Abstract
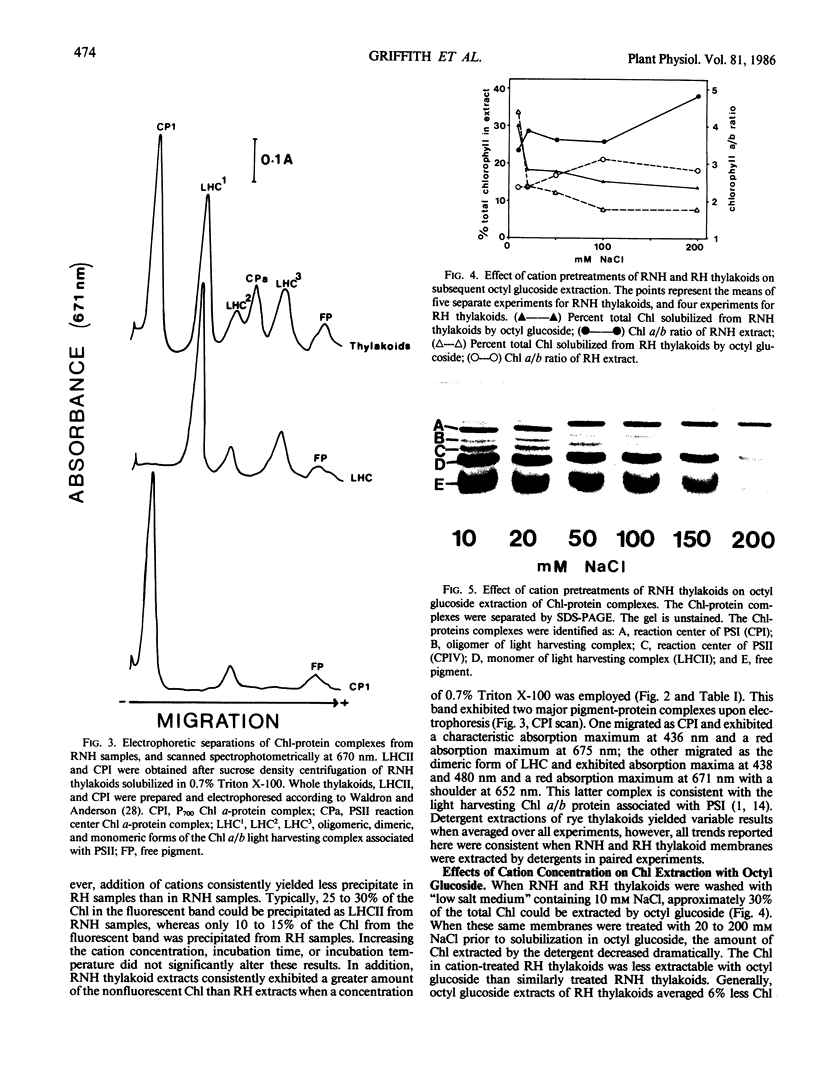
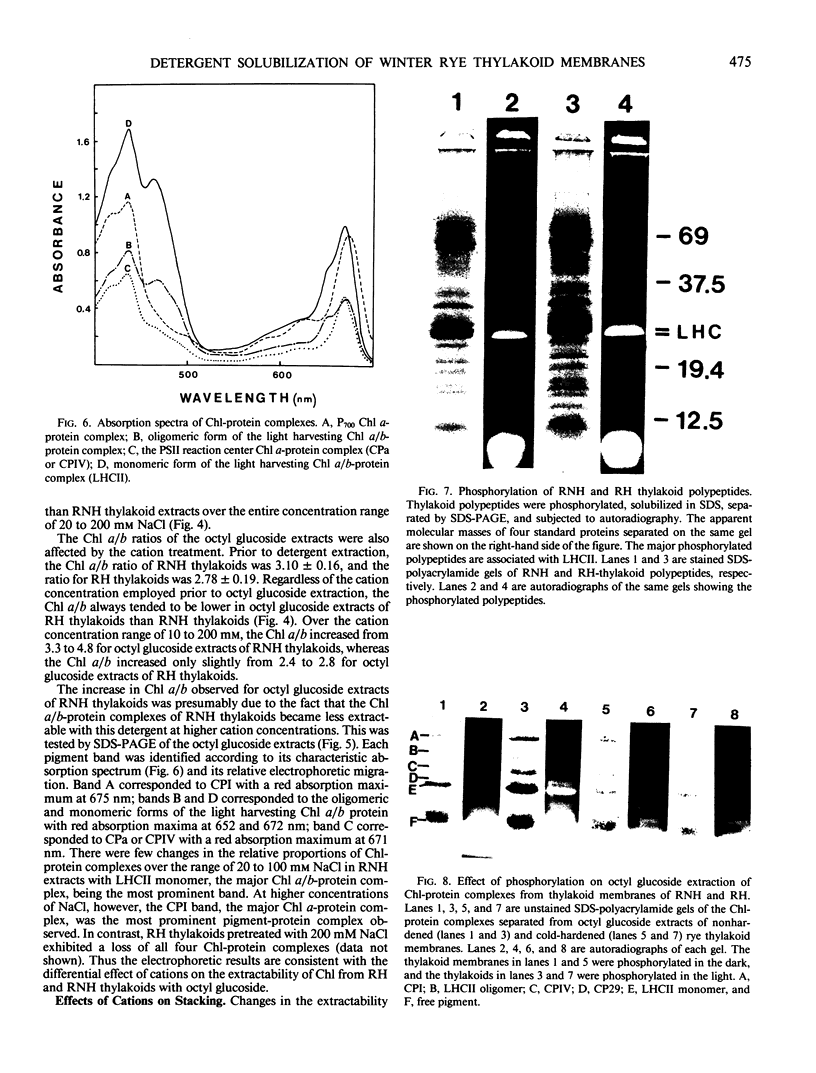
Thylakoids isolated from leaves of winter rye (Secale cereale L. cv Puma) grown at either 20 or 5°C were extracted with the nonionic detergents Triton X-100 and octyl glucoside. Less total chlorophyll was extracted from 5°C thylakoids by these detergents under all conditions, including pretreatment with cations. Thylakoids from either 20 or 5°C leaves were solubilized in 0.7% Triton X-100 and centrifuged on sucrose gradients to purify the light harvesting complex (LHCII). Greater yields of LHCII were obtained by cation precipitation of particles derived from 20°C thylakoids than from 5°C thylakoids. When 20 and 5°C thylakoids were phosphorylated and completely solubilized in sodium dodecyl sulfate, no differences were observed in the 32Pi-labeling characteristics of the membrane polypeptides. However, when phosphorylated thylakoids were extracted with octyl glucoside, extraction of LHCII associated with the 5°C thylakoids was markedly reduced in comparison with the extraction of LHCII from 20°C membranes. Since 20 and 5°C thylakoids exhibited significant differences in the Chl content and Chl a/b ratios of membrane fractions produced after solubilization with either Triton X-100 or octyl glucoside, and since few differences between the proteins of the two membranes could be observed following complete denaturation in sodium dodecyl sulfate, we conclude that the integral structure of the thylakoid membrane is affected during rye leaf development at low temperature.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burke J. J., Ditto C. L., Arntzen C. J. Involvement of the light-harvesting complex in cation regulation of excitation energy distribution in chloroplasts. Arch Biochem Biophys. 1978 Apr 15;187(1):252–263. doi: 10.1016/0003-9861(78)90031-0. [DOI] [PubMed] [Google Scholar]
- Camm E. L., Green B. R. Fractionation of Thylakoid Membranes with the Nonionic Detergent Octyl-beta-d-glucopyranoside: RESOLUTION OF CHLOROPHYLL-PROTEIN COMPLEX II INTO TWO CHLOROPHYLL-PROTEIN COMPLEXES. Plant Physiol. 1980 Sep;66(3):428–432. doi: 10.1104/pp.66.3.428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camm E. L., Green B. R. The effects of cations and trypsin on extraction of chlorophyll-protein complexes by octyl glucoside. Arch Biochem Biophys. 1982 Apr 1;214(2):563–572. doi: 10.1016/0003-9861(82)90061-3. [DOI] [PubMed] [Google Scholar]
- Chow W. S., Thorne S. W., Duniec J. T., Sculley M. J., Boardman N. K. The stacking of chloroplast thylakoids. Effects of cation screening and binding, studied by the digitonin method. Arch Biochem Biophys. 1980 Apr 15;201(1):347–355. doi: 10.1016/0003-9861(80)90520-2. [DOI] [PubMed] [Google Scholar]
- Griffith M., Brown G. N., Huner N. P. Structural Changes in Thylakoid Proteins during Cold Acclimation and Freezing of Winter Rye (Secale cereale L. cv. Puma). Plant Physiol. 1982 Aug;70(2):418–423. doi: 10.1104/pp.70.2.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffith M., Elfman B., Camm E. L. Accumulation of Plastoquinone A during Low Temperature Growth of Winter Rye. Plant Physiol. 1984 Mar;74(3):727–729. doi: 10.1104/pp.74.3.727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffith M., Huner N. P., Kyle D. J. Fluorescence Properties Indicate that Photosystem II Reaction Centers and Light-Harvesting Complex Are Modified by Low Temperature Growth in Winter Rye. Plant Physiol. 1984 Oct;76(2):381–385. doi: 10.1104/pp.76.2.381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helenius A., Simons K. Solubilization of membranes by detergents. Biochim Biophys Acta. 1975 Mar 25;415(1):29–79. doi: 10.1016/0304-4157(75)90016-7. [DOI] [PubMed] [Google Scholar]
- Huner N. P., Macdowall D. H. Chloroplastic proteins of wheat and rye grown at warm and cold-hardening temperatures. Can J Biochem. 1976 Oct;54(10):848–853. doi: 10.1139/o76-122. [DOI] [PubMed] [Google Scholar]
- Mullet J. E., Arntzen C. J. Simulation of grana stacking in a model membrane system. Mediation by a purified light-harvesting pigment-protein complex from chloroplasts. Biochim Biophys Acta. 1980 Jan 4;589(1):100–117. doi: 10.1016/0005-2728(80)90135-8. [DOI] [PubMed] [Google Scholar]
- Mullet J. E., Burke J. J., Arntzen C. J. Chlorophyll proteins of photosystem I. Plant Physiol. 1980 May;65(5):814–822. doi: 10.1104/pp.65.5.814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinback K. E., Bose S., Kyle D. J. Phosphorylation of the light-harvesting chlorophyll-protein regulates excitation energy distribution between photosystem II and photosystem I. Arch Biochem Biophys. 1982 Jun;216(1):356–361. doi: 10.1016/0003-9861(82)90221-1. [DOI] [PubMed] [Google Scholar]
- Steinback K. E., Burke J. J., Arntzen C. J. Evidence for the role of surface-exposed segments of the light-harvesting complex in cation-mediated control of chloroplast structure and function. Arch Biochem Biophys. 1979 Jul;195(2):546–557. doi: 10.1016/0003-9861(79)90381-3. [DOI] [PubMed] [Google Scholar]
- Waldron J. C., Anderson J. M. Chlorophyll-protein complexes from thylakoids of a mutant barley lacking chlorophyll b. Eur J Biochem. 1979 Dec 17;102(2):357–362. doi: 10.1111/j.1432-1033.1979.tb04250.x. [DOI] [PubMed] [Google Scholar]