Abstract
Background
Various treatments for chronic low back pain (LBP) have been reported; among them, platelet‐rich plasma (PRP) as a regenerative medicine has attracted much attention. Although Modic type 1 change (MC1) is associated with LBP, no treatment has been established so far. In addition, no studies have administered PRP to intervertebral discs (IVDs) in patients with LBP, targeting MC1 only. Thus, the purpose of this study was to determine the safety and efficacy of PRP administration to the IVDs in patients with MC1 experiencing LBP.
Methods
PRP was injected intradiscally to 10 patients with MC1 experiencing LBP. Patients were followed prospectively for up to 24 weeks after primary administration. Physical condition, laboratory data, and lumbar x‐ray images were evaluated for safety assessment. Furthermore, to evaluate the effectiveness of PRP, patient‐reported outcomes were considered. In addition, changes in MC1 were assessed using magnetic resonance imaging (MRI).
Results
There were no adverse events in the laboratory data or lumbar X‐ray images after administration. The mean visual analog scale, which was 70.0 ± 13.3 before the treatment, significantly decreased 1 week after PRP administration and was 39.0 ± 28.8 at the last observation. Oswestry disability index and Roland Morris disability questionnaire scores promptly improved after treatment, and both improved significantly 24 weeks after PRP administration. Follow‐up MRI 24 weeks after treatment showed a significant decrease in the mean high‐signal intensity of fat‐suppressed T2‐weighted imaging from 10.1 to 7.90 mm2 compared with that before PRP administration.
Conclusions
The safety and efficacy of PRP administration to the IVDs of patients with MC1 experiencing LBP were identified. Post‐treatment MRI suggested improvement in inflammation, speculating that PRP suppressed inflammation and consequently relieved the patient's symptoms. Despite the small number of patients, this treatment is promising for patients with MC1 experiencing LBP. The study protocol has been reviewed and approved by the Certified Committee for Regenerative Medicine and the Japanese Ministry of Health, Labor and Welfare (Japan Registry of Clinical Trials [jRCT] No. jRCTb042210159).
Keywords: intervertebral disc (IVDs), low back pain (LBP), Modic type 1change (MC1), platelet‐rich plasma (PRP), regenerative medicines
This is the first prospective study evaluating the safety and efficacy of platelet‐rich plasma (PRP) administration to the intervertebral discs of patients with Modic type 1 change (MC1) experiencing low back pain. PRP treatment is promising for patients with MC1.

1. INTRODUCTION
Low back pain (LBP) is a highly prevalent symptom. LBP develops in all countries and all age groups around the world; between 1990 and 2015, the number of people living their daily lives with disabilities due to LBP has increased by 54% worldwide. 1 A systematic analysis for the Global Burden of Disease Study 2015 revealed that LBP and neck pain were the leading causes of disability worldwide in most countries. 2 A systematic review showed the highest prevalence of LBP, particularly among women and those aged 40–80 years. 3 Musculoskeletal pain often recurs throughout life. 4 As chronic pain is often intractable, appropriate treatment must be initiated as early as possible.
Modic et al. classified three types of signal changes in the endplate and subchondral bone on magnetic resonance imaging (MRI) and named them Modic changes (MC). 5 , 6 MC on MRI is one of the various causes of LBP. Overall, the frequency of MC is higher in LBP patients (median frequency of 43% in a meta‐analysis) compared with the asymptomatic population (median frequency of 6%). 7 In particular, Modic type 1 change (MC1) has been reported to be more strongly associated with LBP than other types. 8 , 9 , 10 MC1 shows low‐signal intensity on T1‐weighted imaging and high‐signal intensity on T2‐weighted imaging (T2WI), indicating inflammation with edema and hypervascularity of the vertebral body. Meanwhile, some previous clinical studies have indicated that patients with pyogenic spondylitis were included among patients with MC1. 11 , 12 Treatment of MC1 remains controversial, with some reports of antimicrobials being effective 13 , 14 and others ineffective. 15 Sairyo et al. reported a patient with MC1 who was successfully treated with endoscopic surgery, flushing, and drainage. 16 However, no treatment for MC1 has yet been established.
Recently, many regenerative medicines for intractable symptoms have been reported. In particular, platelet‐rich plasma (PRP) has been widely applied in clinical practice for tissue regeneration and anti‐inflammatory effects because of its simple preparation. In the orthopedics field, it has been reported that soft tissues such as damaged tendons, ligaments, and cartilage have been repaired following PRP administration. 17 , 18 PRP contains high levels of growth factors that enhance cell anabolism, inflammatory mediators that provide anti‐inflammatory effects, and fibrinogen that functions as a scaffold for biomaterials. 18 , 19 These activities are expected to have tissue regeneration effects. Taking advantage of these effects, many studies have reported that PRP was a safe and effective treatment for discogenic pain. 20 , 21 However, the safety and efficacy of PRP in patients with MC1 experiencing LBP have not yet been established, although we previously reported two cases. 22 Considering the possibility that MC1 may contain infected cases, PRP injections may promote iatrogenic bacterial discitis. Therefore, this study aimed to determine the effect on reducing pain and disability outcomes of intradiscal injection of autologous PRP in patients with LBP associated with MC1 and to assess the impact of PRP injection on MRI observations.
2. MATERIALS AND METHODS
2.1. Institutional review board statement
All participants provided written informed consent. The study protocol has been reviewed and approved by the Certified Committee for Regenerative Medicine and the Japanese Ministry of Health, Labor and Welfare (Japan Registry of Clinical Trials [jRCT] No. jRCTb042210159). One of the authors (Y.S.) is an employee of Canon Medical Systems Corporation but did not have control over any of the data used in this study.
2.2. Patients
This study included 10 patients with MC1 experiencing LBP who met the same inclusion and exclusion criteria as in the previous study. 22 The main inclusion criteria were patients aged 20–70 years, with visual analog scale (VAS) pain intensity of ≥50 mm that persists for at least 3 months, and with only one level of MC1 change. Patients with other spinal cord diseases were excluded. Details of these criteria are shown in Table 1. Discography was not performed before the inclusion of the patient.
TABLE 1.
Inclusion and exclusion criteria.
| Inclusion criteria |
|
| Exclusion criteria |
|
A flowchart from patient selection to analysis is shown in Figure 1. Written informed consent was obtained from all patients, and they were followed for up to 24 weeks from the primary administration.
FIGURE 1.
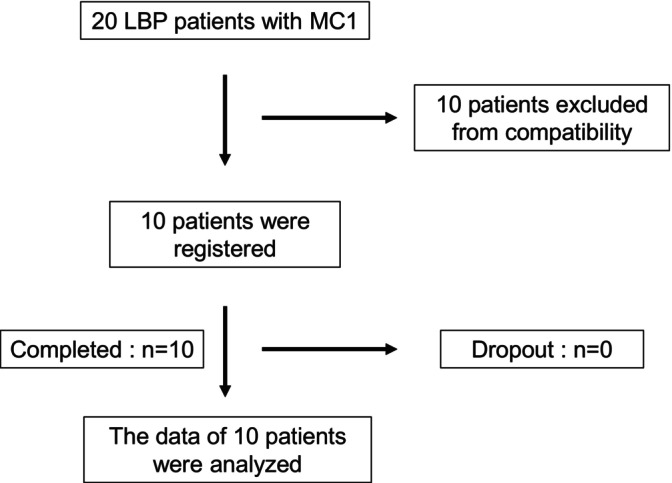
Flowchart from patient selection to analysis. Ten patients were registered in the prospective study, and none dropped out. LBP, low back pain; MC1, Modic type 1 change.
We also evaluated the correlation between fat‐suppressed T2WI and T2* values within the vertebral body and T2* values within the intervertebral disc on MRI and VAS, Oswestry disability index (ODI), and Roland Morris disability questionnaire (RDQ) before administration.
2.3. PRP preparation and procedures
PRP preparation and procedures were performed as previously described. 22 To create PRP, an anticoagulant citrate dextrose solution (Solution A Citra Anticoagulant, Inc., Braintree, MA) was added to the blood collected from the patient (Anticoagulant Citrate Dextrose Solution, 4 mL; venous blood, 26 mL). Samples were gently agitated to ensure adequate mixing of the anticoagulant and blood. Subsequently, 10‐mL blood samples for baseline measurements were collected in a different syringe. Then, 4 mL of PRP was purified from the anticoagulant blood by centrifuging at 3200 rpm for 15 min using the GPS III system (Zimmer Biomet, Warsaw, IN). The PRP used in this study has a high white blood cell (WBC) concentration and is referred to as leukocyte‐rich PRP (LR‐PRP). Of these, 2 mL was administered to the patient and 1 mL was analyzed for components. The patient was placed in the left lateral position and the puncture was made from the right side under fluoroscopy. After disinfection, local anesthesia was injected and a Hijikata discographic needle (19G) was inserted. The needle was advanced to the target disc and 2‐mL PRP was injected. The patient was admitted to a medical institution for follow‐up and discharged the day after treatment.
2.4. Outcome measures
Primarily, physical condition, laboratory data, and changes in disc height on lumbar x‐ray images were evaluated to assess safety. Specifically, adverse events were defined as worsening of physical conditions (worsening of neurological symptoms, fever >37.5°C, redness, swelling, heat at the site of administration, or marked worsening of back pain), deviation in laboratory data from baseline, and worsening of findings on x‐ray images according to the study protocol. Furthermore, we assessed whether analgesic medications were taken during the trial.
Secondarily, to assess the efficacy of PRP, patient‐reported outcomes were examined, including the VAS (0–100 mm) for back pain and the ODI, 23 and RDQ 24 for back pain‐related disability. Furthermore, whether laboratory data and PRP composition were significantly associated with the improvement rate in the VAS, ODI, and RDQ was evaluated.
2.5. MRI protocols
All magnetic resonance (MR) examinations were performed using 3T MR systems (Vantage Centurian, Canon Medical Systems Corporation, Otawara, Tochigi, Japan). In each patient, fat‐suppressed T2WI was performed using the following scanning parameters: Repetition Time (TR)/Echo Time (TE), 4435 ms/99 ms; echo train length, 19; slice thickness, 3 mm; number of excitation (NEX), 1; field of view (FOV), 300 × 300 mm; 384 × 224 matrix; 768 × 768 reconstruction matrix. For T2* measurements in each patient, MRI with ultra‐short echo times (UTEs) was performed with 3D radial gradient‐echo sequence using the following parameters: TR/TE, 14 ms/0.14, 2.74, 5.34, and 7.94 ms; flip angle, 5°; slice thickness, 1.5 mm; NEX, 1; FOV, 256 × 256 mm; parallel imaging factor, 1; number of slices, 150; 256 × 256 acquisition matrix; 256 × 256 reconstruction matrix. Reconstructed multiple TE images were transferred from the MRI scanner to the image analysis workstation (Vitrea, Canon Medical Systems) and automatically identified and fitted to the relaxation model pixel‐by‐pixel using T2* map analysis software (Relaxometry). To assess the effect of PRP administration on MC1, MRI was performed 24 weeks after treatment and the degree of inflammation was measured using fat‐suppressed T2WI and T2* values. To measure the high‐signal intensity region on fat‐suppressed T2WI, the high‐signal region was quantified in all sagittal images that showed high‐signal intensity and calculated by adding them all together. To measure T2* values, the entire two vertebrae exhibiting MC1 were considered as regions of interest (Figure 2). These were performed following previously described methods. 22 Furthermore, to evaluate the effect of PRP on the target intervertebral discs, T2* values of the discs were evaluated before and 24 weeks after PRP administration. Furthermore, the presence of progressive degeneration was evaluated using the Pfirrmann grades.
FIGURE 2.
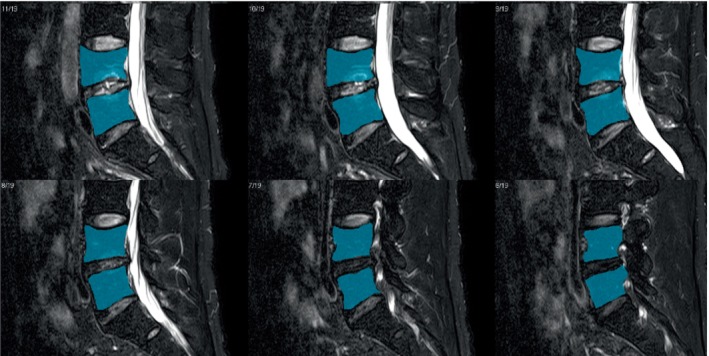
Region of interest for measuring T2* values. The entirety of the two vertebrae showing Modic changes was set as the region of interest (blue regions) when measuring T2* values.
2.6. Image analysis
To measure high‐signal intensity regions on fat‐suppressed T2WI and T2* values, all fat‐suppressed T2WI and UTEs data were transferred to a workstation (Vitrea, Canon Medical Systems) for analysis using quantitative analysis software (Analysis, Canon Medical Systems). The volume of the high‐signal intensity regions on fat‐suppressed T2WI was measured using the segmentation tool on the analysis software. To measure T2* values, volume of interests were placed over the target vertebra and disc.
2.7. Statistical analysis and data collection
Comparisons in T2* MRI value, T2WI high‐signal volume, and disc height on lumbar x‐ray were performed using t‐tests. The Wilcoxon signed‐rank test was used to analyze the Pfirrmann grades. Comparisons over time in blood tests and VAS, ODI, and RDQ were performed using mixed‐effects models for repeated measures. Spearman's correlation coefficient was used to assess the correlation between variables. SAS 9.4 (SAS Institute, Cary, NC) was used for the analysis. The means ± standard deviations of the data are presented. To minimize bias, participant randomization and data collection, management, and analysis were conducted by a third‐party contract research organization company (Soiken Inc., Tokyo, Japan).
3. RESULTS
3.1. Patient characteristics
This study prospectively enrolled 10 patients with MC1 experiencing LBP (Figure 1). Ten patients who did not meet the inclusion or exclusion criteria or did not wish to participate in the study were excluded. The baseline characteristics of all patients, including blood data of whole blood and PRP, are shown in Table 2. Detailed participant information including smoking and alcohol history, occupation, and sports history is shown in Table 3.
TABLE 2.
Patients baseline characteristics.
| Age | 53.3 ± 8.7 |
| Height (cm) | 166.5 ± 10.1 |
| Body weight (kg) | 62.4 ± 13.0 |
| Body mass index (kg/m2) | 22.3 ± 3.0 |
| Pain duration (months) | 15.0 ± 10.2 |
| VAS (mm) | 70.0 ± 13.3 |
| ODI (%) | 41.8 ± 11.9 |
| RDQ | 12.5 ± 4.3 |
| Blood data of whole blood | |
| WBC (×103mL/μL) | 5.43 ± 1.69 |
| PLT (×104 mL/μL) | 24.1 ± 4.7 |
| AST (U/L) | 20.3 ± 4.8 |
| ALT (U/L) | 17.4 ± 5.9 |
| BUN/Cr | 20.2 ± 5.9 |
| CRP (mg/dL) | 0.043 ± 0.045 |
| Blood data of PRP | |
| WBC (×103mL/μL) | 20.7 ± 9.7 |
| PLT (×104 mL/μL) | 97.7 ± 46.5 |
| Distribution of levels (no. of cases) | |
| L2/3 | 1 |
| L4/5 | 5 |
| L5/S | 4 |
Abbreviations: ODI, Oswestry disability index; PRP, platelet‐rich plasma; RDQ, Roland Morris disability questionnaire; VAS, visual analog scale; WBC, white blood cell.
TABLE 3.
Detailed information of the patients.
| Sex | |
| Male | 4 |
| Female | 6 |
| Smoking history | |
| Never | 3 |
| Used to smoke | 3 |
| <1 pack per day | 2 |
| 1–2 packs per day | 2 |
| >2 packs per day | 0 |
| Alcohol history | |
| Don't drink | 2 |
| 1–3 times per month | 4 |
| Once per week | 1 |
| 3–4 times per week | 1 |
| 6–7 times per week | 2 |
| Occupation | |
| Desk worker | 2 |
| Sales staff | 4 |
| Housewife/househusband | 1 |
| Others | 3 |
| Sports history | |
| Yes | 7 |
| No | 3 |
| Baseball | 1 |
| Soccer | 3 |
| Volleyball | 1 |
| Swimming | 2 |
| Others | 7 |
No significant correlation was observed between fat‐suppressed T2WI and T2* values in the vertebral body and T2* values in the intervertebral disc on MRI and VAS, ODI, and RDQ before PRP administration (Table 4).
TABLE 4.
Relationship between magnetic resonance findings and visual analog scale (VAS), Oswestry disability index (ODI), and Roland Morris disability questionnaire (RDQ) before administration.
| Spearman correlation | |||
|---|---|---|---|
| Correlation coefficient | p Value | ||
| Fat‐suppressed T2WI in the vertebral body | VAS | −0.21 | 0.56 |
| ODI | −0.21 | 0.57 | |
| RDQ | −0.24 | 0.51 | |
| T2* values in the vertebral body | VAS | 0.54 | 0.13 |
| ODI | 0.13 | 0.75 | |
| RDQ | 0.03 | 0.95 | |
| T2* values in the intervertebral disc | VAS | 0.18 | 0.64 |
| ODI | 0.42 | 0.26 | |
| RDQ | 0.48 | 0.19 | |
Abbreviation: T2WI, T2‐weighted imaging.
3.2. Physical condition, laboratory data, and x‐ray data
PRP administration was performed without complications. No adverse events in physical condition, laboratory data, or disc height on lumbar x‐ray data occurred after PRP administration. Specifically, no significant differences in WBC count and levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), blood urea nitrogen (BUN)/creatinine (Cr), and CRP were observed over time. Disc height also did not significantly differ over time (Table 5). In one patient, LBP worsened after the injection but eventually improved. Only three patients used analgesics as needed. One took them only once, and the other two took them multiple times. No patients requested additional treatment 24 weeks after PRP administration.
TABLE 5.
Magnetic resonance findings and disc height changes on x‐ray.
| Observation points | Value | p Value | ||
|---|---|---|---|---|
| Mean T2* MRI value of the entire two vertebrae | T2 value (ms) | Before administration | 4.47 ± 0.64 | |
| 24 Weeks after administration | 4.30 ± 0.67 | |||
| Mean changes | 24 Weeks after administration | −0.16 ± 0.34 | 0.19 | |
| Fat‐suppressed T2WI high‐signal volume of the two vertebrae | Volume (mm2) | Before administration | 10.07 ± 5.73 | |
| 24 Weeks after administration | 7.89 ± 5.01 | |||
| Mean changes | 24 Weeks after administration | −2.18 ± 1.96 | 0.007 | |
| Mean T2* MRI value of the targeted disc | T2 value (ms) | Before administration | 16.85 ± 3.24 | |
| 24 Weeks after administration | 16.47 ± 2.58 | |||
| Mean changes | 24 Weeks after administration | −0.38 ± 2.54 | 0.67 | |
| Mean intervertebral disc height of the target on x‐ray | Disc height (mm) | Before administration | 4.96 ± 2.05 | |
| 4 Weeks after administration | 4.90 ± 2.06 | |||
| 12 Weeks after administration | 4.89 ± 1.91 | |||
| 24 Weeks after administration | 4.93 ± 1.86 | |||
| Mean changes | 4 Weeks after administration | −0.06 ± 0.35 | 0.62 | |
| 12 Weeks after administration | −0.07 ± 0.57 | 0.71 | ||
| 24 Weeks after administration | −0.03 ± 0.42 | 0.83 | ||
Abbreviations: MRI, magnetic resonance imaging; T2WI, T2‐weighted imaging.
3.3. VAS, ODI, and RDQ
Changes in the mean VAS, ODI, and RDQ are shown in Figure 3. The mean VAS was 70.0 ± 13.3 before administration, which significantly decreased 1 week after PRP administration to 39.0 ± 28.8 at the last observation (p = 0.008). The ODI and RDQ promptly improved after PRP administration. The pre‐administration ODI and RDQ were 41.8% ± 11.9% and 12.5 ± 4.3, respectively; however, they significantly improved to 26.2% ± 17.8% (p = 0.039) and 7.2 ± 5.5 (p = 0.047), respectively, at the last observation. Table 6 shows the relationship between laboratory data and PRP composition and the improvements rate in VAS, ODI, and RDQ. Significant improvements in VAS and ODI were observed in patients with higher PLT counts on pretreatment blood tests. Furthermore, significant improvement in VAS was observed in patients with higher WBC counts in PRP.
FIGURE 3.
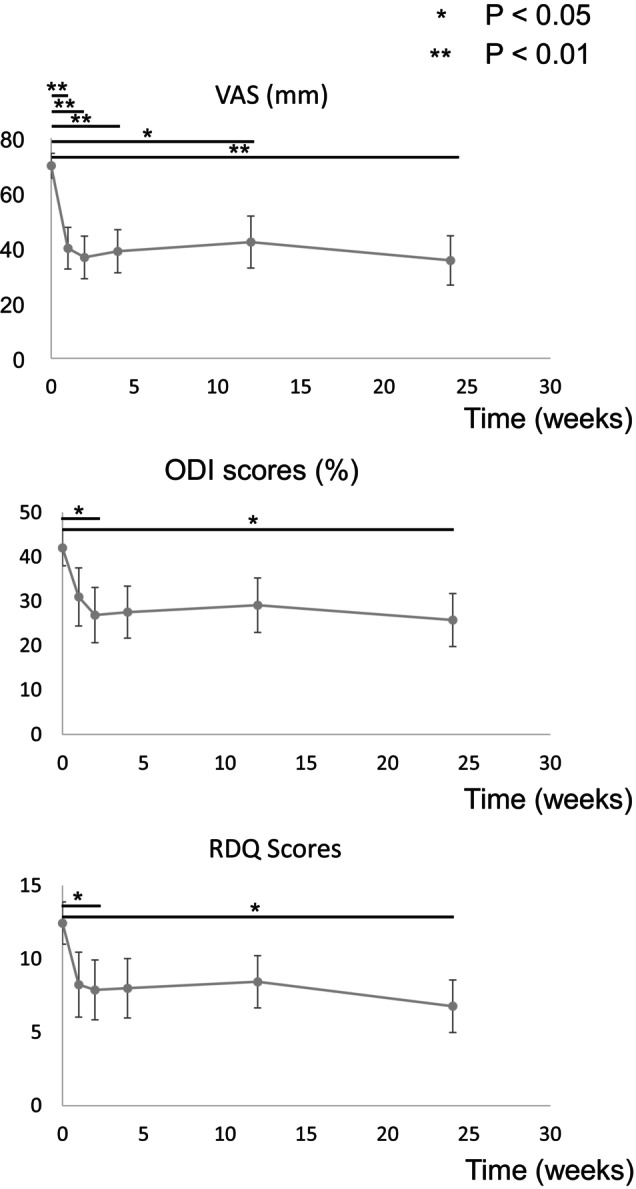
Changes in mean visual analog scale (VAS), Oswestry disability index (ODI), and Roland Morris disability questionnaire (RDQ). Values are presented as mean ± standard deviations. The mean VAS before treatment was 70.0 ± 13.3, which decreased significantly 1 week after administration, and was 39.0 ± 28.8 at the last observation. ODI and RDQ improved promptly after administration. At the last observation, both improved with significant differences.
TABLE 6.
Relationship between laboratory data and platelet‐rich plasma (PRP) composition and the rate of improvement in visual analog scale (VAS), Oswestry disability index (ODI), and Roland Morris disability questionnaire (RDQ).
| Improvement rate | Spearman correlation | |||
|---|---|---|---|---|
| Correlation coefficient | p Value | |||
| Laboratory data | WBC (×103/μL) | VAS | −0.18 | 0.65 |
| ODI | −0.30 | 0.43 | ||
| RDQ | −0.33 | 0.38 | ||
| PLT (×104/μL) | VAS | −0.84 | 0.005 | |
| ODI | −0.73 | 0.025 | ||
| RDQ | −0.43 | 0.24 | ||
| AST (U/L) | VAS | −0.41 | 0.27 | |
| ODI | 0.06 | 0.88 | ||
| RDQ | −0.06 | 0.88 | ||
| ALT (U/L) | VAS | −0.13 | 0.74 | |
| ODI | 0.06 | 0.88 | ||
| RDQ | 0.10 | 0.80 | ||
| BUN/Cr | VAS | 0.44 | 0.23 | |
| ODI | 0.17 | 0.67 | ||
| RDQ | 0.15 | 0.70 | ||
| CRP (mg/dL) | VAS | 0.13 | 0.74 | |
| ODI | 0.13 | 0.74 | ||
| RDQ | 0.03 | 0.95 | ||
| PRP composition | WBC (×103/μL) | VAS | −0.79 | 0.006 |
| ODI | −0.48 | 0.16 | ||
| RDQ | −0.28 | 0.43 | ||
| PLT (×104/μL) | VAS | −0.40 | 0.25 | |
| ODI | −0.43 | 0.21 | ||
| RDQ | −0.42 | 0.23 | ||
Abbreviation: WBC, white blood cell.
3.4. Magnetic resonance imaging
Follow‐up MRI 24 weeks after treatment revealed a significant decrease in the mean high‐signal intensity on fat‐suppressed T2WI from 10.1 mm2 before PRP administration to 7.90 mm2 (p = 0.007) (Table 5). Representative images are shown in Figure 4. Mean T2* values tended to decrease after PRP administration, but not significantly. No patient changed from MC1 to any other type. The mean T2* values in the intervertebral disc were not altered with PRP administration (Table 5). No alterations in the Pfirrmann grade were observed after PRP administration.
FIGURE 4.
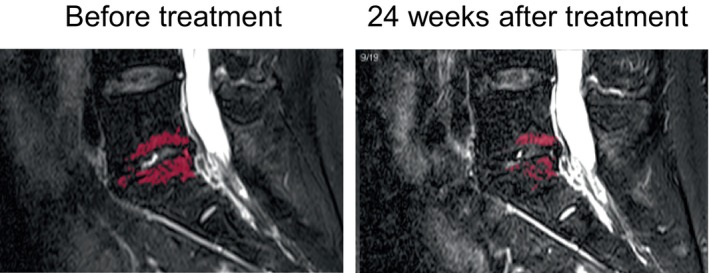
Representative images of fat*suppressed T2‐weighted imaging (T2WI) before and 24 weeks after treatment. Fat‐suppressed T2WI high volume (red color regions) decreased from 6.55 to 3.13 mm2.
4. DISCUSSION
To the best of our knowledge, this is the first prospective study to evaluate the safety and efficacy of PRP administration to IVDs in patients with LBP, targeting MC1 only. A number of studies have documented that much of chronic LBP is caused by discogenic pain. 25 Previous treatments have included pharmacotherapy, physical therapy, injections, and surgical approaches. In recent years, several regenerative medicine‐based treatments for LBP have been proposed, including the injection of mesenchymal stem cells (MSCs) and PRP. According to a 2023 systematic review of PRP therapy, 26 the first clinical trial of intradiscal administration of PRP was conducted by Tuakli‐Wosornu in 2016, 21 and 13 trials have been reported to date. Two randomized controlled studies, five prospective cohort studies, three retrospective cohort studies, and three case reports have been published. Most target diseases were discogenic LBP. This review summarizes that intradiscal administration of PRP is safe and effective for managing pain. Note that two trials included several patients with MC1 27 , 28 ; however, no clinical trials have included only patients with MC1, as in this study. Other systematic reviews and meta‐analyses showed that both MSC and PRP may be effective in the management of discogenic LBP; however, the level of evidence varied, and no high‐quality randomized controlled trials have been conducted. 26 , 29 , 30 , 31 , 32 More powered high‐quality studies are needed to truly assess the long‐term safety and efficacy of regenerative medicine approaches in discogenic LBP.
In this study, we have successfully demonstrated the efficacy of intradiscal administration of PRP in patients with MC1, who are known to have more refractory chronic LBP. Platelets have three main granules including dense granules, α‐granules, and lysosomes. 33 Of these, α‐granules are the largest and the most abundant platelet granules, in one platelet contain 50–80 granules. Vesicles called exosomes are present in the α‐granules and are released when platelets are activated. 34 Proteomic studies have shown that >300 bioactive proteins are released from activated α‐granules, 35 , 36 and these proteins could play multiple roles in hemostasis, anti‐inflammation, antimicrobial activity, angiogenesis, and wound healing. 33 In this study, these effects of PRP are considered to have reduced the inflammation of the MC1‐affected spine and consequently improved LBP. In addition, MRI results quantitatively validated the decreased vertebral inflammation.
PRP is divided into two categories according to leukocyte concentration: PRP with low WBC concentrations are called leukocyte‐poor PRP (LP‐PRP), and those with higher WBC concentrations are called LR‐PRP. With the GPS III system used in this study, LR‐PRP is created. The use of a kit like GPS III system is expected to reduce complications and produce a more stable PRP. In particular, the GPS III system can be used to purify stable LR‐PRP in a single centrifugation. Furthermore, in vitro experiments using the GPS III system have revealed that PRP suppressed cytokine‐induced inflammatory degradative enzymes and mediators in human nucleus pulposus cells. 37 Because of its simplicity and in vitro data, the GPS III system was used in this study. A variety of studies have reported on the influence of leukocyte concentration on the efficacy of PRP; however, there is no consensus yet. 38 , 39 Clinical research studies for patellar tendonitis, 40 Achilles tendinopathy, 41 and lateral epicondylitis 42 have reported no difference in results between LP‐PRP and LR‐PRP. In this study, in patients with more WBCs in PRP, the VAS significantly improved, suggesting that LR‐PRP is useful in the administration of PRP to patients with MC1. According to Intravia et al., both LP‐PRP and LR‐PRP showed antimicrobial activity in in vivo experiments; however no significant difference was observed. 43 In contrast, López et al. reported that LR‐PRP showed better bacteriostatic effect against Methicillin‐Susceptible Staphylococcus Aureus (MSSA). 44 MC1 includes a few cases of pyogenic discitis, and the antimicrobial action of PRP is speculated to be beneficial. A less than two‐fold to an 8.5‐fold increase from baseline in platelet concentration ratios in PRP have been reported. 45 , 46 A constant platelet concentration is important because low platelet concentrations tend to decrease the number of growth factors. In this study, higher PLT counts in blood tests were more effective for LBP, suggesting that the PLT count in blood samples influence the effectiveness of PRP. Cost‐effectiveness is often a hot topic in PRP treatments. Although this study was conducted on patients with lateral epicondylitis, Klifto et al. reported that PRP injections were superior to steroid injections in terms of cost‐effectiveness. 47 Furthermore, intradiscal administration is expected to be highly cost‐effective if its efficacy is further demonstrated in the future.
Modic and Ross noted that LBP in patients with MC1 may be related to unusual stresses, micro‐ or macroinstability, or microtrauma. 48 Patients with MC experiencing LBP had more frequent, longer‐lasting LBP episodes and more frequent hospital visits. 7 , 49 A recent study showed that conservative treatment for chronic LBP patients with MC1 is associated with poor prognosis. 50 MC1 patients have also been reported to have a poor prognosis after discectomy, 51 which implies that the vertebrae may be the source of the pain. In this study, PRP administration resulted in signal changes in the bone marrow but not in the intervertebral discs. In pathology, fibrous tissue replaces normal bone marrow between trabecular bones in MC1. In MC1, vascular granulation tissue is proliferating at the bone‐disc junction, suggesting inflammation. Thus, MC1 is not merely a coincidental MRI finding in LBP patients, but is likely to indicate a condition that should be targeted for treatment. 52 Non‐surgical treatments, including intradiscal steroid injections, anti‐tumor necrosis factor (TNF)‐α antibodies, and antibiotic administration, have been reported to have short‐term efficacy in MC patients, but long‐term results are unknown. Albert et al. treated patients with MC1 with antibiotics for 100 days and reported that the VAS of patient with LBP improved from 67 to 37 mm after 1 year. 13 Zhou et al. reported the short‐term benefit of calcitonin for LBP in patients with MC1. After 3 months of treatment, they reported that the VAS improved from 62.5 to 37.0 mm in the calcitonin group, whereas the VAS improved only from 63.4 to 45 mm in the control diclofenac group. 53 Although not directly comparable, the results of this study suggest that PRP administration is as effective or more effective than these treatments. Although the detailed mechanism of vertebral pain in MC is unclear, higher numbers of Protein Gene Product (PGP)‐9.5 nerve fibers and TNF‐α‐positive cells have been reported in MC1 and MC2 endplates. 54 , 55 Nerves are reported to be more abundant in endplates with fibrovascular marrow, and nerve density is significantly higher in fibrovascular endplate marrow than in normal endplate marrow. 55 Differences in nerve density may contribute to vertebrogenic pain in MC. In this study, we speculated that the PRP injected into the IVDs passed through the endplate defect and reached and acted on the bone marrow, which is the target of treatment. We believe that disc degeneration‐related reduction in cells and nutrient availability may affect the action of PRP 56 , 57 ; however, assessing this in this study was difficult because of the lack of sufficient cases. Therefore, further research is required from the view point of both basic science and clinical medicine in the future. Furthermore, based on the MRI results, which showed a significant reduction in high‐signal intensity on fat‐suppressed T2WI after PRP administration, we believe that we were able to quantify the improvement in inflammation. Meanwhile, Crockett et al. reported that MC1 vertebral endplate changes were secondary to injury, inflammation, or infection and require appropriate treatment. 58 A small number of patients with MC1 have pyogenic discitis. 11 , 12 One patient had worsening back pain after PRP administration. However, follow‐up x‐ray and MRI imaging and blood tests showed no findings suggestive of discitis. The pain appeared immediately after administration and was considered likely iatrogenic pain caused by needle puncture. The severe pain gradually improved and was not considered a serious adverse event. Despite the limited number of cases, the most significant study finding was the confirmation of the safety of PRP administration in all patients with MC1. Kerttula et al. reported a 1‐year follow‐up of patients with MC1 and a significant decrease in disc height. 59 Furthermore, disc puncture itself may promote disc degeneration. 53 , 60 Therefore, although the transpedicular approach is an option, 61 we considered it more invasive and performed disc puncture. As a result, although the follow‐up period for this study was only 6 months, PRP administration did not reduce the disc height. Therefore, it is possible that PRP administration may have inhibited the progression of disc degeneration. However, more cases are needed to further confirm safety.
This study has several limitations to consider. First, the study population is quite small. In future research, the number of cases must be increased. Second, no control group was employed with which to compare outcomes. To confirm our promising results, a larger prospective randomized controlled trial is needed. Third, determining that MC 1 caused LBP was difficult. Discography may be necessary to accurately diagnose MC1 as the cause of LBP. However, discography, an invasive test, was not performed in this study because a significant association was established between MC1 and LBP. Despite these limitations, this is the first prospective study of PRP administration to the IVDs of patients with MC1 experiencing LBP. In particular, we believe that the validated safety of this treatment would lead to further clinical research.
5. CONCLUSION
The safety and efficacy of PRP administered to the IVDs of patients with MC1 experiencing LBP were identified. No major adverse events due to PRP administration were observed, and pyogenic discitis did not occur. Furthermore, MRI after PRP administration showed improvements in imaging features indicative of inflammation, suggesting that PRP could reduce the inflammation area caused by MC1 and relieve the patient's symptoms. Considering the small number of patients in this study, further studies are required to validate the safety and efficacy of PRP treatment.
AUTHOR CONTRIBUTIONS
Each author has read and approved the final submitted manuscript. NF and SoK designed the study. SoK and NF wrote the initial draft of the manuscript. SN, KI, HT, DI, YK, YukS, YuiS, YO, and ShK contributed to data analysis and interpretation. All the authors provided critical revision of the manuscript for important intellectual content. All authors are responsible for the overall contents of the article.
CONFLICT OF INTEREST STATEMENT
The research institution received research support from Zimmer Biomet. This study was technically supported by Canon Medical Systems Corporation. Dr. Yoshiharu Ohno has a research grant from Canon Medical Systems Corporation.
ACKNOWLEDGMENTS
This research is funded by Zimmer Biomet, Warsaw, IN (grant number: IRA2021‐31B).
Kawabata S, Nagai S, Ito K, et al. Intradiscal administration of autologous platelet‐rich plasma in patients with Modic type 1 associated low back pain: A prospective pilot study. JOR Spine. 2024;7(1):e1320. doi: 10.1002/jsp2.1320
REFERENCES
- 1. Hartvigsen J, Hancock MJ, Kongsted A, et al. What low back pain is and why we need to pay attention. Lancet. 2018;391(10137):2356‐2367. doi: 10.1016/s0140-6736(18)30480-x [DOI] [PubMed] [Google Scholar]
- 2. GBD 2017 Disease and Injury Incidence and Prevalence Collaborators , James SL, Abate D, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990‐2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. 2016;388(10053):1545‐1602. doi: 10.1016/s0140-6736(16)31678-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Hoy D, Bain C, Williams G, et al. A systematic review of the global prevalence of low back pain. Arthritis Rheum. 2012;64(6):2028‐2037. doi: 10.1002/art.34347 [DOI] [PubMed] [Google Scholar]
- 4. Kamper SJ, Henschke N, Hestbaek L, Dunn KM, Williams CM. Musculoskeletal pain in children and adolescents. Braz J Phys Ther. 2016;20(3):275‐284. doi: 10.1590/bjpt-rbf.2014.0149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Modic MT, Masaryk TJ, Ross JS, Carter JR. Imaging of degenerative disk disease. Radiology. 1988;168(1):177‐186. doi: 10.1148/radiology.168.1.3289089 [DOI] [PubMed] [Google Scholar]
- 6. Modic MT, Steinberg PM, Ross JS, Masaryk TJ, Carter JR. Degenerative disk disease: assessment of changes in vertebral body marrow with MR imaging. Radiology. 1988;166(1):193‐199. doi: 10.1148/radiology.166.1.3336678 [DOI] [PubMed] [Google Scholar]
- 7. Jensen TS, Karppinen J, Sorensen JS, Niinimäki J, Leboeuf‐Yde C. Vertebral endplate signal changes (Modic change): a systematic literature review of prevalence and association with non‐specific low back pain. Eur Spine J. 2008;17(11):1407‐1422. doi: 10.1007/s00586-008-0770-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Brinjikji W, Diehn FE, Jarvik JG, et al. MRI findings of disc degeneration are more prevalent in adults with low back pain than in asymptomatic controls: a systematic review and meta‐analysis. AJNR Am J Neuroradiol. 2015;36(12):2394‐2399. doi: 10.3174/ajnr.A4498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Thompson KJ, Dagher AP, Eckel TS, Clark M, Reinig JW. Modic changes on MR images as studied with provocative diskography: clinical relevance – a retrospective study of 2457 disks. Radiology. 2009;250(3):849‐855. doi: 10.1148/radiol.2503080474 [DOI] [PubMed] [Google Scholar]
- 10. Järvinen J, Karppinen J, Niinimäki J, et al. Association between changes in lumbar Modic changes and low back symptoms over a two‐year period. BMC Musculoskelet Disord. 2015;16(1):98. doi: 10.1186/s12891-015-0540-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ohtori S, Koshi T, Yamashita M, et al. Existence of pyogenic spondylitis in Modic type 1 change without other signs of infection: 2‐year follow‐up. Eur Spine J. 2010;19(7):1200‐1205. doi: 10.1007/s00586-010-1358-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ninomiya K, Fujita N, Hosogane N, et al. Presence of Modic type 1 change increases risk of postoperative pyogenic discitis following decompression surgery for lumbar canal stenosis. J Orthop Sci. 2017;22(6):988‐993. doi: 10.1016/j.jos.2017.07.003 [DOI] [PubMed] [Google Scholar]
- 13. Albert HB, Sorensen JS, Christensen BS, Manniche C. Antibiotic treatment in patients with chronic low back pain and vertebral bone edema (Modic type 1 changes): a double‐blind randomized clinical controlled trial of efficacy. Eur Spine J. 2013;22:697‐707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wong AYL, Mallow GM, Pinto SM, et al. The efficacy and safety of oral antibiotic treatment in patients with chronic low back pain and Modic changes: a systematic review and meta‐analysis. JOR Spine. 2023;7(1):e1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bråten LCH, Rolfsen MP, Espeland A, et al. Efficacy of antibiotic treatment in patients with chronic low back pain and Modic changes (the AIM study): double blind, randomised, placebo controlled, multicentre trial. BMJ. 2019;367:l5654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sairyo K, Maeda T, Yamashita K, et al. A new surgical strategy for the intractable chronic low back pain due to type 1 Modic change using transforaminal full‐endoscopic disc cleaning (FEDC) surgery under the local anesthesia: a case report and literature review. J Med Invest. 2021;68:1‐5. [DOI] [PubMed] [Google Scholar]
- 17. Alsousou J, Ali A, Willett K, Harrison P. The role of platelet‐rich plasma in tissue regeneration. Platelets. 2013;24(3):173‐182. doi: 10.3109/09537104.2012.684730 [DOI] [PubMed] [Google Scholar]
- 18. Xie X, Zhang C, Tuan RS. Biology of platelet‐rich plasma and its clinical application in cartilage repair. Arthritis Res Ther. 2014;16(1):204. doi: 10.1186/ar4493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Anitua E, Andia I, Ardanza B, Nurden P, Nurden A. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91(1):4‐15. doi: 10.1160/th03-07-0440 [DOI] [PubMed] [Google Scholar]
- 20. Akeda K, Ohishi K, Takegami N, et al. Platelet‐rich plasma releasate versus corticosteroid for the treatment of discogenic low back pain: a double‐blind randomized controlled trial. J Clin Med. 2022;11(2):304. doi: 10.3390/jcm11020304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Tuakli‐Wosornu YA, Terry A, Boachie‐Adjei K, et al. Lumbar Intradiskal platelet‐rich plasma (PRP) injections: a prospective, double‐blind, randomized controlled study. PM and R. 2016;8(1):1‐10; quiz 10. doi: 10.1016/j.pmrj.2015.08.010 [DOI] [PubMed] [Google Scholar]
- 22. Kawabata S, Hachiya K, Nagai S, et al. Autologous platelet‐rich plasma administration on the intervertebral disc in low back pain patients with modic type 1 change: report of two cases. Medicina. 2023;59(1):112. doi: 10.3390/medicina59010112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Fairbank JC, Couper J, Davies JB. The Oswestry low back pain disability questionnaire. Physiotherapy. 1980;66:271‐273. [PubMed] [Google Scholar]
- 24. Roland M, Morris R. A study of the natural history of back pain. Part I: development of a reliable and sensitive measure of disability in low‐back pain. Spine. 1983;8(2):141‐144. [DOI] [PubMed] [Google Scholar]
- 25. Zhao L, Manchikanti L, Kaye AD, Abd‐Elsayed A. Treatment of discogenic low back pain: current treatment strategies and future options – a literature review. Curr Pain Headache Rep. 2019;23(11):86. doi: 10.1007/s11916-019-0821-x [DOI] [PubMed] [Google Scholar]
- 26. Kawabata S, Akeda K, Yamada J, et al. Advances in platelet‐rich plasma treatment for spinal diseases: a systematic review. Int J Mol Sci. 2023;24(8):7677. doi: 10.3390/ijms24087677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Levi D, Horn S, Tyszko S, Levin J, Hecht‐Leavitt C, Walko E. Intradiscal platelet‐rich plasma injection for chronic discogenic low back pain: preliminary results from a prospective trial. Pain Med. 2016;17:1010‐1022. [DOI] [PubMed] [Google Scholar]
- 28. Zhang J, Liu D, Gong Q, Chen J, Wan L. Intradiscal autologous platelet‐rich plasma injection for discogenic low back pain: a clinical trial. Biomed Res Int. 2022;2022:9563693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Sanapati J, Manchikanti L, Atluri S, et al. Do regenerative medicine therapies provide long‐term relief in chronic low back pain: a systematic review and metaanalysis. Pain Phys. 2018;21(6):515‐540. [PubMed] [Google Scholar]
- 30. Basso M, Cavagnaro L, Zanirato A, et al. What is the clinical evidence on regenerative medicine in intervertebral disc degeneration? Musculoskelet Surg. 2017;101(2):93‐104. doi: 10.1007/s12306-017-0462-3 [DOI] [PubMed] [Google Scholar]
- 31. Wu T, Song HX, Dong Y, Li JH. Cell‐based therapies for lumbar discogenic low back pain: systematic review and single‐arm meta‐analysis. Spine. 2018;43(1):49‐57. doi: 10.1097/brs.0000000000001549 [DOI] [PubMed] [Google Scholar]
- 32. Sakai D, Schol J, Watanabe M. Clinical development of regenerative medicine targeted for intervertebral disc disease. Medicina. 2022;58(2):267. doi: 10.3390/medicina58020267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Flaumenhaft R, Sharda A. Platelet secretion. In: Michelson A, ed. Platelets. Academic Press; 2019:349‐370. [Google Scholar]
- 34. Heijnen HF, Schiel AE, Fijnheer R, Geuze HJ, Sixma JJ. Activated platelets release two types of membrane vesicles: microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha‐granules. Blood. 1999;94(11):3791‐3799. [PubMed] [Google Scholar]
- 35. Coppinger JA, Cagney G, Toomey S, et al. Characterization of the proteins released from activated platelets leads to localization of novel platelet proteins in human atherosclerotic lesions. Blood. 2004;103(6):2096‐2104. doi: 10.1182/blood-2003-08-2804 [DOI] [PubMed] [Google Scholar]
- 36. Nurden AT. Platelets, inflammation and tissue regeneration. Thromb Haemost. 2011;105(Suppl 1):S13‐S33. doi: 10.1160/THS10-11-0720 [DOI] [PubMed] [Google Scholar]
- 37. Kim HJ, Yeom JS, Koh YG, et al. Anti‐inflammatory effect of platelet‐rich plasma on nucleus pulposus cells with response of TNF‐α and IL‐1. J Orthop Res. 2014;32:551‐556. [DOI] [PubMed] [Google Scholar]
- 38. Rossi LA, Murray IR, Chu CR, Muschler GF, Rodeo SA, Piuzzi NS. Classification systems for platelet‐rich plasma. Bone Joint J. 2019;101‐B(8):891‐896. doi: 10.1302/0301-620x.101b8.Bjj-2019-0037.R1 [DOI] [PubMed] [Google Scholar]
- 39. D'Asta F, Halstead F, Harrison P, Orlandini SZ, Moiemen N, Lord J. The contribution of leucocytes to the antimicrobial activity of platelet‐rich plasma preparations: a systematic review. Platelets. 2018;29(1):9‐20. doi: 10.1080/09537104.2017.1317731 [DOI] [PubMed] [Google Scholar]
- 40. Scott A, LaPrade RF, Harmon KG, et al. Platelet‐rich plasma for patellar tendinopathy: a randomized controlled trial of leukocyte‐rich PRP or leukocyte‐poor PRP versus saline. Am J Sports Med. 2019;47(7):1654‐1661. doi: 10.1177/0363546519837954 [DOI] [PubMed] [Google Scholar]
- 41. Hanisch K, Wedderkopp N. Platelet‐rich plasma (PRP) treatment of noninsertional Achilles tendinopathy in a two case series: no significant difference in effect between leukocyte‐rich and leukocyte‐poor PRP. Orthop Res Rev. 2019;11:55‐60. doi: 10.2147/orr.S187638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Yerlikaya M, Talay Çaliş H, Tomruk Sütbeyaz S, et al. Comparison of effects of leukocyte‐rich and leukocyte‐poor platelet‐rich plasma on pain and functionality in patients with lateral epicondylitis. Arch Rheumatol. 2018;33(1):73‐79. doi: 10.5606/ArchRheumatol.2018.6336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Intravia J, Allen DA, Durant TJ, et al. In vitro evaluation of the anti‐bacterial effect of two preparations of platelet rich plasma compared with cefazolin and whole blood. Muscles Ligaments Tendons J. 2014;4(1):79‐84. [PMC free article] [PubMed] [Google Scholar]
- 44. López C, Alvarez ME, Carmona JU. Temporal bacteriostatic effect and growth factor loss in equine platelet components and plasma cultured with methicillin‐sensitive and methicillin‐resistant Staphylococcus aureus: a comparative in vitro study. Vet Med Int. 2014;2014:525826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Kushida S, Kakudo N, Morimoto N, et al. Platelet and growth factor concentrations in activated platelet‐rich plasma: a comparison of seven commercial separation systems. J Artif Organs. 2014;17:186‐192. [DOI] [PubMed] [Google Scholar]
- 46. Eppley BL, Pietrzak WS, Blanton M. Platelet‐rich plasma: a review of biology and applications in plastic surgery. Plast Reconstr Surg. 2006;118:147e‐159e. [DOI] [PubMed] [Google Scholar]
- 47. Klifto KM, Colbert SH, Richard MJ, Anakwenze OA, Ruch DS, Klifto CS. Platelet‐rich plasma vs. corticosteroid injections for the treatment of recalcitrant lateral epicondylitis: a cost‐effectiveness Markov decision analysis. J Shoulder Elbow Surg. 2022;31:991‐1004. [DOI] [PubMed] [Google Scholar]
- 48. Modic MT, Ross JS. Lumbar degenerative disk disease. Radiology. 2007;245:43‐61. [DOI] [PubMed] [Google Scholar]
- 49. Kjaer P, Korsholm L, Bendix T, Sorensen JS, Leboeuf‐Yde C. Modic changes and their associations with clinical findings. Eur Spine J. 2006;15:1312‐1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Jensen RK, Leboeuf‐Yde C. Is the presence of modic changes associated with the outcomes of different treatments? A systematic critical review. BMC Musculoskelet Disord. 2011;12:183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Lurie JD, Moses RA, Tosteson AN, et al. Magnetic resonance imaging predictors of surgical outcome in patients with lumbar intervertebral disc herniation. Spine. 1976;2013(38):1216‐1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Dudli S, Fields AJ, Samartzis D, Karppinen J, Lotz JC. Pathobiology of Modic changes. Eur Spine J. 2016;25:3723‐3734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Zhou J, Li T, Li L, Xue Y. Clinical efficacy of calcitonin compared to diclofenac sodium in chronic nonspecific low back pain with type I Modic changes: a retrospective study. J Pain Res. 2018;11:1335‐1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Ohtori S, Inoue G, Ito T, et al. Tumor necrosis factor‐immunoreactive cells and PGP 9.5‐immunoreactive nerve fibers in vertebral endplates of patients with discogenic low back pain and Modic type 1 or type 2 changes on MRI. Spine. 2006;31(9):1026‐1031. [DOI] [PubMed] [Google Scholar]
- 55. Fields AJ, Liebenberg EC, Lotz JC. Innervation of pathologies in the lumbar vertebral end plate and intervertebral disc. Spine J. 2014;14:513‐521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Shalash W, Ahrens SR, Bardonova LA, Byvaltsev VA, Giers MB. Patient‐specific apparent diffusion maps used to model nutrient availability in degenerated intervertebral discs. JOR Spine. 2021;4:e1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. McDonnell EE, Wilson N, Barcellona MN, et al. Preclinical to clinical translation for intervertebral disc repair: effects of species‐specific scale, metabolism, and matrix synthesis rates on cell‐based regeneration. JOR Spine. 2023;6:e1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Crockett MT, Kelly BS, van Baarsel S, Kavanagh EC. Modic type 1 vertebral endplate changes: injury, inflammation, or infection? AJR Am J Roentgenol. 2017;209:167‐170. [DOI] [PubMed] [Google Scholar]
- 59. Kerttula L, Luoma K, Vehmas T, Grönblad M, Kääpä E. Modic type I change may predict rapid progressive, deforming disc degeneration: a prospective 1‐year follow‐up study. Eur Spine J. 2012;21:1135‐1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Carragee EJ, Don AS, Hurwitz EL, Cuellar JM, Carrino J, Herzog R. 2009 ISSLS prize winner: does discography cause accelerated progression of degeneration changes in the lumbar disc: a ten‐year matched cohort study. Spine. 2009;34:2338‐2345. [DOI] [PubMed] [Google Scholar]
- 61. Vadalà G, Russo F, Pattappa G, et al. The transpedicular approach as an alternative route for intervertebral disc regeneration. Spine. 1976;2013(38):E319‐E324. [DOI] [PubMed] [Google Scholar]


