Abstract
Background
Helicobacter pylori infection has been identified to cause constantly recurring inflammation, leading to gastrointestinal tract disorders, including carcinoma. The standard triple therapy (STT), used to eradicate H. pylori, includes two antimicrobials and a proton pump inhibitor for two weeks. Other drug regimens have also been developed since H. pylori exhibits antimicrobial resistance. These regimens, including probiotics, have been shown to lower adverse drug reactions (ADR), improve drug adherence, exert bacteriostatic effect, and reduce inflammation.
Objective
This study intended to explore probiotic intervention for improving eradication rates and mitigating adverse effects while administrating STT.
Methods
This prospective study was conducted from May to December, 2021, in the Department of Gastroenterology of Ship International Hospital, Dhaka, Bangladesh, to observe the effects of probiotics inclusion along with STT on H. pylori eradication. A total of 100 patients aged ≥18 years who tested positive for H. pylori were included. The experimental group (n=50) was given STT and probiotics, and the control group (n=50) was given only STT without probiotics for 14 days. Necessary follow-up was done six weeks after treatment. An independent sample t-test, chi-square test, and multiple regression analysis were used for statistical analysis.
Result
The odds of getting rapid urease test (RUT) negative results from positive were 2.06 times higher (95%CI= 0.95, 3.22, p=0.054) in the experimental group. ADRs were crucially towering in the control group (p=0.045) compared to the probiotics group. The probiotics group had a lower risk of having adverse effects by 0.54 times (95%CI=0.19, 0.84, p=0.032) than the control group.
Conclusion
Using probiotics and STT together to eradicate H. pylori may lower ADR and improve treatment adherence. It may also help terminate H. pylori infection more effectively. More research is required as H. pylori is very contagious and can ultimately cause life-threatening gastric cancer.
Keywords: h. pylori treatment, helicobacter pylori, triple therapy, bangladesh, pud, peptic ulcer disease, probiotic microflora, probiotic bacterium
Introduction
Peptic ulcer disease (PUD) is described as hydrochloric acid-provoked damage of the epithelial lining of the gastrointestinal tract, arising predominantly in the stomach and duodenum called gastric ulcer (GU) and duodenal ulcer (DU), respectively [1,2]. Australia-born Professor (Dr.) Barry James Marshall and Dr. John Robin Warren first reported in 1982 that the principal cause of PUD is infective disorders caused by curved gram-negative bacillus Helicobacter pylori, and later, in 2005, both obtained the Nobel Prize jointly for this discovery [3-5]. Earlier, it was believed that regular ingestion of alcohol and tobacco, psychological multiple disorders, nonsteroidal anti-inflammatory drugs (NSAIDs), and lifestyle issues were principal causes of PUD [6-8]. Marshall and Warren refuted the age-old concept of these causes of PUD [4,5,9].
It is known that H. pylori causes around 90-95% and 80-85% of DU and GU, respectively [4,10,11]. Multiple studies reported that more than 50% of the globe's populace is infected with H. afpylori [12,13], with a considerable difference in the frequency among countries and the prevalence observed within a country [12-15]. Nevertheless, all patients infected with H. pylori do not acquire PUD; only 10% of individuals develop PUD [10,16,17]. H. pylori is responsible for the chronic infection that triggers a chronic inflammatory process in the superficial epithelial layer of the stomach; later, it causes several gastrointestinal disorders, including carcinoma [18-21].
Hooi et al. steered a systematic review and meta-analysis to make public that the African (70.1%; 95%CI, 62.6-77.7) and Australian (Oceania) (24.4%; 95%CI, 18.5-30.4) continents had the maximum and minimum pooled generality of H. pylori infection, respectively. The pervasiveness rate of H. pylori infection varies in different nations with the lowest in Switzerland (18.9%) and the highest in Nigeria (87.7%) [15]. Congedi et al. published a scoping review that reported a decreased tendency of H. pylori infection among Australian citizens. Nevertheless, the scoping review could not confirm the infectious potential of H. pylori among susceptible clusters [22]. Another systematic review conducted by Peleteiro et al. reported that the highest and lowest rates of H pylori infection were found in Mexico (90%) and Finland (13.1%), respectively [23]. Although distinct differences are observed regarding H. pylori infection rates across the world, the general preponderance of H. pylori infection is around 50% of the worldwide populace [24,25].
The mortality rate because of H. pylori infection was considerably higher among the non-pharmacologically intervened group than in treated clusters (p<0.001) [26]. It has been ascertained that the deaths among pharmacologically intervened and non-intervened clusters were 4.1-5.9% and 5.5-7.6%, respectively [26,27]. H. pylori has been identified as the foremost jeopardizing factor in gastric carcinoma [28]. Stomach cancer is considered the fifth leading malignancy and stands in third position for carcinoma-related mortality around the globe [29]. It has been reported that 770,000 [30] to 800,000 [31] people passed away because of stomach cancer, and around 1.1 million fresh cases were seen in 2020 [30]. The rate of frequency of stomach carcinoma is influenced by sex. H. pylori and related diseases affect male subjects two-fold more than female counterparts [30]. It has been appraised that stomach cancer alone accounts for 7.7% of all carcinoma-related deaths [31]. It is thus strongly advocated to eradicate H. pylori, especially in seropositive individuals with cytotoxin-associated gene A (CagA) [32-36]. CagA is an external cancer-stimulating protein for humans generated by some strains of H. pylori [34,37].
Australian Dr. Thomas J. Borody introduced the bismuth-based triple therapy containing bismuth and two antimicrobials. This was the first successful effort to manage H. pylori abolition [38,39]. Goh et al. put forward the theory that the therapeutic intervention for H. pylori infection should include two antimicrobials (clarithromycin and amoxicillin) and a proton pump inhibitor (PPI) for two weeks [40]. This combination is often called standard triple therapy (STT). Additionally, in those cases of H. pylori infection that are isensitive to penicillin (amoxicillin), it is replaced with metronidazole [40,41]. Other than the mentioned antimicrobials, tetracyclines and fluoroquinolones are commonly used for H. pylori eradication [42-44].
The National Institutes of Health brought the first consensus report regarding eradicating H. pylori infection in 1994 [45]. Subsequently, the European H. pylori Study Group endorsed STT as the leading therapeutic strategy during the first Maastricht conference in 1997 [46]. Furthermore, multiple consensus guidelines published in the last two decades or more for the therapeutic intervention of H. pylori infection, such as Maastricht VI/Florence [47], the Toronto [48], Kyoto Global [49], Hong Kong recommendation [50], Taipei Global [51] and many more [52-56].
H. pylori is a gram-negative, spiral-shaped, microaerophilic, and highly infective pathogenic microbe; mounting antimicrobial resistance around the globe creates an alarming human health threat [57,58]. Many treatment regimes and attempts at eradicating H. pylori were unsuccessful because of resistance [42,59-61]. H. pylori attained 100-1000 times more resistance to multiple antimicrobials when H. pylori were grown in the matching floating form or cholesterol [62-65]. Another study revealed that H. pylori's resistance to clarithromycin was 22.2%. Amoxicillin and metronidazole resistance in H. pylori were 1.2% and 69.2%, respectively. The resistance patterns in the United States and Europe were similar and resistance to metronidazole was found to be the highest (50-79%) and the least resistance was to amoxicillin (equal to or lower than 5%) [66]. Global resistance pattern against commonly prescribed antimicrobials for H. pylori infection eradication is high and ranges from 15-50% [67]. Multiple studies reported that among eight South Asian countries (Bhutan, Bangladesh, India, Indonesia, South Korea, Nepal, Sri Lanka, and Thailand), the antimicrobial resistance of H. pylori patterns is much higher (98%). There is a high incidence of self-purchasing of antimicrobials from community pharmacies, poor antimicrobial stewardship programs, and rapid alteration of geopolitical scenery and urbanization [68-71]. One global systematic review and meta-analysis also reported that 15% of H. pylori possesses either primary or secondary resistance towards clarithromycin, metronidazole, and levofloxacin in practically all world regions [72]. Nahar et al. reported from Bangladesh that H. pylori isolates were resistant to amoxicillin, clarithromycin, tetracycline, and metronidazole, and the resistance was determined to be 6.6%, 10%, 15%, and 77.5%, respectively [73]. Over a decade after the study by Mahar et al. [72], Aftab et al. reported that H. pylori isolates in Bangladesh showed resistance to metronidazole, levofloxacin, and clarithromycin, and resistance rates were 94.6%, 66.1%, and 39.3%, respectively [74].
Our planet faces treatment difficulties regarding infectious diseases because of antimicrobial resistance to almost all available antimicrobials, which poses an enormous global health threat [75,76]. Overall, antimicrobial resistance around the globe is the foremost public health issue, which equally affects H. pylori and related diseases because of the evolution of drug resistance. Moreover, H. pylori has been notified as the principal factor for gastric carcinoma with fatal consequences [77]. In this antagonist situation, researchers developed multiple other regimens, such as bismuth-containing quadruple (hybrid therapy (HT) including a PPI, bismuth, metronidazole, and tetracycline), sequential (PPI plus amoxicillin followed by PPI, clarithromycin, and an imidazole), PCN (PPI, clarithromycin and nitroimidazoles), and concurrent or accompaniment (non-bismuth quadruple including PPI, clarithromycin, amoxicillin, and metronidazole) therapy to combat antimicrobial-resistant H. pylori infection beside SST [78-83]. However, even with all these efforts, H. pylori eradication often failed because of resistance to multiple antimicrobials [84], such as amoxicillin, clarithromycin, metronidazole, and levofloxacin [41,85].
Diarrhea, constipation, nausea, vomiting, epigastric pain, flatulence, metallic taste, and pain in the abdomen, especially in the epigastric region, are frequently occurring adverse drug reactions (ADRs) following SST therapy to combat H. pylori infection [86,87]. Another study revealed that over 26% of the research participants had experienced ADRs [88]. Among them, 85% had gastrointestinal issues, such as gastrointestinal distress, nausea, uncomfortable or infrequent bowel movements (typically less than three times per week), loose motions, stomach upset, an eating disorder frequently accompanied by unrestrained body weight loss, and headache [88,89]. Another study revealed that over 45% of participants encountered principally gastrointestinal ADRs [90]. Again, around 5% of patients were unable to continue therapeutic intentions because of profound ADRs, and approximately 3% of cases were unable to take less than 80% of medication because of ADRs. Total or partial discontinuation or medication adherence to H. pylori therapeutic intervention frequently leads to eradication failure and promotes antimicrobial resistance [91-93]. Consequently, research analyses narrated that the foremost cause of H. pylori abolition failure is necessitous treatment incompliance that is related to STT-induced ADRs of prescribed drugs [88,94,95].
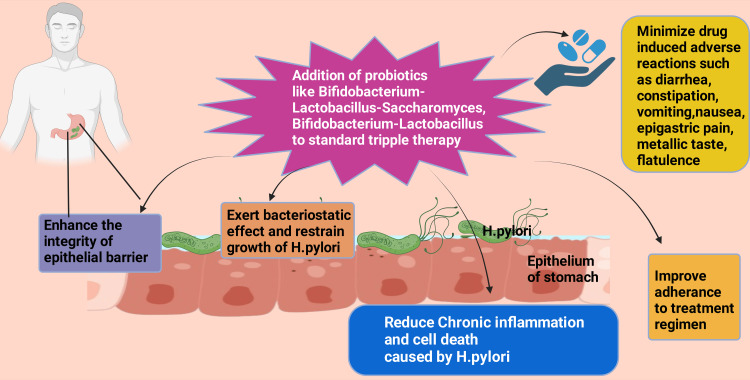
One metanalysis comprising 34 randomized control trials and over 9000 cases showed that the addition of Bifidobacterium-Lactobacillus-Saccharomyces and Bifidobacterium-Lactobacillus-based probiotics with diverse regimens of H. pylori extinction program resulted in minimizing ADRs and promoting medication adherence [96]. Probiotics competitively restrain the growth of H. pylori in the stomach with their bacteriostatic effect. Additionally, probiotics improve gastrointestinal microbiome status [97]. It has been reported that Saccharomyces boulardii, Bacillus licheniformis, Lactobacillus acidophilus, Bifidobacterium triple viable bacteria, and Bacillus subtilis dual viable bacteria are currently in clinical use in the management of H. pylori eradication program. These probiotics increase medicine adherence, cut back ADRs, especially antimicrobial-persuaded, mitigate the stomach mucosal chronic inflammation instigated by H. pylori, and increase the abolition rate of H. pylori when administered with various regimes as an adjuvant [97-107].
Problem statements of this study
Addressing the following problem statements can contribute significantly to developing more effective and patient-friendly H. pylori eradication therapies, ultimately improving treatment outcomes and reducing the global burden of H. pylori-associated diseases.
Efficacy Challenges in STT
One significant problem to address is the suboptimal extinction rate of H. pylori with the STT, consisting of a PPI, amoxicillin, and clarithromycin/levofloxacin. Despite being a widely employed treatment, the efficacy is compromised due to antimicrobial resistance and other factors [108].
Antibiotic-Related ADR
Another critical issue is the high incidence of ADRs associated with antibiotics, particularly clarithromycin and levofloxacin. Gastrointestinal disturbances, allergic reactions, and the development of antibiotic resistance are among the adverse effects that impact patient tolerance and compliance during H. pylori eradication therapy [109].
Impact of Dysbiosis on Treatment Outcome
The STT often disrupts the gut microbiota balance, leading to dysbiosis. This disturbance in the natural microbial community may contribute to prolonged recovery, increased susceptibility to infections, and other complications, necessitating exploring interventions to mitigate dysbiosis [110].
Need for Improved Treatment Strategies
Given the global rise in antibiotic resistance and the limitations of the STT, there is an urgent need to explore adjunctive therapies that can enhance eradication rates. Probiotics represent a promising avenue, but their specific role and mechanisms in improving treatment outcomes remain understudied and require comprehensive investigation [111].
Patient Non-Adherence and Treatment Failure
Poor patient compliance poses a significant challenge in H. pylori eradication therapy. Understanding the impact of probiotics on patient adherence and exploring strategies to improve compliance is crucial for achieving better treatment outcomes and reducing the risk of antibiotic resistance [94].
Variability in Probiotic Strains and Formulations
The variability in probiotic strains and formulations available in the market raises questions about their consistent effectiveness. Addressing the optimal selection, dosage, and duration of probiotic supplementation is essential to establish evidence-based recommendations for integration into H. pylori eradication regimens [112,113].
Objectives of the study
This study explores probiotic intervention for improving eradication rates and mitigating side effects in STT. It assesses the impact of probiotic supplementation on the eradication rate of H. pylori infection when combined with STT and evaluates the reduction in adverse effects of antibiotics of triple therapy, such as nausea, diarrhea, abdominal discomfort, metallic taste, headache, and joint pain and in determining the potential influence of probiotics on patient compliance and adherence to the prescribed treatment regimen. It also provides evidence-based recommendations regarding incorporating probiotics into STT for H. pylori extinction in clinical situations.
Materials and methods
This was a prospective study conducted at the Department of Gastroenterology, Ship International Hospital, Dhaka, Bangladesh, from May to December, 2021. The study obtained ethical approval from the Institutional Review Board of Ship International Hospital (Formerly, Japan East West Medical College Hospital), Dhaka, Bangladesh (approval number: JEWMCH/IEC/01, dated March 5, 2021). Furthermore, written informed consent was obtained from all participants and the study adhered to the World Medical Association's Declaration of Helsinki, ethical principles for medical research involving human subjects. Additionally, research objectives and future publication plans were explained in detail to patients and guardians.
Inclusion and exclusion criteria
Inclusion Criteria
The following inclusion criteria defined the patients eligible for the study and provided a clear framework for selecting study participants: 1. Patients who were candidates for diagnostic endoscopy of the upper gastrointestinal tract (UGIT) with rapid urease test (RUT); 2. Patients who tested conclusively for H. pylori infection by RUT during the endoscopy; 3. Patients aged 18 years or older.
Exclusion Criteria
The following exclusion criteria ensured that the study population was well-defined and that the results were not confounded by factors that could affect the interpretation of the study outcomes: 1. Patients with a history of taking NSAIDs; 2. Patients with a previous history or report of hepatic, renal, or neoplastic diseases; 3. Pregnant women or lactating mothers; 4. Patients who received antibiotics or probiotics within four weeks before the study enrollment; 5. Patients with a known sensitivity or allergy to any drugs used in this study; 6. Patients who were unwilling to participate voluntarily in the study.
Sample size
A simple random probability sampling technique was practiced to allocate the patients to the investigational and control groups. The sample size was calculated at a 5% level of significance and a confidence interval of 95%. The sample size was calculated by the following formula: N= Z2pq/ e2. Using the formula, sample size was n=(1.96)2x (0.5) x (0.5)/ (0.05)2 = 384. However, our sample size was kept at 100 due to financial constraints and the COVID-19 pandemic (Figure 1).
Figure 1. Schematic diagram showing the methodology of this study.
Image credit: Sismita Sinha; with the premium version of BioRender [114] with the license number HM26K5BNK0
Sampling method
Simple random sampling was done from patients who underwent endoscopy of the upper gastrointestinal tract (UGIT) and tested positive for RUT. We used simple random sampling methods to ensure an unbiased representation of patients who undergo endoscopy of the upper UGIT and test positive for the RUT.
Data collection and intervention
Questionnaire
Information on clinical history and adverse effects during triple therapy was collected using a questionnaire administered by the investigator.
Intervention
Patients meeting the inclusion criteria were randomly assigned into two different therapeutic intervention programs: (i) Group A (experimental group) (n = 50), which was given amoxicillin (1000 mg, two times a day), levofloxacin (500 mg once daily), rabeprazole (20 mg two times a day) with probiotics Lactobacillus plantarum LA 301, Lactobacillus salivarius LA 302 (once daily) for 14 days, and (ii) Group B (control group) (n = 50), which was given amoxicillin (1000 mg, two times a day), levofloxacin (500 mg once daily), and rabeprazole (20 mg two times a day for 14 days).
Follow-up was done six weeks after therapy completion. This follow-up time point allowed for assessing ADRs and abrogation rates of H. pylori following the intervention. During the follow-up, a second review endoscopy of UGIT with RUT was conducted to determine the status of H. pylori infection. During the follow-up, any ADRs experienced by the patients during the triple therapy were recorded.
Statistical analysis
No participants missed the follow-up after the intervention; thus, intention-to-treat analysis was introduced here. An independent sample t-test for continuous observation and a chi-square test for categorical observation were used to assess the demographic features. A multiple logistics regression model was used to estimate the risk of adverse effects in triple therapy with probiotics group compared to triple therapy without probiotics. The independent factors influencing the model >5% were used as covariates in the final regression model, i.e., age, sex, and history of hypertension, multiple comorbidities, smoking, and taking NSAIDs. Longitudinal multilevel mixed-effect models were used to assess the overall change in RUT after triple therapy with probiotics intervention. Models were adjusted with covariates (age, sex, history of morbidities, and smoking history) that affected the model R2 by 5% or more in the best-fitted regression model; additionally, time was used as a covariate to reduce the multicollinearity. For statistical analysis, Stata Statistical Software: Release 15 (StataCorp LLC, College Station, Texas, United States) and GraphPad Prism version 8.3.0 (Insightful Science, LLC, San Diego, California, United States) were used for graphical presentation. A p-value of p<0.05 was considered as significant.
Results
The participants in Group A (probiotics group) had an average age of 37.4 years, with a standard deviation of 13.4 years, while those in Group B (non-probiotics control group) had an average age of 38.3 years, with a standard deviation of 14.1 years. Regarding gender distribution, 52% of the probiotics group were male, and 48% were female. In the non-probiotics group, 54% were male, and 46% were female (Table 1).
Table 1. Demographic characteristics of the study participants .
Data have been presented as n (%) except for age, which has been presented as mean±SD. An Independent sample t-test for continuous data and chi-square for categorical data was used to estimate the p-value. An unpaired t-test was conducted.
NSAID: non-steroidal anti-inflammatory drugs
| Variables | Triple therapy with probiotics (N=50) | Triple therapy without probiotics (N=50) | p-value |
| Age (years), mean±SD | 37.4±13.4 | 38.3±14.1 | 0.744 |
| Sex, n (%) | |||
| Male | 26 (52.0%) | 27 (54.0%) | 0.841 |
| Female | 24 (48.0%) | 23 (46.0%) | |
| History of comorbidities, n (%) | |||
| Diabetes | 6 (12.0%) | 7 (14.0%) | 0.766 |
| Hypertension | 13 (26.0%) | 7 (14.0%) | 0.134 |
| Others | 4 (8.00%) | 2 (4.00%) | 0.400 |
| Multiple comorbidities | 1 (2.00%) | 2 (4.00%) | 0.558 |
| Nil | 33 (66.0%) | 38 (76.0%) | 0.271 |
| Personal history, n (%) | |||
| Smoking | 11 (22.0%) | 9 (18.0%) | 0.617 |
| Alcohol | 2 (4.0%) | 1 (2.0%) | 0.558 |
| Tea/coffee | 18 (36.0%) | 16 (32.0%) | 0.673 |
| Betel nut | 8 (16.0%) | 15 (30.0%) | 0.096 |
| Drug history, n (%) | |||
| NSAIDs | 7 (14.0%) | 8 (16.0%) | 0.779 |
The breakdown of comorbidities in both groups showed a nearly identical distribution, covering conditions such as diabetes, hypertension, others, and multiple comorbidities. The category 'Nill' indicated participants with no comorbidities. Among the participants in the probiotics group, 22% were smokers, while in the non-probiotics group, 18% were smokers. Additionally, 36% of the probiotics group reported consuming tea or coffee, compared to 32% in the non-probiotics group. Regarding betel nut consumption, 30% of participants in the non-probiotics group reported its intake, whereas only 16% of participants from the probiotics group had a history of betel nut consumption. The history of NSAIDs showed a similar distribution in both groups (Table 1).
In the group receiving triple therapy with probiotics (N=50), 50% experienced abdominal pain, 16% had anorexia, 34% reported nausea, 8% had episodes of vomiting, 56% suffered from dyspepsia, and 56% had constipation. In the group receiving triple therapy without probiotics (N=50), 48% experienced abdominal pain, 22% had anorexia, 20% reported nausea, 4% had episodes of vomiting, 42% suffered from dyspepsia, and 44% had constipation (Table 2).
Table 2. History of present illness of the study subjects.
Data have been presented as n (%). A chi-square test was conducted.
| Triple therapy with probiotics (N=50), n (%) | Triple therapy without probiotics (N=50), n (%) | p-value | |
| Abdominal pain | 25 (50.0) | 24 (48.0) | 0.841 |
| Anorexia | 8 (16.0) | 11 (22.0) | 0.444 |
| Nausea | 17 (34.0) | 10 (20.0) | 0.115 |
| Vomiting | 4 (8.0) | 2 (4.0) | 0.400 |
| Dyspepsia | 28 (56.0) | 21 (42.0) | 0.161 |
| Constipation | 27 (56.0) | 22 (44.0) | 0.317 |
The history of past illnesses, including hematemesis and melena, exhibited no significant difference between the two groups (p=1.000). Personal habits such as smoking, alcohol consumption, tea/coffee consumption, betel nut use, and consumption of spicy and oily food also did not significantly differ between the two groups (p>0.05). The family history of PUD was reported by 32% of subjects in the probiotics group and 28% in the non-probiotics group, with no significant difference (p=0.663). The history of taking NSAIDs was also similar between the two groups (p=0.779) (Table 3).
Table 3. History of past illness, personal history, family history of PUD, and history of drugs.
Data have been presented as n (%). A chi-square test was conducted.
PUD: peptic ulcer disease
| Triple therapy with probiotics (N=50), n (%) | Triple therapy without probiotics (N=50), n (%) | p-value | |
| History of past Illness | |||
| Hematemesis | 0 (0.0) | 1 (2.0) | 1.000 |
| Melena | 4 (8.0) | 5 (10.0) | 1.000 |
| Personal history | |||
| Smoking | 11 (22.0) | 9 (18.0) | 0.617 |
| Alcohol | 2 (4.0) | 1 (2.0) | 1.000 |
| Tea/coffee | 18 (36.0) | 16 (32.0) | 0.673 |
| Betel nut | 8 (16.0) | 15 (30.0) | 0.096 |
| Spicy and oily food | 18 (36.0) | 19 (38.0) | 0.836 |
| Family history of PUD | 16 (32.0) | 14 (28.0) | 0.663 |
| History of taking NSAID | 7 (14.0) | 8 (16.0) | 0.779 |
Table 4 categorizes findings into reflux esophagitis, erosive gastritis, gastric ulcer, and duodenal ulcer, providing the respective percentages within each group. The p-values, derived from the Chi-Square test, are included to assess the statistical significance of differences in endoscopic findings between the two treatment groups. However, the results indicate no statistically significant variations in the prevalence of reflux esophagitis, erosive gastritis, gastric ulcer, or duodenal ulcer between the groups, as all the p-values surpass the conventional significance threshold of 0.05.
Table 4. Findings of initial endoscopy.
The p-value was estimated using the Chi-Square test.
| Triple therapy with probiotics (N=50), n (%) | Triple therapy without probiotics (N=50), n (%) | p-value | |
| Reflux esophagitis | 25 (50.0) | 24 (48.0) | 0.952 |
| Erosive gastritis | 17 (34.0) | 18 (36.0) | 0.822 |
| Gastric ulcer | 5 (10.0) | 6 (12.0) | 0.911 |
| Duodenal ulcer | 3 (6.0) | 2 (4.0) | 0.871 |
When we assessed the endoscopy findings during the follow-up, it was detected that there was no significant variation between the two groups (Table 5).
Table 5. Findings of the review endoscopy during follow-up.
The P-value was estimated by using the Chi-Square test.
| Triple therapy with probiotics (N=50), n (%) | Triple therapy without probiotics (N=50), n (%) | p-value | |
| Reflux esophagitis | 10 (20.0) | 11 (22.0) | 0.910 |
| Erosive gastritis | 21 (42.0) | 24 (48.0) | 0.899 |
| Gastric ulcer | 8 (16.0) | 5 (10.0) | 0.523 |
| Duodenal ulcer | 2 (4.0) | 2 (4.0) | 0.999 |
| Normal | 9 (18.0) | 8 (16.0) | 0.912 |
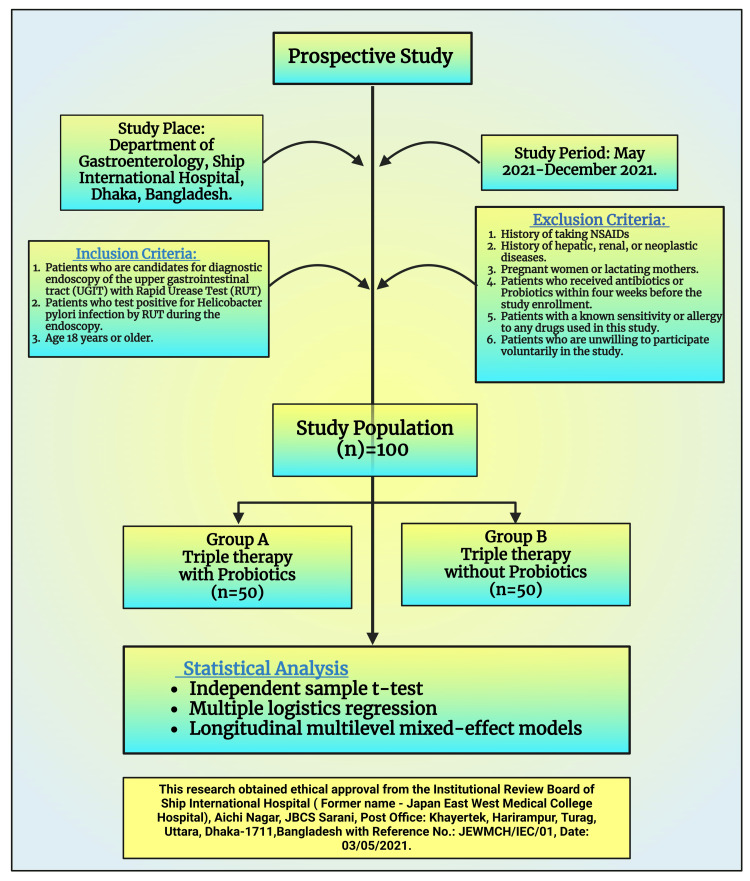
Longitudinal multilevel was used to assess the overall change in RUT after triple therapy with probiotics intervention. The odds of RUT becoming negative from positive were 2.06 times higher (95%CI 0.95-3.22, p=0.054) in the probiotics group compared to the non-probiotics control group (Figure 2). When the association was checked with a non-parametric approach, ADRs were significantly higher in the non-probiotics group (p=0.045) than in the probiotics group (Table 6).
Table 6. Adverse Effects of Triple Therapy on the Study Subjects (N=100).
Notes: A Chi-Square Test was Conducted.
| Adverse Drug Reactions of Triple Therapy | Triple Therapy with Probiotics (n=50) | Triple Therapy without Probiotics (n=50) | p-value |
| Yes | 19 (38.0) | 29 (58.0) | 0.045 |
| No | 31 (62.0) | 21 (42.0) |
Figure 2. The odds ratio of RUT risk becoming negative in the supplementation group compared to the group without probiotics.
A multilevel mixed-effect model was used to analyze the OR of RUT. The analysis considered potential confounders that affected the model by >5% (age, sex, history of morbidities, and smoking history).
RUT: rapid urease test
Image Credit: Md. Ahsanul Haq
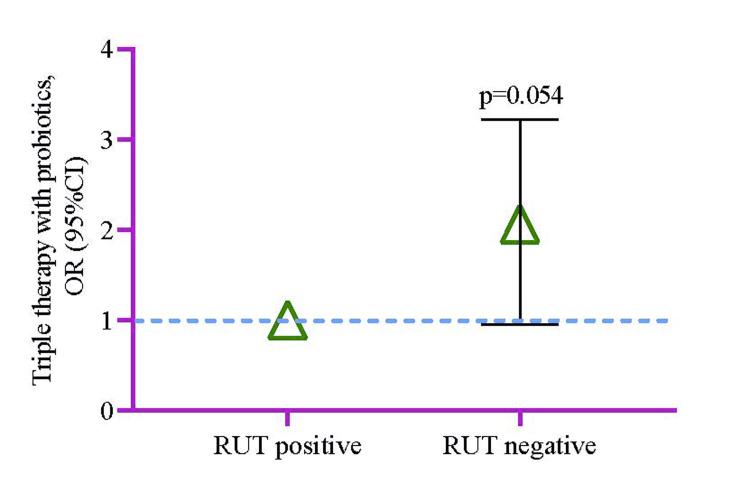
When the history of ADRs in the probiotics group was compared to the non-probiotics control group by the logistic regression model, it was observed that the probiotics group had a lower risk of having detrimental effects by 0.54 times (95%CI 0.19-0.84, p=0.032) (Figure 3).
Figure 3. Risk of having adverse effects of triple therapy in the probiotics group compared to the non-probiotic group.
The logistic regression model was used to estimate the p-value, and the regression model was adjusted by age, sex, history of hypertension, history of multiple comorbidities, history of smoking, and history of taking NSAIDs.
NSAID: non-steroidal anti-inflammatory drugs
Image Credit: Md. Ahsanul Haque.
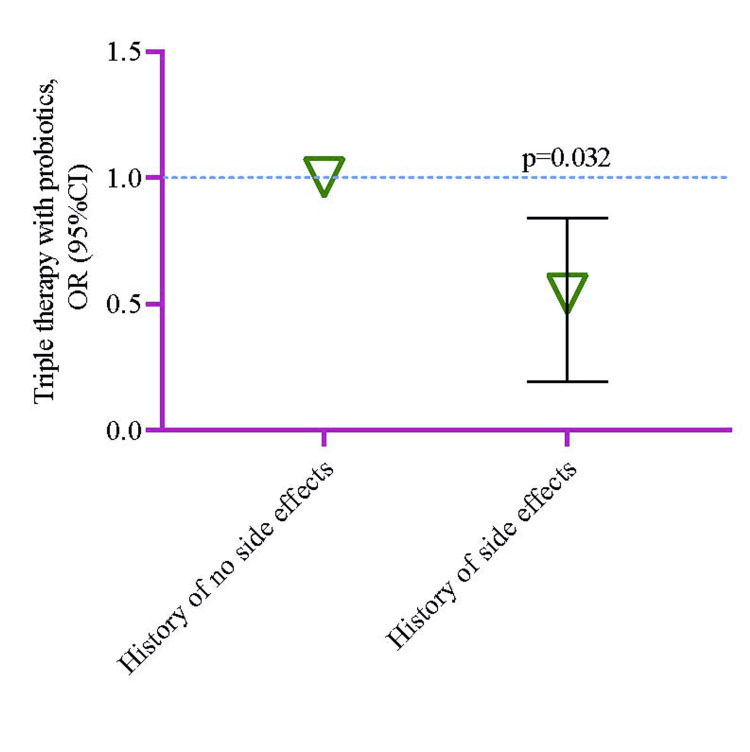
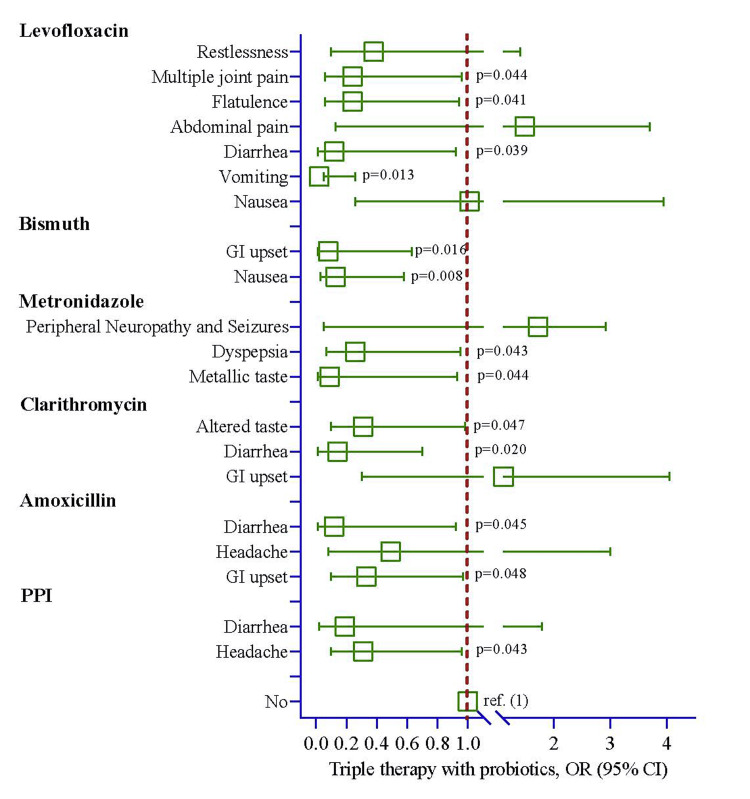
Upon stratifying the risk of ADRs based on antibiotic usage and employing a logistic regression model, the analysis revealed significantly lower odds of experiencing headaches in the group using PPI, antibiotics with probiotics, with an OR of 0.31 (95%CI 0.10-0.96, p=0.043) as depicted in Table 7 and Figure 4. Furthermore, in the amoxicillin group, the likelihood of GI upset and diarrhea was reduced by 0.33 and 0.12 times, respectively, in the presence of probiotics. For those study participants taking clarithromycin, the risk of diarrhea was 0.14 times lower in the group with probiotics compared to the group without probiotics. The analysis also indicated a 0.26 times lower risk of dyspepsia in the group with probiotics for individuals using metronidazole. Bismuth users exhibited a decreased risk of nausea and GI upset by 0.13 and 0.08 times, respectively. Moreover, probiotic users demonstrated lower odds of experiencing vomiting (OR=0.02), diarrhea (OR=0.12), flatulence (OR=0.24), and multiple joint pain (OR=0.24) when using levofloxacin as antibiotics (Table 7 and Figure 4).
Table 7. Risk of adverse effects of the antibiotics in triple therapy with probiotics compared to triple therapy without probiotics .
Multinomial logistic regression was used to estimate the p-value, and the regression model was adjusted by age (category), sex (type, male and female), history of hypertension (category), history of multiple comorbidities (categorical), smoking history (categorical), history of taking NSAID (categorical).
NSAID: non-steroidal anti-inflammatory drug; PPI: proton pump inhibitor
| Drug | Adverse Drug Reactions | Triple therapy without probiotics (N=50) | Triple therapy with probiotics (N=50), OR (95%CI) | p-value |
| PPI | Headache | 1 | 0.31 (0.10, 0.96) | 0.043 |
| Diarrhea | 1 | 0.19 (0.02, 1.79) | 0.145 | |
| Amoxicillin | GI upset | 1 | 0.33 (0.10, 0.97) | 0.048 |
| Headache | 1 | 0.49 (0.08, 3.00) | 0.437 | |
| Diarrhea | 1 | 0.12 (0.01, 0.92) | 0.045 | |
| Clarithromycin | GI upset | 1 | 1.11 (0.30, 4.05) | 0.882 |
| Diarrhea | 1 | 0.14 (0.01, 0.70) | 0.020 | |
| Altered taste | 1 | 0.31 (0.10, 0.98) | 0.047 | |
| Metronidazole | Metallic taste | 1 | 0.09 (0.01, 0.93) | 0.044 |
| Dyspepsia | 1 | 0.26 (0.07, 0.95) | 0.043 | |
| Peripheral neuropathy and seizures | 1 | 1.72 (0.05, 2.92) | 0.766 | |
| Bismuth | Darkening of the tongue and stool | 1 | - | |
| Nausea | 1 | 0.13 (0.03, 0.58) | 0.008 | |
| GI upset | 1 | 0.08 (0.01, 0.63) | 0.016 | |
| Levofloxacin | Nausea | 1 | 1.01 (0.26, 3.94) | 0.983 |
| Vomiting | 1 | 0.02 (0.05, 0.26) | 0.013 | |
| Diarrhea | 1 | 0.12 (0.01, 0.92) | 0.039 | |
| Abdominal pain | 1 | 1.48 (0.13, 6.69) | 0.754 | |
| Flatulence | 1 | 0.24 (0.06, 0.94) | 0.041 | |
| Multiple joint pain | 1 | 0.24 (0.06, 0.96) | 0.044 | |
| Restlessness | 1 | 0.38 (0.10, 1.40) | 0.145 |
Figure 4. Risk of adverse effects in triple therapy with probiotics group compared to triple medications excluding probiotics. No adverse impact was used as reference (1) for all measured events.
The logistic regression model was used to estimate the p-value, and the regression model was adjusted by age, sex, history of hypertension, history of multiple comorbidities, history of smoking, and history of taking NSAIDs.
NSAID: non-steroidal anti-inflammatory drug; PPI: proton pump inhibitor
Image Credit: Md. Ahsanul Haq
Discussion
Table 8 gives details of selected studies from the literature regarding H. Pylori eradication and dysbiosis. The STT for eliminating H. pylori often upsets intestinal microbiota, especially the antimicrobial component of the therapy [113-117]. Additionally, it causes vitamin (vitamin A, B, C, K, α-tocopherol, vitamin B12, and folic acid) insufficiency in the human body [116-118]. Various studies revealed that H. pylori extinction treatment regimens have raised the possibility of several pathogenic microbes such as Streptococcus, Klebsiella, and Shigella [119,120]. These pathogens instigate chronic inflammation and promote the synthesis of genotoxins or carcinogenic metabolites [121-123]. This chronic inflammation of stomach-related gastric dysbiosis bed frequently causes DNA alteration and upholds precancerous ulceration [28,124-128]. H. pylori-related gastric precancerous situation ultimately turned to atrophic gastritis and finally carcinoma with a very high mortality rate [129,130]. Conversely, multiple studies reported that administering probiotics before and after eradication therapy for H. pylori rapidly reinstates intestinal microbiota (Figure 5), especially Bacillus and Lactic acid bacteria [117,131,132]. Another issue is antimicrobial resistance regarding H. pylori management that leads to pharmacological intervention failure throughout the globe [66,80,133-136].
Table 8. Selected randomized control trials, systematic review, review, and meta-analysis regarding H. Pylori eradication and dysbiosis.
Keywords were “H. Pylori,” “Eradication,” and “Dysbiosis.” Filter Applied: Randomized Control Trials, Systematic Review, Review, and Meta-Analysis. Indexed in PubMed.
H. pylori: Helicobacter pylori
| Type of Study | Authors, year, reference | Background | Result | Conclusion |
| Randomized Controlled Trials | He et al., 2022 [131] | The use of antibiotics to treat H. pylori in the gut causes derangement in microbiota. Probiotics reduce the adverse effects of antibiotics, but their impact on allaying microbiota is not well-established | The elimination rate of H. Pylori was almost similar in the placebo and probiotic groups. Normal commensals suppressed following eradication were replaced by probiotics, and microbiota was progressively restored within two weeks. Adverse events were also less in the probiotic cluster. | Concurrent use of probiotics in H. pylori elimination mitigates unwanted effects and restores normal commensals in the gut and oral cavity. |
| Yuan et al., 2021 [137] | The effect of probiotics on gut microbiota in both H. pylori-positive and H. pylori-negative patients is not apparent. | Clusters treated with quadruple therapy and probiotics had increased levels of commensals, e.g., Bifidobacterium and lactobacillus. Probiotics alone were not successful in the elimination of H. pylori. Only quadruple therapy caused significant disruption of the gut microbiome. | H. pylori elimination causes alleviation of normal flora in GIT. This disruption can be minimized if probiotics are added to the treatment protocol. | |
| Chen et al., 2021 [138] | Disruption of gut commensals during H pylori. The use of probiotics can alleviate H.pylori treatment. However, data on the role of probiotics alone in suppressing Helicobacter infection is scarce. | H. pylori load was reduced with Lactobacillus acidophilus and L. rhamnosus, but the gut microbiome did not exhibit any significant variation. | Specific probiotics may mitigate H. pylori load without altering the gut microbiota. | |
| Dore et al., 2022 [139] | Treatment of H. pylori is always accompanied by the development of adverse effects, primarily on the gut microbiome. The use of probiotics can reduce this side effect. | There was no significant difference in the eradication rate of H. pylori. Gut microbiota was similarly deranged by L. reuteri and bismuth and restored after 30-40 days. | L.reuteri can be used in patients who cannot tolerate bismuth along with low-dose quadruple therapy, but disturbance of gut microbiome was unavoidable | |
| Guillemard et al., 2021 [140] | Alteration of gut microbiota during H. pylori treatment is now well established. This derangement can be mitigated using probiotics, but it is poorly documented. A fermented milk product containing several strains was assessed in this research. | The test group did not have a significant impact on GIT symptoms. Still, beta diversity was less, the number of pathogenic bacteria was reduced, and that of short-chain fatty acid (SCFA) was increased. | This seven-strain fermented milk can be helpful to ensure brisk retrieval of the gut microbiome after H. pylori treatment. | |
| Yakovenko et al., 2021 [141] | H. pylori removal by quadruple therapy containing bismuth also causes various unwanted GI symptoms. The addition of probiotics may alleviate these adverse events | When a Bifiform capsule was added to the regimen, disruption of colonic microbiota was less, plasma cells were increased, and IgA level was stable. | Incorporation of probiotics in the treatment regimen can reduce side effects and increase the immunity of the gut. | |
| Systematic Review and Meta-analysis | Guo et al., 2022 [142] | Alteration of gut microbiome is noticed during H. Pylori elimination. Whether gut microbiota is restored in its pre-infection state after treatment is not documented. | Successful elimination of H. pylori was noticed in both quadruple and triple therapy. Alpha diversity and alpha and beta diversity were documented in quadruple and triple treatment, respectively. | H. pylori, the available regimen can successfully eliminate pylori, but a conclusion on restoration of gut commensals could not be drawn. |
| Review | Suzuki et al., 2022 [143] | H. pylori is responsible for gastric inflammation, peptic ulcer, and stomach cancer. Treatment protocols show diversity in different provinces of the world. | Based on present evidence, quadruple without bismuth and vonoprazan-based triple therapy was 90% effective but still caused a disturbance in gut microbiota. | A less costly, more straightforward regimen that will not cause the risk of dysbiosis is needed. Dual therapy with amoxicillin and vonoprazan may be considered. |
Figure 5. Beneficial effects of adding probiotics to the standard triple therapy on patients with H. pylori infection.
H. pylori: Helicobacter pylori
Image credit: Rahnuma Ahmad; with the premium version of BioRender [114] with license number CH26GB4NBY.
Tables 9-10 give the list of studies reviewed on H. Pylori resistance in Bangladesh and low- and middle-income countries, respectively.
Table 9. Selected studies indexed in PubMed on Helicobacter pylori resistance in Bangladesh.
Keywords were “Helicobacter Pylori,” Resistance,” and “Bangladesh.”
| Study (Authors, year, reference) | Background | Result | Conclusion |
| Fauzia et al., 2023 [144] | Due to the development of resistance to usual antibiotics, treatment against H. pylori has become less effective. Clinical detection is formidable. So, understanding the fundamental mechanism is critical to fight against resistance. | Resistance was highest against clarithromycin, followed by levofloxacin, amoxicillin, and metronidazole. Multi-drug resistance was observed in most of the cases. | These findings may facilitate fast decisions on the susceptibility of antibiotics against H. pylori. |
| Aftab et al., 2016 [74] | Knowledge of antibiotic resistance during H.pylori treatment is crucial, but the unavailability of current data makes treatment arduous. | Metronidazole and clarithromycin were the topmost resistant antibiotics, and levofloxacin is rising. Lower resistance was documented in the case of tetracycline and amoxicillin. | In Bangladesh, resistance against metronidazole, clarithromycin, and levofloxacine were highest, making the triple therapy futile. |
| Shrestha et al., 2023 [145] | Antimicrobial resistance against H. pylori has become common in South Asia, but data is limited. | The top five antibiotics found to be resistant were Metronidazole, levofloxacin, clarithromycin, amoxicillin, and tetracycline. India, Pakistan, and Bangladesh were the most prevalent countries regarding resistance. | Antibiotic resistance is on the rise among South Asian countries, and most include commonly used drugs. |
| Miftahussurur et al., 2019 [146] | Antibiotic resistance against H. pylori is highly prevalent in Bangladesh and Nepal. Finding an alternative protocol is now a time-demanding issue. For that, knowledge of the resistance rate and its mechanism is crucial. | Rifabutin, furazolidone, and sitafloxacin were highly susceptible, and rifaximin was resistant in both countries. Samples from Bangladesh showed higher resistance against garenoxacin. Mutations in A87, D91, and gyrA were responsible for developing resistance. | Furazolidone and sitafloxacin can be added to the treatment regimen in resistant cases. Rifabutin should be used. |
| Qaria et al., 2018 [147] | H. pylori cell wall contains Cholesteryl glucosides (CGs), which makes the bacteria pathogenic and virulent. Deleting the gene responsible for this enzyme formation may cause the bacteria to be vulnerable to available antibiotics. | Morphological alterations were perceived following the deletion of the responsible gene, making the bacteria more vulnerable to commonly used antibiotics. | The absence of CGs converts resistant H. pylori to a susceptible one due to cell wall modification. |
| Fauzia et al. , 2023 [148] | H. pylori is a highly biofilm-forming organism that ensures protection against antibiotics. Evaluation of the underlying mechanism of biofilm formation is, therefore, very crucial | Most of the strains were found to be low biofilm forms. G160S, N156K, and A223V mutations among the nucleotide polymorphisms (SNP) were responsible for high biofilm formation in Bangladeshi people. | SNPs have a vital role in biofilm construction, and the method applied in the research can be used to detect mutations. |
| Reza et al., 2023 [149] | Effective treatment of H. Pylori infection is vital as it may result in gastric carcinoma. Due to the development of resistance, antimicrobials have become less potent. Gene therapy can be an efficient alternative. | Specifically designed siRNA were successful in silencing the sequence-specific gene. | These designed siRNAs can be incorporated in treating H. pylori causing gastric carcinoma. |
| Banatvala et al., 1994 [150] | Antibiotic resistance, especially of metronidazole, is well reported in people of developing countries, but data is scarce on the transfer of these resistant varieties through migrants. | The resistant variant was higher in migrants, with the majority from Bangladesh. Women born in the United Kingdom and previous treatment with nitroimidazole, metronidazole, and tinidazole were other risk factors. | Migrants affected by H. pylori from developing countries will likely have resistant varieties. |
| Khan et al., 2004 [151] | Treatment against H. pylori often fails due to resistance against clarithromycin. Alteration in genomic sequence is responsible for developing resistance, but data is limited in cases from Bangladesh. | A T-to-C transition mutation at position 2182 was found in every clarithromycin-resistant case that was analyzed. | Resistance against clarithromycin develops due to a T-to-C transition mutation at position 2182 of the H. pylori genome. |
| Nahar et al., 2004 [73] | Eliminating H. pylori often fails due to antimicrobial resistance against commonly used antibiotics, but data is scarce in Bangladesh. | Metronidazole, followed by tetracycline, clarithromycin, and amoxicillin, were primarily resistant. | Higher resistance against common antibiotics in Bangladesh proves the need for extended monitoring. |
| Qumar et al., 2021 [152] | H. pylori infection is mostly prevalent in developing countries, and its treatment has become arduous due to the emergence of resistant varieties. The genotype of the predominant variety is not well documented in Bangladesh. | Isolated strains showed two separate varieties, e.g., HpAsia2 and HpEurope, where the prior one was more virulent. | The genotype of the bacteria, as well as environmental and host factors, are crucial to determine the clinical prognosis. |
| Khan et al. , 2008 [153] | Mutations in the 16S rRNA gene are thought to be responsible for clarithromycin-resistant H. pylori infection cases. Data from Bangladesh were limited. | In the tetracycline-resistant variety, none showed a mutation in the 16S rRNA gene. | Tetracycline resistance may also appear due to other causes rather than RNA mutation. |
| Rahman et al., 2009 [154] | Cytotoxin-associated gene A (CagA) of H. pylori is a risk factor for developing coronary heart disease (CHD). Data on this association among Asian Indians are sparse. | H. pylori-positive cases with or without CHD had lower levels of HDL, while fasting plasma glucose was markedly increased in HP+ve patients with CHD. Insulin secretory dysfunction (ISD) was higher in all infected cases compared to control groups. | H. pylori infection is a risk factor for developing CHD and Diabetes mellitus. |
| Akash et al. , 2023 [155] | Inflammation of gastric mucosa by H. pylori may lead to the development of gastric cancer. The rising tendency of antimicrobial resistance has further increased the risk, prompting the need for alternative treatment. | Among the eight phytocompounds, Sarsasapogenin and Diosgenin showed the highest binding affinity, and the rest showed superiority over mitomycin. | Gastric carcinoma, induced by H. pylori, may respond to these natural compounds, but further studies are required. |
| Rimbara et al., 2007 [156] | Nine genomic mutations were thought to be responsible for the development of clarithromycin resistance of H. pylori, but documents from Japan are not well-concluded | An adenine → guanine transition at position 2143 (A2143G) or 2142 (A2142G) was detected in every clarithromycin-resistant case. | In Japan, mutations in positions 2142 and 2143 were associated with resistance to clarithromycin. |
Table 10. Selected consequential papers on Helicobacter pylori resistance in low and middle-income countries.
Keywords were “Helicobacter Pylori,” “Resistance,” and “Low and middle-income countries.” Filters Applied: Free full text, Meta-Analysis, Randomized Controlled Trial, Review, Systematic Review in the last five years. Indexed in PubMed.
| Type of Study | Authors, year, reference | Background | Result | Conclusion |
| Review | Qiu et al., 2021 [157] | Noninvasive investigations, e.g., urea breath test, are preferable for detecting H. pylori. Due to the high cost, lack of expertise, and non-compliance of child patients, detecting bacteria from stool samples may be a suitable alternative. | Commercially available kits for stool antigen tests (SAT) showed promising results for mass screening. Immunoassay also demonstrated higher specificity and sensitivity. PCR is also advantageous but costly. | Identifying H. pylori from stool samples can be considered an efficient method, although it is not free of disadvantages. |
| Review | Nguyen et al., 2023 [158] | Children infected by H. pylori may remain symptom-free for a longer period but are at risk for the development of various gastrointestinal disorders later. Comprehensive data are lacking on childhood infection. | The prevalence of H. pylori is still high in the pediatric age group. Lack of symptoms makes diagnosis difficult, followed by the development of antibiotic resistance. | PCR-based diagnosis and detection of susceptibility are essential for successful eradication. During prevention, host and environmental factors should be taken into consideration. |
| Review | Mestre et al., 2022 [97] | Incorporating probiotics with standard treatment protocol for eradicating H. pylori exerts better results and mitigates side effects. | Probiotics alone contribute to the restoration of the gut microbiome only, but when added to the treatment regimen, the treatment success rate is higher, and adverse events are less. | Data from the Asian population has revealed the efficacy of probiotics, but more studies are needed from other parts of the world. |
| Review | Garrido-Treviño et al., 2022 [159] | Treatment of H. pylori infection is often given empirically, which has played a vital role in developing resistant strains. Noninvasive tests such as molecular methods may alleviate this disadvantage. | Higher sensitivity and specificity were observed in molecular methods in detecting H. pylori. | New-generation molecular methods can help mitigate antimicrobial resistance. |
| Systematic review and meta-analysis | Wang et al., 2023 [160] | Antibiotic-resistant varieties constantly disrupt the elimination of H. pylori. Data on primary antibiotic resistance is not well documented in China. | Resistance was found to be higher in the case of metronidazole, clarithromycin, and levofloxacin, followed by amoxicillin. | Commonly used antibiotics showing resistance may result in treatment failure and require rapid action. |
| Systematic Review | Rojas et al., 2021 [161] | There are several diagnostic methods for H. pylori infection, but they differ in cost and effectiveness. | Rapid, noninvasive methods are now replacing invasive tests. Detection of antibiotic resistance by various methods is also being included at a higher rate, but studies on the effectiveness of different techniques are still limited. | Using diagnostic methods instead of empirical therapy may lessen the economic burden. |
| Systematic Review | Sukri et al., 2021 [162] | Antimicrobial resistance, especially against clarithromycin, is a significant obstacle in eradicating H. pylori. Alternative treatment is now needed, especially for people with limited resources. | Data from different Southeast Asian countries showed higher resistance to metronidazole, clarithromycin, and levofloxacin. Antimicrobial peptides (AMP) were found to be effective in resistant cases. | AMPs can be incorporated into treatment protocol to combat resistant variety. |
| Randomized Control Trial | Vilaichone et al., 2020 [163] | In Bhutan, gastric cancer induced by H. pylori infection is the commonest among malignancies. Therefore, a cost-effective method for H. pylori elimination is critical in reducing this malignancy. | Fourteen-day regimens showed a higher eradication rate; females and patients over 40 were more responsive. | A 14-day regimen can be more effective in economically constrained areas like Bhutan. |
| Meta-Analysis | Fontes et al., 2019 [164] | As antibiotic resistance against H. pylori is on the rise, alternative therapy such as N-acetylcysteine (NAC) is now very important, especially for regions with limited sources. | The quality of methodology was poor in the included studies. However, NAC did not show significant efficacy over conventional treatment. | A definitive conclusion could not be drawn on the efficacy of NAG regarding H. pylori treatment. |
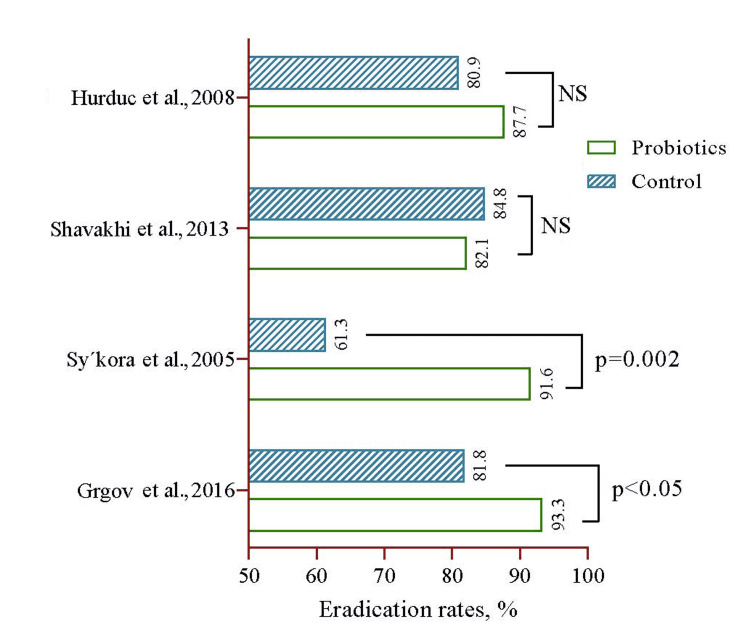
Additionally, Grgov et al. [165] and Sýkora et al. [166], in their randomized control trials, revealed that probiotic treatment led to significantly higher eradication rates of 93.3% and 91.6%, respectively, compared to the control groups. On the contrary, Shavakhi et al. [167] and Hurduc et al. [168] observed no significant difference between the two treatment groups (Figure 6).
Figure 6. Selected consequential papers showing the benefits of adding probiotics with standard therapy for Helicobacter pylori eradication.
Notes: Keywords were “Benefits,” “Probiotics,” “Helicobacter pylori,” “Eradication,” and “Triple therapy.” Filters Applied: Randomized Controlled Trials. Indexed in PubMed.
Image Credit: Md. Ahsanul Haq
One earlier study reported that usual ADRs observed in the combined triple therapy + probiotics group were dyspepsia (4.4%), nausea and/or vomiting (2.9%), dry mouth (2.6%), diarrhea (1.5%), and abdominal pain (1.1%) [169]. However, the current study uncovered that among patients who received triple therapy with probiotics, common ADRs included abdominal pain, anorexia, nausea, vomiting, and dyspepsia. So, our study's findings showed that the types of ADRs and the incidence rate were similar. Furthermore, patients receiving triple therapy without probiotics exhibited higher levels of common similar ADRs (Table 6). Akcam et al. reported identical observations [170]. Furthermore, Lü et al. reported in their meta-analysis comprising 13 randomized controlled trials that adding probiotics throughout H. pylori eradication therapy reduces ADRs [171].
No significant differences were observed in the initial and follow-up endoscopic findings regarding reflux esophagitis, erosive gastritis, gastric ulcer, and duodenal ulcer between the experimental (STT + probiotics) and control (only STT) group in the current study (Table 4) and (Table 5). One earlier study reported that H pylori extermination remedial measures escalate the possibility of reflux esophagitis, which is unrelated to previous records of esophagitis. Additionally, no considerable consequence can be comprehended regarding reflux-associated issues [172]. One Japanese study reported that nearly 10% of H. pylori-positive cases develop reflux esophagitis after pharmacological intervention and successful eradication. This observation was principally noticed among cases with severe reflux-related symptoms before medical intervention. Meanwhile, after efficacious elimination, patients with H. pylori had definite improvement regarding peptic-ulcer-related manifestation. Nonetheless, the possibility of the reflux esophagitis evolvement remains [173].
Model analysis revealed positive clinical outcomes concerning H. pylori biomarker (RUT) after triple therapy with probiotics intervention. The odds of RUT becoming negative from positive were more than two-folds higher among the experimental group (SST + probiotics) in comparison to the control group (only STT) (Figure 2). One meta-analysis by Lu et al. comprising 21 RCTs reported that additional probiotics with STT do not show clinically better outcomes regarding H. pylori's extinction frequency when equated to the inactive medicinal agents [174]. Akcam et al. reported similarly that no substantial confirmation was obtained about the abolition of H. pylori and the minimization of ADRs [170]. Additionally, two more RCTs revealed no statistically significant differences observed between experimental (STT + probiotics) and control (only STT) [167,168].
In contrast, multiple studies reported that adding probiotics with standard therapy improves the degree of H. pylori eradication and diminishes antimicrobial-induced ADRs, especially gastrointestinal issues [170,175,176]. Two more RCTs revealed that adding probiotic treatment led to considerably higher eradication rates [165,166]. However, when the association was checked with a non-parametric approach, ADRs were significantly higher in the non-probiotics group than in the probiotics group (Table 6). Multiple earlier research published papers reported that the addition of probiotics with standard therapy improves the H. pylori eradication rate and diminishes ADRs [97,177]. One meta-analysis comprising 13 RCTs and 2306 patients revealed that adding probiotics with H. pylori antagonist’s regimen improves extinction percentages, parallelly lowers ADRs, and relieves most PUD-allied clinical indicators [171]. Wang et al. reported that traditional triple therapy with probiotics, especially Bifidobacterium-Lactobacillus and Bifidobacterium-Lactobacillus-Saccharomyces therapy recuperates abolition proportions and reduces ADRs [96]. Another RCT by Hauser et al. revealed that supplementation of probiotics with conventional triple therapy improves pharmacodynamic properties and decreases ADRs and patient compliance equally noticeably [178].
The logistic regression model analysis observed that the experimental group (probiotics) had a lesser risk of adverse effects when compared to the control group (without probiotics) regarding the risk of a history of ADRs in traditional triple therapy (Figure 3). Multiple studies reported that socio-anthropological differences between individual cases, history of PUD and non-communicable chronic disorders, tobacco and alcohol consumption, and the existence of hereditary issues affect extermination H. pylori treatment outcomes [95,179,180]. Gebeyehu et al. reported that ADRs while receiving conventional triple therapy were dependent on the following changeable features: history of PUD over 21 days, location of dwelling, body mass index (BMI), consumption of alcohol, and long intervals between meals causing abdominal pain, when conducting bivariate and multiple logistic regression analysis [88].
The logistic regression model analysis revealed significantly lower odds of experiencing headaches in the group using PPI antibiotics with probiotics (Table 7 and Figure 4). Although the exact mechanism remains obscure, adding probiotics reduces migraine-associated headaches [181]. Li et al. reported that supplementing probiotics with traditional triple therapy reduces headaches with or without vomiting [182]. Additionally, both amoxicillin and clarithromycin antimicrobials receivers the possibility of GI issues, especially diarrhea, which were reduced with the supplementation of probiotics. Lactobacillus strains are well-known as noble probiotics and are most widely used among humans [183-185]. Another study claimed probiotics benefit pharmacology in diverse intestinal disorders, including diarrhea [182].
Additionally, probiotics, especially Lactobacillus strains such as L. acidophilus, L. casei, L. salivarius, L. reuteri, L. johnsonii, and L. gasseri [186], possess the necessary pharmacodynamics to inhibit H. pylori [97,186-188]. This analysis also detected a lower risk of dyspepsia in the group with probiotics for those cases receiving metronidazole. Muresan et al. reported that Lactobacillus strains, mainly L. reuteri, minimize chronic dyspepsia [188]. Bismuth salt receivers of the current study exhibited a decreased risk of nausea and GI-related disorders. Bismuth subsalicylate (BSS) was permitted by the United States Food and Drug Administration (FDA) in 1939 for following clinical disorders such as diarrhea, heartburn, indigestions, nausea, and stomach upsets [189]. Furthermore, BSS possesses pharmacological properties in minimizing GI distress and traveler's diarrhea [190]. BSS also decreases the austerity and frequency of diarrhea and flatulence [189]. Moreover, probiotic + levofloxacin recipients of the current study demonstrated lower odds of experiencing vomiting, flatulence, diarrhea, and multiple joint pain (Table 7 and Figure 4). In their research, Pugh et al. confirmed that probiotic therapeutic intervention frequently abolishes GI indicators, especially bringing up wind, biliousness, vomiting, passing intestinal gas, and loose motion [191]. Furthermore, Sheffield et al. reported that adding probiotics with fluoroquinolones does not increase ADRs [192].
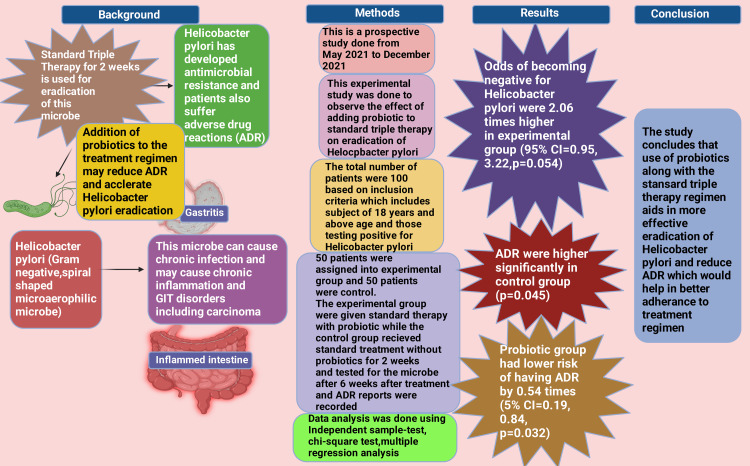
The principal findings of the current study are illustrated in Figure 7.
Figure 7. Principal findings of the current study.
ADR: adverse drug reaction
Image Credit: Rahnuma Ahmad; with the premium version of BioRender [114] with license number XA26G8WP6M
Limitations of the study
This study has limitations, including a small sample size, potentially compromising the generalizability of findings. Reliance on self-reported data raises the possibility of recall bias, impacting the accuracy of information. Additional limitations include the short follow-up duration and lack of assessment for publication bias. Careful consideration of these factors is crucial for a nuanced interpretation of the study's outcomes.
Conclusions
H. pylori causes persistent inflammation as well as infection in the stomach's superficial epithelial layer, leading to GI diseases such as cancer. Probiotics significantly enhance the condition of the gut microbiota and the bacteriostatic activity of probiotics hinders the replication of H. pylori in the stomach. Positive clinical effects for the H. pylori biomarker (RUT) during triple therapy with the probiotic intervention were found by model analysis in the current study. In contrast with the control group (only STT), the probability of going from positive to negative RUT was more than two times higher in the experimental group (SST + probiotics). In terms of the risk of ADRs, the logistic regression model analysis found that the experimental group (STT + probiotics) had a lower risk of side effects than the control group (only STT). Ongoing exploration is an urgent requirement among Bangladeshi patients, as H. pylori is highly infectious, easily transferable from person to person, and life-threatening microbes that may ultimately lead to gastric cancer.
Acknowledgments
I extend heartfelt thanks to the junior doctors and staff in the endoscopy suits at Ship International Hospital, Dhaka, Bangladesh, for their invaluable contributions to this research. The authors are very grateful to Dr. Namrata Dagli for helping us to correct our figures.
The authors have declared that no competing interests exist.
Author Contributions
Concept and design: Mainul Haque, Taslima Zaman, Ahsanul Haq, Rahnuma Ahmad, Susmita Sinha, Sultana Parvin, Mostofa Imran, Zaman U. Humayra, Santosh Kumar, Kona Chowdhury
Acquisition, analysis, or interpretation of data: Mainul Haque, Taslima Zaman, Ahsanul Haq, Rahnuma Ahmad, Susmita Sinha, Sultana Parvin, Mostofa Imran, Zaman U. Humayra, Santosh Kumar, Kona Chowdhury
Drafting of the manuscript: Mainul Haque, Taslima Zaman, Ahsanul Haq, Rahnuma Ahmad, Susmita Sinha, Sultana Parvin, Mostofa Imran, Zaman U. Humayra, Santosh Kumar, Kona Chowdhury
Critical review of the manuscript for important intellectual content: Mainul Haque, Taslima Zaman, Ahsanul Haq, Rahnuma Ahmad, Susmita Sinha, Sultana Parvin, Mostofa Imran, Zaman U. Humayra, Santosh Kumar, Kona Chowdhury
Supervision: Mainul Haque, Taslima Zaman, Ahsanul Haq, Rahnuma Ahmad, Susmita Sinha, Sultana Parvin, Mostofa Imran, Zaman U. Humayra, Santosh Kumar, Kona Chowdhury
Human Ethics
Consent was obtained or waived by all participants in this study. Institutional Review Board of Ship International Hospital (Former name - Japan East West Medical College Hospital), Dhaka, Bangladesh issued approval JEWMCH/IEC/01, dated March 5, 2021. The study adhered to the World Medical Association's Declaration of Helsinki. Research objectives and future publication plans were explained in detail to patients and guardians
Animal Ethics
Animal subjects: All authors have confirmed that this study did not involve animal subjects or tissue.
References
- 1.East Asian-specific and cross-ancestry genome-wide meta-analyses provide mechanistic insights into peptic ulcer disease. He Y, Koido M, Sutoh Y, et al. Nat Genet. 2023;55:2129–2138. doi: 10.1038/s41588-023-01569-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Peptic ulcer disease: a brief review of conventional therapy and herbal treatment options. Kuna L, Jakab J, Smolic R, Raguz-Lucic N, Vcev A, Smolic M. J Clin Med. 2019;8:179. doi: 10.3390/jcm8020179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.The prevalence and incidence of Helicobacter pylori-associated peptic ulcer disease and upper gastrointestinal bleeding throughout the world. Wang AY, Peura DA. Gastrointest Endosc Clin N Am. 2011;21:613–635. doi: 10.1016/j.giec.2011.07.011. [DOI] [PubMed] [Google Scholar]
- 4.23 years of the discovery of Helicobacter pylori: is the debate over? Ahmed N. Ann Clin Microbiol Antimicrob. 2005;4:17. doi: 10.1186/1476-0711-4-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Helicobacter pylori--a Nobel pursuit? Marshall B. Can J Gastroenterol. 2008;22:895–896. doi: 10.1155/2008/459810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.The association between peptic ulcer diseases and mental health problems: a population-based study: a STROBE compliant article. Lee YB, Yu J, Choi HH, et al. Medicine (Baltimore) 2017;96:0. doi: 10.1097/MD.0000000000007828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perforated peptic ulcer disease: a review of history and treatment. Bertleff MJ, Lange JF. Dig Surg. 2010;27:161–169. doi: 10.1159/000264653. [DOI] [PubMed] [Google Scholar]
- 8.Systematic analysis of monoterpenes: advances and challenges in the treatment of peptic ulcer diseases. Périco LL, Emílio-Silva MT, Ohara R, et al. Biomolecules. 2020;10:265. doi: 10.3390/biom10020265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rethinking the history of peptic ulcer disease and its relevance for network epistemology. Radomski BM, Šešelja D, Naumann K. Hist Philos Life Sci. 2021;43:113. doi: 10.1007/s40656-021-00466-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.The association of Helicobacter pylori infection and nonsteroidal anti-inflammatory drugs in peptic ulcer disease. Zapata-Colindres JC, Zepeda-Gómez S, Montaño-Loza A, Vázquez-Ballesteros E, de Jesús Villalobos J, Valdovinos-Andraca F. Can J Gastroenterol. 2006;20:277–280. doi: 10.1155/2006/175217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Prevalence of Helicobacter pylori infection and the incidence of the associated malignant and peptic ulcer disease (PUD) at Nelson Mandela Academic Hospital: a retrospective analysis. Molaoa SZ. J Drug Assess. 2021;10:57–61. doi: 10.1080/21556660.2020.1854560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Overview of Helicobacter pylori Infection: clinical features, treatment, and nutritional aspects. Öztekin M, Yılmaz B, Ağagündüz D, Capasso R. Diseases. 2021;9:66. doi: 10.3390/diseases9040066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Changing prevalence of Helicobacter pylori infection in children and adolescents. Park JS, Jun JS, Seo JH, Youn HS, Rhee KH. Clin Exp Pediatr. 2021;64:21–25. doi: 10.3345/cep.2019.01543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Prevalence and ethnic distribution of Helicobacter pylori infection among endoscoped patients in north eastern peninsular Malaysia. Kaur G, Naing NN. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3561889/pdf/mjms-10-2-066.pdf. Malays J Med Sci. 2003;10:66–70. [PMC free article] [PubMed] [Google Scholar]
- 15.Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Hooi JK, Lai WY, Ng WK, et al. Gastroenterology. 2017;153:420–429. doi: 10.1053/j.gastro.2017.04.022. [DOI] [PubMed] [Google Scholar]
- 16.Helicobacter pylori infection. Malfertheiner P, Camargo MC, El-Omar E, et al. Nat Rev Dis Primers. 2023;9:19. doi: 10.1038/s41572-023-00431-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Peptic ulcer disease and Helicobacter pylori infection. Narayanan M, Reddy KM, Marsicano E. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6140150/pdf/ms115_p0219.pdf. Mo Med. 2018;115:219–224. [PMC free article] [PubMed] [Google Scholar]
- 18.Helicobacter pylori SabA adhesin in persistent infection and chronic inflammation. Mahdavi J, Sondén B, Hurtig M, et al. Science. 2002;297:573–578. doi: 10.1126/science.1069076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Effect of Helicobacter pylori on gastric epithelial cells. Alzahrani S, Lina TT, Gonzalez J, Pinchuk IV, Beswick EJ, Reyes VE. World J Gastroenterol. 2014;20:12767–12780. doi: 10.3748/wjg.v20.i36.12767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Role of the Helicobacter pylori-induced inflammatory response in the development of gastric cancer. Lamb A, Chen LF. J Cell Biochem. 2013;114:491–497. doi: 10.1002/jcb.24389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pathogenesis of Helicobacter pylori infection. Kusters JG, van Vliet AH, Kuipers EJ. Clin Microbiol Rev. 2006;19:449–490. doi: 10.1128/CMR.00054-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Epidemiology of Helicobacter pylori in Australia: a scoping review. Congedi J, Williams C, Baldock KL. PeerJ. 2022;10:0. doi: 10.7717/peerj.13430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Prevalence of Helicobacter pylori infection worldwide: a systematic review of studies with national coverage. Peleteiro B, Bastos A, Ferro A, Lunet N. Dig Dis Sci. 2014;59:1698–1709. doi: 10.1007/s10620-014-3063-0. [DOI] [PubMed] [Google Scholar]
- 24.Worldwide prevalence and risk factors of Helicobacter pylori infection in children. Borka Balas R, Meliț LE, Mărginean CO. Children (Basel) 2022;9:1359. doi: 10.3390/children9091359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Global estimate of gastric cancer in Helicobacter pylori-infected population: a systematic review and meta-analysis. Pormohammad A, Mohtavinejad N, Gholizadeh P, Dabiri H, Salimi Chirani A, Hashemi A, Nasiri MJ. J Cell Physiol. 2019;234:1208–1218. doi: 10.1002/jcp.27114. [DOI] [PubMed] [Google Scholar]
- 26.Effect of Helicobacter pylori treatment on long-term mortality in patients with hypertension. Kim YI, Kim YA, Lee JW, et al. Gut Liver. 2020;14:47–56. doi: 10.5009/gnl18510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Can Helicobacter pylori eradication affect long-term mortality? Lee HL. Korean J Intern Med. 2021;36:539–540. doi: 10.3904/kjim.2021.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Helicobacter pylori and its role in gastric cancer. Reyes VE. Microorganisms. 2023;11:1312. doi: 10.3390/microorganisms11051312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Current prevalence status of gastric cancer and recent studies on the roles of circular RNAs and methods used to investigate circular RNAs. Jiang F, Shen X. Cell Mol Biol Lett. 2019;24:53. doi: 10.1186/s11658-019-0178-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.The current and future incidence and mortality of gastric cancer in 185 countries, 2020-40: a population-based modelling study. Morgan E, Arnold M, Camargo MC, et al. EClinicalMedicine. 2022;47:101404. doi: 10.1016/j.eclinm.2022.101404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Epidemiology of stomach cancer. Ilic M, Ilic I. World J Gastroenterol. 2022;28:1187–1203. doi: 10.3748/wjg.v28.i12.1187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Development of Helicobacter pylori treatment: how do we manage antimicrobial resistance? Suzuki S, Esaki M, Kusano C, Ikehara H, Gotoda T. World J Gastroenterol. 2019;25:1907–1912. doi: 10.3748/wjg.v25.i16.1907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Update on the first-line treatment for Helicobacter pylori infection - a continuing challenge from an old enemy. Huang CC, Tsai KW, Tsai TJ, Hsu PI. Biomark Res. 2017;5:23. doi: 10.1186/s40364-017-0103-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Structure and function of Helicobacter pylori CagA, the first-identified bacterial protein involved in human cancer. Hatakeyama M. Proc Jpn Acad Ser B Phys Biol Sci. 2017;93:196–219. doi: 10.2183/pjab.93.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Helicobacter pylori multiplex serology and risk of non-cardia and cardia gastric cancer: a case-cohort study and meta-analysis. Yao P, Kartsonaki C, Butt J, et al. Int J Epidemiol. 2023;52:1197–1208. doi: 10.1093/ije/dyad007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.The relative and attributable risks of cardia and non-cardia gastric cancer associated with Helicobacter pylori infection in China: a case-cohort study. Yang L, Kartsonaki C, Yao P, et al. Lancet Public Health. 2021;6:0–96. doi: 10.1016/S2468-2667(21)00164-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pathophysiological functions of the CagA oncoprotein during infection by Helicobacter pylori. Murata-Kamiya N. Microbes Infect. 2011;13:799–807. doi: 10.1016/j.micinf.2011.03.011. [DOI] [PubMed] [Google Scholar]
- 38.Recurrence of duodenal ulcer and Campylobacter pylori infection after eradication. Borody TJ, Cole P, Noonan S, et al. Med J Aust. 1989;151:431–435. doi: 10.5694/j.1326-5377.1989.tb101251.x. [DOI] [PubMed] [Google Scholar]
- 39.How to effectively use bismuth quadruple therapy: the good, the bad, and the ugly. Graham DY, Lee SY. Gastroenterol Clin North Am. 2015;44:537–563. doi: 10.1016/j.gtc.2015.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.A Malaysian consensus report on the diagnosis and treatment of Helicobacter pylori infection. Goh KL, Lee YY, Leow AH, et al. JGH Open. 2023;7:261–271. doi: 10.1002/jgh3.12886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Antibiotic treatment for Helicobacter pylori: is the end coming? Kim SY, Choi DJ, Chung JW. World J Gastrointest Pharmacol Ther. 2015;6:183–198. doi: 10.4292/wjgpt.v6.i4.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Evolution of Helicobacter pylori resistance to antibiotics: a topic of increasing concern. Boyanova L, Hadzhiyski P, Gergova R, Markovska R. Antibiotics (Basel) 2023;12:332. doi: 10.3390/antibiotics12020332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Efficacy and safety of gemifloxacin containing treatment regimen in first-line treatment of Helicobacter pylori. Alanli R, Kucukay MB, Aydin MF, Ergül B, Yakaryilmaz F. Arq Gastroenterol. 2023;60:350–355. doi: 10.1590/S0004-2803.230302-23-51. [DOI] [PubMed] [Google Scholar]
- 44.Eradication treatment of Helicobacter pylori infection based on molecular pathologic antibiotic resistance. Gao C, Du SY, Fang L, Fan YH, Song AP, Chen H. Infect Drug Resist. 2020;13:69–79. doi: 10.2147/IDR.S232169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Helicobacter pylori in peptic ulcer disease. Yamada T, Searle JG, Ahnen D, et al. JAMA. 1994;272:65–69. [Google Scholar]
- 46.Current European concepts in the management of Helicobacter pylori infection. The Maastricht consensus report. European Helicobacter pylori study group. Gut. 1997;41:8–13. doi: 10.1136/gut.41.1.8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Malfertheiner P, Megraud F, Rokkas T, et al. Gut. 2022 doi: 10.1136/gutjnl-2012-302084. [DOI] [PubMed] [Google Scholar]
- 48.The Toronto consensus for the treatment of Helicobacter pylori infection in adults. Fallone CA, Chiba N, van Zanten SV, et al. Gastroenterology. 2016;151:51–69. doi: 10.1053/j.gastro.2016.04.006. [DOI] [PubMed] [Google Scholar]
- 49.Current guidelines for Helicobacter pylori treatment in East Asia 2022: differences among China, Japan, and South Korea. Cho JH, Jin SY. World J Clin Cases. 2022;10:6349–6359. doi: 10.12998/wjcc.v10.i19.6349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Consensus recommendations for the screening, diagnosis, and management of Helicobacter pylori infection in Hong Kong. Leung WK, Cheung KS, Sham PC, et al. Hong Kong Med J. 2023;29:532–541. doi: 10.12809/hkmj2210321. [DOI] [PubMed] [Google Scholar]
- 51.Screening and eradication of Helicobacter pylori for gastric cancer prevention: the Taipei global consensus. Liou JM, Malfertheiner P, Lee YC, et al. Gut. 2020;69:2093–2112. doi: 10.1136/gutjnl-2020-322368. [DOI] [PubMed] [Google Scholar]
- 52.Treatment for the eradication of Helicobacter pylori. Recommendations of the Spanish Consensus Conference. Gisbert JP, Calvet X, Gomollon F, et al. Med Clin (Barc) 2000;114:185–195. doi: 10.1016/s0025-7753(00)71237-1. [DOI] [PubMed] [Google Scholar]
- 53.Canadian Helicobacter pylori consensus conference update: infections in adults. Canadian Helicobacter study group. Hunt RH, Fallone CA, Thomson AB. Can J Gastroenterol. 1999;13:213–217. doi: 10.1155/1999/180751. [DOI] [PubMed] [Google Scholar]
- 54.Current concepts in the management of Helicobacter pylori infection--the Maastricht 2-2000 consensus report. Malfertheiner P, Mégraud F, O'Morain C, et al. Aliment Pharmacol Ther. 2002;16:167–180. doi: 10.1046/j.1365-2036.2002.01169.x. [DOI] [PubMed] [Google Scholar]
- 55.Management of Helicobacter pylori infection. The Maastricht IV/ Florence consensus report. Malfertheiner P, Megraud F, O'Morain CA, et al. Gut. 2012;61:646–664. doi: 10.1136/gutjnl-2012-302084. [DOI] [PubMed] [Google Scholar]
- 56.American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Chey WD, Wong BC. Am J Gastroenterol. 2007;102:1808–1825. doi: 10.1111/j.1572-0241.2007.01393.x. [DOI] [PubMed] [Google Scholar]
- 57.Helicobacter pylori infection and antibiotic resistance - from biology to clinical implications. Tshibangu-Kabamba E, Yamaoka Y. Nat Rev Gastroenterol Hepatol. 2021;18:613–629. doi: 10.1038/s41575-021-00449-x. [DOI] [PubMed] [Google Scholar]
- 58.Epidemiology, diagnosis and risk factors of helicobacter pylori infection. Kotilea K, Bontems P, Touati E. Adv Exp Med Biol. 2019;1149:17–33. doi: 10.1007/5584_2019_357. [DOI] [PubMed] [Google Scholar]
- 59.Helicobacter pylori eradication - an update on the latest therapies. Yaxley J, Chakravarty B. https://www.racgp.org.au/getattachment/91c724f0-02a2-4ac9-9186-e59f772b2d67/Helicobacter-pylori-eradication.aspx. Aust Fam Physician. 2014;43:301–305. [PubMed] [Google Scholar]
- 60.Regular review: treatment of Helicobacter pylori infection. de Boer WA, Tytgat GN. BMJ. 2000;320:31–34. doi: 10.1136/bmj.320.7226.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.From antibiotic resistance to antibiotic renaissance: a new era in helicobacter pylori treatment. Godavarthy PK, Puli C. Cureus. 2023;15:0. doi: 10.7759/cureus.36041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Antibiotic resistance in Helicobacter pylori: from potential biomolecular mechanisms to clinical practice. Lin Y, Shao Y, Yan J, Ye G. J Clin Lab Anal. 2023;37:0. doi: 10.1002/jcla.24885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Identification of Helicobacter pylori biofilms in human gastric mucosa. Carron MA, Tran VR, Sugawa C, Coticchia JM. J Gastrointest Surg. 2006;10:712–717. doi: 10.1016/j.gassur.2005.10.019. [DOI] [PubMed] [Google Scholar]
- 64.Biofilm-specific antibiotic tolerance and resistance. Olsen I. Eur J Clin Microbiol Infect Dis. 2015;34:877–886. doi: 10.1007/s10096-015-2323-z. [DOI] [PubMed] [Google Scholar]
- 65.Cholesterol enhances Helicobacter pylori resistance to antibiotics and LL-37. McGee DJ, George AE, Trainor EA, Horton KE, Hildebrandt E, Testerman TL. Antimicrob Agents Chemother. 2011;55:2897–2904. doi: 10.1128/AAC.00016-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Rates of antimicrobial resistance in Helicobacter pylori isolates from clinical trial patients across the US and Europe. Mégraud F, Graham DY, Howden CW, et al. Am J Gastroenterol. 2023;118:269–275. doi: 10.14309/ajg.0000000000002045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Review article: the global emergence of Helicobacter pylori antibiotic resistance. Thung I, Aramin H, Vavinskaya V, Gupta S, Park JY, Crowe SE, Valasek MA. Aliment Pharmacol Ther. 2016;43:514–533. doi: 10.1111/apt.13497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Asian Pacific Alliance on Helicobacter and Microbiota. Primary antibiotic resistance in Helicobacter pylori in the Asia-Pacific region: a systematic review and meta-analysis. Kuo YT, Liou JM, El-Omar EM, et al. Lancet Gastroenterol Hepatol. 2017;2:707–715. doi: 10.1016/S2468-1253(17)30219-4. [DOI] [PubMed] [Google Scholar]
- 69.Self-medication with antibiotics in WHO Southeast Asian region: a systematic review. Nepal G, Bhatta S. Cureus. 2018;10:0. doi: 10.7759/cureus.2428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Clinical significance of various diagnostic techniques and emerging antimicrobial resistance pattern of Helicobacter pylori from gastric biopsy samples. Vagarali MA, Metgud SC, Bannur H, Karadesai SG, Nagmoti JM. Indian J Med Microbiol. 2015;33:560–564. doi: 10.4103/0255-0857.167349. [DOI] [PubMed] [Google Scholar]
- 71.Emerging trends of antimicrobial resistance in Helicobacter pylori isolates obtained from Pakistani patients: the need for consideration of amoxicillin and clarithromycin. Siddiqui TR, Ahmed W, Arif A, Bibi S, Khan A. http://www.jpma.org.pk/PdfDownload/7788.pdf. J Pak Med Assoc. 2016;66:710–716. [PubMed] [Google Scholar]
- 72.Prevalence of antibiotic resistance in Helicobacter pylori: a systematic review and meta-analysis in World Health Organization regions. Savoldi A, Carrara E, Graham DY, Conti M, Tacconelli E. Gastroenterology. 2018;155:1372–1382. doi: 10.1053/j.gastro.2018.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Antimicrobial susceptibility of Helicobacter pylori strains isolated in Bangladesh. Nahar S, Mukhopadhyay AK, Khan R, et al. J Clin Microbiol. 2004;42:4856–4858. doi: 10.1128/JCM.42.10.4856-4858.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Helicobacter pylori antibiotic susceptibility patterns in Bangladesh: emerging levofloxacin resistance. Aftab H, Miftahussurur M, Subsomwong P, Ahmed F, Khan AK, Yamaoka Y. J Infect Dev Ctries. 2016;10:245–253. doi: 10.3855/jidc.7713. [DOI] [PubMed] [Google Scholar]
- 75.Antimicrobial resistance: a growing serious threat for global public health. Salam MA, Al-Amin MY, Salam MT, Pawar JS, Akhter N, Rabaan AA, Alqumber MA. Healthcare (Basel) 2023;11:1946. doi: 10.3390/healthcare11131946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Antimicrobial resistance: a top ten global public health threat. EClinicalMedicine EClinicalMedicine. EClinicalMedicine. 2021;41:101221. doi: 10.1016/j.eclinm.2021.101221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Helicobacter pylori oncogenicity: mechanism, prevention, and risk factors. Muzaheed Muzaheed. ScientificWorldJournal. 2020;2020:3018326. doi: 10.1155/2020/3018326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Is hybrid therapy more efficient in the eradication of Helicobacter pylori infection? A systematic review and meta-analysis. Temido MJ, Mbanze D, Almeida N, Oliveiros B, Gravito-Soares E, Figueiredo P. Ann Clin Microbiol Antimicrob. 2023;22:54. doi: 10.1186/s12941-023-00582-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Comparison of multiple treatment regimens in children with Helicobacter pylori infection: a network meta-analysis. Liang M, Zhu C, Zhao P, Zhu X, Shi J, Yuan B. Front Cell Infect Microbiol. 2023;13:1068809. doi: 10.3389/fcimb.2023.1068809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Helicobacter pylori resistance to antibiotics before and after treatment: Incidence of eradication failure. Nestegard O, Moayeri B, Halvorsen FA, et al. PLoS One. 2022;17:0. doi: 10.1371/journal.pone.0265322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Single capsule bismuth quadruple therapy for eradication of H. pylori infection: a real-life study. Gravina AG, Priadko K, Granata L, et al. Front Pharmacol. 2021;12:667584. doi: 10.3389/fphar.2021.667584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Randomized controlled trial for Helicobacter pylori eradication in a naive Portuguese population: is sequential treatment superior to triple therapy in real world clinical setting? Boal Carvalho P, Magalhães J, Dias de Castro F, Rosa B, Cotter J. Acta Med Port. 2017;30:185–189. doi: 10.20344/amp.8072. [DOI] [PubMed] [Google Scholar]
- 83.Standard bismuth quadruple therapy versus concomitant therapy for the first-line treatment of Helicobacter pylori infection: a systematic review and meta-analysis of randomized controlled trials. Zagari RM, Dajti E, Cominardi A, et al. J Clin Med. 2023;12:3258. doi: 10.3390/jcm12093258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Helicobacter pylori antimicrobial resistance during a 5-year period (2013-2017) in northern Spain and its relationship with the eradication therapies. Cosme A, Torrente Iranzo S, Montes Ros M, Fernández-Reyes Silvestre M, Alonso Galán H, Lizasoain J, Bujanda L. Helicobacter. 2019;24:0. doi: 10.1111/hel.12557. [DOI] [PubMed] [Google Scholar]
- 85.The antibiotic resistance of Helicobacter pylori to five antibiotics and influencing factors in an area of China with a high risk of gastric cancer. Wang D, Guo Q, Yuan Y, Gong Y. BMC Microbiol. 2019;19:152. doi: 10.1186/s12866-019-1517-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Helicobacter pylori eradication therapy: still a challenge. Hafeez M, Qureshi ZA, Khattak AL, Saeed F, Asghar A, Azam K, Khan MA. Cureus. 2021;13:0. doi: 10.7759/cureus.14872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.The effect of probiotic plus prebiotic supplementation on the tolerance and efficacy of Helicobacter pylori eradication quadruple therapy: a randomized prospective double blind controlled trial. Shafaghi A, Pourkazemi A, Khosravani M, Fakhrie Asl S, Amir Maafi A, Atrkar Roshan Z, Abaspour Rahimabad J. Middle East J Dig Dis. 2016;8:179–188. doi: 10.15171/mejdd.2016.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Self-reported adverse drug effects and associated factors among H. pylori infected patients on standard triple therapy: prospective follow up study. Gebeyehu E, Nigatu D, Engidawork E. PLoS One. 2019;14:0. doi: 10.1371/journal.pone.0225585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Helicobacter pylori eradication rate of standard triple therapy and factors affecting eradication rate at Bahir Dar city administration, northwest Ethiopia: a prospective follow up study. Gebeyehu E, Nigatu D, Engidawork E. PLoS One. 2019;14:0. doi: 10.1371/journal.pone.0217645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Empirical modified sequential therapy containing levofloxacin and high-dose esomeprazole in second-line therapy for Helicobacter pylori infection: a multicentre clinical trial. Liou JM, Chen CC, Chen MJ, et al. J Antimicrob Chemother. 2011;66:1847–1852. doi: 10.1093/jac/dkr217. [DOI] [PubMed] [Google Scholar]
- 91.Exploring alternative treatments for Helicobacter pylori infection. Ayala G, Escobedo-Hinojosa WI, de la Cruz-Herrera CF, Romero I. World J Gastroenterol. 2014;20:1450–1469. doi: 10.3748/wjg.v20.i6.1450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.High effectiveness of a 14-day concomitant therapy for Helicobacter pylori treatment in primary care. an observational multicenter study. Olmedo L, Azagra R, Aguyé A, Pascual M, Calvet X, Gené E. J Clin Med. 2020;9:2410. doi: 10.3390/jcm9082410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Treatment approach of refractory helicobacter pylori infection: a comprehensive review. Cortés P, Nelson AD, Bi Y, et al. J Prim Care Community Health. 2021;12:21501327211014087. doi: 10.1177/21501327211014087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Improving compliance with helicobacter pylori eradication therapy: when and how? O'Connor JP, Taneike I, O'Morain C. Therap Adv Gastroenterol. 2009;2:273–279. doi: 10.1177/1756283X09337342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.The patients' adherence and adverse drug reactions (ADRs) which are caused by Helicobacter pylori eradication regimens. Abbasinazari M, Sahraee Z, Mirahmadi M. J Clin Diagn Res. 2013;7:462–466. doi: 10.7860/JCDR/2013/4673.2799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Comparative effectiveness of different probiotics supplements for triple helicobacter pylori eradication: a network meta-analysis. Wang Y, Wang X, Cao XY, Zhu HL, Miao L. Front Cell Infect Microbiol. 2023;13:1120789. doi: 10.3389/fcimb.2023.1120789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Role of probiotics in the management of Helicobacter pylori. Mestre A, Sathiya Narayanan R, Rivas D, et al. Cureus. 2022;14:0. doi: 10.7759/cureus.26463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Saccharomyces boulardii in Helicobacter pylori eradication in children: a randomized trial from Iran. Namkin K, Zardast M, Basirinejad F. Iran J Pediatr. 2016;26:0. doi: 10.5812/ijp.3768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Saccharomyces boulardii CNCM I-745 in different clinical conditions. Dinleyici EC, Kara A, Ozen M, Vandenplas Y. Expert Opin Biol Ther. 2014;14:1593–1609. doi: 10.1517/14712598.2014.937419. [DOI] [PubMed] [Google Scholar]
- 100.Relationship between Helicobacter pylori infection and gastrointestinal microecology. Xu W, Xu L, Xu C. Front Cell Infect Microbiol. 2022;12:938608. doi: 10.3389/fcimb.2022.938608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Probiotic bifunctionality of Bacillus subtilis-rescuing lactic acid bacteria from desiccation and antagonizing pathogenic Staphylococcus aureus. Kimelman H, Shemesh M. Microorganisms. 2019;7 doi: 10.3390/microorganisms7100407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Comparison of safety, effectiveness and serum inflammatory factor indexes of Saccharomyces boulardii versus Bifidobacterium triple viable in treating children with chronic diarrhea: a randomized trial. Zhang J, Wan S, Gui Q. Transl Pediatr. 2021;10:1677–1685. doi: 10.21037/tp-21-195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Probiotics as the live microscopic fighters against Helicobacter pylori gastric infections. Keikha M, Karbalaei M. BMC Gastroenterol. 2021;21:388. doi: 10.1186/s12876-021-01977-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Probiotic effect on Helicobacter pylori attachment and inhibition of inflammation in human gastric epithelial cells. Song H, Zhou L, Liu D, Ge L, Li Y. Exp Ther Med. 2019;18:1551–1562. doi: 10.3892/etm.2019.7742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.In vitro antimicrobial activity and downregulation of virulence gene expression on Helicobacter pylori by reuterin. Urrutia-Baca VH, Escamilla-García E, de la Garza-Ramos MA, Tamez-Guerra P, Gomez-Flores R, Urbina-Ríos CS. Probiotics Antimicrob Proteins. 2018;10:168–175. doi: 10.1007/s12602-017-9342-2. [DOI] [PubMed] [Google Scholar]
- 106.Use of probiotics as an adjuvant to sequential H. pylori eradication therapy: impact on eradication rates, treatment resistance, treatment-related side effects, and patient compliance. Çekin AH, Şahintürk Y, Akbay Harmandar F, Uyar S, Yolcular BO, Çekin Y. Turk J Gastroenterol. 2017;28:3–11. doi: 10.5152/tjg.2016.0278. [DOI] [PubMed] [Google Scholar]
- 107.Exopolysaccharide-producing Streptococcus thermophilus CRL1190 reduces the inflammatory response caused by Helicobacter pylori. Marcial G, Villena J, Faller G, Hensel A, de Valdéz GF. Benef Microbes. 2017;8:451–461. doi: 10.3920/BM2016.0186. [DOI] [PubMed] [Google Scholar]
- 108.Best Helicobacter pylori eradication strategy in the era of antibiotic resistance. Kim SY, Chung JW. Antibiotics (Basel) 2020;9:436. doi: 10.3390/antibiotics9080436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Safety and tolerability of antimicrobial agents in the older patient. Soraci L, Cherubini A, Paoletti L, et al. Drugs Aging. 2023;40:499–526. doi: 10.1007/s40266-023-01019-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Antibiotic-therapy-induced gut dysbiosis affecting gut microbiota-brain axis and cognition: restoration by intake of probiotics and synbiotics. Dahiya D, Nigam PS. Int J Mol Sci. 2023;24 doi: 10.3390/ijms24043074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.The impacts of probiotics in eradication therapy of Helicobacter pylori. Bai X, Zhu M, He Y, Wang T, Tian D, Shu J. Arch Microbiol. 2022;204:692. doi: 10.1007/s00203-022-03314-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Utilizing probiotics for the prevention and treatment of gastrointestinal diseases. Milner E, Stevens B, An M, et al. Front Microbiol. 2021;12:689958. doi: 10.3389/fmicb.2021.689958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Effects of anti-H. pylori triple therapy and a probiotic complex on intestinal microbiota in duodenal ulcer. Wu L, Wang Z, Sun G, et al. Sci Rep. 2019;9:12874. doi: 10.1038/s41598-019-49415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.BioRender. [ Feb; 2024 ]. 2024. https://biorender.com https://biorender.com
- 115.Probiotics and autoprobiotics for treatment of Helicobacter pylori infection. Baryshnikova NV, Ilina AS, Ermolenko EI, Uspenskiy YP, Suvorov AN. World J Clin Cases. 2023;11:4740–4751. doi: 10.12998/wjcc.v11.i20.4740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Antibiotics as major disruptors of gut microbiota. Ramirez J, Guarner F, Bustos Fernandez L, Maruy A, Sdepanian VL, Cohen H. Front Cell Infect Microbiol. 2020;10:572912. doi: 10.3389/fcimb.2020.572912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Probiotics, their prophylactic and therapeutic applications in human health development: a review of the literature. Tegegne BA, Kebede B. Heliyon. 2022;8:0. doi: 10.1016/j.heliyon.2022.e09725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Vitamins and Helicobacter pylori: an updated comprehensive meta-analysis and systematic review. Cai X, Li X, Jin Y, et al. Front Nutr. 2021;8:781333. doi: 10.3389/fnut.2021.781333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.The effect of probiotics supplementation on gut microbiota after Helicobacter pylori eradication: a multicenter randomized controlled trial. Tang B, Tang L, Huang C, et al. Infect Dis Ther. 2021;10:317–333. doi: 10.1007/s40121-020-00372-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Can probiotics play a role in Helicobacter pylori (H. pylori) eradication? Fakhry SM, Kandyl MA, Hashish AF, et al. Egypt Liver J. 2023;13:64. [Google Scholar]
- 121.Interplay and cooperation of Helicobacter pylori and gut microbiota in gastric carcinogenesis. Bakhti SZ, Latifi-Navid S. BMC Microbiol. 2021;21:258. doi: 10.1186/s12866-021-02315-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Oral microbiota and Helicobacter pylori in gastric carcinogenesis: what do we know and where next? Bakhti SZ, Latifi-Navid S. BMC Microbiol. 2021;21:71. doi: 10.1186/s12866-021-02130-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Dysbiosis of the microbiome in gastric carcinogenesis. Castaño-Rodríguez N, Goh KL, Fock KM, Mitchell HM, Kaakoush NO. Sci Rep. 2017;7:15957. doi: 10.1038/s41598-017-16289-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Humphreys C. Textbook of Natural Medicine. St. Louis, Missouri: Elsevier; 2020. Intestinal permeability; pp. 166–177. [Google Scholar]
- 125.Precancerous lesions of the stomach, gastric cancer and hereditary gastric cancer syndromes. Gullo I, Grillo F, Mastracci L, et al. Pathologica. 2020;112:166–185. doi: 10.32074/1591-951X-166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Genetic and molecular aspects of Helicobacter pylori in gastritis, pre- cancerous conditions and gastric adenocrcinoma . Shadifar M, Ataee R, Ataie A, Heydari Gorgi AM, Nasri Nasrabadi N, Nouri S. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4495423/pdf/GHFBB-8-S15.pdf . Gastroenterol Hepatol Bed Bench. 2015;8:0–22. [PMC free article] [PubMed] [Google Scholar]
- 127.Inflammation and gastric cancer. Jaroenlapnopparat A, Bhatia K, Coban S. Diseases. 2022;10:35. doi: 10.3390/diseases10030035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Linking dysbiosis to precancerous stomach through inflammation: deeper than and beyond imaging. Lopes C, Almeida TC, Pimentel-Nunes P, Dinis-Ribeiro M, Pereira C. Front Immunol. 2023;14:1134785. doi: 10.3389/fimmu.2023.1134785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Helicobacter pylori and gastric cancer: adaptive cellular mechanisms involved in disease progression. Díaz P, Valenzuela Valderrama M, Bravo J, Quest AF. Front Microbiol. 2018;9:5. doi: 10.3389/fmicb.2018.00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Eradication of Helicobacter pylori and gastric cancer: a controversial relationship. Piscione M, Mazzone M, Di Marcantonio MC, Muraro R, Mincione G. Front Microbiol. 2021;12:630852. doi: 10.3389/fmicb.2021.630852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Probiotics modulate gastrointestinal microbiota after Helicobacter pylori eradication: a multicenter randomized double-blind placebo-controlled trial. He C, Xie Y, Zhu Y, et al. Front Immunol. 2022;13:1033063. doi: 10.3389/fimmu.2022.1033063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Probiotics in Helicobacter pylori eradication therapy: a systematic review and meta-analysis. Zhang MM, Qian W, Qin YY, He J, Zhou YH. World J Gastroenterol. 2015;21:4345–4357. doi: 10.3748/wjg.v21.i14.4345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Clinical implication of drug resistance for H. pylori management. Argueta EA, Ho JJ, Elfanagely Y, D'Agata E, Moss SF. Antibiotics (Basel) 2022;11:1684. doi: 10.3390/antibiotics11121684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Antibiotic resistance and therapeutic efficacy of Helicobacter pylori infection in pediatric patients-a tertiary center experience. Galoș F, Boboc C, Ieșanu MI, et al. Antibiotics (Basel) 2023;12:146. doi: 10.3390/antibiotics12010146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Global antimicrobial resistance gene study of helicobacter pylori: comparison of detection tools, ARG and efflux pump gene analysis, worldwide epidemiological distribution, and information related to the antimicrobial-resistant phenotype. Alfaray RI, Saruuljavkhlan B, Fauzia KA, et al. Antibiotics (Basel) 2023;12:1118. doi: 10.3390/antibiotics12071118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Research on antibiotic resistance in Helicobacter pylori: a bibliometric analysis of the past decade. Yuan C, Yu C, Sun Q, Xiong M, Zhou S, Zeng M, Song H. Front Microbiol. 2023;14:1208157. doi: 10.3389/fmicb.2023.1208157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.The impact of Helicobacter pylori infection, eradication therapy, and probiotics intervention on gastric microbiota in young adults. Yuan Z, Xiao S, Li S, et al. Helicobacter. 2021;26:0. doi: 10.1111/hel.12848. [DOI] [PubMed] [Google Scholar]
- 138.The efficacy of Lactobacillus acidophilus and rhamnosus in the reduction of bacterial load of Helicobacter pylori and modification of gut microbiota-a double-blind, placebo-controlled, randomized trial. Chen MJ, Chen CC, Huang YC, et al. Helicobacter. 2021;26:0. doi: 10.1111/hel.12857. [DOI] [PubMed] [Google Scholar]
- 139.Metagenomic changes of gut microbiota following treatment of Helicobacter pylori infection with a simplified low-dose quadruple therapy with bismuth or Lactobacillus reuteri. Dore MP, Sau R, Niolu C, et al. Nutrients. 2022;14:2789. doi: 10.3390/nu14142789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.A randomised, controlled trial: effect of a multi-strain fermented milk on the gut microbiota recovery after Helicobacter pylori therapy. Guillemard E, Poirel M, Schäfer F, et al. Nutrients. 2021;13:3171. doi: 10.3390/nu13093171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.A prospective randomized comparative study of the efficacy and safety of a two-week bismuth-based quadrotherapy of Helicobacter pylori infection with the inclusion of the probiotic containing Bifidobacterium longum BB-46 and Enterococcus faecium ENCfa-68 [Article in Russian] Yakovenko EP, Strokova TV, Iakovenko AV, Ivanov AN, Soluyanova IP, Vasilyev NN. Ter Arkh. 2021;93:916–922. doi: 10.26442/00403660.2021.08.200996. [DOI] [PubMed] [Google Scholar]
- 142.Effect of Helicobacter pylori eradication on human gastric microbiota: a systematic review and meta-analysis. Guo Y, Cao XS, Guo GY, Zhou MG, Yu B. Front Cell Infect Microbiol. 2022;12:899248. doi: 10.3389/fcimb.2022.899248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.The ideal Helicobacter pylori treatment for the present and the future. Suzuki S, Kusano C, Horii T, Ichijima R, Ikehara H. Digestion. 2022;103:62–68. doi: 10.1159/000519413. [DOI] [PubMed] [Google Scholar]
- 144.Mutations related to antibiotics resistance in Helicobacter pylori clinical isolates from Bangladesh. Fauzia KA, Aftab H, Tshibangu-Kabamba E, et al. Antibiotics (Basel) 2023;12:279. doi: 10.3390/antibiotics12020279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Drug resistance patterns of commonly used antibiotics for the treatment of Helicobacter pylori infection among south Asian countries: a systematic review and meta-analysis. Shrestha AB, Pokharel P, Sapkota UH, et al. Trop Med Infect Dis. 2023;8:172. doi: 10.3390/tropicalmed8030172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Effective therapeutic regimens in two South Asian countries with high resistance to major Helicobacter pylori antibiotics. Miftahussurur M, Aftab H, Shrestha PK, et al. Antimicrob Resist Infect Control. 2019;8:40. doi: 10.1186/s13756-019-0482-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Roles of cholesteryl-α-glucoside transferase and cholesteryl glucosides in maintenance of Helicobacter pylori morphology, cell wall integrity, and resistance to antibiotics. Qaria MA, Kumar N, Hussain A, Qumar S, Doddam SN, Sepe LP, Ahmed N. mBio. 2018;9 doi: 10.1128/mBio.01523-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Genetic determinants of Biofilm formation of Helicobacter pylori using whole-genome sequencing. Fauzia KA, Aftab H, Miftahussurur M, et al. BMC Microbiol. 2023;23:159. doi: 10.1186/s12866-023-02889-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Gene silencing of Helicobacter pylori through newly designed siRNA convenes the treatment of gastric cancer. Reza MN, Mahmud S, Ferdous N, Ahammad I, Hossain MU, Al Amin M, Mohiuddin AK. Cancer Med. 2023;12:22407–22419. doi: 10.1002/cam4.6772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.High prevalence of Helicobacter pylori metronidazole resistance in migrants to east London: relation with previous nitroimidazole exposure and gastroduodenal disease. Banatvala N, Davies GR, Abdi Y, Clements L, Rampton DS, Hardie JM, Feldman RA. Gut. 1994;35:1562–1566. doi: 10.1136/gut.35.11.1562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.T2182C mutation in 23S rRNA is associated with clarithromycin resistance in Helicobacter pylori isolates obtained in Bangladesh. Khan R, Nahar S, Sultana J, Ahmad MM, Rahman M. Antimicrob Agents Chemother. 2004;48:3567–3569. doi: 10.1128/AAC.48.9.3567-3569.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.A comparative whole genome analysis of Helicobacter pylori from a human dense South Asian setting. Qumar S, Nguyen TH, Nahar S, et al. Helicobacter. 2021;26:0. doi: 10.1111/hel.12766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Isolation of tetracycline-resistant clinical Helicobacter pylori without mutations in 16S rRNA gene in Bangladesh. Khan R, Nahar S, Mukhopadhyay AK, et al. Microbiol Immunol. 2008;52:508–511. doi: 10.1111/j.1348-0421.2008.00062.x. [DOI] [PubMed] [Google Scholar]
- 154.Helicobacter pylori infection and inflammation: implication for the pathophysiology of diabetes and coronary heart disease in Asian Indians. Rahman MA, Cope MB, Sarker SA, Garvey WT, Chaudhury HS, Khaled MA. J Life Sci. 2009;1:45–50. doi: 10.1080/09751270.2009.11885133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Mechanistic inhibition of gastric cancer-associated bacteria Helicobacter pylori by selected phytocompounds: a new cutting-edge computational approach. Akash S, Bayıl I, Mahmood S, et al. Heliyon. 2023;9:0. doi: 10.1016/j.heliyon.2023.e20670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mutations in the 23S rRNA gene of clarithromycin-resistant Helicobacter pylori from Japan. Rimbara E, Noguchi N, Kijima H, Yamaguchi T, Kawai T, Sasatsu M. Int J Antimicrob Agents. 2007;30:250–254. doi: 10.1016/j.ijantimicag.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 157.Methods for detection of Helicobacter pylori from stool sample: current options and developments. Qiu E, Li Z, Han S. Braz J Microbiol. 2021;52:2057–2062. doi: 10.1007/s42770-021-00589-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Helicobacter pylori infections in children. Nguyen J, Kotilea K, Bontems P, Miendje Deyi VY. Antibiotics (Basel) 2023;12:1440. doi: 10.3390/antibiotics12091440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Empiric treatment vs susceptibility-guided treatment for eradicating H. pylori: Is it possible to change that paradigm using modern molecular methods? Garrido-Treviño LF, López-Martínez M, Flores-Hinojosa JA, Tijerina-Rodríguez L, Bosques-Padilla F. Rev Gastroenterol Mex (Engl Ed) 2022;87:330–341. doi: 10.1016/j.rgmxen.2022.06.003. [DOI] [PubMed] [Google Scholar]
- 160.Primary antibiotic resistance in Helicobacter pylori in China: a systematic review and meta-analysis. Wang Y, Du J, Zhang D, et al. J Glob Antimicrob Resist. 2023;34:30–38. doi: 10.1016/j.jgar.2023.05.014. [DOI] [PubMed] [Google Scholar]
- 161.Efficiency of diagnostic testing for Helicobacter pylori infections-a systematic review. Rojas García P, van der Pol S, van Asselt AD, et al. Antibiotics (Basel) 2021;10:55. doi: 10.3390/antibiotics10010055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.The emergence of multidrug-resistant Helicobacter pylori in southeast Asia: a systematic review on the trends and intervention strategies using antimicrobial peptides. Sukri A, Lopes BS, Hanafiah A. Antibiotics (Basel) 2021;10:1061. doi: 10.3390/antibiotics10091061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Efficacy of omeprazole, tetracycline, and 4 times daily dosing of amoxicillin in Helicobacter pylori eradication in limited resource area in Bhutan: a prospective randomized trial (BHUTAN Study) Vilaichone RK, Aumpan N, Ratanachu-Ek T, et al. Asian Pac J Cancer Prev. 2020;21:1109–1114. doi: 10.31557/APJCP.2020.21.4.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.N-acetylcysteine as an adjuvant therapy for Helicobacter pylori eradication. Fontes LE, Martimbianco AL, Zanin C, Riera R. Cochrane Database Syst Rev. 2019;2:0. doi: 10.1002/14651858.CD012357.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Can probiotics improve efficiency and safety profile of triple Helicobacter pylori eradication therapy? A prospective randomized study. Grgov S, Tasić T, Radovanović-Dinić B, Benedeto-Stojanov D. Vojnosanit Pregl. 2016;73:1044–1049. doi: 10.2298/VSP150415127G. [DOI] [PubMed] [Google Scholar]
- 166.Effects of a specially designed fermented milk product containing probiotic Lactobacillus casei DN-114 001 and the eradication of H. pylori in children: a prospective randomized double-blind study. Sýkora J, Valecková K, Amlerová J, et al. J Clin Gastroenterol. 2005;39:692–698. doi: 10.1097/01.mcg.0000173855.77191.44. [DOI] [PubMed] [Google Scholar]
- 167.The effects of multistrain probiotic compound on bismuth-containing quadruple therapy for Helicobacter pylori infection: a randomized placebo-controlled triple-blind study. Shavakhi A, Tabesh E, Yaghoutkar A, et al. Helicobacter. 2013;18:280–284. doi: 10.1111/hel.12047. [DOI] [PubMed] [Google Scholar]
- 168.A randomized, open trial evaluating the effect of Saccharomyces boulardii on the eradication rate of Helicobacter pylori infection in children. Hurduc V, Plesca D, Dragomir D, Sajin M, Vandenplas Y. Acta Paediatr. 2009;98:127–131. doi: 10.1111/j.1651-2227.2008.00977.x. [DOI] [PubMed] [Google Scholar]
- 169.Multidrug resistance: the clinical dilemma of refractory Helicobacter pylori infection. Kuo CJ, Lee CH, Chang ML, et al. J Microbiol Immunol Infect. 2021;54:1184–1187. doi: 10.1016/j.jmii.2021.03.006. [DOI] [PubMed] [Google Scholar]
- 170.The effects of probiotics on treatment of Helicobacter pylori eradication in children. Akcam M, Koca T, Salman H, Karahan N. Saudi Med J. 2015;36:286–290. doi: 10.15537/smj.2015.3.10124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Efficacy of probiotic supplementation therapy for Helicobacter pylori eradication: a meta-analysis of randomized controlled trials. Lü M, Yu S, Deng J, Yan Q, Yang C, Xia G, Zhou X. PLoS One. 2016;11:0. doi: 10.1371/journal.pone.0163743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Endoscopic reflux esophagitis and reflux-related symptoms after Helicobacter pylori eradication therapy: meta-analysis. Sugimoto M, Murata M, Mizuno H, Iwata E, Nagata N, Itoi T, Kawai T. J Clin Med. 2020;9:3007. doi: 10.3390/jcm9093007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Risk of reflux-related symptoms and reflux esophagitis after Helicobacter pylori eradication treatment in the Japanese population. Sugimoto M, Murata M, Iwata E, Nagata N, Itoi T, Kawai T. J Clin Med. 2021;10:1434. doi: 10.3390/jcm10071434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Probiotic supplementation does not improve eradication rate of Helicobacter pylori infection compared to placebo based on standard therapy: a meta-analysis. Lu C, Sang J, He H, et al. Sci Rep. 2016;6:23522. doi: 10.1038/srep23522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Probiotics for the treatment of pediatric Helicobacter pylori infection: a randomized double blind clinical trial. Khodadad A, Farahmand F, Najafi M, Shoaran M. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3574996/pdf/IJPD-23-079.pdf. Iran J Pediatr. 2013;23:79–84. [PMC free article] [PubMed] [Google Scholar]
- 176.Systematic review and meta-analysis: Multi-strain probiotics as adjunct therapy for Helicobacter pylori eradication and prevention of adverse events. McFarland LV, Huang Y, Wang L, Malfertheiner P. United European Gastroenterol J. 2016;4:546–561. doi: 10.1177/2050640615617358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Helicobacter pylori treatment: antibiotics or probiotics. Goderska K, Agudo Pena S, Alarcon T. Appl Microbiol Biotechnol. 2018;102:1–7. doi: 10.1007/s00253-017-8535-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Probiotics for standard triple Helicobacter pylori eradication: a randomized, double-blind, placebo-controlled trial. Hauser G, Salkic N, Vukelic K, JajacKnez A, Stimac D. Medicine (Baltimore) 2015;94:0. doi: 10.1097/MD.0000000000000685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Factors influencing the eradication of Helicobacter pylori with triple therapy. Graham DY, Lew GM, Malaty HM, et al. Gastroenterology. 1992;102:493–496. doi: 10.1016/0016-5085(92)90095-g. [DOI] [PubMed] [Google Scholar]
- 180.Factors affecting the development of adverse drug reactions (review article) Alomar MJ. Saudi Pharm J. 2014;22:83–94. doi: 10.1016/j.jsps.2013.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Gut-brain Axis and migraine headache: a comprehensive review. Arzani M, Jahromi SR, Ghorbani Z, et al. J Headache Pain. 2020;21:15. doi: 10.1186/s10194-020-1078-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Comparative effectiveness and tolerance of treatments for Helicobacter pylori: systematic review and network meta-analysis. Li BZ, Threapleton DE, Wang JY, et al. BMJ. 2015;351:0. doi: 10.1136/bmj.h4052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.The scientific basis for probiotic strains of Lactobacillus. Reid G. Appl Environ Microbiol. 1999;65:3763–3766. doi: 10.1128/aem.65.9.3763-3766.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Lactobacillus spp. for gastrointestinal health: current and future perspectives. Dempsey E, Corr SC. Front Immunol. 2022;13:840245. doi: 10.3389/fimmu.2022.840245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Global analysis of clinical trials with probiotics. Dronkers TM, Ouwehand AC, Rijkers GT. Heliyon. 2020;6:0. doi: 10.1016/j.heliyon.2020.e04467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.A highly acid-resistant novel strain of Lactobacillus johnsonii No. 1088 has antibacterial activity, including that against Helicobacter pylori, and inhibits gastrin-mediated acid production in mice. Aiba Y, Nakano Y, Koga Y, Takahashi K, Komatsu Y. Microbiologyopen. 2015;4:465–474. doi: 10.1002/mbo3.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Effects of Lactobacillus salivarius LN12 in combination with amoxicillin and clarithromycin on Helicobacter pylori biofilm in vitro. Jin F, Yang H. Microorganisms. 2021;9:1611. doi: 10.3390/microorganisms9081611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Lactobacillus reuteri versus triple therapy for the eradication of Helicobacter pylori in functional dyspepsia. Muresan IA, Pop LL, Dumitrascu DL. Med Pharm Rep. 2019;92:352–355. doi: 10.15386/mpr-1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Effect of bismuth subsalicylate on gastrointestinal tolerability in healthy volunteers receiving oral delayed-release dimethyl fumarate: prevent, a randomized, multicenter, double-blind, placebo-controlled study. Koulinska I, Riester K, Chalkias S, Edwards MR. Clin Ther. 2018;40:2021–2030. doi: 10.1016/j.clinthera.2018.10.013. [DOI] [PubMed] [Google Scholar]
- 190.Budisak P, Abbas M. StatPearls [Internet] Treasure Island (FL): StatPearls Publishing; [ Jan; 2024 ]. 2023. Bismuth subsalicylate. [PubMed] [Google Scholar]
- 191.Four weeks of probiotic supplementation reduces GI symptoms during a marathon race. Pugh JN, Sparks AS, Doran DA, et al. Eur J Appl Physiol. 2019;119:1491–1501. doi: 10.1007/s00421-019-04136-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Influence of probiotics on the development of clostridioides difficile infection in patients receiving fluoroquinolones. Sheffield ME, Jones BM, Terrell B, Wagner JL, Bland CM. Pharmacy (Basel) 2021;9:141. doi: 10.3390/pharmacy9030141. [DOI] [PMC free article] [PubMed] [Google Scholar]