Abstract
Background
Toenail onychomycosis affects approximately 6.7% of Canadians. Symptoms include nail discolouration/disfiguration and pain; psychosocial impacts contribute to reduced health-related quality-of-life. Comorbid diabetes increases the risk of complications and exacerbates burden. Treatment may include topical therapy and/or oral agents.
Purpose
To understand toenail onychomycosis treatment preferences, and to quantify the impact of toenail onychomycosis, with or without diabetes, on patient well-being.
Methods
Adults living in Canada with self-reported, physician-diagnosed, toenail onychomycosis were recruited online. A discrete choice experiment was used to quantify treatment preferences. Scenarios were randomized; data were analyzed using conditional logit regression. Health state utilities were estimated using the Health Utilities Index Mark 3®. Results were stratified by diabetes status and toenail onychomycosis severity; the Wilcoxon Rank Sum test was used to assess between-group utility differences.
Results
Three-hundred thirteen participants with toenail onychomycosis were included (161 had comorbid diabetes; 61.3%, severe onychomycosis). The mean age was 57.7 years; 55.9% were male. Treatment attributes with statistically significant impacts on patient preferences were efficacy (odds ratio [OR],1.04; 95% confidence interval [CI], 1.02–1.05 per 1% increased treatment success), administration method (one pill versus topical nail lacquer reference, 1.14; 1.04–1.26; topical solution applicator versus reference: 1.15; 1.03–1.29), severe adverse events (0.85; 0.80–0.90 per 1% increased risk), and risk of potential pharmacodynamic (0.80; 0.76–0.85) and alcohol (0.93; 0.88–0.98) interactions; preferences were more pronounced for efficacy and avoiding severe adverse events among toenail onychomycosis patients with comorbid diabetes. The mean (95% CI) utility value was 0.73 (0.70–0.75) overall, and statistically significantly lower (p=0.02) for toenail onychomycosis patients with diabetes (0.70; CI, 0.66–0.73) than those without (0.76; CI, 0.72–0.79).
Conclusion
Among patients with toenail onychomycosis, the presence of diabetes was associated with differing treatment-related preferences. Utility values for patients with toenail onychomycosis represent a significant decline from full health that is exacerbated by comorbid diabetes.
Keywords: health-related-quality-of-life, utilities, discrete choice experiment, fungus
Introduction
Toenail onychomycosis has an estimated prevalence of 6.7% in Canada.1 Prevalence increases with age, while comorbidities (eg, diabetes mellitus, human immunodeficiency virus) and lifestyle factors (eg, smoking, participation in some sports) increase susceptibility.2–8 Approximately 33% of people living with type I/II diabetes mellitus are afflicted with toenail onychomycosis.9 Furthermore, those with diabetes are at increased risk of developing complications secondary to onychomycosis including foot ulcer, cellulitis, and peripheral artery disease which may lead to lower limb amputation and death.3,10–15 Physical and social consequences of onychomycosis can include pain, paresthesia, difficulties performing activities of daily life, and impaired social interaction.4,5 Although not life threatening, high prevalence and significant clinical, social, and financial impact contribute to toenail onychomycosis burden.3
Adherence to toenail onychomycosis oral and topical treatments is known to be suboptimal.3 Treatment modalities are associated with prolonged treatment regimens and slow toenail growth, leading to poor management and frequent recurrence.2,3 Treatment selection is multifactorial; considerations include severity of nail involvement, mode of administration, efficacy, side effects, and costs.2,3 Selection between topical or oral systemic agents depends on disease severity, organism responsible, risk of adverse events, comorbidities, and treatment success history.3 While both topical and systemic agents can effectively treat toenail onychomycosis, oral antifungals are often recognized as more efficacious.16 However, systemic agents are associated with more severe adverse events than topicals, including headaches, gastrointestinal symptoms, rash, increased drug-drug interactions, and risk of hepatotoxicity.3,17,18 Health Canada-approved prescription products for onychomycosis include: ciclopirox (Penlac®; topical nail lacquer); efinaconazole (Jublia®; topical solution brush applicator); terbinafine (Lamisil® and generics; oral); and itraconazole (Sporanox® and generics; oral). Over-the-counter products are also available. Less common therapeutic interventions include fluconazole (off-label use), laser therapy, photodynamic therapy, and surgical nail removal.
Topical therapies may be a safer choice for patients for whom systemic therapies are not an option (eg, where there is a risk of drug-drug interactions). However, patient preferences for different treatment modalities, and how these preferences vary across key subpopulations with predisposing conditions for onychomycosis, are not well understood. This is of particular relevance for patients with diabetes given considerations around comorbidities, polypharmacy, and increased risk of onychomycosis.19,20
Utility values, a key input of economic evaluations comparing different treatments, quantify the physical and social impacts of a disease allowing overall disease impact to be measured. One common method for estimating utilities is the Health Utilities Index 15-item questionnaire (HUI-15Q®).21 Utility values range from less than 0 (worse than being dead, or “0”) to 1 (equivalent to full health).22 Health-state utility estimates are lacking for those with toenail onychomycosis. To date, utility values stratified by toenail onychomycosis severity or presence of comorbidities are lacking, and only one mean estimate of health state utility for onychomycosis is published.23 Given the small sample size (n=6) from which that estimate was obtained, and that the utility value estimate for persons living with onychomycosis was higher than that for the general Canadian population, estimation of a more robust utility value is warranted.
The objectives of this study were to (1) understand patient preferences regarding oral and topical toenail onychomycosis treatment features, and (2) estimate utility values associated with toenail onychomycosis, and how these varied according to diabetes status and toenail onychomycosis severity.
Materials and Methods
A cross-sectional survey evaluating treatment preferences using a discrete choice experiment (DCE), and utility estimation of adult patients with toenail onychomycosis living in Canada was conducted.
Qualitative Phase Methods
To help inform the DCE, a series of qualitative, semi-structured interviews were conducted among individuals with self-reported toenail onychomycosis. Participants were asked to describe the following: disease history and severity; comorbidities; impact on daily activities and health-related quality-of-life (HRQoL); treatment experience; and perceptions regarding treatment attributes. Participants were asked if each of a set of treatment attributes were important to them (efficacy, extra doctor visits, extra lab tests, frequency and duration, interactions with other medications, oral versus topical, side effects, and costs), then asked to rank the attributes. Differences in the perceived importance of attributes between those with, and without, diabetes and treatment history were explored. Ethics approval for the qualitative phase of this study was provided by Veritas institutional review board (IRB) on June 9, 2020 (#16575-15:38:275–06-2020). This research study involved human subjects; as such, the ethical principles outlined in the World Medical Association (WMA)’s Declaration of Helsinki were followed.24
Quantitative Phase Methods
Participants with self-reported, physician-diagnosed, toenail onychomycosis were recruited through Facebook or email advertisements and redirected to the survey landing page and screening questionnaire.
For study inclusion, participants were required to (1) live in Canada; (2) be aged 18 years or older; (3) have a self-reported diagnosis of onychomycosis of the toenail (active infection or cured within the three months before screening); (4) be able to read English fluently; and (5) be willing to provide written informed consent.
The survey was administered via an internet-based portal and comprised three distinct sections to assess: (1) patient and disease characteristics; (2) treatment preferences via a DCE; and (3) utility estimates using the HUI-15Q®.21
Sample size considerations were based on the primary objective (DCE) and calculated using standard methods.25 A minimum of n=63 per strata was required. Ethics approval for the quantitative phase was provided by Veritas IRB on July 27, 2020 (#16592-12:06:1924–07-2020), with an amendment to the protocol approved on February 15, 2022 (# 2022–2743-9764-1). The amendment outlined the use of different recruiting methods to obtain the required sample size. Ethical principles outlined in the WMA’s Declaration of Helsinki were followed.24
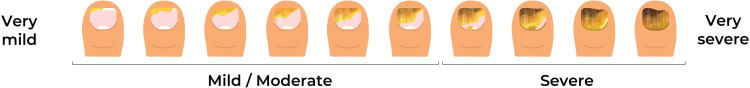
Toenail onychomycosis disease severity was assessed based on the extent of disease of the most affected toenail using a severity visual aid developed with clinical expert input (Figure 1). Clinical and demographic characteristics were summarized using patient counts and percentages, mean (standard deviation [SD]), or median (interquartile range [IQR]) where relevant.
Figure 1.
Toenail onychomycosis severity visual aid.
Six DCE treatment attributes (one with four, and five with two levels) were constructed: 1) mode of administration, frequency, and duration of treatment. These were amalgamated into one attribute and were constructed to represent treatments available in Canada: ciclopirox as topical nail lacquer once a day for 48 weeks with weekly home filing/trimming and monthly removal of the unattached/infected nail (by a health care professional; however, this was not specified in the DCE attribute description); efinaconazole as topical solution once a day for 48 weeks; terbinafine as one pill once a day for 12 weeks; and itraconazole as 2 pills twice a day for 1 week on and 3 weeks off over 12–16 weeks. Other attributes were 2) mild adverse effects; 3) severe adverse effects; 4) efficacy; 5) interaction with alcohol consumption (included based on clinical expert opinion); and 6) interaction with other medications (Figure 2).
Figure 2.
Discrete choice experiment: toenail onychomycosis treatment attributes and levels.
The DCE comprised 16 choice scenarios developed using the Hahn Shapiro catalogue.26 Both choice questions and attribute order within choices were randomized to minimize impact of respondent fatigue. The data obtained from the DCE were analyzed according to best practice guidelines.27 Data were modelled using conditional fixed-effects logit regression models.28 All DCE models were fit using the mclogit (version 0.9.6) package in R (version 4.2.1).29 Results were presented as odds ratios (ORs) with 95% confidence intervals (CIs) and p-values, stratified by diabetes status and toenail onychomycosis disease severity.
Utility values are anchored at 1 (equivalent to a state of full health) and 0 (equivalent to being dead), while states worse than dead are represented by negative values. Utility values were estimated from HUI-15Q® scores using the HUI Mark 3 (HUI3®) health status classification system and presented as means with 95% CIs.30 Utility values were presented for all participants and stratified by diabetes status and disease severity.
All continuous variables were evaluated for normality using the Shapiro–Wilk test. Between group differences were assessed using standard two sample t-tests for normally distributed variables, and the non-parametric Wilcoxon Rank Sum test for non-normally distributed data. Differences between categorical data were assessed using Chi-squared tests. P-values less than 0.05 were considered statistically significant.
Results
Qualitative Results
Fourteen interviews were conducted among individuals with self-reported toenail onychomycosis from across Canada, seven with comorbid diabetes and 11 with prior or current prescription treatment experience.
Participants described varying types of pain, emotions centered around feeling self-conscious about exposed toes, and frustration with treatments that did not appear to work. Participants noted that being able to go swimming, to the beach, get a pedicure, and wear sandals were aspects of daily life that were affected. Participants reported that their physicians appeared hesitant to prescribe oral onychomycosis therapies, regardless of underlying comorbidities.
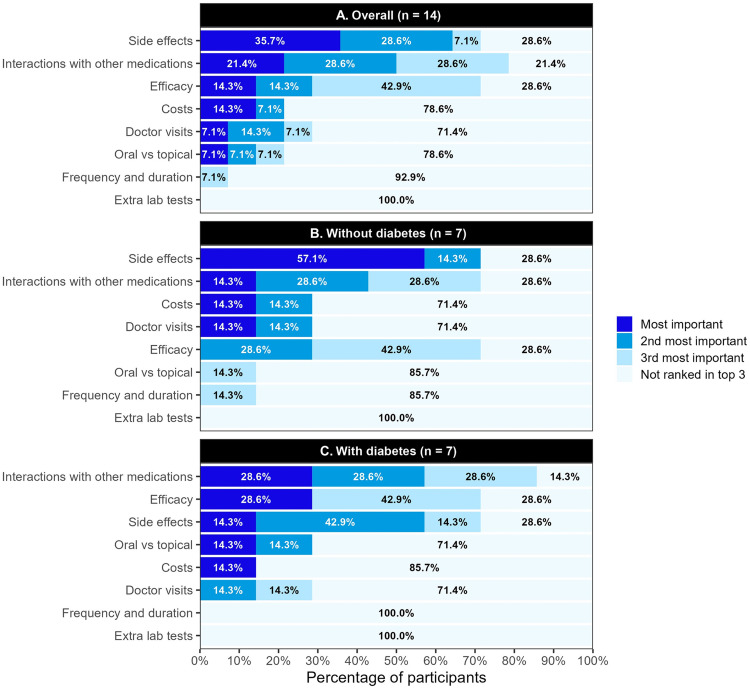
Side effects (35.7%) and interactions with other medications (21.4%) were most frequently identified as the most important attributes during the ranking exercise. After stratification by diabetes status, participants without comorbid diabetes chose potential side effects as the most important attribute 57.1% of the time. Those with diabetes chose interactions with other medications and efficacy as the most important attribute equally (28.6%; Figure 3).
Figure 3.
Attribute rankings among toenail onychomycosis (A) overall, (B) without diabetes, and (C) with diabetes.
Quantitative Results
Three hundred and thirteen adults with toenail onychomycosis were included, 161 (51.4%) with comorbid diabetes (type I or II), 192 (61.3%) with severe onychomycosis, and 113 (36.1%) with both comorbid diabetes and severe toenail onychomycosis. The mean (SD) age was 57.7 (14.7) years, and a slightly higher proportion of participants were male (55.9%; Table 1).
Table 1.
Baseline Patient Demographic and Clinical Characteristics, Overall and by Diabetes Status, Toenail Onychomycosis Severity, and Prescription Onychomycosis Treatment Experience
| Overall | Diabetes Status | Toenail Onychomycosis Severity | |||||
|---|---|---|---|---|---|---|---|
| No | Yes | Difference (p-value) | Mild/ Moderate | Severe | Difference (p-value) | ||
| n=313 | n=152 | n=161 | n=121 | n=192 | |||
| Demographic characteristics | |||||||
| Male sex, n (%) | 175 (55.9) | 78 (51.3) | 97 (60.2) | 0.14 | 67 (55.4) | 108 (56.2) | 0.97 |
| Age (years) | |||||||
| Mean, SD | 57.7 (14.7) | 56.1 (16.6) | 59.1 (12.5) | 0.07 | 56.4 (16.5) | 58.5 (13.4) | 0.23 |
| Median, IQR | 59.0 (50.0, 67.0) | 59.0 (44.8, 67.2) | 60.0 (52.0, 67.0) | 58.0 (46.0, 66.0) | 60.0 (51.0, 68.0) | ||
| Marital status, n (%) | |||||||
| Single | 76 (24.3) | 35 (23.0) | 41 (25.5) | 0.13 | 33 (27.3) | 43 (22.4) | 0.17 |
| Married/Common law | 185 (59.1) | 100 (65.8) | 85 (52.8) | 68 (56.2) | 117 (60.9) | ||
| Widowed/Divorced | 51 (16.3) | 17 (11.2) | 34 (21.1) | 20 (16.5) | 31 (16.1) | ||
| Gross household income ($ CAD) | |||||||
| $0 - $40,000 | 88 (28.1) | 31 (20.4) | 57 (35.4) | <0.01 | 28 (23.1) | 60 (31.2) | 0.20 |
| $40,000 - $60,000 | 62 (19.8) | 32 (21.1) | 30 (18.6) | 24 (19.8) | 38 (19.8) | ||
| $60,000 - $80,000 | 47 (15.0) | 22 (14.5) | 25 (15.5) | 15 (12.4) | 32 (16.7) | ||
| $80,000 + | 95 (30.4) | 60 (39.5) | 35 (21.7) | 43 (35.5) | 52 (27.1) | ||
| Prefer not to answer | 21 (6.7) | 7 (4.6) | 14 (8.7) | 11 (9.1) | 10 (5.2) | ||
| Clinical characteristics | |||||||
| Comorbid diabetes mellitus type I/II, n (%) | 161 (51.4) | 0 (0.0) | 161 (100.0) | <0.01 | 48 (39.7) | 113 (58.9) | <0.01 |
| Currently taking medication for diabetes | 152 (48.6) | 0 (0.0) | 152 (94.4) | 44 (36.4) | 108 (56.2) | ||
| Currently using toenail fungus prescription medication, n (%) | 113 (36.1) | 42 (27.6) | 71 (44.1) | <0.01 | 38 (31.4) | 75 (39.1) | 0.21 |
| Prescribed by family doctor | 97 (85.8) | 34 (81.0) | 63 (88.7) | 30 (78.9) | 67 (89.3) | ||
| Oral prescription | 26 (26.8) | 1 (2.9) | 25 (39.7) | 3 (10.0) | 23 (34.3) | ||
| Topical prescription | 86 (88.7) | 33 (97.1) | 53 (84.1) | 28 (93.3) | 58 (86.6) | ||
| Prescribed by dermatologist | 8 (7.1) | 3 (7.1) | 5 (7.0) | 4 (10.5) | 4 (5.3) | ||
| Oral prescription | 1 (12.5) | 0 (0.0) | 1 (20.0) | 0 (0.0) | 1 (25.0) | ||
| Topical prescription | 8 (100.0) | 3 (100.0) | 5 (100.0) | 4 (100.0) | 4 (100.0) | ||
| Prescribed by other | 8 (7.1) | 5 (11.9) | 3 (4.2) | 4 (10.5) | 4 (5.3) | ||
| Oral prescription | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||
| Topical prescription | 8 (100.0) | 5 (100.0) | 3 (100.0) | 4 (100.0) | 4 (100.0) | ||
| Used toenail fungus prescription medication in the past, n (%) | 76 (24.3) | 46 (30.3) | 30 (18.6) | 0.02 | 30 (24.8) | 46 (24.0) | 0.97 |
| Prior use of oral prescription | 33 (43.4) | 23 (50.0) | 10 (33.3) | 7 (23.3) | 26 (56.5) | ||
| Prior use of topical prescription | 56 (73.7) | 33 (71.7) | 23 (76.7) | 23 (76.7) | 33 (71.7) | ||
| Time since diagnosis (years), mean (SD) | 11.0 (12.3) | 12.5 (13.7) | 9.5 (10.6) | 0.03 | 11.9 (14.8) | 10.5 (10.4) | 0.36 |
| Number of toenails affected, mean (SD) | 3.6 (2.6) | 3.4 (2.4) | 3.8 (2.7) | 0.14 | 2.9 (2.2) | 4.1 (2.7) | <0.01 |
| At least one big toenail affected, n (%) | 277 (88.5) | 129 (84.9) | 148 (91.9) | 0.07 | 106 (87.6) | 171 (89.1) | 0.83 |
| Big toenail most affected, n (%) | 264 (84.3) | 118 (77.6) | 146 (90.7) | <0.01 | 100 (82.6) | 164 (85.4) | 0.62 |
| Severity based on most affected toenail, n (%) | |||||||
| Mild (0 to 20%) | 7 (2.2) | 5 (3.3) | 2 (1.2) | <0.01 | 7 (5.8) | 0 (0.0) | <0.01 |
| Moderate (21% to 60%) | 114 (36.4) | 68 (44.7) | 46 (28.6) | 114 (94.2) | 0 (0.0) | ||
| Severe (61%+) | 192 (61.3) | 79 (52.0) | 113 (70.2) | 0 (0.0) | 192 (100.0) | ||
Note: Statistically significant p-values (<0.05) are bold.
Abbreviations: CAD, Canadian dollar; IQR, interquartile range; SD, standard deviation.
The cohort with diabetes included a slightly higher proportion of males (p-value for difference, 0.14) and was slightly older (p=0.07). Among toenail onychomycosis severity and diabetes subgroups, demographic characteristics (eg, marital status and household income) were similar. More patients with diabetes were currently using prescription medication (either topical or oral) for their toenail fungus (44.1%) than those without (27.6%; p<0.01). Severe toenail fungus was more common among those with diabetes (70.2%) than those without (52.0%; p<0.01).
The DCE analysis set comprised 309 participants who completed all 16 choice scenarios. Treatment preferences overall and stratified by diabetes status and toenail onychomycosis severity are presented in Table 2. Several treatment attributes had statistically significant impacts on patient preferences.
Table 2.
Treatment Preference results from the DCE, Overall and Stratified by Diabetes Status and Toenail Onychomycosis Severity
| Overall | p-value | Diabetes Status | Toenail Onychomycosis Severity | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No | Yes | Mild/ Moderate | Severe | |||||||
| n=309 | n=149 | n=160 | n=111 | n=188 | ||||||
| OR (95% CI) | OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | OR (95% CI) | p-value | ||
| Efficacy | ||||||||||
| Efficacy* | 1.04 (1.02, 1.05) | <0.01 | 1.01 (0.99, 1.03) | 0.37 | 1.06 (1.04, 1.08) | <0.01 | 1.02 (0.99, 1.05) | 0.13 | 1.05 (1.02, 1.07) | <0.01 |
| Treatments | ||||||||||
| One pill** | 1.14 (1.04, 1.26) | 0.01 | 1.16 (1.01, 1.34) | 0.03 | 1.12 (0.98, 1.29) | 0.10 | 1.16 (0.99, 1.35) | 0.07 | 1.13 (1.00, 1.28) | 0.05 |
| Topical applicator** | 1.15 (1.03, 1.29) | 0.01 | 1.28 (1.09, 1.50) | <0.01 | 1.05 (0.90, 1.23) | 0.55 | 1.12 (0.94, 1.35) | 0.20 | 1.17 (1.02, 1.36) | 0.03 |
| Two pills** | 1.03 (0.94, 1.14) | 0.51 | 1.08 (0.94, 1.24) | 0.27 | 0.99 (0.86, 1.14) | 0.88 | 0.93 (0.80, 1.09) | 0.37 | 1.11 (0.98, 1.26) | 0.11 |
| Adverse events | ||||||||||
| Mild adverse events* | 1.00 (0.99, 1.00) | 0.13 | 1.00 (1.00, 1.00) | 0.80 | 1.00 (0.99, 1.00) | 0.06 | 1.00 (0.99, 1.00) | 0.36 | 1.00 (0.99, 1.00) | 0.22 |
| Severe adverse events* | 0.85 (0.80, 0.90) | <0.01 | 0.88 (0.81, 0.95) | <0.01 | 0.82 (0.76, 0.89) | <0.01 | 0.87 (0.79, 0.95) | <0.01 | 0.84 (0.78, 0.90) | <0.01 |
| Interactions | ||||||||||
| Medication interactions | 0.80 (0.76, 0.85) | <0.01 | 0.86 (0.79, 0.93) | <0.01 | 0.76 (0.70, 0.82) | <0.01 | 0.78 (0.72, 0.86) | <0.01 | 0.82 (0.76, 0.88) | <0.01 |
| Alcohol interactions | 0.93 (0.88, 0.98) | 0.01 | 1.04 (0.96, 1.13) | 0.36 | 0.83 (0.77, 0.90) | <0.01 | 0.92 (0.84, 1.01) | 0.08 | 0.93 (0.86, 1.00) | 0.04 |
Notes: *Per 1% increase; **Topical nail lacquer was selected as the reference treatment, ie, the treatment against which other treatments were compared; statistically significant p-values (<0.05) are bold.
Abbreviations: CI, confidence interval; OR, odds ratio.
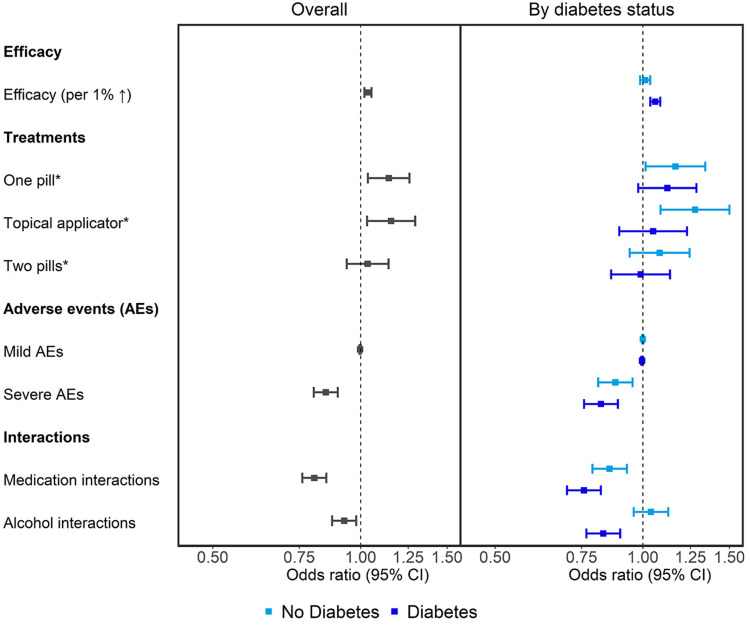
Overall, a 1% increase in efficacy increased the odds of the treatment being preferred by 4% (OR, 1.04; 95% CI, 1.02 to 1.05; p<0.01). Compared to using topical nail lacquer daily for 48 weeks that requires weekly nail filing and monthly nail filing/removal, taking one pill daily for 12 weeks increased the odds of an individual preferring a treatment by 14% (OR, 1.14; CI, 1.04 to 1.26; p=0.01), and using a topical solution (brush applicator) daily for 48 weeks by 15% (OR, 1.15; CI, 1.03 to 1.29; p=0.01). Participants were 15% less likely to prefer a treatment with a 1% risk of severe adverse events (liver, kidney, or heart damage resulting in hospitalization; OR, 0.85; CI, 0.80 to 0.90; p<0.01). The risk of interaction with other medications decreased the odds of treatment preference by 20% (OR, 0.80; CI, 0.76 to 0.85; p<0.01), and interaction with alcohol by 7% (OR, 0.93; CI; 0.88 to 0.98; 0.01).
Among those without diabetes, compared to using a topical nail lacquer daily for 48 weeks with its related nail filing/removal, participants were more likely to prefer taking one pill daily over 12 weeks (OR, 1.16; CI 1.01 to 1.34; p=0.03) or using a topical solution (brush applicator) daily for 48 weeks (OR, 1.28; CI, 1.09 to 1.50; p<0.01). Odds of treatment preference decreased by 12% with a 1% risk of severe adverse events relative to no risk (OR, 0.88; CI, 0.81 to 0.95; p<0.01), and by 14% if the treatment could interact with other medications (OR, 0.86; CI, 0.79 to 0.93; p<0.01).
Among participants with comorbid diabetes, a 1% increase in treatment efficacy resulted in 6% higher odds of treatment preference (OR, 1.06; CI, 1.04 to 1.08; p<0.01). The odds of preferring a treatment decreased by 18% with a 1% increase in risk of severe adverse events (OR, 0.82; CI, 0.76 to 0.89; p<0.01), by 24% with the risk of medical interactions (OR, 0.76; CI, 0.70 to 0.82; p<0.01), and 17% with the risk of interactions with alcohol (OR, 0.83; CI, 0.77 to 0.90; p<0.01; Table 2; Figure 4).
Figure 4.
Toenail onychomycosis treatment preferences (odds ratios with 95% Cis) overall any by diabetes status. *Topical nail lacquer was selected as the reference treatment, i.e, the treatment against which other treatments were compared.
Abbreviations: AE, adverse event; CI, confidence interval.
Among participants with mild/moderate toenail onychomycosis, the odds of preferring a treatment decreased by 13% with a 1% increase in risk of severe adverse events (OR, 0.87; CI, 0.79 to 0.95; p<0.01), and by 22% with the risk of medical interactions (OR, 0.78; CI, 0.72 to 0.86; p<0.01).
Participants with severe toenail onychomycosis were 5% more likely to prefer a treatment that conveyed 1% higher efficacy (OR, 1.05; CI, 1.02 to 1.07; p<0.01). Compared to a topical nail lacquer, participants preferred to use a topical solution (OR, 1.17; CI, 1.02 to 1.36; p=0.03). The odds of treatment preference decreased by 16% with a 1% risk of severe adverse events (OR, 0.84; CI, 0.78 to 0.90; p<0.01), by 18% if the treatment could interact with other medications (OR, 0.82; CI, 0.76 to 0.88; p<0.01), and 7% if the treatment could interact with alcohol consumption (OR, 0.93; CI, 0.86 to 1.00; p=0.04; Table 2).
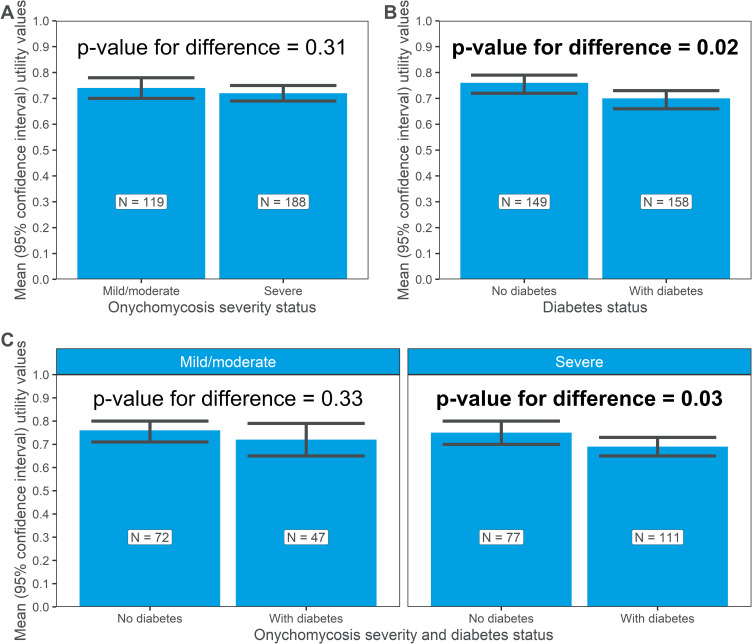
Three hundred and seven participants with toenail onychomycosis completed all 15 items of the HUI3® survey and were thus included in the utility estimation. The mean utility estimate for the sample was 0.73 (95% CI: 0.70 to 0.75; Table 3).
Table 3.
Utility Estimates, Overall and Stratified by Diabetes Status and Toenail Onychomycosis Severity
| Group | n | Mean Utility (95% CI) | p-value for Difference |
|---|---|---|---|
| Overall | 307 | 0.73 (0.70, 0.75) | - |
| Mild/moderate toenail onychomycosis | 119 | 0.74 (0.70, 0.78) | 0.31 |
| Severe toenail onychomycosis | 188 | 0.72 (0.69, 0.75) | |
| No diabetes | 149 | 0.76 (0.72, 0.79) | 0.02 |
| With diabetes | 158 | 0.70 (0.66, 0.73) | |
| Mild/moderate toenail onychomycosis; no diabetes | 72 | 0.76 (0.71, 0.80) | 0.33 |
| Mild/moderate toenail onychomycosis; with diabetes | 47 | 0.72 (0.65, 0.79) | |
| Severe toenail onychomycosis; no diabetes | 77 | 0.75 (0.70, 0.80) | 0.03 |
| Severe toenail onychomycosis; with diabetes | 111 | 0.69 (0.65, 0.73) |
Note: Statistically significant differences (p<0.05) are bold.
Abbreviation: CI, confidence interval.
After stratification by toenail onychomycosis severity, utility estimates were higher among those with mild/moderate toenail onychomycosis (n=119; mean, 0.74; CI, 0.70 to 0.78) compared to those with severe disease (n=188; mean, 0.72; CI, 0.69 to 0.75); however, the difference between the groups was not statistically significant (p=0.31; Table 3; Figure 5A).
Figure 5.
Utilities estimates (mean, 95% CI), stratified by (A) toenail onychomycosis disease severity, (B) the presence or absence of diabetes, and (C) both toenail onychomycosis disease severity and the presence or absence of diabetes.
Note: statistically significant differences (p<0.05) are bold.
When stratified according to diabetes status (Table 3; Figure 5B), utility estimates were statistically significantly lower (p=0.02) among those with comorbid diabetes (n=158; mean, 0.70; CI, 0.66 to 0.73) than those without (n=149; mean, 0.76; CI, 0.72 to 0.79).
After stratification by both toenail onychomycosis severity and the presence or absence of diabetes (Table 3; Figure 5C), among those with mild/moderate toenail onychomycosis, utility estimates among those with (n=47; mean, 0.72; CI, 0.65 to 0.79) and without diabetes (n=72; mean, 0.76; CI, 0.71 to 0.80) were similar (p=0.33). Among those with severe toenail onychomycosis (Table 3; Figure 5C), the difference between those with diabetes (n=111; mean, 0.69; CI, 0.65 to 0.73) and those without diabetes (n=77; mean, 0.75; CI, 0.70 to 0.80) was statistically significant (p=0.03), again with lower estimates among those with comorbid diabetes than without.
Discussion
Toenail onychomycosis is highly prevalent in the general population, and notably more so among those with type I or II diabetes mellitus.1,9 The presence of comorbid diabetes complicates treatment-making decisions and is associated with an increased risk of potentially significant complications secondary to onychomycosis.9,13,31 A key finding of this study lies in the observation that preferences for treatment attributes differ substantially among toenail onychomycosis patients with and without diabetes. Furthermore, a quantifiable and important reduction in estimated utility values among those with toenail onychomycosis in relation to the general Canadian adult population mean utility of 0.86 was observed;32 this decrease was further exacerbated in the presence of comorbid diabetes mellitus.
The modality chosen to treat toenail onychomycosis in some sub-populations requires particular consideration. Oral medications must be used with caution in patients on concomitant medications, and those who are at heightened risk for adverse events.19,20,33 This study revealed that patient preferences for treatment attributes vary by underlying diabetes status, with those with diabetes having stronger preferences for treatment attributes related to efficacy, risk of severe adverse events, and potential for multi-drug or alcohol interactions, than those without. These factors could be considered during reimbursement decisions regarding toenail onychomycosis therapy coverage in Canada. Currently, several topical and oral medications are approved for use in Canada; however, only oral agents are publicly reimbursed for the management of toenail onychomycosis. For patients who are not suitable candidates for systemic therapy, no alternate topical treatments are covered by public drug plans. However, the findings from this study highlight an unmet need among patients with comorbid diabetes, who were more likely to prefer treatment attributes associated with topical therapies (no risk of adverse events, and no medication or alcohol interactions).
Utility value estimates for Canadians with toenail onychomycosis were lower than Canadian population normal values, and statistically significantly lower among those with comorbid diabetes mellitus type I/II. While the estimate for those with severe disease was slightly lower than those with mild/moderate toenail onychomycosis, the difference was not statistically significant.
Our findings align with several other studies reporting reduced HRQoL in onychomycosis patients, with contributing factors including difficulty cutting nails, limited footwear options, embarrassment, physical discomfort, and the potential of spread.34–39 However, to the best of our knowledge, only one study has reported a utility estimate for this population. This utility value of 0.99, estimated from a sample of six onychomycosis patients using the HUI3®,21,23 is higher than Canadian adult population normal mean of 0.86.32 Given the documented reduction in HRQoL among those with onychomycosis, and the larger sample size of 307, the value estimated from this study of 0.73 may be more robust for use in cost-utility analyses evaluating current and future therapies. It should be noted that the sample in the present study purposely oversampled those with diabetes to ensure robust estimates from this subgroup, therefore population-specific weighting should be applied depending on the target population under evaluation.
This is also the first study to report utility estimates stratified by the presence or absence of comorbid diabetes. Diabetes itself is associated with a reduced HRQoL, with Statistics Canada reporting HUI3® mean utility values for Canadians with diabetes of 0.77.40 The utility values presented here for toenail onychomycosis patients without (0.76) and with (0.70) comorbid diabetes may be useful for assessing therapies for use in these sub-populations.
Strengths of this study include a large sample size, onychomycosis- and diabetes comorbidity-specific estimates, and the use of an appropriate instrument validated for use in estimating health state utilities in Canadians (HUI3®). Limitations to this study include self, rather than physician-confirmed toenail onychomycosis and diabetes diagnosis and the recruitment of a convenience sample; however, effort was made to recruit patients from across Canada. Lastly, as out-of-pocket costs vary from person to person in Canada based on coverage, the impact of cost was not explored.
Conclusion
Among patients with toenail onychomycosis from Canada, presence of diabetes was associated with differences in treatment-related preferences that could be useful for decision-makers when assessing treatment options. In addition, the utility estimates presented highlight the reduced utility estimates among those with toenail onychomycosis, with and without diabetes mellitus type I/II, and can be used to inform cost-utility analyses of new toenail onychomycosis therapies for Canadians, and that may be applicable to other jurisdictions.
Acknowledgments
The DCE and utilities findings were independently published as abstracts and subsequently presented as posters at ISPOR Europe 2022.41,42
Funding Statement
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ATM, GLO, and EP are employees of Broadstreet HEOR, which received funds from Bausch Health, Canada Inc. for this work. The design of this study was developed in consultation with Dr. Steve Mathieu (Dermatologist), who received an honorarium from Bausch Health, Canada Inc. for his expert input. However, Dr. Steve Mathieu did not receive any compensation for the preparation and review of this manuscript.
Disclosure
VG and MB are employees of Bausch Health, Canada Inc., the manufacturer of Penlac® and Jublia®. Bausch Health, Canada, Inc. was not involved in data collection, analysis, or interpretation of the data. The authors report no other conflicts of interest in this work.
References
- 1.Gupta AK, Gupta G, Jain HC, et al. The prevalence of unsuspected onychomycosis and its causative organisms in a multicentre Canadian sample of 30 000 patients visiting physicians’ offices. J Eur Acad Dermatol Venereol. 2016;30(9):1567–1572. doi: 10.1111/jdv.13677 [DOI] [PubMed] [Google Scholar]
- 2.Gupta AK, Versteeg SG, Shear NH. Onychomycosis in the 21st century: an update on diagnosis, epidemiology, and treatment. J Cutan Med Surg. 2017;21(6):525–539. doi: 10.1177/1203475417716362 [DOI] [PubMed] [Google Scholar]
- 3.Christenson JK, Peterson GM, Naunton M, et al. Challenges and opportunities in the management of onychomycosis. J Fungi. 2018;4(3):87. doi: 10.3390/jof4030087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Elewski BE, Tosti A. Risk factors and comorbidities for onychomycosis: implications for treatment with topical therapy. J Clin Aesthet Dermatol. 2015;8(11):38–42. [PMC free article] [PubMed] [Google Scholar]
- 5.Lipner SR, Scher RK. Onychomycosis: clinical overview and diagnosis. J Am Acad Dermatol. 2019;80(4):835–851. [DOI] [PubMed] [Google Scholar]
- 6.Gupta AK, Stec N, Summerbell RC, et al. Onychomycosis: a review. J Eur Acad Dermatol Venereol. 2020;34(9):1972–1990. [DOI] [PubMed] [Google Scholar]
- 7.Rosen T, Friedlander SF, Kircik L, et al. Onychomycosis: epidemiology, diagnosis, and treatment in a changing landscape. J Drugs Dermatol. 2015;14(3):223–233. [PubMed] [Google Scholar]
- 8.Baran R. The nail in the elderly. Clin Dermatol. 2011;29(1):54–60. doi: 10.1016/j.clindermatol.2010.07.008 [DOI] [PubMed] [Google Scholar]
- 9.Winston JA, Miller JL. Treatment of onychomycosis in diabetic patients. Clin Diabetes. 2006;24(4):160–166. doi: 10.2337/diaclin.24.4.160 [DOI] [Google Scholar]
- 10.Akkus G, Evran M, Gungor D, Karakas M, Sert M, Tetiker T. Tinea pedis and onychomycosis frequency in diabetes mellitus patients and diabetic foot ulcers. A cross sectional - observational study. Pak J Med Sci. 2016;32(4):891–895. doi: 10.12669/pjms.324.10027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rossaneis MA, Haddad MD, Mantovani MF, Marcon SS, Pissinati PS. Foot ulceration in patients with diabetes: a risk analysis. Br J Nurs. 2017;26(6):S6–S14. doi: 10.12968/bjon.2017.26.6.S6 [DOI] [PubMed] [Google Scholar]
- 12.Papini M, Cicoletti M, Fabrizi V, Landucci P. Skin and nail mycoses in patients with diabetic foot. Giornale Ital Di Dermatol E Venereol. 2013;148(6):603–608. [PubMed] [Google Scholar]
- 13.Foley K, Gupta AK, Versteeg S, Mays R, Villanueva E, John D. Topical and device-based treatments for fungal infections of the toenails. Cochrane Database Syst Rev. 2020;1(1):Cd012093. doi: 10.1002/14651858.CD012093.pub2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fukunaga A, Washio K, Ogura K, et al. Onychomycosis as a warning sign for peripheral arterial disease. Acta Dermato Venereol. 2013;93(6):747–748. doi: 10.2340/00015555-1576 [DOI] [PubMed] [Google Scholar]
- 15.Ozkan F, Ozturk P, Ozyurt K, et al. Frequency of peripheral arterial disease and venous insufficiency in toenail onychomycosis. J Dermatol. 2013;40(2):107–110. doi: 10.1111/1346-8138.12020 [DOI] [PubMed] [Google Scholar]
- 16.Gupta AK, Foley KA, Mays RR, Shear NH, Piguet V. Monotherapy for toenail onychomycosis: a systematic review and network meta-analysis. Br J Dermatol. 2020;182(2):287–299. doi: 10.1111/bjd.18155 [DOI] [PubMed] [Google Scholar]
- 17.Yan J, Wang X, Chen S. Systematic review of severe acute liver injury caused by terbinafine. Int J Clin Pharm. 2014;36(4):679–683. doi: 10.1007/s11096-014-9969-y [DOI] [PubMed] [Google Scholar]
- 18.Aggarwal R, Targhotra M, Kumar B, Sahoo PK, Chauhan MK. Treatment and management strategies of onychomycosis. Journal de mycologie medicale. 2020;30(2):100949. doi: 10.1016/j.mycmed.2020.100949 [DOI] [PubMed] [Google Scholar]
- 19.Mayser P, Freund V, Budihardja D. Toenail onychomycosis in diabetic patients: issues and management. Am J Clin Dermatol. 2009;10(4):211–220. doi: 10.2165/00128071-200910040-00001 [DOI] [PubMed] [Google Scholar]
- 20.Ameen M, Lear JT, Madan V, Mohd Mustapa MF, Richardson M. British Association of Dermatologists’ guidelines for the management of onychomycosis 2014. Br J Dermatol. 2014;171(5):937–958. doi: 10.1111/bjd.13358 [DOI] [PubMed] [Google Scholar]
- 21.Torrance GW, Furlong W, Feeny D, Boyle M. Multi-attribute preference functions. Health Utilities Index. Pharmacoeconomics. 1995;7(6):503–520. doi: 10.2165/00019053-199507060-00005 [DOI] [PubMed] [Google Scholar]
- 22.Tolley KP. What are Health Utilities? Hayward Medical Communications; 2009. [Google Scholar]
- 23.Chen SC, Bayoumi AM, Soon SL, et al. A catalog of dermatology utilities: a measure of the burden of skin diseases. J Investig Dermatol Symp Proc. 2004;9(2):160–168. doi: 10.1046/j.1087-0024.2003.09112.x [DOI] [PubMed] [Google Scholar]
- 24.World Medical Association. WMA Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects; 2022. Available from: https://www.wma.net/policies-post/wma-declaration-of-helsinki-ethical-principles-for-medical-research-involving-human-subjects/. Accessed February 14, 2024.
- 25.Orme B. Sample Size Issues for Conjoint Analysis Studies. Sequim: Sawtooth Software Technical Paper; 1998. [Google Scholar]
- 26.Hahn GHS. A catalog and computer program for the design and analysis of orthogonal symmetric and asymmetric fractional factorial experiments; 1966.
- 27.Reed Johnson F, Lancsar E, Marshall D, et al. Constructing experimental designs for discrete-choice experiments: report of the ISPOR conjoint analysis experimental design good research practices task force. Value Health. 2013;16(1):3–13. doi: 10.1016/j.jval.2012.08.2223 [DOI] [PubMed] [Google Scholar]
- 28.Hauber AB, Gonzalez JM, Groothuis-Oudshoorn CG, et al. Statistical methods for the analysis of discrete choice experiments: a report of the ISPOR conjoint analysis good research practices task force. Value Health. 2016;19(4):300–315. doi: 10.1016/j.jval.2016.04.004 [DOI] [PubMed] [Google Scholar]
- 29.R Core Team. A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing; 2022. Available from: https://www.R-project.org/. Accessed February 14, 2024. [Google Scholar]
- 30.Horsman J, Furlong W, Feeny D, Torrance G. The Health Utilities Index (HUI): concepts, measurement properties and applications. Health Qual Life Outcomes. 2003;1(1):54. doi: 10.1186/1477-7525-1-54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cathcart S, Cantrell W, Elewski B. Onychomycosis and diabetes. J Eur Acad Dermatol Venereol. 2009;23(10):1119–1122. doi: 10.1111/j.1468-3083.2009.03225.x [DOI] [PubMed] [Google Scholar]
- 32.Guertin JR, Feeny D, Tarride JE. Age- and sex-specific Canadian utility norms, based on the 2013–2014 Canadian community health survey. CMAJ. 2018;190(6):E155–E161. doi: 10.1503/cmaj.170317 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gupta AK, Ryder JE, Skinner AR. Treatment of onychomycosis: pros and cons of antifungal agents. J Cutan Med Surg. 2004;8(1):25–30. doi: 10.1177/120347540400800107 [DOI] [PubMed] [Google Scholar]
- 34.Milobratović D, Janković S, Vukičević J, Marinković J, Janković J, Railić Z. Quality of life in patients with toenail onychomycosis. Mycoses. 2013;56(5):543–551. doi: 10.1111/myc.12072 [DOI] [PubMed] [Google Scholar]
- 35.Elewski BE. The effect of toenail onychomycosis on patient quality of life. Int J Dermatol. 1997;36(10):754–756. doi: 10.1046/j.1365-4362.1997.00163.x [DOI] [PubMed] [Google Scholar]
- 36.Szepietowski JC, Reich A, Pacan P, Garlowska E, Baran E. Evaluation of quality of life in patients with toenail onychomycosis by Polish version of an international onychomycosis-specific questionnaire. J Eur Acad Dermatol Venereol. 2007;21(4):491–496. doi: 10.1111/j.1468-3083.2006.02004.x [DOI] [PubMed] [Google Scholar]
- 37.Lubeck DP, Patrick DL, McNulty P, Fifer SK, Birnbaum J. Quality of life of persons with onychomycosis. Qual Life Res. 1993;2(5):341–348. doi: 10.1007/BF00449429 [DOI] [PubMed] [Google Scholar]
- 38.Lubeck DP. Measuring health-related quality of life in onychomycosis. J Am Acad Dermatol. 1998;38(5 Pt 3):S64–68. doi: 10.1016/S0190-9622(98)70487-6 [DOI] [PubMed] [Google Scholar]
- 39.Whittam LR, Hay RJ. The impact of onychomycosis on quality of life. Clin Exp Dermatol. 1997;22(2):87–89. doi: 10.1111/j.1365-2230.1997.tb02626.x [DOI] [PubMed] [Google Scholar]
- 40.Statistics Canada. Utility score norms for each of the chronic conditions examined in the 2013 and 2014 Canadian community health survey, household population aged 12 and older, Canada, 2013 and 2014; 2018. Available from: https://www150.statcan.gc.ca/n1/pub/82-003-x/2018011/article/00001/tbl/tbl03-eng.htm. Accessed February 14, 2024.
- 41.Lozano-Ortega G, Mickle A, Gaudet V, Popoff E, Barbeau M. PCR146 preferences for toenail onychomycosis treatments among Canadians with and without diabetes mellitus: a discrete choice experiment. Value Health. 2022;25(12):S419. doi: 10.1016/j.jval.2022.09.2081 [DOI] [Google Scholar]
- 42.Mickle A, Lozano-Ortega G, Gaudet V, Popoff E, Barbeau M. PCR28 utility values derived from Canadian adults with toenail onychomycosis with and without comorbid diabetes mellitus: a study using the health utilities index®. Value Health. 2022;25(12):S395. doi: 10.1016/j.jval.2022.09.1963 [DOI] [Google Scholar]