Abstract
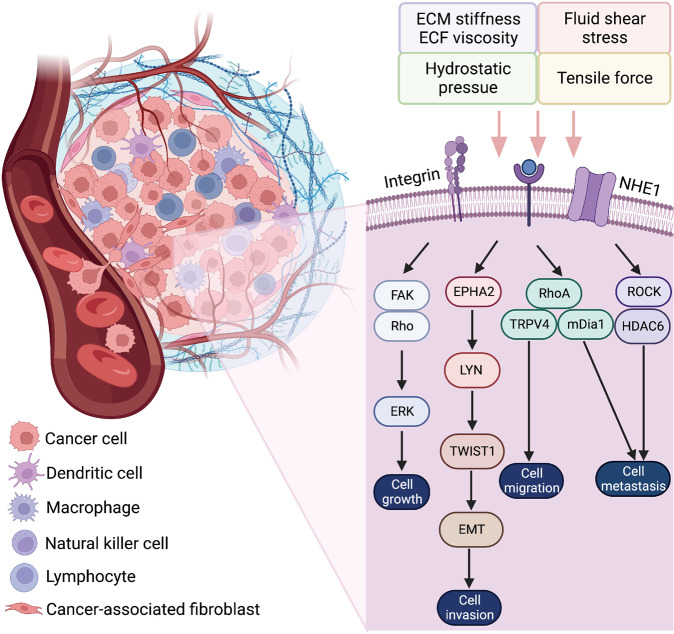
Cellular mechanotransduction, a critical regulator of numerous biological processes, is the conversion from mechanical signals to biochemical signals regarding cell activities and metabolism. Typical mechanical cues in organisms include hydrostatic pressure, fluid shear stress, tensile force, extracellular matrix stiffness or tissue elasticity, and extracellular fluid viscosity. Mechanotransduction has been expected to trigger multiple biological processes, such as embryonic development, tissue repair and regeneration. However, prolonged excessive mechanical stimulation can result in pathological processes, such as multi-organ fibrosis, tumorigenesis, and cancer immunotherapy resistance. Although the associations between mechanical cues and normal tissue homeostasis or diseases have been identified, the regulatory mechanisms among different mechanical cues are not yet comprehensively illustrated, and no effective therapies are currently available targeting mechanical cue-related signaling. This review systematically summarizes the characteristics and regulatory mechanisms of typical mechanical cues in normal conditions and diseases with the updated evidence. The key effectors responding to mechanical stimulations are listed, such as Piezo channels, integrins, Yes-associated protein (YAP) /transcriptional coactivator with PDZ-binding motif (TAZ), and transient receptor potential vanilloid 4 (TRPV4). We also reviewed the key signaling pathways, therapeutic targets and cutting-edge clinical applications of diseases related to mechanical cues.
Subject terms: Cancer microenvironment, Molecular biology, Cell biology, Respiratory tract diseases, Cancer
Introduction
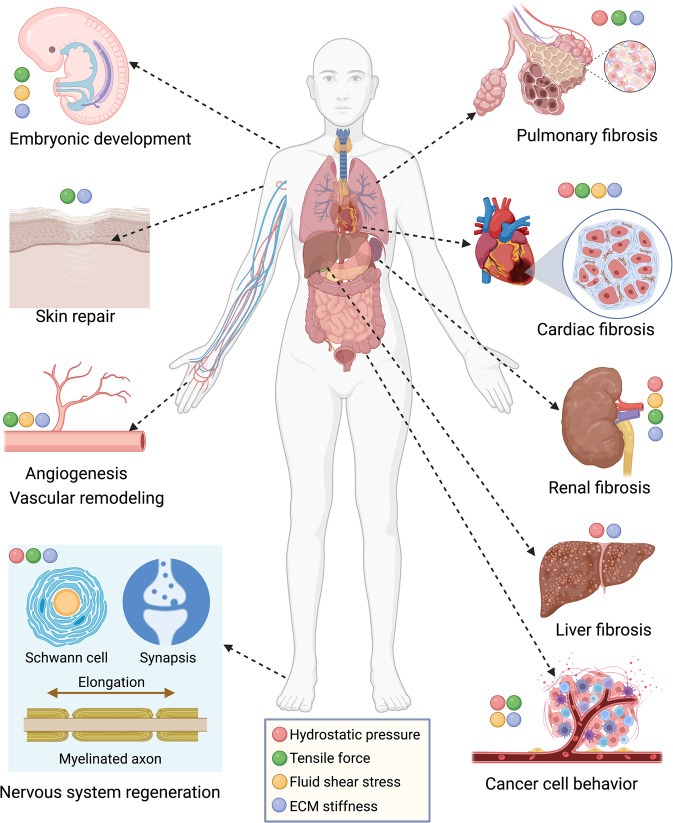
Cellular mechanotransduction is an important biological process in living organisms. It was first studied based on the Wolff’s Law on mechanical load in tissue homeostasis and has extended to the growth and development of tissues and organisms.1 To date, the study of cellular mechanotransduction is being investigated for its impact of multiple mechanical cues on multiple pathophysiological processes, including embryonic development,2 tissue repair and wound healing,3 neural regeneration,4 fibrosis,5 tumorigenesis,6 and cancer immunotherapy resistance7 (Fig. 1). Many ongoing studies are focusing on the mechanism and therapeutic targets for mechanical cue-induced tissue homeostasis, and corresponding diseases.
Fig. 1.
Global overview of the mechanical cue-associated pathophysiological processes. The main components of biomechanical cues engage in several biological processes and diseases, such as pulmonary fibrosis, cardiac fibrosis, renal fibrosis, liver fibrosis, cancer cell behaviors, embryonic development, skin and wound repair, angiogenesis and vascular remodeling, and nervous system regeneration. ECM extracellular matrix. This figure was created using Biorender.com
Biological mechanical cues mainly include hydrostatic pressure (HP), tensile force (TF) or stretching force, fluid shear stress (FSS), extracellular matrix (ECM) stiffness or tissue elasticity, and extracellular fluid (ECF) viscosity. These mechanical cues are involved in the communications in cell–ECM, cell hemodynamics, and cell–ECM–cell crosstalk.5 However, the crosstalk between the normal tissue homeostasis and lesions is also closely associated with mechanical stimulation, which restricts the investigation of therapeutic targets. Hence, finding strategies for balancing the mechanical cues in a normal range is of great importance.
Cells can sense the mechanical cues and respond to the changes in the biomechanical environment. The functional ion channels and receptors on the cell membrane can sense the biomechanical signals, and trigger changes in cytoskeleton structure and downstream biochemical signaling cascades.8 The nuclear membrane can also sense the alteration of cytoskeleton, thereby influencing downstream gene transcription. An intricate regulatory network of the cellular mechanotransduction process has already been established. These signaling pathways extensively intervene in the pathophysiological process of the human body.
Recent studies on the biological function of cellular mechanotransduction have provided a broader view of the interaction between mechanical cues and biological processes. However, the regulatory mechanisms of mechanical cues are complex and how these mechanical cues interact with each other is still poorly studied. Meanwhile, many signaling pathways have been identified without fully illustrating the cell function alteration, including cytoskeleton swelling and contraction, cell polarization, and cell adhesion. The understanding of cellular response to mechanical cues can assist in determining the precise approach targeting cellular mechanotransduction.
There have been several reviews investigating the communication between mechanotransduction and diseases. Nevertheless, these reviews almost focus on specific mechanical situations, technology tools or diseases, lacking discussion of therapeutic intervention and clinical application. In this review, we intend to summarize the characteristics and molecular mechanisms of cellular mechanotransduction in normal conditions and diseases with an updated and comprehensive interpretation. In addition, the mechanosensitive effectors, signaling pathways and clinical applications targeting mechanical cues-induced diseases are presented in light of the most recent advances.
Characteristics and mechanisms of cellular mechanotransduction
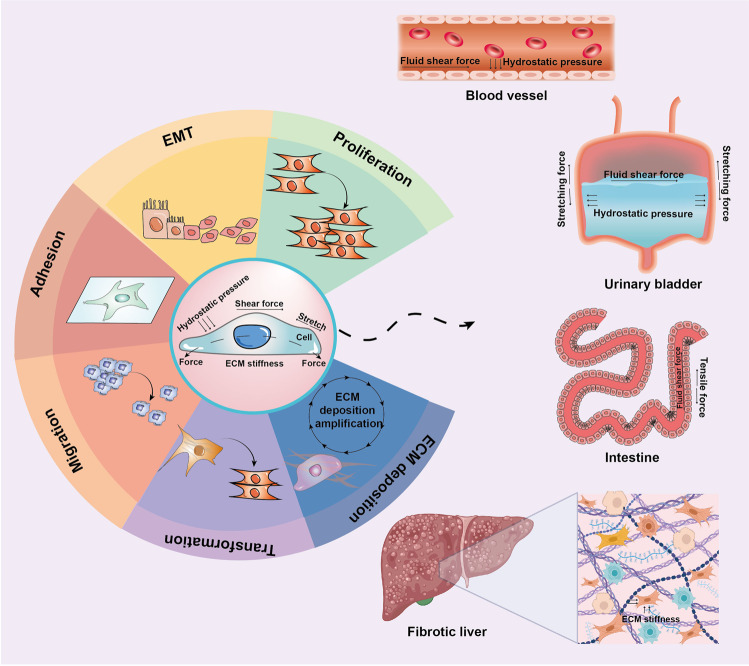
HP, FSS, TF, and ECM stiffness are the main biological mechanical cues affecting cell-matrix communications and signal transduction. These mechanical cues regulate multiple biological processes, including cell development, transformation, differentiation, adhesion, migration, proliferation, and ECM generation (Fig. 2). Mechanical cues can also trigger sophisticated biological modulated network-dependent tissue and organ development, regeneration, repair, tumorigenesis, tumor invasion, and metastasis. Herein, we focus on the molecular biology of these mechanical cues.
Fig. 2.
Cellular mechanotransduction in tissues and organs. Typically, fluid shear force and hydrostatic pressure exist in blood vessels. Tensile force, hydrostatic pressure, and fluid shear force function in the urinary bladder. The fluid shear force and tensile force function in the intestine. The ECM stiffness functions in fibrotic liver. ECM extracellular matrix, EMT epithelial–to-mesenchymal transition. This figure was partly created using Biorender.com
Hydrostatic pressure
HP generally exists in tissues and organs with fluids, such as blood vessels, heart, eye, joint cavity, and urinary bladder. The mechanical cues in hollow organs include HP, sinusoidal stress, and interstitial fluid pressure.9 HP in the interstitial cavity is approximately −4 cmH2O10; in solid tumors and edematous tissues, the pressure is 25–40 cmH2O11; in the liver, an increase of intrahepatic resistance induces augments in the pressure gradient between the inferior vena cava and the portal vein (>5 mmHg).12
HP in the normal range, also being classified as physiological HP, can promote the development and repair of tissues. Studies have revealed that periodic HP promotes bone growth and organization in developmental models.13,14 However, HP beyond the normal range, also being classified as pathological HP, can lead to decompensated lesions in tissues and organs, such as bladder fibrosis and decreased sperm quality.15 Osteogenesis and bone density can also be enhanced by HP.13 Additionally, HP can change the conformation of ion channels and regulate ion transmembrane transportation,16 thereby affecting pathophysiological processes. For example, HP induces the opening of transmembranal channels in Hela cells, resulting in an influx of calcium ions (Ca2+).17 As a mechanosensitive ion channel protein, Piezo1 can respond to HP, activate mitogen-activated protein kinases (MAPK) and p38 signaling pathways, and facilitate the expression of bone morphogenetic protein 2 (BMP2) to affect the phenotype of mesenchymal stem cells.18 Furthermore, high HP (40 mmHg) promotes atrial electrophysiological remodeling and inflammatory response by regulating the ion flow, reducing atrial fibrillation.19
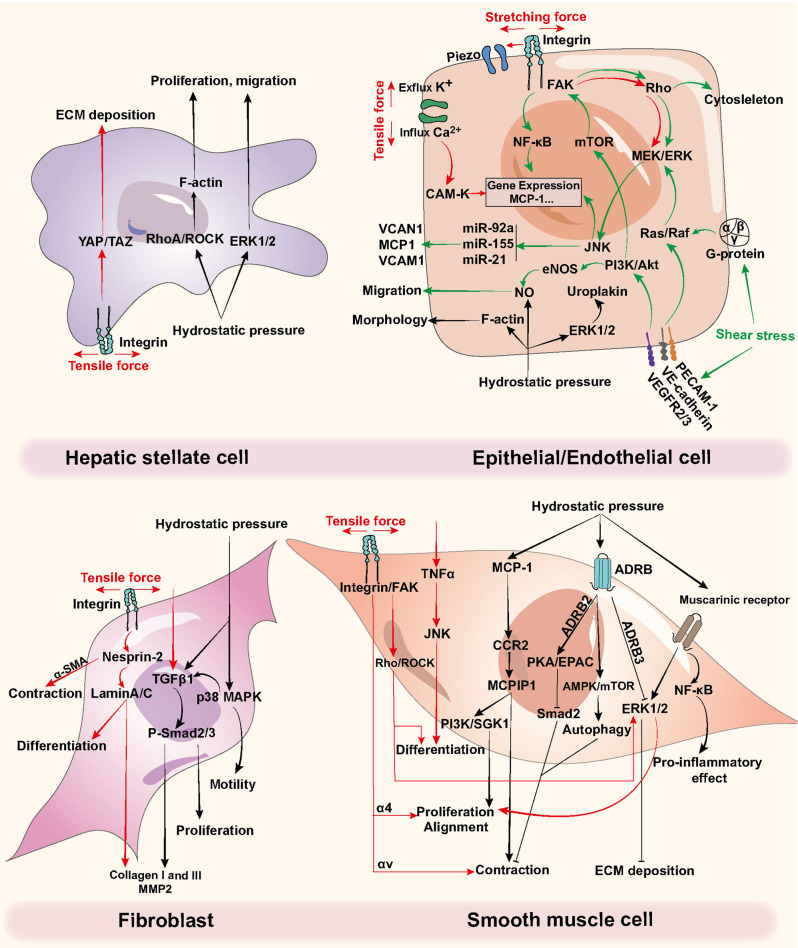
HP signals transduce through various functional proteins and signaling pathways (Fig. 3). For parenchymal cells such as hepatocytes and hepatic stellate cells (HSCs), the subcapsular HP affects the biochemical processes.20 The cytoskeleton-related signals (i.e., RhoA, ROCK, α-SMA) are enhanced by 50 mmHg of HP on HSCs. The increase of interstitial fluid pressure activates the HSCs to facilitate fibrosis progression.21 Moreover, in hollow organs such as the urinary bladder, the uroplakins (Ia/Ib/II/III) on the epithelial cell membrane, which play a pivotal role in cell differentiation,22,23 are the main components of the high-resistance barrier of urinary bladder urothelium.24 Intravesical sustained HP above 40 cmH2O is generally known as pathological pressure and is a potential cause of fibrosis.25 It has been reported that 200 cmH2O HP can facilitate the expression of uroplakin Ia and uroplakin II protein in urothelial cells, which are the key factors for extracellular signal-related kinase (ERK) 1/2 pathway activation.26
Fig. 3.
Regulatory mechanisms of tensile force, hydrostatic pressure, and shear stress on different cell types. Red arrows refer to stretching force, green arrows refer to shear stress, and black arrows refer to hydrostatic pressure. ADRB β adrenoceptors, ECM extracellular matrix, ERK extracellular signal-related kinase, MCP1 monocyte chemotactic protein 1, MCPIP1 MCP1-induced protein, MMP matrix metalloprotease, mTOR mammalian target of rapamycin, NF-κB nuclear factor-κB, TGFβ transforming growth factor β, TNFα tumor necrosis factor α, VCAM-1 vascular cell adhesion molecule 1
All the mechanosensitive parenchymal cells and nonparenchymal cells (i.e., fibroblasts and immune cells) serve as effectors responding to HP. For example, endothelin-1 (ET-1) and nitric oxide are increased under HP and identified as novel insights for vascular remodeling.27 The M3 muscarinic receptor subtypes are mainly distributed in the neck and dome region of the urinary bladder. M2 and M3 muscarinic receptors are involved in the cholinergic transmission process in the human detrusor muscle or dome.28,29 Pathological HP also induces atrial fibroblast proliferation and collagen deposition through the transforming growth factor β1 (TGFβ1)/Smad3 pathway.30 Similarly, the motility and proliferation of fibroblast are promoted by increased HP-induced p38 MAPK cascades.31,32
Fluid shear stress
The intra-organ or intra-capsule fluid can both generate FSS by the fluid flow. Typical FSS exists in human vasculature (i.e., vessel bifurcations, stenosis, aortic aneurysms, heart valves, and capillary networks), including shear and extensional flow.33 The shear flow is classified as laminar flow and turbulent flow influenced by the structure of the lumen. Uniaxial extensional (elongational) flow is the flow acceleration parallel to the vascular wall. Extensional stress is common in regions with sudden contractions or expansions of fluid flow. Stable flow or laminar flow functions in anti-inflammation, anti-adhesion, and anti-thrombosis in the vascular wall.34 However, persistent turbulent flow in the vascular wall can increase the endothelial permeability (i.e., junctional proteins alteration) and proinflammatory signaling (i.e., nuclear factor κB [NF-κB] signaling, adhesion molecules activation) to promote the formation of lesions.35,36
FSS determines the tissue homeostasis such as blood vessels, the heart, the airway, and the urinary bladder37–39 (Fig. 3). The frictional forces generated by the bloodstream, also known as the wall FSS, can lead to the luminal stress of the vascular wall.40 High FSS can induce anti-inflammatory effects, such as Klf2/4 or endothelial nitric oxide synthase (eNOS). The turbulent flow, oscillatory, and low FSS trigger pro-inflammatory responses. In the circulatory system, the bloodstream is generated by the heart contract, which produces FSS. In this situation, FSS is determined by wall shear rate and blood viscosity.41 FSS in large blood vessels and arterioles are approximately 10 dyn/cm2 and 50 dyn/cm2, respectively.42 Many studies have shown that FSS facilitates epithelial cells and triggers atherosclerosis, lipid deposition, and vessel wall thickening,43 while the endothelial cells remain polygonal in shape and unorganized in turbulent flow or low FSS (≤5 dyn/cm2).44
The blood flow-induced hemodynamic changes regulate multiple signaling pathways in various vascular wall cells.45–47 The recently reported signaling pathways related to the FSS mainly include vascular endothelial growth factor (VEGF), Notch, PDGF, Klf2, eNOS, endothelin, Rho family signaling molecules of TGFβ/BMP/Smad pathway, MAPK signaling pathway, NF-κB signaling pathway, and GTPase signaling pathway.48,49 When the cells receive the FSS mechanical signals, several mechanosensors will be triggered, including integrins, the glycocalyx, primary cilia, G-protein-coupled receptors, and ion channels (K+, Ca2+).50–57 Piezo channels are important sensors for mechanical stimulation. Piezo1 initially senses SS and transmits biomechanical signals to the nucleus to promote nuclear contraction.58 In response to FSS, several mechanosensory complexes are also activated, including vascular endothelial cell cadherin (VE-cadherin), VEGF receptor 2 (VEGFR2), and platelet endothelial cell adhesion molecule (PECAM-1, or CD31). As a result, the mechanosensory complexes transmit the biomechanical signal into the endothelial cell.55 PECAM-1 activates Src, and binds to type III intermediate filament, in which process VE-cadherin serves as an adapter.59 In addition, G-protein-coupled receptor is also triggered to enhance Ras and Rho GTPase signaling cascades.60 The enhanced Rho activity increased endothelial cell migration through FSS and modulated the traction force.61 In addition, PI3K/AKT/mammalian target of rapamycin (mTOR) can also be activated by VEGFR2, which ultimately induces integrin activation.62 A recent study concluded that β integrin, a specific sensor of unidirectional FSS but not bidirectional, drives the endothelial cell alignment and downstream cascade.63 The integrin/NF-κB-associated cascades and adhesion of endothelial cells are initiated by FSS.64 Furthermore, the long-term presence of NF-κB and other inflammatory molecules increases the expression of intercellular adhesion molecule (ICAM-1) and vascular adhesion molecule (VCAM-1), further recruiting monocytes to aggravating epithelial inflammation and inducing atherosclerosis under FSS.65 SH2-containing protein tyrosine phosphatase-2 (SHP-2) has been reported to bind to PECAM-1 to activate the ERK1/2 cascades.66 In the hydrodynamic model, the carbon monoxide synthesis increases, and endothelial cell structure is remodeled by FSS.27 It has also been reported to promote endothelial cell migration through Tie2/PI3K/AKT/eNOS pathway.67
Apart from the functional proteins and signaling pathways, noncoding RNAs (ncRNAs) also serve as sensors of FSS. ncRNA is essential in the gene-regulatory process by pairing to the mRNAs and is composed of small and long noncoding RNAs.68 In addition, the response of ncRNAs to biomechanical signal is involved in physiological changes in atherosclerosis, atherogenesis,69 various cardiovascular diseases,70 and coronary syndromes.71–73 Similarly, some small RNAs also engage in mechanotransduction to promote diseases progression. Small RNAs include microRNAs (miRNAs), PIWI-interacting RNAs (piRNAs), and short-interfering RNAs (siRNAs).74 Some miRNAs have been identified as mechanical cues-induced reactions, including miR-10a, miR-19a, miR-23b, miR-101, and miR-143/145.75,76 For example, atherogenesis is promoted by mechanical stimuli-induced miRNA-associated signaling pathways in endothelial cells. Several miRNAs have been illustrated in atherosclerosis, such as miR-17-3p, miR-92, miR-126, miR-712, miR-205, miR-143, miR-145, miR-31, and others.77 Novel therapeutic approaches targeting ncRNAs and miRNAs are necessary for disease treatment.
Tensile force
TF, also known as stretching force, is important in muscle and joint movement, atherogenesis, cardiovascular remodeling,78,79 and cell behaviors (i.e., proliferation, transformation, and development).80 For instance, the dynamic tension of the joint can affect the final behaviors of muscles.81 In the cardiovascular system, endothelial cells, smooth muscle cells, and cardiac myocytes are the principal effector cells responding to TF generated by blood flow. TF can induce cardiomyocyte hypertrophy through nuclear factor-like 2 (Nrf2) and interferon-regulated transcription factors in myocardial tissue.82 In the vascular wall, TF promotes vascular remodeling and contraction.83 Besides, TF can induce urothelium proliferation through α6-focal adhesion kinase (FAK) signaling.84 It is also a factor leading to the development of animal neurons by regulating gene transcription.85,86 Hence, a comprehensive interpretation of the TF in the human body is of great significance.
The magnitude, frequency, and duration of the TF can affect the alignment, differentiation, migration, proliferation, survival, apoptosis, and autocrine and paracrine functions of cells (Fig. 3). Once the integrin receives the biomechanical signals, it will transduce the signals into cells. Then the p38MAPK signaling, NO, and reactive oxygen species (ROS) are activated to trigger downstream cascades.87,88 Meanwhile, the α-smooth muscle actin (α-SMA) expression and promoter activities are enhanced through JNK and p38MAPK signaling pathways.89 Rho also responds to integrins to activate ERK signaling.90 Ultimately the phenotype and alignment of cells are regulated.
The nuclear envelope is also an important effector of cellular mechanotransduction. The nucleus membrane senses TF and reacts more rapidly than common biochemical transduction. The interactions between the nuclear envelope and actin have been reported in nuclear migration,91 cell polarization,92 nuclear deformation,93 Piezo1/endoplasmic reticulum (ER) response,94 chromosomes gathering,95 and chromatin organization.96 Significantly, lamins located in the nuclear envelope can form interacting meshwork with a highly branched structure97,98 and modulate gene transcription, DNA replication, and chromatin organization.99 Lamins also regulate the nucleus assembly, nuclear shape, stiffness, and structure in cytoskeleton arrangement.100,101 The nesprins interact with laminA/C to transmit biomechanical signals into the nucleus. When Nesprin2 is inhibited, the cell transformation process and collagen synthesis are blocked.102 During this process, the mechanosensitive pathways in mature tissues do not respond to intercellular tension. Thus, these signaling pathways do not influence fibroblast differentiation in subsequent fibrosis.103 It is worth noting that different TF intensities induce various effects on the chondrocyte mechanotransduction process. Mechanically activated ion channel transient receptor potential vanilloid 4 (TRPV4)-mediated Ca2+ signaling is significant in response to the physiological strain levels (3% and 8%). However, Piezo2-mediated Ca2+ signaling is substantial in response to pathological high strain levels (18%).104
Like FSS, miRNAs are involved as post-transcriptional regulators in the bone remodeling process and function in cell differentiation in the orthodontic tooth movement process. For instance, it has been confirmed that the miRNA-21 deficiency inhibits orthodontic tooth movement and promotes the remodeling of bone in mice.105 Cyclic stretch activates alkaline phosphatase (ALP) and osteogenesis biomarkers such as osteopontin (OPN), runt-related transcription factor 2 (RUNX2), type I collagen, ALP, osteocalcin, and osterix.106 The ALP activity is inhibited by miR-146a and miR-34a targeted CUGBP Elav-like family member 3 (Celf3), which can regulate the life cycle of mRNAs from transcription to translation.106–108
ECM stiffness
ECM, a non-cellular component presented in extracellular capsules, is a complex that mainly consists of collagen, fibronectin, elastin, lamin, proteoglycan, glycoprotein, and glycosaminoglycan.109 Fibrillar proteins (i.e., collagens) are featured by high tensile strength but low elasticity. However, elastic fibers (i.e., elastin) are featured by high elasticity and low tensile strength. Therefore, the components of ECM greatly affect the tissue mechanical properties.110
ECM components are critical in cell growth, differentiation, and apoptosis. The cells communicate via the intracellular skeleton and cell surface adhesion molecules with the ECM and neighboring cells. These properties of ECM are also engaged in the occurrence and development of diseases. ECM remodeling is associated with complicated physiological conditions. Pathological ECM has a prolonged impact on the morphology and functions of cells and forms an amplification effect to strengthen the ECM deposition process further, resulting in severe fibrosis.111 Excessive deposition of ECM can lead to even worse results. For example, myocardial fibrosis is developed from the fibrotic scar due to myocardial infarction and matrix deposition in the interstitial and perivascular areas. It causes heart function impairment and accelerates the progression of heart failure.112 The available evidence demonstrated a predominant increase in type I and type III collagen fibers observed in hypertensive heart disease and aortic stenosis-associated heart failure.113,114 Moreover, liver fibrosis has been indicated to be triggered by myofibroblasts from chronic hepatotoxic injury (i.e.,hepatitis HBV or hepatitis HCV, alcohol abuse) or cholestatic injury (i.e., bile duct obstruction). The HSCs-secreted ECM promotes transformation from fibroblasts to myofibroblasts.115,116 As a result, the normal liver tissues were replaced by the cross-linked type I and type III collagen fibers.
The mechanical properties of ECM are also critical in biological processes. ECM stiffness indicates the resistance to the deformation of tissue and induces an increase in tissue elasticity. Apart from the normal deposition of ECM, post-translational modifications of ECM components also modulate stiffness. For instance, the nonenzymatic glycation and cross-linking of collagen can increase the elasticity of the matrix. The stiffness of tissues varies in different organs. For example, normal lung tissues have Young’s Modulus of approximately 1 kPa required for respiration.117 However, lung elasticity increases to 30–50 kPa in idiopathic pulmonary fibrosis (IPF) patients due to excessive ECM deposition and contractile myofibroblasts transformation.118 Besides, the normal liver tissue represents a stiffness of 1.5–4.5 kPa, and that in the early and late stages of liver fibrosis are 4.1–12.9 and 16.3–48 kPa, respectively.119–121
ECM stiffness is involved in multiple pathophysiological processes. In the central nervous system, neurons tend to live in an environment with soft stiffness while stiff substrates are required by glial cells.122,123 In addition, pathological ECM stiffness triggers multiple organ fibrosis,64,124,125 vascular smooth muscle disorders,126 benign pancreatic diseases,127 osteoblast differentiation,128 tumor development, invasion and metastasis.129–134 Studies have shown that cancer-associated fibroblasts (CAFs) can change the composition and physicochemical properties of ECM, thereby regulating tumor metastasis and adjusting the metastasis path.135,136
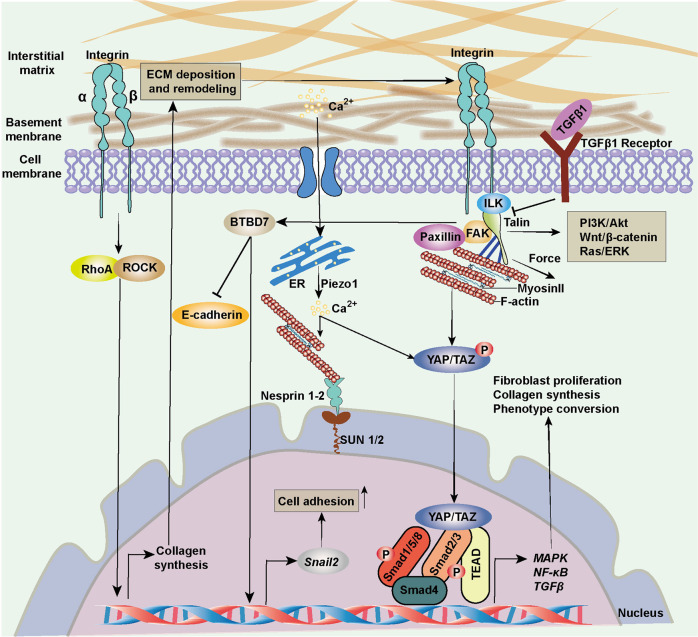
Mechanistically, increasing tissue elasticity activates the integrins, which belong to the cell surface receptors family, to enhance cell-cell communication (Fig. 4). The integrins are abundant in fibroblasts and can receive the signals from ECM components, convey mechanical and biochemical signals into cells, and facilitate cell proliferation, differentiation, migration, and invasion.137–140 Once the integrins are activated, the RhoA/ROCK pathway is initiated, enhancing collagen and fibronectin accumulation.141,142 The transcriptional regulator BTBD7 is activated to trigger epithelial-to-mesenchymal transition (EMT) through Snail2 while E-cadherin is inhibited, and hence the cell adhesion is weakened.143 Meanwhile, talin binds to the cytoplasmic tail of the β integrin and facilitates the assembly of F-actin to promote signal transduction.138,144,145 The FAK also interacts with integrins and triggers downstream cascades in fibroblast phenotype conversion.146,147 The integrin-linked kinase (ILK) and paxillin have also been reported to be essential linkages of integrin-mediated fibrosis.148 The actin connects with myosin II and conveys the biomechanical signals to the nucleus. In this process, Yes-associated protein (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ) are translocated into the nucleus to promote transcription of downstream genes in cell proliferation, collagen synthesis, and cell differentiation. This action inversely enhances ECM stiffness. Significantly, YAP/TAZ-mediated ECM stiffness-induced biochemical transduction is independent of Hippo signaling cascades and the large tumor suppressor kinases.149 TGFβ, Wnt/β-catenin, MAPK/ERK, and NF-κB signaling pathways also engage in this process.150–152 For example, in liver cancer cells, ECM stiffness can activate TGFβ by regulating the cytoskeletal tension induced by the integrin-FAK-Rho GTPase pathway.133 In addition, ECM stiffness regulates mesenchymal stem cell differentiation through the Wnt/β-catenin signaling pathway.153 Reducing ECM stiffness can promote M2 macrophage activation and enhance peroxisome proliferator-activated receptor gamma expression.154 And pathological ECM stiffness can up-regulate Ca2+ ion channels, improve intracellular Ca2+ concentration, and ultimately activate the ERK signaling pathway to promote cell proliferation and vascular remodeling.155 Intriguingly, a recent study reported that the curved nanofibers in the ECM microenvironment facilitated cell proliferation and osteogenic differentiation.156 Differing from a straight fiber network in forming continuous adhesion, a curved fiber structure tends to trigger discrete adhesion. Therefore, the cell bridge forming function of curved matrix supplements the current research in ECM stiffness.
Fig. 4.
Cellular mechanotransduction of ECM stiffness. The integrins convey mechanical and biochemical signals from ECM into cells and facilitate cell proliferation, differentiation, migration, and invasion. The RhoA/ROCK pathway is activated, enhancing collagen and fibronectin accumulation. Talin/FAK facilitates the assembly of F-actin to promote signal transduction. The actin connects with myosin II and conveys the mechanical cues to the nucleus. YAP/TAZ is translocated into the nucleus to promote the transcription of downstream genes, collagen synthesis, and cell differentiation. ECM extracellular matrix, ER endoplasmic reticulum, ERK extracellular signal-related kinase, FAK focal adhesion kinase, ILK integrin-linked kinase, P phosphate, TGFβ transforming growth factor β
The mechanical cues are transduced through the cytoskeleton in a much faster manner than that through biochemical signaling. The nuclear envelope connects with the actin between the cytoskeleton and nucleoskeleton.157,158 The transcriptional alteration better responds to nucleoskeleton change with the assistance of lamins, emerin, and transcriptional regulators.159–161 The modifications of ECM and cytoskeleton trigger the mechanotransduction in the cytoplasm and nucleus network to proceed regulation of cell function.162
ECF viscosity
A recent study identified a novel mechanical cue, ECF viscosity, which could interact with ECM stiffness to induce cell migration and substrate mechanotransduction. The macromolecules in the crowded ECF environment determined the density, osmotic pressure, and viscosity. Intuitively, high ECF viscosity declines the motility of various cell types. However, Konstantopoulos et al. found that cell migration and cancer dissemination are facilitated by increased ECF viscosity.163 The crosstalk between ECF and cells induces the actin-related protein 2/3 (ARP2/3)-complex-dependent actin network, which triggers Na+/H+ exchanger 1 (NHE1) polarization. NHE1, an isoform of NHE, is a membrane transporter that exchanges intracellular proton for extracellular Na+.164 The activation of NHE1 promotes cell swelling and increases cell tension, then facilitates TRPV4-mediated Ca2+ influx. Subsequently, RhoA-dependent contractility is increased, thereby enhancing the motility of cells. High ECF viscosity increases the motility of cells on two dimensional (2D) surfaces. ECF viscosity has also been confirmed to promote cell spreading dynamics based on integrin/YAP signaling.165 High ECF viscosity triggers integrin-based adhesion, enhancing cell migration. ECF can also interact with ECM stiffness to improve cellular mechanotransduction. Despite these, the mechanisms of the impact of ECF viscosity on cells are largely understudied. The recent studies have provided a highlight for future focus on the mechanosensing process.
In the ECM microenvironment, the cells interacting with a reconstituted three-dimensional (3D) environment have a lower level of actin-associated filamin and tension than that with a 2D system, which unfolds the filamin and interacts more with ER.166 Moreover, a rigid 2D substrate induces more integrins than a 3D matrix.167 Although the 2D system simulates ECM mechanical cues to a large extent and has long been recognized as a classical foundation,168 the 3D system with more compliable components is still preferred for research in the future. The mechanical cues function synergistically or organ-specifically in various all stages of diseases. Therefore, investigating mechanical cues in normal conditions and diseases is essential and provides insights for therapies targeting multiple diseases.
Critical effectors in mechanical signaling
Mechanical effectors have been widely investigated in recent years, mainly including ion channels,169–178 receptors,179–181 integrins,182,183 and transcriptional factors.184–187 These sensors behave differently in the mechanotransduction process and some mechanisms are still poorly elaborated. They function dependently or cooperatively and the interactive networks are sophisticated in regulating cell function. Hence, several typical and highlighted sensors are depicted in the current review.
Piezo channels in mechanotransduction
Piezo proteins (Piezo1 and Piezo2) are mechanosensitive ion channels encoded by two genes at chromosomes 16 and 18 in humans. Piezo proteins are initially identified as evolutionary conserved mechanically activated cation channels, producing the most stable current under mechanical stimulation.188,189 Human transmembrane Piezo1 consists of 2521 amino acids, and Piezo2 consists of 2752 amino acids with a molecular weight of about 300 kDa.190,191 The Piezo channels with three kinetic states (open, closed, and inactivated) are responsible for converting the mechanical cues into biochemical signals.192,193 It has been proved that Piezo1 senses bilayer tension in bleb membranes that can be modulated by cytoskeletal proteins and ECM stiffness.194–196 Piezo1 selectively conducts cations including K+, Na+, Mg2+, and especially Ca2+, while Piezo2 indicates non-selective properties in ions conduction.188,197
Piezo channels function in the cardiovascular, gastrointestinal, nervous, respiratory, exercise, and urinary systems of the human body198–225 (Table 1). Piezo channels have been reported to be related to several pathophysiological processes, including erythrocyte volume regulation,201 cell division,226 cell migration,227 and innate immunity.228 Piezo channels are presented in various mechanically sensitive cells coupling Ca2+ transmembrane conversion.229 In addition, Piezo1 is mainly associated with Ca2+ signaling, and Piezo2 functions in touching sensation, tactile pain, balance, respiration, blood pressure, and urinary bladder fullness.218,230–232 For instance, the ablation of Piezo2 results in impaired bladder filling in humans and mice.221,225 In the innate immune system, immune cells respond to the local environment and physiological changes, including temperature, pH, oxygen, and nutrition. Mechanical cues are highlighted as contributing factors of immune cell activation.228 In the orthodontic tooth movement mice model, Piezo1/AKT/Ccnd1 is essential in bone marrow-derived macrophage proliferation.233
Table 1.
Functions of Piezo channels in cellular mechanotransduction
| Type | Target | Mechanical stimulation | Mechanism | Reference |
|---|---|---|---|---|
| Piezo1 | Vascular endothelium development | Shear stress | Ca2+influx→MTP-MMP signaling→focal adhesion and endothelial cell sprout formation; | 232 |
| Vascular tone | Shear stress | Ca2+influx→G-coupled endothelial adrenomedullin receptor→cAMP→ eNOS→NO; Ca2+influx→ATP→PI3K/AKT→eNOS→NO | 198–200 | |
| Vascular remodeling | Stretch | Ca2+influx→transglutaminase →ECM remodeling | 222 | |
| Erythrocytes | Shear stress | Ca2+influx→K+ efflux→red blood cells dehydration | 201 | |
| Erythrocytes | Shear stress | Ca2+influx→pannexin-1→ATP release | 202 | |
| Nervous system | Traction force | Ca2+influx→neural differentiation →neuron-astrocyte interaction | 203,204 | |
| Gastric mucosa | Antrum distension | Activated G cells→gastrin secretion | 205 | |
| Lung endothelium | Shear stress | Ca2+influx→calpain→Src cleavage→stabilization of adherens junctions | 206 | |
| Lung endothelium | Hydrostatic pressure | Ca2+influx→calpain→disruption of adherens junctions | 207 | |
| Aoveoli | Stretch | Ca2+influx→Bcl-2 pathway→type II epithelial cells apoptosis | 208 | |
| Urinary bladder | Stretch | Ca2+influx→ATP→attenuate storage disorders | 209 | |
| Tumor | ECM stiffness | YAP-Piezo1→proliferation; Ca2+influx→AKT/mTOR phosphorylation→proliferation; Piezo1–mitochondrial calcium uniporter–HIF-1α–VEGF axis→metastasis | 210,211,213 | |
| Piezo2 | Gastrointestinal epithelium | Mucosal force | 5-HT pathway→mucosal secretion | 212 |
| Airway | Stretch | Ablation of Piezo2→Airway-innervating sensory neurons→ respiratory distress and death in newborn mice | 214,215 | |
| Urinary bladder | Stretch | Sensory neuron→bladder filling sensation | 221 | |
| Piezo1/2 | Baroreceptor reflex | Shear stress | Elevated blood pressure→Piezo1/2 →nodose-petrosal-jugular-ganglion complex→ decreased blood pressure and heart rate | 216–218 |
| Chondrocyte anabolic and biosynthesis | Mechanical stress | GsMTx4→Piezo1/2 inhibition→ alleviate chondrocyte injury | 219,220 |
ECM extracellular matrix, MMP matrix metalloprotease, 5-HT 5-hydroxytryptamine
Many mechanical cues behave based on Piezo channels, such as ultrasonic stimulation,234 mechanical pulling,235 and fluid flow.222,236 For instance, the Piezo1 channel expresses in endothelial cells and is involved in vascular development and vascular tone.232,237 However, the increase in blood pressure of a Piezo1 disruption mice model contradicts the results above.238 Piezo1 triggers the elevation of blood pressure in the body-activated state instead of the body-inactivated state. In addition, the central nervous system can detect mechanical cues that modulate cell differentiation, cell migration, cell adhesion, gene expression, ion conversion, vesicular transportation, and fluid homeostasis.239,240 Mechanical stretch can also induce apoptosis in dense cell regions and cell differentiation in sparse cell regions through Piezo1.226 The fundamental senses, such as touch, proprioception, and mechanical pain, are closely associated with Piezo2 channels.241–245 In glaucoma patients, ECM deposition has been detected in the trabecular meshwork.246 The activation of Piezo1 by HP triggers ECM degradation and suppresses fibronectin synthesis.247 However, some contradicting results showed that stretching force induces human trabecular meshwork cells’ ECM production. This is possibly due to the alleviation of the effect of mechanical force by Piezo1 activation. TF and ECM stiffness regulate Piezo1 activation to different degrees. Further investigations are required on how Piezo channels mediate mechanical cues-induced diseases and which pathway plays a predominant role in disease progression. Pharmacological modulators can also activate Piezo channels. For instance, Jedi1/2 and Yoda1 can trigger Piezo1 independent of mechanical cues.248,249 In the urinary system, Piezo1 increases in the partial bladder outlet obstruction model, contributing to bladder injury. The inhibition of Piezo1 might be an alternative therapy to ameliorate bladder storage dysfunction.250 In addition, Piezo1 and Piezo2 often function synergistically. Piezo2 is reported to engage in the bladder filling process, and lack of Piezo2 in sensory neurons led to bladder dysfunction to some extent. However, the ablation of Piezo2 in mice does not cause complete urinary tract function loss, and Piezo2-deficient humans are still able to urinate.221 The co-function of Piezo1 in mediating stretching responses might be the reason.
Integrins in mechanotransduction
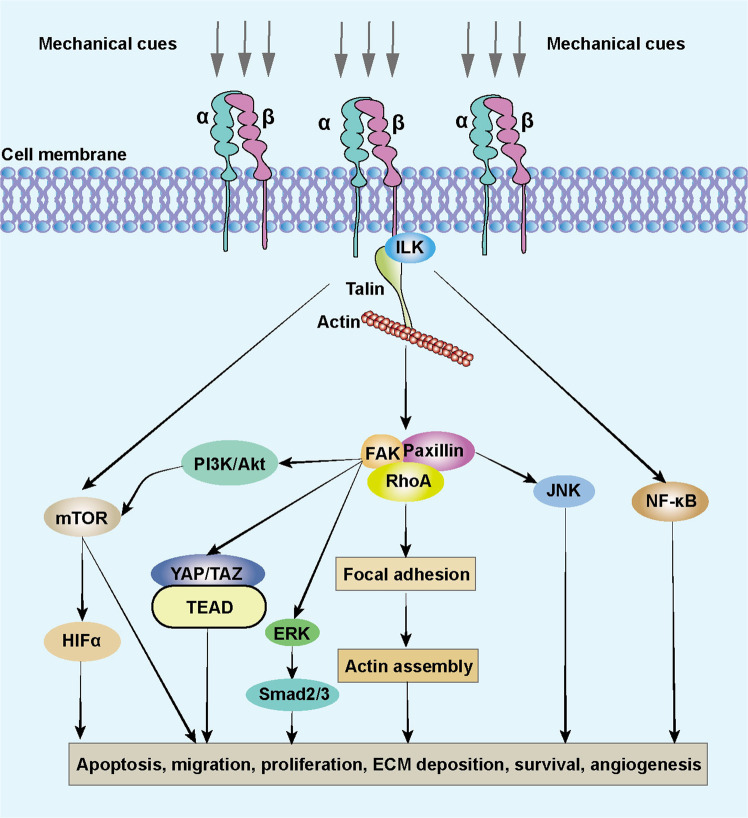
Integrins are identified as transmembranal receptors on the cell surface that function in cell adhesion and biochemical signal transduction. In mammals, there are a total of 24 different pairs of heterodimeric integrins comprising 18 α and 8 β subunits.251,252 In general, the cytoplasmic domain of many β subunits is highly homologous, and the integrins bind cytoskeletal structures through β subunits.253 The integrin complexes express cell-specifically depending on the cell type and developmental stage. Different integrin complexes function in various phenotypes. For instance, the integrin αvβ6 has been identified to trigger spatially restricted activation of TGFβ1 to prevent fibrosis progression.254 In both humans and animals, the loss of integrin v6 activity can lead to baldness, amelogenesis imperfecta, periodontal disease, and altered immunological responses in the lungs and skin.255 In contrast, α1β1, α5β1, and α7β1, which function in collagen, fibronectin, and laminin-binding receptors, are highly expressed in the cardiomyocyte to promote fibrosis.256–258 Besides, cardiac fibroblast expresses α1β1, α2β1, α11β1, and β3 integrins, which act as collagen-binding integrins.259–262 The integrin αv is recognized as a principal pathway in multiple organ fibrosis. The attenuation of integrin αv can alleviate liver and lung fibrosis.263 The integrins α1β1, α2β1, α3β1, α4β1, α5β1, α6β1, αvβ1, αvβ3, and αvβ5 have all been identified in cardofibroblast that play important role in cardiac remodeling. Apart from the above statement, the integrins α8β1 and α11β1 are selectively expressed HSCs that induce liver fibrosis.264,265
Integrins are also recognized as mechanotransducers in cell-ECM and cell-cell communication processes.258,266 Integrins can respond to various mechanical cues, especially the ECM stiffness (Fig. 5). Integrin signaling is assisted by many signaling molecules, including talin, paxillin, vinculin, FAK, ILK, and α-actin, to form links between intracellular and extracellular signals.267,268 Integrin-associated signaling pathways are activated by mechanical cues to promote cell proliferation, cell apoptosis, cell migration, cell survival, angiogenesis, and ECM deposition with positive feedback. For example, matrix crosslinking can enhance the integrin signaling cascades to facilitate tumor progression and tissue fibrosis.269,270 In renal fibrosis, ECM deposition and cross-linking result in pathological fibrotic change induced by mechanical stress.271 The integrin αvβ3 serves as a mechanosensor to promote the development of keloid.272 A stiffening keloid matrix activates integrins to induce gene expression through FAK/ERK signaling pathway and interacts with TGFβ/Smad cascades as well.273 In cellular mechanotransduction process, integrin-dependent RhoA/ROCK signaling is required in signal transduction and the related gene expression of fibrosis. Besides, RhoA/ROCK also triggers nonmuscle myosin II activity that influences the architecture of cytoskeleton. TF triggers integrins to interact with FAK and Src family kinases and promotes cell stiffening.274 The change of cytoskeleton transduces the signal to activate YAP/TAZ dephosphorylation and translocate into the nucleus to promote downstream gene expression through FAK, Src, and JNK signaling pathways.275–277
Fig. 5.
Mechanisms of integrins responding to mechanical stimulation. The β integrin interacts with ILK and talin to trigger downstream cascades. ECM extracellular matrix, ILK integrin-linked kinase
Taken together, integrins are important sensors in mechanotransduction, which can interact with many functional domains and signaling pathways to promote pathophysiological processes. Given that many emerging therapies targeting integrins (i.e., α4β7, αvβ6, and α5β1) in multiple diseases are in process (Table 2), whether the interventions affect normal tissue or organs is uncertain. Therefore, more tissue- or organ-specific therapies targeting integrins are needed. In addition, how these integrins communicate with other receptors, ion channels, or transmembranal proteins still needs further discussion.
Table 2.
Typical clinical trials targeting integrins
| Integrin subtype | Intervention/treatment | Disease type | Phase | Current status | ClinicalTrials.gov identifier |
|---|---|---|---|---|---|
| α5β1 | Volociximab | Metastatic renal cell carcinoma | 2 | Terminated | NCT00100685 |
| Pancreatic cancer | 2 | Completed | NCT00401570 | ||
| Ovarian cancer, primary peritoneal cancer | 1/2 | Completed | NCT00635193 | ||
| α4β7 | Vedolizumab | Ulcerative colitis | 4 | Recruiting | NCT05481619 |
| Crohn’s disease; ulcerative colitis | Not applicable | Completed | NCT02862132 | ||
| Inflammatory bowel disease | Not applicable | Completed | NCT02712866 | ||
| Type 1 diabetes | 1 | Recruiting | NCT05281614 | ||
| αvβ1; αvβ3; αvβ6 | IDL-2965 oral capsule | Idiopathic pulmonary fibrosis | 1 | Terminated | NCT03949530 |
| αvβ6; αvβ1 | PLN-74809 | Idiopathic pulmonary fibrosis | 2 | Completed | NCT04072315 |
| αLβ2; α4β1 | 7HP349 | Solid tumor | 1 | Completed | NCT04508179 |
YAP/TAZ in mechanotransduction
YAP was previously isolated from the interacting protein of Yes kinase278 and acted as a transcriptional coactivator.279 In general, YAP binds to TAZ to interact with the TEAD family of transcriptional enhancers (TEAD1-4) to regulate gene transcription.280 YAP functions in tissue and organ development, cell migration, tissue regeneration, and homeostasis.281 YAP receives both biochemical and biomechanical signals dependent or independent of the Hippo signaling pathway. Once YAP is activated, it is dephosphorylated and translocates into the nucleus to regulate downstream gene expression. As a transcriptional coactivator, YAP often functions with the assistance of several transcriptional factors, such as Smad, RUNX, signal transducers and activators of transcription (STAT), and others.
As a critical downstream effector of the Hippo signaling pathway, Hippo/YAP signaling pathway regulates the regeneration of organs and induces the occurrence and progression of tumors and fibrotic diseases.282–285 For instance, in gastric cancer, Hippo/YAP promotes cancer cell survival and metastasis by activating SIRT/Mfn2/mitochondrial signaling.286 Current studies have shown that ECM is one of the crucial factors promoting fibrosis.287,288 ECM stiffness can significantly enhance matrix deposition, cell adhesion, and tissue remodeling.118,289 YAP/TAZ is highly expressed in fibrotic tissues and upregulates the expression of ECM-related genes through TGFβ/Smad signaling pathway.151 High expression of YAP in adherent cells promotes normal cell proliferation in vitro.290 However, the excessive dephosphorylation of YAP results in uncontrolled cell proliferation and, ultimately, organ overgrowth and diseases.290 Studies demonstrated that Yap knock-out mice presented attenuation in cardiac hypertrophy and progression in fibrosis and apoptosis. This indicated that YAP could promote hypertrophy and survival in adaptive response.291 It was reported that YAP-regulated proliferation of small intestinal epithelial cells in the nucleus controls post-inflammatory cell regeneration and serves as a potential therapeutic target for ulcerative colitis.292 Under various conditions, YAP/TAZ also reprograms mature cells to a poorly differentiated state both in vivo and in vitro.283 Besides, YAP/TAZ also plays a crucial role in EMT,293 promotion of angiogenesis,294 hypoxic stress-induced cancer glycolysis,295 and metabolism.281,296
More importantly, YAP/TAZ responds to mechanical cues, including ECM stiffness, skeletal TF, cell geometry, cell density, substrate adhesion, and non-directional FSS, thereby regulating tumor and fibrosis progression.149,297,298 The currently known biological stress-promoting effects of YAP are mainly manifested in cell self-renewal,299,300 osteocyte differentiation,149,301 epithelium and vascular remodeling,149,302,303 epithelial damage repair,304,305 fibrosis,151,306–308 and others. Additionally, mechanical cues can modulate YAP/TAZ activity via large tumor suppressor kinase (LATS)-dependent or LATS-independent pathways. For example, it has been reported that ECM stiffness interacts with TGFβ depending on YAP/TAZ and Smad2/3. The inhibition of YAP/YAZ lowers the accumulation of Smad2/3 and alleviates renal fibrosis.309 NUAK family kinase1 (NUAK1) expression is triggered by YAP/TAZ to create a positive feedback loop of fibrosis.310 Fibroblast activation, a key result of fibrosis, is mediated by CD44/RhoA/YAP signaling under mechanical stimulation in crystalline silica-induced silicosis.311 ECM deposition and smooth muscle actin expression are initiated, associated with fibroblast generation and transformation.312 YAP has been identified to respond to ECM stiffness-induced bladder smooth muscle proliferation in urinary bladder fibrosis. Prolonged high ECM stiffness results in YAP shuttle to the nucleus and bound to Smad3 to trigger a downstream proliferative cascade.313 Moreover, the inhibition of mechanotransduction by YAP ablation restricts the activation of Engrailed-1 to promote wound healing mediated by Engrailed-1-negative fibroblast without scarring.314 Scarring is attenuated by the limitation of ECM/YAP/integrin-triggered pro-fibroblast phenotype, induced by P21-activated kinase (PAK) protein inhibition.315 The potential YAP/TAZ-targeted drugs have been ubiquitously investigated in several clinical trials (Table 3).
Table 3.
Typical clinical trials targeting YAP/TAZ
| Target | Intervention/treatment | Disease type | Phase | Current status | ClinicalTrials.gov identifier |
|---|---|---|---|---|---|
| YAP | Simvastatin | Prostate cancer | 2 | Recruiting | NCT05586360 |
| ION537 | Advanced solid tumors | 1 | Completed | NCT04659096 | |
| YAP/TAZ | Zoledronate | Breast cancer | 2 | Terminated | NCT02347163 |
| TEAD | IK-930 | Solid tumors | 1 | Recruiting | NCT05228015 |
Besides, TRPV4 is another important mechanosensitive ion channel that responds to mechanical cues, such as ECM stiffness. For instance, ECM stiffness can induce the EMT process by activating TRPV4/AKT/YAP signaling.316,317 Recently, mechanical transduction has been highlighted in hearing and balance transduction. Mechanical stimulation activates transmembrane inner ear (TMIE) through direct or membrane contact with the auxiliary subunit, which binds to the transmembrane-like protein 1 (TMC-1) mechanical transduction complex, thereby opening the pore for ion permeation.318–320 Moreover, the six missense mutations of TMC-1 in mice all indicated a decrease in Ca2+ permeability.321 The outcomes supported that TMC-1 was the pore of the mechanoelectrical transducer channel critical in cellular mechanotransduction.
In addition, humidity receptors are also identified as mechanosensitive and thermosensitive molecules. The sensory effectors of Or42b olfactory sensory neurons are present in cuticular deformations in moist air conditions, which demonstrates the transformation from humidity into mechanical signals. Intriguingly, TMEM63 showed functional conservation in rescuing the moisture response in Tmem63-deficient mutant flies.322 Furthermore, TMEM63 is also sensitive to osmotic stress, stretching force, and negative pressure.323,324 However, the in-depth biochemical mechanisms of these ion channels in mechanical transduction are poorly studied, which provides novel research insights into the related fields.
Signaling pathways in cellular mechanotransduction
Current research indicates that mechanotransduction-related diseases are associated with various signaling cascades, including TGFβ/Smad signaling pathway, Janus Kinase (JAK)/STAT signaling pathway, Wnt/β-catenin signaling pathway, ERK1/2 signaling pathway, RhoA/ROCK signaling pathway, and epigenetics signaling.325 Here, we elucidated several crucial signaling pathways associated with cellular mechanotransduction in normal conditions and diseases.
RhoA/ROCK signaling pathway
RhoA belongs to the Rho-family small GTPases, which is a critical modulator of cell adhesion and cytoskeleton, and many cellular processes, such as cell migration, proliferation, and survival.326,327 A major downstream of RhoA is the serine-threonine kinase ROCK, which triggers actomyosin contractile force.328,329 The RhoA/ROCK signaling mediates cell skeleton remodeling, cell contractility, and cell death process in response to multiple biochemical and biomechanical signals.330
RhoA/ROCK signaling pathway is engaged in many diseases, including osteoarthritis,331 Alzheimer’s disease,332 ischemic stroke,333 hepatic and pulmonary fibrosis,334,335 and cancer.336 For example, the RhoA/ROCK signaling regulates the cardiac fibroblast-to-myofibroblast transformation (FMT) process.337 The inhibition of RhoA/ROCK attenuates the myocardial fibrosis in type II diabetes rats through JNK and TGFβ signaling pathways.338 The TGFβ-induced EMT process is also mediated by RhoA/ROCK signaling pathway.339
In response to mechanical stimulation, ECM-induced remodeling activates integrins to facilitate the RhoA/ROCK signaling pathway in cellular mechanotransduction and osteogenetic differentiation of mesenchymal stem cells.340 Stiff ECM-mediated integrin-induced directional migration through RhoA/ROCK signaling cascades provides new insights into inhibiting cancer metastasis.341 A study on mechanical stress-associated cardiac valve remodeling revealed that the mechanical stress-induced valvular fibrosis through RhoC signaling but was independent of RhoA signaling.342
TGFβ/Smad signaling pathway
TGFβ/Smad is considered the canonical pathway of fibrosis.343 TGFβ induces the phosphorylation of Smad into the nucleus to promote downstream gene transcription. Three isoforms of TGFβ (1/2/3) have been identified. TGFβ1 and TGFβ2 are the most critical in function.344,345 TGFβ1 promotes the expression of transient receptor potential melastatin 7 (TRPM7) through the TGFβ/Smad3 signaling pathway and facilitates the airway smooth muscle cell proliferation.346
The Smad family proteins, which are the central transducers of the TGFβ signaling pathway, are classified as R-Smad, inhibitory Smad (I-Smad), and universal Smad (co-Smad, Smad4).347 In R-Smad, Smad2/3 are considered the main mediators of the TGFβ signaling pathway, and Smad1/5/8 mediate the BMP signaling pathway.348 R-Smad dissociates from phosphorylated receptors and binds to co-Smad (Smad4). This complex is a heterotrimer, with two R-Smad binding to one Smad4349 and one R-Smad binding to one co-Smad.350 Many transcription factors are reported to bind to Smad proteins.351 In addition, other proteins can also interact with or even modify Smad proteins. These modifications often affect the strength of signal output and interact with different signaling pathways. Smads can be modified by phosphorylation, ubiquitination, acetylation, silylation, and ADP ribosylation.352,353
The TGFβ/Smad signaling pathway is essential in inflammatory bone destruction,354 kidney disease,355–357 immune system,358 malignancy,359 neurological disease,360 and inflammatory bowel disease.361 Mechanical stress can activate TGFβ/Smad signaling, which drives the progression of lung fibrosis.362 It has been demonstrated that high mechanical stress can activate TGFβ1/connective tissue growth factor (CCN2)/integrin to induce fibrosis.363 TGFβ1 is modulated by cell-generated TF, promoting the disruption between TGFβ1 and latency-associated peptide (LAP).364 In this process, integrins interact with LAP, and integrin-mediated stretching force changes the conformation of LAP, thus triggering the release of latent TGFβ1,365 thereby contributing to severe fibrotic changes.
Although there are many challenges in treatment targeting TGFβ, the antagonists and monoclonal antibodies are under investigation.366,367 It is worth mentioning that the resolution may be targeting biochemical signaling cascades rather than TGFβ. Some studies have revealed that small molecule inhibitors can attenuate TGFβ-induced fibrosis as well.368–371
JAK/STAT signaling pathway
JAK signaling pathway, also known as the interleukin (IL)-6 signaling pathway, functions in multiple biochemical processes, such as cell proliferation, cell transformation, cell apoptosis, tissue remodeling, immune regulation, and hematopoiesis.372 JAK is activated by binding cytokines, growth factors, or interferon to facilitate dimerization.373 JAK phosphorylates STAT in the cytoplasm, and the phosphorylated STAT is translocated into the nucleus to regulate gene transcriptiton.374
JAK/STAT pathway has been reported to be engaged in innate immunity and adaptive immune responses,375,376 including rheumatoid arthritis,377 Parkinson’s disease,378,379 multiple sclerosis,380 inflammatory bowel disease,381 sepsis,382 liver cirrhosis383 and tumors.384 In the fibrotic process, STAT3 activation triggers ECM deposition and MMPs transcriptional control.385–387 The interactions between TGFβ/JAK/STAT3 fibrosis, independent of Smad, have been identified.373,388 Besides, TGFβ can also function as a target gene of STAT. STAT3 has been reported to enhance liver fibrosis by upregulating TGFβ.389,390
Research from two decades ago illustrated that JAK/STAT signaling is activated by mechanical stretch in cardiomyocytes of rats, partly depending on angiotensin II.391 Then the JAK/STAT signaling was investigated widely, i.e., cyclical stretch triggered the expression of MMP14 and MMP2 in neonatal rat cardiomyocytes.392 JAK/STAT related to fibroblast activation is also identified as the downstream cascades of ROCK in mechanotransduction.393 TGFβ/integrin signaling cascades have been reported to be involved in the ECM alteration positive feedback process. JAK/STAT, the response of TGFβ, also functions in the cellular mechanotransduction process. Studies demonstrated that mechanical stress triggers osteogenic differentiation through JAK/STAT and PI3K/AKT signaling cascades.394 Mechanical stress can also communicate with intracellular signals, including JAK/STAT, signaling, and MAPK signaling, via integrins, cytoskeleton, and sarcolemmal proteins.395 However, some mechanisms of JAK/STAT-associated diseases in cellular mechanotransduction still need further studies.
Wnt/β-catenin signaling pathway
As an essential downstream cascade of TGFβ-mediated fibrosis, Wnt signaling is indispensable in embryonic development and cell proliferation and migration, tumorigenesis, and cell survival through β-catenin activation.396–398 The Wnt proteins interact with receptor complexes on the cell surface to initiate intracellular Wnt/β-catenin signaling.399 β-catenin is also a transcription factor that functions through its translocation into the nucleus.
Many studies demonstrated that the Wnt/β-catenin signaling pathway involves in the fibrotic process. For instance, studies identified that the ablation of Wnt/β-catenin signaling attenuated age-associated mitochondrial dysfunction and renal fibrosis.400 Wnt/β-catenin signaling can facilitate wound repair of kidney injury. However, excess activation of Wnt/β-catenin in renal tubular epithelial cells and fibroblasts result in renal fibrosis.401 In this process, Snail1, the initial step of EMT, is activated by β-catenin in the renal tubular epithelial cells.402 Wnt/β-catenin can be triggered by the dynamic stimulation of ECM involved in the EMT-to-fibrosis process.403 In addition, the Wnt/β-catenin signaling pathway activates IL-4 and TGF-induced macrophage cell (M2) polarization to promote renal fibrosis.404 The activation of Wnt/β-catenin cascades induces fibroblast proliferation and differentiation to myofibroblast.405 Further studies revealed that the TGFβ/Smad3 signaling pathway also interacts with the Wnt/β-catenin signaling pathway in vascular remodeling.406
Mechanistically, Wnt/β-catenin signaling is a critical effector in mechanical transduction, including osteogenesis, cardiovascular disease, and osteoarthritis.407–410 Wnt/β-catenin cascades are associated with YAP/TAZ, which are important effectors of mechanical cues. Besides, Wnt/β-catenin signaling is also regulated by integrins. Wnt/β-catenin signaling was initially identified in loading-triggered osteogenesis.411 Osteocytes and osteoblasts respond to SS, TF, and mechanical load by triggering canonical Wnt/β-catenin cascades.412 Compressive mechanical stress attenuates Wnt signaling, impairing chondrocyte proliferation and cartilage matrix degradation.413 Studies demonstrated that β-catenin stabilization responds to tension or ECM stiffness to activate cell adhesion and E-cadherin-dependent mechanism.414–416 ECM stiffness also promotes EMT-induced aortic valve fibrosis through the activation of β-catenin.417
ERK1/2 signaling pathway
ERK signaling pathway activates ERK1/2 phosphorylation to promote the transcription of genes.418 ERK1 and ERK2 belong to a family of structurally related kinases, which are also known as MAPKs. In general, the ERK signaling pathway functions in cell proliferation, cell survival, cell growth, cell metabolism, cell motility, cell differentiation, and cell development.419 For instance, RAS-induced ERK cascades modulate the G1/S phase transition in proliferative cells. The ablation of Erk1/2 in mouse embryonic fibroblast triggers cell cycle arrest in the G1 phase.420,421 ERK1/2 can also suppress the proapoptotic factors to maintain the cell survival.422 Studies revealed that RAS-ERK signaling was partly engaged in Myc-induced cell growth and cell size control.423 ERK can phosphorylate the transcription factor HIF1α. Besides, HIF1α regulates the process of glucose metabolism.424–426 ERK signaling pathway activation triggers the mobility program in response to mechanical stimulation.427 Studies also demonstrated that ERK signaling enhancement promotes the differentiation of mouse embryonic stem cells.428
Furthermore, the ERK1/2 signaling pathway is closely related to fibro-proliferative disorders. The inhibition of ERK alleviates pathological vascular remodeling429 and biological stress regulates the proliferation of bladder smooth muscle cells through β-receptor regulation of the cAMP (EPAC)-ERK1/2 signaling pathway.430 In the airway remodeling of asthma model mice, IL-13 induces airway smooth muscle proliferation and hypertrophy through the ERK1/2 signaling pathway431; and pulmonary hypertension regulates the pulmonary artery smooth muscle proliferation through the p38MAPK/ERK signaling pathway.432 Rap1 regulates vascular smooth muscle proliferation through the ERK1/2 pathway.433 In addition, IL-11 has been reported to respond to TGFβ stimulation. In mice, fibrogenesis protein synthesis depends on the activation of IL-11 and its receptor, which leads to multiorgan fibrosis.434 Cytokines such as IL-9, IL-11, and IL-17A can provide novel therapeutic targets for fibrosis prevention.435–437
Importantly, ERK1 and ERK2 are dominant features in mechanical cues-induced diseases. The ERK signaling functions independently or cooperatively under the regulation of mechanical stress. For example, the fibrotic scar on the skin of mice, which is induced by mechanical strain, promotes the expression of leucine-rich-α-glycoprotein 1 (LRG-1). ERK inhibition attenuates the expression of LRG-1 to alleviate the scarring.429 The pathologic scar induced by mechanical stress is regulated by FAK/ERK pathways.438
Other signaling pathways for mechanical cue-related diseases
Epigenetic regulation in various cell types functions extensively in fibrogenetic process.439 Epigenetics includes DNA methylation, histone modification, ncRNAs, miRNAs, RNA modification, and chromatin remodeling, which contributes to tumorigenesis, aging, and other diseases.440–446 In epigenetics-induced fibrosis, JMJD1 has been identified as an ECM stiffness-dependent factor regulating the transcription of YAP/TAZ and fibroblast activation.447 Epigenetic silencing is crucial for ECM stiffness-induced fibroblast activation.448
Hedgehog (Hh) signaling is a mediator for fibroblast transformation in fibrosis and consists of a series of signaling pathways in organ development, homeostasis, fibrosis, and regeneration.449–453 Hh signaling can be activated during macrophage cell activation and other biological responses.454–456 A recent study identified mechanical stress/Hh as a novel signaling cascade in fibrotic disease.457 Targeting Hh signaling can be a sight for the treatment of mechanical cues-induced diseases.
Hippo/YAP pathway is associated with tissue and organ development, remodeling, and several diseases. Angiotensin A2 activates YAP/TAZ, the critical downstream target of Hippo signaling, and promotes the migration and proliferation of vascular smooth muscle cells.302 YAP also interacts with cardiac proteins to regulate vascular remodeling.458 As a mechanosensitive signaling pathway, different mechanical stimulation can modulate the proliferation and apoptosis of vascular smooth muscle cells through Hippo/YAP signaling.459
There is evidence showing that hypoxia,460 Rho/ROCK signaling,342,461 PI3K/AKT signaling pathway,462 NF-κB signaling pathway,463 STAT3/Pim-1/NDAT signaling pathway,464 NLRP3 inflammasome-dependent pathway,465 and Notch signaling pathway466 are all related to the mechanical force-induced pathophysiological processes. Collectively, biomechanical signals have been extensively studied in health and diseases, but many unknown mechanisms remain to be explored.
Cellular mechanotransduction in tissue development and repair
Mechanical stimulation is engaged in many biological activities, such as tissue development, regeneration, and remodeling. The mechanisms of cellular mechanotransduction are well-studied in skin repair, nerve regeneration, angiogenesis, vascular remodeling, and others, all of which are important in maintaining the physiological homeostasis of the human body.
Mechanotransduction in embryonic development
Mechanotransduction in embryonic development plays a vital role in cell shape response by morphogenetic movements of tissue.467 Embryonic morphogenesis is closely associated with biomechanical signals upon cells and/or tissues that lead to morphological changes.468
ECM stiffness, FSS, and TF are principal mechanical cues in embryonic development.469,470 For example, in cardiovascular development, the embryonic vasculature system can sense the mechanical stimulation in vascular remodeling and certain aspects of sprouting angiogenesis.471 Furthermore, the mechanical properties of the embryonic heart are soon shaped from a tube that assists the nascent vasculature in the early embryogenesis process.472 In cardiac maturation, the increase of ECM is deposited by proliferating fibroblasts along with more sarcomeric proteins expressed by cardiomyocytes so as to increase the contractility of the heart. In addition, ECM stiffness can regulate short-term and long-term deformations of embryonic tissues through integrin-mediated adhesion and cell-cell interaction.473 Mechanical cues are also involved in nervous system development. FSS and HP participate in the development of cerebral ventricles. The tissue stiffness also regulates neural tube closure, neural progenitor proliferation and differentiation, and neural crest cell migration in embryonic development.474 Besides, embryonic tendon development is also closely correlated with tissue stiffness and dynamic mechanical loading.475
Mechanotransduction in skin repair
As the largest organ in the human body, the skin maintains a strong and pliable physical structure for integrity and flexibility. Skin is mechanosensitive in human body,476 and multiple biomechanical properties regulate skin repair process through mechanosensitive signaling.477 During skin natural repair and surgery repair, skin soft tissue expansion encounters many unsolved problems, such as skin quality, tissue retraction rate, and long-term treatment.478
ECM is abundant in dermis, especially collagens, which provide sufficient TF in the skin. The skin TF on the actin filaments and intermediate filaments integrates integrin-mediated pathways to trigger the maturation of focal adhesion. In addition, the long-term TF facilitates papillary fibroblast proliferation, ECM deposition, and TGFβ expression, thereby promoting a satisfactory regeneration of the skin. For example, the tension within the epidermis promotes gene expression through DNA methyltransferase 3A (DNMT3A) nuclear translocation,479 which is a potential regulator of skin repair. However, the persistent excessive TF triggers premature papillary fibroblast, leading to poor regeneration of the skin.480 Many studies show that the stiffness of skin tissue ensures the regenerative ability during the wound healing process. The ECM secreted and remodeled by matrix metalloproteases realizes a complex balance for the dermis in tissue injury.481 Of note, the skin repair process is always followed by hypertrophic scar formation, which is characterized by skin proliferative disease. For example, fibroblast is highly mechanosensitive, which is responsible for scar formation.482 Piezo1 is highly expressed in hypertrophic scar,483 so targeting mechanosensitive effectors may be a potential therapeutic targets for the scar.
Mechanotransduction in nervous system regeneration
Recent studies have been focusing on the impact of biomechanical cues on the nervous system. Mechanical cues are engaged in neuron development, proliferation, differentiation, migration, and axon extension. Multiple mechanics-associated signaling pathways (i.e., Hippo signaling pathway) are essential for nervous system development and nerve function maintenance.484,485 For example, YAP/TAZ can integrate biochemical and biomechanical signals to promote myelination via the activation of Schwann cells proliferation and transcription of basal lamina receptor genes, both of which are essential for myelination. ECM stiffness-induced YAP/TAZ activation triggers the axon outgrowth by actin-adhesion coupling.486,487 A recent study demonstrated that mechanosensitive ion channels, such as Piezo1 and Piezo2, are rich in Schwann cells. Piezo1 is a transient inhibitor of radial and longitudinal myelination, but Piezo2 may be essential for myelination in Schwann cells.488 Researchers also found that human motor neurons tended to survive in rigid conditions with the elasticity of muscle, but human forebrain neurons preferred brain tissue-like elasticity.489
For neural development, the embryonic cerebrospinal fluid (CSF) is secreted into the lumen of the brain and ventricles. The CSF pressure triggers a dramatic dynamic expansion of the brain.490 The telencephalon developing to left and right lobes are associated with CSF-induced mechanical environment.491 The central nervous system is mechanically induced only by tangential growth but not radial growth. This suggests that the mechanosensors and transducers are different in the nervous system.492,493
Apart from endogenous mechanical stimulation, TF can also promote axon growth.494,495 Cell motility and adhesion are both involved in the determination of the extension of axon and growth cone activities.496,497 In general, axonal transport is driven by mechanical methods with the help of several motor proteins such as myosin, kinesin, and dynesin.498 Hence, more studies of mechanical cues-regulated mechanisms are to be conducted for the nervous system.
Mechanotransduction in angiogenesis and vascular remodeling
Angiogenesis is a multi-stage process characterized by endothelial cells activated by angiogenin, VEGF, platelet-derived growth factor (PDGF), epidermal growth factor (EGF), fibroblast growth factor, TGFβ, and tumor necrosis factor α (TNFα).499 For morphological changes in endothelial cells, pericytes detach from endothelial cells that release metalloproteases to degrade ECM.500 Vascular tubes are formed by tip cell migration and stalk cells proliferation.501 The mechanisms of angiogenesis above also meet the requirements in embryonic development.
Mechanical cues, such as ECM stiffness, TF, and FSS, play important roles in angiogenesis and vascular remodeling. Stiffing ECM induces pro-angiogenesis gene expression via YAP activation, such as Vegfa, Vegfb,502 thereby promoting angiogenesis. In addition, TF in blood vessels generated by blood circulation activates mechanosensitive proteins (i.e., Piezo1 and TRPV4) and facilitates biomechanical signaling pathways. For instance, the activation of Piezo1 upregulates membrane type1 MMP, AKT, and mTOR signaling,503,504 thereby increasing VEGF expression. Besides, FSS in blood vessels remodels endothelial cells by activating Piezo1, TRPV4, tyrosine kinase receptors (TKRs), G-protein-coupled receptors, and integrins.505,506 Branches and bends of arteries have intricate blood flow patterns that can trigger vascular dysfunction. However, physiologically high FSS is protective for angiogenesis and vascular remodeling by regulating BMP-TGFβ, WNT, Notch, hypoxia-inducible factor 1α (HIF1α), TWIST1, and HOX family genes.507 A previous study showed that higher FSS reduced vascular density in veins but not in arteries during vascular development.508
Mechanical strain and FSS in blood vessels are the main mechanical cues in vascular remodeling. In several disease progression stages, the increased FSS induces vascular remodeling to maintain enough tissue perfusion. VEGFR2, VEGFR3, and VE-cadherin communicate with transmembrane domains (TMDs) within the plasma membrane. FSS activates PECAM-1, leading to the activation of Src and VEGFRs.509 In the vascular remodeling process, ROS, NO, NF-κB, epidermal growth factor receptor (EGFR), MAPK, and PKC signaling pathways are all activated by mechanical cues.510,511 For (myo)fibroblasts, mechanical stretch can significantly increase the expression of β1 integrin, α-SMA, Nesprin2, and laminA/C. TF modulates the assembly of α-SMA through the Rho kinase pathway512 and the fibroblasts are transformed into α-SMA+ myofibroblasts. Other studies have found that the TF can promote fibroblast differentiation by activating TGFβ1, thereby promoting the remodeling of tissues and organs.103 Cyclic stretching can also rearrange microfilaments in human periodontal ligament (PDL) cells, thereby regulating cytoskeleton-related changes of gene expression.513 This process may be affected by the Rho signaling pathway,514 but the mechanism is still unknown.
Mechanotransduction in diseases
Although the normal range of physiological mechanical stimulation exhibits a critical impact on the sustainment of cell function, it is important to note that continuous excessive mechanical stimulation is associated with a wide range of diseases and pathologic conditions, including tissue injury and fibrosis, tumor behaviors, cardiovascular diseases, and aging.
Tissue injury and fibrosis
Fibrosis is an excessive tissue repair process induced by multiple tissue damage factors. The combined annualized incidence is approximately 4968 out of 100,000 person-years globally.515 Although the fibrotic response may only cause minor changes to tissues in the early stage, parenchymal sclerosis and cell dysfunction may occur long after. The fibrotic responses generally consist of the primary organ injury stage, effector cells (fibroblasts, myofibroblasts, bone marrow, fibrocytes, epithelial-derived cells in EMT) activation stage, ECM deposition, and loss of cellular homeostasis, which ultimately leads to organ failure.325,516 When the tissue receives a single or minor injury, the tissue repair process will be activated to rebuild the tissue structure. If the tissue suffers repeated or severe damage, the ECM will excessively deposit, eventually leading to fibrosis.
Etiology and mechanism of fibrosis
Fibrosis is often the sign of the final disease stage and is not easily reversed.517 The etiologies of fibrosis include persistent infections, toxin exposures, inherited disorders, chronic autoimmune inflammations, myocardial diseases, abnormal serum cholesterol, obesity, smoking, severe diabetes, and blood hypertension.518 Many organ disorders can induce fibrotic diseases, such as liver cirrhosis, kidney fibrosis, bladder outlet obstruction, IPF, cardiac fibrosis, corneal trauma, glaucoma, atherosclerosis, scleroderma, skin disorders, burns, radiation-induced fibrosis, chemotherapy-induced fibrosis, and surgical complications.116,429,477,519,520 Besides, chronic autoimmune diseases are also identified as potential causes of fibrosis, including rheumatoid arthritis, scleroderma, inflammatory bowel disease, myelosclerosis, and systemic lupus erythematosus.521–523 Since fibrosis causes irreversible damage to organs, identifying the exact mechanisms would provide effective therapies for preventing fibrosis.
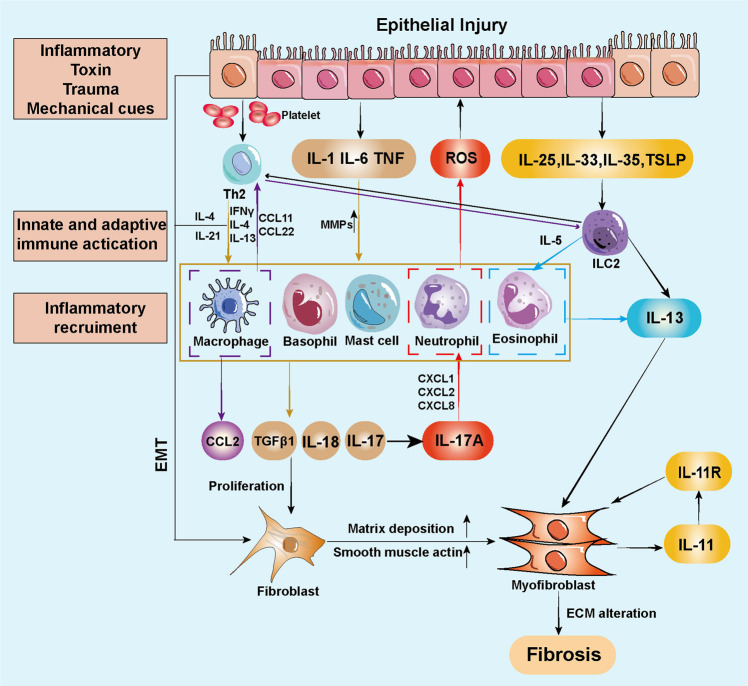
During fibrosis, myofibroblasts have long been recognized as core cell components in remodeling tissue. Therefore, FMT is critical in ECM accumulation and scarring processes. Multiple studies have shown that fibrosis triggers (myo)fibroblasts,524 muscle cells,525 and also recruits inflammatory cells (i.e., macrophages and T cells).526 When the tissue suffers an injury, the innate and adaptive immune responses will be activated527 (Fig. 6). Type I and Type II immunity are cross-activated for defense and metabolic homeostasis maintenance. Type II immunity involves cytokines (i.e., IL-4, IL-5, IL-9, IL-13, IL-25, and IL-33), thymic stromal lymphopoietin, eosinophils, basophils, mast cells, T helper 2 (Th2) cells, group 2 innate lymphoid cells (ILC2s), and IL-4/13-activated macrophages.528–531 The pro-inflammatory chemokines and cytokines (i.e., IL-1, IL-6, and TNF) secreted by macrophage cells, trigger the inflammatory recruitment process.532 TGFβ1, IL-17, and IL-18 can promote fibroblast proliferation as well.533 Meanwhile, IL-17A strengthens the neutrophil responses by CXCL1/2/8 and aggravates tissue injury by synthesizing reactive oxygen species (ROS).534 The IL-17A signaling increases the TGFβ1 receptors on fibroblasts and facilitates the secretion of ECM. The adaptive immune CD4+ Th2 cell can directly activate fibroblasts via IL-4 and IL-21, independent of TGFβ1-induced fibrosis.535,536 Intriguingly, it has been demonstrated that IL-4 is a potent profibrotic cytokine that is more effective than TGFβ.537,538 Similarly, LC2s-originated IL-5 can recruit and activate eosinophils. IL-5 has been identified in attenuating liver cirrhosis539 and intestinal fibrosis540 in mice. Eosinophils secrete IL-13 and TGFβ1 and both can facilitate the fibrotic function of the myofibroblast. Besides, IL-13 acts together with IL-4 to promote fibroblast proliferation and FMT.541 However, the efficacy of anti-IL-13 therapy in ulcerative colitis is controversial,542 and further investigations are required to identify the effect of IL-13 in the fibrotic alterations of Crohn’s disease.543,544 Fibroblasts can be activated directly by etiologies such as inflammations, toxins, mechanical force, and trauma through EMT process.545–547 However, renal fibrosis is only alleviated when IL-33/ILC2 is pre-inhibited in the unilateral ureteral obstruction model, while post-inhibitions tend to be useless.548
Fig. 6.
A schematic view of the fibrotic response. The innate and adaptive immune systems are activated at the tissue repair stage. The pro-inflammatory chemokines and cytokines secreted by macrophage cells trigger the inflammatory recruitment process. TGFβ1, IL-17, and IL-18 from immune cells promote fibroblast proliferation and transformation. In addition, type I and type II immunity are cross-activated for defense and metabolic homeostasis maintenance. The adaptive immune CD4+ Th2 cell can directly activate fibroblasts by IL-4 and IL-21, independent of TGFβ1 induced fibrosis. Excessive ECM deposition leads to fibrosis ultimately. ECM extracellular matrix, EMT epithelial-to-mesenchymal transition, IL interleukin, ILC2 group 2 innate lymphoid cells, MMP matrix metalloprotease, Th2 T helper 2, TGFβ1 transforming growth factor β1, TNF tumor necrosis factor
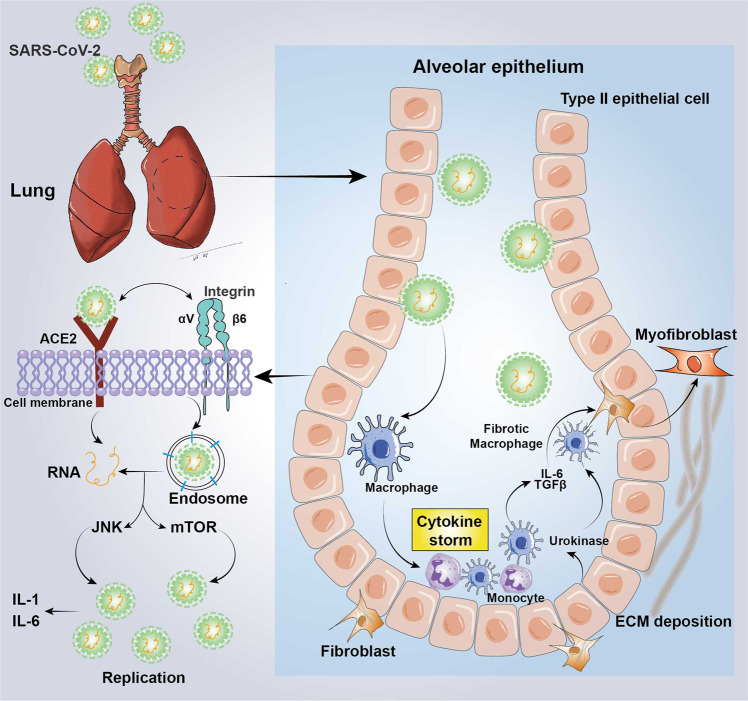
Apart from the common etiologies, the newly emerged coronavirus disease 2019 (COVID-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), can also lead to pulmonary fibrosis (Fig. 7). COVID-19 is manifested as a severe respiratory syndrome and leads to a worldwide pandemic.549 To date (Apr.5, 2023), there are already more than 761 million cases of COVID-19 occurrence and more than 6.89 million deaths from COVID-19 worldwide. An abrupt increase in transmission has arisen because of the Omicron variant (Nov. 26, 2021, WHO).550 The main symptoms of COVID-19 are generally asymptomatic infections, such as sore throat, headache, and fatigue.551 In some cases, the conditions progress to severe pulmonary infections requiring intensive care or even mechanical ventilation (MV).552 It is worth noticing that even though COVID-19-related fibrosis was observed to be relieved in one-third of the patients after four months from onset,553 most post-COVID-19 fibrotic lesions still cannot be cured.
Fig. 7.
Biochemical mechanisms of SARS-CoV-2-induced lung fibrosis. SARS-CoV-2 initially binds to ACE2 of the epithelial cells to activate integrins or CD98. Integrins, especially αVβ6, can assist the SARS-CoV-2 binding to ACE2, thus enhancing the ability of viral infectivity. After SARS-CoV-2 is inhaled, the virus replicates through the JNK and mTOR signaling, which facilitate the generation of NLRP3 inflammasome. Meanwhile, the type II alveolar epithelium cell injury induces pro-inflammatory recruitment of immune cells, such as macrophages. The cytokine storm will then be released and triggers the proliferation and migration of fibroblasts. Besides, the activation of TGFβ triggered by integrins, IL-1, and IL-6 promotes ECM deposition and FMT. ACE2 angiotensin-converting enzyme 2, CAMK calmodulin kinase, SARS-CoV-2 severe acute respiratory syndrome coronavirus 2, ECM extracellular matrix, FMT fibroblast-to-myofibroblast transformation, IL interleukin, TGFβ1 transforming growth factor β1
Overview of mechanical cues-induced fibrosis
ECM deposition destabilizes the tissue’s mechanical environment,554 along with the change of cell survival.555 The expansion and contraction of hollow organs and the fluid flow in the cavities produce mechanical stimulation on tissues and organs, such as HP, FSS, and TF.556–559 In addition, ECM deposition can exacerbate intrastromal cross-linking, increase tension of the tissues, and induce fibrosis.118 These common mechanical cues function as a double-edged sword: physiological mechanical signals ensure the normal development and repair of organs, while prolonged pathological mechanical stimulation causes irreversible damage.
Fibrosis-induced alteration of ECM environments promotes abnormal biological processes. In adipose tissue, ECM-associated mechanical cues do not induce regional apoptosis and inflammation, but trigger system-wide lipotoxicity and insulin resistance.560 Both integrins and Piezo channels are located in the cell membrane, which can sense the mechanical cues in regulating different biological processes. The integrins mediate biomechanical signals to affect cell adhesion, migration, proliferation, and differentiation.561,562 For example, MV can aggravate sepsis-related pulmonary fibrosis, where MV leads to lung fibroblast proliferation, ECM deposition in lung tissue, and increase in procollagen type I carboxy-terminal propeptide in the bronchoalveolar lavage fluid mediated by β3 integrin.563 Another hypothesis demonstrated that MV accelerates acute respiratory distress syndrome (ARDS)-associated pulmonary fibrosis via Piezo1-mediated Ca2+ influx and ATP release in lung epithelial cells.564 In addition, physiological stress promotes the development and structural stability of tissues and organs, while pathological stress disrupts this balance, thus resulting in irreversible damage to anatomical functions.565
Mechanical cues can target nearly all types of cells in the human body. Fibroblast, a common functional cell type in fibrosis, responds to the mechanical cues in fibrosis progression. In the early stage of tissue injury, mechanical cues integrates with inflammatory factors to enhance tissue adhesion and fibrosis.566 For example, the elevated ECM stiffness regulates fibroblast DNA methylation and chromatin condensation.567 Persistent fibroblast phenotype forms after the first week’s rapid response to ECM stiffness. In myocardial fibrosis, Selenbp1 knockdown enhances fibroblast activation and deters fibroblast transformation to matrix-degrading form.568 Mechanical cues activate TGFβ1, which is latent in ECM, subsequently activating fibroblasts.569 The activated fibroblasts are transformed to injury-related fibroblasts subtype, promoting ECM secretion. In addition, epithelial and endothelial cells can also sense changes in intracellular mechanical signals during tissue damage. Mechanosensors of epithelial and endothelial cells include adhesion protein complexes, primary cilia, and mechanosensitive ion channels.199,570 The activation of sensors can lead to functional alterations of the endothelial and epithelial barrier, inflammatory signaling, arteriogenesis, cell migration, invasion, adhesion, and proliferation.61,571 Endothelial cells transduce signals to the surrounding microenvironment through growth factors and chemokines.572 ECM can activate the cell status and promote fibrosis progression through biomechanical signals in a continuous and persistent manner.573 Apart from epithelium and endothelium, smooth muscle cells, HSCs, and renal tubular cells can also serves in cellular mechanotransduction.
Furthermore, several mechanical cues-induced signaling pathways are engaged in muscarinic receptor-mediated fibrosis. Muscarinic receptor-mediated HP/NF-κB induces the pro-inflammatory processes in the urothelium and smooth muscle of the urinary bladder.25,574 Similar performances are also observed in β adrenoceptors and integrin/FAK signaling.430,575,576 However, the β2 adrenoceptors in the heart can strengthen heart contraction and heartbeat by coupling with adenylyl cyclase, which is contrary to the observations in the urinary bladder.577,578 It is speculated that tissue heterogeneity is a possible reason for this and further validations are required. In addition, the monocyte chemotactic protein 1 (MCP1) and its receptor chemokine (C-C motif) receptor 2 (CCR2), and MCP1-induced protein (MCPIP1) are also engaged in mechanical cues-induced fibrosis. Pathological HP regulates the human bladder smooth muscle cell proliferation through MCP1/CCR2-SGK-1 signaling and contraction through MCP1/CCR2-MCPIP1.579,580 Therefore, HP is important in tissue repair and reconstruction, also in the exploration of therapeutic targets for fibrosis.
Mechanotransduction in pulmonary fibrosis
IPF is an interstitial pulmonary disease affecting gas exchange, and eventually leads to respiratory failure.581 Globally, the annual prevalence of IPF increases annually, with 8.65 per 100,000 per year in the United Kingdom (UK) from 2000 to 2012, and 1.2–4.6 per 100,000 per year in East Asia.582 Pulmonary fibrosis is evaluated by pulmonary functional testing, such as forced vital capacity (FVC), which reflects respiratory compliance.583 To date, the etiologies and mechanisms of IPF have not been clarified and no effective treatments have been found.
The mechanical cues in the respiratory cycle include air stress and tissue strain. These two mechanical cues act on ECM to facilitate the crosstalk between the matrix and the cell. In the lung, the ECM compartments, including basement membranes, are composed of glycoproteins, located on the epithelium and endothelium, and interstitial matrices, with loose and fibril-like meshwork with cross-linking structures and mesenchymal cells to maintain mechanical homeostasis.584 Persistent excessive stress and strain may cause abnormal deposition of ECM, leading to changes in ECM stiffness. In the lung, Young’s modulus of normal tissue is approximately 1.8 kPa (range ~0.2–3.7 kPa) while in pulmonary fibrotic tissue approximately 15.5 kPa (range ~3.6–54.1 kPa).585
All cells in the lung are regulated by mechanical stimulation. In general, the characteristics of IPF are epithelial cells aging and ECM deposition. In the normal tissue repair process, myofibroblasts are activated to secret ECM so as to facilitate AEC2 progenitor proliferation and differentiation, and thus repairing epithelium.586 However, a failed repair of alveolar epithelium repair may cause aberrant mesenchymal activation. Meanwhile, innate immune cells, such as monocyte-derived alveolar macrophages are important for pulmonary fibrosis.587 Mechanical stress can induce mast cell degranulation to activate TGFβ signaling in pulmonary fibrosis progression.588
For signaling transduction of pulmonary fibrosis, tyrosine kinase, and serine-threonine kinase pathways, G-protein-coupled pathways are associated with myofibroblast differentiation, matrix deposition, fibroblast proliferation, and reduced apoptosis. ECM stiffness and mechanical signaling (i.e., RhoA/ROCK, myocardin-related transcription factor-A [MRTF-A], and YAP/TAZ) promote pulmonary fibrosis via TRPV4-related signaling and others. Besides ECM stiffness, insufficient tissue repair leads to ECM component alteration, resulting in a mechanically stretched niche. The MV facilitates the TF to the lung and initiates acute respiratory distress syndrome (ARDS)-related pulmonary fibrosis. Mechanical stretch impairs the tight junction between alveolar cells. The loss of tight junction leads to the degradation of the cytoskeleton and cell-cell attachment. Tang et al. demonstrated that an elevated mechanical TF activates the TGFβ signaling loop in alveolar stem (AT2) cells.362 Tang et al. found that AT2 cells in lungs distribute less uniformly under high pressure than under low pressure. Moreover, a recent study demonstrated that mechanical stress induces midkine (MK), a novel cytokine, to promote the pathogenesis of pulmonary fibrosis, which is a potential therapeutic target in mechanical stress-induced fibrosis.589
Generally, pulmonary fibrosis initiates from the lung periphery. According to the pathological features of pulmonary fibrosis, researchers hypothesized that the peripheral region of the lung withstand higher TF, thereby increasing ECM stiffness and contributing to pulmonary fibrosis progression and lung cancer.590 Mechanistically, YAP responds to TF to activate mTOR/PI3K/AKT signaling to regulate epithelial cell proliferation, differentiation, and migration.591 In addition, ECM components and stiffness are also important regulators for pulmonary fibrosis. Cross-linking enzymes, including LOX, LOXL, and transglutaminases (TGs), enhance fibroblast accumulation and increase resistance to ECM.592 Meanwhile, TF from alveolar epithelial cells induces ECM deposition, leading to an increase in ECM stiffness. Increased ECM stiffness and TF activate TGFβ and integrin to promote the activation of fibroblasts and epithelial cells in the lung tissue. For example, the scarring tissue in pulmonary fibrosis is from excessive deposition of ECM. The elevated ECM stiffness enhances tank binding protein kinase 1 (TBK1) phosphorylation to activate YAP/TAZ, which is independent of the Hippo signaling pathway. In addition, mouse minute 4 homolog (MDM4) is identified as an ECM stiffness-modulated endogenous inhibitor of p53.593 Targeting MDM4-p53 (myo)fibroblast can help reduce pulmonary matrix through nonenzymatic cross-linking or genetic ablation. Interestingly, fibroblast can also be activated by CD44/RhoA/YAP-mediated mechanics-induced pulmonary fibrosis.311 Recent research demonstrated that ANKED42, a novel circRNA-ankyrin repeat domain 42, sponges miR324-5p to activate AJUBA expression, inhibiting the interaction between phosphorylated YAP and LATS1/2, thus resulting in YAP translocation to the nucleus. Eventually, pulmonary fibrosis is strengthened by mechanical cues-mediated YAP activation.594
Mechanotransduction in liver fibrosis
Liver fibrosis is a pathological process induced by multiple factors and eventually leads to liver dysfunction. The etiologies of liver fibrosis include viral hepatitis B/C, autoimmune liver diseases, non-alcoholic fatty liver disease (NAFLD), hereditary diseases (i.e., Wilson’s disease), and liver cancer.595 Portal hypertension (PH) and ECM stiffness are of the two main mechanical cues in liver fibrosis progression. PH is categorized by presinusoidal, sinusoidal, and post-sinusoidal.596 Sinusoidal compression increase and vascular compliance reduction can lead to elevated portal pressure. In the HP-induced liver fibrosis model, losartan could alleviates mechanotransduction-associated fibrosis in HSCs.21 As a result of continuous abnormal PH stimulation, elevated ECM deposition increases the stiffness of the matrix, which is also a promoter of liver fibrosis. Losartan has already been applied as the therapy for hepatitis C with mild liver fibrosis (NCT00298714) and non-alcoholic steatohepatitis (NCT01051219).
HSCs, hepatocytes, portal fibroblasts, and immunocytes are the main cell types activated during liver fibrosis. Various studies showed different behaviors of hepatocytes on soft supports and dedifferentiated behaviors on stiff supports in liver fibrosis progression. ECM stiffness has also been reported to influence the hepatocytes’ response to growth factors and fibronectin density.597 HSCs are typical mesenchymal cells with the characteristics of both fibroblasts and pericytes. As a major precursor of the myofibroblasts in the liver, quiescent HSCs can be activated by mechanical cues, such as ECM stiffness. The mechanical stimulation transforms HSCs to myofibroblasts.598
Meanwhile, the features of cell proliferation, cell migration, and cell contraction are facilitated by mechanical cues, leading to high secretion of ECM macromolecule. During ECM deposition in the space of Disse, the ECM composition changes from collagen type IV, heparan sulfate proteoglycan, and lamin to fibrillar collagen type I and III.599 The integrinαv, cytoskeleton-dependent actin-myosin, and Rho/GTPases can respond to mechanical stimulations in the liver.263,600 Moreover, ECM stiffness also activates myofibroblastic differentiation in vivo. Both ECM stiffness and TGFβ trigger portal fibroblast activation in biliary fibrosis.601 A recent study showed that ECM stiffness-induced plasma membrane tension can be orchestrated by the integrinβ1/RhoA axis, which promotes the expression of the tissue inhibitors, metalloproteinase 1 (TIMP-1) and caveolin-1, in liver cirrhosis.602 In addition, increased ECM also binds to, hepatocyte growth factor (HGF), fibroblast growth factor (FGF), epidermal growth factor (EGF), and VEGF, facilitating HSC proliferation.603 The crosslinking enzyme lysyl oxidase-like-2 (LOXL2), mainly expressed by HSCs, catalyzes collagens and elastins crosslinking.604,605 LOXL2 is regarded as a therapeutic target in the simtuzumab (GS-6624) testing of phase II clinical trials, such as NCT01707472, NCT01672853, NCT01672866, and NCT01672879.
Mechanotransduction in renal fibrosis
Renal fibrosis is generally caused by renal tubule injury and may result in glomerulosclerosis, tubulointerstitial fibrosis, and angiosclerosis.606 One of the features of renal fibrosis is tissue scarring caused by excessive ECM deposition, which is a common outcome of multiple chronic kidney diseases.607
The kidney cells are living in a blood-shear environment sheltered by the nephron. FSS in the nephron ranges from 0.06 to 0.3 dyn/cm2.608,609 FSS is a typical mechanical cue being discussed in many fibrotic processes. Essig et al. found that FSS (0.17 dyn/cm2) regulates the fibrotic process through decreasing the expression of plasminogen activator and urokinase.609 Moreover, the TGFβ-induced EMT of renal epithelial cells is modulated by mechanical responses, in which process the stretching of renal cells activates MAPK signaling.610
Apart from FSS, TF from urological obstruction also plays an important role in renal fibrosis. The ureteral obstruction-related hydronephrosis leads to elevated intratubular pressure.611 The obstruction leads to a sharp increase in fluid pressure that damages the renal tubules. Although the compensatory dilation of the pelvis can alleviate the immediate pressure increase, the pressure will still cause damage to the nephron gradually. The nephron cells sense the FSS and HP through activated ion channels, thereby promoting renal fibrosis.612 TF is reported to activate TGFβ-induced EMT as well.613,614 The mechanical TF upregulates fibronectin and TGFβ, activates the signal transducer and transcription factor 3 (STAT3), thereby promoting renal fibrosis.615 Meanwhile, excessive ROS can also be generated by the activation of TF, which results in renal injury.616 ROS in turn facilitates the cytoplasmic proline-rich tyrosine kinase 2 (Pyk2), which is parallel to the expression of TGFβ.
There are three main mechanisms between renal epithelial cells and mechanical cues: the torque of a large number of apical microvilli, the bending moment of primary cilia, and the activation of mechanically sensitive ion channels that activate specific signaling.617 It has been reported that cilia might be associated with fluid resorption in response to FSS.618 This indicates that primary cilia may be engaged in the mechanotransduction in renal epithelial cells. Nevertheless, superficial luminal FSS of kidney cells triggers a Ca2+ response without cilium.619,620
In general, fibrotic lesions are distributed at localized fibrogenic niches.621 The ECM-regulated specialized microenvironment mainly includes ECM macromolecules, resident and infiltrating inflammatory cells of the kidney, extracellular vesicles, soluble factors, and metabolites. The pathological ECM environment promotes renal fibroblast proliferation, tubular injury, macrophage activation, and endothelial cell depletion. The ECM mechanical signals can interact with integrin αv, which further activates TGFβ/Smad signaling to promote renal fibrosis.622 In addition, macrophages in the kidney can sense FSS and ECM stiffness to promote renal fibrosis. Mechanical cues-activated Piezo1 channel facilitates the vascular formation and blood pressure modulation to inflammatory responses. A recent study demonstrated that the depletion of Piezo1 can inhibit macrophage inflammation, thereby alleviating renal fibrosis and EMT process.623 YAP is another important sensor in mechanotransduction and has long been considered a critical regulator in myofibroblast transformation. A recent study revealed that inhibition of YTHDF1 can alleviate renal fibrosis by targeting YAP. However, the effective drugs targeting renal fibrosis still remains challenging.
Mechanotransduction in cardiac fibrosis
Cardiac fibrosis is characterized by excessive ECM deposition in the cardiac interstitium, which impairs both systolic and diastolic functions of the cardiac system,624 eventually leading to heart failure.625 Although cardiac fibrosis is always associated with adverse outcomes of myocardial diseases, it is not the primary cause of cardiac dysfunction. Initially, cardiac fibrosis presents a reparative behavior, the formation of collagen-based scar substitutes the myocardiocytes to maintain the elasticity and prevent the infiltration of injury and cardiac rupture,626 such as myocardial infarction. However, fibrosis involves only the interstitium in other cardiac diseases without impairments to cardiomyocytes.
In the human heart, the cardiac ECM is a three-dimensional scaffold that defines the geometry and muscular structure of the cardiac chambers and transmits forces generated during cardiac systolation and diastolation.627 Generally, the cardiac matrix is divided into epimysium, perimysium and endomysium.628 The valve leaflets, chordae tendineae, and collagen matrix of the myocardium are connected by type I collagen and type III collagen fibers. The abnormal mechanical strength initiates the heart remodeling process, with elevated ECM stiffness, fibroblast proliferation, and differentiation, and cardiomyocytes pathological hypertrophy. For instance, blood hypertension triggers the deposition of ECM and disturbance of the mechanical environment of the heart progressively, leading to diastolic dysfunction.
The mechanical environment in the cardiac system is complex, including HP, TF, FSS, and ECM stiffness. Ventricular hypertension is an important factor contributing to cardiac remodeling and fibrosis. For example, single-cell RNA-seq (scRNA-seq), spatial transcriptomics, and genetic perturbation demonstrated that the pressure overload downregulates the expression of high-temperature requirement A serine peptidase 3 (HTRA3) in cardiac fibroblasts and triggers TGFβ-mediated signaling cascades, eventually leading to cardiac fibrosis.629 Similar to other mechanics-induced remodeling processes, the pressure overload activates YAP/TAZ-dependent fibrotic signaling transduction.630 Long-term overload pressure and stretch lead to abnormal ECM deposition in the cardiac interstitium, which results in elevated ECM stiffness. The stiff matrix imposes mechanical stimulation on myofibroblasts to facilitate cardiac fibrosis progression. TRP family proteins are sensitive to mechanical stimulation. For example, ECM stiffness facilitates myofibroblast activation and differentiation.631 In cardiac fibrosis progression, stiff matrix enhances the proliferation of cardiac fibroblasts by activating YAP-mediated TEA-binding domain (TEAD) and RUNX2 transcription.632 More importantly, the cardiac tissue scarring in fibrosis can prevent the heart from rupture. In turn, cardiac fibrosis can not be reversed, eventually leading to heart failure. Hence, recent studies suggested that cultured cardiac fibroblasts can be transformed into cardiomyocytes, which are exciting evidence for the protection of cardiac function.633
Fibroblasts are the main effectors of cardiac fibrosis. Cardiac fibroblast can sense mechanical cues through mechanosensitive receptors, and channels, hence promoting intracellular fibrogenic signaling.634 Previous studies demonstrated that cyclic stretch promotes fibroblast activation and FMT. The inhibition of TGFβ significantly deters TF-induced expression of SMA and collagens by attenuating the phosphorylation of Smad2.635 The cyclic strain of fibroblasts was shown to elevate ECM deposition (i.e., collagens and fibronectin) via protein kinase C and tyrosine kinase signaling.636,637 In addition, ERK1/2 and NF-κB signaling also respond to the cyclic strain on cardiac fibroblasts.638,639 Besides, atrial fibrillation (AF)-associated with abnormal atrial stretch is also a factor contributing to cardiac fibrosis. The chronic atrial overload caused by AF leads to cardiac fibroblast proliferation, apoptosis, and morphology change. The activation of the G-protein-coupled receptor triggers ERK1/2 and JNK signaling via phosphorylation of c-Jun, c-Fos, and Fra-1.640 Further studies are warranted to identify the mechanisms and therapeutic targets for cardiac fibrosis.
Mechanotransduction in cancer cell behavior
Tumor microenvironment (TME) is an important factor affecting pathological changes for multiple tumors. As a type of TME, organ or tissue mechanical properties can change the cancer cell behaviors (i.e., growth, migration, invasion, metastasis, dedifferentiation) and are different in tumors and normal tissues. Hence, cancer cell mechanotransduction signaling cascades are important therapeutic targets for anti-cancer therapy. However, the mechanisms between mechanical cues and cancer cell behaviors are still not fully elucidated.
Mechanisms of mechanical cues-induced cancer cell behavior
Multiple studies have reported that ECM functions in cancer biology. ECM microenvironment serves as not only a nest for cancer and stroma cells but also a reservoir for cytokines and growth factors.641 Apart from the crosstalk between cancer cells and matrix, ECM stiffness is also engaged in the regulation of cancer cells, such as cell proliferation, cell differentiation, cell migration, cell invasion, EMT, and metabolic reprogramming642,643 (Fig. 8). ECM stiffness typically affects the movements of the lipid bilayer and the activation of ion channels. The ion channels transmit biomechanical signals to cell biochemical signaling to promote cancer cell migration.644,645
Fig. 8.
The mechanisms of mechanical cues-induced cancer cell behavior in tumor microenvironment. Mechanical cues induced cancer cell growth, invasion, migration and metastasis in multiple signaling pathways. Cancer-associated fibroblasts engage in ECM remodeling process, which trigger cancer cell migration and dissemination. ECF extracellular fluid, ECM extracellular matrix, EMT epithelial-to-mesenchymal transition, HDAC6 histone deacetylase 6, NHE1 Na+/H+ exchanger 1, TRPV4 transient receptor potential vanilloid 4. This figure was created using Biorender.com
The elasticity of tumor tissue is higher than that of the surrounding normal tissue, which aroused focus from the researchers.646 For tumor growth, the elevated ECM stiffness triggers Rho-dependent cytoskeletal tension that enhances cell adhesion and disrupts cell junctions, thereby promoting cancer cell growth.647 ECM stiffness transmits prolactin signals to protumorigenic Src/FAK and MMP activation.648 Moreover, ECM stiffness could also induces epithelial cell growth via the FAK/Rho/ERK signaling pathway.649 In general, ECM cross-linking is regarded as the barrier preventing cancer cells from migration. However, it has been found that cancer cell migration is facilitated by elevated ECM stiffness. Cancer cell migration is initiated by CAFs, which secret protease to remodel the ECM and break through the ECM barrier.650 CAFs commonly express high-level α-SMA, which may be activated by TGFβ.651 CAFs can also be activated by mechanical cues via MRTF-SRF and YAP-TEAD signaling pathways.652 Together, ECM stiffness, FSS, HP, and TF are mechanical properties that trigger cancer cell behaviors.653
In addition, cancer cell migration from one site to another requires cytoskeleton remodeling. Stiff ECM triggers actomyosin contractility to activate RhoA/mDia1 signaling and microtubule network reconstruction, promoting cancer metastasis.654 EPHA2/LYN protein complex has responded to mechanical cues to regulate EMT and metastasis of cancer. The ligand-independent phosphorylation of ephrin receptor EPHA2 is triggered by high ECM stiffness through recruiting and activating the LYN kinase.655 EMT transcription factor TWIST1 is phosphorylated by LYN and releases TWIST1 to the nucleus, thereby facilitating EMT and invasion. More and more researchers started to regard the mechanical properties of microtubules and their dynamics as the major mechanism for tubulin.656 The change in microtubule properties affects the migration of cancer cells. A recent study showed that ECM stiffness-activated of glutamine catabolism can induce microtubule glutamylation to stabilize the MT network, thereby promoting cancer aggressiveness.657
For cancer dissemination, blood, and lymphatic circulations are important factors contributing to the efficiency of cancer cell transit from the primary tumor, metastasis, and extravasation.658 In the circulatory system, flow rates and FSS affect the survival of cancer cells. After being exposed to FSS in the circulatory system, anti-anoikis properties of the circulating tumor cells (CTCs) can be improved.659 Specific circulatory transition phenotype cancer cells can survive prolonged FSS and lead to cancer dissemination with a shorter survival.660 It has been demonstrated that low FSS can activate β1 integrin trafficking through cytoskeleton reconstruction and ROCK/histone deacetylase 6 (HDAC6)-mediated deacetylation of MT.661 In addition, intratumor pressure, ECM compression, and lack of functional lymphatic vessels can all lead to an increase in HP.662 The increased ECM macromolecules also contribute to elevated ECF elevation, which enhances cell migration and cancer dissemination through NHE1/RhoA/TRPV4-dependent signaling.163
Mechanisms of anti-cancer therapy resistance induced by mechanical cues
Cellular mechanotransduction in TME are critical in cancer immunotherapy and contribute to immunotherapy resistance.663 Mechanical cues lead to immunotherapy resistance by being engaged in the anti-tumor immunity cascade.
The ECM stiffness in the tumor is often 5–10 times that in normal tissue. For example, the normal stiffness of breast tissue is 0.4–2.0 kPa but increases to 12.0 kPa for breast cancer.664 Notably, the stiffness of a tumor changes at different tumor stages. For example, the elasticity of stage III lung adenocarcinoma is higher than that of the stage II.665 Currently, numerous studies have focused on the impact of ECM stiffness on cancer immunotherapy. PD-L1 overexpression is an adaptive immune resistance strategy for immune surveillance escape of cancer cells. It has been demonstrated that lung cancer cells cultured on a 25 kPa medium exhibited an increased expression of PD-L1 compared with those cultured on a softer medium, possibly due to F-actin polymerization.666 Furthermore, high ECM stiffness in TME will also affect immune cell behaviors in the immunotherapy against cancer. In response to ECM stiffness, the natural killer (NK) cells lyse cancer cells to release antigens. Dendritic cells (DCs) and macrophage cells are transformed into immunosuppressive phenotypes, which lead to immunotherapy resistance. ECM stiffness also triggers DCs differentiation and maturation.667 The migration of immune DCs (iDCs) through ECM depends on integrin-based adhesion structure. C-type lectin receptors (CLRs), which are found on the surface of iDCs, are downregulated by the stiff ECM in TME to prevent it from binding with internalized antigens. On the other hand, the degradation of ECM is essential for the migration and accumulation of NK cells.668 Elevated ECM stiffness breaks the balance between NK cell secretions of TIMP and MMP, thereby deterring NK cells to proceed with tumor-killer functions.663 In addition, tumor-associated macrophages (TAMs) are major immune cells in TME. Through a ROCK-independent, podosome-dependent mesenchymal migratory mechanism, the stiff ECM facilitates the transition of TAMs from an M1 to an M2 phenotype. The damage of phagocytosis and the migration ability of TAMs leads to immunotherapy resistance to cancer. For example, macrophage cells are activated by ECM stiffness via RhoA/ROCK signaling to promote the secretion of inhibitory immune factor IL-10. Then M2 phenotype macrophage cells are up-regulated, contributing to immunoresistance.669,670 However, the regulatory mechanisms between immune cells and ECM stiffness in immunotherapy resistance against cancers are still understudied.
Apart from ECM stiffness, other mechanical cues, such as blood or lymphatic vascular FSS in TME, can affect tumor cell biology. The rapidly structured and complicated vessels, as well as the compression of vessels, increase the geometric resistance to flow. Immunotherapy resistance is primarily brought on by high vascular FSS, which reduces T cell extravasation and immune monitoring for cancer cells.
Cancer stem cells (CSCs) are specific cancer cells that are characterized by self-renew and chemoresistance.671 Recently, EMT has been found to contribute to the generation of CSCs. During EMT, the phenotypes of cancer cells transform from epithelial properties (i.e., apical-basal polarity and cell junctions) to mesenchymal properties (i.e., invasion and migration),672 which reduce adhesion and connection of cells and facilitate cancer metastasis. More importantly, mechanical cues are critical factors contributing to the EMT of cancer cells. On one hand, mechanical cues directly drive the EMT process via mechanosensitive effectors (i.e., Piezo, YAP/TAZ, and integrin). For example, YAP promotes the expression of EMT-associated genes to facilitate cancer cell differentiation into CSCs.673 On the other hand, CAFs and TAMs in the CSCs can secret TGFβ to trigger EMT.674 Researchers have found that EMT is a developmental process being exploited by tumor cells. During this process, Axl kinase expression is critical in cancer metastasis.675 The inhibition of Axl can significantly reduce the EMT level, attenuate metastasis, and increase overall survival in breast cancer.676 Several clinical trials are ongoing in anti-cancer therapies targeting Axl-induced EMT, which can be driven by mechanical cues (Table 4).
Table 4.
Clinical trials of anti-cancer therapies targeting Axl
| Methods of drug application | Disease type | Phase | Current status | ClinicalTrials.gov identifier |
|---|---|---|---|---|
| Bemcentinib (before surgery); Bemcentinib (after surgery) | Brain and central nervous system tumors | 1 | Active | NCT03965494 |
| Bemcentinib; docetaxel | Non-small cell lung cancer | 1 | Active | NCT02922777 |
| Bemcentinib; pembrolizumab | Triple-negative breast cancer; inflammatory breast cancer stage IV | 2 | Terminated | NCT03184558 |
| Bemcentinib; cytarabine; decitabine | Acute myeloid leukemia; myelodysplastic syndromes | 1/2 | Active | NCT02488408 |
| Bemcentinib; pembrolizumab | Lung cancer metastatic; NSCLC stage IV; adenocarcinoma of lung | 2 | Completed | NCT03184571 |
| Bemcentinib+pembrolizumab; Bemcentinib+dabrafenib and trametinib; pembrolizumab; dabrafenib and trametinib | Melanoma | 1/2 | Active | NCT02872259 |
| Erlotinib; bemcentinib | Non-small cell lung cancer | 1/2 | Completed | NCT02424617 |
| Bemcentinib | Acute myeloid leukemia; high-risk myelodysplastic syndrome; low-risk myelodysplastic syndrome | 2 | Completed | NCT03824080 |
| Rucaparib; abemaciclib; pembrolizumab and bemcentinib | Mesothelioma, malignant | 2 | Active | NCT03654833 |
Bemcentinib (BGB324) a highly selective inhibitor of Axl
Conclusions and perspectives
Based on the advanced research, the pivotal roles of cellular mechanotransduction in health and diseases are identified in this review. Furthermore, important effectors (i.e., Piezo, integrin, TRPV, and YAP/TAZ) and signaling pathways (i.e., TGFβ/Smad, JAK/STAT, Wnt/β-catenin, ERK1/2, and RhoA/ROCK) responding to mechanical stimulation have been summarized. The comprehensive review of cellular mechanotransduction in biological processes shall assist us in finding mechanisms and therapeutic targets associated with mechanical cues.
Following the development of scRNA-seq, researchers have identified different subtypes of fibroblast, which provides opportunities for precise inhibition of fibrogenic fibroblast.677 Researchers found that diverse subtypes of fibroblasts distribute differently, which function differently as well. Intriguingly, the tissue-specific fibroblast transformed to myofibroblast unexhaustedly. Hence, we speculate that mechanical cues in tissues may drive the migration of stem cells and storage fibroblasts. In addition, during the wound healing process, myofibroblasts aggregate at the frontier location of the wound with high tension and ECM with high elasticity deposition. Meanwhile, the fibroblasts with collagen enrich at the bottom of the wound.678 Hence, by learning the track of (myo)fibroblast in the tissue fibrosis, we may be able to detect a potential therapeutic target that does not affect the function of normal fibroblast.
Recently, the adeno-associated virus (AAV)-mediated gene therapy aroused the attention of researchers for its high safety, low immunogenicity, and long-term efficacy.679–684 With designed promoters, AAV targets specific tissues or organs and inhibits fibrosis. AAV serotype2 vector with miR19b transgene targeting collagen α I promoter was applied in the male Sprague Dawley rat hepatic fibrosis mode, and hepatic injuries were reduced in the treated group.685 Hence, AAV therapy may be a potential treatment targeting fibrosis in the future.
Besides, attention has been paid to the relationship between cellular mechanotransduction and stem cells in regenerative medicine.686 Mechanical cues can regulate stem cell differentiation for potential regenerative therapies for fibrotic diseases.687 Mechanical stress can also modulate stem cell differentiation to cardiovascular cell types,688 mechanical forces improve myocardium regeneration, and mechanical stretch can promote stem cell migration.689
A recent study suggested that mechanosensitive metabolism played a vital role in cancer cell migration and metastasis.690 Interestingly, cooperatively migrating cells alter leaders dynamically to reduce the thermodynamic costs of invasion.691 Thermodynamic evidence provides novel insights into the association between mechanotransduction and cancer cellular energy metabolism during migration, which indicates that cancer cell migrates through the minimal energy intake pathway. These findings provide novel ideas for preventing cancer dissemination. In addition, for the precise therapies of cancer, biomechanical cues are regarded as a novel strategy to reverse cancer immunotherapy resistance. Further research is warranted to investigate the mechanisms between mechanical cues and immunotherapy resistance. The investigation targeting mechanical properties of the tumor will provide approaches on the restriction of cancer cell behaviors.
Mechanobiology is also being widely investigated in 2D configurations. However, limited studies of cell-ECM matrix forces have been performed in 3D systems. Although the 2D system is considered the foundation of mechanical cues-associated pathophysiology, the 3D platform is more similar to the microenvironment of the human body.692,693 A recent study has reported a novel 3D platform consisting of a well-defined synthetic hydrogel system and 3D traction force microscopy to evaluate the matrix environment and force response. The platform provides a novel insight into cell behaviors in mechanotransduction in a 3D system.694 Hence, mechanical transduction in the 3D microenvironment needs more attention in the future.
In summary, as an essential factor contributing to health and diseases in organisms, cellular mechanotransduction widely affects tissue homeostasis, fibrotic diseases, tumorigenesis, metabolism, and others. Therefore, a broader overview of mechanical cues is a great challenge for researchers. This current review presents the mechanisms of mechanical cues-associated pathophysiological processes in organisms and sheds light on the therapeutic targets of multiple diseases.
Acknowledgements
This work was supported by the National Science Fund of China (Grant Number: 82270720); National Clinical Research Center for Geriatrics, West China Hospital, Sichuan University (Grant Number: Y2021LC005); 1.3.5 project for disciplines of excellence, West China Hospital, Sichuan University (Grant Number: ZYGD18011); The National Science Fund of China (Grant Number: 81873601). Many thanks to ClinicalTrials.gov (https://clinicaltrials.gov/ct2/home), which is maintained by the National Library of Medicine (NLM) at the National Institutes of Health (NIH), for the support of clinical trials collection. We appreciate the support from Biorender (https://www.biorender.com) for helping us create some of the figures. We thank Xintong Bao for her kind and excellent help in this study.
Author contributions
K.W. and D.L. designed and supervised the review; X.D. and X.G. drafted the manuscript, X.D., L.P., and J.A. generated figures and tables; S.Q., X.J., and H.L. performed manuscript reviewing and editing. All authors have read and approved the article.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Xingpeng Di, Xiaoshuai Gao, Liao Peng
Contributor Information
Kunjie Wang, Email: wangkj@scu.edu.cn.
Deyi Luo, Email: luodeyi1985@163.com.
References
- 1.Tschumperlin DJ. Mechanotransduction. Compr. Physiol. 2011;1:1057–1073. doi: 10.1002/cphy.c100016. [DOI] [PubMed] [Google Scholar]
- 2.Fernandez-Sanchez M-E, Brunet T, Röper J-C, Farge E. Mechanotransduction’s impact on animal development, evolution, and tumorigenesis. Annu. Rev. Cell Dev. Biol. 2015;31:373–397. doi: 10.1146/annurev-cellbio-102314-112441. [DOI] [PubMed] [Google Scholar]
- 3.Mascharak S, et al. Multi-omic analysis reveals divergent molecular events in scarring and regenerative wound healing. Cell Stem Cell. 2022;29:315–327.e6. doi: 10.1016/j.stem.2021.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marinval N, Chew SY. Mechanotransduction assays for neural regeneration strategies: a focus on glial cells. APL Bioeng. 2021;5:021505. doi: 10.1063/5.0037814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Long Y, Niu Y, Liang K, Du Y. Mechanical communication in fibrosis progression. Trends Cell Biol. 2022;32:70–90. doi: 10.1016/j.tcb.2021.10.002. [DOI] [PubMed] [Google Scholar]
- 6.Cooper J, Giancotti FG. Integrin signaling in cancer: mechanotransduction, stemness, epithelial plasticity, and therapeutic resistance. Cancer Cell. 2019;35:347–367. doi: 10.1016/j.ccell.2019.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhu P, et al. Targeted mechanical forces enhance the effects of tumor immunotherapy by regulating immune cells in the tumor microenvironment. Cancer Biol. Med. 2023;20:44–55. doi: 10.20892/j.issn.2095-3941.2022.0491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Burridge K, Monaghan-Benson E, Graham DM. Mechanotransduction: from the cell surface to the nucleus via RhoA. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2019;374:20180229. doi: 10.1098/rstb.2018.0229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li N, et al. Multiscale biomechanics and mechanotransduction from liver fibrosis to cancer. Adv. Drug Deliv. Rev. 2022;188:114448. doi: 10.1016/j.addr.2022.114448. [DOI] [PubMed] [Google Scholar]
- 10.Heldin C-H, Rubin K, Pietras K, Ostman A. High interstitial fluid pressure - an obstacle in cancer therapy. Nat. Rev. Cancer. 2004;4:806–813. doi: 10.1038/nrc1456. [DOI] [PubMed] [Google Scholar]
- 11.Berg A, Aas P, Gustafsson T, Reed RK. Effect of alpha-trinositol on interstitial fluid pressure, oedema generation and albumin extravasation in experimental frostbite in the rat. Br. J. Pharmacol. 1999;126:1367–1374. doi: 10.1038/sj.bjp.0702442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Abraldes JG, Sarlieve P, Tandon P. Measurement of portal pressure. Clin. Liver Dis. 2014;18:779–792. doi: 10.1016/j.cld.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 13.Henstock JR, Rotherham M, Rose JB, El Haj AJ. Cyclic hydrostatic pressure stimulates enhanced bone development in the foetal chick femur in vitro. Bone. 2013;53:468–477. doi: 10.1016/j.bone.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 14.Neßler KHL, et al. The influence of hydrostatic pressure on tissue engineered bone development. J. Theor. Biol. 2016;394:149–159. doi: 10.1016/j.jtbi.2015.12.020. [DOI] [PubMed] [Google Scholar]
- 15.Karimi N, Kamangar PB, Azadbakht M, Amini A, Amiri I. Effects of hydrostatic pressure on mouse sperm. Bratisl. Lek. Listy. 2014;115:70–75. doi: 10.4149/bll_2014_015. [DOI] [PubMed] [Google Scholar]
- 16.Xu Y, Yazbeck R, Duan C. Anomalous mechanosensitive ion transport in nanoparticle-blocked nanopores. J. Chem. Phys. 2021;154:224702. doi: 10.1063/5.0046086. [DOI] [PubMed] [Google Scholar]
- 17.Fukuchi M, et al. Hydrostatic pressure-regulated cellular calcium responses. Langmuir. 2021;37:820–826. doi: 10.1021/acs.langmuir.0c03141. [DOI] [PubMed] [Google Scholar]
- 18.Sugimoto A, et al. Piezo type mechanosensitive ion channel component 1 functions as a regulator of the cell fate determination of mesenchymal stem cells. Sci. Rep. 2017;7:17696. doi: 10.1038/s41598-017-18089-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li X, et al. High hydrostatic pressure induces atrial electrical remodeling through upregulation of inflammatory cytokines. Life Sci. 2020;242:117209. doi: 10.1016/j.lfs.2019.117209. [DOI] [PubMed] [Google Scholar]
- 20.Huang Z, Khalifa MO, Gu W, Li T-S. Hydrostatic pressure induces profibrotic properties in hepatic stellate cells via the RhoA/ROCK signaling pathway. FEBS Open Bio. 2022;12:1230–1240. doi: 10.1002/2211-5463.13405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang Z, et al. Angiotensin receptor blocker alleviates liver fibrosis by altering the mechanotransduction properties of hepatic stellate cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2022;322:G446–G456. doi: 10.1152/ajpgi.00238.2021. [DOI] [PubMed] [Google Scholar]
- 22.Salker MS, et al. Activation of SGK1 in endometrial epithelial cells in response to PI3K/AKT inhibition impairs embryo implantation. Cell. Physiol. Biochem. 2016;39:2077–2087. doi: 10.1159/000447903. [DOI] [PubMed] [Google Scholar]
- 23.Mihic A, et al. The effect of cyclic stretch on maturation and 3D tissue formation of human embryonic stem cell-derived cardiomyocytes. Biomaterials. 2014;35:2798–2808. doi: 10.1016/j.biomaterials.2013.12.052. [DOI] [PubMed] [Google Scholar]
- 24.Matuszewski MA, et al. Uroplakins and their potential applications in urology. Cent. Eur. J. Urol. 2016;69:252–257. doi: 10.5173/ceju.2016.638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liang Z, et al. Hydrostatic pressure and muscarinic receptors are involved in the release of inflammatory cytokines in human bladder smooth muscle cells. Neurourol. Urodyn. 2017;36:1261–1269. doi: 10.1002/nau.23104. [DOI] [PubMed] [Google Scholar]
- 26.Gao X, et al. Cyclic hydrostatic pressure promotes uroplakin expression in human urothelial cells through activation of ERK1/2 signaling. Biochem. Biophys. Res. Commun. 2018;503:2499–2503. doi: 10.1016/j.bbrc.2018.07.006. [DOI] [PubMed] [Google Scholar]
- 27.Vozzi F, Bianchi F, Ahluwalia A, Domenici C. Hydrostatic pressure and shear stress affect endothelin-1 and nitric oxide release by endothelial cells in bioreactors. Biotechnol. J. 2014;9:146–154. doi: 10.1002/biot.201300016. [DOI] [PubMed] [Google Scholar]
- 28.Sigala S, et al. Differential gene expression of cholinergic muscarinic receptor subtypes in male and female normal human urinary bladder. Urology. 2002;60:719–725. doi: 10.1016/S0090-4295(02)01819-8. [DOI] [PubMed] [Google Scholar]
- 29.Hegde SS, Eglen RM. Muscarinic receptor subtypes modulating smooth muscle contractility in the urinary bladder. Life Sci. 1999;64:419–428. doi: 10.1016/S0024-3205(98)00581-5. [DOI] [PubMed] [Google Scholar]
- 30.Wei W, et al. Involvement of Smad3 pathway in atrial fibrosis induced by elevated hydrostatic pressure. J. Cell. Physiol. 2018;233:4981–4989. doi: 10.1002/jcp.26337. [DOI] [PubMed] [Google Scholar]
- 31.Kao Y-C, Chen Z-H, Wang W-Y, Lee C-H, Kuo P-L. Hydrostatic pressure promotes migration and filamin-A activation in fibroblasts with increased p38 phosphorylation and TGF-β production. Biochem. Biophys. Res. Commun. 2021;568:15–22. doi: 10.1016/j.bbrc.2021.06.055. [DOI] [PubMed] [Google Scholar]
- 32.Acevedo AD, Bowser SS, Gerritsen ME, Bizios R. Morphological and proliferative responses of endothelial cells to hydrostatic pressure: role of fibroblast growth factor. J. Cell. Physiol. 1993;157:603–614. doi: 10.1002/jcp.1041570321. [DOI] [PubMed] [Google Scholar]
- 33.Zainal Abidin NA, et al. A microfluidic method to investigate platelet mechanotransduction under extensional strain. Res. Pract. Thromb. Haemost. 2023;7:100037. doi: 10.1016/j.rpth.2023.100037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Feinberg MW, Moore KJ. MicroRNA regulation of atherosclerosis. Circ. Res. 2016;118:703–720. doi: 10.1161/CIRCRESAHA.115.306300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yamawaki H, Pan S, Lee RT, Berk BC. Fluid shear stress inhibits vascular inflammation by decreasing thioredoxin-interacting protein in endothelial cells. J. Clin. Investig. 2005;115:733–738. doi: 10.1172/JCI200523001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chiu J-J, et al. Analysis of the effect of disturbed flow on monocytic adhesion to endothelial cells. J. Biomech. 2003;36:1883–1895. doi: 10.1016/S0021-9290(03)00210-0. [DOI] [PubMed] [Google Scholar]
- 37.Gray KM, Stroka KM. Vascular endothelial cell mechanosensing: New insights gained from biomimetic microfluidic models. Semin. Cell Dev. Biol. 2017;71:106–117. doi: 10.1016/j.semcdb.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 38.Tarbell JM, Shi Z-D, Dunn J, Jo H. Fluid mechanics, arterial disease, and gene expression. Annu. Rev. Fluid Mech. 2014;46:591–614. doi: 10.1146/annurev-fluid-010313-141309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhou J, Li Y-S, Chien S. Shear stress-initiated signaling and its regulation of endothelial function. Arterioscler. Thromb. Vasc. Biol. 2014;34:2191–2198. doi: 10.1161/ATVBAHA.114.303422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Roux E, Bougaran P, Dufourcq P, Couffinhal T. Fluid shear stress sensing by the endothelial layer. Front. Physiol. 2020;11:861. doi: 10.3389/fphys.2020.00861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kutikhin AG, Sinitsky MY, Yuzhalin AE, Velikanova EA. Shear stress: an essential driver of endothelial progenitor cells. J. Mol. Cell. Cardiol. 2018;118:46–69. doi: 10.1016/j.yjmcc.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 42.Papaioannou TG, Stefanadis C. Vascular wall shear stress: basic principles and methods. Hellenic J. Cardiol. 2005;46:9–15. [PubMed] [Google Scholar]
- 43.Caro CG. Discovery of the role of wall shear in atherosclerosis. Atheroscler. Thromb. Vasc. Biol. 2009;29:158–161. doi: 10.1161/ATVBAHA.108.166736. [DOI] [PubMed] [Google Scholar]
- 44.Fujiwara K. Platelet endothelial cell adhesion molecule-1 and mechanotransduction in vascular endothelial cells. J. Intern. Med. 2006;259:373–380. doi: 10.1111/j.1365-2796.2006.01623.x. [DOI] [PubMed] [Google Scholar]
- 45.Liu Z, et al. Single-cell RNA sequencing reveals endothelial cell transcriptome heterogeneity under homeostatic laminar flow. Arterioscler. Thromb. Vasc. Biol. 2021;41:2575–2584. doi: 10.1161/ATVBAHA.121.316797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Shinge SAU, Zhang D, Achu Muluh T, Nie Y, Yu F. Mechanosensitive Piezo1 channel evoked-mechanical signals in atherosclerosis. J. Inflamm. Res. 2021;14:3621–3636. doi: 10.2147/JIR.S319789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Paudel R, Fusi L, Schmidt M. The MEK5/ERK5 pathway in health and disease. Int. J. Mol. Sci. 2021;22:7594. doi: 10.3390/ijms22147594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Givens C, Tzima E. Endothelial mechanosignaling: does one sensor fit all? Antioxid. Redox Signal. 2016;25:373–388. doi: 10.1089/ars.2015.6493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Poelmann RE, Gittenberger-de Groot AC. Hemodynamics in cardiac development. J. Cardiovasc Dev. Dis. 2018;5:54. doi: 10.3390/jcdd5040054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nilius B, Viana F, Droogmans G. Ion channels in vascular endothelium. Annu. Rev. Physiol. 1997;59:145–170. doi: 10.1146/annurev.physiol.59.1.145. [DOI] [PubMed] [Google Scholar]
- 51.Olesen SP, Clapham DE, Davies PF. Haemodynamic shear stress activates a K+ current in vascular endothelial cells. Nature. 1988;331:168–170. doi: 10.1038/331168a0. [DOI] [PubMed] [Google Scholar]
- 52.Schwarz G, Droogmans G, Nilius B. Shear stress induced membrane currents and calcium transients in human vascular endothelial cells. Pflug. Arch. 1992;421:394–396. doi: 10.1007/BF00374230. [DOI] [PubMed] [Google Scholar]
- 53.Baeyens N, et al. Syndecan 4 is required for endothelial alignment in flow and atheroprotective signaling. Proc. Natl Acad. Sci. USA. 2014;111:17308–17313. doi: 10.1073/pnas.1413725111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chen J, et al. αvβ3 Integrins mediate flow-induced NF-κB activation, proinflammatory gene expression, and early atherogenic inflammation. Am. J. Pathol. 2015;185:2575–2589. doi: 10.1016/j.ajpath.2015.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Tzima E, et al. A mechanosensory complex that mediates the endothelial cell response to fluid shear stress. Nature. 2005;437:426–431. doi: 10.1038/nature03952. [DOI] [PubMed] [Google Scholar]
- 56.Tzima E, del Pozo MA, Shattil SJ, Chien S, Schwartz MA. Activation of integrins in endothelial cells by fluid shear stress mediates Rho-dependent cytoskeletal alignment. EMBO J. 2001;20:4639–4647. doi: 10.1093/emboj/20.17.4639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yamamoto K, Ando J. New molecular mechanisms for cardiovascular disease:blood flow sensing mechanism in vascular endothelial cells. J. Pharm. Sci. 2011;116:323–331. doi: 10.1254/jphs.10R29FM. [DOI] [PubMed] [Google Scholar]
- 58.Jetta D, Gottlieb PA, Verma D, Sachs F, Hua SZ. Shear stress-induced nuclear shrinkage through activation of Piezo1 channels in epithelial cells. J. Cell Sci. 2019;132:jcs226076. doi: 10.1242/jcs.226076. [DOI] [PubMed] [Google Scholar]
- 59.Conway DE, et al. Fluid shear stress on endothelial cells modulates mechanical tension across VE-cadherin and PECAM-1. Curr. Biol. 2013;23:1024–1030. doi: 10.1016/j.cub.2013.04.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tzima E. Role of small GTPases in endothelial cytoskeletal dynamics and the shear stress response. Circ. Res. 2006;98:176–185. doi: 10.1161/01.RES.0000200162.94463.d7. [DOI] [PubMed] [Google Scholar]
- 61.Shiu Y-T, et al. Rho mediates the shear-enhancement of endothelial cell migration and traction force generation. Biophys. J. 2004;86:2558–2565. doi: 10.1016/S0006-3495(04)74311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Obi S, et al. Fluid shear stress induces differentiation of circulating phenotype endothelial progenitor cells. Am. J. Physiol. Cell Physiol. 2012;303:C595–C606. doi: 10.1152/ajpcell.00133.2012. [DOI] [PubMed] [Google Scholar]
- 63.Xanthis I, et al. β1 integrin is a sensor of blood flow direction. J. Cell Sci. 2019;132:jcs229542. doi: 10.1242/jcs.229542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kang N. Mechanotransduction in liver diseases. Semin Liver Dis. 2020;40:84–90. doi: 10.1055/s-0039-3399502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chatzizisis YS, et al. Role of endothelial shear stress in the natural history of coronary atherosclerosis and vascular remodeling: molecular, cellular, and vascular behavior. J. Am. Coll. Cardiol. 2007;49:2379–2393. doi: 10.1016/j.jacc.2007.02.059. [DOI] [PubMed] [Google Scholar]
- 66.Noguchi T, Matozaki T, Horita K, Fujioka Y, Kasuga M. Role of SH-PTP2, a protein-tyrosine phosphatase with Src homology 2 domains, in insulin-stimulated Ras activation. Mol. Cell Biol. 1994;14:6674–6682. doi: 10.1128/mcb.14.10.6674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Yang Z, et al. Shear stress-induced activation of Tie2-dependent signaling pathway enhances reendothelialization capacity of early endothelial progenitor cells. J. Mol. Cell Cardiol. 2012;52:1155–1163. doi: 10.1016/j.yjmcc.2012.01.019. [DOI] [PubMed] [Google Scholar]
- 68.Di Ieva A, et al. MicroRNAs as biomarkers in pituitary tumors. Neurosurgery. 2014;75:181–9. doi: 10.1227/NEU.0000000000000369. [DOI] [PubMed] [Google Scholar]
- 69.Schober A, Maleki SS, Nazari-Jahantigh M. Regulatory non-coding RNAs in atherosclerosis. Handb. Exp. Pharmacol. 2022;270:463–492. doi: 10.1007/164_2020_423. [DOI] [PubMed] [Google Scholar]
- 70.de Gonzalo-Calvo D, et al. Circulating non-coding RNAs in biomarker-guided cardiovascular therapy: a novel tool for personalized medicine? Eur. Heart J. 2019;40:1643–1650. doi: 10.1093/eurheartj/ehy234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Marin T, et al. Mechanosensitive microRNAs-role in endothelial responses to shear stress and redox state. Free Radic. Biol. Med. 2013;64:61–68. doi: 10.1016/j.freeradbiomed.2013.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cardona-Monzonís A, et al. Non-coding RNAs and coronary artery disease. Adv. Exp. Med. Biol. 2020;1229:273–285. doi: 10.1007/978-981-15-1671-9_16. [DOI] [PubMed] [Google Scholar]
- 73.Wang T, et al. Comprehensive analysis of differentially expressed mRNA and circRNA in ankylosing spondylitis patients’ platelets. Exp. Cell Res. 2021;409:112895. doi: 10.1016/j.yexcr.2021.112895. [DOI] [PubMed] [Google Scholar]
- 74.De Rosa S, et al. Flow-responsive noncoding RNAs in the vascular system: basic mechanisms for the clinician. J. Clin. Med. 2022;11:459. doi: 10.3390/jcm11020459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Neth P, Nazari-Jahantigh M, Schober A, Weber C. MicroRNAs in flow-dependent vascular remodelling. Cardiovasc. Res. 2013;99:294–303. doi: 10.1093/cvr/cvt096. [DOI] [PubMed] [Google Scholar]
- 76.Zakkar M, Angelini GD, Emanueli C. Regulation of vascular endothelium inflammatory signalling by shear stress. Curr. Vasc. Pharm. 2016;14:181–186. doi: 10.2174/1570161114666151202205139. [DOI] [PubMed] [Google Scholar]
- 77.Suárez Y, Wang C, Manes TD, Pober JS. Cutting edge: TNF-induced microRNAs regulate TNF-induced expression of E-selectin and intercellular adhesion molecule-1 on human endothelial cells: feedback control of inflammation. J. Immunol. 2010;184:21–25. doi: 10.4049/jimmunol.0902369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Leychenko A, Konorev E, Jijiwa M, Matter ML. Stretch-induced hypertrophy activates NFkB-mediated VEGF secretion in adult cardiomyocytes. PLoS ONE. 2011;6:e29055. doi: 10.1371/journal.pone.0029055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lemarié CA, Tharaux P-L, Lehoux S. Extracellular matrix alterations in hypertensive vascular remodeling. J. Mol. Cell Cardiol. 2010;48:433–439. doi: 10.1016/j.yjmcc.2009.09.018. [DOI] [PubMed] [Google Scholar]
- 80.Tijore A, et al. Selective killing of transformed cells by mechanical stretch. Biomaterials. 2021;275:120866. doi: 10.1016/j.biomaterials.2021.120866. [DOI] [PubMed] [Google Scholar]
- 81.Opplert J, Babault N. Acute effects of dynamic stretching on muscle flexibility and performance: an analysis of the current literature. Sports Med. 2018;48:299–325. doi: 10.1007/s40279-017-0797-9. [DOI] [PubMed] [Google Scholar]
- 82.Rysä J, Tokola H, Ruskoaho H. Mechanical stretch induced transcriptomic profiles in cardiac myocytes. Sci. Rep. 2018;8:4733. doi: 10.1038/s41598-018-23042-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Albinsson S, Bhattachariya A, Hellstrand P. Stretch-dependent smooth muscle differentiation in the portal vein-role of actin polymerization, calcium signaling, and microRNAs. Microcirculation. 2014;21:230–238. doi: 10.1111/micc.12106. [DOI] [PubMed] [Google Scholar]
- 84.Gao X, et al. Physiological stretch induced proliferation of human urothelial cells via integrin α6-FAK signaling pathway. Neurourol. Urodyn. 2018;37:2114–2120. doi: 10.1002/nau.23572. [DOI] [PubMed] [Google Scholar]
- 85.Loverde JR, Tolentino RE, Soteropoulos P, Pfister BJ. Biomechanical forces regulate gene transcription during stretch-mediated growth of mammalian neurons. Front. Neurosci. 2020;14:600136. doi: 10.3389/fnins.2020.600136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Purohit PK, Smith DH. A model for stretch growth of neurons. J. Biomech. 2016;49:3934–3942. doi: 10.1016/j.jbiomech.2016.11.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Liu B, et al. Role of cyclic strain frequency in regulating the alignment of vascular smooth muscle cells in vitro. Biophys. J. 2008;94:1497–1507. doi: 10.1529/biophysj.106.098574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chen Q, Li W, Quan Z, Sumpio BE. Modulation of vascular smooth muscle cell alignment by cyclic strain is dependent on reactive oxygen species and P38 mitogen-activated protein kinase. J. Vasc. Surg. 2003;37:660–668. doi: 10.1067/mva.2003.95. [DOI] [PubMed] [Google Scholar]
- 89.Tock J, Van Putten V, Stenmark KR, Nemenoff RA. Induction of SM-alpha-actin expression by mechanical strain in adult vascular smooth muscle cells is mediated through activation of JNK and p38 MAP kinase. Biochem. Biophys. Res. Commun. 2003;301:1116–1121. doi: 10.1016/S0006-291X(03)00087-1. [DOI] [PubMed] [Google Scholar]
- 90.Numaguchi K, Eguchi S, Yamakawa T, Motley ED, Inagami T. Mechanotransduction of rat aortic vascular smooth muscle cells requires RhoA and intact actin filaments. Circ. Res. 1999;85:5–11. doi: 10.1161/01.RES.85.1.5. [DOI] [PubMed] [Google Scholar]
- 91.Kirkland NJ, et al. Tissue mechanics regulate mitotic nuclear dynamics during epithelial development. Curr. Biol. 2020;30:2419–2432.e4. doi: 10.1016/j.cub.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Chang W, et al. Imbalanced nucleocytoskeletal connections create common polarity defects in progeria and physiological aging. Proc. Natl Acad. Sci. USA. 2019;116:3578–3583. doi: 10.1073/pnas.1809683116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Petrie RJ, Koo H, Yamada KM. Generation of compartmentalized pressure by a nuclear piston governs cell motility in a 3D matrix. Science. 2014;345:1062–1065. doi: 10.1126/science.1256965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Nava MM, et al. Heterochromatin-driven nuclear softening protects the genome against mechanical stress-induced damage. Cell. 2020;181:800.e22–817.e22. doi: 10.1016/j.cell.2020.03.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Booth A, Jr, et al. Contractile acto-myosin network on nuclear envelope remnants positions human chromosomes for mitosis. Elife. 2019;8:e46902. doi: 10.7554/eLife.46902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Janin A, Bauer D, Ratti F, Millat G, Méjat A. Nuclear envelopathies: a complex LINC between nuclear envelope and pathology. Orphanet J. Rare Dis. 2017;12:147. doi: 10.1186/s13023-017-0698-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Xie W, et al. A-type lamins form distinct filamentous networks with differential nuclear pore complex associations. Curr. Biol. 2016;26:2651–2658. doi: 10.1016/j.cub.2016.07.049. [DOI] [PubMed] [Google Scholar]
- 98.Shimi T, et al. Structural organization of nuclear lamins A, C, B1, and B2 revealed by superresolution microscopy. Mol. Biol. Cell. 2015;26:4075–4086. doi: 10.1091/mbc.E15-07-0461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.de Leeuw R, Gruenbaum Y, Medalia O. Nuclear lamins: thin filaments with major functions. Trends Cell Biol. 2018;28:34–45. doi: 10.1016/j.tcb.2017.08.004. [DOI] [PubMed] [Google Scholar]
- 100.Dechat T, Adam SA, Taimen P, Shimi T, Goldman RD. Nuclear lamins. Cold Spring Harb. Perspect. Biol. 2010;2:a000547. doi: 10.1101/cshperspect.a000547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Shimi T, et al. The A- and B-type nuclear lamin networks: microdomains involved in chromatin organization and transcription. Genes Dev. 2008;22:3409–3421. doi: 10.1101/gad.1735208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Xu Q, et al. Silencing of Nesprin-2 inhibits the differentiation of myofibroblasts from fibroblasts induced by mechanical stretch. Int. Wound J. 2022;19:978–986. doi: 10.1111/iwj.13694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Walker M, Godin M, Pelling AE. Mechanical stretch sustains myofibroblast phenotype and function in microtissues through latent TGF-β1 activation. Integr. Biol. 2020;12:199–210. doi: 10.1093/intbio/zyaa015. [DOI] [PubMed] [Google Scholar]
- 104.Du G, et al. Roles of TRPV4 and piezo channels in stretch-evoked Ca response in chondrocytes. Exp. Biol. Med. 2020;245:180–189. doi: 10.1177/1535370219892601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chen N, et al. microRNA-21 contributes to orthodontic tooth movement. J. Dent. Res. 2016;95:1425–1433. doi: 10.1177/0022034516657043. [DOI] [PubMed] [Google Scholar]
- 106.Meng X, Wang W, Wang X. MicroRNA-34a and microRNA-146a target CELF3 and suppress the osteogenic differentiation of periodontal ligament stem cells under cyclic mechanical stretch. J. Dent. Sci. 2022;17:1281–1291. doi: 10.1016/j.jds.2021.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Dasgupta T, Ladd AN. The importance of CELF control: molecular and biological roles of the CUG-BP, Elav-like family of RNA-binding proteins. Wiley Interdiscip. Rev. RNA. 2012;3:104–121. doi: 10.1002/wrna.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ladd AN. CUG-BP, Elav-like family (CELF)-mediated alternative splicing regulation in the brain during health and disease. Mol. Cell Neurosci. 2013;56:456–464. doi: 10.1016/j.mcn.2012.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Theocharis AD, Skandalis SS, Gialeli C, Karamanos NK. Extracellular matrix structure. Adv. Drug Deliv. Rev. 2016;97:4–27. doi: 10.1016/j.addr.2015.11.001. [DOI] [PubMed] [Google Scholar]
- 110.Marchioni A, et al. Pulmonary stretch and lung mechanotransduction: implications for progression in the fibrotic lung. Int. J. Mol. Sci. 2021;22:6443. doi: 10.3390/ijms22126443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Karsdal MA, et al. The good and the bad collagens of fibrosis - their role in signaling and organ function. Adv. Drug Deliv. Rev. 2017;121:43–56. doi: 10.1016/j.addr.2017.07.014. [DOI] [PubMed] [Google Scholar]
- 112.López B, et al. Diffuse myocardial fibrosis: mechanisms, diagnosis and therapeutic approaches. Nat. Rev. Cardiol. 2021;18:479–498. doi: 10.1038/s41569-020-00504-1. [DOI] [PubMed] [Google Scholar]
- 113.López B, et al. Association of cardiotrophin-1 with myocardial fibrosis in hypertensive patients with heart failure. Hypertension. 2014;63:483–489. doi: 10.1161/HYPERTENSIONAHA.113.02654. [DOI] [PubMed] [Google Scholar]
- 114.Echegaray K, et al. Role of myocardial collagen in severe aortic stenosis with preserved ejection fraction and symptoms of heart failure. Rev. Esp. Cardiol. 2017;70:832–840. doi: 10.1016/j.recesp.2016.12.018. [DOI] [PubMed] [Google Scholar]
- 115.Kisseleva T, Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat. Rev. Gastroenterol. Hepatol. 2021;18:151–166. doi: 10.1038/s41575-020-00372-7. [DOI] [PubMed] [Google Scholar]
- 116.Bataller R, Brenner DA. Liver fibrosis. J. Clin. Invest. 2005;115:209–218. doi: 10.1172/JCI24282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Chuliá-Peris L, et al. Matrix metalloproteinases and their inhibitors in pulmonary fibrosis: EMMPRIN/CD147 comes into play. Int. J. Mol. Sci. 2022;23:6894. doi: 10.3390/ijms23136894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Liu F, et al. Feedback amplification of fibrosis through matrix stiffening and COX-2 suppression. J. Cell Biol. 2010;190:693–706. doi: 10.1083/jcb.201004082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Beekmans SV, Iannuzzi D. Characterizing tissue stiffness at the tip of a rigid needle using an opto-mechanical force sensor. Biomed. Microdevices. 2016;18:15. doi: 10.1007/s10544-016-0039-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Xia T, et al. Effect of substrate stiffness on hepatocyte migration and cellular Young’s modulus. J. Cell. Physiol. 2018;233:6996–7006. doi: 10.1002/jcp.26491. [DOI] [PubMed] [Google Scholar]
- 121.Takeda T, et al. Usefulness of noninvasive transient elastography for assessment of liver fibrosis stage in chronic hepatitis C. World J. Gastroenterol. 2006;12:7768–7773. doi: 10.3748/wjg.v12.i48.7768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Georges PC, Janmey PA. Cell type-specific response to growth on soft materials. J. Appl. Physiol. 2005;98:1547–1553. doi: 10.1152/japplphysiol.01121.2004. [DOI] [PubMed] [Google Scholar]
- 123.Flanagan LA, Ju Y-E, Marg B, Osterfield M, Janmey PA. Neurite branching on deformable substrates. Neuroreport. 2002;13:2411–2415. doi: 10.1097/00001756-200212200-00007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.van Putten S, Shafieyan Y, Hinz B. Mechanical control of cardiac myofibroblasts. J. Mol. Cell Cardiol. 2016;93:133–142. doi: 10.1016/j.yjmcc.2015.11.025. [DOI] [PubMed] [Google Scholar]
- 125.Júnior C, et al. Baseline stiffness modulates the non-linear response to stretch of the extracellular matrix in pulmonary fibrosis. Int. J. Mol. Sci. 2021;22:12928. doi: 10.3390/ijms222312928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Shao Y, et al. Effects of extracellular matrix softening on vascular smooth muscle cell dysfunction. Cardiovasc. Toxicol. 2020;20:548–556. doi: 10.1007/s12012-020-09580-8. [DOI] [PubMed] [Google Scholar]
- 127.Zhang W, et al. Matrix stiffness and its influence on pancreatic diseases. Biochim. Biophys. Acta Rev. Cancer. 2021;1876:188583. doi: 10.1016/j.bbcan.2021.188583. [DOI] [PubMed] [Google Scholar]
- 128.Mullen CA, Haugh MG, Schaffler MB, Majeska RJ, McNamara LM. Osteocyte differentiation is regulated by extracellular matrix stiffness and intercellular separation. J. Mech. Behav. Biomed. Mater. 2013;28:183–194. doi: 10.1016/j.jmbbm.2013.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Piersma B, Hayward MK, Weaver VM. Fibrosis and cancer: a strained relationship. Biochim. Biophys. Acta Rev. Cancer. 2020;1873:188356. doi: 10.1016/j.bbcan.2020.188356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Najafi M, Farhood B, Mortezaee K. Extracellular matrix (ECM) stiffness and degradation as cancer drivers. J. Cell Biochem. 2019;120:2782–2790. doi: 10.1002/jcb.27681. [DOI] [PubMed] [Google Scholar]
- 131.Zanotelli MR, Reinhart-King CA. Mechanical forces in tumor angiogenesis. Adv. Exp. Med. Biol. 2018;1092:91–112. doi: 10.1007/978-3-319-95294-9_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Gensbittel V, et al. Mechanical adaptability of tumor cells in metastasis. Dev. Cell. 2021;56:164–179. doi: 10.1016/j.devcel.2020.10.011. [DOI] [PubMed] [Google Scholar]
- 133.Pang M, et al. Substrate stiffness promotes latent TGF-β1 activation in hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2017;483:553–558. doi: 10.1016/j.bbrc.2016.12.107. [DOI] [PubMed] [Google Scholar]
- 134.Weniger M, Honselmann KC, Liss AS. The extracellular matrix and pancreatic cancer: a complex relationship. Cancers. 2018;10:316. doi: 10.3390/cancers10090316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Eble JA, Niland S. The extracellular matrix in tumor progression and metastasis. Clin. Exp. Metastasis. 2019;36:171–198. doi: 10.1007/s10585-019-09966-1. [DOI] [PubMed] [Google Scholar]
- 136.Xie Y, et al. Extracellular matrix remodeling in stem cell culture: a potential target for regulating stem cell function. Tissue Eng. Part B Rev. 2022;28:542–554. doi: 10.1089/ten.teb.2021.0066. [DOI] [PubMed] [Google Scholar]
- 137.Kechagia JZ, Ivaska J, Roca-Cusachs P. Integrins as biomechanical sensors of the microenvironment. Nat. Rev. Mol. Cell Biol. 2019;20:457–473. doi: 10.1038/s41580-019-0134-2. [DOI] [PubMed] [Google Scholar]
- 138.Harburger DS, Calderwood DA. Integrin signalling at a glance. J. Cell Sci. 2009;122:159–163. doi: 10.1242/jcs.018093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Moretti L, Stalfort J, Barker TH, Abebayehu D. The interplay of fibroblasts, the extracellular matrix, and inflammation in scar formation. J. Biol. Chem. 2022;298:101530. doi: 10.1016/j.jbc.2021.101530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Maldonado H, Hagood JS. Cooperative signaling between integrins and growth factor receptors in fibrosis. J. Mol. Med. 2021;99:213–224. doi: 10.1007/s00109-020-02026-2. [DOI] [PubMed] [Google Scholar]
- 141.Brownfield DG, et al. Patterned collagen fibers orient branching mammary epithelium through distinct signaling modules. Curr. Biol. 2013;23:703–709. doi: 10.1016/j.cub.2013.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Bonnans C, Chou J, Werb Z. Remodelling the extracellular matrix in development and disease. Nat. Rev. Mol. Cell Biol. 2014;15:786–801. doi: 10.1038/nrm3904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Onodera T, et al. Btbd7 regulates epithelial cell dynamics and branching morphogenesis. Science. 2010;329:562–565. doi: 10.1126/science.1191880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wegener KL, et al. Structural basis of integrin activation by talin. Cell. 2007;128:171–182. doi: 10.1016/j.cell.2006.10.048. [DOI] [PubMed] [Google Scholar]
- 145.Kim C, Ye F, Hu X, Ginsberg MH. Talin activates integrins by altering the topology of the β transmembrane domain. J. Cell Biol. 2012;197:605–611. doi: 10.1083/jcb.201112141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Mitra SK, Schlaepfer DD. Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr. Opin. Cell Biol. 2006;18:516–523. doi: 10.1016/j.ceb.2006.08.011. [DOI] [PubMed] [Google Scholar]
- 147.Clemente CFMZ, et al. Targeting focal adhesion kinase with small interfering RNA prevents and reverses load-induced cardiac hypertrophy in mice. Circ. Res. 2007;101:1339–1348. doi: 10.1161/CIRCRESAHA.107.160978. [DOI] [PubMed] [Google Scholar]
- 148.Li R, Frangogiannis NG. Integrins in cardiac fibrosis. J. Mol. Cell Cardiol. 2022;172:1–13. doi: 10.1016/j.yjmcc.2022.07.006. [DOI] [PubMed] [Google Scholar]
- 149.Dupont S, et al. Role of YAP/TAZ in mechanotransduction. Nature. 2011;474:179–183. doi: 10.1038/nature10137. [DOI] [PubMed] [Google Scholar]
- 150.Lampi MC, Reinhart-King CA. Targeting extracellular matrix stiffness to attenuate disease: From molecular mechanisms to clinical trials. Sci. Transl. Med. 2018;10:eaao0475. doi: 10.1126/scitranslmed.aao0475. [DOI] [PubMed] [Google Scholar]
- 151.Liu F, et al. Mechanosignaling through YAP and TAZ drives fibroblast activation and fibrosis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015;308:L344–L357. doi: 10.1152/ajplung.00300.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Santos A, Lagares D. Matrix stiffness: the conductor of organ fibrosis. Curr. Rheumatol. Rep. 2018;20:2. doi: 10.1007/s11926-018-0710-z. [DOI] [PubMed] [Google Scholar]
- 153.Du J, et al. Extracellular matrix stiffness dictates Wnt expression through integrin pathway. Sci. Rep. 2016;6:20395. doi: 10.1038/srep20395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Okamoto T, et al. Reduced substrate stiffness promotes M2-like macrophage activation and enhances peroxisome proliferator-activated receptor γ expression. Exp. Cell Res. 2018;367:264–273. doi: 10.1016/j.yexcr.2018.04.005. [DOI] [PubMed] [Google Scholar]
- 155.Jia X, et al. Stimulation of vascular smooth muscle cell proliferation by stiff matrix via the IK channel-dependent Ca signaling. J. Cell. Physiol. 2021;236:6897–6906. doi: 10.1002/jcp.30349. [DOI] [PubMed] [Google Scholar]
- 156.Sun Q, et al. Curved nanofiber network induces cellular bridge formation to promote stem cell mechanotransduction. Adv. Sci. 2022;10:e2204479. doi: 10.1002/advs.202204479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Wang N, Tytell JD, Ingber DE. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat. Rev. Mol. Cell Biol. 2009;10:75–82. doi: 10.1038/nrm2594. [DOI] [PubMed] [Google Scholar]
- 158.Davidson PM, Cadot B. Actin on and around the nucleus. Trends Cell Biol. 2021;31:211–223. doi: 10.1016/j.tcb.2020.11.009. [DOI] [PubMed] [Google Scholar]
- 159.Fedorchak GR, Kaminski A, Lammerding J. Cellular mechanosensing: getting to the nucleus of it all. Prog. Biophys. Mol. Biol. 2014;115:76–92. doi: 10.1016/j.pbiomolbio.2014.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Iyer KV, Pulford S, Mogilner A, Shivashankar GV. Mechanical activation of cells induces chromatin remodeling preceding MKL nuclear transport. Biophys. J. 2012;103:1416–1428. doi: 10.1016/j.bpj.2012.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Ho CY, Lammerding J. Lamins at a glance. J. Cell Sci. 2012;125:2087–2093. doi: 10.1242/jcs.087288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Maurer M, Lammerding J. The driving force: nuclear mechanotransduction in cellular function, fate, and disease. Annu. Rev. Biomed. Eng. 2019;21:443–468. doi: 10.1146/annurev-bioeng-060418-052139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Bera K, et al. Extracellular fluid viscosity enhances cell migration and cancer dissemination. Nature. 2022;611:365–373. doi: 10.1038/s41586-022-05394-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Xia H, et al. Na+/H+ exchanger 1, a potential therapeutic drug target for cardiac hypertrophy and heart failure. Pharm. (Basel) 2022;15:875. doi: 10.3390/ph15070875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Gonzalez-Molina J, et al. Extracellular fluid viscosity enhances liver cancer cell mechanosensing and migration. Biomaterials. 2018;177:113–124. doi: 10.1016/j.biomaterials.2018.05.058. [DOI] [PubMed] [Google Scholar]
- 166.Kai F, et al. ECM dimensionality tunes actin tension to modulate endoplasmic reticulum function and spheroid phenotypes of mammary epithelial cells. EMBO J. 2022;41:e109205. doi: 10.15252/embj.2021109205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Delcommenne M, Streuli CH. Control of integrin expression by extracellular matrix. J. Biol. Chem. 1995;270:26794–26801. doi: 10.1074/jbc.270.45.26794. [DOI] [PubMed] [Google Scholar]
- 168.Baker BM, Chen CS. Deconstructing the third dimension: how 3D culture microenvironments alter cellular cues. J. Cell Sci. 2012;125:3015–3024. doi: 10.1242/jcs.079509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Brierley SM, et al. TRPA1 contributes to specific mechanically activated currents and sensory neuron mechanical hypersensitivity. J. Physiol. 2011;589:3575–3593. doi: 10.1113/jphysiol.2011.206789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Zhao H, Sokabe M. Tuning the mechanosensitivity of a BK channel by changing the linker length. Cell Res. 2008;18:871–878. doi: 10.1038/cr.2008.88. [DOI] [PubMed] [Google Scholar]
- 171.Beyder A, et al. Mechanosensitivity of Nav1.5, a voltage-sensitive sodium channel. J. Physiol. 2010;588:4969–4985. doi: 10.1113/jphysiol.2010.199034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Kraichely RE, Strege PR, Sarr MG, Kendrick ML, Farrugia G. Lysophosphatidyl choline modulates mechanosensitive L-type Ca2+ current in circular smooth muscle cells from human jejunum. Am. J. Physiol. Gastrointest. Liver Physiol. 2009;296:G833–G839. doi: 10.1152/ajpgi.90610.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Calabrese B, Tabarean IV, Juranka P, Morris CE. Mechanosensitivity of N-type calcium channel currents. Biophys. J. 2002;83:2560–2574. doi: 10.1016/S0006-3495(02)75267-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Hilaire C, Lucas O, Valmier J, Scamps F. Neurotrophin-4 modulates the mechanotransducer Cav3.2 T-type calcium current in mice down-hair neurons. Biochem. J. 2012;441:463–471. doi: 10.1042/BJ20111147. [DOI] [PubMed] [Google Scholar]
- 175.Zhang WK, et al. Mechanosensitive gating of CFTR. Nat. Cell Biol. 2010;12:507–512. doi: 10.1038/ncb2053. [DOI] [PubMed] [Google Scholar]
- 176.Hong K, Driscoll M. A transmembrane domain of the putative channel subunit MEC-4 influences mechanotransduction and neurodegeneration in C. elegans. Nature. 1994;367:470–473. doi: 10.1038/367470a0. [DOI] [PubMed] [Google Scholar]
- 177.Arnadóttir J, O’Hagan R, Chen Y, Goodman MB, Chalfie M. The DEG/ENaC protein MEC-10 regulates the transduction channel complex in Caenorhabditis elegans touch receptor neurons. J. Neurosci. 2011;31:12695–12704. doi: 10.1523/JNEUROSCI.4580-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Pan B, et al. TMC1 and TMC2 are components of the mechanotransduction channel in hair cells of the mammalian inner ear. Neuron. 2013;79:504–515. doi: 10.1016/j.neuron.2013.06.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Murthy SE. Glycine-bound NMDA receptors are stretch-activated. Trends Neurosci. 2022;45:794–795. doi: 10.1016/j.tins.2022.08.002. [DOI] [PubMed] [Google Scholar]
- 180.Jiang S, et al. Down-regulation of NR2B receptors contributes to the analgesic and antianxiety effects of enriched environment mediated by endocannabinoid system in the inflammatory pain mice. Behav. Brain Res. 2022;435:114062. doi: 10.1016/j.bbr.2022.114062. [DOI] [PubMed] [Google Scholar]
- 181.Fran‡a-Neto Ad, Couto GK. Xavier, F. E. & Rossoni, L. V. Cyclooxygenase-2 is a critical determinant of angiotensin II-induced vascular remodeling and stiffness in resistance arteries of ouabain-treated rats. J. Hypertens. 2022;40:2180–2191. doi: 10.1097/HJH.0000000000003242. [DOI] [PubMed] [Google Scholar]
- 182.Discher DE, et al. Matrix mechanosensing: from scaling concepts in ‘omics data to mechanisms in the nucleus, regeneration, and cancer. Annu. Rev. Biophys. 2017;46:295–315. doi: 10.1146/annurev-biophys-062215-011206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Chen Y, Ju L, Rushdi M, Ge C, Zhu C. Receptor-mediated cell mechanosensing. Mol. Biol. Cell. 2017;28:3134–3155. doi: 10.1091/mbc.e17-04-0228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Zhong H-Y, et al. Mechanical stretch aggravates vascular smooth muscle cell apoptosis and vascular remodeling by downregulating EZH2. Int. J. Biochem. Cell Biol. 2022;151:106278. doi: 10.1016/j.biocel.2022.106278. [DOI] [PubMed] [Google Scholar]
- 185.Lee W, et al. The osteogenic differentiation of human dental pulp stem cells through G0/G1 arrest and the p-ERK/Runx-2 pathway by sonic vibration. Int. J. Mol. Sci. 2021;22:10167. doi: 10.3390/ijms221810167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Di Meglio I, et al. Pressure and curvature control of the cell cycle in epithelia growing under spherical confinement. Cell Rep. 2022;40:111227. doi: 10.1016/j.celrep.2022.111227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Freeberg MAT, et al. Mechanical feed-forward loops contribute to idiopathic pulmonary fibrosis. Am. J. Pathol. 2021;191:18–25. doi: 10.1016/j.ajpath.2020.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Coste B, et al. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science. 2010;330:55–60. doi: 10.1126/science.1193270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Coste B, et al. Piezo proteins are pore-forming subunits of mechanically activated channels. Nature. 2012;483:176–181. doi: 10.1038/nature10812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Beech DJ, Kalli AC. Force sensing by piezo channels in cardiovascular health and disease. Arterioscler. Thromb. Vasc. Biol. 2019;39:2228–2239. doi: 10.1161/ATVBAHA.119.313348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Wu J, Lewis AH, Grandl J. Touch, tension, and transduction - the function and regulation of piezo ion channels. Trends Biochem. Sci. 2017;42:57–71. doi: 10.1016/j.tibs.2016.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Xu X, et al. Piezo channels: awesome mechanosensitive structures in cellular mechanotransduction and their role in bone. Int. J. Mol. Sci. 2021;22:6429. doi: 10.3390/ijms22126429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Bae C, Sachs F, Gottlieb PA. Protonation of the human PIEZO1 ion channel stabilizes inactivation. J. Biol. Chem. 2015;290:5167–5173. doi: 10.1074/jbc.M114.604033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Syeda R, et al. Piezo1 channels are inherently mechanosensitive. Cell Rep. 2016;17:1739–1746. doi: 10.1016/j.celrep.2016.10.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Cox CD, et al. Removal of the mechanoprotective influence of the cytoskeleton reveals PIEZO1 is gated by bilayer tension. Nat. Commun. 2016;7:10366. doi: 10.1038/ncomms10366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Fujii S, et al. The TRPV4-AKT axis promotes oral squamous cell carcinoma cell proliferation via CaMKII activation. Lab. Invest. 2020;100:311–323. doi: 10.1038/s41374-019-0357-z. [DOI] [PubMed] [Google Scholar]
- 197.Guo J, et al. Trends in Piezo channel research over the past decade: a bibliometric analysis. Front. Pharm. 2021;12:668714. doi: 10.3389/fphar.2021.668714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.John L, et al. The Piezo1 cation channel mediates uterine artery shear stress mechanotransduction and vasodilation during rat pregnancy. Am. J. Physiol. Heart Circ. Physiol. 2018;315:H1019–H1026. doi: 10.1152/ajpheart.00103.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Wang S, et al. Endothelial cation channel PIEZO1 controls blood pressure by mediating flow-induced ATP release. J. Clin. Investig. 2016;126:4527–4536. doi: 10.1172/JCI87343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Lhomme A, et al. Stretch-activated Piezo1 channel in endothelial cells relaxes mouse intrapulmonary arteries. Am. J. Respir. Cell Mol. Biol. 2019;60:650–658. doi: 10.1165/rcmb.2018-0197OC. [DOI] [PubMed] [Google Scholar]
- 201.Cahalan SM, et al. Piezo1 links mechanical forces to red blood cell volume. Elife. 2015;4:e07370. doi: 10.7554/eLife.07370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Cinar E, et al. Piezo1 regulates mechanotransductive release of ATP from human RBCs. Proc. Natl Acad. Sci. USA. 2015;112:11783–11788. doi: 10.1073/pnas.1507309112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Pathak MM, et al. Stretch-activated ion channel Piezo1 directs lineage choice in human neural stem cells. Proc. Natl Acad. Sci. USA. 2014;111:16148–16153. doi: 10.1073/pnas.1409802111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Blumenthal NR, Hermanson O, Heimrich B, Shastri VP. Stochastic nanoroughness modulates neuron-astrocyte interactions and function via mechanosensing cation channels. Proc. Natl Acad. Sci. USA. 2014;111:16124–16129. doi: 10.1073/pnas.1412740111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Lang K, Breer H, Frick C. Mechanosensitive ion channel Piezo1 is expressed in antral G cells of murine stomach. Cell Tissue Res. 2018;371:251–260. doi: 10.1007/s00441-017-2755-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Zhong M, et al. Alveolar stretch activation of endothelial piezo1 protects adherens junctions and lung vascular barrier. Am. J. Respir. Cell Mol. Biol. 2020;62:168–177. doi: 10.1165/rcmb.2019-0024OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Friedrich EE, et al. Endothelial cell Piezo1 mediates pressure-induced lung vascular hyperpermeability via disruption of adherens junctions. Proc. Natl Acad. Sci. USA. 2019;116:12980–12985. doi: 10.1073/pnas.1902165116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Liang G-P, et al. Piezo1 induced apoptosis of type II pneumocytes during ARDS. Respir. Res. 2019;20:118. doi: 10.1186/s12931-019-1083-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Miyamoto T, et al. Functional role for Piezo1 in stretch-evoked Ca²+ influx and ATP release in urothelial cell cultures. J. Biol. Chem. 2014;289:16565–16575. doi: 10.1074/jbc.M113.528638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Hasegawa K, et al. YAP signaling induces PIEZO1 to promote oral squamous cell carcinoma cell proliferation. J. Pathol. 2021;253:80–93. doi: 10.1002/path.5553. [DOI] [PubMed] [Google Scholar]
- 211.Han Y, et al. Mechanosensitive ion channel Piezo1 promotes prostate cancer development through the activation of the Akt/mTOR pathway and acceleration of cell cycle. Int. J. Oncol. 2019;55:629–644. doi: 10.3892/ijo.2019.4839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Wang F, et al. Mechanosensitive ion channel Piezo2 is important for enterochromaffin cell response to mechanical forces. J. Physiol. 2017;595:79–91. doi: 10.1113/JP272718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Sun Y, et al. The function of Piezo1 in colon cancer metastasis and its potential regulatory mechanism. J. Cancer Res. Clin. Oncol. 2020;146:1139–1152. doi: 10.1007/s00432-020-03179-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Nonomura K, et al. Piezo2 senses airway stretch and mediates lung inflation-induced apnoea. Nature. 2017;541:176–181. doi: 10.1038/nature20793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Chesler AT, et al. The role of PIEZO2 in human mechanosensation. N. Engl. J. Med. 2016;375:1355–1364. doi: 10.1056/NEJMoa1602812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Wehrwein EA, Joyner MJ. Regulation of blood pressure by the arterial baroreflex and autonomic nervous system. Handb. Clin. Neurol. 2013;117:89–102. doi: 10.1016/B978-0-444-53491-0.00008-0. [DOI] [PubMed] [Google Scholar]
- 217.Kirchheim HR. Systemic arterial baroreceptor reflexes. Physiol. Rev. 1976;56:100–177. doi: 10.1152/physrev.1976.56.1.100. [DOI] [PubMed] [Google Scholar]
- 218.Zeng W-Z, et al. PIEZOs mediate neuronal sensing of blood pressure and the baroreceptor reflex. Science. 2018;362:464–467. doi: 10.1126/science.aau6324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Fitzgerald JB, et al. Mechanical compression of cartilage explants induces multiple time-dependent gene expression patterns and involves intracellular calcium and cyclic AMP. J. Biol. Chem. 2004;279:19502–19511. doi: 10.1074/jbc.M400437200. [DOI] [PubMed] [Google Scholar]
- 220.Lee W, et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc. Natl Acad. Sci. USA. 2014;111:E5114–E5122. doi: 10.1073/pnas.1414298111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Marshall KL, et al. PIEZO2 in sensory neurons and urothelial cells coordinates urination. Nature. 2020;588:290–295. doi: 10.1038/s41586-020-2830-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Retailleau K, et al. Piezo1 in smooth muscle cells is involved in hypertension-dependent arterial remodeling. Cell Rep. 2015;13:1161–1171. doi: 10.1016/j.celrep.2015.09.072. [DOI] [PubMed] [Google Scholar]
- 223.Davis MJ, Donovitz JA, Hood JD. Stretch-activated single-channel and whole cell currents in vascular smooth muscle cells. Am. J. Physiol. 1992;262:C1083–C1088. doi: 10.1152/ajpcell.1992.262.4.C1083. [DOI] [PubMed] [Google Scholar]
- 224.McCarter GC, Reichling DB, Levine JD. Mechanical transduction by rat dorsal root ganglion neurons in vitro. Neurosci. Lett. 1999;273:179–182. doi: 10.1016/S0304-3940(99)00665-5. [DOI] [PubMed] [Google Scholar]
- 225.Dalghi MG, et al. Expression and distribution of PIEZO1 in the mouse urinary tract. Am. J. Physiol. Ren. Physiol. 2019;317:F303–F321. doi: 10.1152/ajprenal.00214.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Gudipaty SA, et al. Mechanical stretch triggers rapid epithelial cell division through Piezo1. Nature. 2017;543:118–121. doi: 10.1038/nature21407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Bagriantsev SN, Gracheva EO, Gallagher PG. Piezo proteins: regulators of mechanosensation and other cellular processes. J. Biol. Chem. 2014;289:31673–31681. doi: 10.1074/jbc.R114.612697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Solis AG, et al. Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity. Nature. 2019;573:69–74. doi: 10.1038/s41586-019-1485-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Murthy SE, Dubin AE, Patapoutian A. Piezos thrive under pressure: mechanically activated ion channels in health and disease. Nat. Rev. Mol. Cell Biol. 2017;18:771–783. doi: 10.1038/nrm.2017.92. [DOI] [PubMed] [Google Scholar]
- 230.Jiang Y, Yang X, Jiang J, Xiao B. Structural designs and mechanogating mechanisms of the mechanosensitive piezo channels. Trends Biochem. Sci. 2021;46:472–488. doi: 10.1016/j.tibs.2021.01.008. [DOI] [PubMed] [Google Scholar]
- 231.Hyman AJ, Tumova S, Beech DJ. Piezo1 channels in vascular development and the sensing of shear stress. Curr. Top. Membr. 2017;79:37–57. doi: 10.1016/bs.ctm.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 232.Li J, et al. Piezo1 integration of vascular architecture with physiological force. Nature. 2014;515:279–282. doi: 10.1038/nature13701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Xu H, et al. Mechanical force modulates macrophage proliferation via Piezo1-AKT-Cyclin D1 axis. FASEB J. 2022;36:e22423. doi: 10.1096/fj.202200314R. [DOI] [PubMed] [Google Scholar]
- 234.Qiu Z, et al. The mechanosensitive ion channel Piezo1 significantly mediates in vitro ultrasonic stimulation of neurons. iScience. 2019;21:448–457. doi: 10.1016/j.isci.2019.10.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Gaub BM, Müller DJ. Mechanical stimulation of Piezo1 receptors depends on extracellular matrix proteins and directionality of force. Nano Lett. 2017;17:2064–2072. doi: 10.1021/acs.nanolett.7b00177. [DOI] [PubMed] [Google Scholar]
- 236.Douguet D, Patel A, Xu A, Vanhoutte PM, Honoré E. Piezo ion channels in cardiovascular mechanobiology. Trends Pharmacol. Sci. 2019;40:956–970. doi: 10.1016/j.tips.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 237.Iring A, et al. Shear stress-induced endothelial adrenomedullin signaling regulates vascular tone and blood pressure. J. Clin. Investig. 2019;129:2775–2791. doi: 10.1172/JCI123825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Rode B, et al. Piezo1 channels sense whole body physical activity to reset cardiovascular homeostasis and enhance performance. Nat. Commun. 2017;8:350. doi: 10.1038/s41467-017-00429-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Tyler WJ. The mechanobiology of brain function. Nat. Rev. Neurosci. 2012;13:867–878. doi: 10.1038/nrn3383. [DOI] [PubMed] [Google Scholar]
- 240.Pfister BJ, Iwata A, Meaney DF, Smith DH. Extreme stretch growth of integrated axons. J. Neurosci. 2004;24:7978–7983. doi: 10.1523/JNEUROSCI.1974-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Delmas P, Hao J, Rodat-Despoix L. Molecular mechanisms of mechanotransduction in mammalian sensory neurons. Nat. Rev. Neurosci. 2011;12:139–153. doi: 10.1038/nrn2993. [DOI] [PubMed] [Google Scholar]
- 242.Murthy SE, et al. The mechanosensitive ion channel Piezo2 mediates sensitivity to mechanical pain in mice. Sci. Transl. Med. 2018;10:eaat9897. doi: 10.1126/scitranslmed.aat9897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Szczot M, et al. PIEZO2 mediates injury-induced tactile pain in mice and humans. Sci. Transl. Med. 2018;10:eaat9892. doi: 10.1126/scitranslmed.aat9892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Arcourt A, et al. Touch receptor-derived sensory information alleviates acute pain signaling and fine-tunes nociceptive reflex coordination. Neuron. 2017;93:179–193. doi: 10.1016/j.neuron.2016.11.027. [DOI] [PubMed] [Google Scholar]
- 245.Mikhailov N, et al. Mechanosensitive meningeal nociception via Piezo channels: implications for pulsatile pain in migraine? Neuropharmacology. 2019;149:113–123. doi: 10.1016/j.neuropharm.2019.02.015. [DOI] [PubMed] [Google Scholar]
- 246.Tektas O-Y, Lütjen-Drecoll E. Structural changes of the trabecular meshwork in different kinds of glaucoma. Exp. Eye Res. 2009;88:769–775. doi: 10.1016/j.exer.2008.11.025. [DOI] [PubMed] [Google Scholar]
- 247.Morozumi W, et al. Piezo 1 is involved in intraocular pressure regulation. J. Pharm. Sci. 2021;147:211–221. doi: 10.1016/j.jphs.2021.06.005. [DOI] [PubMed] [Google Scholar]
- 248.Wang Y, et al. A lever-like transduction pathway for long-distance chemical- and mechano-gating of the mechanosensitive Piezo1 channel. Nat. Commun. 2018;9:1300. doi: 10.1038/s41467-018-03570-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Botello-Smith WM, et al. A mechanism for the activation of the mechanosensitive Piezo1 channel by the small molecule Yoda1. Nat. Commun. 2019;10:4503. doi: 10.1038/s41467-019-12501-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Michishita M, Yano K, Tomita K-I, Matsuzaki O, Kasahara K-I. Piezo1 expression increases in rat bladder after partial bladder outlet obstruction. Life Sci. 2016;166:1–7. doi: 10.1016/j.lfs.2016.10.017. [DOI] [PubMed] [Google Scholar]
- 251.Humphries JD, Byron A, Humphries MJ. Integrin ligands at a glance. J. Cell Sci. 2006;119:3901–3903. doi: 10.1242/jcs.03098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Meagher PB, et al. Cardiac fibrosis: key role of integrins in cardiac homeostasis and remodeling. Cells. 2021;10:770. doi: 10.3390/cells10040770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Chen C, Li R, Ross RS, Manso AM. Integrins and integrin-related proteins in cardiac fibrosis. J. Mol. Cell Cardiol. 2016;93:162–174. doi: 10.1016/j.yjmcc.2015.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Nolte M, Margadant C. Controlling immunity and inflammation through integrin-dependent regulation of TGF-β. Trends Cell Biol. 2020;30:49–59. doi: 10.1016/j.tcb.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 255.Koivisto L, Bi J, Häkkinen L, Larjava H. Integrin αvβ6: structure, function and role in health and disease. Int. J. Biochem. Cell Biol. 2018;99:186–196. doi: 10.1016/j.biocel.2018.04.013. [DOI] [PubMed] [Google Scholar]
- 256.Simpson DG, et al. Modulation of cardiac myocyte phenotype in vitro by the composition and orientation of the extracellular matrix. J. Cell Physiol. 1994;161:89–105. doi: 10.1002/jcp.1041610112. [DOI] [PubMed] [Google Scholar]
- 257.Mittal A, Pulina M, Hou S-Y, Astrof S. Fibronectin and integrin alpha 5 play requisite roles in cardiac morphogenesis. Dev. Biol. 2013;381:73–82. doi: 10.1016/j.ydbio.2013.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Israeli-Rosenberg S, Manso AM, Okada H, Ross RS. Integrins and integrin-associated proteins in the cardiac myocyte. Circ. Res. 2014;114:572–586. doi: 10.1161/CIRCRESAHA.114.301275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Gullberg D, Turner DC, Borg TK, Terracio L, Rubin K. Different beta 1-integrin collagen receptors on rat hepatocytes and cardiac fibroblasts. Exp. Cell Res. 1990;190:254–264. doi: 10.1016/0014-4827(90)90194-F. [DOI] [PubMed] [Google Scholar]
- 260.Gullberg D, et al. Analysis of alpha 1 beta 1, alpha 2 beta 1 and alpha 3 beta 1 integrins in cell–collagen interactions: identification of conformation dependent alpha 1 beta 1 binding sites in collagen type I. EMBO J. 1992;11:3865–3873. doi: 10.1002/j.1460-2075.1992.tb05479.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Civitarese RA, et al. The α11 integrin mediates fibroblast-extracellular matrix-cardiomyocyte interactions in health and disease. Am. J. Physiol. Heart Circ. Physiol. 2016;311:H96–H106. doi: 10.1152/ajpheart.00918.2015. [DOI] [PubMed] [Google Scholar]
- 262.Balasubramanian S, et al. β3 integrin in cardiac fibroblast is critical for extracellular matrix accumulation during pressure overload hypertrophy in mouse. PLoS ONE. 2012;7:e45076. doi: 10.1371/journal.pone.0045076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Henderson NC, et al. Targeting of αv integrin identifies a core molecular pathway that regulates fibrosis in several organs. Nat. Med. 2013;19:1617–1624. doi: 10.1038/nm.3282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Yokosaki Y, Nishimichi N. New therapeutic targets for hepatic fibrosis in the integrin family, α8β1 and α11β1, induced specifically on activated stellate cells. Int. J. Mol. Sci. 2021;22:12794. doi: 10.3390/ijms222312794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Nishimichi N, et al. Induced hepatic stellate cell integrin, α8β1, enhances cellular contractility and TGFβ activity in liver fibrosis. J. Pathol. 2021;253:366–373. doi: 10.1002/path.5618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Iwamoto DV, Calderwood DA. Regulation of integrin-mediated adhesions. Curr. Opin. Cell Biol. 2015;36:41–47. doi: 10.1016/j.ceb.2015.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Sun Z, Costell M, Fässler R. Integrin activation by talin, kindlin and mechanical forces. Nat. Cell Biol. 2019;21:25–31. doi: 10.1038/s41556-018-0234-9. [DOI] [PubMed] [Google Scholar]
- 268.Sun Z, Guo SS, Fässler R. Integrin-mediated mechanotransduction. J. Cell Biol. 2016;215:445–456. doi: 10.1083/jcb.201609037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Levental KR, et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell. 2009;139:891–906. doi: 10.1016/j.cell.2009.10.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Li M, et al. Integrins as attractive targets for cancer therapeutics. Acta Pharm. Sin. B. 2021;11:2726–2737. doi: 10.1016/j.apsb.2021.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Rayego-Mateos S, et al. Interplay between extracellular matrix components and cellular and molecular mechanisms in kidney fibrosis. Clin. Sci. 2021;135:1999–2029. doi: 10.1042/CS20201016. [DOI] [PubMed] [Google Scholar]
- 272.Song H, et al. Tension enhances cell proliferation and collagen synthesis by upregulating expressions of integrin αvβ3 in human keloid-derived mesenchymal stem cells. Life Sci. 2019;219:272–282. doi: 10.1016/j.lfs.2018.12.042. [DOI] [PubMed] [Google Scholar]
- 273.Feng F, et al. Biomechanical regulatory factors and therapeutic targets in keloid fibrosis. Front. Pharm. 2022;13:906212. doi: 10.3389/fphar.2022.906212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Guilluy C, et al. The Rho GEFs LARG and GEF-H1 regulate the mechanical response to force on integrins. Nat. Cell Biol. 2011;13:722–727. doi: 10.1038/ncb2254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Codelia VA, Sun G, Irvine KD. Regulation of YAP by mechanical strain through Jnk and Hippo signaling. Curr. Biol. 2014;24:2012–2017. doi: 10.1016/j.cub.2014.07.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Mohseni M, et al. A genetic screen identifies an LKB1-MARK signalling axis controlling the Hippo-YAP pathway. Nat. Cell Biol. 2014;16:108–117. doi: 10.1038/ncb2884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Elbediwy A, et al. Integrin signalling regulates YAP and TAZ to control skin homeostasis. Development. 2016;143:1674–1687. doi: 10.1242/dev.133728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Sudol M. Yes-associated protein (YAP65) is a proline-rich phosphoprotein that binds to the SH3 domain of the Yes proto-oncogene product. Oncogene. 1994;9:2145–2152. [PubMed] [Google Scholar]
- 279.Vassilev A, Kaneko KJ, Shu H, Zhao Y, DePamphilis ML. TEAD/TEF transcription factors utilize the activation domain of YAP65, a Src/Yes-associated protein localized in the cytoplasm. Genes Dev. 2001;15:1229–1241. doi: 10.1101/gad.888601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Kaan HYK, Sim AYL, Tan SKJ, Verma C, Song H. Targeting YAP/TAZ-TEAD protein-protein interactions using fragment-based and computational modeling approaches. PLoS ONE. 2017;12:e0178381. doi: 10.1371/journal.pone.0178381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Koo JH, Guan K-L. Interplay between YAP/TAZ and metabolism. Cell Metab. 2018;28:196–206. doi: 10.1016/j.cmet.2018.07.010. [DOI] [PubMed] [Google Scholar]
- 282.Noguchi S, Saito A, Nagase T. YAP/TAZ signaling as a molecular link between fibrosis and cancer. Int. J. Mol. Sci. 2018;19:3674. doi: 10.3390/ijms19113674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Moya IM, Halder G. Hippo-YAP/TAZ signalling in organ regeneration and regenerative medicine. Nat. Rev. Mol. Cell Biol. 2019;20:211–226. doi: 10.1038/s41580-018-0086-y. [DOI] [PubMed] [Google Scholar]
- 284.Nguyen CDK, Yi C. YAP/TAZ signaling and resistance to cancer therapy. Trends Cancer. 2019;5:283–296. doi: 10.1016/j.trecan.2019.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Guo Y, et al. CK2-induced cooperation of HHEX with the YAP-TEAD4 complex promotes colorectal tumorigenesis. Nat. Commun. 2022;13:4995. doi: 10.1038/s41467-022-32674-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Yan H, et al. Yap regulates gastric cancer survival and migration via SIRT1/Mfn2/mitophagy. Oncol. Rep. 2018;39:1671–1681. doi: 10.3892/or.2018.6252. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 287.Booth AJ, et al. Acellular normal and fibrotic human lung matrices as a culture system for in vitro investigation. Am. J. Respir. Crit. Care Med. 2012;186:866–876. doi: 10.1164/rccm.201204-0754OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Li Y, et al. Severe lung fibrosis requires an invasive fibroblast phenotype regulated by hyaluronan and CD44. J. Exp. Med. 2011;208:1459–1471. doi: 10.1084/jem.20102510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Georges PC, et al. Increased stiffness of the rat liver precedes matrix deposition: implications for fibrosis. Am. J. Physiol. Gastrointest. Liver Physiol. 2007;293:G1147–G1154. doi: 10.1152/ajpgi.00032.2007. [DOI] [PubMed] [Google Scholar]
- 290.Ma S, Meng Z, Chen R, Guan K-L. The Hippo pathway: biology and pathophysiology. Annu. Rev. Biochem. 2019;88:577–604. doi: 10.1146/annurev-biochem-013118-111829. [DOI] [PubMed] [Google Scholar]
- 291.Byun J, et al. Yes-associated protein (YAP) mediates adaptive cardiac hypertrophy in response to pressure overload. J. Biol. Chem. 2019;294:3603–3617. doi: 10.1074/jbc.RA118.006123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Deng F, et al. YAP triggers the Wnt/β-catenin signalling pathway and promotes enterocyte self-renewal, regeneration and tumorigenesis after DSS-induced injury. Cell Death Dis. 2018;9:153. doi: 10.1038/s41419-017-0244-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Wang S, et al. Yes-associated protein (YAP) expression is involved in epithelial-mesenchymal transition in hepatocellular carcinoma. Clin. Transl. Oncol. 2016;18:172–177. doi: 10.1007/s12094-015-1353-4. [DOI] [PubMed] [Google Scholar]
- 294.Boopathy GTK, Hong W. Role of Hippo Ppathway-YAP/TAZ signaling in angiogenesis. Front. Cell Dev. Biol. 2019;7:49. doi: 10.3389/fcell.2019.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Zhang X, et al. Yes-associated protein (YAP) binds to HIF-1α and sustains HIF-1α protein stability to promote hepatocellular carcinoma cell glycolysis under hypoxic stress. J. Exp. Clin. Cancer Res. 2018;37:216. doi: 10.1186/s13046-018-0892-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Ibar C, Irvine KD. Integration of Hippo-YAP signaling with metabolism. Dev. Cell. 2020;54:256–267. doi: 10.1016/j.devcel.2020.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Zhang Q, et al. Yes-associated protein (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ) mediate cell density-dependent proinflammatory responses. J. Biol. Chem. 2018;293:18071–18085. doi: 10.1074/jbc.RA118.004251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Brusatin G, Panciera T, Gandin A, Citron A, Piccolo S. Biomaterials and engineered microenvironments to control YAP/TAZ-dependent cell behaviour. Nat. Mater. 2018;17:1063–1075. doi: 10.1038/s41563-018-0180-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Ohgushi M, Minaguchi M, Sasai Y. Rho-signaling-directed YAP/TAZ activity underlies the long-term survival and expansion of human embryonic stem cells. Cell Stem Cell. 2015;17:448–461. doi: 10.1016/j.stem.2015.07.009. [DOI] [PubMed] [Google Scholar]
- 300.Beyer TA, et al. Switch enhancers interpret TGF-β and Hippo signaling to control cell fate in human embryonic stem cells. Cell Rep. 2013;5:1611–1624. doi: 10.1016/j.celrep.2013.11.021. [DOI] [PubMed] [Google Scholar]
- 301.Tang Y, et al. MT1-MMP-dependent control of skeletal stem cell commitment via a β1-integrin/YAP/TAZ signaling axis. Dev. Cell. 2013;25:402–416. doi: 10.1016/j.devcel.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Feng X, et al. Thromboxane A2 activates YAP/TAZ protein to induce vascular smooth muscle cell proliferation and migration. J. Biol. Chem. 2016;291:18947–18958. doi: 10.1074/jbc.M116.739722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Lin M, et al. Yes-associated protein mediates angiotensin II-induced vascular smooth muscle cell phenotypic modulation and hypertensive vascular remodelling. Cell Prolif. 2018;51:e12517. doi: 10.1111/cpr.12517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Totaro A, et al. YAP/TAZ link cell mechanics to Notch signalling to control epidermal stem cell fate. Nat. Commun. 2017;8:15206. doi: 10.1038/ncomms15206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Morikawa Y, et al. Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice. Sci. Signal. 2015;8:ra41. doi: 10.1126/scisignal.2005781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Bertero T, et al. Matrix remodeling promotes pulmonary hypertension through feedback mechanoactivation of the YAP/TAZ-miR-130/301 circuit. Cell Rep. 2015;13:1016–1032. doi: 10.1016/j.celrep.2015.09.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Noguchi S, et al. TAZ contributes to pulmonary fibrosis by activating profibrotic functions of lung fibroblasts. Sci. Rep. 2017;7:42595. doi: 10.1038/srep42595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Mooring M, et al. Hepatocyte stress increases expression of Yes-associated protein and transcriptional coactivator with PDZ-binding motif in hepatocytes to promote parenchymal inflammation and fibrosis. Hepatology. 2020;71:1813–1830. doi: 10.1002/hep.30928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Szeto SG, et al. YAP/TAZ are mechanoregulators of TGF–Smad signaling and renal fibrogenesis. J. Am. Soc. Nephrol. 2016;27:3117–3128. doi: 10.1681/ASN.2015050499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Zhang T, et al. NUAK1 promotes organ fibrosis via YAP and TGF-β/SMAD signaling. Sci. Transl. Med. 2022;14:eaaz4028. doi: 10.1126/scitranslmed.aaz4028. [DOI] [PubMed] [Google Scholar]
- 311.Li S, et al. Targeting mechanics-induced fibroblast activation through CD44-RhoA-YAP pathway ameliorates crystalline silica-induced silicosis. Theranostics. 2019;9:4993–5008. doi: 10.7150/thno.35665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Panciera T, Azzolin L, Cordenonsi M, Piccolo S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 2017;18:758–770. doi: 10.1038/nrm.2017.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Di X-P, et al. YAP/Smad3 promotes pathological extracellular matrix microenviroment-induced bladder smooth muscle proliferation in bladder fibrosis progression. MedComm. 2022;3:e169. doi: 10.1002/mco2.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Mascharak S, et al. Preventing activation in fibroblasts yields wound regeneration without scarring. Science. 2021;372:eaba2374. doi: 10.1126/science.aba2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Martin K, et al. PAK proteins and YAP-1 signalling downstream of integrin beta-1 in myofibroblasts promote liver fibrosis. Nat. Commun. 2016;7:12502. doi: 10.1038/ncomms12502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Sharma S, Goswami R, Zhang DX, Rahaman SO. TRPV4 regulates matrix stiffness and TGFβ1-induced epithelial-mesenchymal transition. J. Cell Mol. Med. 2019;23:761–774. doi: 10.1111/jcmm.13972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Lee WH, et al. TRPV4 plays a role in breast cancer cell migration via Ca2+-dependent activation of AKT and downregulation of E-cadherin cell cortex protein. Oncogenesis. 2017;6:e338. doi: 10.1038/oncsis.2017.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Zhao B, et al. TMIE is an essential component of the mechanotransduction machinery of cochlear hair cells. Neuron. 2014;84:954–967. doi: 10.1016/j.neuron.2014.10.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Cunningham CL, et al. TMIE defines pore and gating properties of the mechanotransduction channel of mammalian cochlear hair cells. Neuron. 2020;107:126.e8–143.e8. doi: 10.1016/j.neuron.2020.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Jeong H, et al. Structures of the TMC-1 complex illuminate mechanosensory transduction. Nature. 2022;610:796–803. doi: 10.1038/s41586-022-05314-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Fettiplace R, Furness DN, Beurg M. The conductance and organization of the TMC1-containing mechanotransducer channel complex in auditory hair cells. Proc. Natl Acad. Sci. USA. 2022;119:e2210849119. doi: 10.1073/pnas.2210849119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Li S, Li B, Gao L, Wang J, Yan Z. Humidity response in Drosophila olfactory sensory neurons requires the mechanosensitive channel TMEM63. Nat. Commun. 2022;13:3814. doi: 10.1038/s41467-022-31253-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Zhang M, et al. Structure of the mechanosensitive OSCA channels. Nat. Struct. Mol. Biol. 2018;25:850–858. doi: 10.1038/s41594-018-0117-6. [DOI] [PubMed] [Google Scholar]
- 324.Murthy SE, et al. OSCA/TMEM63 are an evolutionarily conserved family of mechanically activated ion channels. Elife. 2018;7:e41844. doi: 10.7554/eLife.41844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Rockey DC, Bell PD, Hill JA. Fibrosis–a common pathway to organ injury and failure. N. Engl. J. Med. 2015;372:1138–1149. doi: 10.1056/NEJMra1300575. [DOI] [PubMed] [Google Scholar]
- 326.Hodge RG, Ridley AJ. Regulating Rho GTPases and their regulators. Nat. Rev. Mol. Cell Biol. 2016;17:496–510. doi: 10.1038/nrm.2016.67. [DOI] [PubMed] [Google Scholar]
- 327.Huang W, et al. Increased intracellular Cl concentration improves airway epithelial migration by activating the RhoA/ROCK pathway. Theranostics. 2020;10:8528–8540. doi: 10.7150/thno.46002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Matsui T, et al. Rho-associated kinase, a novel serine/threonine kinase, as a putative target for small GTP binding protein Rho. EMBO J. 1996;15:2208–2216. doi: 10.1002/j.1460-2075.1996.tb00574.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Amano M, Nakayama M, Kaibuchi K. Rho-kinase/ROCK: a key regulator of the cytoskeleton and cell polarity. Cytoskeleton. 2010;67:545–554. doi: 10.1002/cm.20472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Mulherkar S, Tolias KF. RhoA-ROCK signaling as a therapeutic target in traumatic brain injury. Cells. 2020;9:245. doi: 10.3390/cells9010245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Deng Z, et al. RhoA/ROCK pathway: implication in osteoarthritis and therapeutic targets. Am. J. Transl. Res. 2019;11:5324–5331. [PMC free article] [PubMed] [Google Scholar]
- 332.Cai R, et al. Role of RhoA/ROCK signaling in Alzheimer’s disease. Behav. Brain Res. 2021;414:113481. doi: 10.1016/j.bbr.2021.113481. [DOI] [PubMed] [Google Scholar]
- 333.Lu W, Chen Z, Wen J. RhoA/ROCK signaling pathway and astrocytes in ischemic stroke. Metab. Brain Dis. 2021;36:1101–1108. doi: 10.1007/s11011-021-00709-4. [DOI] [PubMed] [Google Scholar]
- 334.Xie Y, et al. Fasudil alleviates hepatic fibrosis in type 1 diabetic rats: involvement of the inflammation and RhoA/ROCK pathway. Eur. Rev. Med. Pharm. Sci. 2018;22:5665–5677. doi: 10.26355/eurrev_201809_15834. [DOI] [PubMed] [Google Scholar]
- 335.Yan J, et al. ROCK inhibitor attenuates carbon blacks-induced pulmonary fibrosis in mice via Rho/ROCK/NF-kappa B pathway. Environ. Toxicol. 2021;36:1476–1484. doi: 10.1002/tox.23135. [DOI] [PubMed] [Google Scholar]
- 336.Wei X, et al. TAGLN mediated stiffness-regulated ovarian cancer progression via RhoA/ROCK pathway. J. Exp. Clin. Cancer Res. 2021;40:292. doi: 10.1186/s13046-021-02091-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Shi J, et al. Notch3 modulates cardiac fibroblast proliferation, apoptosis, and fibroblast to myofibroblast transition via negative regulation of the RhoA/ROCK/Hif1α axis. Front. Physiol. 2020;11:669. doi: 10.3389/fphys.2020.00669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 338.Zhou H, et al. Involvement of RhoA/ROCK in myocardial fibrosis in a rat model of type 2 diabetes. Acta Pharm. Sin. 2011;32:999–1008. doi: 10.1038/aps.2011.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Korol A, Taiyab A, West-Mays JA. RhoA/ROCK signaling regulates TGFβ-induced epithelial-mesenchymal transition of lens epithelial cells through MRTF-A. Mol. Med. 2016;22:713–723. doi: 10.2119/molmed.2016.00041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Yu L, et al. Self-strengthening adhesive force promotes cell mechanotransduction. Adv. Mater. 2020;32:e2006986. doi: 10.1002/adma.202006986. [DOI] [PubMed] [Google Scholar]
- 341.Peng Y, et al. ROCK isoforms differentially modulate cancer cell motility by mechanosensing the substrate stiffness. Acta Biomater. 2019;88:86–101. doi: 10.1016/j.actbio.2019.02.015. [DOI] [PubMed] [Google Scholar]
- 342.Blomme B, et al. Mechanical strain induces a pro-fibrotic phenotype in human mitral valvular interstitial cells through RhoC/ROCK/MRTF-A and Erk1/2 signaling pathways. J. Mol. Cell Cardiol. 2019;135:149–159. doi: 10.1016/j.yjmcc.2019.08.008. [DOI] [PubMed] [Google Scholar]
- 343.Meng X-M, Nikolic-Paterson DJ, Lan HY. TGF-β: the master regulator of fibrosis. Nat. Rev. Nephrol. 2016;12:325–338. doi: 10.1038/nrneph.2016.48. [DOI] [PubMed] [Google Scholar]
- 344.Bossé Y, Rola-Pleszczynski M. Controversy surrounding the increased expression of TGF beta 1 in asthma. Respir. Res. 2007;8:66. doi: 10.1186/1465-9921-8-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Torrego A, Hew M, Oates T, Sukkar M, Fan Chung K. Expression and activation of TGF-beta isoforms in acute allergen-induced remodelling in asthma. Thorax. 2007;62:307–313. doi: 10.1136/thx.2006.063487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Chen M, Zhang W, Shi J, Jiang S. TGF-β1-induced airway smooth muscle cell proliferation involves TRPM7-dependent calcium influx via TGFβR/SMAD3. Mol. Immunol. 2018;103:173–181. doi: 10.1016/j.molimm.2018.09.015. [DOI] [PubMed] [Google Scholar]
- 347.Derynck R, Budi EH. Specificity, versatility, and control of TGF-β family signaling. Sci. Signal. 2019;12:eaav5183. doi: 10.1126/scisignal.aav5183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 348.Wrighton KH, Feng X-H. To (TGF)beta or not to (TGF)beta: fine-tuning of Smad signaling via post-translational modifications. Cell. Signal. 2008;20:1579–1591. doi: 10.1016/j.cellsig.2008.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 349.Feng X-H, Derynck R. Specificity and versatility in tgf-beta signaling through Smads. Annu. Rev. Cell Dev. Biol. 2005;21:659–693. doi: 10.1146/annurev.cellbio.21.022404.142018. [DOI] [PubMed] [Google Scholar]
- 350.Inman GJ, Hill CS. Stoichiometry of active smad-transcription factor complexes on DNA. J. Biol. Chem. 2002;277:51008–51016. doi: 10.1074/jbc.M208532200. [DOI] [PubMed] [Google Scholar]
- 351.Ross S, Hill CS. How the Smads regulate transcription. Int. J. Biochem. Cell Biol. 2008;40:383–408. doi: 10.1016/j.biocel.2007.09.006. [DOI] [PubMed] [Google Scholar]
- 352.Xu P, Lin X, Feng X-H. Posttranslational regulation of Smads. Cold Spring Harb. Perspect. Biol. 2016;8:a022087. doi: 10.1101/cshperspect.a022087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Bruce DL, Sapkota GP. Phosphatases in SMAD regulation. FEBS Lett. 2012;586:1897–1905. doi: 10.1016/j.febslet.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 354.Fennen M, Pap T, Dankbar B. Smad-dependent mechanisms of inflammatory bone destruction. Arthritis Res. Ther. 2016;18:279. doi: 10.1186/s13075-016-1187-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Meng X-M, Chung ACK, Lan HY. Role of the TGF-β/BMP-7/Smad pathways in renal diseases. Clin. Sci. 2013;124:243–254. doi: 10.1042/CS20120252. [DOI] [PubMed] [Google Scholar]
- 356.Chen L, et al. Central role of dysregulation of TGF-β/Smad in CKD progression and potential targets of its treatment. Biomed. Pharmacother. 2018;101:670–681. doi: 10.1016/j.biopha.2018.02.090. [DOI] [PubMed] [Google Scholar]
- 357.Lan HY, Chung ACK. TGF-β/Smad signaling in kidney disease. Semin. Nephrol. 2012;32:236–243. doi: 10.1016/j.semnephrol.2012.04.002. [DOI] [PubMed] [Google Scholar]
- 358.Malhotra N, Kang J. SMAD regulatory networks construct a balanced immune system. Immunology. 2013;139:1–10. doi: 10.1111/imm.12076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Zhao B, et al. Overexpression of lncRNA ANRIL promoted the proliferation and migration of prostate cancer cells via regulating let-7a/TGF-β1/ Smad signaling pathway. Cancer Biomark. 2018;21:613–620. doi: 10.3233/CBM-170683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Hiew L-F, Poon C-H, You H-Z, Lim L-W. TGF-β/Smad signalling in neurogenesis: implications for neuropsychiatric diseases. Cells. 2021;10:1382. doi: 10.3390/cells10061382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 361.Chandrasinghe P, et al. Role of SMAD proteins in colitis-associated cancer: from known to the unknown. Oncogene. 2018;37:1–7. doi: 10.1038/onc.2017.300. [DOI] [PubMed] [Google Scholar]
- 362.Wu H, et al. Progressive pulmonary fibrosis is caused by elevated mechanical tension on alveolar stem cells. Cell. 2020;180:107.e17–121.e17. doi: 10.1016/j.cell.2019.11.027. [DOI] [PubMed] [Google Scholar]
- 363.Huang YZ, et al. Interrupting TGF-β1/CCN2/integrin-α5β1 signaling alleviates high mechanical-stress caused chondrocyte fibrosis. Eur. Rev. Med. Pharm. Sci. 2021;25:1233–1241. doi: 10.26355/eurrev_202102_24827. [DOI] [PubMed] [Google Scholar]
- 364.Munger JS, Sheppard D. Cross talk among TGF-β signaling pathways, integrins, and the extracellular matrix. Cold Spring Harb. Perspect. Biol. 2011;3:a005017. doi: 10.1101/cshperspect.a005017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Wipff P-J, Rifkin DB, Meister J-J, Hinz B. Myofibroblast contraction activates latent TGF-beta1 from the extracellular matrix. J. Cell Biol. 2007;179:1311–1323. doi: 10.1083/jcb.200704042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Connolly EC, Freimuth J, Akhurst RJ. Complexities of TGF-β targeted cancer therapy. Int. J. Biol. Sci. 2012;8:964–978. doi: 10.7150/ijbs.4564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 367.Akhurst RJ, Hata A. Targeting the TGFβ signalling pathway in disease. Nat. Rev. Drug Discov. 2012;11:790–811. doi: 10.1038/nrd3810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 368.Zhou B, et al. Troglitazone attenuates TGF-β1-induced EMT in alveolar epithelial cells via a PPARγ-independent mechanism. PLoS ONE. 2012;7:e38827. doi: 10.1371/journal.pone.0038827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 369.Gomez EW, Chen QK, Gjorevski N, Nelson CM. Tissue geometry patterns epithelial-mesenchymal transition via intercellular mechanotransduction. J. Cell Biochem. 2010;110:44–51. doi: 10.1002/jcb.22545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 370.Johnson LA, et al. Novel Rho/MRTF/SRF inhibitors block matrix-stiffness and TGF-β-induced fibrogenesis in human colonic myofibroblasts. Inflamm. Bowel Dis. 2014;20:154–165. doi: 10.1097/01.MIB.0000437615.98881.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 371.Velasquez LS, et al. Activation of MRTF-A-dependent gene expression with a small molecule promotes myofibroblast differentiation and wound healing. Proc. Natl Acad. Sci. USA. 2013;110:16850–16855. doi: 10.1073/pnas.1316764110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Bolli R, Dawn B, Xuan Y-T. Role of the JAK-STAT pathway in protection against myocardial ischemia/reperfusion injury. Trends Cardiovasc. Med. 2003;13:72–79. doi: 10.1016/S1050-1738(02)00230-X. [DOI] [PubMed] [Google Scholar]
- 373.Montero P, Milara J, Roger I, Cortijo J. Role of JAK/STAT in interstitial lung diseases; molecular and cellular mechanisms. Int. J. Mol. Sci. 2021;22:6211. doi: 10.3390/ijms22126211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.Xin P, et al. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int. Immunopharmacol. 2020;80:106210. doi: 10.1016/j.intimp.2020.106210. [DOI] [PubMed] [Google Scholar]
- 375.O’Shea JJ, Plenge R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity. 2012;36:542–550. doi: 10.1016/j.immuni.2012.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.O’Shea JJ, et al. The JAK-STAT pathway: impact on human disease and therapeutic intervention. Annu. Rev. Med. 2015;66:311–328. doi: 10.1146/annurev-med-051113-024537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Hodge JA, et al. The mechanism of action of tofacitinib - an oral Janus kinase inhibitor for the treatment of rheumatoid arthritis. Clin. Exp. Rheumatol. 2016;34:318–328. [PubMed] [Google Scholar]
- 378.Benveniste EN, Liu Y, McFarland BC, Qin H. Involvement of the janus kinase/signal transducer and activator of transcription signaling pathway in multiple sclerosis and the animal model of experimental autoimmune encephalomyelitis. J. Interferon Cytokine Res. 2014;34:577–588. doi: 10.1089/jir.2014.0012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Qin H, et al. Inhibition of the JAK/STAT pathway protects against α-synuclein-induced neuroinflammation and dopaminergic neurodegeneration. J. Neurosci. 2016;36:5144–5159. doi: 10.1523/JNEUROSCI.4658-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Liu Y, et al. Therapeutic efficacy of suppressing the Jak/STAT pathway in multiple models of experimental autoimmune encephalomyelitis. J. Immunol. 2014;192:59–72. doi: 10.4049/jimmunol.1301513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Zundler S, Neurath MF. Integrating immunologic signaling networks: the JAK/STAT pathway in colitis and colitis-associated cancer. Vaccines. 2016;4:5. doi: 10.3390/vaccines4010005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 382.Ling L, et al. MicroRNA-30e promotes hepatocyte proliferation and inhibits apoptosis in cecal ligation and puncture-induced sepsis through the JAK/STAT signaling pathway by binding to FOSL2. Biomed. Pharmacother. 2018;104:411–419. doi: 10.1016/j.biopha.2018.05.042. [DOI] [PubMed] [Google Scholar]
- 383.Gao B, Wang H, Lafdil F, Feng D. STAT proteins - key regulators of anti-viral responses, inflammation, and tumorigenesis in the liver. J. Hepatol. 2012;57:430–441. doi: 10.1016/j.jhep.2012.01.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Macha MA, et al. Guggulsterone decreases proliferation and metastatic behavior of pancreatic cancer cells by modulating JAK/STAT and Src/FAK signaling. Cancer Lett. 2013;341:166–177. doi: 10.1016/j.canlet.2013.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Sun X, Zhang J, Wang L, Tian Z. Growth inhibition of human hepatocellular carcinoma cells by blocking STAT3 activation with decoy-ODN. Cancer Lett. 2008;262:201–213. doi: 10.1016/j.canlet.2007.12.009. [DOI] [PubMed] [Google Scholar]
- 386.Wang H, et al. Tissue inhibitor of metalloproteinase 1 (TIMP-1) deficiency exacerbates carbon tetrachloride-induced liver injury and fibrosis in mice: involvement of hepatocyte STAT3 in TIMP-1 production. Cell Biosci. 2011;1:14. doi: 10.1186/2045-3701-1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Matsui F, et al. Mesenchymal stem cells protect against obstruction-induced renal fibrosis by decreasing STAT3 activation and STAT3-dependent MMP-9 production. Am. J. Physiol. Ren. Physiol. 2017;312:F25–F32. doi: 10.1152/ajprenal.00311.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 388.Zhang Y, et al. Inhibition of casein kinase II reduces TGFβ induced fibroblast activation and ameliorates experimental fibrosis. Ann. Rheum. Dis. 2015;74:936–943. doi: 10.1136/annrheumdis-2013-204256. [DOI] [PubMed] [Google Scholar]
- 389.Ogata H, et al. Loss of SOCS3 in the liver promotes fibrosis by enhancing STAT3-mediated TGF-beta1 production. Oncogene. 2006;25:2520–2530. doi: 10.1038/sj.onc.1209281. [DOI] [PubMed] [Google Scholar]
- 390.Zhao J, Qi Y-F, Yu Y-R. STAT3: a key regulator in liver fibrosis. Ann. Hepatol. 2021;21:100224. doi: 10.1016/j.aohep.2020.06.010. [DOI] [PubMed] [Google Scholar]
- 391.Pan J, et al. Mechanical stretch activates the JAK/STAT pathway in rat cardiomyocytes. Circ. Res. 1999;84:1127–1136. doi: 10.1161/01.RES.84.10.1127. [DOI] [PubMed] [Google Scholar]
- 392.Wang T-L, Yang Y-H, Chang H, Hung C-R. Angiotensin II signals mechanical stretch-induced cardiac matrix metalloproteinase expression via JAK-STAT pathway. J. Mol. Cell Cardiol. 2004;37:785–794. doi: 10.1016/j.yjmcc.2004.06.016. [DOI] [PubMed] [Google Scholar]
- 393.Tschumperlin DJ, Lagares D. Mechano-therapeutics: targeting mechanical signaling in fibrosis and tumor stroma. Pharm. Ther. 2020;212:107575. doi: 10.1016/j.pharmthera.2020.107575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Chen S, et al. Combined use of leptin and mechanical stress has osteogenic effects on ossification of the posterior longitudinal ligament. Eur. Spine J. 2018;27:1757–1766. doi: 10.1007/s00586-018-5663-4. [DOI] [PubMed] [Google Scholar]
- 395.Ruwhof C, van der Laarse A. Mechanical stress-induced cardiac hypertrophy: mechanisms and signal transduction pathways. Cardiovasc. Res. 2000;47:23–37. doi: 10.1016/S0008-6363(00)00076-6. [DOI] [PubMed] [Google Scholar]
- 396.Mill C, George SJ. Wnt signalling in smooth muscle cells and its role in cardiovascular disorders. Cardiovasc. Res. 2012;95:233–240. doi: 10.1093/cvr/cvs141. [DOI] [PubMed] [Google Scholar]
- 397.Liu J, et al. Wnt/β-catenin signalling: function, biological mechanisms, and therapeutic opportunities. Signal. Transduct. Target. Ther. 2022;7:3. doi: 10.1038/s41392-021-00762-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Yu F, et al. Wnt/β-catenin signaling in cancers and targeted therapies. Signal. Transduct. Target. Ther. 2021;6:307. doi: 10.1038/s41392-021-00701-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Niehrs C, Shen J. Regulation of Lrp6 phosphorylation. Cell Mol. Life Sci. 2010;67:2551–2562. doi: 10.1007/s00018-010-0329-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Miao J, et al. Wnt/β-catenin/RAS signaling mediates age-related renal fibrosis and is associated with mitochondrial dysfunction. Aging Cell. 2019;18:e13004. doi: 10.1111/acel.13004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 401.Schunk SJ, Floege J, Fliser D, Speer T. WNT-β-catenin signalling - a versatile player in kidney injury and repair. Nat. Rev. Nephrol. 2021;17:172–184. doi: 10.1038/s41581-020-00343-w. [DOI] [PubMed] [Google Scholar]
- 402.Tan RJ, Zhou D, Zhou L, Liu Y. Wnt/β-catenin signaling and kidney fibrosis. Kidney Int. Suppl. 2014;4:84–90. doi: 10.1038/kisup.2014.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 403.Gonzalez DM, Medici D. Signaling mechanisms of the epithelial-mesenchymal transition. Sci. Signal. 2014;7:re8. doi: 10.1126/scisignal.2005189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Feng Y, et al. Wnt/-catenin-promoted macrophage alternative activation contributes to kidney fibrosis. J. Am. Soc. Nephrol. 2018;29:182–193. doi: 10.1681/ASN.2017040391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 405.Zhou D, et al. Tubule-derived Wnts are required for fibroblast activation and kidney fibrosis. J. Am. Soc. Nephrol. 2017;28:2322–2336. doi: 10.1681/ASN.2016080902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 406.DiRenzo DM, et al. A crosstalk between TGF-β/Smad3 and Wnt/β-catenin pathways promotes vascular smooth muscle cell proliferation. Cell. Signal. 2016;28:498–505. doi: 10.1016/j.cellsig.2016.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 407.Vincent KR, Conrad BP, Fregly BJ, Vincent HK. The pathophysiology of osteoarthritis: a mechanical perspective on the knee joint. PM R. 2012;4:S3–S9. doi: 10.1016/j.pmrj.2012.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 408.Chiu J-J, Chien S. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 2011;91:327–387. doi: 10.1152/physrev.00047.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Robinson JA, et al. Wnt/beta-catenin signaling is a normal physiological response to mechanical loading in bone. J. Biol. Chem. 2006;281:31720–31728. doi: 10.1016/S0021-9258(19)84086-3. [DOI] [PubMed] [Google Scholar]
- 410.Armstrong VJ, et al. Wnt/beta-catenin signaling is a component of osteoblastic bone cell early responses to load-bearing and requires estrogen receptor alpha. J. Biol. Chem. 2007;282:20715–20727. doi: 10.1074/jbc.M703224200. [DOI] [PubMed] [Google Scholar]
- 411.Mullender MG, Huiskes R. Proposal for the regulatory mechanism of Wolff’s law. J. Orthop. Res. 1995;13:503–512. doi: 10.1002/jor.1100130405. [DOI] [PubMed] [Google Scholar]
- 412.Warboys CM. Mechanoactivation of Wnt/β-catenin pathways in health and disease. Emerg. Top. Life Sci. 2018;2:701–712. doi: 10.1042/ETLS20180042. [DOI] [PubMed] [Google Scholar]
- 413.Jiang Y-Y, et al. BIO alleviated compressive mechanical force-mediated mandibular cartilage pathological changes through Wnt/β-catenin signaling activation. J. Orthop. Res. 2018;36:1228–1237. doi: 10.1002/jor.23748. [DOI] [PubMed] [Google Scholar]
- 414.Samuel MS, et al. Actomyosin-mediated cellular tension drives increased tissue stiffness and β-catenin activation to induce epidermal hyperplasia and tumor growth. Cancer Cell. 2011;19:776–791. doi: 10.1016/j.ccr.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Mouw JK, et al. Tissue mechanics modulate microRNA-dependent PTEN expression to regulate malignant progression. Nat. Med. 2014;20:360–367. doi: 10.1038/nm.3497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Benham-Pyle BW, Pruitt BL, Nelson WJ. Cell adhesion. Mechanical strain induces E-cadherin-dependent Yap1 and β-catenin activation to drive cell cycle entry. Science. 2015;348:1024–1027. doi: 10.1126/science.aaa4559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Zhong A, Mirzaei Z, Simmons CA. The roles of matrix stiffness and ß-catenin signaling in endothelial-to-mesenchymal transition of aortic valve endothelial cells. Cardiovasc. Eng. Technol. 2018;9:158–167. doi: 10.1007/s13239-018-0363-0. [DOI] [PubMed] [Google Scholar]
- 418.Patel AL, Shvartsman SY. Outstanding questions in developmental ERK signaling. Development. 2018;145:dev143818. doi: 10.1242/dev.143818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Lavoie H, Gagnon J, Therrien M. ERK signalling: a master regulator of cell behaviour, life and fate. Nat. Rev. Mol. Cell Biol. 2020;21:607–632. doi: 10.1038/s41580-020-0255-7. [DOI] [PubMed] [Google Scholar]
- 420.Voisin L, Saba-El-Leil MK, Julien C, Frémin C, Meloche S. Genetic demonstration of a redundant role of extracellular signal-regulated kinase 1 (ERK1) and ERK2 mitogen-activated protein kinases in promoting fibroblast proliferation. Mol. Cell Biol. 2010;30:2918–2932. doi: 10.1128/MCB.00131-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Drosten M, et al. Genetic analysis of Ras signalling pathways in cell proliferation, migration and survival. EMBO J. 2010;29:1091–1104. doi: 10.1038/emboj.2010.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Kale J, Osterlund EJ, Andrews DW. BCL-2 family proteins: changing partners in the dance towards death. Cell Death Differ. 2018;25:65–80. doi: 10.1038/cdd.2017.186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Johnston LA, Prober DA, Edgar BA, Eisenman RN, Gallant P. Drosophila myc regulates cellular growth during development. Cell. 1999;98:779–790. doi: 10.1016/S0092-8674(00)81512-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Ying H, et al. Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell. 2012;149:656–670. doi: 10.1016/j.cell.2012.01.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Richard DE, Berra E, Gothié E, Roux D, Pouysségur J. p42/p44 mitogen-activated protein kinases phosphorylate hypoxia-inducible factor 1alpha (HIF-1alpha) and enhance the transcriptional activity of HIF-1. J. Biol. Chem. 1999;274:32631–32637. doi: 10.1074/jbc.274.46.32631. [DOI] [PubMed] [Google Scholar]
- 426.Schödel J, et al. High-resolution genome-wide mapping of HIF-binding sites by ChIP-seq. Blood. 2011;117:e207–e217. doi: 10.1182/blood-2010-10-314427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Tanimura S, Takeda K. ERK signalling as a regulator of cell motility. J. Biochem. 2017;162:145–154. doi: 10.1093/jb/mvx048. [DOI] [PubMed] [Google Scholar]
- 428.Nett IR, Mulas C, Gatto L, Lilley KS, Smith A. Negative feedback via RSK modulates Erk-dependent progression from naïve pluripotency. EMBO Rep. 2018;19:e45642. doi: 10.15252/embr.201745642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 429.Gao Y, et al. Mechanical strain promotes skin fibrosis through LRG-1 induction mediated by ELK1 and ERK signalling. Commun. Biol. 2019;2:359. doi: 10.1038/s42003-019-0600-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Lai J, et al. β-Adrenoceptor signaling regulates proliferation and contraction of human bladder smooth muscle cells under pathological hydrostatic pressure. J. Cell Biochem. 2019;120:17872–17886. doi: 10.1002/jcb.29056. [DOI] [PubMed] [Google Scholar]
- 431.Wang J, Shang Y-X, Cai X-X, Liu L-Y. Vasoactive intestinal peptide inhibits airway smooth muscle cell proliferation in a mouse model of asthma via the ERK1/2 signaling pathway. Exp. Cell Res. 2018;364:168–174. doi: 10.1016/j.yexcr.2018.01.042. [DOI] [PubMed] [Google Scholar]
- 432.Liu D, et al. TMEM16A regulates pulmonary arterial smooth muscle cells proliferation via p38MAPK/ERK pathway in high pulmonary blood flow-induced pulmonary arterial hypertension. J. Vasc. Res. 2020;58:27–37. doi: 10.1159/000511267. [DOI] [PubMed] [Google Scholar]
- 433.Li Q, et al. Rap1 promotes proliferation and migration of vascular smooth muscle cell via the ERK pathway. Pathol. Res. Pract. 2018;214:1045–1050. doi: 10.1016/j.prp.2018.04.007. [DOI] [PubMed] [Google Scholar]
- 434.Schafer S, et al. IL-11 is a crucial determinant of cardiovascular fibrosis. Nature. 2017;552:110–115. doi: 10.1038/nature24676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Weng C-H, et al. Interleukin-17A induces renal fibrosis through the ERK and Smad signaling pathways. Biomed. Pharmacother. 2020;123:109741. doi: 10.1016/j.biopha.2019.109741. [DOI] [PubMed] [Google Scholar]
- 436.Ng B, Cook SA, Schafer S. Interleukin-11 signaling underlies fibrosis, parenchymal dysfunction, and chronic inflammation of the airway. Exp. Mol. Med. 2020;52:1871–1878. doi: 10.1038/s12276-020-00531-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Guo X, Cen Y, Wang J, Jiang H. CXCL10-induced IL-9 promotes liver fibrosis via Raf/MEK/ERK signaling pathway. Biomed. Pharmacother. 2018;105:282–289. doi: 10.1016/j.biopha.2018.05.128. [DOI] [PubMed] [Google Scholar]
- 438.Wong VW, et al. Focal adhesion kinase links mechanical force to skin fibrosis via inflammatory signaling. Nat. Med. 2011;18:148–152. doi: 10.1038/nm.2574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Xue T, et al. Epigenetic regulation in fibrosis progress. Pharm. Res. 2021;173:105910. doi: 10.1016/j.phrs.2021.105910. [DOI] [PubMed] [Google Scholar]
- 440.Peixoto P, Cartron P-F, Serandour AA, Hervouet E. From 1957 to nowadays: a brief history of epigenetics. Int. J. Mol. Sci. 2020;21:7571. doi: 10.3390/ijms21207571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Cavalli G, Heard E. Advances in epigenetics link genetics to the environment and disease. Nature. 2019;571:489–499. doi: 10.1038/s41586-019-1411-0. [DOI] [PubMed] [Google Scholar]
- 442.Dawson MA, Kouzarides T. Cancer epigenetics: from mechanism to therapy. Cell. 2012;150:12–27. doi: 10.1016/j.cell.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 443.Pal S, Tyler JK. Epigenetics and aging. Sci. Adv. 2016;2:e1600584. doi: 10.1126/sciadv.1600584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 444.O’Reilly S. Epigenetics in fibrosis. Mol. Asp. Med. 2017;54:89–102. doi: 10.1016/j.mam.2016.10.001. [DOI] [PubMed] [Google Scholar]
- 445.Leeper NJ, Maegdefessel L. Non-coding RNAs: key regulators of smooth muscle cell fate in vascular disease. Cardiovasc. Res. 2018;114:611–621. doi: 10.1093/cvr/cvx249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 446.Cheng C, et al. LncRNA MALAT1 regulates proliferation and apoptosis of vascular smooth muscle cells by targeting miRNA-124-3p/PPARα axis. Eur. Rev. Med. Pharm. Sci. 2019;23:9025–9032. doi: 10.26355/eurrev_201910_19304. [DOI] [PubMed] [Google Scholar]
- 447.Kaukonen R, et al. Normal stroma suppresses cancer cell proliferation via mechanosensitive regulation of JMJD1a-mediated transcription. Nat. Commun. 2016;7:12237. doi: 10.1038/ncomms12237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Ligresti G, et al. CBX5/G9a/H3K9me-mediated gene repression is essential to fibroblast activation during lung fibrosis. JCI Insight. 2019;5:e127111. doi: 10.1172/jci.insight.127111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Bangs F, Anderson KV. Primary cilia and mammalian Hedgehog signaling. Cold Spring Harb. Perspect. Biol. 2017;9:a028175. doi: 10.1101/cshperspect.a028175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 450.Wang J, Cao J, Dickson AL, Poss KD. Epicardial regeneration is guided by cardiac outflow tract and Hedgehog signalling. Nature. 2015;522:226–230. doi: 10.1038/nature14325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.Lee RTH, Zhao Z, Ingham PW. Hedgehog signalling. Development. 2016;143:367–372. doi: 10.1242/dev.120154. [DOI] [PubMed] [Google Scholar]
- 452.Wehner D, Weidinger G. Signaling networks organizing regenerative growth of the zebrafish fin. Trends Genet. 2015;31:336–343. doi: 10.1016/j.tig.2015.03.012. [DOI] [PubMed] [Google Scholar]
- 453.Yang J-J, Tao H, Li J. Hedgehog signaling pathway as key player in liver fibrosis: new insights and perspectives. Expert Opin. Ther. Targets. 2014;18:1011–1021. doi: 10.1517/14728222.2014.927443. [DOI] [PubMed] [Google Scholar]
- 454.Edeling M, Ragi G, Huang S, Pavenstädt H, Susztak K. Developmental signalling pathways in renal fibrosis: the roles of Notch, Wnt and Hedgehog. Nat. Rev. Nephrol. 2016;12:426–439. doi: 10.1038/nrneph.2016.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 455.de Oliveira FL, et al. Galectin-3, histone deacetylases, and Hedgehog signaling: possible convergent targets in schistosomiasis-induced liver fibrosis. PLoS Negl. Trop. Dis. 2017;11:e0005137. doi: 10.1371/journal.pntd.0005137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Effendi WI, Nagano T. The Hedgehog signaling pathway in idiopathic pulmonary fibrosis: resurrection time. Int. J. Mol. Sci. 2021;23:171. doi: 10.3390/ijms23010171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Sun C, Ma Q, Yin J, Zhang H, Liu X. WISP-1 induced by mechanical stress contributes to fibrosis and hypertrophy of the ligamentum flavum through Hedgehog-Gli1 signaling. Exp. Mol. Med. 2021;53:1068–1079. doi: 10.1038/s12276-021-00636-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 458.Xie C, et al. Yap1 protein regulates vascular smooth muscle cell phenotypic switch by interaction with myocardin. J. Biol. Chem. 2012;287:14598–14605. doi: 10.1074/jbc.M111.329268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 459.Wang, X. et al. Yes-associated protein reacts differently in vascular smooth muscle cells under different intensities of mechanical stretch. Aging14, 286–296 (2022). [DOI] [PMC free article] [PubMed]
- 460.Lee J, Kang H. Hypoxia promotes vascular smooth muscle cell proliferation through microRNA-mediated suppression of cyclin-dependent kinase inhibitors. Cells. 2019;8:802. doi: 10.3390/cells8080802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Liu W, et al. Sodium aescinate significantly suppress postoperative peritoneal adhesion by inhibiting the RhoA/ROCK signaling pathway. Phytomedicine. 2020;69:153193. doi: 10.1016/j.phymed.2020.153193. [DOI] [PubMed] [Google Scholar]
- 462.Fang H, et al. Notoginsenoside R1 inhibits vascular smooth muscle cell proliferation, migration and neointimal hyperplasia through PI3K/Akt signaling. Sci. Rep. 2018;8:7595. doi: 10.1038/s41598-018-25874-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Cheng W, et al. ABCA1 inhibits PDGF-induced proliferation and migration of rat airway smooth muscle cell through blocking TLR2/NF-κB/NFATc1 signaling. J. Cell Biochem. 2018;119:7388–7396. doi: 10.1002/jcb.27046. [DOI] [PubMed] [Google Scholar]
- 464.Liu Y, et al. Nur77 suppresses pulmonary artery smooth muscle cell proliferation through inhibition of the STAT3/Pim-1/NFAT pathway. Am. J. Respir. Cell Mol. Biol. 2014;50:379–388. doi: 10.1165/rcmb.2013-0198OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Lv Z, et al. NLRP3 inflammasome activation contributes to mechanical stretch-induced endothelial-mesenchymal transition and pulmonary fibrosis. Crit. Care Med. 2018;46:e49–e58. doi: 10.1097/CCM.0000000000002799. [DOI] [PubMed] [Google Scholar]
- 466.Zhang Q, et al. Signaling pathways and targeted therapy for myocardial infarction. Signal. Transduct. Target. Ther. 2022;7:78. doi: 10.1038/s41392-022-00925-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 467.Farge E. Mechanotransduction in development. Curr. Top. Dev. Biol. 2011;95:243–265. doi: 10.1016/B978-0-12-385065-2.00008-6. [DOI] [PubMed] [Google Scholar]
- 468.Sutlive J, et al. Generation, transmission, and regulation of mechanical forces in embryonic morphogenesis. Small. 2022;18:e2103466. doi: 10.1002/smll.202103466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Wozniak MA, Chen CS. Mechanotransduction in development: a growing role for contractility. Nat. Rev. Mol. Cell Biol. 2009;10:34–43. doi: 10.1038/nrm2592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 470.Goodwin K, Nelson CM. Mechanics of development. Dev. Cell. 2021;56:240–250. doi: 10.1016/j.devcel.2020.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Roman BL, Pekkan K. Mechanotransduction in embryonic vascular development. Biomech. Model Mechanobiol. 2012;11:1149–1168. doi: 10.1007/s10237-012-0412-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Majkut S, Dingal PCDP, Discher DE. Stress sensitivity and mechanotransduction during heart development. Curr. Biol. 2014;24:R495–R501. doi: 10.1016/j.cub.2014.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Dzamba BJ, DeSimone DW. Extracellular matrix (ECM) and the sculpting of embryonic tissues. Curr. Top. Dev. Biol. 2018;130:245–274. doi: 10.1016/bs.ctdb.2018.03.006. [DOI] [PubMed] [Google Scholar]
- 474.Abuwarda H, Pathak MM. Mechanobiology of neural development. Curr. Opin. Cell Biol. 2020;66:104–111. doi: 10.1016/j.ceb.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Schiele NR, Marturano JE, Kuo CK. Mechanical factors in embryonic tendon development: potential cues for stem cell tenogenesis. Curr. Opin. Biotechnol. 2013;24:834–840. doi: 10.1016/j.copbio.2013.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Wong VW, Akaishi S, Longaker MT, Gurtner GC. Pushing back: wound mechanotransduction in repair and regeneration. J. Invest. Dermatol. 2011;131:2186–2196. doi: 10.1038/jid.2011.212. [DOI] [PubMed] [Google Scholar]
- 477.Hsu C-K, et al. Mechanical forces in skin disorders. J. Dermatol. Sci. 2018;90:232–240. doi: 10.1016/j.jdermsci.2018.03.004. [DOI] [PubMed] [Google Scholar]
- 478.Guo Y, et al. Mechanical stretch induced skin regeneration: molecular and cellular mechanism in skin soft tissue expansion. Int. J. Mol. Sci. 2022;23:9622. doi: 10.3390/ijms23179622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 479.Bhatt T, et al. Initiation of wound healing is regulated by the convergence of mechanical and epigenetic cues. PLoS Biol. 2022;20:e3001777. doi: 10.1371/journal.pbio.3001777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Tan P-C, et al. Mechanical stretching can modify the papillary dermis pattern and papillary fibroblast characteristics during skin regeneration. J. Invest. Dermatol. 2022;142:2384.e8–2394.e8. doi: 10.1016/j.jid.2021.11.043. [DOI] [PubMed] [Google Scholar]
- 481.Eckes B, Nischt R, Krieg T. Cell-matrix interactions in dermal repair and scarring. Fibrogenesis Tissue Repair. 2010;3:4. doi: 10.1186/1755-1536-3-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Rustad KC, Wong VW, Gurtner GC. The role of focal adhesion complexes in fibroblast mechanotransduction during scar formation. Differentiation. 2013;86:87–91. doi: 10.1016/j.diff.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 483.He J, et al. Mechanical stretch promotes hypertrophic scar formation through mechanically activated cation channel Piezo1. Cell Death Dis. 2021;12:226. doi: 10.1038/s41419-021-03481-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 484.Poitelon Y, et al. YAP and TAZ control peripheral myelination and the expression of laminin receptors in Schwann cells. Nat. Neurosci. 2016;19:879–887. doi: 10.1038/nn.4316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Gerber D, et al. Transcriptional profiling of mouse peripheral nerves to the single-cell level to build a sciatic nerve ATlas (SNAT) Elife. 2021;10:e58591. doi: 10.7554/eLife.58591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 486.Kubo Y, et al. Shootin1-cortactin interaction mediates signal-force transduction for axon outgrowth. J. Cell Biol. 2015;210:663–676. doi: 10.1083/jcb.201505011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 487.Abe K, et al. Mechanosensitive axon outgrowth mediated by L1-laminin clutch interface. Biophys. J. 2021;120:3566–3576. doi: 10.1016/j.bpj.2021.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 488.Acheta J, et al. Piezo channels contribute to the regulation of myelination in Schwann cells. Glia. 2022;70:2276–2289. doi: 10.1002/glia.24251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 489.Nichol RH, Catlett TS, Onesto MM, Hollender D, Gómez TM. Environmental elasticity regulates cell-type specific RHOA signaling and neuritogenesis of human neurons. Stem Cell Rep. 2019;13:1006–1021. doi: 10.1016/j.stemcr.2019.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 490.Desmond ME, Levitan ML, Haas AR. Internal luminal pressure during early chick embryonic brain growth: descriptive and empirical observations. Anat. Rec. A Discov. Mol. Cell Evol. Biol. 2005;285:737–747. doi: 10.1002/ar.a.20211. [DOI] [PubMed] [Google Scholar]
- 491.Garcia KE, Stewart WG, Espinosa MG, Gleghorn JP, Taber LA. Molecular and mechanical signals determine morphogenesis of the cerebral hemispheres in the chicken embryo. Development. 2019;146:dev174318. doi: 10.1242/dev.174318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 492.Motz CT, Kabat V, Saxena T, Bellamkonda RV, Zhu C. Neuromechanobiology: an expanding field driven by the force of greater focus. Adv. Health. Mater. 2021;10:e2100102. doi: 10.1002/adhm.202100102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 493.Kong L, et al. Biomechanical microenvironment in peripheral nerve regeneration: from pathophysiological understanding to tissue engineering development. Theranostics. 2022;12:4993–5014. doi: 10.7150/thno.74571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 494.Dennerll TJ, Lamoureux P, Buxbaum RE, Heidemann SR. The cytomechanics of axonal elongation and retraction. J. Cell Biol. 1989;109:3073–3083. doi: 10.1083/jcb.109.6.3073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 495.Zheng J, et al. Tensile regulation of axonal elongation and initiation. J. Neurosci. 1991;11:1117–1125. doi: 10.1523/JNEUROSCI.11-04-01117.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 496.Myers JP, Santiago-Medina M, Gomez TM. Regulation of axonal outgrowth and pathfinding by integrin-ECM interactions. Dev. Neurobiol. 2011;71:901–923. doi: 10.1002/dneu.20931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.Bard L, et al. A molecular clutch between the actin flow and N-cadherin adhesions drives growth cone migration. J. Neurosci. 2008;28:5879–5890. doi: 10.1523/JNEUROSCI.5331-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 498.Block SM, Goldstein LS, Schnapp BJ. Bead movement by single kinesin molecules studied with optical tweezers. Nature. 1990;348:348–352. doi: 10.1038/348348a0. [DOI] [PubMed] [Google Scholar]
- 499.Zimta A-A, Baru O, Badea M, Buduru SD, Berindan-Neagoe I. The role of angiogenesis and pro-angiogenic exosomes in regenerative dentistry. Int. J. Mol. Sci. 2019;20:406. doi: 10.3390/ijms20020406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 500.Van Hove AH, Benoit DSW. Depot-based delivery systems for pro-angiogenic peptides: a review. Front. Bioeng. Biotechnol. 2015;3:102. doi: 10.3389/fbioe.2015.00102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 501.Blanco R, Gerhardt H. VEGF and Notch in tip and stalk cell selection. Cold Spring Harb. Perspect. Med. 2013;3:a006569. doi: 10.1101/cshperspect.a006569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 502.Mammoto A, et al. A mechanosensitive transcriptional mechanism that controls angiogenesis. Nature. 2009;457:1103–1108. doi: 10.1038/nature07765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 503.Kang H, et al. Piezo1 mediates angiogenesis through activation of MT1-MMP signaling. Am. J. Physiol. Cell Physiol. 2019;316:C92–C103. doi: 10.1152/ajpcell.00346.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 504.Huang J-Q, et al. Mechanically activated calcium channel PIEZO1 modulates radiation-induced epithelial-mesenchymal transition by forming a positive feedback with TGF-β1. Front. Mol. Biosci. 2021;8:725275. doi: 10.3389/fmolb.2021.725275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 505.Chachisvilis M, Zhang Y-L, Frangos JA. G protein-coupled receptors sense fluid shear stress in endothelial cells. Proc. Natl Acad. Sci. USA. 2006;103:15463–15468. doi: 10.1073/pnas.0607224103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 506.Ranade SS, et al. Piezo1, a mechanically activated ion channel, is required for vascular development in mice. Proc. Natl Acad. Sci. USA. 2014;111:10347–10352. doi: 10.1073/pnas.1409233111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 507.Souilhol C, et al. Endothelial responses to shear stress in atherosclerosis: a novel role for developmental genes. Nat. Rev. Cardiol. 2020;17:52–63. doi: 10.1038/s41569-019-0239-5. [DOI] [PubMed] [Google Scholar]
- 508.Chouinard-Pelletier G, Jahnsen ED, Jones EAV. Increased shear stress inhibits angiogenesis in veins and not arteries during vascular development. Angiogenesis. 2013;16:71–83. doi: 10.1007/s10456-012-9300-2. [DOI] [PubMed] [Google Scholar]
- 509.Baeyens N, Schwartz MA. Biomechanics of vascular mechanosensation and remodeling. Mol. Biol. Cell. 2016;27:7–11. doi: 10.1091/mbc.E14-11-1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 510.Kouri FM, Eickelberg O. Transforming growth factor-alpha, a novel mediator of strain-induced vascular remodeling. Circ. Res. 2006;99:348–350. doi: 10.1161/01.RES.0000239429.72678.e1. [DOI] [PubMed] [Google Scholar]
- 511.Birukov KG. Cyclic stretch, reactive oxygen species, and vascular remodeling. Antioxid. Redox Signal. 2009;11:1651–1667. doi: 10.1089/ars.2008.2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 512.Zhao X-H, et al. Force activates smooth muscle alpha-actin promoter activity through the Rho signaling pathway. J. Cell Sci. 2007;120:1801–1809. doi: 10.1242/jcs.001586. [DOI] [PubMed] [Google Scholar]
- 513.Wu Y, et al. Cyclic stretch-induced the cytoskeleton rearrangement and gene expression of cytoskeletal regulators in human periodontal ligament cells. Acta Odontol. Scand. 2017;75:507–516. doi: 10.1080/00016357.2017.1347823. [DOI] [PubMed] [Google Scholar]
- 514.Pan J, Wang T, Wang L, Chen W, Song M. Cyclic strain-induced cytoskeletal rearrangement of human periodontal ligament cells via the Rho signaling pathway. PLoS ONE. 2014;9:e91580. doi: 10.1371/journal.pone.0091580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Zhao X, Kwan JYY, Yip K, Liu PP, Liu F-F. Targeting metabolic dysregulation for fibrosis therapy. Nat. Rev. Drug Discov. 2020;19:57–75. doi: 10.1038/s41573-019-0040-5. [DOI] [PubMed] [Google Scholar]
- 516.Thannickal VJ, Zhou Y, Gaggar A, Duncan SR. Fibrosis: ultimate and proximate causes. J. Clin. Investig. 2014;124:4673–4677. doi: 10.1172/JCI74368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 517.Montesi SB, Désogère P, Fuchs BC, Caravan P. Molecular imaging of fibrosis: recent advances and future directions. J. Clin. Investig. 2019;129:24–33. doi: 10.1172/JCI122132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 518.Wynn TA. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008;214:199–210. doi: 10.1002/path.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 519.Zhang L, Huang C, Fan S. Mangiferin and organ fibrosis: a mini review. Biofactors. 2021;47:59–68. doi: 10.1002/biof.1693. [DOI] [PubMed] [Google Scholar]
- 520.Wynn TA. Integrating mechanisms of pulmonary fibrosis. J. Exp. Med. 2011;208:1339–1350. doi: 10.1084/jem.20110551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 521.Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat. Med. 2012;18:1028–1040. doi: 10.1038/nm.2807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 522.Rieder F, Fiocchi C. Intestinal fibrosis in IBD–a dynamic, multifactorial process. Nat. Rev. Gastroenterol. Hepatol. 2009;6:228–235. doi: 10.1038/nrgastro.2009.31. [DOI] [PubMed] [Google Scholar]
- 523.Kinchen J, et al. Structural remodeling of the human colonic mesenchyme in inflammatory bowel disease. Cell. 2018;175:372.e17–386.e17. doi: 10.1016/j.cell.2018.08.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 524.Kumar S, et al. HIMF (hypoxia-induced mitogenic factor)-IL (interleukin)-6 signaling mediates cardiomyocyte-fibroblast crosstalk to promote cardiac hypertrophy and fibrosis. Hypertension. 2019;73:1058–1070. doi: 10.1161/HYPERTENSIONAHA.118.12267. [DOI] [PubMed] [Google Scholar]
- 525.Hu S, Liu H, Hu Z, Li L, Yang Y. Follistatin-like 1: a dual regulator that promotes cardiomyocyte proliferation and fibrosis. J. Cell Physiol. 2020;235:5893–5902. doi: 10.1002/jcp.29588. [DOI] [PubMed] [Google Scholar]
- 526.Nosalski R, et al. T-cell-derived miRNA-214 mediates perivascular fibrosis in hypertension. Circ. Res. 2020;126:988–1003. doi: 10.1161/CIRCRESAHA.119.315428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 527.Xue D, et al. Expansion of Fcγ receptor IIIa-positive macrophages, ficolin 1-positive monocyte-derived dendritic cells, and plasmacytoid dendritic cells associated with severe skin disease in systemic sclerosis. Arthritis Rheumatol. 2022;74:329–341. doi: 10.1002/art.41813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 528.Gieseck RL, Wilson MS, Wynn TA. Type 2 immunity in tissue repair and fibrosis. Nat. Rev. Immunol. 2018;18:62–76. doi: 10.1038/nri.2017.90. [DOI] [PubMed] [Google Scholar]
- 529.Wynn TA, Vannella KM. Macrophages in tissue repair, regeneration, and fibrosis. Immunity. 2016;44:450–462. doi: 10.1016/j.immuni.2016.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 530.Pelly VS, et al. IL-4-producing ILC2s are required for the differentiation of T2 cells following Heligmosomoides polygyrus infection. Mucosal Immunol. 2016;9:1407–1417. doi: 10.1038/mi.2016.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 531.Gibbs BF, et al. Purified human peripheral blood basophils release interleukin-13 and preformed interleukin-4 following immunological activation. Eur. J. Immunol. 1996;26:2493–2498. doi: 10.1002/eji.1830261033. [DOI] [PubMed] [Google Scholar]
- 532.Sun K, Li Y-Y, Jin J. A double-edged sword of immuno-microenvironment in cardiac homeostasis and injury repair. Signal. Transduct. Target. Ther. 2021;6:79. doi: 10.1038/s41392-020-00455-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 533.Nie Y-J, Wu S-H, Xuan Y-H, Yan G. Role of IL-17 family cytokines in the progression of IPF from inflammation to fibrosis. Mil. Med. Res. 2022;9:21. doi: 10.1186/s40779-022-00382-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 534.Kalyan S, Kabelitz D. When neutrophils meet T cells: beginnings of a tumultuous relationship with underappreciated potential. Eur. J. Immunol. 2014;44:627–633. doi: 10.1002/eji.201344195. [DOI] [PubMed] [Google Scholar]
- 535.Haddad E-B, et al. Current and emerging strategies to inhibit type 2 inflammation in atopic dermatitis. Dermatol. Ther. 2022;12:1501–1533. doi: 10.1007/s13555-022-00737-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 536.Pesce J, et al. The IL-21 receptor augments Th2 effector function and alternative macrophage activation. J. Clin. Investig. 2006;116:2044–2055. doi: 10.1172/JCI27727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 537.Fertin C, et al. Interleukin-4 stimulates collagen synthesis by normal and scleroderma fibroblasts in dermal equivalents. Cell Mol. Biol. 1991;37:823–829. [PubMed] [Google Scholar]
- 538.Letterio JJ, Roberts AB. Regulation of immune responses by TGF-beta. Annu. Rev. Immunol. 1998;16:137–161. doi: 10.1146/annurev.immunol.16.1.137. [DOI] [PubMed] [Google Scholar]
- 539.Reiman RM, et al. Interleukin-5 (IL-5) augments the progression of liver fibrosis by regulating IL-13 activity. Infect. Immun. 2006;74:1471–1479. doi: 10.1128/IAI.74.3.1471-1479.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 540.Takemura N, et al. Eosinophil depletion suppresses radiation-induced small intestinal fibrosis. Sci. Transl. Med. 2018;10:eaan0333. doi: 10.1126/scitranslmed.aan0333. [DOI] [PubMed] [Google Scholar]
- 541.Passalacqua G, et al. IL-13 and idiopathic pulmonary fibrosis: possible links and new therapeutic strategies. Pulm. Pharm. Ther. 2017;45:95–100. doi: 10.1016/j.pupt.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 542.Karmele EP, et al. Anti-IL-13Rα2 therapy promotes recovery in a murine model of inflammatory bowel disease. Mucosal Immunol. 2019;12:1174–1186. doi: 10.1038/s41385-019-0189-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 543.Giuffrida P, Caprioli F, Facciotti F, Di Sabatino A. The role of interleukin-13 in chronic inflammatory intestinal disorders. Autoimmun. Rev. 2019;18:549–555. doi: 10.1016/j.autrev.2019.03.012. [DOI] [PubMed] [Google Scholar]
- 544.Jacobs I, et al. Role of eosinophils in intestinal inflammation and fibrosis in inflammatory bowel disease: an overlooked villain? Front. Immunol. 2021;12:754413. doi: 10.3389/fimmu.2021.754413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 545.Quan TE, Cowper SE, Bucala R. The role of circulating fibrocytes in fibrosis. Curr. Rheumatol. Rep. 2006;8:145–150. doi: 10.1007/s11926-006-0055-x. [DOI] [PubMed] [Google Scholar]
- 546.Zeisberg EM, et al. Endothelial-to-mesenchymal transition contributes to cardiac fibrosis. Nat. Med. 2007;13:952–961. doi: 10.1038/nm1613. [DOI] [PubMed] [Google Scholar]
- 547.Willis BC, duBois RM, Borok Z. Epithelial origin of myofibroblasts during fibrosis in the lung. Proc. Am. Thorac. Soc. 2006;3:377–382. doi: 10.1513/pats.200601-004TK. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 548.Nagashima R, Ishikawa H, Kuno Y, Kohda C, Iyoda M. IL-33 attenuates renal fibrosis via group2 innate lymphoid cells. Cytokine. 2022;157:155963. doi: 10.1016/j.cyto.2022.155963. [DOI] [PubMed] [Google Scholar]
- 549.Zhu N, et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 550.World Health Organization (WHO). WHO coronavirus (COVID-19) dashboard. https://covid19.who.int/ (2023).
- 551.Aghamirza Moghim Aliabadi H, et al. COVID-19: a systematic review and update on prevention, diagnosis, and treatment. MedComm. 2022;3:e115. doi: 10.1002/mco2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 552.John AE, Joseph C, Jenkins G, Tatler AL. COVID-19 and pulmonary fibrosis: a potential role for lung epithelial cells and fibroblasts. Immunol. Rev. 2021;302:228–240. doi: 10.1111/imr.12977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 553.Li X, et al. Pulmonary fibrosis and its related factors in discharged patients with new corona virus pneumonia: a cohort study. Respir. Res. 2021;22:203. doi: 10.1186/s12931-021-01798-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 554.Humphrey JD, Dufresne ER, Schwartz MA. Mechanotransduction and extracellular matrix homeostasis. Nat. Rev. Mol. Cell Biol. 2014;15:802–812. doi: 10.1038/nrm3896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 555.Qu J, Ouyang L, Kuo C-C, Martin DC. Stiffness, strength and adhesion characterization of electrochemically deposited conjugated polymer films. Acta Biomater. 2016;31:114–121. doi: 10.1016/j.actbio.2015.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 556.Pattappa G, Zellner J, Johnstone B, Docheva D, Angele P. Cells under pressure - the relationship between hydrostatic pressure and mesenchymal stem cell chondrogenesis. Eur. Cell Mater. 2019;37:360–381. doi: 10.22203/eCM.v037a22. [DOI] [PubMed] [Google Scholar]
- 557.Navis A, Bagnat M. Developing pressures: fluid forces driving morphogenesis. Curr. Opin. Genet. Dev. 2015;32:24–30. doi: 10.1016/j.gde.2015.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 558.Casares L, et al. Hydraulic fracture during epithelial stretching. Nat. Mater. 2015;14:343–351. doi: 10.1038/nmat4206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 559.Wang N. Review of cellular mechanotransduction. J. Phys. D Appl. Phys. 2017;50:233002. doi: 10.1088/1361-6463/aa6e18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 560.Sun K, Li X, Scherer PE. Extracellular matrix (ECM) and fibrosis in adipose tissue: overview and perspectives. Compr. Physiol. 2023;13:4387–4407. doi: 10.1002/cphy.c220020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 561.Barczyk M, Carracedo S, Gullberg D. Integrins. Cell Tissue Res. 2010;339:269–280. doi: 10.1007/s00441-009-0834-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 562.Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell. 2002;110:673–687. doi: 10.1016/S0092-8674(02)00971-6. [DOI] [PubMed] [Google Scholar]
- 563.Mei S, et al. Integrin β3 mediates sepsis and mechanical ventilation-associated pulmonary fibrosis through glycometabolic reprogramming. Lab. Investig. 2023;103:100021. doi: 10.1016/j.labinv.2022.100021. [DOI] [PubMed] [Google Scholar]
- 564.Fang X-Z, et al. Mechanosensitive ion channel Piezo1 mediates mechanical ventilation-exacerbated ARDS-associated pulmonary fibrosis. J. Adv. Res. 2022;S2090–1232:00285–00285. doi: 10.1016/j.jare.2022.12.006. [DOI] [PubMed] [Google Scholar]
- 565.Hinz B, Lagares D. Evasion of apoptosis by myofibroblasts: a hallmark of fibrotic diseases. Nat. Rev. Rheumatol. 2020;16:11–31. doi: 10.1038/s41584-019-0324-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 566.Cao C, et al. Cadherin-11 cooperates with inflammatory factors to promote the migration and invasion of fibroblast-like synoviocytes in pigmented villonodular synovitis. Theranostics. 2020;10:10573–10588. doi: 10.7150/thno.48666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 567.Sumey JL, Johnston PC, Harrell AM, Caliari SR. Hydrogel mechanics regulate fibroblast DNA methylation and chromatin condensation. Biomater. Sci. 2023;11:2886–2897. doi: 10.1039/D2BM02058K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 568.Shiraishi M, Suzuki K, Yamaguchi A. Effect of mechanical tension on fibroblast transcriptome profile and regulatory mechanisms of myocardial collagen turnover. FASEB J. 2023;37:e22841. doi: 10.1096/fj.202201899R. [DOI] [PubMed] [Google Scholar]
- 569.Spanoudes K, Gaspar D, Pandit A, Zeugolis DI. The biophysical, biochemical, and biological toolbox for tenogenic phenotype maintenance in vitro. Trends Biotechnol. 2014;32:474–482. doi: 10.1016/j.tibtech.2014.06.009. [DOI] [PubMed] [Google Scholar]
- 570.Li Y-SJ, Haga JH, Chien S. Molecular basis of the effects of shear stress on vascular endothelial cells. J. Biomech. 2005;38:1949–1971. doi: 10.1016/j.jbiomech.2004.09.030. [DOI] [PubMed] [Google Scholar]
- 571.Heil M, Eitenmüller I, Schmitz-Rixen T, Schaper W. Arteriogenesis versus angiogenesis: similarities and differences. J. Cell Mol. Med. 2006;10:45–55. doi: 10.1111/j.1582-4934.2006.tb00290.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 572.Ding B-S, et al. Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature. 2014;505:97–102. doi: 10.1038/nature12681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 573.Tschumperlin DJ. Matrix, mesenchyme, and mechanotransduction. Ann. Am. Thorac. Soc. 2015;12:S24–S29. doi: 10.1513/AnnalsATS.201407-320MG. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 574.Chen S, et al. M receptor modulates extracellular matrix synthesis via ERK1/2 signaling pathway in human bladder smooth muscle cells. J. Cell Biochem. 2020;121:4496–4504. doi: 10.1002/jcb.29688. [DOI] [PubMed] [Google Scholar]
- 575.Chen G, et al. β-Adrenoceptor regulates contraction and inflammatory cytokine expression of human bladder smooth muscle cells via autophagy under pathological hydrostatic pressure. Neurourol. Urodyn. 2020;39:2128–2138. doi: 10.1002/nau.24517. [DOI] [PubMed] [Google Scholar]
- 576.Luo D-Y, et al. Integrin αv mediates contractility whereas integrin α4 regulates proliferation of human bladder smooth muscle cells via FAK pathway under physiological stretch. J. Urol. 2013;190:1421–1429. doi: 10.1016/j.juro.2013.04.027. [DOI] [PubMed] [Google Scholar]
- 577.Brodde OE, Bruck H, Leineweber K, Seyfarth T. Presence, distribution and physiological function of adrenergic and muscarinic receptor subtypes in the human heart. Basic Res. Cardiol. 2001;96:528–538. doi: 10.1007/s003950170003. [DOI] [PubMed] [Google Scholar]
- 578.Mittmann C, Eschenhagen T, Scholz H. Cellular and molecular aspects of contractile dysfunction in heart failure. Cardiovasc. Res. 1998;39:267–275. doi: 10.1016/S0008-6363(98)00139-4. [DOI] [PubMed] [Google Scholar]
- 579.Chen G, et al. Monocyte chemotactic protein-1 regulates proliferation and contractility of human bladder smooth muscle cells under hydrostatic pressure. J. Interferon Cytokine Res. 2020;40:245–253. doi: 10.1089/jir.2019.0186. [DOI] [PubMed] [Google Scholar]
- 580.Chen L, et al. Simulated bladder pressure stimulates human bladder smooth muscle cell proliferation via the PI3K/SGK1 signaling pathway. J. Urol. 2012;188:661–667. doi: 10.1016/j.juro.2012.03.112. [DOI] [PubMed] [Google Scholar]
- 581.Lederer DJ, Martinez FJ. Idiopathic pulmonary fibrosis. N. Engl. J. Med. 2018;378:1811–1823. doi: 10.1056/NEJMra1705751. [DOI] [PubMed] [Google Scholar]
- 582.Hutchinson J, Fogarty A, Hubbard R, McKeever T. Global incidence and mortality of idiopathic pulmonary fibrosis: a systematic review. Eur. Respir. J. 2015;46:795–806. doi: 10.1183/09031936.00185114. [DOI] [PubMed] [Google Scholar]
- 583.Karimi-Shah BA, Chowdhury BA. Forced vital capacity in idiopathic pulmonary fibrosis–FDA review of pirfenidone and nintedanib. N. Engl. J. Med. 2015;372:1189–1191. doi: 10.1056/NEJMp1500526. [DOI] [PubMed] [Google Scholar]
- 584.Burgstaller G, et al. The instructive extracellular matrix of the lung: basic composition and alterations in chronic lung disease. Eur. Respir. J. 2017;50:1601805. doi: 10.1183/13993003.01805-2016. [DOI] [PubMed] [Google Scholar]
- 585.Deng Z, et al. The extracellular matrix and mechanotransduction in pulmonary fibrosis. Int. J. Biochem. Cell Biol. 2020;126:105802. doi: 10.1016/j.biocel.2020.105802. [DOI] [PubMed] [Google Scholar]
- 586.Thannickal VJ, Toews GB, White ES, Lynch JP, Martinez FJ. Mechanisms of pulmonary fibrosis. Annu. Rev. Med. 2004;55:395–417. doi: 10.1146/annurev.med.55.091902.103810. [DOI] [PubMed] [Google Scholar]
- 587.Misharin AV, et al. Monocyte-derived alveolar macrophages drive lung fibrosis and persist in the lung over the life span. J. Exp. Med. 2017;214:2387–2404. doi: 10.1084/jem.20162152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 588.Shimbori C, et al. Mechanical stress-induced mast cell degranulation activates TGF-β1 signalling pathway in pulmonary fibrosis. Thorax. 2019;74:455–465. doi: 10.1136/thoraxjnl-2018-211516. [DOI] [PubMed] [Google Scholar]
- 589.Zhang R, et al. Mechanical stress and the induction of lung fibrosis via the midkine signaling pathway. Am. J. Respir. Crit. Care Med. 2015;192:315–323. doi: 10.1164/rccm.201412-2326OC. [DOI] [PubMed] [Google Scholar]
- 590.Wang C, Yang J. Mechanical forces: the missing link between idiopathic pulmonary fibrosis and lung cancer. Eur. J. Cell Biol. 2022;101:151234. doi: 10.1016/j.ejcb.2022.151234. [DOI] [PubMed] [Google Scholar]
- 591.Gokey JJ, et al. Active epithelial Hippo signaling in idiopathic pulmonary fibrosis. JCI Insight. 2018;3:e98738. doi: 10.1172/jci.insight.98738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 592.Philp CJ, et al. Extracellular matrix cross-linking enhances fibroblast growth and protects against matrix proteolysis in lung fibrosis. Am. J. Respir. Cell Mol. Biol. 2018;58:594–603. doi: 10.1165/rcmb.2016-0379OC. [DOI] [PubMed] [Google Scholar]
- 593.Qu J, et al. Targeting mechanosensitive MDM4 promotes lung fibrosis resolution in aged mice. J. Exp. Med. 2021;218:e202033. doi: 10.1084/jem.20202033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 594.Xu P, et al. hnRNPL-activated circANKRD42 back-splicing and circANKRD42-mediated crosstalk of mechanical stiffness and biochemical signal in lung fibrosis. Mol. Ther. 2022;30:2370–2387. doi: 10.1016/j.ymthe.2022.01.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 595.Parola M, Pinzani M. Liver fibrosis: pathophysiology, pathogenetic targets and clinical issues. Mol. Asp. Med. 2019;65:37–55. doi: 10.1016/j.mam.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 596.Nababan SHH, Lesmana CRA. Portal hypertension in nonalcoholic fatty liver disease: from pathogenesis to clinical practice. J. Clin. Transl. Hepatol. 2022;10:979–985. doi: 10.14218/JCTH.2021.00593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 597.Semler EJ, Lancin PA, Dasgupta A, Moghe PV. Engineering hepatocellular morphogenesis and function via ligand-presenting hydrogels with graded mechanical compliance. Biotechnol. Bioeng. 2005;89:296–307. doi: 10.1002/bit.20328. [DOI] [PubMed] [Google Scholar]
- 598.Olsen AL, et al. Hepatic stellate cells require a stiff environment for myofibroblastic differentiation. Am. J. Physiol. Gastrointest. Liver Physiol. 2011;301:G110–G118. doi: 10.1152/ajpgi.00412.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 599.Higashi T, Friedman SL, Hoshida Y. Hepatic stellate cells as key target in liver fibrosis. Adv. Drug Deliv. Rev. 2017;121:27–42. doi: 10.1016/j.addr.2017.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 600.Yee HF. Rho directs activation-associated changes in rat hepatic stellate cell morphology via regulation of the actin cytoskeleton. Hepatology. 1998;28:843–850. doi: 10.1002/hep.510280336. [DOI] [PubMed] [Google Scholar]
- 601.Li Z, et al. Transforming growth factor-beta and substrate stiffness regulate portal fibroblast activation in culture. Hepatology. 2007;46:1246–1256. doi: 10.1002/hep.21792. [DOI] [PubMed] [Google Scholar]
- 602.Lachowski D, et al. Substrate stiffness-driven membrane tension modulates vesicular trafficking via Caveolin-1. ACS Nano. 2022;16:4322–4337. doi: 10.1021/acsnano.1c10534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 603.Friedman SL, Sheppard D, Duffield JS, Violette S. Therapy for fibrotic diseases: nearing the starting line. Sci. Transl. Med. 2013;5:167sr161. doi: 10.1126/scitranslmed.3004700. [DOI] [PubMed] [Google Scholar]
- 604.Barry-Hamilton V, et al. Allosteric inhibition of lysyl oxidase-like-2 impedes the development of a pathologic microenvironment. Nat. Med. 2010;16:1009–1017. doi: 10.1038/nm.2208. [DOI] [PubMed] [Google Scholar]
- 605.Chen W, et al. Lysyl oxidase (LOX) family members: rationale and their potential as therapeutic targets for liver fibrosis. Hepatology. 2020;72:729–741. doi: 10.1002/hep.31236. [DOI] [PubMed] [Google Scholar]
- 606.Yu SMW, Bonventre JV. Acute kidney injury and maladaptive tubular repair leading to renal fibrosis. Curr. Opin. Nephrol. Hypertens. 2020;29:310–318. doi: 10.1097/MNH.0000000000000605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 607.Li L, Fu H, Liu Y. The fibrogenic niche in kidney fibrosis: components and mechanisms. Nat. Rev. Nephrol. 2022;18:545–557. doi: 10.1038/s41581-022-00590-z. [DOI] [PubMed] [Google Scholar]
- 608.Cai Z, Xin J, Pollock DM, Pollock JS. Shear stress-mediated NO production in inner medullary collecting duct cells. Am. J. Physiol. Ren. Physiol. 2000;279:F270–F274. doi: 10.1152/ajprenal.2000.279.2.F270. [DOI] [PubMed] [Google Scholar]
- 609.Essig M, Terzi F, Burtin M, Friedlander G. Mechanical strains induced by tubular flow affect the phenotype of proximal tubular cells. Am. J. Physiol. Ren. Physiol. 2001;281:F751–F762. doi: 10.1152/ajprenal.2001.281.4.F751. [DOI] [PubMed] [Google Scholar]
- 610.Nguyen HT, et al. JNK/SAPK and p38 SAPK-2 mediate mechanical stretch-induced apoptosis via caspase-3 and -9 in NRK-52E renal epithelial cells. Nephron Exp. Nephrol. 2006;102:e49–e61. doi: 10.1159/000088401. [DOI] [PubMed] [Google Scholar]
- 611.Quinlan MR, Docherty NG, Watson RWG, Fitzpatrick JM. Exploring mechanisms involved in renal tubular sensing of mechanical stretch following ureteric obstruction. Am. J. Physiol. Ren. Physiol. 2008;295:F1–F11. doi: 10.1152/ajprenal.00576.2007. [DOI] [PubMed] [Google Scholar]
- 612.Numata T, Shimizu T, Okada Y. TRPM7 is a stretch- and swelling-activated cation channel involved in volume regulation in human epithelial cells. Am. J. Physiol. Cell Physiol. 2007;292:C460–C467. doi: 10.1152/ajpcell.00367.2006. [DOI] [PubMed] [Google Scholar]
- 613.Miyajima A, et al. Antibody to transforming growth factor-beta ameliorates tubular apoptosis in unilateral ureteral obstruction. Kidney Int. 2000;58:2301–2313. doi: 10.1046/j.1523-1755.2000.00414.x. [DOI] [PubMed] [Google Scholar]
- 614.Yoshigi M, Clark EB, Yost HJ. Quantification of stretch-induced cytoskeletal remodeling in vascular endothelial cells by image processing. Cytom. A. 2003;55:109–118. doi: 10.1002/cyto.a.10076. [DOI] [PubMed] [Google Scholar]
- 615.Hamzeh MT, Sridhara R, Alexander LD. Cyclic stretch-induced TGF-β1 and fibronectin expression is mediated by β1-integrin through c-Src- and STAT3-dependent pathways in renal epithelial cells. Am. J. Physiol. Ren. Physiol. 2015;308:F425–F436. doi: 10.1152/ajprenal.00589.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 616.Sonomura K, et al. The kinase Pyk2 is involved in renal fibrosis by means of mechanical stretch-induced growth factor expression in renal tubules. Kidney Int. 2012;81:449–457. doi: 10.1038/ki.2011.403. [DOI] [PubMed] [Google Scholar]
- 617.Grabias BM, Konstantopoulos K. The physical basis of renal fibrosis: effects of altered hydrodynamic forces on kidney homeostasis. Am. J. Physiol. Ren. Physiol. 2014;306:F473–F485. doi: 10.1152/ajprenal.00503.2013. [DOI] [PubMed] [Google Scholar]
- 618.Cattaneo I, et al. Shear stress reverses dome formation in confluent renal tubular cells. Cell Physiol. Biochem. 2011;28:673–682. doi: 10.1159/000335813. [DOI] [PubMed] [Google Scholar]
- 619.Duan Y, et al. Shear-induced reorganization of renal proximal tubule cell actin cytoskeleton and apical junctional complexes. Proc. Natl Acad. Sci. USA. 2008;105:11418–11423. doi: 10.1073/pnas.0804954105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 620.Woda CB, Leite M, Rohatgi R, Satlin LM. Effects of luminal flow and nucleotides on [Ca(2+)](i) in rabbit cortical collecting duct. Am. J. Physiol. Ren. Physiol. 2002;283:F437–F446. doi: 10.1152/ajprenal.00316.2001. [DOI] [PubMed] [Google Scholar]
- 621.Yuan Q, Tan RJ, Liu Y. Myofibroblast in kidney fibrosis: origin, activation, and regulation. Adv. Exp. Med. Biol. 2019;1165:253–283. doi: 10.1007/978-981-13-8871-2_12. [DOI] [PubMed] [Google Scholar]
- 622.Tsou P-S, Haak AJ, Khanna D, Neubig RR. Cellular mechanisms of tissue fibrosis. 8. Current and future drug targets in fibrosis: focus on Rho GTPase-regulated gene transcription. Am. J. Physiol. Cell Physiol. 2014;307:C2–C13. doi: 10.1152/ajpcell.00060.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 623.He Y, et al. Myeloid Piezo1 deletion protects renal fibrosis by restraining macrophage infiltration and activation. Hypertension. 2022;79:918–931. doi: 10.1161/HYPERTENSIONAHA.121.18750. [DOI] [PubMed] [Google Scholar]
- 624.Berk BC, Fujiwara K, Lehoux S. ECM remodeling in hypertensive heart disease. J. Clin. Investig. 2007;117:568–575. doi: 10.1172/JCI31044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 625.Xiang F-L, Fang M, Yutzey KE. Loss of β-catenin in resident cardiac fibroblasts attenuates fibrosis induced by pressure overload in mice. Nat. Commun. 2017;8:712. doi: 10.1038/s41467-017-00840-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 626.Gao X-M, White DA, Dart AM, Du X-J. Post-infarct cardiac rupture: recent insights on pathogenesis and therapeutic interventions. Pharmacol. Ther. 2012;134:156–179. doi: 10.1016/j.pharmthera.2011.12.010. [DOI] [PubMed] [Google Scholar]
- 627.Leonard BL, Smaill BH, LeGrice IJ. Structural remodeling and mechanical function in heart failure. Microsc. Microanal. 2012;18:50–67. doi: 10.1017/S1431927611012438. [DOI] [PubMed] [Google Scholar]
- 628.Weber KT. Cardiac interstitium in health and disease: the fibrillar collagen network. J. Am. Coll. Cardiol. 1989;13:1637–1652. doi: 10.1016/0735-1097(89)90360-4. [DOI] [PubMed] [Google Scholar]
- 629.Ko T, et al. Cardiac fibroblasts regulate the development of heart failure via Htra3-TGF-β-IGFBP7 axis. Nat. Commun. 2022;13:3275. doi: 10.1038/s41467-022-30630-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 630.Garoffolo G, et al. Reduction of cardiac fibrosis by interference with YAP-dependent transactivation. Circ. Res. 2022;131:239–257. doi: 10.1161/CIRCRESAHA.121.319373. [DOI] [PubMed] [Google Scholar]
- 631.Inoue R, Kurahara L-H, Hiraishi K. TRP channels in cardiac and intestinal fibrosis. Semin. Cell Dev. Biol. 2019;94:40–49. doi: 10.1016/j.semcdb.2018.11.002. [DOI] [PubMed] [Google Scholar]
- 632.Ebrahimighaei R, et al. Combined role for YAP-TEAD and YAP-RUNX2 signalling in substrate-stiffness regulation of cardiac fibroblast proliferation. Biochim. Biophys. Acta Mol. Cell Res. 2022;1869:119329. doi: 10.1016/j.bbamcr.2022.119329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 633.Chen W, Bian W, Zhou Y, Zhang J. Cardiac fibroblasts and myocardial regeneration. Front. Bioeng. Biotechnol. 2021;9:599928. doi: 10.3389/fbioe.2021.599928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 634.Frangogiannis NG. Cardiac fibrosis. Cardiovasc. Res. 2021;117:1450–1488. doi: 10.1093/cvr/cvaa324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 635.Watson CJ, et al. Mechanical stretch up-regulates the B-type natriuretic peptide system in human cardiac fibroblasts: a possible defense against transforming growth factor-β mediated fibrosis. Fibrogenesis Tissue Repair. 2012;5:9. doi: 10.1186/1755-1536-5-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 636.Gupta V, Grande-Allen KJ. Effects of static and cyclic loading in regulating extracellular matrix synthesis by cardiovascular cells. Cardiovasc. Res. 2006;72:375–383. doi: 10.1016/j.cardiores.2006.08.017. [DOI] [PubMed] [Google Scholar]
- 637.Husse B, Briest W, Homagk L, Isenberg G, Gekle M. Cyclical mechanical stretch modulates expression of collagen I and collagen III by PKC and tyrosine kinase in cardiac fibroblasts. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007;293:R1898–R1907. doi: 10.1152/ajpregu.00804.2006. [DOI] [PubMed] [Google Scholar]
- 638.Papakrivopoulou J, Lindahl GE, Bishop JE, Laurent GJ. Differential roles of extracellular signal-regulated kinase 1/2 and p38MAPK in mechanical load-induced procollagen alpha1(I) gene expression in cardiac fibroblasts. Cardiovasc. Res. 2004;61:736–744. doi: 10.1016/j.cardiores.2003.12.018. [DOI] [PubMed] [Google Scholar]
- 639.Song J, et al. Pellino1-mediated TGF-β1 synthesis contributes to mechanical stress induced cardiac fibroblast activation. J. Mol. Cell Cardiol. 2015;79:145–156. doi: 10.1016/j.yjmcc.2014.11.006. [DOI] [PubMed] [Google Scholar]
- 640.O’Donnell A, Odrowaz Z, Sharrocks AD. Immediate-early gene activation by the MAPK pathways: what do and don’t we know? Biochem. Soc. Trans. 2012;40:58–66. doi: 10.1042/BST20110636. [DOI] [PubMed] [Google Scholar]
- 641.Jiang Y, et al. Targeting extracellular matrix stiffness and mechanotransducers to improve cancer therapy. J. Hematol. Oncol. 2022;15:34. doi: 10.1186/s13045-022-01252-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 642.Dong Y, et al. Higher matrix stiffness as an independent initiator triggers epithelial-mesenchymal transition and facilitates HCC metastasis. J. Hematol. Oncol. 2019;12:112. doi: 10.1186/s13045-019-0795-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 643.Pankova D, et al. Cancer-associated fibroblasts induce a collagen cross-link switch in tumor stroma. Mol. Cancer Res. 2016;14:287–295. doi: 10.1158/1541-7786.MCR-15-0307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 644.De Felice D, Alaimo A. Mechanosensitive piezo channels in cancer: focus on altered calcium signaling in cancer cells and in tumor progression. Cancers. 2020;12:1780. doi: 10.3390/cancers12071780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 645.Lewis AH, Grandl J. Piezo1 ion channels inherently function as independent mechanotransducers. Elife. 2021;10:e70988. doi: 10.7554/eLife.70988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 646.Gkretsi V, Stylianopoulos T. Cell adhesion and matrix stiffness: coordinating cancer cell invasion and metastasis. Front. Oncol. 2018;8:145. doi: 10.3389/fonc.2018.00145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 647.Li X, Wang J. Mechanical tumor microenvironment and transduction: cytoskeleton mediates cancer cell invasion and metastasis. Int. J. Biol. Sci. 2020;16:2014–2028. doi: 10.7150/ijbs.44943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 648.Barcus CE, Keely PJ, Eliceiri KW, Schuler LA. Stiff collagen matrices increase tumorigenic prolactin signaling in breast cancer cells. J. Biol. Chem. 2013;288:12722–12732. doi: 10.1074/jbc.M112.447631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 649.Provenzano PP, Inman DR, Eliceiri KW, Keely PJ. Matrix density-induced mechanoregulation of breast cell phenotype, signaling and gene expression through a FAK-ERK linkage. Oncogene. 2009;28:4326–4343. doi: 10.1038/onc.2009.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 650.Erdogan B, et al. Cancer-associated fibroblasts promote directional cancer cell migration by aligning fibronectin. J. Cell Biol. 2017;216:3799–3816. doi: 10.1083/jcb.201704053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 651.Papageorgis P, Stylianopoulos T. Role of TGFβ in regulation of the tumor microenvironment and drug delivery (review) Int. J. Oncol. 2015;46:933–943. doi: 10.3892/ijo.2015.2816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 652.Foster CT, Gualdrini F, Treisman R. Mutual dependence of the MRTF-SRF and YAP-TEAD pathways in cancer-associated fibroblasts is indirect and mediated by cytoskeletal dynamics. Genes Dev. 2017;31:2361–2375. doi: 10.1101/gad.304501.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 653.Kim H, Ishibashi K, Okada T, Nakamura C. Mechanical property changes in breast cancer cells induced by stimulation with macrophage secretions in vitro. Micromachines. 2019;10:738. doi: 10.3390/mi10110738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 654.Wang T, Hamilla S, Cam M, Aranda-Espinoza H, Mili S. Extracellular matrix stiffness and cell contractility control RNA localization to promote cell migration. Nat. Commun. 2017;8:896. doi: 10.1038/s41467-017-00884-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 655.Fattet L, et al. Matrix rigidity controls epithelial-mesenchymal plasticity and tumor metastasis via a mechanoresponsive EPHA2/LYN complex. Dev. Cell. 2020;54:302.e7–316.e7. doi: 10.1016/j.devcel.2020.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 656.Hamant O, Inoue D, Bouchez D, Dumais J, Mjolsness E. Are microtubules tension sensors? Nat. Commun. 2019;10:2360. doi: 10.1038/s41467-019-10207-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 657.Torrino S, et al. Mechano-induced cell metabolism promotes microtubule glutamylation to force metastasis. Cell Metab. 2021;33:1342.e10–1357.e10. doi: 10.1016/j.cmet.2021.05.009. [DOI] [PubMed] [Google Scholar]
- 658.Follain G, et al. Fluids and their mechanics in tumour transit: shaping metastasis. Nat. Rev. Cancer. 2020;20:107–124. doi: 10.1038/s41568-019-0221-x. [DOI] [PubMed] [Google Scholar]
- 659.Chen X, et al. Shear stress enhances anoikis resistance of cancer cells through ROS and NO suppressed degeneration of Caveolin-1. Free Radic. Biol. Med. 2022;193:95–107. doi: 10.1016/j.freeradbiomed.2022.10.271. [DOI] [PubMed] [Google Scholar]
- 660.Alvarado-Estrada K, et al. Circulatory shear stress induces molecular changes and side population enrichment in primary tumor-derived lung cancer cells with higher metastatic potential. Sci. Rep. 2021;11:2800. doi: 10.1038/s41598-021-82634-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 661.Tang K, et al. Shear stress stimulates integrin β1 trafficking and increases directional migration of cancer cells via promoting deacetylation of microtubules. Biochim. Biophys. Acta Mol. Cell Res. 2020;1867:118676. doi: 10.1016/j.bbamcr.2020.118676. [DOI] [PubMed] [Google Scholar]
- 662.Ariffin AB, Forde PF, Jahangeer S, Soden DM, Hinchion J. Releasing pressure in tumors: what do we know so far and where do we go from here? A review. Cancer Res. 2014;74:2655–2662. doi: 10.1158/0008-5472.CAN-13-3696. [DOI] [PubMed] [Google Scholar]
- 663.Zhang T, et al. Targeting the tumor biophysical microenvironment to reduce resistance to immunotherapy. Adv. Drug Deliv. Rev. 2022;186:114319. doi: 10.1016/j.addr.2022.114319. [DOI] [PubMed] [Google Scholar]
- 664.Cha YJ, Youk JH, Kim BG, Jung WH, Cho NH. Lymphangiogenesis in breast cancer correlates with matrix stiffness on shear-wave elastography. Yonsei Med. J. 2016;57:599–605. doi: 10.3349/ymj.2016.57.3.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 665.Panzetta V, et al. Mechanical phenotyping of cells and extracellular matrix as grade and stage markers of lung tumor tissues. Acta Biomater. 2017;57:334–341. doi: 10.1016/j.actbio.2017.05.002. [DOI] [PubMed] [Google Scholar]
- 666.Miyazawa A, et al. Regulation of PD-L1 expression by matrix stiffness in lung cancer cells. Biochem. Biophys. Res. Commun. 2018;495:2344–2349. doi: 10.1016/j.bbrc.2017.12.115. [DOI] [PubMed] [Google Scholar]
- 667.Mennens SFB, et al. Substrate stiffness influences phenotype and function of human antigen-presenting dendritic cells. Sci. Rep. 2017;7:17511. doi: 10.1038/s41598-017-17787-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 668.Kim MH, et al. Secreted and membrane-associated matrix metalloproteinases of IL-2-activated NK cells and their inhibitors. J. Immunol. 2000;164:5883–5889. doi: 10.4049/jimmunol.164.11.5883. [DOI] [PubMed] [Google Scholar]
- 669.Sridharan R, Cavanagh B, Cameron AR, Kelly DJ, O’Brien FJ. Material stiffness influences the polarization state, function and migration mode of macrophages. Acta Biomater. 2019;89:47–59. doi: 10.1016/j.actbio.2019.02.048. [DOI] [PubMed] [Google Scholar]
- 670.Jain N, Moeller J, Vogel V. Mechanobiology of macrophages: how physical factors coregulate macrophage plasticity and phagocytosis. Annu. Rev. Biomed. Eng. 2019;21:267–297. doi: 10.1146/annurev-bioeng-062117-121224. [DOI] [PubMed] [Google Scholar]
- 671.Wu J, Cang S, Liu C, Ochiai W, Chiao JW. Development of human prostate cancer stem cells involves epigenomic alteration and PI3K/AKT pathway activation. Exp. Hematol. Oncol. 2020;9:12. doi: 10.1186/s40164-020-00168-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 672.Diepenbruck M, Christofori G. Epithelial-mesenchymal transition (EMT) and metastasis: yes, no, maybe? Curr. Opin. Cell Biol. 2016;43:7–13. doi: 10.1016/j.ceb.2016.06.002. [DOI] [PubMed] [Google Scholar]
- 673.Zhu P, et al. LncBRM initiates YAP1 signalling activation to drive self-renewal of liver cancer stem cells. Nat. Commun. 2016;7:13608. doi: 10.1038/ncomms13608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 674.Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2019;20:69–84. doi: 10.1038/s41580-018-0080-4. [DOI] [PubMed] [Google Scholar]
- 675.Sheridan C. First Axl inhibitor enters clinical trials. Nat. Biotechnol. 2013;31:775–776. doi: 10.1038/nbt0913-775a. [DOI] [PubMed] [Google Scholar]
- 676.Gjerdrum C, et al. Axl is an essential epithelial-to-mesenchymal transition-induced regulator of breast cancer metastasis and patient survival. Proc. Natl Acad. Sci. USA. 2010;107:1124–1129. doi: 10.1073/pnas.0909333107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 677.Buechler MB, et al. Cross-tissue organization of the fibroblast lineage. Nature. 2021;593:575–579. doi: 10.1038/s41586-021-03549-5. [DOI] [PubMed] [Google Scholar]
- 678.Kollmannsberger P, Bidan CM, Dunlop JWC, Fratzl P, Vogel V. Tensile forces drive a reversible fibroblast-to-myofibroblast transition during tissue growth in engineered clefts. Sci. Adv. 2018;4:eaao4881. doi: 10.1126/sciadv.aao4881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 679.Bu F-T, et al. Emerging therapeutic potential of adeno-associated virus-mediated gene therapy in liver fibrosis. Mol. Ther. Methods Clin. Dev. 2022;26:191–206. doi: 10.1016/j.omtm.2022.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 680.Sobrevals L, Enguita M, Quiroga J, Prieto J, Fortes P. Insulin-like growth factor I (IGF-I) expressed from an AAV1 vector leads to a complete reversion of liver cirrhosis in rats. PLoS ONE. 2016;11:e0162955. doi: 10.1371/journal.pone.0162955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 681.Xu M, et al. LECT2, a ligand for Tie1, plays a crucial role in liver fibrogenesis. Cell. 2019;178:1478.e20–1492.e20. doi: 10.1016/j.cell.2019.07.021. [DOI] [PubMed] [Google Scholar]
- 682.Tsui T-Y, et al. rAAV-mediated stable expression of heme oxygenase-1 in stellate cells: a new approach to attenuate liver fibrosis in rats. Hepatology. 2005;42:335–342. doi: 10.1002/hep.20803. [DOI] [PubMed] [Google Scholar]
- 683.Suzumura K, et al. Adeno-associated virus vector-mediated production of hepatocyte growth factor attenuates liver fibrosis in mice. Hepatol. Int. 2008;2:80–88. doi: 10.1007/s12072-007-9042-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 684.Chen H, et al. Targeting Nestin hepatic stellate cells ameliorates liver fibrosis by facilitating TβRI degradation. J. Hepatol. 2021;74:1176–1187. doi: 10.1016/j.jhep.2020.11.016. [DOI] [PubMed] [Google Scholar]
- 685.Brandon-Warner E, et al. Adeno-associated virus serotype 2 vector-mediated reintroduction of microRNA-19b attenuates hepatic fibrosis. Hum. Gene Ther. 2018;29:674–686. doi: 10.1089/hum.2017.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 686.Monemian Esfahani A, et al. Tissue regeneration from mechanical stretching of cell-cell adhesion. Tissue Eng. Part C Methods. 2019;25:631–640. doi: 10.1089/ten.tec.2019.0098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 687.Vining KH, Mooney DJ. Mechanical forces direct stem cell behaviour in development and regeneration. Nat. Rev. Mol. Cell Biol. 2017;18:728–742. doi: 10.1038/nrm.2017.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 688.Lammerding J, Kamm RD, Lee RT. Mechanotransduction in cardiac myocytes. Ann. NY Acad. Sci. 2004;1015:53–70. doi: 10.1196/annals.1302.005. [DOI] [PubMed] [Google Scholar]
- 689.Park JS, et al. Differential effects of equiaxial and uniaxial strain on mesenchymal stem cells. Biotechnol. Bioeng. 2004;88:359–368. doi: 10.1002/bit.20250. [DOI] [PubMed] [Google Scholar]
- 690.Zanotelli MR, Zhang J, Reinhart-King CA. Mechanoresponsive metabolism in cancer cell migration and metastasis. Cell Metab. 2021;33:1307–1321. doi: 10.1016/j.cmet.2021.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 691.Zhang J, et al. Energetic regulation of coordinated leader-follower dynamics during collective invasion of breast cancer cells. Proc. Natl Acad. Sci. USA. 2019;116:7867–7872. doi: 10.1073/pnas.1809964116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 692.Doyle AD, Wang FW, Matsumoto K, Yamada KM. One-dimensional topography underlies three-dimensional fibrillar cell migration. J. Cell Biol. 2009;184:481–490. doi: 10.1083/jcb.200810041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 693.Fraley SI, et al. A distinctive role for focal adhesion proteins in three-dimensional cell motility. Nat. Cell Biol. 2010;12:598–604. doi: 10.1038/ncb2062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 694.Colasurdo M, Nieves EB, Fernández-Yagüe MA, Franck C, García AJ. Adhesive peptide and polymer density modulate 3D cell traction forces within synthetic hydrogels. Biomaterials. 2022;288:121710. doi: 10.1016/j.biomaterials.2022.121710. [DOI] [PubMed] [Google Scholar]