Abstract
Purpose:
This study aims to assess the effectiveness and safety of combining the Preserflo™ MicroShunt implant (MicroShunt) with a simultaneous Ologen™ implant in patients with glaucoma.
Methods:
We conducted a retrospective study on consecutive patients with medically uncontrolled glaucoma who underwent MicroShunt + Ologen implantation as a standalone procedure or in combination with phacoemulsification (combined procedure). Success was defined as achieving an intraocular pressure (IOP) of 6–15 mmHg at 18 months post surgery, with a preoperative IOP reduction of at least 20%, and without (complete success) or with (qualified success) the need for antiglaucoma medications. The primary endpoint was the success rate.
Results:
Forty-eight eyes from 47 patients were included, with 28 eyes (58.3%) undergoing the standalone procedure and 20 eyes (41.7%) undergoing the combined procedure. Overall, there was a significant reduction in preoperative IOP from 19.7 ± 5.8 mmHg to 11.4 ± 2.6 mmHg at 18 months (P < 0.0001). In the standalone procedure group, preoperative IOP decreased from 21.5 ± 5.2 mmHg to 11.7 ± 2.5 mmHg (P < 0.0001), and in the combined procedure group, preoperative IOP decreased from 17.1 ± 5.8 mmHg to 10.9 ± 2.7 mmHg (P = 0.0002), with no significant difference between the two groups regarding final IOP. The mean number of antiglaucoma medications significantly decreased from 3.2 ± 1.1 to 0.3 ± 0.7 in the overall study population (P < 0.0001). At 18 months, 40 eyes (83.3%) were classified as successful. Regarding safety, out of the total number of eyes, two (4.2%) experienced choroidal detachment without visual impairment, two (4.2%) had transient hyphema, one (2.1%) showed reactivation of a corneal herpetic ulcer, one (2.1%) had diplopia, and one (2.1%) exhibited a shallow anterior chamber during the first week.
Conclusion:
The combination of Ologen™ and Preserflo™ MicroShunt, either alone or in conjunction with phacoemulsification, demonstrated a favorable profile in terms of IOP reduction and safety.
Keywords: Glaucoma, glaucoma surgery, intraocular pressure, MIGS, Ologen, Preserflo MicroShunt
Glaucoma is a progressive and often asymptomatic group of diseases that affect the optic nerve, leading to optic-nerve cupping, loss of retinal ganglion cells, and visual field loss, which can ultimately result in blindness. The primary modifiable risk factor for glaucoma is increased intraocular pressure (IOP), making IOP reduction a key goal in glaucoma treatment, including surgery.[1,2]
Angle-based minimally invasive glaucoma surgeries (MIGS) and bleb-forming MIGS have emerged as viable options for managing IOP and reducing medication dependence, particularly in cases of mild-to-moderate glaucoma damage.[3]
Different ab interno and ab externo MIGS use filtration either into Schlemm's canal or the suprachoroidal space, whereas bleb-forming surgeries use filtration to the subconjunctival space, aiming to lower IOP in a safer way as compared with conventional and time-proven surgeries such as trabeculectomy and long-tube glaucoma implants.[4,5]
However, most existing MIGS procedures offer only moderate IOP reduction, leaving a gap in the availability of a safer and more predictable option for eyes with severe damage or complicated secondary glaucoma that necessitate lower-target IOPs to prevent further optic nerve deterioration.
Especially during the COVID-19 pandemic, conventional and bleb-forming MIGS devices have gained popularity due to their reduced surgical duration and lower risk of sight-threatening complications, resulting in a reduced number of postoperative visits.[6] In this context, the Preserflo® MicroShunt (Preserflo MicroShunt, Santen Pharmaceutical Co., Osaka, Japan) has emerged as a novel bleb-forming device. The MicroShunt is made of poly (styrene-block-isobutylene-block-styrene) or SIBS, a biocompatible material that exhibits resistance to biodegradation, biologically inert properties, softness, and flexibility, thereby minimizing inflammation and scar tissue formation.[7] Clinical reports have demonstrated promising mid- and long-term IOP control with few self-limiting complications.[7]
Ologen™ (Aeon Astron Europe B. V., Netherlands) is a porous matrix implant composed of collagen–glycosaminoglycan. It serves as both a spacer and a wound modulator in glaucoma surgeries.[8] Although it has been primarily used in trabeculectomy procedures, it has also been employed in combination with other glaucoma implants for both adult and pediatric patients.[9,10] A recent publication examined the combination of OlogenTM and 0.2 mg/mL Mitomycin C (MMC) in Preserflo™ surgery and found no significant additional benefit compared to the manufacturer’s recommended 0.2 mg/mL MMC alone after a 6-month follow-up in eyes with primary open-angle glaucoma (POAG).[11] We have hypothesized that a thinner implant would induce less tension on the filtration bleb, which would translate into a lower risk of fibrosis. In addition, a larger surface area of the implant would mean greater filtration and, therefore, lower IOP.
This study aimed to assess the impact of combining a thinner and larger version of Ologen™ with Preserflo™ after an 18-month follow-up period. To the best of our knowledge, this is the first report investigating the use of this specific Ologen™ model in different types of glaucoma.
Methods
Study design
This retrospective, single-center study was conducted on patients who underwent Preserflo™ MicroShunt implant surgery combined with simultaneous Ologen™ implant. The follow-up period for the study was 18 months.
Ethical considerations
All patients provided written informed consent for their surgery, in accordance with the requirements of our institution Hospital Virgen de la Luz de Cuenca. The study complied with local and European legislation and ensured the use of clinical data without any identifiers. Furthermore, the study adhered to the principles outlined in the Declaration of Helsinki.
Participants
The study included consecutive patients aged 18 years or older, with glaucoma, who had insufficiently medically controlled IOP, poor treatment adherence, or intolerance to topical hypotensive medication. Patients with conjunctival pathologies (except for previous glaucoma surgery) that could potentially compromise the outcomes of the procedure, as determined by the surgeon, were excluded from the study.
Medication withdrawal
Prior to surgery, patients were instructed to discontinue the use of topical and systemic ocular hypotensive medications.
Surgical technique
All surgeries were performed by a single surgeon MRA under local anesthesia by using a combination of topical and sub-Tenon’s bupivacaine 0.5% and mupivacaine 2%.
The Preserflo™ MicroShunt implantation technique followed the manufacturer’s recommendation, with the additional implantation of the Ologen™ model 862051. This model, chosen due to its availability at our institution, features a round shape with a diameter of 12 mm and a height of 1 mm, placed under Tenon’s capsule. The intention behind using this model was to achieve a larger extension area and a lower height of bleb filtration.
The surgical procedure involved a 70–90° fornix-based peritomy in the selected quadrant, followed by the use of light bipolar cautery and careful elevation of Tenon’s capsule to expose the episclera adequately. Topical MMC at concentrations of either 0.2 or 0.4 mg/mL was applied using the provided 3 LASIK shields for a duration of 2–4 min. The choice of MMC concentration and exposure time was based on the surgeon’s preference and patient characteristics. To prevent excessively elevated limbus bleb and reduce the chances of bleb leakage, the shields were placed as far away from the limbus as possible.
A marker was used to measure 3 mm from the limbus, and a small 1-mm-wide shallow scleral pocket was dissected using a triangular-shaped knife. Subsequently, a 27-G needle tract was created to enter the anterior chamber (AC) just above the iris, away from the corneal endothelium. The MicroShunt device was then inserted through the needle tract until the fins of the implant fixated it to the wider part of the scleral pocket. This ensures a predictable length inside the AC and prevents rotation, allowing the bevel to remain with the opening away from the iris.[12]
Visual confirmation of adequate flow through the device was obtained, and if necessary, flow was promoted by increasing pressure through corneal indentation or by injecting balanced saline solution (BSS) into the anterior chamber. The Ologen™ implant was inserted above the outer tip of the MicroShunt and under Tenon’s capsule, taking care to prevent it from occluding the distal portion of the MicroShunt. Conjunctival closure was achieved using nylon 10-0 sutures with a running mattress technique until no aqueous leaks were observed. Postoperative medication included a combination of topical tobramycin and dexamethasone for the first 2 weeks, followed by a switch to prednisolone acetate 10 mg/mL with a gradually reducing regimen over the next 8–12 weeks.
Study groups
The study sample was divided into two different groups: the MicroShunt solo procedure group, consisting of eyes that underwent Preserflo MicroShunt + Ologen standalone surgery, and the MicroShunt + Phaco group, comprising eyes that underwent combined surgery (MicroShunt + Ologen + phacoemulsification).
Definitions
Success was defined as achieving an IOP of 6–15 mmHg, with an IOP reduction of at least 20% relative to the preoperative value, either without any hypotensive medication (complete success) or with the use of hypotensive medication (qualified success) at the month-18 visit.
Patients with an IOP of <6 mmHg for more than two consecutive visits, those requiring further glaucoma surgery, or those undergoing surgery due to complications were considered treatment failures.
Outcomes
The primary endpoint of the study was the proportion of patients classified as successful at the month-18 visit.
Secondary endpoints included the mean IOP and mean IOP reduction at the month-18 visit, the mean number of antiglaucoma medications used and changes from baseline, the identification of predictive factors associated with success, and the incidence of adverse events.
Statistical analysis
Standard statistical analysis was conducted using MedCalc® Statistical Software version 20.2.18 (MedCalc Software Ltd, Ostend, Belgium; https://www.medcalc.org; 2023).
Descriptive statistics were reported as mean ± standard deviation (SD), mean ± standard error of the mean (SE), median (interquartile range) (IQR), or number (percentage) as appropriate.
The normal distribution of data was assessed using the Shapiro–Wilk test.
Patients with more than 10% missing data were excluded from the analysis, and case-wise deletion was applied to the remaining dataset. Patients with ≤10% missing data were not withdrawn from the study. In these cases, missing values were allocated using an algorithm of multiple imputation.[13]
Changes in IOP and the number of antiglaucoma medications were assessed using repeated measures analysis of variance (ANOVA) or Friedman’s two-way analysis test, as appropriate. Post-hoc analysis for pairwise comparisons was conducted using Scheffé’s method (ANOVA) or the Conover method (Friedman).
Repeated analysis of covariance (MANCOVA) was performed to evaluate changes in IOP between the study groups. The model included “type of surgery” (MicroShunt solo or MicroShunt + Phaco) as a factor, with age, preoperative IOP, pachymetry, and previous glaucoma surgery as covariates.
To assess preoperative differences between cohorts, the Mann–Whitney test was used for continuous variables.
Survival rates of success were analyzed using Kaplan–Meier analysis for the MicroShunt-solo and MicroShunt + Phaco groups, with comparison conducted using the log-rank test.
A logistic regression model was utilized to estimate and test factors associated with success. A backward strategy was adopted, with a statistically significant cutoff of 0.05 for variable screening. Factors demonstrating an association with progression in the univariate analysis at P ≤ 0.1 were included in the multivariate analysis.
Categorical variables were compared using the Chi-square test and Fisher’s exact test, as necessary. A P value of less than 0.05 was considered statistically significant.
Results
A total of 48 eyes from 47 patients were included in the study, with 28 (58.3%) eyes undergoing a standalone procedure (MicroShunt solo) and 20 (41.7%) eyes undergoing a combined procedure (MicroShunt + Phaco).
Preoperative demographic and clinical characteristics:
In the overall study population, the mean age was 72.5 ± 10.2 years, and 32 (66.7%) patients were women. The most frequent diagnoses were POAG and chronic angle-closure glaucoma (CACG), accounting for 33 (68.7%) and 9 (18.8%) eyes, respectively.
Fourteen eyes (29.2%) had previously undergone antiglaucomatous surgery. A majority of the eyes, 38 (79.2%), received MMC at a concentration of 0.4 mg/mL.
The mean preoperative IOP was significantly higher in the MicroShunt-solo group (21.5 ± 5.2 mmHg) compared to the MicroShunt + Phaco group (17.1 ± 5.8 mmHg), with a median difference of 4.0 mmHg (95% confidence interval, 95% CI: 4.0–7.0 mmHg, P = 0.0025).
Eyes that had previously undergone a surgical procedure underwent a MicroShunt standalone procedure.
The main demographic and clinical characteristics of the study sample are summarized in Table 1.
Table 1.
Baseline demographic and clinical characteristics of the study population
| Characteristic | Total population (n=48) | MicroShunt Solo* (n=28) | MicroShunt* + Phaco (n=20) | P | |
|---|---|---|---|---|---|
| Age, years | |||||
| Mean (SD) | 72.5±10.2 | 72.5±10.4 | 72.4±10.3 | 0.9913a | |
| Median (IqR) | 73.0 (63.3–81.0) | 73.0 (67.0–81.0) | 78.0 (63.8–81.0) | ||
| Sex, n (%) | |||||
| Female | 32 (66.7) | 18 (64.3) | 14 (70.0) | 0.7626b | |
| Male | 16 (33.3) | 10 (35.7) | 6 (30.0) | ||
| Eye, n (%) | |||||
| Right | 28 (58.3) | 16 (57.1) | 12 (60.0) | 1.0000b | |
| Left | 20 (41.7) | 12 (42.9) | 8 (40.0) | ||
| BCVA, LogMar | |||||
| Mean (SD) | 0.30±0.47 | 0.23±0.57 | 0.39±0.26 | 0.0008a | |
| Median (IqR) | 0.15 (0.03–0.40) | 0.08 (0.0–0.23) | 0.40 (0.25–0.50) | ||
| Pachymetry, µm | |||||
| Mean (SD) | 544.9±40.1 | 551.0±36.0 | 536.5±44.8 | 0.6529a | |
| Median (IqR) | 542.5 (522.5–563.5) | 542.5 (522.5–563.5) | 542.5 (518.5–565.0) | ||
| Diagnosis, n (%) | |||||
| POAG | 33 (68.7) | 21 (75.0) | 12 (60.0) | 0.1378c | |
| CACG | 9 (18.8) | 6 (21.4) | 3 (15.0) | ||
| NTG | 3 (6.2) | 0 (0.0) | 3 (15.0) | ||
| PG | 1 (2.1) | 0 (0.0) | 1 (5.0) | ||
| Other | 2 (4.2) | 1 (3.6) | 1 (5.0) | ||
| Previous Glaucoma surgery, n (%) | |||||
| Yes | 14 (29.2) | 14 (50.0) | 0 (0.0) | 0.0001b | |
| No | 34 (70.8) | 14 (50.0) | 20 (100.0) | ||
| MMC dose, n (%) | |||||
| 0.02% | 10 (20.8) | 5 (17.9) | 5 (25.0) | 0.7209b | |
| 0.04% | 38 (79.2) | 23 (82.1) | 15 (75.0) | ||
| Initial** IOP, mmHg | |||||
| Mean (SD) | 28.5±7.3 | 30.0±7.3 | 26.5±6.9 | 0.0855a | |
| Median (IqR) | 27.5 (25.0–29.5) | 28.0 (26.0–31.0) | 25.5 (22.5–28.5) | ||
| Preop IOP, mmHg | |||||
| Mean (SD) | 19.7±5.8 | 21.5±5.2 | 17.1±5.8 | 0.0025a | |
| Median (IqR) | 18.0 (16.0–22.0) | 20.0 (18.0–23.5) | 16.0 (13.5–20.0) | ||
| NOHM | |||||
| Mean (SD) | 3.2±1.1 | 3.4±1.0 | 2.9±1.1 | 0.0902a | |
| Median (IqR) | 3.0 (3.0–4.0) | 3.5 (3.0–4.0) | 3.0 (2.0–4.0) | ||
| NOHM, n (%) | |||||
| 0 | 2 (4.2) | 1 (3.6) | 1 (5.0) | <0.1024c | |
| 1 | 1 (2.1) | 0 (0.0) | 1 (5.0) | ||
| 2 | 7 (14.6) | 3 (10.7) | 4 (20.0) | ||
| 3 | 18 (37.5) | 10 (35.7) | 8 (40.0) | ||
| 4 | 18 (37.5) | 12 (42.9) | 6 (30.0) | ||
| 5 | 2 (4.2) | 2 (7.1) | 0 (0.0) |
aMann–Whitney test. bFisher’s exact test. cChi-squared test for trend. *PreserfloTM MicroShunt + OlogenTM implant. **Untreated intraocular pressure, when the patient arrived at the clinic. SD: Standard deviation; CI: Confidence interval; POAG: Primary open-angle glaucoma; CACG: Chronic angle-closure glaucoma; NTG: Normal-tension glaucoma; PG: Pigmentary glaucoma; MMC: Mitomycin-C; IOP: Intraocular pressure; NOHM. Number of ocular hypotensive medications; Phaco: Phacoemulsification
Success rate
The overall success rate in the study population was 83.3% (40/48) eyes. Specifically, the success rate was 92.9% (26/28) eyes in the MicroShunt-solo group and 70.0% (14/20) eyes in the MicroShunt + Phaco group (P = 0.0532). Kaplan–Meier survival analysis demonstrated significantly greater survival in the MicroShunt-solo group compared to the MicroShunt + Phaco group (Mean hazard ratio: 4.56; 95% CI: 1.06–19.59; P = 0.0414) [Fig. 1].
Figure 1.

Kaplan–Meier survival analysis. The mean hazard ratio was 4.56 (95% CI: 1.06–19.59, P = 0.0414)
Intraocular pressure
In the overall study sample, there was a significant reduction in mean IOP from 19.7 ± 5.8 mmHg to 11.4 ± 2.6 mmHg [Fig. 2a], with a mean difference of − 8.3 ± 5.7 mmHg (95% CI: −10.0 to − 6.7 mmHg; P < 0.0001, repeated measures ANOVA).
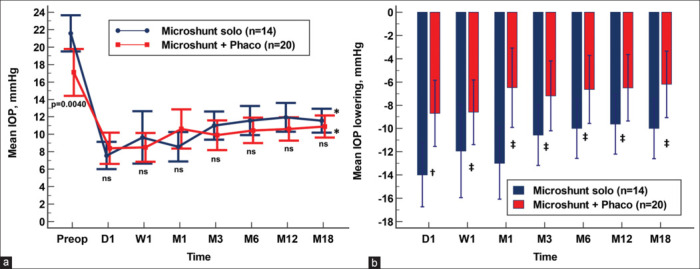
Figure 2.

Mean intraocular pressure (IOP) over the course of follow-up. The vertical bars represent the 95% confidence interval. (a) Overall study population. *P < 0.0001 as compared to preoperative value (repeated measures ANOVA and the Greenhouse-Geisser correction). (b) Comparison between eyes that underwent MicroShunt-solo and those that underwent MicroShunt + phacoemulsification (MicroShunt + Phaco) surgery. The preoperative mean IOP was significantly lower in the MicroShunt + Phaco group (P = 0.0025). No significant differences were observed at any other measured IOP time points (Mann–Whitney U test). Compared to preoperative values, IOP was significantly lowered at all different time points. **P < 0.0001 as compared to preoperative value (Friedman test with the Conover method). IOP: Intraocular pressure; MicroShunt solo: Preserflo™ MicroShunt + Ologen™; MicroShunt + Phaco: Preserflo™ MicroShunt + Ologen™ + Phacoemulsification; Preop: Preoperative; D: Day; W: Week; M: Month; ns: not significant
Both the MicroShunt-solo and MicroShunt + Phaco groups showed a significant decrease in mean preoperative IOP from 21.5 ± 5.2 mmHg and 17.1 ± 5.8 mmHg to 11.7 ± 2.5 mmHg and 10.9 ± 2.7 mmHg, respectively (P < 0.0001 for each, repeated measures ANOVA).
No significant differences in mean IOP were observed between the two study groups at any of the measured time points [Fig. 2b].
Although unadjusted IOP lowering was significantly greater in the MicroShunt-solo group compared to the MicroShunt + Phaco group [Fig. 3], after adjusting for different covariates (age, preoperative IOP, pachymetry, and previous glaucoma surgery), there were no significant differences in IOP lowering between the groups [Table 2].
Figure 3.

Comparison of the mean intraocular pressure (IOP) lowering from preoperative values in PreserfloTM MicroShunt + OlogenTM (MicroShunt-solo) and PreserfloTM MicroShunt + OlogenTM + Phacoemulsification (MicroShunt + Phaco) groups. *p < 0.0001, **p < 0.05. Statistical significance was determined using the Mann–Whitney test. IOP: Intraocular pressure
Table 2.
Adjusted Mean changes in intraocular pressure (IOP) over the course of follow-up in eyes that underwent PreserfloTM MicroShunt + OlogenTM (MicroShunt solo) versus those who underwent PreserfloTM MicroShunt + OlogenTM + Phacoemulsification (MicroShunt + Phaco)
| Mean change in IOP, mmHg | MicroShunt solo |
MicroShunt + Phaco |
Difference |
||||
|---|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | Mean (SE) | 95% CI | P | |
| Day 1 | −12.4 | 0.7 | −10.7 | 0.8 | −1.7 (1.2) | −4.1 to 0.7 | 0.1573 |
| Week 1 | −10.4 | 0.8 | −11.1 | 1.1 | 0.7 (1.5) | −2.3 to 3.7 | 0.6649 |
| Month 1 | −10.6 | 0.8 | −8.2 | 0.9 | −2.5 (1.3) | −0.4 to 5.0 | 0.0860 |
| Month 3 | −9.0 | 0.6 | −9.5 | 0.8 | 0.5 (1.1) | −1.6 to 2.7 | 0.6341 |
| Month 6 | −8.5 | 0.6 | −9.1 | 0.7 | 0.6 (1.0) | −1.4 to 2.5 | 0.5418 |
| Month 12 | −8.3 | 0.4 | −8.8 | 0.6 | 0.5 (0.8) | −1.0 to 2.1 | 0.4890 |
| Month 18 | −8.2 | 0.4 | −8.4 | 0.5 | 0.2 (0.8) | −1.4 to 1.8 | 0.7860 |
| Significance* | P<0.0001 | P<0.0001 | |||||
aBonferroni corrected. *Repeated measures ANCOVA and the Greenhouse–Geisser correction. aRepeated measures analysis of covariance (MANCOVA). The model included “Type of surgery” (MicroShunt-solo vs. MicroShunt + Phaco) as a factor and age, preoperative IOP, pachymetry; and previous glaucoma surgery as covariates. IOP=Intraocular pressure; SE=Standard error; CI=Confidence interval
Ocular hypotensive medications
The mean number of ocular hypotensive medications significantly decreased from 3.2 ± 1.1 drugs preoperatively to 0.3 ± 0.8 drugs at the month-18 visit. However, there were no statistically significant differences in the mean reduction of ocular hypotensive medications between the MicroShunt-solo (3.1 ± 1.1 drugs) and MicroShunt + Phaco (2.5 ± 1.3 drugs) groups (Hodges–Lehmann median difference: −1.0; 95% CI: −1.0 to 0.0; P = 0.1197) [Fig. 4].
Figure 4.

Mean number of ocular hypotensive medications over the course of follow-up. Statistical significance was determined using the Mann–Whitney test. Preop: Preoperative; ns: Not-significant; MicroShunt-solo: PreserfloTM MicroShunt + OlogenTM; MicroShunt + Phaco: PreserfloTM MicroShunt + OlogenTM + Phacoemulsification
Predictive factors for success
In logistic regression analysis, no factors were found to be significantly related to success in the univariate analysis. However, there was a tendency for performing cataract surgery and having an IOP of >8 mmHg on postoperative day 1 to reduce the probability of success (odds ratio = 0.18; P = 0.0513 and odds ratio = 0.123; P = 0.0682, respectively), although these did not reach statistical significance [Table 3].
Table 3.
Univariate and multivariate analysis of the 48 eyes included in the study, to evaluate potential factors for success. Factors associated with success in the univariate analysis at P<0.1 were included in the multivariate analysis
| Variable | Success |
|||
|---|---|---|---|---|
| Univariate |
Multivariate |
|||
| OR (95% CI) | P | OR (95% CI) | P | |
| Age* | ||||
| >73 years | 1.67 (0.35–7.93) | 0.5209 | ||
| Sex | ||||
| Ref: Female | 0.80 (0.17–3.88) | 0.7845 | ||
| Male | ||||
| Eye | ||||
| Ref: Right | 0.67 (0.14–3.05) | 0.6019 | ||
| Left | ||||
| Device implantation | ||||
| Ref: ST | 1.80 (0.19–16.98) | 0.6077 | ||
| SN | 0.20 (0.01–3.66) | 0.2779 | ||
| IN | ||||
| Previous glaucoma surgery | ||||
| Ref: No | 1.29 (0.23–7.31) | 0.7768 | ||
| Yes | ||||
| Cataract surgery | ||||
| Ref: No | 0.18 (0.04–1.01) | 0.0513 | 0.19 (0.03–1.13) | 0.0679 |
| Yes | ||||
| Pachymetry | ||||
| >542.5 µm | 1.00 (0.22–4.56) | 1.0000 | ||
| MMC dose | ||||
| Ref: 0.02% | 1.33 (0.23–7.89) | 0.7511 | ||
| 0.04% | ||||
| Initial IOP | ||||
| >27.5 mmHg | 0.54 (0.11–2.58) | 0.4428 | ||
| Preop IOP | ||||
| >18 mmHg | 3.32 (0.60–18.45) | 0.1711 | ||
| Day 1 | ||||
| IOP >8 mmHg | 0.23 (0.05–1.12) | 0.0682 | 0.24 (0.05–1.29) | 0.0973 |
| NOHM | ||||
| Per medication | 0.64 (0.27–1.52) | 0.3070 | ||
*Reference group ≤ Median. ST: Superotemporal; SN: Superonasal; IN: Inferonasal; MMC: Mitomycin-C; IOP: Intraocular pressure; NOHM: Number of ocular hypotensive medications
Visual acuity
In the overall study sample, there were no significant changes in LogMAR mean best-corrected visual acuity (BCVA) from preoperative to month-18 visit (0.30 ± 0.47 vs. 0.24 ± 0.61; P = 0.2147). However, in the MicroShunt + Phaco group, LogMAR BCVA significantly improved from 0.39 ± 0.26 preoperatively to 0.17 ± 0.19 at the month-18 visit (Hodges–Lehmann median difference: −0.21; 95% CI: −0.35 to − 0.13; P = 0.0003). In contrast, in the MicroShunt-solo group, BCVA remained stable (Hodges–Lehmann median difference: −0.00; 95% CI: −0.03 to 0.05; P = 0.5693).
Eyes with previous glaucoma surgery
Fourteen eyes (19.2%) had undergone previous glaucoma surgery, with 12 eyes undergoing one antiglaucomatous surgical procedure and two eyes undergoing two glaucoma surgeries [Table 4]. All of these eyes underwent a MicroShunt standalone procedure. The mean age was 72.6 ± 11.4 years (range: 39–86 years), with seven (50.0%) women, and 13 (92.9%) underwent surgery with MMC 0.04%. Among these eyes, 12 (85.7%) were classified as a success, with 10 (71.4%) as complete success and 2 (14.3%) as qualified success. The mean preoperative IOP significantly reduced from 21.5 ± 6.5 mmHg to 11.9 ± 2.7 mmHg at month 18 (Hodges–Lehmann median difference: −9.0; 95% CI: −13.0 to − 6.0 mmHg; P = 0.0001). Similarly, the mean preoperative number of ocular hypotensive medications significantly reduced from 3.3 ± 1.3 to 0.4 ± 0.8 at month 18 (Hodges–Lehmann median difference: −3.0; 95% CI: −3.5 to − 2.0; P = 0.0002). There were no significant differences in either preoperative BCVA (Hodges–Lehmann median difference: 0.05; 95% CI: −0.05 to 0.15; P = 0.3825) or month-18 BCVA (Hodges–Lehmann median difference: 0.0; 95% CI: −0.1 to 0.1; P = 0.9435) between the two time points.
Table 4.
Overview of the main demographic and clinical characteristics of the eyes that had undergone previous glaucoma surgery
| n | Age | Sex | Eye | Dx | Previous Surgery | Surgery | BCVA |
IOP |
NOHM |
Complications* | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Preop | Month-18 | Initial | Preop | Month-18 | Preop | Month-18 | ||||||||
| 1 | 39 | F | R | POAG | NPDS | Standalone | 0.5 | 0.3 | 50 | 34 | 15 | 4 | 0 | No |
| 2 | 81 | F | L | POAG | CP | Standalone | 0.05 | 0.05 | 26 | 22 | 12 | 3 | 0 | No |
| 3 | 63 | M | L | POAG | AhV | Standalone | 0.4 | 1.5 | 50 | 35 | 17 | 5 | 2 | No |
| 4 | 81 | M | L | POAG | XEN | Standalone | 0 | 0 | 28 | 20 | 8 | 4 | 0 | CD |
| 5 | 66 | M | R | CACG | TRAB | Standalone | 0 | 0 | 26 | 18 | 14 | 4 | 0 | SAC |
| 6 | 72 | F | L | POAG | CP | Standalone | 0.1 | 0.1 | 25 | 16 | 9 | 3 | 0 | No |
| 7 | 81 | F | R | POAG | CP | Standalone | 0.15 | 0.1 | 26 | 19 | 11 | 2 | 0 | Fibrosis |
| 8 | 73 | F | R | POAG | NPDS & AhV | Standalone | 0.05 | 0.2 | 28 | 20 | 12 | 4 | 0 | No |
| 9 | 86 | F | R | CACG | NPDS | Standalone | 0.5 | 0.4 | 34 | 30 | 11 | 4 | 2 | No |
| 10 | 77 | F | R | POAG | NPDS | Standalone | 0 | 0.15 | 28 | 18 | 8 | 4 | 0 | Diplopia |
| 11 | 75 | M | R | POAG | NPDS & AhV | Standalone | 0 | 0 | 28 | 16 | 14 | 4 | 0 | No |
| 12 | 73 | M | L | POAG | CP | Standalone | 0.05 | 0 | 28 | 17 | 14 | 0 | 0 | No |
| 13 | 77 | F | R | POAG | TRAB | Standalone | 0 | 0.15 | 24 | 18 | 10 | 3 | 1 | Hyphema |
| 14 | 72 | M | R | CACG | XEN | Standalone | 0 | 0 | 25 | 18 | 11 | 2 | 0 | No |
*MicroShunt post-operative complications. N: Case number; IOP: Intraocular pressure; F: Female; M: Male; Dx: Diagnosis; POAG: Primary open-angle glaucoma; CACG: Chronic angle-closure glaucoma; NPDS: Non-penetrating deep sclerectomy; AhV: Ahmed valve; CP: Canaloplasty; TRAB: Trabeculectomy; Preop: Preoperative; BCVA: Best corrected visual acuity; IOP: Intraocular pressure; Initial: Baseline, untreated IOP, when the patient arrived to the clinic; NOHM: Number of ocular hypotensive medications; SAC: Shallow anterior chamber; CD: Choroidal detachment
Eyes without previous surgery
To minimize potential biases, a subanalysis was performed on eyes that had not undergone previous surgery. Except for preoperative IOP, which was significantly higher in the MicroShunt-solo group, no significant differences were observed at any of the different time points measured throughout the study follow-up [Fig. 5a]. However, the mean IOP lowering was significantly greater in the MicroShunt-solo group than in the MicroShunt + Phaco group [Fig. 5b]. In the overall study population, the preoperative mean IOP significantly reduced from 18.9 ± 5.4 mmHg to 11.2 ± 2.6 mmHg (mean difference: −7.8 ± 5.8 mmHg; 95% CI: −9.8 to − 5.8 mmHg; P < 0.0001, Friedman test). In the MicroShunt-solo group, preoperative IOP significantly reduced from 21.6 ± 3.6 mmHg to 11.6 ± 2.4 mmHg at month 18 (P < 0.0001) [Fig. 5a]. Similarly, there was a significant IOP reduction in the MicroShunt + Phaco group from preoperative (17.1 ± 5.8 mmHg) to month 18 (10.9 ± 2.7 mmHg) [Fig. 5a]. Unadjusted IOP lowering was significantly greater in the MicroShunt-solo group compared to the MicroShunt + Phaco group at all the different time points measured [Fig. 5b].
Figure 5.
Mean intraocular pressure (IOP) (a) and mean IOP lowering (b) over the course of follow-up in the eyes that did not undergo previous glaucoma surgery. The vertical bars represent the 95% confidence interval. (a) A comparison of mean IOP between the eyes that underwent MicroShunt-solo and those that underwent MicroShunt + phacoemulsification (MicroShunt + Phaco) surgery. The preoperative mean IOP was significantly lower in the MicroShunt + Phaco group. No significant differences were observed at any other measured IOP time points (Mann–Whitney U test). IOP was significantly lowered at all different time points compared to preoperative values (*p < 0.0001, Friedman test with the Conover method). (b) Comparison of the mean IOP lowering from preoperative values in MicroShunt-solo and MicroShunt + Phaco groups. Statistical significance was determined using the Mann–Whitney test (†p < 0.0001, ‡p < 0.05). IOP: Intraocular pressure; MicroShunt-solo: PreserfloTM MicroShunt + OlogenTM; MicroShunt + Phaco: PreserfloTM MicroShunt + OlogenTM + Phacoemulsification; Preop: Preoperative; D: Day; W: Week; M: Month; ns: Not Significant
Safety
In terms of surgical complications, two (4.2%) eyes experienced choroidal detachment without visual impairment, two (4.2%) eyes had transient hyphema, one (2.1%) eye had a reactivation of a corneal herpetic ulcer, one (2.1%) eye had diplopia, and one (2.1%) eye had a shallow anterior chamber during the first week. All cases of postoperative surgical complications were successfully managed with medical therapy.
No eyes required needling, and only one (2.1%) eye (diagnosed with chronic angle-closure glaucoma) needed repositioning of the implant due to the tip being too close to the corneal endothelium. After 18 months of follow-up, this eye had an IOP of 10 mmHg with the use of two medications.
Discussion
In recent years, significant advancements have been made in the field of glaucoma surgery. MIGS devices have emerged as safer and less traumatic alternatives for lowering IOP in glaucoma patients.[14] Among the various MIGS devices available, the Preserflo™ MicroShunt is an ab-externo and subconjunctival device that has demonstrated favorable efficacy and safety profiles.[15,16,17,18,19,20,21,22,23,24,25]
In this study, the overall success rate in the study population was 83.3%. The MicroShunt-solo group achieved a success rate of 92.9%, whereas the MicroShunt + Phaco group had a success rate of 70.0%. Although there were no significant differences between the two groups (P = 0.0532), both the standalone use of the MicroShunt and its combination with phacoemulsification demonstrated significant reductions in IOP and the number of ocular hypotensive medications in glaucoma patients treated in a real clinical setting.
Initially, the unadjusted analysis showed significantly greater IOP lowering in the MicroShunt-solo group at all measured time points. However, after adjusting for various covariates, there were no significant differences in IOP reduction between the standalone MicroShunt implant and its combination with cataract surgery at any of the measured time points.
Despite the increasing evidence supporting the use of the MicroShunt implant in glaucoma patients,[15,16,17,18,19,20,21,22,23,24,25] there are limited studies analyzing its effectiveness in combination with the collagen–glycosaminoglycan porous matrix implant, Ologen™. To the best of our knowledge, only one study has evaluated the clinical outcomes of the MicroShunt in pseudophakic eyes with moderate to advanced POAG.[11]
Vastardis et al.[11] reported a 6-month success rate of 91.8% using the Preserflo device with a success criterion of IOP ≥5 and ≤16 mmHg. In our study, we observed similar success rates of 92.9% in eyes that underwent the solo procedure, although our success criteria were stricter (IOP ≥6 and ≤15 mmHg) and our follow-up was three times longer (18 months compared to Vastardis et al.’s 6 months).
In terms of IOP reduction, Vastardis et al. reported a significant decrease from 26.04 ± 8.76 mmHg preoperatively to 11.75 ± 3.37 mmHg at 6 months. In our study, the mean preoperative IOP in the MicroShunt-solo group significantly reduced from 21.5 ± 5.2 mmHg to 10.9 ± 3.0 mmHg at 6 months and 11.7 ± 2.5 mmHg at 18 months.
Regarding the reduction of ocular hypotensive medications, our study yielded comparable results to Vastardis et al.,[11] despite our much longer follow-up duration.
It is worth noting that unlike Vastardis et al.,[11] who performed surgical revision in three eyes in the Ologen™ group, our study did not encounter any cases of failed or flat blebs. Although we did have two failures due to elevated IOP, which might have benefited from needling, this procedure was not included as part of our post-surgical management protocol. This disparity can be partially explained by differences in the use of MMC as we used a concentration of 0.04% in 82.1% of the eyes that underwent the solo procedure.
Another potential explanation for the differences is the Ologen™ model itself. In our study, we used a 12-mm diameter × 1-mm thickness implant, which could increase the total filtering area and place less stress on the conjunctiva, whereas Vastardis et al.[11] used a 6-mm diameter × 2-mm thickness implant.
Direct comparisons with previous studies are challenging as they did not utilize the OlogenTM implant.[15,16,17,18,19,20,21,22,23,24,25] Nevertheless, the observed good IOP lowering effect in our study was in line with those previous reports and did not significantly differ [Table 5].
Table 5.
Overview of the intraocular pressure (IOP) and number of hypotensive medications in eyes that underwent PreserfloTM MicroShunt implant surgery in different studies
| Study | n | Follow-up | Preoperative IOP, mmHg | Final IOP, mmHg | Mean preoperative medications | Mean postoperative medications |
|---|---|---|---|---|---|---|
| Vastardis et al.[11] | ||||||
| MMC 0.02% | 25 | 6M | 23.5±5.8* | 11.6±3.1* | 2.5±0.9* | 0.04±0.2* |
| MMC 0.02% + OlogenTM | 25 | 6M | 26.0±8.8* | 11.8±3.4* | 2.6±0.8* | 0.2±0.8* |
| Riss et al.[15] | ||||||
| MMC 0.02% | 31 | 12 M | 27.9±6.7* | 13.3±3.3* | 2.5±1.4* | 0.5±1.0* |
| MMC 0.04% | 23 | 12 M | 23.8±5.3* | 10.7±2.8* | 2.4±0.9* | 0.3±0.8* |
| Batlle et al.[16] | 23 | 12M | 23.8±5.3* | 10.7±2.8* | 2.4±0.9* | 0.3±0.8* |
| 22 | 24M | 23.8±5.3* | 11.9±3.7* | 2.4±0.9* | 0.4±1.0* | |
| 22 | 36M | 23.8±5.3* | 10.7±3.5* | 2.4±0.9* | 0.7±1.1* | |
| Scheres et al.[17] | 41 | 18M | 20.1±5.0* | 12.5±3.3* | 2.3±1.5* | 0.6±0.9* |
| Batlle et al.[18] | 21 | 48M | 23.8±5.3* | 12.8±5.6* | 2.4±1.0* | 0.5±1.1* |
| 21 | 60M | 23.8±5.3* | 12.4±6.5* | 2.4±1.0* | 0.8±1.3* | |
| Martínez-de-la-Casa et al.[20] | ||||||
| MicroShunt | 35 | 12M | 21.3±3.2* | 14.4±3.4* | 2.3±0.5* | −2.1±0.7** |
| MicroShunt + Phaco | 23 | 12M | 21.7±3.5* | 14.9±3.6* | 2.4±0.5* | −2.1±0.6** |
| Quaranta et al.[21] | 31 | 12M | 24.1±3.1* | 12.6±2.6* | 3.3±0.6* | 0.5±0.8* |
| Fea et al.[22] | 104 | 12M | 25.1±6.5* | 14.1±3.4* | 3.0±1.0* | 0.8±1.0* |
| Beckers et al.[23] | 81 | 12M | 21.7±3.4* | 14.5±4.6* | 2.1±1.3* | N.A. |
| 81 | 24M | 21.7±3.4* | 14.1±3.2* | 2.1±1.3* | 0.5±0.9* | |
| Ibarz-Barberá et al.[24] | ||||||
| MicroShunt | 51 | 9M | 22.5±0.9* | 12.8±0.4*,† | 2.7±0.7*,‡ | 0.19±0.5*,‡ |
| MicroShunt + Phaco | 13 | 9M | 20.1±1.3* | 12.5±0.6*,† | ||
| Bhayani et al.[25] | 100 | 12M | 21.5 (19–28)⁂ | 13 (11–16)⁂ | 3 (2–3)⁂ | 0 (0–1)⁂ |
| Current study | ||||||
| Overall | 48 | 18M | 19.7±5.8* | 11.4±2.6* | 3.2±1.1* | 0.3±0.8* |
| MicroShunt | 28 | 18M | 21.5±5.2* | 11.7±2.5* | 3.4±1.0* | 0.3±0.7* |
| MicroShunt + Phaco | 20 | 18M | 17.1±5.8* | 10.9±2.7* | 2.9±1.1* | 0.4±0.9* |
*Mean±Standard deviation. **Mean±Standard deviation reduction. †”Last follow-up visit,” without specifying follow-up. ‡Overall study sample. ⁂Median (Interquartile range). IOP: Intraocular pressure; M: Month; NA: Not available
Our study did not identify any factors significantly associated with surgery success. However, we observed a trend suggesting that a day-1 IOP of > 8 mmHg or the performance of combined surgery (MicroShunt + Phacoemulsification) may reduce the probability of success.
The preoperative IOP of 11 (5.0%) eyes who underwent combined surgery was ≤ 16 mmHg. This means that the main indication in these cases was cataract surgery and, secondarily, the elimination/reduction of ocular hypotensive medication instead of lowering IOP.
Regarding eyes with previous glaucoma surgery, among the 28 eyes that underwent the solo procedure, 14 (50.0%) had undergone a previous glaucoma surgery, whereas 14 (50.0%) had not. The median preoperative IOP was slightly higher in eyes that did not undergo previous glaucoma surgery, but the difference was not statistically significant (Hodges–Lehmann median difference: 2.0 mmHg; 95% CI: −2.0 to 4.0 mmHg, P = 0.2856). Similarly, there were no significant differences in month-18 IOP between the two groups (Hodges–Lehmann median difference: 0.0 mmHg; 95% CI: −2.0 to 2.0 mmHg, P = 0.9443) or in the median IOP lowering from preoperative values (Hodges–Lehmann median difference: 0.0 mmHg; 95% CI: −4.0 to 3.0 mmHg, P = 0.7636).
In terms of success rates, 14 (100%) eyes without previous glaucoma surgery and 12 (85.7%) eyes with previous glaucoma surgery were classified as successful (either complete or qualified) (P = 0.1493). In addition, no significant differences were observed in either preoperative or month-18 best-corrected visual acuity (BCVA) between both groups.
These results are consistent with those published by Quaranta et al.,[21] who reported that the Preserflo™ MicroShunt can be effectively and safely used in eyes with a single failed trabeculectomy. It is important to note that our study included patients with different types of glaucoma, including 11 eyes with POAG and three eyes with chronic angle-closure glaucoma (CACG), whereas Quaranta et al.’s study focused on patients with POAG.
These findings support the hypothesis that MicroShunt + Ologen implantation may serve as a safe and effective alternative in patients who have previously undergone glaucoma surgery, presenting new possibilities for its application in clinical practice. However, despite the promising results, further studies are necessary to thoroughly analyze this option.
Previous studies have indicated that the dose and use of MMC can have a positive effect on surgical success.[16,17,18,19] Higher MMC doses have been associated with a lower risk of MicroShunt failure.[19] In our study, however, MMC dose was not found to be a predictor of surgery success. Furthermore, there were no significant differences in IOP lowering between MMC 0.02% and MMC 0.04% at 18 months (Hodges–Lehmann median difference: 0.0 mmHg; 95% CI: −3.0 to 4.0 mmHg, P = 0.8986). This could be due to variability in the application time or the fact that only 10 eyes received MMC 0.02%.
Study limitations
The study has certain limitations. First, its retrospective design introduces the possibility of selection bias and confounding factors, which are inherent to such studies. Nevertheless, the use of strict inclusion and exclusion criteria, as well as a large number of eyes, helps mitigate these concerns. The absence of a control group is another limitation. However, various subanalyses were performed (solo procedure vs. combined procedure and previous glaucoma surgery vs. primary surgery), providing valuable clinical information. It is also important to note that nine (18.8%) have been diagnosed with CACG. Among them, 6 eyes underwent a solo procedure because they had previously undergone cataract surgery, while 3 eyes underwent combined surgery (MicroShunt + Phaco) to facilitate the opening of the chamber angle.[26]
The lack of difference in success rates and IOP lowering between different MMC doses may be attributed to the heterogeneity of our study sample or the limited number of eyes in the 0.02% MMC group. Therefore, further studies will be necessary to elucidate the role of MMC dose and application time on clinical outcomes.
Conclusions
The findings of this study suggest that the use of the Preserflo™ MicroShunt implant, either alone or in combination with cataract surgery, in conjunction with the Ologen™ implant, leads to a significant reduction in preoperative IOP and a decreased reliance on ocular hypotensive drugs in patients with glaucoma. In addition, this technique has shown favorable clinical outcomes in eyes with a history of previous glaucoma surgery. Although no factors were found to be significantly associated with surgery success in our study, further research is needed to validate or revise this finding. Importantly, no unexpected adverse events were observed in our study.
Ethical approval
“All procedures performed in studies involving human participants were in accordance with the ethical standards of the Ethics Committee of the Hospital Virgen de la Luz and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards”.
Informed consent
“Due to the characteristics of the study, the Ethics Committee of the Hospital Virgen de la Luz waived the need for written informed consent.” Nevertheless, all the patients signed a written informed consent for their surgery.
Data sharing statement
Data not published here are obtainable on reasonable request from the corresponding author.
Financial support and sponsorship:
Support for Editorial assistance was funded by Santen. It should be noted that Santen was not involved in the preparation of the manuscript nor did the company influence in any way the scientific conclusions reached.
Conflicts of interest:
There are no conflicts of interest.
Acknowledgments
Medical writing and Editorial assistant services have been provided by Antonio Martínez (MD) of Ciencia y Deporte S.L.
References
- 1.European Glaucoma Society Terminology and Guidelines for Glaucoma, 4th Edition—Chapter 3: Treatment principles and options. Supported by the EGS Foundation: Part 1: Foreword; Introduction; Glossary; Chapter 3 Treatment principles and options. Br J Ophthalmol. 2017;101:130–95. doi: 10.1136/bjophthalmol-2016-EGSguideline.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Joshi AB, Parrish RK, 2nd, Feuer WF. 2002 survey of the American Glaucoma Society: Practice preferences for glaucoma surgery and antifibrotic use. J Glaucoma. 2005;14:172–4. doi: 10.1097/01.ijg.0000151684.12033.4d. [DOI] [PubMed] [Google Scholar]
- 3.Saheb H, Ahmed II. Micro-invasive glaucoma surgery: current perspectives and future directions. Curr Opin Ophthalmol. 2012;23:96–104. doi: 10.1097/ICU.0b013e32834ff1e7. [DOI] [PubMed] [Google Scholar]
- 4.Jampel HD, Musch DC, Gillespie BW, Lichter PR, Wright MM, Guire KE, et al. Perioperative complications of trabeculectomy in the collaborative initial glaucoma treatment study (CIGTS) Am J Ophthalmol. 2005;140:16–22. doi: 10.1016/j.ajo.2005.02.013. [DOI] [PubMed] [Google Scholar]
- 5.Gedde SJ, Feuer WJ, Lim KS, Barton K, Goyal S, Ahmed IIK, et al. Treatment outcomes in the primary tube versus trabeculectomy study after 3 years of follow-up. Ophthalmology. 2020;127:333–45. doi: 10.1016/j.ophtha.2019.10.002. [DOI] [PubMed] [Google Scholar]
- 6.Holland LJ, Kirwan JF, Mercieca KJ. Effect of COVID-19 pandemic on glaucoma surgical practices in the UK. Br J Ophthalmol. 2022;106:1406–10. doi: 10.1136/bjophthalmol-2021-319062. [DOI] [PubMed] [Google Scholar]
- 7.Sadruddin O, Pinchuk L, Angeles R, Palmberg P. Ab externo implantation of the MicroShunt, a poly (styrene- block-isobutylene- block-styrene) surgical device for the treatment of primary open-angle glaucoma: A review. Eye Vis (Lond) 2019;6:36.. doi: 10.1186/s40662-019-0162-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.He M, Wang W, Zhang X, Huang W. Ologen implant versus mitomycin C for trabeculectomy: A systematic review and meta-analysis. PLoS One. 2014;9:e85782.. doi: 10.1371/journal.pone.0085782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sastre-Ibáñez M, Cabarga C, Canut MI, Pérez-Bartolomé F, Urcelay-Segura JL, Cordero-Ros R, et al. Efficacy of Ologen matrix implant in Ahmed glaucoma valve implantation. Sci Rep. 2019;9:3178.. doi: 10.1038/s41598-019-38656-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jacobson A, Rojas C, Bohnsack BL. Ologen augmentation of Ahmed glaucoma drainage devices in pediatric glaucomas. BMC Ophthalmol. 2021;21:72.. doi: 10.1186/s12886-021-01827-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vastardis I, Fili S, Perdikakis G, Kontopoulou K, Balidis M, Gatzioufas Z, et al. Preliminary results of Preserflo Microshunt versus Preserflo Microshunt and Ologen implantation. Eye Vis (Lond) 2021;8:33.. doi: 10.1186/s40662-021-00253-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ibarz-Barberá M, Morales-Fernández L, Corroto-Cuadrado A, Martinez-Galdón F, Tañá-Rivero P, Gómez de Liaño R, et al. Corneal endothelial cell loss after PRESERFLO™ MicroShunt implantation in the anterior chamber: Anterior segment OCT tube location as a risk factor. Ophthalmol Ther. 2022;11:293–310. doi: 10.1007/s40123-021-00428-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lang KM, Little TD. Principled missing data treatments. Prev Sci. 2018;19:284–94. doi: 10.1007/s11121-016-0644-5. [DOI] [PubMed] [Google Scholar]
- 14.Bar-David L, Blumenthal EZ. Evolution of glaucoma surgery in the last 25 years. Rambam Maimonides Med J. 2018;9:e0024. doi: 10.5041/RMMJ.10345. doi: 10.5041/RMMJ.10345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Riss I, Batlle J, Pinchuk L, Kato YP, Weber BA, Parel JM. Résultats à un an de l’efficacité et de l’innocuité du MicroShunt InnFocus selon l’emplacement et la concentration de MMC [One-year results on the safety and efficacy of the InnFocus MicroShunt™ depending on placement and concentration of mitomycin C] J Fr Ophtalmol. 2015;38:855–60. doi: 10.1016/j.jfo.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 16.Batlle JF, Fantes F, Riss I, Pinchuk L, Alburquerque R, Kato YP, et al. Three-Year follow-up of a novel aqueous humor MicroShunt. J Glaucoma. 2016;25:e58–65. doi: 10.1097/IJG.0000000000000368. [DOI] [PubMed] [Google Scholar]
- 17.Scheres LMJ, Kujovic-Aleksov S, Ramdas WD, de Crom RMPC, Roelofs LCG, Berendschot TTJM, et al. XEN<sup>®</sup>Gel Stent compared to PRESERFLO™ MicroShunt implantation for primary open-angle glaucoma: Two-year results. Acta Ophthalmol. 2021;99:e433–40. doi: 10.1111/aos.14602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Batlle JF, Corona A, Albuquerque R. Long-term results of the PRESERFLO MicroShunt in patients with primary open-angle glaucoma from a single-center nonrandomized study. J Glaucoma. 2021;30:281–6. doi: 10.1097/IJG.0000000000001734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schlenker MB, Durr GM, Michaelov E, Ahmed IIK. Intermediate outcomes of a novel standalone Ab Externo SIBS microshunt with mitomycin C. Am J Ophthalmol. 2020;215:141–53. doi: 10.1016/j.ajo.2020.02.020. [DOI] [PubMed] [Google Scholar]
- 20.Martínez-de-la-Casa JM, Saenz-Francés F, Morales-Fernandez L, Perucho L, Mendez C, Fernandez-Vidal A, et al. Clinical outcomes of combined Preserflo Microshunt implantation and cataract surgery in open-angle glaucoma patients. Sci Rep. 2021;11:15600.. doi: 10.1038/s41598-021-95217-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Quaranta L, Micheletti E, Carassa R, Bruttini C, Fausto R, Katsanos A, et al. Efficacy and safety of PreserFlo® MicroShunt after a failed trabeculectomy in eyes with primary open-angle glaucoma: A retrospective study. Adv Ther. 2021;38:4403–12. doi: 10.1007/s12325-021-01811-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fea AM, Laffi GL, Martini E, Economou MA, Caselgrandi P, Sacchi M, et al. Effectiveness of MicroShunt in patients with primary open-angle and pseudoexfoliative glaucoma: A Retrospective European multicenter study. Ophthalmol Glaucoma. 2022;5:210–8. doi: 10.1016/j.ogla.2021.08.005. [DOI] [PubMed] [Google Scholar]
- 23.Beckers HJM, Aptel F, Webers CAB, Bluwol E, Martínez-de-la-Casa JM, García-Feijoó J, et al. Safety and effectiveness of the PRESERFLO® MicroShunt in primary open-angle glaucoma: Results from a 2-year multicenter study. Ophthalmol Glaucoma. 2022;5:195–209. doi: 10.1016/j.ogla.2021.07.008. [DOI] [PubMed] [Google Scholar]
- 24.Ibarz Barberá M, Martínez-Galdón F, Caballero-Magro E, Rodríguez-Piñero M, Tañá-Rivero P. Efficacy and safety of the Preserflo microshunt with mitomycin C for the treatment of open angle glaucoma. J Glaucoma. 2022;31:557–66. doi: 10.1097/IJG.0000000000002052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bhayani R, Martínez de la Casa JM, Figus M, Klabe K, Rabiolo A, Mercieca K. Short-term safety and efficacy of Preserflo™ Microshunt in glaucoma patients: A multicentre retrospective cohort study. Eye (Lond) 2023;37:644–9. doi: 10.1038/s41433-022-01995-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Azuara-Blanco A, Burr J, Ramsay C, Cooper D, Foster PJ, Friedman DS, et al. Effectiveness of early lens extraction for the treatment of primary angle-closure glaucoma (EAGLE): A randomised controlled trial. Lancet. 2016;388:1389–97. doi: 10.1016/S0140-6736(16)30956-4. [DOI] [PubMed] [Google Scholar]


