Abstract
Objective
To systematically summarise and evaluate the existing evidence on the effect of diet on the management of type 2 diabetes and prevention of complications.
Design
Umbrella review of systematic reviews with meta-analyses of randomised controlled trials.
Data sources
PubMed, Embase, Epistemonikos, and Cochrane, from inception up to 5 June 2022.
Eligibility criteria for selecting studies
Systematic reviews with meta-analyses of randomised controlled trials reporting summary effect estimates on the effect of diet on any health outcome in populations with type 2 diabetes were included in the review. Only meta-analyses with randomised controlled trials with the duration of at least 12 weeks were eligible for inclusion. Summary data were extracted by two investigators independently. Summary effect estimates with 95% confidence intervals were recalculated with a random effects model if the information provided was insufficient. Methodological quality was assessed with the A MeaSurement Tool to Assess systematic Reviews (AMSTAR) 2 tool and the certainty of evidence with the Grading of Recommendations Assessment, Development, and Evaluations (GRADE) approach.
Results
88 publications with 312 meta-analyses of randomised controlled trials were included. Methodological quality was high to moderate in 23% and low to very low in 77% of the included publications. A high certainty of evidence was found for the beneficial effects of liquid meal replacement on reducing body weight (mean difference −2.37 kg, 95% confidence interval −3.30 to −1.44; n=9 randomised controlled trials included in the meta-analysis) and body mass index (−0.87, −1.32 to −0.43; n=8 randomised controlled trials), and of a low carbohydrate diet (<26% of total energy) on levels of haemoglobin A1c (−0.47%, −0.60% to −0.34%; n=17 randomised controlled trials) and triglycerides (−0.30 mmol/L, −0.43 to −0.17; n=19 randomised controlled trials). A moderate certainty of evidence was found for the beneficial effects of liquid meal replacement, plant based, Mediterranean, high protein, low glycaemic index, and low carbohydrate diets (<26% total energy) on various cardiometabolic measures. The remaining results had low to very low certainty of evidence.
Conclusions
The evidence indicated that diet has a multifaceted role in the management of type 2 diabetes. An energy restricted diet can reduce body weight and improve cardiometabolic health. Beyond energy restriction, dietary approaches such as plant based, Mediterranean, low carbohydrate (<26% total energy), or high protein diets, and a higher intake of omega 3 fatty acids can be beneficial for cardiometabolic health in individuals with type 2 diabetes.
Systematic review registration
PROSPERO CRD42021252309.
Keywords: Diabetes mellitus, Dietetics, Nutritional sciences, Epidemiology, Clinical trial
WHAT IS ALREADY KNOWN ON THIS TOPIC
Type 2 diabetes is a major global health problem that can cause further health complications
Diet is essential in managing type 2 diabetes and preventing further diabetes related complications
To provide evidence based dietary recommendations for people with type 2 diabetes, the existing evidence needs to be systematically summarised and evaluated with established tools
WHAT THIS STUDY ADDS
This umbrella review of systematic reviews with meta-analyses of randomised controlled trials identified the dietary factors that can improve surrogate markers of disease and health in people with type 2 diabetes
Robust evidence indicated that in addition to energy restriction, dietary approaches, such as plant based, Mediterranean, low carbohydrate (<26% total energy), and high protein diets are beneficial for cardiometabolic health in individuals with type 2 diabetes
HOW THIS STUDY MIGHT AFFECT RESEARCH, PRACTICE, OR POLICY
This study identified the dietary recommendations that can be effective in improving surrogate markers of disease and health in people with type 2 diabetes
More randomised controlled trials with long term interventions, focusing exclusively on complications related to type 2 diabetes, are needed to provide robust evidence based recommendations for other dietary factors in the management of type 2 diabetes
Introduction
Type 2 diabetes is a major global health problem. In 2021, the International Diabetes Federation estimated that a total of 536.6 million individuals worldwide lived with diabetes, and this number is expected to increase.1 Type 2 diabetes carries a huge financial burden on health systems and people with diabetes.2 3 Type 2 diabetes is typically associated with obesity, abnormalities of lipid metabolism, and an increased risk of numerous complications and comorbidities.4 5 These include cardiovascular and liver disease, kidney disease, diabetes related eye disease, and lower limb amputations.6 7 Also, people with type 2 diabetes have about a 2.5 times higher risk of all cause mortality than people without diabetes.8 9
Lifestyle factors, such as diet, form the basis of the management of type 2 diabetes,1 4 10 and over the past decades, the effects of different diets on managing type 2 diabetes have been studied. Meta-analyses of randomised controlled trials showed that Mediterranean,11 vegetarian,12 and ketogenic diets13 lowered levels of haemoglobin A1c (HbA1c), whereas a higher intake of dietary fibre14 and soy isoflavones,15 and magnesium supplementation16 improved concentrations of blood lipids. These findings suggest that changes in diet might be important for preventing further complications and progression of disease in individuals with type 2 diabetes. To provide evidence based dietary recommendations, however, the existing evidence needs to be systematically summarised and evaluated based on state-of-the-art approaches.
So far, several umbrella reviews have been conducted summarising the evidence on selected dietary factors and specific outcomes in diabetes.17–20 None of the previous umbrella reviews, however, included meta-analyses with randomised controlled trials lasting at least 12 weeks,17–20 not all focused exclusively on people with type 2 diabetes,18 and not all assessed the certainty of evidence with the Grading of Recommendations Assessment, Development, and Evaluations (GRADE) approach.19 There is an urgent need to summarise all available evidence on this topic and, most importantly, to assess the certainty of evidence using the GRADE approach. Our aim in this umbrella review was to systematically summarise the most recent evidence on dietary interventions, including dietary patterns, food groups, and dietary supplements, on health outcomes in people with type 2 diabetes. The evidence was derived from systematic reviews with meta-analyses of randomised controlled trials that lasted at least 12 weeks. We also evaluated the certainty of evidence of these effects.
Methods
This umbrella review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines21 and the preferred reporting items for overviews of reviews (PRIOR statement) for healthcare interventions.22
Literature search
A systematic literature search was conducted in PubMed, Embase, Cochrane, and Epistemonikos, with predefined search terms, from inception to 24 April 2021, with an update on 23 May 2022. None of the studies included in the in 2021 screening were exclusively from Embase, and therefore this database was omitted from the update in 2022. No filters were applied during the literature search process. Online supplemental table S1 shows the detailed search strategies. We also reviewed studies received from PubMed Alert at weekly intervals up to 5 June 2022 and screened the reference lists of all included publications. The literature search originally identified meta-analyses of prospective cohort studies and randomised controlled trials, but for this study, we focused on meta-analyses of randomised controlled trials. The literature screening was conducted by two authors independently. Disagreements were resolved by consensus with a third author.
bmjmed-2023-000664supp001.pdf (6.3MB, pdf)
Eligibility criteria
The systematic reviews with (network) meta-analyses of randomised controlled trials included in our study met the following criteria: the effects of dietary factors (dietary patterns, food groups, and dietary supplements) were investigated; the intervention in randomised controlled trials lasted ≥12 weeks for any health outcome (eg, mortality, incidence of cardiovascular disease, body weight, levels of HbA1c, and heath related quality of life); only individuals with type 2 diabetes were included; effect estimates were reported (eg, for continuous outcomes, mean difference or standardised mean difference with corresponding 95% confidence intervals, or standard error; or for dichotomous outcomes, risk ratio, including hazard ratio or odd ratio, with 95% confidence intervals).
We excluded studies that were not systematic (including narrative reviews), not meta-analyses, not based exclusively on populations with type 2 diabetes, and single study findings. We also excluded publications reporting on alternative dietary treatments or medical nutrition (eg, Chinese medicine, plant extracts, herbs, and Ramadan fasting), and meta-analyses of randomised controlled trials with exclusively active control groups (comparing two intervention arms with each other). We did not exclude studies based on the publication language.
If we identified more than one meta-analysis on the same effect, we chose one meta-analysis for each intervention to avoid inclusion of duplicate results. In this case, we included the meta-analysis with the largest number of randomised controlled trials and participants. If more than one meta-analysis had the same number of randomised controlled trials, we selected the meta-analysis with the largest number of participants. Because we only included meta-analyses that were based on a systematic literature search, we assumed that newer and larger meta-analyses included the same randomised controlled trials as older and smaller meta-analyses looking at the same research question. We always prioritised evidence from direct comparisons over network meta-analyses.
Data extraction
Data were extracted by one author and double checked by a second author. Discrepancies were discussed between the authors, involving a third author if necessary. Data extracted from each publication were name of the first author, publication year, intervention type, comparison, outcome, duration of the study, number and study design of randomised controlled trials, and number of participants in the intervention groups. Online supplemental table S2 lists the definitions and cut-off values for categorising dietary patterns in terms of the amount of energy derived from macronutrients. We also extracted summary effect estimates (mean difference, standardised mean difference, or risk ratio) with corresponding 95% confidence intervals, the statistical model used (fixed effect or random effects model), a measure of inconsistency between randomised controlled trials (I2), publication bias, risk of bias, and certainty of evidence (if assessed according to the GRADE approach). Online supplemental table S2 has further details on data extraction.
Assessment of methodological quality and certainty of evidence
The methodological quality of each included publication was assessed with the validated A MeaSurement Tool to Assess systematic Reviews (AMSTAR) 2 by two reviewers independently.23 Online supplemental table S3 lists a detailed description of the tool, included domains, and grading system.
The certainty of evidence was evaluated with the GRADEpro tool.24 25 GRADE rates the certainty of evidence as high, moderate, low, or very low. A high certainty of evidence means that it is very likely that the true effect lies on one side of a specified threshold or within a chosen range, a moderate certainty of evidence indicates moderate confidence that the true effect lies on one side of a specified threshold or within a chosen range and further studies might change the results, and low or very low certainty of evidence means that confidence in the meta-evidence available is limited.26 27 Three reviewers, in pairs of two, independently rated the certainty of the evidence. Online supplemental table S4 lists a detailed description of the grading of the certainty of evidence.
Data analysis
For meta-analyses that used a random effects model and provided I² values and forest plots, we extracted the reported summary effect estimates with 95% confidence intervals. For meta-analyses that used a fixed effect model, or did not provide I2 values or forest plots, we recalculated the mean difference, standardised mean difference, and summary risk ratio with corresponding 95% confidence intervals with the random effects model by DerSimonian and Laird.28 Information on small study effects and publications bias was extracted from the included publications or, if not reported, was assessed by funnel plots and Egger's test (if ≥10 studies were included).29 30 Online supplemental table S5 presents further details on the statistical methods. All analyses were conducted with Stata version 14.1.
Patient and public involvement
No patients or public were involved in setting the research question, the design of the study, the interpretation of the results, or the writing of the manuscript. The results of this study will be spread to the public via social media or press release.
Results
Literature search
Of 25 221 publications, 764 eligible articles were retrieved for screening of the full text of the article (online supplemental figure S1). Online supplemental table S6 lists the excluded studies and the reasons for exclusion. We identified 88 eligible publications of systematic reviews with meta-analyses of randomised controlled trials for inclusion in our umbrella review.
Description of published meta-analyses
Online supplemental table S7 presents the characteristics and results of all of the included systematic reviews with meta-analyses of randomised controlled trials. Eighty eight publications, with 310 meta-analyses of randomised controlled trials, investigated the effectiveness of dietary patterns (liquid meal replacement, energy, carbohydrate, and fat restriction, low glycaemic index and glycaemic load, high protein, Mediterranean, plant based, vegetarian, and ketogenic diets, and intermittent fasting), foods (mixed nuts, tree nuts, walnuts, almonds, blueberries and cranberries, olive oil, cocoa products, cinnamon, curcumin, ginger, soy and soy isoflavones, and non-nutritive sweeteners), nutrients (monounsaturated fatty acids, omega 3 fatty acids, α-lipolenic acid, L-carnitine, and salt), micronutrients (magnesium, chromium, and zinc), vitamins (niacin, and vitamins E, C, and D), phytochemicals (anthocyanins, flavonoids, polyphenols, and resveratrol), fibre (psyllium, soluble fibre, and guar gum), and probiotics (online supplemental table S7).
Outcomes included anthropometric, glycaemic, and lipid markers, blood pressure, kidney, liver, and inflammation parameters, as well as quality of life, reduced use of drug treatments, constipation, and major cardiovascular events. The number of participants in meta-analyses of randomised controlled trials ranged from 44 to 28 199, and intervention time from 12 weeks to 96 months. In total, 293 of the included meta-analyses were pairwise meta-analyses and 17 were network meta-analyses,12 31 and 15% of the meta-analyses included ≥10 randomised controlled trials. Publication bias was detected for 2.2% of the results of meta-analyses of randomised controlled trials, but 59.4% of meta-analyses included ≤5 studies (online supplemental table S7).
Methodological quality was rated as high for 18% (n=16), moderate for 5% (n=4), low for 22% (n=19), and critically low for the remaining 55% (n=49) of publications (online supplemental table S8). The most common reasons for downgrading the methodological quality were missing study protocol, inappropriate methods for the search strategy (literature search only by one investigator, search strategy not shown, or included reports restricted to the English language), and lack of evaluation or discussion, or both, of the effect of risk of bias on the results.
Evidence for the effect of diet on managing type 2 diabetes: meta-analyses of randomised controlled trials
The certainty of evidence was rated as high for seven (2%), moderate for 40 (13%), low for 120 (39%), and very low for 143 (46%) outcomes (online supplemental tables S9–S32). Table 1 summarises the results of the meta-analyses with high to moderate certainty of evidence and with clinically meaningful changes. Figures 1–6 show the summary effect estimates of the included meta-analyses of randomised controlled trials with high and moderate certainty of evidence. Online supplemental figures S2–S10 show the results of meta-analyses of randomised controlled trials with low and very low certainty of evidence.
Table 1.
Summary of results with high to moderate certainty of evidence from umbrella review of systematic reviews with meta-analyses of randomised controlled trials on diet in the management of type 2 diabetes
| Outcomes | Type of dietary intervention | Effect estimates (mean difference, risk ratio, or risk difference (95% CI)) |
Certainty of evidence* |
| Changes within the clinically meaningful range | |||
| Body weight (kg) |
|
−2.37 (−3.30 to −1.44) | High ⨁⨁⨁⨁ |
|
−1.34 (−1.77 to −0.91) | Moderate ⨁⨁⨁〇 | |
| Body mass index |
|
−0.87 (−1.32 to −0.43) | High ⨁⨁⨁⨁ |
|
−1.13 (−1.88 to −0.38) | Moderate ⨁⨁⨁〇 | |
| Waist circumference (cm) |
|
−2.24 (−3.71 to −0.76) | Moderate ⨁⨁⨁〇 |
|
−2.41 (−3.50 to −1.32) | Moderate ⨁⨁⨁〇 | |
| Haemoglobin A1c (%) |
|
−0.47 (−0.60 to −0.34) | High ⨁⨁⨁⨁ |
| Fasting blood glucose (mmol/L) |
|
−0.63 (−0.99 to −0.27) | Moderate ⨁⨁⨁〇 |
|
−1.18 (−1.47 to −0.88) | Moderate ⨁⨁⨁〇 | |
|
−1.78 (−2.33 to −1.23) | Moderate ⨁⨁⨁〇 | |
| Fasting insulin (µIU/mL) |
|
−1.97 (−3.88 to −0.07) | Moderate ⨁⨁⨁〇 |
| HOMA-IR |
|
−0.82 (−1.38 to −0.25) | Moderate ⨁⨁⨁〇 |
|
−0.90 (−1.34 to −0.46) | Moderate ⨁⨁⨁〇 | |
|
−1.08 (−1.86 to −0.30) | Moderate ⨁⨁⨁〇 | |
| Triglycerides (mmol/L) |
|
−0.30 (−0.43 to −0.17) | High ⨁⨁⨁⨁ |
|
−0.41 (−0.72 to −0.10) | Moderate ⨁⨁⨁〇 | |
| Total cholesterol (mmol/L) |
|
−0.21 (−0.31 to −0.11) | Moderate ⨁⨁⨁〇 |
|
−0.19 (−0.33 to −0.05) | Moderate ⨁⨁⨁〇 | |
|
−0.24 (−0.31 to −0.16) | Moderate ⨁⨁⨁〇 | |
| Low density lipoprotein cholesterol (mmol/L) |
|
−0.10 (−0.18, -0.02) | Moderate ⨁⨁⨁〇 |
|
−0.23 (−0.37 to −0.09) | Moderate ⨁⨁⨁〇 | |
|
−0.18 (−0.31 to −0.05) | Moderate ⨁⨁⨁〇 | |
| High density lipoprotein cholesterol (mmol/L) |
|
0.06 (0.01 to 0.10) | Moderate ⨁⨁⨁〇 |
| Systolic blood pressure (mm Hg) |
|
−4.97 (−7.32 to −2.62) | Moderate ⨁⨁⨁〇 |
| Diastolic blood pressure (mm Hg) |
|
−1.98 (−3.05 to −0.91) | Moderate ⨁⨁⨁〇 |
| Reduced use of drug treatments |
|
Risk ratio 0.24 (0.12 to 0.35) | Moderate ⨁⨁⨁〇 |
| Changes not within the clinically meaningful range | |||
| Body weight (kg) |
|
−0.25 (−0.99 to 0.49) | Moderate ⨁⨁⨁〇 |
| Body mass index |
|
−0.36 (−0.53 to −0.20) | Moderate ⨁⨁⨁〇 |
| Haemoglobin A1c (%) |
|
−0.11 (−0.17 to −0.04) | High ⨁⨁⨁⨁ |
|
0.02 (−0.13 to 0.17) | Moderate ⨁⨁⨁〇 | |
| Fasting blood glucose (mmol/L) |
|
−0.03 (−0.17 to 0.12) | Moderate ⨁⨁⨁〇 |
| QUICKI |
|
0.02 (0.01 to 0.03) | Moderate ⨁⨁⨁〇 |
| Triglycerides (mmol/L) |
|
−0.12 (−0.22 to −0.01) | High ⨁⨁⨁⨁ |
|
−0.13 (−0.24 to −0.02) | Moderate ⨁⨁⨁〇 | |
| Low density lipoprotein cholesterol (mmol/L) |
|
0.02 (−0.10 to 0.14) | Moderate ⨁⨁⨁〇 |
|
−0.03 (−0.08 to 0.02) | Moderate ⨁⨁⨁〇 | |
|
−0.26 (−0.43 to −0.09) | Moderate ⨁⨁⨁〇 | |
| Non-high density lipoprotein cholesterol (mmol/L) |
|
−0.02 (−0.11 to 0.07) | Moderate ⨁⨁⨁〇 |
| High density lipoprotein cholesterol (mmol/L) |
|
−0.04 (−0.08 to −0.00) | Moderate ⨁⨁⨁〇 |
|
−0.02 (−0.07 to 0.03) | Moderate ⨁⨁⨁〇 | |
|
0.01 (−0.03 to 0.05) | Moderate ⨁⨁⨁〇 | |
| Systolic blood pressure (mm Hg) |
|
0.03 (−1.77 to 1.84) | Moderate ⨁⨁⨁〇 |
|
−0.11 (−1.10 to 0.88) | Moderate ⨁⨁⨁〇 | |
| Diastolic blood pressure (mm Hg) |
|
−0.21 (−1.21 to 0.79) | Moderate ⨁⨁⨁〇 |
| Blood urea nitrogen (mg/dL) |
|
−1.22 (−1.94 to −0.51) | Moderate ⨁⨁⨁〇 |
| C reactive protein (standardised mean difference) |
|
−0.87 (−1.11 to −0.63) | Moderate ⨁⨁⨁〇 |
| Major cardiovascular events |
|
Risk difference 0.94 (0.87 to 1.02) | High ⨁⨁⨁⨁ |
CI=confidence interval; HOMA-IR=homeostatic model assessment-insulin resistance; QUICKI=quantitative insulin sensitivity check index.
*Assessed with the GRADEpro tool.
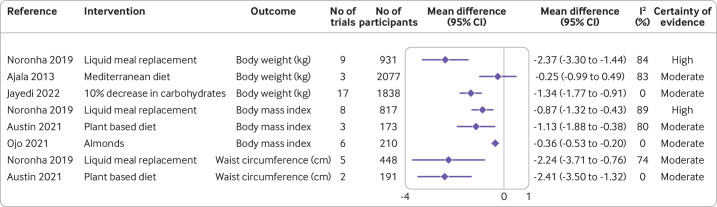
Figure 1.
Effect of different dietary factors on anthropometric measures (body weight, body mass index, and waist circumference) in people with type 2 diabetes.32–36 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval
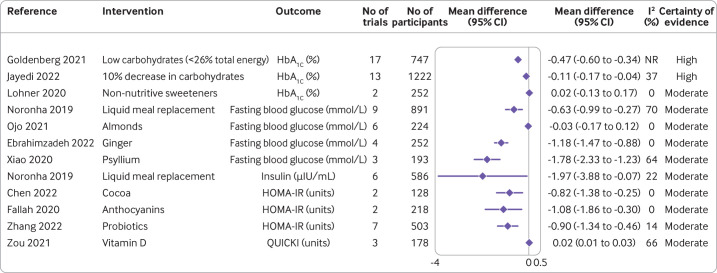
Figure 2.
Effect of different dietary factors on glycaemic measures in people with type 2 diabetes.14 32 34 36–41 61 62 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval; HbA1c=haemoglobin A1c; HOMA-IR=homeostatic model assessment-insulin resistance; NR=not reported; QUICKI=quantitative insulin sensitivity check index
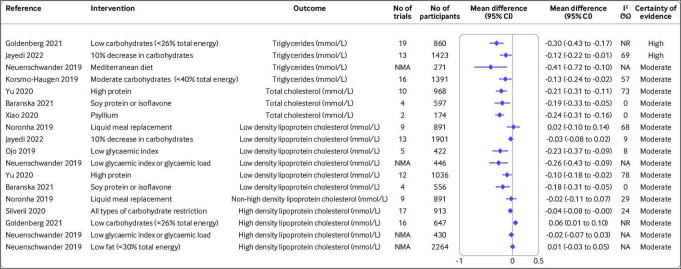
Figure 3.
Effect of different dietary factors on blood lipid measures in people with type 2 diabetes.14 15 31 32 34 37 42–44 63 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval; NA=not available; NR=not reported; NMA=network meta-analysis
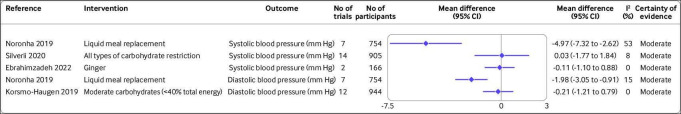
Figure 4.
Effect of different dietary factors on blood pressure in people with type 2 diabetes.32 38 42 63 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval
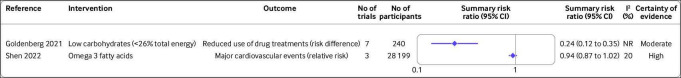
Figure 5.
Effect of different dietary factors on risk reduction in people with type 2 diabetes.37 45 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval; NR=not reported
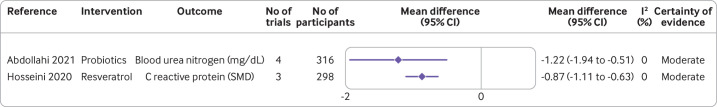
Figure 6.
Effect of different dietary factors on other outcomes in people with type 2 diabetes.46 47 Results from meta-analyses of randomised controlled trials with high to moderate certainty of evidence. CI=confidence interval; SMD=standardised mean difference
Anthropometric measures
The certainty of evidence was high for the efficacy of liquid meal replacement on clinically relevant reductions in body weight and body mass index (figure 1). Liquid meal replacement reduced body weight by 2.37 kg (95% confidence interval −3.30 to −1.44; I2=84%; n=9 randomised controlled trials) and body mass index by 0.87 (−1.32 to −0.43; I2=89%; n=8 randomised controlled trials) compared with a control diet.32 Plant based diets also effectively reduced body mass index by 1.13 (−1.88 to −0.38; I2=80%; n=3 randomised controlled trials) with moderate certainty of evidence.33
Moderate certainty of evidence was found for a reduction in waist circumference after liquid meal replacement (−2.24 cm, 95% confidence interval −3.71 to −0.76; I2=74%; n=5 randomised controlled trials) and plant based diets (−2.41 cm, −3.50 to −1.32; I2=0%; n=2 randomised controlled trials).32 33 Improvement in anthropometric measures was also seen for a decrease in carbohydrate intake of 10%,34 adherence to a Mediterranean diet,35 and increased intake of almonds,36 but effect estimates were small and not clinically relevant (figure 1). The certainty of evidence for the effect of other dietary factors was low to very low (online supplemental figure S2).
Glycaemic markers
A low carbohydrate diet (<26% total energy) produced a clinically relevant reduction in levels of HbA1c of 0.47% (95% confidence interval −0.60% to −0.34%; I2 not reported; n=17 randomised controlled trials) with a high certainty of evidence (figure 2).37 HbA1c levels were also reduced after a decrease in carbohydrate intake of 10%,34 but the effect estimate was small and not clinically relevant.
A moderate certainty of evidence was found for reduced concentrations of fasting blood glucose after liquid meal replacement (−0.63 mmol/L, 95% confidence interval −0.99 to −0.27; I2=70%; n=9 randomised controlled trials), and supplementation with ginger (−1.18 mmol/L, −1.47 to −0.88; I2=0%; n=4 randomised controlled trials) and psyllium (−1.78 mmol/L, −2.33 to −1.23; I2=64%; n=3 randomised controlled trials).14 32 38 Liquid meal replacement reduced insulin concentrations by 1.97 µIU/mL (−3.88 to −0.07; I2=22%; n=6 randomised controlled trials), with moderate certainty of evidence.32 Supplementation with cocoa, probiotics, and anthocyanins decreased the homeostatic model assesment-insulin resistance (HOMA-IR) index with moderate certainty of evidence by −0.82 units (−1.38 to −0.25; I2=0%; n=2 randomised controlled trials), −0.90 units (−1.34 to −0.46; I2=14%; n=7 randomised controlled trials), and −1.08 units (−1.86 to −0.30; I2=0%; n=2 randomised controlled trials), respectively.39–41 The certainty of evidence for the effect of other dietary factors was low to very low (online supplemental figure S3).
Blood lipids
The certainty of evidence was high for a decrease in carbohydrate intake of 10% and low carbohydrate diet (<26% total energy) to lower concentrations of triglycerides by 0.12 mmol/L (95% confidence interval −0.22 to −0.01; I2=69%; n=13 randomised controlled trials) and 0.30 mmol/L (−0.43 to −0.17; I2 not reported; n=19 randomised controlled trials), respectively (figure 3).34 37 Moderate carbohydrate restriction (<45% total energy) and a Mediterranean diet reduced concentrations of triglycerides by −0.13 mmol/L (−0.24 to −0.02; I2=57%; n=16 randomised controlled trials) and −0.41 mmol/L (−0.72 to −0.10; I2 not available; network meta-analysis), respectively, with moderate certainty of evidence.31 42 A moderate certainty of evidence was found for reduction in total cholesterol concentrations after a high protein diet (mean difference −0.21 mmol/L, −0.31 to −0.11; I2=73%; n=10 randomised controlled trials), supplementation with psyllium (mean difference −0.24 mmol/L, −0.31 to −0.16; I2=0%; n=2 randomised controlled trials), and soy protein (mean difference −0.19 mmol/L, −0.33 to −0.05; I2=0%; n=4 randomised controlled trials).14 15 43
The certainty of evidence was moderate for the low density lipoprotein cholesterol lowering effect of a low glycaemic index diet (mean difference −0.23 mmol/L, 95% confidence interval −0.37 to −0.09; I2=8%; n=5 randomised controlled trials), a high protein diet (mean difference −0.10 mmol/L, −0.18 to −0.02; I2=78%; n=12 randomised controlled trials), and a higher intake of soy protein (mean difference −0.18 mmol/L, −0.31 to −0.05; I2=0%; n=4 randomised controlled trials).15 43 44 A low carbohydrate diet (<26% total energy) might increase concentrations of high density lipoprotein cholesterol with moderate certainty of evidence (mean difference 0.06 mmol/L, 0.01 to 0.10; I2 not reported; n=16 randomised controlled trials).37 Evidence for the effect of other dietary factors was low to very low (online supplemental figure S4).
Blood pressure
Liquid meal replacement diets reduced both systolic and diastolic blood pressure by 4.97 mm Hg (95% confidence interval −7.32 to −2.62; I2=53%; n=7 randomised controlled trials) and 1.98 mm Hg (−3.05 to −0.91; I2=15%; n=7 randomised controlled trials), respectively, with moderate certainty of evidence (figure 4).32 Evidence for the effect of other dietary factors was low to very low (online supplemental figure S5).
Other outcomes
A high certainty of evidence was found for supplementation with omega 3 fatty acids and risk of major cardiovascular events, but the 95% confidence interval was imprecisely estimated and the risk reduction was marginal (figure 5; summary risk ratio 0.94, 0.87 to 1.02; I2=20%; n=3 randomised controlled trials).45 A low carbohydrate diet (<26% total energy) reduced the use of drug treatments by an additional 24 per 100 individuals (risk difference 0.24, 0.12 to 0.35; I2 not reported; n=7 randomised controlled trials, moderate certainty of evidence).37 Probiotic supplements reduced concentrations of blood urea nitrogen, a marker of kidney function (mean difference −1.22 mg/dL, −1.94 to −0.51; I2=0%; n=4 randomised controlled trials). Supplementation with resveratrol reduced concentrations of C reactive protein (standardised mean difference −0.87, −1.11 to −0.63; I2=0%; n=3 randomised controlled trials), with moderate certainty of evidence (figure 6).46 47 Evidence for the effect of other dietary factors was low to very low (online supplemental figures S2–S10).
Discussion
Main findings
In this umbrella review, we included 88 publications, with 310 meta-analyses of randomised controlled trials for different dietary factors and health outcomes, mainly surrogate markers of disease and health, in populations with type 2 diabetes. We found a high certainty of evidence and clinically important changes for liquid meal replacement diets in reducing body weight and body mass index, as well as for a low carbohydrate diet (<26% total energy) in lowering levels of HbA1c and triglycerides. A moderate certainty of evidence with clinically important changes was found for plant based diets and reduced anthropometric measures; liquid meal replacement diets, and supplementation with fibre, ginger, anthocyanins, or probiotics improved glycaemic measures; and a high protein diet, soya, or fibre improved blood lipids. For other results, no clinically relevant differences were found or the certainty of evidence was low or very low.
Comparison with existing dietary guidelines
Dietary interventions based on energy restriction for weight loss are the focus of current guidelines for the management of type 2 diabetes.48–53 Clinically important weight loss of 5-10% can improve insulin sensitivity, glycaemic control, blood pressure, and dyslipidaemia.48 49 53 Also, the guidelines suggest that low calorie meal replacement plans are relatively easy to adhere to and allow for clinically significant weight loss in people with type 2 diabetes.48 50 53 We found a reduction in body weight of 2.4 kg after at least 12 weeks of liquid meal replacement diet.
Current guidelines for type 2 diabetes recommend vegetarian or plant based diets and Mediterranean diets for reducing body weight.48 49 52 Our umbrella review confirmed the beneficial effect of plant based diets on reducing body mass index and waist circumference. Also, some of the current guidelines conclude that short term low carbohydrate interventions have a beneficial effect on anthropometric measures, but no clear recommendations exist for the amount of carbohydrates that should be restricted.48 49 Our study showed that a decrease in body weight was greater in interventions with low (<26% total energy) or very low (<15% total energy) carbohydrate intake than in interventions with moderate (<45% total energy) carbohydrate intake for at least 12 weeks.
We found a low certainty of evidence for the effects of a Mediterranean or plant based diet on glycaemic measures. Our findings of the beneficial effect of low carbohydrate diets on levels of HbA1c, however, are in line with current guidelines.49 50 Increased intake of dietary fibre is emphasised in all guidelines, and our results are consistent with the positive effect of fibre rich foods, especially psyllium supplements, on fasting blood glucose levels.49 53 Adding to the current guidelines, we identified a moderate certainty of evidence for the beneficial effects of ginger supplements, as a plant, capsule, or powder, on fasting blood glucose levels, and the potential importance of polyphenols or polyphenol rich products (eg, cocoa) on HOMA-IR.
Our umbrella review supports evidence of the beneficial effects of a Mediterranean diet on triglyceride levels, but we found only a low certainty of evidence for its effect in reducing levels of high and low density lipoprotein cholesterol. In agreement with current guidelines, we also found beneficial effects of low carbohydrate and low glycaemic index diets on levels of triglycerides and low and high density lipoprotein cholesterol, and of higher fibre intake on levels of total cholesterol.48–50 53 Current Canadian, German, and European guidelines recommend no change in protein intake, which for most people with normal kidney function is 15-20% and 10-25% of total energy.48 50 53 Thus our findings of a beneficial effect of a high protein diet (>25% total energy) on levels of total cholesterol and low density lipoprotein cholesterol provide novel evidence in this context. A previous meta-analysis, however, showed that the health benefits are from an increased intake of plant based rather than animal based protein sources.54 Furthermore, our study provides evidence of the beneficial effects of soy proteins on levels of total cholesterol and low density lipoprotein cholesterol. Pulses are considered a good alternative to meat and a good source of fibre and are therefore recommended for controlling levels of blood lipids.48 50
In common with current guidelines, we found insufficient evidence to recommend reducing total fat intake to improve cardiometabolic measures in people with type 2 diabetes.48 50 American and European guidelines recommend a higher intake of monounsaturated and polyunsaturated fatty acids to improve glucose metabolism, but the evidence supporting this recommendation, according to our results, is weak.49 53 We found that studies of supplementation with monounsaturated or polyunsaturated fatty acids showed improvements in levels of HbA1c and insulin, and in HOMA-IR, but the estimates were imprecisely estimated and the number of participants included was small (≤800). Also, a subgroup analysis comparing different doses of omega 3 fatty acid supplements was performed in one meta-analysis,45 and a stronger risk reduction of major cardiovascular events was seen in the group receiving >3 g/day of omega 3 fatty acids, whereas no precisely estimated effect was found in the group receiving lower doses. This finding might explain the observed effect of omega 3 fatty acids on the incidence of major cardiovascular events in people with type 2 diabetes in our umbrella review. Moreover, a recent systematic review and meta-analysis of randomised controlled trials with an intervention duration of at least 3.9 years showed that supplementation with omega 3 fatty acids reduced cardiovascular events in people with type 1 and type 2 diabetes (risk ratio 0.93, 95% confidence interval 0.90 to 0.97; n=8 randomised controlled trials). The effect was marginal but precisely estimated.55
Strengths and limitations
Our study has several strengths. Our umbrella review included meta-analyses of all possible dietary interventions and their effect on many health outcomes in populations with type 2 diabetes. We summarised the evidence from randomised controlled trials that lasted at least 12 weeks, giving a more reliable assessment of the effectiveness of the interventions. We recalculated the results of meta-analyses that used a fixed effect model, or did not provide I² values or forest plots, to ensure valid meta-estimates with 95% confidence intervals to provide an evaluation of the certainty of evidence based on the GRADEpro approach. Finally, we detected gaps in the evidence that indicate the need for future research.
The main limitation of our umbrella review was that recently published randomised controlled trials not yet included in systematic reviews and meta-analyses were not considered in our report.56–58 Also, we did not explore subgroup analyses (eg, by sex, race, or duration of diabetes) or sensitivity analyses (eg, excluding studies with a high risk of bias). We used the DerSimonian and Laird approach28 for recalculation of extracted estimates with the random effects meta-analyses, but the Hartung and Knapp method might perform better in terms of more adequate error rates, especially when the number of studies was small.59
In terms of the limitations of the publications included in our umbrella review, studies of interventions lasting at least 12 weeks provide more reliable estimates than shorter trials, but these studies do not represent long term interventions. Also, the number of randomised controlled trials in most meta-analyses was small (13 meta-analyses with ≥10 randomised controlled trials). Consequently, the number of participants was also low (n<800 for 83% of meta-analyses of randomised controlled trials). This limitation was one of the main reasons for downgrading the certainty of evidence because of imprecision. Other reasons for downgrading the certainty of evidence were the high risk of bias in the randomised controlled trials and inconsistency of the results. Furthermore, the methodological quality of most included publications was low to very low.
In meta-analyses of randomised controlled trials, a high risk of bias caused by lack of blinding of participants to the dietary intervention is a concern. For dietary interventions based on health promoting changes in diet (eg, advice on a Mediterranean diet, where participants are actively instructed and encouraged to change their diet), however, blinding to the intervention might not be feasible. Furthermore, low compliance with the assigned dietary regimens and high dropout rates were commonly seen in randomised controlled trials, especially for low carbohydrate and ketogenic diets,60 potentially resulting in underestimation of their actual effect. For example, one meta-analysis found greater clinically significant weight loss in those with greater adherence than in those with lower adherence to a very low carbohydrate diet,37 and another meta-analysis reported inadequate compliance with the ketogenic diet, as assessed by urinary measurements of ketones.13 Systematic reviews of other dietary interventions lacked a description of compliance with the intervention. Also, study arms did not always receive isocaloric diets, making it difficult to differentiate between the effects of change in dietary patterns, foods, or nutrients on the reduction in energy intake. Some of the meta-analyses conducted subgroup analyses, however, and did not find differences when calories were restricted or matched with controls in the trials.33 34 37
Study implications
Robust evidence exists that in people with type 2 diabetes, liquid meal replacements decrease energy intake and thus body weight. For people who prefer a dietary approach, plant based or carbohydrate restricted diets are also effective in reducing anthropometric measure. Other dietary regimens, such as a ketogenic diet or intermittent fasting, reduced body weight, but the certainty of evidence was low or very low.
Many dietary interventions effectively improved glycaemic measures, but the certainty of the evidence was robust only for liquid meal replacement diets and restricting carbohydrates. Intake of fibre rich foods, polyphenols, probiotics, and ginger was also beneficial for glycaemic control. For control of blood lipids, a low carbohydrate (<26% total energy), Mediterranean, or high protein diet is recommended for people with type 2 diabetes, with the advice to change the protein source to plant based alternatives. Energy restriction was the only effective approach in reducing blood pressure with robust evidence. Restriction of salt intake also showed beneficial effects, but the certainty of evidence was very low and thus recommendations cannot be made from the current body of evidence.
For clinical outcomes, robust evidence exists only for a low carbohydrate diet and reduction in the use of drug treatments. For other outcomes, including kidney function parameters, inflammatory markers, liver enzymes, and patient relevant outcomes (eg, remission of diabetes, health related quality of life, and incidence of cardiovascular disease), meta-analyses are already available, but the certainty of evidence was rated as low or very low, and definitive conclusions on these findings cannot be drawn. In summary, people with type 2 diabetes can be advised to reduce their energy intake if they have obesity or overweight, decrease their carbohydrate intake, or increase their consumption of foods from plant sources (especially plant based proteins) or from Mediterranean-style diets (foods high in polyunsaturated fatty acids, such as fish and nuts, or foods high in polyphenols, such as fruit, vegetables, and legumes).
To strengthen the certainty of evidence for many of the findings on diet and the management of type 2 diabetes and its complications, future studies with a low risk of bias are needed. More randomised controlled trials should investigate the effects of long term (at least six months) dietary interventions, as well as considering isocaloric comparisons between intervention and control arms. Also, more research on dietary patterns (eg, DASH (dietary approaches to stop hypertension), Nordic, and portfolio diets), fasting approaches, single foods (eg, dairy products, fish, or meat), and single nutrients (eg, specific fatty acids) is needed. Finally, future systematic reviews and meta-analyses should follow current guidelines for conducting and reporting on included studies (eg, reports should be transparent about the methods used and critically examine the effect of risk of bias on meta-findings).
Conclusions
In this umbrella review, we identified the dietary factors with robust evidence of health benefits, expressed as surrogate disease and health markers in people with type 2 diabetes. An energy restricted diet can reduce body weight and improve cardiometabolic health. In addition to energy restriction, dietary approaches, such as a plant based, Mediterranean, low carbohydrate (<26% total energy), or high protein diet, are beneficial for cardiometabolic health in individuals with type 2 diabetes. For evidence based recommendations of the effectiveness of other dietary factors in the management of type 2 diabetes, more randomised controlled trials and systematic reviews with meta-analyses focusing on long term interventions are needed.
Acknowledgments
We thank Andrew Reynolds, Department of Medicine, University of Otago, Dunedin, New Zealand; Grace Austin, Hunter Medical Research Institute, University of Newcastle, New Lambton, Australia; Likun Ma, Department of Cardiology, the First Affiliated Hospital of USTC, Division of Life Sciences and Medicine, University of Science and Technology of China, Hefei, China; Eunyoung Kim, Evidence‑Based and Clinical Research Laboratory, Department of Health, Social, and Clinical Pharmacy, College of Pharmacy, Chung-Ang University, Seoul, Republic of Korea; Kaijian Hou, Endocrine Department, Longhu Hospital, First Affiliated Hospital of Shantou University Shantou City, China; Shaun Mason, School of Exercise and Nutrition Sciences, Deakin University, Melbourne, Victoria, Australia; Jean-Philippe Drouin-Chartier, Department of Nutrition, Harvard T H Chan School of Public Health, Boston, MA, USA; and Sakineh Shab-Bidar, Department of Community Nutrition, School of Nutritional Sciences and Dietetics, Tehran University of Medical Sciences, Tehran, Iran, for sending additional data needed for the meta-analysis.
Footnotes
Twitter: @Edyta_Szcz
Contributors: SS designed the research. ES and TS conducted the literature search and literature screening. ES and TS extracted the data. ES, AS-P, and TS assessed the methodological quality of the included publications. ES, SS, and LS evaluated the certainty of evidence. ES, JB, and SS analysed the data and wrote the first draft of the paper. All authors interpreted the data, read the manuscript, and approved the final version. ES and SS are the guarantors. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria were omitted. Transparency: The lead authors (the manuscript’s guarantors) affirm that the manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned have been explained.
Funding: The German Diabetes Centre is funded by the German Federal Ministry of Health and the Ministry of Innovation, Science, Research, and Technology of the State North Rhine Westphalia. This study was also supported in part by a grant from the German Federal Ministry of Education and Research to the German Centre for Diabetes Research. The funders had no role in considering the study design or in the collection, analysis, interpretation of data, writing of the report, or decision to submit the article for publication.
Competing interests: All authors have completed the ICMJE uniform disclosure form at www.icmje.org/disclosure-of-interest/ and declare: support from the German Federal Ministry of Health and the Ministry of Innovation, Science, Research, and Technology of the State North Rhine Westphalia, and the German Federal Ministry of Education and Research for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. All data are available upon reasonable request. All data for this study were extracted from published meta-analyses, all of which are available and accessible.
Ethics approval
Not required because only (secondary) data from published studies were used.
References
- 1.International Diabetes Federation . IDF diabetes atlas, 10th edn. Brussels, Belgium, 2021. [Google Scholar]
- 2.American Diabetes A. Economic costs of diabetes in the U.S. in 2017. Diabetes Care 2018;41:917–28. 10.2337/dci18-0007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Peters ML, Huisman EL, Schoonen M, et al. The current total economic burden of diabetes mellitus in the Netherlands. Neth J Med 2017;75:281–97. [PubMed] [Google Scholar]
- 4.Organization WH . Global reports on diabetes; 2016. Available: https://www.who.int/publications/i/item/9789241565257
- 5.Jaiswal M, Schinske A, Pop-Busui R. Lipids and lipid management in diabetes. Best Pract Res Clin Endocrinol Metab 2014;28:325–38. 10.1016/j.beem.2013.12.001 [DOI] [PubMed] [Google Scholar]
- 6.Roden M, Shulman GI. The integrative biology of type 2 diabetes. Nature 2019;576:51–60. 10.1038/s41586-019-1797-8 [DOI] [PubMed] [Google Scholar]
- 7.Harding JL, Pavkov ME, Magliano DJ, et al. Global trends in diabetes complications: a review of current evidence. Diabetologia 2019;62:3–16. 10.1007/s00125-018-4711-2 [DOI] [PubMed] [Google Scholar]
- 8.An Y, Zhang P, Wang J, et al. Cardiovascular and all-cause mortality over a 23-year period among Chinese with newly diagnosed diabetes in the DA Qing IGT and diabetes study. Diabetes Care 2015;38:1365–71. 10.2337/dc14-2498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barr ELM, Zimmet PZ, Welborn TA, et al. Risk of cardiovascular and all-cause mortality in individuals with diabetes mellitus, impaired fasting glucose, and impaired glucose tolerance: the Australian diabetes, obesity, and lifestyle study (Ausdiab). Circulation 2007;116:151–7. 10.1161/CIRCULATIONAHA.106.685628 [DOI] [PubMed] [Google Scholar]
- 10.Neuenschwander M, Ballon A, Weber KS, et al. Role of diet in type 2 diabetes incidence: umbrella review of meta-analyses of prospective observational studies. BMJ 2019;366:l2368. 10.1136/bmj.l2368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huo R, Du T, Xu Y, et al. Effects of mediterranean-style diet on glycemic control, weight loss and cardiovascular risk factors among type 2 diabetes individuals: a meta-analysis. Eur J Clin Nutr 2015;69:1200–8. 10.1038/ejcn.2014.243 [DOI] [PubMed] [Google Scholar]
- 12.Schwingshackl L, Chaimani A, Hoffmann G, et al. A network meta-analysis on the comparative efficacy of different dietary approaches on glycaemic control in patients with type 2 diabetes mellitus. Eur J Epidemiol 2018;33:157–70. 10.1007/s10654-017-0352-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rafiullah M, Musambil M, David SK. Effect of a very low-carbohydrate ketogenic diet vs recommended diets in patients with type 2 diabetes: a meta-analysis. Nutr Rev 2022;80:488–502. 10.1093/nutrit/nuab040 [DOI] [PubMed] [Google Scholar]
- 14.Xiao Z, Chen H, Zhang Y, et al. The effect of psyllium consumption on weight, body mass index, lipid profile, and glucose metabolism in diabetic patients: a systematic review and dose-response meta-analysis of randomized controlled trials. Phytother Res 2020;34:1237–47. 10.1002/ptr.6609 [DOI] [PubMed] [Google Scholar]
- 15.Barańska A, Błaszczuk A, Polz-Dacewicz M, et al. Effects of soy isoflavones on glycemic control and lipid profile in patients with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Nutrients 2021;13:1886. 10.3390/nu13061886 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Asbaghi O, Moradi S, Nezamoleslami S, et al. The effects of magnesium supplementation on lipid profile among type 2 diabetes patients: a systematic review and meta-analysis of randomized controlled trials. Biol Trace Elem Res 2021;199:861–73. 10.1007/s12011-020-02209-5 [DOI] [PubMed] [Google Scholar]
- 17.Churuangsuk C, Hall J, Reynolds A, et al. Diets for weight management in adults with type 2 diabetes: an umbrella review of published meta-analyses and systematic review of trials of diets for diabetes remission. Diabetologia 2022;65:14–36. 10.1007/s00125-021-05577-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kahleova H, Salas-Salvadó J, Rahelić D, et al. Dietary patterns and cardiometabolic outcomes in diabetes: a summary of systematic reviews and meta-analyses. Nutrients 2019;11:2209. 10.3390/nu11092209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xu B, Fu J, Qiao Y, et al. Higher intake of microbiota-accessible carbohydrates and improved cardiometabolic risk factors: a meta-analysis and umbrella review of dietary management in patients with type 2 diabetes. Am J Clin Nutr 2021;113:1515–30. 10.1093/ajcn/nqaa435 [DOI] [PubMed] [Google Scholar]
- 20.Zeraattalab-Motlagh S, Jayedi A, Shab-Bidar S. The effects of resveratrol supplementation in patients with type 2 diabetes, metabolic syndrome, and nonalcoholic fatty liver disease: an umbrella review of meta-analyses of randomized controlled trials. Am J Clin Nutr 2021;114:1675–85. 10.1093/ajcn/nqab250 [DOI] [PubMed] [Google Scholar]
- 21.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. 10.1136/bmj.n71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gates M, Gates A, Pieper D, et al. Reporting guideline for overviews of reviews of healthcare interventions: development of the PRIOR statement. BMJ 2022;378:e070849. 10.1136/bmj-2022-070849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017;358:j4008. 10.1136/bmj.j4008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Y, Akl EA, Schünemann HJ. Using systematic reviews in guideline development: the GRADE approach. Res Synth Methods 2018;10:312–29. 10.1002/jrsm.1313 [DOI] [PubMed] [Google Scholar]
- 25.Guyatt GH, Oxman AD, Vist GE, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336:924–6. 10.1136/bmj.39489.470347.AD [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schwingshackl L, Schünemann HJ, Meerpohl JJ. Improving the trustworthiness of findings from nutrition evidence syntheses: assessing risk of bias and rating the certainty of evidence. Eur J Nutr 2021;60:2893–903. 10.1007/s00394-020-02464-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hultcrantz M, Rind D, Akl EA, et al. The GRADE working group clarifies the construct of certainty of evidence. J Clin Epidemiol 2017;87:4–13. 10.1016/j.jclinepi.2017.05.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials 1986;7:177–88. 10.1016/0197-2456(86)90046-2 [DOI] [PubMed] [Google Scholar]
- 29.Egger M, Davey Smith G, Schneider M, et al. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315:629–34. 10.1136/bmj.315.7109.629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sterne JA, Egger M, Smith GD. Systematic reviews in health care: investigating and dealing with publication and other biases in meta-analysis. BMJ 2001;323:101–5. 10.1136/bmj.323.7304.101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Neuenschwander M, Hoffmann G, Schwingshackl L, et al. Impact of different dietary approaches on blood lipid control in patients with type 2 diabetes mellitus: a systematic review and network meta-analysis. Eur J Epidemiol 2019;34:837–52. 10.1007/s10654-019-00534-1 [DOI] [PubMed] [Google Scholar]
- 32.Noronha JC, Nishi SK, Braunstein CR, et al. The effect of liquid meal replacements on cardiometabolic risk factors in overweight/obese individuals with type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Diabetes Care 2019;42:767–76. 10.2337/dc18-2270 [DOI] [PubMed] [Google Scholar]
- 33.Austin G, Ferguson JJA, Garg ML. Effects of plant-based diets on weight status in type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials. Nutrients 2021;13:4099. 10.3390/nu13114099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jayedi A, Zeraattalab-Motlagh S, Jabbarzadeh B, et al. Dose-dependent effect of carbohydrate restriction for type 2 diabetes management: a systematic review and dose-response meta-analysis of randomized controlled trials. Am J Clin Nutr 2022;116:40–56. 10.1093/ajcn/nqac066 [DOI] [PubMed] [Google Scholar]
- 35.Ajala O, English P, Pinkney J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am J Clin Nutr 2013;97:505–16. 10.3945/ajcn.112.042457 [DOI] [PubMed] [Google Scholar]
- 36.Ojo O, Wang XH, Ojo OO, et al. The effects of almonds on gut microbiota, glycometabolism, and inflammatory markers in patients with type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials. Nutrients 2021;13:3377. 10.3390/nu13103377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Goldenberg JZ, Day A, Brinkworth GD, et al. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ 2021;372:m4743. 10.1136/bmj.m4743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ebrahimzadeh A, Ebrahimzadeh A, Mirghazanfari SM, et al. The effect of ginger supplementation on metabolic profiles in patients with type 2 diabetes mellitus: a systematic review and meta-analysis of randomized controlled trials. Complement Ther Med 2022;65:102802. 10.1016/j.ctim.2022.102802 [DOI] [PubMed] [Google Scholar]
- 39.Chen X, Guan X, Tang Y, et al. Effects of cocoa products intake on cardiometabolic biomarkers of type 2 diabetes patients: a systematic review and meta-analysis based on both long-term and short-term randomised controlled trials. Int J Food Sci Nutr 2022;73:571–87. 10.1080/09637486.2022.2046711 [DOI] [PubMed] [Google Scholar]
- 40.Fallah AA, Sarmast E, Jafari T. Effect of dietary anthocyanins on biomarkers of glycemic control and glucose metabolism: a systematic review and meta-analysis of randomized clinical trials. Food Res Int 2020;137:109379. 10.1016/j.foodres.2020.109379 [DOI] [PubMed] [Google Scholar]
- 41.Zhang C, Jiang J, Wang C, et al. Meta-analysis of randomized controlled trials of the effects of probiotics on type 2 diabetes in adults. Clin Nutr 2022;41:365–73. 10.1016/j.clnu.2021.11.037 [DOI] [PubMed] [Google Scholar]
- 42.Korsmo-Haugen HK, Brurberg KG, Mann J, et al. Carbohydrate quantity in the dietary management of type 2 diabetes: a systematic review and meta-analysis. Diabetes Obes Metab 2019;21:15–27. 10.1111/dom.13499 [DOI] [PubMed] [Google Scholar]
- 43.Yu Z, Nan F, Wang LY, et al. Effects of high-protein diet on glycemic control, insulin resistance and blood pressure in type 2 diabetes: a systematic review and meta-analysis of randomized controlled trials. Clin Nutr 2020;39:1724–34. 10.1016/j.clnu.2019.08.008 [DOI] [PubMed] [Google Scholar]
- 44.Ojo O, Ojo OO, Wang XH, et al. The effects of a low GI diet on cardiometabolic and inflammatory parameters in patients with type 2 and gestational diabetes: a systematic review and meta-analysis of randomised controlled trials. Nutrients 2019;11:1584. 10.3390/nu11071584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shen S, Gong C, Jin K, et al. Omega-3 fatty acid supplementation and coronary heart disease risks: a meta-analysis of randomized controlled clinical trials. Front Nutr 2022;9:809311. 10.3389/fnut.2022.809311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Abdollahi S, Meshkini F, Clark CCT, et al. The effect of probiotics/synbiotics supplementation on renal and liver biomarkers in patients with type 2 diabetes: a systematic review and meta-analysis of randomised controlled trials. Br J Nutr 2021;1-31:625–35. 10.1017/S0007114521003780 [DOI] [PubMed] [Google Scholar]
- 47.Hosseini H, Koushki M, Khodabandehloo H, et al. The effect of resveratrol supplementation on C-reactive protein (CRP) in type 2 diabetic patients: results from a systematic review and meta-analysis of randomized controlled trials. Complement Ther Med 2020;49:102251. 10.1016/j.ctim.2019.102251 [DOI] [PubMed] [Google Scholar]
- 48.Sievenpiper JL, Chan CB, Dworatzek PD, et al. Nutrition therapy. Can J Diabetes 2018;42 Suppl 1:S64–79. 10.1016/j.jcjd.2017.10.009 [DOI] [PubMed] [Google Scholar]
- 49.American Diabetes Association Professional Practice Committee . Facilitating behavior change and well-being to improve health outcomes: standards of medical care in diabetes—2022. Diabetes Care 2022;45:S60–82. 10.2337/dc22-S005 [DOI] [PubMed] [Google Scholar]
- 50.Skurk T, Bosy-Westphal A, Grünerbel A, et al. Dietary recommendations for persons with type 2 diabetes mellitus. Exp Clin Endocrinol Diabetes 2022;130(S 01):S151–84. 10.1055/a-1624-5095 [DOI] [PubMed] [Google Scholar]
- 51.Davies MJ, Aroda VR, Collins BS, et al. Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2022;65:1925–66. 10.1007/s00125-022-05787-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dyson PA, Twenefour D, Breen C, et al. Diabetes UK evidence-based nutrition guidelines for the prevention and management of diabetes. Diabet Med 2018;35:541–7. 10.1111/dme.13603 [DOI] [PubMed] [Google Scholar]
- 53.Diabetes and Nutrition Study Group (DNSG) of the European Association for the Study of Diabetes (EASD) . Evidence-based European recommendations for the dietary management of diabetes. Diabetologia 2023;66:965–85. 10.1007/s00125-023-05894-8 [DOI] [PubMed] [Google Scholar]
- 54.Naghshi S, Sadeghi O, Willett WC, et al. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020;370:m2412. 10.1136/bmj.m2412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Huang L, Zhang F, Xu P, et al. Corrigendum to 'Effect of omega-3 polyunsaturated fatty acids on cardiovascular outcomes in patients with diabetes: a meta-analysis of randomized controlled trials'. Adv Nutr 2023;14:1250–1. 10.1016/j.advnut.2023.08.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xiao Y, Liu Y, Zhao L, et al. Effect of 5:2 fasting diet on liver fat content in patients with type 2 diabetic with nonalcoholic fatty liver disease. Metab Syndr Relat Disord 2022;20:459–65. 10.1089/met.2022.0014 [DOI] [PubMed] [Google Scholar]
- 57.Wang W, Wang X, Cao S, et al. Dietary antioxidant indices in relation to all-cause and cause-specific mortality among adults with diabetes: a prospective cohort study. Front Nutr 2022;9:849727. 10.3389/fnut.2022.849727 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wang H-W, Huang Y-T, Jiang M-Y. Association of dietary magnesium intake and glycohemoglobin with mortality risk in diabetic patients. PLoS One 2022;17:e0277180. 10.1371/journal.pone.0277180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.IntHout J, Ioannidis JPA, Borm GF. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard Dersimonian-Laird method. BMC Med Res Methodol 2014;14:25. 10.1186/1471-2288-14-25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mirmiran P, Bahadoran Z, Gaeini Z. Common limitations and challenges of dietary clinical trials for translation into clinical practices. Int J Endocrinol Metab 2021;19:e108170. 10.5812/ijem.108170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lohner S, Kuellenberg de Gaudry D, Toews I, et al. Non-nutritive sweeteners for diabetes mellitus. Cochrane Database Syst Rev 2020;5:CD012885. 10.1002/14651858.CD012885.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zou Y, Guo B, Yu S, et al. Effect of vitamin D supplementation on Glycose homeostasis and islet function in vitamin D deficient or insufficient diabetes and Prediabetes: a systematic review and meta-analysis. J Clin Biochem Nutr 2021;69:229–37. 10.3164/jcbn.20-165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Silverii GA, Botarelli L, Dicembrini I, et al. Low-carbohydrate diets and type 2 diabetes treatment: a meta-analysis of randomized controlled trials. Acta Diabetol 2020;57:1375–82. 10.1007/s00592-020-01568-8 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjmed-2023-000664supp001.pdf (6.3MB, pdf)
Data Availability Statement
Data are available upon reasonable request. All data relevant to the study are included in the article or uploaded as supplementary information. All data are available upon reasonable request. All data for this study were extracted from published meta-analyses, all of which are available and accessible.