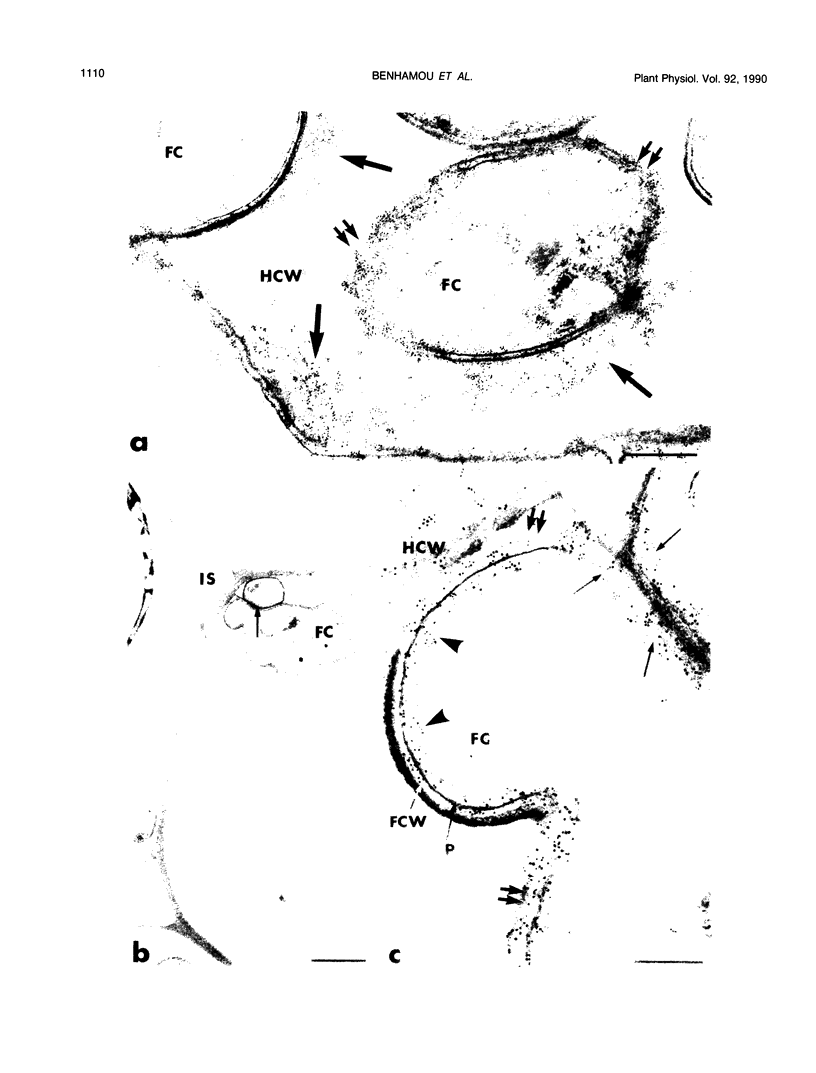
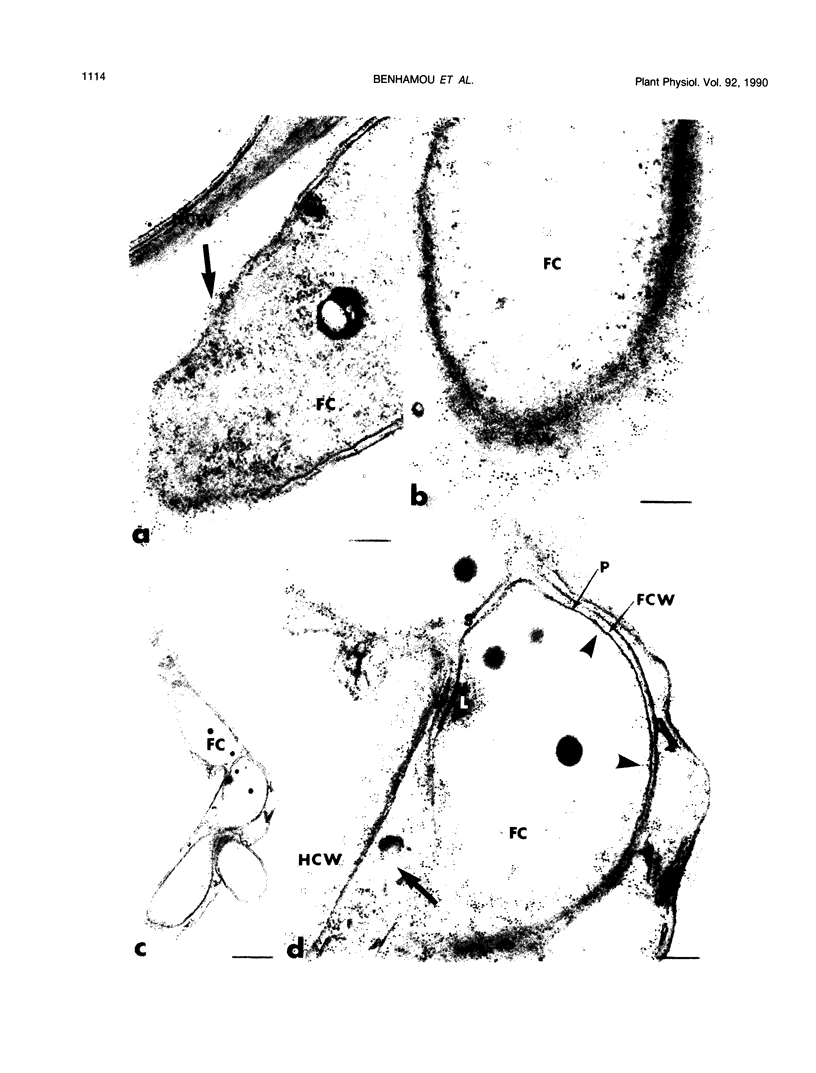
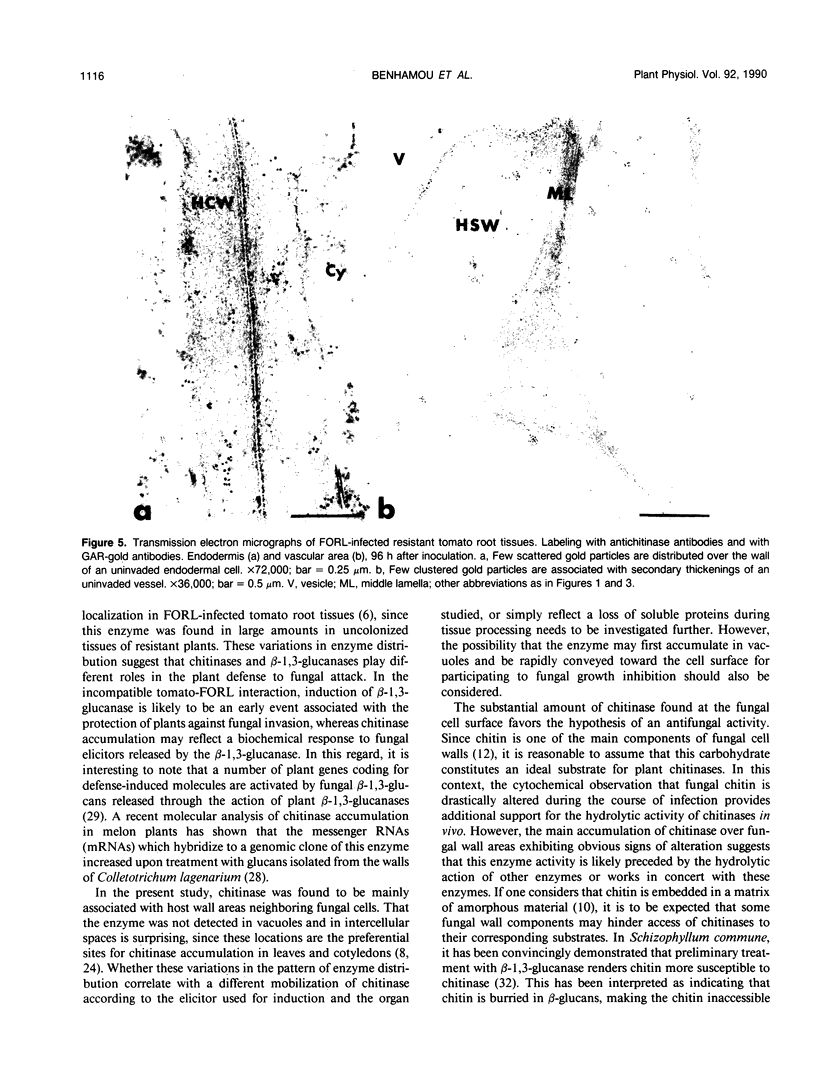
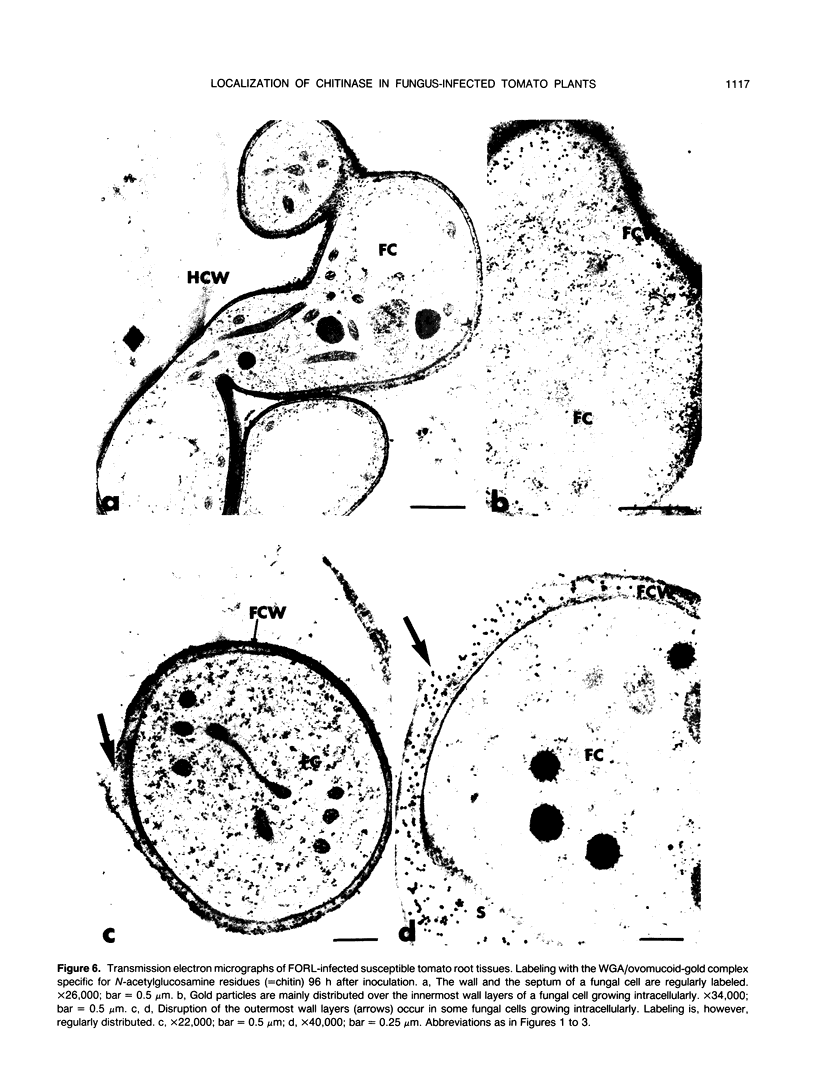
Abstract
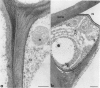
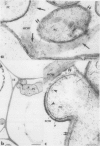
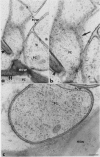
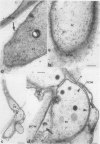
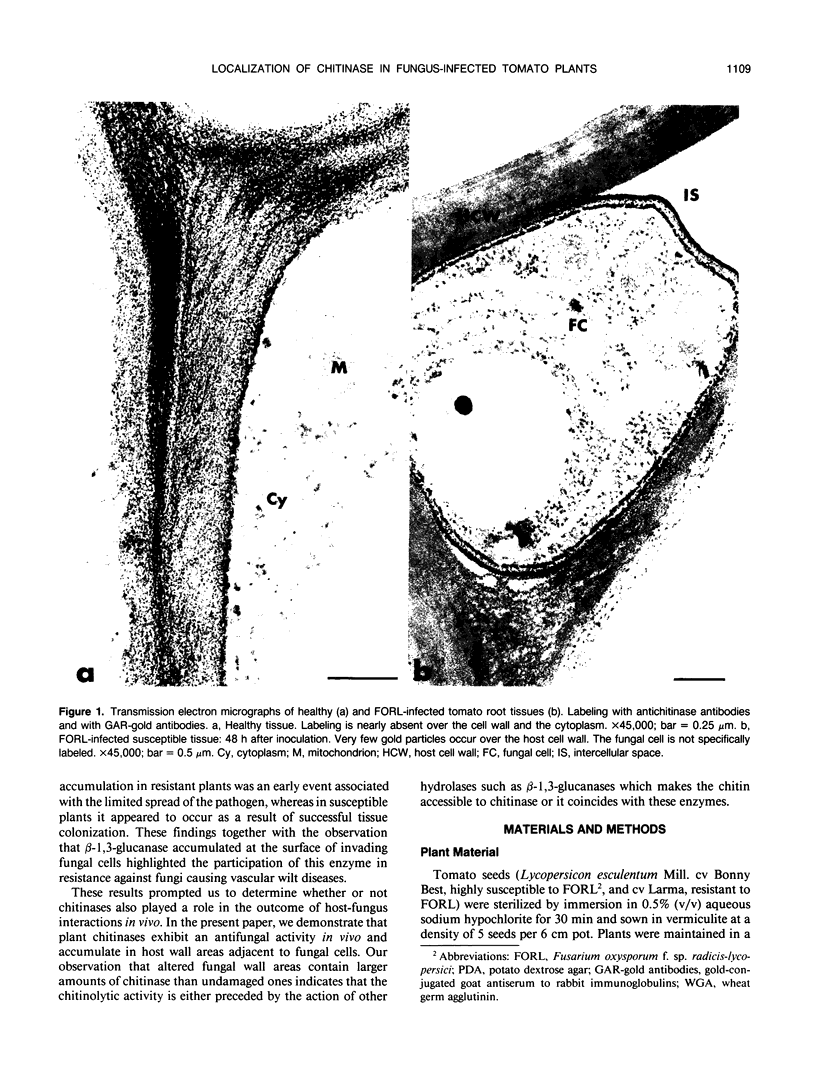
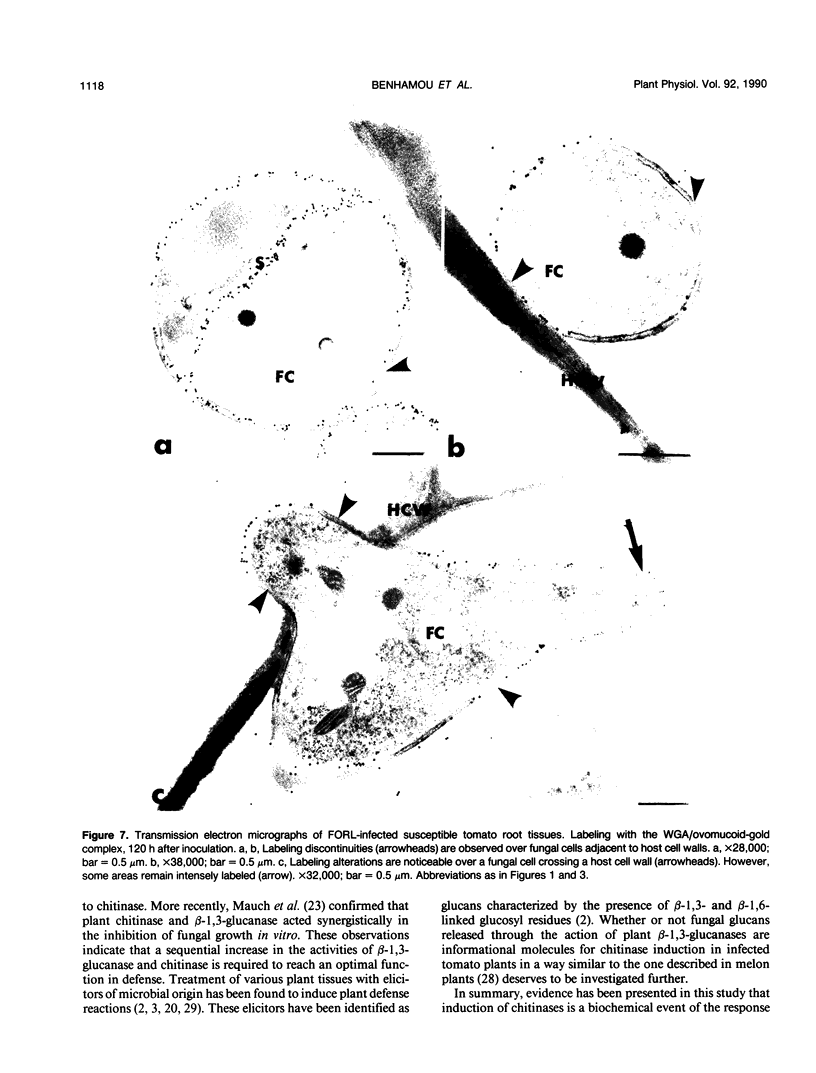
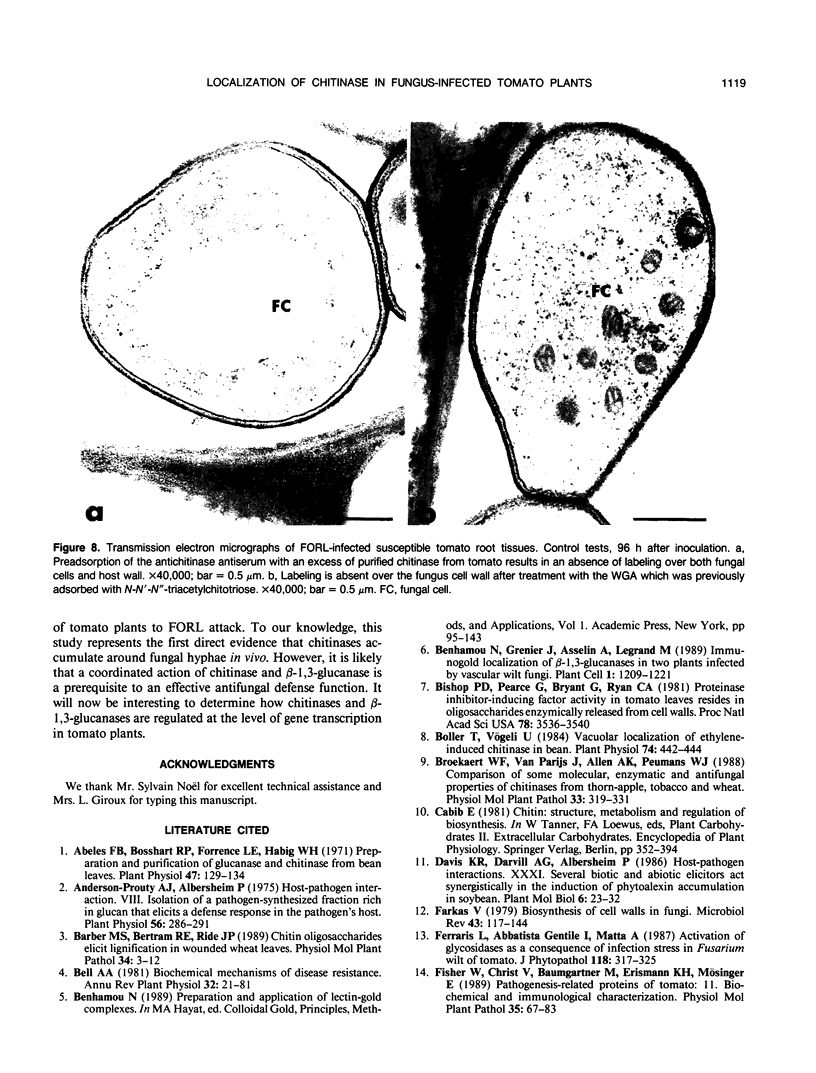
Antiserum raised against a tomato (Lycopersicon esculentum Mill.) chitinase (molecular mass of 26 kilodaltons) was used as a probe to study the subcellular localization of this enzyme in tomato root tissues infected with Fusarium oxysporum f. sp. radicis-lycopersici. A time-course experiment revealed that chitinase accumulated earlier in the incompatible interaction than in the compatible one. However, in both systems, chitinase deposition was largely correlated with pathogen distribution. The enzyme was found to accumulate in areas where host walls were in close contact with fungal cells. In contrast, the enzyme could not be detected in vacuoles and intracellular spaces. The substantial amount of chitinase found at the fungus cell surface supports the view of an antifungal activity. However, the preferential association of the enzyme with altered fungal wall areas indicates that chitinase activity is either preceded by the hydrolytic action of other enzymes such as β-1,3-glucanases or coincides with these enzymes. The possibility that fungal glucans released through the action of β-1,3-glucanases may act as elicitors of chitinase production is discussed.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abeles F. B., Bosshart R. P., Forrence L. E., Habig W. H. Preparation and purification of glucanase and chitinase from bean leaves. Plant Physiol. 1971 Jan;47(1):129–134. doi: 10.1104/pp.47.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson-Prouty A. J., Albersheim P. Host-Pathogen Interactions: VIII. Isolation of a Pathogen-synthesized Fraction Rich in Glucan That Elicits a Defense Response in the Pathogen's Host. Plant Physiol. 1975 Aug;56(2):286–291. doi: 10.1104/pp.56.2.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benhamou N., Grenier J., Asselin A., Legrand M. Immunogold localization of beta-1,3-glucanases in two plants infected by vascular wilt fungi. Plant Cell. 1989 Dec;1(12):1209–1221. doi: 10.1105/tpc.1.12.1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop P. D., Makus D. J., Pearce G., Ryan C. A. Proteinase inhibitor-inducing factor activity in tomato leaves resides in oligosaccharides enzymically released from cell walls. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3536–3540. doi: 10.1073/pnas.78.6.3536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boller T., Vögeli U. Vacuolar localization of ethylene-induced chitinase in bean leaves. Plant Physiol. 1984 Feb;74(2):442–444. doi: 10.1104/pp.74.2.442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farkas V. Biosynthesis of cell walls of fungi. Microbiol Rev. 1979 Jun;43(2):117–144. doi: 10.1128/mr.43.2.117-144.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grandmaison J., Benhamou N., Furlan V., Visser S. A. Ultrastructural localization of N-acetylglucosamine residues in the cell wall of Gigaspora margarita throughout its life-cycle. Biol Cell. 1988;63(1):89–100. [PubMed] [Google Scholar]
- Hadwiger L. A., Beckman J. M. Chitosan as a Component of Pea-Fusarium solani Interactions. Plant Physiol. 1980 Aug;66(2):205–211. doi: 10.1104/pp.66.2.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joosten M. H., De Wit P. J. Identification of Several Pathogenesis-Related Proteins in Tomato Leaves Inoculated with Cladosporium fulvum (syn. Fulvia fulva) as 1,3-beta-Glucanases and Chitinases. Plant Physiol. 1989 Mar;89(3):945–951. doi: 10.1104/pp.89.3.945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keen N. T., Yoshikawa M. beta-1,3-Endoglucanase from Soybean Releases Elicitor-Active Carbohydrates from Fungus Cell Walls. Plant Physiol. 1983 Mar;71(3):460–465. doi: 10.1104/pp.71.3.460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kombrink E., Schröder M., Hahlbrock K. Several "pathogenesis-related" proteins in potato are 1,3-beta-glucanases and chitinases. Proc Natl Acad Sci U S A. 1988 Feb;85(3):782–786. doi: 10.1073/pnas.85.3.782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb C. J., Lawton M. A., Dron M., Dixon R. A. Signals and transduction mechanisms for activation of plant defenses against microbial attack. Cell. 1989 Jan 27;56(2):215–224. doi: 10.1016/0092-8674(89)90894-5. [DOI] [PubMed] [Google Scholar]
- Legrand M., Kauffmann S., Geoffroy P., Fritig B. Biological function of pathogenesis-related proteins: Four tobacco pathogenesis-related proteins are chitinases. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6750–6754. doi: 10.1073/pnas.84.19.6750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauch F., Hadwiger L. A., Boller T. Antifungal Hydrolases in Pea Tissue : I. Purification and Characterization of Two Chitinases and Two beta-1,3-Glucanases Differentially Regulated during Development and in Response to Fungal Infection. Plant Physiol. 1988 Jun;87(2):325–333. doi: 10.1104/pp.87.2.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauch F., Mauch-Mani B., Boller T. Antifungal Hydrolases in Pea Tissue : II. Inhibition of Fungal Growth by Combinations of Chitinase and beta-1,3-Glucanase. Plant Physiol. 1988 Nov;88(3):936–942. doi: 10.1104/pp.88.3.936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauch F., Staehelin L. A. Functional Implications of the Subcellular Localization of Ethylene-Induced Chitinase and [beta]-1,3-Glucanase in Bean Leaves. Plant Cell. 1989 Apr;1(4):447–457. doi: 10.1105/tpc.1.4.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan C. A. Oligosaccharide signalling in plants. Annu Rev Cell Biol. 1987;3:295–317. doi: 10.1146/annurev.cb.03.110187.001455. [DOI] [PubMed] [Google Scholar]