Abstract
Objectives
Among primary prevention-type adults not on lipid-lowering therapy, conflicting results exist on the relationship between low-density lipoprotein cholesterol (LDL-C) and long-term mortality. We evaluated this relationship in a real-world evidence population of adults.
Design
Retrospective cohort study.
Setting
Electronic medical record data for adults, from 4 January 2000 through 31 December 2022, were extracted from the University of Pittsburgh Medical Center healthcare system.
Participants
Adults without diabetes aged 50–89 years not on statin therapy at baseline or within 1 year and classified as primary prevention-type patients. To mitigate potential reverse causation, patients who died within 1 year or had baseline total cholesterol (T-C) ≤120 mg/dL or LDL-C <30 mg/dL were excluded.
Main exposure measure
Baseline LDL-C categories of 30–79, 80–99, 100–129, 130–159, 160–189 or ≥190 mg/dL.
Main outcome measure
All-cause mortality with follow-up starting 365 days after baseline cholesterol measurement.
Results
177 860 patients with a mean (SD) age of 61.1 (8.8) years and mean (SD) LDL-C of 119 (31) mg/dL were evaluated over a mean of 6.1 years of follow-up. A U-shaped relationship was observed between the six LDL-C categories and mortality with crude 10-year mortality rates of 19.8%, 14.7%, 11.7%, 10.7%, 10.1% and 14.0%, respectively. Adjusted mortality HRs as compared with the referent group of LDL-C 80–99 mg/dL were: 30–79 mg/dL (HR 1.23, 95% CI 1.17 to 1.30), 100–129 mg/dL (0.87, 0.83–0.91), 130–159 mg/dL (0.88, 0.84–0.93), 160–189 mg/dL (0.91, 0.84–0.98) and ≥190 mg/dL (1.19, 1.06–1.34), respectively. Unlike LDL-C, both T-C/HDL cholesterol (high-density lipoprotein cholesterol) and triglycerides/HDL cholesterol ratios were independently associated with long-term mortality.
Conclusions
Among primary prevention-type patients aged 50–89 years without diabetes and not on statin therapy, the lowest risk for long-term mortality appears to exist in the wide LDL-C range of 100–189 mg/dL, which is much higher than current recommendations. For counselling these patients, minimal consideration should be given to LDL-C concentration.
Keywords: epidemiology, primary prevention, adult cardiology
STRENGTHS AND LIMITATIONS OF THIS STUDY.
The cohort consisted of a large, ‘real-world’ sample of adults across a large health system with long-term follow-up and sufficient precision for subgroup analyses.
The study design mitigated potential for reverse causation of mortality by excluding patients who died within 1 year of baseline cholesterol measurement or had exceptionally low total or low-density lipoprotein cholesterol levels at baseline.
The analysis was limited to all-cause mortality and thus was unable to assess cause-specific mortality.
Introduction
Heart disease (HD), which includes atherosclerotic cardiovascular disease (ASCVD) as its primary component, is the leading cause of death in the USA.1 2 A near-universal but not absolute belief3 is that high total cholesterol (T-C), low-density lipoprotein cholesterol (LDL-C) in particular (the so-called ‘bad’ cholesterol), is a root cause of ASCVD,4 and that ‘lower is better’ with a suggested optimal LDL-C level at or below 100 mg/dL.5 6 In this regard, the American College of Cardiology (ACC) unequivocally implicates elevated LDL-C as a de facto cause of ASCVD (and hence mortality) by stating that lowering of LDL-C with moderate intensity generic statins allows for efficacious and cost-effective primary prevention for those patients with an estimated 10-year risk of ASCVD ≥7.5%.7 Risk of ASCVD is often estimated using the online ACC-ASCVD Risk Estimator,8 and as seen in online supplemental table 1, all males ages 59 and older even in the presence of ‘normal’ ASCVD risk factors (lipids included) may be classified at intermediate or high risk of ASCVD, and thus candidates for LDL-C lowering therapy.
bmjopen-2023-077949supp003.pdf (76.5KB, pdf)
bmjopen-2023-077949supp002.pdf (101.8KB, pdf)
bmjopen-2023-077949supp001.pdf (153.9KB, pdf)
The overall belief that ‘lower LDL-C is better’ for primary prevention of ASCVD is supported by the 25.5% estimated prevalence of use of statins in this setting for adults aged 40–75 years.9 Despite the generally accepted belief that ‘lower LDL-C is better,’ meta-analyses indicate that high LDL-C is associated with at most a small increased absolute risk of ASCVD or premature mortality. First, in brief, in an extensive recent meta-analysis published in 2023 of 60 randomised controlled trials that compared either placebo, usual care or less-intensive therapy to active or more potent lipid-lowering therapy, the number needed to treat to reduce one death with active or more potent lipid-lowering therapy was exceptionally high at 754 persons. Moreover, there was no relationship between LDL-C per cent lowering and risk of cardiovascular mortality.10 Similarly, whereas an earlier meta-analysis published in 2010 indicated that both use and dose of statin therapy reduced the relative risk of major vascular events and all-cause mortality, absolute risk reductions were very small (eg, 0.2% absolute risk reduction in all-cause mortality per 1.0 mmol/L reduction in LDL-C).11 In the context of lipid-lowering therapy, these findings call into question the prevailing belief that ‘lower LDL-C is better’ at least in terms of any appreciable clinical benefit.
Second, acute coronary syndromes (ACSs) routinely occur in patients with ‘normal’ LDL-C. For example, in a large cohort of 1 36 905 patients hospitalised with coronary artery disease (CAD) (79% attributed to ACS), of whom, 21% were on lipid-lowering therapy at admission, less than one-quarter had an admission LDL-C >130 mg/dL.12 In addition, women are generally considered to be at overall lower risk of coronary HD mortality than men,13 yet tend to have higher T-C and LDL-C,14 which is counterintuitive to higher LDL-C being associated with ASCVD and premature mortality.
Third, the field of life insurance medicine, which focuses principally on predicting mortality hazards,15 arguably conducts the most robust actuarial analyses of life expectancy. Notably, in this field, the T-C/HDL-C (high-density lipoprotein cholesterol) ratio has been shown to be the best single measure of all-cause mortality risk among various lipid tests, including LDL-C.16 This is further supported by examination of selected life insurance underwriting guidelines (obtained publicly and summarised) from a large US insurance company.17 As seen in online supplemental table 2, T-C and HDL-C are used jointly in policy underwriting, whereas LDL-C is not used, and lipid-lowering therapy is not emphasised. Moreover, notwithstanding other important patient factors (blood pressure, smoking), online supplemental table 2 shows that a person 70 years of age or older can potentially qualify for a ‘preferred-plus’ life insurance policy having a T-C value as high as 300 mg/dL so long as the T-C/HDL-C ratio is 5.0 or lower (ie, HDL-C ≥60 mg/dL). This aligns with meta-analyses/systematic reviews that report HDL-C to be inversely associated with all-cause and cardiovascular disease (CVD) mortality risks.18 19
The above-described examples of conflicting beliefs and findings, along with general propensity for health professionals to prescribe LDL-C lowering therapies for primary prevention based in part through routine risk assessment with the ACC-ASCVD Risk Estimator, call for a critical appraisal and analysis of the relationship between LDL-C and long-term risk of mortality in adults. Therefore, within a large, ‘real-world’ healthcare system, we evaluated the association between LDL-C and all-cause long-term mortality among primary prevention-type adults without diabetes aged 50–89 years. The analysis did not focus on the use of statin therapy for primary prevention.
Methods
We conducted a retrospective cohort study of adults aged 50–89 years with hospital and/or office visit data captured through the University of Pittsburgh Medical Center (UPMC) electronic medical record (EMR) system. The date period for analysis was 4 January 2000 through 31 December 2022. Conduct and dissemination of results from this observational study were performed in accordance with the STROBE (STrengthening the Reporting of OBservational studies in Epidemiology) statement.
Data sources
Health-related data captured in the UPMC EMR and its ancillary clinical systems were aggregated and harmonised in a clinical data warehouse, as previously described.20 21 For all patients, we accessed sociodemographic data, medical history and billing charges for all outpatient and inpatient encounters with diagnoses and procedures coded based on the International Classification of Diseases, 9th and 10th Revisions.22 23 Deaths were identified using hospital discharge dispositions of ‘ceased to breathe’ sourced from the inpatient medical record system; deaths after discharge were identified externally via the Death Master File from the Social Security Administration’s National Technical Information Service.24 Cause of death was unavailable for analysis. In secondary analyses, a composite outcome of ASCVD was ascertained from UPMC hospital admission/discharge records, defined as the occurrence of myocardial infarction, stroke, percutaneous coronary intervention, coronary artery bypass graft surgery or peripheral vascular disease.
Eligibility criteria
The index date for selection and analysis of patients aged 50–89 years was the first date of cholesterol measurement performed whether through hospitalisation or in conjunction with an office visit (online supplemental figure 1). For analysis, we required non-missing laboratory values for T-C, LDL-C and HDL-C. The patient population was restricted to ‘primary prevention’ patients, defined as no history of diabetes, CAD, carotid artery disease, peripheral vascular disease, cardiac arrest, haemorrhagic or ischaemic stroke or transient ischaemic attack. Other eligibility criteria included: self-reported race of either white or black (due to very low prevalence of other races), and not on statin therapy at baseline or within 1 year of follow-up. In addition, to help offset potential bias due to reverse causation (ie, very low cholesterol being a marker for malnutrition and overall poor health), we excluded patients who died within 1 year of the baseline cholesterol measurement, as well as those with baseline T-C and/or LDL-C values of ≤120 or <30 mg/dL, respectively.
bmjopen-2023-077949supp004.pdf (81.3KB, pdf)
Classification of lipid levels
From the baseline measurement, we classified patients into mutually exclusive lipid-level categories using common clinical thresholds25 including LDL-C (30–79, 80–99, 100–129, 130–159, 160–189 or 190 mg/dL or higher) and T-C (121–160, 161–200, 201–240, 241–280 or 281 mg/dL or higher). In supplemental analyses, we classified the T-C/HDL-C ratio as ≤3.0, >3.0–4.0, >4.0–5.0, >5.0–6.0 or >6.0, and triglycerides/HDL-C ratio into quintiles. Again, to potentially mitigate potential bias due to reverse causation, we selected the LDL-C category of 80–99 mg/dL as the referent group, rather than the lowest LDL-C group (30–79 mg/dL).
Outcome measures
The main outcome measure was all-cause mortality with the number of days and years of follow-up calculated starting 365 days after the baseline cholesterol measurement. For patients who did not die, their length of follow-up was calculated starting 365 days after the baseline cholesterol measurement and until their last record in the EMR system. In secondary analyses, the composite outcome of occurrence of ASCVD was evaluated.
Statistical analysis
For patients within the respective study-defined baseline LDL-C categories, median and IQR for continuous variables and counts and percentages for categorical variables are presented. For each LDL-C category, the Kaplan-Meier method was used to calculate cumulative mortality rates at 1-, 5- and 10-year follow-up, with survival curves plotted at 6-month intervals out to 12 years. Patients who did not die were censored at last date of follow-up. Cox regression was used to estimate HRs (and corresponding 95% CIs) of mortality over the full follow-up period by baseline LDL-C. A crude model was first fit followed by an adjusted model that included covariates selected by a forward stepwise approach using an entry p value of <0.01 and initiation of statin use any time after 1 year of follow-up. Separate estimates for the relationship between initiation of statin use and mortality are not presented due to expected immortal time bias (ie, requirement to be alive during follow-up to initiate statin use). Secondary analyses of lipid parameters used the same methods as for LDL-C and included categories of the T-C/HDL-C and triglycerides/HDL-C ratios.
In addition to the clinical categories used to define and evaluate baseline lipid levels, in secondary analyses, each lipid parameter was evaluated in relation to mortality risk by use of non-parametric generalised additive models using smoothing splines adjusting for the same covariates used in the Cox regression models. The smoothing parameters including the number of df were optimised by use of generalised cross-validation.
We used SAS, V.9.4 (SAS Institute) for all analyses.
Subgroup analyses
Subgroup analyses for estimation of the relationship between LDL-C category and mortality included age (50–69, 70–89), sex (female, male) and baseline ASCVD risk classification (low/borderline, intermediate, high, risk not determined).
Patient and public involvement
None.
Results
The mean (SD) LDL-C was 119 (31) mg/dL, and the prevalence of patients within the six LDL-C categories was as follows: 30–79 (9.1%), 80–99 (18.3%), 100–129 (39.1%), 130–159 (24.4%), 160–189 (7.1%) or 190 mg/dL or higher (2.0%) (table 1). The median age of patients was 59 years and mean age ranged nominally across the six LDL-C categories from 60.7 to 61.7 years. There was a general indication of overall higher baseline risk in the group of patients with LDL-C from 30 to 79 mg/dL (table 1) (consistent with the stated concern of potential reverse causation). This included a numerically higher prevalence of current smokers and those with a history of various comorbidities (eg, atrial fibrillation, arrhythmia, congestive heart failure, chronic obstructive pulmonary disease), as well as nominally higher prevalence of selected medication use (eg, ACE inhibitors, beta-blockers, diuretics, opioids, direct oral anticoagulants). History of cancer was slightly higher in the two lowest LDL-C categories, whereas estimated 10-year ASCVD risk was highest in those with baseline LDL-C ≥190 mg/dL.
Table 1.
Baseline characteristics of study population by baseline LDL cholesterol value
| Characteristic | Baseline LDL cholesterol value (mg/dL) | |||||
| 30–79 (n=16 162) |
80–99 (n=32 517) |
100–129 (n=69 399) |
130–159 (n=43 333) |
160–189 (n=12 663) |
190 or higher (n=3586) |
|
| Age (years), median (IQR) | 59 (54–67) | 59 (54–67) | 59 (54–66) | 59 (54–65) | 59 (54–65) | 60 (54–67) |
| Age (years), n (%) | ||||||
| 50–59 | 8167 (50.5) | 16 551 (50.9) | 35 706 (51.5) | 22 811 (52.6) | 6694 (52.9) | 1765 (49.2) |
| 60–69 | 4686 (29.0) | 9742 (30.0) | 21 632 (31.2) | 13 797 (31.8) | 4029 (31.8) | 1162 (32.4) |
| 70–79 | 2221 (13.7) | 4399 (13.5) | 8808 (12.7) | 5103 (11.8) | 1439 (11.4) | 514 (13.3) |
| 80 and older | 1088 (6.7) | 1825 (5.6) | 3253 (4.7) | 1622 (3.7) | 501 (4.0) | 145 (4.0) |
| Sex | ||||||
| Female | 9027 (55.9) | 18 965 (58.3) | 42 697 (61.5) | 28 034 (64.7) | 8654 (68.3) | 2562 (71.4) |
| Male | 7135 (44.1) | 13 552 (41.7) | 26 702 (38.5) | 15 299 (35.3) | 4009 (31.7) | 1024 (28.6) |
| Race | ||||||
| Black | 1700 (10.5) | 2350 (7.2) | 3855 (5.6) | 2076 (4.8) | 607 (4.8) | 208 (5.8) |
| White | 14 462 (89.5) | 30 167 (92.8) | 65 544 (94.4) | 41 257 (95.2) | 12 056 (95.2) | 3378 (94.2) |
| Former smoker, n (%) | 4172 (27.3) | 8270 (26.9) | 16 871 (25.7) | 10 354 (25.3) | 2933 (24.5) | 858 (25.5) |
| Current smoker, n (%) | 3287 (21.5) | 5430 (17.6) | 9822 (15.0) | 6274 (15.3) | 1998 (16.7) | 668 (19.8) |
| Body Mass Index, median (IQR) | 25.8 (25.2–33.2) | 26.3 (25.2–33.8) | 26.6 (25.2–34.0) | 26.9 (25.2–33.9) | 26.9 (25.2–33.6) | 26.7 (25.2–33.1) |
| History of obesity, n (%) | 6011 (37.2) | 12 438 (38.3) | 26 946 (38.8) | 16 949 (39.1) | 4899 (38.7) | 1326 (37.0) |
| History of obstructive sleep apnoea, n (%) | 932 (5.8) | 1831 (5.6) | 3619 (5.2) | 1931 (4.5) | 507 (4.0) | 136 (3.8) |
| History of hypertension, n (%) | 5540 (34.3) | 11 331 (34.8) | 23 634 (34.1) | 13 435 (31.0) | 3621 (28.6) | 1060 (29.6) |
| History of atrial fibrillation, n (%) | 687 (4.3) | 1181 (3.6) | 1930 (2.8) | 845 (2.0) | 214 (1.7) | 60 (1.7) |
| History of arrhythmia, n (%) | 1178 (7.3) | 2254 (6.9) | 4143 (6.0) | 2054 (4.7) | 528 (4.2) | 133 (3.7) |
| History of valvular heart disease, n (%) | 431 (2.7) | 834 (2.6) | 1505 (2.2) | 798 (1.8) | 246 (1.9) | 60 (1.7) |
| History of congestive heart failure, n (%) | 251 (1.6) | 375 (1.2) | 597 (0.9) | 245 (0.6) | 80 (0.6) | 15 (0.4) |
| History of deep vein thrombosis, n (%) | 184 (1.1) | 323 (1.0) | 667 (1.0) | 356 (0.8) | 93 (0.8) | 25 (0.7) |
| History of cancer, n (%) | 1554 (9.6) | 2916 (9.0) | 5597 (8.0) | 3348 (7.7) | 912 (7.2) | 281 (7.8) |
| History of chronic obstructive pulmonary disease, n (%) | 1147 (7.1) | 1783 (5.5) | 3156 (4.5) | 1666 (3.8) | 474 (3.7) | 146 (4.1) |
| History of chronic kidney disease, n (%) | 329 (2.0) | 424 (1.3) | 695 (1.0) | 356 (0.8) | 126 (1.0) | 42 (1.2) |
| History of depression, n (%) | 1985 (12.3) | 3981 (12.2) | 8327 (12.0) | 5214 (12.0) | 1606 (12.7) | 440 (12.3) |
| Systolic BP (mm Hg), median (IQR) | 128 (118–140) | 127 (118–138) | 128 (118–139) | 128 (120–140) | 128 (120–140) | 130 (120–140) |
| Diastolic BP (mm Hg), median (IQR) | 78 (70–84) | 78 (70–84) | 80 (71–84) | 80 (72–84) | 80 (72–84) | 80 (72–86) |
| HDL cholesterol (mg/dL), median (IQR) | 57 (45–73) | 56 (44–70) | 55 (45–68) | 55 (45–66) | 54 (45–65) | 53 (45–64) |
| Total/HDL cholesterol, median (IQR) | 2.5 (2.2–3.0) | 3.0 (2.5–3.6) | 3.5 (3.0–4.2) | 4.0 (3.4–4.8) | 4.6 (4.0–5.5) | 5.5 (4.6–6.5) |
| Triglycerides (mg/dL), median (IQR) | 90 (63–137) | 91 (67–131) | 100 (74–140) | 111 (83–151) | 125 (94–167) | 149 (110–201) |
| Haemoglobin (g/dL), median (IQR) | 13.7 (12.6–14.7) | 13.9 (12.9–14.8) | 14.0 (13.1–14.9) | 14.1 (13.3–15.0) | 14.2 (13.4–15.0) | 14.1 (13.3–15.0) |
| Glucose (mg/dL), median (IQR) | 94 (87–104) | 94 (87–103) | 94 (88–102) | 94 (88–102) | 95 (89–103) | 96 (89–105) |
| ACE inhibitor, n (%) | 2060 (12.7) | 3992 (12.3) | 8024 (11.6) | 4454 (10.3) | 1205 (9.5) | 328 (9.1) |
| Angiotensin receptor blocker, n (%) | 1028 (6.4) | 2017 (6.2) | 3927 (5.7) | 2018 (4.7) | 558 (4.4) | 156 (4.4) |
| Beta blocker, n (%) | 2747 (17.0) | 4827 (14.8) | 8969 (12.9) | 4709 (10.9) | 1352 (10.7) | 430 (12.0) |
| Calcium blocker, n (%) | 1931 (11.9) | 3501 (10.8) | 6612 (9.5) | 3534 (8.2) | 956 (7.5) | 297 (8.3) |
| Diuretic, n (%) | 2662 (16.5) | 4763 (14.6) | 8814 (12.7) | 4717 (10.9) | 1257 (9.9) | 390 (10.9) |
| Antidepressant, n (%) | 3497 (21.6) | 6504 (20.0) | 13 784 (19.9) | 8624 (19.9) | 2628 (20.8) | 797 (22.2) |
| Opioids, n (%) | 3319 (20.5) | 5400 (16.6) | 9688 (14.0) | 5711 (13.2) | 1599 (12.6) | 523 (14.2) |
| Antiplatelet agent, n (%) | 2209 (13.7) | 4319 (13.3) | 9006 (13.0) | 5057 (11.7) | 1267 (10.0) | 402 (11.2) |
| Aspirin, n (%) | 3082 (19.1) | 6087 (18.7) | 12 511 (18.0) | 7117 (16.4) | 1922 (15.2) | 586 (16.3) |
| Direct oral anticoagulant, n (%) | 423 (2.6) | 684 (2.1) | 1086 (1.6) | 479 (1.1) | 133 (1.1) | 33 (0.9) |
| ASCVD 10-year risk, median (IQR) | 5.8 (2.3–12.6) | 5.8 (2.5–12.7) | 5.9 (2.8–12.3) | 6.3 (3.1–12.2) | 6.8 (3.6–13.0) | 8.7 (4.6–15.7) |
| ASCVD 10-year risk, n (%) | ||||||
| Low | 6204 (58.8) | 12 166 (58.3) | 25 457 (58.6) | 15 048 (57.3) | 4144 (54.1) | 900 (43.0) |
| Intermediate | 2887 (27.4) | 5804 (27.8) | 12 514 (28.8) | 8161 (31.1) | 2596 (33.9) | 839 (40.0) |
| High | 1459 (13.8) | 2888 (13.8) | 5472 (12.6) | 3045 (11.6) | 913 (11.9) | 356 (17.0) |
| Started statin use >1 year after baseline measurement, n (%) | 484 (3.0) | 921 (2.8) | 2948 (4.2) | 3448 (8.0) | 1600 (12.6) | 644 (18.0) |
ASCVD, atherosclerotic cardiovascular disease; BP, blood pressure; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Patient follow-up
The mean and median follow-up after excluding the study requirement to have survived at least 1 year after baseline cholesterol measurement was 6.1 and 5.9 years, respectively, and 17% of patients had 10 or more years of follow-up. Across the six LDL-C categories, the mean years of follow-up among patients who did not die ranged from 5.8 to 6.4 years. In total, 48.9%–55.5% of patients had their first LDL-C measurement in calendar year 2015 or earlier, and the percentage of patients with their last follow-up extending into calendar year 2023 ranged from 57.8% to 63.4%, thereby suggesting non-informative censoring.
Overall assessment of mortality
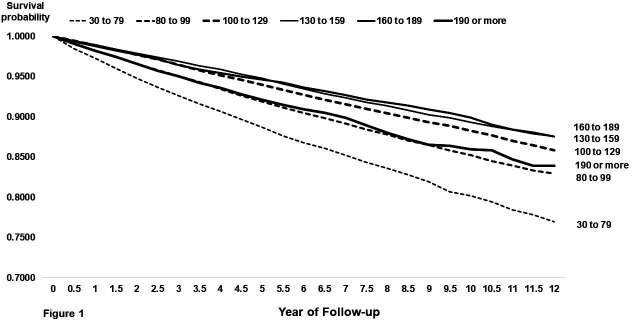
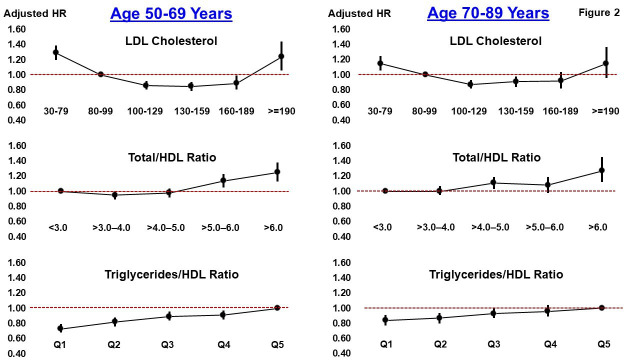
In ascending order from lowest LDL-C category (30–79 mg/dL) to highest LDL-C category (≥190 mg/dL), 10-year cumulative mortality rates were U-shaped at 19.8%, 14.7%, 11.7%, 10.7%, 10.1% and 14.0% (table 2, figures 1 and 2). Adjusted mortality HRs and 95% CIs (table 2), as compared with the referent group of LDL-C 80–99 mg/dL, were as follows: 30–79 mg/dL (1.23, 95% CI 1.17 to 1.30), 100–129 mg/dL (0.87, 95% CI 0.83 to 0.91), 130–159 mg/dL (0.88, 95% CI 0.84 to 0.93), 160–189 mg/dL (0.91, 95% CI 0.84 to 0.98), ≥190 mg/dL (1.19, 95% CI 1.06 to 1.34), respectively. Thus, the three LDL-C categories within the range of 100–189 mg/dL showed similar, slightly lower mortality risk compared with the referent group of LDL-C 80–99 mg/dL. When evaluated as a continuous variable, the relationship between LDL-C and mortality was mostly U-shaped, with the lowest risk of mortality in the range of approximately 110–190 mg/dL (online supplemental figure 2, upper left).
Table 2.
Risks and HRs for death by LDL cholesterol level at baseline
| LDL cholesterol (mg/dL) | n | Cumulative incidence (%) | Total | Crude HR | Adjusted HR | 95% CI | ||
| 1 year | 5 years | 10 years | # deaths | |||||
| 30–79 | 16 162 | 2.7 | 11.3 | 19.8 | 2159 | 1.41 | 1.23 | 1.17 to 1.30 |
| 80–99 | 32 517 | 1.7 | 8.1 | 14.7 | 3232 | 1.0 | 1.0 | — |
| 100–129 | 69 399 | 1.1 | 6.0 | 11.7 | 5415 | 0.77 | 0.87 | 0.83 to 0.91 |
| 130–159 | 43 333 | 1.0 | 5.2 | 10.7 | 2971 | 0.69 | 0.88 | 0.84 to 0.93 |
| 160–189 | 12 663 | 1.2 | 5.4 | 10.1 | 821 | 0.68 | 0.91 | 0.84 to 0.98 |
| 190 or higher | 3586 | 1.8 | 7.9 | 14.0 | 317 | 0.96 | 1.19 | 1.06 to 1.34 |
| Patients aged 50–69 years | ||||||||
| 30–79 | 12 853 | 1.8 | 8.1 | 14.2 | 1241 | 1.52 | 1.20 | 1.20 to 1.39 |
| 80–99 | 26 293 | 1.1 | 5.2 | 9.6 | 1745 | 1.0 | 1.0 | — |
| 100–129 | 57 338 | 0.7 | 3.9 | 7.6 | 2924 | 0.76 | 0.86 | 0.81 to 0.92 |
| 130–159 | 36 608 | 0.7 | 3.4 | 6.9 | 1653 | 0.69 | 0.85 | 0.79 to 0.91 |
| 160–189 | 10 723 | 0.9 | 3.7 | 6.5 | 472 | 0.70 | 0.89 | 0.81 to 0.99 |
| 190 or higher | 2927 | 1.2 | 5.7 | 9.4 | 181 | 1.01 | 1.24 | 1.06 to 1.44 |
| Patients aged 70–89 years | ||||||||
| 30–79 | 3309 | 6.3 | 24.3 | 42.7 | 918 | 1.25 | 1.15 | 1.06 to 1.25 |
| 80–99 | 6224 | 4.5 | 20.5 | 37.2 | 1487 | 1.0 | 1.0 | — |
| 100–129 | 12 061 | 2.7 | 16.0 | 31.4 | 2491 | 0.80 | 0.87 | 0.82 to 0.93 |
| 130–159 | 6725 | 2.8 | 15.3 | 30.8 | 1318 | 0.76 | 0.91 | 0.84 to 0.98 |
| 160–189 | 1940 | 2.9 | 15.0 | 29.7 | 349 | 0.75 | 0.92 | 0.82 to 1.04 |
| 190 or higher | 659 | 4.5 | 17.5 | 34.2 | 136 | 0.90 | 1.15 | 0.96 to 1.37 |
Model adjusted for age, race, sex, BMI, current smoker, former smoker, history of the following in the past year: hypertension, atrial fibrillation, arrhythmia, congestive heart failure, cancer, chronic obstructive pulmonary disease, chronic kidney disease, baseline systolic and diastolic blood pressure, glucose and the following medications in the past year: ACE inhibitors, beta-blockers, calcium blockers, any SBP lowering medication, diuretics, aspirin, DOACs, antidepressants, opioids and statin initiation >1 year after baseline cholesterol measurement.
BMI, Body Mass Index; DOAC, direct oral anticoagulant; LDL, low-density lipoprotein; SBP, systolic blood pressure.
Figure 1.
Plot of cumulative mortality rates in 6-month intervals over 12 years of follow-up by baseline low-density lipoprotein cholesterol (LDL-C) category. Dashed lines depict the three lowest LDL-C categories (30–79, 80–99 and 100–129 mg/dL) and solid lines depict the highest LDL-C categories (130–159, 160–189 and ≥190 mg/dL).
Figure 2.
Plot of mortality HRs (filled circles) and 95% CIs (vertical lines) across categories of LDL cholesterol (top), total cholesterol to HDL cholesterol ratio (middle) and triglycerides to HDL cholesterol ratio (bottom). The left side of the graph is for patients aged 50–69 years; the right side is for patients aged 70–89 years. The dashed line reflects the referent group null value (1.0) for the HR. Q: quintile. Each model is adjusted for age, race, sex, BMI, current smoker, former smoker and history of the following in the past year: hypertension, atrial fibrillation, arrhythmia, congestive heart failure, cancer, chronic obstructive pulmonary disease, chronic kidney disease, baseline systolic and diastolic blood pressure, glucose and the following medications in the past year: ACE inhibitors, beta-blockers, calcium blockers, any SBP lowering medication, diuretics, aspirin, DOACs, antidepressants, opioids and statin initiation >1 year after baseline cholesterol measurement. BMI, Body Mass Index; DOAC, direct oral anticoagulant; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SBP, systolic blood pressure.
Assessment of ASCVD
In ascending order from lowest LDL-C category (30–79 mg/dL) to highest LDL-C category (≥190 mg/dL), 10-year cumulative rates of ASCVD were U-shaped at 6.5%, 5.3%, 4.7%, 4.8%, 5.1% and 7.6% (table 3, top half). Adjusted HRs of risk of ASCVD as compared with the referent group of LDL-C 80–99 mg/dL were as follows: 30–79 mg/dL (1.10, 95% CI 1.00 to 1.20), 100–129 mg/dL (0.94, 95% CI 0.88 to 1.00), 130–159 mg/dL (0.96, 95% CI 0.89 to 1.03), 160–189 mg/dL (0.98, 95% CI 0.88 to 1.08), ≥190 mg/dL (1.23, 95% CI 1.06 to 1.43), respectively. Thus, the three LDL-C categories within the range of 100–189 mg/dL showed similar yet nominally lower risk of ASCVD compared with the referent group of LDL-C 80–99 mg/dL. Similar results were observed for the composite outcome of ASCVD/mortality (table 3, bottom half). Baseline ASCVD risk categories of low, medium and high risk were strongly associated with 10-year rates of ASCVD (1.9%, 4.9% and 9.8%, respectively).
Table 3.
Risks and HRs for ASCVD and ASCVD/mortality by LDL cholesterol levels at baseline
| LDL cholesterol (mg/dL) | n | Cumulative incidence (%) | Total | Crude HR | Adj. HR model | 95% CI | ||
| 1 year | 5 years | 10 years | # events | |||||
| ASCVD | ||||||||
| 30–79 | 16 162 | 0.8 | 3.9 | 6.5 | 816 | 1.25 | 1.10 | 1.00 to 1.20 |
| 80–99 | 32 517 | 0.5 | 2.8 | 5.3 | 1341 | 1.0 | 1.0 | — |
| 100–129 | 69 399 | 0.6 | 2.5 | 4.7 | 2509 | 0.87 | 0.94 | 0.88 to 1.00 |
| 130–159 | 43 333 | 0.5 | 2.4 | 4.8 | 1586 | 0.89 | 0.96 | 0.89 to 1.03 |
| 160–189 | 12 663 | 0.5 | 2.7 | 5.1 | 490 | 0.98 | 0.98 | 0.88 to 1.08 |
| 190 or higher | 3586 | 0.9 | 4.7 | 7.6 | 205 | 1.50 | 1.23 | 1.06 to 1.43 |
| ASCVD/mortality | ||||||||
| 30–79 | 16 162 | 1.0 | 11.8 | 21.4 | 2590 | 1.36 | 1.19 | 1.14 to 1.26 |
| 80–99 | 32 517 | 0.8 | 8.4 | 16.5 | 4014 | 1.0 | 1.0 | — |
| 100–129 | 69 399 | 0.6 | 6.4 | 13.5 | 6952 | 0.79 | 0.89 | 0.85 to 0.92 |
| 130–159 | 43 333 | 0.5 | 5.8 | 12.8 | 4005 | 0.74 | 0.90 | 0.86 to 0.94 |
| 160–189 | 12 663 | 0.5 | 6.5 | 12.6 | 1160 | 0.77 | 0.93 | 0.87 to 0.99 |
| 190 or higher | 3586 | 0.9 | 9.9 | 18.5 | 452 | 1.12 | 1.20 | 1.08 to 1.32 |
Model: Adjusted for age, race, BMI, current smoker, former smoker, history of the following in the past year: hypertension, atrial fibrillation, arrhythmia, congestive heart failure, cancer, chronic obstructive pulmonary disease, chronic kidney disease, baseline systolic and diastolic blood pressure, glucose and the following medications in the past year: ACE inhibitors, beta-blockers, calcium blockers, any SBP lowering medication, diuretics, aspirin, DOACs, antidepressants, opioids and statin initiation >1 year after baseline cholesterol measurement.
ASCVD, atherosclerotic cardiovascular disease; BMI, Body Mass Index; DOAC, direct oral anticoagulant; LDL, low-density lipoprotein; SBP, systolic blood pressure.
Subgroup analyses
For the two different age groups, the three LDL-C categories within the range of 100–189 mg/dL showed relatively similar and slightly lower mortality risk compared with the referent group of LDL-C 80–99 mg/dL (table 2, figure 2). In a similar manner for both females and males, the three LDL-C categories within the range of 100–189 mg/dL showed relatively similar and slightly lower mortality risk compared with the referent group of LDL-C 80–99 mg/dL (online supplemental table 3). Males with LDL-C ≥190 mg/dL did not have a significantly higher risk of mortality than those with LDL-C 80–99 mg/dL (adjusted HR=1.06, 95% CI 0.85 to 1.32). When stratified by 10-year ASCVD risk score, again, the three LDL-C categories within the range of 100–189 mg/dL showed relatively similar and statistically lower mortality risk compared with the referent group of LDL-C 80–99 mg/dL (online supplemental table 4).
Secondary lipid measures
Patients with a T-C/HDL-C ratio >6.0 had a significantly higher risk of mortality than those with a T-C/HDL-C ratio ≤3.0 (adjusted HR=1.28, 95% CI 1.18 to 1.38, online supplemental table 5), with similar results by age (figure 2). For the three T-C/HDL-C ratio categories ≤3.0, >3.0–4.0 and >4.0–5.0, risk of mortality was similar. The triglycerides/HDL-C ratio showed the most consistent evidence of a gradient relationship with mortality with lower values (quintiles) progressively conferring lower risk of mortality (online supplemental table 6) and similar results by age (figure 2). Compared with patients in the highest quintile of triglycerides/HDL-C ratio (value of ≥3.44), those in the lowest quintile (value of ≤1.06) had an estimated 24% lower risk of mortality (adjusted HR=0.76, 95% CI 0.72 to 0.81). Thus, in aggregate and irrespective of age, the secondary lipid measures of T-C/HDL-C ratio and triglycerides/HDL-C ratio appeared to be more predictive of mortality than LDL-C, and a triglycerides/HDL-C ratio of about 1 or lower appears to be optimal.
When evaluated as continuous variables, the relationship between T-C and adjusted risk of mortality was mostly U-shaped (similar to LDL-C), whereas other lipid/mortality relationships presented in a mostly gradient manner (online supplemental figure 2). Specifically, lower HDL-C generally indicated higher adjusted risk of mortality, whereas higher triglycerides, total to HDL-C ratio and triglycerides to HDL-C ratio indicated higher adjusted risk of mortality.
Evaluation of potential reverse causation
By study design, the 2494 patient deaths that occurred from baseline LDL-C measurement to 365 days were excluded from the primary analysis. Among these excluded patients, the percentage of deaths distributed by LDL-C (mg/dL) category was: 30–79 (30.4%), 80–99 (20.1%), 100–129 (26.5%), 130–159 (14.6%), 160–189 (5.9%), 190 or higher (2.5%). The 30.4% of deaths in the 30–79 mg/dL category is much higher than the 9.1% prevalence of patients in the 30–79 mg/dL category (see table 1) observed in the primary analysis. Similarly, 14.1% of deaths excluded in the first year had a total cholesterol value of 40–120 mg/dL compared with 1.6% prevalence of patients in the primary analysis. These results validated the need to remove the influence of potential reverse causality and early deaths and patients with very low baseline cholesterol values from the analysis.
Discussion
In this analysis among primary prevention-type patients without diabetes aged 50–89 years not on statin therapy at baseline or within 1 year, we found no evidence of a gradient relationship between LDL-C and long-term mortality risk. Instead, we observed that within the entire LDL-C range of 100–189 mg/dL (about two-thirds of the total patient population), mortality risk was similar and slightly lower than the referent LDL-C category of 80–99 mg/dL. These data conflict with the prevailing belief that ‘lower LDL-C is better’5 6 yet align with results from multiple studies. A large general population study of adults from Denmark showed a U-shaped relationship between LDL-C and long-term mortality, with lowest risk of all-cause mortality (among individuals not receiving lipid-lowering treatment) being an LDL-C value of 140 mg/dL.26 Similarly, a large cohort study among Korean adults not on statin therapy showed a U-shaped relationship between LDL-C and CVD mortality, with an optimal LDL-C range of 90–149 mg/dL.27 Moreover, in a 20-year prospective cohort study of adults ages 18 and older derived from the National Health and Nutrition Examination Survey III (NHANES III), the lowest relative risk for all-cause mortality was for LDL-C in the range of 130 to <190 mg/dL.28 Collectively, these results indicate that the ‘optimal’ or ‘normal’ range for LDL-C for primary prevention of mortality among adults is likely wide and considerably higher than the suggested optimal LDL-C level of ≤100 mg/dL.5 6
For multiple reasons, we chose to evaluate a population of primary prevention-type adults without diabetes aged 50–89 years not on statin therapy. First, both the prevalence and potential indication for initiating lipid-lowering therapy are relatively high in this population.9 29 30 Second, prevailing guidelines and philosophy for initiating lipid-lowering therapy for secondary prevention of ASCVD and among persons with diabetes are well entrenched.31–33 Third, consideration of initiating lipid-lowering therapy for primary prevention, particularly among older adults, should be carefully weighed based on empirical data34 35 and potential side effects, including but not limited to muscle pain or weakness36 and increased risk of developing diabetes.37–39
Beyond our principal finding of no indication that ‘lower LDL-C is better,’ other prominent findings were that overall and independent of age, the T-C/HDL-C and triglycerides/HDL-C ratios were predictive of long-term mortality risk, the latter of which presented in a gradient manner. A study derived from NHANES data showed a U-shaped relationship between T-C/HDL-C ratio and risk of all-cause mortality,40 whereas results from our analysis were unidirectional with elevated risk of mortality evident among adults with a T-C/HDL-C ratio more than 5.0. Similar to our results, a large study among Korean adults showed a gradient relationship between triglycerides/HDL-C ratio and risk of ischaemic HD.41 Importantly, the triglyceride/HDL-C ratio has recently been reported to be a stronger predictor of 10-year development of type 2 diabetes (strongly associated with mortality risk) than LDL-C, HDL-C or triglycerides alone.42
The importance of high HDL-C alone, or in conjunction with other lipids, has been extensively recognised. In brief, oxidative stress and inflammation are integral in the pathophysiology of atherosclerosis and cardiovascular disease.43 Importantly, HDL-C exerts several physiological roles, prevents oxidation of LDL and inhibits expression of proinflammatory cytokines by macrophages, as well as expression of adhesion molecules by endothelial cells,44–46 and it is inversely associated with both all-cause and CVD mortality risks.18 19 Moreover, it is likely not coincidental nor trivial that the field of life insurance medicine recognises and prioritises the importance of HDL-C over LDL-C in determining underwriting classifications.16 17 47 Unfortunately, from a public health perspective, a meta-analysis of 31 randomised controlled trials on the use of HDL-C modifying treatments showed little to no effect on cardiovascular and all-cause mortality.48
There is an overall lack of consensus on the magnitude and statistical and clinical interpretation of the reduction in mortality risk potentially achieved with the use of LDL-C lowering therapies. Multiple reviews suggest that absolute mortality risk reductions from treatment with statins are small as compared with the more frequent reporting and emphasis on relative risk reductions.49–52 Moreover, mortality reductions with recent use of proprotein convertase subtilisin/kexin type 9 (PCSK-9) inhibitors to lower LDL-C have been mixed and of low absolute risk.53 54 Our postulate from both this review10 and empirical analysis is that whatever small absolute reductions in mortality risk may occur with use of LDL-C lowering therapies, they are most likely not causally related to LDL-C lowering, but potentially to more broad pleiotropic effects. For example, statin use has been shown to reduce inflammatory markers,55 reduce vascular endothelial growth factor concentrations,56 reduce platelet activity57 and increase nitric oxide bioavailability and stabilise atherosclerotic plaques.58 These potential mechanisms of statins, rather than concomitant lowering of LDL-C, per se, may be expected to result in some reduction of ASCVD events.
Arguably, it is irrelevant to patients as to the exact mechanism(s) by which use of statins and other lipid-lowering therapies may result in small absolute reductions in mortality risk. Rather than focusing on LDL-C level, per se, we submit that health professionals should promote established (causal) mechanisms that reduce future risk of major ASCVD events, including weight, blood pressure and blood sugar control, physical activity, avoidance of smoking and stress reduction. Similarly, our results suggest that adult patients without diabetes counselled for primary prevention of ASCVD be apprised of their estimated future risk of ASCVD with minimal consideration of their LDL-C concentration and more consideration of the T-C/HDL and triglyceride/HDL-C ratios along with other known causes of ASCVD (eg, smoking, physical inactivity). Moreover, use of coronary artery calcium scoring in primary prevention is supported by a wealth of data showing that it substantially improves risk prediction including when combined with traditional risk factors and scores.59–61
Limitations
Our study has limitations. First, we were unable to assess cause-specific mortality which would have provided additional insight into the relationship between LDL-C and CVD mortality. Similarly, our assessment of risk of ASCVD in relation to baseline LDL-C levels is based on ascertainment of events within UPMC hospitals and not external facilities—there is certainly some unknown level of ascertainment of ASCVD events. Second, we chose the index date for follow-up mortality assessment to begin 1 year after baseline cholesterol measurement to ideally minimise potential bias due to reverse causation (ie, low LDL-C being an overall marker of malnutrition and poor health). However, low LDL-C has been frequently reported in patients with cancer25 62 63 and many cancers have a viral etiological component64 and with potentially long latency. Theoretically, some patients with the lowest LDL-C values in our analysis may have been in the early stages of cancer development and hence at elevated long-term mortality risk. This is why we chose LDL-C 80–99 mg/dL as the referent group (rather than 30–79 mg/dL), and the observation that mortality risk was similar across a wide range of LDL-C values (100–189 mg/dL) argues against appreciable bias due to reverse causation. Third, absence of statin use at baseline and within the first year of the study (inclusion criterion) was based on patient-reported data in the EMR and not from prescription data—this leaves open the possibility for some misclassification. In addition, the study requirement for absence of statin use at baseline or within 1 year may have resulted in a patient population generally less likely to initiate lipid-lowering therapy in long-term. Lastly, we cannot rule out potential residual confounding despite statistical adjustment for a large set of covariates associated with mortality.
Conclusions
Our analysis indicates that among primary prevention-type patients without diabetes aged 50–89 years and not on statin therapy, the lowest risk for long-term mortality exists in the wide LDL-C range of 100–189 mg/dL which is much higher than current recommendations. Our analysis also shows that lower T-C/HDL-C and triglycerides/HDL-C ratios are independently associated with lower mortality risk, whereas LDL-C appears to be of limited to no predictive value. Collectively, these observations suggest that adult patients without diabetes counselled for primary prevention of ASCVD be apprised of their estimated future risk of ASCVD with minimal consideration of their LDL-C concentration and more consideration of the T-C/HDL and triglycerides/HDL-C ratios along with other established causes of ASCVD (eg, high blood pressure, smoking, physical inactivity) and potentially coronary artery calcium scoring.
Supplementary Material
Footnotes
Contributors: KEK: Conception, statistical analysis, writing and editing. DD: Conception, critical review and editing. SM: Critical review and editing. OCM: Conception, critical review and editing. K.E.K serves as guarantor and accepts full responsibility for the work and/or the conduct of the study, had access to the data, and controlled the decision to publish.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. Study protocol: No separate study protocol was required a priori, as this retrospective analysis was deemed a quality improvement initiative with ethical review and approval granted by the UPMC Quality Improvement Review Committee and Institutional Review Board. Statistical code: Selected statistical code may be requested from Dr Kevin Kip (e-mail, kipke2@upmc.edu). Data set: The data set contains protected health information and will not be available.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The Quality Improvement Review Committee and Institutional Review Board at the University of Pittsburgh Medical Center provided ethical review and approval of the study as an exempt protocol (Project ID: 4565), and all data remained deidentified for this analysis.
References
- 1. Heron M. Deaths: leading causes for 2019. Centers for Disease Control and Prevention. Natl Vital Stat Rep 2021;70:9. Available: https://www.cdc.gov/nchs/data/nvsr/nvsr70/nvsr70-09-508.pdf [PubMed] [Google Scholar]
- 2. US Centers for Disease Control and Prevention . Leading causes of death and injury, Available: https://www.cdc.gov/injury/wisqars/LeadingCauses.html
- 3. Ravnskov U, de Lorgeril M, Diamond DM, et al. LDL-C does not cause cardiovascular disease: a comprehensive review of the current literature. Expert Rev Clin Pharmacol 2018;11:959–70. 10.1080/17512433.2018.1519391 [DOI] [PubMed] [Google Scholar]
- 4. Ference BA, Ginsberg HN, Graham I, et al. Low-density lipoproteins cause Atherosclerotic cardiovascular disease. 1. evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis society consensus panel. Eur Heart J 2017;38:2459–72. 10.1093/eurheartj/ehx144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. American Heart Association . What is cholesterol?, Available: https://www.heart.org/-/media/Files/Health-Topics/Cholesterol/What-is-Cholesterol.pdf
- 6. O’Keefe JH, Cordain L, Harris WH, et al. Optimal low-density lipoprotein is 50 to 70 mg/dl: lower is better and Physiologically normal. J Am Coll Cardiol 2004;43:2142–6. 10.1016/j.jacc.2004.03.046 [DOI] [PubMed] [Google Scholar]
- 7. Grundy SM, Stone NJ, Bailey AL, et al. Guideline on the management of blood cholesterol: A report of the American college of cardiology/American heart Association task force on clinical practice guidelines. Circulation 2019;139:e1082–143. 10.1161/CIR.0000000000000625 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. American College of Cardiology . ASCVD Risk Estimate Plus, Available: https://tools.acc.org/ascvd-risk-estimator-plus/#!/calculate/estimate
- 9. Jacobs JA, Addo DK, Zheutlin AR, et al. Prevalence of Statin use for primary prevention of Atherosclerotic cardiovascular disease by race, Ethnicity, and 10-year disease risk in the US. JAMA Cardiol 2023;8:443. 10.1001/jamacardio.2023.0228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Ennezat PV, Guerbaai RA, Maréchaux S, et al. Extent of low-density lipoprotein cholesterol reduction and all-cause and cardiovascular mortality benefit: A systematic review and meta-analysis. J Cardiovasc Pharmacol 2023;81:35–44. 10.1097/FJC.0000000000001345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Cholesterol Treatment Trialists’ (CTT) Collaboration, Baigent C, Blackwell L, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 2010;376:1670–81. 10.1016/S0140-6736(10)61350-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sachdeva A, Cannon CP, Deedwania PC, et al. Lipid levels in patients hospitalized with coronary artery disease: an analysis of 136,905 hospitalizations in get with the guidelines. Am Heart J 2009;157:111–7. 10.1016/j.ahj.2008.08.010 [DOI] [PubMed] [Google Scholar]
- 13. Bots SH, Peters SAE, Woodward M. Sex differences in coronary heart disease and stroke mortality: a global assessment of the effect of ageing between 1980 and 2010. BMJ Glob Health 2017;2:e000298. 10.1136/bmjgh-2017-000298 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Shohaimi S, Boekholdt MS, Luben R, et al. Distribution of lipid parameters according to different socio-economic Indicators- the EPIC-Norfolk prospective population study. BMC Public Health 2014;14:782. 10.1186/1471-2458-14-782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Kneepkens RF, Lindeboom R. Age dependent decline of relative risks in life insurance medical underwriting. J Insur Med 2014;44:170–83. [PubMed] [Google Scholar]
- 16. Fulks M, Stout RL, Dolan VF. Association of cholesterol, LDL, HDL, cholesterol/HDL and Triglyceride with all-cause mortality in life insurance applicants. J Insur Med 2009;41:244–53. [PubMed] [Google Scholar]
- 17. MetLife . The Condensed Underwriting Guide, 2013. Available: http://www.cassaniinsurance.com/wp-content/uploads/2018/02/Met-Life-condensed_uw_guide.pdf
- 18. Liu L, Han M, Qie R, et al. A dose-response meta-analysis to evaluate the relationship between high-density lipoprotein cholesterol and all-cause and cardiovascular disease mortality. J Endocrinol Invest 2022;45:551–62. 10.1007/s40618-021-01690-6 [DOI] [PubMed] [Google Scholar]
- 19. Ravnskov U, Diamond DM, Hama R, et al. Lack of an association or an inverse association between low-density-lipoprotein cholesterol and mortality in the elderly: a systematic review. BMJ Open 2016;6:e010401. 10.1136/bmjopen-2015-010401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kip KE, McCreary EK, Collins K, et al. Evolving real-world effectiveness of Monoclonal antibodies for treatment of COVID-19: A cohort study. Ann Intern Med 2023;176:496–504. 10.7326/M22-1286 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Reitz KM, Seymour CW, Vates J, et al. Strategies to promote resiliency (SPRY): a randomised embedded Multifactorial Adaptative platform (REMAP) clinical trial protocol to study interventions to improve recovery after surgery in high-risk patients. BMJ Open 2020;10:e037690. 10.1136/bmjopen-2020-037690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Centers for Disease Control and Prevention . n.d. International classification of diseases, ninth revision, clinical modification (ICD-9-CM). Available: www.cdc.gov/nchs/icd/icd9cm.htm
- 23. Centers for Disease Control and Prevention . n.d. International classification of diseases, tenth revision, clinical modification (ICD-10-CM). :www.
- 24. Social Security Administration . Requesting SSA’s Death Information.Available: www.ssa.gov/dataexchange/request_dmf.html [Google Scholar]
- 25. MedlinePlus [Internet]. Bethesda (MD): National Library of Medicine (US). LDL: The “Bad” Cholesterol, Available: https://medlineplus.gov/ldlthebadcholesterol.html
- 26. Johannesen CDL, Langsted A, Mortensen MB, et al. Association between low density lipoprotein and all cause and cause specific mortality in Denmark: prospective cohort study. BMJ 2020;371:m4266. 10.1136/bmj.m4266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Yi SW, An SJ, Park HB, et al. Association between low-density lipoprotein cholesterol and cardiovascular mortality in Statin non-users: a prospective cohort study in 14.9 million Korean adults. Int J Epidemiol 2022;51:1178–89. 10.1093/ije/dyac029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Rong S, Li B, Chen L, et al. Association of low-density lipoprotein cholesterol levels with more than 20-year risk of cardiovascular and all-cause mortality in the general population. J Am Heart Assoc 2022;11:e023690. 10.1161/JAHA.121.023690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Arnett DK, Blumenthal RS, Albert MA, et al. ACC/AHA guideline on the primary prevention of cardiovascular disease: A report of the American college of cardiology/American heart Association task force on clinical practice guidelines. Circulation 2019;140:e596–646. 10.1161/CIR.0000000000000678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lloyd-Jones DM, Braun LT, Ndumele CE, et al. Use of risk assessment tools to guide decision-making in the primary prevention of Atherosclerotic cardiovascular disease: A special report from the American heart Association and American college of cardiology. Circulation 2019;139:e1162–77. 10.1161/CIR.0000000000000638 [DOI] [PubMed] [Google Scholar]
- 31. Banach M, Penson PE. Statins and LDL-C in secondary prevention-so much progress, so far to go. JAMA Netw Open 2020;3:e2025675. 10.1001/jamanetworkopen.2020.25675 [DOI] [PubMed] [Google Scholar]
- 32. Tecson KM, Kluger AY, Cassidy-Bushrow AE, et al. Usefulness of Statins as secondary prevention against recurrent and terminal major adverse cardiovascular events. Am J Cardiol 2022;176:37–42. 10.1016/j.amjcard.2022.04.018 [DOI] [PubMed] [Google Scholar]
- 33. Stone NJ, Robinson JG, Lichtenstein AH, et al. ACC/AHA guideline on the treatment of blood cholesterol to reduce Atherosclerotic cardiovascular risk in adults: a report of the American college of cardiology/American heart Association task force on practice guidelines. J Am Coll Cardiol 2014;63:2889–934. 10.1016/j.jacc.2013.11.002 [DOI] [PubMed] [Google Scholar]
- 34. Gurwitz JH, Go AS, Fortmann SP. Statins for primary prevention in older adults: uncertainty and the need for more evidence. JAMA 2016;316:1971–2. 10.1001/jama.2016.15212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Han BH, Sutin D, Williamson JD, et al. ALLHAT collaborative research group. effect of Statin treatment vs. usual care on primary cardiovascular prevention among older adults: the ALLHAT-LLT randomized clinical trial. JAMA Intern Med 2017;177:955–65. 10.1001/jamainternmed.2017.1442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Cholesterol Treatment Trialists’ Collaboration . Effect of Statin therapy on muscle symptoms: an individual participant data meta-analysis of large-scale, randomised, double-blind trials. Erratum in: lancet 2022; 400:1194. Lancet 2022;400:832–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Mansi IA, Chansard M, Lingvay I, et al. Association of Statin therapy initiation with diabetes progression: A retrospective matched-cohort study. JAMA Intern Med 2021;181:1562–74. 10.1001/jamainternmed.2021.5714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Crandall JP, Mather K, Rajpathak SN, et al. Statin use and risk of developing diabetes: results from the diabetes prevention program. BMJ Open Diab Res Care 2017;5:e000438. 10.1136/bmjdrc-2017-000438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Abbasi F, Lamendola C, Harris CS, et al. Statins are associated with increased insulin resistance and secretion. Arterioscler Thromb Vasc Biol 2021;41:2786–97. 10.1161/ATVBAHA.121.316159 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Zhou D, Liu X, Lo K, et al. The effect of total cholesterol/high-density lipoprotein cholesterol ratio on mortality risk in the general population. Front Endocrinol (Lausanne) 2022;13:1012383. 10.3389/fendo.2022.1012383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Park B, Jung DH, Lee HS, et al. Triglyceride to HDL-cholesterol ratio and the incident risk of ischemic heart disease among Koreans without diabetes: A longitudinal study using national health insurance data. Front Cardiovasc Med 2021;8:716698. 10.3389/fcvm.2021.716698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Yuge H, Okada H, Hamaguchi M, et al. Triglycerides/HDL cholesterol ratio and type 2 diabetes incidence: Panasonic cohort study 10. Cardiovasc Diabetol 2023;22:308. 10.1186/s12933-023-02046-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Ho E, Karimi Galougahi K, Liu CC, et al. Biological markers of oxidative stress: applications to cardiovascular research and practice. Redox Biol 2013;1:483–91. 10.1016/j.redox.2013.07.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mackness B, Mackness M. The antioxidant properties of high-density lipoproteins in Atherosclerosis. Panminerva Med 2010;54:83–90. [PubMed] [Google Scholar]
- 45. Cockerill GW, Rye K-A, Gamble JR, et al. High-density lipoproteins inhibit cytokine-induced expression of endothelial cell adhesion molecules. ATVB 1995;15:1987–94. 10.1161/01.ATV.15.11.1987 [DOI] [PubMed] [Google Scholar]
- 46. Nicholls SJ, Rye KA, Barter PJ. High-density lipoproteins as therapeutic targets. Curr Opin Lipidol 2005;16:345–9. 10.1097/01.mol.0000169356.58513.25 [DOI] [PubMed] [Google Scholar]
- 47. Gleeson R. Atherogenic Dyslipidemia--a medical underwriter’s iceberg. J Insur Med 2009;41:264–9. [PubMed] [Google Scholar]
- 48. Riaz H, Khan SU, Rahman H, et al. Effects of high-density lipoprotein targeting treatments on cardiovascular outcomes: A systematic review and meta-analysis. Eur J Prev Cardiol 2019;26:533–43. 10.1177/2047487318816495 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Byrne P, Demasi M, Jones M, et al. Evaluating the association between low-density lipoprotein cholesterol reduction and relative and absolute effects of Statin treatment: A systematic review and meta-analysis. JAMA Intern Med 2022;182:474–81. 10.1001/jamainternmed.2022.0134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Diamond DM, Leaverton PE. Historical review of the use of relative risk statistics in the portrayal of the purported hazards of high LDL cholesterol and the benefits of lipid-lowering therapy. Cureus 2023;15:e38391. 10.7759/cureus.38391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Ravnskov U, Alabdulgader A, de Lorgeril M, et al. The new European guidelines for prevention of cardiovascular disease are misleading. Expert Rev Clin Pharmacol 2020;13:1289–94. 10.1080/17512433.2020.1841635 [DOI] [PubMed] [Google Scholar]
- 52. Diamond DM, Ravnskov U. How statistical deception created the appearance that Statins are safe and effective in primary and secondary prevention of cardiovascular disease. Expert Rev Clin Pharmacol 2015;8:201–10. 10.1586/17512433.2015.1012494 [DOI] [PubMed] [Google Scholar]
- 53. Dicembrini I, Giannini S, Ragghianti B, et al. Effects of PCSK9 inhibitors on LDL cholesterol, cardiovascular morbidity and all-cause mortality: a systematic review and meta-analysis of randomized controlled trials. J Endocrinol Invest 2019;42:1029–39. 10.1007/s40618-019-01019-4 [DOI] [PubMed] [Google Scholar]
- 54. Steg PG, Szarek M, Bhatt DL, et al. Effect of Alirocumab on mortality after acute coronary syndromes. Circulation 2019;140:103–12. 10.1161/CIRCULATIONAHA.118.038840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Tabrizi R, Tamtaji OR, Mirhosseini N, et al. The effects of Statin use on inflammatory markers among patients with metabolic syndrome and related disorders: A systematic review and meta-analysis of randomized controlled trials. Pharmacol Res 2019;141:85–103. 10.1016/j.phrs.2018.12.010 [DOI] [PubMed] [Google Scholar]
- 56. Sahebkar A, Ponziani MC, Goitre I, et al. Does Statin therapy reduce plasma VEGF levels in humans? A systematic review and meta-analysis of randomized controlled trials. Metabolism 2015;64:1466–76. 10.1016/j.metabol.2015.08.002 [DOI] [PubMed] [Google Scholar]
- 57. Puccetti L, Pasqui AL, Pastorelli M, et al. Time-dependent effect of Statins on platelet function in Hypercholesterolaemia. Eur J Clin Invest 2002;32:901–8. 10.1046/j.1365-2362.2002.01086.x [DOI] [PubMed] [Google Scholar]
- 58. Davignon J. Beneficial cardiovascular pleiotropic effects of Statins. Circulation 2004;109:III39–43. 10.1161/01.CIR.0000131517.20177.5a [DOI] [PubMed] [Google Scholar]
- 59. Blumenthal RS, Cainzos-Achirica M. The Ever-Growing Role of Coronary Artery Calcium in Primary Prevention, Available: https://www.acc.org/Latest-in-Cardiology/Articles/2021/06/21/13/05/The-Ever-Growing-Role-of-CAC-in-Primary-Prevention
- 60. Nasir K, Cainzos-Achirica M. Role of coronary artery calcium score in the primary prevention of cardiovascular disease. BMJ 2021;373:776. 10.1136/bmj.n776 [DOI] [PubMed] [Google Scholar]
- 61. Bittencourt MS, Nasir K, Santos RD, et al. Very high LDL cholesterol: the power of zero passes another test. Atherosclerosis 2020;292:207–8. 10.1016/j.atherosclerosis.2019.11.019 [DOI] [PubMed] [Google Scholar]
- 62. Tanne JH. Meta-analysis says low LDL cholesterol may be associated with greater risk of cancer. BMJ 2007;335:177. 10.1136/bmj.39287.415347.DB [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Benn M, Tybjærg-Hansen A, Stender S, et al. Low-density lipoprotein cholesterol and the risk of cancer: a Mendelian randomization study. J Natl Cancer Inst 2011;103:508–19. 10.1093/jnci/djr008 [DOI] [PubMed] [Google Scholar]
- 64. Plummer M, de Martel C, Vignat J, et al. Global burden of cancers attributable to infections in 2012: a synthetic analysis. Lancet Glob Health 2016;4:e609–16. 10.1016/S2214-109X(16)30143-7 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2023-077949supp003.pdf (76.5KB, pdf)
bmjopen-2023-077949supp002.pdf (101.8KB, pdf)
bmjopen-2023-077949supp001.pdf (153.9KB, pdf)
bmjopen-2023-077949supp004.pdf (81.3KB, pdf)
Data Availability Statement
Data are available upon reasonable request. Study protocol: No separate study protocol was required a priori, as this retrospective analysis was deemed a quality improvement initiative with ethical review and approval granted by the UPMC Quality Improvement Review Committee and Institutional Review Board. Statistical code: Selected statistical code may be requested from Dr Kevin Kip (e-mail, kipke2@upmc.edu). Data set: The data set contains protected health information and will not be available.