Abstract
Polyphenols are compounds found in various plants and foods, known for their antioxidant and anti-inflammatory properties. Recently, researchers have been exploring the therapeutic potential of marine polyphenols and other minor nutrients that are found in algae, fish and crustaceans. These compounds have unique chemical structures and exhibit diverse biological properties, including anti-inflammatory, antioxidant, antimicrobial and antitumor action. Due to these properties, marine polyphenols are being investigated as possible therapeutic agents for the treatment of a wide variety of conditions, such as cardiovascular disease, diabetes, neurodegenerative diseases and cancer. This review focuses on the therapeutic potential of marine polyphenols and their applications in human health, and also, in marine phenolic classes, the extraction methods, purification techniques and future applications of marine phenolic compounds.
Keywords: marine polyphenols, therapeutics, antioxidants, anti-inflammatories, health, cardiovascular diseases, diabetes, neurodegenerative diseases, cancer
1. Introduction
The maritime environment encompasses more than 70% of the Earth’s surface and is the world’s biggest ecosystem, with very changeable and hostile physicochemical conditions (low temperature, restricted light availability, high salinity and high pressure). The world’s oceans and seas contain approximately 90% of our planet’s biological biomass, which is dominated by unicellular microbes [1].
The search for natural alternatives for the treatment and prevention of diseases has been increasingly relevant, and marine polyphenols have aroused the interest of researchers in this field. These compounds are bioactive molecules that have antioxidant, anti-inflammatory and antitumor properties, in addition to other beneficial health effects [2]. One of the main sources of marine polyphenols is algae, which contains a diverse range of substances, including flavonoids, phenols and organic acids. Other important sources include fish and crustaceans, which are also rich in marine polyphenols such as catechins and phenolic acids [3].
Marine polyphenols have shown potential for treating and preventing a variety of health conditions. For example, studies indicate that by lowering oxidative stress and inflammation, these substances may help reduce the chance of cardiovascular disease. In addition, marine polyphenols have demonstrated antidiabetic properties, contributing to glycemic control and improving insulin sensitivity [2]. There is also evidence that these compounds may be beneficial for brain health, as they have neuroprotective and anti-inflammatory properties, which may help prevent neurodegenerative diseases such as Alzheimer’s [4]. In addition, marine polyphenols have demonstrated antitumor effects, showing promise in the treatment of several types of cancer. These compounds are believed to help prevent the development of cancer cells, as well as inhibit the growth and proliferation of existing tumors [5].
Due to the therapeutic potential of marine polyphenols, there is a growing interest in the development of nutraceuticals and pharmaceuticals that contain these compounds as active ingredients. However, more studies are required to assess the safety and effectiveness of these compounds in people, as well as to identify the optimal dose for therapeutic use [6].
This review aims to provide a comprehensive understanding of marine organism phenolic compounds and other important compounds, from their origin, highlighting the potential activities as new potential therapeutics to be applied in cardiovascular diseases, diabetes, neurodegenerative diseases and cancer. Furthermore, it will exploit the circular approach: from mechanism of action, safety measures, challenges and extraction/purification methods of the marine-based phenolic compounds.
2. Methodology
Data were gathered mostly from internet sources, namely Web of Science, Google Scholar, Science Direct and Scopus, and included research papers, books, chapters, news, websites and reviews. The following subjects were chosen: seaweed, macroalgae, fish, fungi, marine plants, marine and phenolic compounds. In addition, we used a laboratory Mendeley group, which includes article regarding marine phenolic, with all the information gathered from 2019 until now. Furthermore, additional terms such as phlorotannin, bromophenol, terpenoids and flavonoids where also searched. We endeavored to collect as much data as possible with scientific backing for analysis.
However, there are references from before 2019, due to be articles cited in the bibliography analyzed and considered important to cite being the original content cited by the recent bibliography.
3. Marine Polyphenols
Marine polyphenols are a group of bioactive compounds that are found in a wide variety of marine organisms, including algae, fish and crustaceans. These compounds are characterized by the presence of multiple hydroxyl groups (-OH) in their molecular structures, which give them antioxidant and anti-inflammatory properties [7]. These compounds have a varied chemical structure and are classified into different groups, such as flavonoids, phenolic acids, tannins, lignans and stilbenes. Flavonoids are one of the most studied classes and include compounds such as catechins, quercetin and rutin, which are commonly found in algae and fish [2].
Marine organisms generate these marine-origin chemicals as a defense strategy against oxidative stress and ultraviolet radiation. Seaweed, for example, is frequently exposed to harsh environmental conditions, and the effects of damage are not visible; as a result, the alga produces a diverse range of metabolites (polyphenols, xanthophylls, tocopherols and polysaccharides) to protect against abiotic and biological factors such as herbivory and mechanical aggression from the sea. Furthermore, marine polyphenols also play an important role in cellular communication and ecological interactions between organisms [8].
Marine polyphenols have aroused the interest of researchers because they have a wide range of health benefits, including anti-inflammatory, antioxidant, antitumor and neuroprotective properties. They have also been investigated as possible therapeutic agents for various conditions such a cardiovascular diseases, diabetes and cancer [9]. Although most studies have fixed their attention on the antioxidant and anti-inflammatory properties of marine polyphenols, recent studies have highlighted the importance of investigating the other mechanisms of action of these compounds, as well as their bioavailability and metabolism in humans [10].
In summary, marine polyphenols are bioactive compounds with promising therapeutic potential, but they are still poorly understood in terms of their properties and effects on human health. Therefore, there is a growing need for additional research to evaluate their safety and efficacy and to develop new therapies based on these compounds [11].
3.1. Sources of Marine Polyphenols and Other Micronutrient
Marine polyphenols are found in a variety of natural sources, including algae, fish, crustaceans and mollusks. Below we will detail the main sources of marine polyphenols and the compounds that can be found in each of them [12].
3.1.1. Algae
These bioactive compounds are found in different types of algae, including green (Chlorophyta), brown (Ochrophyta, Phaeophyceae) and red (Rhodophyta) macroalgae [13]. Each type of seaweed has different chemical compositions, with different types and concentrations of polyphenols. They are rich in various types of polyphenols, such as fucoxanthins, phlorotannins and fucoidans [8]. Fucoxanthins are a type of carotenoid found in brown algae and have antioxidant, anti-inflammatory and anti-obesity properties [14]. Phlorotannins are unique phenolic compounds found in brown seaweed that have antioxidant, anti-inflammatory and anti-tumor properties [15]. Fucoidans are sulfated polysaccharides found in brown algae and have antitumor, anticoagulant and anti-inflammatory properties [16].
The polyphenols found in algae are phenolic compounds, which include catechins [17], phlorotannins, fucoidans and fucoxanthins [18]. Catechins are a type of flavonoid that have antioxidant and anti-inflammatory activity, being found mainly in red algae. Phlorotannins are a unique group of polyphenols found in brown seaweed, with antioxidant and anti-inflammatory activity [19]. Fucoidans are sulfated polysaccharides found in brown algae, with anticoagulant, anticancer, anti-inflammatory and immunomodulatory properties [16]. Fucoxanthins are a type of carotenoid unique to brown algae, with antioxidant, anti-inflammatory, anti-obesity and antitumor activity [20].
Seaweed polyphenols have several beneficial properties for human health. In addition to antioxidant and anti-inflammatory activities, these compounds also exhibit antiviral, antifungal and antibacterial activities. Furthermore, studies have shown that seaweed polyphenols exhibit anti-obesity, anti-hypertension, anti-diabetes and anti-cancer activities [21]. Macroalgae polyphenols are also used in cosmetic products such as skin creams and lotions. These compounds have anti-aging, moisturizing and UV-protective properties [22].
Many previous studies have been performed where phenolic compounds were isolated from seaweed and include single phenolic compounds or polyphenols such as flavonoids, phlorotannins, mycosporine-like amino acids (MAAs), bromophenols and terpenoids [23]. The biological action of phenolic compounds is determined by the position of the hydroxyl groups and the number of phenyl rings in the structure [24].
Brown algae species contain a large amount of phlorotannins, while green and red algae mainly produce flavonoids, bromophenols, terpenoids and mycosporin amino acids in response to environmental conditions [22]. In the cosmetic industry, phlorotannins enable the activation of hyaluronidase, with antiallergic, anti-wrinkle, anti-aging, skin whitening, photoprotection and improved skin health benefits. Thus, seaweed-derived phenolic compounds and their chemical structures, along with their skin benefits, are extremely useful in the skincare industry [25].
Seaweed-derived phenolic compounds have a wide range of applications, including enzyme inhibition (e.g., tyrosinase inhibition, elastase inhibition, collagenase inhibition, matrix metalloproteinase inhibition in photoprotection, angiotensin-converting enzyme inhibition, 1 (ACE-1), pro-inflammatory cyclooxygenase and lipoxygenase (COX-1, 2 and 5-LOX), as well as inhibition of dipeptidyl peptidase-4 (DPP-4) and inhibition of hydroxymethyl glutaryl coenzyme A reductase (hMGCR)) and antibacterial, antifungal, antioxidant and anti-inflammatory qualities that are appealing when used in makeup and cosmeceutical product formulations [19,22].
It is important to emphasize that the concentrations of polyphenols in seaweed vary according to the species, habitat, environmental conditions, stage of development and extraction method. Therefore, it is important to carry out studies to identify the best sources of polyphenols and the best extraction conditions to ensure obtaining products with a high concentration of bioactive compounds [13]. Among the seaweed species with the greatest potential (see Table 1), the red macroalgae stand out (Rhodophyta): Neorhodomela larix, Rhodomela confervoides, Callophycus serratus, Tichocarpus crinitus, Chondrus crispus, Kappaphycus spp., Porphyra/Pyropia spp. and Symphyocladia latiuscula; the brown macroalgae (Ochrophyta, Phaeophyceae): Ecklonia cava, E. cava subsp. stolonifera, E. cava subsp. kurome, Eisenia bicyclis, Ishige okamurae, Fucus vesiculosus, F. spiralis, Gongolaria nodicaulis, G. usneoides, Laminaria digitata, Sargassum muticum, S. vulgare, S. thunbergii, Lessonia spicata, Durvillaea antarctica, Vidalia colensoi, Padina gymnospora, Macrocystis pyrifera; and the green macroalgae (Chlorophyta): Caulerpa racemosa, Cladophora socialis, Monostroma grevillei, Ulva clathrata, U. compressa, U. intestinalis, U. linza, U. flexuosa, U. australis, Capsosiphon fulvescens, Chaetomorpha moniligera.
Table 1.
Phenolic compounds and other micronutrients from some marine macroalgae and their bioactivities.
| Species | Phenolic Compounds and Other Micronutrients | Bioactivities | References |
|---|---|---|---|
| Callophycus serratus (R) | Phenolic terpenoids: diterpenes and sesquiterpenes | Antibacterial, antifungal and anticancer | [23,26] |
| Capsosiphon fulvescens (C) | Bromophenols and flavonoids | Antioxidant | [27] |
| Caulerpa racemosa (C) (Figure 1a) | Catechin, epicatechin, epigallocatechin, catechin gallate, epicatechin gallate | Antidiabetic, Antiproliferative, anti-inflammatory and antioxidant | [19] |
| Chaetomorpha moniligera (C) | Bromophenols and flavonoids | Antioxidant | [22,27] |
| Chondrus crispus (R) (Figure 1b) | Isoflavones | Antioxidant, antiproliferative and antidiabetic | [28] |
| Cladophora socialis (C) | Cladophorol | Antibiotic | [29] |
| Durvillaea antarctica (P) | Phlorotannins, tocopherol | Antioxidant | [15,30,31] |
| Ecklonia cava (P) | Polyphenol extract, phlorotannins, cholinesterase, dieckol | Antioxidant, anti-obesity, neuroprotection | [32,33] |
| E. cava subsp. stolonifera (P) | Phlorotannins, phlorofucofuroeckol | Anti-inflammatory, antioxidant, anti-hyperlipidemic chemo-preventive | [34,35] |
| E. cava subsp. kurome (P) | Phlorotannins | Antibacterial, anti-proliferative, anti-inflammatory and anti-adipogenic | [19,36] |
| Eisenia bicyclis (P) | Phlorotannins, fucofuroeckol-A | Antioxidant, anti-inflammatory and neuroprotective | [23,37] |
| Fucus spiralis (P) (Figure 1c) | Phlorotannins | Antioxidant, photoprotective; anti-enzymatic, anti-inflammatory and cytoprotective | [38,39,40] |
| F. vesiculosus (P) (Figure 1d) | Phlorotannins | Antioxidant, antibacterial and antidiabetic | [41,42] |
| Gongolaria nodicaulis (P) (Figure 1e) | Phlorotannins | Antimicrobial | [23,43] |
| G. usneoides (P) (Figure 1f) | Phlorotannins | Anti-inflammatory, antioxidant, and antimicrobial | [19,44] |
| Ishige okamurae (P) | Phlorotannins | Antioxidant, anti-inflammatory, photoprotective | [45,46] |
| Kappaphycus alvarezii (R) (Figure 1g) | Chlorogenic and salicylic acid | Antioxidant, antimicrobial | [47,48,49] |
| Laminaria digitata (P) (Figure 1h) | Phlorotannins | Antioxidant | [50,51] |
| Lessonia spicata (P) | Phlorotannins | Antioxidant, photoprotective | [52,53] |
| Macrocystis pyrifera (P) | Phlorotannins: phloroeckol and phloroglucinol | Antioxidant and antidiabetic | [54,55] |
| Monostroma grevillei (C) | Polyphenol extract | Antiviral | [23] |
| Neorhodomela larix (R) | Polyphenol extract | Antioxidant | [19,56] |
| Padiana boryana (P) | Ellagic acid and velutin | Antimicrobial and antiprotozoal | [57] |
| Padina boergesenii (P) | Phenolic compounds | Antioxidant and photo-protective | [58] |
| Padina gymnospora (P) (Figure 1i) | Phenolic compounds, flavonoids | Antioxidant, antibacterial | [59,60] |
| Polysiphonia morrowii (R) | 5-bromo-3,4-dihydroxybenzaldehyde | Anti-adipogenesis | [61] |
| Polycladia myrica (P) | Phlorotannins | Antioxidant, Antibacterial and photo-protective | [62] |
| Rhodomela confervoides (R) | Bromophenols | Antioxidant, antibacterial, cytotoxic | [63,64,65] |
| Sargassum muticum (P) (Figure 1j) | Phlorotannins, dieckol | Antioxidant, antibacterial, tyrosinase and elastase inhibition | [66,67] |
| S. vulgare (P) (Figure 1k) | Phlorotannins | Antioxidant, antidiabetic, antifungal, pancreatic lipase and anti-inflammatory | [13,68,69] |
| S. thunbergii (P) | Phenolic compounds, phlorotannins | Antioxidant, anti- inflammatory, antibacterial and photoprotective | [70,71,72] |
| Symphyocladia latiuscula (R) | Phenolic compounds, bromophenols | Antioxidant, neuroprotective | [73,74,75] |
| Tichocarpus crinitus (R) | Bromophenols, phenylpropanoids, tichocarpol | Antioxidant, feeding-deterrent activity | [76,77,78] |
| Ulva australis (C) | Phenolic compounds, bromophenols, flavonoids, tannins | Antioxidant, antidiabetic | [19,79] |
| U. clathrata (C) (Figure 1l) | Phenolic compounds, flavonoids | Antioxidant | [80,81,82] |
| U. compressa (C) (Figure 1m) | Phenolic compounds | Antioxidant | [83,84] |
| U. flexuosa (C) | Phlobatanins | Antifungal, antibacterial | [85,86] |
| U. intestinalis (C) (Figure 1n) | Phenolic compounds, flavonoids | Antioxidant, antibacterial | [87,88,89] |
| U. lactuca (C) (Figure 1o) | Ellagic acid and velutin | Antimicrobial | [57] |
| U. linza (C) (Figure 1p) | Phenolic compounds, flavonoids | Antioxidant, anti-inflammatory | [19,81,90] |
| U. rigida (C) | Phenolic compounds | Antifungal, antibacterial, antioxidant and AChE inhibitory capacity | [91,92] |
| Vidalia colensoi (P) | Bromophenols | Antibacterial | [19,23,93] |
C—Chlorophyta; R—Rhodophyta; P—Phaeophyceae.
Figure 1.
Seaweed species images: (a) Caulerpa racemosa (C); (b) Chondrus crispus (R); (c) Fucus spiralis (P); (d) Fucus vesiculosus (P); (e) Gongolaria nodicaulis (P); (f) Gongolaria usneoides (P); (g) Kappaphycus alvarezii (R); (h) Laminaria digitata (P); (i) Padina gymnospora (P); (j) Sargassum muticum (P); (k) Sargassum vulgare (P); (l) Ulva clathrata (C); (m) Ulva compressa (C); (n) Ulva intestinalis (C); (o) Ulva lactuca (C); (p) Ulva linza (C); (C) Chlorophyta; (R) Rhodophyta; (P) Phaeophyceae. Scale Bar = 1 cm.
3.1.2. Fish
Fish are also an important source of marine polyphenols and other minor nutrients, particularly fatty fish such as salmon (Salmo salar), tuna (Thunnus orientalis) and sardines (Sardina pilchardus) [94]. Polyphenols found in fish include compounds such as catechins, phenolic acids and carotenoids [95]. Catechins are a type of flavonoid that have antioxidant and anti-inflammatory properties. Phenolic acids are common compounds that are also found in fruits, vegetables and plants that also have antioxidant and anti-inflammatory properties. Carotenoids, such as astaxanthin, are natural pigments found in some types of fish that have antioxidant and anti-inflammatory properties [96].
Curcumin is a natural polyphenol that is found in some fish, such as Tambaqui (Colossoma macropomum) [97]. It is responsible for the yellow color of turmeric root, a plant widely used in cooking and traditional medicine [98]. Curcumin has been the subject of many scientific studies due to its antioxidant and anti-inflammatory properties. Curcumin is thought to help prevent or treat a variety of inflammatory conditions, such as arthritis, inflammatory bowel disease and even cardiovascular disease [99]. Additionally, studies suggest that curcumin may help lower blood cholesterol levels. High cholesterol is a major risk factor for heart disease, and curcumin may be helpful in preventing these conditions [100]. Curcumin is considered safe and well tolerated in moderate doses. However, it is important to note that the absorption of curcumin by the body is limited, which can limit its effectiveness in some situations [101].
Catechins are a group of polyphenols with antioxidant and anti-inflammatory properties that are found in many foods, including fish such as tuna and salmon [102]. Catechins are known for their ability to neutralize free radicals, which are unstable molecules naturally produced by the body in response to stress, pollution and other factors. The accumulation of free radicals can lead to cell damage and increase the risk of chronic diseases such as cancer, heart disease and neurodegenerative diseases [103]. Additionally, catechins have anti-inflammatory properties that can help reduce inflammation in the body, which is a natural immune system response to injury and infection, but when persistent can lead to a number of illnesses [104]. Catechins also have anticancer activities, as they can help prevent the growth of cancer cells and inhibit the formation of new blood vessels that feed tumors [105] A study published in the scientific publication “Nutrients” found that eating catechin-rich fish, such as salmon, was associated with a reduced risk of cardiovascular disease [106]. Another study published in “Antioxidants” showed that catechins found in fish can help prevent cellular aging and protect DNA [106]. Although catechins can be found in some fish, most research into their health benefits has been with green tea, which is a rich source of catechins. However, including catechin-rich fish in your diet can be a delicious way to increase your intake of these healthy compounds [107].
Quercetin is a flavonol, a type of flavonoid that is found in many plant foods, including fruits, vegetables and some herbs [108]. Furthermore, quercetin can also be found in some fish such, as salmon and trout. This compound is known for its antioxidant and anti-inflammatory properties and is one of the most studied flavonoids in relation to human health. Quercetin acts as an antioxidant, helping to neutralize free radicals, which are unstable molecules naturally produced by the body that can damage cells and lead to chronic disease [108,109]. Additionally, quercetin has anti-inflammatory properties that can help reduce inflammation in the body, which is a natural immune system response to injury and infection, but which can lead to a host of illnesses when it becomes chronic [110]. Moreover, quercetin may help protect cardiovascular health. It helps to lower LDL cholesterol (“bad cholesterol”) and increase HDL cholesterol (“good cholesterol”), which can help prevent cardiovascular diseases such as heart attacks and strokes. Quercetin also helps lower blood pressure and protects heart cells and blood vessels from damage [111]. Quercetin has also been studied for its potential to prevent and treat cancer. In vivo and in vitro studies have shown that quercetin can help prevent the growth of cancer cells and inhibit the formation of new blood vessels that feed tumors. Additionally, quercetin may help increase the effectiveness of other cancer treatments, such as chemotherapy [112].
Ellagic acid is a naturally occurring phenolic acid that is found in various foods, including fruits, vegetables and some types of fish. Phenolic acids are a type of organic compound that are known for their antioxidant properties and have been associated with a range of health benefits [113] In the case of ellagic acid, research has suggested that it may have anticancer properties and may be beneficial in the prevention and treatment of various types of cancer [114]. Ellagic acid is also believed to have anti-inflammatory and antimicrobial effects, which may further contribute to its potential health benefits [115]. While ellagic acid is most commonly found in fruits and vegetables, such as strawberries, raspberries and pomegranates, it has also been identified in some species of fish. For example, research has shown that ellagic acid can be found in the muscle tissue of salmon and trout (Oncorhynchus mykiss) [116]. It is important to note, however, that the amount of ellagic acid present in fish is typically much lower than that found in fruits and vegetables. Therefore, while including fish in one’s diet may provide some small amount of ellagic acid, it is unlikely to have a significant impact on overall ellagic acid intake [117]. Overall, while ellagic acid may be a beneficial compound with potential health benefits, it is important to consider a variety of dietary sources, including fruits, vegetables, fish and other foods, to ensure adequate intake of this and other important nutrients [118].
Fisetin is a natural flavonoid that can be found in a variety of plants and fruits, such as strawberries, grapes, apples, persimmons, onions and cucumbers. It is also present in some fish, including salmon. Research has shown that fisetin possesses powerful antioxidant, anti-inflammatory and neuroprotective properties that may help defend the body against various diseases and health conditions [119]. Antioxidants help to neutralize harmful free radicals in the body, which can damage cells and contribute to the development of chronic diseases such as cancer, heart disease, and Alzheimer’s disease. By reducing oxidative stress, fisetin may help to prevent these conditions from developing [120]. Fisetin has also been found to have cardioprotective effects, meaning it can help protect the heart and cardiovascular system from damage. It may help lower blood pressure and reduce the risk of heart disease by improving blood flow and reducing inflammation in the arteries [121]. In addition to its potential cardiovascular benefits, fisetin has also been studied for its cancer-fighting properties. Some research has shown that fisetin can inhibit the growth and spread of certain types of cancer cells, including prostate, breast and colon cancer cells [122]. Furthermore, fisetin has also been shown to improve cognitive function and memory in some in vivo studies, suggesting that it may have potential benefits for brain health as well [123].
Overall, while more research is needed to fully understand the potential health benefits of fisetin, the current evidence suggests that this natural compound may have a range of health-promoting properties, including antioxidant and anti-inflammatory effects, cardiovascular protection, cancer prevention and potential benefits for brain health [119].
3.1.3. Shellfish
Shellfish, such as shrimps, clams and oysters, are also a source of marine polyphenols and other minor nutrients. The most common compounds found in shellfish are carotenoids such as astaxanthin and zeaxanthin, which have antioxidant and anti-inflammatory properties [124]. These polyphenols are derived from algae and other marine organisms that are consumed by shellfish as part of their diet [3]. One example of a marine polyphenol are the catechins, which are also found in tea, and procyanidins, which are found in various fruits, vegetables and brown seaweeds [17]. These polyphenols are believed to have a range of health benefits, including antioxidant and anti-inflammatory effects [125].
Anther minor nutrient found in shellfish is fucoxanthin, which is a type of carotenoid that is found in brown seaweed. Fucoxanthin has been shown to have antioxidant, anti-inflammatory and anti-obesity properties [126]
3.1.4. Sponges
Despite being a rich source of highly bioactive chemicals [127], there has been little research in the literature on the extraction and identification of polyphenols in sponges. Traditionally, methanol and dichloromethane were utilized for extraction; however, some novel phenolic compounds have been discovered. Bisabolenes are polyphenolic chemicals discovered in sponges that are particularly fascinating. All sponge bisabolenes have a distinct 7S structure, whereas other marine and terrestrial bisabolenes have a 7R structure [127]. (S)-(+)-curcuphenol, a member of this family discovered in sponges, has a variety of biological activities [128].
3.1.5. Marine Fungi
Several Benzaldehyde compounds produced from marine fungus have also sparked interest due to their scavenging characteristics. Wang et al. discovered and characterized chaetopyramin, a scavenging metabolite isolated from the marine fungus Chaetomium globosum (Ascomycota) and the red algae Polysiphonia stricta (formerly Polysiphonia urceolata). Chaetopyramin was synthesized along with known derivatives isotetrahydroauroglaucin and 2-(2′,3′-epoxy-1′,3′-heptadienyl)-6-hydroxy-5-(3-methyl-2-butenyl)benzaldehyde, having DPPH IC50 values of 35, 26 and 88 g/mL, respectively [128].
In this case, two additional benzaldehyde derivatives, flavoglaucin and isodihydroauroglaucin, were obtained from the marine fungus Microsporum sp. These metabolites, renowned for their DPPH scavenging capacity due to the inclusion of two phenolic hydroxyl groups, demonstrated considerable action, with IC50 values in the range of 11.3 and 11.5 g/mL, making them more effective than ascorbic acid (20 g/mL) [128].
The hydroquinone farnesylhydroquinone and its oxidized counterpart, sesquiterpene quinone, were discovered from the marine fungus Penicillium sp., and Farnesylhydroquinone (IC50 12.5 M) was shown to be a greater DPPH radical scavenger than ascorbic acid (IC50 22.5 M) [129].
3.1.6. Sea Urchins
The existence of polyhydroxylated naphthoquinone (PHNQ) pigments in sea urchins has long been recognized and investigated [130]. They are concentrated in the shells or gonads, and it has been proposed that they, like other polyphenolic components from edible plants, may be used as antioxidants. Indeed, PHNQs extracted from sea urchin gonads have been demonstrated to be potent antioxidants in lipid peroxidation and food systems [131,132].
However, their use may be hampered by their poor yield and restricted by their brown/orange coloration. The structures of polyhydroxylated naphthoquinone pigments reveal that they are easily reduced and re-oxidized. As a result, their stability is critical for future medical applications. Alternatively, their distinctive quinone structure, along with their structural diversity, may lead to the discovery of novel bioactivities that are more relevant to biological applications [130,133].
3.2. Phenolic Compounds Metabolomics
There is a natural necessity of extrinsic or intrinsic drivers to make seaweed cellular systems to create naturally and/or enhance/trigger its production from one molecule or a class of chemical to be generated by a specimen in nature or in aquaculture. Primarily (primary metabolites), phenolic compounds (primary and secondary metabolites) are produced naturally and inherently in basic conformations. When seaweed cells are activated in stressful settings, they develop more complex forms [19] As a result, the presence of phenolic chemicals is invariably recognized in cells [19]
Extrinsic factors, on the other hand, activate cellular defensive responses, which can shift the molecular mechanism to produce greater quantities and a wider range of conformations of a specific compound class, particularly when it is a defensive compound synthesized to protect against external attacks [134,135].
If the drivers of seaweed compound production are fully understood, the exploitation of phenolic compounds and their bioactivity can be moved into kinetic models, providing more exploitation safety and information on how to explore phenolic compounds efficiently with lower costs and higher quality [136,137]. Thus, the cultivation of the marine organism under controlled conditions can be a feasible system to produce and obtain a natural phenolic compound that can be applied commercially. One of the examples is the Dieckol from the brown seaweed Ecklonia radiata, which is already applied in cardiovascular therapeutics [19].
4. Structure and Properties of Marine Polyphenols
The basic structure of marine polyphenols consists of multiple phenolic rings linked together by various chemical bonds. These rings can be modified with other chemical groups, such as sugars or sulfates, which can further influence their properties [138].
One of the unique features of marine polyphenols is their ability to form complex aggregates or “tannins” through intermolecular interactions such as hydrogen bonding and hydrophobic interactions. These tannins can have different physical and chemical properties compared to their monomeric counterparts, including increased solubility and stability [139].
Another important property of marine polyphenols is their potential to be used as natural food preservatives. Some marine polyphenols, such as the phlorotannins found in brown seaweed, have been shown to inhibit the growth of various bacteria and fungi, which can help to extend the shelf life of food products [15].
Because of their structural variety and unpredictability, phenolic compounds from marine creatures are significantly less researched than those from terrestrial sources. However, their biological significance and prospective features make them an appealing category deserving of more scientific investigation. The utilization of effective extraction and, in certain circumstances, purifying processes can provide new bio-actives valuable for food, nutraceutical, cosmeceutical and pharmaceutical applications. The bioactivity of marine phenolics is due to their enzyme inhibitory action as well as antibacterial, antiviral, anticancer, antidiabetic, antioxidant or anti-inflammatory properties [3]. The marine ecosystem can be exploited by aquaculture techniques, causing less impact in terrestrial ecosystem.
4.1. Some Phenolic Compound Structures and Bioactivities
4.1.1. Phenolic Acids (PAs)
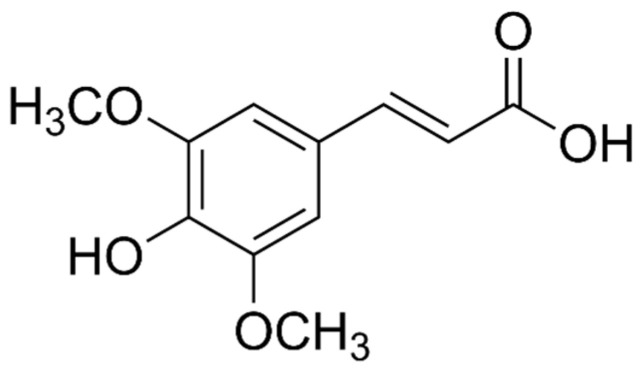
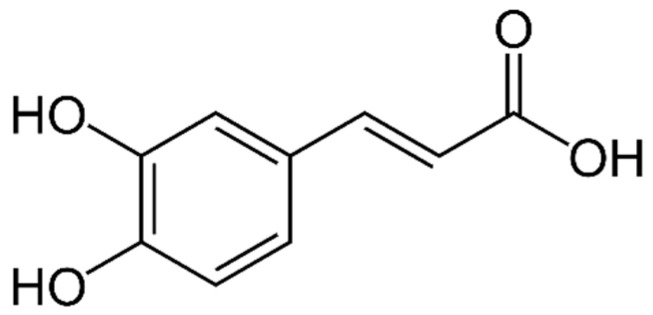
There are two main types of PAs: hydroxybenzoic acids (HBAs) (Figure 2) and hydroxycinnamic acids (HCAs). HBAs include compounds such as gallic acid (Figure 3), protocatechuic acid (Figure 4) and syringic acid (Figure 5), while HCAs include compounds such as caffeic acid (Figure 6), ferulic acid and sinapic acid (Figure 7) [68].
Figure 2.

Hydroxybenzoic acid (HBA).
Figure 3.

Gallic acid.
Figure 4.
Protocatechuic acid.
Figure 5.
Syringic acid.
Figure 6.

Caffeic acid.
Figure 7.

Sinapic acid.
The properties of PAs can vary depending on their structure and the position of the hydroxyl and carboxylic acid groups on the phenolic ring. Some common characteristics of PAs include:
Antioxidant activity: PAs are known to have strong antioxidant activity due to their ability to scavenge free radicals and inhibit lipid peroxidation [140].
Anti-inflammatory activity: PAs have been shown to have anti-inflammatory effects, which may be due to their ability to inhibit the production of inflammatory mediators such as cytokines and prostaglandins [141].
Antimicrobial activity: Some PAs have been shown to have antimicrobial activity against various bacteria and fungi, which may be due to their ability to disrupt microbial cell membranes or inhibit enzyme activity [142].
Absorption and metabolism: PAs are absorbed in the small intestine and metabolized by the liver. The degree of absorption and metabolism can vary depending on the structure of the PA and the presence of other dietary components [143].
4.1.2. Phlorotannins
Phlorotannins, as mentioned earlier, are phenolic compounds that are primarily found in brown algae (Phaeophyceae). Here are some of their characteristics and structures:
Chemical structure: Phlorotannins are phloroglucinol polymers that are formed by the bonding of phloroglucinol units through ether linkages. There are various types of phlorotannins, based on the number of phloroglucinol units they contain and the nature of the linkages between these units [144].
Antioxidant properties: Phlorotannins are known for their strong antioxidant properties, which make them useful in a variety of medical and cosmetic applications [145].
Potential antimicrobial activity: Some studies indicate that phlorotannins may have antimicrobial activity, which could make them useful in the treatment of infections [146].
Potential anti-inflammatory activity: Some studies suggest that phlorotannins may have anti-inflammatory properties, which could make them useful in the treatment of inflammatory conditions [145].
Phlorotannins are characterized by their complex structure, which typically consists of multiple phloroglucinol units (Figure 8) linked by ether or carbon–carbon bonds. Phlorotannins can vary in size and degree of polymerization, with some larger molecules containing more than 20 phloroglucinol units [144].
Figure 8.

Phloroglucinol.
4.1.3. Catechins
Catechins are a type of flavonoid polyphenol found in green tea, but they are also present in some marine algae. They are characterized by a structure that consists of two phenolic rings linked by a carbon–carbon bond, with hydroxyl groups attached to the rings. Catechins can have various substitutions on the rings, which can affect their biological activity [2]. Both rings have hydroxyl groups (-OH) in positions 3 and 4, and in ring B there may be a hydroxyl group in position 5. The position of hydroxyl groups and other substitutions in ring B and ring C can vary, generating different types of catechins with specific biological activities. Some examples of catechins are: epicatechin (EC), epicatechin-3-gallate (ECG) (Figure 9), epigallocatechin (EGC), epigallocatechin-3-gallate (EGCG) (Figure 10), gallocatechin (GC) (Figure 11) and catechin (C) (Figure 12) [147].
Figure 9.

Epicatechin (-3-gallate ECG).
Figure 10.
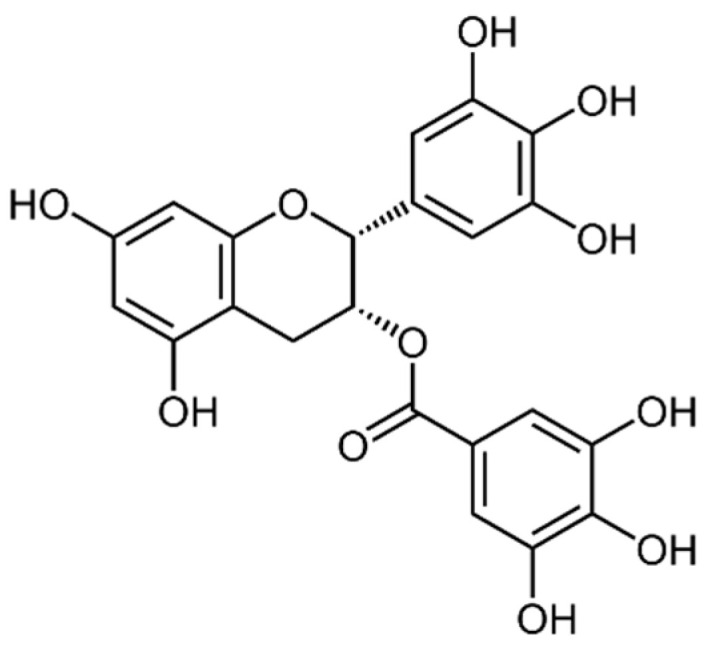
Epigallocatechin-3-gallate (EGCG).
Figure 11.

Gallocatechin (GC).
Figure 12.

Catechin (C).
Some of the main bioactivities of catechins include:
Antioxidant activity: Catechins have strong antioxidant properties and can scavenge free radicals and reactive oxygen species, which can cause oxidative damage to cells and contribute to various diseases [148]
Anti-inflammatory activity: Catechins have been shown to have anti-inflammatory effects, which may help to reduce the risk of chronic diseases such as cardiovascular disease, diabetes and cancer [149].
Anti-cancer activity: Several studies have suggested that catechins may have anti-cancer properties, particularly in reducing the risk of breast, prostate and colon cancer [150].
Anti-obesity activity: Catechins have been shown to have an anti-obesity effect, particularly by promoting fat oxidation and reducing fat accumulation in the body (2023).
Neuroprotective activity: Catechins have been shown to have neuroprotective effects, which may help to reduce the risk of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease [151].
Cardiovascular protection: Catechins may help to protect against cardiovascular disease by reducing the risk of hypertension, lowering LDL cholesterol levels and improving endothelial function [152].
Anti-diabetic activity: Catechins may help to regulate blood sugar levels and improve insulin sensitivity, which may be beneficial for people with type 2 diabetes [149].
4.1.4. Bromophenols
Bromophenols (Figure 13) are a type of polyphenol that contain one or more bromine atoms in addition to the phenolic rings. They are found in some marine organisms such as red algae and sponges [153]. Bromophenols can have various structures, with some containing one phenolic ring and others containing two or more rings [23].
Figure 13.

Bromophenol.
Some of the main bioactivities of bromophenols include:
Antioxidant activity: Bromophenols have been shown to have strong antioxidant properties, which can help to protect cells from oxidative damage caused by free radicals and reactive oxygen species [154].
Anti-inflammatory activity: Bromophenols have been shown to have anti-inflammatory effects, which may help to reduce the risk of chronic diseases such as cardiovascular disease, diabetes and cancer [155].
Anti-tumor activity: Several studies have suggested that bromophenols may have anti-tumor properties, particularly in reducing the growth and proliferation of cancer cells [138].
Antibacterial and antiviral activity: Bromophenols have been shown to have antibacterial and antiviral properties, which may help to prevent and treat infections [156].
Neuroprotective activity: Bromophenols have been shown to have neuroprotective effects, which may help to reduce the risk of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease [157].
Cardiovascular protection: Bromophenols may help to protect against cardiovascular disease by reducing the risk of hypertension, lowering LDL cholesterol levels and improving endothelial function [158].
Anti-diabetic activity: Bromophenols may help to regulate blood sugar levels and improve insulin sensitivity, which may be beneficial for people with type 2 diabetes [159].
4.1.5. Flavonoids
Flavonoids are a diverse class of naturally occurring compounds found in many marine algae, fruits, vegetables and herbs. They are characterized by their unique chemical structure, which consists of two aromatic rings linked by a three-carbon bridge [2]. Flavonoids have a wide range of bioactivities, including:
Antioxidant activity: Flavonoids are well-known for their antioxidant properties, which help to protect cells from oxidative damage caused by free radicals and reactive oxygen species [160].
Anti-inflammatory activity: Many flavonoids have been shown to have anti-inflammatory effects, which may help to reduce the risk of chronic diseases such as cardiovascular disease, diabetes and cancer [161].
Anti-cancer activity: Some flavonoids have been shown to have anti-cancer properties, particularly in reducing the growth and proliferation of cancer cells [162].
Neuroprotective activity: Flavonoids have been shown to have neuroprotective effects, which may help to reduce the risk of neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease [163].
Cardiovascular protection: Flavonoids may help to protect against cardiovascular disease by reducing the risk of hypertension, lowering LDL cholesterol levels and improving endothelial function [164].
Anti-diabetic activity: Flavonoids may help to regulate blood sugar levels and improve insulin sensitivity, which may be beneficial for people with type 2 diabetes [159].
Some examples of flavonoids and their bioactivities include:
Quercetin (Figure 14): Quercetin is a flavonoid found in many fruits and vegetables, including onions, apples and berries. It has been shown to have antioxidant, anti-inflammatory, anti-cancer and neuroprotective properties [165].
Figure 14.

Quercetin.
Epigallocatechin gallate (EGCG) (Figure 10): EGCG is a flavonoid found in green tea. It has been shown to have antioxidant, anti-inflammatory, anti-cancer and cardiovascular protective properties [166].
Hesperidin (Figure 15): Hesperidin is a flavonoid found in citrus fruits. It has been shown to have antioxidant, anti-inflammatory and cardiovascular protective properties [167].
Figure 15.
Hesperidin.
Kaempferol (Figure 16): Kaempferol is a flavonoid found in many plants, including broccoli, kale and tea. It has been shown to have antioxidant, anti-inflammatory, anti-cancer and neuroprotective properties [168].
Figure 16.

Kaempferol.
4.1.6. Phenolic Terpenoids
Phenolic terpenoids, also known as terpenophenolics, are a class of natural compounds that consist of a terpenoid backbone (a linear or cyclic hydrocarbon chain) and one or more phenolic groups [19]. They are produced by a wide range of plants and brown and red seaweeds, and they have a diverse array of bioactivities, including:
Antioxidant activity: Phenolic terpenoids are potent antioxidants that can protect cells from oxidative stress caused by free radicals and reactive oxygen species [169].
Anti-inflammatory activity: Many phenolic terpenoids have anti-inflammatory effects, which can help to reduce inflammation in the body and prevent chronic diseases [170].
Anti-cancer activity: Some phenolic terpenoids have been shown to have anti-cancer properties, including inhibiting tumor growth and inducing cancer cell death [171].
Cardiovascular protection: Phenolic terpenoids may help to protect against cardiovascular disease by reducing oxidative stress, inflammation and lipid peroxidation, and improving vascular function [172].
Anti-microbial activity: Some phenolic terpenoids have been shown to have anti-microbial properties, which can help to prevent and treat infections [173].
Neuroprotective activity: Phenolic terpenoids may have neuroprotective effects, including protecting against oxidative damage, reducing inflammation and improving cognitive function [174].
Examples of phenolic terpenoids and their bioactivities include:
Rosmarinic acid (Figure 17): Rosmarinic acid is a phenolic terpenoid found in many herbs, including rosemary and sage. It has antioxidant, anti-inflammatory and anti-microbial properties, and may also have neuroprotective effects [175].
Figure 17.
Rosmarinic acid.
Ursolic acid (Figure 18): Ursolic acid is a pentacyclic triterpenoid found in many fruits and herbs, including apples, rosemary and basil. It has anti-inflammatory, anti-cancer and neuroprotective properties, and may also help to improve cardiovascular health [176].
Figure 18.
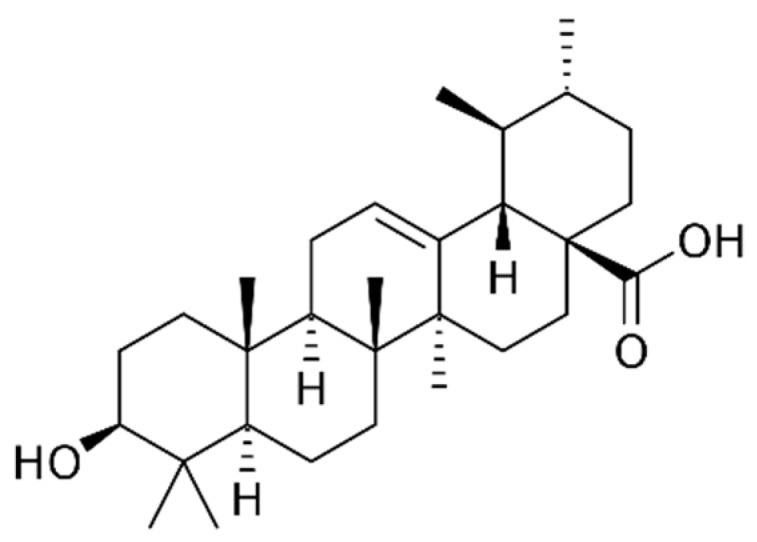
Ursolic acid.
Carnosic acid (Figure 19): Carnosic acid is a phenolic diterpene found in rosemary. It has antioxidant, anti-inflammatory and neuroprotective properties, and may also have anti-cancer effects [177].
Figure 19.

Carnosic acid.
Curcumin (Figure 20): Curcumin is a polyphenolic terpenoid found in turmeric. It has antioxidant, anti-inflammatory, anti-cancer and neuroprotective properties, and may also help to improve cardiovascular health [178].
Figure 20.
Curcumin.
Overall, phenolic terpenoids have a wide range of bioactivities that may help to promote health and prevent chronic diseases. However, more research is needed to fully understand the mechanisms of action and potential therapeutic applications of these compounds [179].
4.1.7. Mycosporine-like Amino Acids (MAA)
Mycosporine-like amino acids (MAAs) are a class of water-soluble, low molecular weight compounds that are widely distributed in marine organisms, including cyanobacteria, algae and some invertebrates [180]. They are produced as a response to UV radiation and act as a photoprotective agent, absorbing UV radiation and dissipating it as heat. MAAs have also been found in some marine and terrestrial organisms, including algae, lichens and fungi [181].
MAAs have a unique structure that consists of a cyclohexenone or cyclohexenimine chromophore linked to one or more amino acids [134]. The specific structure and number of amino acids can vary depending on the organism and environmental conditions. Some examples of MAAs and their bioactivities include:
Shinorine (Figure 21): Shinorine is an MAA found in red algae. It has been shown to have antioxidant, anti-inflammatory and UV-protective properties [182].
Figure 21.

Shinorine.
Porphyra-334: Porphyra-334 is an MAA found in red algae (Rhodophyta). It has been shown to have UV-protective properties and may also have anti-inflammatory effects [183].
Mycosporine-glycine (Figure 22): Mycosporine-glycine is an MAA found in many marine organisms, including cyanobacteria and algae. It has been shown to have antioxidant and anti-inflammatory properties and may also have neuroprotective effects [184].
Figure 22.

Mycosporine-glycine.
Palythine (Figure 23): Palythine is an MAA found in some invertebrates, including jellyfish and sea anemones. It has been shown to have antioxidant and anti-inflammatory properties and may also have neuroprotective effects [185].
Figure 23.

Palythine.
MAAs are known to have several bioactivities, including:
UV-protective activity: MAAs are known for their ability to protect organisms from UV radiation by absorbing UV light and dissipating it as heat. This helps to prevent damage to DNA and other cellular structures caused by UV radiation [181].
Antioxidant activity: MAAs have been shown to have antioxidant properties, which can help to protect cells from oxidative damage caused by free radicals and other reactive oxygen species [186].
Anti-inflammatory activity: Some MAAs have been shown to have anti-inflammatory effects, which may help to reduce inflammation in the body and prevent chronic diseases [187].
Neuroprotective activity: MAAs may have neuroprotective effects, including protecting against oxidative damage and reducing inflammation in the brain [155].
Overall, MAAs are a unique class of compounds with a wide range of bioactivities that are important for the survival of marine organisms in UV-rich environments. More research is needed to fully understand the mechanisms of action and potential therapeutic applications of these compounds [188].
4.1.8. Non-Typical Phenolic Compounds
Some examples of non-typical phenolic compounds and their bioactivities:
The class of oligomeric polyphenolic compounds known as Cladophorols (Figure 24) were initially discovered and characterized in the green algae Cladophora socialis (Chlorophyta) [29]. These compounds have exhibited noteworthy antimicrobial properties, particularly against methicillin-resistant Staphylococcus aureus (MRSA). Cladophorol C, a specific compound within this class, has displayed strong selective antibacterial activity against pathogenic MRSA, with a minimum inhibitory concentration (MIC) of 1.4 µg/mL [29].
Figure 24.

Cladophorol.
Several phenolic compounds have been identified in different seaweed species. Colpol, a phenolic compound, has been identified in brown seaweeds, while tichocarpols, a phenylpropanoid derivative, have been identified in the red algae species Tichocarpus crinitus (Rhodophyta) [77].
5. Phenolic Compound Extraction and Isolation
Pre-treatment with seaweed is advised, such as a washing step to remove stones, sand, epiphytes or other contaminants. As a result, algal biomass can be utilized fresh, dried (air drying or at 30–40 °C with aeration for 3–5 days) or freeze dried [189]. Freeze-dried is preferable because it preserves the integrity of the biomolecules and allows for higher extraction yields [190].
A milling or grinding step is also advised to lower particle size, which would enhance the exposure area between the seaweed biomass and the solvent used for extraction [191]. As a result, the extraction yield will rise.
To avoid co-extraction of pigments or fatty acids [28] with low polar solvents—n-hexane, n-hexane:acetone, n-hexane:ethyl acetate or dichloromethane—a pre-extraction step is usually necessary [19]. The next step is to choose an extraction method, as these approaches vary greatly.
Soxhlet, solid–liquid and liquid–liquid extractions are examples of traditional extraction procedures. Organic solvents (e.g., hexane, petroleum ether, cyclohexane, ethanol, methanol, acetone, benzene, dichloromethane, ethyl acetate, chloroform) are often utilized in the listed techniques. Nonetheless, the solvent used in extraction processes should be non-toxic and inexpensive [192]. Because of its cheaper cost, ethanol is used as an extraction solvent in the industrial sector.
These approaches have changed throughout time to increase extraction efficiency and sustainability as technology has advanced. Currently, ultrasound and microwave-assisted extraction are low-cost, large-scale methods [19].
Following the extraction procedure, the isolated and quantified target phenolic component must be isolated. Depending on the type of substance to be separated, several techniques might be used.
In general, the source of phenolic compounds, the extraction and purification processes used, the sample particle size, the storage conditions and the presence of interfering components in extracts such as fatty acids or pigments all impact the results [19].
Today, phenolic compounds are isolated using preparative chromatography techniques such as column chromatography, high-pressure liquid chromatography (HPLC) or thin-layer chromatography (TLC). However, these chromatographic methods have been developed to be employed for the separation, isolation, purification, identification and quantification of many phenolic substances [193].
Due to these costly procedures, they are still in the initial stage to exploit marine phenolics compounds in an efficient way, although they are being studied to be further applied in pharmaceutics.
6. Marine Polyphenols Action Mechanisms
Marine polyphenols are a diverse group of compounds that include flavonoids, phenolic acids and stilbenes, among others. They are synthesized by marine organisms as a defense mechanism against environmental stressors, such as UV radiation, pathogens and predators [3]. Marine polyphenols have been found to exhibit a wide range of biological activities, including anti-inflammatory, anticancer, antiviral, antimicrobial and neuroprotective effects [19].
One of the key mechanisms by which marine polyphenols exert their biological effects is through their ability to interact with cellular signaling pathways. For example, marine polyphenols have been found to modulate the activity of the enzymes involved in cell proliferation, differentiation and apoptosis [194]. This can lead to the inhibition of cancer cell growth and the induction of cell death. Marine polyphenols can also regulate the expression of genes involved in inflammation, such as cytokines and chemokines, thereby reducing inflammation [195].
One of the primary mechanisms of action of marine polyphenols is their ability to scavenge free radicals and reactive oxygen species (ROS) in the body. Free radicals and ROS can damage cells and tissues, leading to inflammation, aging, and chronic diseases. Marine polyphenols have been shown to neutralize free radicals and prevent oxidative stress, thereby protecting cells and tissues from damage [196].
A mechanism by which marine polyphenols exert their effects is through their interaction with cellular membranes. Polyphenols can interact with the lipid bilayer of the membrane, altering its physical properties, such as its fluidity and permeability. This can lead to changes in membrane-associated signaling pathways, affecting cellular functions such as ion transport, receptor activity, and intracellular signaling [197].
Another mechanism of action of marine polyphenols is their ability to modulate the expression of genes and proteins involved in various cellular pathways. For example, marine polyphenols can activate or inhibit enzymes, such as kinases and phosphatases, involved in signal transduction pathways, leading to altered cellular responses. Marine polyphenols can also regulate the expression of transcription factors, such as nuclear factor-kappa B (NF-κB), which plays a critical role in inflammation and immune responses [195,198].
Marine polyphenols can also modulate the gut microbiota, which has important implications for human health. The gut microbiota plays a critical role in nutrient absorption, immune function and metabolic homeostasis [199]. Polyphenols can affect the composition and activity of the gut microbiota, promoting the growth of beneficial bacteria and reducing the growth of harmful bacteria. This can lead to improved gut health and a reduction in the risk of chronic diseases such as inflammatory bowel disease, obesity and type 2 diabetes [200].
Most of the marine phenolic compounds actuated in enzymes, such as cyclooxygenase (COX), work in tandem with nonsteroidal anti-inflammatory medicines (NSAIDs) to suppress the activity or gene expression of pro-inflammatory mediators. Various phenolic compounds can also operate on transcription factors such as nuclear factor-B (NF-B) or nuclear factor-erythroid factor 2-related factor 2 (Nrf-2) to upregulate or downregulate components in antioxidant response pathways. Phenolic chemicals have been utilized to treat a variety of common human disorders, including hypertension, metabolic difficulties, incendiary infections and neurodegenerative diseases, because they can block the enzymes involved in the development of human diseases. Phenolic chemicals have been used to treat hypertension by inhibiting the angiotensin-converting enzyme (ACE). Carbohydrate hydrolyzing enzyme inhibition is a type 2 diabetes mellitus medication, and cholinesterase inhibition is used to treat Alzheimer’s disease [201].
In addition to their biological activities, marine polyphenols have been found to have applications in various industries, such as food, pharmaceuticals and cosmetics [22]. For example, marine polyphenols are used as natural food preservatives due to their antimicrobial activity [202]. They are also used in the development of new drugs and therapies for various diseases, such as cancer and neurodegenerative disorders. Marine polyphenols are also used in the cosmetic industry due to their antioxidant and anti-aging properties [203].
6.1. Therapeutic Potential of Marine Polyphenols
6.1.1. Cardiovascular Diseases
Cardiovascular diseases (CVDs) are a leading cause of morbidity and mortality worldwide, and marine polyphenols have been studied extensively for their potential therapeutic effects in CVDs. Some of the ways in which marine polyphenols may be beneficial in CVDs [115] are as follows:
Antioxidant activity: Marine polyphenols have strong antioxidant properties, which can help reduce oxidative stress in the cardiovascular system. Oxidative stress has been implicated in the development and progression of CVDs, and reducing it may help improve cardiovascular health. Some of the main marine polyphenolic compounds with antioxidant activity include [204]:
Phlorotannins: These are a group of complex polyphenolic compounds found in brown seaweeds. Phlorotannins are known for their potent antioxidant activity, and they have been shown to have a wide range of health benefits, including anti-inflammatory and anti-cancer properties [15].
Catechins: These are flavonoid polyphenolic compounds found in green tea and some marine sources, such as seaweed [17]. Catechins have been shown to have potent antioxidant properties, and they may help reduce the risk of cardiovascular disease and other chronic diseases [158].
Flavonoids: These are a group of polyphenolic compounds found in a variety of plant and marine sources. Flavonoids have strong antioxidant properties, and they have been shown to have numerous health benefits, including reducing inflammation, improving cardiovascular health and reducing the risk of certain types of cancer [205].
Phenolic acids: These are a group of polyphenolic compounds found in a variety of marine sources, including marine algae. Phenolic acids have potent antioxidant properties, and they may help reduce the risk of cardiovascular disease and other chronic diseases by reducing oxidative stress [19].
Anti-inflammatory effects: Chronic inflammation is a key factor in the development of CVDs, and marine polyphenols have been shown to possess anti-inflammatory effects. By reducing inflammation, these compounds may help protect against CVDs [206]. Some of the most commonly studied compounds in this regard include:
Fucoidan: This is a sulfated polysaccharide found in brown seaweed and has been shown to possess anti-inflammatory effects by inhibiting the production of pro-inflammatory cytokines [207].
Phlorotannins: These are polyphenolic compounds found in brown seaweed and have been shown to possess anti-inflammatory effects by inhibiting the production of pro-inflammatory enzymes such as cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) [145].
Fucoxanthin: This is a carotenoid pigment found in brown seaweed and has been shown to possess anti-inflammatory effects by inhibiting the production of pro-inflammatory cytokines and reducing oxidative stress [20].
Eckol: This is a phlorotannin found in brown seaweed and has been shown to possess anti-inflammatory effects by inhibiting the production of pro-inflammatory cytokines and reducing oxidative stress [208].
Astaxanthin: This is a carotenoid pigment found in microalgae and has been shown to possess anti-inflammatory effects by inhibiting the production of pro-inflammatory cytokines and reducing oxidative stress [209].
Regulation of lipid metabolism: Dyslipidemia, or abnormal lipid levels in the blood, is a major risk factor for CVDs. Marine polyphenols have been shown to regulate lipid metabolism, potentially reducing the risk of CVDs [210]. Some of the main marine polyphenolic and other minor compounds that have been shown to regulate lipid metabolism and potentially reduce the risk of CVDs are:
Fucoxanthin: This is a carotenoid pigment found in brown seaweed. Fucoxanthin has been shown to reduce body weight, decrease total cholesterol and improve lipid metabolism in animal studies. It works by inhibiting the enzymes involved in the synthesis of cholesterol and triglycerides [211].
Phlorotannins: These are a group of polyphenolic compounds found in brown seaweed. Phlorotannins have been shown to reduce serum lipid levels by inhibiting the absorption of dietary fat and cholesterol. They also exhibit antioxidant and anti-inflammatory properties [15].
Fucoidan: This is a sulfated polysaccharide found in brown seaweed. Fucoidan has been shown to decrease triglyceride levels and improve lipid metabolism in animal studies. It works by inhibiting the activity of the enzymes involved in the synthesis of triglycerides [212].
Astaxanthin: This is a carotenoid pigment found in microalgae, yeast, salmon, trout, krill, shrimp, crayfish, crustaceans and the feathers of some birds. Astaxanthin has been shown to improve lipid metabolism by decreasing serum triglyceride and cholesterol levels. It also exhibits antioxidant and anti-inflammatory properties [213].
Vasodilatory effects: Some marine polyphenols have been shown to have vasodilatory effects, meaning they can help relax blood vessels and improve blood flow. This can help reduce blood pressure and improve cardiovascular health [214]. Some of the main marine polyphenolic and other minor nutrients that have been shown to regulate lipid metabolism and potentially reduce the risk of CVDs are:
Fucoxanthin: This is a carotenoid pigment found in brown seaweed. Fucoxanthin has been shown to reduce body weight, decrease total cholesterol and improve lipid metabolism in animal studies. It works by inhibiting the enzymes involved in the synthesis of cholesterol and triglycerides [215].
Phlorotannins: These are a group of polyphenolic compounds found in brown seaweed. Phlorotannins have been shown to reduce serum lipid levels by inhibiting the absorption of dietary fat and cholesterol. They also exhibit antioxidant and anti-inflammatory properties [216].
Fucoidan: This is a sulfated polysaccharide found in brown seaweed. Fucoidan has been shown to decrease triglyceride levels and improve lipid metabolism in animal studies. It works by inhibiting the activity of the enzymes involved in the synthesis of triglycerides [217].
Platelet inhibition: Platelet activation and aggregation play a key role in the development of thrombosis, which can lead to heart attacks and strokes. Marine polyphenols and other minor nutrients have been shown to inhibit platelet aggregation, potentially reducing the risk of thrombosis [218]. Some of the main ones are:
Fucoidan: Fucoidan is a sulfated polysaccharide found in various types of brown seaweed. It has been shown to inhibit platelet aggregation by inhibiting the binding of platelet activating factors to platelet receptors [219].
Phlorotannins: Phlorotannins have been shown to inhibit platelet aggregation by interfering with the release of platelet activating factors [220].
Catechins: Catechins, a type of flavonoid found in many types of seaweed, can inhibit platelet aggregation by inhibiting the activity of platelet-activating factors and reducing the adhesion of platelets to the blood vessel wall [221,222].
Eckol: Eckol is a type of phlorotannin found in brown seaweeds. It has been shown to inhibit platelet aggregation by interfering with the binding of platelet activating factors to platelet receptors [223].
6.1.2. Diabetes
Among the marine polyphenols that have been studied for their potential therapeutic effects in diabetes (Table 2), some of the most commonly studied include:
Fucoxanthin: This polyphenol has been shown to have anti-diabetic effects by improving insulin sensitivity and glucose metabolism in animal studies [224].
Phlorotannins: These polyphenols have been shown to have anti-diabetic effects by reducing blood glucose levels and improving insulin sensitivity in animal studies [225].
Fucoidan: This polysaccharide has been shown to have anti-diabetic effects by improving glucose metabolism and insulin sensitivity in animal studies [6].
Bromophenols: These polyphenols have been shown to have anti-diabetic effects by reducing blood glucose levels and improving insulin sensitivity in animal studies [226].
Catechins: These polyphenols have been shown to have anti-diabetic effects by improving insulin sensitivity and glucose metabolism in animal studies.
While these marine polyphenols have shown promising potential in animal studies, further research is needed to determine their efficacy and safety in humans before they can be recommended as a therapeutic option for diabetes [227].
Table 2.
Therapeutic potential of marine polyphenols for Diabetes.
| Seaweed | Compound | Animal/Cell Line | Effect | Reference |
|---|---|---|---|---|
| E. cava | fucodiphloroethol G, dieckol, 6,6′-bieckol, 7-phloroeckol, phlorofucofuroeckol-A | In vitro assay: α-glucosidase and α-amylase inhibitory activity | Inhibition of α-glucosidase (IC50 values ranged from 10.8 μM for dieckol to 49.5 μM for 7-phloroeckol) and α-amylase (IC50 values ranged from 125 μM for dieckol to <500 μM for the rest of compounds, except 7-phloroeckol with a value of 250 μM) activities |
[228] |
| Lessonia trabeculate | Polyphenol-rich extracts | In vitro assay: α-glucosidase and lipase activity | Inhibition of α-glucosidase and lipase activities (IC50 < 0.25 mg/mL) | [229] |
| F. vesiculosus | Crude extract and semi-purified phlorotannins composed by fucols, fucophlorethols, fuhalols and several other phlorotannin derivatives | In vitro assay: α-glucosidase, α-amylase and pancreatic lipase inhibitory activity | Inhibition of α-amylase (IC50~28.8–2.8 μg/mL), α-glucosidase (IC50~4.5–0.82 μg/mL) and pancreatic lipase (IC50~45.9–19.0 μg/mL) activities | [230] |
| Rhodomela confervoides | 3,4-dibromo-5-(2-bromo-3,4-dihydroxy-6-(ethoxymethyl)benzyl)benzene-1,2-diol) | In vitro: insulin resistant C2C12 cells treated with bromophenol (0.1–0.5 μM for phenol) | Inhibition of PTP1B activity (IC50~0.84 μM) Activation of insulin signaling and potentiate insulin sensitivity |
[231] |
| Rhodomela confervoides | 3-Bromo-4,5-bis(2,3-dibromo-4,5-dihydroxybenzyl)-1,2-benzenediol | In vitro: palmitate-induced insulin resistance in C2C12 cells treated with bromophenol (0.5–2.0 μM for phenol) | Inhibition of PTP1B activity (IC50~2 μM) Activation of insulin signaling and prevent palmitate-induced insulin resistance |
[232] |
| E. stolonifera | Phlorofucofuroeckol-A | In vitro assay for non-enzymatic insulin glycation | Inhibition of AGEs formation (IC50 29.50–43.55 μM for D-ribose and D-glucose-induced insulin glycation, respectively) | [233] |
| Ishige foliacea | Octaphlorethol A | In vitro: STZ-induced pancreatic β-cell damage (RINm5F pancreatic β-cells) (12.5–50.0 μg/mL for phenol) | Decreased the death of STZ-treated pancreatic β-cells Decreased the TBARS and ROS Increased the activity of antioxidant enzymes |
[234] |
| E. cava | 6,6-Bieckol, phloroeckol, dieckol and phlorofucofuroeckol | In vivo: high glucose-stimulated oxidative stress in zebrafish, a vertebrate model (10–20 μM of phenols) | Inhibition of high glucose-induced ROS and cell death Dieckol reduced the heart rates, ROS, NO and lipid peroxidation Dieckol reduced the overexpression of iNOS and COX-2 |
[235] |
| Ulva prolifera | Extract rich in flavonoids | In vivo: STZ-induced diabetic rats (150 mg/kg/day bw of phenol for 4 weeks by gavage) | Diminished the fasting blood glucose and improved oral glucose tolerance Hypoglycemic effect by increasing IRS1/PI3K/Akt and suppressing JNK1/2 in liver |
[236] |
6.1.3. Neurodegenerative Diseases
Neurodegenerative diseases are a group of chronic and progressive disorders that affect the nervous system and lead to the gradual loss of function of neurons. They include Alzheimer’s disease, Parkinson’s disease and Huntington’s disease, among others. The pathogenesis of these diseases is multifactorial and involves oxidative stress, inflammation and the accumulation of misfolded proteins [237].
Marine polyphenols are natural compounds found in various marine organisms, including seaweeds (Table 3), algae and marine animals. They have been shown to possess a wide range of biological activities, including antioxidant, anti-inflammatory and neuroprotective effects. Therefore, marine polyphenols have been investigated for their therapeutic potential in the prevention and treatment of neurodegenerative diseases [9].
The antioxidant properties of marine polyphenols can help reduce oxidative stress in neurons, which is a major contributor to neurodegeneration [238]. These compounds have been shown to scavenge free radicals, prevent lipid peroxidation and enhance the activity of antioxidant enzymes. Moreover, marine polyphenols can also modulate inflammatory pathways, reducing the release of pro-inflammatory cytokines and chemokines that contribute to neuronal damage [2].
Marine polyphenols have also been found to have neuroprotective effects by inhibiting the aggregation of misfolded proteins, such as amyloid-beta and tau in Alzheimer’s disease and alpha-synuclein in Parkinson’s disease. By preventing the accumulation of these proteins, marine polyphenols can help maintain neuronal function and prevent neuronal death [239].
Overall, the therapeutic potential of marine polyphenols in neurodegenerative diseases is promising, but more research is needed to fully understand their mechanisms of action and to develop effective treatments. Further studies should focus on identifying the most potent marine polyphenols and optimizing their delivery to the brain to maximize their therapeutic effects [240].
Phenolic compounds and other minor nutrients from marine sources have shown potential in the treatment of neurodegenerative diseases due to their antioxidant and anti-inflammatory properties [241]. Some of the main phenolic compounds of marine origin with potential in the treatment of neurodegenerative diseases include:
Phlorotannins: These are a type of polyphenol found in brown seaweed that have been shown to have neuroprotective effects. They have been shown to reduce oxidative stress and inflammation in the brain, which are two factors that contribute to neurodegeneration [242].
Fucoxanthin: This is a carotenoid pigment found in brown seaweed that has been shown to have anti-inflammatory and antioxidant properties. It has been shown to reduce inflammation in the brain and to protect against oxidative stress [243].
Fucoidan: This is a sulfated polysaccharide found in brown seaweed that has been shown to have neuroprotective effects. It has been shown to reduce inflammation in the brain and to protect against oxidative stress [244].
Halogenated phenols: These are phenolic compounds that are found in marine sponges and have been shown to have neuroprotective effects. They have been shown to protect against oxidative stress and to reduce inflammation in the brain [3].
Bromophenols: These are phenolic compounds that are found in marine algae and have been shown to have neuroprotective effects. They have been shown to protect against oxidative stress and to reduce inflammation in the brain [245].
Table 3.
Therapeutic potential of marine polyphenols for neurodegenerative diseases.
| Seaweed | Compound | Animal/Cell Line | Effect | Reference |
|---|---|---|---|---|
| E. cava | dieckol, 6,6′-bieckol, 8,8′-bieckol, eckol and phlorofucofuroeckol-A | In vitro: assays of AChE, BChE and BACE-1 activities -- In vitro: Jurkat clone E1–6 cells (GSK3β activity at 50 μM) |
Inhibition of AChE and BChE activities (IC50 16.0–96.3 μM and 0.9–29.0 μM, respectively) Inhibition of BACE-1 activity (18.6–58.3% at 1 μM) Inhibition of GSK3β activity (14.4–39.7% at 50 μM) |
[246] |
| E. bicyclis | eckols | In vitro: assays of AChE and BChE activities | Inhibition of AChE and BChE activities (IC50 2.78 and 3.48 μg/mL, respectively) | [247] |
| Gracilaria beckeri, Gelidium pristoides, U. rigida and E. maxima | Aqueous extracts composed by phloroglucinol, catechin and epicatechin 3-glucoside | In vitro: assays of AChE and BChE activities | High antioxidant potency Inhibition of AChE and BChE activities (IC50 49.41 and 52.11 μg/mL, respectively, for E. maxima) Inhibition of Aβ aggregation |
[248] |
| E. maxima, G. pristoides, Gracilaria gracilis and Ulva lactuca | Aqueous-ethanolic extracts containing phlorotannins, flavonoids and phenolic acids | In vitro: assays of AChE, BChE and BACE-1 activities | Inhibition of AChE and BChE activities (IC50 1.74–2.42 and 1.55–2.04 mg/mL, respectively) Inhibition of BACE-1 activity (IC50 0.052–0.062 mg/mL) Inhibition of Aβ aggregation |
[249] |
| E. cava | Phlorofucofuroeckol | In vitro: Glutamate-stimulated PC12 cells (10 μM of phenol) |
Increased the cell viability and attenuated glutamate excitotoxicity Inhibited the apoptosis in a caspase-dependent manner Regulated the production of ROS and attenuated mitochondrial dysfunction |
[250] |
| E. cava | Phloroglucinol | In vitro: Aβ-induced neurotoxicity in HT-22 cells (10 μg/mL) --- In vivo: 5XFAD mice, model of AD (acute, 1.2 μmol of phenol bilaterally delivery) |
Reduced the Aβ-induced ROS accumulation in HT-22 cells Ameliorated the reduction in dendritic spine density --- Attenuated the impairments in cognitive dysfunction |
[251] |
| E. maxima | Eckmaxol | In vitro: Aβ oligomer-induced neurotoxicity in SH-SY5Y cells (5–20 μM of phenol) | Prevented the Aβ oligomer-induced neurotoxicity Inhibition of GSK3β and ERK signaling pathway |
[252] |
| E. cava | eckol, 8,80-bieckol and dieckol | In vitro: Aβ 25–35-induced damage in PC12 Cells (1–50 μM of phenol) | Inhibition of pro-inflammatory enzymes preventing Aβ production and neurotoxicity on the brain | [253] |
| E. cava | dieckol, 6,6′-bieckol, 8,8′-bieckol, eckol and phlorofucofuroeckol-A | In vitro: assays of AChE, BChE and BACE-1 activities -- In vitro: Jurkat clone E1–6 cells (GSK3β activity at 50 μM) |
Inhibition of AChE and BChE activities (IC50 16.0–96.3 μM and 0.9–29.0 μM, respectively) Inhibition of BACE-1 activity (18.6–58.3% at 1 μM) Inhibition of GSK3β activity (14.4–39.7% at 50 μM) |
[246] |
| E. bicyclis | eckols | In vitro: assays of AChE and BChE activities | Inhibition of AChE and BChE activities (IC50 2.78 and 3.48 μg/mL, respectively) | [247] |
| Gracilaria beckeri, Gelidium pristoides, U. rigida and E. maxima | Aqueous extracts composed by phloroglucinol, catechin and epicatechin 3-glucoside | In vitro: assays of AChE and BChE activities | High antioxidant potency Inhibition of AChE and BChE activities (IC50 49.41 and 52.11 μg/mL, respectively, for E. maxima) Inhibition of Aβ aggregation |
[248] |
6.1.4. Cancer
As described earlier, polyphenols (Table 4) and other micronutrients are bioactive compounds found in plants and animals, and recently there has been a growing interest in marine polyphenols due to their therapeutic potential in several areas of health, including cancer [2,254].
Marine polyphenols are extracted from marine organisms such as algae, mollusks, corals, sponges and fish. They have a wide variety of health benefits, including antioxidant, anti-inflammatory, anticancer and immunomodulatory activities [255].
The anticancer activity of marine polyphenols has been observed in several in vitro and in vivo studies. They are able to induce cell death in cancer cells, inhibit cell proliferation, inhibit angiogenesis and modulate the immune response. These effects are important because uncontrolled cell proliferation, excessive angiogenesis and suppression of the immune response are hallmarks of tumor development [256].
Ellagic acid is a polyphenol present in kelp that has been shown to cause cell death in breast and colorectal cancer. It functions by blocking the expression of pro-inflammatory and pro-angiogenic genes in cancer cells. It has also been shown to boost the production of tumor suppressor proteins [257].
Phloroglucinol acid is another polyphenol found in marine sponges with anticancer activity against lung and prostate cancer cells. This polyphenol induces apoptosis (programmed cell death) in cancer cells and inhibits the formation of capillaries that are necessary for angiogenesis [138].
Another micronutrient of marine origin with therapeutic potential is fucoidan, a sulfated polysaccharide found in brown algae. Studies suggest that fucoidan has anticancer activity against several cancer cell lines, including breast, lung and colon cancer cells. This sulfated polysaccharide inhibits angiogenesis, modulates the immune response and induces apoptosis in cancer cells [258].
Fucoxanthin is a carotenoid pigment found in brown algae that has also been shown to have anticancer activity. This compound is capable of inhibiting the growth of liver and colon cancer cells, inhibiting cell proliferation and inducing apoptosis [211].
The eckol-family of phlorotannins stands out among the various phlorotannin structures due to its exceptional bioactivity, particularly its anti-tumoral properties [254].
Despite the therapeutic potential of marine polyphenols in cancer, more research is needed to fully understand their mechanisms of action and to develop new anticancer therapies based on these compounds. Furthermore, it is important to evaluate the safety and efficacy of these compounds in human clinical trials [259].
Table 4.
Therapeutic potential of marine polyphenols for cancer.
| Specie | Compound | Animal/Cell Line | Effect | Reference |
|---|---|---|---|---|
| E. bicyclis | Phlorofucofuroeckol A | In vitro: LoVo, HT-29, SW480 and HCT116 cells (25–100 μM of phenol) | Antiproliferative and pro-apoptotic properties Induced the apoptosis on colorectal cancer cells by ATF3 signalling pathway |
[260] |
| E. cava | Phloroglucinol | In vitro: MCF7, SKBR3 and BT549 cells (10–100 μM of phenol) In vivo: MDA-MB231 breast cancer cells implanted into mammary fat pads of NOD-scid gamma (NSG) mice, treated with phloroglucinol 4 times on alternate days (25 mg/kg bw by intratumoral injections) |
Antiproliferative effect by KRAS inhibition and its downstream PI3K/Akt and RAF-1/ERK signalling pathways | [261] |
| E. cava | Dieckol | In vivo: N-nitrosodiethylamime-induced hepatocarcinogenesis rats (40 mg/kg bw/day for 15 weeks administered orally) | Regulated the xenobiotic-metabolizing enzymes Induced the apoptosis by mitochondrial pathway Inhibited the invasion by decreasing PCNA expression Inhibited the angiogenesis by changing MMP-2 and MMP-9 activity and VEGF expression Anti-inflammatory activity by inhibiting NF-kB and COX2 |
[262] |
| E. cava | Dieckol | In vitro: EA.hy926 cells (10–100 μM of phenol) | Antiangiogenic activity by inhibiting the proliferation and migration of cells through MAPK, ERK and p38 signaling pathways | [263] |
| E. cava | Eckol | In vitro: on human HaCaT keratinocytes against PM2.5-induced cell damage (30 μM of phenol for 17 days) | Decreased ROS generation Protected the cells from apoptosis by inhibiting MAPK signaling pathway |
[264] |
| E. cava | Dieckol | In vivo: N-nitrosodiethylamime-induced hepatocarcinogenesis rats (40 mg/kg bw/day for 15 weeks administered orally) | Regulated the xenobiotic-metabolizing enzymes Induced the apoptosis by mitochondrial pathway Inhibited the invasion by decreasing PCNA expression Inhibited the angiogenesis by changing MMP-2 and MMP-9 activity and VEGF expression Anti-inflammatory activity by inhibiting NF-kB and COX2 |
[262] |
7. Safety and Toxicity of Marine Polyphenols
As previously stated, marine polyphenols are natural substances found in a variety of aquatic creatures, including seaweed, algae and shellfish. These compounds have received a significant amount of attention because of their possible health advantages, which include antioxidant, anti-inflammatory and anti-cancer properties. However, concerns have been raised regarding their safety and toxicity [9,18], mostly regarding their extraction and isolation methods, which can change their relative safety and toxicity; due to the diverse chemical structure and impurities, there is a need to standardize the procedure from extraction until the safety/toxicity assays.
Several studies have investigated the safety of marine polyphenols and their potential toxicity. Overall, the available evidence suggests that these compounds are generally safe for human consumption. However, there are some concerns regarding their potential toxicity at high doses [19]. To date, the bioavailability of seaweeds has not been well researched. More research and study are required in this sector. The majority of seaweed phenolic pharmacological and biological bioavailability investigations have used mice models. Animal investigations and in vitro studies have provided evidence that seaweed phenols protect against various illnesses. As a result, fresh research investigations are required to investigate and completely comprehend their bioavailability in humans (the proportion of the chemical that reaches the human circulatory system and has an active impact). Furthermore, there is more pharmacokinetics required in order to fully understand the marine phenolic potential in in vivo models, due to a general lack of information, where the therapeutics in use do not have full public data regarding this topic [19,201,265,266]. Although phenolic compounds appear to have a wide range of biological actions, the problem of safe dose must be addressed. Indeed, phenolic substances have bimodal pharmacological effects, in that they can be beneficial at low doses while being poisonous at large concentrations. However, much current research on the negative effects of phenolic chemicals is focused on cell investigations and animal models, with few human trials [266].
One of the main concerns is the potential for heavy metal contamination in marine organisms. Heavy metals such as lead, mercury and cadmium can accumulate in the tissues of marine organisms and can pose a health risk if consumed in large quantities. Therefore, it is essential to ensure that marine polyphenol supplements are sourced from reputable suppliers that test for heavy metal contamination [267].
Another concern is the potential for allergic reactions to marine polyphenols. Some people may be allergic to certain types of marine organisms or their products, which could lead to adverse reactions. Therefore, it is important to check for any allergies before consuming marine polyphenol supplements [268].
Furthermore, the effects of marine polyphenols on pregnant and breastfeeding women are not yet fully understood, and caution should be exercised when consuming these compounds during these periods [269].
In conclusion, marine polyphenols have shown potential health benefits, but it is essential to ensure their safety and minimize any potential toxicity. It is recommended to consume marine polyphenols in moderation and to obtain them from reputable sources. Furthermore, consulting with a healthcare expert before beginning any new supplement routine is always recommended [270].
Phenolic Compound Pharmacodynamics
Pharmacodynamics and pharmacokinetics depend on the bioavailability of themarine phenolic compounds and are conducted by the absorptive process across the intestine into the circulatory system, after food ingestion. Thus, bioavailability involves several processes, including liberation from a food matrix, absorption, distribution, metabolism and elimination phases. Several polyphenols can be ingested as either purified, isolated substances or in foods. During the absorption process, gastric acid from the stomach can cause initial modifications to oligomeric polyphenols. Following ingestion, glycosidic polyphenols are cleaved in the small intestine, releasing the glycoside radical. Lactase phlorizin hydrolase and cytosolic glucosidase are enzymes with an affinity for glucose, xylose and galactose. However, polyphenols that are not cleaved by these enzymes are not absorbed by the small intestine and can be cleaved into small molecules known as phenolic acids produced by intestinal bacteria. Polyphenol structures can also be involved in conjugation reactions, resulting in methyl, glucuronide or sulfate groups. The remaining polyphenols, especially those attached to rhamnose, can be processed by rhamnosidase released by the colonic microbiota. Following these absorptive processes, phenolics will typically follow one of four paths: (1) Excretion in the feces; (2) absorption by the mucosa of the intestines or the colon, followed by entry into the portal vein for delivery to the liver; (3) further conjugation in the liver can result in the addition of with methyl, glucuronide or sulfate groups, followed by release into the bloodstream for tissue absorption; and (4) excretion in the urine. However, the absorption kinetic is mostly determined by the physical and chemical properties of the bioactive substances, but it can also be impacted by the subject’s physiology (age, genetic profile, gender, lifestyle, etc.), resulting in a unique bioavailability profile. As a result, the half-life of bioactive chemicals can range from minutes (e.g., gallic acid) to hours (e.g., rutin) [201,266].
8. Challenges and Opportunities in the Use of Marine Polyphenols as a Therapy
Due to these activities, there has been increasing interest in using marine polyphenols as a therapy for various diseases. However, the use of marine polyphenols as a therapy also presents several challenges. One of the challenges is the identification and isolation of specific marine polyphenols with therapeutic potential [9,18]. Marine organisms contain a vast array of compounds, and it can be challenging to isolate specific polyphenols with therapeutic potential. Additionally, there is a general absence of standardized techniques for the extraction and purification of marine polyphenols, which can affect the quality and consistency of the final product [12].
Another challenge is the limited knowledge of the pharmacokinetics and pharmacodynamics of marine polyphenols. Unlike synthetic drugs, marine polyphenols have complex structures that can affect their bioavailability, absorption, distribution, metabolism and excretion. This complexity can make it challenging to determine the optimal dose and frequency of administration of marine polyphenols [271].
Despite these challenges, there are several opportunities in the use of marine polyphenols as a therapy. One of the opportunities is the development of new therapies for diseases that currently have limited treatment options. For example, marine polyphenols have shown promising results as a therapy for diverse types of cancer, including breast, colon and prostate cancer. They have also shown potential as a therapy for neurodegenerative diseases such as Alzheimer’s and Parkinson’s [2].
Another opportunity is the development of new products for the food and cosmetic industries. Marine polyphenols have been shown to possess anti-aging and skin-whitening properties, making them attractive ingredients for the cosmetic industry [22]. Additionally, marine polyphenols have been shown to have antimicrobial properties, making them potential additives for food preservation [202].
Therefore, the use of marine polyphenols as a therapy presents both challenges and opportunities. Despite the challenges, the potential benefits of marine polyphenols in the treatment of various diseases and the development of new products make them a promising area of research. Further studies are needed to address the challenges and fully exploit the opportunities in the use of marine polyphenols as a therapy [18].
However, to obtain the benefits of phenolic compounds’ biological activities, they must be consumed. These chemicals have traditionally been included directly into meals, but their instability during food processing, distribution and storage, as well as their limited absorption and bioavailability in the gastrointestinal system, restrict their activity and health effects. Similarly, topical polyphenol usage is restricted due to their rapid oxidation, which causes food browning and the creation of undesirable aromas, as well as a decrease in activity [266,272]. Their encapsulation or application as typical drugs on a delivery system can potentiate their bioactivity and respective benefits [265,272,273].
Approved Polyphenolic Therapeutics
The most explored seaweed components are phenolic compounds, which are currently used in commercial solutions (for example, cosmetic items). Normally, phenolic compounds are not separated because commercial seaweed extracts include a high concentration of phenols [19].
The European Food Safety Authority has certified SeapolynolTM (Botamedi Inc, Seoul, Korea) as a food supplement. This supplement is based on dieckol and other polyphenols derived from E. cava; it has been evaluated and shown to be effective as an anti-hyperlipidemic and cardioprotective agent against doxorubicin-induced cardiotoxicity. Furthermore, SeapolynolTM improved insulin sensitivity in type 2 diabetes and may play an important role in the prevention of metabolic diseases [19,274,275,276,277] These tests, however, were carried out only on mice. The primary goals of phlorotannin supplements in cardiovascular illness are to avoid arteriosclerosis and enhance protective high-density lipoprotein cholesterol (HDL-C). HealSeaTM (made by Diana Naturals in Rennes, France), IdAlgTM (manufactured by Bio Serae in Bram, France) and SeanolTM (produced by LiveChem in Jeju-do, South Korea and sold by Simple Health in Maitland, USA) are phlorotannin-containing products [19,278]. InSea2TM (Rimouski, QC, Canada), a commercial combination of A. nodosum and F. vesiculosus phlorotannins, promotes a 90% decrease in postprandial blood glucose while lowering peak insulin production by 40% [19].
Among the sea urchin pigments, a cardiovascular drug, Histochrome (solution of Echinochrome A sodium salt), is the only compound that has been approved for clinical use. It is used in solutions of 10 mg/mL for cardiology and 2 mg/mL for ophthalmology. It is recommended for clinical use in patients with acute myocardial infarction to decrease incidence of ventricular extra systole and episodes of accelerated idioventricular rhythm after thrombolytic therapy [133].
9. Conclusions and Future Perspectives
The therapeutic potential of polyphenols and other micronutrients of marine origin has acquired significant attention in recent years due to their numerous health benefits. Polyphenols are a class of natural compounds found in several marine organisms that have antioxidant and anti-inflammatory properties. Other micronutrients found in marine sources include omega-3 fatty acids, vitamins and minerals, all of which have been linked to various health benefits [2].
Polyphenols and other micronutrients of marine origin have been shown in studies to help avoid and cure a variety of health problems, including cardiovascular disease, cancer, diabetes and neurodegenerative illnesses. Omega-3 fatty acids, for example, have been shown to promote cardiac health by decreasing inflammation, improving blood lipid levels, and lowering blood pressure. Polyphenols have also been shown to have anti-cancer properties by inhibiting the growth of cancer cells and promoting their death [279].
In addition to their therapeutic potential, marine-based polyphenols and micronutrients are also being investigated for their potential use in cosmetic and skincare products. Studies have shown that marine-derived compounds can have a helpful influence on skin health by decreasing inflammation, improving collagen production and protecting against UV damage [22].
Despite the promising potential of marine-based polyphenols and other micronutrients, there is still much research to be done to completely identify their mechanisms of action and potential side effects. However, the growing body of evidence suggests that marine-based compounds have significant therapeutic potential and should be further investigated for their potential use in disease prevention and treatment [13,254].
In conclusion, the therapeutic potential of marine-based polyphenols and other micronutrients is a promising area of research that has the potential to significantly impact human health [2]. Ongoing research into the mechanisms of action and potential side effects of these compounds will provide valuable insights into their therapeutic potential and pave the way for the development of new therapies and preventive measures. As such, marine-based compounds should be further investigated as a valuable resource for disease prevention and treatment [280].
Author Contributions
Writing—original draft preparation, L.P.; writing—review and editing, L.P. and J.C.; visualization, J.C.; supervision, L.P. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was financed by national funds through the FCT—Foundation for Science and Technology, I.P., within the scope of the project LA/P/0069/2020 granted to the Associate Laboratory ARNET, UIDB/04292/2020 granted to MARE—Marine and Environmental Sciences Centre.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Vitale G.A., Coppola D., Palma Esposito F., Buonocore C., Ausuri J., Tortorella E., de Pascale D. Antioxidant Molecules from Marine Fungi: Methodologies and Perspectives. Antioxidants. 2020;9:1183. doi: 10.3390/antiox9121183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rathod N.B., Elabed N., Punia S., Ozogul F., Kim S.-K., Rocha J.M. Recent Developments in Polyphenol Applications on Human Health: A Review with Current Knowledge. Plants. 2023;12:1217. doi: 10.3390/plants12061217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mateos R., Pérez-Correa J.R., Domínguez H. Bioactive Properties of Marine Phenolics. Mar. Drugs. 2020;18:501. doi: 10.3390/md18100501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Caruso G., Godos J., Privitera A., Lanza G., Castellano S., Chillemi A., Bruni O., Ferri R., Caraci F., Grosso G. Phenolic Acids and Prevention of Cognitive Decline: Polyphenols with a Neuroprotective Role in Cognitive Disorders and Alzheimer’s Disease. Nutrients. 2022;14:819. doi: 10.3390/nu14040819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dyshlovoy S.A. Recent Updates on Marine Cancer-Preventive Compounds. Mar. Drugs. 2021;19:558. doi: 10.3390/md19100558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang C., Jia J., Zhang P., Zheng W., Guo X., Ai C., Song S. Fucoidan from Laminaria Japonica Ameliorates Type 2 Diabetes Mellitus in Association with Modulation of Gut Microbiota and Metabolites in Streptozocin-Treated Mice. Foods. 2022;12:33. doi: 10.3390/foods12010033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Elbandy M. Anti-Inflammatory Effects of Marine Bioactive Compounds and Their Potential as Functional Food Ingredients in the Prevention and Treatment of Neuroinflammatory Disorders. Molecules. 2022;28:2. doi: 10.3390/molecules28010002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.El-Beltagi H.S., Mohamed A.A., Mohamed H.I., Ramadan K.M.A., Barqawi A.A., Mansour A.T. Phytochemical and Potential Properties of Seaweeds and Their Recent Applications: A Review. Mar. Drugs. 2022;20:342. doi: 10.3390/md20060342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Besednova N.N., Andryukov B.G., Zaporozhets T.S., Kryzhanovsky S.P., Fedyanina L.N., Kuznetsova T.A., Zvyagintseva T.N., Shchelkanov M.Y. Antiviral Effects of Polyphenols from Marine Algae. Biomedicines. 2021;9:200. doi: 10.3390/biomedicines9020200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vladkova T., Georgieva N., Staneva A., Gospodinova D. Recent Progress in Antioxidant Active Substances from Marine Biota. Antioxidants. 2022;11:439. doi: 10.3390/antiox11030439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karthikeyan A., Joseph A., Nair B.G. Promising Bioactive Compounds from the Marine Environment and Their Potential Effects on Various Diseases. J. Genet. Eng. Biotechnol. 2022;20:14. doi: 10.1186/s43141-021-00290-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gunathilake T., Akanbi T.O., Suleria H.A.R., Nalder T.D., Francis D.S., Barrow C.J. Seaweed Phenolics as Natural Antioxidants, Aquafeed Additives, Veterinary Treatments and Cross-Linkers for Microencapsulation. Mar. Drugs. 2022;20:445. doi: 10.3390/md20070445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lomartire S., Cotas J., Pacheco D., Marques J.C., Pereira L., Gonçalves A.M.M. Environmental Impact on Seaweed Phenolic Production and Activity: An Important Step for Compound Exploitation. Mar. Drugs. 2021;19:245. doi: 10.3390/md19050245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Peng J., Yuan J.-P., Wu C.-F., Wang J.-H. Fucoxanthin, a Marine Carotenoid Present in Brown Seaweeds and Diatoms: Metabolism and Bioactivities Relevant to Human Health. Mar. Drugs. 2011;9:1806–1828. doi: 10.3390/md9101806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zheng H., Zhao Y., Guo L. A Bioactive Substance Derived from Brown Seaweeds: Phlorotannins. Mar. Drugs. 2022;20:742. doi: 10.3390/md20120742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Usov A.I., Bilan M.I., Ustyuzhanina N.E., Nifantiev N.E. Fucoidans of Brown Algae: Comparison of Sulfated Polysaccharides from Fucus Vesiculosus and Ascophyllum Nodosum. Mar. Drugs. 2022;20:638. doi: 10.3390/md20100638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yoshie Y., Wang W., Petillo D., Suzuki T. Distribution of Catechins in Japanese Seaweeds. Fish. Sci. 2000;66:998–1000. doi: 10.1046/j.1444-2906.2000.00160.x. [DOI] [Google Scholar]
- 18.Besednova N.N., Andryukov B.G., Zaporozhets T.S., Kryzhanovsky S.P., Kuznetsova T.A., Fedyanina L.N., Makarenkova I.D., Zvyagintseva T.N. Algae Polyphenolic Compounds and Modern Antibacterial Strategies: Current Achievements and Immediate Prospects. Biomedicines. 2020;8:342. doi: 10.3390/biomedicines8090342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cotas J., Leandro A., Monteiro P., Pacheco D., Figueirinha A., Gonçalves A.M.M., da Silva G.J., Pereira L. Seaweed Phenolics: From Extraction to Applications. Mar. Drugs. 2020;18:384. doi: 10.3390/md18080384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Méresse S., Fodil M., Fleury F., Chénais B. Fucoxanthin, a Marine-Derived Carotenoid from Brown Seaweeds and Microalgae: A Promising Bioactive Compound for Cancer Therapy. Int. J. Mol. Sci. 2020;21:9273. doi: 10.3390/ijms21239273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jaworowska A., Murtaza A. Seaweed Derived Lipids Are a Potential Anti-Inflammatory Agent: A Review. Int. J. Environ. Res. Public Health. 2022;20:730. doi: 10.3390/ijerph20010730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kalasariya H.S., Pereira L. Dermo-Cosmetic Benefits of Marine Macroalgae-Derived Phenolic Compounds. Appl. Sci. 2022;12:11954. doi: 10.3390/app122311954. [DOI] [Google Scholar]
- 23.Gomes L., Monteiro P., Cotas J., Gonçalves A.M.M., Fernandes C., Gonçalves T., Pereira L. Seaweeds’ Pigments and Phenolic Compounds with Antimicrobial Potential. Biomol. Concepts. 2022;13:89–102. doi: 10.1515/bmc-2022-0003. [DOI] [PubMed] [Google Scholar]
- 24.Pereira D., Valentão P., Pereira J., Andrade P. Phenolics: From Chemistry to Biology. Molecules. 2009;14:2202–2211. doi: 10.3390/molecules14062202. [DOI] [Google Scholar]
- 25.Jesumani V., Du H., Aslam M., Pei P., Huang N. Potential Use of Seaweed Bioactive Compounds in Skincare—A Review. Mar. Drugs. 2019;17:688. doi: 10.3390/md17120688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kubanek J., Prusak A.C., Snell T.W., Giese R.A., Hardcastle K.I., Fairchild C.R., Aalbersberg W., Raventos-Suarez C., Hay M.E. Antineoplastic Diterpene−Benzoate Macrolides from the Fijian Red Alga Callophycus Serratus. Org. Lett. 2005;7:5261–5264. doi: 10.1021/ol052121f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cho M., Kang I.-J., Won M.-H., Lee H.-S., You S. The Antioxidant Properties of Ethanol Extracts and Their Solvent-Partitioned Fractions from Various Green Seaweeds. J. Med. Food. 2010;13:1232–1239. doi: 10.1089/jmf.2010.1124. [DOI] [PubMed] [Google Scholar]
- 28.Santos S.A.O., Félix R., Pais A.C.S., Rocha S.M., Silvestre A.J.D. The Quest for Phenolic Compounds from Macroalgae: A Review of Extraction and Identification Methodologies. Biomolecules. 2019;9:847. doi: 10.3390/biom9120847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lavoie S., Sweeney-Jones A.M., Mojib N., Dale B., Gagaring K., McNamara C.W., Quave C.L., Soapi K., Kubanek J. Antibacterial Oligomeric Polyphenols from the Green Alga Cladophora Socialis. J. Org. Chem. 2019;84:5035–5045. doi: 10.1021/acs.joc.8b03218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Olate-Gallegos C., Barriga A., Vergara C., Fredes C., García P., Giménez B., Robert P. Identification of Polyphenols from Chilean Brown Seaweeds Extracts by LC-DAD-ESI-MS/MS. J. Aquat. Food Prod. Technol. 2019;28:375–391. doi: 10.1080/10498850.2019.1594483. [DOI] [Google Scholar]
- 31.Ortiz J., Romero N., Robert P., Araya J., Lopez-Hernández J., Bozzo C., Navarrete E., Osorio A., Rios A. Dietary Fiber, Amino Acid, Fatty Acid and Tocopherol Contents of the Edible Seaweeds Ulva Lactuca and Durvillaea Antarctica. Food Chem. 2006;99:98–104. doi: 10.1016/j.foodchem.2005.07.027. [DOI] [Google Scholar]
- 32.Eo H., Jeon Y., Lee M., Lim Y. Brown Alga Ecklonia Cava Polyphenol Extract Ameliorates Hepatic Lipogenesis, Oxidative Stress, and Inflammation by Activation of AMPK and SIRT1 in High-Fat Diet-Induced Obese Mice. J. Agric. Food Chem. 2015;63:349–359. doi: 10.1021/jf502830b. [DOI] [PubMed] [Google Scholar]
- 33.Nho J.A., Shin Y.S., Jeong H.-R., Cho S., Heo H.J., Kim G.H., Kim D.-O. Neuroprotective Effects of Phlorotannin-Rich Extract from Brown Seaweed Ecklonia Cava on Neuronal PC-12 and SH-SY5Y Cells with Oxidative Stress. J. Microbiol. Biotechnol. 2020;30:359–367. doi: 10.4014/jmb.1910.10068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kim A.-R., Shin T.-S., Lee M.-S., Park J.-Y., Park K.-E., Yoon N.-Y., Kim J.-S., Choi J.-S., Jang B.-C., Byun D.-S., et al. Isolation and Identification of Phlorotannins from Ecklonia Stolonifera with Antioxidant and Anti-Inflammatory Properties. J. Agric. Food Chem. 2009;57:3483–3489. doi: 10.1021/jf900820x. [DOI] [PubMed] [Google Scholar]
- 35.Lee M.-S., Kwon M.-S., Choi J.-W., Shin T., No H.K., Choi J.-S., Byun D.-S., Kim J.-I., Kim H.-R. Anti-Inflammatory Activities of an Ethanol Extract of Ecklonia Stolonifera in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage Cells. J. Agric. Food Chem. 2012;60:9120–9129. doi: 10.1021/jf3022018. [DOI] [PubMed] [Google Scholar]
- 36.Nagayama K. Bactericidal Activity of Phlorotannins from the Brown Alga Ecklonia Kurome. J. Antimicrob. Chemother. 2002;50:889–893. doi: 10.1093/jac/dkf222. [DOI] [PubMed] [Google Scholar]
- 37.Lee S.-H., Eom S.-H., Yoon N.-Y., Kim M.-M., Li Y.-X., Ha S.K., Kim S.-K. Fucofuroeckol-A from Eisenia Bicyclis Inhibits Inflammation in Lipopolysaccharide-Induced Mouse Macrophages via Downregulation of the MAPK/NF-κ B Signaling Pathway. J. Chem. 2016;2016:6509212. doi: 10.1155/2016/6509212. [DOI] [Google Scholar]
- 38.Freitas R., Martins A., Silva J., Alves C., Pinteus S., Alves J., Teodoro F., Ribeiro H.M., Gonçalves L., Petrovski Ž., et al. Highlighting the Biological Potential of the Brown Seaweed Fucus Spiralis for Skin Applications. Antioxidants. 2020;9:611. doi: 10.3390/antiox9070611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pinteus S., Silva J., Alves C., Horta A., Thomas O., Pedrosa R. Antioxidant and Cytoprotective Activities of Fucus Spiralis Seaweed on a Human Cell In Vitro Model. Int. J. Mol. Sci. 2017;18:292. doi: 10.3390/ijms18020292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Obluchinskaya E.D., Pozharitskaya O.N., Zakharov D.V., Flisyuk E.V., Terninko I.I., Generalova Y.E., Shikov A.N. Biochemical Composition, Antiradical Potential and Human Health Risk of the Arctic Edible Brown Seaweed Fucus Spiralis L. J. Appl. Phycol. 2023;35:365–380. doi: 10.1007/s10811-022-02885-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Agregán R., Munekata P.E., Domínguez R., Carballo J., Franco D., Lorenzo J.M. Proximate Composition, Phenolic Content and In Vitro Antioxidant Activity of Aqueous Extracts of the Seaweeds Ascophyllum Nodosum, Bifurcaria Bifurcata and Fucus Vesiculosus. Effect of Addition of the Extracts on the Oxidative Stability of Canola Oil Unde. Food Res. Int. 2017;99:986–994. doi: 10.1016/j.foodres.2016.11.009. [DOI] [PubMed] [Google Scholar]
- 42.Obluchinskaya E.D., Pozharitskaya O.N., Zakharov D.V., Flisyuk E.V., Terninko I.I., Generalova Y.E., Smekhova I.E., Shikov A.N. The Biochemical Composition and Antioxidant Properties of Fucus Vesiculosus from the Arctic Region. Mar. Drugs. 2022;20:193. doi: 10.3390/md20030193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jégou C., Connan S., Bihannic I., Cérantola S., Guérard F., Stiger-Pouvreau V. Phlorotannin and Pigment Content of Native Canopy-Forming Sargassaceae Species Living in Intertidal Rockpools in Brittany (France): Any Relationship with Their Vertical Distribution and Phenology? Mar. Drugs. 2021;19:504. doi: 10.3390/md19090504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lopes G., Sousa C., Silva L.R., Pinto E., Andrade P.B., Bernardo J., Mouga T., Valentão P. Can Phlorotannins Purified Extracts Constitute a Novel Pharmacological Alternative for Microbial Infections with Associated Inflammatory Conditions? PLoS ONE. 2012;7:e31145. doi: 10.1371/journal.pone.0031145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yang F., Hyun J., Nagahawatta D.P., Kim Y.M., Heo M.-S., Jeon Y.-J. Cosmeceutical Effects of Ishige Okamurae Celluclast Extract. Antioxidants. 2022;11:2442. doi: 10.3390/antiox11122442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zou Y., Qian Z.-J., Li Y., Kim M.-M., Lee S.-H., Kim S.-K. Antioxidant Effects of Phlorotannins Isolated from Ishige Okamurae in Free Radical Mediated Oxidative Systems. J. Agric. Food Chem. 2008;56:7001–7009. doi: 10.1021/jf801133h. [DOI] [PubMed] [Google Scholar]
- 47.Rajaram R., Muralisankar T., Paray B.A., Al-Sadoon M.K. Phytochemical Profiling and Antioxidant Capacity of Kappaphycus Alvarezii (Doty) Doty Collected from Seaweed Farming Sites of Tropical Coastal Environment. Aquac. Res. 2021;52:3438–3448. doi: 10.1111/are.15188. [DOI] [Google Scholar]
- 48.Rajasulochana P., Krishnamoorthy P., Dhamotharan R. Identification of Bromophenol Compound and Antibacterial Activity of Kappaphycus sp. Int. J. Pharm. Bio. Sci. 2012;3:173–186. [Google Scholar]
- 49.Rudke A.R., da Silva M., Andrade C.J., de Vitali L., Ferreira S.R.S. Green Extraction of Phenolic Compounds and Carrageenan from the Red Alga Kappaphycus Alvarezii. Algal Res. 2022;67:102866. doi: 10.1016/j.algal.2022.102866. [DOI] [Google Scholar]
- 50.Heffernan N., Smyth T.J., Soler-Villa A., Fitzgerald R.J., Brunton N.P. Phenolic Content and Antioxidant Activity of Fractions Obtained from Selected Irish Macroalgae Species (Laminaria digitata, Fucus serratus, Gracilaria gracilis and Codium fragile) J. Appl. Phycol. 2015;27:519–530. doi: 10.1007/s10811-014-0291-9. [DOI] [Google Scholar]
- 51.D’Este M., Alvarado-Morales M., Ciofalo A., Angelidaki I. Macroalgae Laminaria digitata and Saccharina latissima as Potential Biomasses for Biogas and Total Phenolics Production: Focusing on Seasonal and Spatial Variations of the Algae. Energy Fuels. 2017;31:7166–7175. doi: 10.1021/acs.energyfuels.7b00853. [DOI] [Google Scholar]
- 52.Zúñiga A., Sáez C.A., Trabal A., Figueroa F.L., Pardo D., Navarrete C., Rodríguez-Rojas F., Moenne F., Celis-Plá P.S.M. Seasonal Photoacclimation and Vulnerability Patterns in the Brown Macroalga Lessonia Spicata (Ochrophyta) Water. 2020;13:6. doi: 10.3390/w13010006. [DOI] [Google Scholar]
- 53.Gómez I., Español S., Véliz K., Huovinen P. Spatial Distribution of Phlorotannins and Its Relationship with Photosynthetic UV Tolerance and Allocation of Storage Carbohydrates in Blades of the Kelp Lessonia Spicata. Mar. Biol. 2016;163:110. doi: 10.1007/s00227-016-2891-1. [DOI] [Google Scholar]
- 54.Leyton A., Pezoa-Conte R., Barriga A., Buschmann A.H., Mäki-Arvela P., Mikkola J.-P., Lienqueo M.E. Identification and Efficient Extraction Method of Phlorotannins from the Brown Seaweed Macrocystis pyrifera Using an Orthogonal Experimental Design. Algal Res. 2016;16:201–208. doi: 10.1016/j.algal.2016.03.019. [DOI] [Google Scholar]
- 55.Pérez-Alva A., Baigts-Allende D.K., Ramírez-Rodrigues M.A., Ramírez-Rodrigues M.M. Effect of Brown Seaweed (Macrocystis pyrifera) Addition on Nutritional and Quality Characteristics of Yellow, Blue, and Red Maize Tortillas. Foods. 2022;11:2627. doi: 10.3390/foods11172627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tenorio-Rodriguez P.A., Murillo-Álvarez J.I., Campa-Cordova Á.I., Angulo C. Antioxidant Screening and Phenolic Content of Ethanol Extracts of Selected Baja California Peninsula Macroalgae. J. Food Sci. Technol. 2017;54:422–429. doi: 10.1007/s13197-016-2478-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Hassan S., Hamed S., Almuhayawi M., Hozzein W., Selim S., AbdElgawad H. Bioactivity of Ellagic Acid and Velutin: Two Phenolic Compounds Isolated from Marine Algae. Egypt. J. Bot. 2020;61:219–231. doi: 10.21608/ejbo.2020.23778.1456. [DOI] [Google Scholar]
- 58.Soleimani S., Yousefzadi M., Nezhad S.B.M., Pozharitskaya O.N., Shikov A.N. Potential of the Ethyl Acetate Fraction of Padina Boergesenii as a Natural UV Filter in Sunscreen Cream Formulation. Life. 2023;13:239. doi: 10.3390/life13010239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Praba N., Sumaya S. Study on Phytochemical and Antioxidant Properties of Padina Gymnospora and Ulva Lactuca. Int. J. Life Sci. Pharma Res. 2022;12:155–160. doi: 10.22376/ijpbs/lpr.2022.12.6.L155-160. [DOI] [Google Scholar]
- 60.Rahman F., Abdul A., Ahsan T., Islam T., Alam M., Hossain M.N. Screening of Potential Bioactive Compounds from Padina Gymnospora Found in the Coast of St. Martin Island of Bangladesh. J. Mar. Biol. Aquac. 2021;6:1–7. doi: 10.15436/2381-0750.21.3010. [DOI] [Google Scholar]
- 61.Ko S.-C., Ding Y., Kim J., Ye B.-R., Kim E.-A., Jung W.-K., Heo S.-J., Lee S.-H. Bromophenol (5-Bromo-3,4-Dihydroxybenzaldehyde) Isolated from Red Alga Polysiphonia Morrowii Inhibits Adipogenesis by Regulating Expression of Adipogenic Transcription Factors and AMP-Activated Protein Kinase Activation in 3T3-L1 Adipocytes. Phytother. Res. 2019;33:737–744. doi: 10.1002/ptr.6266. [DOI] [PubMed] [Google Scholar]
- 62.Soleimani S., Yousefzadi M., Nezhad S.B.M., Pozharitskaya O.N., Shikov A.N. Evaluation of Fractions Extracted from Polycladia Myrica: Biological Activities, UVR Protective Effect, and Stability of Cream Formulation Based on It. J. Appl. Phycol. 2022;34:1763–1777. doi: 10.1007/s10811-022-02705-2. [DOI] [Google Scholar]
- 63.Li K., Li X.-M., Gloer J.B., Wang B.-G. Isolation, Characterization, and Antioxidant Activity of Bromophenols of the Marine Red Alga Rhodomela Confervoides. J. Agric. Food Chem. 2011;59:9916–9921. doi: 10.1021/jf2022447. [DOI] [PubMed] [Google Scholar]
- 64.Lijun H., Nianjun X., Jiangong S., Xiaojun Y., Chengkui Z. Isolation and Pharmacological Activities of Bromophenols FromRhodomela Confervoides. Chin. J. Oceanol. Limnol. 2005;23:226–229. doi: 10.1007/BF02894243. [DOI] [Google Scholar]
- 65.Wang B.-G., Zhang W.-W., Duan X.-J., Li X.-M. In Vitro Antioxidative Activities of Extract and Semi-Purified Fractions of the Marine Red Alga, Rhodomela Confervoides (Rhodomelaceae) Food Chem. 2009;113:1101–1105. doi: 10.1016/j.foodchem.2008.08.078. [DOI] [Google Scholar]
- 66.Puspita M., Déniel M., Widowati I., Radjasa O.K., Douzenel P., Marty C., Vandanjon L., Bedoux G., Bourgougnon N. Total Phenolic Content and Biological Activities of Enzymatic Extracts from Sargassum Muticum (Yendo) Fensholt. J. Appl. Phycol. 2017;29:2521–2537. doi: 10.1007/s10811-017-1086-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tanniou A., Vandanjon L., Incera M., Serrano Leon E., Husa V., Grand J.L., Nicolas J.-L., Poupart N., Kervarec N., Engelen A., et al. Assessment of the Spatial Variability of Phenolic Contents and Associated Bioactivities in the Invasive Alga Sargassum Muticum Sampled along Its European Range from Norway to Portugal. J. Appl. Phycol. 2013;26:1215–1230. doi: 10.1007/s10811-013-0198-x. [DOI] [Google Scholar]
- 68.Afnan, Saleem A., Akhtar M.F., Sharif A., Akhtar B., Siddique R., Ashraf G.M., Alghamdi B.S., Alharthy S.A. Anticancer, Cardio-Protective and Anti-Inflammatory Potential of Natural-Sources-Derived Phenolic Acids. Molecules. 2022;27:7286. doi: 10.3390/molecules27217286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chouh A., Nouadri T., Catarino M.D., Silva A.M.S., Cardoso S.M. Phlorotannins of the Brown Algae Sargassum Vulgare from the Mediterranean Sea Coast. Antioxidants. 2022;11:1055. doi: 10.3390/antiox11061055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chen B., Chen H., Qu H., Qiao K., Xu M., Wu J., Su Y., Shi Y., Liu Z., Wang Q. Photoprotective Effects of Sargassum Thunbergii on Ultraviolet B-Induced Mouse L929 Fibroblasts and Zebrafish. BMC Complement. Med. Ther. 2022;22:144. doi: 10.1186/s12906-022-03609-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Heo S.J., Lee G.W., Song C.B., Jeon Y.J. Antioxidant Activity of Enzymatic Extracts from Brown Seaweeds. ALGAE. 2003;18:71–81. doi: 10.4490/ALGAE.2003.18.1.071. [DOI] [Google Scholar]
- 72.Park J.-S., Han J.-M., Surendhiran D., Chun B.-S. Physicochemical and Biofunctional Properties of Sargassum Thunbergii Extracts Obtained from Subcritical Water Extraction and Conventional Solvent Extraction. J. Supercrit. Fluids. 2022;182:105535. doi: 10.1016/j.supflu.2022.105535. [DOI] [Google Scholar]
- 73.Duan X.-J., Li X.-M., Wang B.-G. Highly Brominated Mono- and Bis-Phenols from the Marine Red Alga Symphyocladia latiuscula with Radical-Scavenging Activity. J. Nat. Prod. 2007;70:1210–1213. doi: 10.1021/np070061b. [DOI] [PubMed] [Google Scholar]
- 74.Samaddar S., Koneri R. Polyphenols of Marine Red Macroalga Symphyocladia latiuscula Ameliorate Diabetic Peripheral Neuropathy in Experimental Animals. Heliyon. 2019;5:e01781. doi: 10.1016/j.heliyon.2019.e01781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhang W.-W., Duan X.-J., Huang H.-L., Zhang Y., Wang B.-G. Evaluation of 28 Marine Algae from the Qingdao Coast for Antioxidative Capacity and Determination of Antioxidant Efficiency and Total Phenolic Content of Fractions and Subfractions Derived from Symphyocladia latiuscula (Rhodomelaceae) J. Appl. Phycol. 2007;19:97–108. doi: 10.1007/s10811-006-9115-x. [DOI] [Google Scholar]
- 76.Ishii T., Okino T., Suzuki M., Machiguchi Y. Tichocarpols A and B, Two Novel Phenylpropanoids with Feeding-Deterrent Activity from the Red Alga Tichocarpus c Rinitus. J. Nat. Prod. 2004;67:1764–1766. doi: 10.1021/np0498509. [DOI] [PubMed] [Google Scholar]
- 77.Jimenez-Lopez C., Pereira A.G., Lourenço-Lopes C., Garcia-Oliveira P., Cassani L., Fraga-Corral M., Prieto M.A., Simal-Gandara J. Main Bioactive Phenolic Compounds in Marine Algae and Their Mechanisms of Action Supporting Potential Health Benefits. Food Chem. 2021;341:128262. doi: 10.1016/j.foodchem.2020.128262. [DOI] [PubMed] [Google Scholar]
- 78.Lee J., Lee T.-K., Kang R.-S., Shin H., Lee H. The In Vitro Antioxidant Activities of the Bromophenols from the Red Alga Tichocarpus Crinitus and Phenolic Derivatives. J. Korean Magn. Reson. Soc. 2007;11:56–63. [Google Scholar]
- 79.Trentin R., Custódio L., Rodrigues M.J., Moschin E., Sciuto K., da Silva J.P., Moro I. Exploring Ulva Australis Areschoug for Possible Biotechnological Applications: In Vitro Antioxidant and Enzymatic Inhibitory Properties, and Fatty Acids Contents. Algal Res. 2020;50:101980. doi: 10.1016/j.algal.2020.101980. [DOI] [Google Scholar]
- 80.Corral-Rosales C., Ricque-Marie D., Cruz-Suárez L.E., Arjona O., Palacios E. Fatty Acids, Sterols, Phenolic Compounds, and Carotenoid Changes in Response to Dietary Inclusion of Ulva Clathrata in Shrimp Litopenaeus Vannamei Broodstock. J. Appl. Phycol. 2019;31:4009–4020. doi: 10.1007/s10811-019-01829-2. [DOI] [Google Scholar]
- 81.Farasat M., Khavari-Nejad R.-A., Nabavi S.M.B., Namjooyan F. Antioxidant Activity, Total Phenolics and Flavonoid Contents of Some Edible Green Seaweeds from Northern Coasts of the Persian Gulf. Iran. J. Pharm. Res. 2014;13:163–170. [PMC free article] [PubMed] [Google Scholar]
- 82.Hong J., Huang P., Sun B., Mi J., Zhang J., Li C., Yang W. Protective Effects of Ulva Clathrata Derived Polyphenols against Oxidative Stress in Lipopolysaccharide-Induced RAW 264.7 Cells via Nrf2/HO-1 Pathway. J. Appl. Phycol. 2022;34:2657–2669. doi: 10.1007/s10811-022-02789-w. [DOI] [Google Scholar]
- 83.Fernandes H., Salgado J.M., Martins N., Peres H., Oliva-Teles A., Belo I. Sequential Bioprocessing of Ulva Rigida to Produce Lignocellulolytic Enzymes and to Improve Its Nutritional Value as Aquaculture Feed. Bioresour. Technol. 2019;281:277–285. doi: 10.1016/j.biortech.2019.02.068. [DOI] [PubMed] [Google Scholar]
- 84.Shanab S.M.M., Shalaby E.A., El-Fayoumy E.A. Enteromorpha Compressa Exhibits Potent Antioxidant Activity. J. Biomed. Biotechnol. 2011;2011:726405. doi: 10.1155/2011/726405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sharmila S., Rebecca L.J. Phytochemical Analysis of Enteromorpha Flexuosa and Ulva Lactuca: A Comparative Study. Int. J. Pharm. Bio Sci. 2014;5:830–834. [Google Scholar]
- 86.Sasikala C., Ramani D.G. Comparative Study on Antimicrobial Activity of Seaweeds. Asian J. Pharm. Clin. Res. 2017;10:384. doi: 10.22159/ajpcr.2017.v10i12.21002. [DOI] [Google Scholar]
- 87.Kosanić M., Ranković B., Stanojković T. Biological Activities of Two Macroalgae from Adriatic Coast of Montenegro. Saudi J. Biol. Sci. 2015;22:390–397. doi: 10.1016/j.sjbs.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Srikong W., Bovornreungroj N., Mittraparparthorn P., Bovornreungroj P. Antibacterial and Antioxidant Activities of Differential Solvent Extractions from the Green Seaweed Ulva Intestinalis. ScienceAsia. 2017;43:88. doi: 10.2306/scienceasia1513-1874.2017.43.088. [DOI] [Google Scholar]
- 89.Wekre M.E., Kåsin K., Underhaug J., Holmelid B., Jordheim M. Quantification of Polyphenols in Seaweeds: A Case Study of Ulva Intestinalis. Antioxidants. 2019;8:612. doi: 10.3390/antiox8120612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kim J.-H., Kim S.A., Edwards M.S., Lee I.-A. Anti-Inflammatory Effects of Polyphenol Extracts from Ulva Linza (Ulvophyceae, Chlorophyta) Toxicol. Environ. Health Sci. 2018;10:212–219. doi: 10.1007/s13530-018-0366-0. [DOI] [Google Scholar]
- 91.Mezghani S., Csupor D., Bourguiba I., Hohmann J., Amri M., Bouaziz M. Characterization of Phenolic Compounds of Ulva Rigida (Chlorophycae) and Its Antioxidant Activity. Eur. J. Med. Plants. 2016;12:1–9. doi: 10.9734/EJMP/2016/22935. [DOI] [Google Scholar]
- 92.Trigui M., Gasmi L., Zouari I., Tounsi S. Seasonal Variation in Phenolic Composition, Antibacterial and Antioxidant Activities of Ulva Rigida (Chlorophyta) and Assessment of Antiacetylcholinesterase Potential. J. Appl. Phycol. 2013;25:319–328. doi: 10.1007/s10811-012-9866-5. [DOI] [Google Scholar]
- 93.Popplewell W.L., Northcote P.T. Colensolide A: A New Nitrogenous Bromophenol from the New Zealand Marine Red Alga Osmundaria Colensoi. Tetrahedron Lett. 2009;50:6814–6817. doi: 10.1016/j.tetlet.2009.09.118. [DOI] [Google Scholar]
- 94.Ashraf S.A., Adnan M., Patel M., Siddiqui A.J., Sachidanandan M., Snoussi M., Hadi S. Fish-Based Bioactives as Potent Nutraceuticals: Exploring the Therapeutic Perspective of Sustainable Food from the Sea. Mar. Drugs. 2020;18:265. doi: 10.3390/md18050265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Méndez L., Medina I. Polyphenols and Fish Oils for Improving Metabolic Health: A Revision of the Recent Evidence for Their Combined Nutraceutical Effects. Molecules. 2021;26:2438. doi: 10.3390/molecules26092438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Gutiérrez-del-Río I., López-Ibáñez S., Magadán-Corpas P., Fernández-Calleja L., Pérez-Valero Á., Tuñón-Granda M., Miguélez E.M., Villar C.J., Lombó F. Terpenoids and Polyphenols as Natural Antioxidant Agents in Food Preservation. Antioxidants. 2021;10:1264. doi: 10.3390/antiox10081264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Saccol E.M.H., Toni C., Pês T.S., Ourique G.M., Gressler L.T., Silva L.V.F., Mourão R.H.V., Oliveira R.B., Baldisserotto B., Pavanato M.A. Anaesthetic and Antioxidant Effects of Myrcia Sylvatica (G. Mey.) DC. and Curcuma Longa L. Essential Oils on Tambaqui (Colossoma Macropomum) Aquac. Res. 2017;48:2012–2031. doi: 10.1111/are.13034. [DOI] [Google Scholar]
- 98.Kocaadam B., Şanlier N. Curcumin, an Active Component of Turmeric (Curcuma Longa), and Its Effects on Health. Crit. Rev. Food Sci. Nutr. 2017;57:2889–2895. doi: 10.1080/10408398.2015.1077195. [DOI] [PubMed] [Google Scholar]
- 99.Peng Y., Ao M., Dong B., Jiang Y., Yu L., Chen Z., Hu C., Xu R. Anti-Inflammatory Effects of Curcumin in the Inflammatory Diseases: Status, Limitations and Countermeasures. Drug Des. Dev. Ther. 2021;15:4503–4525. doi: 10.2147/DDDT.S327378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cox F.F., Misiou A., Vierkant A., Ale-Agha N., Grandoch M., Haendeler J., Altschmied J. Protective Effects of Curcumin in Cardiovascular Diseases—Impact on Oxidative Stress and Mitochondria. Cells. 2022;11:342. doi: 10.3390/cells11030342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Cas M.D., Ghidoni R. Dietary Curcumin: Correlation between Bioavailability and Health Potential. Nutrients. 2019;11:2147. doi: 10.3390/nu11092147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Aubourg S.P. Enhancement of Lipid Stability and Acceptability of Canned Seafood by Addition of Natural Antioxidant Compounds to the Packing Medium—A Review. Antioxidants. 2023;12:245. doi: 10.3390/antiox12020245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lobo V., Patil A., Phatak A., Chandra N. Free Radicals, Antioxidants and Functional Foods: Impact on Human Health. Pharmacogn. Rev. 2010;4:118. doi: 10.4103/0973-7847.70902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Kim J.M., Heo H.J. The Roles of Catechins in Regulation of Systemic Inflammation. Food Sci. Biotechnol. 2022;31:957–970. doi: 10.1007/s10068-022-01069-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Jiang Y., Jiang Z., Ma L., Huang Q. Advances in Nanodelivery of Green Tea Catechins to Enhance the Anticancer Activity. Molecules. 2021;26:3301. doi: 10.3390/molecules26113301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ambroselli D., Masciulli F., Romano E., Catanzaro G., Besharat Z.M., Massari M.C., Ferretti E., Migliaccio S., Izzo L., Ritieni A., et al. New Advances in Metabolic Syndrome, from Prevention to Treatment: The Role of Diet and Food. Nutrients. 2023;15:640. doi: 10.3390/nu15030640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Bae J., Kim N., Shin Y., Kim S.-Y., Kim Y.-J. Activity of Catechins and Their Applications. Biomed. Dermatol. 2020;4:8. doi: 10.1186/s41702-020-0057-8. [DOI] [Google Scholar]
- 108.D’Andrea G. Quercetin: A Flavonol with Multifaceted Therapeutic Applications? Fitoterapia. 2015;106:256–271. doi: 10.1016/j.fitote.2015.09.018. [DOI] [PubMed] [Google Scholar]
- 109.Vrânceanu M., Galimberti D., Banc R., Dragoş O., Cozma-Petruţ A., Hegheş S.-C., Voştinaru O., Cuciureanu M., Stroia C.M., Miere D., et al. The Anticancer Potential of Plant-Derived Nutraceuticals via the Modulation of Gene Expression. Plants. 2022;11:2524. doi: 10.3390/plants11192524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Saeedi-Boroujeni A., Mahmoudian-Sani M.-R. Anti-Inflammatory Potential of Quercetin in COVID-19 Treatment. J. Inflamm. 2021;18:3. doi: 10.1186/s12950-021-00268-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Carrasco-Pozo C., Tan K.N., Reyes-Farias M., De La Jara N., Ngo S.T., Garcia-Diaz D.F., Llanos P., Cires M.J., Borges K. The Deleterious Effect of Cholesterol and Protection by Quercetin on Mitochondrial Bioenergetics of Pancreatic β-Cells, Glycemic Control and Inflammation: In Vitro and In Vivo Studies. Redox Biol. 2016;9:229–243. doi: 10.1016/j.redox.2016.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yang D., Wang T., Long M., Li P. Quercetin: Its Main Pharmacological Activity and Potential Application in Clinical Medicine. Oxidative Med. Cell. Longev. 2020;2020:8825387. doi: 10.1155/2020/8825387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kaczmarek-Szczepańska B., Grabska-Zielińska S., Michalska-Sionkowska M. The Application of Phenolic Acids in The Obtainment of Packaging Materials Based on Polymers—A Review. Foods. 2023;12:1343. doi: 10.3390/foods12061343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Ceci C., Lacal P., Tentori L., De Martino M., Miano R., Graziani G. Experimental Evidence of the Antitumor, Antimetastatic and Antiangiogenic Activity of Ellagic Acid. Nutrients. 2018;10:1756. doi: 10.3390/nu10111756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Sharifi-Rad J., Quispe C., Castillo C.M.S., Caroca R., Lazo-Vélez M.A., Antonyak H., Polishchuk A., Lysiuk R., Oliinyk P., De Masi L., et al. Ellagic Acid: A Review on Its Natural Sources, Chemical Stability, and Therapeutic Potential. Oxidative Med. Cell. Longev. 2022;2022:3848084. doi: 10.1155/2022/3848084. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 116.Aqilah N.M.N., Rovina K., Felicia W.X.L., Vonnie J.M. A Review on the Potential Bioactive Components in Fruits and Vegetable Wastes as Value-Added Products in the Food Industry. Molecules. 2023;28:2631. doi: 10.3390/molecules28062631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Alfei S., Marengo B., Zuccari G. Oxidative Stress, Antioxidant Capabilities, and Bioavailability: Ellagic Acid or Urolithins? Antioxidants. 2020;9:707. doi: 10.3390/antiox9080707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Majdan M., Bobrowska-Korczak B. Active Compounds in Fruits and Inflammation in the Body. Nutrients. 2022;14:2496. doi: 10.3390/nu14122496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Hassan S.S.U., Samanta S., Dash R., Karpiński T.M., Habibi E., Sadiq A., Ahmadi A., Bungau S. The Neuroprotective Effects of Fisetin, a Natural Flavonoid in Neurodegenerative Diseases: Focus on the Role of Oxidative Stress. Front. Pharmacol. 2022;13:1015835. doi: 10.3389/fphar.2022.1015835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Mucha P., Skoczyńska A., Małecka M., Hikisz P., Budzisz E. Overview of the Antioxidant and Anti-Inflammatory Activities of Selected Plant Compounds and Their Metal Ions Complexes. Molecules. 2021;26:4886. doi: 10.3390/molecules26164886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kozłowska A., Szostak-Węgierek D. Targeting Cardiovascular Diseases by Flavonols: An Update. Nutrients. 2022;14:1439. doi: 10.3390/nu14071439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kumar R.M., Kumar H., Bhatt T., Jain R., Panchal K., Chaurasiya A., Jain V. Fisetin in Cancer: Attributes, Developmental Aspects, and Nanotherapeutics. Pharmaceuticals. 2023;16:196. doi: 10.3390/ph16020196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Maher P. Preventing and Treating Neurological Disorders with the Flavonol Fisetin. Brain Plast. 2021;6:155–166. doi: 10.3233/BPL-200104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Nag M., Lahiri D., Dey A., Sarkar T., Pati S., Joshi S., Bunawan H., Mohammed A., Edinur H.A., Ghosh S., et al. Seafood Discards: A Potent Source of Enzymes and Biomacromolecules with Nutritional and Nutraceutical Significance. Front. Nutr. 2022;9:879929. doi: 10.3389/fnut.2022.879929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zhang Z., Li X., Sang S., McClements D.J., Chen L., Long J., Jiao A., Jin Z., Qiu C. Polyphenols as Plant-Based Nutraceuticals: Health Effects, Encapsulation, Nano-Delivery, and Application. Foods. 2022;11:2189. doi: 10.3390/foods11152189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Miyashita K., Nishikawa S., Beppu F., Tsukui T., Abe M., Hosokawa M. The Allenic Carotenoid Fucoxanthin, a Novel Marine Nutraceutical from Brown Seaweeds. J. Sci. Food Agric. 2011;91:1166–1174. doi: 10.1002/jsfa.4353. [DOI] [PubMed] [Google Scholar]
- 127.Costa M., Coello L., Urbatzka R., Pérez M., Thorsteinsdottir M. New Aromatic Bisabolane Derivatives with Lipid-Reducing Activity from the Marine Sponge Myrmekioderma sp. Mar. Drugs. 2019;17:375. doi: 10.3390/md17060375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Cichewicz R.H., Clifford L.J., Lassen P.R., Cao X., Freedman T.B., Nafie L.A., Deschamps J.D., Kenyon V.A., Flanary J.R., Holman T.R., et al. Stereochemical Determination and Bioactivity Assessment of (S)-(+)-Curcuphenol Dimers Isolated from the Marine Sponge Didiscus Aceratus and Synthesized through Laccase Biocatalysis. Bioorg. Med. Chem. 2005;13:5600–5612. doi: 10.1016/j.bmc.2005.06.020. [DOI] [PubMed] [Google Scholar]
- 129.Wang S., Li X.-M., Teuscher F., Li D.-L., Diesel A., Ebel R., Proksch P., Wang B.-G. Chaetopyranin, a Benzaldehyde Derivative, and Other Related Metabolites from Chaetomium Globosum, an Endophytic Fungus Derived from the Marine Red Alga Polysiphonia Urceolata. J. Nat. Prod. 2006;69:1622–1625. doi: 10.1021/np060248n. [DOI] [PubMed] [Google Scholar]
- 130.Powell C., Hughes A.D., Kelly M.S., Conner S., McDougall G.J. Extraction and Identification of Antioxidant Polyhydroxynaphthoquinone Pigments from the Sea Urchin, Psammechinus Miliaris. LWT—Food Sci. Technol. 2014;59:455–460. doi: 10.1016/j.lwt.2014.05.016. [DOI] [Google Scholar]
- 131.Kuwahara R., Hatate H., Chikami A., Murata H., Kijidani Y. Quantitative Separation of Antioxidant Pigments in Purple Sea Urchin Shells Using a Reversed-Phase High Performance Liquid Chromatography. LWT—Food Sci. Technol. 2010;43:1185–1190. doi: 10.1016/j.lwt.2010.03.005. [DOI] [Google Scholar]
- 132.Kuwahara R., Hatate H., Yuki T., Murata H., Tanaka R., Hama Y. Antioxidant Property of Polyhydroxylated Naphthoquinone Pigments from Shells of Purple Sea Urchin Anthocidaris Crassispina. LWT—Food Sci. Technol. 2009;42:1296–1300. doi: 10.1016/j.lwt.2009.02.020. [DOI] [Google Scholar]
- 133.Shikov A.N., Pozharitskaya O.N., Krishtopina A.S., Makarov V.G. Naphthoquinone Pigments from Sea Urchins: Chemistry and Pharmacology. Phytochem. Rev. 2018;17:509–534. doi: 10.1007/s11101-018-9547-3. [DOI] [Google Scholar]
- 134.Carreto J.I., Carignan M.O. Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects. Mar. Drugs. 2011;9:387–446. doi: 10.3390/md9030387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Cotas J., Figueirinha A., Pereira L., Batista T. The Effect of Salinity on Fucus Ceranoides (Ochrophyta, Phaeophyceae) in the Mondego River (Portugal) J. Oceanol. Limnol. 2019;37:881–891. doi: 10.1007/s00343-019-8111-3. [DOI] [Google Scholar]
- 136.García-Poza S., Leandro A., Cotas C., Cotas J., Marques J.C., Pereira L., Gonçalves A.M.M. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. Int. J. Environ. Res. Public Health. 2020;17:6528. doi: 10.3390/ijerph17186528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Whitfield F.B., Helidoniotis F., Shaw K.J., Svoronos D. Distribution of Bromophenols in Species of Marine Algae from Eastern Australia. J. Agric. Food Chem. 1999;47:2367–2373. doi: 10.1021/jf981080h. [DOI] [PubMed] [Google Scholar]
- 138.Matulja D., Vranješević F., Markovic M.K., Pavelić S.K., Marković D. Anticancer Activities of Marine-Derived Phenolic Compounds and Their Derivatives. Molecules. 2022;27:1449. doi: 10.3390/molecules27041449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Soares S., Brandão E., Guerreiro C., Soares S., Mateus N., de Freitas V. Tannins in Food: Insights into the Molecular Perception of Astringency and Bitter Taste. Molecules. 2020;25:2590. doi: 10.3390/molecules25112590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Saeed N., Khan M.R., Shabbir M. Antioxidant Activity, Total Phenolic and Total Flavonoid Contents of Whole Plant Extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012;12:221. doi: 10.1186/1472-6882-12-221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Bustos-Salgado P., Andrade-Carrera B., Domínguez-Villegas V., Díaz-Garrido N., Rodríguez-Lagunas M.J., Badía J., Baldomà L., Mallandrich M., Calpena-Campmany A., Garduño-Ramírez M.L. Screening Anti-Inflammatory Effects of Flavanones Solutions. Int. J. Mol. Sci. 2021;22:8878. doi: 10.3390/ijms22168878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Baran A., Kwiatkowska A., Potocki L. Antibiotics and Bacterial Resistance—A Short Story of an Endless Arms Race. Int. J. Mol. Sci. 2023;24:5777. doi: 10.3390/ijms24065777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Azman M., Sabri A.H., Anjani Q.K., Mustaffa M.F., Hamid K.A. Intestinal Absorption Study: Challenges and Absorption Enhancement Strategies in Improving Oral Drug Delivery. Pharmaceuticals. 2022;15:975. doi: 10.3390/ph15080975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Catarino M.D., Pires S.M.G., Silva S., Costa F., Braga S.S., Pinto D.C.G.A., Silva A.M.S., Cardoso S.M. Overview of Phlorotannins’ Constituents in Fucales. Mar. Drugs. 2022;20:754. doi: 10.3390/md20120754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Kumar L.R.G., Paul P.T., Anas K.K., Tejpal C.S., Chatterjee N.S., Anupama T.K., Mathew S., Ravishankar C.N. Phlorotannins–Bioactivity and Extraction Perspectives. J. Appl. Phycol. 2022;34:2173–2185. doi: 10.1007/s10811-022-02749-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Pradhan B., Nayak R., Bhuyan P.P., Patra S., Behera C., Sahoo S., Ki J.-S., Quarta A., Ragusa A., Jena M. Algal Phlorotannins as Novel Antibacterial Agents with Reference to the Antioxidant Modulation: Current Advances and Future Directions. Mar. Drugs. 2022;20:403. doi: 10.3390/md20060403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Heim K.E., Tagliaferro A.R., Bobilya D.J. Flavonoid Antioxidants: Chemistry, Metabolism and Structure-Activity Relationships. J. Nutr. Biochem. 2002;13:572–584. doi: 10.1016/S0955-2863(02)00208-5. [DOI] [PubMed] [Google Scholar]
- 148.Bernatoniene J., Kopustinskiene D. The Role of Catechins in Cellular Responses to Oxidative Stress. Molecules. 2018;23:965. doi: 10.3390/molecules23040965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wen L., Wu D., Tan X., Zhong M., Xing J., Li W., Li D., Cao F. The Role of Catechins in Regulating Diabetes: An Update Review. Nutrients. 2022;14:4681. doi: 10.3390/nu14214681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Trisha A.T., Shakil M.H., Talukdar S., Rovina K., Huda N., Zzaman W. Tea Polyphenols and Their Preventive Measures against Cancer: Current Trends and Directions. Foods. 2022;11:3349. doi: 10.3390/foods11213349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Mandel S.A., Amit T., Kalfon L., Reznichenko L., Youdim M. Targeting Multiple Neurodegenerative Diseases Etiologies with Multimodal-Acting Green Tea Catechins. J. Nutr. 2008;138:1578S–1583S. doi: 10.1093/jn/138.8.1578S. [DOI] [PubMed] [Google Scholar]
- 152.Chen X.-Q., Hu T., Han Y., Huang W., Yuan H.-B., Zhang Y.-T., Du Y., Jiang Y.-W. Preventive Effects of Catechins on Cardiovascular Disease. Molecules. 2016;21:1759. doi: 10.3390/molecules21121759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Dong H., Dong S., Erik Hansen P., Stagos D., Lin X., Liu M. Progress of Bromophenols in Marine Algae from 2011 to 2020: Structure, Bioactivities, and Applications. Mar. Drugs. 2020;18:411. doi: 10.3390/md18080411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Meulmeester F.L., Luo J., Martens L.G., Mills K., van Heemst D., Noordam R. Antioxidant Supplementation in Oxidative Stress-Related Diseases: What Have We Learned from Studies on Alpha-Tocopherol? Antioxidants. 2022;11:2322. doi: 10.3390/antiox11122322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Barbalace M.C., Malaguti M., Giusti L., Lucacchini A., Hrelia S., Angeloni C. Anti-Inflammatory Activities of Marine Algae in Neurodegenerative Diseases. Int. J. Mol. Sci. 2019;20:3061. doi: 10.3390/ijms20123061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Liu M., Hansen P.E., Lin X. Bromophenols in Marine Algae and Their Bioactivities. Mar. Drugs. 2011;9:1273–1292. doi: 10.3390/md9071273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Pangestuti R., Kim S.-K. Neuroprotective Effects of Marine Algae. Mar. Drugs. 2011;9:803–818. doi: 10.3390/md9050803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Gómez-Guzmán M., Rodríguez-Nogales A., Algieri F., Gálvez J. Potential Role of Seaweed Polyphenols in Cardiovascular-Associated Disorders. Mar. Drugs. 2018;16:250. doi: 10.3390/md16080250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Al-Ishaq R.K., Abotaleb M., Kubatka P., Kajo K., Büsselberg D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules. 2019;9:430. doi: 10.3390/biom9090430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Panche A.N., Diwan A.D., Chandra S.R. Flavonoids: An Overview. J. Nutr. Sci. 2016;5:e47. doi: 10.1017/jns.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Ginwala R., Bhavsar R., Chigbu D.I., Jain P., Khan Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants. 2019;8:35. doi: 10.3390/antiox8020035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kopustinskiene D.M., Jakstas V., Savickas A., Bernatoniene J. Flavonoids as Anticancer Agents. Nutrients. 2020;12:457. doi: 10.3390/nu12020457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Evans J.A., Mendonca P., Soliman K.F.A. Neuroprotective Effects and Therapeutic Potential of the Citrus Flavonoid Hesperetin in Neurodegenerative Diseases. Nutrients. 2022;14:2228. doi: 10.3390/nu14112228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Ciumărnean L., Milaciu M.V., Runcan O., Vesa Ștefan C., Răchișan A.L., Negrean V., Perné M.-G., Donca V.I., Alexescu T.-G., Para I., et al. The Effects of Flavonoids in Cardiovascular Diseases. Molecules. 2020;25:4320. doi: 10.3390/molecules25184320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Deepika, Maurya P.K. Health Benefits of Quercetin in Age-Related Diseases. Molecules. 2022;27:2498. doi: 10.3390/molecules27082498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mokra D., Joskova M., Mokry J. Therapeutic Effects of Green Tea Polyphenol (−)-Epigallocatechin-3-Gallate (EGCG) in Relation to Molecular Pathways Controlling Inflammation, Oxidative Stress, and Apoptosis. Int. J. Mol. Sci. 2022;24:340. doi: 10.3390/ijms24010340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Parhiz H., Roohbakhsh A., Soltani F., Rezaee R., Iranshahi M. Antioxidant and Anti-Inflammatory Properties of the Citrus Flavonoids Hesperidin and Hesperetin: An Updated Review of Their Molecular Mechanisms and Experimental Models. Phytother. Res. 2015;29:323–331. doi: 10.1002/ptr.5256. [DOI] [PubMed] [Google Scholar]
- 168.Calderon-Montaño J.M., Burgos-Morón E., Perez-Guerrero C., Lopez-Lazaro M. A Review on the Dietary Flavonoid Kaempferol. Mini-Rev. Med. Chem. 2011;11:298–344. doi: 10.2174/138955711795305335. [DOI] [PubMed] [Google Scholar]
- 169.Ponnampalam E.N., Kiani A., Santhiravel S., Holman B.W.B., Lauridsen C., Dunshea F.R. The Importance of Dietary Antioxidants on Oxidative Stress, Meat and Milk Production, and Their Preservative Aspects in Farm Animals: Antioxidant Action, Animal Health, and Product Quality—Invited Review. Animals. 2022;12:3279. doi: 10.3390/ani12233279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Masyita A., Sari R.M., Astuti A.D., Yasir B., Rumata N.R., Emran T.B., Nainu F., Simal-Gandara J. Terpenes and Terpenoids as Main Bioactive Compounds of Essential Oils, Their Roles in Human Health and Potential Application as Natural Food Preservatives. Food Chem. X. 2022;13:100217. doi: 10.1016/j.fochx.2022.100217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Esmeeta A., Adhikary S., Dharshnaa V., Swarnamughi P., Maqsummiya Z.U., Banerjee A., Pathak S., Duttaroy A.K. Plant-Derived Bioactive Compounds in Colon Cancer Treatment: An Updated Review. Biomed. Pharmacother. 2022;153:113384. doi: 10.1016/j.biopha.2022.113384. [DOI] [PubMed] [Google Scholar]
- 172.Ghani M.A.A., Ugusman A., Latip J., Zainalabidin S. Role of Terpenophenolics in Modulating Inflammation and Apoptosis in Cardiovascular Diseases: A Review. Int. J. Mol. Sci. 2023;24:5339. doi: 10.3390/ijms24065339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Mahizan N.A., Yang S.-K., Moo C.-L., Song A.A.-L., Chong C.-M., Chong C.-W., Abushelaibi A., Lim S.-H.E., Lai K.-S. Terpene Derivatives as a Potential Agent against Antimicrobial Resistance (AMR) Pathogens. Molecules. 2019;24:2631. doi: 10.3390/molecules24142631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Montenegro Z.J.S., Álvarez-Rivera G., Sánchez-Martínez J.D., Gallego R., Valdés A., Bueno M., Cifuentes A., Ibáñez E. Neuroprotective Effect of Terpenoids Recovered from Olive Oil By-Products. Foods. 2021;10:1507. doi: 10.3390/foods10071507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Noor S., Mohammad T., Rub M.A., Raza A., Azum N., Yadav D.K., Hassan M.I., Asiri A.M. Biomedical Features and Therapeutic Potential of Rosmarinic Acid. Arch. Pharmacal Res. 2022;45:205–228. doi: 10.1007/s12272-022-01378-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Woźniak Ł., Skąpska S., Marszałek K. Ursolic Acid—A Pentacyclic Triterpenoid with a Wide Spectrum of Pharmacological Activities. Molecules. 2015;20:20614–20641. doi: 10.3390/molecules201119721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Satoh T., Trudler D., Oh C.-K., Lipton S.A. Potential Therapeutic Use of the Rosemary Diterpene Carnosic Acid for Alzheimer’s Disease, Parkinson’s Disease, and Long-COVID through NRF2 Activation to Counteract the NLRP3 Inflammasome. Antioxidants. 2022;11:124. doi: 10.3390/antiox11010124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Tagde P., Tagde P., Islam F., Tagde S., Shah M., Hussain Z.D., Rahman H., Najda A., Alanazi I.S., Germoush M.O., et al. The Multifaceted Role of Curcumin in Advanced Nanocurcumin Form in the Treatment and Management of Chronic Disorders. Molecules. 2021;26:7109. doi: 10.3390/molecules26237109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Proshkina E., Plyusnin S., Babak T., Lashmanova E., Maganova F., Koval L., Platonova E., Shaposhnikov M., Moskalev A. Terpenoids as Potential Geroprotectors. Antioxidants. 2020;9:529. doi: 10.3390/antiox9060529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Geraldes V., Pinto E. Mycosporine-Like Amino Acids (MAAs): Biology, Chemistry and Identification Features. Pharmaceuticals. 2021;14:63. doi: 10.3390/ph14010063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Singh A., Čížková M., Bišová K., Vítová M. Exploring Mycosporine-Like Amino Acids (MAAs) as Safe and Natural Protective Agents against UV-Induced Skin Damage. Antioxidants. 2021;10:683. doi: 10.3390/antiox10050683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Becker K., Hartmann A., Ganzera M., Fuchs D., Gostner J. Immunomodulatory Effects of the Mycosporine-Like Amino Acids Shinorine and Porphyra-334. Mar. Drugs. 2016;14:119. doi: 10.3390/md14060119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kasanah N., Ulfah M., Imania O., Hanifah A.N., Marjan M.I.D. Rhodophyta as Potential Sources of Photoprotectants, Antiphotoaging Compounds, and Hydrogels for Cosmeceutical Application. Molecules. 2022;27:7788. doi: 10.3390/molecules27227788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Kageyama H., Waditee-Sirisattha R. Antioxidative, Anti-Inflammatory, and Anti-Aging Properties of Mycosporine-Like Amino Acids: Molecular and Cellular Mechanisms in the Protection of Skin-Aging. Mar. Drugs. 2019;17:222. doi: 10.3390/md17040222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Figueroa F.L. Mycosporine-Like Amino Acids from Marine Resource. Mar. Drugs. 2021;19:18. doi: 10.3390/md19010018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Xu D., Hu M.-J., Wang Y.-Q., Cui Y.-L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules. 2019;24:1123. doi: 10.3390/molecules24061123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Suh S.-S., Hwang J., Park M., Seo H., Kim H.-S., Lee J., Moh S., Lee T.-K. Anti-Inflammation Activities of Mycosporine-Like Amino Acids (MAAs) in Response to UV Radiation Suggest Potential Anti-Skin Aging Activity. Mar. Drugs. 2014;12:5174–5187. doi: 10.3390/md12105174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Chrapusta E., Kaminski A., Duchnik K., Bober B., Adamski M., Bialczyk J. Mycosporine-Like Amino Acids: Potential Health and Beauty Ingredients. Mar. Drugs. 2017;15:326. doi: 10.3390/md15100326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Stengel D.B., Connan S., editors. Natural Products from Marine Algae. Volume 1308. Springer; New York, NY, USA: 2015. [Google Scholar]
- 190.Pádua D., Rocha E., Gargiulo D., Ramos A.A. Bioactive Compounds from Brown Seaweeds: Phloroglucinol, Fucoxanthin and Fucoidan as Promising Therapeutic Agents against Breast Cancer. Phytochem. Lett. 2015;14:91–98. doi: 10.1016/j.phytol.2015.09.007. [DOI] [Google Scholar]
- 191.Michalak I. Experimental Processing of Seaweeds for Biofuels. WIREs Energy Environ. 2018;7:e288. doi: 10.1002/wene.288. [DOI] [Google Scholar]
- 192.Michalak I., Chojnacka K. Algal Extracts: Technology and Advances. Eng. Life Sci. 2014;14:581–591. doi: 10.1002/elsc.201400139. [DOI] [Google Scholar]
- 193.Mekinić I.G., Skroza D., Šimat V., Hamed I., Čagalj M., Perković Z.P. Phenolic Content of Brown Algae (Pheophyceae) Species: Extraction, Identification, and Quantification. Biomolecules. 2019;9:244. doi: 10.3390/biom9060244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Li A.-N., Li S., Zhang Y.-J., Xu X.-R., Chen Y.-M., Li H.-B. Resources and Biological Activities of Natural Polyphenols. Nutrients. 2014;6:6020–6047. doi: 10.3390/nu6126020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Heydarzadeh S., Kia S.K., Zarkesh M., Pakizehkar S., Hosseinzadeh S., Hedayati M. The Cross-Talk between Polyphenols and the Target Enzymes Related to Oxidative Stress-Induced Thyroid Cancer. Oxidative Med. Cell. Longev. 2022;2022:2724324. doi: 10.1155/2022/2724324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Rudrapal M., Khairnar S.J., Khan J., Bin Dukhyil A., Ansari M.A., Alomary M.N., Alshabrmi F.M., Palai S., Deb P.K., Devi R. Dietary Polyphenols and Their Role in Oxidative Stress-Induced Human Diseases: Insights into Protective Effects, Antioxidant Potentials and Mechanism(s) of Action. Front. Pharmacol. 2022;13:283. doi: 10.3389/fphar.2022.806470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Gugleva V., Ivanova N., Sotirova Y., Andonova V. Dermal Drug Delivery of Phytochemicals with Phenolic Structure via Lipid-Based Nanotechnologies. Pharmaceuticals. 2021;14:837. doi: 10.3390/ph14090837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Pereira Q.C., dos Santos T.W., Fortunato I.M., Ribeiro M.L. The Molecular Mechanism of Polyphenols in the Regulation of Ageing Hallmarks. Int. J. Mol. Sci. 2023;24:5508. doi: 10.3390/ijms24065508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Bié J., Sepodes B., Fernandes P.C.B., Ribeiro M.H.L. Polyphenols in Health and Disease: Gut Microbiota, Bioaccessibility, and Bioavailability. Compounds. 2023;3:40–72. doi: 10.3390/compounds3010005. [DOI] [Google Scholar]
- 200.Wang X., Qi Y., Zheng H. Dietary Polyphenol, Gut Microbiota, and Health Benefits. Antioxidants. 2022;11:1212. doi: 10.3390/antiox11061212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Rahman M., Rahaman S., Islam R., Rahman F., Mithi F.M., Alqahtani T., Almikhlafi M.A., Alghamdi S.Q., Alruwaili A.S., Hossain S., et al. Role of Phenolic Compounds in Human Disease: Current Knowledge and Future Prospects. Molecules. 2021;27:233. doi: 10.3390/molecules27010233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Martinengo P., Arunachalam K., Shi C. Polyphenolic Antibacterials for Food Preservation: Review, Challenges, and Current Applications. Foods. 2021;10:2469. doi: 10.3390/foods10102469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Fonseca S., Amaral M.N., Reis C.P., Custódio L. Marine Natural Products as Innovative Cosmetic Ingredients. Mar. Drugs. 2023;21:170. doi: 10.3390/md21030170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Dubois-Deruy E., Peugnet V., Turkieh A., Pinet F. Oxidative Stress in Cardiovascular Diseases. Antioxidants. 2020;9:864. doi: 10.3390/antiox9090864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Ullah A., Munir S., Badshah S.L., Khan N., Ghani L., Poulson B.G., Emwas A.-H., Jaremko M. Important Flavonoids and Their Role as a Therapeutic Agent. Molecules. 2020;25:5243. doi: 10.3390/molecules25225243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Gasmi A., Mujawdiya P.K., Noor S., Lysiuk R., Darmohray R., Piscopo S., Lenchyk L., Antonyak H., Dehtiarova K., Shanaida M., et al. Polyphenols in Metabolic Diseases. Molecules. 2022;27:6280. doi: 10.3390/molecules27196280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Du B., Zhao Q., Cheng C., Wang H., Liu Y., Zhu F., Yang Y. A Critical Review on Extraction, Characteristics, Physicochemical Activities, Potential Health Benefits, and Industrial Applications of Fucoidan. eFood. 2022;3:e19. doi: 10.1002/efd2.19. [DOI] [Google Scholar]
- 208.Catarino M.D., Amarante S.J., Mateus N., Silva A.M.S., Cardoso S.M. Brown Algae Phlorotannins: A Marine Alternative to Break the Oxidative Stress, Inflammation and Cancer Network. Foods. 2021;10:1478. doi: 10.3390/foods10071478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Patil A.D., Kasabe P.J., Dandge P.B. Pharmaceutical and Nutraceutical Potential of Natural Bioactive Pigment: Astaxanthin. Nat. Prod. Bioprospect. 2022;12:25. doi: 10.1007/s13659-022-00347-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Feldman F., Koudoufio M., Desjardins Y., Spahis S., Delvin E., Levy E. Efficacy of Polyphenols in the Management of Dyslipidemia: A Focus on Clinical Studies. Nutrients. 2021;13:672. doi: 10.3390/nu13020672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Din N.A.S., Alayudin S.M., Sofian-Seng N.-S., Rahman H.A., Razali N.S.M., Lim S.J., Mustapha W.A.W. Brown Algae as Functional Food Source of Fucoxanthin: A Review. Foods. 2022;11:2235. doi: 10.3390/foods11152235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Wang K., Xu X., Wei Q., Yang Q., Zhao J., Wang Y., Li X., Ji K., Song S. Application of Fucoidan as Treatment for Cardiovascular and Cerebrovascular Diseases. Ther. Adv. Chronic Dis. 2022;13:204062232210768. doi: 10.1177/20406223221076891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Šimat V., Rathod N.B., Čagalj M., Hamed I., Mekinić I.G. Astaxanthin from Crustaceans and Their Byproducts: A Bioactive Metabolite Candidate for Therapeutic Application. Mar. Drugs. 2022;20:206. doi: 10.3390/md20030206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Grosso G., Godos J., Currenti W., Micek A., Falzone L., Libra M., Giampieri F., Forbes-Hernández T.Y., Quiles J.L., Battino M., et al. The Effect of Dietary Polyphenols on Vascular Health and Hypertension: Current Evidence and Mechanisms of Action. Nutrients. 2022;14:545. doi: 10.3390/nu14030545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Mumu M., Das A., Emran T.B., Mitra S., Islam F., Roy A., Karim M., Das R., Park M.N., Chandran D., et al. Fucoxanthin: A Promising Phytochemical on Diverse Pharmacological Targets. Front Pharmacol. 2022;13:929442. doi: 10.3389/fphar.2022.929442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Venkatesan J., Keekan K.K., Anil S., Bhatnagar I., Kim S.-K. Encyclopedia of Food Chemistry. Elsevier; Amsterdam, The Netherlands: 2019. Phlorotannins; pp. 515–527. [Google Scholar]
- 217.Shin D., Shim S.R., Wu Y., Hong G., Jeon H., Kim C.-G., Lee K.J. How Do Brown Seaweeds Work on Biomarkers of Dyslipidemia? A Systematic Review with Meta-Analysis and Meta-Regression. Mar. Drugs. 2023;21:220. doi: 10.3390/md21040220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Ed Nignpense B., Chinkwo K.A., Blanchard C.L., Santhakumar A.B. Polyphenols: Modulators of Platelet Function and Platelet Microparticle Generation? Int. J. Mol. Sci. 2019;21:146. doi: 10.3390/ijms21010146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Manne B.K., Getz T.M., Hughes C.E., Alshehri O., Dangelmaier C., Naik U.P., Watson S.P., Kunapuli S.P. Fucoidan Is a Novel Platelet Agonist for the C-Type Lectin-like Receptor 2 (CLEC-2) J. Biol. Chem. 2013;288:7717–7726. doi: 10.1074/jbc.M112.424473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Wei Y., Wang C., Li J., Guo Q., Qi H. Inhibitory Effects and Mechanisms of High Molecular-Weight Phlorotannins from Sargassum Thunbergii on ADP-Induced Platelet Aggregation. Chin. J. Oceanol. Limnol. 2009;27:558–563. doi: 10.1007/s00343-009-9166-3. [DOI] [Google Scholar]
- 221.Biegańska-Hensoldt S., Rosołowska-Huszcz D. Polyphenols in Preventing Endothelial Dysfunction. Postep. Hig. Med. Dosw. 2017;71:227–235. doi: 10.5604/01.3001.0010.3808. [DOI] [PubMed] [Google Scholar]
- 222.Tanna B., Brahmbhatt H.R., Mishra A. Phenolic, Flavonoid, and Amino Acid Compositions Reveal That Selected Tropical Seaweeds Have the Potential to Be Functional Food Ingredients. J. Food Process. Preserv. 2019;43:e14266. doi: 10.1111/jfpp.14266. [DOI] [Google Scholar]
- 223.Olsthoorn S.E.M., Wang X., Tillema B., Vanmierlo T., Kraan S., Leenen P.J.M., Mulder M.T. Brown Seaweed Food Supplementation: Effects on Allergy and Inflammation and Its Consequences. Nutrients. 2021;13:2613. doi: 10.3390/nu13082613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Oliyaei N., Moosavi-Nasab M., Tamaddon A.M., Tanideh N. Antidiabetic Effect of Fucoxanthin Extracted from Sargassum Angustifolium on Streptozotocin-nicotinamide-induced Type 2 Diabetic Mice. Food Sci. Nutr. 2021;9:3521–3529. doi: 10.1002/fsn3.2301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Lopes G., Andrade P., Valentão P. Phlorotannins: Towards New Pharmacological Interventions for Diabetes Mellitus Type 2. Molecules. 2016;22:56. doi: 10.3390/molecules22010056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Aryaeian N., Sedehi S.K., Arablou T. Polyphenols and Their Effects on Diabetes Management: A Review. Med. J. Islam. Repub. Iran. 2017;31:886–892. doi: 10.14196/mjiri.31.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Sun C., Zhao C., Guven E.C., Paoli P., Simal-Gandara J., Ramkumar K.M., Wang S., Buleu F., Pah A., Turi V., et al. Dietary Polyphenols as Antidiabetic Agents: Advances and Opportunities. Food Front. 2020;1:18–44. doi: 10.1002/fft2.15. [DOI] [Google Scholar]
- 228.Lee S.-H., Li Y., Karadeniz F., Kim M.-M., Kim S.-K. α-Glucosidase and α-Amylase Inhibitory Activities of Phloroglucinal Derivatives from Edible Marine Brown Alga, Ecklonia Cava. J. Sci. Food Agric. 2009;89:1552–1558. doi: 10.1002/jsfa.3623. [DOI] [Google Scholar]
- 229.Yuan Y., Zheng Y., Zhou J., Geng Y., Zou P., Li Y., Zhang C. Polyphenol-Rich Extracts from Brown Macroalgae Lessonia Trabeculate Attenuate Hyperglycemia and Modulate Gut Microbiota in High-Fat Diet and Streptozotocin-Induced Diabetic Rats. J. Agric. Food Chem. 2019;67:12472–12480. doi: 10.1021/acs.jafc.9b05118. [DOI] [PubMed] [Google Scholar]
- 230.Catarino M., Silva A., Mateus N., Cardoso S. Optimization of Phlorotannins Extraction from Fucus Vesiculosus and Evaluation of Their Potential to Prevent Metabolic Disorders. Mar. Drugs. 2019;17:162. doi: 10.3390/md17030162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Xu Q., Luo J., Wu N., Zhang R., Shi D. BPN, a Marine-Derived PTP1B Inhibitor, Activates Insulin Signaling and Improves Insulin Resistance in C2C12 Myotubes. Int. J. Biol. Macromol. 2018;106:379–386. doi: 10.1016/j.ijbiomac.2017.08.042. [DOI] [PubMed] [Google Scholar]
- 232.Luo J., Hou Y., Xie M., Ma W., Shi D., Jiang B. CYC31, A Natural Bromophenol PTP1B Inhibitor, Activates Insulin Signaling and Improves Long Chain-Fatty Acid Oxidation in C2C12 Myotubes. Mar. Drugs. 2020;18:267. doi: 10.3390/md18050267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Seong S.H., Paudel P., Jung H.A., Choi J.S. Identifying Phlorofucofuroeckol-A as a Dual Inhibitor of Amyloid-Β25-35 Self-Aggregation and Insulin Glycation: Elucidation of the Molecular Mechanism of Action. Mar. Drugs. 2019;17:600. doi: 10.3390/md17110600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Lee S.-H., Kang S.-M., Ko S.-C., Kang M.-C., Jeon Y.-J. Octaphlorethol A, a Novel Phenolic Compound Isolated from Ishige Foliacea, Protects against Streptozotocin-Induced Pancreatic β Cell Damage by Reducing Oxidative Stress and Apoptosis. Food Chem. Toxicol. 2013;59:643–649. doi: 10.1016/j.fct.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 235.Kim E.-A., Kang M.-C., Lee J.-H., Kang N., Lee W., Oh J.-Y., Yang H.-W., Lee J.-S., Jeon Y.-J. Protective Effect of Marine Brown Algal Polyphenols against Oxidative Stressed Zebrafish with High Glucose. RSC Adv. 2015;5:25738–25746. doi: 10.1039/C5RA00338E. [DOI] [Google Scholar]
- 236.Yan X., Yang C., Lin G., Chen Y., Miao S., Liu B., Zhao C. Antidiabetic Potential of Green Seaweed Enteromorpha Prolifera Flavonoids Regulating Insulin Signaling Pathway and Gut Microbiota in Type 2 Diabetic Mice. J. Food Sci. 2019;84:165–173. doi: 10.1111/1750-3841.14415. [DOI] [PubMed] [Google Scholar]
- 237.Tanaka M., Toldi J., Vécsei L. Exploring the Etiological Links behind Neurodegenerative Diseases: Inflammatory Cytokines and Bioactive Kynurenines. Int. J. Mol. Sci. 2020;21:2431. doi: 10.3390/ijms21072431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Ashok A., Andrabi S.S., Mansoor S., Kuang Y., Kwon B.K., Labhasetwar V. Antioxidant Therapy in Oxidative Stress-Induced Neurodegenerative Diseases: Role of Nanoparticle-Based Drug Delivery Systems in Clinical Translation. Antioxidants. 2022;11:408. doi: 10.3390/antiox11020408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Rojas-García A., Fernández-Ochoa Á., Cádiz-Gurrea M.D.L.L., Arráez-Román D., Segura-Carretero A. Neuroprotective Effects of Agri-Food By-Products Rich in Phenolic Compounds. Nutrients. 2023;15:449. doi: 10.3390/nu15020449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Gentile M.T., Camerino I., Ciarmiello L., Woodrow P., Muscariello L., De Chiara I., Pacifico S. Neuro-Nutraceutical Polyphenols: How Far Are We? Antioxidants. 2023;12:539. doi: 10.3390/antiox12030539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Vasarri M., Degl’Innocenti D. Antioxidant and Anti-Inflammatory Agents from the Sea: A Molecular Treasure for New Potential Drugs. Mar. Drugs. 2022;20:132. doi: 10.3390/md20020132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Barbosa M., Valentão P., Andrade P.B. Polyphenols from Brown Seaweeds (Ochrophyta, Phaeophyceae): Phlorotannins in the Pursuit of Natural Alternatives to Tackle Neurodegeneration. Mar. Drugs. 2020;18:654. doi: 10.3390/md18120654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Karpiński T.M., Adamczak A. Fucoxanthin—An Antibacterial Carotenoid. Antioxidants. 2019;8:239. doi: 10.3390/antiox8080239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Dimitrova-Shumkovska J., Krstanoski L., Veenman L. Potential Beneficial Actions of Fucoidan in Brain and Liver Injury, Disease, and Intoxication—Potential Implication of Sirtuins. Mar. Drugs. 2020;18:242. doi: 10.3390/md18050242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Hannan A., Dash R., Haque N., Mohibbullah, Sohag A., Rahman A., Uddin J., Alam M., Moon I.S. Neuroprotective Potentials of Marine Algae and Their Bioactive Metabolites: Pharmacological Insights and Therapeutic Advances. Mar. Drugs. 2020;18:347. doi: 10.3390/md18070347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Choi B.W., Lee H.S., Shin H.-C., Lee B.H. Multifunctional Activity of Polyphenolic Compounds Associated with a Potential for Alzheimer’s Disease Therapy from Ecklonia Cava. Phytother. Res. 2015;29:549–553. doi: 10.1002/ptr.5282. [DOI] [PubMed] [Google Scholar]
- 247.Choi J.S., Haulader S., Karki S., Jung H.J., Kim H.R., Jung H.A. Acetyl- and Butyryl-Cholinesterase Inhibitory Activities of the Edible Brown Alga Eisenia Bicyclis. Arch. Pharmacal Res. 2015;38:1477–1487. doi: 10.1007/s12272-014-0515-1. [DOI] [PubMed] [Google Scholar]
- 248.Olasehinde T.A., Olaniran A.O., Okoh A.I. Phenolic Composition, Antioxidant Activity, Anticholinesterase Potential and Modulatory Effects of Aqueous Extracts of Some Seaweeds on β-Amyloid Aggregation and Disaggregation. Pharm. Biol. 2019;57:460–469. doi: 10.1080/13880209.2019.1634741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Olasehinde T.A., Olaniran A.O., Okoh A.I. Aqueous–Ethanol Extracts of Some South African Seaweeds Inhibit Beta-amyloid Aggregation, Cholinesterases, and Beta-secretase Activities In Vitro. J. Food Biochem. 2019;43:e12870. doi: 10.1111/jfbc.12870. [DOI] [PubMed] [Google Scholar]
- 250.Kim J.-J., Kang Y.-J., Shin S.-A., Bak D.-H., Lee J.W., Lee K.B., Yoo Y.C., Kim D.-K., Lee B.H., Kim D.W., et al. Phlorofucofuroeckol Improves Glutamate-Induced Neurotoxicity through Modulation of Oxidative Stress-Mediated Mitochondrial Dysfunction in PC12 Cells. PLoS ONE. 2016;11:e0163433. doi: 10.1371/journal.pone.0163433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Yang E.-J., Ahn S., Ryu J., Choi M.-S., Choi S., Chong Y.H., Hyun J.-W., Chang M.-J., Kim H.-S. Phloroglucinol Attenuates the Cognitive Deficits of the 5XFAD Mouse Model of Alzheimer’s Disease. PLoS ONE. 2015;10:e0135686. doi: 10.1371/journal.pone.0135686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Wang J., Zheng J., Huang C., Zhao J., Lin J., Zhou X., Naman C.B., Wang N., Gerwick W.H., Wang Q., et al. Eckmaxol, a Phlorotannin Extracted from Ecklonia Maxima, Produces Anti-β-Amyloid Oligomer Neuroprotective Effects Possibly via Directly Acting on Glycogen Synthase Kinase 3β. ACS Chem. Neurosci. 2018;9:1349–1356. doi: 10.1021/acschemneuro.7b00527. [DOI] [PubMed] [Google Scholar]
- 253.Lee S., Youn K., Kim D., Ahn M.-R., Yoon E., Kim O.-Y., Jun M. Anti-Neuroinflammatory Property of Phlorotannins from Ecklonia Cava on Aβ25-35-Induced Damage in PC12 Cells. Mar. Drugs. 2018;17:7. doi: 10.3390/md17010007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Monteiro P., Lomartire S., Cotas J., Marques J.C., Pereira L., Gonçalves A.M.M. Call the Eckols: Present and Future Potential Cancer Therapies. Mar. Drugs. 2022;20:387. doi: 10.3390/md20060387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Montuori E., de Pascale D., Lauritano C. Recent Discoveries on Marine Organism Immunomodulatory Activities. Mar. Drugs. 2022;20:422. doi: 10.3390/md20070422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Ali M., Benfante V., Stefano A., Yezzi A., Di Raimondo D., Tuttolomondo A., Comelli A. Anti-Arthritic and Anti-Cancer Activities of Polyphenols: A Review of the Most Recent In Vitro Assays. Life. 2023;13:361. doi: 10.3390/life13020361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Çetinkaya M., Baran Y. Therapeutic Potential of Luteolin on Cancer. Vaccines. 2023;11:554. doi: 10.3390/vaccines11030554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Jin J.-O., Yadav D., Madhwani K., Puranik N., Chavda V., Song M. Seaweeds in the Oncology Arena: Anti-Cancer Potential of Fucoidan as a Drug—A Review. Molecules. 2022;27:6032. doi: 10.3390/molecules27186032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Santaniello G., Nebbioso A., Altucci L., Conte M. Recent Advancement in Anticancer Compounds from Marine Organisms: Approval, Use and Bioinformatic Approaches to Predict New Targets. Mar. Drugs. 2022;21:24. doi: 10.3390/md21010024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Eo H., Kwon T.-H., Park G., Song H., Lee S.-J., Park N.-H., Jeong J. In Vitro Anticancer Activity of Phlorofucofuroeckol A via Upregulation of Activating Transcription Factor 3 against Human Colorectal Cancer Cells. Mar. Drugs. 2016;14:69. doi: 10.3390/md14040069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Kim R.-K., Uddin N., Hyun J.-W., Kim C., Suh Y., Lee S.-J. Novel Anticancer Activity of Phloroglucinol against Breast Cancer Stem-like Cells. Toxicol. Appl. Pharmacol. 2015;286:143–150. doi: 10.1016/j.taap.2015.03.026. [DOI] [PubMed] [Google Scholar]
- 262.Sadeeshkumar V., Duraikannu A., Ravichandran S., Kodisundaram P., Fredrick W.S., Gobalakrishnan R. Modulatory Efficacy of Dieckol on Xenobiotic-Metabolizing Enzymes, Cell Proliferation, Apoptosis, Invasion and Angiogenesis during NDEA-Induced Rat Hepatocarcinogenesis. Mol. Cell. Biochem. 2017;433:195–204. doi: 10.1007/s11010-017-3027-8. [DOI] [PubMed] [Google Scholar]
- 263.Li Y.-X., Li Y., Je J.-Y., Kim S.-K. Dieckol as a Novel Anti-Proliferative and Anti-Angiogenic Agent and Computational Anti-Angiogenic Activity Evaluation. Environ. Toxicol. Pharmacol. 2015;39:259–270. doi: 10.1016/j.etap.2014.11.027. [DOI] [PubMed] [Google Scholar]
- 264.Zhen A.X., Hyun Y.J., Piao M.J., Fernando P.D.S.M., Kang K.A., Ahn M.J., Yi J.M., Kang H.K., Koh Y.S., Lee N.H., et al. Eckol Inhibits Particulate Matter 2.5-Induced Skin Keratinocyte Damage via MAPK Signaling Pathway. Mar. Drugs. 2019;17:444. doi: 10.3390/md17080444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Karakaya S. Bioavailability of Phenolic Compounds. Crit. Rev. Food Sci. Nutr. 2004;44:453–464. doi: 10.1080/10408690490886683. [DOI] [PubMed] [Google Scholar]
- 266.Cosme P., Rodríguez A.B., Espino J., Garrido M. Plant Phenolics: Bioavailability as a Key Determinant of Their Potential Health-Promoting Applications. Antioxidants. 2020;9:1263. doi: 10.3390/antiox9121263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Zaynab M., Al-Yahyai R., Ameen A., Sharif Y., Ali L., Fatima M., Khan K.A., Li S. Health and Environmental Effects of Heavy Metals. J. King Saud Univ. Sci. 2022;34:101653. doi: 10.1016/j.jksus.2021.101653. [DOI] [Google Scholar]
- 268.Mildenberger J., Stangeland J.K., Rebours C. Antioxidative Activities, Phenolic Compounds and Marine Food Allergens in the Macroalgae Saccharina Latissima Produced in Integrated Multi-Trophic Aquaculture Systems. Aquaculture. 2022;546:737386. doi: 10.1016/j.aquaculture.2021.737386. [DOI] [Google Scholar]
- 269.Salinas-Roca B., Rubió-Piqué L., Montull-López A. Polyphenol Intake in Pregnant Women on Gestational Diabetes Risk and Neurodevelopmental Disorders in Offspring: A Systematic Review. Nutrients. 2022;14:3753. doi: 10.3390/nu14183753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Duda-Chodak A., Tarko T. Possible Side Effects of Polyphenols and Their Interactions with Medicines. Molecules. 2023;28:2536. doi: 10.3390/molecules28062536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Stromsnes K., Lagzdina R., Olaso-Gonzalez G., Gimeno-Mallench L., Gambini J. Pharmacological Properties of Polyphenols: Bioavailability, Mechanisms of Action, and Biological Effects in In Vitro Studies, Animal Models, and Humans. Biomedicines. 2021;9:1074. doi: 10.3390/biomedicines9081074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Munin A., Edwards-Lévy F. Encapsulation of Natural Polyphenolic Compounds; a Review. Pharmaceutics. 2011;3:793–829. doi: 10.3390/pharmaceutics3040793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Rein M.J., Renouf M., Cruz-Hernandez C., Actis-Goretta L., Thakkar S.K., da Silva Pinto M. Bioavailability of Bioactive Food Compounds: A Challenging Journey to Bioefficacy. Br. J. Clin. Pharmacol. 2013;75:588–602. doi: 10.1111/j.1365-2125.2012.04425.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Jeon H.-J., Yoon K.-Y., Koh E.-J., Choi J., Kim K.-J., Choi H.-S., Lee B.-Y. Seapolynol and Dieckol Improve Insulin Sensitivity through the Regulation of the PI3K Pathway in C57BL/KsJ-Db/Db Mice. J. Food Nutr. Res. 2015;3:648–652. doi: 10.12691/jfnr-3-10-5. [DOI] [Google Scholar]
- 275.Jeon H.-J., Choi H.-S., Lee Y.-J., Hwang J.-H., Lee O.-H., Seo M.-J., Kim K.-J., Lee B.-Y. Seapolynol Extracted from Ecklonia Cava Inhibits Adipocyte Differentiation In Vitro and Decreases Fat Accumulation In Vivo. Molecules. 2015;20:21715–21731. doi: 10.3390/molecules201219796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Hwang H.J. Skin Elasticity and Sea Polyphenols. Seanol Sci. Cent. Rev. 2010;1:1–10. [Google Scholar]
- 277.Origin of SEANOL. [(accessed on 30 April 2020)]. Available online: http://seanolinstitute.org/ssc/origin.html.
- 278.Apostolidis E., Lee C.M. Brown Seaweed-Derived Phenolic Phytochemicals and Their Biological Activities for Functional Food Ingredients with Focus on Ascophyllum Nodosum. In: Kim S., editor. Handbook of Marine Macroalgae. John Wiley & Sons, Ltd.; Chichester, UK: 2011. pp. 356–370. [Google Scholar]
- 279.Li K., Wang C., Wang Y., Fu L., Zhang N. Future Foods, Dietary Factors and Healthspan. J. Future Foods. 2023;3:75–98. doi: 10.1016/j.jfutfo.2022.12.001. [DOI] [Google Scholar]
- 280.Zuo W., Kwok H.F. Development of Marine-Derived Compounds for Cancer Therapy. Mar. Drugs. 2021;19:342. doi: 10.3390/md19060342. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.