Abstract
Hyperoside is a flavonol glycoside mainly found in plants of the genera Hypericum and Crataegus, and also detected in many plant species such as Abelmoschus manihot, Ribes nigrum, Rosa rugosa, Agrostis stolonifera, Apocynum venetum and Nelumbo nucifera. This compound exhibits a multitude of biological functions including anti-inflammatory, antidepressant, antioxidative, vascular protective effects and neuroprotective effects, etc. This review summarizes the quantification, original plant, chemical structure and property, structure–activity relationship, pharmacologic effect, pharmacokinetics, toxicity and clinical application of hyperoside, which will be significant for the exploitation for new drug and full utilization of this compound.
Keywords: hyperoside, original plant, chemical structure, pharmacology, pharmacokinetics, toxicity
Introduction
Humans have used plants as the basis of traditional medical system for many years.1 However, even with the rise of modern medicine, traditional medicine is being drawn upon. As the discovery and synthesis of traditional chemical drugs are facing great obstacles and challenges, more and more researchers have turned their attention to the application of natural drugs and their extracts.2,3 As the source of natural drugs often comes from plants, animals or other natural products,4,5 they always show a superiority in side effects, metabolic burden and other aspects compared with traditional chemical synthetic drugs.6,7 The research boom gained momentum after Tu Youyou was awarded the Nobel Prize in 2015. Different from the traditional chemical drugs, natural drugs and their extracts have been considered to be good resources for new drugs, especially in plants. Because of the wide variety of plants and extracts they have become the main resources of natural drugs.
With the further study of natural medicinal chemistry, the active components in plants and their natural extracts have been isolated and analyzed.8,9 After screening several dozen commonly-used natural herbs, many compounds from plant extracts are reported, which efficiently exhibited anti-inflammatory responses.10–14
While the flavonoids are one of the most important natural compounds including flavanones, isoflavones, etc. possess a multitude of biological effects including anti-bacteria, anti-inflammatory, anti-allergy, anti-oxidation,15,16 cytoprotective, anti-thrombotic and anti-platelet.17,18
With the development of natural medicinal chemistry, many natural compounds or drugs derived from natural compounds have used in clinic.19,20 For instance, paclitaxel has been used to treat ovarian, breast, and lung cancer,21 artemisinin has been used to treat malaria,22 and silibinin from Silybum marianum has been applied to treat hepatitis,23 etc.
Quercetin-3-O-β-D-galactopyranoside, known as hyperoside, is a flavonol glycoside mainly found in plants of the genera Hypericum and Crataegus,24 and also detected in many plant species such as Abelmoschus manihot, Ribes nigrum, Rosa rugosa, Agrostis stolonifera, Apocynum venetum and Nelumbo nucifera. This compound exhibits a multitude of biological functions including as an anti-inflammatory,25 antidepressant,26 antioxidative,27 a vascular protector,28 and neuroprotector.29
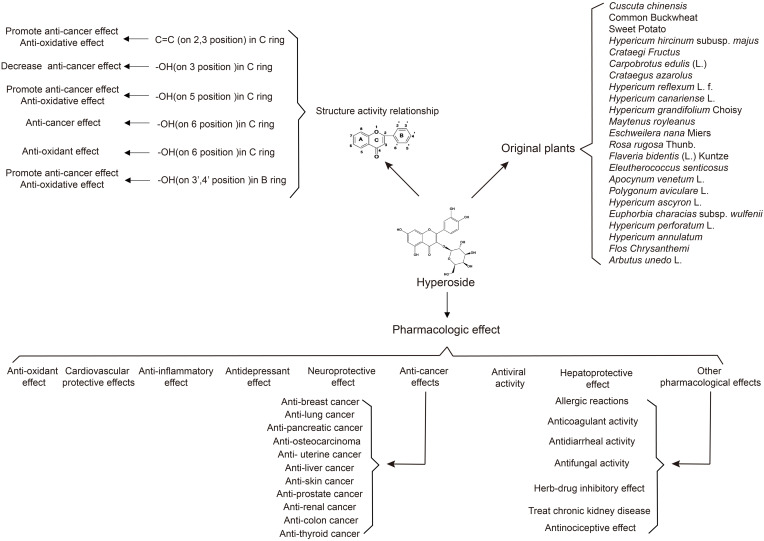
In this review we summarize the advancements of hyperoside from six aspects including quantification and original plant, chemical structure and property, structure–activity relationship, pharmacologic effect, pharmacokinetics, toxicity and clinical application, which will be significant for the exploitation for new drug and full utilization of this compound (Figure 1). Additionally, possible tendency and perspective for future investigations of hyperoside is also discussed in this review.
Figure 1.
The outline of review.
Extraction and Original Plant
Since 1960, hyperoside has been isolated from red osier dogwood (Cornus stolonifera Michx.). And with the further study of natural medicinal chemistry, many natural drugs have been found to contain flavonoids and hyperoside is one of the most important components. With the development of chromatographic analysis methods including column chromatography, high performance counter-current chromatography, principal component analysis, high performance liquid chromatography (HPLC) has been widely used in quantification of hyperoside. At present HPLC is also the most commonly used extraction and identification method recorded in Chinese Pharmacopoeia.
There are a variety of herbal plants have been applied in the extraction of hyperoside like Crataegus pinnatifida Bunge (hawthorn), Fagopyrum tataricum (L.) Gaertn. (Common buckwheat), etc. And hyperoside could also extracted from different parts of plants. For example, the leaf of Crataegus pinnatifida Bunge (Hawthorn), Crataegus azarolus L., Maytenus royleana Cufod., Eschweilera nana Miers, Flaveria bidentis (L.) Kuntze and Arbutus unedo L. The seed of Cuscuta chinensis var. chinensis, Hypericum perforatum L. and Hypericum annulatum Moris. The flower or blossom of Hypericum reflexum L.f., Hypericum canariense L. and Hypericum grandifolium Choisy. The fruit or hull of Fagopyrum tataricum (L.) Gaertn. (Common Buckwheat), Crataegus pinnatifida Bunge (Hawthorn), Carpobrotus edulis (L.) N.E.Br. The root of Dioscorea esculenta (Lour.) Burkill (Sweet Potato), Eleutherococcus senticosus Maxim. And the plant or the aerial part of Hypericum hircinum subsp. majus (Aiton) N.Robson, Polygonum aviculare L., Hypericum ascyron L., Euphorbia characias subsp. wulfenii (Hoppe ex W.D.J.Koch) Radcl.-Sm. and Hypericum perforatum L. In this section, we summarized the isolated original plants in Table 1.
Table 1.
Original Plants and Parts
| Num | Plants | Parts | Reference |
|---|---|---|---|
| 1 | Cuscuta chinensis var. chinensis | Seeds | [30,31] |
| 2 | Fagopyrum tataricum (L.) Gaertn. (Common Buckwheat) | Hull | [32] |
| 3 | Dioscorea esculenta (Lour.) Burkill (Sweet Potato) | Storage root | [33] |
| 4 | Hypericum hircinum subsp. majus (Aiton) N.Robson | Plants | [34] |
| 5 | Crataegus pinnatifida Bunge (Hawthorn) | Fruit Leaf |
[35,36] |
| 6 | Carpobrotus edulis (L.) N.E.Br. | Fruit | [37] |
| 7 | Crataegus azarolus L. | Leaf | [38] |
| 8 | Hypericum reflexum L.f. | Blossom | [39] |
| 9 | Hypericum canariense L. | ||
| 10 | Hypericum grandifolium Choisy | ||
| 11 | Maytenus royleana Cufod. | Leaf | [40] |
| 12 | Eschweilera nana Miers | Leaf | [41] |
| 13 | Rosa rugosa Thunb. (Family Rosaceae) | Flower | [42] |
| 14 | Flaveria bidentis (L.) Kuntze | Leaf | [43] |
| 15 | Eleutherococcus senticosus Maxim. | Root | [44] |
| 16 | Apocynum venetum L. | Leaf | [44] |
| 17 | Polygonum aviculare L. | Aerial part | [45] |
| 18 | Hypericum ascyron L. | Aerial part | [46] |
| 19 | Euphorbia characias subsp. wulfenii (Hoppe ex W.D.J.Koch) Radcl.-Sm. | Aerial part | [47] |
| 20 | Hypericum perforatum L. (St. John’swort) | Seed Aerial part |
[48,49] |
| 21 | Hypericum annulatum Moris | Seed | [48] |
| 22 | Chrysanthemum morifolium Ramat. | Flower | [50] |
| 23 | Arbutus unedo L. | Leaf | [51] |
Chemical Structure and Property
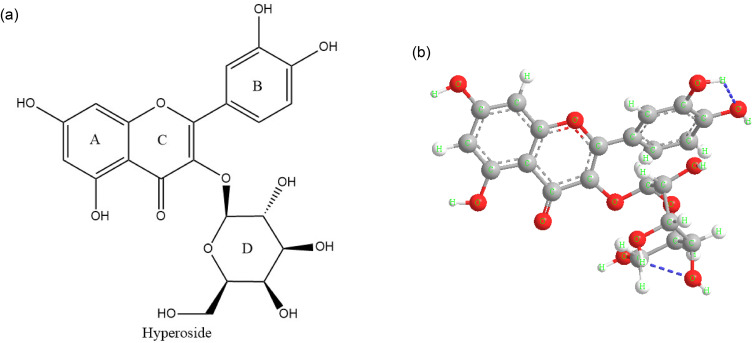
Hyperoside also called quercetin-3-O-β-D-galactoside pyranoside, belongs to flavonol glycosides. It’s composed of two phenyl rings (A and B rings), a six-membered oxygen heterocycle (C ring) and a galactopyranosid (D ring) (Figure 2a). With the deepening of research, Liu et al predicted the most stable conformation according to the minimum energy principle which is shown in Figure 2b, by calculating the Self-Consistent Field (SCF) energy level of twenty different chair and boat conformations of galactopyranosid (D ring).52
Figure 2.
Structure and structure–activity of hyperoside. Chemical structural formula of hyperoside (a), Spatial structure diagram of hyperoside (b).
It has been widely reported that hyperoside has a variety of bioactive functions, such as anti-bacteria, anti-inflammatory, anti-allergy, anti-oxidation, etc. The molecule of hyperoside contains lots of polar groups, including three ether bonds, one carbonyl group and eight hydroxyl groups. Based on its chemical structure these groups make hyperoside has high activity and could interact with multiple functional monomers.
Structure–Activity Relationship
Hyperoside as a kind of compound of flavonoids, due to its structure and properties exhibit some biological activity. Hyperoside could exhibit a variety of pharmacologic effects, based on its structure especially the substitutional groups.
The C2-3 double bond in C ring promotes the anti-cancer effects. And when there is a hydroxyl substituent(-OH) on the third carbon, the anti-cancer effects may be decreased.53 As aglycone of hyperoside is quercetin, they may have similar properties. And 5-OH also showed the same effect.53 On the contrary, 6-OH showed an enhancement on the anti-cancer effects, when the compound contains 3’,4’-OH the anti-cancer effect will be increased.54 Another factor that influences the anti-cancer effect is the differences of substituent.55
Meanwhile, hyperoside also has anti-oxidant effect, and the position of -OH is one of the most important factors,56 while hyperoside contains a 3’,4’-o-diphenol hydroxyl structure on B ring demonstrating the strong anti-oxidant effect. In addition, 5-OH and 7-OH also make hyperoside shows a strong anti-oxidant effect.57 And the double bond between C-2,3 has also been proved to be relative to the anti-oxidant.58
Pharmacologic Effect
Hyperoside is a biological active compound with great application prospect and there have been some clinical applications because of its effects in anti-inflammatory, anti-oxidative, anti-depressant and vascular protective effects and so on.59 So, in this part we reviewed the pharmacologic effects of hyperoside (Table 2, Figures 3 and 4).
Table 2.
Pharmacologic Effects
| Num | Classification | Materials | Dose | Mechanism | Reference |
|---|---|---|---|---|---|
| 1 | Anti-oxidant | Saccharomyces Cerevisiae | 5, 20 mg/L | Decreased LPO and the level of ROS | [60] |
| 2 | Human Spermatozoa | - | Decreased the level of LPO | [61] | |
| 3 | PC12 cell | 100 μg/mL | Inhibition the shrinking and apoptosis induced by H2O2 and TBHP | [62] | |
| 160 μg/mL | |||||
| 4 | ECV304 cells | 128 μM | Increased SIRT1, inhibition the translocation of Bax | [63] | |
| 5 | B16 melanoma cells | 18.2 μM | Inhibition the expression of tyrosinase and oxidative stress-induced melanogenesis | [64] | |
| 6 | B16 cells | 5, 10, 50 μM | Against RS, • O2, NO •, ONOO −, enhance the GSH/GSSG ratio, inhibited oxidative stress-induced melanogenesis | [65] | |
| 7 | MT3C3-E1 cells | 5, 10, 50 μmol/L | Decreased the expression of p-JNK and p-p38, regulated the MAPK-mediated responses | [66] | |
| 8 | SH-SY5Y Cells | 0.25, 0.5, 1, 2 μM | Upregulation of Nrf2 and HO-1, activation of Nrf2/HO-1 signaling | [67] | |
| 9 | L02 cell | 10, 50, 100, 200, 500, 800 μM | Upregulation of HO-1 via the regulation of MAPK-dependent Keap1–Nrf2–ARE signaling pathway | [68] | |
| 10 | Female Wistar rats | - | Increased transcription of CAT and GSH | [69] | |
| 11 | V79-4 cells | 5 μM | Downregulation of ROS, induction of catalase and glutathione peroxidase activities | [70] | |
| 12 | Cardiovascular protective effect | H9C2 cells | 1, 10, 50, 100 μM | Upregulated miR-138, inhibited the expression of MLK3 and Lnc2 to inhibit apoptosis | [71] |
| Male C57BL/6 mice | 10, 50, 100, 200 mg/kg | ||||
| 13 | RAW 264.7 cells | 10, 30, 100 μM | Inhibited the iNOS protein expression | [72] | |
| 14 | HUVECs | 10, 20, 50 μM | Reduced IL-1β, IL-8, TF, ICAM1, VCAM1, activated autophagy and suppressed mTOR/S6K and TLR/Myd88/NF-κB signaling | [73] | |
| 15 | HUVECs | 5, 10, 25 μM | Increased Bcl-2, decreased Bax, induced phosphorylation of ERK1/2 | [74] | |
| 16 | HUVECs, mice | 5, 10, 20, 50 μM | Blocked HG-induced vascular inflammation via inhibition of NF-κB | [75] | |
| 17 | HUVECs, mice | 5, 10, 20, 50 μM | Inhibition of the HMGB1 signaling pathway | [76] | |
| 18 | VSMCs, male C57BL/6N mice | 5 μM in vitro 40 mg/kg in vivo |
Upregulation of Nur77, inhibited both cell proliferation of VSMCs in vitro and carotid artery ligation-induced neointimal formation in vivo | [28] | |
| 19 | VSMCs | 10, 25, 50, 100 μg/mL | Inhibited oxLDL-induced LOX1 expression, ERK activation and cell proliferation via the regulation of oxLDL-LOX1-ERK pathway | [77] | |
| 20 | Male SD rats | 1–100 μM | Endothelium-dependent and endothelium-independent mechanisms | [78] | |
| 21 | Male adult SD rats | 50 μM | Activation of ERK-dependent signal pathway | [79] | |
| 22 | Male KM mice | 9, 18, 36 mg/kg | Upregulated autophagy, suppressed NLRP1 inflammation pathway | [80] | |
| 23 | NRCMs, Male C57/BL6 mice | 1, 5, 10 μM | Inhibition of the AKT signal pathway | [81] | |
| 24 | H9C2 cells, Adult male Wistar rats | 10 μM 100, 200 mg/kg |
Attenuating myocardial apoptosis and inducing autophagy | [82] | |
| 25 | Anti-inflammation | HT22 cells | 20 μM | Alleviates the level of IL-1β, IL-6, IL-8, TNF-α, ROS, MDA, Bax, and caspase-3; increases the expression of CAT, SOD, GSH, Bcl-2, BDNF, TrkB, and NGF. Alleviated LPS-induced inflammation, oxidative stress, and apoptosis by upregulating SIRT1 to activate Wnt/β-catenin and sonic hedgehog pathways. | [83] |
| 26 | Rat peritoneal macrophages | 100 μM | Inhibited NO production via inhibition of expression of iNOS by attenuation of p44/p42 MAPK, p38 MAPK and JNK | [84] | |
| 27 | Chondrocytes, Male C57BL/6 mice | 10, 20, 40 μM; 20 mg/kg | Suppression of PI3K/AKT/NF-κB and MAPK pathways Enhanced Nrf2/HO-1 |
[85] | |
| 28 | HK-2 cells | 10, 50, 100 μM | Activation of miR-499a-5p/NRIP1 signal pathway | [86] | |
| 29 | Male SD rats, BMSCs | 40 μg/mL | Activation of NF-κB pathway to proliferation and osteogenic differentiation | [87] | |
| 30 | Mouse peritoneal macrophages | 5 μM | Inhibition of TNF-α, IL-6, NO production, NF-κB activation | [25] | |
| 31 | Antidepressant effect | C6 glioblastoma cells | 1 μM | Reduced β2-adrenergic sensitivity | [88] |
| 32 | C6 glioblastoma cells | 1 μM | Reduction in β2-AR density in plasma membrane and decrease in corresponding downstream signaling | [89] | |
| 33 | PC12 cells | 2.5, 5, 10 μg/mL | Decreased Ca2+, upregulation of CREB and BNDF through the cAMP-CREB signal pathway | [90] | |
| 34 | Male Albino Swiss mice | 0.94 mg/kg, 3.75 mg/kg | Mediated by monoaminergic system and the upregulation of BDNF level | [91] | |
| 35 | Adult male CF1 mice, Male Wistar rats | 10, 20, 40 mg/kg(i.p.) or 20 and 40 mg/kg(P.O.) in mice, 1.8 mg/kg/day (P.O.) in rats | D2-like receptor activation | [26] | |
| 36 | Male ICR mice | 10, 20, 30 mg/kg | Be related to the serotoninergic system including 5-HT2A, 5-HT2 receptor | [92] | |
| 37 | Adult male SD rats | 5, 10, 15 mg/kg | Regulation on hypothalamus- pituitary- adrenal (HPA) axis activity | [93] | |
| 38 | ICR mice | - | Promoted the level of NE and 5-HT in brain and decreased the activity of MAO | [94] | |
| 39 | Neuroprotective effect | Primary cortical neuron | 2.5, 5, 10, 20 μM | Inhibited PI3K/Akt/Bad/Bclxl-regulated mitochondrial apoptotic pathway | [95] |
| 40 | Primary cortical neuron | 10 μM | Inhibited the activation of NF-κB, lessened the expression of iNOS, ameliorated ERK, JNK and Bcl-2 family-related apoptotic signaling pathway | [29] | |
| 41 | PC12 cells | 10, 50, 100 μM | Inhibited ROS, the activation of caspase-3 and PARP to inhibit apoptosis | [96] | |
| 42 | ICR mice | 2.5 mg/kg | Inhibited AchE activity | [97] | |
| 43 | SD rats | 25, 50, 100 mg/kg | Upregulated the level of H2S, decreased the synthesis and release of NO | [98] | |
| 44 | C57BL /6J mice (CSE+/+, CSE−/−) | 25, 50, 100 mg/kg | Upregulation of cystathionine γ-lyase (CES)-H2S signal pathway | [99] | |
| 45 | SD rats | 1, 10, 100 μM | Upregulation of H2S, activation of KCa and opening KCa channels, blocked Ca2+ influx | [100] | |
| 46 | Male SD rats | 10, 20, 40 mg/kg | Promote the anti-oxidant activity of SOD and CAT to alleviate oxidative stress injury | [101] | |
| 47 | Anti-breast cancer | 4T1 cells, MCF-7 cells, Balb/c mice | 25, 50, 100 μM in vitro 50 mg/kg in vivo |
Inhibition of NF-κB signal pathway, activation of Bax-caspase 3 axis to promote ROS induced apoptosis | [102] |
| 48 | MDA-MB-231 cells | 5, 10, 50, 100 μg/mL | Blocking TLR4-mediated pro-survival and inflammatory cytokine expression to promote the sensitivity of breast cancer cells to paclitaxel | [103] | |
| 49 | Anti-lung cancer | A549 cells, Balb/c-nude mice | 15, 20, 25 μM in vitro 15, 20, 25 mg/kg in vivo |
Activation of caspase-3 to motivate apoptosis and inactivation of NF-κB to inhibit inflammatory | [104] |
| 50 | A549 cells | 10, 50, 100 μM | Upregulation of AMPK signal pathway and HO-1 expression to suppressed the survival and proliferation of A549 cells | [105] | |
| 51 | A549 cells | 10, 50, 100 μM | Upregulated apoptosis via activation of the p38 MAPK- and JNK-induced mitochondrial death pathway | [106] | |
| 52 | A549 cells, H466 cells, C57BL/6J mice | – | Upregulated the expression of p38 MAPK, caspase 3, caspase 9, cleaved caspase 3, cleaved caspase 9 and Bax, downregulated the expression of Cu/Zn SOD, CAT, Nrf2, NQO1, HO-1 and Bcl-2 | [107] | |
| 53 | A549 cells | 10, 20, 50, 100, 200, 400 μg/mL | Inhibited the process of G1/S phase to inhibit proliferation | [108] | |
| 54 | NCI-H1975 cells, PC-9 cells, Nude male mice | 30, 60, 90, 120, 150 μM in vitro, 25 mg/kg in vivo | Upregulation of FoxO1 via CCAT1 to inhibit proliferation and induce apoptosis | [109] | |
| 55 | A549 cells | 0.5, 1, 2 mM | Induced autophagy through inhibiting the Akt/mTOR/p70S6K signal pathway, induced apoptosis | [110] | |
| 56 | H1975 cells, A549 cells, male Balb/c nude mice | 20, 40, 60, 80, 100 μg/mL | Inhibited NF-κB transcriptional activity, caspase-9/caspase 3 activation, cell cycle arrest and suppression of cell proliferation | [111] | |
| 57 | A549 cells | 1, 2, 5 μM | Upregulation of Akt/PI3K and p38 MAPK signal pathway, downregulation of nm23-H1, MTA1, TIMP-2 and MMP-2 | [112] | |
| 58 | Anti-pancreatic cancer | PANC-1 cells, BxPC-3 cells, Female nude Balb/c mice | 100, 300, 500 μM in vitro 20 mg/kg in vivo |
Promote Bax/Bcl-2 and Bax/Bcl-xL ratios and inhibit NF-κB activation, promoted of apoptosis and suppressed of proliferation | [113] |
| 59 | Anti-osteocarcinoma | ARP cells, H929 cells MC3T3-E1 cells, Raw264.7 cells | 0.05, 5 μM | Promoted osteoblastogenesis and suppressed osteoclastogenesis, thus improving the bone marrow microenvironment to inhibit MM cell proliferation | [114] |
| 60 | U2OS cells, MG63 cells | 150 μg/mL | Activation of TGF-β signal pathway to induce G0/G1 arrest to inhibit cell proliferation | [115] | |
| 61 | Anti-uterine cancer | Hela cells, C33-A cells |
0.25, 0.5, 1, 2, 4, 8 μM | Downregulation of C-MYC gene expression | [116] |
| 62 | RL952 cells | 10, 20, 100 μM | Induced apoptosis through mitochondria-dependent and death receptor-dependent apoptotic pathways | [117] | |
| 63 | SKOV-3 cells, HO-8901 cells |
50, 100 μM | Overexpression of PGRMC1 enhanced the autophagy and apoptosis induced by hyperoside | [118] | |
| 64 | Anti-liver cancer | HepG2 cells | 10, 20, 50 nM | Upregulation of p53/caspase signal pathway to induce apoptosis and inhibit proliferation | [119] |
| 65 | HepG2 cells | 5, 10, 20, 40, 80 μM | Inhibiting the BMP-7 dependent-PI3K/AKT pathway to suppress cell proliferation | [120] | |
| 66 | CBRH-7919 cells, Balb/c nude mice | 1, 1.25, 5, 20, 60 μg/mL in vitro, 6 mg/kg in vivo | Upregulation of caspase 3 and caspase 9 induced apoptosis to inhibit liver cancer in vitro and vivo | [59] | |
| 67 | Anti-skin cancer | A431 cells, A432 cells, HS-4 cells, female mice | 1, 5, 10, 25, 50, 100 μM in vitro, 8.16 μM in vivo | Inhibition of PI3K/Akt/mTOR/p38MAPK signal pathway and activation of AMPK to inhibit cell proliferation and induced apoptosis and autophagy | [121] |
| 68 | Anti-prostate cancer | PC3 cells | - | Reduced the expression of miR-21 to inhibit cell growth and metastasis | [122] |
| 70 | Anti-renal cancer | 786-O cells | - | Downregulation of ROS to induce caspase-3 cleavage and PARP cleavage, decreased miR-27A and induced ZBTB10 and downregulated Sp1, Sp3 and Sp4 | [123] |
| 71 | Anti-colon cancer | SW620 human colorectal cancer cells | 12.5, 25, 50 μM | Upregulation of p21, p53 to induce G2/M phase arrest and apoptosis | [124] |
| 72 | HT-29 human colon cancer cells | 100, 200 μM | Activation of mitochondria-dependent apoptotic pathway | [125] | |
| 73 | Anti-thyroid cancer | SW579 cells | 5, 10, 20 μg/mL | Upregulated the expression of Fas and FasL mRNAs, downregulated the expression of the survivin protein | [126] |
| 74 | Antiviral effect | HepG2.2.15 cells, Peking ducklings | 0.2, 0.1, 0.05, 0.025, 0.0125 g/L in vitro 0.02, 0.05, 0.1 g/kg/d in vivo |
Hyperoside showed an anti-HBV effect both in vitro and in vivo | [127] |
| 75 | HepG2.2.15 cells | 50 μg/mL | Hyperoside showed an anti-HBV effect both in vitro | [128] | |
| 76 | Huh-7 cells | - | Hyperoside from Nymphaea alba extracts showed anti-HCV effect | [129] | |
| 77 | Hepatoprotective effect | L02 cells, C57BL/6 mice | 5, 10, 20 μM in vitro 25, 50, 100 mg/kg in vivo |
Activated Nrf2 to downregulate LDH and ALT | [130] |
| 78 | Male Balb/c mice, L02 cells | 50, 100 mg/kg in vivo 50, 100, 200 μM in vitro |
Activated ERK1/2-Crm1 pathway to promote the nuclear export of Bach 1 to upregulate Nrf2 binding to ARE | [131] | |
| 79 | Male ICR mice | 100 mg/kg | Upregulation of HO-1 and Nrf2, downregulation of iNOS, COX2, TNF-α | [132] | |
| 80 | Female Wistar rats | - | Increased transcription of CAT and GSH to show an antioxidant effect | [69] | |
| 81 | L02 cells, Male SD rats | 100 μM in vitro 15, 30, 60 mg/kg in vivo |
Reduced PHLPP2 expression, activated AKT phosphorylation, induced GSK-3β phosphorylation, increased Nrf2 translocation, promoted HO-1 expression | [133] | |
| 82 | C57BL/6 mice, Nr4A1 knockout mice | 50 mg/kg | Upregulation of Nr4A1 expression to macrophage polarization and HFD-induced NAFLD progression | [134] | |
| 83 | Male Wistar rats | 100, 200 mg/kg | Inhibiting the activation of TGF-β1/Smad pathway | [135] | |
| 84 | Allergic reactions | HMC-1 cells | 100, 160 μg/mL | Inhibited the level of TSLP by downregulation of intracellular calcium/RIP2/caspase 1/NF-κB signal | [136] |
| 85 | RBL-2H3 cells | 0.35 μM | Hyperoside could covalently modify with Bovine β-Lactoglobulin | [137] | |
| 86 | Anticoagulant activity | HUVECs ICR mice |
0.5, 1, 2, 5, 10, 20, 50 μM in vitro, 2.3 mg/kg in vivo | Prolonged APTT and PT and inhibited the activities of thrombin and FXa, inhibited production of thrombin and FXa, inhibited TNF-α induced production of PAI-1 | [138] |
| 87 | Herb-drug inhibitory effect | Human liver microsomes | 0.5, 1, 2, 5 μM | Selectively inhibited CYP2D6 | [139] |
| 88 | Treat chronic kidney disease | SD rats | - | Reduction in CaOx formation | [140] |
| 89 | SD rats Male SDT rats |
2.5, 5, 10 μg/mL | Inhibited the interaction between PDGF-BB and PDGFR-β | [141] | |
| 90 | SD male rats NRK-52E cells |
20 mg/kg/d in vivo 5, 10, 15 μg/mL |
Inhibited AMPK-ULK1 signaling-mediated autophagy to attenuate renal aging and injury | [142] |
Abbreviations: AchE, anti-acetyl-cholinesterase; Akt, Akt kinase; AMPK, adenosine monophosphate-activated protein kinase; APTT, activated partial thromboplastin time; ARE, antioxidant response element; BDNF, brain-derived neurotrophic factor; BMP-7, bone morphogenetic protein-7; CAT, catalase; CREB, cAMP response element binding protein; COX2, cyclooxygenase 2; ERK, extra cellular signal-regulated protein kinase; FoxO1, fork head box protein O1; GSH, reduced glutathione; GSK-3β, glycogen synthase kinase 3β; GSSG, Glutathione Oxidized; HBV, hepatitis B virus; HCV, hepatitis C virus; HFD, high-fat diet; HO-1, heme oxygenase-1; ICAM1, intercellular cell adhesion molecule-1; iNOS, inducible nitric oxide synthase; JNK, c-Jun N-terminal kinase; Keap1, kelch-like ECH-associated protein 1; LDH, lactate dehydrogenase; LPO, lipid peroxidation; LPS, lipopolysaccharide; MAO, monoamine oxidase; MTA1, Metastasis Associated 1; MAPK, mitogen-activated proteinkinase; MDA, malondialdehyde; MMP-2, matrix metalloproteinase 2; mTOR, mammalian target of rapamycin; NAFLD, non-alcoholic fatty liver disease; NGF, nerve growth factor; Nrf2, nuclear erythroid 2-related factor 2; NF-κB, nuclear factor kappa-B; NO, nitric oxide; NO•, nitric oxide radical; NQO1, NAD(P)H quinone dehydrogenase 1; Nr4A1, nuclear receptor subfamily 4 group A member 1; •O2, superoxide radical; ONOO−, peroxynitrite anion; PAI-1, Plasminogen activator inhibitor 1; PARP, poly-ADP-ribose-polymerase; PHLPP2, PH domain leucine-rich repeat protein phosphatase 2; PI3K, phosphatidylinositol 3-kinase; p-JNK, phosphorylated c-Jun N-terminal kinase; PGRMC1, progesterone receptor membrane component 1; PT, prothrombin time; RIP2, receptor-interacting protein 2; RS, reactive species; ROS, reactive oxygen species; PDGF-BB, platelet-derived growth factor-BB; PDGFR-β, platelet-derived growth factor-B receptor; SD, Sprague-Dawley; SIRT1, silent mating type information regulation 2 homolog-1; SOD, superoxide dismutase; TBHP, tert-butyl hydroperoxide; TNF-α, tumor necrosis factor-α; TIMP-2, Tissue inhibitors metalloproteinases; TrkB, Tyrosine Kinase receptor B; TGF-β, transforming growth factor beta; TLR4, toll-like receptor-4; TSLP, thymic stromal lymphopoietin; ULK1, unc-51-like kinase 1; VCAM1, vascular cell adhesion molecule-1.
Figure 3.
The possible molecular mechanisms of hyperoside in anti-oxidant effect, anti-inflammatory effect, cardiovascular protective effects, anti-depressant effect, neuroprotective effect and hepatoprotective effect.
Figure 4.
The possible molecular mechanisms of hyperoside in anti-cancer (breast, lung, pancreatic, skin, thyroid, renal, colon, liver, prostate and uterine).
Anti-Oxidant Effect
Anti-oxidant effect is an essential effect of hyperoside. Several studies demonstrated that hyperoside showed a strong activity of anti-oxidant.97,143–146 In a model of oxidative damage induced by hydrogen peroxide (H2O2), carbon tetrachloride and cadmium in Saccharomyces cerevisiae demonstrated that hyperoside could significantly increase cell viability, decrease the lipid peroxidation (LPO) and intracellular reactive oxygen species (ROS) levels.60 In human spermatozoa LPO model induced by H2O2, C. sativa leaf extract containing high concentration hyperoside showed great antioxidant effect by reducing LPO level. And hyperoside showed a protective effect on damage induced by LPO particularly at the plasma membrane level.61 In the tert-butyl hydroperoxide (TBHP) induced model, hyperoside could protect PC12 and ECV-304 cells against cytotoxicity induced by TBHP.62,63 In B16F10 melanoma cells (B16 cells), hyperoside was reported to suppress oxidative stress-induced melanogenesis.64,65
Radical scavenging activity and enhancement of antioxidant enzyme play an important role in the cellular and molecular mechanisms of hyperoside’s antioxidant effect. In Qi et al research that hyperoside could resist the oxidative stress and dysfunction induced by H2O2 in osteoblastic MC3T3-E1 via regulation of mitogen-activated proteinkinase (MAPK)-mediated responses.66 Hyperoside could also play an anti-oxidant effect in dopaminergic neurons induced by 6-hydroxydopamine (6-HODA) via activation of nuclear erythroid 2-related factor 2 (Nrf2)/heme oxygenase-1 (HO-1) signaling.67 Xing et al reported hyperoside further enhanced the cellular antioxidant defense system through MAPK-dependent Kelch-like ECH-associated protein 1 (Keap1)-Nrf2-antioxidant response element (ARE) signaling pathway to up-regulating HO-1 expression.68 In carbon tetrachloride-treated Wistar female rat, hyperoside extracted from Rourea induta Planch. (RIEE) was found to reduce histopathologic alterations observed in the liver and levels of oxidative stress markers.69 In H2O2-induced damage model in Chinese hamster fibroblast (V79-4) cells, hyperoside possessed cytoprotective properties against oxidative stress by scavenging intracellular ROS and enhancing anti-oxidant enzyme activity.70 Besides, Liu et al demonstrated hyperoside exhibited significant radical scavenging activation 1.1-diphenyl-2-picrylhydrazyl (DPPH) radical, hydroxyl radical and ABTS radical, respectively.27
Cardiovascular Protective Effect
Cardiovascular disease has gradually become a serious threat to human health. Many factors including inflammatory, play a very important role in the occurrence and progression of cardiovascular disease. Many studies have shown that hyperoside has a certain effect on the inhibition of cardiovascular related-disease.
As a vital organ to pump blood, heart is in need of a lot of oxygen and also most sensitive to lack of oxygen, so a hypoxic environment could cause damage to the cardiomyocytes even to the cardiovascular system, like coronary and cyanotic congenital heart disease.147 He et al conducted a study that hyperoside could perform a protective effect in hypoxic H9C2 cells cardiomyocytes. Meanwhile, they also found that hyperoside upregulated miR-138 expression levels and inhibited the downstream expression of mixed lineage kinase 3 (MLK3) and lipocalin-2 (Lcn2) to alleviate apoptosis to promote the survival of cardiomyocytes.71
It’s widely acknowledged that vascular endothelial cells play a pivotal role in maintaining normal cardiovascular system function.148 Hyperoside isolated from Acanthopanax chiisanensis Roots exhibited a significant inhibition effect in acetic acid-induced vascular permeability in vivo. And the inhibited effect might be relative with the inhibition of the production of prostaglandin E2 (PEG2) and NO in immune cells.72 Researchers also found that hyperoside could help to against the vascular endothelial injury induced by anticardiolipin antibody in human umbilical vein endothelial cells (HUVECs) via activating mammalian target of rapamycin (mTOR)-mediated autophagy.73 Hyperoside protected HUVECs against H2O2 damage, at least partially, by activating the extra cellular signal-regulated protein kinase (ERK) signaling pathway.74 Hyperoside suppressed vascular inflammatory processes induced by high glucose (HG) in HUVECs and in C57BL/6 mice.75 Besides, hyperoside suppressed lipopolysaccharide (LPS)-mediated release of High-mobility group box 1 (HMGB1) and HMGB1-mediated cytoskeletal rearrangement.76
In vascular smooth muscle cells (VSMCs) model, Huo et al reported hyperoside increased the expression of Nur77 (an orphan nuclear receptor) in rat VSMCs and inhibited VSMCs proliferation and the carotid artery ligation-induced neointimal formation.28 And hyperoside was found to exhibit the effects in preventing atherosclerosis via the oxidized low-density lipoprotein (oxLDL)-lectin-like oxLDL receptor-1 (LOX1)-ERK signal pathway to inhibit VSMC proliferation.77
Hyperoside produced significant hyperpolarization and relaxation in rat basilar artery smooth muscle cells through both endothelium-dependent and endothelium-independent mechanisms.78 Furthermore, in the myocardial ischemia/reperfusion (I/R) injury model in Sprague-Dawley rat hearts, hyperoside could protect cardiomyocytes from I/R-induced oxidative stress through the activation of ERK-dependent signaling.79
Myocardial infarction (MI) has a high morbidity in cardiovascular diseases. In a model conducted by a ligating surgery of the left anterior descending (LAD) coronary artery in KM mice, hyperoside obviously showed its protective effect on heart injury in MI mice. Furthermore, the protective effect may partial due to the up-regulation of autophagy and suppression of NLRP1 inflammation pathway.80 Cardiac hypertrophy is a condition leading to many cardiac diseases.149 The experiments in vitro and in vivo indicated that hyperoside could alleviate established cardiac remodeling and even prevent the occurrence of cardiac remodeling, via inhibition of protein kinase (AKT) pathway.81 In heart failure rats, hyperoside showed a protective effect via improving the cardiac function of heart failure, meanwhile hyperoside could also repress apoptosis and induce autophagy in H9C2 cells.82
Anti-Inflammatory
In the HT22 murine neuronal cell, hyperoside showed a protective effect in LPS-induced inflammatory model via the regulation of a series of inflammation-related molecules. For instance, the downregulation of IL-1β, IL-6, IL-8, etc. And the upregulation of the expression of catalase (CAT), superoxide dismutase (SOD), reduced glutathione (GSH) and so on. Meanwhile the expression of silent mating type information regulation 2 homolog-1 (SIRT1) was also increased which is the key molecule to active Wnt/β-Catenin and Sonic Hedgehog pathways. All in all, hyperoside could alleviate LPS-induced HT22 murine neuronal cell inflammatory model by the activation of Wnt/β-Catenin and Sonic Hedgehog pathways via upregulating of SIRT1.83 In the LPS-stimulated rat peritoneal macrophages model, hyperoside isolated from the extract of Acanthopanax chiisanensis Nakai root was shown to inhibit nitric oxide (NO) production through inhibition of the expression of inducible nitric oxide synthase (iNOS) by attenuation of p44/p42 MAPK, p38 MAPK and c-Jun N-terminal kinase (JNK).84 Hyperoside is also reported to ameliorate the progression of osteoarthritis via the suppression of phosphatidylinositol 3-kinase (PI3K)/AKT/NF-κB and the MAPK signaling pathways and the enhancement of Nrf2/HO-1.85 Hyperoside also could against the apoptosis and inflammatory in human renal proximal tubule (HK-2) cells exposed to high glucose via miR-499a-5p/nuclear receptor-interacting protein 1 (NRIP1) axis.86 Xu et al reported the therapeutic potential of hyperoside in periodontitis, and the activation of the nuclear factor-κB (NF-κB) pathway was considered to be the key point.87 And in the mouse peritoneal macrophages, hyperoside could inhibit the inflammation through the suppression of the activation of NF-κB signaling pathway.25
Antidepressant Effect
In C6 glioblastoma cell model, hyperoside from St. John’s Wort reduce β2-adrenergic sensitivity,88 and β1-adrenergic receptor density in the plasma membrane.89 Besides, Zheng et al reported hyperoside showed antidepressant effect in the corticosterone-induced neurotoxicity in PC12 cell, and the possible mechanisms is related to elevate BDNF and cAMP response element binding protein (CREB) through AC-cAMP-CREB signal pathway.90
In the study of Orzelska-Górka et al, the researchers conducted tail suspension test (TST) and forced swimming test (FST) on mice and confirmed that hyperoside could significantly reduce immobility, and had no effect to locomotor activity of mice. And the antidepressant effect was observed to be mediated by monoaminergic system and the upregulation of BDNF.91 In another FST on mice and rats, Hass et al found hyperoside (10 and 20 mg/kg i.p. in mice; 1.8 mg/kg/day P.O. in rats) presented a depressor effect on the central nervous system as well as an antidepressant-like effect which is mediated by the dopaminergic system.26 The antidepressant effect of hyperoside was also reported to be related to the serotoninergic system including 5-HT2A, 5-HT2 receptor.92 In a chronic unpredicted mild stress inducing depressive behavior rat model, hyperoside possessed antidepressant-like activity due to its antioxidant effect and regulation on hypothalamus- pituitary- adrenal (HPA) axis activity.93 In a reserpine injection-induced depressant model in mice, hyperoside from the total flavonoid in hypericumperforatum promoted the level of norepinephrine (NE) and 5-hydroxytryptophan (5-HT) in brain and decreased the activity of monoamine oxidase (MAO) to display antidepressant-like effect.94
Neuroprotective Effect
A variety of neuroprotective effects of hyperoside has been confirmed in many studies. In Amyloid β-protein25-35 (Aβ25-35)-induced primary cultured cortical neurons, the neuroprotective effects of hyperoside were investigated. Hyperoside protected Aβ25-35-induced primary cultured cortical neurons via PI3K/Akt/Bad/Bclxl-regulated mitochondrial apoptotic pathway, suggesting hyperoside could be developed into a clinically valuable treatment for Alzheimer’s disease and other neuronal degenerative diseases associated with mitochondrial dysfunction.95 Hyperoside was found to significantly protect neurons from injury induced by reperfusion, while the mechanism may be associated to NO signaling pathway. That hyperoside could inhibit the activation of NF-κB to lessen the expression of iNOS, then ameliorated ERK, JNK and Bcl-2 family-related apoptotic signaling pathway.29 In the model of CoCl2-induced hypoxic/ischemic PC12 cells, hyperoside exhibited neuroprotective effects on hypoxic/ischemic neural injuries through inhibiting apoptosis.96
In vivo, hyperoside, isolated from Cortex Acanthopanacis Radicis, inhibited anti-acetyl-cholinesterase (AchE) activity and potently ameliorated scopolamine-induced memory impairment model in Institute of Cancer Research (ICR) mice. Additionally, the effect may be partially mediated by the acetylcholine-enhancing cholinergic nervous system.97 In a cerebral ischemia reperfusion model in rats, hyperoside was found to up-regulate the level of H2S to causes vasodilation against cerebral ischemia injury. And hyperoside could decreased the synthesis and release of NO to protect brain.98 And researchers also found hyperoside could help against the injury induced by cerebral ischemia reperfusion model in mice and the mechanism may be relative to the upregulation of cystathionine γ-lyase (CES)-H2S signal pathway.99 And further study elaborated the upregulation of H2S could activate KCa and opening KCa channels, leading to the hyperpolarization of VSMC membrane and block Ca2+ influx, then results in vasodilatation.100 Hyperoside could promote the anti-oxidant activity of SOD and CAT to alleviate oxidative stress injury, and contribute to attenuate memory impairment induced by hypobaric hypoxia in rats.101
Anti-Cancer Effects
Anti-Breast Cancer
Breast cancer is the most common cancer in women around the world. In a recent study, Qiu et al found that hyperoside has an anti-breast cancer effect in vivo and in vitro. And they demonstrated that hyperoside could reduce the produce of ROS and the expression of Bcl-2 and X-linked inhibitor of apoptosis (XIAP), resulting in the inhibition of the activation of NF-κB signal pathway to promote the apoptosis of breast cancer cell. Meanwhile the downregulation of ROS could also activate caspase-3 to promote apoptosis.102
As the traditional treatment facing great challenge, combination drug therapy seems to be a new treatment. Sun et al conducted a study to see if hyperoside could be used in combination drug therapy. Besides the anti-breast cancer effect, hyperoside could also attenuate paclitaxel-mediated anti-apoptotic Bcl-2 expression, meanwhile enhanced the expression of Bax. Hyperoside also reversed the toll-like receptor-4 (TLR4)-NF-κB signaling induced by paclitaxel. So hyperoside could enhance the sensitive of breast cancer cells to paclitaxel.103
Anti-Lung Cancer
Inflammatory has been proved to be an important response in the progress of cancers. Lü found that hyperoside induced apoptosis and suppressed inflammatory response in vivo and in vitro. The key point is the activation of caspase-3 to motivate apoptosis and inactivation of NF-κB to inhibit inflammatory.104 Hypoxia is an important factor in the survival and proliferation of lung cancer cells. In a A549 human non-small cell lung cancer (NSCLC) cell model, hyperoside could reverse the downregulation of phosphorylation of adenosine monophosphate-activated protein kinase (AMPK) and expression of HO-1 induced by hypoxia.105 Hyperoside inhibited cell viability of A549 cells in a dose and time dependent-manner and upregulated apoptosis via activation of the p38 MAPK- and JNK-induced mitochondrial death pathway.106
As the activation of ROS could induced apoptosis, hyperoside upregulated ROS in A549 cells leading to the upregulation of p38 MAPK, caspase-3, caspase-9 and Bax. And all the effects above resulting in the promotion of apoptosis in lung cancer cells.107 In A549 cells, hyperoside combined with microRNA-let-7, a tumor suppressor, could perform a synergistic effect on anti-cancer. And the mechanism relative to induce apoptosis and inhibition of proliferation via blocking the process of G1/S phase.108 Hyperoside was confirmed to inhibit the proliferation and induce the apoptosis of T790M-positive NSCLC cells by upregulating the expression of fork head box protein O1 (FoxO1).109 Besides apoptosis, hyperoside could also inhibit Akt/mTOR/p70S6K signal pathway to induce autophagy in A549 cells to perform an anti-cancer effect.110 Another study demonstrated that hyperoside induced apoptosis and inhibited proliferation through caspase-3 and p53 signal pathway to show a preventive effect on lung cancer cells.111
Migration and invasion are the important factors of the poor prognosis of malignancies including NSCLC. Hyperoside could also inhibit the migration and invasion of A549 cells via upregulating PI3K/Akt and p38 MAPK pathways.112
In a H466 human small cell lung cancer (SCLC) cells, hyperoside also exhibited an inhibition effect of cell viability by the activation of ROS/p38 MAPK pathway to promote the apoptosis of cancer cells.107
Anti-Pancreatic Cancer
Pancreatic cancer is the fourth leading cause of death relative to cancer. Boukes, G. J et al primarily proved that hyperoside could be a potential anti-pancreatic cancer drug.107,150 Hyperoside exhibited an effect in inhibiting proliferation and promoting apoptosis in PANC-1 and BxPC-3 cell lines and also inhibited the growth of tumor in vivo. Mechanically, the anti-cancer effect may be associated with the upregulation of Bax/Bcl-2 and Bcl-xL and down-regulation of NF-κB level and the downstream gene expression.113
Anti-Osteocarcinoma
The effects of hyperoside in osteosarcoma cells have been investigated. Hyperoside isolated from Abelmoschus manihot L. alleviated the progression of multiple myeloma in mouse model. The mechanism was found to be relative to the osteoblast genesis by inducing the differentiation of murine pre-osteoblast cells.114 In another model in vitro, hyperoside inhibited the proliferation of osteosarcoma cells by inducing G0/G1 arrest, without causing obvious cell death. Additionally, hyperoside may stimulates osteoblastic differentiation in osteosarcoma cells.115
Anti-Uterine Cancer
Gou et al demonstrated that hyperoside could inhibit proliferation of cervical cancer cells (Hela cells and C-33A cells) by downregulation of C-MYC gene expression.116 Li et al reported hyperoside may play an important role in tumor growth suppression with IC50 value was 38.67 μg/mL after 72 h treatment, 47.82 μg/mL after 48 h, and 69.14 μg/mL after 24 h, respectively. The inhibition may be associated with Ca2+-related mitochondrion apoptotic pathway in RL952 cells.117 Cisplatin is a traditional chemotherapy drug in many cancers including ovarian cancer. But drug resistance is a great challenge for cisplatin. Hyperoside induced apoptosis via the progesterone receptor membrane component 1 (PGRMC1) dependent autophagy. And hyperoside could also sensitize the cell to cisplatin in PGRMC1 overexpressed cells.118
Anti-Liver Cancer
Sen et al’s study primarily demonstrated that hyperoside could inhibit the proliferation of HepG2 cells and arise the apoptosis and the mechanism may be associated to the activation of p53/Caspase signal pathway.119
Bone morphogenetic protein-7 (BMP-7) has been found to have an anti-cancer effect in hepatocellular carcinoma (HCC). Hyperoside suppressed HepG2 cell proliferation by inhibiting BMP-7 dependent PI3K/Akt signal pathway to against HCC.120 Another study demonstrated that hyperoside inhibiting CHRH-7919 cells by the upregulation of caspase 3 and caspase 9 induced apoptosis in vitro and animal tumor in vivo.59
Anti-Skin Cancer
In 7.12 dimethylbenzanthracene and 12-O-tetradecanoylphorbol-13-acetate induced skin cancer model, hyperoside exhibited an anti-cancer effect via the inhibition of PI3K/Akt/mTOR/p38MAPK signal pathway and activation of AMPK to inhibit cell proliferation and induced apoptosis and autophagy.121
Anti-Prostate Cancer
Hyperoside combined with quercetin which has a familiar structure of hyperoside could inhibit prostate cancer cells (PC3 cell) growth via the activation of caspase-3 and cleavage of poly (adenosine ribose) polymerase. Furthermore, hyperoside reduced the expression of prostate tumor-associated microRNAs including microRNA-21 to inhibit metastasis.122
Anti-Renal Cancer
In another study on renal cancer cells. Hyperoside combined with quercetin could inhibit 786-O cells via the downregulation of ROS to induce caspase-3 cleavage and PARP cleavage. Hyperoside could also decrease microRNA-27A and induce zinc finger protein ZBTB10 resulting in the downregulation of specificity protein (Sp) transcription factors including Sp1, Sp3 and Sp4 which overexpressed in cancer cells and inhibit 786-O cells.123
Anti-Colon Cancer
In the SW620 human colorectal cancer cell line, hyperoside from Zanthoxylum bungeanum leaves showed a significant anti-proliferation effect and induced apoptosis. The mechanism may be relative to the upregulation of p53 and p21.124
In HT-29 human colon cancer cells, hyperoside significantly decreased the cell viability in a dose- and time-dependent manner via the activation of mitochondria-dependent apoptotic pathway inhibition of colon cancer.125
Anti-Thyroid Cancer
Nowadays, the morbidity of thyroid cancer is increasing, so it is necessary to get an effective agent in treating thyroid cancer. In SW579 cells, a human thyroid squamous cell carcinoma cell line, apoptosis was induced by hyperoside in a dose-dependent manner. And hyperoside could upregulate the expression of Fas and FasL mRNA and downregulate survivin protein expression to induce apoptosis of cancer cells.126
Antiviral Activity
Duck hepatitis B virus (DHBV) infection model in human hepatoma HepG2.2.15 cell was established to examine anti-hepatitis B virus (HBV) effect of hyperoside extracted from Abelmoschus Manihot (L) medik. The data showed the inhibition rates of hyperoside (0.05 g/L) on HBeAg and HBsAg in the cells were 86.41% and 82.27% on day 8, respectively. Additionally, the DHBV-DNA levels significantly decreased in the treatment of 0.05 g/kg/d and 0.10 g/kg/d of hyperoside. These results suggest hyperoside possess the anti-HBV effect in vitro and in vivo.127 Besides, hyperoside isolated from ethanol extract of Geranium carolinianum L. also exhibited anti-HBV effects in HepG2.2.15 cell.128 Nymphaea alba L. extract in which contains hyperoside showed an anti-hepatitis C virus activity in Huh-7 cell line.129
Hepatoprotective effect
In a N-acetyl-para-amino-phenol (APAP)-induced acute hepatic injury, hyperoside could activate Nrf2 to downregulate LDH and ALT to protect L02 cells.130 As Nrf2 was considered to be important in treatment of acute hepatic injury, exogenous BTB-CNC homolog 1 (Bach1) is an important molecule that regulates Nrf2 pathway. Researchers found hyperoside performed a hepatoprotective effect based on the improvement of Bach1 nuclear export, depending on ERK1/2-Crm1 to upregulate the level of Nrf2 binding to ARE.131 In vivo, hyperoside showed a protective effect against CCl4-induced acute liver injury in ICR mice. The protective effect may be due to enhancement of the antioxidative defense system and suppression of the inflammatory response.132 Kalegari et al showed Rourea induta Planch (RIEE) exhibits antioxidant and hepatic protective activities in vivo, which may be related to hyperoside (flavonoids composition of RIEE).69 Xing et al have conducted research both in vivo and in vitro demonstrating that hyperoside showed a protective effect in CCl4 induced rat liver injury model. Hyperoside could reverse the decrease of SOD and the upregulation of MAD. Hyperoside could protect against oxidative stress-induced liver injury via the PHLPP2-AKT-GSK-3β signaling pathway.133
Non-alcoholic fatty liver disease (NAFLD) is a metabolic disease and there is no medication for it. Sun et al proved that hyperoside performed a protective effect in NAFLD induced by high-fat diet (HFD). After treated with hyperoside, hepatic steatosis, insulin resistance, and inflammatory responses were significantly ameliorated. And the protective effect of hyperoside could be relative to the upregulation of Nr4A1 and leading to macrophage polarization.134
Liver is an organ with abundant blood supply, and heart failure was reported to lead to a liver injury and finally may result in a liver fibrosis. Gou et al found that hyperoside could correct heart failure and improve the liver fibrosis and injury in aortocaval fistula model (ACF) in rats. And the mechanism was proved to be associated with the inhibition of TGF-β1/Smad pathway.135
Other Pharmacological Effects
Allergic Reactions
The regulation of thymic stromal lymphopoietin (TSLP) levels in human mast cell line-1 (HMC-1) cells by hyperoside was investigated. TSLP plays an important role in the pathogenesis of allergic reactions. The results showed hyperoside significantly decreased production and mRNA expression of TSLP and the level of intracellular calcium, receptor-interacting protein 2 (RIP2), active caspase-1, NF-κB, IL-1β and IL-6 in stimulated HMC-1 cells. In conclusion, these investigations establish hyperoside as a potential agent for the treatment of allergic reactions.136 Flavonoids including hyperoside could covalent modify bovine β-lactoglobulin (BLG), so as to alters human intestinal microbiota, might result in the reduction of allergenicity.137
Anticoagulant activity
The potential anticoagulant activities of hyperoside from Oenanthe javanica, were tested in HUVECs. As results showed, hyperoside significantly prolonged APTT and PT and inhibition of the activities of thrombin and FXa, and inhibited production of thrombin and FXa in HUVECs. The report indicates hyperoside possesses antithrombotic activities and offer bases for development of a novel anticoagulant.138
Antidiarrheal Activity
In an experiment, treatment of standardized ethyl acetate fraction of Rhododendron arboreum (EFRA) flowers (the concentration of hyperoside was found to be 0.148%) at 100, 200 and 400 mg/kg exhibited dose-dependent and significant antidiarrheal potential in castor oil and magnesium sulfate-induced diarrhea. Moreover, EFRA at doses of 100, 200 and 400 mg/kg also produced significant dose-dependent reduction in propulsive movement in castor oil-induced gastrointestinal transit using charcoal meal in rats. These results demonstrate that hyperoside has potent antidiarrheal activity for the treatment of diarrhea.151
Antifungal Activity
The camptothecin (CPT), trifolin and hyperoside isolated from amptotheca effectively control fungal pathogens in vitro, including Alternaria alternata, Epicoccum nigrum, Pestalotia guepinii, Drechslera sp., and Fusarium avenaceum. The flavonoids (trifolin and hyperoside) were less effective than CPT at 50 μg/mL, particularly within 20 days after treatment, but more effective at 100 or 150 μg/mL. These investigations showed hyperoside possessed potential antifungal activity.152 Extracts from Hypericum hircinum subsp.majus Exert showed an antifungal effects and further study demonstrated that hyperoside from the extract plays an important part in the antifungal effect.34
Herb-Drug Inhibitory Effect
In vitro, the potential herb-drug inhibitory effects of hyperoside on nine cytochrome P450 (CYP) isoforms in pooled human liver microsomes (HLMs) was investigated. Hyperoside strongly inhibited CYP2D6-catalyzed dextromethorphan O-demethylation, with IC50 values of 1.2 and 0.81 μM after 0 and 15 min of preincubation, and a Ki value of 2.01 μM in HLMs, respectively. In addition, hyperoside decreased CYP2D6-catalyzed dextromethorphan O-demethylation activity of human recombinant cDNA-expressed CYP2D6, with an IC50 value of 3.87 μM. However, other CYPs were not inhibited significantly by hyperoside. As data showed, hyperoside is a potent selective CYP2D6 inhibitor in HLMs, and might cause herb-drug interactions when co-administrated with CYP2D substrates.139
Treat Chronic Kidney Disease
Quercetin and hyperoside treatment (20 mg/kg/day) possess an inhibitory effect on the deposition of calcium oxalate (CaOx) formation in ethylene glycol (EG)-fed rats and might have an prevent effect for stone-forming disease.140 Diabetes is a kind of metabolism disease and has been proved to induce a renal injury. Osteomeles schwerinae Extract was found to prevent diabetes, in which hyperoside was found to be the biological activate compound. In diabetic rats’ model, hyperoside inhibited the platelet-derived growth factor-BB (PDGF-BB)/platelet-derived growth factor-B receptor (PDGFR-β) ligand binding.141 In a D-galactose induced renal aging and injury model, hyperoside was significantly useful in treating renal aging and injury. Further study demonstrated that hyperoside could attenuated renal aging and injury via inhibiting AMPK-autophagy activating kinase 1 (ULK1)-associated autophagy.142
Antinociceptive Effect
According to a study, aqueous extract of Roureainduta has significant antinociceptive action, which seems to be associated with an inhibition of pro-inflammatory cytokines activated pathways. Hyperoside was isolated from aqueous extract of Roureainduta and may be responsible for its effect.153 Periaqueductal gray (PAG) is a structure plays an important role in pain transmission and modulation. Research found that noxious stimuli enhanced the N-methyl-D-aspartate receptor (NMDAR) expression in the PAG. Hyperoside isolated from Rhododendron ponticum L. significantly reversed up-regulation of NR2B-containing NMDAR in PAG and showed an antinociceptive effect in a persistent inflammatory stimulus.154 In research about the acute inflammatory pain model, hyperoside could exhibit an inhibition on the pain induced by acute inflammatory models.155
Pharmacokinetics
In the experiment conducted by Wu et al, HPLC was applied to analyze the pharmacokinetic behavior of hyperoside isolated from hawthorn leaf. The results demonstrated that after intravenous administration, the hyperoside presented a 3 rooms model. And the absorption and elimination of hyperoside was fast as the hyperoside could be detected 3 min after administration, and eliminated in 2 h.156 Ai et al carried out another experiment to study the absorption, distribution and excretion of hyperoside after intragastric administration to rats. The results revealed that hyperoside was absorbed rapidly after intragastric administration with a long half-life of about 4 hours. And the absolute bioavailability was 26% demonstrating that hyperoside could be made into an oral preparation for clinical application.157 In Tan et al’s study showed the half-life of intravenous administration of hyperoside was 264.96±145.80 min detecting with LC/MS.158
Toxicity
Recently the application of natural drugs or the chemical drugs from natural product has become a hotspot, hyperoside has been proved to have many kinds of pharmacological effect, but the toxicity of hyperoside also need to be evaluated.
In research conducted in Wistar rats, the researchers demonstrated that in a long-term oral administration lasted for 6 months, hyperoside has a good safety. And the possible target organ of toxicity is kidney and the damage is reversible.159 Ai et al finished a further study in beagle dogs, the results indicated that hyperoside also had a good safety in a dog model. While in some individuals damage to liver and kidney was observed, suggesting liver and kidney could also be the target organ.160 When applied on healthy pregnant rats, the results showed that hyperoside in a dose of 1000 mg/kg influenced the body weight, the length of the embryos and the length of tail. The results suggested that hyperoside may do harm to the pregnant.161
Clinical Application
Hyperoside is a flavonoids compound isolated from many natural products, but it has not been used in clinical as a drug independently, so in this part we under view the medicinal materials or drugs that hyperoside was the main biological active compound or play an important part in the process in which drugs function. Hypericum perforatum L. what is also call St. John’s wort has effects in anti-virus, anti-inflammatory, diuretic therapy and so forth and has been applied as a drug of first choice in treating depressant.162 Dried rose petals from Rose damascene Mill. is a traditional medicine in Uyghur medicine, hyperoside is the main active compound. It has been used in neurasthenia, dizziness, brain distension, etc.163
At the same time, there are also many Chinese patent medicines containing hyperoside in the market widely used. Jianer Qingjie solution an oral preparation made up of Honeysuckle, chrysanthemum, forsythia, hawthorn, bitter almond, tangerine peel and so on, applied on cough, sore throat, loss of appetite, etc.164 Chaijin Jieyu granules, apreparation composed of six Chinese herbs including bupleurum, Hypericum perforatum, jujube seed, coptis, etc. was used to treat depressant.165 Qianbai Biyan Capsule used for stuffy nose, itching gas heat, runny nose yellow thick, or continuous stuffy nose, slow sense of smell, acute and chronic rhinitis, acute and chronic sinusitis, etc. And hyperoside is indicative component and the main ingredient of the preparation.166
Prospects and Conclusion
The last 200 years, based on the rapid development of chemistry, has brought us a new way to gain new drugs which is quite different from the ancient drug discovery that we can only gain drugs from natural plants, animals or mineral. With the help of chemistry, we can synthesize some of the new drugs that we need, or we can modify the structure of the existing drugs to get new drugs based on the pharmacological action of different pharmacophores. As the drug resistance and side effects of traditional chemical treatment has faced great challenges. So, natural medicine has gradually entered the vision of researchers, but the complex components of natural medicine has brought great obstacle to the pharmacologic effects clarification and drug quality control that made natural medicine has not been as widely used as traditional chemical medicine. With the development and progress of extraction and separation technology. It provides the foundation for the further research and application of natural medicine and natural monomeric drugs have become the focus of research.
Hyperoside as a member of the flavonoids is distributed in many plants including hawthorn, common buckwheat, etc. and abundant in content. Based on the structure of hyperoside, it showed a variety of bioactive effects especially anti-oxidant effect, anti-inflammatory and anti-allergy effects, etc. While the further studies suggested that hyperoside plays an important role in various cancer models, and its mechanism of action is also very diverse. Meanwhile, hyperoside also showed a long half-life for 4 hours and the safety experiments also proves that it has good safety. All the results above indicated that hyperoside has broad application prospect.
However, the knowledge and systemic data is still incomplete and requires extensive research. Therefore, it is important to further investigate the pharmacokinetics, pharmacodynamics, toxicity and molecular mechanisms of hyperoside based on modern concepts of diseases pathophysiology. Moreover, bioactivity-guided isolation and quantification of hyperoside and subsequent investigation of pharmacological effects will promote the development and wide the usage of this compound. Moreover, just like most natural drug monomers, hyperoside, due to its chemical properties, makes it a large gap in stability from traditional synthetic small molecule drugs, is also an important factor restricting the clinical use of many natural compounds. In addition, due to the lack of clinical application demand, there is a lack of research on the synthesis process of corresponding molecules, and relying on natural extraction alone is often difficult to meet the needs of large-scale application, which further limits the possibility of its clinical application.
Based on the problems above, hyperoside has not been made into a single drug for clinical application, only performed as an important active gradient in a drug which indicated that the application of hyperoside needs further study. Expanding this knowledge will make for a more practical application of hyperoside in medicine and society.
In addition to the application of hyperoside, we also have other gains from this review we believe that except constantly discovering new natural compounds, we can also improve the stability of existing potential drug molecules by means of multidisciplinary crossover, structural improvement, or preparation of certain dosage forms, so as to enhance their application prospects. So as to realize the transformation from laboratory to clinical application, so that our research results can benefit everybody.
Funding Statement
This project is supported by a grant from the Chongqing Clinical Pharmacy Key Specialties Construction Project, Technology Project of Chongqing Health and Family Planning Commission (2019MSXM010) and Natural Science Foundation of Chongqing (No. cstc2020jcyj-msxmX0223).
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Cragg GM, Newman DJ. Natural products: a continuing source of novel drug leads. Biochim Biophys Acta Gen Subjects. 2013;1830(6):3670–3695. doi: 10.1016/j.bbagen.2013.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Demir Y. Naphthoquinones, benzoquinones, and anthraquinones: molecular docking, ADME and inhibition studies on human serum paraoxonase‐1 associated with cardiovascular diseases. Drug Dev Res. 2020;81:628–636. doi: 10.1002/ddr.21667 [DOI] [PubMed] [Google Scholar]
- 3.Demir Y, Durmaz L, Taslimi P, et al. Antidiabetic properties of dietary phenolic compounds: inhibition effects on α‐amylase, aldose reductase, and α‐glycosidase. Biotechnol Appl Biochem. 2019;66(5):781–786. doi: 10.1002/bab.1781 [DOI] [PubMed] [Google Scholar]
- 4.Aslan HE, Demir Y, Özaslan MS, et al. The behavior of some chalcones on acetylcholinesterase and carbonic anhydrase activity. Drug Chem Toxicol. 2019;42(6):634–640. doi: 10.1080/01480545.2018.1463242 [DOI] [PubMed] [Google Scholar]
- 5.Çağlayan C, Taslimi P, Demir Y, et al. The effects of zingerone against vancomycin‐induced lung, liver, kidney and testis toxicity in rats: the behavior of some metabolic enzymes. J Biochem Mol Toxicol. 2019;33:e22381. doi: 10.1002/jbt.22381 [DOI] [PubMed] [Google Scholar]
- 6.Mahmudov I, Demir Y, Sert Y, et al. Synthesis and inhibition profiles of N-benzyl- and N-allyl aniline derivatives against carbonic anhydrase and acetylcholinesterase – a molecular docking study. Arab J Chem. 2022;15(3):103645. doi: 10.1016/j.arabjc.2021.103645 [DOI] [Google Scholar]
- 7.Anil DA, Aydin BO, Demir Y, et al. Design, synthesis, biological evaluation and molecular docking studies of novel 1H-1,2,3-Triazole derivatives as potent inhibitors of carbonic anhydrase, acetylcholinesterase and aldose reductase. J Mol Struct. 2022;1257:132613. doi: 10.1016/j.molstruc.2022.132613 [DOI] [Google Scholar]
- 8.Ceylan H, Demir Y, Beydemir S. Inhibitory effects of Usnic and Carnosic Acid on some metabolic enzymes: an in vitro study. Protein Pept Lett. 2019;26:364–370. doi: 10.2174/0929866526666190301115122 [DOI] [PubMed] [Google Scholar]
- 9.Demir Y, Ceylan H, Türkeş C, et al. Molecular docking and inhibition studies of vulpinic, carnosic and usnic acids on polyol pathway enzymes. J Biomol Struct Dyn. 2022;40:12008–12021. doi: 10.1080/07391102.2021.1967195 [DOI] [PubMed] [Google Scholar]
- 10.Persson PB, Persson AB. Age your garlic for longevity! Acta Physiol. 2012;205(1):1–2. doi: 10.1111/j.1748-1716.2012.02424.x [DOI] [PubMed] [Google Scholar]
- 11.Öztürk C, Bayrak S, Demir Y, et al. Some indazoles as alternative inhibitors for potato polyphenol oxidase. Biotechnol Appl Biochem. 2022;69(5):2249–2256. doi: 10.1002/bab.2283 [DOI] [PubMed] [Google Scholar]
- 12.Özaslan MS, Sağlamtaş R, Demir Y, et al. Isolation of some phenolic compounds from plantago subulata L. and determination of their antidiabetic, anticholinesterase, antiepileptic and antioxidant activity. Chem Biodivers. 2022;19(8). doi: 10.1002/cbdv.202200280 [DOI] [PubMed] [Google Scholar]
- 13.Bae J. Role of high mobility group box 1 in inflammatory disease: focus on sepsis. Arch Pharm Res. 2012;35(9):1511–1523. doi: 10.1007/s12272-012-0901-5 [DOI] [PubMed] [Google Scholar]
- 14.Türkeş C, Demir Y, Beydemir Ş. In vitro inhibitory activity and molecular docking study of selected natural phenolic compounds as AR and SDH inhibitors. ChemistrySelect. 2022;7(48):e202204050. doi: 10.1002/slct.202204050 [DOI] [Google Scholar]
- 15.Hollman P, Katan M. Absorption, metabolism of dietary flavonoids. Biomed&Pharmacother. 1997;51:305–310. [DOI] [PubMed] [Google Scholar]
- 16.Rice-Evans CA, Miller NJ, Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med. 1996;20(7):933–956. doi: 10.1016/0891-5849(95)02227-9 [DOI] [PubMed] [Google Scholar]
- 17.Middleton EJ, Drzewiecki G. Flavonoid inhibition of human basophil histamine release stimulated by various agents. Biochem Pharmacol. 1984;33(21):3333–3338. doi: 10.1016/0006-2952(84)90102-3 [DOI] [PubMed] [Google Scholar]
- 18.Mukaida N. Interleukin-8: an expanding universe beyond neutrophil chemotaxis and activation. Int J Hematol. 2000;72(4):391–398. [PubMed] [Google Scholar]
- 19.Palabıyık E, Sulumer AN, Uguz H, et al. Assessment of hypolipidemic and anti-inflammatory properties of walnut (Juglans regia) seed coat extract and modulates some metabolic enzymes activity in triton WR-1339-induced hyperlipidemia in rat kidney, liver, and heart. J Mol Recognit. 2023;36(3). doi: 10.1002/jmr.3004 [DOI] [PubMed] [Google Scholar]
- 20.Bayrak S, Öztürk C, Demir Y, et al. Purification of polyphenol oxidase from potato and investigation of the inhibitory effects of phenolic acids on enzyme activity. Protein Pept Lett. 2020;27(3):187–192. doi: 10.2174/0929866526666191002142301 [DOI] [PubMed] [Google Scholar]
- 21.Weaver BA, Bement W, Bement W. How Taxol/paclitaxel kills cancer cells. Mol Biol Cell. 2014;25(18):2677–2681. doi: 10.1091/mbc.E14-04-0916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang J, He Y, Li Y, et al. Advances in the research on the targets of anti-malaria actions of artemisinin. Pharmacol Ther. 2020;216:107697. doi: 10.1016/j.pharmthera.2020.107697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Federico A, Dallio M, Loguercio C. Silymarin/silybin and chronic liver disease: a marriage of many years. Molecules. 2017;22(2):191. doi: 10.3390/molecules22020191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zou Y, Lu Y, Wei D. Antioxidant activity of a flavonoid-rich extract of hypericum perforatum L. in vitro. J Agric Food Chem. 2004;52(16):5032–5039. doi: 10.1021/jf049571r [DOI] [PubMed] [Google Scholar]
- 25.Kim S, Um J-Y, Hong S-H, et al. Anti-Inflammatory activity of hyperoside through the suppression of nuclear factor-κB activation in mouse peritoneal macrophages. Am J Chin Med. 2012;39:171–181. doi: 10.1142/S0192415X11008737 [DOI] [PubMed] [Google Scholar]
- 26.Haas JS, Dischkaln Stolz E, Heemann Betti A, et al. The anti-immobility effect of hyperoside on the forced swimming test in rats is mediated by the D2-like receptors activation. Planta Med. 2011;77(04):334–339. doi: 10.1055/s-0030-1250386 [DOI] [PubMed] [Google Scholar]
- 27.Liu X, Zhu L, Tan J, et al. Glucosidase inhibitory activity and antioxidant activity of flavonoid compound and triterpenoid compound from agrimonia pilosa ledeb. BMC Complement Altern Med. 2014;14(1):12. doi: 10.1186/1472-6882-14-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huo Y, Yi B, Chen M, et al. Induction of Nur77 by hyperoside inhibits vascular smooth muscle cell proliferation and neointimal formation. Biochem Pharmacol. 2014;92(4):590–598. doi: 10.1016/j.bcp.2014.09.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu R, Xiong Q-J, Shu Q, et al. Hyperoside protects cortical neurons from oxygen–glucose deprivation–reperfusion induced injury via nitric oxide signal pathway. Brain Res. 2012;1469:164–173. doi: 10.1016/j.brainres.2012.06.044 [DOI] [PubMed] [Google Scholar]
- 30.Wang H, Hou X, Li B, et al. Study on active components of cuscuta chinensis promoting neural stem cells proliferation: bioassay-guided fractionation. Molecules. 2021;26(21):6634. doi: 10.3390/molecules26216634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yang L, Chen Q, Wang F, et al. Antiosteoporotic compounds from seeds of Cuscuta chinensis. J Ethnopharmacol. 2011;135(2):553–560. doi: 10.1016/j.jep.2011.03.056 [DOI] [PubMed] [Google Scholar]
- 32.Cui Y, Zhao Z, Liu Z, et al. Purification and identification of buckwheat hull flavonoids and its comparative evaluation on antioxidant and cytoprotective activity in vitro. Food Sci Nutr. 2020;8(7):3882–3892. doi: 10.1002/fsn3.1683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sun Y, Pan Z, Yang C, et al. Comparative assessment of phenolic profiles, cellular antioxidant and antiproliferative activities in ten varieties of sweet potato (Ipomoea Batatas) storage roots. Molecules (Basel, Switzerland). 2019;24(24):4476. doi: 10.3390/molecules24244476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tocci N, Perenzoni D, Iamonico D, et al. Extracts from hypericum hircinum subsp. majus exert antifungal activity against a panel of sensitive and drug-resistant clinical strains. Front Pharmacol. 2018;9. doi: 10.3389/fphar.2018.00382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wu P, Li F, Zhang J, et al. Phytochemical compositions of extract from peel of hawthorn fruit, and its antioxidant capacity, cell growth inhibition, and acetylcholinesterase inhibitory activity. BMC Complement Altern Med. 2017;17(1). doi: 10.1186/s12906-017-1662-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lund JA, Brown PN, Shipley PR. Quantification of North American and European Crataegus flavonoids by nuclear magnetic resonance spectrometry. Fitoterapia. 2020;143:104537. doi: 10.1016/j.fitote.2020.104537 [DOI] [PubMed] [Google Scholar]
- 37.Hafsa J, Hammi KM, Khedher MRB, et al. Inhibition of protein glycation, antioxidant and antiproliferative activities of Carpobrotus edulis extracts. Biomed Pharmacother. 2016;84:1496–1503. doi: 10.1016/j.biopha.2016.11.046 [DOI] [PubMed] [Google Scholar]
- 38.Mustapha N, Mokdad-Bzéouich I, Sassi A, et al. Immunomodulatory potencies of isolated compounds from Crataegus azarolus through their antioxidant activities. Tumor Biol. 2016;37(6):7967–7980. doi: 10.1007/s13277-015-4517-5 [DOI] [PubMed] [Google Scholar]
- 39.Zorzetto C, Sánchez-Mateo CC, Rabanal RM, et al. Phytochemical analysis and in vitro biological activity of three hypericum species from the canary Islands (Hypericum reflexum, Hypericum canariense and Hypericum grandifolium). Fitoterapia. 2015;100:95–109. doi: 10.1016/j.fitote.2014.11.013 [DOI] [PubMed] [Google Scholar]
- 40.Shabbir M, Afsar T, Razak S, et al. Phytochemical analysis and Evaluation of hepatoprotective effect of Maytenus royleanus leaves extract against anti-tuberculosis drug induced liver injury in mice. Lipids Health Dis. 2020;19(1). doi: 10.1186/s12944-020-01231-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cardoso ML, Bersani Amado C, Outuki,P, et al. A high performance liquid chromatography with ultraviolet method for Eschweilera nana leaves and their anti-inflammatory and antioxidant activities. Pharmacogn Mag. 2015;11(43):619–626. doi: 10.4103/0973-1296.160464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mansur S, Abdulla R, Ayupbec A, et al. Chemical fingerprint analysis and quantitative analysis of Rosa rugosa by UPLC-DAD. Molecules. 2016;21(12):1754. doi: 10.3390/molecules21121754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xie Q, Ding L, Wei Y, et al. Determination of major components and fingerprint analysis of flaveria bidentis (L.) Kuntze. J Chromatogr Sci. 2014;52(3):252–257. doi: 10.1093/chromsci/bmt020 [DOI] [PubMed] [Google Scholar]
- 44.Shen J, Yang K, Jiang C, et al. Development and application of a rapid HPLC method for simultaneous determination of hyperoside, isoquercitrin and eleutheroside E in Apocynum venetum L. and Eleutherococcus senticosus. BMC Chem. 2020;14(1). doi: 10.1186/s13065-020-00687-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li L. Simulation determination of eight active components in Polygonum aviculare L. by HPLC. J Taishan Med Coll. 2021;42:61–65. [Google Scholar]
- 46.Wang H. Determination of 5 flavonoids in Hypericum ascyron L. by HPLC. Cent South Pharm. 2021;19:1911–1914. [Google Scholar]
- 47.Ozbilgin S, Acıkara ÖB, Akkol EK, et al. In vivo wound-healing activity of Euphorbia characias subsp. wulfenii: isolation and quantification of quercetin glycosides as bioactive compounds. J Ethnopharmacol. 2018;224:400–408. doi: 10.1016/j.jep.2018.06.015 [DOI] [PubMed] [Google Scholar]
- 48.Dresler S, Kováčik J, Strzemski M, et al. Methodological aspects of biologically active compounds quantification in the genus Hypericum. J Pharm Biomed Anal. 2018;155:82–90. doi: 10.1016/j.jpba.2018.03.048 [DOI] [PubMed] [Google Scholar]
- 49.Zeliou K, Kontaxis NI, Margianni E, et al. Optimized and Validated HPLC Analysis of St. John’s wort extract and final products by simultaneous determination of major ingredients. J Chromatogr Sci. 2017;55(8):805–812. doi: 10.1093/chromsci/bmx040 [DOI] [PubMed] [Google Scholar]
- 50.Jia Q, Huang X, Yao G, et al. Pharmacokinetic study of thirteen ingredients after the oral administration of flos chrysanthemi extract in rats by UPLC-MS/MS. Biomed Res Int. 2020;2020:1–10. doi: 10.1155/2020/8420409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Males Z, Saric D, Bojic M. Quantitative determination of flavonoids and chlorogenic acid in the leaves of arbutus unedo L. using thin layer chromatography. J Anal Methods Chem. 2013;2013:385473. doi: 10.1155/2013/385473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu J, Zhang Z, Yang L, et al. Molecular structure and spectral characteristics of hyperoside and analysis of its molecular imprinting adsorption properties based on density functional theory. J Mol Graph Model. 2019;88:228–236. doi: 10.1016/j.jmgm.2019.01.005 [DOI] [PubMed] [Google Scholar]
- 53.Cheng Y. Inhibition effect of nine flavonoids on human cancer cells and their structure-activity relationship analysis. Northwest Pharm J. 2014;29:187–190. [Google Scholar]
- 54.Chen J, Zhu Z-Q, Hu T-X, et al. Structure-activity relationship of natural flavonoids in hydroxyl radical scavenging effects. Acta Pharmacol Sin. 2002;23:667–672. [PubMed] [Google Scholar]
- 55.Plochmann K, Korte G, Koutsilieri E, et al. Structure–activity relationships of flavonoid-induced cytotoxicity on human leukemia cells. Arch Biochem Biophys. 2007;460(1):1–9. doi: 10.1016/j.abb.2007.02.003 [DOI] [PubMed] [Google Scholar]
- 56.Zhang Y, Wu X, Ding X. Studies on the relationship between the structure of flavonoids and their scavenging capacity on active oxygen radicals by means of chemiluminescence. Nat Prod Res Dev. 1998;10:26–33. [Google Scholar]
- 57.Zhao J. Study of 6 flavonoids compounds for the scavenging superoxide anion free radical ability and the structure-activity relationships. China Med Herald. 2014;11:7–10, 14. [Google Scholar]
- 58.Cai YZ, Sun M, Xing J, et al. Structure-radical scavenging activity relationships of phenolic compounds from traditional Chinese medicinal plants. Life Sci. 2006;78(25):2872–2888. doi: 10.1016/j.lfs.2005.11.004 [DOI] [PubMed] [Google Scholar]
- 59.Feng Y, Qin G, Chang S, et al. Antitumor effect of hyperoside loaded in charge reversed and mitochondria-targeted liposomes. Int J Nanomedicine. 2021;16:3073–3089. doi: 10.2147/IJN.S297716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gao Y, Fang L, Wang X, et al. Antioxidant activity evaluation of dietary flavonoid hyperoside using saccharomyces cerevisiae as a model. Molecules. 2019;24(4):788. doi: 10.3390/molecules24040788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Biagi M, Noto D, Corsini M, et al. Antioxidant effect of the castanea sativa mill. Leaf extract on oxidative stress induced upon human spermatozoa. Oxid Med Cell Longev. 2019;2019:8926075. doi: 10.1155/2019/8926075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu Z, Tao X, Zhang C, et al. Protective effects of hyperoside (quercetin-3-o-galactoside) to PC12 cells against cytotoxicity induced by hydrogen peroxide and tert-butyl hydroperoxide. Biomed Pharmacother. 2005;59(9):481–490. doi: 10.1016/j.biopha.2005.06.009 [DOI] [PubMed] [Google Scholar]
- 63.Li HB, Yi X, Gao JM, et al. The mechanism of hyperoside protection of ECV-304 cells against tert-butyl hydroperoxide-induced injury. Pharmacology. 2008;82(2):105–113. doi: 10.1159/000139146 [DOI] [PubMed] [Google Scholar]
- 64.Ohguchi K, Nakajima C, Oyama M, et al. Inhibitory effects of flavonoid glycosides isolated from the peel of Japanese persimmon (Diospyros kaki ‘Fuyu’) on melanin biosynthesis. Biol Pharm Bull. 2010;33:122–124. doi: 10.1248/bpb.33.122 [DOI] [PubMed] [Google Scholar]
- 65.Kim YJ. Hyperin and quercetin modulate oxidative stress-induced melanogenesis. Biol Pharm Bull. 2012;35(11):2023–2027. doi: 10.1248/bpb.b12-00592 [DOI] [PubMed] [Google Scholar]
- 66.Qi X, Li B, Wu WL, et al. Protective effect of hyperoside against hydrogen peroxide-induced dysfunction and oxidative stress in osteoblastic MC3T3-E1 cells. Artif Cells Nanomed Biotechnol. 2020;48:377–383. doi: 10.1080/21691401.2019.1709851 [DOI] [PubMed] [Google Scholar]
- 67.Kwon SH, Lee SR, Park YJ, et al. Suppression of 6-hydroxydopamine-induced oxidative stress by hyperoside via activation of Nrf2/HO-1 signaling in dopaminergic neurons. Int J Mol Sci. 2019. doi: 10.3390/ijms20235832 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Xing H, Liu Y, Chen J-H, et al. Hyperoside attenuates hydrogen peroxide-induced L02 cell damage via MAPK-dependent Keap1-Nrf2-ARE signaling pathway. Biochem Biophys Res Commun. 2011;410:759–765. doi: 10.1016/j.bbrc.2011.06.046 [DOI] [PubMed] [Google Scholar]
- 69.Kalegari M, Gemin CAB, Araújo-Silva G, et al. Chemical composition, antioxidant activity and hepatoprotective potential of Rourea induta Planch. (Connaraceae) against CCl4-induced liver injury in female rats. Nutrition. 2014;30(6):713–718. doi: 10.1016/j.nut.2013.11.004 [DOI] [PubMed] [Google Scholar]
- 70.Piao MJ, Kang KA, Zhang R, et al. Hyperoside prevents oxidative damage induced by hydrogen peroxide in lung fibroblast cells via an antioxidant effect. Biochim Biophys Acta Gen Subjects. 2008;1780(12):1448–1457. doi: 10.1016/j.bbagen.2008.07.012 [DOI] [PubMed] [Google Scholar]
- 71.He S, Yin X, Wu F, et al. Hyperoside protects cardiomyocytes against hypoxiainduced injury via upregulation of microRNA138. Mol Med Rep. 2021;23. doi: 10.3892/mmr.2021.11925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lee S, Jung SH, Lee YS, et al. Antiinflammatory activity of hyperin from Acanthopanax chiisanensis roots. Arch Pharm Res. 2004;27:628–632. doi: 10.1007/BF02980162 [DOI] [PubMed] [Google Scholar]
- 73.Wei A, Xiao H, Xu G, et al. Hyperoside protects human umbilical vein endothelial cells against anticardiolipin antibody-induced injury by activating autophagy. Front Pharmacol. 2020;11. doi: 10.3389/fphar.2020.00762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Li Z, Liu J-C, Hu J, et al. Protective effects of hyperoside against human umbilical vein endothelial cell damage induced by hydrogen peroxide. J Ethnopharmacol. 2012;139(2):388–394. doi: 10.1016/j.jep.2011.11.020 [DOI] [PubMed] [Google Scholar]
- 75.Ku S, Kwak S, Kwon O-J, et al. Hyperoside inhibits high-glucose-induced vascular inflammation in vitro and in vivo. Inflammation. 2014;37(5):1389–1400. doi: 10.1007/s10753-014-9863-8 [DOI] [PubMed] [Google Scholar]
- 76.Ku S, Zhou W, Lee W, et al. Anti-inflammatory effects of hyperoside in human endothelial cells and in mice. Inflammation. 2015;38(2):784–799. doi: 10.1007/s10753-014-9989-8 [DOI] [PubMed] [Google Scholar]
- 77.Zhang Z, Zhang D, Du B, et al. Hyperoside inhibits the effects induced by oxidized low-density lipoprotein in vascular smooth muscle cells via oxLDL-LOX-1-ERK pathway. Mol Cell Biochem. 2017;433(1–2):169–176. doi: 10.1007/s11010-017-3025-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Fan Y, Chen Z-W, Guo Y, et al. Cellular mechanisms underlying Hyperin-induced relaxation of rat basilar artery. Fitoterapia. 2011;82(4):626–631. doi: 10.1016/j.fitote.2011.01.023 [DOI] [PubMed] [Google Scholar]
- 79.Li Z, Hu J, Li Y-L, et al. The effect of hyperoside on the functional recovery of the ischemic/reperfused isolated rat heart: potential involvement of the extracellular signal-regulated kinase 1/2 signaling pathway. Free Radic Biol Med. 2013;57:132–140. doi: 10.1016/j.freeradbiomed.2012.12.023 [DOI] [PubMed] [Google Scholar]
- 80.Yang Y, Li J, Rao T, et al. The role and mechanism of hyperoside against myocardial infarction in mice by regulating autophagy via NLRP1 inflammation pathway. J Ethnopharmacol. 2021;276:114187. doi: 10.1016/j.jep.2021.114187 [DOI] [PubMed] [Google Scholar]
- 81.Wang X, Liu Y, Xiao L, et al. Hyperoside protects against pressure overload-induced cardiac remodeling via the AKT signaling pathway. Cell Physiol Biochem. 2018;51(2):827–841. doi: 10.1159/000495368 [DOI] [PubMed] [Google Scholar]
- 82.Guo X, Zhang Y, Lu C, et al. Protective effect of hyperoside on heart failure rats via attenuating myocardial apoptosis and inducing autophagy. Biosci Biotechnol Biochem. 2020;84(4):714–724. doi: 10.1080/09168451.2019.1685369 [DOI] [PubMed] [Google Scholar]
- 83.Huang J, Zhou L, Chen J, et al. Hyperoside attenuate inflammation in HT22 cells via upregulating SIRT1 to activities Wnt/β-catenin and sonic hedgehog pathways. Neural Plast. 2021;2021:1–10. doi: 10.1155/2021/8706400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lee S, Park HS, Notsu Y, et al. Effects of hyperin, isoquercitrin and quercetin on lipopolysaccharide-induced nitrite production in rat peritoneal macrophages. Phytother Res. 2008;22:1552–1556. doi: 10.1002/ptr.2529 [DOI] [PubMed] [Google Scholar]
- 85.Sun K, Luo J, Jing X, et al. Hyperoside ameliorates the progression of osteoarthritis: an in vitro and in vivo study. Phytomedicine. 2021;80:153387. doi: 10.1016/j.phymed.2020.153387 [DOI] [PubMed] [Google Scholar]
- 86.Zhou J, Zhang S, Sun X, et al. Hyperoside Protects HK-2 cells against high glucose-induced apoptosis and inflammation via the miR-499a-5p/NRIP1 pathway. Pathol Oncol Res. 2021;27. doi: 10.3389/pore.2021.629829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Xu T, Wu X, Zhou Z, et al. Hyperoside ameliorates periodontitis in rats by promoting osteogenic differentiation of BMSCs via activation of the NF‐κB pathway. FEBS Open Bio. 2020;10:1843–1855. doi: 10.1002/2211-5463.12937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Prenner L, Sieben A, Zeller K, et al. Reduction of high-affinity β 2-adrenergic receptor binding by hyperforin and hyperoside on rat C6 Glioblastoma cells measured by fluorescence correlation spectroscopy. Biochemistry. 2007;46:5106–5113. doi: 10.1021/bi6025819 [DOI] [PubMed] [Google Scholar]
- 89.Jakobsa D, Hage-Hülsmann A, Prenner L, et al. Downregulation of b1-adrenergic receptors in rat C6 glioblastoma cells by hyperforin and hyperoside from St John’s wort. J Pharm Pharmacol. 2013;65:907–915. doi: 10.1111/jphp.12050 [DOI] [PubMed] [Google Scholar]
- 90.Zheng M, Liu C, Pan F, et al. Antidepressant-like effect of hyperoside isolated from Apocynum venetum leaves: possible cellular mechanisms. Phytomedicine. 2012;19:145–149. doi: 10.1016/j.phymed.2011.06.029 [DOI] [PubMed] [Google Scholar]
- 91.Orzelska-Górka J, Szewczyk K, Gawrońska-Grzywacz M, et al. Monoaminergic system is implicated in the antidepressant-like effect of hyperoside and protocatechuic acid isolated from Impatiens glandulifera Royle in mice. Neurochem Int. 2019;128:206–214. doi: 10.1016/j.neuint.2019.05.006 [DOI] [PubMed] [Google Scholar]
- 92.Zhu ZM, Fan YJ, Pan Y, et al. Studies on the antidepressant-like effect of hyperoside on the possible mechanism of 5-HT system. J Changchun Normal Univ. 2018;37:83–87. [Google Scholar]
- 93.Yan Z, Chen-chen Z. Effect of hyperin on depressive behavior of rats induced by chronic unpredicted mild stress. Chin J New Drugs Clin Rem. 2017;36:150–156. [Google Scholar]
- 94.Li X, Wei CE, Zhao MB, et al. Experimental study of the total flavonoid in Hypericum perforatum on depression. China J Chin Mater Med. 2005;30:1184–1188. [PubMed] [Google Scholar]
- 95.Zeng K, Wang X-M, Ko H, et al. Hyperoside protects primary rat cortical neurons from neurotoxicity induced by amyloid β-protein via the PI3K/Akt/Bad/BclXL-regulated mitochondrial apoptotic pathway. Eur J Pharmacol. 2011;672(1–3):45–55. doi: 10.1016/j.ejphar.2011.09.177 [DOI] [PubMed] [Google Scholar]
- 96.Zeng K, Wang X, Fu H, et al. Protective effects and mechanism of hyperin on CoCl2-induced PC12 cells. China J Chin Mater Med. 2011;36:2409–2412. [PubMed] [Google Scholar]
- 97.Nam Y, Lee D. Ameliorating effects of constituents from cortex acanthopanacis radicis on memory impairment in mice induced by scopolamine. J Trad Chin Med. 2014;34(1):57–62. doi: 10.1016/S0254-6272(14)60055-8 [DOI] [PubMed] [Google Scholar]
- 98.Han J. Mechanism of hyperoside- induced dilatation in middle cerebral arteries of rats subjected to cerebral ischemia reperfusion. Chin Pharm J. 2015;50:595–601. [Google Scholar]
- 99.Shanshan G, Shuo C, Zhiwu C. The H2S mechanism of Hyp against cerebral ischemia reperfusion injury in mice. Acta Univ Med Anhui. 2016;51:1292–1296. [Google Scholar]
- 100.Han J, Xuan JL, Hu HR, et al. Effects and mechanisms of hyperoside on vascular endothelium function in middle cerebral arteries of rats ex vivo. China J Chin Mater Med. 2014;39:4849–4855. [PubMed] [Google Scholar]
- 101.Jin-song L, Jian-hong C, Min-jie M. Hyperoside attenuated hypoxia-induced memory impairment by antioxidative activity. J Reg Anat Oper Surg. 2015;24:181–184. [Google Scholar]
- 102.Qiu J, Zhang T, Zhu X, et al. Hyperoside induces breast cancer cells apoptosis via ROS-Mediated NF-κB signaling pathway. Int J Mol Sci. 2020;21:131. doi: 10.3390/ijms21010131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sun T, Liu Y, Li M, et al. Administration with hyperoside sensitizes breast cancer cells to paclitaxel by blocking the TLR4 signaling. Mol Cell Probes. 2020;53:101602. doi: 10.1016/j.mcp.2020.101602 [DOI] [PubMed] [Google Scholar]
- 104.Lü P. Inhibitory effects of hyperoside on lung cancer by inducing apoptosis and suppressing inflammatory response via caspase-3 and NF-κB signaling pathway. Biomed Pharmacother. 2016;82:216–225. doi: 10.1016/j.biopha.2016.05.006 [DOI] [PubMed] [Google Scholar]
- 105.Chen D, Wu Y-X, Qiu Y-B, et al. Hyperoside suppresses hypoxia-induced A549 survival and proliferation through ferrous accumulation via AMPK/HO-1 axis. Phytomedicine. 2020;67:153138. doi: 10.1016/j.phymed.2019.153138 [DOI] [PubMed] [Google Scholar]
- 106.Yang Y, Tantai J, Sun Y, et al. Effect of hyperoside on the apoptosis of A549 human non-small cell lung cancer cells and the underlying mechanism. Mol Med Rep. 2017;16(5):6483–6488. doi: 10.3892/mmr.2017.7453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Jia X, Zhang Q, Xu L, et al. Lotus leaf flavonoids induce apoptosis of human lung cancer A549 cells through the ROS/p38 MAPK pathway. Biol Res. 2021;54(1). doi: 10.1186/s40659-021-00330-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Li J, Liao XH, Xiang Y, et al. Hyperoside and let-7a-5p synergistically inhibits lung cancer cell proliferation via inducing G1/S phase arrest. Gene. 2018;679:232–240. doi: 10.1016/j.gene.2018.09.011 [DOI] [PubMed] [Google Scholar]
- 109.Hu Z, Zhao P, Xu H. Hyperoside exhibits anticancer activity in nonsmall cell lung cancer cells with T790M mutations by upregulating FoxO1 via CCAT1. Oncol Rep. 2020;43:617–624. doi: 10.3892/or.2019.7440 [DOI] [PubMed] [Google Scholar]
- 110.Fu T, Wang L, Jin XN, et al. Hyperoside induces both autophagy and apoptosis in non-small cell lung cancer cells in vitro. Acta Pharmacol Sin. 2016;37:505–518. doi: 10.1038/aps.2015.148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Liu Y, Liu G-H, Mei JJ, et al. The preventive effects of hyperoside on lung cancer in vitro by inducing apoptosis and inhibiting proliferation through Caspase-3 and P53 signaling pathway. Biomed Pharmacother. 2016;83:381–391. doi: 10.1016/j.biopha.2016.06.035 [DOI] [PubMed] [Google Scholar]
- 112.Yang Y, Sun Y, Guo X, et al. Hyperoside inhibited the migration and invasion of lung cancer cells through the upregulation of PI3K/AKT and p38 MAPK pathways. Int J Clin Exp Pathol. 2017;10:9382–9390. [PMC free article] [PubMed] [Google Scholar]
- 113.Li Y, Wang Y, Li L, et al. Hyperoside induces apoptosis and inhibits growth in pancreatic cancer via Bcl-2 family and NF-κB signaling pathway both in vitro and in vivo. Tumor Biol. 2016;37(6):7345–7355. doi: 10.1007/s13277-015-4552-2 [DOI] [PubMed] [Google Scholar]
- 114.Hou J, Qian J, Li Z, et al. Bioactive compounds from abelmoschus manihot L. alleviate the progression of multiple myeloma in mouse model and improve bone marrow microenvironment. OncoTargets and Therapy. 2020;13:959–973. doi: 10.2147/OTT.S235944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhang N, Ying M-D, Wu Y-P, et al. Hyperoside, a flavonoid compound, inhibits proliferation and stimulates osteogenic differentiation of human osteosarcoma cells. PLoS One. 2014;9(7):e98973. doi: 10.1371/journal.pone.0098973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Guo W, Yu H, Zhang L, et al. Effect of hyperoside on cervical cancer cells and transcriptome analysis of differentially expressed genes. Cancer Cell Int. 2019;19(1). doi: 10.1186/s12935-019-0953-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Li F, Yu F-X, Yao S-T, et al. Hyperin extracted from Manchurian rhododendron leaf induces apoptosis in human endometrial cancer cells through a mitochondrial pathway. Asian Pac J Cancer Prev. 2012;13(8):3653–3656. doi: 10.7314/APJCP.2012.13.8.3653 [DOI] [PubMed] [Google Scholar]
- 118.Zhu X, Ji M, Han Y, et al. PGRMC1-dependent autophagy by hyperoside induces apoptosis and sensitizes ovarian cancer cells to cisplatin treatment. Int J Oncol. 2017;50(3):835–846. doi: 10.3892/ijo.2017.3873 [DOI] [PubMed] [Google Scholar]
- 119.Jiang S, Xiong HS, Wu HK, et al. Regulatory effect of hyperoside on proliferation and apoptosis of hepatic carcinoma cell HepG2 via mitochondrial P53 /Caspase signaling pathway. Chin J Immunol. 2018;34:1832–1836. [Google Scholar]
- 120.Wei S, Sun Y, Wang L, et al. Hyperoside suppresses BMP-7-dependent PI3K/AKT pathway in human hepatocellular carcinoma cells. Ann Transl Med. 2021;9(15):1233. doi: 10.21037/atm-21-2980 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kong Y, Sun W, Wu P. Hyperoside exerts potent anticancer activity in skin cancer. Front Biosci. 2020;25(3):463–479. doi: 10.2741/4814 [DOI] [PubMed] [Google Scholar]
- 122.Yang F, Liu M, Li W, et al. Combination of quercetin and hyperoside inhibits prostate cancer cell growth and metastasis via regulation of microRNA-21. Mol Med Rep. 2015;11(2):1085–1092. doi: 10.3892/mmr.2014.2813 [DOI] [PubMed] [Google Scholar]
- 123.Li W, Liu M, Xu Y-F, et al. Combination of quercetin and hyperoside has anticancer effects on renal cancer cells through inhibition of oncogenic microRNA-27a. Oncol Rep. 2014;31(1):117–124. doi: 10.3892/or.2013.2811 [DOI] [PubMed] [Google Scholar]
- 124.Zhang Y, Dong H, Zhang J, et al. Inhibitory effect of hyperoside isolated from Zanthoxylum bungeanum leaves on SW620 human colorectal cancer cells via induction of the p53 signaling pathway and apoptosis. Mol Med Rep. 2017;16(2):1125–1132. doi: 10.3892/mmr.2017.6710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Guon TE, Chung HS. Hyperoside and rutin of Nelumbo nucifera induce mitochondrial apoptosis through a caspase-dependent mechanism in HT-29 human colon cancer cells. Oncol Lett. 2016;11(4):2463–2470. doi: 10.3892/ol.2016.4247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Liu Z, Liu G, Liu X, et al. The effects of hyperoside on apoptosis and the expression of Fas/FasL and survivin in SW579 human thyroid squamous cell carcinoma cell line. Oncol Lett. 2017;14(2):2310–2314. doi: 10.3892/ol.2017.6453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wu L, Yang X-B, Huang ZM, et al. In vivo and in vitro antiviral activity of hyperoside extracted from Abelmoschus manihot (L) medik1. Acta Pharmacol Sin. 2007;28(3):404–409. doi: 10.1111/j.1745-7254.2007.00510.x [DOI] [PubMed] [Google Scholar]
- 128.Li J, Huang H, Zhou W, et al. Anti-hepatitis B virus activities of Geranium carolinianum L. extracts and identification of the active components. Biol Pharm Bull. 2008;31(4):743–747. doi: 10.1248/bpb.31.743 [DOI] [PubMed] [Google Scholar]
- 129.Rehmana S, Ashfaq UA, Ijaz B, et al. Anti-hepatitis C virus activity and synergistic effect of Nymphaea alba extracts and bioactive constituents in liver infected cells. Microb Pathog. 2018;121:198–209. doi: 10.1016/j.micpath.2018.05.023 [DOI] [PubMed] [Google Scholar]
- 130.Jiang Z, Wang J, Liu C, et al. Hyperoside alleviated N-acetyl-para-amino-phenol-induced acute hepatic injury via Nrf2 activation. Int J Clin Exp Pathol. 2019;12(1):64–76. [PMC free article] [PubMed] [Google Scholar]
- 131.Cai Y, Li B, Peng D, et al. Crm1-dependent nuclear export of bach1 is involved in the protective effect of hyperoside on oxidative damage in hepatocytes and CCl4-induced acute liver injury. J Inflamm Res. 2021;14:551–565. doi: 10.2147/JIR.S279249 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Choi J, Kim D-W, Yun N, et al. Protective effects of hyperoside against carbon tetrachloride-induced liver damage in mice. J Nat Prod. 2011;74(5):1055–1060. doi: 10.1021/np200001x [DOI] [PubMed] [Google Scholar]
- 133.Xing H, Fu R, Cheng C, et al. Hyperoside protected against oxidative stress-induced liver injury via the PHLPP2-AKT-GSK-3β signaling pathway in vivo and in vitro. Front Pharmacol. 2020;11. doi: 10.3389/fphar.2020.01065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Sun B, Zhang R, Liang Z, et al. Hyperoside attenuates non-alcoholic fatty liver disease through targeting Nr4A1 in macrophages. Int Immunopharmacol. 2021;94:107438. doi: 10.1016/j.intimp.2021.107438 [DOI] [PubMed] [Google Scholar]
- 135.Guo X, Zhu C, Liu X, et al. Hyperoside protects against heart failure-induced liver fibrosis in rats. Acta Histochem. 2019;121(7):804–811. doi: 10.1016/j.acthis.2019.07.005 [DOI] [PubMed] [Google Scholar]
- 136.Han N, Go J-H, Kim H-M, et al. Hyperoside regulates the level of thymic stromal lymphopoietin through intracellular calcium signalling. Phytother Res. 2014;28(7):1077–1081. doi: 10.1002/ptr.5099 [DOI] [PubMed] [Google Scholar]
- 137.Liu J, Wang Y, Tu Z-C, et al. Bovine β-lactoglobulin covalent modification by flavonoids: effect on the allergenicity and human intestinal microbiota. J Agric Food Chem. 2021;69(24):6820–6828. doi: 10.1021/acs.jafc.1c02482 [DOI] [PubMed] [Google Scholar]
- 138.Ku S, Kim TH, Lee S, et al. Antithrombotic and profibrinolytic activities of isorhamnetin-3-O-galactoside and hyperoside. Food Chem Toxicol. 2013;53:197–204. doi: 10.1016/j.fct.2012.11.040 [DOI] [PubMed] [Google Scholar]
- 139.Song M, Hong M, Lee MY, et al. Selective inhibition of the cytochrome P450 isoform by hyperoside and its potent inhibition of CYP2D6. Food Chem Toxicol. 2013;59:549–553. doi: 10.1016/j.fct.2013.06.055 [DOI] [PubMed] [Google Scholar]
- 140.Zhu W, Xu Y-F, Feng Y, et al. Prophylactic effects of quercetin and hyperoside in a calcium oxalate stone forming rat model. Urolithiasis. 2014;42(6):519–526. doi: 10.1007/s00240-014-0695-7 [DOI] [PubMed] [Google Scholar]
- 141.Sohn E, Kim J, Kim C-S, et al. Osteomeles schwerinae extract prevents diabetes-induced renal injury in spontaneously diabetic torii rats. Evid Based Complement Alternat Med. 2018;2018:1–8. doi: 10.1155/2018/6824215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Liu B, Tu Y, He W, et al. Hyperoside attenuates renal aging and injury induced by D-galactose via inhibiting AMPK-ULK1 signaling-mediated autophagy. Aging (Albany NY). 2018;10(12):4197–4212. doi: 10.18632/aging.101723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Lee D, Shrestha S, Seo WD, et al. Structural and quantitative analysis of antioxidant and low-density lipoprotein-antioxidant flavonoids from the grains of sugary rice. J Med Food. 2012;15(4):399–405. doi: 10.1089/jmf.2011.1905 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Liaudanskas M, Viškelis P, Raudonis R, et al. Phenolic composition and antioxidant activity of Malus domestica leaves. ScientificWorldJournal. 2014;2014:10. doi: 10.1155/2014/306217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wu Y, Zheng L-J, Wu J-G, et al. Antioxidant activities of extract and fractions from receptaculum nelumbinis and related flavonol glycosides. Int J Mol Sci. 2012;13(6):7163–7173. doi: 10.3390/ijms13067163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Yao X, Zhang D-Y, Luo M, et al. Negative pressure cavitation-microwave assisted preparation of extract of Pyrola incarnata Fisch. rich in hyperin, 2′-O-galloylhyperin and chimaphilin and evaluation of its antioxidant activity. Food Chem. 2015;169:270–276. doi: 10.1016/j.foodchem.2014.07.115 [DOI] [PubMed] [Google Scholar]
- 147.Azzouzi HE, Leptidis S, Doevendans PA, et al. HypoxamiRs: regulators of cardiac hypoxia and energy metabolism. Trends Endocrinol Metab. 2015;26(9):502–508. doi: 10.1016/j.tem.2015.06.008 [DOI] [PubMed] [Google Scholar]
- 148.Bećarević M. TNF-alpha and annexin A2: inflammation in thrombotic primary antiphospholipid syndrome. Rheumatol Int. 2016;36(12):1649–1656. doi: 10.1007/s00296-016-3569-1 [DOI] [PubMed] [Google Scholar]
- 149.Li Q, Xie J, Wang B, et al. Overexpression of microRNA-99a attenuates cardiac hypertrophy. PLoS One. 2016;11:e148480. doi: 10.1371/journal.pone.0148480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Boukes GJ, van de Venter M. The apoptotic and autophagic properties of two natural occurring prodrugs, hyperoside and hypoxoside, against pancreatic cancer cell lines. Biomed Pharmacother. 2016;83:617–626. doi: 10.1016/j.biopha.2016.07.029 [DOI] [PubMed] [Google Scholar]
- 151.Verma N, Singh AP, Gupta A, et al. Antidiarrheal potential of standardized extract of rhododendron arboreum smith flowers in experimental animals. India J Pharmacol. 2011;43:689–693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Li S, Zhang Z, Cain A, et al. Antifungal activity of camptothecin, trifolin, and hyperoside isolated from camptotheca acuminata. J Agric Food Chem. 2005;53(1):32–37. doi: 10.1021/jf0484780 [DOI] [PubMed] [Google Scholar]
- 153.Kalegari M, Cerutti ML, Macedo-Júnior SJ, et al. Chemical composition and antinociceptive effect of aqueous extract from Rourea induta Planch. leaves in acute and chronic pain models. J Ethnopharmacol. 2014;153(3):801–809. doi: 10.1016/j.jep.2014.03.045 [DOI] [PubMed] [Google Scholar]
- 154.Hu J, Wang Z, Guo -Y-Y, et al. A role of periaqueductal grey NR2B-containing NMDA receptor in mediating persistent inflammatory pain. Mol Pain. 2009;5:71. doi: 10.1186/1744-8069-5-71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Shi N. Effects of hyperin on mice with acute inflammatory pain. Drug Eval Res. 2016;39:768–771. [Google Scholar]
- 156.Wu C, Chai J, Zhang W. Study on the pharmacokinetics of hyperoside in rats. J Liaoning Univ TCM. 2016;18:23–26. [Google Scholar]
- 157.Guo AI, Huang ZM, Liu CX, et al. Pharmacokinetics study of hyperoside in rats. Chin J Exp Trad Med Formulae. 2013;19:157–161. [Google Scholar]
- 158.Tan A, Lin P, Zhang F. Determination of hyperoside in rat plasma after intravenous administration by UPLC-MS. J Chin Pharm Sci. 2013;22:516–520. doi: 10.5246/jcps.2013.06.075 [DOI] [Google Scholar]
- 159.Ai G, Huang ZM, Wang DW, Zhang HD. Toxicity of hyperoside after long-term oral administration in Wistar rats. Chin J New Drugs. 2012;21:2811–6, 2828. [Google Scholar]
- 160.Ai G, Wang D, Huang Z, Zhang H. Long-term toxicity of hyperoside in Beagle dogs. Chin J New Drugs. 2015;24:1641–1647. [Google Scholar]
- 161.Ai G, Huang Z, Wang D, et al. Study on toxicity of hyperoside in rat embryo-fetal development. China J Chin Mater Med. 2012;37(16):2452–2455. [PubMed] [Google Scholar]
- 162.Ren X. Simulation determination of hyperoside and hypericin in hypericum perforatum L. and Related drugs by HPLC. Trad Chin Drug Res Clin Pharmacol. 2019;30:1374–1378. [Google Scholar]
- 163.Wu G, Yan S, Yan L. Determination of hyperoside, isoquercitrin and quercitrin in rose petal by HPLC. Mod Chin Med. 2020;22:1607–1610. [Google Scholar]
- 164.Zhang S. Determination of chlorogenic acid, hesperidin, hyperin and benzoic acid in jianer qingjie solution by HPLC. China Pharmacist. 2020;23:1429–1431. [Google Scholar]
- 165.Jia Y. Qualitative and quantitative study of Chaijin Jieyu granules. Cent South Pharm. 2018;16:1604–1608. [Google Scholar]
- 166.Zhu X. Determination of hyperoside in the extract of senecionis scandens herba and qianbai biyan capsule by HPLC and its pharmacokinetics in rats. Shandong Chem Indus. 2019;48:71–74. [Google Scholar]