Abstract
Introduction
Respiratory syncytial virus (RSV) causes a substantial disease burden among infants. In older children and adults, incidence is underestimated due to nonspecific symptoms and limited standard-of-care testing. We aimed to estimate RSV-attributable hospitalizations and deaths in Spain during 2016–2019.
Methods
Nationally representative hospitalization and mortality databases were obtained from the Ministry of Health and the National Statistical Office. A quasi-Poisson regression model was fitted to estimate the number of hospitalizations and deaths attributable to RSV as a function of periodic and aperiodic time trends and viral activity, while allowing for potential overdispersion.
Results
In children, the RSV-attributable respiratory hospitalization incidence was highest among infants aged 0–5 months (3998–5453 cases/100,000 person-years, representing 72% of all respiratory hospitalizations) and decreased with age. In 2019, estimated rates in children 0–5, 6–11, 12–23 months and 6–17 years were approximately 1.3, 1.4, 1.5, and 6.5 times higher than those based on standard-of-care RSV-specific codes. In adults, the RSV-attributable cardiorespiratory hospitalization rate increased with age and was highest among persons ≥ 80 years (1325–1506 cases/100,000, 6.5% of all cardiorespiratory hospitalizations). In 2019, for persons aged 18–49, 50–59, 60–79, and ≥ 80 years, estimated rates were approximately 8, 6, 8, and 16 times higher than those based on standard-of-care RSV-specific codes. The RSV-attributable cardiorespiratory mortality rate was highest among ≥ 80 age group (126–150 deaths/100,000, 3.5–4.1% of all cardiorespiratory deaths), when reported mortality rate ranged between 0 and 0.5/100,000.
Conclusions
When accounting for under-ascertainment, estimated RSV-attributable hospitalizations were higher than those reported based on standard-of-care RSV-specific codes in all age groups but particularly among older children and older adults. Like other respiratory viruses, RSV contributes to both respiratory and cardiovascular complications. Efficacious RSV vaccines could have a high public health impact in these age and risk groups.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40121-024-00920-7.
Keywords: Disease burden, Epidemiology, Hospitalization, Mortality, Respiratory syncytial virus
Key Summary Points
| Why carry out the study? |
| Respiratory syncytial virus (RSV) causes a substantial disease burden among infants. In older children and adults, incidence is underestimated due to nonspecific symptoms and limited standard-of-care testing. |
| We aimed to estimate RSV-attributable hospitalizations and deaths in Spain during 2016–2019 using a quasi-Poisson regression model. |
| What was learned from the study? |
| Estimated respiratory RSV rates in children were up to 6.5 times higher than those based on standard-of-care RSV-specific codes. |
| Estimated cardiorespiratory RSV rates in adults were up to 16 times higher than those based on standard-of-care RSV-specific codes. |
| When accounting for under-ascertainment, estimated RSV-attributable hospitalizations were substantially higher than those reported in all age groups but particularly among older children and older adults. |
| Our study also indicates that, like other respiratory viruses such as influenza and SARS-CoV-2, RSV may contribute to both respiratory and cardiovascular complications in adults. |
| Efficacious RSV vaccines could have a high public health impact in these age groups. |
Introduction
Respiratory syncytial virus (RSV) causes a substantial disease burden, typically with an annual seasonality, peaking during the autumn/winter months in temperate climates [1]. High-risk groups for severe infection include young children and older adults, particularly those with underlying cardiorespiratory conditions [2, 3]. Approximately 33 million annual RSV cases occur globally in children < 5 years, of which 11% are hospitalized [4]. Among older adults, an estimated 787,000 RSV-related hospitalizations are projected to be occurring annually in high-income countries alone [5]. In Spain, RSV-related hospitalization rates were reported to be between 2500 per 100,000 population [6] in children aged < 1 year and 1.7 per 100,000 population [7] in adults aged ≥ 60 years, based on the count of RSV-specific hospital discharge codes. However, these estimates are likely many fold below the true burden for older children and adults due to methodological limitations [8, 9]. Although RSV infection is frequently diagnosed in infants, the disease burden in older children and adults is mostly underestimated due to nonspecific symptoms, limited standard-of-care testing among adults, inadequate diagnostic capacity and lower sensitivity of tests compared to infants [8, 10–17]. In addition, several studies reported that RSV infection not only causes respiratory diseases but often generates new events or acute exacerbations of chronic cardiovascular diseases (such as congestive heart failure, acute coronary syndrome, and arrhythmias) [18–20], which are not typically considered when assessing RSV burden. Therefore, the estimates based on laboratory-confirmed RSV cases generally underestimate the incidence.
To overcome this limitation, several retrospective statistical model-based approaches have been developed to estimate the burden of RSV disease using secondary data sources [21–31]. These models indirectly estimate the proportion of a health outcome attributable to RSV by examining the relationship between changes in the viral activity indicator (e.g., laboratory-confirmed cases or virus-specific hospitalizations) and the corresponding changes in the incidence of the health outcome. Studied outcomes ranged from very broad (e.g., all-cause hospitalizations, all-cause mortality) [26, 32] to more specific (e.g., bronchitis/bronchiolitis hospitalizations, influenza/pneumonia deaths) [25, 33]. These estimates are substantially closer to prospective study estimates than incidence estimates based on standard-of-care testing and RSV-specific ICD-10 codes [34].
In this study, we aimed to estimate the RSV-attributable incidence of cardiorespiratory hospitalizations and deaths in Spain between 2016 and 2019 by applying comprehensive statistical modeling.
Methods
Study Design
This observational retrospective database analysis used a quasi-Poisson regression model to estimate the incidence of RSV-attributable cardiorespiratory hospitalizations and deaths. The model links the temporal variability in RSV represented through a viral activity indicator (RSV-related ICD-coded hospitalizations) with the variability in the number of selected hospitalizations or deaths (outcome), while adjusting for variation in the activity of influenza and baseline seasonality, to estimate the number of hospitalizations or deaths (outcome) associated with RSV.
Data Sources
We obtained anonymized individual-level data on daily hospitalizations from the Ministry of Health’s national hospital discharge database (“Conjunto Mínimo Básico de Datos”, CMBD) that reports more than 90% of all admissions in both public and private institutions [35] and has been validated for data quality [36, 37]; and mortality data from the National Statistical Office’s mortality database (“Base de datos de mortalidad del Instituto Nacional de Estadística”, BDM) that reports 100% of deaths [38] between 2016 and 2019. All diagnoses were coded according to the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) [39].
To estimate the incidence of RSV-attributable hospitalizations and deaths in adults, we selected two primary outcomes: all respiratory events, and cardiorespiratory events (all respiratory plus selected subgroup cardiovascular events) that were previously reported to be linked with RSV [18–20, 40–42] and used in other model-based studies [23, 25, 30, 31]. In addition, we defined four respiratory outcomes (influenza or pneumonia; bronchitis or bronchiolitis; chronic lower respiratory diseases and upper respiratory diseases) and four subgroup cardiovascular outcomes (chronic heart failure exacerbations; ischemic heart diseases; arrhythmias and cerebrovascular diseases) to investigate further the contribution of RSV to those specific outcomes (ICD-10-CM codes provided in Supplementary Table 1). Other model-based studies also investigated similar outcomes [11, 21, 43]. For children, only respiratory events were included.
We considered primary and secondary diagnoses, as reported in hospital discharge records, and the underlying cause of death in death certificates. Hospitalization was defined as an overnight stay in the hospital and was characterized by the admission date. Only the initial hospitalization was counted if readmission for the same outcome occurred within 30 days of admission.
We analyzed nine age categories: 0–5 months, 6–11 months, 12–23 months, 2–5 years, 6–17 years, 18–49 years, 50–59 years, 60–79 years, and ≥ 80 years.
The indicator for RSV activity (an independent variable in model) was defined as weekly (for modeling hospitalizations) or monthly (for modeling mortality) RSV-related hospitalization counts (B97.4, J21.0, J12.1, J20.5, J21.9) among children < 2 years of age. The indicator for influenza activity (another independent variable in model) was defined as weekly or monthly influenza-specific hospitalization counts (J09, J10, J11) in children < 2 years (pediatric model) or in adults ≥ 60 years (adult’s model). We selected these indicators as they were found to be better proxies of viral activity in previous studies [29] because these are ages that have the highest level of testing/illness to track the specific viral activity (e.g., < 2 for infants have high rate of RSV testing and hospitalization for RSV-related bronchiolitis). We included J21.9 (acute bronchiolitis, unspecified) because RSV is the leading cause of bronchiolitis in this age group, causing the majority of bronchiolitis hospitalizations, and because it is used as an indicator of the start of the RSV season [44–48].
To compare the difference between the observed (reported in the database based on standard-of-care testing) versus the attributable (model-based) RSV events, we also counted weekly RSV-specific hospitalizations and deaths (B97.4, J21.0, J12.1, J20.5) per age group.
Statistical Methods
Time series data were stratified by age group and aggregated weekly (hospitalization) or monthly (mortality). A quasi-Poisson regression model was fitted to each age stratum to associate the outcome with aperiodic and periodic time trends and circulation of viral activity (RSV and influenza) while allowing for potential overdispersion. The identity link function was used to reflect the most biologically plausible link between viral circulation and the outcome of interest. The baseline aperiodic trend in the outcome of interest was accounted for in the model through polynomial terms up to the fourth order. To account for the baseline periodic trend, harmonic terms were included in the model. Circulation of RSV and influenza was represented in the model by viral proxies. To account for potential delays between viral testing and the occurrence of the outcome of interest, lagged viral proxies were considered when modeling weekly data.
The general model fitted to weekly data is given by:
with .
where represents the expected number of events in week t with the running week index, the total number of weeks in the study period, and the overdispersion parameter. Parameter is the coefficient associated with the baseline number of events, to are coefficients associated with the aperiodic time trend, to are coefficients associated with the periodic time trend, and are coefficients associated with the appropriately lagged activity of viral pathogen (VP) l with where (RSV and influenza), and the pathogen-specific time lag where .
In the general model fitted to monthly data, is modeled as:
where represents the expected number of events in month t with the running month index, the total number of months in the study period, and parameters , and to defined as before. Parameters are coefficients associated with activity of viral pathogen (VP) l with where L = 2 (RSV and influenza).
Each final model was built in a step-by-step manner. A model containing only the periodic and aperiodic trends was fitted in the first step. When possible (significance level 0.05), the polynomial order was reduced. Next, the viral pathogens were added to the model. When modeling weekly data, all possible lags of all pathogens not yet included in the model were considered for inclusion (one at a time). The lagged pathogen with the highest test statistic was selected for inclusion in the model. This approach has been chosen as it prioritizes positive over negative associations, given that it is biologically implausible for the viral pathogens (RSV and influenza) to protect against the outcome of interest [49, 50]. The viral proxy selection was repeated until all pathogens were included in the final model once.
The weekly (or monthly) number of events attributable to RSV was calculated as the difference between the total model-estimated number of events (using the full model) and the model-estimated number of events under the hypothetical absence of RSV circulation (by setting the parameter associated with RSV to zero). The age-specific annual number of RSV-attributable events was obtained by summing over the included weeks (or months).
RSV-attributable proportions (%) were calculated by dividing the yearly model-based age-specific number of RSV-attributable events by the yearly observed age-specific total number of events (e.g., respiratory hospitalizations).
Age-specific incidence rates of RSV-attributable events per year were obtained by dividing the yearly model-based age-specific number of RSV-attributable events by the yearly age-specific population at risk of the event (from July 1st national census data) [38] and expressed as the number of events per 100,000 person-years. Confidence intervals were obtained through residual bootstrapping. Data management and statistical analysis were performed in R version 4.2.2.
Ethical Considerations
This study involved data in an anonymized structured format and contained no personal information of the patients; therefore, no ethical approval was required.
Results
Observed Events
From January 2016 to December 2019, a total of 263,102 hospitalizations and 175 deaths were coded with a primary respiratory cause in children < 18 years (data not shown). In adults, 5,227,970 hospitalizations and 553,332 deaths were coded with a primary respiratory or cardiovascular cause during the same period (data not shown).
The observed RSV-specific hospitalization rate based on standard-of-care RSV diagnostic coding was highest among infants aged 0–5 months and decreased substantially in older children. Conversely, in adults, the incidence rate increased with age. All rates showed an increasing trend over the study period (Table 1). During the study period, the RSV-specific deaths were rarely reported in children (five deaths) and adults (48 deaths) based on routine underlying cause-of-death data (data not shown). Observed mortality rates ranged between 0 and 0.6 deaths per 100,000 person-years in children aged < 1 year, 0–0.1 deaths per 100,000 person-years in adults aged 60–79 years and between 0–0.5 deaths per 100,000 person-years in those aged ≥ 80 years (data not shown).
Table 1.
RSV-specific hospitalization rate based on standard-of-care diagnostic codes per 100,000 person-years (number of cases) by age group and year, 2016–2019, Spain
| Age group | 2016 | 2017 | 2018 | 2019 |
|---|---|---|---|---|
| 0–5 months | 3292.7 (6751) | 3331.2 (6601) | 4031.3 (7619) | 4150.2 (7509) |
| 6–11 months | 811.1 (1663) | 863.5 (1711) | 1048.2 (1981) | 1250.8 (2263) |
| 12–23 months | 295.6 (1259) | 326.0 (1367) | 406.9 (1656) | 485.6 (1886) |
| 2–5 years | 38.5 (697) | 47.3 (838) | 56.3 (981) | 80.4 (1388) |
| 6–17 years | 1.4 (82) | 1.9 (107) | 1.6 (92) | 2.2 (131) |
| 18–49 years | 0.4 (88) | 0.8 (152) | 0.9 (175) | 1.2 (231) |
| 50–59 years | 1.7 (111) | 3.1 (208) | 3.9 (270) | 4.8 (335) |
| 60–79 years | 5.9 (502) | 11.0 (954) | 14.9 (1330) | 20.1 (1844) |
| ≥ 80 years | 17.4 (492) | 46.2 (1326) | 56.8 (1633) | 95.0 (2717) |
IR incidence rate
Estimated RSV-Attributable Incidence in Children
Hospitalizations
Hospitalization rates of RSV-attributable respiratory events in infants aged 0–5 months ranged from 3998 to 5453 cases per 100,000 person-years in 2016 and 2019, respectively. RSV-attributable cases represented between 65% (2016) and 72% (2019) of all respiratory hospitalizations in this age group, and while the proportion due to RSV decreased with increasing age, it remained substantial: approximately half of respiratory hospitalizations were due to RSV among infants 6–11 months of age, and one-third among those 12–23 months. Among age stratifications < 2 years, both RSV incidence and the proportion of respiratory hospitalizations attributable to RSV increased slightly from 2016 to 2019, while remaining more stable among children 2–17 years (Table 2).
Table 2.
Estimated incidence rate (per 100,000 person-years) and percentage of all respiratory hospitalizations attributable to RSV infections in children by age group and year, 2016–2019, Spain
| 2016 | 2017 | 2018 | 2019 | |||||
|---|---|---|---|---|---|---|---|---|
| Age group | IR [95% CI] | % | IR [95% CI] | % | IR [95% CI] | % | IR [95% CI] | % |
| 0–5 months | 3998.3 [3812.9; 4151.5] | 64.5 | 4329.9 [4129.1; 4495.8] | 69.0 | 5390.0 [5140.1; 5596.6] | 69.1 | 5453.2 [5200.4; 5662.2] | 72.0 |
| 6–11 months | 1285.3 [1151.8;1423.6] | 47.5 | 1391.9 [1247.3;1541.6] | 51.2 | 1732.7 [1552.7; 1919.1] | 51.2 | 1753.0 [1570.9; 1941.6] | 51.8 |
| 12–23 months | 543.9 [460.9; 637.5] | 29.0 | 578.3 [490.0; 677.9] | 30.5 | 707.3 [599.4; 829.1] | 31.1 | 717.9 [608.4; 841.5] | 33.5 |
| 2–5 years | 11.7 [0; 74.9] | 0.9 | 11.0 [0; 70.2] | 1.0 | 11.9 [0; 75.8] | 0.9 | 13.1 [0; 83.3] | 1.1 |
| 6–17 years | 14.1 [0.3; 28.6] | 5.0 | 12.8 [0.3; 25.9] | 4.6 | 13.5 [0.3; 27.3] | 4.7 | 14.6 [0.3; 29.5] | 5.1 |
CI confidence interval, IR incidence rate
In 2019, compared with observed rates, estimated rates were 1.3, 1.4, 1.5, and 6.5 times higher in the 0–5, 6–11, 12–23 months, and 6–17 years age groups, respectively (Fig. 1). Estimated rates in 2–5 years were lower than observed and the model had a suboptimal fit, as the respiratory hospitalizations had less seasonal pattern when compared to infants (Supplementary Fig. 1).
Fig. 1.
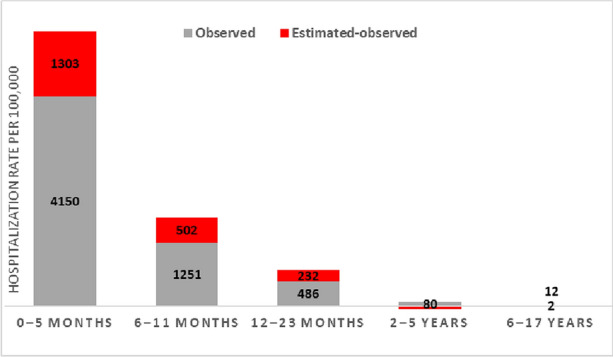
Difference between the observed rate based on standard-of-care diagnoses and estimated RSV-attributable respiratory hospitalization rate (per 100,000 person-years) in children, 2019, Spain
Estimated RSV-attributable incidence in adults
Hospitalizations
Estimated RSV-attributable hospitalization rates for all outcomes are presented in Table 3. In adults aged ≥ 60 years, estimated cardiorespiratory hospitalizations ranged from 438 to 476 cases per 100,000 person-years. Rates increased with age, with the highest one observed among ≥ 80 years (1506 per 100,000 person-years in 2019, representing 6.4% of all cardiorespiratory hospitalizations).
Table 3.
Estimated incidence rate (per 100,000 person-years) and percentage of all hospitalizations attributable to RSV infections in adults by age group and year, 2016–2019, Spain
| 2016 | 2017 | 2018 | 2019 | |||||
|---|---|---|---|---|---|---|---|---|
| IR [95% CI] | % | IR [95% CI] | % | IR [95% CI] | % | IR [95% CI] | % | |
| Primary outcomes | ||||||||
| RSV-attributable cardiorespiratory hospitalizations (all respiratory + selected subgroup cardiovascular outcomes) | ||||||||
| 18–49 years | 9.1 [0.8; 17.3] | 2.4 | 8.5 [0.8; 16.0] | 2.2 | 9.1 [0.8; 17.2] | 2.2 | 9.9 [0.9; 18.7] | 2.4 |
| 50–59 years | 27.0 [5.6; 49.9] | 2.0 | 24.3 [5.1; 44.8] | 1.8 | 25.4 [5.3; 46.9] | 1.7 | 27.2 [5.7; 50.1] | 1.9 |
| 60–79 years | 149.0 [83.4; 218.7] | 2.7 | 144.7 [81.0; 212.4] | 2.6 | 156.1 [87.3; 229.1] | 2.7 | 155.2 [86.8; 227.7] | 2.8 |
| ≥ 80 years | 1361.4 [1035.9; 1696.6] | 6.5 | 1324.9 [1008.1; 1651.1] | 6.0 | 1460.9 [1111.6; 1820.6] | 6.2 | 1505.7 [1145.7; 1876.4] | 6.4 |
| ≥ 60 yearsa | 450.3 | 4.9 | 437.6 | 4.5 | 474.5 | 4.7 | 475.8 | 4.8 |
| RSV-attributable respiratory hospitalizations | ||||||||
| 18–49 years | 5.1 [0; 11,5] | 2.1 | 4.8 [0; 10,7] | 1.9 | 5.1 [0; 11,4] | 1.9 | 5.5 [0; 12,4] | 2.1 |
| 50–59 years | 18.2 [7.1; 29.2] | 3.2 | 16.4 [6.4; 26.3] | 2.9 | 17.1 [6.7; 27.5] | 2.8 | 18.3 [7.1; 29.4] | 3.1 |
| 60–79 years | 114.5 [85.4; 145.2] | 5.8 | 116.0 [86.5; 147.1] | 5.8 | 130.9 [97.6; 165.9] | 6.1 | 125.8 [93.8; 159.4] | 6.1 |
| ≥ 80 years | 702.5 [540.4; 871.0] | 9.5 | 683.6 [525.9; 847.7] | 8.7 | 753.8 [579.9; 934.7] | 8.9 | 776.9 [597.7; 963.3] | 9.4 |
| ≥ 60 yearsa | 260.7 | 7.8 | 256.9 | 7.4 | 282.9 | 7.7 | 280.4 | 7.9 |
| Subgroup outcomes | ||||||||
| RSV-attributable influenza or pneumonia hospitalizations | ||||||||
| 18–49 years | 0.7 [0; 3.2] | 1.3 | 0.8 [0; 3.4] | 1.6 | 0.9 [0; 4.1] | 1.6 | 0.9 [0; 4.0] | 1.5 |
| 50–59 years | 1.6 [0; 7.8] | 1.1 | 1.5 [0; 7.0] | 1.0 | 1.5 [0; 7.3] | 0.9 | 1.6 [0; 7.8] | 1.0 |
| 60–79 years | 36.4 [25.3; 46.8] | 7.3 | 36.8 [25.6; 47.4] | 7.0 | 41.5 [28.9; 53.5] | 7.1 | 39.9 [27.8; 51.4] | 7.0 |
| ≥ 80 years | 229.1 [176.3; 283.5] | 11.8 | 223.0 [171.5; 275.9] | 9.8 | 245.9 [189.1; 304.2] | 10.2 | 253.4 [194.9; 313.5] | 10.9 |
| ≥ 60 yearsa | 84.3 | 9.8 | 83.0 | 8.7 | 91.4 | 8.8 | 90.6 | 9.2 |
| RSV attributable bronchitis or bronchiolitis hospitalizations | ||||||||
| 18–49 years | 1.3 [0.6; 2.1] | 6.9 | 1.3 [0.6; 2.1] | 6.2 | 1.4 [0.6; 2.2] | 5.9 | 1.5 [0.6; 2.4] | 6.7 |
| 50–59 years | 8.3 [5.6; 11.1] | 10.6 | 8.1 [5.4; 10.8] | 9.7 | 8.8 [5.9; 11.7] | 8.7 | 8.8 [5.9; 11.8] | 9.3 |
| 60–79 years | 46.1 [37.0; 55.9] | 13.2 | 46.7 [37.5; 56.6] | 12.1 | 52.7 [42.2; 63.9] | 12.2 | 50.6 [40.6; 61.4] | 12.5 |
| ≥ 80 years | 270.4 [217.4; 331.8] | 16.4 | 263.1 [211.5; 322.9] | 13.4 | 290.1 [233.2; 356.1] | 13.7 | 299.0 [240.4; 367.0] | 14.7 |
| ≥ 60 yearsa | 101.8 | 15.2 | 100.4 | 12.9 | 110.6 | 13.1 | 109.6 | 13.8 |
| RSV-attributable chronic lower respiratory disease hospitalizations | ||||||||
| 18–49 years | 3.1 [1.6; 4.7] | 5.8 | 2.9 [1.5; 4.4] | 5.1 | 3.1 [1.6; 4.7] | 5.1 | 3.4 [1.7; 5.1] | 5.8 |
| 50–59 years | 14.1 [8.5; 19.6] | 5.8 | 12.7 [7.6; 17.6] | 5.2 | 13.2 [8.0; 18.4] | 5.0 | 14.2 [8.5; 19.6] | 5.7 |
| 60–79 years | 58.0 [41.8; 75.7] | 5.5 | 58.8 [42.4; 76.7] | 5.5 | 66.3 [47.8; 86.5] | 6.0 | 63.7 [45.9; 83.2] | 6.0 |
| ≥ 80 years | 213.5 [160.2; 271.2] | 7.6 | 207.7 [155.9; 263.9] | 7.0 | 229.1 [171.9; 291.0] | 7.5 | 236.1 [177.1; 299.9] | 7.9 |
| ≥ 60 yearsa | 96.7 | 6.5 | 95.8 | 6.2 | 106.0 | 6.7 | 104.7 | 6.9 |
| RSV-attributable upper respiratory disease hospitalizations | ||||||||
| 18–49 years | 1.3 [0; 6.0] | 1.2 | 1.2 [0; 5.6] | 1.1 | 1.3 [0; 6.0] | 1.1 | 1.4 [0; 6.5] | 1.2 |
| 50–59 years | 1.8 [0; 6.2] | 1.9 | 1.6 [0; 5.6] | 1.6 | 1.7 [0; 5.8] | 1.6 | 1.8 [0; 6.3] | 1.8 |
| 60–79 years | 2.8 [0; 6.2] | 2.4 | 2.6 [0; 5.6] | 2.1 | 2.7 [0; 5.8] | 2.1 | 2.8 [0; 6.1] | 2.2 |
| ≥ 80 years | 18.4 [13.0; 23.9] | 10.4 | 17.9 [12.7; 23.2] | 9.2 | 19.7 [14.0; 25.6] | 9.8 | 20.3 [14.4; 26.4] | 10.3 |
| ≥ 60 yearsa | 6.7 | 5.1 | 6.4 | 4.5 | 6.8 | 4.7 | 7.0 | 4.9 |
| RSV-attributable chronic heart failure exacerbation hospitalizations | ||||||||
| 18–49 years | 1.1 [0.5; 1.7] | 4.9 | 1.0 [0.4; 1.6] | 4.2 | 1.1 [0.5; 1.7] | 4.5 | 1.2 [0.5; 1.8] | 4.6 |
| 50–59 years | 6.2 [2.9; 9.6] | 4.1 | 5.6 [2.6; 8.6] | 3.5 | 5.8 [2.8; 9.0] | 3.6 | 6.2 [3.0; 9.6] | 3.8 |
| 60–79 years | 24.9 [13.6; 36.2] | 2.8 | 24.1 [13.2; 35.2] | 2.7 | 26.0 [14.3; 37.9] | 2.9 | 25.9 [14.2; 37.7] | 2.9 |
| ≥ 80 years | 277.4 [208.4; 353.9] | 6.0 | 270.0 [202.8; 344.4] | 5.5 | 297.7 [223.6; 379.7] | 5.7 | 306.8 [230.4; 391.4] | 5.8 |
| ≥ 60 yearsa | 87.6 | 4.8 | 85.1 | 4.5 | 92.3 | 4.7 | 92.6 | 4.8 |
| RSV-attributable ischemic heart disease hospitalizations | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | 2.4 [0; 8.0] | 0.8 | 2.2 [0; 7.2] | 0.7 | 2.3 [0; 7.5] | 0.7 | 2.4 [0; 8.0] | 0.8 |
| 60–79 years | 26.7 [12.4; 41.1] | 2.8 | 24.0 [11.2; 37.1] | 2.5 | 24.9 [11.6; 38.4] | 2.5 | 26.3 [12.2; 40.6] | 2.7 |
| ≥ 80 years | 92.4 [61.0; 128.4] | 4.0 | 89.9 [59.4; 124.9] | 3.7 | 99.1 [65.5; 137.8] | 4.0 | 102.2 [67.5; 142.0] | 4.2 |
| ≥ 60 yearsa | 43.0 | 3.3 | 40.4 | 3.0 | 43.0 | 3.2 | 44.3 | 3.4 |
| RSV-attributable arrythmias hospitalizations | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | 6.0 [1.1; 11.2] | 3.1 | 5.4 [1.0; 10.1] | 2.7 | 5.6 [1.0; 10.6] | 2.8 | 6.0 [1.1; 11.3] | 2.9 |
| 60–79 years | 29.0 [11.2; 47.1] | 2.5 | 26.1 [10.1; 42.4] | 2.2 | 27.1 [10.4; 44.0] | 2.2 | 28.6 [11.0; 46.4] | 2.4 |
| ≥ 80 years | 272.5 [198.1; 345.2] | 5.4 | 265.2 [192.8; 335.9] | 4.9 | 292.4 [212.6; 370.4] | 5.1 | 301.3 [219.1; 381.8] | 5.2 |
| ≥ 60 yearsa | 89.5 | 4.2 | 85.4 | 3.9 | 91.8 | 4.0 | 93.3 | 4.1 |
| RSV-attributable cerebrovascular hospitalizations | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 60–79 years | 10.3 [3.7; 17.0] | 2.2 | 9.3 [3.3; 15.3] | 1.9 | 9.7 [3.5; 15.9] | 2.0 | 10.2 [3.7; 16.8] | 2.0 |
| ≥ 80 years | 20.9 [4.3; 37.6] | 1.4 | 18.9 [3.8; 33.9] | 1.2 | 20.0 [4.1; 36.0] | 1.3 | 21.9 [4.5; 39.4] | 1.4 |
| ≥ 60 yearsa | 13.0 | 1.8 | 11.7 | 1.6 | 12.2 | 1.6 | 13.0 | 1.7 |
Myocarditis outcome was NA in all categories
CI confidence interval, IR incidence rate, NA not applicable since data is unsuitable for modeling or insufficient model fit
aBased on pooling of results for models fitted to adults, thus confidence intervals are not provided. Negative estimates were suppressed to zero because of biological implausibility
In 2019, compared with the observed ones, estimated RSV-attributable cardiorespiratory hospitalization rates were approximately 8, 6, 8, and 16 times higher in the 18–49, 50–59, 60–79, and ≥ 80 age groups, respectively (Fig. 2).
Fig. 2.
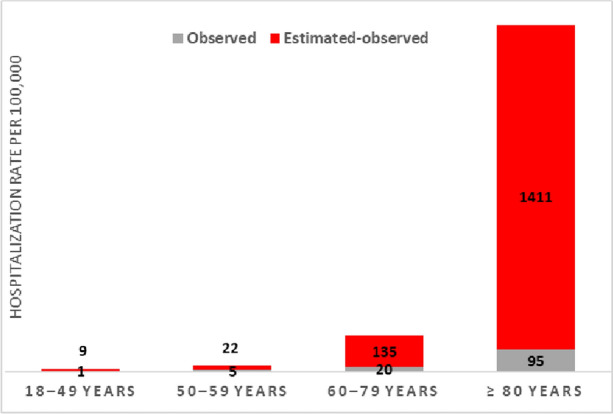
Difference between the observed standard-of-care and estimated RSV-attributable cardiorespiratory hospitalization rate (per 100,000 person-years) in adults, 2019, Spain
The incidence of subgroup outcomes was highest for chronic heart failure exacerbations, arrhythmias, and bronchitis/bronchiolitis in the oldest age group. In contrast, younger age groups had higher rates for chronic lower respiratory diseases and bronchitis/bronchiolitis (Table 3). Upper respiratory and cerebrovascular diseases were characterized by low RSV-attributable incidence and lack of seasonality in these youngest age groups.
Mortality
Estimated RSV-attributable mortality rates for all outcomes are presented in Table 4. The highest RSV-attributable mortality rates among all age groups were for selected cardiorespiratory diseases, followed by all respiratory diseases. Among subgroup outcomes, estimated deaths caused by ischemic heart diseases (in age groups ≥ 80 and 60–79 years) and chronic heart failure (in age group ≥ 80 years) accounted for the highest rates. In the youngest age group (18–49 years), the mortality data for most of secondary outcomes were randomly distributed without clear seasonal pattern, thus not modeled.
Table 4.
Estimated mortality rate (per 100,000 person-years) and percentage of all deaths attributable to RSV infections in adults by age group and year, 2016–2019, Spain
| 2016 | 2017 | 2018 | 2019 | |||||
|---|---|---|---|---|---|---|---|---|
| MR [95% CI] | % | MR [95% CI] | % | MR [95% CI] | % | MR [95% CI] | % | |
| Primary outcomes | ||||||||
| RSV-attributable cardiorespiratory deaths (all respiratory + selected cardiovascular subgroup outcomes) | ||||||||
| 18–49 years | 0.3 [0.0; 0.7] | 2.4 | 0.3 [0.0; 0.7] | 2.6 | 0.4 [0.0; 0.8] | 3 | 0.4 [0.0; 0.8] | 3 |
| 50–59 years | 1.5 [0.0; 3.1] | 2.1 | 1.5 [0.0; 3.1] | 2.2 | 1.8 [0.0; 3.6] | 2.4 | 1.7 [0.0; 3.4] | 2.3 |
| 60–79 years | 8.7 [1.7; 15.2] | 2.4 | 8.8 [1.7; 15.2] | 2.5 | 10.2 [2.0; 17.6] | 2.9 | 9.3 [1.8; 16.1] | 2.8 |
| ≥ 80 years | 125.9 [47.6; 200.9] | 3.7 | 126.9 [47.9; 202.4] | 3.5 | 150.0 [56.7; 239.3] | 4.1 | 142.1 [53.7; 226.6] | 4.2 |
| ≥ 60 yearsa | 37.9 | 3.4 | 38.1 | 3.3 | 44.3 | 3.9 | 40.8 | 3.9 |
| RSV-attributable respiratory deaths | ||||||||
| 18–49 years | 0.0 [0; 0.2] | 0.5 | 0.0 [0; 0.2] | 0.6 | 0.0 [0; 0.2] | 0.6 | 0.0 [0; 0.2] | 0.5 |
| 50–59 years | 0.5 [0; 1.2] | 3 | 0.5 [0; 1.2] | 3.1 | 0.6 [0; 1.4] | 3.2 | 0.6 [0; 1.3] | 3 |
| 60–79 years | 3.2 [0; 6.6] | 2.7 | 3.3 [0; 6.6] | 2.7 | 3.8 [0; 7.7] | 3 | 3.4 [0; 7.0] | 3.1 |
| ≥ 80 years | 49.0 [0; 100.1] | 4 | 49.4 [0; 100.8] | 3.6 | 58.4 [0; 119.2] | 4.1 | 55.3 [0; 112.9] | 4.5 |
| ≥ 60 yearsa | 14.6 | 3.7 | 14.7 | 3.4 | 17.1 | 3.9 | 15.8 | 4.2 |
| Subgroup outcomes | ||||||||
| RSV-attributable influenza or pneumonia deaths | ||||||||
| 18–49 years | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 50–59 years | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 60–79 years | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| ≥ 80 years | 3.1 [0; 18.3] | 1.2 | 3.1 [0; 18.4] | 1 | 3.7 [0; 21.8] | 1.1 | 3.5 [0; 20.6] | 1.2 |
| ≥ 60 yearsa | NA | NA | NA | NA | NA | NA | NA | NA |
| RSV-attributable bronchitis or bronchiolitis deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 60–79 years | 0.2 [0.0; 0.3] | 10.9 | 0.2 [0.0; 0.3] | 10 | 0.2 [0.0; 0.4] | 11.9 | 0.2 [0.0; 0.4] | 10.1 |
| ≥ 80 years | 3.8 [0.6; 7.0] | 9.4 | 3.9 [0.6; 7.1] | 7.5 | 4.6 [0.7; 8.4] | 8.8 | 4.3 [0.7; 7.9] | 8.1 |
| ≥ 60 yearsa | 1.1 | 9.6 | 1.1 | 7.7 | 1.3 | 9.1 | 1.2 | 8.3 |
| RSV-attributable chronic lower respiratory disease deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | 0.1 [0; 0.4] | 1.1 | 0.1 [0; 0.4] | 1 | 0.1 [0; 0.4] | 1.3 | 0.1 [0; 0.4] | 1.1 |
| 60–79 years | 2.2 [0.2; 4.3] | 4.1 | 2.2 [0.2; 4.3] | 4.2 | 2.5 [0.3; 5.0] | 5.1 | 2.3 [0.3; 4.6] | 4.8 |
| ≥ 80 years | 15.5 [4.4; 26.4] | 4.3 | 15.6 [4.4; 26.6] | 4.3 | 18.5 [5.2; 31.4] | 5.4 | 17.5 [4.9; 29.8] | 5.6 |
| ≥ 60 yearsa | 5.5 | 4.2 | 5.5 | 4.2 | 6.4 | 5.3 | 5.9 | 5.3 |
| RSV-attributable upper respiratory disease deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 60–79 years | NA | NA | NA | NA | NA | NA | NA | NA |
| ≥ 80 years | 0.3 [0.1; 0.6] | 28.5 | 0.3 [0.1; 0.7] | 16 | 0.4 [0.1; 0.8] | 20.3 | 0.4 [0.1; 0.7] | 19.8 |
| ≥ 60 yearsa | NA | NA | NA | NA | NA | NA | NA | NA |
| RSV-attributable chronic heart failure exacerbation deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | 0.3 [0.0; 0.7] | 3.2 | 0.3 [0.0; 0.7] | 3.3 | 0.4 [0.0; 0.8] | 3.7 | 0.4 [0.0; 0.7] | 3 |
| 60–79 years | 1.5 [0.4; 2.7] | 3.6 | 1.5 [0.4; 2.7] | 3.7 | 1.8 [0.5; 3.2] | 4.5 | 1.6 [0.4; 2.9] | 4.1 |
| ≥ 80 years | 21.3 [9.7; 32.9] | 3.6 | 21.5 [9.8; 33.2] | 3.4 | 25.4 [11.6; 39.2] | 4 | 24.0 [11.0; 37.2] | 3.9 |
| ≥ 60 yearsa | 6.5 | 3.6 | 6.5 | 3.5 | 7.5 | 4.1 | 7.0 | 3.9 |
| RSV-attributable ischemic heart disease deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | 0.6 [0; 1.4] | 2 | 0.6 [0; 1.4] | 2.1 | 0.8 [0; 1.6] | 2.5 | 0.7 [0; 1.5] | 2.4 |
| 60–79 years | 3.4 [1.5; 5.4] | 3 | 3.5 [1.5; 5.4] | 3.1 | 4.0 [1.8; 6.3] | 3.7 | 3.7 [1.6; 5.7] | 3.6 |
| ≥ 80 years | 26.3 [12.0; 39.4] | 3.8 | 26.5 [12.1; 39.7] | 3.8 | 31.3 [14.3; 46.9] | 4.8 | 29.7 [13.5; 44.4] | 4.9 |
| ≥ 60 yearsa | 9.1 | 3.6 | 9.2 | 3.6 | 10.7 | 4.4 | 9.8 | 4.5 |
| RSV-attributable arrythmias deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 60–79 years | 0.1 [0; 0.5] | 1.1 | 0.1 [0; 0.5] | 1.2 | 0.2 [0; 0.6] | 1.2 | 0.2 [0; 0.6] | 1.2 |
| ≥ 80 years | 7.6 [1.3; 13.3] | 3 | 7.6 [1.4; 13.4] | 2.8 | 9.0 [1.6; 15.9] | 3.2 | 8.5 [1.5; 15.0] | 3.2 |
| ≥ 60 yearsa | 2.0 | 2.7 | 2.0 | 2.6 | 2.3 | 2.9 | 2.1 | 2.9 |
| RSV-attributable cerebrovascular disease deaths | ||||||||
| 18–49 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 50–59 years | NA | NA | NA | NA | NA | NA | NA | NA |
| 60–79 years | 0.7 [0; 1.9] | 1 | 0.7 [0; 1.9] | 1 | 0.8 [0; 2.2] | 1.2 | 0.7 [0; 2.0] | 1.1 |
| ≥ 80 years | 20.0 [8.9; 29.4] | 3 | 20.1 [9.0; 29.7] | 3.1 | 23.8 [10.6; 35.1] | 3.7 | 22.5 [10.1; 33.2] | 3.6 |
| ≥ 60 yearsa | 5.5 | 2.5 | 5.5 | 2.6 | 6.4 | 3.1 | 5.9 | 3.0 |
Myocarditis outcome was NA in all categories
CI confidence interval, MR mortality rate, NA not applicable since data is unsuitable for modeling or insufficient model fit
aBased on pooling of results for models fitted to adults, thus confidence intervals are not provided Negative estimates were suppressed to be zero because of biological implausibility
Discussion
Time-series studies compare the variability in pathogen-indicators (here RSV and influenza) to variability in broad all-cause health outcomes that are could be related to these pathogens (e.g., all-cause respiratory hospitalizations), to estimate the proportion of these all-cause events that can be attributed to these pathogens. Our model-based study found a high burden of RSV-attributable hospitalizations in Spain, with incidences substantially higher than those reported based on RSV-specific codes in all ages, particularly among older children and adults. We also found that a considerable burden of adult cardiovascular hospitalizations and deaths, specifically for chronic heart failure and arrhythmias, was attributed to RSV by the time-series model.
As expected, our estimates diverged substantially from results in another Spanish study that used the same database but did not incorporate time-series modeling to account for untested/undiagnosed events. The annual RSV-specific hospitalization rate in adults aged ≥ 60 years was 1.7 per 100,000 for the 2012–2020 period [7], while our estimates ranged from 257–283 per 100,000 (if we only consider respiratory hospitalizations) and 438–476 per 100,000 (if we consider cardiorespiratory hospitalizations). Our estimates in turn were comparable to prospective study results from two recent systematic literature reviews and meta-analyses that adjusted for diagnostic testing based under-ascertainment: a United States study with a pooled annual incidence estimate of 282 cases per 100,000 population [34] and a global study of high-income countries with a pooled estimate of 347 cases per 100,000 population [5] in those aged ≥ 65 years. By contrast, two model-based studies from the United Kingdom reported lower incidences [11, 49], a result that may be explained primarily by two methodological differences. First, we observed that using surveillance data as RSV indicator (as these studies did, rather than pediatric RSV-related hospitalization) can reduce peak outcome estimates and decrease capture of RSV activity outside of the influenza season, when testing is more frequent [9]. Deriving the RSV proxy from the same dataset as the outcomes ensures an exact match regarding geographic representation of the proxy and outcome data, which may not be present when using sentinel viral surveillance data as the proxy. Second, our respiratory and cardiorespiratory disease outcomes groupings used both primary and secondary diagnosis codes. This allows for events with other primary codes (e.g., viral illness or sepsis) to be included in the modeling. This approach has been used in other recent publications [11, 29]. Nevertheless, our calculated proportions of RSV-attributable cases of hospitalizations were comparable to these studies, ranging from 4 to 10%, depending on age [11, 34].
We found that under-ascertainment based on standard-of-care diagnostic codes increased as age increased. This observation can be explained by the increased use of nonspecific lower respiratory tract infection codes –such as unspecified bronchiolitis, bronchitis or pneumonia– as age increased [51], exacerbated by infrequent testing. Studies from Spain and other countries also support lower testing frequency and reporting in older children [52, 53] and adults [8]. The opposite was seen for children aged 2–5 years, among whom the estimated incidence rates (11–13 cases per 100,000) were lower than those based on RSV-specific codes (39–80 cases per 100,000). This is likely due to the fact that the observed respiratory hospitalizations frequency in this age group did not have a clearly seasonal pattern, which prevents the model from accurately attributing a percent of the outcome to RSV. The reason for the lack of seasonality is not clear but it may be that more respiratory hospitalizations are associated with chronic disease exacerbations which can have non-infectious triggers, e.g., asthma exacerbations can have allergic and infectious triggers. Similar limitations were also observed in other modeling studies, where estimates for RSV-attributable hospitalizations in children aged 5–17 years were negative [33] or equal to zero [22, 54].
As expected, the RSV-attributable hospitalization rates of respiratory events in children obtained from our model were slightly higher than those reported in other Spanish observational studies using RSV-specific codes [6, 55, 56]. Nevertheless, although comparisons must be made with caution since the study periods and regions were not the same, our estimates are in line with results from other countries, such as the United Kingdom, where the modeled incidence for 0–5 months was 4184 per 100,000 [54] (range of 3998–5453 in our study). In the United States, modeled results were slightly lower (1500–2800 cases [29] and 1,896 per 100,000 [28] in children < 1 year of age).
Based on the literature establishing that some cardiovascular outcomes can be provoked/related to RSV infections [19, 21, 22, 30, 31], we included cardiovascular and respiratory diseases in our modeled outcomes. This study’s definition included only selected specific cardiovascular outcomes since not all of them showed marked seasonality. The seasonal variation allows the linkage between the season viral activity and the variability in the all-cause outcomes through time-series modeling. We estimated that in adults ≥ 80 years, RSV may have been related to approximately 6%, 4% and 5% of all hospitalizations for chronic heart failure exacerbations, ischemic heart diseases, and arrhythmias, respectively. This finding from our model is supported by previous literature that has established that respiratory viruses, including RSV, can exacerbate chronic cardiovascular diseases, such as coronary artery disease, and trigger new cardiovascular events [20, 57]. It has been described that inflammation, prothrombotic imbalance, and increased metabolic demands of the myocardium can worsen or trigger myocardial ischemia and arrhythmias [19, 42, 58]. Respiratory viruses were also found to be one of the most common etiological factors of myocarditis [59]. However, myocarditis hospitalizations did not show a clear seasonal pattern in our study.
As expected, RSV-attributable mortality rates were highest in the oldest age group, among whom RSV was related with 4–5% of selected cardiorespiratory deaths. Although our estimates were slightly lower than those reported in the United Kingdom (1, 8, 37, and 198 deaths per 100,000 in people 18–49, 50–64, 65–74, and ≥ 75 years, respectively), the proportion of deaths attributable to RSV in older adults was similar for cardiorespiratory and respiratory causes (ranged between 2 and 5%) [11]. This might be also related to a different definition of cardiorespiratory deaths, as we selected more specific outcomes than all cardiorespiratory causes (i.e., J00-J99I00-I99) [11]. Spain has the second highest life expectancy in Europe [60], which may in part account for the lower mortality rate. However, our estimates (14.6–17.1 deaths per 100,000 person-years) of RSV-attributable mortality rate of underlying respiratory deaths in adults ≥ 60 years were comparable to results of a model-based study in the United States (14.7 deaths per 100,000 in people ≥ 65 years of age) [61]. Due to a low number of deaths among children, data were not modeled.
The main strength of our study was the inclusion of two population-based, nationally representative databases, which reduced the risk of selection bias. In addition, primary and subgroup outcome definitions, including cardiovascular causes, allowed us to explore a wider spectrum of conditions described in the literature associated with RSV infection. Our final statistical model was based on an extensive literature review and experts’ input.
However, several limitations can be described. Firstly, we captured only severe cases (hospitalizations and deaths) and thus likely underestimated the total burden of RSV disease in the population. Secondly, we could not stratify by risk groups due to data limitations. Thirdly, as an RSV activity proxy, we used hospitalization data from children aged < 2 years (consistent with other publication [29]), which may not reflect the exact temporality of circulation of the virus in the adult population. To account for this, we selected the best lag between the proxy in children and the outcome in adults to reflect the sequential pattern of infection, usually starting in children and spreading to older age groups [62]. In addition, we only included RSV and influenza in our analysis, implicitly assuming that these are the only two pathogens that show a relevant association with the outcome of interest. Nevertheless, even without explicitly modeling other potentially relevant pathogens, they are indirectly accounted for in the model through the periodic component and the overdispersion parameter to a great extent. Lastly, by using a modeling approach, we cannot confirm causal inference.
In conclusion, our study suggests that a high number of RSV-related hospitalizations and deaths may not have been correctly identified in Spain, largely due to low rates of standard-of-care RSV testing and diagnosis outside of the infant/toddler period. This underestimation, which is higher in older children and older adults, should be considered when assessing the potential benefit of preventive measures such as vaccination. Our study also indicates that, like other respiratory viruses such as influenza and SARS-CoV-2, RSV may contribute to both respiratory and cardiovascular complications. In these age and risk groups, implementation of preventive measures such as vaccines and effective RSV antivirals would have a substantial impact on health.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Editorial assistance in the preparation of this article was provided by Dr. Somsuvro Basu of P95. Support for this assistance was funded by Pfizer Ltd. The datasets generated during and/or analyzed during the current study are not publicly available due to restriction of data provider.
Funding
This study was funded by Pfizer Ltd., including the journal's Rapid Service Fee.
Data Availability
The datasets analyzed during the current study are available, upon request, from the Ministry of Health and National Statistical Office repositories. Registro de Actividad de Atención Especializada RAE-CMBD 2022 Ministerio de Sanidad; 2022 [Available from: https://www.sanidad.gob.es/estadEstudios/estadisticas/cmbdhome.htm]. Instituto Nacional de Estadística. Base de datos de mortalidad (Demografía y población/Fenómenos demográficos/Tablas de mortalidad); 2022 [Available from: https://www.ine.es].
Declarations
Conflict of Interest
Mariana Haeberer, Caihua Liang, Charles Nuttens, Jessica Atwell, Cristina Mendez Diez, Bradford D. Gessner, Elizabeth Begier are employees of Pfizer and may own Pfizer stock. Robin Bruyndonckx, Aleksandra Polkowska-Kramek, Maribel Casas, Francesca Lemme, Worku Biyadgie Ewnetu, and Thao Mai Phuong Tran are employees of P95, which received funding from Pfizer to conduct this study and for manuscript development. Antoni Torres received funding to be a speaker for Pfizer, MSD y Janssen.
Ethical Approval
This study involved data in an anonymized structured format and contained no patient personal information; therefore, no ethical approval was required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Chadha M, Hirve S, Bancej C, Barr I, Baumeister E, Caetano B, et al. Human respiratory syncytial virus and influenza seasonality patterns-early findings from the WHO global respiratory syncytial virus surveillance. Influenza Other Respir Viruses. 2020;14(6):638–646. doi: 10.1111/irv.12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Stein RT, Bont LJ, Zar H, Polack FP, Park C, Claxton A, et al. Respiratory syncytial virus hospitalization and mortality: systematic review and meta-analysis. Pediatr Pulmonol. 2017;52(4):556–569. doi: 10.1002/ppul.23570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nguyen-Van-Tam JS, O'Leary M, Martin ET, Heijnen E, Callendret B, Fleischhackl R, et al. Burden of respiratory syncytial virus infection in older and high-risk adults: a systematic review and meta-analysis of the evidence from developed countries. Eur Respir Rev. 2022;31(166):220105. doi: 10.1183/16000617.0105-2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li Y, Wang X, Blau DM, Caballero MT, Feikin DR, Gill CJ, et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet. 2022;399(10340):2047–2064. doi: 10.1016/S0140-6736(22)00478-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li Y, Kulkarni D, Begier E, Wahi-Singh P, Wahi-Singh B, Gessner B, et al. Adjusting for case under-ascertainment in estimating RSV hospitalisation burden of older adults in high-income countries: a systematic review and modelling study. Infect Dis Ther. 2023;12(4):1137–1149. doi: 10.1007/s40121-023-00792-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Martinón-Torres F, Carmo M, Platero L, Drago G, López-Belmonte JL, Bangert M, et al. Clinical and economic burden of respiratory syncytial virus in Spanish children: the BARI study. BMC Infect Dis. 2022;22(1):759. doi: 10.1186/s12879-022-07745-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heppe-Montero M, Gil-Prieto R, Del Diego SJ, Hernández-Barrera V, Gil-de-Miguel Á. Impact of respiratory syncytial virus and influenza virus infection in the adult population in Spain between 2012 and 2020. Int J Environ Res Public Health. 2022;19(22):14680. doi: 10.3390/ijerph192214680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rozenbaum MH, Judy J, Tran D, Yacisin K, Kurosky SK, Begier E. Low levels of RSV testing among adults hospitalized for lower respiratory tract infection in the United States. Infect Dis Ther. 2023;12(2):677–685. doi: 10.1007/s40121-023-00758-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Datta S, Walsh EE, Peterson DR, Falsey AR. Can analysis of routine viral testing provide accurate estimates of respiratory syncytial virus disease burden in adults? J Infect Dis. 2017;215(11):1706–1710. doi: 10.1093/infdis/jix196. [DOI] [PubMed] [Google Scholar]
- 10.Muscatello DJ, Amin J, MacIntyre CR, Newall AT, Rawlinson WD, Sintchenko V, et al. Inaccurate ascertainment of morbidity and mortality due to influenza in administrative databases: a population-based record linkage study. PLoS ONE. 2014;9(5):e98446. doi: 10.1371/journal.pone.0098446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fleming DM, Taylor RJ, Lustig RL, Schuck-Paim C, Haguinet F, Webb DJ, et al. Modelling estimates of the burden of respiratory syncytial virus infection in adults and the elderly in the United Kingdom. BMC Infect Dis. 2015;15:443. doi: 10.1186/s12879-015-1218-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Branche AR, Falsey AR. Respiratory syncytial virus infection in older adults: an under-recognized problem. Drugs Aging. 2015;32(4):261–269. doi: 10.1007/s40266-015-0258-9. [DOI] [PubMed] [Google Scholar]
- 13.Lee N, Walsh EE, Sander I, Stolper R, Zakar J, Wyffels V, et al. Delayed diagnosis of respiratory syncytial virus infections in hospitalized adults: individual patient data, record review analysis and physician survey in the United States. J Infect Dis. 2019;220(6):969–979. doi: 10.1093/infdis/jiz236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Talbot HK, Belongia EA, Walsh EE, Schaffner W. Respiratory syncytial virus in older adults. A hidden annual epidemic. Infect Dis Clin Pract. 2016;24(6):295–302. doi: 10.1097/IPC.0000000000000455. [DOI] [Google Scholar]
- 15.Chartrand C, Tremblay N, Renaud C, Papenburg J. Diagnostic accuracy of rapid antigen detection tests for respiratory syncytial virus infection: systematic review and meta-analysis. J Clin Microbiol. 2015;53(12):3738–3749. doi: 10.1128/JCM.01816-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Onwuchekwa C, Atwell J, Moreo LM, Menon S, Machado B, Siapka M, et al. Pediatric RSV diagnostic testing performance: a systematic review and meta-analysis. J Infect Dis. 2023;228:1516–1527. doi: 10.1093/infdis/jiad185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ramirez J, Carrico R, Wilde A, Junkins A, Furmanek S, Chandler T, et al. Diagnosis of respiratory syncytial virus in adults substantially increases when adding sputum, saliva, and serology testing to nasopharyngeal swab RT-PCR. Infect Dis Ther. 2023;12(6):1593–1603. doi: 10.1007/s40121-023-00805-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kujawski SA, Whitaker M, Ritchey MD, Reingold AL, Chai SJ, Anderson EJ, et al. Rates of respiratory syncytial virus (RSV)-associated hospitalization among adults with congestive heart failure-United States, 2015–2017. PLoS ONE. 2022;17(3):e0264890. doi: 10.1371/journal.pone.0264890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ivey KS, Edwards KM, Talbot HK. Respiratory syncytial virus and associations with cardiovascular disease in adults. J Am Coll Cardiol. 2018;71(14):1574–1583. doi: 10.1016/j.jacc.2018.02.013. [DOI] [PubMed] [Google Scholar]
- 20.Franczuk P, Tkaczyszyn M, Kulak M, Domenico E, Ponikowski P, Jankowska EA. Cardiovascular complications of viral respiratory infections and COVID-19. Biomedicines. 2022 doi: 10.3390/biomedicines11010071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schanzer DL, Langley JM, Tam TW. Role of influenza and other respiratory viruses in admissions of adults to Canadian hospitals. Influenza Other Respir Viruses. 2008;2(1):1–8. doi: 10.1111/j.1750-2659.2008.00035.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matias G, Taylor RJ, Haguinet F, Schuck-Paim C, Lustig RL, Shinde V. Estimates of hospitalization attributable to influenza and RSV in the US during 1997–2009, by age and risk status. BMC Public Health. 2017;17(1):271. doi: 10.1186/s12889-017-4177-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou H, Thompson WW, Viboud CG, Ringholz CM, Cheng PY, Steiner C, et al. Hospitalizations associated with influenza and respiratory syncytial virus in the United States, 1993–2008. Clin Infect Dis. 2012;54(10):1427–1436. doi: 10.1093/cid/cis211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Muller-Pebody B, Crowcroft NS, Zambon MC, Edmunds WJ. Modelling hospital admissions for lower respiratory tract infections in the elderly in England. Epidemiol Infect. 2006;134(6):1150–1157. doi: 10.1017/S0950268806006376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Matias G, Taylor RJ, Haguinet F, Schuck-Paim C, Lustig RL, Shinde V. Estimates of mortality attributable to influenza and RSV in the United States during 1997–2009 by influenza type or subtype, age, cause of death, and risk status. Influenza Other Respir Viruses. 2014;8(5):507–515. doi: 10.1111/irv.12258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hardelid P, Pebody RG, Andrews N. Mortality caused by influenza and respiratory syncytial virus by age group in England and Wales 1999–2010. Influenza Other Respir Viruses. 2013;7(1):35–45. doi: 10.1111/j.1750-2659.2012.00345.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Griffin MR, Coffey CS, Neuzil KM, Mitchel EF, Jr, Wright PF, Edwards KM. Winter viruses: influenza- and respiratory syncytial virus-related morbidity in chronic lung disease. Arch Intern Med. 2002;162(11):1229–1236. doi: 10.1001/archinte.162.11.1229. [DOI] [PubMed] [Google Scholar]
- 28.Goldstein E, Greene SK, Olson DR, Hanage WP, Lipsitch M. Estimating the hospitalization burden associated with influenza and respiratory syncytial virus in New York City, 2003–2011. Influenza Other Respir Viruses. 2015;9(5):225–233. doi: 10.1111/irv.12325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zheng Z, Warren JL, Shapiro ED, Pitzer VE, Weinberger DM. Estimated incidence of respiratory hospitalizations attributable to RSV infections across age and socioeconomic groups. Pneumonia (Nathan) 2022;14(1):6. doi: 10.1186/s41479-022-00098-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mullooly J, Bridges C, Thompson W, Chen J, Weintraub E, Jackson L, et al. Influenza- and RSV-associated hospitalizations among adults. Vaccine. 2007;25(5):846–855. doi: 10.1016/j.vaccine.2006.09.041. [DOI] [PubMed] [Google Scholar]
- 31.Thompson WW, Shay DK, Weintraub E, Brammer L, Cox N, Anderson LJ, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA. 2003;289(2):179–186. doi: 10.1001/jama.289.2.179. [DOI] [PubMed] [Google Scholar]
- 32.Karstaedt AS. Influenza- and respiratory syncytial virus-associated adult mortality in Soweto. S Afr Med J. 2009;99(10):750–754. [PubMed] [Google Scholar]
- 33.Nazareno AL, Muscatello DJ, Turner RM, Wood JG, Moore HC, Newall AT. Modelled estimates of hospitalisations attributable to respiratory syncytial virus and influenza in Australia, 2009–2017. Influenza Other Respir Viruses. 2022;16(6):1082–1090. doi: 10.1111/irv.13003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.McLaughlin JM, Khan F, Begier E, Swerdlow DL, Jodar L, Falsey AR. Rates of medically attended RSV among US adults: a systematic review and meta-analysis. Open Forum Infect Dis. 2022;9(7):ofac300. doi: 10.1093/ofid/ofac300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Registro de Actividad de Atención Especializada. RAE-CMBD 2022 Ministerio de Sanidad; 2022. https://www.sanidad.gob.es/estadEstudios/estadisticas/cmbdhome.htm.
- 36.Quirós-González V, Bernal JL, Haro-Pérez AM, Maderuelo-Fernández JA, Santos-Jiménez MT, García-Barrio N, et al. Validity and usefulness of the RAE-CMBD studying patients hospitalised with influenza. Rev Esp Quimioter. 2023;36(2):160–168. doi: 10.37201/req/074.2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rodríguez del Águila M P-ME, Librero J, BuzónBarrera M, Rivas Ruiz Atlas VPM Nº 4. Análisis del control de calidad del Conjunto Mínimo de Datos Básicos de Andalucía en los años 2000 a 2003. Sevilla, 2004.
- 38.Instituto Nacional de Estadística. Base de datos de mortalidad, Base de datos de mortalidad (Demografía y población/Fenómenos demográficos/Tablas de mortalidad), 2022. Available from: https://www.ine.es.
- 39.WHO . International statistical classification of diseases and related health problems. 10. World Health Organization; 2016. [Google Scholar]
- 40.Eisenhut M. Extrapulmonary manifestations of severe respiratory syncytial virus infection–a systematic review. Crit Care. 2006;10(4):R107. doi: 10.1186/cc4984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kawashima H, Inagaki N, Nakayama T, Morichi S, Nishimata S, Yamanaka G, et al. Cardiac complications caused by respiratory syncytial virus infection: questionnaire survey and a literature review. Glob Pediatr Health. 2021 doi: 10.1177/2333794X211044114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Corrales-Medina VF, Madjid M, Musher DM. Role of acute infection in triggering acute coronary syndromes. Lancet Infect Dis. 2010;10(2):83–92. doi: 10.1016/S1473-3099(09)70331-7. [DOI] [PubMed] [Google Scholar]
- 43.Schanzer DL, Saboui M, Lee L, Nwosu A, Bancej C. Burden of influenza, respiratory syncytial virus, and other respiratory viruses and the completeness of respiratory viral identification among respiratory inpatients, Canada, 2003–2014. Influenza Other Respir Viruses. 2018;12(1):113–121. doi: 10.1111/irv.12497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Calvo C, Pozo F, García-García ML, Sanchez M, Lopez-Valero M, Pérez-Breña P, et al. Detection of new respiratory viruses in hospitalized infants with bronchiolitis: a three-year prospective study. Acta Paediatr. 2010;99(6):883–887. doi: 10.1111/j.1651-2227.2010.01714.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Mansbach JM, Piedra PA, Teach SJ, Sullivan AF, Forgey T, Clark S, et al. Prospective multicenter study of viral etiology and hospital length of stay in children with severe bronchiolitis. Arch Pediatr Adolesc Med. 2012;166(8):700–706. doi: 10.1001/archpediatrics.2011.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kenmoe S, Kengne-Nde C, Ebogo-Belobo JT, Mbaga DS, Fatawou Modiyinji A, Njouom R. Systematic review and meta-analysis of the prevalence of common respiratory viruses in children < 2 years with bronchiolitis in the pre-COVID-19 pandemic era. PLoS ONE. 2020;15(11):e0242302. doi: 10.1371/journal.pone.0242302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Walton NA, Poynton MR, Gesteland PH, Maloney C, Staes C, Facelli JC. Predicting the start week of respiratory syncytial virus outbreaks using real time weather variables. BMC Med Inform Decis Mak. 2010;10:68. doi: 10.1186/1472-6947-10-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Murray J, Bottle A, Sharland M, Modi N, Aylin P, Majeed A, et al. Risk factors for hospital admission with RSV bronchiolitis in England: a population-based birth cohort study. PLoS ONE. 2014;9(2):e89186. doi: 10.1371/journal.pone.0089186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sharp A, Minaji M, Panagiotopoulos N, Reeves R, Charlett A, Pebody R. Estimating the burden of adult hospital admissions due to RSV and other respiratory pathogens in England. Influenza Other Respir Viruses. 2022;16(1):125–131. doi: 10.1111/irv.12910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.van Asten L, van den Wijngaard C, van Pelt W, van de Kassteele J, Meijer A, van der Hoek W, et al. Mortality attributable to 9 common infections: significant effect of influenza A, respiratory syncytial virus, influenza B, norovirus, and parainfluenza in elderly persons. J Infect Dis. 2012;206(5):628–639. doi: 10.1093/infdis/jis415. [DOI] [PubMed] [Google Scholar]
- 51.Heppe-Montero M, Gil-Prieto R, Walter S, Aleixandre-Blanquer F, De Gil MÁ. Burden of severe bronchiolitis in children up to 2 years of age in Spain from 2012 to 2017. Hum Vaccin Immunother. 2022;18(1):1879. doi: 10.1080/21645515.2021.1883379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.de González DJ, Ochoa SC. Study of variability in the management of acute bronchiolitis in Spain in relation to age of patients. National multicenter study (aBREVIADo project) An Pediatr (Barc) 2010;72(1):4–18. doi: 10.1016/j.anpedi.2009.10.006. [DOI] [PubMed] [Google Scholar]
- 53.Arriola CS, Kim L, Langley G, Anderson EJ, Openo K, Martin AM, et al. Estimated burden of community-onset respiratory syncytial virus-associated hospitalizations among children aged <2 years in the United States, 2014–15. J Pediatric Infect Dis Soc. 2020;9(5):587–595. doi: 10.1093/jpids/piz087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Taylor S, Taylor RJ, Lustig RL, Schuck-Paim C, Haguinet F, Webb DJ, et al. Modelling estimates of the burden of respiratory syncytial virus infection in children in the UK. BMJ Open. 2016;6(6):e009337. doi: 10.1136/bmjopen-2015-009337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Martinón-Torres F, Carmo M, Platero L, Drago G, López-Belmonte JL, Bangert M, et al. Clinical and economic hospital burden of acute respiratory infection (BARI) due to respiratory syncytial virus in Spanish children, 2015–2018. BMC Infect Dis. 2023;23(1):385. doi: 10.1186/s12879-023-08358-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wildenbeest JG, Billard MN, Zuurbier RP, Korsten K, Langedijk AC, van de Ven PM, et al. The burden of respiratory syncytial virus in healthy term-born infants in Europe: a prospective birth cohort study. Lancet Respir Med. 2023;11(4):341–353. doi: 10.1016/S2213-2600(22)00414-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kwong JC, Schwartz KL, Campitelli MA, Chung H, Crowcroft NS, Karnauchow T, et al. Acute myocardial infarction after laboratory-confirmed influenza infection. N Engl J Med. 2018;378(4):345–353. doi: 10.1056/NEJMoa1702090. [DOI] [PubMed] [Google Scholar]
- 58.Musher DM, Abers MS, Corrales-Medina VF. Acute infection and myocardial infarction. N Engl J Med. 2019;380(2):171–176. doi: 10.1056/NEJMra1808137. [DOI] [PubMed] [Google Scholar]
- 59.Kindermann I, Barth C, Mahfoud F, Ukena C, Lenski M, Yilmaz A, et al. Update on myocarditis. J Am Coll Cardiol. 2012;59(9):779–792. doi: 10.1016/j.jacc.2011.09.074. [DOI] [PubMed] [Google Scholar]
- 60.World Health Organization. Life expectancy at birth, 2023. Available from: https://www.who.int/data/gho/data/indicators/indicator-details/GHO/life-expectancy-at-birth-(years.
- 61.Hansen CL, Chaves SS, Demont C, Viboud C. Mortality associated with influenza and respiratory syncytial virus in the US, 1999–2018. JAMA Netw Open. 2022;5(2):e220527. doi: 10.1001/jamanetworkopen.2022.0527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zambon MC, Stockton JD, Clewley JP, Fleming DM. Contribution of influenza and respiratory syncytial virus to community cases of influenza-like illness: an observational study. Lancet. 2001;358(9291):1410–1416. doi: 10.1016/S0140-6736(01)06528-X. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets analyzed during the current study are available, upon request, from the Ministry of Health and National Statistical Office repositories. Registro de Actividad de Atención Especializada RAE-CMBD 2022 Ministerio de Sanidad; 2022 [Available from: https://www.sanidad.gob.es/estadEstudios/estadisticas/cmbdhome.htm]. Instituto Nacional de Estadística. Base de datos de mortalidad (Demografía y población/Fenómenos demográficos/Tablas de mortalidad); 2022 [Available from: https://www.ine.es].


