Abstract
Plants synthesize betaine by a two-step oxidation of choline (choline → betaine aldehyde → betaine). Protoplast-derived chloroplasts of spinach (Spinacia oleracea L.) carry out both reactions, more rapidly in light than in darkness (AD Hanson et al. 1985 Proc Natl Acad Sci USA 82: 3678-3682). We investigated the light-stimulated oxidation of choline, using spinach chloroplasts isolated directly from leaves. The rates of choline oxidation obtained (dark and light rates: 10-50 and 100-300 nanomoles per hour per milligram chlorophyll, respectively) were approximately 20-fold higher than for protoplast-derived chloroplasts. Betaine aldehyde was the main product. Choline oxidation in darkness and light was suppressed by hypoxia. Neither uncouplers nor the Calvin cycle inhibitor glyceraldehyde greatly affected choline oxidation in the light, and maximal choline oxidation was attained far below light saturation of CO2 fixation. The light stimulation of choline oxidation was abolished by the PSII inhibitors DCMU and dibromothymoquinone, and was partially restored by adding reduced diaminodurene, an electron donor to PSI. Both methyl viologen and phenazine methosulfate prevented choline oxidation. Adding dihydroxyacetone phosphate, which can generate NADPH in organello, doubled the dark rate of choline oxidation. These results indicate that choline oxidation in chloroplasts requires oxygen, and reducing power generated from PSI. Enzymic reactions consistent with these requirements are discussed.
Full text
PDF


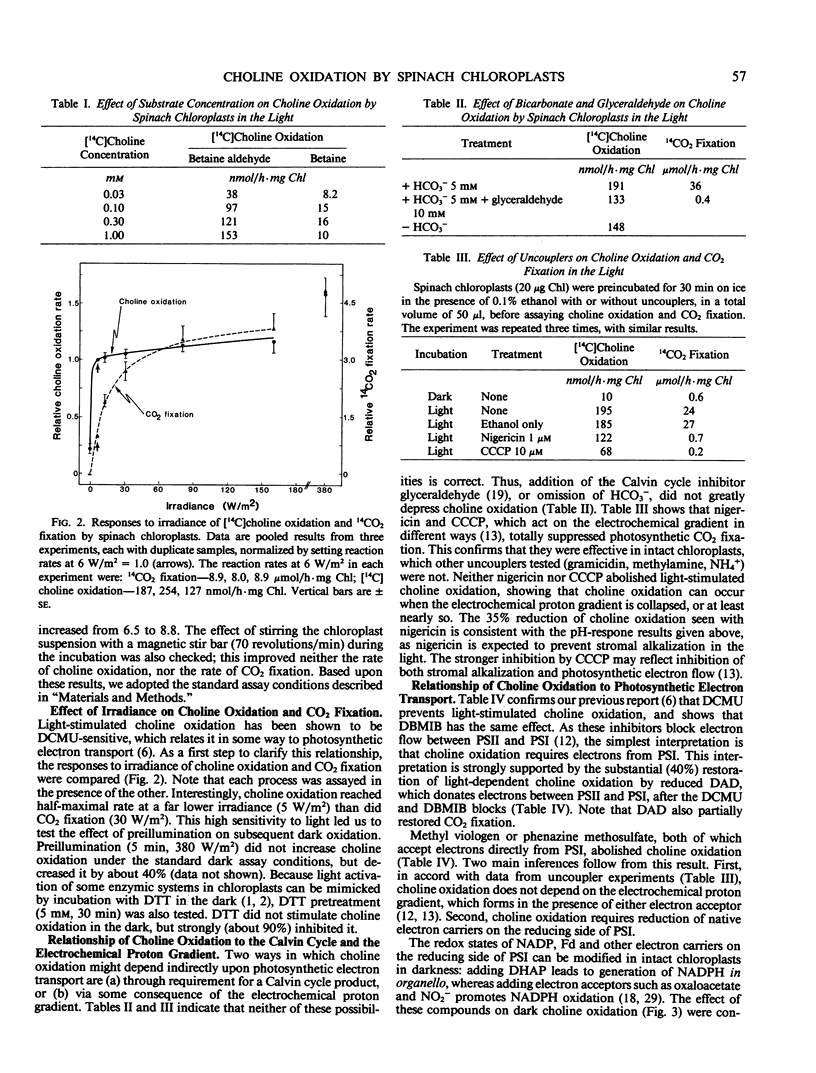



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Hanson A. D., May A. M., Grumet R., Bode J., Jamieson G. C., Rhodes D. Betaine synthesis in chenopods: Localization in chloroplasts. Proc Natl Acad Sci U S A. 1985 Jun;82(11):3678–3682. doi: 10.1073/pnas.82.11.3678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikuta S., Imamura S., Misaki H., Horiuti Y. Purification and characterization of choline oxidase from Arthrobacter globiformis. J Biochem. 1977 Dec;82(6):1741–1749. doi: 10.1093/oxfordjournals.jbchem.a131872. [DOI] [PubMed] [Google Scholar]
- JELLINEK M., STRENGTH D. R., THAYER S. A. Isolation and identification of the products of the oxidation of choline. J Biol Chem. 1959 May;234(5):1171–1173. [PubMed] [Google Scholar]
- Keilin D., Hartree E. F. Properties of catalase. Catalysis of coupled oxidation of alcohols. Biochem J. 1945;39(4):293–301. [PMC free article] [PubMed] [Google Scholar]
- Kortstee G. J. The aerobic decomposition of choline by microorganisms. I. The ability of aerobic organisms, particularly coryneform bacteria, to utilize choline as the sole carbon and nitrogen source. Arch Mikrobiol. 1970;71(3):235–244. [PubMed] [Google Scholar]
- Landfald B., Strøm A. R. Choline-glycine betaine pathway confers a high level of osmotic tolerance in Escherichia coli. J Bacteriol. 1986 Mar;165(3):849–855. doi: 10.1128/jb.165.3.849-855.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leegood R. C., Walker D. A. Activation of fructose 1,6-bisphosphatase in darkened intact chloroplasts by NADPH. Arch Biochem Biophys. 1981 Dec;212(2):644–650. doi: 10.1016/0003-9861(81)90408-2. [DOI] [PubMed] [Google Scholar]
- Loechler E. L., Hollocher T. C. Letter: Mechanism of the reaction of dithiols with flavins. J Am Chem Soc. 1975 May 28;97(11):3235–3237. doi: 10.1021/ja00844a062. [DOI] [PubMed] [Google Scholar]
- McKeon T. A., Stumpf P. K. Purification and characterization of the stearoyl-acyl carrier protein desaturase and the acyl-acyl carrier protein thioesterase from maturing seeds of safflower. J Biol Chem. 1982 Oct 25;257(20):12141–12147. [PubMed] [Google Scholar]
- Rhodes D., Rich P. J., Myers A. C., Reuter C. C., Jamieson G. C. Determination of Betaines by Fast Atom Bombardment Mass Spectrometry : Identification of Glycine Betaine Deficient Genotypes of Zea mays. Plant Physiol. 1987 Jul;84(3):781–788. doi: 10.1104/pp.84.3.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuge H., Nakano Y., Onishi H., Futamura Y., Ohashi K. A novel purification and some properties of rat liver mitochondrial choline dehydrogenase. Biochim Biophys Acta. 1980 Aug 7;614(2):274–284. doi: 10.1016/0005-2744(80)90217-x. [DOI] [PubMed] [Google Scholar]
- Weigel P., Weretilnyk E. A., Hanson A. D. Betaine aldehyde oxidation by spinach chloroplasts. Plant Physiol. 1986 Nov;82(3):753–759. doi: 10.1104/pp.82.3.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Werdan K., Heldt H. W., Milovancev M. The role of pH in the regulation of carbon fixation in the chloroplast stroma. Studies on CO2 fixation in the light and dark. Biochim Biophys Acta. 1975 Aug 11;396(2):276–292. doi: 10.1016/0005-2728(75)90041-9. [DOI] [PubMed] [Google Scholar]
- Yancey P. H., Clark M. E., Hand S. C., Bowlus R. D., Somero G. N. Living with water stress: evolution of osmolyte systems. Science. 1982 Sep 24;217(4566):1214–1222. doi: 10.1126/science.7112124. [DOI] [PubMed] [Google Scholar]