Abstract
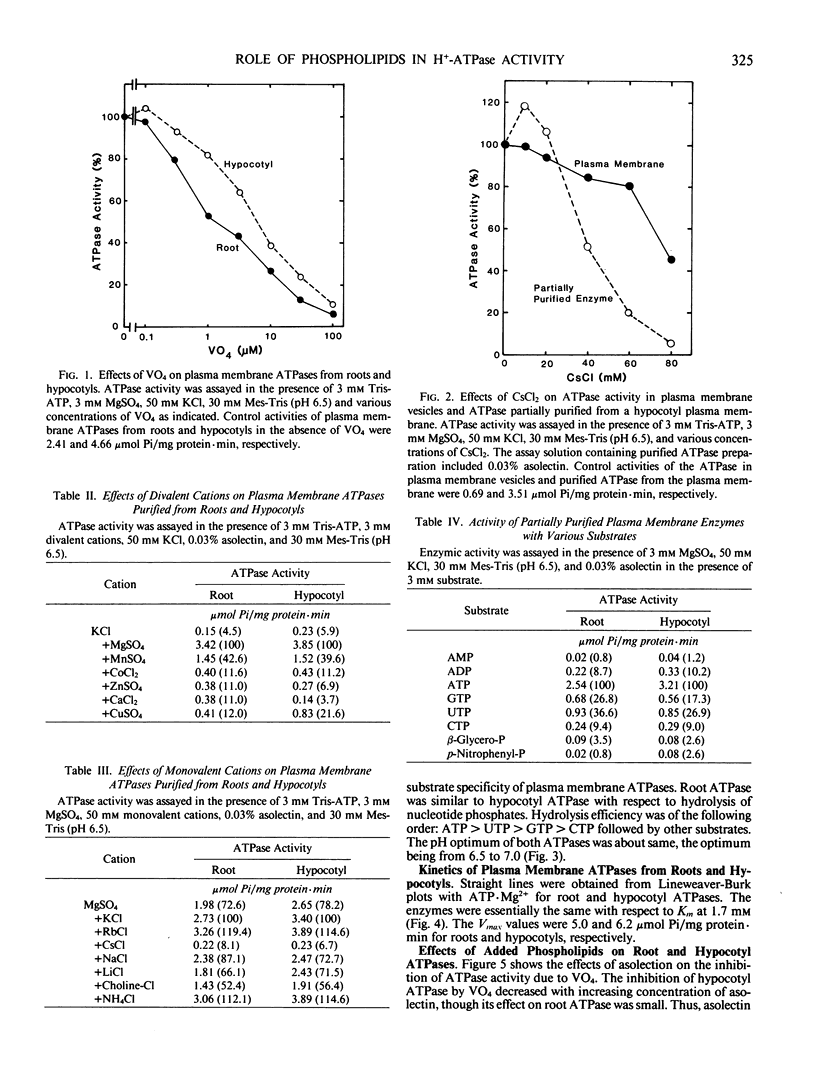
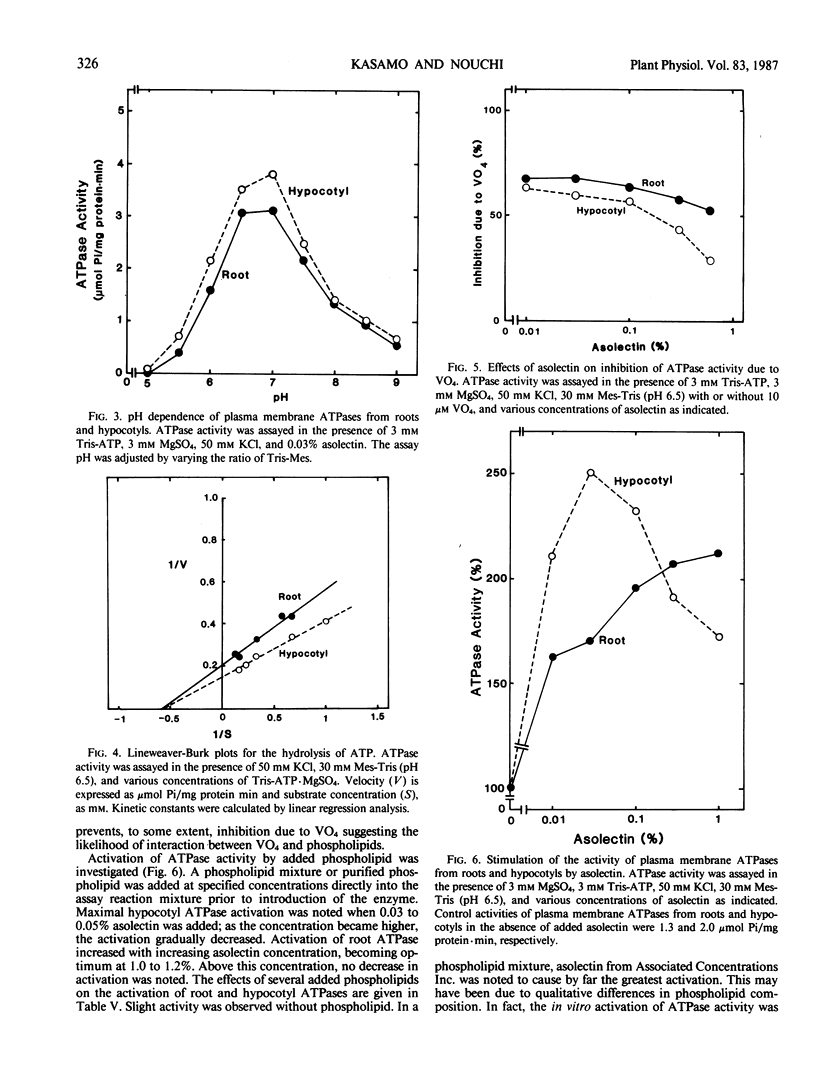
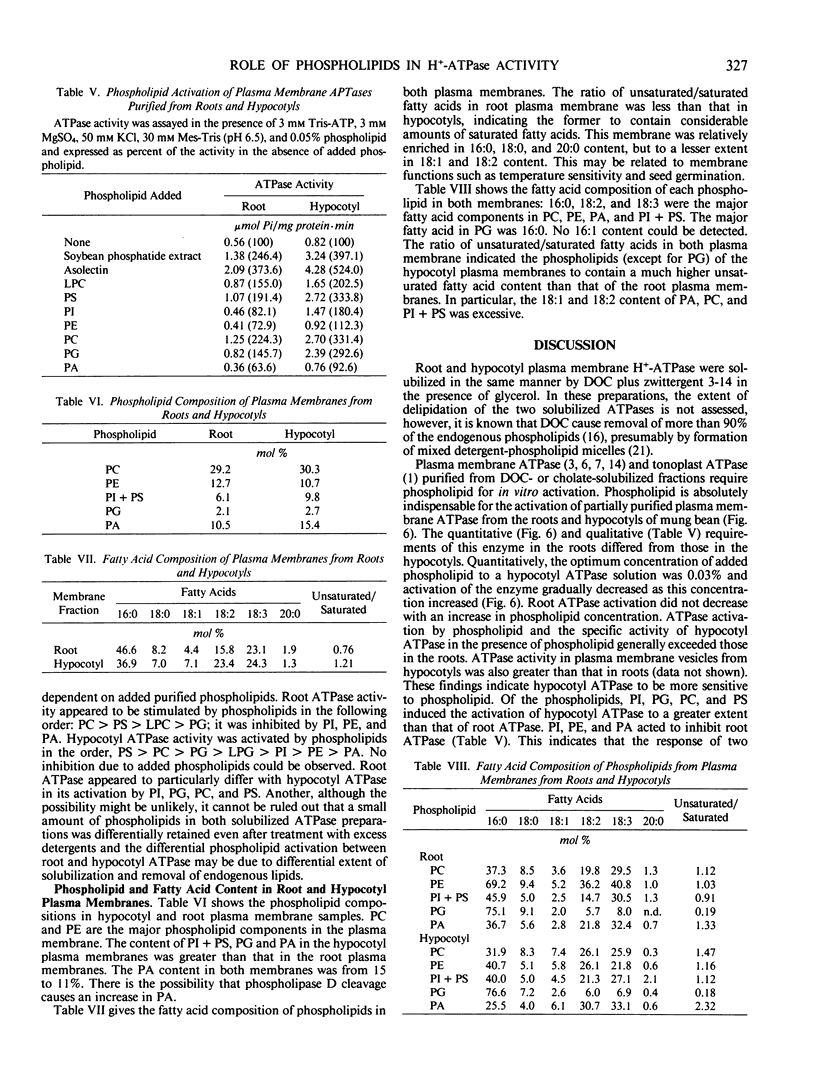
Root and hypocotyl plasma membrane H+-ATPases were partially purified from deoxycholate-solubilized fractions of microsomes in mung bean (Vigna radiata L.) plants in the presence of glycerol. Certain properties of the ATPases and the manner in which phospholipids affect their activity were compared. Root ATPase was similar to hypocotyl ATPase with respect to substrate specificity, salt stimulation, pH dependence, Km for ATP·Mg2+ and inhibitor sensitivity, except for inhibition by vanadate. Both purified ATPases required phospholipids for their activation. Optimum concentrations of exogenously added phospholipid mixture (asolectin) to hypocotyl and root ATPase mixture were 0.03% and 1.0%, respectively. Root ATPase activation did not decrease if more than 1.0% asolectin was added. Qualitatively, phosphatidylserine and phosphatidylcholine brought about greater ATPase activation than other phospholipids. The hypocotyl ATPase was activated by phosphatidylinositol, phosphatidylserine and phosphatidylglycerol to a greater extent than the root ATPase. Root, but not hypocotyl ATPase, was slightly inhibited by the addition of phosphatidylinositol, phosphatidylethanolamine, and phosphatidic acid. The hypocotyl plasma membrane contained phosphatidylinositol + phosphatidylserine, phosphatidylglycerol and phosphatidic acid, and unsaturated fatty acids in greater abundance than the root plasma membrane. The differential activation of the plasma membrane ATPases may arise from these differences.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BLIGH E. G., DYER W. J. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959 Aug;37(8):911–917. doi: 10.1139/o59-099. [DOI] [PubMed] [Google Scholar]
- Bowman B. J., Blasco F., Slayman C. W. Purification and characterization of the plasma membrane ATPase of Neurospora crassa. J Biol Chem. 1981 Dec 10;256(23):12343–12349. [PubMed] [Google Scholar]
- Cantley L. C., Jr, Josephson L., Warner R., Yanagisawa M., Lechene C., Guidotti G. Vanadate is a potent (Na,K)-ATPase inhibitor found in ATP derived from muscle. J Biol Chem. 1977 Nov 10;252(21):7421–7423. [PubMed] [Google Scholar]
- Cocucci M., Ballarin-Denti A. Effect of polar lipids on ATPase activity of membrane preparations from germinating radish seeds. Plant Physiol. 1981 Aug;68(2):377–381. doi: 10.1104/pp.68.2.377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasamo K. Purification and Properties of the Plasma Membrane H-Translocating Adenosine Triphosphatase of Phaseolus mungo L. Roots. Plant Physiol. 1986 Apr;80(4):818–824. doi: 10.1104/pp.80.4.818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimelberg H. K., Papahadjopoulos D. Phospholipid requirements for (Na + + K + )-ATPase activity: head-group specificity and fatty acid fluidity. Biochim Biophys Acta. 1972 Sep 1;282(1):277–292. doi: 10.1016/0005-2736(72)90334-3. [DOI] [PubMed] [Google Scholar]
- Mandala S., Taiz L. Partial purification of a tonoplast ATPase from corn coleoptiles. Plant Physiol. 1985 Jun;78(2):327–333. doi: 10.1104/pp.78.2.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Philippot J. Study of human red blood cell membrane using sodium deoxycholate. I. Mechanism of the solubilization. Biochim Biophys Acta. 1971 Feb 2;225(2):201–213. doi: 10.1016/0005-2736(71)90213-6. [DOI] [PubMed] [Google Scholar]
- Roughan P. G. Phosphatidylglycerol and chilling sensitivity in plants. Plant Physiol. 1985 Mar;77(3):740–746. doi: 10.1104/pp.77.3.740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandermann H., Jr Regulation of membrane enzymes by lipids. Biochim Biophys Acta. 1978 Sep 29;515(3):209–237. doi: 10.1016/0304-4157(78)90015-1. [DOI] [PubMed] [Google Scholar]
- Whitman C. E., Travis R. L. Phospholipid composition of a plasma membrane-enriched fraction from developing soybean roots. Plant Physiol. 1985 Oct;79(2):494–498. doi: 10.1104/pp.79.2.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida S., Kawata T., Uemura M., Niki T. Properties of Plasma Membrane Isolated from Chilling-Sensitive Etiolated Seedlings of Vigna radiata L. Plant Physiol. 1986 Jan;80(1):152–160. doi: 10.1104/pp.80.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida S., Uemura M. Protein and Lipid Compositions of Isolated Plasma Membranes from Orchard Grass (Dactylis glomerata L.) and Changes during Cold Acclimation. Plant Physiol. 1984 May;75(1):31–37. doi: 10.1104/pp.75.1.31. [DOI] [PMC free article] [PubMed] [Google Scholar]


