Abstract
Traumatic encephalopathy syndrome (TES) is used to describe the clinical manifestations of chronic traumatic encephalopathy (CTE). However, effective treatment and prevention strategies are lacking. Increasing evidence has shown that rehabilitation training could prevent cognitive decline, enhance brain plasticity, and effectively improve neurological function in neurodegenerative diseases. Therefore, the mechanisms involved in the effects of rehabilitation exercise therapy on the prognosis of CTE are worth exploring. The aim of this article is to review the pathogenesis of CTE and provide a potential clinical intervention strategy for CTE.
Keywords: Traumatic encephalopathy syndrome, Chronic traumatic encephalopathy, Rehabilitation exercise, Neuropathology, Nerve regeneration
Introduction
In recent years, the concept of traumatic encephalopathy syndrome (TES) has been proposed to describe the clinical manifestations of CTE. TES is a progressive process in which patients present with cognitive dysfunction and neurobehavioral disorders (Katz et al. 2021). However, the current understanding of TES remains still not completely clear, and there is a lack of effective preventive and therapeutic strategies.
Exercise has been shown to be beneficial in the prognosis of a variety of diseases (Dibben et al. 2021; Kanaley et al. 2022). The pathophysiology of CTE may be related to abnormal protein accumulation, neuroinflammation, microcirculation injury. Studies have demonstrated that rehabilitation exercise can reduce abnormal protein deposition by enhancing signal pathway transduction, promote neurogenesis, promote synaptogenesis, and increase synaptic plasticity (Xu et al. 2022a; Horowitz et al. 2020; Mu et al. 2022). Neuroinflammation is a common pathological manifestation of a variety of nervous system diseases, including neurodegenerative diseases and central nervous system tumors, and rehabilitation exercises also help to reduce neuroinflammation (Miguel et al. 2021a; Pang et al. 2022a). Several studies have demonstrated the effectiveness of rehabilitation exercise in the treatment of neurodegenerative diseases (Ruiz-González et al. 2021a; López-Ortiz et al. 2021a; Johansson et al. 2022), and rehabilitation exercise, as a less expensive and convenient treatment, may also have a good effect on improving the prognosis of patients with CTE, so it is necessary to investigate this area. This article discusses some of the identified and possible molecular mechanisms of CTE pathogenesis, and explores the potential of rehabilitation exercise to improve the clinical manifestations of TES.
Clinical phenotype and possible molecular mechanisms of traumatic encephalopathy syndrome
CTE has a unique pathological profile, and its diagnosis can currently only be confirmed by neuropathological examination at autopsy (Murray et al. 2022a). The specific molecular mechanism of CTE pathogenesis is not fully understood, and there are few studies on CTE from the perspective of neurovascular units (NVU), so some of the identified and possible pathogenesis of CTE are listed below.
Pathological and clinical features of CTE
Although the consensus defines CTE-related clinical syndromes as TES, the diagnosis of TES does not represent the diagnosis of CTE, and the diagnosis of CTE still requires neuropathological examination (Katz et al. 2021). In addition, the existence of TES remains difficult to determine, and the subjective judgment of the physician is sometimes required in the TES diagnostic process, which can introduce bias (Cullum and LoBue 2021). Patients with CTE are mostly veterans, contact sports athletes, and civilians who have suffered long-term head violence in various situations (Mez et al. 2021). At present, the etiology of CTE is not clear, and it may be highly related to repetitive head blows, and a small number of patients have no clear history of neurotrauma (McKee et al. 2023). McKee divided CTE into four stages according to the severity of p-tau lesions. With progression through the stages, the patients' clinical symptoms gradually worsen (McKee et al. 2013). The first stage mainly manifests as cognitive impairment and headache; psychiatric features (usually depression and suicide) persist in the second to fourth stages; the third stage usually shows behavior disorder; dementia, motor problems and severe mental disorders are common in the fourth stage (McKee et al. 2013). It should be noted that the relationship between pathological changes and clinical symptoms has not been confirmed (Iverson et al. 2019).
Molecular mechanisms of CTE
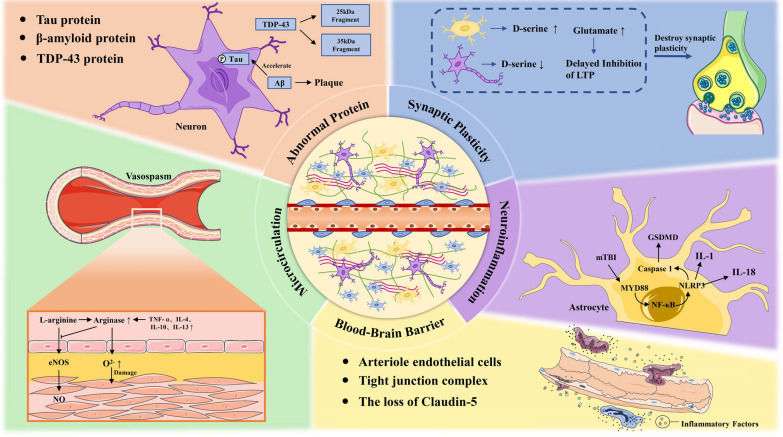
The gross pathology can show ventricular dilation and brain atrophy, usually located in the frontal and temporal lobes (McKee et al. 2015). The pathological changes of CTE mainly involve abnormal protein deposition, neuroinflammation, and microcirculation disorders, among which the typical microscopic pathological features are the aggregation of abnormal P-tau protein in the neurons and astroglia distributed around vessels in the deep cerebral cortical groove (McKee et al. 2016; Kaufman et al. 2021). The misfolded tau protein can induce the template misfolding and aggregation of healthy tau molecules in healthy cells, spread in a prion like manner, and then develop the lesions to other regions of the brain (Brunello et al. 2020; Falcon et al. 2019). P-tau lesions most often affect five regions of the brain: the dorsolateral frontal cortex, the superior temporal cortex, the entorhinal cortex, the amygdala, and the locus coeruleus (Alosco et al. 2020, 2019). In addition, diffuse amyloid β (A β) plaque and pathological inclusion bodies composed of transactive response DNA-binding protein can be seen in some CTE cases (Smith et al. 2019). Aβ deposition may result from axonal damage and loss, resulting in increased release of amyloid precursor protein (APP) (Chaves et al. 2021; Ikonomovic et al. 2017).
Microglia are the main cells mediating inflammation in various diseases of the central nervous system (Pang et al. 2022b; Khan et al. 2023). The activation of microglia in CTE may create conditions for maintaining chronic neuroinflammation and promote the accumulation of P-tau (Verboon et al. 2021). In addition, abnormal protein deposition can cause neuroinflammation, and RHIs can also directly cause neuroinflammation, such as the activation of Toll-like receptor myeloid difference factor 88 (MYD88) after RHIs, the promotion of nuclear factor kappa B (NF kappa B) transcription, and an increase in the expression of nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing-3 (NLRP3), interleukin 1 (IL-1), and interleukin 18 (IL-18) (Bauernfeind et al. 2009). Elevated inflammatory cytokines, such as tumor necrosis factor- α (TNF- α) and interleukin-4, can induce an increase in arginase activity and expression (Thornhill and Haskard 1990). Arginase has an inhibitory effect on nitric oxide synthase (eNOS), which can reduce the production of eNOS-derived nitric oxide (NO), resulting in vasodilation dysfunction that affects parenchymal perfusion (Shin et al. 2019; Mahdi et al. 2020). In addition, neuropathological examination of CTE patients reveals disruption of the blood–brain barrier, which may be associated with loss of tight junction complex 5 (claudin-5) (Farrell et al. 2019). This may lead to a vicious circle of neuroinflammation, microcirculatory disorder and neuronal death.
Inflammation and microcirculation damage can reduce synaptic plasticity. The mechanism may be related to up-regulation of D-serine level by astrocytes, and an increase in excitatory neurotransmitter glutamate levels (Wolosker et al. 2016; Tapanes et al. 2022). Therefore, how to inhibit neuroinflammation, microcirculation disorder and neuron death, promote neurogenesis and increase synaptic plasticity are the keys to improving the prognosis of CTE (Fig. 1).
Fig. 1.
The pathological mechanisms of CTE. The accumulation of abnormal protein deposition, reduction in synaptic plasticity, and abnormal activation of the inflammatory reaction, reduced microcirculation function, and impaired blood–brain barrier are the main pathological features of CTE. It worsens the brain microenvironment and leads to extensive neuronal degeneration
Potential mechanisms by which rehabilitation exercises improve CTE
Rehabilitation exercise can improve cognitive function, clinical symptoms such as depression and anxiety (Pearce et al. 2022a), and can also protect the nervous system by inducing the production of brain-derived neurotrophic factor (BDNF), insulin-like growth factor I and vascular endothelial growth factor (VEGF), which has a beneficial effect on brain plasticity (Ruiz-González et al. 2021a; Morland et al. 2017a). For example, light and moderate aerobic treadmill exercise can reduce fatigue, improve neonatal hippocampal neuron survival, and improve neurobehavioral outcomes after neurological disease (Karelina et al. 2021). Regular physical exercise has been shown to be an effective way to slow the progression of neurodegenerative diseases such as Alzheimer’s disease (Pearce et al. 2022a).
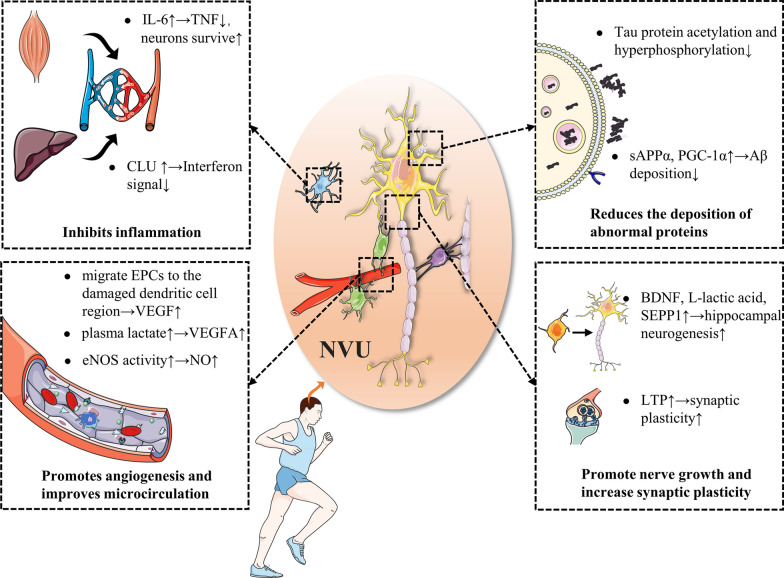
Repeated concussions can be a trigger for CTE (McKee et al. 2023), and rehabilitative physical activity for post-concussion patients has been shown to improve symptoms and speed recovery (Howell et al. 2021; Leddy et al. 2021). CTE progresses slowly, and as a neurodegenerative disease, rehabilitation exercises may also be effective for it (Murray et al. 2022b). In this section, we aim to introduce the potential mechanism of rehabilitative physical exercise in improving CTE. It may be related to preventing abnormal protein deposition, alleviating neuroinflammation, regulating microcirculation, and promoting neurogenesis and synaptic plasticity (Table 1) (Fig. 2).
Table 1.
The main beneficial mechanisms of rehabilitation exercise for CTE
| Sorting | Subclass | Mechanism | References |
|---|---|---|---|
| Reduction of abnormal protein deposition | Tau protein | Rehabilitation exercises can reduce tau protein acetylation and promote Wnt/GSK3 β and PI3K/Akt signaling pathways, reducing P-Tau production and increasing its clearance | Mankhong et al. (2020); Chen et al. (2020); Xu et al. (2022b) |
| β-amyloid protein | Rehabilitation exercise increases ADAM10 expression and promote PGC-1 α / FNDC5 pathway thereby reduces Aβ production | Yu et al. (2021a); McMeekin et al. (2020) | |
| Antiinflammation and oxidation stres | IL-6, CLU | Rehabilitation exercise promotes the release of IL-6 from skeletal muscles and the release of CLUs from the liver to reduce neuroinflammation | Chow et al. (2022); Bateman et al. (2016) |
| Promote angiogenesis and improve micro-circulation | VEGF | Rehabilitation exercise can increase plasma lactic acid to promote ERK1/2 and Akt signal transduction and promote EPC to secretion VEGF | Morland et al. (2017b); Ross et al. (2014) |
| Promotion of neurogenesis | BDNF | BDNF promotes neuronal development and differentiation through the BDNF/TrkB signaling pathway | Colucci-D’Amato et al. (2020) |
| L-lactic acid | L-lactate activates HCA1 to promote the AKT/PK pathway to promote cell survival and value-added | Lambertus et al. (2021) | |
| MCT2 | Rehabilitation exercise increases MCT2 expression and improves neuronal energy metabolism | Lev-Vachnish et al. (2019); Yu et al. (2021b) | |
| The promotion of synaptogenesis and the increase of synaptic plasticity | LTP | Regular rehabilitation exercises can increase LTP | Vivar and Praag (2017) |
| Glutamate | Rehabilitation exercise increases the excitatory neurotransmitter glutamate | Andersen et al. (2021); Maddock et al. (2016) |
Fig. 2.
The mechanisms of exercise in CTE. CTE triggers neurodegenerative changes. However, rehabilitation exercise reduces the pathological processes of CTE through a variety of mechanisms (such as reducing abnormal protein accumulation, promoting neurogenesis, synaptic formation, increasing synaptic plasticity and promoting angiogenesis, improving microcirculation, and resisting micro-inflammation)
Rehabilitation exercises reduce abnormal protein deposition
The deposition of p-tau is an important pathological feature of CTE. Deposition of other proteins such as Aβ can accelerate p-tau deposition, aggravating pathological outcomes. However, rehabilitation exercise can reduce the deposition of abnormal proteins and thus could benefit those with CTE.
Rehabilitation exercises reduce P-tau generation
Rehabilitation exercises can reduce P-tau synthesis and increase P-tau clearance. Tau protein acetylation can promote Tau protein aggregation and hyperphosphorylation, and make P-tau more difficult to degrade, while regular aerobic rehabilitation exercise can reduce Tau protein acetylation and improve the pathological process of CTE (Mankhong et al. 2020). In addition, rehabilitation exercise can inhibit tau protein hyperphosphorylation. Glycogen synthase kinase-3 (GSK-3) is involved in the regulation of many key cell biological pathways, and one of its subforms, GSK-3β, is involved in phosphorylation of Tau protein after CTE (Hernandez et al. 2013; Moszczynski et al. 2018). Rehabilitation exercises reduce GSK-3β activity and reduce Tau phosphorylation by inhibiting the expression of Dickkopf-1, an inhibitor of the Wnt/GSK3β pathway, by upregulating the Wnt/GSK3β pathway (Chen et al. 2020). Finally, rehabilitation can activate the PI3K/Akt pathway to increase the expression of its downstream protein HSP70, a key protein that makes up the ubiquitin–proteasome system, so rehabilitation can enhance this system and promote P-tau clearance (Xu et al. 2022b).
Rehabilitation exercise reduces Aβ deposition
Amyloid precursor protein (APP) is an important protein for the growth and development of neurons and the implementation of various activities (Liu et al. xxxx). APP processing pathways are generally divided into the amyloid pathway (mediated by secretary enzyme β and γ) and non-amyloid (mainly mediated by secretary enzyme α) (Cho et al. 2022; Wilkins and Swerdlow 2017). Research on the APP processing pathway shows that exercise can increase the secretion of a disintegrin and metalloprotease 10 (ADAM10), the main member of the α secretary enzyme, which increases the expression of soluble APP by increasing the non-amyloid pathway α (sAPP α) (Yu et al. 2021a). In addition, the increase in sAPPα can inhibit BACE1, thereby reducing the amyloid pathway and Aβ production (Nigam et al. 2017). Rehabilitation exercise can also reduce Aβ-induced neurological dysfunction by increasing muscle expression of proliferator-activated receptor-γ coactivator-1α (PGC-1α) and through the PGC-1α/FNDC5/BDNF pathway (Azimi et al. 2018; Neto et al. 2023). Generally speaking, rehabilitation exercises can reduce abnormal proteins (P-tau and Aβ), therefore rehabilitation exercises may be used as a potential clinical intervention strategy for the treatment of CTE.
Rehabilitation exercise inhibits the vicious cycle of neuroinflammation—microcirculation disorders—neuronal death
Neuroinflammation is the initiating factor of secondary brain injury, which can lead to microcirculation disorders. Brain tissue and neuronal ischemia and hypoxia aggravate inflammatory reaction, forming a vicious cycle of neuroinflammation-microcirculation disorders-neuronal death. Many studies have shown that rehabilitation exercise could effectively inhibit this vicious circle, thereby improving the prognosis of patients.
Rehabilitation exercise has an anti-inflammatory effect
The mechanism of rehabilitation exercise to reduce neuroinflammation may be related to reducing microglia activation and promoting the release of anti-inflammatory factors from skeletal muscle and the liver. Glia mediates neuroimmunity, and intervention in glial cells through different pathways may alter disease progression (Goenka et al. 2023; Xuan et al. 2022). For example, microglia-targeted immunotherapy has good promise in tumor treatment (Pang et al. 2022a). Similarly, rehabilitation exercise helps to regulate microglia metabolic status, reduce microglia activation, and thus reduce neuroinflammation. IL-6 released from skeletal muscle after exercise can enter the brain through the blood brain barrier. IL-6 demonstrates a dual effect of promoting inflammation and exerting anti-inflammatory actions at various levels (Forcina et al. 2022), on the one hand, a transient increase in IL-6 after exercise can combine with other anti-inflammatory factors to inhibit inflammation such as inhibiting the pro-inflammatory effect of TNF (Chow et al. 2022; Bateman et al. 2016), on the other hand, long-term high systemic levels of IL-6 have pro-inflammatory effects and are associated with the occurrence of a variety of diseases, however, exercise individuals have lower baseline levels of IL-6 (Pedersen and Febbraio 2012), and functional neurogenesis refilling of post-CTE microglia may also be mediated by interleukin 6 (IL-6) trans-signaling pathways (Willis et al. 2020). IL-6 may be the key to enhancing the survival of neurons after CTE. In addition, some studies have shown that rehabilitation exercise can promote clusterin (CLU) production in the liver, targets brain endothelial cells, inhibits interferon signals, and plays an anti-inflammatory role (Miguel et al. 2021b).
Promotes angiogenesis and improves microcirculation
Rehabilitation exercise with a certain intensity can improve cerebral blood flow perfusion (Steventon et al. 2020). As a heparin binding growth factor, VEGF can target vascular endothelial cells, promote their proliferation and induce angiogenesis in vivo (Ahmad and Nawaz 2022). The increase in VEGF caused by exercise may be achieved through the following two ways: (1) Exercise increases the plasma lactate concentration, activates ERK1/2 and Akt signals by acting on the lactate receptor, thereby increasing the expression of vascular endothelial growth factor A (VEGFA) (Morland et al. 2017b); (2) rehabilitation exercise can promote the migration of endothelial progenitor cells (EPCs) to the area of injured dendothelial cells, and the secretion of VEGF to promote vascular growth and repair (Ross et al. 2014). In addition, rehabilitation exercise can promote EPCs to secrete EPC-derived exosomes and promote microvascular regeneration, thereby improving microcirculation (Wang et al. 2020; Ma et al. 2018). Rehabilitation exercises can also help improve vascular function. Exercise can increase vascular laminar shear stress, which can induce increased calcium-mediated eNOS enzyme activity in vascular endothelial cells, thereby increasing NO production (Tryfonos et al. 2022). Therefore, rehabilitation exercise is conducive to avoiding vascular endothelial dysfunction and reducing vascular oxidative damage. To sum up, rehabilitation exercise may induce angiogenesis, and VEGF may improve microcirculation disorders and nerve function, which may be a potential therapeutic target for improving microcirculation disorders in CTE patients.
Rehabilitation exercises can promote nerve growth and increase synaptic plasticity
The brain is structurally and functionally highly plastic, and central nervous system diseases can have harmful effects through this property, such as neurodegenerative diseases, where the hippocampus is remodeled and cognitive and functional impairment can be caused (Weerasinghe-Mudiyanselage et al. 2022). Physical exercise has been shown to promote beneficial brain remodeling and improve neural function by regulating epigenetic inheritance and promoting the expression of neurotrophic factors (Liang et al. 2021). The positive effects of rehabilitation exercise on the brain plasticity of CTE patients are mainly reflected in promoting hippocampal neurogenesis and increasing synaptic plasticity.
Rehabilitation exercises contribute to hippocampal neurogenesis
During the pathological development of CTE, degenerative necrosis of neurons often occurs (Bauernfeind et al. 2009). However, the adult hippocampus can produce a type of neuron (dentate gyrus granule cells) that can eventually differentiate into new neurons (Zhou et al. 2022). Rehabilitation exercises can improve the hippocampal environment and promote adult hippocampal neurogenesis through various mechanisms, thereby improving the pathological outcome of CTE and the prognosis of patients. Rehabilitation exercise raises the level of BDNF, and its mechanism may be related to the Wnt/ β-catenin signal pathway (Cheng et al. 2020). Rehabilitation exercise can change the morphology of astrocytes and promote the expression of BDNF (Li et al. 2021). BDNF is an important neurodynamic factor in the brain, which promotes the development and differentiation of neurons and plays an active role in the repair of nerve injury (Walsh and Tschakovsky 2018). Its role in adult hippocampal neurogenesis is mediated by BDNF/TrkB signaling pathway (Colucci-D'Amato et al. 2020).
Rehabilitation exercise may promote neurogenesis by mediating L-lactic acid, and its mechanism may be associated with the hydroxycarboxylic acid receptor 1 (HCA1)-mediated AKt/PK pathway (Lambertus et al. 2021). Also, L-lactic acid can activate monocarboxylate transporter 2 (MCT2) on newborn neurons (Lev-Vachnish et al. 2019). However, overexpression of MCT2 can increase mitochondrial biogenesis, thereby improving neuronal energy metabolism (Yu et al. 2021b). Rehabilitation training can also promote hippocampal neurogenesis in adults by altering blood composition. Selenium is an indispensable trace element in the human body. The secreting selenoprotein P (SEPP1) metabolized in the liver is the organic form of selenium in the human body, which has antioxidant and anti-inflammatory effects (Hariharan and Dharmaraj 2020; Burk and Hill 2015). Studies have shown that rehabilitation exercise can significantly increase the concentration of SEPP1 in plasma and promote the binding of SEPP1 to the receptor. Researchers have reported that low-density lipoprotein receptor related protein 8 promoted hippocampal neurogenesis (Leiter et al. 2022). In addition, rehabilitation exercise can increase the concentration of glycosylphosphatidylinositol (GPI)-specific phospholipase D1 in plasma, and alter the signal cascade downstream of GPI anchor substrate lysis, thereby affecting neurogenesis, improving age-related regeneration and reducing cognitive impairment (Leiter et al. 2022; Fujihara and Ikawa 2016). Finally, rehabilitation training can also induce platelet activation, promote the secretion of platelet factor 4, and contribute to the neurogenesis of the dentate gyrus of the hippocampus (Leiter et al. 2019).
Rehabilitation exercises help to increase synaptic plasticity
Synaptic plasticity is an important component of learning and memory (Magee and Grienberger 2020). The progress of CTE is usually accompanied by the loss of synaptic connections, the rupture of neuronal axes and the loss of dendritic spines, which seriously damage the plasticity of synapses. In recent years, more and more data have shown that hippocampal synaptic plasticity can be achieved through regular rehabilitation exercise. Regular rehabilitation exercises can increase long-term enhancement (LTP), a form of synaptic plasticity and an important component of memory formation and maintenance (Vivar and Praag 2017). The mechanism of rehabilitation exercise-enhanced LTP is complex and may be related to a variety of exercise-induced products (Vints et al. 2022). Rehabilitation exercise can also remotely regulate synaptic plasticity through exosomes, studies have shown that rehabilitation exercise can promote exosome release and promote synaptic remodeling through microglia (Li et al. 2022; Jiang et al. 2023). Finally, rehabilitation exercises also affect the expression of neurotransmitters, thereby altering synaptic plasticity, especially levels of glutamate, which is thought to be the main excitatory neurotransmitter involved in synaptic formation and synaptic communication (Andersen et al. 2021; Maddock et al. 2016). Therefore, rehabilitation exercise may promote hippocampal neurogenesis and synaptic plasticity in a variety of ways to improve the prognosis of CTE.
At present, there are still few clinical studies and basic studies on rehabilitation exercise to improve CTE, and many patients with CTE are athletes and soldiers, compared with other patients such as civilians who have suffered from long-term head violence, they have performed long-term regular exercise before CTE, which will have an impact on the prognosis of CTE is unknown, so the specific molecular mechanism of rehabilitation exercise to improve CTE still needs to be explored and verified.
Clinical therapeutic intervention strategy of rehabilitation exercise for CTE
As the pathogenic factors and pathological mechanism of CTE have not been fully defined, and the diagnosis of its corresponding clinical syndrome TES is also challenging, the treatment of CTE is still in an emerging field. P-tau accumulation is a key pathological mechanism of CTE, so blocking P-tau deposition is also regarded as a promising target for the treatment of CTE (Kim et al. 2023). There have been studies to counter the accumulation of P-Tau by introducing adeno-associated virus vectors encoding anti-P-Tau antibodies to the central nervous system (Sacramento et al. 2020), and there have been studies using nanocapsules to deliver immunoglobulins to the central nervous system, which also help reduce the accumulation of P-tau (Zhang et al. 2023). Although these studies have shown good effects, they are still confined to animal experiments, and the effectiveness of treatment for CTE patients may still need to be verified for a long time. At the present stage, rehabilitation exercise may still be one of the most potential and easiest to implement CTE treatment. Rehabilitation exercise also has a good therapeutic effect on the complications of CTE, including related metabolic disorders such as hypopituitarism, neurobehavioral disorders, cognitive dysfunction, dementia and other adverse consequences (Fig. 3).
Fig. 3.
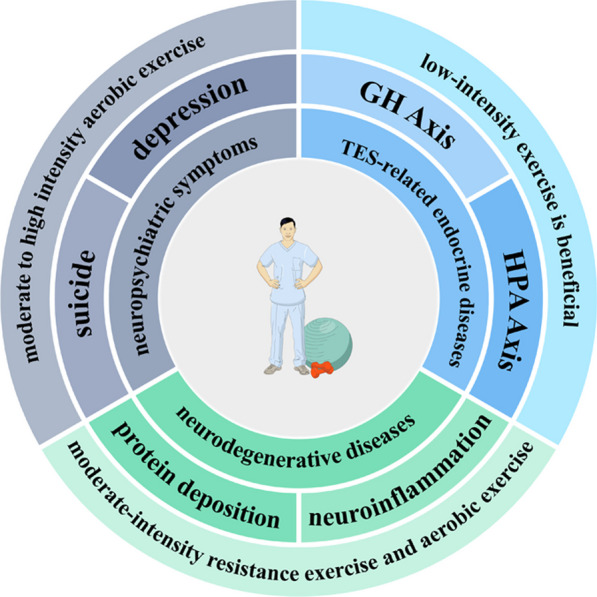
Rehabilitation exercise has obvious clinical benefits for CTE patients, which can effectively improve the symptoms of CTE-related endocrine diseases, and can significantly improve early neuropsychiatric symptoms and late diseases that are difficult to effectively treat with drugs such as AD
Rehabilitation exercises may improve CTE-related endocrine diseases
Pituitary dysfunction is a common concomitant endocrine complication after CTE, which can lead to the loss of pituitary hormones in patients, including growth hormone, adrenocorticotropic hormone, and thyroid-stimulating hormone deficiency (Costanza et al. 2011). For these patients, different intensities and different types of exercise have different effects on patients lacking different kinds of hormones. Studies have shown that high intensity exercise is effective in increasing growth-hormone axis (GH Axis) activity, while the effect of resistance exercise on hypothalamic–pituitary–adrenal axis (HPA Axis) has not been confirmed (Haunhorst et al. 2022), but moderate and high-intensity aerobic exercise seems to increase HPA Axis activity (Hill et al. 2008; Takahashi et al. 2022). The effect of exercise on hypothalamic-pituitary-thyroid axis is uncertain (Babić Leko et al. 2021). The above evidence suggests that rehabilitation exercise may improve endocrine diseases related to CTE.
Rehabilitation exercises may improve neuropsychiatric symptoms caused by CTE
The early manifestations of CTE in patients are neuropsychiatric symptoms, mainly depression and suicide. The effect of rehabilitation exercise on neuropsychiatric symptoms is positive (Kim 2022), and close in magnitude to that of traditional drug treatment (Smith and Merwin 2021). The mechanism of rehabilitation exercise to improve neuropsychiatric symptoms may be related to the change in neural plasticity (Smith and Merwin 2021). Rehabilitation exercise has a good effect on patients of different ages (Philippot et al. 2022; Hidalgo and Sotos 2021). Even low-intensity exercise has obvious benefits for improving neuropsychiatric symptoms (Pearce et al. 2022b). Therefore, it is recommended that TES patients start rehabilitation exercise as soon as possible.
Rehabilitation exercise may improve neurodegenerative diseases caused by CTE
Dementia is the terminal manifestation of CTE, which is similar to the clinical presentations of many neurodegenerative diseases. CTE is also considered to be associated with neurodegenerative diseases, especially Alzheimer’s disease (AD) (Gardner and Yaffe 2015; Yu et al. 2020). CTE and AD have similar main neuropathological characteristics: P-tau protein has different structures but similar immunophenotypes. Aβ deposition can also be seen in some patients with TES (Falcon et al. 2019; Johnson et al. 2020). The clinical characteristics of CTE and AD are similar, which often leads to misdiagnosis. The effect of rehabilitation exercise on Alzheimer’s disease has been well established (Valenzuela et al. 2020). It can decrease abnormal protein deposition (Tan et al. 2021), alleviate neuroinflammation and oxidative stress (Maddock et al. 2016; Miguel et al. 2021b), improve microcirculation (López-Ortiz et al. 2021b), reduce neuronal apoptosis (Zhao et al. 2021), promote neural repair (Ruiz-González et al. 2021b), and improve the prognosis of Alzheimer’s disease caused by CTE. The recommended modality for AD is at least 45 min of moderate-intensity resistance exercise and aerobic exercise (Northey et al. 2018).
It is important to note that different types of rehabilitation exercise, such as aerobic exercise or resistance exercise, as well as the time, intensity and frequency of rehabilitation exercise, often have different effects on the mitigation of neurological diseases. Therefore, follow-up studies are needed to determine the most effective rehabilitation exercise treatment strategy for CTE. In addition, it is necessary to develop personalized strategies for patients with different behavioral abilities in the clinical implementation process to facilitate the smooth development of treatment. For some patients with obvious cognitive impairment, cognitive rehabilitation may be more conducive to symptom relief and progress. At present, there are still few studies on drug development targeting the pathological mechanism of CTE, most of which focus on immunotherapy to slow down P-tau deposition. The therapeutic targets of other pathogenic pathways remain to be discovered, and the therapeutic effects of rehabilitation therapy for CTE, including rehabilitation exercise, still need to be further verified although they have good prospects.
Conclusion
The clinical syndrome of CTE, TES, begins early in injury and is difficult to relieve with existing means. This article reviews the potential mechanism of physical rehabilitation exercise in the treatment of CTE from various aspects, aiming to explore the prospect of rehabilitation exercise, an effective and easy-to-implement treatment, for the application of CTE.
Further research into the neuropathology of CTE patients is needed, as it can provide valuable information for the development of disease biomarkers and the evaluation of potential treatments. In addition, the impact of rehabilitation exercises on brain health in CTE patients that needs to be validated is important for improving our understanding of the mechanisms of neurodegenerative diseases, and the search for the most appropriate exercise strategy for CTE patients should continue, which may be a promising area of research in the future.
Acknowledgements
None.
Abbreviations
- CTE
Chronic traumatic encephalopathy
- P-Tau
Hyperphosphorylated tau protein
- TES
Traumatic encephalopathy syndrome
- Aβ
Amyloid-β
- APP
Amyloid precursor protein
- BACE1
B-site APP cleaving enzyme 1
- MYD88
Myeloid difference factor 88
- NF kappa B
Nuclear factor kappa B
- NLRP3
Nucleotide-binding oligomerization domain-like receptor family pyrin domain-containing-3
- IL-1
Interleukin 1
- IL-18
Interleukin 18
- TNF-α
Tumor necrosis factor-alpha
- NO
Nitric oxide
- eNOS
Nitric oxide synthase
- ET-1
Endothelin-1
- MMPs
Matrix metalloproteinases
- NFAT
Nuclear cell membrane proteases
- Claudin-5
Complex 5
- BDNF
Brain-derived neurotrophic factor
- VEGF
Vascular endothelial growth factor
- GSK-3
Glycogen synthase kinase-3
- ADAM10
A disintegrin and metalloprotease 10
- sAPPα
Souble APP α
- PGC-1α
Proliferator to activate receptor gamma coactivator-1α
- IL-6
Interleukin 6
- CLU
Clusterin
- SOD
Superoxide dismutase
- VEGFA
Vascular endothelial growth factor A
- EPCs
Endothelial progenitor cells
- MCT2
Monocarboxylate transporter 2
- SEPP1
Selenoprotein P
- GPI
Glycosylphosphatidylinositol
- PDE-4
Phosphodiesterase-4
- cAMP
Cyclic adenosine monophosphate
- LTP
Long-term potentiation
- GH Axis
Increasing growth-hormone axis
- HPA Axis
Hypothalamic–pituitary–adrenal axis
- AD
Alzheimer’s disease
Author contributions
YH and ZW contributed equally to this manuscript. YH and ZW contributed to the literature search and manuscript writing. SL and XC contributes to the study design and manuscript writing. All the authors read and approved the final manuscript. All the authors read and approved the final manuscript.
Funding
This work was supported in part by Grants from the Major Scientific Research Program for Young and Middle-aged Health Professionals of Fujian Province, China (Grant No. 2022ZQNZD007); Fujian Province Scientific Foundation (Grant No. 2023J01725); National Natural Science Foundation of China (Grant No. 82371390).
Availability of data and materials
All relevant data is contained within the article. Further inquiries can be directed to the corresponding author.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors have read and consented to the publication of the article.
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yin-qiong Huang and Zhe Wu contributed equally to this work.
Contributor Information
Shu Lin, Email: shulin1956@126.com.
Xiang-rong Chen, Email: xiangrong_chen281@126.com.
References
- Ahmad A, Nawaz MI. Molecular mechanism of VEGF and its role in pathological angiogenesis. J Cell Biochem. 2022;123(12):1938–1965. doi: 10.1002/jcb.30344. [DOI] [PubMed] [Google Scholar]
- Alosco ML, Stein TD, Tripodis Y, Chua AS, Kowall NW, Huber BR, et al. Association of white matter rarefaction, arteriolosclerosis, and tau with dementia in chronic traumatic encephalopathy. JAMA Neurol. 2019;76(11):1298–1308. doi: 10.1001/jamaneurol.2019.2244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alosco ML, Cherry JD, Huber BR, Tripodis Y, Baucom Z, Kowall NW, et al. Characterizing tau deposition in chronic traumatic encephalopathy (CTE): utility of the McKee CTE staging scheme. Acta Neuropathol. 2020;140(4):495–512. doi: 10.1007/s00401-020-02197-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen JV, Markussen KH, Jakobsen E, Schousboe A, Waagepetersen HS, Rosenberg PA, et al. Glutamate metabolism and recycling at the excitatory synapse in health and neurodegeneration. Neuropharmacology. 2021;196:108719. doi: 10.1016/j.neuropharm.2021.108719. [DOI] [PubMed] [Google Scholar]
- Azimi M, Gharakhanlou R, Naghdi N, Khodadadi D, Heysieattalab S. Moderate treadmill exercise ameliorates amyloid-β-induced learning and memory impairment, possibly via increasing AMPK activity and up-regulation of the PGC-1α/FNDC5/BDNF pathway. Peptides. 2018;102:78–88. doi: 10.1016/j.peptides.2017.12.027. [DOI] [PubMed] [Google Scholar]
- Babić Leko M, Gunjača I, Pleić N, Zemunik T. Environmental factors affecting thyroid-stimulating hormone and thyroid hormone levels. Int J Mol Sci. 2021;22(12):6521. doi: 10.3390/ijms22126521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bateman RM, Sharpe MD, Jagger JE, Ellis CG, Solé-Violán J, López-Rodríguez M, et al. 36th International Symposium on Intensive Care and Emergency Medicine: Brussels, Belgium. 15–18 March 2016. Crit Care. 2016;20 (Suppl 2):94. [DOI] [PMC free article] [PubMed]
- Bauernfeind FG, Horvath G, Stutz A, Alnemri ES, MacDonald K, Speert D, et al. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. J Immunol. 2009;183(2):787–791. doi: 10.4049/jimmunol.0901363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunello CA, Merezhko M, Uronen RL, Huttunen HJ. Mechanisms of secretion and spreading of pathological tau protein. Cell Mol Life Sci. 2020;77(9):1721–1744. doi: 10.1007/s00018-019-03349-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burk RF, Hill KE. Regulation of selenium metabolism and transport. Annu Rev Nutr. 2015;35:109–134. doi: 10.1146/annurev-nutr-071714-034250. [DOI] [PubMed] [Google Scholar]
- Chaves RS, Tran M, Holder AR, Balcer AM, Dickey AM, Roberts EA, et al. Amyloidogenic processing of amyloid precursor protein drives stretch-induced disruption of axonal transport in hiPSC-derived neurons. J Neurosci. 2021;41(49):10034–10053. doi: 10.1523/JNEUROSCI.2553-20.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D, Zhang Y, Zhang M, Chang J, Zeng Z, Kou X, et al. Exercise attenuates brain aging by rescuing down-regulated Wnt/β-catenin signaling in aged rats. Front Aging Neurosci. 2020;12:105. doi: 10.3389/fnagi.2020.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng J, Shen W, Jin L, Pan J, Zhou Y, Pan G, et al. Treadmill exercise promotes neurogenesis and myelin repair via upregulating Wnt/β-catenin signaling pathways in the juvenile brain following focal cerebral ischemia/reperfusion. Int J Mol Med. 2020;45(5):1447–1463. doi: 10.3892/ijmm.2020.4515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho Y, Bae H-G, Okun E, Arumugam TV, Jo D-G. Physiology and pharmacology of amyloid precursor protein. Pharmacol Ther. 2022;235:108122. doi: 10.1016/j.pharmthera.2022.108122. [DOI] [PubMed] [Google Scholar]
- Chow LS, Gerszten RE, Taylor JM, Pedersen BK, van Praag H, Trappe S, et al. Exerkines in health, resilience and disease. Nat Rev Endocrinol. 2022;18(5):273–289. doi: 10.1038/s41574-022-00641-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colucci-D'Amato L, Speranza L, Volpicelli F. Neurotrophic factor BDNF, physiological functions and therapeutic potential in depression, neurodegeneration and brain cancer. Int J Mol Sci. 2020;21(20):7777. doi: 10.3390/ijms21207777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costanza A, Weber K, Gandy S, Bouras C, Hof PR, Giannakopoulos P, et al. Review: contact sport-related chronic traumatic encephalopathy in the elderly: clinical expression and structural substrates. Neuropathol Appl Neurobiol. 2011;37(6):570–584. doi: 10.1111/j.1365-2990.2011.01186.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullum CM, LoBue C. Defining traumatic encephalopathy syndrome—advances and challenges. Nat Rev Neurol. 2021;17(6):331–332. doi: 10.1038/s41582-021-00500-0. [DOI] [PubMed] [Google Scholar]
- De Miguel Z, Khoury N, Betley MJ, Lehallier B, Willoughby D, Olsson N, et al. Exercise plasma boosts memory and dampens brain inflammation via clusterin. Nature. 2021;600(7889):494–499. doi: 10.1038/s41586-021-04183-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dibben G, Faulkner J, Oldridge N, Rees K, Thompson DR, Zwisler A-D, et al. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst Rev. 2021;11(11):CD001800. doi: 10.1002/14651858.CD001800.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falcon B, Zivanov J, Zhang W, Murzin AG, Garringer HJ, Vidal R, et al. Novel tau filament fold in chronic traumatic encephalopathy encloses hydrophobic molecules. Nature. 2019;568(7752):420–423. doi: 10.1038/s41586-019-1026-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farrell M, Aherne S, O'Riordan S, O'Keeffe E, Greene C, Campbell M. Blood-brain barrier dysfunction in a boxer with chronic traumatic encephalopathy and schizophrenia. Clin Neuropathol. 2019;38(2):51–58. doi: 10.5414/NP301130. [DOI] [PubMed] [Google Scholar]
- Forcina L, Franceschi C, Musarò A. The hormetic and hermetic role of IL-6. Ageing Res Rev. 2022;80:101697. doi: 10.1016/j.arr.2022.101697. [DOI] [PubMed] [Google Scholar]
- Fujihara Y, Ikawa M. GPI-AP release in cellular, developmental, and reproductive biology. J Lipid Res. 2016;57(4):538–545. doi: 10.1194/jlr.R063032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner RC, Yaffe K. Epidemiology of mild traumatic brain injury and neurodegenerative disease. Mol Cell Neurosci. 2015;66(Pt B):75–80. doi: 10.1016/j.mcn.2015.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goenka A, Khan F, Verma B, Sinha P, Dmello CC, Jogalekar MP, et al. Tumor microenvironment signaling and therapeutics in cancer progressio n. Cancer Commun (lond). 2023;43(5):525–561. doi: 10.1002/cac2.12416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hariharan S, Dharmaraj S. Selenium and selenoproteins: it’s role in regulation of inflammation. Inflammopharmacology. 2020;28(3):667–695. doi: 10.1007/s10787-020-00690-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haunhorst S, Bloch W, Ringleb M, Fennen L, Wagner H, Gabriel HHW, et al. Acute effects of heavy resistance exercise on biomarkers of neuroendocrine-immune regulation in healthy adults: a systematic review. Exerc Immunol Rev. 2022;28:36–52. [PubMed] [Google Scholar]
- Hernandez F, Lucas JJ, Avila J. GSK3 and tau: two convergence points in Alzheimer’s disease. J Alzheimers Dis. 2013;33(Suppl 1):S141–S144. doi: 10.3233/JAD-2012-129025. [DOI] [PubMed] [Google Scholar]
- Hidalgo JL-T, Sotos JR. Effectiveness of physical exercise in older adults with mild to moderate depression. Ann Fam Med. 2021;19(4):302–309. doi: 10.1370/afm.2670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill EE, Zack E, Battaglini C, Viru M, Viru A, Hackney AC. Exercise and circulating cortisol levels: the intensity threshold effect. J Endocrinol Invest. 2008;31(7):587–591. doi: 10.1007/BF03345606. [DOI] [PubMed] [Google Scholar]
- Horowitz AM, Fan X, Bieri G, Smith LK, Sanchez-Diaz CI, Schroer AB, et al. Blood factors transfer beneficial effects of exercise on neurogenesis and cognition to the aged brain. Science (new York, NY) 2020;369(6500):167–173. doi: 10.1126/science.aaw2622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howell DR, Hunt DL, Aaron SE, Meehan WP, 3rd, Tan CO. Influence of aerobic exercise volume on postconcussion symptoms. Am J Sports Med. 2021;49(7):1912–1920. doi: 10.1177/03635465211005761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikonomovic MD, Mi Z, Abrahamson EE. Disordered APP metabolism and neurovasculature in trauma and aging: combined risks for chronic neurodegenerative disorders. Ageing Res Rev. 2017;34:51–63. doi: 10.1016/j.arr.2016.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iverson GL, Gardner AJ, Shultz SR, Solomon GS, McCrory P, Zafonte R, et al. Chronic traumatic encephalopathy neuropathology might not be inexorably progressive or unique to repetitive neurotrauma. Brain. 2019;142(12):3672–3693. doi: 10.1093/brain/awz286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang X-H, Li H-F, Chen M-L, Zhang Y-X, Chen H-B, Chen R-H, et al. Treadmill exercise exerts a synergistic effect with bone marrow mesenchymal stem cell-derived exosomes on neuronal apoptosis and synaptic-axonal remodeling. Neural Regen Res. 2023;18(6):1293–1299. doi: 10.4103/1673-5374.357900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansson ME, Cameron IGM, Van der Kolk NM, de Vries NM, Klimars E, Toni I, et al. Aerobic exercise alters brain function and structure in Parkinson’s disease: a randomized controlled trial. Ann Neurol. 2022;91(2):203–216. doi: 10.1002/ana.26291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson VE, Stewart W, Trojanowski JQ, Lee VMY, Robinson JL, Irwin DJ, et al. Tau immunophenotypes in chronic traumatic encephalopathy recapitulate those of ageing and Alzheimer’s disease. Brain. 2020;143(5):1572–1587. doi: 10.1093/brain/awaa071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanaley JA, Colberg SR, Corcoran MH, Malin SK, Rodriguez NR, Crespo CJ, et al. Exercise/physical activity in individuals with type 2 diabetes: a consensus statement from the American college of sports medicine. Med Sci Sports Exerc. 2022;54(2):353–368. doi: 10.1249/MSS.0000000000002800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karelina K, Schneiderman K, Shah S, Fitzgerald J, Cruz RV, Oliverio R, et al. Moderate intensity treadmill exercise increases survival of newborn hippocampal neurons and improves neurobehavioral outcomes after traumatic brain injury. J Neurotrauma. 2021;38(13):1858–1869. doi: 10.1089/neu.2020.7389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz DI, Bernick C, Dodick DW, Mez J, Mariani ML, Adler CH, et al. National institute of neurological disorders and stroke consensus diagnostic criteria for traumatic encephalopathy syndrome. Neurology. 2021;96(18):848–863. doi: 10.1212/WNL.0000000000011850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufman SK, Svirsky S, Cherry JD, McKee AC, Diamond MI. Tau seeding in chronic traumatic encephalopathy parallels disease severity. Acta Neuropathol. 2021;142(6):951–960. doi: 10.1007/s00401-021-02373-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan F, Pang L, Dunterman M, Lesniak MS, Heimberger AB, Chen P. Macrophages and microglia in glioblastoma: heterogeneity, plasticity, and therapy. J Clin Invest. 2023;133(1):e163446. doi: 10.1172/JCI163446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J-H. Regular physical exercise and its association with depression: a population-based study short title: exercise and depression. Psychiatry Res. 2022;309:114406. doi: 10.1016/j.psychres.2022.114406. [DOI] [PubMed] [Google Scholar]
- Kim J, de Haro M, Al-Ramahi I, Garaicoechea LL, Jeong H-H, Sonn JY, et al. Evolutionarily conserved regulators of tau identify targets for new therapies. Neuron. 2023;111(6):824–838. doi: 10.1016/j.neuron.2022.12.012. [DOI] [PubMed] [Google Scholar]
- Lambertus M, Øverberg LT, Andersson KA, Hjelden MS, Hadzic A, Haugen ØP, et al. L-lactate induces neurogenesis in the mouse ventricular-subventricular zone via the lactate receptor HCA. Acta Physiol (oxf) 2021;231(3):e13587. doi: 10.1111/apha.13587. [DOI] [PubMed] [Google Scholar]
- Leddy JJ, Master CL, Mannix R, Wiebe DJ, Grady MF, Meehan WP, et al. Early targeted heart rate aerobic exercise versus placebo stretching f or sport-related concussion in adolescents: a randomised controlled trial. Lancet Child Adolesc Health. 2021;5(11):792–799. doi: 10.1016/S2352-4642(21)00267-4. [DOI] [PubMed] [Google Scholar]
- Leiter O, Seidemann S, Overall RW, Ramasz B, Rund N, Schallenberg S, et al. Exercise-induced activated platelets increase adult hippocampal precursor proliferation and promote neuronal differentiation. Stem Cell Reports. 2019;12(4):667–679. doi: 10.1016/j.stemcr.2019.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leiter O, Zhuo Z, Rust R, Wasielewska JM, Grönnert L, Kowal S, et al. Selenium mediates exercise-induced adult neurogenesis and reverses learning deficits induced by hippocampal injury and aging. Cell Metab. 2022;34(3):408. doi: 10.1016/j.cmet.2022.01.005. [DOI] [PubMed] [Google Scholar]
- Lev-Vachnish Y, Cadury S, Rotter-Maskowitz A, Feldman N, Roichman A, Illouz T, et al. L-lactate promotes adult hippocampal neurogenesis. Front Neurosci. 2019;13:403. doi: 10.3389/fnins.2019.00403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F, Geng X, Yun HJ, Haddad Y, Chen Y, Ding Y. Neuroplastic effect of exercise through astrocytes activation and cellular crosstalk. Aging Dis. 2021;12(7):1644–1657. doi: 10.14336/AD.2021.0325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li C, Hu J, Liu W, Ke C, Huang C, Bai Y, et al. Exercise intervention modulates synaptic plasticity by inhibiting excessive microglial activation via exosomes. Front Cell Neurosci. 2022;16:953640. doi: 10.3389/fncel.2022.953640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang J, Wang H, Zeng Y, Qu Y, Liu Q, Zhao F, et al. Physical exercise promotes brain remodeling by regulating epigenetics, neuroplasticity and neurotrophins. Rev Neurosci. 2021;32(6):615–629. doi: 10.1515/revneuro-2020-0099. [DOI] [PubMed] [Google Scholar]
- Liu T, Zhang T, Nicolas M, Boussicault L, Rice H, Soldano A, et al. The amyloid precursor protein is a conserved Wnt receptor. Elife. 2021;10. [DOI] [PMC free article] [PubMed]
- López-Ortiz S, Valenzuela PL, Seisdedos MM, Morales JS, Vega T, Castillo-García A, et al. Exercise interventions in Alzheimer’s disease: a systematic review and meta-analysis of randomized controlled trials. Ageing Res Rev. 2021;72:101479. doi: 10.1016/j.arr.2021.101479. [DOI] [PubMed] [Google Scholar]
- López-Ortiz S, Pinto-Fraga J, Valenzuela PL, Martín-Hernández J, Seisdedos MM, García-López O, et al. Physical exercise and Alzheimer’s disease: effects on pathophysiological molecular pathways of the disease. Int J Mol Sci. 2021;22(6):2897. doi: 10.3390/ijms22062897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma C, Wang J, Liu H, Chen Y, Ma X, Chen S, et al. Moderate exercise enhances endothelial progenitor cell exosomes release and function. Med Sci Sports Exerc. 2018;50(10):2024–2032. doi: 10.1249/MSS.0000000000001672. [DOI] [PubMed] [Google Scholar]
- Maddock RJ, Casazza GA, Fernandez DH, Maddock MI. Acute modulation of cortical glutamate and GABA content by physical activity. J Neurosci. 2016;36(8):2449–2457. doi: 10.1523/JNEUROSCI.3455-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Magee JC, Grienberger C. Synaptic plasticity forms and functions. Annu Rev Neurosci. 2020;43:95. doi: 10.1146/annurev-neuro-090919-022842. [DOI] [PubMed] [Google Scholar]
- Mahdi A, Kövamees O, Pernow J. Improvement in endothelial function in cardiovascular disease—is arginase the target? Int J Cardiol. 2020;301:207–214. doi: 10.1016/j.ijcard.2019.11.004. [DOI] [PubMed] [Google Scholar]
- Mankhong S, Kim S, Moon S, Lee KH, Jeon HE, Hwang BH, et al. Effects of aerobic exercise on tau and related proteins in rats with the middle cerebral artery occlusion. Int J Mol Sci. 2020;21(16):5842. doi: 10.3390/ijms21165842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee AC, Stern RA, Nowinski CJ, Stein TD, Alvarez VE, Daneshvar DH, et al. The spectrum of disease in chronic traumatic encephalopathy. Brain. 2013;136(Pt 1):43–64. doi: 10.1093/brain/aws307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee AC, Stein TD, Kiernan PT, Alvarez VE. The neuropathology of chronic traumatic encephalopathy. Brain Pathol (zurich, Switzerland) 2015;25(3):350–364. doi: 10.1111/bpa.12248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee AC, Cairns NJ, Dickson DW, Folkerth RD, Keene CD, Litvan I, et al. The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol. 2016;131(1):75–86. doi: 10.1007/s00401-015-1515-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKee AC, Stein TD, Huber BR, Crary JF, Bieniek K, Dickson D, et al. Chronic traumatic encephalopathy (CTE): criteria for neuropathological diagnosis and relationship to repetitive head impacts. Acta Neuropathol. 2023;145(4):371–394. doi: 10.1007/s00401-023-02540-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMeekin LJ, Bartley AF, Bohannon AS, Adlaf EW, van Groen T, Boas SM, et al. A role for PGC-1α in transcription and excitability of neocortical and hippocampal excitatory neurons. Neuroscience. 2020;435:73–94. doi: 10.1016/j.neuroscience.2020.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mez J, Alosco ML, Daneshvar DH, Saltiel N, Baucom Z, Abdolmohammadi B, et al. Validity of the 2014 traumatic encephalopathy syndrome criteria for CTE pathology. Alzheimers Dement. 2021;17(10):1709–1724. doi: 10.1002/alz.12338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morland C, Andersson KA, Haugen ØP, Hadzic A, Kleppa L, Gille A, et al. Exercise induces cerebral VEGF and angiogenesis via the lactate recept or HCAR1. Nat Commun. 2017;8:15557. doi: 10.1038/ncomms15557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morland C, Andersson KA, Haugen OP, Hadzic A, Kleppa L, Gille A, et al. Exercise induces cerebral VEGF and angiogenesis via the lactate receptor HCAR1. Nat Commun. 2017;8:15557. doi: 10.1038/ncomms15557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moszczynski AJ, Strong W, Xu K, McKee A, Brown A, Strong MJ. Pathologic Thr tau phosphorylation in CTE and CTE with ALS. Neurology. 2018;90(5):e380–e387. doi: 10.1212/WNL.0000000000004899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mu L, Cai J, Gu B, Yu L, Li C, Liu Q-S, et al. Treadmill exercise prevents decline in spatial learning and memory in 3×Tg-AD mice through enhancement of structural synaptic plasticity of the hippocampus and prefrontal cortex. Cells. 2022;11(2):244. doi: 10.3390/cells11020244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray HC, Osterman C, Bell P, Vinnell L, Curtis MA. Neuropathology in chronic traumatic encephalopathy: a systematic review of comparative post-mortem histology literature. Acta Neuropathol Commun. 2022;10(1):108. doi: 10.1186/s40478-022-01413-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neto IVS, Pinto AP, Muñoz VR, de Cássia Marqueti R, Pauli JR, Ropelle ER, et al. Pleiotropic and multi-systemic actions of physical exercise on PGC-1α signaling during the aging process. Ageing Res Rev. 2023;87:101935. doi: 10.1016/j.arr.2023.101935. [DOI] [PubMed] [Google Scholar]
- Nigam SM, Xu S, Kritikou JS, Marosi K, Brodin L, Mattson MP. Exercise and BDNF reduce Aβ production by enhancing α-secretase processing of APP. J Neurochem. 2017;142(2):286–296. doi: 10.1111/jnc.14034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Northey JM, Cherbuin N, Pumpa KL, Smee DJ, Rattray B. Exercise interventions for cognitive function in adults older than 50: a systematic review with meta-analysis. Br J Sports Med. 2018;52(3):154–160. doi: 10.1136/bjsports-2016-096587. [DOI] [PubMed] [Google Scholar]
- Pang L, Khan F, Dunterman M, Chen P. Pharmacological targeting of the tumor-immune symbiosis in glioblastom a. Trends Pharmacol Sci. 2022;43(8):686–700. doi: 10.1016/j.tips.2022.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pang L, Khan F, Heimberger AB, Chen P. Mechanism and therapeutic potential of tumor-immune symbiosis in glioblastoma. Trends Cancer. 2022;8(10):839–854. doi: 10.1016/j.trecan.2022.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearce M, Garcia L, Abbas A, Strain T, Schuch FB, Golubic R, et al. Association between physical activity and risk of depression: a systematic review and meta-analysis. JAMA Psychiat. 2022;79(6):550–559. doi: 10.1001/jamapsychiatry.2022.0609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedersen BK, Febbraio MA. Muscles, exercise and obesity: skeletal muscle as a secretory organ. Nat Rev Endocrinol. 2012;8(8):457–465. doi: 10.1038/nrendo.2012.49. [DOI] [PubMed] [Google Scholar]
- Philippot A, Dubois V, Lambrechts K, Grogna D, Robert A, Jonckheer U, et al. Impact of physical exercise on depression and anxiety in adolescent inpatients: a randomized controlled trial. J Affect Disord. 2022;301:145–153. doi: 10.1016/j.jad.2022.01.011. [DOI] [PubMed] [Google Scholar]
- Ross MD, Wekesa AL, Phelan JP, Harrison M. Resistance exercise increases endothelial progenitor cells and angiogenic factors. Med Sci Sports Exerc. 2014;46(1):16–23. doi: 10.1249/MSS.0b013e3182a142da. [DOI] [PubMed] [Google Scholar]
- Ruiz-González D, Hernández-Martínez A, Valenzuela PL, Morales JS, Soriano-Maldonado A. Effects of physical exercise on plasma brain-derived neurotrophic factor in neurodegenerative disorders: a systematic review and meta-analysis of randomized controlled trials. Neurosci Biobehav Rev. 2021;128:394–405. doi: 10.1016/j.neubiorev.2021.05.025. [DOI] [PubMed] [Google Scholar]
- Sacramento CB, Sondhi D, Rosenberg JB, Chen A, Giordano S, Pey E, et al. Anti-phospho-tau gene therapy for chronic traumatic encephalopathy. Hum Gene Ther. 2020;31(1–2):57–69. doi: 10.1089/hum.2019.174. [DOI] [PubMed] [Google Scholar]
- Shin N, Kim H-G, Shin HJ, Kim S, Kwon HH, Baek H, et al. Uncoupled endothelial nitric oxide synthase enhances p-tau in chronic traumatic encephalopathy mouse model. Antioxid Redox Signal. 2019;30(13):1601–1620. doi: 10.1089/ars.2017.7280. [DOI] [PubMed] [Google Scholar]
- Smith PJ, Merwin RM. The role of exercise in management of mental health disorders: an integrative review. Annu Rev Med. 2021;72:45–62. doi: 10.1146/annurev-med-060619-022943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DH, Johnson VE, Trojanowski JQ, Stewart W. Chronic traumatic encephalopathy—confusion and controversies. Nat Rev Neurol. 2019;15(3):179–183. doi: 10.1038/s41582-018-0114-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steventon JJ, Foster C, Furby H, Helme D, Wise RG, Murphy K. Hippocampal blood flow is increased after 20 min of moderate-intensity exercise. Cereb Cortex. 2020;30(2):525–533. doi: 10.1093/cercor/bhz104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Shima T, Soya M, Yook JS, Koizumi H, Jesmin S, et al. Exercise-induced adrenocorticotropic hormone response is cooperatively regulated by hypothalamic arginine vasopressin and corticotrophin-releasing hormone. Neuroendocrinology. 2022;112(9):894–903. doi: 10.1159/000521237. [DOI] [PubMed] [Google Scholar]
- Tan Z-X, Dong F, Wu L-Y, Feng Y-S, Zhang F. The beneficial role of exercise on treating Alzheimer’s disease by inhibiting β-amyloid peptide. Mol Neurobiol. 2021;58(11):5890–5906. doi: 10.1007/s12035-021-02514-7. [DOI] [PubMed] [Google Scholar]
- Tapanes SA, Arizanovska D, Díaz MM, Folorunso OO, Harvey T, Brown SE, et al. Inhibition of glial D-serine release rescues synaptic damage after brain injury. Glia. 2022;70(6):1133–1152. doi: 10.1002/glia.24161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornhill MH, Haskard DO. IL-4 regulates endothelial cell activation by IL-1, tumor necrosis factor, or IFN-gamma. J Immunol. 1990;145(3):865–872. [PubMed] [Google Scholar]
- Tryfonos A, Rasoul D, Sadler D, Shelley J, Mills J, Green DJ, et al. Elevated shear rate-induced by exercise increases eNOS ser1177 but not PECAM-1 Tyr713 phosphorylation in human conduit artery endothelial cells. Eur J Sport Sci. 2022;23:561. doi: 10.1080/17461391.2022.2046175. [DOI] [PubMed] [Google Scholar]
- Valenzuela PL, Castillo-García A, Morales JS, de la Villa P, Hampel H, Emanuele E, et al. Exercise benefits on Alzheimer’s disease: state-of-the-science. Ageing Res Rev. 2020;62:101108. doi: 10.1016/j.arr.2020.101108. [DOI] [PubMed] [Google Scholar]
- Verboon LN, Patel HC, Greenhalgh AD. The immune system’s role in the consequences of mild traumatic brain injury (Concussion) Front Immunol. 2021;12:620698. doi: 10.3389/fimmu.2021.620698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vints WAJ, Levin O, Fujiyama H, Verbunt J, Masiulis N. Exerkines and long-term synaptic potentiation: mechanisms of exercise-induced neuroplasticity. Front Neuroendocrinol. 2022;66:100993. doi: 10.1016/j.yfrne.2022.100993. [DOI] [PubMed] [Google Scholar]
- Vivar C, van Praag H. Running changes the brain: the long and the short of it. Physiology (bethesda) 2017;32(6):410–424. doi: 10.1152/physiol.00017.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsh JJ, Tschakovsky ME. Exercise and circulating BDNF: mechanisms of release and implications for the design of exercise interventions. Appl Physiol Nutr Metab. 2018;43(11):1095–1104. doi: 10.1139/apnm-2018-0192. [DOI] [PubMed] [Google Scholar]
- Wang J, Liu H, Chen S, Zhang W, Chen Y, Yang Y. Moderate exercise has beneficial effects on mouse ischemic stroke by enhancing the functions of circulating endothelial progenitor cell-derived exosomes. Exp Neurol. 2020;330:113325. doi: 10.1016/j.expneurol.2020.113325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weerasinghe-Mudiyanselage PDE, Ang MJ, Kang S, Kim J-S, Moon C. Structural plasticity of the hippocampus in neurodegenerative diseases. Int J Mol Sci. 2022;23(6):3349. doi: 10.3390/ijms23063349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkins HM, Swerdlow RH. Amyloid precursor protein processing and bioenergetics. Brain Res Bull. 2017;133:71–79. doi: 10.1016/j.brainresbull.2016.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willis EF, MacDonald KPA, Nguyen QH, Garrido AL, Gillespie ER, Harley SBR, et al. Repopulating microglia promote brain repair in an IL-6-dependent manner. Cell. 2020;180(5):833–846. doi: 10.1016/j.cell.2020.02.013. [DOI] [PubMed] [Google Scholar]
- Wolosker H, Balu DT, Coyle JT. The rise and fall of the d-serine-mediated gliotransmission hypothesis. Trends Neurosci. 2016;39(11):712–721. doi: 10.1016/j.tins.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu L, Li M, Wei A, Yang M, Li C, Liu R, et al. Treadmill exercise promotes E3 ubiquitin ligase to remove amyloid β and P-tau and improve cognitive ability in APP/PS1 transgenic mice. J Neuroinflammation. 2022;19(1):243. doi: 10.1186/s12974-022-02607-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu L, Li M, Wei A, Yang M, Li C, Liu R, et al. Treadmill exercise promotes E3 ubiquitin ligase to remove amyloid beta and P-tau and improve cognitive ability in APP/PS1 transgenic mice. J Neuroinflammation. 2022;19(1):243. doi: 10.1186/s12974-022-02607-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xuan W, Hsu W-H, Khan F, Dunterman M, Pang L, Wainwright DA, et al. Circadian regulator CLOCK drives immunosuppression in glioblastoma. Cancer Immunol Res. 2022;10(6):770–784. doi: 10.1158/2326-6066.CIR-21-0559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J-T, Xu W, Tan C-C, Andrieu S, Suckling J, Evangelou E, et al. Evidence-based prevention of Alzheimer’s disease: systematic review and meta-analysis of 243 observational prospective studies and 153 randomised controlled trials. J Neurol Neurosurg Psychiatry. 2020;91(11):1201–1209. doi: 10.1136/jnnp-2019-321913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu H, Zhang C, Xia J, Xu B. Treadmill exercise ameliorates adult hippocampal neurogenesis possibly by adjusting the APP proteolytic pathway in APP/PS1 transgenic mice. Int J Mol Sci. 2021;22(17):9570. doi: 10.3390/ijms22179570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X, Zhang R, Wei C, Gao Y, Yu Y, Wang L, et al. MCT2 overexpression promotes recovery of cognitive function by increasing mitochondrial biogenesis in a rat model of stroke. Anim Cells Syst (seoul). 2021;25(2):93. doi: 10.1080/19768354.2021.1915379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C, Wei C, Huang X, Hou C, Liu C, Zhang S, et al. MPC-n (IgG) improves long-term cognitive impairment in the mouse model of repetitive mild traumatic brain injury. BMC Med. 2023;21(1):199. doi: 10.1186/s12916-023-02895-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao N, Xia J, Xu B. Physical exercise may exert its therapeutic influence on Alzheimer’s disease through the reversal of mitochondrial dysfunction via SIRT1-FOXO1/3-PINK1-Parkin-mediated mitophagy. J Sport Health Sci. 2021;10(1):1–3. doi: 10.1016/j.jshs.2020.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Y, Su Y, Li S, Kennedy BC, Zhang DY, Bond AM, et al. Molecular landscapes of human hippocampal immature neurons across lifespan. Nature. 2022;607(7919):527–533. doi: 10.1038/s41586-022-04912-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All relevant data is contained within the article. Further inquiries can be directed to the corresponding author.