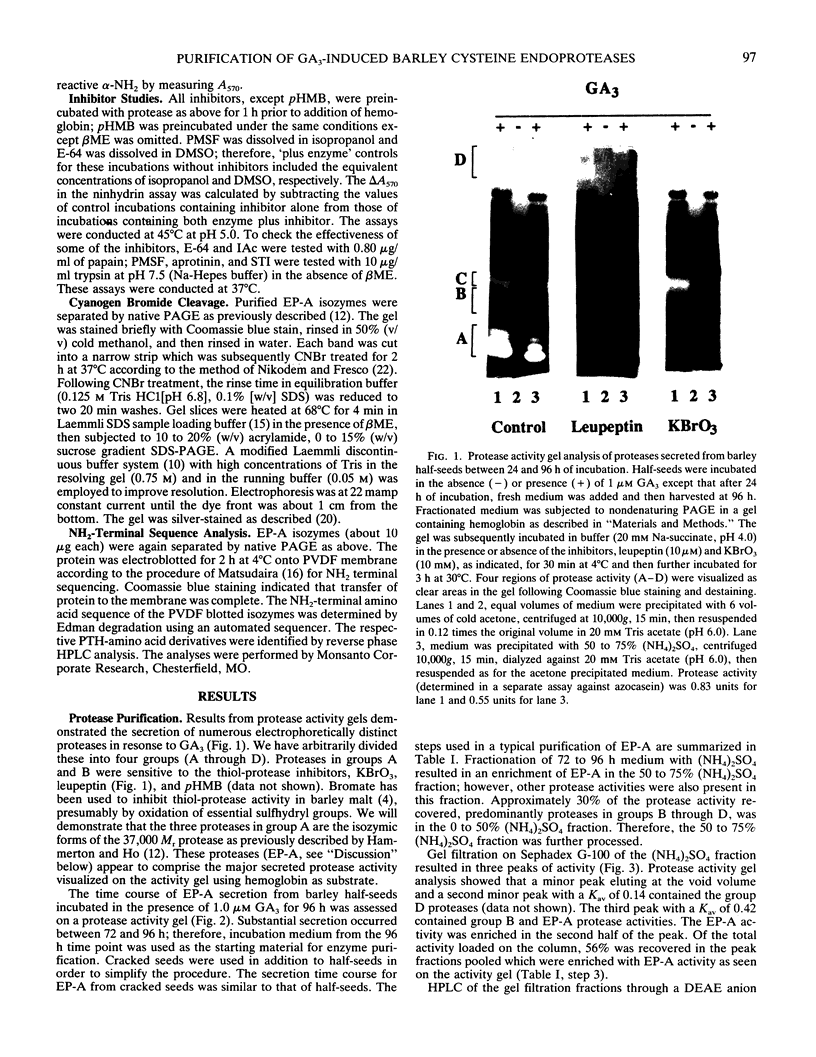
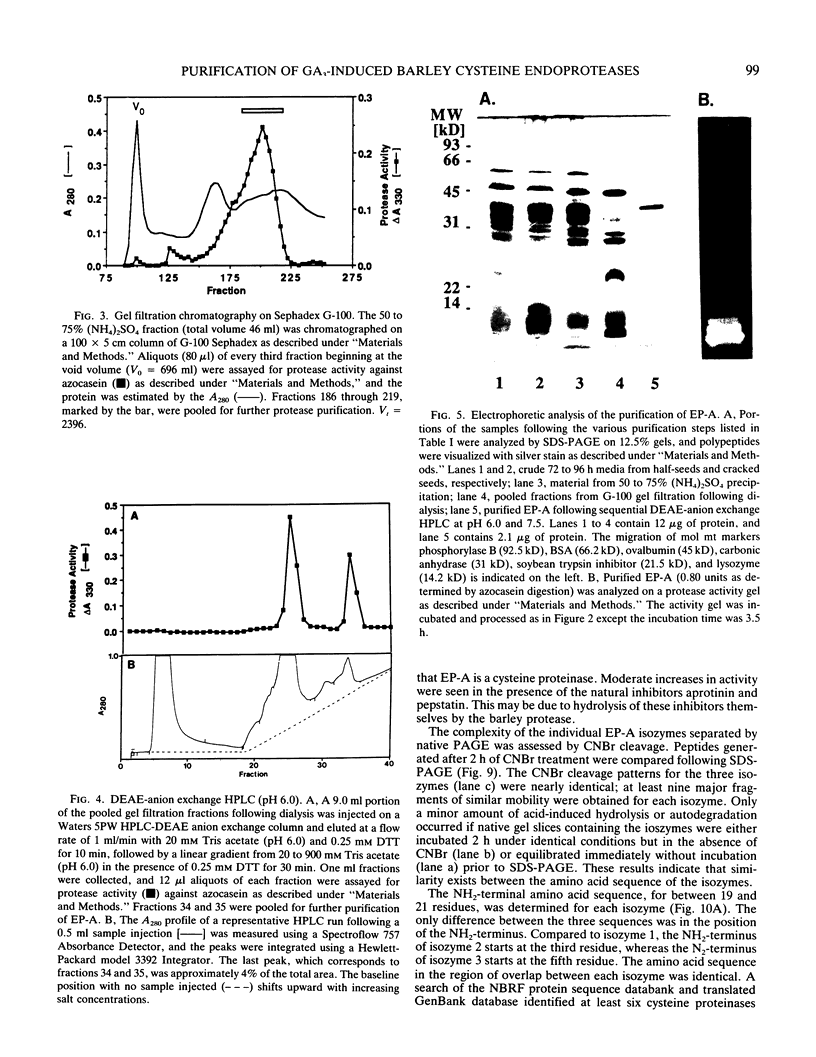
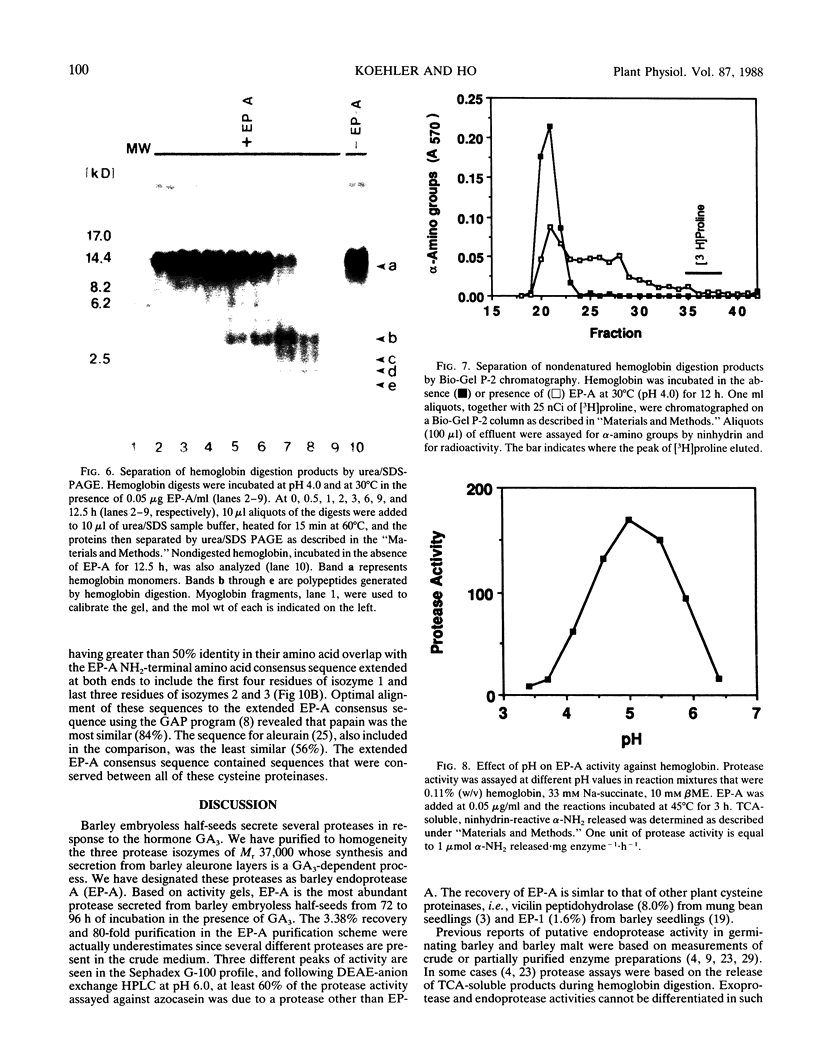
Abstract
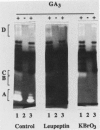
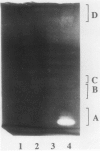
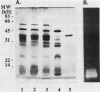
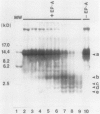
Using in series ammonium sulfate precipitation, gel filtration, and DEAE anion exchange high performance liquid chromatography, we have purified to homogeneity a protease of Mr 37,000 secreted from barley (Hordeum vulgare L. cv Himalaya) embryoless half-seeds. This protease exists in three isozymic forms whose synthesis and secretion from barley aleurone layers was shown to be a gibberellic acid (GA3)-dependent process (R Hammerton, T-HD Ho 1986 Plant Physiol 80: 692-697). This protease constitutes a major portion of the protease activity secreted from half-seeds between 72 to 96 hours of incubation in the presence of GA3 as detected on activity gels containing hemoglobin as the substrate. Analysis of digestion products by urea/sodium dodecyl sulfate-polyacrylamide gel electrophoresis and gel filtration indicated that this protease is an endoprotease, therefore it is designated as barley endoprotease-A (EP-A). Inhibitor studies demonstrated that EP-A belongs to the cysteine class of endoproteases. The optimum pH for EP-A activity was 5.0, and the temperature optimum was 45°C. Comparison of cyanogen bromide generated peptide fragments and NH2-terminal sequence analyses of the three individual EP-A isozymes demonstrates that they are very similar to each other. The NH2-terminal sequence shows extensive sequence homology to the NH2-terminal sequence of papain and several other cysteine proteinases. We also provide evidence that EP-A is not `aleurain,' a putative cysteine proteinase encoded by a GA3-induced barley cDNA clone (JC Rogers, D Dean, GR Heck 1985 Proc Natl Acad Sci USA 82:6512-6516).
Full text
PDF


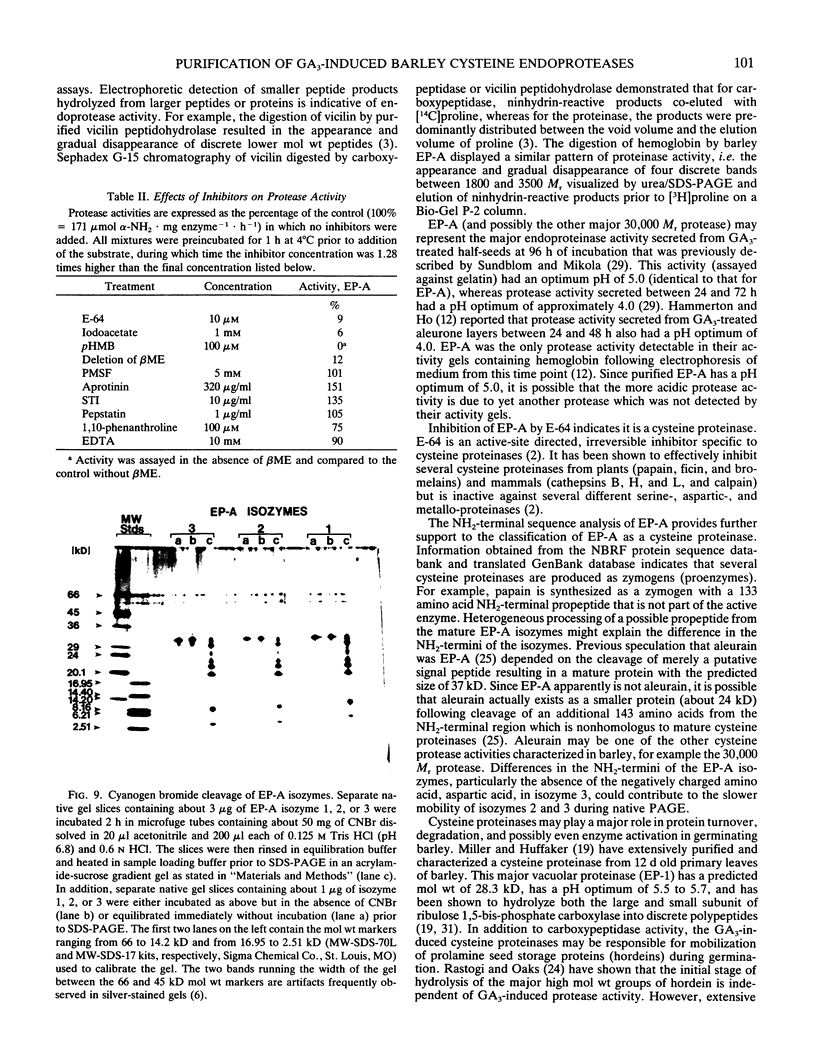


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barrett A. J., Kembhavi A. A., Brown M. A., Kirschke H., Knight C. G., Tamai M., Hanada K. L-trans-Epoxysuccinyl-leucylamido(4-guanidino)butane (E-64) and its analogues as inhibitors of cysteine proteinases including cathepsins B, H and L. Biochem J. 1982 Jan 1;201(1):189–198. doi: 10.1042/bj2010189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumgartner B., Chrispeels M. J. Purification and characterization of vicilin peptidohydrolase, the major endopeptidase in the cotyledons of mung-bean seedlings. Eur J Biochem. 1977 Jul 15;77(2):223–233. doi: 10.1111/j.1432-1033.1977.tb11661.x. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Brown P. H., Ho T. H. Barley aleurone layers secrete a nuclease in response to gibberellic Acid : purification and partial characterization of the associated ribonuclease, deoxyribonuclease, and 3'-nucleotidase activities. Plant Physiol. 1986 Nov;82(3):801–806. doi: 10.1104/pp.82.3.801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chrispeels M. J., Varner J. E. Gibberellic Acid-enhanced synthesis and release of alpha-amylase and ribonuclease by isolated barley and aleurone layers. Plant Physiol. 1967 Mar;42(3):398–406. doi: 10.1104/pp.42.3.398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enari T. M., Mikola J. Peptidases in germinating barley grain: properties, localization and possible functions. Ciba Found Symp. 1977;(50):335–352. [PubMed] [Google Scholar]
- Gribskov M., Burgess R. R. Sigma factors from E. coli, B. subtilis, phage SP01, and phage T4 are homologous proteins. Nucleic Acids Res. 1986 Aug 26;14(16):6745–6763. doi: 10.1093/nar/14.16.6745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammerton R. W., Ho T. H. Hormonal regulation of the development of protease and carboxypeptidase activities in barley aleurone layers. Plant Physiol. 1986 Mar;80(3):692–697. doi: 10.1104/pp.80.3.692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobsen J. V., Varner J. E. Gibberellic Acid-induced synthesis of protease by isolated aleurone layers of barley. Plant Physiol. 1967 Nov;42(11):1596–1600. doi: 10.1104/pp.42.11.1596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Matsudaira P. Sequence from picomole quantities of proteins electroblotted onto polyvinylidene difluoride membranes. J Biol Chem. 1987 Jul 25;262(21):10035–10038. [PubMed] [Google Scholar]
- Miller B. L., Huffaker R. C. Partial purification and characterization of endoproteinases from senescing barley leaves. Plant Physiol. 1981 Oct;68(4):930–936. doi: 10.1104/pp.68.4.930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- Needleman S. B., Wunsch C. D. A general method applicable to the search for similarities in the amino acid sequence of two proteins. J Mol Biol. 1970 Mar;48(3):443–453. doi: 10.1016/0022-2836(70)90057-4. [DOI] [PubMed] [Google Scholar]
- Nikodem V., Fresco J. R. Protein fingerprinting by SDS-gel electrophoresis after partial fragmentation with CNBr. Anal Biochem. 1979 Sep 1;97(2):382–386. doi: 10.1016/0003-2697(79)90089-7. [DOI] [PubMed] [Google Scholar]
- Rastogi V., Oaks A. Hydrolysis of storage proteins in barley endosperms : analysis of soluble products. Plant Physiol. 1986 Jul;81(3):901–906. doi: 10.1104/pp.81.3.901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers J. C., Dean D., Heck G. R. Aleurain: a barley thiol protease closely related to mammalian cathepsin H. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6512–6516. doi: 10.1073/pnas.82.19.6512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stuart I. M., Loi L., Fincher G. B. Development of (1-->3,1-->4)-beta-d-Glucan Endohydrolase Isoenzymes in Isolated Scutella and Aleurone Layers of Barley (Hordeum vulgare). Plant Physiol. 1986 Feb;80(2):310–314. doi: 10.1104/pp.80.2.310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swank R. T., Munkres K. D. Molecular weight analysis of oligopeptides by electrophoresis in polyacrylamide gel with sodium dodecyl sulfate. Anal Biochem. 1971 Feb;39(2):462–477. doi: 10.1016/0003-2697(71)90436-2. [DOI] [PubMed] [Google Scholar]