Abstract
Purpose:
Young women are typically thought to be protected from cardiovascular disease (CVD) before menopause. However, posttraumatic stress disorder (PTSD) increases CVD risk in women by up to three-fold. Data in predominantly male cohorts point to physiological mechanisms such as vascular and autonomic derangements as contributing to increased CVD risk. The purpose of the current work was to determine if young women diagnosed with PTSD, compared to those without, present with arterial stiffness and impaired autonomic control of the heart.
Methods:
73 healthy young women (18–40 years) with a history of trauma exposure were included in this study, 32 with and 41 without a clinical PTSD diagnosis. We measured resting pulse wave velocity (PWV), central hemodynamics, augmentation pressure and index (AI) via pulse wave analysis using applanation tonometry. Additionally, heart rate variability was assessed via peripheral arterial tone.
Results:
In comparison to controls, women with PTSD showed higher central arterial pressure [systolic, 104±8 vs 97±8 mmHg, p<0.001; diastolic, 72±7 vs 67±7 mmHg, p=0.003], PWV (6±0.3 vs 5±0.6 m/s, p<0.001) and AI (22±13 vs 15±12%, p=0.007) but lower standard deviation of normal-to-normal intervals (SDNN, 44±17 vs 54±18 ms, p=0.005) and root mean square of successive differences between normal heartbeats (RMSSD, 37±17 vs 51±22 ms, p=0.002).
Conclusion:
PTSD in young women is associated with higher brachial and central pressures, increased arterial stiffness, and blunted parasympathetic control of the heart. These findings illustrate potential mechanisms underlying high risk for CVD in young women with PTSD, suggesting potential treatment targets for this at-risk group.
Keywords: PTSD, young, women, arterial stiffness, central arterial pressure
INTRODUCTION
Cardiovascular disease is considered the leading cause of death among women in the United States and the prevalence is increasing with about 1 in every 5 female deaths in 2021 attributed to heart disease [1, 2]. Posttraumatic stress disorder (PTSD) is a debilitating disorder that can develop in individuals after either witnessing or experiencing a traumatic event and is often complicated by the co-occurrence of conditions such as depression, drug and alcohol use, and eating disorders [3, 4].There is increasing evidence of an association between PTSD and the development of cardiovascular disease (CVD)[5]. Women are twice more likely to develop PTSD than men [6] and have higher prevalence of more prolonged PTSD symptoms than men [7]. Furthermore, the development of PTSD particularly before menopause in women may increase cardiovascular disease risk [8]. However, the underlying mechanisms linking PTSD to increased CVD risk remains unknown.
Recent studies demonstrate that women with PTSD have high sympathetic nervous system activity leading to elevated resting heart rate and impaired blood pressure (BP) regulation [9] and greater risk of ischemic heart disease [10]. Furthermore, reduced heart rate variability, heart rate oscillations within the beat-to-beat variability often used as a primary measure of parasympathetic nervous system activity, is linked to increased cardiovascular disease risk [11]. Prior literature points to vascular dysfunction, particularly increased arterial stiffness in PTSD [12], with the magnitude of pulse wave velocity increasing with the severity of PTSD. Arterial stiffness is an important and independent predictor of cardiovascular morbidity and mortality [13], even among asymptomatic individuals [14]. A recent study in 61 young adults (29 women) revealed sex differences in the association of PTSD symptoms and subclinical atherosclerosis, with women displaying reduced vasomotor function as PTSD symptoms increased.
Few studies have explored the underlying vascular mechanisms linking PTSD and cardiovascular risk in premenopausal women. Ebrahimi et al. in 2021 published the largest and most comprehensive study to date evaluating the association between PTSD and ischemic heart disease in women. In this longitudinal cohort study of 398,769 women veterans (132,923 with PTSD matched 1:2 to 265,846 without PTSD), they found that those with PTSD had a 44% higher rate of developing incident ischemic heart disease. More importantly, the elevated ischemic heart disease risk associated with PTSD was most prominent for younger women, particularly women younger than 40 years, suggesting growing cardiovascular disease risk in women before menopause by virtue of PTSD. There is growing evidence that PTSD before menopause increases women’s long-term cardiovascular risk [15, 16] yet the underlying physiological mechanisms are not clear.
Currently, only a limited body of work exists examining PTSD and peripheral vascular function in young women, prior to menopause. To date, no study has examined this relationship in a sample of premenopausal women free of confounding characteristics that can reduce peripheral vascular function independent of PTSD, such as advanced age, high body mass index, comorbidities, and negative lifestyle behaviors such as smoking and sedentary lifestyle. In our very recent work by Tashin et al. [27], we conducted a dimensional analysis that evaluated associations between PTSD symptom severity and two markers of vascular function (i.e. microvascular endothelial function and arterial stiffness) in a group on young trauma-exposed women, regardless of a clinical diagnosis of PTSD. Therefore, the goal of the current study, in a larger sample, was to determine if young women with PTSD present with cardiovascular disease risk factors (i.e., reduced parasympathetic control of the heart, increased arterial stiffness, and higher central hemodynamics) compared to young trauma-exposed women without PTSD. We hypothesized that a PTSD diagnosis would be associated with higher resting heart rate and aortic arterial pressure, lower heart rate variability, and higher pulse wave velocity.
METHODS
The literature on the effects of PTSD on cardiovascular health in young premenopausal women is sparse. Hence the focus of our study sample on premenopausal women. Given the biological sex, gender, and age group of our participants, the results of our study may not be generalizable to other populations. However, because young women are underrepresented in the research and are increasingly at risk for PTSD and its related complications, including CVD, the value of studying this population far outweighs any limitations of generalizability.
Ethical Approval
This study conformed to the standards set by the Declaration of Helsinki and all procedures in this study were approved by the Institutional Review Board of the University of Minnesota. All participants provided written informed consent for study participation via forms approved by the regulatory committee mentioned above.
Study Sample
Seventy-three healthy young women (18–40 years) with a history of trauma exposure recruited over two years were included in this study, 32 with and 41 without a PTSD diagnosis. Participants were recruited from the University of Minnesota Twin Cities campus, the surrounding community, women’s shelters, and through the University of Minnesota Medical Center, Fairview Hospital. Eligibility requirements for all phases of the study included a history of trauma, age (18–40 years of age), premenopausal women, free from any known CVD, and the ability to give informed consent. Exclusion criteria included trans women and men and non-binary individuals; pregnant, breastfeeding, or unwilling to practice birth control during participation in the study; medical conditions such as hypertension, diabetes, heart disease, vascular disease, ongoing illicit drug use, excessive alcohol use (>2 drinks per day), hyperlipidemia, autonomic dysfunction, any serious systemic disease, medications for PTSD, or other CVD; psychiatric comorbidities such as ongoing substance abuse, severe traumatic brain injury, and the inability or unwillingness to abstain from nicotine use for at least 12 hours before physiological studies. The goal of these exclusion criteria was to limit heterogeneity in circulating estrogen levels and comorbid conditions that could affect our exposures and outcome variables. Eligibility criteria were assessed via online screening survey and phone interviews. Women were asked on the screening survey to list all current mental health disorders they have been clinically diagnosed with, and based on their answers, we categorized them as PTSD or controls. The control group in our study reported mental health diagnoses such as depression and anxiety but not PTSD (current or lifetime). Trauma exposure was self-reported and stated in writing by the participants, as part of our standardized measure for PTSD symptoms severity (see Measures below). Given the comorbidity of depression and anxiety with PTSD, we also assessed depression and anxiety symptoms severity in all participants. Although participants were asked to report the trauma that currently affects them the most, more than one traumatic event could be reported.
Experimental Design
We used a cross-sectional study design for the current study. After completing the screening survey online, participants who met the inclusion criteria were invited to the laboratory for the first of the two visits. During their first visit, after obtaining written informed consent, we measured vital signs and collected anthropomorphic data. Next, participants completed mental health questionnaires assessing PTSD symptoms severity and co-morbid anxiety and depression symptoms. Three seated blood pressures (BP) and heart rate were measured during this visit with an appropriately sized cuff placed on the upper arm with the arm resting at heart level after at least 5 min of quiet rest using an automated digital BP device (Omron, HEM-907XL, Omron Healthcare, Kyoto, Japan). Resting respiratory rate was measured in between BP measures by visually counting the number of breaths taken over the course of 1 min in the supine position. During the second laboratory visit, we collected our primary variables of interest listed and described below. All participants were instructed to abstain from smoking, exercise, alcohol, caffeine, and over-the-counter medication affecting BP for 12 h before the visit and a minimum of 6 h for food. This visit was scheduled within 1–5 days after the onset of actual or planned menstruation, to ensure that vascular measurements were recorded during the early follicular phase of the menstrual cycle. Women who had irregular cycles were asked to contact the laboratory at onset of menstruation for their second visit. For each participant, a urine pregnancy test was performed to exclude pregnancy.
Outcome Measures
Aortic arterial stiffness and central hemodynamics.
We quantified arterial stiffness via applanation tonometry (SphygmoCor XCEL, Atcor Medical, Sydney, Australia) with the participant lying in the supine position. Pulse-wave velocity (PWV) measurements were performed noninvasively using a tonometer placed over the carotid artery and a BP cuff over the femoral artery. The distance from the carotid to femoral artery measurement sites was measured, and PWV was calculated as the quotient of the distance and the time delay between the carotid and femoral pressure waveforms (m/s). The SphygmoCor method uses the foot of the waveform as an onset point for calculating the time differences between the R wave and the pulse waveforms at each site [17]. Using the same device, aortic arterial pressure, augmentation pressure and index (AI) were determined by the technique of noninvasive aortic pulse wave analysis as described here [18]. AI is based on blood pulse-wave reflection and calculated as the pressure difference between the second peak of the aortic reflection wave and the first peak of the incident wave. It is expressed as a percentage of central pulse pressure. The heart rate-corrected AI is called AI@75. We collected two to three measures of PWV and AI per participant and calculated the average value.
Heart rate variability (HRV).
Time domains of heart variability [standard deviation of normal-to-normal intervals (SDNN) and the root mean square of difference between normal heart beats (RMSSD) to measure heart rate variability] were measured by recording finger arterial pulsatile volume changes from peripheral arterial tonometry using the EndoPAT2000 device (Itamar Medical Ltd., Caesarea, Israel). Inflatable pressure cuffs were attached to the index fingers of both hands while the participants rested in the supine position in a quiet laboratory. A BP cuff was placed on the participant’s non-dominant forearm. After a 6-min resting baseline, the BP cuff was inflated to a suprasystolic pressure (>220 mmHg) for a period of 5 min, followed by rapid cuff deflation and data collection (recovery) for another 5 min. HRV is calculated from the baseline period, based on the European Society of Cardiology and North American Society of Pacing Electrophysiology task force standards [19].
Exposure Measures
PTSD checklist for DSM-5 with criterion A (traumatic event).
The PTSD checklist for DSM-5 (PCL5) is a 20-item self-reported questionnaire based on the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) symptoms of PTSD [20]. As part of this standardized measure, participants were asked in criterion A to “briefly identify their worst traumatic event (if they felt comfortable doing so).” Next, participants reported how much they were bothered over the past month by symptoms related to that traumatic event, using a five-point Likert scale (0 = “Not at all,” 1 = “A little bit,” 2 = “Moderately,” 3 = “Quite a bit,” 4 = “Extremely”). This questionnaire is one of the most widely used screening measures for PTSD and findings support PCL5 as a psychometrically sound measure in individuals at high risk for exposure to trauma [21]. The total score for PCL5 ranges from 0 to 80. A PCL5 cutoff score between 31 and 33 is indicative of probable PTSD [22].
Comorbid Exposure Measures
Spielberger state-trait anxiety inventory for adults.
The state-trait anxiety inventory (STAI; “Form Y”) assesses self-reported anxiety (both state and trait anxiety) using a validated 40-item four-point Likert scale questionnaire [23]. State anxiety reflects transient (i.e., current moment) emotional anxiety due to situational stress. Trait anxiety assesses an individual’s predisposition to react with anxiety in any stressful event. Together, the STAI allows the quantification of personal characteristic anxiety reactivity, as well as transient fluctuations dependent on the situation. Both subscales have been shown to have good internal reliability and test-retest reliability [24]. STAI scores vary from a minimum score of 20 to a maximum score of 80 on both the STAI trait (STAI-T) and STAI state (STAI-S) subscales. STAI scores are commonly classified no or low anxiety for scores between 20 to 37 [23, 25].
Beck depression inventory-II.
The Beck depression inventory-II (BDI-II) is a well-validated 21-item self-report measure, scored from 0 to 3, of current depressive symptoms [26]. In the first portion of the test, psychological symptoms are assessed, whereas the second portion assesses physical symptoms. In this study, items were summed to create an overall, continuous depression severity score and to compute a categorical diagnostic of current depression where a cutoff score greater than 18 suggests probable depression [26]. Total raw score can range from 0 to 63, with cutoff scores of 0 to 9 for none to minimal depression.
Statistical Analysis
Data were analyzed statistically using commercial software (SPSS 25.0; IBM SPSS, Armonk, NY) and the Statistical Analysis System (SAS). All data were tested for normal distribution. We analyzed demographic characteristics using descriptive statistics. Independent t-tests were used to compare mental health questionnaires data and our outcome variables (BP, HR, PWV, augmentation pressure, AI, AI@75, SDNN and RMSSD) between women with PTSD and trauma-exposed controls without PTSD. Values are reported as mean ± SD. Significance level was set at α < 0.05. A subset of the current sample of participants was included in our previously published paper [27] as well as in a conference abstract, APS Summit 2023 [28].
RESULTS
For all graphed variables, to avoid repetition of values presented in figures, we are reporting instead the t-statistic as follow: t(degrees of freedom)= t-statistic, p-value.
Demographic characteristics and mental health questionnaires:
32 women with PTSD (PTSD+) and 41 trauma-exposed women without PTSD (PTSD-) were enrolled in the study. As seen in Table 1, PTSD+ women were aged 28 ± 7 vs 24 ± 6 yr for PTSD-, with a BMI of 28 ± 6 vs 24 ± 4 kg/m2 and an abdominal circumference of 87 ± 23 vs 75 ± 15 cm for PTSD+ and PTSD-, respectively. Resting respiratory rates were 15 ± 3 vs 16 ± 3 breaths/min for PTSD+ and PTSD-, respectively.
Table 1.
Descriptive characteristics of all 73 participants by group.
| PTSD+ (n=32) | PTSD− (n=41) | P-Value | |||
|---|---|---|---|---|---|
| Mean±SD | Min-Max | Mean±SD | Min-Max | ||
| Age (years) | 28 ± 7 | 19 – 40 | 24 ± 6 | 18 – 38 | 0.004 |
| BMI (Kg/m2) | 28 ± 6 | 13 – 40 | 24 ± 4 | 17 – 35 | <0.001 |
| Abdominal circumference (cm) | 87 ± 23 | 41 – 126 | 75 ± 15 | 27 – 103 | 0.013 |
| Respiratory rate (breaths/min) | 15 ± 3 | 7 – 24 | 16 ± 3 | 7 – 20 | 0.243 |
PTSD+, Women diagnosed with posttraumatic stress disorder (PTSD) diagnosis; PTSD-, trauma-exposed women without PTSD diagnosis; n, number of participants included; SD, Standard Deviation; BMI, Body Mass Index.
As shown in Table 2, participants in the PTSD+ group had higher scores of PCL5 (41 ± 15 vs 29 ± 15 a.u, p=0.001), and BDI (21 ± 13 vs 14 ± 9 a.u, p=0.017) compared to PTSD− (Table 2). STAI-Trait was also higher in the PTSD+ group (48 ± 14 vs 42 ± 13 a.u, p=0.038). STAI-State (49 ± 14 vs 46 ± 13 a.u, p=0.370) scores were not different between PTSD+ and PTSD-, respectively.
Table 2:
Comparison of mental health questionnaires severity scores between the groups.
| PTSD+ (n=32) | PTSD− (n=41) | P-value | |||
|---|---|---|---|---|---|
| Mean±SD | Min-Max | Mean±SD | Min-Max | ||
| PTSD checklist for DSM-5 criterion A (PCL5) | 41 ± 15 | 10 – 69 | 29 ± 15 | 5 – 62 | 0.001 |
| State-Trait Anxiety Inventory Trait (STAI-T) | 48 ± 14 | 26 – 73 | 42 ± 13 | 6 – 63 | 0.038 |
| State-Trait Anxiety Inventory State (STAI-S) | 49± 14 | 27 – 69 | 46 ± 13 | 24 – 71 | 0.370 |
| Beck’s Depression Inventory-II (BDI-II) | 21 ± 13 | 0 – 50 | 14 ± 9 | 0 – 33 | 0.017 |
PTSD+, Women diagnosed with posttraumatic stress disorder (PTSD) diagnosis; PTSD-, trauma-exposed women without PTSD diagnosis; n, number of participants included; SD, Standard Deviation; Min, Minimum; Max, Maximum.
Brachial Arterial Pressure and Heart Rate:
Figure 1 shows resting peripheral systolic arterial BP [panel A, t(71)= 2.93, p=0.002], diastolic arterial BP [panel B, t(71)= 2.61, p=0.006] and heart rate [panel C, t(71)= 2.27, p=0.013] were higher in PTSD+ compared to PTSD-.
Figure 1.
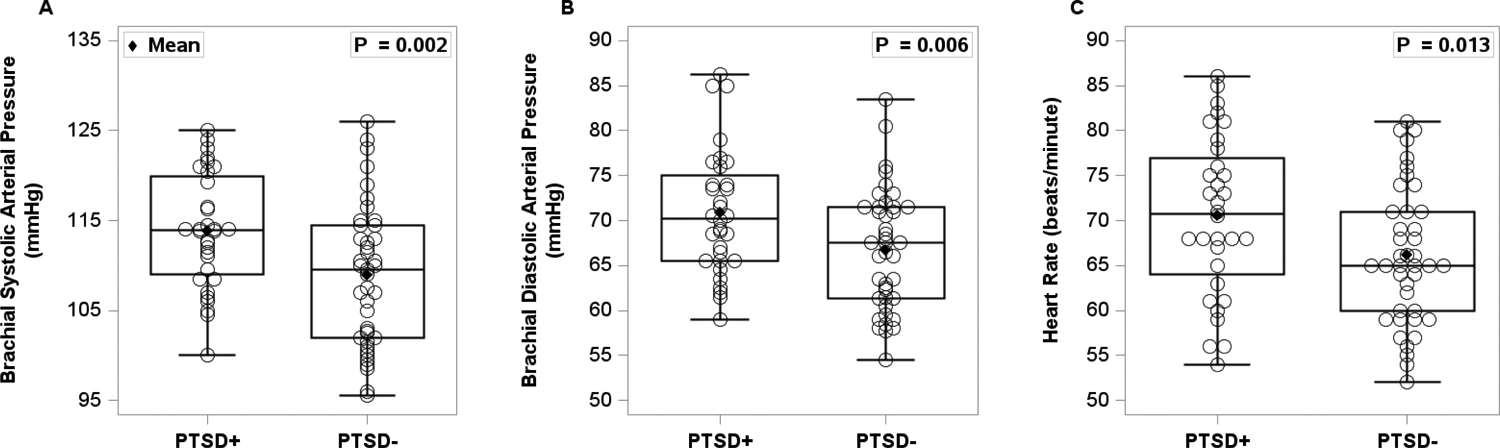
Brachial systolic and diastolic blood pressure (panel A and B) in 32 women with PTSD (PTSD+) and 41 trauma-exposed women without PTSD (PTSD-). Baseline heart rate (panel C) was also measured in 32 PTSD+ and 41 PTSD-. An independent t-test was used to compare the two groups. Women with PTSD had significantly elevated brachial arterial pressure and heart rate compared to women without PTSD.
Central (Aortic) Arterial Pressure and Augmentation Pressure:
Likewise, central systolic arterial BP [panel A, t(71)= 3.56, p<0.001] and diastolic arterial BP [panel B, t(71)= 2.84, p=0.003] were higher in PTSD+ compared to PTSD− (Figure 2). Finally, aortic augmentation pressure [panel C, t(71)= 3.10, p=0.001] was also higher in PTSD+ compared to PTSD-.
Figure 2.
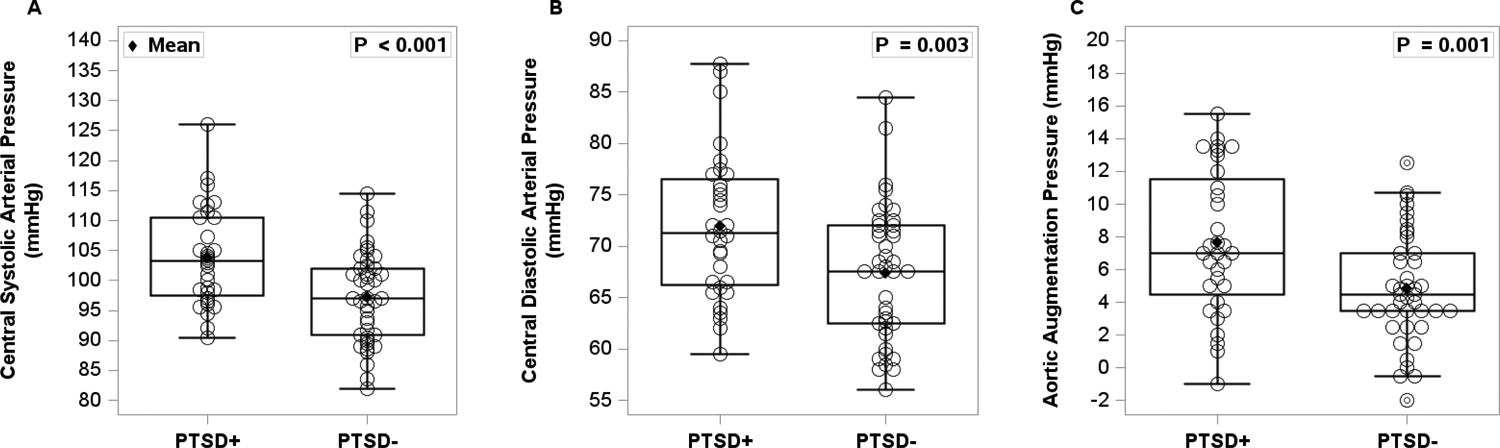
Central (aortic) systolic and diastolic arterial pressure (panel A and B) in 32 women with PTSD (PTSD+) and 41 trauma-exposed women without PTSD (PTSD-). Aortic augmentation pressure (panel C) was also measured in 32 PTSD+ women and 41 PTSD− women. An independent t-test was used to compare the two groups. Women with PTSD had significantly elevated aortic systolic and diastolic arterial pressures, as well aortic augmentation pressure compared to women without PTSD.
Pulse Wave Velocity and Augmentation Index:
As seen in Figure 3, PWV was higher in PTSD+ [panel A, t(71)= 5.79, p<0.001] compared to PTSD-. Similarly, AI was higher in PTSD+ [panel B, t(71)= 2.51, p=0.007] compared to PTSD-. Lastly, after adjusting for heart rate, AI@75 [panel C, t(71)= 3.44, p<0.001] remained higher in PTSD+ compared to PTSD-.
Figure 3.
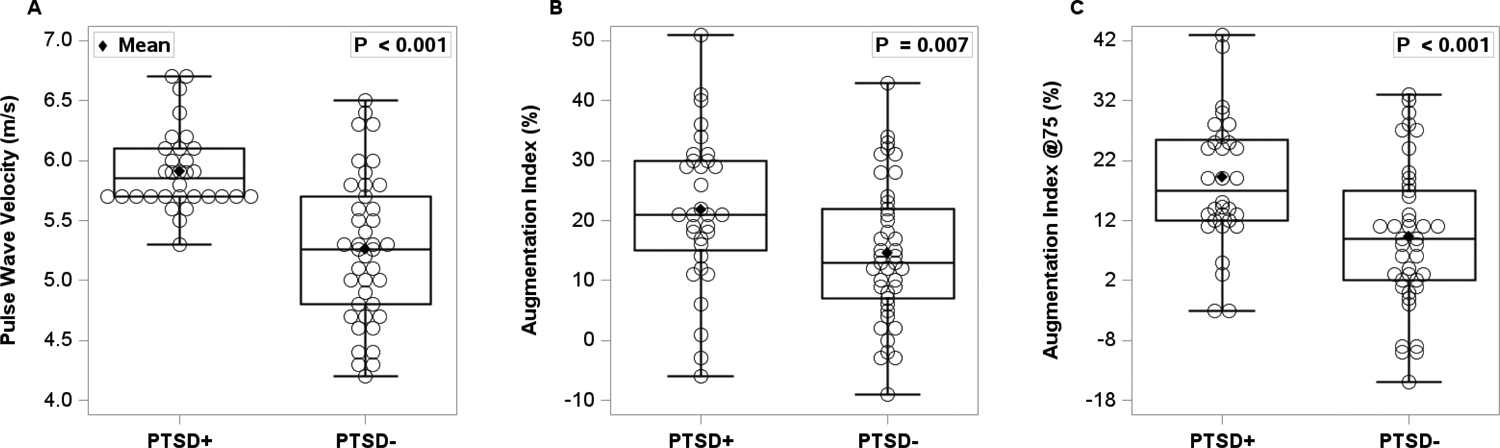
Pulse wave velocity (panel A), augmentation index (panel B), as well as augmentation index normalized to 75 bpm (panel C) was obtained from 32 women with PTSD (PTSD+) and 41 trauma-exposed women without PTSD (PTSD-). An independent t-test was used to compare the two groups. Women with PTSD had significantly elevated pulse wave velocity and augmentation index compared to women without PTSD.
Baseline Heart Rate Variability:
Time domains of heart rate variability (SDNN and RMSSD) were blunted in PTSD+ compared to PTSD− (Figure 4). Specifically, Baseline SDNN measurements were ~20% lower in PTSD+ [panel A, t(71)= −2.62, p=0.005] compared to PTSD-. Similarly, baseline RMSSD [panel B, t(71)= −3.03, p=0.002] was ~30% lower in PTSD+ compared to PTSD-.
Figure 4.
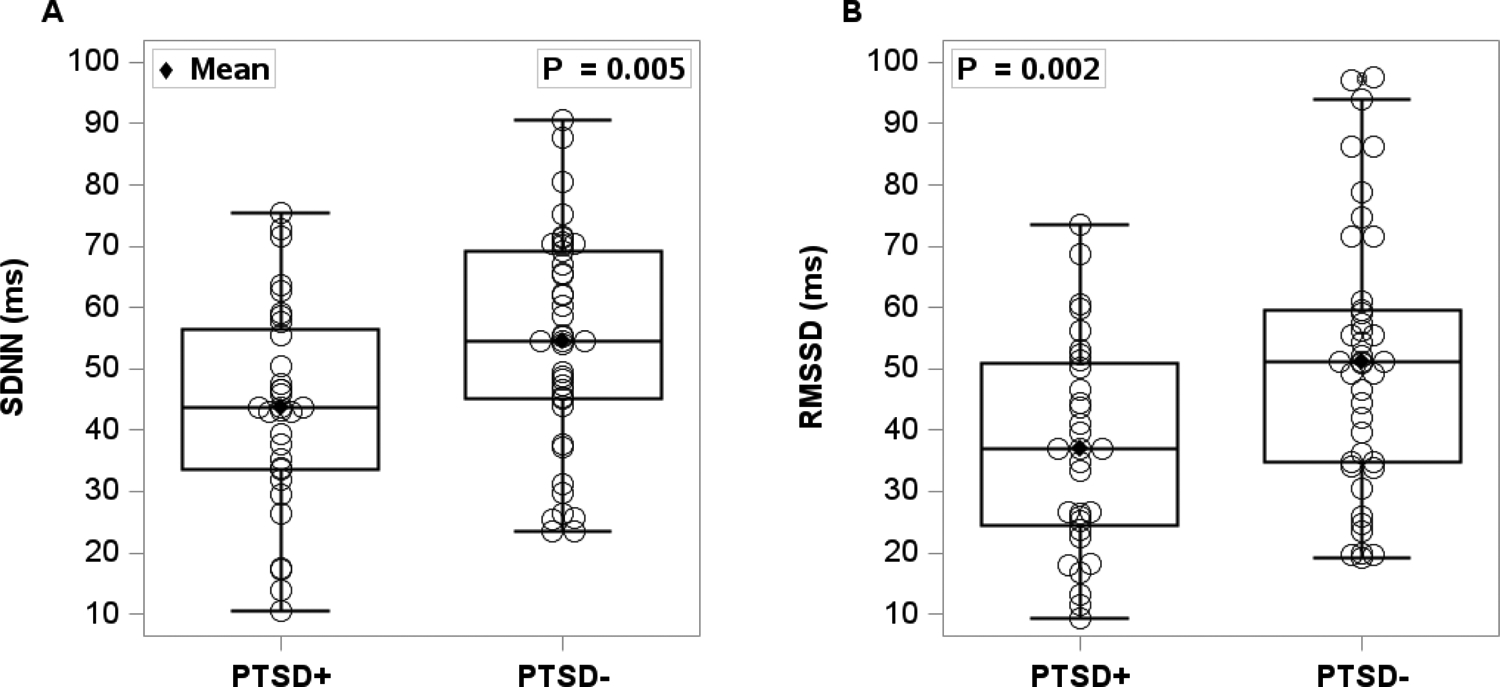
Resting time domains of heart rate variability [standard deviation of normal R-R intervals (SDNN) (panel A) and root mean square of the successive differences (RMSSD) (panel B)] was measured in 32 women with PTSD (PTSD+) and 41 trauma-exposed women without PTSD (PTSD-). An independent t-test was used to compare the two groups. Women with PTSD had significantly lower SDNN and RMSSD compared to women without PTSD.
When controlling for age and BMI, PWV (p<0.001), AI@75 (p=0.008), HR (p<0.001) remained higher in young women with a clinical diagnosis of PTSD compared to those without. Likewise, SDNN (p=0.025) and RMSSD (p=0.018) remained blunted in young women with a clinical diagnosis of PTSD compared to those without. Central arterial systolic pressure (p=0.067) but not diastolic (p=0.097) tended to be statistically higher in young women with a clinical diagnosis of PTSD after controlling for age and BMI.
Correlations between physiological outcomes and mental health outcomes.
STAI was correlated with PWV (r=0.30, p=0.035) and not SDNN, RMSSD, SBP or DBP. PCL5 and BDI-II scores did not show significant correlations with either PWV, SDNN, RMSSD, SBP or DBP (all P>0.1).
DISCUSSION
In the current study, we examined arterial stiffness, peripheral and central hemodynamics and vagal control of the heart due to PTSD. We report for the first time that among trauma-exposed women, those diagnosed with PTSD present with greater arterial stiffness, higher peripheral and central hemodynamics, and blunted vagal control of the heart compared to those without PTSD. Specifically, we report that: 1) brachial arterial pressure and heart rate were higher in women with PTSD; 2) aortic arterial pressure and augmentation pressure were also higher in women with PTSD; 3) pulse wave velocity, augmentation index and augmentation index @75 were all higher in women with PTSD; and finally 4) time domains measures of heart rate variability (SDNN and RMSSD) were lower in women with PTSD compared to trauma-exposed women without PTSD. Our current findings may help establish a mechanistic link between PTSD and cardiovascular health in trauma-exposed women.
PTSD is a psychiatric disorder with a prevalence higher in women Veterans (11.4%) than in male Veterans (5.2%) [29]. PTSD include four clusters of symptoms: re-experiencing of trauma, avoidance, negative thoughts and emotions, and hyperarousal [30]. In addition to a greater prevalence in women, evidence is accumulating for association between PTSD and CVD. In a representative sample of almost 200,000 Veterans of the wars in Iraq and Afghanistan, Burg et al (2017) observed a 24%–46% greater risk for incident hypertension associated with untreated PTSD. Currently, the pathophysiological mechanisms by which PTSD is independently related to increased hypertension and CVD risk are still under investigation, but there are many candidate mechanisms. Thus far, the literature strongly supports the roles of autonomic dysfunction in the association between PTSD and CVD [9, 31–33]. Park et al. [33] provided the first evidence of overactivity of sympathetic nervous system in veterans with PTSD using the gold standard technique of microneurography. There is growing evidence that women with PTSD have aberrant (increased or decreased) sympathetic neural drive compared to female controls [9, 32, 34, 35]. Some demonstrates greater sympathetic nervous activity (SNA) reactivity in women with PTSD [9, 32], while others demonstrate that there is blunted SNA reactivity [35, 36]. These studies highlight that there is clear SNA-related dysfunction in women with PTSD; however, it may not just be sympathetic overactivity.
Similarly, the evidence for vascular dysfunction as a potential underlying mechanism is also growing [37–39], with some studies suggesting a bidirectional causal relationship between endothelial dysfunction and PTSD. Our previous study in young trauma-exposed women revealed that PTSD symptoms severity predicts endothelial dysfunction [27]. In the current study, we report that arterial stiffness, the other main contributor to vascular dysfunction, might already be present in these young women by virtue of having PTSD. Specifically, we measured higher augmentation index and [40] PWV in young women with PTSD than in controls. The potential mechanisms behind vascular dysfunction in PTSD have yet to be thoroughly elucidated, particularly in these young women with PTSD. However, increased inflammation [41–43] and oxidative stress [40, 44] have been suggested to induce reductions in peripheral vascular function in people with PTSD.
Further, we recently showed that in trauma-exposed women, vagal control of the heart moderates the association between vascular dysfunction and PTSD symptoms [45]. In line with that finding, here we report that young women with PTSD in the present study also have blunted heart rate variability at rest, suggestive of blunted vagal control of the heart and impaired ability of the parasympathetic nervous system to effectively regulate sympathetic stress response states. Prior data in predominantly men and older women [46, 47] provide strong evidence for autonomic imbalance in PTSD, with reports of increased sympathetic activity and blunted parasympathetic activity at rest and in response to acute stress [9, 33, 48]. The chronic increase in sympathetic arousal observed in PTSD might exert an allostatic load on the vascular wall, leading to smooth muscle remodeling and stiffening of the arterial wall over time. When arousal becomes chronic, the body adapts by thickening arteriolar smooth muscle and increasing the vascular wall-to-lumen ratio for more effective higher blood pressure maintenance, resulting in arterial stiffness [49, 50]. This smooth muscle remodeling might explain why young women diagnosed with PTSD present with arterial stiffness in addition to lower vagal control. Trauma exposure has been reported to lead to a cascade of biological changes and stress responses. Casada and Roache [51] examined autonomic (skin conductance and heart rate) changes accompanying behavioral inhibition and activation using a stop signal task in 10 PTSD and 13 trauma-exposed controls, and reported a dissociation between autonomic reactivity and behavioral response in PTSD but not in trauma-exposed controls. This suggest that although trauma-exposure presents with physiological consequences, this response is unlike the response observed in PTSD. Taken together, prior and current results provide insight into the physiological changes preceding the overt development of hypertension in this population.
Finally, in the current study we found that along with arterial stiffness and impaired vagal control, young women with PTSD had higher brachial and aortic arterial pressures compared to controls. These findings are similar to those of Moazen-Zadeh et al. in a sample of 50 male veterans aged 41 to 73 years compared to age-matched controls [52]. As expected, their older male population had a slightly higher mean central systolic arterial pressure than young women in our study (117 vs 104 mmHg). As arterial stiffness increases, elevations in central systolic blood pressure can occur due to increased forward and reflected wave amplitudes and earlier return of the reflected wave to the proximal aorta [53]. Likewise, although resting HR was within the normal of 60 to 100, it was also higher in young women diagnosed with PTSD, and this increase might be driven by reduced parasympathetic control as well as heightened sympathetic stimulation. Our recently published work showed an increase in HR with increased PTSD symptom severity in a primarily male cohort [43]. In the present study, PTSD symptoms severity in women with PTSD was almost double that of trauma-exposed women without PTSD and could explain the difference in HR. This is a clinically relevant finding since higher resting HR has previously been shown to be a marker of future CVD risk [54–56]. Importantly, our results suggest a greater impairment in autonomic control of heart rate and blood pressure may be linked to greater CVD risk in young women with PTSD. Of note, we uncovered few statistically significant correlations between our mental health scores and our physiological outcome variables. Consistent with our recently published paper [27], only PWV was correlated with STAI However, the lack of significant correlations, except for PWV and STAI could be because these symptoms severity score (PCL5, BDI and STAI) focus on the experience of symptoms for last 3 weeks, while physiological changes take months and years to develop.
We recognize several limitations to our study. First, our cohort was primarily comprised of Caucasian cis-women from the Twin-Cities area; therefore, our results may not be generalizable to other geographical locations, racial groups, or trans- populations. However, young trauma-exposed women remain an understudied population at increasing risk for hypertension and CVD. Our findings may help advance the care of young women living with PTSD, who are overlooked in CVD prevention. Second, we did not diagnose our participants but instead relied on their self-reported current clinician diagnosed mental health conditions. Rather, we assessed PTSD symptoms severity using a well-validated questionnaire. Third, our study is cross-sectional in nature and can only explore associations and not causation. Fourth, we did not have a control group of women without trauma exposure in the current study so we are not able to assess how our findings compare to healthy controls. However, given prior reports that trauma can lead to various physical disorders including cardiovascular and neurological, our current data informs on the additive physiological burden of a PTSD diagnosis. Further, in our current study we merely collected PCL5 data to measure symptoms severity and did not use the score to categorize our participants for data analysis. It is then possible that many in our PTSD− group are individuals that are not yet clinically diagnosed with PTSD. Given that the gold standard for PTSD diagnosis is the Clinician-Administered PTSD Scale, we are careful to categorize our control group as having moderate or severe PTSD based on PCL5. Finally, we did not account for differences in physical activity that could influence autonomic control and arterial stiffness [57].
In summary, a growing number of studies [47] in the recent years have highlighted the role of autonomic nervous system and baroreflex sensitivity as an early mechanism contributing to the development of CVD in PTSD. Our findings demonstrate that in addition to autonomic dysfunction (i.e. blunted parasympathetic control of the heart), young women diagnosed with PTSD display higher arterial stiffness coupled with higher aortic arterial pressure. PTSD may impose a high allostatic load on the heart of premenopausal women typically thought to be protected from CVD. Available literature support that increased arterial stiffness in young adults is associated with left ventricular mass index independently of traditional CV risk factors. Therefore, these results may provide a mechanistic link into cardiovascular risk in a younger premenopausal population of women often thought to be protected from CVD. Further, consistent with prior reports suggesting that the increase in CVD risk is proportionate to increased PTSD severity [58], we observed in the current study that among a group of young trauma-exposed women, those with a diagnosis of PTSD had higher PTSD symptoms severity and impaired autonomic and cardiovascular biomarkers. Further research is needed to optimize the care and mitigate the increased CVD risk in trauma-exposed women before menopause. Importantly research is needed to evaluate the potential for intervention strategies targeting autonomic and vascular function in premenopausal women with PTSD to prevent future development of hypertension and ultimately to reduce CVD morbidity and mortality in this overlooked population.
Acknowledgements:
We are grateful for our amazing participants and the dedicated Neurobiology of Emotion, Sleep and Trauma (NEST) lab team.
This study was supported by the following grants: National Institutes of Health (NIH) K01HL161027, NIH Programs to Increase Diversity among Individuals Engaged in Health-Related Research (PRIDE) R25 HL105400 and NIH UL1TR002494.
Footnotes
Statement: On behalf of all authors, the corresponding author states that there is no conflict of interest.
REFERENCES
- 1.Garcia M, Mulvagh SL, Merz CNB, et al. (2016) Cardiovascular Disease in Women: Clinical Perspectives. Circ Res 118:1273–1293. 10.1161/CIRCRESAHA.116.307547 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tsao CW, Aday AW, Almarzooq ZI, et al. (2023) Heart disease and stroke statistics—2023 update: A report from the American Heart Association. Circulation 147:. 10.1161/cir.0000000000001123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.María-Ríos CE, Morrow JD (2020) Mechanisms of shared vulnerability to post-traumatic stress disorder and substance use disorders. Front Behav Neurosci 14:6. 10.3389/fnbeh.2020.00006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reyes-Rodríguez ML, Von Holle A, Ulman TF, et al. (2011) Posttraumatic stress disorder in anorexia nervosa. Psychosom Med 73:491–497. 10.1097/PSY.0b013e31822232bb [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Levine GN, Cohen BE, Commodore-Mensah Y, et al. (2021) Psychological health, well-being, and the mind-heart-body connection: A scientific statement from the American Heart Association. Circulation 143:. 10.1161/cir.0000000000000947 [DOI] [PubMed] [Google Scholar]
- 6.Resnick EM, Mallampalli M, Carter CL (2012) Current challenges in female veterans’ health. J Womens Health (Larchmt) 21:895–900. 10.1089/jwh.2012.3644 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pooley AE, Benjamin RC, Sreedhar S, et al. (2018) Sex differences in the traumatic stress response: PTSD symptoms in women recapitulated in female rats. Biol Sex Differ 9:. 10.1186/s13293-018-0191-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Remch M, Laskaris Z, Flory J, et al. (2018) Post-traumatic stress disorder and cardiovascular diseases. Circ Cardiovasc Qual Outcomes 11:. 10.1161/circoutcomes.117.004572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yoo J-K, Badrov MB, Huang M, et al. (2020) Abnormal sympathetic neural recruitment patterns and hemodynamic responses to cold pressor test in women with posttraumatic stress disorder. Am J Physiol Heart Circ Physiol 318:H1198–H1207. 10.1152/ajpheart.00684.2019 [DOI] [PubMed] [Google Scholar]
- 10.Ebrahimi R, Lynch KE, Beckham JC, et al. (2021) Association of Posttraumatic Stress Disorder and Incident Ischemic Heart Disease in Women Veterans. JAMA Cardiol 6:642–651. 10.1001/jamacardio.2021.0227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Binici Z, Mouridsen MR, Køber L, Sajadieh A (2011) Decreased nighttime heart rate variability is associated with increased stroke risk. Stroke 42:3196–3201. 10.1161/strokeaha.110.607697 [DOI] [PubMed] [Google Scholar]
- 12.Walczewska J, Rutkowski K, Wizner B, et al. (2011) Stiffness of large arteries and cardiovascular risk in patients with post-traumatic stress disorder. Eur Heart J 32:730–736. 10.1093/eurheartj/ehq354 [DOI] [PubMed] [Google Scholar]
- 13.Cecelja M, Chowienczyk P (2012) Role of arterial stiffness in cardiovascular disease. JRSM Cardiovasc Dis 1(4):cvd.2012.012016. 10.1258/cvd.2012.012016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Redheuil A, Wu CO, Kachenoura N, et al. (2014) Proximal aortic distensibility is an independent predictor of all-cause mortality and incident CV events: the MESA study. J Am Coll Cardiol 64:2619–2629. 10.1016/j.jacc.2014.09.060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vaccarino V, Bremner JD (2017) Behavioral, emotional and neurobiological determinants of coronary heart disease risk in women. Neurosci Biobehav Rev 74:297–309. 10.1016/j.neubiorev.2016.04.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Remch M, Laskaris Z, Flory J, et al. (2018) Post-traumatic stress disorder and cardiovascular diseases: A cohort study of men and women involved in cleaning the debris of the world Trade Center complex. Circ Cardiovasc Qual Outcomes 11:e004572. 10.1161/CIRCOUTCOMES.117.004572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Doupis J, Papanas N, Cohen A, et al. (2016) Pulse Wave Analysis by Applanation Tonometry for the Measurement of Arterial Stiffness. Open Cardiovasc Med J 10:188–195. 10.2174/1874192401610010188 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.O’Rourke MF, Pauca A, Jiang XJ (2001) Pulse wave analysis. Br J Clin Pharmacol 51:507–522. 10.1046/j.0306-5251.2001.01400.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Camm AJ, Malik M, Bigger JT, et al. (1996) Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North …. Circulation [PubMed] [Google Scholar]
- 20.Weathers F, Litz B, Keane T, et al. (2013) The PTSD Checklist for DSM-5 (PCL-5) - Extended Criterion A [Measurement Instrument
- 21.Morrison K, Su S, Keck M, Beidel DC (2021) Psychometric properties of the PCL-5 in a sample of first responders. J Anxiety Disord 77:102339. 10.1016/j.janxdis.2020.102339 [DOI] [PubMed] [Google Scholar]
- 22.Blanchard EB, Jones-Alexander J, Buckley TC, Forneris CA (1996) Psychometric properties of the PTSD Checklist (PCL). Behav Res Ther 34:669–673. 10.1016/0005-7967(96)00033-2 [DOI] [PubMed] [Google Scholar]
- 23.Spielberger C, Gorsuch R, Lushene R, et al. (1983) State-Trait Anxiety Inventory for Adults (Form Y): Manual, Test, Scoring Key. Mind Garden Inc, Redwood City, CA [Google Scholar]
- 24.Ortuño-Sierra J, García-Velasco L, Inchausti F, et al. (2016) New approaches on the study of the psychometric properties of the STAI. Actas Esp Psiquiatr 44:83–92 [PubMed] [Google Scholar]
- 25.Kayikcioglu O, Bilgin S, Seymenoglu G, Deveci A (2017) State and Trait Anxiety Scores of Patients Receiving Intravitreal Injections. Biomed Hub 2:1–5. 10.1159/000478993 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beck AT, Steer RA, Brown GK (1996) Manual for the Beck Depression Inventory-II. Psychological Corporation, San Antonio, TX [Google Scholar]
- 27.Tahsin CT, Michopoulos V, Powers A, et al. (2023) Sleep efficiency and PTSD symptom severity predict microvascular endothelial function and arterial stiffness in young, trauma-exposed women. American Journal of Physiology-Heart and Circulatory Physiology 325:H739–H750. 10.1152/ajpheart.00169.2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ahmed Z, Michopoulos V, Medina Banuelo S, et al. (2023) Premenopausal women with PTSD display blunted vagal control of the heart and elevated central hemodynamics. Physiology (Bethesda) 38:. 10.1152/physiol.2023.38.s1.5728859 [DOI] [Google Scholar]
- 29.Mano T, Iwase S, Toma S (2006) Microneurography as a tool in clinical neurophysiology to investigate peripheral neural traffic in humans. Clin Neurophysiol 117:2357–2384. 10.1016/j.clinph.2006.06.002 [DOI] [PubMed] [Google Scholar]
- 30.Rudas L, Crossman AA, Morillo CA, et al. (1999) Human sympathetic and vagal baroreflex responses to sequential nitroprusside and phenylephrine. Am J Physiol Heart Circ Physiol 276:H1691–H1698. 10.1152/ajpheart.1999.276.5.h1691 [DOI] [PubMed] [Google Scholar]
- 31.Fu Q (2022) Autonomic dysfunction and cardiovascular risk in post-traumatic stress disorder. Auton Neurosci 237:102923. 10.1016/j.autneu.2021.102923 [DOI] [PubMed] [Google Scholar]
- 32.Yoo J-K, Badrov MB, Parker RS, et al. (2020) Early onset neurocirculatory response to static handgrip is associated with greater blood pressure variability in women with posttraumatic stress disorder. Am J Physiol Heart Circ Physiol 318:H49–H58. 10.1152/ajpheart.00490.2019 [DOI] [PubMed] [Google Scholar]
- 33.Park J, Marvar PJ, Liao P, et al. (2017) Baroreflex dysfunction and augmented sympathetic nerve responses during mental stress in veterans with post-traumatic stress disorder. J Physiol 595:4893–4908. 10.1113/JP274269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.D’Souza AW, Yoo J-K, Takeda R, et al. (2022) Neurocardiovascular dysregulation during orthostasis in women with posttraumatic stress disorder. Circulation 146:1483–1485. 10.1161/CIRCULATIONAHA.122.061705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.D’Souza AW, Yoo J-K, Takeda R, et al. (2022) Impaired sympathetic neural recruitment during exercise pressor reflex activation in women with post-traumatic stress disorder. Clin Auton Res 32:115–129. 10.1007/s10286-022-00858-1 [DOI] [PubMed] [Google Scholar]
- 36.D’Souza AW, Yoo J-K, Takeda R, et al. (2022) Neurocardiovascular dysregulation during orthostasis in women with posttraumatic stress disorder. Circulation 146:1483–1485. 10.1161/circulationaha.122.061705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sfera A, Osorio C, Rahman L, et al. (2021) PTSD as an endothelial disease: Insights from COVID-19. Front Cell Neurosci 15:770387. 10.3389/fncel.2021.770387 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Grenon SM, Owens CD, Alley H, et al. (2016) Posttraumatic stress disorder is associated with worse endothelial function among veterans. J Am Heart Assoc 5:e003010. 10.1161/JAHA.115.003010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Edmondson D, von Känel R (2017) Post-traumatic stress disorder and cardiovascular disease. Lancet Psychiatry 4:320–329. 10.1016/S2215-0366(16)30377-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Miller MW, Lin AP, Wolf EJ, Miller DR (2018) Oxidative stress, inflammation, and neuroprogression in chronic PTSD. Harv Rev Psychiatry 26:57–69. 10.1097/HRP.0000000000000167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hori H, Kim Y (2019) Inflammation and post-traumatic stress disorder. Psychiatry Clin Neurosci 73:143–153. 10.1111/pcn.12820 [DOI] [PubMed] [Google Scholar]
- 42.Moshfegh CM, Elkhatib SK, Collins CW, et al. (2019) Autonomic and redox imbalance correlates with T-lymphocyte inflammation in a model of chronic social defeat stress. Front Behav Neurosci 13:103. 10.3389/fnbeh.2019.00103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fonkoue IT, Marvar PJ, Norrholm S, et al. (2020) Symptom severity impacts sympathetic dysregulation and inflammation in post-traumatic stress disorder (PTSD). Brain Behav Immun 83:260–269. 10.1016/j.bbi.2019.10.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Miller MW, Sadeh N (2014) Traumatic stress, oxidative stress and post-traumatic stress disorder: neurodegeneration and the accelerated-aging hypothesis. Mol Psychiatry 19:1156–1162. 10.1038/mp.2014.111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Seligowski AV, Fonkoue IT, Noble NC, et al. (2022) Vagal control moderates the association between endothelial function and PTSD symptoms in women with T2DM. Brain Behav Immun Health 26:100527. 10.1016/j.bbih.2022.100527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fonkoue IT, Michopoulos V, Park J (2020) Sex differences in post-traumatic stress disorder risk: autonomic control and inflammation. Clin Auton Res 30:409–421. 10.1007/s10286-020-00729-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brudey C, Park J, Wiaderkiewicz J, et al. (2015) Autonomic and inflammatory consequences of posttraumatic stress disorder and the link to cardiovascular disease. Am J Physiol Regul Integr Comp Physiol 309:R315–21. 10.1152/ajpregu.00343.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fonkoue IT, Norrholm SD, Marvar PJ, et al. (2018) Elevated resting blood pressure augments autonomic imbalance in posttraumatic stress disorder. Am J Physiol Regul Integr Comp Physiol 315:R1272–R1280. 10.1152/ajpregu.00173.2018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ganzel BL, Morris PA, Wethington E (2010) Allostasis and the human brain: Integrating models of stress from the social and life sciences. Psychol Rev 117:134–174. 10.1037/a0017773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jaminon A, Reesink K, Kroon A, Schurgers L (2019) The role of vascular smooth muscle cells in arterial remodeling: Focus on calcification-related processes. Int J Mol Sci 20:5694. 10.3390/ijms20225694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Casada JH, Roache JD (2006) Dissociation of physiology and behavior in PTSD. Int J Psychophysiol 62:243–248. 10.1016/j.ijpsycho.2006.04.005 [DOI] [PubMed] [Google Scholar]
- 52.Moazen-Zadeh E, Khoshdel A, Avakh F, Rahmani A (2016) Increased blood pressures in veterans with post traumatic stress disorder. Int J Psychiatry Med 51:576–586. 10.1177/0091217417696734 [DOI] [PubMed] [Google Scholar]
- 53.Muth BJ, Brian MS, Chirinos JA, et al. (2017) Central systolic blood pressure and aortic stiffness response to dietary sodium in young and middle-aged adults. J Am Soc Hypertens 11:627–634. 10.1016/j.jash.2017.07.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hansen TW, Thijs L, Boggia J, et al. (2008) Prognostic value of ambulatory heart rate revisited in 6928 subjects from 6 populations. Hypertension 52:229–235. 10.1161/HYPERTENSIONAHA.108.113191 [DOI] [PubMed] [Google Scholar]
- 55.Woodward M, Webster R, Murakami Y, et al. (2014) The association between resting heart rate, cardiovascular disease and mortality: evidence from 112,680 men and women in 12 cohorts. Eur J Prev Cardiol 21:719–726. 10.1177/2047487312452501 [DOI] [PubMed] [Google Scholar]
- 56.Zhang D, Shen X, Qi X (2016) Resting heart rate and all-cause and cardiovascular mortality in the general population: a meta-analysis. CMAJ 188:E53–E63. 10.1503/cmaj.150535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Saz-Lara A, Cavero-Redondo I, Álvarez-Bueno C, et al. (2021) The acute effect of exercise on arterial stiffness in healthy subjects: A meta-analysis. J Clin Med 10:291. 10.3390/jcm10020291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Scherrer JF, Salas J, Schneider FD, et al. (2020) PTSD improvement and incident cardiovascular disease in more than 1000 veterans. J Psychosom Res 134:110128. 10.1016/j.jpsychores.2020.110128 [DOI] [PMC free article] [PubMed] [Google Scholar]


