Abstract
There has been an increase in the number of women using marijuana whilst pregnant. Previous studies have shown that children with prenatal marijuana exposure have developmental deficits in memory and decreased attentiveness. In this study, we assess whether prenatal marijuana exposure is associated with alterations in brain regional morphometry and functional and structural connectivity in adolescents. We downloaded behavioural scores and subject image files from the Adolescent Brain Cognitive DevelopmentSM Study. A total of 178 anatomical and diffusion magnetic resonance imaging files (88 prenatal marijuana exposure and 90 age- and gender-matched controls) and 152 resting-state functional magnetic resonance imaging files (76 prenatal marijuana exposure and 76 controls) were obtained. Behavioural metrics based on the parent-reported child behavioural checklist were also obtained for each subject. The associations of prenatal marijuana exposure with 17 subscales of the child behavioural checklist were calculated. We assessed differences in brain morphometry based on voxel-based and surface-based morphometry in adolescents with prenatal marijuana exposure versus controls. We also evaluated group differences in structural and functional connectivity in adolescents for region-to-region connectivity and graph theoretical metrics. Interactions of prenatal marijuana exposure and graph networks were assessed for impact on behavioural scores. Multiple comparison correction was performed as appropriate. Adolescents with prenatal marijuana exposure had greater abnormal or borderline child behavioural checklist scores in 9 out of 17 subscales. There were no significant differences in voxel- or surface-based morphometry, structural connectivity or functional connectivity between prenatal marijuana exposure and controls. However, there were significant differences in prenatal marijuana exposure–graph network interactions with respect to behavioural scores. There were three structural prenatal marijuana exposure–graph network interactions and seven functional prenatal marijuana exposure–graph network interactions that were significantly associated with behavioural scores. Whilst this study was not able to confirm anatomical or functional differences between prenatal marijuana exposure and unexposed pre-adolescent children, there were prenatal marijuana exposure–brain structural and functional graph network interactions that were significantly associated with behavioural scores. This suggests that altered brain networks may underlie behavioural outcomes in adolescents with prenatal marijuana exposure. More work needs to be conducted to better understand the prognostic value of brain structural and functional network measures in prenatal marijuana exposure.
Keywords: ABCD, prenatal marijuana exposure, functional connectivity, structural connectivity, graph networks
Vishnubhotla et al. demonstrate that prenatal marijuana exposure is associated with worse behavioural scores in adolescents. Prenatal marijuana exposure also impacts the association between several structural and functional graph network measures and behavioural scores, suggesting that altered brain networks could underlie adverse behavioural outcomes in adolescents with prenatal marijuana exposure.
Graphical Abstract
Graphical Abstract.
Introduction
Several studies have shown an increasing prevalence of marijuana use following legalization trends, and the United Nations Office on Drugs and Crime reported marijuana as the most commonly used illicit drug globally in 2016.1 Along with increasing rates of marijuana use, there has been an increase in the number of women using marijuana whilst pregnant. Alleviation of nausea, vomiting and morning sickness have been cited as common reasons for marijuana use during pregnancy. Additionally, there is an increasing perception of safety with prenatal marijuana exposure (PME), as many pregnant women report being unaware of any adverse effects associated with PME.2-4 Marijuana is the most common recreational drug used in pregnancy.5 A cross-sectional study found that out of 367,403 pregnancies between 2009 and 2017 in the Kaiser Permanente Northern California population, self-reported daily cannabis use during pregnancy was 3.38% in 2017.3 In a follow-up study, cannabis-only use during pregnancy rose to 6.3% in 2018.6 The 2020 National Survey of Drug Use and Health reported marijuana use in the past month among pregnant women aged 15–44 as 8%.7 The National Institute on Drug Abuse estimates that rates may be higher since women were twice as likely to screen positive for marijuana use in a urine test than self-report.8 Therefore, these are likely underestimates because data from most studies is self-reported.
Despite the increasing prevalence of PME, there is a relative lack of knowledge on the long-term effects of PME. Previous studies have shown that PME leads to developmental deficits—such as gaps in problem-solving skills and memory, increased depressive and anxiety symptoms, and decreased ability to remain attentive in school-aged children with PME compared with controls.9-12 The Ottawa Prenatal Prospective Study13,14 showed developmental challenges in children with PME that persisted from birth until 12 years of age. These developmental challenges included decreased verbal memory, poorer sustained attention, increased risk for hyperactivity and impulsivity and poorer executive functioning. Other researchers have shown that these cognitive and behavioural challenges continued onto adulthood as those with PME showed decreased response inhibition, and significantly greater brain activity compared with unexposed controls when performing executive functioning tasks.15
Effects of PME on the brain have been assessed through neuroimaging studies. Functional magnetic resonance imaging (fMRI) studies reveal impaired executive functioning,15 response inhibition16 and visuospatial working memory17 in children with PME compared with controls. Additionally, prenatal drug exposure, including marijuana, has shown to impact brain functional network connectivity.18-21 Assessing brain network connectivity could help predict those at risk for developmental conditions. For example, children with attention-deficit hyperactivity disorder (ADHD) are shown to have altered brain network connectivity,22-24 and risk of behavioural problems in children with ADHD children are associated with altered functional connectivity.25 Assessing structural connectivity is also important as white matter microstructural alterations have been associated with disruptive behaviours.26
Brain structure and function can be assessed through direct connections between regions of interest (ROIs) and interactions between regions or through organized frameworks called graph networks. Graph networks are relationships or connections between multiple brain regions that are mathematically assessed utilizing systems composed of nodes (specific brain region) and edges (connections between nodes). Several metrics are used to understand how inter-connected these nodes are, that may be global, comprising the entire network, or local, in a particular region of the network. These graph network metrics provide information on global and local network connectivity that cannot be assessed by simple ROI-to-ROI connectivity, which assesses each connection independently.
Graph theory metrics have been applied in neuroimaging to understand networks in the human brain.27-31 For example, individuals with ADHD had lower global and local efficiencies (measures of how well a network exchanges information), along with a longer characteristic path length (indicating lower efficiency).32 In the adult population, individuals with ADHD had lower local efficiencies and altered clustering coefficients (a measure of the extent regions cluster together) in multiple regions.33 Additionally, reduced values in another graph network measure, betweenness centrality (a measure of the amount of influence a region has in the flow of information), in the hippocampus and prefrontal cortex were associated with greater stress.34
Since brain structural and functional connectivity could be an important measure of developmental and behavioural problems and PME can be associated with adverse behavioural and developmental outcomes, we assess (i) the impact of PME on developmental and behavioural problems, and (ii) the impact of PME on the associations of altered brain morphology, structural connectivity and functional connectivity with behavioural outcomes in adolescents.
Methods
Gathering data
Data were acquired from the Adolescent Brain Cognitive DevelopmentSM (ABCD®) Study (https://abcdstudy.org), held in the NIMH Data Archive.35,36 Subjects were screened for PME, excluding exposure to substances such as alcohol, tobacco and other illicit drugs. Controls were screened and excluded for exposure to marijuana, alcohol, tobacco and other illicit drugs. Additionally, controls were selected to have a similar gender composition to the PME subjects. Demographic characteristics, such as age, sex, mean age, mean birth weight, maternal prescription medications, premature birth and maternal education, were compared between the PME and control groups. The parent-reported child behavioural checklist (CBCL)37 scores were also downloaded for each subject.
Minimally processed resting-state fMRI (rs-fMRI) images, diffusion-weighted images and T1-weighted anatomical images were also downloaded. Image samples were processed as described by Hagler et al.38 Briefly, fMRI data had B0 correction, unwarping and motion correction. Diffusion images had B0 correction, unwarping, motion correction and eddy current correction. Anatomical images had unwarping, intensity inhomogeneity correction and rigid body registration.
Behavioural metrics
Behavioural metrics were based on a parent-reported CBCL37 and used 17 subscales. These include anxiety/depression, withdrawn/depression, somatic complaints, social problems, thought problems, attention problems, rule-breaking behaviour, aggressive behaviour, internalizing problems, externalizing problems, ADHD, oppositional disorder, conduct disorder, sluggish cognition, obsessive-compulsive disorder, stress problems and total problems. Internalizing problems included scales for anxiety, withdrawal, depression and somatic complaints whilst externalizing problems included scales for rule-breaking and aggressive behaviours.39 There were specific scales for the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition criteria including ADHD, oppositional and conduct disorders.40 The scales used were normative, where 50 was considered average and clinical cut-offs of 65–69 for borderline and 70+ for clinically abnormal values. The impact of demographic characteristics such as age, sex, presence of PME, maternal education and partner education was assessed. Differences in CBCL clinical categories between PME and control groups were also assessed.
Voxel-based and surface-based morphometry
Voxel-based morphometry and surface-based morphometry were calculated from T1-weighted anatomical images using Computational Anatomy Toolbox 12.6 (CAT12) (Jena, Germany),41 a toolbox for Statistical Parametric Mapping, version 12 (SPM12).42 Steps for voxel-based morphometry processing include denoising,43 affine pre-processing, local adaptive segmentation, skull stripping and spatial normalization.44 Voxel-based morphometry grey matter segmentations were then smoothed at 8-mm full width at half maximum. Additional steps for surface-based morphometry processing utilize the projection-based thickness method45 along with topology correction and spherical mapping.46 Surface data were resampled based on a 32-K vertices surface mesh47 and smoothed to 15-mm full width at half maximum. For statistical analysis, two contrasts were set—PME > control and control > PME. Clusters less than 10 voxels were excluded. Total intracranial volume was used as a covariate for voxel-based morphometry.
Tract-based spatial statistics
Diffusion-weighted images, b-value, and b-vector files were downloaded from the ABCD® Study database. Tract-based spatial statistics48 were performed using the FMRIB (for fMRI of the brain) software library (FSL, Oxford, UK).49 First, fractional anisotropy (FA) images were created by fitting a tensor model using FMRIB’s diffusion toolbox and then brain extracted using FSL’s brain extraction tool.50 Subjects’ data were aligned into a common space using FMRIB’s non-linear image registration tool51,52 using a b-spline representation of the registration warp field.53 A mean FA skeleton was generated representing the centre of group tracts. Each subject’s FA data was projected onto the skeleton and voxelwise cross-subject statistics were generated. Data for mean diffusivity and axial diffusivity were obtained and data for radial diffusivity were calculated by taking the mean of the second and third eigenvalues. Mean diffusivity, axial diffusivity and radial diffusivity data were projected onto mean FA skeleton and cross-subject statistics were generated similar to FA. Comparisons were made between PME and unexposed controls using a general linear model and voxelwise statistics were performed using threshold-free cluster enhancement in randomize.54
Tractography
Diffusion-weighted images, b-value, and b-vector files were downloaded from the ABCD® Study database. The b-tables were imported and corrected using DSI Studio (http://dsi-studio.labsolver.org) using a population average template.55 Diffusion data were reconstructed in the Montreal Neurological Institute space using q-space diffeomorphic reconstruction56 and aligned with the International Consortium for Brain Mapping template.57,58 Tractography was performed on the whole brain with DSI Studio using a deterministic fibre tracking algorithm59 with a diffusion sampling ratio of 1.25. Three million tracts were calculated for each subject. The quantitative anisotropy threshold was set to software optimization. The angular threshold was set at 45° and the step size was 0.75 mm. Track lengths shorter than 20 mm or longer than 200 mm were discarded. The International Consortium for Brain Mapping template57,58 was registered to subject space through non-linear transformation. Brain parcellation regions were based on the automated anatomical labelling version 2 atlas.60 Connectivity matrices and graph network measures were calculated in DSI Studio based on fibre count.
Structural pairwise connectivity
Connectivity matrices with 120 ROIs were collected for each subject based on fibre count. Regions involving the cerebellum and vermis were excluded, leaving 94 ROIs for the analysis. The count data were firstly normalized via square root transformation. Robust linear regression was employed to assess the significance of PME on connectivity, where predictors include age, sex, PME, maternal education and partner education.
Structural graph network measures
Network measures for fibre count were collected for each subject within DSI Studio based on the Brain Connectivity Toolbox.61 Prior studies have shown altered graph networks in developmental conditions.32-34 Therefore, we evaluated network measures including average path length, global efficiency, local efficiency, betweenness centrality and clustering coefficient. Networks were based on weighted values. The significance of PME on network measures was assessed using a linear regression. Age, sex, PME, maternal education and partner education were predictors and network measures were the response variables.
rs-fMRI pre-processing
Pre-processing and analysis of rs-fMRI data were performed with CONN Toolbox (Cambridge, MA).62,63 Samples obtained from the ABCD database were already minimally processed with B0 correction, unwarping and motion correction as described by Hagler et al.38 that we used for further analysis. Functional and structural MRI data were normalized to the standard Montreal Neurological Institute T1 template using a direct normalization process. Data were segmented into grey matter, white matter and cerebrospinal fluid.64,65 Isotropic resolution of 1 mm for structural images and 2 mm for functional images were used. Next, the data were smoothed using spatial convolution with a Gaussian kernel of 8-mm full width at half maximum.63
Denoising involved removing of noise from white matter and cerebrospinal fluid,66,67 scrubbing68 and session effects. For temporal band pass filtering, the lower frequency threshold was 0.008 Hz, and the upper frequency threshold was 0.09 Hz. Filtering was performed after regression to avoid mismatch in nuisance regressor procedure.63,69
Independent component analysis
Independent component analysis was conducted using CONN Toolbox62 based on the methodology as described by Calhoun et al.70 Comparisons were made between PME and unexposed controls. Clusters less than five voxels in size were discarded.
rs-fMRI analysis
Functional connectivity was assessed between ROIs using CONN Toolbox.62 ROI-to-ROI analysis was performed based on 106 regions of the Harvard–Oxford atlas,71-74 distributed by Conn Toolbox. Cerebellar regions were excluded from the analysis. Statistical analysis was also performed in CONN Toolbox. Comparisons were made between PME and unexposed controls using a weighted general linear model.75 Analyses were conducted on Fisher z-transformed correlations.
Functional graph network measures
Functional graph network measures were obtained using CONN Toolbox based on the Brain Connectivity Toolbox.61 Prior studies have shown altered graph networks in developmental conditions.32-34 Therefore, we evaluated network measures including average path length, global efficiency, local efficiency, betweenness centrality and clustering coefficient. The significance of PME on network measures was assessed using a robust linear regression. Age, sex, PME, maternal education and partner education were the predictors and network measures were the response variables.
Correlating behavioural data with graph network measures
Clinically relevant CBCL metrics were correlated with graph network measures and PME to assess their impact on clinical scores. The significance of network measures and PME on clinical scores was assessed using linear regression for structural and functional data. Age, sex, PME, maternal education, partner education and network measure were the predictors and CBCL scores were the response variables. An interaction between PME and network measures was included in the analysis so that significance was assessed for each group separately, as well as for the interaction term.
Statistical analysis
Statistical analyses were performed using R Studio (posit.co/products/open-source/rstudio/), MATLAB® (mathworks.com/products/matlab.html; MathWorks; Natick, MA, USA), and within individual processing packages.
The impact of demographic characteristics such as age, sex, presence of PME, maternal education and partner education on behavioural measures was assessed using a general linear model. Differences in clinical categories were calculated using a Fisher’s exact test. Multiplicity was corrected following the Benjamini–Hochberg procedure to control for the false discovery rate.76 An adjusted P-value of <0.05 was considered significant.
Statistics for voxel-based and surface-based morphometry were calculated in SPM, statistics for TBBS were calculated in FSL, and statistics for fMRI independent component analysis were calculated in CONN Toolbox. Structural ROI-to-ROI connectivity was calculated using the sum of square errors followed by a robust linear regression. Functional ROI-to-ROI analysis was conducted in CONN Toolbox. Structural and functional graph network measures were calculated in DSI Studio and CONN Toolbox, respectively. Statistical comparisons of graph network measures were performed using linear regressions. Multiplicity was corrected following the Benjamini–Hochberg procedure to control for the false discovery rate.76 In all cases, significance was determined by an adjusted P-value of <0.05.
Results
Demographics
There were 178 subjects included in this study—88 (35 males) prenatally exposed to marijuana and 90 (37 males) without prenatal marijuana exposure. Both groups did not have history of prenatal exposure to other substances, such as alcohol, tobacco and other illicit drugs. Maternal education level was significantly different for PME versus unexposed controls (P = 0.0013). Factors such as birth weight, maternal prescription medications and premature birth were not statistically different. The demographic data are summarized in Table 1.
Table 1.
Demographic information of children with PME and unexposed controls
| PME | Controls | P-value | |
|---|---|---|---|
| Number | 88 | 90 | |
| Males | 35 | 37 | 0.977b |
| Mean age (years) (SD) | 9.9 (0.59) | 9.9 (0.65) | 0.879a |
| Mean birth weight (lbs.) | 6.9 | 7.1 | 0.405a |
| Taking prescription medications | 9 | 11 | 0.813b |
| Premature birth | 15 | 13 | 0.684b |
| Maternal college degree | 18 | 39 | 0.001b,* |
| Number (fMRI cohort) | 76 | 76 | |
| Males (fMRI cohort) | 30 | 27 | 0.738b |
| Mean age of fMRI cohort (years) (SD) | 9.9 (0.58) | 9.9 (0.68) | 0.694a |
aCalculated by an unpaired t-test.
bCalculated using a Fisher’s exact test.
*Significant.
Behavioural metrics
Behavioural metrics were based on the parent-reported CBCL37 from which we used data from 17 subscales. These scales had clinical cut-offs of 65–69 for borderline, and 70+ for clinical cases. Mean values were significantly greater in those with PME for all scales except the anxiety/depression and somatic complaint scales. Based on a general linear model, PME had a significant effect on 16 out of the 17 scales after correcting for multiple comparisons. No other variables were significant after multiple comparison corrections (Table 2). Clinical outcomes were significantly different in those with PME compared with unexposed controls in 9 of 17 scales. These include thought problems, attention problems, rule-breaking behaviour, aggressive behaviour, externalizing problems, ADHD, conduct disorder, sluggish cognition and total problems. The data are summarized in Table 3. Full data for this table are shown in Supplementary Table 1.
Table 2.
A linear regression was performed with age, sex, maternal education, partner education and PME as predictors and behavioural scores as response variables
| t-stat | P-value | P-FDR | |
|---|---|---|---|
| Anxiety/depression | 2.04 | 0.043 | 0.046* |
| Withdrawn/depressed | 2.53 | 0.012 | 0.014* |
| Somatic complaints | 1.23 | 0.219 | 0.219 |
| Social problems | 3.69 | <0.001 | <0.001* |
| Thought problems | 4.08 | <0.001 | <0.001* |
| Attention problems | 4.43 | <0.001 | <0.001* |
| Rule breaking behaviour | 3.74 | <0.001 | <0.001* |
| Aggressive behaviour | 3.88 | <0.001 | <0.001* |
| Internalizing problems | 3.10 | 0.002 | 0.003* |
| Externalizing problems | 4.67 | <0.001 | <0.001* |
| ADHD | 4.67 | <0.001 | <0.001* |
| Oppositional disorder | 3.55 | <0.001 | <0.001* |
| Conduct disorder | 3.64 | <0.001 | <0.001* |
| Sluggish cognitive | 4.28 | <0.001 | <0.001* |
| Obsessive-compulsive disorder | 2.79 | 0.006 | 0.007* |
| Stress problems | 3.45 | <0.001 | <0.001* |
| Total problems | 5.47 | <0.001 | <0.001* |
All predictors except for PME were not significant after multiple comparison corrections. Behavioural measures for which PME was a significant predictor are shown in this table. A false discovery rate less than 0.05 was considered significant.
*Significant.
Table 3.
CBCL clinical classifications based on sub-scale scores for PME and unexposed children
| P-value | P-FDR | |
|---|---|---|
| Anxiety/depression | 0.316 | 0.335 |
| Withdrawn/depressed | 0.185 | 0.225 |
| Somatic complaints | 0.230 | 0.261 |
| Social problems | 0.109 | 0.16 |
| Thought problems | 0.002 | 0.02a |
| Attention problems | 0.006 | 0.025a |
| Rule breaking behaviour | 0.021 | 0.042a |
| Aggressive behaviour | 0.021 | 0.042a |
| Internalizing problems | 0.113 | 0.16 |
| Externalizing problems | 0.007 | 0.025a |
| ADHD | 0.002 | 0.02a |
| Oppositional disorder | 0.034 | 0.058 |
| Conduct disorder | 0.022 | 0.042a |
| Sluggish cognitive | 0.014 | 0.04a |
| Obsessive-compulsive disorder | 0.180 | 0.225 |
| Stress problems | 0.371 | 0.371 |
| Total problems | 0.007 | 0.025a |
Comparisons were calculated using a Fisher’s exact test and multiple comparison correction was based on false discovery rate. A false discovery rate less than 0.05 was considered significant.
aSignificant.
Voxel-based and surface-based morphometry
Brain anatomical measurements for volume and surface thickness were assessed using voxel-based and surface-based morphometry, respectively, for 88 PME and 90 unexposed children. There were no clusters or regions showing significant differences. The results are displayed in Supplementary Figs 1 and 2.
Voxelwise tract-based spatial statistics
Tract-based spatial statistics were performed for diffusion-weighted images of 88 PME children and 90 unexposed children. There were no regions or tracts showing significant differences when accounting for multiple comparisons.
Structural pairwise connectivity
Differences in structural connectivity were measured in terms of fibre count in 88 PME children and 90 unexposed children; 73 connections were significantly different in PME versus unexposed children including 51 with greater connectivity and 22 with lower connectivity in PME children. However, these did not maintain significance after correcting for multiple comparisons. The data are visualized in Supplementary Fig. 3.
Structural graph network measures
Differences in structural graph network measures were measured in 88 PME children and 90 unexposed children. None of the global graph network metrics showed significant differences. There were regional differences in graph network measures in local efficiency for three regions, betweenness centrality for six regions and clustering coefficient for six regions. However, the significance for these metrics was not sustained when corrected for multiple comparisons. The data are summarized in Supplementary Table 2.
Connectivity in independent component analysis functional networks
Brain rs-fMRI images were available for 152 subjects—76 PME and 76 unexposed children. Voxelwise clusters were compared between PME and unexposed controls. Clusters under five voxels in size were discarded. There were no regions showing significant differences between the PME and control groups.
Functional pairwise connectivity
Brain rs-fMRI images were obtained for 152 subjects—76 PME and 76 unexposed children. Brain functional connectivity was assessed between 106 ROIs. Whilst 511 connections showed significance for individual comparisons, significance was not maintained after correcting for multiple comparisons for all connections. This is shown in Supplementary Fig. 4.
Functional graph network measures
Differences in structural graph network measures were measured in 76 PME children and 76 unexposed children. None of the global graph network metrics showed significant differences. There were regional differences in graph network measures in local efficiency for two regions, betweenness centrality for three regions and clustering coefficient for three regions. However, the significance for these metrics was not sustained when corrected for multiple comparisons. The data are summarized in Supplementary Table 3.
Prediction of behavioural measures based on graph networks and PME
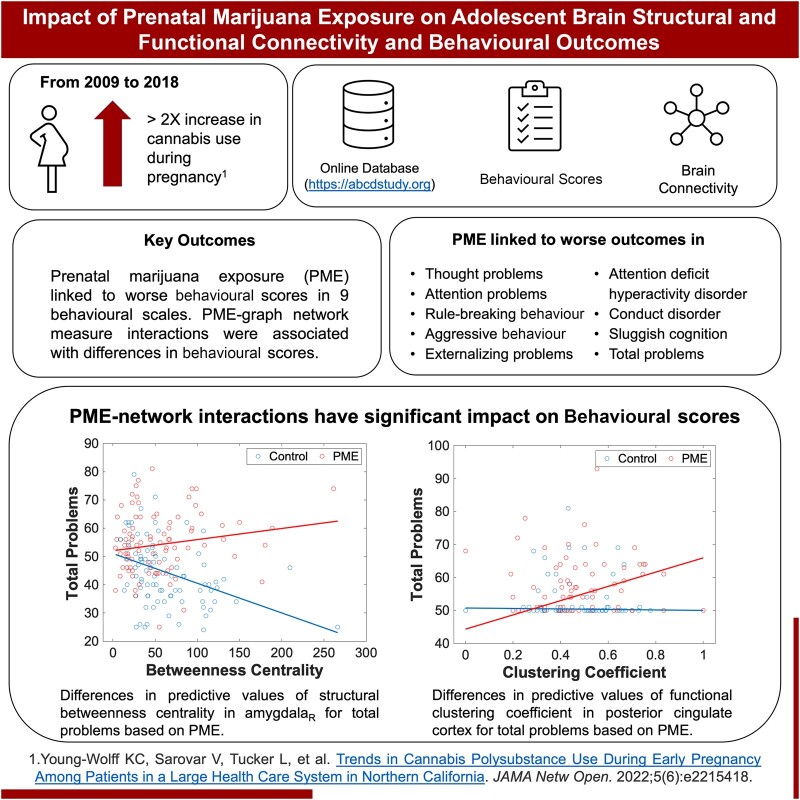
We assessed the predictive value of graph network measures on behavioural scores using linear models. For this, we assess the significance of network–PME interactions for nine behavioural scores that demonstrated significant differences between PME and unexposed controls. In our predictive interaction plot, differences in slopes between PME and controls represent different effects of graph network measures on behavioural scores. For structural networks, there was a significant difference in PME–network interactions in local efficiency (Fig. 1A) and clustering coefficient in the right lateral orbitofrontal cortex (OFC) (Fig. 1B) for the externalizing problems scale. Additionally, there was a significant difference in PME–network interactions in betweenness centrality in the right amygdala for the total problems scale (Fig. 1C). Full data can be found in Supplementary Tables 4–12.
Figure 1.
PME interactions for structural graph networks. Plots of PME–graph network interactions for structural connectivity using a linear model (n = 178). (A) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for externalizing problems scores versus local efficiency in the right lateral OFC grouped by exposure. There were significant differences in interactions between PME and control groups (t = −3.69). (B) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for externalizing problems scores versus clustering coefficient in the right lateral OFC grouped by exposure. There were significant differences in interactions between PME and control groups (t = −3.68). (C) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for total problems scores versus betweenness centrality in the right amygdala grouped by exposure. There were significant differences in interactions between PME and control groups (t = 3.89). A false discovery rate less than 0.05 was considered significant.
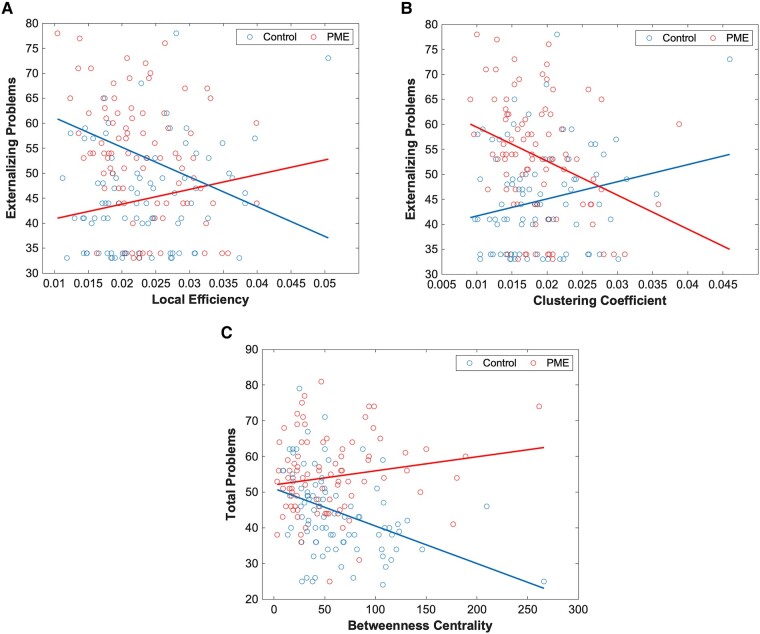
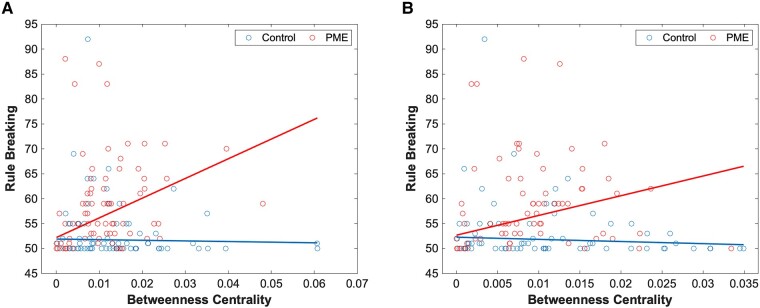
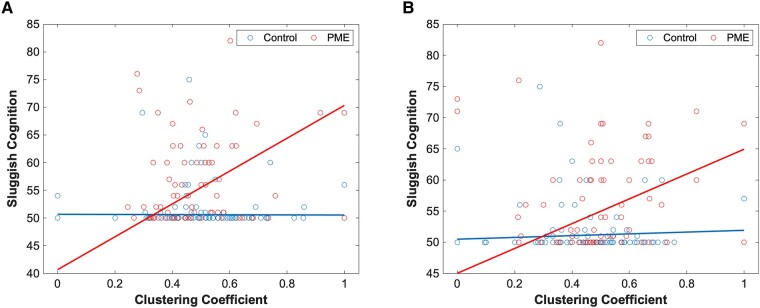
A similar assessment was performed for functional graph networks. Here, there was a significant difference in PME–network interactions in betweenness centrality in the right inferior temporal gyrus (Fig. 2A) and the left cuneus (Fig. 2B) for the rule-breaking behaviour scale. There were also several significant PME–network interactions that were significant predictors for sluggish cognition. Networks where this was observed include betweenness centrality in the left calcarine sulcus (Fig. 3), clustering coefficient in the left middle gyrus (Fig. 4A) and clustering coefficient in the left inferior temporal gyrus (Fig. 4B). Finally, PME–network interactions for the clustering coefficient in the posterior cingulate cortex were a significant predictor for total problems (Fig. 5). Full data can be found in Supplementary Tables 13–21.
Figure 2.
PME interactions for rule-breaking scores. Plots of PME–graph network interactions for functional connectivity using a robust linear model (n = 152). (A) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for rule-breaking scores versus betweenness centrality in the right fusiform gyrus grouped by exposure. There were significant differences in interactions between PME and control groups (t = 5.02). (B) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for rule-breaking scores versus betweenness centrality in the left cuneus grouped by exposure. There were significant differences in interactions between PME and control groups (t = 5.89). A false discovery rate less than 0.05 was considered significant.
Figure 3.
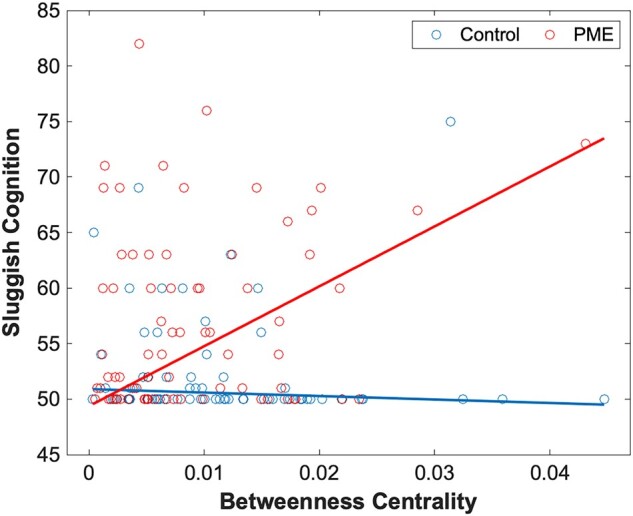
Sluggish cognition and betweenness centrality. Plot of PME–graph network interactions for functional connectivity using a robust linear model (n = 152). Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for sluggish cognition scores versus betweenness centrality in the left calcarine sulcus grouped by exposure. There were significant differences in interactions between PME and control groups (t = 6.68). A false discovery rate less than 0.05 was considered significant.
Figure 4.
Sluggish cognition and clustering coefficient. Plots of PME–graph network interactions for functional connectivity using a robust linear model (n = 152). (A) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for sluggish cognition scores versus clustering coefficient in the left anterior middle temporal gyrus grouped by exposure. There were significant differences in interactions between PME and control groups (t = 6.29). (B) Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for sluggish cognition scores versus clustering coefficient in the left anterior inferior temporal gyrus grouped by exposure. There were significant differences in interactions between PME and control groups (t = 3.69). A false discovery rate less than 0.05 was considered significant.
Figure 5.
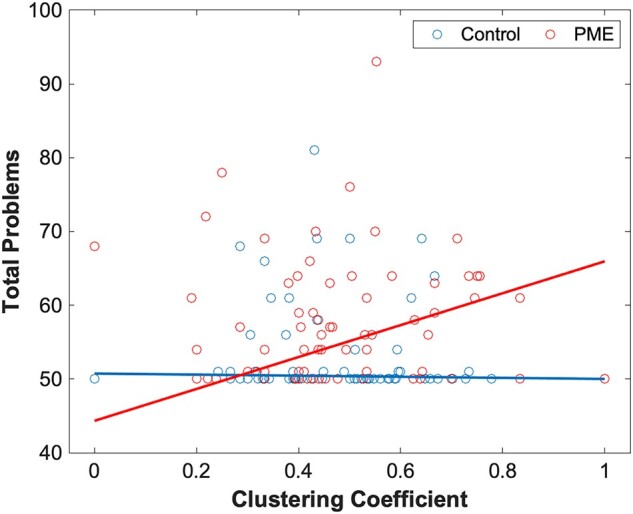
PME interactions for total problems scores. Plot of PME–graph network interactions for functional connectivity using a robust linear model (n = 152). Chart represents a scatter plot with raw data overlayed with an interaction plot based on adjusted data for total problems scores versus clustering coefficient in the posterior cingulate cortex grouped by exposure. There were significant differences in interactions between PME and control groups (t = 4.71). A false discovery rate less than 0.05 was considered significant.
Discussion
This study assesses the impact of PME on behaviour and neurodevelopment in subjects from the Adolescent Brain Cognitive Development database. Overall, PME was a significant contributor to worse behavioural scores in 16 out of 17 CBCL scales, with the somatic complaint scale being the only exception. In this analysis, none of the other factors (age, sex, maternal education and partner education) were significant predictors for behavioural outcomes. Based on clinical thresholds, 9 out of 17 behavioural scales were significantly influenced by PME, after correcting for multiple comparisons. Effected scales were related to attention and behavioural problems. PME was less strongly associated with internalizing factors such as anxiety and depression. Our results are concordant with prior findings showing PME being associated with a higher risk of developing behavioural problems and ADHD.11,77
Despite significant differences in behavioural scores between adolescents with PME and non-PME controls, differences in neuroimaging measurements were not statistically significant. There were no significant differences in brain structure on both voxel-based and surface-based morphometry after multiple comparison corrections. Similarly, there were no significant differences in FA, mean diffusivity, axial diffusivity or radial diffusivity. However, other studies have shown that children with PME demonstrated greater thickness in the frontal cortices78 and alterations in brain volume. These different results may be related to different ages at evaluation, socioenvironmental confounders or image analysis methods.79
Results for differences in structural and functional connectivity were mixed. We assessed connectivity using both ROI-to-ROI and graph network connectivity. ROI-to-ROI connectivity involves direct connections, either anatomical or functional, from one region to another. On the other hand, graph networks are organizational frameworks involving nodes and edges that are centred around a particular brain region or the whole brain. From our results, there were no significant differences of ROI-to-ROI or graph network connectivity based on the presence of PME. However, several PME–graph network interactions were significant predictors for multiple behavioural scales.
Graph network metrics that were involved with significant differences between PME and control groups include clustering coefficient, local efficiency and betweenness centrality. All significant metrics were significant at the nodal level (anatomical ROI). Clustering coefficient is a measure of how nodes tend to cluster together. At the level of a node, or brain region, this is the proportion of connections that are being utilized by its neighbouring regions compared with the total number of possible connections.80 Local efficiency is involved with how well information is exchanged between its neighbouring regions in the absence of the node.81 Increased levels of both indicate greater connectivity. Betweenness centrality is a metric that indicates the amount of influence a region has for the overall network. This is assessed by the number of shortest paths that travel through the brain region.82
There were several structural graph networks that had significantly different correlations with behavioural scores based on the presence of PME. Correlations between graph network metrics (clustering coefficient and local efficiency) of the right lateral OFC with externalizing problems in the PME versus control groups. This indicates that local connectivity of the right lateral OFC and neighbouring regions may have different association to externalizing problems in those with PME. The OFC is an important structure for decision-making and the lateral OFC has been linked to conforming to social values,83 suppression of responses,84 evaluation of punishment85 and devaluation.86 Another node that showed differences in correlation of structural metrics with behavioural scores was betweenness centrality of the right amygdala for the total problems scale. This suggests that the influence of the right amygdala may have a different association with total problems scores in those with PME compared with controls. The amygdala has been associated with emotions such as fear87-89 and memory89,90 and is a component of the rewards network.91,92 The amygdala is also functionally connected to the OFC,93 and this connection is shown to be responsible for with social anxiety,94,95 immediate rewards,96 goal-directed behaviour84,97 and reinforcement.98 Additionally, betweenness centrality in the amygdala has been associated with increased stress.34
Our results suggest that PME differentially impacts the association of several functional graph networks and behavioural scores. First, we identified differences in correlations of betweenness centrality in the right fusiform gyrus and left cuneus with the rule-breaking scale in those with PME compared with controls. The fusiform gyrus is an important region for facial recognition, and connectivity in this region has been linked to learning disabilities such as dyslexia.99 The cuneus is involved with visual processing and atypical connectivity in this region has been linked with autism spectrum disorders.100 Increased significance of these regions in those specifically with PME is noteworthy.
Second, we identified differences in associations of functional graph network metrics (betweenness centrality in the left calcarine sulcus and clustering coefficient in the left anterior middle and inferior temporal gyri) with the sluggish cognition scale. Increased influence of the left calcarine sulcus is associated with poorer outcomes of sluggish cognition in those with PME compared with controls. Reduced connectivity in the neighbouring regions may imply that these systems are more poorly regulated. The calcarine sulcus is a prominent component in the visual cortex and functional connectivity in the region has been linked with eye misalignment.101 The calcarine sulcus may play a more significant role in cognition in those with PME.
Increased regional connectivities in the left anterior middle and inferior temporal gyri were associated with poorer outcomes of sluggish cognition in those with PME compared with controls. The middle and inferior temporal gyri are involved with semantic processing and visual perception. The association between increased regional connectivity and increased symptoms, even if the overall correlation is weak, is somewhat unexpected. Greater connectivity would typically be associated with improved functioning. However, it is possible that this greater regional connectivity could be a form of overcompensation for functional deficits.
Finally, we identified differences associations of the functional graph network clustering coefficient in the posterior cingulate cortex with the total problems scale in PME versus controls. The posterior cingulate cortex is a highly connected region that is an important region of the default mode network. Considering that the default mode network is associated with mind wandering,102 increased clustering coefficient may be associated with sluggish outcomes. However, this effect was notable in the PME population and not the controls. Altered connectivity in the default mode network has also been linked to many disorders including ADHD,103 depression,104 schizophrenia105 and other mental disorders.
In those with PME, there was a significant association of multiple graph measures in regions involving visual processing with behavioural outcomes. This is unsurprising given that PME has been linked with impaired visual processing in infants and toddlers.106 This may contribute to impaired learning as children with PME can increase the risk of learning disabilities and ADHD.9-12,107
Other studies have demonstrated important findings on the short- and long-term impact of PME. The Ottawa Prenatal Prospective Study found that PME impacted neurocognition at various stages of life.13 Whilst no correlation between PME and overall IQ was found, the study did find that children with PME tend to score more poorly on tasks related to executive functioning. Neonates with PME demonstrated decreased visual habituation and increased tremors. As children, the PME group had poorer verbal skills, memory, attention, visual perception and executive functioning. Similar issues seemed to continue into adulthood as they exhibited reduced concentration and inhibition along with impaired visual memory analytical skills.9,13 From this study, 31 subjects underwent fMRI using four tasks15 with the results demonstrating that the PME group had increased brain activity in left posterior brain regions for similar tasks compared with unexposed controls.
Other studies have shown differences in structural and functional connectivity in infants with PME.21,108 The lack of persistence of these structural and functional alterations may be at least partly explained by developmental neuroplasticity and postnatal factors. However, we identified differences in associations of multiple structural and functional graph network measures for behavioural scores based on the presence of PME. This would suggest that despite neuroplasticity, there may be some persistent focal developmental impacts of prenatal marijuana on the brain.
There have been other studies examining the long-term effects of PME based on subjects with the ABCD database. Based on a larger sample size from the ABCD® Study, differences in behavioural outcomes were observed in subjects with PME.109 Additionally, in the same study, significantly lower brain volumes were reported after multiple comparison corrections. Positive confirmatory findings for brain volume, which were not found in our study, may be due to a larger sample size and greater statistical power. Another study by Cioffredi et al.110 looked at PME in the context of three fMRI tasks and CBCL scores from the ABCD® Study. Similar to our study, they identified adolescents with PME having greater attention, externalizing and total problems scores. Additionally, they did not observe differences in terms of cognitive performance or patterns of brain activation during tasks. This falls in line with our results in that there were significant differences in behavioural scores, particularly externalizing scores, but there were no confirmatory findings regarding differences in MRI measurements.
Overall, this study showed a significant influence of PME on behavioural scores for 16 out of 17 scales and clinical categories for 9 out of 17 scales. Furthermore, several PME–graph network interactions were identified as significant predictors for multiple behavioural scales. This indicates that network measures in these regions could be biomarkers for those with PME who may be at risk for developmental and behavioural problems. On the other hand, we were not able to find a significant difference in anatomical measurements or brain connectivity between the PME and control groups after correcting for multiple comparisons. However, other studies have shown significant differences in these factors. Additionally, considering that there were several interactions terms that were significant for multiple behavioural scales, it is likely that PME has some effect on connectivity even though these could not pass the statistical threshold in our study due to limitations due to retrospective nature and probable undetermined environmental effects in the ABCD data.
There are several limitations that need to be noted. For the ABCD database, the proportion of adolescents with PME was relatively small, and being a retrospective assessment, there may be recall bias. For future studies, we would recommend using a prospective design and follow-up of a larger cohort tracking children from the neonatal stage to adolescence to better assess the causality of PME on developmental conditions such as behavioural problems or ADHD and more accurately assess brain developmental alterations. Whilst we assessed the differences between PME and unexposed children, we did not account for the amount of cannabis used. Although we had 178 subjects with behavioural scores, only 152 subjects had fMRI data. We used different atlases for different imaging modalities. For structural connectivity, we used the automated anatomical labelling version 2 atlas, and for functional connectivity, we used the Harvard–Oxford atlas. Since we did not correlate data from the different imaging modalities, there is limited effect for these differences. Finally, this study also did not take into account postnatal effects, as these have also shown to impact behavioural outcomes for children with prenatal substance exposure.111
Conclusions
This study, based on images and data from the Adolescent Brain Cognitive Development study, assessed the impact of PME on neurodevelopment in pre-adolescent children. There were significant differences in multiple CBCL scales in PME children compared with unexposed controls. There were no statistically significant differences in anatomical measurements and brain connectivity between the PME and control groups. However, there were PME–graph network interactions that were significant for behavioural scores. This suggests that altered brain networks may underlie behavioural outcomes in adolescents with PME. More work needs to be conducted to better understand the prognostic value of brain structural and functional network measures in PME.
Supplementary Material
Acknowledgements
The authors would like to acknowledge the contributions of Shrey Ramnath in image processing. Data used in the preparation of this article were obtained from the ABCD® Study (https://abcdstudy.org), held in the NIMH Data Archive. This is a multisite, longitudinal study designed to recruit more than 10 000 children aged 9–10 and follow them over 10 years into early adulthood. The ABCD® Study is supported by the National Institutes of Health and additional federal partners under award numbers U01DA041048, U01DA050989, U01DA051016, U01DA041022, U01DA051018, U01DA051037, U01DA050987, U01DA041174, U01DA041106, U01DA041117, U01DA041028, U01DA041134, U01DA050988, U01DA051039, U01DA041156, U01DA041025, U01DA041120, U01DA051038, U01DA041148, U01DA041093, U01DA041089, U24DA041123, and U24DA041147. A full list of supporters is available online (https://abcdstudy.org/federal-partners.html). A listing of participating sites and a complete listing of the study investigators can be found online (https://abcdstudy.org/consortium_members/). ABCD consortium investigators designed and implemented the study and/or provided data but did not necessarily participate in the analysis or writing of this report. This manuscript reflects the views of the authors and may not reflect the opinions or views of the NIH or ABCD consortium investigators.
Contributor Information
Ramana V Vishnubhotla, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, Indianapolis, IN 46202, USA.
Sidra T Ahmad, Indiana University School of Medicine, Indianapolis, IN 46202, USA.
Yi Zhao, Department of Biostatistics and Health Data Science, Indiana University School of Medicine, Indianapolis, IN 46202, USA.
Rupa Radhakrishnan, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, Indianapolis, IN 46202, USA.
Supplementary material
Supplementary material is available at Brain Communications online.
Funding
R.R. and R.V. were supported by the National Institutes of Health R01DA059321 (PI RR) and grant 2021258 from the Doris Duke Charitable Foundation (PI RR) through the COVID-19 Fund to Retain Clinical Scientists collaborative grant program that was made possible through the support of grant 62288 from the John Templeton Foundation. S.A. was supported by the Indiana Clinical and Translational Sciences Institute funded, in part by UL1TR002529 from the National Institutes of Health. Y.Z. was funded through the National Institutes of Health R01MH126970. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The opinions expressed in this publication are those of the authors and do not necessarily reflect the view of the John Templeton Foundation.
Competing interests
The authors have conducted this research in the absence of any commercial, financial or personal relationships that would be considered a conflict of interest.
Data availability
Data for this study were obtained from ABCD® Study (https://abcdstudy.org), held in the NIMH Data Archive.35,36
References
- 1. Tavella RA, DE Abreu VOM, Muccillo-Baisch AL, DA Silva Junior FMR. Prevalence of illicit drug use during pregnancy: A global perspective. An Acad Bras Cienc. 2020;92(4):e20200302. [DOI] [PubMed] [Google Scholar]
- 2. Metz TD, Borgelt LM. Marijuana use in pregnancy and while breastfeeding. Obstet Gynecol. 2018;132(5):1198–1210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Young-Wolff KC, Sarovar V, Tucker LY, et al. Self-reported daily, weekly, and monthly cannabis use among women before and during pregnancy. JAMA Netw Open. 2019;2(7):e196471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Stickrath E. Marijuana use in pregnancy: An updated look at marijuana use and its impact on pregnancy. Clin Obstet Gynecol. 2019;62(1):185–190. [DOI] [PubMed] [Google Scholar]
- 5. Conner SN, Bedell V, Lipsey K, Macones GA, Cahill AG, Tuuli MG. Maternal marijuana use and adverse neonatal outcomes. Obstet Gynecol. 2016;128(4):713–723. [DOI] [PubMed] [Google Scholar]
- 6. Young-Wolff KC, Sarovar V, Tucker LY, et al. Trends in cannabis polysubstance use during early pregnancy among patients in a large health care system in northern California. JAMA Netw Open. 2022;5(6):e2215418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.2020 National Survey on Drug Use and Health: Women. 2020. https://search.library.wisc.edu/catalog/999953798802121
- 8. Young-Wolff KC, Tucker LY, Alexeeff S, et al. Trends in self-reported and biochemically tested marijuana use among pregnant females in California from 2009–2016. JAMA. 2017;318(24):2490–2491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Thompson R, DeJong K, Lo J. Marijuana use in pregnancy: A review. Obstet Gynecol Surv. 2019;74(7):415–428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. de Moraes Barros MC, Guinsburg R, Mitsuhiro S, Chalem E, Laranjeira RR. Neurobehavioral profile of healthy full-term newborn infants of adolescent mothers. Early Hum Dev. 2008;84(5):281–287. [DOI] [PubMed] [Google Scholar]
- 11. Goldschmidt L, Day NL, Richardson GA. Effects of prenatal marijuana exposure on child behavior problems at age 10. Neurotoxicol Teratol. 2000;22(3):325–336. [DOI] [PubMed] [Google Scholar]
- 12. Leech SL, Larkby CA, Day R, Day NL. Predictors and correlates of high levels of depression and anxiety symptoms among children at age 10. J Am Acad Child Adolesc Psychiatry. 2006;45(2):223–230. [DOI] [PubMed] [Google Scholar]
- 13. Fried PA, Watkinson B, Gray R. Differential effects on cognitive functioning in 9- to 12-year olds prenatally exposed to cigarettes and marihuana. Neurotoxicol Teratol. 1998;20(3):293–306. [DOI] [PubMed] [Google Scholar]
- 14. Fried PA. The Ottawa Prenatal Prospective Study (OPPS): Methodological issues and findings–it’s easy to throw the baby out with the bath water. Life Sci. 1995;56(23–24):2159–2168. [DOI] [PubMed] [Google Scholar]
- 15. Smith AM, Mioduszewski O, Hatchard T, Byron-Alhassan A, Fall C, Fried PA. Prenatal marijuana exposure impacts executive functioning into young adulthood: An fMRI study. Neurotoxicol Teratol. 2016;58:53–59. [DOI] [PubMed] [Google Scholar]
- 16. Smith AM, Fried PA, Hogan MJ, Cameron I. Effects of prenatal marijuana on response inhibition: An fMRI study of young adults. Neurotoxicol Teratol. 2004;26(4):533–542. [DOI] [PubMed] [Google Scholar]
- 17. Smith AM, Fried PA, Hogan MJ, Cameron I. Effects of prenatal marijuana on visuospatial working memory: An fMRI study in young adults. Neurotoxicol Teratol. 2006;28(2):286–295. [DOI] [PubMed] [Google Scholar]
- 18. Salzwedel A, Chen G, Chen Y, Grewen K, Gao W. Functional dissection of prenatal drug effects on baby brain and behavioral development. Hum Brain Mapp. 2020;41(17):4789–4803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Faraj MM, Evanski J, Zundel CG, et al. Impact of prenatal cannabis exposure on functional connectivity of the salience network in children. J Neurosci Res. 2023;101(1):162–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Thomason ME, Palopoli AC, Jariwala NN, et al. Miswiring the brain: Human prenatal Delta9-tetrahydrocannabinol use associated with altered fetal hippocampal brain network connectivity. Dev Cogn Neurosci. 2021;51:101000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Grewen K, Salzwedel AP, Gao W. Functional connectivity disruption in neonates with prenatal marijuana exposure. Front Hum Neurosci. 2015;9:601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Wang M, Hu Z, Liu L, Li H, Qian Q, Niu H. Disrupted functional brain connectivity networks in children with attention-deficit/hyperactivity disorder: Evidence from resting-state functional near-infrared spectroscopy. Neurophotonics. 2020;7(1):015012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. McNorgan C, Judson C, Handzlik D, Holden JG. Linking ADHD and behavioral assessment through identification of shared diagnostic task-based functional connections. Front Physiol. 2020;11:583005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Sudre G, Norman L, Bouyssi-Kobar M, Price J, Shastri GG, Shaw P. A mega-analytic study of white matter microstructural differences across 5 cohorts of youths with attention-deficit/hyperactivity disorder. Biol Psychiatry. 2023;94(1):18–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Rohr CS, Bray SL, Dewey DM. Functional connectivity based brain signatures of behavioral regulation in children with ADHD, DCD, and ADHD-DCD. Dev Psychopathol. 2023;35(1):85–94. [DOI] [PubMed] [Google Scholar]
- 26. Bolhuis K, Muetzel RL, Stringaris A, et al. Structural brain connectivity in childhood disruptive behavior problems: A multidimensional approach. Biol Psychiatry. 2019;85(4):336–344. [DOI] [PubMed] [Google Scholar]
- 27. Mears D, Pollard HB. Network science and the human brain: Using graph theory to understand the brain and one of its hubs, the amygdala, in health and disease. J Neurosci Res. 2016;94(6):590–605. [DOI] [PubMed] [Google Scholar]
- 28. Lee JM, Kim PJ, Kim HG, et al. Analysis of brain connectivity during nitrous oxide sedation using graph theory. Sci Rep. 2020;10(1):2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Bullmore E, Sporns O. Complex brain networks: Graph theoretical analysis of structural and functional systems. Nat Rev Neurosci. 2009;10(3):186–198. [DOI] [PubMed] [Google Scholar]
- 30. Sporns O. Graph theory methods: Applications in brain networks. Dialogues Clin Neurosci. 2018;20(2):111–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bassett DS, Bullmore ET. Human brain networks in health and disease. Curr Opin Neurol. 2009;22(4):340–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Chen Y, Huang X, Wu M, et al. Disrupted brain functional networks in drug-naive children with attention deficit hyperactivity disorder assessed using graph theory analysis. Hum Brain Mapp. 2019;40(17):4877–4887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Sidlauskaite J, Caeyenberghs K, Sonuga-Barke E, Roeyers H, Wiersema JR. Whole-brain structural topology in adult attention-deficit/hyperactivity disorder: Preserved global—Disturbed local network organization. Neuroimage Clin. 2015;9:506–512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Wheelock MD, Rangaprakash D, Harnett NG, et al. Psychosocial stress reactivity is associated with decreased whole-brain network efficiency and increased amygdala centrality. Behav Neurosci. 2018;132(6):561–572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Alcohol Research: Current Reviews Editorial Staff . NIH’s Adolescent Brain Cognitive Development (ABCD) Study. Alcohol Res. 2018;39(1):97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Casey BJ, Cannonier T, Conley MI, et al. The Adolescent Brain Cognitive Development (ABCD) Study: Imaging acquisition across 21 sites. Dev Cogn Neurosci. 2018;32:43–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Achenbach TM, Ruffle TM. The Child Behavior Checklist and related forms for assessing behavioral/emotional problems and competencies. Pediatr Rev. 2000;21(8):265–271. [DOI] [PubMed] [Google Scholar]
- 38. Hagler DJ Jr, Hatton S, Cornejo MD, et al. Image processing and analysis methods for the Adolescent Brain Cognitive Development study. Neuroimage. 2019;202:116091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Achenbach TM, Rescorla LA. Manual for ASEBA school-age forms & profiles. University of Vermont, Research Center for Children, Youth & Families; 2001. [Google Scholar]
- 40. Nakamura BJ, Ebesutani C, Bernstein A, Chorpita BF. A psychometric analysis of the child behavior checklist DSM-oriented scales. J Psychopathol Behav Assess. 2009;31:178–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Christian G, Robert D, Paul MT, Florian K, Eileen L. CAT – A Computational Anatomy Toolbox for the analysis of structural MRI data. bioRxiv. 2023:2022.06.11.495736. doi: 10.1101/2022.06.11.495736 [DOI] [Google Scholar]
- 42. Andersson JL, Hutton C, Ashburner J, Turner R, Friston K. Modeling geometric deformations in EPI time series. Neuroimage. 2001;13(5):903–919. [DOI] [PubMed] [Google Scholar]
- 43. Manjon JV, Coupe P, Marti-Bonmati L, Collins DL, Robles M. Adaptive non-local means denoising of MR images with spatially varying noise levels. J Magn Reson Imaging. 2010;31(1):192–203. [DOI] [PubMed] [Google Scholar]
- 44. Ashburner J. A fast diffeomorphic image registration algorithm. Neuroimage. 2007;38(1):95–113. [DOI] [PubMed] [Google Scholar]
- 45. Dahnke R, Yotter RA, Gaser C. Cortical thickness and central surface estimation. Neuroimage. 2013;65:336–348. [DOI] [PubMed] [Google Scholar]
- 46. Yotter RA, Dahnke R, Thompson PM, Gaser C. Topological correction of brain surface meshes using spherical harmonics. Hum Brain Mapp. 2011;32(7):1109–1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Glasser MF, Sotiropoulos SN, Wilson JA, et al. The minimal preprocessing pipelines for the Human Connectome Project. Neuroimage. 2013;80:105–124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Smith SM, Jenkinson M, Johansen-Berg H, et al. Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. Neuroimage. 2006;31(4):1487–1505. [DOI] [PubMed] [Google Scholar]
- 49. Woolrich MW, Jbabdi S, Patenaude B, et al. Bayesian analysis of neuroimaging data in FSL. Neuroimage. 2009;45(1 Suppl):S173–S186. [DOI] [PubMed] [Google Scholar]
- 50. Smith SM. Fast robust automated brain extraction. Hum Brain Mapp. 2002;17(3):143–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Andersson JLR, Jenkinson M, Smith S. Non-linear Optimisation. FMRIB Technial Report TR07JA1. 2007:1–16.
- 52. Andersson JLR, Jenkinson M, Smith S. Non-linear Registration aka Spatial Normalisation. FMRIB Technial Report TR07JA2. 2007:1–21.
- 53. Rueckert D, Sonoda LI, Hayes C, Hill DL, Leach MO, Hawkes DJ. Nonrigid registration using free-form deformations: Application to breast MR images. IEEE Trans Med Imaging. 1999;18(8):712–721. [DOI] [PubMed] [Google Scholar]
- 54. Winkler AM, Ridgway GR, Webster MA, Smith SM, Nichols TE. Permutation inference for the general linear model. Neuroimage. 2014;92:381–397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Yeh FC, Panesar S, Fernandes D, et al. Population-averaged atlas of the macroscale human structural connectome and its network topology. Neuroimage. 2018;178:57–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Yeh FC, Tseng WY. NTU-90: A high angular resolution brain atlas constructed by q-space diffeomorphic reconstruction. Neuroimage. 2011;58(1):91–99. [DOI] [PubMed] [Google Scholar]
- 57. Fonov V, Evans AC, Botteron K, et al. Unbiased average age-appropriate atlases for pediatric studies. Neuroimage. 2011;54(1):313–327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Fonov VS, Evans AC, McKinstry RC, Almli CR, Collins DL. Unbiased nonlinear average age-appropriate brain templates from birth to adulthood. Neuroimage. 2009;47:S102. [Google Scholar]
- 59. Yeh FC, Verstynen TD, Wang Y, Fernandez-Miranda JC, Tseng WY. Deterministic diffusion fiber tracking improved by quantitative anisotropy. PLoS One. 2013;8(11):e80713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Rolls ET, Joliot M, Tzourio-Mazoyer N. Implementation of a new parcellation of the orbitofrontal cortex in the automated anatomical labeling atlas. Neuroimage. 2015;122:1–5. [DOI] [PubMed] [Google Scholar]
- 61. Rubinov M, Sporns O. Complex network measures of brain connectivity: Uses and interpretations. Neuroimage. 2010;52(3):1059–1069. [DOI] [PubMed] [Google Scholar]
- 62. Whitfield-Gabrieli S, Nieto-Castanon A. Conn: A functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2012;2:125–141. [DOI] [PubMed] [Google Scholar]
- 63. Nieto-Castanon A. Handbook of functional connectivity Magnetic Resonance Imaging methods in CONN. Hilbert Press; 2020. doi: 10.56441/hilbertpress.2207.6598 [DOI] [Google Scholar]
- 64. Ashburner J, Friston K. Multimodal image coregistration and partitioning–a unified framework. Neuroimage. 1997;6(3):209–217. [DOI] [PubMed] [Google Scholar]
- 65. Ashburner J, Friston KJ. Unified segmentation. Neuroimage. 2005;26(3):839–851. [DOI] [PubMed] [Google Scholar]
- 66. Behzadi Y, Restom K, Liau J, Liu TT. A component based noise correction method (CompCor) for BOLD and perfusion based fMRI. Neuroimage. 2007;37(1):90–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Chai XJ, Castanon AN, Ongur D, Whitfield-Gabrieli S. Anticorrelations in resting state networks without global signal regression. Neuroimage. 2012;59(2):1420–1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Power JD, Mitra A, Laumann TO, Snyder AZ, Schlaggar BL, Petersen SE. Methods to detect, characterize, and remove motion artifact in resting state fMRI. Neuroimage. 2014;84:320–341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Hallquist MN, Hwang K, Luna B. The nuisance of nuisance regression: Spectral misspecification in a common approach to resting-state fMRI preprocessing reintroduces noise and obscures functional connectivity. Neuroimage. 2013;82:208–225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Calhoun VD, Adali T, Pearlson GD, Pekar JJ. A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp. 2001;14(3):140–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Makris N, Goldstein JM, Kennedy D, et al. Decreased volume of left and total anterior insular lobule in schizophrenia. Schizophr Res. 2006;83(2–3):155–171. [DOI] [PubMed] [Google Scholar]
- 72. Frazier JA, Chiu S, Breeze JL, et al. Structural brain magnetic resonance imaging of limbic and thalamic volumes in pediatric bipolar disorder. Am J Psychiatry. 2005;162(7):1256–1265. [DOI] [PubMed] [Google Scholar]
- 73. Desikan RS, Segonne F, Fischl B, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage. 2006;31(3):968–980. [DOI] [PubMed] [Google Scholar]
- 74. Goldstein JM, Seidman LJ, Makris N, et al. Hypothalamic abnormalities in schizophrenia: Sex effects and genetic vulnerability. Biol Psychiatry. 2007;61(8):935–945. [DOI] [PubMed] [Google Scholar]
- 75. Nieto-Castanon A. General linear model. Handbook of functional connectivity magnetic resonance imaging methods in CONN. Hilbert Press; 2020:63–82. [Google Scholar]
- 76. Benjamini Y, Hochberg Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J R Stat Soc Series B Methodol. 1995;57(1):289–300. [Google Scholar]
- 77. Baranger DAA, Paul SE, Colbert SMC, et al. Association of mental health burden with prenatal cannabis exposure from childhood to early adolescence: Longitudinal findings from the Adolescent Brain Cognitive Development (ABCD) Study. JAMA Pediatr. 2022;176(12):1261–1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. El Marroun H, Tiemeier H, Franken IH, et al. Prenatal cannabis and tobacco exposure in relation to brain morphology: A prospective neuroimaging study in young children. Biol Psychiatry. 2016;79(12):971–979. [DOI] [PubMed] [Google Scholar]
- 79. Hiraoka D, Makita K, Hamatani S, Tomoda A, Mizuno Y. Effects of prenatal cannabis exposure on developmental trajectory of cognitive ability and brain volumes in the Adolescent Brain Cognitive Development (ABCD) Study. Dev Cogn Neurosci. 2023;60:101209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Watts DJ, Strogatz SH. Collective dynamics of ‘small-world’ networks. Nature. 1998;393(6684):440–442. [DOI] [PubMed] [Google Scholar]
- 81. Latora V, Marchiori M. Efficient behavior of small-world networks. Phys Rev Lett. 2001;87(19):198701. [DOI] [PubMed] [Google Scholar]
- 82. Freeman L. A set of measures of centrality based on betweenness. Sociometry. 1977;40:35–41. [Google Scholar]
- 83. Campbell-Meiklejohn DK, Kanai R, Bahrami B, et al. Structure of orbitofrontal cortex predicts social influence. Curr Biol. 2012;22(4):R123–R124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Elliott R, Dolan RJ, Frith CD. Dissociable functions in the medial and lateral orbitofrontal cortex: Evidence from human neuroimaging studies. Cereb Cortex. 2000;10(3):308–317. [DOI] [PubMed] [Google Scholar]
- 85. Kringelbach ML, Rolls ET. The functional neuroanatomy of the human orbitofrontal cortex: Evidence from neuroimaging and neuropsychology. Prog Neurobiol. 2004;72(5):341–372. [DOI] [PubMed] [Google Scholar]
- 86. Tobia MJ, Guo R, Schwarze U, et al. Neural systems for choice and valuation with counterfactual learning signals. Neuroimage. 2014;89:57–69. [DOI] [PubMed] [Google Scholar]
- 87. Feinstein JS, Adolphs R, Damasio A, Tranel D. The human amygdala and the induction and experience of fear. Curr Biol. 2011;21(1):34–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Krabbe S, Grundemann J, Luthi A. Amygdala inhibitory circuits regulate associative fear conditioning. Biol Psychiatry. 2018;83(10):800–809. [DOI] [PubMed] [Google Scholar]
- 89. Ressler RL, Maren S. Synaptic encoding of fear memories in the amygdala. Curr Opin Neurobiol. 2019;54:54–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Roesler R, Parent MB, LaLumiere RT, McIntyre CK. Amygdala-hippocampal interactions in synaptic plasticity and memory formation. Neurobiol Learn Mem. 2021;184:107490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Baxter MG, Murray EA. The amygdala and reward. Nat Rev Neurosci. 2002;3(7):563–573. [DOI] [PubMed] [Google Scholar]
- 92. Murray EA. The amygdala, reward and emotion. Trends Cogn Sci. 2007;11(11):489–497. [DOI] [PubMed] [Google Scholar]
- 93. Carmichael ST, Price JL. Limbic connections of the orbital and medial prefrontal cortex in macaque monkeys. J Comp Neurol. 1995;363(4):615–641. [DOI] [PubMed] [Google Scholar]
- 94. Mao Y, Zuo XN, Ding C, Qiu J. OFC and its connectivity with amygdala as predictors for future social anxiety in adolescents. Dev Cogn Neurosci. 2020;44:100804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Sladky R, Hoflich A, Kublbock M, et al. Disrupted effective connectivity between the amygdala and orbitofrontal cortex in social anxiety disorder during emotion discrimination revealed by dynamic causal modeling for FMRI. Cereb Cortex. 2015;25(4):895–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Bari AA, Sparks H, Levinson S, et al. Amygdala structural connectivity is associated with impulsive choice and difficulty quitting smoking. Front Behav Neurosci. 2020;14:117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Padoa-Schioppa C, Conen KE. Orbitofrontal cortex: A neural circuit for economic decisions. Neuron. 2017;96(4):736–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Roberts AC, Reekie Y, Braesicke K. Synergistic and regulatory effects of orbitofrontal cortex on amygdala-dependent appetitive behavior. Ann N Y Acad Sci. 2007;1121:297–319. [DOI] [PubMed] [Google Scholar]
- 99. Liu T, Thiebaut de Schotten M, Altarelli I, Ramus F, Zhao J. Maladaptive compensation of right fusiform gyrus in developmental dyslexia: A hub-based white matter network analysis. Cortex. 2021;145:57–66. [DOI] [PubMed] [Google Scholar]
- 100. Xiao Y, Wen TH, Kupis L, et al. Atypical functional connectivity of temporal cortex with precuneus and visual regions may be an early-age signature of ASD. Mol Autism. 2023;14(1):11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. He X, Hong J, Liu Z, et al. Decreased functional connectivity of the primary visual cortex and the correlation with clinical features in patients with intermittent exotropia. Front Neurol. 2021;12:638402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Zhou X, Lei X. Wandering minds with wandering brain networks. Neurosci Bull. 2018;34(6):1017–1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Hoekzema E, Carmona S, Ramos-Quiroga JA, et al. An independent components and functional connectivity analysis of resting state fMRI data points to neural network dysregulation in adult ADHD. Hum Brain Mapp. 2014;35(4):1261–1272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Wise T, Marwood L, Perkins AM, et al. Instability of default mode network connectivity in major depression: A two-sample confirmation study. Transl Psychiatry. 2017;7(4):e1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Whitfield-Gabrieli S, Ford JM. Default mode network activity and connectivity in psychopathology. Annu Rev Clin Psychol. 2012;8:49–76. [DOI] [PubMed] [Google Scholar]
- 106. Bailey BA, Osborne JB. Prenatal marijuana exposure and visual perception in toddlers: Evidence of a sensory processing deficit. Front Pediatr. 2023;11:1113047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Roncero C, Valriberas-Herrero I, Mezzatesta-Gava M, Villegas JL, Aguilar L, Grau-Lopez L. Cannabis use during pregnancy and its relationship with fetal developmental outcomes and psychiatric disorders. A systematic review. Reprod Health. 2020;17(1):25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Peterson BS, Rosen T, Dingman S, et al. Associations of maternal prenatal drug abuse with measures of newborn brain structure, tissue organization, and metabolite concentrations. JAMA Pediatr. 2020;174(9):831–842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Paul SE, Hatoum AS, Fine JD, et al. Associations between prenatal cannabis exposure and childhood outcomes: Results from the ABCD Study. JAMA Psychiatry. 2021;78(1):64–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Cioffredi LA, Anderson H, Loso H, et al. Prenatal cannabis exposure predicts attention problems, without changes on fMRI in adolescents. Neurotoxicol Teratol. 2022;91:107089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Dixon DR, Kurtz PF, Chin MD. A systematic review of challenging behaviors in children exposed prenatally to substances of abuse. Res Dev Disabil. 2008;29(6):483–502. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data for this study were obtained from ABCD® Study (https://abcdstudy.org), held in the NIMH Data Archive.35,36