Abstract
Objective
A large body of literature has examined the links between the use of dopamine replacement therapy (DRT) in Parkinson’s disease (PD) and the development of “impulsive-compulsive behaviors (ICBs).” Little is known regarding the link between the development of ICBs and health-related quality of life (HRQOL). We aimed to explore the factors that are associated with poorer HRQOL, especially in relation to DRT-induced ICBs, in a sample of PD patients.
Methods
This PARKADD (PARK: PARKinson’s disease; ADD: behavioral ADDictions) study was a prospective case‒control study initially designed to assess the factors associated with ICBs in PD patients. A prospective clinical follow-up was added, aiming to capture the long-term evolution of HRQOL in relation to ICBs occurring or worsening after the beginning of PD. We focused on sociodemographic and PD characteristics and the history or presence of ICBs. HRQOL was measured using the Parkinson’s Disease Questionnaire-8. A multivariate linear regression was performed to identify factors related to poorer HRQOL.
Results
A total of 169 patients were eligible for the follow-up study. The presence of an ICB, a higher levodopa equivalent daily dose (LEDD) and a longer PD duration were significantly associated with poorer HRQOL, with an interaction between LEDD and PD duration.
Conclusion
The presence of an ICB was related to poorer HRQOL and should be considered a crucial factor for the management of PD patients. Several studies were recently published that provide guidelines for the management of these patients, with recommendations based on two key principles: prevention and specific treatment.
Keywords: Parkinson’s disease, Impulse control disorder, Behavioral addiction, Dopamine replacement therapy, Health-related quality of life, L-DOPA equivalent daily dose
INTRODUCTION
Parkinson’s disease (PD), the most prevalent neurodegenerative disease after Alzheimer’s disease, affects more than 1.5% of people aged over 65 years. Due to the gradual loss of dopaminergic neurons in the substantia nigra and the degeneration of nerve terminals in the striatum, motor symptoms, mainly bradykinesia, tremor and muscle rigidity, emerge several years after the appearance of lesions in the nigrostriatal tract. In addition to these classic motor symptoms, PD is also associated with a broad spectrum of nonmotor symptoms, including autonomic dysfunction, depression and cognitive impairment.
As a chronic and progressive disease, PD naturally tends to worsen with time, having strong repercussions on health-related quality of life (HRQOL) [1]. Both motor and nonmotor symptoms have a significant impact on HRQOL in PD patients, and many studies have investigated their role in worsening HRQOL [2-5]. Although motor symptom intensity correlates significantly with HRQOL scores, it seems to have a limited contribution when nonmotor symptoms are taken into account [3,4,6]. Thus, several studies have found that nonmotor symptoms of PD have a greater impact on HRQOL than motor symptoms [7-9], particularly depression, which emerged as one of the main predictors of low HRQOL in the course of PD [4,7,9].
Disease progression is managed using long-term treatments, mostly consisting of dopamine replacement therapy (DRT) to increase the dopamine concentration in the brain. However, these treatments can induce several complications, including certain impulse control disorders (regarding eating, gambling, buying, or sex, so that they are called behavioral addictions) and obsessive-compulsive disorders (such as obsessive hobbying, hoarding, punding and compulsive medication use in the context of dopaminergic dysregulation syndrome). These disorders are commonly grouped under the term “impulsive-compulsive behaviors (ICBs)” [10] and can occur in up to 20% of PD patients over the course of their illness [11], which is significantly higher than the prevalence in the general population [12].
Although many studies have explored HRQOL in patients with PD, the specific role of ICBs in this association has been less investigated and has led to inconsistent results [13-19]. The aim of our study was to explore the link between HRQOL and ICBs in PD patients treated with DRT. We hypothesized that HRQOL would be poorer in patients with a history of or current DRT-induced ICBs.
MATERIALS & METHODS
Study setting and participants
The present work is a follow-up to the PARKADD (PARK: PARKinson’s disease; ADD: behavioral ADDictions; NCT 01733199) study.
The PARKADD study was a prospective case‒control study initially designed to assess the factors associated with ICBs in PD patients. For this purpose, 225 patients treated with usual DRT for idiopathic PD for at least six months were recruited from the Neurology Department of the Nantes University Hospital (France), which is a reference center for PD. Those with a history of deep brain stimulation surgery or cognitive impairment were not eligible for the study. Patients were divided into two groups according to the presence of ICBs: the “ICB-” group (patients with no ICBs occurring after the beginning of PD, n = 150) and the “ICB+” group (patients with ICBs occurring or worsening after the beginning of PD, n = 75). Recruitment took place from October 2012 to February 2016. Each patient meeting the inclusion criteria was invited for a research visit and underwent a neurological assessment, participated in a structured psychiatric interview and completed self-report questionnaires. The results of the princeps study were recently published [10].
Moreover, a prospective clinical follow-up was added to the initial procedure during the course of the study, specifically to capture the long-term evolution of HRQOL in relation to the occurrence or worsening of ICBs after the beginning of PD. Patients who agreed to take part in the follow-up, provided they were free of cognitive impairment, were given self-rated questionnaires at least 12 months after inclusion. This second assessment took place from October 2016 to January 2017. Figure 1 illustrates the study design.
Figure 1.
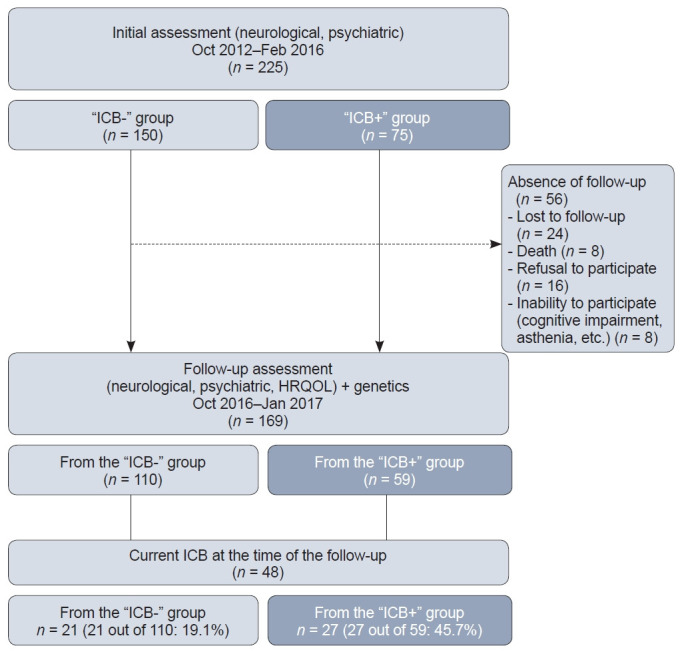
Flow diagram of the study. ICB- group, patients with no ICBs occurring after the beginning of PD; ICB+ group, patients with ICBs occurring or worsening after the beginning of PD; ICB, impulsive-compulsive behavior; PD, Parkinson’s disease; HRQOL, health-related quality of life.
The PARKADD study was conducted in accordance with the Good Clinical Practice Guidelines and the Declaration of Helsinki and was approved by the Tours Ethics Committee for the patient recruitment and follow-up (No. 2012-R17). Written informed consent was collected from all participants.
Data collection
Sociodemographic characteristics
We collected data on age and sex.
Neurological characteristics
We collected the following data related to PD: age of PD onset, PD duration, and current DRT (medication, dosage, number of pills per day). The standardized conversion formula described by Tomlinson et al. [20] was used to calculate the levodopa equivalent daily dose (LEDD), which reflects the levodopa requirement [21,22].
During the inclusion process, a face-to-face interview with a trained examiner explored the presence of lifetime and current ICBs (Minnesota Impulsive Disorder Interview [23], Giovanni criteria [24]) to establish the initial “ICB+” and “ICB-” groups. During the follow-up visit, the presence of current ICBs (de novo ICBs, ICB recurrence or persistence diagnosed at inclusion) was assessed using an adapted version of the initial interview, shortened [25] and designed to be completed as a self-report questionnaire exploring the period of the last 12 months.
Quality of life
Current HRQOL was assessed using the short form of the Parkinson’s Disease Questionnaire-8 (PDQ-8) [26]. It is a self-completion “patient-reported outcome” questionnaire designed to address aspects of functioning and well-being for individuals affected by PD. Each item assesses a domain of the core aspects of functioning: mobility; activities of daily living; emotional well-being; social support; cognitions; communication; bodily discomfort; and stigma. Each question is scored between 0 (never) and 4 (always or cannot do at all). The total score is obtained by adding the individual scores and then transformed into a score ranging from 0 to 100. The higher the score, the poorer the HRQOL.
Statistical analysis
Descriptive statistics were provided for dichotomous (number, percentage) and continuous (mean, standard deviation) variables. The links between the PDQ-8 score on the one hand and sociodemographic and clinical characteristics on the other hand were explored following a backward regression procedure, testing all variables, as well as all the interactions of order 2 between these variables. Regarding collinear variables, only one of them was tested in the model. The backward selection procedure was applied by removing nonsignificant variables one at a time, starting with the least significant variable, to select only those that were significantly associated with a higher PDQ-8 score. A two-sided p value < 0.05 was considered statistically significant. As it is known to predict HRQOL [27], PD duration was forced in the model so that the potential links observed between poorer HRQOL and other variables would be independent of PD duration.
The statistical analysis was carried out with SAS 9.4 (SAS Institute, Inc., Cary, NC, USA).
RESULTS
Description of the sample
A detailed description of the whole sample and the two groups of patients at inclusion was recently published [11]. One hundred sixty-nine patients were eligible for the follow-up study. The mean time between the inclusion and follow-up visits was 2.79 years. Approximately three-quarters of each initial group were followed-up: 110 out of the 150 patients in the “ICB-” group (73.3%) and 59 out of the 75 patients in the “ICB+” group (78.6%).
Just over two-thirds (69.8%) of patients were men, and the mean age was 65.3 (± 7.6) years. On average, PD had begun 10 years before (55.3 ± 8 years). Patients took, on average, 8 (± 3.6) pills per day for their PD, with an LEDD of 1,020 (± 594) mg. Dopamine agonists were currently used by 73.8% of the patients.
Forty-eight patients reported a current ICB at the time of the follow-up. Among them, 36 patients currently received at least one dopamine agonist medication, and 27 were initially included in the “ICB+” group, suggesting persistence or recurrence of the ICB during the period between inclusion and follow-up. The remaining 21 patients were considered to have a de novo ICB. There were no significant differences between the characteristics of patients with de novo ICBs and those with persistent or recurrent ICBs (Supplementary Table 1 in the online-only Data Supplement).
Table 1.
Health-related quality of life scores: comparison according to the dichotomous variables (n = 169)
| Variable | HRQOL | p value |
|---|---|---|
| Sex | 0.8080 | |
| Male (n = 118) | 38.8 ± 17.7 | |
| Female (n = 51) | 38.1 ± 17.5 | |
| History of ICB (at inclusion) | 0.0182 | |
| ICB- (n = 110) | 36.2 ± 16.7 | |
| ICB+ (n = 59) | 42.9 ± 18.5 | |
| Current ICB (at the time of follow-up) | 0.0089 | |
| No (n = 121) | 36.3 ± 17.7 | |
| Yes (n = 48) | 44.1 ± 16.2 |
Values are presented as mean ± standard deviation.
HRQOL, health-related quality of life; ICB-, patients with no ICBs occurring after the beginning of PD; ICB+, patients with ICBs occurring or worsening after the beginning of PD; ICB, impulsive-compulsive behavior; PD, Parkinson’s disease.
The average PDQ-8 score in the entire sample was 38.6/100 (± 17.6).
Factors associated with HRQOL
Table 1 shows the results of the univariate first step of analysis for dichotomous variables. Globally, no difference was found between men and women regarding HRQOL. Conversely, both a history of an ICB (occurring or worsening after the beginning of PD) reported at inclusion and a current ICB reported at the time of follow-up were significantly associated with poorer HRQOL at the time of follow-up.
Furthermore, the results of the Pearson correlation coefficients between the PDQ-8 score and other continuous variables collected at the time of follow-up (age, age of PD onset, PD duration, number of pills per day, LEDD) are displayed in Table 2. A significant relationship was evidenced between PDQ-8 score on the one hand and LEDD and number of pills taken per day on the other hand.
Table 2.
Health-related quality of life scores: correlation with the continuous variables’ scores (n = 169)
| Variable (at the time of follow-up) | PDQ-8 score |
|
|---|---|---|
| r * | p value | |
| Age (n = 168) | -0.07 | 0.4019 |
| Age of PD onset | -0.14 | 0.0791 |
| PD duration (n = 168) | 0.14 | 0.0695 |
| Number of pills/day | 0.21 | 0.0071 |
| LEDD | 0.25 | 0.0010 |
Pearson correlation coefficients (Prob > |r| under H0: Rho = 0).
LEDD, levodopa equivalent daily dose; PD, Parkinson’s disease; PDQ, Parkinson’s Disease Questionnaire.
The last step of the analysis consisted of building a multivariate model to explain the higher PDQ-8 scores. Due to collinearity problems, two variables selected in the first univariate step were excluded from the multivariate model: a history of an ICB at inclusion (collinear with a current ICB at the time of follow-up: 75% of the participants had both a history of an ICB and a current ICB or had no history of an ICB and no current ICB, p < 0.0001) and the number of pills taken per day (collinear with the LEDD: Pearson correlation coefficient = 0.77, p < 0.0001). In each case, we selected the variable that displayed the highest significance level in the univariate analysis. As a consequence, the multivariate analysis included the following variables: sex of the patient, presence of a current ICB at the time of follow-up, age of PD onset, PD duration at the time of follow-up, LEDD at the time of follow-up, and all the interactions of order 2 between these variables. After the backward selection procedure, the final model showed that poorer HRQOL was significantly associated with the presence of a current ICB, a higher LEDD, and a longer PD duration. Furthermore, we found a significant interaction between the last two variables (Table 3). This interaction can be understood in two ways. First, the longer the PD duration, the less the LEDD impaired the HRQOL for an equivalent LEDD. Second, the higher the LEDD, the less the PD duration impaired the HRQOL, for an equivalent PD duration.
Table 3.
Factors associated with poorer health-related quality of life (n = 169)
| Variable (at the time of follow-up) | Coefficient | CI 95% | p value |
|---|---|---|---|
| Current ICB | 6.998 | [1.223; 12.774] | 0.018 |
| LEDD | 1.647 | [0.515; 2.779] | 0.005 |
| PD duration | 1.494 | [0.321; 2.667] | 0.013 |
| LEDD * PD duration | -0.108 | [-0.211; -0.005] | 0.040 |
CI, confidence interval; ICB, impulsive-compulsive behavior; LEDD, levodopa equivalent daily dose; PD, Parkinson’s disease.
DISCUSSION
Main results
This follow-up study focused on the links between ICBs and HRQOL in PD patients. Several key findings of this work should be highlighted.
First, as expected, we found that a higher PDQ-8 score was significantly associated with the presence of a current ICB. To date, few studies have focused on the specific link between HRQOL and ICBs in PD patients, and most of them have identified ICBs as a predictor of impaired HRQOL [15-17,28-30], which our results seem to confirm. Phu et al. [17] pointed out that ICBs particularly impacted the emotional well-being dimension of HRQOL, assessed by the PDQ-39 [31], even after adjusting for the presence of depression. In our study, we used the PDQ-8, which was derived from the PDQ-39. This shortened version has been found to have good psychometric properties but does not allow for the assessment of individual domains. However, for descriptive and exploratory purposes, we calculated the mean score for each item, according to the absence or presence of a current ICB and the type of ICB (a persistent/recurrent or de novo ICB) (Supplementary Figure 1 in the online-only Data Supplement). The highest score was obtained for the Bodily Discomfort item, especially when an ICB was present. The links among PD, ICBs and HRQOL are probably complex and likely to be bidirectional. According to some authors, the unpredictable nature of PD and the inability to stop or slow its progression may result in uncertainty and psychological stress [32]. One might imagine that the development of ICBs is a coping mechanism against the stigma, distress, anxiety, or depression associated with the evolution of the disease for some patients. In this case, PD would induce negative emotions and cognitions, decreasing HRQOL, which would in turn lead to some specific behaviors (eating, gambling, etc.) to address psychological disturbances, with the progressive development of ICBs. Conversely, the presence of an ICB could be an adverse effect of DRT [12] and may impair HRQOL due to its specific damage (negative consequences on financial, social, professional, or marital domains, etc.), then inducing secondary negative cognitions and emotions.
Second, the higher the LEDD, the poorer the HRQOL, placing our results in line with those of previous works [16,29,33]. A recent report showed that the onset of motor complications as well as some nonmotor fluctuations were associated with a higher LEDD and poorer quality of life [21].
Third, the longer the PD duration, the poorer the HRQOL, as previously found by a systematic review of HRQOL determinants in PD patients [4]. Interestingly, Benge et al. [27] emphasized the importance of considering HRQOL not as a unidimensional construct but as covering several domains, with Activities in daily life, Stigma, and Communication being the three domains most strongly affected by PD duration.
Finally, HRQOL was significantly associated with the interaction between LEDD and PD duration. Two ways of understanding this interaction can be proposed. First, the longer the PD duration, the less the LEDD impaired the HRQOL for an equivalent LEDD. Second, the higher the LEDD, the less the PD duration impaired the HRQOL, for an equivalent PD duration. This interaction could reflect the highly heterogeneous presentation of PD, which evolves over time, as well as the levodopa requirement. Thus, certain symptoms appear or worsen throughout the course of the disease, in particular levodopa-resistant motor symptoms and nonmotor symptoms. For example, tremor or stiffness, symptoms that respond best to levodopa, tend to be mentioned less and less by patients with PD when asked to identify the symptoms they would most like to see improvement in as the disease progresses. Conversely, balance failure or freezing gait—motor symptoms that are levodopa-resistant—tend to be increasingly mentioned [34]. Furthermore, we cannot exclude an effect of habituation of the patients to the constraints related to their treatment over time (number of pills per day, number of times taken during the day, adverse effects, etc.).
Strengths and weaknesses
The results must be interpreted by taking the strengths and weaknesses of the study into consideration. The first limitation arose from the absence of an initial assessment of HRQOL that did not allow us to explore longitudinal changes. The second limitation is linked to the choice of the assessment method, mainly motivated by the need to minimize the duration of the evaluation and to avoid moving patients: patients were only given self-rated questionnaires, without a face-to-face clinical assessment, and the presence of pain, depression or mild cognitive impairment was not assessed, so we were not able to adjust our analyses for these symptoms that could be confounders for ICBs. Finally, HRQOL could be influenced by other chronic diseases, such as diabetes or hypertension, and their specific treatments, which were not taken into account. For these reasons, the present results should be interpreted with caution.
However, these limits were compensated by the strengths of the study. One of the most important strengths is the large number of patients, making our study one of those exploring the characteristics of PD patients who develop ICBs, with the highest power. Furthermore, just over 75% of the initial sample completed the follow-up, which was a high participation rate. Finally, this is one of the few studies exploring HRQOL related to the course of PD through ICBs, paying attention to the distinction between persistent/recurrent and de novo ICBs.
Perspectives
This follow-up PARKADD study showed that DRT-induced ICBs have an impact on HRQOL in patients with PD. It is therefore crucial to take ICBs into account to better prevent their occurrence during the course of PD and to favor remission. It is alarming that nearly half of the patients who reported an ICB at the beginning of our study still reported it at the time of follow-up, despite the therapeutic options available to regulate the behaviors, and that a de novo ICB was reported for 19% of the followed-up sample without a history of ICBs. Identifying patients at higher risk of developing an ICB is crucial to encourage predictive medicine and to better adjust treatment strategies for them. Guidelines on the best ways to monitor and manage ICBs in patients undergoing DRT are already available [35]. The first stage aims at optimizing DRT, for example, by avoiding drugs that are more likely to induce an ICB, which seem to be the most selective D3 receptors (such as pramipexole) [11]. The second stage is to propose nonpharmacological approaches, especially cognitive behavioral therapy [36,37]. In the event of a negative outcome, the third stage involves fewer conventional treatment options, particularly specific pharmacological treatments, such as antiepilectic drugs (topiramate, valproate, and zonisamide) and anti-craving drugs (naltrexone) [37,38]. It is also important to provide full and clear information on these potential adverse drug reactions to patients and to encourage them to notify their caregivers of any sign of an ICB from a preventive and participatory medicine perspective. Given that an ICB can potentially alter HRQOL by itself, it is even more important to take it into account.
The Proactive and Integrated Management and Empowerment in PD (PRIME-Parkinson) model is a new integrated model of care designed to help manage problems proactively, deliver integrated, multidisciplinary care, and empower patients and their caregivers [39]. It could represent an innovative approach for treating PD and is expected to improve HRQOL [40].
Footnotes
Conflicts of Interest
The authors have no financial conflicts of interest.
Funding Statement
The PARKADD study was supported by a grant from the French National Research Agency (ANR-SAMENTA call for proposal, 2012, decision number: ANR 12 SAMA 013 01). It received additional funding as part of labeling by the Atlanpole Biotherapies competitiveness cluster.
This research was conducted at the initiative of and coordinated by the UIC “Psychiatrie et Santé Mentale” of Nantes University Hospital. Nantes University Hospital is the sponsor of this study. There were no constraints on publishing.
Author Contributions
Conceptualization: Marie Grall-Bronnec, Gaëlle Challet-Bouju, Tiphaine Rouaud, Pascal Derkinderen. Data curation: Marie Grall-Bronnec, Gaëlle Challet-Bouju, Tiphaine Rouaud, Pascal Derkinderen, Juliette Leboucher, Elsa Thiabaud, Monica Roy. Formal analysis: Marie Grall-Bronnec, Caroline Victorri-Vigneau, Jean-Benoit Hardouin. Funding acquisition: Marie Grall-Bronnec, Gaëlle Challet-Bouju. Investigation: Marie Grall-Bronnec, Gaëlle Challet-Bouju, Tiphaine Rouaud, Pascal Derkinderen. Methodology: Marie Grall-Bronnec, Gaëlle Challet-Bouju, Tiphaine Rouaud, Pascal Derkinderen, Juliette Leboucher, Jean-Benoit Hardouin. Project administration: Gaëlle Challet-Bouju, Juliette Leboucher. Writing—original draft: Marie Grall-Bronnec, Audrey Verholleman. Writing—review & editing: all authors.
Supplementary Materials
The online-only Data Supplement is available with this article at https://doi.org/10.14802/jmd.23042.
Characteristics of patients with de novo ICB and those whose ICB persisted or re-occurred (n = 48)
HRQOL according to the absence or presence of a current ICB, be it was persistent/recurrent or de novo (n = 169). HRQOL, health-related quality of life; ICB: impulsive-compulsive behavior.
REFERENCES
- 1.Opara JA, Brola W, Leonardi M, Błaszczyk B. Quality of life in Parkinson’s disease. J Med Life. 2012;5:375–381. [PMC free article] [PubMed] [Google Scholar]
- 2.Park HR, Youn J, Cho JW, Oh ES, Kim JS, Park S, et al. Characteristic motor and nonmotor symptoms related to quality of life in drug-naïve patients with late-onset Parkinson disease. Neurodegener Dis. 2018;18:19–25. doi: 10.1159/000484249. [DOI] [PubMed] [Google Scholar]
- 3.Schrag A, Jahanshahi M, Quinn N. What contributes to quality of life in patients with Parkinson’s disease? J Neurol Neurosurg Psychiatry. 2000;69:308–312. doi: 10.1136/jnnp.69.3.308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Soh SE, Morris ME, McGinley JL. Determinants of health-related quality of life in Parkinson’s disease: a systematic review. Parkinsonism Relat Disord. 2011;17:1–9. doi: 10.1016/j.parkreldis.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 5.Martínez-Martín P, Rodríguez-Blázquez C, Forjaz MJ, Alvarez-Sánchez M, Arakaki T, Bergareche-Yarza A, et al. Relationship between the MDSUPDRS domains and the health-related quality of life of Parkinson’s disease patients. Eur J Neurol. 2014;21:519–524. doi: 10.1111/ene.12349. [DOI] [PubMed] [Google Scholar]
- 6.He L, Lee EY, Sterling NW, Kong L, Lewis MM, Du G, et al. The key determinants to quality of life in Parkinson’s disease patients: results from the Parkinson’s disease biomarker program (PDBP) J Parkinsons Dis. 2016;6:523–532. doi: 10.3233/JPD-160851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li H, Zhang M, Chen L, Zhang J, Pei Z, Hu A, et al. Nonmotor symptoms are independently associated with impaired health-related quality of life in Chinese patients with Parkinson’s disease. Mov Disord. 2010;25:2740–2746. doi: 10.1002/mds.23368. [DOI] [PubMed] [Google Scholar]
- 8.Martinez-Martin P, Rodriguez-Blazquez C, Kurtis MM, Chaudhuri KR; NMSS Validation Group. The impact of non-motor symptoms on healthrelated quality of life of patients with Parkinson’s disease. Mov Disord. 2011;26:399–406. doi: 10.1002/mds.23462. [DOI] [PubMed] [Google Scholar]
- 9.Santos-García D, Sanjurjo LF, Macías M, Llaneza M, Carpintero P, de la Fuente-Fernández R. Long-term exposure to duodenal levodopa/carbidopa infusion therapy improves quality of life in relation especially to mobility, activities of daily living, and emotional well-being. Acta Neurol Scand. 2012;125:187–191. doi: 10.1111/j.1600-0404.2011.01523.x. [DOI] [PubMed] [Google Scholar]
- 10.Grall-Bronnec M, Victorri-Vigneau C, Rouaud T, Verholleman A, Schreck B, Leboucher J, et al. Parkinson’s disease and iatrogenic impulsive-compulsive behaviors: a case/non-case study to build a complete model of individual vulnerability. J Behav Addict. 2022;11:766–777. doi: 10.1556/2006.2022.00051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Weintraub D, Claassen DO. Impulse control and related disorders in Parkinson’s disease. Int Rev Neurobiol. 2017;133:679–717. doi: 10.1016/bs.irn.2017.04.006. [DOI] [PubMed] [Google Scholar]
- 12.Grall-Bronnec M, Victorri-Vigneau C, Donnio Y, Leboucher J, Rousselet M, Thiabaud E, et al. Dopamine agonists and impulse control disorders: a complex association. Drug Saf. 2018;41:19–75. doi: 10.1007/s40264-017-0590-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Balestrino R, Martinez-Martin P. Neuropsychiatric symptoms, behavioural disorders, and quality of life in Parkinson’s disease. J Neurol Sci. 2017;373:173–178. doi: 10.1016/j.jns.2016.12.060. [DOI] [PubMed] [Google Scholar]
- 14.Dujardin K, Sgambato V. Neuropsychiatric disorders in Parkinson’s disease: what do we know about the role of dopaminergic and non-dopaminergic systems? Front Neurosci. 2020;14:25. doi: 10.3389/fnins.2020.00025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Erga AH, Alves G, Tysnes OB, Pedersen KF. Impulsive and compulsive behaviors in Parkinson’s disease: impact on quality of and satisfaction with life, and caregiver burden. Parkinsonism Relat Disord. 2020;78:27–30. doi: 10.1016/j.parkreldis.2020.07.007. [DOI] [PubMed] [Google Scholar]
- 16.Leroi I, Ahearn DJ, Andrews M, McDonald KR, Byrne EJ, Burns A. Behavioural disorders, disability and quality of life in Parkinson’s disease. Age Ageing. 2011;40:614–621. doi: 10.1093/ageing/afr078. [DOI] [PubMed] [Google Scholar]
- 17.Phu AL, Xu Z, Brakoulias V, Mahant N, Fung VS, Moore GD, et al. Effect of impulse control disorders on disability and quality of life in Parkinson’s disease patients. J Clin Neurosci. 2014;21:63–66. doi: 10.1016/j.jocn.2013.02.032. [DOI] [PubMed] [Google Scholar]
- 18.Rieu I, Houeto JL, Pereira B, De Chazeron I, Bichon A, Chéreau I, et al. Impact of mood and behavioral disorders on quality of life in Parkinson’s disease. J Parkinsons Dis. 2016;6:267–277. doi: 10.3233/JPD-150747. [DOI] [PubMed] [Google Scholar]
- 19.Vela L, Martínez Castrillo JC, García Ruiz P, Gasca-Salas C, Macías Macías Y, Pérez Fernández E, et al. The high prevalence of impulse control behaviors in patients with early-onset Parkinson’s disease: a cross-sectional multicenter study. J Neurol Sci. 2016;368:150–154. doi: 10.1016/j.jns.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 20.Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord. 2010;25:2649–2653. doi: 10.1002/mds.23429. [DOI] [PubMed] [Google Scholar]
- 21.Julien C, Hache G, Dulac M, Dubrou C, Castelnovo G, Giordana C, et al. The clinical meaning of levodopa equivalent daily dose in Parkinson’s disease. Fundam Clin Pharmacol. 2021;35:620–630. doi: 10.1111/fcp.12646. [DOI] [PubMed] [Google Scholar]
- 22.Nyholm D, Karlsson E, Lundberg M, Askmark H. Large differences in levodopa dose requirement in Parkinson’s disease: men use higher doses than women. Eur J Neurol. 2010;17:260–266. doi: 10.1111/j.1468-1331.2009.02866.x. [DOI] [PubMed] [Google Scholar]
- 23.Grant JE, Chamberlain SR. Impulsive action and impulsive choice across substance and behavioral addictions: cause or consequence? Addict Behav. 2014;39:1632–1639. doi: 10.1016/j.addbeh.2014.04.022. [DOI] [PubMed] [Google Scholar]
- 24.Giovannoni G, O’Sullivan JD, Turner K, Manson AJ, Lees AJ. Hedonistic homeostatic dysregulation in patients with Parkinson’s disease on dopamine replacement therapies. J Neurol Neurosurg Psychiatry. 2000;68:423–428. doi: 10.1136/jnnp.68.4.423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee JY, Kim JM, Kim JW, Cho J, Lee WY, Kim HJ, et al. Association between the dose of dopaminergic medication and the behavioral disturbances in Parkinson disease. Parkinsonism Relat Disord. 2010;16:202–207. doi: 10.1016/j.parkreldis.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 26.Jenkinson C, Fitzpatrick R, Peto V, Greenhall R, Hyman N. The PDQ-8:development and validation of a short-form Parkinson’s disease questionnaire. Psych Health. 1997;12:805–814. [Google Scholar]
- 27.Benge JF, Kekecs Z, Encarnacion E, Ainslie M, Herff C, Elkins G, et al. Duration of disease does not equally influence all aspects of quality of life in Parkinson’s disease. J Clin Neurosci. 2016;28:102–106. doi: 10.1016/j.jocn.2015.09.019. [DOI] [PubMed] [Google Scholar]
- 28.Jesús S, Labrador-Espinosa MA, Adarmes AD, Méndel-Del Barrio C, Martínez-Castrillo JC, Alonso-Cánovas A, et al. Non-motor symptom burden in patients with Parkinson’s disease with impulse control disorders and compulsive behaviours: results from the COPPADIS cohort. Sci Rep. 2020;10:16893. doi: 10.1038/s41598-020-73756-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lubomski M, Davis RL, Sue CM. Health-related quality of life for Parkinson’s disease patients and their caregivers. J Mov Disord. 2021;14:42–52. doi: 10.14802/jmd.20079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Santos-García D, de Deus Fonticoba T, Cores Bartolomé C, Suárez Castro E, Jesús S, Mir P, et al. Depression is associated with impulse-compulsive behaviors in Parkinson’s disease. J Affect Disord. 2021;280(Pt B):77–89. doi: 10.1016/j.jad.2020.11.075. [DOI] [PubMed] [Google Scholar]
- 31.Peto V, Jenkinson C, Fitzpatrick R, Greenhall R. The development and validation of a short measure of functioning and well being for individuals with Parkinson’s disease. Qual Life Res. 1995;4:241–248. doi: 10.1007/BF02260863. [DOI] [PubMed] [Google Scholar]
- 32.Austin KW, Ameringer SW, Starkweather AR, Cloud LJ, Sturgill JL, Elswick RK., Jr Biobehavioral framework of symptom and health outcomes of uncertainty and psychological stress in Parkinson disease. J Neurosci Nurs. 2016;48:E2–E9. doi: 10.1097/JNN.0000000000000244. [DOI] [PubMed] [Google Scholar]
- 33.Behari M, Srivastava AK, Pandey RM. Quality of life in patients with Parkinson’s disease. Parkinsonism Relat Disord. 2005;11:221–226. doi: 10.1016/j.parkreldis.2004.12.005. [DOI] [PubMed] [Google Scholar]
- 34.Port RJ, Rumsby M, Brown G, Harrison IF, Amjad A, Bale CJ. People with Parkinson’s disease: what symptoms do they most want to improve and how does this change with disease duration? J Parkinsons Dis. 2021;11:715–724. doi: 10.3233/JPD-202346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.National Institute for Health and Care Excellence . Parkinson’s disease in adults: diagnosis and management. London: National Institute for Health and Care Excellence (NICE); 2017. pp. 12–14. [Google Scholar]
- 36.Jiménez-Murcia S, Bove FI, Israel M, Steiger H, Fernández-Aranda F, Alvarez-Moya E, et al. Cognitive-behavioral therapy for pathological gambling in Parkinson’s disease: a pilot controlled study. Eur Addict Res. 2012;18:265–274. doi: 10.1159/000337442. [DOI] [PubMed] [Google Scholar]
- 37.Seppi K, Ray Chaudhuri K, Coelho M, Fox SH, Katzenschlager R, Perez Lloret S, et al. Update on treatments for nonmotor symptoms of Parkinson’s disease-an evidence-based medicine review. Mov Disord. 2019;34:180–198. doi: 10.1002/mds.27602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ramirez-Zamora A, Gee L, Boyd J, Biller J. Treatment of impulse control disorders in Parkinson’s disease: practical considerations and future directions. Expert Rev Neurother. 2016;16:389–399. doi: 10.1586/14737175.2016.1158103. [DOI] [PubMed] [Google Scholar]
- 39.Tenison E, Smink A, Redwood S, Darweesh S, Cottle H, van Halteren A, et al. Proactive and integrated management and empowerment in Parkinson’s disease: designing a new model of care. Parkinsons Dis. 2020;2020:8673087. doi: 10.1155/2020/8673087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bloem BR, Henderson EJ, Dorsey ER, Okun MS, Okubadejo N, Chan P, et al. Integrated and patient-centred management of Parkinson’s disease: a network model for reshaping chronic neurological care. Lancet Neurol. 2020;19:623–634. doi: 10.1016/S1474-4422(20)30064-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Characteristics of patients with de novo ICB and those whose ICB persisted or re-occurred (n = 48)
HRQOL according to the absence or presence of a current ICB, be it was persistent/recurrent or de novo (n = 169). HRQOL, health-related quality of life; ICB: impulsive-compulsive behavior.


