Abstract
Anthracycline-based chemotherapy is associated with increased subsequent breast cancer (SBC) risk in female childhood cancer survivors, but the current evidence is insufficient to support early breast cancer screening recommendations for survivors treated with anthracyclines. In this study, we pooled individual patient data of 17,903 survivors from six well-established studies, of whom 782 (4.4%) developed a SBC, and analyzed dose-dependent effects of individual anthracycline agents on developing SBC and interactions with chest radiotherapy. A dose-dependent increased SBC risk was seen for doxorubicin (hazard ratio (HR) per 100 mg m−2: 1.24, 95% confidence interval (CI): 1.18–1.31), with more than twofold increased risk for survivors treated with ≥200 mg m−2 cumulative doxorubicin dose versus no doxorubicin (HR: 2.50 for 200–299 mg m−2, HR: 2.33 for 300–399 mg m−2 and HR: 2.78 for ≥400 mg m−2). For daunorubicin, the associations were not statistically significant. Epirubicin was associated with increased SBC risk (yes/no, HR: 3.25, 95% CI: 1.59–6.63). For patients treated with or without chest irradiation, HRs per 100 mg m−2 of doxorubicin were 1.11 (95% CI: 1.02–1.21) and 1.26 (95% CI: 1.17–1.36), respectively. Our findings support that early initiation of SBC surveillance may be reasonable for survivors who received ≥200 mg m−2 cumulative doxorubicin dose and should be considered in SBC surveillance guidelines for survivors and future treatment protocols.
Subject terms: Risk factors, Breast cancer, Quality of life, Paediatric cancer, Cancer epidemiology
In a pooled analysis of six international studies involving about 17,900 female survivors of childhood cancer, the use of doxorubicin was associated with a dose-dependent risk of subsequent breast cancer, irrespective of prior chest radiotherapy exposure.
Main
Over the past six decades, survival rates for childhood cancer have improved markedly in resource-rich countries due to improvements in treatment and supportive care. Unfortunately, the life expectancy and quality of life of long-term survivors are compromised by long-term adverse effects of treatments such as subsequent neoplasms1–4. Breast cancer is one of the most frequent subsequent malignant neoplasms among female childhood cancer survivors5–7. Based on strong evidence regarding the effect of chest radiotherapy on subsequent breast cancer (SBC) risk, the International Guideline Harmonization Group (IGHG) recommends initiation of annual breast cancer surveillance for female survivors who received ≥10 Gray (Gy) chest radiotherapy at age 25 years or ≥8 years from radiation8, which is earlier than the population screening programs for breast cancer that typically recommend initiation of screening at age 40 years or 50 years9.
Over time, childhood cancer treatments have been modified to include decreased radiation doses and volumes and increased use of chemotherapy, especially anthracyclines10. Several previous studies have shown that anthracycline exposure is associated with increased SBC risk7,11–15, including several Childhood Cancer Survivor Study (CCSS) reports7,11,14,15, the St. Jude Lifetime Cohort Study (SJLIFE)13 and the Dutch Childhood Cancer Survivor Study LATER (DCCSS-LATER)12. These studies investigated the dose-dependent associations between the sum of anthracycline agents dose and SBC risk. Only a few studies evaluated the dose effects of doxorubicin, an individual anthracycline agent, on SBC risk12,14,16. However, these studies all used tertiles of cumulative doxorubicin dose, which are derived from cumulative dose distributions that are study dependent. There is currently no data on dose effects with regard to SBC risks for other individual anthracycline agents (for example, daunorubicin). Moreover, there is little information on the joint effects of anthracyclines and chest radiotherapy14. The current evidence is insufficient to alter the SBC screening recommendations because there was inconsistent evidence on dose thresholds for determining which survivors are at moderate or high risk. Furthermore, there were no data on possible differences in dose effects with regard to SBC risk for the different individual anthracycline agents owing to the limited number of survivors treated with these specific chemotherapy agents in individual cohort studies.
To address these knowledge gaps, detailed treatment data from a large number of individuals are required. Therefore, we pooled individual patient data from six well-established childhood cancer survivor studies in Europe and North America with the aim of estimating the dose-dependent effects of specific anthracycline agents on developing SBC in female childhood cancer survivors, as well as interactions with chest radiotherapy and age at primary cancer diagnosis.
Results
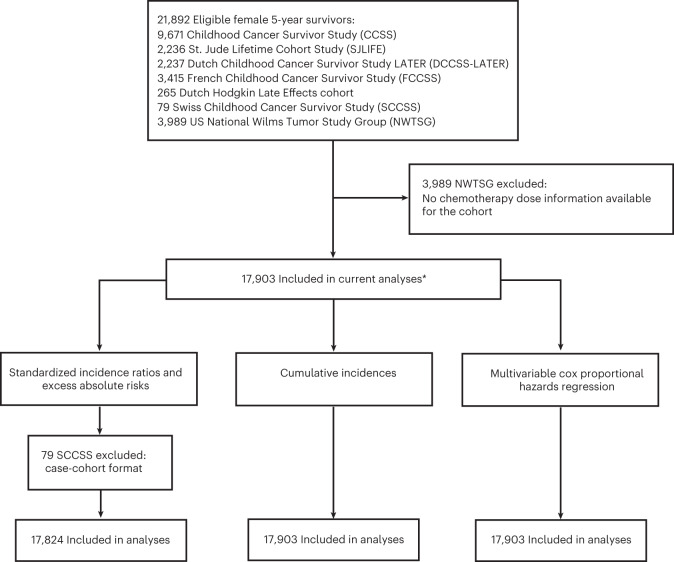
In total, our pooled cohort included 17,903 5-year survivors, with data from five cohort studies (CCSS: 9,671 women, SJLIFE: 2,236 women, DCCSS-LATER: 2,237 women, French Childhood Cancer Survivor Study (FCCSS): 3,415 women and Dutch Hodgkin Late Effects cohort: 265 women), and one case–cohort study (Swiss Childhood Cancer Survivor Study (SCCSS): 79 women) in Europe and North America (Fig. 1).
Fig. 1. Cohort composition diagram of eligible female 5-year childhood cancer survivors in each analysis.
*The number of included survivors in each analysis may vary due to missing values of analysis variables.
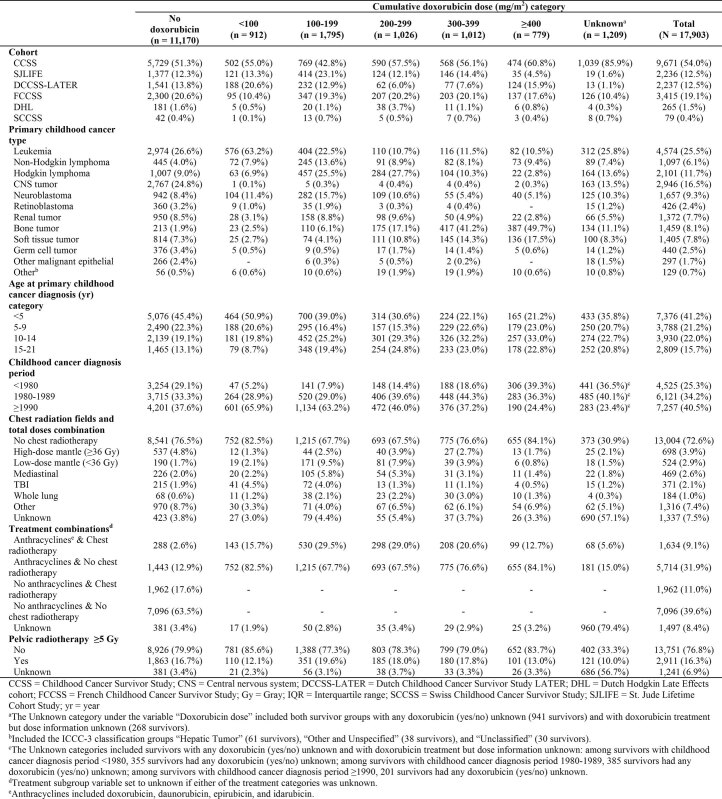
Among the eligible 17,903 5-year survivors, the median age at primary childhood cancer diagnosis was 6.7 years (interquartile range (IQR): 2.8–13.0), with leukemia (25.5%), central nervous system tumor (16.5%) and Hodgkin lymphoma (11.7%) as the most frequent childhood cancer types (Table 1 and Supplementary Tables 1 and 2), with some variations by cohort (Supplementary Table 3). Of all survivors, 5,714 (31.9%) received anthracyclines without chest radiotherapy, 1,962 (11.0%) received chest radiotherapy without anthracyclines, 1,634 (9.1%) received both anthracyclines and chest radiotherapy, 7,096 (39.6%) received neither treatment and for 1,497 (8.4%), it was unclear whether they received anthracycline treatment and/or chest radiotherapy treatment.
Table 1.
Demographic and treatment characteristics of 17,903 female 5-year childhood cancer survivors (primary cancer diagnosis year 1946–2012) overall and by subsequent breast cancer status
| Characteristic | Total (n = 17,903) | Subsequent breast cancera (n = 782) | No subsequent breast cancer (n = 17,121) |
|---|---|---|---|
| No. (%) | No. (%) | No. (%) | |
| Primary childhood cancerb | |||
| Leukemia | 4,574 (25.5) | 81 (10.4) | 4,493 (26.2) |
| Non-Hodgkin lymphoma | 1,097 (6.1) | 37 (4.7) | 1,060 (6.2) |
| Hodgkin lymphoma | 2,101 (11.7) | 405 (51.8) | 1,696 (9.9) |
| Central nervous system tumor | 2,946 (16.5) | 14 (1.8) | 2,932 (17.1) |
| Neuroblastoma | 1,657 (9.3) | 15 (1.9) | 1,642 (9.6) |
| Retinoblastoma | 426 (2.4) | 2 (0.3) | 424 (2.5) |
| Renal tumor | 1,372 (7.7) | 45 (5.8) | 1,327 (7.8) |
| Bone tumor | 1,459 (8.1) | 106 (13.6) | 1,353 (7.9) |
| Soft tissue tumor | 1,405 (7.8) | 55 (7.0) | 1,350 (7.9) |
| Germ cell tumor | 440 (2.5) | 9 (1.2) | 431 (2.5) |
| Other malignant epithelial | 297 (1.7) | 11 (1.4) | 286 (1.7) |
| Otherc | 129 (0.7) | 2 (0.2) | 127 (0.8) |
| Cumulative doxorubicin dose (mg m−2) | |||
| 0 | 11,170 (62.4) | 431 (55.1) | 10,739 (62.7) |
| <100 | 912 (5.1) | 16 (2.0) | 896 (5.2) |
| 100–199 | 1,795 (10.0) | 69 (8.8) | 1,726 (10.1) |
| 200–299 | 1,026 (5.7) | 67 (8.6) | 959 (5.6) |
| 300–399 | 1,012 (5.7) | 64 (8.2) | 948 (5.5) |
| ≥400 | 779 (4.4) | 58 (7.4) | 721 (4.2) |
| Unknownd | 1,209 (6.8) | 77 (9.8) | 1,132 (6.6) |
| Cumulative daunorubicin dose (mg m−2) | |||
| 0 | 14,630 (81.7) | 684 (87.5) | 13,946 (81.5) |
| <100 | 623 (3.5) | 7 (0.9) | 616 (3.6) |
| 100–199 | 953 (5.3) | 16 (2.0) | 937 (5.5) |
| ≥200 | 645 (3.6) | 17 (2.2) | 628 (3.7) |
| Unknowne | 1,052 (5.9) | 58 (7.4) | 994 (5.8) |
| Epirubicin | |||
| No | 16,637 (92.9) | 717 (91.7) | 15,920 (93.0) |
| Yes | 325 (1.8) | 9 (1.2) | 316 (1.8) |
| Unknown | 941 (5.3) | 56 (7.2) | 885 (5.2) |
| Idarubicin | |||
| No | 16,843 (94.1) | 725 (92.7) | 16,118 (94.1) |
| Yes | 107 (0.6) | 1 (0.1) | 106 (0.6) |
| Unknown | 953 (5.3) | 56 (7.2) | 897 (5.2) |
| CEDf (mg m−2) | |||
| 0 | 7,951 (44.4) | 301 (38.5) | 7,650 (44.7) |
| <6,000 | 3,069 (17.1) | 94 (12.0) | 2,975 (17.4) |
| 6,000–17,999 | 3,899 (21.8) | 192 (24.6) | 3,707 (21.7) |
| ≥18,000 | 1,117 (6.2) | 47 (6.0) | 1,070 (6.2) |
| Unknown | 1,867 (10.4) | 148 (18.9) | 1,719 (10.0) |
| Chest radiotherapy fields and dosesg | |||
| No chest radiotherapy | 13,004 (72.6) | 250 (32.0) | 12,754 (74.5) |
| High-dose mantle (≥36 Gy; median: 40 Gy, IQR: 39–44 Gy)h | 698 (3.9) | 238 (30.4) | 460 (2.7) |
| Low-dose mantle (<36 Gy; median: 26 Gy, IQR: 21–30 Gy)h | 524 (2.9) | 93 (11.9) | 431 (2.5) |
| Mediastinal (median: 26 Gy, IQR: 21–36 Gy)h | 469 (2.6) | 33 (4.2) | 436 (2.5) |
| TBI (median: 12 Gy, IQR: 11–13 Gy)h | 371 (2.1) | 22 (2.8) | 349 (2.0) |
| Whole lung (median: 16 Gy, IQR: 12–23 Gy)h | 184 (1.0) | 23 (2.9) | 161 (0.9) |
| Other (median: 28 Gy, IQR: 21–36 Gy)h | 1,316 (7.4) | 63 (8.1) | 1,253 (7.3) |
| Unknown | 1,337 (7.5) | 60 (7.7) | 1,277 (7.5) |
| Pelvic radiotherapy dosei | |||
| No pelvic radiotherapy | 13,727 (76.7) | 505 (64.6) | 13,222 (77.2) |
| <10 Gy | 142 (0.8) | 6 (0.8) | 136 (0.8) |
| 10–19 Gy | 594 (3.3) | 24 (3.1) | 570 (3.3) |
| 20–29 Gy | 719 (4.0) | 38 (4.9) | 681 (4.0) |
| 30–39 Gy | 767 (4.3) | 82 (10.5) | 685 (4.0) |
| ≥40 Gy | 713 (4.0) | 72 (9.2) | 641 (3.7) |
| Unknown | 1,241 (6.9) | 55 (7.0) | 1,186 (6.9) |
| Age at diagnosis of primary cancer (years) | |||
| <5 | 7,376 (41.2) | 66 (8.4) | 7,310 (42.7) |
| 5–9 | 3,788 (21.2) | 65 (8.3) | 3,723 (21.7) |
| 10–14 | 3,930 (22.0) | 273 (34.9) | 3,657 (21.4) |
| 15–21 | 2,809 (15.7) | 378 (48.3) | 2,431 (14.2) |
| Treatment subgroupsj | |||
| Anthracyclinek and chest radiotherapy | 1,634 (9.1) | 163 (20.8) | 1,471 (8.6) |
| Anthracycline and no chest radiotherapy | 5,714 (31.9) | 156 (19.9) | 5,558 (32.5) |
| No anthracycline and chest radiotherapy | 1,962 (11.0) | 294 (37.6) | 1,668 (9.7) |
| No anthracycline and no chest radiotherapy | 7,096 (39.6) | 83 (10.6) | 7,013 (41.0) |
| Unknown | 1,497 (8.4) | 86 (11.0) | 1,411 (8.2) |
aIncluded both invasive and ductal carcinoma in situ breast cancer.
bBecause of the eligibility criteria of the cohort, the composition of primary cancer diagnosis groups in our pooled data may differ from the composition in underlying populations of childhood cancer survivors.
cIncluded the ICCC-3 classification groups ‘hepatic tumor’ (0 case/61 survivors), ‘other and unspecified’ (1 case/38 survivors), and ‘unclassified’ (1 case/30 survivors).
dThe unknown category under the variable ‘doxorubicin dose’ included both survivor groups with any doxorubicin (yes/no) unknown (56 cases/941 survivors) and with doxorubicin treatment but dose information unknown (21 cases/268 survivors).
eThe unknown category under the variable ‘daunorubicin dose’ included both survivor groups with any daunorubicin (yes/no) unknown (56 cases/953 survivors) and with daunorubicin treatment but dose information unknown (2 cases/99 survivors).
fCED calculation: CED (mg m−2) = 1.0 (cumulative cyclophosphamide dose (mg m−2)) + 0.244 (cumulative ifosfamide dose (mg m−2)) + 0.857 (cumulative procarbazine dose (mg m−2)) + 14.286 (cumulative chlorambucil dose (mg m−2)) + 15.0 (cumulative BCNU (carmustine) dose (mg m−2)) + 16.0 (cumulative CCNU (lomustine) dose (mg m−2)) + 40 (cumulative melphalan dose (mg m−2)) + 50 (cumulative Thio-TEPA (thiotepa) dose (mg m−2)) + 100 (cumulative nitrogen mustard dose (mg m−2)) + 8.823 (cumulative busulfan dose (mg m−2)).
gIncluded radiotherapy fields exposing (parts of) the chest. Radiation dose referred to the cumulative prescribed dose (including boost doses, if applicable), or slight variations, depending on definitions in the underlying cohorts25. Chest radiotherapy was categorized as the combination of chest radiation fields with the associated maximum chest radiotherapy dose below or above the median. The variable was classified as follows: high-dose mantle (median: 40 Gy, IQR: 39–44 Gy), low-dose mantle (median: 26 Gy, IQR: 21–30 Gy), mediastinal (median: 26 Gy, IQR: 21–36 Gy), TBI (median: 12 Gy, IQR: 11–13 Gy), whole lung (median: 16 Gy, IQR: 12–23 Gy), other (median: 28 Gy, IQR: 21–36 Gy) and unknown.
hDose represents the maximum cumulative prescribed chest dose (including boost doses, if applicable) of survivors classified in this group. This could include doses to chest field other than this category.
iIncluded radiotherapy fields exposing (parts of) the pelvis (including TBI). Radiation dose referred to the cumulative prescribed dose (including boost doses, if applicable), or slight variations, depending on definitions in the underlying cohorts25. The unknown category under the variable ‘pelvic radiotherapy dose’ included both survivor groups with any pelvic radiotherapy (yes/no) unknown (54 cases/1,212 survivors) and with pelvic radiotherapy treatment but dose information unknown (1 case/29 survivors).
jTreatment subgroup variable set to unknown if either of the treatment categories was unknown.
kAnthracyclines included doxorubicin, daunorubicin, epirubicin and idarubicin.
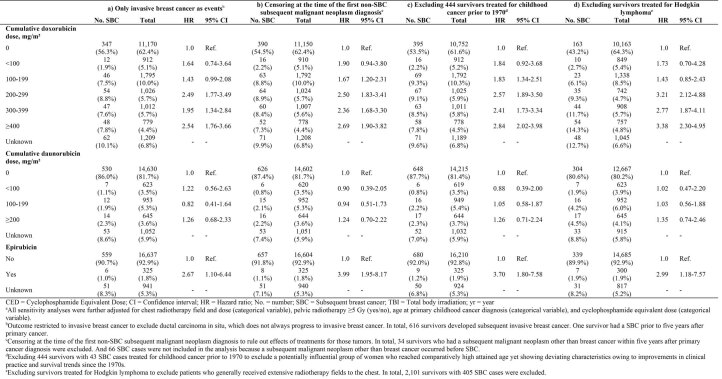
Among survivors with a cumulative doxorubicin dose of ≥200 mg m−2, the most common childhood cancer types were bone tumors, Hodgkin lymphoma and soft tissue sarcomas (Extended Data Table 1). The highest percentage of survivors who received a cumulative doxorubicin dose of ≥200 mg m−2 was observed in the Dutch Hodgkin Late Effects cohort (20.8%), followed by the CCSS (16.9%) and the FCCSS (16.0%). Although the percentage of survivors who received any doxorubicin treatment was higher in the diagnosis period ≥1990 (39.3%) compared to the diagnosis periods 1980–1989 (33.0%) and <1980 (20.2%), the percentage of survivors who received ≥200 mg m−2 cumulative doxorubicin dose was fairly similar across diagnosis period ≥1990 (14.3%), diagnosis period 1980–1989 (18.6%) and diagnosis period <1980 (14.2%). The median follow-up time after primary cancer diagnosis was 24.9 years (IQR: 19.1–33.2). In total, 782 survivors developed the first SBC at a median age of 39.7 years (IQR: 34.3–44.9), including 616 invasive breast cancer and 166 ductal carcinoma in situ (DCIS) cases. The median attained age at the end of follow-up was 33.7 years (IQR: 25.9–41.6) and 29.6% of survivors attained an age of 40 years or more.
Extended Data Table 1.
Demographic and treatment characteristics of female five-year childhood cancer survivors by cumulative doxorubicin dose categories
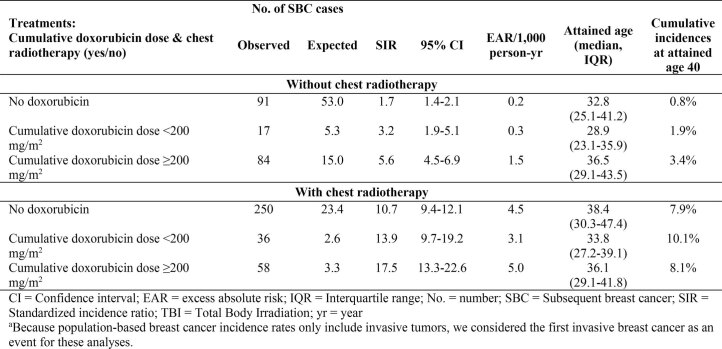
Comparison with the general population
In Extended Data Table 2, breast cancer risk by doxorubicin and chest radiotherapy treatment is provided. Compared with the general female population, the risk of invasive breast cancer was most elevated in the survivor group that received a cumulative doxorubicin dose ≥200 mg m−2 and chest radiotherapy (standardized incidence ratio (SIR): 17.5, 95% confidence interval (CI): 13.3–22.6; median attained age, 36.1 years), followed by the cumulative doxorubicin dose <200 mg m−2 and chest radiotherapy group (SIR: 13.9, 95%: CI: 9.7–19.2; median attained age, 33.8 years), then by the chest radiotherapy-only with no doxorubicin group (SIR: 10.7, 95%: CI 9.4–12.1; median attained age, 38.4 years), then by the cumulative doxorubicin dose ≥200 mg m−2 with no chest radiotherapy group (SIR: 5.6, 95% CI: 4.5–6.9; median attained age, 36.5 years), then by the cumulative doxorubicin dose <200 mg m−2 with no chest radiotherapy group (SIR: 3.2, 95% CI: 1.9–5.1; median attained age, 28.9 years) and finally by the group receiving neither doxorubicin nor chest radiotherapy (SIR: 1.7, 95% CI: 1.4–2.1; median attained age, 32.8 years). The highest excess absolute risk (EAR) was observed in the cumulative doxorubicin dose ≥200 mg m−2 and chest radiotherapy group with 5.0 excess cases per 1,000 person-years.
Extended Data Table 2.
Standardized incidence ratios, excess absolute risks, and cumulative incidences by treatmentsa
Risk factors for SBC
In multivariable Cox regression analyses, cumulative doxorubicin dose was associated with an increased risk of SBC, with a hazard ratio (HR) of 1.76 (95% CI: 0.88–3.51) for <100 mg m−2, HR of 1.77 (95% CI: 1.30–2.42) for 100–199 mg m−2, HR of 2.50 (95% CI: 1.85–3.40) for 200–299 mg m−2, HR of 2.33 (95% CI: 1.68–3.23) for 300–399 mg m−2 and HR of 2.78 (95% CI: 1.99–3.88) for ≥400 mg m−2 cumulative doxorubicin dose category compared to the no doxorubicin treatment (Table 2 Model I; survivor characteristics by cumulative doxorubicin dose categories are shown in Extended Data Table 1). Compared to those not treated with daunorubicin, HRs were close to one for those with cumulative doses of daunorubicin <200 mg m−2 (HR: 0.98, 95% CI: 0.46–2.09 for <100 mg m−2 and HR: 0.98, 95% CI: 0.55–1.75 for 100–199 mg m−2), and the highest cumulative dose group, ≥200 mg m−2, conferred a nonstatistically significant association (HR: 1.22, 95% CI: 0.69–2.17). When the continuous cumulative doxorubicin and daunorubicin dose information was included in the model, the risk of developing SBC in survivors treated with doxorubicin increased 1.24-fold (HR per 100 mg m−2 1.24, 95% CI: 1.18–1.31) for every 100 mg m−2 increase in cumulative doxorubicin dose after adjustments (Table 2, model II). Cumulative daunorubicin dose and risk of SBC were not statistically significant (HR per 100 mg m−2 1.10, 95% CI: 0.95–1.29). Epirubicin treatment was associated with an increased SBC risk (yes versus no, HR: 3.25, 95% CI: 1.59–6.63).
Table 2.
Multivariable Cox proportional hazard regression analyses for subsequent breast cancer in female 5-year childhood cancer survivors (primary cancer diagnosis year 1946–2012)
| Characteristic | Total (n) | % | No. of SBC (n)c | % | Model Ia | Model IIb | ||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | HR | 95% CI | |||||
| Cumulative doxorubicin dose (mg m−²) | ||||||||
| 0 | 11,170 | 62.4 | 431 | 55.1 | 1.0 | Ref. | – | |
| <100 | 912 | 5.1 | 16 | 2.0 | 1.76 | 0.88–3.51 | ||
| 100–199 | 1,795 | 10.0 | 69 | 8.8 | 1.77 | 1.30–2.42 | ||
| 200–299 | 1,026 | 5.7 | 67 | 8.6 | 2.50 | 1.85–3.40 | ||
| 300–399 | 1,012 | 5.7 | 64 | 8.2 | 2.33 | 1.68–3.23 | ||
| ≥400 | 779 | 4.4 | 58 | 7.4 | 2.78 | 1.99–3.88 | ||
| Unknown | 1,209 | 6.8 | 77 | 9.8 | – | – | ||
| Continuous variable: cumulative doxorubicin dose (per 100 mg m−²) | – | – | – | – | – | – | 1.24 | 1.18–1.31 |
| Cumulative daunorubicin dose (mg m−²) | ||||||||
| 0 | 14,630 | 81.7 | 684 | 87.5 | 1.0 | Ref. | – | |
| <100 | 623 | 3.5 | 7 | 0.9 | 0.98 | 0.46–2.09 | ||
| 100–199 | 953 | 5.3 | 16 | 2.0 | 0.98 | 0.55–1.75 | ||
| ≥200 | 645 | 3.6 | 17 | 2.2 | 1.22 | 0.69–2.17 | ||
| Unknown | 1,052 | 5.9 | 58 | 7.4 | – | – | ||
| Continuous variable: cumulative daunorubicin dose (per 100 mg m−²) | – | – | – | – | – | – | 1.10 | 0.95–1.29 |
| Epirubicin | ||||||||
| No | 16,637 | 92.9 | 717 | 91.7 | 1.0 | Ref. | 1.0 | Ref. |
| Yes | 325 | 1.8 | 9 | 1.2 | 3.40 | 1.66–6.98 | 3.25 | 1.59–6.63 |
| Unknown | 941 | 5.3 | 56 | 7.2 | – | – | – | – |
| Chest radiotherapy field and dose | ||||||||
| No chest radiotherapy | 13,004 | 72.6 | 250 | 32.0 | 1.0 | Ref. | 1.0 | Ref. |
| High-dose mantle (≥36 Gy; median: 40 Gy, IQR: 39–44 Gy) | 698 | 3.9 | 238 | 30.4 | 8.99 | 7.00–11.53 | 9.12 | 7.09–11.75 |
| Low-dose mantle (<36 Gy; median: 26 Gy, IQR: 21–30 Gy) | 524 | 2.9 | 93 | 11.9 | 4.72 | 3.48–6.41 | 5.23 | 3.86–7.09 |
| Mediastinal (median: 26 Gy, IQR: 21–36 Gy) | 469 | 2.6 | 33 | 4.2 | 1.65 | 1.02–2.67 | 1.71 | 1.06–2.78 |
| TBI (median: 12 Gy, IQR: 11–13 Gy) | 371 | 2.1 | 22 | 2.8 | 7.05 | 4.11–12.10 | 7.18 | 4.18–12.33 |
| Whole lung (median: 16 Gy, IQR: 12–23 Gy) | 184 | 1.0 | 23 | 2.9 | 7.58 | 4.68–12.27 | 8.00 | 4.94–12.95 |
| Other (median: 28 Gy, IQR: 21–36 Gy) | 1,316 | 7.4 | 63 | 8.1 | 2.61 | 1.87–3.64 | 2.68 | 1.91–3.75 |
| Unknown | 1,337 | 7.5 | 60 | 7.7 | – | – | – | – |
| Pelvic radiotherapy ≥5 Gy | ||||||||
| No | 13,751 | 76.8 | 505 | 64.6 | 1.0 | Ref. | 1.0 | Ref. |
| Yes | 2,911 | 16.3 | 222 | 28.4 | 0.95 | 0.78–1.17 | 0.94 | 0.77–1.16 |
| Unknown | 1,241 | 6.9 | 55 | 7.0 | – | – | – | – |
| Age at primary childhood cancer diagnosis (year) | ||||||||
| <5 | 7,376 | 41.2 | 66 | 8.4 | 1.0 | Ref. | 1.0 | Ref. |
| 5–9 | 3,788 | 21.2 | 65 | 8.3 | 1.13 | 0.76–1.69 | 1.12 | 0.75–1.67 |
| 10–14 | 3,930 | 22.0 | 273 | 34.9 | 2.03 | 1.48–2.79 | 2.04 | 1.49–2.81 |
| 15–21 | 2,809 | 15.7 | 378 | 48.3 | 1.83 | 1.31–2.55 | 1.84 | 1.32–2.57 |
| CEDd (mg m−2) | ||||||||
| None | 7,951 | 44.4 | 301 | 38.5 | 1.0 | Ref. | 1.0 | Ref. |
| <6,000 | 3,069 | 17.1 | 94 | 12.0 | 0.87 | 0.67–1.14 | 0.95 | 0.73–1.25 |
| 6,000–17,999 | 3,899 | 21.8 | 192 | 24.6 | 1.02 | 0.82–1.27 | 1.07 | 0.86–1.32 |
| ≥18,000 | 1,117 | 6.2 | 47 | 6.0 | 1.20 | 0.83–1.74 | 1.23 | 0.85–1.77 |
| Unknown | 1,867 | 10.4 | 148 | 18.9 | – | – | – | – |
aModel I included categorical variables of cumulative doxorubicin and daunorubicin dose by steps of 100 mg m−2.
bModel II included continuous variables of cumulative doxorubicin and daunorubicin dose per 100 mg m−2.
cOne survivor had SBC before 5 years after primary cancer.
dCED calculation: CED (mg m−2) = 1.0 (cumulative cyclophosphamide dose (mg m−2)) + 0.244 (cumulative ifosfamide dose (mg m−2)) + 0.857 (cumulative procarbazine dose (mg m−2)) + 14.286 (cumulative chlorambucil dose (mg m−2)) + 15.0 (cumulative BCNU (carmustine) dose (mg m−2)) + 16.0 (cumulative CCNU (lomustine) dose (mg m−2)) + 40 (cumulative melphalan dose (mg m−2)) + 50 (cumulative Thio-TEPA (thiotepa) dose (mg m−2)) + 100 (cumulative nitrogen mustard dose (mg m−2)) + 8.823 (cumulative busulfan dose (mg m−2)).
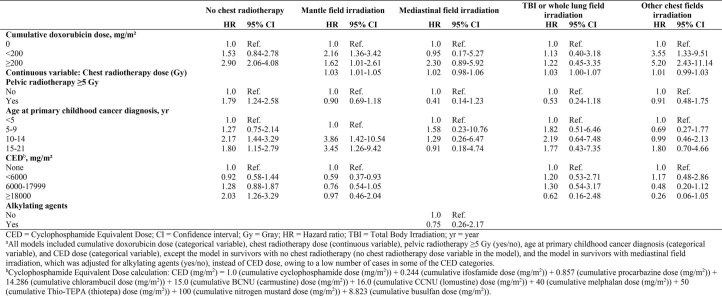
Additionally, all chest radiotherapy field and dose categories were significantly associated with increased SBC risk, with the highest HRs for those treated with high-dose mantle field (HR: 8.99, 95% CI: 7.00–11.53), followed by whole lung irradiation (HR: 7.58, 95% CI: 4.68–12.27), and total body irradiation (TBI; HR: 7.05, 95% CI: 4.11–12.10; Table 2, model I). Survivors with a primary cancer diagnosis at ages 10–14 or 15–21 years had an elevated risk of SBC with HRs of 2.03 (95% CI: 1.48–2.79) for 10–14 years and 1.83 (95% CI: 1.31–2.55) for 15–21 years compared with the survivors who were diagnosed at ages 0–4. We did not observe significant effects of pelvic radiotherapy or alkylating agents (cyclophosphamide equivalent dose (CED)) on SBC risk. HR per 100 mg m−2 of cumulative doxorubicin dose was 1.11 (95% CI: 1.02–1.21) for survivors who received chest radiotherapy and 1.26 (95% CI: 1.17–1.36) for survivors who did not receive chest radiotherapy (Table 3).
Table 3.
Multivariable Cox proportional hazard regression analyses for subsequent breast cancer by chest radiotherapy status among female 5-year childhood cancer survivors (primary cancer diagnosis year 1946–2012)
| Models without interaction | ||||
|---|---|---|---|---|
| Characteristic | With chest radiotherapya | Without chest radiotherapya | ||
| HR | 95% CI | HR | 95% CI | |
|
Continuous variable: cumulative doxorubicin dose (per 100 mg m−²) |
1.11 | 1.02–1.21 | 1.26 | 1.17–1.36 |
|
Continuous variable: cumulative daunorubicin dose (per 100 mg m−²) |
0.95 | 0.74–1.21 | 1.12 | 0.93–1.36 |
| Epirubicin | ||||
| No | 1.0 | Ref. | 1.0 | Ref. |
| Yes | 2.28 | 1.00–5.21 | 2.13 | 0.49–9.17 |
| Models with a multiplicative interaction | ||||
| – | Interaction: cumulative doxorubicin dose × chest radiotherapy status (yes/no) | Interaction: cumulative daunorubicin dose × chest radiotherapy status (yes/no) | ||
| HR | 95% CI | HR | 95% CI | |
|
Continuous variable: Cumulative doxorubicin dose (per 100 mg m−²) |
1.28 | 1.21–1.37 | 1.21 | 1.15–1.28 |
|
Continuous variable: cumulative daunorubicin dose (per 100 mg m−²) |
1.04 | 0.89–1.23 | 1.15 | 0.96–1.37 |
| Epirubicin | ||||
| No | 1.0 | Ref. | 1.0 | Ref. |
| Yes | 2.54 | 1.22–5.30 | 2.47 | 1.17–5.23 |
| Interaction: cumulative doxorubicin dose (per 100 mg m−2) × chest radiotherapy status (yes/no) | 0.86 | 0.78–0.96 | – | – |
| Interaction: cumulative daunorubicin dose (per 100 mg m−2) × chest radiotherapy status (yes/no) | – | – | 0.77 | 0.57–1.05 |
aModels were further adjusted for pelvic radiotherapy ≥5 Gy (yes/no), age at primary childhood cancer diagnosis (categorical variable), and cyclophosphamide equivalent dose (categorical variable).
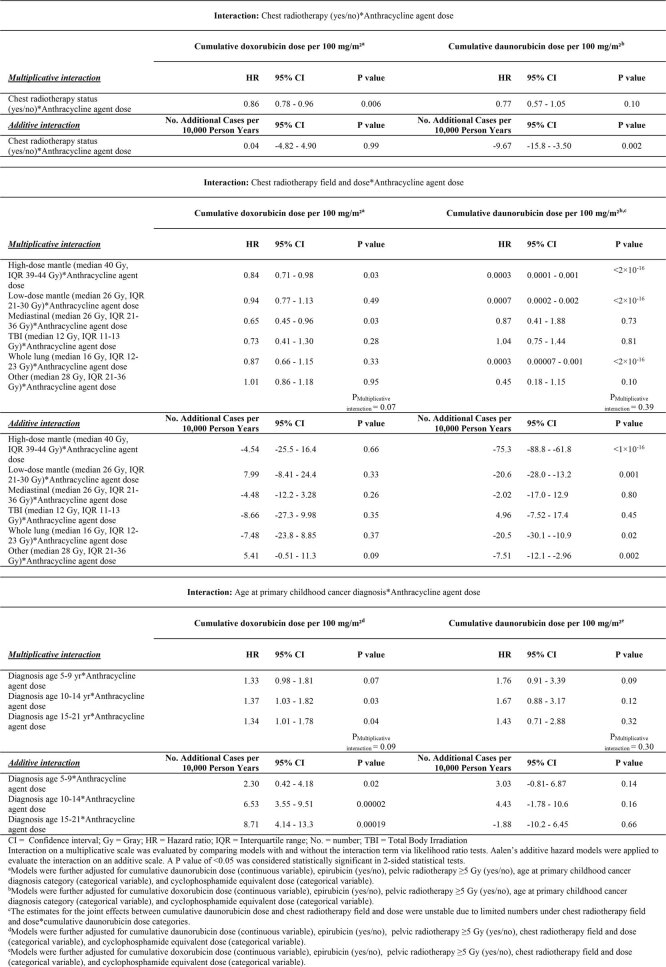
Joint effects of continuous cumulative doxorubicin dose and chest radiation (yes versus no) were submultiplicative (HRmultiplicative interaction: 0.86, 95% CI: 0.78–0.96, Pmultiplicative interaction = 0.006) and compatible with additive effects (Padditive interaction = 0.99; Extended Data Table 3). The effect of cumulative doxorubicin dose on SBC risk was significantly less strong among those with high-dose mantle field (HRmultiplicative interaction: 0.84, 95% CI: 0.71–0.98, Pmultiplicative interaction = 0.03) and mediastinal field irradiation (HRmultiplicative interaction: 0.65, 95% CI: 0.45–0.96, Pmultiplicative interaction = 0.03), compared to those treated without chest radiotherapy. On an additive scale, the joint effects of cumulative doxorubicin dose and chest radiotherapy fields were equal to the sum of these two individual effects (all Padditive interaction > 0.05). Joint effects of daunorubicin and chest radiation were on a multiplicative scale (Pmultiplicative interaction = 0.10) and significantly less than additive (no. of additional cases per 10,000 person-years: −9.67, Padditive interaction = 0.002).
Extended Data Table 3.
Joint effects of cumulative doxorubicin and daunorubicin doses and other relevant risk factors on subsequent breast cancer among female five-year childhood cancer survivors
Age at childhood cancer diagnosis did not significantly modify the effects of cumulative doxorubicin and daunorubicin dose on SBC risk on a multiplicative scale (Pmultiplicative interaction = 0.09 and Pmultiplicative interaction = 0.30, respectively). However, on an additive scale, the joint effects of cumulative doxorubicin dose and age at childhood cancer diagnosis (5–9; 10–14; 15–21 versus 0–4 years) were all significantly greater than the sum of the individual effects (all Padditive interaction < 0.05). Such an effect was not found for cumulative daunorubicin dose.
To rule out potential effects of other treatments that have been associated with SBC, such as chest radiotherapy and alkylating agents, we performed separate analyses in survivors who received neither chest radiotherapy nor alkylating agents; the effects of high cumulative doxorubicin dose on SBC risk remained statistically significant, with HR of 2.67 (95% CI: 1.08–6.59) for 300–399 mg m−2 and HR of 3.58 (95% CI: 1.66–7.71) for ≥400 mg m−2 cumulative doxorubicin dose category (Table 4).
Table 4.
Multivariable Cox proportional hazard regression analyses for subsequent breast cancer in female 5-year childhood cancer survivors whose treatment history did not include chest radiotherapy nor alkylating agent chemotherapy (primary cancer diagnosis year 1946–2012)a
| Total (n) | % | No. of SBC (n) | % | HRb | 95% CI | |
|---|---|---|---|---|---|---|
| Cumulative doxorubicin dose (mg m−²) | ||||||
| 0 | 5,880 | 90.5 | 61 | 71.8 | 1.0 | Ref. |
| <300c | 305 | 4.7 | 7 | 8.2 | 2.35 | 0.96–5.71 |
| 300–399 | 131 | 2.0 | 5 | 5.9 | 2.67 | 1.08–6.59 |
| ≥400 | 136 | 2.1 | 8 | 9.4 | 3.58 | 1.66–7.71 |
| Unknown | 47 | 0.7 | 4 | 4.7 | – | – |
| Daunorubicin | ||||||
| No | 6,020 | 92.6 | 81 | 95.3 | 1.0 | Ref. |
| Yes | 479 | 7.4 | 4 | 4.7 | 1.43 | 0.48–4.24 |
| Epirubicin | ||||||
| No | 6,362 | 97.9 | 84 | 98.8 | 1.0 | Ref. |
| Yes | 137 | 2.1 | 1 | 1.2 | 3.46 | 0.41–29.10 |
aIn total, 6,499 female 5-year survivors did not receive chest radiotherapy nor alkylating agent chemotherapy, among whom 85 developed SBC during follow-up.
bModels were further adjusted for pelvic radiotherapy ≥5 Gy (yes/no) and age at primary childhood cancer diagnosis (categorical variable).
cThe cumulative doxorubicin dose (mg m−2) categories <100 (0 case/46 survivors), 100–199 (1 case/129 survivors) and 200–299 (6 cases/130 survivors) were collapsed due to low numbers of SBC cases.
Cumulative incidences
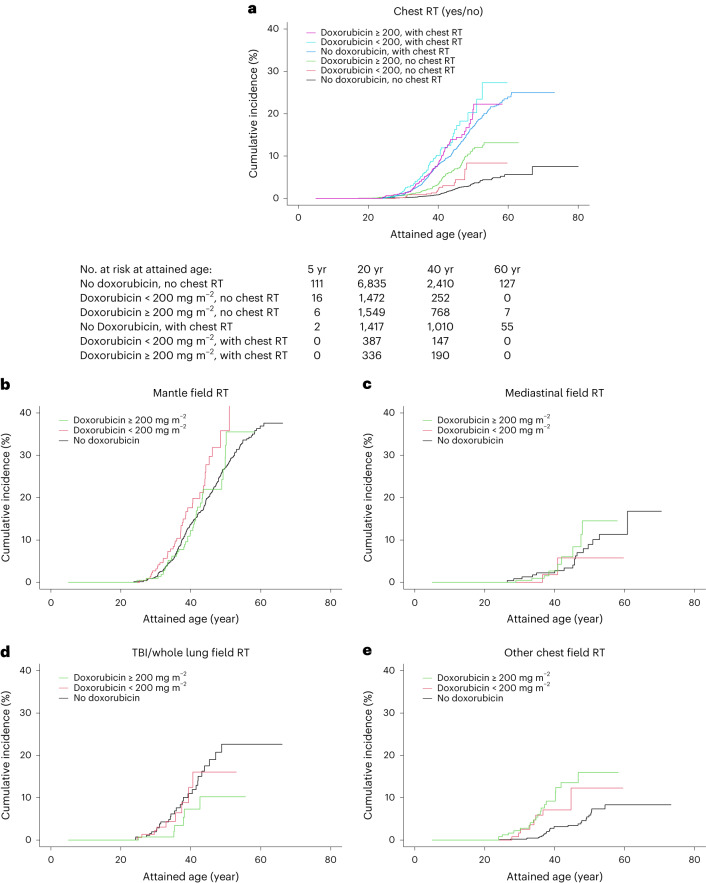
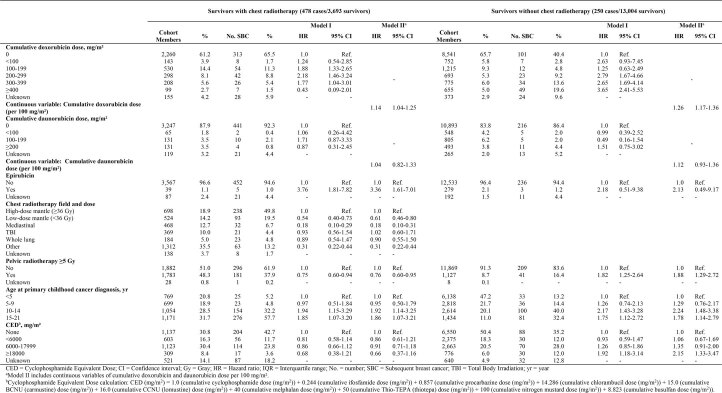
For survivors who did not receive chest radiotherapy, cumulative incidences at the age of 40 years were 0.8% for no doxorubicin treatment group, 1.9% for <200 mg m−2 cumulative doxorubicin dose group and 3.4% for ≥200 mg m−2 cumulative doxorubicin dose group; for survivors who received chest radiotherapy, corresponding cumulative incidences at age 40 for the three cumulative dose groups were 7.9% for no doxorubicin treatment group, 10.1% for <200 mg m−2 cumulative doxorubicin dose group and 8.1% for ≥200 mg m−2 cumulative doxorubicin dose group (Fig. 2a), with some variation by chest radiation field (Fig. 2b–e). In Extended Data Table 4, multivariable Cox regression results for these cumulative doxorubicin dose categories are presented.
Fig. 2. Cumulative incidence of subsequent breast cancer in female 5-year childhood cancer survivors by cumulative doxorubicin dose, stratified by chest radiotherapy status (primary cancer diagnosis year 1946–2012).
Stratification by a, chest radiotherapy status; b, Mantle field; c, Mediastinal field; d, TBI/whole lung field; e, Other chest field. The SCCSS was excluded from cumulative incidence analyses due to the case–cohort design. The cumulative incidence figures represent univariable comparisons. Multivariable Cox regression results for the cumulative doxorubicin dose categories presented in this figure are shown in Extended Data Table 4. No., number; RT, radiotherapy.
Extended Data Table 4.
Multivariable Cox proportional hazard regression analyses for subsequent breast cancer in female five-year childhood cancer survivors with different chest radiotherapy fields irradiationa
Sensitivity analyses
Sensitivity analyses including (a) only invasive breast cancer as an outcome, (b) censoring at the time of first non-SBC subsequent malignant tumor, (c) excluding females treated before 1970, (d) excluding patients with Hodgkin lymphoma (all in Extended Data Table 5) and (e) excluding each cohort on a one-by-one basis (Supplementary Table 4) yielded similar results. The results of the models conducted in each cohort are shown in Supplementary Table 5.
Extended Data Table 5.
Sensitivity analyses for risk of subsequent breast cancer on subgroups with different treatments or eventsa
Discussion
Previous studies based on single cohorts reported that anthracycline exposure may increase the risk of SBC, but had relatively smaller sample sizes and case numbers7,11–15. We are able to estimate precise dose thresholds for doxorubicin and identify the effects of other types of anthracyclines on SBC risk in a pooled cohort analysis of large numbers of childhood cancer survivors. These pooled analyses demonstrate a relationship between increasing cumulative doxorubicin dose and SBC risk, as well as an association between epirubicin exposure (yes versus no) and an increased SBC risk. We observed that treatment with doxorubicin increases SBC risk both in survivors who received chest radiotherapy and in survivors treated without chest radiotherapy. Furthermore, the joint effects between doxorubicin and chest radiotherapy appear to be additive. In addition, our results did not show a statistically significant association between daunorubicin and increased SBC risk.
The mechanisms underpinning anthracycline-related SBC risk have not been elucidated. Known mutagenic properties of anthracycline agents that might contribute to SBC risk include topoisomerase II inhibition, DNA intercalation, oxidative stress and chromatin damage17. In regard to our identification of differential risk between different anthracycline agents, potential differences in the mechanisms of developing subsequent neoplasms are unclear. Animal studies indicate that both doxorubicin and daunorubicin can induce mammary tumors18,19. The antineoplastic properties of doxorubicin and daunorubicin have both been assumed to result from DNA damage and chromatin damage17, and based on limited studies, the anticancer efficacies are thought to be similar20,21. Evidence from murine models and human cells suggests that chemically separating those activities by reducing the DNA damage effect while retaining chromatin damage could detoxify the anthracycline variants while maintaining anticancer efficacy22. A possible factor that might underlie the differences in dose effects observed between doxorubicin and daunorubicin is the lower number of individuals and SBC cases among those exposed to daunorubicin, which might have limited power to detect a substantial dose–response relationship. For epirubicin (nine SBC cases exposed), we identified an association with SBC increased risk, but for idarubicin (one SBC case exposed), the number of cases was too low. Future experimental and animal studies that elucidate mechanisms underlying breast carcinogenicity among the various anthracycline agents are warranted.
Childhood cancer treatments often feature multimodality regimens23, which challenge the elucidation of joint and independent effects of different treatments. Our study provides evidence of the joint effects of chest radiotherapy and individual anthracycline agents. Our findings indicate that the joint effects of doxorubicin and chest radiation are submultiplicative and compatible with additive effects, which implies that the combined effects of doxorubicin and chest radiation are not equal to the product of their individual effects, but to the sum of their individual effects. A previous CCSS case–control study showed that the joint effects of radiotherapy dose to the breast and anthracycline exposure (yes/no) were more than additive14. However, they did not investigate the interaction between individual anthracycline agents and chest radiotherapy, and further comparison between the studies is difficult because the CCSS study used a case–control design with estimated radiation dose to breast cancer location.
We did not observe a statistically significant reduction of SBC risk among survivors with radiotherapy delivered to the pelvic region (≥ 5 Gy versus no pelvic radiotherapy or <5 Gy, as an indicator of ovarian dose) in our entire cohort (Table 2), which aligns with a SJLIFE study (pelvic radiotherapy yes versus no) that was also included in our pooled cohort13. However, when we restricted our analyses to survivors who received chest radiotherapy (Extended Data Table 6), we found a decreased SBC risk for pelvic radiotherapy, consistent with previous reports that showed reduced SBC risk associated with absorbed ovarian radiation dose ≥5 Gy in survivors treated with chest radiation, likely due to suppression of hormonal stimulation of breast tissue16.
Extended Data Table 6.
Multivariable Cox proportional hazard regression analyses for subsequent breast cancer in female five-year childhood cancer survivors stratified by chest radiotherapy status (yes/no)
Some limitations should be taken into account when interpreting our study findings. For SIR/EAR and cumulative incidence analyses, one should be cautious with interpreting differences between categories, as there might be differences in the duration of follow-up and pelvic radiotherapy exposure between the categories. As we did not have complete data on treatments for subsequent malignant tumors before SBC (66 survivors had subsequent malignant tumors before SBC diagnosis), we were not able to explore the effects of those treatments on SBC risk. However, our sensitivity analyses censoring at the time of the first subsequent malignant tumor (Extended Data Table 5b) were consistent with the results in our main analyses. Our results of a 1.7-time increased risk of SBC compared to the general female population for survivors who received neither chest radiotherapy nor doxorubicin (Extended Data Table 2) suggest that other factors, such as genetic predisposition, may also have a role. As we had incomplete information on genetic cancer predispositions in our study, we were not able to evaluate genetic effects and possible gene–treatment interactions. The SJLIFE study, however, demonstrated that anthracycline effects are independent of cancer predisposition gene mutations13. Future studies with germline genetic sequencing data may help to further elucidate the interplay of genetic modifiers and individual chemotherapeutic agent exposure on SBC risk. Furthermore, we did not have information on some other SBC risk factors, such as unhealthy lifestyle (for example, low-level physical activity, obesity and excessive alcohol use) and reproductive history, which could potentially lead to some degree of confounding bias. Our analyses did not identify associations between gonadotoxic therapies (pelvic radiotherapy and alkylating agents as proxies for reproductive history) and SBC risk, making it unlikely that reproductive factors are strong confounders. Also, most lifestyle factors are not very likely to be associated with anthracycline exposure. This has also been described in a previous study showing that anthracyclines were not associated with being insufficiently active or having high-risk health behaviors24. Therefore, we assume that the risk of confounding caused by lifestyle factors is very low. However, it is important to evaluate this in future studies.
According to the current IGHG breast cancer screening guideline, survivors with a relative risk more than two times higher than survivors not exposed to a specific treatment are considered to be at moderate or high risk for SBC. Recommendations for SBC screening in survivors are based on these risk levels8. The current IGHG guideline was not able to formulate SBC screening recommendations for survivors treated with anthracyclines because there was inconsistent evidence on dose thresholds for classifying survivors as moderate or high risk and no data on possible dose–effect differences in risks for the different individual anthracycline agents. In our study, we observed a more than two times higher risk of SBC for survivors treated with ≥200 mg m−2 cumulative doxorubicin dose compared to the no doxorubicin treatment. Given that trends in childhood cancer treatments include reduced use of chest radiation therapy doses and increased exposure to anthracyclines since the 1970s7, our findings support that early initiation of breast cancer surveillance may be reasonable for childhood cancer survivors who have received ≥200 mg m−2 cumulative doxorubicin dose. We believe that these findings should be implemented in an update of the SBC surveillance guideline for survivors. Our study also provided insufficient information on the dose–response relation of epirubicin on SBC risk to advise on screening recommendations for this anthracycline agent.
In conclusion, doxorubicin is associated with a dose-dependent increase of SBC, both in women treated with and without chest radiotherapy. Epirubicin is also associated with an increased SBC risk. Our findings support that it may be reasonable to initiate early breast cancer screening in female childhood cancer survivors who have received ≥200 mg m−2 cumulative doxorubicin dose. We believe that the results of our study should be considered in updates of the SBC surveillance guidelines for survivors and can inform future treatment protocols for newly diagnosed childhood cancer patients.
Methods
Study population
We pooled data from five cohort studies (CCSS (9,671 women diagnosed in period 1970–1999), SJLIFE (2,236 women diagnosed in period 1962–2012), DCCSS-LATER (2,237 women diagnosed in period 1963–2001), FCCSS (3,415 women diagnosed in period 1943–2000) and Dutch Hodgkin Late Effects cohort (265 women diagnosed in period 1965–1995)) and one case–cohort study (SCCSS, 79 women diagnosed in period 1976–2007) in Europe and North America with available data on radiotherapy cumulative dose and fields and cumulative dose for chemotherapy (Fig. 1). Details of the study design and methodology have been previously described25. Briefly, eligibility criteria included a primary cancer diagnosis at <21 years of age, survival ≥5 years from primary cancer diagnosis, follow-up data on the presence and type of subsequent primary neoplasms.
Ethics approval
The contributing cohort study teams obtained institutional review board and/or Ethics Committee approval or exemption in their respective contributing institute (CCSS: The St. Jude Children’s Research Hospital Institutional Review Board (ref. 021289), SJLIFE: The St. Jude Children’s Research Hospital Institutional Review Board (ref. 021898); DCCSS-LATER: Medical Ethical Committee Academic Medical Center, Amsterdam (ref. MEC 08/2014); FCCSS: ethics committee of the INSERM (ref. 12-077); Dutch Hodgkin Late Effects cohort: the NKI-AVL Institutional Review Board (ref. IRBd20-155); SCCSS: the cantonal ethics committee Bern (ref. KEK BE 166/2014 and KEK BE 183/11)). The pooling effort is exempt from review in compliance with Dutch law and regulations for health research involving human beings. Data sharing agreements between the Princess Máxima Center for Pediatric Oncology and all data providers are in place. Written consent was obtained from all patients of the CCSS and the SJLIFE. Specific informed consent for retrospective data collections for selected groups of patients for the DCCSS-LATER, the Dutch Hodgkin Late Effects cohort, the FCCSS and the SCCSS cohorts was waived in accordance with the country’s legislation.
Ascertainment of treatment information and SBC diagnosis
For each patient in the individual cohorts, diagnostic information of the childhood cancer and treatment details of primary cancer and recurrences were ascertained by medical record abstraction25. CED was calculated and used as the cumulative exposure of alkylating agents26. Radiotherapy fields involving the chest, collectively referred to as ‘chest radiotherapy’ included whole lung, TBI, mantle, mediastinal and other chest-exposing fields (for example, axilla and spine). Pelvic radiotherapy included any field involving the pelvis, including TBI.
Methods for ascertainment and validation of SBC differed among the included cohorts. The study teams applied various combinations of cancer registry linkage, self-reported survey data with medical record validation for survivors who reported SBC and/or information extracted from pathology reports or medical records (Supplementary Table 6). Details regarding cohort-specific methodology for definitions of treatment exposures and subsequent tumor ascertainment were reported previously25. Vital status was ascertained by linkages to national death registries and/or by medical records.
Statistical analysis
Childhood cancer survivors were considered at risk for developing SBC from 5 years after a primary cancer diagnosis until the date of the first SBC, death or the date of the last follow-up observation, whichever occurred first.
The incidence of SBC in the pooled cohort was compared with the general female population using country-specific incidence rates of the Cancer Incidence in Five Continents database (CI5, https://ci5.iarc.fr/), a database from the International Agency for Research on Cancer containing information from cancer registries worldwide27. Because no French nationwide incidence data were available from the CI5, we used data from the French cancer registry network Francim for the incidence rate of breast cancer in France28–30. SIRs were calculated as the ratio of the observed number of SBC to the expected number of female breast cancers. Expected numbers were estimated by accumulating cohort-specific person-years at risk by country, age (5-year bands), and calendar year (1-year bands)-specific strata and multiplying by the corresponding female breast cancer incidence rates in the general population. EARs were calculated as the differences between observed and expected numbers of female breast cancer per 1,000 person-years at risk. Because population-based breast cancer incidence rates only include invasive tumors, we considered the first invasive breast cancer as an event for these analyses. Cumulative incidences of SBC overall and by treatment subgroups were calculated, treating death as a competing risk.
Multivariable Cox proportional hazards regression analyses, stratified by cohort, were used to compute HRs and 95% CIs of SBC, either invasive breast cancer or DCIS, according to treatment exposure categories using a one-stage approach and stratifying the analyses by cohort. Attained age was used as the time scale31. Weights were applied to account for the case–cohort data from the SCCSS and for the under-sampling of acute lymphoblastic leukemia cases in the CCSS data. The proportional hazards assumption was checked with scaled Schoenfeld residuals in Cox models; it was not violated. The base multivariable model included specific anthracycline agents, age at primary cancer diagnosis, the combination of the chest radiation field and its associated maximum dose, pelvic radiation dose ≥5 Gy and alkylating agent CED exposure, all of which have been shown or suggested to be associated with breast cancer risk in previous studies6,11,16,32,33. We modeled cumulative doxorubicin dose and daunorubicin dose (categories by steps of 100 mg m−2 to ≥400 mg m−2 for doxorubicin dose and to ≥200 mg m−2 for daunorubicin dose due to statistical power reasons, respectively, and continuously per 100 mg m−2 increase) and epirubicin (yes/no); this proved infeasible for idarubicin owing to limited numbers of females treated with this agent. We first categorized chest radiotherapy as the combination of each eligible radiation field (defined above) with the associated maximum chest radiotherapy dose below or above the median categorized as low-dose or high-dose, respectively. Because results were comparable for fields with similar levels of potential radiation exposure to the breast, we categorized chest radiotherapy as follows: no chest radiotherapy, high-dose mantle, low-dose mantle, mediastinal, TBI, whole lung and other. Because there is only evidence for associations between anthracyclines and alkylating agents on SBC risk, we applied the following selection procedure to evaluate other chemotherapeutic agents: we added binary indicators for epipodophyllotoxins, vinca alkaloids, platinum compounds and antimetabolites to the base model. If addition of each variable changed any HRs of cumulative doxorubicin and/or daunorubicin dose by >10% compared to a model without the variable, it was included in the final models (Supplementary Table 7). Our final multivariable analyses did not include any of the additional classes of chemotherapeutic agents indicated above.
Interaction between cumulative doxorubicin/daunorubicin doses and chest radiotherapy and age at primary childhood cancer diagnosis on a multiplicative scale was evaluated by comparing models with and without the interaction term via likelihood ratio tests. Aalen’s additive hazard models were applied to evaluate the interaction of cumulative doxorubicin/daunorubicin exposures and chest radiotherapy and age at primary childhood cancer diagnosis on an additive scale34.
A series of prespecified sensitivity analyses were conducted by applying the same regression models to the data with (a) outcome restricted to invasive breast cancer to exclude DCIS, which does not always progress to invasive breast cancer; (b) censoring at the time of the first non-SBC subsequent malignant neoplasm diagnosis to rule out effects of treatments for those tumors; (c) excluding 444 survivors treated for childhood cancer before 1970 to exclude a potentially influential group of women who reached comparatively high attained age yet showing deviating characteristics owing to improvements in clinical practice and survival trends since the 1970s; (d) excluding survivors treated for Hodgkin lymphoma to exclude patients who generally received extensive radiotherapy fields to the chest (Extended Data Table 5) (e) excluding each cohort on a one-by-one basis to evaluate robustness of findings (Supplementary Table 4) and each cohort to evaluate between-cohort differences (Supplementary Table 5).
All analyses were conducted in R software (version 4.0.3). A P value of <0.05 was considered statistically significant in two-sided statistical tests.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Online content
Any methods, additional references, Nature Portfolio reporting summaries, source data, extended data, supplementary information, acknowledgements, peer review information; details of author contributions and competing interests; and statements of data and code availability are available at 10.1038/s41591-023-02514-1.
Supplementary information
Supplementary Tables 1–7.
Acknowledgements
This work was supported by the Children Cancer Free Foundation (KiKa; grant 325 title: risk factors for female breast cancer after treatment for childhood and adolescent cancer: individual patient data analyses of an internationally pooled cohort) to C.M.R., F.E.v.L., L.C.M.K. (principal investigators). The CCSS and SJLIFE cohorts are supported by the National Cancer Institute (CA55727 to G.T.A. (principal investigator) and CA195547 to M.M.H. and K.K.N. (principal investigators)) as well as support to St. Jude Children’s Research Hospital also provided by the Cancer Center Support (CORE) grant (CA21765 to C.R. (principal investigator)) and the American Lebanese-Syrian Associated Charities (ALSAC). The NWTSG was supported by the National Cancer Institute (CA054498 to N.E.B. and W.L. (principal investigator)). The DCCSS-LATER is supported by the Dutch Cancer Society (DCOG2011-5027 to C.M.R., F.E.v.L. and W.T. (principal investigators) and UVA2012-5517 to C.M.R. and L.C.M.K. (principal investigators)). The FCCSS is funded by the Fondation ARC (PopHARC Grant to F.d.V. (principal investigator)), the Agence Nationale pour la Recherche Médicale (ANR, Hope-Epi Grant to F.d.V. (principal investigator)), and the Gustave Roussy Foundation (Pediatric Program ‘Guérir le Cancer de l’Enfant’ to F.d.V. (principal investigator)). The SCCSS has been supported by the Swiss Cancer League and the Swiss Cancer Research foundation (KFS-02783-02-2011, KLS-3412-02-2014, KFS-4157-02-2017, KLS/KFS-4825-01-2019, KFS-4722-02-2019, KFS-5027-02-2020, KFS-5302-02-2021 and KLS-5432-08-2021 to C.E.K. (principal investigator)), Kinderkrebs Schweiz (www.kinderkrebs-schweiz.ch) and Kinderkrebshilfe Schweiz (www.kinderkrebshilfe.ch). The Dutch Hodgkin Late Effects cohort has been supported by the Dutch Cancer Society (NKI 2010-4720 to F.E.v.L. (principal investigator)).
Extended data
Author contributions
Y.W., J.C.T., L.C.M.K., C.M.R. and F.E.v.L. designed and conceptualized the study. J.C.T., L.C.M.K., C.M.R. and F.E.v.L. acquired funding and supervised the project. G.T.A., M.M.H., W.L., F.v.D., L.C.M.K., F.E.v.L. and C.E.K. provided administrative, technical or material support. Y.W., J.C.T., C.S.M. and W.L. performed the statistical analyses. Y.W., J.C.T., L.C.M.K., C.M.R. and F.E.v.L. drafted the manuscript. All authors contributed to data interpretation and manuscript revision and approved the final version for publication.
Peer review
Peer review information
Nature Medicine thanks Joachim Schuz, Giuseppe Curigliano and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ming Yang, in collaboration with the Nature Medicine team.
Data availability
The International Consortium for Pooled Studies on Subsequent Malignancies after Childhood and Adolescent Cancer database is not an open-access database due to ethical and data protection constraints. The pseudonymized data are managed by the Princess Máxima Center for Pediatric Oncology in the Netherlands and cannot be shared with investigators outside the institute without consent from all involved parties. However, potential collaborators are welcome to submit proposals to J.C.T. (j.c.teepen@prinsesmaximacentrum.nl), which will be considered by the consortium. The consortium will come up with a decision about submitted proposals within 3 months after application. The country-specific female breast cancer rates of the Cancer Incidence in Five Continents database were used as the general female population in our study. These data are publicly available: https://ci5.iarc.fr/.
Code availability
The statistical code for our main analyses is available at https://github.com/yuehanwanggitub/IPDanthracyclines.git.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of authors and their affiliations appears at the end of the paper.
Contributor Information
Yuehan Wang, Email: y.wang-2@prinsesmaximacentrum.nl.
The International Consortium for Pooled Studies on Subsequent Malignancies after Childhood and Adolescent Cancer:
Flora E. van Leeuwen, Florent de Vathaire, Helena J. H. van der Pal, Nadia Haddy, Ibrahima Diallo, K. Scott Baker, Amy Berrington de González, Miriam R. Conces, Louis S. Constine, Mike Hawkins, Jacqueline J. Loonen, Marloes Louwerens, Geert O. Janssens, Lene Mellemkjaer, Raoul Reulen, and Jeanette F. Winther
Extended data
is available for this paper at 10.1038/s41591-023-02514-1.
Supplementary information
The online version contains supplementary material available at 10.1038/s41591-023-02514-1.
References
- 1.Meadows AT, et al. Second neoplasms in survivors of childhood cancer: findings from the childhood cancer survivor study cohort. J. Clin. Oncol. 2009;27:2356–2362. doi: 10.1200/JCO.2008.21.1920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Reulen RC, et al. Long-term cause-specific mortality among survivors of childhood cancer. JAMA. 2010;304:172–179. doi: 10.1001/jama.2010.923. [DOI] [PubMed] [Google Scholar]
- 3.Armstrong GT, et al. Reduction in late mortality among 5-year survivors of childhood cancer. N. Engl. J. Med. 2016;374:833–842. doi: 10.1056/NEJMoa1510795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yeh JM, et al. Life expectancy of adult survivors of childhood cancer over 3 decades. JAMA Oncol. 2020;6:350–357. doi: 10.1001/jamaoncol.2019.5582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bhatia S, et al. Breast cancer and other second neoplasms after childhood Hodgkin’s disease. N. Engl. J. Med. 1996;334:745–751. doi: 10.1056/NEJM199603213341201. [DOI] [PubMed] [Google Scholar]
- 6.Moskowitz CS, et al. Breast cancer after chest radiation therapy for childhood cancer. J. Clin. Oncol. 2014;32:2217–2223. doi: 10.1200/JCO.2013.54.4601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Henderson TO, et al. Association of changes in cancer therapy over 3 decades with risk of subsequent breast cancer among female childhood cancer survivors: a report from the childhood cancer survivor study (CCSS) JAMA Oncol. 2022;8:1765–1774. doi: 10.1001/jamaoncol.2022.4649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mulder RL, et al. Updated breast cancer surveillance recommendations for female survivors of childhood, adolescent, and young adult cancer from the international guideline harmonization group. J. Clin. Oncol. 2020;38:4194–4207. doi: 10.1200/JCO.20.00562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ren W, Chen M, Qiao Y, Zhao F. Global guidelines for breast cancer screening: a systematic review. Breast. 2022;64:85–99. doi: 10.1016/j.breast.2022.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Turcotte LM, et al. Temporal trends in treatment and subsequent neoplasm risk among 5-year survivors of childhood cancer, 1970–2015. JAMA. 2017;317:814–824. doi: 10.1001/jama.2017.0693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Henderson TO, et al. Breast cancer risk in childhood cancer survivors without a history of chest radiotherapy: a report from the childhood cancer survivor study. J. Clin. Oncol. 2016;34:910–918. doi: 10.1200/JCO.2015.62.3314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Teepen JC, et al. Long-term risk of subsequent malignant neoplasms after treatment of childhood cancer in the DCOG LATER study cohort: role of chemotherapy. J. Clin. Oncol. 2017;35:2288–2298. doi: 10.1200/JCO.2016.71.6902. [DOI] [PubMed] [Google Scholar]
- 13.Ehrhardt MJ, et al. Subsequent breast cancer in female childhood cancer survivors in the St Jude Lifetime Cohort Study (SJLIFE) J. Clin. Oncol. 2019;37:1647–1656. doi: 10.1200/JCO.18.01099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Veiga LH, et al. Association of breast cancer risk after childhood cancer with radiation dose to the breast and anthracycline use: a report from the childhood cancer survivor study. JAMA Pediatr. 2019;173:1171–1179. doi: 10.1001/jamapediatrics.2019.3807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Turcotte LM, et al. Chemotherapy and risk of subsequent malignant neoplasms in the childhood cancer survivor study cohort. J. Clin. Oncol. 2019;37:3310–3319. doi: 10.1200/JCO.19.00129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Inskip PD, et al. Radiation dose and breast cancer risk in the childhood cancer survivor study. J. Clin. Oncol. 2009;27:3901–3907. doi: 10.1200/JCO.2008.20.7738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.van der Zanden SY, Qiao X, Neefjes J. New insights into the activities and toxicities of the old anticancer drug doxorubicin. FEBS J. 2021;288:6095–6111. doi: 10.1111/febs.15583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bucclarelli E. Mammary tumor induction in male and female Sprague-Dawley rats by adriamycin and daunomycin. J. Natl Cancer Inst. 1981;66:81–84. [PubMed] [Google Scholar]
- 19.Solcia E, Ballerini L, Bellini O, Sala L, Bertazzoli C. Mammary tumors induced in rats by adriamycin and daunomycin. Cancer Res. 1978;38:1444–1446. [PubMed] [Google Scholar]
- 20.Buckley JD, et al. Remission induction in children with acute non-lymphocytic leukemia using cytosine arabinoside and doxorubicin or daunorubicin: a report from the Childrens Cancer Study Group. Med. Pediatr. Oncol. 1989;17:382–390. doi: 10.1002/mpo.2950170507. [DOI] [PubMed] [Google Scholar]
- 21.Kaspers GL, et al. In vitro cytotoxicity of mitoxantrone, daunorubicin and doxorubicin in untreated childhood acute leukemia. Leukemia. 1994;8:24–29. [PubMed] [Google Scholar]
- 22.Qiao X, et al. Uncoupling DNA damage from chromatin damage to detoxify doxorubicin. Proc. Natl Acad. Sci. USA. 2020;117:15182–15192. doi: 10.1073/pnas.1922072117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Erdmann F, et al. Childhood cancer: survival, treatment modalities, late effects and improvements over time. Cancer Epidemiol. 2021;71:101733. doi: 10.1016/j.canep.2020.101733. [DOI] [PubMed] [Google Scholar]
- 24.Lown EA, et al. Patterns and predictors of clustered risky health behaviors among adult survivors of childhood cancer: a report from the childhood cancer survivor study. Cancer. 2016;122:2747–2756. doi: 10.1002/cncr.30106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang Y, et al. Cohort profile: risk and risk factors for female breast cancer after treatment for childhood and adolescent cancer: an internationally pooled cohort. BMJ Open. 2022;12:e065910. doi: 10.1136/bmjopen-2022-065910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Green DM, et al. The cyclophosphamide equivalent dose as an approach for quantifying alkylating agent exposure: a report from the childhood cancer survivor study. Pediatr. Blood Cancer. 2014;61:53–67. doi: 10.1002/pbc.24679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bray, F. et al. Cancer Incidence in Five Continents (Electronic Version) Vol. 11 (International Agency for Research on Cancer, 2017).
- 28.Binder-Foucard F, et al. Cancer incidence and mortality in France over the 1980–2012 period: solid tumors. Rev. Epidemiol. Sante Publique. 2014;62:95–108. doi: 10.1016/j.respe.2013.11.073. [DOI] [PubMed] [Google Scholar]
- 29.Defossez G, et al. Cancer incidence and mortality trends in France over 1990–2018 for solid tumors: the sex gap is narrowing. BMC Cancer. 2021;21:726. doi: 10.1186/s12885-021-08261-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ménégoz F, et al. Cancer incidence and mortality in France in 1975–95. Eur. J. Cancer Prev. 1997;6:442–466. doi: 10.1097/00008469-199710000-00005. [DOI] [PubMed] [Google Scholar]
- 31.Yasui Y, et al. A methodological issue in the analysis of second-primary cancer incidence in long-term survivors of childhood cancers. Am. J. Epidemiol. 2003;158:1108–1113. doi: 10.1093/aje/kwg278. [DOI] [PubMed] [Google Scholar]
- 32.Swerdlow AJ, et al. Breast cancer risk after supradiaphragmatic radiotherapy for Hodgkin’s lymphoma in England and Wales: a National Cohort Study. J. Clin. Oncol. 2012;30:2745–2752. doi: 10.1200/JCO.2011.38.8835. [DOI] [PubMed] [Google Scholar]
- 33.Moskowitz CS, et al. Radiation-associated breast cancer and gonadal hormone exposure: a report from the childhood cancer survivor study. Br. J. Cancer. 2017;117:290–299. doi: 10.1038/bjc.2017.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rod NH, Lange T, Andersen I, Marott JL, Diderichsen F. Additive interaction in survival analysis: use of the additive hazards model. Epidemiology. 2012;23:733–737. doi: 10.1097/EDE.0b013e31825fa218. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Tables 1–7.
Data Availability Statement
The International Consortium for Pooled Studies on Subsequent Malignancies after Childhood and Adolescent Cancer database is not an open-access database due to ethical and data protection constraints. The pseudonymized data are managed by the Princess Máxima Center for Pediatric Oncology in the Netherlands and cannot be shared with investigators outside the institute without consent from all involved parties. However, potential collaborators are welcome to submit proposals to J.C.T. (j.c.teepen@prinsesmaximacentrum.nl), which will be considered by the consortium. The consortium will come up with a decision about submitted proposals within 3 months after application. The country-specific female breast cancer rates of the Cancer Incidence in Five Continents database were used as the general female population in our study. These data are publicly available: https://ci5.iarc.fr/.
The statistical code for our main analyses is available at https://github.com/yuehanwanggitub/IPDanthracyclines.git.