Abstract
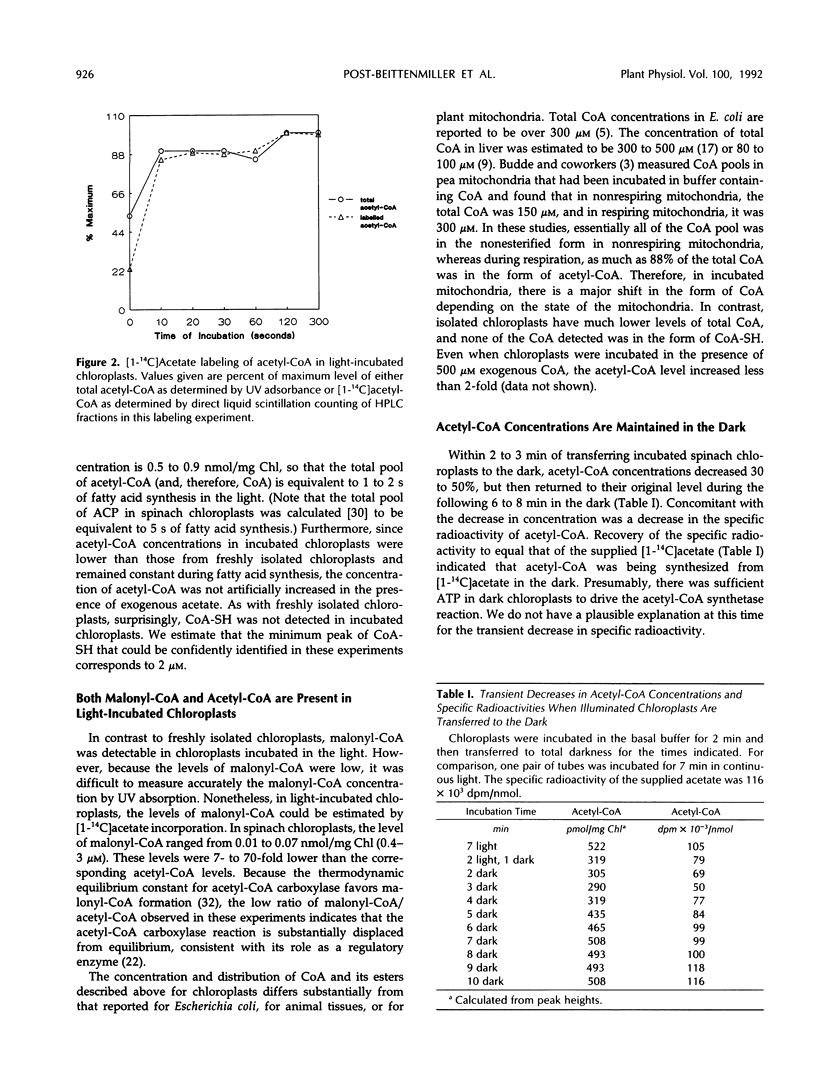
In previous work (D. Post-Beittenmiller, J.G. Jaworski, J.B. Ohlrogge [1991] J Biol Chem 266: 1858-1865), the in vivo acyl-acyl carrier protein (ACP) pools were measured in spinach (Spinacia oleracea) leaves and changes in their levels were compared to changes in the rates of fatty acid biosynthesis. To further examine the pools of substrates and cofactors for fatty acid biosynthesis and to evaluate metabolic regulation of this pathway, we have now examined the coenzyme A (CoA) and short chain acyl-CoA pools, including acetyl- and malonyl-CoA, in isolated spinach and pea (Pisum sativum) chloroplasts. In addition, the relationships of the acetyl- and malonyl-CoA pools to the acetyl- and malonyl-ACP pools have been evaluated. These studies have led to the following conclusions: (a) Essentially all of the CoA (31-54 μm) in chloroplasts freshly isolated from light-grown spinach leaves or pea seedling was in the form of acetyl-CoA. (b) Chloroplasts contain at least 77% of the total leaf acetyl-CoA, based on comparison of acetyl-CoA levels in chloroplasts and total leaf. (c) CoA-SH was not detected either in freshly isolated chloroplasts or in incubated chloroplasts and is, therefore, less than 2 μm in the stroma. (d) The malonyl-CoA:ACP transacylase reaction is near equilibrium in both light- and dark-incubated chloroplasts, whereas the acetyl-CoA:ACP transacylase reaction is far from equilibrium in light-incubated chloroplasts. However, the acetyl-CoA:ACP transacylase reaction comes nearer to equilibrium when chloroplasts are incubated in the dark. (e) Malonyl-CoA and -ACP could be detected in isolated chloroplasts only during light incubations, and increased with increased rates of fatty acid biosynthesis. In contrast, both acetyl-CoA and acetyl-ACP were detectable in the absence of fatty acid biosynthesis, and acetyl-ACP decreased with increased rates of fatty acid biosynthesis. Together these data have provided direct in situ evidence that acetyl-CoA carboxylase plays a regulatory role in chloroplast fatty acid biosynthesis.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browse J., Roughan P. G., Slack C. R. Light control of fatty acid synthesis and diurnal fluctuations of fatty acid composition in leaves. Biochem J. 1981 Apr 15;196(1):347–354. doi: 10.1042/bj1960347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Budde R. J., Fang T. K., Randall D. D., Miernyk J. A. Acetyl-coenzyme a can regulate activity of the mitochondrial pyruvate dehydrogenase complex in situ. Plant Physiol. 1991 Jan;95(1):131–136. doi: 10.1104/pp.95.1.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerović Z. G., Plesnicar M. An improved procedure for the isolation of intact chloroplasts of high photosynthetic capacity. Biochem J. 1984 Oct 15;223(2):543–545. doi: 10.1042/bj2230543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corkey B. E. Analysis of acyl-coenzyme A esters in biological samples. Methods Enzymol. 1988;166:55–70. doi: 10.1016/s0076-6879(88)66011-3. [DOI] [PubMed] [Google Scholar]
- Eastwell K. C., Stumpf P. K. Regulation of Plant Acetyl-CoA Carboxylase by Adenylate Nucleotides. Plant Physiol. 1983 May;72(1):50–55. doi: 10.1104/pp.72.1.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardiner S. E., Heinz E., Roughan P. G. Rates and products of long-chain Fatty Acid synthesis from [1-C]acetate in chloroplasts isolated from leaves of 16:3 and 18:3 plants. Plant Physiol. 1984 Apr;74(4):890–896. doi: 10.1104/pp.74.4.890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guynn R. W., Veloso D., Veech R. L. The concentration of malonyl-coenzyme A and the control of fatty acid synthesis in vivo. J Biol Chem. 1972 Nov 25;247(22):7325–7331. [PubMed] [Google Scholar]
- Heinz E., Roughan P. G. Similarities and differences in lipid metabolism of chloroplasts isolated from 18:3 and 16:3 plants. Plant Physiol. 1983 Jun;72(2):273–279. doi: 10.1104/pp.72.2.273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaworski J. G., Clough R. C., Barnum S. R. A Cerulenin Insensitive Short Chain 3-Ketoacyl-Acyl Carrier Protein Synthase in Spinacia oleracea Leaves. Plant Physiol. 1989 May;90(1):41–44. doi: 10.1104/pp.90.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Law M. Y., Charles S. A., Halliwell B. Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts. The effect of hydrogen peroxide and of Paraquat. Biochem J. 1983 Mar 15;210(3):899–903. doi: 10.1042/bj2100899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lund H., Stakkestad J. A., Skrede S. Effects of thyroid state and fasting on the concentrations of CoA and malonyl-CoA in rat liver. Biochim Biophys Acta. 1986 May 21;876(3):685–687. doi: 10.1016/0005-2760(86)90058-5. [DOI] [PubMed] [Google Scholar]
- Murphy D. J., Leech R. M. Photosynthesis of Lipids from CO(2) in Spinacia oleracea. Plant Physiol. 1981 Sep;68(3):762–765. doi: 10.1104/pp.68.3.762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuburger M., Day D. A., Douce R. Transport of coenzyme A in plant mitochondria. Arch Biochem Biophys. 1984 Feb 15;229(1):253–258. doi: 10.1016/0003-9861(84)90151-6. [DOI] [PubMed] [Google Scholar]
- Post-Beittenmiller D., Jaworski J. G., Ohlrogge J. B. In vivo pools of free and acylated acyl carrier proteins in spinach. Evidence for sites of regulation of fatty acid biosynthesis. J Biol Chem. 1991 Jan 25;266(3):1858–1865. [PubMed] [Google Scholar]
- Roughan G., Nishida I. Concentrations of long-chain acyl-acyl carrier proteins during fatty acid synthesis by chloroplasts isolated from pea (Pisum sativum), safflower (Carthamus tinctoris), and amaranthus (Amaranthus lividus) leaves. Arch Biochem Biophys. 1990 Jan;276(1):38–46. doi: 10.1016/0003-9861(90)90007-l. [DOI] [PubMed] [Google Scholar]
- Roughan P. G., Mudd J. B., McManus T. T., Slack C. R. Linoleate and alpha-linolenate synthesis by isolated spinach (Spinacia oleracea) chloroplasts. Biochem J. 1979 Dec 15;184(3):571–574. doi: 10.1042/bj1840571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roughan P. G., Slack C. R., Holland R. High rates of [1-14C]acetate incorporation into the lipid of isolated spinach chloroplasts. Biochem J. 1976 Sep 15;158(3):593–601. doi: 10.1042/bj1580593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stumpf P. K., Boardman N. K. Fat metabolism in higher plants. XXXIX. Effect of adenosine triphosphate and triton X-100 on lipid synthesis by isolated spinach chloroplasts. J Biol Chem. 1970 May 25;245(10):2579–2587. [PubMed] [Google Scholar]
- Weber A. L. Origin of fatty acid synthesis: thermodynamics and kinetics of reaction pathways. J Mol Evol. 1991;32:93–100. doi: 10.1007/BF02515381. [DOI] [PubMed] [Google Scholar]
- Williamson I. P., Wakil S. J. Studies on the mechanism of fatty acid synthesis. XVII. Preparation and general properties of acetyl coenzyme A and malonyl coenzyme A-acyl carrier protein transacylases. J Biol Chem. 1966 May 25;241(10):2326–2332. [PubMed] [Google Scholar]