Abstract

Lack of horizontal and vertical bone at the site of an implant can lead to significant clinical problems that need to be addressed before implant treatment can take place. Guided bone regeneration (GBR) is a commonly used surgical procedure that employs a barrier membrane to encourage the growth of new bone tissue in areas where bone has been lost due to injury or disease. It is a promising approach to achieve desired repair in bone tissue and is widely accepted and used in approximately 40% of patients with bone defects. In this Review, we provide a comprehensive examination of recent advances in resorbable membranes for GBR including natural materials such as chitosan, collagen, silk fibroin, along with synthetic materials such as polyglycolic acid (PGA), polycaprolactone (PCL), polyethylene glycol (PEG), and their copolymers. In addition, the properties of these materials including foreign body reaction, mechanical stability, antibacterial property, and growth factor delivery performance will be compared and discussed. Finally, future directions for resorbable membrane development and potential clinical applications will be highlighted.
Keywords: Guided bone regeneration, Barrier membranes, Tissue engineering, Biomaterials, Biocompatibility
Introduction
Aging, congenital diseases, cancer, trauma, tooth loss, and periodontal disease are considered as common causes of bone loss and disorders in our maxillofacial region. Lack of horizontal and vertical bone in the site of an implant may lead to major clinical problems and needs to be augmented prior to implant treatment.1 To achieve desirable repair in bone tissue, methods such as guided bone regeneration (GBR), distraction osteogenesis, and split-crest surgery have been applied by clinicians. Among all methods, GBR has been widely accepted and used in approximately 40% of patients with bone defects.2 GBR is a surgical procedure that uses a barrier membrane to encourage the growth of new bone tissue in areas where bone has been lost due to injury or disease (Figure 1a).3 The barrier membrane acts as a physical barrier to prevent the ingrowth of soft tissue, such as fibrolasts, while allowing bone cells to migrate into the area and form new bone. The goal is to block and exclude the fast migration of epithelial cells and fibroblasts to the defect site by placing a membrane between hard tissue and soft tissue.4 GBR is commonly used in dental implant surgery, where it is used to increase the amount of bone available to support the implant. It can also be used to repair bone defects caused by periodontal disease or traumatic injury. The procedure typically involves making a small incision in the soft tissue, creating a space for the barrier membrane and bone graft material and then suturing the tissue closed (Figure 1b).5,6 Recovery time can vary depending on the extent of the bone loss and the specific procedure used.3
Figure 1.

(a) Schematic illustrations of the principle of guided bone regeneration (GBR)218 and (b) the combination of guided tissue regeneration (GTR) and bone graft techniques for periodontal defects management.219 (c) Illustration of the cellular and molecular cascades occur during GBR process.218 Reprinted with permission from ref (218). Copyright 2017 John Wiley & Sons. Adapted with permission from ref (219). Copyright 2010 Elsevier.
To ensure a successful GBR, four biologic principles must be achieved as follows: “primary wound closure” to establish uninterrupted wound healing, “exclusion of Epithelium and connective tissue” without interfering with angiogenesis to provide enough blood supply, “space maintenance” to provide suitable space for bone regeneration, and “stability” in the fibrin clot and implanted material to prevent disturbance in the healing process.7
Osseous regeneration, the process of growing new bone tissue to fill in areas of bone loss, relies on the migration of osteogenic cells to the defect region (Figure 1c).8,9 This can be achieved through the use of various techniques and materials including the usage of growth factors.10 Growth factors are substances that promote the growth and differentiation of cells such as bone cells. They can be derived from various sources, including human or animal tissue, or artificially created.11 They are used to enhance the body’s natural healing process and accelerate the growth of new bone tissue. The GBR procedure aims to create an environment that is conducive to bone regeneration by isolating the bone defect from surrounding soft tissue and providing the necessary growth factors and bone graft materials.12 Once the new bone tissue has formed, it integrates with the surrounding natural bone, providing a stable foundation for dental implants and other restorations.
After GBR surgery, a sequence of events leads to bone regeneration: Immediately after the surgery, the body’s immune system responds by initiating an inflammatory response.13 This process is necessary for removing debris and bacteria and recruiting cells needed for healing. During the first 24 h, the grafted space would become filled with blood clots which are enriched by growth factors and chemoactive agents such as cytokines. After the blood clot absorption, cells called osteoblasts and osteoclasts start to migrate to the bone defect site. Mesenchymal stem cells (MSCs) with osteogenic differentiation capability would be transported to this area via newly formed vessels and contribute to forming osteoid structures (Figure 1c).14−16 Osteoblasts and osteoclasts are pivotal cells involved in bone formation and resorption, respectively. Upon injury, these cells proliferate and begin to form a clot, which serves as a scaffold for new bone formation.
Barrier Membranes
Barrier membranes, which are used to separate the bone defect from surrounding soft tissue,17,18 are considered a key element in GBR as they create a physical barrier that prevents the ingrowth of unwanted cells such as gingival tissue (Figure 2a).19 They also serve as scaffolds for the migration of osteogenic cells, which are responsible for forming new bone. Meanwhile, according to recent research focused on the biological processes occurring within membranes, it has been suggested that these membranes may have the potential to actively participate in the regeneration of bone in areas where there are defects.20 Five main criteria have been identified to describe an ideal barrier membrane intended for GBR:3 biocompatibility, space maintenance, occlusive functionality, easy handling, and bioactivity (Figure 2b).21 Several generations of barrier membranes have been developed for use in GBR procedures. The first-generation membranes are made of highly purified, synthetic materials such as polytetrafluoroethylene (PTFE), offering great resistance to displacement, excellent mechanical stability, and long-term stability. However, they are nonresorbable, which means a second step of surgery is required for membrane removal, adding extra cost and burden for the patient.22 Yet another constraint is the excessive rigidity of these materials, which may result in the separation of soft tissue and subsequent membrane exposure, potentially leading to complications.23
Figure 2.
(a) Schematic illustration of the placement of an electrospun barrier membrane for GBR in scenarios involving periodontitis and dental loss. Reprinted with permission from ref (19). Copyright 2020 Elsevier. (b) Five main criteria to define practical barrier membranes for GBR.
Based on these facts, the second generation of barrier membranes, including naturally sourced and synthetic absorbable membranes, has been developed to overcome the shortcomings of nonabsorbable membranes.24,25 The former is fabricated from natural biodegradable biomaterials such as chitosan, collagen, gelatin, silk etc., as it is desirable for an implanted membrane to be gradually replaced by the patient’s own tissue during the recovery phase.3 However, poor mechanical stability and limited time of barrier functionality due to the resorption process are their main drawbacks, restricting their real-world applications. The latter can be tailored to have specific mechanical and degradation properties, allowing customization to suit different tissue types and healing rates. They also eliminate the need for a second surgical procedure to remove the membrane, reducing patient discomfort and healthcare costs.
The third generation involves the use of membranes produced via tissue engineering, as previously proposed by Sam and Pillai26 as multifunctional membranes that not only serve as barriers but also enable the controlled release of specific agents such as antibiotics, growth factors, and adhesion factors at the wound site, that could help facilitate and regulate the natural wound healing process in a more effective manner. Later, Lee et al.27 emphasized on the need to move beyond the fundamental principles of GBR by introducing this generation of membranes that incorporate the release of growth factors and antibiotics In fact, in the third generation, resorbable membranes and bioactive agents such as antibiotics, growth factors, and adhesion factors are combined to enhance cell migration, proliferation, differentiation, and extracellular matrix deposition, leading to faster and more efficient tissue regeneration.28 It can also provide a controlled release of growth factors, which can reduce the required dose and minimize potential side effects. The growth factors can be either incorporated into the membrane matrix during the manufacturing process or loaded onto the membrane surface after fabrication.6,29−,37 However, these newly developed membranes are relatively more expensive than other types of membranes.
Natural Resorbable Membranes
Natural resorbable barrier membranes are biological materials designed to be used in medical procedures and dissolve or break down over time in the body. They are made from natural materials such as collagen, gelatin (which is derived from collagen), chitosan, silk, etc38 and are used in a variety of applications, including in wound healing, in tissue engineering, and as a barrier in GBR procedures, to separate the bone defect from the surrounding soft tissue and promote bone formation (Figure 3).39
Figure 3.

Classification of resorbable membranes based on their natural or synthetic origins in the outer layer. The inner layer displays the current substances used for bioactivity enhancement.
Collagen
Collagen’s etymology originates from “colla”, meaning glue in Greek and “gene”, meaning producing or generating.40 Collagens are the most abundant structural proteins in mammals (30% of total protein mass),41 comprising 28 types and at least 46 polypeptide chains in vertebrates.42 One-third of the total protein in humans and 75% of the dry weight of the skin is composed of collagen, making it the most ubiquitous component in the extracellular matrix (ECM).43 The discovery of collagen dates back to the 1890s when Mall and Young produced gelatin by boiling reticular tissue in water. A decade later, Siegfried found that reticular tissue split up into gelatin,44 and later, in 1904, Tebb proved that reticulin is a type of fiber in connective tissue artifact from collagen.45
Collagen consists of three parallel left-handed polyproline type II polypeptide strands called α chains. These triple helix polypeptides stagger by one residue to form a unique right-handed triple helix structure,46 presented in Figure 4a. ECM contains many classes of collagenous structures, including fibrils, beaded filaments, anchoring fibrils, and networks. Most collagen fibrils are heterotypic, consisting of at least two different fibrillar collagen types. In cartilage, collagen fibrils are largely made of collagen type II cross-linked with collagen types XI, IX, and III.47 In the skin, the most abundant types of collagens are types I and III, while in the cornea they are types I and IV.48 The main sources of collagen are the skin, tendon, and cartilage which could be extracted from different animals like calves, mice, fish, chickens, and birds.49 Collagen-based membranes available on the market are biocompatible and biodegradable. However, they suffer from rapid loss of mechanical stability particularly in a liquid or high humidity environment due to their rapid degradation rate.50 In addition, they are relatively expensive with limited supply sources, making it a challenge to apply them in bone regeneration applications.51 Different degradation patterns and rates are reported for the commercially available collagen membranes.52 The resorption rate is associated with collagenic tissue structure, which would be different based on collagen origin (porcine, bovine, etc.) and the organ from which it is derived (such as skin, tendon, pericardium, etc.). In addition, other characteristics, including porosity, thickness, and weight, could influence the resorption rate and degradation pattern.53 Increasing numbers of studies are now focused on enhancing the bioactivity properties, loading the collagen membrane with active organic and inorganic compounds. Organic compounds include growth factors, cytokines, and anti-inflammatory agents. Inorganic compounds such as calcium phosphate substrates and bioceramics have shown promising results.54
Figure 4.
(a) Collagen, (b) chitosan, and (c) and silk fibroin. (a-i) Some sea creatures are exemplary sources for collagen. Reprinted from ref (220). Copyright 2021 American Chemical Society. (a-ii) Hierarchical microstructure of the collagen fiber. Adapted with permission under a Creative Commons Attribution 4.0 International License from ref (221). (a-iii) In the chemical composition of type I bovine collagen, the triple helix formation is composed of three frequently recurring amino acid sequences: (GlyPro/Hyp-Ala). (a-iv) 3D models of the type I bovine collagen: model at the left is highlighted by nitrogen atoms in blue and oxygen atoms in red, while the model at the right is color-coded to emphasize the three chains.222 (a-v) Chemical structure of collagen. Reprinted with permission under a Creative Commons Attribution 4.0 International License from ref (223). Copyright 2021 MDPI. (b-i) Hierarchical structure of the lobster cuticle. Reprinted with permission from ref (224). Copyright 2010 John Wiley & Sons. (b-ii) 3D structure of chitosan. Adapted with permission under a Creative Commons Attribution 4.0 International License from ref (225). (b-iii) Chitosan chemical structure. Reprinted with permission under a Creative Commons Attribution 4.0 International License from ref (226). Copyright 2014 MDPI. (c-i) Structure of Bombyx mori silk227 from the cocoon spun by B. mori silkworm.228 Reprinted with permission from ref (227). Copyright 2018 John Wiley & Sons. Reprinted from ref (228). Copyright 2022 American Chemical Society. Silk fibroin displays two different structures, silk I and silk II, with distinct configurations. Silk I is characterized by an α helix formation, while silk II has an antiparallel β-sheet nanocrystal conformation. Reprinted with permission from ref (227). Copyright 2018 John Wiley & Sons. The 3D modeling229 (c-ii) and chemical structure230 (c-iii) of silk fibroin. Reprinted with permission from ref (229). Copyright 2012 Elsevier. Reprinted with permission from ref (230). Copyright 2021 Springer Nature.
Collagen has gained significant attention for use in GBR. From a biological perspective, collagen is an important component of the ECM and provides a structural scaffold for cell attachment and proliferation. When used in GBR, collagen can promote cell adhesion and proliferation, as well as enhance tissue regeneration and bone formation.55 Additionally, collagen can act as a carrier for various bioactive molecules, such as growth factors, which can further enhance bone regeneration.43 From a material perspective, collagen possesses several desirable properties for GBR, including biocompatibility, biodegradability, and low immunogenicity. Moreover, it can be processed into various forms, including membranes, sponges, and gels, allowing for versatility in its use.49
Chitosan
Chitosan (Figure 4b) is a cationic polymer that is not widely available in the environment but can be extracted from chitin through the process of deacetylation.56 As a biocompatible, biodegradable, nontoxic, antioxidant, anticancer, anti-inflammation, and hemocompatible material chitosan has attracted extensive attention for biomedical applications including tissue regeneration. In addition, chitosan has a unique feature of attracting both negatively and positively charged molecules due to the presence of amino groups and a hydroxyl group in its chemical structure.57 These functional groups can significantly enhance the mechanical and biological properties of chitosan, making it suitable for a wide range of biomedical and pharmaceutical applications. In addition, it could be modified with other bioactive molecules and made into different forms and shapes, including membranes, hydrogels, sponges, and fiber forms.58 There are numerous techniques to produce chitosan-based biomaterials including solvent casting,59−61 compression molding,62,63 freeze-drying,64,65 electrospinning,66,67 dense gas foaming,68,69 and microwave assisted method.70,71 Despite recent advances in developing mechanically stable and biodegradable chitosan-based membranes,72 chitosan is not commonly used as a natural resorbable membrane for GBR yet as more research is needed to evaluate its efficacy and safety.
Chitosan has been reported as a suitable material for GBR applications, has been shown to promote cell adhesion and proliferation, and can stimulate bone formation and regeneration.69 Chitosan also has antibacterial properties, which can be beneficial in preventing infection during the bone healing process.73 From a material perspective, chitosan is biocompatible and biodegradable and has low immunogenicity. Additionally, chitosan can be processed into various forms, such as films, sponges, and microspheres, allowing for versatility in its use.72 However, some challenges associated with the use of chitosan in GBR include poor mechanical properties and limited control over degradation ratea. Despite these challenges, chitosan-based materials have shown potential in promoting bone regeneration and continue to be an area of active research.66
Silk Fibroin
Silk fibroin is a natural polymer derived from Bombyx mori silkworm (Figure 4c) and is considered as an excellent bioinspired material for tissue engineering and bone regeneration applications, both in vitro and in vivo environments.74,75 It has outstanding biocompatibility, biodegradability, oxygen and water vapor permeability, and mechanical and physical properties,76,77 and has been used as a biodegradable scaffold to induce osteogenesis, mineral resorption, and bone regeneration in vivo,78 with minimum inflammatory reaction and great cellular response (comparable to collagen).79 When it degrades, silk fibroin produces nontoxic amino acids which can be easily absorbed in vivo, with a tunable degradation time from a few days to several months.51,80,81 In vivo studies of silk fibroin scaffolds on Lewis rats demonstrated decomposition after 8 weeks and full resorption after 1 year,82 indicating promising biodegradability and bioresorbability for this natural polymer. However, the main disadvantage of using silk fibroin in GBR applications is that the material can become brittle when it dries.83 This can make it difficult to handle and can also reduce its mechanical strength and durability.84 Some strategies to overcome this problem include adding plasticizers, increasing water content, and controlling the manufacturing process in order to eliminate the formation of rigid β-sheet structures.85,86 Additionally, silk fibroin membranes can be sensitive to changes in pH and temperature, which can also affect their mechanical properties.87
Due to its ease of processing, silk fibroin has been fabricated in a wide range of forms and shapes, including hydrogels, mats, films, and sponges, making it an excellent material for GBR application in biomedical and pharmaceutical applications, particularly in dentistry.88,89 In addition, the active sites on the silk surface can act as potential bonding spots for bioactive molecules, such as growth factors and stem cells, facilitating bone regeneration and osteodifferentiation.90 A wide range of manufacturing techniques, such as electrospinning, spin-coating, freeze-drying, micropatterning, and bioprinting, have been introduced to fabricate silk-based biomaterials for tissue engineering and bone regeneration.80 The main challenge, however, is to enhance the osteogenic characteristics of the silk-based material’s GBR application. To tackle this issue, silk-based composite materials made of polymers, hydroxyapatite,78,91,92 magnesium oxide,93,94 graphene,95,96 and antibacterial agents such as silver nanoparticles97,98 and curcumin nanoparticles99,100 were incorporated into silk. Using an electrospinning technique, Ghafouri et al.51 fabricated functionally graded membranes (FGMs) made of electrospun nanofibers of silk fibroin (extracted from sericin protein) and poly(ethylene oxide) (PEO), with fiber diameters ranging from 300 to 550 nm. The membrane layout consisted of two layers, which were mechanically pressed to each other to form an FGM. The loose layer was designed to guide osteogenic cell growth and bone regeneration, while the dense layer provided a better barrier effect, prohibiting epithelial tissue growth into bone defects. The fabricated membranes demonstrated excellent stability and degradability performance in artificial saliva solution, featuring a negligible mass loss of <0.003 g after 4 weeks, with retained bilayer structure after 8 weeks.51
Synthetic Resorbable Membranes
Synthetic resorbable membranes are designed to be biocompatible and to degrade over time, allowing the body to absorb them and promoting the regeneration of new bone tissue. Some examples of synthetic resorbable membranes for GBR include polylactic acid (PLA),101−103 polyglycolide (PGA),3,104 poly(lactic-co-glycolic) acid (PLGA),103,105 polydioxanone (PDO),106,107 polycaprolactone (PCL),108−110 and polyethylene glycol (PEG) membranes.111,112 However, synthetic bioresorbable membranes also suffer from lack of control over the resorption rate dictated by factors like local pH and lack of inertness because of unfavorable reactions during degradation (Figure 3).113
PLA
PLA is a biodegradable polymer used to synthesize barrier membranes for biomedical applications, particularly GBR. It is a synthetic polymer made from repeating units of lactic acid (Figure 5) and is a member of the polyester family. PLA membranes are suitable barriers for preventing the migration of cells from the surrounding soft tissue into the bone defect while allowing for the ingrowth of new bone cells from the surrounding tissue. PLA can be fabricated into different forms, including films, fibers, and meshes, and can be combined with other materials to improve its properties, such as chitosan, hydroxyapatite, or growth factors.114 Additionally, the physical and mechanical properties of PLA can be adjusted by varying the molecular weight and degree of crystallinity, which can improve the handling, stability, and barrier properties of the membrane.25,115 Despite its advantages, PLA has some disadvantages such as a slow degradation rate, which may result in long-term inflammation, and its acidic degradation products and increased risk of foreign body reaction.114,116 To overcome these limitations, studies focused on different modifications and blends of PLA, including copolymers and composite materials, to improve its degradation rate and biocompatibility for GBR applications.17,101
Figure 5.
Chemical formulas of the classical synthesized polymers used as barrier membranes.
PGA
PGA is a biodegradable polymer that has been extensively studied for its use in GBR as a barrier membrane.117,118 It is a synthetic polymer made from repeating units of glycolic acid (Figure 5) and is another member of the polyester family.118 PGA starts to degrade within a few weeks after implantation and is entirely absorbed by the body within a few months. This eliminates the need for removal of the implant and reduces the risk of infection and other complications associated with nondegradable materials.119 PGA has a high tensile strength, which makes it suitable for a wide range of medical applications such as sutures, surgical mesh, scaffolds for tissue engineering, and barrier membrane in GBR.104 When used in combination with other materials, such as collagen or calcium phosphate (Figure 3), PGA can improve its mechanical properties and degradation rate. Studies have demonstrated that PGA-based membranes can promote new bone formation in GBR procedures, and their degradation products have been found to be biocompatible and nontoxic.119 Additionally, it has high thermal stability and a high melting point which makes it suitable for sterilization process.120 However, PGA can cause an inflammatory response in vivo due to its acidic degradation products. To overcome this issue, various modifications of PGA have been explored, such as blending it with other polymers or surface-modifying with hydroxyapatite.118,119
PLGA
PLGA is a biodegradable and biocompatible copolymer commonly used in the medical field for drug delivery and tissue engineering applications. It is composed of repeating units of lactic acid and glycolic acid (Figure 5), which are biodegraded by hydrolysis in the body over time.121,122 The rate of degradation can be controlled by adjusting the ratio of lactic acid to glycolic acid in the copolymer.122 PLGA has several desirable properties, such as good biocompatibility, controlled degradation, and tunable mechanical properties, making it a popular choice for various biomedical applications. For example, its microspheres can be used for sustained drug delivery,123,124 whereas its scaffolds can be used for tissue engineering.125,126 It is a thermoplastic polymer, which means it can be melted and molded into various shapes,127 making it appealing for a range of biomedical applications. In the context of GBR, PLGA has been used as a scaffold material to provide a temporary support structure for the growth of new bone tissue.125,126 The scaffolds serve as physical support and provide a surface for cell attachment and proliferation, which helps to promote the formation of new bone tissue.125 They can be designed to have specific mechanical properties and pore sizes to optimize conditions for bone growth. The degradation rate of the PLGA scaffold can be adjusted to match the rate of new bone growth,128 ensuring that the scaffold is present for the right amount of time to support the growth of new bone tissue.129
PDO
PDO is a biocompatible, absorbable suture material that can also be used in GBR as a barrier membrane, which can be broken down by the body over time. It is a polymer made from repeating units of p-dioxanone (Figure 5) and is also a member of the polyester family.130 PDO sutures are flexible and easy to handle, which makes them suitable for a wide range of surgical procedures, including general surgery, orthopedics, and ophthalmology. PDO sutures start to lose their strength within the first few days after surgery and can be completely absorbed by the body within 4–6 months. PDO is also known for its good handling properties and minimal tissue reaction.131 Additionally, PDO sutures are nonantigenic, which means that they are unlikely to cause an immune response and have minimal tissue reaction, which reduces the risk of infection and wound healing complications.131 PDO has a good balance of mechanical and degradation properties, meaning it has good strength while also breaking down at a suitable rate, making it a promising option for GBR.132 Despite its advantages, PDO has some disadvantages such as high cost and the fact that it can cause some tissue inflammation.133
PCL
PCL is a biodegradable and biobased thermoplastic polymer that has a wide range of uses, including as a biomedical material for controlled drug delivery,134,135 as a bioresorbable suture,136,137 and as a component in 3D printing filaments.138,139 It has a linear, aliphatic polyester structure consisting repeating units of caprolactone, which is a six-carbon ester.140 The chemical formula for PCL is [−(C6H10O2)n−], where n represents the number of repeating units in the polymer chain (Figure 5). The repeating units are connected by ester bonds, and the resulting polymer chain has a flexible and amorphous structure. This structure gives PCL distinctive mechanical properties, such as good toughness, flexibility, and low melting temperature,140 making it an attractive alternative to petroleum-based plastics. From a material perspective, PCL has desirable properties, such as slow degradation, ease of processing, biocompatibility, and biodegradability. The degradation rate of PCL can be controlled by modifying the molecular weight and blending it with other polymers.141 Additionally, it is resistant to deformation and can maintain its strength and shape even when bent or twisted. From a biological perspective, PCL has been used as a carrier for various bioactive agents including growth factors, antibiotics, and stem cells, which can further enhance the bone regeneration process.142 In the context of osteogenesis, PCL can be made into porous scaffolds, which provides a suitable environment for cell growth and helps to promote the formation of new bone tissue.143,144 PCL has a high resistance to water and most organic materials, making it suitable for biomedical, tissue engineering, and wound healing applications.145−147
PEG
PEG is a bioresorbable polymer (Figure 5) with great biocompatibility, low toxicity, and ease of processing. This material could be efficiently used in composite membranes and blended with other polymers in making various new options for GBR membrane purpose.148,149 The effectiveness of PEG hydrogel as a barrier membrane for GBR in animal models, such as rabbits,150 has been explored featuring the potential of PEG hydrogel to successfully promote bone regeneration in defect areas.148 PEG-hydroxyapatite hybrid hydrogel has also been investigated as a scaffold for bone tissue engineering and has shown the ability to support bone cell growth and promote bone regeneration in vitro and in vivo.151 These findings suggest that the PEG hydrogel may have potential as an effective material for GBR and bone tissue engineering applications. Furthermore, PEG has been used as a plasticizer to enhance the material properties of other polymers including PCL.112 This is done in order to decrease the hardness of PCL, lower the glass transition temperature, and reduce the amount of solvent required for solvent-involved manufacturing processes. In a multicentered randomized clinical trial by Jung et al.,111 clinical outcomes of PEG versus native collagen membranes covering bony dehiscence type defects around bone level dental titanium implants were assessed. In this study, vertical bone fill was measured at baseline and after 6 months of healing as a primary parameter. Marginal bone level (MBL) and soft tissue conditions were recorded as secondary parameters after 18 months of loading. Results demonstrated that both PEG and collagen groups had comparable vertical bone fill (59% and 64%) and a change in defect height, and a comparison between two groups did not show a statistical difference.111
Bioactive Composite Membranes
Bioactive composite membranes refer to a type of material that is designed to have biological activity and can be used in various medical applications such as tissue engineering, drug delivery, and wound healing. These membranes typically combine biocompatible and biodegradable polymers with bioactive molecules such as growth factors, peptides, and proteins. The bioactive molecules incorporated into the composite membrane can interact with cells and tissues, promoting cell proliferation, differentiation, and migration. This can be especially useful in tissue engineering applications, where the goal is to create new tissues or organs by growing cells on a scaffold or membrane. Pal et al.152 developed a hydrogel composed of a collagen and elastin-like polypeptide interpenetrating network that encapsulates BMP-2, doxycycline, and 45S5 Bioglass, that enhanced human adipose-derived stem cell attachment, proliferation, and differentiation toward the osteogenic lineage. In vitro culture has revealed the deposition of hydroxyapatite particles within a collagenous fibrillar structure. When implanted in a rat cranial defect model, the hydrogel fosters the development of mature mineralized tissue that links with the surrounding bone, indicating significant potential for improved bone healing following cranioplasty.152 Bioactive composite membranes can also be used for drug delivery, as the bioactive molecules can be designed to release drugs or other therapeutic agents over time, providing sustained and localized delivery to the desired tissue. Here, we present a short review of the latest achievements and research directions on key requirements of bioactive composite membranes for GBR application:
Biocompatibility
The introduction of collagen material in the surgical site has been reported to provoke foreign body reaction (FBR),13,153 an inevitable immune response that occurs when a foreign material (such as a membrane or a bone substitute) is placed in the body.154 By recognizing the implanted material as a foreign body, the inflammatory response is activated, which can lead to polymorphonuclear leukocyte (PMN) recruitment.155 The inflammatory response is a complex process that is mediated by various cells and molecules, including immune cells (such as neutrophils and monocytes) and inflammatory mediators (such as cytokines and chemokines).154 During acute inflammation, the blood vessels in the area of injury or foreign material become more permeable, allowing immune cells and other inflammatory mediators to migrate to the site. The immune cells then begin to phagocytose and remove the foreign material, while the inflammatory mediators help to recruit more immune cells and promote tissue repair. The acute inflammation can last for a few days to a week and is followed by the resolution and the remodeling phase.154
Monocytes and macrophages are closely related types of white blood cells that play important roles in the immune response and inflammation,156 and could be polarized into proinflammatory (M1) or proregenerative (M2) phenotypes.157 The efficient and timely switch from M1 to M2 phenotype is reported to be responsible for successful healing and regeneration,158 while a prolonged M1 polarization can lead to an increase in fibrous encapsulation and bone regeneration failure.159 To minimize FBR, biocompatible materials and techniques are used in GBR. Chu et al.32,160 demonstrated the potential of epigallocatechin-3-gallate (EGCG)-modified collagen membrane (E-Col membrane) as an excellent barrier membrane to minimize FBR and enhance bone regeneration in vivo (Figure 6a). EGCG is the most abundant catechin in green tea, has demonstrated remarkable anticancer, anti-inflammatory, antifibrosis, and pro-osteogenesis characteristics, and has been proposed to modulate the macrophage polarization in biomaterials.161 The results showed that the migration of keratinocytes was successfully prevented in EGCG-modified collagen membranes (Figure 6b), providing enough room for osteoblasts with no adverse effect on the viability and adhesion of osteoblasts. A significantly higher cell viability was observed in the modified membrane with cell seeds on day 1 compared to the unmodified one (Figure 6c), with comparable live/dead staining results (Figure 6b, insets). Quantitative polymerase chain reaction (qPCR) analysis demonstrated that the expression of CD206, CD163, RUNX2, OPN, and colla1 markers significantly increased in E-Col membranes compared to that of Col (Figure 6e). The highest bone healing was observed in the modified membrane with EGCG with a thick and ordered bone formation and the closure of the bone defect, while the most fibrous tissue was found in the pure collagen membrane, which is unfavorable (Figure 6d). These findings demonstrate that the EGCG-modified collagen membranes are favorable for switching from M1 to M2 phenotype and can act as a suitable barrier against the invasion of keratinocytes to the bone defect, thus promoting in vivo bone regeneration for guided bone regeneration practice (Figure 6f).
Figure 6.
(a) In vivo promotion of bone regeneration after membrane implantation. Inset: the comparison of defect size with the bone volume using Micro-CT 4 weeks post membrane implantation of Col and E-Col. (b) SEM images of keratinocytes and osteoblast seeded on EGCG-modified collagen membranes with insets to show their corresponding live/dead staining. (c) Cell viability and (d) H&E staining of membrane sites 8 weeks post membrane implantation of Col (red rectangle circled) and E-Col (green rectangle circled), NB represents new regenerated bone. (e) Results of qPCR, which demonstrated that expression of CD206, CD163, RUNX2, OPN, and colla1 was significantly upregulated in E-Col compared to that of Col. (f) Schematic diagrams on the function of EGCG-modified collagen membrane. Adapted with permission from ref (32). Copyright 2019 Elsevier.
The immune response is considered among the most critical aspects of biomaterial-mediated reconstruction of hard and soft tissue, which could be modulated by modification of the properties of biomaterial. It was previously believed that a biomaterial is better to be “inert”, but according to a paradigm shift in the nature of biomaterials, recent advances are more focused on “immunomodulatory” materials.162−165 There are several nutrient elements proved to be efficient in osteo-immunomodulation including calcium (Ca), zinc (Zn), and silicon (Si) (Figure 3).166,167 Using pulsed laser deposition (PLD) technique, Chen et al.168 deposited a highly homogeneous, uniform layer of Ca2ZnSi2O7 nanoparticles, as a bioactive glass, on a commonly used Bio-Gide collagen membrane with a rough surface on one side to manipulate the osteo-immunomodulatory property of the barrier collagen membrane. The other side, however, was made of a smooth surface of a dense collagen fiber network. As presented in Figure 7a,b, the smooth surface could successfully prevent the migration of soft tissue into the defect site, while cell migration into the membrane scaffold was observed in the rough surface (Figure 7e,f), forming a monocyte infiltration zone. The osteo-immunomodulatory properties of collagen membrane showed that the smooth surface acts like a foreign body reaction toward inert biomaterials, with macrophages from the M2 phenotype, and more anti-inflammatory cytokine profile was seen in the smooth surface (Figure 7c,d). The shift toward the M2 macrophage phenotype is reported to be a key component of fibro-encapsulation.158 On the rough surface, the FBR was more like a bioactive material. Fibroblasts were declined with significantly lower fibro-encapsulation response on the rough surface (Figure 7g,h).168 The dominant phenotype presented in situ was M1 which plays a key role in FBR. Moreover, a proinflammatory cytokine profile was observed in the rough surface with increased revascularization compared to a smooth surface profile.168
Figure 7.
Host subcutaneous tissue response to collagen membranes 7 days post implantation by H&E staining and cell attachment of macrophages on (a–d) smooth and (e–h) rough sides of the collagen membranes. (a,b) The smooth surface prevents the cells from invading into the membranes, and (c,d) the macrophages grow on the smooth surface without migrating into the collagen network. (e,f) The cells migrate into the membrane scaffold on the rough side, generating a monocyte infiltration zone. (g) Macrophages migrate into the collagen network with (h) extended pseudopodia attach firmly on the collagen fibers. Adapted with permission from ref (168). Copyright 2018 Royal Society of Chemistry.
Effective regulation in osteogenesis was reported by incorporating nutrient elements significantly enhancing osteogenic differentiation. Calcium ions have been found to trigger the calcium-sensing receptor (CaSR), a member of the G protein-coupled receptor superfamily and a key player in the maintenance of calcium homeostasis.169 This leads to a reduction in Wnt5A protein and consequently suppresses inflammation.170 Zinc ions, on the other hand, are responsible for the normal function and development of immune cells, while deficiency in Zn can lead to impairment in production and inflammatory cytokines signaling process.171 It is speculated that Si ions released from the membrane could activate the Wnt3 pathway, a signaling molecule that plays a crucial role in the regulation of bone formation and remodeling. These findings demonstrated that the next generation of membranes need a paradigm shift from just a physical barrier to a bioactive barrier with osteo-immunomodulation as an effective way to improve bone regeneration.168
Mechanical Stability
Mechanical stability is one of the most critical characteristics of GBR barrier membranes as the amount of regenerated bone in the bone defect would be reduced if the membranes collapse into the defect space. So, the utilization of barrier membranes in biomedical and pharmaceutical applications is limited by their mechanical instability in aqueous environments. Generally, cross-linked membranes and scaffolds are less susceptible to rapid enzymatic degradation and show better mechanical stability compared to non-cross-linked biomaterials.172,173 However, many cross-linkers are toxic or immunogenic, hindering use in in vivo applications.174 To address this issue, Acevedo et al.73 proposed the application of UV radiation as a nontoxic cross-linking technique to upgrade the mechanical properties of a biodegradable gelatin-chitosan membrane (Figure 8a,b). The membranes were made of the biopolymers of gelatin-chitosan (56% gelatin, 42% chitosan) and a homogeneous distribution of titania (TiO2, 1%) and hydroxyapatite (HAp, 1%) nanoparticles (Figure 8c,d). Gelatin is a naturally driven polymer obtained by incomplete denaturation of collagen. Compared to collagen, gelatin is less expensive with reduced antigenicity, and shows similar cell adhesion and proliferation. Bonded together, gelatin and chitosan form a polyelectrolyte complex with enhanced physical and mechanical properties, enabling the copolymer to be embedded with biological molecules such as growth factors. The results showed a significant enhancement in the membrane’s stiffness upon increasing UV radiation doses, indicating efficient cross-linking of the material. The optimal mechanical property was reported to occur under an optimum UV dosage of 20 J·cm–2 (Figure 8e), demonstrating the maximum cross-linking state. Similarly, an increment in cell adhesion, spreading, and growth velocity was observed on UV-irradiated membranes (Figure 8g), demonstrating the acceptable biocompatibility of the cross-linked membranes. However, the degradation percentage decreased to zero for the UV-treated membranes with more than 10 J·cm–2 radiation doses (Figure 8f). In addition, serious concerns could be raised regarding nonresorbable titania nanoparticles, as they have been classified as a possible carcinogen to humans, according to the World Health Organization’s International Agency for Research on Cancer (IARC).175
Figure 8.

SEM images of the nanocomposed gelatin-chitosan membrane: (a) top view and (b) transversal section of the membrane without nanoparticles (red rectangle circled) and corresponding (c) top and (d) side views of membrane incorporated with nanoparticles (green rectangle circled). (e) Mechanical and (f) degradation behavior of the UV-irradiated nanocomposed gelatin-chitosan membranes. (g) Cell spreading on UV-irradiated nanocomposed gelatin-chitosan membranes at different time intervals. Scale bars are 100 μm. MEF cells in suspension (2,5 × 105 cells/film) were allowed to attach and spread onto UV-irradiated membranes (0, 5, and 10 J·cm–2) for 15 and 120 min. Adapted with permission from ref (73). Copyright 2019 Elsevier.
In a similar approach, Ansarizadeh et al.176 developed a novel barrier membrane for GBR application by combining collagen-chitosan and lyophilized advanced platelet rich fibrin (A-PRF), featuring excellent mechanical stability, cell viability, degradability, and reasonable osteogenic differentiation. Increasing the Chit/Col weight ratio from 0 to 4 and decreasing the A-PRF concentration from 5 to 0 resulted in a significant enhancement in the Young’s modulus of the fabricated membranes (from 3 to 8 MPa, Figure 9a), which peaked at 8 MPa for the membrane with a Chit/Col weight ratio and A-PRF concentration of 4 and 0.58 mg·mL–1, respectively. This enhanced mechanical property could be ascribed to a higher cross-linking stage between collagen and chitosan chains caused by free amine group in chitosan leading to morphology deformation, pore size reduction, and greater interconnectivity within collagen-based membranes (Figure 9c–e). Enhanced mechanical properties, cell infiltration, nutrient diffusion, and consequently cell survival rate have been previously reported for membranes with smaller pore size.177,178 Cell viability of MSCs was also improved by both increasing the chit/col weight ratio and A-PRF concentration, which could be attributed to the higher number of amine groups in the membrane with a higher Chit-Col weight ratio, leading to a better cell adhesion property. In addition, increasing the A-PRF concentration resulted in an increase in the degradation rate of the fabricated membranes (Figure 9b), which is attributed to a higher degradability of fibrins, and consequently higher release rate of growth factors, compared to that of collagen and chitosan. Using a response surface methodology, a polynomial model was designed to mathematically explore the correlation between two independent variables and predict the optimum Col/Chit/A-PRF ratios.176
Figure 9.
(a) Effect of Chit/Col weight ratio and A-PRF concentration on Young’s modulus of the membrane. (b) Effect of Chit/Col weight ratio and A-PRF concentration on degradation rate of membrane. The cross-sectional SEM images of membranes made of (c) collagen, (d) collagen-chitosan (Chit/Col weight ratio = 1), and (e) collagen/chitosan (Chit/Col weight ratio = 4). Adapted with permission from ref (176). Copyright 2019 Elsevier.
Chen et al.179 developed a new composite scaffold developed using a combination of elastin-like polypeptide (ELP) and silk fibroin (SF). The scaffold was created by using a dehydrothermal treatment that is both simple and environmentally friendly. The resulting scaffold showed unique mechanical properties and was able to support the adhesion, proliferation, and differentiation of both bone mesenchymal stem cells and chondrocytes in vitro. The scaffold also demonstrated its ability to enhance cartilage and subchondral bone repair in vivo, making it a potential biomaterial for repairing tissues such as skin and for use in osteochondral repair.179 Omar et al.180 compared the efficacy of two resorbable collagen membranes for GBR in rat femurs. They found that elastin-collagen had a higher percentage of new bone formation in the central defect area compared to that of pure collagen. Molecular analysis also revealed differences in BMP2 expression, with higher expression observed in the collagen group and higher expression of BMP2 and remodeling activity observed in the elastin-collagen group.180 These findings suggest that the healing process of GBR can be influenced by the type of resorbable collagen membrane used. Gurumurthy et al.181 reinforced collagen with ELP and optimized the composite composition using response surface methodology. The optimized composite exhibited an improved tensile strength, elastic modulus, and toughness. Physical property analysis demonstrated that ELP lowered the residual water content and interacted with collagen. The collagen-ELP composite demonstrated a dense microstructure, which improved the mechanical properties. These composites can be utilized to construct rigid, porous structures that are suited for tissue engineering applications.
Another approach to enhance the mechanical properties of barrier membranes is adding PCL, a nontoxic, nonallergenic, biodegradable, and biocompatible polymer. Several studies reported the development of natural-synthetic based polymers, using PCL, featuring enhanced mechanical stability and modulated degradation rate.109,182−184 However, one of the main drawbacks of PCL is its slow degradation rate, as previous studies reported a 24-month degradation time, which is not practical for GBR applications.185 To improve the degradation profile of PCL-modified gelatin membranes, Joo et al.186 fabricated dual-layered membrane embedding PCL and gelatin into alginate sheet and followed by an activation stage (Figure 10a–c) where beta-tricalcium phosphate (β-TCP) molecules were embedded into the membrane. β-TCP is a biocompatible ceramic material that has been used in a variety of medical applications, including as a bone substitute or scaffold for bone tissue engineering. It can also be used in dental implants and as a filler for bone defects.186,187
Figure 10.
SEM images of (a) a dual-layered membrane made of (b) an alginate sheet and (c) the electrospun cross-linked PCL/Gel/β-TCP membrane. The yellow arrow indicates the alginate-membrane interface. (d) Quantitative study of in vitro post degradation weight loss of electrospun membrane and casted alginate sheet. (e) H&E stained images of electrospun membrane after 4 and 8 weeks of implantation. M: membrane, H: host, NB: new bone formation. Asterisk (*) denotes the bone ingrowth via pores of the membrane. Adapted with permission from ref (186). Copyright 2022 IOP Publishing Ltd.
Alginate-based materials have been used in GBR to protect the bone defect, but also to act as a source of growth factors, promoting bone formation and remodeling.188 They can also improve bone repair and regeneration and reduce the risk of complications such as bacterial infection.189,190 Another unique aspect of alginate is its pH-dependent strength properties, so the membrane would immediately become firm within the initial stages after implantation upon being exposed to the acidic environment of the wound area.186,191 In vitro degradation showed approximately 50% and 22% weight loss after 2 weeks with visible cracks and deformations observed on alginate and electrospun membranes, respectively (Figure 10d).186 The combination of PCL and gelatin can enhance cell attachment to the membrane, and β-TCP is a substrate that could improve cell attachment in bone area and increase osteogenic differentiation and cell proliferation. In vivo histologic analysis results showed good space maintenance at the defect site with impressive new bone formation after 8 weeks (Figure 10e). At 8 weeks, the membranes maintained their shape and thickness with great integration into the surrounding tissues. No inflammatory cells were detected with minor penetration of soft tissue into the new bone area after 4 weeks of implantation (Figure 10e).186
The main concern about PCL, however, is its poor cell adhesion and inadequate wettability properties, which could be attributed to the low surface energy and hydrophobicity of PCL, which makes it difficult for cells to adhere to the surface.192 To address this issue, β-TCP was included in the composite membrane due to its great osteo-conductivity and biocompatibility.193 Additionally, β-TCP could act as a precursor for Ca2+ and PO43– ions, encouraging bone regeneration in osteogenesis.194 Using the electrospinning technique, Ezati et al.141 fabricated polymeric nanofibers with a diameter range of 200–500 nm, resulting in highly porous membranes with efficient nutrient exchange and cell migration into the scaffold. The mechanical stability analysis showed significant improvement in the elastic modulus, elongation, and ultimate tensile strength (UTS) of the membranes upon addition of β-TCP, with the optimal mechanical properties peaking for the 5% β-TCP composite (200 MPa, 38% and 8.5 MP for modulus, elongation, and UTS, respectively). This could be attributed to cross-linking between polymer chains caused by β-TCP content and, consequently, pore size reduction, as previously reported by Tsuru et al.142 In addition to mechanical properties, higher wettability and roughness were observed in the fabricated membranes upon addition of β-TCP. The MTT assay demonstrated a higher cell affinity and proliferation rate for membranes with 3–5% β-TCP, with more cell contacts and extent of filopodia and lamellipodia compared to the control sample. This enhanced cell adhesion could be attributed to the higher hydrophilicity and surface roughness of the modified composite membranes. Gene expression tests were performed to examine two osteogenic markers: alkaline phosphatase (ALP) and type I collagen. The former is for early stage differentiation, and the latter is for later differentiation of MG63 cells. MG63 cells expressed more maturation markers and higher expression of type I collagen by increasing the β-TCP content up to 3% β-TCP, which could be due to the release of calcium ions from the composite membrane. Moreover, samples with a higher concentration of β-TCP showed greater biodegradation rate and weight loss (up to 67% after 4 weeks of incubation), which could be related to higher hydrophilicity of membranes with β-TCP content.195
Silk fibroin is a protein found in silk that can be processed into various materials with different properties, including films, fibers, and hydrogels. One of the challenges in working with silk fibroin is that it becomes brittle when dried, resulting in high tensile strength but poor elongation. This is because the protein molecules lose their ability to interact with each other and form a stable network, which leads to a decrease in the mechanical properties. Various strategies have been developed to overcome this brittleness, such as cross-linking, blending with other polymers, or incorporating plasticizers.88,196 To tackle this problem, Geão et al.197 designed a flexible SF membrane by adding glycerol (GLY) and polyvinyl alcohol (PVA) as plasticizers. GLY is a clear, colorless, and odorless liquid that is widely added to plastics to increase their flexibility and durability, and to personal skincare products as a moisturizer.198 PVA is a water-soluble synthetic polymer that is commonly added to cement and plaster-based products to improve their flexibility and durability, and to adhesives and coatings to increase their viscosity and workability.199 It is considered a nontoxic and environmentally friendly plasticizer, as it is water-soluble and can be easily removed during cleaning.200 Benefiting from GLY and PVA properties, Geão et al.197 managed to decrease the membrane’s UTS from 40 MPa in pure SF membrane to 22 and 12 MPa in membranes with 10% GLY and 10% PVA, respectively (Figure 11a). As expected, a significant enhancement in membrane’s elongation was achieved upon the addition of 10% plasticizer from 147% for SF, to 228% and 249% for 10% GLY and PVA, respectively (Figure 11b). Higher surface roughness was also achieved by increasing the GLY and PVA contents (up to 30%) with an acceptable degree of transparency. The SEM images depicted that the control membrane had a smooth surface after undergoing thermal treatment (Figure 11c, i). However, when GLY was added to the membrane, a globular microstructure was formed, which was more pronounced at a concentration of 30% (Figure 11d,e, i). In vitro cellular studies showed enhanced human periodontal ligament cell (hPDLs) adhesion, viability, and proliferation in samples with SF and GLY, while no visible cells were observed on the membrane with PVA content (Figure 11c–e, (ii). This higher cell viability properties of GLY could be attributed to the higher hydrophilicity of the surface caused by hydroxy (−OH) groups.201
Figure 11.

Representative (a) ultimate tensile stress (UTS) and (b) ultimate strain of membranes with different GLY and PVA content. (i) SEM micrographs and (ii) confocal images of the produced SF-based membranes: (c) pure SF, (d) 70SF:30GLY, and (e) 70SF:30PVA dried for 12 h. Adapted with permission from ref (197). Copyright 2019 Springer Nature.
Antibacterial Activity
Bacterial infection in GBR membranes is another major problem, leading to complications in the procedure. The membrane is used to cover the bone defect and protect it from bacterial contamination, but if it becomes infected, it can impede the healing process and lead to potential failure.202,203 The most common bacterial species that cause infection in GBR membranes are Streptococcus spp, Staphylococcus spp, and Porphyromonas gingivalis.204,205 One common solution reported is to directly dope the membrane with antibiotics to minimize the risk of infection.206,207 However, the uncontrollable continuous release of antibiotics was reported as the main drawback, hindering their real-world application as anti-infectious membranes.
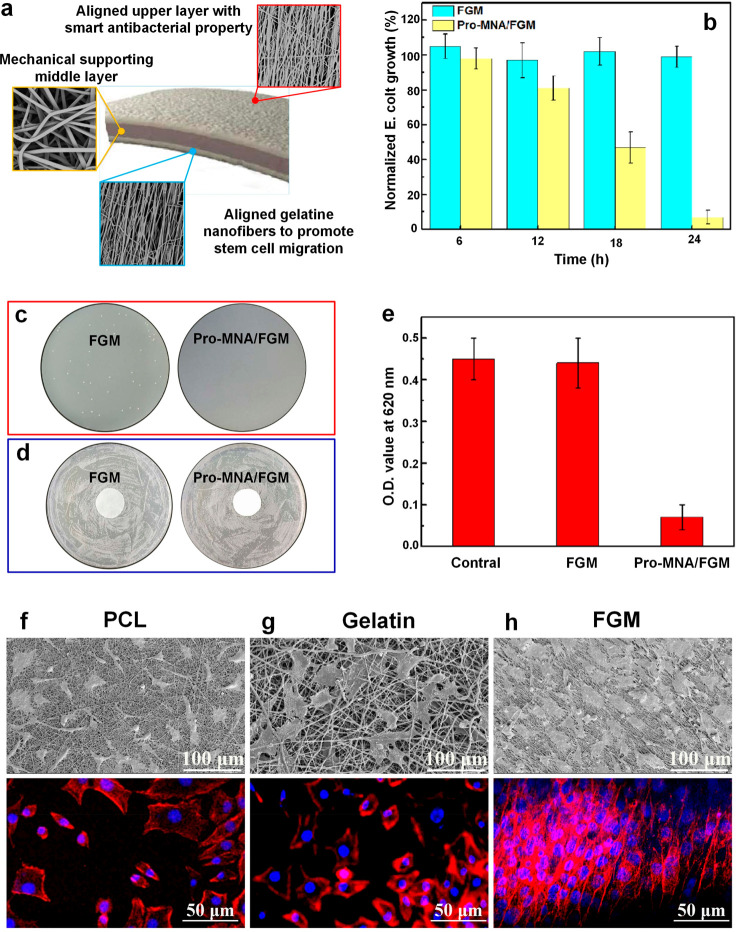
Recently, He et al.208 designed a two-layered FGM with gelatin as the surface layer to provide biocompatibility and PCL as the core material (Figure 12a), to simultaneously enhance the mechanical properties of the fabricated membranes. A strong tensile strength of 6 MPa and strain at failure of 490% were achieved for the developed membrane, which was significantly higher compared to the ones for gelatin (2.6 MPa and 210%, respectively). However, a dramatic reduction in mechanical properties was observed after 5 weeks in the cell culture medium, which could be attributed to the breakdown of the PCL molecules.113 The interfacing space was loaded with pro-metronidazole (Pro-MNA), a hydrophobic and water-insoluble prodrug form of metronidazole, to develop a self-defensive membrane with controllable antibiotic release that has been used to treat a variety of bacterial and protozoal infections. In contrast to bacterial infection in the control sample, significantly restrained bacterial growth efficiency was observed for Pro-MNA/FGM within 24 h of incubation (Figure 12b). The mechanism of infection responsive release in Pro-MNA membrane showed a trigger in MNA release, due to the accumulation of esterase and decrease in pH value. The antibiotic effect reached the optimum level at 24 h after incubation, inhibiting the formation of any bacterial colonies in the Pro-MNA/FGM sample (Figure 12c,d). Pro-MNA/FGM inhibited bacterial growth and prevented bacterial adhesion on cells compared to the control (Figure 12e), suggesting that Pro-MNA loaded FGM has an inherent self-defensive antibacterial property. Epithelial cell adhesion and proliferation increased continuously within 7 days of incubation, with a consistent ranking of FGM > R-gelatin > R-PCL (Figure 12f–h). This result could be attributed to the synergistic effect of chemical composition and fibers alignment on cell proliferation and adhesion.208
Figure 12.
(a) Sandwich structured multifunctionalized graded membrane. (b) Normalized growth of E. coli incubated with the membranes for different durations. (c, left) Plate samples showing colonies of S. aureus and inhibition zone of the membrane: the sample incubated with FGM (without Pro-MNA loading) shows some bacterial colonies were formed. (c, right) No bacterial colonies formed for the sample incubated with Pro-MNA/FGM. (d) Nonetheless, both the control sample and Pro-MNA/FGM did not exhibit a consistently stable inhibition zone, which suggests that no MNA diffused from the membrane. (e) Quantitative study of the OD. values in the bacteria-cell infection model to investigate the bacterial growth. (f-h) SEM micrographs (up) and confocal fluorescent images (bottom) of cells proliferated on R-PCL, R-gelatin, and FGM membranes for 7 days. Adapted with permission from ref (208). Copyright 2020 Elsevier.
Abe et al.209 found that the PLCL bilayer membrane had reduced bacterial adhesion and blocked bacterial penetration, compared to the membranes of collagen and PLGA (Figure 13a), leading to a favorable outcome for regenerative treatments. Moreover, colony-forming units (CFU) counts of the incubated bacteria throughout the experimental period up to 72 h (Figure 13b), revealing that (1) P. gingivalis showed that the PLCL membrane had fewer adherent cells compared to the PLGA or Col membranes. While the PLCL membrane had significantly lower numbers of adherent P. gingivalis than the Col membrane, there was no statistical difference between the PLCL and PLGA membranes until after 72 h of incubation with P. gingivalis. In contrast, there were no significant differences in the adherence of P. gingivalis to PLGA and Col membranes at any time point. (2) For Streptococcus mutans, the PLCL membrane exhibited significantly fewer adherent cells than the PLGA membrane after 6, 24, and 72 h of incubation, and a lower number of adherent cells than the Col membrane, although no statistical significance was found. (3) CFU counts of membranes incubated with bacteria from human saliva displayed that the PLCL compact layer had significantly fewer CFUs than PLGA and Col at 6 h, and after 72 h of incubation there were no statistical differences in CFU counts between any of the groups (Figure 13b).209 The properties of the PLCL membrane, including reduced bacterial adhesion and penetration, may be beneficial for regenerative treatments, and in the case of exposure at surgical sites, efficient barrier function may facilitate the management of complications in guided tissue and bone regeneration.
Figure 13.
(a) SEM images of the P. gingivalis, S. mutans, and multiple bacteria obtained from human saliva cultured on PLGA, Col, and PLCL membranes after 24 h. White arrows indicate the bacterial cells (colored in orange for a clear vision). All scale bars are10 μm. (b) CFU counts of cultured P. gingivalis, S. mutans, and multiple bacteria obtained from human saliva on the three membranes at different time intervals. Adapted with permission from ref (209). Copyright 2022 John Wiley & Sons.
Cell Proliferation Enhancement
Growth factors are naturally occurring proteins that play a crucial role in stimulating and regulating bone growth and healing. In GBR, growth factors are usually delivered directly to the site of the defect via a carrier material, such as a collagen sponge or a synthetic scaffold, to stimulate the formation of new bone tissue, reduce the healing time, and improve the final outcomes of the procedure. The most common growth factors used in barrier membranes for GBR procedures in dentistry and oral surgery are typically obtained from either platelet-rich plasma (PRP) including Platelet-derived growth factor (PDGF), Growth/differentiation factor 5 (GDF-5) and Fibroblast growth factor-2 (FGF-2) or BMP including recombinant human bone morphogenetic protein-2 (rhBMP-2) and recombinant human bone morphogenetic protein-9 (rhBMP-9). In the field of regenerative medicine, these growth factors have been studied for their potential to promote tissue regeneration and repair including the regeneration of bone, connective tissues, and blood vessels. They have shown enormous potential to enhance tissue regeneration and repair, including the regeneration of bone, cartilage, and other connective tissues. They have been studied to stimulate the proliferation and differentiation of bone-forming cells (osteoblasts) and promote the formation of new blood vessels (angiogenesis), which are necessary for delivering oxygen and nutrients to the repair sites. The main challenge, however, is dosing, as determining the optimal dose of growth factor to use in GTR and GBR procedures can be challenging, and there is a lack of consensus on the best approach. In addition, there is a lack of standardization in the production and preparation of growth factors for use in GTR and GBR procedures, which can affect their effectiveness and safety. Furthermore, using growth factors can significantly increase the cost of GTR and GBR procedures, which can be a burdento some patients.
Shirzaei Sani et al.210 reported the potential use of hydrogel-based biomaterials in tissue engineering in promoting tissue regeneration in vivo due to their high-water content, permeability, and ability to create a supportive environment for cells. However, the clinical application of hydrogels has been limited by factors such as poor mechanical performance, low adhesive strength, uncontrolled degradation rates, and susceptibility to bacterial colonization. To tackle this issue, they introduced a new type of hydrogel made from methacrylated hyaluronic acid (MeHA) and an ELP, which is elastic, antimicrobial, and adhesive. This hydrogel can be rapidly photo-cross-linked in situ, making it suitable for the regeneration and repair of various tissues. Different physical properties of the hydrogels were engineered by varying the concentrations of MeHA and ELP. The MeHA/ELP hydrogels exhibited a higher adhesive strength to tissue than commercially available tissue adhesives. Zinc oxide nanoparticles were incorporated into the hydrogel to make it antimicrobial, and it was found to significantly inhibit the growth of methicillin-resistant Staphylococcus aureus. The hydrogel also supported the growth, spreading, and proliferation of mammalian cells in vitro. In vivo subcutaneous implantation demonstrated that MeHA/ELP hydrogels did not elicit a significant inflammatory response but could be efficiently biodegraded and promoted the integration of new autologous tissue.
Yamano et al.55 evaluated the effect of using bioactive collagen membranes with PDGF and GDF-5 on bone regeneration in a preclinical animal model. The results showed that both PDGF and GDF-5 released from the bioactive collagen membranes significantly increased bone regeneration compared to control groups without growth factor treatment, with GDF-5 demonstrating an accelerated yet dose-dependent bone regeneration behavior compared to PDGF. In addition, the collagen membranes loaded with GDF-5 showed significantly higher ALP and cell proliferation activities and osteogenic gene expression of transcription factors compared to the ones loaded with PDGF or without any growth factor. In another approach, Thoma and co-workers211,212 compared the bone regeneration performance of recombinant human PDFG-BB (rhPDGF-BB) and rhBMP-2, which are the synthesized versions of PDGF and BMP-2, respectively. In dental surgery, these growth factors are usually used in conjunction with other materials, such as bone substitute materials or barrier membranes, to promote new bone growth and improve the volume and quality of the surrounding bone. Using dental implants on the buccal sides of an edentulous ridge in ten dogs, they reported a significant increase in bone volume and density around the dental implants, leading to its higher stability and longevity. As presented in Figure 14a–d, the mean percentages of regenerated area within total defect area increased to 56.95% and 48.86% for deproteinized bovine bone mineral (DBBM) mixed with rhBMP-2 and a collagen membrane (CM) (DBBM/BMP-2) (Figure 14a) and DBBM mixed with rhPDGF-BB and CM (DBBM/PDGF) (Figure 14b), respectively, which is significantly higher compared to 33.44% (Figure 14c) and 1.59% (Figure 14d) for DBBM and control samples at 8 weeks, respectively. In another study, they investigated the use of rBMP-2 and PDGF-BB for localized bone regeneration in rabbits and assessed the effectiveness of this treatment using histologic and radiographic methods. At 8 weeks, the area of bone regeneration (histology; AAHisto) peaked at 96.29% for DBBM/BMP-2, in which bone formation filled the entire space between the DBBM particles, reaching every corner of the cylinders (Figure 14e). The cylinders were therefore densely packed with mineralized tissues. In DBBM/PDFG, bone formation partially reached the top of the cylinder with mean AAHisto of 46.37% (Figure 14f), which was slightly higher than DBBM (Figure 14g) and control (Figure 14h) samples with 39.66% and 35.98%, respectively. The highest bone regeneration was achieved for DBBM/BMP-2 (35.62%), with statistically significant differences compared to all other groups (P < 0.05). The results showed that the use of rhBMP-2 significantly enhanced bone regeneration compared to all other groups including those with rhPDGF-BB. In another approach, the effect of FGF-2 on the preservation of the alveolar ridge after tooth extraction was investigated using acellular dermal matrix (ADM) membranes. ADM membranes are semisynthetic membranes derived from natural sources (human or animal skin tissue) but undergo a process of decellularization to remove cells and cellular components and potential modification with other materials such as growth factors. Using bovine xenografts as a bone substitute material, combined with FGF-2-loaded ADM membrane, Wang et al.213 examined the changes in the width and height of alveolar ridges and extraction socket bone healing 8 weeks postextraction. They reported a significant inhibitory effect on alveolar bone width reduction for the FGF-2-loaded membrane (Figure 14i,j) when compared with the unloaded membrane and empty control.
Figure 14.
Histological bone growth at 8 weeks on different membrane groups: (a) DBBM/BMP-2, (b) DBBM/PDGF, (c) DBBM, and (d) control. Reprinted with permission from ref (211). Copyright 2016 John Wiley & Sons. H&E stained images of the histologic slide of different group cylinders after 8 weeks of healing: (e) DBBM/BMP-2, (f) DBBM/PDGF, (g) DBBM, and (h) empty control. Reprinted with permission from ref (212). Copyright 2017 John Wiley & Sons. (i) CBCT observation of extraction socket healing between control, ADM, and FGF-2@ADM immediately and 8 weeks after surgery. (j) 3D reconstruction images of the central region of extraction socket corresponding to the three groups mentioned above 8 weeks after surgery. Adapted with permission from ref (213). Copyright 2021 John Wiley & Sons.
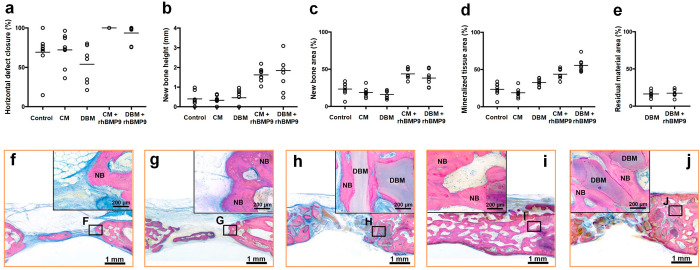
BMP-9 is another member of the BMP family of growth factors that plays a key role in the regulation of bone formation and has been shown to have a potent osteogenic effect in various animal models. To find out the best carrier system for BMP-9, Kobayashi et al.214 studied the effect of two carrier systems, porcine-derived CM and DBBM on the effective delivery of BMP-9 on bone formation in a rabbit calvarial defect model (10 adult female New Zealand white rabbits). The results showed that the CM treated with rhBMP-9 led to complete resolution of the horizontal bone deficiency in all specimens (Figure 15a) with no signs of multinucleated giant cells (MNGC). New bone height and new bone area were both significantly increased in the CM-rhBMP9 and the DBBM-rhBMP9 groups when compared to the control, CM, and DBM groups (Figure 15b,c and f–j). When evaluating the overall amount of mineralized tissue in the treated bone sites, the combination of DBM-rhBMP-9 led to a significantly greater amount of mineralized tissue compared to just DBM (Figure 15c,d). There was no difference in the residual material area between the groups treated with DBM and DBM-rhBMP-9 (Figure 15e). These findings suggest that the use of CM, rather than DBM, in combination with rhBMP-9 may result in a more consistent stimulation of bone growth and complete coverage of the affected area. In a very similar work,36 the team compared the bone regeneration performance of rhBMP-9 loaded on DBBM and CM in a rabbit model. They found that the CM/rhBMP-9 samples achieved close to complete closure of horizontal bone defects, as evidenced by the formation of new bone layers in the CM-implanted areas and the bone growth surrounding DBBM materials (Figure 16a–e). Of note, both DBBM/rhBMP-9 and CM/rhBMP-9 significantly enhanced new bone formation, as demonstrated by an increase in mineralized tissue volume (Figure 16f) and new bone height and area (Figure 16f–h). However, the combination of rhBMP9 with CM showed to be more effective in promoting wound closure compared to DBBM-rhBMP9, which could be attributed to the tendency of BMP9 to be absorbed by CM rather than other bone grafts, including HAp215,216 and ceramics.217 However, further research is necessary, utilizing large animal studies and a variety of bone grafts and barrier membranes, to further explore these results.
Figure 15.
Histomorphometrical measurements of linear parameters: (a) percentage of horizontal defect closure in each group; (b) length of maximum new bone height in the middle area of defect; (c) percentage of new bone area, (d) mineralized tissue area, and (e) residual material area in DBM and DBM1rhBMP9 groups. Dot plots demonstrate data for all specimens and the bars indicate the average of the values in each group. Images of the toluidine blue and fuchsin staining in bone defects at 8 weeks in (f) control, (g) CM, (h) DBM, (i) CM+rhBMP9, and (j) DBM+rhBMP9 groups. Insets correspond to the rectangles accordingly. NB shows new bone, and DBM refers to residual materials. Adapted with permission from ref (214). Copyright 2017 John Wiley & Sons.
Figure 16.
Toluidine blue and fuchsin staining on the middle sections in the bone defects at 8 weeks in (a) control, (b) DBBM/CM alone, (c) DBBM(BMP9)+CM, and (d) DBBM+CM(BMP9) groups. Insets: μCT images in bone defects from the top view at 8 weeks. (e) Statistical study of the horizontal bone defect closure. (f) Mineralized tissue volume measured by μCT, (g) length of new bone height in the middle area of the defect, and (h) new bone area. Dot plots show data for all specimens, and bars indicate the average and SD of the values in each group. Adapted with permission from ref (36). Copyright 2017 John Wiley & Sons.
Summary and Outlook
The use of resorbable membranes for GBR has been a significant development in the field of dentistry and orthopedics. The ability to selectively prevent the growth of nonosteogenic cells and allow the infiltration of osteogenic cells has shown great promise in promoting bone regeneration. Natural barrier membranes, such as collagen-based membranes, are widely used due to their biocompatibility and ability to support cellular attachment and growth. However, their mechanical properties are often inferior to those of synthetic membranes. Synthetic barrier membranes, such as PLGA and PCL, have superior mechanical properties and can be tailored to meet specific requirements. Bioactive nanocomposite-based barrier membranes, such as natural or synthetic polymers reinforced with HAp and TCP, have been developed to enhance the osteogenic potential of membranes and improve bone regeneration. However, there is still a need to optimize the design and properties of these membranes to maximize their efficacy. While resorbable membranes have shown promise in promoting bone regeneration, their success depends on various factors, including the membrane’s degradation rate, mechanical properties, biocompatibility, and ability to support cellular attachment and growth.
There is a need for continued research to optimize the design and properties of resorbable membranes. One important aspect of membrane designs is the degradation rate. Resorbable membranes that degrade too quickly may not provide sufficient support for bone regeneration, while those that degrade too slowly will impede bone formation. Therefore, the development of membranes with controlled degradation rates could improve their effectiveness in promoting bone regeneration.
Another important aspect of membrane design is their mechanical properties. Resorbable membranes should have sufficient strength to withstand the forces of the surrounding tissue, while allowing for cellular infiltration. Furthermore, the incorporation of bioactive materials such as HAp, TCP, and growth factors into resorbable membranes could further enhance their osteogenic potential. The development of new materials and manufacturing techniques may also lead to improved resorbable membranes for GBR. For instance, 3D printing technology has been used to fabricate customized implants for bone regeneration, and this technology could potentially be used to create resorbable membranes with tailored properties. Additionally, clinical studies are needed to evaluate the long-term safety and efficacy of modified resorbable membranes in promoting bone regeneration. Such studies could provide valuable information about the optimal membrane design and properties for various clinical applications.
Author Contributions
⊥ S.A. and X.C.: co-first authors. CRediT: Sara Abtahi conceptualization (equal), methodology (equal), visualization (equal), writing-original draft (equal), writing-review & editing (equal); Xiaohu Chen methodology (equal), project administration (equal), visualization (equal), writing-review & editing (equal); Sima Shahabi conceptualization (equal), supervision (equal); Noushin Nasiri conceptualization (equal), investigation (equal), methodology (equal), project administration (equal), supervision (equal), visualization (equal), writing-original draft (equal), writing-review & editing (equal).
The authors declare no competing financial interest.
References
- Liu J. P. Molecular mechanisms of ageing and related diseases. Clin. Exp. Pharmacol. Physiol. 2014, 41, 445–458. 10.1111/1440-1681.12247. [DOI] [PubMed] [Google Scholar]
- Buser D.; Bornstein M. M.; Weber H. P.; Grütter L.; Schmid B.; Belser U. C. Early implant placement with simultaneous guided bone regeneration following single-tooth extraction in the esthetic zone: A cross-sectional, retrospective study in 45 subjects with a 2-to 4-year follow-up. J. Periodontol. 2008, 79, 1773–1781. 10.1902/jop.2008.080071. [DOI] [PubMed] [Google Scholar]
- Aprile P.; Letourneur D.; Simon-Yarza T. Membranes for guided bone regeneration: a road from bench to bedside. Adv. Healthc. Mater. 2020, 9, 2000707 10.1002/adhm.202000707. [DOI] [PubMed] [Google Scholar]
- Sheikh Z.; Hamdan N.; Ikeda Y.; Grynpas M.; Ganss B.; Glogauer M. Natural graft tissues and synthetic biomaterials for periodontal and alveolar bone reconstructive applications: a review. Biomater. Res. 2017, 21, 1–20. 10.1186/s40824-017-0095-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zumstein T.; Schütz S.; Sahlin H.; Sennerby L. Factors influencing marginal bone loss at a hydrophilic implant design placed with or without GBR procedures: a 5-year retrospective study. Clin Implant Dent Relat Res. 2019, 21, 817–826. 10.1111/cid.12826. [DOI] [PubMed] [Google Scholar]
- Kim H. Y.; Park J. H.; Byun J.-H.; Lee J. H.; Oh S. H. BMP-2-immobilized porous matrix with leaf-stacked structure as a bioactive GBR membrane. ACS Appl. Mater. Interfaces 2018, 10, 30115–30124. 10.1021/acsami.8b09558. [DOI] [PubMed] [Google Scholar]
- Wang H.-L.; Boyapati L. PASS” principles for predictable bone regeneration. Implant Dent. 2006, 15, 8–17. 10.1097/01.id.0000204762.39826.0f. [DOI] [PubMed] [Google Scholar]
- Zakrzewski W.; Dobrzynski M.; Rybak Z.; Szymonowicz M.; Wiglusz R. J. Selected nanomaterials’ application enhanced with the use of stem cells in acceleration of alveolar bone regeneration during augmentation process. Nanomaterials 2020, 10, 1216. 10.3390/nano10061216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasan A.; Byambaa B.; Morshed M.; Cheikh M. I.; Shakoor R. A.; Mustafy T.; Marei H. E. Advances in osteobiologic materials for bone substitutes. J. Tissue Eng. Regen Med. 2018, 12, 1448–1468. 10.1002/term.2677. [DOI] [PubMed] [Google Scholar]
- Leonida A.; Favero G.; Caccianiga P.; Ceraulo S.; Rodella L. F.; Rezzani R.; Caccianiga G. Concentrated Growth Factors (CGF) Combined with Melatonin in Guided Bone Regeneration (GBR): A Case Report. Diagnostics 2022, 12, 1257. 10.3390/diagnostics12051257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gadelkarim M.; Abushouk A. I.; Ghanem E.; Hamaad A. M.; Saad A. M.; Abdel-Daim M. M. Adipose-derived stem cells: effectiveness and advances in delivery in diabetic wound healing. Biomed. Pharmacother. 2018, 107, 625–633. 10.1016/j.biopha.2018.08.013. [DOI] [PubMed] [Google Scholar]
- Zhang W.; Wang N.; Yang M.; Sun T.; Zhang J.; Zhao Y.; Huo N.; Li Z. Periosteum and development of the tissue-engineered periosteum for guided bone regeneration. J. Orthop. Transl. 2022, 33, 41–54. 10.1016/j.jot.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu C.; Zhao X.; Rung S.; Xiao W.; Liu L.; Qu Y.; Man Y. Application of biomaterials in periodontal tissue repair and reconstruction in the presence of inflammation under periodontitis through the foreign body response: Recent progress and perspectives. J. Biomed. Mater. Res. 2022, 110, 7–17. 10.1002/jbm.b.34891. [DOI] [PubMed] [Google Scholar]
- Javed F.; Al-Askar M.; Al-Rasheed A.; Al-Hezaimi K. Significance of the platelet-derived growth factor in periodontal tissue regeneration. Arch. Oral Biol. 2011, 56, 1476–1484. 10.1016/j.archoralbio.2011.06.020. [DOI] [PubMed] [Google Scholar]
- Seong J. M.; Kim B.-C.; Park J.-H.; Kwon I. K.; Mantalaris A.; Hwang Y.-S. Stem cells in bone tissue engineering. Biomed. Mater. 2010, 5, 062001 10.1088/1748-6041/5/6/062001. [DOI] [PubMed] [Google Scholar]
- Jin S.-S.; He D.-Q.; Luo D.; Wang Y.; Yu M.; Guan B.; Fu Y.; Li Z.-X.; Zhang T.; Zhou Y.-H.; et al. A biomimetic hierarchical nanointerface orchestrates macrophage polarization and mesenchymal stem cell recruitment to promote endogenous bone regeneration. ACS Nano 2019, 13, 6581–6595. 10.1021/acsnano.9b00489. [DOI] [PubMed] [Google Scholar]
- Ma B.; Han J.; Zhang S.; Liu F.; Wang S.; Duan J.; Sang Y.; Jiang H.; Li D.; Ge S.; et al. Hydroxyapatite nanobelt/polylactic acid Janus membrane with osteoinduction/barrier dual functions for precise bone defect repair. Acta Biomater. 2018, 71, 108–117. 10.1016/j.actbio.2018.02.033. [DOI] [PubMed] [Google Scholar]
- Li H.; Zheng J.; Zhang S.; Yang C.; Kwon Y.-D.; Kim Y.-J. Experiment of GBR for repair of peri-implant alveolar defects in beagle dogs. Sci. Rep. 2018, 8, 16532. 10.1038/s41598-018-34805-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- dos Santos V. I.; Merlini C.; Aragones Á.; Cesca K.; Fredel M. C. Influence of calcium phosphates incorporation into poly(lactic-co-glycolic acid) electrospun membranes for guided bone regeneration. Polym. Degrad. Stab. 2020, 179, 109253 10.1016/j.polymdegradstab.2020.109253. [DOI] [Google Scholar]
- Omar O.; Elgali I.; Dahlin C.; Thomsen P. Barrier membranes: More than the barrier effect?. J. Clin. Periodontol. 2019, 46, 103–123. 10.1111/jcpe.13068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caballe-Serrano J.; Munar-Frau A.; Ortiz-Puigpelat O.; Soto-Penaloza D.; Penarrocha M.; Hernandez-Alfaro F. On the search of the ideal barrier membrane for guided bone regeneration. J. Clin. Exp. Dent. 2018, 0–0. 10.4317/jced.54767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Weijden F.; Dell’Acqua F.; Slot D. E. Alveolar bone dimensional changes of post-extraction sockets in humans: a systematic review. J. Clin. Periodontol. 2009, 36, 1048–1058. 10.1111/j.1600-051X.2009.01482.x. [DOI] [PubMed] [Google Scholar]
- Zhang M.; Zhou Z.; Yun J.; Liu R.; Li J.; Chen Y.; Cai H.; Jiang H. B.; Lee E.-S.; Han J.; Sun Y. Effect of Different Membranes on Vertical Bone Regeneration: A Systematic Review and Network Meta-Analysis. Biomed. Res. Int. 2022, 2022, 1–16. 10.1155/2022/7742687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li S.; Zhao J.; Xie Y.; Tian T.; Zhang T.; Cai X. Hard tissue stability after guided bone regeneration: a comparison between digital titanium mesh and resorbable membrane. Int. J. Oral Sci. 2021, 13, 37. 10.1038/s41368-021-00143-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alauddin M. S.; Abdul Hayei N. A.; Sabarudin M. A.; Mat Baharin N. H. Barrier membrane in regenerative therapy: a narrative review. Membranes 2022, 12, 444. 10.3390/membranes12050444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sam G.; Pillai B. R. M. Evolution of barrier membranes in periodontal regeneration-“are the third generation membranes really here?. J. Clin Diagn Res. 2014, 8, ZE14 10.7860/JCDR/2014/9957.5272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H.-S.; Byun S.-H.; Cho S.-W.; Yang B.-E. Past, present, and future of regeneration therapy in oral and periodontal tissue: A review. Appl. Sci. 2019, 9, 1046. 10.3390/app9061046. [DOI] [Google Scholar]
- Solomon S.-M.; Sufaru I.-G.; Teslaru S.; Ghiciuc C. M.; Stafie C. S. Finding the perfect membrane: Current knowledge on barrier membranes in regenerative procedures: A descriptive review. Appl. Sci. 2022, 12, 1042. 10.3390/app12031042. [DOI] [Google Scholar]
- Lee C.-H.; Liu K.-S.; Cheng C.-W.; Chan E.-C.; Hung K.-C.; Hsieh M.-J.; Chang S.-H.; Fu X.; Juang J.-H.; Hsieh I.-C.; et al. Codelivery of sustainable antimicrobial agents and platelet-derived growth factor via biodegradable nanofibers for repair of diabetic infectious wounds. ACS Infect. Dis. 2020, 6, 2688–2697. 10.1021/acsinfecdis.0c00321. [DOI] [PubMed] [Google Scholar]
- Caballé-Serrano J.; Abdeslam-Mohamed Y.; Munar-Frau A.; Fujioka-Kobayashi M.; Hernández-Alfaro F.; Miron R. Adsorption and release kinetics of growth factors on barrier membranes for guided tissue/bone regeneration: a systematic review. Arch. Oral Biol. 2019, 100, 57–68. 10.1016/j.archoralbio.2019.02.006. [DOI] [PubMed] [Google Scholar]
- Mandelaris G. A.; DeGroot B. Bone construction surgery: A case report using recombinant human platelet-derived growth factor-BB. Clin. adv. periodontics. 2022, 12, 262. 10.1002/cap.10220. [DOI] [PubMed] [Google Scholar]
- Chu C.; Wang Y.; Wang Y.; Yang R.; Liu L.; Rung S.; Xiang L.; Wu Y.; Du S.; Man Y.; et al. Evaluation of epigallocatechin-3-gallate (EGCG) modified collagen in guided bone regeneration (GBR) surgery and modulation of macrophage phenotype. Mater. Sci. Eng., C 2019, 99, 73–82. 10.1016/j.msec.2019.01.083. [DOI] [PubMed] [Google Scholar]
- Zhang Y.; Wang J.; Xiao J.; Fang T.; Hu N.; Li M.; Deng L.; Cheng Y.; Zhu Y.; Cui W. An electrospun fiber-covered stent with programmable dual drug release for endothelialization acceleration and lumen stenosis prevention. Acta Biomater. 2019, 94, 295–305. 10.1016/j.actbio.2019.06.008. [DOI] [PubMed] [Google Scholar]
- Um C. Y.; Fedirko V.; Flanders W. D.; Höflich C.; Wirthgen E.; Bostick R. M. Circulating insulin-like growth factor-related biomarkers: Correlates and responses to calcium supplementation in colorectal adenoma patients. Mol. Carcinog. 2017, 56, 2127–2134. 10.1002/mc.22669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sousa L.; Guarda M.; Meneses M. J.; Macedo M. P.; Vicente Miranda H. Insulin-degrading enzyme: an ally against metabolic and neurodegenerative diseases. J. Pathol 2021, 255, 346–361. 10.1002/path.5777. [DOI] [PubMed] [Google Scholar]
- Saulacic N.; Fujioka-Kobayashi M.; Kobayashi E.; Schaller B.; Miron R. J. Guided bone regeneration with recombinant human bone morphogenetic protein 9 loaded on either deproteinized bovine bone mineral or a collagen barrier membrane. Clin Implant Dent Relat Res. 2017, 19, 600–607. 10.1111/cid.12491. [DOI] [PubMed] [Google Scholar]
- Hsu Y. T.; Al-Hezaimi K.; Galindo-Moreno P.; O’valle F.; Al-Rasheed A.; Wang H. L. Effects of recombinant human bone morphogenetic protein-2 on vertical bone augmentation in a canine model. J. Periodontol. 2017, 88, 896–905. 10.1902/jop.2017.160516. [DOI] [PubMed] [Google Scholar]
- Chen Y.; Chen M.; Gao Y.; Zhang F.; Jin M.; Lu S.; Han M. Biological efficacy comparison of natural tussah silk and mulberry silk nanofiber membranes for guided bone regeneration. ACS omega 2022, 7, 19979–19987. 10.1021/acsomega.2c01784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elgali I.; Turri A.; Xia W.; Norlindh B.; Johansson A.; Dahlin C.; Thomsen P.; Omar O. Guided bone regeneration using resorbable membrane and different bone substitutes: Early histological and molecular events. Acta Biomater. 2016, 29, 409–423. 10.1016/j.actbio.2015.10.005. [DOI] [PubMed] [Google Scholar]
- Kielty C. M.; Grant M. E. The collagen family: structure, assembly, and organization in the extracellular matrix. Connective tissue and its heritable disorders: molecular, genetic, and medical aspects 2002, 159–221. 10.1002/0471221929.ch2. [DOI] [Google Scholar]
- Ricard-Blum S. The collagen family. Cold Spring Harb. Perspect. Biol. 2011, 3, a004978 10.1101/cshperspect.a004978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fidler A. L.; Boudko S. P.; Rokas A.; Hudson B. G. The triple helix of collagens–an ancient protein structure that enabled animal multicellularity and tissue evolution. J. Cell Sci. 2018, 131, jcs203950 10.1242/jcs.203950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessing B.; Lettner S.; Zechner W. Guided bone regeneration with collagen membranes and particulate graft materials: a systematic review and meta-analysis. Int. J. Oral Maxillofac Implants 2018, 33, 87–100. 10.11607/jomi.5461. [DOI] [PubMed] [Google Scholar]
- Siegfried M. Reticulin and collagen. Physiol. J. 1902, 28, 319. 10.1113/jphysiol.1902.sp000919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- H. W. D. Studien über die Albuminoide mit besonderer Berücksichtigung des Spongin und der Keratine. Nature 1904, 71, 174–175. 10.1038/071174c0. [DOI] [Google Scholar]
- Shoulders M. D.; Raines R. T. Collagen structure and stability. Annu. Rev. Biochem. 2009, 78, 929–958. 10.1146/annurev.biochem.77.032207.120833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J.-J.; Weis M. A.; Kim L. S.; Eyre D. R. Type III collagen, a fibril network modifier in articular cartilage. J. Biol. Chem. 2010, 285, 18537–18544. 10.1074/jbc.M110.112904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruckner P. Suprastructures of extracellular matrices: paradigms of functions controlled by aggregates rather than molecules. Cell Tissue Res. 2010, 339, 7. 10.1007/s00441-009-0864-0. [DOI] [PubMed] [Google Scholar]
- Schmidt M.; Dornelles R.; Mello R.; Kubota E.; Mazutti M.; Kempka A.; Demiate I. Collagen extraction process. Int. Food Res. J. 2016, 23, 913. [Google Scholar]
- Thanka Rajan S.; Subramanian B.; Arockiarajan A. A comprehensive review on biocompatible thin films for biomedical application. Ceram. Int. 2022, 48, 4377–4400. 10.1016/j.ceramint.2021.10.243. [DOI] [Google Scholar]
- Ghafouri S.; Sadeghi-avalshahr A. R.; Molavi A. M.; Hassanzadeh H. Fabrication of Functionally Graded Electrospun Membranes Based on Silk Fibroin for Using as Dental Barrier Membranes in Guided Bone Regeneration. Fibers Polym. 2022, 23, 2549–2556. 10.1007/s12221-022-4304-z. [DOI] [Google Scholar]
- Vallecillo-Rivas M.; Toledano-Osorio M.; Vallecillo C.; Toledano M.; Osorio R. The collagen origin influences the degradation kinetics of guided bone regeneration membranes. Polymers 2021, 13, 3007. 10.3390/polym13173007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vallecillo C.; Toledano-Osorio M.; Vallecillo-Rivas M.; Toledano M.; Osorio R. In vitro biodegradation pattern of collagen matrices for soft tissue augmentation. Polymers 2021, 13, 2633. 10.3390/polym13162633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ren Y.; Fan L.; Alkildani S.; Liu L.; Emmert S.; Najman S.; Rimashevskiy D.; Schnettler R.; Jung O.; Xiong X.; Barbeck M. Barrier Membranes for Guided Bone Regeneration (GBR): A Focus on Recent Advances in Collagen Membranes. Int. J. Mol. Sci. 2022, 23, 14987. 10.3390/ijms232314987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamano S.; Haku K.; Yamanaka T.; Dai J.; Takayama T.; Shohara R.; Tachi K.; Ishioka M.; Hanatani S.; Karunagaran S.; et al. The effect of a bioactive collagen membrane releasing PDGF or GDF-5 on bone regeneration. Biomaterials 2014, 35, 2446–2453. 10.1016/j.biomaterials.2013.12.006. [DOI] [PubMed] [Google Scholar]
- Di Nardo T.; Hadad C.; Nguyen Van Nhien A.; Moores A. Synthesis of high molecular weight chitosan from chitin by mechanochemistry and aging. Green Chem. 2019, 21, 3276–3285. 10.1039/C9GC00304E. [DOI] [Google Scholar]
- EL Knidri H.; Dahmani J.; Addaou A.; Laajeb A.; Lahsini A. Rapid and efficient extraction of chitin and chitosan for scale-up production: Effect of process parameters on deacetylation degree and molecular weight. Int. J. Biol. Macromol. 2019, 139, 1092–1102. 10.1016/j.ijbiomac.2019.08.079. [DOI] [PubMed] [Google Scholar]
- Wang D.; Zhang N.; Meng G.; He J.; Wu F. The effect of form of carboxymethyl-chitosan dressings on biological properties in wound healing. Colloids Surf., B 2020, 194, 111191 10.1016/j.colsurfb.2020.111191. [DOI] [PubMed] [Google Scholar]
- De Masi A.; Tonazzini I.; Masciullo C.; Mezzena R.; Chiellini F.; Puppi D.; Cecchini M. Chitosan films for regenerative medicine: Fabrication methods and mechanical characterization of nanostructured chitosan films. Biophys. Rev. 2019, 11, 807–815. 10.1007/s12551-019-00591-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fakhri E.; Eslami H.; Maroufi P.; Pakdel F.; Taghizadeh S.; Ganbarov K.; Yousefi M.; Tanomand A.; Yousefi B.; Mahmoudi S.; et al. Chitosan biomaterials application in dentistry. Int. J. Biol. Macromol. 2020, 162, 956–974. 10.1016/j.ijbiomac.2020.06.211. [DOI] [PubMed] [Google Scholar]
- Bakhtiari S. S. E.; Karbasi S.; Tabrizi S. A. H.; Ebrahimi-Kahrizsangi R. Chitosan/MWCNTs composite as bone substitute: Physical, mechanical, bioactivity, and biodegradation evaluation. Polym. Compos. 2019, 40, E1622–E1632. 10.1002/pc.25104. [DOI] [Google Scholar]
- Guerrero P.; Muxika A.; Zarandona I.; De La Caba K. Crosslinking of chitosan films processed by compression molding. Carbohydr. Polym. 2019, 206, 820–826. 10.1016/j.carbpol.2018.11.064. [DOI] [PubMed] [Google Scholar]
- Wang K.; Lim P. N.; Tong S. Y.; Thian E. S. Development of grapefruit seed extract-loaded poly (ε-caprolactone)/chitosan films for antimicrobial food packaging. Food Packag. 2019, 22, 100396 10.1016/j.fpsl.2019.100396. [DOI] [Google Scholar]
- Valencia C.; Valencia C. H.; Zuluaga F.; Valencia M. E.; Mina J. H.; Grande-Tovar C. D. Synthesis and application of scaffolds of chitosan-graphene oxide by the freeze-drying method for tissue regeneration. Molecules 2018, 23, 2651. 10.3390/molecules23102651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahbazarab Z.; Teimouri A.; Chermahini A. N.; Azadi M. Fabrication and characterization of nanobiocomposite scaffold of zein/chitosan/nanohydroxyapatite prepared by freeze-drying method for bone tissue engineering. Int. J. Biol. Macromol. 2018, 108, 1017–1027. 10.1016/j.ijbiomac.2017.11.017. [DOI] [PubMed] [Google Scholar]
- Kalantari K.; Afifi A. M.; Jahangirian H.; Webster T. J. Biomedical applications of chitosan electrospun nanofibers as a green polymer–Review. Carbohydr. Polym. 2019, 207, 588–600. 10.1016/j.carbpol.2018.12.011. [DOI] [PubMed] [Google Scholar]
- Mengistu Lemma S.; Bossard F.; Rinaudo M. Preparation of pure and stable chitosan nanofibers by electrospinning in the presence of poly (ethylene oxide). Int. J. Mol. Sci. 2016, 17, 1790. 10.3390/ijms17111790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Udeni Gunathilake T. M. S.; Ching Y. C.; Chuah C. H. Enhancement of curcumin bioavailability using nanocellulose reinforced chitosan hydrogel. Polymers 2017, 9, 64. 10.3390/polym9020064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pandey A. R.; Singh U. S.; Momin M.; Bhavsar C. Chitosan: Application in tissue engineering and skin grafting. J. Polym. Res. 2017, 24, 1–22. 10.1007/s10965-017-1286-4. [DOI] [Google Scholar]
- dos Santos D. M.; de Lacerda Bukzem A.; Campana-Filho S. P. Response surface methodology applied to the study of the microwave-assisted synthesis of quaternized chitosan. Carbohydr. Polym. 2016, 138, 317–326. 10.1016/j.carbpol.2015.11.056. [DOI] [PubMed] [Google Scholar]
- Sebastian J.; Rouissi T.; Brar S. K.; Hegde K.; Verma M. Microwave-assisted extraction of chitosan from Rhizopus oryzae NRRL 1526 biomass. Carbohydr. Polym. 2019, 219, 431–440. 10.1016/j.carbpol.2019.05.047. [DOI] [PubMed] [Google Scholar]
- Islam M. M.; Shahruzzaman M.; Biswas S.; Sakib M. N.; Rashid T. U. Chitosan based bioactive materials in tissue engineering applications-A review. Bioact. Mater. 2020, 5, 164–183. 10.1016/j.bioactmat.2020.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acevedo C. A.; Olguín Y.; Briceño M.; Forero J. C.; Osses N.; Díaz-Calderón P.; Jaques A.; Ortiz R. Design of a biodegradable UV-irradiated gelatin-chitosan/nanocomposed membrane with osteogenic ability for application in bone regeneration. Mater. Sci. Eng., C 2019, 99, 875–886. 10.1016/j.msec.2019.01.135. [DOI] [PubMed] [Google Scholar]
- Kapoor S.; Kundu S. C. Silk protein-based hydrogels: Promising advanced materials for biomedical applications. Acta Biomater. 2016, 31, 17–32. 10.1016/j.actbio.2015.11.034. [DOI] [PubMed] [Google Scholar]
- Wang F.; Wang Y.; Tian C.; Xu S.; Wang R.; Hou K.; Chen W.; Zhao P.; Yu L.; Lu Z.; et al. Fabrication of the FGF1-functionalized sericin hydrogels with cell proliferation activity for biomedical application using genetically engineered Bombyx mori (B. mori) silk. Acta Biomater. 2018, 79, 239–252. 10.1016/j.actbio.2018.08.031. [DOI] [PubMed] [Google Scholar]
- Wen D.-L.; Sun D.-H.; Huang P.; Huang W.; Su M.; Wang Y.; Han M.-D.; Kim B.; Brugger J.; Zhang H.-X.; et al. Recent progress in silk fibroin-based flexible electronics. Microsyst. Nanoeng. 2021, 7, 35. 10.1038/s41378-021-00261-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mehrabani M. G.; Karimian R.; Mehramouz B.; Rahimi M.; Kafil H. S. Preparation of biocompatible and biodegradable silk fibroin/chitin/silver nanoparticles 3D scaffolds as a bandage for antimicrobial wound dressing. Int. J. Biol. Macromol. 2018, 114, 961–971. 10.1016/j.ijbiomac.2018.03.128. [DOI] [PubMed] [Google Scholar]
- Song J. E.; Tripathy N.; Lee D. H.; Park J. H.; Khang G. Quercetin inlaid silk fibroin/hydroxyapatite scaffold promotes enhanced osteogenesis. ACS Appl. Mater. Interfaces 2018, 10, 32955–32964. 10.1021/acsami.8b08119. [DOI] [PubMed] [Google Scholar]
- Yang N.; Moore M. J.; Michael P. L.; Santos M.; Lam Y. T.; Bao S.; Ng M. K.; Rnjak-Kovacina J.; Tan R. P.; Wise S. G. Silk Fibroin Scaffold Architecture Regulates Inflammatory Responses and Engraftment of Bone Marrow-Mononuclear Cells. Adv. Healthc. Mater. 2021, 10, 2100615 10.1002/adhm.202100615. [DOI] [PubMed] [Google Scholar]
- Melke J.; Midha S.; Ghosh S.; Ito K.; Hofmann S. Silk fibroin as biomaterial for bone tissue engineering. Acta Biomater. 2016, 31, 1–16. 10.1016/j.actbio.2015.09.005. [DOI] [PubMed] [Google Scholar]
- Umuhoza D.; Yang F.; Long D.; Hao Z.; Dai J.; Zhao A. Strategies for tuning the biodegradation of silk fibroin-based materials for tissue engineering applications. ACS Biomater. Sci. Eng. 2020, 6, 1290–1310. 10.1021/acsbiomaterials.9b01781. [DOI] [PubMed] [Google Scholar]
- Wang Y.; Rudym D. D.; Walsh A.; Abrahamsen L.; Kim H.-J.; Kim H. S.; Kirker-Head C.; Kaplan D. L. In vivo degradation of three-dimensional silk fibroin scaffolds. Biomaterials 2008, 29, 3415–3428. 10.1016/j.biomaterials.2008.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C.; Song D.; Lu Q.; Hu X.; Kaplan D. L.; Zhu H. Flexibility regeneration of silk fibroin in vitro. Biomacromolecules 2012, 13, 2148–2153. 10.1021/bm300541g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ha S.-W.; Park Y. H.; Hudson S. M. Dissolution of Bombyx m ori silk fibroin in the calcium nitrate tetrahydrate– methanol system and aspects of wet spinning of fibroin solution. Biomacromolecules 2003, 4, 488–496. 10.1021/bm0255948. [DOI] [PubMed] [Google Scholar]
- Su D.; Yao M.; Liu J.; Zhong Y.; Chen X.; Shao Z. Enhancing mechanical properties of silk fibroin hydrogel through restricting the growth of β-sheet domains. ACS Appl. Mater. Interfaces 2017, 9, 17489–17498. 10.1021/acsami.7b04623. [DOI] [PubMed] [Google Scholar]
- Ni T.; Liu M.; Zhang Y.; Cao Y.; Pei R. 3D bioprinting of bone marrow mesenchymal stem cell-laden silk fibroin double network scaffolds for cartilage tissue repair. Bioconjugate Chem. 2020, 31, 1938–1947. 10.1021/acs.bioconjchem.0c00298. [DOI] [PubMed] [Google Scholar]
- Kaushik S.; Thungon P. D.; Goswami P. Silk fibroin: an emerging biocompatible material for application of enzymes and whole cells in bioelectronics and bioanalytical sciences. ACS Biomater. Sci. Eng. 2020, 6, 4337–4355. 10.1021/acsbiomaterials.9b01971. [DOI] [PubMed] [Google Scholar]
- Sergi R.; Bellucci D.; Cannillo V. A review of bioactive glass/natural polymer composites: State of the art. Materials 2020, 13, 5560. 10.3390/ma13235560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon K.-J.; Seok H. Silk protein-based membrane for guided bone regeneration. Appl. Sci. 2018, 8, 1214. 10.3390/app8081214. [DOI] [Google Scholar]
- Qiu Y.; Xu X.; Guo W.; Zhao Y.; Su J.; Chen J. Mesoporous hydroxyapatite nanoparticles mediate the release and bioactivity of BMP-2 for enhanced bone regeneration. ACS Biomater. Sci. Eng. 2020, 6, 2323–2335. 10.1021/acsbiomaterials.9b01954. [DOI] [PubMed] [Google Scholar]
- Saleem M.; Rasheed S.; Yougen C. Silk fibroin/hydroxyapatite scaffold: a highly compatible material for bone regeneration. Sci. Technol. Adv. Mate 2020, 21, 242–266. 10.1080/14686996.2020.1748520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Z. H.; Ma X. L.; Zhao B.; Tian P.; Ma J. X.; Kang J. Y.; Zhang Y.; Guo Y.; Sun L. Naringin-inlaid silk fibroin/hydroxyapatite scaffold enhances human umbilical cord-derived mesenchymal stem cell-based bone regeneration. Cell Prolif. 2021, 54, e13043 10.1111/cpr.13043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eivazzadeh-Keihan R.; Khalili F.; Aliabadi H. A. M.; Maleki A.; Madanchi H.; Ziabari E. Z.; Bani M. S. Alginate hydrogel-polyvinyl alcohol/silk fibroin/magnesium hydroxide nanorods: A novel scaffold with biological and antibacterial activity and improved mechanical properties. Int. J. Biol. Macromol. 2020, 162, 1959–1971. 10.1016/j.ijbiomac.2020.08.090. [DOI] [PubMed] [Google Scholar]
- Wu Z.; Meng Z.; Wu Q.; Zeng D.; Guo Z.; Yao J.; Bian Y.; Gu Y.; Cheng S.; Peng L.; et al. Biomimetic and osteogenic 3D silk fibroin composite scaffolds with nano MgO and mineralized hydroxyapatite for bone regeneration. J. Tissue Eng. 2020, 11, 204173142096779 10.1177/2041731420967791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding X.; Huang Y.; Li X.; Liu S.; Tian F.; Niu X.; Chu Z.; Chen D.; Liu H.; Fan Y. Three-dimensional silk fibroin scaffolds incorporated with graphene for bone regeneration. J. Biomed Mater. Res. A 2021, 109, 515–523. 10.1002/jbm.a.37034. [DOI] [PubMed] [Google Scholar]
- Wang W.; Liu Y.; Yang C.; Jia W.; Qi X.; Liu C.; Li X. Delivery of salvianolic acid B for efficient osteogenesis and angiogenesis from silk fibroin combined with graphene oxide. ACS Biomater. Sci. Eng. 2020, 6, 3539–3549. 10.1021/acsbiomaterials.0c00558. [DOI] [PubMed] [Google Scholar]
- Fei X.; Jia M.; Du X.; Yang Y.; Zhang R.; Shao Z.; Zhao X.; Chen X. Green synthesis of silk fibroin-silver nanoparticle composites with effective antibacterial and biofilm-disrupting properties. Biomacromolecules 2013, 14, 4483–4488. 10.1021/bm4014149. [DOI] [PubMed] [Google Scholar]
- Patil S.; Singh N. Antibacterial silk fibroin scaffolds with green synthesized silver nanoparticles for osteoblast proliferation and human mesenchymal stem cell differentiation. Colloids Surf., B 2019, 176, 150–155. 10.1016/j.colsurfb.2018.12.067. [DOI] [PubMed] [Google Scholar]
- Zhang X.; Chen Z.; Bao H.; Liang J.; Xu S.; Cheng G.; Zhu Y. Fabrication and characterization of silk fibroin/curcumin sustained-release film. Materials 2019, 12, 3340. 10.3390/ma12203340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng Z.; Liu Y.; Xu K.; Sun X.; Yu Q.; Wu Z.; Zhao Z. Biomimetic polydopamine-modified silk fibroin/curcumin nanofibrous scaffolds for chemo-photothermal therapy of bone tumor. ACS omega 2021, 6, 22213–22223. 10.1021/acsomega.1c02903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang C.; Park S.; Kang I.-G.; Kim H.-E.; Han C.-M. Tantalum-coated polylactic acid fibrous membranes for guided bone regeneration. Mater. Sci. Eng., C 2020, 115, 111112 10.1016/j.msec.2020.111112. [DOI] [PubMed] [Google Scholar]
- de Moura N. K.; Martins E. F.; Oliveira R. L. M. S.; de Brito Siqueira I. A. W.; Machado J. P. B.; Esposito E.; Amaral S. S.; de Vasconcellos L. M. R.; Passador F. R.; de Sousa Trichês E. Synergistic effect of adding bioglass and carbon nanotubes on poly (lactic acid) porous membranes for guided bone regeneration. Mater. Sci. Eng., C 2020, 117, 111327 10.1016/j.msec.2020.111327. [DOI] [PubMed] [Google Scholar]
- Abe G. L.; Sasaki J.-I.; Katata C.; Kohno T.; Tsuboi R.; Kitagawa H.; Imazato S. Fabrication of novel poly (lactic acid/caprolactone) bilayer membrane for GBR application. Dent Mater. 2020, 36, 626–634. 10.1016/j.dental.2020.03.013. [DOI] [PubMed] [Google Scholar]
- Budak K.; Sogut O.; Aydemir Sezer U. A review on synthesis and biomedical applications of polyglycolic acid. J. Polym. Res. 2020, 27, 1–19. 10.1007/s10965-020-02187-1. [DOI] [Google Scholar]
- Wang Z.; Liang R.; Jiang X.; Xie J.; Cai P.; Chen H.; Zhan X.; Lei D.; Zhao J.; Zheng L. Electrospun PLGA/PCL/OCP nanofiber membranes promote osteogenic differentiation of mesenchymal stem cells (MSCs). Mater. Sci. Eng., C 2019, 104, 109796 10.1016/j.msec.2019.109796. [DOI] [PubMed] [Google Scholar]
- Pitol-Palin L.; Frigério P. B.; Moura J.; Pilatti L.; de Oliveira L. M. J.; Matsubara E. Y.; Tunchel S.; Shibli J. A.; Blay A.; Saska S.; et al. Performance of Polydioxanone-Based Membrane in Association with 3D-Printed Bioceramic Scaffolds in Bone Regeneration. Polymers 2023, 15, 31. 10.3390/polym15010031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saska S.; Pilatti L.; Silva E. S. d. S.; Nagasawa M. A.; Câmara D.; Lizier N.; Finger E.; Dyszkiewicz Konwińska M.; Kempisty B.; Tunchel S.; et al. Polydioxanone-based membranes for bone regeneration. Polymers 2021, 13, 1685. 10.3390/polym13111685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soltani Dehnavi S.; Mehdikhani M.; Rafienia M.; Bonakdar S. Preparation and in vitro evaluation of polycaprolactone/PEG/bioactive glass nanopowders nanocomposite membranes for GTR/GBR applications. Mater. Sci. Eng., C 2018, 90, 236–247. 10.1016/j.msec.2018.04.065. [DOI] [PubMed] [Google Scholar]
- Ren K.; Wang Y.; Sun T.; Yue W.; Zhang H. Electrospun PCL/gelatin composite nanofiber structures for effective guided bone regeneration membranes. Mater. Sci. Eng., C 2017, 78, 324–332. 10.1016/j.msec.2017.04.084. [DOI] [PubMed] [Google Scholar]
- Van T. T. T.; Makkar P.; Farwa U.; Lee B.-T. Development of a novel polycaprolactone based composite membrane for periodontal regeneration using spin coating technique. J. Biomater. Sci. Polym. Ed. 2022, 33, 783–800. 10.1080/09205063.2021.2020414. [DOI] [PubMed] [Google Scholar]
- Jung R. E.; Mihatovic I.; Cordaro L.; Windisch P.; Friedmann A.; Blanco Carrion J.; Sanz Sanchez I.; Hallman M.; Quirynen M.; Hammerle C. H. F. Comparison of a polyethylene glycol membrane and a collagen membrane for the treatment of bone dehiscence defects at bone level implants—A prospective, randomized, controlled, multicenter clinical trial. Clin. Oral Impl. Res. 2020, 31, 1105–1115. 10.1111/clr.13657. [DOI] [PubMed] [Google Scholar]
- Soltani Dehnavi S.; Mehdikhani M.; Rafienia M.; Bonakdar S. Preparation and in vitro evaluation of polycaprolactone/PEG/bioactive glass nanopowders nanocomposite membranes for GTR/GBR applications. Mater. Sci. Eng., C 2018, 90, 236–247. 10.1016/j.msec.2018.04.065. [DOI] [PubMed] [Google Scholar]
- Tayebi L.; Rasoulianboroujeni M.; Moharamzadeh K.; Almela T. K.; Cui Z.; Ye H. 3D-printed membrane for guided tissue regeneration. Mater. Sci. Eng., C 2018, 84, 148–158. 10.1016/j.msec.2017.11.027. [DOI] [PubMed] [Google Scholar]
- Ramot Y.; Haim-Zada M.; Domb A. J.; Nyska A. Biocompatibility and safety of PLA and its copolymers. Adv. Drug Delivery Rev. 2016, 107, 153–162. 10.1016/j.addr.2016.03.012. [DOI] [PubMed] [Google Scholar]
- Bee S. L.; Hamid Z. A. A. Asymmetric resorbable-based dental barrier membrane for periodontal guided tissue regeneration and guided bone regeneration: A review. J. Biomed. Mater. Res. 2022, 110, 2157–2182. 10.1002/jbm.b.35060. [DOI] [PubMed] [Google Scholar]
- Kharbikar B. N.; Chendke G. S.; Desai T. A. Modulating the foreign body response of implants for diabetes treatment. Adv. Drug Delivery Rev. 2021, 174, 87–113. 10.1016/j.addr.2021.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim B. N.; Ko Y.-G.; Yeo T.; Kim E. J.; Kwon O. K.; Kwon O. H. Guided regeneration of rabbit calvarial defects using silk fibroin nanofiber–poly (glycolic acid) hybrid scaffolds. ACS Biomater. Sci. Eng. 2019, 5, 5266–5272. 10.1021/acsbiomaterials.9b00678. [DOI] [PubMed] [Google Scholar]
- Lin C.-C.; Chiu J.-Y. A novel γ-PGA composite gellan membrane containing glycerol for guided bone regeneration. Mater. Sci. Eng., C 2021, 118, 111404 10.1016/j.msec.2020.111404. [DOI] [PubMed] [Google Scholar]
- Low Y. J.; Andriyana A.; Ang B. C.; Zainal Abidin N. I. Bioresorbable and degradable behaviors of PGA: Current state and future prospects. Polym. Eng. Sci. 2020, 60, 2657–2675. 10.1002/pen.25508. [DOI] [Google Scholar]
- Lin C.-C.; Chiu J.-Y. Glycerol-modified γ-PGA and gellan composite hydrogel materials with tunable physicochemical and thermal properties for soft tissue engineering application. Polymer 2021, 230, 124049 10.1016/j.polymer.2021.124049. [DOI] [Google Scholar]
- Li J.; Washington M. A.; Bell K. L.; Weiss R. M.; Rothstein S. N.; Little S. R.; Edenborn H. M.; Meyer T. Y., Engineering hydrolytic degradation behavior of poly(lactic-co-glycolic acid) through precise control of monomer sequence. In Sequence-Controlled Polymers: Synthesis, Self-Assembly, and Properties; ACS Publications: 2014; pp 271–286. [Google Scholar]
- Li J.; Rothstein S. N.; Little S. R.; Edenborn H. M.; Meyer T. Y. The effect of monomer order on the hydrolysis of biodegradable poly (lactic-co-glycolic acid) repeating sequence copolymers. J. Am. Chem. Soc. 2012, 134, 16352–16359. 10.1021/ja306866w. [DOI] [PubMed] [Google Scholar]
- Yang Z.; Liu L.; Su L.; Wu X.; Wang Y.; Liu L.; Lin X. Design of a zero-order sustained release PLGA microspheres for palonosetron hydrochloride with high encapsulation efficiency. Int. J. Pharm. 2020, 575, 119006 10.1016/j.ijpharm.2019.119006. [DOI] [PubMed] [Google Scholar]
- Zhang Z.; Wang X.; Li B.; Hou Y.; Cai Z.; Yang J.; Li Y. Paclitaxel-loaded PLGA microspheres with a novel morphology to facilitate drug delivery and antitumor efficiency. RSC Adv. 2018, 8, 3274–3285. 10.1039/C7RA12683B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babilotte J.; Martin B.; Guduric V.; Bareille R.; Agniel R.; Roques S.; Héroguez V.; Dussauze M.; Gaudon M.; Le Nihouannen D.; et al. Development and characterization of a PLGA-HA composite material to fabricate 3D-printed scaffolds for bone tissue engineering. Mater. Sci. Eng., C 2021, 118, 111334 10.1016/j.msec.2020.111334. [DOI] [PubMed] [Google Scholar]
- Tang Y.; Wang H.; Sun Y.; Jiang Y.; Fang S.; Kan Z.; Lu Y.; Liu S.; Zhou X.; Li Z. Using platelet-rich plasma hydrogel to deliver mesenchymal stem cells into three-dimensional PLGA scaffold for cartilage tissue engineering. ACS Appl. Bio Mater. 2021, 4, 8607–8614. 10.1021/acsabm.1c01160. [DOI] [PubMed] [Google Scholar]
- Rahman C. V.; Kuhn G.; White L. J.; Kirby G. T.; Varghese O. P.; McLaren J. S.; Cox H. C.; Rose F. R.; Müller R.; Hilborn J.; et al. PLGA/PEG-hydrogel composite scaffolds with controllable mechanical properties. J. Biomed. Mater. Res. 2013, 101, 648–655. 10.1002/jbm.b.32867. [DOI] [PubMed] [Google Scholar]
- Du B.; Yin H.; Chen Y.; Lin W.; Wang Y.; Zhao D.; Wang G.; He X.; Li J.; Li Z.; et al. A waterborne polyurethane 3D scaffold containing PLGA with a controllable degradation rate and an anti-inflammatory effect for potential applications in neural tissue repair. J. Mater. Chem. B 2020, 8, 4434–4446. 10.1039/D0TB00656D. [DOI] [PubMed] [Google Scholar]
- Sun F.; Sun X.; Wang H.; Li C.; Zhao Y.; Tian J.; Lin Y. Application of 3D-printed, PLGA-based scaffolds in bone tissue engineering. Int. J. Mol. Sci. 2022, 23, 5831. 10.3390/ijms23105831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giammanco G.; Martínez de Ilarduya A.; Alla A.; Muñoz-Guerra S. Hydrolyzable aromatic copolyesters of p-dioxanone. Biomacromolecules 2010, 11, 2512–2520. 10.1021/bm1007025. [DOI] [PubMed] [Google Scholar]
- Wang B.; Qin C.; Liu Y.; Zhang Y.; Feng C.; Mi F.; Zhu H. Positive space acquiring asymmetric membranes for guiding alveolar bone regeneration under infectious conditions. Biomaterials Advances 2023, 145, 213252 10.1016/j.bioadv.2022.213252. [DOI] [PubMed] [Google Scholar]
- Heidari B. S.; Chen P.; Ruan R.; Davachi S. M.; Al-Salami H.; Pardo E. D. J.; Zheng M.; Doyle B. A novel biocompatible polymeric blend for applications requiring high toughness and tailored degradation rate. J. Mater. Chem. B 2021, 9, 2532–2546. 10.1039/D0TB02971H. [DOI] [PubMed] [Google Scholar]
- Martins J. A.; Lach A. A.; Morris H. L.; Carr A. J.; Mouthuy P.-A. Polydioxanone implants: a systematic review on safety and performance in patients. J. Biomater. Appl. 2020, 34, 902–916. 10.1177/0885328219888841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Surnar B.; Jayakannan M. Structural engineering of biodegradable PCL block copolymer nanoassemblies for enzyme-controlled drug delivery in cancer cells. ACS Biomater. Sci. Eng. 2016, 2, 1926–1941. 10.1021/acsbiomaterials.6b00310. [DOI] [PubMed] [Google Scholar]
- Holländer J.; Genina N.; Jukarainen H.; Khajeheian M.; Rosling A.; Mäkilä E.; Sandler N. Three-dimensional printed PCL-based implantable prototypes of medical devices for controlled drug delivery. J. Pharm. Sci. 2016, 105, 2665–2676. 10.1016/j.xphs.2015.12.012. [DOI] [PubMed] [Google Scholar]
- Archer E.; Torretti M.; Madbouly S. Biodegradable polycaprolactone (PCL) based polymer and composites. Phys. Sci. Rev. 2023, 000010151520200074. 10.1515/psr-2020-0074. [DOI] [Google Scholar]
- Jing X.; Mi H.-Y.; Huang H.-X.; Turng L.-S. Shape memory thermoplastic polyurethane (TPU)/poly (ε-caprolactone)(PCL) blends as self-knotting sutures. J. Mech Behav Biomed Mater. 2016, 64, 94–103. 10.1016/j.jmbbm.2016.07.023. [DOI] [PubMed] [Google Scholar]
- Liu F.; Vyas C.; Poologasundarampillai G.; Pape I.; Hinduja S.; Mirihanage W.; Bartolo P. Structural evolution of PCL during melt extrusion 3D printing. Macromol. Mater. Eng. 2018, 303, 1700494 10.1002/mame.201700494. [DOI] [Google Scholar]
- Wang F.; Tankus E. B.; Santarella F.; Rohr N.; Sharma N.; Märtin S.; Michalscheck M.; Maintz M.; Cao S.; Thieringer F. M. Fabrication and characterization of PCL/HA filament as a 3D printing material using thermal extrusion technology for bone tissue engineering. Polymers 2022, 14, 669. 10.3390/polym14040669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espinoza S. M.; Patil H. I.; San Martin Martinez E.; Casañas Pimentel R.; Ige P. P. Poly-ε-caprolactone (PCL), a promising polymer for pharmaceutical and biomedical applications: Focus on nanomedicine in cancer. Int. J. Polym. Mater. Polym. 2020, 69, 85–126. 10.1080/00914037.2018.1539990. [DOI] [Google Scholar]
- Ezati M.; Safavipour H.; Houshmand B.; Faghihi S. Development of a PCL/gelatin/chitosan/β-TCP electrospun composite for guided bone regeneration. Prog. Biomater 2018, 7, 225–237. 10.1007/s40204-018-0098-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bang L. T.; Tsuru K.; Munar M.; Ishikawa K.; Othman R. Mechanical behavior and cell response of PCL coated α-TCP foam for cancellous-type bone replacement. Ceram. Int. 2013, 39, 5631–5637. 10.1016/j.ceramint.2012.12.079. [DOI] [Google Scholar]
- Yin S.; Zhang W.; Zhang Z.; Jiang X. Recent advances in scaffold design and material for vascularized tissue-engineered bone regeneration. Adv. Healthc. Mater. 2019, 8, 1801433 10.1002/adhm.201801433. [DOI] [PubMed] [Google Scholar]
- Lee S. J.; Lee D.; Yoon T. R.; Kim H. K.; Jo H. H.; Park J. S.; Lee J. H.; Kim W. D.; Kwon I. K.; Park S. A. Surface modification of 3D-printed porous scaffolds via mussel-inspired polydopamine and effective immobilization of rhBMP-2 to promote osteogenic differentiation for bone tissue engineering. Acta Biomater. 2016, 40, 182–191. 10.1016/j.actbio.2016.02.006. [DOI] [PubMed] [Google Scholar]
- Jafari A.; Amirsadeghi A.; Hassanajili S.; Azarpira N. Bioactive antibacterial bilayer PCL/gelatin nanofibrous scaffold promotes full-thickness wound healing. Int. J. Pharm. 2020, 583, 119413 10.1016/j.ijpharm.2020.119413. [DOI] [PubMed] [Google Scholar]
- Cui C.; Sun S.; Li X.; Chen S.; Wu S.; Zhou F.; Ma J. Optimizing the chitosan-PCL based membranes with random/aligned fiber structure for controlled ciprofloxacin delivery and wound healing. Int. J. Biol. Macromol. 2022, 205, 500–510. 10.1016/j.ijbiomac.2022.02.118. [DOI] [PubMed] [Google Scholar]
- Rather H. A.; Thakore R.; Singh R.; Jhala D.; Singh S.; Vasita R. Antioxidative study of Cerium Oxide nanoparticle functionalised PCL-Gelatin electrospun fibers for wound healing application. Bioact. Mater. 2018, 3, 201–211. 10.1016/j.bioactmat.2017.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z.; Wu C.; Shi H.; Luo X.; Sun H.; Wang Q.; Zhang D. Advances in barrier membranes for guided bone regeneration techniques. Front. Bioeng. Biotechnol. 2022, 10, 1047. 10.3389/fbioe.2022.921576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J.; Wang L.; Zhou Z.; Lai H.; Xu P.; Liao L.; Wei J. Biodegradable Polymer Membranes Applied in Guided Bone/Tissue Regeneration: A Review. Polymers 2016, 8, 115. 10.3390/polym8040115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoma D. S.; Weber F. E.; Bienz S. P.; Ge Y.; Hämmerle C. H. F.; Jung R. E. Biodegradation and tissue integration of various polyethylene glycol matrices: a comparative study in rabbits. Clin. Oral Impl. Res. 2017, 28, e244–e251. 10.1111/clr.13004. [DOI] [PubMed] [Google Scholar]
- Yue S.; He H.; Li B.; Hou T. Hydrogel as a Biomaterial for Bone Tissue Engineering: A Review. Nanomaterials 2020, 10, 1511. 10.3390/nano10081511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal P.; Tucci M. A.; Fan L. W.; Bollavarapu R.; Lee J. W.; Salazar Marocho S. M.; Janorkar A. V. Functionalized Collagen/Elastin-like Polypeptide Hydrogels for Craniofacial Bone Regeneration. Adv. Healthc. Mater. 2023, 12, 2202477 10.1002/adhm.202202477. [DOI] [PubMed] [Google Scholar]
- Zhang D.; Chen Q.; Shi C.; Chen M.; Ma K.; Wan J.; Liu R. Dealing with the foreign-body response to implanted biomaterials: strategies and applications of new materials. Adv. Funct. Mater. 2021, 31, 2007226 10.1002/adfm.202007226. [DOI] [Google Scholar]
- Chandorkar Y.; K R.; Basu B. The foreign body response demystified. ACS Biomater. Sci. Eng. 2019, 5, 19–44. 10.1021/acsbiomaterials.8b00252. [DOI] [PubMed] [Google Scholar]
- Ekdahl K. N.; Lambris J. D.; Elwing H.; Ricklin D.; Nilsson P. H.; Teramura Y.; Nicholls I. A.; Nilsson B. Innate immunity activation on biomaterial surfaces: a mechanistic model and coping strategies. Adv. Drug Delivery Rev. 2011, 63, 1042–1050. 10.1016/j.addr.2011.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin K.; Luo Z.; Zhang B.; Pang Z. Biomimetic nanoparticles for inflammation targeting. Acta Pharm. Sin. B 2018, 8, 23–33. 10.1016/j.apsb.2017.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isali I.; McClellan P.; Shankar E.; Gupta S.; Jain M.; Anderson J. M.; Hijaz A.; Akkus O. Genipin guides and sustains the polarization of macrophages to the pro-regenerative M2 subtype via activation of the pSTAT6-PPAR-gamma pathway. Acta Biomater. 2021, 131, 198–210. 10.1016/j.actbio.2021.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown B. N.; Badylak S. F. Expanded applications, shifting paradigms and an improved understanding of host–biomaterial interactions. Acta Biomater. 2013, 9, 4948–4955. 10.1016/j.actbio.2012.10.025. [DOI] [PubMed] [Google Scholar]
- Dewey M. J.; Harley B. A. Biomaterial design strategies to address obstacles in craniomaxillofacial bone repair. RSC Adv. 2021, 11, 17809–17827. 10.1039/D1RA02557K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu C.; Liu L.; Wang Y.; Wei S.; Wang Y.; Man Y.; Qu Y. Macrophage phenotype in the epigallocatechin-3-gallate (EGCG)-modified collagen determines foreign body reaction. J. Tissue Eng. Regen Med. 2018, 12, 1499–1507. 10.1002/term.2687. [DOI] [PubMed] [Google Scholar]
- Almatroodi S. A.; Almatroudi A.; Khan A. A.; Alhumaydhi F. A.; Alsahli M. A.; Rahmani A. H. Potential therapeutic targets of epigallocatechin gallate (EGCG), the most abundant catechin in green tea, and its role in the therapy of various types of cancer. Molecules 2020, 25, 3146. 10.3390/molecules25143146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kharaziha M.; Baidya A.; Annabi N. Rational design of immunomodulatory hydrogels for chronic wound healing. Adv. Mater. 2021, 33, 2100176 10.1002/adma.202100176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin Yavari S.; Castenmiller S. M.; Strijp J. A. G.; Croes M. Combating implant infections: shifting focus from bacteria to host. Adv. Mater. 2020, 32, 2002962 10.1002/adma.202002962. [DOI] [PubMed] [Google Scholar]
- Lee J.; Byun H.; Madhurakkat Perikamana S. K.; Lee S.; Shin H. Current advances in immunomodulatory biomaterials for bone regeneration. Adv. Healthc. Mater. 2018, 8, 1801106 10.1002/adhm.201801106. [DOI] [PubMed] [Google Scholar]
- Wang Y.; Zhang H.; Hu Y.; Jing Y.; Geng Z.; Su J. Bone Repair Biomaterials: A Perspective from Immunomodulatory. Adv. Funct. Mater. 2022, 32, 2208639. 10.1002/adfm.202208639. [DOI] [Google Scholar]
- Li S.; Cui Y.; Liu H.; Tian Y.; Wang G.; Fan Y.; Wang J.; Wu D.; Wang Y. Application of bioactive metal ions in the treatment of bone defects. J. Mater. Chem. B 2022, 10, 9369. 10.1039/D2TB01684B. [DOI] [PubMed] [Google Scholar]
- Akshaya S.; Rowlo P. K.; Dukle A.; Nathanael A. J. Antibacterial Coatings for Titanium Implants: Recent Trends and Future Perspectives. Antibiotics 2022, 11, 1719. 10.3390/antibiotics11121719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Z.; Chen L.; Liu R.; Lin Y.; Chen S.; Lu S.; Lin Z.; Chen Z.; Wu C.; Xiao Y. The osteoimmunomodulatory property of a barrier collagen membrane and its manipulation via coating nanometer-sized bioactive glass to improve guided bone regeneration. Biomater. Sci. 2018, 6, 1007–1019. 10.1039/C7BM00869D. [DOI] [PubMed] [Google Scholar]
- Goltzman D.; Hendy G. N. The calcium-sensing receptor in bone—mechanistic and therapeutic insights. Nat. Rev. Endocrinol. 2015, 11, 298–307. 10.1038/nrendo.2015.30. [DOI] [PubMed] [Google Scholar]
- Hao J.; Tang J.; Zhang L.; Li X.; Hao L. The crosstalk between calcium ions and aldosterone contributes to inflammation, apoptosis, and calcification of VSMC via the AIF-1/NF-κB pathway in uremia. Oxid. Med. Cell. Longev. 2020, 2020, 1. 10.1155/2020/3431597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skrajnowska D.; Bobrowska-Korczak B. Role of zinc in immune system and anti-cancer defense mechanisms. Nutrients 2019, 11, 2273. 10.3390/nu11102273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Veríssimo D.; Leitão R.; Ribeiro R.; Figueiró S.; Sombra A.; Góes J.; Brito G. Polyanionic collagen membranes for guided tissue regeneration: Effect of progressive glutaraldehyde cross-linking on biocompatibility and degradation. Acta Biomater. 2010, 6, 4011–4018. 10.1016/j.actbio.2010.04.012. [DOI] [PubMed] [Google Scholar]
- Wu Y.; Chen S.; Luo P.; Deng S.; Shan Z.; Fang J.; Liu X.; Xie J.; Liu R.; Wu S.; et al. Optimizing the bio-degradability and biocompatibility of a biogenic collagen membrane through cross-linking and zinc-doped hydroxyapatite. Acta Biomater. 2022, 143, 159–172. 10.1016/j.actbio.2022.02.004. [DOI] [PubMed] [Google Scholar]
- Krishnakumar G. S.; Sampath S.; Muthusamy S.; John M. A. Importance of crosslinking strategies in designing smart biomaterials for bone tissue engineering: A systematic review. Mater. Sci. Eng., C 2019, 96, 941–954. 10.1016/j.msec.2018.11.081. [DOI] [PubMed] [Google Scholar]
- Markowska-Szczupak A.; Endo-Kimura M.; Paszkiewicz O.; Kowalska E. Are titania photocatalysts and titanium implants safe? Review on the toxicity of titanium compounds. Nanomaterials 2020, 10, 2065. 10.3390/nano10102065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ansarizadeh M.; Mashayekhan S.; Saadatmand M. Fabrication, modeling and optimization of lyophilized advanced platelet rich fibrin in combination with collagen-chitosan as a guided bone regeneration membrane. Int. J. Biol. Macromol. 2019, 125, 383–391. 10.1016/j.ijbiomac.2018.12.078. [DOI] [PubMed] [Google Scholar]
- Karageorgiou V.; Kaplan D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. 10.1016/j.biomaterials.2005.02.002. [DOI] [PubMed] [Google Scholar]
- Takahashi Y.; Tabata Y. Effect of the fiber diameter and porosity of non-woven PET fabrics on the osteogenic differentiation of mesenchymal stem cells. J. Biomater. Sci. Polym. Ed. 2004, 15, 41–57. 10.1163/156856204322752228. [DOI] [PubMed] [Google Scholar]
- Chen Z.; Zhang Q.; Li H.; Wei Q.; Zhao X.; Chen F. Elastin-like polypeptide modified silk fibroin porous scaffold promotes osteochondral repair. Bioact. Mater. 2021, 6, 589–601. 10.1016/j.bioactmat.2020.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omar O.; Dahlin A.; Gasser A.; Dahlin C. Tissue dynamics and regenerative outcome in two resorbable non-cross-linked collagen membranes for guided bone regeneration: A preclinical molecular and histological study in vivo. Clin. Oral Implants Res. 2018, 29, 7–19. 10.1111/clr.13032. [DOI] [PubMed] [Google Scholar]
- Gurumurthy B.; Griggs J. A.; Janorkar A. V. Optimization of collagen-elastin-like polypeptide composite tissue engineering scaffolds using response surface methodology. J. Mech Behav Biomed Mater. 2018, 84, 116–125. 10.1016/j.jmbbm.2018.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter P.; Rahman S. M.; Bhattarai N. Facile fabrication of aloe vera containing PCL nanofibers for barrier membrane application. J. Biomater. Sci. Polym. Ed. 2016, 27, 692–708. 10.1080/09205063.2016.1152857. [DOI] [PubMed] [Google Scholar]
- Hadj-Hamou A. S.; Metref F.; Yahiaoui F. Thermal stability and decomposition kinetic studies of antimicrobial PCL/nanoclay packaging films. Polym. Bull. 2017, 74, 3833–3853. 10.1007/s00289-017-1929-y. [DOI] [Google Scholar]
- Liberato M.; Kogikoski S.; Da Silva E.; De Araujo D.; Guha S.; Alves W. Polycaprolactone fibers with self-assembled peptide micro/nanotubes: a practical route towards enhanced mechanical strength and drug delivery applications. J. Mater. Chem. B 2016, 4, 1405–1413. 10.1039/C5TB02240A. [DOI] [PubMed] [Google Scholar]
- Hamlekhan A.; Moztarzadeh F.; Mozafari M.; Azami M.; Nezafati N. Preparation of laminated poly (ε-caprolactone)-gelatin-hydroxyapatite nanocomposite scaffold bioengineered via compound techniques for bone substitution. Biomatter 2011, 1, 91–101. 10.4161/biom.1.1.17445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joo G.; Park M.; Park S.-s.; Tripathi G.; Lee B.-T. Tailored alginate/PCL-gelatin-β-TCP membrane for guided bone regeneration. Biomed. Mater. 2022, 17, 045011 10.1088/1748-605X/ac6bd8. [DOI] [PubMed] [Google Scholar]
- Shen H.; Zhang C.; Zhang C.; Shi J.; Shen S. G.; Yuan Y.; Si J. A Novel Immunoregulatory PEGylated Poly (glycerol sebacate)/β-TCP Membrane for Application in Guided Bone Regeneration. Adv. Mater. Interfaces 2022, 9, 2101218 10.1002/admi.202101218. [DOI] [Google Scholar]
- Gnanasampanthan T.; Karthäuser J. F.; Spöllmann S.; Wanka R.; Becker H.-W.; Rosenhahn A. Amphiphilic Alginate-Based Layer-by-Layer Coatings Exhibiting Resistance against Nonspecific Protein Adsorption and Marine Biofouling. ACS Appl. Mater. Interfaces 2022, 14, 16062–16073. 10.1021/acsami.2c01809. [DOI] [PubMed] [Google Scholar]
- Lv C.; Zhou X.; Wang P.; Li J.; Wu Z.; Jiao Z.; Guo M.; Wang Z.; Wang Y.; Wang L.; et al. Biodegradable alginate-based sponge with antibacterial and shape memory properties for penetrating wound hemostasis. Compos. B. Eng. 2022, 247, 110263 10.1016/j.compositesb.2022.110263. [DOI] [Google Scholar]
- Ahmed A.; Getti G.; Boateng J. Medicated multi-targeted alginate-based dressings for potential treatment of mixed bacterial-fungal infections in diabetic foot ulcers. Int. J. Pharm. 2021, 606, 120903 10.1016/j.ijpharm.2021.120903. [DOI] [PubMed] [Google Scholar]
- Gyles D. A.; Castro L. D.; Silva J. O. C. Jr; Ribeiro-Costa R. M. A review of the designs and prominent biomedical advances of natural and synthetic hydrogel formulations. Eur. Polym. J. 2017, 88, 373–392. 10.1016/j.eurpolymj.2017.01.027. [DOI] [Google Scholar]
- Kiran A. S. K.; Kumar T. S.; Sanghavi R.; Doble M.; Ramakrishna S. Antibacterial and bioactive surface modifications of titanium implants by PCL/TiO2 nanocomposite coatings. Nanomaterials 2018, 8, 860. 10.3390/nano8100860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park H. J.; Min K. D.; Lee M. C.; Kim S. H.; Lee O. J.; Ju H. W.; Moon B. M.; Lee J. M.; Park Y. R.; Kim D. W.; et al. Fabrication of 3D porous SF/β-TCP hybrid scaffolds for bone tissue reconstruction. J. Biomed Mater. Res. A 2016, 104, 1779–1787. 10.1002/jbm.a.35711. [DOI] [PubMed] [Google Scholar]
- Ye X.; Zhang Y.; Liu T.; Chen Z.; Chen W.; Wu Z.; Wang Y.; Li J.; Li C.; Jiang T.; et al. Beta-tricalcium phosphate enhanced mechanical and biological properties of 3D-printed polyhydroxyalkanoates scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2022, 209, 1553–1561. 10.1016/j.ijbiomac.2022.04.056. [DOI] [PubMed] [Google Scholar]
- Cao L.; Duan P.-G.; Wang H.-R.; Li X.-L.; Yuan F.-L.; Fan Z.-Y.; Li S.-M.; Dong J. Degradation and osteogenic potential of a novel poly (lactic acid)/nano-sized β-tricalcium phosphate scaffold. Int. J. Nanomedicine 2012, 5881–5888. 10.2147/IJN.S38127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paladini F.; Pollini M. Novel Approaches and Biomaterials for Bone Tissue Engineering: A Focus on Silk Fibroin. Materials 2022, 15, 6952. 10.3390/ma15196952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geão C.; Costa-Pinto A. R.; Cunha-Reis C.; Ribeiro V. P.; Vieira S.; Oliveira J. M.; Reis R. L.; Oliveira A. L. Thermal annealed silk fibroin membranes for periodontal guided tissue regeneration. J. Mater. Sci. Mater. Med. 2019, 30, 1–19. 10.1007/s10856-019-6225-y. [DOI] [PubMed] [Google Scholar]
- Zhu C.; Chiu S.; Nakas J. P.; Nomura C. T. Bioplastics from waste glycerol derived from biodiesel industry. J. Appl. Polym. Sci. 2013, 130, 1–13. 10.1002/app.39157. [DOI] [Google Scholar]
- Khabbaz B.; Solouk A.; Mirzadeh H. Polyvinyl alcohol/soy protein isolate nanofibrous patch for wound-healing applications. Prog. Biomater 2019, 8, 185–196. 10.1007/s40204-019-00120-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Julinová M.; Vaňharová L.; Jurča M. Water-soluble polymeric xenobiotics–Polyvinyl alcohol and polyvinylpyrrolidon–And potential solutions to environmental issues: A brief review. J. Environ. Manage. 2018, 228, 213–222. 10.1016/j.jenvman.2018.09.010. [DOI] [PubMed] [Google Scholar]
- Golbaten-Mofrad H.; Salehi M. H.; Jafari S. H.; Goodarzi V.; Entezari M.; Hashemi M. Preparation and properties investigation of biodegradable poly (glycerol sebacate-co-gelatin) containing nanoclay and graphene oxide for soft tissue engineering applications. J. Biomed. Mater. Res. 2022, 110, 2241–2257. 10.1002/jbm.b.35073. [DOI] [PubMed] [Google Scholar]
- Mathew A.; Vaquette C.; Hashimi S.; Rathnayake I.; Huygens F.; Hutmacher D. W.; Ivanovski S. Antimicrobial and immunomodulatory surface-functionalized electrospun membranes for bone regeneration. Adv. Healthc. Mater. 2017, 6, 1601345 10.1002/adhm.201601345. [DOI] [PubMed] [Google Scholar]
- L. Abe G.; Tsuboi R.; Kitagawa H.; Sasaki J. I.; Li A.; Kohno T.; Imazato S. Poly (lactic acid/caprolactone) bilayer membrane blocks bacterial penetration. J. Periodontal Res. 2022, 57, 510–518. 10.1111/jre.12980. [DOI] [PubMed] [Google Scholar]
- Jin S.; Li J.; Wang J.; Jiang J.; Zuo Y.; Li Y.; Yang F. Electrospun silver ion-loaded calcium phosphate/chitosan antibacterial composite fibrous membranes for guided bone regeneration. Int. J. Nanomedicine 2018, 13, 4591. 10.2147/IJN.S167793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S.-Y.; Kim Y.-K.; Jang Y.-S.; Lee M.-H. Enhancement of Biofunctionalization by Loading Manuka Oil on TiO2 Nanotubes. Nanomaterials 2022, 12, 569. 10.3390/nano12030569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Giau V.; An S. S. A.; Hulme J. Recent advances in the treatment of pathogenic infections using antibiotics and nano-drug delivery vehicles. Drug Des Devel Ther 2019, 13, 327–343. 10.2147/DDDT.S190577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mi G.; Shi D.; Wang M.; Webster T. J. Reducing bacterial infections and biofilm formation using nanoparticles and nanostructured antibacterial surfaces. Adv. Healthc. Mater. 2018, 7, 1800103 10.1002/adhm.201800103. [DOI] [PubMed] [Google Scholar]
- He M.; Wang Q.; Xie L.; Wu H.; Zhao W.; Tian W. Hierarchically multi-functionalized graded membrane with enhanced bone regeneration and self-defensive antibacterial characteristics for guided bone regeneration. J. Chem. Eng. 2020, 398, 125542 10.1016/j.cej.2020.125542. [DOI] [Google Scholar]
- L. Abe G.; Tsuboi R.; Kitagawa H.; Sasaki J. I.; Li A.; Kohno T.; Imazato S. Poly(lactic acid/caprolactone) bilayer membrane blocks bacterial penetration. J. Periodontal Res. 2022, 57, 510–518. 10.1111/jre.12980. [DOI] [PubMed] [Google Scholar]
- Shirzaei Sani E.; Portillo-Lara R.; Spencer A.; Yu W.; Geilich B. M.; Noshadi I.; Webster T. J.; Annabi N. Engineering adhesive and antimicrobial hyaluronic acid/elastin-like polypeptide hybrid hydrogels for tissue engineering applications. ACS Biomater. Sci. Eng. 2018, 4, 2528–2540. 10.1021/acsbiomaterials.8b00408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thoma D. S.; Cha J. K.; Sapata V. M.; Jung R. E.; Hüsler J.; Jung U. W. Localized bone regeneration around dental implants using recombinant bone morphogenetic protein-2 and platelet-derived growth factor-BB in the canine. Clin. Oral Implants Res. 2017, 28, 1334–1341. 10.1111/clr.12989. [DOI] [PubMed] [Google Scholar]
- Thoma D. S.; Lim H. C.; Sapata V. M.; Yoon S. R.; Jung R. E.; Jung U. W. Recombinant bone morphogenetic protein-2 and platelet-derived growth factor-BB for localized bone regeneration. Histologic and radiographic outcomes of a rabbit study. Clin. Oral Implants Res. 2017, 28, e236–e243. 10.1111/clr.13002. [DOI] [PubMed] [Google Scholar]
- Wang R.; Liu W.; Guo H.; Ge S.; Huang H.; Yang P. Alveolar ridge preservation with fibroblast growth factor-2 modified acellular dermal matrix membrane and a bovine-derived xenograft: An experimental in vivo study. Clin Oral Implants Res. 2021, 32, 808–817. 10.1111/clr.13749. [DOI] [PubMed] [Google Scholar]
- Fujioka-Kobayashi M.; Kobayashi E.; Schaller B.; Mottini M.; Miron R. J.; Saulacic N. Effect of recombinant human bone morphogenic protein 9 (rhBMP9) loaded onto bone grafts versus barrier membranes on new bone formation in a rabbit calvarial defect model. J. Biomed Mater. Res. A 2017, 105, 2655–2661. 10.1002/jbm.a.36125. [DOI] [PubMed] [Google Scholar]
- Nasiri N.; Ceramidas A.; Mukherjee S.; Panneerselvan A.; Nisbet D. R.; Tricoli A. Ultra-Porous Nanoparticle Networks: A Biomimetic Coating Morphology for Enhanced Cellular Response and Infiltration. Sci. Rep. 2016, 6, 24305. 10.1038/srep24305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasiri N.; Mukherjee S.; Panneerselvan A.; Nisbet D. R.; Tricoli A. Optimally Hierarchical Nanostructured Hydroxyapatite Coatings for Superior Prosthesis Biointegration. ACS Appl. Mater. Interfaces 2018, 10, 24840–24849. 10.1021/acsami.8b08029. [DOI] [PubMed] [Google Scholar]
- Zhang Y.; Yang S.; Zhou W.; Fu H.; Qian L.; Miron R. J. Addition of a synthetically fabricated osteoinductive biphasic calcium phosphate bone graft to BMP2 improves new bone formation. Clin Implant Dent Relat Res. 2016, 18, 1238–1247. 10.1111/cid.12384. [DOI] [PubMed] [Google Scholar]
- Elgali I.; Omar O.; Dahlin C.; Thomsen P. Guided bone regeneration: materials and biological mechanisms revisited. Eur. J. Oral Sci. 2017, 125, 315–337. 10.1111/eos.12364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen F.-M.; Zhang J.; Zhang M.; An Y.; Chen F.; Wu Z.-F. A review on endogenous regenerative technology in periodontal regenerative medicine. BIOMATERIALS 2010, 31, 7892–7927. 10.1016/j.biomaterials.2010.07.019. [DOI] [PubMed] [Google Scholar]
- Fassini D.; Wilkie I. C.; Pozzolini M.; Ferrario C.; Sugni M.; Rocha M. S.; Giovine M.; Bonasoro F.; Silva T. H.; Reis R. L. Diverse and Productive Source of Biopolymer Inspiration: Marine Collagens. BIOMACROMOLECULES 2021, 22, 1815–1834. 10.1021/acs.biomac.1c00013. [DOI] [PubMed] [Google Scholar]
- Nijhuis W. H.; Eastwood D. M.; Allgrove J.; Hvid I.; Weinans H. H.; Bank R. A.; Sakkers R. J. Current concepts in osteogenesis imperfecta: Bone structure, biomechanics and medical management. J. Child Orthop 2019, 13, 1–11. 10.1302/1863-2548.13.180190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodsell D.http://pdb101.rcsb.org/motm/4, Accessed 28 May 2023. Data Bank April 2000.
- Mbese Z.; Alven S.; Aderibigbe B. A. Collagen-Based Nanofibers for Skin Regeneration and Wound Dressing Applications. Polymers 2021, 13, 4368. 10.3390/polym13244368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikolov S.; Petrov M.; Lymperakis L.; Friák M.; Sachs C.; Fabritius H.-O.; Raabe D.; Neugebauer J. Revealing the Design Principles of High-Performance Biological Composites Using Ab initio and Multiscale Simulations: The Example of Lobster Cuticle. Adv. Mater. 2010, 22, 519–526. 10.1002/adma.200902019. [DOI] [PubMed] [Google Scholar]
- Ribeiro D.; Yuto G.; Titotto S. L. M. C. Analysis about the behavior and modeling of PH-sensitive hydrogels with different ratios of chitosan and polyvinylpyrrolidone. J. mater. sci. chem. eng. 2019, 7, 64–76. 10.4236/msce.2019.78008. [DOI] [Google Scholar]
- López-García J.; Lehocký M.; Humpolíček P.; Sáha P. HaCaT Keratinocytes Response on Antimicrobial Atelocollagen Substrates: Extent of Cytotoxicity, Cell Viability and Proliferation. J. Fun. Biomater. 2014, 5, 43–57. 10.3390/jfb5020043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debari M. K.; Abbott R. D. Microscopic considerations for optimizing silk biomaterials. Wiley Interdiscip. Rev.: Nanomed. Nanobiotechnology 2019, 11, e1534 10.1002/wnan.1534. [DOI] [PubMed] [Google Scholar]
- Tan X. H.; Liu L.; Mitryashkin A.; Wang Y.; Goh J. C. H. Silk Fibroin as a Bioink – A Thematic Review of Functionalization Strategies for Bioprinting Applications. ACS Biomater. Sci. Eng. 2022, 8, 3242. 10.1021/acsbiomaterials.2c00313. [DOI] [PubMed] [Google Scholar]
- He Y.-X.; Zhang N.-N.; Li W.-F.; Jia N.; Chen B.-Y.; Zhou K.; Zhang J.; Chen Y.; Zhou C.-Z. N-Terminal Domain of Bombyx mori Fibroin Mediates the Assembly of Silk in Response to pH Decrease. J. Mol. Biol. 2012, 418, 197–207. 10.1016/j.jmb.2012.02.040. [DOI] [PubMed] [Google Scholar]
- Dadras Chomachayi M.; Jalali-Arani A.; Martínez Urreaga J. A Comparison of the Effect of Silk Fibroin Nanoparticles and Microfibers on the Reprocessing and Biodegradability of PLA/PCL Blends. J. Polym. Environ. 2021, 29, 2585–2597. 10.1007/s10924-021-02053-1. [DOI] [Google Scholar]