Abstract
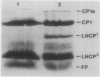
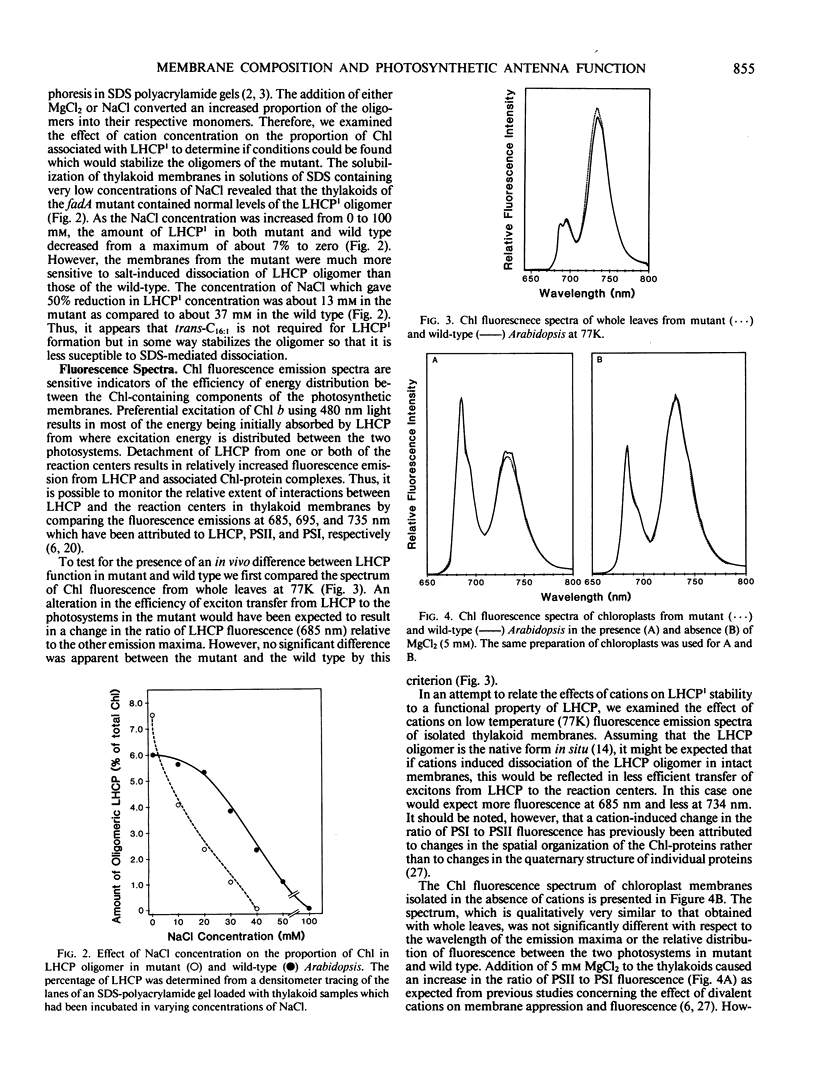
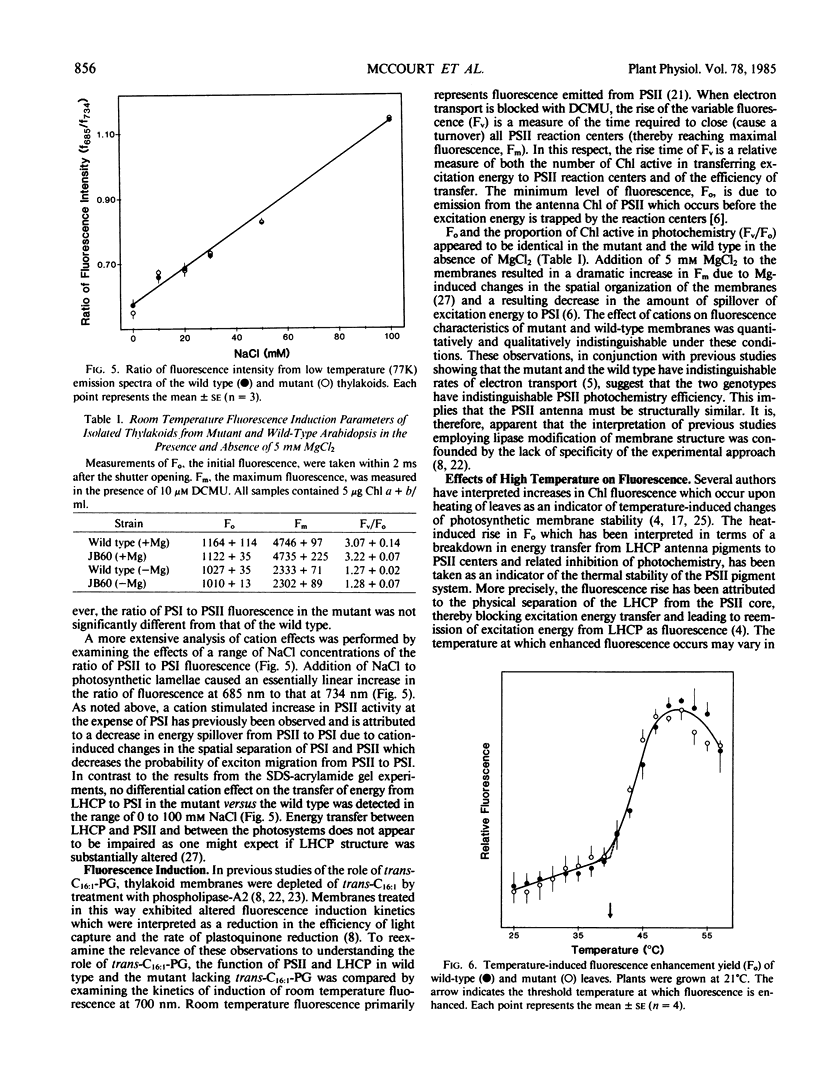
Several lines of evidence support the proposal that the unusual chloroplast-specific lipid acyl group Δ3,trans-hexadecenoic acid (trans-C16:1) stimulates the formation or maintenance of the oligomeric form of the light-harvesting chlorophyll a/b complex (LHCP). To assess the functional significance of this apparent association we have analyzed LHCP structure and function in a mutant of Arabidopsis thaliana (L.) which lacks trans-C16:1 by electrophoretic analysis of the protein-chlorophyll complexes and by measurements of chlorophyll fluorescence under a variety of conditions. By these criteria the putative oligomeric form of LHCP appears to be slightly more labile to detergent-mediated dissociation in the mutant. The oligomeric PSI chlorophyll-protein complex, associated with PSI, was also more labile to detergent-mediated dissociation in the mutant, suggesting a previously unsuspected association of trans-C16:1 with the PSI complex. However, no significant effect of the mutation on the efficiency of energy transfer from LHCP to the photochemical reaction centers was observed under any of the various conditions imposed. Also, the stability of the chlorophyll-protein complexes to temperature-induced dissociation was unaffected in the mutant. The role of trans-C16:1 is very subtle or is only conditionally expressed.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Armond P. A., Schreiber U., Björkman O. Photosynthetic Acclimation to Temperature in the Desert Shrub, Larrea divaricata: II. Light-harvesting Efficiency and Electron Transport. Plant Physiol. 1978 Mar;61(3):411–415. doi: 10.1104/pp.61.3.411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browse J., McCourt P., Somerville C. R. A mutant of Arabidopsis lacking a chloroplast-specific lipid. Science. 1985 Feb 15;227(4688):763–765. doi: 10.1126/science.227.4688.763. [DOI] [PubMed] [Google Scholar]
- Izawa S., Good N. E. Effect of Salts and Electron Transport on the Conformation of Isolated Chloroplasts. II. Electron Microscopy. Plant Physiol. 1966 Mar;41(3):544–552. doi: 10.1104/pp.41.3.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lam E., Oritz W., Mayfield S., Malkin R. Isolation and Characterization of a Light-Harvesting Chlorophyll a/b Protein Complex Associated with Photosystem I. Plant Physiol. 1984 Mar;74(3):650–655. doi: 10.1104/pp.74.3.650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch D. V., Thompson G. A. Chloroplast Phospholipid Molecular Species Alterations during Low Temperature Acclimation in Dunaliella. Plant Physiol. 1984 Feb;74(2):198–203. doi: 10.1104/pp.74.2.198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawyler A., Siegenthaler P. A. Transmembrane distribution of phospholipids and their involvement in electron transport, as revealed by phospholipids A2 treatment of spinach thylakoids. Biochim Biophys Acta. 1981 Apr 13;635(2):348–358. doi: 10.1016/0005-2728(81)90033-5. [DOI] [PubMed] [Google Scholar]
- Staehelin L. A., Arntzen C. J. Regulation of chloroplast membrane function: protein phosphorylation changes the spatial organization of membrane components. J Cell Biol. 1983 Nov;97(5 Pt 1):1327–1337. doi: 10.1083/jcb.97.5.1327. [DOI] [PMC free article] [PubMed] [Google Scholar]