Abstract
Aging is the leading risk factor for Alzheimer's disease and other neurodegenerative diseases. We now understand that a breakdown in the neuronal cytoskeleton, mainly underpinned by protein modifications leading to the destabilization of microtubules, is central to the pathogenesis of Alzheimer's disease. This is accompanied by morphological defects across the somatodendritic compartment, axon, and synapse. However, knowledge of what occurs to the microtubule cytoskeleton and morphology of the neuron during physiological aging is comparatively poor. Several recent studies have suggested that there is an age-related increase in the phosphorylation of the key microtubule stabilizing protein tau, a modification, which is known to destabilize the cytoskeleton in Alzheimer's disease. This indicates that the cytoskeleton and potentially other neuronal structures reliant on the cytoskeleton become functionally compromised during normal physiological aging. The current literature shows age-related reductions in synaptic spine density and shifts in synaptic spine conformation which might explain age-related synaptic functional deficits. However, knowledge of what occurs to the microtubular and actin cytoskeleton, with increasing age is extremely limited. When considering the somatodendritic compartment, a regression in dendrites and loss of dendritic length and volume is reported whilst a reduction in soma volume/size is often seen. However, research into cytoskeletal change is limited to a handful of studies demonstrating reductions in and mislocalizations of microtubule-associated proteins with just one study directly exploring the integrity of the microtubules. In the axon, an increase in axonal diameter and age-related appearance of swellings is reported but like the dendrites, just one study investigates the microtubules directly with others reporting loss or mislocalization of microtubule-associated proteins. Though these are the general trends reported, there are clear disparities between model organisms and brain regions that are worthy of further investigation. Additionally, longitudinal studies of neuronal/cytoskeletal aging should also investigate whether these age-related changes contribute not just to vulnerability to disease but also to the decline in nervous system function and behavioral output that all organisms experience. This will highlight the utility, if any, of cytoskeletal fortification for the promotion of healthy neuronal aging and potential protection against age-related neurodegenerative disease. This review seeks to summarize what is currently known about the physiological aging of the neuron and microtubular cytoskeleton in the hope of uncovering mechanisms underpinning age-related risk to disease.
Keywords: age-related changes, aging, cytoskeleton, microtubules, neuronal morphology
Introduction
Age is the primary risk factor for developing neurodegenerative diseases such as Alzheimer's disease (AD). With an ever-increasing aging population, AD is now one of the leading causes of death globally and the 6th leading cause of death in the US, making it vital to understand the early mechanisms of disease (AAR, 2022). Understanding the cellular and molecular drivers of neuronal aging can provide vital insights into how this physiological phenomenon predisposes to the pathological changes that characterize age-related neurodegenerative diseases like AD. Though many aspects of neuronal aging have been investigated, our knowledge of how neuronal morphology changes as we age are not well established. Furthermore, how the neuronal cytoskeleton ages and how changes to the cytoskeleton contribute to age-related changes in neuronal morphology is even less well-known. In the context of disease, we understand that a compromised cytoskeleton contributes to neuronal degeneration; however, within physiological aging we have a poor understanding of how cytoskeletal changes influence the neuron. This review will explore what is currently known about age-related changes to neuronal morphology (in three distinct neuronal compartments) and the neuronal cytoskeleton, how these two elements relate to one another, and how knowledge of the mechanisms that underpin neuronal aging can help to promote healthy brain aging.
Search Strategy
The papers reviewed here are centered around age-related changes to the morphology of the neuron in each of the distinct neuronal compartments and additionally what age-related changes might occur to the cytoskeleton to potentially influence such morphological changes. PubMed and Web of Science (WOS) online databases were used to search for articles used in this review. Search terms used in PubMed database were: Aging, neuron, morphology, axon, synapse, cell body, dendrite, cytoskeleton, microtubules, actin, Tau, Tau-phosphorylation. Searches were targeted towards papers published between 2018 and 2023. However, literature searches highlighted a large body of important articles that were published before these dates that describe age-related changes to neurons. Combined search terms were used in the WOS database to compliment PubMed searches. Aging (AND) neuronal morphology, aging (AND) microtubules, aging (AND) cytoskeleton, aging (AND) axon morphology, aging (AND) dendrite morphology, aging (AND) cell body morphology, aging (AND) synaptic morphology, aging (AND) tau, aging (AND Tau phosphorylation search terms were used. Once again, literature searches were targeted to the years of 2018–2023. However, further analysis of older articles revealed important papers prior to the initial search dates. All literature was searched for between May 2022 and July 2023.
Anatomy of the Neuronal Cytoskeleton
The neuronal cytoskeleton is a multiplexed structure comprising of three major components: tubulin containing microtubules, neurofilaments (intermediate filaments), and actin containing microfilaments. Each component has a very distinct structure specialized to sustain important neurobiological functions ranging from coordination of proliferation, migration, and differentiation of developing neurons, provision of intracellular structural and mechanical support, supplying tracks for intracellular transport including organelle targeting and facilitating signal transduction in sub-cellular compartments in mature neurons. Due to its importance in disease and physiology, the microtubule cytoskeleton will be the focus of this review. However, both neurofilaments and actin also form important components of the cytoskeleton and are described briefly in this section.
Microtubules
Microtubules (MTs) are the largest of the cytoskeletal components with polarized elongated hollow tubules formed from dimers of α and β-tubulin (25 nm across) that sit parallel within axons and dendrites (Bryan and Wilson, 1971; Baas and Lin, 2011). The polarized nature of the MTs dictates their intra-neuronal orientation, with axonal microtubules arranged with their plus ends pointing outwards, away from the soma, in both vertebrates and invertebrates (Baas et al., 1988). However, in the proximal dendrites of vertebrate neurons, nearly 50% of the MTs were arranged with plus ends pointing away from the soma with the remaining MTs oriented with the plus ends point towards the soma (Baas et al., 1988). In contrast, most of the MTs are oriented with their minus ends pointing away from the soma in invertebrate neurons, though some mixed orientations have also been reported (Stone et al., 2008). This specific orientation and organization of MTs enable the microtubular cytoskeleton to subserve an important function, that of compartment-specific sorting and transportation of proteins and cargo either to dendrites or axons. A key contributor to intra-neuronal protein and organelle sorting, and therefore functional compartmentalization of the neuron is the axon initial segment (AIS). The MT cytoskeleton displays several unique features in this important structure (Eichel and Shen, 2022) that may play a role in enabling the AIS to act as a gatekeeper for axon-specific cargo (Kapitein and Hoogenraad, 2015).
Another characteristic feature of MTs is that they undergo “dynamic instability”, which is the growth and catastrophe of the microtubule labile domain, mostly at the MT plus end, caused by the hydrolysis of stable GTP bound β-tubulin to volatile GDP-bound units (Mitchison and Kirschner, 1984). MT dynamics are regulated by several MT-related proteins including +end tracking proteins, end binding proteins, microtubule-associated proteins (MAPs) including Tau, MAP2, MAP1B, MAP1C amongst others (Sayas and Ávila, 2014; Penazzi et al., 2016). Synchronization of MT dynamics (destabilization and stability) by such microtubule-related proteins plays a key role in the morphological changes that underlie neuronal differentiation and axonal guidance during neuronal development (Kapitein and Hoogenraad, 2015). Post-translational modifications of tubulin itself as well as the MAPs that bind to the tubulin also play a role in regulating MT dynamics and stabilization, ultimately shaping the MT cytoskeleton.
Amongst the many MT interacting proteins, one key MAP which will be described to a greater extent in this review, because of its role in age-related neurodegenerative diseases, is Tau. Tau is a phospho-protein, expressed as six alternatively spliced isoforms (although there are now suggestions of more isoforms), all of which bind to microtubules between tubulin-heterodimers via 3 or 4 microtubule-binding domains in a phosphorylation dependent manner (Corsi et al., 2022). Tau binding to MT promotes suppression of microtubule shortening and reduces the concentration of tubulin required for polymerization (Weingarten et al., 1975; Panda et al., 1995; Kadavath et al., 2015). Tau condensates (envelopes) preferentially form on stabilized microtubule lattices with specific lattice architecture, shortening the distance between tubulin heterodimers, and creating permeable barriers to the binding of other MAPs as well as regulating motor protein motility (Tan et al., 2019; Siahaan et al., 2022). Recent studies suggest that Tau preferentially interacts with the microtubule labile domain instead of the entire structure (Qiang et al., 2018); however, it is likely that it binds along the entire microtubule but holds specificity in aiding polymerization at the labile domain (Barbier et al., 2019). As described in later sections of this review, Tau is particularly intriguing from an age-related perspective because there is evidence of age and disease-related changes in Tau localization and MT binding, which may contribute to some of the age-related changes in neuronal and microtubular structure and function.
Though a lot is known about how MT cytoskeleton changes during development and how this underpins key developmental processes, relatively little is understood about how this important neuronal structure changes during physiological aging. This is surprising given that in age-related neurodegenerative diseases like AD, there is evidence for structural and functional changes in both the MT network itself and in MAPs like Tau. This review therefore seeks to highlight our gaps in knowledge in this area and potentially provide some causal links between MT cytoskeletal aging and vulnerability to age-related neurodegenerative disease.
Neurofilaments and actin
Neurofilaments and actin similarly contribute to cytoskeletal function within neurons. Neurofilaments form 10 nm wide filaments from Neurofilament- heavy, medium, and light chains that aid in neuronal stability (particularly within axons), and in the promotion of axon growth (Yuan et al., 2012). Like microtubules, they can be subjected to post-translation modifications such as phosphorylation and are known to be involved in neurodegeneration (Didonna and Opal, 2019). However, little work has been done on age-related changes to neurofilaments although a large body of biomarker literature has shown increases in certain NF chains within blood serum and CSF with age and in disease, potentially reflecting age or disease-related neuronal death (Kuhle et al., 2011; Vågberg et al., 2015; Idland et al., 2017; Khalil et al., 2020; Nyberg et al., 2020). Like microtubules and neurofilaments, actin is also found within the axon. Actin is the smallest of the three cytoskeletal components (7 nm diameter) and exists in both a monomeric and filamentous state (F-actin) (Dominguez and Holmes, 2011). F-actin is found in abundance within the dendrites and synapses to help in the organization of the presynaptic site, axon initial segment organization, and dendritic plasticity (Konietzny et al., 2017). The use of ultrastructural imaging, stochastic optical reconstruction microscopy has revealed that spectrin-associated actin forms “ring-like structures” periodically along the length of the axon (every 180 nm) in a structure termed the “periodic-actin-cytoskeleton” which is thought to contribute to axonal fortification (Xu et al., 2013). Although there are reports of disease-related changes to actin, little has been reported about the actin cytoskeleton in an aging neuron (Lai and Wong, 2020).
This review will address what is known about changes to the morphology of the synapse, somatodendritic compartment, and axon with physiological aging. Alongside this, a description of what is known about age-related changes to the microtubular cytoskeleton will be discussed as a possible mechanism to explain the age-related morphological changes in the different neuronal compartments. The literature used in this review is summarized in Additional Table 1.
Additional Table 1.
Summary of age-related morphological and cytoskeletal changes covered in this review
| Neuronal compartment | Summary of age-related changes |
|---|---|
| Dendrites | Morphological changes |
|
Changes in dendrite length A 13% reduction in layer 3 pyramidal cells, 37% reduction in level 5 basal dendrites of humans (Nakamura et al., 1984; Nakamura et al., 1985b), and 83% reduction in rodent dentate gyrus dendrite length (Wang et al., 2017) and dendrite length within rodent hippocampus (Aguilar-Hernández et al., 2020). Dendrite length reduction is also reported in PFC dendrites of monkeys and rodents (Kabaso et al., 2009; Sotoudeh et al., 2020). However, no dendritic regression is seen in the human prefrontal cortex (de Brabander et al., 1998), monkey layer 3 pyramidal neurons (Duan et al., 2003), or superior temporal cortex pyramidal dendrites (Luebke et al., 2015). Conversely, dendrite length is seen to increase in the human parahippocampal gyrus (Buell and Coleman, 1979) and rodent amygdala (Sotoudeh et al., 2020). |
|
|
Reduction in dendrite volume/diameter Dendrite surface area/volume is reduced in long projecting PFC dendrites (Kabaso et al., 2009)) and diameter is reduced in lumbar spinal cord motor neuron dendrites (Castro et al., 2023). |
|
|
Reductions in dendrite branching/space A 50% reduction in dendritic branching is seen in rodent dentate gyrus neurons (Wang et al., 2017) and a 20% reduction in synaptic coverage of dendrites in rodent spinal cord motor neurons (Castro et al., 2023). |
|
| Cytoskeletal changes | |
|
Changes to microtubule integrity Up to a 55% reduction in microtubule density between the ages of 62 and 80 in humans (Cash et al., 2003). |
|
|
Reduction in tubulin CacyBP/SIP and beta-tubulin staining reduces with age in rodents (Filipek et al., 2008). |
|
|
Reductions in Tau Phosphorylated tau (Niewiadomska and Baksalerska-Pazera, 2003; Niewiadomska et al., 2005; Filipek et al., 2008) and non- phosphorylated tau (Niewiadomska, 2003) staining is reduced in processes (some thought to be dendrites) with age in the rodent hippocampus and cortex. |
|
|
Reductions in MAP2 MAP2 immuno-staining is reduced in dendritic processes in the rodent dentate gyrus and CA1 (Di Stefano et al., 2001; Di Stefano et al., 2006). |
|
| Soma | Morphological changes |
|
Changes to soma size/volume A 33% reduction in soma volume reported in human super frontal gyrus cells between the ages of 52 and 94 years (Uemura and Hartmann, 1978), soma size reduces in the cerebral cortex and PFC of rodents (Ueno et al., 2018; Sotoudeh et al., 2020) and spinal motor neurons of cats (Liu et al., 1996). Additionally, soma size decreases by 30% in touch receptor neurons between 1 and 8 days of age (Hess et al., 2019). Conversely, soma size remains consistent in human and rodent spinal cord motor neurons (Castro et al., 2023), monkey visual cortex neurons (Luebke et al., 2015) and rodent amygdala (Sotoudeh et al., 2020). |
|
|
Appearance of lipofuscin granules Lipofuscin granules are seen to accumulate in many regions of the aged brain (Brody, 1960; Robles, 1978; Gray and Woulfe, 2005; Pannese, 2011; Merlo et al., 2015) although this is not reported in hypothalamic neurons (Brody, 1970). |
|
| Cytoskeletal changes | |
|
Increases in staining for cytoskeletal proteins in the soma Usually found to be an axonal MAP, both tau and phosphorylated tau are shown to mislocalize to the soma in rodents whilst soma tubulin staining is reduced (Filipek, Schneider 2008, Niewadomska 2003, 2006). |
|
| Axon | Morphological changes |
|
Changes to axonal diameter A 1.5-1.6 fold increase in axon diameter is seen in human male skin sensory neurons (Metzner et al., 2022)) and an increase in axonal diameter index is shown in the corpus callosum of aged humans (Fan et al., 2019)) whilst rodent optic nerve axon volume increases by 50% and diameter increases between 1 and 12 months (Stahon et al., 2016). However, no change in axon diameter is seen in the corpus callosum, cingulate gyrus, and visual cortex of monkeys (Peters et al., 2001; Bowley et al., 2010). |
|
|
Appearance of axonal swellings “Torpedo” axonal swellings are seen in aged human (Kato and Hirano, 1985) and rodent (Bäurle and Grüsser-Cornehls, 1994) Purkinje cells. Thickened fibers and ballooned terminals are also seen in the anterior cingulate cortex, entorhinal cortex, parietal lobe, and temporal gyrus of aged humans (Geula et al., 2008). |
|
| Cytoskeletal changes | |
|
Changes to microtubule integrity In the aged human retina, up to 30% of rods have microtubules with altered morphology such as non-uniformed alignments and wavy appearances (Nag et al., 2020). |
|
|
Changes to microtubule staining Staining for tubulin, tau, and P-tau (phospho-tau) are all reduced within processes of aged rodents (Filipek 2008, Niewiadomska 2003, 2005) but NFH, tubulin and actin are shown to increase within human sensory axons with age (Metzner et al., 2022). |
|
|
Changes to axonal transport A reduction in axonal transport is reported in the CNS and PNS of rodents (Niewiadomska and Baksalerska-Pazera, 2003; Milde et al., |
|
| 2015; Takihara et al., 2015) and the Drosophila wing sensory neurons (Vagnoni et al., 2016). | |
| Synapses | Morphological changes |
|
Reductions in spine density In humans, spine density/volume in pyramidal neurons and the frontal cortex does not change into old age (Jacobs et al., 1997; Scheff et al., 2001) although this is argued as more recent work shows an age-related reduction in spine density within the human prefrontal cortex (Boros et al., 2019). In rodents, overall spine count/density is lost in multiple regions (Bloss et al., 2011; Mostany et al., 2013; Calì et al., 2018; Aguilar-Hernández et al., 2020). In monkeys, a reduction of spine density is also observed in the prefrontal and visual cortices (Kabaso et al., 2009; Dumitriu et al., 2010; Young et al., 2014; Luebke et al., 2015). |
|
|
Change to spine type In rodents a loss of thin and stubby spines is widely documented (Bloss et al., 2011; Mostany et al., 2013) but conversely, an increase in stubby spines (Aguilar-Hernández et al., 2020) and 13% increase in spine head volume is also reported (Chen and Hillman, 1999). In monkeys, a loss of thin spines alongside reductions in spine density is observed in the prefrontal and visual cortices (Kabaso et al., 2009; Dumitriu et al., 2010; Young et al., 2014; Luebke et al., 2015). |
|
| Cytoskeletal changes | |
| • Reduced membrane-association of Rho GTPases in mice (critical to actin remodeling) (Afshordel et al., 2014); • Reduced synaptic levels of actin and synaptophysin in rodents (Ve et al., 2020); • Reduction and increased inactivation of cofilin in the aging mouse brain (cofilin is important for actin remodeling) (Barone et al., 2014). |
|
| Biochemical changes to MT cytoskeletal components (not clear what compartment as difficult to determine this from biochemical fractions) |
Tubulin levels Soluble tubulin decreases by up to 75% in aged human cortex between the ages of 0-90 years (Yan et al., 1985), decreases also shown by (Labisso et al., 2018). In rodents, there is evidence for either a reduction (Shimada et al., 2006) or a trend for a decrease (Fifkova and Morales, 1992; Kneynsberg and Kanaan, 2017) in tubulin. |
| Tubulin PTMs Age-related decreases in acetylated tubulin (suggesting decreased MT integrity) reported by some in human studies (Larrayoz et al., 2017); no change reported by others in rodents (Larrayoz et al., 2017). Interestingly, some report increases in acetylated tubulin in rodents (Marton et al., 2010). | |
| MAP2 levels Reduction in MAP2 in human retina (Nag et al., 2020) and rodent brain (Chauhan and Siegel, 1997; Di Stefano et al., 2001; Himeda et al., 2005). | |
| MAP1 and 5 levels MAP1 and MAP5 reduced in rodent hippocampus (Chauhan and Siegel 1997). | |
| Total Tau levels | |
| Shown to decrease with age in humans (Mukaetova-Ladinska et al., 1996). | |
| Biochemical changes to MT cytoskeletal components (not clear what compartment as difficult to determine this from biochemical fractions) |
P-Tau levels Phosphoproteomic studies show an increase in phosphorylated cytoskeletal proteins (Andrés-Benito et al., 2023); significant increases shown in rodents (Larrayoz et al., 2017). Evidence for increases in monkeys (Datta et al., 2021; Leslie et al., 2021). |
| Overall neuronal loss: | No more than 10% overall neuronal loss in healthy physiological aging (Pannese, 2011) though there are regional differences with cerebellar Purkinje cells more vulnerable and showing up to 40% neuronal loss in some regions (Zhang et al., 2010). |
CNS: Central nervous system; MAP: microtubule associated protein; MTs: microtubules; NFH: neurofilament-heavy; PFC: pre-frontal cortex; PNS: peripheral nervous system; P- Tau: phosphorylated-Tau.
Age-Related Synaptic Changes
The structure and function of the synapse
The human brain contains trillions of synapses which are formed in excess during development before being pruned as functional networks develop. Following development, the majority of synaptic connections are relatively stable but new connections can form, or be abolished, in response to experience (Ackerman, 1992). Moreover, individual synaptic strength is modulated (termed synaptic plasticity) allowing for the high storage and working capacity of our brains that underlies our everyday function (Bartol et al., 2015). Chemical synapses form the connections between neurons that are dedicated to fast, highly regulated directional information transfer that underpins the functioning of our brains. They are the basic units that enable neuronal network function and consist of a presynaptic site that contains the conserved neurotransmitter release machinery and a postsynaptic site containing arrays of neurotransmitter receptors. The two sites are connected by a series of cell adhesion molecules, which ensures trans-synaptic juxtaposition of release and detection machineries and the intracellular organization of the synapses. Actin contributes to pre- and postsynaptic function in both membrane trafficking and structural organization. A sustained increase in synaptic strength critically requires postsynaptic actin remodeling. In this regard, postsynaptic spine structure is indicative of stable versus transient connectivity and the importance of the cytoskeleton in synaptic structure and function is highlighted (Kasai et al., 2003).
Age-related change in synaptic morphology and function
Compared with other neuronal compartments more is known about the aging of synapses, perhaps due to their vital importance in cellular connectivity. As described here, there are two key changes noted with age: loss of spine density and selective loss of spine types (Figure 1). Within the human prefrontal cortex, Golgi impregnation revealed that the spine density of pyramidal neurons decreases around the end of the fourth decade of life, but appears to remain relatively stable thereafter (Jacobs et al., 1997). Additionally, an electron microscopy-based approach has suggested that the volume density of spines in the human frontal cortex remains constant across ages (Scheff et al., 2001). In line with this, a global view of the human brain using PET (positron emission tomography) tracer labeling of the synaptic vesicle (glycoprotein 2A-SV2A) shows no synaptic loss associated with aging across the human brain (Michiels et al., 2021). However, 3D morphological analysis of synapses of the prefrontal cortex in cognitively normal humans shows an age-related reduction in spine density (Boros et al., 2019). Specifically, thin and stubby spines are lost, leading to an overall lower spine count across apical and basal dendritic trees whilst the density of the more stable mushroom spines is retained (Figure 1; Boros et al., 2019). This is also seen in the rat prefrontal cortex, where spine morphology and density change with aging and are accompanied by a loss in experience-dependent plasticity (Bloss et al., 2011).
Figure 1.
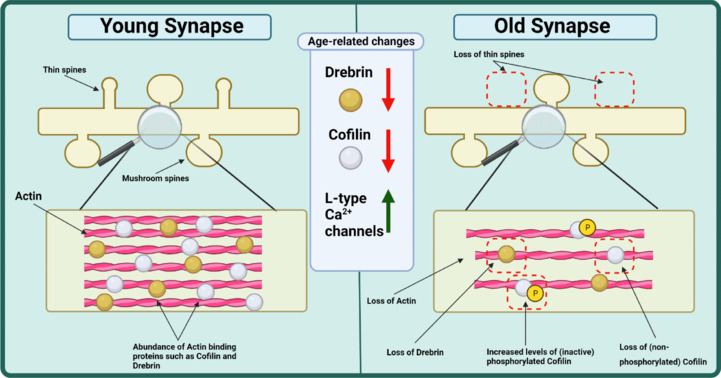
Graphical summary of age-related changes that occur to the morphology of synaptic spines.
Research shows a general decrease in density of synaptic spines with age which is often characterized by the selective loss of thin spine types. Though it has not been studied extensively, the aging cytoskeleton within the synapse sees decreases in actin itself alongside actin binding proteins Drebrin and Cofilin. This is accompanied by an increase in the non-functional phosphorylated cofilin. Functionally, there is an increase in L-type calcium channels with age. White circles show cofilin proteins whilst brown circles represent Drebrin. Yellow (P) symbols represent phosphorylation. Created with BioRender.com.
Other studies also report the loss of dendritic spines in aging rodents, within the hippocampus, but also show an increase in stubby spines (Aguilar-Hernández et al., 2020). A 16.5% loss of synaptic density is similarly documented in the mouse somatosensory cortex (Calì et al., 2018). Within the somatosensory cortex of aged rodents, a loss of thin spine types and a reduction in spine volume is also reported (Mostany et al., 2013). Interestingly in the Purkinje cells of aging rodents, although a 30% reduction in synapses is seen, a 13% increase in spine head volume is documented (Chen and Hillman, 1999). The consequence of these reported changes in rodents is potentially demonstrated in the aging rodent cortex, where a loss of spine density correlates with reduced calcium activity and motor learning ability (Huang et al., 2020). The selective loss of thin spines and reduction in spine density is also observed within the prefrontal cortex and visual cortex of aged monkeys (Kabaso et al., 2009; Dumitriu et al., 2010; Young et al., 2014; Luebke et al., 2015). This is suggestive of a loss of synaptic plasticity with aging, which may be accompanied by some reduction in synaptic number.
In line with changes in synaptic plasticity, long-term potentiation induction and maintenance deficits are observed in the hippocampus of aged rats (Landfield et al., 1978; Barnes, 2003). Experiments in rodents have highlighted a shift in the regulation of calcium homeostasis with aging. Specifically, aged rats show an upregulation of L-type Ca2+ channels, which is correlated with poorer working memory (Figure 1; Thibault and Landfield, 1996). In the prefrontal cortex of monkeys, reduced firing in aged animals could be restored by inhibiting cAMP-PKA signaling or by blockers of select potassium channels (Wang et al., 2011). The efficiency of protein degradation pathways also declines with aging (Kuijpers, 2022). Indeed, the degradative capacity of late endocytic and lysosomal organelles declines with aging, possibly due to lower vATPase levels, and the less effective organelles accumulating at synaptic sites (Burrinha et al., 2023). The impairment of proteostasis particularly affects the presynaptic sites, where rapid membrane fusion and recycling of the fusion machinery is key to sustained synaptic transmission. Accordingly, disruption of presynaptic homeostasis leads to use-dependent synaptic loss (Fernández-Chacón et al., 2004). This cellular and molecular evidence for reduced function within synapses, alongside anecdotal/primary evidence of age-related decline in memory, suggests that age-related morphological changes to synapses (documented here) are correlated with functional change. As the synapses rely upon a proficient cytoskeleton for both structure and function (particularly actin), could age-related cytoskeletal decline contribute towards the phenotypes documented here?
Age-related change in the synaptic cytoskeleton
As discussed above, actin rearrangements are central to post-synaptic structural plasticity. The Rho family GTPases play a critical role in actin remodeling. Interestingly, in the aging mouse brain, reduced membrane association of these GTPases is detected (Afshordel et al., 2014), potentially underlying the reduced structural plasticity observed in aged rodents. In aged mice, synaptic levels of actin and synaptophysin are reduced while α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor subunits and postsynaptic density protein-95 remain largely unaltered (Ve et al., 2020), further suggesting a decline in release efficiency and structural remodeling within the aging brain prior to/instead of widespread loss. Further evidence in the aging mouse brain comes from reduced levels of actin regulatory proteins drebrin (Hatanpaa et al., 1999) and cofilin that declines age (Figure 1), whilst levels of inactive phospho-cofilin increase (Barone et al., 2014). Changes related to actin dynamics and structure have been covered in studies of non-neuronal cell types (Mack et al., 2016; Higuchi-Sanabria et al., 2018) but as demonstrated here our understanding of actin, its modulators, and effectors in the aging brain/neuron is limited. Moreover, it is difficult to infer if age-related synaptic morphological alterations documented here are due to a change in the integrity and carefully balanced active remodeling process of the cytoskeleton.
Age-Related Changes in the Somatodendritic Compartment
The structure and function of the somatodendritic compartment
Being the housekeeping hub of the neuron, the somatodendritic region represents a vital neuronal compartment. The soma (or cell body) contains the cell's nucleus, housing genetic information and regulating cellular activity. Importantly, it connects to the axon via the AIS, modulating the trafficking of cargo (such as mitochondria) to distal regions of the neuron via the axon. Additionally, the cell body is directly connected to dendritic projections. Dendrites are specialized processes, charged with receiving and processing synaptic information through the activation of post-synaptic elements found on the dendrites (Häusser et al., 2000). As such, dendrites are crucial components of a neuron's communication architecture. As already discussed, dendritic post-synaptic spines see age-related changes but here we discuss morphological changes that are reported to occur within the dendritic shafts and the soma.
Age-related changes in dendrite shaft morphology
It is generally accepted that, with age, there is a loss of and change in the morphology of dendrites (Pannese, 2011). Evidence for changes to dendritic morphology comes from early studies conducted on human brain tissue. Regressive changes are seen in the dendritic length of pyramidal cells in the cortex with age (Figure 2a). However, the extent of this regression in the cortex is layer-dependent. The dendritic field of basal dendrites in layer 3 pyramidal cells in the motor cortex has been found to regress by approximately 13%, whereas layer 5 basal dendrites by 37% (Nakamura et al., 1984; Nakamura et al., 1985a). Others report a more profound reduction with an 76% reduction in dendritic length of dentate gyrus neurons in 12-month-old rodent brains compared to 3 months (Wang et al., 2017). This may indicate that dendrites in certain locations are more susceptible to the effects of aging than others. However, this reduction in dendritic field is not uniform throughout the brain as shown by layer 3 pyramidal neuron dendrites in the prefrontal cortex where regression is not evident whilst dendrite length is seen to increase in the parahippocampal gyrus with age (Buell and Coleman, 1979; de Brabander et al., 1998).
Figure 2.
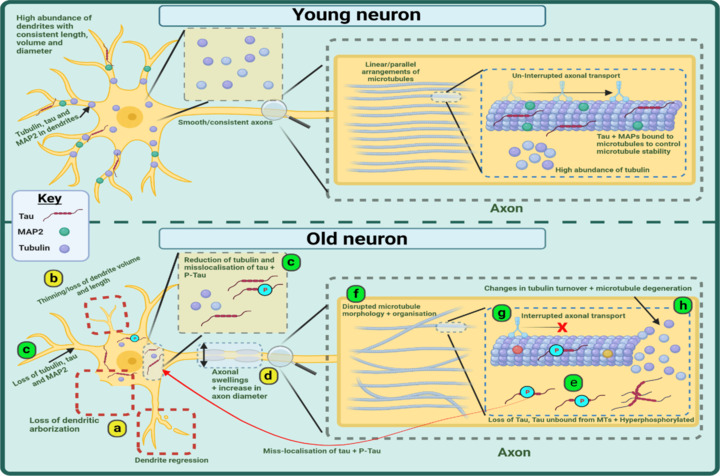
Graphical representation of age-related changes occurring to the morphology of the somatodendritic and axonal neuronal compartments and the cytoskeleton.
Evidence shows an age-related loss of and regression to dendrites (a) alongside a thinning and reduction in dendritic volume (b). Within the dendrites there is a loss of tubulin, tau, and MAP2 whilst tubulin is reduced in the soma and tau + P-Tau is mislocalized to the soma (c). In the axon, there can be increases in axonal diameter and the appearance of swellings (d). Normal tau is reduced and phosphorylated tau is increased whilst both also appear to mislocalize to the cell soma (e). EM studies show a disruption in the organization and morphology of microtubules (f) whilst axonal transport is interrupted (g) and GTPase derived tubulin turnover altered (h). Green circles represent MAP2 proteins, Red symbols represent tau proteins, purple circles represent tubulin, and blue (P) symbols represent phosphorylation. Created with BioRender.com. EM: Electron-microscopy; MAP: microtubule-associated protein; MTs: microtubules; p-Tau: phosphorylated tau.
Changes to dendritic morphology are also described in Rhesus monkeys. 3D-morphometric analysis of apical and basal dendritic trees in the prefrontal cortex (PFC) reveals that total dendritic length, surface area, and volume all reduce with age in long projecting neurons whilst remaining unchanged in locally projecting cells (Figure 2b; Kabaso et al., 2009). However, ultrastructural analysis of aged layer 3 pyramidal cells with electron microscopy and retrograde labeling of neurons in the superior temporal cortex pyramidal neurons show no significant age-related change in dendritic length or diameter, although these studies show a trend for reduction in these parameters (Duan et al., 2003; Luebke et al., 2015). In the rodent brain, there are reductions in synaptic coverage of dendrites (20% loss) and dendrite diameter at the most proximal regions within lumbar spinal cord motor neurons (Castro et al., 2023). Additionally, total dendrite length is significantly lower in the pre-frontal cortex but significantly higher in the amygdala of aged rodents (Sotoudeh et al., 2020). Like in humans, there are region-specific differences in the age-related somatodendritic changes. Nonetheless, there appears to be a reduction in dendritic arborisation in aged neurons across the species.
Age-related changes in soma morphology
Several papers have investigated age-related changes to dendrites and their spines, but comparatively fewer studies have interrogated the cell soma. In the cerebellar Purkinje cells, it is generally accepted that there is a shrinkage of the cell soma with age (alongside degeneration and retraction of dendritic arborizations) (Zhang et al., 2010). Beyond these better-studied Purkinje cells, there is also evidence for age-related change. In humans, comparisons of cell somas within the superior frontal gyrus showed a 33% reduction in volume (5721 µm3 to 3551 µm3) between 52 and 94 years (Uemura and Hartmann, 1978). More recently, fluorescent imaging of immuno-stained human cervical spinal cord motor neurons has shown no significant age-related change in soma size (although a trend for an increase in soma size is apparent) (Castro et al., 2023). Similarly, ultrastructural electron-microscopy analysis of the aged rhesus monkey visual cortex shows no significant change in soma length or width with age (Luebke et al., 2015).
Studies on non-primate neurons provide slightly more evidence for age-related changes to soma morphology. In lumbar spinal cord motor neurons and amygdala neurons of rodents, soma size appears unchanged with age (Sotoudeh et al., 2020; Castro et al., 2023). However, in the rodent cerebral cortex, one-third of the parvalbumin-positive neurons display an age-related reduction in soma area whilst soma diameter increases in the PFC (Ueno et al., 2018; Sotoudeh et al., 2020). This region-specific difference in soma morphology is also represented in cats; although both show decreases in soma cross-section, large discrepancies are seen in spinal motor neurons of aged animals (Liu et al., 1996). It must also be noted that soma volume is reported to decrease by 30% with age (between 1 and 8 days) in C. elegans anterior lateral microtubule (touch receptor) neurons with long-living mutants showing a smaller decrease (27.44%), perhaps indicating soma volume to be a marker of health (Hess et al., 2019). Collectively, this literature shows changes in soma volume, width, and height to be brain region-specific. However, more work is needed to truly understand age-related changes to somatodendritic morphology, especially in the human brain. Moreover, an understanding of why changes in morphology occur and what their functional consequences are is unknown. One possibility is that morphological changes arise due to a destabilization of the neuronal cytoskeleton.
As well as these structural changes to the soma, several studies have documented age-related increases in lipofuscin granules in neurons, in both vertebrates and invertebrates (Robles, 1978; Pannese, 2011; Merlo et al., 2015). The cytoplasmic volume of motor neurons in some human centenarians consists of up to 75% lipofuscin granules (Gray and Woulfe, 2005). When visualizing neurons in an aged brain, lipofuscin granules can be found in most types of neurons but are most abundant in motor neurons in the anterior horn of the brainstem and spinal cord, inferior olivary nucleus, dentate nucleus of the cerebellum, the globus pallidus and the dentate nucleus of the cerebellum. Interestingly, lipofuscin accumulation is not seen in hypothalamic neurons (Brody, 1955, 1960). As lipofuscin accumulation occurs in lysosomes, its accumulation in an age-related manner is thought to arise due to age-related deficits in clearance mechanisms (Terman and Brunk, 1998; Kritsilis et al., 2018). Such deficits in clearance are also thought to predispose to age-related proteinopathies (Hardy, 2019).
Age-related cytoskeletal changes within the somatodendritic compartment
Microtubules play a prominent role in maintaining the structural integrity of the somatodendritic compartment. Structural analysis of the cytoskeleton in human cortical pyramidal cell dendrites using electron-microscopy reports up to a 55% reduction in microtubule density between the ages of 62 years and 80 years in non-cognitively impaired individuals (Cash et al., 2003). Rodent studies show an age-related change in the abundance of tubulin and MAPs within the somatodendritic compartment (Figure 2c). Fluorescent imaging of rodent hippocampal and cortical tissue in young (4 months old) vs. old (28 months old) animals shows that several proteins become reduced in neuronal processes with some mislocalizing to the soma with age (Figure 2c). These include CacBP/SIP (which co-localizes within tubulin) and phosphorylated tau (anti P-Tau404) whilst beta-tubulin staining is reduced in dendrites and the soma (Filipek et al., 2008). Another study also showed an age-dependent loss of MAP2 immunoreactivity from the dendritic regions of the dentate gyrus and CA1 region of the rodent brain (Figure 2c; Di Stefano et al., 2001, 2006). Similarly, in the CA3 area of rodent brains, non-phosphorylated tau species (detected by the Tau1 antibody) appear to mislocalize from cellular processes (neuropil) to the cell body between the ages of 4 and 28 months. Phosphorylated tau species (detected by the AT180 antibody) in hippocampal and cortical neurons show a similar pattern across the same time frame (Niewiadomska and Baksalerska-Pazera, 2003; Niewiadomska et al., 2005). However, it should be noted that studies on accelerated aging models (such as Fischer 344 rodents) do not replicate findings from wild-type rodents (Kneynsberg and Kanaan, 2017). Collectively, this limited research begins to show that elements constituting the microtubule cytoskeleton may become lost from dendritic processes with age and exhibit a tendency to accumulate in the cell body. However, it is unknown if the loss of dendritic microtubule proteins is due to a loss of dendritic projections or if a loss of these proteins results in a reduction in dendritic fortification which leads to dendrite loss and morphological changes. Due to the lack of concurrent research on dendrite morphology and cytoskeletal health, it is difficult to infer the relationship between the dendrites and the mislocalization of microtubule proteins reported here.
Age-Related Changes in the Axon
The structure and function of the axon
The axon is vital to the functioning of a healthy neuron. These long projections aid in the propagation of electrical signals to target cells, forming neuronal networks. Mammalian axon diameter can vary vastly from approximately 0.08 µm to 20 µm whilst length can vary between 1 mm and 1 m depending on anatomical location, function, and myelination status although measurements can vary beyond those listed here (Debanne et al., 2011; Muzio and Cascella, 2023). The axon is perhaps the least understood when it comes to age-related morphological changes, despite the fact that axonal dysfunction is implicated in a plethora of neurodegenerative diseases, some of which are age-related (Kanaan et al., 2013; Correale et al., 2019). Outside of neurodegenerative research, two age-related axonal phenotypes that have been documented are changes to axonal diameter and the accumulation of swellings within the axon.
Age-related changes in axon morphology
Depending on anatomical location, differences are seen when comparing axonal diameter in young and old animals Research on peripheral sensory axons found within human skin shows a 1.5-to-1.6-fold increase in axon diameter at proximal and distal locations when comparing biopsies from healthy men aged between 23 years and 79 years old with confocal microscopy (Figure 2d; Metzner et al., 2022). In the central nervous system, a novel MRI-based technique created for measuring axonal diameter and density in human brains in vivo has shown that the axonal diameter index increases within the corpus callosum with age (Fan et al., 2019). However, electron microscopy of axons in the analogous corpus callosum as well as the cingulate gyrus and primary visual cortex of Rhesus monkeys aged 4 years to 35 years old shows no significant difference in average axon diameter, although an increase in axons displaying a degenerative phenotype (darkened axoplasm, vacuoles and dense debris) and increase in myelin sheath diameter is reported (Peters et al., 2001; Bowley et al., 2010). In a more isolated compartment of the central nervous system, post-mortem analysis of the rodent optic nerve using three-dimensional electron microscopy shows a 50% increase in axon volume between the ages of 1 and 12 months with significantly larger axon diameters (0.4–1.6 µm in young vs. 0.5–4.1 µm in aged; Stahon et al., 2016). When comparing studies, it is difficult to draw conclusions due to the sparsity of data and the conflicting nature of the results. It is likely that different regions of the brain have contrasting vulnerabilities to changes in axonal diameter, but more work is needed to fully understand this. The age-related appearance of axonal swellings is another aspect of neuronal morphology that has been reported.
Axonal swellings are known to be a common hallmark of neurodegeneration and are often seen in studies of disease, as demonstrated by transgenic rodents expressing mutant Huntington protein where axonal swellings are found to be early age-related morphological markers preceding other pathologies (Marangoni et al., 2014). There is also evidence that swellings appear throughout aging, as reported in Purkinje cells (Figure 2d). Evidence for this comes from an early study, where the appearance of torpedoes (rounded axonal swellings) in the proximal section of Purkinje axons of aged humans is described (Kato and Hirano, 1985). In rodent Purkinje axons, a similar phenomenon is observed with a 13.7% increase in “torpedo” bearing axons between the ages of 6 to 32 months (Bäurle and Grüsser-Cornehls, 1994). However, a 2009 study by Louis and colleagues disputes that Purkinje cell axon torpedoes do not form as a factor of age but instead a marker of cerebellar injury in humans (Louis et al., 2009). Whether their appearance in age-related injury is due to the axons becoming more vulnerable with increasing age is a matter of debate. Increases in thickened fibers and ballooned terminal have also been reported in cholinergic axons (anterior cingulate cortex, entorhinal cortex, parietal lobe, and temporal gyrus) of non-demented human brains when compared to younger neurons (Geula et al., 2008). However, like changes in axonal diameter, this is an area with a distinct lack of information. More work is needed to understand if axonal swellings universally occur in all neurons, why they occur and what the possible implications of such changes are. Whilst it is likely that there is a cumulative effect of age-related injury manifesting in these age-related changes, there is also a likely contribution of age-related vulnerability to injury.
Are changes in axonal morphology associated with cytoskeletal abnormalities?
The discussed literature indicates possible age-related axonal morphological abnormalities, but it is difficult to deduce a cause for such changes. One possibility is that a change in the integrity of the neuronal cytoskeleton is resulting in a deficit in vital processes like axonal transport inside the axon which manifests as morphological alterations. Several papers do indeed show morphological abnormalities and cytoskeletal aberrations in tandem, and in experimental models where cytoskeletal integrity is compromised (Cowan et al., 2010), there are axonal transport deficits that give rise to disruptions in axonal morphology reminiscent of those reported in aging brains (Mudher et al., 2004). In a previously discussed paper, it was found that changes to the diameter of human sensory neurons are associated with increases in neurofilament-heavy chain immunoreactivity, microtubule mass, and filamentous actin at the protein level and differential expression of cytoskeletal genes when analyzed with transcriptomics (Metzner et al., 2022). In-vitro human neural stem cell models of traumatic brain injury demonstrate axonal swellings that are associated with a dysregulation of the periodic actin cytoskeleton and a change in the spatial organization of tubulin (Pozo Devoto et al., 2022). Similarly, spastin mutations (SPG4 deletion) in cortical cultures have been found to cause axonal swellings that are accompanied by severely interrupted axonal transport, and un-bundled, misoriented/curly microtubules with these phenotypes rescued by supplementation of sub-stoichiometric concentrations of microtubule stabilizing compounds (such as nocodazole; Kasher et al., 2009; Fassier et al., 2013). These studies provide a link between reductions in cytoskeletal integrity and the appearance of axonal swellings, but it is unknown if age-related morphological and cytoskeletal changes correlate. Swellings seen in these studies, their pathology, and cause may differ significantly from swellings seen with age. Research aimed specifically at age-related changes to the microtubule cytoskeleton of the axon might help us to draw comparisons between the cytoskeletal changes and neuronal morphology.
Age-related cytoskeletal changes within the axon
Like the somatodendritic compartment, studies investigating the localization of microtubule proteins in the rodent brain report age-related changes in the abundance of microtubule proteins within neuronal processes to the cell soma (Figure 2c; protein mislocalization) (Niewiadomska and Baksalerska-Pazera, 2003; Niewiadomska et al., 2005; Filipek et al., 2008; Additional Table 1). These findings seem to suggest that tubulin and microtubule-related proteins are reduced within axons with age (Figure 2e). This is opposed by work on human peripheral sensory neurons (discussed above) showing an age-related increase in cytoskeletal proteins (NFH, tubulin, actin) within the axon (Metzner et al., 2022). Although providing information on the localization of cytoskeletal proteins, these studies provide little knowledge of the integrity/morphology of the microtubules.
One way to determine changes in microtubule integrity within the axon is to directly visualize them at high resolution. However, only a handful of studies to date have conducted such research. One paper has observed changes in axonal microtubules within the aging human retina (photoreceptors). Here it was found that up to 30% of rods had an altered morphology of microtubules within the retina by the 8th decade of life whilst 10% exhibited this change by the 6th decade (Figure 2f; Nag et al., 2020). Electron microscopy highlighted that the once linear and parallel arrangement of microtubules became “bendy” or “wavy” in morphology (like electron-microscopy of spastin-disrupted microtubules previously discussed; Fassier et al., 2013), adopting non-uniformed and scattered orientations with age. However, a caveat to these results is that photoreceptors are specialized, meaning comparisons with other neurons should be made carefully. This limited research begins to show that aging might be associated with a change in the morphology of microtubules/microtubule bundles within the axon. However, it is unknown if changes in microtubule integrity during physiological aging result in changes to the morphology of the axon. It is also difficult to draw conclusions on the cytoskeleton within the somatodendritic compartment and axon due to the sparsity of data available. Studies observing functional changes within the axon, linked to cytoskeletal integrity, might provide more clarity as to the state of the cytoskeleton with age. Furthermore, it is not known if there are age-related changes to key axonal structures like the AIS, and whether this is responsible for the loss of neuronal polarity that the mislocalization of proteins implies.
Age-related changes in axonal function (axonal transport)
Evidence for age-related impairments in axonal function, linked to cytoskeletal integrity, comes from studies of axonal transport. Axonal transport is facilitated by interactions between microtubules and motor proteins, age-dependent disruptions to this process have been reported in both rodents and Drosophila (Figure 2g). Rodent studies show that between the ages of 1.5 and 25 months, there are interruptions in both anterograde and retrograde transport when conducting live imaging of tagged vesicles (Nicotinamide nucleotide adenylyltransferase 2-venus and tagged Mitochondria) in neurons from the central nervous system and peripheral nervous system (Milde et al., 2015; Takihara et al., 2015). These experiments report phenotypes such as reductions in average particle velocity, decreases in mitochondria travel length and increases in the total amount of free space not occupied by transported cargo. Additionally, fluorescent imaging of fixed rodent hippocampal and cortex tissue, stained with fluorogold, shows an age-related reduction in axonal transport (Niewiadomska and Baksalerska-Pazera, 2003). Similarly, live imaging of the Drosophila wing sensory neurons has revealed an age-dependent disruption in mitochondrial transport (deficits in transport motility and accumulated cargo), reminiscent of observations made in rodents (Vagnoni et al., 2016).
The studies reported here demonstrate an age-dependent disruption in axonal transport which might arise due to a reduction in microtubule stability. The reliance of axonal transport on a stable microtubule network is shown in Drosophila larvae, where a reduction of microtubule stability via the abolition of Stathmin (a regulatory protein, which disrupts microtubule stability) results in an increase in the aggregation of cargo and reduction in lifespan in comparison to controls (Duncan et al., 2013). However, it is not understood if age-related changes to microtubule integrity influence axonal transport and whether these transport deficits lead to changes in neuronal morphology. Further evidence for age-related cytoskeletal change comes from research on un-specified compartments of the neuron.
Further Evidence for Age-Related Cytoskeletal Changes
Changes to tubulin/MAP concentration
Although not specifying if changes are within the axon, soma, dendrite, or synapse, western blot and low-resolution immunofluorescent studies suggest that microtubules become compromised with age. As shown in Additional Table 1, the consensus is that tubulin is reduced with age within the brains of humans and rodents (Yan et al., 1985; Shimada et al., 2006; Labisso et al., 2018). Additionally, reductions in acetylated tubulin (which marks long-lived stable microtubules) are shown in both human and rodent brains (Larrayoz, 2017), but here to there are some opposing findings as acetylated tubulin has been shown to increase in the cerebellum of aged Wistar rodents (Marton et al., 2010). When observing age-related changes to MAPs, it is reported that MAP2, 1 and 5 are reduced in the hippocampus of rodents (Additional Table 1; Chauhan and Siegel, 1997; Di Stefano et al., 2001; Himeda et al., 2005) whilst the rodent cerebellar cortex curiously appears impervious to change (Chauhan and Siegel, 1997; Di Stefano et al., 2001). Similarly, MAP2 immunoreactivity is reduced in the photoreceptors of aged humans (Nag et al., 2020). From these studies, we see that there are likely region-specific reductions in microtubules and MAPs, although more work is needed to truly understand this. It is unclear whether reductions in these proteins influence the integrity of the cytoskeleton or morphology of the neuron or even what part of the neuron these changes occur within. Additionally, associating tubulin reductions to impairments in cytoskeletal integrity should be made sparingly as several of the studies investigate soluble tubulin which might not reflect polymerized tubulin integrated into microtubule arrays. Evidence for changes to the integrity of the microtubule cytoskeleton comes from research on tubulin turnover.
Age-related changes in microtubule turnover
Age-related decreases in microtubule integrity can be further investigated by observing changes in the balance of the dynamic instability process that governs microtubule stability. GTPase activity, alongside the binding of microtubule-associated proteins, directs the balance of this. Hippocampal and temporal tissue from young (average of 32 years) and old (average of 79 years) healthy aged humans have revealed an age-related change in tubulin polymerization and a trend for increased critical tubulin concentration (concentration of tubulin required for microtubule assembly) (Figure 2h; Rajaei et al., 2020). This is concurrent with a trend for increased GTPase activity, change in micro-environment, reduced hydrophobicity of tubulin and alpha-helix content and an apparent reduction in tau-microtubule interactions. These results agree with an earlier rodent study that also described GTPase-dependent age-related changes in tubulin kinetics/assembly (Qian et al., 1993). However, it is unknown whether changes in microtubule turnover affect the integrity of microtubules or cellular morphology. As noted by Rajaei, there is a reduction in tau-microtubule interactions alongside GTPase changes. Tau-related changes might be influencing the stability of microtubules with age which is especially relevant given taus relationship with neurodegenerative diseases for which age is the greatest risk factor.
Age-related changes in Tau
Tau biology is especially important when investigating the cytoskeleton due to its relationship with disease and its important role in influencing microtubule stability. Soluble tau has been shown to reduce by 14% per decade in non-cognitively impaired humans whilst insoluble tau contained within PHF (paired helical filaments) fractions remains unchanged despite low levels of tangle pathology being reported in most elderly brains (Mukaetova-Ladinska et al., 1996). This loss of tau appeared selective to distinct regions and mainly affected the hippocampus with up to 90% less soluble tau in aged vs. younger groups (18 to 88 years). Changes in the phosphorylation status of tau might be as important as the total amount of the protein. If total tau levels remain constant but the ratio of phosphorylated/normal tau differs, then this would indicate a mechanism by which microtubules could be disrupted. This is because hyperphosphorylated tau proteins are known to have reduced binding to microtubules and lead to cytoskeletal disruption in vivo (Cowan et al., 2010).
Age-related changes to patterns of Tau phosphorylation
The hyperphosphorylation of tau has been shown to reduce tau-microtubule binding and negatively influence microtubule stability and is seen at high levels in AD (Biernat et al., 1993; Bramblett et al., 1993; Alonso et al., 1994). There is also evidence from humans and rodents that tau phosphorylation increases with age, unrelated to disease (Figure 2e). Research, conducted on the human retina revealed that younger individuals had restriction of AT8 staining to small amounts of cone cells within localized retinal regions, but with age, this AT8 immunostaining became apparent within more distant regions and increased in intensity implying greater phosphorylation of tau at this epitope (Nag et al., 2020). Proteomic and phosphoproteomic analysis of the frontal cortex of normally aged humans has also shown significant levels of altered neuronal proteins with a large proportion of change coming from cytoskeletal/tubulin binding proteins when compared with young brains (Andrés-Benito et al., 2023). Changes in the phosphorylation of tau are also found in the brains of rhesus macaque monkeys. Pyramidal cells of aged Rhesus (30 years) show AT8 staining and reduction in tau solubility (indicates tau phosphorylation) whilst the frontal cortex shows age-related increases in tau phosphorylation (specifically at serine 396 and 235) using a single-shot phosphoproteomic analysis of tau (Datta et al., 2021; Leslie et al., 2021). Similarly, studies of the rodent nervous system describe age-related changes in AT180 and P-TauS404 staining in the hippocampus, cortex, and basal forebrain cholinergic neurons of rodents (4 vs. 28 months) with an increase in intensity within the cell bodies and a significant reduction in dendritic/axonal processes (Niewiadomska et al., 2005). Western blots of rodent tissue (3 vs. 18 months) show age-related increases in tau phosphorylation (PHF1 and AT8) within the hippocampus and frontal cortex (Larrayoz et al., 2017).
Collectively, this work provides evidence that normal aging is associated with an increase in the phosphorylation of tau and a reduction in non-phosphorylated tau. This is thought to be less drastic than the phosphorylation increases seen in disease, since radioimmunoassays and fluorescent imaging of the human cortex/entorhinal cortex and comparisons of CSF have shown AD brains to have significantly more tau phosphorylation than aged-matched controls (Khatoon et al., 1994; Sjögren et al., 2001; Neddens et al., 2018). The more subtle increase in tau phosphorylation seen with physiological aging, alongside reductions in total tau, tubulin, and MAPs and changes in tubulin turnover, could all have the potential to disrupt the microtubule cytoskeleton in the aging neuron. As these studies do not indicate distinct compartments that the changes occur within, it is hard to infer where these changes might influence morphology and function. However, it is more likely to lead to changes in axons and dendrites due to the high density of microtubules there. These phenotypes might represent a change in cytoskeletal stability that manifests as morphological and functional abnormalities previously reported in aging neurons.
Does the Aging Microtubule Cytoskeleton Contribute to Risk of Alzheimer's Disease?
There are many tenuous links between the age-related cytoskeletal changes described here and pathogenic processes evident in AD. As described above, age-related increases in phosphorylated tau may represent an early pathogenic step, especially since phosphorylation is associated with misfolding of tau, which in turn underpins more pathogenic properties like seeding and aggregation potential (Dujardin et al., 2018). Furthermore, the increased age-related tau phosphorylation, whilst not significant enough to cause dramatic MT destabilization and axonal transport deficits, may prime further tau phosphorylation (Bertrand et al., 2010). This could lead to more extensive and abnormal hyper-phosphorylation culminating in MT destabilization and greater axonal transport disruptions evident in diseased neurons (Cash et al., 2003; Wang et al., 2015). Finally, the mislocalization of phosphorylated tau, from the axonal to the dendritic compartments, which is evident in aged neurons, may facilitate another pathogenic event: retrograde propagation of pathology. Retrograde movement of pathogenic, often highly phosphorylated tau species, is now emerging as a novel mechanism of tau-toxicity (Hu et al., 2022), which may be rooted in this peculiar phenomenon seen in aging neurons.
Whether physiological age-related increases in tau phosphorylation, potentially initiating cytoskeletal breakdown seen in AD, can be restored with MT stabilizing drugs is still unknown. The utility of MT stabilization has been demonstrated in animal models of tauopathy (Brunden et al., 2010; Quraishe et al., 2013, 2016) whilst improvements in memory scores were described in humans with mild cognitive impairment following treatment with MT stabilizing agents (Morimoto et al., 2013).
Is There a Relationship between Age-Related Changes in Cytoskeletal Integrity and Neuronal Regeneration Capabilities?
Aside from the greater risk of protopathic neurodegenerative diseases, aged neurons are also characterized by reduced regenerative capabilities. Axonal regeneration following injury is dependent upon several processes, including axon intrinsic signaling pathways such as axotomy-induced axon-to-nucleus signaling, anterograde transport of mRNAs and proteins, retrograde importin signaling, growth cone formation, and axon growth (Smith et al., 2020). All of these processes are heavily reliant on efficient microtubule structure, function, and plasticity, which invariably declines with age. It is therefore conceivable that this is why older neurons have reduced regenerative potential. Indeed tubulin deacetylation, suggestive of more dynamic MTs occurs more prominently in peripheral neurons following injury, than in central neurons, which are less amenable to regeneration following axotomy (Cho and Cavalli, 2012). This is further supported by the observation that artificial MT stabilization of central neurons, using the MT stabilizing drug epothilone B, enhances their regeneration following injury (Ruschel et al., 2015).
Summary
From the discussed literature, there is clear evidence to suggest there are structural and functional deficits in the microtubular cytoskeleton with increasing age. Though this does not happen to the same extent in all regions of the brain, and does not occur uniformly in all species, we describe several reports of consistent changes in quantities and/or localizations of microtubule components and MAPs in an age-related manner. Limited structural studies indicate that the density and morphology of microtubules become disrupted, whilst axonal transport assays provide evidence for a reduction in cytoskeletal function. Collectively, these findings suggest that the microtubule cytoskeleton becomes compromised with age. However, more work is needed to determine the molecular causes and contributors to these structural and functional changes during healthy physiological aging. Additionally, it is still unknown if changes to the microtubular cytoskeleton (and indeed the actin and neurofilament cytoskeletons not discussed here) contribute towards age-related morphological changes reported in the somatodendritic and axonal compartments of neurons. Neither is it known whether age-related reductions in behavioral and cognitive functions underpinned by neuronal circuitry are in any way precipitated by the age-dependent deficits in microtubular structure and function discussed here, before overt age-related neurodegeneration inevitably develops. Studies comparing longitudinal changes to all aspects of cytoskeletal structure and function, and its relationship to overall neuronal morphology, and functional output during physiological aging are needed to draw these conclusions. Further studies are also required to simulate age-related neuronal changes in young neurons to study how this may lead to pathogenic processes that are clearly linked to neuronal aging, whilst not being a continuum of aging. If such studies reveal important causal links between cytoskeletal integrity, neuronal morphology, and functional output. This could unveil age-related vulnerabilities within neurons that could be reversed through fortification of cytoskeletal integrity thus protecting against neuronal aging and age-related risk of neurodegenerative disease.
Additional file:
Additional Table 1: Summary of age-related morphological and cytoskeletal changes covered in this review.
Acknowledgments:
We would like to thank the GKT for supporting this work.
Funding Statement
Funding: This work was funded by the Gerald Kerkut Charitable Trust (GKT) (to BR).
Footnotes
Conflicts of interest: The authors declare no conflicts of interest.
Data availability statement: All data relevant to the work are included in the article or uploaded as Additional files.
C-Editors: Zhao M, Liu WJ, Qiu Y; T-Editor: Jia Y
References
- Ackerman S. Washington (DC): National Academies Press (US); 1992. Discovering the brain. [PubMed] [Google Scholar]
- Afshordel S, Wood WG, Igbavboa U, Muller WE, Eckert GP. Impaired geranylgeranyltransferase-I regulation reduces membrane-associated Rho protein levels in aged mouse brain. J Neurochem. 2014;129:732–742. doi: 10.1111/jnc.12654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguilar-Hernández L, Vázquez-Hernández AJ, de-Lima-Mar DF, Vázquez-Roque RA, Tendilla-Beltrán H, Flores G. Memory and dendritic spines loss, and dynamic dendritic spines changes are age-dependent in the rat. J Chem Neuroanat. 2020;110:101858. doi: 10.1016/j.jchemneu.2020.101858. [DOI] [PubMed] [Google Scholar]
- Alonso AC, Zaidi T, Grundke-Iqbal I, Iqbal K. Role of abnormally phosphorylated tau in the breakdown of microtubules in Alzheimer disease. Proc Natl Acad Sci U S A. 1994;91:5562–5566. doi: 10.1073/pnas.91.12.5562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrés-Benito P, Íñigo-Marco I, Brullas M, Carmona M, Rio JAD, Fernández-Irigoyen J, Santamaría E, Povedano M, Ferrer I. Proteostatic modulation in brain aging without associated Alzheimer's disease-and age-related neuropathological changes. Aging (Albany NY) 2023;15:3295–3330. doi: 10.18632/aging.204698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baas PW, Lin S. Hooks and comets: the story of microtubule polarity orientation in the neuron. Dev Neurobiol. 2011;71:403–418. doi: 10.1002/dneu.20818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baas PW, Deitch JS, Black MM, Banker GA. Polarity orientation of microtubules in hippocampal neurons: uniformity in the axon and nonuniformity in the dendrite. Proc Natl Acad Sci U S A. 1988;85:8335–8339. doi: 10.1073/pnas.85.21.8335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbier P, Zejneli O, Martinho M, Lasorsa A, Belle V, Smet-Nocca C, Tsvetkov PO, Devred F, Landrieu I. Role of Tau as a microtubule-associated protein: structural and functional aspects. Front Aging Neurosci. 2019;11:204. doi: 10.3389/fnagi.2019.00204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes CA. Long-term potentiation and the ageing brain. Philos Trans R Soc Lond B Biol Sci. 2003;358:765–772. doi: 10.1098/rstb.2002.1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barone E, Mosser S, Fraering PC. Inactivation of brain Cofilin-1 by age, Alzheimer's disease and γ-secretase. Biochim Biophys Acta. 20141842:2500–2509. doi: 10.1016/j.bbadis.2014.10.004. [DOI] [PubMed] [Google Scholar]
- Bartol TM, Bromer C, Kinney J, Chirillo MA, Bourne JN, Harris KM, Sejnowski TJ. Nanoconnectomic upper bound on the variability of synaptic plasticity. Elife. 2015;4:e10778. doi: 10.7554/eLife.10778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bäurle J, Grüsser-Cornehls U. Axonal torpedoes in cerebellar Purkinje cells of two normal mouse strains during aging. Acta Neuropathol. 1994;88:237–245. doi: 10.1007/BF00293399. [DOI] [PubMed] [Google Scholar]
- Bertrand J, Plouffe V, Sénéchal P, Leclerc N. The pattern of human tau phosphorylation is the result of priming and feedback events in primary hippocampal neurons. Neuroscience. 2010;168:323–334. doi: 10.1016/j.neuroscience.2010.04.009. [DOI] [PubMed] [Google Scholar]
- Biernat J, Gustke N, Drewes G, Mandelkow EM, Mandelkow E. Phosphorylation of Ser262 strongly reduces binding of tau to microtubules: distinction between PHF-like immunoreactivity and microtubule binding. Neuron. 1993;11:153–163. doi: 10.1016/0896-6273(93)90279-z. [DOI] [PubMed] [Google Scholar]
- Bloss EB, Janssen WG, Ohm DT, Yuk FJ, Wadsworth S, Saardi KM, McEwen BS, Morrison JH. Evidence for reduced experience-dependent dendritic spine plasticity in the aging prefrontal cortex. J Neurosci. 2011;31:7831–7839. doi: 10.1523/JNEUROSCI.0839-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boros BD, Greathouse KM, Gearing M, Herskowitz JH. Dendritic spine remodeling accompanies Alzheimer's disease pathology and genetic susceptibility in cognitively normal aging. Neurobiol Aging. 2019;73:92–103. doi: 10.1016/j.neurobiolaging.2018.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowley MP, Cabral H, Rosene DL, Peters A. Age changes in myelinated nerve fibers of the cingulate bundle and corpus callosum in the rhesus monkey. J Comp Neurol. 2010;518:3046–3064. doi: 10.1002/cne.22379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bramblett GT, Goedert M, Jakes R, Merrick SE, Trojanowski JQ, Lee VM. Abnormal tau phosphorylation at Ser396 in Alzheimer's disease recapitulates development and contributes to reduced microtubule binding. Neuron. 1993;10:1089–1099. doi: 10.1016/0896-6273(93)90057-x. [DOI] [PubMed] [Google Scholar]
- Brody H. Organization of the cerebral cortex. III. A study of aging in the human cerebral cortex. J Comp Neurol. 1955;102:511–516. doi: 10.1002/cne.901020206. [DOI] [PubMed] [Google Scholar]
- Brody H. The deposition of aging pigment in the human cerebral cortex. J Gerontol. 1960;15:258–261. doi: 10.1093/geronj/15.3.258. [DOI] [PubMed] [Google Scholar]
- Brunden KR, Zhang B, Carroll J, Yao Y, Potuzak JS, Hogan AM, Iba M, James MJ, Xie SX, Ballatore C, Smith AB, 3rd, Lee VM, Trojanowski JQ. Epothilone D improves microtubule density, axonal integrity, and cognition in a transgenic mouse model of tauopathy. J Neurosci. 2010;30:13861–13866. doi: 10.1523/JNEUROSCI.3059-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryan J, Wilson L. Are cytoplasmic microtubules heteropolymers? Proc Natl Acad Sci U S A. 1971;68:1762–1766. doi: 10.1073/pnas.68.8.1762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buell SJ, Coleman PD. Dendritic growth in the aged human brain and failure of growth in senile dementia. Science. 1979;206:854–856. doi: 10.1126/science.493989. [DOI] [PubMed] [Google Scholar]
- Burrinha T, Cunha C, Hall MJ, Lopes-da-Silva M, Seabra MC, Guimas Almeida C. Deacidification of endolysosomes by neuronal aging drives synapse loss. Traffic. 2023;24:334–354. doi: 10.1111/tra.12889. [DOI] [PubMed] [Google Scholar]
- Calì C, Wawrzyniak M, Becker C, Maco B, Cantoni M, Jorstad A, Nigro B, Grillo F, De Paola V, Fua P, Knott GW. The effects of aging on neuropil structure in mouse somatosensory cortex-A 3D electron microscopy analysis of layer 1. PLoS One. 2018;13:e0198131. doi: 10.1371/journal.pone.0198131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cash AD, Aliev G, Siedlak SL, Nunomura A, Fujioka H, Zhu X, Raina AK, Vinters HV, Tabaton M, Johnson AB, Paula-Barbosa M, Avíla J, Jones PK, Castellani RJ, Smith MA, Perry G. Microtubule reduction in Alzheimer's disease and aging is independent of tau filament formation. Am J Pathol. 2003;162:1623–1627. doi: 10.1016/s0002-9440(10)64296-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro RW, Lopes MC, Settlage RE, Valdez G. Aging alters mechanisms underlying voluntary movements in spinal motor neurons of mice, primates, and humans. JCI Insight. 2023;8:e168448. doi: 10.1172/jci.insight.168448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chauhan N, Siegel G. Age-dependent organotypic expression of microtubule-associated proteins (MAP1, MAP2, and MAP5) in rat brain. Neurochem Res. 1997;22:713–719. doi: 10.1023/a:1027306227402. [DOI] [PubMed] [Google Scholar]
- Chen S, Hillman DE. Dying-back of Purkinje cell dendrites with synapse loss in aging rats. J Neurocytol. 1999;28:187–196. doi: 10.1023/a:1007015721754. [DOI] [PubMed] [Google Scholar]
- Cho Y, Cavalli V. HDAC5 is a novel injury-regulated tubulin deacetylase controlling axon regeneration. EMBO J. 2012;31:3063–3078. doi: 10.1038/emboj.2012.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Correale J, Marrodan M, Ysrraelit MC. Mechanisms of neurodegeneration and axonal dysfunction in progressive multiple sclerosis. Biomedicines. 2019;7:14. doi: 10.3390/biomedicines7010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corsi A, Bombieri C, Valenti MT, Romanelli MG. Tau isoforms: gaining insight into MAPT alternative splicing. Int J Mol Sci. 2022;23:15383. doi: 10.3390/ijms232315383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan CM, Bossing T, Page A, Shepherd D, Mudher A. Soluble hyper-phosphorylated tau causes microtubule breakdown and functionally compromises normal tau in vivo. Acta Neuropathol. 2010;120:593–604. doi: 10.1007/s00401-010-0716-8. [DOI] [PubMed] [Google Scholar]
- Datta D, Leslie SN, Wang M, Morozov YM, Yang S, Mentone S, Zeiss C, Duque A, Rakic P, Horvath TL, van Dyck CH, Nairn AC, Arnsten AFT. Age-related calcium dysregulation linked with tau pathology and impaired cognition in non-human primates. Alzheimers Dement. 2021;17:920–932. doi: 10.1002/alz.12325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Brabander JM, Kramers RJ, Uylings HB. Layer-specific dendritic regression of pyramidal cells with ageing in the human prefrontal cortex. Eur J Neurosci. 1998;10:1261–1269. doi: 10.1046/j.1460-9568.1998.00137.x. [DOI] [PubMed] [Google Scholar]
- Debanne D, Campanac E, Bialowas A, Carlier E, Alcaraz G. Axon physiology. Physiol Rev. 2011;91:555–602. doi: 10.1152/physrev.00048.2009. [DOI] [PubMed] [Google Scholar]
- Di Stefano G, Casoli T, Fattoretti P, Gracciotti N, Solazzi M, Bertoni-Freddari C. Distribution of MAP2 in hippocampus and cerebellum of young and old rats by quantitative immunohistochemistry. J Histochem Cytochem. 2001;49:1065–1066. doi: 10.1177/002215540104900818. [DOI] [PubMed] [Google Scholar]
- Di Stefano G, Casoli T, Fattoretti P, Balietti M, Grossi Y, Giorgetti B, Bertoni-Freddari C. Level and distribution of microtubule-associated protein-2 (MAP2) as an index of dendritic structural dynamics. Rejuvenation Res. 2006;9:94–98. doi: 10.1089/rej.2006.9.94. [DOI] [PubMed] [Google Scholar]
- Didonna A, Opal P. The role of neurofilament aggregation in neurodegeneration: lessons from rare inherited neurological disorders. Mol Neurodegener. 2019;14:19. doi: 10.1186/s13024-019-0318-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominguez R, Holmes KC. Actin structure and function. Annu Rev Biophys. 2011;40:169–186. doi: 10.1146/annurev-biophys-042910-155359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duan HL, Wearne SL, Rocher AB, Macedo A, Morrison JH, Hof PR. Age-related dendritic and spine changes in corticocortically projecting neurons in macaque monkeys. Cerebral Cortex. 2003;13:950–961. doi: 10.1093/cercor/13.9.950. [DOI] [PubMed] [Google Scholar]
- Dujardin S, Bégard S, Caillierez R, Lachaud C, Carrier S, Lieger S, Gonzalez JA, Deramecourt V, Déglon N, Maurage CA, Frosch MP, Hyman BT, Colin M, Buée L. Different tau species lead to heterogeneous tau pathology propagation and misfolding. Acta Neuropathol Commun. 2018;6:132. doi: 10.1186/s40478-018-0637-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dumitriu D, Hao J, Hara Y, Kaufmann J, Janssen WG, Lou W, Rapp PR, Morrison JH. Selective changes in thin spine density and morphology in monkey prefrontal cortex correlate with aging-related cognitive impairment. J Neurosci. 2010;30:7507–7515. doi: 10.1523/JNEUROSCI.6410-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan JE, Lytle NK, Zuniga A, Goldstein LS. The microtubule regulatory protein stathmin is required to maintain the integrity of axonal microtubules in drosophila. PLoS One. 2013;8:e68324. doi: 10.1371/journal.pone.0068324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eichel K, Shen K. The function of the axon initial segment in neuronal polarity. Dev Biol. 2022;489:47–54. doi: 10.1016/j.ydbio.2022.05.016. [DOI] [PubMed] [Google Scholar]
- Fan Q, Tian Q, Ohringer NA, Nummenmaa A, Witzel T, Tobyne SM, Klawiter EC, Mekkaoui C, Rosen BR, Wald LL, Salat DH, Huang SY. Age-related alterations in axonal microstructure in the corpus callosum measured by high-gradient diffusion MRI. Neuroimage. 2019;191:325–336. doi: 10.1016/j.neuroimage.2019.02.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fassier C, Tarrade A, Peris L, Courageot S, Mailly P, Dalard C, Delga S, Roblot N, Lefèvre J, Job D, Hazan J, Curmi PA, Melki J. Microtubule-targeting drugs rescue axonal swellings in cortical neurons from spastin knockout mice. Dis Model Mech. 2013;6:72–83. doi: 10.1242/dmm.008946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernández-Chacón R, Wölfel M, Nishimune H, Tabares L, Schmitz F, Castellano-Muñoz M, Rosenmund C, Montesinos ML, Sanes JR, Schneggenburger R, Südhof TC. The synaptic vesicle protein CSP alpha prevents presynaptic degeneration. Neuron. 2004;42:237–251. doi: 10.1016/s0896-6273(04)00190-4. [DOI] [PubMed] [Google Scholar]
- Fifkova E, Morales M. Aging and the neurocytoskeleton. Exp Gerontol. 1992;27:125–136. doi: 10.1016/0531-5565(92)90034-w. [DOI] [PubMed] [Google Scholar]
- Filipek A, Schneider G, Mietelska A, Figiel I, Niewiadomska G. Age-dependent changes in neuronal distribution of CacyBP/SIP: comparison to tubulin and the tau protein. J Neural Transm. 2008;115:1257–1264. doi: 10.1007/s00702-008-0062-3. [DOI] [PubMed] [Google Scholar]
- Geula C, Nagykery N, Nicholas A, Wu CK. Cholinergic neuronal and axonal abnormalities are present early in aging and in Alzheimer disease. J Neuropathol Exp Neurol. 2008;67:309–318. doi: 10.1097/NEN.0b013e31816a1df3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray DA, Woulfe J. Lipofuscin and aging: a matter of toxic waste. Sci Aging Knowledge Environ. 2005;2005:re1. doi: 10.1126/sageke.2005.5.re1. [DOI] [PubMed] [Google Scholar]
- Hardy J. Failures in protein clearance partly underlie late onset neurodegenerative diseases and link pathology to genetic risk. Front Neurosci. 2019;13:1304. doi: 10.3389/fnins.2019.01304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatanpaa K, Isaacs KR, Shirao T, Brady DR, Rapoport SI. Loss of proteins regulating synaptic plasticity in normal aging of the human brain and in Alzheimer disease. J Neuropathol Exp Neurol. 1999;58:637–643. doi: 10.1097/00005072-199906000-00008. [DOI] [PubMed] [Google Scholar]
- Häusser M, Spruston N, Stuart GJ. Diversity and dynamics of dendritic signaling. Science. 2000;290:739–744. doi: 10.1126/science.290.5492.739. [DOI] [PubMed] [Google Scholar]
- Hess M, Gomariz A, Goksel O, Ewald CY. In-vivo quantitative image analysis of age-related morphological changes of C. elegans neurons reveals a correlation between neurite bending and novel neurite outgrowths. eNeuro. 2019;6 doi: 10.1523/ENEURO.0014-19.2019. ENEURO.0014-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higuchi-Sanabria R, Paul JW, 3rd, Durieux J, Benitez C, Frankino PA, Tronnes SU, Garcia G, Daniele JR, Monshietehadi S, Dillin A. Spatial regulation of the actin cytoskeleton by HSF-1 during aging. Mol Biol Cell. 2018;29:2522–2527. doi: 10.1091/mbc.E18-06-0362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Himeda T, Mizuno K, Kato H, Araki T. Effects of age on immunohistochemical changes in the mouse hippocampus. Mech Ageing Dev. 2005;126:673–677. doi: 10.1016/j.mad.2004.12.004. [DOI] [PubMed] [Google Scholar]
- Hu W, Liu F, Gong CX, Iqbal K. Does proteopathic tau propagate trans-synaptically in the brain? Mol Neurodegener. 2022;17:21. doi: 10.1186/s13024-022-00527-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang L, Zhou H, Chen K, Chen X, Yang G. Learning-dependent dendritic spine plasticity is reduced in the aged mouse cortex. Front Neural Circuits. 2020;14:581435. doi: 10.3389/fncir.2020.581435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Idland AV, Sala-Llonch R, Borza T, Watne LO, Wyller TB, Braekhus A, Zetterberg H, Blennow K, Walhovd KB, Fjell AM. CSF neurofilament light levels predict hippocampal atrophy in cognitively healthy older adults. Neurobiol Aging. 2017;49:138–144. doi: 10.1016/j.neurobiolaging.2016.09.012. [DOI] [PubMed] [Google Scholar]
- Jacobs B, Driscoll L, Schall M. Life-span dendritic and spine changes in areas 10 and 18 of human cortex: a quantitative Golgi study. J Comp Neurol. 1997;386:661–680. [PubMed] [Google Scholar]
- Kabaso D, Coskren PJ, Henry BI, Hof PR, Wearne SL. The electrotonic structure of pyramidal neurons contributing to prefrontal cortical circuits in macaque monkeys is significantly altered in aging. Cereb Cortex. 2009;19:2248–2268. doi: 10.1093/cercor/bhn242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadavath H, Hofele RV, Biernat J, Kumar S, Tepper K, Urlaub H, Mandelkow E, Zweckstetter M. Tau stabilizes microtubules by binding at the interface between tubulin heterodimers. Proc Natl Acad Sci U S A. 2015;112:7501–7506. doi: 10.1073/pnas.1504081112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanaan NM, Pigino GF, Brady ST, Lazarov O, Binder LI, Morfini GA. Axonal degeneration in Alzheimer's disease: when signaling abnormalities meet the axonal transport system. Exp Neurol. 2013;246:44–53. doi: 10.1016/j.expneurol.2012.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kapitein LC, Hoogenraad CC. Building the neuronal microtubule cytoskeleton. Neuron. 2015;87:492–506. doi: 10.1016/j.neuron.2015.05.046. [DOI] [PubMed] [Google Scholar]
- Kasai H, Matsuzaki M, Noguchi J, Yasumatsu N, Nakahara H. Structure-stability-function relationships of dendritic spines. Trends Neurosci. 2003;26:360–368. doi: 10.1016/S0166-2236(03)00162-0. [DOI] [PubMed] [Google Scholar]
- Kasher PR, De Vos KJ, Wharton SB, Manser C, Bennett EJ, Bingley M, Wood JD, Milner R, McDermott CJ, Miller CC, Shaw PJ, Grierson AJ. Direct evidence for axonal transport defects in a novel mouse model of mutant spastin-induced hereditary spastic paraplegia (HSP) and human HSP patients. J Neurochem. 2009;110:34–44. doi: 10.1111/j.1471-4159.2009.06104.x. [DOI] [PubMed] [Google Scholar]
- Kato T, Hirano A. A Golgi study of the proximal portion of the human Purkinje cell axon. Acta Neuropathol. 1985;68:191–195. doi: 10.1007/BF00690193. [DOI] [PubMed] [Google Scholar]
- Khalil M, Pirpamer L, Hofer E, Voortman MM, Barro C, Leppert D, Benkert P, Ropele S, Enzinger C, Fazekas F, Schmidt R, Kuhle J. Serum neurofilament light levels in normal aging and their association with morphologic brain changes. Nature Communications. 2020;11:812. doi: 10.1038/s41467-020-14612-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khatoon S, Grundke-Iqbal I, Iqbal K. Levels of normal and abnormally phosphorylated tau in different cellular and regional compartments of Alzheimer disease and control brains. FEBS Lett. 1994;351:80–84. doi: 10.1016/0014-5793(94)00829-9. [DOI] [PubMed] [Google Scholar]
- Kneynsberg A, Kanaan NM. Aging does not affect axon initial segment structure and somatic localization of Tau protein in hippocampal neurons of Fischer 344 rats. eNeuro. 2017;4 doi: 10.1523/ENEURO.0043-17.2017. ENEURO.0043-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konietzny A, Bär J, Mikhaylova M. Dendritic actin cytoskeleton: structure, functions, and regulations. Front Cell Neurosci. 2017;11:147. doi: 10.3389/fncel.2017.00147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kritsilis M, V Rizou S, Koutsoudaki PN, Evangelou K, Gorgoulis VG, Papadopoulos D. Ageing, cellular senescence and neurodegenerative disease. Int J Mol Sci. 2018:19. doi: 10.3390/ijms19102937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhle J, Leppert D, Petzold A, Regeniter A, Schindler C, Mehling M, Anthony DC, Kappos L, Lindberg RL. Neurofilament heavy chain in CSF correlates with relapses and disability in multiple sclerosis. Neurology. 2011;76:1206–1213. doi: 10.1212/WNL.0b013e31821432ff. [DOI] [PubMed] [Google Scholar]
- Kuijpers M. Keeping synapses in shape: degradation pathways in the healthy and aging brain. Neuronal Signal. 2022;6:Ns20210063. doi: 10.1042/NS20210063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labisso WL, Raulin AC, Nwidu LL, Kocon A, Wayne D, Erdozain AM, Morentin B, Schwendener D, Allen G, Enticott J, Gerdes HK, Johnson L, Grzeskowiak J, Drizou F, Tarbox R, Osna NA, Kharbanda KK, Callado LF, Carter WG. The loss of α- and β-tubulin proteins are a pathological hallmark of chronic alcohol consumption and natural brain ageing. Brain Sci. 2018;8:175. doi: 10.3390/brainsci8090175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai WF, Wong WT. Roles of the actin cytoskeleton in aging and age-associated diseases. Ageing Res Rev. 2020;58:101021. doi: 10.1016/j.arr.2020.101021. [DOI] [PubMed] [Google Scholar]
- Landfield PW, McGaugh JL, Lynch G. Impaired synaptic potentiation processes in the hippocampus of aged, memory-deficient rats. Brain Res. 1978;150:85–101. doi: 10.1016/0006-8993(78)90655-8. [DOI] [PubMed] [Google Scholar]
- Larrayoz IM, Ferrero H, Martisova E, Gil-Bea FJ, Ramírez MJ, Martínez A. Adrenomedullin contributes to age-related memory loss in mice and is elevated in aging human brains. Front Mol Neurosci. 2017;10:384. doi: 10.3389/fnmol.2017.00384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leslie SN, Kanyo J, Datta D, Wilson RS, Zeiss C, Duque A, Lam TT, Arnsten AFT, Nairn AC. Simple, single-shot phosphoproteomic analysis of heat-stable tau identifies age-related changes in pS235- and pS396-Tau levels in non-human primates. Front Aging Neurosci. 2021;13:767322. doi: 10.3389/fnagi.2021.767322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu RH, Bertolotto C, Engelhardt JK, Chase MH. Age-related changes in soma size of neurons in the spinal cord motor column of the cat. Neurosci Lett. 1996;211:163–166. doi: 10.1016/0304-3940(96)12742-7. [DOI] [PubMed] [Google Scholar]
- Louis ED, Faust PL, Vonsattel JP, Erickson-Davis C. Purkinje cell axonal torpedoes are unrelated to advanced aging and likely reflect cerebellar injury. Acta Neuropathol. 2009;117:719–721. doi: 10.1007/s00401-009-0534-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luebke JI, Medalla M, Amatrudo JM, Weaver CM, Crimins JL, Hunt B, Hof PR, Peters A. Age-related changes to layer 3 pyramidal cells in the rhesus monkey visual cortex. Cereb Cortex. 2015;25:1454–1468. doi: 10.1093/cercor/bht336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mack TG, Kreis P, Eickholt BJ. Defective actin dynamics in dendritic spines: cause or consequence of age-induced cognitive decline? Biol Chem. 2016;397:223–229. doi: 10.1515/hsz-2015-0185. [DOI] [PubMed] [Google Scholar]
- Marangoni M, Adalbert R, Janeckova L, Patrick J, Kohli J, Coleman MP, Conforti L. Age-related axonal swellings precede other neuropathological hallmarks in a knock-in mouse model of Huntington's disease. Neurobiol Aging. 2014;35:2382–2393. doi: 10.1016/j.neurobiolaging.2014.04.024. [DOI] [PubMed] [Google Scholar]
- Marton O, Koltai E, Nyakas C, Bakonyi T, Zenteno-Savin T, Kumagai S, Goto S, Radak Z. Aging and exercise affect the level of protein acetylation and SIRT1 activity in cerebellum of male rats. Biogerontology. 2010;11:679–686. doi: 10.1007/s10522-010-9279-2. [DOI] [PubMed] [Google Scholar]
- Merlo S, Nakayama AB, Brusco J, Rossi MA, Carlotti CG, Jr., Moreira JE. Lipofuscin granules in the epileptic human temporal neocortex with age. Ultrastruct Pathol. 2015;39:378–384. doi: 10.3109/01913123.2015.1043416. [DOI] [PubMed] [Google Scholar]
- Metzner K, Darawsha O, Wang M, Gaur N, Cheng Y, Rödiger A, Frahm C, Witte OW, Perocchi F, Axer H, Grosskreutz J, Brill MS. Age-dependent increase of cytoskeletal components in sensory axons in human skin. Front Cell Dev Biol. 2022;10:965382. doi: 10.3389/fcell.2022.965382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michiels L, Delva A, van Aalst J, Ceccarini J, Vandenberghe W, Vandenbulcke M, Koole M, Lemmens R, Laere KV. Synaptic density in healthy human aging is not influenced by age or sex: a (11)C-UCB-J PET study. Neuroimage. 2021;232:117877. doi: 10.1016/j.neuroimage.2021.117877. [DOI] [PubMed] [Google Scholar]
- Milde S, Adalbert R, Elaman MH, Coleman MP. Axonal transport declines with age in two distinct phases separated by a period of relative stability. Neurobiol Aging. 2015;36:971–981. doi: 10.1016/j.neurobiolaging.2014.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchison T, Kirschner M. Dynamic instability of microtubule growth. Nature. 1984;312:237–242. doi: 10.1038/312237a0. [DOI] [PubMed] [Google Scholar]
- Morimoto BH, Schmechel D, Hirman J, Blackwell A, Keith J, Gold M. A double-blind, placebo-controlled, ascending-dose, randomized study to evaluate the safety, tolerability and effects on cognition of AL-108 after 12 weeks of intranasal administration in subjects with mild cognitive impairment. Dement Geriatr Cogn Disord. 2013;35:325–336. doi: 10.1159/000348347. [DOI] [PubMed] [Google Scholar]
- Mostany R, Anstey JE, Crump KL, Maco B, Knott G, Portera-Cailliau C. Altered synaptic dynamics during normal brain aging. J Neurosci. 2013;33:4094–4104. doi: 10.1523/JNEUROSCI.4825-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukaetova-Ladinska EB, Harrington CR, Roth M, Wischik CM. Alterations in tau protein metabolism during normal aging. Dementia. 1996;7:95–103. doi: 10.1159/000106861. [DOI] [PubMed] [Google Scholar]
- Muzio MR, Cascella M. StatPearls. Treasure Island (FL): StatPearls Publishing; 2023. Histology, Axon. [PubMed] [Google Scholar]
- Nag TC, Kathpalia P, Wadhwa S. Microtubule alterations may destabilize photoreceptor integrity: Age-related microtubule changes and pattern of expression of MAP-2, Tau and hyperphosphorylated Tau in aging human photoreceptor cells. Experimental Eye Research. 2020:198. doi: 10.1016/j.exer.2020.108153. [DOI] [PubMed] [Google Scholar]
- Nakamura S, Akiguchi I, Kameyama M, Mizuno N. Age-related changes of pyramidal cell basal dendrites in layers III and V of human motor cortex: a quantitative Golgi study. Acta Neuropathol. 1985a;65:281–284. doi: 10.1007/BF00687009. [DOI] [PubMed] [Google Scholar]
- Nakamura S, Akiguchi I, Kameyama M, Mizuno N. Age-related-changes of pyramidal cell basal dendrites in layers-III and layers-V of human motor cortex - a quantitative golgi-study. Acta Neuropathologica. 1985b;65:281–284. doi: 10.1007/BF00687009. [DOI] [PubMed] [Google Scholar]
- Nakamura S, Akiguchi I, Kameyama M, Kudo M, Mizuno N. Senile changes in the basal dendrites of the pyramidal cells in the human motor cortex--a quantitative Golgi study. Rinsho Shinkeigaku. 1984;24:85–95. [PubMed] [Google Scholar]
- Neddens J, Temmel M, Flunkert S, Kerschbaumer B, Hoeller C, Loeffler T, Niederkofler V, Daum G, Attems J, Hutter-Paier B. Phosphorylation of different tau sites during progression of Alzheimer's disease. Acta Neuropathol Commun. 2018;6:52. doi: 10.1186/s40478-018-0557-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niewiadomska G, Baksalerska-Pazera M. Age-dependent changes in axonal transport and cellular distribution of Tau 1 in the rat basal forebrain neurons. Neuroreport. 2003;14:1701–1706. doi: 10.1097/00001756-200309150-00009. [DOI] [PubMed] [Google Scholar]
- Niewiadomska G, Baksalerska-Pazera M, Riedel G. Altered cellular distribution of phospho-tau proteins coincides with impaired retrograde axonal transport in neurons of aged rats. Ann N Y Acad Sci. 20051048:287–295. doi: 10.1196/annals.1342.026. [DOI] [PubMed] [Google Scholar]
- No authors listed 2022 Alzheimer's disease facts and figures. Alzheimers Dement. 2022;18:700–789. doi: 10.1002/alz.12638. [DOI] [PubMed] [Google Scholar]
- Nyberg L, Lundquist A, Nordin Adolfsson A, Andersson M, Zetterberg H, Blennow K, Adolfsson R. Elevated plasma neurofilament light in aging reflects brain white-matter alterations but does not predict cognitive decline or Alzheimer's disease. Alzheimers Dement (Amst) 2020;12:e12050. doi: 10.1002/dad2.12050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Panda D, Daijo JE, Jordan MA, Wilson L. Kinetic stabilization of microtubule dynamics at steady state in vitro by substoichiometric concentrations of tubulin-colchicine complex. Biochemistry. 1995;34:9921–9929. doi: 10.1021/bi00031a014. [DOI] [PubMed] [Google Scholar]
- Pannese E. Morphological changes in nerve cells during normal aging. Brain Struct Funct. 2011;216:85–89. doi: 10.1007/s00429-011-0308-y. [DOI] [PubMed] [Google Scholar]
- Penazzi L, Bakota L, Brandt R. Microtubule dynamics in neuronal development, plasticity, and neurodegeneration. Int Rev Cell Mol Biol. 2016;321:89–169. doi: 10.1016/bs.ircmb.2015.09.004. [DOI] [PubMed] [Google Scholar]
- Peters A, Sethares C, Killiany RJ. Effects of age on the thickness of myelin sheaths in monkey primary visual cortex. J Comp Neurol. 2001;435:241–248. doi: 10.1002/cne.1205. [DOI] [PubMed] [Google Scholar]
- Pozo Devoto VM, Lacovich V, Feole M, Bhat P, Chovan J, Čarna M, Onyango IG, Dragišić N, Sűsserová M, Barrios-Llerena ME, Stokin GB. Unraveling axonal mechanisms of traumatic brain injury. Acta Neuropathol Commun. 2022;10:140. doi: 10.1186/s40478-022-01414-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian A, Burton PR, Himes RH. A comparison of microtubule assembly in brain extracts from young and old rats. Brain Res Mol Brain Res. 1993;18:100–106. doi: 10.1016/0169-328x(93)90177-q. [DOI] [PubMed] [Google Scholar]
- Qiang L, Sun X, Austin TO, Muralidharan H, Jean DC, Liu M, Yu W, Baas PW. Tau does not stabilize axonal microtubules but rather enables them to have long labile domains. Curr Biol. 2018;28:2181–2189. doi: 10.1016/j.cub.2018.05.045. [DOI] [PubMed] [Google Scholar]
- Quraishe S, Cowan CM, Mudher A. NAP (davunetide) rescues neuronal dysfunction in a Drosophila model of tauopathy. Mol Psychiatry. 2013;18:834–842. doi: 10.1038/mp.2013.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quraishe S, Sealey M, Cranfield L, Mudher A. Microtubule stabilising peptides rescue tau phenotypes in-vivo. Sci Rep. 2016;6:38224. doi: 10.1038/srep38224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajaei S, Karima S, Sepasi Tehrani H, Shateri S, Mahmoodi Baram S, Mahdavi M, Mokhtari F, Alimohammadi A, Tafakhori A, Amiri A, Aghamollaii V, Fatemi H, Rajabibazl M, Kobarfard F, Gorji A. Conformational change and GTPase activity of human tubulin: A comparative study on Alzheimer's disease and healthy brain. J Neurochem. 2020;155:207–224. doi: 10.1111/jnc.15009. [DOI] [PubMed] [Google Scholar]
- Robles LJ. Accumulation and identification of lipofuscin-like pigment in the neurons of Bulla gouldiana (Gastropoda: Opisthobranchia) Mech Ageing Dev. 1978;7:53–64. doi: 10.1016/0047-6374(78)90052-0. [DOI] [PubMed] [Google Scholar]
- Ruschel J, Hellal F, Flynn KC, Dupraz S, Elliott DA, Tedeschi A, Bates M, Sliwinski C, Brook G, Dobrindt K, Peitz M, Brüstle O, Norenberg MD, Blesch A, Weidner N, Bunge MB, Bixby JL, Bradke F. Axonal regeneration. Systemic administration of epothilone B promotes axon regeneration after spinal cord injury. Science. 2015;348:347–352. doi: 10.1126/science.aaa2958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayas CL, Ávila J. Crosstalk between axonal classical microtubule-associated proteins and end binding proteins during axon extension: possible implications in neurodegeneration. J Alzheimers Dis 40 Suppl. 2014;1:S17–22. doi: 10.3233/JAD-132315. [DOI] [PubMed] [Google Scholar]
- Scheff SW, Price DA, Sparks DL. Quantitative assessment of possible age-related change in synaptic numbers in the human frontal cortex. Neurobiol Aging. 2001;22:355–365. doi: 10.1016/s0197-4580(01)00222-6. [DOI] [PubMed] [Google Scholar]
- Shimada A, Tsuzuki M, Keino H, Satoh M, Chiba Y, Saitoh Y, Hosokawa M. Apical vulnerability to dendritic retraction in prefrontal neurones of ageing SAMP10 mouse: a model of cerebral degeneration. Neuropathol Appl Neurobiol. 2006;32:1–14. doi: 10.1111/j.1365-2990.2006.00632.x. [DOI] [PubMed] [Google Scholar]
- Siahaan V, Tan R, Humhalova T, Libusova L, Lacey SE, Tan T, Dacy M, Ori-McKenney KM, McKenney RJ, Braun M, Lansky Z. Microtubule lattice spacing governs cohesive envelope formation of tau family proteins. Nat Chem Biol. 2022;18:1224–1235. doi: 10.1038/s41589-022-01096-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sjögren M, Vanderstichele H, Agren H, Zachrisson O, Edsbagge M, Wikkelsø C, Skoog I, Wallin A, Wahlund LO, Marcusson J, Nägga K, Andreasen N, Davidsson P, Vanmechelen E, Blennow K. Tau and Abeta42 in cerebrospinal fluid from healthy adults 21-93 years of age: establishment of reference values. Clin Chem. 2001;47:1776–1781. [PubMed] [Google Scholar]
- Smith TP, Sahoo PK, Kar AN, Twiss JL. Intra-axonal mechanisms driving axon regeneration. Brain Res. 2020;1740:146864. doi: 10.1016/j.brainres.2020.146864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sotoudeh N, Namavar MR, Zarifkar A, Heidarzadegan AR. Age-dependent changes in the medial prefrontal cortex and medial amygdala structure, and elevated plus-maze performance in the healthy male Wistar rats. IBRO Rep. 2020;9:183–194. doi: 10.1016/j.ibror.2020.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stahon KE, Bastian C, Griffith S, Kidd GJ, Brunet S, Baltan S. Age-related changes in axonal and mitochondrial ultrastructure and function in white matter. J Neurosci. 2016;36:9990–10001. doi: 10.1523/JNEUROSCI.1316-16.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone MC, Roegiers F, Rolls MM. Microtubules have opposite orientation in axons and dendrites of Drosophila neurons. Mol Biol Cell. 2008;19:4122–4129. doi: 10.1091/mbc.E07-10-1079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takihara Y, Inatani M, Eto K, Inoue T, Kreymerman A, Miyake S, Ueno S, Nagaya M, Nakanishi A, Iwao K, Takamura Y, Sakamoto H, Satoh K, Kondo M, Sakamoto T, Goldberg JL, Nabekura J, Tanihara H. In vivo imaging of axonal transport of mitochondria in the diseased and aged mammalian CNS. Proc Natl Acad Sci U S A. 2015;112:10515–10520. doi: 10.1073/pnas.1509879112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan R, Lam AJ, Tan T, Han J, Nowakowski DW, Vershinin M, Simó S, Ori-McKenney KM, McKenney RJ. Microtubules gate tau condensation to spatially regulate microtubule functions. Nat Cell Biol. 2019;21:1078–1085. doi: 10.1038/s41556-019-0375-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terman A, Brunk UT. Lipofuscin: mechanisms of formation and increase with age. Apmis. 1998;106:265–276. doi: 10.1111/j.1699-0463.1998.tb01346.x. [DOI] [PubMed] [Google Scholar]
- Thibault O, Landfield PW. Increase in single L-type calcium channels in hippocampal neurons during aging. Science. 1996;272:1017–1020. doi: 10.1126/science.272.5264.1017. [DOI] [PubMed] [Google Scholar]
- Uemura E, Hartmann HA. RNA content and volume of nerve cell bodies in human brain. I. Prefrontal cortex in aging normal and demented patients. J Neuropathol Exp Neurol. 1978;37:487–496. doi: 10.1097/00005072-197809000-00004. [DOI] [PubMed] [Google Scholar]
- Ueno H, Takao K, Suemitsu S, Murakami S, Kitamura N, Wani K, Okamoto M, Aoki S, Ishihara T. Age-dependent and region-specific alteration of parvalbumin neurons and perineuronal nets in the mouse cerebral cortex. Neurochem Int. 2018;112:59–70. doi: 10.1016/j.neuint.2017.11.001. [DOI] [PubMed] [Google Scholar]
- Vågberg M, Norgren N, Dring A, Lindqvist T, Birgander R, Zetterberg H, Svenningsson A. Levels and age dependency of neurofilament light and glial fibrillary acidic protein in healthy individuals and their relation to the brain parenchymal fraction. PLoS One. 2015;10:e0135886. doi: 10.1371/journal.pone.0135886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vagnoni A, Hoffmann PC, Bullock SL. Reducing Lissencephaly-1 levels augments mitochondrial transport and has a protective effect in adult Drosophila neurons. J Cell Sci. 2016;129:178–190. doi: 10.1242/jcs.179184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ve H, Cabana VC, Gouspillou G, Lussier MP. Quantitative immunoblotting analyses reveal that the abundance of actin, tubulin, synaptophysin and EEA1 proteins is altered in the brains of aged mice. Neuroscience. 2020;442:100–113. doi: 10.1016/j.neuroscience.2020.06.044. [DOI] [PubMed] [Google Scholar]
- Wang M, Gamo NJ, Yang Y, Jin LE, Wang XJ, Laubach M, Mazer JA, Lee D, Arnsten AF. Neuronal basis of age-related working memory decline. Nature. 2011;476:210–213. doi: 10.1038/nature10243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Romine JL, Gao X, Chen J. Aging impairs dendrite morphogenesis of newborn neurons and is rescued by 7, 8-dihydroxyflavone. Aging Cell. 2017;16:304–311. doi: 10.1111/acel.12553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang ZX, Tan L, Yu JT. Axonal transport defects in Alzheimer's disease. Mol Neurobiol. 2015;51:1309–1321. doi: 10.1007/s12035-014-8810-x. [DOI] [PubMed] [Google Scholar]
- Weingarten MD, Lockwood AH, Hwo SY, Kirschner MW. A protein factor essential for microtubule assembly. Proc Natl Acad Sci U S A. 1975;72:1858–1862. doi: 10.1073/pnas.72.5.1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu K, Zhong G, Zhuang X. Actin, spectrin, and associated proteins form a periodic cytoskeletal structure in axons. Science. 2013;339:452–456. doi: 10.1126/science.1232251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan SC, Hwang S, Rustan TD, Frey WH. Human brain tubulin purification: decrease in soluble tubulin with age. Neurochem Res. 1985;10:1–18. doi: 10.1007/BF00964768. [DOI] [PubMed] [Google Scholar]
- Young ME, Ohm DT, Dumitriu D, Rapp PR, Morrison JH. Differential effects of aging on dendritic spines in visual cortex and prefrontal cortex of the rhesus monkey. Neuroscience. 2014;274:33–43. doi: 10.1016/j.neuroscience.2014.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan A, Rao MV, Veeranna, Nixon RA. Neurofilaments at a glance. J Cell Sci. 2012;125:3257–3263. doi: 10.1242/jcs.104729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang C, Zhu Q, Hua T. Aging of cerebellar Purkinje cells. Cell Tissue Res. 2010;341:341–347. doi: 10.1007/s00441-010-1016-2. [DOI] [PubMed] [Google Scholar]


