Abstract
Worldwide, the incidence of major depressive disorder (MDD) is increasing annually, resulting in greater economic and social burdens. Moreover, the pathological mechanisms of MDD and the mechanisms underlying the effects of pharmacological treatments for MDD are complex and unclear, and additional diagnostic and therapeutic strategies for MDD still are needed. The currently widely accepted theories of MDD pathogenesis include the neurotransmitter and receptor hypothesis, hypothalamic-pituitary-adrenal (HPA) axis hypothesis, cytokine hypothesis, neuroplasticity hypothesis and systemic influence hypothesis, but these hypothesis cannot completely explain the pathological mechanism of MDD. Even it is still hard to adopt only one hypothesis to completely reveal the pathogenesis of MDD, thus in recent years, great progress has been made in elucidating the roles of multiple organ interactions in the pathogenesis MDD and identifying novel therapeutic approaches and multitarget modulatory strategies, further revealing the disease features of MDD. Furthermore, some newly discovered potential pharmacological targets and newly studied antidepressants have attracted widespread attention, some reagents have even been approved for clinical treatment and some novel therapeutic methods such as phototherapy and acupuncture have been discovered to have effective improvement for the depressive symptoms. In this work, we comprehensively summarize the latest research on the pathogenesis and diagnosis of MDD, preventive approaches and therapeutic medicines, as well as the related clinical trials.
Subject terms: Diseases of the nervous system, Cellular neuroscience
Introduction
Major depressive disorder (MDD), a main cause of disability worldwide, is characterized by physical changes such as tiredness, weight loss, and appetite loss. Anhedonia is a classic feature of MDD, and MDD is also accompanied by a lack of drive, sleep issues, cognitive challenges, and emotional symptoms such as guilt.1 The prevalence of depression is increasing yearly. About 300 million people in the world are affected by MDD, which has become one of the main causes of disability.2 In 2018, MDD ranked third in terms of disease burden according to the WHO, and it is predicted to rank first by 2030.3 Pregnant women, elderly people, children, and others have a higher incidence rate of MDD, which may be related to genetic, psychological, and social factors.4 Depression can be accompanied by recurrent seizures, which may occur even during remission or persist for longer than the disease itself.5 Pharmacological therapies for MDD can effectively control symptoms; thus, patients may experience recurrence within a short time after discontinuing medication.6 During recurrence, the patient experiences symptoms of low mood, loss of interest in life, fatigue, delayed thinking, and repeated fluctuations in mental state.7
There is a certain correlation between the occurrence of MDD and social development.8 A survey reported that with the development of the economy and increased life pressure, MDD has begun to emerge at a younger age, and the incidence of MDD in women is approximately twice that in men.9 Specifically, women are more likely to develop depressive symptoms when they encounter social emergencies or are under significant stress.8 Additionally, autumn and winter have been reported to be associated with a high incidence of MDD, namely, seasonal depression.10
The clinical symptoms of MDD include a depressed mood, loss of interest, changes in weight or appetite, and increased likelihood of committing suicide.11 These symptoms are also listed as the criteria for MDD in the Diagnostic and Statistical Manual of Mental Disorders (DSM-5).12 In addition to the criteria listed in the DSM-5, the criteria reported in the International Classification of Diseases (ICD-10) are also used to guide clinical diagnosis.13 However, due to the lack of characteristic symptoms and objective diagnostic evidence for MDD, identification and early prevention are difficult in the clinic.14
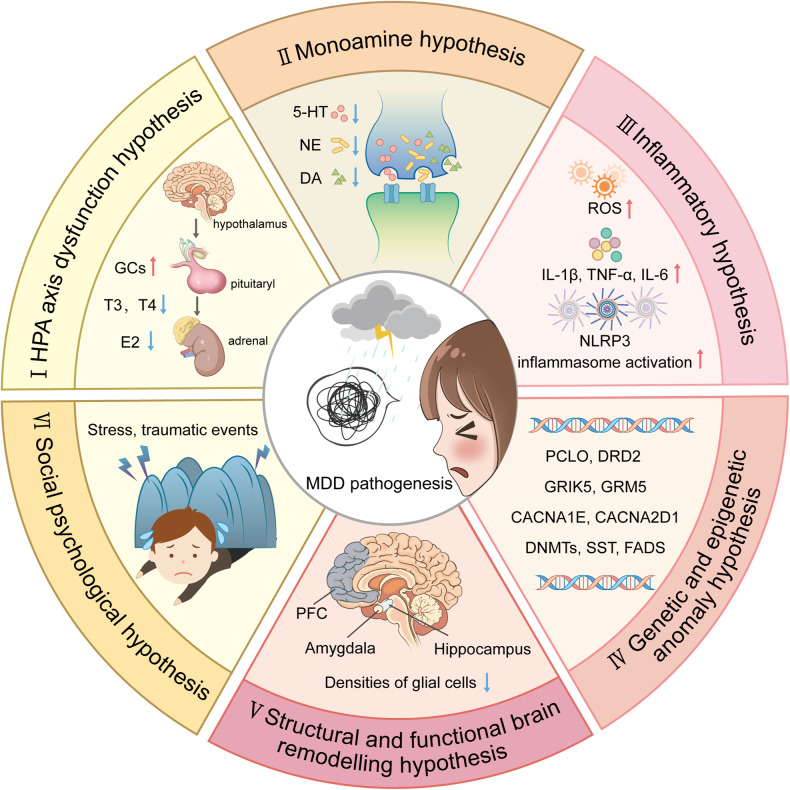
Due to the complexity of the pathological mechanism of MDD, accurate diagnostic approaches and pharmacological therapeutic strategies are relatively limited. Several hypothesis were developed to explain MDD pathogenesis pathogenic including (i) the hypothalamic‒pituitary‒adrenal (HPA) axis dysfunction hypothesis, (ii) the monoamine hypothesis, (iii) the inflammatory hypothesis, (iv) the genetic and epigenetic anomaly hypothesis, (v) the structural and functional brain remodeling hypothesis, and (vi) the social psychological hypothesis3,15,16 (Fig. 1). However, none of these hypotheses alone can fully explain the pathological basis of MDD, while many mechanisms proposed by these hypotheses interact with each other. In recent years, great progress has been made in identifying novel pharmacological therapies, diagnostic criteria, and nonpharmacological preventive measures for MDD, initiating related clinical trials. Specifically, increasing evidence suggests that astrocytic dysfunction plays a substantial role in MDD.17 Pharmacological ablation of astrocytes in the medial prefrontal cortex (mPFC) causes depressive-like symptoms in experimental animals,18 and postmortem studies of patients with MDD have shown reduced densities of glial cells in the prefrontal cortex (PFC), hippocampus and amygdala.19 In addition, glial fibrillary acidic protein (GFAP), one of the markers of astrocytes, is expressed at various levels,20 and the levels of connexins,21 glutamine synthase (GS), glutamate transporter-1 (GLT-1),21,22 and aquaporin-4 (AQP4)23 are reduced in patients with MDD.
Fig. 1.
An outline map of the hypotheses to explain MDD pathogenesis. (I) HPA axis dysfunction hypothesis: high levels of glucocorticoids (GCs) play a core role in the pathogenesis of MDD, and thyroid hormone (TH) and estrogen are also involved in functions of the HPA axis; (II) the monoamine hypothesis: the functional deficiency of serotonin (5-HT), dopamine (DA) and norepinephrine (NE) are the main pathogenesis of MDD; (III) the inflammatory hypothesis: the neuro-inflammation induced by reactive oxygen species (ROS), inflammatory cytokines and inflammasomes activation is suggested to promote the occurrence of MDD; (IV) the genetic and epigenetic anomaly hypothesis: some genes are susceptible in the patients with MDD, including presynaptic vesicle trafficking (PCLO), D2 subtype of the dopamine receptor (DRD2), glutamate ionotropic receptor kainate type subunit 5 (GRIK5), metabotropic glutamate receptor 5 (GRM5), calcium voltage-gated channel subunit alpha1 E (CACNA1E), calcium voltage-gated channel auxiliary subunit alpha2 delta1(CACNA2D1), DNA methyltransferases (DNMTs), transcription levels of somatostatin (SST), fatty acid desaturase (FADS); (V) the structural and functional brain remodeling hypothesis: the postmortem results of patients with MDD are mostly associated with the reduced densities of glial cells in the prefrontal cortex (PFC), hippocampus, and amygdala; (VI) the social psychological hypothesis: the traumatic or stressful life events are the high risks of the occurrence of MDD. Adobe Illustrator was used to generate this figure
In this review, we summarize the latest research on the etiology, pathogenesis, diagnosis, prevention, mechanism, and pharmacological and nonpharmacological treatment of MDD as well as related clinical experiments.
Potential etiologies and pathogenic hypotheses
The common pathogenic factors
Although the etiology of MDD is still unclear, it is widely accepted that MDD is associated with multiple pathogenic factor. In addition to well-known mental factors, MDD is also related to genetic factors, social stress, and even other common chronic diseases. Therefore, the etiology of MDD cannot be described from the perspective of a single factor.
Genetic factors
Although the etiology of MDD is still unclear, numerous studies have been performed and various models have been employed to explore the genetic factors, environmental factors and gene-environment interactions related to the disease.24 Recent family, twin, and adoption studies suggests that genetic factors play a crucial role in the occurrence of MDD.25 As a genetically diverse illness, MDD has a heritability of 30–50%.26 Over 100 gene loci, including those associated with presynaptic vesicle trafficking (PCLO), dopaminergic neurotransmission (a primary target of antipsychotics), glutamate ionotropic receptor kainate type subunit 5 (GRIK5), and metabotropic glutamate receptor 5 (GRM5), and neuronal calcium signaling such as calcium voltage-gated channel subunit alpha1 E (CACNA1E) and calcium voltage-gated channel auxiliary subunit alpha2 delta1 (CACNA2D1), are found to be associated with an increased risk of MDD by genome-wide association studies.19,27,28 In addition, rare copy number variants are also identified to be related to MDD risk, there may be three copy number variants (CNV) loci associated with Prader-Willis syndrome: 1q21.1 duplication, 15q11-13, and 16p11.2. However, no single genetic variation has been found to increase the risk of MDD thus far.26 Genome Wide Association Studies (GWAS) identified 178 genetic risk loci and proposed over 200 candidate gene, using of biobank data, novel imputation methods, combined with clinical cases improved the ability to identify MDD specific pathways.29 In the study of human MDD transcriptome, there are defects in the transcription levels of somatostatin (SST) in the subgenus anterior cingulate cortex and amygdala of MDD patients,30,31 and SST levels are directly involved in the cellular processes that affect the synaptic output of intermediate neuronal circuits.32 Recent studies revealed that gender specific genomic differences in MDD patients, the downregulation of the MDD-related gene Dusp6 in females leads to an increased susceptibility to stress, but this expression is not present in male mice.33 In addition, studies of drug gene interactions, transcriptional genes associated with the risk of MDD are also reported, such as D2 subtype of the dopamine receptor (DRD2) and fatty acid desaturase (FADS),34 which may serve as promising new targets for therapeutic intervention points. Thus, genetic variants are expected to have only minor effects on the overall risk of disease, and various hereditary factors combined with environmental factors such as stress are likely more essential for the development of MDD.35
Stress factors
In addition to heritable factors, environmental influences such as stress also significantly contribute to the development of MDD, both independently and in conjunction with genetic factors.26 Numerous studies have suggested that adverse life events can lead to the development of MDD.18 A major depressive episode always follows a traumatic or stressful life event. In particular, severe events such as job loss, extramarital affairs and divorce are known to provoke the onset of the disease.36 The exact pathological mechanism by which social stress results in the development of MDD is still not known, mainly due to the difficulty of separating social factors from genetic factors in patients and the impracticality of exposing disease model animals to relevant environmental factors. It has been proved that the changes in the structure and function of neurons may occur under the chronic stress and lead to the occurrence of MDD.37,38 In some MDD patients, stress leads to long-term elevated glucocorticoids, resulting in synaptic structural changes and remodeling, and the stress-induced hyperactivity of the HPA axis leads to negative feedback imbalance of the HPA axis, which is also related to depression.39 Studies on damage to microglia and astrocytes suggest the significance of glial cells in the development of environmental factor-induced depression-like behaviors in mice.40 In addition, our previous studies proved that chronic environmental stress-induced depressive-like behaviors in mice can be dependent on purinergic ligand-gated ion channel 7 receptor (P2X7R) activation in astrocytes.41
Comorbidity factors
The existence of various physiological and psychological comorbidities in patients with depression reveals a clear link between physical and mental health, which has given us a better understanding of MDD. The presence of MDD is a risk factor for a variety of complications, including neurodegenerative diseases (such as dementia, Alzheimer’s disease, and Parkinson’s disease), cardiovascular diseases (such as ischemic coronary artery disease and myocardial infarction), metabolic and endocrine diseases (such as obesity in females and diabetes in males), and some autoimmune diseases.42,43 The relationship between the onset of MDD and several diseases is complex and potentially bidirectional in nature.44 The impact of depression on society and the economy is increased by the existence of comorbidities.45 Specifically, in 2018, comorbid disorders rather than MDD itself were responsible for 63% of all costs related to MDD in the United States.46,47 Furthermore, compared to people without depression, patients with MDD have been demonstrated to have a shorter life expectancy.48 Additionally, the worsening of comorbidities could be a factor in the premature mortality of MDD patients.44
Neurotransmitter and receptor hypothesis
The traditional monoamine theory contends that in addition to common pathogenic factors, deficiencies in monoamine neurotransmitters, such as serotonin (5-HT), dopamine (DA) and norepinephrine (NE), are the root cause of clinical depression.49 Selective serotonin reuptake inhibitors (SSRIs), a class of antidepressants that have been proven to successfully treat clinical depression, were developed in response to this hypothesis, which was derived primarily on the basis of the pharmacological mechanism of drug that were accidentally discovered to act as antidepressants. It is also crucial to note that astrocytes express NE transporter (NETT) and 5-HT transporter (SERT), which are the targets of some traditional antidepressants.50 A previous study suggested that the function of astrocytes can be directly regulated by SSRIs.51 Monoamine oxidase (MAO) activates the metabolism of adrenaline and triggers calcium signaling in astrocytes,52 which suggests that antidepressants may directly affect astrocytes by preventing them from reabsorbing monoamines.
Serotonin (5-HT)
An essential neuromodulatory transmitter with specific neuroplastic properties is serotonin. Numerous investigations have demonstrated that 5-HT is intimately related to the pathophysiological process of major depression. The 5-HT hypothesis primarily asserts that a decrease in the 5-HT level is a risk factor for depression.53 In addition, low levels of 5-HT and L-tryptophan, which is a precursor of 5-HT,54 in blood platelets are also found in depressed people. Additionally, long-term treatment with fluoxetine, a typical SSRIs, reverses the stress-induced reduction in the quantity of astrocytic cells in the hippocampus in a tree shrew model of depression.55
5-HT receptors, which are mostly found on the bodies and dendrites of neurons, play a role in the pathogenesis of MDD.56 To date, 5-HT receptor subfamilies comprising 14 different receptor subunits expressed in various brain regions, namely, 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1E, 5-HT1F, 5-HT2A, 5-HT2B, 5-HT2C, 5-HT3, 5-HT4, 5-HT5A, 5-HT5B, 5-HT6 and 5-HT7, have been reported. Among these 5-HT receptor subtypes, the 5-HT1, 5-HT2, 5-HT6, and 5-HT7 subtypes are expressed on brain and spinal astrocytes in humans and rodents. Numerous 5-HT receptors expressed on astrocytes are G-coupled proteins that are associated with changes in the concentration of free cytosolic calcium ([Ca2+]i). These changes may trigger the release of a variety of astrocyte-derived signaling modulators, which may control neuronal activity.57 In astrocytes, 5-HT has a strong effect on the 5-HT2B receptor.58 5-HT receptors have been extensively studied to determine the pharmacological mechanism of antidepressants, and many novel pharmaceutical preparations are being investigated. For example, some novel antidepressants function as agonists of the 5-HT1A, 5-HT2B, or 5-HT4 receptor or antagonists of the 5-HT1B, 5-HT2A, 5-HT2C, 5-HT3, 5-HT6, or 5-HT7 receptor.59
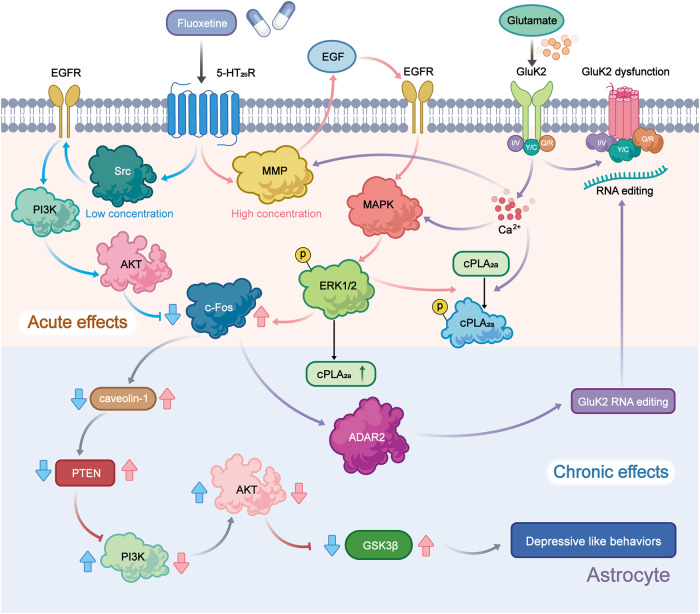
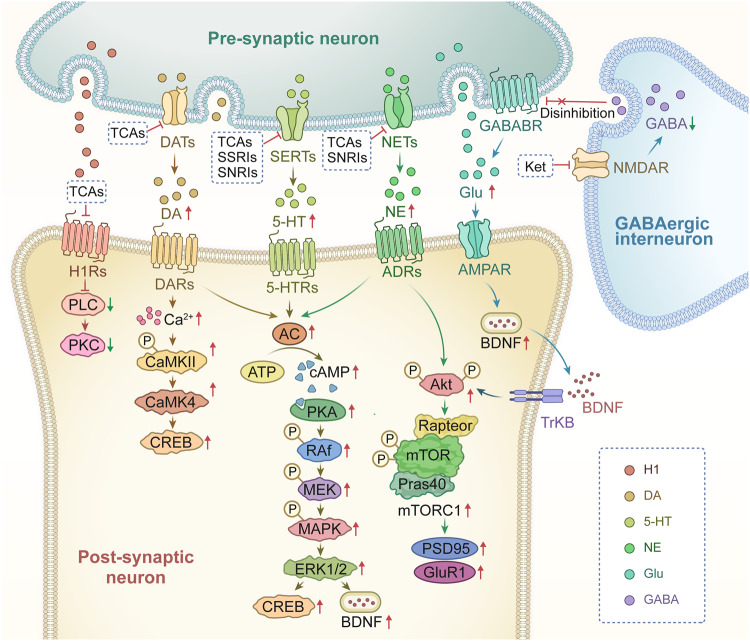
Administration of fluoxetine in different concentrations to astrocytes expressing the 5-HT2B receptor may activate distinct signaling pathways to control gene expression. Fluoxetine reduces the mRNA expression of c-Fos through the PI3K/AKT signaling pathway after acute application at concentrations below 1 μM, while the treatments with the higher doses (above 5 μM), it increases the gene expression of c-Fos via the MAPK/ERK signaling pathway in astrocytes.60 Then, in the nucleus, the altered transcription factor c-Fos can further biphasic change the expression of caveoline under the chronic treatments, thus the alteration levels of caveoline on cellular membrane can finally affect the downstream activation of PTEN/PI3K/AKT/GSK3β60. The GSK3β polymorphisms are associated with the high risk of MDD in Chinese Han Population.61 In our recent reports, the activation of GSK3β is also increased in the sorted astrocytes from the MDD-related stress-treated mice model and MDD clinic patients’ plasma.62 In addition, after fluoxetine-mediated stimulation of the 5-HT2B receptor in astrocytes, epidermal growth factor receptor (EGFR) is transactivated and subsequently activates the MAPK/ERK and PI3K/AKT signaling cascades, which control the expression of mRNA or proteins that may be linked to mood disorders, such as SERT. Ca2+-dependent phospholipase A2 (cPLA2), adenosine deaminase acting on RNA 2 (ADAR2), and kainate receptor subtype 2 (GluK2) are all involved in kainate receptor signaling.63,64 These discoveries promise astrocytic 5-HT2B receptors can be the potential pharmacological target of SSRIs (Fig. 2).
Fig. 2.
Schematic illustration of the pharmacological mechanism of fluoxetine in astrocytes. Acute treatment with fluoxetine at low concentrations (green arrows) stimulates Src, which phosphorylates EGF receptors by activating 5-HT2B receptors (5-HT2BR) and activates the PI3K/AKT signaling pathway. AKT phosphorylation induced by fluoxetine at low concentrations inhibits the expression of cFos and subsequently decreases the expression of caveolin-1 expression (chronic effects), which in turn decreases the membrane content of PTEN, induces phosphorylation and stimulation of PI3K and increases the phosphorylation of GSK3β, thus suppressing its activity. At higher concentrations, fluoxetine (red arrows) stimulates metalloproteinases (MMP) by activating 5-HT2BR and induces the release of growth factors, which stimulates EGF receptors and activates the mitogen-activated protein kinases (MAPK)/ERK1/2 signaling pathway. ERK1/2 phosphorylation induced by fluoxetine at high concentrations stimulates the expression of cFos and subsequently increases the expression of caveolin-1 (chronic effects), which inhibits PTEN/PI3K/AKT/GSK3β,60 ultimately leading to MDD like behavior. At high concentration, fluoxetine can also stimulate the activation of cPLA2a by the transactivation of EGFR/MAPK/ERK1/2 pathway, and the activated ERK1/2 can also increases the expression of cPLA2a at chronic treatments.61 In addition, the increased expression of cFos induced by fluoxetine can further increases the RNA editing of GluK2 by increasing the expression of ADAR2 at the chronic treatments, the function of the edited GluK2 by fluoxetine is down-regulated, which causes the acute glutamated induced Ca2+-dependent ERK phosphorylation is suppressed.63 Adobe Illustrator was used to generate this figure
Norepinephrine (NE)
NE released by the locus coeruleus (LC) can participate in regulating various neural functions, such as smell, movement, and sensation.65 It is significant to note that after being released, noradrenaline (NA) is not restricted to the area around the synaptic cleft and can reach nearby glial cells.66 Atomoxetine is a norepinephrine reuptake inhibitor (NRI) clinically used for the treatment of MDD. After systemic inflammatory attack with bacterial lipopolysaccharide (LPS), atomoxetine can decrease neuroinflammation in the rat cerebral cortex.67
The bioavailability of 5-HT and NE are increased by antidepressants called serotonin/norepinephrine reuptake inhibitors (SNRIs), which belong to antidepressants. Currently, new SNRIs, including duloxetine (DXT),68 desvenlafaxine (DVS),69 and venlafaxine,70 are widely used in MDD patients resistant to other treatments. Chronic treatment with DXT increases the expression of connexin 43 (Cx43), a crucial component of astrocyte gap junctions, in the rat PFC, preventing chronic unpredictable stress-induced dysfunction of astrocyte gap junctions and reversing the depressive-like behaviors caused by gap junction inhibition.71 A novel therapeutic target for MDD is transforming growth factor β1 (TGF-β1), the expression of which is controlled by antidepressants. Venlafaxine has also been found to exert neuroprotection by boosting the production of type 2 fibroblast growth factor (FGF-2) and transforming growth factor 1 TGF-β1 in astrocytes following stroke.72 However, the expression of protein markers of astrocytes and neurons is unaffected by DVS, and the chronic unpredictable mild stress (CUMS)-induced reduction in the levels of myelin- and oligodendrocyte-related proteins can be prevented by DVS.69 DVS may reduce oligodendrocyte dysfunction in the CUMS mouse model by altering cholesterol production and reducing depression-like phenotypes.69
Dopamine (DA)
There is increasing evidence that people with depression have reduced dopamine neurotransmission.73 Astrocytes in the lateral habeula are involved in regulating depressive-like behavior,74 whereas the reward circuit is mediated by the striatum.75 The dorsolateral part of the striatum is linked to the drug-seeking behavior and drug addiction associated with psychiatric disorders. As the major input to the basal ganglia, the striatum and related nuclei are linked to psychiatric morbidity, while the chronic stress reduces dopamine levels in areas such as the striatum and hippocampus.76 Due to processes involving dopamine D2 receptor signaling,77 the glutamine level increases in the presence of dopaminergic lesions and decreases in the presence of a high DA level.78 DA signaling is considered to play a key role in astrocyte-neuron crosstalk in the striatum.79 Sulpiride is an antidepressant that blocks the ability of the GLT-1 inhibitor TFB-TBOA to induce synaptic depression80 and partly attenuates the impact of fluorocitrate (a metabolic uncoupler that blocks aconitase in the tricarboxylic acid (TCA) cycle) on synaptic output. According to these results, astrocyte dysfunction results in an increase in DA levels, which decreases neuronal activity resulting from the binding of DA to dopamine D2 receptors,80 which generates neuronal depolarization, reducing DA selectivity at dopamine D1-like receptors and promoting DA inhibition through dopamine D2 receptors, which may contribute to increasing extracellular glutamate levels.81 An increase in DA signaling brought on by compromised astrocyte activity may induce a long-lasting change in striatal neurotransmission80 since DA signaling is crucial for both structural and synaptic plasticity.82
Glutamate
Glutamate is the main excitatory neurotransmitter in the central nervous system (CNS)83 and can be released by neurons through exocytosis, which in turn activates extracellular N-methyl-D-aspartate receptors (eNMDARs) in neurons, leading to synaptic loss.84 Exosynaptic glutamate also contributes to metabolism in neurons and astrocytes. When exosynaptic glutamate is taken up by astrocytes, it can become a substrate for glutamine synthesis or be metabolized by astrocytes and neurons.85 In addition, extracellular glutamate can also promote glucose uptake by astrocytes and inhibit glucose uptake by neurons. Therefore, glutamate is an important signal that mediates the interaction between central neurons and astrocytes, and its normal release and transport are the result of the functional cooperation between neurons and astrocytes. Glutamate homeostasis and neurotransmission play a major role in the onset of depression and anxiety. Studies have shown that glutamate levels in frontal cortex samples from autopsied patients with severe depression are increased, and antidepressants can restore normal glutamate levels.86 It has been observed in animal models that sustained glucocorticoid stimulation can increase the excitability of glutamatergic neurons and simultaneously decrease the number and plasticity of astrocytes, in addition to decreasing neuronal dendrite connectivity in the hippocampus and frontal cortex, leading to depression.87
It is well-documented that astrocytes have a wide range of modulatory functions that may either increase or decrease the release of many different neurotransmitters. Specifically, astrocytes are essential regulators of glutamatergic neurotransmission, and reuptake of glutamate by astrocytes regulates excitatory synaptic activity.85 When a large amount of glutamate is released from neuronal vesicles, glutamate clearance is mainly achieved by glutamate transporters (EAATs) on the membrane of astrocytes, which transport excess glutamate into astrocytes, where it is converted to glutamylamine through the action of glutamine synthase, reducing damage to neurons.88,89 In the classic glutamate-glutamine cycle, astrocytes and neurons convert glutamate to the nonexcitatory amino acid glutamine, which is then released back into the extracellular space and absorbed by neurons. Alterations in astrocytic glutamate clearance are known to occur in schizophrenia and other psychiatric illnesses, and mice with glutamate/aspartate transporter (GLAST) deletion show phenotypic abnormalities such as mental and behavioral deficits.90,91
Adenosine triphosphate (ATP)
Ectonucleotidases that are found in synapses can catabolize extracellular ATP to produce adenosine, and synapses also contain bidirectional nucleoside transporters that can release adenosine.92 Adenosine primarily stimulates inhibitory A1 and facilitatory adenosine receptors (A2AR) to play function.93 Notably, depressive behavior is linked to purinergic signaling. Depressive-like symptoms are exacerbated by activation of P2X7R in glial cells.94 Polimorphisms at P2X7R increase vulnerability to mood disorders whereas P2X2R-mediated neuronal activity is decreased in mice exposed to chronic stress due to insufficient ATP release from astrocytes.95 According to our earlier studies, chronic sleep deprivation (SD) can cause depressive-like behaviors by increasing extracellular ATP levels in vivo.41 Acting through P2X7R and FoxO3a cascade ATP inhibits expression of the 5-HT2B receptor, the decrease in extracellular ATP levels caused by chronic stress and an increase in ATP levels caused by SD are both linked to depressive-like behaviors.41 In detail, the elevated extracellular ATP induced by SD stress stimulates P2×7R and down-regulates the expression of 5-HT2BR by suppressing the activation of AKT, which inhibits the phosphorylation of FoxO3a and promotes its transportation into the nucleus, the reduced 5-HT2BR alleviates the inhibition of STAT3 to cPLA2, the activated cPLA2 further increases the release of AA and PGE2, these indicators have high relationship with the depressive-like behaviors, because in P2X7R knockout mice, the above changes of these indicators and behavioral performance are all eliminated.41 This increased activation of cPLA2 and the elevated levels of AA and PGE2 in astrocytes are supported by our discoveries in MDD patients’ plasma.62
After building a stress injury model in rats through maternal separation (MS), it is found that MS obviously reduces the total length of apical dendrites, however, the use of A2AR antagonists could prevent synaptic loss96 and reverse behavioral, electrophysiological, and morphological damage caused by MS,97 this is related to the activity reconstruction of the HPA axis. In another study, the abnormally increased A2AR in the lateral septum(LS) is a key factor in recurrent stress for leading to depressive-like behaviors. This function is mainly achieved by the increased activity of A2AR-positive neurons and the inhibited activity of ambient neurons, associating with the neural circuits of dorsomedial hypothalamus(DMH) and lateral habenular(LHb).98
Caffeine is an adenosine receptor antagonist, and epidemiological studies have shown that the intake of caffeine is closely related to the occurrence of suicide99 and depression.100 Since A2AR polymorphisms are associated with emotional problems, adenosine A2AR overexpression leads to emotional dysfunction, and A2AR blockade protects against the persistent emotional disturbance brought on by stress.101 Moreover, animal experiments have demonstrated that A2AR are upregulated in chronic stress animal models.102 Additionally, neuronal A1 receptors exhibit hypofunction caused by a decrease in astrocyte-derived adenosine levels;103 this decrease, as well as depressive-like behavior, can be reversed by certain antidepressants.104,105
HPA axis hypothesis
Stress and MDD are closely related, and stressful life events can often lead to depressive episodes. The activation of the HPA axis by stress can cause cognitive and emotional changes.106 An increase in HPA activity is one of the most common neurobiological alterations in depressed people. Studies have shown that the main factor contributing to the elevation of hypothalamic-pituitary activity is the increased production of corticotropin-releasing hormone (CRH). In addition, pituitary adrenal corticotropic hormone (ACTH) is released in response to CRH, which in turn triggers the adrenal cortex to release glucocorticoids (GCs).
Glucocorticoids
The HPA axis, a component of the neuroendocrine system, is commonly associated with the stress response. Hyperactivity of the HPA axis is thought to be an important pathophysiological mechanism underlying depression. High HPA activity is among the most typical neurobiological alterations in depressed individuals. The HPA axis is the primary stress response system that produces GCs, which are a class of steroid hormones. There is evidence that GCs, which are released in response to stress, are harmful to neurons in various brain regions. The hypothalamic paraventricular nucleus (PVN) rapidly secretes CRH and arginine vasopressin (AVP)107 when the HPA axis is activated by stress. The anterior pituitary is stimulated by CRH and AVP to produce ACTH, which in turn increases the release of GCs into the bloodstream.108
The GC and mineralocorticoid (MC) receptors GR and MR are members of the nuclear receptor (NR) superfamily. Both NRs can be triggered by binding to either MCs (such as aldosterone) or GCs (such as cortisol). However, the affinity of MR for its ligands is 10 times higher than that of GR for its ligands.109,110 GRs are expressesd at higher levels and particularly concentrated in the pituitary and hypothalamus, as well as a variety of regions of the limbic system (including the amygdala, hippocampus, and PFC), which are important for cognitive and psychological functions.
To prevent loss of control over the HPA axis, GCs exert negative feedback on the axis in all regions involved (the limbic system, hypothalamus, and pituitary). Some data suggest that HPA axis imbalance and high levels of GCs play a core role in the pathogenesis of MDD and suggest that GR may serve as an important target for treating depression.111
Thyroid hormone
Thyroxine (T4) and triiodothyronine (T3) are the two primary Thyroid hormones (THs) that regulate metabolism, protein synthesis, the growth of bones, and nervous system development. Thyrotropin-releasing hormone (TRH), which regulates the synthesis of thyroid-stimulating hormone (TSH) by the anterior pituitary gland, is mostly produced by neurons in the PVN. TSH stimulates the thyroid gland to produce T3 and T4. The levels of serum-free T4 and free T3 are regulated by negative feedback from pituitary TSH release. Tissue deiodinase mostly transforms T4 into the less physiologically active metabolite reverse T3 and the more biologically active metabolite T4.112
Overactivity of the HPA axis may be caused by damaged astrocytes and aberrant GR function. The HPA and hypothalamic-pituitary-thyroid (HPT) axes are inextricably linked. The most important related finding is that cortisol directly affects TRH secretion (which regulates TSH release), potentially through the response of GCs to TRH mRNA expression in neurons. According to research, hypercortisolemia may result in a reduction in TRH mRNA levels in the mid-caudal PVN.113 TRH expression in the PVN is lower in nonpsychiatric patients treated with corticosteroids, and the mRNA levels of TRH are lower in the PVN of depressed patients who have recurrent suicidal thoughts. This suggests that the effect of hypothalamic TRH is weaker in these individuals.
THs are required for neuronal growth and function not only in the periphery but also in the CNS,114 where they promote the formation of microglia, astrocytes, including radial glial cells, and oligodendrocytes. The role of THs in glial cells is becoming clear because of new discoveries in the field of glial cell biology. THs affect the shape and proliferation of astrocytes, as well as the organization and expression of GFAP/vimentin, and boost GS activity.115 T3 has an effect on glial morphology and hence on glial function in the adult brain; therefore, it also has an effect on neuron-glia interactions.115,116 It has been shown that T3 induces astrocyte proliferation by autocrine production of growth factors such as epidermal growth factor (EGF) and FGF-2. Apart from their proliferation-promoting impact, these growth factors increase and modify the pattern of deposition of the extracellular matrix components laminin and fibronectin, therefore boosting cell adherence and attachment to the substratum. Together with the discovery that animals with hypothyroidism and mice with TH receptor mutations display significant defects in glial development, these findings indicate that astrocytes are TH targets and that TH can protect neurons and astrocytes from glutamate toxicity.115
Estrogens
The hippocampus is closely related to memory and learning, and estrogen plays an important role in these processes. Estrogen increases the proliferation, migration, and differentiation of neurons in the dentate gyrus to maintain hippocampal function and is also important for controlling the HPA axis.117
Estrone (E1), estradiol (E2), and estriol (E3) are the three physiological estrogens; among these estrogens, E2 is the most active, and its level quickly decreases throughout menopause.118 E2 has been demonstrated in numerous studies to alter systems involved in the pathophysiology of depression, including the serotonin and norepinephrine systems, and to considerably alleviate depressive symptoms in animal models. Estrogen therapy can decrease the quantity of 5-HT1 and β-adrenergic receptors while increasing the quantity of 5-HT1 receptors.119 In addition, estradiol may influence the pathogenesis of male MDD patients.120 In animal models, E2 has been shown to alleviate depressive-like behavior.121,122 Estrogen receptor 1 (ER1) and estrogen receptor 2 (ER2) are transcription factors that are members of the NR family. Activating ER2 with a range of ER2 agonists has been reported to reduce stress-induced HPA activity and anxiety-like behaviors.123,124
Astrocytes are estrogen targets,125 as both ER1 and ER2 receptors are present on the astrocyte membrane or intracellularly in astrocytes. The transmembrane receptors ER and GPR30 have been shown to facilitate nongenomic and fast estrogen signaling in astrocytes, contributing to the neuroprotective effects of E2. In mature astrocytes differentiated from human induced pluripotent stem cells (iPSC)-derived astrocyte progenitors, ketamine can exert rapid antidepressant effects through the activation of amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) glutamate receptors, and estrogen enhances this effect of ketamine by increasing the gene expression of AMPA receptor subunits.126
Leptin
The obese gene (OB) encodes the hormone leptin, which is derived from adipocytes and the stomach and exerts its function through a specific receptor (OB-R). Leptin controls the function of the HPA axis127 via its receptor in the hypothalamus. The cerebral cortex, hippocampus, hypothalamus, dorsal raphe (DR) nucleus, arcuate nucleus, and solitary tract nucleus are some regions of the brain that can express leptin receptors. Increasing experimental data have recently shown that leptin is linked to the pathological and physiological processes of numerous mental illnesses and plays a vital regulatory role in the CNS.128,129 According to our previous reports, leptin can enhance the pharmacological effects of fluoxetine in astrocytes sorting from GFAP-GFP transgenic mice.130 Leptin selectively increases the expression of the astrocytic 5-HT2B receptor by activating the JAK2/STAT3 pathway, and fluoxetine in turn stimulates the 5-HT2B receptor and increases the secretion of brain-derived neurotrophic factor (BDNF) from astrocytes in vivo, thus ameliorating depressive-like behaviors.130 All of these findings indicate leptin’s potential to boost protein expression and functionally stimulate SERT.
Cytokine hypothesis
MDD is accompanied by changes in the levels of proinflammatory cytokines and trophic factors, including BDNF, interleukins (IL-1β, IL-6), and tumor necrosis factor alpha (TNF-α). Increasing data suggest that the production of certain cytokines by brain astrocytes plays a significant role in the pathogenesis of MDD.
Oxidative stress
Oxidative stress (OS), which is caused by an imbalance between antioxidants and reactive oxygen species (ROS), can harm proteins, lipids, or DNA. The activity of monoamine oxidase, the enzymes that break down monoamines such as DA, 5-HT and NE, is influenced by ROS and in turn can increase ROS production in mitochondria. The brain is more vulnerable to OS than other organs. In depression, OS plays a crucial role.131,132 The brain is particularly sensitive to OS due to numerous variables, including rapid oxidative energy metabolism (a process through which ROS, which are harmful molecules, are constantly produced), high levels of unsaturated fatty acids (which are vulnerable to lipid peroxidation), and relatively low intrinsic antioxidant capability.133 Adults with MDD exhibit ROS-mediated reductions in nitric oxide (NO)-dependent dilation.134
Thioredoxin reductase, heme-oxygenase 1, glutathione, and glutathione peroxidase are only a few of the ROS-detoxifying enzymes that are abundant in astrocytes.135 Astrocytes are the major producers of glutathione in the brain because they express a system xc-cyttine/glutamate antiporter, which does not exist in neurons; hence, neurons cannot synthesize glutathione. Notably, astrocytes can protect nearby neurons against toxic dosages of NO, H2O2, and superoxide anion in combination with NO, iron, or 6-hydroxydopamine in coculture systems,135 indicating that neurons rely on the strong antioxidant capacity of astrocytes for protection against OS. Nuclear factor erythroid 2 (Nrf2), a redox-sensitive transcription factor required for coordinating the cellular antioxidant response, can be activated by astrocytes. In our recent study, lithium salt (Li+) was found to effectively alleviate ischemia-induced anhedonia in mice by suppressing the production of mitochondrial ROS in glial cells.136
Recent investigations have indicated that MDD is caused by increased ROS production and promotes inflammation.137 The brain has weak antioxidative defenses and a high oxygen consumption rate, making it particularly susceptible to OS. Inflammasomes in microglia can be activated by ROS, which causes inflammatory cytokines, including TNF-α, IL-1β, and IFN-γ, to be produced.138 Neuroendocrine-immune activities can be compromised by inflammation, which can also result in numerous disorders, such as MDD. Proinflammatory cytokines have become pathological indicators of MDD, and using the right antioxidants to combat ROS may be a useful method for treating MDD.
Proinflammatory cytokines
Higher levels of inflammation increase the chance of developing new-onset depression.138,139 Although depression can cause inflammation, its cause is still unclear and may be influenced and regulated by immune cells, inflammatory cytokines, and the nervous system. In addition to contributing to the etiology of depression, activation of proinflammatory signaling pathways occurs as a result of elevated OS.140 Evidence suggests that MDD is associated with the immune response, as shown by increased levels of IL-1β, TNF-α, and IL-6.141 LPS-induced astrocyte activation also contributes to the symptoms of MDD. Systemic treatment with LPS induces depressive-like behaviors and increases the production of inducible nitric oxide synthase (iNOS), IL-1β, TNF-α, and GFAP in the hippocampus and cortex. Inhibition of activated astrocytes reduces neuroinflammation. These alterations are followed by amelioration of LPS-induced depressive-like behaviors.142
Neurotrophic factors
In the vast majority of patients with severe depression, antidepressants affect the levels of neurotrophic factors. For example, the primary regultaory factor of neuronal survival, growth, and differentiation during development is BDNF. For the treatment of depression, targeting signaling transduction by BDNF and its receptor, tropomycin receptor kinase B (TrkB), is essential.143,144 Recent research has shown a link between decreased hippocampal neurogenesis and low levels of BDNF and glial-derived neurotrophic factor (GDNF) in the brains of depressed individuals.145 Under normal conditions, astrocytes release various nutrients and cytokines. After cell reactivation, the secretion of these factors is further increased.146 According to previous studies, fluoxetine stimulates c-Fos expression and ERK1/2 phosphorylation, which in turn promotes BDNF production in astrocytes sorting from GFAP-GFP transgenic mice.147 Imipramine acts as an antidepressant by increasing the mRNA expression of BDNF in astrocytes. Fluoxetine also induces BDNF expression by activating cAMP-response element binding protein(CREB) through the PKA and/or ERK pathways.148
BDNF is an essential molecule for neural plasticity and development and is related to several CNS diseases. Currently, it is known that BDNF can regulate the activity of neurons and that it is produced not only by neurons but also by astrocytes.149 SSRIs and tricyclic antidepressants increase BDNF expression in cultured primary astrocytes, and BDNF overexpression in mouse hippocampal astrocytes is sufficient to promote neurogenesis and causes anxiolytic behavior.149 By promoting neurotransmitter release, facilitating vesicle docking, and upregulating the expression of synaptic vesicle proteins, BDNF, which is released by astrocytes in response to long-term antidepressant therapy, may assist in increasing synaptic plasticity at presynaptic terminals.150 In addition, astrocyte-secreted BDNF can stimulate adult hippocampal neurogenesis and may contribute to synaptic and structural plasticity that underlies the long-lasting behavioral effects of antidepressants.150 Astrocytes can secrete numerous nerve growth factors. Vascular endothelial growth factor (VEGF) is a member of the vasoactive growth factor family. It exerts its unique molecular effects by binding and activating endothelial cell tyrosine kinase receptors. VEGF is traditionally associated with angiogenesis and its stimulation. Recent evidence indicates, however, that it also influences nerve cells and plays a crucial role in hippocampal neurogenesis and neuroprotection.151
Inflammasomes
Neuroinflammation is a central pathophysiological mechanism and defining characteristic of MDD. Numerous elements in the periphery and CNS interact to generate neuroinflammation, thereby stimulating astrocytes. The nucleotide-binding domain and leucine-rich repeat protein-3 (NLRP3) inflammasome is one of the largest typical inflammasomes discovered thus far. It is composed of pro-Casp-1 protein, NLRP3, and apoptosis-associated speck-like protein (ASC).152 The sensitization of the NLRP3 inflammasome and the suppression of BDNF synthesis result in MDD.153 In our research, SD is found to reduce BDNF levels and induce depressive-like behaviors in the sorted astrocytes from GFAP-GFP transgenic mice by activating the NLRP3 inflammasome.130 NLRP3 inflammasome activation causes astrocytes to produce more IL-1β and IL-18.154,155
The release of proinflammatory cytokines is the primary consequence of the activation of caspase-1, a component of the NLRP3 inflammasome. In addition, it has been observed that stimulating NLRP3 inflammasome assembly can induce depression-like behaviors in rodents exposed to LPS or CUMS.156,157 Research on the effect of astrocyte-specific NLRP3 knockout suggests that the astrocytic NLRP3 inflammasome exerts a significant effect on astrocytic pyroptosis via the Casp-1/GSDMD pathway in depression.156,157 Therefore, efficient NLRP3 inflammasome inhibitors are novel therapeutic agents for MDD. As we previously reported, chronic SD can specifically activate the NLRP3 inflammasome and decrease the level of BDNF in astrocytes to ameliorate depressive-like behaviors. Fluoxetine can suppress the effects of SD on astrocytes by stimulating astrocytic 5-HT2B receptors directly.147 Additionally, in the middle cerebral artery occlusion (MCAO) stroke model of mice, Li+ can significantly attenuate GSDMD-mediated glial pyroptosis by regulating the AKT/GSK3β/TCF4/β-catenin signaling pathway, in which, the activation of AKT induced by Li+ can also increase the phosphorylation of FoxO3a and promote the transportation of FoxO3a from nucleus into cytoplasm, the reduced FoxO3a in nucleus dissolves its competition with TCF4 in order to confirm more β-catenin/TCF4 complex. The increased latter complex further up-regulates the expression and activation of STAT3 in nucleus, the latter further inhibits the activation of the NLRP3 inflammasome by increase UCP2 which can decrease the production of ROS from mitochondrion.136 This neuroprotective mechanism of Li+ after ischemia-reperfusion injuries contributes to the improved depressive-like behaviors, besides of motor and cognitive capacities.136
In conclusion, there have been so many hypothesis to explain the pathogenesis of MDD associating with many booming researches (Fig. 3). However, it is still hard to adopt only one above hypothesis to completely reveal pathophysiology of MDD. The main problem may contribute to the limitations of the theoretical perspective and the limitations of detection methods. Some key scientific problems in the neurobiology of neurological and psychiatric disorders are still unclear, such as how to identify the pathological characteristic changes for mood disorders, how to metabolize the cerebral metabolic waste under the pathological condition,how to observe the instant interactions of neural cells and the real-time changes of intracellular organelles in the patients of MDD? In the pathological conditions, conducting research from the perspective of comprehensive collaboration of the whole body and increasing the proportion of new technological applications in research will open up the new paths to reveal the pathogenesis of MDD in the future.
Fig. 3.
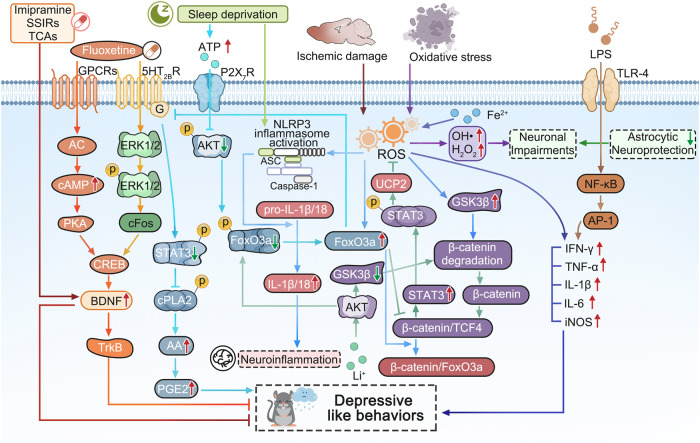
The molecular signaling schematic of cytokine hypothesis in the pathogenesis of MDD. The rodent performed the depressive like behaviors are impaired by some widely accepted risk factors, such as long-term sleep deprivation (SD), oxidative stress, lipopolysaccharide (LPS), ischemic damage and so on. Long-term SD can increase the extracellular ATP level, the latter inhibits the activation of AKT and the followed phosphorylation of FoxO3a by stimulating P2X7 receptors (P2X7R), the dephosphorylated FoxO3a translocates into the astrocytic nucleus, then the increased FoxO3a decreases the expression of 5-HT2BR expression, which results the reduced phosphorylation of STAT3 which increases the activation of cPLA2 and the followed release of arachidonic acid (AA) and prostaglandin E2 (PGE2), finally causing the depressive-like behaviors.41 Thus, antidepressant fluoxetine activates ERK1/2/cFos pathway by stimulating 5-HT2BR and AC/cAMP/PKA pathway by activating GPCRs in order to increase the activation of CREB and the level of BDNF and TrkB, which can alleviate the depressive like behaviors induced by long-term SD.147,148 As well as, imipramine, other SSIRs, and TCAs can also play antidepressive roles by increasing BDNF mRNA expression in astrocytes.148 Ischemic stroke can trigger the increase of reactive oxygen species (ROS) which can induce the activation of NLRP3 inflammasome and the release of IL-1β/18, resulting in the neuroinflammation, however, Li+ salt inhibits the activation of GSK3β and increases the phosphorylation of FoxO3a by activating AKT, which promotes the more FoxO3a transportation from nucleus into cytoplasm, and the reduced FoxO3a in nucleus lacks the competition with TCF4, the increased complex level of β-catenin and TCF4 further stimulates the expression and the phosphorylation of STAT3, which further induce the mRNA and protein expression of UCP2, then in mitochondrion, the increased UCP2 suppresses the production of ROS and results in the deactivation of NLRP3 inflammasomeincreases.136 Superoxidation of Fe2+ stimulates an increase in ROS, resulting in the production of inflammatory cytokines (including IFN-γ, TNF-α, IL-1β, IL-6) and inducible nitric oxide synthase (iNOS).138 While, the treatments of oxidative stress (OS) can produce a large number of ROS, such as OH• and H2O2, resulting in neuronal impairments, while astrocytes can play their neuroprotective role by antioxidation.135 Additionally, LPS can also increase TNF-α, IL-1β, and IL-6 by TLR-4/NFkB/AP-1 pathway and cause depressive-like behavior.142 Adobe Illustrator was used to generate this figure
Interactions of multi-cells and multi-organs
Recently, increasing evidence has shown that pathological changes in a single cell type or brain region limited are insufficient explain the pathogenesis of MDD. This section mainly introduces the latest research on the pathogenesis of MDD, discussing the multiple interactions among neural cells and the multiple regulatory mechanisms between the brain and peripheral organs in detail.
The interaction between neuron and glial cell
Over the past few decades, studies on MDD have identified decreased PFC activity and excitatory/inhibitory (E/I) imbalance as probable mechanisms underlying depression.158 Astrocytes are recognized to be essential for controlling neural network activity and to take part in higher brain activities.159 To explore efficient treatments for MDD, it is important to focus on how to regulate the E/I balance and neuronal remodeling.160
MDD-related marker proteins in neural cells
Astrocytes in the CNS form the neurovascular unit with neurons and blood vessels. The neurovascular unit mediates the exchange of nutrients and other functional substances between its components.161 The blood-brain barrier (BBB) consists of endothelial cells tight junctions, a continuous basement membrane and astrocytic end-feet. Two proteins expressed on astrocytes, connexin 30 (Cx30) and Cx43, have been linked to the pathogenesis of depression.162 Gap junctions that enable communication between astrocytes are formed by the membrane proteins Cx30 and Cx43.163 Chronic unpredictable stress (CUS) and acute stress both specifically reduce the expression of the gap junction-forming proteins Cx30 and Cx43,164 and the integrity of the BBB is weakened in mice lacking Cx30 and Cx43.165
In addition to being an essential component of the developing astrocyte cytoskeleton, GFAP serves as the main intermediate filament protein in adult astrocytes. Although increased expression of GFAP is commonly observed in reactive astrogliosis, postmortem results suggest that the frequency and intensity of reactive astrogliosis are decreased in the brains of patients with MDD.166 Accompanied by a decreased astrocyte density, the levels of GFAP and the GFAP intermediate filament domain are also reduced in brain samples from patients with MDD.167 Researchers have even proposed that the GFAP content in serum can be used to determine the severity of MDD,168 but this point is controversial.
AQP4, a kind of water channel, is mainly expressed on astrocytic end-feet in contact with blood vessels. The water channel AQP4 regulates the equilibrium of ions and water in the brain and is an essential part of the neurovascular unit. The vascular coverage of AQP4-immunopositive astrocytes in the orbitofrontal cortex (OFC) is lower in people with clinically significant depression than in psychiatrically healthy control patients.169 In another postmortem study, it was found that the coverage of blood vessels by AQP4-positive astrocyte terminals was reduced in the OFC of MDD patients.170 In addition, the K+-buffering capacity and presumably synaptic transmission are impaired in mice lacking AQP4, and impairment of these processes is associated with depressive-like behaviors.171 In our previous study, we reported that the expression of AQP4 was decreased by exposure to CUMS, which contributed to dysfunction of glymphatic circulation and depressive-like behaviors in mice.172 Additionally, the coverage of blood vessels by AQP4-positive astrocytic endfeet is decreased by 50% in MDD patients, indicating that decreased levels or mislocalization of AQP4 may contribute to the pathogenesis of MDD.169,173
S100B is produced and secreted by astrocytes in the gray matter,174 and changes in the levels of S100B in the blood and cerebrospinal fluid (CSF) of patients with MDD can cause glial cell dysfunction and damage.175,176 In individuals with MDD, the number of S100B-immunopositive astrocytes in the pyramidal layer of the bilateral hippocampal CA1 region is decreased.177 S100B secreted by damaged astrocytes can enter the extracellular space and CSF,178 and the level of S100B is increased in the dorsolateral prefrontal cortex (dlPFC) of patients with MDD.179 S100B levels are elevated in the CSF or serum of patients with MDD,180 which suggests that S100B is a potential diagnostic biomarker for depressive episodes associated with MDD.
Communication between neurons and microglia plays an important role in the pathogenesis of depression. C-X3-C Motif Chemokine Ligand 1 (CX3CL1)- C-X3-C Motif Chemokine Ligand 1 receptor (CX3CR1) and OX-2 membrane glycoprotein (CD200)-OX-2 membrane glycoprotein receptor (CD200R) form ligand-receptor pairs, and these molecules are the most important chemokines and clusters of differentiation in maintaining CNS homeostasis.181 CX3CL1 and CD200 are mainly expressed in neurons, and their receptors CX3CR1 and CD200R are expressed on microglia.182 Activated microglia and decreased expression of CX3CL1 in the hippocampus were observed in an LPS-induced depression model.183 CX3CR1-deficient mice show a temporary decrease in the number of microglia and a resulting deficiency of synaptic pruning, which may be related to neurodevelopmental and neuropsychiatric disorders.184 However, CX3CR1-deficient mice show significant resistance to stress-induced depressive-like behaviors.185 The level of CX3CL1 in the serum is increased in patients with moderate-severe depression compared with healthy subjects; thus, CX3CL1 could be used as a target for depression treatment.186 Patients diagnosed with MDD with comorbid cocaine addiction show higher serum levels of CX3CL1.187 Additionally, in a rat early-life social isolation (ESI) model, the expression of CD200 receptors in microglia is significantly reduced.188 Exposure to unavoidable tail shock causes a decrease in CD200R expression in the hippocampus and amygdala,189 and stress was also discovered to suppress CD200R expression in the hippocampus of rats.190
Synaptic plasticity
Long-term potentiation (LTP) serves as the physiological basis for learning and conditioned responses.191 Ketamine has a quick antidepressant effect, as it is a noncompetitive channel blocker of N-methyl-D-aspartate receptors (NMDARs).192 Excessive glutamate in the synaptic cleft activates synaptic metabotropic glutamate receptors (mGluRs), which lead to neural excitotoxicity.193 In a mouse model of chronic social defeat stress (CSDS), which causes depression, mGluR5 was shown to induce long-term depression (LTD). The major process responsible for synaptic plasticity is the mGluR-mediated LTD, which likely plays a significant role in the pathophysiological changes underlying depressive-like behaviors in the CSDS-induced depression paradigm.194
ATP can mediate the activity of the astrocyte-neuron network, and ATP is a signaling molecule that also controls synaptic plasticity.195 ATP can increase the expression of amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors (AMPARs) by stimulating P2X7R and increasing the amplitude of miniature excitatory postsynaptic currents.196 Stress exposure is a major pathogenic factor in disease models and can increase Ca2+-dependent release of ATP from neurons, which causes excitotoxicity.197,198
Regulated in development and DNA damage response-1 (REDD1) is a stress response gene that can regulate development and the response to DNA damage. Virus-mediated overexpression of REDD1 in the rat PFC is sufficient to cause anxiety- and depressive-like behaviors and neuronal atrophy.199 According to postmortem studies, the volume of the dlPFC is smaller and the density of neurons in the dlPFC is lower in MDD.200 BDNF can modulate synaptic plasticity in the brain. TrkB is a functional receptor of BDNF.201 BDNF produces antidepressant-like effects by increasing synaptic plasticity in a mouse model of CUMS.202
Neuron-glia integrity
The term “tripartite synapse” was initially used to describe the intimate relationship between astrocytes and neurons at glutamatergic synapses, similar to the glutamate-glutamine cycle described above.203 Moreover, glutamic acid decarboxylase, an enzyme that transforms glutamate into γ-aminobutyric acid (GABA), also exists in inhibitory GABAergic neurons. Increased inhibitory neurotransmission, glutamatergic/GABAergic E/I imbalance, and chronic stress-related emotional dysfunction reduce PFC activity.204,205 In local circuits, various glutamatergic and GABAergic neurons interact in complicated ways to achieve E/I balance.206 A meta-regression analysis indicated that glutamine and glutamate levels are decreased in the PFC, which is correlated with the therapies to MDD.207 Global topological E/I imbalance in MDD is discovered through gene and protein expression of molecules related to inhibitory GABAergic and excitatory glutamatergic signaling in the postmortem MDD brains.22,208,209 It shows the imbalance in cortical-subcortical limbic regions with decreased GABAergic signaling and increased glutamatergic signaling.210,211 Meanwhile, GABAergic signaling is decreased in regions comprising the default mode network (DMN), while it is increased in the lateral prefrontal cortex (LPFC).212,213 Stimulating P2X7R in neocortical nerve terminals can block the reuptake of GABA and glutamate by the presynaptic membrane and promote the release of these two neurotransmitters in the cerebral cortex of rats and humans,214,215 and activation of P2X7R reduces the expression of GLAST.216 This results in neuronal damage, a reduced number of synapses, decreased neurogenesis, and even impairment of key cerebral circuits that regulate mood.
Astrocytes are fundamental elements in synapses, participate in synaptogenesis and maturation, and maintain synaptic homeostasis. Ionic homeostasis in the extracellular space is critical for central nervous system function.217 Astrocytes play an important role in maintaining extracellular K+ homeostasis in the CNS, as well as H+, Cl-, and Ca2+ homeostassis.218 In addition, it also plays an important role in maintaining transmitter homeostasis, in which glutamate and GABA play particularly important roles.219
In addition to the tripartite synapse, the more recent concepts of the four-part extracellular matrix and the microglial five-part synapse220 also support the idea that glial dysfunction plays key roles in the early pathological features common to psychiatric disorders.221,222 Under physiological conditions, microglia can play a neuroprotective role by producing cytokines. However, under pathological conditions, microglia can also affect the balance between excitatory and inhibitory synapses by phagocytosing synapses223 and activating inflammatory factors in microglia.224 In addition, the extracellular matrix (ECM) plays a significant role in maintaining normal communication in mature neural networks, which can limit the synaptic restriction of glutamate.225 The components of the ECM are mainly produced by neurons and astrocytes, and microglia can also regulate the remodeling of the ECM.226
Interaction mechanism in multi-organs
Abnormalities in cytokine levels in the brain and peripheral organs, disruption of the brain/immune system balance, and dysfunction of communication between the peripheral organs and the brain can cause neuroinflammation and depressive symptoms. For instance, cirrhosis and depression have been linked to intestinal dysbiosis, which results in intestinal barrier disruption, increasing bacterial translocation. Increased bacterial translocation then activates circulating immune cells, which produce cytokines and induce systemic inflammation.227 In comparison with the healthy population, MDD patients have a much higher incidence and prevalence of chronic liver disease.228 Inflammatory bowel disease (IBD) and irritable bowel syndrome (IBS) with increased intestinal permeability, which may have both inflammatory and autoimmune sources, are common comorbidities of MDD and anxiety.229,230
Neuroendocrine-immune axis
Microglia secrete chemokines that disrupt the integrity of the BBB and increase the ability of immune cells to enter the brain parenchyma.231 The stress response is a complex array of behavioral, neuroendocrine, autonomic, and immunological responses that enable adaptation to unpleasant psychological and physiological stimuli.232 The HPA axis is a crucial endocrine system that orchestrates this response.233 Stress can activate microglia, which are considered important immunocytes of the CNS. Mediators released by activated microglia can stimulate the HPA axis and induce GC production.39 Similarly, high levels of GCs can also activate microglia, creating a vicious cycle.234
Tryptophan (TRP) can be converted into a variety of biologically active molecules, and more than 95% of TRP is metabolized to kynurenine (KYN) and its breakdown products, with only a small portion of TRP being converted to 5-HT.235 Indoleamine 2,3 dioxygenase (IDO) is an immune inducible enzyme that metabolizes TRP through the KYN pathway and plays an important role in the immune response.236 In the brain, KYN is metabolized to the neurotoxic substance quinolinic acid (QUIN).237
The primary GC in the HPA axis, corticosterone, plays a role in regulating the stress response in rodents. Stress, high GC levels, and serious depression are all linked. Analysis of transcriptomic changes associated with corticosterone-induced cytotoxicity revealed an association of neurite outgrowth-related genes with depression. Therapies for MDD may target the expression of genes involved in neurite formation, such as calpain 2 (Capn2), vesicle-associated membrane protein (Vamp7), and c-type natriuretic peptide (Cnp).238
Consumption of a high-fat diet (HFD; for approximately 16 weeks) results in anxiety and anhedonic behaviors, and 4 months of HFD consumption results in increased levels of corticosterone and blood glucose, which also activate the innate immune system, increasing the release of inflammatory cytokines (i.e., IL-6, IL-1β, TNF-α). The behavioral abnormalities that arise from long-term consumption of a HFD are quickly reversed by ketamine. Additionally, giving HFD-fed rats a P2X7R antagonist greatly alleviates their anxiety.239
Microbiota-gut–brain axis
In recent years, the microbiota-gut-brain axis has been reported to be disrupted in MDD. Stress stimulation can affect the gut microbiota, which in turn induces the production of inflammatory mediators (mainly IL-6 and IFN-γ) and a reduction in short-chain fatty acid levels.240 The increased level of inflammatory cytokines may be caused by disturbance of the gut microbiota, which may also disrupt the gut barrier.241 Alterations in the gut microbiota and inflammatory agents have an impact on the KYN pathway, metabolism, and toxin metabolism in the periphery.242 Proinflammatory cytokines or toxic byproducts resulting from microbiota alterations may pass through the BBB and enter the brain.243 This increases the levels of cytokines such as IL-1β and IL-6 and NLRP3 inflammasome activation in brain-resident cells.244 In particular, microglia and astrocytes are activated and undergo atrophy, respectively. These glial cell changes, which affect the brain networks involved in learning and memory, mood regulation, and emotional regulation, may cause depressive symptoms or anxiety episodes.245
According to clinical research, TRP and tryptophan catabolites (TRYCATs) may play a crucial role in psychiatric illnesses, including MDD. Peripheral and central inflammation can both stimulate the KYN pathway and trigger TRP metabolism and subsequent synthesis of various TRYCATs, including the toxic NMDAR activator QUIN,246 which influences glutamate transmission, has a variety of immunomodulatory effects and has both neurotoxic and neuroprotective effects on the CNS.141 Studies have proven that peripherally injected LPS increases the central and peripheral metabolism of TRP via the KYN pathway by exerting neurotoxic effects, inducing reactivation of microglia and astrocytes in the CNS.247 Excessive production of QUIN, an NMDAR agonist, stimulates the release of glutamate and inhibits reuptake, leading to neuronal excitotoxicity.248
Liver-brain axis
Patients with liver diseases often struggle with depression. According to one study on the frequency of liver disease and major depression in the United States, liver disease is linked to both major depression and suicidal thoughts.249 A further population-based cohort study discovered that patients with MDD had much higher prevalence and incidence rates of chronic liver disease than the general population.228 The incidence of depression is high in cirrhosis patients; moreover, depression is an independent predictor of mortality from cirrhosis.250
An internal metabolic mechanism regulated by the liver can control depressive-like behavior. A crucial enzyme in epoxyeicosatrienoic acid (EET) signaling in the liver is epoxide hydrolase (sEH). Chronic stress selectively exacerbates sEH-induced depression-related changes in the liver while dramatically lowering the plasma levels of 14,15-EET. Deletion of hepatic epoxide hydrolase 2 (Ephx2) (which encodes sEH) rescues the chronic mild stress (CMS)-induced decrease in 14,15-EET plasma levels.251 In a rat model of CUMS, electroacupuncture (EA) was found to downregulate P2X7R, NLRP3, and IL-1β expression in the prefrontal cortex and liver and relieved depression-like behavior.252
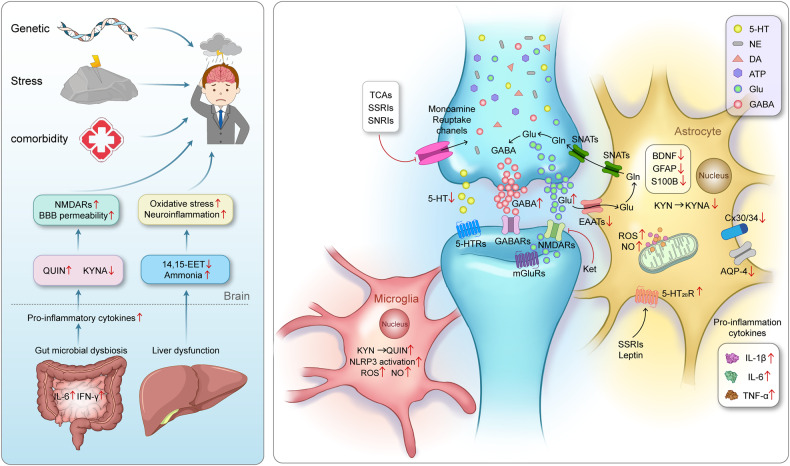
In summary, as shown in Fig. 4, although the etiology of MDD is still unclear, it is widely accepted that the common pathogenic factors of MDD are genetic, stress, and comorbidity.3 The levels of monoamine neurotransmitters (5-HT, NE, and DA) are insufficient in the synaptic cleft of MDD patients, correspondingly, the explored antidepressants such as tricyclic antidepressants(TCAs), SSRIs and SNRIs almostly act on the channels responsible for inhibiting reuptake of these neurotransmitters.51 Thus, according to these traditional pharmacological theories, these antidepressants always have the delayed clinical efficacy, this promises the potential new pharmacological mechanism still requires further study. As the well-known glutamate-glutamine cycle, astrocytes play key roles in resolving neuronal glutamate toxicity. However, under the MDD pathological condition, due to the decreased expression of EAATs in astrocytes, excessive glutamate in the synaptic cleft activates synaptic mGluRs, which leads to neuronal excitotoxicity.194 In addition, the overdose glutamate can also be decarboxylated by glutamate decarboxylase (GAD) to GABA and activates the GABA receptors on the postsynaptic membranes.206 In our previous studies, the expression of 5-HT2B is selectively decreased in the sorting astrocytes from MDD model mice.64 The antidepressants SSRIs and leptin can increase the expression of the astrocytic 5-HT2B receptor.147 Furthermore, OS plays a crucial role in the emergence of depression, including by elevating the levels of ROS and NO in the mitochondrion of astrocytes.253 Proinflammatory signaling pathways are activated as a result of elevated OS, the mitochondrial dysfunction results in an increased generation of ROS and NO.137 As well as, the pathogenesis of MDD are associated with the inflammatory-immune response, as shown by elevated levels of proinflammatory cytokines, mainly IL-1β, TNF-α, and IL-6.141 The expression of neural cell marker proteins in neural cells, including Cx30/43,162 GFAP,167 AQP4,172 and S100B,177 are all decreased under MDD pathological conditions. In brain, KYN is metabolized by microglia to the neurotoxic metabolite QUIN and by astrocytes to the beneficial metabolite kynurenic acid (KynA), thus, QUIN is increased and KynA is decreased in MDD patients’ brain.141,254,255 Recently, growing evidence support that the occurrence of MDD are the results of the correlational disorders from multiple systems or organs, not only limiting in brain.227,228 The comorbidities of MDD have attracted widespread attention, the intestinal gut microbial dysbiosis, liver dysfunction, immune system disorders all play important roles in the pathogenesis of MDD. Stressful conditions can affect the gut microbiota, which in turn induces the production of inflammatory mediators (mainly IL-6 and IFN-γ).256 Proinflammatory cytokines or toxic QUIN resulting from alterations in the microbiota may pass through the BBB and activate NMDARs.243 Under the dysfunction of liver, the level of ammonia is increased in the brain.257 The pathogenic factors of various organs at the body level and the pathological changes of glial cells at the cellular level should attract more attention to explain the pathogenesis of MDD.
Fig. 4.
The pathogenesis of MDD is closely related to synapses, astrocytes, microglia, and their interactions as well as interactions among organ. Genetic factors, stress and comorbidities are considered the most common pathogenic factors of MDD3. The traditional monoamine theory contends that MDD may cause by the deficits in monoamine neurotransmitters.49 Moreover, the other abnormal increase of neurotransmitters in the synaptic cleft, such as glutamate, GABA and ATP, has the high relationship with the pathogenesis of MDD.41,496 The interaction between neurons and glial cells can induce the oxidative stress, pro-inflammatory cytokines released, the reduction of neurotrophic factors. The microbiota-gut-brain axis is clearly disrupted in MDD.243,248 When liver dysfunction occurs and causes OS and neuroinflammation in the brain, which also contribute to the pathophysiology of MDD.497 Adobe Illustrator was used to generate this figure
New diagnostic approaches
MDD is a prevalent psychiatric disorder worldwide and is expected to become one of top disease in terms of burden by 2030.258 However, the current clinical diagnostic criteria for MDD are subjective, and diagnoses are mainly based on clinical symptoms, leading to high rates of missed and incorrect diagnoses. This section summarizes the newest research on diagnostic approaches for MDD, including serum indicators, neuroimaging indicators and multimodality scales. Research on new diagnostic approaches for MDD has the potential to improve our understanding of MDD pathogenesis and the accuracy of clinical diagnosis.
Potential serum indicators
The pathological mechanism of MDD can be studied in two ways: by exploring the pathophysiology of the disease and by identifying MDD-related neurobiological indicators4. Hence, identifying potential biomarkers for MDD could allow accurate diagnosis, faster treatment and effective monitoring of the disease. Recently, an increasing number of studies have confirmed the involvement of OS and neuroinflammation in MDD pathology.259,260 Two novel biomarkers, serum nicotinamide adenine dinucleotide phosphate oxidase 1 (NOX1) and Raftlin, are reported to have good diagnostic value in MDD patients. The effectiveness of elevated NOX1 and Raftlin levels in diagnosing MDD has been evaluated in clinical trials; the related mechanism is that NOX1 can regulate the ROS-antioxidant balance in patients with MDD through OS and the inflammatory repsonse.261 The serum level of the chemokine-like protein TAFA-5 (FAM19A5) has also been reported to be increased in patients with MDD, and increased serum FAM19A5 levels are associated with reactive astrogliosis, neuroinflammation, and neurodegeneration.262 In addition, the level of serum FAM19A5 was shown to have a negative correlation with cortical thickness in specific brain regions. These findings suggest that serum FAM19A5 could be a potential biomarker for neurodegenerative changes in MDD.
Functional magnetic resonance imaging indicators
In addition to serum indicators, neuroimaging metrics are potential objective tools for improving the accuracy of MDD diagnosis and must be studied in death. In recent years, many researchers have tried to diagnose MDD using MRI by identifying disease-specific functional and/or structural abnormalities in patients with MDD compared with healthy subjects.263 Structural MRI techniques, such as voxel-based morphometry (VBM), can be used to detect volume changes in gray matter.264 It has been reported that abnormal gray matter volume (GMV) in several brain regions is positively correlated with MDD.265,266 Regarding functional MRI, recent studies have revealed that cerebral functional abnormalities are not limited to specific brain regions in patients with MDD. These differences are also associated with hypoconnectivity within the frontoparietal network (FN), the DMN, and midline cortical regions.267,268 Furthermore, resting-state functional magnetic resonance imaging (R-fMRI) is an emerging neuroimaging technique used to study functional connectivity in the brain and holds great potential in aiding clinical diagnosis.269 It has the benefits of being noninvasive and easy to perform and offering high temporal and spatial resolution.270 As a result, it has played a significant role in MDD research and is a superior technique for researching MDD pathogenesis and identifying neuroimaging markers for MDD.271 Thus, indicators such as amplitude of low-frequency fluctuation (ALFF), fractional amplitude of low-frequency fluctuation (fALFF), regional homogeneity and functional connectivity (FC) have shown promise as neuroimaging markers for MDD. Recently, a study reported that increased average values of ALFF and fALFF in the right caudate and corpus callosum may serve as potential markers for diagnosing MDD.272 Another study based on the largest R-fMRI database of MDD patients confirmed that the DMN plays a crucial role in MDD diagnosis, as DMN FC is reduced in patients with recurrent MDD.273 These findings also suggest that the DMN should continue to be a prominent focus of MDD research.
New multi-modal evaluation scales
Given that structural and functional abnormalities are associated with MDD,274 using multimodal approaches is more appropriate than relying on a single feature for the diagnosis of MDD. However, research results related to the effectiveness of neuroimaging techniques in diagnosing MDD remain inconsistent.275 This may be attributed to variations in the types of structural and functional features examined; however, more importantly, very few studies have used multimodal approaches to diagnose MDD.276 Recently, in a study utilizing multimodal MRI data, patients with MDD were successfully distinguished from healthy controls by radiomics analysis.276 Radiomics is a rapidly developing field involving the extraction of quantitative information from diagnostic images, and it can be mainly divided into three steps: image acquisition, analysis and model building.277 Additionally, omics and neuroimaging techniques can be combined to construct models for diagnosing MDD; specifically, 5-hydroxytryptamine receptor 1 A/1B methylation data can be integrated with resting-state functional connectivity (rsFC) data. It was shown that this combination could be used to more accurately distinguish patients with MDD from healthy subjects than R-fMRI data or DNA methylation data alone.278
By now, the widely accepted objective diagnostic indicators or methods for MDD are still deficient. In addition to the unclear pathogenesis of MDD, insufficient sensitivity and accuracy of detection instruments are also the main reasons, especially the correlation between imaging characterization and disease-specific changes that need to be discussed.
Preventing the occurrence and recurrence of MDD
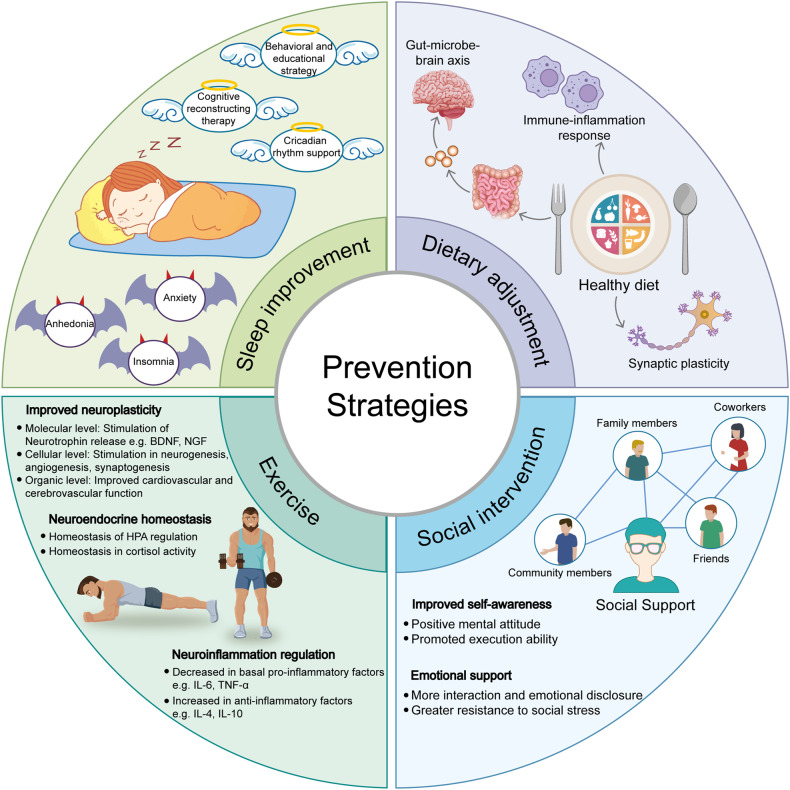
MDD is a disease with a high prevalence worldwide,279 and preventing its occurrence and recurrence is crucial. Lifestyle medicine is an evolving medical specialty that aims to prevent chronic, noncommunicable diseases through lifestyle interventions. The goal of lifestyle medicine is to prevent the occurrence and recurrence of disease by improving sleep hygiene and diet, increasing physical exercise, avoiding sedentary behavior, increasing social support, and improving mood. In recent years, an increasing number of studies have demonstrated that the occurrence and recurrence of MDD can be prevented by means of lifestyle medicine;280 we summarize these reports in this section.
Sleep improvements
Improving sleep is an important strategy to prevent the occurrence of depression. Insomnia is included in the diagnostic criteria for MDD.281 However, few studies have examined whether treating insomnia can prevent the exacerbation of depressive symptoms. Treating insomnia can prevent the worsening of depressive symptoms, and cognitive behavioral therapy for insomnia (CBT-I) is a recommended intervention for treating insomnia to improve sleep and mood.282–284 As a first-line treatment for insomnia, CBT-I includes cognitive therapy, stimulus management, sleep restriction, improved sleep hygiene, and relaxation.282,285 CBT-I can also lead to sustained remission of insomnia-related disorders, and continuous treatment of insomnia with CBT-I can also reduce the occurrence and recurrence of MDD.286 Circadian rhythm support (CRS) can strengthen the circadian rhythm by means of scheduled bright light exposure, physical activity, and body warming.287 Although CRS has been reported to have only an indirect effect in alleviating sleep disturbance and depressive symptoms,288 treatment with CRS may help maintain the beneficial effects of CBT-I.288,289 In one study, 44% of untreated patients but 38%, 28% and 9% of patients treated with CRS, CBT-I, and CBT-I + CRS, respectively, experienced clinically significant worsening of depressive symptoms during a 1-year follow-up period. Between-group comparisons showed that the percentage of patients who experienced worsening of depressive symptoms was significantly different between the CBT-I + CRS group and the nontreated and CRS groups.289 In a randomized controlled trial, exacerbation of depressive symptoms over one year was decreased in insomia patients with an increased risk of depression and insomnia patients treated by therapist-guided CBT-I combined with CRS; however, untreated insomnia patients with a high risk of depression experienced clinically significant worsening of depressive symptoms.288,289
Disrupted sleep is a common symptom of depressive episodes and increases the risk of MDD,290 but the correlation between the onset of sleep disturbance and MDD is still unclear. Additionally, patients with symptoms of sleep disturbance have a greater risk of MDD occurrence and recurrence.290,291 One study suggests that disrupted sleep may affect monoamine function and the HPA axis,292 even causing hyperarousal and inflammation.293 Additional studies on the pathological mechanism of depression have suggested that the HPA axis is hyperactive in MDD patients and that sensitivity to negative feedback is decreased.15 Additionally, one prospective cohort study reported that a history of sleep disorders can increase the risk of depression later in life and that subjective sleep problems are associated with clinically significant depressive symptoms.294
Dietary adjustment
Dietary adjustment is an effective, safe, and widely applicable method for preventing MDD, especially by inhibiting MDD-related pathological inflammation.295 Various nutrients can possess different anti-inflammatory properties; in contrast, there are many proinflammatory foods, such as those high in refined starch, sugar, and saturated fat and low in fiber and omega-3 fatty acids,296 which can promote the occurrence of inflammation to increase the risk of MDD.297 One study reported that the chance of being diagnosed with depression is higher among individuals who consume a proinflammtory diet than among those who consume an anti-inflammatory diet.295 Stimulation of the innate immune system by proinflammatory foods can result in mild inflammation and chronic illness, which may contribute to an increased risk of MDD.298 Furthermore, an increasing number of studies suggest that at the molecular and cellular levels, dietary factors have effects on neuronal function and synaptic plasticity, which may be implicated in the etiology of MDD.299,300 Therefore, adherence to a healthier diet can reduce the incidence of MDD, which is of great significance for the clinical treatment and prevention of depression.295
In addition, an increasing number of studies have identified the importance of the interaction among the microbiota, gut permeability, and immune-inflammatory processes in the pathophysiology of MDD.301 Because the interaction of bacteria of some taxa in the gut with peripheral inflammation with the brain may be related to depression pathophysiology,302,303 regulating the gut-microbe-brain axis may be a therapeutic and preventive strategy for psychiatric disorders.304 Restoration of the gut eubiosis can prevent the occurrence of MDD, and probiotics can normalize the gut ecosystem. Additionally, by altering the microbiota and regulating gut permeability, a gluten-free diet can alter the activity of the gut-microbe-brain axis, which has been discovered to be related to the pathogenesis of MDD.305–307 Other studies report that consuming a gluten-free diet and probiotic supplements together may inhibit the immune-inflammatory cascade in MDD patients, and decreased inflammation can improve the integrity of the gut barrier and alleviate depressive symptoms.307 Similarly, dietary fiber can also improve immune function by regulating the gut microbiota to prevent the occurrence of MDD,308 which is attributed to the inhibition of OS and inflammation.
Exercise
Increasing evidence suggests that physical exercise can prevent some mental disorders in addition to cardiovascular disease.280,309 This finding suggests that physical exercise may be able to prevent MDD. As reported in some studies, physical exercise can effectively prevent depression by affecting many molecular and cellular pathways; for instance, physical exercise can stimulate VEGF expression,310,311 leading to cellular level changes, such as stimulation of angiogenesis, increased delivery of neurotrophic factors and oxygen by the vascular system,312 an increase in the neurogenesis rate and induction of synaptogenesis.312,313 Ultimately, VEGF improves function in the hippocampus, which is one of the brain regions related to depression and stress regulation.314–316 Exercise also reduces the levels of proinflammatory factors (e.g., IL-6) and increases the levels of anti-inflammatory factors (e.g., IL-10), which is beneficial for preventing the occurrence of MDD.317–319 Furthermore, physical exercise for approximately 45 minutes per day can significantly reduce the risk of MDD.320,321 High-intensity activity, such as aerobic exercise, dancing, and the usage of exercise machines, and low-intensity exercises, including yoga and stretching, can all reduce the occurrence of MDD.322 Specifically, the combination of aerobic exercise and stretching as a multimodal therapeutic strategies has a significant antidepressant effect in depressed inpatients.323
Patients with MDD have significantly more sedentary than ordinary people, and they engage in less physical activity than what is recommended, i.e., an average of 150 min of moderate- to high-intensity physical activity weekly.324 This finding suggests that decreasing sedentary behavior or increasing physical activity levels should be a priority to prevent the occurrence of disease. In psychiatric centers, aerobic exercise has received increasing attention as a valuable method of prevention.324 Studies report that reduced depressive symptoms in MDD patients can be observed after increasing aerobic exercise and stretching exercise, with more significant alleviation of depressive symptoms after 8 weeks of aerobic exercise.325 Reward positivity (RewP) and error-related negativity (ERN) were identified as potential biomarkers of the exercise treatment response in depression.325 In individuals with MDD, aerobic exercise was found to be beneficial in ameliorating depressive symptoms, particularly in those with more severe depressive symptoms and a higher baseline RewP.325,326 RewP may be useful for identifying those who will benefit from exercise as a treatment for depression.325
Social intervention
Social support refers to the help provided by social relations and transactions.327 Social support may be obtained from a variety of individuals, including family members, friends, coworkers, and community members.328 Furthermore, a variety of factors, including the quantity and quality of support as well as subjectively perceived social support by individuals, impact the level of social support.329 It has been reported that MDD patients often lack social support, and receiving adequate social support can confer greater resistance to stress and prevent the occurrence and recurrence of MDD.330,331 Low-functioning social support or self-perceived poor social support causes worse symptoms and treatment outcomes in depressed patients.332–334 A previous study also reported that patients who lack adequate social support are more likely to experience MDD.335 Social support may have an influence on depression through neuroendocrine pathways,336,337 and social support can improve a person’s psychological wellbeing and make the individual more resistant to stress.337
Studies on structural social support, social network size, and mental health disorders have shown that less social contact and loneliness can cause more severe depressive symptoms.338 For individuals with MDD, it is necessary not only to increase the frequency of social contact but also to improve self-awareness and foster close functional supporttive relationships.335,339 Studies have reported that when controlling for all other variables, each aspect of social support is clearly associated with MDD, and to some extent, the occurrence of panic disorder in patients with MDD is more strongly associated with poor functional support. This finding suggests that functional support may be an important protective factor against MDD.331,335 Social support itself, especially emotional support,340 may alleviate and prevent depressive symptoms, and support from family members or friends can replace formal health care.341
In general, the pathological development of MDD is a gradual transition from subclinical state to clinical pathological changes. It is crucial to identify the core targets that lead to pathological changes from quantitative to qualitative changes during this process, and the above preventive interventions, sleep improvement, physical exercise, dietary regulation, and social intervention, may prolong or reverse the subclinical pathological stage (Fig. 5).
Fig. 5.
Schematic of prevention strategies for the occurrence and reoccurrence of MDD. An outline of various prevention strategies for MDD includes sleep improvement, dietary adjustment, exercise, and social intervention. Sleep disturbances have the high relationship with the occurrence of MDD, the anhedonia, anxiety and insomnia are the main symptoms of patients with MDD. The behavioral and educational strategies, cognitive reconstructing therapy and circadian rhythm support can be applied to improve sleep quality.281,289 Dietary adjustments are also suggested to have the potential effects to prevent the occurrence or re-occurrence of MDD, the improvement mechanism of diet may involve in the regulated immune-inflammatory responses, the improved gut-microbe-brain axis and synaptic plasticity.295,299,304 In addition, xxercise is an effective way to improve neuroplasticity, to maintain neuroendocrine homeostasis, and to regulate neuroinflammation, in order to effectively prevent the occurrence or re-occurrence of MDD.280,309 Importantly, getting social support from family members, friends, coworkers and community members can be helpful for the MDD patients’ recovery, these social interventions can let patients get emotional support and improve their self-awareness.328,340 Adobe Illustrator was used to generate this figure
Therapeutic drugs and strategies
This section summarizes new advances in research on the pharmacological mechanisms of common antidepressants and novel therapeutic strategies. Moreover, as laboratory animal models of MDD and other mental diseases are lacking, hindering the development of strategies for evaluating pharmacological effects and studying pathological mechanisms, we also discuss recent research on animal models.
The molecular mechanism of antidepressants
Tricyclic antidepressants
In the late 1950s, the first TCAs were approved and used for the treatment of depression.342 TCAs have a common three-ring chemical structure, and the main TCAs are imipramine, amitriptyline, clomipramine, desipramine and doxepin. The pharmacological mechanism of TCAs mainly involves its interaction with neurotransmitters in the brain, resulting in changes in neurotransmitter levels and an antidepressant effect. First, TCAs can inhibit the reuptake of neurotransmitters, leading to antidepressant effects. For example, they can influence the levels of 5-HT, NE, and to a lesser degree, DA, causing an increase in neurotransmitter concentrations in the synaptic gap and increasing neurotransmitter signaling to exert pharmacological effects.343 However, different TCAs inhibit 5-HT and NE reuptake to varying degrees. For instance, amitriptyline, imipramine, and desipramine strongly inhibit 5-HT reuptake,344 clomipramine specifically inhibits NE reuptake, and nortriptyline can inhibit both NE and 5-HT reuptake while also exerting central anticholinergic effects.345–347 Additionally, TCAs can antagonize 5-HT2A and 5-HT2C, thereby increasing the release of NE and DA in cortical areas.348–350 TCAs can bind to histamine receptors, especially H1 receptors, as well.351 By blocking H1 receptors, they can induce sedation and drowsiness, which may benefit depressed patients with sleep disorders.352 Furthermore, TCAs can also block muscarinic acetylcholine receptors, exerting anticholinergic effects and resulting in side effects such as dry mouth and constipation.353
In addition to the above-known pharmacological mechanisms, some recent studies have reported that amitriptyline can induce the activation of fibroblast growth factor receptor (FGFR), leading to the production of GDNF.354 In addition, amitriptyline can increase the expression of Cx43 to promote gap junction intercellular communication (GJIC) between astrocytes, thereby relieving depressive symptoms.355 This suggests that TCAs may also ameliorate severe depression through additional mechanisms involving astrocytes that are independent of the monoamine system to some extent. Further exploration is needed to fully understand the specific mechanism. Another study demonstrated that FKBP51, a crucial modulator of the glucocorticoid receptor (GR) pathway, can bind to clomipramine and impede its interaction with PIAS4. Inhibition of this interaction subsequently hinders sumoylation; this alteration represents a newly discovered mechanism by which the antidepressant drug exerts its effect.356
Selective serotonin reuptake inhibitor
According to a study, most severe depression patients are still advised to consider SSRIs as the initial choice for treatment.350 The main representative SSRIs drugs include fluoxetine, sertraline, paroxetine, and escitalopram. The mechanisms of action of SSRIs are commonly known as follows: first, SSRIs can selectively inhibit SERT, inhibiting the reuptake of 5-HT in the synaptic cleft and thereby exerting pharmacological effects.357 Second, SSRIs can impact the 5-HT signaling pathway, activating 5-HT1A.358,359 In addition, studies have shown that antagonism of 5-HT2A/2C receptors can enhance the effects of SSRIs such as fluoxetine.360,361 Third, long-term use of SSRIs can increase 5-HT transmission in the LC,362 thereby increasing the release of GABA to exert inhibitory effects on NA neurons.363 Fourth, long-term use of SSRIs is associated with neuroplasticity and neurogenesis in certain brain regions.364 SSRIs have been found to increase the expression of BDNF, a protein crucial for neuronal growth and survival, by acting on TrkB,365 which may contribute to the long-term therapeutic effects of SSRIs. Thus, our previous reports and others researches all suggested that astrocytic 5-HT2B receptors may be the potential pharmacological target of SSIRs.59,60,366–368
According to previous studies by our group, in the absence of SERT, SSRIs such as fluoxetine can act as direct agonists of astrocytic 5-HT2B receptors to exert antidepressant-like effects.60,64,179,366,369 In astroglia isolated from mice exposed to CUMS, fluoxetine activates the 5-HT2B receptor, promoting ERK1/2 phosphorylation. This increases downstream c-Fos expression, which in turn boosts BDNF synthesis.147 Furthermore, administration of fluoxetine effectively inhibits SD-induced stimulation of the NLRP3 inflammasome by the AKT/STAT3 and ERK/STAT3 pathways in vivo, and SD dramatically triggers depressive-like behaviors by stimulating astrocytic P2X7Rs.41,155 As previously mentioned, leptin may increase the expression of the 5-HT2B receptor in astrocytes via the LepR/JAK2/STAT3 pathway, and fluoxetine may be more effective in increasing BDNF levels and alleviating depressive-like behaviors due to the leptin-mediated increase in 5-HT2B receptor expression.130 Both in vivo and in vitro, fluoxetine’s inhibitory actions on A1 reactive astrocytes depend on astrocytic 5-HT2BR.55 Recently, fluoxetine was shown to act as a 5-HT2B agonist, and this finding is also supported by research by other groups. Fluoxetine has been reported to suppress the activation of A1 reactive astrocytes and decrease unusual behaviors in CMS-exposed mice. In vitro, Gq protein and b-arrestin1 are not necessary for fluoxetine’s effects on A1 astrocyte activation, and downstream signaling through astrocytic 5-HT2BR is responsible for fluoxetine’s inhibitory effects on A1 astrocyte activation in primary culture.55
Serotonin/norepinephrine reuptake inhibitors
SNRIs are often recommended as the initial choice for the treatment of MDD. Representative SNRIs include milnacipran, DXT, DVS, and venlafaxine. The molecular mechanisms of SNRIs can be summarized as follows: First, SNRIs inhibit the norepinephrine transporter (NET), which prevents the reuptake of NE into presynaptic neurons, leading to an increased concentration of NE in the synaptic cleft.370 Second, similar to SSRIs, SNRIs also inhibit SERT, resulting in an increased concentration of 5-HT in the synaptic cleft.371 For example, paroxetine and venlafaxine can inhibit SERT and, to a lesser extent, NET.372 Third, SNRIs inhibit the reuptake of both NE and 5-HT; thus, they have a dual mechanism of action. This dual inhibitory effect is believed to contribute to the broader therapeutic effects of SNRIs compared to SSRIs.373 Chronic treatment with fluoxetine has been shown to increase the expression of Cx43 in the rat PFC, which further prevents the dysfunction of astrocytic gap junctions induced by CUS and reverses the depressive-like behaviors caused by gap junction blockade.71
In a randomized controlled trial, MRI scan were taken after treatment with duloxetine and desvenlafaxine, and the results showed that the thalamo-cortico-periaqueductal network, which is associated with the experience of pain, may be an important target of action of antidepressant drugs.374
New potential pharmacological targets
The abovementioned antidepressants have been utilized as clinical therapies for MDD, but it is difficult to elucidate the exact pharmacological mechanisms of every medicine due to delayed clinical efficacy, poor treatment response to some patients, and difficulty in effectively controlling the incidence of suicide. Recently, several pharmacological agents have been discovered as potential antidepressants.
Ketamine, a noncompetitive antagonist of the NMDAR, has been shown to induce rapid and significant antidepressant effects within a few hours.375 Due to the rapid antidepressant effects of ketamine, unlike the delayed effects of traditional antidepressant drugs,376 research on this drug has continued and has revealed its mechanisms of action and potential drug targets. Ketamine can increase the level of BDNF in the prefrontal cortex, especially in the hippocampus, to exert antidepressant-like effects.377 Studies have suggested that ketamine can increase the synthesis of synaptic proteins through BDNF signaling dependent on the activate protein kinase B (Akt) and mammalian target of rapamycin complex 1 (mTORC1) signaling cascades.378,379 Ketamine may induce the activation of mTOR by the upstream kinase Akt, regulate the phosphorylation of GSK-3β, and exert antidepressant effects.380 Ketamine can block NMDARs in postsynaptic principal neurons in the PFC and hippocampus, increase synaptic function through homeostatic mechanisms, and reverse synaptic defects caused by chronic stress.381,382 Furthermore, by inhibiting NMDARs, ketamine can reduce the excitation of specific cortical GABAergic interneurons, resulting in a temporary increase in glutamate release that stimulates postsynaptic AMPA glutamate receptors. This, in turn, leads to the release of BDNF, activation of the TrkB receptor, and subsequent activation of the Akt/mTORC1 signaling pathway. These molecular events ultimately contribute to an increase in the number and functionality of synapses, leading to amelioration of depressive symptoms.383
Similar as ketamine, some other psychedelics can also produce fast and persistent antidepressant effects.384 Psilocybin, a classical psychedelic, can play its antidepressant roles by activating 5-HT2A receptors (5-HT2AR).385 Thus, to block the 5-HT2AR can not produce the antidepressant effects of psilocybin, only induce the hallucinogenic-like behaviors in mice.386 This proposes 5-HT2AR may not be the real pharmacological target for its antidepressant effects. Another study reports that the combination of lysergic acid diethylamide (LSD) and psilocybin may exert long-term antidepressant effects by promoting neural plasticity, which dose not involve in the hallucinogenic effects.384 Additionally, to target 5-HT2AR, the combination of LSD and psilocybin can lead to biased activation of the mediated signaling pathway and produce antidepressant effects without the side effects of hallucinations.387 Thus, the administration of psilocybin can rapidly and persistently induce neuronal dendritic remodeling in the medial frontal cortex of mice, and the psilocybin-induced newly formed dendritic spines can successfully transform functional synapses, suggesting that synaptic rewiring may also be one pharmacological mechanism of the rapid antidepressant effects of psilocybin.388 To further dissociate the hallucinogens effects from the psychedelics can be beneficial to develop more specific antidepressants with better therapeutic capacities.
Additionally, some novel potential therapeutic targets for MDD have also been reported, such as TGF-β1389 and growth-associated protein 43 (GAP-43).390 Multiple studies have shown that antidepressants may cause changes in TGF-β1 expression. Fluoxetine, paroxetine, venlafaxine, and sertraline have been shown to have the potential to increase the levels of TGF-β1, which may contribute to their antidepressant effects.391,392 Venlafaxine has also been reported to exert neuroprotection by increasing the production of FGF-2 and TGF-β1 in astrocytes following stroke.72 Then, chronic administration of desipramine has been shown to upregulate the expression of GAP-43 in the hippocampus of rats, potentially influencing neuronal plasticity in the CNS.390 GAP-43 has been suggested as a relevant target for the pharmacological effects of antidepressants.393,394
The most of above antidepressants have been widely used for the MDD patients according to the respective potential pharmacological actions (Fig. 6). Thus, the exactly neuromolecular mechanisms require deep studied and the new potential therapeutic targets and strategies still need further exploration.
Fig. 6.
The molecular mechanisms of tricyclic antidepressants (TCAs), selective serotonin reuptake inhibitors (SSRIs), serotonin/norepinephrine reuptake inhibitors (SNRIs) and ketamine. TCAs can inhibit the protein kinase C (PKC) pathway by blocking the H1 receptors (H1Rs),351,352 TCAs decreases the reuptake of dopamine (DA) by inhibiting dopamine transporters (DATs) in the presynaptic membrane, and increases the DA concentration in the synaptic gap, increase the effect of DA on dopamine receptors (DARs) of postsynaptic membrane.343 The activated DARs increase Ca2+ dependent CaMKII and CaMK4, as well as, the secretion of CREB.498,499 In another way, the stimulated DARs by DA can also activate the cAMP-PKA pathway, which in turn activates the levels of CREB and BDNF by stimulating MAPK/ERK1/2 pathway.500 TCAs, SSRIs, and SNRIs can all inhibit the reuptake of 5-HT by SERTs, specially SSRIs have the selective inhibition on SERTs, which increase the concentration of 5-HT in the synaptic gap and play antidepressive roles by effecting on 5-HTRs in postsynaptic membrane,343,344 which also activate the cAMP-PKA pathway.49,501 Moreover, TCAs and SNRIs can also inhibit the reuptake of NE by NETs, which also increase the concentration of NE in the synaptic gap, and in turn activate the effect of NE on adrenoceptors (ADRs) and activate the cAMP-PKA pathway in postsynaptic membrane.502 Besides of the AC/cAMP/PKA pathway, the effect of NE on ADRs can also activate protein kinase B (Akt) phosphorylation and mammalian target of rapamycin complex 1 (mTORC1) by stimulating TrkB, in order to promote the secretion of postsynaptic density 95 (PSD95) and glutamate receptor 1 (GluR1).502 Ketamine works as the antagonist of NMDAR on GABAergic interneurons, it suppresses the excitation of subsets of GABAergic interneurons, which reduces the gamma aminobutyric acid (GABA) effects on gamma aminobutyric acid type B receptor (GABABR), and relieves the inhibition of GABAergic interneurons on the release of glutamate, the latter further stimulates AMPAR on postsynaptic membrane and increases the level of BDNF, even the release of BDNF stimulates the above TrkB/AKT/mTORC1 pathway.503,504 Adobe Illustrator was used to generate this figure
Novel therapeutic strategies
New animal models
Establishing animal models with pathological features representative of those seen in humans is key for advancing MDD research. Currently, the widely utilized animal models of MDD include CUMS, behavioral despair (BD), learned helplessness (LH), and CSDS, drug withdrawal, and transgenic animal models.395 The CUMS model, one of the most commonly used animal models for MDD,64,172 exhibits depressive-like behaviors.396,397 According to a meta-analysis of 408 papers involving stress protocols, the most commonly used stressors for CUMS models are food and water deprivation, light cycle modification, wet bedding, cage tilting, social stress, and forced swimming.398 Recently, we constructed an improved depression model named the chronic unpredictable mild restraint (CUMR) model by using environmental interference.62 The stressors used to construct this CUMR mouse model included activity restriction, damp bedding, cage shaking, tail suspension, forced swimming, and 45° cage tilting. These stressors all restrict the activity of the mice; moreover, stressors that disturb physiological rhythms, chronic unpredictable rhythm disturbance (CURD), can cause manic-like behaviors in mice (Fig. 7). The disease-related pathological changes and serum indicators in the CUMR and CURD models are highly similar to those in patients in the clinic, and therapeutic medicines can effectively improve brain function and behavior in these models.62
Fig. 7.
The protocol and stressors used for CURD and CUMR. In order to establish the CUMR model, a combination of various stressors includes interference of constraint (a), damp bedding (b), cage shaking (c), tail suspension (d), forced swimming (e), and cage tilting (f). Among these six stressors, two were randomly selected and administered daily for a duration of 3 weeks. On the other hand, to establish the CUMR model, a set of behavioral constraints includes circadian rhythm (g), sleep deprivation (h), interference of cone light (i), interference of followed spotlight (j), high temperature stress (k), stroboscopic illumination (l), noise disturbance (m), and foot shock (n). Similarly, two out of these eight constraints were randomly chosen and applied daily for a period of 3 weeks62
Phototherapy
Phototherapy plays a significant role in regulating emotional behavior399 and can have strong and rapid effects on mood and alertness.400–402 There is increasing evidence for the therapeutic efficacy of phototherapy for MDD.403,404 The combination of phototherapy and antidepressants has better effects than antidepressants alone.402,405 Phototherapy utilizes bright light with a specific wavelength to stimulate the retina and affect the production of 5-HT and hormones in the brain.406 Furthermore, phototherapy can alleviate depressive-like behavior by targeting the retinal-thalamic ventral lateral geniculate nucleus/intergeniculate leaflet-lateral habenula (retinal-vlGN/IGL-LHb) circuit; this mechanism may explain how phototherapy alleviates MDD.407
Repetitive transcranial magnetic stimulation
Repetitive transcranial magnetic stimulation (rTMS) is an effective method used in clinical practice for treating patients with MDD.408 Multiple evaluations and analyses have shown that rTMS can effectively treat MDD in patients from different age groups, including children and adolescents,409,410 adults,411,412 and elderly patients.413,414 It is suggested that early use of rTMS in the treatment of depression in elderly patients may yield better results.415 Furthermore, research has indicated that rTMS can effectively treat perinatal depression.416 Increasing evidence suggests that rTMS of the anterior stimulation site of the left dlPFC can yield optimal treatment outcomes.417–419 A randomized controlled trial demonstrated that the efficacy of rTMS in treating depression is linked to precise targeting of the dlPFC, the activity of which exhibits a negative correlation with subgenual cingulate cortex activity.420 Identifying the optimal site for stimulation may further enhance the ability of rTMS to treat depression.421 Recently, a retrospective study was conducted, which included 29 systematic evaluations and reanalyzed 15 meta-analyses to assess the effectiveness and safety of transcranial magnetic stimulation (TMS) for treating MDD in adults.422 The results of the study indicated significant variations in the efficacy of TMS for MDD across different settings and revealed poor tolerability in certain populations, the further research is necessary to identify specific beneficiary populations for TMS in treating MDD and to personalize treatment based on comprehensive and detailed information.422
Psychological intervention
MDD is characterized by a gradual onset and a high risk of relapse.421 The American Medical Association recommends psychological interventions for individuals who are at a high risk of MDD. Some of the interventions commonly used for depression treatment include acceptance and commitment therapy, cognitive therapy, cognitive behavioral therapy (CBT), interpersonal therapy, and psychodynamic therapies.423 Specifically, the combination of psychological interventions and antidepressants effectively decreases the risk of relapse in cases of MDD.424–426
Acupuncture
Acupuncture, which mainly includes traditional body needling, moxibustion, EA, and laser acupuncture, is a traditional Chinese treatment modality used to treat various diseases.427 Compared with pharmacological therapies, acupuncture is more cost-effective and has fewer side effects.428 EA stimulation can effectively treat MDD;429–431 however, the specific mechanism by which acupuncture treats depression remains unclear. In previous research, EA at the ST36 acupoint was shown to prevent shrinkage of the prefrontal cortical astrocytes and alleviate depressive-like behavior in mice exposed to CUMS.432 The results of an 8-week clinical study involving 46 female patients with severe depression suggested that acupuncture may achieve therapeutic effects by modulating the corticostriatal reward/motivation circuit in patients with severe depression.433 Additionally, studies indicate that EA may have the potential to promote neuronal regeneration and exert antidepressant effects by elevating the phosphorylation of cyclic adenosine monophosphate response element binding protein and the levels of BDNF.434 Acupuncture at the GV20 and GV24 acupoints may alleviate depression symptoms by regulating the calmodulin-dependent protein kinase (CaMK) signaling pathway.435 The antidepressant effect of EA may also be associated with increased synaptic transmission in the ventromedial prefrontal cortex (vmPFC).436 A recent meta-analysis of 43 randomized controlled trials involving adult subjects with acupuncture for MDD demonstrated that acupuncture, either alone or in combination with antidepressants, significantly reduced the hamilton depression scal scores and had fewer adverse effects compared to antidepressants, however, further rigorous experiments are still required to determine the optimal frequency of acupuncture for MDD in order to achieve better efficacy.437
In conclusion, the common antidepressants can improve some depressive symptoms in some patients with depression, but are always associated with the risk of adverse effects or recurrence. Although some new developed treatment methods can improve depression symptoms in a certain program, the compatibility between potential treatment mechanisms and pathological mechanisms still needs further research. In particular, the therapeutic principle of acupuncture still needs to be explored in depth, and the accompanied therapeutic mechanism and application potential of traditional Chinese medicine in depression deserve to be explored in depth.
Clinical research progress
In summary, the pathological features of MDD and pharmacological mechanism of antidepressants have been widely studied. Furthermore, there have been many clinical studies on MDD, and studies of human postmortem tissues and clinical medical images, multomics studies, and preclinical/clinical trials of new therapeutic drugs have improved our understanding of the disease mechanism.
Transcriptional studies of human postmortem tissue
A recent meta-analysis of eight transcriptome datasets identified 566 disease-related genes that are consistently up- or downregulated in patients with MDD. The brain regions in which these genes are expressed include the amygdala, subgenual anterior cingulate, and dorsolateral prefrontal cortex, and the associated molecular pathways include reduced neurotrophic support, neural signaling, and GABA function.438 Through the discovery of nonoverlapping proteins that bind to calcium parvalbumin, calretinin, and the neural peptide somatostatin, subgroups of GABA interneurons that govern main pyramidal neurons differently were identified.439 Decreased cortical levels of GABA and specific populations of GABA neurons have been reported in investigations of postmortem MDD patient tissues,440 and the SST mRNA level is specifically decreased in patients with MDD.213
The DR nucleus is the largest and most significant conduit of forebrain serotonergic input.441 In postmortem samples of the human brain, several transcriptional regulators are dysregulated within the DR, including transcription-related elements (such as EGR1, TOB1, and CSDA), which bind to genes to stimulate their expression directly or in response to environmental cues, and NRs (NR4A2, NR4A3, THRA, and THRB), which are activated by ligands and regulate translation by targeting genes.442 In addition, transporters for GRs generally regulate the activity of the HPA axis by negative feedback.443 According to studies of postmortem brain tissues, hyperactivity of the HPA axis in MDD patients could be caused by methylation-mediated changes in GR transcription.444 The expression of nerve growth factor-inducible protein A (NGFI-A), an enzyme that bindss exon 1 F of GR, is reduced in the hippocampus of patients with MDD, which may contribute to low methylation levels in the brain.444 Moreover, in postmortem MDD patients, total GR levels are unchanged, while level of GRα in the amygdala and cingulate gyrus is decreased.
Sex-related molecular markers of MDD
Women are more likely than males to experience recurring MDD445 and are twice as likely to experience MDD throughout their lifetimes.446 Compared with male patients, female patients with MDD have symptoms that manifest sooner in the disease course, last longer, and are more severe; in addition, they experience hunger changes, weight fluctuations, and sleep difficulties more frequently.447,448
In postmortem samples of patients who committed suicide due to MDD, the expression of DNA methyltransferases (DNMTs) in the frontopolar cortex was found to be more significantly increased in women than in men; elevated methylation is associated with decreased levels of the GABAA receptor alpha-1 subunit in men, which supports sex-related epigenetic alterations in transcription.449 A gene array meta-analysis also revealed sex differences in MDD, with depressed females being more likely than depressed men to have lower production of somatostatin, a GABA neuron biomarker in corticolimbic brain regions according to postmortem analysis.450 X-linked chromosomal polymorphisms affect the expression of the GABA-synthesizing enzyme and somatostatin.450 Analyses of postmortem brain tissues showed an increase in the transcription of numerous glutamate-related genes in the prefrontal cortex in depressed women but not in depressed men; depressed women exhibited more alterations in glutamate receptor expression, while depressed men showed only GRM5 downregulation.451
In postmortem brain specimens, there were no transcription differences between MDD men and controls, and the levels of 5-HT1D receptors and the transcription factors NUDR and REST, which regulate 5-HT activity, in 5-HT-containing neurons in the ventral raphe nuclei were found to be higher in MDD females.452 5-HT receptors and regulators were shown to exhibit sex-specific alterations in expression at the protein level, and postmortem investigations have largely focused on female subjects. The protein levels of 5-HT1AR and NUDR, which regulate 5-HT signaling, in the prefrontal cortex were found to be lower in MDD women than in control subjects; however, this difference was not observed in MDD males compared with controls.453 The NA/NE system, especially in the LC, is another monoaminergic system that exhibits sex-related variations and influences MDD risk. In fact, some researchers have found that the levels of microRNAs (miRNAs), short RNA molecules that control the expression of genes and play roles in psychological disorders,454 are higher in the LC of suicidal female subjects than in the LC of suicidal male subjects. MiR-1179 is associated with GRIA3 and MAOA, which are involved in neuropsychiatric diseases.455
OS is commonly linked to the onset of MDD. A study found that whereas cysteine and 1-methylinosine levels were much higher in males with MDD, they were significantly lower in females with MDD.456 These metabolites are related to OS. Furthermore, several studies found a significant link between MDD and lipid metabolism;457 for example, as 1-Oalkyl-2-acyl-PEs levels are decreased in MDD, showing a negative correlation with the extent of depression, lysophospholipid (LPC) and phospholipid (PC) levels are increased in MDD, exhibiting a substantial positive correlation with depression severity.458 Similarly, a study found that men and women had different lipid concentrations.456 These clinical data suggest that sex differences in MDD may result from differences in OS and lipid metabolism, but further research is required to make this connection.
Multiomics studies
Transcriptome studies, which explore relationships among the expression of genes and diseases, are regarded as an essential for investigating disease-causing mutations in genes, the mechanisms of disease development and progression, and disease-related target genes.459 Dorsolateral prefrontal cortex tissues have been employed to identify genes and miRNAs that show changes in expression and biological processes that are altered in patients with MDD.460 Serpin Family H Member 1 (SERPINH1), IL-8, humanin like-8 (MTRNRL8), and chemokine ligand 4 (CCL4) are among the genes whose expression is altered in MDD.460,461 According to Gene Ontology (GO) enrichment analysis, MDD is related to decreased expression of genes related to oligodendrocyte development, glutamatergic neurotransmission modulation, and oxytocin receptor expression. These findings confirm that impairment of the blood-brain barrier and microglial, endothelial cell, ATPase, and astrocyte function exacerbate MDD; the involvement of these cells, molecules, and structures in MDD should be further investigated.460
The field of study known as genomics focuses on the transcription of genes, the precise interactions among genes, and the control of gene activity. MDD has been linked to numerous biological processes, including energy metabolism. When the transcription of genes involved in glycolysis and glycogen synthesis was examined in the hippocampus of depressed rats, it was found that the mRNA expression of Slc2a3, which codes for GLUT3, is considerably increased.462 Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and lactate dehydrogenase B (LDHB) mRNA levels were found to be substantially decreased in MDD.462 The transcription of genes in the brain tissues of IL18-/- mice was examined with the use of genome-wide microarrays, and the results revealed that urocortin 3 (Ucn3) expression was increased.463 Ucn3 controls how the body processes glucose;464 therefore, a change in Ucn3 expression will result in energy imbalance. Gene comethylation analysis was performed in the brains of individuals with MDD. The findings revealed that the methylation of genes associated with mitochondria was dramatically decreased, indicating impaired mitochondrial function.465
Metabolomics has recently emerged as a useful technique for identifying markers and pathways associated with a wide range of diseases.466 It is often used to analyze the mechanisms underlying disease occurrence and progression and the effects of small-molecule compounds. In one study, targeted metabolomic analysis of the CSF of 14 MDD patients who were not taking medication, 14 MDD patients in remission, and 18 healthy controls was performed.467 An analysis of the tryptophan, tyrosine, purine pathways, and associated pathways revealed that in patients in remission, methionine levels were higher, while tryptophan and tyrosine levels were lower. The same group of patients also showed changes in the methionine-to-glutathione ratio, indicating alterations in OS and methylation. The levels of these same metabolites were altered in MDD patients who were not taking medication, although not to a significant degree.467
Clinical medical imaging studies
MRI has been widely employed in research in recent years to pinpoint patterns of brain alterations linked to MDD. Many studies have demonstrated that structural and fMRI has outstanding potential as trustworthy imaging modalities for monitoring MDD treatment responses. A study indicated that MDD patients had large volume decreases in various frontal areas, such as the anterior cingulate cortex and OFC, which were linked to problems with stress management and emotional processing.468 People with MDD also exhibited structural changes in their parietal lobes.469 Alterations in the total gray matter volume and an increase in cortical thickness are the two findings that are most consistent.470
The functional changes in the frontal lobe in MDD are hotly contested. A study discovered lower precuneus, supragenual anterior cingulate cortex, dorsomedial PFC, and dorsomedial thalamus lower activity when processing pleasant stimuli in MDD patients.471 Another study found that during the processing of favorable self-indulgent information, individuals with MDD displayed higher activity in the medial PFC and anterior cingulate cortex.472 The right hippocampus, parahippocampal gyrus, left amygdala, and the whole caudate nucleus all had functional changes in activity in MDD patients compared to healthy controls, indicating that the temporal lobe might be involved in the pathogenesis of MDD.473
Although it is not feasible to evaluate synapse density directly in people in vivo, positron emission tomography (PET) can be utilized to gather useful information. It is thought that impairments of functional connections and synaptic atrophy are two factors that contribute to the symptoms of MDD. An indirect method of estimating synaptic density is to count the number of nerve terminals using synaptic vesicle glycoprotein 2 A (SV2A). The researchers examined synaptic density in MDD patients who were not taking any medication using positron emission PET with the SV2A radioligand [11C] UCB-J.474 The results revealed that reductions in the synapse density in areas connected with various processes, such as emotion control and thought (the dorsolateral prefrontal cortex, anterior cingulate cortex, and hippocampus), are related to to the severity of depressive disorders. Additionally, it was shown that compared with healthy subjects, subjects with MDD had reduced dlPFC resting-state connectivity throughout the brain. It was found that the dlPFC-posterior cingulate cortex connection was inversely negatively linked to the severity of depression symptoms and connected with synapse activity in the dlPFC, indicating that synaptic loss may impair antagonistism within the centers of both networks, which are typically at odds.474
Preclinical and clinical trials of new therapeutic drugs
Esmethadone is a new, noncompetitive NMDAR antagonist475 that exhibits fast antidepressant-like action by improving performance of rats in the forced swim test.476 Esmethadone can also alleviate neural dysfunction linked to symptoms of depression by boosting the synapse and spine density and restoring spinogenesis, in addition to correcting depressive-like behaviors in animal models of depression.378,477 Esmethadone was found to reduce cognitive symptoms in individuals with MDD in a stage II clinical study478 and to increase the levels of circulating BDNF in normal individuals in a stage I clinical investigation.479 In a phase II study involving patients who had received insufficient benefit from conventional antidepressants, esmethadone demonstrated immediate, strong, and long-lasting antidepressant benefits.478
Ketamine is the most well-known rapid-acting antidepressant and an NMDAR antagonist.383 GluN1, GluN2, and GluN3 are NMDAR subunits.480 Ketamine exerts a quick and effective antidepressant effect by binding to the asparagine 616 residue of GluN1 and the leucine 642 residue of GluN2A.192 In a clinical experiment, the effect of supplementary injection of subanesthetic doses of ketamine on thoughts of suicide in MDD patients was evaluated, and the results showed that the reduction in thoughts of suicide among MDD patients receiving ketamine was mostly sustained.481 In several studies, a single dose of ketamine reduced immobility in the forced swim test immediately after injection and had effects similar to those of an antidepressant.482,483
The S-enantiomer of ketamine, esketamine, has been approved by the U.S. Food and Drug Administration (FDA) for depression treatment.383 Moreover, formulations of ketamine are also being developed, and intranasal esketamine spray has shown high efficacy in treating MDD.484 Additionally, hydroxynorketamine (HNK), a metabolite of ketamine, can exert its anti-depressive effects by an NMDAR-independent mechanism.377 One of these mechanisms involves increasing BDNF levels; an increasing number of studies have shown that BDNF signaling is an important target of antidepressants.377 Thus, ketamine can also exert anti-inflammatory effects, a large amount of evidence suggests a tight relationship between neuroinflammation and the pathogenesis of MDD.485–487 A summary of clinical trials related to new therapeutic drugs for MDD is shown in Table 1.
Table 1.
Clinical trials of new therapeutic drugs for MDD
| Study | Duration | Mean age (SD) in years | Mood disorder type | Diagnostic tool | Interventions | Control | Outcome indicators | Blinding of participants | Outcomes |
|---|---|---|---|---|---|---|---|---|---|
| Fava M et al.478 | 7 days | NR | MED, with an inadequate response to one to three courses of antidepressant treatment | DSM-5; HAMD |
(1) REL-1017 75 mg (Day 1), 25 mg/day (Days 2-7) (2) REL-1017 100 mg (Day 1), 50 mg/day (Days 2-7) |
Placebo | MADRS; SDQ; CGI-S; CGI-I | Double blind | REL-1017 may have rapid and sustained antidepressant effects in patients with inadequate response to antidepressant treatment. |
| De Martin S et al.479 | 10 days | 39 (8) | Healthy | / | REL-1017 25 mg | Placebo | BDNF plasma levels; systolic BP; diastolic BP | Double blind | Administration of 25 mg of REL-1017 significantly increased BDNF plasma levels and significantly decreased diastolic blood pressure. |
| Hochschild A et al.481 | 2 days | 38.4 (13.2) | MDE, unipolar depression | DSM-IV; HAMD-17; SSI | Ketamine 0.5 mg/kg | Midazolam 0.02 mg/kg | SSI; HAMD-24; POMS; BDI | Double blind | Ketamine resulted in greater improvements in HDRS, HDRS, BDI and POMS scores and reduced suicidal ideation in patients. |
| Daly EJ et al.484 | 10 weeks, with an additional 8 weeks of post-treatment follow-up | 44.7 (10.0) | TRD | DSM-IV-TR | Esketamine 28 mg, 56 mg, or 84 mg twice weekly | Placebo, an inactive substance | MADRS | Double blind | Antidepressant effects of intranasal esketamine in the treatment of TRD are rapid and dose-related. |
| Abbasi SH et al.488 | 6 weeks | NR | MDD | DSM-IV-TR; HAMD-17 | Celecoxib 200 mg twice daily plus sertraline 200 mg/day | Placebo plus sertraline 200 mg/day | HAMD; IL-6 concentrations in the sera | Double blind | The serum IL-6 concentration in the celecoxib group was significantly reduced, which may be related to its antidepressant activity and can be used as an auxiliary antidepressant drug. |
| Akhondzadeh S et al.489 | 6 weeks | 34.6 (6.8) | MDD | DSM-IV-TR; HAMD | Celecoxib 400 mg/day plus fluoxetine 40 mg/day | Placebo plus fluoxetine 40 mg/day | HAMD | Double blind | Celecoxib combined with fluoxetine is more effective than fluoxetine alone in treating major depression. Celecoxib may be an effective adjunct to treatment of patients with major depressive disorder. |
| Nettis MA et al.490 | 4 weeks | 47.0 (10.0) | MDD with peripheral inflammation (CRP ≥ 1 mg/L) | DSM-5; MINI; HAMD-17; levels of serum CRP | Minocycline 200 mg/day | Placebo | HAMD-17; BDI- II; CGI; PSS; SHAPS; STAI-S; STAI-T: levels of inflammatory biomarkers | Double blind | Add-on therapy with minocycline may be effective in patients with MDD in patients with low-grade inflammation and CRP ≥ 3 mg/L |
| Hasebe K et al.491 | 12 weeks | 51.7 (14.4) | MDD | MINI-PLUS 5; MADRS | Minocycline 200 mg/day | Placebo | HAMA; Q-LES-Q-SF; LIFE-RIFT; PGI; CGI-I; levels of IL-6, LBP and BDNF in blood samples | Double blind | There were no overall changes in IL-6, LBP or BDNF following adjunctive minocycline treatment. |
| Su KP et al.492 | 2 weeks | 53 (10) | Depression induced by IFN-α | DSM-IV | Omega-3 fatty acids: EPA 3.5 g/day or DHA 1.75 g/day | Placebo (high oleic oil) | HAMD-21; NTRS; percentage of participants with MDE induced by IFN-α | Double blind | EPA is effective in preventing depression in HCV patients receiving IFN-α. |
| Berk M et al.493 | 12 weeks | 20.2 (2.6) | MDD | SCID-I/P; MADRS | Rosuvastatin 10 mg/day or aspirin 100 mg/day | Placebo | MADRS; QIDS-SR; GAD-7; CGI-I/S; PGI; Q-LES-Q-SF; SAS-SR; SOFAS | Triple blind | The addition of aspirin or rosuvastatin did not produce any beneficial effects in the treatment of depression in young adults, but rosuvastatin may have potential therapeutic role in adolescent depression. |
| Meltzer-Brody S et al.494 | 3 days, with an additional 4 weeks of post-treatment follow-up | NR | PPD | SCID-I; HAMD | Brexanolone 90 μg/kg/h or brexanolone 60 μg/kg/h | Placebo | HAMD-17; CGI-I; MADRS; EPDS; PHQ; GAD-7 | Double blind | Compared with placebo, after 60 hours of intravenous infusion of brexanolone, the total HAMD score of patients with postpartum depression was significantly reduced, and the drug effect was rapid and long-lasting. |
| Leal GC et al.495 | 7 days | NR | TRD; and failure to respond to at least two adequate antidepressant trials in the current episode | MINI; DSM-5; MADRS | (R)-ketamine 0.5 mg/kg | Placebo (saline solution) | MADRS; CGI-S; CGI-I | Double blind | (R)-ketamine is capable of producing rapid and potent antidepressant effects in TRD subjects. |
SD standard deviation, NR not reported, MDE major depressive episode, DSM Diagnostic and Statistical Manual of Mental Disorders, HAMD Hamilton Depression Scale, MADRS Montgomery-Asberg Depression Rating Scale, SDQ Symptoms of Depression Questionnaire, CGI-S Clinical Global Impressions Severity Scale, CGI-I Clinical Global Impressions Improvement Scale, BDNF brain-derived neurotrophic factor, BP blood pressure, SSI Beck Scale for Suicidal Ideation, POMS Profile of Mood States, BDI Beck Depression Inventory, TRD treatment resistant depression, MDD major depressive disorder, IL-6 interleukin-6, CRP C-reactive protein, MINI Mini International Neuropsychiatric Interview, PSS Perceived Stress Scale, SHAPS Snaith–Hamilton Pleasure Scale, STAI-S Spielberger State-Trait Anxiety Rating Scale-State, STAI-T Spielberger State-Trait Anxiety Rating Scale-Trait, HAMA Hamilton Anxiety Scale, Q-LES-Q-SF Quality of Life Enjoyment and Satisfaction Questionnaire Short Form, LIFE-RIFT Range of Impaired Functioning Tool, PGI Patient Global Impression, LBP lipopolysaccharide binding protein, IFN interferon, EPA eicosapentaenoic acid, DHA docosahexaenoic acid, NTRS Neurotoxicity Rating Scale, SCID Structured Clinical Interview, QIDS-SR Quick Inventory of Depression Symptomatology–Self Report, GAD-7 Generalised Anxiety Disorder seven-item scale, SAS-SR Social Adjustment Scale–Self Report, SOFAS Social and Occupational Functioning Scale, PPD postpartum depression, EPDS Edinburgh Postnatal Depression Scale, PHQ Patient Health Questionnaire
The development of the present therapeutic medicines in clinic mainly targets the discovered pharmacological targets, mainly focusing on the key receptors or enzymes. However, at the organelle level of neural cells, the disturbed energy metabolism of mitochondria and the related RNA drugs, as well as the dysfunctions of lipid and glucose metabolism in psychopathological condition, still need deep exploration. Totally, the research on the mechanism of therapeutic drugs always requires the development of pathological mechanisms as support.
Conclusions and future perspectives
MDD is a heterogeneous disease, its pathological and pharmacological mechanisms are still unclear, and diagnostic and therapeutic methods for MDD are limited. SSRIs and SNRIs are the first-line treatments for MDD in the clinic; however, a sizable portion of MDD patients do not respond well to the currently available antidepressants. According to research on real-world sequential therapies, even after numerous treatment attempts, almost 30% of MDD patients do not experience remission. This suggests that the existing theories and hypotheses cannot completely explain the pathogenesis of MDD and that more research on the pharmacological mechanisms of currently available antidepressants is still needed. We mainly discussed the potential etiology and pathogenesis of MDD from the perspective of widely accepted theories, including the neurotransmitter and receptor hypothesis, HPA axis hypothesis, cytokine hypothesis, neuroplasticity hypothesis and systemic influence hypothesis. A more comprehensive understanding of the pathophysiological mechanisms of MDD might significantly improve our capacity to develop preventive and more effective therapeutic methods that can help reduce the burden of and pain caused by major depression. Knowledge of the cellular processes that drive these alterations and the symptoms they cause may offer crucial will provide insight for new treatments.
MDD is connected with several cellular and structural modifications in the nervous system. Nonetheless, in the majority of these alterations cannot be consistently observed in vivo. Therefore, several issues need to be considered in future research: (i) Studies of animal models have made important contributions to our understanding of the pathophysiology of major depression, and more representative animal models of MDD should be developed. (ii) Because of our incomplete understanding of the disease and the disease’s intrinsic intricacy, there is an urgent need to develop updated imaging technologies and imaging software to allow advances in our understanding of the disease. (iii) The therapeutic shortcomings of traditional antidepressants have prompted the need for further drug discovery and development. (iv) MDD is strongly associated with many systems, and it will be important to further elucidate the mechanisms associated with MDD and other pathological conditions.
Acknowledgements
This work was supported by the National Natural Science Foundation of China, MX [grant number 32271038] and BL [grant number 81871852]; Shenyang Science and Technology Innovation Talents Project, BL [grant number RC210251]; ‘ChunHui’ Program of Education Ministry, BL [grant number 2020703]; National Natural Science Foundation of China-Russian Science Foundation (NSFC-RSF), YT [grant number 82261138557]; Sichuan Provincial Administration of Traditional Chinese Medicine, YT [grant number 2023zd024].
Author contributions
L.C., X.W., and B.L. provided direction and guidance throughout the preparation of this manuscript. L.C., X.W., and B.L. wrote and edited the manuscript. L.C., S.L., S.W., M.X., and B.L. reviewed and made significant revisions to the manuscript. L.C., S.L., S.W., X.W., Y.L., W.Y., Y.W., Y.T., M.X., and B.L. collected and prepared the related papers. All authors have read and approved the article.
Competing interests
The authors declare no competing interests.
Contributor Information
Maosheng Xia, Email: xiamaosheng1981@163.com.
Baoman Li, Email: bmli@cmu.edu.cn.
References
- 1.Disease GBD, Injury I, Prevalence C. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1789–1858. doi: 10.1016/S0140-6736(18)32279-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nagy C, et al. Single-nucleus transcriptomics of the prefrontal cortex in major depressive disorder implicates oligodendrocyte precursor cells and excitatory neurons. Nat. Neurosci. 2020;23:771–781. doi: 10.1038/s41593-020-0621-y. [DOI] [PubMed] [Google Scholar]
- 3.Malhi GS, Mann JJDepression. Lancet. 2018;392:2299–2312. doi: 10.1016/S0140-6736(18)31948-2. [DOI] [PubMed] [Google Scholar]
- 4.Fellinger M, et al. Seasonality in major depressive disorder: effect of sex and age. J. Affect Disord. 2022;296:111–116. doi: 10.1016/j.jad.2021.09.051. [DOI] [PubMed] [Google Scholar]
- 5.Liu CH, et al. Role of inflammation in depression relapse. J. Neuroinflammation. 2019;16:90. doi: 10.1186/s12974-019-1475-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burcusa SL, Iacono WG. Risk for recurrence in depression. Clin. Psychol. Rev. 2007;27:959–985. doi: 10.1016/j.cpr.2007.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Monroe SM, Harkness KL. Life stress, the “kindling” hypothesis, and the recurrence of depression: considerations from a life stress perspective. Psychol. Rev. 2005;112:417–445. doi: 10.1037/0033-295X.112.2.417. [DOI] [PubMed] [Google Scholar]
- 8.Albert KM, Newhouse PA. Estrogen, stress, and depression: cognitive and biological interactions. Annu. Rev. Clin. Psychol. 2019;15:399–423. doi: 10.1146/annurev-clinpsy-050718-095557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kessler RC, Chiu WT, Demler O, Merikangas KR, Walters EE. Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch. Gen. Psychiatry. 2005;62:617–627. doi: 10.1001/archpsyc.62.6.617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Monteleone P, Maj M. The circadian basis of mood disorders: recent developments and treatment implications. Eur. Neuropsychopharmacol. 2008;18:701–711. doi: 10.1016/j.euroneuro.2008.06.007. [DOI] [PubMed] [Google Scholar]
- 11.Rice F, et al. Adolescent and adult differences in major depression symptom profiles. J. Affect Disord. 2019;243:175–181. doi: 10.1016/j.jad.2018.09.015. [DOI] [PubMed] [Google Scholar]
- 12.Hasin DS, et al. Epidemiology of adult DSM-5 major depressive disorder and its specifiers in the United States. JAMA Psychiatry. 2018;75:336–346. doi: 10.1001/jamapsychiatry.2017.4602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Howard DM, et al. Genome-wide association study of depression phenotypes in UK Biobank identifies variants in excitatory synaptic pathways. Nat. Commun. 2018;9:1470. doi: 10.1038/s41467-018-03819-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kovacs M, Lopez-Duran N. Prodromal symptoms and atypical affectivity as predictors of major depression in juveniles: implications for prevention. J. Child Psychol. Psychiatry. 2010;51:472–496. doi: 10.1111/j.1469-7610.2010.02230.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stetler C, Miller GE. Depression and hypothalamic-pituitary-adrenal activation: a quantitative summary of four decades of research. Psychosom. Med. 2011;73:114–126. doi: 10.1097/PSY.0b013e31820ad12b. [DOI] [PubMed] [Google Scholar]
- 16.Milaneschi Y, Lamers F, Berk M, Penninx B. Depression heterogeneity and its biological underpinnings: toward immunometabolic depression. Biol. Psychiatry. 2020;88:369–380. doi: 10.1016/j.biopsych.2020.01.014. [DOI] [PubMed] [Google Scholar]
- 17.Zhou X, et al. Astrocyte, a promising target for mood disorder interventions. Front. Mol. Neurosci. 2019;12:136. doi: 10.3389/fnmol.2019.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Monroe SM, Harkness KL. Major depression and its recurrences: life course matters. Annu. Rev. Clin. Psychol. 2022;18:329–357. doi: 10.1146/annurev-clinpsy-072220-021440. [DOI] [PubMed] [Google Scholar]
- 19.Wray NR, et al. Genome-wide association analyses identify 44 risk variants and refine the genetic architecture of major depression. Nat. Genet. 2018;50:668–681. doi: 10.1038/s41588-018-0090-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gittins RA, Harrison PJ. A morphometric study of glia and neurons in the anterior cingulate cortex in mood disorder. J. Affect. Disord. 2011;133:328–332. doi: 10.1016/j.jad.2011.03.042. [DOI] [PubMed] [Google Scholar]
- 21.Miguel-Hidalgo JJ, et al. Glial and glutamatergic markers in depression, alcoholism, and their comorbidity. J. Affect. Disord. 2010;127:230–240. doi: 10.1016/j.jad.2010.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sequeira A, et al. Global brain gene expression analysis links glutamatergic and GABAergic alterations to suicide and major depression. PLoS ONE. 2009;4:e6585. doi: 10.1371/journal.pone.0006585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rajkowska G, Stockmeier CA. Astrocyte pathology in major depressive disorder: insights from human postmortem brain tissue. Curr. Drug Targets. 2013;14:1225–1236. doi: 10.2174/13894501113149990156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsuang MT, Taylor L, Faraone SV. An overview of the genetics of psychotic mood disorders. J. Psychiatr. Res. 2004;38:3–15. doi: 10.1016/S0022-3956(03)00096-7. [DOI] [PubMed] [Google Scholar]
- 25.Lohoff FW. Overview of the genetics of major depressive disorder. Curr. Psychiatry Rep. 2010;12:539–546. doi: 10.1007/s11920-010-0150-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kendall KM, et al. The genetic basis of major depression. Psychol. Med. 2021;51:2217–2230. doi: 10.1017/S0033291721000441. [DOI] [PubMed] [Google Scholar]
- 27.Power RA, et al. Genome-wide association for major depression through age at onset stratification: Major Depressive Disorder Working Group of the psychiatric genomics consortium. Biol. Psychiatry. 2017;81:325–335. doi: 10.1016/j.biopsych.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Huang C, et al. Proteomic analysis of olfactory bulb suggests CACNA1E as a promoter of CREB signaling in microbiota-induced depression. J. Proteom. 2019;194:132–147. doi: 10.1016/j.jprot.2018.11.023. [DOI] [PubMed] [Google Scholar]
- 29.Flint J. The genetic basis of major depressive disorder. Mol. Psychiatry. 2023;28:2254–2265. doi: 10.1038/s41380-023-01957-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tripp A, Kota RS, Lewis DA, Sibille E. Reduced somatostatin in subgenual anterior cingulate cortex in major depression. Neurobiol. Dis. 2011;42:116–124. doi: 10.1016/j.nbd.2011.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Seney ML, Tripp A, McCune S, Lewis DA, Sibille E. Laminar and cellular analyses of reduced somatostatin gene expression in the subgenual anterior cingulate cortex in major depression. Neurobiol. Dis. 2015;73:213–219. doi: 10.1016/j.nbd.2014.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hicks EM, et al. Integrating genetics and transcriptomics to study major depressive disorder: a conceptual framework, bioinformatic approaches, and recent findings. Transl. Psychiatry. 2023;13:129. doi: 10.1038/s41398-023-02412-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Labonte B, et al. Sex-specific transcriptional signatures in human depression. Nat. Med. 2017;23:1102–1111. doi: 10.1038/nm.4386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Levey DF, et al. Bi-ancestral depression GWAS in the Million Veteran Program and meta-analysis in >1.2 million individuals highlight new therapeutic directions. Nat. Neurosci. 2021;24:954–963. doi: 10.1038/s41593-021-00860-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cohen-Woods S, Craig IW, McGuffin P. The current state of play on the molecular genetics of depression. Psychol. Med. 2013;43:673–687. doi: 10.1017/S0033291712001286. [DOI] [PubMed] [Google Scholar]
- 36.Slavich GM, Sacher J. Stress, sex hormones, inflammation, and major depressive disorder: Extending Social Signal Transduction Theory of Depression to account for sex differences in mood disorders. Psychopharmacology. 2019;236:3063–3079. doi: 10.1007/s00213-019-05326-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McEwen BS, Nasca C, Gray JD. Stress effects on neuronal structure: hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology. 2016;41:3–23. doi: 10.1038/npp.2015.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fries GR, Saldana VA, Finnstein J, Rein T. Molecular pathways of major depressive disorder converge on the synapse. Mol. Psychiatry. 2023;28:284–297. doi: 10.1038/s41380-022-01806-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Keller J, et al. HPA axis in major depression: cortisol, clinical symptomatology and genetic variation predict cognition. Mol. Psychiatry. 2017;22:527–536. doi: 10.1038/mp.2016.120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dong L, Li B, Verkhratsky A, Peng L. Cell type-specific in vivo expression of genes encoding signalling molecules in the brain in response to chronic mild stress and chronic treatment with fluoxetine. Psychopharmacology. 2015;232:2827–2835. doi: 10.1007/s00213-015-3921-2. [DOI] [PubMed] [Google Scholar]
- 41.Xia M, et al. Sleep deprivation selectively down-regulates astrocytic 5-HT(2B) receptors and triggers depressive-like behaviors via stimulating P2X(7) receptors in mice. Neurosci. Bull. 2020;36:1259–1270. doi: 10.1007/s12264-020-00524-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hare DL, Toukhsati SR, Johansson P, Jaarsma T. Depression and cardiovascular disease: a clinical review. Eur. Heart J. 2014;35:1365–1372. doi: 10.1093/eurheartj/eht462. [DOI] [PubMed] [Google Scholar]
- 43.Dunbar JA, et al. Depression: an important comorbidity with metabolic syndrome in a general population. Diabetes Care. 2008;31:2368–2373. doi: 10.2337/dc08-0175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Arnaud AM, et al. Impact of major depressive disorder on comorbidities: a systematic literature review. J. Clin. Psychiatry. 2022;83:21r14328. doi: 10.4088/JCP.21r14328. [DOI] [PubMed] [Google Scholar]
- 45.Sato S, Yeh TL. Challenges in treating patients with major depressive disorder: the impact of biological and social factors. CNS Drugs. 2013;27:S5–10. doi: 10.1007/s40263-012-0028-8. [DOI] [PubMed] [Google Scholar]
- 46.Greenberg PE, Fournier AA, Sisitsky T, Pike CT, Kessler RC. The economic burden of adults with major depressive disorder in the United States (2005 and 2010) J. Clin. Psychiatry. 2015;76:155–162. doi: 10.4088/JCP.14m09298. [DOI] [PubMed] [Google Scholar]
- 47.Greenberg PE, et al. The economic burden of adults with major depressive disorder in the United States (2010 and 2018) Pharmacoeconomics. 2021;39:653–665. doi: 10.1007/s40273-021-01019-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dean J, Keshavan M. The neurobiology of depression: an integrated view. Asian J. Psychiatr. 2017;27:101–111. doi: 10.1016/j.ajp.2017.01.025. [DOI] [PubMed] [Google Scholar]
- 49.Bhatt S, Devadoss T, Manjula SN, Rajangam J. 5-HT(3) receptor antagonism a potential therapeutic approach for the treatment of depression and other disorders. Curr. Neuropharmacol. 2021;19:1545–1559. doi: 10.2174/1570159X18666201015155816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Inazu M, Takeda H, Matsumiya T. Functional expression of the norepinephrine transporter in cultured rat astrocytes. J. Neurochem. 2003;84:136–144. doi: 10.1046/j.1471-4159.2003.01514.x. [DOI] [PubMed] [Google Scholar]
- 51.Schipke CG, Heuser I, Peters O. Antidepressants act on glial cells: SSRIs and serotonin elicit astrocyte calcium signaling in the mouse prefrontal cortex. J. Psychiatr. Res. 2011;45:242–248. doi: 10.1016/j.jpsychires.2010.06.005. [DOI] [PubMed] [Google Scholar]
- 52.Novikova IN, et al. Adrenaline induces calcium signal in astrocytes and vasoconstriction via activation of monoamine oxidase. Free Radic. Biol. Med. 2020;159:15–22. doi: 10.1016/j.freeradbiomed.2020.07.011. [DOI] [PubMed] [Google Scholar]
- 53.Martin, H. et al. Insulin modulates emotional behavior through a serotonin-dependent mechanism. Mol. Psychiatry (2022). [Oline ahead of print]. [DOI] [PubMed]
- 54.Ogawa S, et al. Plasma L-tryptophan concentration in major depressive disorder: new data and meta-analysis. J. Clin. Psychiatry. 2014;75:e906–915. doi: 10.4088/JCP.13r08908. [DOI] [PubMed] [Google Scholar]
- 55.Fang Y, et al. Fluoxetine inhibited the activation of A1 reactive astrocyte in a mouse model of major depressive disorder through astrocytic 5-HT(2B)R/beta-arrestin2 pathway. J. Neuroinflammation. 2022;19:23. doi: 10.1186/s12974-022-02389-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Savitz J, Lucki I, Drevets WC. 5-HT(1A) receptor function in major depressive disorder. Prog. Neurobiol. 2009;88:17–31. doi: 10.1016/j.pneurobio.2009.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Verkhratsky A, Rodriguez JJ, Parpura V. Astroglia in neurological diseases. Future Neurol. 2013;8:149–158. doi: 10.2217/fnl.12.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhang S, et al. 5-HT2B receptors are expressed on astrocytes from brain and in culture and are a chronic target for all five conventional ‘serotonin-specific reuptake inhibitors. Neuron Glia Biol. 2010;6:113–125. doi: 10.1017/S1740925X10000141. [DOI] [PubMed] [Google Scholar]
- 59.Verkhratsky A, Parpura V, Scuderi C, Li B. Astroglial serotonin receptors as the central target of classic antidepressants. Adv. Neurobiol. 2021;26:317–347. doi: 10.1007/978-3-030-77375-5_13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Li B, et al. Biphasic regulation of caveolin-1 gene expression by fluoxetine in astrocytes: opposite effects of PI3K/AKT and MAPK/ERK signaling pathways on c-fos. Front. Cell. Neurosci. 2017;11:335. doi: 10.3389/fncel.2017.00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Li B, Zhang S, Li M, Hertz L, Peng L. Chronic treatment of astrocytes with therapeutically relevant fluoxetine concentrations enhances cPLA2 expression secondary to 5-HT2B-induced, transactivation-mediated ERK1/2 phosphorylation. Psychopharmacology. 2009;207:1–12. doi: 10.1007/s00213-009-1631-3. [DOI] [PubMed] [Google Scholar]
- 62.Li X, et al. A novel murine model of mania. Mol. Psychiatry. 2023;28:3044–3054. doi: 10.1038/s41380-023-02037-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Li B, Zhang S, Zhang H, Hertz L, Peng L. Fluoxetine affects GluK2 editing, glutamate-evoked Ca(2+) influx and extracellular signal-regulated kinase phosphorylation in mouse astrocytes. J. Psychiatry Neurosci. 2011;36:322–338. doi: 10.1503/jpn.100094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Li B, et al. Cell type-specific gene expression and editing responses to chronic fluoxetine treatment in the in vivo mouse brain and their relevance for stress-induced anhedonia. Neurochem. Res. 2012;37:2480–2495. doi: 10.1007/s11064-012-0814-1. [DOI] [PubMed] [Google Scholar]
- 65.Breton-Provencher V, Drummond GT, Feng J, Li Y, Sur M. Spatiotemporal dynamics of noradrenaline during learned behaviour. Nature. 2022;606:732–738. doi: 10.1038/s41586-022-04782-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mori K, et al. Effects of norepinephrine on rat cultured microglial cells that express alpha1, alpha2, beta1 and beta2 adrenergic receptors. Neuropharmacology. 2002;43:1026–1034. doi: 10.1016/S0028-3908(02)00211-3. [DOI] [PubMed] [Google Scholar]
- 67.O’Sullivan JB, Ryan KM, Curtin NM, Harkin A, Connor TJ. Noradrenaline reuptake inhibitors limit neuroinflammation in rat cortex following a systemic inflammatory challenge: implications for depression and neurodegeneration. Int. J. Neuropsychopharmacol. 2009;12:687–699. doi: 10.1017/S146114570800967X. [DOI] [PubMed] [Google Scholar]
- 68.Choi HS, et al. The anti-inflammatory activity of duloxetine, a serotonin/norepinephrine reuptake inhibitor, prevents kainic acid-induced hippocampal neuronal death in mice. J. Neurol. Sci. 2015;358:390–397. doi: 10.1016/j.jns.2015.10.001. [DOI] [PubMed] [Google Scholar]
- 69.Wang J, et al. Desvenlafaxine prevents white matter injury and improves the decreased phosphorylation of the rate-limiting enzyme of cholesterol synthesis in a chronic mouse model of depression. J. Neurochem. 2014;131:229–238. doi: 10.1111/jnc.12792. [DOI] [PubMed] [Google Scholar]
- 70.Luo Y, Kataoka Y, Ostinelli EG, Cipriani A, Furukawa TA. National prescription patterns of antidepressants in the treatment of adults with major depression in the US Between 1996 and 2015: a population representative survey based analysis. Front. Psychiatry. 2020;11:35. doi: 10.3389/fpsyt.2020.00035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sun JD, Liu Y, Yuan YH, Li J, Chen NH. Gap junction dysfunction in the prefrontal cortex induces depressive-like behaviors in rats. Neuropsychopharmacology. 2012;37:1305–1320. doi: 10.1038/npp.2011.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zepeda R, et al. Venlafaxine treatment after endothelin-1-induced cortical stroke modulates growth factor expression and reduces tissue damage in rats. Neuropharmacology. 2016;107:131–145. doi: 10.1016/j.neuropharm.2016.03.011. [DOI] [PubMed] [Google Scholar]
- 73.Bekhbat M, et al. Correction to: functional connectivity in reward circuitry and symptoms of anhedonia as therapeutic targets in depression with high inflammation: evidence from a dopamine challenge study. Mol. Psychiatry. 2022;27:4122. doi: 10.1038/s41380-022-01754-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Cui Y, et al. Astroglial Kir4.1 in the lateral habenula drives neuronal bursts in depression. Nature. 2018;554:323–327. doi: 10.1038/nature25752. [DOI] [PubMed] [Google Scholar]
- 75.Skupio U, et al. Astrocytes determine conditioned response to morphine via glucocorticoid receptor-dependent regulation of lactate release. Neuropsychopharmacology. 2020;45:404–415. doi: 10.1038/s41386-019-0450-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rasheed N, et al. Differential response of central dopaminergic system in acute and chronic unpredictable stress models in rats. Neurochem. Res. 2010;35:22–32. doi: 10.1007/s11064-009-0026-5. [DOI] [PubMed] [Google Scholar]
- 77.Morales I, Fuentes A, Ballaz S, Obeso JA, Rodriguez M. Striatal interaction among dopamine, glutamate and ascorbate. Neuropharmacology. 2012;63:1308–1314. doi: 10.1016/j.neuropharm.2012.08.007. [DOI] [PubMed] [Google Scholar]
- 78.Solis O, Garcia-Sanz P, Herranz AS, Asensio MJ, Moratalla R. L-DOPA reverses the increased free amino acids tissue levels induced by dopamine depletion and rises GABA and tyrosine in the striatum. Neurotox. Res. 2016;30:67–75. doi: 10.1007/s12640-016-9612-x. [DOI] [PubMed] [Google Scholar]
- 79.Corkrum M, et al. Dopamine-evoked synaptic regulation in the nucleus accumbens requires astrocyte activity. Neuron. 2020;105:1036–1047.e1035. doi: 10.1016/j.neuron.2019.12.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Adermark L, et al. Astrocytes modulate extracellular neurotransmitter levels and excitatory neurotransmission in dorsolateral striatum via dopamine D2 receptor signaling. Neuropsychopharmacology. 2022;47:1493–1502. doi: 10.1038/s41386-021-01232-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Agren R, Sahlholm K. Voltage-dependent dopamine potency at D(1)-like dopamine receptors. Front Pharmacol. 2020;11:581151. doi: 10.3389/fphar.2020.581151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fasano C, et al. Dopamine facilitates dendritic spine formation by cultured striatal medium spiny neurons through both D1 and D2 dopamine receptors. Neuropharmacology. 2013;67:432–443. doi: 10.1016/j.neuropharm.2012.11.030. [DOI] [PubMed] [Google Scholar]
- 83.Hao Y, Plested AJR. Seeing glutamate at central synapses. J. Neurosci. Methods. 2022;375:109531. doi: 10.1016/j.jneumeth.2022.109531. [DOI] [PubMed] [Google Scholar]
- 84.Trudler D, et al. alpha-synuclein oligomers induce glutamate release from astrocytes and excessive extrasynaptic NMDAR activity in neurons, thus contributing to synapse loss. J. Neurosci. 2021;41:2264–2273. doi: 10.1523/JNEUROSCI.1871-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Schmitz F, et al. Methylphenidate decreases ATP levels and impairs glutamate uptake and Na(+),K(+)-ATPase activity in juvenile rat hippocampus. Mol. Neurobiol. 2017;54:7796–7807. doi: 10.1007/s12035-016-0289-1. [DOI] [PubMed] [Google Scholar]
- 86.Kucukibrahimoglu E, et al. The change in plasma GABA, glutamine and glutamate levels in fluoxetine- or S-citalopram-treated female patients with major depression. Eur. J. Clin. Pharmacol. 2009;65:571–577. doi: 10.1007/s00228-009-0650-7. [DOI] [PubMed] [Google Scholar]
- 87.Chiba S, et al. Chronic restraint stress causes anxiety- and depression-like behaviors, downregulates glucocorticoid receptor expression, and attenuates glutamate release induced by brain-derived neurotrophic factor in the prefrontal cortex. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2012;39:112–119. doi: 10.1016/j.pnpbp.2012.05.018. [DOI] [PubMed] [Google Scholar]
- 88.Sullivan R, et al. Cloning, transport properties, and differential localization of two splice variants of GLT-1 in the rat CNS: implications for CNS glutamate homeostasis. Glia. 2004;45:155–169. doi: 10.1002/glia.10317. [DOI] [PubMed] [Google Scholar]
- 89.Zou J, et al. Glutamine synthetase down-regulation reduces astrocyte protection against glutamate excitotoxicity to neurons. Neurochem. Int. 2010;56:577–584. doi: 10.1016/j.neuint.2009.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Karlsson RM, et al. Assessment of glutamate transporter GLAST (EAAT1)-deficient mice for phenotypes relevant to the negative and executive/cognitive symptoms of schizophrenia. Neuropsychopharmacology. 2009;34:1578–1589. doi: 10.1038/npp.2008.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Karlsson RM, et al. Reduced alcohol intake and reward associated with impaired endocannabinoid signaling in mice with a deletion of the glutamate transporter GLAST. Neuropharmacology. 2012;63:181–189. doi: 10.1016/j.neuropharm.2012.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Di Castro MA, et al. Local Ca2+ detection and modulation of synaptic release by astrocytes. Nat. Neurosci. 2011;14:1276–1284. doi: 10.1038/nn.2929. [DOI] [PubMed] [Google Scholar]
- 93.Matos M, et al. Deletion of adenosine A2A receptors from astrocytes disrupts glutamate homeostasis leading to psychomotor and cognitive impairment: relevance to schizophrenia. Biol. Psychiatry. 2015;78:763–774. doi: 10.1016/j.biopsych.2015.02.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yue N, et al. Activation of P2X7 receptor and NLRP3 inflammasome assembly in hippocampal glial cells mediates chronic stress-induced depressive-like behaviors. J. Neuroinflammation. 2017;14:102. doi: 10.1186/s12974-017-0865-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Cao X, et al. Astrocyte-derived ATP modulates depressive-like behaviors. Nat. Med. 2013;19:773–777. doi: 10.1038/nm.3162. [DOI] [PubMed] [Google Scholar]
- 96.Cunha GM, Canas PM, Oliveira CR, Cunha RA. Increased density and synapto-protective effect of adenosine A2A receptors upon sub-chronic restraint stress. Neuroscience. 2006;141:1775–1781. doi: 10.1016/j.neuroscience.2006.05.024. [DOI] [PubMed] [Google Scholar]
- 97.Batalha VL, et al. Adenosine A(2A) receptor blockade reverts hippocampal stress-induced deficits and restores corticosterone circadian oscillation. Mol. Psychiatry. 2013;18:320–331. doi: 10.1038/mp.2012.8. [DOI] [PubMed] [Google Scholar]
- 98.Wang M, et al. Lateral septum adenosine A(2A) receptors control stress-induced depressive-like behaviors via signaling to the hypothalamus and habenula. Nat. Commun. 2023;14:1880. doi: 10.1038/s41467-023-37601-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Lucas M, et al. Coffee, caffeine, and risk of completed suicide: results from three prospective cohorts of American adults. World J. Biol. Psychiatry. 2014;15:377–386. doi: 10.3109/15622975.2013.795243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Lucas M, et al. Coffee, caffeine, and risk of depression among women. Arch. Intern. Med. 2011;171:1571–1578. doi: 10.1001/archinternmed.2011.393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Coelho JE, et al. Overexpression of adenosine A2A receptors in rats: effects on depression, locomotion, and anxiety. Front. Psychiatry. 2014;5:67. doi: 10.3389/fpsyt.2014.00067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kaster MP, et al. Caffeine acts through neuronal adenosine A2A receptors to prevent mood and memory dysfunction triggered by chronic stress. Proc. Natl Acad. Sci. USA. 2015;112:7833–7838. doi: 10.1073/pnas.1423088112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hines DJ, Schmitt LI, Hines RM, Moss SJ, Haydon PG. Antidepressant effects of sleep deprivation require astrocyte-dependent adenosine mediated signaling. Transl. Psychiatry. 2013;3:e212. doi: 10.1038/tp.2012.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Etievant A, et al. Astroglial control of the antidepressant-like effects of prefrontal cortex deep brain stimulation. EBioMedicine. 2015;2:898–908. doi: 10.1016/j.ebiom.2015.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Serchov T, et al. Increased signaling via adenosine A1 receptors, sleep deprivation, imipramine, and ketamine inhibit depressive-like behavior via induction of Homer1a. Neuron. 2015;87:549–562. doi: 10.1016/j.neuron.2015.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Dostal CR, Carson Sulzer M, Kelley KW, Freund GG, McCusker RH. Glial and tissue-specific regulation of Kynurenine Pathway dioxygenases by acute stress of mice. Neurobiol. Stress. 2017;7:1–15. doi: 10.1016/j.ynstr.2017.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yang W, et al. Dex modulates the balance of water-electrolyte metabolism by depressing the expression of AVP in PVN. Front. Pharmacol. 2022;13:919032. doi: 10.3389/fphar.2022.919032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Jacobson L. Forebrain glucocorticoid receptor gene deletion attenuates behavioral changes and antidepressant responsiveness during chronic stress. Brain Res. 2014;1583:109–121. doi: 10.1016/j.brainres.2014.07.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Perez P. Glucocorticoid receptors, epidermal homeostasis and hair follicle differentiation. Dermatoendocrinol. 2011;3:166–174. doi: 10.4161/derm.15332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Reichrath J. Ancient friends, revisited: new aspects on the important role of nuclear receptor signalling for skin physiology and for the treatment of skin diseases. Dermatoendocrinol. 2011;3:121–124. doi: 10.4161/derm.17544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Herbert J, Lucassen PJ. Depression as a risk factor for Alzheimer’s disease: genes, steroids, cytokines and neurogenesis - What do we need to know? Front. Neuroendocrinol. 2016;41:153–171. doi: 10.1016/j.yfrne.2015.12.001. [DOI] [PubMed] [Google Scholar]
- 112.Rosenthal LJ, Goldner WS, O’Reardon JP. T3 augmentation in major depressive disorder: safety considerations. Am. J. Psychiatry. 2011;168:1035–1040. doi: 10.1176/appi.ajp.2011.10030402. [DOI] [PubMed] [Google Scholar]
- 113.Mokrani MC, Duval F, Erb A, Gonzalez Lopera F, Danila V. Are the thyroid and adrenal system alterations linked in depression? Psychoneuroendocrinology. 2020;122:104831. doi: 10.1016/j.psyneuen.2020.104831. [DOI] [PubMed] [Google Scholar]
- 114.Stenzel D, Huttner WB. Role of maternal thyroid hormones in the developing neocortex and during human evolution. Front. Neuroanat. 2013;7:19. doi: 10.3389/fnana.2013.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mendes-de-Aguiar CB, et al. Thyroid hormone increases astrocytic glutamate uptake and protects astrocytes and neurons against glutamate toxicity. J. Neurosci. Res. 2008;86:3117–3125. doi: 10.1002/jnr.21755. [DOI] [PubMed] [Google Scholar]
- 116.Rastogi L, Godbole MM, Sinha RA, Pradhan S. Reverse triiodothyronine (rT3) attenuates ischemia-reperfusion injury. Biochem. Biophys. Res. Commun. 2018;506:597–603. doi: 10.1016/j.bbrc.2018.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Isgor C, Watson SJ. Estrogen receptor alpha and beta mRNA expressions by proliferating and differentiating cells in the adult rat dentate gyrus and subventricular zone. Neuroscience. 2005;134:847–856. doi: 10.1016/j.neuroscience.2005.05.008. [DOI] [PubMed] [Google Scholar]
- 118.Russell JK, Jones CK, Newhouse PA. The role of estrogen in brain and cognitive aging. Neurotherapeutics. 2019;16:649–665. doi: 10.1007/s13311-019-00766-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Biegon A, Reches A, Snyder L, McEwen BS. Serotonergic and noradrenergic receptors in the rat brain: modulation by chronic exposure to ovarian hormones. Life Sci. 1983;32:2015–2021. doi: 10.1016/0024-3205(83)90053-X. [DOI] [PubMed] [Google Scholar]
- 120.Arinami H, Suzuki Y, Tajiri M, Tsuneyama N, Someya T. Role of insulin-like growth factor 1, sex and corticosteroid hormones in male major depressive disorder. BMC Psychiatry. 2021;21:157. doi: 10.1186/s12888-021-03116-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Walf AA, Rhodes ME, Frye CA. Antidepressant effects of ERbeta-selective estrogen receptor modulators in the forced swim test. Pharm. Biochem Behav. 2004;78:523–529. doi: 10.1016/j.pbb.2004.03.023. [DOI] [PubMed] [Google Scholar]
- 122.Frye CA, Walf AA. Estrogen and/or progesterone administered systemically or to the amygdala can have anxiety-, fear-, and pain-reducing effects in ovariectomized rats. Behav. Neurosci. 2004;118:306–313. doi: 10.1037/0735-7044.118.2.306. [DOI] [PubMed] [Google Scholar]
- 123.Hughes ZA, et al. WAY-200070, a selective agonist of estrogen receptor beta as a potential novel anxiolytic/antidepressant agent. Neuropharmacology. 2008;54:1136–1142. doi: 10.1016/j.neuropharm.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 124.Weiser MJ, Wu TJ, Handa RJ. Estrogen receptor-beta agonist diarylpropionitrile: biological activities of R- and S-enantiomers on behavior and hormonal response to stress. Endocrinology. 2009;150:1817–1825. doi: 10.1210/en.2008-1355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Irwin RW, et al. Selective oestrogen receptor modulators differentially potentiate brain mitochondrial function. J. Neuroendocrinol. 2012;24:236–248. doi: 10.1111/j.1365-2826.2011.02251.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ho MF, et al. Ketamine and ketamine metabolites as novel estrogen receptor ligands: Induction of cytochrome P450 and AMPA glutamate receptor gene expression. Biochem. Pharmacol. 2018;152:279–292. doi: 10.1016/j.bcp.2018.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Hochol A, et al. Effects of neuropeptides B and W on the rat pituitary-adrenocortical axis: in vivo and in vitro studies. Int J. Mol. Med. 2007;19:207–211. [PubMed] [Google Scholar]
- 128.Kurosawa N, Shimizu K, Seki K. The development of depression-like behavior is consolidated by IL-6-induced activation of locus coeruleus neurons and IL-1beta-induced elevated leptin levels in mice. Psychopharmacology. 2016;233:1725–1737. doi: 10.1007/s00213-015-4084-x. [DOI] [PubMed] [Google Scholar]
- 129.Lee JY, et al. The association between serum leptin levels and post-stroke depression: a retrospective clinical study. Ann. Rehabil. Med. 2015;39:786–792. doi: 10.5535/arm.2015.39.5.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Li Z, et al. Fluoxetine improves behavioural deficits induced by chronic alcohol treatment by alleviating RNA editing of 5-HT(2C) receptors. Neurochem. Int. 2020;134:104689. doi: 10.1016/j.neuint.2020.104689. [DOI] [PubMed] [Google Scholar]
- 131.de Morais H, et al. Increased oxidative stress in prefrontal cortex and hippocampus is related to depressive-like behavior in streptozotocin-diabetic rats. Behav. Brain Res. 2014;258:52–64. doi: 10.1016/j.bbr.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 132.Rawdin BJ, et al. Dysregulated relationship of inflammation and oxidative stress in major depression. Brain Behav. Immun. 2013;31:143–152. doi: 10.1016/j.bbi.2012.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Dringen R. Metabolism and functions of glutathione in brain. Prog. Neurobiol. 2000;62:649–671. doi: 10.1016/S0301-0082(99)00060-X. [DOI] [PubMed] [Google Scholar]
- 134.Greaney JL, Saunders EFH, Alexander LM. Short-term salicylate treatment improves microvascular endothelium-dependent dilation in young adults with major depressive disorder. Am. J. Physiol. Heart Circ. Physiol. 2022;322:H880–H889. doi: 10.1152/ajpheart.00643.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Belanger M, Magistretti PJ. The role of astroglia in neuroprotection. Dialogues Clin. Neurosci. 2009;11:281–295. doi: 10.31887/DCNS.2009.11.3/mbelanger. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Chen B, et al. The neuroprotective mechanism of lithium after ischaemic stroke. Commun. Biol. 2022;5:105. doi: 10.1038/s42003-022-03051-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Maes M, et al. Major differences in neurooxidative and neuronitrosative stress pathways between major depressive disorder and types I and II bipolar disorder. Mol. Neurobiol. 2019;56:141–156. doi: 10.1007/s12035-018-1051-7. [DOI] [PubMed] [Google Scholar]
- 138.Early JO, et al. Circadian clock protein BMAL1 regulates IL-1beta in macrophages via NRF2. Proc. Natl Acad. Sci. USA. 2018;115:E8460–E8468. doi: 10.1073/pnas.1800431115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Pasco JA, et al. Association of high-sensitivity C-reactive protein with de novo major depression. Br. J. Psychiatry. 2010;197:372–377. doi: 10.1192/bjp.bp.109.076430. [DOI] [PubMed] [Google Scholar]
- 140.Bakunina N, Pariante CM, Zunszain PA. Immune mechanisms linked to depression via oxidative stress and neuroprogression. Immunology. 2015;144:365–373. doi: 10.1111/imm.12443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Paul ER, et al. Peripheral and central kynurenine pathway abnormalities in major depression. Brain Behav. Immun. 2022;101:136–145. doi: 10.1016/j.bbi.2022.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Wang Y, et al. Inhibition of activated astrocyte ameliorates lipopolysaccharide- induced depressive-like behaviors. J. Affect Disord. 2019;242:52–59. doi: 10.1016/j.jad.2018.08.015. [DOI] [PubMed] [Google Scholar]
- 143.Lenk T, Rabet S, Sprick M, Raabe G, Schroder U. Insight into the interaction of furfural with metallic surfaces in the electrochemical hydrogenation process. Chemphyschem. 2023;24:e202200614. doi: 10.1002/cphc.202200614. [DOI] [PubMed] [Google Scholar]
- 144.Zhang JC, Yao W, Hashimoto K. Brain-derived neurotrophic factor (BDNF)-TrkB signaling in inflammation-related depression and potential therapeutic targets. Curr. Neuropharmacol. 2016;14:721–731. doi: 10.2174/1570159X14666160119094646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Kennis M, et al. Prospective biomarkers of major depressive disorder: a systematic review and meta-analysis. Mol. Psychiatry. 2020;25:321–338. doi: 10.1038/s41380-019-0585-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Norden DM, Trojanowski PJ, Villanueva E, Navarro E, Godbout JP. Sequential activation of microglia and astrocyte cytokine expression precedes increased Iba-1 or GFAP immunoreactivity following systemic immune challenge. Glia. 2016;64:300–316. doi: 10.1002/glia.22930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Li X, et al. Leptin increases expression of 5-HT(2B) receptors in astrocytes thus enhancing action of fluoxetine on the depressive behavior induced by sleep deprivation. Front. Psychiatry. 2018;9:734. doi: 10.3389/fpsyt.2018.00734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Takano K, Yamasaki H, Kawabe K, Moriyama M, Nakamura Y. Imipramine induces brain-derived neurotrophic factor mRNA expression in cultured astrocytes. J. Pharmacol. Sci. 2012;120:176–186. doi: 10.1254/jphs.12039FP. [DOI] [PubMed] [Google Scholar]
- 149.Zhang Y, et al. BDNF enhances electrophysiological activity and excitatory synaptic transmission of RA projection neurons in adult male zebra finches. Brain Res. 2023;1801:148208. doi: 10.1016/j.brainres.2022.148208. [DOI] [PubMed] [Google Scholar]
- 150.Quesseveur G, et al. BDNF overexpression in mouse hippocampal astrocytes promotes local neurogenesis and elicits anxiolytic-like activities. Transl. Psychiatry. 2013;3:e253. doi: 10.1038/tp.2013.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Zor F, et al. Effect of VEGF gene therapy and hyaluronic acid film sheath on peripheral nerve regeneration. Microsurgery. 2014;34:209–216. doi: 10.1002/micr.22196. [DOI] [PubMed] [Google Scholar]
- 152.Sun L, et al. Propofol directly induces caspase-1-dependent macrophage pyroptosis through the NLRP3-ASC inflammasome. Cell Death Dis. 2019;10:542. doi: 10.1038/s41419-019-1761-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Alcocer-Gomez E, Cordero MD. NLRP3 inflammasome: a new target in major depressive disorder. CNS Neurosci. Ther. 2014;20:294–295. doi: 10.1111/cns.12230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Beckwith KS, et al. Plasma membrane damage causes NLRP3 activation and pyroptosis during Mycobacterium tuberculosis infection. Nat. Commun. 2020;11:2270. doi: 10.1038/s41467-020-16143-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Xia M, et al. The ameliorative effect of fluoxetine on neuroinflammation induced by sleep deprivation. J. Neurochem. 2018;146:63–75. doi: 10.1111/jnc.14272. [DOI] [PubMed] [Google Scholar]
- 156.Jeon SA, et al. NLRP3 inflammasome contributes to lipopolysaccharide-induced depressive-like behaviors via indoleamine 2,3-dioxygenase induction. Int. J. Neuropsychopharmacol. 2017;20:896–906. doi: 10.1093/ijnp/pyx065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Wong ML, et al. Inflammasome signaling affects anxiety- and depressive-like behavior and gut microbiome composition. Mol. Psychiatry. 2016;21:797–805. doi: 10.1038/mp.2016.46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Page CE, Coutellier L. Prefrontal excitatory/inhibitory balance in stress and emotional disorders: Evidence for over-inhibition. Neurosci. Biobehav. Rev. 2019;105:39–51. doi: 10.1016/j.neubiorev.2019.07.024. [DOI] [PubMed] [Google Scholar]
- 159.Santello M, Toni N, Volterra A. Astrocyte function from information processing to cognition and cognitive impairment. Nat. Neurosci. 2019;22:154–166. doi: 10.1038/s41593-018-0325-8. [DOI] [PubMed] [Google Scholar]
- 160.Castillo-Gomez E, Perez-Rando M, Vidueira S, Nacher J. Polysialic acid acute depletion induces structural plasticity in interneurons and impairs the excitation/inhibition balance in medial prefrontal cortex organotypic cultures. Front. Cell. Neurosci. 2016;10:170. doi: 10.3389/fncel.2016.00170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Haydon PG, Carmignoto G. Astrocyte control of synaptic transmission and neurovascular coupling. Physiol. Rev. 2006;86:1009–1031. doi: 10.1152/physrev.00049.2005. [DOI] [PubMed] [Google Scholar]
- 162.Huang D, et al. Dysfunction of astrocytic connexins 30 and 43 in the medial prefrontal cortex and hippocampus mediates depressive-like behaviours. Behav. Brain Res. 2019;372:111950. doi: 10.1016/j.bbr.2019.111950. [DOI] [PubMed] [Google Scholar]
- 163.Nagy C, Torres-Platas SG, Mechawar N, Turecki G. Repression of astrocytic connexins in cortical and subcortical brain regions and prefrontal enrichment of H3K9me3 in depression and suicide. Int J. Neuropsychopharmacol. 2017;20:50–57. doi: 10.1093/ijnp/pyw071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Murphy-Royal C, et al. Stress gates an astrocytic energy reservoir to impair synaptic plasticity. Nat. Commun. 2020;11:2014. doi: 10.1038/s41467-020-15778-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ezan P, et al. Deletion of astroglial connexins weakens the blood-brain barrier. J. Cereb. Blood Flow Metab. 2012;32:1457–1467. doi: 10.1038/jcbfm.2012.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Torres-Platas SG, Nagy C, Wakid M, Turecki G, Mechawar N. Glial fibrillary acidic protein is differentially expressed across cortical and subcortical regions in healthy brains and downregulated in the thalamus and caudate nucleus of depressed suicides. Mol. Psychiatry. 2016;21:509–515. doi: 10.1038/mp.2015.65. [DOI] [PubMed] [Google Scholar]
- 167.Cobb JA, et al. Density of GFAP-immunoreactive astrocytes is decreased in left hippocampi in major depressive disorder. Neuroscience. 2016;316:209–220. doi: 10.1016/j.neuroscience.2015.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Steinacker P, et al. Glial fibrillary acidic protein as blood biomarker for differential diagnosis and severity of major depressive disorder. J. Psychiatr. Res. 2021;144:54–58. doi: 10.1016/j.jpsychires.2021.09.012. [DOI] [PubMed] [Google Scholar]
- 169.Rajkowska G, Hughes J, Stockmeier CA, Javier Miguel-Hidalgo J, Maciag D. Coverage of blood vessels by astrocytic endfeet is reduced in major depressive disorder. Biol. Psychiatry. 2013;73:613–621. doi: 10.1016/j.biopsych.2012.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Rajkowska G, Miguel-Hidalgo JJ. Glial pathology in major depressive disorder: an approach to investigate the coverage of blood vessels by astrocyte endfeet in human postmortem brain. Methods Mol. Biol. 2019;1938:247–254. doi: 10.1007/978-1-4939-9068-9_17. [DOI] [PubMed] [Google Scholar]
- 171.Kong H, et al. Aquaporin-4 knockout exacerbates corticosterone-induced depression by inhibiting astrocyte function and hippocampal neurogenesis. CNS Neurosci. Ther. 2014;20:391–402. doi: 10.1111/cns.12222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Xia M, Yang L, Sun G, Qi S, Li B. Mechanism of depression as a risk factor in the development of Alzheimer’s disease: the function of AQP4 and the glymphatic system. Psychopharmacology. 2017;234:365–379. doi: 10.1007/s00213-016-4473-9. [DOI] [PubMed] [Google Scholar]
- 173.Di Benedetto B, et al. Fluoxetine requires the endfeet protein aquaporin-4 to enhance plasticity of astrocyte processes. Front. Cell. Neurosci. 2016;10:8. doi: 10.3389/fncel.2016.00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Du J, et al. S100B is selectively expressed by gray matter protoplasmic astrocytes and myelinating oligodendrocytes in the developing CNS. Mol. Brain. 2021;14:154. doi: 10.1186/s13041-021-00865-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Schroeter ML, Abdul-Khaliq H, Krebs M, Diefenbacher A, Blasig IE. Serum markers support disease-specific glial pathology in major depression. J. Affect Disord. 2008;111:271–280. doi: 10.1016/j.jad.2008.03.005. [DOI] [PubMed] [Google Scholar]
- 176.Michel M, et al. Increased GFAP concentrations in the cerebrospinal fluid of patients with unipolar depression. Transl. Psychiatry. 2021;11:308. doi: 10.1038/s41398-021-01423-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Gos T, et al. S100B-immunopositive astrocytes and oligodendrocytes in the hippocampus are differentially afflicted in unipolar and bipolar depression: a postmortem study. J. Psychiatr. Res. 2013;47:1694–1699. doi: 10.1016/j.jpsychires.2013.07.005. [DOI] [PubMed] [Google Scholar]
- 178.Michetti F, et al. The S100B protein in biological fluids: more than a lifelong biomarker of brain distress. J. Neurochem. 2012;120:644–659. doi: 10.1111/j.1471-4159.2011.07612.x. [DOI] [PubMed] [Google Scholar]
- 179.Zhang L, et al. Changes in glial gene expression in the prefrontal cortex in relation to major depressive disorder, suicide and psychotic features. J. Affect Disord. 2021;295:893–903. doi: 10.1016/j.jad.2021.08.098. [DOI] [PubMed] [Google Scholar]
- 180.Gules E, Iosifescu DV, Tural U. Plasma neuronal and glial markers and anterior cingulate metabolite levels in major depressive disorder: a pilot study. Neuropsychobiology. 2020;79:214–221. doi: 10.1159/000505782. [DOI] [PubMed] [Google Scholar]
- 181.Chamera K, Szuster-Gluszczak M, Trojan E, Basta-Kaim A. Maternal Immune Activation Sensitizes Male Offspring Rats to Lipopolysaccharide-Induced Microglial Deficits Involving the Dysfunction of CD200-CD200R and CX3CL1-CX3CR1 systems. Cells. 2020;9:1676. doi: 10.3390/cells9071676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Jiang X, et al. Asperosaponin VI ameliorates the CMS-induced depressive-like behaviors by inducing a neuroprotective microglial phenotype in hippocampus via PPAR-gamma pathway. J. Neuroinflammation. 2022;19:115. doi: 10.1186/s12974-022-02478-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Tang MM, Lin WJ, Pan YQ, Li YC. Fibroblast growth factor 2 modulates hippocampal microglia activation in a neuroinflammation induced model of depression. Front. Cell. Neurosci. 2018;12:255. doi: 10.3389/fncel.2018.00255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Zhan Y, et al. Deficient neuron-microglia signaling results in impaired functional brain connectivity and social behavior. Nat. Neurosci. 2014;17:400–406. doi: 10.1038/nn.3641. [DOI] [PubMed] [Google Scholar]
- 185.Hellwig S, et al. Altered microglia morphology and higher resilience to stress-induced depression-like behavior in CX3CR1-deficient mice. Brain Behav. Immun. 2016;55:126–137. doi: 10.1016/j.bbi.2015.11.008. [DOI] [PubMed] [Google Scholar]
- 186.Merendino RA, et al. Involvement of fractalkine and macrophage inflammatory protein-1 alpha in moderate-severe depression. Mediat. Inflamm. 2004;13:205–207. doi: 10.1080/09511920410001713484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Garcia-Marchena N, et al. Inflammatory mediators and dual depression: Potential biomarkers in plasma of primary and substance-induced major depression in cocaine and alcohol use disorders. PLoS ONE. 2019;14:e0213791. doi: 10.1371/journal.pone.0213791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Wang HT, et al. Early-Life Social Isolation-Induced Depressive-Like Behavior in Rats Results in Microglial Activation and Neuronal Histone Methylation that Are Mitigated by Minocycline. Neurotox. Res. 2017;31:505–520. doi: 10.1007/s12640-016-9696-3. [DOI] [PubMed] [Google Scholar]
- 189.Frank MG, Fonken LK, Annis JL, Watkins LR, Maier SF. Stress disinhibits microglia via down-regulation of CD200R: A mechanism of neuroinflammatory priming. Brain Behav. Immun. 2018;69:62–73. doi: 10.1016/j.bbi.2017.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Fonken LK, et al. Neuroinflammatory priming to stress is differentially regulated in male and female rats. Brain Behav. Immun. 2018;70:257–267. doi: 10.1016/j.bbi.2018.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Popov A, et al. Astrocyte dystrophy in ageing brain parallels impaired synaptic plasticity. Aging Cell. 2021;20:e13334. doi: 10.1111/acel.13334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Zhang Y, et al. Structural basis of ketamine action on human NMDA receptors. Nature. 2021;596:301–305. doi: 10.1038/s41586-021-03769-9. [DOI] [PubMed] [Google Scholar]
- 193.Wang Q, Jie W, Liu JH, Yang JM, Gao TM. An astroglial basis of major depressive disorder? An overview. Glia. 2017;65:1227–1250. doi: 10.1002/glia.23143. [DOI] [PubMed] [Google Scholar]
- 194.Jiang X, Lin W, Cheng Y, Wang D. mGluR5 facilitates long-term synaptic depression in a stress-induced depressive mouse model. Can. J. Psychiatry. 2020;65:347–355. doi: 10.1177/0706743719874162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Guo W, et al. Iptakalim alleviates synaptic damages via targeting mitochondrial ATP-sensitive potassium channel in depression. FASEB J. 2021;35:e21581. doi: 10.1096/fj.202100124RR. [DOI] [PubMed] [Google Scholar]
- 196.Curro D, Navarra P, Samengo I, Martire M. P2X7 receptors exert a permissive effect on the activation of presynaptic AMPA receptors in rat trigeminal caudal nucleus glutamatergic nerve terminals. J. Headache Pain. 2020;21:83. doi: 10.1186/s10194-020-01153-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Iwata M, et al. Psychological stress activates the inflammasome via release of adenosine triphosphate and stimulation of the purinergic type 2X7 receptor. Biol. Psychiatry. 2016;80:12–22. doi: 10.1016/j.biopsych.2015.11.026. [DOI] [PubMed] [Google Scholar]
- 198.He Y, Taylor N, Fourgeaud L, Bhattacharya A. The role of microglial P2X7: modulation of cell death and cytokine release. J. Neuroinflammation. 2017;14:135. doi: 10.1186/s12974-017-0904-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Ota KT, et al. REDD1 is essential for stress-induced synaptic loss and depressive behavior. Nat. Med. 2014;20:531–535. doi: 10.1038/nm.3513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Kang HJ, et al. Decreased expression of synapse-related genes and loss of synapses in major depressive disorder. Nat. Med. 2012;18:1413–1417. doi: 10.1038/nm.2886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Tomassoni-Ardori F, et al. Rbfox1 up-regulation impairs BDNF-dependent hippocampal LTP by dysregulating TrkB isoform expression levels. Elife. 2019;8:e49673. doi: 10.7554/eLife.49673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Wang G, et al. Antidepressant-like effect of ginsenoside Rb1 on potentiating synaptic plasticity via the miR-134-mediated BDNF signaling pathway in a mouse model of chronic stress-induced depression. J. Ginseng Res. 2022;46:376–386. doi: 10.1016/j.jgr.2021.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Lalo U, Koh W, Lee CJ, Pankratov Y. The tripartite glutamatergic synapse. Neuropharmacology. 2021;199:108758. doi: 10.1016/j.neuropharm.2021.108758. [DOI] [PubMed] [Google Scholar]
- 204.Shepard R, Page CE, Coutellier L. Sensitivity of the prefrontal GABAergic system to chronic stress in male and female mice: Relevance for sex differences in stress-related disorders. Neuroscience. 2016;332:1–12. doi: 10.1016/j.neuroscience.2016.06.038. [DOI] [PubMed] [Google Scholar]
- 205.Shepard R, Coutellier L. Changes in the prefrontal glutamatergic and parvalbumin systems of mice exposed to unpredictable chronic stress. Mol. Neurobiol. 2018;55:2591–2602. doi: 10.1007/s12035-017-0528-0. [DOI] [PubMed] [Google Scholar]
- 206.Bouamrane L, et al. Reelin-haploinsufficiency disrupts the developmental trajectory of the E/I Balance in the Prefrontal Cortex. Front. Cell. Neurosci. 2016;10:308. doi: 10.3389/fncel.2016.00308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Arnone D, Mumuni AN, Jauhar S, Condon B, Cavanagh J. Indirect evidence of selective glial involvement in glutamate-based mechanisms of mood regulation in depression: meta-analysis of absolute prefrontal neuro-metabolic concentrations. Eur. Neuropsychopharmacol. 2015;25:1109–1117. doi: 10.1016/j.euroneuro.2015.04.016. [DOI] [PubMed] [Google Scholar]
- 208.Hu YT, Tan ZL, Hirjak D, Northoff G. Brain-wide changes in excitation-inhibition balance of major depressive disorder: a systematic review of topographic patterns of GABA- and glutamatergic alterations. Mol. Psychiatry. 2023;28:3257–3266. doi: 10.1038/s41380-023-02193-x. [DOI] [PubMed] [Google Scholar]
- 209.Zhao J, et al. Prefrontal alterations in GABAergic and glutamatergic gene expression in relation to depression and suicide. J. Psychiatr. Res. 2018;102:261–274. doi: 10.1016/j.jpsychires.2018.04.020. [DOI] [PubMed] [Google Scholar]
- 210.Douillard-Guilloux G, Lewis D, Seney ML, Sibille E. Decrease in somatostatin-positive cell density in the amygdala of females with major depression. Depress Anxiety. 2017;34:68–78. doi: 10.1002/da.22549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Scifo E, et al. Sustained molecular pathology across episodes and remission in major depressive disorder. Biol. Psychiatry. 2018;83:81–89. doi: 10.1016/j.biopsych.2017.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Oh DH, Son H, Hwang S, Kim SH. Neuropathological abnormalities of astrocytes, GABAergic neurons, and pyramidal neurons in the dorsolateral prefrontal cortices of patients with major depressive disorder. Eur. Neuropsychopharmacol. 2012;22:330–338. doi: 10.1016/j.euroneuro.2011.09.001. [DOI] [PubMed] [Google Scholar]
- 213.Sibille E, Morris HM, Kota RS, Lewis DA. GABA-related transcripts in the dorsolateral prefrontal cortex in mood disorders. Int J. Neuropsychopharmacol. 2011;14:721–734. doi: 10.1017/S1461145710001616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Barros-Barbosa AR, Lobo MG, Ferreirinha F, Correia-de-Sa P, Cordeiro JM. P2X7 receptor activation downmodulates Na(+)-dependent high-affinity GABA and glutamate transport into rat brain cortex synaptosomes. Neuroscience. 2015;306:74–90. doi: 10.1016/j.neuroscience.2015.08.026. [DOI] [PubMed] [Google Scholar]
- 215.Barros-Barbosa AR, et al. Up-regulation of P2X7 receptor-mediated inhibition of GABA uptake by nerve terminals of the human epileptic neocortex. Epilepsia. 2016;57:99–110. doi: 10.1111/epi.13263. [DOI] [PubMed] [Google Scholar]
- 216.Liu YP, Yang CS, Chen MC, Sun SH, Tzeng SF. Ca(2+)-dependent reduction of glutamate aspartate transporter GLAST expression in astrocytes by P2X(7) receptor-mediated phosphoinositide 3-kinase signaling. J. Neurochem. 2010;113:213–227. doi: 10.1111/j.1471-4159.2010.06589.x. [DOI] [PubMed] [Google Scholar]
- 217.Macaulay N, Zeuthen T. Glial K(+) clearance and cell swelling: key roles for cotransporters and pumps. Neurochem. Res. 2012;37:2299–2309. doi: 10.1007/s11064-012-0731-3. [DOI] [PubMed] [Google Scholar]
- 218.Larsen BR, et al. Contributions of the Na(+)/K(+)-ATPase, NKCC1, and Kir4.1 to hippocampal K(+) clearance and volume responses. Glia. 2014;62:608–622. doi: 10.1002/glia.22629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Verkhratsky A, Nedergaard M. Astroglial cradle in the life of the synapse. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2014;369:20130595. doi: 10.1098/rstb.2013.0595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Aramideh JA, Vidal-Itriago A, Morsch M, Graeber MB. Cytokine signalling at the microglial penta-partite synapse. Int J. Mol. Sci. 2021;22:13186. doi: 10.3390/ijms222413186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Chung WS, et al. Astrocytes mediate synapse elimination through MEGF10 and MERTK pathways. Nature. 2013;504:394–400. doi: 10.1038/nature12776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Yang J, et al. Astrocytes contribute to synapse elimination via type 2 inositol 1,4,5-trisphosphate receptor-dependent release of ATP. Elife. 2016;5:e15043. doi: 10.7554/eLife.15043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Petrache AL, et al. Aberrant excitatory-inhibitory synaptic mechanisms in entorhinal cortex microcircuits during the pathogenesis of Alzheimer’s disease. Cereb. Cortex. 2019;29:1834–1850. doi: 10.1093/cercor/bhz016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Arioz BI, et al. Melatonin attenuates LPS-Induced acute depressive-like behaviors and microglial NLRP3 inflammasome activation through the SIRT1/Nrf2 pathway. Front. Immunol. 2019;10:1511. doi: 10.3389/fimmu.2019.01511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Bikbaev A, Frischknecht R, Heine M. Brain extracellular matrix retains connectivity in neuronal networks. Sci. Rep. 2015;5:14527. doi: 10.1038/srep14527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Strackeljan L, et al. Microglia depletion-induced remodeling of extracellular matrix and excitatory synapses in the hippocampus of adult mice. Cells. 2021;10:1862. doi: 10.3390/cells10081862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Leclercq S, De Saeger C, Delzenne N, de Timary P, Starkel P. Role of inflammatory pathways, blood mononuclear cells, and gut-derived bacterial products in alcohol dependence. Biol. Psychiatry. 2014;76:725–733. doi: 10.1016/j.biopsych.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 228.Hsu JH, Chien IC, Lin CH. Increased risk of chronic liver disease in patients with major depressive disorder: a population-based study. J. Affect Disord. 2019;251:180–185. doi: 10.1016/j.jad.2019.03.070. [DOI] [PubMed] [Google Scholar]
- 229.Chen, D., Zhang, Y., Huang, T. & Jia, J. Depression and risk of gastrointestinal disorders: a comprehensive two-sample Mendelian randomization study of European ancestry. Psychol. Med. 53, 7309–7321 (2023). [DOI] [PubMed]
- 230.Facanali CBG, et al. The relationship of major depressive disorder with Crohn’s disease activity. Clinics (Sao Paulo) 2023;78:100188. doi: 10.1016/j.clinsp.2023.100188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Ronaldson PT, Davis TP. Regulation of blood-brain barrier integrity by microglia in health and disease: A therapeutic opportunity. J. Cereb. Blood Flow. Metab. 2020;40:S6–S24. doi: 10.1177/0271678X20951995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Qing H, et al. Origin and Function of Stress-Induced IL-6 in Murine Models. Cell. 2020;182:1660. doi: 10.1016/j.cell.2020.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Labad J, et al. Hypothalamic-pituitary-adrenal axis function and exposure to stress factors and cannabis use in recent-onset psychosis. World J. Biol. Psychiatry. 2020;21:564–571. doi: 10.1080/15622975.2019.1628301. [DOI] [PubMed] [Google Scholar]
- 234.Frank MG, Miguel ZD, Watkins LR, Maier SF. Prior exposure to glucocorticoids sensitizes the neuroinflammatory and peripheral inflammatory responses to E. coli lipopolysaccharide. Brain Behav. Immun. 2010;24:19–30. doi: 10.1016/j.bbi.2009.07.008. [DOI] [PubMed] [Google Scholar]
- 235.Deng Y, et al. Involvement of the microbiota-gut-brain axis in chronic restraint stress: disturbances of the kynurenine metabolic pathway in both the gut and brain. Gut Microbes. 2021;13:1–16. doi: 10.1080/19490976.2020.1869501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Munn DH, Mellor AL. Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol. 2013;34:137–143. doi: 10.1016/j.it.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Gos T, et al. Reduced microglial immunoreactivity for endogenous NMDA receptor agonist quinolinic acid in the hippocampus of schizophrenia patients. Brain Behav. Immun. 2014;41:59–64. doi: 10.1016/j.bbi.2014.05.012. [DOI] [PubMed] [Google Scholar]
- 238.Li M, et al. Transcriptome profiles of corticosterone-induced cytotoxicity reveals the involvement of neurite growth-related genes in depression. Psychiatry Res. 2019;276:79–86. doi: 10.1016/j.psychres.2019.04.017. [DOI] [PubMed] [Google Scholar]
- 239.Dutheil S, Ota KT, Wohleb ES, Rasmussen K, Duman RS. High-fat diet induced anxiety and anhedonia: impact on brain homeostasis and inflammation. Neuropsychopharmacology. 2016;41:1874–1887. doi: 10.1038/npp.2015.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Skonieczna-Zydecka K, et al. Faecal short chain fatty acids profile is changed in polish depressive women. Nutrients. 2018;10:1939. doi: 10.3390/nu10121939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Sampson TR, et al. Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell. 2016;167:1469–1480.e1412. doi: 10.1016/j.cell.2016.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Fan L, et al. Total glycosides from stems of Cistanche tubulosa alleviate depression-like behaviors: bidirectional interaction of the phytochemicals and gut microbiota. Phytomedicine. 2021;83:153471. doi: 10.1016/j.phymed.2021.153471. [DOI] [PubMed] [Google Scholar]
- 243.Zhang JC, et al. Blockade of interleukin-6 receptor in the periphery promotes rapid and sustained antidepressant actions: a possible role of gut-microbiota-brain axis. Transl. Psychiatry. 2017;7:e1138. doi: 10.1038/tp.2017.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Pan Y, Chen XY, Zhang QY, Kong LD. Microglial NLRP3 inflammasome activation mediates IL-1beta-related inflammation in prefrontal cortex of depressive rats. Brain Behav. Immun. 2014;41:90–100. doi: 10.1016/j.bbi.2014.04.007. [DOI] [PubMed] [Google Scholar]
- 245.Woodburn SC, Bollinger JL, Wohleb ES. Synaptic and behavioral effects of chronic stress are linked to dynamic and sex-specific changes in microglia function and astrocyte dystrophy. Neurobiol. Stress. 2021;14:100312. doi: 10.1016/j.ynstr.2021.100312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Zhang K, Liu R, Gao Y, Ma W, Shen W. Electroacupuncture relieves LPS-induced depression-like behaviour in rats through IDO-Mediated tryptophan-degrading pathway. Neuropsychiatr. Dis. Treat. 2020;16:2257–2266. doi: 10.2147/NDT.S274778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Parrott JM, Redus L, O’Connor JC. Kynurenine metabolic balance is disrupted in the hippocampus following peripheral lipopolysaccharide challenge. J. Neuroinflammation. 2016;13:124. doi: 10.1186/s12974-016-0590-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Alzarea S, Abbas M, Ronan PJ, Lutfy K, Rahman S. The Effect of an alpha-7 nicotinic allosteric modulator PNU120596 and NMDA receptor antagonist memantine on depressive-like behavior induced by lps in mice: the involvement of brain microglia. Brain Sci. 2022;12:1493. doi: 10.3390/brainsci12111493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Le Strat Y, Le Foll B, Dubertret C. Major depression and suicide attempts in patients with liver disease in the United States. Liver Int. 2015;35:1910–1916. doi: 10.1111/liv.12612. [DOI] [PubMed] [Google Scholar]
- 250.Buganza-Torio E, et al. Depression in cirrhosis - a prospective evaluation of the prevalence, predictors and development of a screening nomogram. Aliment. Pharmacol. Ther. 2019;49:194–201. doi: 10.1111/apt.15068. [DOI] [PubMed] [Google Scholar]
- 251.Qin XH, et al. Liver soluble epoxide hydrolase regulates behavioral and cellular effects of chronic stress. Cell Rep. 2019;29:3223–3234.e3226. doi: 10.1016/j.celrep.2019.11.006. [DOI] [PubMed] [Google Scholar]
- 252.Pang F, et al. Electroacupuncture alleviates depressive-like behavior by modulating the expression of P2X7/NLRP3/IL-1beta of prefrontal cortex and liver in rats exposed to chronic unpredictable mild stress. Brain Sci. 2023;13:436. doi: 10.3390/brainsci13030436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Zhang Y, et al. Effects of hydrogen-rich water on depressive-like behavior in mice. Sci. Rep. 2016;6:23742. doi: 10.1038/srep23742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Brown, S. J. et al. Sex- and suicide-specific alterations in the kynurenine pathway in the anterior cingulate cortex in major depression. Neuropsychopharmacology (2023). Online ahead of print. [DOI] [PMC free article] [PubMed]
- 255.Guillemin GJ, et al. Characterisation of kynurenine pathway metabolism in human astrocytes and implications in neuropathogenesis. Redox Rep. 2000;5:108–111. doi: 10.1179/135100000101535375. [DOI] [PubMed] [Google Scholar]
- 256.Park HJ, Kim SA, Kang WS, Kim JW. Early-life stress modulates gut microbiota and peripheral and central inflammation in a sex-dependent manner. Int J. Mol. Sci. 2021;22:1899. doi: 10.3390/ijms22041899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Ninan J, Feldman L. Ammonia levels and hepatic encephalopathy in patients with known chronic liver disease. J. Hosp. Med. 2017;12:659–661. doi: 10.12788/jhm.2794. [DOI] [PubMed] [Google Scholar]
- 258.Kautzky A, et al. The influence of the rs6295 gene polymorphism on serotonin-1A receptor distribution investigated with PET in patients with major depression applying machine learning. Transl. Psychiatry. 2017;7:e1150. doi: 10.1038/tp.2017.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Das R, et al. Higher levels of serum IL-1beta and TNF-alpha are associated with an increased probability of major depressive disorder. Psychiatry Res. 2021;295:113568. doi: 10.1016/j.psychres.2020.113568. [DOI] [PubMed] [Google Scholar]
- 260.Lu Z, et al. Oxidative stress and psychiatric disorders: evidence from the bidirectional mendelian randomization study. Antioxidants (Basel) 2022;11:1386. doi: 10.3390/antiox11071386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261.Hursitoglu O, et al. Serum NOX1 and Raftlin as new potential biomarkers of Major Depressive Disorder: a study in treatment-naive first episode patients. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2023;121:110670. doi: 10.1016/j.pnpbp.2022.110670. [DOI] [PubMed] [Google Scholar]
- 262.Han KM, et al. Serum FAM19A5 levels: A novel biomarker for neuroinflammation and neurodegeneration in major depressive disorder. Brain Behav. Immun. 2020;87:852–859. doi: 10.1016/j.bbi.2020.03.021. [DOI] [PubMed] [Google Scholar]
- 263.Winter NR, et al. Quantifying deviations of brain structure and function in major depressive disorder across neuroimaging modalities. JAMA Psychiatry. 2022;79:879–888. doi: 10.1001/jamapsychiatry.2022.1780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Zhao Y, et al. Gray matter abnormalities in non-comorbid medication-naive patients with major depressive disorder or social anxiety disorder. EBioMedicine. 2017;21:228–235. doi: 10.1016/j.ebiom.2017.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Wise T, et al. Common and distinct patterns of grey-matter volume alteration in major depression and bipolar disorder: evidence from voxel-based meta-analysis. Mol. Psychiatry. 2017;22:1455–1463. doi: 10.1038/mp.2016.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Liu Y, et al. Abnormal brain gray matter volume in patients with major depressive disorder: Associated with childhood trauma? J. Affect Disord. 2022;308:562–568. doi: 10.1016/j.jad.2022.04.083. [DOI] [PubMed] [Google Scholar]
- 267.Kaiser RH, Andrews-Hanna JR, Wager TD, Pizzagalli DA. Large-scale network dysfunction in major depressive disorder: a meta-analysis of resting-state functional connectivity. JAMA Psychiatry. 2015;72:603–611. doi: 10.1001/jamapsychiatry.2015.0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Wang Y, et al. Topologically convergent and divergent functional connectivity patterns in unmedicated unipolar depression and bipolar disorder. Transl. Psychiatry. 2017;7:e1165. doi: 10.1038/tp.2017.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Kremneva EI, Sinitsyn DO, Dobrynina LA, Suslina AD, Krotenkova MV. [Resting state functional MRI in neurology and psychiatry] Zh. Nevrol. Psikhiatr Im. S S Korsakova. 2022;122:5–14. doi: 10.17116/jnevro20221220215. [DOI] [PubMed] [Google Scholar]
- 270.Luo Z, et al. Shared and specific dynamics of brain segregation and integration in bipolar disorder and major depressive disorder: A resting-state functional magnetic resonance imaging study. J. Affect. Disord. 2021;280:279–286. doi: 10.1016/j.jad.2020.11.012. [DOI] [PubMed] [Google Scholar]
- 271.Drysdale AT, et al. Resting-state connectivity biomarkers define neurophysiological subtypes of depression. Nat. Med. 2017;23:28–38. doi: 10.1038/nm.4246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Chen Q, et al. Regional amplitude abnormities in the major depressive disorder: A resting-state fMRI study and support vector machine analysis. J. Affect. Disord. 2022;308:1–9. doi: 10.1016/j.jad.2022.03.079. [DOI] [PubMed] [Google Scholar]
- 273.Yan CG, et al. Reduced default mode network functional connectivity in patients with recurrent major depressive disorder. Proc. Natl Acad. Sci. USA. 2019;116:9078–9083. doi: 10.1073/pnas.1900390116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Pizzagalli DA, Roberts AC. Prefrontal cortex and depression. Neuropsychopharmacology. 2022;47:225–246. doi: 10.1038/s41386-021-01101-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Muller VI, et al. Altered brain activity in unipolar depression revisited: meta-analyses of neuroimaging studies. JAMA Psychiatry. 2017;74:47–55. doi: 10.1001/jamapsychiatry.2016.2783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Sun K, et al. A two-center radiomic analysis for differentiating major depressive disorder using multi-modality MRI data under different parcellation methods. J. Affect. Disord. 2022;300:1–9. doi: 10.1016/j.jad.2021.12.065. [DOI] [PubMed] [Google Scholar]
- 277.Mayerhoefer ME, et al. Introduction to radiomics. J. Nucl. Med. 2020;61:488–495. doi: 10.2967/jnumed.118.222893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Xu Z, et al. Combined HTR1A/1B methylation and human functional connectome to recognize patients with MDD. Psychiatry Res. 2022;317:114842. doi: 10.1016/j.psychres.2022.114842. [DOI] [PubMed] [Google Scholar]
- 279.Verduijn J, et al. Reconsidering the prognosis of major depressive disorder across diagnostic boundaries: full recovery is the exception rather than the rule. BMC Med. 2017;15:215. doi: 10.1186/s12916-017-0972-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Blumenthal JA, Rozanski A. Exercise as a therapeutic modality for the prevention and treatment of depression. Prog. Cardiovasc. Dis. 2023;77:50–58. doi: 10.1016/j.pcad.2023.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Baglioni C, Spiegelhalder K, Nissen C, Riemann D. Clinical implications of the causal relationship between insomnia and depression: how individually tailored treatment of sleeping difficulties could prevent the onset of depression. EPMA J. 2011;2:287–293. doi: 10.1007/s13167-011-0079-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Riemann D, et al. European guideline for the diagnosis and treatment of insomnia. J. Sleep. Res. 2017;26:675–700. doi: 10.1111/jsr.12594. [DOI] [PubMed] [Google Scholar]
- 283.Soh HL, Ho RC, Ho CS, Tam WW. Efficacy of digital cognitive behavioural therapy for insomnia: a meta-analysis of randomised controlled trials. Sleep. Med. 2020;75:315–325. doi: 10.1016/j.sleep.2020.08.020. [DOI] [PubMed] [Google Scholar]
- 284.Ho FY, Chan CS, Lo WY, Leung JC. The effect of self-help cognitive behavioral therapy for insomnia on depressive symptoms: an updated meta-analysis of randomized controlled trials. J. Affect. Disord. 2020;265:287–304. doi: 10.1016/j.jad.2020.01.062. [DOI] [PubMed] [Google Scholar]
- 285.Qaseem A, et al. Management of chronic insomnia disorder in adults: a clinical practice guideline from the american college of physicians. Ann. Intern. Med. 2016;165:125–133. doi: 10.7326/M15-2175. [DOI] [PubMed] [Google Scholar]
- 286.Irwin MR, et al. Prevention of incident and recurrent major depression in older adults with insomnia: a randomized clinical trial. JAMA Psychiatry. 2022;79:33–41. doi: 10.1001/jamapsychiatry.2021.3422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Scheer FA, Pirovano C, Van Someren EJ, Buijs RM. Environmental light and suprachiasmatic nucleus interact in the regulation of body temperature. Neuroscience. 2005;132:465–477. doi: 10.1016/j.neuroscience.2004.12.012. [DOI] [PubMed] [Google Scholar]
- 288.Dekker K, et al. Combined internet-based cognitive-behavioral and chronobiological intervention for insomnia: a randomized controlled trial. Psychother. Psychosom. 2020;89:117–118. doi: 10.1159/000503570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Leerssen J, et al. Treating insomnia with high risk of depression using therapist-guided digital cognitive, behavioral, and circadian rhythm support interventions to prevent worsening of depressive symptoms: a randomized controlled trial. Psychother. Psychosom. 2022;91:168–179. doi: 10.1159/000520282. [DOI] [PubMed] [Google Scholar]
- 290.Zhai L, Zhang H, Zhang D. Sleep duration and depression among adults: a meta-analysis of prospective studies. Depress. Anxiety. 2015;32:664–670. doi: 10.1002/da.22386. [DOI] [PubMed] [Google Scholar]
- 291.Baglioni C, et al. Insomnia as a predictor of depression: a meta-analytic evaluation of longitudinal epidemiological studies. J. Affect. Disord. 2011;135:10–19. doi: 10.1016/j.jad.2011.01.011. [DOI] [PubMed] [Google Scholar]
- 292.Utge SJ, et al. Systematic analysis of circadian genes in a population-based sample reveals association of TIMELESS with depression and sleep disturbance. PLoS ONE. 2010;5:e9259. doi: 10.1371/journal.pone.0009259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Irwin MR. Sleep and inflammation: partners in sickness and in health. Nat. Rev. Immunol. 2019;19:702–715. doi: 10.1038/s41577-019-0190-z. [DOI] [PubMed] [Google Scholar]
- 294.Hill Almeida LM, et al. Disrupted sleep and risk of depression in later life: A prospective cohort study with extended follow up and a systematic review and meta-analysis. J. Affect. Disord. 2022;309:314–323. doi: 10.1016/j.jad.2022.04.133. [DOI] [PubMed] [Google Scholar]
- 295.Tolkien K, Bradburn S, Murgatroyd C. An anti-inflammatory diet as a potential intervention for depressive disorders: A systematic review and meta-analysis. Clin. Nutr. 2019;38:2045–2052. doi: 10.1016/j.clnu.2018.11.007. [DOI] [PubMed] [Google Scholar]
- 296.Galland L. Diet and inflammation. Nutr. Clin. Pr. 2010;25:634–640. doi: 10.1177/0884533610385703. [DOI] [PubMed] [Google Scholar]
- 297.Oddy WH, et al. Dietary patterns, body mass index and inflammation: Pathways to depression and mental health problems in adolescents. Brain Behav. Immun. 2018;69:428–439. doi: 10.1016/j.bbi.2018.01.002. [DOI] [PubMed] [Google Scholar]
- 298.Bosma-den Boer MM, van Wetten ML, Pruimboom L. Chronic inflammatory diseases are stimulated by current lifestyle: how diet, stress levels and medication prevent our body from recovering. Nutr. Metab. (Lond.) 2012;9:32. doi: 10.1186/1743-7075-9-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Sanchez-Villegas A, et al. The effect of the Mediterranean diet on plasma brain-derived neurotrophic factor (BDNF) levels: the PREDIMED-NAVARRA randomized trial. Nutr. Neurosci. 2011;14:195–201. doi: 10.1179/1476830511Y.0000000011. [DOI] [PubMed] [Google Scholar]
- 300.Jiang C, Salton SR. The role of neurotrophins in major depressive disorder. Transl. Neurosci. 2013;4:46–58. doi: 10.2478/s13380-013-0103-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Ogbonnaya ES, et al. Adult hippocampal neurogenesis is regulated by the microbiome. Biol. Psychiatry. 2015;78:e7–9. doi: 10.1016/j.biopsych.2014.12.023. [DOI] [PubMed] [Google Scholar]
- 302.Zheng P, et al. Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host’s metabolism. Mol. Psychiatry. 2016;21:786–796. doi: 10.1038/mp.2016.44. [DOI] [PubMed] [Google Scholar]
- 303.Jiang H, et al. Altered fecal microbiota composition in patients with major depressive disorder. Brain Behav. Immun. 2015;48:186–194. doi: 10.1016/j.bbi.2015.03.016. [DOI] [PubMed] [Google Scholar]
- 304.Burokas A, et al. Targeting the microbiota-gut-brain axis: prebiotics have anxiolytic and antidepressant-like effects and reverse the impact of chronic stress in mice. Biol. Psychiatry. 2017;82:472–487. doi: 10.1016/j.biopsych.2016.12.031. [DOI] [PubMed] [Google Scholar]
- 305.Bonder MJ, et al. The influence of a short-term gluten-free diet on the human gut microbiome. Genome Med. 2016;8:45. doi: 10.1186/s13073-016-0295-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Mohan M, et al. Dietary gluten-induced gut dysbiosis is accompanied by selective upregulation of microRNAs with intestinal tight junction and bacteria-binding motifs in rhesus macaque model of celiac disease. Nutrients. 2016;8:684. doi: 10.3390/nu8110684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Karakula-Juchnowicz H, et al. The study evaluating the effect of probiotic supplementation on the mental status, inflammation, and intestinal barrier in major depressive disorder patients using gluten-free or gluten-containing diet (SANGUT study): a 12-week, randomized, double-blind, and placebo-controlled clinical study protocol. Nutr. J. 2019;18:50. doi: 10.1186/s12937-019-0475-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Khosravi M, et al. The relationship between dietary patterns and depression mediated by serum levels of Folate and vitamin B12. BMC Psychiatry. 2020;20:63. doi: 10.1186/s12888-020-2455-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Schuch FB, et al. Exercise as a treatment for depression: a meta-analysis adjusting for publication bias. J. Psychiatr. Res. 2016;77:42–51. doi: 10.1016/j.jpsychires.2016.02.023. [DOI] [PubMed] [Google Scholar]
- 310.Morland C, et al. Exercise induces cerebral VEGF and angiogenesis via the lactate receptor HCAR1. Nat. Commun. 2017;8:15557. doi: 10.1038/ncomms15557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Coelho FG, et al. Physical exercise modulates peripheral levels of brain-derived neurotrophic factor (BDNF): a systematic review of experimental studies in the elderly. Arch. Gerontol. Geriatr. 2013;56:10–15. doi: 10.1016/j.archger.2012.06.003. [DOI] [PubMed] [Google Scholar]
- 312.Stimpson NJ, Davison G, Javadi AH. Joggin’ the noggin: towards a physiological understanding of exercise-induced cognitive benefits. Neurosci. Biobehav Rev. 2018;88:177–186. doi: 10.1016/j.neubiorev.2018.03.018. [DOI] [PubMed] [Google Scholar]
- 313.De Rossi P, et al. A critical role for VEGF and VEGFR2 in NMDA receptor synaptic function and fear-related behavior. Mol. Psychiatry. 2016;21:1768–1780. doi: 10.1038/mp.2015.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Bugg JM, Head D. Exercise moderates age-related atrophy of the medial temporal lobe. Neurobiol. Aging. 2011;32:506–514. doi: 10.1016/j.neurobiolaging.2009.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Cole RC, et al. Cardiorespiratory fitness and hippocampal volume predict faster episodic associative learning in older adults. Hippocampus. 2020;30:143–155. doi: 10.1002/hipo.23151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Zotcheva E, et al. Associations of changes in cardiorespiratory fitness and symptoms of anxiety and depression with brain volumes: The HUNT Study. Front Behav. Neurosci. 2019;13:53. doi: 10.3389/fnbeh.2019.00053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Dowlati Y, et al. A meta-analysis of cytokines in major depression. Biol. Psychiatry. 2010;67:446–457. doi: 10.1016/j.biopsych.2009.09.033. [DOI] [PubMed] [Google Scholar]
- 318.Valkanova V, Ebmeier KP, Allan CL. CRP, IL-6 and depression: a systematic review and meta-analysis of longitudinal studies. J. Affect Disord. 2013;150:736–744. doi: 10.1016/j.jad.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 319.Kohler CA, et al. Peripheral cytokine and chemokine alterations in depression: a meta-analysis of 82 studies. Acta Psychiatr. Scand. 2017;135:373–387. doi: 10.1111/acps.12698. [DOI] [PubMed] [Google Scholar]
- 320.Choi KW, et al. Assessment of bidirectional relationships between physical activity and depression among adults: a 2-sample mendelian randomization study. JAMA Psychiatry. 2019;76:399–408. doi: 10.1001/jamapsychiatry.2018.4175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 321.Schuch FB, et al. Physical activity and incident depression: a meta-analysis of prospective cohort studies. Am. J. Psychiatry. 2018;175:631–648. doi: 10.1176/appi.ajp.2018.17111194. [DOI] [PubMed] [Google Scholar]
- 322.Choi KW, et al. Physical activity offsets genetic risk for incident depression assessed via electronic health records in a biobank cohort study. Depress Anxiety. 2020;37:106–114. doi: 10.1002/da.22967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Imboden C, et al. Aerobic exercise or stretching as add-on to inpatient treatment of depression: Similar antidepressant effects on depressive symptoms and larger effects on working memory for aerobic exercise alone. J. Affect Disord. 2020;276:866–876. doi: 10.1016/j.jad.2020.07.052. [DOI] [PubMed] [Google Scholar]
- 324.Vancampfort D, et al. Sedentary behavior and physical activity levels in people with schizophrenia, bipolar disorder and major depressive disorder: a global systematic review and meta-analysis. World Psychiatry. 2017;16:308–315. doi: 10.1002/wps.20458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Brush CJ, et al. A randomized trial of aerobic exercise for major depression: examining neural indicators of reward and cognitive control as predictors and treatment targets. Psychol. Med. 2022;52:893–903. doi: 10.1017/S0033291720002573. [DOI] [PubMed] [Google Scholar]
- 326.Kujawa A, et al. Reduced reward responsiveness predicts change in depressive symptoms in anxious children and adolescents following treatment. J. Child Adolesc. Psychopharmacol. 2019;29:378–385. doi: 10.1089/cap.2018.0172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Kandola A, Ashdown-Franks G, Hendrikse J, Sabiston CM, Stubbs B. Physical activity and depression: towards understanding the antidepressant mechanisms of physical activity. Neurosci. Biobehav Rev. 2019;107:525–539. doi: 10.1016/j.neubiorev.2019.09.040. [DOI] [PubMed] [Google Scholar]
- 328.Yildirim Y, Kocabiyik S. The relationship between social support and loneliness in Turkish patients with cancer. J. Clin. Nurs. 2010;19:832–839. doi: 10.1111/j.1365-2702.2009.03066.x. [DOI] [PubMed] [Google Scholar]
- 329.Lin J, et al. Perceived stressfulness mediates the effects of subjective social support and negative coping style on suicide risk in Chinese patients with major depressive disorder. J. Affect. Disord. 2020;265:32–38. doi: 10.1016/j.jad.2020.01.026. [DOI] [PubMed] [Google Scholar]
- 330.Smith L, Hill N, Kokanovic R. Experiences of depression, the role of social support and its impact on health outcomes. J. Ment. Health. 2015;24:342–346. doi: 10.3109/09638237.2014.954693. [DOI] [PubMed] [Google Scholar]
- 331.Wang X, Cai L, Qian J, Peng J. Social support moderates stress effects on depression. Int. J. Ment. Health Syst. 2014;8:41. doi: 10.1186/1752-4458-8-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Hybels CF, Pieper CF, Blazer DG, Steffens DC. Heterogeneity in the three-year course of major depression among older adults. Int. J. Geriatr. Psychiatry. 2016;31:775–782. doi: 10.1002/gps.4391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 333.Backs-Dermott BJ, Dobson KS, Jones SL. An evaluation of an integrated model of relapse in depression. J. Affect. Disord. 2010;124:60–67. doi: 10.1016/j.jad.2009.11.015. [DOI] [PubMed] [Google Scholar]
- 334.Holma IA, Holma KM, Melartin TK, Rytsala HJ, Isometsa ET. A 5-year prospective study of predictors for disability pension among patients with major depressive disorder. Acta Psychiatr. Scand. 2012;125:325–334. doi: 10.1111/j.1600-0447.2011.01785.x. [DOI] [PubMed] [Google Scholar]
- 335.Stewart RA, Patel TA, McDermott KA, Cougle JR. Functional and structural social support in DSM-5 mood and anxiety disorders: a population-based study. J. Affect Disord. 2022;308:528–534. doi: 10.1016/j.jad.2022.04.026. [DOI] [PubMed] [Google Scholar]
- 336.Ouakinin SRS, Barreira DP, Gois CJ. Depression and obesity: integrating the role of stress, neuroendocrine dysfunction and inflammatory pathways. Front. Endocrinol. (Lausanne) 2018;9:431. doi: 10.3389/fendo.2018.00431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Hallgren M, Lundin A, Tee FY, Burstrom B, Forsell Y. Somebody to lean on: Social relationships predict post-treatment depression severity in adults. Psychiatry Res. 2017;249:261–267. doi: 10.1016/j.psychres.2016.12.060. [DOI] [PubMed] [Google Scholar]
- 338.van Beljouw IM, Verhaak PF, Cuijpers P, van Marwijk HW, Penninx BW. The course of untreated anxiety and depression, and determinants of poor one-year outcome: a one-year cohort study. BMC Psychiatry. 2010;10:86. doi: 10.1186/1471-244X-10-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 339.Dour HJ, et al. Perceived social support mediates anxiety and depressive symptom changes following primary care intervention. Depress Anxiety. 2014;31:436–442. doi: 10.1002/da.22216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 340.Teo AR, Choi H, Valenstein M. Social relationships and depression: ten-year follow-up from a nationally representative study. PLoS ONE. 2013;8:e62396. doi: 10.1371/journal.pone.0062396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Griffiths KM, Crisp DA, Barney L, Reid R. Seeking help for depression from family and friends: a qualitative analysis of perceived advantages and disadvantages. BMC Psychiatry. 2011;11:196. doi: 10.1186/1471-244X-11-196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 342.Hillhouse TM, Porter JH. A brief history of the development of antidepressant drugs: from monoamines to glutamate. Exp. Clin. Psychopharmacol. 2015;23:1–21. doi: 10.1037/a0038550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Sulser F, Vetulani J, Mobley PL. Mode of action of antidepressant drugs. Biochem. Pharmacol. 1978;27:257–261. doi: 10.1016/0006-2952(78)90226-5. [DOI] [PubMed] [Google Scholar]
- 344.Gillman PK. Tricyclic antidepressant pharmacology and therapeutic drug interactions updated. Br. J. Pharmacol. 2007;151:737–748. doi: 10.1038/sj.bjp.0707253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Taylor C, Fricker AD, Devi LA, Gomes I. Mechanisms of action of antidepressants: from neurotransmitter systems to signaling pathways. Cell Signal. 2005;17:549–557. doi: 10.1016/j.cellsig.2004.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 346.Jann MW, Slade JH. Antidepressant agents for the treatment of chronic pain and depression. Pharmacotherapy. 2007;27:1571–1587. doi: 10.1592/phco.27.11.1571. [DOI] [PubMed] [Google Scholar]
- 347.Shultz E, Malone DA., Jr A practical approach to prescribing antidepressants. Cleve Clin. J. Med. 2013;80:625–631. doi: 10.3949/ccjm.80a.12133. [DOI] [PubMed] [Google Scholar]
- 348.Croom KF, Perry CM, Plosker GL. Mirtazapine: a review of its use in major depression and other psychiatric disorders. CNS Drugs. 2009;23:427–452. doi: 10.2165/00023210-200923050-00006. [DOI] [PubMed] [Google Scholar]
- 349.Benjamin S, Doraiswamy PM. Review of the use of mirtazapine in the treatment of depression. Expert Opin. Pharmacother. 2011;12:1623–1632. doi: 10.1517/14656566.2011.585459. [DOI] [PubMed] [Google Scholar]
- 350.Cleare A, et al. Evidence-based guidelines for treating depressive disorders with antidepressants: a revision of the 2008 British Association for Psychopharmacology guidelines. J. Psychopharmacol. 2015;29:459–525. doi: 10.1177/0269881115581093. [DOI] [PubMed] [Google Scholar]
- 351.Guloglu C, Orak M, Ustundag M, Altunci YA. Analysis of amitriptyline overdose in emergency medicine. Emerg. Med. J. 2011;28:296–299. doi: 10.1136/emj.2009.076596. [DOI] [PubMed] [Google Scholar]
- 352.David DJ, Gourion D. [Antidepressant and tolerance: determinants and management of major side effects] Encephale. 2016;42:553–561. doi: 10.1016/j.encep.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 353.Richelson E. Antimuscarinic and other receptor-blocking properties of antidepressants. Mayo Clin. Proc. 1983;58:40–46. [PubMed] [Google Scholar]
- 354.Hisaoka K, et al. Tricyclic antidepressant amitriptyline activates fibroblast growth factor receptor signaling in glial cells: involvement in glial cell line-derived neurotrophic factor production. J. Biol. Chem. 2011;286:21118–21128. doi: 10.1074/jbc.M111.224683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 355.Morioka N, et al. Amitriptyline up-regulates connexin43-gap junction in rat cultured cortical astrocytes via activation of the p38 and c-Fos/AP-1 signalling pathway. Br. J. Pharmacol. 2014;171:2854–2867. doi: 10.1111/bph.12614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Budzinski ML, et al. Tricyclic antidepressants target FKBP51 SUMOylation to restore glucocorticoid receptor activity. Mol. Psychiatry. 2022;27:2533–2545. doi: 10.1038/s41380-022-01491-0. [DOI] [PubMed] [Google Scholar]
- 357.Malison RT, et al. Reduced brain serotonin transporter availability in major depression as measured by [123I]-2 beta-carbomethoxy-3 beta-(4-iodophenyl)tropane and single photon emission computed tomography. Biol. Psychiatry. 1998;44:1090–1098. doi: 10.1016/S0006-3223(98)00272-8. [DOI] [PubMed] [Google Scholar]
- 358.Samuels BA, et al. 5-HT1A receptors on mature dentate gyrus granule cells are critical for the antidepressant response. Nat. Neurosci. 2015;18:1606–1616. doi: 10.1038/nn.4116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Scorza MC, et al. Preclinical and clinical characterization of the selective 5-HT(1A) receptor antagonist DU-125530 for antidepressant treatment. Br. J. Pharmacol. 2012;167:1021–1034. doi: 10.1111/j.1476-5381.2011.01770.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Cremers TI, et al. Inactivation of 5-HT(2C) receptors potentiates consequences of serotonin reuptake blockade. Neuropsychopharmacology. 2004;29:1782–1789. doi: 10.1038/sj.npp.1300474. [DOI] [PubMed] [Google Scholar]
- 361.Boothman LJ, Mitchell SN, Sharp T. Investigation of the SSRI augmentation properties of 5-HT(2) receptor antagonists using in vivo microdialysis. Neuropharmacology. 2006;50:726–732. doi: 10.1016/j.neuropharm.2005.11.020. [DOI] [PubMed] [Google Scholar]
- 362.Haddjeri N, de Montigny C, Blier P. Modulation of the firing activity of noradrenergic neurones in the rat locus coeruleus by the 5-hydroxtryptamine system. Br. J. Pharmacol. 1997;120:865–875. doi: 10.1038/sj.bjp.0700968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Szabo ST, de Montigny C, Blier P. Modulation of noradrenergic neuronal firing by selective serotonin reuptake blockers. Br. J. Pharmacol. 1999;126:568–571. doi: 10.1038/sj.bjp.0702343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 364.Jang SW, et al. Amitriptyline is a TrkA and TrkB receptor agonist that promotes TrkA/TrkB heterodimerization and has potent neurotrophic activity. Chem. Biol. 2009;16:644–656. doi: 10.1016/j.chembiol.2009.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 365.Casarotto PC, et al. Antidepressant drugs act by directly binding to TRKB neurotrophin receptors. Cell. 2021;184:1299–1313.e1219. doi: 10.1016/j.cell.2021.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Li B, et al. Down-regulation of GluK2 kainate receptor expression by chronic treatment with mood-stabilizing anti-convulsants or lithium in cultured astrocytes and brain, but not in neurons. Neuropharmacology. 2009;57:375–385. doi: 10.1016/j.neuropharm.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 367.Diaz SL, Narboux-Neme N, Boutourlinsky K, Doly S, Maroteaux L. Mice lacking the serotonin 5-HT2B receptor as an animal model of resistance to selective serotonin reuptake inhibitors antidepressants. Eur. Neuropsychopharmacol. 2016;26:265–279. doi: 10.1016/j.euroneuro.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 368.Sviridova A, et al. The role of 5-HT(2B)-receptors in fluoxetine-mediated modulation of Th17- and Th1-cells in multiple sclerosis. J. Neuroimmunol. 2021;356:577608. doi: 10.1016/j.jneuroim.2021.577608. [DOI] [PubMed] [Google Scholar]
- 369.Hertz L. Isotope-based quantitation of uptake, release, and metabolism of glutamate and glucose in cultured astrocytes. Methods Mol. Biol. 2012;814:305–323. doi: 10.1007/978-1-61779-452-0_20. [DOI] [PubMed] [Google Scholar]
- 370.Arakawa R, et al. Venlafaxine ER blocks the norepinephrine transporter in the brain of patients with major depressive disorder: a PET study using [18F]FMeNER-D2. Int J. Neuropsychopharmacol. 2019;22:278–285. doi: 10.1093/ijnp/pyz003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 371.Gould GG, Javors MA, Frazer A. Effect of chronic administration of duloxetine on serotonin and norepinephrine transporter binding sites in rat brain. Biol. Psychiatry. 2007;61:210–215. doi: 10.1016/j.biopsych.2006.02.029. [DOI] [PubMed] [Google Scholar]
- 372.Owens MJ, et al. Estimates of serotonin and norepinephrine transporter inhibition in depressed patients treated with paroxetine or venlafaxine. Neuropsychopharmacology. 2008;33:3201–3212. doi: 10.1038/npp.2008.47. [DOI] [PubMed] [Google Scholar]
- 373.Sramek JJ, et al. Exploratory biomarker study of the triple reuptake inhibitor sep-432 compared to the dual reuptake inhibitor duloxetine in healthy normal subjects. CNS Neurosci. Ther. 2016;22:404–412. doi: 10.1111/cns.12513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.Wang Y, et al. The association between antidepressant treatment and brain connectivity in two double-blind, placebo-controlled clinical trials: a treatment mechanism study. Lancet Psychiatry. 2019;6:667–674. doi: 10.1016/S2215-0366(19)30179-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Berman RM, et al. Antidepressant effects of ketamine in depressed patients. Biol. Psychiatry. 2000;47:351–354. doi: 10.1016/S0006-3223(99)00230-9. [DOI] [PubMed] [Google Scholar]
- 376.Murrough JW, et al. Antidepressant efficacy of ketamine in treatment-resistant major depression: a two-site randomized controlled trial. Am. J. Psychiatry. 2013;170:1134–1142. doi: 10.1176/appi.ajp.2013.13030392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Zanos P, et al. NMDAR inhibition-independent antidepressant actions of ketamine metabolites. Nature. 2016;533:481–486. doi: 10.1038/nature17998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 378.Li N, et al. mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science. 2010;329:959–964. doi: 10.1126/science.1190287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Li N, et al. Glutamate N-methyl-D-aspartate receptor antagonists rapidly reverse behavioral and synaptic deficits caused by chronic stress exposure. Biol. Psychiatry. 2011;69:754–761. doi: 10.1016/j.biopsych.2010.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 380.Beurel E, Grieco SF, Amadei C, Downey K, Jope RS. Ketamine-induced inhibition of glycogen synthase kinase-3 contributes to the augmentation of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid (AMPA) receptor signaling. Bipolar Disord. 2016;18:473–480. doi: 10.1111/bdi.12436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Miller OH, Moran JT, Hall BJ. Two cellular hypotheses explaining the initiation of ketamine’s antidepressant actions: direct inhibition and disinhibition. Neuropharmacology. 2016;100:17–26. doi: 10.1016/j.neuropharm.2015.07.028. [DOI] [PubMed] [Google Scholar]
- 382.Mingardi J, et al. miR-9-5p is involved in the rescue of stress-dependent dendritic shortening of hippocampal pyramidal neurons induced by acute antidepressant treatment with ketamine. Neurobiol. Stress. 2021;15:100381. doi: 10.1016/j.ynstr.2021.100381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 383.Krystal JH, Charney DS, Duman RS. A new rapid-acting antidepressant. Cell. 2020;181:7. doi: 10.1016/j.cell.2020.02.033. [DOI] [PubMed] [Google Scholar]
- 384.Moliner R, et al. Psychedelics promote plasticity by directly binding to BDNF receptor TrkB. Nat. Neurosci. 2023;26:1032–1041. doi: 10.1038/s41593-023-01316-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 385.Halberstadt AL, Koedood L, Powell SB, Geyer MA. Differential contributions of serotonin receptors to the behavioral effects of indoleamine hallucinogens in mice. J. Psychopharmacol. 2011;25:1548–1561. doi: 10.1177/0269881110388326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 386.Hesselgrave N, Troppoli TA, Wulff AB, Cole AB, Thompson SM. Harnessing psilocybin: antidepressant-like behavioral and synaptic actions of psilocybin are independent of 5-HT2R activation in mice. Proc. Natl Acad. Sci. USA. 2021;118:e2022489118. doi: 10.1073/pnas.2022489118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 387.Cao D, et al. Structure-based discovery of nonhallucinogenic psychedelic analogs. Science. 2022;375:403–411. doi: 10.1126/science.abl8615. [DOI] [PubMed] [Google Scholar]
- 388.Shao LX, et al. Psilocybin induces rapid and persistent growth of dendritic spines in frontal cortex in vivo. Neuron. 2021;109:2535–2544 e2534. doi: 10.1016/j.neuron.2021.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 389.Caraci F, et al. Neurobiological links between depression and AD: The role of TGF-beta1 signaling as a new pharmacological target. Pharmacol. Res. 2018;130:374–384. doi: 10.1016/j.phrs.2018.02.007. [DOI] [PubMed] [Google Scholar]
- 390.Chen B, Wang JF, Sun X, Young LT. Regulation of GAP-43 expression by chronic desipramine treatment in rat cultured hippocampal cells. Biol. Psychiatry. 2003;53:530–537. doi: 10.1016/S0006-3223(02)01551-2. [DOI] [PubMed] [Google Scholar]
- 391.Lee KM, Kim YK. The role of IL-12 and TGF-beta1 in the pathophysiology of major depressive disorder. Int. Immunopharmacol. 2006;6:1298–1304. doi: 10.1016/j.intimp.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 392.Sutcigil L, et al. Pro- and anti-inflammatory cytokine balance in major depression: effect of sertraline therapy. Clin. Dev. Immunol. 2007;2007:76396. doi: 10.1155/2007/76396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Zhao YN, et al. Nelumbo nucifera gaertn stems (Hegeng) improved depression Behavior in CUMS mice by regulating NCAM and GAP-43 expression. Evid. Based Complement Altern. Med. 2020;2020:3056954. doi: 10.1155/2020/3056954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Zavvari F, Nahavandi A, Goudarzi M. Fluoxetine attenuates stress-induced depressive-like behavior through modulation of hippocampal GAP43 and neurogenesis in male rats. J. Chem. Neuroanat. 2020;103:101711. doi: 10.1016/j.jchemneu.2019.101711. [DOI] [PubMed] [Google Scholar]
- 395.Wang Q, Timberlake MA, 2nd, Prall K, Dwivedi Y. The recent progress in animal models of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2017;77:99–109. doi: 10.1016/j.pnpbp.2017.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Aten S, et al. Chronic stress impairs the structure and function of astrocyte networks in an animal model of depression. Neurochem. Res. 2023;48:1191–1210. doi: 10.1007/s11064-022-03663-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Liang S, et al. Iron aggravates the depressive phenotype of stressed mice by compromising the glymphatic system. Neurosci. Bull. 2020;36:1542–1546. doi: 10.1007/s12264-020-00539-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Antoniuk S, Bijata M, Ponimaskin E, Wlodarczyk J. Chronic unpredictable mild stress for modeling depression in rodents: meta-analysis of model reliability. Neurosci. Biobehav Rev. 2019;99:101–116. doi: 10.1016/j.neubiorev.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 399.Fernandez DC, et al. Light affects mood and learning through distinct retina-brain pathways. Cell. 2018;175:71–84.e18. doi: 10.1016/j.cell.2018.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 400.Smolders KC, de Kort YA, Cluitmans PJ. A higher illuminance induces alertness even during office hours: findings on subjective measures, task performance and heart rate measures. Physiol. Behav. 2012;107:7–16. doi: 10.1016/j.physbeh.2012.04.028. [DOI] [PubMed] [Google Scholar]
- 401.Liu A, et al. Encoding of environmental illumination by primate melanopsin neurons. Science. 2023;379:376–381. doi: 10.1126/science.ade2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 402.Lam RW, et al. Efficacy of bright light treatment, fluoxetine, and the combination in patients with nonseasonal major depressive disorder: a randomized clinical trial. JAMA Psychiatry. 2016;73:56–63. doi: 10.1001/jamapsychiatry.2015.2235. [DOI] [PubMed] [Google Scholar]
- 403.Rutten S, et al. Bright light therapy for depression in Parkinson disease: a randomized controlled trial. Neurology. 2019;92:e1145–e1156. doi: 10.1212/WNL.0000000000007090. [DOI] [PubMed] [Google Scholar]
- 404.Li VW, et al. Functional outcomes with bright light in monotherapy and combined with fluoxetine in patients with major depressive disorder: results from the LIFE-D trial. J. Affect. Disord. 2022;297:396–400. doi: 10.1016/j.jad.2021.10.035. [DOI] [PubMed] [Google Scholar]
- 405.Guzel Ozdemir P, et al. Comparison of venlafaxine alone versus venlafaxine plus bright light therapy combination for severe major depressive disorder. J. Clin. Psychiatry. 2015;76:e645–654. doi: 10.4088/JCP.14m09376. [DOI] [PubMed] [Google Scholar]
- 406.Lambert GW, Reid C, Kaye DM, Jennings GL, Esler MD. Effect of sunlight and season on serotonin turnover in the brain. Lancet. 2002;360:1840–1842. doi: 10.1016/S0140-6736(02)11737-5. [DOI] [PubMed] [Google Scholar]
- 407.Huang L, et al. A visual circuit related to habenula underlies the antidepressive effects of light therapy. Neuron. 2019;102:128–142.e128. doi: 10.1016/j.neuron.2019.01.037. [DOI] [PubMed] [Google Scholar]
- 408.McClintock SM, et al. Consensus recommendations for the clinical application of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression. J. Clin. Psychiatry. 2018;79:16cs10905. doi: 10.4088/JCP.16cs10905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Wall CA, et al. Adjunctive use of repetitive transcranial magnetic stimulation in depressed adolescents: a prospective, open pilot study. J. Clin. Psychiatry. 2011;72:1263–1269. doi: 10.4088/JCP.11m07003. [DOI] [PubMed] [Google Scholar]
- 410.Qiu H, et al. Efficacy and safety of repetitive transcranial magnetic stimulation in children and adolescents with depression: a systematic review and preliminary meta-analysis. J. Affect. Disord. 2023;320:305–312. doi: 10.1016/j.jad.2022.09.060. [DOI] [PubMed] [Google Scholar]
- 411.Mutz J, et al. Comparative efficacy and acceptability of non-surgical brain stimulation for the acute treatment of major depressive episodes in adults: systematic review and network meta-analysis. BMJ. 2019;364:l1079. doi: 10.1136/bmj.l1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 412.Faller J, et al. Daily prefrontal closed-loop repetitive transcranial magnetic stimulation (rTMS) produces progressive EEG quasi-alpha phase entrainment in depressed adults. Brain Stimul. 2022;15:458–471. doi: 10.1016/j.brs.2022.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Kaster TS, et al. Efficacy, tolerability, and cognitive effects of deep transcranial magnetic stimulation for late-life depression: a prospective randomized controlled trial. Neuropsychopharmacology. 2018;43:2231–2238. doi: 10.1038/s41386-018-0121-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 414.Desbeaumes Jodoin V, Miron JP, Lesperance P. Safety and efficacy of accelerated repetitive transcranial magnetic stimulation protocol in elderly depressed unipolar and bipolar patients. Am. J. Geriatr. Psychiatry. 2019;27:548–558. doi: 10.1016/j.jagp.2018.10.019. [DOI] [PubMed] [Google Scholar]
- 415.Wathra RA, et al. Effect of prior pharmacotherapy on remission with sequential bilateral theta-burst versus standard bilateral repetitive transcranial magnetic stimulation in treatment-resistant late-life depression. Br. J. Psychiatry. 2023;223:504–506. doi: 10.1192/bjp.2023.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Cox EQ, et al. Repetitive transcranial magnetic stimulation for the treatment of postpartum depression. J. Affect. Disord. 2020;264:193–200. doi: 10.1016/j.jad.2019.11.069. [DOI] [PubMed] [Google Scholar]
- 417.Pascual-Leone A, Rubio B, Pallardo F, Catala MD. Rapid-rate transcranial magnetic stimulation of left dorsolateral prefrontal cortex in drug-resistant depression. Lancet. 1996;348:233–237. doi: 10.1016/S0140-6736(96)01219-6. [DOI] [PubMed] [Google Scholar]
- 418.Zrenner B, et al. Brain oscillation-synchronized stimulation of the left dorsolateral prefrontal cortex in depression using real-time EEG-triggered TMS. Brain Stimul. 2020;13:197–205. doi: 10.1016/j.brs.2019.10.007. [DOI] [PubMed] [Google Scholar]
- 419.Fitzgerald PB. Targeting repetitive transcranial magnetic stimulation in depression: do we really know what we are stimulating and how best to do it? Brain Stimul. 2021;14:730–736. doi: 10.1016/j.brs.2021.04.018. [DOI] [PubMed] [Google Scholar]
- 420.Rosen AC, et al. Targeting location relates to treatment response in active but not sham rTMS stimulation. Brain Stimul. 2021;14:703–709. doi: 10.1016/j.brs.2021.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 421.Moffitt TE, et al. How common are common mental disorders? Evidence that lifetime prevalence rates are doubled by prospective versus retrospective ascertainment. Psychol. Med. 2010;40:899–909. doi: 10.1017/S0033291709991036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 422.Brini S, et al. Efficacy and safety of transcranial magnetic stimulation for treating major depressive disorder: An umbrella review and re-analysis of published meta-analyses of randomised controlled trials. Clin. Psychol. Rev. 2023;100:102236. doi: 10.1016/j.cpr.2022.102236. [DOI] [PubMed] [Google Scholar]
- 423.Qaseem A, Barry MJ, Kansagara D, Clinical Guidelines Committee of the American College of Physicians Nonpharmacologic versus pharmacologic treatment of adult patients with major depressive disorder: a clinical practice guideline from the american college of physicians. Ann. Intern. Med. 2016;164:350–359. doi: 10.7326/M15-2570. [DOI] [PubMed] [Google Scholar]
- 424.Biesheuvel-Leliefeld KE, et al. Effectiveness of psychological interventions in preventing recurrence of depressive disorder: meta-analysis and meta-regression. J. Affect. Disord. 2015;174:400–410. doi: 10.1016/j.jad.2014.12.016. [DOI] [PubMed] [Google Scholar]
- 425.Breedvelt JJF, et al. Psychological interventions as an alternative and add-on to antidepressant medication to prevent depressive relapse: systematic review and meta-analysis. Br. J. Psychiatry. 2021;219:538–545. doi: 10.1192/bjp.2020.198. [DOI] [PubMed] [Google Scholar]
- 426.Guidi J, Fava GA. Sequential combination of pharmacotherapy and psychotherapy in major depressive disorder: a systematic review and meta-analysis. JAMA Psychiatry. 2021;78:261–269. doi: 10.1001/jamapsychiatry.2020.3650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 427.Tang Y, Yin HY, Rubini P, Illes P, Acupuncture-Induced Analgesia: a neurobiological basis in purinergic signaling. Neuroscientist. 2016;22:563–578. doi: 10.1177/1073858416654453. [DOI] [PubMed] [Google Scholar]
- 428.Chan YY, Lo WY, Yang SN, Chen YH, Lin JG. The benefit of combined acupuncture and antidepressant medication for depression: a systematic review and meta-analysis. J. Affect Disord. 2015;176:106–117. doi: 10.1016/j.jad.2015.01.048. [DOI] [PubMed] [Google Scholar]
- 429.Mischoulon D, Brill CD, Ameral VE, Fava M, Yeung AS. A pilot study of acupuncture monotherapy in patients with major depressive disorder. J. Affect Disord. 2012;141:469–473. doi: 10.1016/j.jad.2012.03.023. [DOI] [PubMed] [Google Scholar]
- 430.Yue N, et al. Electro-acupuncture alleviates chronic unpredictable stress-induced depressive- and anxiety-like behavior and hippocampal neuroinflammation in rat model of depression. Front. Mol. Neurosci. 2018;11:149. doi: 10.3389/fnmol.2018.00149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Jung J, et al. Lipidomics reveals that acupuncture modulates the lipid metabolism and inflammatory interaction in a mouse model of depression. Brain Behav. Immun. 2021;94:424–436. doi: 10.1016/j.bbi.2021.02.003. [DOI] [PubMed] [Google Scholar]
- 432.Lin SS, et al. Electroacupuncture prevents astrocyte atrophy to alleviate depression. Cell Death Dis. 2023;14:343. doi: 10.1038/s41419-023-05839-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 433.Wang Z, et al. Acupuncture treatment modulates the corticostriatal reward circuitry in major depressive disorder. J. Psychiatr. Res. 2017;84:18–26. doi: 10.1016/j.jpsychires.2016.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Duan DM, Tu Y, Liu P, Jiao S. Antidepressant effect of electroacupuncture regulates signal targeting in the brain and increases brain-derived neurotrophic factor levels. Neural Regen. Res. 2016;11:1595–1602. doi: 10.4103/1673-5374.193238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Bai L, et al. Mechanisms underlying the antidepressant effect of acupuncture via the CaMK signaling pathway. Front Behav. Neurosci. 2020;14:563698. doi: 10.3389/fnbeh.2020.563698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 436.Cai X, et al. Electroacupuncture alleviated depression-like behaviors in ventromedial prefrontal cortex of chronic unpredictable mild stress-induced rats: Increasing synaptic transmission and phosphorylating dopamine transporter. CNS Neurosci. Ther. 2023;29:2608–2620. doi: 10.1111/cns.14200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Xu G, et al. Clinical evidence for association of acupuncture with improved major depressive disorder: a systematic review and meta-analysis of randomized control trials. Neuropsychobiology. 2023;82:1–13. doi: 10.1159/000527903. [DOI] [PubMed] [Google Scholar]
- 438.Ding Y, et al. Molecular and genetic characterization of depression: overlap with other psychiatric disorders and aging. Mol. Neuropsychiatry. 2015;1:1–12. doi: 10.1159/000369974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 439.Kay RB, Brunjes PC. Diversity among principal and GABAergic neurons of the anterior olfactory nucleus. Front. Cell. Neurosci. 2014;8:111. doi: 10.3389/fncel.2014.00111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Karolewicz B, et al. Reduced level of glutamic acid decarboxylase-67 kDa in the prefrontal cortex in major depression. Int. J. Neuropsychopharmacol. 2010;13:411–420. doi: 10.1017/S1461145709990587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 441.Azmitia EC. Serotonin neurons, neuroplasticity, and homeostasis of neural tissue. Neuropsychopharmacology. 1999;21:33S–45S. doi: 10.1038/sj.npp.1395346. [DOI] [PubMed] [Google Scholar]
- 442.Kerman IA, et al. Evidence for transcriptional factor dysregulation in the dorsal raphe nucleus of patients with major depressive disorder. Front. Neurosci. 2012;6:135. doi: 10.3389/fnins.2012.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Keller-Wood M. Hypothalamic-pituitary–adrenal axis-feedback control. Compr. Physiol. 2015;5:1161–1182. doi: 10.1002/cphy.c140065. [DOI] [PubMed] [Google Scholar]
- 444.Alt SR, et al. Differential expression of glucocorticoid receptor transcripts in major depressive disorder is not epigenetically programmed. Psychoneuroendocrinology. 2010;35:544–556. doi: 10.1016/j.psyneuen.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 445.van Loo HM, Aggen SH, Gardner CO, Kendler KS. Sex similarities and differences in risk factors for recurrence of major depression. Psychol. Med. 2018;48:1685–1693. doi: 10.1017/S0033291717003178. [DOI] [PubMed] [Google Scholar]
- 446.Salk RH, Hyde JS, Abramson LY. Gender differences in depression in representative national samples: meta-analyses of diagnoses and symptoms. Psychol. Bull. 2017;143:783–822. doi: 10.1037/bul0000102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 447.Cavanagh A, Wilson CJ, Kavanagh DJ, Caputi P. Differences in the expression of symptoms in men versus women with depression: a systematic review and meta-analysis. Harv. Rev. Psychiatry. 2017;25:29–38. doi: 10.1097/HRP.0000000000000128. [DOI] [PubMed] [Google Scholar]
- 448.Marcus SM, et al. Gender differences in depression: findings from the STAR*D study. J. Affect Disord. 2005;87:141–150. doi: 10.1016/j.jad.2004.09.008. [DOI] [PubMed] [Google Scholar]
- 449.Poulter MO, et al. GABAA receptor promoter hypermethylation in suicide brain: implications for the involvement of epigenetic processes. Biol. Psychiatry. 2008;64:645–652. doi: 10.1016/j.biopsych.2008.05.028. [DOI] [PubMed] [Google Scholar]
- 450.Seney ML, et al. The role of genetic sex in affect regulation and expression of GABA-related genes across species. Front. Psychiatry. 2013;4:104. doi: 10.3389/fpsyt.2013.00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 451.Gray AL, Hyde TM, Deep-Soboslay A, Kleinman JE, Sodhi MS. Sex differences in glutamate receptor gene expression in major depression and suicide. Mol. Psychiatry. 2015;20:1057–1068. doi: 10.1038/mp.2015.91. [DOI] [PubMed] [Google Scholar]
- 452.Goswami DB, May WL, Stockmeier CA, Austin MC. Transcriptional expression of serotonergic regulators in laser-captured microdissected dorsal raphe neurons of subjects with major depressive disorder: sex-specific differences. J. Neurochem. 2010;112:397–409. doi: 10.1111/j.1471-4159.2009.06462.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 453.Szewczyk B, et al. Gender-specific decrease in NUDR and 5-HT1A receptor proteins in the prefrontal cortex of subjects with major depressive disorder. Int. J. Neuropsychopharmacol. 2009;12:155–168. doi: 10.1017/S1461145708009012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Fiori LM, et al. miR-323a regulates ERBB4 and is involved in depression. Mol. Psychiatry. 2021;26:4191–4204. doi: 10.1038/s41380-020-00953-7. [DOI] [PubMed] [Google Scholar]
- 455.Roy B, Wang Q, Palkovits M, Faludi G, Dwivedi Y. Altered miRNA expression network in locus coeruleus of depressed suicide subjects. Sci. Rep. 2017;7:4387. doi: 10.1038/s41598-017-04300-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 456.Zheng P, et al. Identification of sex-specific urinary biomarkers for major depressive disorder by combined application of NMR- and GC-MS-based metabonomics. Transl. Psychiatry. 2016;6:e955. doi: 10.1038/tp.2016.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 457.Liu X, et al. Discovery and validation of plasma biomarkers for major depressive disorder classification based on liquid chromatography-mass spectrometry. J. Proteome Res. 2015;14:2322–2330. doi: 10.1021/acs.jproteome.5b00144. [DOI] [PubMed] [Google Scholar]
- 458.Liu X, et al. Plasma lipidomics reveals potential lipid markers of major depressive disorder. Anal. Bioanal. Chem. 2016;408:6497–6507. doi: 10.1007/s00216-016-9768-5. [DOI] [PubMed] [Google Scholar]
- 459.Wang Z, Gerstein M, Snyder M. RNA-Seq: a revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009;10:57–63. doi: 10.1038/nrg2484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 460.Xu Y, et al. Serum cytokines-based biomarkers in the diagnosis and monitoring of therapeutic response in patients with major depressive disorder. Int. Immunopharmacol. 2023;118:110108. doi: 10.1016/j.intimp.2023.110108. [DOI] [PubMed] [Google Scholar]
- 461.Pantazatos SP, et al. Whole-transcriptome brain expression and exon-usage profiling in major depression and suicide: evidence for altered glial, endothelial and ATPase activity. Mol. Psychiatry. 2017;22:760–773. doi: 10.1038/mp.2016.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 462.Herbet M, et al. Altered expression of genes involved in brain energy metabolism as adaptive responses in rats exposed to chronic variable stress; changes in cortical level of glucogenic and neuroactive amino acids. Mol. Med. Rep. 2019;19:2386–2396. doi: 10.3892/mmr.2019.9865. [DOI] [PubMed] [Google Scholar]
- 463.Yamanishi K, et al. Analysis of genes linked to depressive-like behaviors in interleukin-18-deficient mice: gene expression profiles in the brain. Biomed. Rep. 2020;12:3–10. doi: 10.3892/br.2019.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 464.Kuperman Y, et al. Perifornical Urocortin-3 mediates the link between stress-induced anxiety and energy homeostasis. Proc. Natl Acad. Sci. USA. 2010;107:8393–8398. doi: 10.1073/pnas.1003969107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 465.Bustamante AC, Armstrong DL, Uddin M. Epigenetic profiles associated with major depression in the human brain. Psychiatry Res. 2018;260:439–442. doi: 10.1016/j.psychres.2017.12.010. [DOI] [PubMed] [Google Scholar]
- 466.Beger RD, et al. Metabolomics enables precision medicine: “A White Paper, Community Perspective”. Metabolomics. 2016;12:149. doi: 10.1007/s11306-016-1094-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 467.Kaddurah-Daouk R, et al. Cerebrospinal fluid metabolome in mood disorders-remission state has a unique metabolic profile. Sci. Rep. 2012;2:667. doi: 10.1038/srep00667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 468.Schmaal L, et al. Cortical abnormalities in adults and adolescents with major depression based on brain scans from 20 cohorts worldwide in the ENIGMA Major Depressive Disorder Working Group. Mol. Psychiatry. 2017;22:900–909. doi: 10.1038/mp.2016.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Schmitgen MM, et al. Aberrant cortical neurodevelopment in major depressive disorder. J. Affect. Disord. 2019;243:340–347. doi: 10.1016/j.jad.2018.09.021. [DOI] [PubMed] [Google Scholar]
- 470.Chen Z, et al. High-field magnetic resonance imaging of structural alterations in first-episode, drug-naive patients with major depressive disorder. Transl. Psychiatry. 2016;6:e942. doi: 10.1038/tp.2016.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 471.Grimm S, et al. Increased self-focus in major depressive disorder is related to neural abnormalities in subcortical-cortical midline structures. Hum. Brain Mapp. 2009;30:2617–2627. doi: 10.1002/hbm.20693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Yoshimura S, et al. Cognitive behavioral therapy for depression changes medial prefrontal and ventral anterior cingulate cortex activity associated with self-referential processing. Soc. Cogn. Affect. Neurosci. 2014;9:487–493. doi: 10.1093/scan/nst009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Yu H, et al. Functional brain abnormalities in major depressive disorder using the Hilbert-Huang transform. Brain Imaging Behav. 2018;12:1556–1568. doi: 10.1007/s11682-017-9816-6. [DOI] [PubMed] [Google Scholar]
- 474.Holmes SE, et al. Lower synaptic density is associated with depression severity and network alterations. Nat. Commun. 2019;10:1529. doi: 10.1038/s41467-019-09562-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Bettini E, et al. Pharmacological comparative characterization of REL-1017 (Esmethadone-HCl) and Other NMDAR channel blockers in human heterodimeric N-methyl-D-aspartate receptors. Pharmaceuticals (Basel) 2022;15:997. doi: 10.3390/ph15080997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 476.Hanania T, Manfredi P, Inturrisi C, Vitolo OV. The N-methyl-D-aspartate receptor antagonist d-methadone acutely improves depressive-like behavior in the forced swim test performance of rats. Exp. Clin. Psychopharmacol. 2020;28:196–201. doi: 10.1037/pha0000310. [DOI] [PubMed] [Google Scholar]
- 477.Fogaca MV, et al. N-Methyl-D-aspartate receptor antagonist d-methadone produces rapid, mTORC1-dependent antidepressant effects. Neuropsychopharmacology. 2019;44:2230–2238. doi: 10.1038/s41386-019-0501-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 478.Fava M, et al. REL-1017 (esmethadone) as adjunctive treatment in patients with major depressive disorder: a phase 2a randomized double-blind trial. Am. J. Psychiatry. 2022;179:122–131. doi: 10.1176/appi.ajp.2021.21020197. [DOI] [PubMed] [Google Scholar]
- 479.De Martin S, et al. REL-1017 (Esmethadone) increases circulating BDNF levels in healthy subjects of a phase 1 clinical study. Front. Pharmacol. 2021;12:671859. doi: 10.3389/fphar.2021.671859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Chou TH, Kang H, Simorowski N, Traynelis SF, Furukawa H. Structural insights into assembly and function of GluN1-2C, GluN1-2A-2C, and GluN1-2D NMDARs. Mol. Cell. 2022;82:4548–4563.e4544. doi: 10.1016/j.molcel.2022.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 481.Hochschild A, et al. Ketamine vs midazolam: Mood improvement reduces suicidal ideation in depression. J. Affect Disord. 2022;300:10–16. doi: 10.1016/j.jad.2021.12.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 482.Burgdorf J, et al. GLYX-13, a NMDA receptor glycine-site functional partial agonist, induces antidepressant-like effects without ketamine-like side effects. Neuropsychopharmacology. 2013;38:729–742. doi: 10.1038/npp.2012.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 483.Gigliucci V, et al. Ketamine elicits sustained antidepressant-like activity via a serotonin-dependent mechanism. Psychopharmacology (Berl.) 2013;228:157–166. doi: 10.1007/s00213-013-3024-x. [DOI] [PubMed] [Google Scholar]
- 484.Daly EJ, et al. Efficacy and safety of intranasal esketamine adjunctive to oral antidepressant therapy in treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2018;75:139–148. doi: 10.1001/jamapsychiatry.2017.3739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Zhao J, et al. Low-dose ketamine inhibits neuronal apoptosis and neuroinflammation in PC12 cells via alpha7nAChR mediated TLR4/MAPK/NF-kappaB signaling pathway. Int. Immunopharmacol. 2023;117:109880. doi: 10.1016/j.intimp.2023.109880. [DOI] [PubMed] [Google Scholar]
- 486.Yang Y, et al. Ketamine relieves depression-like behaviors induced by chronic postsurgical pain in rats through anti-inflammatory, anti-oxidant effects and regulating BDNF expression. Psychopharmacology (Berl.) 2020;237:1657–1669. doi: 10.1007/s00213-020-05490-3. [DOI] [PubMed] [Google Scholar]
- 487.Shibakawa YS, et al. Effects of ketamine and propofol on inflammatory responses of primary glial cell cultures stimulated with lipopolysaccharide. Br. J. Anaesth. 2005;95:803–810. doi: 10.1093/bja/aei256. [DOI] [PubMed] [Google Scholar]
- 488.Abbasi, S., Hosseini, F., Modabbernia, A., Ashrafi, M. & Akhondzadeh, S. Effect of celecoxib add-on treatment on symptoms and serum IL-6 concentrations in patients with major depressive disorder: randomized double-blind placebo-controlled study. J. Affect. Disord.141, 308–314 (2012). [DOI] [PubMed]
- 489.Akhondzadeh, S. et al. Clinical trial of adjunctive celecoxib treatment in patients with major depression: a double blind and placebo controlled trial. Depress Anxiety26, 607–611 (2009). [DOI] [PubMed]
- 490.Nettis, M. A. et al. Augmentation therapy with minocycline in treatment-resistant depression patients with low-grade peripheral inflammation: results from a double-blind randomised clinical trial. Neuropsychopharmacology46, 939–948 (2021). [DOI] [PMC free article] [PubMed]
- 491.Hasebe, K. et al. Exploring interleukin-6, lipopolysaccharide-binding protein and brain-derived neurotrophic factor following 12 weeks of adjunctive minocycline treatment for depression. Acta Neuropsychiatr.34, 220–227 (2022). [DOI] [PubMed]
- 492.Su, K. et al. Omega-3 fatty acids in the prevention of interferon-alpha-induced depression: results from arandomized, controlled trial. Biol. Psychiatry76, 559–566 (2014). [DOI] [PubMed]
- 493.Berk, M. et al. Youth Depression Alleviation with Anti-inflammatory Agents (YoDA-A): a randomised clinical trial of rosuvastatin and aspirin. BMC Med. 18, 16 (2020). [DOI] [PMC free article] [PubMed]
- 494.Meltzer-Brody, S. et al. Brexanolone injection in post-partum depression: two multicentre, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet392, 1058–1070 (2018). [DOI] [PubMed]
- 495.Leal, G. C. et al. Arketamine as adjunctive therapy for treatment-resistant depression: A placebo-controlled pilot study. J Affect. Disord.330, 7–15 (2023). [DOI] [PubMed]
- 496.Kuga N, Sasaki T, Takahara Y, Matsuki N, Ikegaya Y. Large-scale calcium waves traveling through astrocytic networks in vivo. J. Neurosci. 2011;31:2607–2614. doi: 10.1523/JNEUROSCI.5319-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 497.Shawcross DL, et al. Infection and systemic inflammation, not ammonia, are associated with Grade 3/4 hepatic encephalopathy, but not mortality in cirrhosis. J. Hepatol. 2011;54:640–649. doi: 10.1016/j.jhep.2010.07.045. [DOI] [PubMed] [Google Scholar]
- 498.Page G, et al. The up-regulation of the striatal dopamine transporter’s activity by cAMP is PKA-, CaMK II- and phosphatase-dependent. Neurochem. Int. 2004;45:627–632. doi: 10.1016/j.neuint.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 499.França AS, et al. D2 dopamine receptor regulation of learning, sleep and plasticity. Eur. Neuropsychopharmacol. 2015;25:493–504. doi: 10.1016/j.euroneuro.2015.01.011. [DOI] [PubMed] [Google Scholar]
- 500.Beaulieu JM, Gainetdinov RR. The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol. Rev. 2011;63:182–217. doi: 10.1124/pr.110.002642. [DOI] [PubMed] [Google Scholar]
- 501.Opal MD, et al. Serotonin 2C receptor antagonists induce fast-onset antidepressant effects. Mol. Psychiatry. 2014;19:1106–1114. doi: 10.1038/mp.2013.144. [DOI] [PubMed] [Google Scholar]
- 502.Seki K, Yoshida S, Jaiswal MK. Molecular mechanism of noradrenaline during the stress-induced major depressive disorder. Neural Regen. Res. 2018;13:1159–1169. doi: 10.4103/1673-5374.235019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 503.Duman RS, Aghajanian GK, Sanacora G, Krystal JH. Synaptic plasticity and depression: new insights from stress and rapid-acting antidepressants. Nat. Med. 2016;22:238–249. doi: 10.1038/nm.4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 504.Yang B, et al. Comparison of R-ketamine and rapastinel antidepressant effects in the social defeat stress model of depression. Psychopharmacology. 2016;233:3647–3657. doi: 10.1007/s00213-016-4399-2. [DOI] [PMC free article] [PubMed] [Google Scholar]