Abstract
Chimeric antigen receptor T (CAR-T) cell therapy achieved advanced progress in the treatment of hematological tumors. However, the application of CAR-T cell therapy for solid tumors still faces many challenges. Competition with tumor cells for metabolic resources in an already nutrient-poor tumor microenvironment is a major contributing cause to CAR-T cell therapy’s low effectiveness. Abnormal metabolic processes are now acknowledged to shape the tumor microenvironment, which is characterized by increased interstitial fluid pressure, low pH level, hypoxia, accumulation of immunosuppressive metabolites, and mitochondrial dysfunction. These factors are important contributors to restriction of T cell proliferation, cytokine release, and suppression of tumor cell-killing ability. This review provides an overview of how different metabolites regulate T cell activity, analyzes the current dilemmas, and proposes key strategies to reestablish the CAR-T cell therapy’s effectiveness through targeting metabolism, with the aim of providing new strategies to surmount the obstacle in the way of solid tumor CAR-T cell treatment.
Keywords: CAR-T cell therapy, Metabolism, Tumor microenvironment, Immunotherapy, Mitochondrial fitness
Introduction
In the past decade, chimeric antigen receptor (CAR)-T cells have been a revolutionary approach in cancer therapy.[1] Currently, ten CAR-T cell therapy products have been approved by the U.S. Food and Drug Administration (FDA) and National Medical Products Administration (NMPA) in China respectively, for treating incurable hematological tumors.[2-4] Although CAR-T cell therapy has achieved great strides and has shown some clinical efficacy in patients with hematologic tumors and a subset of solid tumors, many obstacles remain in the field of tumor therapy,[5-8] such as tumor antigen heterogeneity, poor immune cell infiltration ability, immunosuppressive microenvironment, metabolic microenvironment disturbance, and T-cell depletion, which have limited further improvement in solid tumor therapy. The effectiveness of CAR-T cells therapy in solid tumor can be improved by searching for new antigens, designing multi-target CARs, overexpressing chemokine receptors and cytokines, knocking down inhibitory signaling molecules, enhancing the metabolic capacity of T cells, combining with immune checkpoint inhibitors, and epigenetic modifications.[9] With the in-depth exploration of the tumor immune system and the development of genetic engineering techniques, high-throughput gene sequencing and other technologies, strategies to improve the efficacy of immune cell therapy are constantly being developed. T cells are sentinels of CAR-T cell therapy that are tailored to identify and eliminate tumor cells in the host. Nutrient uptake and metabolism in tumors are very different from those in healthy tissues, and support fundamental cellular processes ranging from bioenergetics to biomass production and T cell fate regulation.[10,11] T cell activation and metabolic reprogramming occur simultaneously. There is growing evidence that the reduced anticancer efficacy of T cells may be caused by disruptions in intratumoral metabolic demands or metabolic reprogramming.[12] T cell persistence and anti-tumor activity are significantly increased by increasing their metabolic fitness, and the effectiveness of these metabolic drugs is being studied in clinical settings.[13,14] Therefore, it is important to pursue new approaches to modify the tumor microenvironment (TME) metabolic profile and customize the metabolic states of CAR-T cells to conquer the existing obstacles.[15] In this review, we introduce the complicated metabolic reprogramming of T cells in the tumor microenvironment and further discuss the latest developments in metabolic interventions exploited to facilitate the efficiency of CAR-T cells, to offer new perspectives on enhancing CAR-T cells therapeutic efficacy.
Overview of T cell Metabolism
Naïve T cells without antigen stimulation circulate in the body, monitoring the body’s external antigen. Once foreign antigens activate T cells, they experience rapid proliferation, triggering the effect to eliminate foreign invaders, which requires changes in metabolic function to adapt. Glucose, fatty acids, amino acids and other nutrients support T cells reaction processes, providing nutrients and energy to different stages of T cells, involving transcription and enzyme activity regulation of multiple genes. Understanding the impact of metabolic reprogramming on the fate of T cells will guide us to discover new strategies to enhance immune function[16-18] [Figure 1].
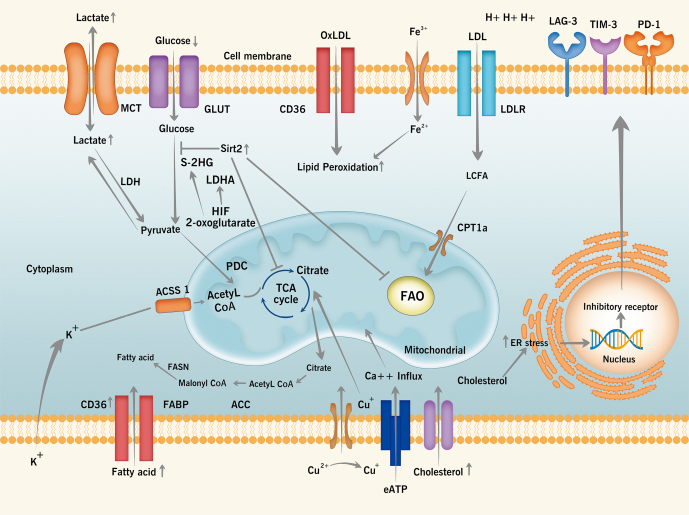
Figure 1.
T cell metabolic crosstalk in TME. In the tumor microenvironment, TCR complex and co-receptor CD28 receive stimuli that activate T cells. GLUT1 expression is upregulated, glucose is lacking in the tumor microenvironment, lactate content is high, and the cells are in an acidic environment. NAD+-dependent deacetylase (Sirt2) inhibits T cell function, lipid accumulation increases, and CD36 expression is upregulated. Cholesterol accumulation induces the expression of immune checkpoints. eATP promotes Ca+ in the cytoplasm and then enhances the mitochondrial activity. LDHA catalyzes 2-oxoglutarate to S-2HG under hypoxia to regulate T cells proliferation. Increased levels of Fe2+ results in lipid peroxidation, which induces ferroptosis. Cu+ can bind to the lipoyl protein in TCA and promote abnormal oligomerization of the lipoyl protein. Increasing potassium levels cause an elevation in the total cellular AcCoA. ACC: Acetyl-CoA carboxylase; AcCoA: Acetyl-coenzyme A; ACSS 1: Acyl-CoA synthetase short chain family member 1; CoA: Coenzyme A; CPT1a: Carnitine palmitoyl-transferase 1A; eATP: Extracellular adenosine 5’-triphosphate; ER: Endoplasmic reticulum; FABP: Fatty acid-binding protein; FAO: Fatty acid oxidation; FASN: Fatty acid synthase; GLUT: Glucose transporter; HIF: Hypoxia inducible factor-1; LAG-3: Lymphocyte activation gene 3; LCFA: Long-chain fatty acids; LDH: Lactate dehydrogenase; LDL: Low density lipoprotein; LDLR: Low density lipoprotein receptor; LDHA: Lactate dehydrogenase A; MCT: Medium-chain triglyceride; NAD: Nicotinamide adenine dinucleotide; OxLDL: Oxidized low-density lipoprotein; PD-1: Programmed death 1; PDC: Pyruvate dehydrogenase complex; S-2HG: S-2-hydroxyglutarate; TCA: Tricarboxylic acid cycle; TCR: T cell receptor; TIM-3: T cell immunoglobulin domain and mucin domain-3; TME: Tumor microenvironment.
Glucose
Distinct activation states of T-cells necessitate metabolic programs that align with their specific functional needs. Upon activation, naïve T cells quickly reestablish connections to their metabolic networks in order to fulfill the requirements of clonal expansion and diverse biochemical processes. T-cell activation is initiated by the T-cell receptor (TCR) complex and co-stimulatory receptors. Upon antigen stimulation, T cells initiate proliferation and effector function, provides energy for this process through nutrient uptake and utilization, and thus promotes anti-tumor immune responses. In the tumor microenvironment, the utilization of glucose by tumor cells imposes metabolic limitations on T cells, leading to a decrease in mechanistic target of rapamycin (mTOR) function, glycolytic ability, and interferon-γ (IFN-γ) production, which results in tumor progression.[19]
Glucose transporter 1 (GLUT1) is the primary glucose transporter protein, which maintains the glycolytic flux in naïve T cells. GLUT1 expression is lower in naïve T cells than in activated T cells. Quiescence or the transition out of quiescence may be influenced by glucose uptake.[20] Glucose is transported into the cell and broken down into two pyruvate molecules through glycolytic reactions. When T-cells are activated, pyruvate is converted to lactate to regenerate nicotinamide adenine dinucleotide (NAD+) followed by glycolytic reactions.[21] The reactant phosphoenolpyruvate (PEP), which produces pyruvate, is an important metabolic checkpoint in antitumor T cells. Ho et al[22] found that PEP plays an important role in supporting the Ca2+/nuclear factor of activated T cells (NFAT) signaling and effector functions of T cell receptors by inhibiting the activity of sarco/endoplasmic reticulum (ER) Ca2+/ATPase. Lactate, a crucial compound generated through the glycolytic breakdown of glucose, exerts intricate impacts on both tumor and T cells within the tumor microenvironment. The exuberant glycolytic metabolism of cancer cells in the tumor microenvironment generates large amounts of lactate, leading to an acidic environment for T cells in tumors, which inhibits the functioning of CD8+ T cells. Mechanistically, lactic acidosis can impair T cell function by inhibiting the T cell receptor-triggered p38 and C-Jun N-terminal kinase (JNK)/c-JUN pathways.[23] Interestingly, Wen et al[24] showed that lactate anions enhance T cell cytokine production and glycolysis and enhance antitumor activity. Feng et al[25] revealed that lactate increases the stemness of CD8+ T cells and enhances anti-tumor immunity. Lactate dehydrogenase (LDH) isozymes are tetramer of the translation products of the LDHA and LDHB genes. Chen et al[26] found that deletion of LDHA inhibited glycolysis, cell proliferation and differentiation, whereas deletion of LDHB promoted glycolysis but inhibited T-cell differentiation. In CD8+ effector T cells, the glycolytic enzyme lactate dehydrogenase A (LDHA) is induced by phosphoinositide 3-kinase (PI3K) signaling, and ablation of LDHA inhibits protein kinase B (AKT) and forkhead box 1 (Foxo1) activity, leading to decreased T cell effector function.[27] T cell metabolism is suppressed by Sirtuin-2 (Sirt2), a deacetylase that relies on NAD+. Sirt2 inhibits T cell metabolism through key enzymes involved in glycolysis, tricarboxylic acid cycle, fatty acid oxidation, and glutamine catabolism.[28]
Antitumor T cells gradually upregulate the PD-1 molecules during tumor immunity, and the action of PD-1 blocking antibodies can restore T cell function to a certain extent. Interestingly, Kumagai et al[29] found that in the low-glucose tumor microenvironment, Tregs actively take up lactic acid to promote the entry of NFAT1 into the nucleus, which enhances the expression of PD-1, while PD-1 expression by effector T cell is inhibited, and PD-1 blockade therapy activates PD-1-expressing Tregs, leading to therapeutic failure.
Reprogramming of glycolysis is a hallmark of tumors. Wu et al[30] found that deletion of two important glycolytic enzymes, GLUT1 and glucose-6-phosphate isomerase 1, resulted in enhanced tumor cell killing by cytotoxic T lymphocytes (CTL). Inactivation of GLUT1 leads to metabolic rewiring toward oxidative phosphorylation, which in turn leads to excessive reactive oxygen species (ROS) production in tumor cells, sensitive to the tumor necrosis factor alpha (TNF-α)-mediated death pathway in CTLs. Zhang et al[31] discovered that during the early recall response, glycogen serves as the primary carbon source for glycolysis. Following antigen stimulation, TCR signaling directly phosphorylates glycogen phosphorylase via lymphocyte-specific protein tyrosine kinase (LCK)-Zeta-chain-associated protein kinase 70 (ZAP70), resulting in glycogenolysis and the release of glucose-6-phosphate, which primarily supply to the cellular glycolytic response, facilitating memory T cells’ early recall response.
Lipid
Nutrition and metabolism are key factors that determine the fate of T cells, and lipids are among these key metabolites. Lipids are important components of the T cell membrane and are intermediates in cellular activities. Extracellular lipids, including fatty acids (FAs) and cholesterol, are important sources of cellular metabolic energy.[32,33] Cellular lipid biosynthesis is linked to glucose metabolism as a component of T cell activation. T cells can also be energized by fatty acid degradation, which is closely related to T cell memory differentiation.[34] Memory T cells require fatty acid oxidation to respond rapidly to restimulation.[35] The acquired immune response depends on the development of immune memory. Metabolic profiling has revealed that memory T cells mainly obtain their energy from fatty acid oxidation and oxidative phosphorylation processes, and that the switch from glycolysis to fatty acid oxidation is essential for memory CD8+ T cell development and survival.[36,37]
A common metabolic change in the tumor microenvironment is increased lipid accumulation, where accumulation of ultra-long-chain fatty acids or cholesterol leads to dysfunction of CD8+ tumor-infiltrating lymphocytes (TIL). CD36, also known as fatty acid translocase, is a transporter of free fatty acids and oxidized lipids such as oxidized low-density lipoproteins (OxLDLs) and phosphocholine-containing phospholipids. In the tumor microenvironment, CD36 expression is upregulated in CD8+ T cells, which results in the upregulation of OxLDL uptake and lipid peroxidation rates, whereas knockdown of CD36 results in decreased OxLDL uptake and lipid peroxidation in CD8+ TIL, increased cytokine production and tumor control.[38,39] In addition, another study showed that CD8+ T cells in the tumor microenvironment ingested fatty acids from the TME via CD36, inducing lipid peroxidation and ferroptosis, as well as a decrease in anti-tumor function; blocking CD36 or inhibiting ferroptosis effectively restored the anti-tumor activity of CD8+ T cells, and the combination of targeting CD36 with anti-PD-1 antibodies had a more potent anti-tumor activity.[40]
Linoleic acid (LA) modulates CD8+ T cell activity by improving metabolic adaptations, stimulating the formation of a memory T cell phenotype with superior effector function, and LA treatment promotes the formation of endoplasmic reticulum-mitochondrial contacts (MERC), which in turn facilitates calcium signaling, mitochondrial energetics, and CTL effector function, leading to enhanced antitumor activity.[41] Lysosomal acid lipase breaks down cholesteryl ester (CE) and triacylglycerol (TAG) to produce free FA and cholesterol in the lysosomes of cells, supporting the metabolic reprogramming of CD8+ memory T cells.[42] A significant proportion of the material obtained from glucose metabolism after T-cell activation is used to synthesize fatty acids de novo. Upregulation of de novo fatty acid synthesis initiated by mechanistic target of rapamycin complex 1 (mTORC1) through induction of the activity of sterol-responsive element-binding proteins (SREBPs) also regulates cholesterol metabolism, and in effector T-cells, the SREBPs induce the expression of acetyl coenzyme A carboxylase, fatty acid synthase and hydroxy methyl glutaryl coenzyme A reductase, which are the rate-limiting enzymes for synthesizing fatty acids and cholesterol, respectively. Fatty acid synthesis plays an important role in T-cell activation.[43]
How the hypoxic and low-glucose tumor microenvironment constrains T cells from exerting their effector functions at the metabolic level is not less well understood. Zhang et al[44] found that under hypoxic and low-glucose conditions, CD8+ TIL enhanced peroxisome proliferator-activated receptor (PPAR)-α signaling and fatty acid (FA) catabolism, and this metabolic alteration partially retained the CD8+ TIL effector function; further promotion of fatty acid catabolism could increase the ability of CD8+ TIL to retard tumor growth. Tumor-infiltrating CD8+ T cells often lose function, however the underlying mechanisms are not fully understood. Ma et al[45] found that cholesterol in the tumor microenvironment induced the upregulation of immune checkpoint expression, including lymphocyte activation gene 3 (LAG-3), 2B4, T cell immunoglobulin domain and mucin domain-3 (TIM3), and programmed death 1 (PD-1), and depletion of CD8+ T cells by increasing endoplasmic reticulum stress in CD8+ T cells. The endoplasmic reticulum stress sensor, X-box binding protein 1 (XBP1), was activated and regulated the transcription of the inhibitory receptor. Inhibition of XBP1 or reduction of cholesterol in CD8+ T cells was effective in restoring anti-tumor activity, which has important implications for the recovery of immunosuppressed CD8+ T cells and provides ideas for new strategies to enhance immunotherapy.[45]
Amino acids
Amino acid metabolism of T cells and their crosstalk affects tumor immunity and therapeutic efficacy in patients with tumors, with T cell maturation, differentiation, and function largely reliance on amino acid metabolism.[46] L-arginine is a multipurpose amino acid that is essential for the synthesis of proteins and a precursor to several metabolites, such as nitric oxide (NO) and polyamines, which are critical for T-cell immunity. Analysis of the metabolome and proteome profiles of activated human T-cells by high-resolution mass spectrometry revealed that arginine metabolism underwent a major alteration, with a decrease in intracellular L-arginine concentration, and that the overall metabolic level of T cells could be altered by adding L-arginine. Three transcriptional regulatory factors (translin [TSN], Bromodomain adjacent to zinc finger domain 1B [BAZ1B], and PC4 and SRSF1 interacting protein 1 [PSIP1]) sensed intracellular L-arginine levels and promoted T-cell survival. These included a shift in activated T cell metabolism from primarily glycolytic metabolism to oxidative phosphorylation, up-regulation of several intermediates of the mitochondrial tricarboxylic acid cycle, enhancement of mitochondrial spare respiratory capacity (SRC) accompanied by the generation of more central memory-like T cells, and enhanced anti-tumor activity in an antitumor mouse model.[47] Tumor-associated arginine deprivation is the primary mechanism via which tumor immunity eludes T cell-mediated anti-tumor immune responses. Proline and arginine metabolism is important for the anticancer activity of T cells, although the precise function of proline metabolism in T cell function is unknown, new research has proven the crucial regulatory role of proline metabolism in T cells.[48]
Activated T cells upregulate enzymes of the serine, glycine, one-carbon metabolic network and rapidly increase the processing of serine into one-carbon metabolism, even when glucose is sufficient. Extracellular serine is required for optimal T cell expansion and is converted to glycine by serine hydroxy methyl transferase, which helps drive the folate cycle by transferring methyl groups from serine to tetrahydrofolate, the active carrier of the one-carbon units used for the de novo synthesis of nucleotides. Serine restriction impairs T-cell expansion after activation.[49] Serine stimulates glutathione (GSH) synthesis and access to the carbon metabolic network necessary for effector T cell (Teff) responses. Glutamine restriction during in vitro T cell activation promotes memory CD8+ T cell differentiation, and activated T cells upregulate glutamine metabolism to support immune responses and cell fate decisions. Extracellular signal-regulated kinase (ERK) activity is necessary to establish a connection to TCR signaling in order to induce glutamine uptake and metabolism.[50] Moreover, glutamate levels can control T cell growth and cytokine production; after TCR activation, both CD4+ and CD8+ T cells upregulate glutamate receptors, which is correlated with higher activation molecule expression and IFN-γ production; on the other hand, high extracellular glutamate concentrations (>100 μmol/L) can prevent T cell activation.[14]
The mechanism by which amino acids play a role in T cells can also directly regulate the activation of T cell signaling pathways in addition to their own metabolic effects. Increasing the level of the non-essential amino acid aspartic acid strongly enhances the activation and anti-tumor response of CD8+ T cells. In contrast, restricting the uptake or lowering the intracellular level of aspartic acid impairs the activity and response of CD8+ T cells. Aspartic acid binds LCK, a member of the SRC-family protein, and increases LCK activity and T cell responsiveness. This indicates even more that LCK is a natural aspartate sensor that alerts T cells to aspartate sufficiency.[51] Roy et al[52] used metabolomics to discover that methionine is quickly absorbed by activated T cells and acts as the main building block for the synthesis of S-adenosyl-L-methionine (SAM), a universal methyl donor. Methionine is an essential molecule for T cell histone methylation and SAM production. Methionine restriction inhibits the growth and function of T helper 17 (Th17) cells by lowering histone H3K4 methylation (H3K4me3) in the promoter region of critical genes involved in Th17 cell proliferation and cytokine generation. Ammonia is processed in the liver through the urea cycle, but recent studies have shown that CD8+ memory T cells also utilize the urea and citrulline cycles to process ammonia by mobilizing mitochondrial arginase 2 (ARG2) to catalyze the production of urea from arginine, and that carbamoyl phosphate (CP) synthase-1 regulation may play a critical role in ammonia processing and affect the formation of memory T cells, and that, as in hepatocytes, all ammonia-detoxifying enzymes are present in memory T cells and are lacking in the initial and effector T cells.[53]
Others
In addition to the previously indicated rationales, metabolites present in the TME may also affect lymphocyte infiltration and function. Extracellular nucleotides are one of the most abundant products and can be metabolized to produce adenosine by ectoenzymes, such as extracellular adenosine 5’-triphosphate (eATP) and NAD+ in the TME. Extracellular adenosine (eADO) inhibits the function of cytotoxic T cells in cancers.[54,55] Another characteristic of the TME is hypoxia, which results in an increase in hypoxia-inducible factor-1 (HIF-1) transcriptional complexes in activated CD8+ T cells to produce S-2-hydroxyglutarate (S-2HG), which may mediate T cell differentiation, anti-tumor activity, and proliferation.[56,57] In addition, immunomodulatory products secreted during tumor cells death can impair the T-cell activity. Prostaglandin E2 (PGE2) released by tumor cells during ferroptosis, a unique type of mediated cell death, has been regarded as an immunosuppressive molecule beneficial to tumor evasion.[58,59] Cuproptosis is a recently reported form of cell death caused by excess copper. Intracellular copper promotes the degradation of Fe-S cluster proteins and protein toxicity by stimulating sulfur-acylation and aggregation process of mitochondria-related proteins, eventually leading to cell death.[60] Higher extracellular potassium concentration triggered by tumor necrosis can reduce Akt-mTOR phosphorylation activated by T-cell receptors to depress the cytotoxic function of CD8+T cells.[61,62] Nitric oxide (NO) produced by inducible nitric oxide synthase (iNOS) in myeloid-derived suppressor cells (MDSCs) may be an essential contributor to the suppression of T-cell proliferation and function.[63,64]
Strategies for Improving the Effectiveness of CAR-T cells by Metabolic Interventions
Despite the fact that CAR-T cell therapy has exhibited promise in treating hematologic tumors, obstacles remain in solid tumors.[65] The accumulation of immunosuppressive metabolites in TME of solid tumor, dynamically changing metabolic flow, and the competition for nutrients from tumor cells leading to an insufficient supply of CAR-T cells are the vital elements. Therefore, rational and effective metabolic intervention approaches are needed to improve the metabolic adaptability and anti-tumor ability of CAR-T cells in TME [Figure 2].
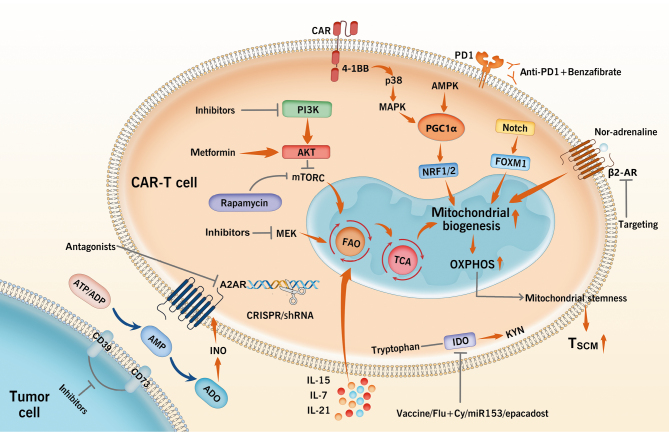
Figure 2.
Metabolic interventions for improving CAR-T cells efficacy in TME. 4-1BB co-stimulation signaling or AMPK or PGC1α agonist activate PGC1α-NRF1/2 and Notch/FOXM1 signaling to facilitate mitochondrial biogenesis and OXPHOS to increase mitochondrial stemness leading to TSCM population elevation. Treatment with the inhibitors of PI3K or mTORC or MEK or AKT activator metformin could also promote FAO and TCA cycle to increase mitochondrial biogenesis. Blocking the binding of β2-AR and Nor-adrenaline could also increase mitochondrial biogenesis. Cytokines such as IL-15, IL-7 and IL-21 could also accelerate FAO and OXPHOS rate. Inhibition of IDO with the vaccine or Flu+Cy or epacadost or overexpression of miR153 could increase tryptophan and restore the activity of CAR-T cells. Treatment with CD39 or CD73 inhibitors could prevent conversion of ATP/ADP to AMP and INO and further block the binding of INO to A2AR expressed on CAR-T cells. Blockade of A2AR using genetic shRNA or CRISPR/Cas9, or distinct antagonists enhanced the anti-tumor efficiency of CAR-T cells. A2AR: A2A receptor; ADO: Adenosine; ADP: Adenosine 5’-diphosphate; AKT: Protein kinase B; AMP: Adenosine monophosphate; AMPK: AMP-activated protein kinase; ATP: Adenosine 5’-triphosphate; β2-AR: β2-adrenergic receptor; CAR: Chimeric antigen receptor; Cy: Cyclophosphamide; FAO: Fatty acid oxidation; Flu: Fludarabine; FOXM1: Forkhead box M1; IDO: Indoleamine-2,3-dioxygenase; IL: Interleukin; INO: Inosine; KYN: Kynurenine; MAPK: Mitogen-activated protein kinase; MEK: Mitogen-activated extracellular signal-regulated kinase; miR: MicroRNA; mTORC: Mechanistic target of rapamycin complex; NRF: Nuclear factor erythroid 2-related factor; OXPHOS: Oxidative phosphorylation; PD1: Programmed death 1; PGC1α: Peroxisome proliferator-activated receptor-γ coactivator (PGC)-1α; PI3K: Phosphoinositide 3-kinase; shRNA: Short hairpin RNA; TCA: Tricarboxylic acid cycle; TME: Tumor microenvironment; Tscm: Stem cell-like memory T cell.
Enhancing CAR-T Cell effectiveness by decreasing the inhibitory effects of immunosuppressive metabolites
Elevated kynurenine (Kyn) levels are frequently associated with poor prognosis and limit effector T cells function. Indoleamine 2,3-dioxigenase 1 (IDO1) and tryptophan 2,3-dioxygenase (TDO) are the key enzymes that catalyze the conversion of tryptophan to Kyn.[66] IDO1 overexpression mediates the inhibition of CD8+ T cells infiltration and function.[67-70] It has been shown that the IDO1 vaccine can strengthen the major histocompatibility complex-II (MHC-II) signal and facilitate the differentiation of the central memory T cells (TCM) CD44hiCD62Lhi population cells. The percentage of TCM that displays increased self-renewal, prolonged survival, and reduced exhaustion is a crucial factor in determining the survival and anti-tumor function of CAR-T cells. An increased TCM percentage in CAR-T cells has been shown to enhance metabolic adaptability and long-term anti-tumor response.[71-74] IDO has been reported to inhibit CD19-CART activity, by inducing the Kyn pathway, and fludarabine and cyclophosphamide could inhibit IDO expression in lymphoma cells before CAR-T cells administration in IDO-positive lymphoma.[67] Huang’s team has demonstrated that microRNA (miR)-153 inhibited the IDO1 expression in colon cancer cells by targeting its 3’ non-coding region, and enhanced the anti-tumor effects of CAR-T cells both in in vitro and in vivo studies.[75] Furthermore, epacadostat (INCB024360), an IDO1 inhibitor that has been utilized in clinical trials over the years, has not shown successful results due to the complicated solid tumor microenvironment. In our previous study, we have demonstrated that Kyn suppressed the cytokine secretion and cytotoxic activity of CAR-T cells. With the utilizing of epacadostat-loaded hyaluronic acid-modified nanomaterial graphene oxide (HA-GO) nanosheets, CAR-T cells exhibit a more powerful anti-tumor ability.[76] To overcome the immunosuppressive impact of TME with high concentrations of Kyn, Yang et al[77] have modified CAR-T cells through overexpression (OE) of KYUN. They found that KYUN-OE CAR-T cells showed a superior anti-tumor activity even in the immunosuppressive TME with high Kyn.[77]
Adenosine is also a pivotal metabolite in the TME that induces immunosuppression and mediates immune escape. Adenosine inhibits T cell function. CD39 and CD73 are the key enzymes which are responsible for the conversion of adenosine 5’-triphosphate (ATP) to adenosine diphosphate (ADP) and adenosine monophosphate (AMP), which eventually generates extracellular adenosine. More importantly, when adenosine binds to the adenosine to adenosine 2A receptors (A2AR), which are expressed on the surface of T cells, the activation of TCR signaling can be inhibited,[78] blocking the cytotoxic function, cytokine secretion, and infiltration of CD8+ T cells,[79,80] and inducing T cells exhaustion.[81] An inhibitory effect of A2AR on human epidermal growth factor 2 (HER2)-CAR-T cells in patients with primary melanomas has been reported by Paul et al,[82] suggesting that A2AR is a potential metabolic target for CAR-T cell therapy.[82] Blockade of A2AR using genetic short hairpin RNA (shRNA),[83] or clustered regularly interspaced short palindromic repeats (CRISPR)/Cas 9,[82,84] or distinct pharmacological compounds[85-87] profoundly enhanced IFN-γ production by CAR-T cells and enhanced activation of CD8+ and CD4+ CAR-T cells both in hematological and solid tumors. However, due to the complicated TME, the delivery of A2AR antagonists is limited. To overcome this limitation, Natnaree et al[88] reported a new drug delivery nanotechnology that enhanced the validity of CAR-T cells through chemically conjugating A2AR antagonist-loaded cross-linked multilamellar liposomal vesicles (cMLVs) ex vivo prior to systemic administration, to facilitate CAR-T cells infiltration into the deep immunosuppressive TME. Adenosine deaminase is an enzyme involved in converting adenosine into inosine, which is necessary for the immune system function.[89] Adenosine deaminase overexpression in CAR-T cells augments the exhaustion resistance, expansion, infiltration of CAR-T cells and remodels the TME to favor antitumor immunity.[90] It is noteworthy that that A2AR antagonists have been tested phase III clinical trials for Parkinson’s disease and ongoing phase I trials for cancer. Combining CAR-T cells with A2AR antagonists or CD73/CD39 blockers may improve the efficacy of clinical treatments.
In the TME, D-2-hydroxyglutarate (D2HG) is also recognized as an immunosuppressive factor with the characteristic that impaired activated T cell function.[91,92] The inducible enzyme D-2-hydroxyglutarate dehydrogenase (D2HGDH) transforms D2HG into endogenous metabolite 2-oxoglutarate. Over-expression of D2HGDH in CAR-T cells exhibited increased anti-tumor effectiveness and improved overall survival in the metabolically immunosuppressive TME.[93]
Promoting CAR-T Cell function through enhanced metabolite uptake
Low arginine levels in the TME affect the production of the arginine resynthesis enzymes ornithine transcarbamylase (OTC) and argininosuccinate synthase (ASS), which are essential for the proper function of CAR-T cells. Based on this principle, ASS or OTC modified CAR-T cells were designed and exhibited an effective increasing proliferation and promoted solid tumor clearance without influencing the cytotoxicity or exhaustion of CAR-T cells.[94] Apart from arginine, proline metabolism is also crucial in the T cell anti-tumor response. Proline dehydrogenase 2 (PRODH2), an extremely tissue-specific enzyme that is normally not expressed in immune cells but is mostly expressed in the liver and kidney, catalyzes the trans-4-hydroxy-L-proline catabolism as the first step in the catabolism of proline. Chen et al[48] exploited a dead-guide RNA (dgRNA)-based CRISPR activation screen method in primary T cells, and subsequently found that overexpression of PRODH2 improved the ability of CAR-T cells to fight tumor cells in multiple cancer types through reprogramming proline metabolism and facilitating oxidative phosphorylation (OXPHOS) and mitochondrial fitness.[47] This suggests that targeting proline metabolism is an important means of strengthening CAR-T cells function.
Tumor cells can regulate their own metabolism and adapt to a metabolically dysregulated microenvironment to support their growth.[95] Tumor cells rely primarily on aerobic glycolysis to consume large amounts of glucose and ingest large amounts of glutamine, which results in the accumulation of lactic acid and the formation of a hypoxic, acidic tumor microenvironment.[96] It is known that tumor cells compete with T cells in the TME for glucose uptake. Given that T-cell activation entails the fast development of aerobic glycolysis and that the metabolic profiles of glycolytic T-cells are linked to improved effector T-cell function,[97-100] the limitation of glucose uptake in the tumor microenvironment results in the incapable function of CAR-T cells. The contribution of glucose to CAR-T cell activity was supported by Cribioli et al.[101] GLUT3 possesses an enhanced transport capacity in addition to a greater affinity for glucose.[22,102,103] Primary CD8+ T cells overexpressing GLUT3 showed improved glucose uptake, and greater glycogen and fatty acid storage. It has also been linked to improved stress tolerance, decreased reactive oxygen species levels, and mitochondrial fitness,[101] suggesting that GLUT3 reinforcement offers a useful tactic to enhance metabolic fitness and maintain effective activity of CAR-T cells. Products of CAR-T cells with high glycolytic metabolism and low terminal differentiation were linked to better outcomes in another cohort of patients with chronic lymphocytic leukemia, which highlights the link between metabolism and outcomes of CAR-T products.[104] Except the modification of CAR-T cells with genetically engineered key metabolic enzymes,[13] controlling the nutrient supplementation during CAR-T cells culturing is an alternative approach. Gross et al[105] have found that replacing the carbon source of culture media from glucose to galactose could increase mitochondrial activity in CAR-T cells, leading to an improved anti-tumor efficacy. Inosine (INO) is the metabolite of adenosine in the TME. INO supplement could also enhanced CAR-T cells function by inducing metabolic reprogramming, diminishing glycolysis and increasing oxidative phosphorylation during scalable manufacturing of CAR-T cells.[106]
Promoting CAR-T cell function by enhancing mitochondrial metabolism
The development of TCM cell subsets, the persistence of CAR-T cells following infusion, and the proliferation, activation, and anti-tumor potential of CAR-T cells are also significantly influenced by mitochondrial dynamics.[107,108] Jaco et al[109] reported that increased mitochondrial mass and biogenesis were observed in CD19-directed CAR-T cells derived from chronic lymphocytic leukemia patients with a complete response, which showed good clinical outcomes compared with non-responders.
Peroxisome proliferator-activated receptor-γ coactivator-1α (PGC-1α) is triggered by enhanced energy demand, which is activated by phosphorylation following AMP-activated protein kinase (AMPK) activation and acts through the binding and activation of NRF1 and NRF2, and involved in the control of excessive reactive oxygen species generated in response to enhanced mitochondrial activity.[110-112] T cell differentiation is regulated by glycolysis, which involves pyruvate oxidation. However, unlike glycolysis, oxidative phosphorylation allows T cells to develop into memory stem cells, which have more potent anti-tumor properties. The mitochondrial pyruvate carrier (MPC), which is expressed on the inner mitochondrial membrane, is the only channel through which pyruvate enters the mitochondria. Targeted inhibition of MPC can aid in the differentiation of memory T cells. It has been demonstrated that adding MPC inhibitors to nutrient-rich cultures during the production of CAR-T cells increases the anti-tumor effects of these cells, accompanied by increased uptake of glutamine and fatty acids.[15] PGC-1α expression is decreased in resting CD8+ T cells in chronic lymphocytic leukemia (CLL) patients. Mitochondria and ROS are the cardinal metabolic features of TCM induction,[113] which is essential for the efficiency of CAR-T cells. Increased PGC-1α expression shows sustained mitochondrial structural and functional fitness and leads to the formation of TCM in the antitumor response.[114] Overexpression of PGC-1α raised CD8+ T cell mitochondrial mass and enhanced tumor-killing ability in a mouse model of melanoma.[115] Combining benzafibrate, a PGC1α agonist, with anti-PD-1 treatment resulted in increased OXPHOS and decreased T-cell apoptosis. Konstantinos et al[116] also found that in CAR-T cells, a modified form of PGC-1α can efficiently stimulate mitochondrial biogenesis, metabolically reprogram CAR-T cells, and result in a substantially improved antitumor efficiency in solid tumors. The costimulatory domain of CAR molecule determines the mitochondrial biogenesis of CAR-T cells. Previous studies demonstrate that 4-1BB costimulatory domain of CD8+ T cells engages in PGC-1α signaling via p38-mitogen-activated protein kinase (MAPK) pathway activation and leads to increased mitochondrial capacity and aids in overcoming the tumor microenvironment’s immunosuppressive metabolic landscape.[117] Complement C1q binding protein (C1QBP), another mitochondrial protein, was also confirmed to play an indispensable role in the antitumor activity of CAR-T cells by maintaining mitochondrial biogenesis and morphology through AMPK-PPAR1 and the mitochondrial dynamics protein dynamin-related protein 1 signaling.[118] β2-adrenergic receptor (β2-AR) is another potential targetable checkpoint in boosting the efficiency of CAR-T cell therapy via modulating metabolic reprogramming,[119] especially in the mitochondrial metabolism. The inhibition of β2-AR signaling increases mitochondrial biogenesis, respiration capacity, and membrane potential.[120-122] As a key metabolic factor which controls the cellular oxidative process, IDH2 is located in mitochondria, and catalyzes the reversible conversion of isocitric acid to α-ketoglutaric acid in the tricarboxylic acid cycle, using NADP+ as a cofactor. Blocking IDH2 activity by gene knockdown or targeted drugs can remodel the central carbon metabolism pattern and epigenetic regulation of CAR T cells and increase the anti-tumor ability of CAR T cells. Si et al[123] have reported that by inhibiting the activity of IDH2 in CAR T cells with Enasidenib, an FDA-approved clinical drug for leukemia, glucose utilization was strongly diverted into the pentose phosphate pathway (PPP), which provides antioxidant capacity and reduces CAR-T cell exhaustion and support acetyl-CoA-mediated epigenetic activation of memory differentiation of CAR-T cells, particularly under nutrient-restricted conditions. This study mechanistically resolved the mechanism of mitochondrial key metabolic steps on the fate decision of CAR-T cells, and provided a safety-guaranteed intervention strategy to enhance the metabolic adaptability and therapeutic efficacy of CAR-T cells. In another study, the researchers have reported that deletion or pharmacological inhibition of protein tyrosine phosphatase, mitochondrial 1 (PTPMT1) significantly reduces the development and clonal expansion of CD8+ effector T cells. In addition, PTPMT1 deletion accelerated CD8+ T cells dysfunction, leading to increased tumor growth. Mechanistically, PTPMT1 deletion severely affected pyruvate utilization, resulting in enhanced fatty acid utilization. Overall, this study reveals the important role of PTPMT1 in promoting mitochondrial carbohydrate utilization and that mitochondrial flexibility in energy source selection is critical for anti-tumor immunity of CD8+ T cells. These results have important implications for CAR-T cells therapeutic efficacy.[124]
During the manufacturing process, cytokines are crucial for the differentiation of CAR-T cells. Various cytokines, such as IL-2, IL-7, and IL-15 are frequently employed to keep CAR-T cells expanding, differentiating, and functioning.[125] Cui et al[126] found that IL-7 favor memory CD8+ T cells to express water/glycerol channel protein aquaporin-9 (AQP9). Long-term memory CD8+ T cell survival depends on AQP9, and its absence inhibits glycerol entry into memory CD8+ T cells for fatty acid esterification and triglyceride (TAG) synthesis and storage. Memory T cell survival is restored by rescuing TAG synthesis, and long-term IL-7-mediated memory T cell survival depends on TAG synthesis. IL-15 regulates mitochondrial SRC and oxidative metabolism by facilitating the CPT1a expression and mitochondrial biogenesis, which in turn maintains the viability of memory CD8+ T cells.[127] Meanwhile, IL-15 preserved CAR-T cells in a T memory stem cell (TSCM)-less differentiated state, mainly expressing CD62L+ CD45RA+ CC chemokine receptor 7 (CCR7)+, and showed low expression of exhaustion molecules, high anti-apoptotic capacity, and proliferative activity. IL-15 promotes the expression of glycolytic enzymes mainly by inhibiting mTORC1 activity and increasing mitochondrial fitness. Rapamycin, an mTORC1 inhibitor, or the AMPK activator metformin, restores the phenotypic features of CAR-T cells by intensifying the rates of fatty acid oxidation and OXPHOS.[128] IL-10 has the function of reprogramming T cell metabolism, and IL-10-Fc fusion protein can significantly enhance the OXPHOS metabolism of intratumor T cells and effectively enhance the proliferation and effector function of terminally depleted T cells, which has great potential for the treatment of solid tumors.[129] In a recent report, the investigators constructed IL-10-expressing CAR-T cells that prevented T cells exhaustion in the TME through sustained mitochondrial fitness and increased OXPHOS in a mitochondrial pyruvate carrier-dependent manner, leading to an enhanced proliferative capacity and effector function of CAR-T cells.[130] There are three ongoing clinical trials (NCT06120166, NCT05715606, and NCT05747157) evaluating the safety and effectiveness of IL-10-producing CAR T cells in patients with relapsed and/or refractory diffuse large B-cell lymphoma or B-cell acute lymphoblastic leukemia.
Other signaling pathways, such as NOTCH-FOXM1, transfer conventional CAR-T cells to those with TSCM characteristics by promoting mitochondrial biogenesis and fatty acid oxidation.[131-134] Inhibition of MEK1/2 signaling was found to maintain T cells activation and proliferation,[135] ameliorate the tumor-killing efficiency of GD2-CAR-T cells,[136] and induce TSCM, which has enhanced multipotency and proliferative capacity achieved by enhancing mitochondrial biogenesis and FAO,[137] indicating that enhancing CAR-T cells treatment may potentially benefit from focusing on MEK1/2 signaling. Idelalisib, the first PI3K inhibitor approved by FDA, and other PI3K inhibitors prolong the survival of CAR-T cells and increase their effectiveness in killing tumor cells in vivo by keeping the cells less differentiated in vitro without affecting their activation.[138-140] Pharmacological inhibition of the PI3Kδ pathway during the CAR-T cell production process may also improve the differentiation of CAR-T cells into CD8+ CAR-T cells with stemness characteristics. This is predicated on the increased enrichment of transcription factors Tcf-1 and Lef-1, exhibiting metabolic plasticity following tumor antigen recognition, as evidenced by enhanced glucose uptake and mitochondrial function.[141] In the meantime, encouraging the expression and epigenetic reprogramming of the mitochondrial fusion proteins mitofusins1 and 2 (MFN1/2) of CAR-T cells enables an increase in the overall mitochondrial cross-sectional area.[142]
Conclusion
Regarding the effectiveness and safety of CAR-T cell treatment in solid tumors, there are still a lot of unanswered questions. Concurrently, a series of metabolic interventions have been investigated both in preclinical and clinical studies of CAR-T cells to strengthen its efficiency. However, it is important to consider the gap between the basic research and clinical success. More interventions need to be explored and confirmed for these patients. With the development of single-cell sequencing, multi-omics, and especially spatial multi-omics technologies, it is beneficial for scientists and clinicians to analyze specimens from patients undergoing clinical CAR-T cell therapy to explore the changes in metabolites in real-world patients with effective and ineffective CAR-T cell therapies. Therefore, more innovative and effective strategies for targeting metabolisms can be developed while ensuring patient safety. By using innovative engineering techniques and combination therapies, we believe that CAR-T cells will overcome various physiological constraints. We remain convinced of the immense promise of CAR-T cell therapy to offer cancer patients a long-term, durable remission. Numerous theoretical applications exist, and the platform is robust.
Funding
This work was supported by grants from National Key Research and Development Program Intergovernmental Key Project for International Science and Technology Innovation Cooperation (No. 2022YFE0141000), Natural Science Foundation of China (No. 82203548), China Postdoctoral Science Foundation Project (No. 2022M712894), Medical Science and Technology Project of Henan Province (No. LHGJ20220385), and Major science and technology project of Henan Province (No. 221100310100).
Conflicts of interest
None.
Footnotes
Shasha Liu and Yuyu Zhao contributed equally to this work.
How to cite this article: Liu SS, Zhao YY, Gao YX, Li F, Zhang Y. Targeting metabolism to improve CAR-T cells therapeutic efficacy. Chin Med J 2024;137:909–920. doi: 10.1097/CM9.0000000000003046
References
- 1.Baker DJ, Arany Z, Baur JA, Epstein JA, June CH. CAR T therapy beyond cancer: the evolution of a living drug. Nature 2023;619:707–715. doi: 10.1038/s41586-023-06243-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Labanieh L, Mackall CL. CAR immune cells: design principles, resistance and the next generation. Nature 2023;614:635–648. doi: 10.1038/s41586-023-05707-3. [DOI] [PubMed] [Google Scholar]
- 3.Liu S, Zhang Y. Challenges and interventions of chimeric antigen receptor-T cell therapy in solid tumors. Chin J Cancer Res 2023;35:239–244. doi: 10.21147/j.issn.1000-9604.2023.03.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.The NMPA approved the Naki Orensei Injection from He Yuan Biotechnology (Tianjin) Co. for the treatment of relapsed or refractory B-cell acute lymphoblastic leukemia in adults. Available from: https://www.nmpa.gov.cn/zhuanti/cxylqx/cxypxx/20231108092415187.html. [Accessed on December 2, 2023].
- 5.Tian Y, Li Y, Shao Y, Zhang Y. Gene modification strategies for next-generation CAR T cells against solid cancers. J Hematol Oncol 2020;13:54. doi: 10.1186/s13045-020-00890-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cappell KM, Kochenderfer JN. Long-term outcomes following CAR T cell therapy: what we know so far. Nat Rev Clin Oncol 2023;20:359–371. doi: 10.1038/s41571-023-00754-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yang Z, Wang Y. Clinical development of chimeric antigen receptor-T cell therapy for hematological malignancies. Chin Med J (Engl) 2023;136:2285–2296. doi: 10.1097/CM9.0000000000002549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang Y Xu Y Dang X Zhu Z Qian W Liang A, et al. Challenges and optimal strategies of CAR T therapy for hematological malignancies. Chin Med J 2023;136:269–279. doi: 10.1097/CM9.0000000000002476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li X, Li W, Xu L, Song Y. Chimeric antigen receptor-immune cells against solid tumors: Structures, mechanisms, recent advances, and future developments. Chin Med J 2023;doi: 10.1097/CM9.0000000000002818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Makowski L, Chaib M, Rathmell JC. Immunometabolism: From basic mechanisms to translation. Immunol Rev 2020;295:5–14. doi: 10.1111/imr.12858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Finley L. What is cancer metabolism. Cell 2023;186:1670–1688. doi: 10.1016/j.cell.2023.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Franco F, Jaccard A, Romero P, Yu YR, Ho PC. Metabolic and epigenetic regulation of T-cell exhaustion. Nat Metab 2020;2:1001–1012. doi: 10.1038/s42255-020-00280-9. [DOI] [PubMed] [Google Scholar]
- 13.Peng JJ, Wang L, Li Z, Ku CL, Ho PC. Metabolic challenges and interventions in CAR T cell therapy. Sci Immunol 2023;8:eabq3016. doi: 10.1126/sciimmunol.abq3016. [DOI] [PubMed] [Google Scholar]
- 14.Li X, Wenes M, Romero P, Huang SC, Fendt SM, Ho PC. Navigating metabolic pathways to enhance antitumour immunity and immunotherapy. Nat Rev Clin Oncol 2019;16:425–441. doi: 10.1038/s41571-019-0203-7. [DOI] [PubMed] [Google Scholar]
- 15.Wenes M Jaccard A Wyss T Maldonado-Pérez N Teoh ST Lepez A, et al. The mitochondrial pyruvate carrier regulates memory T cell differentiation and antitumor function. Cell Metab 2022;34:731–746.e9. doi: 10.1016/j.cmet.2022.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pearce EL, Poffenberger MC, Chang CH, Jones RG. Fueling immunity: insights into metabolism and lymphocyte function. Science 2013;342:1242454. doi: 10.1126/science.1242454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ho PC Bihuniak JD Macintyre AN Staron M Liu X Amezquita R, et al. Phosphoenolpyruvate Is a Metabolic Checkpoint of Anti-tumor T Cell Responses. Cell 2015;162:1217–1228. doi: 10.1016/j.cell.2015.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhang Z, Liu S, Zhang B, Qiao L, Zhang Y, Zhang Y. T Cell Dysfunction and Exhaustion in Cancer. Front Cell Dev Biol 2020;8:17. doi: 10.3389/fcell.2020.00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chang CH Qiu J O’Sullivan D Buck MD Noguchi T Curtis JD, et al. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression. Cell 2015;162:1229–1241. doi: 10.1016/j.cell.2015.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chapman NM, Boothby MR, Chi H. Metabolic coordination of T cell quiescence and activation. Nat Rev Immunol 2020;20:55–70. doi: 10.1038/s41577-019-0203-y. [DOI] [PubMed] [Google Scholar]
- 21.Palmer CS, Ostrowski M, Balderson B, Christian N, Crowe SM. Glucose metabolism regulates T cell activation, differentiation, and functions. Front Immunol 2015;6:1. doi: 10.3389/fimmu.2015.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Macintyre AN Gerriets VA Nichols AG Michalek RD Rudolph MC Deoliveira D, et al. The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab 2014;20:61–72. doi: 10.1016/j.cmet.2014.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang ZH, Peng WB, Zhang P, Yang XP, Zhou Q. Lactate in the tumour microenvironment: From immune modulation to therapy. EBioMedicine 2021;73:103627. doi: 10.1016/j.ebiom.2021.103627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wen J Cheng S Zhang Y Wang R Xu J Ling Z, et al. Lactate anions participate in T cell cytokine production and function. Sci China Life Sci 2021;64:1895–1905. doi: 10.1007/s11427-020-1887-7. [DOI] [PubMed] [Google Scholar]
- 25.Feng Q Liu Z Yu X Huang T Chen J Wang J, et al. Lactate increases stemness of CD8 + T cells to augment anti-tumor immunity. Nat Commun 2022;13:4981. doi: 10.1038/s41467-022-32521-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen X, Liu L, Kang S, Gnanaprakasam JR, Wang R. The lactate dehydrogenase (LDH) isoenzyme spectrum enables optimally controlling T cell glycolysis and differentiation. Sci Adv 2023;9:eadd9554. doi: 10.1126/sciadv.add9554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Xu K Yin N Peng M Stamatiades EG Shyu A Li P, et al. Glycolysis fuels phosphoinositide 3-kinase signaling to bolster T cell immunity. Science 2021;371:405–410. doi: 10.1126/science.abb2683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hamaidi I Zhang L Kim N Wang MH Iclozan C Fang B, et al. Sirt2 Inhibition Enhances Metabolic Fitness and Effector Functions of Tumor-Reactive T Cells. Cell Metab 2020;32:420–436.e12. doi: 10.1016/j.cmet.2020.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kumagai S Koyama S Itahashi K Tanegashima T Lin YT Togashi Y, et al. Lactic acid promotes PD-1 expression in regulatory T cells in highly glycolytic tumor microenvironments. Cancer Cell 2022;40:201–218.e9. doi: 10.1016/j.ccell.2022.01.001. [DOI] [PubMed] [Google Scholar]
- 30.Wu L Jin Y Zhao X Tang K Zhao Y Tong L, et al. Tumor aerobic glycolysis confers immune evasion through modulating sensitivity to T cell-mediated bystander killing via TNF-α. Cell Metab 2023;35:1580–1596.e9. doi: 10.1016/j.cmet.2023.07.001. [DOI] [PubMed] [Google Scholar]
- 31.Zhang H Liu J Yang Z Zeng L Wei K Zhu L, et al. TCR activation directly stimulates PYGB-dependent glycogenolysis to fuel the early recall response in CD8+ memory T cells. Mol Cell 2022;82:3077–3088.e6. doi: 10.1016/j.molcel.2022.06.002. [DOI] [PubMed] [Google Scholar]
- 32.Lim SA, Su W, Chapman NM, Chi H. Lipid metabolism in T cell signaling and function. Nat Chem Biol 2022;18:470–481. doi: 10.1038/s41589-022-01017-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu X Hartman CL Li L Albert CJ Si F Gao A, et al. Reprogramming lipid metabolism prevents effector T cell senescence and enhances tumor immunotherapy. Sci Transl Med 2021;13:doi: 10.1126/scitranslmed.aaz6314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lochner M, Berod L, Sparwasser T. Fatty acid metabolism in the regulation of T cell function. Trends Immunol 2015;36:81–91. doi: 10.1016/j.it.2014.12.005. [DOI] [PubMed] [Google Scholar]
- 35.van der Windt GJ O’Sullivan D Everts B Huang SC Buck MD Curtis JD, et al. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc Natl Acad Sci U S A 2013;110:14336–14341. doi: 10.1073/pnas.1221740110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Endo Y, Kanno T, Nakajima T. Fatty acid metabolism in T-cell function and differentiation. Int Immunol 2022;34:579–587. doi: 10.1093/intimm/dxac025. [DOI] [PubMed] [Google Scholar]
- 37.Howie D, Ten Bokum A, Necula AS, Cobbold SP, Waldmann H. The Role of Lipid Metabolism in T Lymphocyte Differentiation and Survival. Front Immunol 2017;8:1949. doi: 10.3389/fimmu.2017.01949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Xu S Chaudhary O Rodríguez-Morales P Sun X Chen D Zappasodi R, et al. Uptake of oxidized lipids by the scavenger receptor CD36 promotes lipid peroxidation and dysfunction in CD8+ T cells in tumors. Immunity 2021;54:1561–1577.e7. doi: 10.1016/j.immuni.2021.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chen Y, Zhang J, Cui W, Silverstein RL. CD36, a signaling receptor and fatty acid transporter that regulates immune cell metabolism and fate. J Exp Med 2022;219:doi: 10.1084/jem.20211314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ma X Xiao L Liu L Ye L Su P Bi E, et al. CD36-mediated ferroptosis dampens intratumoral CD8+ T cell effector function and impairs their antitumor ability. Cell Metab 2021;33:1001–1012.e5. doi: 10.1016/j.cmet.2021.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nava Lauson CB Tiberti S Corsetto PA Conte F Tyagi P Machwirth M, et al. Linoleic acid potentiates CD8+ T cell metabolic fitness and antitumor immunity. Cell Metab 2023;35:633–650.e9. doi: 10.1016/j.cmet.2023.02.013. [DOI] [PubMed] [Google Scholar]
- 42.O’Sullivan D van der Windt G Huang SC Curtis JD Chang CH Buck MD, et al. Memory CD8+ T Cells Use Cell-Intrinsic Lipolysis to Support the Metabolic Programming Necessary for Development. Immunity 2018;49:375–376. doi: 10.1016/j.immuni.2018.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Raud B, McGuire PJ, Jones RG, Sparwasser T, Berod L. Fatty acid metabolism in CD8+ T cell memory: Challenging current concepts. Immunol Rev 2018;283:213–231. doi: 10.1111/imr.12655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang Y Kurupati R Liu L Zhou XY Zhang G Hudaihed A, et al. Enhancing CD8+ T Cell Fatty Acid Catabolism within a Metabolically Challenging Tumor Microenvironment Increases the Efficacy of Melanoma Immunotherapy. Cancer Cell 2017;32:377–391.e9. doi: 10.1016/j.ccell.2017.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ma X Bi E Lu Y Su P Huang C Liu L, et al. Cholesterol Induces CD8+ T Cell Exhaustion in the Tumor Microenvironment. Cell Metab 2019;30:143–156.e5. doi: 10.1016/j.cmet.2019.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yu M, Zhang S. Influenced tumor microenvironment and tumor immunity by amino acids. Front Immunol 2023;14:1118448. doi: 10.3389/fimmu.2023.1118448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Geiger R Rieckmann JC Wolf T Basso C Feng Y Fuhrer T, et al. L-Arginine Modulates T Cell Metabolism and Enhances Survival and Anti-tumor Activity. Cell 2016;167:829–842.e13. doi: 10.1016/j.cell.2016.09.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ye L Park JJ Peng L Yang Q Chow RD Dong MB, et al. A genome-scale gain-of-function CRISPR screen in CD8 T cells identifies proline metabolism as a means to enhance CAR-T therapy. Cell Metab 2022;34:595–614.e14. doi: 10.1016/j.cmet.2022.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ma EH Bantug G Griss T Condotta S Johnson RM Samborska B, et al. Serine Is an Essential Metabolite for Effector T Cell Expansion. Cell Metab 2017;25:345–57. doi: 10.1016/j.cmet.2016.12.011. [DOI] [PubMed] [Google Scholar]
- 50.Carr EL Kelman A Wu GS Gopaul R Senkevitch E Aghvanyan A, et al. Glutamine uptake and metabolism are coordinately regulated by ERK/MAPK during T lymphocyte activation. J Immunol 2010;185:1037–1044. doi: 10.4049/jimmunol.0903586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu J, Li G, Li L, Li D, Dong Z, Jiang P. Asparagine enhances LCK signalling to potentiate CD8+ T-cell activation and anti-tumour responses. Nat Cell Biol 2021;23:75–86. doi: 10.1038/s41556-020-00615-4. [DOI] [PubMed] [Google Scholar]
- 52.Roy DG Chen J Mamane V Ma EH Muhire BM Sheldon RD, et al. Methionine Metabolism Shapes T Helper Cell Responses through Regulation of Epigenetic Reprogramming. Cell Metab 2020;31:250–266.e9. doi: 10.1016/j.cmet.2020.01.006. [DOI] [PubMed] [Google Scholar]
- 53.Tang K Zhang H Deng J Wang D Liu S Lu S, et al. Ammonia detoxification promotes CD8+ T cell memory development by urea and citrulline cycles. Nat Immunol 2023;24:162–173. doi: 10.1038/s41590-022-01365-1. [DOI] [PubMed] [Google Scholar]
- 54.Griffiths H, Williams C, King SJ, Allison SJ. Nicotinamide adenine dinucleotide (NAD+): essential redox metabolite, co-substrate and an anti-cancer and anti-ageing therapeutic target. Biochem Soc Trans 2020;48:733–744. doi: 10.1042/BST20190033. [DOI] [PubMed] [Google Scholar]
- 55.Vigano S Alatzoglou D Irving M Ménétrier-Caux C Caux C Romero P, et al. Targeting Adenosine in Cancer Immunotherapy to Enhance T-Cell Function. Front Immunol 2019;10:925. doi: 10.3389/fimmu.2019.00925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Clever D Roychoudhuri R Constantinides MG Askenase MH Sukumar M Klebanoff CA, et al. Oxygen Sensing by T Cells Establishes an Immunologically Tolerant Metastatic Niche. Cell 2016;166:1117–1131.e14. doi: 10.1016/j.cell.2016.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Davila ML Riviere I Wang X Bartido S Park J Curran K, et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci Transl Med 2014;6:224ra25. doi: 10.1126/scitranslmed.3008226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Dixon SJ Lemberg KM Lamprecht MR Skouta R Zaitsev EM Gleason CE, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 2012;149:1060–1072. doi: 10.1016/j.cell.2012.03.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yang WS SriRamaratnam R Welsch ME Shimada K Skouta R Viswanathan VS, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell 2014;156:317–331. doi: 10.1016/j.cell.2013.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Xiong C, Ling H, Hao Q, Zhou X. Cuproptosis: p53-regulated metabolic cell death. Cell Death Differ 2023;30:876–884. doi: 10.1038/s41418-023-01125-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Eil R Vodnala SK Clever D Klebanoff CA Sukumar M Pan JH, et al. Ionic immune suppression within the tumour microenvironment limits T cell effector function. Nature 2016;537:539–543. doi: 10.1038/nature19364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vodnala SK Eil R Kishton RJ Sukumar M Yamamoto TN Ha NH, et al. T cell stemness and dysfunction in tumors are triggered by a common mechanism. Science 2019;363:doi: 10.1126/science.aau0135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Mazzoni A Bronte V Visintin A Spitzer JH Apolloni E Serafini P, et al. Myeloid suppressor lines inhibit T cell responses by an NO-dependent mechanism. J Immunol 2002;168:689–695. doi: 10.4049/jimmunol.168.2.689. [DOI] [PubMed] [Google Scholar]
- 64.Aydin E, Johansson J, Nazir FH, Hellstrand K, Martner A. Role of NOX2-Derived Reactive Oxygen Species in NK Cell-Mediated Control of Murine Melanoma Metastasis. Cancer Immunol Res 2017;5:804–811. doi: 10.1158/2326-6066.CIR-16-0382. [DOI] [PubMed] [Google Scholar]
- 65.Albelda SM. CAR T cell therapy for patients with solid tumours: key lessons to learn and unlearn. Nat Rev Clin Oncol 2024;21:47–66. doi: 10.1038/s41571-023-00832-4. [DOI] [PubMed] [Google Scholar]
- 66.Triplett TA Garrison KC Marshall N Donkor M Blazeck J Lamb C, et al. Reversal of indoleamine 2,3-dioxygenase-mediated cancer immune suppression by systemic kynurenine depletion with a therapeutic enzyme. Nat Biotechnol 2018;36:758–764. doi: 10.1038/nbt.4180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ninomiya S Narala N Huye L Yagyu S Savoldo B Dotti G, et al. Tumor indoleamine 2,3-dioxygenase (IDO) inhibits CD19-CAR T cells and is downregulated by lymphodepleting drugs. Blood 2015;125:3905–3916. doi: 10.1182/blood-2015-01-621474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Spranger S Spaapen RM Zha Y Williams J Meng Y Ha TT, et al. Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med 2013;5:200ra116. doi: 10.1126/scitranslmed.3006504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhai L Ladomersky E Lauing KL Wu M Genet M Gritsina G, et al. Infiltrating T Cells Increase IDO1 Expression in Glioblastoma and Contribute to Decreased Patient Survival. Clin Cancer Res 2017;23:6650–6660. doi: 10.1158/1078-0432.CCR-17-0120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Noonepalle SK Gu F Lee EJ Choi JH Han Q Kim J, et al. Promoter Methylation Modulates Indoleamine 2,3-Dioxygenase 1 Induction by Activated T Cells in Human Breast Cancers. Cancer Immunol Res 2017;5:330–344. doi: 10.1158/2326-6066.CIR-16-0182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sommermeyer D Hudecek M Kosasih PL Gogishvili T Maloney DG Turtle CJ, et al. Chimeric antigen receptor-modified T cells derived from defined CD8+ and CD4+ subsets confer superior antitumor reactivity in vivo. Leukemia 2016;30:492–500. doi: 10.1038/leu.2015.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Crompton JG Sukumar M Roychoudhuri R Clever D Gros A Eil RL, et al. Akt inhibition enhances expansion of potent tumor-specific lymphocytes with memory cell characteristics. Cancer Res 2015;75:296–305. doi: 10.1158/0008-5472.CAN-14-2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Araki K Turner AP Shaffer VO Gangappa S Keller SA Bachmann MF, et al. mTOR regulates memory CD8 T-cell differentiation. Nature 2009;460:108–112. doi: 10.1038/nature08155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Abu Eid R Ahmad S Lin Y Webb M Berrong Z Shrimali R, et al. Enhanced Therapeutic Efficacy and Memory of Tumor-Specific CD8 T Cells by Ex Vivo PI3K-δ Inhibition. Cancer Res 2017;77:4135–4145. doi: 10.1158/0008-5472.CAN-16-1925. [DOI] [PubMed] [Google Scholar]
- 75.Huang Q Xia J Wang L Wang X Ma X Deng Q, et al. miR-153 suppresses IDO1 expression and enhances CAR T cell immunotherapy. J Hematol Oncol 2018;11:58. doi: 10.1186/s13045-018-0600-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Shao J Hou L Liu J Liu Y Ning J Zhao Q, et al. Indoleamine 2,3-Dioxygenase 1 Inhibitor-Loaded Nanosheets Enhance CAR-T Cell Function in Esophageal Squamous Cell Carcinoma. Front Immunol 2021;12:661357. doi: 10.3389/fimmu.2021.661357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Yang Q Hao J Chi M Wang Y Xin B Huang J, et al. Superior antitumor immunotherapy efficacy of kynureninase modified CAR-T cells through targeting kynurenine metabolism. Oncoimmunology 2022;25;11:2055703. doi: 10.1080/2162402X.2022.2055703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Newick K O’Brien S Sun J Kapoor V Maceyko S Lo A, et al. Augmentation of CAR T-cell Trafficking and Antitumor Efficacy by Blocking Protein Kinase A Localization. Cancer Immunol Res 2016;4:541–551. doi: 10.1158/2326-6066.CIR-15-0263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Ma SR Deng WW Liu JF Mao L Yu GT Bu LL, et al. Blockade of adenosine A2A receptor enhances CD8+ T cells response and decreases regulatory T cells in head and neck squamous cell carcinoma. Mol Cancer 2017;16:99. doi: 10.1186/s12943-017-0665-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Young A Ngiow SF Barkauskas DS Sult E Hay C Blake SJ, et al. Co-inhibition of CD73 and A2AR Adenosine Signaling Improves Anti-tumor Immune Responses. Cancer Cell 2016;30:391–403. doi: 10.1016/j.ccell.2016.06.025. [DOI] [PubMed] [Google Scholar]
- 81.Leone RD Sun IM Oh MH Sun IH Wen J Englert J, et al. Inhibition of the adenosine A2a receptor modulates expression of T cell coinhibitory receptors and improves effector function for enhanced checkpoint blockade and ACT in murine cancer models. Cancer Immunol Immunother 2018;67:1271–1284. doi: 10.1007/s00262-018-2186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Giuffrida L Sek K Henderson MA Lai J Chen A Meyran D, et al. CRISPR/Cas9 mediated deletion of the adenosine A2A receptor enhances CAR T cell efficacy. Nat Commun 2021;12:3236. doi: 10.1038/s41467-021-23331-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Liu G Zhang Q Liu G Li D Zhang L Gu Z, et al. Disruption of adenosine 2A receptor improves the anti-tumor function of anti-mesothelin CAR T cells both in vitro and in vivo. Exp Cell Res 2021;409:112886. doi: 10.1016/j.yexcr.2021.112886. [DOI] [PubMed] [Google Scholar]
- 84.Li N Tang N Cheng C Hu T Wei X Han W, et al. Improving the anti-solid tumor efficacy of CAR-T cells by inhibiting adenosine signaling pathway. Oncoimmunology 2020;9:1824643. doi: 10.1080/2162402X.2020.1824643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Masoumi E Jafarzadeh L Mirzaei HR Alishah K Fallah-Mehrjardi K Rostamian H, et al. Genetic and pharmacological targeting of A2a receptor improves function of anti-mesothelin CAR T cells. J Exp Clin Cancer Res 2020;39:49. doi: 10.1186/s13046-020-01546-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Fallah-Mehrjardi K Mirzaei HR Masoumi E Jafarzadeh L Rostamian H Khakpoor-Koosheh M, et al. Pharmacological targeting of immune checkpoint A2aR improves function of anti-CD19 CAR T cells in vitro. Immunol Lett 2020;223:44–52. doi: 10.1016/j.imlet.2020.04.005. [DOI] [PubMed] [Google Scholar]
- 87.Beavis PA Henderson MA Giuffrida L Mills JK Sek K Cross RS, et al. Targeting the adenosine 2A receptor enhances chimeric antigen receptor T cell efficacy. J Clin Invest 2017;127:929–941. doi: 10.1172/JCI89455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Siriwon N Kim YJ Siegler E Chen X Rohrs JA Liu Y, et al. CAR-T Cells Surface-Engineered with Drug-Encapsulated Nanoparticles Can Ameliorate Intratumoral T-cell Hypofunction. Cancer Immunol Res 2018;6:812–824. doi: 10.1158/2326-6066.CIR-17-0502. [DOI] [PubMed] [Google Scholar]
- 89.Parkman R, Gelfand EW. Severe combined immunodeficiency disease, adenosine deaminase deficiency and gene therapy. Curr Opin Immunol 1991;3:547–551. doi: 10.1016/0952-7915(91)90019-w. [DOI] [PubMed] [Google Scholar]
- 90.Qu Y Dunn ZS Chen X MacMullan M Cinay G Wang HY, et al. Adenosine Deaminase 1 Overexpression Enhances the Antitumor Efficacy of Chimeric Antigen Receptor-Engineered T Cells. Hum Gene Ther 2022;33:223–236. doi: 10.1089/hum.2021.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bunse L Pusch S Bunse T Sahm F Sanghvi K Friedrich M, et al. Suppression of antitumor T cell immunity by the oncometabolite (R)-2-hydroxyglutarate. Nat Med 2018;24:1192–1203. doi: 10.1038/s41591-018-0095-6. [DOI] [PubMed] [Google Scholar]
- 92.Zhang L, Sorensen MD, Kristensen BW, Reifenberger G, McIntyre TM, Lin F. D-2-Hydroxyglutarate Is an Intercellular Mediator in IDH-Mutant Gliomas Inhibiting Complement and T Cells. Clin Cancer Res 2018;24:5381–5391. doi: 10.1158/1078-0432.CCR-17-3855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Yang Q Hao J Chi M Wang Y Li J Huang J, et al. D2HGDH-mediated D2HG catabolism enhances the anti-tumor activities of CAR-T cells in an immunosuppressive microenvironment. Mol Ther 2022;30:1188–1200. doi: 10.1016/j.ymthe.2022.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Fultang L Booth S Yogev O Martins da Costa B Tubb V Panetti S, et al. Metabolic engineering against the arginine microenvironment enhances CAR-T cell proliferation and therapeutic activity. Blood 2020;136:1155–1160. doi: 10.1182/blood.2019004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pavlova NN, Thompson CB. The Emerging Hallmarks of Cancer Metabolism. Cell Metab 2016;23:27–47. doi: 10.1016/j.cmet.2015.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science 2009;324:1029–1033. doi: 10.1126/science.1160809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ping Y, Shen C, Huang B, Zhang Y. Reprogramming T-Cell Metabolism for Better Anti-Tumor Immunity. Cells 2022;11:doi: 10.3390/cells11193103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Shi R, Tang YQ, Miao H. Metabolism in tumor microenvironment: Implications for cancer immunotherapy. MedComm (2020) 2020;1:47–68. doi: 10.1002/mco2.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Yu W Lei Q Yang L Qin G Liu S Wang D, et al. Contradictory roles of lipid metabolism in immune response within the tumor microenvironment. J Hematol Oncol 2021;14:187. doi: 10.1186/s13045-021-01200-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Heintzman DR, Fisher EL, Rathmell JC. Microenvironmental influences on T cell immunity in cancer and inflammation. Cell Mol Immunol 2022;19:316–326. doi: 10.1038/s41423-021-00833-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Cribioli E Giordano Attianese G Ginefra P Signorino-Gelo A Vuillefroy de Silly R Vannini N, et al. Enforcing GLUT3 expression in CD8+ T cells improves fitness and tumor control by promoting glucose uptake and energy storage. Front Immunol 2022;13:976628. doi: 10.3389/fimmu.2022.976628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Beckermann KE Hongo R Ye X Young K Carbonell K Healey D, et al. CD28 costimulation drives tumor-infiltrating T cell glycolysis to promote inflammation. JCI Insight 2020;5:doi: 10.1172/jci.insight.138729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Simpson IA, Dwyer D, Malide D, Moley KH, Travis A, Vannucci SJ. The facilitative glucose transporter GLUT3: 20 years of distinction. Am J Physiol Endocrinol Metab 2008;295:E242–253. doi: 10.1152/ajpendo.90388.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Fraietta JA Lacey SF Orlando EJ Pruteanu-Malinici I Gohil M Lundh S, et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 2018;24:563–571. doi: 10.1038/s41591-018-0010-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Gross G Alkadieri S Meir A Itzhaki O Aharony-Tevet Y Yosef SB, et al. Improved CAR-T cell activity associated with increased mitochondrial function primed by galactose. bioRxiv 2023;doi: 10.1101/2023.09.23.559091. [DOI] [PubMed] [Google Scholar]
- 106.Klysz DD Fowler C Malipatlolla M Stuani L Freitas KA Meier S, et al. Inosine Induces Stemness Features in CAR T cells and Enhances Potency. bioRxiv 2023;doi: 10.1101/2023.04.21.537859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Buck MD O’Sullivan D Klein Geltink RI Curtis JD Chang CH Sanin DE, et al. Mitochondrial Dynamics Controls T Cell Fate through Metabolic Programming. Cell 2016;166:63–76. doi: 10.1016/j.cell.2016.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Huang Y, Si X, Shao M, Teng X, Xiao G, Huang H. Rewiring mitochondrial metabolism to counteract exhaustion of CAR-T cells. J Hematol Oncol 2022;15:38. doi: 10.1186/s13045-022-01255-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.van Bruggen J Martens A Fraietta JA Hofland T Tonino SH Eldering E, et al. Chronic lymphocytic leukemia cells impair mitochondrial fitness in CD8+ T cells and impede CAR T-cell efficacy. Blood 2019;134:44–58. doi: 10.1182/blood.2018885863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chamoto K Chowdhury PS Kumar A Sonomura K Matsuda F Fagarasan S, et al. Mitochondrial activation chemicals synergize with surface receptor PD-1 blockade for T cell-dependent antitumor activity. Proc Natl Acad Sci U S A 2017;114:E761–761E770. doi: 10.1073/pnas.1620433114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Austin S, St-Pierre J. PGC1α and mitochondrial metabolism--emerging concepts and relevance in ageing and neurodegenerative disorders. J Cell Sci 2012;125:4963–4971. doi: 10.1242/jcs.113662. [DOI] [PubMed] [Google Scholar]
- 112.Ventura-Clapier R, Garnier A, Veksler V. Transcriptional control of mitochondrial biogenesis: the central role of PGC-1alpha. Cardiovasc Res 2008;79:208–217. doi: 10.1093/cvr/cvn098. [DOI] [PubMed] [Google Scholar]
- 113.Fernandez-Marcos PJ, Auwerx J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am J Clin Nutr 2011;93:884S–90. doi: 10.3945/ajcn.110.001917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Dumauthioz N Tschumi B Wenes M Marti B Wang H Franco F, et al. Enforced PGC-1α expression promotes CD8 T cell fitness, memory formation and antitumor immunity. Cell Mol Immunol 2021;18:1761–1771. doi: 10.1038/s41423-020-0365-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Scharping NE Menk AV Moreci RS Whetstone RD Dadey RE Watkins SC, et al. The Tumor Microenvironment Represses T Cell Mitochondrial Biogenesis to Drive Intratumoral T Cell Metabolic Insufficiency and Dysfunction. Immunity 2016;45:701–703. doi: 10.1016/j.immuni.2016.08.009. [DOI] [PubMed] [Google Scholar]
- 116.Lontos K Wang Y Joshi SK Frisch AT Watson MJ Kumar A, et al. Metabolic reprogramming via an engineered PGC-1α improves human chimeric antigen receptor T-cell therapy against solid tumors. J Immunother Cancer 2023;11:doi: 10.1136/jitc-2022-006522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Menk AV Scharping NE Rivadeneira DB Calderon MJ Watson MJ Dunstane D, et al. 4-1BB costimulation induces T cell mitochondrial function and biogenesis enabling cancer immunotherapeutic responses. J Exp Med 2018;215:1091–1100. doi: 10.1084/jem.20171068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Tian H Chai D Wang G Wang Q Sun N Jiang G, et al. Mitochondrial C1QBP is essential for T cell antitumor function by maintaining mitochondrial plasticity and metabolic fitness. Cancer Immunol Immunother 2023;72:2151–2168. doi: 10.1007/s00262-023-03407-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Farooq MA, Ajmal I, Hui X, Chen Y, Ren Y, Jiang W. β2-Adrenergic Receptor Mediated Inhibition of T Cell Function and Its Implications for CAR-T Cell Therapy. Int J Mol Sci 2023;24:doi: 10.3390/ijms241612837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Qiao G Bucsek MJ Winder NM Chen M Giridharan T Olejniczak SH, et al. β-Adrenergic signaling blocks murine CD8+ T-cell metabolic reprogramming during activation: a mechanism for immunosuppression by adrenergic stress. Cancer Immunol Immunother 2019;68:11–22. doi: 10.1007/s00262-018-2243-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Qiao G Chen M Mohammadpour H MacDonald CR Bucsek MJ Hylander BL, et al. Chronic Adrenergic Stress Contributes to Metabolic Dysfunction and an Exhausted Phenotype in T Cells in the Tumor Microenvironment. Cancer Immunol Res 2021;9:651–664. doi: 10.1158/2326-6066.CIR-20-0445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Jensen A, Carnaz Simões AM, Thor Straten P, Holmen Olofsson G. Adrenergic Signaling in Immunotherapy of Cancer: Friend or Foe. Cancers (Basel) 2021;13:doi: 10.3390/cancers13030394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Si X Shao M Teng X Huang Y Meng Y Wu L, et al. Mitochondrial isocitrate dehydrogenase impedes CAR T cell function by restraining antioxidant metabolism and histone acetylation. Cell Metab 2024;36:176–192.e10. doi: 10.1016/j.cmet.2023.12.010. [DOI] [PubMed] [Google Scholar]
- 124.Chen C Zheng H Horwitz EM Ando S Araki K Zhao P, et al. Mitochondrial metabolic flexibility is critical for CD8+ T cell antitumor immunity. Sci Adv 2023;9:eadf9522. doi: 10.1126/sciadv.adf9522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Li L Li Q Yan ZX Sheng LS Fu D Xu P, et al. Transgenic expression of IL-7 regulates CAR-T cell metabolism and enhances in vivo persistence against tumor cells. Sci Rep 2022;12:12506. doi: 10.1038/s41598-022-16616-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Cui G Staron MM Gray SM Ho PC Amezquita RA Wu J, et al. IL-7-Induced Glycerol Transport and TAG Synthesis Promotes Memory CD8+ T Cell Longevity. Cell 2015;161:750–761. doi: 10.1016/j.cell.2015.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.van der Windt GJ Everts B Chang CH Curtis JD Freitas TC Amiel E, et al. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity 2012;36:68–78. doi: 10.1016/j.immuni.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Alizadeh D Wong RA Yang X Wang D Pecoraro JR Kuo CF, et al. IL15 Enhances CAR-T Cell Antitumor Activity by Reducing mTORC1 Activity and Preserving Their Stem Cell Memory Phenotype. Cancer Immunol Res 2019;7:759–772. doi: 10.1158/2326-6066.CIR-18-0466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Guo Y Xie YQ Gao M Zhao Y Franco F Wenes M, et al. Metabolic reprogramming of terminally exhausted CD8+ T cells by IL-10 enhances anti-tumor immunity. Nat Immunol 2021;22:746–756. doi: 10.1038/s41590-021-00940-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Zhao Y Chen J Andreatta M Feng B Xie YQ Wenes M, et al. IL-10-expressing CAR T cells resist dysfunction and mediate durable clearance of solid tumors and metastases. Nat Biotechnol 2024;doi: 10.1038/s41587-023-02060-8. [DOI] [PubMed] [Google Scholar]
- 131.Kondo T Ando M Nagai N Tomisato W Srirat T Liu B, et al. The NOTCH-FOXM1 Axis Plays a Key Role in Mitochondrial Biogenesis in the Induction of Human Stem Cell Memory-like CAR-T Cells. Cancer Res 2020;80:471–483. doi: 10.1158/0008-5472.CAN-19-1196. [DOI] [PubMed] [Google Scholar]
- 132.Kondo T Morita R Okuzono Y Nakatsukasa H Sekiya T Chikuma S, et al. Notch-mediated conversion of activated T cells into stem cell memory-like T cells for adoptive immunotherapy. Nat Commun 2017;8:15338. doi: 10.1038/ncomms15338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ando M Kondo T Tomisato W Ito M Shichino S Srirat T, et al. Rejuvenating Effector/Exhausted CAR T Cells to Stem Cell Memory-Like CAR T Cells By Resting Them in the Presence of CXCL12 and the NOTCH Ligand. Cancer Res Commun 2021;1:41–55. doi: 10.1158/2767-9764.CRC-21-0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kondo T Imura Y Chikuma S Hibino S Omata-Mise S Ando M, et al. Generation and application of human induced-stem cell memory T cells for adoptive immunotherapy. Cancer Sci 2018;109:2130–2140. doi: 10.1111/cas.13648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Choi H Deng J Li S Silk T Dong L Brea EJ, et al. Pulsatile MEK Inhibition Improves Anti-tumor Immunity and T Cell Function in Murine Kras Mutant Lung Cancer. Cell Rep 2019;27:806–819.e5. doi: 10.1016/j.celrep.2019.03.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tomida A Yagyu S Nakamura K Kubo H Yamashima K Nakazawa Y, et al. Inhibition of MEK pathway enhances the antitumor efficacy of chimeric antigen receptor T cells against neuroblastoma. Cancer Sci 2021;112:4026–4036. doi: 10.1111/cas.15074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Verma V Jafarzadeh N Boi S Kundu S Jiang Z Fan Y, et al. MEK inhibition reprograms CD8+ T lymphocytes into memory stem cells with potent antitumor effects. Nat Immunol 2021;22:53–66. doi: 10.1038/s41590-020-00818-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zheng W O’Hear CE Alli R Basham JH Abdelsamed HA Palmer LE, et al. PI3K orchestration of the in vivo persistence of chimeric antigen receptor-modified T cells. Leukemia 2018;32:1157–1167. doi: 10.1038/s41375-017-0008-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Petersen CT Hassan M Morris AB Jeffery J Lee K Jagirdar N, et al. Improving T-cell expansion and function for adoptive T-cell therapy using ex vivo treatment with PI3Kδ inhibitors and VIP antagonists. Blood Adv 2018;2:210–223. doi: 10.1182/bloodadvances.2017011254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Dwyer CJ Arhontoulis DC Rangel Rivera GO Knochelmann HM Smith AS Wyatt MM, et al. Ex vivo blockade of PI3K gamma or delta signaling enhances the antitumor potency of adoptively transferred CD8+ T cells. Eur J Immunol 2020;50:1386–1399. doi: 10.1002/eji.201948455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Rangel Rivera GO Dwyer CJ Knochelmann HM Smith AS Aksoy BA Cole AC, et al. Progressively enhancing stemness of adoptively transferred T cells with PI3Kdelta blockade improves metabolism and anti-tumor immunity. Cancer Res 2024;84:69–83. doi: 10.1158/0008-5472.CAN-23-0801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Funk CR Wang S Chen KZ Waller A Sharma A Edgar CL, et al. PI3Kδ/γ inhibition promotes human CART cell epigenetic and metabolic reprogramming to enhance antitumor cytotoxicity. Blood 2022;139:523–537. doi: 10.1182/blood.2021011597. [DOI] [PMC free article] [PubMed] [Google Scholar]