Abstract
Objective
To evaluate the efficacy and safety of 0.01% atropine alone and in combination with orthokeratology for myopia control using a meta-analysis.
Methods
PubMed, Cochrane Library, and EMBASE were searched. We included eligible randomized controlled trials (RCTs), non-RCTs, and retrospective cohort studies, published up to August 1, 2022. We calculated the weighted mean difference (WMD) and 95% confidence interval (CI) for all outcomes and plotted them in forest plots.
Results
Fourteen studies were included; 4 and 11 in the 0.01% atropine monotherapy and atropine-orthokeratology (AOK) groups, respectively. Compared with orthokeratology (OK) alone, 0.01% atropine alone had similar effects on slowing the axial elongation (WMD: −0.00 mm; 95% CI: −0.05–0.04, p<0.31), while AOK significantly lowered axial growth. Moreover, the baseline myopic degree and duration of treatment were influential for the change in axial elongation (WMD: −0.12 mm; 95% CI: −0.17–−0.07, p = 0.00001 and WMD: −0.11 mm; 95% CI: −0.15–−0.108, p<0.00001, respectively). Additionally, the AOK may reduce the change rate of the spherical equivalent refraction and the accommodation amplitude (WMD: −0.13 D; 95% CI: 0.07–0.19, p<0.001 and WMD: −1.08 mm; 95% CI: −1.73–−0.43, p<0.0001, respectively), and cause a slight increase in the diameter of the pupil (WMD: 0.56 mm; 95% CI: 0.43–0.70, p = 0.007). No significant differences in the uncorrected distant visual acuity, best corrected visual acuity, intraocular pressure, tear film break-up time, lipid layer thickness, and corneal endothelial cell density were found between the OK and AOK groups.
Conclusion
In slowing the axial elongation, 0.01% atropine alone and OK alone have similar effects, while AOK is more effective than OK alone in slowing down the axial elongation. Furthermore, the baseline degree of myopia and treatment duration may affect changes in axial elongation.
Introduction
Myopia is among the most common eye diseases globally. The prevalence in the adult population is 10%–30% in many countries. In some parts of East and Southeast Asia, the prevalence in young adults is 80%–90% [1]. The development of severe myopia leads to excessive axial growth, and severe and irreversible vision loss including retinal detachment [2], macular dystrophy, and glaucoma [3]. Therefore, it is particularly important to take effective measures to slow the axial elongation to control the progression of myopia.
Among the current myopic interventions, atropine eye drops and orthokeratology (ortho-K) are considered effective in inhibiting the progression of myopia [4]. At present, the mechanism by which atropine controls myopia progression is not clear. Studies have suggested that atropine can slow myopia progression through a non-regulatory mechanism. It may act on M1 and M4 subtypes of the muscarinic receptors of the retina and sclera and slow the growth of the axial length, and may also promote the release of dopamine and other neurotransmitters, thus affecting the signal transduction of the retina and slow the growth of the axial length [5]. The atropine for the treatment of childhood myopia studies 1 and 2 have suggested that atropine has a dose-dependent clinical efficacy in controlling myopia progression and that low-concentration atropine was the most appropriate dosage, with fewer side effects of pupil dilation, photophobia, and cycloplegia [6, 7]. At present, 0.01% atropine is most commonly used for myopia control in East Asia, especially in Taiwan and Singapore [8]. However, atropine treatment must be combined with corrective spectacles, contact lenses, and orthokeratology (OK) lenses to provide refractive correction.
Orthokeratology (OK) is a special type of rigid contact lens with a reverse geometric design. These are worn at night to change the corneal curvature in patients, thus, reversibly reducing the degree of myopia and improving daytime vision [9]. The mechanism of the OK lens involves corneal epithelial remodeling (thinning of the central cornea and thickening of the peripheral cornea), which causes myopia defocusing in the peripheral retina, thus, controlling the progression of myopia [10]. Most studies have confirmed that OK can reduce axial elongation by 32%-63% over two years [11].
Some studies have compared atropine alone and OK alone for juvenile myopia, but the results remain controversial [12–14]. Due to the different mechanisms of atropine and OK in controlling myopia progression, the combination of atropine and OK may produce an additive effect. Several studies have confirmed that the combination of 0.01% atropine and OK is more effective than OK alone in slowing axial elongation in children with myopia [15–25]. In this meta-analysis, we aimed to further evaluate the efficacy and safety of 0.01% atropine alone and in combination with orthokeratology for myopia control.
Methods
Search strategy
The databases, PubMed, Cochrane Library, and EMBASE, were searched. Potentially eligible randomized controlled trials (RCTs), non-randomized controlled trials (non-RCTs), and retrospective cohort studies (REs) published from the establishment of the database to August 1, 2022, were considered. The search terms, using Medical Subject Headings and free words, were as follows: orthokeratology procedures, orthokeratology, ortho-k, OK lens, orthokeratology lens, atropine, atropine sulfate, myopia, near sight, refractive errors, and nearsightedness. The primary treatment efficacy was measured via the mean change in axial length (AL) and spherical equivalent refraction (SER) after therapy. The primary measures of treatment safety included the mean change in uncorrected distant visual acuity (UCVA), best corrected visual acuity (BCVA), pupil diameter (PD), accommodation amplitude (AA), intraocular pressure (IOP), tear film break-up time (TBUT), lipid layer thickness (LLT), and corneal endothelial cell density (CECD). Furthermore, the reference lists of published reviews were carefully screened to identify applicable studies.
Eligibility criteria
We selected all studies according to the criteria as follows. (1) Study population: children, aged under 18 years, with myopia, equivalent spherical refraction of less than -6.0D, and binocular anisometropia of less than -1.5D at baseline. (2) Intervention measures: the experimental group was treated with 0.01% atropine combined with OK or 0.01% atropine alone, whereas the control group received OK only. (3) Study Design: RCTs, non-RCTs, and REs. (4) The study described at least one of the following outcomes: a mean change in AL, SER, UCDA, BCDA, PD, AA, IOP, TBUT, LLT, or CECD after treatment, and which was statistically significant. (5) Treatment duration: at least 6 months. Studies that did not meet the inclusion criteria, or had incorrect or incomplete data, conference reviews, case reports, and duplicate publications were excluded.
Data extraction and quality assessment
Two reviewers (WZY and YZP) independently performed data extraction and quality assessment. All extracted data were recorded in a Microsoft Excel spreadsheet including first author, year of publication, study design, intervention, follow-up time, baseline participant characteristics (age, sample size, AL, and SER), and measurement methods. The RCT quality was evaluated according to the Cochrane Collaboration’s tool for assessing the risk of bias [26], which included seven domains as follows: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases. In each domain, the reviewers assessed each domain as “low risk” of bias, “high risk” of bias, or “clear risk” of bias, according to the given criteria. If insufficient details are reported in the study, an “unclear risk” of bias was considered. We used the Newcastle-Ottawa Scale to assess the quality of the observational studies [27]. A score of seven or more on the Newcastle-Ottawa Scale was considered high quality, four to six as moderate quality, and less than three as low quality. The quality of each included non-RCT was assessed based on the methodological index for non-randomized studies (MINORS) [28]. Disagreements were resolved by discussion with third-party experts or by focused discussions that involved at least three reviewers.
Statistical analysis
Statistical analyses were performed using Review Manager (version 5.3). The weighted mean difference (WMD), and 95% confidence interval (CI), were calculated for all the outcomes. Heterogeneity was assessed using I2 statistics. If I2 was ≥50%, the random-effects model was used for meta-analysis. Otherwise, the fixed-effects model was chosen. Sensitivity analysis was performed by sequentially eliminating one study. Publication bias was detected using a funnel plot test.
Results
Literature search and characteristics of the included studies
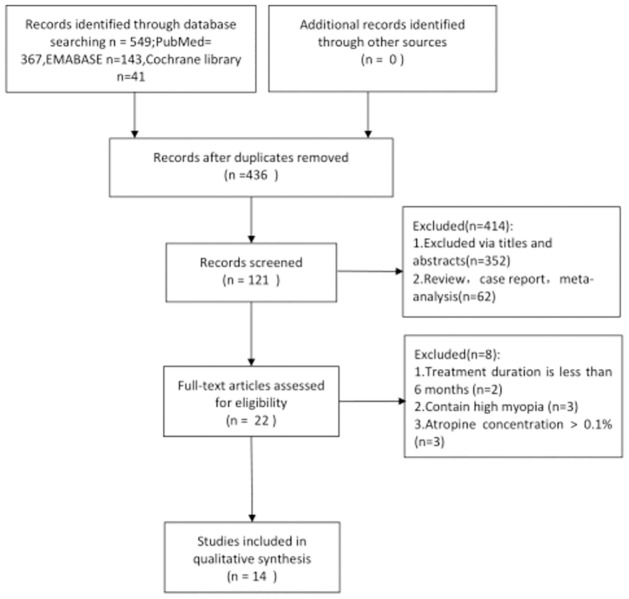
A total of 549 studies were obtained from the searched databases based on the specified search strategy. After eliminating 111 duplicates, we reviewed the abstracts of 436 studies, of which 414 were irrelevant. The final 22 studies were reviewed in full, and 8 were excluded because they did not meet our inclusion criteria. Finally, 14 studies were included in this meta-analysis, including 8 RCTs, 3 non-RCTs, and 3 REs. The screening process for eligible studies is shown in a flow diagram in Fig 1.
Fig 1. A flow diagram of the included studies eligible for meta-analysis.
Table 1 presents the characteristics of the 14 eligible studies. Four studies were categorized into the 0.01% atropine monotherapy group [12–14, 25], and 11 were classified into the AOK group [15–25]. The study by Zhao (2021) [25] was included in both the 0.01% atropine monotherapy and AOK groups. Two studies (Kinoshita, 2018 [18] and Kinoshita, 2020 [17]) were conducted in Japan, and the remainder were conducted in China. A total of 1230 children with myopia were included in this study. Of the 1230 children, 395 were in the 0.01% atropine monotherapy group and 865 were in the AOK group. In the 0.01% atropine monotherapy group, the children were prescribed single-vision spectacle lenses that were worn constantly, in addition to administering one drop of 0.01% atropine in each eye before bedtime every night. In 11 studies, self-prepared low-concentration atropine was used, while in three studies, low-concentration, preservative-free atropine eye drops were used.
Table 1. Baseline characteristics of all included studies.
| First author | Country | Study design | Sample size(E/C) | Age(years) | Group | Follow-up(month) | SER,D(E/C) | Axial Length, mm(E/C) | Axial Length Measurements |
|---|---|---|---|---|---|---|---|---|---|
| Ji N2022 | China | RE | 46/49 | 9.96±1.75/10.14±1.84 | SA/OK | 12 | H:-3.78±1.21/-4.01±1.13 | H: 24.71±0.89/24.80±0.81 | IOL master |
| L: -2.18±1.08/-1.94±1.33 | L:24.01±0.84/24.08±1.32 | ||||||||
| Jiang J2018 | China | Non-RCT | 80/80 | 11.50±2.30/12.10±2.10 | SA/OK | 18 | -2.85±0.54/-2.75±0.52 | 23.90±1.30/23.93±1.07 | - |
| Ren QL2017 | China | RCT | 50/50 | 11.96±2.58 | SA/OK | 12 | -3.16±0.28/-3.08±0.11 | 24.32±1.16/24.28±1.32 | - |
| Zhao Q2021 | China | RCT | 20/20 | 9.65±1.53/11.00±1.17 | SA/OK | 6,12 | -1.98±0.45/-2.75±0.46 | 24.17±0.68/24.42±0.48 | LS-900 |
| Zhao Q2022 | China | RCT | 20/21 | 10.9±1.29/11.00±1.17 | AOK/OK | 6,13 | -2.85±0.45/-2.75±0.46 | 24.56±0.39/24.42±0.48 | LS-900 |
| Tan Q2020 | China | RCT | 29/30 | 9.20±1.00/9.10±1.20 | AOK/OK | 6,12 | -2.65±0.92/-2.84±0.96 | 24.43±0.62/24.43±0.81 | IOL master |
| Tan Q2022 | China | RCT | 34/35 | 9.20±1.00/9.10±1.20 | AOK/OK | 6,12,24 | -2.76±0.88/-2.83±1.01 | 24.56±0.71/24.50±0.92 | IOL master |
| Chen Z2019 | China | RE | 28/29 | 7.50±1.20/8.30±1.50 | AOK/OK | 12 | -2.70±1.10/-2.50±0.94 | 24.80±0.87/24.53±0.61 | IOL master |
| Chen Z2021 | China | RE | 37/36 | 8.90±1.40/8.80±1.21 | AOK/OK | 12,24 | -2.85±1.08/-2.38±1.10 | 24.49±0.96/24.26±0.90 | IOL master |
| Kinoshita N2018 | Japan | RCT | 20/20 | 10.87±1.38/10.40±1.86 | AOK/OK | 12 | − 2.81±1.43/− 2.95±1.43 | 24.73±0.58/24.95±0.92 | IOL master |
| Kinoshita N2020 | Japan | RCT | 38/35 | 10.33±1.59/10.37±1.65 | AOK/OK | 24 | -2.60±1.29/-2.72±1.31 | 24.69±0.58/24.86±0.81 | IOL master |
| Vincent J2020 | China | RCT | 25/38 | 8.90±1.20/9.10±1.10 | AOK/OK | 6 | -2.38±0.81/-2.58±0.91 | 24.38±0.62/24.44±0.84 | IOL master |
| Tang WT2020 | China | RCT | L: 20/22 H: 43/41 | 11.05±2.13 | AOK/OK | 12 | L:-2.56±1.15/-2.59±1.12 | L:23.72±0.31/23.70±0.29 | IOL master |
| H:-4.90±1.16/-4.92±1.21 | H:24.69±0.34/24.71±0.37 | ||||||||
| Niu YL2019 | China | Non-RCT | 80/72 | 13.73±2.52/13.61±2.41 | AOK/OK | 12 | -1.40±0.25/-1.38±0.23 | 23.58±3.39/23.61±3.42 | IOL master |
| Luo Y2021 | China | Non-RCT | 60/60 | 10.02±2.35/10.77±1.91 | AOK/OK | 12 | -3.26±1.35/-3.18±1.32 | 25.12±0.81/25.01±0.79 | IOL master |
Bias risk assessment
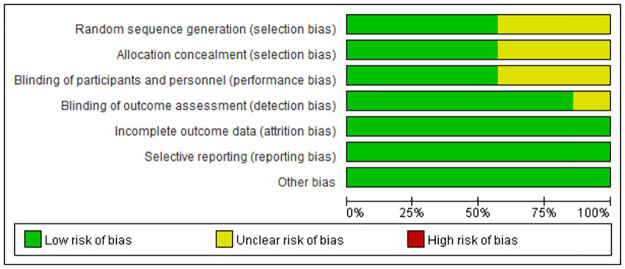
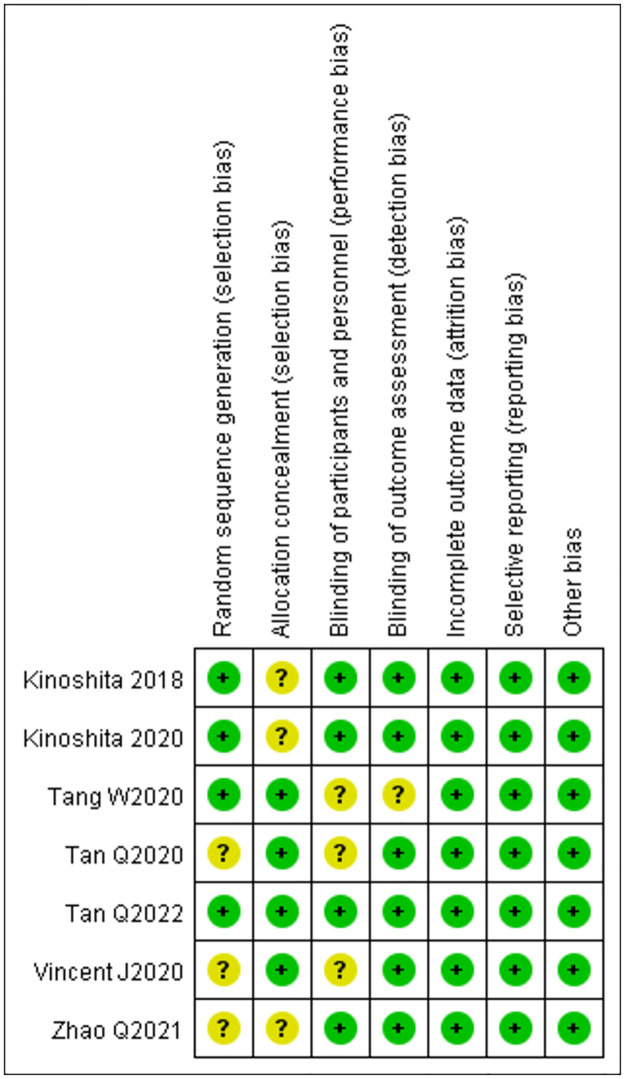
According to the Cochrane Collaboration’s tool for assessing the risk of bias, the quality of the RCTs was high (Figs 2 and 3). Four studies adopted randomization methods [17, 18, 20, 22], while the remaining studies did not. Only four studies used blinding methods [20–23]. The included studies completely reported all outcome data. Other biases in all the studies were regarded as low risk. The Newcastle-Ottawa Scale was used to evaluate the cohort studies. The three cohort studies scored six, six, and eight, respectively on the Newcastle-Ottawa Scale, and therefore, were assessed to be of moderate quality (Table 2). The MINORS was used to evaluate the non-randomized studies, and all studies demonstrated a high quality with a score of 19 (Table 3).
Fig 2. Risk of bias graph.
Quality evaluation of included randomised controlled trials.
Fig 3. Risk of bias summary.
Quality evaluation of included randomised controlled trials.
Table 2. Results of quality assessment using the Newcastle-Ottawa scale for cohort studies.
| Selection | Comparabiliy | Outcome | Score | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Representativeness of the exposed cohort | Seletion of the non exposed cohort | Ascertainment of exposure | Demonstration that outcome of interest was not present at start of study | Comparability of cohorts on the basis of the design or analysis | Assessment of outcome | Was follow-up long enough for outcomes to occur | Adequacy of follow up of cohorts | ||
| Chen Z 2019 | * | * | * | - | - | * | * | * | 6 |
| Chen Z 2021 | * | * | * | - | * | * | * | * | 8 |
| Ji N 2022 | * | * | * | - | * | * | * | * | 8 |
A study can be awarded a maximum of one asterisk for each numbered item within the Selection and Outcome categories. A maximum of two asterisks can be given for Comparability.
Table 3. The revised and validated version of MINORS.
| Methodological items for non-randomized studies | Score† | ||
|---|---|---|---|
| Luo Y2021 | Niu YL2019 | Jing J2018 | |
| 1. A clearly stated aim: the question addressed should be precise and relevant in the light of available literature | 2 | 2 | 2 |
| 2. Inclusion of consecutive patients: all patients potentially fit for inclusion (satisfying the criteria for inclusion) have been included in the study during the study period (no exclusion or details about the reasons for exclusion) | 1 | 1 | 1 |
| 3. Prospective collection of data: data were collected according to a protocol established before the beginning of the study | 2 | 2 | 2 |
| 4. Endpoints appropriate to the aim of the study: unambiguous explanation of the criteria used to evaluate the main outcome which should be in accordance with the question addressed by the study. Also, the endpoints should be assessed on an intention-to-treat basis. | 2 | 2 | 2 |
| 5. Unbiased assessment of the study endpoint: blind evaluation of objective endpoints and double-blind evaluation of subjective endpoints. Otherwise the reasons for not blinding should be stated | 0 | 0 | 0 |
| 6. Follow-up period appropriate to the aim of the study: the follow-up should be sufficiently long to allow the assessment of the main endpoint and possible adverse events | 2 | 2 | 2 |
| 7. Loss to follow up less than 5%: all patients should be included in the follow up. Otherwise, the proportion lost to follow up should not exceed the proportion experiencing the major endpoint | 2 | 2 | 2 |
| 8. Prospective calculation of the study size: information of the size of detectable difference of interest with a calculation of 95% confidence interval, according to the expected incidence of the outcome event, and information about the level for statistical significance and estimates of power when comparing the outcomes | 0 | 0 | 0 |
| Additional criteria in the case of comparative study | |||
| 9. An adequate control group: having a gold standard diagnostic test or therapeutic intervention recognized as the optimal intervention according to the available published data | 2 | 2 | 2 |
| 10. Contemporary groups: control and studied group should be managed during the same time period (no historical comparison) | 2 | 2 | 2 |
| 11. Baseline equivalence of groups: the groups should be similar regarding the criteria other than the studied endpoints. Absence of confounding factors that could bias the interpretation of the results | 2 | 2 | 2 |
| 12. Adequate statistical analyses: whether the statistics were in accordance with the type of study with calculation of confidence intervals or relative risk | 2 | 2 | 2 |
| 总分 | 19 | 19 | 19 |
†The items are scored 0 (not reported), 1 (reported but inadequate) or 2 (reported and adequate). The global ideal score being 16 for non-comparative studies and 24 for comparative studies.
Atropine monotherapy group
Change in AL
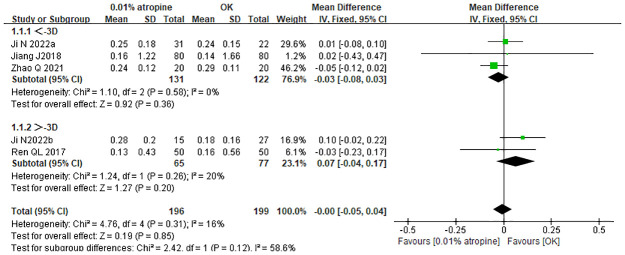
All four studies analyzed the changes in AL with 0.01% atropine alone and OK alone. Based on an I2 of 16%, the fixed-effects model was selected for data merging. The WMD in the change in AL was -0.00 mm (95% CI: −0.05–0.04), which was statistically significant (p = 0.31). The meta-analysis indicated that the change in AL in the 0.01% atropine group was similar to that in the OK group.
We performed a subgroup analysis according to whether the SER was greater than 3, to examine the effect of the baseline myopic degree on axial elongation. The corresponding WMDs for the aforementioned subgroups were as follows: −0.03 mm (95% CI: −0.08–−0.03) and 0.07 mm (95% CI: −0.04–0.17), respectively. These results indicated that the mean axial elongation in the 0.01% atropine alone group was similar to that in the OK alone group (Fig 4).
Fig 4. Forest plot of WMD of axial elongation in the atropine monotherapy group and the ortho-k monotherapy group and subgroup analysis by SER.
WMD, weighted mean difference.
Atropine combined with ortho-k therapy group
Change in AL
All 11 studies reported changes in the AL at baseline versus different follow-up periods in the AOK and OK groups. Of note, we chose a random-effects model because of the significant heterogeneity (I2 = 95%).
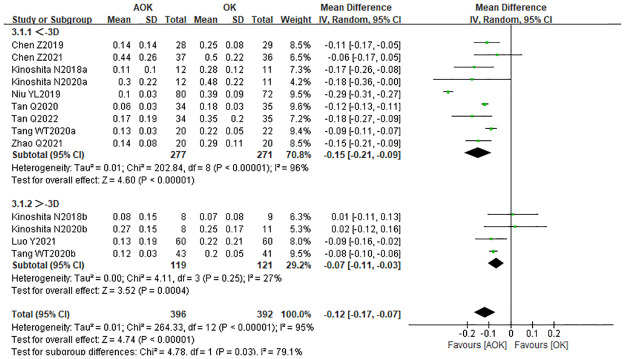
To investigate the effect of baseline refractive error (diopter) on axial elongation, we performed a subgroup analysis. We divided the included studies into two subgroups: low myopia (−1 to −3 D) and medium myopia (−3 to −6 D). There were nine studies with a baseline SER between −1 to −3 D. The combined results showed that the WMD in the low myopia subgroup was −0.15 mm (95% CI: −0.21–−0.09). There were four studies with a baseline SER between −3 to −6 D. The WMD of the medium myopia subgroup was −0.07 mm (95% CI: −0.11–0.03; Fig 5). The results confirmed that the AOK group significantly slowed axial elongation in children with low to moderate myopia, relative to the OK group.
Fig 5. Forest plot of WMD of axial elongation in the AOK group and the OK group and subgroup analysis by baseline SER.
WMD, weighted mean difference.
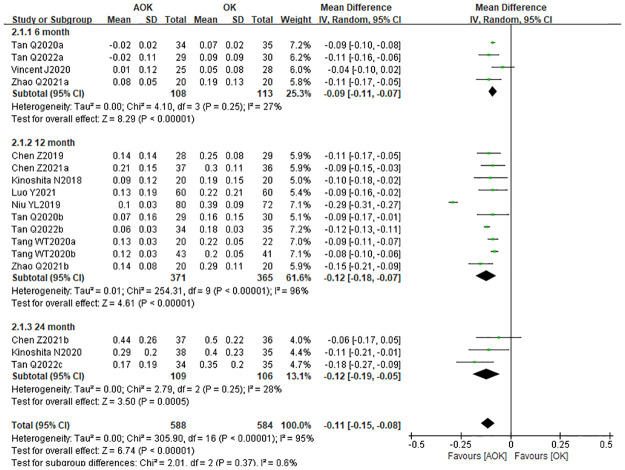
We performed another subgroup analysis to investigate the impact of the follow-up period on axial elongation. The studies were divided into three groups according to the follow-up times of 6, 12, and 24 months. The corresponding WMD for each of the subgroups was as follows: −0.09 mm (95% CI: −0.11–−0.07), −0.12 mm (95% CI: −0.18–−0.07), and −0.12 mm (95% CI: −0.19–−0.05), respectively (Fig 6). The results indicated that AOK was more effective in slowing axial elongation than OK alone. The findings also suggest that the longer the follow-up period, the more significant and stable the effect of AOK on slowing the axial elongation.
Fig 6. Forest plot of WMD of axial elongation in the AOK group and the OK group and subgroup analysis by follow-up periods.
WMD, weighted mean difference.
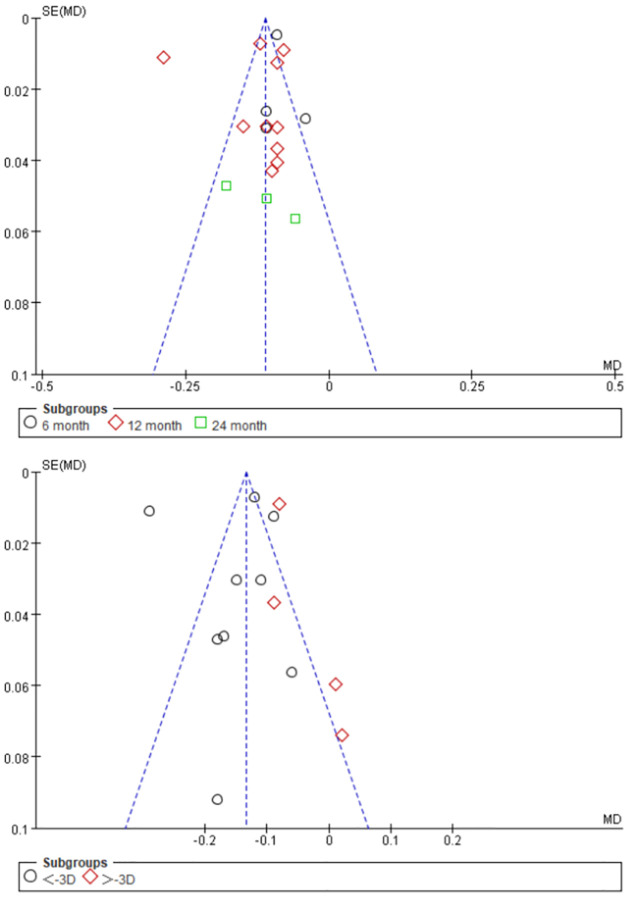
Publication bias
A funnel plot was used to assess publication bias (Fig 7). The results showed no publication bias among the studies included in this meta-analysis.
Fig 7. Funnel plot of axial length (AL).
Sensitivity analysis
A sensitivity analysis was conducted to estimate the effects of a single study on the overall results by removing one study at a time (Table 4). The analysis revealed that no single study significantly changed the overall results, thus, indicating that the pooled effect of the meta-analysis was stable and reliable.
Table 4. Subgroup sensitivity analysis.
| Total (95%CI) | Heterogeneity | Test for overall effect | |
|---|---|---|---|
| Removal of Chen Z 2019 | -0.13 [-0.14, -0.12] | I2 = 96% | P < 0.00001 |
| Removal of Chen Z 2021 | -0.13 [-0.14, -0.12] | I2 = 96% | P < 0.00001 |
| Removal of Kinoshita N 2018 | -0.13 [-0.14, -0.12] | I2 = 96% | P < 0.00001 |
| Removal of Kinoshita N 2020 | -0.13 [-0.14, -0.12] | I2 = 96% | P < 0.00001 |
| Removal of Tan Q 2020 | -0.14 [-0.15, -0.13] | I2 = 96% | P<0.00001 |
| Removal of Tan Q2022 | -0.13 [-0.14, -0.12] | I2 = 96% | P<0.00001 |
| Removal of Tang WT2020 | -0.14 [-0.15, -0.13] | I2 = 96% | P<0.00001 |
| Removal of Zhao Q 2021 | -0.13 [-0.14, -0.12] | I2 = 96% | P<0.00001 |
| Removal of Niu YL2019 | -0.10 [-0.11, -0.09] | I2 = 61% | P = 0.003 |
| Removal of Luo Y2021 | -0.13 [-0.14, -0.12] | I2 = 96% | P<0.00001 |
| Removal of Vincent J2020 | -0.11 [-0.12, -0.11] | I2 = 95% | P<0.00001 |
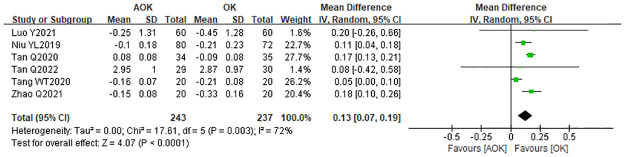
Change in SER
Six studies compared OK with AOK in terms of the change in SER after treatment. Due to significant heterogeneity (I2 = 72%), a randomized effect model was selected, with the results showing WMD = 0.13 D (95% CI 0.07–0.19, p = 0.003; Fig 8). The results from this meta-analysis suggest that AOK is more effective in slowing SER progression than OK was.
Fig 8. Forest plot of the comparison of change in SER.
WMD, weighted mean difference.
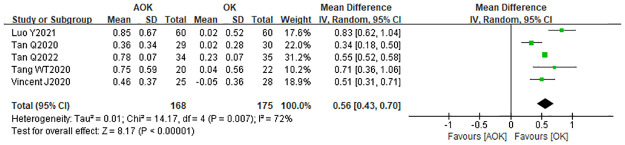
Change in PD
Five studies compared OK with AOK in terms of the change in PD after treatment. Owing to significant heterogeneity (I2 = 72%), a randomized effect model was selected, with the results showing WMD = 0.56 mm (95% CI: 0.43–0.70, p = 0.007; Fig 9). The results suggest that AOK may slightly increase the PD, compared with OK alone.
Fig 9. Forest plot of the comparison of change in PD.
WMD, weighted mean difference.
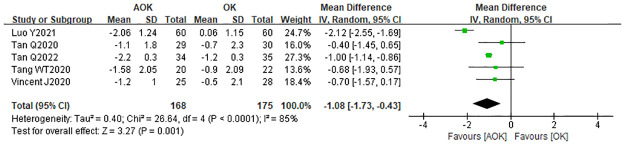
Change in AA
Three studies included the change in AA. Based on an I2 of 85%, a randomized effects model was used. The WMD for the change in AL was −1.08 mm (95% CI: −1.73–−0.43), which was statistically significant (p<0.0001; Fig 10). These results suggest that AOK may slightly reduce the AA, compared with the effect of OK alone.
Fig 10. Forest plot of the comparison of change in AA.
WMD, weighted mean difference.
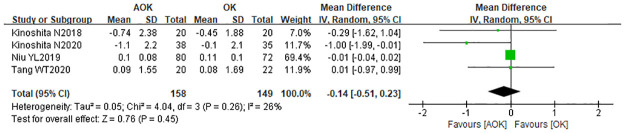
Change in IOP
The change in IOP between the two groups was reported by four studies. The combined results revealed a WMD of -0.14 (95% CI: −0.51–0.23), which suggested that the IOP was not significantly different between the two groups, as shown in Fig 11. Furthermore, there was no statistical heterogeneity between the two groups (p = 0.51, I2 = 0%).
Fig 11. Forest the comparison of change in IOP.
WMD, weighted mean difference.
Change in TBUT
Four studies reported the change in TBUT between the OK and AOK groups. These studies showed no statistical heterogeneity (p = 0.09). The results indicated WMD = −0.01 s (95% CI: −0.48–0.45), which suggests that TBUT was not significantly different between the AOK and OK groups (Fig 12).
Fig 12. Forest plot of the comparison of change in TBUT.
WMD, weighted mean difference.
Change in LLT
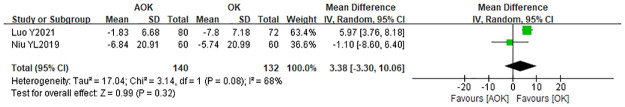
Two studies reported the change in LLT between the combination and monotherapy groups. The combined results indicated that there was no statistical difference in LLT between the two groups (95% CI: −3.30–10.06; Fig 13). Moreover, there was no significant between-study heterogeneity (p = 0.08, I2 = 68%).
Fig 13. Forest plot of the comparison of change in LLT.
WMD, weighted mean difference.
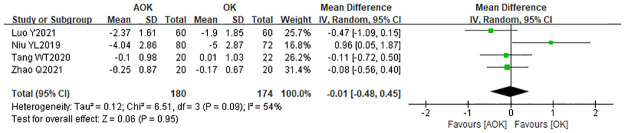
Change in CECD
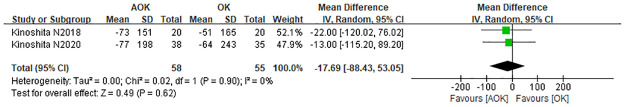
Two studies reported the change in CECD between the OK and AOK groups. The results showed WMD = –17.69 (95% CI: –88.43−53.05), which suggests that CECD did not differ significantly between the AOK and OK groups (Fig 14).
Fig 14. Forest plot of the comparison of change in CECD.
WMD, weighted mean difference.
Change in UCVA
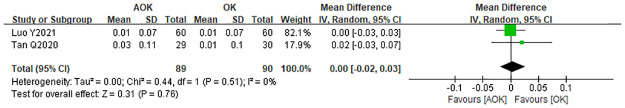
Two studies included the change in UCVA. The results indicated no significant difference in UCVA between the groups (95% CI: –0.02−0.03; Fig 15). Also, no statistical heterogeneity was observed between the groups (p = 0.51, I2 = 0%).
Fig 15. Forest plot of the comparison of change in UCVA.
WMD, weighted mean difference.
Change in BCVA
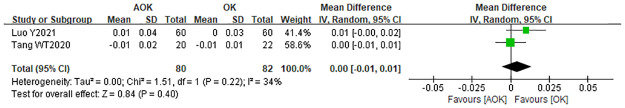
Two studies reported the change in BCVA. The results indicated no significant difference in BCVA between the two groups (95% CI: –0.01−0.01), as shown in Fig 16. There was no statistical heterogeneity between the two groups (p = 0.90, I2 = 0%).
Fig 16. Forest plot of the comparison of change in BCVA.
WMD, weighted mean difference.
Discussion
The results of this meta-analysis reveal that the axial growth rate of children who wear overnight OK alone is similar to that of children who wear single-vision spectacle lenses during the day and use one drop of 0.01% atropine before bedtime every night. Moreover, 0.01% atropine combined with OK is more effective than OK alone in slowing the axial elongation. The subgroup analysis demonstrated that the change in AL was influenced by the baseline myopic degree and duration of treatment. In contrast, there was no significant difference in the IOP, TBUT, LLT, CECD, UCDA, or BCDA between the AOK and OK treatment groups. However, the combination therapy is associated with a slight increase in PD and a decrease in the AA relative to OK therapy alone.
Presently, both atropine and OK are considered effective treatment strategies for myopia control. However, some controversies remain. Huang et al. reported that OK was worse than high- and medium-dose atropine, and equal to low-dose atropine for myopia control [29]. The latest SUCRA analysis revealed that in terms of hierarchy, OK was similar to 0.01%−0.025% atropine in AL inhibition over a one-year follow-up period [30]. There have been many reports that OK is more effective in slowing axial elongation in children with high myopia than in children with low myopia [31, 32]. Our meta-analysis shows that 0.01% atropine alone and OK alone have similar effects on slowing the axial elongation. However, with OK alone, the effect was greater in children with high myopia.
Synergistic effects of OK and atropine have been observed in previous studies [15–25]. Wan et al. demonstrated that the potential mechanisms underlying the combined effect of atropine and OK lenses relate to a large PD that increases retinal illumination, which in turn, lowers the myopic shift in the peripheral retina and enhances the effect of the OK lens [33]. Since all study participants wore OK lenses, we can assume that the difference between the two therapies, in terms of the change in AL, was primarily due to atropine. Low-concentration atropine can also lead to slight pupil enlargement, which may facilitate the optical effect of OK lenses [34]. Kinoshita et al. [17, 18] found that the additive effects of OK and 0.01% atropine on slowing axial elongation were greater in children with low myopia, whereas OK monotherapy was as effective as the combination therapy in children with high myopia [17, 18]. Conversely, the results of this meta-analysis conflicted with these previous studies. In this study, the effect of AOK in slowing axial elongation was greater in children with low myopia. This may be because the more peripheral myopia defocus was induced in high myopia compared with low myopia during OK treatment as described by previous studies. The effect of OK monotherapy in slowing the progression of high myopia may be adequate, whereas in children with low initial myopia, the amount of peripheral myopia defocusing may be insufficient. Additionally, the use of low-concentration atropine can compensate for the control effect of ortho-k in low myopia and contribute synergistically [17, 18, 31, 32].
AOK therapy significantly slowed the axial elongation at 6 and 12 months. However, when the treatment duration was extended to 24 months, the combination therapy did not lead to a further decrease in the axial elongation rate. Previous studies suggest that this may be due to temporary slight choroidal thickening after using atropine, which exaggerates the axial elongation control effect [35, 36]. Some studies also reported that the OK lens treatment effect was most marked within the first 12 months, and the effect becomes less marked but still significant in the second and third years [37].
No significant differences were found in the IOP, TBUT, LLT, CECD, UCDA, and BCDA between the AOK and OK groups, indicating that combination therapy for children with myopia has no negative influence on clinical results. The main complications of atropine use were photophobia, blurred vision, and allergic reactions [38]. The main complications of OK included infectious keratitis and allergic conjunctivitis [39]. These adverse reactions completely resolved when treatment stopped. Hence, the two interventions are considered relatively safe.
Our meta-analysis had some limitations. First, studies on 0.01% atropine alone and OK alone are still limited, and publication biases might have occurred. We only used axial elongation as the primary evaluation factor. We could not include refraction, PD, and other outcomes as the majority of the included studies did not report them. Second, due to the concentration dependence of atropine, different doses of atropine alone and OK alone must be analyzed. Third, the participants were all Asian children. The rate of myopia progression in Chinese children is generally higher than that in Caucasian children; thus, ethnicity may impact myopia progression and may influence the intervention effect. Future studies should consider ethnic differences. Finally, we did not consider all possible factors; for example, environmental and genetic factors, outdoor activities, and close work.
Conclusions
For children with myopia, 0.01% atropine alone, and OK alone, have similar effects on slowing axial elongation. Conversely, 0.01% atropine combined with OK is more effective than OK alone in slowing axial elongation. Furthermore, baseline myopic degree and duration of treatment may affect the changes in axial elongation.
Supporting information
(DOC)
(DOC)
(DOC)
Data Availability
All relevant data are within the manuscript and its Supporting information files.
Funding Statement
The author(s) received no specific funding for this work.
References
- 1.Baird PN, Saw SM, Lanca C, Guggenheim JA, Smith EL Iii, Zhou X, et al. Myopia. Nat Rev Dis Primers. 2020;6(1):99. Epub 2020/12/18. doi: 10.1038/s41572-020-00231-4 . [DOI] [PubMed] [Google Scholar]
- 2.Crim N, Esposito E, Monti R, Correa LJ, Serra HM, Urrets-Zavalia JA. Myopia as a risk factor for subsequent retinal tears in the course of a symptomatic posterior vitreous detachment. BMC ophthalmology. 2017;17(1):226. Epub 2017/12/02. doi: 10.1186/s12886-017-0629-6 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ha A, Kim CY, Shim SR, Chang IB, Kim YK. Degree of Myopia and Glaucoma Risk: A Dose-Response Meta-analysis. American journal of ophthalmology. 2022;236:107–19. Epub 2021/10/15. doi: 10.1016/j.ajo.2021.10.007 . [DOI] [PubMed] [Google Scholar]
- 4.Ang M, Flanagan JL, Wong CW, Muller A, Davis A, Keys D, et al. Review: Myopia control strategies recommendations from the 2018 WHO/IAPB/BHVI Meeting on Myopia. The British journal of ophthalmology. 2020;104(11):1482–7. Epub 2020/02/28. doi: 10.1136/bjophthalmol-2019-315575 . [DOI] [PubMed] [Google Scholar]
- 5.Upadhyay A, Beuerman RW. Biological Mechanisms of Atropine Control of Myopia. Eye & contact lens. 2020;46(3):129–35. Epub 2020/01/04. doi: 10.1097/ICL.0000000000000677 cofounder and chief scientific officer at SinSaLabs. The remaining author has no funding or conflicts of interest to disclose. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chia A, Chua WH, Cheung YB, Wong WL, Lingham A, Fong A, et al. Atropine for the treatment of childhood myopia: safety and efficacy of 0.5%, 0.1%, and 0.01% doses (Atropine for the Treatment of Myopia 2). Ophthalmology. 2012;119(2):347–54. Epub 2011/10/04. doi: 10.1016/j.ophtha.2011.07.031 . [DOI] [PubMed] [Google Scholar]
- 7.Chua WH, Balakrishnan V, Chan YH, Tong L, Ling Y, Quah BL, et al. Atropine for the treatment of childhood myopia. Ophthalmology. 2006;113(12):2285–91. Epub 2006/09/26. doi: 10.1016/j.ophtha.2006.05.062 . [DOI] [PubMed] [Google Scholar]
- 8.Galvis V, Tello A, Parra MM, Merayo-Lloves J, Larrea J, Julian Rodriguez C, et al. Topical Atropine in the Control of Myopia. Medical hypothesis, discovery & innovation ophthalmology journal. 2016;5(3):78–88. Epub 2017/03/16. . [PMC free article] [PubMed] [Google Scholar]
- 9.Nti AN, Berntsen DA. Optical changes and visual performance with orthokeratology. Clinical & experimental optometry. 2020;103(1):44–54. Epub 2019/08/06. doi: 10.1111/cxo.12947 . [DOI] [PubMed] [Google Scholar]
- 10.<Pauline2011.Peripheral Refraction in Myopic Children Wearing__Orthokeratology and Gas-Permeable Lenses.pdf>.
- 11.Hiraoka T. Myopia Control With Orthokeratology: A Review. Eye & contact lens. 2022;48(3):100–4. Epub 2021/12/04. doi: 10.1097/ICL.0000000000000867 . [DOI] [PubMed] [Google Scholar]
- 12.Ji N, Niu Y, Qin J, Fu AC, Cui C. Orthokeratology Lenses Versus Administration of 0.01% Atropine Eye Drops for Axial Length Elongation in Children With Myopic Anisometropia. Eye & contact lens. 2022;48(1):45–50. Epub 2021/12/21. doi: 10.1097/ICL.0000000000000848 . [DOI] [PubMed] [Google Scholar]
- 13.Jiang J. Effect of orthokeratology, low concentration atropine and frame glasses on juvenile myopia prevention and control. International Eye Science. 2018;18(7):1349–52. doi: 10.3980/j.issn.1672-5123.2018.7.46 [DOI] [Google Scholar]
- 14.Ren QJ, Yue H, Wang P, Liu RJ, Lü P. Effects of low concentration atropine and orthokeratology on myopia prevention and control. International Eye Science. 2017;17(4):794–6. doi: 10.3980/j.issn.1672-5123.2017.4.57 [DOI] [Google Scholar]
- 15.Chen Z, Huang S, Zhou J, Xiaomei Q, Zhou X, Xue F. Adjunctive effect of orthokeratology and low dose atropine on axial elongation in fast-progressing myopic children-A preliminary retrospective study. Contact lens & anterior eye: the journal of the British Contact Lens Association. 2019;42(4):439–42. Epub 2018/11/30. doi: 10.1016/j.clae.2018.10.026 . [DOI] [PubMed] [Google Scholar]
- 16.Chen Z, Zhou J, Xue F, Qu X, Zhou X. Two-year add-on effect of using low concentration atropine in poor responders of orthokeratology in myopic children. The British journal of ophthalmology. 2022;106(8):1069–72. Epub 2021/03/13. doi: 10.1136/bjophthalmol-2020-317980 . [DOI] [PubMed] [Google Scholar]
- 17.Kinoshita N, Konno Y, Hamada N, Kanda Y, Shimmura-Tomita M, Kaburaki T, et al. Efficacy of combined orthokeratology and 0.01% atropine solution for slowing axial elongation in children with myopia: a 2-year randomised trial. Scientific reports. 2020;10(1):12750. Epub 2020/07/31. doi: 10.1038/s41598-020-69710-8 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kinoshita N, Konno Y, Hamada N, Kanda Y, Shimmura-Tomita M, Kakehashi A. Additive effects of orthokeratology and atropine 0.01% ophthalmic solution in slowing axial elongation in children with myopia: first year results. Japanese journal of ophthalmology. 2018;62(5):544–53. Epub 2018/07/06. doi: 10.1007/s10384-018-0608-3 . [DOI] [PubMed] [Google Scholar]
- 19.Luo Y, Luo WQ, Lu PF, Mao HJ, Wei DF, Huang YY. Clinical observation of orthokeratology combined with 0.01% Atropine for controlling low to moderate myopia in adolescents. International Eye Science. 2021;21(1):47–52. doi: 10.3980/j.issn.1672-5123.2021.1.09 [DOI] [Google Scholar]
- 20.Tan Q, Ng AL, Cheng GP, Woo VC, Cho P. Combined 0.01% atropine with orthokeratology in childhood myopia control (AOK) study: A 2-year randomized clinical trial. Contact lens & anterior eye: the journal of the British Contact Lens Association. 2022:101723. Epub 2022/06/03. doi: 10.1016/j.clae.2022.101723 . [DOI] [PubMed] [Google Scholar]
- 21.Tan Q, Ng AL, Choy BN, Cheng GP, Woo VC, Cho P. One-year results of 0.01% atropine with orthokeratology (AOK) study: a randomised clinical trial. Ophthalmic & physiological optics: the journal of the British College of Ophthalmic Opticians (Optometrists). 2020;40(5):557–66. Epub 2020/08/11. doi: 10.1111/opo.12722 . [DOI] [PubMed] [Google Scholar]
- 22.Tang WT, Tian M, Li SB, Yu Q. Clinical observation of low-dose Atropine combined with orthokeratology in the treatment of myopia. International Eye Science. 2020;20(6):1044–7. doi: 10.3980/j.issn.1672-5123.2020.6.26 [DOI] [Google Scholar]
- 23.Vincent SJ, Tan Q, Ng ALK, Cheng GPM, Woo VCP, Cho P. Higher order aberrations and axial elongation in combined 0.01% atropine with orthokeratology for myopia control. Ophthalmic & physiological optics: the journal of the British College of Ophthalmic Opticians (Optometrists). 2020;40(6):728–37. Epub 2020/09/06. doi: 10.1111/opo.12730 . [DOI] [PubMed] [Google Scholar]
- 24.Yu-Ling N, Ru-Shan Y, Ting-Ming D, Ling J, Jing-Yi N. Effect of low grade atropine combined with keratoplasty on middle grade myopia in adolescents. International Eye Science. 2019;19(11):1940–4. doi: 10.3980/j.issn.1672-5123.2019.11.29 [DOI] [Google Scholar]
- 25.Zhao Q, Hao Q. Clinical efficacy of 0.01% atropine in retarding the progression of myopia in children. International ophthalmology. 2021;41(3):1011–7. Epub 2020/11/19. doi: 10.1007/s10792-020-01658-0 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ (Clinical research ed). 2019;366:l4898. Epub 2019/08/30. doi: 10.1136/bmj.l4898 . [DOI] [PubMed] [Google Scholar]
- 27.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. European journal of epidemiology. 2010;25(9):603–5. Epub 2010/07/24. doi: 10.1007/s10654-010-9491-z . [DOI] [PubMed] [Google Scholar]
- 28.Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for non-randomized studies (minors): development and validation of a new instrument. ANZ journal of surgery. 2003;73(9):712–6. Epub 2003/09/06. doi: 10.1046/j.1445-2197.2003.02748.x . [DOI] [PubMed] [Google Scholar]
- 29.Huang J, Wen D, Wang Q, McAlinden C, Flitcroft I, Chen H, et al. Efficacy Comparison of 16 Interventions for Myopia Control in Children: A Network Meta-analysis. Ophthalmology. 2016;123(4):697–708. Epub 2016/02/02. doi: 10.1016/j.ophtha.2015.11.010 . [DOI] [PubMed] [Google Scholar]
- 30.Tsai HR, Wang JH, Huang HK, Chen TL, Chen PW, Chiu CJ. Efficacy of atropine, orthokeratology, and combined atropine with orthokeratology for childhood myopia: A systematic review and network meta-analysis. Journal of the Formosan Medical Association = Taiwan yi zhi. 2022. Epub 2022/06/11. doi: 10.1016/j.jfma.2022.05.005 . [DOI] [PubMed] [Google Scholar]
- 31.Li SM, Kang MT, Wu SS, Liu LR, Li H, Chen Z, et al. Efficacy, Safety and Acceptability of Orthokeratology on Slowing Axial Elongation in Myopic Children by Meta-Analysis. Current eye research. 2016;41(5):600–8. Epub 2015/08/04. doi: 10.3109/02713683.2015.1050743 . [DOI] [PubMed] [Google Scholar]
- 32.Kakita T, Hiraoka T, Oshika T. Influence of overnight orthokeratology on axial elongation in childhood myopia. Investigative ophthalmology & visual science. 2011;52(5):2170–4. Epub 2011/01/08. doi: 10.1167/iovs.10-5485 . [DOI] [PubMed] [Google Scholar]
- 33.Wan L, Wei CC, Chen CS, Chang CY, Lin CJ, Chen JJ, et al. The Synergistic Effects of Orthokeratology and Atropine in Slowing the Progression of Myopia. Journal of clinical medicine. 2018;7(9). Epub 2018/09/13. doi: 10.3390/jcm7090259 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen Z, Niu L, Xue F, Qu X, Zhou Z, Zhou X, et al. Impact of pupil diameter on axial growth in orthokeratology. Optometry and vision science: official publication of the American Academy of Optometry. 2012;89(11):1636–40. Epub 2012/10/03. doi: 10.1097/OPX.0b013e31826c1831 . [DOI] [PubMed] [Google Scholar]
- 35.Ye L, Shi Y, Yin Y, Li S, He J, Zhu J, et al. Effects of Atropine Treatment on Choroidal Thickness in Myopic Children. Investigative ophthalmology & visual science. 2020;61(14):15. Epub 2020/12/16. doi: 10.1167/iovs.61.14.15 Zhu, None; X. Xu, None. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li W, Jiang R, Zhu Y, Zhou J, Cui C. Effect of 0.01% atropine eye drops on choroidal thickness in myopic children. Journal francais d’ophtalmologie. 2020;43(9):862–8. Epub 2020/08/24. doi: 10.1016/j.jfo.2020.04.023 . [DOI] [PubMed] [Google Scholar]
- 37.Hiraoka T, Kakita T, Okamoto F, Takahashi H, Oshika T. Long-term effect of overnight orthokeratology on axial length elongation in childhood myopia: a 5-year follow-up study. Investigative ophthalmology & visual science. 2012;53(7):3913–9. Epub 2012/05/12. doi: 10.1167/iovs.11-8453 . [DOI] [PubMed] [Google Scholar]
- 38.Gong Q, Janowski M, Luo M, Wei H, Chen B, Yang G, et al. Efficacy and Adverse Effects of Atropine in Childhood Myopia: A Meta-analysis. JAMA Ophthalmol. 2017;135(6):624–30. Epub 2017/05/12. doi: 10.1001/jamaophthalmol.2017.1091 ICMJE Form for Disclosure of Potential Conflicts of Interest and none were reported. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu YM, Xie P. The Safety of Orthokeratology—A Systematic Review. Eye & contact lens. 2016;42(1):35–42. Epub 2015/12/26. doi: 10.1097/ICL.0000000000000219 . [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOC)
(DOC)
(DOC)
Data Availability Statement
All relevant data are within the manuscript and its Supporting information files.