Abstract
Background:
In Canada, more than 2 million people live with osteoporosis, a disease that increases the risk for fractures, which result in excess mortality and morbidity, decreased quality of life and loss of autonomy. This guideline update is intended to assist Canadian health care professionals in the delivery of care to optimize skeletal health and prevent fractures in postmenopausal females and in males aged 50 years and older.
Methods:
This guideline is an update of the 2010 Osteoporosis Canada clinical practice guideline on the diagnosis and management of osteoporosis in Canada. We followed the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework and quality assurance as per Appraisal of Guidelines for Research and Evaluation (AGREE II) quality and reporting standards. Primary care physicians and patient partners were represented at all levels of the guideline committees and groups, and participated throughout the entire process to ensure relevance to target users. The process for managing competing interests was developed before and continued throughout the guideline development, informed by the Guideline International Network principles. We considered benefits and harms, patient values and preferences, resources, equity, acceptability and feasibility when developing recommendations; the strength of each recommendation was assigned according to the GRADE framework.
Recommendations
The 25 recommendations and 10 good practice statements are grouped under the sections of exercise, nutrition, fracture risk assessment and treatment initiation, pharmacologic interventions, duration and sequence of therapy, and monitoring. The management of osteoporosis should be guided by the patient’s risk of fracture, based on clinical assessment and using a validated fracture risk assessment tool. Exercise, nutrition and pharmacotherapy are key elements of the management strategy for fracture prevention and should be individualized.
Interpretation:
The aim of this guideline is to empower health care professionals and patients to have meaningful discussions on the importance of skeletal health and fracture risk throughout older adulthood. Identification and appropriate management of skeletal fragility can reduce fractures, and preserve mobility, autonomy and quality of life.
Fracture risk increases with advancing age, as a result of declining skeletal strength and increased risk of falling. In Canada, more than 2 million people live with osteoporosis.1 Every year in Canada, about 150 people per 100 000 suffer a hip fracture, which is considered among the most serious fractures associated with osteoporosis. Fractures lead to increased morbidity, excess mortality, decreased quality of life and loss of autonomy.2 Although osteoporosis is often considered a disease of older females, males are remarkably underevaluated and undertreated for the condition despite suffering worse outcomes following fracture,3 highlighting the importance of providing guidance in males.
Osteoporosis, defined as a bone mineral density (BMD) of 2.5 or more standard deviations below the peak bone mass (i.e., T-score ≤ −2.5), is an indicator of increased fracture risk; this risk is modified by age, sex and other factors.4 A clinical diagnosis of osteoporosis can be made in people aged 50 years and older if they have sustained a low-trauma hip, vertebral, humerus or pelvic fracture after the age of 40 years, or if they have an absolute fracture risk of 20% or more over the next 10 years, using a fracture risk assessment tool (FRAX or the Canadian Association of Radiologists and Osteoporosis Canada [CAROC]).5–7
Advances in risk assessment and nonpharmacologic and pharmacologic management warranted an update to the Osteoporosis Canada 2010 clinical practice guideline for the diagnosis and management of osteoporosis in Canada.8
Scope
Developed by the Osteoporosis Canada 2023 Guideline Update Group, this updated guideline is intended to assist Canadian primary health care professionals in screening community-dwelling postmenopausal females and males aged 50 years and older for the presence of risk factors for osteoporosis and fractures, and in providing interventions to optimize skeletal health and fracture prevention. Throughout the guideline, we use “females” and “males” to refer to biological sex.
The focus of the guideline recommendations for treatment is on people with primary osteoporosis. The guideline does not address the complexity of patients with cancer, serious illnesses or active conditions that can induce bone loss (e.g., hyperparathyroidism, multiple myeloma, or newly diagnosed rheumatoid arthritis or other inflammatory conditions requiring high-dose glucocorticoid therapy). Conditions known to cause secondary osteoporosis should be sought and referral to specialists with appropriate expertise for co-management considered (Appendix 1, Supplementary Table 5, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content).
Falls are a leading cause of fractures among older adults in Canada.9 Because detailed recommendations for fall prevention were beyond the scope of this guideline, we refer the reader to guidance from the Canadian Task Force on Preventive Health Care on falls prevention.10 Specific recommendations on preventing fracture in people who reside in long-term care have been published.11
Recommendations
We formulated 25 recommendations and 10 good practice statements (GPS) using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework (Table 1). The recommendations are grouped under the sections of exercise, nutrition, fracture risk assessment and treatment initiation, pharmacologic interventions, duration and sequence of therapy, and monitoring.12,13
Table 1:
| Criteria | Interpretation by patients | Interpretation by health care providers | Interpretation by policy-makers |
|---|---|---|---|
| Strong recommendation for or against | |||
| Desirable consequences CLEARLY OUTWEIGH the undesirable consequences in most settings (or vice versa) | Most individuals in this situation would want the recommended course of action, and only a small proportion would not. | Most individuals should receive the recommended course of action. Adherence to this recommendation according to the guideline could be used as a quality criterion or performance indicator. Formal decision aids are not likely to be needed to help individuals make decisions consistent with their values and preferences. |
Recommendation can be adopted as policy in most situations. |
| Conditional recommendation for or against | |||
| Desirable consequences PROBABLY OUTWEIGH undesirable consequences in most settings (or vice versa) | Most individuals in this situation would want the suggested course of action, but many would not. | Clinicians should recognize that different choices will be appropriate for each person and that clinicians must help each person arrive at a management decision consistent with the person’s values and preferences. Decision aids may be useful to help people make decisions consistent with their values and preferences. |
Policy-making will require substantial debate and involvement of various stakeholders. |
| Good practice statement | |||
| |||
Note: GRADE = Grading of Recommendations Assessment, Development and Evaluation.
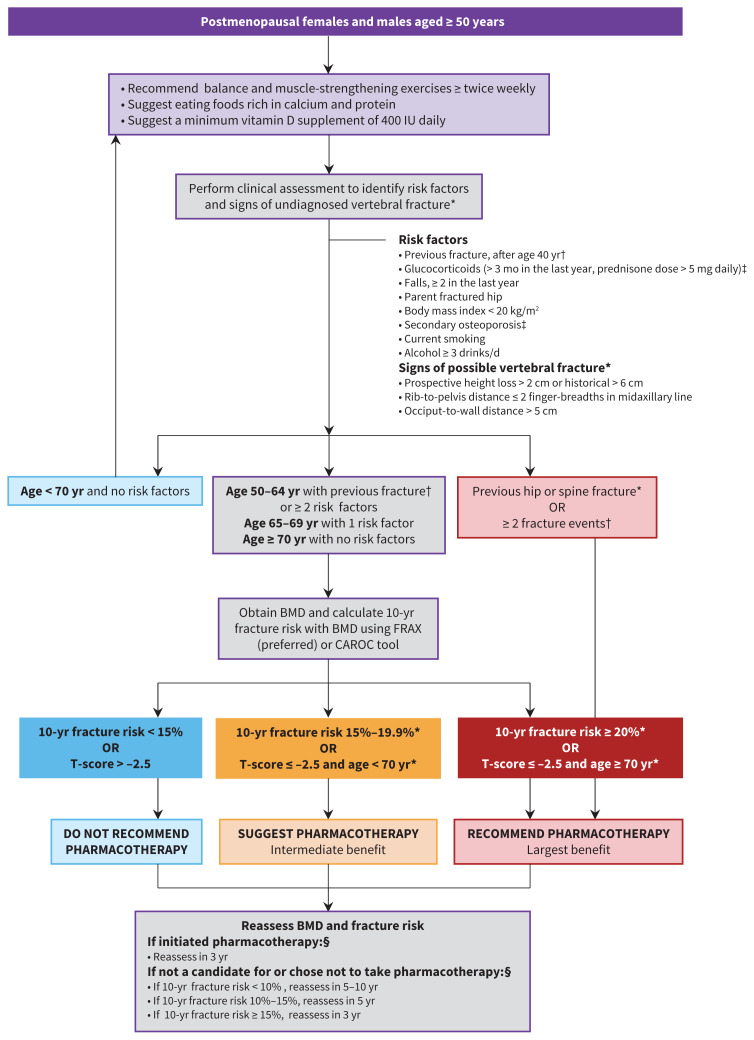
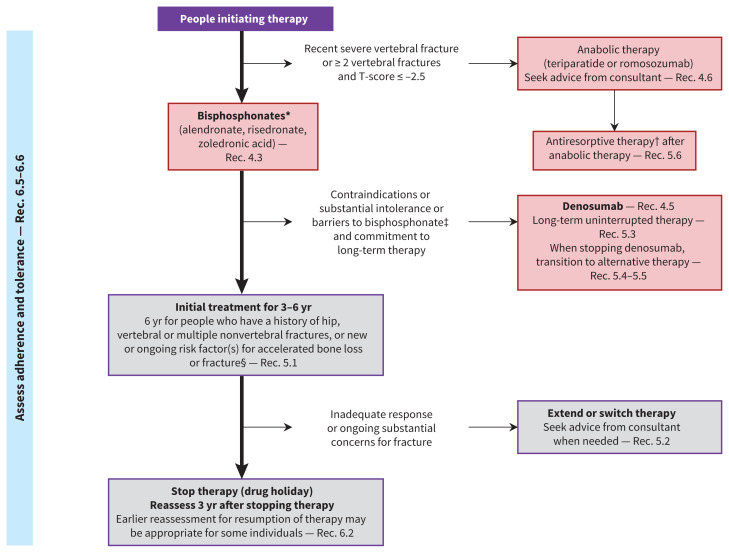
Figure 1 describes an integrated approach to bone health and fracture prevention in postmenopausal females and in males aged 50 years and older, and Figure 2 presents an approach to pharmacologic treatment, when required. Appendix 2 (available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content) provides evidence-to-decision tables (starting on p. 50) that explain our rationales for strength of recommendations and certainty of evidence.
Figure 1:
Integrated approach to the management of bone health and fracture prevention in postmenopausal females and males aged 50 years and older. See Appendix 1, Supplementary Tables 1–6 (available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content) for more information on exercise and nutrition, secondary causes of osteoporosis and vertebral fracture assessment. Note: BMD = bone mineral density. *Consider lateral spine imaging to identify vertebral fracture(s). Finding of undiagnosed vertebral fracture(s) can guide appropriate choice and duration of therapy (Appendix 1, Supplementary Table 6). †Fractures that occur after the age of 40 years, in the setting of low trauma. Fractures of the hands, feet and craniofacial bones are not considered osteoporotic fractures. ‡Conditions known to cause secondary osteoporosis (Appendix 1, Supplementary able 5); consider referral to specialists with expertise for co-management. §Should be reassessed earlier if patient develops secondary causes (Appendix 1, Supplementary Table 5), new fracture or other risk factor for rapid bone loss.
Figure 2:
Approach to pharmacotherapy to prevent fractures. Note: Rec. = recommendation (see Tables 4, 6 and 7 for full recommendations). * Menopausal hormone therapy is a suggested alternative for females younger than 60 years or within 10 years after menopause who prioritize alleviation of substantial menopausal symptoms (Rec. 4.4). †Antiresorptive therapy includes bisphosphonates (alendronate, risedronate and zoledronic acid), denosumab, raloxifene and menopausal hormone therapy. ‡Raloxifene is suggested rather than no treatment for females who have contraindications or substantial intolerance to, or who choose not to take, other suggested therapies) (Rec. 4.7). §See Figure 1 for list of risk factors and Appendix 1, Supplementary Table 5, for causes of secondary osteoporosis.
We considered certainty of evidence, benefits and harms of interventions on clinical outcomes in the decision-making process. Males were under-represented in the clinical evidence of certain interventions and outcomes; we therefore report the strength of the recommendations based on the certainty of the evidence separately for males and females, where appropriate. All recommendations and GPS were informed by input from primary care physicians and patient partners, and include their values and preferences. Although for this guideline, we did not carry out cost-effectiveness analyses, we have considered cost-effectiveness information, when available, in the development of the recommendations and documented this in the specific evidence-to-decision tables.14–16
In adults older than 40 years, osteoporosis can result in fractures. Often referred to as fragility or osteoporosis-related fractures, they occur as a result of a fall from standing height or when the force applied to the bone is judged to be insufficient to fracture normal bone.17 Fractures of the hip, vertebra, humerus and distal forearm are categorized as major osteoporotic fractures, whereas fractures of the hands, feet and craniofacial bones are not considered osteoporotic fractures.1 Vertebral fractures, whether clinically silent or symptomatic, are associated with increased risk of fractures at all skeletal sites and can be identified or verified by radiologic imaging.18,19
Exercise
The recommendations on exercise for fall and fracture prevention for postmenopausal females and males aged 50 years and older are outlined in Table 2 (also see Appendix 1, Supplementary Table 1: key points on exercise and definitions). Evidence-to-decision tables are in Appendix 2 (p. 50).
Table 2:
Recommendations on exercise and nutrition for fall and fracture prevention in postmenopausal females and males aged 50 years and older*
| Recommendations | Strength of recommendation and certainty of evidence† |
|---|---|
| 1. Exercise ‡ | |
| 1.1. We recommend balance and functional training ≥ twice weekly to reduce the risk of falls. Remark: Increase difficulty, pace, frequency, volume (sets, reps) or resistance over time. Balance exercises challenge aspects of balance, such as:
|
Strong recommendation; moderate-certainty evidence |
| 1.2. We suggest progressive resistance training ≥ twice weekly, including exercises targeting abdominal and back extensor muscles. Remark: Resistance training involves exercises in which major muscle groups (e.g., upper and lower extremities, chest, shoulders, back) work against resistance (e.g., squats, lunges and push-ups). Increase volume (e.g., sets, reps, weight), frequency or difficulty to achieve progressive overload. Many resistance-training exercises would be considered functional exercises. |
Conditional recommendation; low-certainty evidence |
| 1.3. We suggest that people who want to participate in other activities (e.g., walking, impact exercise, yoga, Pilates) for enjoyment or other benefits be encouraged to do them, if they can be done safely or modified for safety. Other activities should be encouraged in addition to, but not instead of, balance, functional and resistance training. Remark: Encourage a variety of types and intensities of physical activity in accordance with the Canadian 24-Hour Movement Guidelines (https://csepguidelines.ca), such as getting ≥ 150 min of moderate to vigorous physical activity per week, but prioritize balance, functional and resistance training. If participating in impact exercise, progress to moderate-impact (e.g., running, racquet sports, skipping) or high-impact (e.g., drop or high vertical jumps) exercise only if appropriate for fracture risk or physical fitness level; safety or efficacy of impact exercise is uncertain in people at high fracture risk (e.g., history of spine fracture or 10-yr fracture risk for major osteoporotic fracture of ≥ 20% calculated by FRAX or CAROC fracture risk assessment tools). |
Conditional recommendation; very low-certainty evidence |
| 1.4. Activities that involve rapid, repetitive, sustained, weighted or end range-of-motion twisting or flexion of the spine may need to be modified, especially in people at high risk of fracture. | Good practice statement |
| 1.5. When available, seek advice from exercise professionals who have training on osteoporosis for exercise selection, intensity and progression, and activity modification, especially after recent fracture or if there is high risk of fracture. When not available, refer to Osteoporosis Canada resources.§ | Good practice statement |
| 2. Nutrition ¶ | |
| 2.1. For people who meet the recommended dietary allowance for calcium with a variety of calcium-rich foods, we suggest no supplementation to prevent fractures. Remark: Health Canada’s recommended dietary allowance for calcium is 1000 mg/d (males aged 51–70 yr) and 1200 mg/d (females > 50 yr and males > 70 yr). |
Conditional recommendation; moderate-to-high-certainty evidence |
| 2.2. We suggest following Health Canada’s recommendation on vitamin D for bone health. Remark: Health Canada’s recommended dietary allowance for vitamin D is 600 IU/d (age 51–70 yr) and 800 IU/d (age > 70 yr) for males and females. Given that it is difficult to achieve this level of intake, as few foods contain vitamin D, Health Canada recommends adults older than 50 yr take a vitamin D supplement of 400 IU daily, in addition to consuming vitamin D–rich foods, to achieve the recommended dietary allowance. For people at risk of vitamin D deficiency, additional supplemental vitamin D should be provided.** |
Conditional recommendation; high-certainty evidence |
| 2.3. For people who follow Canada’s Food Guide (food-guide.canada.ca), we suggest no supplementation of protein, vitamin K or magnesium to prevent fractures. | Conditional recommendation; low-certainty evidence (protein, vitamin K), very low-certainty evidence (magnesium) |
| 2.4. For people initiating pharmacotherapy, it is good practice to individualize intake of calcium and vitamin D. Although participants in most pharmacotherapy trials received a minimum of 400 IU/d of vitamin D and up to 1000 mg/d of calcium supplements, food sources or supplementation should be individualized according to risk factors for insufficiency.** | Good practice statement |
Note: CAROC = Canadian Association of Radiologists and Osteoporosis Canada tool, FRAX = Fracture Risk Assessment Tool.
Integrated approach is shown in Figure 1.
See Table 1 for definitions.
See Appendix 1, Supplementary Table 1 (available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content) for key points on exercise and definitions.
See osteoporosis.ca/exercise/
See Appendix 1, Supplementary Table 2, for key points on nutrition.
See Appendix 1, Supplementary Table 4 for risk factors for vitamin D insufficiency.
High-quality evidence from a Cochrane systematic review shows that functional and balance training in adults older than 50 years can reduce the number of falls (rate ratio 0.76, 95% confidence interval [CI] 0.70–0.80; 41 studies, 7290 participants; high-certainty evidence) and the number of people who fall (rate ratio 0.87, 95% CI 0.82–0.91; 38 studies, 8288 participants; high-certainty evidence). Some evidence exists that functional and balance training may reduce the number of people who have fall-related fractures (rate ratio 0.44, 95% CI 0.25–0.76; 7 studies, 2139 participants; low-certainty evidence), and may improve quality of life (mean difference 2.48 points, 95% CI 1.31–3.64; 8 studies, 854 participants; low-certainty evidence) and physical functioning.20
A systematic review of randomized controlled trials (RCTs) suggests that resistance training may improve quality of life (standardized mean difference 0.75, 95% CI 0.54–0.95; 8 studies, 421 participants; moderate-certainty evidence), physical functioning and BMD, and reduce mortality.21 When combined with functional and balance training in older adults, resistance training can reduce the number of falls and number of people who fall.
Evidence from a systematic review of RCTs shows that in people with hyperkyphosis, exercises targeting abdominal, back extensor and shoulder muscles may result in small improvements in spinal curvature, quality of life, physical functioning and back extensor strength;22 however, effects on spinal curvature may not occur with fixed kyphosis (e.g., from vertebral fracture).
Observational studies suggest that walking may reduce mortality in older adults,23 but effects on fractures, falls, quality of life, physical functioning or harms in people at risk of fracture are unknown, or effects are uncertain.24 A systematic review suggests that impact exercise may improve BMD and physical functioning, and may reduce mortality,25 but the benefit of this type of exercise in isolation is uncertain, as it is often combined with resistance exercise or other exercise.
We performed systematic reviews on the effects of yoga and Pilates on health-related outcomes and found that these types of exercise may improve physical functioning and quality of life in older adults, but effects on BMD, falls and fractures are uncertain, and evidence for people at risk of fracture is limited.
Males were underrepresented in the evidence for some of the exercise outcomes. We chose not to downgrade the strength of the evidence for indirectness when applied to males because there was no reason to expect that the direction or magnitude of the effects on falls, fractures, physical functioning, quality of life or adverse events would be different in males.
Nutrition
Table 2 summarizes the recommendations on nutrition for postmenopausal females and males aged 50 years and older to prevent falls and fracture (Appendix 1, Supplementary Table 2: key points on nutrition). Evidence-to-decision tables are in Appendix 2 (p. 114).
For people who are consuming a balanced diet (https://food-guide.canada.ca/en/food-guide-snapshot/) and not receiving pharmacotherapy for osteoporosis, supplementation with calcium, vitamin D and protein is likely to have little to no beneficial or detrimental effect on fractures (Appendix 1, Supplementary Table 2). In systematic reviews and recent individual studies on calcium and vitamin D use, conducted predominantly in people not receiving osteoporosis pharmacotherapy and without deficiency, supplementation was associated with small, unimportant benefits in fracture reduction regardless of fracture site.26,27 Minimal decreases in falls were observed for vitamin D supplementation alone or in combination with calcium.27,28 Evidence for supplementation with magnesium and vitamin K in relation to bone health is limited (Appendix 2, p. 161).
Reduction in hip fracture rates with protein supplementation may be minimal.29,30 The evidence is uncertain because the studies assessed dietary rather than supplemental levels, often at or above the recommended level (0.8–1.3 g protein/kg body weight/d) and in people who were well nourished. Whether protein source matters with respect to bone health is unclear, because most participants had low intakes of plant protein. The effect on functional outcomes with higher dietary protein intakes was minimal.31
Fracture risk assessment
The recommendations on fracture risk assessment are presented in Table 3, with evidence-to-decision tables in Appendix 2 (p. 182). Figure 1 shows the clinical application of these recommendations.
Table 3:
Fracture risk assessment and treatment initiation in postmenopausal females and males aged 50 years and older*
| Recommendations | Strength of recommendation and certainty of evidence† |
|---|---|
| 3. Fracture risk assessment and treatment initiation | |
| 3.1. A clinical assessment for osteoporosis and fracture includes identifying risk factors and assessing for signs of undiagnosed vertebral fracture(s).‡ | Good practice statement |
| 3.2. We suggest that the Canada-specific FRAX tool is the preferred tool for fracture risk estimation. Remark: CAROC is an alternative fracture risk assessment tool. FRAX and CAROC may underestimate fracture risk in the presence of specific risk factors such as recency of fractures, recurrent falls, other comorbidities or very low BMD at the lumbar spine and total hip sites.§ |
Conditional recommendation; moderate-certainty evidence |
3.3. We suggest BMD testing in postmenopausal females and males who
|
Conditional recommendation; low-certainty evidence (females), very low-certainty evidence (males) |
3.4. We suggest vertebral imaging with lateral spine radiograph or vertebral fracture assessment in postmenopausal females and males without known vertebral fractures who
|
Conditional recommendation; moderate-certainty evidence (females), low-certainty evidence (males) |
3.5. We recommend initiating pharmacotherapy in postmenopausal females and males aged ≥ 50 yr who
|
Strong recommendation; high-certainty evidence (females: a and c), moderate-certainty evidence (females: b; males: a, b and c) |
3.6. We suggest initiating pharmacotherapy in postmenopausal females and males aged ≤ 50 yr who
|
Conditional recommendation; moderate-certainty evidence (females), very low-certainty evidence (males) |
3.7. We suggest that for individuals who do not meet the threshold for initiating pharmacotherapy or choose not to initiate therapy, BMD testing can be repeated at:
|
Conditional recommendation; low-certainty evidence (females), very low-certainty evidence (males) |
| 3.8. We recommend that postmenopausal females and males aged ≥ 50 yr presenting with a recent fracture have access to a Fracture Liaison Service to improve identification and treatment initiation for osteoporosis.** | Strong recommendation; high-certainty evidence |
Note: BMD = bone mineral density, CAROC = Canadian Association of Radiologists and Osteoporosis Canada, FRAX = Fracture Risk Assessment Tool.
Integrated approach is shown in Figure 1.
See Table 1 for definitions.
For list of risk factors, see Figure 1 and Appendix 1, Supplementary Table 5 (causes of secondary osteoporosis) and Supplementary Table 6 (Clinical Assessment of Vertebral Fractures), available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content
For information on fracture risk assessment tools, see www.osteoporosis.ca
See Appendix 1, Supplementary Table 6 (clinical assessment of vertebral fractures).
See https://fls.osteoporosis.ca/for list of Fracture Liaison Services in Canada.
We suggest an approach based on the assessment of age and the presence of clinical risk factors (i.e., a “targeted” approach) for identifying people who should undergo BMD measurement (low-certainty evidence in postmenopausal females aged 50–64 years; moderate-certainty evidence in females aged ≥ 65 years;32–34 very low-certainty evidence in males).14,32,35 This strategy delays BMD testing in most people until age 70 years and allows for appropriate categorization of those at high fracture risk with the fracture risk assessment tools available in Canada.32 Patient partners stated that providing guidance in identifying both females and males for appropriate BMD testing based on the presence of clinical risk factors was of high importance.
In Canada, validated 10-year major osteoporotic fracture assessment tools include FRAX and CAROC. FRAX’s performance is as good as or slightly better than that of other tools,36 and results in better fracture risk classification than CAROC (net reclassification improvement in risk category by 2% and in treatment recommendation by 1%).37 We suggest the use of the Canada-specific FRAX tool as the preferred tool for fracture risk estimation; however, this recommendation is conditional, owing to the moderate certainty of the evidence and indirectness of evidence in males.37
Treatment initiation
Recommendations on initiating treatment are provided in Table 3, with evidence-to-decision tables in Appendix 2 (p. 190 and p. 314).
No consensus exists on the optimal approach to setting a treatment threshold.38 In setting pharmacotherapy initiation thresholds, we considered the burden of fractures in the Canadian population, how fracture outcomes were affected by baseline fracture risk, the efficacy of pharmacotherapy on fracture outcomes, the importance of identifying those at high fracture risk to prevent the most fractures and the importance of limiting overtreatment in those at lower risk, from population and patient perspectives.38
Previous fracture of the vertebra (clinical or documented on imaging) or the hip, and more than 1 fracture, indicate high risk for future fractures.8 Randomized controlled trials provide a moderate-to-high certainty of evidence showing important benefits of fracture reduction with pharmacotherapy in people with osteoporosis (defined as T-score ≤ −2.5 or history of previous fracture), with a risk reduction of about 50% for vertebral, 30% for hip, and 20% for nonvertebral fractures after 3 years of treatment.39–41 In addition, given observational data from a large clinical registry, an intervention threshold of 20% for 10-year major fracture risk (as measured by FRAX or CAROC) was also selected (as a conditional recommendation), as this strategy was highly ranked in terms of number of fractures prevented among females aged 50 years and older and the number of females treated (to limit overtreatment).38 We did not rate down the certainty of the evidence in males, as evidence suggests that there are no differences in treatment benefits and harms by sex.39,41–44
Fracture Liaison Service programs (https://fls.osteoporosis.ca/), which provide postfracture investigation and treatment initiation and are currently in place in a few Canadian jurisdictions, increase the appropriate use of fracture risk assessment and antifracture treatment, and are cost-effective.45 We recommend that postmenopausal females and males aged 50 years and older who present with a recent fracture have access to a Fracture Liaison Service to improve identification and treatment initiation for osteoporosis.
Pharmacologic interventions
Table 4 presents the recommendations on pharmacotherapy in those initiating therapy (additional information in Table 5), with an algorithm for clinical application in Figure 2. Evidence-to-decision tables are in Appendix 2 (p. 323).
Table 4:
Pharmacologic interventions for postmenopausal females and males aged 50 years and older for whom anti-osteoporosis treatment is recommended or suggested*
| Recommendations | Strength of recommendation and certainty of evidence† |
|---|---|
| 4. Pharmacologic interventions | |
| 4.1. Before initiating pharmacotherapy, good practice includes assessing for secondary causes of osteoporosis, and for potential limitations when considering specific osteoporosis pharmacotherapy.‡ | Good practice statement |
| 4.2. When uncertainty exists about fracture risk or treatment, such as possible secondary causes of osteoporosis, comorbidities that complicate management and important adverse effects from pharmacotherapy, good practice includes seeking advice from a consultant with expertise in osteoporosis (e.g., family physician, general internist, endocrinologist, rheumatologist, geriatrician). | Good practice statement |
| 4.3. For people who meet criteria for initiation of pharmacotherapy, we recommend bisphosphonates (alendronate, risedronate or zoledronic acid). Remark: Oral bisphosphonates may be preferred, as drug coverage, costs and access to an infusion centre may be barriers to zoledronic acid. |
Strong recommendation; high-certainty evidence (females), moderate-certainty evidence (males) |
| 4.4. For postmenopausal females aged < 60 yr or within 10 yr of menopause initiating pharmacotherapy who prioritize alleviation of substantial menopausal symptoms, we suggest menopausal hormone therapy as an alternative option to bisphosphonate therapy. Remark: The choice will also depend on individualized risks of menopausal hormone therapy, which consists of an estrogen dose equivalent of conjugated equine estrogens of 0.625 mg daily (plus progestogen in those with an intact uterus). |
Conditional recommendation; moderate-certainty evidence |
| 4.5. For people meeting criteria for initiation of pharmacotherapy who have contraindications, substantial intolerance or barriers to bisphosphonates, we suggest denosumab. Remark: Despite the benefits of denosumab, a careful assessment of indications is required because of the risk of rapid bone loss and vertebral fractures with delayed dosing or discontinuation of denosumab. It is important to communicate the need for commitment to long-term therapy and the need to transition to alternative antiresorptive therapy if discontinuing denosumab. Denosumab may be preferred when there is a high burden of oral medications, gastrointestinal intolerance, contraindication to oral bisphosphonates or barriers to accessing intravenous zoledronic acid. |
Conditional recommendation; high-certainty evidence (females), moderate-certainty evidence (males) |
| 4.6. For people meeting criteria for initiation of pharmacotherapy who have had a recent severe vertebral fracture, or > 1 vertebral fracture, AND a T-score ≤ −2.5, we suggest seeking advice from a consultant with expertise in osteoporosis about anabolic therapy (teriparatide or romosozumab). Remark: “Recent fracture” is defined as a fracture occurring within the past 2 yr, and “severe vertebral fracture” as vertebral body height loss of > 40%. Clinicians may seek advice from radiologists to clarify the degree of severity of the vertebral fracture. The choice of anabolic therapy may depend on affordability and feasibility of injection schedule. |
Conditional recommendation; high-certainty evidence (females), moderate-certainty evidence (males) |
| 4.7. For postmenopausal females initiating pharmacotherapy who have contraindications or substantial intolerance to, or who choose not to take other suggested therapies, we suggest raloxifene rather than no treatment. Remark: Raloxifene should be used only in those who are not at high risk of venous thromboembolism. |
Conditional recommendation; moderate-certainty evidence |
Integrated approach shown in Figure 2. Recommendations on criteria for initiating therapy are in Table 3.
See Table 1 for definitions.
See Appendix 1, Supplementary Table 3 (biochemical testing) and Supplementary Table 5 (causes of secondary osteoporosis), available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content
Table 5:
Medications for the treatment of osteoporosis*
| Drug | Route and dosing | Potential adverse effects | Contraindications | Other considerations | Cost† |
|---|---|---|---|---|---|
| Antiresorptive agents | |||||
| Bisphosphonates | |||||
| Alendronate | Oral: 70 mg weekly or 10 mg daily |
|
|
|
$ |
| Risedronate | Oral: 35 mg weekly or1 150 mg monthly or 5 mg daily |
|
|
|
$ |
| Zoledronic acid | Intravenous: 5 mg yearly |
|
|
|
$$ |
| RANK-ligand inhibitor (monoclonal antibody) | |||||
| Denosumab | Subcutaneous: 60 mg every 6 mo |
|
|
|
$$$ |
| Hormonal therapy | |||||
| Menopausal hormonal therapy | Multiple regimens |
|
|
|
$–$$ |
| Raloxifene (SERM) | Oral: 60 mg daily |
|
|
|
$ |
| Anabolic agents | |||||
| Parathyroid hormone analog | |||||
| Teriparatide | Subcutaneous: 20 μg daily for 24 mo |
|
|
|
$$$$$ |
| Sclerostin inhibitor (monoclonal antibody) | |||||
| Romosozumab | Subcutaneous: 210 mg monthly for 12 mo |
|
|
|
$$$$$ |
Note: AFF = atypical femoral fracture, ALP = alkaline phosphatase, CrCl = creatinine clearance, CVD = cardiovascular disease, GI = gastrointestinal, MSK = musculoskeletal, ONJ = osteonecrosis of the jaw, RANK = receptor activator of nuclear factor κ-β, SERM = selective estrogen receptor modulator, VTE = venous thromboembolism.
Information in this table is not meant to be exhaustive and should not replace complete details provided by drug monographs (available in the Compendium of Pharmaceuticals and Specialties at myrxtx.ca). Further information on some medications available in selected references.15,43,46–51
Relative cost.
Pharmacologic therapies are classified as antiresorptive therapy, which inhibits osteoclastic activity, and anabolic therapy, which stimulates new bone formation. Antiresorptive therapy includes bisphosphonates (alendronate, risedronate and zoledronic acid), denosumab, raloxifene and menopausal hormone therapy; anabolic therapy includes teriparatide and romosozumab (Table 5).
There was evidence of moderate-to high certainty in pharmacotherapy effects.39 Although most studies were performed in postmenopausal females, the evidence available in males with primary or hypogonadal osteoporosis shows similar effects to females on fractures with bisphosphonates and denosumab; therefore, the evidence from females was used for males but rated as moderate certainty from some indirectness.39,41,42
Overall, bisphosphonate therapy for 3 years results in 20–30 fewer vertebral, 10 fewer nonvertebral, and 3 fewer hip fractures per 1000 people than no treatment.39,41 Compared with placebo, there may be very few harms with short-term (≤ 3 yr) use of oral bisphosphonate therapy, including gastrointestinal events such as esophagitis and ulcers (< 1% difference), and transient flu-like symptoms with zoledronic acid infusions, as well as very uncertain evidence for an increased risk of atrial fibrillation.43 The benefits of denosumab are similar to those of zoledronic acid,39 but there may be greater harms with denosumab: 7% more serious adverse events (such as infections requiring hospital admission) than with placebo47 and 14% and 7% more when compared with alendronate and zoledronic acid, respectively. 15 Delayed dosing or discontinuation of denosumab is associated with rapid bone loss and may lead to vertebral fractures.52
In females with higher risk of fractures (e.g., recent severe vertebral fracture, or > 1 vertebral fracture and T-score ≤ −2.5),53 there is high-certainty evidence that anabolic therapy (teriparatide or romosozumab) results in greater reductions in vertebral, nonvertebral and hip fractures than bisphosphonates (35, 18 and 5 fewer, respectively, per 1000 people).39,43 This evidence is indirect in males and therefore of moderate certainty. Stopping anabolic treatment without subsequent antiresorptive therapy risks the loss of bone density gains.54,55 For most people, the downsides of teriparatide, romosozumab or denosumab (such as injection schedules, the risks associated with and need for transition therapy when stopping the medication, and costs) probably outweigh the benefits compared with bisphosphonates.56 However, for people at higher risk of fractures, the benefits may outweigh these downsides.
Duration and sequence of therapy
Recommendations on duration and sequence of therapy are outlined in Table 6. Evidence-to-decision tables are in Appendix 2 (p. 330).
Table 6:
Duration and sequence of therapy in postmenopausal females and males aged 50 years and older who receive anti-osteoporosis medications*
| Recommendations | Strength of recommendation and certainty of evidence† |
|---|---|
| 5. Duration and sequence of therapy | |
| 5.1. For people on bisphosphonates, we suggest initial therapy for a duration of 3–6 yr. Remark: Six years of therapy is appropriate for individuals with a history of hip, vertebral or multiple nonvertebral fractures, or new or ongoing risk factor(s) for accelerated bone loss or fracture.‡ When using zoledronic acid, dosing less frequently than annually may be appropriate.46 |
Conditional recommendation; low-certainty evidence |
| 5.2. When there is inadequate response or ongoing substantial concern for fracture during bisphosphonate therapy, good practice includes extending or switching therapy, reassessing for secondary causes and seeking advice from a consultant with expertise in osteoporosis, if needed. Remark: Inadequate response to treatment should be considered when > 1 fracture or substantial bone density decline (e.g., ≥ 5%) occurs despite adherence to an adequate course of treatment (typically > 1 yr). However, fractures or bone density decline during therapy do not always indicate inadequate response to treatment (e.g., secondary causes of osteoporosis, falls, BMD imprecision errors). |
Good practice statement |
| 5.3. For people on denosumab, we suggest long-term uninterrupted therapy. Remarks: The injection schedule of every 6 mo should not be delayed by more than 1 mo because of the risk of rapid bone loss and vertebral fractures. Duration of therapy may be assessed after 6–10 yr and may be dependent on previous bisphosphonate therapy and individualized risk for atypical femoral fracture and osteonecrosis of the jaw. |
Conditional recommendation; low-certainty evidence |
| 5.4. For people discontinuing denosumab after ≤ 4 doses, we suggest transitioning to a bisphosphonate 6 mo after the last dose of denosumab to reduce the risk of rapid bone loss. We suggest bisphosphonate therapy for 1 yr and then reassessing the need for ongoing transition therapy. Remark: Discontinuation of denosumab may be appropriate for people for whom treatment with denosumab is no longer warranted or for those who develop intolerance or contraindications to denosumab. |
Conditional recommendation; low-certainty evidence |
| 5.5. For people discontinuing denosumab after ≥ 5 doses where the risk of rapid bone loss or vertebral fractures is high (e.g., those with prevalent vertebral fractures), good practice includes seeking advice from a consultant with expertise in osteoporosis on how to transition to an alternative therapy. | Good practice statement |
| 5.6. After a course of anabolic therapy, we suggest transitioning to an antiresorptive agent to maintain bone density gains. | Conditional recommendation; low-certainty evidence |
Note: BMD = bone mineral density.
See Figure 2 for integrated approach.
See Table 1 for definitions.
See risk factors in Figure 1 and Appendix 1, Supplementary Table 5 (causes of secondary osteoporosis), available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content
Taking oral bisphosphonates for 5 years or more (e.g., for as long as 10 yr in the oral alendronate extension study57), compared with shorter durations, likely results in no difference in hip or overall number of fractures, but a moderate-to-small reduction in clinically (22 fewer per 1000) and radiologically (17 fewer per 1000) identified vertebral fractures.43,58 Taking zoledronic acid annually for 6 years, compared with 3 years annually, likely results in no difference in hip and nonvertebral fractures, but radiologically confirmed vertebral fractures may be substantially reduced, although the evidence is uncertain (56 fewer per 1000, from 88 to 5 fewer).48
Harms may be increased with longer durations of bisphosphonates: after 6 years, there are 39–131 atypical femur fractures (a stress or insufficiency fracture occurring in the femoral shaft) per 100 000 person-years, compared with 25 at 3–5 years, and higher risk in females who self-report Asian race or ethnicity;49 and the risk of osteonecrosis of the jaw (a condition in which ≥ 1 parts of the jaw bone becomes necrotic and exposed to the oral cavity) is 25 per 100 000 person-years, and approximately doubles with use longer than 5 years.50 At 6 years, these harms likely outweigh the benefits of continued therapy, except in people at higher risk of fractures (e.g., previous hip or vertebral fractures, recent fracture, multiple fractures).
Evidence suggests that the benefits of denosumab do not wane at 10 years of therapy and the incidence of atypical femur fracture and osteonecrosis of the jaw is relatively stable at 8 and 52 per 100 000 patient-years, respectively.51 There are insufficient data comparing the effects of continuing or switching medications when there is concern of inadequate response to bisphosphonates (e.g., substantial BMD decline).59–62
Monitoring
Table 7 presents the recommendations on monitoring in patients who initiate pharmacotherapies or stop bisphosphonates. Evidence-to-decision tables are in Appendix 2 (p. 249).
Table 7:
Monitoring of therapy in postmenopausal females and males aged 50 years and older who receive anti-osteoporosis medication*
| Recommendations | Strength of recommendation and certainty of evidence† |
|---|---|
| 6. Monitoring | |
| 6.1. We suggest BMD measurement 3 yr after initiating pharmacotherapy. Remarks: BMD measurement may be repeated at a shorter interval in people with secondary causes of osteoporosis, new fracture or new clinical risk factors associated with rapid bone loss.‡ |
Conditional recommendation; very low-certainty evidence |
| 6.2. Three years after stopping bisphosphonate therapy (i.e., drug holiday), we suggest repeating BMD measurement and clinical assessment of fracture risk to determine the need for resumption of therapy. We suggest following the recommendations for risk assessment and initiation of pharmacotherapy. Remark: A shorter interval for reassessment to resume therapy may be appropriate in people with higher risk of fracture (such as previous hip fracture or high FRAX or CAROC score), secondary causes of osteoporosis, new fracture or new clinical risk factors associated with rapid bone loss.‡ |
Conditional recommendation; very low-certainty evidence |
| 6.3. We suggest against monitoring using bone turnover markers for fracture prevention or for deciding on resumption of therapy in people who have stopped bisphosphonates (drug holiday). | Conditional recommendation; very low-certainty evidence |
| 6.4. We suggest against using a fracture risk assessment tool (FRAX or CAROC) for monitoring response to pharmacotherapy. | Conditional recommendation; very low-certainty evidence |
| 6.5. Good practice includes regular clinical assessment for new fractures and new or active risk factors such as falls, as well as adherence to therapy, tolerability and adverse effects. Remark: Adherence to osteoporosis medications is known to be low and may be lower in people who have multiple comorbidities or medications, adverse effects, no drug coverage or misconceptions about osteoporosis therapy. |
Good practice statement |
| 6.6. Good practice includes counselling on and monitoring for symptoms of AFF and ONJ with bisphosphonates or denosumab therapy. Remark: Risk factors for AFF include glucocorticoid use, longer duration of therapy. The risk is also higher in females who self-report Asian race or ethnicity. Unexplained thigh or groin pain should be evaluated. Poor dental health, invasive dental surgery and glucocorticoid use are risk factors for ONJ; oral cavity lesions should be evaluated by a dentist. |
Good practice statement |
Note: AFF = atypical femur fracture, BMD = bone mineral density, CAROC = Canadian Association of Radiologists and Osteoporosis Canada tool, FRAX = Fracture Risk Assessment Tool, ONJ = osteonecrosis of the jaw.
See Table 1 for definitions.
See risk factors in Figure 1 and Appendix 1, Supplementary Table 5 (causes of secondary osteoporosis), available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content
Very low-certainty direct and strong indirect evidence in females and males support a possible improvement in fracture risk in people who undergo BMD monitoring about 3 years after initiation of therapy.63,64 Bone turnover markers measure the remodelling process (e.g., C-terminal telopeptide of type 1 collagen for bone resorption and bone-specific bone alkaline phosphatase for bone formation).65 Inadequate evidence exists to support the clinical use of bone turnover markers to reduce fracture risk or to predict fracture in those on a bisphosphonate drug holiday.66
Methods
This guideline was developed by the volunteer Osteoporosis Canada 2023 Guideline Update Group to address advances in risk assessment, nonpharmacologic and pharmacologic osteoporosis management since the publication of the 2010 Osteoporosis Canada guideline.8 We followed the GRADE framework13 and quality assurance as per Appraisal of Guidelines for Research and Evaluation (AGREE II) quality and reporting standards.67 We began the guideline work in June 2017 and completed it in November 2022.
Guideline panel composition
The Osteoporosis Canada 2023 Guideline Update Group consisted of a steering committee (S.N.M., S.K., N.S., S.F., L.G., W.W., H.M.-B. [ex officio] and L.F.) selected by the Osteoporosis Canada Scientific Advisory Council, a conflict-of-interest oversight committee (H.M.-B., L.F. and R.R.), 4 working groups (chairs: exercise [L.G.], nutrition [W.W.], fracture risk assessment [S.F.], pharmacotherapy [S.K. and N.S.]) and a knowledge translation committee. Primary care physicians and 5 patient partners were represented on the steering committee and were on the working groups, to ensure relevance to target users. A methodologist (N.S.) with expertise in guideline development and GRADE methodology was a member of the steering committee and the Pharmacotherapy Working Group, and consulted with the other working groups.
The steering committee was responsible for the organization of the guideline processes, setting of priorities, recruiting of working group participants, oversight of the working group activities and preparation for publication. We applied the principles of equity, diversity and inclusion in the recruitment of working group participants, ensuring appropriate representation for the following: sex, age, expertise (e.g., physicians, pharmacists, physiotherapists, nutritionist, patient partners) and location in Canada. Working group chairs were tasked with recruiting participants to their group with support from the steering committee. We recruited patient partners from the Canadian Osteoporosis Patient Network across the country (https://osteoporosis.ca/copn-patient-network/).
Selection of priority topics
We established the priority areas and all research questions from surveys of more than 1000 patient partners68 and primary care physicians, and through direct input from people with lived osteoporosis experience, primary care physicians and bone health experts at an initial face-to-face meeting (June 2, 2017; 35 attendees). Each working group formulated questions using the PICO framework (patient, intervention, comparison, outcome), which the steering committee then reviewed (Appendix 2, Questions, p. 22).
Outcomes of interest comprised hip fractures, vertebral fractures, all fractures, fracture-related mortality, physical functioning and disability, quality of life or well-being, and harms such as adverse events from medication, including atypical femur fractures and osteonecrosis of the jaw.68 The Exercise Working Group also considered falls. We agreed, through consensus, on the variation in absolute risk that would be considered clinically important by patients and clinicians, depending on the outcome (Appendix 2, Outcomes of Interest, p. 20).
Literature search and quality assessment
We conducted literature searches (Appendix 2, Search Strategies, p. 27), initially looking for recent systematic reviews of RCTs. In the absence of recent systematic reviews, we conducted a systematic review of the primary literature (initially of RCTs and, if not available, of observational studies).21,22,24,25,69,70 Each working group had the support of a research librarian for the search strategies. The development of certain recommendations was supported by the results of analyses from a large observational Canadian cohort registry (the Manitoba Bone Mineral Density registry).32,38 We searched for systematic reviews for patient values and preferences. We considered indirect evidence when direct evidence was not available or was of very low quality.
The latest systematic search dates were October 2020; each working group surveyed the literature for pertinent RCTs until the fall of 2022 (or publication of their systematic reviews, as for the Exercise Working Group). We verified the results of the searches for the presence of retracted articles (Appendix 2, Pharmacotherapy Working Group, p. 49). Each working group summarized the evidence, appraised it for biases and developed evidence-to-decision tables (Appendix 2, Evidence-to-Decision Tables, p. 50).
Development of recommendations
After the development of the evidence-to-decision tables, each working group formulated the recommendations using the GRADE framework.13 The working groups considered benefits and harms, patient values and preferences, resources, equity, acceptability and feasibility when making decisions and presented their initial work at a face-to-face meeting (Nov. 15, 2019; 28 attendees). Initial decision-making on the recommendations took place separately in each working group, by voting (email or using GRADEpro software [https://gradepro.org]) or by consensus, depending on the working group size and its membership.
The strength of each recommendation was assigned according to the GRADE system, as strong (“We recommend …”) or conditional (“We suggest …”) (Table 1). Some recommendations met the GRADE Working Group criteria for GPS, where the guideline group agreed that the benefits of implementing the GPS would result in large positive consequences (even if supported by indirect evidence), and did not require further collection and summary of the evidence.12 Recommendations and GPS from each working group were reviewed and approved by the steering committee and thereafter by the Osteoporosis Canada 2023 Guideline Update Group as a whole.
External review
Relevant stakeholders (Appendix 2, Stakeholder Participants, p. 18), as determined by the steering committee, provided feedback in October 2022 regarding clarity and clinical utility of the recommendations. Each working group collated and reviewed feedback received for the recommendations they had developed; the steering committee then reviewed the suggested changes and incorporated them as appropriate into the guideline, followed by review and approval from the Osteoporosis Canada 2023 Guideline Update Group.
Management of competing interests
The process for managing competing interests was developed before and continued throughout the guideline development, informed by Guideline International Network principles and overseen by the conflict-of-interest oversight committee (Appendix 2, Conflict of Interest, p. 6).71 Working group chairs and at least half of each working group’s membership were required to be free of financial competing interests during the guideline development process. Members with financial competing interests were excluded from formulating and voting on recommendations. Competing interests were adjudicated via annual submission by email (Appendix 2, Annual Conflict-of-Interest Declaration Template, p. 12) to the conflict-of-interest oversight committee and by verbal updates to respective chairs at each working group meeting.
Funding came from the Canadian Institutes of Health Research (Planning and Dissemination Grant program 2017) and Osteoporosis Canada. Osteoporosis Canada, a patient-focused organization, supports, educates and advocates at the individual, community and governmental levels regarding bone health, fracture risk reduction and management strategies for osteoporosis. It receives about 8% of its funding from corporations and foundations (including pharmaceutical industry and lobby groups) and the remainder from government sources (68.5%), individuals (23%) and other (0.5%) (https://osteoporosis.ca/our-impact/).
We managed the funders’ competing interests through an arm’s-length relationship with Osteoporosis Canada leadership or staff, who did not sit on committees nor any of the working groups. The funding sources had no direct contact with any member of the steering committee or working groups. The views of the funding bodies did not influence the process, nor the content of the recommendations of the guideline. Funding supported research librarians via Cochrane Canada and the GRADE methodologist at McMaster University via institutional mechanisms.
Implementation
We are developing knowledge transfer tools to support patients and clinicians in their discussions on exercise, nutrition, fracture risk and osteoporosis treatment, including a free digital mobile application to provide point of care and educational support (https://osteoporosis.ca/). We will ensure timely dissemination of the tools and guideline content through the Osteoporosis Canada website and network, podcast, and initiatives with patients and primary care providers. Osteoporosis Canada will monitor downloads of the digital application, traffic to its website, the Canadian FRAX tool and implementation of the guideline recommendations in Fracture Liaison Services across the country.
Updates will be published as peer-reviewed, structured guidance statements when new data pertinent to clinical assessment and management of osteoporosis emerge.
Other guidelines
Changes from the 2010 Osteoporosis Canada guideline8 include the use of the GRADE framework in the development of the recommendations, the addition of expanded recommendations on exercise and types of exercise, and guidance on nutrients other than calcium and vitamin D. We also provide clearer guidance on thresholds for treatment initiation, duration of therapy and monitoring.
Based on evidence from a systematic review of RCTs, the Canadian Task Force on Preventive Health Care guideline recommends risk assessment — first screening for the primary prevention of fragility fractures in females aged 65 years and older with the initial application of the FRAX tool without BMD.72 The task force guideline recommends against screening younger females and males of any age. Of note, the task force’s recommendations apply to community-dwelling people who are not on anti-osteoporosis pharmacotherapy.
The United States Preventive Services Task Force found convincing evidence for the accuracy of BMD measurements and risk assessment tools at identifying risk of osteoporotic fractures, and that drug therapies reduce fracture rates in postmenopausal females.73 The US Bone Health and Osteoporosis Foundation uses a similar approach as our update, to screening (i.e., screening postmenopausal females and males aged ≥ 50 yr with a history of fracture or clinical risk factors) and recommends treatment based on any of the following: vertebral or hip fracture; humerus, pelvis or distal forearm fracture with low bone mass; a BMD T-score ≤ −2.5; and low bone mass and a US FRAX 10-year probability of a hip fracture ≥ 3% or of major osteoporosis-related fracture ≥ 20%.74 The United Kingdom National Osteoporosis Guidelines Group uses a 2-step screening process for identifying people at high fracture risk, with age-dependent cut-offs for pharmacologic intervention.75 Most clinical guidelines recommend vertebral fracture assessment in people with selected risk factors.74,76
Recent guidelines, such as those of the American College of Physicians, the US Bone Health and Osteoporosis Foundation, and the UK National Osteoporosis Guidelines Group, recommend bisphosphonates as first-line therapy in postmenopausal females and males with osteoporosis, whereas denosumab and anabolic therapies are mostly suggested as second-line therapy for people with intolerance, contraindications or higher risk.44,74,75,77 In addition, these guidelines support adequate calcium and vitamin D intake, and exercise for falls and fracture reduction.
Gaps in knowledge
More evidence is required to guide management in males and with regard to optimal screening of younger people (age 50–64 yr) for fracture risk reduction. The FRAX and CAROC tools have not been validated in transgender and gender-nonconforming people. Dietary patterns and the optimal level of dietary protein for fracture prevention are areas for future study. Further research is also warranted on duration of pharmacotherapy, bisphosphonate interruptions, and sequence of treatments, particularly in those who remain at high risk for fracture.
Limitations
We were not able to incorporate all topics of high relevance to skeletal health, such as fall prevention or identification of frailty, within this guideline. We also recognize that primary health care professionals may care for people with secondary causes of osteoporosis and multiple comorbidities. However, we determined that these situations were beyond the scope of this guideline, as they would require additional expertise, and therefore we recommended consultation with a specialist as feasible.
Although we did not update our searches of the literature on an ongoing basis (such as in living guidelines), we did keep abreast of any new or pivotal trials before publication. For some recommendations, we used indirect evidence to inform our guidance (e.g., screening, treatment and monitoring in males). In addition, we made recommendations based on magnitude of effects on fractures and harms that we assessed to be clinically important a priori, through consensus. We consider our guideline group was representative and would reflect values important to people at risk of fractures.
Conclusion
The aim of this guideline is to empower health care professionals and patients to have meaningful discussions on the importance of skeletal health and fracture risk in older adulthood. Identification and appropriate management of skeletal fragility in Canadians can reduce fractures, and preserve mobility, autonomy and quality of life in this population.
Supplementary Material
Acknowledgements
The authors thank the support team for their dedication to the guideline project and acknowledge their contributions. Fracture risk assessment working group: Hajar Abu Alrob, Arnav Agarwal, Lindsie Blencowe, Marija Djekic-Ivankovic and Jeevitha Srighanthan. Exercise working group: Zachary Fielding, Seyedeh Zeinab Hosseini, Kawon Victoria Kim and Emily McLaughlin. Nutrition working group: Ina Ilse. Pharmacotherapy working group: Lindsay Wong. Project administrative liaison: Kerry Grady (Osteoporosis Canada). Research librarians: Yuhong Yuan (McMaster University) and Jackie Stapleton (University of Waterloo). The authors thank the external reviewers and stakeholders (named in Appendix 2, available at www.cmaj.ca/lookup/doi/10.1503/cmaj.221647/tab-related-content) for their contributions to the guideline.
Footnotes
Competing interests: Lora Giangregorio reports receiving an honorarium from Amgen Inc. for a lecture on exercise with no mention of medications, outside the current manuscript. Dr. Giangregorio is also a member of the Scientific Advisory Council of Osteoporosis Canada and co-lead of the Vertebral Fracture Special Interest Group, Fragility Fracture Network. Sandra Kim reports receiving support from Osteoporosis Canada for a Grading of Recommendations, Assessment, Development and Evaluations (GRADE) methodologist at McMaster University, during the conduct of the study. Dr. Kim was also a volunteer member of the Osteoporosis Canada Board of Directors from 2017 to 2020. Nancy Santesso reports receiving payment to provide GRADE expertise for guideline development and preparation of materials paid to institution (McMaster University), during the conduct of the study. Dr. Santesso has also received support from Osteoporosis Canada for travel to guideline-related meetings. Heather McDonald-Blumer reports receiving consulting fees from Eli Lilly and Novartis, for attendance on advisory boards, outside the submitted work. Rowena Ridout has been a member of the Osteoporosis Canada Board of Directors since 2020 (no payment received). Neil Binkley reports receiving a research grant (paid to institution) from Radius, and consulting fees and honoraria from Amgen. Angela Cheung has received honoraria for consultancy work from Amgen and Paladin Laboratories. Dr. Cheung has also held the following roles: member of the Clinical Guidelines Committee of the Endocrine Society; co-chair of the postmenopausal health pathway, American College of Obstetrics and Gynecology; and vice-president and co-chair of the 2023 Position Development Conference, International Society for Clinical Densitometry. Robert Josse reports receiving consulting fees from Amgen Canada, Paladin Laboratories and Alexion, and honoraria from Amgen and Alexion. Dr. Josse has also participated on advisory boards for Amgen, Paladin, Alexion and Ultragenyx. Aliya Khan has received research grants from Alexion, Amgen Canada, Takeda, Ascendis, Chugai, Radius, Amolyt and Ultragenyx, and speaker honoraria from Amgen, Alexion, Ascendis, Takeda and Ultragenyx. Dr. Khan has also participated on advisory boards for Amgen Canada, Alexion, Amolyt, Ascendis and Takeda. E. Lynn Izumi Nash reports receivin an honorarium from the Ontario College of Family Physicians for designing continuing medical education workshops on osteoporosis. Dr. Nash is also a member of the Scientific Advisory Council for Osteoporosis Canada (voluntary unpaid position). Zahra Bardai reports receiving honoraria as a member of the Pri-Med Canada Scientific Planning Committee (2011–present) and as an author of the Foundation for Medical Practice Education Small Group Practice–Based Learning Program, at McMaster University. Dr. Bardai is also a volunteer member of the Osteoporosis Canada Scientific Advisory Council. Suzanne Cadarette reports receiving research funds (paid to institution) as co–principal investigator on Canadian Institutes of Health Research and National Institutes of Health project grants. Heather Frame is a former member of the Osteoporosis Canada Board of Directors (term ended November 2020). Kaleen Hayes reports receiving researcher-initiated grants from the National Institute on Aging, Insight Therapeutics, Genentech and Sanofi (all paid directly to institution), and consulting fees from the Canadian Agency for Drugs and Technologies in Health. Alexandra Papaioannou has participated on an advisory board and in speakers’ bureaus and received honoraria from Amgen Canada. Christine Thomas has been a member of the Osteoporosis Canada Board of Directors at various time points.
This article has been peer reviewed.
Steering Committee: Suzanne N. Morin (chair), Sidney Feldman, Larry Funnell, Lora Giangregorio, Wendy Ward, Sandra Kim, Nancy Santesso, Heather McDonald-Blumer (ex-officio).
Conflict-of-Interest Oversight Committee: Heather McDonald-Blumer (Chair), Larry Funnell, Rowena Ridout.
Stakeholder Committee: Heather McDonald-Blumer (chair), Rowena Ridout, Larry Funnell.
Fracture Risk Assessment Working Group: Sidney Feldman (chair), Neil Binkley, Steven Burrell, Angela M. Cheung, Carol Holmes, George Ioannidis, Robert Josse, Aliya A. Khan, Virginia McIntyre, Suzanne N. Morin, Lynn Nash, Ahmed Negm, Lianne Tile.
Exercise Working Group: Lora Giangregorio (chair), Maureen C. Ashe, Zahra Bardai, Joan Bartley, Debra Butt, Phil Chilibeck, Isabel B. Rodrigues, Lehana Thabane, Matteo Ponzano, John D. Wark.
Nutrition Working Group: Wendy Ward (chair), William Gittings, Lynn Nash.
Pharmacotherapy Working Group: Sandra Kim (co-chair), Nancy Santesso (co-chair), Suzanne M. Cadarette, Sheila Dunn, Jamie Falk, Heather Frame, Kaleen Hayes, Susan B. Jaglal, Alexandra Papaioannou, Rowena Ridout, Christine A. Thomas.
Contributors: All of the authors made substantial contributions to the conception and design of the work, as well as acquisition, analysis and interpretation of data. Members of the Steering Committee wrote the first draft and all of the authors contributed to subsequent versions of the manuscript, including reviewing them critically for important intellectual content. All of the authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
Funding: Funding came from the Canadian Institutes of Health Research (Planning and Dissemination Grant program, 2017), and Osteoporosis Canada. The views of the funding bodies did not influence the process, the content of this guideline or the preparation of the manuscript.
References
- 1.Osteoporosis and related fractures in Canada: report from the Canadian Chronic Disease Surveillance System. Ottawa: Public Health Agency of Canada; 2020:1–85. Available: https://www.canada.ca/content/dam/phac-aspc/documents/services/publications/diseases-conditions/osteoporosis-related-fractures-2020/osteoporosis-related-fractures-2020.pdf (accessed 2023 May 15). [Google Scholar]
- 2.Hopkins RB, Burke N, Von Keyserlingk C, et al. The current economic burden of illness of osteoporosis in Canada. Osteoporos Int 2016;27:3023–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Adler RA. Update on osteoporosis in men. Best Pract Res Clin Endocrinol Metab 2018;32:759–72. [DOI] [PubMed] [Google Scholar]
- 4.Siminoski K, Leslie WD, Frame H, et al. Canadian Association of Radiologists. Recommendations for bone mineral density reporting in Canada. Can Assoc Radiol J 2005;56:178–88. [PubMed] [Google Scholar]
- 5.Siris ES, Adler R, Bilezikian J, et al. The clinical diagnosis of osteoporosis: a position statement from the National Bone Health Alliance Working Group. Osteoporos Int 2014;25:1439–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Leslie WD, Lix LM, Langsetmo L, et al. Construction of a FRAX model for the assessment of fracture probability in Canada and implications for treatment. Osteoporos Int 2011;22:817–27. [DOI] [PubMed] [Google Scholar]
- 7.Leslie WD, Berger C, Langsetmo L, et al. Canadian Multicentre Osteoporosis Study Research Group. Construction and validation of a simplified fracture risk assessment tool for Canadian women and men: results from the CaMos and Manitoba cohorts. Osteoporos Int 2011;22:1873–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Papaioannou A, Morin S, Cheung AM, et al. Scientific Advisory Council of Osteoporosis Canada. 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ 2010;182:1864–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Seniors’ falls in Canada, second report: protecting Canadians from illness. Ottawa: Public Health Agency of Canada; 2014:1–62. Available: https://www.canada.ca/content/dam/phac-aspc/migration/phac-aspc/seniors-aines/publications/public/injury-blessure/seniors_falls-chutes_aines/assets/pdf/seniors_falls-chutes_aines-eng.pdf (accessed 2023 April 11). [Google Scholar]
- 10.Pillay J, Riva JJ, Tessier LA, et al. Fall prevention interventions for older community-dwelling adults: systematic reviews on benefits, harms, and patient values and preferences. Syst Rev 2021;10:18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Papaioannou A, Santesso N, Morin SN, et al. Scientific Advisory Council of Osteoporosis Canada. Recommendations for preventing fracture in long-term care. CMAJ 2015;187:1135–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dewidar O, Lotfi T, Langendam MW, et al. eCOVID-19 recommendations map collaborators. Good or best practice statements: proposal for the operationalisation and implementation of GRADE guidance. BMJ Evid Based Med 2023; 28:189–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schünemann HJ, Wiercioch W, Brozek J, et al. GRADE Evidence to Decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE-ADOLOPMENT. J Clin Epidemiol 2017;81: 101–10. [DOI] [PubMed] [Google Scholar]
- 14.Li N, Beaudart C, Cauley JA, et al. Cost effectiveness analyses of interventions for osteoporosis in men: a systematic literature review. PharmacoEconomics 2023;41:363–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Davis S, Simpson E, Hamilton J, et al. Denosumab, raloxifene, romosozumab and teriparatide to prevent osteoporotic fragility fractures: a systematic review and economic evaluation. Health Technol Assess 2020;24:1–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Coyle D. Cost-effectiveness of pharmacological treatments for osteoporosis consistent with the revised economic evaluation guidelines for Canada. MDM Policy Pract 2019;4:2381468318818843. doi: 10.1177/2381468318818843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kanis JA, Johnell O, De Laet C, et al. A meta-analysis of previous fracture and subsequent fracture risk. Bone 2004;35:375–82. [DOI] [PubMed] [Google Scholar]
- 18.Lentle B, Koromani F, Brown JP, et al. Vertebral Fracture Research Groups of the CaMos, STOPP, and Rotterdam Studies. The radiology of osteoporotic vertebral fractures revisited. J Bone Miner Res 2019;34:409–18. [DOI] [PubMed] [Google Scholar]
- 19.Johansson L, Johansson H, Axelsson KF, et al. Improved fracture risk prediction by adding VFA-identified vertebral fracture data to BMD by DXA and clinical risk factors used in FRAX. Osteoporos Int 2022;33:1725–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sherrington C, Fairhall NJ, Wallbank GK, et al. Exercise for preventing falls in older people living in the community. Cochrane Database Syst Rev 2019;1: CD012424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ponzano M, Rodrigues IB, Hosseini Z, et al. Progressive resistance training for improving health-related outcomes in people at risk of fracture: a systematic review and meta-analysis of randomized controlled trials. Phys Ther 2021;101: pzaa221. doi: 10.1093/ptj/pzaa221. [DOI] [PubMed] [Google Scholar]
- 22.Ponzano M, Tibert N, Bansal S, et al. Exercise for improving age-related hyperkyphosis: a systematic review and meta-analysis with GRADE assessment. Arch Osteoporos 2021;16:140. [DOI] [PubMed] [Google Scholar]
- 23.Kelly P, Kahlmeier S, Götschi T, et al. Systematic review and meta-analysis of reduction in all-cause mortality from walking and cycling and shape of dose response relationship. Int J Behav Nutr Phys Act 2014;11:132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rodrigues IB, Ponzano M, Butt DA, et al. The effects of walking or nordic walking in adults 50 years and older at elevated risk of fractures: a systematic review and meta-analysis. J Aging Phys Act 2021;29:886–99. [DOI] [PubMed] [Google Scholar]
- 25.Rodrigues IB, Ponzano M, Hosseini Z, et al. The effect of impact exercise (alone or multicomponent intervention) on health-related outcomes in individuals at risk of fractures: a systematic review and meta-analysis of randomized controlled trials. Sports Med 2021;51:1273–92. [DOI] [PubMed] [Google Scholar]
- 26.Zhao J-G, Zeng X-T, Wang J, et al. Association between calcium or vitamin D supplementation and fracture incidence in community-dwelling older adults. JAMA 2017;318:2466–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bolland MJ, Grey A, Avenell A. Effects of vitamin D supplementation on musculoskeletal health: a systematic review, meta-analysis, and trial sequential analysis. Lancet Diabetes Endocrinol 2018;6:847–58. [DOI] [PubMed] [Google Scholar]
- 28.Bolland MJ, Grey A, Gamble GD, et al. Vitamin D supplementation and falls: a trial sequential meta-analysis. Lancet Diabetes Endocrinol 2014;2:573–80. [DOI] [PubMed] [Google Scholar]
- 29.Groenendijk I, den Boeft L, van Loon LJC, et al. High versus low dietary protein intake and bone health in older adults: a systematic review and meta-analysis. Comput Struct Biotechnol J 2019;17:1101–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Darling AL, Manders RJF, Sahni S, et al. Dietary protein and bone health across the life-course: an updated systematic review and meta-analysis over 40 years. Osteoporos Int 2019;30:741–61. [DOI] [PubMed] [Google Scholar]
- 31.Ten Haaf DSM, Nuijten MAH, Maessen MFH, et al. Effects of protein supplementation on lean body mass, muscle strength, and physical performance in non-frail community-dwelling older adults: a systematic review and meta-analysis. Am J Clin Nutr 2018;108:1043–59. [DOI] [PubMed] [Google Scholar]
- 32.Leslie WD, Morin SN, Lix LM, et al. Targeted bone density testing for optimizing fracture prevention in Canada. Osteoporos Int 2020;31:1291–7. [DOI] [PubMed] [Google Scholar]
- 33.Shepstone L, Lenaghan E, Cooper C, et al. SCOOP Study Team. Screening in the community to reduce fractures in older women (SCOOP): a randomised controlled trial. Lancet 2018;391:741–7. [DOI] [PubMed] [Google Scholar]
- 34.Merlijn T, Swart KMA, van der Horst HE, et al. Fracture prevention by screening for high fracture risk: a systematic review and meta-analysis. Osteoporos Int 2020;31:251–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liu H, Paige NM, Goldzweig CL, et al. Screening for osteoporosis in men: a systematic review for an American College of Physicians guideline. Ann Intern Med 2008;148:685–701. [DOI] [PubMed] [Google Scholar]
- 36.Beaudoin C, Moore L, Gagné M, et al. Performance of predictive tools to identify individuals at risk of non-traumatic fracture: a systematic review, meta-analysis, and meta-regression. Osteoporos Int 2019;30:721–40. [DOI] [PubMed] [Google Scholar]
- 37.Leslie WD, Majumdar SR, Lix LM, et al. Direct comparison of FRAX® and a simplified fracture risk assessment tool in routine clinical practice: a registry-based cohort study. Osteoporos Int 2016;27:2689–95. [DOI] [PubMed] [Google Scholar]
- 38.Leslie WD, Morin SN, Lix LM, et al. Comparison of treatment strategies and thresholds for optimizing fracture prevention in Canada: a simulation analysis. Arch Osteoporos 2019;15:4. [DOI] [PubMed] [Google Scholar]
- 39.Barrionuevo P, Kapoor E, Asi N, et al. Efficacy of pharmacological therapies for the prevention of fractures in postmenopausal women: a network meta-analysis. J Clin Endocrinol Metab 2019;104:1623–30. [DOI] [PubMed] [Google Scholar]
- 40.Black D, Lui L-Y, Bauer D, et al. Do women with lower BMD benefit more from anti-fracture treatment? An analysis pooling individual patient data from 134,000 women in the FNIH-SABRE RCT Database. J Bone Miner Res 2020;35. [Google Scholar]
- 41.Nayak S, Greenspan SL. Osteoporosis treatment efficacy for men: a systematic review and meta-analysis. J Am Geriatr Soc 2017;65:490–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li P, Wu X, Li Y, et al. Denosumab versus bisphosphonates for the prevention of the vertebral fractures in men with osteoporosis: an updated network meta-analysis. Clin Invest Med 2022;45:E14–22. [DOI] [PubMed] [Google Scholar]
- 43.Crandall CJ, Newberry SJ, Diamant WG, et al. Treatment to prevent fractures in men and women with low bone density or osteoporosis: update of a 2007 report. Report No 12-EHC023-EF. Rockville (MD): Agency for Healthcare Research and Quality (US); 2012. [PubMed] [Google Scholar]
- 44.Qaseem A, Hicks LA, Etxeandia-Ikobaltzeta I, et al. Pharmacologic treatment of primary osteoporosis or low bone mass to prevent fractures in adults: a living clinical guideline from the American College of Physicians. Ann Intern Med 2023;176:224–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wu C-H, Tu S-T, Chang Y-F, et al. Fracture liaison services improve outcomes of patients with osteoporosis-related fractures: a systematic literature review and meta-analysis. Bone 2018;111:92–100. [DOI] [PubMed] [Google Scholar]
- 46.Reid IR, Horne AM, Mihov B, et al. Fracture prevention with zoledronate in older women with osteopenia. N Engl J Med 2018;379:2407–16. [DOI] [PubMed] [Google Scholar]
- 47.Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab on bone mineral density and bone turnover in postmenopausal women. J Clin Endocrinol Metab 2008;93:2149–57. [DOI] [PubMed] [Google Scholar]
- 48.Black DM, Reid IR, Boonen S, et al. The effect of 3 versus 6 years of zoledronic acid treatment of osteoporosis: a randomized extension to the HORIZON-Pivotal Fracture Trial (PFT). J Bone Miner Res 2012;27:243–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Black DM, Geiger EJ, Eastell R, et al. Atypical femur fracture risk versus fragility fracture prevention with bisphosphonates. N Engl J Med 2020;383:743–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Eiken PA, Prieto-Alhambra D, Eastell R, et al. Surgically treated osteonecrosis and osteomyelitis of the jaw and oral cavity in patients highly adherent to alendronate treatment: a nationwide user-only cohort study including over 60,000 alendronate users. Osteoporos Int 2017;28:2921–8. [DOI] [PubMed] [Google Scholar]
- 51.Bone HG, Wagman RB, Brandi ML, et al. 10 years of denosumab treatment in postmenopausal women with osteoporosis: results from the phase 3 randomised FREEDOM trial and open-label extension. Lancet Diabetes Endocrinol 2017;5:513–23. [DOI] [PubMed] [Google Scholar]
- 52.Tsourdi E, Zillikens MC, Meier C, et al. Fracture risk and management of discontinuation of denosumab therapy: a systematic review and position statement by ECTS. J Clin Endocrinol Metab 2020. Oct. 26 [Epub ahead of print]. doi: 10.1210/clinem/dgaa756. [DOI] [PubMed] [Google Scholar]
- 53.Kanis JA, Johansson H, Odén A, et al. Characteristics of recurrent fractures. Osteoporos Int 2018;29:1747–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.McClung MR, Brown JP, Diez-Perez A, et al. Effects of 24 months of treatment with romosozumab followed by 12 months of denosumab or placebo in postmenopausal women with low bone mineral density: a randomized, double-blind, phase 2, parallel group study. J Bone Miner Res 2018;33:1397–406. [DOI] [PubMed] [Google Scholar]
- 55.Leder BZ, Neer RM, Wyland JJ, et al. Effects of teriparatide treatment and discontinuation in postmenopausal women and eugonadal men with osteoporosis. J Clin Endocrinol Metab 2009;94:2915–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Barrionuevo P, Gionfriddo MR, Castaneda-Guarderas A, et al. Women’s values and preferences regarding osteoporosis treatments: a systematic review. J Clin Endocrinol Metab 2019;104:1631–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Black DM, Schwartz AV, Ensrud KE, et al. FLEX Research Group. Effects of continuing or stopping alendronate after 5 years of treatment: the Fracture Intervention Trial Long-term Extension (FLEX): a randomized trial. JAMA 2006;296:2927–38. [DOI] [PubMed] [Google Scholar]
- 58.Fink HA, MacDonald R, Forte ML, et al. Long-term drug therapy and drug discontinuations and holidays for osteoporosis fracture prevention: a systematic review. Ann Intern Med 2019;171:37–50. [DOI] [PubMed] [Google Scholar]
- 59.Ensrud KE, Schousboe JT. Anabolic therapy for osteoporosis. JAMA 2021;326:350–1. [DOI] [PubMed] [Google Scholar]
- 60.Eiken P, Vestergaard P. Treatment of osteoporosis after alendronate or risedronate. Osteoporos Int 2016;27:1–12. [DOI] [PubMed] [Google Scholar]
- 61.Miller PD, Pannacciulli N, Brown JP, et al. Denosumab or zoledronic acid in postmenopausal women with osteoporosis previously treated with oral bisphosphonates. J Clin Endocrinol Metab 2016;101:3163–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Obermayer-Pietsch BM, Marin F, McCloskey EV, et al. EUROFORS Investigators. Effects of two years of daily teriparatide treatment on BMD in postmenopausal women with severe osteoporosis with and without prior antiresorptive treatment. J Bone Miner Res 2008;23:1591–600. [DOI] [PubMed] [Google Scholar]
- 63.Bouxsein ML, Eastell R, Lui L-Y, et al. FNIH Bone Quality Project. Change in bone density and reduction in fracture risk: a meta-regression of published trials. J Bone Miner Res 2019;34:632–42. [DOI] [PubMed] [Google Scholar]
- 64.Leslie WD, Morin SN, Martineau P, et al. Association of bone density monitoring in routine clinical practice with anti-osteoporosis medication use and incident fractures: a matched cohort study. J Bone Miner Res 2019;34:1808–14. [DOI] [PubMed] [Google Scholar]
- 65.Bauer DC. Clinical use of bone turnover markers. JAMA 2019;322:569–70. [DOI] [PubMed] [Google Scholar]
- 66.Burch J, Rice S, Yang H, et al. Systematic review of the use of bone turnover markers for monitoring the response to osteoporosis treatment: the secondary prevention of fractures, and primary prevention of fractures in high-risk groups. Health Technol Assess 2014;18:1–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Brouwers MC, Kho ME, Browman GP, et al. AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ 2010;182:E839–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Morin SN, Djekic-Ivankovic M, Funnell L, et al. Patient engagement in clinical guidelines development: input from > 1000 members of the Canadian Osteoporosis Patient Network. Osteoporos Int 2020;31:867–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kim KV, Bartley J, Ashe MC, et al. Effect of yoga on health-related outcomes in people at risk of fractures: a systematic review. Appl Physiol Nutr Metab 2022;47: 215–26. [DOI] [PubMed] [Google Scholar]
- 70.McLaughlin EC, Bartley J, Ashe MC, et al. The effects of pilates on health-related outcomes in individuals at risk of fracture: a systematic review. Appl Physiol Nutr Metab 2022;47:369–78. [DOI] [PubMed] [Google Scholar]
- 71.Schünemann HJ, Al-Ansary LA, Forland F, et al. Board of Trustees of the Guidelines International Network. Guidelines International Network: principles for disclosure of interests and management of conflicts in guidelines. Ann Intern Med 2015;163:548–53. [DOI] [PubMed] [Google Scholar]
- 72.Thériault G, Limburg H, Klarenbach S, et al. Canadian Task Force on Preventive Health Care. Recommendations on screening for primary prevention of fragility fractures. CMAJ 2023;195:E639–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.US Preventive Services Task Force;Curry SJ, Krist AH, Owens DK, et al. Screening for osteoporosis to prevent fractures: US Preventive Services Task Force recommendation statement. JAMA 2018;319:2521–31. [DOI] [PubMed] [Google Scholar]
- 74.LeBoff MS, Greenspan SL, Insogna KL, et al. The clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 2022;33:2049–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gregson CL, Armstrong DJ, Bowden J, et al. UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos 2022;17:58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Schousboe JT, Vokes T, Broy SB, et al. Vertebral fracture assessment: the 2007 ISCD Official Positions. J Clin Densitom 2008;11:92–108. [DOI] [PubMed] [Google Scholar]
- 77.Shoback D, Rosen CJ, Black DM, et al. Pharmacological management of osteoporosis in postmenopausal women: an endocrine society guideline update. J Clin Endocrinol Metab 2020;105:dgaa04810.1210/clinem/dgaa048. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.