Abstract
We developed a flow cell apparatus and method for streamlined, real-time measurements of nanopore conductance (G) in response to pH changes. By time-resolving the measurements of interfacial kinetics, we were able to probe nanopore surface coating presence and properties more thoroughly than in our previous work. Nanopores have emerged as a prominent tool for single-molecule sensing, characterization, and sequencing of DNA, proteins, and carbohydrates. Nanopore surface chemistry affects analyte passage, signal characteristics, and sensor lifetime through a range of electrostatic, electrokinetic, and chemical phenomena, and optimizing nanopore surface chemistry has become increasingly important. Our work makes nanopore surface chemistry characterizations more accessible as a complement to routine single-pH conductance measurements used to infer nanopore size. We detail the design and operation of the apparatus and discuss the trends in G and capacitance. Characteristic G vs pH curves matching those obtained in previous work could be obtained with the addition of time-resolved interfacial kinetic information. We characterized native and chemically functionalized (carboxylated) silicon nitride (SiNx) nanopores, illustrating how the method can inform of thin film compositions, interfacial kinetics, and nanoscale chemical phenomena.
INTRODUCTION
Nanopores are a powerful tool to manipulate, sense, and characterize matter.1,2 In materials science, they have been used to fabricate, count, and surface-profile nanoparticles.3–6 In the life sciences, they have been used to detect, characterize, and even sequence a range of biopolymers, including RNA, proteins, carbohydrates, and, most predominantly, DNA.1,7–11 They support fundamental studies in physics, such as measuring the binding strength of biological receptors and electrophoretic forces on single molecules, and support novel applications, such as selective ion filtering.12–15 Fabrication of solid-state nanopores in conventional micro- and nanofabrication-compatible thin films offers numerous advantages over the use of protein nanopores, including size tunability, comparatively simple fabrication, and stability in harsh chemical environments.16–19 Characterization methods, however, must be developed and improved for solid-state nanopores formed in amorphous thin films, such as silicon nitride, because fabrication is not generally reproducible at the atomic scale, and outstanding questions about the stability of chemically functionalized nanopores can be even more complicated to address.1,12,16,20–22
We have used nanopore conductance, G, vs pH to characterize nanopore surface chemistry for a multitude of reasons. First, nanopore conductance at a fixed solution pH is widely used to characterize nanopore diameter, d, with accuracy well-established by direct comparison to results from electron microscopy and related approaches,23–26
| (1) |
where K is the solution conductivity and L is the membrane thickness. These measurements are generally carried out at high ionic strength to suppress the effect of any surface charge on the measurement. The implementation requires nothing more than is required for conventional nanopore resistive-pulse sensing—measurement of the current vs voltage for an ohmic pore in a suitable electrolyte. It thus introduces no additional cost, no additional burden for user expertise, no need for additional instrumentation, and no surface contamination that could occur during electron microscopy. When examining nanopores with organic coatings, in particular, conductance avoids potential film damage or loss by beam-based surface science tools.12,20,27
In the high-ionic-strength limit where Eq. (1) holds, a surface coating of thickness δ will reduce the nanopore diameter by 2δ, thereby causing a reduction in conductance from which the presence of the coating can be inferred.22 Such changes do not inherently reveal the nature of the surface terminal group, however, which is what dominates the behavior of the nanopore–solution interface. Worse, short of that ionic strength limiting behavior, a new surface coating might reduce the conductance by volume exclusion while increasing it by charge density so that nanopore surface chemical modifications may be difficult to detect and characterize by a single conductance measurement. The measurement of G vs pH is designed to circumvent this limitation while also revealing characteristic information about the coating.
Previous work has pointed out that measurements of G at several different solution ionic strengths provided additional nanopore characterization information, such as nanopore shape and surface charge.15,23,25 The key enabling aspect of such work is that, at sufficiently low solution ionic strength, the total nanopore G can contain an appreciable contribution from any nanopore surface conductance, Gsurface. This relationship is shown in Eq. (2), where n is the bulk concentration of ions in solution and Gaccess is the inverse of the pore access resistance,1,2
| (2) |
For nanopores surface-terminated in a (solution-facing) functional group that can undergo pH-dependent (de)protonation, the surface charge, σ, is dictated by the nature of the surface chemistry (pKa) and the solution pH. In such cases, the explicit dependence has been used to assess nanopore surface chemistry, as extensively detailed in earlier published work.20,22 In Fig. 1, we illustrate the key outcomes of the original quasi-steady-state experimental measurements. We recapitulate the findings for four different nanopore surfaces: –COOH, –OH, –NH2-terminated organic coatings on an SiNx nanopore, and a native –OH/—NH2-terminated SiNx surface. For the two acidic surfaces [–COOH and –OH, Figs. 1(a) and 1(b)], there is a plateau at values of the pH where each surface species is expected to be appreciably protonated, thereby contributing nothing to the surface conductance because they are uncharged. The conductance increases as the acids are deprotonated, and this occurs at a lower pH value for the –COOH termination than for the –OH termination because pKa(COOH) < pKa(OH). In contrast, the amine-terminated surface shows an opposite trend, befitting its identity as a base: a plateau at basic pH where it is uncharged, increasing at acidic pH as protonation to –NH3+ increases the surface charge and, thus, surface conductance [Fig. 1(c)]. The amphoteric SiNx surface in Fig. 1(d) requires two pKa values to describe its profile and is marked by a minimum conductance at its isoelectric point (pI ∼ 4.3), where the surface is net uncharged, so Gsurface = 0. The conductance increases when the surface becomes charged (Gsurface > 0) at acidic pH (Si–NH3+) and basic pH (Si–O−).
FIG. 1.
Plots of four different nanopore surfaces characterized by G vs pH. (a) Carboxylic acid-terminated SiNx, (b) hydroxyl-terminated SiNx, (c) amine-terminated SiNx, and (d) native SiNx surfaces. These <10 nm diameter nanopores were profiled in pH ≈ 7, 1.0M KCl solutions, with data taken from Ref. 20.
While providing much-needed insight into nanopore surface chemistry, each curve in Fig. 1 was acquired at a single time-point. In earlier work, Rant and co-workers measured the zeta potential of a nanopore surface over 28 h in response to a change in solution pH.28 While they reported 30 min and ∼18 h response timescales, they chose equilibration times of at least 12 h. Most of their reported changes occurred over 30 min, though. We were interested in a real-time observation of the conductance response and whether, if early-time measurements did not display the characteristic profile, they could still provide useful surface characterization information on a timescale that allows for the method to be used routinely. In this work, we developed an apparatus and method to analyze the time evolution of G in response to pH changes over 1 h of measurement at each pH. Improvements to the experimental tools and implementation support further streamlining and more general use, including changing solution ionic strengths for alternative nanopore characterization methods or establishing salt gradients to enhance analyte detection rates.23,25,29 Having ensured chemical compatibility between the particular reagents and the 3D-printed cell—at levels suitable for single-molecule sensing—one could also use this apparatus for nanopore chemical treatments. In the case of the photohydrosilylation reaction employed here, the flow cell would need to be further developed to allow for suitable optical access to the nanopore membrane.
EXPERIMENTAL
Nanopores were fabricated by controlled (dielectric) breakdown (CBD) in 15 ± 2 nm-thick SiNx membranes in 1M KCl and 10 mM HEPES solutions at pH 7, as described previously.11,16,24 Chemically functionalized nanopores were coated within 30 min of formation by photohydrosilylation with neat four-pentenoic acid, leaving a –COOH surface termination, as described in the supplementary material, Fig. S1, and the preceding text.1,20 Nanopore diameters were calculated using their conductance, G, obtained from I/V measurements in 1 M KCl and 10 mM HEPES solutions at pH 7 and calculated with Eq. (1), unless otherwise stated.24,26 Conventional I/V measurements of the nanopores used in this work are found in the supplementary material, Fig. S11. The design, fabrication, and operation of the nanopore flow cell and fluid handling apparatus are a focus of the present work and are thus described in the main body of the manuscript. More information can be found in the supplementary material, including the supplementary material, Figs. S1–S4.
RESULTS AND DISCUSSION
The G vs pH curves in Fig. 1 were obtained in prior work20 using an open nanopore housing design without flow and with manual exchange of solutions.30 To facilitate the G vs pH characterization workflow, we decided to transition to a nanopore housing that supports flow. We adopted stereolithography (SLA) 3D-printing to fabricate the millifluidic flow cell that is outlined in Fig. 2.24,31,32
FIG. 2.
(a) 3D printed polymethacrylate (PMA) nanopore flow cell. (b) The transparent schematic shows the interior fluid channels and volumes. (1) Twist-fit connection ports, (2) threaded electrode ports, (3) silicon nanopore support wafer, (4) silicone gaskets, (5) alignment guides, (6) flow channels, (7) screw through-holes, (8) nanopore sealing surfaces, (9) nanopore sensing volume, (10) fluid inlet, (11) fluid outlet, and (12) resin drainage hole.
We constructed the flow cell in two halves, drawing upon the sealing surface configuration reported by Waugh and co-workers.24 Each half of the cell had three ports: custom twist-fit connector ports for the fluid inlet and outlet and one threaded port for the electrode. The interior fluid channels were 1.1 mm in diameter, meeting in a 6 mm-long, 3.3 mm-diameter sensing chamber in front of the nanopore. The total fluid volume per half-cell was ∼79 µL.
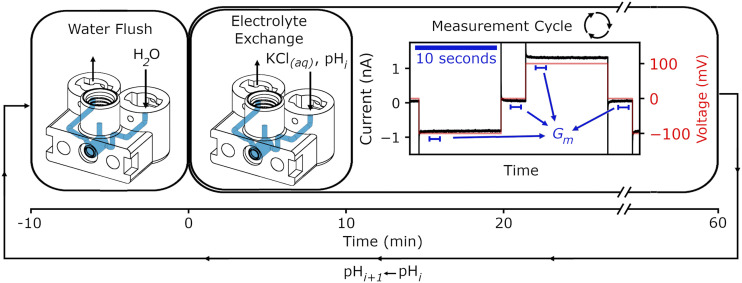
The design of the flow cell was tightly integrated with the design of the fluid handling apparatus and the G vs pH method, with the final result shown in Fig. 3 and Scheme 1. Several key points emerged during the extensive design and testing cycle and are addressed in the final design and operation: bubble management, flow control quality, and electromagnetic interference (EMI) mitigation. Connectors that could be quickly and easily connected and disconnected while offering good sealing reduced measurement time directly and by reducing the likelihood of problems during operation. We designed custom twist-fit connectors (supplementary material, Fig. S3) for this purpose. The connectors were reusable, and their lifetime could be increased by changing the gasket durometer over time to compensate for wear. Commercial connections were all sealed with PTFE tape and/or chlorobutyl/PTFE gaskets. Priming the apparatus was essential. We ran ultra-pure water through the apparatus before the nanopore flow cell was installed. The waste vials were slightly filled to submerge the outlet of the waste tubing in liquid. Without this step, the waste tubing would empty by dripping, causing inconsistencies in the flow rate and current measurements, as shown in the supplementary material, Fig. S5. A visual inspection of the lines was done to check for obvious bubbles, and then the nanopore flow cell was installed. If there were obvious bubbles in the lines, the nanopore flow cell was not connected, and the apparatus was re-primed. After the nanopore flow cell was connected, a test measurement was done, measuring the current with fluid flow. As illustrated in the supplementary material, Fig. S6, the presence of bubbles in the fluid handling apparatus could be detected by significant increases in the current noise. If the apparatus passed the visual inspection but still displayed increased current noise levels, the nanopore flow cell would be disconnected and the solvent introduction process outlined in the supplementary material repeated, along with re-priming the apparatus. Bubble traps provided an in-line, operational buttress against the effects of bubbles. A commercial bubble trap design was modified with rounded edges and reduced internal fluid volumes to minimize the volumes used and the risk of any bubbles being trapped. Finally, all fluid-filled lines were housed within a Faraday cage to shield against electromagnetic interference (EMI). Figure S7 of the supplementary material illustrates how electrolyte-filled fluid lines can couple EMI into the measurement. To isolate components and fluid lines inside the Faraday cage, fluid flow was driven by pressurizing the solvent vials with air from a syringe pump, with only the air-filled portion of tubing outside of the Faraday cage. In the closed system, air was simultaneously withdrawn from the waste vials at the same rate as it was driven into the fluid lines.
FIG. 3.
(a) Diagram of the fluid handling apparatus. The black lines indicate fluid-filled lines, the green lines indicate vacuum or suction lines, and the blue lines indicate pressurized air lines. Four syringes were used in a synchronized push–pull configuration to drive fluid flow. Fluid and air lines to each half of the apparatus were of equal length to ensure synchronized arrival times. (b) Image of apparatus in an open Faraday cage (top-down image). The air-filled drive lines are shown exiting the Faraday cage at the top of the image, while all fluid-filled components are housed within the Faraday cage.
SCHEME 1.
Flow chart of the streamlined nanopore characterization implementation. The flow is driven at 0.5 mL/min through both halves of the flow cell simultaneously (only one half is shown). The current is measured over the last ∼50 min of immersion without flow while the voltage is cycled. This was repeated across a range of pH values. The first second of data from each voltage state was omitted from the conductance analysis because of the capacitive spike. The conductance of the pore for each 26 s voltage cycle was calculated using one second of current data from each voltage state and performing a linear fit. This calculation was repeated for all iterations of the voltage cycle for each pH. These data, in series, then marked the time evolution of the pore conductance at each pH.
In operation, a flow rate of 0.5 mL/min yielded timely changes in pH measured when exchanging between pH 2.5 and pH 9 solutions with the pH electrode flow cell in place of the millifluidic flow cell (supplementary material, Fig. S8). We show in the supplementary material, Fig. S9, that G vs pH profiles were more consistent with established profiles (cf. Fig. 1) when we introduced a 10 min wash of ultra-pure water in between changes in solution pH. We thus adhered to that practice. During the exchange of solution vials, the syringe pump was turned off, the air-filled drive tubing was clamped, and the solution vials were exchanged for the new liquid before turning the pump on and unclamping the tubing. Scheme 1 outlines the measurement: 10 min of rinsing, followed by 10 min of active solution exchange, and then 50 min of measurement in the solution of fixed pH without flow. The conductance for each 26 s voltage cycle was calculated by omitting the first 1 s of data at each voltage to abate the effect of the capacitive spike, averaging for the next full second, and then performing a linear fit. Suitable measurements could be performed either at an acquisition rate of 100 kHz with a 10 kHz low-pass Bessel filter or at 10 kHz filtered at 1 kHz.
We performed G vs pH measurements on two nanopores: a native SiNx nanopore (diameter 16.81 ± 0.04 nm), Fig. 4(a), and a nanopore that had been functionalized with 4-pentenoic acid (diameter 9.49 ± 0.02 nm), Fig. 4(b).20 If the flow based measurement process were inaccurate, the two distinct coatings would allow us to assess if the approach could nevertheless be used to distinguish between surface coatings. This more limited capability would support applications where one needs to simply confirm that the surface chemistry has changed or that a coating is still present. Given the observations of time-resolved nanopore zeta potential by Rant and co-workers, with 30 min and 18 h response times (and a choice of 12 h equilibration times),28 we examined the G vs pH curves at two different time points. Figure 4 shows the G vs pH curves for each nanopore surface averaged across the 10–20 and 50–60 min windows of the hour-long experiments. Measurements of the two surfaces showed good agreement with expected trends at both early and late times. For native SiNx, there was a conductance minimum around pH ∼ 4.3—the isoelectric point of SiNx—with increasing conductance above and below pH ∼ 4.3.20,28,33 For the carboxylic-acid termination, the plateau at low pH is consistent with an uncharged –COOH surface and the increased conductance at more basic pH is consistent with a charged —COO− surface.20 The G vs pH curves could differentiate between the two different coatings. In addition, given the concordance between the results here and earlier results, as illustrated in Fig. 1, they can also be used to confirm or identify the nature of that surface coating. Moreover, these two capabilities are possible even at earlier measurement times.
FIG. 4.
Conductance vs pH plots from measurements averaged over 10-minute windows (red: 10–20 min and black: 50–60 min) for (a) a native SiNx nanopore (16.81 ± 0.04 nm diameter) and (b) a 4-pentenoic acid-functionalized nanopore (9.49 ± 0.02 nm diameter). The characterizations were performed in 0.1 M KCl with an acquisition rate of 10 kHz and a low-pass Bessel filter set to 1 kHz.
Figure 5 shows the evolution of G vs time for pH values selected to probe signal states of native SiNx surface chemistry: the isoelectric point (i.e., a net uncharged surface with a mix of positive and negative surface charges, pI ∼ 4.3), pH > 6 (a negatively charged surface), and pH < 3 (a positively charged SiNx surface but an uncharged COOH surface). For both coatings, the low-pH values plateaued over the 60 min of measurement, while those closer to neutral pH continued to increase, although without changing the overall trend of G vs pH (Fig. 4). Figure S10 of the supplementary material provides G vs time traces for more pH values and shows that the slower time to plateau persists at basic solution pH values. The data in the shaded region in Fig. 5 agree with the global trends but have much larger standard deviations because they were recorded during the exchange of water for electrolyte. We conservatively excluded these data points from the presentation of G vs pH profiles (Fig. 4) to allow for the complete exchange of solutions. Taken together, Figs. 4 and 5 show that this implementation of G vs pH can usefully inform of nanopore surface chemistry in a manner consistent with routine use.
FIG. 5.
Conductance vs time plots at three representative pH values for (a) a native SiNx nanopore (16.81 ± 0.04 nm diameter) and (b) a 4-pentenoic acid functionalized nanopore (9.49 ± 0.02 nm diameter). σ denotes the surface charge. Each data point represents the mean of the conductance values calculated over 10-min windows of each pH measurement. The error bars represent the standard deviation of the conductance calculations over the 10-min windows. The shaded region shows the first ten minutes of the measurement where electrolyte was being flowed to exchange for water. Measurements were carried out in 0.1M KCl with an acquisition rate of 10 kHz and a low-pass Bessel filter set to 1 kHz.
In conventional characterizations of nanopores using responses to voltage changes—I/V curves for size and measurements of G vs pH for surface profiling—the capacitive spike that occurs upon the voltage change is omitted from the analysis. Such spikes can be seen in the current traces in Scheme 1 and the supplementary material, Figs. S5 and S6 and Scheme S1. We were interested in whether or not the nanopore surface chemistry would modify the characteristics of the capacitive decay. Using voltage switches between +100, 0, and −100 mV, we adjusted the amplifier gain to capture more of the capacitive spike magnitude at the cost of resolution of the steady-state current levels (iss) and fit the decays to a single exponential,34
| (3) |
We plotted the time constant, τ, vs pH (Fig. 6 and the supplementary material, Fig. S12). The time constants for positive-going (Fig. 6) and negative-going (the supplementary material, Fig. S12) capacitive spikes averaged over the 10–20 and 50–60 min windows for the carboxylic-acid-terminated pore showed greater disparity at a given pH than for the native SiNx pore. Interestingly, the τ vs pH curves for both the native SiNx and COOH-terminated pores were similar in profile to the G vs pH curve for the native SiNx nanopore. Interpreting this trend in the same manner, by reference to native SiNx surface chemistry, there was a minimum in the time constant magnitude near the SiNx isoelectric point and an increase in magnitude above and below the pI that scaled with the magnitude of the pH-induced surface charge. Such an explanation would suggest that while measurements of the nanopore conductance vs pH can be used to assay the internal nanopore surface chemistry that the hydrosilylation reaction is selective for, the related measurements of the capacitance could be used to assay the outer membrane chemistry and any membrane-specific modifications. Tabard–Cossa and co-workers coated the outer membrane surface of a nanopore with polydimethylsiloxane, without modification of the nanopore interior surface chemistry.30 They showed that this modification affected the capacitive noise during nanopore measurements. Photohydrosilylation chemistry for planar SiNx thin films requires hydrofluoric acid etching to prepare the membrane surface for functionalization.35–37 In the SiNx nanopore context, CBD forms and prepares the necessary SiNx interior pore surface for attachment, such that the outer membrane is not expected to be amenable to hydrosilylation functionalization—that is, it is expected to remain native SiNx. Given the much larger surface area of the nanopore outer membranes compared to the nanopore surface, it is perhaps not surprising that the native SiNx surface chemistry of the outer membrane would dominate the interior functionalized surface chemistry in terms of the capacitive response timescale.
FIG. 6.
Positive-going capacitive decay fit time constant, τ, vs pH for two nanopore surfaces over the 10- to 20-min window (red) and the final 50- to 60-min window (black) across a range of pHs for (a) a native SiNx nanopore and (b) a 4-pentenoic-acid-coated nanopore. Each data point is the average of 10 min of voltage cycles from 0 to +100 mV or 23 positive voltage capacitive spike time constants. The error bars represent the standard deviation over each window. The measurements were performed in 0.1M KCl with an acquisition rate of 10 kHz and a low-pass Bessel filter set to 1 kHz.
CONCLUSION
In summary, a nanopore flow cell, fluid handling apparatus, and method were created to characterize chemically functionalized SiNx nanopores. The measurements substantively replicated those of earlier quasi-steady-state measurements of native and chemically functionalized SiNx nanopores and provided further insights with the added time-resolution capability. Time-resolved measurements of the nanopore conductance showed that, in response to pH, characteristic values of nanopore conductance change on the order of tens of minutes. An analysis was also performed on portions of nanopore data that are commonly not analyzed, such as capacitive spikes. The results suggest that capacitance spike decay times hold information about the composition of the freestanding membrane regardless of the inner nanopore surface composition. To conclude, we have streamlined the operation of an already validated method for characterizing nanopore surface chemistry and explored the underlying equilibration timescales. This will help make the characterization of nanopore surface chemistry more routine, much as the inference of nanopore diameter from nanopore conductance is the prevalent means to quickly characterize nanopore sizes.
SUPPLEMENTARY MATERIAL
See the supplementary material for more detailed explanations of the apparatus fabrication, individual components, method development, and additional data.
ACKNOWLEDGMENTS
The authors would like to thank Dr. James Hagan for his work fabricating and maintaining the nanopore fabrication equipment and his input in developing the reported apparatus and method. This research was supported by NIH Award No. NIH 5R21HG011096 (J.R.D.).
AUTHOR DECLARATIONS
Conflict of Interest
The authors have no conflicts to disclose.
Author Contributions
Brian S. Sheetz: Conceptualization (supporting); Data curation (lead); Formal analysis (lead); Investigation (lead); Methodology (lead); Software (lead); Writing – original draft (supporting); Writing – review & editing (equal). Jason R. Dwyer: Conceptualization (lead); Formal analysis (supporting); Funding acquisition (lead); Investigation (supporting); Methodology (supporting); Supervision (lead); Writing – original draft (lead); Writing – review & editing (equal).
DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1.Hagan J. T., Sheetz B. S., Bandara Y. N. D., Karawdeniya B. I., Morris M. A., Chevalier R. B., and Dwyer J. R., “Chemically tailoring nanopores for single-molecule sensing and glycomics,” Anal. Bioanal. Chem. 412(25), 6639–6654 (2020). 10.1007/s00216-020-02717-2 [DOI] [PubMed] [Google Scholar]
- 2.Karawdeniya B. I., Bandara Y. M. N. D. Y., Nichols J. W., Chevalier R. B., Hagan J. T., and Dwyer J. R., “Challenging nanopores with analyte scope and environment,” J. Anal. Test 3(1), 61–79 (2019). 10.1007/s41664-019-00092-1 [DOI] [Google Scholar]
- 3.Venta K., Wanunu M., and Drndić M., “Electrically controlled nanoparticle synthesis inside nanopores,” Nano Lett. 13(2), 423–429 (2013). 10.1021/nl303576q [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guo P., Huang J., Zhao Y., Martin C. R., Zare R. N., and Moses M. A., “Nanomaterial preparation by extrusion through nanoporous membranes,” Small 14(18), 1703493 (2018). 10.1002/smll.201703493 [DOI] [PubMed] [Google Scholar]
- 5.German S. R., Luo L., White H. S., and Mega T. L., “Controlling nanoparticle dynamics in conical nanopores,” J. Phys. Chem. C 117(1), 703–711 (2013). 10.1021/jp310513v [DOI] [Google Scholar]
- 6.Davenport M., Healy K., Pevarnik M., Teslich N., Cabrini S., Morrison A. P., Siwy Z. S., and Létant S. E., “The role of pore geometry in single nanoparticle detection,” ACS Nano 6(9), 8366–8380 (2012). 10.1021/nn303126n [DOI] [PubMed] [Google Scholar]
- 7.Sohi A. N., Beamish E., Tabard-Cossa V., and Godin M., “DNA capture by nanopore sensors under flow,” Anal. Chem. 92(12), 8108–8116 (2020). 10.1021/acs.analchem.9b05778 [DOI] [PubMed] [Google Scholar]
- 8.Xia K., Hagan J. T., Fu L., Sheetz B. S., Bhattacharya S., Zhang F., Dwyer J. R., and Linhardt R. J., “Synthetic heparan sulfate standards and machine learning facilitate the development of solid-state nanopore analysis,” Proc. Natl. Acad. Sci. U. S. A. 118(11), e2022806118 (2021). 10.1073/pnas.2022806118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hagan J. T., Gonzalez A., Shi Y., Han G. G. D., and Dwyer J. R., “Photoswitchable binary nanopore conductance and selective electronic detection of single biomolecules under wavelength and voltage polarity control,” ACS Nano 16, 5537 (2022). 10.1021/acsnano.1c10039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.O’Donohue M., Saharia J., Bandara N., Alexandrakis G., and Kim M. J., “Use of a solid-state nanopore for profiling the transferrin receptor protein and distinguishing between transferrin receptor and its ligand protein,” Electrophoresis 44, 349 (2022). 10.1002/elps.202200147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Karawdeniya B. I., Bandara Y. M. N. D. Y., Nichols J. W., Chevalier R. B., and Dwyer J. R., “Surveying silicon nitride nanopores for glycomics and heparin quality assurance,” Nat. Commun. 9(1), 3278 (2018). 10.1038/s41467-018-05751-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chou Y. C., Masih Das P., Drndić M., Monos D. S., and Drndić M., “Lifetime and stability of silicon nitride nanopores and nanopore arrays for ionic measurements,” ACS Nano 14(6), 6715–6728 (2020). 10.1021/acsnano.9b09964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lin K., Li Z., Tao Y., Li K., Yang H., Ma J., Li T., Sha J., and Chen Y., “Surface charge density inside a silicon nitride nanopore,” Langmuir 37(35), 10521–10528 (2021). 10.1021/acs.langmuir.1c01504 [DOI] [PubMed] [Google Scholar]
- 14.Van Dorp S., Keyser U. F., Dekker N. H., Dekker C., and Lemay S. G., “Origin of the electrophoretic force on DNA in solid-state nanopores,” Nat. Phys. 5(5), 347–351 (2009). 10.1038/nphys1230 [DOI] [Google Scholar]
- 15.Lin C. Y., Yeh L. H., and Siwy Z. S., “Voltage-induced modulation of ionic concentrations and ion current rectification in mesopores with highly charged pore walls,” J. Phys. Chem. Lett. 9(2), 393–398 (2018). 10.1021/acs.jpclett.7b03099 [DOI] [PubMed] [Google Scholar]
- 16.Kwok H., Briggs K., and Tabard-Cossa V., “Nanopore fabrication by controlled dielectric breakdown,” PLoS One 9(3), e92880 (2014). 10.1371/journal.pone.0092880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Leung C., Briggs K., Laberge M. P., Peng S., Waugh M., and Tabard-Cossa V., “Mechanisms of solid-state nanopore enlargement under electrical stress,” Nanotechnology 31(44), 44LT01 (2020). 10.1088/1361-6528/aba86e [DOI] [PubMed] [Google Scholar]
- 18.Dekker C., “Solid-state nanopores,” Nat. Nanotechnol. 2(4), 209–215 (2007). 10.1038/nnano.2007.27 [DOI] [PubMed] [Google Scholar]
- 19.Fried J. P., Swett J. L., Nadappuram B. P., Mol J. A., Edel J. B., Ivanov A. P., and Yates J. R., “In situ solid-state nanopore fabrication,” Chem. Soc. Rev. 50(8), 4974–4992 (2021). 10.1039/d0cs00924e [DOI] [PubMed] [Google Scholar]
- 20.Bandara Y. M. N. D. Y., Karawdeniya B. I., Hagan J. T., Chevalier R. B., and Dwyer J. R., “Chemically functionalizing controlled dielectric breakdown silicon nitride nanopores by direct photohydrosilylation,” ACS Appl. Mater. Interfaces 11(33), 30411–30420 (2019). 10.1021/acsami.9b08004 [DOI] [PubMed] [Google Scholar]
- 21.Yanagi I., Hamamura H., Akahori R., and Takeda K. I., “Two-step breakdown of a SiN membrane for nanopore fabrication: Formation of thin portion and penetration,” Sci. Rep. 8(1), 10129 (2018). 10.1038/s41598-018-28524-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Frament C. M., Bandara N., and Dwyer J. R., “Nanopore surface coating delivers nanopore size and shape through conductance-based sizing,” ACS Appl. Mater. Interfaces 5(19), 9330–9337 (2013). 10.1021/am4026455 [DOI] [PubMed] [Google Scholar]
- 23.Frament C. M. and Dwyer J. R., “Conductance-based determination of solid-state nanopore size and shape: An exploration of performance limits,” J. Phys. Chem. C 116(44), 23315–23321 (2012). 10.1021/jp305381j [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Waugh M., Briggs K., Gunn D., Gibeault M., King S., Ingram Q., Jimenez A. M., Berryman S., Lomovtsev D., Andrzejewski L., and Tabard-Cossa V., “Solid-state nanopore fabrication by automated controlled breakdown,” Nat. Protoc. 15(1), 122–143 (2020). 10.1038/s41596-019-0255-2 [DOI] [PubMed] [Google Scholar]
- 25.Liebes Y., Drozdov M., Avital Y. Y., Kauffmann Y., Rapaport H., Kaplan W. D., and Ashkenasy N., “Reconstructing solid state nanopore shape from electrical measurements,” Appl. Phys. Lett. 97(22), 223105 (2010). 10.1063/1.3521411 [DOI] [Google Scholar]
- 26.Kowalczyk S. W., Grosberg A. Y., Rabin Y., and Dekker C., “Modeling the conductance and DNA blockade of solid-state nanopores,” Nanotechnology 22(31), 315101 (2011). 10.1088/0957-4484/22/31/315101 [DOI] [PubMed] [Google Scholar]
- 27.Xia Z., Scott A., Keneipp R., Chen J., Niedzwiecki D. J., Dipaolo B., and Drndić M., “Silicon nitride nanopores formed by simple chemical etching: DNA translocations and TEM imaging,” ACS Nano 16, 18648 (2022). 10.1021/acsnano.2c07240 [DOI] [PubMed] [Google Scholar]
- 28.Firnkes M., Pedone D., Knezevic J., Döblinger M., and Rant U., “Electrically facilitated translocations of proteins through silicon nitride nanopores: Conjoint and competitive action of diffusion, electrophoresis, and electroosmosis,” Nano Lett. 10(6), 2162–2167 (2010). 10.1021/nl100861c [DOI] [PubMed] [Google Scholar]
- 29.Wanunu M., Morrison W., Rabin Y., Grosberg A. Y., and Meller A., “Electrostatic focusing of unlabelled DNA into nanoscale pores using a salt gradient,” Nat. Nanotechnol. 5, 160–165 (2010). 10.1038/nnano.2009.379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tabard-Cossa V., Trivedi D., Wiggin M., Jetha N. N., and Marziali A., “Noise analysis and reduction in solid-state nanopores,” Nanotechnology 18(30), 305505 (2007). 10.1088/0957-4484/18/30/305505 [DOI] [Google Scholar]
- 31.Gross B., Lockwood S. Y., and Spence D. M., “Recent advances in analytical chemistry by 3D printing,” Anal. Chem. 89(1), 57–70 (2017). 10.1021/acs.analchem.6b04344 [DOI] [PubMed] [Google Scholar]
- 32.Baden T., Chagas A. M., Gage G., Marzullo T., Prieto-Godino L. L., and Euler T., “Open labware: 3-D printing your own lab equipment,” PLoS Biol. 13(3), e1002112 (2015). 10.1371/journal.pbio.1002086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hoogerheide D. P., Garaj S., and Golovchenko J. A., “Probing surface charge fluctuations with solid-state nanopores,” Phys. Rev. Lett. 102(25), 256804 (2009). 10.1103/PhysRevLett.102.256804 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Smeets R. M. M., Keyser U. F., Dekker N. H., and Dekker C., “Noise in solid-state nanopores,” Proc. Natl. Acad. Sci. U. S. A. 105(2), 417–421 (2008). 10.1073/pnas.0705349105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sieval A. B., Demirel A. L., Nissink J. W. M., Linford M. R., Van Der Maas J. H., De Jeu W. H., Zuilhof H., and Sudhölter E. J. R., “Highly stable Si−C linked functionalized monolayers on the silicon (100) surface,” Langmuir 14(7), 1759–1768 (1998). 10.1021/la971139z [DOI] [Google Scholar]
- 36.Sieval A. B., Vleeming V., Zuilhof H., and Sudhölter E. J. R., “An improved method for the preparation of organic monolayers of 1-alkenes on hydrogen-terminated silicon surfaces,” Langmuir 15(23), 8288–8291 (1999). 10.1021/la9904962 [DOI] [Google Scholar]
- 37.Rosso M., Giesbers M., Arafat A., Schroën K., and Zuilhof H., “Covalently attached organic monolayers on SiC and SixN4 surfaces: Formation using UV light at room temperature,” Langmuir 25(4), 2172–2180 (2009). 10.1021/la803094y [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
See the supplementary material for more detailed explanations of the apparatus fabrication, individual components, method development, and additional data.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.