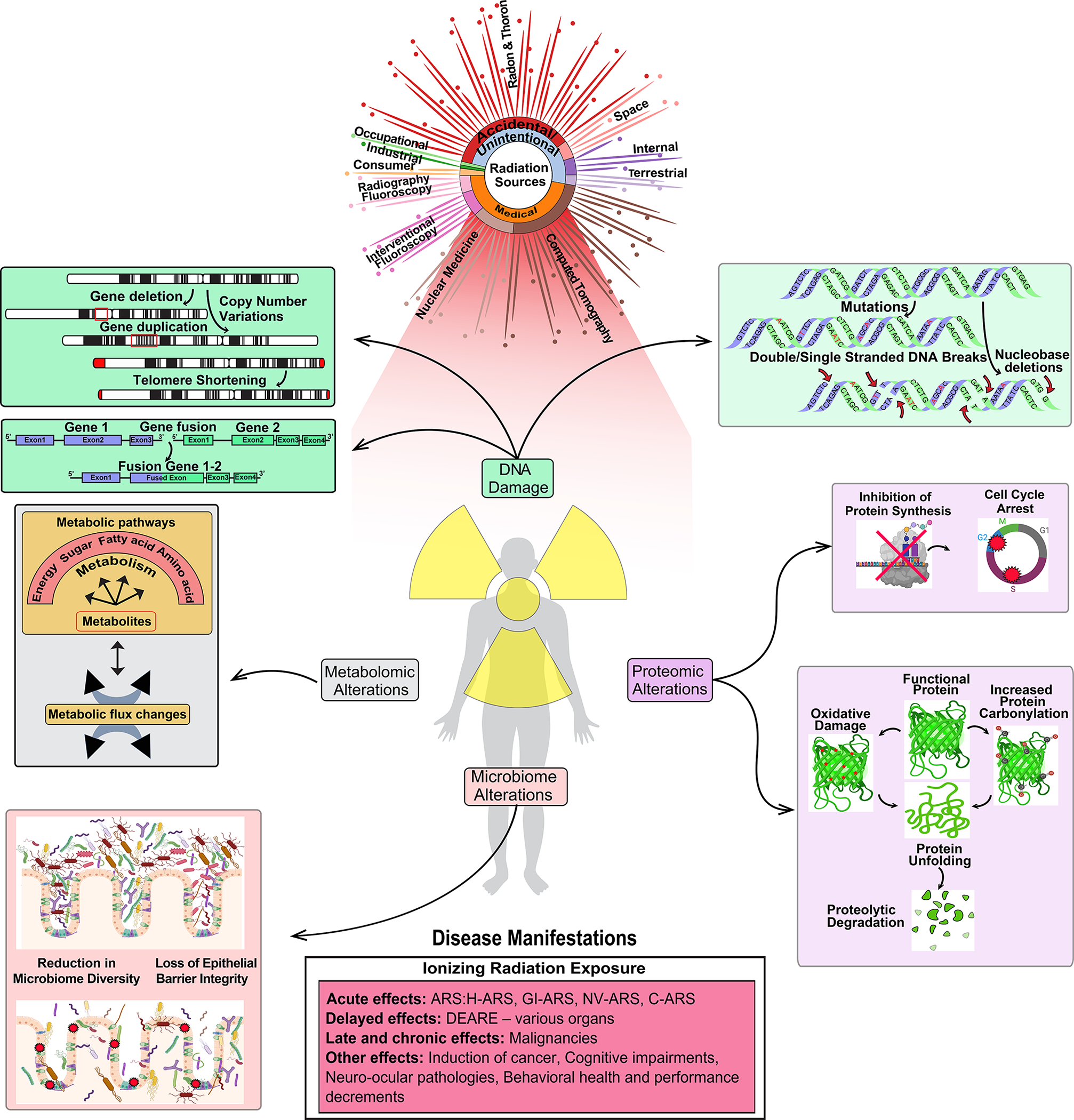
Overview of the sources of radiation exposure and their impact on human biology. The damaging effects of irradiation on DNA, proteins, metabolites/lipidomes, microbiome, and the resulting health consequence in humans are depicted. Significant sources of natural and medical IR are mentioned here. Exposure to radiation induces DNA damage, leading to chromosomal aberrations, gene fusions, DNA breaks, and mutations. Proteomic alterations can lead to inhibition of protein synthesis, changes in protein folding and degradation. Radiation exposure also affects gut health through impairment of gut epithelial barrier integrity, villus shortening, and causes reduction in microbiome diversity. Irradiation also leads to a lowered activity in key metabolic pathways including glycolysis and energy metabolism. Exposure to high dose IR causes acute, delayed, late, and chronic health effects. Only the major health consequence from both high and low dose radiation exposure is represented in this figure.
Identification of early radiation response genes by using RNAseq and whole human genome DNA-microarrays have been major approaches to study radiation biology. Currently, there are several approaches for screening biomarkers of IR, which involve measuring blood cell counts, protein concentrations, or gene expression, and cytogenetic approaches in irradiated cells. In addition to tracking DNA lesions and DNA repair proteins, other radiation-induced changes such as genome-wide copy number variants (CNVs) and mutations have also been exploited as radiation biomarkers. CNV changes after radiation exposure are distributed non-randomly across the genome and are also found recurrent in some loci. Genome-wide hotspots that harbor recurrent CNVs have been effectively utilized as biomarkers of radiation exposure (5). Chromosomal microarray analysis (CMA), single nucleotide polymorphism (SNP) arrays, comparative genome hybridization (CGH) arrays, or Next-generation genome sequencing methods were utilized for studying genome-wide CNV changes and quantifying the exposure (6, 7). Similarly, germline mutations in the offspring of individuals exposed to radiation were used as biomarkers for estimating parental radiation exposure and biomonitoring (8).
A detailed account of different exposure sources and resulting damages on DNA, proteins, metabolites, and the microbiome is depicted in Figure 1. This review covers the consequences of radiation on genomic, transcriptomic, metabolomic, proteomic, and microbiome profiles. We performed a literature search to cover radiation-based omics studies to identify biomarkers. Specifically, the keywords such as “gene/miRNA/protein biomarkers in radiation”, “omic-based biomarkers in radiation injuries”, “radiation omics”, and “radiation countermeasures”, “radiation drugs” were used to search the literature. The information on omics-based biomarkers and radiation drugs studied in any injured tissue/organ in human, mouse, nonhuman primates (NHPs), or any other model organism, were saved. In the latter part of the review, we focused on the literature covering the promising radiation countermeasures that include both radioprotectors and radiomitigators. Hundreds of abstracts extracted from keyword searches were manually curated for their suitability and included in the present review.
Radiation-induced genomic alterations:
Experiments in a controlled setting exploring the impact of IR on laboratory animals or cell lines can help understand the extent of radiation damage possible in humans. A genome-wide study on the impact of IR on germline changes in mice noted increased de novo CNVs and indels in offspring due to irradiation at 3 Gy (9). This study also identified a significantly higher number of clustered mutations in the progeny of irradiated male mice. A signature of radiation exposure is the association of these mutations with clustered damage sites. These clustered DNA lesions occur within one or two helical turns of DNA, i.e., within a short distance. DNA lesions are mostly seen as an aftermath of radiation exposure. Complex double-stranded and single-stranded DNA breaks and clustered mutations in specific genomic areas are the most damaging as the repair process of these types of changes are challenging. Single-stranded DNA breaks in the clustered damage sites can lead to inefficient base excision repair pathways resulting in persistent lesions during subsequent replication cycles (10, 11). Complex clusters of DNA lesions caused by IR initiate further mutagenesis and genomic instability, which could eventually lead to cell death (12). This property of IR is exploited in medicine to target and kill tumor cells. The increased number of DNA breaks induced by γ-radiation (<2.0 Gy) and the increased replication rate in tumor cells act in synergy to eliminate cancerous cells (13). Cells with a high proliferation rate are prone to radiation-induced DNA damage clusters (14), suggesting that different tissues might respond to different dose, dose rates, and radiation quality.
In vitro studies have also demonstrated the ability of radiation exposure to induce CNVs (5, 15). Experiments conducted on human fibroblast cells detected genome-wide distributions of CNVs associated with radiation exposure (<=3 Gy), with several hotspots corresponding to aphidicolin (APH) and hydroxyurea (HU) induced replication stress (5). This study also reported a higher percentage of duplications than other stressors and that CNVs were detected more than seven days post-exposure. The aftermath of radiation exposure on the germline of male mice identified an eight-fold increase in CNVs in offspring, indicating a transgenerational impact (9). Several other studies using different radiation sources, such as laser-driven electron accelerators or X-rays, identified increasing CNVs in chromosome hotspots (15, 16). It was also observed that radiation-induced tumors in Trp53 mutant mice developed CNV changes in chromosome hotspots associated with specific tumor types (7). For example, radiation-induced mammary tumors exhibited amplification of the MET locus on chromosome 6. Consistent with this observation, radiation-induced gliomas in Trp53/PTEN mutant mice also developed recurrent amplification of the locus containing the RTK and MET gene in chromosome 6 (17), changes that were associated with cancer stem cell maintenance in both of these cancers (18, 19).
High doses of γ-radiation on hematopoietic cells induced gene fusions associated with leukemogenesis, including AML1-ETO, BCR-ABL, DEK-CAN, and DEK-ABL, indicated that DNA breakage and misrepair is a significant risk factor for leukemia (20). Low and therapeutic dose radiation on hematopoietic stem and progenitor cells (HSPC) isolated from human umbilical cord blood also led to increased formation of preleukemic fusion genes associated with radiation-induced DNA damage (21). A significant increase in BCR-ABL fusion was reported in this study compared to controls, suggesting that the risk of developing leukemia exists even at low radiation doses.
Exposure to high-intensity radiation is also known to cause excessive skin damage. A study investigating the impact of γ-irradiation (53 Gy, dose rate 0.6 Gy/minute) on minipig skins found elevated apoptosis and weakened stem cell replication after irradiation (22). A higher percentage of ɣH2AX phosphorylation and increased frequency of radiation-induced 53PB1 foci indicated radiation-induced DNA damage. Extensive radiation-induced fibrosis (RIFs) persisted for weeks in a minor fraction of cells (<1%) and reflect the complex DNA damage, including single-strand and double-strand DNA breaks, and base lesions that are refractory to DNA repair mechanisms. A similar observation of persistent RIFs after low-intensity in vivo irradiation of human skin was observed 24 h after 4 Gy exposure with significant inter-individual variation in RIF levels that could be associated with differences in DNA repair mechanisms (23).
Recent advancements that aid in studying three-dimensional (3D) DNA organization have helped the research community to understand the effect of radiation stress on genome-wide 3D organizational structure. CCCTC-binding factor (CTCF) and cohesin, involved in chromatin organization, are early responders to DNA damage induced by radiation (24, 25). In this context, incipient role of G protein-coupled receptors (GPCRs) signaling system in DNA damage response has opened up a new direction to controlling the damage due to radiation and subsequently developing target-specific therapeutics (26). A previous study showed that protein G2A (member of GPCR family) responds to DNA damages when induced in lymphocytes (27). Similariliy, the combined role of GPCR relaxin family peptide 3 receptor (RXFP3) and GPCR interacting protein 2 (GIT2) in response to oxidative stress and DNA damage have been identified by Gastel and collegues (28, 29). These studies also concluded that RXFP3-GIT2 system plays a significant role in regulating cellular degrdation associated with DNA damage. Nevertheless, further studies are needed to identify the involvement of GPCRs in radiation-caused DNA damage. There are currently no reports that explore the consequences of γ-radiation on chromatin organization, but one study revealed the ability of 5 Gy X-ray irradiation to induce changes in the genome-wide 3D organization in human fibroblasts and lymphoblastoid cells (30). A strengthening of topologically associating domain (TAD) formation was observed after X-ray exposure, suggesting noticeable 3D genome changes after exposure. TADs, formed through the interaction of CTCF, play a crucial role in bringing genes and regulatory elements in close contact with each other, leading to preserving genome integrity after irradiation. This impact lasted for days and was more pronounced in lymphoblastoid cells than in fibroblasts (30), pointing to differences in radiosensitivity across cell types. This study also linked the structural change to the Ataxia Telangiectasia Mutated (ATM) DNA repair pathway. These reports noted that radiation-induced DNA damage was extensive and included point mutations, DNA breaks, and changes in the genome-wide 3D organization, highlighting the importance of functional DNA repair mechanisms to mitigate against the lasting effects of such radiation-induced genomic insults.
Transcriptomic changes induced by radiation:
Messenger RNA (mRNA)-expression:
Genome-wide expression analysis of the peripheral blood (PB) in different mammalian systems has shown radiation-induced alterations at different exposure intensities. Experiments in partial-body irradiated at 0.5 Gy, 2 Gy and 10 Gy mice identified PB expression signatures that differentiated radiation damage with 79–100% accuracy (31). Additional studies on global gene expression profiles in the blood of male C57BL/6 mice identified a 74-gene signature associated with moderate to low radiation doses (at 0.5, 2, 5, and 8 Gy). More than one-third of these genes were regulated by TP53, suggesting that TP53 plays a vital role in radiation responses (32). The most marked response was in genes associated with natural killer (NK) cell functions, reflecting a relative loss of NK cells from the population. T- and B-cell mediated immunity genes were also significantly influenced by 48 h after γ-ray irradiation (0, 0.5, 2, 5, 8 Gy). Downregulation of NK-cytotoxicity-associated genes was observed, including NKG7, GNLY, and GZMA (33). Several of these genes were further validated in independent studies, including human PB, indicating that these genes could be further explored as reliable radiation biomarkers (34, 35) (Table 1). Other reports that analyzed the global expression profile of irradiated mice have identified trends in gene expression. An increasing number of genes were differentially expressed within days after irradiation with 2, 3, and 5 Gy, and gene expression increased with increasing radiation doses (21). Specifically, the expression of SLC25A51 and CCNA2 showed an initial suppression followed by recovery of the transcript levels by seven days after exposure to mice with doses less than 10 Gy (21). Another study focused on differential expression of genes in mice after irradiation at 4 and 8 Gy found many upregulated genes including CDKN1A, MDM2, BBC3, and CCNG1, and downregulated genes including TCF4 and MYC. The same study also identified genes, DDB2, PCNA, GADD45A, SESN1, RRM2B, KCNN4, IFI30, and PTPRO, which are downregulated in mice but uprgulated in human (36). The differnence in gene expression were also observed in wild-type C57BL/6 (8 Gy) and DNA double-strand break repair-deficient Atm−/− (4 Gy) and Prkdcscid (3 Gy) mutants of C57BL/6, concluding that regulators including TP53 and NFκB are activated by radiation exposure only in wild type mice (37). The study further identified that mutant strains show inflammatory responses after radiation exposure. The source of the radiation can be a key factor for the transcriptomic changes, as discussed by Broustas et al., 2017, based on the experiments where 7,285 and 5,045 genes were differentially expressed in irradiated blood of mice at 0.25 or 1 Gy of neutron or 1 or 4 Gy x-ray radiation, respectively (38). Similarly, effects of age on transcriptomics profile have been observed after irradiating (4 Gy x-rays) young (2 months) and old (21 months) male mice for 24h. The microarray-based gene expression analysis suggested that young mice were more active against radiation by upregulating pathways related to apoptosis and phagocytosis. The pathways associated with fibroblast growth factor signaling were underrepresented in old mice, while hematologic malignancies related pathways were enriched (39).
Table 1:
List of potential mRNA biomarkers in radiation-caused injury in different animal models.
| Gene | Full name | Effect | Study model | References |
|---|---|---|---|---|
| GDF15 | Growth Differentiation Factor 15 | Cell-cycle arrest | Mouse | (47) |
| CKAP2 | Cytoskeleton-associated protein | Cell-cycle arrest | Mouse | (47) |
| PTPN1 | Protein Tyrosine Phosphatase Non-receptor Type 1 | Survival improvement in mice | Mouse | (48) |
| RALB | RAS like proto-oncogene | Shows radio resistiveness to different tumor types | Mouse | (49) |
| CKB | Creatine Kinase B | Prognostic biomarker for dose dependent radiation exposure | Mouse | (50) |
| CCNA2 | Cyclin A2 | G1/S and G2/M cell cycle regulators | Mouse | (51, 52) |
| SLC25A51 | Solute Carrier Family 25 Member 51 | Mitochondrial NAD+ transporter | Mouse | (21, 52) |
| ACE | Angiotensin-converting enzyme | Inhibition of ACE is associated with hematopoietic recovery following radiation | Mouse | (53) |
| DDA3 | Differential Display And Activated By P53 | Supressing cell growth | Mouse | (54, 55) |
| MPL | MPL Proto-Oncogene, Thrombopoietin Receptor | Associated with survival in radiation-exposed mice | Mouse | (56) |
| GZMH | Granzyme H | NK-cytotoxicity | Baboons | (57, 58) |
| NCR3 | Natural Cytotoxicity Triggering Receptor 3 | NK-cytolysis | Baboons | (57, 58) |
| PRF1 | Perforin 1 | Membrane pore in cytolysis | Baboons | (57, 58) |
| KLRF1 | Killer cell lectin-like receptor F1 | NK-cytolysis | Baboons | (57, 58) |
| GBP2 | Guanylate Binding Protein 2 | Oxidative killing and antiviral activity | Baboons | (59) |
| GLUL | Glutamate-Ammonia Ligase | Synthesis of glutamine | Baboons | (59) |
| CCR7 | C-C Motif Chemokine Receptor 7 | Migration of memory T-cells | Baboons | (57) |
| CD117 | KIT Proto-Oncogene, Receptor Tyrosine Kinase | Regulation of cell survival and proliferation | Baboons | (57) |
| RNASE3 | Ribonuclease A Family Member 3 | Antimicrobial function | Baboons | (57) |
| VSIG4 | V-Set And Immunoglobulin Domain Containing 4 | Negative regulator of T-cell proliferation | Baboons | (57) |
| ARG2 | Arginase 2 | Regulation availability of L-arginine to nitric oxid synthase | Baboons | (60) |
| CD177 | CD177 Molecule | Promote neutrophil activation | Baboons | (60) |
| WLS | Wnt Ligand Secretion Mediator | Regulates Wnt proteins sorting | Baboons | (60) |
| PPP3CC | Protein Phosphatase 3 Catalytic Subunit Gamma | T-cell and B cell immune response | Baboons | (59) |
| SH2D1A | SH2 Domain Containing 1A | Stimulation of B- and T-cells | Baboons | (57, 58) |
| ARHGAP45 | Rho GTPase Activating Protein 45 | Migrating cancer and hematopoietic cells | Baboons | (59) |
| HERC5 | HECT Domain And RCC1-Like Domain-Containing Protein 5 | T-cell and B-cell immune response, migrating cancer and hematopoietic cells | Baboons | (59) |
| CDCA7L | Cell Division Cycle Associated 7 Like | Cell cycle regulation | Baboons | (59) |
| NKG7 | Natural Killer Cell Granule Protein 7 | Natural killer (NK)-cytotoxicity | Human | (33, 61) |
| FDXR | Ferredoxin Reductase | Radiation dose estimator | Human | (62–64) |
| ERBB2 | Erb-B2 Receptor Tyrosine Kinase 2 | Reducing apoptosis by activating NFκB-related signaling | Human | (65, 66) |
| FOXM1 | Forkhead box protein M1 | Inhibition of FOXM1 elevate radiation sensitivity | Human | (66) |
| TP53 | Tumor protein p53 | Transcription factors modulating genes which are involved in regulation of irradiated cell cycle | Human | (32, 66) |
| GNLY | Granulysin | NK-cytotoxicity | Human | (33, 61) |
| GZMA | Granzyme A | NK-cytotoxicity | Human | (33, 61) |
| CBR1 | Carbonyl Reductase 1 | Radioprotective in radiotherapy on head and neck squamous cell carcinoma, radiosensitivity predictor in oesophageal cancer | Human | (67–70) |
| IGF1R | Insulin-Like Growth Factor 1 Receptor | Increased radio resistance in chemotherapy if IGF1R is overexpressed | Human | (71) |
| ATF3 | Activating transcription factor 3 | Regulate expression of other genes human myeloid tumor cell line | Human | (72, 73) |
| DDB2 | Damage Specific DNA Binding Protein 2 | Dose-dependent response | Human | (64, 74, 75) |
| XPC | XPC Complex Subunit, DNA Damage Recognition And Repair Factor | Dose-dependent response | Human | (74, 75) |
| GADD45A | Growth Arrest And DNA Damage Inducible Alpha | Radiation induced gene expression in human peripheral blood lymphocytes | Human | (72, 74) |
| FDXR and DDB2 Combined with WNT3 and POU2AF1 | FDXR- Ferredoxin Reductase; DDB2- Damage Specific DNA Binding Protein 2; WNT3- Wnt Family Member 3; POU2AF1- POU Class 2 Homeobox Associating Factor 1 | Up-regulation of FDXR and DDB2 Combined with down-regulation of WNT3 and POU2AF1: predictive biomarkers via T- and B-lymphocytes regulation | Human | (76, 77) |
| GPX | Glutathione Peroxidase 1 | Elevated expression in human lymphoblastoid TK6 cells for high doses γ-radiation injuries | Human | (78) |
| P21 | Protein 21 | Elevated expression in human lymphoblastoid TK6 cells for high doses γ-radiation injuries, upregulation at protein level in irradiated apoptotic PBMCs | Human | (78–80) |
| BAD | BCL2 Associated Agonist Of Cell Death | Upregulation at protein level in irradiated apoptotic PBMCs | Human | (79, 80) |
| XIAP | X-Linked Inhibitor Of Apoptosis | Upregulation at protein level in irradiated apoptotic PBMCs | Human | (79, 80) |
| BAX | BCL2 Associated X, Apoptosis Regulator | Upregulation at protein level in irradiated apoptotic PBMCs | Human | (79, 80) |
| NNMT | Nicotinamide N-Methyltransferase | Prevention of Mesenchymal Cancer stem cells in case of radiation-caused injuries | Human | (81) |
| TFPI | Tissue Factor Pathway Inhibitor | Inhibition of TFPI supress hemophilia, coagulopathy due to radiation exposure decreases clotting ability | Human | (82, 83) |
| MMP9 | Matrix metalloproteinase-9 | Involve in degradation of extracellular matrix proteins and activates cytokines for tissue remodelling | Human | (69, 70) |
| ANXA2 | Annexin A2 | Regulate nuclear factor κB nuclear translocation to prevent radiation-induced apoptosis | Human | (84, 85) |
| TNFSF4 | TNF Superfamily Member 4 | Lymphocyte activation | Human | (86) |
| KIF20A | Kinesin Family Member 20A | Provide radio-resistance | Human | (86) |
| PSRC1 | Proline/serine-rich coiled-coil protein 1 | Regulation of mitotic spindle dynamics | Human | (86) |
| CDCA3 | Cell division cycle-associated 3 | Modulation of cell cycle progression | Human | (86) |
| NFKBIA | NF-Kappa-B Inhibitor Alpha | Decreasing growth in cancer lineages and increase in apoptosis by Blocking NF-kB | Human | (69, 70) |
| AURKA | Aurora Kinase A | Increase in radiosensitivity due to inhibition of AURKA | Human | (87, 88) |
| ANXA1 | Annexin A1 | Improving prognosis in radiation-caused lung injuries | Human | (89) |
| ZAP70 | Zeta Chain of T-Cell Receptor Associated Protein Kinase 70 | Deficiency is associated with loss of T-cells | Human | (69, 70, 90) |
| GADD45 | Growth arrest and DNA damage-inducible protein GADD45 alpha | Inhibits entry of cells into S phase | Human | (72, 73, 75, 86) |
| PCNA | Proliferating Cell Nuclear Antigen | Control of eukaryotic DNA replication, elevated expression in human lymphoblastoid TK6 cells for high doses γ-radiation injuries | Human | (33, 36, 78) |
| BRD4 | Bromodomain-Containing Protein 4 | BRD4 inhibition improve cancer cell survival following irradiation | Human | (69, 70, 91) |
| MCL1 | MCL1 Apoptosis Regulator | Protects against apoptosis caused by radiation exposure | Human | (92) |
| MDM2 | E3 ubiquitin-protein ligase Mdm2 | Ubiquitination of p53/TP53 | Human | (72–74) |
| MYD88 | Innate Immune Signal Transduction Adaptor | Prevent fibrosis and long term damage due to radiation exposure | Human | (93) |
| XRCC1 | X-ray repair cross-complementing protein 1 | DNA repair | Human | (78) |
| IDH3B | Isocitrate Dehydrogenase (NAD(+) 3 Beta | IDH3B helps in generating NADH which is a radioprotective for mouse intestine | Human | (69, 70, 94) |
| STAT4 | Signal Transducer And Activator Of Transcription 4 | Promotion of IL-12 response | Baboons, Mouse | (57, 95) |
| WNT3 | Wnt Family Member 3 | Canonical Wnt signalling pathway | Baboons, Human | (57, 64) |
| POU2AF1 | POU domain class 2-associating factor 1 | Response of B-cells to antigens | Baboons, Human | (57, 64, 76, 77) |
| CDKN1A | Cyclin-dependent kinase inhibitor 1 | Inhibition of cellular proliferation in response to DNA damage, radiation-induced gene expression in human peripheral blood lymphocytes, dose-dependent response, cell-cycle arrest | Mouse, Human | (47, 69, 70, 72, 74, 75, 86) |
| LGALS1 | Lectin Galactoside-Binding Soluble | Induction of cell death | Mouse, Human | (54) |
In a ex vivo irradiated (0.56 Gy, 2.23 Gy and 4.45 Gy, acute dose rate = 1.03 Gy/min, low dose-rate = 3.1 mGy/min) human whole blood, 454 genes were differentially expressed 24h after exposure to all doses, while 598 genes were differentially expressed after acute exposure. These genes were mainly enriched in functions related to immune response, B-cell mediated immunity, cell-to-cell signaling, and natural killer cell activation (40). Similarly, in the whole-thorax irradiated (single dose of 10 Gy) NHP, 1,187 mRNA transcripts were significantly dysregulated 30 days after exposure. The differentially regulated genes mostly belonged to functions related to immune responses (41). The transcriptomics effect of radiation dose (2.5 Gy and 0.1 Gy of low-LET protons) were also observed in a human three dimensional tissue model EPI-200 at 4, 16 and 24 h after exposure. The study found that high dose reduced terminal differentiation and structural integrity, while low dose were mainly associated with recovery and tissue repair (42). A systematic review included 27 previous radiation-based studies and identified 27 potential genes that have significant correlation with radiation dose. Top five discriminatory genes, TNFSF4, FDXR, MYC, ZMAT3, and GADD45A were idenfied when compared between doses < 2 Gy and ≥ 2 Gy (43). The roles of several other long ncRNAs in radiation responses have been discussed in previous studies (44–46); however not covered in present review.
Experiments in NHPs, such as baboons, can model radiation-induced changes comparable to humans. Baboons that developed the clinically relevant disease, hematologic acute radiation syndrome (HARS, classified based on severity ranging from H1–3), had a persistent change in peripheral gene expression within 1 or 2 days post-irradiation to 2.5 or 5 Gy γ-radiation (57). Of these, three genes (WNT3, POU2AF1, and ZZZ3) showed a persistent change in gene expression over time. Several genes associated with immune-related functions that impact T-cell migration (CCR7, CD117) and T-cell responses (VSIG4), antimicrobial function (RNASE3), promotion of Interleukin-12 (IL-12) response signal transducer and activator of transcription 4 protein (STAT4), stimulation of B- and T-cells (SH2D1A), and initiation of apoptosis in pathogen-infected cells (GZMH) and cytolysis (PRF1, NCR3, and KLRF1) were associated with the radiation-induced response (58). Another study in the same mammalian system identified radiation-induced overexpression of genes involved in cell cycle regulation (CDCA7L), modification of the T-cell and B-cell immune responses (GBP2, GLUL, HERC5, and PPP3CC), erythropoiesis (GBP2), and cell migration of cancer and hematopoietic cells (HERC5, HMHA1) (59) when irradiated with 2.5 or 5 Gy. A recent study using NHPs showed that IL-3 signaling, ephrin receptor signaling, ErbB signaling, nitric oxide signaling in the cardiovascular system, Wnt/β-catenin signaling, and inflammasome pathways were associated with positive survival outcomes in NHPs after acute exposure to 6.5 Gy γ-radiation (96).
Experiments to study the effect of radiation (γ-rays of 0.1 Gy) on gene expression in a human myeloid tumor cell line (ML-1) identified genes that responded in a dose-dependent manner (72, 73). Among the set of genes identified in these studies (CDKN1A, GADD45, MDM2, ATF3, and BAX), CDKN1A, GADD45A, and MDM2 were also overexpressed with low-dose γ-radiation. Of the transcriptionally active genes identified after radiation exposure, induction of CDKN1A and GADD45A was proportional to exposure intensity. Another study that explored the transcriptional changes in human peripheral blood lymphocytes (PBL) also identified CDNK1A and GADD45A as radiation-induced genes after exposure to 2 Gy using X-ray (74). This study also identified a linear dose-dependent response in the expression of DDB2, CDKN1A, and XPC. Human PB signatures are of interest because radiation exposure biomarkers can distinguish between the irradiated and non-irradiated samples with 100% accuracy (54).
Exposure to high doses of γ-radiation (5, 10 and 20 Gy) in human lymphoblastoid TK6 cells elevated mRNA levels of GPX, GADD45, P21, and PCNA, moderately repressed XRCC1 expression, and strongly down-regulated the expression of KU80 (78). A similar observation was noted when non-immortalized T-cells were exposed to low to moderate γ-radiation doses (between 0.15–12 Gy), with transcriptional induction of several genes, including CDKN1A, GADD45A, TNFSF4, KIF20A, PSRC1, and CDCA3 (86). Another in silico network analysis identified eight ARS-associated genes (BRD4, NFKBIA, CDKN1A, TFPI, MMP9, CBR1, ZAP70, IDH3B) and confirmed through literature mining (69, 70). In these experiments, commonly studied genes such as CDKN1A and GADD45 in mice, NHPs, and human cell lines showed promise to be effectively utilized as radiation biomarkers.
MicroRNA (miRNA) expression:
Serum microRNA (miRNA) signatures indicate the long-term impact of total-body irradiation (TBI, 6.5 Gy and 8 Gy) in mice when measured within 24 h of exposure (97). These miRNA signatures distinguished mice exposed to radiation from unirradiated animals, which correlated with the impact of irradiation on hematopoietic stem cells (HSCs). Several studies in mice have explored the miRNA signatures associated with radiation exposure. An analysis identified long term elevation of miR-21 in the brain when hippocampal cells and brain tissue from mice were irradiated at 0.5 Gy using X ray at different times for 1 year (98), and upregulation of miR-145 and miR-663 in AHH-1 cells and HPBLs, when irradiated at 4 Gy using γ-ray for 4 h or 24 h (99). Radiation-induced miRNA profiles were also found to be time- and dose-dependent. Serum miRNAs (miR-27a-3p, miR-187–3p, miR-30a-3p, and miR-30c-5p) were detectable within 24 h after radiation exposure in mice. Of these miRNAs, the expression of miR-30a-3p and miR-30c-5p were able to differentiate between radiation doses (6.5 Gy vs 8 Gy ) after a week of post-exposure (97). The microRNAs were also correlated with injuries to human bone marrow cells bassed on serum profiling of mice exposed to sublethal (6.5 Gy) and lethal (8 Gy) doses of radiation (97). In a different study, 600 miRNAs from serum of irradiated (at 1 to 12 Gy) mice were compared in a dose dependent manner at time points of 24 and 48 hr (100). Similarly, serum miRNA signatures containing miR-130a-3p, miR-150-5p, miR-142-5p, miR-706, and miR-342-3p can be used to identify low-dose (2 Gy) radiation cohorts exposed to radiation within 24 h (97). The dose reconstruction algorithms that were developed based on serum miR-150-5p depletion normalized with miR-23a-3p in mouse models can approximate the absorbed dose at various time points during the recovery phase (101). Leukemia specimens from patients treated with fractionated radiation showed depletion of miR-150-5p in blood (101). Changes in plasma miRNAs such as miR-34a-5p, miR-100-5p, and miR-150-5p were associated with pro-inflammatory NF-κB-mediated functions as studied in heart and lung damaged C3H mouse after irradiation. In the same study, expression of miRNAs, miR-34b-3p, miR-96-5p, and miR-802-5p, were significantly altered in C57Bl/6 mice after exposing their heart and lung at 13.99 Gy (102). In a separate study using rats, miRNAs, miR-144-5p, miR-144-3p, miR-142-5p and miR-19a-3p, were differentially regulated in the blood after lung-specific injuries caused by a single dose of radiation at 15 Gy (dose rate of 1.43 Gy/min) radiation (103). miRNA miR-150 found in blood and lung also showed significant downregulation after thoracic irradiation in female WAG/RijCmcr rats (103). Similarly, thoracic exposure of NHPs to radiation causing injuries in lung and heart have shown differences in the expression profile of blood miRNAs, miR-199a-3p and miR-25-3p, after a single exposure to whole-thorax and lung irradiation (WTLI) at 9.8 or 10.7 Gy (104). In C3H mice afte whole thorax irradiation at dose 13.92 Gy, differentially expressed miRNAs miR-34a-5p, miR-100-5p, and miR-150-5p were identified associated with survival (102). These studies have improved our ability to consider circulatory miRNAs as organ-specific markers upon IR damage.
Similarly, microarray analysis identified seven miRNA signatures altered by irradiation at 5.8, 6.5, or 7.2 Gy (dose rate of 0.6 Gy/min) in NHPs (105). Conserved serum miRNA signatures have the potential to serve as predictive biomarkers for radiation injury in humans, NHPs, and mice. A combination of three miRNAs (miR-133b, miR-215, and miR-375) was found to identify radiated versus unexposed NHPs, accurately. Two microRNAs, miR-199a-3p and miR-25-3p, from blood of whole-thorax lung irradiated NHPs at 10.7 Gy were differentially expressed and associated with survival (104). Similarily in another study, microRNAs associated with neutropenia were identified in NHPs after TBI (at 2–6.5 Gy) and whole thorax lung irradiation (at 9.8 or 10.7 Gy) (106). Radiation-induced mortality could also be predicted by a five-miRNA (miR-133b, miR-215, miR-375,miR-126, and miR-30a) composite signature in macaques (105). The same study identified a set of two miRNAs (miR-30a and miR-126), which together can be used as a survival indicator (107).
Global miRNA expression changes measured in the PB of baboons within the days after irradiation at 2.5 or 5 Gy (dose rate- 0.08 Gy/min for 5 Gy TBI and 5 Gy 50% partial body irradiation, and 0.32 Gy/min for other) identified miR-425-5p expression that distinguished different HARS groups (108). Six miRNA species (miR-133, miR-124, miR-29c, miR-378, miR-574-3p, and rno-miR-7) were identified as promising candidates that can distinguish HARS groups with and without pancytopenia (59). Many miRNA species were already known to be linked with radiosensitivity (e.g., miR-22, miR-29c, miR-195, miR-212) or chemotherapy resistance (e.g., miR-331-5p). In particular, miR-212 involved in radiosensitivity and immune modulation was upregulated 48 to 77-fold over an extended period (60). Another study that compared radiation-induced global gene expression profiles in baboons when exposed to total-body radiation at 2.5 or 5 Gy (dose rate- 0.08 Gy/min for 5 Gy TBI and 5 Gy 50% partial-body irradiation, and 0.32 Gy/min for other), identified a significant downregulation of miR-342-3p, which allowed an almost complete separation of HARS categories (109). These miRNAs can be further explored as reliable radiation-biomarkers for accurate screening and evaluation of radiation exposure in humans.
A systematic review and meta-analysis study showed that seven miRNAs that include miR-150, miR-29a, miR-29b, miR-30c, miR-200b, miR-320a, and miR-30a showed significant correlation with given dose of radiation across different species that mainly include human, mouse, rat and NHPs (110, 111). These studies support the hypothesis that miRNAs from body fluids can be used as biomarkers for detecting organ-specific injuries caused by high dose radiation exposures. Studies in humanized mice engrafted with human CD34+ HSCs showed that the expression profile of a serum miRNA signature containing miR-27a-3p, miR-187-3p, miR-30a-3p, and miR-30c-5p was altered in response to irradiation at 6.5 Gy and 8 Gy. This indicates that radiation-induced miRNA signatures may be conserved between mice and humans and can serve as viable biomarkers of radiation injury in humans, however, further studies are needed to validate their utility as biomarkers (97).
An extensive list of important mRNAs, miRNAs, and their tested effects in different animal models is provided in Table 1 and Table 2, respectively. The summary of miRNA and gene-based biomarkers studied in different animal models is illustrated in Figure 2A. Note that the overlap among the gene or miRNA biomarkers across the species is little to none warranting the need to further investigate and validate their utility as effective radiation biomarkers.
Table 2:
List of potential miRNA biomarkers of radiation-caused injuries in different animal models.
| miRNA | Full name | Effect | Study model | References |
|---|---|---|---|---|
| miR-124 | MicroRNA 124 | Modulator of immunity and inflammation, associated with anti-inflammatory responses post-TBI | Baboons | (59, 112) |
| miR-212 | MicroRNA 212 | Radiosensitivity and immune modulation | Baboobs | (88, 109) |
| miR-322-3p | MicroRNA 322 3p | Distinguisher between high dose (6.5 Gy) and low-dose (2 Gy) sublethal groups | Human | (97) |
| miR-34b-3p | MicroRNA 34b 3p | Distinguisher between high dose (6.5 Gy) and low-dose (2 Gy) sublethal groups | Human | (97, 102) |
| miR-27a-3p | MicroRNA 27a-3p | Proliferation and growth signaling pathways | Human | (97) |
| miR-136-5p | MicroRNA 136 5p | Distinguisher between high dose (6.5 Gy) and low-dose (2 Gy) sublethal groups | Human | (97) |
| miR-17-3p, miR-17-5p | MicroRNA 17 3p/5p | Distinguisher between high dose (6.5 Gy) and low-dose (2 Gy) sublethal groups, cardiovascular atrophy caused by lethal dose of radiation, differentially expressed and associated with TGF-beta in early phase after TBI | Human | (97, 113, 114) |
| miR-187-3p | MicroRNA 187-3p | IL-10–mediated suppression of TNF-α | Human | (97) |
| miR-21 | MicroRNA 21 | Long term elevation in the brain after radiation exposure, radiation caused bystander effect, epithelium-to-mesenchymal transition, associated with anti-inflammatory responses post-TBI | Human | (98, 112, 115–117) |
| miR-145 | MicroRNA 145 | Dose dependent upregulation in AHH-1 cells and HPBLs irradiated ex vivo | Human | (99) |
| miR-145-5p | MicroRNA 145 5p | Associated with anti-inflammatory responses post-TBI | Human | (112) |
| miR-663 | MicroRNA 663 | Dose dependent upregulation in AHH-1 cells and HPBLs irradiated ex vivo | Human | (99) |
| miR-223-3p | MicroRNA 223 3p | Associated with anti-inflammatory responses post-TBI | Human | (112) |
| miR-181a-5p | MicroRNA 181a 5p | Associated with anti-inflammatory responses post-TBI | Human | (112) |
| miR-1307 | MicroRNA 1307 | Significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99) |
| miR-3197 | MicroRNA 3197 | Significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99) |
| miR-4267 | MicroRNA 4267 | Significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99) |
| miR-5096 | MicroRNA 5096 | Significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99) |
| miR-7641 | MicroRNA 7641 | Significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99) |
| miR-150 | MicroRNA 150 | Circulatory miRNAs correlated with dose of radiation, radiation-induced bystander effect, involved in adaptive response caused by radiation | Human | (97, 101, 103, 110, 118) |
| miR-1246 | MicroRNA 1246 | Targeting Lig4 and inducing bystander DNA damage, significantly upregulated by 4 Gy γ-rays and associated biological functions | Human | (99, 119) |
| miR-122-5p | MicroRNA 122 5p | Radiation-induced bystander effect, incolved in adaptive response caused by radiation | Human | (118) |
| miR-122b-3p | MicroRNA 122b 3p | Radiation-induced bystander effect, incolved in adaptive response caused by radiation | Human | (118) |
| mir-155-5p | MicroRNA 155b 5p | Elevated expression after TBI, and association with anti- and pro-inflammatory functions | Human | (120) |
| mir-378 | MicroRNA 378 | Cardiovascular atrophy caused by lethal dose of radiation | Human | (59, 113) |
| miR-16-5p | MicroRNA 16 5p | Cardiovascular atrophy caused by lethal dose of radiation | Human | (113) |
| miR-140-3p | MicroRNA 140 3p | Cardiovascular atrophy caused by lethal dose of radiation | Human | (113) |
| miR-19b-3p | MicroRNA 19b 3p | Cardiovascular atrophy caused by lethal dose of radiation | Human | (113) |
| miR-199a-3p | MicroRNA 199a 3p | Cardiovascular atrophy caused by lethal dose of radiation | Human | (104, 113) |
| miR-125-5p | MicroRNA 125 5p | Pro-inflammatory biomarker candiate post-TBI | Human | (121) |
| miR-23a-3p | MicroRNA 23a 3p | Pro-inflammatory biomarker candiate post-TBI | Human | (101, 121) |
| miR-34a-5p | MicroRNA 34a 5p | Pro-inflammatory NF-κB-mediated functions, circulatory biomarker for lung specific injury | Human | (102) |
| miR-133a/b | MicroRNA 133 a/b | Muscle development, Targeting radiation-associated gene IGF1R, biomarker for irradiated vs unirradiated NHPs | Baboons, NHP | (59, 105, 110) |
| miR-375 | MicroRNA 375 | Radioprotective efficacy of GT3, regulate pyruvate dehydrogenase kinase, isozyme 1 (Pdk1) and myotrophin (Mtpn) to control production of beta cells and insulin, biomarker for irradiated vs unirradiated NHPs | Human, mouse | (105, 110, 122) |
| miR-7-5p | MicroRNA 7 5p | Targeting EGFR and Bcl2 and inducing autophagy | Human, mouse | (123, 124) |
| miR-215 | MicroRNA 215 | Targeting radiation-associated gene IGF1R, biomarker for irradiated vs unirradiated NHPs | Human, NHP | (105, 110) |
| miR-126, miR-126a-5p | MicroRNA 126 | Radioprotective efficacy of GT3, survival indicators, radiation-induced bystander effect, involved in adaptive response caused by radiation, radiation caused fatality indicator in NHPs, distinguisher between high dose (6.5 Gy) and low-dose (2 Gy) sublethal groups, cardiovascular atrophy caused by lethal dose of radiation | Human, NHP | (97, 105, 110, 113, 118) |
| miR-30a-3p, miR-30 | MicroRNA 30a-3p | Inhibit cancer cell proliferation, Targeting radiation-associated gene IGF1R, radioprotective efficacy of GT3, survival indicators, circulatory miRNAs correlated with dose of radiation, radiation caused fatality indicator in NHPs, biomarker for lethal (8 Gy) and sublethal (6.5 Gy) groups | Human, NHP | (97, 105, 110) |
| miR-30c-5p, miR-30c | MicroRNA 30c-5p | Regulates proliferation, apoptosis, and differentiation, circulatory miRNAs correlated with dose of radiation, biomarker for lethal (8 Gy) and sublethal (6.5 Gy) groups, cardiovascular atrophy caused by lethal dose of radiation | Human, NHP | (97, 110, 113) |
| miR-29a/b/c | MicroRNA 29 a/b/c | Circulatory miRNAs correlated with dose of radiation, distinguisher of HARS groups with and without pancytopenia | Baboons, human, mouse, and NHP | (59, 110) |
| miR-200 | MicroRNA 200 | Circulatory miRNAs correlated with dose of radiation | Human, mouse, and NHP | (110) |
| miR-320 | MicroRNA 320 | Circulatory miRNAs correlated with dose of radiation | Human, mouse, and NHP | (110) |
Figure 2:
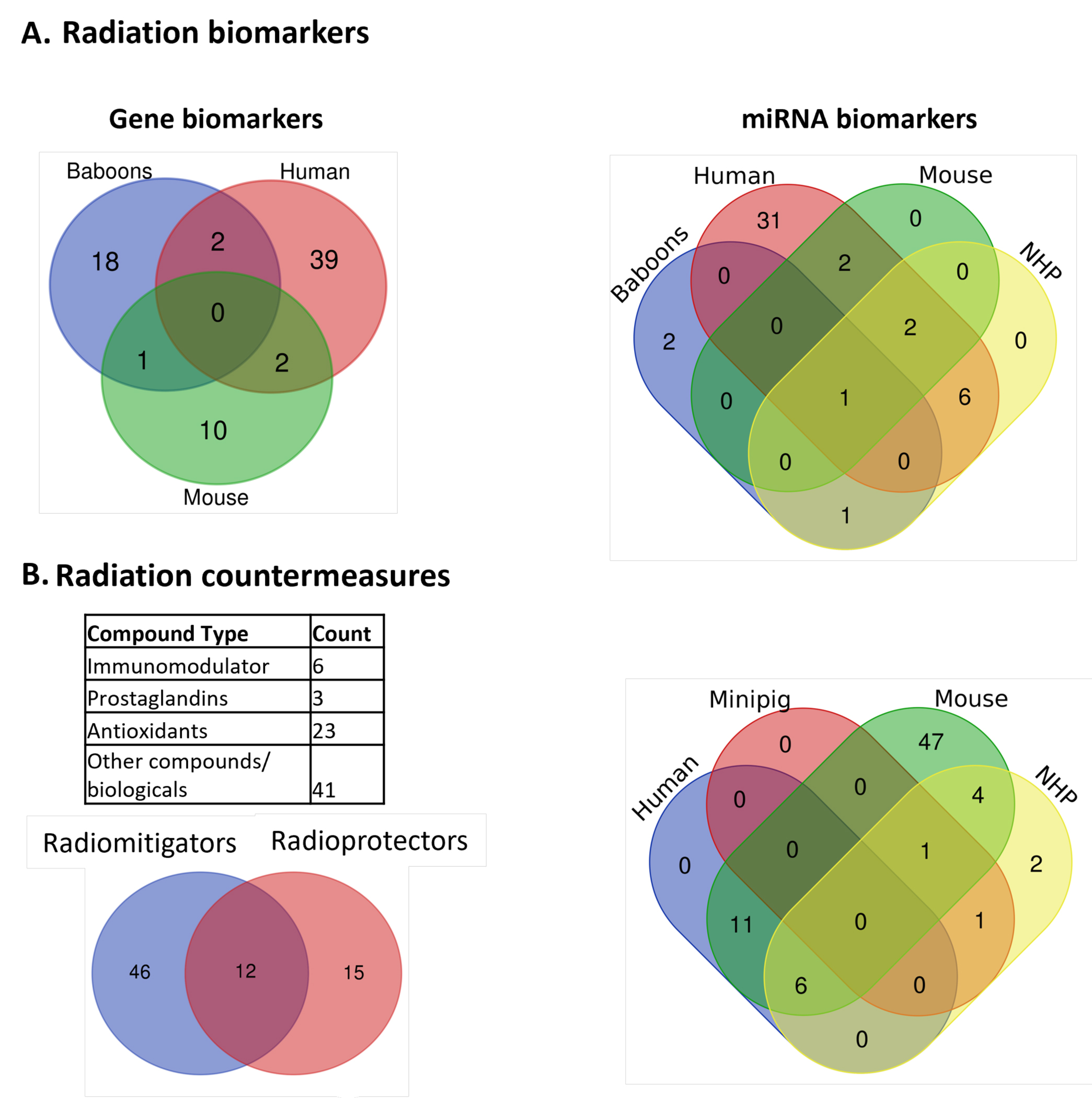
A) Summary of genes and miRNAs biomarkers studied in different animal models. The genes or miRNAs studied in different animal models such as baboons, human, mouse, and NHP were compared. B) Statistics of radiation countermeasures, their classification, and experimental studies in different animal models. The drugs studied in animal models e.g. NHP, human, mouse/rat, and minipig were compared to identify unique and common ones in each model.
Radiation effects on mammalian microbiota:
Microorganisms are essential to host cell maintenance in health and disease; therefore, the human gut microbiota has attracted increasing attention in recent years. The gastrointestinal (GI) tract hosts the largest and most diverse microbiome in the human body, where the microbiome plays a vital role in several metabolic processes that are essential for human health. ARS occurs after TBI, and death is frequently attributed to poor hematopoietic recovery and/or death of epithelial cells lining the GI tract. The GI tract is the fastest-renewing adult tissue, making it highly sensitive to radiation (125). It also harbors a diverse microbial community comprising 10 to 100 trillion microorganisms, raising the possibility that the gut microbiota is highly prone to radiation-induced damage (126–130). Although some studies have shown associations between gut microbiota and radiation-induced damage (130–132), possible mechanisms are poorly understood.
Most cancer patients undergoing pelvic irradiation experience side effects such as fatigue and diarrhea; however, the impact of radiation on the microorganisms colonizing the GI tract’s mucosal surfaces is unexplored. Severe diarrhea may also complicate radiotherapy as there is no current clinical or experimental information on the role of gut microbiota in this pathogenesis. A study focusing on the fecal microbiota of patients receiving pelvic radiotherapy and suffering from acute post-radiotherapy diarrhea revealed changes in microbial diversity. In healthy volunteers and patients without diarrhea, microbial diversity was stable throughout the study. However, patients exhibiting diarrhea showed a progressive modification in their microbial diversity. Patients who developed diarrhea harbored bacterial phyla, Actinobacteria, and some Firmicutes, which were either not detected or least abundant in patients who did not develop diarrhea. On the other hand, patients who developed diarrhea had a reduced abundance of some Clostridia species, which are members of the phylum Firmicutes (131).
Irradiation-induced changes in the bacterial compositions of the large and small intestines at the genus level are one of the hallmarks of radiation injury. For instance, irradiation with a single dose of 8 Gy increased several genera, including Alistipes, Lactobacillus, and Akkermansia in the large intestine and Corynebacterium and Turicibacter in the small intestine. Compared with the corresponding unexposed control group, the abundance of the genera Prevotella was lower in the irradiated large intestine, while levels of Alistipes were lower in the irradiated small intestine (132).
The gut microbiota may contribute to radiation-induced pathogenesis, and it differs between patients with and without radiation enteropathy. Reports also indicated that microbial diversity decreased over time in patients with rising radiation enteropathy. A consistent association between bacterial diversity and late radiation enteropathy was observed, and higher counts of Clostridium IV, Roseburia, and Phascolarctobacterium were significantly associated with radiation enteropathy (133).
A study that analyzed ‘elite survivors’ in a population of mice that recovered from exposure to total-body radiation at high-dose (8.0 to 9.2 Gy) had overrepresented members of the bacterial taxa, Lachnospiraceae and Enterococcaceae. These microorganisms were associated with post-irradiation restoration of hematopoiesis and GI repair. These bacteria were also found to be more abundant in leukemia patients undergoing radiotherapy, who also displayed milder GI dysfunction (134). A different study in the radiation enteritis (RE) mice model, considering a single dose of 18 Gy X-ray irradiation at a rate of 5 Gy/min, identified an abundance of bacteria representing 12 genera from Alloprevotella, Alistipes, Akkermansia, Bacteroides, Dubosiella, Eggerthellaceae, Enterococcus, Escherichia-Shigella, Lactobacillus, Lachnospiraceae, Muribaculaceae, and Rikenellaceae after radiation exposure (135). These studies highlight the importance of GI microbiota in radiation-induced pathogenesis and potentially pave the way for novel treatment protocols involving alterations of human gut microbiota.
In a recent study, the bacterial 16S rRNA amplicon sequencing and untargeted metabolomics were accomplished to investigate the effects of BIO 300, a promising radiation countermeasure under advanced development, on the gut microbiome and metabolome of CD2F1 male mice exposed to 60Co gamma-radiation (9.2 Gy, 0.6 Gy/min, LD70/30 dose). Irradiation changed the ratio of Firmicutes/Bacteroidetes and also decreased the relative abundance of Lactobacillus in BIO 300-treated as well as control mice after irradiation (136). The ratio returned to pre-irradiated levels in BIO 300-treated animals by day 14 post-irradiation. Concurrently, there was corrective shifts in metabolic pathways that were altered after radiation exposure. In brief, these results demonstrated that irradiation resulted in a relative depletion of commensals like Lactobacillus leading to an inflammatory metabolic phenotype in untreated control mice while the BIO 300-treated mice demonstrated alleviation of this condition by restoring normal gut microbiota.
Alterations to the gut microbiome of NHPs exposed to high doses of radiation detected several bacterial species. Post-irradiation, significant increases in the relative abundance of Treponema and Helicobacter genera were observed in rhesus macaques when exposed to 6.8 Gy of radiation (137). In another study, relative abundances of Prevotella, Lactobacillus, Clostridium XIVa, Oscillibacter, and Treponema were found to be highly correlated with radiation intensity. Prevotella, Oscillibacter, and Treponema were closely associated with the overall survival, while Streptococcus was associated with death in macaques (138). The Firmicutes/Bacteriodetes ratio, a factor related to the disruption of metabolic homeostasis, declined from 1.2 to below one post-radiation exposure at 7.4 Gy. Actinobacillus, Bacteroides, Prevotella, and Veillonella genera were significantly increased by more than 2-fold, and Acinetobacter and Aerococcus genera were diminished by greater than 10-fold post-irradiation (139).
Other omic profiles altered by radiation:
Altered lipid profiles:
Concerns over a potential exposure of IR in large populations have emphasized the need for rapid and reliable biodosimetry methods to determine the absorbed dose and required triage. Indirect consequences of radiation exposure include the generation of reactive oxygen species (ROS) through water hydrolysis, enhanced NADPH oxidase activity, compromised mitochondrial function leading to damaged cellular lipids. Since lipidomics is a powerful technique for large-scale identification and quantification of lipids, rapid identification of altered lipid molecules are promising biomarkers for biodosimetry.
Experiments conducted in mice and NHP models to characterize lipidomic profiles that respond to radiation exposure have helped identify important radiation biomarkers. A study that analyzed global lipid profiling from mouse serum identified significant molecular alterations following γ-radiation exposure. A Low-abundance of oxygenated, polyunsaturated fatty acids (PUFAs) was observed after irradiation at 8 Gy of γ rays (dose rate- 1.67 Gy/min). Exposure to γ-radiation induced a significant increase in the serum levels of phosphatidylcholines (PCs) and arachidonic acid, while levels of diacyl PCs carrying PUFAs were decreased (140).
Radiation exposure in NHPs caused significant perturbations in lipid metabolism, affecting all major lipid species, including free fatty acids, glycerolipids, glycerophospholipids, and esterified sterols. In particular, a significant increase in the levels of PUFA-containing lipids and polyunsaturated triglycerides in the serum of NHPs exposed to 10 Gy radiation was identified. These TGs contained primarily arachidonic acid and docosahexaenoic acid acyl moieties (141). Temporal changes in the serum lipidome from hours to days after radiation exposure in NHPs have also been reported. Marked lipidomic changes occurred within 24 h post-irradiation with 6.5 Gy, along with increased levels of cytokine, free fatty acids, monoacylglycerides, and C-reactive protein. Simultaneous decreases in di- and tri-acylglycerides, sphingomyelins, lysophosphatidylcholines, and esterified sterols were also observed in this study (142). Decreased sphingomyelins and increased levels of lysophosphatidylcholines may be important markers for biodosimetry within days following irradiation. The biphasic and dynamic alterations linked to radiation exposure in the serum lipidome emphasized the importance of determining the long-term temporal response of these promising radiation biomarkers (142).
Global lipidomic analysis using ultra-high performance liquid chromatography-mass spectrometry (UHPLC-MS) performed on intestinal tissue specimens from acute RE at 18 Gy X-irradiation at a dose rate of 5 Gy/min revealed distinct lipid metabolite fingerprints. Several lipids were significantly altered by the occurrence of peroxidation in the acute or chronic RE group compared with the control, which included glucosylceramide, phosphatidylethanolamine, lysophosphatidylcholine, lysophosphoglycerol, lysophosphatidyl-inositol, phosphatidylcholinephosphatidylglycerol, phosphatidylinositol, and sphingosine (135). Metabolism of sphingolipids is a very complex process that involves the catabolism of several lipids by enzymes such as glucosylceramide synthase and sphingosine kinase 1 (143). Though current technologies can perform lipid profiling for various exposure conditions, data interpretation becomes very difficult because of their complex biological roles; hence, lipid profiling is not widely used for biomarker identification in radiation studies.
Altered metabolic profiles:
The rapid identification of radiation-induced metabolomic markers in biological samples, such as urine, blood, tissue, and saliva, have also been investigated for developing countermeasures to a radiological or nuclear public health emergency (144). Radiation metabolomics has primarily focused on the mass spectrometry (MS) analysis of samples from radiation-exposed animals to monitor an altered biological response. In this manner, metabolic studies are valuable for understanding the systems-level biological impact of radiation exposure. Moreover, radiosensitive biodosimetry methods were developed to accurately measure exposure levels and understand the impact of radiation exposure on tissues and organs. Metabolomic studies helped to understand changes in metabolite levels in normal and irradiated organs in mice, NHPs, and humans. For example, Gao et al. measured 31 low molecular weight metabolites such as lactamide, 1,2,4-benzenetriol, taurine, and piperine to assess the metabolic changes in rat lungs when exposed to single dose of 10 or 20 Gy radiation (145). Similarly, age-dependent correlation was observed for N(1)-acetylspermidine and 2’-deoxyuridine, but a poor correlation for elevated xanthine and N(1)-acetylspermidine in older irradiated mice when exposed to 3 Gy γ-radiation (146). These findings suggested that aging might be associated with higher levels of oxidative stress and a decline in DNA damage-repair efficiency while also implying a specific role for polyamine metabolism. Other studies have identified a time- and dose-dependent response in the GI metabolites to 4–8 Gy of IR in mice (147) or in the urinary metabolites, β-thymidine and N-hexanoylglycine, in mice when exposed to doses of 0, 3 and 8 Gy (2.57 Gy/min) for 24 h (148). Altered metabolic profiles of tissues, including bone marrow, ileum, liver, muscle, and lung, were also observed within 12 h of radiation exposure (6 Gy, dose rate 0.92 Gy/min), which are associated with DNA methylation, energy metabolism, and amino acid metabolism (149). Li and colleagues analyzed T-cells from irradiated mice when exposed to 0.1, 0.5, or 3 Gy TBI at a dose rate of 1.7 Gy/min, and concluded that radiation effects were correlated with a decrease in key metabolic pathways such as glycolysis and energy metabolism (150).
A sex-specific study confirmed that thirteen dose-responsive metabolic biomarkers that include L-carnitine, xanthine, L-acetylcarnitine, and xanthosine were increased in male NHPs when exposed to 2, 4, 6, 7 and 10 Gy for 7 days (151). A follow-up study demonstrated that several derivatives of carnitine and acylcarnitines were also significantly altered in NHPs after exposure to γ-radiation (152). Numerous LC-MS-based studies have been used to measure radiation-induced metabolic markers pertaining to energy metabolism, DNA damage, organ injuries, and inflammation (153–155). Metabolic profiles have also been evaluated for their use in the therapeutic intervention of post-radiation exposure. There is a recent report for conducting a study in irradiated NHP to evaluate metabolic changes in plasma and plasma-derived exosomes. NHP were exposed to gamma-radiation and blood samples were collected at various time points in relation to 5.8 Gy or 6.5 Gy TBI. Exosomes were isolated and analyzed for untargeted metabolomic and lipidomic profiling. Plasma profiling demonstrated markers of dyslipidemia, inflammation and oxidative stress post-irradiation. Based on the differences in metabolite composition between plasma and exosomes, it was suggested that exosomal profiling may augment the identification of low abundance biomarkers that would otherwise be obscured in plasma (156). In another study with 7.2 Gy TBI NHP serum, authors reported temporal fluctuations of metabolites within 96 h post-irradiation and higher fold changes of altered metabolites at 7.2 Gy compared to 6.5 Gy at 24 h post-irradiation. These findings highlight the importance of biofluid collection timepoint for successful interpretation of serum metabolic profiles (157, 158). The profound elevation of long-chain acylcarnitines in irradiated NHPs across multiple tissue types underscored the role of this class of metabolites as a generic indicator of radiation-induced tissue injury (159).
Metabolomics studies have also been applied to biodosimetry-based analyses (160, 161). For example, in human cell line studies, depleted metabolites such as glutathione, adenosine monophosphate (AMP), and nicotinamide adenine dinucleotide (NAD) were linked to oxidative stress and DNA repair pathways 1 h after radiation exposure to IR doses of 0.5 to 8.0 Gy (162).
Low-dose irradiated human keratinocytes also exhibited a disruption in energy metabolism when exposed to 0.03, 0.1 and 2 Gy of X-ray for 3, 24 and 48 h (163). An integrated study involving transcriptomics and metabolomics of irradiated human bronchial epithelial (HBE) cells measured 326 differentially expressed genes and 147 altered metabolites 24 h after 4 Gy exposure. These omics results suggested that post-irradiation cellular metabolism may be regulated by p53 (164). There is also a report for metabolomic and lipidomic profiles in multiple tissues (liver, kidney, jejunum, heart, lung, and spleen) of NHP exposed to 7.2 Gy γ-TBI. There were robust metabolic changes in the kidney and liver and modest changes in other tissues. Overall, metabolomics has identified numerous potential biomarkers that may be useful for analyzing, diagnosing, or treating ARS or other symptoms of radiation exposure.
Altered proteomic profiles:
Like metabolomics profiling, proteomics analyses can also be exploited to measure the detrimental effects of radiation exposure. Radiation might alter regulatory networks or cause proteomic modifications such as decarboxylation, disulfide bonds, or aggregation (165) and subsequently affect the steady-state levels of specific proteins. Several studies have analyzed different tissues to measure changes in protein abundance resulting from radiation exposure. For example, 19 differentially expressed proteins associated with biological functions such as the DNA damage response, stress response, and cytoskeleton system were observed in the intestines of irradiated mice (exposure to 9 Gy for 24 h and 72 h) when analyzed using two-dimensional gel electrophoresis (166). A single dose exposure study on the liver proteome of mice (from 0.02 to 1 Gy) showed a remarkable downregulation of glycolysis and pyruvate dehydrogenase availability. A subsequent long-term outcome was an increase in liver inflammation (167). Another study focused on the spleen proteome of tocotrienol (GT3) treated mice 24 h prior to 7 Gy TBI, which showed a difference in the expression of several proteins, including the upregulation of Wnt signaling pathways (168). This study also identified significant alterations in the levels of metabolic enzymes such as aldehyde dehydrogenase and propionyl coenzyme A, guanylate cyclase, and glycine amidinotransferase, which suggested possible changes in the carbon and amino acid metabolism due to radiation. Other differentially expressed proteins suggested that cell signaling proteins attenuated radiation-induced injuries.
Previous studies characterized the radiation-induced proteome changes in different body fluids such as urine and blood plasma (169) from NHPs, when exposed to 6.7 Gy and 7.4 Gy doses of radiation (169, 170). Some of the differentially expressed proteins based on dose and time points in both the samples from the same animal included ferritin (F6ZV45), angiotensinogen preproprotein (G7MFR4), putative uncharacterized proteins (G7NQN4 and G7MJ28), angiotensin-converting enzyme isoform 1 (H9FJ99), and other uncharacterized proteins (F6TLR3, F7DHQ1, and F7GRY2). Further, the resulting urine proteome profile identified many proteins associated with cell adhesion, disease progression, and key metabolic pathways (170). Proteomic changes in the serum of high-dose irradiated NHPs after administrating BIO 300, a synthetic genistein nanosuspension, identified upregulated proteins, e.g., tubulin α chain (B3KT06), cDNA FLJ57036 (B4E3P1), CP protein (A5PL27), glutathione peroxidase (V9HWN8), carbonic anhydrase I (V9HWE3), glutathione S-transferase pi 1 (V9HWE9), which helped understand the metabolic changes associated with drug effects (171).
Several previous studies also focused on understanding the proteome changes in human tissues such as endothelial cells (exposure to <2.5 Gy of γ-radiation) (172, 173), peripheral lymphocytes (exposure to 1, 2, and 4 Gy γ-radiation) (174), skin fibroblast cell lines (exposure to 10 Gy γ-radiation) (175). Proteomics-based studies have also suggested that tumor-derived factors are upregulated by radiation exposure and may serve as potential therapeutic targets during radiation treatment (176). The changes in the proteomic profiles due to radiation exposure is associated with biological phenotype such as inflammation, toxicity, immune response and retinoic acid signaling in human (177, 178). Some studies have noted that protein expression changes were associated with radiotherapy and tumor resistance in different organs such as the breast, lungs, and brain (176, 179–182). A study that treated breast cancer cell lines with a single dose of 10 Gy of γ-radiation showed the top differentially regulated proteins such as C-type mannose receptor 2, arginino-succinate synthase 1, gelsolin, peroxiredoxin 5, and cathepsin D (183), while the lung cells in the same study showed cofilin-1, HSPB1, annexin A4, and vimentin as the top upregulated proteins (184). Overall, these reports have demonstrated the potential of proteins as reliable biomarkers for high-dose radiation exposure.
Radiation countermeasures: radioprotectors and radiomitigators
As discussed above, exposure to high or low doses of radiation can affect human health in many ways, primarily resulting in ARS, cutaneous radiation syndrome (CRS), neurovascular syndrome, or even death. These health effects are caused by radiation-induced damage such as genomic alterations or direct DNA damage, destruction of blood-forming stem cells, and damaging the immune system and other metabolic functions.
Current drug discovery efforts are focused on developing radioprotectors and radiomitigators for their use during pre- and post-radiation exposure treatments, respectively. To date, several agents have been tested as potential radioprotectors and/or radiomitigators to counter ARS. The manifestation of ARS is related to disorders of the GI, neurovascular, hematopoietic, and cutaneous systems (185).
Radiomitigators
Under this category, the thrombomodulin (Thbd)-activated protein C (aPC) pathway has been shown to stimulate blood cell production and help attenuate radiation injuries in mice (dose rates- 1.37 Gy/min and 0.52 Gy/min for two separate mouse models) (186). In general, aPC has an association with several cellular activities such as anti-coagulation, anti-inflammation in blood, and other cytoprotective properties such as endothelial barrier protection, protection against vascular leakage, inflammation, apoptosis, and inflammasome activation (187). The drug TP508 (rusalatide acetate, Chrysalin) also mitigates the effects of radiation by activating radioresistant stem cells in the intestines and colonic regions of mice when treated 24 h after lethal radiation exposure of 9 Gy (188). In another study, TP508 was found to stimulate cellular events linked with the repair of bone, skin, and muscle tissues in rats (189). Among the FDA-approved radiomitigators, Neupogen and Neulasta are the most efficient for treating radiation damage and neutropenia (190, 191). These drugs help improve granulocyte colony-stimulating factor (G-CSF) pharmacokinetics and subsequently empower the immune response against radiation injuries (192). Another FDA-approved drug, Leukine (Sargramostim), has also shown potential radiomitigatitve effects to treat neutropenia in radiation-exposed patients (193–196). Apart from G-CSF, NPLATE (Romiplostim) is the first FDA-approved thrombopoietin receptor agonist used as radiomitigator to treat low platelet counts due to acute radiation exposure as studied in (197). In a recent study, Singh and colleagues analyzed metabolic and lipidomic profiles in the serum of NHPs (exposed to 7.2 Gy of γ-radiation) after treatment of a candidate drug, Ex-Rad (ON01210), which showed significant alterations in biochemical pathways towards the recovery of radiation-injured organs (198). Several other studies and reviews were focused on repurposing of drugs as radiomitigators (199–203).
Radioprotectors
Unlike radiomitigators, radioprotectors are administered before radiation exposure and investigated for their counter effects against ARS in different models, including mice and NHPs. BIO 300 is one example of a promising candidate that causes alterations in the metabolic profile, which further helped identify potential biomarkers associated with lung and other radiation-induced tissue injuries in the mice model when exposed to a single dose of 11.0 or 12.5 Gy whole thorax lung irradiation (204–206). In NHPs, serum-based global lipidomics and metabolomic changes were observed after treatment with BIO 300, which were again used to understand the regulation of different pathways associated with the radiation-caused injuries. Specifically, significant alterations in the levels of important metabolites such as tyrosine, glycerophosphoserine, glycerophosphocholine, and phenylalanine were observed (207). A similar study with proteomics concluded that BIO 300 causes elevation of actin by minimizing its nitration in NHP model (171).
Similarly, amifostine (WR-2721), as a radioprotecting candidate, showed significant positive impacts against ARS in humans. Metabolomics and lipidomics analyses of control and amifostine-treated mice, when exposed to whole-body at 9.6 Gy radiation, revealed that radiation exposure caused the dysregulation of 1,614 metabolites in bone marrow, jejunum, and lung samples. Further analysis concluded that bone marrow exhibited a heightened response to the protective outcomes of amifostine, while jejunum and lung had a modest response (208). In another study, reduced levels of metabolites such as hypoxanthine, glutamic acid, and L-valine due to radiation exposure was mitigated, while, some elevated metabolites like PS (18:0/20:4) and L-arginine were corrected after treatment by amifostine in mice, which were exposed to 9.6 Gy γ-radiation (209). Similarly, MS and NMR studies of serum of mice (exposed to 14 Gy γ-radiation) and NHPs (exposed to 5.8 Gy or 7.2 Gy γ-radiation, dose rate- 0.6 Gy/min) showed dysregulation of twenty-three pathways after exposure to γ-radiation. These altered metabolic pathways mainly included lipid biosynthesis, glycolysis, and nucleotide metabolism (210). This study further observed reversed metabolic signatures of ARS progression through pretreatment of Amifostine, showing the potential of the drug as a radioprotector; however, side effects such as nausea and vomiting are still a major concern. Amifostine has been tried to treat ARS but showed toxic side effects, however later approved by FDA for xerostomia (211–213).
Antioxidants, especially the group of vitamin E compounds, have been widely studied as radioprotectors (214, 215). For example, α-tocopherol enhanced the survival of mice when administered 24 h before γ-irradiation (dose rate of 0.6 Gy/min) (215). Similarly, GT3 has also been investigated as radioprotective agent. This agent exerted metabolic changes in NHPs, which were linked with increasing levels of antioxidants, suggesting a protective benefit to countermeasure radiation-induced injuries (216). Global metabolomic changes were analyzed in another study using irradiated NHP treated with GT3. Analysis of serum samples identified several altered metabolites after irradiation, including compounds involved in fatty acid beta-oxidation, purine catabolism, and amino acid metabolism. A machine-learning algorithm, Random Forest, separated control, irradiated GT3-treated, and irradiated vehicle-treated NHPs at 12 h and 24 h. Primary metabolites validated included carnitine/acylcarnitines, amino acids, creatine, and xanthine. Overall, GT3 administration reduced high fluctuations in serum metabolite levels, suggesting an overall beneficial effect on animals exposed to radiation (158). Recently conducted studies with serum and jejunum samples of GT3-treated mice exposed to 11 Gy TBI demonstrated the restoration of irradiation-induced proteomic changes by GT3 (217, 218). Likewise, an injection of selenium-containing compounds, such as sodium selenite or selenomethionine, resulted in the improved survival of mice after radiation exposure.
Agents that serve both as radiomitigators or radioprotectors
Some of the drugs that have been investigated as radioprotectors and radiomitigators to modulate ARS effectively include glucans, 5-androstenediol (5-AED), and meloxicam (219, 220). In animal trials, β-glucan has shown potential both as a radioprotector and a radiomitigator when mice were exposed to the doses of 6, 7, and 8 Gy γ-radiation and doses of 4 Gy (dose rate- 0.15 Gy/min) in two different studies (221, 222). β-glucan combined with other radioprotectors, such as cystamine or WR-2721, showed a positive impact in protecting from radiation damage (223). The administration of β-glucan with WR-2721 and selenium also resulted in favorable outcomes (224). Several other studies revealed similar successes in the use of β-glucan either pre- or post-irradiation to treat radiation toxicity and improve animal survivability (225–228). A pre- or post-irradiation treatment with 5-AED was also effective in modulating the immune response to counter the impact of radiation. 5-AED stimulated myelopoiesis and the recovery from infections in mice (exposed to 3 Gy total-body γ-radiation) and NHPs (TBI 4 Gy γ) (219, 229). Increased G-CSF production in 5-AED treated mice 24–48 h prior to TBI with 7.5 Gy (dose rate- 0.6 Gy/min), synergistically improved the activation of monocytes, granulocytes, and NK cells, along with an increase in the number of innate immune cells (230–232). 5-AED is an open Investigational New Drug (IND) used as a radioprotector or radiomitigator for radiation-related injuries (233).
The mechanism of action and efficacy of a variety of such compounds and biomolecules were comprehensively discussed in previous reviews (202, 234, 235). Various highlighted compounds, especially the repurposed ones, that can be used to modulate ARS pre- and post-radiation exposure, which include immunomodulators, are listed in Supplementary Table S1. A short summary of radiation countermeasures studied in different animal models is provided in Figure 2B. Different radiation drugs were categorized into four main classes i.e. immunomodulator, prostaglandin, antioxidant and other biologicals. Interestingly, our literature search showed 12 compounds that have been studied as radioprotectors and radiomitigators [Figure 2B and Table S1].
In the recent past, countermeasures have been used for studying metabolomics in NHP models with different doses of TBI. The metabolomic profile in serum samples of NHPs treated with Ex-Rad after irradiation (7.2 Gy, 0.6 Gy/min, LD70/60) has been investigated. Two different dose administration schedules (24 and 36 h post-irradiation as well as 48 and 60 h post-irradiation) were used to investigate the global profiling approach. Alterations in the biochemical pathways relating to inflammation and oxidative stress after irradiation were alleviated in animals that received Ex-Rad treatments (201).
Conclusions
Decades of research into the effects of radiation damage in several model systems, along with the generation, analysis, and interpretation of radiation-altered omics data, have identified several promising candidates as biomarkers. These biomarkers were identified from various data sources, including genomics, transcriptomics, proteomics, metabolomics, lipidomics, and microbiome-based studies. Here, we discussed current progress in the identification of these biomarkers for radiation exposure-associated disease manifestations. Several of these radiation-induced transcriptomic signatures, including GADD45, PCNA, CCNA2, CDKN1A, and MDM2, could be employed as rapid and cost-effective biomarkers to detect the extent of radiation damage. Similarly, lipid biomarkers, such as glucosylceramide, phosphatidylethanolamine, and sphingosine have the potential to be population-based biodosimetry biomarkers, owing to their dynamic and reliable response to radiation damage and the availability of robust analytical assays for detection. There has also been significant progress in identifying proteomic and metabolic markers associated with radiation damage. In this context, radiation-altered proteins such as FDXR, DDB2, and ACTN1, and metabolites such as β-thymidine and N-hexanoylglycine were associated with dysregulated molecular pathways that included glycolysis, DNA methylation, central carbon metabolism, and nucleic acid and amino acid metabolism. Furthermore, these pathway-level alterations and the associated system-level phenotypic changes may provide a rapid assessment of radiation exposure. The use of multiple biomarkers from different data sources would help enhance the prediction accuracy of diagnostic approaches compared to using a single biomarker.
Biomarkers of radiation injuries are extensively used for optimizing and tracking the damage induced by radiotherapy (236, 237). Since differences in tissue sensitivity to radiation exposure are reported, the optimization of an accurate dose for each tissue is essential for the efficacy of radiotherapy with minimal side effects. Also, identifying the extent of exposure associated with unfortunate nuclear accidents, such as the Chernobyl and the Fukushima Daiichi incidents or occupational radiation exposures, requires continuous monitoring using the biomarkers discussed here. To summarize, radiation biomarkers are essential for monitoring both unforeseen radiation accidents as well as the side effects of calibrated exposures such as radiation therapy. Recent advancements in high-throughput biomedical research technologies that generate multi-omics data coupled with the development of efficient data-analytical tools and computational algorithms have the potential to detect novel and reliable biomarkers associated with radiation exposure. Analysis of publicly available data and literature mining of radiation-injury data will also provide new therapeutic targets to help develop appropriate radiation countermeasures.
Supplementary Material
Acknowledgements
This material is based upon work supported by the Defense Health Agency and U.S. Strategic Command under Contract No. FA4600-12-D-9000 and Contract No. FA4600-18-D-9001, and in part from the Nebraska Center for Integrated Biomolecular Communication (P20 GM113126, NIGMS). Any opinions, findings and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the Defense Health Agency, U.S. Strategic Command, or 55th Contracting Squadron. Similarly, the opinions or assertions contained herein are the private views of the authors and are not necessarily those of the Uniformed Services University of the Health Sciences, or the Department of Defense. The mention of specific therapeutic agents does not constitute endorsement by the U.S. Department of Defense, and trade names are used only for the purpose of clarification. We apologize to those having contributed substantially to the topics discussed herein that we were unable to cite because of space constraints.
References:
- 1.Nikitaki Z, Hellweg CE, Georgakilas AG, Ravanat JL, Stress-induced DNA damage biomarkers: applications and limitations. Front Chem 2015; 3, 35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.!!! INVALID CITATION !!! (2).
- 3.Oh YJ, Kwak MS, Sung MH, Protection of Radiation-Induced DNA Damage by Functional Cosmeceutical Poly-Gamma-Glutamate. J Microbiol Biotechnol 2018; 28, 527–33. [DOI] [PubMed] [Google Scholar]
- 4.Santivasi WL, Xia F, Ionizing radiation-induced DNA damage, response, and repair. Antioxid Redox Signal 2014; 21, 251–9. [DOI] [PubMed] [Google Scholar]
- 5.Arlt MF, Rajendran S, Birkeland SR, Wilson TE, Glover TW, Copy number variants are produced in response to low-dose ionizing radiation in cultured cells. Environ Mol Mutagen 2014; 55, 103–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Costa EOA, Pinto IP, Goncalves MW, da Silva JF, Oliveira LG, da Cruz AS, et al. , Small de novo CNVs as biomarkers of parental exposure to low doses of ionizing radiation of caesium-137. Sci Rep 2018; 8, 5914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li W, Olivier M, Current analysis platforms and methods for detecting copy number variation. Physiol Genomics 2013; 45, 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ricarte-Filho JC, Li S, Garcia-Rendueles ME, Montero-Conde C, Voza F, Knauf JA, et al. , Identification of kinase fusion oncogenes in post-Chernobyl radiation-induced thyroid cancers. J Clin Invest 2013; 123, 4935–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Adewoye AB, Lindsay SJ, Dubrova YE, Hurles ME, The genome-wide effects of ionizing radiation on mutation induction in the mammalian germline. Nat Commun 2015; 6, 6684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gollapalle E, Wang R, Adetolu R, Tsao D, Francisco D, Sigounas G, et al. , Detection of oxidative clustered DNA lesions in X-irradiated mouse skin tissues and human MCF-7 breast cancer cells. Radiat Res 2007; 167, 207–16. [DOI] [PubMed] [Google Scholar]
- 11.Georgakilas AG, Bennett PV, Wilson DM 3rd, Sutherland BM, Processing of bistranded abasic DNA clusters in gamma-irradiated human hematopoietic cells. Nucleic Acids Res 2004; 32, 5609–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Georgakilas AG, O’Neill P, Stewart RD, Induction and repair of clustered DNA lesions: what do we know so far? Radiat Res 2013; 180, 100–9. [DOI] [PubMed] [Google Scholar]
- 13.Rube CE, Dong X, Kuhne M, Fricke A, Kaestner L, Lipp P, et al. , DNA double-strand break rejoining in complex normal tissues. Int J Radiat Oncol Biol Phys 2008; 72, 1180–7. [DOI] [PubMed] [Google Scholar]
- 14.Redon CE, Nakamura AJ, Zhang YW, Ji JJ, Bonner WM, Kinders RJ, et al. , Histone gammaH2AX and poly(ADP-ribose) as clinical pharmacodynamic biomarkers. Clin Cancer Res 2010; 16, 4532–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Harutyunyan T, Hovhannisyan G, Sargsyan A, Grigoryan B, Al-Rikabi AH, Weise A, et al. , Analysis of copy number variations induced by ultrashort electron beam radiation in human leukocytes in vitro. Mol Cytogenet 2019; 12, 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Flunkert J, Maierhofer A, Dittrich M, Muller T, Horvath S, Nanda I, et al. , Genetic and epigenetic changes in clonal descendants of irradiated human fibroblasts. Exp Cell Res 2018; 370, 322–32. [DOI] [PubMed] [Google Scholar]
- 17.Todorova PK, Fletcher-Sananikone E, Mukherjee B, Kollipara R, Vemireddy V, Xie XJ, et al. , Radiation-Induced DNA Damage Cooperates with Heterozygosity of TP53 and PTEN to Generate High-Grade Gliomas. Cancer Res 2019; 79, 3749–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chiche A, Moumen M, Romagnoli M, Petit V, Lasla H, Jezequel P, et al. , p53 deficiency induces cancer stem cell pool expansion in a mouse model of triple-negative breast tumors. Oncogene 2017; 36, 2355–65. [DOI] [PubMed] [Google Scholar]
- 19.Joo KM, Jin J, Kim E, Ho Kim K, Kim Y, Gu Kang B, et al. , MET signaling regulates glioblastoma stem cells. Cancer Res 2012; 72, 3828–38. [DOI] [PubMed] [Google Scholar]
- 20.Deininger MW, Bose S, Gora-Tybor J, Yan XH, Goldman JM, Melo JV, Selective induction of leukemia-associated fusion genes by high-dose ionizing radiation. Cancer Res 1998; 58, 421–5. [PubMed] [Google Scholar]
- 21.Kosik P, Durdik M, Jakl L, Skorvaga M, Markova E, Vesela G, et al. , DNA damage response and preleukemic fusion genes induced by ionizing radiation in umbilical cord blood hematopoietic stem cells. Sci Rep 2020; 10, 13722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ahmed EA, Agay D, Schrock G, Drouet M, Meineke V, Scherthan H, Persistent DNA damage after high dose in vivo gamma exposure of minipig skin. PLoS One 2012; 7, e39521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chua ML, Somaiah N, Bourne S, Daley F, A’Hern R, Nuta O, et al. , Inter-individual and inter-cell type variation in residual DNA damage after in vivo irradiation of human skin. Radiother Oncol 2011; 99, 225–30. [DOI] [PubMed] [Google Scholar]
- 24.Lang F, Li X, Zheng W, Li Z, Lu D, Chen G, et al. , CTCF prevents genomic instability by promoting homologous recombination-directed DNA double-strand break repair. Proc Natl Acad Sci U S A 2017; 114, 10912–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hilmi K, Jangal M, Marques M, Zhao T, Saad A, Zhang C, et al. , CTCF facilitates DNA double-strand break repair by enhancing homologous recombination repair. Sci Adv 2017; 3, e1601898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leysen H, van Gastel J, Hendrickx JO, Santos-Otte P, Martin B, Maudsley S, G Protein-Coupled Receptor Systems as Crucial Regulators of DNA Damage Response Processes. Int J Mol Sci 2018; 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Weng Z, Fluckiger AC, Nisitani S, Wahl MI, Le LQ, Hunter CA, et al. , A DNA damage and stress inducible G protein-coupled receptor blocks cells in G2/M. Proc Natl Acad Sci U S A 1998; 95, 12334–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.van Gastel J, Leysen H, Santos-Otte P, Hendrickx JO, Azmi A, Martin B, et al. , The RXFP3 receptor is functionally associated with cellular responses to oxidative stress and DNA damage. Aging (Albany NY) 2019; 11, 11268–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chadwick W, Martin B, Chapter MC, Park SS, Wang L, Daimon CM, et al. , GIT2 acts as a potential keystone protein in functional hypothalamic networks associated with age-related phenotypic changes in rats. PLoS One 2012; 7, e36975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sanders JT, Freeman TF, Xu Y, Golloshi R, Stallard MA, Hill AM, et al. , Radiation-induced DNA damage and repair effects on 3D genome organization. Nat Commun 2020; 11, 6178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Meadows SK, Dressman HK, Daher P, Himburg H, Russell JL, Doan P, et al. , Diagnosis of partial body radiation exposure in mice using peripheral blood gene expression profiles. PLoS One 2010; 5, e11535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Paul S, Amundson SA, Development of gene expression signatures for practical radiation biodosimetry. Int J Radiat Oncol Biol Phys 2008; 71, 1236–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Paul S, Smilenov LB, Amundson SA, Widespread decreased expression of immune function genes in human peripheral blood following radiation exposure. Radiat Res 2013; 180, 575–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Paul S, Amundson SA, Gene expression signatures of radiation exposure in peripheral white blood cells of smokers and non-smokers. Int J Radiat Biol 2011; 87, 791–801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Paul S, Ghandhi SA, Weber W, Doyle-Eisele M, Melo D, Guilmette R, et al. , Gene expression response of mice after a single dose of 137CS as an internal emitter. Radiat Res 2014; 182, 380–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ghandhi SA, Smilenov L, Shuryak I, Pujol-Canadell M, Amundson SA, Discordant gene responses to radiation in humans and mice and the role of hematopoietically humanized mice in the search for radiation biomarkers. Sci Rep 2019; 9, 19434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rudqvist N, Laiakis EC, Ghandhi SA, Kumar S, Knotts JD, Chowdhury M, et al. , Global Gene Expression Response in Mouse Models of DNA Repair Deficiency after Gamma Irradiation. Radiat Res 2018; 189, 337–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Broustas CG, Xu Y, Harken AD, Garty G, Amundson SA, Comparison of gene expression response to neutron and x-ray irradiation using mouse blood. BMC Genomics 2017; 18, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Broustas CG, Duval AJ, Amundson SA, Impact of aging on gene expression response to x-ray irradiation using mouse blood. Sci Rep 2021; 11, 10177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ghandhi SA, Smilenov LB, Elliston CD, Chowdhury M, Amundson SA, Radiation dose-rate effects on gene expression for human biodosimetry. BMC Med Genomics 2015; 8, 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ghandhi SA, Turner HC, Shuryak I, Dugan GO, Bourland JD, Olson JD, et al. , Whole thorax irradiation of non-human primates induces persistent nuclear damage and gene expression changes in peripheral blood cells. PLoS One 2018; 13, e0191402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mezentsev A, Amundson SA, Global gene expression responses to low- or high-dose radiation in a human three-dimensional tissue model. Radiat Res 2011; 175, 677–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lacombe J, Sima C, Amundson SA, Zenhausern F, Candidate gene biodosimetry markers of exposure to external ionizing radiation in human blood: A systematic review. PLoS One 2018; 13, e0198851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.May JM, Bylicky M, Chopra S, Coleman CN, Aryankalayil MJ, Long and short non-coding RNA and radiation response: a review. Transl Res 2021; 233, 162–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Aryankalayil MJ, Chopra S, Levin J, Eke I, Makinde A, Das S, et al. , Radiation-Induced Long Noncoding RNAs in a Mouse Model after Whole-Body Irradiation. Radiat Res 2018; 189, 251–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang Y, Wang Q, Chen S, Hu Y, Yu C, Liu R, et al. , Screening of Long Noncoding RNAs Induced by Radiation Using Microarray. Dose Response 2020; 18, 1559325820916304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Aryankalayil MJ, Martello S, Bylicky MA, Chopra S, May JM, Shankardass A, et al. , Analysis of lncRNA-miRNA-mRNA expression pattern in heart tissue after total body radiation in a mouse model. J Transl Med 2021; 19, 336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Mojena M, Pimentel-Santillana M, Povo-Retana A, Fernandez-Garcia V, Gonzalez-Ramos S, Rada P, et al. , Protection against gamma-radiation injury by protein tyrosine phosphatase 1B. Redox Biol 2018; 17, 213–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kidd AR 3rd, Snider JL, Martin TD, Graboski SF, Der CJ, Cox AD, Ras-related small GTPases RalA and RalB regulate cellular survival after ionizing radiation. Int J Radiat Oncol Biol Phys 2010; 78, 205–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sersa I, Kranjc S, Sersa G, Nemec-Svete A, Lozar B, Sepe A, et al. , Study of radiation induced changes of phosphorus metabolism in mice by (31)P NMR spectroscopy. Radiol Oncol 2010; 44, 174–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Paul S, Kleiman NJ, Amundson SA, Transcriptomic responses in mouse blood during the first week after in vivo gamma irradiation. Sci Rep 2019; 9, 18364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Broustas CG, Harken AD, Garty G, Amundson SA, Identification of differentially expressed genes and pathways in mice exposed to mixed field neutron/photon radiation. BMC Genomics 2018; 19, 504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Davis TA, Landauer MR, Mog SR, Barshishat-Kupper M, Zins SR, Amare MF, et al. , Timing of captopril administration determines radiation protection or radiation sensitization in a murine model of total body irradiation. Exp Hematol 2010; 38, 270–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Meadows SK, Dressman HK, Muramoto GG, Himburg H, Salter A, Wei Z, et al. , Gene expression signatures of radiation response are specific, durable and accurate in mice and humans. PLoS One 2008; 3, e1912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hsieh SC, Lo PK, Wang FF, Mouse DDA3 gene is a direct transcriptional target of p53 and p73. Oncogene 2002; 21, 3050–7. [DOI] [PubMed] [Google Scholar]
- 56.Yamaguchi M, Hirouchi T, Yokoyama K, Nishiyama A, Murakami S, Kashiwakura I, The thrombopoietin mimetic romiplostim leads to the complete rescue of mice exposed to lethal ionizing radiation. Sci Rep 2018; 8, 10659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Port M, Herodin F, Valente M, Drouet M, Lamkowski A, Majewski M, et al. , First Generation Gene Expression Signature for Early Prediction of Late Occurring Hematological Acute Radiation Syndrome in Baboons. Radiat Res 2016; 186, 39–54. [DOI] [PubMed] [Google Scholar]
- 58.Schwartz JL, Jordan R, Sun J, Ma H, Hsieb AW, Dose-dependent changes in the spectrum of mutations induced by ionizing radiation. Radiat Res 2000; 153, 312–7. [DOI] [PubMed] [Google Scholar]
- 59.Port M, Herodin F, Valente M, Drouet M, Lamkowski A, Majewski M, et al. , Gene expression signature for early prediction of late occurring pancytopenia in irradiated baboons. Ann Hematol 2017; 96, 859–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Port M, Herodin F, Valente M, Drouet M, Ostheim P, Majewski M, et al. , Persistent mRNA and miRNA expression changes in irradiated baboons. Sci Rep 2018; 8, 15353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Paul S SL, Amundson SA, Widespread Decreased Expression of Immune Function Genes in Human Peripheral Blood Following Radiation Exposure. Radiation Research 2013; 180, 575–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Abend M, Badie C, Quintens R, Kriehuber R, Manning G, Macaeva E, et al. , Examining Radiation-Induced In Vivo and In Vitro Gene Expression Changes of the Peripheral Blood in Different Laboratories for Biodosimetry Purposes: First RENEB Gene Expression Study. Radiat Res 2016; 185, 109–23. [DOI] [PubMed] [Google Scholar]
- 63.Badie C, Kabacik S, Balagurunathan Y, Bernard N, Brengues M, Faggioni G, et al. , Laboratory intercomparison of gene expression assays. Radiat Res 2013; 180, 138–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Port M, Ostheim P, Majewski M, Voss T, Haupt J, Lamkowski A, et al. , Rapid High-Throughput Diagnostic Triage after a Mass Radiation Exposure Event Using Early Gene Expression Changes. Radiat Res 2019; 192, 208–18. [DOI] [PubMed] [Google Scholar]
- 65.Guo G, Wang T, Gao Q, Tamae D, Wong P, Chen T, et al. , Expression of ErbB2 enhances radiation-induced NF-kappaB activation. Oncogene 2004; 23, 535–45. [DOI] [PubMed] [Google Scholar]
- 66.Lu TP, Hsu YY, Lai LC, Tsai MH, Chuang EY, Identification of gene expression biomarkers for predicting radiation exposure. Sci Rep 2014; 4, 6293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yun M, Choi AJ, Lee YC, Kong M, Sung JY, Kim SS, et al. , Carbonyl reductase 1 is a new target to improve the effect of radiotherapy on head and neck squamous cell carcinoma. J Exp Clin Cancer Res 2018; 37, 264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhang Q, Bing Z, Tian J, Wang X, Liu R, Li Y, et al. , Integrating radiosensitive genes improves prediction of radiosensitivity or radioresistance in patients with oesophageal cancer. Oncol Lett 2019; 17, 5377–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Moore R, Puniya BL, Powers R, Guda C, Bayles KW, Berkowitz DB, et al. , Author Correction: Integrative network analyses of transcriptomics data reveal potential drug targets for acute radiation syndrome. Sci Rep 2021; 11, 7057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Moore R, Puniya BL, Powers R, Guda C, Bayles KW, Berkowitz DB, et al. , Integrative network analyses of transcriptomics data reveal potential drug targets for acute radiation syndrome. Sci Rep 2021; 11, 5585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Valenciano A, Henriquez-Hernandez LA, Moreno M, Lloret M, Lara PC, Role of IGF-1 receptor in radiation response. Transl Oncol 2012; 5, 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Amundson SA, Do KT, Fornace AJ Jr., Induction of stress genes by low doses of gamma rays. Radiat Res 1999; 152, 225–31. [PubMed] [Google Scholar]
- 73.Amundson SA, Lee RA, Koch-Paiz CA, Bittner ML, Meltzer P, Trent JM, et al. , Differential responses of stress genes to low dose-rate gamma irradiation. Mol Cancer Res 2003; 1, 445–52. [PubMed] [Google Scholar]
- 74.Amundson SA, Fornace AJ Jr., Monitoring human radiation exposure by gene expression profiling: possibilities and pitfalls. Health Phys 2003; 85, 36–42. [DOI] [PubMed] [Google Scholar]
- 75.Amundson SA, Do KT, Shahab S, Bittner M, Meltzer P, Trent J, et al. , Identification of potential mRNA biomarkers in peripheral blood lymphocytes for human exposure to ionizing radiation. Radiat Res 2000; 154, 342–6. [DOI] [PubMed] [Google Scholar]
- 76.Ostheim P, Don Mallawaratchy A, Muller T, Schule S, Hermann C, Popp T, et al. , Acute radiation syndrome-related gene expression in irradiated peripheral blood cell populations. Int J Radiat Biol 2021; 97, 474–84. [DOI] [PubMed] [Google Scholar]
- 77.Manning G, Kabacik S, Finnon P, Bouffler S, Badie C, High and low dose responses of transcriptional biomarkers in ex vivo X-irradiated human blood. Int J Radiat Biol 2013; 89, 512–22. [DOI] [PubMed] [Google Scholar]
- 78.Akerman GS, Rosenzweig BA, Domon OE, Tsai CA, Bishop ME, McGarrity LJ, et al. , Alterations in gene expression profiles and the DNA-damage response in ionizing radiation-exposed TK6 cells. Environ Mol Mutagen 2005; 45, 188–205. [DOI] [PubMed] [Google Scholar]
- 79.Buscaglia LE, Li Y, Apoptosis and the target genes of microRNA-21. Chin J Cancer 2011; 30, 371–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Beer L, Seemann R, Ristl R, Ellinger A, Kasiri MM, Mitterbauer A, et al. , High dose ionizing radiation regulates micro RNA and gene expression changes in human peripheral blood mononuclear cells. BMC Genomics 2014; 15, 814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.D’Andrea FP, Safwat A, Kassem M, Gautier L, Overgaard J, Horsman MR, Cancer stem cell overexpression of nicotinamide N-methyltransferase enhances cellular radiation resistance. Radiother Oncol 2011; 99, 373–8. [DOI] [PubMed] [Google Scholar]
- 82.Peterson JA, Maroney SA, Mast AE, Targeting TFPI for hemophilia treatment. Thromb Res 2016; 141 Suppl 2, S28–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kennedy AR, Maity A, Sanzari JK, A Review of Radiation-Induced Coagulopathy and New Findings to Support Potential Prevention Strategies and Treatments. Radiat Res 2016; 186, 121–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Waters KM, Stenoien DL, Sowa MB, von Neubeck C, Chrisler WB, Tan R, et al. , Annexin A2 modulates radiation-sensitive transcriptional programming and cell fate. Radiat Res 2013; 179, 53–61. [DOI] [PubMed] [Google Scholar]
- 85.Wang CY, Lin CF, Annexin A2: its molecular regulation and cellular expression in cancer development. Dis Markers 2014; 2014, 308976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pogosova-Agadjanyan EL, Fan W, Georges GE, Schwartz JL, Kepler CM, Lee H, et al. , Identification of radiation-induced expression changes in nonimmortalized human T cells. Radiat Res 2011; 175, 172–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Liu N, Wang YA, Sun Y, Ecsedy J, Sun J, Li X, et al. , Inhibition of Aurora A enhances radiosensitivity in selected lung cancer cell lines. Respir Res 2019; 20, 230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jiang J, Guo Z, Xu J, Sun T, Zheng X, Identification of Aurora Kinase A as a Biomarker for Prognosis in Obesity Patients with Early Breast Cancer. Onco Targets Ther 2020; 13, 4971–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Han G, Lu K, Xu W, Zhang S, Huang J, Dai C, et al. , Annexin A1-mediated inhibition of inflammatory cytokines may facilitate the resolution of inflammation in acute radiation-induced lung injury. Oncol Lett 2019; 18, 321–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Chinn IK, Sanders RP, Stray-Pedersen A, Coban-Akdemir ZH, Kim VH, Dadi H, et al. , Novel Combined Immune Deficiency and Radiation Sensitivity Blended Phenotype in an Adult with Biallelic Variations in ZAP70 and RNF168. Front Immunol 2017; 8, 576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Floyd SR, Pacold ME, Huang Q, Clarke SM, Lam FC, Cannell IG, et al. , The bromodomain protein Brd4 insulates chromatin from DNA damage signalling. Nature 2013; 498, 246–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Skvara H, Thallinger C, Wacheck V, Monia BP, Pehamberger H, Jansen B, et al. , Mcl-1 blocks radiation-induced apoptosis and inhibits clonogenic cell death. Anticancer Res 2005; 25, 2697–703. [PubMed] [Google Scholar]
- 93.Brickey WJ, Neuringer IP, Walton W, Hua X, Wang EY, Jha S, et al. , MyD88 provides a protective role in long-term radiation-induced lung injury. Int J Radiat Biol 2012; 88, 335–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Wang JT, Xie WQ, Liu FQ, Bi Y, Zhu XJ, Wang QE, et al. , NADH protect against radiation enteritis by enhancing autophagy and inhibiting inflammation through PI3K/AKT pathway. Am J Transl Res 2018; 10, 1713–21. [PMC free article] [PubMed] [Google Scholar]
- 95.Thierfelder WE, van Deursen JM, Yamamoto K, Tripp RA, Sarawar SR, Carson RT, et al. , Requirement for Stat4 in interleukin-12-mediated responses of natural killer and T cells. Nature 1996; 382, 171–4. [DOI] [PubMed] [Google Scholar]
- 96.Li Y, Singh J, Varghese R, Zhang Y, Fatanmi OO, Cheema AK, et al. , Transcriptome of rhesus macaque (Macaca mulatta) exposed to total-body irradiation. Sci Rep 2021; 11, 6295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Acharya SS, Fendler W, Watson J, Hamilton A, Pan Y, Gaudiano E, et al. , Serum microRNAs are early indicators of survival after radiation-induced hematopoietic injury. Sci Transl Med 2015; 7, 287ra69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shi Y, Zhang X, Tang X, Wang P, Wang H, Wang Y, MiR-21 is continually elevated long-term in the brain after exposure to ionizing radiation. Radiat Res 2012; 177, 124–8. [DOI] [PubMed] [Google Scholar]
- 99.Song M, Xie D, Gao S, Bai CJ, Zhu MX, Guan H, et al. , A Biomarker Panel of Radiation-Upregulated miRNA as Signature for Ionizing Radiation Exposure. Life (Basel) 2020; 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Jacob NK, Cooley JV, Yee TN, Jacob J, Alder H, Wickramasinghe P, et al. , Identification of sensitive serum microRNA biomarkers for radiation biodosimetry. PLoS One 2013; 8, e57603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Yadav M, Bhayana S, Liu J, Lu L, Huang J, Ma Y, et al. , Two-miRNA-based finger-stick assay for estimation of absorbed ionizing radiation dose. Sci Transl Med 2020; 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Rogers CJ, Lukaszewicz AI, Yamada-Hanff J, Micewicz ED, Ratikan JA, Starbird MA, et al. , Identification of miRNA signatures associated with radiation-induced late lung injury in mice. PLoS One 2020; 15, e0232411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gao F, Liu P, Narayanan J, Yang M, Fish BL, Liu Y, et al. , Changes in miRNA in the lung and whole blood after whole thorax irradiation in rats. Sci Rep 2017; 7, 44132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Rogers CJ, Kyubwa EM, Lukaszewicz AI, Yamada-Hanff J, Starbird MA, Miller TA, et al. , Identification of miRNA Associated with Reduced Survival after Whole-Thorax Lung Irradiation in Non-Human Primates. Radiat Res 2021; 196, 510–22. [DOI] [PubMed] [Google Scholar]
- 105.Fendler W, Malachowska B, Meghani K, Konstantinopoulos PA, Guha C, Singh VK, et al. , Evolutionarily conserved serum microRNAs predict radiation-induced fatality in nonhuman primates. Sci Transl Med 2017; 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Rogers CJ, Kyubwa EM, Lukaszewicz AI, Starbird MA, Nguyen M, Copeland BT, et al. , Observation of Unique Circulating miRNA Signatures in Non-Human Primates Exposed to Total-Body vs. Whole Thorax Lung Irradiation. Radiat Res 2021; 196, 547–59. [DOI] [PubMed] [Google Scholar]
- 107.Singh VK, Pollard HB, Ionizing radiation-induced altered microRNA expression as biomarkers for assessing acute radiation injury. Expert review of molecular diagnostics 2017; 17, 871–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Port M, Herodin F, Valente M, Drouet M, Ullmann R, Majewski M, et al. , Pre-Exposure Gene Expression in Baboons with and without Pancytopenia after Radiation Exposure. Int J Mol Sci 2017; 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Port M, Herodin F, Valente M, Drouet M, Ullmann R, Doucha-Senf S, et al. , MicroRNA Expression for Early Prediction of Late Occurring Hematologic Acute Radiation Syndrome in Baboons. PLoS One 2016; 11, e0165307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Malachowska B, Tomasik B, Stawiski K, Kulkarni S, Guha C, Chowdhury D, et al. , Circulating microRNAs as Biomarkers of Radiation Exposure: A Systematic Review and Meta-Analysis. Int J Radiat Oncol Biol Phys 2020; 106, 390–402. [DOI] [PubMed] [Google Scholar]
- 111.Tomasik B, Fendler W, Chowdhury D, Serum microRNAs - potent biomarkers for radiation biodosimetry. Oncotarget 2018; 9, 14038–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tahamtan A, Teymoori-Rad M, Nakstad B, Salimi V, Anti-Inflammatory MicroRNAs and Their Potential for Inflammatory Diseases Treatment. Front Immunol 2018; 9, 1377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Thum T, Catalucci D, Bauersachs J, MicroRNAs: novel regulators in cardiac development and disease. Cardiovasc Res 2008; 79, 562–70. [DOI] [PubMed] [Google Scholar]
- 114.Suzuki HI, MicroRNA Control of TGF-beta Signaling. Int J Mol Sci 2018; 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liu Z, Liang X, Li X, Liu X, Zhu M, Gu Y, et al. , MiRNA-21 functions in ionizing radiation-induced epithelium-to-mesenchymal transition (EMT) by downregulating PTEN. Toxicol Res (Camb) 2019; 8, 328–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Xu S, Ding N, Pei H, Hu W, Wei W, Zhang X, et al. , MiR-21 is involved in radiation-induced bystander effects. RNA Biol 2014; 11, 1161–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Yin X, Tian W, Wang L, Wang J, Zhang S, Cao J, et al. , Radiation quality-dependence of bystander effect in unirradiated fibroblasts is associated with TGF-beta1-Smad2 pathway and miR-21 in irradiated keratinocytes. Sci Rep 2015; 5, 11373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Nowicka Zuzanna, Tomasik Bartłomiej, Kozono David, Stawiski Konrad, Johnson Thomas, Haas-Kogan Daphne, et al. , Serum miRNA-based signature indicates radiation exposure and dose in humans: a multicenter diagnostic biomarker study. medRxiv 2021. [DOI] [PubMed] [Google Scholar]
- 119.Mo LJ, Song M, Huang QH, Guan H, Liu XD, Xie DF, et al. , Exosome-packaged miR-1246 contributes to bystander DNA damage by targeting LIG4. Br J Cancer 2018; 119, 492–502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Wu XY, Fan WD, Fang R, Wu GF, Regulation of microRNA-155 in endothelial inflammation by targeting nuclear factor (NF)-kappaB P65. J Cell Biochem 2014; 115, 1928–36. [DOI] [PubMed] [Google Scholar]
- 121.Rebane A, Akdis CA, MicroRNAs: Essential players in the regulation of inflammation. J Allergy Clin Immunol 2013; 132, 15–26. [DOI] [PubMed] [Google Scholar]
- 122.Chiba M, Monzen S, Iwaya C, Kashiwagi Y, Yamada S, Hosokawa Y, et al. , Serum miR-375-3p increase in mice exposed to a high dose of ionizing radiation. Sci Rep 2018; 8, 1302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Song M, Wang Y, Shang ZF, Liu XD, Xie DF, Wang Q, et al. , Bystander autophagy mediated by radiation-induced exosomal miR-7-5p in non-targeted human bronchial epithelial cells. Sci Rep 2016; 6, 30165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Cai S, Shi GS, Cheng HY, Zeng YN, Li G, Zhang M, et al. , Exosomal miR-7 Mediates Bystander Autophagy in Lung after Focal Brain Irradiation in Mice. Int J Biol Sci 2017; 13, 1287–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Yu J, Intestinal stem cell injury and protection during cancer therapy. Transl Cancer Res 2013; 2, 384–96. [PMC free article] [PubMed] [Google Scholar]
- 126.Shadad AK, Sullivan FJ, Martin JD, Egan LJ, Gastrointestinal radiation injury: prevention and treatment. World J Gastroenterol 2013; 19, 199–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Kau AL, Ahern PP, Griffin NW, Goodman AL, Gordon JI, Human nutrition, the gut microbiome and the immune system. Nature 2011; 474, 327–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Crawford PA, Gordon JI, Microbial regulation of intestinal radiosensitivity. Proc Natl Acad Sci U S A 2005; 102, 13254–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Ciorba MA, Riehl TE, Rao MS, Moon C, Ee X, Nava GM, et al. , Lactobacillus probiotic protects intestinal epithelium from radiation injury in a TLR-2/cyclo-oxygenase-2-dependent manner. Gut 2012; 61, 829–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wang A, Ling Z, Yang Z, Kiela PR, Wang T, Wang C, et al. , Gut microbial dysbiosis may predict diarrhea and fatigue in patients undergoing pelvic cancer radiotherapy: a pilot study. PLoS One 2015; 10, e0126312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Manichanh C, Varela E, Martinez C, Antolin M, Llopis M, Dore J, et al. , The gut microbiota predispose to the pathophysiology of acute postradiotherapy diarrhea. Am J Gastroenterol 2008; 103, 1754–61. [DOI] [PubMed] [Google Scholar]
- 132.Kim YS, Kim J, Park SJ, High-throughput 16S rRNA gene sequencing reveals alterations of mouse intestinal microbiota after radiotherapy. Anaerobe 2015; 33, 1–7. [DOI] [PubMed] [Google Scholar]
- 133.Reis Ferreira M, Andreyev HJN, Mohammed K, Truelove L, Gowan SM, Li J, et al. , Microbiota- and Radiotherapy-Induced Gastrointestinal Side-Effects (MARS) Study: A Large Pilot Study of the Microbiome in Acute and Late-Radiation Enteropathy. Clin Cancer Res 2019; 25, 6487–500. [DOI] [PubMed] [Google Scholar]
- 134.Guo H, Chou WC, Lai Y, Liang K, Tam JW, Brickey WJ, et al. , Multi-omics analyses of radiation survivors identify radioprotective microbes and metabolites. Science 2020; 370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Li Y, Yan H, Zhang Y, Li Q, Yu L, Li Q, et al. , Alterations of the Gut Microbiome Composition and Lipid Metabolic Profile in Radiation Enteritis. Front Cell Infect Microbiol 2020; 10, 541178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Cheema AK, Li Y, Singh J, Johnson R, Girgis M, Wise SY, et al. , Microbiome study in irradiated mice treated with BIO 300, a promising radiation countermeasure. Anim Microbiome 2021; 3, 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Carbonero F, Mayta-Apaza AC, Yu JZ, Lindeblad M, Lyubimov A, Neri F, et al. , A comparative analysis of gut microbiota disturbances in the Gottingen minipig and rhesus macaque models of acute radiation syndrome following bioequivalent radiation exposures. Radiat Environ Biophys 2018; 57, 419–26. [DOI] [PubMed] [Google Scholar]
- 138.Carbonero F, Mayta A, Bolea M, Yu JZ, Lindeblad M, Lyubimov A, et al. , Specific Members of the Gut Microbiota are Reliable Biomarkers of Irradiation Intensity and Lethality in Large Animal Models of Human Health. Radiat Res 2019; 191, 107–21. [DOI] [PubMed] [Google Scholar]
- 139.Kalkeri R, Walters K, Van Der Pol W, McFarland BC, Fisher N, Koide F, et al. , Changes in the gut microbiome community of nonhuman primates following radiation injury. BMC Microbiol 2021; 21, 93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Laiakis EC, Strassburg K, Bogumil R, Lai S, Vreeken RJ, Hankemeier T, et al. , Metabolic phenotyping reveals a lipid mediator response to ionizing radiation. J Proteome Res 2014; 13, 4143–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Pannkuk EL, Laiakis EC, Mak TD, Astarita G, Authier S, Wong K, et al. , A Lipidomic and Metabolomic Serum Signature from Nonhuman Primates Exposed to Ionizing Radiation. Metabolomics 2016; 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Pannkuk EL, Laiakis EC, Singh VK, Fornace AJ, Lipidomic Signatures of Nonhuman Primates with Radiation-Induced Hematopoietic Syndrome. Sci Rep 2017; 7, 9777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Bataller M, Sanchez-Garcia A, Garcia-Mayea Y, Mir C, Rodriguez I, ME LL, The Role of Sphingolipids Metabolism in Cancer Drug Resistance. Front Oncol 2021; 11, 807636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Menon N, Rogers CJ, Lukaszewicz AI, Axtelle J, Yadav M, Song F, et al. , Detection of acute radiation sickness: A feasibility study in non-human primates circulating miRNAs for triage in radiological events. PLoS ONE 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Gao Y, Li X, Gao J, Zhang Z, Feng Y, Nie J, et al. , Metabolomic Analysis of Radiation-Induced Lung Injury in Rats: The Potential Radioprotective Role of Taurine. Dose-Response 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Manna SK, Krausz KW, Bonzo JA, Idle JR, Gonzalez FJ, Metabolomics reveals aging-associated attenuation of noninvasive radiation biomarkers in mice: Potential role of polyamine catabolism and incoherent DNA damage-repair. Journal of Proteome Research 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ghosh SP, Singh R, Chakraborty K, Kulkarni S, Uppal A, Luo Y, et al. , Metabolomic changes in gastrointestinal tissues after whole body radiation in a murine model. Molecular BioSystems 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Tyburski JB, Patterson AD, Krausz KW, Slavík J, Fornace AJ, Gonzalez FJ, et al. , Radiation metabolomics. 1. Identification of minimally invasive urine biomarkers for gamma-radiation exposure in mice. Radiation Research 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Golla S, Golla JP, Krausz KW, Manna SK, Simillion C, Beyoǧlu D, et al. , Metabolomic Analysis of Mice Exposed to Gamma Radiation Reveals a Systemic Understanding of Total-Body Exposure. Radiation Research 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Li HH, Wang YW, Chen R, Zhou B, Ashwell JD, Fornace AJ, Ionizing radiation impairs T cell activation by affecting metabolic reprogramming. International Journal of Biological Sciences 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Pannkuk EL, Laiakis EC, Authier S, Wong K, Fornace AJ, Global metabolomic identification of long-term dose-dependent urinary biomarkers in nonhuman primates exposed to ionizing radiation. Radiation Research 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Pannkuk EL, Laiakis EC, Authier S, Wong K, Fornace AJ Jr., Targeted Metabolomics of Nonhuman Primate Serum after Exposure to Ionizing Radiation: Potential Tools for High-throughput Biodosimetry. RSC Adv 2016; 6, 51192–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Menon SS, Uppal M, Randhawa S, Cheema MS, Aghdam N, Usala RL, et al. , Radiation Metabolomics: Current Status and Future Directions. Frontiers in Oncology 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Johnson CH, Patterson AD, Krausz KW, Lanz C, Kang DW, Luecke H, et al. , Radiation metabolomics. 4. UPLC-ESI-QTOFMS-based metabolomics for urinary biomarker discovery in gamma-irradiated rats. Radiation Research 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Johnson CH, Patterson AD, Krausz KW, Kalinich JF, Tyburski JB, Kang DW, et al. , Radiation metabolomics. 5. Identification of urinary biomarkers of ionizing radiation exposure in nonhuman primates by mass spectrometry-based metabolomics. Radiation Research 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Cheema AK, Hinzman CP, Mehta KY, Hanlon BK, Garcia M, Fatanmi OO, et al. , Plasma derived exosomal biomarkers of exposure to ionizing radiation in nonhuman primates. Int J Mol Sci 2018; 19, 3427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Pannkuk EL, Laiakis EC, Garcia M, Fornace AJ Jr., Singh VK, Nonhuman primates with acute radiation syndrome: Results from a global serum metabolomics study after 7.2 Gy total-body irradiation. Radiat Res 2018; 190, 576–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Pannkuk EL, Laiakis EC, Fornace AJ Jr., Fatanmi OO, Singh VK, A metabolomic serum signature from nonhuman primates treated with a radiation countermeasure, gamma-tocotrienol, and exposed to ionizing radiation. Health Phys 2018; 115, 3–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Cheema AK, Mehta KY, Rajagopal MU, Wise SY, Fatanmi OO, Singh VK, Metabolomic studies of tissue injury in nonhuman primates exposed to gamma-radiation. Int J Mol Sci 2019; 20, 3360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Roh C, Metabolomics in radiation-induced biological dosimetry: A mini-review and a polyamine study. 2018. [DOI] [PMC free article] [PubMed]
- 161.Vicente E, Vujaskovic Z, Jackson IL, A systematic review of metabolomic and lipidomic candidates for biomarkers in radiation injury. 2020. [DOI] [PMC free article] [PubMed]
- 162.Patterson AD, Li H, Eichler GS, Krausz KW, Weinstein JN, Fornace AJ, et al. , UPLC-ESI-TOFMS-based metabolomics and gene expression dynamics inspector self-organizing metabolomic maps as tools for understanding the cellular response to ionizing radiation. Analytical Chemistry 2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Hu ZP, Kim YM, Sowa MB, Robinson RJ, Gao X, Metz TO, et al. , Metabolomic response of human skin tissue to low dose ionizing radiation. Molecular BioSystems 2012. [DOI] [PubMed] [Google Scholar]
- 164.Huang R, Liu X, Li H, Zhou Y, Zhou PK, Integrated analysis of transcriptomic and metabolomic profiling reveal the p53 associated pathways underlying the response to ionizing radiation in HBE cells. Cell and Bioscience 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Reisz JA, Bansal N, Qian J, Zhao W, Furdui CM, Effects of ionizing radiation on biological molecules - mechanisms of damage and emerging methods of detection. 2014. [DOI] [PMC free article] [PubMed]
- 166.Bajaj S, Singh A, Kalita B, Yashavarddhan MH, Ranjan R, Farooqi H, et al. , Gamma Radiation Induced Intestinal Proteomic Modulation in Mice: A Two Dimensional Electrophoretic Analysis. Defence Life Science Journal 2017. [Google Scholar]
- 167.Bakshi MV, Azimzadeh O, Barjaktarovic Z, Kempf SJ, Merl-Pham J, Hauck SM, et al. , Total body exposure to low-dose ionizing radiation induces long-term alterations to the liver proteome of neonatally exposed mice. Journal of Proteome Research 2015. [DOI] [PubMed] [Google Scholar]
- 168.Cheema AK, Byrum SD, Sharma NK, Altadill T, Kumar VP, Biswas S, et al. , Proteomic Changes in Mouse Spleen after Radiation-Induced Injury and its Modulation by Gamma-Tocotrienol. Radiat Res 2018; 190, 449–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Byrum SD, Burdine MS, Orr L, Mackintosh SG, Authier S, Pouliot M, et al. , Time- and radiation-dose dependent changes in the plasma proteome after total body irradiation of non-human primates: Implications for biomarker selection. PLoS One 2017; 12, e0174771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Byrum S D, Burdine M S, A Quantitative Proteomic Analysis of Urine from Gamma-Irradiated Non- Human Primates. Journal of Proteomics & Bioinformatics 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Girgis M, Li Y, Ma J, Sanda M, Wise SY, Fatanmi OO, et al. , Comparative proteomic analysis of serum from nonhuman primates administered BIO 300: a promising radiation countermeasure. Scientific Reports 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Pluder F, Barjaktarovic Z, Azimzadeh O, Mörtl S, Krämer A, Steininger S, et al. , Low-dose irradiation causes rapid alterations to the proteome of the human endothelial cell line EA.hy926. Radiation and Environmental Biophysics 2011. [DOI] [PubMed] [Google Scholar]
- 173.Sriharshan A, Boldt K, Sarioglu H, Barjaktarovic Z, Azimzadeh O, Hieber L, et al. , Proteomic analysis by SILAC and 2D-DIGE reveals radiation-induced endothelial response: Four key pathways. Journal of Proteomics 2012. [DOI] [PubMed] [Google Scholar]
- 174.Turtoi A, Sharan RN, Srivastava A, Schneeweiss FHA, Proteomic and genomic modulations induced by γ-irradiation of human blood lymphocytes. International Journal of Radiation Biology 2010. [DOI] [PubMed] [Google Scholar]
- 175.Moore HM, Bai B, Boisvert FM, Latonen L, Rantanen V, Simpson JC, et al. , Quantitative proteomics and dynamic imaging of the nucleolus reveal distinct responses to UV and ionizing radiation. Molecular and Cellular Proteomics 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kim MH, Jung SY, Ahn J, Hwang SG, Woo HJ, An S, et al. , Quantitative proteomic analysis of single or fractionated radiation-induced proteins in human breast cancer MDA-MB-231 cells. Cell and Bioscience 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Huang W, Yu J, Jones JW, Carter CL, Pierzchalski K, Tudor G, et al. , Proteomic Evaluation of the Acute Radiation Syndrome of the Gastrointestinal Tract in a Murine Total-body Irradiation Model. Health Phys 2019; 116, 516–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Simonian M, Shirasaki D, Lee VS, Bervini D, Grace M, Loo RRO, et al. , Proteomics identification of radiation-induced changes of membrane proteins in the rat model of arteriovenous malformation in pursuit of targets for brain AVM molecular therapy. Clin Proteomics 2018; 15, 43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Guo L, Xiao Y, Fan M, Li JJ, Wang Y, Profiling global kinome signatures of the radioresistant MCF-7/C6 breast cancer cells using MRM-based targeted proteomics. Journal of Proteome Research 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Wei R, Zhang Y, Shen L, Jiang W, Li C, Zhong M, et al. , Comparative proteomic and radiobiological analyses in human lung adenocarcinoma cells. Molecular and Cellular Biochemistry 2012. [DOI] [PubMed] [Google Scholar]
- 181.Feng XP, Yi H, Li MY, Li XH, Yi B, Zhang PF, et al. , Identification of biomarkers for predicting nasopharyngeal carcinoma response to radiotherapy by proteomics. Cancer Research 2010. [DOI] [PubMed] [Google Scholar]
- 182.Wu P, Zhang H, Qi L, Tang Q, Tang Y, Xie Z, et al. , Identification of ERp29 as a biomarker for predicting nasopharyngeal carcinoma response to radiotherapy. Oncology Reports 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kim MH, Jung SY, Ahn J, Hwang SG, Woo HJ, An S, et al. , Quantitative proteomic analysis of single or fractionated radiation-induced proteins in human breast cancer MDA-MB-231 cells. Cell Biosci 2015; 5, 2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Wei R, Zhang Y, Shen L, Jiang W, Li C, Zhong M, et al. , Comparative proteomic and radiobiological analyses in human lung adenocarcinoma cells. Mol Cell Biochem 2012; 359, 151–9. [DOI] [PubMed] [Google Scholar]
- 185.Dörr H, Meineke V, Acute radiation syndrome caused by accidental radiation exposure - therapeutic principles. 2011. [DOI] [PMC free article] [PubMed]
- 186.Geiger H, Pawar SA, Kerschen EJ, Nattamai KJ, Hernandez I, Liang HPH, et al. , Pharmacological targeting of the thrombomodulin-activated protein C pathway mitigates radiation toxicity. Nature Medicine 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Mosnier LO, Gale AJ, Yegneswaran S, Griffin JH, Activated protein C variants with normal cytoprotective but reduced anticoagulant activity. Blood 2004; 104, 1740–4. [DOI] [PubMed] [Google Scholar]
- 188.Kantara C, Moya SM, Houchen CW, Umar S, Ullrich RL, Singh P, et al. , Novel regenerative peptide TP508 mitigates radiation-induced gastrointestinal damage by activating stem cells and preserving crypt integrity. Laboratory Investigation; 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Ryaby JT, Thrombin Peptide TP508 Stimulates Cellular Events Leading to Angiogenesis, Revascularization, and Repair of Dermal and Musculoskeletal Tissues. The Journal of Bone and Joint Surgery (American) 2006. [DOI] [PubMed] [Google Scholar]
- 190.Singh VK, Romaine PLP, Seed TM, Medical countermeasures for radiation exposure and related injuries: Characterization of medicines, FDA-approval status and inclusion into the strategic national stockpile. 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Singh VK, Romaine PLP, Newman VL, Seed TM, Medical countermeasures for unwanted CBRN exposures: part II radiological and nuclear threats with review of recent countermeasure patents. 2016. [DOI] [PMC free article] [PubMed]
- 192.Strohl WR, Fusion Proteins for Half-Life Extension of Biologics as a Strategy to Make Biobetters. 2015. [DOI] [PMC free article] [PubMed]
- 193.Singh VK, Seed TM, An update on sargramostim for treatment of acute radiation syndrome. Drugs Today (Barc) 2018; 54, 679–93. [DOI] [PubMed] [Google Scholar]
- 194.FDA approves Leukine for Acute Radiation Syndrome. 2018.
- 195.Sanofi-Aventis U.S. LLC. LEUKINE® (sargramostim) for injection, forsubcutaneous or intravenous use.; 2018.
- 196.Zhong Y, Pouliot M, Downey AM, Mockbee C, Roychowdhury D, Wierzbicki W, et al. , Efficacy of delayed administration of sargramostim up to 120 hours post exposure in a nonhuman primate total body radiation model. Int J Radiat Biol 2020, 1–17. [DOI] [PubMed] [Google Scholar]
- 197.Bunin DI, Bakke J, Green CE, Javitz HS, Fielden M, Chang PY, Romiplostim (Nplate((R))) as an effective radiation countermeasure to improve survival and platelet recovery in mice. Int J Radiat Biol 2020; 96, 145–54. [DOI] [PubMed] [Google Scholar]
- 198.Li Y, Girgis M, Wise SY, Fatanmi OO, Seed TM, Maniar M, et al. , Analysis of the metabolomic profile in serum of irradiated nonhuman primates treated with Ex-Rad, a radiation countermeasure. Scientific Reports 2021; 11, 11449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Micewicz ED, Damoiseaux RD, Deng G, Gomez A, Iwamoto KS, Jung ME, et al. , Classes of Drugs that Mitigate Radiation Syndromes. Front Pharmacol 2021; 12, 666776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Singh VK, Seed TM, Repurposing Pharmaceuticals Previously Approved by Regulatory Agencies to Medically Counter Injuries Arising Either Early or Late Following Radiation Exposure. Front Pharmacol 2021; 12, 624844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Li Y, Girgis M, Wise SY, Fatanmi OO, Seed TM, Maniar M, et al. , Analysis of the metabolomic profile in serum of irradiated nonhuman primates treated with Ex-Rad, a radiation countermeasure. Sci Rep 2021; 11, 11449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Singh VK, Garcia M, Seed TM, A review of radiation countermeasures focusing on injury-specific medicinals and regulatory approval status: part II. Countermeasures for limited indications, internalized radionuclides, emesis, late effects, and agents demonstrating efficacy in large animals with or without FDA IND status. Int J Radiat Biol 2017; 93, 870–84. [DOI] [PubMed] [Google Scholar]
- 203.DiCarlo AL, Cassatt DR, Dowling WE, Esker JL, Hewitt JA, Selivanova O, et al. , Challenges and Benefits of Repurposing Products for Use during a Radiation Public Health Emergency: Lessons Learned from Biological Threats and other Disease Treatments. Radiat Res 2018; 190, 659–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Jackson IL, Zodda A, Gurung G, Pavlovic R, Kaytor MD, Kuskowski MA, et al. , BIO 300, a nanosuspension of genistein, mitigates pneumonitis/fibrosis following high-dose radiation exposure in the C57L/J murine model. Br J Pharmacol 2017; 174, 4738–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Jones JW, Jackson IL, Vujaskovic Z, Kaytor MD, Kane MA, Targeted Metabolomics Identifies Pharmacodynamic Biomarkers for BIO 300 Mitigation of Radiation-Induced Lung Injury. Pharm Res 2017; 34, 2698–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Singh VK, Seed TM, BIO 300: a promising radiation countermeasure under advanced development for acute radiation syndrome and the delayed effects of acute radiation exposure. Expert Opin Investig Drugs 2020; 29, 429–41. [DOI] [PubMed] [Google Scholar]
- 207.Cheema AK, Mehta KY, Santiago PT, Fatanmi OO, Kaytor MD, Singh VK, Pharmacokinetic and Metabolomic Studies with BIO 300, a Nanosuspension of Genistein, in a Nonhuman Primate Model. Int J Mol Sci 2019; 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Cheema AK, Li Y, Girgis M, Jayatilake M, Simas M, Wise SY, et al. , Metabolomic studies in tissues of mice treated with amifostine and exposed to gamma-radiation. Scientific Reports 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Cheema AK, Li Y, Girgis M, Jayatilake M, Fatanmi OO, Wise SY, et al. , Alterations in Tissue Metabolite Profiles with Amifostine-Prophylaxed Mice Exposed to Gamma Radiation. Metabolites 2020; 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Crook A, De Lima Leite A, Payne T, Bhinderwala F, Woods J, Singh VK, et al. , Radiation exposure induces cross-species temporal metabolic changes that are mitigated in mice by amifostine. Sci Rep 2021; 11, 14004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Mell LK, Movsas B, Pharmacologic normal tissue protection in clinical radiation oncology: Focus on amifostine. Expert Opinion on Drug Metabolism and Toxicology 2008. [DOI] [PubMed] [Google Scholar]
- 212.Upadhyay SN, Ghosef A, Radioprotection by chemical means with the help of combined regimen radio-protectors - A short review. 2017.
- 213.Singh VK, Seed TM, The efficacy and safety of amifostine for the acute radiation syndrome. Expert Opin Drug Saf 2019; 18, 1077–90. [DOI] [PubMed] [Google Scholar]
- 214.Cherdyntseva N, Shishkina A, Butorin I, Murase H, Gervas P, Kagiya TV, Effect of tocopherol-monoglucoside (TMG), a water-soluble glycosylated derivate of vitamin E, on hematopoietic recovery in irradiated mice. Journal of Radiation Research 2005. [DOI] [PubMed] [Google Scholar]
- 215.Kumar KS, Srinivasan V, Toles R, Jobe L, Seed TM, Nutritional approaches to radioprotection: Vitamin E. Mil Med 2002; 167, 57–9. [PubMed] [Google Scholar]
- 216.Cheema AK, Mehta KY, Fatanmi OO, Wise SY, Hinzman CP, Wolff J, et al. , A Metabolomic and Lipidomic Serum Signature from Nonhuman Primates Administered with a Promising Radiation Countermeasure, Gamma-Tocotrienol. Int J Mol Sci 2017; 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Rosen E, Fatanmi OO, Wise SY, Rao VA, Singh VK, Tocol prophylaxis for total-body irradiation: A proteomic analysis in murine model. Health Phys 2020. [DOI] [PubMed] [Google Scholar]
- 218.Rosen E, Fatanmi OO, Wise SY, Rao VA, Singh VK, Gamma-tocotrienol, a radiation countermeasure, reverses proteomic changes in serum following total-body gamma irradiation in mice. Sci Rep 2022; 12, 3387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Whitnall MH, Elliott TB, Harding RA, Inal CE, Landauer MR, Wilhelmsen CL, et al. , Androstenediol stimulates myelopoiesis and enhances resistance to infection in gamma-irradiated mice. International Journal of Immunopharmacology 2000. [DOI] [PubMed] [Google Scholar]
- 220.Whitnall MH, Inal CE, Jackson WE, Miner VL, Villa V, Seed TM, In vivo radioprotection by 5-androstenediol: Stimulation of the innate immune system. Radiation Research 2001. [DOI] [PubMed] [Google Scholar]
- 221.Tabeie F, Tabatabaei SM, Mahmoud-Pashazadeh A, Assadi M, Radioprotective effect of beta D-glucan and vitamin E on gamma irradiated mouse. Journal of Clinical and Diagnostic Research 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Hofer M, Pospíšil M, Dušek L, Hoferová Z, Weiterová L, Inhibition of cyclooxygenase-2 promotes the stimulatory action of adenosine A 3 receptor agonist on hematopoiesis in sublethally γ-irradiated mice. Biomedicine and Pharmacotherapy 2011. [DOI] [PubMed] [Google Scholar]
- 223.Pospisil M, Netikova J, Pipalova I, Jary J, Combined radioprotection by preirradiation peroral cystamine and postirradiation glucan administration. Folia Biologica 1991. [PubMed] [Google Scholar]
- 224.Patchen ML, Macvittie TJ, Weiss JF, Combined modality radioprotection: The use of glucan and selenium with WR-2721. International Journal of Radiation Oncology, Biology, Physics 1990. [DOI] [PubMed] [Google Scholar]
- 225.Cramer DE, Allendorf DJ, Baran JT, Hansen R, Marroquin J, Li B, et al. , β-Glucan enhances complement-mediated hematopoietic recovery after bone marrow injury. Blood 2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Salama SF, ß-Glucan ameliorates gamma-rays induced oxidative injury in male swiss albino rats. Pakistan Journal of Zoology 2011. [Google Scholar]
- 227.Pillai TG, Uma Devi P, Mushroom beta glucan: Potential candidate for post irradiation protection. Mutation Research - Genetic Toxicology and Environmental Mutagenesis 2013. [DOI] [PubMed] [Google Scholar]
- 228.Rondanelli M, Opizzi A, Monteferrario F, [The biological activity of beta-glucans]. Minerva medica; 2009. [PubMed] [Google Scholar]
- 229.Stickney DR, Dowding C, Garsd A, Ahlem C, Whitnall M, McKeon M, et al. , 5-androstenediol stimulates multilineage hematopoiesis in rhesus monkeys with radiation-induced myelosuppression. International Immunopharmacology 2006. [DOI] [PubMed] [Google Scholar]
- 230.Singh VK, Shafran RL, Inal CE, Jackson WE, Whitnall MH, Effects of whole-body gamma irradiation and 5-androstenediol administration on serum G-CSF. Immunopharmacology and Immunotoxicology 2005. [DOI] [PubMed] [Google Scholar]
- 231.Singh VK, Grace MB, Jacobsen KO, Chang CM, Parekh VI, Inal CE, et al. , Administration of 5-androstenediol to mice: Pharmacokinetics and cytokine gene expression. Experimental and Molecular Pathology 2008. [DOI] [PubMed] [Google Scholar]
- 232.Aerts-Kaya FSF, Visser TP, Arshad S, Frincke J, Stickney DR, Reading CL, et al. , 5-androstene-3β,17β-diol promotes recovery of immature hematopoietic cells following myelosuppressive radiation and synergizes with thrombopoietin. International Journal of Radiation Oncology Biology Physics 2012. [DOI] [PubMed] [Google Scholar]
- 233.Singh VK, Newman VL, Romaine PLP, Wise SY, Seed TM, Radiation countermeasure agents: An update (2011–2014). 2014. [DOI] [PMC free article] [PubMed]
- 234.Singh VK, Hanlon BK, Santiago PT, Seed TM, A review of radiation countermeasures focusing on injury-specific medicinals and regulatory approval status: part III. Countermeasures under early stages of development along with ‘standard of care’ medicinal and procedures not requiring regulatory approval for use. Int J Radiat Biol 2017; 93, 885–906. [DOI] [PubMed] [Google Scholar]
- 235.Rosen EM, Day R, Singh VK, New approaches to radiation protection. Front Oncol 2014; 4, 381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.De Ruysscher D, Jin J, Lautenschlaeger T, She JX, Liao Z, Kong FS, Blood-based biomarkers for precision medicine in lung cancer: precision radiation therapy. Transl Lung Cancer Res 2017; 6, 661–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Rutten EA, Badie C, Radiation Biomarkers: Silver Bullet, or Wild Goose Chase? J Pers Med 2021; 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


