Abstract
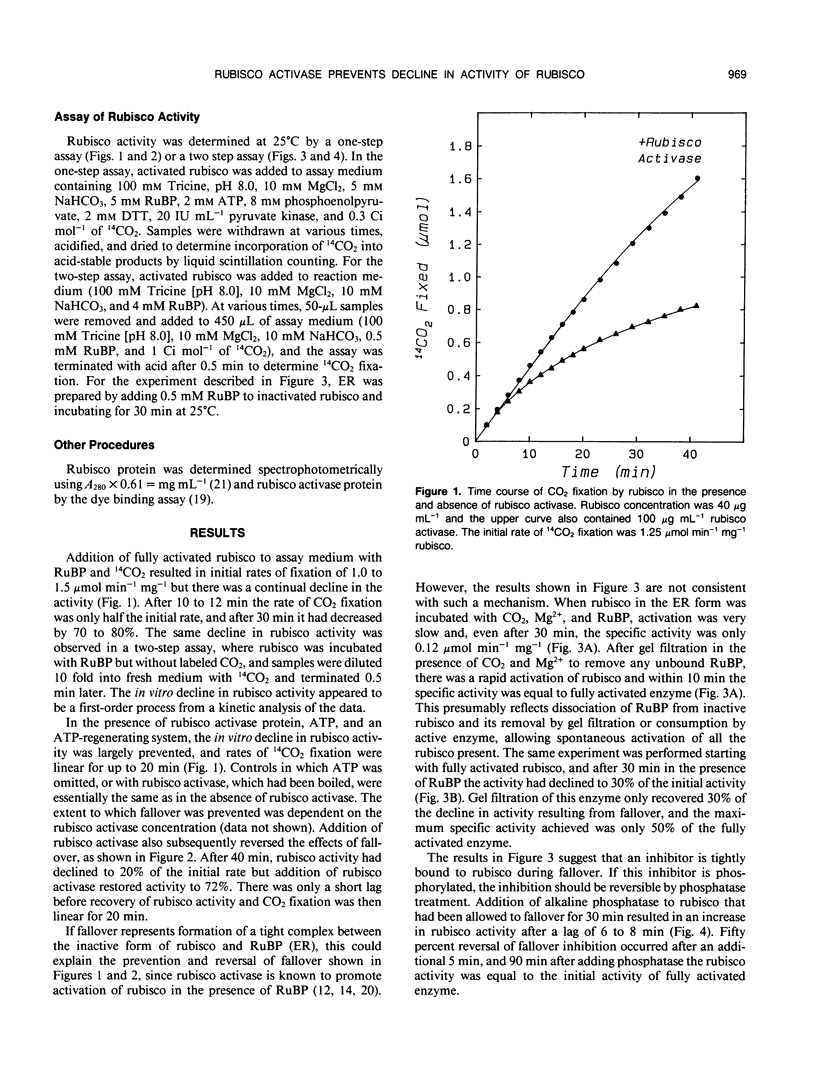
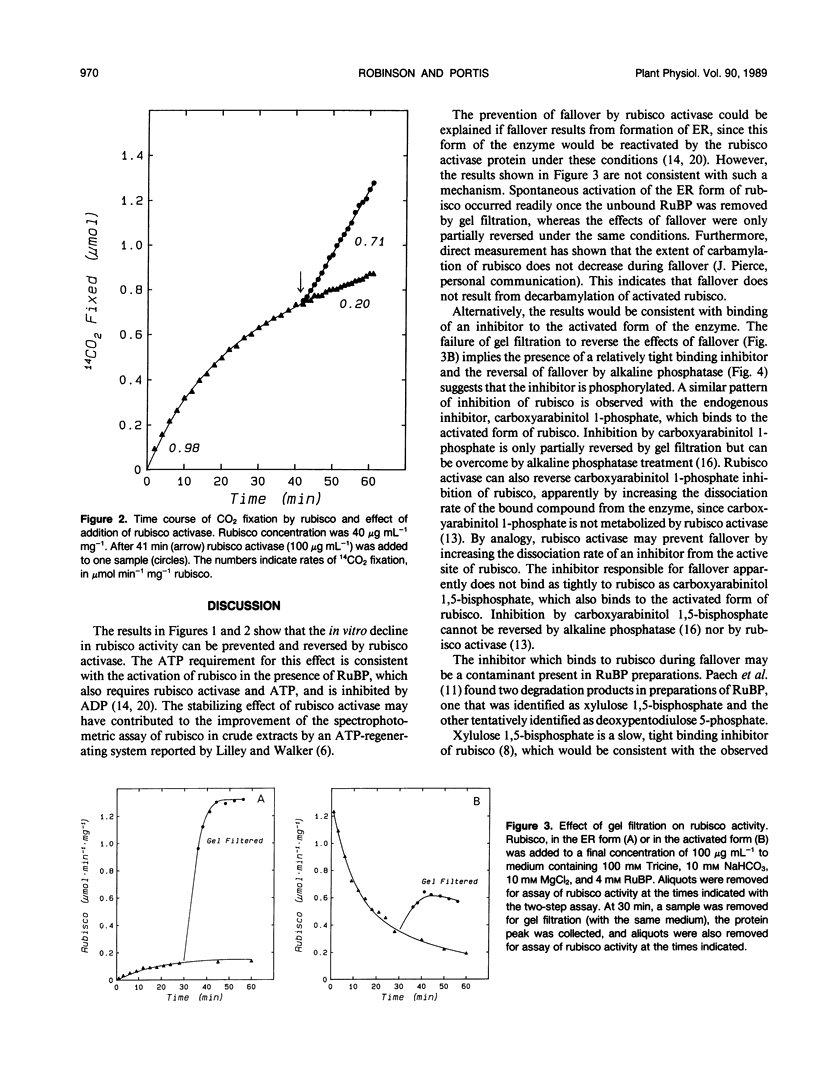
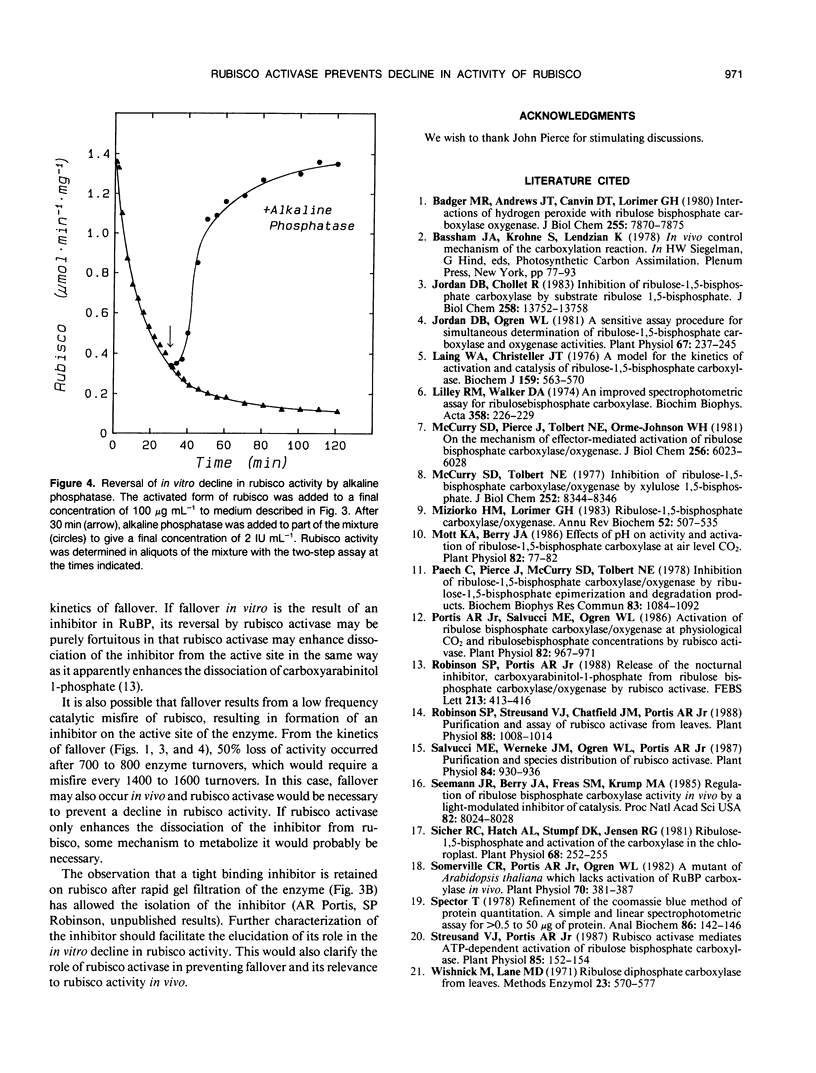
The rate of CO2 fixation by ribulose-1,5-bisphosphate carboxylase/oxygenase (rubisco) following addition of ribulose 1,5-bisphosphate (RuBP) to fully activated enzyme, declined with first-order kinetics, resulting in 50% loss of rubisco activity after 10 to 12 minutes. This in vitro decline in rubisco activity, termed fall-over, was prevented if purified rubisco activase protein and ATP were added, allowing linear rates of CO2 fixation for up to 20 minutes. Rubisco activase could also stimulate rubisco activity if added after fallover had occurred. Gel filtration of the RuBP-rubisco complex to remove unbound RuBP allowed full activation of the enzyme, but the inhibition of activated rubisco during fallover was only partially reversed by gel filtration. Addition of alkaline phosphatase completely restored rubisco activity following fallover. The results suggest that fallover is not caused by binding of RuBP to decarbamylated enzyme, but results from binding of a phosphorylated inhibitor to the active site of rubisco. The inhibitor may be a contaminant in preparations of RuBP or may be formed on the active site but is apparently removed from the enzyme in the presence of the rubisco activase protein.
Full text
PDF
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Badger M. R., Andrews T. J., Canvin D. T., Lorimer G. H. Interactions of hydrogen peroxide with ribulose bisphosphate carboxylase oxygenase. J Biol Chem. 1980 Aug 25;255(16):7870–7875. [PubMed] [Google Scholar]
- Bassham J. A., Krohne S., Lendzian K. In vivo control mechanism of the carboxylation reaction. Basic Life Sci. 1978;11:77–93. doi: 10.1007/978-1-4684-8106-8_6. [DOI] [PubMed] [Google Scholar]
- Jordan D. B., Chollet R. Inhibition of ribulose bisphosphate carboxylase by substrate ribulose 1,5-bisphosphate. J Biol Chem. 1983 Nov 25;258(22):13752–13758. [PubMed] [Google Scholar]
- Jordan D. B., Ogren W. L. A Sensitive Assay Procedure for Simultaneous Determination of Ribulose-1,5-bisphosphate Carboxylase and Oxygenase Activities. Plant Physiol. 1981 Feb;67(2):237–245. doi: 10.1104/pp.67.2.237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laing W. A., Christeller J. T. A model for the kinetics of activation and catalysis of ribulose 1,5-bisphosphate carboxylase. Biochem J. 1976 Dec 1;159(3):563–570. doi: 10.1042/bj1590563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lilley R. M., Walker D. A. An improved spectrophotometric assay for ribulosebisphosphate carboxylase. Biochim Biophys Acta. 1974 Jul 17;358(1):226–229. doi: 10.1016/0005-2744(74)90274-5. [DOI] [PubMed] [Google Scholar]
- McCurry S. D., Tolbert N. E. Inhibition of ribulose-1,5-bisphosphate carboxylase/oxygenase by xylulose 1,5-bisphosphate. J Biol Chem. 1977 Dec 10;252(23):8344–8346. [PubMed] [Google Scholar]
- Miziorko H. M., Lorimer G. H. Ribulose-1,5-bisphosphate carboxylase-oxygenase. Annu Rev Biochem. 1983;52:507–535. doi: 10.1146/annurev.bi.52.070183.002451. [DOI] [PubMed] [Google Scholar]
- Mott K. A., Berry J. A. Effects of pH on Activity and Activation of Ribulose 1,5-Bisphosphate Carboxylase at Air Level CO(2). Plant Physiol. 1986 Sep;82(1):77–82. doi: 10.1104/pp.82.1.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paech C., Pierce J., McCurry S. D., Tolbert N. E. Inhibition of ribulose-1,5-biphosphate carboxylase/oxygenase by ribulose-1,5-bisphosphate epimerization and degradation products. Biochem Biophys Res Commun. 1978 Aug 14;83(3):1084–1092. doi: 10.1016/0006-291x(78)91506-1. [DOI] [PubMed] [Google Scholar]
- Portis A. R., Salvucci M. E., Ogren W. L. Activation of Ribulosebisphosphate Carboxylase/Oxygenase at Physiological CO(2) and Ribulosebisphosphate Concentrations by Rubisco Activase. Plant Physiol. 1986 Dec;82(4):967–971. doi: 10.1104/pp.82.4.967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson S. P., Streusand V. J., Chatfield J. M., Portis A. R. Purification and assay of rubisco activase from leaves. Plant Physiol. 1988 Dec;88(4):1008–1014. doi: 10.1104/pp.88.4.1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salvucci M. E., Werneke J. M., Ogren W. L., Portis A. R. Purification and species distribution of rubisco activase. Plant Physiol. 1987 Jul;84(3):930–936. doi: 10.1104/pp.84.3.930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seemann J. R., Berry J. A., Freas S. M., Krump M. A. Regulation of ribulose bisphosphate carboxylase activity in vivo by a light-modulated inhibitor of catalysis. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8024–8028. doi: 10.1073/pnas.82.23.8024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sicher R. C., Hatch A. L., Stumpf D. K., Jensen R. G. Ribulose 1,5-bisphosphate and activation of the carboxylase in the chloroplast. Plant Physiol. 1981 Jul;68(1):252–255. doi: 10.1104/pp.68.1.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somerville C. R., Portis A. R., Ogren W. L. A Mutant of Arabidopsis thaliana Which Lacks Activation of RuBP Carboxylase In Vivo. Plant Physiol. 1982 Aug;70(2):381–387. doi: 10.1104/pp.70.2.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978 May;86(1):142–146. doi: 10.1016/0003-2697(78)90327-5. [DOI] [PubMed] [Google Scholar]
- Streusand V. J., Portis A. R. Rubisco Activase Mediates ATP-Dependent Activation of Ribulose Bisphosphate Carboxylase. Plant Physiol. 1987 Sep;85(1):152–154. doi: 10.1104/pp.85.1.152. [DOI] [PMC free article] [PubMed] [Google Scholar]


