Abstract
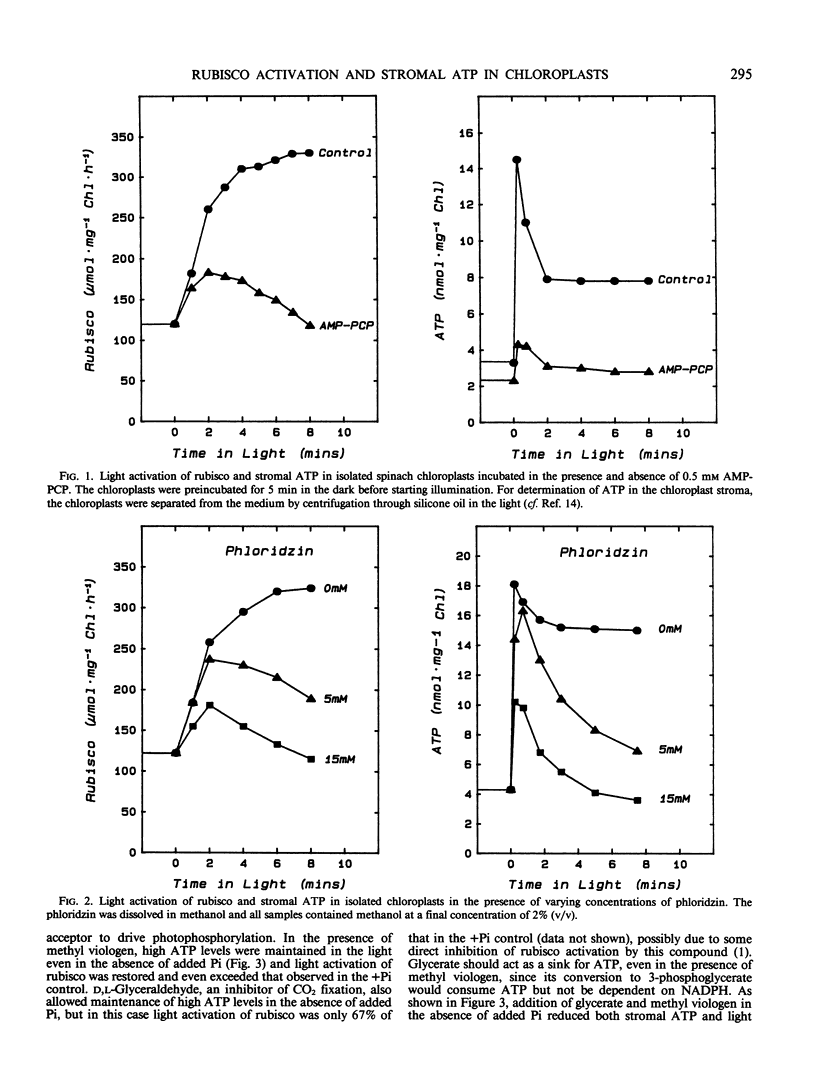
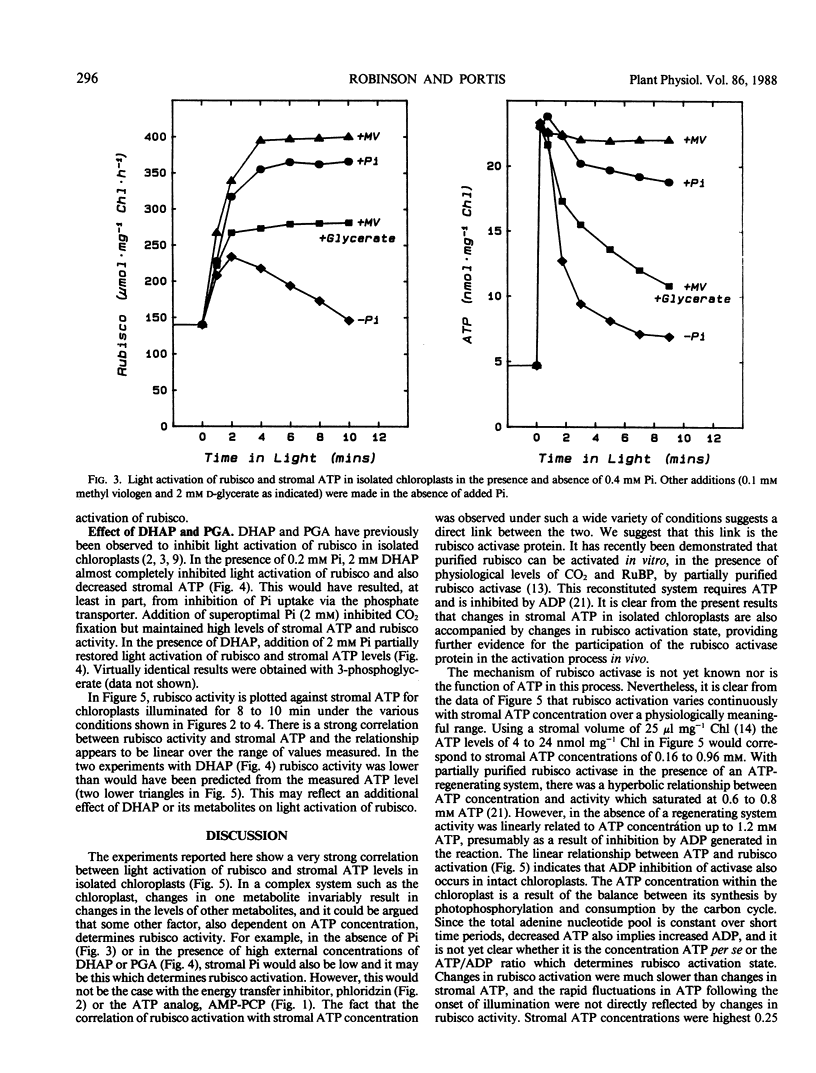
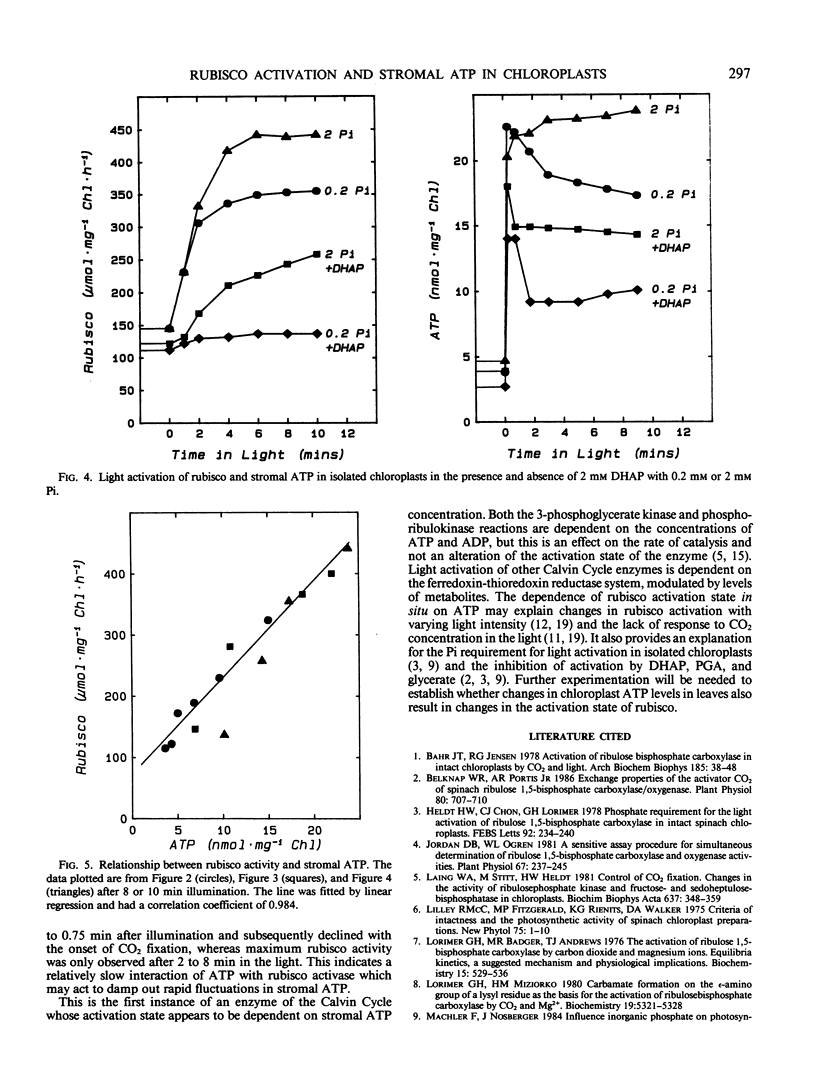
Light activation of ribulose-1,5-bisphosphate carboxylase/oxygenase (rubisco) and stromal ATP content were measured in intact isolated spinach chloroplasts. Treatments which decreased stromal ATP, such as incubation with the ATP analog β,γ-methylene adenosine triphosphate or with the energy transfer inhibitor phloridzin inhibited the light activation of rubisco. In the absence of added inorganic phosphate (Pi), light activation of rubisco was inhibited, coincident with low stromal ATP. Addition of methyl viologen restored both stromal ATP and rubisco activity to levels observed in the presence of Pi. Activation of rubisco was inhibited in the presence of 2 millimolar dihydroxyacetone phosphate or 3-phosphoglycerate and stromal ATP was also decreased under these conditions. Both were partially restored by increasing the Pi concentration. The strong correlation between activation state of rubisco and stromal ATP concentration in intact chloroplasts under a wide variety of experimental conditions indicates that light activation of rubisco is dependent on ATP and proportional to the ATP concentration. These observations can be explained in terms of the rubisco activase protein, which mediates activation of rubisco at physiological concentrations of CO2 and ribulose-1,5-bisphosphate and is dependent upon ATP.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bahr J. T., Jensen R. G. Activation of ribulose bisphosphate carboxylase in intact chloroplasts by CO2 and light. Arch Biochem Biophys. 1978 Jan 15;185(1):39–48. doi: 10.1016/0003-9861(78)90141-8. [DOI] [PubMed] [Google Scholar]
- Belknap W. R., Portis A. R. Exchange Properties of the Activator CO(2) of Spinach Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase. Plant Physiol. 1986 Mar;80(3):707–710. doi: 10.1104/pp.80.3.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan D. B., Ogren W. L. A Sensitive Assay Procedure for Simultaneous Determination of Ribulose-1,5-bisphosphate Carboxylase and Oxygenase Activities. Plant Physiol. 1981 Feb;67(2):237–245. doi: 10.1104/pp.67.2.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorimer G. H., Badger M. R., Andrews T. J. The activation of ribulose-1,5-bisphosphate carboxylase by carbon dioxide and magnesium ions. Equilibria, kinetics, a suggested mechanism, and physiological implications. Biochemistry. 1976 Feb 10;15(3):529–536. doi: 10.1021/bi00648a012. [DOI] [PubMed] [Google Scholar]
- Lorimer G. H., Miziorko H. M. Carbamate formation on the epsilon-amino group of a lysyl residue as the basis for the activation of ribulosebisphosphate carboxylase by CO2 and Mg2+. Biochemistry. 1980 Nov 11;19(23):5321–5328. doi: 10.1021/bi00564a027. [DOI] [PubMed] [Google Scholar]
- Miziorko H. M., Lorimer G. H. Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem. 1983;52:507–535. doi: 10.1146/annurev.bi.52.070183.002451. [DOI] [PubMed] [Google Scholar]
- Perchorowicz J. T., Jensen R. G. Photosynthesis and Activation of Ribulose Bisphosphate Carboxylase in Wheat Seedlings : Regulation by CO(2) and O(2). Plant Physiol. 1983 Apr;71(4):955–960. doi: 10.1104/pp.71.4.955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perchorowicz J. T., Raynes D. A., Jensen R. G. Light limitation of photosynthesis and activation of ribulose bisphosphate carboxylase in wheat seedlings. Proc Natl Acad Sci U S A. 1981 May;78(5):2985–2989. doi: 10.1073/pnas.78.5.2985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Portis A. R., Salvucci M. E., Ogren W. L. Activation of Ribulosebisphosphate Carboxylase/Oxygenase at Physiological CO(2) and Ribulosebisphosphate Concentrations by Rubisco Activase. Plant Physiol. 1986 Dec;82(4):967–971. doi: 10.1104/pp.82.4.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson S. P., Wiskich J. T. Uptake of ATP analogs by isolated pea chloroplasts and their effect on CO2 fixation and electron transport. Biochim Biophys Acta. 1977 Jul 7;461(1):131–140. doi: 10.1016/0005-2728(77)90075-5. [DOI] [PubMed] [Google Scholar]
- Salvucci M. E., Portis A. R., Ogren W. L. Light and CO(2) Response of Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase Activation in Arabidopsis Leaves. Plant Physiol. 1986 Mar;80(3):655–659. doi: 10.1104/pp.80.3.655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somerville C. R., Portis A. R., Ogren W. L. A Mutant of Arabidopsis thaliana Which Lacks Activation of RuBP Carboxylase In Vivo. Plant Physiol. 1982 Aug;70(2):381–387. doi: 10.1104/pp.70.2.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Streusand V. J., Portis A. R. Rubisco Activase Mediates ATP-Dependent Activation of Ribulose Bisphosphate Carboxylase. Plant Physiol. 1987 Sep;85(1):152–154. doi: 10.1104/pp.85.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]


