Abstract
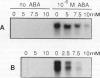
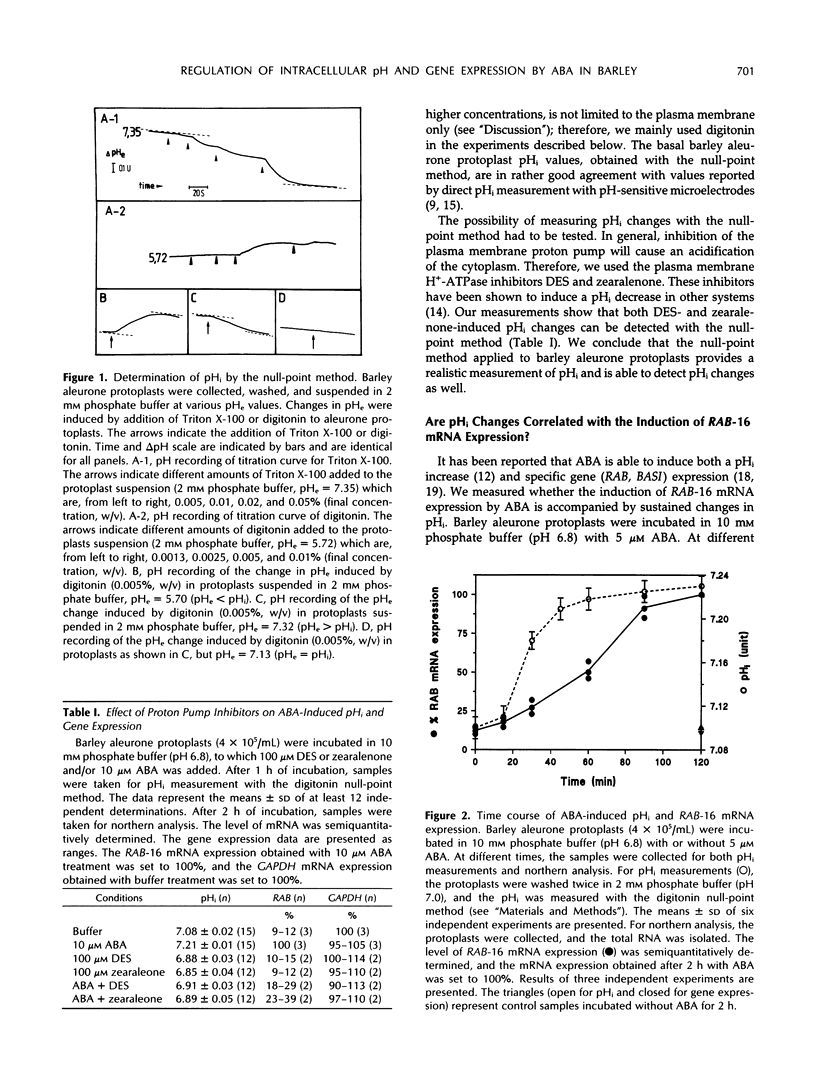
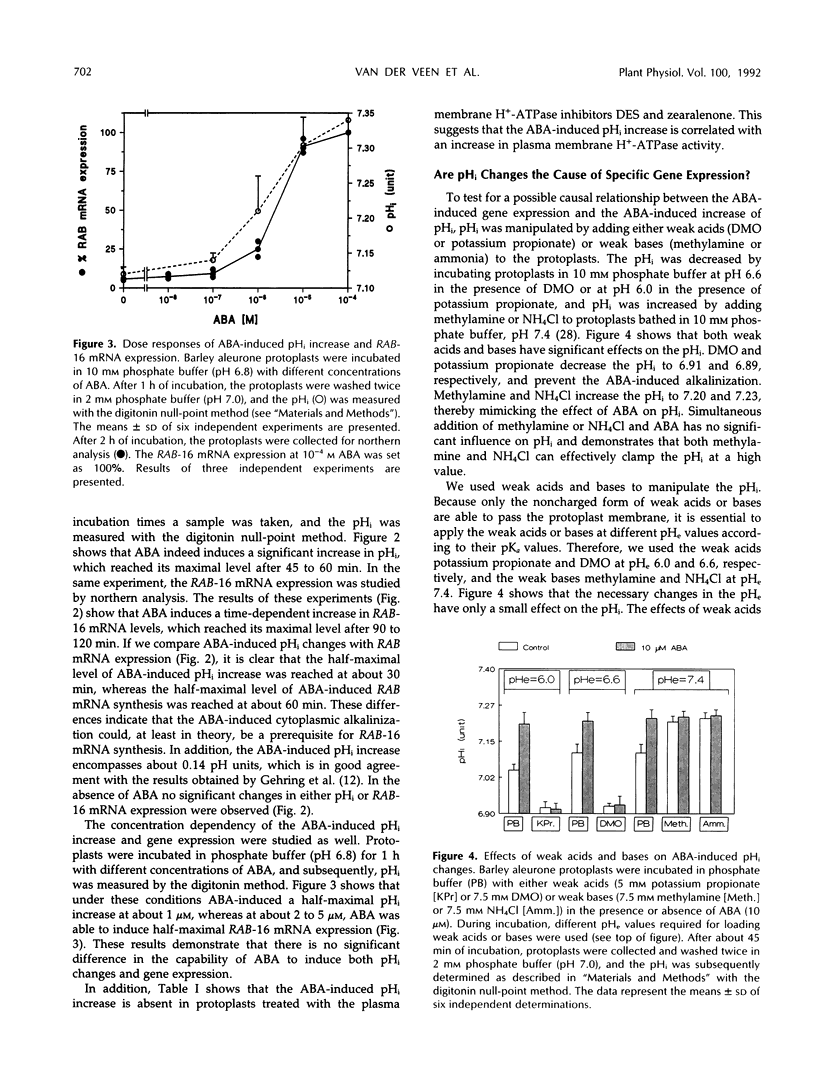
We investigated whether intracellular pH (pHi) is a causal mediator in abscisic acid (ABA)-induced gene expression. We measured the change in pHi by a “null-point” method during stimulation of barley (Hordeum vulgare cv Himalaya) aleurone protoplasts with ABA and found that ABA induces an increase in pHi from 7.11 to 7.30 within 45 min after stimulation. This increase is inhibited by plasma membrane H+-ATPase inhibitors, which induce a decrease in pHi, both in the presence and absence of ABA. This ABA-induced pHi increase precedes the expression of RAB-16 mRNA, as measured by northern analysis. ABA-induced pHi changes can be bypassed or clamped by addition of either the weak acids 5,5-dimethyl-2,4-oxazolidinedione and propionic acid, which decrease the pHi, or the weak bases methylamine and ammonia, which increase the pHi. Artificial pHi increases or decreases induced by weak bases or weak acids, respectively, do not induce RAB-16 mRNA expression. Clamping of the pHi at a high value with methylamine or ammonia treatment affected the ABA-induced increase of RAB-16 mRNA only slightly. However, inhibition of the ABA-induced pHi increase with weak acid or proton pump inhibitor treatments strongly inhibited the ABA-induced RAB-16 mRNA expression. We conclude that, although the ABA-induced the pHi increase is correlated with and even precedes the induction of RAB-16 mRNA expression and is an essential component of the transduction pathway leading from the hormone to gene expression, it is not sufficient to cause such expression.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aerts R. J., Durston A. J., Moolenaar W. H. Cytoplasmic pH and the regulation of the Dictyostelium cell cycle. Cell. 1985 Dec;43(3 Pt 2):653–657. doi: 10.1016/0092-8674(85)90237-5. [DOI] [PubMed] [Google Scholar]
- Akiyama T., Takagi S., Sankawa U., Inari S., Saitô H. Saponin-cholesterol interaction in the multibilayers of egg yolk lecithin as studied by deuterium nuclear magnetic resonance: digitonin and its analogues. Biochemistry. 1980 Apr 29;19(9):1904–1911. doi: 10.1021/bi00550a027. [DOI] [PubMed] [Google Scholar]
- Bright G. R., Fisher G. W., Rogowska J., Taylor D. L. Fluorescence ratio imaging microscopy: temporal and spatial measurements of cytoplasmic pH. J Cell Biol. 1987 Apr;104(4):1019–1033. doi: 10.1083/jcb.104.4.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cross J. W., Briggs W. R. Auxin receptors of maize coleoptile membranes do not have ATPase activity. Plant Physiol. 1978 Apr;61(4):581–584. doi: 10.1104/pp.61.4.581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dure L., 3rd, Greenway S. C., Galau G. A. Developmental biochemistry of cottonseed embryogenesis and germination: changing messenger ribonucleic acid populations as shown by in vitro and in vivo protein synthesis. Biochemistry. 1981 Jul 7;20(14):4162–4168. doi: 10.1021/bi00517a033. [DOI] [PubMed] [Google Scholar]
- Felle H., Brummer B., Bertl A., Parish R. W. Indole-3-acetic acid and fusicoccin cause cytosolic acidification of corn coleoptile cells. Proc Natl Acad Sci U S A. 1986 Dec;83(23):8992–8995. doi: 10.1073/pnas.83.23.8992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fiskum G. Intracellular levels and distribution of Ca2+ in digitonin-permeabilized cells. Cell Calcium. 1985 Apr;6(1-2):25–37. doi: 10.1016/0143-4160(85)90032-6. [DOI] [PubMed] [Google Scholar]
- Gehring C. A., Irving H. R., Parish R. W. Effects of auxin and abscisic acid on cytosolic calcium and pH in plant cells. Proc Natl Acad Sci U S A. 1990 Dec 15;87(24):9645–9649. doi: 10.1073/pnas.87.24.9645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcotte W. R., Jr, Russell S. H., Quatrano R. S. Abscisic acid-responsive sequences from the em gene of wheat. Plant Cell. 1989 Oct;1(10):969–976. doi: 10.1105/tpc.1.10.969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mundy J., Chua N. H. Abscisic acid and water-stress induce the expression of a novel rice gene. EMBO J. 1988 Aug;7(8):2279–2286. doi: 10.1002/j.1460-2075.1988.tb03070.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rink T. J., Tsien R. Y., Pozzan T. Cytoplasmic pH and free Mg2+ in lymphocytes. J Cell Biol. 1982 Oct;95(1):189–196. doi: 10.1083/jcb.95.1.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders D., Hansen U. P., Slayman C. L. Role of the plasma membrane proton pump in pH regulation in non-animal cells. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5903–5907. doi: 10.1073/pnas.78.9.5903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroeder J. I., Hedrich R. Involvement of ion channels and active transport in osmoregulation and signaling of higher plant cells. Trends Biochem Sci. 1989 May;14(5):187–192. doi: 10.1016/0968-0004(89)90272-7. [DOI] [PubMed] [Google Scholar]
- Skriver K., Mundy J. Gene expression in response to abscisic acid and osmotic stress. Plant Cell. 1990 Jun;2(6):503–512. doi: 10.1105/tpc.2.6.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timmers A. C., Reiss H. D., Schel J. H. Digitonin-aided loading of Fluo-3 into embryogenic plant cells. Cell Calcium. 1991 Jul;12(7):515–521. doi: 10.1016/0143-4160(91)90033-b. [DOI] [PubMed] [Google Scholar]
- Van Duijn B., Inouye K. Regulation of movement speed by intracellular pH during Dictyostelium discoideum chemotaxis. Proc Natl Acad Sci U S A. 1991 Jun 1;88(11):4951–4955. doi: 10.1073/pnas.88.11.4951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang M., Van Duijn B., Schram A. W. Abscisic acid induces a cytosolic calcium decrease in barley aleurone protoplasts. FEBS Lett. 1991 Jan 14;278(1):69–74. doi: 10.1016/0014-5793(91)80086-i. [DOI] [PubMed] [Google Scholar]