Abstract
In clinical organ transplantation, donor and recipient age may differ substantially. Old donor organs accumulate senescent cells that have the capacity to induce senescence in naïve cells. We hypothesized that the engraftment of old organs may induce senescence in younger recipients, promoting age-related pathologies. When performing isogeneic cardiac transplants between age-mismatched C57BL/6 old donor mice (18-months) and young and middle-aged C57BL/6 recipients (3- or 12-months), we observed augmented frequencies of senescent cells in draining lymph nodes, adipose tissue, livers, and hindlimb muscles 30 days after transplantation. These observations went along with compromised physical performance and impaired spatial learning and memory abilities. Systemic levels of SASP factors including mitochondrial DNA (mt-DNA) were elevated in recipients. Of mechanistic relevance, injections of mt-DNA phenocopied effects of age-mismatched organ transplantation in accelerating aging. Single treatment of old donor animals with senolytics prior to transplantation attenuated mt-DNA release and improved physical capacities in young recipients.
Collectively, we show that transplanting older organs induces senescence in transplant recipients, resulting in compromised physical and cognitive capacities. Depleting senescent cells with senolytics, in turn represents a promising approach to improve outcomes of older organs.
Introduction
Aging is linked to a decline in physical capacity and cognitive functions leading to immobility, frailty, and declining learning and memory abilities(1–5). In transplantation, donor age constitutes a significant risk factor for transplant outcomes, yet the utilization of older organs remains critical to address insufficient organ supply(6, 7). Notably, most transplanted organs today are from donors ≥50(8).
We have recently delineated the accumulation of senescent cells as drivers of compromised transplant outcomes(9). Activated by various stressors, senescence is characterized by stable proliferative arrest and the senescence-associated secretory phenotype (SASP), including interleukin-6 (IL-6), IL-8, GRO-1α, and IGFBP7, capable of inducing a senescent phenotype in neighboring cells (10–13). We found that engraftment of senescent adipose mesenchymal cells into the peritoneal cavity led to augmented populations of senescent cells, inducing muscle weakness and other frailty-like disabilities that persisted for at least 6-months(14). Likewise, senescent cells injected into knee-joints of young mice accelerated osteoarthritis-like symptoms(15). Moreover, heterochronic blood exchange between old and young animals induced senescence in several organs(16).
During transplantation, donor cells are transferred to recipients, leading to a microchimeric state with donor-derived passenger leucocytes detectable for years(17–19). We postulated that the transplantation of old organs into younger recipients promotes senescence either through the transfer of senescent passenger leucocytes or through an augmented release of SASP-factors(20). When transplanting older cardiac grafts, we observed an amplified burden of senescent cells in recipients associated with an increased release of SASP factors including mt-DNA that resulted in compromised physical and cognitive competencies. Depleting senescent cells prior to transplantation with senolytics, in turn reduced mt-DNA levels and restored physical capacity.
Material and Methods
Animals
Young (3-months) and middle-aged (12-months) male C57BL/6-mice were purchased from Charles River Laboratory, Wilmington, MA. Old (18-months) male C57BL/6-mice from the same colony were obtained through the National Institute of Aging. All animal experiments were performed according to protocols approved by the Institutional Animal Care and Use Committees (IACUC) at Brigham and Womeńs Hospital, Harvard Medical School, and Mayo Clinic. Age-matched p16Ink4a-GFP-reporter mice (21-months) were bred at Mayo Clinic. Animals were maintained under specific pathogen-free conditions at the animal facilities of BWH and Mayo Clinic in accordance with federal, state, and institutional guidelines. Animals were maintained on 12-hr light/dark-cycles at an ambient temperature of 19–22 °C at 40–60% humidity with free access to water and standard chow. Euthanasia was performed by cervical dislocation under anesthesia with isoflurane.
Heterotopic Heart Transplantation
Mice were anesthetized with ketamine (100-mg/kg) and xylazine (10-mg/kg). Using a modified cuff technique, vascularized cardiac grafts were heterotopically transplanted into the cervical region of recipients, facilitating reliable functional assessment through daily palpation. Ischemic times were kept consistently at 40-min with an anastomosis time of 12-min.
Mt-DNA isolation and injection
Mitochondria were isolated from whole liver tissue of wild-type male C57BL/6 mice using Mitochondria Isolation Kit for Tissue (Thermo Fisher Scientific). Mitochondrial-DNA was subsequently extracted from isolated mitochondria using QIAmp-Blood & Tissue (Qiagen). Next, middle-aged or old C57BL/6 mice were injected (i.v.) with either 30 μg mt-DNA or PBS; thereafter, physical capacities were tested. We used Cytochrome c oxidase subunit III and NADH-ubiquinone-oxidoreductase-chain-6 (mt-CO3 and mt-ND6) for our analysis.
Immunohistochemistry
For immunofluorescence, organs were kept in formalin for 18-hrs and subsequently embedded in paraffin; paraffin sections were deparaffinized and rehydrated, followed by antigen-retrieval using sodium-citrate-buffer. After 3-washes with TBS, sections were first incubated with 5% normal donkey-serum (Jackson ImmunoResearch) for 1-hr at room temperature, then incubated with mouse anti-p16Ink4a (1:500, Abcam) and rabbit anti-p21Cip1 (1:200, Abcam) primary antibodies overnight at 4°C. Thereafter, slides were washed 3-times and incubated with Cy3-conjugated Donkey anti-rabbit secondary antibody (Jackson ImmunoResearch, 1:300) and Alexa-Fluor-647 conjugated Donkey anti-mouse secondary antibody (Invitrogen, 1:300). Samples were counterstained with Hoechst dye, washed 3-times with TBS, and mounted with Prolong-Gold anti-fade mounting media (Invitrogen).
qPCR
Mt-DNA extraction from plasma was performed using an mt-DNA isolation Kit (QIAamp-DNA-Mini and Blood-Mini-Kit, Qiagen). For qPCR-reactions, mouse cytochrome-c-subunit III (qMmuCEP0060078) and mouse NADH-dehydrogenase-6 (qMmuCEP0062889) measurements were performed using Taqman primers and probes (Bio-Rad). Samples that produced no PCR products after 40-cycles were considered undetectable. For quantification of serum mt-DNA levels, gene-specific synthetic DNA templates (Bio-Rad) were used to obtain standard curves. For analysis of gene expression, RNA was extracted from selected organs using the RNAqueous-extraction-kit (Applied Biosystems). After successive washes, RNA was eluted, and reverse transcription was performed using the i-Script®-cDNA synthesis-kit (Bio-Rad). For qPCR reactions, p16INK4a/Cdkn2a (Mm00494449_m1), p21 (Mm6671726a1), IL-6 (Mm00446190_m1), and TNF-α (Mm00443258_m1) expression measurements were performed with Taqman primers and probes from Applied Biosystems. Relative gene expression was determined using the housekeeping gene GAPDH (Mm99999915_g1) and abundant cytoskeletal housekeeping protein ACTB (NM_007393.1) as controls.
Detection of donor derived senescent cells in recipient animals
To identify potential donor derived senescent cells in recipient mice, hearts from old (21-months) p16Ink4a-GFP-reporter mice were transplanted into young (3-months) C57Bl/6 recipient mice. After 30-days, total-RNA was isolated from muscle, kidney, and inguinal adipose tissue and GFP-expression was quantified using qPCR.
SASP factor quantification in serum
SASP factors were quantified in the serum of recipient animals that had received a (3-months) or old (18-months) cardiac transplant; an additional cohort of recipients received mt-DNA injections. Serum samples were analyzed using the Mouse Cytokine-32-Plex Discovery-Assay (Eve Technologies).
Drug administration
18-month-old C57BL/6 donor mice were treated with a single dose of Dasatinib and Quercetin (D+Q) by oral gavage (5 and 50 mg/kg, respectively; both from Sigma); 24-hrs after treatment, hearts were procured and used for heterotopic HTx.
Neurobehavioral and physical function measurements
A subset of 5–6 mice/group were analyzed consecutively (1–4 months) after HTx. Prior to the tests, mice were randomized and given 15-min to acclimatize.
Grip Strength Test:
Once all paws were on the grip strength meter (Bioseb, Vitrolles, France), mice were pulled back slowly. The force required to make the mouse release its paws was measured. Mice were given 5-consecutive trials with a 20-s inter-trial interval in a holding cage. In addition to the maximum and minimum grip strength measured in these trials, the adjusted average grip strength was calculated after eliminating the lowest and highest force measurements; data are provided in gram (g).
RotaRod Test:
Mice underwent a habituation session at 4-rpm for 5-min on the apparatus, falls were recorded (TSE system). The acceleration rate was at 4–40 rpm over a 3-min interval/trial. The inter-trial interval was 10-min. The latency to fall was recorded in seconds (s) and averaged across 3 trials.
Locomotor Activity:
Arenas consisting of clear Plexiglas (27.3cm × 27.3cm) and equipped with infrared arrays (Med Associates) were used. Mice were placed in the center and allowed to explore freely for 60-min. Distance traveled, vertical beam breaks, and time spent in either the center or the peripheral area were recorded using Med Associates software (Activity Monitor).
Open Field Test:
Mice were placed individually in the center of a Plexiglas-chamber (50cm × 50cm) and allowed to explore freely for 30-min. Mice were video recorded; distance traveled and time spent in either the center or the peripheral area were analyzed by Ethovision Software (Noldus).
Y-maze:
This apparatus consists of three clear acrylic arms (1-start arm and 2-test arms) and a removable partition to block individual arms (Fig.4A). During the first run, one arm was blocked. Mice were placed in the start arm and allowed to explore for 3-min before being placed back in the holding cage for 2-min. Afterwards, mice performed a second trial exploring all arms freely for 3-min. Time spent in the familiar arm and the novel arm was measured using Topscan-software (CleverSys). Percentage of distance and time used to explore the novel arm during the free choice trial was calculated using the formula: %-novel = novel / (novel+familiar)×100.
Figure 4. Recipients of old donor hearts display inferior cognitive performance.
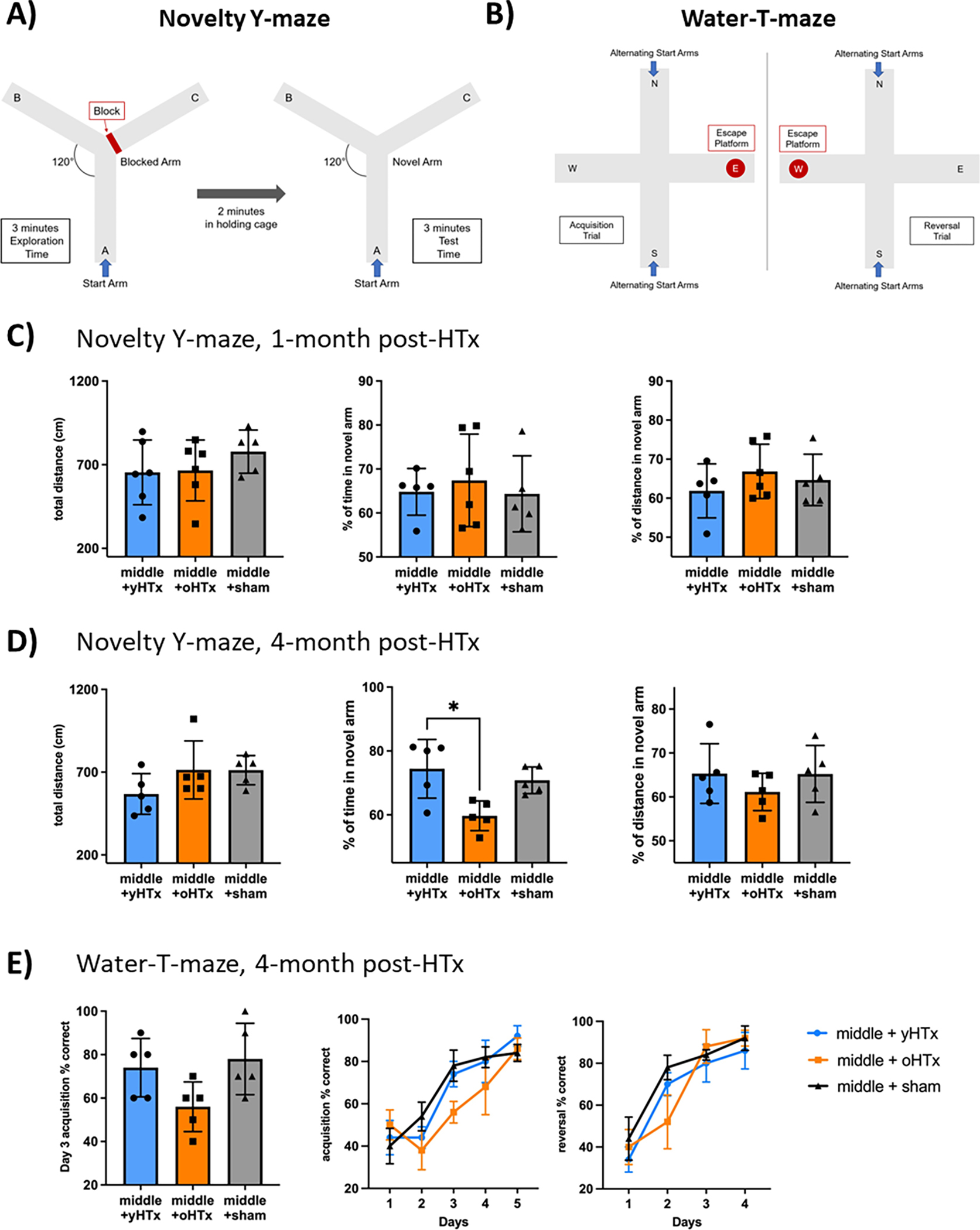
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice served as controls. One and four months after transplant, cognitive tests were performed. (A) + (B) Schematic explanation of Novelty-Y-maze and Water-T-maze. (C) Novelty-Y-maze test after 1-month: Total distance, distance in novel arm, and time in novel arm. (D) Novelty-Y-maze after 4-months. (E) Water-T-maze: Correct responses for each day. Acquisition day three, the acquisition test and the reversal test. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant; cm, centimeter.
Water T-Maze:
A plus-maze with each arm designated as north(N), south(S), east(E), or west(W) was filled with opaque water and a 1-cm submerged escape platform; a divider was placed resulting in a T-maze (Fig.4B). For acquisition, the platform was placed in the E-arm and mice were given 10-trials/day with semi-randomized starting points from the N- and S-positions. Each trial documented whether the mouse found the E-arm at the first attempt. The criterion for acquisition was ≥80% correct responses averaged across 10-trials over 2-consecutive days. After all mice had reached the acquisition criterion, reversal training began. The platform was moved to the W-arm and the same procedure was repeated.
Metabolic Cages:
Metabolic measurements and locomotor activity were assessed using the Columbus-Instruments-Comprehensive-Lab-Animal-Monitoring-System (Columbus Instruments). Mice were individually housed for 4-consecutive days with access to food and water ad-libitum. Lights were on from 7-a.m. to 7-p.m. Activity, respiratory exchange ratios (RER), volumetric rate of oxygen consumption (VO2), and carbon dioxide production (VCO2) were measured. CalR software60 was used for analyzation(21).
Statistics
Kolmogorov-Smirnov and d’Agostino & Pearson-omnibus-normality tests were applied to verify Gaussean distribution before using Student’s T-test with Tukey’s post-test or Dunnett’s Multiple Comparison test for proving statistical significance. Non-parametric data were analyzed by Mann-Whitney-U test. The level of significance was chosen to be at p<0.05 (GraphPadPrism V8).
Results
Transplantation of old cardiac isografts induces cellular senescence in young recipients
We previously discovered an increased burden of senescent cells in old donor organs linked to augmented systemic SASP and mt-DNA levels that increased following IRI(9). Moreover, we have demonstrated that transplanting senescent pre-adipocytes into the abdominal cavity of young mice promoted frailty, inducing senescent cells at distant sites from the injection(14).
To test whether augmented frequencies of senescent cells in donor organs promote senescence in recipients, we transplanted hearts from either young or old C57BL/6 (3- and 18-months) donor mice heterotopically into young syngeneic recipients and quantified senescent cell frequencies using immunofluorescence. Staining for the cellular senescence markers p21Cip1 and p16Ink4a(22) 30-days after transplantation, we detected an augmented frequency of senescent cells in draining lymph nodes and livers of young recipients receiving old hearts compared to recipients of young grafts (Fig.1A). Similarly, p16Ink4a RNA in situ hybridization of abdominal fat depots revealed a trend towards increased expression of p16Ink4a (Fig.1B). Moreover, we detected increased expression of p16Ink4a in muscles of young recipient mice consistent with an augmented burden of senescent cells (Fig.1C). Of note, global p16Ink4a-expression in total-brain tissue of recipient animals was not altered (Fig.1D). Taken together, these findings indicate that transplanting old donor hearts promotes cellular senescence in young recipients.
Figure 1. Transplantation of old cardiac isografts induces cellular senescence in young recipient organs.

Hearts from either young or old C57BL/6 (3- and 18-months) donors were transplanted into young syngeneic recipients. (A) Liver and draining lymph nodes were collected from donors 30 days after engraftment, cut into slides, and co-stained for p16Ink4a, p21Cip1, and DAPI; cell size and percentage of senescent cells was defined as the number of p16/p21 double positive cells for lymph nodes and p16 positive cells of DAPI-stained cells for livers using a confocal microscope. (B) Expression of p16 in inguinal adipose tissue of animals receiving a cardiac transplant from either a young or old INK-ATTAC donor mouse was quantified using p16-RNA in situ hybridization. (C) Furthermore, p16 expression was quantified in femoral muscle (D) and the total brain tissue of young recipient mice by qPCR. Column plots display mean with standard deviation and individual data points are shown. Statistical significance was determined by Student’s T-test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Transplantation of old cardiac isografts compromises physical capacity
When transplanting senescent pre-adipocytes into the abdominal cavity of young mice, we observed a compromised physical capacity by 30-days after transplantation(14). Having observed increased frequencies of senescent cells in several tissues, we next tested whether this phenomenon is also of relevance when transplanting old organs into young recipients (Fig.2A). Strikingly, when performing RotaRod tests in young mice that had received an old cardiac isograft 1-month prior, we found a significantly compromised physical capacity with limited running times. Notably, a trend for compromised physical activity was also observed 1.5-months after transplantation (Fig.2B). Since the transplants had been heterotopic, compromised cardiodynamic function of older hearts as a confounder for the observed effects can be neglected.
Figure 2. Recipients of old donor hearts display inferior physical performance.

(A) Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into young (3-months) and middle-aged (12-months) syngeneic recipients. Sham surgery treated recipients served as controls. Physical tests were performed over a 4-month period after transplantation. (B) RotaRod performance of young recipients. (C) Average, minimum, and maximum grip strength of middle-aged recipients are shown. (D) RotaRod performance of middle-aged recipients. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant; s, seconds; g, gram.
Clinically most recipients today are ≥50-years associated with a significant deterioration of the immune system termed “immunosenesecence”, which has been linked to compromised clearance of senescent cells(23, 24). As older organs are more likely to be transplanted into older recipients, and to preclude excessive clearance of senescent cells in young recipients, we continued our studies in middle-aged recipient mice.
Strikingly, transplanting old organs to middle-aged mice led to compromised four-paw grip-strength, especially impairing minimal strength as recorded by the minimum force required to break the mouse’s grip (Fig.2C). Likewise, RotaRod performance was compromised by 30-days after transplantation, consistent with our observation in younger recipients. Strength differences after the transplantation of older organs were not present by 4-months, suggesting transient effects (Fig.2D). Of relevance, physical performance of sham-treated middle-aged animals was comparable to that of recipients of young hearts. Bodyweights of animals that received either a young or old heart were comparable before and after transplantation (Supp.Fig.1).
Collectively, these results suggest that transplanting older donor hearts impairs physical capacity in both young and middle-aged recipient mice.
Transplantation of old cardiac isografts affects behavior of middle-aged recipient mice
In line with clinical observations, old mice are more likely to suffer from anxiety compared to young mice(25), with senescent cells being contributors(26). Simultaneously, aging is associated with cognitive decline affecting short-term spatial recognition memory, which has been linked to increased numbers of senescent cells within the hippocampus(27). Hence, we next determined if transplanting older organs affects behavior and cognitive function. We assessed the activity of recipients in a locomotor-arena in addition to an open-field-arena, both constituting established methods to evaluate murine locomotion, anxiety, and stereotypical behaviors(28). Notably, recipients of old organs displayed augmented thigmotaxis, spending less time in the center rather than at the walls of the test arenas, reflective of augmented anxiety levels in aging(29). Although this observation was consistent in both tests 30-days after transplantation (Fig.3A+B), no difference was observed after 4-months (Supp. Fig.2A+B). Despite compromised walking distance throughout the experiments, recipients of old hearts demonstrated increased activity levels after initial exposure to the open field arena, suggestive of increased agitation related to anxiety(30) (Fig.3C).
Figure 3. Recipients of old donor hearts display altered behavior.

Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice served as controls. Behavioral tests were performed over the 4-months after transplantation. (A) Total distance of 60-minutes locomotor activity, time, and distance spent in the center are shown 1-month post HTx. (B) 30-minutes open field test, total distance, time, and distance spent in the center 1-month post HTx. (C) Distance in the first 5-minutes of the open field test. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant; cm, centimeter.
To assess potential cognitive implications, we used a Novelty-Y-maze, a test based on the innate curiosity of rodents, which enjoy exploring new environments(31) (Fig.4A). After blocking one arm of the maze during the training period, recipients of old hearts spent less time in the novel arm, indicating worse short-term memory. This phenomenon was observed long-term (4-months) after transplantation, but not at earlier time-points (Fig.4C+D). To further delineate the impact of an older organ transplant on cognitive function, we next performed a Water-maze test 4-months after transplantation. This test requires swimming to find a hidden platform in opaque water, allowing animals to rest outside the water. Mice with greater spatial memory function learn to recognize the position of the platform faster after being randomly placed at different starting points(32) (Fig.4B). Strikingly, recipient mice that received an old organ were more likely to pick the wrong arm during both the acquisition and reversal phases, indicating impaired memory and learning performance as found in old or unhealthy animals(33) (Fig.4E).
Physical impairment following the transplantation of old cardiac isografts is not mediated by metabolic changes
Several studies have demonstrated that metabolic dysfunction including insulin resistance that has been associated with the accumulation of senescent cells in aging(34) can impact the physical capacity of mice(35).
To determine if the observed decline in physical capacity is mediated through systemic metabolic alterations or muscle-intrinsic changes, we performed a CLAMS-analysis in middle-aged recipients. We did not observe any significant differences in VO2, VCO2, RER, metabolic-rate, or food-intake related to the age of organ grafts (Fig.5A+B). These observations were consistent during both day- and nighttime (Supp.Fig.4 A–D). Remarkably, locomotor activity monitored during the metabolic analyses by CLAMS declined in recipient animals of old cardiac isografts by 1- and 4-months after transplantation (Fig.5C), consistent with our observations from the other physical tests. Hence, declined physical capacity is not driven by metabolic impairment but rather by muscle-intrinsic or neurologic dysfunction, supported by our finding of increased p16Ink4a expression in hind limb muscle.
Figure 5. Recipients of old and young donor organs exhibit comparable metabolic profiles.

Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice were included as controls. One and four months after transplant, metabolic cage analysis over 24-hours was performed. Hereby, average oxygen consumption (milliliter/hour), average carbon dioxide production (milliliter/hour), average respiratory exchange ratio (carbon dioxide production/oxygen consumption), and average food consumption were analyzed. (A) Analysis 1-month after transplantation. (B) Analysis 4-months after transplantation. (C) Locomotor activity (beam breaks) of animals while in metabolic cages one and 4-months after transplantation. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Induction of senescence following transplantation is linked to augmented release of SASP-factors including mt-DNA
As the augmented burden of senescent cells in young recipients of old donor hearts correlated with physical and cognitive dysfunction following transplantation, we explored underlying mechanisms. Both an increased number of donor-derived senescent passenger leukocytes(17) and increased SASP-factors related to IRI(36) may explain symptoms of accelerated senescence subsequent to the transplantation of older organs.
To delineate the impact of senescent donor-derived passenger cells, we used p16Ink4a-GFP reporter mice that co-express the senescent cell marker p16Ink4a linked to GFP-protein(37) that can be quantified by qPCR in recipient organs following transplantation. Thirty days after engraftment, we quantified p16Ink4a-GFP expression in recipient kidneys, muscle, and adipose tissue by qPCR. Notably, we did not observe significant GFP-expression, indicating limited migration of donor-derived senescent cells into recipient animals (Fig.6A).
Figure 6. Transplanting older organs promotes senescence in recipients through augmented release of SASP-factors including mt-DNA.

(A) Hearts from either young or old p16Ink4a-GFP reporter mice (3- and 18-months) were transplanted into young or middle-aged syngeneic recipients. After 30 days, GFP expression was quantified in kidneys, muscle, and adipose tissue by qPCR. (B) Hearts from either young or old C57BL/6 donor mice (3- and 18-months) were transplanted into young or middle-aged syngeneic recipients (3- and 12-months). 24-hours after transplantation, mitochondrial DNA was quantified in plasma by real-time PCR according to standard curve results after DNA isolation. (C) Middle-aged mice were exposed to either isolated mt-DNA or PBS injections and physical performance was subsequently assessed using a standardized rotarod tests. (D) By two weeks, p21, p16, IL-6, and TNF-α levels were quantified in brain sections from the hippocampus by qPCR. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by Student’s T-test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.
SASP-factors have the capacity to induce senescence in adjacent and distant cells(10–12). Furthermore, we have recently identified mt-DNA as a SASP-factor originating from senescent cells that is augmented during IRI(9). Next, we determined if the augmented senescence in recipients of old organs was a result of augmented SASP-factor release and quantified SASP factors and systemic mt-DNA levels in recipient animals by 1-month after transplantation. Several SASP factors including eotaxin, IP-10, Gro-α, MCP-1, MIP, IL-13, and VEGF that have been shown to induce senescence and age-related pathologies in young mice(10, 16, 36, 38–40) were increased in recipients of old hearts (Supp.Fig.5). Moreover, systemic mt-DNA levels were significantly elevated (Fig.6B). To evaluate augmented mt-DNA levels as a potential driver of senescence induction, we tested if isolated mt-DNA injections could imitate the effects of age-mismatched organ transplantation. Indeed, mt-DNA injections led to an increase of SASP factors comparable to effects observed when transplanting older hearts (Supp.Fig.6). More importantly, we observed a compromised RotaRod-performance of animals that had received mt-DNA injections (Fig.6C) comparable to recipients of old organs (Fig.2B+D). Since we had not observed altered p16Ink4a-levels in our total brain analysis (Fig.1D), we next analyzed the expression of senescence and inflammaging markers in brain tissue derived from the hippocampus as a center for memory regulation(41). Remarkably, animals injected with mt-DNA demonstrated localized elevated levels of p16Ink4a, p21Cip1, IL-6, and TNF-α mRNA 14-days after injection (Fig.6D).
These results indicate that advanced donor age promotes the release of various SASP factors including mt-DNA inducing senescence in recipient animals that contribute to physical and cognitive dysfunction following transplantation.
Treating old donor mice with senolytics improves physical capacity of recipients
Senolytics deplete senescent cells through inhibiting senescence related anti-apoptotic pathways, alleviating a broad range of age-associated pathologies, including frailty and cognitive impairment(42). Moreover, senolytics applied during cardiac IRI have been shown to decrease SASP-driven induction of senescence(36, 43). To test if senolytics could impede induction of senescence following engraftment of old organs, we treated old donor mice with D+Q prior to organ procurement. Subsequently, we quantified systemic mt-DNA levels by qPCR and assessed the physical capacity of recipient mice over a 1.5-month period. Both young and middle-aged recipient mice with grafts from an old donor treated with senolytics showed significantly reduced mt-DNA levels (Fig.7A). Moreover, young animals receiving an old donor heart that had been treated with senolytics exhibited enhanced RotaRod-performance 4- and 6-weeks after transplantation (Fig.7B). These results indicate that senolytics have the capacity to attenuate the spread of senescence caused by age-mismatched organ transplantation.
Figure 7. Treating old donor mice with senolytics improves physical performance in recipients of organs transplanted from the old donors.

Young and old (3- and 18-months) C57BL/6 mice as well as old C57BL/6 donor mice that had been treated with a single dose of Dasatinib and Quercetin prior to transplantation served as donor mice. (A) 24 hours following syngeneic transplantation of cardiac isografts into young or middle-aged recipient mice, systemic mt-DNA levels were quantified by qPCR. (B) 30 days following transplantation, physical capacity of recipient animals was assessed by a RotaRod test. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by Student’s T-test or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.
Discussion
We have shown that the engraftment of old donor organs impacts both physical and cognitive capacity through an augmented burden of senescent cells in recipients. Increased p16Ink4a-expression in femoral muscles was linked to compromised physical performance as normally observed in old animals(44). Moreover, transplantation of old organs impacted the cognitive capacity of recipient animals, with augmented anxiety and compromised spatial working memory, consistent with a phenotype usually observed in old mice(45, 46). Our total-brain analysis was not able to detect increased p16Ink4a-levels in mice receiving an old donor heart, requiring further regional anatomic analyzes, particularly focusing on the hippocampus as a center for memory(41). Notably, when injecting mt-DNA, we observed elevated p16Ink4-levels in the hippocampus along with other senescence markers including p21Cip1, IL-6 and TNF-α. While the association between elevated IL-6 and cognitive dysfunction are well accepted(47, 48), TNF-α also seems to impair the function of the hippocampus, a region critically involved in learning with recent studies highlighting the importance of age-stratified TNF-α analysis(49, 50). In line with our findings, advanced senescence in the hippocampus have been associated with compromised Y-maze performance in a murine chronic stress model(51).
Mechanistically, we excluded donor-derived passenger leukocytes as mediators of senescence induction, yet observed a release of SASP factors, including a prominent increase of systemic mt-DNA levels following the transplantation of older organs. Several of these SASP-factors, including eotaxin, IP-10, Gro-α, MCP-1, MIP, IL-13, and VEGF have been reported to induce senescence and age-related pathologies(10, 16, 36, 38–40). Accordingly, heterochronic blood exchange from old into young mice resulted in increased systemic SASP-factor levels, inducing senescence across several tissues including liver and muscle(16). Our data suggest that the engraftment of old organs promotes an augmented release of SASP-factors in recipient animals, including mt-DNA, potentially contributing to increased senescent cell burden, ultimately causing physical and cognitive dysfunction.
Restored RotaRod-performances by 4-months indicate that physical impairments are particularly pronounced shortly after transplantation and at least partly related to perioperative conditions, including IRI. We have previously shown that mt-DNA is released subsequent to IRI in old organs(9). Based on our data showing mt-DNA to induce senescence, we hypothesize that IRI during transplantation of old organs enhances the release of SASP factors and mt-DNA, advancing senescence in recipients. Future experiments will need to determine if effects of an accelerated aging are time-limited or continue to persist long-term. Of additional relevance, immune cell-mediated mechanisms eliminating senescent cells may help to attenuate long-term consequences(23).
Some of our results suggest long-term consequences of heterochronic transplants as demonstrated by the compromised distance traveled within 24-hours and the impairment in cognitive function 4-months after transplantation. We have clinically and experimentally shown that older organs contain increased numbers of senescent cells as a source of mt-DNA(9). Hence, transplanting older organs could lead to continuous release of SASP factors including mt-DNA, potentially promoting durable senescence, while compromised capacities for clearing senescent cells with age appear disadvantageous(52–54).
Previously, we reported that the amount of mt-DNA rather than the age of the animal from which mt-DNA was isolated triggered SASP production(9). Considering the significantly augmented release of mt-DNA subsequent to isolated IRI or transplantation of older organs(9), we submit that the quantity rather than the ‘quality’ of mt-DNA drives senescence. Injecting young mice with mt-DNA isolated from young animals phenocopied effects of age-mismatched organs indicating that high quantities of mt-DNA per se are sufficient to induce senescence.
Senolytics have the potential to inhibit senescence following organ transplantation. Administration of senolytics prevented systemic induction of senescence in young mice receiving old blood(16) and in a model of cardiac IRI(36) while reducing the release of mt-DNA following IRI(9). In our current experiments, senolytics diminished mt-DNA levels and enhanced physical capacity in recipients of older organs, emphasizing on a potential therapeutic approach for inhibiting the induction of senescence. Senolytic treatment has been shown to improve cognitive function in a murine model of Alzheimer’s disease(55) while improving physical and cognitive function in wire hanging and Stone’s T-maze test, respectively(14, 27). Of translational relevance, our recent open label clinical trial investigating the impact of D+Q on idiopathic pulmonal fibrosis also suggested improved physical function(56).
Previous studies have shown that IRI causes upregulation of SASP proteins including IL-6, IP-10, and eotaxin, all of which were also upregulated in analysis after the transplantation of old organs(36). While in our living donor transplant model both warm and cold ischemic times have been short, ischemic times are often longer in clinical reality(57, 58). We submit that prolonged ischemic periods – either warm or cold - will likely amplify the effects observed in our experiments. Likewise, our results emphasize on an acceleration of aging early after transplantation while the question of long-term effects remains to be defined. As common immunosuppressants appear to interact with senescence(59–61), future studies are also needed to determine the effects of immunosuppression on the induction of senescence.
Of additional relevance may be the investigation of methylation and histone acetylation of mt-DNA since altered patterns have been suggested to regulate cellular senescence(62–65).
While our study provides novel insights, there remain open questions regarding clinical impact and mechanisms involved. Experiments assessing cognitive and physical exercise by nature do not always lead to largely different effects, yet the consistency of our results across different scenarios support validity and robustness.
Collectively, our study reveals that senescence can be triggered by transplanting old organs, leading to molecular and cellular changes as well as accelerated aging-related physical and cognitive impairments. Senolytics prove effective in preventing senescence when given to old donors. These findings have potential clinical implications as older organs are increasingly used in transplantation.
Supplementary Material
Supplementary Figure 1. Recipients of old and young donor organs have comparable body weight
Body weight of recipient animals was measured (A) prior to and (B) after HTx. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 5. Transplanting older organs promotes senescence in recipients through augmented release of SASP factors
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. After 30 days, systemic SASP factor concentrations were assessed by Multi-ELISA. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 6. Increased mt-DNA levels induce senescence with augmented levels of systemic SASP factors
Middle-aged C57BL/6 mice (12-months) were subjected to mt-DNA or PBS injections. 2 weeks after injection, systemic SASP factor concentrations were assessed by Multi-ELISA. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: mtDNA, mitochondrial DNA.
Supplementary Figure 2. Recipients of old donor hearts have compromised hanging endurance 4 months after transplantation
Hearts from either young or old C57BL/6 (2- and 18-months) were transplanted into young (3-months) and middle-aged (12 months) syngeneic recipients. Sham surgery treated recipients served as controls. Physical tests were performed over a 4-months period after transplantation. (A) Average wire hanging endurance of middle-aged recipients. (B) Average grid hanging endurance of middle-aged recipients. Column plots display mean with standard deviation. Statistical significance was determined by Student’s T-test. Asterisks indicate p-values * = p≤0.05, *= p≤0.01 and ** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant; s, seconds; g, gram.
Supplementary Figure 3. Behavior is not altered significantly in recipients of old donor organs 4-months after transplantation
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice served as controls. Behavioral tests were performed 4-months after transplantation. (A) Total distance of 60-minutes locomotor activity, time, and distance spent in the center are shown. (B) 30-minutes Open field test, total distance, time, and distance spent in the center. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 4. Recipients of old and young donor organs exhibit comparable metabolic profiles during daytime and night
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice were included as controls. One and four months after transplant, metabolic cage analysis over 24-hours was performed. Average oxygen consumption (milliliter/hour), average carbon dioxide production (milliliter/hour), average respiratory exchange ratio (carbon dioxide production/oxygen consumption), and average food consumption were analyzed. (A) Analysis 1-month after transplantation during daytime. (B) Analysis 1-month after transplantation during nighttime. (C) Analysis 4-months after transplantation during daytime. (D) Analysis 4-month after transplantation during nighttime. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.
Acknowledgements
SGT is supported by grants from the National Institutes of Health (R01AG064165). T.T. and J.L.K. are supported by NIH grants R37 AG013925 and R33AG 61456. J.I. was supported by the Junior Clinician Scientist Program of the Berlin Institute of Health. J.I., M.J.R., and A.S. were supported by the Biomedical Education Program (BMEP) of the German Academic Exchange Service. T.M. and R.M. were supported by the Osaka Medical Foundation. U.T. was supported by an American Heart Association predoctoral fellowship (917775). Y.N. was supported by the Chinese Scholarship Council (201606370196) and Central South University. T.T. and J.L.K. were supported by the Connor Group, Robert J. and Theresa W. Ryan, and the Noaber and Ted Nash Long Life Foundations.
The Transplant Surgery Research Laboratory is supported by an unrestricted grant of the Pablo and Almudena Legorreta Kidney Health Research Fund.
Abbreviations:
- IRI
ischemia reperfusion injury
- Mt-DNA
mitochondrial DNA
- SASP
senescence-associated secretory phenotype
- NIA
National Institute of Aging
- IACUC
Institutional Animal Care and Use Committees
- D+Q
Dasatinib and Quercetin
- RER
respiratory exchange ratios
- VO2
volumetric rate of oxygen consumption
- VCO2
carbon dioxide production
- IPF
Idiopathic Pulmonary Fibrosis
Footnotes
Competing interests
J.L.K., T.T., and S.G.T. have a financial interest related to this research. Patents on senolytic drugs are held by Mayo Clinic and Brigham and Women’s Hospital. This research has been reviewed by the Mayo Clinic Conflict of Interest Review Board and was conducted in compliance with Mayo Clinic Conflict of Interest policies. No conflicts of interest, financial or otherwise, are declared by the other authors.
Declaration of interests
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:
Stefan G. Tullius has patent Methods And Materials For Improving Transplant Outcomes licensed to Stefan G. Tullius.
James L. Kirkland has patent Methods And Materials For Improving Transplant Outcomes licensed to Stefan G. Tullius.
Tamar Tchkonia has patent Methods And Materials For Improving Transplant Outcomes licensed to Stefan G. Tullius.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Daley MJ, Spinks WL. Exercise, mobility and aging. Sports Med 2000;29(1):1–12. [DOI] [PubMed] [Google Scholar]
- 2.Groessl EJ, Kaplan RM, Rejeski WJ, Katula JA, Glynn NW, King AC et al. Physical Activity and Performance Impact Long-term Quality of Life in Older Adults at Risk for Major Mobility Disability. Am J Prev Med 2019;56(1):141–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Clark R, Freedberg M, Hazeltine E, Voss MW. Are There Age-Related Differences in the Ability to Learn Configural Responses? PLoS One 2015;10(8):e0137260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Foster TC, Defazio RA, Bizon JL. Characterizing cognitive aging of spatial and contextual memory in animal models. Front Aging Neurosci 2012;4:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hou Y, Dan X, Babbar M, Wei Y, Hasselbalch SG, Croteau DL et al. Ageing as a risk factor for neurodegenerative disease. Nat Rev Neurol 2019;15(10):565–581. [DOI] [PubMed] [Google Scholar]
- 6.Tullius SG, Milford E. Kidney allocation and the aging immune response. N Engl J Med 2011;364(14):1369–1370. [DOI] [PubMed] [Google Scholar]
- 7.Saidi RF, Hejazii Kenari SK. Challenges of organ shortage for transplantation: solutions and opportunities. Int J Organ Transplant Med 2014;5(3):87–96. [PMC free article] [PubMed] [Google Scholar]
- 8.Hart A, Smith JM, Skeans MA, Gustafson SK, Stewart DE, Cherikh WS et al. OPTN/SRTR 2015 Annual Data Report: Kidney. Am J Transplant 2017;17 Suppl 1:21–116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iske J, Seyda M, Heinbokel T, Maenosono R, Minami K, Nian Y et al. Senolytics prevent mt-DNA-induced inflammation and promote the survival of aged organs following transplantation. Nat Commun 2020;11(1):4289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Acosta JC, O’Loghlen A, Banito A, Guijarro MV, Augert A, Raguz S et al. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 2008;133(6):1006–1018. [DOI] [PubMed] [Google Scholar]
- 11.Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 2008;133(6):1019–1031. [DOI] [PubMed] [Google Scholar]
- 12.Wajapeyee N, Serra RW, Zhu X, Mahalingam M, Green MR. Oncogenic BRAF induces senescence and apoptosis through pathways mediated by the secreted protein IGFBP7. Cell 2008;132(3):363–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Herranz N, Gil J. Mechanisms and functions of cellular senescence. J Clin Invest 2018;128(4):1238–1246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xu M, Pirtskhalava T, Farr JN, Weigand BM, Palmer AK, Weivoda MM et al. Senolytics improve physical function and increase lifespan in old age. Nat Med 2018;24(8):1246–1256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xu M, Bradley EW, Weivoda MM, Hwang SM, Pirtskhalava T, Decklever T et al. Transplanted Senescent Cells Induce an Osteoarthritis-Like Condition in Mice. J Gerontol A Biol Sci Med Sci 2017;72(6):780–785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jeon OH, Mehdipour M, Gil TH, Kang M, Aguirre NW, Robinson ZR et al. Systemic induction of senescence in young mice after single heterochronic blood exchange. Nat Metab 2022;4(8):995–1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Monaco AP. Chimerism in organ transplantation: conflicting experiments and clinical observations. Transplantation 2003;75(9 Suppl):13s–16s. [DOI] [PubMed] [Google Scholar]
- 18.Richter N, Raddatz G, Graeter T, Schäfers HJ, Schlitt HJ. Allogeneic lymphocyte chimerism after clinical lung transplantation. Transpl Immunol 1995;3(1):74–80. [DOI] [PubMed] [Google Scholar]
- 19.Schlitt HJ, Kanehiro H, Raddatz G, Steinhoff G, Richter N, Nashan B et al. Persistence of donor lymphocytes in liver allograft recipients. Transplantation 1993;56(4):1001–1007. [DOI] [PubMed] [Google Scholar]
- 20.Iske J, Matsunaga T, Zhou H, Tullius SG. Donor and Recipient Age-Mismatches: The Potential of Transferring Senescence. Front Immunol 2021;12:671479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mina AI, LeClair RA, LeClair KB, Cohen DE, Lantier L, Banks AS. CalR: A Web-Based Analysis Tool for Indirect Calorimetry Experiments. Cell Metab 2018;28(4):656–666 e651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hernandez-Segura A, Nehme J, Demaria M. Hallmarks of Cellular Senescence. Trends Cell Biol 2018;28(6):436–453. [DOI] [PubMed] [Google Scholar]
- 23.Prata L, Ovsyannikova IG, Tchkonia T, Kirkland JL. Senescent cell clearance by the immune system: Emerging therapeutic opportunities. Semin Immunol 2018;40:101275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Karin O, Agrawal A, Porat Z, Krizhanovsky V, Alon U. Senescent cell turnover slows with age providing an explanation for the Gompertz law. Nat Commun 2019;10(1):5495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shoji H, Takao K, Hattori S, Miyakawa T. Age-related changes in behavior in C57BL/6J mice from young adulthood to middle age. Mol Brain 2016;9:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ogrodnik M, Zhu Y, Langhi LGP, Tchkonia T, Kruger P, Fielder E et al. Obesity-Induced Cellular Senescence Drives Anxiety and Impairs Neurogenesis. Cell Metab 2019;29(5):1061–1077 e1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ogrodnik M, Evans SA, Fielder E, Victorelli S, Kruger P, Salmonowicz H et al. Whole-body senescent cell clearance alleviates age-related brain inflammation and cognitive impairment in mice. Aging Cell 2021;20(2):e13296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kraeuter AK, Guest PC, Sarnyai Z. The Open Field Test for Measuring Locomotor Activity and Anxiety-Like Behavior. Methods Mol Biol 2019;1916:99–103. [DOI] [PubMed] [Google Scholar]
- 29.Meeker HC, Chadman KK, Heaney AT, Carp RI. Assessment of social interaction and anxiety-like behavior in senescence-accelerated-prone and -resistant mice. Physiol Behav 2013;118:97–102. [DOI] [PubMed] [Google Scholar]
- 30.Mintzer JE, Brawman-Mintzer O. Agitation as a possible expression of generalized anxiety disorder in demented elderly patients: toward a treatment approach. J Clin Psychiatry 1996;57 Suppl 7:55–63; discussion 73–55. [PubMed] [Google Scholar]
- 31.Prieur EAK, Jadavji NM. Assessing Spatial Working Memory Using the Spontaneous Alternation Y-maze Test in Aged Male Mice. Bio Protoc 2019;9(3):e3162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Davis KE, Burnett K, Gigg J. Water and T-maze protocols are equally efficient methods to assess spatial memory in 3xTg Alzheimer’s disease mice. Behav Brain Res 2017;331:54–66. [DOI] [PubMed] [Google Scholar]
- 33.de Fiebre NC, Sumien N, Forster MJ, de Fiebre CM. Spatial learning and psychomotor performance of C57BL/6 mice: age sensitivity and reliability of individual differences. Age (Dordr) 2006;28(3):235–253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xu M, Palmer AK, Ding H, Weivoda MM, Pirtskhalava T, White TA et al. Targeting senescent cells enhances adipogenesis and metabolic function in old age. Elife 2015;4:e12997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ostler JE, Maurya SK, Dials J, Roof SR, Devor ST, Ziolo MT et al. Effects of insulin resistance on skeletal muscle growth and exercise capacity in type 2 diabetic mouse models. Am J Physiol Endocrinol Metab 2014;306(6):E592–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dookun E, Walaszczyk A, Redgrave R, Palmowski P, Tual-Chalot S, Suwana A et al. Clearance of senescent cells during cardiac ischemia-reperfusion injury improves recovery. Aging Cell 2020;19(10):e13249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 2011;479(7372):232–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Coppé JP, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol 2008;6(12):2853–2868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Acosta JC, Banito A, Wuestefeld T, Georgilis A, Janich P, Morton JP et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat Cell Biol 2013;15(8):978–990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhu M, Min S, Mao X, Zhou Y, Zhang Y, Li W et al. Interleukin-13 promotes cellular senescence through inducing mitochondrial dysfunction in IgG4-related sialadenitis. Int J Oral Sci 2022;14(1):29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Anderson ML, Kinnison J, Pessoa L. Describing functional diversity of brain regions and brain networks. Neuroimage 2013;73:50–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Al-Naggar IMA, Kuchel GA, Xu M. Senolytics: targeting senescent cells for age-associated diseases. Curr Mol Biol Rep 2020;6(4):161–172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lee JR, Park BW, Park JH, Lim S, Kwon SP, Hwang JW et al. Local delivery of a senolytic drug in ischemia and reperfusion-injured heart attenuates cardiac remodeling and restores impaired cardiac function. Acta Biomater 2021;135:520–533. [DOI] [PubMed] [Google Scholar]
- 44.Graber TG, Maroto R, Fry CS, Brightwell CR, Rasmussen BB. Measuring Exercise Capacity and Physical Function in Adult and Older Mice. J Gerontol A Biol Sci Med Sci 2021;76(5):819–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shoji H, Miyakawa T. Age-related behavioral changes from young to old age in male mice of a C57BL/6J strain maintained under a genetic stability program. Neuropsychopharmacol Rep 2019;39(2):100–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bromley-Brits K, Deng Y, Song W. Morris water maze test for learning and memory deficits in Alzheimer’s disease model mice. J Vis Exp 2011(53). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Maggio M, Guralnik JM, Longo DL, Ferrucci L. Interleukin-6 in aging and chronic disease: a magnificent pathway. J Gerontol A Biol Sci Med Sci 2006;61(6):575–584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Weaver JD, Huang MH, Albert M, Harris T, Rowe JW, Seeman TE. Interleukin-6 and risk of cognitive decline: MacArthur studies of successful aging. Neurology 2002;59(3):371–378. [DOI] [PubMed] [Google Scholar]
- 49.Takahashi S, Fukushima H, Yu Z, Tomita H, Kida S. Tumor necrosis factor α negatively regulates the retrieval and reconsolidation of hippocampus-dependent memory. Brain Behav Immun 2021;94:79–88. [DOI] [PubMed] [Google Scholar]
- 50.Mygind L, Bergh MS, Tejsi V, Vaitheeswaran R, Lambertsen KL, Finsen B et al. Tumor Necrosis Factor (TNF) Is Required for Spatial Learning and Memory in Male Mice under Physiological, but Not Immune-Challenged Conditions. Cells 2021;10(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lin YF, Wang LY, Chen CS, Li CC, Hsiao YH. Cellular senescence as a driver of cognitive decline triggered by chronic unpredictable stress. Neurobiol Stress 2021;15:100341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 2014;15(7):482–496. [DOI] [PubMed] [Google Scholar]
- 53.Song P, An J, Zou MH. Immune Clearance of Senescent Cells to Combat Ageing and Chronic Diseases. Cells 2020;9(3). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yousefzadeh MJ, Flores RR, Zhu Y, Schmiechen ZC, Brooks RW, Trussoni CE et al. An aged immune system drives senescence and ageing of solid organs. Nature 2021;594(7861):100–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhang P, Kishimoto Y, Grammatikakis I, Gottimukkala K, Cutler RG, Zhang S et al. Senolytic therapy alleviates Aβ-associated oligodendrocyte progenitor cell senescence and cognitive deficits in an Alzheimer’s disease model. Nat Neurosci 2019;22(5):719–728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Justice JN, Nambiar AM, Tchkonia T, LeBrasseur NK, Pascual R, Hashmi SK et al. Senolytics in idiopathic pulmonary fibrosis: Results from a first-in-human, open-label, pilot study. EBioMedicine 2019;40:554–563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhao H, Alam A, Soo AP, George AJT, Ma D. Ischemia-Reperfusion Injury Reduces Long Term Renal Graft Survival: Mechanism and Beyond. EBioMedicine 2018;28:31–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Salvadori M, Rosso G, Bertoni E. Update on ischemia-reperfusion injury in kidney transplantation: Pathogenesis and treatment. World J Transplant 2015;5(2):52–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Koppelstaetter C, Kern G, Leierer G, Mair SM, Mayer G, Leierer J. Effect of cyclosporine, tacrolimus and sirolimus on cellular senescence in renal epithelial cells. Toxicol In Vitro 2018;48:86–92. [DOI] [PubMed] [Google Scholar]
- 60.Kim YC, Guan KL. mTOR: a pharmacologic target for autophagy regulation. J Clin Invest 2015;125(1):25–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Singh AK, Singh S, Garg G, Rizvi SI. Rapamycin mitigates erythrocyte membrane transport functions and oxidative stress during aging in rats. Arch Physiol Biochem 2018;124(1):45–53. [DOI] [PubMed] [Google Scholar]
- 62.Sakaki M, Ebihara Y, Okamura K, Nakabayashi K, Igarashi A, Matsumoto K et al. Potential roles of DNA methylation in the initiation and establishment of replicative senescence revealed by array-based methylome and transcriptome analyses. PLOS ONE 2017;12(2):e0171431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang Y, Gao J, Wu F, Lai C, Li Y, Zhang G et al. Biological and epigenetic alterations of mitochondria involved in cellular replicative and hydrogen peroxide-induced premature senescence of human embryonic lung fibroblasts. Ecotoxicology and Environmental Safety 2021;216:112204. [DOI] [PubMed] [Google Scholar]
- 64.Yu D, Du Z, Pian L, Li T, Wen X, Li W et al. Mitochondrial DNA Hypomethylation Is a Biomarker Associated with Induced Senescence in Human Fetal Heart Mesenchymal Stem Cells. Stem Cells Int 2017;2017:1764549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Di Giorgio E, Paluvai H, Dalla E, Ranzino L, Renzini A, Moresi V et al. HDAC4 degradation during senescence unleashes an epigenetic program driven by AP-1/p300 at selected enhancers and super-enhancers. Genome Biol 2021;22(1):129. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1. Recipients of old and young donor organs have comparable body weight
Body weight of recipient animals was measured (A) prior to and (B) after HTx. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 5. Transplanting older organs promotes senescence in recipients through augmented release of SASP factors
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. After 30 days, systemic SASP factor concentrations were assessed by Multi-ELISA. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 6. Increased mt-DNA levels induce senescence with augmented levels of systemic SASP factors
Middle-aged C57BL/6 mice (12-months) were subjected to mt-DNA or PBS injections. 2 weeks after injection, systemic SASP factor concentrations were assessed by Multi-ELISA. Column plots display mean with standard deviation with individual data points being shown. Abbreviations: mtDNA, mitochondrial DNA.
Supplementary Figure 2. Recipients of old donor hearts have compromised hanging endurance 4 months after transplantation
Hearts from either young or old C57BL/6 (2- and 18-months) were transplanted into young (3-months) and middle-aged (12 months) syngeneic recipients. Sham surgery treated recipients served as controls. Physical tests were performed over a 4-months period after transplantation. (A) Average wire hanging endurance of middle-aged recipients. (B) Average grid hanging endurance of middle-aged recipients. Column plots display mean with standard deviation. Statistical significance was determined by Student’s T-test. Asterisks indicate p-values * = p≤0.05, *= p≤0.01 and ** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant; s, seconds; g, gram.
Supplementary Figure 3. Behavior is not altered significantly in recipients of old donor organs 4-months after transplantation
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice served as controls. Behavioral tests were performed 4-months after transplantation. (A) Total distance of 60-minutes locomotor activity, time, and distance spent in the center are shown. (B) 30-minutes Open field test, total distance, time, and distance spent in the center. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTx, old heart transplant; yHTx, young heart transplant.
Supplementary Figure 4. Recipients of old and young donor organs exhibit comparable metabolic profiles during daytime and night
Hearts from either young or old C57BL/6 (3- and 18-months) were transplanted into middle-aged (12-months) syngeneic recipients. Sham surgery treated middle-aged mice were included as controls. One and four months after transplant, metabolic cage analysis over 24-hours was performed. Average oxygen consumption (milliliter/hour), average carbon dioxide production (milliliter/hour), average respiratory exchange ratio (carbon dioxide production/oxygen consumption), and average food consumption were analyzed. (A) Analysis 1-month after transplantation during daytime. (B) Analysis 1-month after transplantation during nighttime. (C) Analysis 4-months after transplantation during daytime. (D) Analysis 4-month after transplantation during nighttime. Column plots display mean with standard deviation with individual data points being shown. Statistical significance was determined by One-Way ANOVA or Friedmann Test. Asterisks indicate p-values * = p≤0.05, **= p≤0.01 and *** = p≤0.001, only significant values are shown. Abbreviations: oHTX, old heart transplant; yHTx, young heart transplant.


