Abstract
INTRODUCTION
Virtual reality (VR) is an advanced technology that creates simulated environments and conditions. By offering the possibility of combining motor, cognitive, and well-being in conjunction with the potential to manipulate multi-sensorial features in a safe environment, VR has emerged as a promising powerful rehabilitation tool. Among advanced VR systems, various authors have highlighted promising effects in the rehabilitation of the computer-assisted rehabilitation environment (CAREN - Motekforce Link; Amsterdam, The Netherlands). In our scoping review, we aimed to map the existing evidence on the use of CAREN in the rehabilitation of neurological patients.
EVIDENCE ACQUISITION
This scoping review was conducted following the PRISMA guidelines. A search was carried out for all peer-reviewed articles published until June 30, 2023, using the following databases: PubMed, Embase, Cochrane Database, PeDro and Web of Science. The following terms have been used: (“Cognitive Rehabilitation” OR “Motor Rehabilitation” OR “CAREN” or “Computer-Assisted Rehabilitation Environment”) AND (“Virtual Reality” OR “Rehab”).
EVIDENCE SYNTHESIS
From the assessed studies, only seven met the inclusion criteria: 1) one study concerned cognitive rehabilitation in patients suffering from Parkinson’s Disease (PD); 2) one was on the usability of CAREN in PD patients; 3) two studies related to the influence of emotional components to CAREN rehabilitation; 4) three studies were related to motor rehabilitation using CAREN, and involved individuals with PD, Multiple Sclerosis, TBI, respectively. Generally, the few assessed studies demonstrate that CAREN is a safe and potentially effective tool to treat different symptoms (including gait and vestibular disturbances, executive function, depressive mood, and anxiety) in patients with different neurological disorders.
CONCLUSIONS
The reviewed literature indicated the potential use of CAREN in improving motor and cognitive skills with conflicting results on emotional aspects. However, since the data comes from few and small sample size studies, further research is needed to confirm the effectiveness of the tool in neurorehabilitation.
Keywords: Key words: Virtual reality exposure therapy, Nervous system diseases, Meta-analysis
Introduction
Virtual reality (VR) is an advanced technology that creates simulated environments and conditions. VR allows users to engage in an immersive, multi-sensory, and interactive simulation of ecological scenarios. Virtual scenarios reproduce real objects and events in a 3-D way, providing the users with audio-visual feedback in response to their movement in front of the screen.1, 2 Additionally, VR gives the user the illusion of actively interacting with the scenarios used in training, encouraging not only the improvement of motor and cognitive functions but also the well-being and participation of patients. In fact, the subject becomes the protagonist of the training, abandoning the passive role in his/her own therapy.3, 4 By offering the possibility of combining motor, cognitive, and well-being in conjunction with the potential to manipulate multi-sensorial features in a safe environment,5 VR has emerged as a promising powerful rehabilitation tool.
There are two key concepts of VR: immersion and presence. Immersion is the objective perception of sensory absorption/immersion in a 3-D computer-generated environment. The perception of immersion in a VR environment allows the user to modify and reshape one’s representations of the world.6, 7 The level of immersion experienced in a VR environment is determined by the quantity and diversity of sensory and motor channels integrated into the VR system and the level of accuracy and responsiveness of sensory inputs and motor outputs to user interactions.8 This process is related to the concept of presence, which consists of a subjective psychological state in which the user is consciously involved in the virtual context. Based on these concepts, it is possible to distinguish three types of VR: fully immersive VR, non-immersive VR, and semi-immersive VR. The fully immersive VR absorbs the user, isolating him from the external environment by blocking out their perception of the real world (i.e., giving the full sensation and illusion of being inside the virtual world). The semi-immersive VR replaces the helmet with a standard monitor/interactive wall or a series of monitors (Cave) which allows, thanks to special glasses, a three-dimensional vision to the patient, who has the impression of seeing the world through a window or a wall (i.e., it combines a certain degree of perception of both real and VR environments). On the contrary, non-immersive VR elicits in the user the role of external observer of a three-dimensional reality simply presented on a monitor.6, 7
These perceptive and experiential aspects activated by VR are especially relevant in the rehabilitation field, as motor awareness and emotional involvement can favor functional execution methods and control of complex behavioral sequences, activating motor functions and cognitive abilities of the patient and boosting their functional recovery. In fact, VR systems can enhance the neuro-rehabilitative processes, through increased sensory feedback, and induce changes in the processes of neural plasticity, which allows the re-acquisition of motor and cognitive functionality.9, 10
Among advanced VR systems, various authors have highlighted promising effects in motor and cognitive rehabilitation of the computer-assisted rehabilitation environment (CAREN - Motekforce Link; Amsterdam, The Netherlands). The first prototype of the CAREN was developed in 1998, but it was used in scientific research starting in 2000 by the University of Groningen. Its use was initially intended for scientific and military organizations due to the sheer complexity of the instrument. Only in recent years, the CAREN has also been used for rehabilitation.
CAREN is a dome-simulator equipped with VR screens (from 180° to 360°) and used for neurological and orthopedic patients as well as amputees. CAREN is composed of a series of software and peripherals, such as a sensorized treadmill with plantar pressure sensors, high-speed infrared cameras that map the position of the reflective markers positioned on the patient, and a mobile platform (Figure 1).
Figure 1.
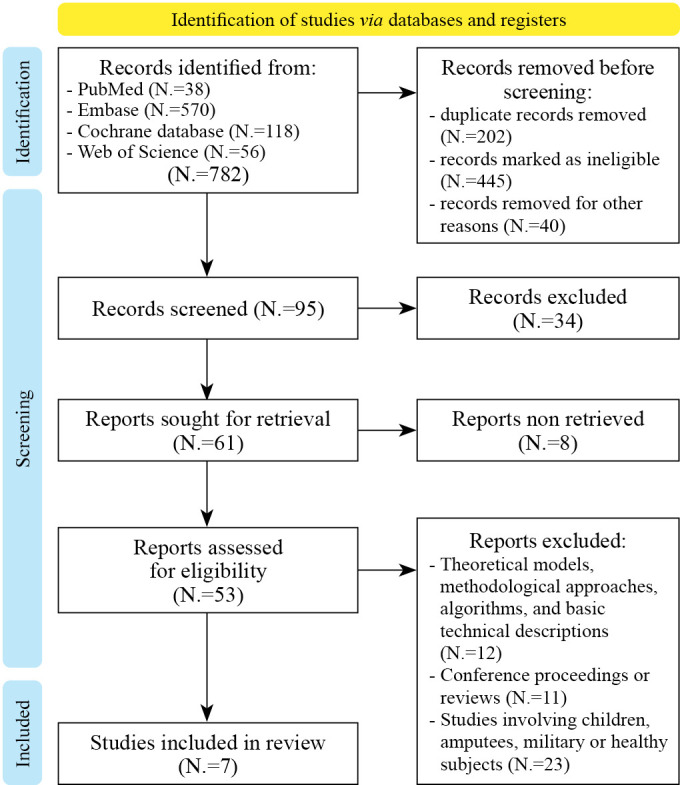
—PRISMA 2020 flow diagram for new systematic reviews which included searches of databases and registers only.
CAREN creates a fully responsive physical and virtual environment for the patient, in which he can feel immersed.11, 12 Because CAREN was specifically designed for rehabilitation and therapy, the tool provides a higher level of customization and control over the simulated environment and can be relatively easily tailored according to the patient’s needs. Therefore, understanding the current state of the art on the use of CAREN in rehabilitation therapy, as well as its effectiveness in neurological diseases, could be important. Nonetheless, in our scoping review, we aimed to map the existing evidence on the use of CAREN in the rehabilitation of neurological patients.
Evidence acquisition
We performed this review, according to the PRISMA extension for scoping reviews, to explore the existing evidence on CAREN as a new rehabilitation strategy (both motor and cognitive) in neurological populations. The use of a scoping review can be the ideal tool for determining the scope or coverage of a body of literature on the use of CAREN in neurorehabilitation, as it provides a clear indication of the volume of literature and studies available, as well as summarizing the results of the trials and the extent of the literature on a topic.13
Search strategy and selections
A search was conducted for all peer-reviewed articles published until June 30, 2023, using the following databases: PubMed, Embase, Cochrane Database, PEDro, and Web of Science. The following terms have been used: (“Cognitive Rehabilitation” OR “Motor Rehabilitation” AND/OR “CAREN” or “Computer-Assisted Rehabilitation Environment”) AND (“Virtual Reality” OR “Rehabilitation” OR “Neurorehabilitation) AND/OR (“Neurological disorders” OR “disease” OR “neurodegenerative” OR “brain/spinal cord injury”).
All articles were reviewed based on titles and abstracts by two investigators (MG.M., R.S.C.), who independently performed data collection to reduce the risk of bias (i.e., missing results bias). These researchers read the full-text articles deemed suitable for the study and, in case of disagreement on the inclusion and exclusion criteria, the final decision was made by a third researcher (A.Q.). The inclusion criteria were: 1) adult patients with neurological disease; 2) an applied approach to cognitive and motor rehabilitation; 3) the English language; and 4) published in a peer-reviewed journal. We excluded articles describing theoretical models, methodological approaches, algorithms, and basic technical descriptions. Additionally, we excluded: 1) animal studies; 2) conference proceedings or reviews; 3) studies involving children, amputees, military, or healthy subjects; and 4) studies that have focused on CAREN only as an assessment tool. The list of articles was then refined for relevance, revised, and summarized, with the key themes identified from the summary based on the inclusion/exclusion criteria.
Data extraction and analysis
After full-text selection, the data extraction from the included studies was summarized in a table (Microsoft Excel – Version 2021). Data summarized were considered for the following information: authors, year, and type of publication (e.g., clinical studies, pilot study); and type of participants (i.e. neurological patients or healthy controls) involved in the study.
Evidence synthesis
The reviewed studies have applied CAREN as an immersive VR tool in the rehabilitation of neurological patients. Thus, we excluded those studies in which the reference populations were healthy subjects, amputees, military without primary neurological disorders, or were related to single cases. Moreover, studies using the tool for the assessment of gait and balance were also excluded.
The initial search returned 782 articles. After thoroughly reviewing the manuscripts, we removed 202 studies because they are duplicate records. Moreover, 485 were excluded for ineligibility (published in a peer-reviewed journal or not in the English language). Then, we excluded 42 studies because they did not have an applied CAREN approach to cognitive and motor rehabilitation; or they were not on neurological patients. Finally, we excluded articles describing theoretical models, methodological approaches, algorithms, and basic technical descriptions (N.=12); animal studies, conference proceedings, or reviews (N.=11); and studies involving children, amputees, the military, or healthy subjects (N.=23). Finally, we considered only seven studies (Supplementary Digital Material 1: Supplementary Table I) (Figure 2).
Figure 2.

—The CAREN tool consisting of a six-DOF platform, with a dual belt treadmill and a 180° VR screen.
From the included studies: 1) one study concerned cognitive rehabilitation in patients suffering from Parkinson’s Disease (PD);14 2) one was on the usability of CAREN in PD patients;15 3) two studies related to the influence of emotional components on CAREN rehabilitation;16, 17 4) two studies were related to motor rehabilitation using CAREN, one on PD patients,18 one on patients affected by Multiple Sclerosis (MS),19 and one on TBI patients.20
Generally, these studies demonstrate that CAREN is a safe and potentially effective tool to treat different symptoms (including gait and vestibular disturbances, executive function, depressive mood, and anxiety) in patients with different neurological disorders. In detail, Formica et al.14 carried out a study on 31 PD patients who underwent 24 training sessions in a computer-assisted rehabilitation environment. The results highlighted that CAREN could promote functional recovery of cognitive and emotional domains, including executive function, anxiety, and depressive symptoms.14
Impellizzeri et al.15 found in a case-control study of Parkinson’s disease that users experienced no severe discomfort. Cybersickness is a subtype of motion sickness that arises from immersion in VR, and it may cause many symptoms, including nausea, vomiting, pallor, eructation, salivation, drowsiness, dizziness, headaches, an illusion of movement, and postural instability. The authors have postulated that walking and moving the CAREN platform (that adapts to the patient’s gait thanks to its six degrees of movement and reproduces the movement of the virtual environment) may have played a role in the reduction of perceived motion sickness since it reduces the sensory mismatch.
Kane et al. found in a sample of seventy-five adults with MS that mood is not affected by the CAREN system,16 whereas Onakomaiya et al.17 have shown that physical performance on CAREN can be effectively influenced by the presence of comorbid emotional disorders, such as post-traumatic stress disorder (PTSD).
Concerning motor outcomes following CAREN training, the studies have found an improvement in posture, gait, and balance.18-20 Calabrò et al.,18 in their preliminary study on 22 PD patients, found that after the training with this tool, patients walked faster and with greater stability, with wider and longer steps.20 In line with these results, Kalron et al.19 showed that CAREN could be very useful in balance training in people with MS, with better results than patients undergoing conventional training.19 Finally, Sessoms et al.20 observed in patients with TBI that vestibular physical therapy using CAREN may lead to greater improvements in gait speed and weight shift than subjects receiving traditional vestibular therapy (Supplementary Table I).
Discussion
In our scoping review, we aimed to map the existing evidence on the use of CAREN in the rehabilitation of neurological patients. The few studies included, indicate that CAREN could be a safe and potentially useful tool to improve different cognitive and motor domains in different neurological populations, but with conflicting results concerning emotional features. Indeed, the treatment with the device was feasible and safe, and there were no reported adverse events, including cybersickness.17 Improving patient safety (i.e., “avoidance, prevention and amelioration of adverse outcomes or injuries stemming from the process of healthcare”) is a major goal in healthcare systems worldwide. Then, finding new safe training approaches in neurorehabilitation is fundamental. Based on the few available data, CAREN may be safely used to train different neurological patients (including PD, TBI, MS) thanks to the hardware feature of the device (the fall arrest harness, sensorized treadmill, and 3-DOF balance real-time interaction with the immersive VR) and constant presence and supervision of the therapist.
The tool seems to improve gait and balance as well as vestibular dysfunction thanks to the six-DOF treadmill’s ability to act as a bottom-up sensory-motor stimulation. Cognition and behavior are instead stimulated by the immersive virtual environment. Reasonably, the potential effects of CAREN may be due to greater sensory involvement (visual and auditory feedback), which stimulates patient compliance and treatment motivation.3, 8, 14-20 The observed benefits of CAREN are in line with the effects of semi-immersive VR tools, suggesting that rehabilitation implemented with VR has multiple benefits for neurological patients. This is likely due to the effectiveness of VR training in inducing neural reorganization and promoting the recovery of motor skills following neurological damage.21 This is very important for the rehabilitation field and could be useful in various neurological disorders, in which pharmacological treatments may be of limited efficacy.4, 22-25 Studies using semi-immersive VR tools, such as BTs Nirvana or Virtual Reality rehabilitation system, on patients suffering from stroke, TBI, PD, or MS showed improvements in motor learning, attention, visuoperceptual skills, memory, and executive functions.23-26 This is possible because VR provides the patient with multi-sensory feedback, which stimulates neural plasticity processes, favoring/improving functional recovery.21 Furthermore, VR could affect the cognitive reserve of patients, improving their response to treatment.27 Cognitive reserve depends both on the premorbid stage (years of education, profession, and social activities) and is stimulated by adulthood activities.28, 29 Thus, further studies would be useful to evaluate the benefit of immersive VR, which could potentially have a greater impact than the semi-immersive modality, as it could allow for greater motor, perceptual, and sensorial involvement. This aspect is underlined by single case studies, highlighting an important functional outcome through rehabilitation with CAREN, compared to other types of rehabilitation, in terms of cognitive and motor enhancement.30-32
Moreover, the reviewed studies support the use of CAREN, which, as an immersive VR tool, can favor not only the improvement of motor and cognitive functions but also their degree of well-being and participation.14-20 Participation and motivation are fundamental during the training and are known to be key factors for functional recovery.
In recent years, VR has found numerous applications in the rehabilitation paths of children and adults affected by motor, cognitive, behavioral, and sensory disabilities.23 Indeed, VR has considerable advantages since it allows calibration of the difficulties of the tasks on the capabilities and potential of the user and monitors the patient’s performance using various feedbacks (visual and auditory). Furthermore, the systems make it possible to increase the quality of the intervention, allowing the therapist to present rehabilitation activities playfully, with an increase in the patient’s motivation and involvement. The users can safely face daily activities otherwise not accessible in hospital settings (such as playing musical instruments, cooking, skating, and driving).3 The experience with VR brings the patient to the center of his/her therapeutic program, allowing him/her to modify and remodel his representations of the world. These perceptive and experiential aspects activated by VR are particularly relevant in the neurorehabilitation field, as motor awareness and emotional involvement can increase the functional execution modalities and the control of complex behavioral sequences.9 Consequently, these aspects can activate the patient’s motor and cognitive skills and promote functional recovery.
A key concept in rehabilitation with CAREN and VR devices is neuroplasticity. Neuroplasticity is the ability of our brain to modify itself through interactions with the external environment for different mechanisms, such as glial cell activation, supersensitivity denervation, and metabolic changes.24 Multi-sensory VR feedback and the repeated execution of cognitive and physical tasks could improve the patient’s functional outcome, amplifying the processes of brain plasticity through motor learning and resulting in profound cortical and subcortical modifications at the cellular level and synaptic.9 These effects may be due to the reactivation/amplification of cerebral neurotransmission and to the involvement of mirror neurons thanks to the visuomotor information coming from the observation of the stimuli presented with VR.33-36 This allows integration between perception, cognition, and action and the recall of memorized motor plans. This process is due to reinforcement learning, which allows for greater efficiency and better performance.37, 38 Therefore, the advantage of using VR, especially with immersive tools such as CAREN, is to create a positive, fun, and motivating learning experience for the patient that requires individual control over several sensory-motor, cognitive, and social domains.3, 4, 9 The increased feedback obtained through exercises performed in a virtual environment allows for the development of the “awareness of the results” of the movements performed and the “awareness of the quality” of the movements themselves with positive effects on a cognitive and motor level.14-20
Limitations of the study
The main limitation of this scoping review is that it involves few studies with a small sample size, and this does not allow for the generalization of the results to the neurological population. However, the aim of this scoping review was to map the existing literature on the role of CAREN in neurorehabilitation, since the body of literature on this issue has not yet been comprehensively reviewed. In addition, a systematic review with meta-analysis to state the efficacy of the tool is still not possible, given the few studies with few neurological patients treated with this innovative device. In the near future, whether new studies on this topic are published, a systematic review is welcomed.
Moreover, we did not focus on a specific neurological disorder, and this may have biased the results since the recovery from neurodegenerative illness and acquired brain injury is completely different. RCT with a more homogeneous sample (e.g., neurodegenerative vs. brain injury) is needed to confirm the positive effect of CAREN on motor and cognitive outcomes.
The review also presents publication bias since only one study by Kalron19 has protocol registration, as well as language bias because we did not include articles that have been written in different languages, but English. Lastly, we did not register the protocol, although also for scoping review, it is recommended. Nonetheless, as far as we know, this is the first ever review dealing with such an important issue.
Nonetheless, given the features of this innovative device and the encouraging data to date published, CAREN could be considered as a complementary treatment in the management of neurological patients. Unfortunately, the use of the tool in clinical practice is far from being realized because of the high costs required for purchase and maintenance, as well as the need for highly specialized personnel for its use. Then, to date, the VR system can be used only for research purposes.
Conclusions
The current literature indicates that CAREN could be a valuable tool in the neurorehabilitation field. Indeed, the few available studies demonstrate the potential role of CAREN in improving motor and cognitive outcomes, with conflicting results on the emotional aspects.
Given that our results come from a small and heterogeneous sample, future studies should involve larger samples, short- and long-term follow-up, and more homogeneous populations. This is fundamental to confirm the effectiveness of the tool in neurological patients and better understand the neural mechanisms subtending the VR-related functional recovery.
Supplementary Digital Material 1
Supplementary Table I o Figure 1 o Text File 1
Main studies concerning CAREN rehabilitation.
Footnotes
Conflicts of interest :The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Funding :This research was supported by the current Research Fund 2023, the Italian Ministry of Health.
References
- 1.Burdea GC. Virtual rehabilitation—benefits and challenges. Methods Inf Med 2003;42:519–23. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=14654886&dopt=Abstract 10.1055/s-0038-1634378 [DOI] [PubMed] [Google Scholar]
- 2.Wade E, Winstein CJ. Virtual reality and robotics for stroke rehabilitation: where do we go from here? Top Stroke Rehabil 2011;18:685–700. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=22436307&dopt=Abstract 10.1310/tsr1806-685 [DOI] [PubMed] [Google Scholar]
- 3.De Luca R, Russo M, Naro A, Tomasello P, Leonardi S, Santamaria F, et al. Effects of virtual reality-based training with BTs-Nirvana on functional recovery in stroke patients: preliminary considerations. Int J Neurosci 2018;128:791–6. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=29148855&dopt=Abstract 10.1080/00207454.2017.1403915 [DOI] [PubMed] [Google Scholar]
- 4.Maggio MG, Maresca G, De Luca R, Stagnitti MC, Porcari B, Ferrera MC, et al. The Growing Use of Virtual Reality in Cognitive Rehabilitation: Fact, Fake or Vision? A Scoping Review. J Natl Med Assoc 2019;111:457–63. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=30739728&dopt=Abstract 10.1016/j.jnma.2019.01.003 [DOI] [PubMed] [Google Scholar]
- 5.Canning CG, Allen NE, Nackaerts E, Paul SS, Nieuwboer A, Gilat M. Virtual reality in research and rehabilitation of gait and balance in Parkinson disease. Nat Rev Neurol 2020;16:409–25. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=32591756&dopt=Abstract 10.1038/s41582-020-0370-2 [DOI] [PubMed] [Google Scholar]
- 6.Riva G, Baños RM, Botella C, Mantovani F, Gaggioli A. Transforming Experience: The Potential of Augmented Reality and Virtual Reality for Enhancing Personal and Clinical Change. Front Psychiatry 2016;7:164. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=27746747&dopt=Abstract 10.3389/fpsyt.2016.00164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sheridan TB. Musing on telepresence and virtual presence. Presence (Camb Mass) 1992;1:120–5. 10.1162/pres.1992.1.1.120 [DOI] [Google Scholar]
- 8.Bohil CJ, Alicea B, Biocca FA. Virtual reality in neuroscience research and therapy. Nat Rev Neurosci 2011;12:752–62. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=22048061&dopt=Abstract 10.1038/nrn3122 [DOI] [PubMed] [Google Scholar]
- 9.Calabrò RS, Naro A, Russo M, Leo A, De Luca R, Balletta T, et al. The role of virtual reality in improving motor performance as revealed by EEG: a randomized clinical trial. J Neuroeng Rehabil 2017;14:53. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=28592282&dopt=Abstract 10.1186/s12984-017-0268-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Calabrò RS, Russo M, Naro A, De Luca R, Leo A, Tomasello P, et al. Robotic gait training in multiple sclerosis rehabilitation: can virtual reality make the difference? Findings from a randomized controlled trial. J Neurol Sci 2017;377:25–30. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=28477702&dopt=Abstract 10.1016/j.jns.2017.03.047 [DOI] [PubMed] [Google Scholar]
- 11.Lees A, Vanrenterghem J, Barton G, Lake M. Kinematic response characteristics of the CAREN moving platform system for use in posture and balance research. Med Eng Phys 2007;29:629–35. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=16952478&dopt=Abstract 10.1016/j.medengphy.2006.06.004 [DOI] [PubMed] [Google Scholar]
- 12.de Groot IJ, Zohar OE, Haspels R, van Keeken H, Otten E. CAREN (computer assisted rehabilitation environment): a novel way to improve shoe efficacy. Prosthet Orthot Int 2003;27:158–62. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=14571947&dopt=Abstract 10.1080/03093640308726673 [DOI] [PubMed] [Google Scholar]
- 13.Tricco AC, Lillie E, Zarin W, O’Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and Explanation. Ann Intern Med 2018;169:467–73. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=30178033&dopt=Abstract 10.7326/M18-0850 [DOI] [PubMed] [Google Scholar]
- 14.Levac D, Colquhoun H, O’Brien KK. Scoping studies: advancing the methodology. Implement Sci 2010;5:69. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=20854677&dopt=Abstract 10.1186/1748-5908-5-69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Formica C, Bonanno L, Latella D, Ferrera MC, Maresca G, Logiudice AL, et al. The effect of Computer Assisted Rehabilitation Environment (CAREN) in cognitive impairment and coping strategies in Parkinson’s disease: a preliminary study. Sci Rep 2023;13:2214. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=36750642&dopt=Abstract 10.1038/s41598-023-29299-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Impellizzeri F, Naro A, Basile G, Bramanti A, Gazia F, Galletti F, et al. Does cybersickness affect virtual reality training using the Computer Assisted Rehabilitation Environment (CAREN)? Preliminary results from a case-control study in Parkinson’s disease. Physiother Theory Pract 2022;38:2603–11. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=34365911&dopt=Abstract 10.1080/09593985.2021.1964117 [DOI] [PubMed] [Google Scholar]
- 17.Kane A, Thompson NR, Sullivan AB. Assessment of Computer Assisted Rehabilitation Environment (CAREN) System Use and Mood in Patients With Multiple Sclerosis. Int J MS Care 2022;24:63–6. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=35462873&dopt=Abstract 10.7224/1537-2073.2020-131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Onakomaiya MM, Kruger SE, Highland KB, Kodosky PN, Pape MM, Roy MJ. Expanding Clinical Assessment for Traumatic Brain Injury and Comorbid Post-Traumatic Stress Disorder: A Retrospective Analysis of Virtual Environment Tasks in the Computer-Assisted Rehabilitation Environment. Mil Med 2017;182(S1):128–36. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=28291464&dopt=Abstract 10.7205/MILMED-D-16-00054 [DOI] [PubMed] [Google Scholar]
- 19.Calabrò RS, Naro A, Cimino V, Buda A, Paladina G, Di Lorenzo G, et al. Improving motor performance in Parkinson’s disease: a preliminary study on the promising use of the computer assisted virtual reality environment (CAREN). Neurol Sci 2020;41:933–41. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=31858331&dopt=Abstract 10.1007/s10072-019-04194-7 [DOI] [PubMed] [Google Scholar]
- 20.Kalron A, Fonkatz I, Frid L, Baransi H, Achiron A. The effect of balance training on postural control in people with multiple sclerosis using the CAREN virtual reality system: a pilot randomized controlled trial. J Neuroeng Rehabil 2016;13:13. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=26925955&dopt=Abstract 10.1186/s12984-016-0124-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sessoms PH, Gottshall KR, Collins JD, Markham AE, Service KA, Reini SA. Improvements in gait speed and weight shift of persons with traumatic brain injury and vestibular dysfunction using a virtual reality computer-assisted rehabilitation environment. Mil Med 2015;180(Suppl):143–9. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=25747645&dopt=Abstract 10.7205/MILMED-D-14-00385 [DOI] [PubMed] [Google Scholar]
- 22.You SH, Jang SH, Kim YH, Hallett M, Ahn SH, Kwon YH, et al. Virtual reality-induced cortical reorganization and associated locomotor recovery in chronic stroke: an experimenter-blind randomized study. Stroke 2005;36:1166–71. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=15890990&dopt=Abstract 10.1161/01.STR.0000162715.43417.91 [DOI] [PubMed] [Google Scholar]
- 23.Son C, Park JH. Ecological Effects of VR-Based Cognitive Training on ADL and IADL in MCI and AD patients: A Systematic Review and Meta-Analysis. Int J Environ Res Public Health 2022;19:15875. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=36497946&dopt=Abstract 10.3390/ijerph192315875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Maggio MG, De Luca R, Manuli A, Buda A, Foti Cuzzola M, Leonardi S, et al. Do patients with multiple sclerosis benefit from semi-immersive virtual reality? A randomized clinical trial on cognitive and motor outcomes. Appl Neuropsychol Adult 2022;29:59–65. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=31920097&dopt=Abstract 10.1080/23279095.2019.1708364 [DOI] [PubMed] [Google Scholar]
- 25.Darekar A. Virtual Reality for Motor and Cognitive Rehabilitation. Curr Top Behav Neurosci 2023;65:337–69. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=37041455&dopt=Abstract 10.1007/7854_2023_418 [DOI] [PubMed] [Google Scholar]
- 26.Maggio MG, De Cola MC, Latella D, Maresca G, Finocchiaro C, La Rosa G, et al. What About the Role of Virtual Reality in Parkinson Disease’s Cognitive Rehabilitation? Preliminary Findings From a Randomized Clinical Trial. J Geriatr Psychiatry Neurol 2018;31:312–8. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=30360679&dopt=Abstract 10.1177/0891988718807973 [DOI] [PubMed] [Google Scholar]
- 27.Leonardi S, Maggio MG, Russo M, Bramanti A, Arcadi FA, Naro A, et al. Cognitive recovery in people with relapsing/remitting multiple sclerosis: A randomized clinical trial on virtual reality-based neurorehabilitation. Clin Neurol Neurosurg 2021;208:106828. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=34332269&dopt=Abstract 10.1016/j.clineuro.2021.106828 [DOI] [PubMed] [Google Scholar]
- 28.Stern Y. Cognitive reserve. Neuropsychologia 2009;47:2015–28. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=19467352&dopt=Abstract 10.1016/j.neuropsychologia.2009.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sumowski JF, Wylie GR, Gonnella A, Chiaravalloti N, Deluca J. Premorbid cognitive leisure independently contributes to cognitive reserve in multiple sclerosis. Neurology 2010;75:1428–31. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=20956787&dopt=Abstract 10.1212/WNL.0b013e3181f881a6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Booth AJ, Rodgers JD, Schwartz CE, Quaranto BR, Weinstock-Guttman B, Zivadinov R, et al. Active cognitive reserve influences the regional atrophy to cognition link in multiple sclerosis. J Int Neuropsychol Soc 2013;19:1128–33. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=24050681&dopt=Abstract 10.1017/S1355617713001082 [DOI] [PubMed] [Google Scholar]
- 31.Cellini R, Paladina G, Mascaro G, Lembo MA, Lombardo Facciale A, Ferrera MC, et al. Effect of Immersive Virtual Reality by a Computer Assisted Rehabilitation Environment (CAREN) in Juvenile Huntington’s Disease: A Case Report. Medicina (Kaunas) 2022;58:919. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=35888638&dopt=Abstract 10.3390/medicina58070919 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.De Luca R, Manuli A, De Domenico C, Lo Voi E, Buda A, Maresca G, et al. Improving neuropsychiatric symptoms following stroke using virtual reality: A case report. Medicine (Baltimore) 2019;98:e15236. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=31083155&dopt=Abstract 10.1097/MD.0000000000015236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.De Luca R, Portaro S, Le Cause M, De Domenico C, Maggio MG, Cristina Ferrera M, et al. Cognitive rehabilitation using immersive virtual reality at young age: A case report on traumatic brain injury. Appl Neuropsychol Child 2020;9:282–7. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=30838889&dopt=Abstract 10.1080/21622965.2019.1576525 [DOI] [PubMed] [Google Scholar]
- 34.Schedlbauer AM, Copara MS, Watrous AJ, Ekstrom AD. Multiple interacting brain areas underlie successful spatiotemporal memory retrieval in humans. Sci Rep 2014;4:6431. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=25234342&dopt=Abstract 10.1038/srep06431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Carrieri M, Petracca A, Lancia S, Basso Moro S, Brigadoi S, Spezialetti M, et al. Prefrontal cortex activation upon a demanding virtual hand controlled task: a new frontier for neuroergonomics. Front Hum Neurosci 2016;10:53. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=26909033&dopt=Abstract 10.3389/fnhum.2016.00053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Allison SL, Fagan AM, Morris JC, Head D. Spatial navigation in preclinical Alzheimer’s disease. J Alzheimers Dis 2016;52:77–90. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=26967209&dopt=Abstract 10.3233/JAD-150855 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rizzo A, Kim GJ. A SWOT analysis of the field of RV rehabilitation and therapy. Presence: Teleoperators and Virtual Envir. 2005;14:119–46. 10.1162/1054746053967094 [DOI]
- 38.Rizzolatti G, Fogassi L. The mirror mechanism: recent findings and perspectives. Philos Trans R Soc Lond B Biol Sci 2014;369:20130420. https://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&list_uids=24778385&dopt=Abstract 10.1098/rstb.2013.0420 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Table I o Figure 1 o Text File 1
Main studies concerning CAREN rehabilitation.


