Abstract
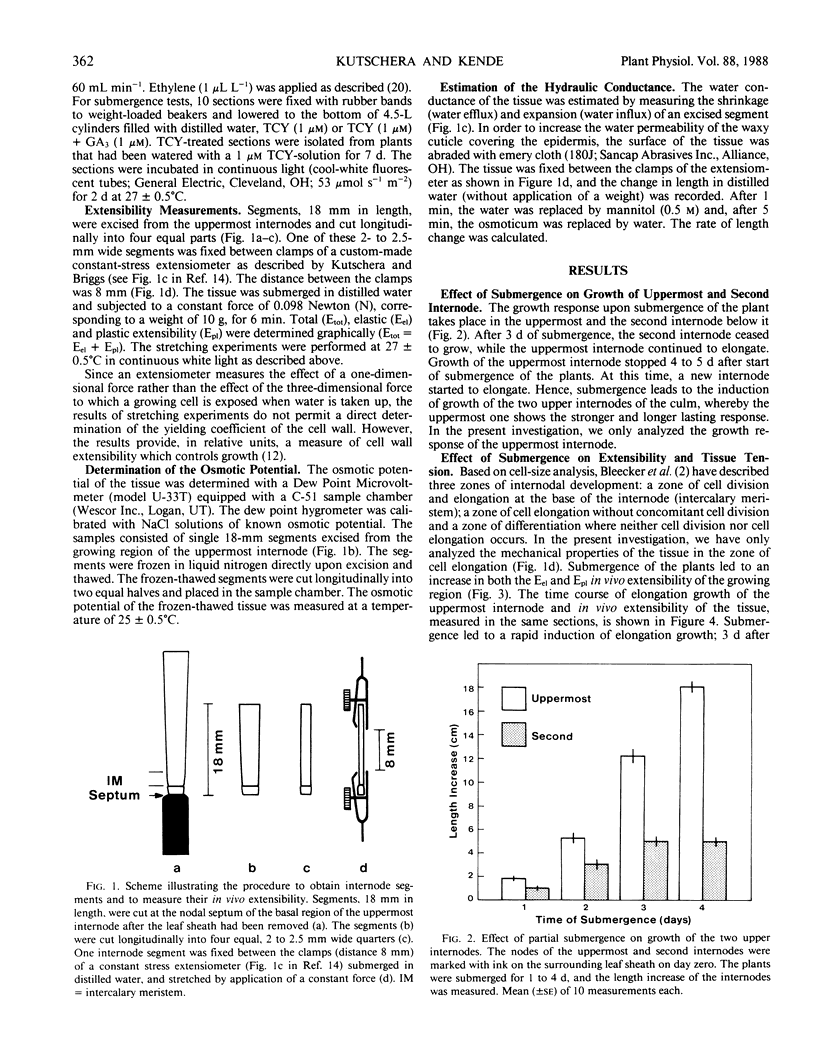
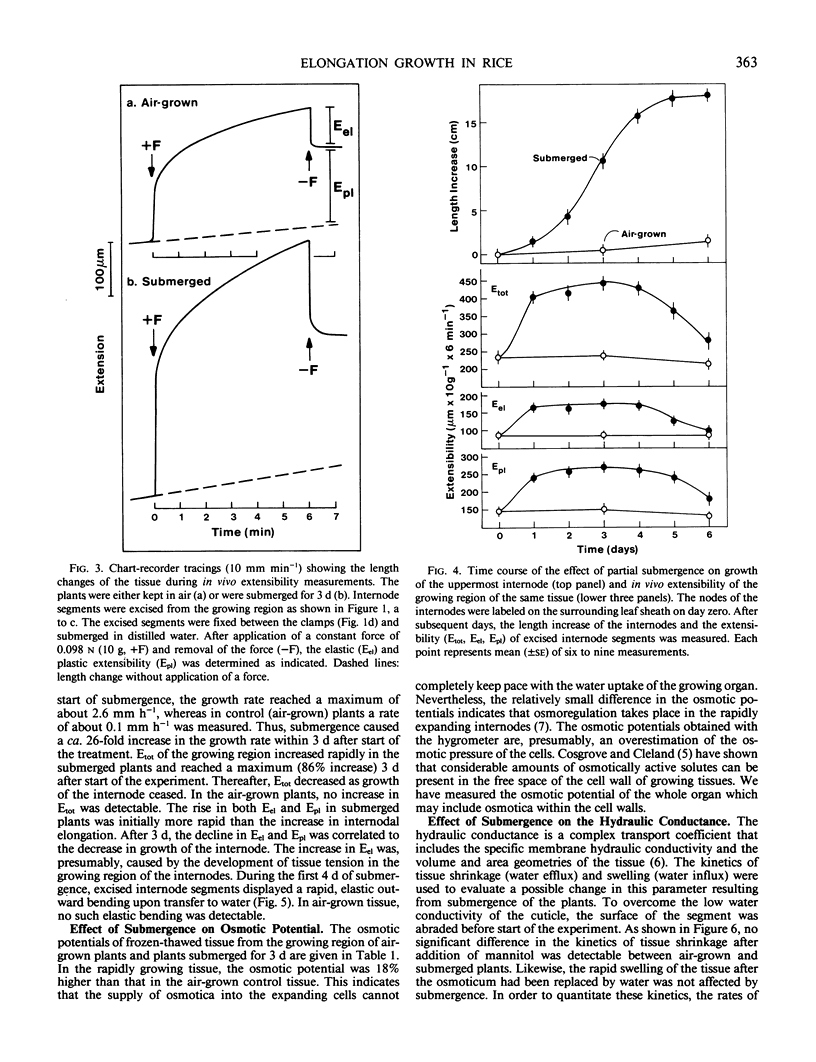
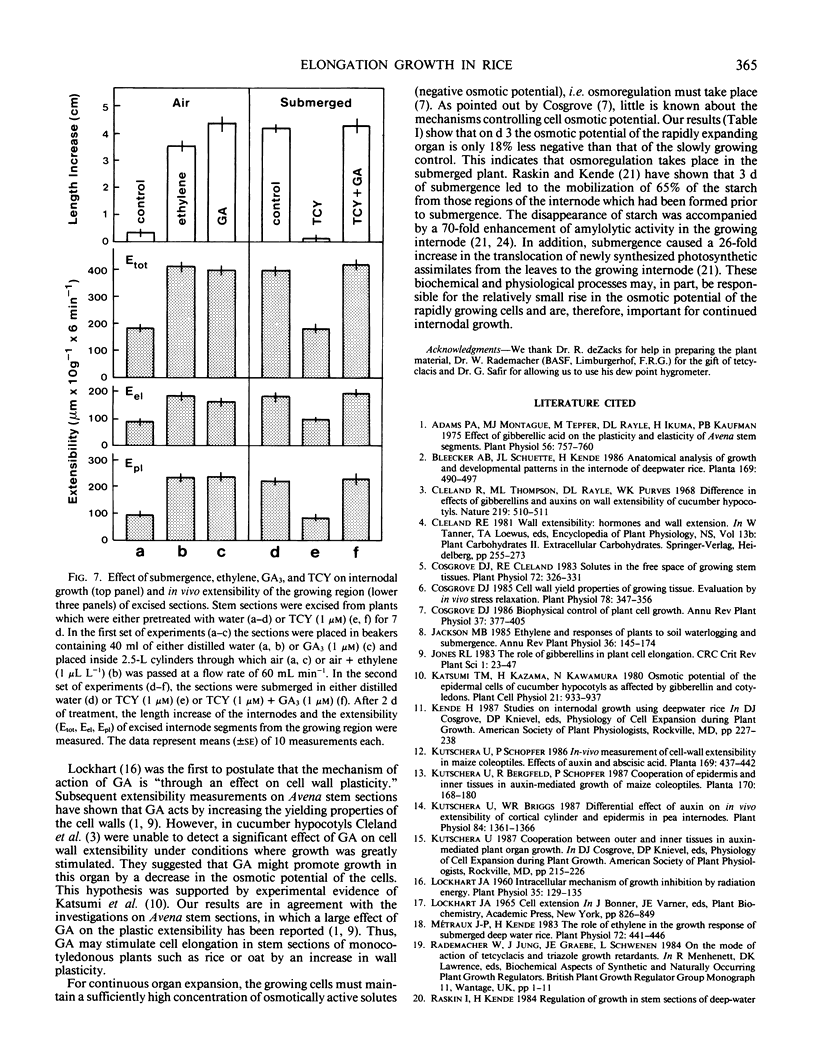
Partial submergence induces rapid internodal elongation in deepwater rice (Oryza sativa L., cv Habiganj Aman II). We measured in vivo extensibility, tissue tension, hydraulic conductance and osmotic potential in the region of cell elongation in the uppermost internode. The in vivo extensibility of the internode, measured by stretching of living tissue with a custom-made constant stress extensiometer, rose rapidly following submergence of the plant. Both the elastic (Eel) and plastic (Epl) extensibility increased when growth of the internode was induced. The submerged internode displayed tissue tension (elastic outward bending of longitudinally split internode sections); in air-grown control internodes, no such bending occurred. The hydraulic conductance, estimated from the kinetics of tissue shrinkage in 0.5 molar mannitol and subsequent swelling in distilled water, was not changed by submergence. The osmotic potential, measured with a dew-point hygrometer using frozen-thawed tissue, was only 18% less negative in the submerged internode than in the air-grown control. This indicates that osmoregulation takes place in rapidly elongating rice internodes. We suggest that the rapid expansion of the newly formed internodal cells of submerged plants is controlled by the yielding properties (Epl) of the cell walls. Experiments with excised stem sections indicate that gibberellin is involved in increasing the Epl of the elongating cell walls.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams P. A., Montague M. J., Tepfer M., Rayle D. L., Ikuma H., Kaufman P. B. Effect of gibberellic Acid on the plasticity and elasticity of Avena stem segments. Plant Physiol. 1975 Dec;56(6):757–760. doi: 10.1104/pp.56.6.757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cleland R., Thompson M. L., Rayle D. L., Purves W. K. Difference in effects of gibberellins and auxins on wall extensibility of cucumber hypocotyls. Nature. 1968 Aug 3;219(5153):510–511. doi: 10.1038/219510a0. [DOI] [PubMed] [Google Scholar]
- Cosgrove D. J. Cell wall yield properties of growing tissue : evaluation by in vivo stress relaxation. Plant Physiol. 1985 Jun;78(2):347–356. doi: 10.1104/pp.78.2.347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosgrove D. J., Cleland R. E. Solutes in the free space of growing stem tissues. Plant Physiol. 1983 Jun;72(2):326–331. doi: 10.1104/pp.72.2.326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cosgrove D. Biophysical control of plant cell growth. Annu Rev Plant Physiol. 1986;37:377–405. doi: 10.1146/annurev.pp.37.060186.002113. [DOI] [PubMed] [Google Scholar]
- Kutschera U., Briggs W. R. Differential effect of auxin on in vivo extensibility of cortical cylinder and epidermis in pea internodes. Plant Physiol. 1987 Aug;84(4):1361–1366. doi: 10.1104/pp.84.4.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lockhart J. A. Intracellular Mechanism of Growth Inhibition by Radiant Energy. Plant Physiol. 1960 Jan;35(1):129–135. doi: 10.1104/pp.35.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Métraux J. P., Kende H. The role of ethylene in the growth response of submerged deep water rice. Plant Physiol. 1983 Jun;72(2):441–446. doi: 10.1104/pp.72.2.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raskin I., Kende H. Role of gibberellin in the growth response of submerged deep water rice. Plant Physiol. 1984 Dec;76(4):947–950. doi: 10.1104/pp.76.4.947. [DOI] [PMC free article] [PubMed] [Google Scholar]