Abstract
tRNAs encoded on the mitochondrial DNA of Physarum polycephalum and Didymium nigripes require insertional editing for their maturation. Editing consists of the specific insertion of a single cytidine or uridine relative to the mitochondrial DNA sequence encoding the tRNA. Editing sites are at 14 different locations in nine tRNAs. Cytidine insertion sites can be located in any of the four stems of the tRNA cloverleaf and usually create a G · C base pair. Uridine insertions have been identified in the T loop of tRNALys from Didymium and tRNAGlu from Physarum. In both tRNAs, the insertion creates the GUUC sequence, which is converted to GTΨC (Ψ = pseudouridine) in most tRNAs. This type of tRNA editing is different from other, previously described types of tRNA editing and resembles the mRNA and rRNA editing in Physarum and Didymium. Analogous tRNAs in Physarum and Didymium have editing sites at different locations, indicating that editing sites have been lost, gained, or both since the divergence of Physarum and Didymium. Although cDNAs derived from single tRNAs are generally fully edited, cDNAs derived from unprocessed polycistronic tRNA precursors often lack some of the editing site insertions. This enrichment of partially edited sequences in unprocessed tRNAs may indicate that editing is required for tRNA processing or at least that RNA editing occurs as an early event in tRNA synthesis.
RNA editing has been shown to be an obligatory step in the gene expression of a number of organisms (26). The most extensively edited RNAs identified to date are produced in mitochondria or chloroplasts. In trypanosome mitochondria, uridines are inserted or deleted in mRNAs (28), and in the mitochondria of the acellular slime mold Physarum polycephalum, cytidines (and occasionally other nucleotides) are inserted in mRNA (6, 13, 20–22) or rRNAs (14). In mitochondria and chloroplasts of plants, some cytidine and uridine residues in RNA are converted by nucleotide modification to uridine and cytidine, respectively (15).
Nucleotide modification in tRNAs has been known for decades (3). Generally, this consists of posttranscriptional modification of existing nucleotides but in the case of the Q base modification may also include the exchange of bases between nucleotides by transglycosylation (23). Usually, conventional ribonucleotides are converted to unconventional ribonucleotides. In a few instances, a conventional nucleotide has been converted to one of the other conventional ribonucleotides, and this has been termed tRNA editing. At least five different types of tRNA editing have been identified, all in mitochondria. Cytidine-to-uridine conversion has been observed in mitochondrial tRNAPhe and tRNACys of angiosperms (2, 17) and mitochondrial tRNAHis of a gymnosperm (18). These editing sites are located in the acceptor or anticodon stems of the tRNA and create A · U base pairs. Another cytidine-to-uridine conversion has been identified in the anticodon of the tRNAAsp of marsupials (7). This conversion changes the 5′-GCC-3′ anticodon to 5′-GUC-3′, allowing the tRNA to recognize aspartic acid codons and to be acylated with aspartic acid (4).
In the mitochondria of the protist Acanthamoeba castellanii (5, 11, 12) and the fungus Spizellomyces punctatus (10), base substitutions occur in the first three nucleotides at the 5′ end of the tRNAs, creating conventional base pairs in the acceptor stem. Generally, G or A is substituted for the U, A, or C inferred from the mitochondrial DNA (mtDNA) sequence. In the mitochondria of snails (32), squid (29), and chickens (34), the 3′ ends of some tRNAs have base substitutions relative to the mtDNA sequence. These tRNAs are produced from the upstream gene of overlapping tRNA genes and have A substitutions. These tRNAs are thought to be truncated by 5′ processing of the downstream overlapping tRNA with subsequent oligo(A) addition to the 3′ end, which is terminated by CCA addition to produce mature tRNAs with 1 to 5 A · U bp at the end of the acceptor stem. A similar type of base substitution occurs in mitochondrial tRNASer of the platypus, in which 3 of the 4 nucleotides at the 3′ end of the tRNA are substituted relative to the mtDNA sequence (33). However, in this case the substituted nucleotides are not all adenosines.
Here we report the identification and characterization of insertional editing in tRNAs of P. polycephalum and Didymium nigripes. These tRNAs have nucleotide insertions of cytidine or uridine relative to the mtDNA genes encoding them. These insertions create classical base pairing or conserved nucleotides in the tRNAs that are not predicted from the DNA sequence. cDNAs corresponding to unprocessed polycistronic tRNAs generally lack many of the nucleotide insertions at the editing sites.
MATERIALS AND METHODS
Cell cultures and nucleic acid preparation.
P. polycephalum M3C was cultured and mitochondrial nucleic acids were prepared essentially as described by Mahendran et al. (14). D. nigripes (ATCC 28363) was cultured as macroplasmodia on half-strength cornmeal agar plates supplemented with approximately 1 g of oat flakes. Cultures were grown for 9 days at 27°C in the dark. D. nigripes was harvested by gently scraping the macroplasmodium from the surfaces of eight agar plates in the presence of 2 ml of diethylpyrocarbonate-treated BEST (0.1% bovine serum albumin, 60 mM EDTA, 300 mM sucrose, 20 mM Tris-HCl, pH 7.5) per plate. The plasmodium was pelleted by centrifugation at 8,000 × g for 10 min at 4°C, and the supernatant fluid was discarded. The pellets were resuspended in BEST containing 2% sodium dodecyl sulfate (SDS) and lysed with vortexing. A cleared lysate was produced by centrifugation at 8,000 × g for 10 min at 4°C. Lysis of the pellet was repeated, and the resulting cleared lysate was pooled with the initial cleared lysate. The pooled lysates were extracted three times with a 1:1 mixture of phenol-chloroform, and nucleic acids were precipitated with 0.5 M sodium acetate (pH 7.8) and ethanol overnight at −20°C.
Northern blot analysis.
Isolation of RNA from Physarum mitochondria was performed as detailed in the work of Mahendran et al. (14). Approximately 20 μg of mitochondrial RNA was separated in either 1.5 or 2% agarose–8 M urea–1× TPE (40 mM Tris-HCl [pH 7.8], 36 mM NaH2PO4, 1 mM EDTA) gels and transferred to diazobenzyloxymethyl-cellulose filters overnight by protocols from the work of Alwine et al. (1). The filters were cut into strips and prehybridized at 42°C for 14 h in 5× SSC (1× SSC is 0.15 M NaCl plus 0.015 M sodium citrate)–50% formamide–1× Denhardt’s buffer–50 mM sodium phosphate (pH 7.4)–0.1% SDS–25 mg of sheared calf thymus DNA per ml. Probes for hybridization were subclones into pUC18 (New England Biolabs) of restriction fragments of pΦm1234, a 10-kb XbaI clone of Physarum mtDNA (9). Probe 1 is a subclone containing a 650-bp HindIII/HindIII fragment which encodes the 5′ end of the mitochondrial large-subunit rRNA. Probe 2 is a 2.5-kb AvaI/HindIII fragment which contains the entire coding region for the mitochondrial small-subunit rRNA. Probe 3 is a 0.4-kb EcoRI/AvaI fragment which has sequences corresponding to three mitochondrial tRNAs (Met2, Lys, and Pro). The three probes were labeled separately by nick translation (Gibco BRL) in the presence of 50 μCi of [α-32P]dATP (ICN). In situ hybridization was performed in fresh hybridization solution at 42°C for 16 h with 3 × 106 cpm per filter strip. After hybridization, the filter strips were washed twice in 2× SSC–20 mM sodium phosphate buffer (pH 7.4)–0.1% SDS at 25°C and twice in 0.1× SSC–1 mM sodium phosphate buffer (pH 7.4)–0.005% SDS at 42°C. Hybridization patterns were visualized by autoradiography.
Oligonucleotide primers and primer extension.
The oligonucleotide primers used in the generation of DNA and cDNA amplification products are as follows: Phy-Met1-5′Eco, 5′-GGAATTCGTTCTATAGTTTAGTGG-3′; Phy-Met1-3′Xba, 5′GCTCTAGAGTAGAATCGAACTACCG-3′; Phy-Glu-5′Eco, 5′-GGAATTCGCCCCTTTCGTCTAGTGG-3′; Phy-Glu-3′Xba, 5′-GCTCTAGAGCCCCTTGAGAGAATC-3′; Phy-Lys-5′Eco, 5′-GGAATTCTGGCTCGTAGCATAAATAG-3′; Phy-Lys-3′Bam, 5′-CGGGATCCTTGGCTCCACAGGACTTGC-3′; Phy-Pro-5′Eco, 5′-GGAATTCTCGGAGCGTAGCT-3′; Phy-Pro-3′Bam, 5′-CGGGATCCAAGATTTGAACTCGC-3′; Phy-Pro-DSBam, 5′-CGGGATCCTAAAACCTAGAGTTCTG-3′; Did-Lys-3′Bam, 5′-CGGGATCCATGGCACCATGGG-3′; Did-Pro-5′Eco, 5′-GGAATTCACGGGGGTAGCTC-3′; Did-Lys-5′Eco, 5′-GGAATTCTGGTACGTAGCATAACTGG-3′; Did-Met2-5′Eco, 5′-GGAATTCGGGTTGGTGTAATTGG-3′; Did-Met2-3′Bam, 5′-CGGGATCCTGGTGGGGGGTTT-3′; SSU-3′Eco, 5′-GGAATTCGAAYCTGYKGYTGGA-3′ (Y = C + T; K = T + G).
The primers designated with the prefix Phy- were synthesized with an ABI 381A DNA synthesizer. All other primers were commercially synthesized (Gibco BRL). Nucleic acids isolated from either Physarum or Didymium were treated with 2 U of RNase-free RQ1 DNase I (Promega) in 40 mM Tris-HCl (pH 7.9)–10 mM NaCl–6 mM MgCl2–10 mM CaCl2 in the presence of 1 U of Inhibit-ACE (5Prime-3Prime, Inc.) for 30 min at 37°C. The RNA was recovered through three extractions with a 1:1 mixture of phenol-chloroform and precipitated with 0.5 M sodium acetate (pH 7.8) and ethanol at −20°C for 15 min. Approximately 15 μg of RNA was hybridized with 1 μg of a specific oligonucleotide primer in 250 mM KCl–10 mM Tris-HCl (pH 8.3), heated to 80°C, and slowly cooled to 45°C over the course of 30 min. After annealing, the primer-RNA hybrid was precipitated with 0.5 M sodium acetate and ethanol. The RNA-primer pellet was resuspended in 50 mM Tris-HCl (pH 8.3)–40 mM KCl–10 mM dithiothreitol–7 mM MgCl2–0.1 mg of bovine serum albumin per ml–25 mM deoxynucleoside triphosphates–1 U of Inhibit-ACE to a final volume of 50 μl. The primer was extended with 200 U of Moloney murine leukemia virus reverse transcriptase (Promega) for 45 min at 37°C, and the RNA was removed by treatment with DNase-free RNase A (Sigma) for 15 min at 37°C. cDNAs were recovered by three phenol-chloroform extractions and precipitated with ethanol in 2.5 M ammonium acetate.
PCR amplification, cloning, and sequence analysis.
Double-stranded cDNAs were produced by the method of Saiki et al. (25). The amplification was performed in Taq buffer (50 mM KCl, 10 mM Tris-HCl [pH 8.3], 2.5 mM MgCl2, 25 μM deoxynucleoside triphosphates, 0.1% [wt/vol] gelatin) with 1 μg of the appropriate 5′-end primer and 2.5 U of Taq DNA polymerase (Fisher Biotech). The thermal cycle regimen consisted of 94°C for 1 min, 55°C for 2 min, and 72°C for 3 min for 30 cycles. During the final cycle, an extension of 10 min at 72°C was performed and the reaction products were slowly cooled to 4°C for complete annealing. The resulting PCR amplification products were cloned into pUC18 (New England Biolabs) with the appropriate restriction enzymes for the recognition site added to a given oligonucleotide primer tail and ligated with pUC18 in 30 mM Tris-HCl (pH 7.8)–10 mM dithiothreitol–10 mM MgCl2–10 mM ATP with 3 U of T4 DNA ligase (Promega) at 16°C for 18 h. The ligase reaction was precipitated with 0.5 M sodium acetate and ethanol. An aliquot was used to transform Escherichia coli (XL1-Blue; Stratagene), and recombinant DNA clones were identified and isolated by conventional methods. Clones of interest were subjected to sequence analysis by typical dideoxynucleotide sequencing methods and reagents supplied with the Sequenase kit (Amersham).
Nucleotide sequence accession numbers.
The mtDNA sequences of the tRNA genes and their flanking sequences from P. polycephalum and D. nigripes which have been annotated with editing site locations have been submitted to GenBank under accession no. AF059032, AF059033, and AF059034.
RESULTS
tRNA editing in P. polycephalum.
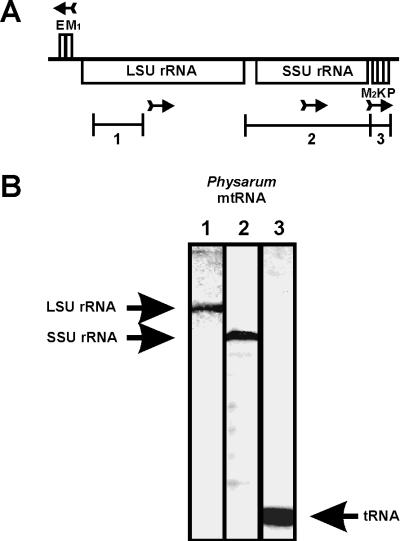
During sequence analysis of the rRNA-encoding region of the mtDNA of P. polycephalum, sequences which could potentially encode tRNAs were identified. These tRNAs were tentatively identified as tRNAGlu, tRNA1Met, tRNA2Met, tRNALys, and tRNAPro on the basis of their potential anticodons, assuming a classical genetic code and classical codon-anticodon interactions. The potential genes for tRNAGlu and tRNA1Met were located upstream of the gene for the large-subunit rRNA, were separated by 5 bp, and were encoded by the strand opposite to that encoding the rRNAs. The genes for tRNA2Met, tRNALys, and tRNAPro were located downstream of the small-subunit rRNA, were separated by 19 and 18 bp, respectively, and were encoded from the same strand as the rRNAs (Fig. 1A).
FIG. 1.
(A) Location on the mtDNA of tRNA genes relative to the large (LSU)- and small (SSU)-subunit rRNA genes. tRNA genes are designated by the single-letter code for their cognate amino acid. Arrows indicate the direction of transcription of these genes. The lines labeled 1, 2, and 3 indicate the location and extent of hybridization probes used in Northern blots. (B) Northern blot of Physarum mitochondrial RNA (mtRNA). Mitochondrial RNA was separated by agarose-urea gel electrophoresis and transferred in situ to diazobenzyloxymethyl-paper. Equivalent strips were hybridized with probes 1, 2, and 3 (A). The detected RNAs are the mitochondrial large-subunit rRNA (lane 1), the mitochondrial small-subunit rRNA (lane 2), and tRNAs (lane 3).
To determine if these sequences were transcribed as tRNA-sized RNAs, Northern blots of mitochondrial RNA were probed with mtDNA sequences specific for the tRNA sequences. tRNA-sized transcripts were detected by using a probe for tRNA2Met, tRNALys, and tRNAPro (Fig. 1B) and a probe for tRNA1Met and tRNAGlu (data not shown). Fragments from other regions of the mtDNA did not detect tRNA-sized transcripts (data not shown).
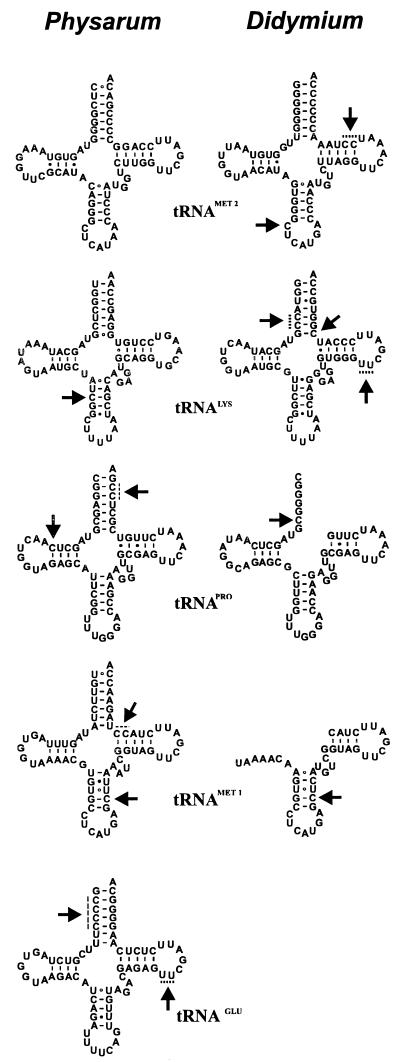
The secondary structure inferred from the mtDNA sequences was consistent with the classical cloverleaf structure associated with tRNAs. However, certain conserved features associated with most tRNAs were missing in four of the five sequences (Fig. 2). In tRNAGlu, the highly conserved GTTC motif was absent and base-pairing opportunities were absent in the acceptor stem of tRNAPro and tRNAGlu, in the dihydrouridine (DHU) stem of tRNAPro, in the anticodon stem of tRNA1Met and tRNALys, and in the GTΨC stem of tRNA1Met.
FIG. 2.
Secondary structure of tRNAs deduced from the sequence of cDNAs. Cloverleaf structures of analogous mitochondrial tRNAs from Physarum and Didymium are compared. Analogous tRNA pairs are labeled with their cognate amino acid based on the tRNA anticodon. Nucleotides inserted in the tRNAs relative to their mtDNA sequence are indicated by arrows. Locations in which the exact site of the insertion is ambiguous due to cytidine insertions next to encoded cytidines or uridine insertions next to encoded uridines are indicated by dotted lines. Nucleotides in Watson-Crick base pairs are connected by a dash, nucleotides in GU wobble base pairs are connected by a closed dot, and nucleotides in other base pairs are connected with an open dot. Some of the sequences for the cloverleaf structures for tRNA1Met and tRNAPro from Didymium are not shown since they were derived from the primer and not from the mtDNA or tRNA template.
In order to determine the sequence of the tRNAs produced from the mtDNA, primers complementary to the 3′ ends of the tRNA were synthesized and were extended on an RNA template with reverse transcriptase. cDNAs produced from the tRNAs were amplified by PCR with the primer used for cDNA synthesis and a primer with a sequence analogous to the 5′ end of the tRNA. Primer pairs were used to amplify cDNAs corresponding to tRNA1Met, tRNAGlu, tRNALys, and tRNAPro. RNA-specific amplification products produced in this way were cloned in the plasmid pUC18. The inserts of plasmids corresponding to two or more independent clones of each amplification product were sequenced and compared to the mtDNA sequence. Differences between cDNA and mtDNA sequences corresponding to RNA editing sites were identified in each case (Fig. 2). Both tRNA1Met and tRNALys had a single cytidine insertion in the anticodon stem, creating a G · C base pair. tRNAPro had a single cytidine insertion in the DHU stem, which also created a G · C base pair. tRNAGlu had a single uridine insertion in the GTΨC loop, creating the highly conserved GUUC motif that is modified to GTΨC in most tRNAs.
Because primers corresponding to the 5′ and 3′ ends of the tRNAs were used for amplification of cDNAs, editing sites at the ends of the tRNAs could not be detected by this approach. To identify editing sites at the ends of tRNAs, primer pairs which would selectively amplify unprocessed tRNA precursors were chosen for cDNA amplification. The Met1-5′ primer was used with the Glu-3′ primer to amplify the unprocessed dicistronic tRNA precursor for tRNA1Met-tRNAGlu, and the Lys-5′ primer was used with a primer (Pro-DS) located downstream of the tRNAPro gene to amplify the unprocessed dicistronic tRNA precursor for tRNALys-tRNAPro. cDNAs amplified by using these primer pairs were cloned, sequenced, and compared with the mtDNA sequence. This approach revealed that tRNAGlu, tRNA1Met, and tRNAPro each had an additional editing site. tRNAGlu had a cytidine insertion in the acceptor stem next to three other cytidines, producing a total of four consecutive C · G base pairs in the acceptor stem. tRNA1Met had an additional cytidine insertion creating a G · C base pair in the GTΨC stem, and tRNAPro had an additional cytidine insertion creating a G · C base pair in the DHU stem.
In all, seven editing sites were identified in the four tRNAs examined, four sites in tRNA1Met-tRNAGlu and three sites in tRNALys-tRNAPro. The distance between the sites in these two groups averaged 43.4 nucleotides, about the same as the average distance between editing sites in the mitochondrial small-subunit rRNA (14). As with the rRNA editing sites, the distance ranged between 74 and 17 nucleotides, the latter being the approximate distance between editing sites in mRNA (6, 13, 20–22).
In contrast to the amplification products corresponding to a single tRNA, which in every case had a nucleotide inserted at a potential editing site, cDNAs corresponding to unprocessed dicistronic tRNA precursors lacked insertions at potential editing sites at a high frequency. Analysis of 30 independent clones of tRNA1Met-tRNAGlu indicated that the majority of the amplification products were unedited at all four potential editing sites. However, 7 of the 15 possible patterns of insertions at editing sites were observed (Table 1). These patterns were inconsistent with a single linear order of nucleotide insertions at editing sites.
TABLE 1.
Patterns of nucleotide insertions at editing sites in dicistronic RNAs from Physarum mitochondria containing tRNA1Met and tRNAGlu
| Pattern type | Insertion at editing sitea
|
|||
|---|---|---|---|---|
| tRNA1Met
|
tRNAGlu
|
|||
| 1 | 2 | 1 | 2 | |
| 1 | C | C | C | U |
| 2 | C | C | C | — |
| 3 | C | — | C | U |
| 4 | C | — | C | — |
| 5 | C | — | — | U |
| 6 | C | — | — | — |
| 7 | — | — | — | U |
| 8 | — | — | — | — |
C or U, cytidine or uridine insertion, respectively, at editing site; —, no nucleotide insertion at editing site. Editing sites for a given tRNA are numbered 5′ to 3′.
Likewise, cDNAs corresponding to partially edited RNAs were observed when tRNALys-tRNAPro amplification products were selected. Again, many (9 of 20) of these amplification products lacked insertions at all three potential editing sites. Five of the seven possible patterns of insertions in the three potential editing sites were observed (Table 2), and these patterns were also inconsistent with a single linear order of nucleotide insertion at editing sites.
TABLE 2.
Patterns of nucleotide insertions at editing sites in dicistronic RNAs containing tRNALys and tRNAPro
| Pattern type | Insertion at editing sitea
|
||
|---|---|---|---|
| tRNALys | tRNAPro
|
||
| 1 | 2 | ||
| 1 | C | C | C |
| 2 | C | C | — |
| 3 | — | C | C |
| 4 | — | C | — |
| 5 | — | — | C |
| 6 | — | — | — |
C, cytidine insertion at editing site; —, no insertion at editing site. Editing sites for a given tRNA are numbered 5′ to 3′.
tRNA editing in D. nigripes.
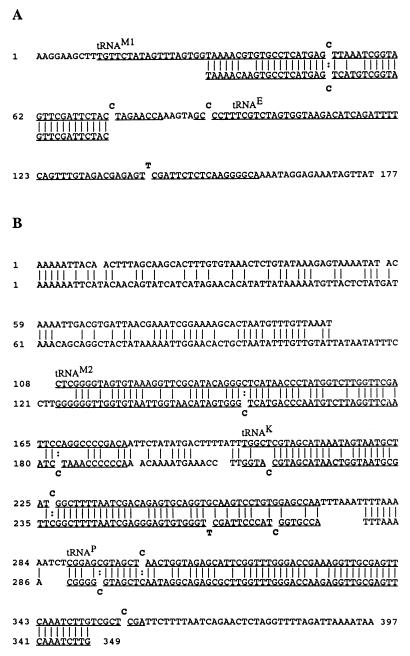
D. nigripes is a member of the Myxomycota and is classified in the same order (Physarales) as Physarum but in a different family (Didymiaceae). Comparison of the sequences of the nuclear large-subunit rRNAs of Physarum and Didymium indicates that they are related but diverged about 400 to 500 million years ago (8). We wished to determine whether the organization of the rRNA and tRNA genes is conserved on the mtDNA of Didymium, whether the tRNAs would require editing for their maturation, and, if editing was required, whether the sites of editing would be conserved in the analogous tRNAs. Since the sequence and organization of the mtDNA of D. nigripes are not known, primers used to amplify portions of the Physarum mtDNA were used in an attempt to amplify D. nigripes mtDNA. A 386-bp amplification product was produced from D. nigripes DNA by using the 3′ primer for Physarum tRNAPro (Pro-3′) and a primer which hybridizes to a conserved portion of the 3′ end of the small-subunit rRNA gene of Physarum (SSU-3′). This amplification product was cloned in plasmid pUC18 and sequenced. Its sequence revealed that three tRNA genes analogous to those in Physarum mtDNA were also located in the same order downstream of the small-subunit rRNA gene. Although the anticodon and the potential for a general cloverleaf structure were conserved, a number of differences were observed between the primary sequences of the tRNA genes (Fig. 2). The intergenic region between tRNAs and the rRNA was even less conserved (Fig. 3). The primers for the tRNA1Met gene of Physarum produced an amplification product from D. nigripes DNA, indicating that the analogous gene was present in Didymium. Primers for the tRNAGlu gene did not produce amplification products from Didymium DNA.
FIG. 3.
Alignment of the regions of the mtDNA from Physarum and Didymium that encode analogous tRNAs. Vertical bars connect identical nucleotides in the alignment. Sequences encoding mature tRNAs are underlined and labeled with the single-letter code for their cognate amino acid. C’s or T’s displaced from the sequence indicate the sites of insertions of cytidines or thymidines, respectively, in the cDNAs of the tRNAs. The top line is the sequence from Physarum mtDNA; the bottom line is the analogous region of Didymium mtDNA. (A) Sequences flanking the tRNA1Met gene. (B) Sequences downstream of the small-subunit rRNA gene.
To determine whether RNA editing was necessary for the maturation of these tRNAs, cDNAs were synthesized on D. nigripes RNA templates and amplified by PCR. New primers (Did-Met2-5′, Did-Met2-3′, Did-Lys-5′, Did-Lys-3′, and Did-Pro-5′) specific for D. nigripes were synthesized based on the sequence of the 386-bp Didymium amplification product and used in the amplification. cDNA amplification products were produced from all primer combinations except those which included the SSU-3′ primers, indicating that either these tRNAs are not cotranscribed with the rRNA or that rRNA-tRNA processing is very rapid.
Sequence analysis of these cDNA amplification products revealed seven editing sites in the mitochondrial tRNAs of Didymium. While the insertions were single cytidines and uridines as in Physarum, with one exception, the location of these sites differed from their location in the analogous genes of Physarum. All of the editing sites in Didymium corresponded to existing nucleotides encoded by the mtDNA in Physarum, and editing sites in Physarum corresponded to existing nucleotides in the mtDNA sequence of D. nigripes (Fig. 2). Two sites in tRNALys and one site in tRNAPro were cytidine insertions in the acceptor stems. In the case of tRNALys, these insertions created G · C base pairs; in the case of tRNAPro, base pairing could not be determined since the potential G nucleotide was covered by the amplification primer. Another C insertion created a G · C base pair in the GTΨC arm of tRNA2Met. A second cytidine insertion was located in the anticodon loop of tRNA2Met, and a uridine insertion was located in the GTΨC loop of tRNALys, creating the conserved 5′-GUUC-3′ motif. The tRNA1Met in D. nigripes had a single cytidine insertion at the site analogous to the position of the cytidine insertion in Physarum tRNA1Met although the sequences of the tRNAs differed at other positions.
The spacing of editing sites in cDNAs corresponding to the unprocessed tRNA2Met-tRNALys-tRNAPro precursor was similar to that found for mRNA editing in Physarum. The average spacing between editing sites was about 27 nucleotides with a standard deviation of about 14 nucleotides. As with Physarum, cDNAs corresponding to unprocessed, polycistronic tRNA precursors lacked insertions at some of the editing sites which invariably had insertions in single tRNA amplification products. The seven different patterns of insertion at editing sites for seven dicistronic and four tricistronic transcripts are shown in Table 3. In contrast to Physarum, none of the polycistronic cDNAs from Didymium lacked insertions at all of the editing sites. As with Physarum, no linear order of insertions was consistent with the insertion pattern of all of the cDNAs.
TABLE 3.
Patterns of nucleotide insertions at editing sites in polycistronic RNAs from Didymium mitochondria containing tRNA2Met, tRNALys, or tRNAPro
| Pattern type | Insertion at editing sitea
|
|||||
|---|---|---|---|---|---|---|
| tRNA2Met
|
tRNALys
|
tRNAPro | ||||
| 1 | 2 | 1 | 2 | 3 | ||
| 1 | C | C | C | U | C | C |
| 2 | — | — | C | U | C | C |
| 3 | — | — | C | — | C | C |
| 4 | U | C | C | |||
| 5 | — | C | C | |||
| 6 | — | C | — | |||
| 7 | U | — | — | |||
C or U, cytidine or uridine insertion, respectively, at editing site; —, no nucleotide insertion at editing site. Editing sites for a given tRNA are numbered from 5′ to 3′.
DISCUSSION
We have identified 14 insertional editing sites in nine tRNAs of P. polycephalum and D. nigripes. These insertions create features in the tRNAs that increase their similarity to nearly all other tRNAs, i.e., the proper number of base pairs in the stems of the cloverleaf secondary structure and conserved motifs such as the GUUC sequence in the GTΨC loop and the conserved pyrimidine at position 32 of the anticodon loop. After editing, these tRNAs have most of the features of the conventional tRNA cloverleaf, although some unusual features still exist.
One unique feature of all of the tRNAs except tRNAPro and tRNA1Met of Physarum is the potential for a non-Watson-Crick base pair at the top of the anticodon stem (position 27:43). Eight different base pairs exist in this position in the nine tRNAs. While tRNAPro and tRNA1Met of Physarum have a U · A base pair and tRNAPro of Didymium has a C · G base pair at this position, the other tRNAs have non-Watson-Crick base pairings: U · G in tRNALys of Didymium, A · C in tRNALys of Physarum, C · A in tRNA2Met of Physarum, G · A in tRNA2Met of Didymium, A · A in tRNA1Met of Didymium, and U · U in tRNAGlu of Physarum. Another unusual feature of tRNA1Met of both organisms is the potential for a U · U base pair at the central base pair (position 29:41) of the anticodon stem. This base alignment has also been observed in the anticodon stem of tRNAIle and tRNACys of Oenothera mitochondria (2). Also, the conserved A at position 21 is a C in tRNA2Met of Physarum, and the conserved purine at position 9 is a cytidine in tRNAGlu of Physarum. The first base pair of the acceptor stem (position 1:72) of tRNA1Met (C · C) and tRNA2Met (U · C) of Physarum is not a Watson-Crick base pair, but there is a precedent for this in the initiator tRNAMet of some prokaryotes and organelles (27). A G · G pairing is located at position 7:66 at the base of the acceptor stem of tRNALys in Physarum. This potential base pairing is predicted from the sequence of the mtDNA in Didymium as well but is corrected by the insertion of two cytidines in the acceptor stem of the tRNA.
The tRNA editing of Physarum and Didymium mitochondria differs in its features from any other tRNA editing described to date. It is the first example of single nucleotide insertions in tRNA relative to the mtDNA sequence. Although it differs from other tRNA editing, it has many of the features associated with the editing identified in mRNA and rRNA of P. polycephalum (6, 13, 14, 20–22). Most of the editing sites (12 of 14) have a single cytidine insertion, while a few (2 of 14) have a single uridine insertion. As with rRNA and mRNA, an unusually large number of cytidine insertions (6 of 12) could follow purine-pyrimidine dinucleotides. (Ambiguity arises from the fact that some cytidine insertions are next to a cytidine encoded by mtDNA.) However, four cytidine insertions follow purine-purine dinucleotides and two follow pyrimidine-pyrimidine dinucleotides. Also, the spacing of the insertions is similar to that observed in rRNA and mRNA of Physarum. Overall, the average spacing between editing sites in unprocessed tRNA precursors is about 35 nucleotides and ranges from 10 to 74 nucleotides. As with mRNAs and rRNAs from Physarum, no editing site is closer than 9 nucleotides.
The editing appears to be both very accurate and efficient. Insertion of the wrong nucleotide at a site or insertion of nucleotides at an incorrect site has not been observed. Additionally, greater than 95% of the potential editing sites in the total mitochondrial RNA population have a nucleotide insertion (6, 20–22, 24). This is consistent with the observation that 100% of the single tRNA cDNA amplification products had a nucleotide inserted at editing sites. In contrast, cDNA amplification products derived from unprocessed tRNAs have a relatively large number of potential editing sites that lack nucleotide insertions. This implies that editing and 5′ and 3′ tRNA processing may be associated.
In Physarum, it is not known precisely when, relative to transcription, RNA editing occurs. Visomirski-Robic and Gott (30, 31) have shown that, in isolated mitochondria provided with limiting concentrations of one exogenous nucleotide, editing can occur in nascent RNAs at sites very close to the mitochondrial transcription complex. This is consistent with our results that RNA editing is associated with tRNA processing, which probably occurs on nascent transcripts concurrent with transcription. Although it is clear that under some conditions editing can be closely associated with transcription, it is still not clear whether editing is cotranscriptional, in the sense that the nontemplated nucleotides are added to the 3′ end of the nascent RNA, or posttranscriptional, in the sense that the nontemplated nucleotides are inserted between adjacent nucleotides in the RNA.
If RNA editing in Physarum and Didymium is cotranscriptional, then the cDNAs that lack insertions at editing sites must be derived from aberrant RNAs that have not been edited at some sites. Visomirski-Robic and Gott (31) have shown that RNAs with missing cytidines at editing sites are produced in isolated mitochondria when CTP concentrations are low. The fact that unprocessed tRNAs are enriched in potential editing sites that do not have insertions would then indicate that editing of tRNAs may be a prerequisite for tRNA processing. Modificational editing of tRNAs in plant mitochondria has been shown to be required for proper tRNA processing (2, 16, 18, 19). However, if partially edited sequences are enriched in unprocessed RNAs because partially edited RNAs cannot be processed, then one would not expect fully edited versions of the cDNAs to also be enriched in these populations. tRNA processing must be rapid and efficient since unprocessed forms cannot be detected in Northern blots of mitochondrial RNA (Fig. 1B), and so fully edited forms would be expected to be rapidly processed. In contrast to this expectation, fully edited sequences (pattern type 1 in Tables 1, 2, and 3) were observed for each of the three unprocessed polycistronic transcripts selected. Another inconsistency with the idea that the enrichment of partially edited cDNAs is derived from aberrantly edited RNAs which cannot be processed is the occurrence of multiple unedited sites in the unprocessed tRNA population. Under normal growth conditions, the frequency of editing sites without insertions is at most 5% (6, 20–22, 24). If insertions at editing sites are independent of one another, this would predict that the probability of multiple editing site errors within a single transcript would be very rare. However, the majority of cDNAs corresponding to partially edited tRNAs are missing nucleotides at multiple potential editing sites.
If RNA editing in Physarum and Didymium is posttranscriptional, then the unprocessed, partially edited transcripts may be not the result of editing errors but the result of incomplete editing of tRNA precursors. In this case, partially edited sequences would be enriched in unprocessed tRNAs because RNA editing and tRNA processing are concurrent, so that unedited sites which are rare or absent in mature tRNAs are present in tRNAs which have not yet been processed. The low level of unedited sites in the RNA population then reflects the rate of RNA editing and not its error frequency. Whether the enrichment of unedited sites in cDNAs derived from unprocessed tRNAs implies that editing is concurrent with processing or that editing is required for processing, it is likely that RNA editing is an early event in tRNA expression in Physarum and Didymium mitochondria.
Comparison of the editing in the tRNAs of Didymium and Physarum indicates that both organisms have the same type of tRNA editing. This implies that this form of RNA editing is at least as ancient as the divergence of Physarum and Didymium (400 to 500 million years ago [8]) and that this type of RNA editing is likely to be distributed throughout the Myxomycota. Although the type of tRNA editing seems to be conserved between Physarum and Didymium, the location of the editing sites in the tRNAs of these two organisms is, with only one exception, different even though tRNA gene location and order as well as tRNA structure and sequence are relatively conserved (75% nucleotide identity). This is the first example of evolutionary variation in editing site location in the Myxomycota. This variation could be the result of the loss of different editing sites in Physarum and Didymium since their divergence from a common ancestor with a higher density of editing sites. Conversely, the variability of editing site location could result from the establishment of different sites after the divergence of these organisms. A third possibility is that sites have been both lost and gained in a coordinated way so as to maintain editing site density. In any case, it is clear that editing sites are not fixed or static but can be lost or gained over time and that the rate of editing site variation can exceed the rate of sequence divergence in mtDNA.
How editing sites are specified is still unknown. It could be that base pairing in the stems of the tRNA acts as the template for editing, as has been proposed by Lonergan and Gray (11, 12) for 5′ tRNA editing in A. castellanii and by Laforest et al. (10) for 5′ tRNA editing in S. punctatus. However, this would not explain how editing sites in loop regions of tRNAs are indicated, and the variation of editing site location between Physarum and Didymium argues that location is not specified by an intrinsic property of the tRNA since tRNA structure and sequence are conserved relative to editing site location. Further comparisons will be necessary to determine if the insertional pattern of editing or RNA editing itself is conserved among the other members of the Myxomycota.
ACKNOWLEDGMENT
This research was supported in part by a research grant to D.M. from the National Institute of General Medical Sciences (GM44223), National Institutes of Health.
REFERENCES
- 1.Alwine J, Kemp D, Stark G. Method for the detection of specific RNAs in agarose gels by transfer to diazobenzyloxymethyl paper and hybridization with DNA probes. Proc Natl Acad Sci USA. 1977;74:5350–5354. doi: 10.1073/pnas.74.12.5350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Binder S, Marchfelder A, Brennicke A. RNA editing of tRNAPhe and tRNACys in mitochondria of Oenothera berteriana is initiated in precursor molecules. Mol Gen Genet. 1994;244:67–74. doi: 10.1007/BF00280188. [DOI] [PubMed] [Google Scholar]
- 3.Björk G R, Ericson J U, Gustafson C E D, Hagervall T G, Jönsson Y H, Wikström P M. Transfer RNA modification. Annu Rev Biochem. 1987;56:263–287. doi: 10.1146/annurev.bi.56.070187.001403. [DOI] [PubMed] [Google Scholar]
- 4.Borner G V, Morl M, Janke A, Pääbo S. RNA editing changes the identity of a mitochondrial tRNA in marsupials. EMBO J. 1996;15:5949–5957. [PMC free article] [PubMed] [Google Scholar]
- 5.Burger G, Plante I, Lonergan K, Gray M W. The mitochondrial DNA of the amoeboid protozoon Acanthamoeba castellanii: complete sequence, gene content and genome organization. J Mol Biol. 1995;245:522–537. doi: 10.1006/jmbi.1994.0043. [DOI] [PubMed] [Google Scholar]
- 6.Gott J M, Visomirski L M, Hunter J L. Substitutional and insertional RNA editing of the cytochrome c oxidase subunit I mRNA of Physarum polycephalum. J Biol Chem. 1993;268:25483–25486. [PubMed] [Google Scholar]
- 7.Janke A, Pääbo S. Editing of a tRNA anticodon in marsupial mitochondria changes its codon recognition. Nucleic Acids Res. 1993;21:1523–1525. doi: 10.1093/nar/21.7.1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johansen S, Johansen T, Haugli F. Extrachromosomal ribosomal DNA of Didymium iridis: sequence analysis of the large subunit ribosomal RNA gene and sub-telomeric region. Curr Genet. 1992;22:305–312. doi: 10.1007/BF00317926. [DOI] [PubMed] [Google Scholar]
- 9.Jones E P, Mahendran R, Spottswood M R, Yang Y, Miller D L. Mitochondrial DNA of Physarum polycephalum: physical mapping, cloning and transcription mapping. Curr Genet. 1990;17:331–337. doi: 10.1007/BF00314881. [DOI] [PubMed] [Google Scholar]
- 10.Laforest M J, Roewer I, Lang B F. Mitochondrial tRNAs in the lower fungus Spizellomyces punctatus: tRNA editing and UAG ‘stop’ codons recognized as leucine. Nucleic Acids Res. 1997;25:626–632. doi: 10.1093/nar/25.3.626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lonergan K, Gray M W. Editing of transfer RNAs in Acanthamoeba castellanii mitochondria. Science. 1993;259:812–816. doi: 10.1126/science.8430334. [DOI] [PubMed] [Google Scholar]
- 12.Lonergan K, Gray M W. Predicted editing of additional transfer RNAs in Acanthamoeba castellanii mitochondria. Nucleic Acids Res. 1993;21:4402. doi: 10.1093/nar/21.18.4402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mahendran R, Spottswood M R, Miller D L. RNA editing by cytidine insertion in mitochondria of Physarum polycephalum. Nature. 1991;349:434–438. doi: 10.1038/349434a0. [DOI] [PubMed] [Google Scholar]
- 14.Mahendran R, Spottswood M, Ghate A, Ling M, Jeng K, Miller D L. Editing of the mitochondrial small subunit rRNA in Physarum polycephalum. EMBO J. 1994;13:232–240. doi: 10.1002/j.1460-2075.1994.tb06253.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maier R M, Zeltz P, Kossel H, Bonnard G, Gualberto J M, Grienenberger J M. RNA editing in plant mitochondria and chloroplasts. Plant Mol Biol. 1996;32:343–365. doi: 10.1007/BF00039390. [DOI] [PubMed] [Google Scholar]
- 16.Marchfelder A, Brennicke A, Binder S. RNA editing is required for efficient excision of tRNAPhe from precursors in plant mitochondria. J Biol Chem. 1996;271:1898–1903. doi: 10.1074/jbc.271.4.1898. [DOI] [PubMed] [Google Scholar]
- 17.Maréchal-Drouard L, Ramamonjisoa D, Cosset A, Weil J H, Dietrich A. Editing corrects mispairing in the acceptor stem of bean and potato mitochondrial phenylalanine transfer RNAs. Nucleic Acids Res. 1993;21:4909–4914. doi: 10.1093/nar/21.21.4909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Maréchal-Drouard L, Kumar R, Remacle C, Small I. RNA editing of larch mitochondrial tRNAHis precursors is a prerequisite for processing. Nucleic Acids Res. 1996;24:3229–3234. doi: 10.1093/nar/24.16.3229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Maréchal-Drouard L, Cosset A, Remacle C, Ramamonjisoa D, Dietrich A. A single editing event is a prerequisite for efficient processing of potato mitochondrial phenylalanine tRNA. Mol Cell Biol. 1996;16:3504–3510. doi: 10.1128/mcb.16.7.3504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Miller D, Mahendran R, Spottswood M, Ling M, Wang S, Yang N, Costandy H. RNA editing in mitochondria of Physarum polycephalum. In: Benne R, editor. RNA editing: the alteration of protein coding sequences of RNA. London, United Kingdom: Ellis Horwood; 1993. pp. 87–103. [Google Scholar]
- 21.Miller D, Ling M, Wang S, Yang N, Costandy H. RNA editing in mitochondria of Physarum polycephalum. In: Brennicke A, Kück U, editors. Plant mitochondria. Weinheim, Germany: VCH Press; 1993. pp. 53–63. [Google Scholar]
- 22.Miller D, Mahendran R, Spottswood M, Costandy H, Wang S, Ling M, Yang N. Insertional editing in mitochondria of Physarum. Semin Cell Biol. 1993;4:261–266. doi: 10.1006/scel.1993.1031. [DOI] [PubMed] [Google Scholar]
- 23.Okada N, Nishimura S. Isolation and characterization of a guanine enzyme, a specific tRNA transglycosylase, from E. coli. J Biol Chem. 1979;254:3061–3066. [PubMed] [Google Scholar]
- 24.Rundquist B A, Gott J M. RNA editing of the coI mRNA throughout the life cycle of Physarum polycephalum. Mol Gen Genet. 1995;247:306–311. doi: 10.1007/BF00293198. [DOI] [PubMed] [Google Scholar]
- 25.Saiki R K, Gelfand D H, Stoffel S, Scharf S J, Higuchi R, Horn G T, Mullis K B, Erlich H A. Primer-directed enzymatic amplifications of DNA with a thermostable DNA polymerase. Science. 1988;239:487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- 26.Smith H C, Gott J M, Hanson M R. A guide to RNA editing. RNA. 1997;3:1105–1123. [PMC free article] [PubMed] [Google Scholar]
- 27.Sprinzl M, Dank N, Nock S, Schön A. Compilation of tRNA sequences and of tRNA genes. Nucleic Acids Res. 1991;19:2127–2171. doi: 10.1093/nar/19.suppl.2127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Stuart K, Allen T E, Heidmann S, Seiwert S D. RNA editing of kinetoplastid protozoa. Microbiol Mol Biol Rev. 1997;61:105–120. doi: 10.1128/mmbr.61.1.105-120.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tomita K, Ueda T, Watanabe K. RNA editing in the acceptor stem of squid mitochondrial tRNATyr. Nucleic Acids Res. 1996;24:4987–4991. doi: 10.1093/nar/24.24.4987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Visomirski-Robic L M, Gott J M. Insertional editing of nascent mitochondrial RNAs in Physarum. Proc Natl Acad Sci USA. 1997;94:4324–4329. doi: 10.1073/pnas.94.9.4324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Visomirski-Robic L M, Gott J M. Insertional editing in isolated Physarum mitochondria is linked to RNA synthesis. RNA. 1997;3:821–827. [PMC free article] [PubMed] [Google Scholar]
- 32.Yokobori S, Pääbo S. Transfer RNA editing in land snail mitochondria. Proc Natl Acad Sci USA. 1995;92:10432–10435. doi: 10.1073/pnas.92.22.10432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yokobori S, Pääbo S. tRNA editing in metazoans. Nature. 1995;377:490. doi: 10.1038/377490a0. [DOI] [PubMed] [Google Scholar]
- 34.Yokobori S, Pääbo S. Polyadenylation creates the discriminator nucleotide of chicken mitochondrial tRNATyr. J Mol Biol. 1997;265:95–96. doi: 10.1006/jmbi.1996.0728. [DOI] [PubMed] [Google Scholar]