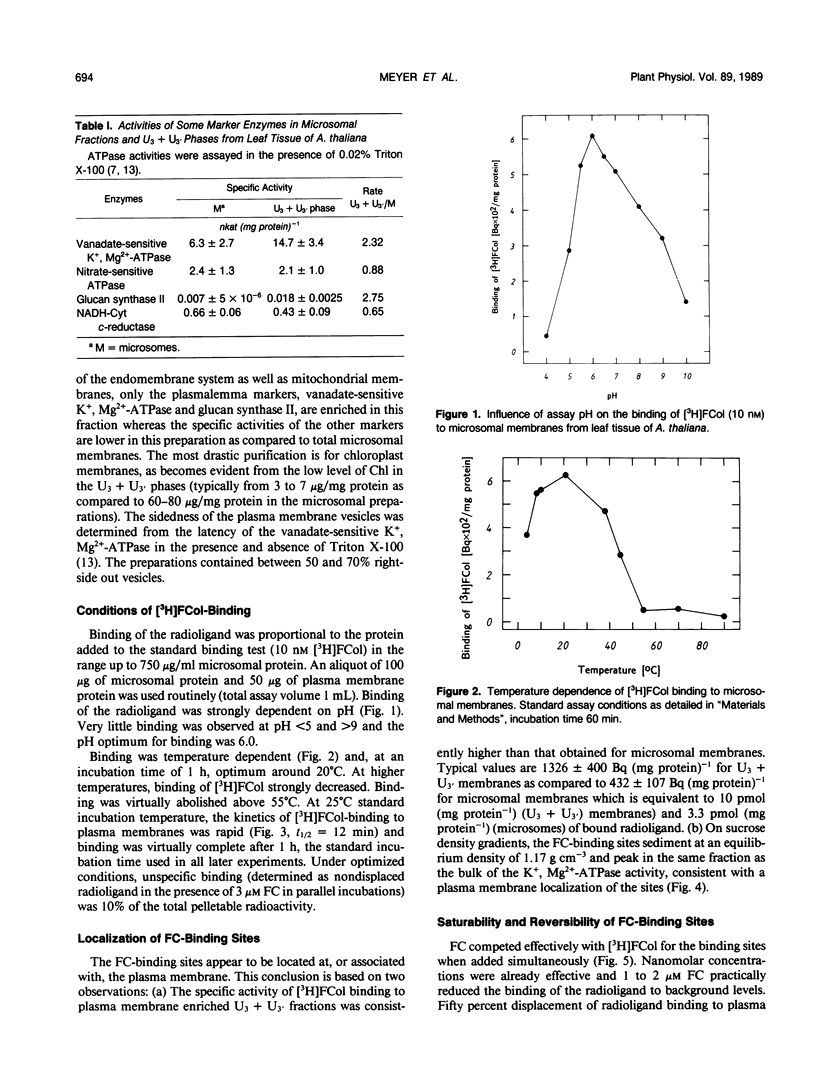
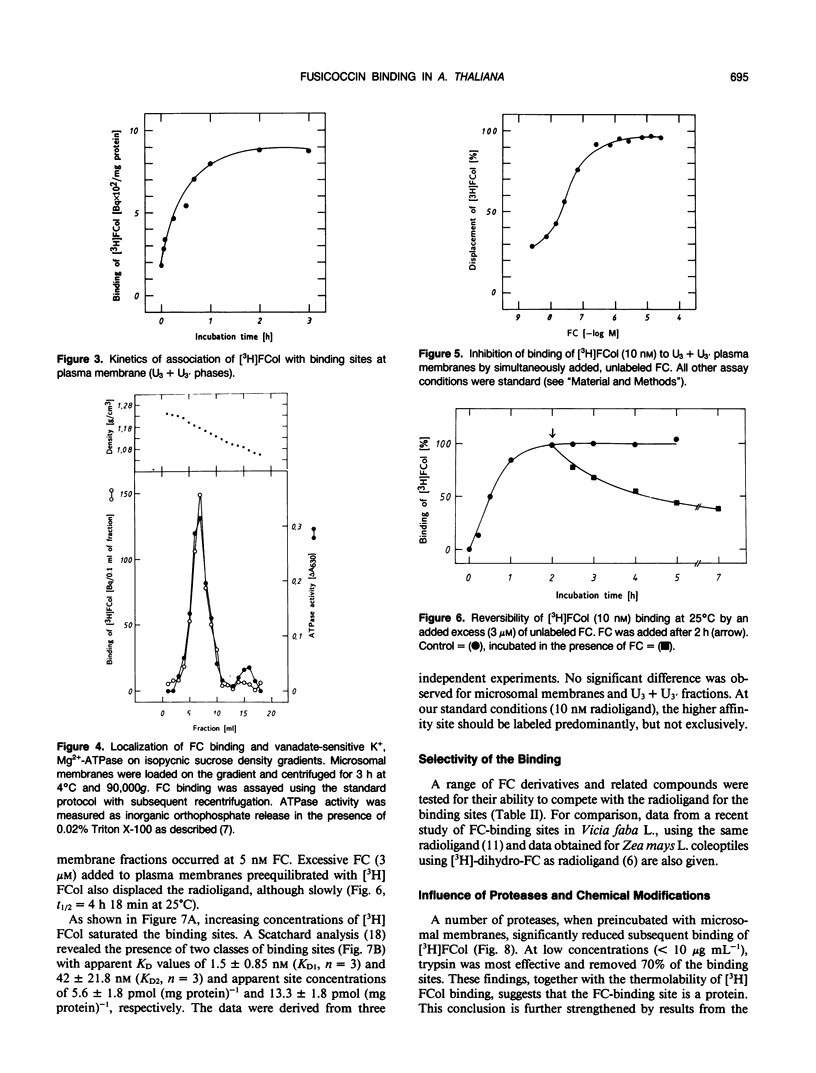
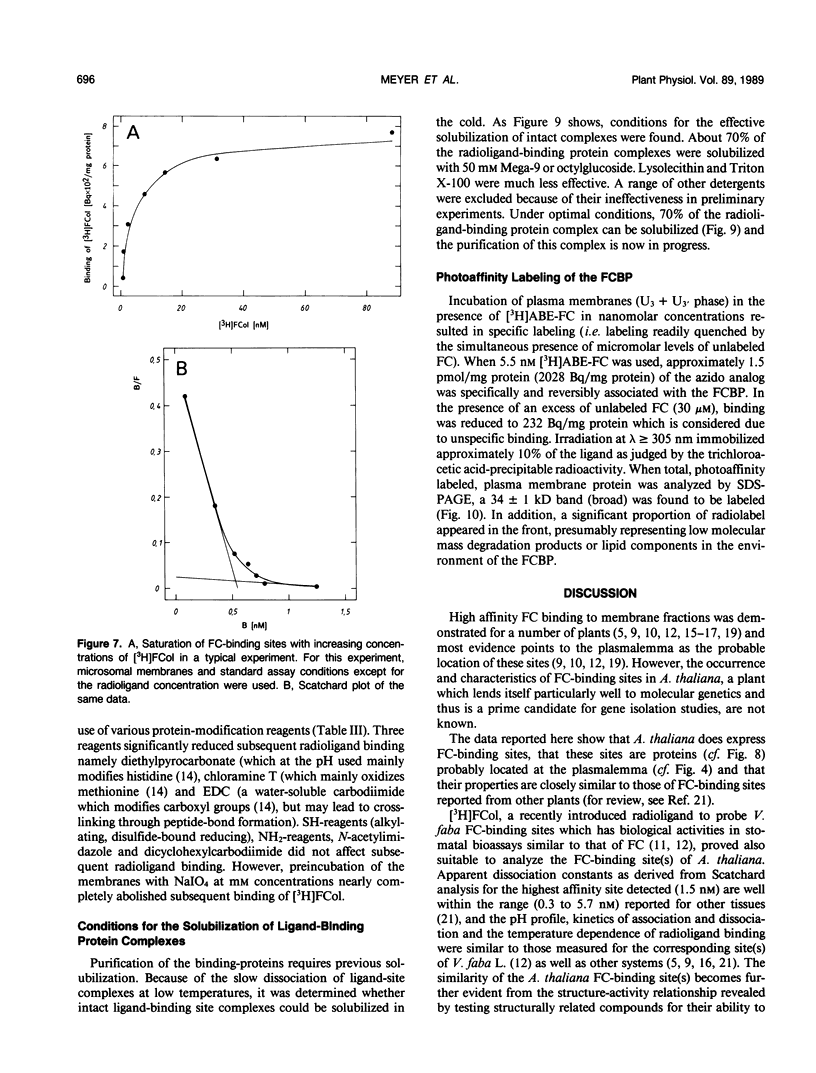
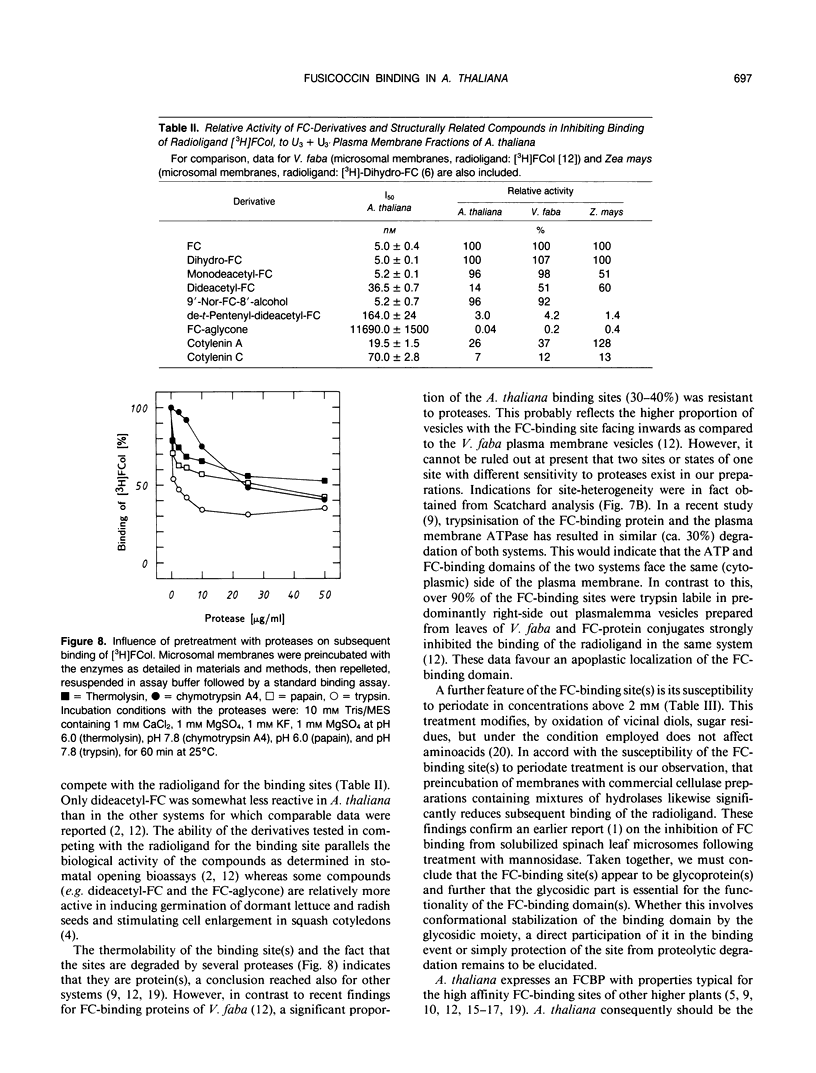
Abstract
Using the novel radioligand, [3H]-9′-nor-fusicoccin-8′-alcohol, high affinity binding sites for fusicoccin were characterized in preparations from leaves of Arabidopsis thaliana (L.) Heynh. The binding site copartitioned with the plasmalemma marker, vanadate-sensitive K+, Mg2+-ATPase, when microsomal fractions were further purified by aqueous two-phase partitioning in polyethylene glycol-dextran phase systems and sedimented at an equilibrium density of 1.17 grams per cubic centimeter in continuous sucrose density gradients, as did the ATPase marker. The binding of [3H]-9′-nor-fusicoccin-8′-alcohol was saturable and Scatchard analysis revealed a biphasic plot with two apparent dissociation constants (KD), KD1 = 1.5 nanomolar and KD2 = 42 nanomolar, for the radioligand. Binding was optimal at pH 6, thermolabile, and was reduced by 70% when the membrane vesicles were pretreated with trypsin. The data are consistent with the presence of one or several binding proteins for fusicoccin at the plasma membrane of A. thaliana. Binding of the radioligand was unaffected by pretreatment of the sites with various alkylating and reducing agents, but was reduced by 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide, diethylpyrocarbonate, chloramine T, and periodate. A number of detergents were tested to find optimum conditions for solubilization. Nonanoyl-N-methylglucamide (50 millimolar) solubilized 70% of the radioligand-binding protein complex in undissociated form. Photoaffinity labeling of membrane preparations with a tritiated azido analog of fusicoccin resulted in the labeling of a 34 ± 1 kilodalton polypeptide. Labeling of this polypeptide, presumably the fusicoccin-binding protein, was severely reduced in the presence of unlabeled fusicoccin.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bruns R. F., Lawson-Wendling K., Pugsley T. A. A rapid filtration assay for soluble receptors using polyethylenimine-treated filters. Anal Biochem. 1983 Jul 1;132(1):74–81. doi: 10.1016/0003-2697(83)90427-x. [DOI] [PubMed] [Google Scholar]
- Feyerabend M., Weiler E. W. Monoclonal antibodies against fusicoccin with binding characteristics similar to the putative fusicoccin receptor of higher plants. Plant Physiol. 1987 Nov;85(3):835–840. doi: 10.1104/pp.85.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stout R. G., Cleland R. E. Partial characterization of fusicoccin binding to receptor sites on oat root membranes. Plant Physiol. 1980 Sep;66(3):353–359. doi: 10.1104/pp.66.3.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Lenten L., Ashwell G. Studies on the chemical and enzymatic modification of glycoproteins. A general method for the tritiation of sialic acid-containing glycoproteins. J Biol Chem. 1971 Mar 25;246(6):1889–1894. [PubMed] [Google Scholar]


