Abstract
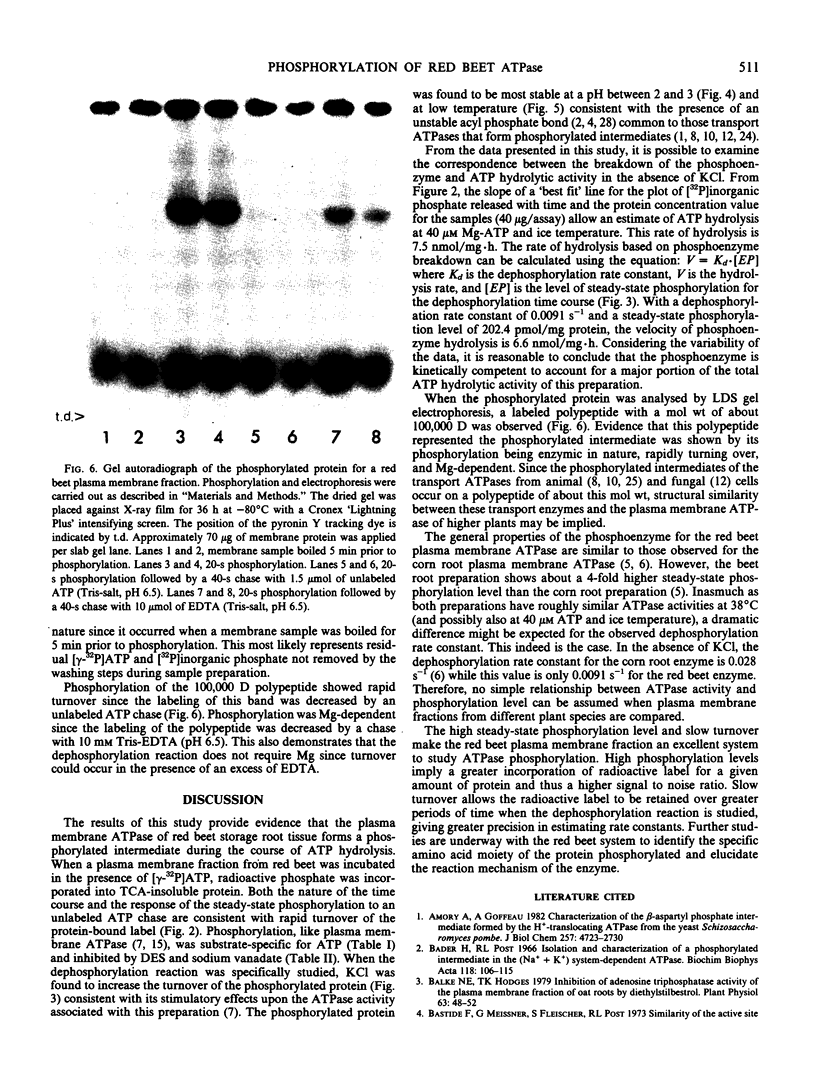
When a plasma membrane-enriched fraction isolated from red beet (Beta vulgaris L.) was incubated in the presence of 40 micromolar [γ-32P] ATP, 40 micromolar MgSO4 at pH 6.5, a rapidly turning over phosphorylated protein was formed. Phosphorylation of the protein was substrate-specific for ATP, sensitive to diethylstilbestrol and vanadate, but insensitive to azide. When the dephosphorylation reaction was specifically studied, KCl was found to increase the turnover of the phosphorylated protein consistent with its stimulatory effect upon plasma membrane ATPase. The protein-bound phosphate was found to be most stable at a pH between 2 and 3 and under cold temperature, suggesting that the protein phosphate bond was an acyl-phosphate. When the phosphorylated protein was analyzed with lithium dodecyl sulfate gel electrophoresis, a labeled polypeptide with a molecular weight of about 100,000 daltons was observed. Phosphorylation of this polypeptide was rapidly turning over and Mg-dependent. It is concluded that the phosphorylation observed represents a reaction intermediate of the red beet plasma membrane ATPase.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amory A., Gofffeau A. Characterization of the beta-aspartyl phosphate intermediate formed by the H+-translocating ATPase from the yeast Schizosaccharomyces pombe. J Biol Chem. 1982 May 10;257(9):4723–4730. [PubMed] [Google Scholar]
- Bader H., Sen A. K., Post R. L. Isolation and characterization of a phosphorylated intermediate in the (Na+ + K+) system-dependent ATPase. Biochim Biophys Acta. 1966 Apr 12;118(1):106–115. doi: 10.1016/s0926-6593(66)80149-2. [DOI] [PubMed] [Google Scholar]
- Balke N. E., Hodges T. K. Inhibition of adenosine triphosphatase activity of the plasma membrane fraction of oat roots by diethylstilbestrol. Plant Physiol. 1979 Jan;63(1):48–52. doi: 10.1104/pp.63.1.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briskin D. P., Leonard R. T. Partial characterization of a phosphorylated intermediate associated with the plasma membrane ATPase of corn roots. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6922–6926. doi: 10.1073/pnas.79.22.6922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briskin D. P., Leonard R. T. Phosphorylation of the adenosine triphosphatase in a deoxycholate-treated plasma membrane fraction from corn roots. Plant Physiol. 1982 Nov;70(5):1459–1464. doi: 10.1104/pp.70.5.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briskin D. P., Poole R. J. Characterization of a k-stimulated adenosine triphosphatase associated with the plasma membrane of red beet. Plant Physiol. 1983 Feb;71(2):350–355. doi: 10.1104/pp.71.2.350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dupont F. M., Burke L. L., Spanswick R. M. Characterization of a partially purified adenosine triphosphatase from a corn root plasma membrane fraction. Plant Physiol. 1981 Jan;67(1):59–63. doi: 10.1104/pp.67.1.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goffeau A., Slayman C. W. The proton-translocating ATPase of the fungal plasma membrane. Biochim Biophys Acta. 1981 Dec 30;639(3-4):197–223. doi: 10.1016/0304-4173(81)90010-0. [DOI] [PubMed] [Google Scholar]
- Gunn R. B. Co- and counter-transport mechanisms in cell membranes. Annu Rev Physiol. 1980;42:249–259. doi: 10.1146/annurev.ph.42.030180.001341. [DOI] [PubMed] [Google Scholar]
- Knowles J. R. Enzyme-catalyzed phosphoryl transfer reactions. Annu Rev Biochem. 1980;49:877–919. doi: 10.1146/annurev.bi.49.070180.004305. [DOI] [PubMed] [Google Scholar]
- Lichtner R., Wolf H. U. Dodecyl sulphate/polyacrylamide-gel electrophoresis at low pH values and low temperatures. Biochem J. 1979 Sep 1;181(3):759–761. doi: 10.1042/bj1810759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maloney P. C. Energy coupling to ATP synthesis by the proton-translocating ATPase. J Membr Biol. 1982;67(1):1–12. doi: 10.1007/BF01868643. [DOI] [PubMed] [Google Scholar]
- Mårdh S., Zetterqvist O. Phosphorylation and dephosphorylation reactions of bovine brain (Na+-K+)-stimulated ATP phosphohydrolase studied by a rapid mixing technique. Biochim Biophys Acta. 1974 Jun 18;350(2):473–483. doi: 10.1016/0005-2744(74)90523-3. [DOI] [PubMed] [Google Scholar]
- Peterson G. L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem. 1977 Dec;83(2):346–356. doi: 10.1016/0003-2697(77)90043-4. [DOI] [PubMed] [Google Scholar]
- Petraglia T., Poole R. J. ATP Levels and their Effects on Plasmalemma Influxes of Potassium Chloride in Red Beet. Plant Physiol. 1980 May;65(5):969–972. doi: 10.1104/pp.65.5.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs G., Berglindh T., Rabon E., Wallmark B., Barcellona M. L., Stewart H. B., Saccomani G. The interaction of K+ with gastric parietal cells and gastric ATPase. Ann N Y Acad Sci. 1980;358:118–137. doi: 10.1111/j.1749-6632.1980.tb15391.x. [DOI] [PubMed] [Google Scholar]
- Sze H., Churchill K. A. Mg/KCl-ATPase of plant plasma membranes is an electrogenic pump. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5578–5582. doi: 10.1073/pnas.78.9.5578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Meis L., Vianna A. L. Energy interconversion by the Ca2+-dependent ATPase of the sarcoplasmic reticulum. Annu Rev Biochem. 1979;48:275–292. doi: 10.1146/annurev.bi.48.070179.001423. [DOI] [PubMed] [Google Scholar]