Abstract
Type 2 diabetes is a chronic and progressive cardiometabolic disorder that affects more than 10% of adults worldwide and is a major cause of morbidity, mortality, disability, and high costs. Over the past decade, the pattern of management of diabetes has shifted from a predominantly glucose centric approach, focused on lowering levels of haemoglobin A1c (HbA1c), to a directed complications centric approach, aimed at preventing short term and long term complications of diabetes, and a pathogenesis centric approach, which looks at the underlying metabolic dysfunction of excess adiposity that both causes and complicates the management of diabetes. In this review, we discuss the latest advances in patient centred care for type 2 diabetes, focusing on drug and non-drug approaches to reducing the risks of complications of diabetes in adults. We also discuss the effects of social determinants of health on the management of diabetes, particularly as they affect the treatment of hyperglycaemia in type 2 diabetes.
Keywords: Diabetes mellitus, Ambulatory care, Endocrinology, Internal medicine, Metabolic diseases, Primary health care
Introduction
Diabetes, a chronic and progressive cardiometabolic disorder, is a major cause of morbidity, disability, and mortality worldwide. Comprehensive person centred management of diabetes requires attention to glycaemic control and risk factors for cardiovascular disease (hyperlipidaemia, hypertension, and tobacco use), weight management, early detection and treatment of microvascular, macrovascular, and metabolic complications of diabetes and mental health concerns, mitigation of burden of treatment, addressing social determinants of health, and improving quality of life.1 The past decade has seen multiple developments in each aspect of the management of diabetes. This review focuses specifically on recent advances in the management of hyperglycaemia in diabetes, including drug and non-drug treatments. People with diabetes, caregivers, clinicians, health systems, payers, and policy makers need to appreciate the complexity and cost associated with optimal care of diabetes to meaningfully improve the health and well being of people living with diabetes.
Epidemiology
The current prevalence of diabetes among adults is 10.5% worldwide (536.6 million adults), with marked variation across regions and countries, and is estimated to reach 12.2% (783.2 million adults) by 2045.2 Diabetes is more prevalent in high income (11.1%) and middle income (10.8%) countries than in low income countries (5.5%). The prevalence of diabetes is rising everywhere, most rapidly in middle income countries where the prevalence is expected to reach 13.1% by 2045,2 probably because of changing diet and lifestyle factors, rising rates of obesity, inadequate resources for early diagnosis and prevention, and potentially greater genetic or epigenetic susceptibility arising from inadequate fetal and childhood nutrition. Data for low and middle income countries are likely to be underestimated because of barriers to screening and timely diagnosis.
More than 90% of people with diabetes have type 2 diabetes,3 characterised by insulin resistance and progressive beta cell failure, and commonly associated with other cardiometabolic disorders, including obesity, hypertension, cardiovascular disease, and hepatic steatosis. Diabetes contributed to 6.7 million deaths in 2021 alone,4 highlighting the urgency of preventing diabetes and optimising its management to improve health outcomes and quality of life for all people at risk of or with the disease.1
Sources and selection criteria
We searched PubMed for articles published in English. We prioritised randomised controlled trials, clinical guidelines, consensus statements, and systematic reviews. Search terms were: ((type 2 diabetes mellitus AND management (medical subject headings (MeSH) terms)) AND (type 2 diabetes mellitus (MeSH terms))) AND (care management, patient (MeSH terms)). Filters applied were: clinical trial, guideline, meta-analysis, practice guideline, randomised controlled trial, and systematic review, from 1 January 2013 to 1 January 2023. The reference lists of these articles were screened for relevant publications.
Goals and targets of management of type 2 diabetes
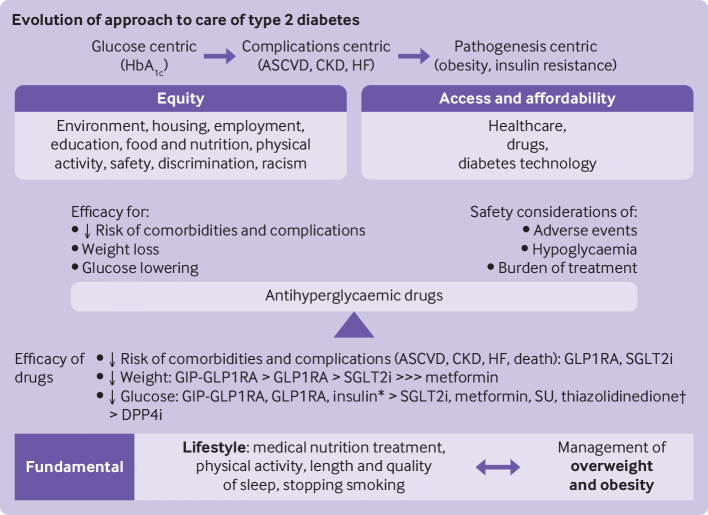
The primary objectives of the management of diabetes are to reduce the incidence and burden of complications and to improve quality of life (figure 1). Historically, these objectives were pursued through control of hyperglycaemia. In this glucose centric approach, clinical practice guidelines recommend targeting haemoglobin A1c (HbA1c) concentrations at <7% (53 mmol/mol) or <6.5% (47.5 mmol/mol) and, more recently, continuous glucose monitoring time in range >70% for most non-pregnant adults with type 2 diabetes, with lower or higher glycaemic thresholds individualised for each person.5–7 These recommendations for levels of HbA1c come from data from randomised controlled trials showing a reduction in microvascular complications with more intensive glycaemic control,8–12 although data for the association between time in range and risk of complications of chronic diabetes are limited but emerging.13 Implementation of glycaemic targets based on continuous glucose monitoring has also been limited by gaps in insurance coverage and accessibility, although continuous glucose monitoring is increasingly recommended for and used by people with type 2 diabetes.14 15
Figure 1.
Shifting pattern of management of type 2 diabetes. HbA1c=haemoglobin A1c; ASCVD=atherosclerotic cardiovascular disease; CKD=chronic kidney disease; HF=heart failure; GIP=glucose dependent insulinotropic polypeptide; GLP1RA=glucagon-like peptide 1 receptor agonist; SGLT2i=sodium glucose cotransporter 2 inhibitor; SU=sulfonylurea; DPP4i=dipeptidyl peptidase 4 inhibitor. *Insulin is preferred for acute management of severe hyperglycaemia; †Thiazolidinediones improve insulin resistance
Randomised controlled trials of older antihyperglycaemic treatments, such as sulfonylureas and insulins, however, have not shown a consistent association between intensive glycaemic control and reduction in macrovascular complications or mortality.16 Nevertheless, longer term follow-up of intensively treated adults provides some evidence of a lower risk of macrovascular events and cardiovascular death.8 17 Conversely, intensive glycaemic control in individuals with frailty, advanced age, and multimorbidity was associated with an increased risk of severe hypoglycaemia and death.18–20 Therefore, future research is needed to examine the effect of intensive glycaemic control when achieved with newer glucose lowering drugs, which have a lower risk of hypoglycaemia and additional cardio-reno-metabolic benefits. Taken together, these data highlight the importance of individualised glycaemic management and the need to shift the emphasis away from the imperfect surrogate of levels of HbA1c towards reducing hard outcomes of the adverse health effects of diabetes, while lessening the burden of treatment.21 22
Over the past decade, multiple randomised controlled trials have shown a reduction in cardiovascular disease, kidney disease, heart failure, and mortality with the use of glucagon-like peptide 1 receptor agonists (GLP1RAs) and sodium glucose cotransporter 2 inhibitors (SGLT2is), independent of a reduction in levels of HbA1c.23 These findings signalled a new complications centric era of the management of diabetes, focused directly on preventing or reducing macrovascular, microvascular, and other emerging complications of diabetes, such as heart failure. Many,6 24 25 although not all,26 clinical practice guidelines recommend treatment with GLP1RAs or SGLT2is, or both, for patients with cardiovascular or kidney disease, or both, or with risk factors for atherosclerotic cardiovascular disease, independent of glycaemic control, although all continue to stress the concurrent importance of achieving HbA1c targets.
More recently, the pattern of management of diabetes has begun to shift further, with a renewed focus on looking at the causes of type 2 diabetes and its metabolic comorbidities and long term complications. This pathogenesis centric approach places the management of obesity at the centre of the prevention and treatment of the disease.27 Even a relatively small amount (5-7%) of weight loss reduced the risk of incident diabetes and improved glycaemic control in people with type 2 diabetes.28–31 Greater amounts of weight loss have been reported to have greater beneficial effects on glycaemic control (including remission of diabetes), metabolic dysfunction, and quality of life.29 30 32–37 Weight loss achieved with metabolic surgery reduced the risks of microvascular and macrovascular complications of diabetes and reduced mortality.38–41
By contrast, intensive lifestyle treatments in the Look AHEAD (Action for Health in Diabetes) randomised controlled trial of 5145 adults with type 2 diabetes and overweight/obesity did not reduce the risk of cardiovascular events compared to usual care.30 The likelihood of detecting differences between the intensive lifestyle and conventional treatment groups might have been reduced because the cardiovascular event rate in the Look AHEAD population was much lower than anticipated (0.7% per year v estimated 3.1% per year).42 A post hoc analysis suggested that those who lost at least 10% of their body weight in the first year had a significantly lower risk of the primary outcome, which was a composite of the first occurrence of death from cardiovascular causes, non-fatal acute myocardial infarction, stroke, and hospital admission for angina (adjusted hazard ratio 0.79, 95% confidence interval 0.64 to 0.98, P=0.034).35 How weight loss achieved with drug treatment, particularly agents such as semaglutide and tirzepatide, compares with metabolic surgery for glycaemic control, microvascular and macrovascular complications, and mortality, should be examined.
Lifestyle treatments: medical nutrition treatment, physical activity, and sleep
Successful management of type 2 diabetes must include consistent attention to behaviours that sustain a healthy lifestyle and are foundational for achieving glycaemic control, preventing complications, supporting quality of life, and preserving optimal health. Medical nutrition treatment for diabetes emphasises a balanced selection of nutrient dense foods while minimising or eliminating added sugar, refined grains, and highly processed foods.6 7 43 Recommendations for optimal carbohydrate intake and composition vary, with the strongest evidence supporting an overall reduction in intake of carbohydrates. This principle can be applied to multiple dietary patterns, including a Mediterranean diet high in monounsaturated and polyunsaturated fats, low carbohydrate, vegetarian, or a plant based diets, and the Dietary Approaches to Stop Hypertension diet, with a focus on non-starchy vegetables, fruits, and legumes, and some dairy in those who are lactose tolerant.6 43 Only the Mediterranean diet has been shown to reduce cardiovascular disease and mortality.44 Also, evidence indicates the beneficial effects of involvement of community health workers to support education in self-management of diabetes and overall care, especially in rural or underserved communities, or both.45 Because hypertension and cardiovascular disease are major causes of mortality in individuals with diabetes, more attention needs to be paid to overall sodium intake and limiting the content of saturated fat and trans fat in the diet.6
Stopping smoking and abstinence from tobacco products is also imperative for cardiovascular health in adults with diabetes, and robust evidence supports the benefit of stopping smoking despite the potential for weight gain.6 46 Although nicotine replacement products and electronic cigarettes might facilitate stopping smoking, nicotine itself can impair glucose tolerance and adversely affect the cardiovascular system through increased sympathetic activation.47
Baseline levels of physical activity should be assessed to set reasonable and realistic behaviour oriented goals. Increasing the duration of physical activity and reducing sedentary time have been reported to improve cardiorespiratory fitness and HbA1c levels.48 Recommendations can be made to increase leisure time physical activity (walking, taking the stairs, and household chores), decrease sedentary time, and introduce physical activity on most days.6 49 Physical activities include both aerobic and resistance training, as well as flexibility and balance training.50
The length and quality of sleep are increasingly recognised as essential components of the management of diabetes and individuals should be screened for sleep related disorders.6 43 Referral for diagnosis and treatment of obstructive sleep apnoea and other sleep disorders should be considered if indicated. Screening for psychosocial factors and social determinants of health that might affect an individual's diabetes care and quality of life should also be performed, with engagement of or referral to relevant clinical team members for further evaluation and care, as appropriate.43
Lifestyle interventions in individuals with obesity or who are overweight are most successful when efforts are intensive and frequent follow-up is available, either in person or virtually.6 49 Weight loss can be achieved in various ways, and is most effective when strategies are combined: caloric restriction, increased caloric expenditure, elimination or substitution of drugs that promote weight gain, use of weight reducing drugs and, in select individuals, metabolic or bariatric surgery. One dietary strategy that has received considerable attention in recent years is time restricted eating,51 although data in adults with type 2 diabetes are limited to one randomised controlled trial52 53 and a larger trial is ongoing (n=344; Using Early Time Restricted Feeding and Timed Light Therapy to Improve Glycemic Control in Adults With Type 2 Diabetes, NCT04155619). Weight management is discussed in more detail below.
Drug treatment of type 2 diabetes
Initial management of type 2 diabetes has traditionally included metformin in most adults because of its glucose lowering effect, neutral effects on weight, minimal risk of hypoglycaemia, safety profile, low cost, and ease of administration. Now, in the light of evidence from trials of cardiovascular and kidney outcomes, decisions on treatment of diabetes with drugs should be made based on cardiac comorbidities (established atherosclerotic cardiovascular disease and heart failure), risk factors for atherosclerotic cardiovascular disease and kidney disease, engaging adults in shared decision making, and prioritising the use of drugs shown to reduce the risk of cardiovascular or kidney adverse outcomes, or both, in adults with specific comorbidities.7 24–26
Adults with atherosclerotic cardiovascular disease or indicators of high risk
In people with established atherosclerotic cardiovascular disease or risk factors for atherosclerotic cardiovascular disease, a GLP1RA or SGLT2i with known cardiovascular benefit should be started, regardless of levels of HbA1c or background glucose lowering treatments.24 Drugs that have been shown to cause significant reductions in major adverse cardiovascular events in cardiovascular outcomes trials compared with placebo include the GLP1RAs dulaglutide (hazard ratio 0.88, 95% confidence interval 0.79 to 0.99), liraglutide (0.87, 0.78 to 0.97), and subcutaneous semaglutide (0.74, 0.58 to 0.95), and the SGLT2is canagliflozin (0.86, 0.75 to 0.97) and empagliflozin (0.85, 0.75 to 0.97).54–58 None of the trials of cardiovascular outcomes involved head-to-head comparisons of GLP1RAs versus SGLT2is.59
Individual components of the composite major adverse cardiovascular events outcome as well as secondary outcomes in the cardiovascular outcomes trials vary between GLP1RAs and SGLT2is. A reduction in stroke was seen in meta-analyses of randomised controlled trials of GLP1RAs compared with placebo (hazard ratio 0.83, 95% confidence interval 0.76 to 0.92) but not with SGLT2is compared with placebo (0.95, 0.85 to 1.05).59 The mechanisms and benefits of GLP1RAs and SGLT2is seem to be complementary, and evidence is emerging to support combination treatment, which might provide more benefit than each used alone.60–62 Currently, guidelines from the American Diabetes Association/European Association for the Study of Diabetes recommend the addition of the alternative class when more glucose lowering is needed.24 25
Adults with heart failure
GLP1RAs have not shown benefit for heart failure outcomes in individual randomised controlled trials of cardiovascular outcomes,55 56 58 although meta-analyses of these studies suggested a potential benefit.59 63 64 SGLT2is, by contrast, have consistently shown significant benefit for heart failure outcomes.54 57 65 Also, dapagliflozin and empagliflozin were beneficial in people with reduced or preserved ejection fraction without type 2 diabetes, and have an indication for improving heart failure outcomes.66–69 Accordingly, in people with heart failure, an SGLT2i with known benefit should be started to reduce the risk of major adverse cardiovascular events and worsening heart failure.24 26
Adults with chronic kidney disease
GLP1RAs have shown benefit for secondary kidney related outcomes in large individual randomised controlled trials55 56 70 and meta-analyses59 63 64 of cardiovascular outcomes, but dedicated kidney outcome trials are ongoing.71 Several SGLT2is, including canagliflozin, dapagliflozin, and empagliflozin, have shown benefit in adults with chronic kidney disease with or without type 2 diabetes and in dedicated kidney outcome trials, and have an indication for improving chronic kidney disease outcomes.24 72 Therefore, SGLT2is with primary evidence are preferred for individuals with an estimated glomerular filtration rate <60 mL/min/1.73 m2 or albuminuria, or both, to reduce the progression of chronic kidney disease. If SGLT2is are not tolerated or cannot be used, GLP1RAs with demonstrated renal benefit are a reasonable alternative.24 26 73 Current prescribing information allows SGLT2is to be started in adults with an estimated glomerular filtration rate of ≥20 mL/min/1.73 m2 for kidney benefit, although the glucose lowering effects are substantially reduced at an estimated glomerular filtration rate <45 mL/min/1.73 m2.74 A small reduction in the estimated glomerular filtration rate can be seen after starting treatment with SGLT2is because of reversal or correction of the previous hyperfiltration state in adults with diabetes, but it does not predict further reductions in estimated glomerular filtration rate or require discontinuation of treatment.
Role of metformin
Although metformin was a commonly used background drug in most large trials of cardiovascular and kidney outcomes,75 several post hoc analyses have demonstrated benefit with GLP1RAs or SGLT2is regardless of background use of metformin.76–82 Current guidelines from the American Diabetes Association/European Association for the Study of Diabetes and the American Association of Clinical Endocrinology no longer recommend metformin as the preferred first line agent for all individuals with type 2 diabetes, and instead suggest consideration of cardiac and kidney comorbidities when selecting first line treatment.6 24 25 Cost is a major consideration in selecting the most appropriate treatment, however, probably contributing to differences in these recommendations from guidelines used in other countries. In the US, insurers have not caught up with the guidelines, and require that metformin is used before other agents. Guidance from the National Institute for Health and Care Excellence (NICE) still recommends metformin as the first line treatment for people with cardiac or kidney comorbidities, or both, with introduction of an SGLT2i in people who cannot tolerate metformin or need intensification of treatment.26 Despite robust outcome data, GLP1RAs are not recommended ny NICE until failure of triple oral drug treatment and only in people with a high body mass index or in whom insulin treatment cannot be used.26 Insurance formulary restrictions on prescribing GLP1RAs and SGLT2is, including the requirement of step treatment starting with metformin, still persist but should be reconsidered to better align with scientific evidence.
Other situations when a drug other than metformin can be considered as first line treatment include severe or symptomatic hyperglycaemia (HbA1c>10%, ketosis, or weight loss), creatinine clearance or estimated glomerular filtration rate <30 mL/min/1.73 m2, or when the person cannot tolerate metformin despite slow up titration of the dose or a trial of the extended release formulation, or both. Sulfonylureas and thiazolidinediones are now less commonly recommended because of their adverse effect profiles. Sulfonylureas can lead to weight gain and are associated with a high risk of hypoglycaemia, and thiazolidinediones can also cause weight gain, as well as fluid retention and osteoporosis. People treated with thiazolidinediones must be monitored for the development of heart failure; thiazolidinediones are not recommended for those with symptoms of heart failure and are contraindicated in class 3 or 4 heart failure. Because generic forms of sulfonylureas and thiazolidinediones are available, however, these drug classes are options when cost is a barrier to accessing other agents or the individual's clinical situation requires these drugs. Pioglitazone, a thiazolidinedione, has beneficial effects in hepatic steatosis and stroke, and can be considered in these contexts.6 83
Effect on weight and weight related comorbidities
Clinicians should also consider the effect of the glucose lowering regimen on weight and weight related comorbidities, including overweight or obesity and non-alcoholic fatty liver disease or non-alcoholic steatohepatitis. Weight loss is greatest with the dual glucose dependent insulinotropic polypeptide (GIP)-GLP1RA, tirzepatide, and subcutaneous semaglutide, followed by dulaglutide and liraglutide.27 Moderate weight loss is seen with the other GLP1RAs and SGLT2is. Drugs with neutral effects on weight include the dipeptidyl peptidase 4 inhibitors (DPP4is) and metformin, whereas the sulfonylureas, thiazolidinediones, and insulin all increase the risk of weight gain (table 1).24 27 84 Recent single centre and population based cross sectional studies in the US estimated that >70% of people with type 2 diabetes have non-alcoholic fatty liver disease and more than half of those with type 2 diabetes and non-alcoholic fatty liver disease have steatohepatitis.85–88 Insulin resistance, impaired lipid and glucose metabolism, and altered insulin secretion play a part in non-alcoholic fatty liver disease and progression of type 2 diabetes, and might indicate why the two diseases are so closely linked.89 Although limited evidence exists so far, current guidelines recommend the use of a GLP1RA or pioglitazone for the treatment of diabetes in people with non-alcoholic steatohepatitis.90 91 Weight management, which is essential for the treatment of hepatic steatosis, is discussed below.
Table 1.
Choosing drug class for type 2 diabetes
| Drug class | Glucose efficacy | Effect on weight | Risk of hypoglycaemia | Route of administration | Cost | Ideal candidates for use |
| Metformin | High | Neutral | No | Oral | Low |
|
| SGLT2i | Intermediate to high | Loss (intermediate) | No | Oral | High |
|
| GLP1RA | High | Loss (intermediate to very high) | No | Subcutaneous (oral semaglutide) | High |
|
| GIP-GLP1RA | High | Loss (very high) |
No | Subcutaneous | High |
|
| DPP4i | Intermediate | Neutral | No | Oral | High |
|
| Sulfonylurea | High | Gain | Yes | Oral | Low |
|
| Thiazolidinedione | High | Gain | No | Oral | Low |
|
GIP=glucose dependent insulinotropic polypeptide; GLP1RA=glucagon-like peptide 1 receptor agonist; SGLT2i=sodium-glucose cotransporter 2 inhibitor; DPP4i=dipeptidyl peptidase 4 inhibitor.
Glucose lowering efficacy
In addition to choosing a drug that targets cardiovascular, kidney, and metabolic outcomes, clinicians should also develop a treatment approach that has sufficient efficacy to achieve glycaemic targets.24 Although some guidelines (most notably, the Australian Diabetes Society) cite a lack of evidence to support substantial differences in glucose lowering between antihyperglycaemic drug classes when used as monotherapy,92 prior meta-analyses, including a meta-analysis of 453 trials assessing nine drug classes, and the recently completed Glycemia Reduction Approaches in Type 2 Diabetes: A Comparative Effectiveness (GRADE) pragmatic randomised clinical trial comparing insulin glargine U-100, the sulfonylurea glimepiride, the GLP1RA liraglutide, and the DPP4i sitagliptin in 5047 individuals with moderately uncontrolled type 2 diabetes found insulin and GLP1RA to be significantly more effective at lowering HbA1c than the other examined drugs.93 94 The American Diabetes Association Standards of Care there categorise drug classes as having very high, high, or intermediate glucose lowering efficacy (table 1).24 The greatest reductions in levels of HbA1c are seen with the dual GIP-GLP1RAs, GLP1RAs, and insulin. In the GLP1RA class, subcutaneous semaglutide and dulaglutide had the highest efficacy for glucose lowering. The recently approved dual GIP-GLP1RA, tirzepatide, seems to have the greatest efficacy for reducing levels of glucose. SGLT2is and DPP4is have less robust HbA1c lowering effects and are classified as intermediate to high (SGLT2is) and intermediate (DPP4is).24 25 93
GRADE, a large scale, comparative effectiveness study of four drugs in combination with metformin, found that insulin glargine and liraglutide achieved and maintained HbA1c targets more effectively than glimepiride and sitagliptin. The study did not, however, include newer agents, such as the SGLT2is, or once weekly GLP1RAs.95 The GRADE study also highlighted the challenges of maintaining glucose targets over time, with 71% of study participants progressing to HbA1c ≥7% within four years, regardless of the treatment option.94 A meta-analysis of 229 randomised controlled trials comprising 121 914 participants suggested that glucose lowering efficacy was highest with GLP1RA and weakest with DPP4i, with other agents in between.96 By contrast, a meta-analysis of 140 randomised trials and 26 observational studies showed that each new class of non-insulin drugs added to metformin monotherapy lowers levels of HbA1c by about 0.7-1%.95 A shift towards earlier use of combination treatment, in contrast with a stepwise approach, to reach glucose targets and provide better glycaemic durability has been reported.24 97 For people with marked hyperglycaemia (eg, HbA1c >10% or with symptoms), clinicians should start insulin, or a combination of insulin with GLP1RAs.98 When improved glycaemic control is achieved, many people with type 2 diabetes can be safely transitioned to non-insulin treatments with close monitoring to prevent hypoglycemia and hyperglycemia.
Safety considerations
Other considerations in the selection of treatment for diabetes are the risks of hypoglycaemia, other adverse effects and safety considerations, as well as cost and administration requirements that often result in barriers to adherence. Therefore, individuals with diabetes, care partners, and clinicians need to engage in shared decision making to identify treatment strategies that are aligned with the individual's goals of care, treatment preferences, the clinical and psychosocial context, and risks and benefits associated with each treatment option. Tables 1 and 2 summarise this information. Some key and controversial safety considerations are discussed below.
Table 2.
Safety and mitigation considerations for each drug class for type 2 diabetes
| Drug class | Safety considerations | Strategies for successful initiation and use | Key education points |
| Metformin | Common adverse effects are gastrointestinal (diarrhoea, stomach upset), metallic taste, vitamin B12 deficiency (with long term use) Use caution:
Do not use:
|
|
|
| SGLT2i | Common adverse effects are genital mycotic infections and urinary tract infections (less common) Use caution:
|
|
|
| GLP1RA | Common adverse effects are gastrointestinal (nausea, vomiting, diarrhoea, constipation) Use caution:
Do not use:
|
|
|
| GIP-GLP1RA | Same as GLP1RA | Same as GLP1RA | Same as GLP1RA |
| DPP4i | Very well tolerated; few common adverse effects Use caution:
|
|
|
| Sulfonylurea | Common adverse effects are weight gain and hypoglycaemia |
|
|
| Thiazolidinedione | Common adverse effects are oedema and weight gain Use caution:
|
|
|
GIP=glucose-dependent insulinotropic polypeptide; GLP1RA=glucagon-like peptide 1 receptor agonist; SGLT2i=sodium-glucose cotransporter 2 inhibitor; DPP4i=dipeptidyl peptidase 4 inhibitor.
Acute pancreatitis
Acute pancreatitis has been reported in individuals who received GLP1RAs, DPP4is, and the GIP-GLP1RA, tirzepatide. After early post-marketing reports, the US Food and Drug Administration warned of a potential link between acute pancreatitis and GLP1RAs and DPP4is.99 Multiple preclinical, observational, and randomised controlled studies were inconsistent, with some showing positive associations and others showing no association.100 Ultimately, the FDA concluded that a causal relation could not be established and insufficient evidence existed to modify treatment. Systematic reviews and meta-analyses of randomised controlled trials (eg, long term cardiovascular outcomes trials) concluded that treatment with GLP1RAs or DPP4is was not associated with an increased risk of pancreatitis or pancreatic cancer.101–103
Nonetheless, current prescribing information, FDA guidance, and treatment guidelines recommend cautious use of these drug classes in people with a history of pancreatitis, in part because these people were excluded from most trials.24 If these drug classes are used, individuals should be monitored for signs and symptoms of pancreatitis and, if pancreatitis develops, treatment should be discontinued and not restarted.24 99 We also suggest caution in starting these drugs in people with a previous history of pancreatitis, particularly when the cause of pancreatitis is unknown or persists. Monitoring of lipase levels in randomised controlled trials showed asymptomatic fluctuations in both groups (intervention and placebo). Hence no evidence exists to suggest ongoing monitoring during treatment.
Gallbladder or biliary disease
GLP1RAs, DPP4is, and GIP-GLP1RAs are also associated with an increased risk of gallbladder and biliary disease, including cholelithiasis and cholecystitis.104–107 Although the absolute risk of biliary or gllbladder disease with GLP1RA therapy seems to be small, with a recent meta-analysis of 76 randomised controlled trials involving 103 371 103 371 participants reporting an additional 27 incidences per 10 000 patients per year,104 this finding might under-represent the true risk, because many studies did not report biliary related events. The risk seems to be higher with higher doses of drugs, longer duration of use, and when used for weight loss rather than glycaemic control. We therefore advise caution with the use of GLP1RAs, DPP4is, and GIP-GLP1RAs in people at high risk of biliary complications.
Diabetic retinopathy
A significant increase in retinopathy complications (3% v 1.8%, P=0.02), including vitreous haemorrhage, blindness, or need for photocoagulation treatment or an intravitreal agent, was seen in people receiving semaglutide during the SUSTAIN-6 (Trial to Evaluate Cardiovascular and Other Long term Outcomes With Semaglutide in Subjects With Type 2 Diabetes) randomised controlled trial with 3297 participants with type 2 diabetes.56 Of those with retinopathy complications, 83.5% had a history of retinopathy at baseline. In a meta-analysis of four cardiovascular outcomes trials of dulaglutide, liraglutide, oral semaglutide, and subcutaneous semaglutide, use of GLP1RAs was associated with an increased risk of rapidly worsening retinopathy (odds ratio 1.23, 95% confidence interval 1.05 to 1.44).108 In another meta-analysis, GLP1RAs were not independently associated with an increased risk of retinopathy, but an association between retinopathy and the magnitude of the reduction in levels of HbA1c was found.109 Rapid glucose lowering has previously been associated with worsening diabetic retinopathy,110 and the GLP1RA cardiovascular outcomes trials were not powered to detect differences in retinopathy complications. Thus whether worsening retinopathy is caused by the drug itself, a change or rate of change in glucose levels, or a combination of both is unclear. We advise caution when GLP1RAs are used, particularly semaglutide, in people with diabetic retinopathy, and individuals should be monitored closely for progression of retinopathy.111
Whether other GLP1RAs similarly increase the risk of progressive diabetic retinopathy is not known. Consultation with an ophthalmologist should be considered before starting GLP1RAs in people with pre-existing retinopathy.111 A large randomised controlled trial (A Research Study to Look at How Semaglutide Compared to Placebo Affects Diabetic Eye Disease in People With Type 2 Diabetes (FOCUS), NCT03811561) evaluating the long term effects of subcutaneous semaglutide on eye disease in 1500 people with type 2 diabetes is ongoing and should provide more evidence.
Amputations
An increased risk of lower limb amputations was first reported in the cardiovascular outcomes trial for canagliflozin that included 10 142 participants with type 2 diabetes and high cardiovascular risk (6.3 v 3.4 participants per 1000 patient years; hazard ratio 1.97, 95% confidence interval 1.41 to 2.75)57 and led to a warning added to the prescribing information for canagliflozin in 2017.112 The FDA removed the warning in 2020 based on more clinical trial data that found that the risk was less than previously described.113 Subsequent real world cohort studies, randomised controlled studies, and meta-analyses have reported conflicting results, with some suggesting an increased risk with all SGLT2is and others finding no increased risk.114–121 Therefore, reasonable steps to take are to consider factors that increase the risk of amputations before starting an SGLT2i, closely monitor people for lower limb ulcers or infections, and discontinue the SGLT2i if these occur. Subgroup and exploratory analyses of the SGLT2i cardiovascular outcomes trials, however, suggest cardiovascular benefit in patients with peripheral arterial disease,122–124 so clinicians should use shared decision making when assessing the benefits and risks of SGLT2is in those at high risk.
Diabetic ketoacidosis
SGLT2is are associated with an increased risk of diabetic ketoacidosis, particularly in people with type 1 diabetes and in the perioperative population.125 126 Rates in adults with type 2 diabetes are low and range from 0.16 to 0.76 events per 1000 patient years.127 In type 2 diabetes, the risk is increased in people who are insulin deficient, in older people, with prolonged use of SGLT2is, or in those with a combination of these factors.128 Guidance on risk management of diabetic ketoacidosis is mainly from individuals with type 1 diabetes, with little guidance specific to type 2 diabetes, and recommendations are mostly extrapolated from the type 1 diabetes context.126 129 People with diabetes should be informed of the importance of adherence to insulin treatment, avoiding very low carbohydrate diets (such as ketotic diets), and excessive intake of alcohol. Education on management of sick days should also be given, and insulin doses should be monitored carefully; basal insulin should not be discontinued completely during illness or planned activity, particularly in those receiving intensive insulin treatment.
Clinicians and people with diabetes should be aware of predisposing factors and the clinical presentation of diabetic ketoacidosis, which often occurs with lower serum glucose levels (so-called euglycaemic diabetic ketoacidosis), sometimes at glucose concentrations of ≤200 mg/dL (11.1 mmol/L). The SGLT2i should be discontinued and treatment started promptly if diabetic ketoacidosis is suspected. SGLT2is should also be discontinued 3-4 days before scheduled surgery, during prolonged fasting or low carbohydrate intake, or during critical illness to lessen the risk of diabetic ketoacidosis.24 Some have suggested that absence of ketosis (<0.6 mmol/L blood ketones, negative urine ketones) should be confirmed in people with type 1 diabetes before the start of treatment if SGLT2is are being used off label in this population,126 but no evidence exists in support of this practice for people with type 2 diabetes.
Starting and titrating insulin treatment
Many people with type 2 diabetes will eventually require insulin because of the progressive nature of the disease. For most people, a GLP1RA should be considered as the first injectable agent before basal insulin, based on the strong evidence of similar efficacy, beneficial effect on weight, and less hypoglycaemia.130 131 If more treatment is needed after a GLP1RA, basal insulin should be started first and titrated to a maximum effective dose in a safe and timely way.7 98 Several steps are necessary to support optimisation of insulin treatment, including clear communication of expectations, adequacy of glucose monitoring (including continuous glucose monitoring for people with basal insulin or intensive insulin treatment, and remote telemonitoring), a feasible dose titration plan, clearly defined glycaemic targets, and education on proper administration of insulin and storage.131–134 Whether the individual can self-titrate the dose or if more support is needed should be assessed. People who can self-titrate can be instructed to continue uptitrating the dose until fasting glucose levels are consistently between 80 and 130 mg/dL (4.4 to 7.2 mmol/L; or an individualised glycaemic target), an anticipated maximum basal dose is reached (eg, 0.5 units/kg/day), or have unexplained hypoglycaemia. Providing these endpoints is key to reducing the risk of being treated with an inappropriately high dose of basal insulin in an attempt to compensate for inadequate post-prandial glycaemic control (ie, overbasalisation) while facilitating continued titration to an effective dose.135 If the individual cannot self-titrate, consider providing weekly follow-up healthcare remotely (ie, telehealth) for timely dose titrations.
If the basal insulin dose has been sufficiently titrated but levels of HbA1c remain above the person's individualised target or concern for overbasalisation exists, targeting postprandial glucose excursions is warranted. Initially, consider adding a GLP1RA or GIP-GLPRA if not already being used. The next step is to add prandial insulin as a separate injection or by switching to a fixed ratio combination. Basal bolus insulin treatment requires more injections, more glucose testing, more education, and carries a higher risk of hypoglycaemia and weight gain.98 Metformin or complication centric drugs (GLP1RAs and SGLT2is), or both, should be continued. Sulfonylureas should be discontinued because of the risk of hypoglycaemia with concurrent insulin treatment.
Weight management in type 2 diabetes
Among adults with diabetes in the US, almost 28% are overweight (body mass index 25.0-29.9), 46% have obesity (body mass index 30.0-39.9), and 16% have severe obesity (body mass index ≥40.0).136 Increasingly recognised as a chronic disease, obesity (termed adiposity based chronic disease)137 138 is characterised by excessive, maldistributed, and dysfunctional adipose tissue, and is associated with increased risks of hyperglycaemia (ie, prediabetes and type 2 diabetes), cardiovascular disease, hyperlipidaemia, hypertension, chronic kidney disease, cancer, urinary incontinence, non-alcoholic fatty liver disease, osteoarthritis, infertility, obstructive sleep apnoea, and gastro-oesophageal reflux disease.137
Obesity is closely related to the pathogenesis and pathophysiology of type 2 diabetes and it also affects the management and outcomes of diabetes.137 139 Strong evidence indicates that weight loss, particularly if >10% of body weight, can prevent, improve, and even reverse type 2 diabetes.140 The Diabetes Prevention Programme showed that people with prediabetes who were randomised to receive an intensive lifestyle intervention had a 16% reduction in the risk of progressing from prediabetes to diabetes for every kilogram of weight loss.37 In the Look AHEAD study of people with type 2 diabetes and overweight or obesity, improvement in fasting glucose and HbA1c levels was found with weight loss as little as ≥2 kg, and improvements were directly proportional to the amount of weight lost.31 After initial weight loss from lifestyle interventions or pharmacotherapy, compensatory physiological responses often make efforts at further weight loss more difficult, less successful, or difficult to maintain, a biological phenomenon referred to as obesity protecting obesity.141 Hence clinicians should provide a supportive approach, recognising personal biases, and avoiding stigma and judgment to facilitate weight management efforts.141
Despite years of commercial availability, obesity drugs are rarely used, with fewer than 5-10% of people with diabetes and obesity receiving obesity drugs in the US.142 This finding could be driven by the relatively low efficacy of historically available drugs for weight loss, with most drugs causing <7% body weight loss.141 Recent developments with incretin treatments have closed this gap, however, with up to 20% weight loss reported with tirzepatide.107 Several studies in people with obesity, with or without type 2 diabetes, treated with semaglutide or tirzepatide have reported reductions in body weight of at least 5-10% in up to 80-90% of people, and reductions of 15-20% in up to 40-50% of people.106 107 143 144 Efforts to lose weight in people with type 2 diabetes and obesity should be supported through preferential use of glucose lowering drugs that are associated with weight loss, avoiding glucose lowering and non-diabetes drugs associated with weight gain, and aiming for weight loss of 12-15% as appropriate, to achieve maximum benefits.7 140
Equity and affordability of diabetes care
Affordability, accessibility, and feasibility of implementing the diabetes care plan are major considerations in shared decision making. In the US, the high and rising costs of insulin and non-insulin drugs145 have contributed to diabetes distress,146 cost related non-adherence147 148 with a detrimental effect on diabetes health outcomes149 and rationing of other vital expenses.150 Therefore, healthcare providers must discuss concerns about affordability with all people with diabetes, ensure that prescribed drugs are available and accessible, and leverage care team and community support systems to reduce the financial burden of the management of diabetes.151 152
To deal with the growing concerns about affordability of insulin in the US, out-of-pocket costs have been capped in 2023 by the Centers for Medicare and Medicaid Services (which oversee publicly funded insurance for seniors, low income individuals, and people with disabilities or end-stage kidney disease), several private insurance plans, and insulin manufacturers, and the effect of these changes on cost related non-adherence and rationing will need to be assessed. The cost of drugs is generally much lower outside of the US because of highly regulated policies on drug pricing and cost effectiveness in other high income countries,153 154 but 80% of people with diabetes live in low and middle income countries155 and half do not have access to recommended diabetes treatments.156 These findings call for multifaceted policy solutions to lower costs, increase supply, and improve accessibility of evidence based diabetes treatments and technologies in all settings and populations.157
Socioeconomic barriers to optimal management of diabetes are multifaceted and include not only the high costs of diabetes drugs, technology, and equipment, but also foundational social determinants of health, such as the home environment with access to healthy food choices and space for physical activity, environmental pollution and endocrine disrupting chemicals, stable housing with access to electricity and refrigeration, employment type and stability, and educational attainment.152 Geographical differences in the quality of care and prevalence of type 2 diabetes and its complications exist across levels of rurality,158 159 neighbourhood disadvantage,158 160 and geopolitical environment.161–163 Several interventions have been shown to be successful in improving the management of diabetes, including community health worker programmes, diabetes prevention and self-management programmes adapted specifically to the needs of underserved and disadvantaged populations, expansion of health insurance as part of the Affordable Care Act, food and housing support programmes, and others.152
We must also be cognisant of pervasive racial and ethnic inequalities in the quality of diabetes care and health outcomes. In the US, racial and ethnic minority populations are disproportionately affected by diabetes164 and its complications.152 165 166 Multiple studies have shown worse glycaemic control165 167 168 and higher rates of acute complications (hypoglycaemia,160 165 169–172 diabetic ketoacidosis, and hyperglycaemic hyperosmolar state),160 165 170 173 chronic complications (kidney disease,165 174–178 amputation,165 175 cardiovascular disease,165 175 and retinopathy),176 179 and mortality180 181 among black people with type 2 diabetes relative to other racial and ethnic groups. People with type 2 diabetes from racial and ethnic minority groups are also substantially less likely to be treated with GLP1RAs and SGLT2is than non-Hispanic white people.182 183 Similar inequalities in the prevalence, management, and health outcomes of diabetes have been described in Europe184 185 and around the world.186 187 These inequalities highlight the need for structural solutions and multisector collaborations that deal with the barriers to optimal diabetes management and health at all levels to ensure that all people, regardless of race, ethnic group, socioeconomic status, or place of residence, receive high quality care.
Conclusions
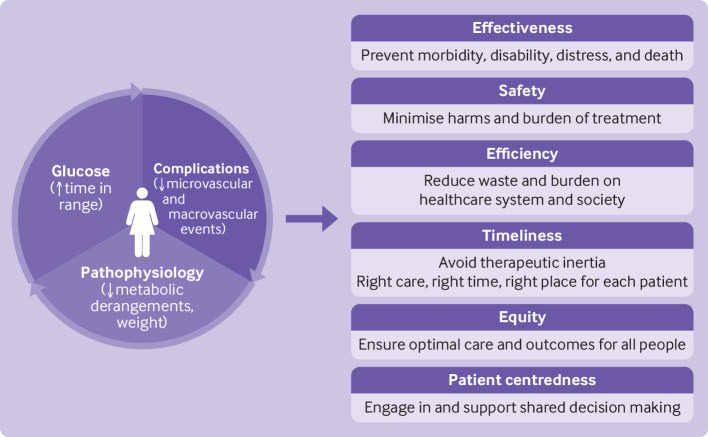
The paradigm of diabetes management has shifted over the past decade from a predominantly glucose-centric approach to approaches that prioritise prevention of diabetes complications and addressing the underlying causes of diabetes and metabolic dysfunction, such as obesity (figure 2). High quality, evidence based management of diabetes therefore requires reducing glucose levels to a safe, patient centred range; using glucose lowering drugs with a strong evidence base for reduction of diabetes complications and excess adiposity, not just lowering levels of HbA1c; minimising burden of treatment and improving quality of life; and implementing care delivery models that support high quality (effective, efficient, safe, equitable, timely, and person centred) care.188 Access and affordability remain major barriers, as is the sustainable implementation of effective lifestyle interventions.
Figure 2.
Person centred goals of treatment of type 2 diabetes
Questions for further research.
What are the short term and long term health outcomes associated with combined GLP1RA and SGLT2i treatment?
What is the optimal weight loss target (>10% or 15%) in the management of type 2 diabetes?
What is the comparative effectiveness and safety of drug treatments for obesity compared with metabolic surgery for long term metabolic, microvascular, and macrovascular complications?
How can effective lifestyle treatments for long term weight loss be implemented effectively, sustainably, and equitably?
What are effective and sustainable ways to engage people with diabetes, care partners, and communities in the prevention and management of diabetes to ensure equitable access to care?
How can structural barriers to optimal metabolic health be removed?
Patient involvement.
Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Footnotes
Twitter: @RozalinaMD
Contributors: All authors designed the study, drafted sections of the manuscript, and reviewed or edited the manuscript. RGM secured funding and supervised the study. RGM is the guarantor of this work and, as such, had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. RGM affirms that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
Funding: The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health (NIH), grant Nos K23DK114497 (to RGM) and K23DK123384 (to RJG). The funder had no role in considering the study design or in the collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication.
Competing interests: We have read and understood the BMJ policy on declaration of interests and declare the following interests: in the past 36 months, RGM received support from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), National Institute on Aging of the National Institutes of Health (NIH), the Patient Centered Outcomes Research Institute (PCORI), the National Center for Advancing Translational Sciences, and AARP. RGM also served as a consultant to Emmi (Wolters Kluwer) on developing patient education materials related to prediabetes and diabetes. In the past 36 months, RJG has received unrestricted research support (to Emory University) from Novo Nordisk, Dexcom, and Eli Lilly, and consulting or personal fees from Sanofi, Eli Lilly, Novo Nordisk, Boehringer-Ingelheim, Bayer, Pfizer, and Weight Watchers, all outside the scope of this work.
Provenance and peer review: Commissioned; externally peer reviewed.
References
- 1.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 4. Comprehensive medical evaluation and assessment of comorbidities. Diabetes Care 2022;46:S49–67. 10.2337/dc23-S004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sun H, Saeedi P, Karuranga S, et al. IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 2022;183:109119. 10.1016/j.diabres.2021.109119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 2. Classification and diagnosis of diabetes. Diabetes Care 2023;46:S19–40. 10.2337/dc23-S002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.International Diabetes Federation . IDF diabetes Atlas. diabetes around the world in 2021. International Diabetes Federation; 2021. Available: https://diabetesatlas.org/atlas/tenth-edition/ [Accessed 15 Aug 2023]. [Google Scholar]
- 5.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 6. Glycemic targets. Diabetes Care 2023;46:S97–110. 10.2337/dc23-S006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Blonde L, Umpierrez GE, Reddy SS, et al. American Association of Clinical Endocrinology clinical practice guideline: developing a diabetes mellitus comprehensive care plan-2022 update. Endocr Pract 2022;28:923–1049. 10.1016/j.eprac.2022.08.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Samson SL, Vellanki P, Blonde L, et al. American Association of Clinical Endocrinology consensus statement: comprehensive type 2 diabetes management algorithm - 2023 update. Endocr Pract 2023;29:305–40. 10.1016/j.eprac.2023.02.001 [DOI] [PubMed] [Google Scholar]
- 8.Holman RR, Paul SK, Bethel MA, et al. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008;359:1577–89. 10.1056/NEJMoa0806470 [DOI] [PubMed] [Google Scholar]
- 9.UKPDS . Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) group. Lancet 1998;352:837–53. 10.1016/S0140-6736(98)07019-6 [DOI] [PubMed] [Google Scholar]
- 10.UKPDS . Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) group. Lancet 1998;352:854–65. 10.1016/S0140-6736(98)07037-8 [DOI] [PubMed] [Google Scholar]
- 11.Duckworth W, Abraira C, Moritz T, et al. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009;360:129–39. 10.1056/NEJMoa0808431 [DOI] [PubMed] [Google Scholar]
- 12.ADVANCE Collaborative Group, Patel A, MacMahon S, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008;358:2560–72. 10.1056/NEJMoa0802987 [DOI] [PubMed] [Google Scholar]
- 13.Raj R, Mishra R, Jha N, et al. Time in range, as measured by continuous glucose monitor, as a predictor of microvascular complications in type 2 diabetes: a systematic review. BMJ Open Diab Res Care 2022;10:e002573. 10.1136/bmjdrc-2021-002573 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Galindo RJ, Parkin CG, Aleppo G, et al. What’s wrong with this picture? A critical review of current centers for Medicare and Medicaid services coverage criteria for continuous glucose monitoring. Diabetes Technol Ther 2021;23:652–60. 10.1089/dia.2021.0107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.ElSayed NA, Aleppo G, Aroda VR, et al. 7. Diabetes technology: standards of care in diabetes—2023. Diabetes Care 2023;46:S111–27. 10.2337/dc23-S007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rodríguez-Gutiérrez R, Montori VM. Glycemic control for patients with type 2 diabetes mellitus: our evolving faith in the face of evidence. Circ Cardiovasc Qual Outcomes 2016;9:504–12. 10.1161/CIRCOUTCOMES.116.002901 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hayward RA, Reaven PD, Emanuele NV, et al. Follow-up of glycemic control and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2015;373:978. 10.1056/NEJMc1508386 [DOI] [PubMed] [Google Scholar]
- 18.Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008;358:2545–59. 10.1056/NEJMoa0802743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Duckworth WC, Abraira C, Moritz TE, et al. The duration of diabetes affects the response to intensive glucose control in type 2 subjects: the VA diabetes trial. J Diabetes Complications 2011;25:355–61. 10.1016/j.jdiacomp.2011.10.003 [DOI] [PubMed] [Google Scholar]
- 20.McCoy RG, Lipska KJ, Yao X, et al. Intensive treatment and severe hypoglycemia among adults with type 2 diabetes. JAMA Intern Med 2016;176:969–78. 10.1001/jamainternmed.2016.2275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rodriguez-Gutierrez R, McCoy RG. Measuring what matters in diabetes. JAMA 2019;321:1865–6. 10.1001/jama.2019.4310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rodríguez-Gutiérrez R, Millan-Alanis JM, Barrera FJ, et al. Value of patient-centered glycemic control in patients with type 2 diabetes. Curr Diab Rep 2021;21:63. 10.1007/s11892-021-01433-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cefalu WT, Kaul S, Gerstein HC, et al. Cardiovascular outcomes trials in type 2 diabetes: where do we go from here? Reflections from a diabetes care editors’ expert forum. Diabetes Care 2018;41:14–31. 10.2337/dci17-0057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 9. Pharmacologic approaches to glycemic treatment. Diabetes Care 2023;46:S140–57. 10.2337/dc23-S009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Davies MJ, Aroda VR, Collins BS, et al. Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2022;65:1925–66. 10.1007/s00125-022-05787-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.NICE . Type 2 Diabetes in Adults: Management (NG28). NICE Guideline, 2022. Available: https://www.nice.org.uk/guidance/ng28 [accessed 15 Aug 2023]. [Google Scholar]
- 27.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 8. Obesity and weight management for the prevention and treatment of type 2 diabetes. Diabetes Care 2023;46:S128–39. 10.2337/dc23-S008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393–403. 10.1056/NEJMoa012512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Look AHEAD Research Group . Eight-year weight losses with an intensive lifestyle intervention: the look AHEAD study. Obesity (Silver Spring) 2014;22:5–13. 10.1002/oby.20662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Look AHEAD Research Group, Wing RR, Bolin P, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med 2013;369:145–54. 10.1056/NEJMoa1212914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wing RR, Lang W, Wadden TA, et al. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 2011;34:1481–6. 10.2337/dc10-2415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lean ME, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (direct): an open-label, cluster-randomised trial. Lancet 2018;391:541–51. 10.1016/S0140-6736(17)33102-1 [DOI] [PubMed] [Google Scholar]
- 33.Schauer PR, Bhatt DL, Kashyap SR. Bariatric surgery or intensive medical therapy for diabetes after 5 years. N Engl J Med 2017;376:1997. 10.1056/NEJMc1703377 [DOI] [PubMed] [Google Scholar]
- 34.Ikramuddin S, Korner J, Lee W-J, et al. Durability of addition of Roux-en-Y gastric bypass to lifestyle intervention and medical management in achieving primary treatment goals for uncontrolled type 2 diabetes in mild to moderate obesity: a randomized control trial. Diabetes Care 2016;39:1510–8. 10.2337/dc15-2481 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Look AHEAD Research Group, Gregg E, Jakicic J, et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol 2016;4:913–21. 10.1016/S2213-8587(16)30162-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Booth H, Khan O, Prevost T, et al. Incidence of type 2 diabetes after bariatric surgery: population-based matched cohort study. Lancet Diabetes Endocrinol 2014;2:963–8. 10.1016/S2213-8587(14)70214-1 [DOI] [PubMed] [Google Scholar]
- 37.Hamman RF, Wing RR, Edelstein SL, et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006;29:2102–7. 10.2337/dc06-0560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Adams TD, Davidson LE, Litwin SE, et al. Health benefits of gastric bypass surgery after 6 years. JAMA 2012;308:1122–31. 10.1001/2012.jama.11164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sjöström L, Peltonen M, Jacobson P, et al. Association of bariatric surgery with long-term remission of type 2 diabetes and with microvascular and macrovascular complications. JAMA 2014;311:2297–304. 10.1001/jama.2014.5988 [DOI] [PubMed] [Google Scholar]
- 40.Sjöström L, Lindroos A-K, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med 2004;351:2683–93. 10.1056/NEJMoa035622 [DOI] [PubMed] [Google Scholar]
- 41.O’Brien R, Johnson E, Haneuse S, et al. Microvascular outcomes in patients with diabetes after bariatric surgery versus usual care: a matched cohort study. Ann Intern Med 2018;169:300–10. 10.7326/M17-2383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pi-Sunyer X. The look AHEAD trial: a review and discussion of its outcomes. Curr Nutr Rep 2014;3:387–91. 10.1007/s13668-014-0099-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 5. Facilitating positive health behaviors and well-being to improve health outcomes. Diabetes Care 2023;46:S68–96. 10.2337/dc23-S005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Martínez-González MA, Ros E, Estruch R. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med 2018;379:1388–9. 10.1056/NEJMc1809971 [DOI] [PubMed] [Google Scholar]
- 45.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 1. Improving care and promoting health in populations. Diabetes Care 2023;46:S10–8. 10.2337/dc23-S001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kos K. Cardiometabolic morbidity and mortality with smoking cessation, review of recommendations for people with diabetes and obesity. Curr Diab Rep 2020;20:82. 10.1007/s11892-020-01352-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eliasson B, Taskinen M-R, Smith U. Long-term use of nicotine gum is associated with hyperinsulinemia and insulin resistance. Circulation 1996;94:878–81. 10.1161/01.CIR.94.5.878 [DOI] [PubMed] [Google Scholar]
- 48.Balducci S, Haxhi J, Sacchetti M, et al. Relationships of changes in physical activity and sedentary behavior with changes in physical fitness and cardiometabolic risk profile in individuals with type 2 diabetes. Diabetes Care 2022;45:213–21. 10.2337/dc21-1505 [DOI] [PubMed] [Google Scholar]
- 49.ElSayed NA, Aleppo G, Aroda VR, et al. 3. Prevention or delay of type 2 diabetes and associated comorbidities: standards of care in diabetes-2023. Diabetes Care 2023;46:S41–8. 10.2337/dc23-ad08a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kanaley JA, Colberg SR, Corcoran MH, et al. Exercise/physical activity in individuals with type 2 diabetes: a consensus statement from the American College of Sports Medicine. Med Sci Sports Exerc 2022;54:353–68. 10.1249/MSS.0000000000002800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Manoogian ENC, Chow LS, Taub PR, et al. Time-restricted eating for the prevention and management of metabolic diseases. Endocr Rev 2022;43:405–36. 10.1210/endrev/bnab027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Obermayer A, Tripolt NJ, Pferschy PN, et al. Efficacy and safety of intermittent fasting in people with insulin-treated type 2 diabetes (INTERFAST-2)-A randomized controlled trial. Diabetes Care 2023;46:463–8. 10.2337/dc22-1622 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Parr EB, Devlin BL, Lim KHC, et al. Time-restricted eating as a nutrition strategy for individuals with type 2 diabetes: a feasibility study. Nutrients 2020;12:3228. 10.3390/nu12113228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med 2015;373:2117–28. 10.1056/NEJMoa1504720 [DOI] [PubMed] [Google Scholar]
- 55.Marso SP, Bain SC, Consoli A, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med 2016;375:1834–44. 10.1056/NEJMoa1607141 [DOI] [PubMed] [Google Scholar]
- 56.Marso SP, Bain SC, Consoli A, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med 2016;375:1834–44. 10.1056/NEJMoa1607141 [DOI] [PubMed] [Google Scholar]
- 57.Neal B, Perkovic V, Matthews DR. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med 2017;377:2099. 10.1056/NEJMc1712572 [DOI] [PubMed] [Google Scholar]
- 58.Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 2019;394:121–30. 10.1016/S0140-6736(19)31149-3 [DOI] [PubMed] [Google Scholar]
- 59.Giugliano D, Scappaticcio L, Longo M, et al. GLP-1 receptor agonists vs. SGLT-2 inhibitors: the gap seems to be leveling off. Cardiovasc Diabetol 2021;20:205. 10.1186/s12933-021-01400-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li C, Luo J, Jiang M, et al. The efficacy and safety of the combination therapy with GLP-1 receptor agonists and SGLT-2 inhibitors in type 2 diabetes mellitus: a systematic review and meta-analysis. Front Pharmacol 2022;13. 10.3389/fphar.2022.838277 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lam CSP, Ramasundarahettige C, Branch KRH, et al. Efpeglenatide and clinical outcomes with and without concomitant sodium-glucose cotransporter-2 inhibition use in type 2 diabetes: exploratory analysis of the AMPLITUDE-O trial. Circulation 2022;145:565–74. 10.1161/CIRCULATIONAHA.121.057934 [DOI] [PubMed] [Google Scholar]
- 62.Dave CV, Kim SC, Goldfine AB, et al. Risk of cardiovascular outcomes in patients with type 2 diabetes after addition of SGLT2 inhibitors versus sulfonylureas to baseline GLP-1Ra therapy. Circulation 2021;143:770–9. 10.1161/CIRCULATIONAHA.120.047965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Giugliano D, Scappaticcio L, Longo M, et al. GLP-1 receptor agonists and cardiorenal outcomes in type 2 diabetes: an updated meta-analysis of eight CVOTs. Cardiovasc Diabetol 2021;20:189. 10.1186/s12933-021-01366-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sattar N, Lee MMY, Kristensen SL, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol 2021;9:653–62. 10.1016/S2213-8587(21)00203-5 [DOI] [PubMed] [Google Scholar]
- 65.Wiviott SD, Raz I, Sabatine MS. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. reply. N Engl J Med 2019;380:1881–2. 10.1056/NEJMc1902837 [DOI] [PubMed] [Google Scholar]
- 66.McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med 2019;381:1995–2008. 10.1056/NEJMoa1911303 [DOI] [PubMed] [Google Scholar]
- 67.Anker SD, Butler J, Filippatos G, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med 2021;385:1451–61. 10.1056/NEJMoa2107038 [DOI] [PubMed] [Google Scholar]
- 68.Packer M, Anker SD, Butler J, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med 2020;383:1413–24. 10.1056/NEJMoa2022190 [DOI] [PubMed] [Google Scholar]
- 69.Solomon SD, McMurray JJV, Claggett B, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med 2022;387:1089–98. 10.1056/NEJMoa2206286 [DOI] [PubMed] [Google Scholar]
- 70.Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet 2019;394:131–8. 10.1016/S0140-6736(19)31150-X [DOI] [PubMed] [Google Scholar]
- 71.Rossing P, Baeres FMM, Bakris G, et al. The rationale, design and baseline data of FLOW, a kidney outcomes trial with once-weekly semaglutide in people with type 2 diabetes and chronic kidney disease. Nephrol Dial Transplant 2023. 10.1093/ndt/gfad009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.ElSayed NA, Aleppo G, Aroda VR, et al. Standards of care in diabetes—2023. Chapter 11. Chronic kidney disease and risk management. Diabetes Care 2023;46:S191–202. 10.2337/dc23-S011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rossing P, Caramori ML, Chan JCN, et al. Clinical practice guideline for diabetes management in chronic kidney disease. Kidney Int 2022;102:S1–127. 10.1016/j.kint.2022.06.008 [DOI] [PubMed] [Google Scholar]
- 74.Heerspink HJL, Cherney DZI. Clinical implications of an acute dip in eGFR after SGLT2 inhibitor initiation. Clin J Am Soc Nephrol 2021;16:1278–80. 10.2215/CJN.02480221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Harrington JL, de Albuquerque Rocha N, Patel KV, et al. Should metformin remain first-line medical therapy for patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease. Curr Diab Rep 2018;18:64. 10.1007/s11892-018-1035-z [DOI] [PubMed] [Google Scholar]
- 76.Inzucchi SE, Fitchett D, Jurišić-Eržen D, et al. Are the cardiovascular and kidney benefits of empagliflozin influenced by baseline glucose-lowering therapy? Diabetes Obes Metab 2020;22:631–9. 10.1111/dom.13938 [DOI] [PubMed] [Google Scholar]
- 77.Ferrannini G, Gerstein H, Colhoun HM, et al. Similar cardiovascular outcomes in patients with diabetes and established or high risk for coronary vascular disease treated with dulaglutide with and without baseline metformin. Eur Heart J 2021;42:2565–73. 10.1093/eurheartj/ehaa777 [DOI] [PubMed] [Google Scholar]
- 78.Crowley MJ, McGuire DK, Alexopoulos A-S, et al. Effects of liraglutide on cardiovascular outcomes in type 2 diabetes patients with and without baseline metformin use: post hoc analyses of the LEADER trial. Diabetes Care 2020;43:e108–10. 10.2337/dc20-0437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Neuen BL, Arnott C, Perkovic V, et al. Sodium-glucose co-transporter-2 inhibitors with and without metformin: a meta-analysis of cardiovascular, kidney and mortality outcomes. Diabetes Obes Metab 2021;23:382–90. 10.1111/dom.14226 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Masson W, Lavalle-Cobo A, Lobo M, et al. Novel antidiabetic drugs and risk of cardiovascular events in patients without baseline metformin use: a meta-analysis. Eur J Prev Cardiol 2021;28:69–75. 10.1093/eurjpc/zwaa074 [DOI] [PubMed] [Google Scholar]
- 81.Husain M, Consoli A, De Remigis A, et al. Semaglutide reduces cardiovascular events regardless of metformin use: a post hoc subgroup analysis of SUSTAIN 6 and PIONEER 6. Cardiovasc Diabetol 2022;21:64. 10.1186/s12933-022-01489-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Baker C, Retzik-Stahr C, Singh V, et al. Should metformin remain the first-line therapy for treatment of type 2 diabetes? Ther Adv Endocrinol Metab 2021;12:2042018820980225. 10.1177/2042018820980225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cusi K, Orsak B, Bril F, et al. Long-term pioglitazone treatment for patients with NASH and prediabetes or type 2 diabetes mellitus: a randomized controlled trial. Ann Intern Med 2016;165:305–15. 10.7326/M15-1774 [DOI] [PubMed] [Google Scholar]
- 84.Tsapas A, Karagiannis T, Kakotrichi P, et al. Comparative efficacy of glucose-lowering medications on body weight and blood pressure in patients with type 2 diabetes: a systematic review and network meta-analysis. Diabetes Obes Metab 2021;23:2116–24. 10.1111/dom.14451 [DOI] [PubMed] [Google Scholar]
- 85.Lomonaco R, Godinez Leiva E, Bril F, et al. Advanced liver fibrosis is common in patients with type 2 diabetes followed in the outpatient setting: the need for systematic screening. Diabetes Care 2021;44:399–406. 10.2337/dc20-1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Ciardullo S, Monti T, Perseghin G. High prevalence of advanced liver fibrosis assessed by transient elastography among U.S. adults with type 2 diabetes. Diabetes Care 2021;44:519–25. 10.2337/dc20-1778 [DOI] [PubMed] [Google Scholar]
- 87.Barb D, Repetto EM, Stokes ME, et al. Type 2 diabetes mellitus increases the risk of hepatic fibrosis in individuals with obesity and nonalcoholic fatty liver disease. Obesity (Silver Spring) 2021;29:1950–60. 10.1002/oby.23263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Harrison SA, Gawrieh S, Roberts K, et al. Prospective evaluation of the prevalence of non-alcoholic fatty liver disease and steatohepatitis in a large middle-aged US cohort. J Hepatol 2021;75:284–91. 10.1016/j.jhep.2021.02.034 [DOI] [PubMed] [Google Scholar]
- 89.Saponaro C, Gaggini M, Gastaldelli A. Nonalcoholic fatty liver disease and type 2 diabetes: common pathophysiologic mechanisms. Curr Diab Rep 2015;15:607. 10.1007/s11892-015-0607-4 [DOI] [PubMed] [Google Scholar]
- 90.Cusi K, Isaacs S, Barb D, et al. American Association of Clinical Endocrinology clinical practice guideline for the diagnosis and management of nonalcoholic fatty liver disease in primary care and endocrinology clinical settings: co-sponsored by the American Association for the Study of Liver Diseases (AASLD). Endocr Pract 2022;28:528–62. 10.1016/j.eprac.2022.03.010 [DOI] [PubMed] [Google Scholar]
- 91.Kanwal F, Shubrook JH, Adams LA, et al. Clinical care pathway for the risk stratification and management of patients with nonalcoholic fatty liver disease. Gastroenterology 2021;161:1657–69. 10.1053/j.gastro.2021.07.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Living evidence guidelines in diabetes. Australian evidence-based clinical guidelines for diabetes Australian Diabetes Society; 2020. Available: https://www.diabetessociety.com.au/living-evidence-guidelines-in-diabetes/ [Google Scholar]
- 93.Tsapas A, Avgerinos I, Karagiannis T, et al. Comparative effectiveness of glucose-lowering drugs for type 2 diabetes: a systematic review and network meta-analysis. Ann Intern Med 2020;173:278–86. 10.7326/M20-0864 [DOI] [PubMed] [Google Scholar]
- 94.GRADE Study Research Group, Nathan DM, Lachin JM, et al. Glycemia reduction in type 2 diabetes - glycemic outcomes. N Engl J Med 2022;387:1063–74. 10.1056/NEJMoa2200433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Bennett WL, Maruthur NM, Singh S, et al. Comparative effectiveness and safety of medications for type 2 diabetes: an update including new drugs and 2-drug combinations. Ann Intern Med 2011;154:602–13. 10.7326/0003-4819-154-9-201105030-00336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Maloney A, Rosenstock J, Fonseca V. A model-based meta-analysis of 24 antihyperglycemic drugs for type 2 diabetes: comparison of treatment effects at therapeutic doses. Clin Pharmacol Ther 2019;105:1213–23. 10.1002/cpt.1307 [DOI] [PubMed] [Google Scholar]
- 97.Matthews DR, Paldánius PM, Proot P, et al. Glycaemic durability of an early combination therapy with vildagliptin and metformin versus sequential metformin monotherapy in newly diagnosed type 2 diabetes (VERIFY): a 5-year, multicentre, randomised, double-blind trial. Lancet 2019;394:1519–29. 10.1016/S0140-6736(19)32131-2 [DOI] [PubMed] [Google Scholar]
- 98.Galindo RJ, Moazzami B, Scioscia MF, et al. A randomized controlled trial comparing the efficacy and safety of ideglira versus basal-bolus in patients with poorly controlled type 2 diabetes and very high Hba1C >/=9-15%: DUAL HIGH trial. Diabetes Care 2023:dc222426. 10.2337/dc22-2426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.FDA . FDA Drug Safety Communication: FDA investigating reports of possible increased risk of pancreatitis and pre-cancerous findings of the pancreas from incretin mimetic drugs for type 2 diabetes. Silver Spring, MD: U.S. Food and Drug Administration, 2013. [Google Scholar]
- 100.Egan AG, Blind E, Dunder K, et al. Pancreatic safety of incretin-based drugs — FDA and EMA assessment. N Engl J Med 2014;370:794–7. 10.1056/NEJMp1314078 [DOI] [PubMed] [Google Scholar]
- 101.Monami M, Nreu B, Scatena A, et al. Safety issues with glucagon-like peptide-1 receptor agonists (pancreatitis, pancreatic cancer and cholelithiasis): data from randomized controlled trials. Diabetes Obes Metab 2017;19:1233–41. 10.1111/dom.12926 [DOI] [PubMed] [Google Scholar]
- 102.Storgaard H, Cold F, Gluud LL, et al. Glucagon-like peptide-1 receptor agonists and risk of acute pancreatitis in patients with type 2 diabetes. Diabetes Obes Metab 2017;19:906–8. 10.1111/dom.12885 [DOI] [PubMed] [Google Scholar]
- 103.Dicembrini I, Montereggi C, Nreu B, et al. Pancreatitis and pancreatic cancer in patientes treated with dipeptidyl peptidase-4 inhibitors: an extensive and updated meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2020;159:107981. 10.1016/j.diabres.2019.107981 [DOI] [PubMed] [Google Scholar]
- 104.He L, Wang J, Ping F, et al. Association of glucagon-like peptide-1 receptor agonist use with risk of gallbladder and biliary diseases: a systematic review and meta-analysis of randomized clinical trials. JAMA Intern Med 2022;182:513–9. 10.1001/jamainternmed.2022.0338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.He L, Wang J, Ping F, et al. Dipeptidyl peptidase-4 inhibitors and gallbladder or biliary disease in type 2 diabetes: systematic review and pairwise and network meta-analysis of randomised controlled trials. BMJ 2022;377:e068882. 10.1136/bmj-2021-068882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Aronne LJ, Bramblette S, Huett-Garcia A, et al. Weight and health - pathophysiology and therapies. N Engl J Med 2022;387:e62. 10.1056/NEJMp2214423 [DOI] [PubMed] [Google Scholar]
- 107.Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med 2021;385:503–15. 10.1056/NEJMoa2107519 [DOI] [PubMed] [Google Scholar]
- 108.Yoshida Y, Joshi P, Barri S, et al. Progression of retinopathy with glucagon-like peptide-1 receptor agonists with cardiovascular benefits in type 2 diabetes - A systematic review and meta-analysis. J Diabetes Complications 2022;36:108255. 10.1016/j.jdiacomp.2022.108255 [DOI] [PubMed] [Google Scholar]
- 109.Bethel MA, Diaz R, Castellana N, et al. HbA1c change and diabetic retinopathy during GLP-1 receptor agonist cardiovascular outcome trials: a meta-analysis and meta-regression. Diabetes Care 2021;44:290–6. 10.2337/dc20-1815 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Jingi AM, Tankeu AT, Ateba NA, et al. Mechanism of worsening diabetic retinopathy with rapid lowering of blood glucose: the synergistic hypothesis. BMC Endocr Disord 2017;17:63. 10.1186/s12902-017-0213-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Albert SG, Wood EM, Ahir V. Glucagon-like peptide 1-receptor agonists and A1C: good for the heart but less so for the eyes. Diabetes Metab Syndr 2023;17:102696. 10.1016/j.dsx.2022.102696 [DOI] [PubMed] [Google Scholar]
- 112.FDA . FDA Drug Safety Communication: FDA confirms increased risk of leg and foot amputations with the diabetes medicine canagliflozin (Invokana, Invokamet, Invokamet XR). Silver Spring, MD: U.S. Food and Drug Administration, Department of Health and Human Services, 2017. [Google Scholar]
- 113.FDA . FDA removes Boxed Warning about risk of leg and foot amputations for the diabetes medicine canagliflozin (Invokana, Invokamet, Invokamet XR). Silver Spring, MD: U.S. Food and Drug Administration, 2020. [Google Scholar]
- 114.Scheen AJ. Lower-limb amputations in patients treated with SGLT2 inhibitors versus DPP-4 inhibitors: a meta-analysis of observational studies. Diabetes Epidemiol Manage 2022;6:100054. 10.1016/j.deman.2022.100054 [DOI] [Google Scholar]
- 115.See RM, Teo YN, Teo YH, et al. Effects of sodium-glucose cotransporter 2 on amputation events: a systematic review and meta-analysis of randomized-controlled trials. Pharmacology 2022;107:123–30. 10.1159/000520903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Scheen AJ. Lower limb amputations: protection with GLP-1 receptor agonists rather than increased risk with SGLT2 inhibitors? Diabetes Metab 2022;48:101325. 10.1016/j.diabet.2022.101325 [DOI] [PubMed] [Google Scholar]
- 117.Lin C, Zhu X, Cai X, et al. SGLT2 inhibitors and lower limb complications: an updated meta-analysis. Cardiovasc Diabetol 2021;20:91. 10.1186/s12933-021-01276-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Dicembrini I, Tomberli B, Nreu B, et al. Peripheral artery disease and amputations with sodium-glucose co-transporter-2 (SGLT-2) inhibitors: a meta-analysis of randomized controlled trials. Diabetes Res Clin Pract 2019;153:138–44. 10.1016/j.diabres.2019.05.028 [DOI] [PubMed] [Google Scholar]
- 119.Ueda P, Svanström H, Melbye M, et al. Sodium glucose cotransporter 2 inhibitors and risk of serious adverse events: nationwide register based cohort study. BMJ 2018;363:k4365. 10.1136/bmj.k4365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Katsiki N, Dimitriadis G, Hahalis G, et al. Sodium-glucose co-transporter-2 inhibitors (SGLT2I) use and risk of amputation: an expert panel overview of the evidence. Metabolism 2019;96:92–100. 10.1016/j.metabol.2019.04.008 [DOI] [PubMed] [Google Scholar]
- 121.Paul SK, Bhatt DL, Montvida O. The association of amputations and peripheral artery disease in patients with type 2 diabetes mellitus receiving sodium-glucose cotransporter type-2 inhibitors: real-world study. Eur Heart J 2021;42:1728–38. 10.1093/eurheartj/ehaa956 [DOI] [PubMed] [Google Scholar]
- 122.Verma S, Mazer CD, Al-Omran M, et al. Cardiovascular outcomes and safety of empagliflozin in patients with type 2 diabetes mellitus and peripheral artery disease. Circulation 2018;137:405–7. 10.1161/CIRCULATIONAHA.117.032031 [DOI] [PubMed] [Google Scholar]
- 123.Bonaca MP, Wiviott SD, Zelniker TA, et al. Dapagliflozin and cardiac, kidney, and limb outcomes in patients with and without peripheral artery disease in DECLARE-TIMI 58. Circulation 2020;142:734–47. 10.1161/CIRCULATIONAHA.119.044775 [DOI] [PubMed] [Google Scholar]
- 124.Barraclough JY, Yu J, Figtree GA, et al. Cardiovascular and renal outcomes with canagliflozin in patients with peripheral arterial disease: data from the CANVAS program and CREDENCE trial. Diabetes Obes Metab 2022;24:1072–83. 10.1111/dom.14671 [DOI] [PubMed] [Google Scholar]
- 125.Auerbach JS, Gershengorn HB, Aljure OD, et al. Postcardiac surgery euglycemic diabetic ketoacidosis in patients on sodium-glucose cotransporter 2 inhibitors. J Cardiothorac Vasc Anesth 2023;37:956–63. 10.1053/j.jvca.2023.01.041 [DOI] [PubMed] [Google Scholar]
- 126.Danne T, Garg S, Peters AL, et al. International consensus on risk management of diabetic ketoacidosis in patients with type 1 diabetes treated with sodium-glucose cotransporter (SGLT) inhibitors. Diabetes Care 2019;42:1147–54. 10.2337/dc18-2316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Blau JE, Tella SH, Taylor SI, et al. Ketoacidosis associated with SGLT2 inhibitor treatment: analysis of FAERS data. Diabetes Metab Res Rev 2017;33. 10.1002/dmrr.2924 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Liu J, Li L, Li S, et al. Sodium-glucose co-transporter-2 inhibitors and the risk of diabetic ketoacidosis in patients with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Obes Metab 2020;22:1619–27. 10.1111/dom.14075 [DOI] [PubMed] [Google Scholar]
- 129.Garg SK, Peters AL, Buse JB, et al. Strategy for mitigating DKA risk in patients with type 1 diabetes on adjunctive treatment with SGLT inhibitors: a STICH protocol. Diabetes Technol Ther 2018;20:571–5. 10.1089/dia.2018.0246 [DOI] [PubMed] [Google Scholar]
- 130.Singh S, Wright EE, Kwan AYM, et al. Glucagon-like peptide-1 receptor agonists compared with basal insulins for the treatment of type 2 diabetes mellitus: a systematic review and meta-analysis. Diabetes Obes Metab 2017;19:228–38. 10.1111/dom.12805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Abd El Aziz MS, Kahle M, Meier JJ, et al. A meta-analysis comparing clinical effects of short- or long-acting GLP-1 receptor agonists versus insulin treatment from head-to-head studies in type 2 diabetic patients. Diabetes Obes Metab 2017;19:216–27. 10.1111/dom.12804 [DOI] [PubMed] [Google Scholar]
- 132.Kuritzky L, Reid TS, Wysham CH. Practical guidance on effective basal insulin titration for primary care providers. Clin Diabetes 2019;37:368–76. 10.2337/cd18-0091 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Garnero TL, Davis NJ, Perez-Nieves M, et al. Insulin non-persistence among people with type 2 diabetes: how to get your patients to stay on insulin therapy. Postgrad Med 2018;130:394–401. 10.1080/00325481.2018.1457396 [DOI] [PubMed] [Google Scholar]
- 134.Patel D, Triplitt C, Trujillo J. Appropriate titration of basal insulin in type 2 diabetes and the potential role of the pharmacist. Adv Ther 2019;36:1031–51. 10.1007/s12325-019-00907-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Johnson EL, Frias JP, Trujillo JM. Anticipatory guidance in type 2 diabetes to improve disease management; next steps after basal insulin. Postgrad Med 2018;130:365–74. 10.1080/00325481.2018.1452515 [DOI] [PubMed] [Google Scholar]
- 136.CDC . National Diabetes Statistics Report. Atlanta, Georgia, U.S.A: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, 2021. [Google Scholar]
- 137.Garvey WT, Mechanick JI, Brett EM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract 2016;22 Suppl 3:1–203. 10.4158/EP161365.GL [DOI] [PubMed] [Google Scholar]
- 138.Burki T. European Commission classifies obesity as a chronic disease. Lancet Diabetes Endocrinol 2021;9:418. 10.1016/S2213-8587(21)00145-5 [DOI] [PubMed] [Google Scholar]
- 139.Bae JP, Lage MJ, Mo D, et al. Obesity and glycemic control in patients with diabetes mellitus: analysis of physician electronic health records in the US from 2009-2011. J Diabetes Complications 2016;30:212–20. 10.1016/j.jdiacomp.2015.11.016 [DOI] [PubMed] [Google Scholar]
- 140.Ryan DH, Yockey SR. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15%, and over. Curr Obes Rep 2017;6:187–94. 10.1007/s13679-017-0262-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Alemán JO, Almandoz JP, Frias JP, et al. Obesity among Latinx people in the United States: a review. Obesity (Silver Spring) 2023;31:329–37. 10.1002/oby.23638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Thomas CE, Mauer EA, Shukla AP, et al. Low adoption of weight loss medications: a comparison of prescribing patterns of antiobesity pharmacotherapies and SGLT2S. Obesity 2016;24:1955–61. 10.1002/oby.21533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wilding JPH, Calanna S, Kushner RF. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med 2021;385. 10.1056/NEJMc2106918 [DOI] [PubMed] [Google Scholar]
- 144.Alkhezi OS, Alahmed AA, Alfayez OM, et al. Comparative effectiveness of glucagon-like peptide-1 receptor agonists for the management of obesity in adults without diabetes: a network meta-analysis of randomized clinical trials. Obes Rev 2023;24:e13543. 10.1111/obr.13543 [DOI] [PubMed] [Google Scholar]
- 145.Zhou X, Shrestha SS, Shao H, et al. Factors contributing to the rising national cost of glucose-lowering medicines for diabetes during 2005–2007 and 2015–2017. Diabetes Care 2020;43:2396–402. 10.2337/dc19-2273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Vitale RJ, Wentzell K, Laffel LMB. "Fear that one day I may not be able to afford insulin": the emotional burden of diabetes costs during emerging adulthood. Diabetes Technol Ther 2022;24:915–9. 10.1089/dia.2022.0170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Taha MB, Valero-Elizondo J, Yahya T, et al. Cost-related medication nonadherence in adults with diabetes in the United States: the national health interview survey 2013-2018. Diabetes Care 2022;45:594–603. 10.2337/dc21-1757 [DOI] [PubMed] [Google Scholar]
- 148.Caraballo C, Valero-Elizondo J, Khera R, et al. Burden and consequences of financial hardship from medical bills among nonelderly adults with diabetes mellitus in the United States. Circ Cardiovasc Qual Outcomes 2020;13:e006139. 10.1161/CIRCOUTCOMES.119.006139 [DOI] [PubMed] [Google Scholar]
- 149.Khunti K, Seidu S, Kunutsor S, et al. Association between adherence to pharmacotherapy and outcomes in type 2 diabetes: a meta-analysis. Diabetes Care 2017;40:1588–96. 10.2337/dc16-1925 [DOI] [PubMed] [Google Scholar]
- 150.Piette JD, Heisler M, Wagner TH. Problems paying out-of-pocket medication costs among older adults with diabetes. Diabetes Care 2004;27:384–91. 10.2337/diacare.27.2.384 [DOI] [PubMed] [Google Scholar]
- 151.Herges JR, Neumiller JJ, McCoy RG. Easing the financial burden of diabetes management: a guide for patients and primary care clinicians. Clin Diabetes 2021;39:427–36. 10.2337/cd21-0004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Hill-Briggs F, Adler NE, Berkowitz SA, et al. Social determinants of health and diabetes: a scientific review. Diabetes Care 2020;44:258–79. 10.2337/dci20-0053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Morgan SG, Good CB, Leopold C, et al. An analysis of expenditures on primary care prescription drugs in the United States versus ten comparable countries. Health Policy 2018;122:1012–7. 10.1016/j.healthpol.2018.07.005 [DOI] [PubMed] [Google Scholar]
- 154.Mulcahy AW, Schwam D, Edenfield N. Comparing insulin prices in the United States to other countries: results from a price index analysis. In: Comparing insulin prices in the United States to other countries: results from a price index analysis. Santa Monica, California, U.S.A: RAND Corporation, 2020. 10.7249/RRA788-1 [DOI] [Google Scholar]
- 155.Zhou B, Lu Y, Hajifathalian K, et al. Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4·4 million participants. Lancet 2016;387:1513–30. 10.1016/S0140-6736(16)00618-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Flood D, Seiglie JA, Dunn M, et al. The state of diabetes treatment coverage in 55 low-income and middle-income countries: a cross-sectional study of nationally representative, individual-level data in 680 102 adults. Lancet Healthy Longev 2021;2:e340–51. 10.1016/s2666-7568(21)00089-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.WHO . Keeping the 100-year-old promise: making insulin access universal. Geneva, Switzerland: World Health Organization, 2021. [Google Scholar]
- 158.Kurani SS, Lampman MA, Funni SA, et al. Association between area-level socioeconomic deprivation and diabetes care quality in US primary care practices. JAMA Netw Open 2021;4:e2138438. 10.1001/jamanetworkopen.2021.38438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Dugani SB, Mielke MM, Vella A. Burden and management of type 2 diabetes in rural United States. Diabetes Metab Res Rev 2021;37:e3410. 10.1002/dmrr.3410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Kurani SS, Heien HC, Sangaralingham LR, et al. Association of area-level socioeconomic deprivation with hypoglycemic and hyperglycemic crises in US adults with diabetes. JAMA Netw Open 2022;5:e2143597. 10.1001/jamanetworkopen.2021.43597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Loop MS, Howard G, de Los Campos G, et al. Heat maps of hypertension, diabetes mellitus, and smoking in the continental United States. Circ Cardiovasc Qual Outcomes 2017;10:e003350. 10.1161/CIRCOUTCOMES.116.003350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Callaghan T, Ferdinand AO, Akinlotan MA, et al. The changing landscape of diabetes mortality in the United States across region and rurality, 1999-2016. J Rural Health 2020;36:410–5. 10.1111/jrh.12354 [DOI] [PubMed] [Google Scholar]
- 163.Akinlotan MA, Primm K, Bolin JN, et al. Racial, rural, and regional disparities in diabetes-related lower-extremity amputation rates, 2009–2017. Diabetes Care 2021;44:2053–60. 10.2337/dc20-3135 [DOI] [PubMed] [Google Scholar]
- 164.Gregg EW, Zhuo X, Cheng YJ, et al. Trends in lifetime risk and years of life lost due to diabetes in the USA, 1985-2011: a modelling study. Lancet Diabetes Endocrinol 2014;2:867–74. 10.1016/S2213-8587(14)70161-5 [DOI] [PubMed] [Google Scholar]
- 165.CDC . Diabetes data & statistics. diabetes Atlas. Atlanta, GA: Division of Diabetes Translation, Centers for Disease Control and Prevention, U.S. Dept of Health and Human Services; 2022. Available: https://www.cdc.gov/diabetes/data/index.html [Accessed 15 Aug 2023]. [Google Scholar]
- 166.Golden SH, Joseph JJ, Hill-Briggs F. Casting a health equity lens on endocrinology and diabetes. J Clin Endocrinol Metab 2021;106:e1909–16. 10.1210/clinem/dgaa938 [DOI] [PubMed] [Google Scholar]
- 167.Kirk JK, D’Agostino RB, Bell RA, et al. Disparities in HbA1C levels between African-American and non-hispanic white adults with diabetes: a meta-analysis. Diabetes Care 2006;29:2130–6. 10.2337/dc05-1973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Lynch CP, Williams JL, Reid J, et al. Racial/ethnic differences in multiple diabetes outcomes in patients with type 2 diabetes in the southeastern United States. Ethn Dis 2014;24:189–94. [PubMed] [Google Scholar]
- 169.McCoy RG, Lipska KJ, Van Houten HK, et al. Association of cumulative multimorbidity, glycemic control, and medication use with hypoglycemia-related emergency department visits and hospitalizations among adults with diabetes. JAMA Netw Open 2020;3:e1919099. 10.1001/jamanetworkopen.2019.19099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Rodriguez-Gutierrez R, Herrin J, Lipska KJ, et al. Racial and ethnic differences in 30-day hospital readmissions among US adults with diabetes. JAMA Netw Open 2019;2:e1913249. 10.1001/jamanetworkopen.2019.13249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Karter AJ, Lipska KJ, O’Connor PJ, et al. High rates of severe hypoglycemia among African American patients with diabetes: the surveillance, prevention, and management of diabetes mellitus (SUPREME-DM) network. J Diabetes Complications 2017;31:869–73. 10.1016/j.jdiacomp.2017.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Pathak RD, Schroeder EB, Seaquist ER, et al. Severe hypoglycemia requiring medical intervention in a large cohort of adults with diabetes receiving care in U.S. integrated health care delivery systems: 2005-2011. Diabetes Care 2016;39:363–70. 10.2337/dc15-0858 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.McCoy RG, Galindo RJ, Swarna KS, et al. Sociodemographic, clinical, and treatment-related factors associated with hyperglycemic crises among adults with type 1 and type 2 diabetes in the United States, 2014-2020. JAMA Netw Open 2021;4:e2123471. 10.1001/jamanetworkopen.2021.23471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Dias JP, Shardell M, Golden SH, et al. Racial/ethnic trends in prevalence of diabetic kidney disease in the United States. Kidney Int Rep 2019;4:334–7. 10.1016/j.ekir.2018.10.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Gregg EW, Williams DE, Geiss L. Changes in diabetes-related complications in the United States, 1990-2010. N Engl J Med 2014;371:286–7. 10.1056/NEJMc1406009 [DOI] [PubMed] [Google Scholar]
- 176.Lanting LC, Joung IMA, Mackenbach JP, et al. Ethnic differences in mortality, end-stage complications, and quality of care among diabetic patients: a review. Diabetes Care 2005;28:2280–8. 10.2337/diacare.28.9.2280 [DOI] [PubMed] [Google Scholar]
- 177.Young BA, Maynard C, Boyko EJ. Racial differences in diabetic nephropathy, cardiovascular disease, and mortality in a national population of veterans. Diabetes Care 2003;26:2392–9. 10.2337/diacare.26.8.2392 [DOI] [PubMed] [Google Scholar]
- 178.Karter AJ, Ferrara A, Liu JY, et al. Ethnic disparities in diabetic complications in an insured population. JAMA 2002;287:2519–27. 10.1001/jama.287.19.2519 [DOI] [PubMed] [Google Scholar]
- 179.Emanuele N, Moritz T, Klein R, et al. Ethnicity, race, and clinically significant macular edema in the veterans affairs diabetes trial (VADT). Diabetes Res Clin Pract 2009;86:104–10. 10.1016/j.diabres.2009.08.001 [DOI] [PubMed] [Google Scholar]
- 180.Mansour O, Golden SH, Yeh H-C. Disparities in mortality among adults with and without diabetes by sex and race. J Diabetes Complications 2020;34:107496. 10.1016/j.jdiacomp.2019.107496 [DOI] [PubMed] [Google Scholar]
- 181.Chang MH, Moonesinghe R, Athar HM, et al. Trends in disparity by sex and race/ethnicity for the leading causes of death in the United States-1999-2010. J Public Health Manag Pract 2016;22 Suppl 1:S13–24. 10.1097/PHH.0000000000000267 [DOI] [PubMed] [Google Scholar]
- 182.McCoy RG, Van Houten HK, Dunlay SM, et al. Race and sex differences in the initiation of diabetes drugs by privately insured US adults. Endocrine 2021;73:480–4. 10.1007/s12020-021-02710-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.McCoy RG, Van Houten HK, Deng Y, et al. Comparison of diabetes medications used by adults with commercial insurance vs medicare advantage, 2016 to 2019. JAMA Netw Open 2021;4:e2035792. 10.1001/jamanetworkopen.2020.35792 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Pham TM, Carpenter JR, Morris TP, et al. Ethnic differences in the prevalence of type 2 diabetes diagnoses in the UK: cross-sectional analysis of the health improvement network primary care database. Clin Epidemiol 2019;11:1081–8. 10.2147/CLEP.S227621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Mathur R, Farmer RE, Eastwood SV, et al. Ethnic disparities in initiation and intensification of diabetes treatment in adults with type 2 diabetes in the UK, 1990–2017: a cohort study. PLOS Med 2020;17:e1003106. 10.1371/journal.pmed.1003106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Zhu Li AP, Whyte MB. Disparities in diabetes care. EMJ Diabetes 2021;9:92–101. 10.33590/emjdiabet/21-00194 [DOI] [Google Scholar]
- 187.Lin X, Xu Y, Pan X, et al. Global, regional, and national burden and trend of diabetes in 195 countries and territories: an analysis from 1990 to 2025. Sci Rep 2020;10:14790. 10.1038/s41598-020-71908-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Jiang DH, O’Connor PJ, Huguet N, et al. Modernizing diabetes care quality measures. Health Affairs 2022;41:955–62. 10.1377/hlthaff.2022.00233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Wharton S, Davies M, Dicker D, et al. Managing the gastrointestinal side effects of GLP-1 receptor agonists in obesity: recommendations for clinical practice. Postgrad Med 2022;134:14–9. 10.1080/00325481.2021.2002616 [DOI] [PubMed] [Google Scholar]
- 190.Nauck MA. A critical analysis of the clinical use of incretin-based therapies: the benefits by far outweigh the potential risks. Diabetes Care 2013;36:2126–32. 10.2337/dc12-2504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Shomali M. Optimizing the care of patients with type 2 diabetes using incretin-based therapy: focus on GLP-1 receptor agonists. Clin Diabetes 2014;32:32–43. 10.2337/diaclin.32.1.32 [DOI] [PMC free article] [PubMed] [Google Scholar]