Abstract
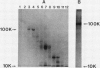
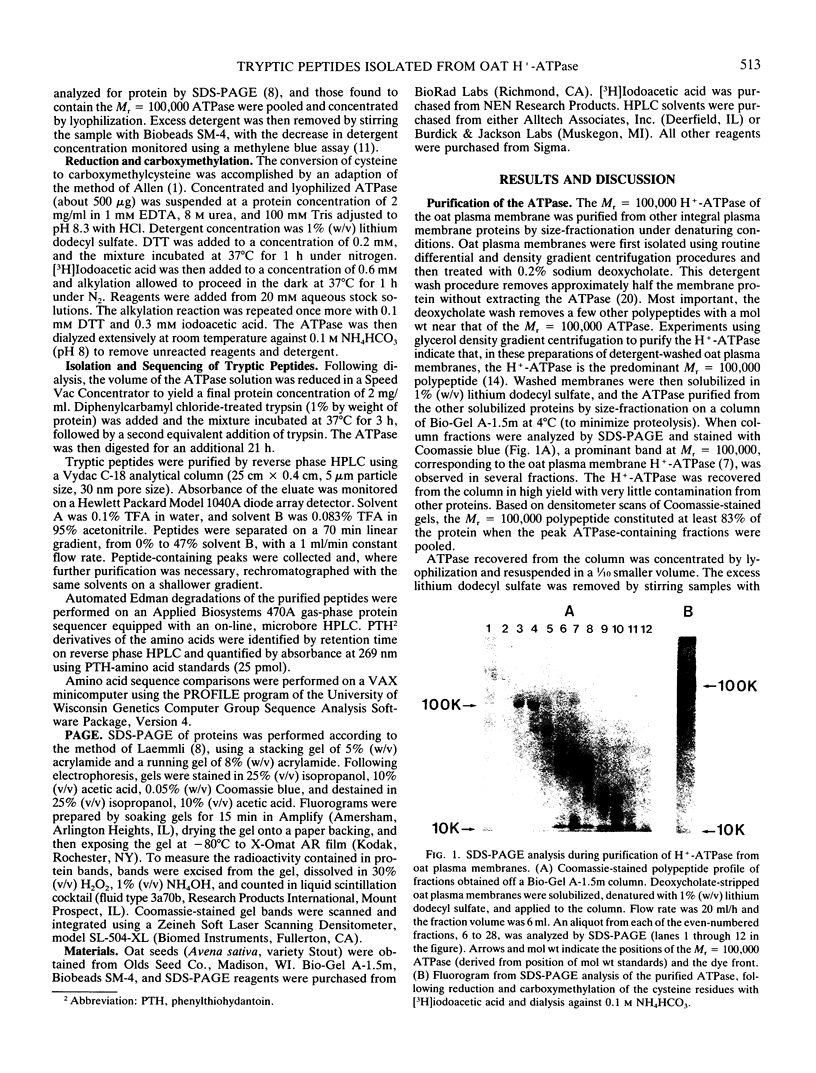
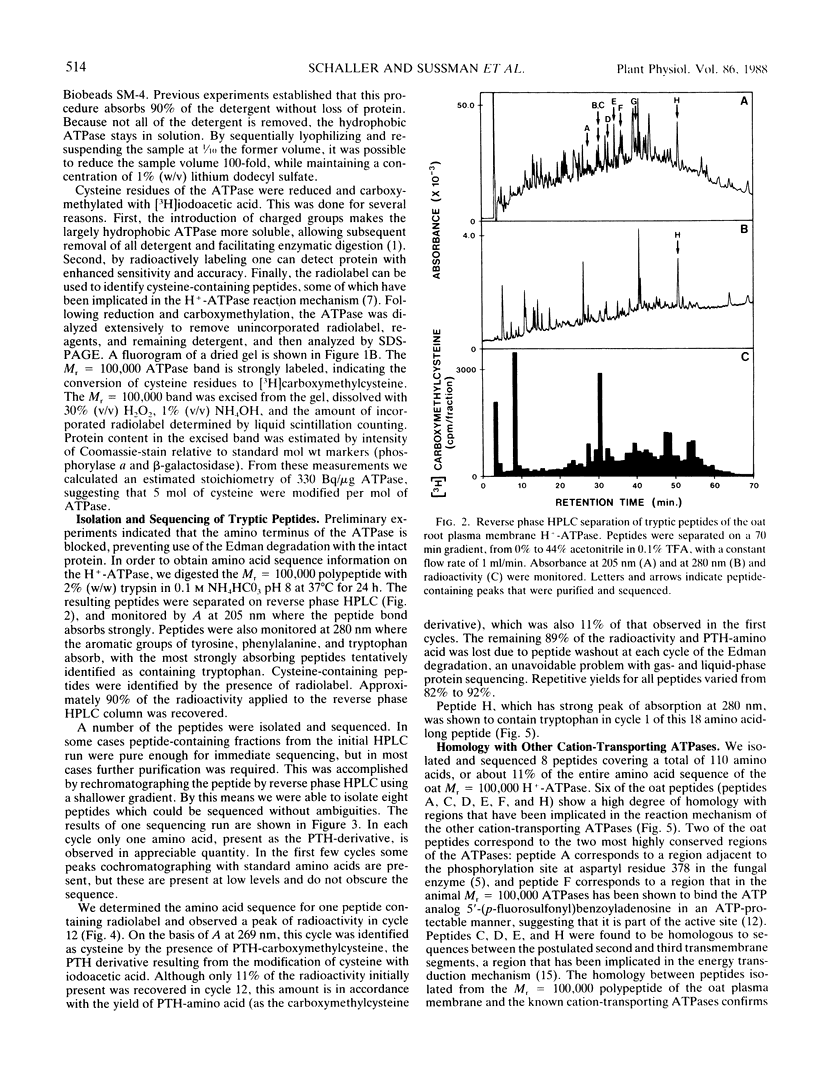
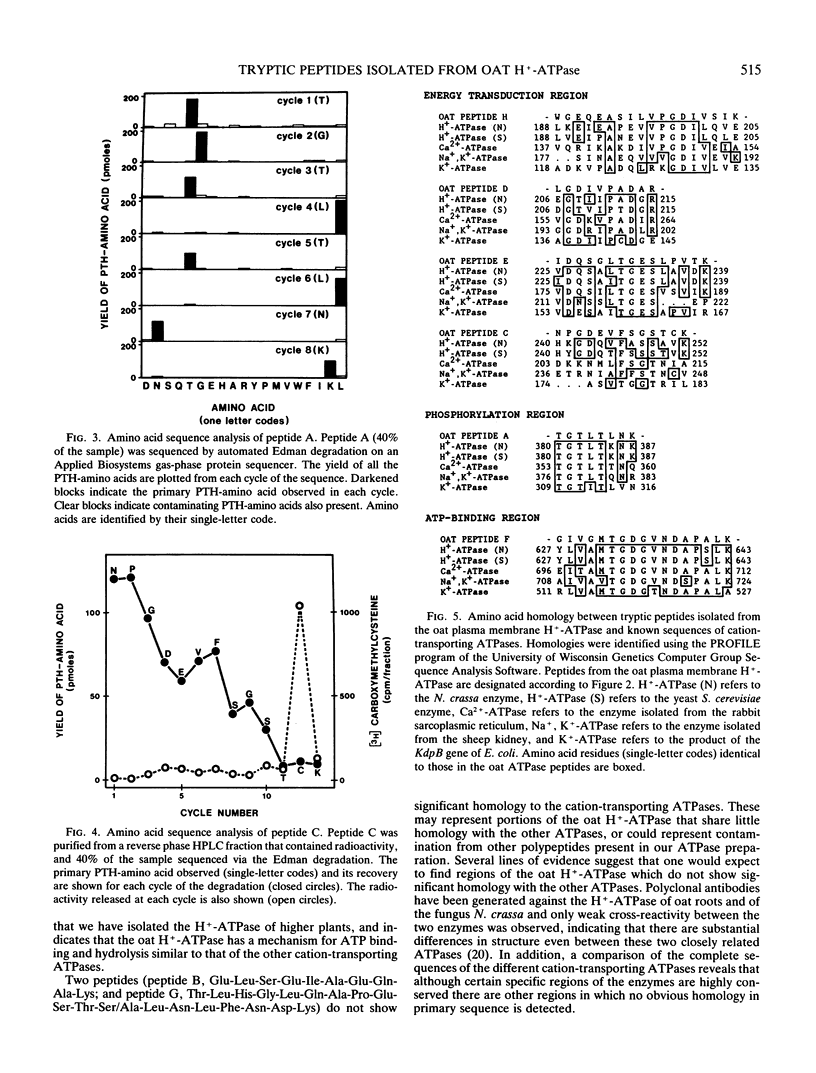
In crude extracts of plant tissue, the Mr = 100,000 proton-pumping ATPase constitutes less than 0.01% of the total cell protein. A large-scale purification procedure is described that has been used to obtain extensive protein sequence information from this enzyme. Plasma membrane vesicles enriched in ATPase activity were obtained from extracts of oat roots by routine differential and density gradient centrifugation. Following a detergent wash, the ATPase was resolved from other integral membrane proteins by size fractionation at 4°C in the presence of lithium dodecyl sulfate. After carboxymethylation of cysteine residues and removal of detergent, the ATPase was digested with trypsin and resultant peptide fragments separated by reverse phase high performance liquid chromatography. Peptides were recovered with high yield and were readily sequenced by automated Edman degradation on a gas-phase sequencer. Of the eight peptides sequenced, six showed strong homology with known amino acid sequences of the fungal proton-pumping and other cation-transporting ATPases.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anthon G. E., Spanswick R. M. Purification and properties of the h-translocating ATPase from the plasma membrane of tomato roots. Plant Physiol. 1986 Aug;81(4):1080–1085. doi: 10.1104/pp.81.4.1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bidwai A. P., Zhang L., Bachmann R. C., Takemoto J. Y. Mechanism of Action of Pseudomonas syringae Phytotoxin, Syringomycin : Stimulation of Red Beet Plasma Membrane ATPase Activity. Plant Physiol. 1987 Jan;83(1):39–43. doi: 10.1104/pp.83.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goffeau A., Slayman C. W. The proton-translocating ATPase of the fungal plasma membrane. Biochim Biophys Acta. 1981 Dec 30;639(3-4):197–223. doi: 10.1016/0304-4173(81)90010-0. [DOI] [PubMed] [Google Scholar]
- Hager K. M., Mandala S. M., Davenport J. W., Speicher D. W., Benz E. J., Jr, Slayman C. W. Amino acid sequence of the plasma membrane ATPase of Neurospora crassa: deduction from genomic and cDNA sequences. Proc Natl Acad Sci U S A. 1986 Oct;83(20):7693–7697. doi: 10.1073/pnas.83.20.7693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hesse J. E., Wieczorek L., Altendorf K., Reicin A. S., Dorus E., Epstein W. Sequence homology between two membrane transport ATPases, the Kdp-ATPase of Escherichia coli and the Ca2+-ATPase of sarcoplasmic reticulum. Proc Natl Acad Sci U S A. 1984 Aug;81(15):4746–4750. doi: 10.1073/pnas.81.15.4746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katz D. B., Sussman M. R. Inhibition and Labeling of the Plant Plasma Membrane H-ATPase with N-Ethylmaleimide. Plant Physiol. 1987 Apr;83(4):977–981. doi: 10.1104/pp.83.4.977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- MacLennan D. H., Brandl C. J., Korczak B., Green N. M. Amino-acid sequence of a Ca2+ + Mg2+-dependent ATPase from rabbit muscle sarcoplasmic reticulum, deduced from its complementary DNA sequence. Nature. 1985 Aug 22;316(6030):696–700. doi: 10.1038/316696a0. [DOI] [PubMed] [Google Scholar]
- Ohta T., Nagano K., Yoshida M. The active site structure of Na+/K+-transporting ATPase: location of the 5'-(p-fluorosulfonyl)benzoyladenosine binding site and soluble peptides released by trypsin. Proc Natl Acad Sci U S A. 1986 Apr;83(7):2071–2075. doi: 10.1073/pnas.83.7.2071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleski N. A., Bennett A. B. H-ATPase Activity from Storage Tissue of Beta vulgaris: IV. N,N'-Dicyclohexylcarbodiimide Binding and Inhibition of the Plasma Membrane H-ATPase. Plant Physiol. 1987 Mar;83(3):569–572. doi: 10.1104/pp.83.3.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott T. L., Shamoo A. E. Disruptiin of energy transductiin in sarcoplasmic reticulum by trypsin cleavage of (Ca2+ + Mg2+)-ATPase. J Membr Biol. 1982;64(3):137–144. doi: 10.1007/BF01870879. [DOI] [PubMed] [Google Scholar]
- Serrano R., Kielland-Brandt M. C., Fink G. R. Yeast plasma membrane ATPase is essential for growth and has homology with (Na+ + K+), K+- and Ca2+-ATPases. Nature. 1986 Feb 20;319(6055):689–693. doi: 10.1038/319689a0. [DOI] [PubMed] [Google Scholar]
- Serrano R. Purification of the proton pumping ATPase from plant plasma membranes. Biochem Biophys Res Commun. 1984 Jun 15;121(2):735–740. doi: 10.1016/0006-291x(84)90243-2. [DOI] [PubMed] [Google Scholar]
- Shull G. E., Schwartz A., Lingrel J. B. Amino-acid sequence of the catalytic subunit of the (Na+ + K+)ATPase deduced from a complementary DNA. Nature. 1985 Aug 22;316(6030):691–695. doi: 10.1038/316691a0. [DOI] [PubMed] [Google Scholar]
- Sussman M. R., Strickler J. E., Hager K. M., Slayman C. W. Location of a dicyclohexylcarbodiimide-reactive glutamate residue in the Neurospora crassa plasma membrane H+-ATPase. J Biol Chem. 1987 Apr 5;262(10):4569–4573. [PubMed] [Google Scholar]
- Vai M., Popolo L., Alberghina L. Immunological cross-reactivity of fungal and yeast plasma membrane H+-ATPase. FEBS Lett. 1986 Sep 29;206(1):135–141. doi: 10.1016/0014-5793(86)81355-2. [DOI] [PubMed] [Google Scholar]
- Walderhaug M. O., Post R. L., Saccomani G., Leonard R. T., Briskin D. P. Structural relatedness of three ion-transport adenosine triphosphatases around their active sites of phosphorylation. J Biol Chem. 1985 Mar 25;260(6):3852–3859. [PubMed] [Google Scholar]