Abstract
Mosquitoes are the deadliest animal in the word, transmitting a variety of insect-borne infectious diseases, such as malaria, dengue fever, yellow fever, and Zika, causing more deaths than any other vector-borne pathogen. Moreover, in the absence of effective drugs and vaccines to prevent and treat insect-borne diseases, mosquito control is particularly important as the primary measure. In recent decades, due to the gradual increase in mosquito resistance, increasing attention has fallen on the mechanisms and effects associated with pathogen infection. This review provides an overview of mosquito innate immune mechanisms in terms of physical and physiological barriers, pattern recognition receptors, signalling pathways, and cellular and humoral immunity, as well as the antipathogenic effects of mosquito symbiotic bacteria. This review contributes to an in-depth understanding of the interaction process between mosquitoes and pathogens and provides a theoretical basis for biological defence strategies against mosquito-borne infectious diseases.
Graphical Abstract
Keywords: Mosquito, Pathogen infection, Innate immune system, Immune priming, Symbiotic bacteria
Background
As an important pathogen vector, mosquitoes transmit a variety of insect-borne infectious diseases, such as malaria, dengue fever, yellow fever, and Zika, causing more deaths than any other vector-borne pathogen [1]. As the world's number one “health killer”, mosquitoes pose a great threat to global public health. Pathogens enter the mosquito body mainly in the following two ways: bacteria and fungi easily enter the body through brakes in the cuticle or exoskeleton; viruses, Plasmodium, and other parasites easily enter the midgut through mosquitoes sucking blood that contains the pathogens and penetrate the midgut tissue barrier through haemolymph diffusion to reach other organs and tissues, such as the trachea, fat body, and salivary glands, which allows them to cross the salivary gland barrier, enter the saliva, and transmit to the next host through bites.
Infection with arboviruses induces an innate immune response in mosquitos that activates a complex mosquito-pathogen interaction, which in turn alters the gene expression profile of mosquitos. Similar to other arthropods [2], mosquitoes lack an acquired immune system and rely primarily on innate immunity to protect against viruses, bacteria, fungi, parasites, and other pathogens [3, 4]. The mosquito innate immune system modulates infection by human pathogens or resistance to insect pathogens and increases the fitness and longevity of infected mosquitoes [1]. According to studies, cell-mediated phagocytosis, melanisation, and lysis are the three main modes of pathogen clearance by mosquitoes [5]. Cellular and humoral immunity are involved in the regulation of pathogen infection in mosquitoes, first through the recognition and binding of pathogens by pattern recognition receptors (PRRs) and pathogen-associated molecular patterns (PAMPs) and then through the activation of immune-associated signalling pathways, thus triggering cellular and humoral immunity in mosquitoes. Cellular immunity defends against pathogen invasion through the circulation of insect peripheral haemolymph, with phagocytosis and encapsulation responses mediated by haemocytes and pericardial cells. Humoral immunity, which also occurs in the haemocoel, involves a variety of mechanisms, including the production of antimicrobial peptides (AMPs), the activation of melanization by the phenoloxidase (PO) cascade, and the production of reactive oxygen species (ROS) and nitric oxide (NO) [1, 3]. There is no clear boundary between the molecular pathways of action of cellular and humoral immunity, and some of the molecules that participate in humoral immunity are produced by haemocytes and participate in cellular immunity at the same time [6]. Although mosquitoes only have innate immunity, there is some evidence that they can induce an enhanced immunity when they encounter reinfection, called “immune priming” (also known as trained immunity). In addition, the immunological role of mosquito symbiotic bacteria (including gut microorganisms and Wolbachia) will be further investigated. This review provides an overview of mosquito innate immune mechanisms in terms of physical and physiological barriers, pattern recognition receptors, signalling pathways, cellular and humoral immunity, and immune priming, as well as the antipathogenic effects of mosquito symbiotic bacteria.
Physical and physiological barriers
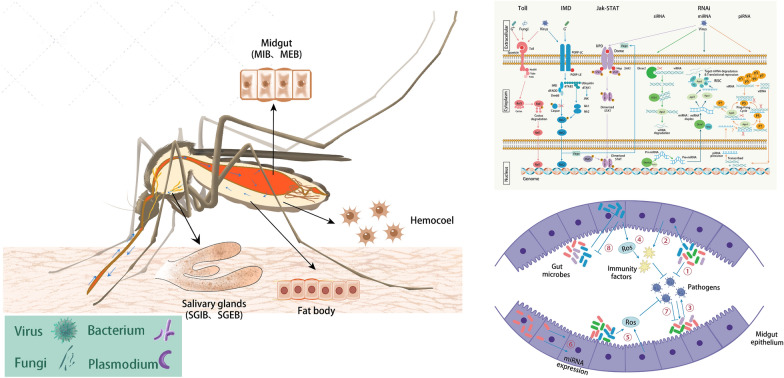
The physical barriers of mosquitoes mainly include the exoskeleton, cuticle, epithelial-epidermal barrier, trachea, fat body, gut, haemocoel (including haemolymph and haemocytes), and salivary glands. Four of these compartments, the midgut, haemocoel, salivary glands, and fat body, play dual roles as important physical and physiological barriers (Fig. 1). There are four main physiological barriers: midgut infection barrier (MIB), midgut escape barrier (MEB), salivary gland infection barrier (SGIB), and salivary gland escape barrier (SGEB). Physiological barriers are reaction processes that occur in mosquitoes in response to pathogens. Depending on the site of infection and the state of infection, different responses occur in mosquitoes with the goal of eliminating the pathogen. In the case of pathogens, the goal is to successfully establish infection in the host.
Fig. 1.
Main immune-related compartments of mosquitoes. The small blue arrows indicate the general circulation pattern of the pathogens in mosquito after the mosquito feeds on blood infected with the pathogen. Pathogens include viruses, bacteria, fungi, Plasmodium, etc. The pathogen enters the infected blood, partly develops in the intestinal epithelium, partly excretes from the intestines, and is passively transported through the mosquito's circulatory system as it crosses the haemocoel to reach the salivary glands. The large black arrows indicate the four main immune compartments, including the midgut [represented by midgut epithelial cells; containing the midgut infection barrier (MIB) and the midgut escape barrier (MEB)], salivary glands [containing the salivary gland infection barrier (SGIB) and the salivary gland escape barrier (SGEB)], haemocoel (represented by haemocytes), and fat body (represented by trophocytes and oenocytes)
Midgut
The midgut consists of a narrow anterior end and a wider posterior end, which are involved in the absorption of sugar and blood, respectively [7]. After the absorbed blood reaches the midgut, drastic changes in the epithelial cells of the midgut can be observed, such as smaller nuclei, enlarged mitochondria, the disappearance of vesicles of the rough endoplasmic reticulum and the appearance of vortices, an increase in the number of residual lysosomes, and the accumulation of charged opaque substances between the cells [8]. The peritrophic membrane (PM, assembled from chitin and proteins) is secreted by the epithelium into the midgut to protect the epithelial cells from damage; it compartmentalises the midgut and therefore the blood meal digestion and regulates the passage of molecules [9, 10]. So, it could act as a protective barrier against pathogens. Pathogen proliferation in the midgut is greatly restricted by mosquitoes; for example, ookinetes can be lost at a rate of 0.05–1.00 × 104-fold in the midgut of different mosquito species [11], and infection of the midgut triggers the production of large amounts of AMPs [12] and immune proteins in other tissues.
The current study demonstrates the presence of MIB and MEB in the midgut. In refractory mosquitoes with MIBs, the pathogen is unable to infect and replicate in mosquito midgut cells. There are several hypotheses that explain the mechanisms leading to MIB, including the transfer of pathogens to chitin-lined sacs in mosquitoes; compartmentalisation of pathogens (e.g. viruses) by the PM [13]; digestion of pathogens by enzymes in the midgut leading to inactivation; pathogen-midgut interactions that prevent binding [14]; lack of surface receptors in the midgut epithelium [9]. In the presence of MEBs, the pathogen can replicate in the midgut, even to high titres, but the virus may not be able to leave the midgut and spread the infection. Factors such as pathogen load or the dose and duration of pathogen transport determine whether pathogens successfully escape from the midgut [15]. The following two main hypotheses explain the MEB: the direct use of basal lamina and tracheal cells as a conduit between the midgut and the haematopoietic sheath [15]. MEB is dose-dependent [16, 17], and MEB also plays an important role the innate immune memory of pathogens in mosquitos [18].
Haemocoel
The haemocoel plays an extremely important role in the innate immunity of mosquitoes as pathogens penetrate the midgut and then enter the haemocoel, where they reach the rest of the body through the autodriven or the haemolymph circulation; this is the site where most cellular and humoral immunity occurs [19]. Once the pathogen enters the haemocoel, it is in an environment rich in immune cells and humoral immune factors consisting of haemocytes, pericardial cells, and fat bodies. Of these, haemocytes are the main immune cells in the mosquito. The mechanism of action in the antiviral immunity of haemocytes is unclear and direct evidence is lacking [20]. However, some studies have shown that arboviruses are tropic for mosquito haemocytes [21–23]. In addition, haemocytes are immune surveillance cells that initiate the immune response and have a variety of functions that may contribute to antiviral immunity, including the production of PRRs and proteins responsible for phagocytosis and nodulation, as well as other molecules (e.g. melanisation modulators and enzymes, signal transduction proteins, stress-responsive proteins, and AMPs) to kill or isolate pathogens [24].
Haemocytes are mainly classified into prohaemocytes, oenocytoids, and granulocytes, which exist as both circulating (within the haemolymph) and sessile (in the tissues) [1, 25, 26]. Granulocytes make up 80%–95% of circulating haemocytes and are phagocytic [27]. Oenocytoids make up about 10% and are the main producers of PO [28]. However, other studies have shown that haemocytes may produce a variety of factors involved in melanisation, including POs, serine proteases (SP), serine protease inhibitors (SRPN), phenylalanine hydroxylase, dopachrome convertase and C-type lectins (CTLs), as well as cytotoxic factors such as ROS and RNS [24, 29–32]. In addition, multiple haemocytes aggregate and bind to bacteria to form multicellular sheaths, called encapsulation or nodulation, which is the primary defence response for the removal bacteria from the haemolymph [33, 34].
Salivary glands
The salivary glands play a major role in disease transmission, and invasion of the salivary gland epithelium and migration to the salivary gland ducts is necessary for most pathogens to complete their life cycle. Salivary glands secrete a variety of proteins involved in different activities, such as CTLs that bind to specific carbohydrates on the surface of microorganisms and are the primary PRRs [35–38]; adenosine triphosphate bisphosphatase (apyrase), which is involved in blood feeding and helps mosquitoes locate blood [39]; and D7, which interferes with haemostasis and vertebrate immune responses [40]. To date, little is known about the role of the salivary glands as immunologically active organs in the anti-pathogen response, but saliva contains complex protein-peptide mixtures, antimicrobial agents, anticoagulants, proteins with angiogenic or anti-inflammatory properties, and immunomodulators, which can act in conjunction with pathogens that infect the host [41].
Furthermore, the molecular mechanisms underlying the SGIB and the SGEB in the salivary glands have not been clearly defined [42]. The concepts of the SGIB and SGEB are tentatively supported by different pathogen infection experiments in different mosquito species, including La Crosse virus (LACV) in Aedes triseriatus, Eastern equine encephalitis virus (EEEV) in Ae. albopictus, and Japanese encephalitis virus (JEV) and West Nile virus (WNV) in Ae. aegypti [43–45]. The SGIB has been demonstrated in Culex annularis infected with Western equine encephalitis virus (WEEV) and EEEV [16]. These viruses spread from the midgut to the fat body but cannot enter the salivary glands. Experiments with Rift Valley fever virus (RVFV) infection in the genus Aedes provide evidence that the basal lamina surrounding the salivary glands acts as a major barrier to infection [46]. Another study described the role of haemolymph in the SGIB, with the haemolymph of Culex tarsalis being more susceptible to WEEV infection compared to refractory females [14]. According to the study, the SGEB appeared in Aedes and Culex mosquitoes transmitting LACV, SINV, and RVFV [47–50]. The study likewise found that AgESP contributes to the passage of Plasmodium through the SGEB of Anopheles gambiae and that Plasmodium successfully completes its life cycle in mosquitoes by modifying the actin cytoskeleton of mosquito epithelial cells, a process that AgESP may be involved in regulating [17].
Fat body
The fat body is a major metabolic organ and a primary source of haemolymph proteins [51]; it participates in energy metabolism and reproduction by providing precursors for flight and yolk protein synthesis [52] and plays an important role in the innate immune response. The expansion of the fat body from the body wall to the visceral organs is hypothesised to increase the surface area of the organs and enhance communication with the haemocoel [53]. The fat body consists primarily of trophocytes and oenocytes but is generally thought to include sessile haemocytes, pericardial cells, peripheral neurons, and associated tracheoles [54, 55]. Trophocytes are mesodermal cells that provide energy and nutrients for locomotion and reproduction [56]. Oenocytes are ectodermal cells whose primary functions are detoxification, lipid metabolism, and, to a lesser extent, the maintenance of homeostasis in the mosquito; they are immunocompetent. Transcriptomic analysis revealed that oenocytes express many immune-related genes, with the most abundant transcript being lysozyme P. In addition, the peripheral position of oenocytes may contribute to the recognition of pathogens to activate the innate immune system, which in turn elicits the expression and secretion of immunity factor [57].
In insects, the fat body is the main organ that responds to microbial invasion and is able to secrete AMPs that can rapidly reach effective concentrations against invading microorganisms [58]. In mosquitoes, the fat body is equally involved in several immune pathways and AMPs synthesis in response to bacterial and Plasmodium infections [59]. Some of the fat body transcripts are thought to encode immune-related proteins, including defensins (DEFs), cecropins (CECs), CTLs, gram-negative binding proteins (GNBPs), and peptidoglycan recognition proteins (PGRPs) [52, 60]. In addition, enhancement of insulin signalling within the fat body of Anopheles stephensi enhances the host immune response to bacterial and Plasmodium falciparum infections [61]. Several studies have shown that altering the immune function of mosquito fat body through transgenic techniques can make mosquitoes susceptible to pathogens or develop resistance [62]. It has also been found that proliferation of oenocytes expressing DBLOX peroxidase maintains immune priming in mosquitoes [63].
Pattern recognition receptors (PRRs) and pathogen-associated molecular patterns (PAMPs)
The activation of the mosquito innate immune system first starts with the recognition of pathogens. When a pathogen infects a mosquito, it is recognised by the host through molecular interactions between PRRs and PAMPs. A genomic analysis of A. gambiae revealed the presence of nearly 150 PRRs, the vast majority of which clustered as members of several gene families. Usually, PRRs are host-secreted proteins found in different body parts, such as the midgut, and have an adhesive structural domain that interacts with PAMPs.
Thioester-containing proteins (TEPs)
TEPs are normally found in haemolymph and are primarily involved in bacterial and Plasmodium infections. Their role as essential pathogen recognition molecules in Drosophila melanogaster, A. gambiae, and Ae. aegypti allows for the neutralisation of pathogens [64–66]. In mosquitoes, studies on TEP have focused on TEP1 produced by haemocytes, which is phagocytic and inactive on its own and requires activation by proteolytic cleavage [67]; TEP1 then stabilises itself by forming a complex with the leucine-rich repeat (LRR) structural domains of the LRIM1 and APL1C proteins [3]. Only after the formation of the complex can stable TEP1 bind to bacteria in the haemolymph and Plasmodium in the midgut, thus leading to clearance [68–70]. Variants or polymorphisms in the TEP1 and APL1 sequences were associated with the efficiency of Plasmodium in killing mosquitoes [71]. The role of TEPs was similarly evaluated in Ae. aegypti infected with dengue virus (DENV) and WNV. RNAi-mediated overexpression and silencing of TEP1 and TEP3 revealed that the viral load was reduced when TEP1 was overexpressed, but TEP3 overexpression did not lead to a reduction in viral load, further illustrating the role of TEP1 in regulating viral infection [65].
Fibrinogen-related proteins or fibrillin (FREPs or FBN)
FREPs are the largest family of pattern recognition receptors in A. gambiae, with 59 putative members identified, and 37 FREPs have been identified in Ae. aegypti, most of which had upregulated expression in response to bacterial, fungal, or Plasmodium infection [10, 72, 73]. Using RNAi-mediated gene silencing, members of the FREP family were found to play a crucial role in the innate immune response and maintenance of immune homeostasis in mosquitoes [74]; furthermore, silencing of fibrinogen-related protein 30 (FBN30) was able to markedly promote parasitic infections, whereas eliminating FBN1 transcripts resulted in the absence of parasites within the mosquito [75]. The most studied member of this family is FBN9, which is associated with antibacterial and Plasmodium immunological mechanisms, as FBN9 interacts with bacteria and binds directly to Plasmodium in midgut epithelial cells, leading to their destruction [76, 77]. A comparative analysis of FNB9 gene sequences in four neotropical anopheline species (Anopheles davidianus, A. nueces, A. salinarius, and A. albopictus) and A. gambiae was also carried out [78], and clustering analysis showed that neotropical anophelines and A. gambiae belonged to two subgenera in different evolutionary branches.
C-type lectins (CTLs)
CTLs are membrane-bound or soluble proteins that bind to carbohydrates in a calcium-dependent manner and recognise pathogens by means of cell adhesion and cell-cell interactions, among other interactions. In addition, CTLs promote the activation of prophenoloxidase and melanin deposition on pathogen surfaces in insects [79]. CTLs can induce both positive and negative feedback regulation in the immune response of mosquitoes. In A. gambiae, CTLMA2 and CTL4 of this family inhibit melanisation in the midgut, whereas in the haemocoel, they can be present as disulfide-linked heterodimers that kill Escherichia coli in a melanisation-independent manner. RNAi-mediated silencing or knockdown of these proteins resulted in increased bacterial loads in the bloodstream and reduced mosquito survival, suggesting that both proteins play a role in the antimicrobial response in mosquitos [38, 80]. In Ae. albopictus, the mannose-binding C-type lectin Aalb_CTL is equally involved in combatting yeast and gram-positive bacterial (G+) infections [15]. However, in Ae. aegypti, galactose-specific C-type lectin 1 (mosGCTL-1) promotes WNV adhesion to cell membranes, and mosGCTL-3 is the main factor that promotes DENV2 infection [37].
Gram-negative binding proteins (GNBPs) and peptidoglycan recognition proteins (PGRPs)
GNBPs and PGRPs are the first PRRs to be studied in mosquitoes and can be found in different tissues, such as the midgut and salivary glands; these proteins play an important role in the recognition of bacteria and parasites [81]. In A. gambiae, six family members are recognised as PRRs, which act by binding to β-1,3-glucan and lipopolysaccharide on the surface of the pathogen. All members of this family are increased after infection but vary in their antimicrobial specificity and activity. GNBP4 is implicated in the regulation of immune signalling and plays an important role in E. coli, Staphylococcus aureus, and Plasmodium berghei infections but not in P. falciparum infections. GNBPA2 is an important factor involved in the killing of E. coli and P. falciparum, but it has little effect on P. berghei and is not involved in S. aureus infections [82]. PGRPs recognise and bind peptidoglycan on the surface of pathogens. In Armigeres subalbatus infected with E. coli and Micrococcus garciniae, AsPGRP-S1 expression was increased after infection, and after silencing by RNAi, it was found that AsPGRP-LCs may be involved in mosquito survivability but they are not the main factor in regulating bacterial infection [83].
Immunoglobulin superfamily (IgSF)
The IgSF specifically recognises and binds to cell surface receptors [6], and the IgSF in A. gambiae consists of 138 genes, 85 of which have upregulated expression after infection and are involved in resistance to Plasmodium and bacterial infections. One of the infection-responsive genes with an immunoglobulin domain (IRID), IRID4 and IRID6, is involved in killing P. falciparum conidia; IRID3 and IRID4 control the growth of opportunistic bacteria, and IRID3, IRID5, and IRID6 are involved in scavenging exogenous bacteria [84]. Another member of this family, AgDSCAM, has been shown to modulate bacterial infection and kill Plasmodium in the midgut [77].
Immune signalling pathways
Immune signalling pathways protect mosquitoes from persistent infection by invading pathogens and opportunistic microorganisms and are involved in the regulation of the natural microbiota, such as the gut flora. Immune signalling is an intermediate process that links pathogen recognition and the immune response. Three main immune signalling pathways exist in mosquitoes, namely, the Toll pathway, immune deficiency (IMD) pathway, and Janus kinase (JAK)/signal transducer and activator of transcription (STAT) pathway (Fig. 2). These pathways are activated and trigger immune effector molecules to neutralise invading pathogens, including bacteria, fungi, Plasmodium, and viruses (Table 1). In addition, the RNA interfering (RNAi) pathway, although not a classical innate immune signalling pathway, also plays a key role in antiviral defence.
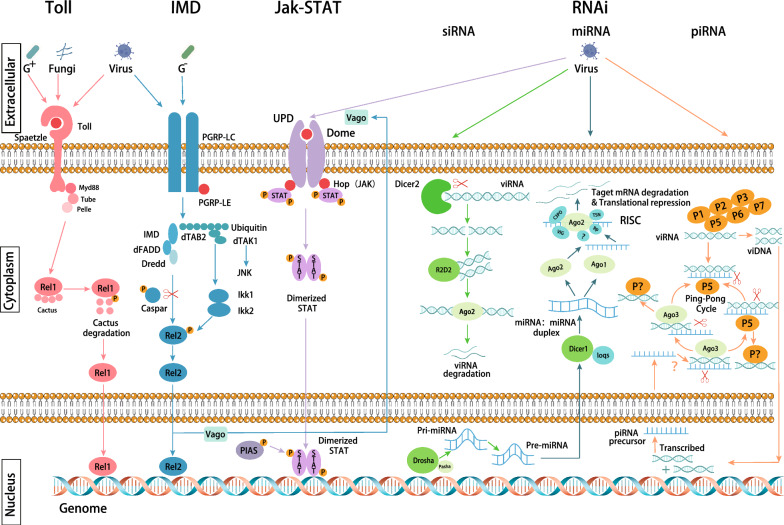
Fig. 2.
Immune-related signalling pathways in mosquitos. In the Toll pathway, the pathogen first binds to PPRs, which triggers the proteolytic cleavage of the cytokine Späetzle and then activates the Toll receptor to which it binds. Subsequent triggering of signals via the adaptor proteins MyD88, Tube, and Pelle leads to the phosphorylation and degradation of the Cactus protein, an inhibitor of Rel1. Finally, the degradation of the Cactus protein translocates Rel1 to the nucleus and activates the Toll pathway to regulate gene transcription. In the IMD pathway, pathogens bind to PGRP-LC and PGRP-LE ligands and trigger signalling by various cysteoaspartases and kinases. One branch triggers the JNK signalling pathway, while the other triggers signals via IMD, FADD, and Dredd, which mediate phosphorylation, as well as the cleavage of Rel2. Caspar proteins act as negative regulators of the IMD pathway. Subsequent translocation of Rel2 to the nucleus activates the IMD pathway to regulate gene transcription. The JAK-STAT pathway is triggered by the binding of Unpaired (Upd) to the receptor protein Dome, which activates the receptor-associated Hop Janus kinase. Activated Hop kinases phosphorylate each other and subsequently recruit phosphorylated STAT transcription factors and form dimers. Phosphorylated STAT dimers are translocated to the nucleus to activate JAK-STAT-regulated transcription. RNAi pathways include the siRNA pathway, miRNA pathway, and piRNA pathway. In the siRNA pathway, exogenous dsRNA is recognised by the RNA-binding structural domain of Dicer2 and is sheared into small dsRNA of 21–28 nt. The Ago2 protein leads to the silencing of complementary transcripts in the cell by degrading or inhibiting the translation of sRNAs by binding to them, and R2D2 is involved in the delivery of the siRNA guide strand from Dicer2 to Ago2. In the miRNA pathway, pri-miRNAs are produced in the nucleus using the genome and processed by Drosha and Pasha proteins to be converted into pre-miRNAs. Pre-miRNA is translocated to the cytoplasm via export protein 5, where it is further processed by the Dicer1 and Loqs complexes to form miRNA-miRNA duplexes, followed by the formation of guide RNAs via Ago1 and Ago2, leading to degradation of the target mRNAs and transcriptional repression through the RISC complexes (composed of Ago2 or Ago1, VIG, TSN, Rp, C3PO, and other unknown proteins). In the piRNA pathway, after viRNA enters the host cell, it binds to unknown PIWI proteins (PIWI 1–7) and directly enters the ping-pong cycle to bind to the complementary antisense RNA of Piwi5 for shearing and methylation. The antisense RNA is then released to bind to an unknown PIWI protein. The processed positive-sense RNA then binds to Ago3 to form a long-stranded antisense piRNA precursor, and the antisense piRNA precursor is sheared and methylated to form an antisense piRNA release, which binds to Piwi5 to start another round of the ping-pong cycle. The other viRNA undergoes reverse transcription and replication and then enters the host cell nucleus to integrate into the genome, where it is subsequently transcribed and translocated into the cytoplasm to enter the ping-pong cycle
Table 1.
Studies on mosquito immune signalling pathways
| Immune signalling pathways | Mosquito/cell line | Pathogen | Key genes/effectors | References |
|---|---|---|---|---|
| Toll pathways | Anopheles gambiae | Plasmodium berghei | Toll, Rel1, Cactus, LRIM1, Tep1 | [87] |
| Aedes aegypti | Plasmodium gallinaceum, fungi, DENV, SINV, WNV | RUNX4, AaREL1, Spz1A, Serpin-27A, AMPs, Dif, Späetzle 5 | [88–92, 94, 95, 97] | |
| IMD pathways | Anopheles gambiae | Bacterium, Plasmodium berghei, P. falciparum | Caspar, Rel2, AMP, cecropin 1, TEP1, APL1, LRIM1 | [98–100] |
| Anopheles stephensi | Plasmodium falciparum | Rel2 | [99, 102] | |
| Anopheles albimanus | Plasmodium falciparum | Rel2 | [99] | |
| Aedes aegypti | DENV, SINV | Caspar, Rel2 | [12, 92, 103, 105] | |
| Aedes albopictus U4.4 cell | SFV | – | [104] | |
| JAK-STAT pathways | Anopheles gambiae | Bacterium | STAT-A, STAT-B | [106] |
| Aedes aegypti | DENV | STAT, Dome, Hop, PIAS | [107] | |
| Anopheles aquasalis | Plasmodium vivax | STAT, PIAS | [108] | |
| siRNA pathways | Anopheles gambiae 4a-2s4 cell | FHV, NoV | Ago2 | [116] |
| Aedes aegypti | YFV, DENV2, SINV | Dicer2, R2D2, Ago2 | [117–119] | |
| Anopheles gambiae | ONNV | Ago2 | [121] | |
| miRNA pathways | Anopheles gambiae | Plasmodium falciparum | aga-miR-305 | [124] |
| Aedes aegypti | ZIKV, DENV | – | [125, 126] | |
| Aedes aegypti Aag2 cell | WNV | KUN-miR-1, GATA4 | [129] | |
| Aedes albopictus C6/36 cell | DENV, WNV | DENV-vsRNA-5, Aae-miR-2940-5p, aal-miR-4728-5p, miR-1767, miR-276-3p, miR-4448, KUN-miR-1, GATA4 | [127–130, 132] | |
| Aedes albopictus | DENV2 | miR-252 | [131] | |
| piRNA pathways | Aedes aegypti | CHIKV | vpiRNA | [137] |
| Aedes albopictus | CHIKV | vpiRNA | [137] | |
| Aedes aegypti Aag2 cell | SINV, SFV | Piwi5, Ago3, vpiRNA, Piwi4 | [135, 139, 141] |
The Toll signalling pathway
The Toll pathway was originally discovered during the screening of genes associated with early embryonic development in D. melanogaster and was later found to play a crucial role in defence against fungi, gram-positive bacteria, and viruses [85–87]. The Toll pathway has reshaped the understanding of the immune system not only in Drosophila but also in other insects and even mammalian systems. In mosquitoes, genes regulated by the Toll pathway are controlled by the transcription factor Rel1 of nuclear factor kappa B (NF-κB). Silencing Cactus, a negative regulator of Rel1, to induce the Toll pathway significantly reduces the extent of P. berghei and Plasmodium gallinaceum infection in the midgut of Anopheles and Aedes mosquitoes. Cosilencing Rel1 and Cactus makes mosquitoes more susceptible, suggesting that Cactus-mediated susceptibility is controlled by Rel1 [88, 89]. Cosilencing of Cactus and LRIM1 or Tep1 also renders mosquitoes susceptible, suggesting that these two effector molecules are induced by the Toll pathway [88].
In addition to the induction of antimalarial activity by Rel1, the Toll pathway is involved in the control of fungal and DENV infections [90, 91]. Knockdown of Rel1 increases mosquito susceptibility to fungus and attenuates the induction of Spaetzle 1A and Serpin-27A [90]. Serpin-27A is overexpressed in heat-killed bacterial Aag2 cells as well as in fungus-infected female Ae. aegypti mosquitoes [92]. DENV infection of Ae. aegypti activates the transcription of Toll pathway-associated factors and putative effectors (Späetzle, Toll, Rel1A, and multiple AMPs) [91, 93, 94]. The Toll pathway begins to exert antiviral effects as early as 3 days after DENV infection, is able to protect against DENV infection in multiple serotypes, and remains active in different Ae. aegypti strains [95]. Furthermore, the symbiotic Wolbachia in mosquitoes can inhibit DENV replication by inducing mosquitoes to produce ROS to activate the Toll pathway and subsequently produce AMPs and DEFs [96]. The transcription of Dif, a Toll pathway transcription factor, is induced early in the infection of Ae. aegypti with Sindbis virus (SINV) [12]. The role of the Toll pathway in defences against WNV infection is unclear; Culex quinquefasciatus infection with WNV does not significantly alter the expression of Toll pathway-associated genes or effectors [97], whereas Ae. aegypti infection downregulates the expression of mosquito homologues of Drosophila Späetzle 5 [98]. In addition, it has been found that activation of Toll signalling in A. gambiae mosquitoes by silencing Cactus modulates haemocyte differentiation leading to a large increase in circulating megacytes (from 2% to 80% of granulocytes) [99]. Moreover, megacytes are recruited to the midgut during Plasmodium infection and release microvesicles that promote the activation of the mosquito complement-like system, thereby eliminating Plasmodium ookinetes and enhancing mosquito immunity.
Imd signalling pathway
The Imd pathway likewise plays a crucial role in mosquito immunity. The molecules acting in this pathway overlap with those in the Toll pathway; together, they trigger an immune response. Similar to Rel1 in the Toll pathway, Rel2, which belongs to the same family, plays a central role in the Imd signalling pathway and is involved in the regulation of major AMPs and cecropin 1. The Imd pathway not only plays an important role in antimicrobial defence but also directs the immune response to Plasmodium [100–103]. This pathway also has an indirect effect on the viral load of Ae. aegypti [104]. Knockdown experiments showed that two isoforms of Rel2 (Rel2-F and Rel2-S) in Anopheles mosquitoes are involved in immune defence against gram-positive and -negative bacteria, respectively. Rel2-F is also involved in regulating the intensity of P. berghei infection [100]. Although both the Toll and Imd pathways are involved in the immune response to P. berghei, the immune response to P. falciparum is mainly controlled through the Imd pathway. Caspar proteins are negative regulators of Rel2, similar to the function of Cactus for Rel1. Several studies have shown that silencing Caspar may enhance the immune response to Plasmodium [102]. In addition, overexpression of Rel2 rendered A. gambiae, A. stephensi, and Anopheles albimanus completely resistant to P. falciparum [101].
Related studies have shown that some Imd pathway-associated proteins and effectors can promote DENV and SINV infection [12, 93]. However, the transient activation of the Imd pathway through RNAi-mediated Caspar silencing did not affect the DENV titre in the midgut of mosquitoes [91]. Furthermore, in experiments where SFV infected Ae. albopictus cells, the addition of heat-inactivated gram-negative bacteria (activating both the Imd and JAK-STAT pathways) prior to infection resulted in a reduction in SFV load [105]. Another study showed that disruption of the Imd pathway in DENV-tolerant strains of Ae. aegypti resulted in a decrease in midgut DENV titres [106], but DENV titres were unchanged in susceptible strains, suggesting that the Imd pathway may be needed for defence against DENV but that in susceptible strains the pathway may already be operating at maximum load.
JAK/STAT signalling pathway
The JAK/STAT pathway, a major signalling pathway induced by interferon, is one of the less studied immune pathways in mosquitoes. In A. gambiae, this pathway is regulated by two transcription factors, STAT-A and STAT-B. STAT-B regulates the transcription of STAT-A, and bacterial infection leads to the translocation of STAT to the nucleus [107]. Whereas only one type of STAT is present in Ae. aegypti, the JAK/STAT pathway plays an important role in the immune response against DENV infection in Ae. aegypti. DENV replication in the mosquito midgut was significantly increased when the pathway was inhibited by RNAi-mediated silencing of transmembrane proteins (Dome receptors) or JAK immediate homologues (Hop proteins). In contrast, viral replication was inhibited when an inhibitor (negative regulator PIAS) of activated STAT in the pathway was silenced [108]. Another study showed that the JAK/STAT pathway is involved in the immune response against Plasmodium intermedius infection in A. aquasalis [109] and found that the transcript levels of STAT and PIAS were elevated 24 and 36 h after Plasmodium infection. In addition, in uninfected Plasmodium mosquitoes, STAT and PIAS are expressed mainly in the fat body. After infection, they are expressed in other tissues, and silencing STAT increases the number of oocysts in the midgut.
RNAi pathway
RNAi is a highly conserved, sequence-specific mechanism of gene silencing at the posttranscription level that is primarily involved in the antiviral immune response of mosquitoes. RNAi is a process by which double-stranded RNA (dsRNA) induces small RNA (sRNA) molecules and thus efficiently and specifically degrades homologous mRNA. The RNAi pathway mainly produces sRNA molecules with different characteristics; endogenous small interfering RNAs (siRNAs, 18–24 nt), microRNAs (miRNAs, 18–24 nt), and PIWI protein-interacting RNAs (piRNAs, 24–30 nt) are produced, with siRNAs being the main molecules in the antiviral response [26, 110, 111]. In addition to the small molecules mentioned above, exogenous siRNAs of viral origin may also trigger antiviral responses. These sRNAs form the RNA-induced silencing complex (RISC) by binding to protein molecules such as AGO, TRBP, and PACT, which recognise the target RNA through a specific family of Argonaute (Ago) proteins, leading to gene silencing.
siRNA
The siRNA pathway is mainly triggered by Dicer2, R2D2, and Ago2 and can be classified as exogenous and endogenous. Exogenous siRNAs are involved in antiviral immunity by processing exogenous RNAs derived from viruses (or viRNAs), and endogenous siRNAs are involved in regulating cellular processes by binding endogenously encoded dsRNAs [112]. Exogenous dsRNA is recognised by the RNA-binding structural domain of Dicer, which has ribonucleic acid endonuclease activity (RNase III) and is sheared into small dsRNAs of 21–28 nt. Biosynthesis of endogenous siRNAs primarily involves RNA polymerase IV and RNA polymerase V [113]. Dicer is a key element in the biosynthesis of most sRNA molecules, and its main role is to shear dsRNA into sRNAs [114]. The Ago family plays a central role in the RNAi pathway by binding to sRNAs and thereby degrading or inhibiting their translation, leading to silencing of the complementary transcripts in cells. R2D2 is involved in the delivery of the siRNA guide strand from Dicer2 to Ago2; thereby, R2D2 acts as a bridge to the RNAi pathway [115]. The antiviral effect of siRNA was first demonstrated in A. gambiae cells, and Ago2 was found to be involved in inhibiting viral replication [116]. Since then, several studies have found that the siRNA pathway inhibits the replication of yellow fever virus (YFV) [117], DENV2 [118] and SINV [119, 120], and the level of viral replication is significantly increased upon the knockdown or silencing of siRNA pathway-related genes [121].
miRNA
miRNAs are small endogenous noncoding RNAs that regulate gene expression by degrading mRNAs or terminating transcription by binding to the noncoding regions at the 3′ ends [122]. The mechanism of miRNAs is similar to that of siRNAs, with the main difference being their location in the cell and the effector proteins involved [123]. siRNAs occur mainly in the cytoplasm, whereas the formation of miRNA precursors (pre-miRNAs) is performed in the nucleus, and they are later transported to the cytoplasm for action. miRNAs are involved in a variety of cellular functions, ranging from development to participation in anti-infection immune mechanisms. miRNAs regulate mosquito immunity mainly by controlling the expression of immune-related genes and genes subject to posttranslational modifications or by directly binding to the genome of pathogens. Studies have shown that aga-miR-305 increases the susceptibility of A. gambiae to P. falciparum, which replicates itself by utilising miRNAs from reservoirs in host cells [124]. Infection with mosquito-borne viruses such as ZIKV [125], DENV [126, 127], and WNV can similarly trigger miRNA responses, leading to the differential expression of multiple miRNAs. Among them, the study confirmed that downregulation of Aae-miR-2940-5p expression inhibited WNV replication [128]; KUN-miR-1 promoted WNV proliferation in Ae. aegypti and Ae. albopictus cells by upregulating the expression of the transcription factor GATA4 [129]; aal-miR-4728-5p enhanced DENV2 replication after overexpression in Ae. albopictus C6/36 cells [130]; miR-252 inhibited DENV2 infection in Ae. albopictus by regulating E protein expression [131]. miR-1767 and miR-276-3p enhanced DENV2 replication in C6/36 cells, whereas miR-4448 inhibited DENV2 replication [132].
piRNA
The piRNA pathway is poorly understood, but it has regulatory roles in germline maintenance and development [133], the protection of transposons [134], and the regulation of viral infections [135]. Unlike siRNAs and miRNAs, piRNAs are dependent on PIWI subfamily proteins, mainly Piwi, Aub, and Ago3, which together form the piRNA-induced silencing complex (piRISC) [136]. piRNAs are mainly derived from repetitive sequence elements or piRNA clusters, but viral genomes are another source of piRNAs (vpiRNAs) [137, 138]. vpiRNA was detected in Ae. aegypti and Ae. albopictus infected with chikungunya virus (CHIKV) [137] as well as in SINV-infected Ae. aegypti cells [135]. In siRNA-deficient mosquito cells, cellular lesions were found to be aggravated by the inhibition of vpiRNA expression, suggesting that piRNAs exert their antiviral effects in the presence of defects in the siRNA pathway, indicating that piRNA is complementary to the RNAi pathway in the immune response [137]. In addition, another study showed that in Ae. aegypti, a noncanonical PIWI protein involved in the antiviral response, Piwi4 knockdown resulted in increased SFV replication in Ae. aegypti Aag2 cells [139]. piRNAs are also involved in host resistance to specific viruses. Endogenous bornavirus-like nucleoprotein elements were found to be reverse transcribed and integrated into primate and rodent genomes, resulting in resistance to bornavirus [140]. In addition, the piRNA pathway may play a role in preventing the transmission of vertically transmitted arboviruses through germ cells [141].
Immune effects
AMPs
AMPs are small molecule proteins with in vitro antimicrobial activity that are mainly produced by the Toll and IMD signalling pathways. These two pathways regulate the expression of AMPs by activating different transcription factors, which are increased in the fat body when mosquitoes are infected with pathogens [91, 142]. DEFs, CECs, and gambicins (GAMs) are the three main AMP families in mosquitoes. CECs and GAMs primarily target gram-positive bacteria, and DEF primarily targets gram-negative bacteria. DEF has antimicrobial activity in vitro, but its role as an essential component of the mosquito immune response is still unclear. In A. gambiae, knockdown of DEF reduced the survival of S. aureus-infected mosquitoes but the mosquitoes did not show resistance to malaria [143]. Sequence analysis of Ae. aegypti examining 17 AMPs found that the mRNA expression of 7 AMPs was significantly increased after DENV2 infection [144]. In addition, there was no effect on the survival of bacteria-infected mosquitoes after silencing DEF in Ae. aegypti [145, 146]. DEF and CEC inhibit DENV replication in Ae. aegypti infected with Wolbachia [96]. The mechanism of the antimicrobial response to CECs is not known, but ectopic expression of CECs can limit P. berghei infection in A. gambiae [147]. Escherichia coli and Streptococcus mucin also stimulate CEC-A, D, E, F, and N expression in Aag2 cells. GAMs exhibited antimalarial activity in the midgut and antimicrobial activity in the blood lumen, and the silencing of GAMs resulted in increased P. berghei loads in A. gambiae [148]. Furthermore, the silencing of key factors of the JAK-STAT pathway in Aag2 cells leads to reduced GAM activity against bacterial infections, suggesting that immune signalling plays a central role in AMP production [149].
NO and ROS
NO and ROS are two important effector components of the mosquito immune response. NO is a multifunctional free radical that is produced during the oxidation of l-arginine to l-guanine and catalysed by nitric oxide synthase (NOS). In the midgut, Plasmodium glycosylphosphatidylinositol and Plasmodium-derived haemagglutinin obtained through infection of the blood meal induce NOS transcription via the STAT pathway, and the resulting NO kills ookinetes by cleavage. NOS is upregulated in the haemocoel after E. coli and Micrococcus luteus infections, and NO is necessary for bacterial killing and mosquito survival during E. coli infection [150]. ROS are involved in the clearance of ookinetes from the midgut and bacteria from the haemocoel, and although the exact mechanism of action on bacteria is unknown, ROS kill Plasmodium via the cleavage and melanisation pathways [151–153]. Oral antioxidants also reduced melanisation [151]. The L3-5 strain of A. gambiae (P. berghei resistant) lives in a state of chronic oxidative stress that promotes the melanisation of ookinetes in the midgut epithelium [152]. In contrast, G3-susceptible strains kill ookinetes through an infection-induced oxidative stress cleavage mechanism that can be maintained by the inhibition of catalase [153]. The silencing of catalase promotes Plasmodium infection in the midgut and reduces the bacterial load. In addition, silencing of the dioxygenase DuoX reduced ROS levels and promoted the proliferation of gut microbes [154].
Melanisation
Melanisation is another important immune response developed by mosquitoes against invading pathogens and is a complex mechanism. The major components involved in melanisation include PO, SP, SRPN, and serine protease hairpin domains (CLIPs). Melanisation initiates the sequential cascade of SPs via PAMPs, which catalyse the hydrolysis of phenol oxidase proteins (proPOs) to active PO. PO is a key enzyme in melanisation, where tyrosine is hydroxylated to DOPA by PO; DOPA is further oxidised to dopaquinone, and in the presence of thiols, dopaquinone forms a cysteine and glutathione coupling to form yellow/red pheomelanin, or in the absence of thiols, dopaquinone is spontaneously converted to dopachrome, which is further converted to eumelanin in brown/black polymers [155], which encapsulate and destroy invading pathogens. SPs and POs are involved in melanisation, and SRPN acts as an inhibitor of serine protease to tightly control the SP cascade [156]. SRPN regulates the activation of proPOs and may activate the Toll pathway to cause Plasmodium lysis [157]. At least three SRPNs were found to be involved in the anti-Plasmodium innate immune response in A. gambiae, with Ag-SRPN6 primarily inhibiting Plasmodium proliferation and transmission [158]. CLIPs are similarly involved in regulating the melanisation of Plasmodium. SPCLIP1, an arthropod-specific noncatalytic serine protease, is involved in TEP1 accumulation on the surface of invading pathogens to regulate the complement pathway in mosquitoes, thereby inducing a peak in the Plasmodium and bacterial melanisation cascade [159]. Another study found that SRPN7 and CLIP2 play a synergistic role in the cascade signalling of mosquitoes during Plasmodium infection, with SRPN7 acting as an inhibitor and CLIP2 acting as a facilitator [160]. However, neither SRPN7 nor CLIP2 were transcriptionally regulated following infection with P. berghei. CTLs also inhibit the melanisation of mosquitoes against P. berghei, and the silencing of CTL4 and CTLMA2 causes LRIM1-dependent ookinete melanisation [80]. The silencing of the key regulators of melanisation TEP1 and CLIPA8 completely prevented mycelial melanisation but not melanisation in conidia and shoot tubes and resulted in more rapid proliferation of the fungus and greater susceptibility to Coccidioides albicans. In addition, amino acid metabolism has an important role in melanisation, and tyrosine synthesis is reduced following the silencing of phenylalanine carboxylase (PAH) of the amino acid metabolic pathway, thus altering melanisation in response to P. berghei. Carbidopa, an inhibitor of DOPA decarboxylase, similarly affects egg melanisation [161]. Intermediates produced during hydrolysis of ProPOs inactivate SFV; conversely, SFV can activate the melanisation cascade of POs to inhibit virus transmission [162].
Apoptosis
Apoptosis leads to programmed cell death (PCD) and is essential for maintaining homeostasis because it removes damaged and infected cells. Apoptosis inhibits viral replication as an intrinsic response to viral infection in vertebrates [163, 164]. Although pathogen infection in insects often leads to non-pathogenic infections, there is growing evidence that apoptosis has an antiviral effects in insects [165]. Apoptosis in mosquitoes was found to be involved in the regulation of pathogen load. An apoptosis inhibitor antagonist gene, michelob-x (mx), was first identified in A. gambiae, which is similar to the reaper-like apoptosis inhibitor antagonist gene in Drosophila and may be involved in regulating the pro-apoptotic response to viral infection [166]. Aedes aegypti (CuniNPV-refractory) larvae infected with CuniNPV rapidly generated mx in the midgut and induced the rapid apoptosis of infected cells at 4–6 h, whereas rapidly induced apoptosis was not detected in the larvae of Cx. quinquefasciatus (CuniNPV-susceptible). This finding suggests that apoptosis plays an important role in mosquito resistance to viral infection [167]. This phenomenon has been demonstrated in WNV-infected Cx. quinquefasciatus [168, 169] and DENV-infected resistant and susceptible Ae. aegypti [165]. There are two important factors in the mosquito apoptosis pathway, Aeiap1 and Aedronc. Aedronc is an RNAi-mediated primary apoptotic cysteinyl asparaginase, and the silencing of Aedronc was found to increase the prevalence of infection in resistant Ae. aegypti mosquitoes [170]. Aeiap1, an inhibitor of apoptosis, activates apoptosis by silencing IAP, leading to increased SINV titres in the midgut and viral spread to other sites in Ae. aegypti; meanwhile, silencing Aedronc has the opposite result [171]. These results are contrary to the hypothesis that apoptosis plays a role in the mosquito antiviral immune response. Apoptosis may limit viral infectivity by disrupting the physical barriers of the cell, but when apoptosis is artificially induced, both viral infectivity and transmission are enhanced.
Autophagy
Autophagy is a fundamental cellular process involved in the maintenance of cellular homeostasis in situations of stress and nutrient deprivation. Autophagy can activate or modulate the immune response and directly eliminate intracellular microorganisms [172]. Autophagy-related gene 5 (ATG5) in the midgut of DENV2-susceptible Ae. aegypti was transcriptionally elevated upon infection with DENV2, in contrast to resistant strains, and this elevation was associated with increased expression of apoptosis-related genes [173]. When these genes were silenced, a concomitant increase in DENV2 load was accompanied by accumulation of Atg8-PE, a marker of autophagy induction and progression [173]; furthermore, apoptosis-related genes are also involved in regulating autophagy.
Phagocytosis
Phagocytosis is a conservative immune process that has evolved to effectively neutralise or remove microorganisms. By recognizing that particles or microorganisms are excited and form phagosomes after being engulfed by phagocytes, the phagosome fuses with the lysosome, and the microorganism is digested by hydrolytic enzymes within the lysosome. In mosquitoes, the granulocyte subpopulation of haemocytes isolates and kills bacteria by phagocytosis [174, 175], approximately 95% of circulating haemocytes are phagocytic, and a single haemocyte can phagocytose > 1000 bacteria 24 h post infection [176]. The main phagocytosis regulators in mosquitoes are PRRs, transmembrane receptors, and intracellular signalling proteins. TEP1, TEP3, and TEP4 are involved in phagocytosis of gram-positive and -negative bacteria in the haemocoel [67, 177]. TEP1 downregulates bacterial prevalence through thioester-mediated binding, which in turn initiates phagocytosis, and the mechanism of action of TEP3 and TEP4 is unknown [177]. In addition, LRIM1 is also involved in phagocytosis, and its exact mechanism has not been elucidated, but it has been shown that the antimicrobial activity of TEP1 in the midgut depends on the complexation of TEP1 with LRIM1 in the haemocoel [68]. Knockdown of AgDSCAM, a highly variable immunoglobulin structural domain containing the receptor for A. gambiae, promoted bacterial proliferation in the haemocoel and reduced mosquito survival [77]; in addition, AgDSCAM recognises bacteria to initiate phagocytosis. Transmembrane receptors trigger phagocytosis by directly recognising pathogens or pathogens regulated by haemocyte proteins, including PGRPs, integrins (BINT2), and low-density lipoprotein receptor-related protein (LRP1) [177, 178]. Intracellular CED proteins can similarly trigger bacterial internalisation. In A. gambiae, knockdown of CED2, CED5, or CED6 reduced phagocytic efficiency to 80% [177]. Analysis showed that TEP1-, TEP3-, LRIM1-, and LRP1-mediated phagocytosis occurs through the CED6 pathway, and TEP4- and BINT2-mediated phagocytosis occurs through the CED2/CED5 pathway. Another study on A. gambiae found that cytoplasmic actin can act as a pathogen recognition factor by interacting with specific extracellular immune factors, which in turn bind to the bacterial surface to mediate phagocytosis [179].
Immune priming
In recent years, researchers have discovered that the mosquito's innate immune response can induce an enhanced immunity when it encounters reinfection, a phenomenon known as "immune priming" (also known as trained immunity). The innate immune memory of A. gambiae for Plasmodium infection is associated with haemocyte differentiation [18], mediated by the systemic release of haemocyte differentiation factor (HDF). Plasmodium infection mediates immune initiation in A. gambiae by inducing the release of lipoxin/lipocalin complex, which is associated with a sustained increase in the expression of a lipocalin lipid carrier called Evokin, which promotes haematopoietic differentiation [180]. Another study similarly found that prostaglandin E2 (PGE2) released from the midgut of A. gambiae attracts haemocytes to the midgut surface and enhances their patrolling activity, resulting in a more effective immune response to re-infection in A. gambiae [181]. Double peroxidase (DBLOX) is essential for HDF synthesis, and histone acetyltransferase AgTip60 is essential for oenocyte number, HDF synthesis, and immune priming [182].
It has been previously shown that the response of Ae. aegypti to bacterial reinfection is predominantly non-specific immune priming [183]. However, another study found that A. gambiae fed with Serratia fonticola S1 and Enterobacter sp. Ag1 sugar meal had a higher 24-h survival rate after re-infection and that this immune priming was specific, suggesting that the previous bacterial sugar meal enhanced the subsequent antibacterial immunity [184]. Bacterial exposure in the larval stage of Ae. aegypti was found to increase the survival of both male and female adult mosquitoes. However, there was sexual immune dimorphism, with higher PO activity in male mosquitoes and higher NO production, higher antibacterial activity, and longer duration in female mosquitoes [185]. Using the Ae. aegypti Aag2 cell line as a model, the investigators found that bacterial-induced immune responses inhibited RVFV replication, and that RVFV infection markedly enhanced subsequent bacterial-stimulated immune responses. RVFV infection affected gene expression of PRRs, which may be involved in immune priming [186].
Mosquitoes are also involved in immune priming during interactions with the gut microorganisms, including Wolbachia [187, 188]. It is a major way to increase the survival of reinfected mosquitoes and to limit Plasmodium invasion. Studies have also shown that immune priming to viruses [189] and Wolbachia [190, 191] sometimes enhances immunity but also suppresses the immune response. The effect of transgenerational immune priming (TGIP) on offspring immune response rates is also being further investigated. Aedes aegypti can pass on antiviral immune memory to their offspring, and the memory persists for several generations. CHIKV-infected Ae. aegypti offspring show lower viral loads when infected, and TGIP is viral RNA-dependent and sequence-specific but RNAi-independent [192]. In female offspring of Ae. aegypti infected with DV, transcripts of the siRNA pathway are reduced and immune priming decreases viral load [193].
Symbiotic bacteria and anti-pathogenic effects
Influence of gut microorganisms on immunity
As an important site for pathogen colonisation and determining the outcome of infection, the gut possesses complex microbial populations that influence multiple aspects of mosquito nutrition, reproduction, metabolism, immunity, and vectorial competence. Mosquitoes acquire viruses by sucking blood, which then infects intestinal epithelial cells and spreads throughout the body to various organs and tissues via the haemocoel. Gut microbes play a role in mosquito immune regulation by initiating host immune surveillance mechanisms and secreting metabolites (Fig. 3) [194, 195]. The mosquito innate immune system mediates the interaction of midgut microbes with DENV infection, and Proteus sp. prsp_P upregulates AMP expression and increases mosquito resistance to DENV infection [196]. Microbes trigger basal immune and antimicrobial responses against DENV infection, and the immune response triggered by DENV infection also affects midgut microbes. Another study found that in A. gambiae, gut microbes reduce infection rates and Plasmodium counts by producing antiparasitic effectors. Chromobacterium Csp_P in the midgut reduced the survival of larvae and adults and suppressed malaria and DENV infection. Additionally, Csp_P mediates in vitro anti-Plasmodium and anti-DENV activity by producing bioactive factors with transmission blocking and therapeutic potential [197]. In Ae. aegypti, Talaromyces alter mosquito physiology by modulating digestive enzymes and trypsin activity to facilitate pathogen infection [198]. Serratia marcescens is a key commensal bacterium that plays a role in the efficient acquisition of arboviruses. Serratia marcescens facilitates arbovirus infection by secreting the SmEnhancin protein, which digests membrane-bound mucin in the gut epithelium, thereby enhancing virus transmission [199]. The findings suggest that tripartite interactions among mosquitoes, gut microbes, and pathogens offer new possibilities for the control of mosquito-borne infectious diseases.
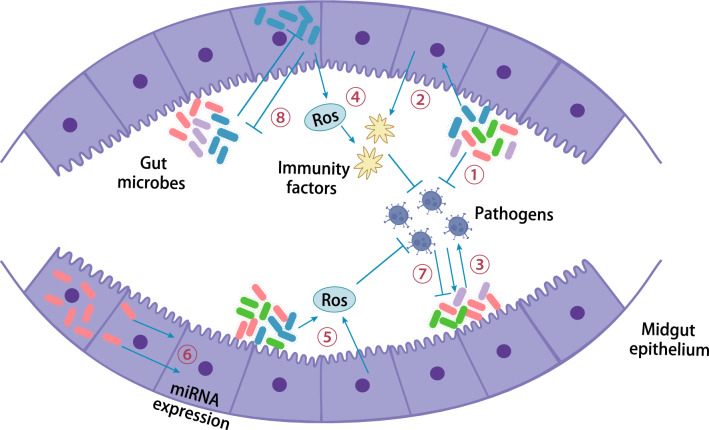
Fig. 3.
Interactions among mosquitoes, gut microbes, and pathogens. Red-, purple-, green-, and blue-coloured rods with hyphae represent various gut microbes. ① Gut microbes directly inhibit viruses; ② gut microbes can inhibit viruses by stimulating basal immunity; ③ gut microbes can potentiate viruses; ④ gut microbes, such as Wolbachia, can stimulate immunity through the production of ROS; ⑤ ROS can also be coproduced by a mosquito host and microbes and inhibit bacteria and pathogens; ⑥ intracellular microbes can modulate the expression of host miRNAs; ⑦ arboviruses can either inhibit or promote gut microbes; ⑧ interactions with gut microbes can affect the microbes themselves
Wolbachia
Currently, the most widely used microbial control strategy for insect-borne viruses is Wolbachia. Wolbachia is a maternally inherited intracellular symbiotic bacterium predicted to infect > 60% of insects [200]. Wolbachia is programmed to control insects by utilising the following two key processes of host biology: cytoplasmic incompatibility (CI) and pathogen blocking (PB) [201]. CI is a reproductive manipulation that manifests as fewer eggs being produced or the failure to hatch after mating between males infected with Wolbachia and uninfected females, the compatibility of females carrying Wolbachia with either infected or uninfected males for successful reproduction, and the possibility of CI between mosquitoes carrying different strains of Wolbachia, referred to as bidirectional CI. In addition, some Wolbachia trans infections also cause PB; Wolbachia inhibits pathogen infection, replication, and the transmission of key pathogens. Mosquito control interventions utilising Wolbachia generally follow two approaches, namely, population suppression and population substitution [202]. Population suppression targets the two main DENV vectors, Ae. aegypti (infection by the wAlbB strain) and Ae. albopictus (triple infection by the wAlbA, wAlbB and wPip strains). By releasing large numbers of Wolbachia-infected male mosquitoes to mate with wild females, CI then leads to a dramatic decline in the size of the target mosquito population by reducing female fecundity [203–206], with suppression rates of local mosquito populations ranging from 78% to 95%. The Wolbachia wMelPop-CLA strain introduced from D. melanogaster into Ae. aegypti reduced the lifespan of female mosquitoes by approximately 50% compared to uninfected mosquitoes [207]. Population substitution is the use of CI to displace Wolbachia-infected populations using mosquito populations that transmit the target virus; therefore, PB limits arbovirus transmission. Studies have shown that population substitution is generally effective and that Wolbachia can spread rapidly at high frequencies and remain stable over time in target populations [208–210]. Successful infection with Wolbachia usually corresponds to a significant reduction in DEF transmission in DENV-endemic areas [209–212]. In addition, Wolbachia increases mosquito resistance to DENV, CHIKV, YFV, and Plasmodium infection [213–216]. The Wolbachia wAlbB strain from Ae. albopictus similarly showed resistance to DENV after midgut colonisation in Ae. aegypti [217], and gene expression analysis showed that wAlbB induced ROS production, which in turn induced activation of the Toll pathway [96].
Conclusions
Mosquitoes, as vectors of many infectious diseases, have received extensive attention worldwide. In recent years, with the development of RNAi and transgenic and CRISPR/Cas9 technologies, there has been an increasing number of studies on mosquito immune mechanisms. This article describes the innate immune mechanisms of mosquitoes in response to pathogens, outlining the physical barriers of the mosquito salivary glands, haemocoel, midgut, and the physiological barriers of MIB, MEB, SGIB, and SGEB, which activate the Toll, Imd, and JAK-STAT immune signalling pathways through the recognition of pathogens by PRRs and PAMPs, thereby triggering cellular and humoral immune responses such as AMPs, melanisation, ROS and NO, apoptosis, autophagy, phagocytosis, and other cellular and humoral immune responses. Finally, the mechanisms and related studies of mosquito immune priming as well as symbiotic bacteria against pathogen infection are described. The study of the molecular mechanisms of mosquito innate immune responses provides new ideas for mosquito density control and the development of mosquito-borne disease control strategies. However, the mechanism of mosquito innate immunity still has many unknowns and needs to be further studied and explored; for example the lacks of knowledge about the molecular and biochemical mechanisms underlying the physiological barriers of MIB, MEB, SGIB, and SGEB; the differentiated mechanisms of innate immunity among mosquito species; the differences in immune mechanisms between larvae and adults; the mechanisms of haemocyte development and differentiation; the complex regulatory mechanisms in response to different pathogens; the exact mechanisms of immune-associated genes and effectors involved in immune regulation; and the search for new mechanisms of immunity, immune genes, and immune effectors all require further investigation. The development of gene editing and genomics and biochemical analysis techniques will further promote the study of mosquito immune mechanisms, which will provide a basis for improving mosquito-borne disease research.
Acknowledgements
Not applicable.
Abbreviations
- PRRs
Pattern recognition receptors
- PAMPs
Pathogen-associated molecular patterns
- AMPs
Antimicrobial peptides
- PO
Phenoloxidase
- ROS
Reactive oxygen species
- NO
Nitric oxide
- PM
Peritrophic membrane
- MIB
Midgut infection barrier
- MEB
Midgut escape barrier
- SGIB
Salivary gland infection barrier
- SGEB
Salivary gland escape barrier
- LACV
La Crosse virus
- EEEV
Eastern equine encephalitis virus
- JEV
Japanese encephalitis virus
- WNV
West Nile virus
- WEEV
Western equine encephalitis virus
- RVFV
Rift Valley fever virus
- TEPs
Thioester-containing proteins
- LRR
Leucine-rich repeat
- DENV
Dengue virus
- FREPs
Fibrinogen-related proteins
- FBN
Fibrillin
- CTLs
C-type lectins
- GNBPs
Gram-negative binding proteins
- PGRPs
Peptidoglycan recognition proteins
- IgSF
Immunoglobulin superfamily
- IRID
Immunoglobulin domain
- IMD
Immune deficiency
- JAK
Janus kinase
- STAT
Signal transducer and activator of transcription
- RNAi
RNA interfering
- NF-κB
Nuclear factor kappa B
- SINV
Sindbis virus
- dsRNA
Double-stranded RNA
- sRNA
Small RNA
- siRNAs
Small interfering RNAs
- miRNAs
MicroRNAs
- piRNAs
PIWI protein-interacting RNAs
- RISC
RNA-induced silencing complex
- Ago
Argonaute
- YFV
Yellow fever virus
- pre-miRNAs
MiRNA precursors
- CHIKV
Chikungunya virus
- DEFs
Defensins
- CECs
Cecropins
- GAMs
Gambicins
- NOS
Nitric oxide synthase
- SP
Serine protease
- SRPN
Serine protease inhibitor
- CLIPs
Serine protease hairpin domains
- proPOs
Phenol oxidase proteins
- PCD
Programmed cell death
- mx
Michelob-x
- ATG5
Autophagy-related gene 5
- LRP1
Lipoprotein receptor-related protein
- HDF
Haemocyte differentiation factor
- PGE2
Prostaglandin E2
- DBLOX
Double peroxidase
- TGIP
Transgenerational immune priming
- CI
Cytoplasmic incompatibility
- PB
Pathogen blocking
Author contributions
MJL, YZ, CJL, and YS conceptualized the review. MJL and YZ drafted the manuscript. MJL, YZ, JC screened literature included in the review, extracted data from articles, and analyzed the data. YQW drew pictures. All authors read and approved submission of the manuscript.
Funding
Not applicable.
Availability of data and materials
The datasets supporting the conclusions of this article are included within the article.
Declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manjin Li and Yang Zhou contributed equally to this work.
Contributor Information
Cejie Lan, Email: lancejie@126.com.
Yuan Shen, Email: sarayshen@163.com.
References
- 1.King JG. Developmental and comparative perspectives on mosquito immunity. Dev Comp Immunol. 2020;103:103458. doi: 10.1016/j.dci.2019.103458. [DOI] [PubMed] [Google Scholar]
- 2.Dimopoulos G. Insect immunity and its implication in mosquito-malaria interactions. Cell Microbiol. 2003;5:3–14. doi: 10.1046/j.1462-5822.2003.00252.x. [DOI] [PubMed] [Google Scholar]
- 3.Cirimotich CM, Dong Y, Garver LS, Sim S, Dimopoulos G. Mosquito immune defenses against Plasmodium infection. Dev Comp Immunol. 2010;34:387–395. doi: 10.1016/j.dci.2009.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang YH, Wang JM, Jiang H, Zhou Z. Research advances on immune mechanism against pathogens in mosquitoes. Chin J Vector Biol Control. 2013;24:477–482. [Google Scholar]
- 5.Hillyer JF. Mosquito immunity. Adv Exp Med Biol. 2010;708:218–238. doi: 10.1007/978-1-4419-8059-5_12. [DOI] [PubMed] [Google Scholar]
- 6.Guo XX, Wang HW. Research progress on the molecular mechanisms of mosquito innate immunity. Chin J Parasitol Parasit Dis. 2015;33:52–57. [PubMed] [Google Scholar]
- 7.Hecker H. Structure and function of midgut epithelial cells in culicidae mosquitoes (insecta, diptera) Cell Tissue Res. 1977;184:321–341. doi: 10.1007/BF00219894. [DOI] [PubMed] [Google Scholar]
- 8.Houk EJ. Midgut ultrastructure of Culex tarsalis (Diptera: Culcidae) before and after a bloodmeal. Tissue Cell. 1977;9:103–118. doi: 10.1016/0040-8166(77)90052-0. [DOI] [PubMed] [Google Scholar]
- 9.Kato N, Mueller CR, Fuchs JF, McElroy K, Wessely V, Higgs S, et al. Evaluation of the function of a type I peritrophic matrix as a physical barrier for midgut epithelium invasion by mosquito-borne pathogens in Aedes aegypti. Vector Borne Zoonotic Dis. 2008;8:701–712. doi: 10.1089/vbz.2007.0270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dong Y, Manfredini F, Dimopoulos G. Implication of the mosquito midgut microbiota in the defense against malaria parasites. PLoS Pathog. 2009;5:e1000423. doi: 10.1371/journal.ppat.1000423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Alavi Y, Arai M, Mendoza J, Tufet-Bayona M, Sinha R, Fowler K, et al. The dynamics of interactions between Plasmodium and the mosquito: a study of the infectivity of Plasmodium berghei and Plasmodium gallinaceum, and their transmission by Anopheles stephensi, Anopheles gambiae and Aedes aegypti. Int J Parasitol. 2003;33:933–943. doi: 10.1016/S0020-7519(03)00112-7. [DOI] [PubMed] [Google Scholar]
- 12.Sanders HR, Foy BD, Evans AM, Ross LS, Beaty BJ, Olson KE, et al. Sindbis virus induces transport processes and alters expression of innate immunity pathway genes in the midgut of the disease vector, Aedes aegypti. Insect Biochem Mol Biol. 2005;35:1293–1307. doi: 10.1016/j.ibmb.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 13.Houk EJ, Obie F, Hardy JL. Peritrophic membrane formation and the midgut barrier to arboviral infection in the mosquito, Culex tarsalis Coquillett (Insecta, Diptera) Acta Trop. 1979;36:39–45. [PubMed] [Google Scholar]
- 14.Hardy JL, Houk EJ, Kramer LD, Reeves WC. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Annu Rev Entomol. 1983;28:229–262. doi: 10.1146/annurev.en.28.010183.001305. [DOI] [PubMed] [Google Scholar]
- 15.Passarelli AL. Barriers to success: how baculoviruses establish efficient systemic infections. Virology. 2011;411:383–392. doi: 10.1016/j.virol.2011.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kramer LD, Hardy JL, Presser SB, Houk EJ. Dissemination barriers for western equine encephalomyelitis virus in Culex tarsalis infected after ingestion of low viral doses. Am J Trop Med Hyg. 1981;30:190–197. doi: 10.4269/ajtmh.1981.30.190. [DOI] [PubMed] [Google Scholar]
- 17.Rodrigues J, Oliveira GA, Kotsyfakis M, Dixit R, Molina-Cruz A, Jochim R, et al. An epithelial serine protease, AgESP, is required for Plasmodium invasion in the mosquito Anopheles gambiae. PLoS ONE. 2012;7:e35210. doi: 10.1371/journal.pone.0035210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rodrigues J, Brayner FA, Alves LC, Dixit R, Barillas-Mury C. Hemocyte differentiation mediates innate immune memory in Anopheles gambiae mosquitoes. Science. 2010;329:1353–1355. doi: 10.1126/science.1190689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Glenn JD, King JG, Hillyer JF. Structural mechanics of the mosquito heart and its function in bidirectional hemolymph transport. J Exp Biol. 2010;213:541–550. doi: 10.1242/jeb.035014. [DOI] [PubMed] [Google Scholar]
- 20.Lewis J, Gallichotte EN, Randall J, Glass A, Foy BD, Ebel GD, et al. Intrinsic factors driving mosquito vector competence and viral evolution: a review. Front Cell Infect Microbiol. 2023;13:1330600. doi: 10.3389/fcimb.2023.1330600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Salazar MI, Richardson JH, Sánchez-Vargas I, Olson KE, Beaty BJ. Dengue virus type 2: replication and tropisms in orally infected Aedes aegypti mosquitoes. BMC Microbiol. 2007;7:9. doi: 10.1186/1471-2180-7-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Parikh GR, Oliver JD, Bartholomay LC. A haemocyte tropism for an arbovirus. J Gen Virol. 2009;90:292–296. doi: 10.1099/vir.0.005116-0. [DOI] [PubMed] [Google Scholar]
- 23.Cheng G, Liu Y, Wang P, Xiao X. Mosquito defense strategies against viral infection. Trends Parasitol. 2016;32:177–186. doi: 10.1016/j.pt.2015.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pinto SB, Lombardo F, Koutsos AC, Waterhouse RM, McKay K, An C, et al. Discovery of Plasmodium modulators by genome-wide analysis of circulating hemocytes in Anopheles gambiae. Proc Natl Acad Sci U S A. 2009;106:21270–21275. doi: 10.1073/pnas.0909463106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Castillo JC, Robertson AE, Strand MR. Characterization of hemocytes from the mosquitoes Anopheles gambiae and Aedes aegypti. Insect Biochem Mol Biol. 2006;36:891–903. doi: 10.1016/j.ibmb.2006.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee WS, Webster JA, Madzokere ET, Stephenson EB, Herrero LJ. Mosquito antiviral defense mechanisms: a delicate balance between innate immunity and persistent viral infection. Parasit Vectors. 2019;12:165. doi: 10.1186/s13071-019-3433-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hillyer JF, Strand MR. Mosquito hemocyte-mediated immune responses. Curr Opin Insect Sci. 2014;3:14–21. doi: 10.1016/j.cois.2014.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nakatogawa S, Oda Y, Kamiya M, Kamijima T, Aizawa T, Clark KD, et al. A novel peptide mediates aggregation and migration of hemocytes from an insect. Curr Biol. 2009;19:779–785. doi: 10.1016/j.cub.2009.03.050. [DOI] [PubMed] [Google Scholar]
- 29.Aliota MT, Fuchs JF, Mayhew GF, Chen CC, Christensen BM. Mosquito transcriptome changes and filarial worm resistance in Armigeres subalbatus. BMC Genomics. 2007;8:463. doi: 10.1186/1471-2164-8-463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bartholomay LC, Mayhew GF, Fuchs JF, Rocheleau TA, Erickson SM, Aliota MT, et al. Profiling infection responses in the haemocytes of the mosquito, Aedes aegypti. Insect Mol Biol. 2007;16:761–776. doi: 10.1111/j.1365-2583.2007.00773.x. [DOI] [PubMed] [Google Scholar]
- 31.Baton LA, Robertson A, Warr E, Strand MR, Dimopoulos G. Genome-wide transcriptomic profiling of Anopheles gambiae hemocytes reveals pathogen-specific signatures upon bacterial challenge and Plasmodium berghei infection. BMC Genomics. 2009;10:257. doi: 10.1186/1471-2164-10-257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Choi YJ, Fuchs JF, Mayhew GF, Yu HE, Christensen BM. Tissue-enriched expression profiles in Aedes aegypti identify hemocyte-specific transcriptome responses to infection. Insect Biochem Mol Biol. 2012;42:729–738. doi: 10.1016/j.ibmb.2012.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Satyavathi VV, Minz A, Nagaraju J. Nodulation: an unexplored cellular defense mechanism in insects. Cell Signal. 2014;26:1753–1763. doi: 10.1016/j.cellsig.2014.02.024. [DOI] [PubMed] [Google Scholar]
- 34.Browne N, Heelan M, Kavanagh K. An analysis of the structural and functional similarities of insect hemocytes and mammalian phagocytes. Virulence. 2013;4:597–603. doi: 10.4161/viru.25906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cheng J, Wang Y, Li F, Liu J, Sun Y, Wu J. Cloning and characterization of a mannose binding C-type lectin gene from salivary gland of Aedes albopictus. Parasit Vectors. 2014;7:337. doi: 10.1186/1756-3305-7-337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cheng G, Cox J, Wang P, Krishnan MN, Dai J, Qian F, et al. A C-type lectin collaborates with a CD45 phosphatase homolog to facilitate West Nile virus infection of mosquitoes. Cell. 2010;142:714–725. doi: 10.1016/j.cell.2010.07.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu Y, Zhang F, Liu J, Xiao X, Zhang S, Qin C, et al. Transmission-blocking antibodies against mosquito C-type lectins for dengue prevention. PLoS Pathog. 2014;10:e1003931. doi: 10.1371/journal.ppat.1003931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Schnitger AK, Yassine H, Kafatos FC, Osta MA. Two C-type lectins cooperate to defend Anopheles gambiae against Gram-negative bacteria. J Biol Chem. 2009;284:17616–17624. doi: 10.1074/jbc.M808298200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thiévent K, Zilio G, Hauser G, Koella JC. Malaria load affects the activity of mosquito salivary apyrase. J Insect Physiol. 2019;116:10–16. doi: 10.1016/j.jinsphys.2019.04.003. [DOI] [PubMed] [Google Scholar]
- 40.Phattanawiboon B, Jariyapan N, Roytrakul S, Paemanee A, Sor-suwan S, Intakhan N, et al. Morphological and protein analyses of adult female salivary glands of Anopheles barbirostris species A1 (Diptera: Culicidae) Trop Biomed. 2014;31:813–827. [PubMed] [Google Scholar]
- 41.Schneider BS, Higgs S. The enhancement of arbovirus transmission and disease by mosquito saliva is associated with modulation of the host immune response. Trans R Soc Trop Med Hyg. 2008;102:400–408. doi: 10.1016/j.trstmh.2008.01.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kumar A, Srivastava P, Sirisena P, Dubey SK, Kumar R, Shrinet J, et al. Mosquito innate immunity. Insects. 2018;9:3. doi: 10.3390/insects9030095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Paulson SL, Grimstad PR, Craig GB., Jr Midgut and salivary gland barriers to La Crosse virus dissemination in mosquitoes of the Aedes triseriatus group. Med Vet Entomol. 1989;3:113–123. doi: 10.1111/j.1365-2915.1989.tb00485.x. [DOI] [PubMed] [Google Scholar]
- 44.Scott TW, Lorenz LH, Weaver SC. Susceptibility of Aedes albopictus to infection with eastern equine encephalomyelitis virus. J Am Mosq Control Assoc. 1990;6:274–278. [PubMed] [Google Scholar]
- 45.Turell MJ, Mores CN, Dohm DJ, Komilov N, Paragas J, Lee JS, et al. Laboratory transmission of Japanese encephalitis and West Nile viruses by molestus form of Culex pipiens (Diptera: Culicidae) collected in Uzbekistan in 2004. J Med Entomol. 2006;43:296–300. doi: 10.1093/jmedent/43.2.296. [DOI] [PubMed] [Google Scholar]
- 46.Romoser WS, Turell MJ, Lerdthusnee K, Neira M, Dohm D, Ludwig G, et al. Pathogenesis of Rift Valley fever virus in mosquitoes—tracheal conduits & the basal lamina as an extra-cellular barrier. Arch Virol Suppl. 2005;19:89–100. doi: 10.1007/3-211-29981-5_8. [DOI] [PubMed] [Google Scholar]
- 47.Beaty BJ, Holterman M, Tabachnick W, Shope RE, Rozhon EJ, Bishop DH. Molecular basis of bunyavirus transmission by mosquitoes: role of the middle-sized RNA segment. Science. 1981;211:1433–1435. doi: 10.1126/science.6781068. [DOI] [PubMed] [Google Scholar]
- 48.Grimstad PR, Paulson SL, Craig GB., Jr Vector competence of Aedes hendersoni (Diptera: Culicidae) for La Crosse virus and evidence of a salivary-gland escape barrier. J Med Entomol. 1985;22:447–453. doi: 10.1093/jmedent/22.4.447. [DOI] [PubMed] [Google Scholar]
- 49.Jupp PG. Culex theileri and Sindbis virus; salivary glands infection in relation to transmission. J Am Mosq Control Assoc. 1985;1:374–376. [PubMed] [Google Scholar]
- 50.Turell MJ, Britch SC, Aldridge RL, Kline DL, Boohene C, Linthicum KJ. Potential for mosquitoes (Diptera: Culicidae) from Florida to transmit Rift Valley fever virus. J Med Entomol. 2013;50:1111–1117. doi: 10.1603/ME13049. [DOI] [PubMed] [Google Scholar]
- 51.Martins GF, Pimenta PF. Structural changes in fat body of Aedes aegypti caused by aging and blood feeding. J Med Entomol. 2008;45:1102–1107. doi: 10.1093/jmedent/45.6.1102. [DOI] [PubMed] [Google Scholar]
- 52.Price DP, Nagarajan V, Churbanov A, Houde P, Milligan B, Drake LL, et al. The fat body transcriptomes of the yellow fever mosquito Aedes aegypti, pre- and post-blood meal. PLoS ONE. 2011;6:e22573. doi: 10.1371/journal.pone.0022573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Martins GF, Serrão JE, Ramalho-Ortigão JM, Pimenta PF. A comparative study of fat body morphology in five mosquito species. Mem Inst Oswaldo Cruz. 2011;106:742–747. doi: 10.1590/S0074-02762011000600015. [DOI] [PubMed] [Google Scholar]
- 54.Martins GF, Serrão JE, Ramalho-Ortigão JM, Pimenta PF. Histochemical and ultrastructural studies of the mosquito Aedes aegypti fat body: effects of aging and diet type. Microsc Res Tech. 2011;74:1032–1039. doi: 10.1002/jemt.20990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ramirez JL, Dunlap CA, Muturi EJ, Barletta ABF, Rooney AP. Entomopathogenic fungal infection leads to temporospatial modulation of the mosquito immune system. PLoS Negl Trop Dis. 2018;12:e0006433. doi: 10.1371/journal.pntd.0006433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Arrese EL, Soulages JL. Insect fat body: energy, metabolism, and regulation. Annu Rev Entomol. 2010;55:207–225. doi: 10.1146/annurev-ento-112408-085356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Martins GF, Ramalho-Ortigão JM, Lobo NF, Severson DW, McDowell MA, Pimenta PF. Insights into the transcriptome of oenocytes from Aedes aegypti pupae. Mem Inst Oswaldo Cruz. 2011;106:308–315. doi: 10.1590/S0074-02762011000300009. [DOI] [PubMed] [Google Scholar]
- 58.Goddard J. Biology of disease vectors. Emerg Infect Dis. 2005;11:1330–1. 10.3201/eid1108.050610.
- 59.Kokoza V, Ahmed A, Woon Shin S, Okafor N, Zou Z, Raikhel AS. Blocking of Plasmodium transmission by cooperative action of Cecropin A and Defensin A in transgenic Aedes aegypti mosquitoes. Proc Natl Acad Sci U S A. 2010;107:8111–8116. doi: 10.1073/pnas.1003056107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Feitosa FM, Calvo E, Merino EF, Durham AM, James AA, de Bianchi AG, et al. A transcriptome analysis of the Aedes aegypti vitellogenic fat body. J Insect Sci. 2006;6:1–26. doi: 10.1673/1536-2442(2006)6[1:ATAOTA]2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hun LV, Cheung KW, Brooks E, Zudekoff R, Luckhart S, Riehle MA. Increased insulin signaling in the Anopheles stephensi fat body regulates metabolism and enhances the host response to both bacterial challenge and Plasmodium falciparum infection. Insect Biochem Mol Biol. 2021;139:103669. doi: 10.1016/j.ibmb.2021.103669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Coutinho-Abreu IV, Zhu KY, Ramalho-Ortigao M. Transgenesis and paratransgenesis to control insect-borne diseases: current status and future challenges. Parasitol Int. 2010;59:1–8. doi: 10.1016/j.parint.2009.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gomes FM, Tyner MDW, Barletta ABF, Yenkoidiok-Douti L, Canepa G, Molina-Cruz A, et al. Proliferation of DBLOX peroxidase-expressing oenocytes maintains innate immune memory in primed mosquitoes. bioRxiv. 2020. [DOI] [PMC free article] [PubMed]
- 64.Blandin S, Shiao SH, Moita LF, Janse CJ, Waters AP, Kafatos FC, et al. Complement-like protein TEP1 is a determinant of vectorial capacity in the malaria vector Anopheles gambiae. Cell. 2004;116:661–670. doi: 10.1016/S0092-8674(04)00173-4. [DOI] [PubMed] [Google Scholar]
- 65.Cheng G, Liu L, Wang P, Zhang Y, Zhao YO, Colpitts TM, et al. An in vivo transfection approach elucidates a role for Aedes aegypti thioester-containing proteins in flaviviral infection. PLoS ONE. 2011;6:e22786. doi: 10.1371/journal.pone.0022786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lagueux M, Perrodou E, Levashina EA, Capovilla M, Hoffmann JA. Constitutive expression of a complement-like protein in toll and JAK gain-of-function mutants of Drosophila. Proc Natl Acad Sci U S A. 2000;97:11427–11432. doi: 10.1073/pnas.97.21.11427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Levashina EA, Moita LF, Blandin S, Vriend G, Lagueux M, Kafatos FC. Conserved role of a complement-like protein in phagocytosis revealed by dsRNA knockout in cultured cells of the mosquito, Anopheles gambiae. Cell. 2001;104:709–718. doi: 10.1016/S0092-8674(01)00267-7. [DOI] [PubMed] [Google Scholar]
- 68.Fraiture M, Baxter RH, Steinert S, Chelliah Y, Frolet C, Quispe-Tintaya W, et al. Two mosquito LRR proteins function as complement control factors in the TEP1-mediated killing of Plasmodium. Cell Host Microbe. 2009;5:273–284. doi: 10.1016/j.chom.2009.01.005. [DOI] [PubMed] [Google Scholar]
- 69.Baxter RH, Steinert S, Chelliah Y, Volohonsky G, Levashina EA, Deisenhofer J. A heterodimeric complex of the LRR proteins LRIM1 and APL1C regulates complement-like immunity in Anopheles gambiae. Proc Natl Acad Sci U S A. 2010;107:16817–16822. doi: 10.1073/pnas.1010575107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Povelones M, Waterhouse RM, Kafatos FC, Christophides GK. Leucine-rich repeat protein complex activates mosquito complement in defense against Plasmodium parasites. Science. 2009;324:258–261. doi: 10.1126/science.1171400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Blandin SA, Wang-Sattler R, Lamacchia M, Gagneur J, Lycett G, Ning Y, et al. Dissecting the genetic basis of resistance to malaria parasites in Anopheles gambiae. Science. 2009;326:147–150. doi: 10.1126/science.1175241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Waterhouse RM, Kriventseva EV, Meister S, Xi Z, Alvarez KS, Bartholomay LC, et al. Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes. Science. 2007;316:1738–1743. doi: 10.1126/science.1139862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dong Y, Dimopoulos G. Anopheles fibrinogen-related proteins provide expanded pattern recognition capacity against bacteria and malaria parasites. J Biol Chem. 2009;284:9835–9844. doi: 10.1074/jbc.M807084200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ferguson HM, Read AF. Why is the effect of malaria parasites on mosquito survival still unresolved? Trends Parasitol. 2002;18:256–261. doi: 10.1016/S1471-4922(02)02281-X. [DOI] [PubMed] [Google Scholar]
- 75.Li J, Wang X, Zhang G, Githure JI, Yan G, James AA. Genome-block expression-assisted association studies discover malaria resistance genes in Anopheles gambiae. Proc Natl Acad Sci U S A. 2013;110:20675–20680. doi: 10.1073/pnas.1321024110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Baker DA, Nolan T, Fischer B, Pinder A, Crisanti A, Russell S. A comprehensive gene expression atlas of sex- and tissue-specificity in the malaria vector, Anopheles gambiae. BMC Genomics. 2011;12:296. [DOI] [PMC free article] [PubMed]
- 77.Dong Y, Taylor HE, Dimopoulos G. AgDscam, a hypervariable immunoglobulin domain-containing receptor of the Anopheles gambiae innate immune system. PLoS Biol. 2006;4:e229. doi: 10.1371/journal.pbio.0040229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Oliveira SB, Ibraim IC, Tadei WP, Ruiz JC, Nahum LA, Brito CF, et al. Identification of a fibrinogen-related protein (FBN9) gene in neotropical anopheline mosquitoes. Malar J. 2011;10:21. doi: 10.1186/1475-2875-10-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ning YY, You MS, Wang CS. Advances in the mechanisms of insect immune recognition and pathogen immune escape. Acta Entomol Sin. 2009;52:567–575. [Google Scholar]
- 80.Osta MA, Christophides GK, Kafatos FC. Effects of mosquito genes on Plasmodium development. Science. 2004;303:2030–2032. doi: 10.1126/science.1091789. [DOI] [PubMed] [Google Scholar]
- 81.Dimopoulos G, Richman A, Müller HM, Kafatos FC. Molecular immune responses of the mosquito Anopheles gambiae to bacteria and malaria parasites. Proc Natl Acad Sci U S A. 1997;94:11508–11513. doi: 10.1073/pnas.94.21.11508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Warr E, Das S, Dong Y, Dimopoulos G. The Gram-negative bacteria-binding protein gene family: its role in the innate immune system of Anopheles gambiae and in anti-Plasmodium defence. Insect Mol Biol. 2008;17:39–51. doi: 10.1111/j.1365-2583.2008.00778.x. [DOI] [PubMed] [Google Scholar]
- 83.Wang S, Beerntsen BT. Insights into the different functions of multiple peptidoglycan recognition proteins in the immune response against bacteria in the mosquito, Armigeres subalbatus. Insect Biochem Mol Biol. 2013;43:533–543. doi: 10.1016/j.ibmb.2013.03.004. [DOI] [PubMed] [Google Scholar]
- 84.Garver LS, Xi Z, Dimopoulos G. Immunoglobulin superfamily members play an important role in the mosquito immune system. Dev Comp Immunol. 2008;32:519–531. doi: 10.1016/j.dci.2007.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spätzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996;86:973–983. doi: 10.1016/S0092-8674(00)80172-5. [DOI] [PubMed] [Google Scholar]
- 86.Rutschmann S, Kilinc A, Ferrandon D. Cutting edge: the toll pathway is required for resistance to gram-positive bacterial infections in Drosophila. J Immunol. 2002;168:1542–1546. doi: 10.4049/jimmunol.168.4.1542. [DOI] [PubMed] [Google Scholar]
- 87.Zambon RA, Nandakumar M, Vakharia VN, Wu LP. The Toll pathway is important for an antiviral response in Drosophila. Proc Natl Acad Sci U S A. 2005;102:7257–7262. doi: 10.1073/pnas.0409181102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Frolet C, Thoma M, Blandin S, Hoffmann JA, Levashina EA. Boosting NF-kappaB-dependent basal immunity of Anopheles gambiae aborts development of Plasmodium berghei. Immunity. 2006;25:677–685. doi: 10.1016/j.immuni.2006.08.019. [DOI] [PubMed] [Google Scholar]
- 89.Zou Z, Shin SW, Alvarez KS, Bian G, Kokoza V, Raikhel AS. Mosquito RUNX4 in the immune regulation of PPO gene expression and its effect on avian malaria parasite infection. Proc Natl Acad Sci U S A. 2008;105:18454–18459. doi: 10.1073/pnas.0804658105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Bian G, Shin SW, Cheon HM, Kokoza V, Raikhel AS. Transgenic alteration of Toll immune pathway in the female mosquito Aedes aegypti. Proc Natl Acad Sci U S A. 2005;102:13568–13573. doi: 10.1073/pnas.0502815102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Xi Z, Ramirez JL, Dimopoulos G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008;4:e1000098. doi: 10.1371/journal.ppat.1000098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shin SW, Kokoza V, Bian G, Cheon HM, Kim YJ, Raikhel AS. REL1, a homologue of Drosophila dorsal, regulates toll antifungal immune pathway in the female mosquito Aedes aegypti. J Biol Chem. 2005;280:16499–16507. doi: 10.1074/jbc.M500711200. [DOI] [PubMed] [Google Scholar]
- 93.Luplertlop N, Surasombatpattana P, Patramool S, Dumas E, Wasinpiyamongkol L, Saune L, et al. Induction of a peptide with activity against a broad spectrum of pathogens in the Aedes aegypti salivary gland, following Infection with Dengue Virus. PLoS Pathog. 2011;7:e1001252. doi: 10.1371/journal.ppat.1001252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sim S, Ramirez JL, Dimopoulos G. Dengue virus infection of the Aedes aegypti salivary gland and chemosensory apparatus induces genes that modulate infection and blood-feeding behavior. PLoS Pathog. 2012;8:e1002631. doi: 10.1371/journal.ppat.1002631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ramirez JL, Dimopoulos G. The Toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev Comp Immunol. 2010;34 6:625–9. [DOI] [PMC free article] [PubMed]
- 96.Pan X, Zhou G, Wu J, Bian G, Lu P, Raikhel AS, et al. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc Natl Acad Sci U S A. 2012;109:E23–31. doi: 10.1073/pnas.1116932108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Bartholomay LC, Waterhouse RM, Mayhew GF, Campbell CL, Michel K, Zou Z, et al. Pathogenomics of Culex quinquefasciatus and meta-analysis of infection responses to diverse pathogens. Science. 2010;330:88–90. doi: 10.1126/science.1193162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Colpitts TM, Cox J, Vanlandingham DL, Feitosa FM, Cheng G, Kurscheid S, et al. Alterations in the Aedes aegypti transcriptome during infection with West Nile, dengue and yellow fever viruses. PLoS Pathog. 2011;7:e1002189. doi: 10.1371/journal.ppat.1002189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Barletta ABF, Saha B, Trisnadi N, Talyuli OAC, Raddi G, Barillas-Mury C. Hemocyte differentiation to the megacyte lineage enhances mosquito immunity against Plasmodium. Elife. 2022;11:e81116. 10.7554/eLife.81116. [DOI] [PMC free article] [PubMed]
- 100.Meister S, Kanzok SM, Zheng XL, Luna C, Li TR, Hoa NT, et al. Immune signaling pathways regulating bacterial and malaria parasite infection of the mosquito Anopheles gambiae. Proc Natl Acad Sci U S A. 2005;102:11420–11425. doi: 10.1073/pnas.0504950102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Garver LS, Dong Y, Dimopoulos G. Caspar controls resistance to Plasmodium falciparum in diverse anopheline species. PLoS Pathog. 2009;5:e1000335. doi: 10.1371/journal.ppat.1000335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Garver LS, Bahia AC, Das S, Souza-Neto JA, Shiao J, Dong Y, et al. Anopheles Imd pathway factors and effectors in infection intensity-dependent anti-Plasmodium action. PLoS Pathog. 2012;8:e1002737. doi: 10.1371/journal.ppat.1002737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Dong Y, Das S, Cirimotich C, Souza-Neto JA, McLean KJ, Dimopoulos G. Engineered Anopheles immunity to Plasmodium infection. PLoS Pathog. 2011;7:e1002458. doi: 10.1371/journal.ppat.1002458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Barletta AB, Nascimento-Silva MC, Talyuli OA, Oliveira JH, Pereira LO, Oliveira PL, et al. Microbiota activates IMD pathway and limits Sindbis infection in Aedes aegypti. Parasit Vectors. 2017;10:103. doi: 10.1186/s13071-017-2040-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Fragkoudis R, Chi Y, Siu RW, Barry G, Attarzadeh-Yazdi G, Merits A, et al. Semliki Forest virus strongly reduces mosquito host defence signaling. Insect Mol Biol. 2008;17:647–656. doi: 10.1111/j.1365-2583.2008.00834.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Sim S, Jupatanakul N, Ramirez JL, Kang S, Romero-Vivas CM, Mohammed H, et al. Transcriptomic profiling of diverse Aedes aegypti strains reveals increased basal-level immune activation in dengue virus-refractory populations and identifies novel virus-vector molecular interactions. PLoS Negl Trop Dis. 2013;7:e2295. doi: 10.1371/journal.pntd.0002295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Barillas-Mury C, Han YS, Seeley D, Kafatos FC. Anopheles gambiae Ag-STAT, a new insect member of the STAT family, is activated in response to bacterial infection. EMBO J. 1999;18:959–967. doi: 10.1093/emboj/18.4.959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Souza-Neto JA, Sim S, Dimopoulos G. An evolutionary conserved function of the JAK-STAT pathway in anti-dengue defense. Proc Natl Acad Sci U S A. 2009;106:17841–17846. doi: 10.1073/pnas.0905006106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bahia AC, Kubota MS, Tempone AJ, Araújo HR, Guedes BA, Orfanó AS, et al. The JAK-STAT pathway controls Plasmodium vivax load in early stages of Anopheles aquasalis infection. PLoS Negl Trop Dis. 2011;5:e1317. doi: 10.1371/journal.pntd.0001317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Ding SW, Voinnet O. Antiviral immunity directed by small RNAs. Cell. 2007;130:413–426. doi: 10.1016/j.cell.2007.07.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Nandety RS, Kuo YW, Nouri S, Falk BW. Emerging strategies for RNA interference (RNAi) applications in insects. Bioengineered. 2015;6:8–19. doi: 10.4161/21655979.2014.979701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Deshpande G, Calhoun G, Schedl P. Drosophila argonaute-2 is required early in embryogenesis for the assembly of centric/centromeric heterochromatin, nuclear division, nuclear migration, and germ-cell formation. Genes Dev. 2005;19:1680–1685. doi: 10.1101/gad.1316805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Lu C, Tej SS, Luo S, Haudenschild CD, Meyers BC, Green PJ. Elucidation of the small RNA component of the transcriptome. Science. 2005;309:1567–1569. doi: 10.1126/science.1114112. [DOI] [PubMed] [Google Scholar]
- 114.Zhang H, Kolb FA, Jaskiewicz L, Westhof E, Filipowicz W. Single processing center models for human Dicer and bacterial RNase III. Cell. 2004;118:57–68. doi: 10.1016/j.cell.2004.06.017. [DOI] [PubMed] [Google Scholar]
- 115.Liu Q, Rand TA, Kalidas S, Du F, Kim HE, Smith DP, et al. R2D2, a bridge between the initiation and effector steps of the Drosophila RNAi pathway. Science. 2003;301:1921–1925. doi: 10.1126/science.1088710. [DOI] [PubMed] [Google Scholar]
- 116.Li WX, Li H, Lu R, Li F, Dus M, Atkinson P, et al. Interferon antagonist proteins of influenza and vaccinia viruses are suppressors of RNA silencing. Proc Natl Acad Sci U S A. 2004;101:1350–1355. doi: 10.1073/pnas.0308308100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Samuel GH, Wiley MR, Badawi A, Adelman ZN, Myles KM. Yellow fever virus capsid protein is a potent suppressor of RNA silencing that binds double-stranded RNA. Proc Natl Acad Sci U S A. 2016;113:13863–13868. doi: 10.1073/pnas.1600544113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Sánchez-Vargas I, Scott JC, Poole-Smith BK, Franz AW, Barbosa-Solomieu V, Wilusz J, et al. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquito's RNA interference pathway. PLoS Pathog. 2009;5:e1000299. doi: 10.1371/journal.ppat.1000299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Khoo CC, Piper J, Sanchez-Vargas I, Olson KE, Franz AW. The RNA interference pathway affects midgut infection- and escape barriers for Sindbis virus in Aedes aegypti. BMC Microbiol. 2010;10:130. doi: 10.1186/1471-2180-10-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Basu S, Aryan A, Overcash JM, Samuel GH, Anderson MA, Dahlem TJ, et al. Silencing of end-joining repair for efficient site-specific gene insertion after TALEN/CRISPR mutagenesis in Aedes aegypti. Proc Natl Acad Sci U S A. 2015;112:4038–4043. doi: 10.1073/pnas.1502370112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Keene KM, Foy BD, Sanchez-Vargas I, Beaty BJ, Blair CD, Olson KE. RNA interference acts as a natural antiviral response to O'nyong-nyong virus (Alphavirus; Togaviridae) infection of Anopheles gambiae. Proc Natl Acad Sci U S A. 2004;101:17240–17245. doi: 10.1073/pnas.0406983101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Abraham EG, Pinto SB, Ghosh A, Vanlandingham DL, Budd A, Higgs S, et al. An immune-responsive serpin, SRPN6, mediates mosquito defense against malaria parasites. Proc Natl Acad Sci U S A. 2005;102:16327–16332. doi: 10.1073/pnas.0508335102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Lee YS, Nakahara K, Pham JW, Kim K, He Z, Sontheimer EJ, et al. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell. 2004;117:69–81. doi: 10.1016/S0092-8674(04)00261-2. [DOI] [PubMed] [Google Scholar]
- 124.Lucas KJ, Zhao B, Liu S, Raikhel AS. Regulation of physiological processes by microRNAs in insects. Current opinion in insect science. 2015;11:1–7. doi: 10.1016/j.cois.2015.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Saldaña MA, Etebari K, Hart CE, Widen SG, Wood TG, Thangamani S, et al. Zika virus alters the microRNA expression profile and elicits an RNAi response in Aedes aegypti mosquitoes. PLoS Negl Trop Dis. 2017;11:e0005760. doi: 10.1371/journal.pntd.0005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Campbell CL, Harrison T, Hess AM, Ebel GD. MicroRNA levels are modulated in Aedes aegypti after exposure to Dengue-2. Insect Mol Biol. 2014;23:132–139. doi: 10.1111/imb.12070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Hussain M, Asgari S. MicroRNA-like viral small RNA from Dengue virus 2 autoregulates its replication in mosquito cells. Proc Natl Acad Sci U S A. 2014;111:2746–2751. doi: 10.1073/pnas.1320123111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Slonchak A, Hussain M, Torres S, Asgari S, Khromykh AA. Expression of mosquito microRNA Aae-miR-2940-5p is downregulated in response to West Nile virus infection to restrict viral replication. J Virol. 2014;88:8457–8467. doi: 10.1128/JVI.00317-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hussain M, Torres S, Schnettler E, Funk A, Grundhoff A, Pijlman GP, et al. West Nile virus encodes a microRNA-like small RNA in the 3' untranslated region which up-regulates GATA4 mRNA and facilitates virus replication in mosquito cells. Nucleic Acids Res. 2012;40:2210–2223. doi: 10.1093/nar/gkr848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Su J, Li C, Zhang Y, Yan T, Zhu X, Zhao M, et al. Identification of microRNAs expressed in the midgut of Aedes albopictus during dengue infection. Parasit Vectors. 2017;10:63. doi: 10.1186/s13071-017-1966-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Yan H, Zhou Y, Liu Y, Deng Y, Chen X. miR-252 of the Asian tiger mosquito Aedes albopictus regulates dengue virus replication by suppressing the expression of the dengue virus envelope protein. J Med Virol. 2014;86:1428–1436. doi: 10.1002/jmv.23815. [DOI] [PubMed] [Google Scholar]
- 132.Su J, Wang G, Li C, Xing D, Yan T, Zhu X, et al. Screening for differentially expressed miRNAs in Aedes albopictus (Diptera: Culicidae) exposed to DENV-2 and their effect on replication of DENV-2 in C6/36 cells. Parasit Vectors. 2019;12:44. doi: 10.1186/s13071-018-3261-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ku HY, Lin H. PIWI proteins and their interactors in piRNA biogenesis, germline development and gene expression. Natl Sci Rev. 2014;1:205–218. doi: 10.1093/nsr/nwu014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Shirayama M, Seth M, Lee HC, Gu W, Ishidate T, Conte D, Jr., et al. piRNAs initiate an epigenetic memory of nonself RNA in the C. elegans germline. Cell. 2012;150:65–77. [DOI] [PMC free article] [PubMed]
- 135.Miesen P, Girardi E, van Rij RP. Distinct sets of PIWI proteins produce arbovirus and transposon-derived piRNAs in Aedes aegypti mosquito cells. Nucleic Acids Res. 2015;43:6545–6556. doi: 10.1093/nar/gkv590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Siomi MC, Sato K, Pezic D, Aravin AA. PIWI-interacting small RNAs: the vanguard of genome defence. Nat Rev Mol Cell Biol. 2011;12:246–258. doi: 10.1038/nrm3089. [DOI] [PubMed] [Google Scholar]
- 137.Morazzani EM, Wiley MR, Murreddu MG, Adelman ZN, Myles KM. Production of virus-derived ping-pong-dependent piRNA-like small RNAs in the mosquito soma. PLoS Pathog. 2012;8:e1002470. doi: 10.1371/journal.ppat.1002470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Miesen P, Joosten J, van Rij RP. PIWIs go viral: arbovirus-derived piRNAs in vector mosquitoes. PLoS Pathog. 2016;12:e1006017. doi: 10.1371/journal.ppat.1006017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Varjak M, Maringer K, Watson M, Sreenu VB, Fredericks AC, Pondeville E, et al. Aedes aegypti Piwi4 Is a noncanonical PIWI protein involved in antiviral responses. mSphere. 2017;2 3. [DOI] [PMC free article] [PubMed]
- 140.Girardi E, Miesen P, Pennings B, Frangeul L, Saleh MC, van Rij RP. Histone-derived piRNA biogenesis depends on the ping-pong partners Piwi5 and Ago3 in Aedes aegypti. Nucleic Acids Res. 2017;45:4881–4892. doi: 10.1093/nar/gkw1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Schnettler E, Donald CL, Human S, Watson M, Siu RWC, McFarlane M, et al. Knockdown of piRNA pathway proteins results in enhanced Semliki Forest virus production in mosquito cells. J Gen Virol. 2013;94:1680–1689. doi: 10.1099/vir.0.053850-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Bartholomay LC, Cho WL, Rocheleau TA, Boyle JP, Beck ET, Fuchs JF, et al. Description of the transcriptomes of immune response-activated hemocytes from the mosquito vectors Aedes aegypti and Armigeres subalbatus. Infect Immun. 2004;72:4114–4126. doi: 10.1128/IAI.72.7.4114-4126.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Blandin S, Moita LF, Köcher T, Wilm M, Kafatos FC, Levashina EA. Reverse genetics in the mosquito Anopheles gambiae: targeted disruption of the Defensin gene. EMBO Rep. 2002;3:852–856. doi: 10.1093/embo-reports/kvf180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Xiao X, Liu Y, Zhang X, Wang J, Li Z, Pang X, et al. Complement-related proteins control the flavivirus infection of Aedes aegypti by inducing antimicrobial peptides. PLoS Pathog. 2014;10:e1004027. doi: 10.1371/journal.ppat.1004027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Bartholomay LC, Fuchs JF, Cheng LL, Beck ET, Vizioli J, Lowenberger C, et al. Reassessing the role of defensin in the innate immune response of the mosquito, Aedes aegypti. Insect Mol Biol. 2004;13:125–132. doi: 10.1111/j.0962-1075.2004.00467.x. [DOI] [PubMed] [Google Scholar]
- 146.Magalhaes T, Leandro DC, Ayres CF. Knock-down of REL2, but not defensin A, augments Aedes aegypti susceptibility to Bacillus subtilis and Escherichia coli. Acta Trop. 2010;113:167–173. doi: 10.1016/j.actatropica.2009.10.013. [DOI] [PubMed] [Google Scholar]
- 147.Kim W, Koo H, Richman AM, Seeley D, Vizioli J, Klocko AD, et al. Ectopic expression of a cecropin transgene in the human malaria vector mosquito Anopheles gambiae (Diptera: Culicidae): effects on susceptibility to Plasmodium. J Med Entomol. 2004;41:447–455. doi: 10.1603/0022-2585-41.3.447. [DOI] [PubMed] [Google Scholar]
- 148.Dong Y, Aguilar R, Xi Z, Warr E, Mongin E, Dimopoulos G. Anopheles gambiae immune responses to human and rodent Plasmodium parasite species. PLoS Pathog. 2006;2:e52. doi: 10.1371/journal.ppat.0020052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhang R, Zhu Y, Pang X, Xiao X, Zhang R, Cheng G. Regulation of antimicrobial peptides in Aedes aegypti Aag2 cells. Front Cell Infect Microbiol. 2017;7:22. doi: 10.3389/fcimb.2017.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hillyer JF, Estévez-Lao TY. Nitric oxide is an essential component of the hemocyte-mediated mosquito immune response against bacteria. Dev Comp Immunol. 2010;34:141–149. doi: 10.1016/j.dci.2009.08.014. [DOI] [PubMed] [Google Scholar]
- 151.Molina-Cruz A, DeJong RJ, Charles B, Gupta L, Kumar S, Jaramillo-Gutierrez G, et al. Reactive oxygen species modulate Anopheles gambiae immunity against bacteria and Plasmodium. J Biol Chem. 2008;283:3217–3223. doi: 10.1074/jbc.M705873200. [DOI] [PubMed] [Google Scholar]
- 152.Kumar S, Christophides GK, Cantera R, Charles B, Han YS, Meister S, et al. The role of reactive oxygen species on Plasmodium melanotic encapsulation in Anopheles gambiae. Proc Natl Acad Sci U S A. 2003;100:14139–14144. doi: 10.1073/pnas.2036262100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Lanz-Mendoza H, Hernández-Martínez S, Ku-López M, Rodríguez Mdel C, Herrera-Ortiz A, Rodríguez MH. Superoxide anion in Anopheles albimanus hemolymph and midgut is toxic to Plasmodium berghei ookinetes. J Parasitol. 2002;88:702–706. doi: 10.1645/0022-3395(2002)088[0702:SAIAAH]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 154.Oliveira JH, Gonçalves RL, Lara FA, Dias FA, Gandara AC, Menna-Barreto RF, et al. Blood meal-derived heme decreases ROS levels in the midgut of Aedes aegypti and allows proliferation of intestinal microbiota. PLoS Pathog. 2011;7:e1001320. doi: 10.1371/journal.ppat.1001320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Beerntsen BT, James AA, Christensen BM. Genetics of mosquito vector competence. Microbiol Mol Biol Rev. 2000;64:115–137. doi: 10.1128/MMBR.64.1.115-137.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Feng XY. Study on immune genes polymorphism in the malaria mosqutio Anopheles sinensis (Diptera: Culicidae); Naval Medical University, 2012.
- 157.Gulley MM, Zhang X, Michel K. The roles of serpins in mosquito immunology and physiology. J Insect Physiol. 2013;59:138–147. doi: 10.1016/j.jinsphys.2012.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.An C, Hiromasa Y, Zhang X, Lovell S, Zolkiewski M, Tomich JM, et al. Biochemical characterization of Anopheles gambiae SRPN6, a malaria parasite invasion marker in mosquitoes. PLoS ONE. 2012;7:e48689. doi: 10.1371/journal.pone.0048689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Povelones M, Bhagavatula L, Yassine H, Tan LA, Upton LM, Osta MA, et al. The CLIP-domain serine protease homolog SPCLIP1 regulates complement recruitment to microbial surfaces in the malaria mosquito Anopheles gambiae. PLoS Pathog. 2013;9:e1003623. doi: 10.1371/journal.ppat.1003623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Blumberg BJ, Trop S, Das S, Dimopoulos G. Bacteria- and IMD pathway-independent immune defenses against Plasmodium falciparum in Anopheles gambiae. PLoS ONE. 2013;8:e72130. doi: 10.1371/journal.pone.0072130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Fuchs S, Behrends V, Bundy JG, Crisanti A, Nolan T. Phenylalanine metabolism regulates reproduction and parasite melanization in the malaria mosquito. PLoS ONE. 2014;9:e84865. doi: 10.1371/journal.pone.0084865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Rodriguez-Andres J, Rani S, Varjak M, Chase-Topping ME, Beck MH, Ferguson MC, et al. Phenoloxidase activity acts as a mosquito innate immune response against infection with Semliki Forest virus. PLoS Pathog. 2012;8:e1002977. doi: 10.1371/journal.ppat.1002977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Upton JW, Chan FK-M. Staying alive: cell death in antiviral immunity. Mol Cell. 2014;54:273–80. [DOI] [PMC free article] [PubMed]
- 164.Lamkanfi M, Dixit VM. Manipulation of host cell death pathways during microbial infections. Cell Host Microbe. 2010;8:44–54. doi: 10.1016/j.chom.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 165.Liu B, Behura SK, Clem RJ, Schneemann A, Becnel J, Severson DW, et al. P53-mediated rapid induction of apoptosis conveys resistance to viral infection in Drosophila melanogaster. PLoS Pathog. 2013;9:e1003137. doi: 10.1371/journal.ppat.1003137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Zhou L, Jiang G, Chan G, Santos CP, Severson DW, Xiao L. Michelob_x is the missing inhibitor of apoptosis protein antagonist in mosquito genomes. EMBO Rep. 2005;6:769–774. doi: 10.1038/sj.embor.7400473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Liu Q, Clem RJ. Defining the core apoptosis pathway in the mosquito disease vector Aedes aegypti: the roles of iap1, ark, dronc, and effector caspases. Apoptosis. 2011;16:105–113. doi: 10.1007/s10495-010-0558-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Vaidyanathan R, Scott TW. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis. 2006;11:1643–1651. doi: 10.1007/s10495-006-8783-y. [DOI] [PubMed] [Google Scholar]
- 169.Girard YA, Schneider BS, McGee CE, Wen J, Han VC, Popov V, et al. Salivary gland morphology and virus transmission during long-term cytopathologic West Nile virus infection in Culex mosquitoes. Am J Trop Med Hyg. 2007;76:118–128. doi: 10.4269/ajtmh.2007.76.118. [DOI] [PubMed] [Google Scholar]
- 170.Ocampo CB, Caicedo PA, Jaramillo G, Ursic Bedoya R, Baron O, Serrato IM, et al. Differential expression of apoptosis related genes in selected strains of Aedes aegypti with different susceptibilities to dengue virus. PLoS ONE. 2013;8:e61187. doi: 10.1371/journal.pone.0061187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Wang H, Gort T, Boyle DL, Clem RJ. Effects of manipulating apoptosis on Sindbis virus infection of Aedes aegypti mosquitoes. J Virol. 2012;86:6546–6554. doi: 10.1128/JVI.00125-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Deretic V, Levine B. Autophagy, immunity, and microbial adaptations. Cell Host Microbe. 2009;5:527–549. doi: 10.1016/j.chom.2009.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Eng MW, van Zuylen MN, Severson DW. Apoptosis-related genes control autophagy and influence DENV-2 infection in the mosquito vector, Aedes aegypti. Insect Biochem Mol Biol. 2016;76:70–83. doi: 10.1016/j.ibmb.2016.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Hillyer JF, Schmidt SL, Christensen BM. Rapid phagocytosis and melanization of bacteria and Plasmodium sporozoites by hemocytes of the mosquito Aedes aegypti. J Parasitol. 2003;89:62–69. doi: 10.1645/0022-3395(2003)089[0062:RPAMOB]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 175.Hillyer JF, Schmidt SL, Christensen BM. Hemocyte-mediated phagocytosis and melanization in the mosquito Armigeres subalbatus following immune challenge by bacteria. Cell Tissue Res. 2003;313:117–127. doi: 10.1007/s00441-003-0744-y. [DOI] [PubMed] [Google Scholar]
- 176.Hillyer JF, Schmidt SL, Fuchs JF, Boyle JP, Christensen BM. Age-associated mortality in immune challenged mosquitoes (Aedes aegypti) correlates with a decrease in haemocyte numbers. Cell Microbiol. 2005;7:39–51. doi: 10.1111/j.1462-5822.2004.00430.x. [DOI] [PubMed] [Google Scholar]
- 177.Moita LF, Wang-Sattler R, Michel K, Zimmermann T, Blandin S, Levashina EA, et al. In vivo identification of novel regulators and conserved pathways of phagocytosis in A. gambiae. Immunity. 2005;23:65–73. [DOI] [PubMed]
- 178.Moita LF, Vriend G, Mahairaki V, Louis C, Kafatos FC. Integrins of Anopheles gambiae and a putative role of a new beta integrin, BINT2, in phagocytosis of E. coli. Insect Biochem Mol Biol. 2006;36:282–90. [DOI] [PubMed]
- 179.Sandiford SL, Dong Y, Pike A, Blumberg BJ, Bahia AC, Dimopoulos G. Cytoplasmic actin is an extracellular insect immune factor which is secreted upon immune challenge and mediates phagocytosis and direct killing of bacteria, and is a Plasmodium antagonist. PLoS Pathog. 2015;11:e1004631. doi: 10.1371/journal.ppat.1004631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Ramirez JL, de Almeida OG, Calvo E, Dalli J, Colas RA, Serhan CN, et al. A mosquito lipoxin/lipocalin complex mediates innate immune priming in Anopheles gambiae. Nat Commun. 2015;6:7403. doi: 10.1038/ncomms8403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Barletta ABF, Trisnadi N, Ramirez JL, Barillas-Mury C. Mosquito midgut prostaglandin release establishes systemic immune priming. iScience. 2019;19:54–62. [DOI] [PMC free article] [PubMed]
- 182.Gomes FM, Tyner MDW, Barletta ABF, Saha B, Yenkoidiok-Douti L, Canepa GE, et al. Double peroxidase and histone acetyltransferase AgTip60 maintain innate immune memory in primed mosquitoes. Proc Natl Acad Sci U S A. 2021;118:e2114242118. [DOI] [PMC free article] [PubMed]
- 183.Vargas V, Moreno-García M, Duarte-Elguea E, Lanz-Mendoza H. Limited specificity in the injury and infection priming against bacteria in Aedes aegypti mosquitoes. Front Microbiol. 2016;7:975. doi: 10.3389/fmicb.2016.00975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Kulkarni A, Pandey A, Trainor P, Carlisle S, Chhilar JS, Yu W, et al. Trained immunity in Anopheles gambiae: antibacterial immunity is enhanced by priming via sugar meal supplemented with a single gut symbiotic bacterial strain. Front Microbiol. 2021;12:649213. doi: 10.3389/fmicb.2021.649213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Moreno-García M, Vargas V, Ramírez-Bello I, Hernández-Martínez G, Lanz-Mendoza H. Bacterial exposure at the larval stage induced sexual immune dimorphism and priming in adult Aedes aegypti mosquitoes. PLoS ONE. 2015;10:e0133240. doi: 10.1371/journal.pone.0133240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Laureti M, Lee RX, Bennett A, Wilson LA, Sy VE, Kohl A, et al. Rift valley fever virus primes immune responses in Aedes aegypti cells. Pathogens (Basel, Switzerland). 2023;12:563. [DOI] [PMC free article] [PubMed]
- 187.Dennison NJ, Jupatanakul N, Dimopoulos G. The mosquito microbiota influences vector competence for human pathogens. Curr Opin Insect Sci. 2014;3:6–13. doi: 10.1016/j.cois.2014.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Pan X, Pike A, Joshi D, Bian G, McFadden MJ, Lu P, et al. The bacterium Wolbachia exploits host innate immunity to establish a symbiotic relationship with the dengue vector mosquito Aedes aegypti. ISME J. 2018;12:277–288. doi: 10.1038/ismej.2017.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Sim S, Dimopoulos G. Dengue virus inhibits immune responses in Aedes aegypti cells. PLoS ONE. 2010;5:e10678. doi: 10.1371/journal.pone.0010678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Dodson BL, Hughes GL, Paul O, Matacchiero AC, Kramer LD, Rasgon JL. Wolbachia enhances West Nile virus (WNV) infection in the mosquito Culex tarsalis. PLoS Negl Trop Dis. 2014;8:e2965. doi: 10.1371/journal.pntd.0002965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Hughes GL, Vega-Rodriguez J, Xue P, Rasgon JL. Wolbachia strain wAlbB enhances infection by the rodent malaria parasite Plasmodium berghei in Anopheles gambiae mosquitoes. Appl Environ Microbiol. 2012;78:1491–1495. doi: 10.1128/AEM.06751-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Mondotte JA, Gausson V, Frangeul L, Suzuki Y, Vazeille M, Mongelli V, et al. Evidence for long-lasting transgenerational antiviral immunity in insects. Cell Rep. 2020;33:108506. doi: 10.1016/j.celrep.2020.108506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Cime-Castillo J, Vargas V, Hernández-Tablas JM, Quezada-Ruiz E, Díaz G, Lanz-Mendoza H. The costs of transgenerational immune priming for homologous and heterologous infections with different serotypes of dengue virus in Aedes aegypti mosquitoes. Front Immunol. 2023;14:1286831. doi: 10.3389/fimmu.2023.1286831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Hegde S, Rasgon JL, Hughes GL. The microbiome modulates arbovirus transmission in mosquitoes. Curr Opin Virol. 2015;15:97–102. doi: 10.1016/j.coviro.2015.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Jupatanakul N, Sim S, Dimopoulos G. The insect microbiome modulates vector competence for arboviruses. Viruses. 2014;6:4294–4313. doi: 10.3390/v6114294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Ramirez JL, Souza-Neto J, Torres Cosme R, Rovira J, Ortiz A, Pascale JM, et al. Reciprocal tripartite interactions between the Aedes aegypti midgut microbiota, innate immune system and dengue virus influences vector competence. PLoS Negl Trop Dis. 2012;6:e1561. doi: 10.1371/journal.pntd.0001561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Ramirez JL, Short SM, Bahia AC, Saraiva RG, Dong Y, Kang S, et al. Chromobacterium Csp_P reduces malaria and dengue infection in vector mosquitoes and has entomopathogenic and in vitro anti-pathogen activities. PLoS Pathog. 2014;10:e1004398. doi: 10.1371/journal.ppat.1004398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Angleró-Rodríguez YI, Talyuli OA, Blumberg BJ, Kang S, Demby C, Shields A, et al. An Aedes aegypti-associated fungus increases susceptibility to dengue virus by modulating gut trypsin activity. eLife. 2017;6:e28844. 10.7554/eLife.28844. [DOI] [PMC free article] [PubMed]
- 199.Wu P, Sun P, Nie K, Zhu Y, Shi M, Xiao C, et al. A gut commensal bacterium promotes mosquito permissiveness to arboviruses. Cell Host Microbe. 2019;25:101–12. [DOI] [PubMed]
- 200.Hilgenboecker K, Hammerstein P, Schlattmann P, Telschow A, Werren JH. How many species are infected with Wolbachia?—A statistical analysis of current data. FEMS Microbiol Lett. 2008;281:215–220. doi: 10.1111/j.1574-6968.2008.01110.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Ross PA. Designing effective Wolbachia release programs for mosquito and arbovirus control. Acta Trop. 2021;222:106045. doi: 10.1016/j.actatropica.2021.106045. [DOI] [PubMed] [Google Scholar]
- 202.Caragata EP, Dutra HLC, Sucupira PHF, Ferreira AGA, Moreira LA. Wolbachia as translational science: controlling mosquito-borne pathogens. Trends Parasitol. 2021;37:1050–1067. doi: 10.1016/j.pt.2021.06.007. [DOI] [PubMed] [Google Scholar]
- 203.Crawford JE, Clarke DW, Criswell V, Desnoyer M, Cornel D, Deegan B, et al. Efficient production of male Wolbachia-infected Aedes aegypti mosquitoes enables large-scale suppression of wild populations. Nat Biotechnol. 2020;38:482–492. doi: 10.1038/s41587-020-0471-x. [DOI] [PubMed] [Google Scholar]
- 204.Mains JW, Kelly PH, Dobson KL, Petrie WD, Dobson SL. Localized Control of Aedes aegypti (Diptera: Culicidae) in Miami, FL, via inundative releases of Wolbachia-infected male mosquitoes. J Med Entomol. 2019;56:1296–1303. doi: 10.1093/jme/tjz051. [DOI] [PubMed] [Google Scholar]
- 205.The Project Wolbachia – Singapore C, Ching NL: Wolbachia-mediated sterility suppresses Aedes aegypti populations in the urban tropics. medRxiv; 2021.
- 206.Zheng X, Zhang D, Li Y, Yang C, Wu Y, Liang X, et al. Incompatible and sterile insect techniques combined eliminate mosquitoes. Nature. 2019;572:56–61. doi: 10.1038/s41586-019-1407-9. [DOI] [PubMed] [Google Scholar]
- 207.McMeniman CJ, Lane RV, Cass BN, Fong AW, Sidhu M, Wang YF, et al. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science. 2009;323:141–144. doi: 10.1126/science.1165326. [DOI] [PubMed] [Google Scholar]
- 208.O'Neill SL, Ryan PA, Turley AP, Wilson G, Retzki K, Iturbe-Ormaetxe I, et al. Scaled deployment of Wolbachia to protect the community from dengue and other Aedes transmitted arboviruses. Gates open research. 2018;2:36. doi: 10.12688/gatesopenres.12844.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Indriani C, Tantowijoyo W, Rancès E, Andari B, Prabowo E, Yusdi D, et al. Reduced dengue incidence following deployments of Wolbachia-infected Aedes aegypti in Yogyakarta, Indonesia: a quasi-experimental trial using controlled interrupted time series analysis. Gates open research. 2020;4:50. doi: 10.12688/gatesopenres.13122.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Pinto SB, Riback TIS, Sylvestre G, Costa G, Peixoto J, Dias FBS, et al. Effectiveness of Wolbachia-infected mosquito deployments in reducing the incidence of dengue and other Aedes-borne diseases in Niterói, Brazil: a quasi-experimental study. PLoS Negl Trop Dis. 2021;15:e0009556. doi: 10.1371/journal.pntd.0009556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Garcia GA, Sylvestre G, Aguiar R, da Costa GB, Martins AJ, Lima JBP, et al. Matching the genetics of released and local Aedes aegypti populations is critical to assure Wolbachia invasion. PLoS Negl Trop Dis. 2019;13:e0007023. doi: 10.1371/journal.pntd.0007023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Nazni WA, Hoffmann AA, NoorAfizah A, Cheong YL, Mancini MV, Golding N, et al. Establishment of Wolbachia strain wAlbB in Malaysian populations of Aedes aegypti for dengue control. Curr Biol. 2019;29:4241–8.e5. doi: 10.1016/j.cub.2019.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Iturbe-Ormaetxe I, Walker T, SL ON. Wolbachia and the biological control of mosquito-borne disease. EMBO Rep. 2011;12:508–18. [DOI] [PMC free article] [PubMed]
- 214.Moreira LA, Iturbe-Ormaetxe I, Jeffery JA, Lu G, Pyke AT, Hedges LM, et al. A Wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and Plasmodium. Cell. 2009;139:1268–1278. doi: 10.1016/j.cell.2009.11.042. [DOI] [PubMed] [Google Scholar]
- 215.Hurk AF, Hall-Mendelin S, Pyke AT, Frentiu FD, McElroy K, Day A, et al. Impact of Wolbachia on infection with chikungunya and yellow fever viruses in the mosquito vector Aedes aegypti. PLoS Negl Trop Dis. 2012;6:e1892. doi: 10.1371/journal.pntd.0001892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Kambris Z, Blagborough AM, Pinto SB, Blagrove MS, Godfray HC, Sinden RE, et al. Wolbachia stimulates immune gene expression and inhibits Plasmodium development in Anopheles gambiae. PLoS Pathog. 2010;6:e1001143. doi: 10.1371/journal.ppat.1001143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Bian G, Xu Y, Lu P, Xie Y, Xi Z. The endosymbiotic bacterium Wolbachia induces resistance to dengue virus in Aedes aegypti. PLoS Pathog. 2010;6:e1000833. doi: 10.1371/journal.ppat.1000833. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article.